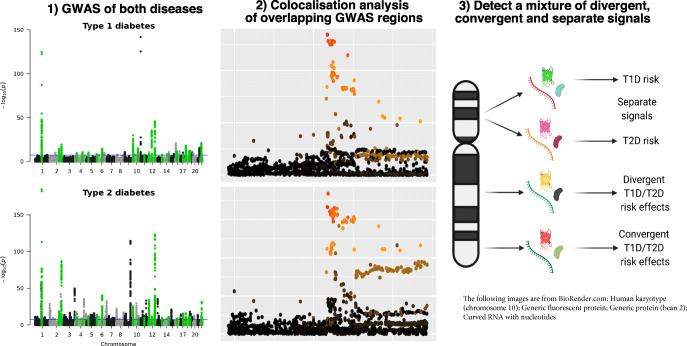
Abstract
Aims/hypothesis
Given the potential shared aetiology between type 1 and type 2 diabetes, we aimed to identify any genetic regions associated with both diseases. For associations where there is a shared signal and the allele that increases risk to one disease also increases risk to the other, inference about shared aetiology could be made, with the potential to develop therapeutic strategies to treat or prevent both diseases simultaneously. Alternatively, if a genetic signal co-localises with divergent effect directions, it could provide valuable biological insight into how the association affects the two diseases differently.
Methods
Using publicly available type 2 diabetes summary statistics from a genome-wide association study (GWAS) meta-analysis of European ancestry individuals (74,124 cases and 824,006 controls) and type 1 diabetes GWAS summary statistics from a meta-analysis of studies on individuals from the UK and Sardinia (7467 cases and 10,218 controls), we identified all regions of 0.5 Mb that contained variants associated with both diseases (false discovery rate <0.01). In each region, we performed forward stepwise logistic regression to identify independent association signals, then examined co-localisation of each type 1 diabetes signal with each type 2 diabetes signal using coloc. Any association with a co-localisation posterior probability of ≥0.9 was considered a genuine shared association with both diseases.
Results
Of the 81 association signals from 42 genetic regions that showed association with both type 1 and type 2 diabetes, four association signals co-localised between both diseases (posterior probability ≥0.9): (1) chromosome 16q23.1, near CTRB1/BCAR1, which has been previously identified; (2) chromosome 11p15.5, near the INS gene; (3) chromosome 4p16.3, near TMEM129 and (4) chromosome 1p31.3, near PGM1. In each of these regions, the effect of genetic variants on type 1 diabetes was in the opposite direction to the effect on type 2 diabetes. Use of additional datasets also supported the previously identified co-localisation on chromosome 9p24.2, near the GLIS3 gene, in this case with a concordant direction of effect.
Conclusions/interpretation
Four of five association signals that co-localise between type 1 diabetes and type 2 diabetes are in opposite directions, suggesting a complex genetic relationship between the two diseases.
Graphical abstract
Supplementary Information
The online version contains peer-reviewed but unedited supplementary material available at 10.1007/s00125-021-05428-0.
Keywords: Analyses, Co-localisation, Genetics, Genome-wide association study, Insulin, Statistics, Systematic, Type 1 diabetes, Type 2 diabetes
Introduction
There is a genetic component to both type 1 and type 2 diabetes, with approximately 60 chromosome regions associated with type 1 diabetes [1] and over 200 associated with type 2 diabetes [2] at genome-wide significance. Examination of regions associated with both diseases could uncover signals that simultaneously alter disease risk for both diseases, termed co-localisation. Uncovering co-localising signals could provide biological insights into shared disease mechanisms, and potentially reveal therapeutic targets effective for both diseases. A recent analysis suggested that the same genetic variant alters risk of both type 1 and type 2 diabetes in five regions, near CENPW, CTRB1/BCAR1, GLIS3, BCL11A and THADA [3].
Here, we identified all regions across the genome that showed evidence of association with both type 1 and type 2 diseases at a false discovery rate (FDR) <0.01 and assessed co-localisation between the two diseases in each of these regions. Furthermore, to account for the possibility of multiple causal variants within an associated region, we extended the analysis to investigate conditionally independent associations within each region, to assess whether any of the associations with one disease co-localised with any associations in the other.
Methods
Type 1 diabetes meta-analysis summary statistics were generated using genome-wide association study (GWAS) data from 3983 cases and 3994 controls from the UK (genotyped using the Illumina Infinium 550K platform), 1926 cases and 3342 controls from the UK (genotyped using the Affymetrix GeneChip 500K platform) and 1558 cases and 2882 controls from Sardinia (genotyped using the Affymetrix 6.0 and Illumina Omni Express platforms), totalling 7467 cases and 10,218 controls (Electronic supplementary material [ESM] Table 1). Genotypes were imputed using the Haplotype Reference Consortium reference panel for the UK collections [4], and a custom Sardinian reference panel of 3514 Sardinians for the Sardinian collection (ESM, Imputation).
Summary statistics for type 2 diabetes were from 74,124 cases and 824,006 controls of European ancestry, imputed using the Haplotype Reference Consortium reference panel [2].
Regions associated with both diseases were identified by selecting all variants with type 1 diabetes and a type 2 diabetes association with an FDR <0.01 (ESM Methods, Type 1 diabetes GWAS). In each such region, windows of approximately 0.5 Mb were taken to examine co-localisation (ESM Methods, Regions associated with both diseases). Within these regions, forward stepwise logistic regressions were carried out for both diseases, and conditional summary statistics were obtained so each conditionally independent signal from both diseases could be tested against each other for co-localisation (ESM Methods, Conditional analyses).
Co-localisation of signals was assessed using coloc [5], a Bayesian method that enumerates the posterior probability that the association signals in a region are shared between traits. The prior probability of association with either disease was taken to be 1×10-4 and the prior probability that the association signal is shared across traits was taken to be 5×10-6, as recommended [6]. The threshold to consider signals as co-localising was conservatively chosen at a posterior probability ≥0.9. Co-localisation was also examined using an alternative approach, as a secondary analysis, eCAVIAR [7] (ESM Methods, eCAVIAR).
Code used to carry out this analysis is available at https://github.com/jinshaw16/t1d-t2d-colocalisation.
Results
Including conditionally independent association signals, 81 co-localisation analyses were carried out across 42 chromosomal regions that showed association with both diseases (ESM Table 2).
Four signals showed evidence of co-localisation using coloc, and these were also the regions with the highest eCAVIAR regional co-localisation posterior probabilities (ESM Table 3). The first was on chromosome 16q23.1, near CTRB1 and BCAR1, with a posterior probability of co-localisation (H4PP hereafter) of 0.98 (ESM Fig. 1). The minor A allele at the type 2 diabetes index variant, rs72802342 (C>A), is protective for type 2 diabetes (OR 0.87, p=4.00×10-32) and susceptible for type 1 diabetes (OR 1.33, p=5.81×10-10).
The second was on chromosome 11p15.5, near INS, where the primary type 2 diabetes association co-localised with the secondary type 1 diabetes association (H4PP=0.95, ESM Fig. 2). The direction of effect was opposite, with the minor A allele at the type 2 diabetes index variant, rs4929965 (G>A), associated with susceptibility to type 2 diabetes (OR 1.07, p=4.80×10-25) and protection from type 1 diabetes (OR 0.87, p=1.89×10-5).
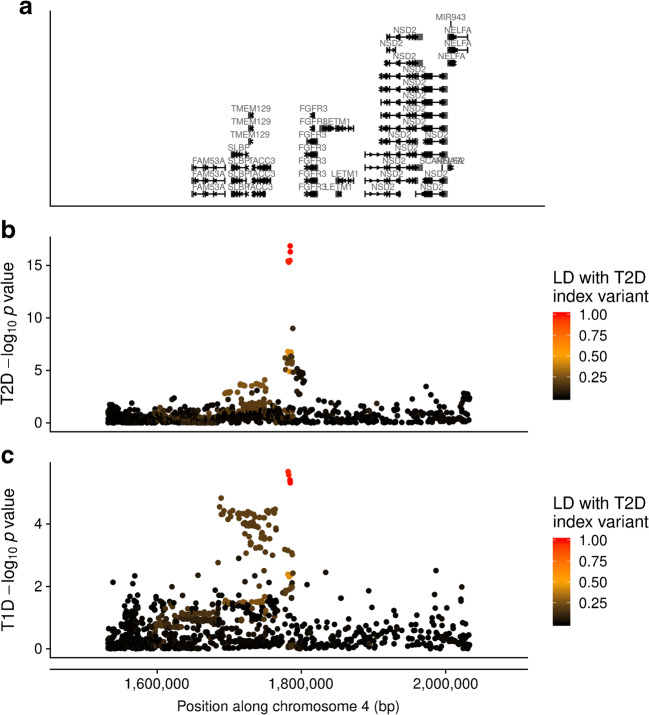
Third, a region on chromosome 4p16.3 co-localised (H4PP=0.97) (Fig. 1), near TMEM129. The minor T allele at the type 2 diabetes index variant, rs56337234 (C>T), was associated with decreased risk of type 2 diabetes (OR 0.94, p=1.4×10-17) and increased risk of type 1 diabetes (OR 1.12, p=4.07×10-6).
Fig. 1.
Manhattan plots showing (a) gene locations and –log10 p value of association for each variant by position along chromosome 4 (genome build 37) in the TMEM129 region for (b) type 2 diabetes (T2D) and (c) type 1 diabetes (T1D), coloured by r2 to the type 2 diabetes index variant, rs56337234
Finally, a region on chromosome 1p31.3, near PGM1, co-localised (H4PP=0.91, ESM Fig. 3), with the minor T allele at the type 2 diabetes index variant rs2269247 (C>T) decreasing risk of type 2 diabetes (OR 0.96, p=4.6×10-7) and increasing risk of type 1 diabetes (OR 1.15, p=1.9×10-6) (Table 1).
Table 1.
Regions with a co-localisation posterior probability of ≥0.9 between type 1 diabetes and type 2 diabetes
| rsID | Proximal gene(s) | chr | pos (gr37) | REF | ALT | T2D conditional on: | T2D OR (95% CI) | T2D p | r2 to T1D index variant (T1D index variant) | T1D conditional on: | T1D OR (95% CI) | T1D p |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| rs2269247 | PGM1 | 1p31.3 | 64107284 | C | T | – | 0.96 (0.94, 0.97) | 4.60×10-7 | 0.86 (rs2269246) | – | 1.14 (1.08, 1.22) | 1.94×10-6 |
| rs56337234 | TMEM129 | 4p16.3 | 1784403 | C | T | – | 0.94 (0.93, 0.96) | 1.40×10-17 | 0.97 (rs6829631) | – | 1.12 (1.07, 1.18) | 4.07×10-6 |
| rs4929965 | INS | 11p15.5 | 2197286 | G | A |
rs11042596, rs555759341, rs571342427, rs10838787 |
1.07 (1.06, 1.09) | 4.80×10-25 | 0.97 (rs7119275) | rs689 | 0.87 (0.81, 0.93) | 1.89×10-5 |
| rs72802342 |
CTRB1/ BCAR1 |
16q23.1 | 75234872 | C | A | rs3115960 | 0.87 (0.85, 0.89) | 4.00×10-32 | 0.89 (rs55993634) | – | 1.33 (1.22, 1.46) | 5.81×10-10 |
Summary statistics given from the perspective of the index type 2 diabetes variant and with respect to the ALT allele
r2 obtained from 1000 Genomes Project European population
ALT, alternative allele; REF, reference allele; T2D, type 2 diabetes; T1D, type 1 diabetes
The OR is for the addition of an ALT allele
We did not replicate the finding that the chromosome regions near CENPW, GLIS3, BCL11A or THADA co-localised between type 1 and type 2 diabetes (H4PP CENPW=0.12, GLIS3=0.29, BCL11A=0.28, THADA not examined as no type 1 diabetes association existed in the region [FDR=0.07]). To investigate these discrepancies, we examined two other large type 2 diabetes meta-analyses: a trans-ethnic study including 1,407,282 individuals [8] and a study of 433,540 individuals of East Asian ancestry [9]. For the CENPW and BCL11A regions, the type 2 diabetes signal is consistent with at least one of the other GWAS studies (measured by linkage disequilibrium [LD] in Europeans to the other study index variants, ESM Table 4), and the type 1 diabetes index variant is not in strong LD (r2<0.41) with any of the index variants for type 2 diabetes across the three GWAS studies. However, at GLIS3, there appears to be a distinct signal in the European study [2] compared with the trans-ethnic and East Asian type 2 diabetes studies (r2=0.65), and the index variants from these two studies are in higher r2 with the type 1 diabetes signal in our analysis (r2=0.68), and even higher r2 with the index variant from a larger type 1 diabetes genetic analysis [1] (r2=0.99), indicating that the signal near GLIS3 does co-localise between type 1 and type 2 diabetes with concordant direction of effect, as previously identified [10].
Discussion
Using genetic association summary statistics from European populations, we identified 42 regions that showed association with both type 1 and type 2 diabetes, with 81 conditionally independent association signals across those regions. Four signals (near CTRB1/BCAR1, INS, TMEM129 and PGM1) co-localised between the diseases, including a signal at the complex INS region for the first time, which was achieved by examining conditional summary statistics. However, in all four cases, the allele increasing risk for one disease was protective against the other. Examination of additional trans-ethnic and East Asian type 2 diabetes genetic analyses indicated that a fifth association, near GLIS3, is likely to co-localise between diseases, with concordant direction of effect.
Given the distinct mechanisms underlying beta cell dysfunction and cell death between the two diseases [11], it is perhaps unsurprising that no additional signals were detected with concordant direction of effect. However, the type 1 diabetes GWAS was much smaller than the type 2 diabetes analysis, and therefore had less statistical power to detect more subtle genetic effects. If a type 1 diabetes GWAS were to be performed with similar power to the type 2 diabetes GWAS, more regions might co-localise between the two diseases, but either the effects of these additional regions on type 1 diabetes would be small compared with the currently known associations or they would be rare variants with larger effect sizes.
That four of five co-localisation signals had opposite directions of effect implies a complex genetic relationship between the two diseases. While the directional discordance offers little hope for effective treatments for both diseases simultaneously at these particular targets, it can offer biological insight into the disease pathways that these regions act upon, and even if there is directional discordance, the genetics could be highlighting the same therapeutic target.
We did not replicate the findings that the associations near BCL11A, CENPW and THADA co-localise between the two diseases [3], despite overlapping samples and similar numbers of cases and controls in the type 1 diabetes GWAS. There are three possible reasons for this: 1) the previous study [3] examined co-localisation using weaker association signals, for example, the co-localisation near THADA was based on a type 1 diabetes association p value of 0.01; 2) we used a more stringent prior for co-localisation between the two diseases, as recently suggested [6] (5×10-6 vs 1×10-5); and 3) we used a more stringent posterior probability threshold to declare co-localisation (0.9 vs 0.5). Our increased stringency compared with the previous analysis [3], while increasing the probability that any identified shared signals will be true positives, may have decreased our sensitivity to detect all co-localisations. For example, by examining other large type 2 diabetes GWAS analyses and a larger type 1 diabetes genetic analysis, we conclude that the association near GLIS3 likely does co-localise between the two diseases, and with concordant directions of effect.
In conclusion, with current GWAS sample sizes, just five associations appear to co-localise between type 1 diabetes and type 2 diabetes, four with opposing direction of effect. Larger sample sizes would be required to identify the depth of genetically identified therapeutic targets to treat or prevent both diseases simultaneously.
Supplementary Information
(PDF 2124 kb)
(XLSX 43 kb)
Acknowledgements
We gratefully acknowledge all participants for allowing the analysis of their genotypic and phenotypic data.
Authors’ relationships and activities
MIM has served on advisory panels for Pfizer, Novo Nordisk and Zoe Global, has received honoraria from Merck, Pfizer, Novo Nordisk and Eli Lilly, and research funding from Abbvie, Astra Zeneca, Boehringer Ingelheim, Eli Lilly, Janssen, Merck, Novo Nordisk, Pfizer, Roche, Sanofi Aventis, Servier and Takeda. As of June 2019, MIM is an employee of Genentech and a holder of Roche stock. AM is an employee of Genentech since January 2020, and a holder of Roche stock. JRJI is an employee of Exploristics since June 2020. JAT serves on the advisory board of GSK. The remaining authors declare that there are no relationships or activities that might bias, or be perceived to bias, their work.
Contribution statement
JRJI carried out the type 1 diabetes meta-analysis and the co-localisation analyses, drafted the manuscript and approved the final version. AM carried out the type 2 diabetes meta-analysis and conditional analyses, revised the article for intellectual content and approved the final version. CS and FC were involved in data collection in the Sardinia collection and carried out the association testing in this collection, revised the article for intellectual content and approved the final version. DJMC provided statistical advice and input, and made contributions to interpretation of the data, revised the article for intellectual content and approved the final version. MIS provided biological insight, contributed towards interpretation of the data, revised the article for intellectual content and approved the final version. MIM and JAT oversaw the research, contributed towards the conception, design and data collection, revised the article for intellectual content and approved the final version. JAT is the guarantor of this work.
Abbreviations
- FDR
False discovery rate
- GWAS
Genome-wide association study
- H4PP
Posterior probability of co-localisation
- LD
Linkage disequilibrium
Funding
This work was funded by the JDRF (9-2011-253, 5-SRA-2015-130-A-N) and Wellcome (091157, 107212) to the Diabetes and Inflammation Laboratory, University of Oxford. Additional funding was obtained from Wellcome (090532, 098381, 106130, 212259) and the National Institute of Diabetes and Digestive and Kidney diseases (U01-DK105535). Computation used the Oxford Biomedical Research Computing (BMRC) facility, a joint development between the Wellcome Centre for Human Genetics and the Big Data Institute supported by Health Data Research UK and the NIHR Oxford Biomedical Research Centre. Financial support was provided by the Wellcome Trust Core Award Grant Number 203141/Z/16/Z. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health. Work was supported from grant U1301.2015/AI.1157.BE from Fondazione di Sardegna to Francesco Cucca.
Data availability
Type 1 diabetes summary statistics will be available through the GWAS catalogue (https://www.ebi.ac.uk/gwas/). Type 2 diabetes summary statistics are already publicly available.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Anubha Mahajan and John A. Todd contributed equally to the work.
Contributor Information
Jamie R. J. Inshaw, Email: jinshaw@well.ox.ac.uk
John A. Todd, Email: jatodd@well.ox.ac.uk
References
- 1.Onengut-Gumuscu S, Chen W-M, Burren O et al (2015) Fine mapping of type 1 diabetes susceptibility loci and evidence for colocalization of causal variants with lymphoid gene enhancers. Nat Genet 47:381–386 [DOI] [PMC free article] [PubMed]
- 2.Mahajan A, Taliun D, Thurner M et al (2018) Fine-mapping type 2 diabetes loci to single-variant resolution using high-density imputation and islet-specific epigenome maps. Nat Genet 50:1505–1513 [DOI] [PMC free article] [PubMed]
- 3.Aylward A, Chiou J, Okino M-L, Kadakia N, Gaulton KJ (2018) Shared genetic risk contributes to type 1 and type 2 diabetes etiology. Hum Mol Genet 10.1093/hmg/ddy314 [DOI] [PubMed]
- 4.McCarthy S, Das S, Kretzschmar W et al (2016) A reference panel of 64,976 haplotypes for genotype imputation. Nat Genet 48:1279–1283 [DOI] [PMC free article] [PubMed]
- 5.Guo H, Fortune MD, Burren OS, Schofield E, Todd JA, Wallace C (2015) Integration of disease association and eQTL data using a Bayesian colocalisation approach highlights six candidate causal genes in immune-mediated diseases. Hum Mol Genet 24:3305–3313 [DOI] [PMC free article] [PubMed]
- 6.Wallace C (2020) Eliciting priors and relaxing the single causal variant assumption in colocalisation analyses. PLoS Genet 16:e1008720. https://journals.plos.org/plosgenetics/article?id=10.1371/journal.pgen.1008720 [DOI] [PMC free article] [PubMed]
- 7.Hormozdiari F, van de Bunt M, Segre AV et al (2016) Colocalization of GWAS and eQTL Signals Detects Target Genes. Am J Hum Genet 99:1245–1260 [DOI] [PMC free article] [PubMed]
- 8.Vujkovic M, Keaton JM, Lynch JA et al (2019) Discovery of 318 novel loci for type-2 diabetes and related micro- and macrovascular outcomes among 1.4 million participants in a multi-ethnic meta-analysis. MedRxiv. 10.1101/19012690
- 9.Spracklen CN, Horikoshi M, Kim YJ et al (2020) Identification of type 2 diabetes loci in 433,540 East Asian individuals. Nature 582:240–245. 10.1038/s41586-020-2263-3 [DOI] [PMC free article] [PubMed]
- 10.Dooley J, Tian L, Schonefeldt S et al (2016) Genetic predisposition for beta cell fragility underlies type 1 and type 2 diabetes. Nat Genet 48:519–527 [DOI] [PMC free article] [PubMed]
- 11.Eizirik DL, Pasquali L, Cnop M (2020) Pancreatic beta-cells in type 1 and type 2 diabetes mellitus: different pathways to failure. Nat Rev Endocrinol 16:349–362. 10.1038/s41574-020-0355-7 [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PDF 2124 kb)
(XLSX 43 kb)
Data Availability Statement
Type 1 diabetes summary statistics will be available through the GWAS catalogue (https://www.ebi.ac.uk/gwas/). Type 2 diabetes summary statistics are already publicly available.