Abstract
Background
About half of patients with Crohn's disease (CD) require surgery within 10 years of diagnosis. Resection of the affected segment is highly effective, however the majority of patients experience clinical recurrence after surgery. Most of these patients have asymptomatic endoscopic recurrence weeks or months before starting with symptoms. This inflammation can be detected by colonoscopy and is a good predictor of poor prognosis.Therapy guided by colonoscopy could tailor the management and improve the prognosis of postoperative CD.
Objectives
To assess the effects of prophylactic therapy guided by colonoscopy in reducing the postoperative recurrence of CD in adults.
Search methods
The following electronic databases were searched up to 17 December 2019: MEDLINE, Embase, CENTRAL, Clinical Trials.gov, WHO Trial Registry and Cochrane IBD specialized register. Reference lists of included articles, as well as conference proceedings were handsearched.
Selection criteria
Randomised controlled trials (RCTs), quasi‐RCTs and cohort studies comparing colonoscopy‐guided management versus management non‐guided by colonoscopy.
Data collection and analysis
Two review authors independently considered studies for eligibility, extracted the data and assessed study quality. Methodological quality was assessed using both the Cochrane 'Risk of bias' tool for RCTs and Newcastle‐Ottawa scale (NOS) for cohort studies. The primary outcome was clinical recurrence. Secondary outcomes included: endoscopic, surgical recurrence and adverse events. We calculated the risk ratio (RR) for each dichotomous outcome and extracted the hazard ratio (HR) for time‐to‐event outcomes. All estimates were reported with their corresponding 95% confidence interval (CI). Data were analysed on an intention‐to‐treat (ITT) basis. The overall quality of the evidence was evaluated using GRADE criteria.
Main results
Two RCTs (237 participants) and five cohort studies (794 participants) met the inclusion criteria. Meta‐analysis was not conducted as the studies were highly heterogeneous. We included two comparisons.
Intensification of prophylactic‐therapy guided by colonoscopy versus intensification guided by clinical recurrence
One unblinded RCT and four retrospective cohort studies addressed this comparison. All participants received the same prophylactic therapy immediately after surgery. In the colonoscopy‐based management group the therapy was intensified in case of endoscopic recurrence; in the control group the therapy was intensified only in case of symptoms.
In the RCT, clinical recurrence (defined as Crohn's Disease Activity Index (CDAI) > 150 points) in the colonoscopy‐based management group was 37.7% (46/122) compared to 46.1% (21/52) in the control group at 18 months' follow up (RR 0.82, 95% CI: 0.56 to 1.18, 174 participants, low‐certainty evidence). There may be a reduction in endoscopic recurrence at 18 months with colonoscopy‐based management (RR 0.73, 95% CI 0.56 to 0.95, 1 RCT, 174 participants, low‐certainty evidence). The certainty of the evidence for surgical recurrence was very low, due to only four cohort studies with inconsistent results reporting this outcome.
Adverse events at 18 months were similar in both groups, with 82% in the intervention group (100/122) and 86.5% in the control group (45/52) (RR 0.95, 95% CI:0.83 to 1.08, 1 RCT, 174 participants, low‐certainty of evidence).The most common adverse events reported were alopecia, wound infection, sensory symptoms, systemic lupus, vasculitis and severe injection site reaction. Perforations or haemorrhages secondary to colonoscopy were not reported.
Initiation of prophylactic‐therapy guided by colonoscopy versus initiation immediately after surgery
An unblinded RCT and two retrospective cohort studies addressed this comparison. The control group received prophylactic therapy immediately after surgery, and in the colonoscopy‐based management group the therapy was delayed up to detection of endoscopic recurrence.
The effects on clinical and endoscopic recurrence are uncertain (clinical recurrence until week 102: RR 1.16, 95% CI 0.73 to 1.84; endoscopic recurrence at week 102: RR 1.16, 95% CI 0.73 to 1.84; 1 RCT, 63 participants, very low‐certainty evidence). Results from one cohort study were similarly uncertain (median follow‐up 32 months, 199 participants). The effects on surgical recurrence at a median follow‐up of 50 to 55 months were also uncertain in one cohort study (RR 0.79, 95% CI 0.38 to 1.62, 133 participants, very low‐certainty evidence).
There were fewer adverse events with colonoscopy‐based management (54.8% (17/31)) compared with the control group (93.8% (30/32)) but the evidence is very uncertain (RR 0.58, 95% CI 0.42 to 0.82; 1 RCT, 63 participants). Common adverse events were infections, gastrointestinal intolerance, leukopenia, pancreatitis and skin lesions. Perforations or haemorrhages secondary to colonoscopy were not reported.
Authors' conclusions
Intensification of prophylactic‐therapy guided by colonoscopy may reduce clinical and endoscopic postoperative recurrence of CD compared to intensification guided by symptoms, and there may be little or no difference in adverse effects. We are uncertain whether initiation of therapy guided by colonoscopy impacts postoperative recurrence and adverse events when compared to initiation immediately after surgery, as the certainty of the evidence is very low. Further studies are necessary to improve the certainty of the evidence of this review.
Plain language summary
Colonoscopy‐based prophylactic therapy for postoperative Crohn’s disease
What is Crohn's disease?
Crohn's disease (CD) is a chronic inflammatory disorder that can affect any segment of the gastrointestinal tract. Symptoms include abdominal pain, non‐bloody diarrhoea and weight loss. CD is characterised by periods of clinical relapse when people experience symptoms and periods of clinical remission when the symptoms stop. Despite the available therapies, about half of patients require surgery and resection of the affected segment within 10 years of diagnosis. This surgery is highly effective, however, the majority require a second surgery after 10 years.
What is colonoscopy?
Colonoscopy is a procedure whereby a physician inserts a viewing tube (colonoscope) into the rectum for the purpose of inspecting the colon.
What did the researchers investigate?
Most patients that experience a clinical relapse of CD have inflammation of the intestinal mucosa weeks or months before starting with symptoms. This asymptomatic inflammation can be detected by colonoscopy and it is usually called endoscopic recurrence. The researchers investigated whether the initiation or intensification of prophylactic‐therapy guided by the detection of endoscopic recurrence may improve the prognosis of postoperative CD.
What are the issues related to start prophylactic‐therapy in asymptomatic patients?
The drugs usually used as prophylactic‐therapy in CD are 5‐aminosalicylates, antibiotics, thiopurines, methotrexate and/or anti‐tumour necrosis factor antibody (anti‐TNFα) drugs. The main issues related to these drugs are costs and adverse effects. The most frequent adverse events are alopecia, gastrointestinal intolerance, infections, sensory symptoms, lupus, vasculitis, leukopenia, pancreatitis, skin lesions and less frequently skin and haematological cancer.
How was this study performed?
A systematic review of the current literature was performed to assess the efficacy of prophylactic‐therapy guided by colonoscopy in reducing the postoperative recurrence of CD. An electronic search of several databases was performed and studies that met our inclusion criteria were selected for further evaluation.
What did the researchers find?
The researchers identified three strategies of management of CD after surgery: prophylactic‐therapy guided by clinical symptoms, prophylactic‐therapy guided by colonoscopy and full‐treatment immediately after surgery.
Five studies compared prophylactic‐therapy guided by colonoscopy versus prophylactic‐therapy guided by clinical symptoms. In these studies, all patients received the same prophylactic therapy immediately after surgery. In the colonoscopy‐based management group, therapy was intensified in the case of asymptomatic mucosal inflammation, and in the control group, therapy was intensified only in case of clinical symptoms. According to these studies, intensification of therapy guided by colonoscopy may reduce postoperative clinical recurrence and endoscopic recurrence when compared to intensification guided by symptoms. Data from the study with the highest methodological quality showed that 378 patients in the colonoscopy‐guided management group experience clinical recurrence compared to 462 patients in the group where the management was guided by symptoms, assuming 18 months of follow‐up and a population of 1000 patients underwent intestinal resection due to CD in each group. At 18 months the benefit is greater in terms of endoscopic recurrence, which is a good predictor of clinical recurrence. Studies of lower methodological quality but longer follow‐up also supported the benefit in terms of clinical recurrence. Additionally, there may be little or no difference in the risk of adverse events in colonoscopy‐guided management compared to intensification guided by symptoms. We are uncertain whether intensification of prophylactic therapy guided by colonoscopy compared to therapy guided by symptoms impacts surgical recurrence as the certainty of the evidence is very low.
Three studies compared prophylactic‐therapy guided by colonoscopy versus full‐treatment immediately after surgery. In the colonoscopy‐based management group, the therapy was delayed up to the detection of asymptomatic mucosal inflammation, while in the other group the drugs were started immediately after surgery. Unfortunately, these studies had many limitations, hence the certainty of the evidence was judged as very low. Considering this, we are uncertain about the effect of initiation of therapy guided by colonoscopy on postoperative recurrence and adverse effects when compared to therapy immediately after surgery.
Conclusions
Prophylactic‐therapy guided by colonoscopy may decrease the risk of clinical and endoscopic recurrence compared to prophylactic‐therapy guided by symptoms with little or no difference the risk of adverse events. We are uncertain whether initiation of therapy guided by colonoscopy compared to initiation immediately after surgery impacts postoperative recurrence and adverse events.
Summary of findings
Background
Description of the condition
Crohn’s disease (CD) is a chronic inflammatory bowel disease of unknown origin. A recent systematic review (Ng 2018), with worldwide data reported incidence rates (per 100,000 inhabitants) ranging from 6.3 to 23.82 in North America, 0 to 15.4 in Europe, 0.06 to 3.91 in Asia, 0 to 3.5 in South America, and 12.96 to 29.3 in Australia. Furthermore, the same systematic review (Ng 2018) presented prevalence rates (per 100,000 inhabitants) ranging from 96.3 to 318.5 in North America, 1.51 to 322 in Europe, 1.05 to 18.6 in Asia, 0.9 to 41.4 in South America, and 155.2 to 197.3 in Oceania. These data agree with two previous systematic reviews (Molodecky 2012; Selvaratnam 2019).
CD is characterised by relapsing, transmural inflammation that can affect any portion of the gastrointestinal tract, but it is usually localised in the terminal ileum or colon (Baumgart 2012; Lamb 2019). Despite the available therapies, only 10% of patients achieve prolonged clinical remission, with up to 60% requiring intestinal resection within 10 years of diagnosis (Cosnes 2002; Peyrin‐Biroulet 2010). This surgery is highly effective for induction of remission, however, 47% of patients evolve with symptomatic recurrence at five years, and up to 61% require a second surgery after 10 years (Baumgart 2012; Bernell 2000; Lamb 2019; Margagnoni 2011; Peyrin‐Biroulet 2010).
Description of the intervention
Evidence supporting the management of postoperative CD is scarce. Small randomised controlled trials (RCTs) have shown that thiopurines (i.e. azathioprine, 6‐mercaptopurine) and anti‐tumour necrosis factor antibodies (i.e. infliximab, adalimumab) decrease the postoperative recurrence of CD (Colombel 2014; Gjuladin‐Hellon 2019; Regueiro 2009; Savarino 2013; Yoshida 2012). However, the limited available evidence and the cost and adverse‐event profiles of these drugs have precluded the establishment of a standard preventive strategy. From a theoretical point of view, there are three preventive strategies: therapy for all patients immediately after surgery; therapy based on classification of patients according to risk of recurrence; or no prophylactic therapy. The most frequent approach involves classifying patients as low or high risk of postoperative recurrence according to clinical risk factors (Blum 2009, Mowat 2011). High‐risk patients receive prophylactic therapy while low‐risk patients usually do not receive any therapy unless they experience symptomatic recurrence. The evidence that supports this course of action is limited (Mowat 2011).
Recurrence of CD is usually confirmed by performing a colonoscopy on symptomatic patients in order to detect erythema, ulcers, or inflammatory stenosis (Baumgart 2012; Lamb 2019; Mowat 2011). Despite this common practice, prospective studies have shown that the majority of patients develop endoscopic lesions before gastrointestinal symptoms, suggesting that recurrence of the disease starts with a subclinical phase. This subclinical phase has been called "endoscopic recurrence". According to the available evidence, up to 60% of patients had endoscopic recurrence one year after surgery, however only 20% reported clinical symptoms. After 24 months only 3% of patients with minimal or no endoscopic lesions developed clinical recurrence, while over 50% of patients with endoscopic recurrence experienced symptomatic recurrence (Baert 2010; Colombel 2014; D'Haens 2008; Frøslie 2007; Rutgeerts 1984; Rutgeerts 1990; Schintzler 2009, Ungaro 2020). Based on this evidence, colonoscopic surveillance may be able to identify those patients at the highest risk of symptomatic recurrence of CD (Lamb 2019).
How the intervention might work
During many years the management of postoperative CD has been based on clinical risk factors of recurrence. Unfortunately, the accuracy of this strategy is low (Lamb 2019; Pascua 2008). Colonoscopic surveillance and diagnosis of endoscopic recurrence would allow identifying those patients at the highest risk of symptomatic recurrence and tailor the initiation of preventative therapy (Lamb 2019).
Why it is important to do this review
An effective treatment strategy is needed for the maintenance of postoperative remission in CD. While the majority of patients will develop postoperative recurrence and require a second surgery, one third of patients will remain in remission without medication five years after resection (Jones 2014; Peyrin‐Biroulet 2010; Watanabe 2014). Furthermore, the available medications are costly and associated with a range of adverse events (Ford 2009; Kandiel 2005; Williams 2014). Preventative therapy is therefore not suitable for all postoperative patients. It is essential to develop accurate techniques for identifying patients who are most likely to relapse after surgery.
Objectives
To assess the effects of prophylactic therapy guided by colonoscopy in reducing the postoperative recurrence of Crohn’s disease (CD) in adults.
Methods
Criteria for considering studies for this review
Types of studies
Randomised controlled trials (RCTs), quasi‐RCTs and cohort studies (prospective and retrospective) were considered for inclusion. We considered a quasi‐RCT as one in which the allocation is not truly random, for example, allocation by date of birth, day of the week, medical record number or alternation. We decided to include quasi‐RCTs and cohort studies because clinical and surgical recurrence are long‐term outcomes, therefore an RCT could be unable to address these effects of the intervention. There were no restrictions based on publication status or language of publication.
Types of participants
Adult participants (>16 years of age) undergoing intestinal resection due to CD that meet the following criteria.
No macroscopic evidence of CD in the remaining gut after surgery.
Endoscopically accessible anastomosis.
There was no restriction regarding gender.
Studies focused on strictureplasty or other surgical procedures without intestinal resection were excluded.
Types of interventions
All studies that compared prophylactic therapy guided by colonoscopic surveillance (intervention group) versus prophylactic therapy non‐guided by colonoscopic surveillance (control group) were considered for inclusion. The inclusion criteria are detailed below.
-
Intervention group:
colonoscopic surveillance;
modification of the baseline preventive strategy in case of detection of endoscopic recurrence.
-
Control group:
no colonoscopic surveillance;
participants remain in the baseline preventive strategy up to clinical recurrence.
There were no restrictions in terms of type of baseline preventive strategy (i.e. prophylaxis for all participants, prophylaxis based on clinical risk factors or no‐prophylaxis), modifications of the prophylactic therapy after endoscopic recurrence detection (i.e. initiation, intensification or change of the prophylactic therapy), or the drugs used for prevention (i.e. 5‐ASA, budesonide, antibiotics, thiopurines or biologic therapy). Additionally, there was no restriction on the scales used for the diagnosis of endoscopic recurrence.
Types of outcome measures
Primary outcomes
The primary outcome was the proportion of participants that experience clinical postoperative recurrence, as defined by the included studies. This outcome was evaluated at fixed intervals, 12 to 24 months, 25 to 36 months and > 36 months after surgery.
Secondary outcomes
Secondary outcome include the following.
Endoscopic recurrence, as defined by the primary studies. The following definitions were considered a priori: Rutgeerts score ≥ i2, simple endoscopic score for CD (SES‐CD) ≥ 4 and CD endoscopic index of severity, (CDEIS) ≥ 3 (Daperno 2004; Rutgeerts 1990; Schoepfer 2010).
Surgical recurrence.
Crohn's Disease Activity Index (CDAI) (at the end of follow‐up).
Harvey Bradshaw index score (at the end of follow‐up).
Mortality by any cause.
Adverse effects of colonoscopy: perforation and haemorrhage.
Adverse events of prophylactic therapy. The following events will be considered a priori: withdrawal due to adverse events, haematological (anaemia, leucopenia (reduction in the number of white blood cells), or low platelet count), infections, neoplasms and any serious adverse event, as defined by the primary studies.
Health‐related quality of life, as defined by the primary studies.
All these outcomes were evaluated at fixed intervals, 12 to 24 months, 25 to 36 months and > 36 months after surgery. Additionally, the following time to event outcomes were evaluated.
Time to clinical recurrence.
Time to surgical recurrence.
Search methods for identification of studies
Electronic searches
The search was not restricted by language, date or publication status. To build the strategy the following terms were considered: postoperative CD, endoscopic and mucosal recurrence, endoscopic and mucosal remission, colonoscopy, sigmoidoscopy and clinical recurrence. We searched the following databases from inception to 17 December 2019:
MEDLINE;
Embase;
Cochrane Central Register of Controlled Trials (CENTRAL);
Clinical Trials.gov;
WHO Trial Registry; and
Cochrane IBD specialized register.
The search strategies for each database are reported in Appendix 1; Appendix 2; Appendix 3, Appendix 4; Appendix 5; Appendix 6, respectively.
Searching other resources
We handsearched conference abstracts from 2010 onwards to identify studies published as abstracts. Conference proceedings included Digestive Disease Week, United European Gastroenterology Week and the European Crohn's and Colitis Organisation. We reviewed all the references of the included studies and the articles that cited them. We searched in Epistemonikos database (www.epistemonikos.org) for previous reviews (similar or related) to identify additional citations. Ongoing trials were identified by searching Clinicaltrials.gov (http://clinicaltrials.gov). We also searched Google scholar and contacted experts in the field.
Data collection and analysis
Selection of studies
Two review authors (GB and HM) independently screened titles and abstracts to identify potentially eligible studies based on the inclusion criteria detailed above. Two review authors (RC and CH) independently screened studies selected for full‐text review. Disagreements at either stage were resolved by discussion and consensus. A third review author (GN) acted as the arbitrator when consensus was not reached. The study selection process was reported in a PRISMA flow diagram.
Data extraction and management
Four review authors (RC, GB, HM and CH) independently extracted data from included studies using forms specially designed for this purpose. Each study was independently reviewed by at least two review authors. Discrepancies were resolved by discussion. A third review author (GN) acted as the arbitrator when consensus was not reached.
For each included study, the following information was collected: source (i.e. citation, study name if applicable and contact details); inclusion and exclusion criteria; methods of the study (as per quality assessment checklist); number of participant centres; characteristics of participants (i.e. age, sex, smoking status, type of surgery, localisation and behaviour of the disease according to Montreal classification), frequency of colonoscopic surveillance, baseline preventive strategy (i.e. prophylactic therapy for all participants, according to clinical risk factors or no prophylactic therapy), drugs used for prevention and intensification, and data relating to the outcomes (i.e. number of participants allocated to each group, and for each outcome of interest, sample size, missing participants, summary data for each group, estimate of effect with confidence interval and P value and subgroup analyses).
Assessment of risk of bias in included studies
Four review authors (RC, GB, HM and CH) independently assessed the risk of bias of each study. Each study was independently reviewed by at least two review authors. Two assessment tools were used based on study design.
We used the Cochrane 'Risk of bias' tool for RCTs (Higgins 2011a). Trials were rated as high, low or unclear risk of bias for each of the following domains: sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective outcome reporting and other sources of bias.
We used the Newcastle‐Ottawa Scale (NOS) for cohort studies. For this tool, each study is evaluated using eight items, categorised into three domains: the selection of the study groups (four items: representativeness of the exposed cohort; selection of the non exposed cohort; ascertainment of exposure; demonstration that outcome of interest was not present at start of study); the comparability of the groups (one item: comparability of cohorts on the basis of the design or analysis); and the ascertainment of the outcome of interest (three items: assessment of the outcome, whether the follow‐up was long enough to observe the outcome, adequacy of follow‐up of cohorts). A star system is used to allow a semi‐quantitative assessment of study quality, such that the highest quality studies are awarded a maximum of one star for each item, with the exception of the item related to comparability that allows the assignment of two stars. The NOS ranges between zero up to nine stars (Wells 2016). The methodological quality of each cohort study was judged as high only if all items were rated with a low risk of bias. If in at least one of the items the risk of bias was high (zero stars), the overall methodological quality of the study was judged as low. In those studies where the risk of bias was judged as low in the domains "selection of the study groups" and "ascertainment of the outcome of interest", but only one star was assigned to the domain "comparability of the groups", the methodological quality was considered moderate. For the assessment of the domain "comparability of the groups", we pre‐specified key confounders of the association of interest. The following key confounders had to be considered in the design or analysis (i.e. paired samples, multivariable analysis, propensity scores analysis, etc) to received two stars: age at diagnosis of CD, behaviour of the disease according to the Montreal classification (non‐structuring non‐penetrating; structuring; penetrating) and smoking status.
We contacted the authors of included studies if the information available in published studies was insufficient to evaluate the risk of bias. Discrepancies were resolved by discussion. A third review author (GN) acted as the arbitrator when consensus was not reached.
Measures of treatment effect
We analysed data using Review Manager (Revman 5.3.5). We calculated the risk ratio (RR) and corresponding 95% confidence interval (95% CI) for dichotomous outcomes. For continuous outcomes, we planned to calculate the mean difference (MD) and corresponding 95% CI. These effect measures were chosen to favour consistency and interpretability of results. For continuous outcomes measured with different scales (e.g. health‐related quality of life), we planned to calculate the standardised mean difference (SMD) and corresponding 95% CI. For time to event outcomes, we calculated the hazard ratio (HR) and corresponding 95% CI when possible.
Unit of analysis issues
When included studies reported more than one observation for the same outcome during follow‐up, the outcomes were grouped by fixed intervals to perform separate analyses (e.g. 12 to 24 months, 25 to 36 months, > 36 months). Additionally, the timing and frequency of colonoscopic surveillance could vary among the included studies (e.g. at 3, 6 or 12 months after surgery). For the primary analysis, the studies were planned to be pooled regardless of the timing or frequency of colonoscopic surveillance. Considering the risk of heterogeneity, subgroup analyses were planned to compare efficacy and safety across different strategies, when possible. Although some studies reported more than one efficacy or safety event per participant, the primary analysis considered the proportion of participants who experienced at least one event. Considering the nature of the intervention under evaluation, we did not expect to find cross‐over studies or designs with multiple active arms. Despite this, in case of cross‐over design, we determined that only the data of the first phase of the study would be included. In case of multiple active arms (e.g. two or more frequency schedules of colonoscopic surveillance), all of these groups were planned to be pooled in one intervention arm, for the primary analysis.
Dealing with missing data
Whenever possible, we contacted the authors of included studies to request missing data. In relation to loss of follow‐up, the analyses were performed according to the intention‐to‐treat (ITT) principle, i.e. all participants randomised to each group were included in the analysis regardless of whether they received the allocated intervention or withdrew from the study. All withdrawals were deemed to be treatment failures. For continuous outcomes where the standard deviation was not available, the standard deviation was estimated from standard errors, P values or confidence intervals as described by Higgins 2011b. We planned to estimate the standard deviation using the methods proposed by Higgins 2011b, and only if the trials with missing data were a small proportion of the studies included in the review. We also planned to report missing data and imputations. Despite this, we did not perform any imputation and did not use imputed data from trialists in the final analysis.
Assessment of heterogeneity
We planned to assess statistical heterogeneity by visual inspection of forest plots, and by using the Chi2 and the I2 statistics. A P value < 0.10 was considered to indicate statistically significant heterogeneity. Additionally, the following guidance was used to quantify heterogeneity using the I2 statistic: a value of 25% indicates low heterogeneity, 50% indicates moderate heterogeneity, and 75% indicates high heterogeneity (Higgins 2003). We planned to conduct subgroup and sensitivity analyses (described below) to evaluate potential explanations for heterogeneity.
Assessment of reporting biases
We planned to assess the potential of publication bias by visual inspection of funnel plots if at least 10 studies were included in a pooled analysis. The risk of selective reporting bias for individual studies was evaluated by comparing outcomes listed in the protocols to published manuscripts. When the protocols were not available; the risk of bias for this domain was deemed to be unclear.
Data synthesis
RCTs and cohort studies were analysed separately. We planned to combine in a meta‐analysis the data from individual trials when the interventions, participants and outcomes are sufficiently similar. This issue was determined by consensus. A priori we planned to use a random‐effects model to pool data. We planned to assess heterogeneity using the I2 statistics.
Subgroup analysis and investigation of heterogeneity
Considering our inclusion criteria, we expected a high heterogeneity across the included studies. The following subgroup analyses were planned to evaluate potential causes of heterogeneity.
a) Risk of recurrence according to clinical risk factors. A priori we planned to compare participants with low and high risk of recurrence, according to clinical criteria defined by the primary studies.
b) Type of prophylactic therapy. A recent network meta‐analysis shows that the preventive effect of biologics seems to be stronger than the preventive effect of thiopurines, 5‐ASA and placebo (Yang 2014). Additionally, thiopurines appear to be superior to 5‐ASA and placebo for prevention of relapse (Gjuladin‐Hellon 2019; Yang 2014). Based on this evidence, we expected to observe a differential effect of colonoscopy according to the drugs started immediately after surgery. We planned to compare the following subgroups:
prophylactic therapy based on biologics (alone or with other drugs);
prophylactic therapy based on thiopurines (alone or with other drugs, except biologics); and
other strategies: no prophylactic therapy, 5‐ASA, or antibiotics.
c) Timing of colonoscopic surveillance. A priori we expected to observe a differential effect in participants undergoing colonoscopy during the first 12 months compared to those studied after 12 months post‐surgery.
Cohort and RCT studies were evaluated separately.
Sensitivity analysis
We planned to conduct sensitivity analyses by replicating the meta‐analysis after exclusion of low methodological quality studies. Cohort studies with seven or less "stars" according to NOS and RCTs with two or more domains with a high risk of bias were deemed low‐quality studies.
A second sensitivity analysis was planned after the exclusion of any obvious outliers detected by visual inspection of forest plots. Outliers were defined by consensus.
Finally, a sensitivity analysis was planned to be performed based on different strategies for addressing missing data (e.g. exclusion versus imputation).
Summary of findings and assessment of the certainty of the evidence
We used the GRADE approach to assess the overall certainty of the evidence supporting the primary outcome (i.e. clinical recurrence) and selected secondary outcomes including endoscopic recurrence, surgical recurrence, mortality, adverse events and quality of life (Guyatt 2008; Schünemann 2011). The overall evidence was rated as high, moderate, low or very low certainty. When the data come from randomised controlled trials the overall evidence begins as high certainty and, when data come from observational studies begins as low certainty. However, it can be downgraded based on the following criteria: risk of bias, indirectness, inconsistency (unexplained heterogeneity), imprecision (sparse data) and publication bias. Moreover, in case of observational studies, it can be upgraded based on the following criteria: strong evidence of association (significant relative risk of > 2 or < 0.5, based on consistent evidence from two or more observational studies, with no plausible confounders), evidence of a dose response gradient, or all plausible confounders would have reduced the effect.
The certainty of the evidence allows establishing how likely it is that the results observed by the studies coincide with reality and to estimate how much they may change with future studies. An effect estimate based on high‐certainty evidence is unlikely to change with future studies. When the overall certainty of evidence is moderate, new studies may change the estimate. Low and very low certainty of evidence means that there is uncertainty about the effect estimate and further research will probably change the effect estimate (Atkins 2004; Guyatt 2008; Schünemann 2011).
Finally, the results of the GRADE analysis were reported in 'Summary of findings' tables and key message was written according to the last suggestion of Cochrane Norway Cochrane Norway 2019.
Results
Description of studies
Results of the search
The search included articles published up to 17 December, 2019. A total of 3810 citations were screened for retrieval. Two articles were detected from another source (references of selected articles). Six hundred and sixty‐nine duplicates were excluded. Among the remaining 3143 citations, 3131 were excluded after scanning the titles and/or abstracts because the design, participants, intervention or outcome did not meet inclusion criteria. Twelve full‐text articles were retrieved for detailed evaluation, five were excluded (Barreiro‐De Acosta 2019; Gonzales‐Lama 2015; Papamichael 2012; Sorrentino 2012; Yamamoto 2009) (Figure 1).
1.
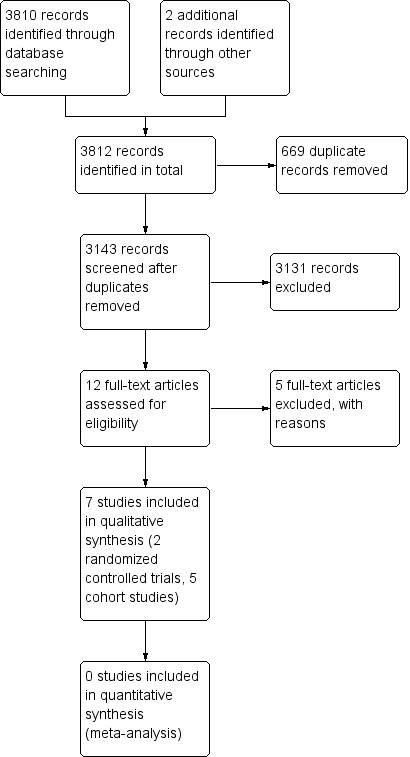
Study flow diagram.
Included studies
The final analysis includes seven studies, two randomised controlled trials (RCTs) (237 participants; De Cruz 2015; Ferrante 2015) and five retrospective cohorts (794 participants; Baudry 2014; Bordeianou 2011; Boucher 2016; De Cruz 2013, Guo 2018). All participants in the included studies had an endoscopic accessible anastomosis, 89.8% (926/1031) ileo‐colonic anastomosis, 3.5% ileo‐ileal anastomosis (36/1031), 2% ileo‐rectal anastomosis (21/1031) and 4.7% colo‐colonic anastomosis (48/1031). The design of the included studies was heterogeneous and two types of comparisons were identified:
Intensification of baseline prophylactic therapy guided by colonoscopy versus intensification guided by clinical recurrence;
Initiation of prophylactic therapy guided by colonoscopy versus initiation of prophylactic therapy immediately after surgery.
Four studies exclusively assessed the first comparison (Baudry 2014; Boucher 2016; De Cruz 2013; De Cruz 2015), and two studies exclusively assessed the second comparison (Bordeianou 2011; Ferrante 2015). One cohort study included three groups and assessed both comparisons (Guo 2018).
The studies were highly heterogeneous in terms of design and result reporting, hence both meta‐analyses and planned sensitivity analyses were not conducted.
Intensification of prophylactic therapy guided by colonoscopy versus intensification guided by clinical recurrence
One RCT (De Cruz 2015) and four cohort studies (Baudry 2014; Boucher 2016; De Cruz 2013; Guo 2018) addressed this comparison. In these studies, all participants received the same prophylactic therapy immediately after surgery. In the colonoscopy‐based management group, the therapy was intensified in case of endoscopic recurrence, and in the control group, the therapy was intensified only in case of clinical recurrence.
De Cruz 2015 was a RCT that included 174 participants. Participants were randomly allocated in a ratio 2:1 to colonoscopy at six months after surgery (intervention group) or no colonoscopy (control group). The main outcome was endoscopic recurrence and the secondary outcomes were clinical recurrence, surgical recurrence and adverse effects. The study was planned to include an 18‐month follow‐up after surgery. Participants with a low and high risk of postoperative recurrence were included, although the majority were high risk (145/174 participants, 83.3%). All participants received metronidazole for three months immediately after surgery, low‐risk participants did not receive another prophylaxis, high‐risk participants received thiopurines. Participants allocated to the intervention group who developed endoscopic recurrence received an intensified scheme: low risk started thiopurines, high risk stepped‐up to biologic therapy (adalimumab) plus thiopurines. Participants allocated to the control group maintained the baseline management up to symptomatic recurrence. For details see Characteristics of included studies.
Baudry 2014, Boucher 2016, and De Cruz 2013 were retrospective cohort studies. Baudry 2014 included 132 participants, 90 received colonoscopy‐based management. Boucher 2016 included 161 participants; 49 received colonoscopy‐based management. De Cruz 2013 included 136 participants, 70 were considered under a colonoscopy‐based management. In the three studies, the colonoscopy was performed within the first year after surgery. The median follow‐up was 51 and 53 months in Baudry 2014 and De Cruz 2013, respectively. In Boucher 2016 the follow‐up ranged from six months to 20 years. The definition of both endoscopic and clinical recurrence was heterogeneous. For endoscopic recurrence, Baudry 2014 and Boucher 2016 used Rutgeerts score ≥ i2. De Cruz 2013 used an unspecific definition: any lesion in the neoterminal ileum or at anastomosis. For clinical recurrence, Baudry 2014 used Harvey‐Bradshaw index > 3. Boucher 2016 and De Cruz 2013 used an unspecific definition: recurrent symptoms requiring a change in medical treatment or surgery, after exclusion of other causes. In the three studies, the participants received a similar prophylactic therapy immediately after surgery, which included thiopurines, antibiotics, 5‐ASA or no prophylaxis. Only in Boucher 2016, did the management include biologics (Infliximab or adalimumab). The intensified management after endoscopic recurrence was heterogeneous. In Baudry 2014, the intensified management consisted of an increasing dose of thiopurines, in Boucher 2016, this consisted of an increasing dose of thiopurines or biologic therapy. In De Cruz 2013 the intensified management consisted of thiopurines, antibiotics, 5‐ASA or methotrexate. In terms of statistical analysis, only Baudry 2014 and Boucher 2016 reported a multivariable‐adjusted analysis. For details see Characteristics of included studies.
Initiation of prophylactic therapy guided by colonoscopy versus initiation of prophylactic therapy immediately after surgery
One RCT (Ferrante 2015) and two cohort studies (Bordeianou 2011; Guo 2018) addressed this comparison. In these studies, the control group received prophylactic therapy immediately after surgery and in the colonoscopy‐based management group, the therapy was delayed up‐to detection of endoscopic recurrence.
Ferrante 2015 was a RCT that included 63 participants. Participants were randomly allocated in a ratio 1:1 to initiation of prophylactic therapy in case of detection of endoscopic recurrence (intervention group) or prophylactic therapy immediately after surgery (control group). The study was planned to include a 102‐week follow‐up after surgery. Only participants with a high risk of recurrence were included. Participants allocated to the control group started prophylactic therapy within two weeks after surgery. Participants allocated to the intervention group did not receive prophylactic therapy immediately after surgery, this was started only after detection of endoscopic recurrence at 26 weeks or 52 weeks. In both groups, the prophylactic therapy was azathioprine 2.0 to 2.5 mg/Kg. The diagnosis of endoscopic recurrence was established with a Rutgeerts score ≥ i2 and clinical recurrence with a Crohn's Disease Activity Index (CDAI) ≥150 points. The authors reported that the original sample size was estimated in 200 participants, however, the study was prematurely stopped due to slow recruitment. For details see Characteristics of included studies.
Bordeianou 2011 was a retrospective cohort study that included 199 participants divided in two groups: 69 participants who received prophylactic therapy immediately after surgery and 130 participants who did not receive prophylactic therapy immediately after surgery. In the second group, all participants underwent colonoscopy approximately three to six months after index surgery, 32 participants started therapy after detection of endoscopic recurrence, and 98 participants never received therapy after surgery. The median follow‐up was 32 months. All participants who required initiation of medical therapy, endoscopic procedures or further surgery to control symptoms were deemed to have clinical recurrence. The definition of endoscopic recurrence was not specified. The prophylactic therapy was heterogeneous, consisted of antibiotics, 5‐ASA, thiopurines and/or biologic drugs. In terms of statistical analysis, the results were reported as univariate and multivariable‐adjusted analyses. For details see Characteristics of included studies.
Finally, Guo 2018 was a retrospective cohort that assessed both comparisons. This study included 166 participants divided in three groups: prophylactic therapy immediately after surgery, guided by endoscopic recurrence and guided by clinical recurrence (91, 42 and 33 participants, respectively). The first group received azathioprine between two to four weeks after surgery; the second group underwent colonoscopy between three to 18 months after surgery and azathioprine was started only in case of endoscopic recurrence (Rutgeerts score ≥ i2); the third group received azathioprine only in case of CDAI ≥150 points and C‐reactive protein > 10 mg/L. In terms of statistical analysis, the results were reported as univariate and multivariable‐adjusted analysis. For details see Characteristics of included studies.
Excluded studies
A detailed description of excluded studies is reported in the Characteristics of excluded studies tables. In summary, five observational studies retrieved as full‐text articles were excluded. Three studies were excluded because all the participants underwent colonoscopy after surgery and only those with endoscopic recurrence were included in the final analysis (Gonzales‐Lama 2015; Sorrentino 2012; Yamamoto 2009). In one study the authors compared therapy immediately after surgery versus initiation after detection of endoscopic recurrence, however, all participants underwent colonoscopy after surgery, only participants with endoscopic recurrence were included in the intervention group and the therapy was modified according endoscopic findings in both groups (Papamichael 2012). Finally, one study described the endoscopic monitoring within the first year after surgery, however the authors did not include outcomes of interest for this review (Barreiro‐De Acosta 2019).
Risk of bias in included studies
Randomised controlled trials
The methodological quality of both RCTs was deemed moderate. In relation to the selection bias domain, the random sequence generation and allocation concealment were considered suitable in both studies. Considering the nature of the intervention (colonoscopic surveillance), both studies were open‐label, hence the risk for performance and detection bias was deemed high for the outcome clinical recurrence. For the outcome endoscopic recurrence, in both studies the outcome assessors were blinded, hence the risk of detection bias was low. In both studies, withdrawals were balanced across the groups and the reasons were detailed, they were not related to the outcomes and the analysis was according to the intention‐to‐treat principle. Despite this, in both studies, the withdrawal rate was over 30%. Additionally, in De Cruz 2015 10 participants were excluded from the analysis after randomisation because they did not receive the allocated medication. For these reasons both articles were evaluated with unclear risk of attrition bias. Protocols of both articles were available on‐line (clinicaltrials.gov), and the reported outcomes coincide with the final report. The risk of selective reporting bias was deemed low for De Cruz 2015. However, for Ferrante 2015 the risk was deemed unclear because the recruitment of participants started in 2005, but the protocol was registered in 2014. We did not detect other sources of bias. For details see Figure 2 and Characteristics of included studies.
2.

'Risk of bias' summary: review authors' judgements about each risk of bias item for each included randomized controlled trial.
Retrospective cohorts
The methodological quality of included cohort studies was assessed by the Newcastle‐Ottawa Scale (NOS). The methodological quality was deemed low in four cohort studies (Baudry 2014, Bordeianou 2011, De Cruz 2013, Guo 2018), and moderate in one study (Boucher 2016).
Baudry 2014 was conducted in a centre where the postoperative treatment was protocolised. In this centre, all participants should be evaluated by colonoscopy between 6 to 12 months after surgery; for this reason the control group was composed of participants who did not undergo colonoscopy because of participant’s refusal or medical omission. Additionally, all participants with early clinical recurrence (<12 months after surgery) were excluded. In our opinion, both conditions could be associated with a high risk of selection bias. Additionally, although the authors conducted a multivariable analysis, the factors included in the regression model were selected only based on a statistical criterion (P < 0.1), hence critical confounders were not specified, for example smoking status and age at diagnosis of CD.
In the case of Bordeianou 2011, two key confounders (smoking status and age at diagnosis of CD) were not reported in both univariate and multivariable analysis (unmeasured confounders). Additionally, the loss of follow‐up was high and not balanced across the groups (11.6% in the control group, 19.8% in the intervention group), and could be related to the outcome. Although these data were censored, a multivariable Cox’s proportional hazard analysis did not resolve this issue.
In the case of De Cruz 2013, the groups showed imbalances in potential confounders (age at diagnosis of CD and proportion of participants with penetrating disease), however, a multivariable analysis was not performed as the univariate analysis did not show differences. Additionally, in this study the decision of colonoscopic surveillance was probably related to the suspicion of recurrence and the endoscopic recurrence was not systematically associated with an intensified management (only 15 of 55 participants (27%) with endoscopic recurrence received intensified management). These issues probably favoured the control group and increased the risk of bias.
Boucher 2016 was considered a cohort study of moderate methodological quality. This was conducted in a unique centre and all participants who underwent intestinal resection for CD were assessed for inclusion. Additionally, the allocation to the groups was probably not linked to the outcomes. The authors performed a multivariable regression model that was constructed using statistical criteria and clinically relevant confounders. Despite this, the factors included in the final model were not reported, which could be related to risk of bias. Finally, the follow‐up was long enough to observe outcomes of interest and they were assessed using appropriate record linkage.
In Guo 2018, the authors retrieved the data from a prospectively maintained database. Despite this, the three management strategies were adopted during different time periods: the participants included in the control group (management guided by clinical recurrence) were mainly treated before 2009, while participants included in the other groups were treated afterwards. Considering this, the risk of selection bias was judged as high. Additionally, although the authors conducted multivariable analyses, the factors included in the regression models were selected only based on a statistical criterion (P < 0.1), hence critical confounders were not specified, for example, the behavior of the disease and smoking status.
For a summary of the risk of bias of the cohort studies see Table 3 and Characteristics of included studies.
1. Methodological quality of included cohort studies.
| Risk of bias domain* | Baudry 2014 | Boucher 2016 | De Cruz 2013 | Bordeianou 2011 | Guo 2018 | |
| Selection | Representativeness of the exposed cohort | ★ | ★ | ★ | ★ | ★ |
| Selection of the non‐exposed cohort | ‐ | ★ | ★ | ★ | ‐ | |
| Ascertainment of exposure | ★ | ★ | ‐ | ★ | ★ | |
| Outcome was not present at start of study | ★ | ★ | ★ | ★ | ★ | |
| Comparability | Comparability of cohorts (design or analysis) * | ★ ‐ | ★ ‐ | ‐ ‐ | ★ ‐ | ★ ‐ |
| Outcome | Assessment of outcome | ★ | ★ | ★ | ★ | ★ |
| Length of follow‐up | ★ | ★ | ★ | ★ | ‐ | |
| Adequacy of follow‐up | ★ | ★ | ★ | ‐ | ★ | |
Methodological quality of included cohort studies, according to the Newcastle‐Ottawa Scale (NOS)*.
* NOS allows a semi‐quantitative assessment of the risk of bias. The scale ranges from zero to nine stars. The overall methodological quality of each study was considered "Low" whether in at least one of the items the risk of bias was judged as high (zero stars) (Baudry 2014; Bordeianou 2011; De Cruz 2013; Guo 2018).
Effects of interventions
Summary of findings 1. Intensification of prophylactic therapy guided by colonoscopy compared to intensification guided by clinical recurrence for the prevention of postoperative recurrence of Crohn’s disease.
| Intensification of prophylactic therapy guided by colonoscopy compared to intensification guided by clinical recurrence for the prevention of postoperative recurrence of Crohn’s disease | ||||||
|
Patient or population: adult participants (> 16 years of age) undergoing intestinal resection due to Crohn's disease
Settings: Hospital outpatients Intervention: Intensification of prophylactic therapy guided by colonoscopy Comparison: Intensification guided by clinical recurrence | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) |
Relative effect (95% CI) |
Number of participants (studies) | Certainty of the evidence (GRADE) | Coments | |
|
WITH clinical recurrence guided therapy |
WITH colonoscopy‐guided therapy |
|||||
|
Clinical recurrence (Follow‐up: 18 months) |
462 per 1000 | 378 per 1000 (259 to 545) |
RR 0.82 (0.56 to 1.18) |
174 (1 RCT) |
⊕⊕⊝⊝a,b LOW |
Clinical recurrence was defined by a CDAI >150 points |
|
Endoscopic recurrence (Follow‐up: 18 months) |
673 per 1000 | 485 per 1000 (377 to 640) |
RR 0.73 (0.56 to 0.95) | 174 (1 RCT) |
⊕⊕⊝⊝c,d LOW | Endoscopic recurrence was defined by a Rutgeerts' score ≥ i2 |
|
Surgical recurrence (Follow‐up: various) |
‐ | ‐ | See comment | 504 (4 cohort studies) |
⊕⊝⊝⊝e VERY LOW | Studies were highly heterogeneous; meta‐analysis not performed. Follow‐up ranged from 51 months to 20 years. In two studies with unadjusted analyses the results were: RR 0.58 (CI 95% 0.17 to 2.06) and RR 1.89 (CI 95% 0.49 to 7.23) In two observational studies with multivariable analysis the results were: HR 0.3 (CI 95% 0.13 to 0.70) and HR 0.35 (CI 95% 0.14 to 0.89) (high‐risk patients) |
|
Mortality (Follow‐up: 18 months) |
‐ | ‐ | ‐ | 174 (1 RCT) |
‐ | No deaths were reported during the follow‐up period |
| Quality of life | ‐ | ‐ | ‐ | ‐ | ‐ | Outcome was not measured or reported |
|
Adverse effects (Follow‐up: 18 months) |
346 per 1000 | 270 per 1000 (169 to 433) | RR 0.78 (0.49 to 1.25) | 174 (1 RCT) | ⊕⊝⊝⊝a,b LOW | Adverse events included drug intolerance, sensory symptoms, alopecia, wound infection, lupus, leukocytoclastic vasculitis and severe injection site reaction (biologic therapy). Perforations or haemorrhages secondary to colonoscopy were not reported. |
|
CI 95%: 95% confidence interval (CI); RR: Risk ratio; GRADE: Evidence grades of the GRADE Working Group (see later); RCT: Randomised controlled trial; CDAI: Crohn's disease activity index. *The risk withclinical recurrence guided therapy is based on the risk in the control group of the trials. The risk with colonoscopy‐guided therapy (and its confidence interval) is calculated from relative effect (and its confidence interval). | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect. | ||||||
a The certainty of the evidence was downgraded in one level for risk of bias because trial was open‐label and unclear risk of attrition bias.
b The certainty of the evidence was downgraded in one level for imprecision because the result at each end of the confidence interval entails a different clinical decision.
c Despite the outcome assessor was blinded for the outcome "endoscopic recurrence", the participants and personnel were not blinded. Additionally, there is an unclear risk of attrition bias. For these reasons, the certainty of the evidence was downgraded in one level for risk of bias.
d The certainty of the evidence was downgraded in one level for imprecision. Although the trial did meet its required sample size, only one study assessed this outcome and the confidence interval was wide.
e The certainty of the evidence was downgraded in one level for inconsistency, because different studies present different conclusions.
Summary of findings 2. Initiation of prophylactic therapy guided by colonoscopy compared to initiation of prophylactic therapy immediately after surgery for the prevention of postoperative recurrence of Crohn’s disease.
| Initiation of prophylactic therapy guided by colonoscopy compared to initiation of prophylactic therapy immediately after surgery for the prevention of postoperative recurrence of Crohn’s disease | ||||||
|
Patient or population: Adult participants (> 16 years of age) undergoing intestinal resection due to Crohn's disease.
Settings: Hospital outpatients Intervention: Prophylactic therapy guided by colonoscopy Comparison: Prophylactic therapy immediately after surgery | ||||||
| Outcomes |
Anticipated absolute effects* (95% CI) |
Relative effect (95% CI) |
Number of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
|
WITH therapy immediately after surgery |
WITH colonoscopy‐guided therapy |
|||||
|
Clinical recurrence (until week 102) |
500 per 1000 | 580 per 1000 (365 to 920) |
RR 1.16 (0.73 to 1.84) | 63 (1 RCT) |
⊕⊝⊝⊝a,b,c VERY LOW | Clinical recurrence was defined by a CDAI >150 points |
|
Endoscopic recurrence (at week 102) |
500 per 1000 | 580 per 1000 (365 to 920) |
RR 1.16 (0.73 to 1.84) |
63 (1 RCT) |
⊕⊝⊝⊝a,b,c VERY LOW | Endoscopic recurrence was defined by a Rutgeerts' score ≥ i2 |
|
Surgical recurrence (Follow‐up: 102 weeks) |
242 per 1000 | 191 per 1000 (92 to 392) | RR 0.79 (0.38 to 1.62) |
133 (1 cohort study) |
⊕⊝⊝⊝d VERY LOW | Unadjusted risk ratio |
|
Mortality (Follow‐up: 102 weeks) |
‐ | ‐ | ‐ | 63 (1 RCT) |
‐ | No deaths were reported during the follow‐up period |
| Quality of life | ‐ | ‐ | ‐ | ‐ | ‐ | Outcome was not measured or reported |
|
Adverse effects (Follow‐up: 102 weeks) |
938 per 1000 | 544 per 1000 (394 to 769) |
RR 0.58 (0.42 to 0.82) |
63 (1 RCT) |
⊕⊝⊝⊝a,c,e VERY LOW | Common adverse effects included infections, gastrointestinal intolerance, leukopenia, pancreatitis, skin lesions and cancer. Perforations or haemorrhages secondary to colonoscopy were not reported |
|
CI 95%: 95% confidence interval (CI); RR: Risk ratio; GRADE: Evidence grades of the GRADE Working Group (see later); RCT: randomised controlled trial; CDAI: Crohn's disease activity index. *The risk WITH therapy immediately after surgery is based on the risk in the control group of the trials. The risk WITH colonoscopy‐guided therapy (and its confidence interval) is calculated from relative effect (and its confidence interval). | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect. | ||||||
a The certainty of the evidence was downgraded in one level for risk of bias because trial was open‐label and unclear risk of attrition bias. Despite the outcome assessor was blinded for the outcome endoscopic recurrence, the participants and personnel were not blinded
b The certainty of the evidence was downgraded in one level for imprecision because the result at each end of the confidence interval entails a different clinical decision and the sample size of the included study was lower than the calculated optimal information size.
c The certainty of the evidence was downgraded in one level for indirectness, because the current practice includes management with biologic therapy and this study only considered thiopurines.
d The certainty of the evidence was downgraded in one level for imprecision because only one study was included and the result at each end of the confidence interval entails a different clinical decision.
e The certainty of the evidence was downgraded in one level for imprecision because the sample size of the included study was lower than the calculated optimal information size.
Intensification of prophylactic therapy guided by colonoscopy versus intensification guided by clinical recurrence
Clinical recurrence
One RCT and three cohort studies addressed this comparison.
In the RCT De Cruz 2015, 128 participants were allocated to the intervention group (colonoscopy‐based management) and 56 to the control group (intensification guided by clinical recurrence). All participants received the same management immediately after surgery, however, 10 participants were excluded after randomisation because they did not receive the allocated medication (six in the intervention group and four in the control group). In the intervention group, the colonoscopy was performed six months after surgery and prophylactic therapy was intensified in case of endoscopic recurrence. After a follow‐up of 18 months and using as a criterion a Crohn's Disease Activity Index (CDAI) >150 points, 37.7% (46/122) of participants in the intervention group developed clinical recurrence versus 46.1% (24/52) in the control group (ITT analysis, risk ratio (RR): 0.82, 95% confidence interval (CI): 0.56 to 1.18, Analysis 1.1. The authors also measured clinical recurrence using as a criterion a CDAI > 200 points, reporting 27% (33/122) of clinical recurrence in the intervention group, compared to 40% (21/52) in the control group (ITT analysis, RR: 0.68, 95% CI: 0.44 to 1.06, Analysis 1.1).
1.1. Analysis.

Comparison 1: Intensification of prophylactic therapy guided by colonoscopic surveillance versus intensification guided by clinical recurrence, Outcome 1: Randomised trial (18 months of follow‐up)
In De Cruz 2015, the authors provided information to conduct a subgroup analysis according to clinical risk factors of recurrence. High risk of recurrence was defined as having one or more of the following factors: smoking, perforating disease or previous resection:
In the high‐risk subgroup, the risk of clinical recurrence (CDAI > 150 points) was 41.6% (42/101) in the colonoscopy‐based management group compared to 47.7% (21/44) in the control group (145 participants, ITT analysis, RR: 0.87, 95% CI: 0.59 to 1.28, Analysis 2.1). Using as a criterion a CDAI > 200 points, the risk of clinical recurrence was 29.7% (30/101) in the colonoscopy‐based management group, compared to 40.9% (18/44) in the control group (145 participants, ITT analysis, RR: 0.73, 95% CI: 0.46 to 1.16, Analysis 2.2).
In the low‐risk subgroup, the risk of clinical recurrence (CDAI > 150 points) was 19% (4/21) in the colonoscopy‐based management group compared to 37.5% (3/8) in the control group (29 participants, ITT analysis, RR: 0.51, 95% CI: 0.14 to 1.78, Analysis 2.1). Using as a criterion a CDAI > 200 points, the risk of clinical recurrence was 14.3% (3/21) in the colonoscopy‐based management group, compared to 37.5% (3/8) in the control group (29 participants, ITT analysis, RR: 0.38, 95% CI: 0.1o to 1.51, Analysis 2.2). Given how few participants were included in the low‐risk subgroup, the findings were only trends and they should be interpreted with caution.
2.1. Analysis.

Comparison 2: Subgroup analysis according to baseline risk: Intensification of prophylactic therapy guided by colonoscopic surveillance versus intensification guided by clinical recurrence, Outcome 1: Clinical recurrence (CDAI >150) at 18 months
2.2. Analysis.

Comparison 2: Subgroup analysis according to baseline risk: Intensification of prophylactic therapy guided by colonoscopic surveillance versus intensification guided by clinical recurrence, Outcome 2: Clinical recurrence (CDAI >200) at 18 months
Three cohort studies addressed this comparison. These studies were highly heterogeneous in terms of design, risk of bias and data reporting, therefore we did not conduct a meta‐analysis.
In De Cruz 2013, the risk of clinical recurrence was similar between the groups (136 participants, median follow‐up 53 months, clinical recurrence in the intervention group 48.6% (34/70), in the control group 48.5% (32/66); univariate analysis, RR: 1.00, 95% CI: 0.71 to 1.42, Analysis 1.2). This study had a high risk of selection and confounding bias and its findings should be interpreted with caution.
1.2. Analysis.

Comparison 1: Intensification of prophylactic therapy guided by colonoscopic surveillance versus intensification guided by clinical recurrence, Outcome 2: Cohort studies (50‐55 months of median follow‐up)
In Baudry 2014, 23.3% (21/90) of participants in the intervention group developed clinical recurrence versus 54.7% (23/42) in the control group (132 participants, median follow‐up of 51 months, univariate analysis, RR: 0.43, 95% CI: 0.27 to 0.68, Analysis 1.2). The median time to clinical recurrence was 25 months in the intervention group and 31 months in the control group. In the multivariable analysis, the management guided by symptoms was associated with a higher risk of clinical recurrence (adjusted odds ratio (OR): 1.9, 95% CI: 1.07 to 3.57). Factors included in the logistic regression model were the behavior of CD (penetrating versus non‐penetrating disease) and extraintestinal manifestations. Despite this, this study was judged as low methodological quality due to a high risk of selection bias.
In Boucher 2016, the authors did not report the proportion of participants that developed clinical recurrence into each group; the data were reported using time‐to‐event analyses. According to univariate analysis, the colonoscopy‐based management was associated with a delayed time to clinical recurrence (33.4 versus 84.5 months). According to the multivariable Cox proportional hazards regression model, the colonoscopy‐guided management decreased the risk of clinical recurrence compared to management guided by symptoms (161 participants, follow‐up ranging from six months to 20 years, adjusted hazard ratio (HR): 0.4, 95% CI: 0.25 to 0.66). This study was considered to have a low risk of selection and outcome assessment bias, however, the factors included in the multivariable regression model were not detailed in the published article.
In summary, the therapy guided by colonoscopy compared to therapy guided by symptoms may reduce postoperative clinical recurrence of CD (low‐certainty evidence) (Table 1). This statement is mainly based on the findings described in De Cruz 2015, The certainty of the evidence in this outcome was low because we downgraded one level due to risk of bias (the trial was open‐label and had an unclear risk of attrition bias), and one level due to imprecision (the result at each end of the confidence interval entails a different clinical decision). The certainty of the evidence given by the cohort studies was not better than the RCT. Despite this, two cohort studies with longer follow‐up and appropriate control of confounding and outcome assessment bias also showed the benefit of a colonoscopy‐guided therapy (Baudry 2014, Boucher 2016).
Endoscopic recurrence
One RCT (De Cruz 2015), reported endoscopic recurrence for this comparison. Using as a criterion a Rutgeerts score ≥ i2 the colonoscopy‐based management (intervention group) decreased the risk of endoscopic recurrence compared to intensification guided by symptoms (control group). At 18 months of follow‐up, the risk of endoscopic recurrence was 49.2% (60/122) in the intervention group compared to 67.3% (35/52) in the control group (174 participants, ITT analysis, RR: 0.73, 95% CI: 0.56 to 0.95, Analysis 1.1). Using as a criterion a Rutgeerts score ≥ i3, 11.5% (14/122) of participants in the intervention group and 15.4% (8/52) in the control group evolved with endoscopic recurrence (ITT analysis, RR: 0.75, 95% CI: 0.33 to 1.67, Analysis 1.1).
The authors provided information to conduct a subgroup analysis according to clinical risk factors of recurrence:
In the high‐risk subgroup (145 participants), the risk of endoscopic recurrence (Rutgeerts score ≥ i2) was lower in the colonoscopy‐based management group (49.5%, 50/101) compared to the control group (70.5%, 31/44) (ITT analysis, RR: 0.70, 95% CI: 0.53 to 0.92, Analysis 2.3),
In the low‐risk subgroup (29 participants), the endoscopic recurrence (Rutgeerts score ≥ i2) was lower in the colonoscopy‐based management group (47.6%, 10/21) compared to the control group (50%, 4/8) (ITT analysis, RR: 0.95, 95% CI: 0.42 to 2.17, Analysis 2.3). Given the scarce number of participants included in the low‐risk subgroup, the findings were only trends and they should be interpreted with caution.
2.3. Analysis.

Comparison 2: Subgroup analysis according to baseline risk: Intensification of prophylactic therapy guided by colonoscopic surveillance versus intensification guided by clinical recurrence, Outcome 3: Endoscopic recurrence (Rutgeerts' score ≥i2) at 18 months
Included cohort studies did not report endoscopic recurrence as an outcome.
In summary, the therapy guided by colonoscopy compared to therapy guided by symptoms may reduce postoperative endoscopic recurrence of CD (low‐certainty evidence) (Table 1). This statement is based on the findings described in De Cruz 2015. The certainty of the evidence in this outcome was low because we downgraded one level due to risk of bias (the participants and personnel were not blinded and unclear risk of attrition bias), and one level due to imprecision (the confidence interval was wide and is based in only one study).
Surgical recurrence
Four cohort studies (Baudry 2014, Boucher 2016; De Cruz 2013 and Guo 2018) reported surgical recurrence for this comparison. They were highly heterogeneous therefore a meta‐analysis was not conducted.
In two studies of low methodological quality (Baudry 2014 and De Cruz 2013), the authors only conducted univariate analyses. In both studies, the effect of the colonoscopy‐based management was uncertain. After a median follow‐up of 51 months, Baudry 2014 reported that 5.6% (5/90) of participants in the intervention group developed surgical recurrence versus 9.5% (4/42) in the control group (unadjusted RR: 0.58, 95% CI 0.17 to 2.06, Analysis 1.2). Similarly, De Cruz 2013 reported that 8.6% (6/70) of participants in the intervention group developed surgical recurrence versus 4.5% (3/66) in the control group (median follow‐up 53 months, unadjusted RR: 1.89, 95% CI 0.49 to 7.23, Analysis 1.2).
In two studies, of moderate‐ (Boucher 2016) and low‐methodological quality (Guo 2018), the authors conducted multivariable analyses (Cox proportional hazards regression models). In both studies, colonoscopy‐based management seemed to decrease the risk of surgical recurrence. In Boucher 2016 the data were only reported as time‐to‐event analysis. According to this study, the colonoscopy‐based management seemed to decrease the risk of surgical recurrence (161 participants, follow‐up ranging from six months to 20 years, adjusted HR: 0.30, 95% CI: 0.13 to 0.70). Factors included in the regression model were not reported. In Guo 2018, the risk of surgical recurrence was similar across the groups in the univariate time‐to‐event analysis (75 participants, median follow‐up of 50 to 55 months, log rank test, P = 0.432). In the high risk of recurrence subgroup, the colonoscopy‐guided management seemed to decrease the risk of surgical recurrence in uni‐ and multivariable analyses (66 participants, 17.9% (7/39) in the intervention group versus 48.1% (13/27) in the control group, unadjusted RR: 0.37, 95% CI: 0.17 to 0.81 (Analysis 2.4); adjusted HR: 0.35, 95% CI: 0.14 to 0.89). Age at diagnosis of CD and type of management after surgery were included in the regression model.
2.4. Analysis.

Comparison 2: Subgroup analysis according to baseline risk: Intensification of prophylactic therapy guided by colonoscopic surveillance versus intensification guided by clinical recurrence, Outcome 4: Surgical recurrence (cohort study, 50‐55 months of median follow‐up)
Participants included in the RCT (De Cruz 2015) did not evolve with surgical recurrence (18 months of follow‐up).
In summary, we are uncertain whether the therapy guided by colonoscopy compared to therapy guided by symptoms reduces surgical recurrence as the certainty of the evidence is very low (Table 1). Although the cohort study with the highest methodological quality showed a benefit of colonoscopy‐guided management (Boucher 2016), the certainty of the evidence in this outcome was very low due to all studies were observational design and their results were inconsistent.
Mortality for any cause
Only in De Cruz 2015 did the authors directly reported mortality. In this study, there was no mortality during the follow‐up at 18 months.
Adverse events
Adverse events were reported according to the definitions used by the authors from two studies, a RCT (De Cruz 2015) and a cohort study (Guo 2018). In De Cruz 2015, the risk of adverse events, minor or severe, was similar between the groups, 82% in the intervention group (100/122) and 86.5% in the control group (45/52) (ITT analysis, RR; 0.95, 95% CI:0.83 to 1.08, Analysis 1.1). The risk of severe adverse events was slightly lower in the colonoscopy‐guided management, 27% in the intervention group (33/122), 34.6% in the control group (18/52) (ITT analysis, RR; 0.78, 95% CI:0.49 to 1.25, Analysis 1.1), despite the participants in the intervention group receiving thiopurines and/or adalimumab earlier than participants in the control group. The most important were drug intolerance (35 participants using metronidazole), sensory symptoms (three participants using metronidazole), alopecia, headache or wound infection (three participants using azathioprine), lupus, leukocytoclastic vasculitis, and severe injection site reaction (four participants using biologic therapy). Other details related to the adverse events were not available.
In Guo 2018, the authors reported azathioprine withdrawal due to adverse reactions. In this study; the risk seemed to be higher in the colonoscopy‐guided management, but this result should be interpreted with caution because the number of events was very low (4.8% in the colonoscopy‐guided group (2/42) and 0% in the control group (0/33) (RR 3.95; 95% CI: 0.20 to 79.64; Analysis 1.2)
Adverse effects of colonoscopy, such as perforations or haemorrhages, were not measured or reported.
In summary, there may be little or no difference in adverse events with intensification of therapy guided by colonoscopy when compared to intensification guided by symptoms (low‐certainty evidence).(Table 1). This statement is mainly based on the findings described in De Cruz 2015, The certainty of the evidence in this outcome was low because we downgraded one level due to risk of bias (the trial was open‐label and unclear risk of attrition bias), and one level due to imprecision (the result at each end of the confidence interval entails a different clinical decision).
Crohn's Disease Activity Index (CDAI), Harvey Bradshaw index score and Quality of life
There was not enough information in the included studies to perform these analyses.
Initiation of prophylactic therapy guided by colonoscopy versus initiation of prophylactic therapy immediately after surgery
Clinical recurrence
One RCT and one cohort study addressed this comparison. In the RCT (Ferrante 2015), 31 participants were allocated to the colonoscopy‐based management group (intervention group) and 32 received therapy immediately after surgery (control group). Only participants with a high risk of recurrence were included. In the intervention group, the colonoscopy was performed at six and 12 months after surgery and prophylactic therapy was delayed up‐to detection of endoscopic recurrence. Using a CDAI > 150 points as a criterion of clinical recurrence, 58% (18/31) of participants in the intervention group developed clinical recurrence until week 102, compared to 50% (16/32) in the control group (ITT analysis, RR: 1.16, 95% CI: 0.73 to 1.84, Analysis 3.1).
3.1. Analysis.

Comparison 3: Initiation of prophylactic therapy guided by colonoscopic surveillance versus initiation of prophylactic therapy immediately after surgery, Outcome 1: Randomised trial (102 weeks of follow‐up)
Bordeianou 2011 was a cohort study of low methodological quality. In this study 199 participants were included, 130 received colonoscopy‐based management (32 started the therapy after endoscopic recurrence and 98 never required therapy), and 69 started therapy immediately after surgery (control group). After a median follow‐up of 32 months, the effect of timing of therapy (colonoscopy‐based management or immediately after surgery) was uncertain. In the univariate analysis, 24.6% (32/130) of participants in the colonoscopy‐based management group developed clinical recurrence compared to 28.9% (20/69) in the control group (unadjusted RR: 0.85, 95% CI: 0.53 to 1.37, Analysis 3.2). According to a multivariable time‐to‐event analysis, the timing of therapy was not considered a predictor of clinical recurrence (Cox proportional hazards regression model, therapy after endoscopic recurrence versus no therapy, adjusted HR: 1.36, 95% CI: 0.66 to 2.78; therapy immediately after surgery versus no therapy, adjusted HR: 1.33, 95% CI: 0.63 to 2.76). The factors included in the regression model were age, Charlson comorbidity index, type of prophylactic therapy, prior resection, active disease (within surgical specimen) and type of anastomosis.
3.2. Analysis.

Comparison 3: Initiation of prophylactic therapy guided by colonoscopic surveillance versus initiation of prophylactic therapy immediately after surgery, Outcome 2: Cohort studies
In summary, we are uncertain whether initiation of therapy guided by colonoscopy compared to therapy immediately after surgery impacts clinical recurrence as the certainty of the evidence is very low. (Table 2). This statement is mainly based on the findings described in Ferrante 2015. The certainty of evidence in this outcome was very low because we downgraded one level due to risk of bias (the trial was open‐label with an unclear risk of attrition bias), one level due to imprecision (the result at each end of the confidence interval entails a different clinical decision and the sample size was lower than the calculated optimal information size), and one level due to for indirectness (the current practice includes management with biologic therapy and this study only considered thiopurines). One cohort study also addressed this comparison, however, its certainty of the evidence was not better than the RCT (Ferrante 2015).
Endoscopic recurrence
One RCT (Ferrante 2015) and one cohort study (Bordeianou 2011) reported endoscopic recurrence for this comparison. Using as a criterion a Rutgeerts score ≥ i2, Ferrante 2015 showed that 58.1% (18/31) of participants in the colonoscopy‐based management group (intervention group) and 50% (16/32) of participants that received therapy immediately after surgery (control group) developed endoscopic recurrence at week 102 (ITT analysis, RR:1.16, 95% CI: 0.73 to 1.84, Analysis 3.1). Using as a criterion a Rutgeerts score ≥ i3 the results were similar, 45.2% (14/31) of participants in the intervention group and 43.7% (14/32) in the control group developed endoscopic recurrence at week 102 (ITT analysis, RR: 1.03, 95% CI: 0.59 to 1.79, Analysis 3.1).
In a cohort study of low methodological quality (Bordeianou 2011), the effect of timing of prophylactic therapy (immediately after surgery or colonoscopy‐based management) on endoscopic recurrence was similar. The authors did not report the proportion of participants that developed endoscopic recurrence in each group, the data were reported using time‐to‐event analyses. In the univariate and multivariable analysis, the timing of prophylactic therapy was not considered a predictor of endoscopic recurrence (199 participants, median follow‐up 32 months, therapy immediately after surgery versus colonoscopy‐based management, unadjusted HR: 0.71, 95% CI: 0.45 to 1.23; adjusted HR: 0.79, 95% CI: 0.49 to 1.31). The factors included in the Cox proportional hazards regression model were age, Charlson comorbidity index, prior resection, active disease (within surgical specimen) and type of anastomosis.
In summary, we are uncertain whether initiation of therapy guided by colonoscopy compared to therapy immediately after surgery impacts endoscopic recurrence as the certainty of the evidence is very low. (Table 2). This statement is mainly based on the findings described in Ferrante 2015. The certainty of evidence in this outcome was very low because we downgraded one level due to risk of bias (the trial was an open‐label with an unclear risk of attrition bias), one level due to imprecision (the result at each end of the confidence interval entails a different clinical decision and the sample size was lower than the calculated optimal information size), and one level due to indirectness (current practice includes management with biologic therapy and this study only considered thiopurines). One cohort study also addressed this comparison however its certainty of the evidence was not better than the RCT (Ferrante 2015).
Surgical recurrence
One cohort study of low methodological quality reported surgical recurrence for this comparison. In Guo 2018, the risk of surgical recurrence was similar between the groups in the univariate analysis (133 participants, median follow‐up of 50 to 55 months, 19% (8/42) in the intervention group versus 24.2% (22/91) in the control group, unadjusted RR: 0.79, 95% CI: 0.38 to 1.62 (Analysis 3.2)). In the high risk of recurrence subgroup, the risk of surgical recurrence was also similar between the groups (108 participants, 17.9% (7/39) in the intervention group versus 24.6% (17/69) in the control group, unadjusted RR: 0.73, 95% CI: 0.33 to 1.60 (Analysis 4.1); adjusted HR: 0.75, 95% CI: 0.32 to 1.78). Age at diagnosis of CD and type of management after surgery were included in the regression model.
4.1. Analysis.

Comparison 4: Subgroup analysis according to baseline risk: Initiation of prophylactic therapy guided by colonoscopic surveillance versus initiation of prophylactic therapy immediately after surgery, Outcome 1: Surgical recurrence (cohort study, 50 months of median follow‐up)
Participants included in Ferrante 2015 did not develop surgical recurrence (102 weeks of follow‐up). The authors of Bordeianou 2011 did not report this outcome.
In summary, we are uncertain whether initiation of therapy guided by colonoscopy compared to therapy immediately after surgery impacts surgical recurrence as the certainty of the evidence is very low. (Table 2).This statement is based on findings described in Guo 2018. The certainty of evidence in this outcome was very low because the study was observational design and we downgraded one level due to imprecision (only one study was included and the result at each end of the confidence interval entails a different clinical decision).
Mortality for any cause
Only in Ferrante 2015 did the authors directly report mortality. In this study, there was no mortality during the follow‐up at 102 weeks.
Adverse effects
Adverse events were reported according to the definitions used by the authors from two studies, one RCT (Ferrante 2015) and one cohort study (Guo 2018).
In Ferrante 2015, six participants withdrew from the study due to adverse events, all of them were under azathioprine therapy at the time of withdrawal. One participant developed azathioprine‐induced leucopenia and one participant developed azathioprine‐induced pancreatitis, both in the therapy immediately after surgery group. Two participants discontinued therapy due to gastrointestinal intolerance, one due to weight gain and one due to thyroid cancer. Ten participants in the intervention group and 16 participants in the control group developed infections, none of them withdrew from the study. Considering all adverse events reported (infections, gastrointestinal intolerance, leucopenia, pancreatitis, skin lesions, cancer and miscellaneous) therapy guided by colonoscopy was associated with a lower risk of adverse events than therapy immediately after surgery (ITT analysis, colonoscopy‐based management group 54.8% (17/31), control group 93.8% (30/32), RR; 0.58, 95% CI:0.42 to 0.82 Analysis 3.1). Despite this, only six participants discontinued the study prematurely due to adverse events while taking the prophylactic therapy (9.5%, 6/63 participants)
In Guo 2018, the authors reported azathioprine withdrawal due to adverse reactions. In this study, the risk was similar between the groups (4.8% in the colonoscopy‐based management group (2/42) and 5.5% in the immediately after surgery group (5/91) (RR; 0.87; CI 95%: 0.18 to 4.29, Analysis 3.2)
Adverse effects of colonoscopy, such as perforations or haemorrhages, were not measured or reported.
In summary, we are uncertain whether initiation of therapy guided by colonoscopy compared to therapy immediately after surgery impacts adverse effects as the certainty of the evidence is very low. (Table 2). This statement is mainly based on the findings described in Ferrante 2015. The certainty of evidence in this outcome was very low because we downgraded one level due to risk of bias (the trial was an open‐label with an unclear risk of attrition bias), one level due to imprecision (the sample size was lower than the calculated optimal information size), and one level due to indirectness (current practice includes management with biologic therapy and this study only considered thiopurines). One cohort study also addressed this comparison, however the certainty of the evidence was not better than the RCT (Ferrante 2015).
Crohn's Disease Activity Index (CDAI), Harvey Bradshaw index score and Quality of life
There was not enough information in the included studies to perform these analyses.
Subgroup analysis
The high heterogeneity and the scarce amount of studies preclude performing a subgroup analysis according to the type of prophylactic therapy. In all included studies colonoscopy was performed within one year after surgery, therefore a subgroup analysis according to the timing of colonoscopic surveillance was not conducted.
Discussion
Summary of main results
Two comparisons using colonoscopy‐guided management of postoperative of Crohn's disease (CD) were identified in this review.
Intensification of prophylactic therapy guided by colonoscopy versus intensification guided by clinical recurrence
The main results for this comparison were that intensification of prophylactic therapy guided by colonoscopy may reduce postoperative endoscopic and clinical recurrence when compared to intensification guided by symptoms. Although participants in the intervention group received thiopurines and/or biologic therapy earlier than participants in the control group, colonoscopy‐guided management may not increase the risk of severe adverse events when compared to intensification guided by symptoms. In terms of surgical recurrence, we are uncertain whether the management guided by colonoscopy has any impact when compared to therapy guided by symptoms as the certainty of the evidence is very low, because the studies were only observational in design and with inconsistent results. (Table 1).
In terms of clinical recurrence, the one randomised controlled trial (RCT) included in this comparison showed there may be benefit with colonoscopy‐based management (De Cruz 2015), although the certainty of the evidence is low and the confidence interval included the possibility of harm. Despite this, in the opinion of the review authors the overall evidence suggests that colonoscopy‐based management may decrease the risk of clinical recurrence when compared to management guided by symptoms.The reasons supporting this conclusion are summarised below.
The point estimator presented an appropriate benefit in favour of the colonoscopy‐guided management. In terms of absolute difference, 84 less patients experience clinical recurrence in the intervention group at 18 months of follow‐up, assuming a recurrence risk of 46.2% in the control group, a population of 1000 patients per arm, and using as a criterion a Crohn's Disease Activity Index (CDAI) > 150 points (Table 1).
There is an appropriate correlation between different forms of clinical recurrence measurement. If we use as a criterion a CDAI > 200 points, the benefit is stronger. In terms of absolute difference, 147 less patients experience clinical recurrence in the intervention group at 18 months of follow‐up, assuming a risk of 46.2% in the control group and a population of 1000 patients per arm.
The number of participants and the follow‐up considered in the RCT were probably insufficient to evaluate clinical recurrence. The primary outcome for this RCT was endoscopic recurrence, therefore the sample size was calculated according to this outcome. Considering this, it is difficult to have accurate results for the outcome clinical recurrence.
Current evidence suggests that endoscopic recurrence is a strong predictor of clinical recurrence and poor prognosis (Baert 2010; Colombel 2014; D'Haens 2008; Frøslie 2007; Rutgeerts 1990; Schintzler 2009, Ungaro 2020). It is therefore appropriate to expect that methods that use colonoscopy to identify endoscopic recurrence will in turn reduce cases of clinical recurrence and poor prognostic outcome. Although this outcome should be considered an intermediate outcome according to a recent classification (Guyatt 2011), expert consensus has established that maintaining a healthy mucosa is a desirable target in CD management (Peyrin‐Biroulet 2015). The findings of this review showed a benefit in terms of endoscopic recurrence (Analysis 1.1), which also supports the benefit in terms of clinical recurrence, although this was low‐certainty evidence.
Finally, evidence from cohort studies with longer follow‐up and an appropriate control of confounding and outcome assessment bias showed that colonoscopy‐based management decreased the risk of clinical recurrence (Baudry 2014, Boucher 2016).
Initiation of prophylactic therapy guided by colonoscopy versus initiation immediately after surgery
The main results for this comparison were that we are uncertain whether initiation of therapy guided by colonoscopy compared to therapy immediately after surgery impacts clinical, endoscopic or surgical recurrence or adverse effects, as the certainty of the evidence is very low. The limitations of the available evidence included risk of bias, imprecision and indirectness. Considering this, it was not possible to make a definitive judgment of the data, therefore the decision should be based on other important elements for decision‐making as proposed by the GRADE group (endoscopy safety, availability, cost, values and patient preferences, etc).
In relation to the other outcomes considered by this review, i.e. mortality and quality of life, the limited information available in the included studies did not allow making conclusions.
Overall completeness and applicability of evidence
The aim of this review was to assess whether the management guided by endoscopic recurrence improved the prognosis of postoperative CD. Unfortunately, the assessment of this intervention is complex, because the management of postoperative CD has been highly heterogeneous. The heterogeneity is given by the pharmacological alternatives, ranging from 5‐ASA and thiopurines to biologic therapy, by the timing when these drugs are started or intensified and the timing of colonoscopic surveillance. To capture all studies evaluating the effect of colonoscopy‐guided management, we used wide inclusion criteria, with no restrictions in terms of the type of baseline management, modifications of the therapy after detection of endoscopic recurrence or the drugs used for prevention. Additionally, our review was not restricted to RCTs, we also considered cohort studies. Despite this, the evidence addressing this issue was scarce and only seven studies met our inclusion criteria. Three strategies of prophylactic therapy were detected, management guided by clinical symptoms, management guided by endoscopic recurrence and full‐management immediately after surgery. According to our findings, the management guided by endoscopic recurrence seems to be superior to the management guided by clinical symptoms, decreasing the risk of recurrence without increasing adverse effects. In general, the drugs used in the groups under comparison were the same, therefore the difference was the timing when these drugs were started or intensified. Probably an accelerated step‐up therapy in participants with asymptomatic endoscopic recurrence explains these findings, however, a GRADE analysis showed that the certainty of this evidence was low for clinical recurrence, endoscopic recurrence and serious adverse effects. In relation to the second comparison, we are uncertain whether the management guided by endoscopic recurrence compared with therapy immediately after surgery impacts the risk of recurrence and adverse effects as the certainty of the evidence is very low. One of the main issues is that biologic therapy was not considered in the study with the best design. In this context, further studies are necessary to answer this question, especially comparing the initiation of anti‐TNFα drugs immediately after surgery versus a delayed initiation guided by endoscopic recurrence.
Considering the available evidence, involving patients in decision‐making is essential, especially for the second comparison. For example, for patients more worried about adverse effects and costs of drugs, strategies focused on a delayed therapy may be a better option. On the other hand, for patients more worried about CD reactivation than side effects of drugs, the best option could be start therapy immediately after surgery. In our opinion, the optimal approach to the management of post‐operative CD should consider the patients' perspective.
Quality of the evidence
The results found in this review should be interpreted with caution because the evidence has important limitations in both comparisons and all outcomes.
Intensification of prophylactic therapy guided by colonoscopy versus intensification guided by clinical recurrence
In this comparison, the global certainty of the evidence was low and very low (Table 1).The following reasons are given for or downgrading the evidence.
Risk of bias: the RCT included in this comparison (De Cruz 2015) was open‐label and the risk of attrition bias was unclear, due to the high rate of loss of follow‐up, and 10 patients were excluded after randomisation (they never received the allocated therapy). Despite the outcome assessor being blinded for the outcome endoscopic recurrence, the participants and personnel were not blinded.
Imprecision: in most of outcomes (clinical recurrence and severe adverse effect), the result at each end of the confidence interval entails a different clinical decision. Additionally, the results in endoscopic recurrence were imprecise because only one study assessed this outcome and the confidence interval was wide.
Inconsistency: in the surgical outcome, the observational studies were highly heterogeneous (different pharmacological interventions, follow‐up, characteristics of participants) and their results showed opposite conclusions.
Observational design: the cohort studies included in this comparison were of low or moderate methodological quality according to the Newcastle‐Ottawa Scale (NOS). Specifically, the evidence on surgical recurrence only came from cohort studies with inconsistent results.
The interpretation of these results should be carried out with caution since they could change in the near future, especially with new RCTs.
Initiation of prophylactic therapy guided by colonoscopy versus initiation of prophylactic therapy immediately after surgery
In this comparison, the global certainty of the evidence was very low (Table 2). The reasons for downgraded the evidence were:
Risk of bias: The RCT included in this comparison (Ferrante 2015) was open‐label and the risk of attrition bias was unclear, due to the high rate of loss of follow‐up. Despite the outcome assessor was blinded for the outcome endoscopic recurrence, the participants and personnel were not blinded.
Imprecision: In the most of outcomes (clinical recurrence, endoscopic recurrence and surgical recurrence), the result at each end of the confidence interval entails a different clinical decision, and the sample size of the RCT was lower than the calculated optimal information size.
Indirectness: The RCT (Ferrante 2015) did not included the overall spectrum of drugs available in the current practice. The study only considered thiopurines and current management also include biologic therapy
Observational design: The cohort studies included in this comparison were of low methodological quality according to the NOS. Specifically, the results of surgical recurrence only came from cohort studies.
In this comparison the limitations are important, therefore the findings should be interpreted with caution.
Potential biases in the review process
This review was conducted with the highest quality standards to reduce the risk of bias. The review process was performed by a comprehensive literature search to identify all eligible studies, without limitations in languages or publication status. In addition, two review authors independently assessed each study for inclusion, extracted data and assessed study quality. Despite this, this review has limitations. The two comparisons analysed in this review were not contemplated in the protocol, which may induce bias, even when the authors consider that the analyses are clinically appropriate. Furthermore, due to the low number of studies, several planned sensitivity analyses were not possible, making it difficult to interpret the impact on the results of the potential bias of each study. Also, there is a very limited judgment of the potential publication bias of the studies.
Agreements and disagreements with other studies or reviews
As far as we know this is the first systematic review to focus on the efficacy of colonoscopy‐based management of postoperative CD. This situation is probably related to the nature of the intervention, which is highly heterogeneous. The effect of colonoscopy‐based management depends on the efficacy of the available drugs, the timing and frequency of colonoscopy after surgery and the characteristics of the control group, among other potential sources of heterogeneity. Despite these difficulties, our review is the first that summarises the evidence supporting the use of endoscopic‐recurrence as a criterion to start or intensify the pharmacological management of postoperative CD.
Authors' conclusions
Implications for practice.
Based on the findings of this review, the intensification of prophylactic therapy guided by colonoscopy may reduce clinical recurrence when compared to therapy guided by symptoms. Similarly, this strategy may reduce endoscopic recurrence, which is a good predictor of poor prognosis in Chrohn's disease, without increasing the risk of adverse events. However, certainty of the evidence is low. Considering this, management guided by colonoscopy may be an alternative for the prevention of postoperative recurrence of Crohn's disease when compared to management guided only by symptoms, but the real magnitude of the effect is uncertain. Given the low certainty of the evidence, decision‐making should take in to account patients' preferences, costs and availability of pharmacological alternatives and colonoscopic surveillance. In the case of initiation of therapy immediately after surgery compared to a delayed initiation guided by colonoscopy, there is more uncertainty. The main hypothesis against immediate initiation is to expose patients to significant adverse effects and costs with marginal benefit in terms of clinical recurrence. The findings of this review support this, however, the certainty of the evidence is very low, due to limitations in terms of risk of bias, imprecision and indirectness. Specifically, the participants in the only one randomised controlled trial (RCT) included in this analysis did not receive biologic therapy, which does not reflect current clinical practice.
Implications for research.
According to the findings of this review, further studies are required to establish with more certainty the effect of colonoscopy‐guided management for the prevention of postoperative recurrence of CD. New randomised trials could increase the precision of the results and correct the risk of bias, especially the risk of attrition bias detected in the RCTs included in this review. Specifically for the comparison initiation of therapy guided by colonoscopy versus initiation immediately after surgery, it is necessary to conduct studies including biologic therapy, which is the current standard in clinical practice. Additionally, it is reasonable that the new studies incorporate the quality of life outcome, which was not reported in the studies analysed in this review. In terms of cost‐effectiveness analysis, this review allows updating or carrying out new economic assessments, especially considering clinical guideline contexts. In this sense, a network meta‐analysis comparing the three alternatives simultaneously could provide additional information to aid decision‐making. Finally, the authors of this review did not find a defined core outcome set for this question nor minimal clinically important difference for the main outcomes. Both can be challenging to work with organisations of patients with Crohn's disease.
History
Protocol first published: Issue 8, 2016 Review first published: Issue 8, 2020
Acknowledgements
We would like to acknowledge to Elizabeth Uleryk for her assistance in the design of the search strategy and Centro de Evidencia UC for its assistance in the search process.
Appendices
Appendix 1. MEDLINE search strategy
1. exp Crohn Disease/ or crohn*.mp.
2. ileitis.mp. or exp Ileitis/
3. inflammatory bowel disease.mp. or exp Inflammatory Bowel Diseases/
4. 1 or 2 or 3
5. Postoperative Period/
6. postoperativ*.mp.
7. surger*.mp.
8. surgic*.mp.
9. postsurgical*.mp.
10. postsurger*.mp.
11. "post‐surgery".mp.
12. "post‐surgical*".mp.
13. "post‐operative*".mp.
14. (bowel adj2 resection*).mp.
15. (intestin* adj2 resection*).mp.
16. (colo* adj2 resection*).mp.
17. operation*.mp.
18. 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17
19. Crohn Disease/su [Surgery]
20. exp Ileitis/su [Surgery]
21. 19 or 20
22. 4 and 18
23. 21 or 22
24. colonoscop*.mp. or exp Colonoscopy/
25. sigmoidoscop*.mp. or exp Sigmoidoscopy/
26. endoscop*.mp. or exp Endoscopy/
27. exp Secondary Prevention/ or prevent*.mp.
28. prophyl*.mp.
29. 24 or 25 or 26 or 27 or 28
30. (intestinal adj2 mucosa*).mp. or exp Intestinal Mucosa/
31. (colo* adj2 mucosa*).mp.
32. ((mucos* or endoscop* or intestin* or colo*) adj3 (recur* or relaps* or heal* or remission* or improv*)).mp.
33. exp Recurrence/ or recur*.mp.
34. relaps*.mp.
35. remission*.mp.
36. 30 or 31 or 32 or 33 or 35
37. 23 and 29 and 36
38. (clinical trial or clinical trial, all).pt. or clinical trials as topic/
39. clinical trial, phase i.pt. or clinical trials, phase i as topic/
40. clinical trial, phase ii.pt. or clinical trials, phase ii as topic/
41. clinical trial, phase iii.pt. or clinical trials, phase iii as topic/
42. clinical trial, phase iv.pt. or clinical trials, phase iv as topic/
43. controlled clinical trial.pt. or controlled clinical trials as topic/
44. meta analysis.pt. or meta analysis as topic/
45. multicentre study.pt. or multicenter studies as topic/
46. randomized controlled trial.pt. or randomized controlled trials as topic/
47. pragmatic clinical trial.pt. or Pragmatic Clinical Trials as Topic/ or ((preference or practical or pragmatic or "real world" or naturalistic) adj5 trial*).ti,ab.
48. Comparative Effectiveness Research/ or ((comparative adj2 effectiveness) or (CER adj5 (research* or method* or framework* or compari* or statement*))).ti,ab.
49. ((clin: adj 5 trial:) or ((singl: or doubl: or tripl: or trebl:) and (mask: or blind:))).ti,ab.
50. ((random: adj5 trial:) or rct or rcts).ti,ab.
51. or/38‐50
52. cohort studies/ or longitudinal studies/ or follow‐up studies/ or prospective studies/ or retrospective studies/
53. 51 or 52
54. 37 and 53
Appendix 2. Embase search strategy
1. exp Crohn disease/ or crohn*.mp.
2. exp ileitis/ or ileitis.mp.
3. inflammatory bowel disease.mp. or inflammatory bowel disease/
4. 1 or 2 or 3
5. postoperative period/
6. postoperativ*.mp.
7. surger*.mp. or surgery/
8. surgic*.mp.
9. postsurgical*.mp.
10. postsurger*.mp.
11. "post‐surger*".mp.
12. "post‐surgical*".mp.
13. "post‐operative*".mp.
14. intestine resection/
15. (intestin* adj2 resection*).mp.
16. (bowel adj2 resection*).mp.
17. (colo* adj2 resection*).mp.
18. operation.mp.
19. 5 or 6 or 7 or 8 or 9 or 10 or 11 or 12 or 13 or 14 or 15 or 16 or 17 or 18
20. exp Crohn disease/su [Surgery]
21. exp ileitis/su [Surgery]
22. 4 and 19
23. 20 or 21
24. 22 or 23
25. colonoscop*.mp. or exp colonoscopy/
26. sigmoidoscop*.mp. or exp sigmoidoscopy/
27. exp endoscopy/ or exp gastrointestinal endoscopy/ or endoscop*.mp.
28. exp secondary prevention/ or exp prevention/ or exp prevention study/ or prevent*.mp.
29. exp prophylaxis/ or prophyl*.mp.
30. 25 or 26 or 27 or 28 or 29
31. (intestinal adj2 mucosa*).mp. or exp intestine mucosa/
32. (colo* adj2 mucosa*).mp.
33. ((mucos* or endoscop* or intestin* or colo*) adj3 (recur* or relaps* or heal* or remission* or improv*)).mp.
34. recurre*.mp. or exp recurrent disease/
35. relaps*.mp.
36. remission*.mp.
37. 31 or 32 or 33 or 34 or 36
38. 24 and 30 and 37
39. ct.fs.
40. clinical trial.pt. or clinical trial/
41. phase 1 clinical trial.pt. or phase 1 clinical trial/
42. phase 2 clinical trial.pt. or phase 2 clinical trial/
43. phase 3 clinical trial.pt. or phase 3 clinical trial/
44. phase 4 clinical trial.pt. or phase 4 clinical trial/
45. controlled clinical trial.pt. or controlled clinical trial/
46. meta analysis/
47. multicenter study/
48. randomized controlled trial.pt. or randomized controlled trial/
49. crossover procedure/
50. outcomes research/
51. double blind procedure/
52. single‐blind procedure/
53. triple blind procedure/
54. ((preference or practical or pragmatic or "real world" or naturalistic) adj5 trial*).ti,ab.
55. comparative effectiveness/ or ((comparative adj2 effectiveness) or (CER adj5 (research* or method* or framework* or compari* or statement*))).ti,ab.
56. ((clin adj10 trial*) or ((singl* or doubl* or tripl* or trebl*) and (random* or blind* or mask*)) or (sham or placebo8 or multicenter* or multicentre: or metaanalys*) or (meta adj2 analys*)).mp.
57. (cochrane or medline or cinahl or embase or CCTR or scopus or "web of science" or lilacs).ti,ab.
58. or/39‐57
59. cohort analysis/ or longitudinal study/ or prospective study/ or retrospective study/
60. 58 or 59
61. 38 and 60
Appendix 3. CENTRAL search strategy
#1 MeSH descriptor: [Crohn Disease] explode all trees
#2 MeSH descriptor: [Ileitis] explode all trees
#3 MeSH descriptor: [Inflammatory Bowel Diseases] explode all trees
#4 "crohn*" or "ileitis" or "Inflammatory Bowel Disease*"
#5 #1 or #2 or #3 or #4
#6 MeSH descriptor: [Postoperative Period] explode all trees
#7 "postoperativ*" or "surger*" or "surgic*" or "postsurgical*" or "postsurger*" or "post‐surger*" or "post‐surgical*" or "post‐operativ*" or "bowel resection*" or "intestin* resection*" or "colo* resection*" or "operation*"
#8 #6 or #7
#9 MeSH descriptor: [Crohn Disease] explode all trees and with qualifier(s): [Surgery ‐ SU]
#10 MeSH descriptor: [Ileitis] explode all trees and with qualifier(s): [Surgery ‐ SU]
#11 #9 or #10
#12 #5 and #8
#13 #11 or #12
#14 MeSH descriptor: [Colonoscopy] explode all trees
#15 MeSH descriptor: [Sigmoidoscopy] explode all trees
#16 MeSH descriptor: [Endoscopy] explode all trees
#17 "colonoscop*" or "sigmoidoscop*" or "endoscop*"
#18 MeSH descriptor: [Secondary Prevention] explode all trees
#19 "prevent*" or "prophyl*"
#20 #14 or #15 or #16 or #17 or #18 or #19
#21 (intestinal adj2 mucosa*)
#22 (colo* adj2 mucosa*)
#23 ((mucos* or endoscop* or intestin* or colo*) adj3 (recur* or relaps* or heal* or remission* or improv*))
#24 MeSH descriptor: [Recurrence] explode all trees
#25 "recur*" or "relaps*" or "remission*"
#26 #21 or #22 or #23 or #24 or #25
#27 #13 and #20 and #26
Appendix 4. Clinical Trials.gov search strategy
#1. Post‐operative recurrence and Crohn’s Disease
#2. Colonoscopy and Crohn’s Disease
Appendix 5. WHO trial registry search strategy
#1. Post‐operative recurrence and Crohn’s Disease
#2. Colonoscopy and Crohn’s Disease
Appendix 6. Cochrane IBD specialized register search strategy
#1. Post‐operative recurrence and Crohn’s Disease
#2. Colonoscopy and Crohn’s Diseas
Data and analyses
Comparison 1. Intensification of prophylactic therapy guided by colonoscopic surveillance versus intensification guided by clinical recurrence.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Randomised trial (18 months of follow‐up) | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.1.1 Clinical recurrence (CDAI > 150) at 18 months | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.1.2 Clinical recurrence (CDAI >200) at 18 months | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.1.3 Endoscopic recurrence (Rutgeerts' score ≥i2) at 18 months | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.1.4 Endoscopic recurrence (Rutgeerts' score ≥i3) at 18 months | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.1.5 Adverse events at 18 months | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.1.6 Severe adverse events at 18 months | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.2 Cohort studies (50‐55 months of median follow‐up) | 3 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.2.1 Clinical recurrence | 2 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.2.2 Surgical recurrence | 3 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 1.2.3 Azathioprine withdrawal due to adverse event | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected |
Comparison 2. Subgroup analysis according to baseline risk: Intensification of prophylactic therapy guided by colonoscopic surveillance versus intensification guided by clinical recurrence.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 2.1 Clinical recurrence (CDAI >150) at 18 months | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.1.1 High‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.1.2 Low‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.2 Clinical recurrence (CDAI >200) at 18 months | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.2.1 High‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.2.2 Low‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.3 Endoscopic recurrence (Rutgeerts' score ≥i2) at 18 months | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.3.1 High‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.3.2 Low‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.4 Surgical recurrence (cohort study, 50‐55 months of median follow‐up) | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.4.1 High‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 2.4.2 Low‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected |
Comparison 3. Initiation of prophylactic therapy guided by colonoscopic surveillance versus initiation of prophylactic therapy immediately after surgery.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 3.1 Randomised trial (102 weeks of follow‐up) | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 3.1.1 Clinical recurrence (CDAI >150) until week 102 | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 3.1.2 Endoscopic recurrence (Rutgeerts' score ≥i2) at week 102 | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 3.1.3 Endoscopic recurrence (Rutgeerts' score ≥i3) at week 102 | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 3.1.4 Adverse events at 102 weeks | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 3.2 Cohort studies | 2 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 3.2.1 Clinical recurrence (32 months of median follow‐up) | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 3.2.2 Surgical recurrence (50 months of median follow‐up) | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 3.2.3 Azathioprine withdrawal due to adverse event (50 months of median follow‐up) | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected |
Comparison 4. Subgroup analysis according to baseline risk: Initiation of prophylactic therapy guided by colonoscopic surveillance versus initiation of prophylactic therapy immediately after surgery.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 4.1 Surgical recurrence (cohort study, 50 months of median follow‐up) | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 4.1.1 High‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected | |
| 4.1.2 Low‐risk participants | 1 | Risk Ratio (IV, Random, 95% CI) | Totals not selected |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Baudry 2014.
| Study characteristics | ||
| Methods | Observational study (retrospective cohort) conducted in France (1 centre). Median follow‐up: 51 months. |
|
| Participants | Adults undergoing bowel resection due to Crohn disease with an endoscopically accessible anastomosis at the Saint‐Louis Hospital (Paris) between 1995 and 2005. Adults with clinical recurrence within 12 months of surgery were excluded. Adults with low and high risk of postoperative recurrence (i.e. previous intestinal or extensive resection) were included. There are not more details about the definition of “high risk”. Control group 42 participants Intervention group 90 participants |
|
| Interventions | All participants received a tailored prophylactic therapy immediately after surgery: Low‐risk participants: no treatment 43.9% (58/132) or 5‐ASA 32.6% (43/132) High‐risk participants: azathioprine or 6‐mercaptopurine 22.7% (30/132) or methotrexate 0.8% (1/132). Control group No colonoscopy. Intensification of therapy only in case of clinical recurrence. Intervention group Colonoscopy at 6 to 12 months after surgery*. Intensification of prophylactic‐therapy in case of endoscopic recurrence (Rutgeerts’ score ≥ i2). Intensification of prophylactic‐therapy Rutgeerts’ score 2: addition or increase dose of 5‐ASA or thiopurines. Rutgeerts’ score 3‐4: addition or increase dose of thiopurines. *The participants were included in the “colonoscopy‐based management” group if they underwent a systematic colonoscopy with no clinical recurrence at the time of endoscopy. |
|
| Outcomes |
|
|
| Notes |
|
|
Bordeianou 2011.
| Study characteristics | ||
| Methods | Observational study (retrospective cohort) conducted in USA (2 centres) Median follow‐up: 32 months. |
|
| Participants | Adults undergoing ileocaecal resection due to Crohn disease without any other concomitant procedure at the Massachusetts General Hospital in Boston (N = 108) and the New York Presbyterian Hospital (N = 91) between September 1993 and April 2008. Control group 69 participants Intervention group 130 participants |
|
| Interventions |
Control group Initiation of prophylactic‐therapy during the first month after surgery: 5‐ASA 46.4% (32/69), antibiotics 53.6% (37/69), azathioprine 24.6% (17/69), 6‐mercaptopurine 86.9% (60/69), biologic therapy 10.1% (7/69) Intervention group No prophylactic‐therapy immediately after surgery Colonoscopy three to six months after surgery. Prophylactic‐therapy in case of endoscopic recurrence. Thirty‐two of 130 participants received prophylactic‐therapy after detection of endoscopic recurrence: 5‐ASA 53.1% (17/32), antibiotics 50% (16/32), azathioprine 21.9% (7/32), 6‐mercaptopurine 87.5% (28/32), biologic therapy 6.3% (2/32). |
|
| Outcomes |
|
|
| Notes |
|
|
Boucher 2016.
| Study characteristics | ||
| Methods | Observational study (retrospective cohort) conducted in France (1 centre) Follow‐up: 6 months to 20 years. |
|
| Participants | Adults undergoing bowel resection due to Crohn disease with a follow‐up of at least 6 months at the Hospital Estaing of Clermont‐Ferrand between 1986 and 2015. Control group 112 participants Intervention group 49 participants |
|
| Interventions | All participants received a similar prophylactic therapy immediately after surgery: 5‐ASA 33.5% (54/161), thiopurines 24.8% (40/161), methotrexate 4.3% (7/161), biologic therapy 25.5% (41/161). Control group No colonoscopy. Intensification of therapy according to clinical symptoms. Intervention group Systematic postoperative colonoscopy in participants with no clinical recurrence at the time of endoscopy (median time of colonoscopy after surgery: 9.5 months) Intensification of prophylactic‐therapy in case of endoscopic recurrence. Intensification of prophylactic‐therapy Addition or increase doses of thiopurines and/or biologic therapy. |
|
| Outcomes |
|
|
| Notes |
|
|
De Cruz 2013.
| Study characteristics | ||
| Methods | Observational study (retrospective cohort) conducted in Australia (2 centres) Median follow‐up: 53 months |
|
| Participants | Adults undergoing bowel resection due to Crohn disease (resection of all macroscopic disease) with an endoscopically accessible anastomosis at the St Vincent’s Public and Private Hospitals (Melbourne) between June 1998 and June 2008. Control group 66 participants Intervention group 70 participants |
|
| Interventions | All participants received a similar prophylactic therapy immediately after surgery: No therapy 19.1% (26/136), antibiotics 24.3% (33/136), 5‐ASA 37.5% (51/136), thiopurines 36% (49/136), methotrexate 5.9% (8/136). Control group No colonoscopy. Intensification of therapy according to clinical symptoms. Intervention group Colonoscopy within one year after surgery*. Intensification of prophylactic‐therapy in case of endoscopic recurrence. Endoscopic recurrence was defined as any lesion seen in the neoterminal ileum or at the anastomosis. Intensification of prophylactic‐therapy Addition or increase doses of antibiotics, 5‐ASA, thiopurines and/or methotrexate. *The participants were included in the “colonoscopy‐based management” group if they underwent a systematic colonoscopy with no clinical recurrence at the time of endoscopy. |
|
| Outcomes |
|
|
| Notes |
|
|
De Cruz 2015.
| Study characteristics | ||
| Methods | Multicentre randomised‐controlled trial conducted in Australia and New Zealand (17 centres) between Oct 13, 2009, and Sept 28, 2011. Trial duration: 18 months |
|
| Participants | Adults undergoing bowel resection due to Crohn disease (resection of all macroscopic disease) with an endoscopically accessible anastomosis. Participants with high and low risk of postoperative recurrence were included. High risk was defined as having one or more of the following factors:
One hundred eighty‐four participants were initially randomised, but 10 participants were excluded from the analysis after randomisation, because they did not receive the allocated management (four in the control group, six in the intervention group). Control group 52 participants (44 high risk, 8 low risk) Intervention group 122 participants (101 high risk, 21 low risk) |
|
| Interventions | All participants received a tailored preventive therapy immediately after surgery: Low‐risk participants: no treatment High‐risk participants: azathioprine (2.0 mg/kg per day) or 6‐mercaptopurine (1.5 mg/kg per day) Control group No colonoscopy. Intensification of therapy only in case of clinical recurrence. Intervention group Colonoscopy at 6 months after surgery. Intensification of prophylactic‐therapy in case of endoscopic recurrence. Intensification therapy Low‐risk participants: azathioprine (2.0 mg/kg per day) or 6‐mercaptopurine (1.5 mg/kg per day) High‐risk participants: adalimumab (160 mg, 80 mg two weeks later, then 40 mg every two weeks) |
|
| Outcomes |
Primary outcome Endoscopic recurrence (Rutgeerts' score ≥ i2) Secondary outcomes Clinical recurrence (CDAI> 150 or 200 points) Surgical recurrence |
|
| Notes |
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation, in blocks of 3 in a 2:1 ratio. |
| Allocation concealment (selection bias) | Low risk | Computer‐generated with block randomisation undertaken for each centre. The allocation list was maintained centrally |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Open‐label study. Participants, personnel and endoscopists were unblinded. |
| Blinding of outcome assessment (detection bias) Clinical recurrence | High risk | Open‐label study. CDAI score has subjective items that are assessed by participants. |
| Blinding of outcome assessment (detection bias) Endoscopic recurrence | Low risk | The assessment was carried out using photographs. Two masked investigators scored the pictures. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | All withdrawals were reported with reasons, were balanced across groups and the analysis was conducted according to ITT principle. Despite this, 32.1% of participants dropped out the study. All withdrawals were included in the final analysis as participants with endoscopic and clinical recurrence.It is not clear the real impact in the final estimate of the effect (this could be inaccurate) Ten participants were excluded from the analysis after randomisation, because they did not receive the allocated management. |
| Selective reporting (reporting bias) | Low risk | Protocol outcomes were the same as final report. ClinicalTrials.gov number NCT00989560 |
| Other bias | Low risk | Comment: No other major risk of bias was noticed |
Ferrante 2015.
| Study characteristics | ||
| Methods | Multicentre randomised‐controlled trial conducted in Belgium, Czech Republic and Greece (4 centres) between 2005 and 2012. Trial duration: 102 weeks |
|
| Participants | Adults (16 to 75 years) undergoing bowel resection due to Crohn disease (resection of all macroscopic disease) with an endoscopically accessible anastomosis. Participants with high risk of postoperative recurrence were included. High risk was defined as having one of the following risk factors:
Control group 32 participants Intervention group 31 participants |
|
| Interventions |
Control group Initiation of prophylactic‐therapy within 14 days from surgery: Azathioprine (2.0 to 2.5 mg/kg per day). Intervention group No prophylactic‐therapy immediately after surgery Colonoscopy at 26 and 102 weeks after surgery. In case of endoscopic recurrence (Rutgeerts’ score ≥ i2), Azathioprine was introduced (2.0 to 2.5 mg/kg per day) |
|
| Outcomes |
Primary outcome Endoscopic recurrence (Rutgeerts score ≥ i2) Secondary outcomes Clinical recurrence (CDAI > 150 points) Surgical recurrence |
|
| Notes |
|
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Computer‐generated randomisation in a 1:1 ratio. |
| Allocation concealment (selection bias) | Low risk | Participants were randomised 1:1 and balanced for age using a central computer‐based randomisation system. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Open‐label study.Participants and personnel were unblinded. |
| Blinding of outcome assessment (detection bias) Clinical recurrence | High risk | Open‐label study. CDAI score has subjective items that are assessed by participants. |
| Blinding of outcome assessment (detection bias) Endoscopic recurrence | Low risk | Endoscopists were blinded to treatment allocation and time interval from surgery. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Withdrawals were balanced across the groups, the reasons were detailed, were not related to the outcomes and the analysis was according to the intention to treat principle. Despite this, the proportion of participants that dropped out was high (33.3%) and the effect estimate could be inaccurate. |
| Selective reporting (reporting bias) | Unclear risk | Primary and secondary outcomes were the same as in protocol published in ClinicalTrials.gov, number NCT02247258. Despite this, the protocol was submitted in Sep 2014, but the recruitment started in 2005 and was prematurely stopped due to slow recruitment. |
| Other bias | Low risk | Comment: No other major risk of bias was noticed |
Guo 2018.
| Study characteristics | ||
| Methods | Observational study (retrospective cohort) conducted in China (1 centre) Mean follow‐up: 50.6 ± 23.8 months |
|
| Participants | Adults undergoing intestinal resection with ileocolic anastomosis and who also received azathioprine as maintenance therapy from 2006 to 2015. Exclusion criteria: previous intestinal resections, other segmental resections, stricturoplasty or repair at the time of the index resection, more than one bowel anastomosis during a single operation and incomplete medical information. Participants with at least one of the following risk factors were considered to be at high risk of recurrence:
Control group (Symptom‐driven) 33 participants (27 high risk, 6 low risk) Intervention group 1 (Endoscopy‐driven) 42 participants (39 high risk, 3 low risk) Intervention group 2 (Immediate) 91 participants (69 high risk, 22 low risk) |
|
| Interventions | This study had three groups. Control group (Symptom‐driven) Symptom‐driven therapy was defined as azathioprine initiated only in the presence of clinical recurrence. Intervention group 1 (Endoscopy‐driven) Endoscopy‐driven therapy was defined as azathioprine initiated only when asymptomatic endoscopic recurrence occurred. The endoscopic follow‐up occurred from 3 to 18 months after surgery. Intervention group 2 (Immediate) Immediate prophylactic therapy was defined as azathioprine initiated within 2 to 4 weeks after resection. The dosage of azathioprine was 1.5to 2.5 mg/kg/day. |
|
| Outcomes | Surgical recurrence (intestinal resection for complications of Crohn disease or symptoms refractory to medical treatments after the index surgery) | |
| Notes |
|
|
anti‐TNFa: anti‐tumour necrosis factor antibodies; CD: Crohn's disease; CDAI: Crohn's Disease Activity Index; ITT: intention‐to‐treat.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Barreiro‐De Acosta 2019 | This was a nationwide retrospective cohort study that evaluated 314 participants who underwent ileocaecal resection for CD. The main aim was to assess endoscopic monitoring and treatment decisions in CD patients in a real‐world setting. This study was excluded because the authors did not include outcomes of interest for this review. |
| Gonzales‐Lama 2015 | This was a retrospective cohort study that evaluated 166 participants who underwent ileocaecal resection for CD. The main aim was to assess the role of colonoscopy‐based management of postoperative CD. This study was excluded because of all participants included in the final analysis were studied with colonoscopy and the definition of “colonoscopy‐based management” and “non‐colonoscopy‐based management” was only established among participants with endoscopic recurrence. |
| Papamichael 2012 | This was a prospective cohort study that included 23 participants who underwent ileocaecal resection for CD. The main aim was to assess the role of adalimumab given immediately after surgery or at six months in case of endoscopic recurrence. This study was excluded because of participants of both groups were studied with colonoscopy at six months after surgery and in both groups the postoperative management was modified according to endoscopic findings. |
| Sorrentino 2012 | This was a prospective cohort study that included 43 participants who underwent bowel resection for CD. The main aim was to investigate the impact of infliximab on early endoscopic lesions after resection for CD. This study was excluded because of all participants were studied with colonoscopy at six months after surgery. In all participants the prophylactic therapy was modified according to the endoscopic findings and only those with endoscopic recurrence were included in the final analysis. |
| Yamamoto 2009 | This was a prospective cohort study that included 26 participants who underwent ileocaecal resection for CD. The main aim was to investigate the impact of infliximab on early endoscopic lesions after resection for CD. This study was excluded because of all participants were studied with colonoscopy at six months after surgery, only those participants with clinical remission and endoscopic recurrence were included in the analysis and the therapy was not modified according to the endoscopic findings. |
CD: Chron's disease
Differences between protocol and review
The main difference between the protocol and the review was the identification of two comparisons that were not contemplated (management guided by colonoscopy compared to management guided by symptoms and full‐treatment immediately after surgery). Although this situation may induce bias to review, the authors consider that the separation is clinically appropriate. Also, the search on Epistemonikos was not planned from the start. We added this search to be more exhaustive.
Contributions of authors
Draft the protocol: Candia R
Develop and run the search strategy: Preliminary search strategy was performed by Candia R with assistance of Elizabeth Uleryk. The final search strategy was performed by the Cochrane IBD Group Information Specialist.
Obtain copies of studies: Candia R
Select which studies to include: Bravo‐Soto G, Monrroy H, Candia R, Hernandez‐Rocha C. Disagreements were resolved by Candia R.
Extract data from studies: Candia R, Bravo G, Monrroy H, Hernandez‐Rocha C. Disagreements were resolved by Candia R.
Enter data into RevMan: Candia R, Bravo‐Soto G.
Carry out the analysis: Candia R, Bravo‐Soto G.
Interpret the analysis: Candia R, Bravo‐Soto G.
Draft the final review: Candia R, Bravo‐Soto G, Nguyen GC
Check correct use of grammar: Nguyen GC
Update the review: Candia R
Sources of support
Internal sources
-
CONICYT, Becas Chile para Magister en el extranjero, Chile
Sponsorship support
Pontificia Universidad Católica de Chile, Chile
Univerisity of Toronto, Canada
External sources
No sources of support supplied
Declarations of interest
The authors of this review have no known conflicts to declare.
New
References
References to studies included in this review
Baudry 2014 {published data only}
- Baudry C, Pariente B, Lourenço N, Simon M, Chirica M, Cattan P, et al. Tailored treatment according to early post-surgery colonoscopy reduces clinical recurrence in Crohn's disease: a retrospective study. Digestive and Liver Disease 2014;46(10):887-92. [DOI] [PubMed] [Google Scholar]
Bordeianou 2011 {published data only}
- Bordeianou L, Stein SL, Ho VP, Dursun A, Sands BE, Korzenik JR, et al. Immediate versus tailored prophylaxis to prevent symptomatic recurrences after surgery for ileocecal Crohn's disease? Surgery 2011;149(1):72-8. [DOI] [PubMed] [Google Scholar]
Boucher 2016 {published data only}
- Boucher AL, Pereira B, Decousus S, Goutte M, Goutorbe F, Dubois A, et al. Endoscopy-based management decreases the risk of postoperative recurrences in Crohn's disease. World Journal of Gastroenterology 2016;22(21):5068-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
De Cruz 2013 {published data only}
- De Cruz P, Bernardi MP, Kamm MA, Allen PB, Prideaux L, Williams J, et al. Postoperative recurrence of Crohn's disease: impact of endoscopic monitoring and treatment step-up. Colorectal Disease 2013;15(2):187-97. [DOI] [PubMed] [Google Scholar]
De Cruz 2015 {published data only}
- De Cruz P, Kamm MA, Hamilton AL, Ritchie KJ, Krejany EO, Gorelik A, et al. Crohn's disease management after intestinal resection: a randomised trial. Lancet 2015;385(9976):1406-17. [DOI] [PubMed] [Google Scholar]
Ferrante 2015 {published data only}
- Ferrante M, Papamichael K, Duricova D, D'Haens G, Vermeire S, Archavlis E, et al. Systematic versus endoscopy-driven treatment with azathioprine to prevent postoperative ileal Crohn's disease recurrence. Journal of Crohn's & Colitis 2015;9(8):617-24. [DOI] [PubMed] [Google Scholar]
Guo 2018 {published data only}
- Guo Z, Cai X, Liu R, Gong J, Li Y, Cao L, et al. Immediate prophylactic vs endoscopic or symptomatic-driven azathioprine treatment to prevent surgical recurrence after intestinal resection for Crohn's disease. Colorectal Disease 2018;20(9):O267-O276. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Barreiro‐De Acosta 2019 {published data only}
- Barreiro-De Acosta M, Domènech E, Martín Arranz MD, García Sánchez V, Gutiérrez Casbas A, Chaparro M, et al. Endoscopic follow-up and therapeutic attitude after ileocolonic resection in a nationwide Spanish cohort of Crohn's disease patients: the Practicrohn study. Expert Review of Gastroenterology & Hepatology 2019 Aug;13(8):807-13. [DOI: 10.1080/17474124.2019.1626717] [PMID: ] [DOI] [PubMed] [Google Scholar]
Gonzales‐Lama 2015 {published data only}
- González-Lama Y, Blázquez I, Suárez CJ, Oliva B, Matallana V, Calvo M, et al. Impact of endoscopic monitoring in postoperative Crohn's disease patients already receiving pharmacological prevention of recurrence. Revista Espanola de Enfermedades Digestivas 2015;107(10):586-90. [DOI] [PubMed] [Google Scholar]
Papamichael 2012 {published data only}
- Papamichael K, Archavlis E, Lariou C, Mantzaris GJ. Adalimumab for the prevention and/or treatment of post-operative recurrence of Crohn's disease: A prospective, two-year, single center, pilot study. Journal of Crohn's & Colitis 2012;6(9):924-31. [DOI] [PubMed] [Google Scholar]
Sorrentino 2012 {published data only}
- Sorrentino D, Terrosu G, Paviotti A, Geraci M, Avellini C, Zoli G, et al. Early diagnosis and treatment of postoperative endoscopic recurrence of Crohn's disease: partial benefit by infliximab--a pilot study. Digestive Diseases and Sciences 2012;57(5):1341-8. [DOI] [PubMed] [Google Scholar]
Yamamoto 2009 {published data only}
- Yamamoto T, Umegae S, Matsumoto K. Impact of infliximab therapy after early endoscopic recurrence following ileocolonic resection of Crohn's disease: a prospective pilot study. Inflammatory Bowel Diseases 2009;15(10):1460-6. [DOI] [PubMed] [Google Scholar]
Additional references
Atkins 2004
- Atkins D, Best D, Briss PA, Eccles M, Falck-Ytter Y, Flottorp S, et al, GRADE Working Group. Grading quality of evidence and strength of recommendations. BMJ 2004;328(7454):1490-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Baert 2010
- Baert F, Moortgat L, Van Assche G, Caenepeel P, Vergauwe P, De Vos M, et al. Mucosal healing predicts sustained clinical remission in patients with early-stage Crohn's disease. Gastroenterology 2010;138(2):463-8. [DOI] [PubMed] [Google Scholar]
Baumgart 2012
- Baumgart DC, Sandborn WJ. Crohn's disease. Lancet 2012;380(9853):1590-605. [DOI] [PubMed] [Google Scholar]
Bernell 2000
- Bernell O, Lapidus A, Hellers G. Risk factors for surgery and recurrence in 907 patients with primary ileocaecal Crohn's disease. British Journal of Surgery 2000;87(12):1697-701. [DOI] [PubMed] [Google Scholar]
Blum 2009
- Blum E, Katz JA. Postoperative therapy for Crohn's disease. Inflammatory Bowel Diseases 2009;15(3):463-72. [DOI] [PubMed] [Google Scholar]
Cochrane Norway 2019
- Cochrane Norway. How to write a plain language summary of a Cochrane intervention review. https://www.cochrane.no/sites/cochrane.no/files/public/uploads/how_to_write_a_cochrane_pls_12th_february_2019.pdf 2019.
Colombel 2014
- Colombel JF, Rutgeerts PJ, Sandborn WJ, Yang M, Camez A, Pollack PF, et al. Adalimumab induces deep remission in patients with Crohn's disease. Clinical Gastroenterology and Hepatology 2014;12(3):414-22. [DOI] [PubMed] [Google Scholar]
Cosnes 2002
- Cosnes J, Cattan S, Blain A, Beaugerie L, Carbonnel F, Parc R, et al. Long-term evolution of disease behavior of Crohn's disease. Inflammatory Bowel Diseases 2002;8(4):244-50. [DOI] [PubMed] [Google Scholar]
D'Haens 2008
- D'Haens G, Baert F, Van Assche G, Caenepeel P, Vergauwe P, Tuynman H, et al. Early combined immunosuppression or conventional management in patients with newly diagnosed Crohn's disease: an open randomised trial. Lancet 2008;371(9613):660-7. [DOI] [PubMed] [Google Scholar]
Daperno 2004
- Daperno M, D'Haens G, Van Assche G, Baert F, Bulois P, Maunoury V, et al. Development and validation of a new, simplified endoscopic activity score for Crohn's disease: the SES-CD. Gastrointestinal Endoscopy 2004;60(4):505-12. [DOI] [PubMed] [Google Scholar]
Ford 2009
- Ford AC, Peyrin-Biroulet L. Opportunistic infections with anti-tumor necrosis factor-α therapy in inflammatory bowel disease: meta-analysis of randomized controlled trials. American Journal of Gastroenterology 2009;108(8):1268-76. [DOI] [PubMed] [Google Scholar]
Frøslie 2007
- Frøslie KF, Jahnsen J, Mourn BA, Vatn MH, IBSEN Group. Mucosal healing in inflammatory bowel disease: results from a Norwegian population-based cohort. Gastroenterology 2007;133(2):412-22. [DOI] [PubMed] [Google Scholar]
Gjuladin‐Hellon 2019
- Gjuladin‐Hellon T, Iheozor‐Ejiofor Z, Gordon M, Akobeng AK. Azathioprine and 6-mercaptopurine for maintenance of surgically-induced remission in Crohn's disease. Cochrane Database of Systematic Reviews 2019, Issue 8. Art. No: CD010233. [DOI: 10.1002/14651858.CD010233.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Guyatt 2008
- Guyatt GH, Oxman AD, Vist GE, Kunz R, Falck-Ytter Y, Alonso-Coello P, et al. GRADE: an emerging consensus on rating high quality evidence and strength of recommendations. BMJ 2008;336(7650):924-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Guyatt 2011
- Guyatt GH, Oxman AD, Kunz R, Atkins D, Brozek J, Vist G, et al. GRADE guidelines: 2. Framing the question and deciding on important outcomes. Journal of Clinical Epidemiology 2011;64(4):395-400. [DOI: 10.1016/j.jclinepi.2010.09.012] [PMID: PMID: 21194891] [DOI] [PubMed] [Google Scholar]
Higgins 2003
- Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ 2003;327(7414):557-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2011a
- Higgins JP, Altman DG, Sterne JA (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Green S, (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.cochrane-handbook.org.
Higgins 2011b
- Higgins JP, Deeks JJ, Altman DG (editors). Chapter 16: Special topics in statistics. In: Higgins JP, Green S, (editors). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.cochrane-handbook.org.
Jones 2014
- Jones GR, Kennedy NA, Lees CW, Arnott ID, Satsangi J. Systematic review: the use of thiopurines or anti-TNF in post-operative Crohn's disease maintenance--progress and prospects. Alimentary Pharmacology and Therapeutics 2014;39(11):1253-65. [DOI] [PubMed] [Google Scholar]
Kandiel 2005
- Kandiel A, Fraser AG, Korelitz BI, Brensinger C, Lewis JD. Increased risk of lymphoma among inflammatory bowel disease patients treated with azathioprine and 6-mercaptopurine. Gut 2005;54(8):1121-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lamb 2019
- Lamb CA, Kennedy NA, Raine T, Hendy PA, Smith PJ, Limdi JK, et al. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults.. Gut 2019;68(Suppl 3):s1-s106. [DOI] [PMC free article] [PubMed] [Google Scholar]
Margagnoni 2011
- Margagnoni G, Aratari A, Mangone M, Moretti A, Spagnolo A, Fascì Spurio F, et al. Natural history of ileo-caecal Crohn's disease after surgical resection. A long term study. Minerva Gastroenterologica e Dietologica 2011;57(4):335-44. [PubMed] [Google Scholar]
Molodecky 2012
- Molodecky NA, Soon IS, Rabi DM, Ghali WA, Ferris M, Chernoff G, et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 2012;142(1):46-54. [DOI: 10.1053/j.gastro.2011.10.001] [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Mowat 2011
- Mowat C, Cole A, Windsor A, Ahmad T, Arnott I, Driscoll R, et al. Guidelines for the management of inflammatory bowel disease in adults. Gut 2011;60(5):571-607. [DOI] [PubMed] [Google Scholar]
Ng 2018
- Ng SC, Shi HY, Hamidi N, Underwood FE, Tang W, Benchimol EI, et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet 2018 Dec 23;390(10114):2769-78. [DOI: 10.1016/S0140-6736(17)32448-0] [PMID: ] [DOI] [PubMed] [Google Scholar]
Pascua 2008
- Pascua M, Su C, Lewis JD, Brensinger C, Lichtenstein GR. Meta-analysis: factors predicting post-operative recurrence with placebo therapy in patients with Crohn's disease. Alimentary Pharmacology & Therapeutics 2008;28(5):545-56. [DOI] [PubMed] [Google Scholar]
Peyrin‐Biroulet 2010
- Peyrin-Biroulet L, Loftus EV Jr, Colombel JF, Sandborn WJ. The natural history of adult Crohn's disease in population-based cohorts. American Journal of Gastroenterology 2010;105(2):289-97. [DOI] [PubMed] [Google Scholar]
Peyrin‐Biroulet 2015
- Peyrin-Biroulet L, Sandborn W, Sands BE et al. Selecting Therapeutic Targets in Inflammatory Bowel Disease (STRIDE): Determining Therapeutic Goals for Treat-to-Target. Am J Gastroenterol 2015;110(9):1324-38. [DOI] [PubMed] [Google Scholar]
Regueiro 2009
- Regueiro M, Schraut W, Baidoo L, Kip KE, Sepulveda AR, Pesci M, et al. Infliximab prevents Crohn's disease recurrence after ileal resection. Gastroenterology 2009;136(2):441-50. [DOI] [PubMed] [Google Scholar]
Rutgeerts 1984
- Rutgeerts P, Geboes K, Vantrappen G, Kerremans R, Coenegrachts JL, Coremans G. Natural history of recurrent Crohn's disease at the ileocolonic anastomosis after curative surgery. Gut 1984;25(6):665-72. [DOI] [PMC free article] [PubMed] [Google Scholar]
Rutgeerts 1990
- Rutgeerts P, Geboes K, Vantrappen G, Beyls J, Kerremans R, Hiele M. Predictability of the postoperative course of Crohn's disease. Gastroenterology 1990;99(4):956-63. [DOI] [PubMed] [Google Scholar]
Savarino 2013
- Savarino E, Bodini G, Dulbecco P, Assandri L, Bruzzone L, Mazza F, et al. Adalimumab is more effective than azathioprine and mesalamine at preventing postoperative recurrence of Crohn's disease: a randomized controlled trial. American Journal of Gastroenterology 2013;108(11):1731-42. [DOI] [PubMed] [Google Scholar]
Schintzler 2009
- Schnitzler F, Fidder H, Ferrante M, Noman M, Arijs I, Van Assche G, et al. Mucosal healing predicts long-term outcome of maintenance therapy with infliximab in Crohn's disease. Inflammatory Bowel Diseases 2009;15(9):1295–301. [DOI] [PubMed] [Google Scholar]
Schoepfer 2010
- Schoepfer AM, Beglinger C, Straumann A, Trummler M, Vavricka SR, Bruegger LE, et al. Fecal calprotectin correlates more closely with the Simple Endoscopic Score for Crohn's disease (SES-CD) than CRP, blood leukocytes, and the CDAI. American Journal of Gastroenterology 2010;105(1):162-9. [DOI] [PubMed] [Google Scholar]
Schünemann 2011
- Schünemann HJ, Oxman AD, Vist GE, Higgins JP, Deeks JJ, Glasziou P, et al. Chapter 12: Interpreting results and drawing conclusions. In: Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.cochrane-handbook.org.
Selvaratnam 2019
- Selvaratnam S, Gullino S, Shim L, Lee E, Lee A, Paramsothy S, et al. Epidemiology of inflammatory bowel disease in South America: a systematic review. World Journal of Gastroenterology 2019 Dec 21;25(47):6866-75. [CENTRAL: PMC6931006] [DOI: 10.3748/wjg.v25.i47.6866] [MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ungaro 2020
- Ungaro RC, Yzet C, Bossuyt P, Baert FJ, Vanasek T, D'Haens GR, et al. Deep remission at 1 year prevents progression of early Crohn's disease. Gastroenterology 2020;pii: S0016-5085(20)30390-5. doi: 10.1053/j.gastro.2020.03.039:[Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
Watanabe 2014
- Watanabe K, Sasaki I, Fukushima K, Futami K, Ikeuchi H, Sugita A, et al. Long-term incidence and characteristics of intestinal failure in Crohn's disease: a multicenter study. Journal of Gastroenterology 2014;49(2):231-8. [DOI] [PubMed] [Google Scholar]
Wells 2016
- Wells GA, Shea B, O'Connell D, Peterson J, Welch V, Losos M, et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of non randomised studies in meta-analyses. www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed 4 May 2016).
Williams 2014
- Williams CJ, Peyrin-Biroulet L, Ford AC. Systematic review with meta-analysis: malignancies with anti-tumour necrosis factor-α therapy in inflammatory bowel disease. Alimentary Pharmacology and Therapeutics 2014;39(5):447-58. [DOI] [PubMed] [Google Scholar]
Yang 2014
- Yang Z, Ye X, Wu Q, Wu K, Fan D. A network meta-analysis on the efficacy of 5-aminosalicylates, immunomodulators and biologics for the prevention of postoperative recurrence in Crohn's disease. International Journal of Surgery 2014;12(5):516-22. [DOI] [PubMed] [Google Scholar]
Yoshida 2012
- Yoshida K, Fukunaga K, Ikeuchi H, Kamikozuru K, Hida N, Ohda Y, et al. Scheduled infliximab monotherapy to prevent recurrence of Crohn's disease following ileocolic or ileal resection: a 3-year prospective randomized open trial. Inflammatory Bowel Diseases 2012;18(9):1617-23. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Candia 2016
- Candia R, Ibañez P, Nguyen GC. Colonoscopy-guided therapy for the prevention of post-operative recurrence of Crohn’s disease. Cochrane Database of Systematic Reviews 2016, Issue 8. Art. No: CD012328. [DOI: 10.1002/14651858.CD012328] [DOI] [PMC free article] [PubMed] [Google Scholar]


