ABSTRACT
More than a year has passed since the first reported case of severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) infection in the city of Wuhan in China's Hubei Province. Until now, few antiviral medications (e.g., remdesivir) or drugs that target inflammatory complications associated with SARS-CoV2 infection have been considered safe by public health authorities. By the end of November 2020, this crisis had led to >1 million deaths and revealed the high susceptibility of people with pre-existing comorbidities (e.g., obesity, diabetes, coronary heart disease, hypertension) to suffer from a severe form of the disease. Elderly people have also been found to be highly susceptible to SARS-CoV2 infection and morbidity. Gastrointestinal manifestations and gut microbial alterations observed in SARS-CoV2–infected hospitalized patients have raised awareness of the potential role of intestinal mechanisms in increasing the severity of the disease. It is therefore critically important to find alternative or complementary approaches, not only to prevent or treat the disease, but also to reduce its growing societal and economic burden. In this review, we explore potential nutritional strategies that implicate the use of polyphenols, probiotics, vitamin D, and ω-3 fatty acids with a focus on the gut microbiome, and that could lead to concrete recommendations that are easily applicable to both vulnerable people with pre-existing metabolic comorbidities and the elderly, but also to the general population.
Keywords: COVID-19, obesity, gut microbiota, nutrition, polyphenols, probiotics, vitamin D, omega-3
This review explores potential nutritional strategies that involve the use of known modulators of the gut microbiome, namely polyphenols, probiotics, vitamin D, or ω-3 PUFAs, to mitigate COVID-19 disease outcomes.
Introduction
Since December 2019, the world's population has been severely affected by the rapid spread of coronavirus disease-19 (COVID-19), an initially unknown viral infection that was detected in patients from the city of Wuhan in China's Hubei Province (1). The disease has further been shown to originate from the severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) (2), for which the genomic sequence has some similarities with the previously known SARS-CoV (∼79% sequence identity) and to a lesser extent, with the Middle East respiratory syndrome coronavirus (MERS-CoV) (∼50% sequence identity) (3). Sepsis, acute respiratory distress, pulmonary thrombosis, and acute kidney and cardiac injuries are some of the complications frequently reported in infected patients, highlighting the scope of disease severity (2). An overproduction of proinflammatory mediators such as IL-6, IL-1β, TNF-α, and monocyte chemoattractant protein 1—a response described as a “cytokine storm”—is in part responsible for the above-mentioned complications (4). The elderly population have been largely affected by this virus leading to high mortality rates (5). In the United States, where the disease has rapidly spread, 8 out of 10 deaths related to SARS-CoV2 infection are in adults aged >65 y (6). Another subgroup of the population, those with pre-existing metabolic comorbidities, have been shown to be highly susceptible to suffer from respiratory and cardiac complications associated with SARS-CoV2 infection, resulting in the need for hospitalization and intensive care, and in some cases leading to death (7, 8). In a retrospective cohort study including 191 hospitalized patients with COVID-19, 48% had pre-existing comorbidities, the most prevalent being hypertension, diabetes, and coronary heart diseases (2).
Even though the current pandemic creates a lot of uncertainty, one consensus remains among epidemiologists: “COVID-19 is here to stay” (9). Therefore, it is of utmost importance to explore complementary or alternative strategies to reduce the burden on the healthcare system in these pandemic times and in the long term. Two decades ago, it was shown that the nutritional status of the host not only affects the capacity of the immune system to dampen the viral load but also can influence the virulence of a virus (10, 11). Interestingly, selenium-deficient mice infected with influenza virus A (H3N2) exhibited severe lung pathology compared with nondeficient mice despite equal lung virus titers between groups (11). This effect has further been attributed to the mutation of the virus due to mice selenium deficiency (10). Thus, the use of nutritional approaches targeting the numerous side effects associated with the COVID-19 pandemic in the most vulnerable populations could be promising. However, the study of the direct relation between our nutritional health and SARS-CoV2 infection in patients can be challenging considering the severity of the pandemic. In vitro and animal studies provide the first degree of evidence but also have their own limits and require specific containment levels in laboratories. It is thus relevant to provide evidence of potential nutritional applications that can target the immune system and improve our health in the context of the COVID-19 pandemic in the hope to stimulate research around this topic to eventually lead to concrete solutions.
Increased Susceptibility to SARS-CoV2 Infection and Disease Severity in People with Pre-existing Metabolic Comorbidities and Elderly Individuals
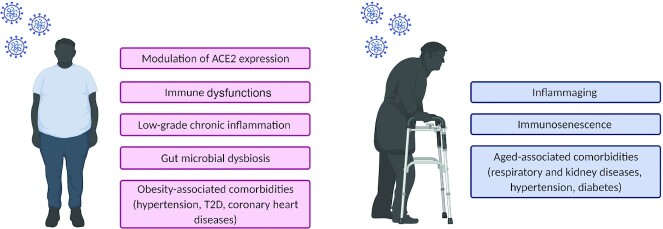
The worldwide rise of SARS-CoV2 infections has revealed the susceptibility of individuals with pre-existing metabolic comorbidities to test positive for COVID-19 (12) and to present a more severe form of the illness (13). Several hypotheses have been put forward to explain this phenomenon (Figure 1). Briefly, SARS-CoV2 enters the cells by binding to the angiotensin-converting enzyme 2 (ACE2) implicated in the renin-angiotensin system (14). The spike glycoprotein present on the outer envelope of the virus binds ≥10 times more efficiently to ACE2 than the previously known SARS-CoV, contributing to the severity of the pandemic (15). Epithelial cells lining the airway and lungs express ACE2 and appear as an easy target for virus transmission. In lung tissues of nonobese diabetic mice, a model that develops type 1 diabetes, ACE2 protein expression was significantly increased compared with nondiabetic mice (16). Insulin treatment significantly reduced the protein expression of ACE2 in the lungs of diabetic mice, suggesting that low circulating insulin concentrations, or reduced insulin action (resistance or poor glycemic control), plays a key role in modulating the susceptibility to SARS-CoV2 infection. The increased ACE2 expression in diabetic animal models has been suggested by Bindom and Lazartigues (17) to act as a compensatory mechanism to mitigate the hyperglycemic induced-activation of the ACE/Angiotensin II (Ang II)/Ang II type 1 receptor axis. The latter has been associated with vasoconstrictive and fibrogenic effects in the pancreas of healthy rats, decreasing the blood flow to the pancreatic islets of Langerhans and therefore delaying the first peak of glucose-stimulated insulin secretion (18). In individuals with obesity, it has been suggested that the upregulation of ACE2 cannot be the sole contributor to the increased susceptibility to suffer from the disease (19). Indeed, ACE2 is not only expressed in the lungs but also in the intestine (20), liver, pancreas, and heart (16) as well as both visceral and subcutaneous adipose tissues (19). Based on a human gene expression database, the expression of ACE2 is higher in visceral and subcutaneous adipose depots than in the lungs, suggesting that the high level of adiposity in people with obesity could contribute to their increased susceptibility to SARS-CoV2 infection (19). In contrast, decreased ACE2 mRNA and protein expression has been observed in SARS-CoV–infected animal models and associated with disease severity (21, 22). Downregulation of ACE2 has also been reported in a mouse model of acute lung injury induced by acid aspirations (23). The membrane fusion that permits SARS-CoV2 entry in the host cell leads to ACE2 endocytosis, contributing to the decreased expression of the membrane-bound receptor (24), the latter being implicated in antithrombotic and anti-inflammatory effects (25). As suggested by Verdecchia et al. (26), a pre-existing ACE2 deficiency in type 2 diabetic (T2D) and/or elderly people could promote a more severe form of the disease.
FIGURE 1.
Potential risk factors influencing the susceptibility to SARS-CoV2 infection and disease severity in vulnerable populations. Risk factors in individuals with obesity (pink); risk factors in elderly people (blue). Figure created with BioRender.com. ACE2, angiotensin-converting enzyme 2; SARS-CoV2, severe acute respiratory syndrome coronavirus 2; T2D, type 2 diabetes.
In obesity, innate and adaptive immune dysfunctions could represent other risk factors that can impair the antiviral response. Before December 2019, no one had ever been exposed to SARS-CoV2. Therefore, a rapid innate immune response is necessary when infected to control the viral replication (27). Rebello et al. (28) have recently suggested leptin to be a possible causal link between the dysregulated metabolic state of people with obesity and the altered immune response. Leptin is an anorexigenic hormone produced by the adipose tissue in amounts proportional to fat mass and is therefore present at high circulating concentrations in people with obesity. This hormone can regulate both the innate and adaptive immune response because many immune cells present the leptin receptor on their cell surface (29). In a mouse model of diet-induced obesity infected with the influenza A (H1N1) virus, intravenous administration of anti–mouse leptin antibody to counteract hyperleptinemia is associated with reduced pulmonary IL-6 concentrations and reduced mortality by 40%, despite similar viral titers between the experimental and control groups (30). In COVID-19 patients, a subset of monocytes correlates positively with circulating leptin concentrations. CD80, an M1 (pro-inflammatory) polarization marker, is upregulated in CD14+ monocytes of severe COVID-19 patients with high leptin concentrations as opposed to those presenting a mild form of the disease (31), suggesting hyperleptinemia to contribute to the exacerbated cytokine storm and vulnerability in individuals with obesity. A dysregulated adaptive immune response in people with obesity is also of concern because most countries rely mainly on vaccination to contain the spread of SARS-CoV2. Karlsson et al. (32) have shown impaired function of memory T cells in the lungs of diet-induced obese mice following a secondary A/H1N1 influenza infection, that was preceded by an A/H3N2 influenza infection. In vaccinated obese persons, the risk of contracting influenza or suffering from an influenza-like illness is twice as high as in healthy vaccinated subjects, an effect that could be attributed to malfunctioning T cells (33).
The evolution of the pandemic has also revealed the susceptibility of the elderly population to COVID-19–related mortality (5). In this population, the immune system undergoes marked adaptive changes due to the aged environment, a process referred to as immunosenescence (34). Therefore, the capacity of the innate and adaptive immune system to properly respond to the presence of SARS-CoV2 in the host cell might be altered. For instance, changes in the cellular metabolism of monocytes in aged individuals have been proposed as a potential link to understand the severity of COVID-19 in this population (35). In addition, the inability in older adults to compensate for the increased production of cellular debris or altered molecules by the disposal mechanisms (36) triggers inflammation, a concept also known as “inflammaging” (37). This process could exacerbate inflammatory events already associated with SARS-CoV2 infection (38). Age-associated comorbidities (e.g., respiratory and kidney diseases, hypertension, diabetes) are also independent risk factors for COVID-19 severity (39) (Figure 1).
The Potential Role of the Gut Microbiota in the Complex Pathogenesis of SARS-CoV2 Infections
The human gut microbiota represents trillions of different microorganisms (e.g., bacteria, viruses, fungi, archaea) that interact with the host to maintain metabolic homeostasis. Mounting evidence has shown the interaction of gut microbiota with the mucosal interface of the intestine through several immune mechanisms (40). However, the impact of microbes on the regulation of the host immune system is far from being restricted to the intestinal tract, but targets many different cell types within peripheral tissues. Indeed, the gut microbiome produces large numbers of microbial metabolites, in part by metabolizing nonabsorbable dietary compounds. SCFAs (the most abundant being acetate, propionate, and butyrate) have been extensively studied (41). Other metabolites such as secondary bile acids produced by the gut microbiota also contribute to the regulation of host immunity (42). Interestingly, the gut–lung axis has emerged as a novel bidirectional connection linking the gut microbiota to the airway immune system through the blood and lymphatic system (43). The recent interest shown toward the gut–lung axis is in part derived from previous studies that demonstrated increased prevalence of pulmonary diseases in patients suffering from inflammatory bowel diseases such as Crohn disease and ulcerative colitis (44–46). The airways possess their own microbiota that is similar in many ways to the mouth microbiome but not throughout its entire taxon composition, suggesting pathways of airway colonization other than the mouth microbiota (47). It has been shown that the absence of microbiota in germ-free (GF) mice was associated with innate immune cell defects in peripheral tissues. When GF mice were infected with Listeria monocytogenes, the survival rate was null compared with the specific-pathogen-free mice, which harbor a gut microbiota (48). The underlying mechanisms by which the gut microbiota can control the immune response in distant organs remain poorly understood, and the same holds true for the implication of the airway microbiome in the interaction between the gut and the lungs. However, there is some evidence pointing toward the production of gut microbial-derived compounds. Indeed, Trompette et al. (49) showed that mice fed a high-fiber diet exposed to house dust mite extract were protected against allergic inflammation in the lungs compared with mice receiving the control diet. The protective effect was attributed to increased plasma SCFA concentrations derived from the high fiber intake modulation of the gut microbiota. Thus, it is suggested from this study that SCFAs impair the capacity of dendritic cells to activate effector type 2 helper T cells responsible for the increased inflammatory response in the lungs.
In addition to the possible connection between the gut and lungs, other evidence points toward the role of the gastrointestinal (GI) tract as being a potential extrapulmonary site implicated in the infection process and the evolution of the disease severity (50). As mentioned above, ACE2 is expressed not only in the lung but also along the entire GI tract (20). The presence of SARS-CoV2 in feces of infected patients (50, 51) was also associated with alterations in the fecal microbiome (52) and mycobiome (53) during the time of hospitalization of those patients. Zuo et al. (52) reported that the baseline gut microbiome was associated with COVID-19 severity. Interestingly, Faecalibacterium prausnitzii was one of the bacteria showing the strongest inverse correlation with disease severity. This observation is in line with reduced Faecalibacterium prausnitzii abundance being reported largely in obese individuals (54). It is important to note that the studies performed by Zuo et al. (52, 53) were within a small cohort of patients, and thus conclusions from those studies should be drawn carefully. Additionally, because the gut–lung axis represents a bidirectional axis, lung inflammation could also induce gut microbial dysbiosis (55). Although more evidence is needed, it is worth considering the potential benefits of gut microbiome and immune regulators such as prebiotics and probiotics, especially in more vulnerable individuals such as people with pre-existing metabolic comorbidities or in the elderly, given their well-known predisposition for more severe COVID-19 owing to an inherently altered gut microbiome community (56–58). GI manifestations are present in SARS-CoV2–infected patients, and are positively associated with the severity of respiratory complications (59), sometimes even preceding their occurrence (60).
The Contribution of the Gut Microbiota to Chronic Low-Grade Inflammation in Assessing COVID-19 Severity
One common feature in individuals highly susceptible to suffer from a more severe form of COVID-19 is the presence of a chronic low-grade inflammatory state. A dysbiotic gut microbiota contributes to this inflammatory profile (61) and is a key feature associated with a dysregulated metabolic state (56, 62). Gut microbial dysbiosis is highly associated with disruption of the intestinal membrane integrity, which contributes to increased intestinal permeability, commonly known as leaky gut. These events can subsequently lead to the passage of bacterial endotoxins such as LPSs derived from Gram-negative bacteria through the intestinal membrane. This process has been described as metabolic endotoxemia (61). When in circulation, LPS binds to the polysaccharide-binding protein (LBP) and is transferred to membrane-bound or soluble CD14 receptor. The LPS from the CD14 receptor is then transferred to the Toll-like receptor 4 (TLR4)/Myeloid differentiation factor 2 complex on the surface of various cells, which activates downstream target genes (e.g., NF-κB) triggering inflammatory responses (63) via the myeloid differentiation primary response 88 or the TIR-domain–containing adapter-inducing interferon-β (TRIF)-dependent pathways (64). It has been reported that TLR4 signaling in a TRIF-dependent manner could determine the susceptibility to acute lung injury. Indeed, genetic deletion of TLR4 in a mouse model improved edema and histological pulmonary changes associated with acute lung injuries (65). Because TLR4 is a sensor for LPS, one could speculate that the gut microbiota contributes to COVID-19 severity in a TLR4 signaling–dependent manner. Moreover, Hoel et al. (66) have observed increased baseline LBP concentrations in the circulation of patients confirmed positive for SARS-CoV2 compared with healthy controls and, more interestingly, in infected patients with adverse cardiac outcomes compared with those who did not experience such complications (66). Thus, the presence of a low-grade chronic inflammatory state in individuals with a dysregulated metabolic state, which is at least partly explained by gut dysbiosis, could increase the risk of greater COVID-19 severity and morbidity (67). As suggested by Onishi et al. (68), it would also be interesting to further investigate the implication of microbial-derived endotoxins in determining the severity of the cytokine storm in people with pre-existing metabolic comorbidities. Recent work from our group showed that bacteria or microbial components can cross the intestinal barrier in morbidly obese individuals (69). This work showed that a specific bacterial signature was found in extraintestinal tissues like the liver, the adipose tissues, and the plasma of people with morbid obesity. More interestingly, the bacterial signature of the mesenteric adipose tissue of normoglycemic individuals was significantly discriminated from individuals with T2D, notably by the increased abundance of Faecalibacterium in the former (69). This is particularly interesting because this bacterium was reported to be associated with a lean phenotype by several research groups (54, 70). Because T2D represents a major risk factor for COVID-19 severity, one could therefore speculate that the types of bacteria or bacterial fragments present in extraintestinal tissues could be a key factor influencing the susceptibility of people to develop more severe complications.
Additional Evidence Suggesting the Use of Nutritional Applications Targeting the Gut Microbiome to Promote Health in the Context of COVID-19
From another perspective, COVID-19 has forced people to quarantine, isolating them from their usual social environment. Many individuals have lost their jobs generating additional stress, which can lead to increased risk of suffering from more profound disorders such as depression and anxiety (71). Wang et al. (72) reported that ≤16.5% and ≤28.8% of the general population experienced moderate to severe depressive and anxiety symptoms, respectively. Increasing evidence suggests that change in the composition and functions of the gut microbiome could be implicated in the onset of mood disorders through a bidirectional gut–brain axis (73, 74). Gut-derived microbial metabolites could play a pivotal role in connecting these 2 organs. Neuroactive compounds (75, 76) and SCFAs (77) produced by bacteria or intestinal cells are some documented examples of gut-derived metabolites that have been associated with brain modulation, reinforcing the use of nutritional approaches that can target the gut microbiota.
Potential Nutritional Approaches Targeting the Gut Microbiota to Alleviate SARS-CoV2 Outcomes
Polyphenols
The potential for plant-derived molecules to treat diseases takes its root in the fact that 2 of the most prescribed drugs in modern medicine worldwide, namely metformin and aspirin, were first derived from extracts of French lilac and willow bark, respectively (78, 79). Polyphenols are biologically active compounds that are abundant in berries, nuts, fruits, vegetables, and whole grains and are part of a healthy lifestyle diet (80). Polyphenols are thought to exert some of their benefits through so-called antioxidant and anti-inflammatory activities. Indeed, the molecular structure of polyphenols makes them good electron or hydrogen atom donors, which can thereby neutralize reactive oxygen species and prevent cellular damage (81). However, in the specific context of SARS-CoV2 infection, the molecular structure of polyphenols could confer them with other novel properties. Polyphenols could act as natural inhibitors of SARS-CoV2 viral activity, as suggested from studies that were performed in the context of the 2003 SARS-CoV epidemic and which reported that some polyphenols (i.e., luteolin) could bind with high affinity to the surface spike glycoprotein of the virus and restrict its entry into host cells, as demonstrated using frontal affinity chromatography coupled with MS detection (82). In addition, an in silico study has proposed that anthocyanidins (i.e., petunidin, delphinidin) and flavonols (i.e., quercetin) could act as antagonists to block ACE2 receptors to prevent the virus entry (83).
In addition to their purported ability to prevent viral entry, polyphenols such as resveratrol could prevent severe COVID-19 as suggested by Horne et al. (84). As previously mentioned, the expression of ACE2 is decreased following SARS-CoV infection and acute lung injuries. However, the administration of resveratrol was shown to increase ACE2 expression levels in the liver of rat offspring born from a high-fat (HF)–fed dam and subsequently fed an HF diet (85), and in the epididymal adipose tissue of HF-fed mice (86) at a dose of 50 and 30 mg/kg/day, respectively. On the other hand, the angiotensin-converting enzyme (ACE), which has opposite functions to ACE2 in the regulation of the renin-angiotensin system, has been shown to promote the disease severity of acute lung injuries (23). Guerrero et al. (87) reported that a concentration of 500 μM of different subtypes of flavonoids could inhibit ACE activity ranging from 17% to 95% efficacy in purified lung ACE from rabbits. Polyphenols could also decrease the disease severity by inhibiting virus replication. High concentrations (150–250 μM) of resveratrol in vitro were shown to reduce MERS-CoV RNA abundance in infected Vero E6 cells (88). One of the proposed mechanisms for this inhibition by polyphenols is their potential actions against specific coronavirus proteases such as 3-chymotrypsin-like protease (3CLPro) (89) and papain-like protease (PLPro) (90), which are implicated in virus replication. 3CLPro and PLPro are present in different types of coronavirus (91), suggesting that previous reported data on the potential inhibitory effects of polyphenols on SARS-CoV and MERS-CoV could apply also to SARS. Viral proteases have also been highly targeted for the development of drugs against coronaviruses (92, 93). Although in vitro experiments can provide interesting insights about the beneficial effects of polyphenols, they also have their limits. The concentration of polyphenols used for cellular experiments are often too high and do not take into consideration that native compounds are poorly absorbed in the gut and strongly metabolized by the gut microbiota. More than 90% of ingested polyphenols are not absorbed in the upper part of the intestine due to their high molecular weight (94). The relative abundance and biological activity of circulatory metabolites derived from the interaction of dietary polyphenols with the gut microbiota likely differ from those of the native compounds used for in vitro studies (95). Moreover, the presence of a dysregulated gut microbiota in COVID-19 patients is likely to impact on the metabolism of polyphenols, their absorption, and thus the availability of both the native molecules and their metabolites. Further experiments are needed to better understand the role of polyphenols in potentially preventing SARS-CoV2 entry into host cells and to reduce disease severity. At the time of writing this review, only 3 clinical studies evaluating the potential benefits of dietary polyphenols against SARS-CoV2 infections were registered in the US National Library of Medicine (96).
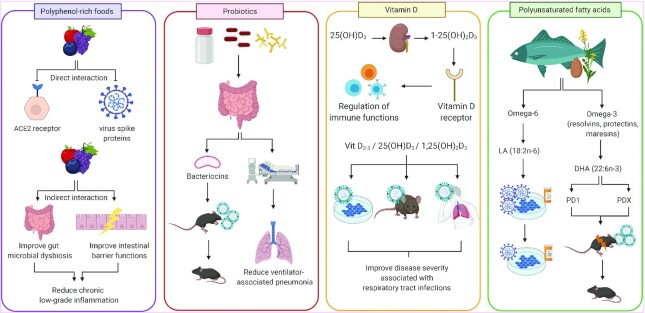
In addition to the direct effect of polyphenols on viral infection, their purported intestinal and peripheral anti-inflammatory activities are also worth mentioning (97, 98). When they have reached the colon, polyphenols exert prebiotic effects, because they are powerful modulators of the gut microbiota leading to several intestinal health benefits such as improvement of the intestinal barrier functions (99, 100). Polyphenols stimulate the growth of beneficial bacterial species such as Akkermansia muciniphila and Barnesiella as shown by our research group (101, 102) and others (103, 104). The latter have been associated with the improvement of many features of the metabolic syndrome (e.g., improved glucose tolerance, decreased visceral adiposity, reduced circulating triglyceride concentrations). As proposed above, a dysregulated gut homeostasis could contribute to the severity of COVID-19. Furthermore, when colonic bacteria metabolize polyphenols, smaller compounds are released in the circulation and reach peripheral organs to modulate host immunity (105). In the context of the pandemic, the potential of plant polyphenols has certainly not been fully explored (Table 1, Figure 2).
TABLE 1.
Summary of evidence suggesting the use of nutrients to prevent or decrease SARS-CoV2 disease severity1
| Nutrients | Evidence | References |
|---|---|---|
| Polyphenols | Natural inhibitors of SARS-CoV2 activity by binding with high affinity to the surface spike proteins | (82, 83) |
| Prevent illness severity by increasing ACE2 expression and inhibiting ACE expression | (85–87) | |
| Inhibit virus replication by their actions on specific coronavirus proteases (3CLPro and PLPro) | (88–90) | |
| Indirect evidence through their intestinal and peripheral anti-inflammatory and antioxidant effects | (101–104) | |
| Probiotics | Reduce disease incidence of ventilator associated-pneumonia | (106, 107) |
| Production of antiviral inhibitory metabolites; bacteriocins | (108, 109) | |
| Immunomodulatory effects against respiratory tract influenza viruses | (110–112) | |
| Vitamin D | Regulates immune responses against respiratory tract viruses through their receptors on immune cells | (113–117) |
| Improves disease severity associated with SARS-CoV2 | (118, 119) | |
| PUFAs | Reduce replication of SARS-CoV2 in combination with remdesivir | (120) |
| Improve disease severity associated with respiratory tract viruses and acute inflammation | (121–123) | |
| Glucoregulatory activities | (124) | |
| Anticoagulant properties | (125, 126) |
1ACE2, angiotensin-converting enzyme 2; PLPro, papain-like protease; SARS-CoV, severe acute respiratory syndrome coronavirus; 3CLPro, chymotrypsin-like protease,.
FIGURE 2.
Evidence suggesting the use of nutritional applications to alleviate SARS-CoV2 infection and adverse outcomes. Polyphenol-rich foods: Polyphenols are suggested to have direct inhibitory effects on virus entry in host cells through the interaction with ACE2 receptors and spike proteins. Polyphenols can indirectly reduce low-grade inflammation by improving gut microbial dysbiosis and intestinal membrane functions. Probiotics: The production of bacteriocins by probiotics improves mortality rate in influenza-infected mice. A prophylactic administration of probiotics has been associated with reduced ventilator-associated pneumonia in patients at high risk of developing this complication. Vitamin D: In circulation, 25(OH)D3 is converted by the kidneys to its active form, 1,25(OH)2D3, which binds to the vitamin D receptor and regulates essential immune functions. In vitro, in vivo, and clinical studies have shown the beneficial effects of vitamin D supplementation against the disease severity of respiratory tract infection. Polyunsaturated fatty acids: Resolvins, protectins, and maresins are ω-3 fatty acid–derived proresolving mediators. LA and DHA are n–6 and n–3 PUFAs, respectively. In vitro, LA reduces SARS-CoV2 replication in combination with remdesivir. Protectins (PD1, PDX) derived from the enzymatic conversion of DHA have been shown to reduce mortality rates in sepsis and influenza-infected mouse models. Figure created with BioRender.com. ACE2, angiotensin-converting enzyme 2; LA, linoleic acid; PD1, protectin D1; PDX, protectin DX; SARS-CoV, severe acute respiratory syndrome coronavirus; Vit D2-3, Vitamin D2 and D3; 1,25(OH)2D3, 1,25-dihydroxycholecalciferol; 25(OH)D3, 25-hydroxycholecalciferol.
Probiotics
Probiotics have been defined as “live microorganisms that, when administered in adequate amounts, confer a health benefit on the host” (127). Many commercial probiotics are specific strains of Lactobacillus and Bifidobacterium (127). In prospective randomized control trials, a prophylactic administration of Lactobacillus rhamnosus GG (106) and a combination of Bacillus subtilis and Enterococcus faecalis (107) in patients at high risk of developing ventilator-associated pneumonia significantly reduced disease incidence compared with patients receiving placebo. Considering that hospitalized patients with pre-existing metabolic comorbidities are more likely to need mechanical ventilation (128), the use of probiotics to help reduce respiratory complications should be further explored in the context of this pandemic (Figure 2). A direct virus–probiotic interaction has been proposed to explain the antiviral potential of probiotics (129, 130), but robust evidence supporting this interaction is lacking. As suggested by Mak et al. (131), it is doubtful that a direct probiotic–SARS-CoV2 interaction occurs. On the other hand, an indirect interaction of a probiotic with SARS-CoV2 through the production of antiviral inhibitory metabolites or immunomodulatory effects is a more likely mechanism. In a recent review, Tiwari et al. (132) suggested using bacteriocins for their potential antiviral benefits as a new therapeutic agent. These are polypeptides produced by probiotics such as lactic acid bacteria. The role of bacteriocins goes beyond their known function as natural food preservatives (133). However, scant attention has been given to their antiviral effects in the context of COVID-19. In a previous in vitro study, semipurified bacteriocins from Enterococcus durans inhibited herpes simplex virus 1 and poliovirus replication at different levels in Vero cells (108). Mice infected with the influenza virus A (H1N1) receiving a probiotic strain (Enterococcus faecium L3) known to produce specific bacteriocins starting 1 wk prior to infection showed a decreased mortality rate compared with the nontreated control group (Figure 2) (109). As for the immunomodulatory capacities of probiotics, commensal bacteria have been shown to regulate the innate and adaptive immune response against respiratory tract influenza A/H1N1 infections (110–112). Indeed, antibiotic-treated mice showed decreased influenza-specific antibody titers and CD4+ and CD8+ T cell responses (110). The administration of a probiotic cocktail in antibiotic-treated mice infected with influenza virus showed improved lung pathology (111). In BALB/c mice challenged with A/H1N1 influenza virus, the administration of Bidifobacterium bifidum prophylactically significantly reduced IL-6 concentration in lung homogenates and increased the survival rate of the mice (112). Based on those preclinical studies, the development of potential probiotic applications should be considered knowing that the commensal gut microbiota composition is altered in people with obesity (56) and in elderly individuals (57). However, evidence supporting their use to prevent or reduce the severity of COVID-19 is currently lacking (Table 1).
Additional Nutritional Approaches to Improve Health in the Context of COVID-19
Vitamin D
Cholecalciferol (vitamin D-3), a fat-soluble vitamin, is naturally synthesized from 7-dehydrocholesterol in the skin when exposed to ultraviolet-B. It is then metabolized in the liver to 25-hydroxycholecalciferol [25(OH)D3] and subsequently by the kidneys to its active form, 1,25-dihydroxycholecalciferol [1,25(OH)2D3] (134). Therefore, in countries with diminished sunlight exposure due to geographic location and cold weather, vitamin D supplementation can be recommended for some groups of the population to achieve the recommended nutritional intake. Indeed, natural dietary sources such as fatty fish or egg yolks can only provide a minimal amount of vitamin D (135). In some countries, systematic vitamin D food fortification is also mandatory (136). Even though these measures are in place and UV radiation is considered enough to avoid deficiency in countries with continuous sunlight exposure, vitamin D insufficiency/deficiency is still a public health issue worldwide (137). Compared with healthy individuals, people with various comorbidities such as T2D (138) and obesity (139) have a greater prevalence of vitamin D deficiency. However, the reason for this discrepancy is not yet well understood. It must be clarified whether a low 25(OH)D3 status is a cause or a consequence of a dysregulated metabolic state, as proposed by Vranić et al. (140). A large proportion of the elderly population are also deficient in vitamin D (141).
Vitamin D acts as an important player in the regulation of metabolism that goes beyond its function in calcium homoeostasis (142). 1,25(OH)2D3 binds to the vitamin D receptor, a nuclear receptor that acts as a transcription factor, implicated in the regulation of essential immune functions (143). Interestingly, in vitro evidence has suggested a protective role of vitamin D in regulating the immune response of respiratory viruses in human alveolar and bronchial epithelial cell lines (113–115). Mice supplemented with 25(OH)D3 in their diet for 7 wk showed reduced lung viral replication 3 d post-A/H1N1influenza viral injection. This effect was, however, transient and no longer statistically significant 5 d postinfection (116). In humans, a meta-analysis including 10,899 subjects suggested protective effects of vitamin D against the risk of acute respiratory tract infection (117). Knowing that individuals with metabolic comorbidities and older people are more prone to suffer from vitamin D deficiency, it is noteworthy that patients who were the most vitamin D deficient were observed to benefit the most from a daily or weekly supplementation (117). In the context of COVID-19, an observational study and a clinical case report have shown beneficial effects of vitamin D supplementation against the progression of the disease severity, such as lower oxygen requirement and reduced support from the intensive care unit (ICU), in hospitalized patients (118, 119). Such studies were, however, performed in small cohorts of patients. Overall, robust evidence for the potential therapeutic use of vitamin D against SARS-CoV2 infection and disease severity is still lacking (Table 1, Figure 2). An important international effort is currently underway to assess these effects. Indeed, many clinical studies are listed by the US National Library of Medicine to test vitamin D products in the context of COVID-19 (96).
Omega-3 fatty acids and their lipid mediators
The health benefits of PUFAs have been largely demonstrated and are thought to be partly related to their anti-inflammatory actions (144). Recent results point toward the use of PUFAs in the treatment of COVID-19 (Table 1, Figure 2). The administration of 50 μM linoleic acid (18:2n–6) in combination with remdesivir (a broad-spectrum antiviral agent) in human epithelial cells has been shown to reduce the replication of SARS-CoV2 greater than the administration of remdesivir alone (120). These results are highly significant considering that remdesivir has been authorized in many countries for the treatment of patients with symptoms of COVID-19 (145). Moreover, long-chain ω-3 PUFAs, such as EPA (20:5n–3) and DHA (22:6n–3), can be synthesized by internal bioconversion of their precursor, α-linolenic acid (ALA; 18:3n–3). However, the bioconversion of ALA to EPA and DHA in humans is limited and so an adequate dietary intake of rich food sources such fish oil, nuts, and seeds (flax, chia, canola) is necessary to achieve recommendations (146). Protectins are lipid mediators derived from DHA and are naturally produced in mammals through enzymatic production (147). They are known to promote resolution of inflammatory processes (148). The administration of the protectin D1 (PD1) isomer [known as protectin DX (PDX)] at a dose of 1000 ng combined with an antiviral drug (peramivir) in A/H1N1 influenza–infected mice 48 h postinfection prevented mortality (100% survival rate) compared with administration of the antiviral drug or PDX alone, which was associated with only 25–30% survival rates (121). Moreover, the administration of high doses of PDX (300 and 1000 ng) has been shown to increase survival rate in a mouse model of sepsis (122). In mice suffering from LPS-induced acute lung injury, the administration of low doses of PDX (1 and 10 ng compared with 100 ng) protected against pulmonary histopathological changes, facilitating the resolution of inflammation and inhibiting neutrophil infiltration (123). Other unique bioactivities of PDX over those of PD1 might also contribute to its ability to reduce COVID-19 illness and its most severe complications. Indeed, we have shown that PDX, but not PD1, has unique glucoregulatory activities and reduces insulin resistance in an obese diabetic mouse model (124). Finally, PDX has a well-recognized anticoagulant activity that is also distinct from PD1 (125, 126), a key attribute when considering the high incidence of thrombotic events in ICU patients with COVID-19 pneumonia (149). These properties, coupled with its antidiabetic effect, point toward the development of an innovative treatment for COVID-19, particularly in more vulnerable diabetic patients, targeting both acute inflammation and hematological complications caused by the viral infection. Historically, the pharmaceutical development of PDX has been hampered by its exorbitant cost of production linked to an inefficient biosynthetic manufacturing method. However, we have recently developed an efficient and complete chemical synthesis of PDX that will allow the synthesis of large quantities of this unique proresolving and metabolically active DHA derivative at reduced cost (150). Other EPA- and DHA-derived proresolving mediators (e.g., resolvins, maresins) have also been suggested for their potential antiviral activities (151) and also need to be investigated for their potential actions against COVID-19 and associated pathologies.
Conclusion
This review presents an overview of potential nutritional approaches that could help limit SARS-CoV2 infection and related pathological outcomes, particularly in patients with pre-existing metabolic comorbidities and in the elderly population. Indeed, many preclinical and clinical studies have suggested the immunomodulatory and/or anti-inflammatory effects of polyphenols, probiotics, vitamin D, and PUFAs, in part through gut microbiota modulation. Even if some of these strategies appear promising to prevent COVID-19 or to limit its severity, the main issue is the lack of well-designed and large-scale clinical studies for us to be able to draw final conclusions and eventually formulate public health recommendations. When writing this review, most of the clinical trials assessing the impact of some of these nutritional strategies have not yet started the recruitment process or are still in the process of enrolling patients. None of these potential applications to improve health in the context of COVID-19 or to treat the disease have yet been approved by public health authorities. Therefore, claims made for the potential of food sources to prevent or attenuate COVID-19 severity should be carefully stated due to the lack of extensive literature and potential adverse effects. One notable example is the proposed use of relatively high doses of vitamin D to protect people from COVID-19, but which can also lead to secondary vascular calcification, a disorder of the blood vessels characterized by a deposit of calcium along the vessel walls (152). Nausea, flatulence, and abdominal cramping are possible mild side effects of probiotic consumption, but they are generally recognized as safe (GRAS) even if overconsumed. However, the intake of probiotics should be supervised by a healthcare provider in immunosuppressed patients, pregnant women, or patients with heart disease, for instance (153, 154). In a similar way, some adverse outcomes associated with a high polyphenol consumption in both animal models and humans (155) and some conflicting evidence associated with the intake of PUFAs and the risk of prostate cancer (156, 157) have been reported. Finally, nutrition and infectious diseases are not typical cross-disciplines but this should be further encouraged considering the growing occurrence of epidemics (e.g., SARS-CoV, MERS-CoV, Ebola, Zika virus) and a pandemic (SARS-CoV2) over the last 20 y.
ACKNOWLEDGEMENTS
We thank Patricia Mitchell for her editorial help with the manuscript.
The authors’ responsibilities were as follows—LD, GP, AM: designed the content of this review; LD: drafted the manuscript; GP, AM: revised the manuscript; and all authors: read and approved the final manuscript.
Notes
AM is funded by a CIHR/Pfizer Research Chair in the pathogenesis of insulin resistance and cardiovascular diseases, and by Sentinel North Program funded by the Canada First Research Excellence Fund. LD is funded by the Jean-Paul Houle fund from Université Laval.
Author disclosures: The authors report no conflicts of interest.
Perspective articles allow authors to take a position on a topic of current major importance or controversy in the field of nutrition. As such, these articles could include statements based on author opinions or point of view. Opinions expressed in Perspective articles are those of the author and are not attributable to the funder(s) or the sponsor(s) or the publisher, Editor, or Editorial Board of Advances in Nutrition. Individuals with different positions on the topic of a Perspective are invited to submit their comments in the form of a Perspectives article or in a Letter to the Editor.
Abbreviations used: ACE, angiotensin-converting enzyme; ALA, α-linolenic acid; Ang II, angiotensin II; COVID-19, coronavirus disease-19; GF, germ-free; GI, gastrointestinal; H1N1, influenza A virus subtype; HF, high-fat; ICU, intensive care unit; LBP, lipopolysaccharide-binding protein; MERS-CoV, Middle East respiratory syndrome coronavirus; PD1, protectin D1; PDX, protectin DX; PLPro, papain-like protease; SARS-CoV, severe acute respiratory syndrome coronavirus; T2D, type 2 diabetes; TLR4, Toll-like receptor 4; TRIF, TIR domain–containing adapter-inducing interferon-β; 1,25(OH)2D3, 1,25-dihydroxycholecalciferol; 3CLPro, chymotrypsin-like protease; 25(OH)D3, 25-hydroxycholecalciferol.
Contributor Information
Laurence Daoust, Quebec Heart and Lung Institute, Laval University, Quebec City, Quebec, Canada; Institute of Nutrition and Functional Foods, Laval University, Quebec City, Quebec, Canada.
Geneviève Pilon, Quebec Heart and Lung Institute, Laval University, Quebec City, Quebec, Canada; Institute of Nutrition and Functional Foods, Laval University, Quebec City, Quebec, Canada.
André Marette, Quebec Heart and Lung Institute, Laval University, Quebec City, Quebec, Canada; Institute of Nutrition and Functional Foods, Laval University, Quebec City, Quebec, Canada.
References
- 1. Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu Ret al. . A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382(8):727–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu Xet al. . Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395(10229):1054–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lu R, Zhao X, Li J, Niu P, Yang B, Wu H, Wang W, Song H, Huang B, Zhu Net al. . Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395(10224):565–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Ragab D, Salah Eldin H, Taeimah M, Khattab R, Salem R. The COVID-19 cytokine storm; what we know so far. Front Immunol. 2020;11:1446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Wang W, Tang J, Wei F. Updated understanding of the outbreak of 2019 novel coronavirus (2019-nCoV) in Wuhan. J Med Virol. 2020;92(4):441–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Centers for Disease Control and Prevention.. Coronavirus disease 2019 (COVID-19). Older adults. [Internet]. U.S. Department of Health & Human Services; 2020. Updated September 11, 2020 [cited November 23, 2020]. Available from: https://www.cdc.gov/coronavirus/2019-ncov/need-extra-precautions/older-adults.html [Google Scholar]
- 7. Petrilli CM, Jones SA, Yang J, Rajagopalan H, O'Donnell L, Chernyak Y, Tobin KA, Cerfolio RJ, Francois F, Horwitz LI. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Samidurai A, Das A. Cardiovascular complications associated with COVID-19 and potential therapeutic strategies. Int J Mol Sci. 2020;21(18):6790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Scudellari M. How the pandemic might play out in 2021 and beyond. Nature. 2020;584(7819):22–5. [DOI] [PubMed] [Google Scholar]
- 10. Nelson HK, Shi Q, Van Dael P, Schiffrin EJ, Blum S, Barclay D, Levander OA, Beck MA. Host nutritional selenium status as a driving force for influenza virus mutations. FASEB J. 2001;15(10):1727–38. [PubMed] [Google Scholar]
- 11. Beck MA, Nelson HK, Shi Q, Van Dael P, Schiffrin EJ, Blum S, Barclay D, Levander OA. Selenium deficiency increases the pathology of an influenza virus infection. FASEB J. 2001;15(8):1481–3. [PubMed] [Google Scholar]
- 12. de Lusignan S, Dorward J, Correa A, Jones N, Akinyemi O, Amirthalingam G, Andrews N, Byford R, Dabrera G, Elliot Aet al. . Risk factors for SARS-CoV-2 among patients in the Oxford Royal College of General Practitioners Research and Surveillance Centre primary care network: a cross-sectional study. Lancet Infect Dis. 2020;20(9):1034–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Atkins JL, Masoli JAH, Delgado J, Pilling LC, Kuo CL, Kuchel GA, Melzer D. Preexisting comorbidities predicting COVID-19 and mortality in the UK Biobank Community Cohort. J Gerontol A Biol Sci Med Sci. 2020;75(11):2224–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, Schiergens TS, Herrler G, Wu NH, Nitsche Aet al. . SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181(2):271–80..e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Wrapp D, Wang N, Corbett KS, Goldsmith JA, Hsieh CL, Abiona O, Graham BS, McLellan JS. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020;367(6483):1260–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Roca-Ho H, Riera M, Palau V, Pascual J, Soler MJ. Characterization of ACE and ACE2 expression within different organs of the NOD mouse. Int J Mol Sci. 2017;18(3):563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bindom SM, Lazartigues E. The sweeter side of ACE2: physiological evidence for a role in diabetes. Mol Cell Endocrinol. 2009;302(2):193–202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Carlsson PO, Berne C, Jansson L. Angiotensin II and the endocrine pancreas: effects on islet blood flow and insulin secretion in rats. Diabetologia. 1998;41(2):127–33. [DOI] [PubMed] [Google Scholar]
- 19. Al-Benna S. Association of high level gene expression of ACE2 in adipose tissue with mortality of COVID-19 infection in obese patients. Obes Med. 2020;19:100283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Zhang H, Kang Z, Gong H, Xu D, Wang J, Li Z, Li Z, Cui X, Xiao J, Zhan Jet al. . Digestive system is a potential route of COVID-19: an analysis of single-cell coexpression pattern of key proteins in viral entry process. Gut. 2020;69(6):1010–18. [Google Scholar]
- 21. Oudit GY, Kassiri Z, Jiang C, Liu PP, Poutanen SM, Penninger JM, Butany J. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur J Clin Invest. 2009;39(7):618–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng Wet al. . A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med. 2005;11(8):875–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Imai Y, Kuba K, Rao S, Huan Y, Guo F, Guan B, Yang P, Sarao R, Wada T, Leong-Poi Het al. . Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436(7047):112–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Gheblawi M, Wang K, Viveiros A, Nguyen Q, Zhong JC, Turner AJ, Raizada MK, Grant MB, Oudit GY. Angiotensin-converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system: celebrating the 20th anniversary of the discovery of ACE2. Circ Res. 2020;126(10):1456–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Santos RAS, Sampaio WO, Alzamora AC, Motta-Santos D, Alenina N, Bader M, Campagnole-Santos MJ. The ACE2/angiotensin-(1-7)/MAS axis of the renin-angiotensin system: focus on angiotensin-(1-7). Physiol Rev. 2018;98(1):505–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Verdecchia P, Cavallini C, Spanevello A, Angeli F. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur J Int Med. 2020;76:14–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Catanzaro M, Fagiani F, Racchi M, Corsini E, Govoni S, Lanni C. Immune response in COVID-19: addressing a pharmacological challenge by targeting pathways triggered by SARS-CoV-2. Sig Transduct Target Ther. 2020;5(1):84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Rebello CJ, Kirwan JP, Greenway FL. Obesity, the most common comorbidity in SARS-CoV-2: is leptin the link?. Int J Obes. 2020;44(9):1810–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. La Cava A, Matarese G. The weight of leptin in immunity. Nat Rev Immunol. 2004;4(5):371–9. [DOI] [PubMed] [Google Scholar]
- 30. Zhang AJ, To KK, Li C, Lau CC, Poon VK, Chan CC, Zheng BJ, Hung IF, Lam KS, Xu Aet al. . Leptin mediates the pathogenesis of severe 2009 pandemic influenza A(H1N1) infection associated with cytokine dysregulation in mice with diet-induced obesity. J Infect Dis. 2013;207(8):1270–80. [DOI] [PubMed] [Google Scholar]
- 31. Wang J, Xu Y, Zhang X, Wang S, Peng Z, Guo J, Jiang H, Liu J, Xie Y, Wang Jet al. . Leptin correlates with monocytes activation and severe condition in COVID-19 patients. J Leukoc Biol. [Internet]2021. doi:10.1002/JLB.5HI1020-704R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Karlsson EA, Sheridan PA, Beck MA. Diet-induced obesity impairs the T cell memory response to influenza virus infection. J Immunol. 2010;184(6):3127–33. [DOI] [PubMed] [Google Scholar]
- 33. Neidich SD, Green WD, Rebeles J, Karlsson EA, Schultz-Cherry S, Noah TL, Chakladar S, Hudgens MG, Weir SS, Beck MA. Increased risk of influenza among vaccinated adults who are obese. Int J Obes. 2017;41(9):1324–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Cunha LL, Perazzio SF, Azzi J, Cravedi P, Riella LV. Remodeling of the immune response with aging: immunosenescence and its potential impact on COVID-19 immune response. Front Immunol. 2020;11:1748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Pence BD. Severe COVID-19 and aging: are monocytes the key?. Geroscience. 2020;42(4):1051–61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Ferrucci L, Fabbri E. Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol. 2018;15(9):505–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Franceschi C, Bonafè M, Valensin S, Olivieri F, De Luca M, Ottaviani E, De Benedictis G. Inflamm-aging. An evolutionary perspective on immunosenescence. Ann N Y Acad Sci. 2006;908:244–54. [DOI] [PubMed] [Google Scholar]
- 38. Akbar AN, Gilroy DW. Aging immunity may exacerbate COVID-19. Science. 2020;369(6501):256–7. [DOI] [PubMed] [Google Scholar]
- 39. Mueller AL, McNamara MS, Sinclair DA. Why does COVID-19 disproportionately affect older people?. Aging. 2020;12(10):9959–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Zheng D, Liwinski T, Elinav E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020;30(6):492–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Ratajczak W, Rył A, Mizerski A, Walczakiewicz K, Sipak O, Laszczyńska M. Immunomodulatory potential of gut microbiome-derived short-chain fatty acids (SCFAs). Acta Biochim Pol. 2019;66(1):1–12. [DOI] [PubMed] [Google Scholar]
- 42. Campbell C, McKenney PT, Konstantinovsky D, Isaeva OI, Schizas M, Verter J, Mai C, Jin WB, Guo CJ, Violante Set al. . Bacterial metabolism of bile acids promotes generation of peripheral regulatory T cells. Nature. 2020;581(7809):475–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Anand S, Mande SS. Diet, microbiota and gut-lung connection. Front Microbiol. 2018;9:2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Yazar A, Atis S, Konca K, Pata C, Akbay E, Calikoglu M, Hafta A. Respiratory symptoms and pulmonary functional changes in patients with irritable bowel syndrome. Am J Gastroenterol. 2001;96(5):1511–16. [DOI] [PubMed] [Google Scholar]
- 45. Rothfuss KS, Stange EF, Herrlinger KR. Extraintestinal manifestations and complications in inflammatory bowel diseases. World J Gastroenterol. 2006;12(30):4819–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Tzanakis N, Samiou M, Bouros D, Mouzas J, Kouroumalis E, Siafakas NM. Small airways function in patients with inflammatory bowel disease. Am J Respir Crit Care Med. 1998;157(2):382–6. [DOI] [PubMed] [Google Scholar]
- 47. Morris A, Beck JM, Schloss PD, Campbell TB, Crothers K, Curtis JL, Flores SC, Fontenot AP, Ghedin E, Huang Let al. . Comparison of the respiratory microbiome in healthy nonsmokers and smokers. Am J Respir Crit Care Med. 2013;187(10):1067–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Khosravi A, Yáñez A, Price JG, Chow A, Merad M, Goodridge HS, Mazmanian SK. Gut microbiota promote hematopoiesis to control bacterial infection. Cell Host Microbe. 2014;15(3):374–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Trompette A, Gollwitzer ES, Yadava K, Sichelstiel AK, Sprenger N, Ngom-Bru C, Blanchard C, Junt T, Nicod LP, Harris NLet al. . Gut microbiota metabolism of dietary fiber influences allergic airway disease and hematopoiesis. Nat Med. 2014;20(2):159–66. [DOI] [PubMed] [Google Scholar]
- 50. Xiao F, Tang M, Zheng X, Liu Y, Li X, Shan H. Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology. 2020;158(6):1831–3.e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Xiao F, Sun J, Xu Y, Li F, Huang X, Li H, Zhao J, Huang J, Zhao J. Infectious SARS-CoV-2 in feces of patient with severe COVID-19. Emerg Infect Dis. 2020;26(8):1920–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Zuo T, Zhang F, Lui GCY, Yeoh YK, Li AYL, Zhan H, Wan Y, Chung A, Cheung CP, Chen Net al. . Alterations in gut microbiota of patients with COVID-19 during time of hospitalization. Gastroenterology. 2020;159:944–55..e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Zuo T, Zhan H, Zhang F, Liu Q, Tso EYK, Lui GCY, Chen N, Li A, Lu W, Chan FKLet al. . Alterations in fecal fungal microbiome of patients with COVID-19 during time of hospitalization until discharge. Gastroenterology. 2020;159:1302–10..e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Thingholm LB, Rühlemann MC, Koch M, Fuqua B, Laucke G, Boehm R, Bang C, Franzosa EA, Hübenthal M, Rahnavard Aet al. . Obese individuals with and without type 2 diabetes show different gut microbial functional capacity and composition. Cell Host Microbe. 2019;26(2):252–264..e10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Sencio V, Barthelemy A, Tavares LP, Machado MG, Soulard D, Cuinat C, Queiroz-Junior CM, Noordine ML, Salomé-Desnoulez S, Deryuter Let al. . Gut dysbiosis during influenza contributes to pulmonary pneumococcal superinfection through altered short-chain fatty acid production. Cell Rep. 2020;30(9):2934–47..e6. [DOI] [PubMed] [Google Scholar]
- 56. Turnbaugh PJ, Hamady M, Yatsunenko T, Cantarel BL, Duncan A, Ley RE, Sogin ML, Jones WJ, Roe BA, Affourtit JPet al. . A core gut microbiome in obese and lean twins. Nature. 2009;457(7228):480–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Xu C, Zhu H, Qiu P. Aging progression of human gut microbiota. BMC Microbiol. 2019;19(1):236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Jensen BA, Marette A. Microbial translocation in type 2 diabetes: when bacterial invaders overcome host defence in human obesity. Gut. 2020;69(10):1724–6. [DOI] [PubMed] [Google Scholar]
- 59. Wan Y, Li J, Shen L, Zou Y, Hou L, Zhu L, Faden HS, Tang Z, Shi M, Jiao Net al. . Enteric involvement in hospitalised patients with COVID-19 outside Wuhan. Lancet Gastroenterol Hepatol. 2020;5(6):534–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Aumpan N, Nunanan P, Vilaichone R-k. Gastrointestinal manifestation as clinical predictor of severe COVID-19: a retrospective experience and literature review of COVID-19 in Association of Southeast Asian Nations (ASEAN). JGH Open 4(6):10961101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Cani PD, Bibiloni R, Knauf C, Waget A, Neyrinck AM, Delzenne NM, Burcelin R. Changes in gut microbiota control metabolic endotoxemia-induced inflammation in high-fat diet-induced obesity and diabetes in mice. Diabetes. 2008;57(6):1470–81. [DOI] [PubMed] [Google Scholar]
- 62. Turnbaugh PJ, Backhed F, Fulton L, Gordon JI. Diet-induced obesity is linked to marked but reversible alterations in the mouse distal gut microbiome. Cell Host Microbe. 2008;3(4):213–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Ryu JK, Kim SJ, Rah SH, Kang JI, Jung HE, Lee D, Lee HK, Lee JO, Park BS, Yoon TYet al. . Reconstruction of LPS transfer cascade reveals structural determinants within LBP, CD14, and TLR4-MD2 for efficient LPS recognition and transfer. Immunity. 2017;46(1):38–50. [DOI] [PubMed] [Google Scholar]
- 64. Sakai J, Cammarota E, Wright JA, Cicuta P, Gottschalk RA, Li N, Fraser IDC, Bryant CE. Lipopolysaccharide-induced NF-κB nuclear translocation is primarily dependent on MyD88, but TNFα expression requires TRIF and MyD88. Sci Rep. 2017;7(1):1428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Imai Y, Kuba K, Neely GG, Yaghubian-Malhami R, Perkmann T, van Loo G, Ermolaeva M, Veldhuizen R, Leung YH, Wang Het al. . Identification of oxidative stress and Toll-like receptor 4 signaling as a key pathway of acute lung injury. Cell. 2008;133(2):235–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Hoel H, Heggelund L, Reikvam DH, Stiksrud B, Ueland T, Michelsen AE, Otterdal K, Muller KE, Lind A, Muller Fet al. . Elevated markers of gut leakage and inflammasome activation in COVID-19 patients with cardiac involvement. J Intern Med. [Internet]2020. doi:10.1111/joim.13178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Belančić A. Gut microbiome dysbiosis and endotoxemia – additional pathophysiological explanation for increased COVID-19 severity in obesity. Obes Med. 2020;20:100302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Onishi JC, Häggblom MM, Shapses SA. Can dietary fatty acids affect the COVID-19 infection outcome in vulnerable populations?. mBio. 2020;11(4):e01723–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Anhê FF, Jensen BAH, Varin TV, Servant F, Van Blerk S, Richard D, Marceau S, Surette M, Biertho L, Lelouvier Bet al. . Type 2 diabetes influences bacterial tissue compartmentalisation in human obesity. Nat Metab. 2020;2(3):233–42. [DOI] [PubMed] [Google Scholar]
- 70. Le Chatelier E, Nielsen T, Qin J, Prifti E, Hildebrand F, Falony G, Almeida M, Arumugam M, Batto JM, Kennedy Set al. . Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500(7464):541–6. [DOI] [PubMed] [Google Scholar]
- 71. Almandoz JP, Xie L, Schellinger JN, Mathew MS, Gazda C, Ofori A, Kukreja S, Messiah SE. Impact of COVID-19 stay-at-home orders on weight-related behaviours among patients with obesity. Clin Obes. 2020:e12386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Wang C, Pan R, Wan X, Tan Y, Xu L, Ho CS, Ho RC. Immediate psychological responses and associated factors during the initial stage of the 2019 coronavirus disease (COVID-19) epidemic among the general population in China. Int J Environ Res Public Health. 2020;17(5):1729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Zheng P, Zeng B, Zhou C, Liu M, Fang Z, Xu X, Zeng L, Chen J, Fan S, Du Xet al. . Gut microbiome remodeling induces depressive-like behaviors through a pathway mediated by the host's metabolism. Mol Psychiatry. 2016;21(6):786–96. [DOI] [PubMed] [Google Scholar]
- 74. Bruce-Keller AJ, Salbaum JM, Luo M, Blanchard E IV, Taylor CM, Welsh DA, Berthoud HR. Obese-type gut microbiota induce neurobehavioral changes in the absence of obesity. Biol Psychiatry. 2015;77(7):607–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Abot A, Wemelle E, Laurens C, Paquot A, Pomie N, Carper D, Bessac A, Mas Orea X, Fremez C, Fontanie Met al. . Identification of new enterosynes using prebiotics: roles of bioactive lipids and mu-opioid receptor signalling in humans and mice. Gut. [Internet]2020. doi:10.1136/gutjnl-2019-320230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Yano JM, Yu K, Donaldson GP, Shastri GG, Ann P, Ma L, Nagler CR, Ismagilov RF, Mazmanian SK, Hsiao EY. Indigenous bacteria from the gut microbiota regulate host serotonin biosynthesis. Cell. 2015;161(2):264–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Dalile B, Van Oudenhove L, Vervliet B, Verbeke K. The role of short-chain fatty acids in microbiota-gut-brain communication. Nat Rev Gastroenterol Hepatol. 2019;16(8):461–78. [DOI] [PubMed] [Google Scholar]
- 78. Montinari MR, Minelli S, De Caterina R. The first 3500 years of aspirin history from its roots – a concise summary. Vasc Pharmacol. 2019;113:1–8. [DOI] [PubMed] [Google Scholar]
- 79. Bailey C, Day C. Metformin: its botanical background. Pract Diab Int. 2004;21(3):115–17. [Google Scholar]
- 80. Anhe FF, Choi BSY, Dyck JRB, Schertzer JD, Marette A. Host-microbe interplay in the cardiometabolic benefits of dietary polyphenols. Trends Endocrinol Metab. 2019;30:384–95. [DOI] [PubMed] [Google Scholar]
- 81. Leopoldini M, Marino T, Russo N, Toscano M. Antioxidant properties of phenolic compounds: H-atom versus electron transfer mechanism. J Phys Chem A. 2004;108(22):4916–22. [Google Scholar]
- 82. Yi L, Li Z, Yuan K, Qu X, Chen J, Wang G, Zhang H, Luo H, Zhu L, Jiang Pet al. . Small molecules blocking the entry of severe acute respiratory syndrome coronavirus into host cells. J Virol. 2004;78(20):11334–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Joshi T, Joshi T, Sharma P, Mathpal S, Pundir H, Bhatt V, Chandra S. In silico screening of natural compounds against COVID-19 by targeting Mpro and ACE2 using molecular docking. Eur Rev Med Pharmacol Sci. 2020;24(8):4529–36. [DOI] [PubMed] [Google Scholar]
- 84. Horne JR, Vohl MC. Biological plausibility for interactions between dietary fat, resveratrol, ACE2, and SARS-CoV illness severity. Am J Physiol Endocrinol Metab. 2020;318(5):E830–e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Tiao MM, Lin YJ, Yu HR, Sheen JM, Lin IC, Lai YJ, Tain YL, Huang LT, Tsai CC. Resveratrol ameliorates maternal and post-weaning high-fat diet-induced nonalcoholic fatty liver disease via renin-angiotensin system. Lipids Health Dis. 2018;17(1):178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Oliveira Andrade JM, Paraíso AF, Garcia ZM, Ferreira AV, Sinisterra RD, Sousa FB, Guimarães AL, de Paula AM, Campagnole-Santos MJ, dos Santos RAet al. . Cross talk between angiotensin-(1-7)/Mas axis and sirtuins in adipose tissue and metabolism of high-fat feed mice. Peptides. 2014;55:158–65. [DOI] [PubMed] [Google Scholar]
- 87. Guerrero L, Castillo J, Quiñones M, Garcia-Vallvé S, Arola L, Pujadas G, Muguerza B. Inhibition of angiotensin-converting enzyme activity by flavonoids: structure-activity relationship studies. PLoS One. 2012;7(11):e49493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88. Lin SC, Ho CT, Chuo WH, Li S, Wang TT, Lin CC. Effective inhibition of MERS-CoV infection by resveratrol. BMC Infect Dis. 2017;17(1):144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Chen L, Li J, Luo C, Liu H, Xu W, Chen G, Liew OW, Zhu W, Puah CM, Shen Xet al. . Binding interaction of quercetin-3-beta-galactoside and its synthetic derivatives with SARS-CoV 3CL(pro): structure-activity relationship studies reveal salient pharmacophore features. Bioorg Med Chem. 2006;14(24):8295–306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Park JY, Yuk HJ, Ryu HW, Lim SH, Kim KS, Park KH, Ryu YB, Lee WS. Evaluation of polyphenols from Broussonetia papyrifera as coronavirus protease inhibitors. J Enzyme Inhib Med Chem. 2017;32(1):504–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Liu Y, Liang C, Xin L, Ren X, Tian L, Ju X, Li H, Wang Y, Zhao Q, Liu Het al. . The development of coronavirus 3C-like protease (3CL(pro)) inhibitors from 2010 to 2020. Eur J Med Chem. 2020;206:112711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Mody V, Ho J, Wills S, Mawri A, Lawson L, Ebert M, Fortin GM, Rayalam S, Taval S. Identification of 3-chymotrypsin like protease (3CLPro) inhibitors as potential anti-SARS-CoV-2 agents. Commun Biol. 2021;4(1):93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Patel D, Athar M, Jha PC. Computational investigation of binding of chloroquinone and hydroxychloroquinone against PLPro of SARS-CoV-2. J Biomol Struct Dyn. [Internet]2020:1–11.. doi:10.1080/07391102.2020.1844804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Cardona F, Andres-Lacueva C, Tulipani S, Tinahones FJ, Queipo-Ortuno MI. Benefits of polyphenols on gut microbiota and implications in human health. J Nutr Biochem. 2013;24(8):1415–22. [DOI] [PubMed] [Google Scholar]
- 95. Espín JC, González-Sarrías A, Tomás-Barberán FA. The gut microbiota: a key factor in the therapeutic effects of (poly)phenols. Biochem Pharmacol. 2017;139:82–93. [DOI] [PubMed] [Google Scholar]
- 96. ClinicalTrials.gov. Homepage. [Internet]. Bethesa (MD): US National Library of Medicine; 2004[cited November 19, 2020]. Available from: https://www.clinicaltrials.gov/ [Google Scholar]
- 97. Anhe FF, Roy D, Pilon G, Dudonne S, Matamoros S, Varin TV, Garofalo C, Moine Q, Desjardins Y, Levy Eet al. . A polyphenol-rich cranberry extract protects from diet-induced obesity, insulin resistance and intestinal inflammation in association with increased Akkermansia spp. population in the gut microbiota of mice. Gut. 2015;64(6):872–83. [DOI] [PubMed] [Google Scholar]
- 98. Kuhn P, Kalariya HM, Poulev A, Ribnicky DM, Jaja-Chimedza A, Roopchand DE, Raskin I. Grape polyphenols reduce gut-localized reactive oxygen species associated with the development of metabolic syndrome in mice. PLoS One. 2018;13(10):e0198716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Van Hul M, Geurts L, Plovier H, Druart C, Everard A, Stahlman M, Rhimi M, Chira K, Teissedre PL, Delzenne NMet al. . Reduced obesity, diabetes, and steatosis upon cinnamon and grape pomace are associated with changes in gut microbiota and markers of gut barrier. Am J Physiol Endocrinol Metab. 2018;314(4):E334–e52. [DOI] [PubMed] [Google Scholar]
- 100. Pierre JF, Heneghan AF, Feliciano RP, Shanmuganayagam D, Roenneburg DA, Krueger CG, Reed JD, Kudsk KA. Cranberry proanthocyanidins improve the gut mucous layer morphology and function in mice receiving elemental enteral nutrition. JPEN J Parenter Enteral Nutr. 2013;37(3):401–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Anhe FF, Nachbar RT, Varin TV, Vilela V, Dudonne S, Pilon G, Fournier M, Lecours MA, Desjardins Y, Roy Det al. . A polyphenol-rich cranberry extract reverses insulin resistance and hepatic steatosis independently of body weight loss. Molecular metabolism. 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Anhe FF, Nachbar RT, Varin TV, Trottier J, Dudonne S, Le Barz M, Feutry P, Pilon G, Barbier O, Desjardins Yet al. . Treatment with camu camu (Myrciaria dubia) prevents obesity by altering the gut microbiota and increasing energy expenditure in diet-induced obese mice. Gut. 2019;68:453–64. [DOI] [PubMed] [Google Scholar]
- 103. Roopchand DE, Carmody RN, Kuhn P, Moskal K, Rojas-Silva P, Turnbaugh PJ, Raskin I. Dietary polyphenols promote growth of the gut bacterium Akkermansia muciniphila and attenuate high-fat diet-induced metabolic syndrome. Diabetes. 2015;64(8):2847–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Henning SM, Yang J, Shao P, Lee RP, Huang J, Ly A, Hsu M, Lu QY, Thames G, Heber Det al. . Health benefit of vegetable/fruit juice-based diet: role of microbiome. Sci Rep. 2017;7(1):2167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Koudoufio M, Desjardins Y, Feldman F, Spahis S, Delvin E, Levy E. Insight into polyphenol and gut microbiota crosstalk: are their metabolites the key to understand protective effects against metabolic disorders?. Antioxidants (Basel). 2020;9(10):982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Morrow LE, Kollef MH, Casale TB. Probiotic prophylaxis of ventilator-associated pneumonia: a blinded, randomized, controlled trial. Am J Respir Crit Care Med. 2010;182(8):1058–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Zeng J, Wang CT, Zhang FS, Qi F, Wang SF, Ma S, Wu TJ, Tian H, Tian ZT, Zhang SLet al. . Effect of probiotics on the incidence of ventilator-associated pneumonia in critically ill patients: a randomized controlled multicenter trial. Intensive Care Med. 2016;42(6):1018–28. [DOI] [PubMed] [Google Scholar]
- 108. Cavicchioli VQ, Carvalho OV, Paiva JC, Todorov SD, Silva Júnior A, Nero LA. Inhibition of herpes simplex virus 1 (HSV-1) and poliovirus (PV-1) by bacteriocins from Lactococcus lactis subsp. lactis and Enterococcus durans strains isolated from goat milk. Int J Antimicrob Agents. 2018;51(1):33–7. [DOI] [PubMed] [Google Scholar]
- 109. Ermolenko EI, Desheva YA, Kolobov AA, Kotyleva MP, Sychev IA, Suvorov AN. Anti-influenza activity of enterocin B in vitro and protective effect of bacteriocinogenic enterococcal probiotic strain on influenza infection in mouse model. Probiotics Antimicrob Proteins. 2019;11(2):705–12. [DOI] [PubMed] [Google Scholar]
- 110. Ichinohe T, Pang IK, Kumamoto Y, Peaper DR, Ho JH, Murray TS, Iwasaki A. Microbiota regulates immune defense against respiratory tract influenza A virus infection. Proc Natl Acad Sci U S A. 2011;108(13):5354–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111. Wu S, Jiang ZY, Sun YF, Yu B, Chen J, Dai CQ, Wu XL, Tang XL, Chen XY. Microbiota regulates the TLR7 signaling pathway against respiratory tract influenza A virus infection. Curr Microbiol. 2013;67(4):414–22. [DOI] [PubMed] [Google Scholar]
- 112. Mahooti M, Abdolalipour E, Salehzadeh A, Mohebbi SR, Gorji A, Ghaemi A. Immunomodulatory and prophylactic effects of Bifidobacterium bifidum probiotic strain on influenza infection in mice. World J Microbiol Biotechnol. 2019;35(6):91. [DOI] [PubMed] [Google Scholar]
- 113. Telcian AG, Zdrenghea MT, Edwards MR, Laza-Stanca V, Mallia P, Johnston SL, Stanciu LA. Vitamin D increases the antiviral activity of bronchial epithelial cells in vitro. Antiviral Res. 2017;137:93–101. [DOI] [PubMed] [Google Scholar]
- 114. Khare D, Godbole NM, Pawar SD, Mohan V, Pandey G, Gupta S, Kumar D, Dhole TN, Godbole MM. Calcitriol [1, 25[OH]2 D3] pre- and post-treatment suppresses inflammatory response to influenza A (H1N1) infection in human lung A549 epithelial cells. Eur J Nutr. 2013;52(4):1405–15. [DOI] [PubMed] [Google Scholar]
- 115. Greiller CL, Suri R, Jolliffe DA, Kebadze T, Hirsman AG, Griffiths CJ, Johnston SL, Martineau AR. Vitamin D attenuates rhinovirus-induced expression of intercellular adhesion molecule-1 (ICAM-1) and platelet-activating factor receptor (PAFR) in respiratory epithelial cells. J Steroid Biochem Mol Biol. 2019;187:152–9. [DOI] [PubMed] [Google Scholar]
- 116. Hayashi H, Okamatsu M, Ogasawara H, Tsugawa N, Isoda N, Matsuno K, Sakoda Y. Oral supplementation of the vitamin D metabolite 25(OH)D(3) against influenza virus infection in mice. Nutrients. 2020;12(7):2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Martineau AR, Jolliffe DA, Hooper RL, Greenberg L, Aloia JF, Bergman P, Dubnov-Raz G, Esposito S, Ganmaa D, Ginde AAet al. . Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta-analysis of individual participant data. BMJ. 2017;356:i6583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Tan CW, Ho LP, Kalimuddin S, Cherng BPZ, Teh YE, Thien SY, Wong HM, Tern PJW, Chandran M, Chay JWMet al. . Cohort study to evaluate effect of vitamin D, magnesium, and vitamin B(12) in combination on severe outcome progression in older patients with coronavirus (COVID-19). Nutrition. 2020;79-80:111017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Ohaegbulam KC, Swalih M, Patel P, Smith MA, Perrin R. Vitamin D supplementation in COVID-19 patients: a clinical case series. Am J Ther. 2020;27(5):e485–e90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Toelzer C, Gupta K, Yadav SKN, Borucu U, Davidson AD, Kavanagh Williamson M, Shoemark DK, Garzoni F, Staufer O, Milligan Ret al. . Free fatty acid binding pocket in the locked structure of SARS-CoV-2 spike protein. Science. 2020;370:725–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Morita M, Kuba K, Ichikawa A, Nakayama M, Katahira J, Iwamoto R, Watanebe T, Sakabe S, Daidoji T, Nakamura Set al. . The lipid mediator protectin D1 inhibits influenza virus replication and improves severe influenza. Cell. 2013;153(1):112–25. [DOI] [PubMed] [Google Scholar]
- 122. Xia H, Chen L, Liu H, Sun Z, Yang W, Yang Y, Cui S, Li S, Wang Y, Song Let al. . Protectin DX increases survival in a mouse model of sepsis by ameliorating inflammation and modulating macrophage phenotype. Sci Rep. 2017;7(1):99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Tan W, Chen L, Wang YX, Hu LS, Xiong W, Shang Y, Yao SL. Protectin DX exhibits protective effects in mouse model of lipopolysaccharide-induced acute lung injury. Chin Med J (Engl). 2018;131(10):1167–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. White PJ, St-Pierre P, Charbonneau A, Mitchell PL, St-Amand E, Marcotte B, Marette A. Protectin DX alleviates insulin resistance by activating a myokine-liver glucoregulatory axis. Nat Med. 2014;20(6):664–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Chen P, Fenet B, Michaud S, Tomczyk N, Véricel E, Lagarde M, Guichardant M. Full characterization of PDX, a neuroprotectin/protectin D1 isomer, which inhibits blood platelet aggregation. FEBS Lett. 2009;583(21):3478–84. [DOI] [PubMed] [Google Scholar]
- 126. Liu M, Boussetta T, Makni-Maalej K, Fay M, Driss F, El-Benna J, Lagarde M, Guichardant M. Protectin DX, a double lipoxygenase product of DHA, inhibits both ROS production in human neutrophils and cyclooxygenase activities. Lipids. 2014;49(1):49–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Hill C, Guarner F, Reid G, Gibson GR, Merenstein DJ, Pot B, Morelli L, Canani RB, Flint HJ, Salminen Set al. . Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat Rev Gastroenterol Hepatol. 2014;11(8):506–14. [DOI] [PubMed] [Google Scholar]
- 128. Richardson S, Hirsch JS, Narasimhan M, Crawford JM, McGinn T, Davidson KW, Barnaby DP, Becker LB, Chelico JD, Cohen SLet al. . Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. JAMA. 2020;323(20):2052–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Wang Z, Chai W, Burwinkel M, Twardziok S, Wrede P, Palissa C, Esch B, Schmidt MF. Inhibitory influence of Enterococcus faecium on the propagation of swine influenza A virus in vitro. PLoS One. 2013;8(1):e53043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130. Botić T, Klingberg TD, Weingartl H, Cencic A. A novel eukaryotic cell culture model to study antiviral activity of potential probiotic bacteria. Int J Food Microbiol. 2007;115(2):227–34. [DOI] [PubMed] [Google Scholar]
- 131. Mak JWY, Chan FKL, Ng SC. Probiotics and COVID-19: one size does not fit all. Lancet Gastroenterol Hepatol. 2020;5(7):644–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132. Tiwari SK, Dicks LMT, Popov IV, Karaseva A, Ermakov AM, Suvorov A, Tagg JR, Weeks R, Chikindas ML. Probiotics at war against viruses: what is missing from the picture?. Front Microbiol. 2020;11:1877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Drider D, Bendali F, Naghmouchi K, Chikindas ML. Bacteriocins: not only antibacterial agents. Probiotics Antimicrob Proteins. 2016;8(4):177–82. [DOI] [PubMed] [Google Scholar]
- 134. Bikle DD. Vitamin D metabolism, mechanism of action, and clinical applications. Chem Biol. 2014;21(3):319–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135. Itkonen ST, Andersen R, Björk AK, Brugård Konde Å, Eneroth H, Erkkola M, Holvik K, Madar AA, Meyer HE, Tetens Iet al. . Vitamin D status and current policies to achieve adequate vitamin D intake in the Nordic countries. Scand J Public Health. [Internet]2020. doi:10.1177/1403494819896878. [DOI] [PubMed] [Google Scholar]
- 136. Pilz S, März W, Cashman KD, Kiely ME, Whiting SJ, Holick MF, Grant WB, Pludowski P, Hiligsmann M, Trummer Cet al. . Rationale and plan for vitamin D food fortification: a review and guidance paper. Front Endocrinol. 2018;9:373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Palacios C, Gonzalez L. Is vitamin D deficiency a major global public health problem?. J Steroid Biochem Mol Biol. 2014;144:138–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Miñambres I, Sánchez-Quesada JL, Vinagre I, Sánchez-Hernández J, Urgell E, de Leiva A, Pérez A. Hypovitaminosis D in type 2 diabetes: relation with features of the metabolic syndrome and glycemic control. Endocr Res. 2015;40(3):160–5. [DOI] [PubMed] [Google Scholar]
- 139. Daniel D, Hardigan P, Bray N, Penzell D, Savu C. The incidence of vitamin D deficiency in the obese: a retrospective chart review. J Community Hosp Intern Med Perspect. 2015;5(1):26069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Vranić L, Mikolašević I, Milić S. Vitamin D deficiency: consequence or cause of obesity?. Medicina (Kaunas). 2019;55(9):541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141. Laird E, O'Halloran AM, Carey D, Healy M, O'Connor D, Moore P, Shannon T, Molloy AM, Kenny RA. The prevalence of vitamin D deficiency and the determinants of 25(OH)D concentration in older Irish adults: data from the Irish Longitudinal Study on Ageing (TILDA). J Gerontol A Biol Sci Med Sci. 2018;73(4):519–25. [DOI] [PubMed] [Google Scholar]
- 142. Takeuti FAC, Guimaraes FSF, Guimaraes PSF. Applications of vitamin D in sepsis prevention. Discov Med. 2018;25(140):291–7. [PubMed] [Google Scholar]
- 143. Pike JW, Meyer MB. The vitamin D receptor: new paradigms for the regulation of gene expression by 1,25-dihydroxyvitamin D(3). Endocrinol Metab Clin North Am. 2010;39(2):255–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Vors C, Allaire J, Mejia SB, Khan TA, Sievenpiper JL, Lamarche B. Comparing the effects of docosahexaenoic and eicosapentaenoic acids on inflammation markers using pairwise and network meta-analyses of randomized controlled trials. Adv Nutr. 2021;12:128–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Coronavirus disease (COVID-19): for health professionals. [Internet]. Government of Canada; 2020[cited November 19, 2020]. Available from: https://www.canada.ca/en/public-health/services/diseases/2019-novel-coronavirus-infection/health-professionals.html [Google Scholar]
- 146. Shahidi F, Ambigaipalan P. Omega-3 polyunsaturated fatty acids and their health benefits. Annu Rev Food Sci Technol. 2018;9:345–81. [DOI] [PubMed] [Google Scholar]
- 147. Levy BD. Resolvins and protectins: natural pharmacophores for resolution biology. Prostaglandins Leukot Essent Fatty Acids. 2010;82(4-6):327–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148. Hansen TV, Vik A, Serhan CN. The protectin family of specialized pro-resolving mediators: potent immunoresolvents enabling innovative approaches to target obesity and diabetes. Front Pharmacol. 2019;9:1582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149. Klok FA, Kruip M, van der Meer NJM, Arbous MS, Gommers D, Kant KM, Kaptein FHJ, van Paassen J, Stals MAM, Huisman MVet al. . Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Sancéau JY, Maltais R, Poirier D, Marette A. Total synthesis of the antidiabetic (type 2) lipid mediator protectin DX/PDX. J Org Chem. 2019;84(2):495–505. [DOI] [PubMed] [Google Scholar]
- 151. Pal A, Gowdy KM, Oestreich KJ, Beck M, Shaikh SR. Obesity-driven deficiencies of specialized pro-resolving mediators may drive adverse outcomes during SARS-CoV-2 infection. Front Immunol. 2020;11:1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. Kang YH, Jin JS, Yi DW, Son SM. Bone morphogenetic protein-7 inhibits vascular calcification induced by high vitamin D in mice. Tohoku J Exp Med. 2010;221(4):299–307. [DOI] [PubMed] [Google Scholar]
- 153. Doron S, Snydman DR. Risk and safety of probiotics. Clin Infect Dis. 2015;60(Suppl 2):S129–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Markowiak P, Śliżewska K. Effects of probiotics, prebiotics, and synbiotics on human health. Nutrients. 2017;9(9):1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Mennen LI, Walker R, Bennetau-Pelissero C, Scalbert A. Risks and safety of polyphenol consumption. Am J Clin Nutr. 2005;81(1 Suppl):326S. [DOI] [PubMed] [Google Scholar]
- 156. Brasky TM, Darke AK, Song X, Tangen CM, Goodman PJ, Thompson IM, Meyskens FL Jr, Goodman GE, Minasian LM, Parnes HLet al. . Plasma phospholipid fatty acids and prostate cancer risk in the SELECT trial. J Natl Cancer Inst. 2013;105(15):1132–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Szymanski KM, Wheeler DC, Mucci LA. Fish consumption and prostate cancer risk: a review and meta-analysis. Am J Clin Nutr. 2010;92(5):1223–33. [DOI] [PubMed] [Google Scholar]