Abstract
Coronavirus disease 2019 (COVID-19) is largely threatening global public health, social stability, and economy. Efforts of the scientific community are turning to this global crisis and should present future preventative measures. With recent trends in polymer science that use plasma to activate and enhance the functionalities of polymer surfaces by surface etching, surface grafting, coating and activation combined with recent advances in understanding polymer-virus interactions at the nanoscale, it is promising to employ advanced plasma processing for smart antiviral applications. This trend article highlights the innovative and emerging directions and approaches in plasma-based surface engineering to create antiviral polymers. After introducing the unique features of plasma processing of polymers, novel plasma strategies that can be applied to engineer polymers with antiviral properties are presented and critically evaluated. The challenges and future perspectives of exploiting the unique plasma-specific effects to engineer smart polymers with virus-capture, virus-detection, virus-repelling, and/or virus-inactivation functionalities for biomedical applications are analysed and discussed.
Keywords: Antiviral polymers, surface modification, plasma processing
Abbreviations: COVID-19, coronavirus disease 2019; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; PPE, personal protective equipment; DC, direct current; MW, microwave; WCA, water contact angle; PC, polycarbonate; PTFE, polytetrafluoroethylene; PP, polypropylene; PECVD, plasma-enhanced chemical vapour deposition; HMDSO, hexamethyldisiloxane; CF4, tetrafluoromethane; H2, hydrogen; PDMS, polydimethylsiloxane; RSV, respiratory syncytial virus; RV, rhinovirus; TEOS-O2, tetraethyl orthosilicate and oxygen; IPNpp, plasma polymerized isopentyl nitrite; PET, polyethene terephthalate; REF, reference; RONS, reactive oxygen and nitrogen species; NO, nitric oxide; ACE2, angiotensin-converting enzyme 2; ΔD, the variation of the dissipation; Δf, the frequency shift; PFM, pentafluorophenyl methacrylate; PS, polystyrene; BSA, bovine serum albumin; RF, radio frequency; MERS-CoV, middle east respiratory syndrome; PVC, polyvinyl chloride; SEM, scanning electron microscopy; PEG, polyethene glycol; RT-PCR, reverse transcription-polymerase chain reaction; HBV, hepatitis B virus; UV, ultraviolet
Graphical Abstract
1. Introduction
Ever since the first outbreak of coronavirus disease 2019 (COVID-19) in December 2019, it took just a few months before it was declared a global pandemic in March 2020 [1,2]. Despite enormous efforts dedicated to COVID-19 control, many countries are still suffering from the ongoing local transmission with a persistent threat to the public health, social stability, and economy. The current pandemic is caused by a novel human coronavirus that is named severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [3]. Although not all mechanisms responsible for the spread of the virus are already fully understood, it is believed that current known routes for many other viruses are also applicable. Similar to other viruses, SARS-CoV-2 spreads through virion-laden respiratory droplets, direct contact, airborne aerosols, and contaminated surfaces [4,5]. It is worth noting that surfaces in hospital rooms of infected patients such as floors, bedside lockers, cardiac tables, and electric switches were found to contain coronavirus particles [6,7], which could be responsible for the nosocomial outbreak at the early stage of the pandemic [8]. Moreover, previous studies have demonstrated that various viruses, including SARS-CoV-2, can remain viable and infectious in aerosols for hours and on surfaces including plastics, stainless steels, copper, and card-boards up to days [9,10], suggesting that surfaces such as food packages, clothes, transports, and building elements could also impose potential threats during the pandemic period. In response, these high-touch surfaces are thus needed to be able to a) capture, b) detect, c) repel, and/or even d) inactivate viruses to provide humans with the necessary protection from viral disease infection. It is imperative to develop and fabricate antiviral surfaces for an effective reduction and prevention of the spread of the current COVID-19 and for the prevention of the outbreak of future novel virus-related pandemics.
In particular, the COVID-19 crisis has raised the global need for the specialized polymeric materials for personal protective equipment (PPE) and virus-detection devices as they are indispensable at the forefront of fighting against SARS-CoV-2 and other infectious diseases. However, the commonly used polymeric materials generally lack sufficient antiviral properties thus making front-line health-care workers vulnerable of being infected [11]. In literature, one of the particularly crucial strategies to control infectious diseases is to exploit modern surface modification techniques to create multifunctional materials with the desired ability to mitigate the spread of infectious pathogens [12], [13], [14]. Considerable research in the past decade has been conducted to combat bacterial pathogens by antibacterial agent release, contact-killing, as well as anti-adhesion and bacteria-repelling surfaces [15,16]. However, literature for a systematic study on broad-range antiviral surfaces is still scarce. Several reviews regarding antiviral surfaces have focused on either the performance of various antiviral agents or the virus-inactivation therapeutic tools [17], [18], [19], [20], but they generally focus on the specific surface engineering methods for engineering antiviral surfaces. There is an emerging trend in developing innovative antiviral surfaces based on the well-established antibacterial experience in the scientific community [17]. In this regard, highlighting an effective and promising surface engineering technique could thus be of critical importance for accelerating the related studies.
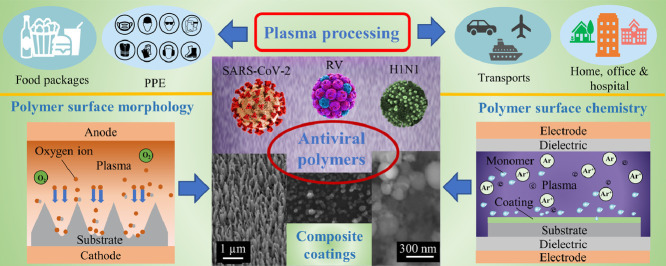
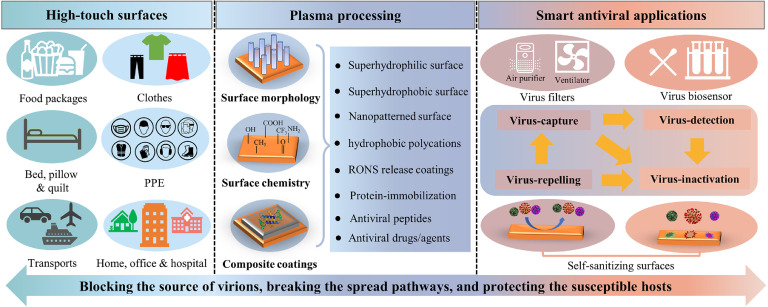
This trend article aims to highlight the state-of-the-art in the development of antiviral surfaces using the diversified functionalities of the low-temperature plasma technology in polymer surface engineering. Both focus and scope of this article differ from the available publications on the intrinsic (e.g. natural or biomimetic) antiviral polymers used as antiviral therapeutics [21] and on plasma methods for surface functionalization of polymers [22], plasma surface chemistry [23], and antibacterial surface modification [24,25]. The distinctive feature of this article is in the systematic discussion of the possibility of processing common polymers with antiviral properties by plasma-based methods. Considering the virus as an object with a characteristic size at the nanometres scale, one could use nanoscale structuring and functionalization of polymer surfaces to prevent the spread of viruses. As indicated in Fig. 1 , plasma structured and functionalized polymers with nanoscale features can be used for protecting humans from virus infection by various antiviral strategies mainly including blocking the source of virions, breaking the spread pathways, and protecting the susceptible hosts. Novel advances in plasma surface engineering make the antiviral process non-toxic and environmentally friendly. As a result, a healthier living and working environment can be established that offers holistic solutions to people with compromised or weakened immune systems.
Fig. 1.
Graphic summary of options for plasma structured and functionalized polymer surfaces with nanoscale features to be used for protecting humans from viral infections. Plasma processing can be used to functionalize the high-touch surfaces, e.g., food packages, PPE, and some high-traffic objects such as doors and curtains in transports, homes, offices, and hospitals to protect people from getting infected. Moreover, plasma functionalized surfaces are promising to be used in smart antiviral applications, e.g., new virus biosensors with enhanced detection performance and novel self-sanitizing surfaces which involve some of the key antiviral processes including virus-capture, virus-detection, virus-repelling, and/or virus-inactivation. All these antiviral strategies meet the requirements of controlling infectious viral diseases by blocking the sources of virions, breaking the spread pathways, and protecting the susceptible hosts.
2. Unique features of plasma processing of polymers
Plasma is an ionized quasi-neutral gas known as the “fourth state of matter” that consists of various charge carriers such as electrons, ions, and reactive radicals. Among a variety of plasmas, non-thermal plasmas have been highly successful in the effective surface modification of polymeric materials for biomedical applications [26].
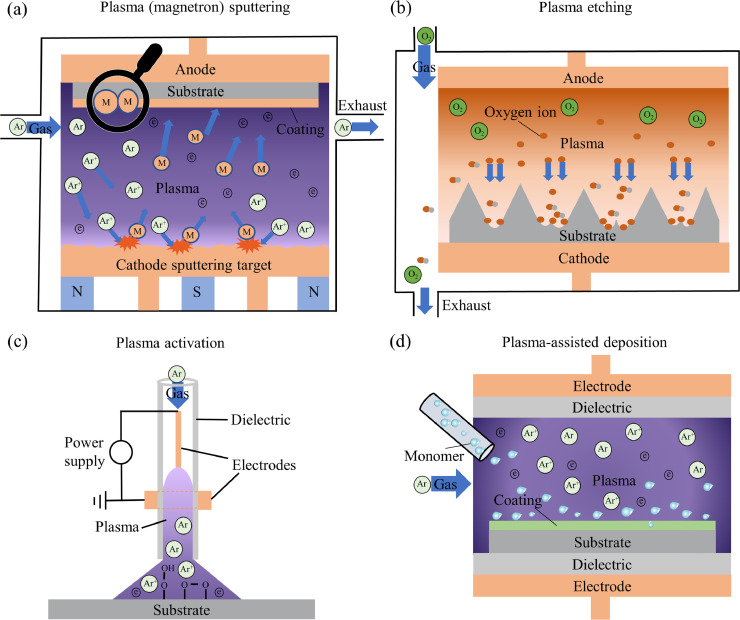
Generally, by taking advantage of high-energy particles in plasmas, several common approaches of plasma-enabled physical and chemical surface modifications can be determined as (i) plasma (magnetron) sputtering; (ii) plasma etching; (iii) plasma activation; and (iv) plasma-assisted deposition, etc. The schematic representation of various types of approaches for plasma processing of polymer surfaces is shown in Fig. 2 . Typically, plasma sputtering has been widely applied for the deposition of hard metallic and non-metallic coatings on flat materials [27]. Plasma etching has been widely used for the engineering of desired surface morphology with precise control at the nanoscale [28]. In contrast, plasma activation and plasma-assisted deposition processes can be employed for tailoring special surface chemistry combined with controlled surface morphology [22,29].
Fig. 2.
Schematic representation of typical approaches for plasma processing of polymers. (a) plasma (magnetron) sputtering, (b) plasma etching (taking reactive ion etching as an example), (c) plasma activation (in atmospheric-pressure open-air), and (d) plasma-assisted deposition.
Importantly, these processes are versatile and can be operated in broad process parameter windows including the frequencies of plasma power (e.g., from direct current (DC) to microwave (MW) and lasers), electrode configurations, gas pressures from ultrahigh vacuum to atmospheric, and spatial scales from atomic to mesoscales [30,31]. Therefore, plasma surface processing allows the fabrication of polymers to be scalable to almost any level with precise control over surface morphology and chemical composition. Moreover, the plasma-based approach is well-known as a solvent-free technique, which is regarded as one of the most promising environmentally friendly methods as an alternative to conventional wet chemistry. The high-speed, stability, repeatability, low-cost, and no damage to the bulk characteristics of treated polymers are also vital advantages over other conventional surface modification methods [32], [33], [34].
With recent advances in nanostructured biomaterials, these plasma methods have collectively emerged as an extremely efficient nanofabrication toolbox for polymer surface processing in biomedicine such as controlled drug release [33], tissue engineering scaffolds [35], and antibacterial surfaces [36]. Accordingly, plasma processing of polymers is expected to be one of the truly unique tools in the nanofabrication of antiviral materials under the current COVID-19 pandemic caused by the SARS-CoV-2. Diverse plasma-based nanofabrication methods make it possible to provide sufficient flexibility to target viruses such as SARS-CoV-2. These viruses can be considered as core-shell particles with relatively fast mutations [37], [38], [39], [40]. The specific targets in this regard include disrupting the spread pathways and affecting the surface-virus interactions [4].
3. Plasma processing of polymers to impart them with antiviral properties
Central to plasma processing of polymers to impart them with antiviral properties is the enhancement of nanotexture formation, functional groups grafting, and composite coating growth with targeted surface-virus interactions. This article aims to highlight the main strategies and the most relevant recent trends in plasma-assisted processing of polymer surfaces that can enable polymers with antiviral properties. Therefore, some of the most relevant plasma-engineered polymer surfaces that could be extended for antiviral applications are selected and discussed below. The subsections below represent the key strategies of plasma antiviral functionalization of polymer surfaces, namely surface morphology (Section 3.1), surface chemistry (Section 3.2), and composite coatings (Section 3.3).
3.1. Plasma processing of surface morphology
In the last decade, considerable progress has been achieved in precise control of surface morphology by different plasma methods, including plasma etching [41,42], plasma sputtering [43], and plasma-assisted deposition [44]. The surface properties such as wettability, adhesion, and mechanical performance that are highly sensitive to surface morphology can thus be precisely tailored to meet the high demand of desired applications in biology, energy, and electronics [28]. For virus-related applicatons, the virus-laden respiratory fluid aerosols or droplets can range from 10 μm to 100 μm in diameter (taking influenza A viruses as examples) [45]. However, a virus is typically in the range of tens to hundreds nm in size, while the size of its features (such as viral spikes or binding domains) is much smaller that is in tens nm range. Accordingly, the antiviral surface response for targeting should be presented at the scales from nanometres to micrometres and even to millimetres when the prevention of virus spread pathways is tackled.
Recent theoretical and experimental studies have suggested that the key to control respiratory virus transmission is to control the virus-containing aerosols or water droplets instead of the virus itself only [4,[46], [47], [48]]. Moreover, a positively charged nanofibrous microfiltration membrane with hydrophilic properties functionalized by wet chemistry processes has demonstrated virus filters with enhanced performance that can meet the specific needs in the purification of contaminated water [49], showing the potential in the prevention of human exposure even if aerosols or water droplets were generated from the water [50]. Therefore, surfaces with the desired wettability can play a crucial role in controlling virus activities. More specifically, the use of hydrophilic surface functionalization with water contact angle (WCA) less than 3° has ensured the collapse of aspirated water droplets containing the MS2 bacteriophage virus as shown for a bi-functional nano-composite coating on a porous Nomex® fabric by a dip-coating method [51]. The study [51] has demonstrated that superhydrophilic fabric can be used to separate the viruses from virus-containing water droplets and prevent viruses from spreading in water droplets over a long distance. Furthermore, a recent model has shown that superhydrophilic surfaces could be used to reduce the dry time of the virus-containing water droplets, and in the case of N95 mask/PPE bodywear, a reduction in WCA to 10° can reduce the risk of getting COVID-19 infection by around 38% [52]. It has to be emphasized that desiccation is an effective way for virus-inactivation as many survival activities of viruses such as the assembly of membrane bilayers for the enveloped viruses could not maintain without water [53]. Superhydrophilic surfaces are thus important during pandemics such as COVID-19 to replace the mass use of chemical disinfection solutions that could cause environmental pollution. However, relatively little effort has been devoted to this field whereas superhydrophilic surfaces specifically designed for antiviral applications can be used as a complimentary approach to chemical disinfection.
On the other hand, plasma technologies have been widely used for tailoring surface wettability of polymer surfaces, which provides ideal tools for relevant studies. Representative examples of plasma nanotexturing polymer surfaces with superhydrophilic properties are present in Fig. 3 (a) [54]. A forest of nanopillars with a number density of 39 8 μm−2 on polycarbonate (PC) surface can be observed at 20 min of 100 W oxygen plasma surface etching which induced a pronounced hydrophilic behaviour with values of 12° and 3° on the surface for the advancing and receding WCA, respectively. It is worth noting that such plasma etched polymer surfaces were subjected to a typical hydrophobic recovery with advancing WCA reaching 78° and receding WCA reaching 19° at 21 days after the processing [54]. However, when it was covered by a thin layer of the silica-like coatings by plasma-assisted deposition, the advancing WCA and receding WCA were slightly changed to 4° and 2°, respectively, and the superhydrophilicity of the covered coating was preserved for as long as 5 months [54], which is promising for practical applications.
Fig. 3.
Representative examples of plasma processing of polymers with controlled surface morphologies. (a) Superhydrophilic PC surface prepared by plasma etching. The scale bar is 1 µm. [54], Copyright 2011. Adapted with permission from John Wiley & Sons Inc. (b) A Cassie-Baxter state robust superhydrophobic PC surface obtained by oxygen plasma etching combined with PECVD using HMDSO as the precursor:(i) Surface morphology, (ii) WCA image showing WCA >170°, and (iii) the sharp bouncing behaviour when hit by 3 µL water droplets at an impact velocity of 1.1 m/s. The scale bars in (i) and (iii) are 1 µm and 1 mm, respectively. [60], Copyright 2014. Adapted with permission from Elsevier. (c) Plasma etching of polymer nanowires with controlled density and length. The figure shows examples of Kapton nanowires by covering the initial surface with 15 nm Au before plasma treatment. The scale bar is 1 µm. [62], Copyright 2009. Adapted with permission from the American Chemical Society. (d) Surface morphologies of ZnO nanowires grown on channel diffused plasma modified regions of PDMS substrate at different magnifications. The scale bars are 2 µm. [63], Copyright 2012. Adapted with permission from the Royal Society of Chemistry.
Likewise, superhydrophobic surfaces with the abilities to repel water droplets could also be targeted in some practical antiviral applications [55]. Recently, superhydrophobic surfaces with viral repellence properties have been achieved using polytetrafluoroethylene (PTFE) nanoparticles by a wet chemistry approach through thermal sintering in a liquid phase to polypropylene (PP) microfibers [56]. The coated textiles reduce the attachment of adenovirus type 4 and 7a virions by 99.2 ± 0.2% and 97.6 ± 0.1% (2.10 and 1.62 log), respectively, as compared with the non-coated controls. The outstanding performance can be attributed to the Cassie-Baxter state of wetting, a state where a liquid drop is located on top of a surface's rough features and traps pockets of gas between these features, leading to a very high WCA greater than 150° and a very low WCA hysteresis below 10° with water repellent and self-cleaning characteristics [57,58]. However, the environmental pollution level during the fabrication steps using solvents as used in [56] are quite high. Moreover, a recent study has shown that the laser-fabricated graphene-coated masks with outstanding superhydrophobic and photo-thermal performance are self-cleaning and reusable after sunlight sterilization [59]. However, such a method is very costly and only applicable at a small-scale without perspective for industrial-scale application, and more importantly, laser-induced surface modification of polymers may cause bulk damage to the material.
Plasma fabricating methods, on the other hand, are viable alternatives to the laser-based approaches. Superhydrophobic surfaces can be achieved by oxygen plasma etching combined with plasma-enhanced chemical vapour deposition (PECVD) using hexamethyldisiloxane (HMDSO) as the precursor (Fig. 3(b)) [60]. Importantly, the surfaces are also in a Cassie-Baxter state, which can result in sharp bouncing when hit by water droplets at high impact velocity (see Fig. 3(b) (iii)) [60]. Such surfaces prepared by plasma methods represent a class of polymers with water repellent and self-cleaning characteristics [61] and can theoretically be used to repel those respiratory viruses such as the above-mentioned influenza A and SARS-CoV-2 that mainly spread through virion-laden droplets. These polymer surfaces should be of high importance for medical equipment, protective clothes, and respiratory masks [55].
Plasma engineered nanopatterned surfaces. In many cases, the required morphology of a surface for many advanced applications is inspired by nature where nanotexturing of the materials is often a case for plants, animals, and insects [64]. The new trends in mimicking biological surfaces have led to the development of novel surfaces with unique properties. It has been reported that the topography of dragonfly wing surfaces has a bactericidal effect on Escherichia coli [65]. Similar behaviour of plasma engineered nanopatterned surfaces has been obtained by tetrafluoromethane (CF4) / hydrogen (H2) plasma texturing on a Si surface [66]. Moreover, some biomimetic plasma nanopatterned polymer surfaces have been demonstrated to convey bactericidal efficacy [67]. The mechanisms of bactericidal activity are mainly attributed to the stressing mechanical killing by nanostructured surfaces, known as contact-killing [68,69]. Despite the well-studied antibacterial effects, experimental evidence of the antiviral functionality of such polymer surfaces is rare. Viricidal effect of nanotexturing polymers should be investigated in the future.
Considering the difference between the bacteria and viruses regarding the structure and size, the requirement on the size and density of the nanostructured polymers for disabling viruses can be very high. Plasma fabrication can provide high flexibility in polymer morphology for subsequent research on antiviral activities. For example, plasma-assisted methods of deposition can provide sufficient flexibility in the fabrication of dense polymer nanowires with a typical length up to 7-8 μm [70]. One-step plasma etching of polymer films covered with a thin layer of metal nanoparticles has been demonstrated to fabricate large-scale polymer nanowire arrays of any organic materials with reasonable control over the density and length (Fig. 3(c)) [62]. Metal oxide nanowires can be fabricated using oxygen plasma site-selective surface modification combined with wet chemistry on polydimethylsiloxane (PDMS) surfaces with desired control over the growth position (Fig. 3(d)) [63].
Recently, it has been reported that 23 nm wide nanostructures randomly aligned as ridges on aluminium surfaces engineered by a wet etching technique exhibited both antibacterial and antiviral effects that are promising for clinical applications [71]. In the study [71], common respiratory viruses, respiratory syncytial virus (RSV) and rhinovirus (RV) have been tested on the fabricated nanoscale surfaces, and the surfaces have significantly reduced the amount of viable RSV recovered as compared with the control surfaces. The structured surfaces were more effective in preventing RV than RSV infection. In the case of RV, there was a 3−4 log reduction observed in viable viruses after 24 h, showing the ability of the nanosurfaces to reduce the potential for surface contact transmission of diseases [71]. Even though it is extremely difficult to exploit such kind of wet etching technique to nanotexture the widely used polymeric materials in the desired manner as it is hard to control and may contaminate the treated polymers [72], the study [71] provides evidence about the viricidal effect of nanostructured surfaces. Therefore, we believe that the versatile plasma fabrication approaches with controllable properties could play a vital role in fabricating similar or even better nanopatterned polymeric antiviral surfaces, and the relevant studies should be carried out immediately.
It should be noticed that the mechanism of virus reduction is still unclear. One possible mechanism can be similar to the bactericidal effect of nanostructured surfaces. We can thus infer that plasma-fabricated nanowires deposited on a polymer surface could have certain antiviral efficiency if their sizes and dimensions could match the characteristic dimensions of the viruses. Another possible mechanism is attributed to the physical adsorption of viruses on surfaces, as the process is a function of surface area and surface activity [51].
It should also be emphasized that bacterial infection is a typical complication of viral infections [73,74]. Therefore, surfaces combined with both bactericidal and viricidal effects are necessary and can be fabricated using plasma-based approaches that could result in multifunctional polymers.
There is no doubt that current advances in fabricating the desired wetting and nanopatterned polymer surfaces as antiviral materials are not sufficient and many observed effects are not yet fully understood. There is a trend and a strong need to employ versatile plasma techniques to tailor surface morphology to target viruses including the novel COVID-19 type. More specifically, the wettability control for desiccating or repelling virus-containing droplets and the nanostructured surface interactions with viruses should be one of the foci in both plasma technology and polymer science.
3.2. Plasma processing of surface chemistry
Plasma methods of processing polymer surfaces can target the morphology of the polymers and provide a convenient way to regulate the chemical composition of the surfaces, which may play a crucial role in the development of a new class of antiviral materials. Variation of the polymer surface chemistries by plasma functionalization can result in surface properties with the desired wettability and specific functional groups without altering mechanical properties.
As discussed in Section 3.1, the control of surface wettability is one of the important antiviral strategies. Targeted surface wettability can also be achieved by tailoring surface chemistry using plasma approaches. As it is known, surface hydrophilicity induced by plasma activation generally shows a hydrophobic recovery that is caused by the reorganization of chemical polar groups to minimize the surface energy.
Multi-layer plasma depositions, however, can be considered as one of the most promising strategies that can overcome such problems. Fig. 4 (a) shows the stable superhydrophilic surfaces fabricated by such multi-layer plasma depositions [75]. In [75], the hydrophobic PDMS block firstly covered by a layer of CH4 plasma coating was then overlaid by a hydrophilic SiOx layer using tetraethyl orthosilicate and oxygen (TEOS-O2) precursors. This approach provided superhydrophilic properties of the polymer surface for over 28 days. The highly cross-linked hydrocarbon layer acted as a physical barrier between the bare PDMS surface and the hydrophilic layer, which possibly hindered the reorganization and migration of chemical polar groups and resulted in the surface that remained stable over a long time.
Fig. 4.
Representative examples of plasma engineering polymers with controlled surface chemistries. (a) Superhydrophilic surfaces by plasma-polymerized TEOS-O2/CH4/PDMS with long-term stability. The inset shows WCA ~ 0° and the scale bar shows 1 µm. [75], Copyright 2012. Adapted with permission from Elsevier. (b) One-step production of rough superhydrophobic plasma coatings with WCA = 167° using He gas plasma containing 0.3 vol % benzene + cyclohexane (8:2) mixture on a flat Si wafer substrate. The scale bar shows 300 nm. [81], Copyright 2011. Adapted with permission from the American Chemical Society. (c) Bacterial proliferation in REF sample (i) and the NO release coating produced by IPNpp (ii) after 14 h incubation. Scale bars show 200 µm. [85], Copyright 2015. Adapted with permission from the Royal Society of Chemistry.
Similarly, stable superhydrophilic surfaces can also be obtained by Ar plasma post-treatment of plasma polymerized HMDSO coatings in the atmospheric-pressure roll-to-roll system that is promising to be scaled-up for industrial applications [76]. It is worth emphasizing that due to the diversities of plasma reactor configurations [77], it is promising to integrate the existing or newly designed atmospheric-pressure plasma reactor into the existing roll-to-roll industrial production line. Typical examples with the potential for mass production of the desired antiviral polymers are presented in the literature [78], [79], [80].
Recent years have also witnessed major advances in the fabrication of superhydrophobic surfaces using the plasma-assisted modification of surface chemistry. As surface wettability is determined by surface chemistry and surface microstructures, it is extremely difficult to achieve superhydrophobicity in a single-step process using conventional fabrication strategies.
Plasma polymerization, one of the most important plasma techniques is based on the fact that the formation of particles is highly dependent on the nature of the precursor monomer and occurs with the assistance of the reactive species in plasmas to induce condensation reactions in the gas phase. Therefore, it allows a one-step process for tailoring both surface chemistry and surface roughness simultaneously. Fig. 4 (b) indicates that one-step production of a rough superhydrophobic plasma coating with WCA of 167° using He gas plasma containing 0.3 vol % benzene + cyclohexane (8:2) mixture on a flat Si wafer substrate [81]. Similar superhydrophobic coatings on different substrates including gold film, Kimwipe, and cotton have also been achieved using He-CF4-H2 plasma at atmospheric-pressure [82]. In addition to these chemicals mentioned above, precursor monomers containing alkyl- or fluoroalkyl- groups that can lead to surface hydrophobicity, such as HMDSO (O[Si(CH₃)₃]₂) [83], 1H,1H,2H,2H-perfluorododecyl acrylate (C13H7F17O2) and dodecyl acrylate (C15H28O2) mixture [84] have also been investigated intensively for producing superhydrophobic surfaces by the method of plasma polymerization.
We emphasize that some of the hydrophobic surfaces tailored by plasma fabrication may not only repel the virus-containing water droplets for some antiviral applications (as described above) but also play a crucial role in inactivating viruses themselves. A previous study has demonstrated that surfaces that are simultaneously hydrophobic and oleophilic are efficient in inactivating influenza A viruses (e.g. for the surface with WCA=111° and hexadecane contact angle = 45° simultaneously, the infection degree can be reduced to below 30%) due to the disruption of the viral membrane upon its contact with the alkyl chains coated on the glass surfaces [86]. More importantly, nanostructured surfaces with the enhanced highly hydrophobic and oleophilic behaviour induced by the plasma (reactive ion) etching can increase the inactivation effects (e.g. the surface with WCA=154° and hexadecane contact angle = 26° simultaneously can lead to a dramatic reduction in the infectivity level of influenza A viruses to below 10%) [86]. Indeed, when some of the virus-containing water droplets failed to be repelled by the functionalized hydrophobic surfaces, such kind of hydrophobic “contact-killing” surfaces could play a role of “the second line of defence” to inactive the attached viruses.
Another type of the known contact-killing microbicidal surfaces functionalized by specific surface chemistry is the polymeric coatings comprising hydrophobic polycations such as branched or linear N, N-dodecyl methyl-polyethylenimines and their derivatives that have shown the capacity to kill influenza A virus with essentially a 100% efficiency (at least a 4 log reduction in the viral titer) within minutes, as well as the airborne human pathogenic bacteria Escherichia coli and Staphylococcus aureus [87]. Generally, these polyethylenimines-based coatings are hydrophobic and positively charged, when viruses owning an inherent negative charge (e.g. influenza A virus) strike the coated surface due to thermal motion, the hydrophobic and electrostatic interactions will make viruses adhere to the surface and then lead them towards inactivation by destroying their genomic content or structures [88,89]. Typically, these hydrophobic polycations are either covalently attached or deposited onto surfaces of diverse materials and regarded as non-leaching, permanently microbicidal coatings with promise for replacing these toxic and easy to be exhausted biocide release products [88]. However, fabricating these covalently modified antiviral surfaces by wet chemistry generally requires harsh reaction conditions, multi-step processes, and specific substrates [88,90].
Atmospheric-pressure aerosol-assisted plasma deposition has been proposed as a direct and substrate-independent approach to fabricate N, N-hexyl,methyl-polyethylenimine coatings for virucidal against human influenza A virus with a reduction of greater than 4 log [90]. Importantly, the coatings have shown thermal stability (up to 150°C) and durability upon exposure to vigorous washes and exposure to detergents. However, the relevant studies still remain at the early stages and a combination of both plasma-based approaches and polyethylenimines-based chemistry should be investigated and applied for more kinds of viruses beyond the influenza A virus.
Chemically active plasma-assisted coatings are proposed as another strategy in the development of antiviral polymers based on the engineering layers capable of releasing reactive oxygen and nitrogen species (RONS). Indeed, it is known that RONS play a vital role in cancer treatment [91], antibacterial [92] and virus-inactivation [93]. Therefore, active materials capable of generating RONS are considered as a new trend in virus-inactivation. Some of these materials are based on the coatings that integrate photosensitizers and photocatalysts capable of RONS generation under light-activation that results in antimicrobial efficiency with multiple advantages including high biocidal effect, broad-spectrum antimicrobial action, and low toxicity with little environmental concern [94]. Nanofibrous membranes grafted with the designed photosensitizer molecules based on benzophenones and polyphenols by wet chemistry approaches have shown the ability to release RONS such as hydroxyl radicals (•OH), superoxide (•O2−), and hydrogen peroxide (H2O2) [95]. This ability was activated by the daylight, showing contact-killing of both Gram-negative Escherichia coli O157:H7 and Gram-positive Listeria innocua bacteria (efficiency > 99.9999%) and T7 bacteriophage viruses (efficiency > 99.999%) either in aerosol or liquid form [95]. The materials are quite promising to produce PPE with the desired antiviral functions. However, such kinds of photosensitizer-based RONS agent release coatings prepared by plasma processing of surface chemistry for both antibacterial and antiviral applications are still rare and are thus worth to be intensively investigated.
In addition to these RONS virucidal agents, RONS could also be effective for stimulating immune responses against viruses in the patients [19]. Hence, RONS release coatings can be used as drug delivery systems. One important such type of RONS is nitric oxide (NO) that is a critical messenger molecule in biological systems and has been applied in biofilm inhibition and dispersal [96], vasodilatation [97], and stem cell behaviour modification [98]. More importantly, NO has shown the high efficiency of antibacterial activities [96] and antiviral effect by improving arterial oxygenation and enabling the reduction of inspired oxygen therapy and airway pressure support in patients carrying SARS or COVID-19 [99,100]. It has been reported that plasma polymerized isopentyl nitrite (IPNpp) coatings on polyethene terephthalate (PET) surfaces can be successfully used for NO release at bacteriostatic concentrations, inhibiting bacterial (Staphylococcus epidermidis) growth without cytotoxic side effects to human mesenchymal stem/stromal cells: the bacteria in the reference (REF) sample continued to multiply and form biofilms and in contrast, bacteria being in contact with the IPNpp neither multiplied nor formed any visible biofilm even after 14 h incubation (Fig. 4 (c)) [85]. Similarly, precursor monomers such as allylamine and diallylamine have also been used for plasma polymerization to load NO that enables excellent cell-surface interactions and continuous release of NO over 48 h [101]. The plasma polymerization approach allows the formation of stable amine polymers with targeted surface anchoring sites for loading the NO molecular which can avoid potentially carcinogenic amine decomposition products.
We emphasize that the antiviral mechanisms of NO are quite different from the antibacterial mechanisms. As a highly reactive free radical, NO may be a modulator of host response against numerous viruses rather than a simple antiviral agent [102,103]. A recent study has shown that acute respiratory distress, caused by the inflammatory cascade inside the lungs, is the leading cause of COVID-19 case-fatality [104]. Thus, NO may soak up cellular signals that enhance inflammation, which is an important factor for antiviral protection against the current pandemic. Despite the promising therapeutic potential of NO, the delivery of NO remains a challenge due to its reactive nature and short half-life in the biological media (typically in the order of seconds). Plasma polymerization of NO release coatings could thus be one of the viable candidates for the delivery of the NO molecules to the infected loci with the controlled release amount and release time. The relevant antiviral effects and in-depth mechanisms of the NO release coatings are thus also needed to be urgently investigated. On the other hand, it also needs to investigate the collective strategies by combining the effective antibacterial effects of NO and other antiviral strategies to prevent the spread of viruses in the environment and reduce concurrent bacterial infections caused by viral infections.
3.3. Plasma processing of composite coatings
Materials-centred approaches that impart antiviral properties onto a polymer surface are also focused on the processing of composite coatings by integrating the desired biomolecules or antiviral drugs/agents into the coating structure. Such techniques are truly versatile in enabling different surface-virus interactions. Plasma-based approaches are also known for the engineering of composite coatings in a controllable and spatially selective manner [105], which could be highly promising for the nanofabrication of antiviral polymers. Various strategies with typical examples of plasma processing of composite coatings, including surface-immobilized bioactive compounds and antiviral drug/agent release coatings, that can be applied for imparting polymers with antiviral properties are discussed below.
3.3.1. Surface-immobilized bioactive compounds
It is known that the multivalent interactions between viruses and their protein receptors on host cells such as interactions between SARS-COV-2 and angiotensin-converting enzyme 2 (ACE2) are the primary step during virus infection [106]. Therefore, protein-immobilization on the polymeric materials could be regarded as one of the antiviral strategies given the specific interactions between a particular protein and its specific binding viral target. During the last decade, multiple plasma-assisted methods of surface modification and activation have been employed to create polymers with surface functional groups that can covalently attach many types of biomolecules [107].
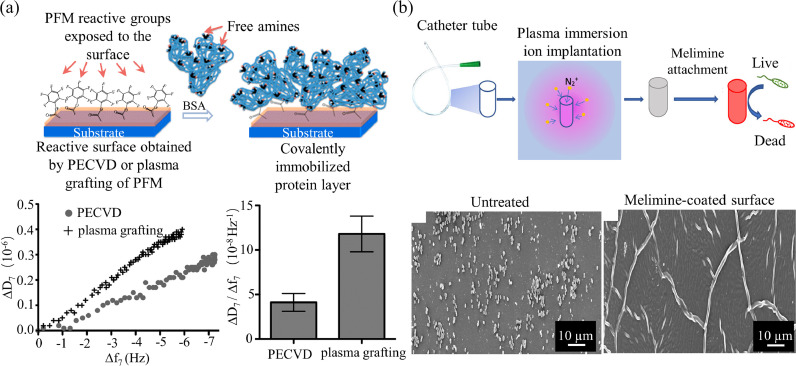
Protein-immobilized surfaces can be one of the important antiviral materials due to their specific interactions with targeted viruses. Fig. 5 (a) presents a typical example of two plasma-based approaches for protein-immobilization [108]. Both PECVD and plasma grafting using pentafluorophenyl methacrylate (PFM) as monomers were investigated to functionalize polystyrene (PS) strips surfaces with reactive ester groups. Such functional groups are highly reactive toward amines, and allow the covalent bonding of bovine serum albumin (BSA) proteins to the surfaces in different ranges (200 ng•cm–2 for PECVD, 100 ng•cm–2 for plasma grafting of PFM) [108]. Importantly, both approaches can maintain the bioactivity of the immobilized proteins despite that PECVD can result in more conformational changes than those induced by the plasma grafting approach [108]. Similarly, various monomers such as allylamine [109] and ethylenediamine [110], and various reactive gases such as oxygen [111] and ammonia [112] were also used as reactive chemicals in plasma polymerization or plasma activation processes for the immobilization of various proteins including antigens and antibodies.
Fig. 5.
Representative examples of plasma-assisted surface-immobilized bioactive compounds. (a) Two plasma-based approaches (PECVD and plasma grafting of PFM) for BSA protein-immobilization. The left plot indicated that the variation of the dissipation (ΔD) vs the frequency shift (Δf) showing a higher increase in the viscoelastic properties per mass unit attached on the grafting PFM-modified surface. The right bar graph showed the final value of the ΔD/Δf ratio confirming that the protein layer formed by PECVD was more rigid and thus had lower water content. [108], Copyright 2013. Adapted with permission from the American Chemical Society. (b) Plasma immersion ion implantation for antimicrobial peptide immobilization. The results showed that the bacteria adhesion on the covalently attached melimine-coated surface was significantly reduced. Scale bars show 10 µm. [113], Copyright 2019. Adapted with permission from the American Chemical Society.
It should be emphasized that some of the antibody-immobilization surfaces prepared by plasma technology have already been employed for virus-capture. Anti-influenza A virus hemagglutinin antibody was anchored via N-succinimidyl 3-(2-pyridyldithio) propionate on the surface of graphite-encapsulated magnetic beads functionalized by radio frequency (RF) ammonia plasma, and the infectivity of captured viruses was maintained, showing that this method is useful for the enhanced detection and isolation of influenza A virus [112]. On the other hand, a recent study has revealed that the single-domain antibodies isolated from a llama immunized with the prefusion-stabilized middle east respiratory syndrome (MERS-CoV) or SARS-CoV coronavirus spikes can be used for neutralizing MERS-CoV or SARS-CoV spike pseudo-type viruses, respectively, showing the promise for the treatment of diseases caused by coronaviruses including SARS-CoV-2 [114]. There is no doubt that polymer surfaces with such kinds of antibodies engineered by plasma technology could also be potentially used for targeting a specific virus.
The antiviral peptide immobilized surface is another important type of bioactive materials that can be applied for fighting against viral infections. Generally, antimicrobial peptides are produced by all microorganisms, plants, and animals as a part of the innate immune response, and display a broad-spectrum of antibacterial, antifungal, and antiviral activities [115,116]. Indeed, antiviral peptides play a vital role in fighting against various virus infections [117,118]. Fig. 5 (b) illustrates an example of a plasma-based approach for the immobilization of antimicrobial peptides on polymer surfaces [113]. In this example, plasma immersion ion implantation was adopted as a linker-free single-step treatment that covalently coupled antimicrobial peptides to polyvinyl chloride (PVC) catheter, showing a strong antimicrobial activity demonstrated by higher than 3 log kill of Staphylococcus aureus [113]. Similarly, oxygen plasma treatment of silicone surfaces has been regarded as a straightforward strategy to immobilize antibacterial peptides in a site-specific way with excellent antibacterial activities [119]. Surfaces functionalized by argon plasma activation followed by silanization or plasma polymerized allyl glycidyl ether coatings were also investigated in-depth for the immobilization of antimicrobial peptides [120,121]. The key point of surfaces functionalized by plasma is the specific reaction between surface functional groups and the chemical composition of targeted bioactive compounds.
Therefore, it is reasonable to transfer from antibacterial towards antiviral surfaces by the fabrication of composite coatings containing antiviral peptides for antiviral applications, as the chemical composition for antibacterial peptides is similar to that of antiviral peptides. Moreover, a recent study has revealed that two types of cellular polymeric sponges made of the derivatives from human lung epithelial type II cells or human macrophages displayed the same protein receptors, both identified and unidentified, required by SARS-CoV-2 for cell entry, and can be used as effective medical countermeasures against SARS-CoV-2 infection [122]. As the plasma-based approach is both scalable and flexible, it is promising to apply plasma technology for the mass production of such kinds of polymeric nanoparticles with bioactive compounds. Nevertheless, all such antiviral applications should be investigated in-depth both in theory and in practice.
3.3.2. Antiviral drug/agent release coatings
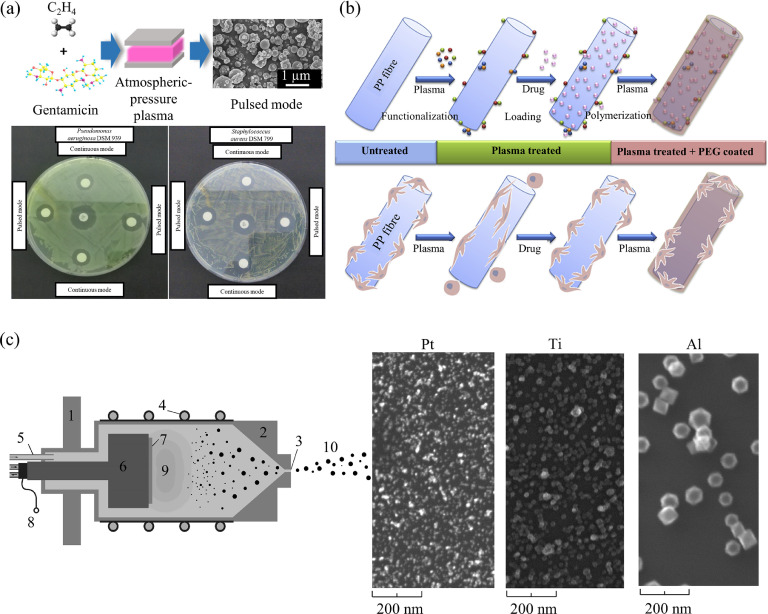
Recently, major attention has been paid to the controlled drug/agent release polymeric coatings as they are regarded as successful tools to modulate surface properties of biomaterials, namely tissue responses and fouling behaviour [123,124]. Specifically, plasma treatment is a novel and advanced method that can play a crucial role in this emerging research area [125]. Different from the aforementioned “contact-binding” strategy through plasma-enabled immobilization of bioactive compounds and virus-recognition, composite drug release coatings aim to inactivate specific viruses by releasing antiviral drugs/agents from the polymeric coatings in a controlled way. Generally, several plasma-based approaches can be applied to fabricate such coatings with specific drugs/agents, as presented in Fig. 6 .
Fig. 6.
Representative examples of plasma engineering of drug/agent release coatings. (a) Single-step aerosol-assisted plasma deposition of gentamicin-containing coatings for drug release applications. Both continuous and pulsed mode plasma-exposed disks produced antimicrobial activity against Pseudomonas aeruginosa DSM939 and Staphylococcus aureus DSM799. The scanning electron microscopy (SEM) image shows the coating morphology which was prepared in the pulse mode for 20 min with 5 slm He, 20 sccm ethylene, and 10 mg/mL gentamicin solution. The scale bar shows 1 µm. [126], Copyright 2018. Adapted with permission from the American Chemical Society. (b) Multi-steps plasma surface engineering drug-loading processes and the relevant summary of the effects of the different plasma treatments performed to PP fibres on the adsorption of drugs and on the cell adhesion and morphology. [127], Copyright 2015. Adapted with permission from Elsevier. (c) Gas-aggregation plasma sputtering system (1-vacuum flange, 2-aggregation chamber, 3-exit orifice, 4-water cooling, 5-gas inlet, 6-magnetron, 7-target, 8-electrical connection, 9-plasma, 10-beam of nanoparticles) for composite surfaces production with the capability of metal ions release and hydrophobic/superhydrophobic behaviour. Scale bars are 200 nm. [128,129]. [128], Copyright 2012. Adapted with permission from Elsevier. [129], Copyright 2014. Adapted with permission from Elsevier.
Aerosol-assisted plasma deposition represents a typical example of a process for preparing drug release coatings. As depicted in Fig. 6 (a), water containing antimicrobial drugs Gentamicin was injected in the form of an aerosol into a dielectric barrier discharge plasma through an atomizer, the ethylene gas was added as a precursor of the matrix simultaneously [126]. With the assistance of the non-thermal plasma, the drug molecules were successfully incorporated into the plasma polymers without altering their chemical structure, and the embedded drug showed excellent antibacterial effects with the controllable release [126]. This single-step procedure performed at the atmospheric-pressure is quite attractive and can be scalable to meet the industrial demands. Importantly, the aerosol-assisted plasma deposition process can keep the bioactivity of the drug molecules unchanged due to the mild fragmentation conditions as featured by the atmospheric-pressure plasma and the shield of the drug by a thin protective solvent shell that preserves its structure and functionality [126,130]. Moreover, aerosol-assisted plasma deposition also shows excellent versatility, as various drugs or bioactive molecules (including proteins and peptides mentioned above) such as Vancomycin and Lysozyme have also been investigated in-depth for biomedical applications [131], [132], [133].
Multi-steps plasma surface engineering processes, in addition to the single-step plasma process for drug release coatings, present another important strategy to achieve improved drug release performance. As indicated in Fig. 6 (b), air plasma activation of polypropylene (PP) surgical meshes were firstly adopted to increase the drug-loading capacity (nearly 3-fold higher Ampicillin loading after 3.5 s treatment), followed by polyethene glycol (PEG)-like coatings using tetraethylene glycol dimethyl ether as the precursor under a low-pressure RF plasma condition [127]. It allowed combining higher drug-loadings and maintaining essentially unchanged fibroblast properties such as chemotaxis or adhesion with respect to untreated PP meshes.
Therefore, there is no doubt that combined with the rapid development of antiviral drugs, plasma techniques could be promising for the production of antiviral drug release coatings even though relevant antiviral studies are currently rare.
Although specific reactions between viruses and antiviral drugs ensure the high efficiency of virus-inactivation, antiviral materials need to have a broad-spectrum activity to enable their binding or interacting with the mutated viral spike as viruses have always been evolving, e.g. the mutation of SARS-CoV-2 spike may potentially produce more aggressive coronaviruses [40]. Functional metal-based nanoparticles including Cu, Au, Ag, TiO2 have shown broad-spectrum antiviral activities [134,135]. A recent study has shown that Cu2O nanoparticles embedded into polymeric coatings can inactive SARS-CoV-2 with about 99.9% reduction of viral titer on average as compared with the uncoated sample [136], implicating that metal-based nanoparticles may inactivate unknown and understudied viruses given their broad virus binding spectrum. As a versatile technique, the aforementioned aerosol-assisted plasma deposition can also be applied to dope polymeric coatings with antiviral metal-based nanoparticles [137,138].
Plasma sputtering, as a recent example, has been employed in a configuration of the gas-aggregation plasma sputtering system for the production of composite surfaces with the capability of metal ions release (such as Pt, Ti and Al) and hydrophobic/superhydrophobic behaviour (Fig. 6 (c)) [128,129]. Such surfaces are expected to fight against infections due to the surface hydrophobicity/superhydrophobicity and the broad-spectrum antibacterial/antiviral activities of the nanoparticles. It should be emphasized that plasma technology can be employed to functionalize not only polymer surfaces but also metal/metal oxide nanoparticles (e.g. ZnO, Al2O3, and ZrO2 in [139]) with specific functional groups or bioactive compounds to enhance their antiviral effect. Implanting the functionalized nanoparticles into the fabricated polymeric coatings (by e.g. the gas-aggregation plasma sputtering system) can produce highly desired materials for inactivation of the targeted virus.
Considering the extreme complexity of virus inhibition and the variability of the viruses, employing plasma techniques for preparing composite antiviral materials is expected to grow as an effective antiviral material engineering strategy in the future thus forming a new trend at the interface of polymer science, plasma science, biology and medicine.
4. Future trends in smart polymers for antiviral applications
As discussed in Section 3, non-thermal plasmas generate active physical and chemical species which make them of considerable value in the production of advanced materials with antiviral properties. Further applications of such materials should be sufficiently smart to involve some of the key processes including virus-capture, virus-detection, virus-repelling, and/or virus-inactivation. To illustrate the potential utility of non-thermal plasmas, below this article highlights the selected further trends in the emerging antiviral applications that are most relevant to plasma polymer processing.
4.1. Plasma-enhanced performance of virus biosensors
Early diagnosis and management of virus spread are crucial for constraining the spread of infectious viral diseases such as COVID-19 [48,140]. Currently, real-time reverse transcription-polymerase chain reaction (RT-PCR) is the primary approach for emerging pathogen diagnosis. However, the protocol of RT-PCR involves multiple steps, and errors in any of these steps such as the DNA/RNA preparation could affect the diagnostic accuracy [141,142]. Hence, the development of advanced highly sensitive immunological diagnostic methods that directly detect viral antigens in clinical samples without sample preparation is necessary for rapid and accurate diagnosis of the emerging pathogens. Basically, immunoassays are based on highly specific antigen-antibody interactions. The key to immunological diagnostic is immobilizing bioactive antibodies on support polymeric substrates by a protein-immobilization technique to enable virus-capture and virus-detection.
As discussed in Section 3.3.1, plasma-based approaches are powerful tools for protein-immobilization that can retain the bioactivity of proteins on the test kit surfaces. Polymer surfaces functionalized by plasma could effectively enhance the virus biosensor performance by enhancing immobilization of receptor antibodies. For example, the sensitivity of a plasma functionalized biosensor for human hepatitis B virus (HBV) is improved as much as 1000-fold in comparison with a commercial one [143]. Although relevant in-depth studies and commercial devices are rare, it is promising to develop new viral biosensors with enhanced performance for detecting emerging pathogens using similar plasma functionalization protocols [144,145]. Therefore, the development of novel viral biosensors by plasma surface functionalization is one of the important trends in antiviral applications. The key attention should be on the relationships between the plasma treatment and the performance of the biosensors for future scalable production and commercial devices.
4.2. Plasma-enabled new sanitizing approaches
Clean air and common surfaces such as food packages, PPE, and building elements without pathogens are essential for human health. Wet chemical solutions containing alcohol or hypochlorite are often used to mitigate pathogens in the air or on these common surfaces through spraying or wiping. However, the sanitizing approach is labour- and materials-intensive, impractical for covering all exposed areas, and needs to be periodically reapplied. In addition, it may cause environmental pollution and then, in turn, negatively influence human health. Therefore, new sanitizing approaches are required for creating a healthier living and working environment.
One possible sanitizing approach is to collect virion-laden water droplets in the air using plasma engineered hydrophilic-hydrophobic patterned polymers by taking advantage of the capacity of water-collection [146], and then one can use physical methods such as ultraviolet (UV) light, sunlight, and cold plasma itself to inactivate the viruses [93,147]. It should be emphasized that the UV sterilization can also be based on the utilization of cold plasmas, with typical examples of the excimer- and Hg-lamps [148,149]. The challenge for such kind of sanitizing approach is the unknown efficiency of the polymers collecting virion-laden water droplets in the air that needs to be precisely quantified.
Self-sanitizing surfaces are ideal alternatives to the conventional sanitizing approaches. Plasma engineered polymers with photocatalytic self-cleaning [150], [151], [152] and/or water-repelling [153] properties can be among the effective self-sanitizing surfaces. It is necessary to pinpoint that the photocatalytic self-cleaning and superhydrophobic water-repelling surfaces could be able to not only repel the virion-laden respiratory droplets but also remove the deactivated or decomposed products. Typical examples of such bi-functional self-sanitizing surfaces can be achieved by combining the superhydrophobic water-repelling polymer coatings with TiO2 nanoparticles [154]. Moreover, plasma engineered superhydrophilic polymers could be useful for self-sanitizing applications to some extent as they have theoretically shown the potential in reducing the risk of getting COVID-19 infection by reducing the dry time of the water droplets on them [52]. Another type of self-sanitizing surfaces can be plasma engineered chemically active coatings capable of RONS release and composite coatings capable of binding viruses or releasing antiviral drugs/agents [155], [156], [157], as described in Section 3.2 and Section 3.3, respectively. Such self-sanitizing coatings should be durable against rubbing and washing, long-lasting, and nontoxic, which represent challenges for future studies.
4.3. Future antiviral polymers for future viruses
As mentioned above, viruses evolve naturally, thus becoming more aggressive to infect and/or more effective to kill. During the past 20 years, various types of coronavirus including SARS-CoV, MERS-CoV, and SARS-CoV-2 have caused epidemics or pandemics, with ACE2 being the primary receptor of SARS-CoV and SARS-CoV-2 [1], and CD26 mediating the entry of MERS-CoV [158]. These together with other receptors such as TMPRSS2 [159], [160], [161] that activate the spike protein of SARS-CoV, MERS-CoV, and SARS-CoV-2, and CD209L [162] that mediates the entry of SARS-CoV, Ebola and Sindbis, form the primary pool to predict the emerging highly prevalent pathologic viruses (especially coronaviruses). The high variety of the receptors and the mechanisms that lead to the entry of viruses into recipients pose a great challenge that complicates the development of specific drugs and vaccines for efficient viral disease control including COVID-19.
Although drugs/vaccines against viral infection and multiplication are the most efficient and canonical approaches for viral disease management, their development is quite time-consuming and has a high risk to fail. The complexity of the specific drug development is also related to the fast evolution of the virus structure that can challenge the efficacy of medical treatment. SARS-CoV-2 has mutated from type D614 to type G614 that is the current dominant type in almost every nation and region affected by the COVID-19 pandemic [40].
To tackle challenges in the establishment of antiviral strategies against emerging viruses in the future, and challenges in the development of novel drugs as well as antiviral polymer surfaces, new approaches and materials with broad-spectrum properties are required [163]. Through computationally modelling virus-host receptor interactions and identifying key factors affecting their receptor binding affinities, one could predict the structure of potential viruses with enhanced transmission ability and corresponding antibodies or antiviral drugs to prevent their potential spread. On the other hand, an integrated pipeline or platform from initial computational predictions of virus-host interactions to downstream polymer fabrications including the plasma-assisted approach should be established for preventing emerging pandemics, which is of fundamental significance to the public health and socio-economic development.
5. Conclusions and outlook
The unpredictable nature of viral diseases and their impacts on the global economy and society pose a major challenge. Despite being recognized as one of the most devastating global problems, studies on the prevention and treatment of emerging viral diseases are sparse. The goal of this article is to provide a critical analysis of the recent progress in the field of plasma surface engineering for developing future polymer based antiviral surfaces. Several current and emerging trends have been emphasized in the areas of polymer science and surface engineering technology based on the use of plasma processing methods.
Impressive recent advances in plasma functionalization of polymer surfaces suggest that plasma-assisted surface functionalization approaches are promising for the production of antiviral polymers with targeted antiviral applications ranging from in-vitro prevention to in-vivo therapy. The strategy of antiviral polymers engineered by plasma processing such as biosensors and virus destruction could be established in advance to respond to the next generation of viruses and evolve faster than any emerging human viral pathogens in the future.
Ideally, the antiviral polymers for disease diagnosis, prevention and therapeutics should be of a broad-spectrum activity against multiple viruses and have multiple functionalities, i.e., in the form of a mixture of multiple types of plasma engineered polymers that could be used for biosensing, disinfection and decontamination. Importantly, new approaches in antiviral polymers development take advantage of computational predictions and are not limited to any particular type of viruses to cope with the fast evolution of viruses in practice. Considering major achievements in the manufacturing of novel antibacterial surfaces using plasma surface engineering the hope in a fast translation of the available approaches for fighting against viruses is arguably high. However, practical implementation of the highlighted trends will require a highly-coordinated cross-disciplinary collaboration involving researchers and engineers from the areas spanning virology, bioinformatics, polymer science, and plasma surface engineering.
CRediT authorship contribution statement
Chuanlong Ma: Data curation, Formal analysis, Visualization, Writing - original draft, Writing - review & editing. Anton Nikiforov: Writing - original draft, Writing - review & editing. Nathalie De Geyter: Project administration, Resources, Writing - review & editing, Supervision. Xiaofeng Dai: Methodology, Writing - review & editing, Supervision. Rino Morent: Project administration, Resources, Writing - review & editing, Supervision. Kostya (Ken) Ostrikov: Conceptualization, Methodology, Writing - review & editing, Project administration, Supervision.
Declaration of Competing Interest
The authors declare no competing interests.
Acknowledgements
C. Ma thanks China Scholarship Council (No.201807090104). A. Nikiforov thanks for partial support of the work by Vlaio project HBC.2019.0157. X.F. Dai was funded by the National Natural Science Foundation of China (Grant No. 81972789), National Science and Technology Major project (Grant No. 2018ZX10302205-004-002), and Fundamental Research Funds for the Central Universities (Grant No. JUSRP22011). K. Ostrikov thanks the Australian Research Council (ARC) and QUT Centre for Materials Science for partial support.
References
- 1.Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cucinotta D, Vanelli M. WHO declares COVID-19 a pandemic. Acta Biomed. 2020;91:157–160. doi: 10.23750/abm.v91i1.9397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gorbalenya AE, Baker SC, Baric RS, de Groot RJ, Drosten C, Gulyaeva AA, et al. The species severe acute respiratory syndrome-related coronavirus: Classifying 2019-nCoV and naming it SARS-CoV-2. Nat Microbiol. 2020;5:536–544. doi: 10.1038/s41564-020-0695-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huang H, Fan C, Li M, Nie HL, Wang FB, Wang H, et al. COVID-19: A call for physical scientists and engineers. ACS Nano. 2020;14:3747–3754. doi: 10.1021/acsnano.0c02618. [DOI] [PubMed] [Google Scholar]
- 5.Liu Y, Ning Z, Chen Y, Guo M, Liu Y, Gali NK, Sun L, et al. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature. 2020;582:557–560. doi: 10.1038/s41586-020-2271-3. [DOI] [PubMed] [Google Scholar]
- 6.Chia PY, Coleman KK, Tan YK, Ong SWX, Gum M, Lau SK, et al. Detection of air and surface contamination by SARS-CoV-2 in hospital rooms of infected patients. Nat Commun. 2020;11:2800. doi: 10.1038/s41467-020-16670-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ong SWX, Tan YK, Chia PY, Lee TH, Ng OT, Wong MSY, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA. 2020;323:1610–1612. doi: 10.1001/jama.2020.3227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, et al. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020;323:1061–1069. doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.van Doremalen N, Bushmaker T, Morris DH, Holbrook MG, Gamble A, Williamson BN, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med. 2020;382:1564–1567. doi: 10.1056/NEJMc2004973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.van Doremalen N, Bushmaker T, Munster VJ. Stability of middle east respiratory syndrome coronavirus (MERS-CoV) under different environmental conditions. Eurosurveillance. 2013;18:20590. doi: 10.2807/1560-7917.es2013.18.38.20590. [DOI] [PubMed] [Google Scholar]
- 11.Nguyen LH, Drew DA, Graham MS, Joshi AD, Guo CG, Ma W, et al. Risk of COVID-19 among front-line health-care workers and the general community: A prospective cohort study. Lancet Public Heal. 2020;5:e475–e483. doi: 10.1016/S2468-2667(20)30164-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hasan J, Crawford RJ, Ivanova EP. Antibacterial surfaces: The quest for a new generation of biomaterials. Trends Biotechnol. 2013;31:295–304. doi: 10.1016/j.tibtech.2013.01.017. [DOI] [PubMed] [Google Scholar]
- 13.Song B, Zhang E, Han X, Zhu H, Shi Y, Cao Z. Engineering and application perspectives on designing an antimicrobial surface. ACS Appl Mater Interfaces. 2020;12:21330–21341. doi: 10.1021/acsami.9b19992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Muñoz-Bonilla A, Fernández-García M. Polymeric materials with antimicrobial activity. Prog Polym Sci. 2012;37:281–339. [Google Scholar]
- 15.Cloutier M, Mantovani D, Rosei F. Antibacterial coatings: Challenges, perspectives, and opportunities. Trends Biotechnol. 2015;33:637–652. doi: 10.1016/j.tibtech.2015.09.002. [DOI] [PubMed] [Google Scholar]
- 16.Ding X, Duan S, Ding X, Liu R, Xu FJ. Versatile antibacterial materials: An emerging arsenal for combatting bacterial pathogens. Adv Funct Mater. 2018;28 [Google Scholar]
- 17.Imani SM, Ladouceur L, Marshall T, Maclachlan R, Soleymani L, Didar TF. Antimicrobial nanomaterials and coatings: Current mechanisms and future perspectives to control the spread of viruses including SARS-CoV-2. ACS Nano. 2020;14:12341–12369. doi: 10.1021/acsnano.0c05937. [DOI] [PubMed] [Google Scholar]
- 18.Zhou J, Hu Z, Zabihi F, Chen Z, Zhu M. Progress and perspective of antiviral protective material. Adv Fiber Mater. 2020;2:123–139. doi: 10.1007/s42765-020-00047-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Garren MR, Ashcraft M, Qian Y, Douglass M, Risbois EJ, Handa H. Nitric oxide and viral infection: Recent developments in antiviral therapies and platforms. Appl Mater Today. 2021;22 doi: 10.1016/j.apmt.2020.100887. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Weiss C, Carriere M, Fusco L, Fusco L, Capua I, Regla-Nava JA, Pasquali M, et al. Toward nanotechnology-enabled approaches against the COVID-19 pandemic. ACS Nano. 2020;14:6383–6406. doi: 10.1021/acsnano.0c03697. [DOI] [PubMed] [Google Scholar]
- 21.Bianculli RH, Mase JD, Schulz MD. Antiviral polymers: Past approaches and future possibilities. Macromolecules. 2020;53:9158–9186. [Google Scholar]
- 22.Vesel A, Mozetic M. New developments in surface functionalization of polymers using controlled plasma treatments. J Phys D Appl Phys. 2017;50 [Google Scholar]
- 23.Khelifa F, Ershov S, Habibi Y, Snyders R, Dubois P. Free-radical-induced grafting from plasma polymer surfaces. Chem Rev. 2016;116:3975–4005. doi: 10.1021/acs.chemrev.5b00634. [DOI] [PubMed] [Google Scholar]
- 24.Múgica-Vidal R, Sainz-García E, Álvarez-Ordóñez A, Prieto M, González-Raurich M, López M, et al. Production of antibacterial coatings through atmospheric pressure plasma: A promising alternative for combatting biofilms in the food industry. Food Bioprocess Technol. 2019;12:1251–1263. [Google Scholar]
- 25.Vasilev K, Griesser SS, Griesser HJ. Antibacterial surfaces and coatings produced by plasma techniques. Plasma Process Polym. 2011;8:1010–1023. [Google Scholar]
- 26.Desmet T, Morent R, De Geyter N, Leys C, Schacht E, Dubruel P. Nonthermal plasma technology as a versatile strategy for polymeric biomaterials surface modification: A review. Biomacromolecules. 2009;10:2351–2378. doi: 10.1021/bm900186s. [DOI] [PubMed] [Google Scholar]
- 27.Sarakinos K, Alami J, Konstantinidis S. High power pulsed magnetron sputtering: A review on scientific and engineering state of the art. Surf Coat Technol. 2010;204:1661–1684. [Google Scholar]
- 28.He B, Yang Y, Yuen MF, Chen XF, Lee CS, Zhang WJ. Vertical nanostructure arrays by plasma etching for applications in biology, energy, and electronics. Nano Today. 2013;8:265–289. [Google Scholar]
- 29.Nikiforov A, Deng X, Xiong Q, Cvelbar U, DeGeyter N, Morent R, et al. Non-thermal plasma technology for the development of antimicrobial surfaces: A review. J Phys D Appl Phys. 2016;49 [Google Scholar]
- 30.Ostrikov K. Colloquium: Reactive plasmas as a versatile nanofabrication tool. Rev Mod Phys. 2005;77:489–511. [Google Scholar]
- 31.Ostrikov K, Neyts EC, Meyyappan M. Plasma nanoscience: From nano-solids in plasmas to nano-plasmas in solids. Adv Phys. 2013;62:113–224. [Google Scholar]
- 32.Černák M, Kováčik D, Ráhel’ J, St'ahel P, Zahoranová A, Kubincová J, et al. Generation of a high-density highly non-equilibrium air plasma for high-speed large-area flat surface processing. Plasma Phys Control Fusion. 2011;53 [Google Scholar]
- 33.Petlin DG, Tverdokhlebov SI, Anissimov YG. Plasma treatment as an efficient tool for controlled drug release from polymeric materials: A review. J Control Release. 2017;266:57–74. doi: 10.1016/j.jconrel.2017.09.023. [DOI] [PubMed] [Google Scholar]
- 34.Morent R, De Geyter N, Verschuren J, De Clerck K, Kiekens P, Leys C. Non-thermal plasma treatment of textiles. Surf Coat Technol. 2008;202:3427–3449. [Google Scholar]
- 35.Morent R, De Geyter N, Desmet T, Dubruel P, Leys C. Plasma surface modification of biodegradable polymers: A review. Plasma Process Polym. 2011;8:171–190. [Google Scholar]
- 36.Bazaka K, Jacob MV, Chrzanowski W, Ostrikov K. Anti-bacterial surfaces: Natural agents, mechanisms of action, and plasma surface modification. RSC Adv. 2015;5:48739–48759. [Google Scholar]
- 37.Hou YJ, Chiba S, Halfmann P, Ehre C, Kuroda M, Dinnon KH, Leist SR, et al. SARS-CoV-2 D614G variant exhibits efficient replication ex vivo and transmission in vivo. Science. 2020;370:1464–1468. doi: 10.1126/science.abe8499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen J, Wang R, Wang M, Wei GW. Mutations strengthened SARS-CoV-2 infectivity. J Mol Biol. 2020;432:5212–5226. doi: 10.1016/j.jmb.2020.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kim S, Lee JH, Lee S, Shim S, Nguyen TT, Hwang J, Kim H, al Et. The progression of SARS coronavirus 2 (SARS-CoV2): Mutation in the receptor binding domain of spike gene. Immune Netw. 2020;20:e41. doi: 10.4110/in.2020.20.e41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Korber B, Fischer WM, Gnanakaran S, Yoon H, Theiler J, Abfalterer W, Hengartner N, et al. Tracking changes in SARS-CoV-2 spike: Evidence that D614G increases infectivity of the COVID-19 virus. Cell. 2020;182:812–827. doi: 10.1016/j.cell.2020.06.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Mozetič M, Primc G, Vesel A, Zaplotnik R, Modic M, Junkar I, Recek N, et al. Application of extremely non-equilibrium plasmas in the processing of nano and biomedical materials. Plasma Sources Sci Technol. 2015;24 [Google Scholar]
- 42.Wohlfart E, Fernández-Blázquez JP, Knoche E, Bello A, Pérez E, Arzt E, et al. Nanofibrillar patterns by plasma etching: The influence of polymer crystallinity and orientation in surface morphology. Macromolecules. 2010;43:9908–9917. [Google Scholar]
- 43.Rani KV, Sarma B, Sarma A. Plasma sputtering process of copper on polyester/silk blended fabrics for preparation of multifunctional properties. Vacuum. 2017;146:206–215. [Google Scholar]
- 44.Wang BB, Zhu MK, Ostrikov K, Levchenko I, Keidar M, Shao RW, Zheng K, et al. Conversion of vertically-aligned boron nitride nanowalls to photoluminescent CN compound nanorods: Efficient composition and morphology control via plasma technique. Carbon. 2016;109:352–362. [Google Scholar]
- 45.Weber TP, Stilianakis NI. Inactivation of influenza A viruses in the environment and modes of transmission: A critical review. J Infect. 2008;57:361–373. doi: 10.1016/j.jinf.2008.08.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Rubino I, Choi HJ. Respiratory protection against pandemic and epidemic diseases. Trends Biotechnol. 2017;35:907–910. doi: 10.1016/j.tibtech.2017.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Leung NHL, Chu DKW, Shiu EYC, Chan KH, McDevitt JJ, Hau BJP, Yen HL, et al. Respiratory virus shedding in exhaled breath and efficacy of face masks. Nat Med. 2020;26:678–680. doi: 10.1038/s41591-020-0843-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Prather KA, Wang CC, Schooley RT. Reducing transmission of SARS-CoV-2. Science. 2020;368:1422–1424. doi: 10.1126/science.abc6197. [DOI] [PubMed] [Google Scholar]
- 49.Ma H, Hsiao BS, Chu B. Functionalized electrospun nanofibrous microfiltration membranes for removal of bacteria and viruses. J Memb Sci. 2014;452:446–452. [Google Scholar]
- 50.Casanova L, Rutala WA, Weber DJ, Sobsey MD. Survival of surrogate coronaviruses in water. Water Res. 2009;43:1893–1898. doi: 10.1016/j.watres.2009.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Maguire-Boyle SJ, Liga M V, Li Q, Barron AR. Alumoxane/ferroxane nanoparticles for the removal of viral pathogens: The importance of surface functionality to nanoparticle activity. Nanoscale. 2012;4:5627–5632. doi: 10.1039/c2nr31117h. [DOI] [PubMed] [Google Scholar]
- 52.Bhardwaj R, Agrawal A. Tailoring surface wettability to reduce chances of infection of COVID-19 by a respiratory droplet and to improve the effectiveness of personal protection equipment. Phys Fluids. 2020;32 doi: 10.1063/5.0020249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cox CS. Roles of water molecules in bacteria and viruses. Orig Life Evol Biosph. 1993;23:29–36. doi: 10.1007/BF01581988. [DOI] [PubMed] [Google Scholar]
- 54.Palumbo F, Di Mundo R, Cappelluti D, d’Agostino R. SuperHydrophobic and superHydrophilic polycarbonate by tailoring chemistry and nano-texture with plasma processing. Plasma Process Polym. 2011;8:118–126. [Google Scholar]
- 55.Meguid SA, Elzaabalawy A. Potential of combating transmission of COVID-19 using novel self-cleaning superhydrophobic surfaces: Part I—protection strategies against fomites. Int J Mech Mater Des. 2020;16:423–431. doi: 10.1007/s10999-020-09513-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Galante AJ, Haghanifar S, Romanowski EG, Shanks RMQ, Leu PW. Superhemophobic and antivirofouling coating for mechanically durable and wash-stable medical textiles. ACS Appl Mater Interfaces. 2020;12:22120–22128. doi: 10.1021/acsami.9b23058. [DOI] [PubMed] [Google Scholar]
- 57.Cassie ABD, Baxter S. Wettability of porous surfaces. Trans Faraday Soc. 1944;40:546–551. [Google Scholar]
- 58.Zhang X, Zhu W, He G, Zhang P, Zhang Z, Parkin IP. Flexible and mechanically robust superhydrophobic silicone surfaces with stable Cassie-Baxter state. J Mater Chem A. 2016;4:14180–14186. [Google Scholar]
- 59.Zhong H, Zhu Z, Lin J, Cheung CF, Lu VL, Yan F, et al. Reusable and recyclable graphene masks with outstanding superhydrophobic and photothermal performances. ACS Nano. 2020;14:6213–6221. doi: 10.1021/acsnano.0c02250. [DOI] [PubMed] [Google Scholar]
- 60.Di Mundo R, Bottiglione F, Carbone G. Cassie state robustness of plasma generated randomly nano-rough surfaces. Appl Surf Sci. 2014;316:324–332. [Google Scholar]
- 61.Dimitrakellis P, Gogolides E. Hydrophobic and superhydrophobic surfaces fabricated using atmospheric pressure cold plasma technology: A review. Adv Colloid Interface Sci. 2018;254:1–21. doi: 10.1016/j.cis.2018.03.009. [DOI] [PubMed] [Google Scholar]
- 62.Fang H, Wu W, Song J, Wang ZL. Controlled growth of aligned polymer nanowires. J Phys Chem C. 2009;113:16571–16574. [Google Scholar]
- 63.George A, Stawski TM, Unnikrishnan S, Veldhuis SA, ten Elshof JE. Micro and nanopatterning of functional materials on flexible plastic substrates via site-selective surface modification using oxygen plasma. J Mater Chem. 2012;22:328–332. [Google Scholar]
- 64.Liu M, Wang S, Jiang L. Nature-inspired superwettability systems. Nat Rev Mater. 2017;2 [Google Scholar]
- 65.Bandara CD, Singh S, Afara IO, Wolff A, Tesfamichael T, Ostrikov K, et al. Bactericidal effects of natural nanotopography of dragonfly wing on Escherichia coli. ACS Appl Mater Interfaces. 2017;9:6746–6760. doi: 10.1021/acsami.6b13666. [DOI] [PubMed] [Google Scholar]
- 66.Vassallo E, Pedroni M, Silvetti T, Morandi S, Toffolatti S, Angella G, et al. Bactericidal performance of nanostructured surfaces by fluorocarbon plasma. Mater Sci Eng C. 2017;80:117–121. doi: 10.1016/j.msec.2017.05.111. [DOI] [PubMed] [Google Scholar]
- 67.Mo S, Mehrjou B, Tang K, Wang H, Huo K, Qasim AM, et al. Dimensional-dependent antibacterial behavior on bioactive micro/nano polyetheretherketone (PEEK) arrays. Chem Eng J. 2020;392 [Google Scholar]
- 68.Bandara CD, Ballerin G, Leppänen M, Tesfamichael T, Ostrikov KK, Whitchurch CB. Resolving bio-nano interactions of E.coli bacteria-dragonfly wing interface with helium ion and 3D-structured illumination microscopy to understand bacterial death on nanotopography. ACS Biomater Sci Eng. 2020;6:3925–3932. doi: 10.1021/acsbiomaterials.9b01973. [DOI] [PubMed] [Google Scholar]
- 69.Modaresifar K, Azizian S, Ganjian M, Fratila-Apachitei LE, Zadpoor AA. Bactericidal effects of nanopatterns: A systematic review. Acta Biomater. 2019;83:29–36. doi: 10.1016/j.actbio.2018.09.059. [DOI] [PubMed] [Google Scholar]
- 70.Yun J, Jeong Y, Lee GH. Direct synthesis of silicon oxide nanowires on organic polymer substrates. Nanotechnology. 2009;20 doi: 10.1088/0957-4484/20/36/365606. [DOI] [PubMed] [Google Scholar]
- 71.Hasan J, Xu Y, Yarlagadda T, Schuetz M, Spann K, Yarlagadda PKDV. Antiviral and antibacterial nanostructured surfaces with excellent mechanical properties for hospital applications. ACS Biomater Sci Eng. 2020;6:3608–3618. doi: 10.1021/acsbiomaterials.0c00348. [DOI] [PubMed] [Google Scholar]
- 72.Puliyalil H, Cvelbar U. Selective plasma etching of polymeric substrates for advanced applications. Nanomaterials. 2016;6 doi: 10.3390/nano6060108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rothberg MB, Haessler SD, Brown RB. Complications of viral influenza. Am J Med. 2008;121:258–264. doi: 10.1016/j.amjmed.2007.10.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhou P, Liu Z, Chen Y, Xiao Y, Huang X, Fan XG. Bacterial and fungal infections in COVID-19 patients: A matter of concern. Infect Control Hosp Epidemiol. 2020;41:1124–1125. doi: 10.1017/ice.2020.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Lee D, Yang S. Surface modification of PDMS by atmospheric-pressure plasma-enhanced chemical vapor deposition and analysis of long-lasting surface hydrophilicity. Sensors Actuators, B Chem. 2012;162:425–434. [Google Scholar]
- 76.Ma C, Wang L, Nikiforov A, Onyshchenko Y, Cools P, Ostrikov K, et al. Atmospheric-pressure plasma assisted engineering of polymer surfaces: From high hydrophobicity to superhydrophilicity. Appl Surf Sci. 2021;535 [Google Scholar]
- 77.Tendero C, Tixier C, Tristant P, Desmaison J, Leprince P. Atmospheric pressure plasmas: A review. Spectrochim Acta Part B At Spectrosc. 2006;61:2–30. [Google Scholar]
- 78.Starostin SA, Creatore M, Bouwstra JB, van de Sanden MCM, de Vries HW. Towards roll-to-roll deposition of high quality moisture barrier films on polymers by atmospheric pressure plasma assisted process. Plasma Process Polym. 2015;12:545–554. [Google Scholar]
- 79.Zhang H, Li H, Fang M, Wang Z, Sang L, Yang L, et al. Roll-to-roll DBD plasma pretreated polyethylene web for enhancement of Al coating adhesion and barrier property. Appl Surf Sci. 2016;388:539–545. [Google Scholar]
- 80.Moreno-Couranjou M, Mauchauffé R, Bonot S, Detrembleur C, Choquet P. Anti-biofouling and antibacterial surfaces via a multicomponent coating deposited from an up-scalable atmospheric-pressure plasma-assisted CVD process. J Mater Chem B. 2018;6:614–623. doi: 10.1039/c7tb02473h. [DOI] [PubMed] [Google Scholar]
- 81.Lee SH, Dilworth ZR, Hsiao E, Barnette AL, Marino M, Kim JH, et al. One-step production of superhydrophobic coatings on flat substrates via atmospheric RF plasma process using non-fluorinated hydrocarbons. ACS Appl Mater Interfaces. 2011:476–481. doi: 10.1021/am101052z. [DOI] [PubMed] [Google Scholar]
- 82.Kim SH, Kim JH, Kang BK, Uhm HS. Superhydrophobic CFx coating via in-line atmospheric RF plasma of He−CF4−H2. Langmuir. 2005;21:12213–12217. doi: 10.1021/la0521948. [DOI] [PubMed] [Google Scholar]
- 83.Nwankire CE, Favaro G, Duong QH, Dowling DP. Enhancing the mechanical properties of superhydrophobic atmospheric pressure plasma deposited siloxane coatings. Plasma Process Polym. 2011;8:305–315. [Google Scholar]
- 84.Mertz G, Delmée M, Bardon J, Martin A, Ruch D, Fouquet T, et al. Atmospheric pressure plasma co-polymerization of two acrylate precursors: Toward the control of wetting properties. Plasma Process Polym. 2018;15 [Google Scholar]
- 85.Michl TD, Coad BR, Doran M, Osiecki M, Kafshgari MH, Voelcker NH, et al. Nitric oxide releasing plasma polymer coating with bacteriostatic properties and no cytotoxic side effects. Chem Commun. 2015;51:7058–7060. doi: 10.1039/c5cc01722j. [DOI] [PubMed] [Google Scholar]
- 86.Mannelli I, Reigada R, Suárez I, Janner D, Carrilero A, Mazumder P, et al. Functionalized surfaces with tailored wettability determine influenza A infectivity. ACS Appl Mater Interfaces. 2016;8:15050–15066. doi: 10.1021/acsami.6b02779. [DOI] [PubMed] [Google Scholar]
- 87.Haldar J, An D, de Cienfuegos LA, Chen J, Klibanov AM. Polymeric coatings that inactivate both influenza virus and pathogenic bacteria. Proc Natl Acad Sci U S A. 2006;103:17667–17671. doi: 10.1073/pnas.0608803103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Klibanov AM. Permanently microbicidal materials coatings. J Mater Chem. 2007;17:2479–2482. [Google Scholar]
- 89.Hsu BB, Wong SY, Hammond PT, Chen J, Klibanov AM. Mechanism of inactivation of influenza viruses by immobilized hydrophobic polycations. Proc Natl Acad Sci U S A. 2011;208:61–66. doi: 10.1073/pnas.1017012108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Liu H, Kim Y, Mello K, Lovaasen J, Shah A, Rice N, et al. Aerosol-assisted plasma deposition of hydrophobic polycations makes surfaces highly antimicrobial. Appl Biochem Biotechnol. 2014;172:1254–1264. doi: 10.1007/s12010-013-0593-4. [DOI] [PubMed] [Google Scholar]
- 91.Yang B, Chen Y, Shi J. Reactive oxygen species (ROS)-based nanomedicine. Chem Rev. 2019;119:4881–4985. doi: 10.1021/acs.chemrev.8b00626. [DOI] [PubMed] [Google Scholar]
- 92.Fang FC. Antimicrobial reactive oxygen and nitrogen species: Concepts and controversies. Nat Rev Microbiol. 2004;2:820–832. doi: 10.1038/nrmicro1004. [DOI] [PubMed] [Google Scholar]
- 93.Filipić A, Gutierrez-Aguirre I, Primc G, Mozetič M, Dobnik D. Cold plasma, a new hope in the field of virus inactivation. Trends Biotechnol. 2020;38:1278–1291. doi: 10.1016/j.tibtech.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Zhuo J. In: Antimicrobial Textiles. Sun G, editor. Woodhead Publishing; Cambridge: 2006. Photoactive chemicals for antimicrobial textiles; pp. 197–223. [Google Scholar]
- 95.Si Y, Zhang Z, Wu W, Fu Q, Huang K, Nitin N, et al. Daylight-driven rechargeable antibacterial and antiviral nanofibrous membranes for bioprotective applications. Sci Adv. 2018;4 doi: 10.1126/sciadv.aar5931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Hetrick EM, Schoenfisch MH. Reducing implant-related infections: Active release strategies. Chem Soc Rev. 2006;35:780–789. doi: 10.1039/b515219b. [DOI] [PubMed] [Google Scholar]
- 97.Wang Q, Akhavan B, Jing F, Cheng D, Sun H, Xie D, et al. Catalytic formation of nitric oxide mediated by Ti-Cu coatings provides multifunctional interfaces for cardiovascular applications. Adv Mater Interfaces. 2018;5 [Google Scholar]
- 98.Wang W, Lee Y, Lee CH. Effects of nitric oxide on stem cell therapy. Biotechnol Adv. 2015;33:1685–1696. doi: 10.1016/j.biotechadv.2015.09.004. [DOI] [PubMed] [Google Scholar]
- 99.Chen L, Liu P, Gao H, Sun B, Chao D, Wang F, et al. Inhalation of nitric oxide in the treatment of severe acute respiratory syndrome: A rescue trial in beijing. Clin Infect Dis. 2004;39:1531–1535. doi: 10.1086/425357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Parikh R, Wilson C, Weinberg J, Gavin D, Murphy J, Reardon CC. Inhaled nitric oxide treatment in spontaneously breathing COVID-19 patients. Ther Adv Respir Dis. 2020;14:1–3. doi: 10.1177/1753466620933510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Ho KKK, Ozcelik B, Willcox MDP, Thissen H, Kumar N. Facile solvent-free fabrication of nitric oxide (NO)-releasing coatings for prevention of biofilm formation. Chem Commun. 2017;53:6488–6491. doi: 10.1039/c7cc02772a. [DOI] [PubMed] [Google Scholar]
- 102.Akaike T, Maeda H. Nitric oxide and virus infection. Immunology. 2000;101:300–308. doi: 10.1046/j.1365-2567.2000.00142.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Abdul-Cader MS, Amarasinghe A, Abdul-Careem MF. Activation of toll-like receptor signaling pathways leading to nitric oxide-mediated antiviral responses. Arch Virol. 2016;161:2075–2086. doi: 10.1007/s00705-016-2904-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Aouba A, Baldolli A, Geffray L, Verdon R, Bergot E, Martin-Silva N, et al. Targeting the inflammatory cascade with anakinra in moderate to severe COVID-19 pneumonia: Case series. Ann Rheum Dis. 2020;79:1381–1382. doi: 10.1136/annrheumdis-2020-217706. [DOI] [PubMed] [Google Scholar]
- 105.Sardella E, Palumbo F, Camporeale G, Favia P. Non-equilibrium plasma processing for the preparation of antibacterial surfaces. Materials. 2016;9 doi: 10.3390/ma9070515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Zhang H, Penninger JM, Li Y, Zhong N, Slutsky AS. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: Molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46:586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Goddard JM, Hotchkiss JH. Polymer surface modification for the attachment of bioactive compounds. Prog Polym Sci. 2007;32:698–725. [Google Scholar]
- 108.Cifuentes A, Borrós S. Comparison of two different plasma surface-modification techniques for the covalent immobilization of protein monolayers. Langmuir. 2013;29:6645–6651. doi: 10.1021/la400597e. [DOI] [PubMed] [Google Scholar]
- 109.Basarir F, Cuong N, Song WK, Yoon TH. Surface modification via plasma polymerization of allylamine for antibody immobilization. Macromol Symp. 2007;249:61–66. [Google Scholar]
- 110.Kim J, Park H, Jung D, Kim S. Protein immobilization on plasma-polymerized ethylenediamine-coated glass slides. Anal Biochem. 2003;313:41–45. doi: 10.1016/s0003-2697(02)00563-8. [DOI] [PubMed] [Google Scholar]
- 111.Grimaldi IA, Testa G, Persichetti G, Loffredo F, Villani F. Plasma functionalization procedure for antibody immobilization for SU-8 based sensor. Biosens Bioelectron. 2016;86:827–833. doi: 10.1016/j.bios.2016.07.090. [DOI] [PubMed] [Google Scholar]
- 112.Sakudo A, Chou H, Ikuta K, Nagatsu M. Integration of antibody by surface functionalization of graphite-encapsulated magnetic beads using ammonia gas plasma technology for capturing influenza A virus. Bioorg Med Chem Lett. 2015;25:1876–1879. doi: 10.1016/j.bmcl.2015.03.036. [DOI] [PubMed] [Google Scholar]
- 113.Tran C, Yasir M, Dutta D, Eswaramoorthy N, Suchowerska N, Willcox M, et al. Single step plasma process for covalent binding of antimicrobial peptides on catheters to suppress bacterial adhesion. ACS Appl Bio Mater. 2019;2:5739–5748. doi: 10.1021/acsabm.9b00776. [DOI] [PubMed] [Google Scholar]
- 114.Wrapp D, De Vlieger D, Corbett KS, Torres GM, Wang N, Van Breedam W, et al. Structural basis for potent neutralization of betacoronaviruses by single-domain camelid antibodies. Cell. 2020;181:1004–1015. doi: 10.1016/j.cell.2020.04.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Mulder KCL, Lima LA, Miranda VJ, Dias SC, Franco OL. Current scenario of peptide-based drugs: The key roles of cationic antitumor and antiviral peptides. Front Microbiol. 2013;4 doi: 10.3389/fmicb.2013.00321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Hilchie AL, Wuerth K, Hancock REW. Immune modulation by multifaceted cationic host defense (antimicrobial) peptides. Nat Chem Biol. 2013;9:761–768. doi: 10.1038/nchembio.1393. [DOI] [PubMed] [Google Scholar]
- 117.Jackman JA, Costa VV, Park S, Real ALCV, Park JH, Cardozo PL, et al. Therapeutic treatment of Zika virus infection using a brain-penetrating antiviral peptide. Nat Mater. 2018;17:971–977. doi: 10.1038/s41563-018-0194-2. [DOI] [PubMed] [Google Scholar]
- 118.Jones JC, Turpin EA, Bultmann H, Brandt CR, Schultz-Cherry S. Inhibition of influenza virus infection by a novel antiviral peptide that targets viral attachment to cells. J Virol. 2006;80:11960–11967. doi: 10.1128/JVI.01678-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Pinese C, Jebors S, Echalier C, Licznar-Fajardo P, Garric X, Humblot V, et al. Simple and specific grafting of antibacterial peptides on silicone catheters. Adv Healthc Mater. 2016;5:3067–3073. doi: 10.1002/adhm.201600757. [DOI] [PubMed] [Google Scholar]
- 120.North SH, Lock EH, Cooper CJ, Franek JB, Taitt CR, Walton SG. Plasma-based surface modification of polystyrene microtiter plates for covalent immobilization of biomolecules. ACS Appl Mater Interfaces. 2010;2:2884–2891. doi: 10.1021/am100566e. [DOI] [PubMed] [Google Scholar]
- 121.Vreuls C, Zocchi G, Thierry B, Garitte G, Griesser SS, Archambeau C, et al. Prevention of bacterial biofilms by covalent immobilization of peptides onto plasma polymer functionalized substrates. J Mater Chem. 2010;20:8092–8098. [Google Scholar]
- 122.Zhang Q, Honko A, Zhou J, Gong H, Downs SN, Vasquez JH, et al. Cellular nanosponges inhibit SARS-CoV-2 Infectivity. Nano Lett. 2020;20:5570–5574. doi: 10.1021/acs.nanolett.0c02278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Rambhia KJ, Ma PX. Controlled drug release for tissue engineering. J Control Release. 2015;219:119–128. doi: 10.1016/j.jconrel.2015.08.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Zelikin AN. Drug releasing polymer thin films: New era of surface-mediated drug delivery. ACS Nano. 2010;4:2494–2509. doi: 10.1021/nn100634r. [DOI] [PubMed] [Google Scholar]
- 125.Yoshida S, Hagiwara K, Hasebe T, Hotta A. Surface modification of polymers by plasma treatments for the enhancement of biocompatibility and controlled drug release. Surf Coat Technol. 2013;233:99–107. [Google Scholar]
- 126.Palumbo F, Treglia A, Lo Porto C, Fracassi F, Baruzzi F, Frache G, et al. Plasma-deposited nanocapsules containing coatings for drug delivery applications. ACS Appl Mater Interfaces. 2018;10:35516–35525. doi: 10.1021/acsami.8b11504. [DOI] [PubMed] [Google Scholar]
- 127.Labay C, Canal JM, Modic M, Cvelbar U, Quiles M, Armengol M, et al. Antibiotic-loaded polypropylene surgical meshes with suitable biological behaviour by plasma functionalization and polymerization. Biomaterials. 2015;71:132–144. doi: 10.1016/j.biomaterials.2015.08.023. [DOI] [PubMed] [Google Scholar]
- 128.Polonskyi O, Solař P, Kylián O, Drábik M, Artemenko A, Kousal J, et al. Nanocomposite metal/plasma polymer films prepared by means of gas aggregation cluster source. Thin Solid Films. 2012;520:4155–4162. [Google Scholar]
- 129.Kylián O, Petr M, Serov A, Solař P, Polonskyi O, Hanuš J, et al. Hydrophobic and super-hydrophobic coatings based on nanoparticles overcoated by fluorocarbon plasma polymer. Vacuum. 2014;100:57–60. [Google Scholar]
- 130.Heyse P, Van Hoeck A, Roeffaers MBJ, Raffin JP, Steinbüchel A, Stöveken T, et al. Exploration of atmospheric pressure plasma nanofilm technology for straightforward bio-active coating deposition: Enzymes, plasmas and polymers, an elegant synergy. Plasma Process Polym. 2011;8:965–974. [Google Scholar]
- 131.Lo Porto C, Palumbo F, Buxadera-Palomero J, Canal C, Jelinek P, Zajickova L, et al. On the plasma deposition of vancomycin-containing nano-capsules for drug-delivery applications. Plasma Process Polym. 2018;15 [Google Scholar]
- 132.Palumbo F, Camporeale G, Yang YW, Wu JS, Sardella E, Dilecce G, et al. Direct plasma deposition of lysozyme-embedded bio-composite thin films. Plasma Process Polym. 2015;12:1302–1310. [Google Scholar]
- 133.Palumbo F, Lo Porto C, Fracassi F, Favia P. Recent advancements in the use of aerosol-assisted atmospheric pressure plasma deposition. Coatings. 2020;10 [Google Scholar]
- 134.Ingle AP, Duran N, Rai M. Bioactivity, mechanism of action, and cytotoxicity of copper-based nanoparticles: A review. Appl Microbiol Biotechnol. 2014;98:1001–1009. doi: 10.1007/s00253-013-5422-8. [DOI] [PubMed] [Google Scholar]
- 135.Rai M, Deshmukh SD, Ingle AP, Gupta IR, Galdiero M, Galdiero S. Metal nanoparticles: The protective nanoshield against virus infection. Crit Rev Microbiol. 2016;42:46–56. doi: 10.3109/1040841X.2013.879849. [DOI] [PubMed] [Google Scholar]
- 136.Behzadinasab S, Chin A, Hosseini M, Poon L, Ducker WA. A surface coating that rapidly inactivates SARS-CoV-2. ACS Appl Mater Interfaces. 2020;12:34723–34727. doi: 10.1021/acsami.0c11425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.De Vietro N, Conte A, Incoronato AL, Del Nobile MA, Fracassi F. Aerosol-assisted low pressure plasma deposition of antimicrobial hybrid organic-inorganic Cu-composite thin films for food packaging applications. Innov Food Sci Emerg Technol. 2017;41:130–134. [Google Scholar]
- 138.Fanelli F, Mastrangelo AM, Fracassi F. Aerosol-assisted atmospheric cold plasma deposition and characterization of superhydrophobic organic-inorganic nanocomposite thin films. Langmuir. 2014;30:857–865. doi: 10.1021/la404755n. [DOI] [PubMed] [Google Scholar]
- 139.Mathioudaki S, Barthélémy B, Detriche S, Vandenabeele C, Delhalle J, Mekhalif Z, et al. Plasma treatment of metal oxide nanoparticles: Development of core-shell structures for a better and similar dispersibility. ACS Appl Nano Mater. 2018;1:3464–3473. [Google Scholar]
- 140.Morales-Narváez E, Dincer C. The impact of biosensing in a pandemic outbreak: COVID-19. Biosens Bioelectron. 2020;163 doi: 10.1016/j.bios.2020.112274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Won J, Lee S, Park M, Kim TY, Park MG, Choi BY, et al. Development of a laboratory-safe and low-cost detection protocol for SARS-CoV-2 of the coronavirus disease 2019 (COVID-19) Exp Neurobiol. 2020;29:107–119. doi: 10.5607/en20009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Seo G, Lee G, Kim MJ, Baek SH, Choi M, Ku KB, et al. Rapid detection of COVID-19 causative virus (SARS-CoV-2) in human nasopharyngeal swab specimens using field-effect transistor-based biosensor. ACS nano. 2020;14:5135–5142. doi: 10.1021/acsnano.0c02823. [DOI] [PubMed] [Google Scholar]
- 143.Choi YH, Lee GY, Ko H, Chang YW, Kang MJ. Development of SPR biosensor for the detection of human hepatitis B virus using plasma-treated parylene-N film. Biosens Bioelectron. 2014;56:286–294. doi: 10.1016/j.bios.2014.01.035. [DOI] [PubMed] [Google Scholar]
- 144.Bilek MM, McKenzie DR. Plasma modified surfaces for covalent immobilization of functional biomolecules in the absence of chemical linkers: Towards better biosensors and a new generation of medical implants. Biophys Rev. 2010;2:55–65. doi: 10.1007/s12551-010-0028-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Hosseini S, Ibrahim F, Djordjevic I, Koole LH. Recent advances in surface functionalization techniques on polymethacrylate materials for optical biosensor applications. Analyst. 2014;139:2933–2943. doi: 10.1039/c3an01789c. [DOI] [PubMed] [Google Scholar]
- 146.Gürso M. All-dry patterning method to fabricate hydrophilic/hydrophobic surface for fog harvesting. Colloid Polym Sci. 2020;298:969–976. [Google Scholar]
- 147.Raeiszadeh M, Adeli B. A critical review on ultraviolet disinfection systems against COVID-19 outbreak: Applicability, validation, and safety considerations. ACS Photonics. 2020;7:2941–2951. doi: 10.1021/acsphotonics.0c01245. [DOI] [PubMed] [Google Scholar]
- 148.Muranyi P, Wunderlich J, Heise M. Sterilization efficiency of a cascaded dielectric barrier discharge. J Appl Microbiol. 2007;103:1535–1544. doi: 10.1111/j.1365-2672.2007.03385.x. [DOI] [PubMed] [Google Scholar]
- 149.Popov OA, Starshinov PV, Ilikeeva RA, Irkhin IV. Ferrite-free closed-loop inductively-coupled low mercury pressure UV lamp. Plasma Res Express. 2020;2 [Google Scholar]
- 150.Qi K, Xin JH, Daoud WA, Mark CL. Functionalizing polyester fiber with a self-cleaning property using anatase TiO2 and low-temperature plasma treatment. Int J Appl Ceram Technol. 2007;4:554–563. [Google Scholar]
- 151.Chemin JB, Bulou S, Baba K, Fontaine C, Sindzingre T, Boscher ND, et al. Transparent anti-fogging and self-cleaning TiO2/SiO2 thin films on polymer substrates using atmospheric plasma. Sci Rep. 2018;8 doi: 10.1038/s41598-018-27526-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Hajkova P, Spatenka P, Horsky J, Horska I, Kolouch A. Photocatalytic effect of TiO2 films on viruses and bacteria. Plasma Process Polym. 2007;4:S397–S401. [Google Scholar]
- 153.Vazirinasab E, Jafari R, Momen G. Evaluation of atmospheric-pressure plasma parameters to achieve superhydrophobic and self-cleaning HTV silicone rubber surfaces via a single-step, eco-friendly approach. Surf Coat Technol. 2019;375:100–111. [Google Scholar]
- 154.Khaleghi H, Eshaghi A. Fabrication of superhydrophobic micro-nano structure Al2O3-13% TiO2/PTFE coating with anti-fuoling and self-cleaning properties. Surf Interfaces. 2020;20 [Google Scholar]
- 155.Canal C, Khurana K, Gallinetti S, Bhatt S, Pulpytel J, Arefi-Khonsari F, et al. Design of calcium phosphate scaffolds with controlled simvastatin release by plasma polymerisation. Polymer (Guildf) 2016;92:170–178. [Google Scholar]
- 156.Körner E, Aguirre MH, Fortunato G, Ritter A, Rühe J, Hegemann D. Formation and distribution of silver nanoparticles in a functional plasma polymer matrix and related Ag+ release properties. Plasma Process Polym. 2010;7:619–625. [Google Scholar]
- 157.Kuzminova A, Beranová J, Polonskyi O, Shelemin A, Kylián O, Choukourov A, et al. Antibacterial nanocomposite coatings produced by means of gas aggregation source of silver nanoparticles. Surf Coat Technol. 2016;294:225–230. [Google Scholar]
- 158.Lu G, Hu Y, Wang Q, Qi J, Gao F, Li Y, et al. Molecular basis of binding between novel human coronavirus MERS-CoV and its receptor CD26. Nature. 2013;500:227–231. doi: 10.1038/nature12328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Matsuyama S, Nagata N, Shirato K, Kawase M, Takeda M, Taguchi F. Efficient activation of the severe acute respiratory syndrome coronavirus spike protein by the transmembrane protease TMPRSS2. J Virol. 2010;84:12658–12664. doi: 10.1128/JVI.01542-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Iwata-Yoshikawa N, Okamura T, Shimizu Y, Hasegawa H, Takeda M, Nagata N. TMPRSS2 contributes to virus spread and immunopathology in the airways of murine models after coronavirus infection. J Virol. 2019;93:e01815–18. doi: 10.1128/JVI.01815-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Hoffmann M, Kleine-Weber H, Schroeder S, Krüger N, Herrler T, Erichsen S, et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Jeffers SA, Tusell SM, Gillim-Ross L, Hemmila EM, Achenbach JE, Babcock GJ, et al. CD209L (L-SIGN) is a receptor for severe acute respiratory syndrome coronavirus. Proc Natl Acad Sci U S A. 2004;101:15748–15753. doi: 10.1073/pnas.0403812101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Cho NJ, Glenn JS. Materials science approaches in the development of broad-spectrum antiviral therapies. Nat Mater. 2020;19:813–816. doi: 10.1038/s41563-020-0698-4. [DOI] [PubMed] [Google Scholar]