Abstract
The aim of this study was to evaluate the effects of cannabidiol (CBD) on the behavioural and gene expression changes in a new animal model of spontaneous cocaine withdrawal. For this purpose, male CD-1 mice were exposed to progressive increasing doses of cocaine for 12 days (15 to 60 mg/kg/day, i.p.), evaluating spontaneous cocaine withdrawal 6 h after the last cocaine administration. The effects of CBD (10, 20, and 40 mg/kg, i.p.) were evaluated on cocaine withdrawal–induced alterations in motor activity, somatic signs, and anxiety-like behaviour. Furthermore, gene expression changes in dopamine transporter (DAT) and tyrosine hydroxylase (TH) in the ventral tegmental area, and in cannabinoid receptors 1 (CNR1) and 2 (CNR2) in the nucleus accumbens, were analysed by real-time PCR. The results obtained in the study showed that mice exposed to the spontaneous cocaine withdrawal model presented increased motor activity, somatic withdrawal signs, and high anxiety-like behaviour. Interestingly, the administration of CBD normalized motor and somatic signs disturbances and induced an anxiolytic effect. Moreover, the administration of CBD blocked the increase of DAT and TH gene expression in mice exposed to the cocaine withdrawal, regulated the decrease of CNR1 and induced an additional upregulation of CNR2 gene expression. Thus, this model of spontaneous cocaine withdrawal induces clear behavioural and gene expression changes in mice. Interestingly, CBD alleviates these behavioural and gene expression alterations suggesting its potential for the management of cocaine withdrawal.
Supplementary Information
The online version contains supplementary material available at 10.1007/s13311-020-00976-6.
Key Words: Cocaine, withdrawal, cannabidiol, mice, mRNA
Introduction
Cocaine use disorder (CUD) is a chronic and relapsing disorder characterized by compulsive drug seeking and drug use despite the negative consequences. The most recent data suggest that CUD remains a major public health problem with an estimated 20 million users, making this drug of abuse the most predominant psychostimulant used in the world [1]. Recent epidemiological reports indicate that the number of cocaine users is increasing, driving an associated rise in socio-economic and legal complications as well as hospitalisations caused by cocaine dependence [2].
Cocaine is a psychostimulant drug with high potential for addiction because of its short half-life and dopaminergic mechanism of action. Up to 5 to 6% of cocaine users will develop cocaine dependence within the first year of use [3] which involves a recurring cycle of intoxication, bingeing, withdrawal, and craving, resulting in excessive drug use [4]. The most common symptoms reported by cocaine users are cardiac with cocaine addiction standing out as the leading cause of death among adults using illicit substances. Indeed, cocaine use was reported as a risk factor in 25% of nonfatal myocardial infarctions in younger people [3]. Moreover, when patients quit using this drug after a long period of consumption, cocaine withdrawal causes both somatic physical and emotional symptoms [5]. People with most severe withdrawal symptoms and the highest scores on the Cocaine Selective Severity Assessment scale are more likely to drop out of treatment prematurely or to have a worse response to the selected treatment [6–10]. The severity of cocaine withdrawal syndrome could be correlated to the degree of dependence or increased sensitivity to its effects [11]. Thus, given the absence of specific pharmacological tools for treating CUD, managing cocaine withdrawal could be an effective way to break the cycle of addiction and prevent relapse.
In this sense, appropriate animal models could be essential tools for improving the knowledge and understanding of the behavioural and neurobiological mechanisms involved in cocaine withdrawal. Unfortunately, only a few current animal models of cocaine withdrawal have been described in the literature. Some studies simulated cocaine abstinence by administering the same dose of cocaine for several days or by increasing cocaine doses once a day combined with periods of abstinence [12–16]. However, human users take cocaine several times a day at increasing doses, and abstinence syndrome appears soon after drug consumption stops. Therefore, these animal models only partially reproduce the principal clinical features of human cocaine addiction [17], highlighting the need for new animal models to better simulate cocaine withdrawal syndrome from a translational point of view. These should reproduce the frequent and escalating doses of cocaine consumed over a long period of time and the appearance of withdrawal symptoms within a few hours after cessation. The development of a suitable animal model of cocaine withdrawal can help generate more precise knowledge of the neurobiological mechanisms underlying this complex phase of cocaine addiction, enabling the identification of new therapeutic targets and specific pharmacological strategies.
No regulatory agencies have approved specific drugs with established efficacy for treating cocaine withdrawal. However, several recent preclinical and clinical studies have shown that cannabidiol (CBD), a main constituent of the Cannabis sativa plant without potential for becoming a drug of abuse [18], may be a promising therapeutic tool for CUD management. CBD can interact with more than 65 different targets, producing anxiolytic, antidepressant, antipsychotic, and neuroprotective effects [19]. In addition, CBD has been proposed as a pharmacological candidate for treating substance use disorders [20–22]. Several preclinical reports have also suggested that CBD attenuates cue-induced cocaine seeking in rats after withdrawal [23] along with cocaine-induced conditioned place preference [24, 25]. Researchers have also pointed to the efficacy of CBD for reducing cocaine consumption and the progressive ratio in a cocaine self-administration paradigm [25]. Therefore, more studies to assess CBD for the pharmacological treatment of CUD are warranted.
The main aim of this study was to evaluate the effects of CBD on behavioural and gene expression in a new model of spontaneous cocaine withdrawal in mice, induced by repeated administration of increasing doses of cocaine for 12 days. Anxiety-like behaviour (light-dark box), motor activity (distance travelled in the open-field test) and somatic withdrawal signs (number of rearings, rubbings, groomings, and diggings) were assessed 6 h after the last cocaine administration. Furthermore, gene expression analyses were carried out by real-time polymerase chain reaction (real-time PCR) to evaluate changes induced by cocaine withdrawal in specific targets involved in cocaine addiction and withdrawal. Relative gene expression analyses of dopamine transporter (DAT) and tyrosine hydroxylase (TH) in the ventral tegmental area (VTA), and cannabinoid receptors 1 (CNR1) and 2 (CNR2) in the nucleus accumbens (NAcc), were also performed.
Methods
Mice
A total of 100 CD1 male mice were purchased from Charles River laboratories (Lille, France). Mice, weighing 20–25 g, were housed in individual cages (40 cm × 25 cm × 22 cm) under controlled environmental conditions (temperature, 21 ± 2 °C, relative humidity, 60 ± 10%, and 12 h light-dark cycle, lights on from 08:00 to 20:00). One week after mice adaptation to the animal room, cocaine administration was initiated. Behavioural evaluations were carried out during the light cycle. All studies complied with the Spanish Royal Decree 53/2013, the Spanish Law 32/2007, and the European Union Directive of 22 September 2010 (2010/63/UE) regulating the care of experimental animals and were approved by the ethics committee of Miguel Hernandez University.
Drugs
Cocaine (cocaine hydrochloride) was obtained from the Spanish Drugs Agency (AEMPS, Madrid, Spain) and dissolved in saline (0.9% sodium chloride) to prepare required doses immediately before intraperitoneal (i.p.) administration. Cannabidiol (CBD) was obtained from STI Pharmaceuticals (Essex, UK) and was dissolved in ethanol:cremophor:saline (1:1:18) immediately before use to obtain the required doses of 10, 20 and 40 mg/kg (i.p.). CBD was given 90 min before any behavioural evaluation, according to the pharmacokinetics of CBD [26] and previous studies from our laboratory [20, 21, 27].
Animal Model of Spontaneous Cocaine Withdrawal
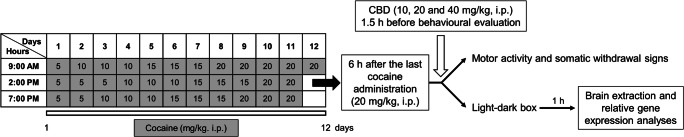
Spontaneous cocaine withdrawal was induced by first administering increasing doses of cocaine starting with 15 mg/kg/day at day 1 and rising to 60 mg/kg/day at day 11 (Fig. 1). Cocaine was administered 3 times a day, every 5 h during the light cycle for 12 consecutive days. Spontaneous cocaine withdrawal-induced behavioural and gene expression alterations were evaluated 6 h after the last cocaine administration at day 12 (20 mg/kg) during the light cycle (Fig. 1).
Fig. 1.
Timeline diagram of the experimental procedure used for the development of an animal model of spontaneous cocaine withdrawal syndrome. Cocaine administration (15 mg/kg/day–60 mg/kg/day, i.p.) was performed during 12 consecutive days. Six hours after last cocaine dose, the effects of CBD (10, 20 and 40 mg/kg, i.p.) on spontaneous cocaine withdrawal syndrome were evaluated by the open-field and light-dark box paradigms
Experimental Design
Motor Activity and Somatic Withdrawal Signs
A set of 50 CD1 mice were used to evaluate motor activity and somatic withdrawal signs in the open-field paradigm. For this purpose, 6 h after the last cocaine administration (day 12, 20 mg/kg), mice were placed into individual methacrylate boxes (25 cm × 25 cm × 25 cm) and videotaped for 15 min to evaluate somatic signs associated with abstinence (number of rearings, rubbings, groomings, and diggings). Simultaneously, motor responses were also evaluated by measuring the total distance travelled for 15 min with the SMART programme (Panlab). Ninety minutes before the behavioural evaluation, CBD (10, 20, and 40 mg/kg) or its corresponding vehicle were administered (i.p.) to assess their effects on spontaneous cocaine withdrawal signs and motor activity.
Light-Dark Box Test
An additional set of 50 CD1 mice were used to evaluate anxiety-like behaviour changes induced by cocaine abstinence. Four and a half hours after last cocaine administration (day 12, 20 mg/kg), CBD (10, 20, and 40 mg/kg) or its corresponding vehicle was administered and 90 min later mice were individually tested for 5 min in the light-dark box paradigm. The time spent in the lighted box and the number of transitions were recorded for each session. After this behavioural evaluation, mice were sacrificed by cervical dislocation and brain samples were removed to analyse relative gene expression in the targets of interest.
Relative Gene Expression Analyses by Real-Time PCR
Relative gene expression analyses of DAT and TH in the VTA, and CNR1, and CNR2 in the NAcc, were carried out in vehicle and cocaine-treated mice to assess the effects of spontaneous cocaine withdrawal on anxiety-like behaviour and its pharmacological modulation by CBD administration. Briefly, mice were sacrificed 150 min after administration of CBD or vehicle and brain samples were removed from the skull and frozen at − 80 °C. These samples were used to obtain coronal sections (500 μm) of regions of interest in a cryostat (− 10 °C) according to Paxinos and Franklin’s atlas [28]. Brain nuclei of interest were microdissected following Palkovits’s method and as previously modified by our group [29, 30]. Total RNA was extracted from brain micropunches with TRI Reagent (Applied Biosystems, Madrid, Spain) and reverse transcription was carried out to obtain the complementary DNA (cDNA) (4374966, High-Capacity cDNA Reverse Transcription Kit with RNase Inhibitor, Applied Biosystems, Madrid, Spain). To perform the real-time PCR, 6.25 μl of water with DEPC (diethyl pyrocarbonate, RNAase inhibitor), 5 μl of the cDNA, 11.25 μl of the TaqmanTM Master Mix (4369514, Applied Biosystems, Madrid, Spain), and 1.25 μl of the corresponding Taqman assay were added in each well (4346907, Applied Biosystems, Madrid, Spain). Quantitative analyses of the relative expression of DAT (Mm00438388_m1), TH (Mm00447546_m1), CNR1 (Mm00432621_s1), and CNR2 (Mm00438286_m1) genes were performed on the StepOne Sequence Detector System (Applied Biosystems, Madrid, Spain). All reagents were used following the manufacturer’s instructions. The reference gene used was 18S rRNA (Mm03928990_g1), and data for each target was normalized to the endogenous reference gene. The fold change in target gene expression was calculated using the 2ΔΔ−Ct method [31].
Data and Statistical Analyses
Statistical analyses were performed using one-way ANOVA followed by Student-Newman-Keul’s test when comparing different experimental groups. Differences were considered significant if the probability of type-I error or alpha was less than 5%. SigmaPlot 11 software (Systat software Inc., Chicago, IL, USA) was used for all statistical analyses.
Results
Effects of CBD on Motor Activity and Somatic Signs Induced by Spontaneous Cocaine Withdrawal
Open-Field Paradigm
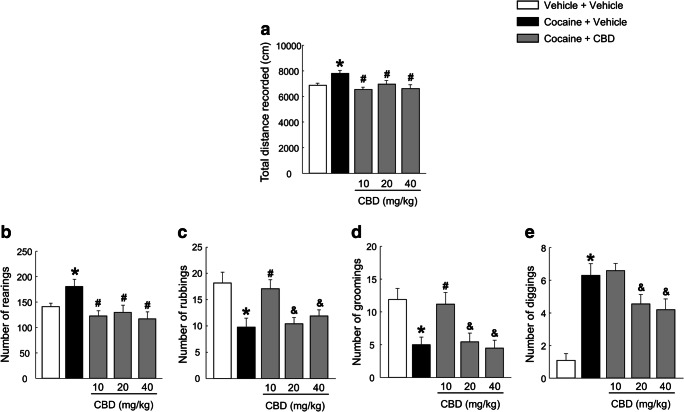
Mice exposed to the spontaneous cocaine withdrawal model showed significantly increased total distance values compared with vehicle-treated mice. CBD administration fully blocked this increase at all three administered doses (Fig. 2(A), one-way ANOVA, F(4, 48) = 4.173, P = 0.006).
Fig. 2.
Evaluation of CBD (10, 20 and 40 mg/kg) effects on motor activity and somatic signs alterations induced by the spontaneous cocaine withdrawal model, in the open-field paradigm. Columns represent the means and vertical lines ± SEM of the total distance recorded (cm) (A) and the number of rearings (B), rubbings (C), groomings (D) and diggings (E). *, values from COCA-VEH-treated group that are significantly different from VEH-VEH controls (one-way ANOVA, P < 0.05). #, values from COCA-CBD-treated groups that are significantly different from COCA-VEH-treated group (one-way ANOVA, P < 0.05). &, values from COCA-CBD (20 and 40 mg/kg)-treated groups that are significantly different from COCA-CBD 10 mg/kg treated group (one-way ANOVA, P < 0.05). Number of mice per group n = 9–10
Withdrawal Somatic Signs
The one-way ANOVA showed an increase in the number of rearings and diggings after cocaine treatment cessation compared with the vehicle-treated group. All three doses of CBD (10, 20, and 40 mg/kg) completely normalized the increased number of rearings (Fig. 2(B), one-way ANOVA, F(4, 48) = 4.589, P = 0.003), whereas only 20 and 40 mg/kg doses of CBD significantly reduced the number of diggings (Fig. 2(E), one-way ANOVA, F(4, 48) = 15.025, P < 0.001). Mice exposed to the spontaneous cocaine withdrawal model presented a reduced number of rubbings (Fig. 2(C), one-way ANOVA, F(4, 48) = 5.856, P < 0.001) and groomings (Fig. 2(D), one-way ANOVA, F(4, 48) = 6.248, P < 0.001), which were normalized only with the lowest employed dose of CBD (10 mg/kg).
Effects of CBD on Anxiety-Like Behaviour Induced by Spontaneous Cocaine Withdrawal
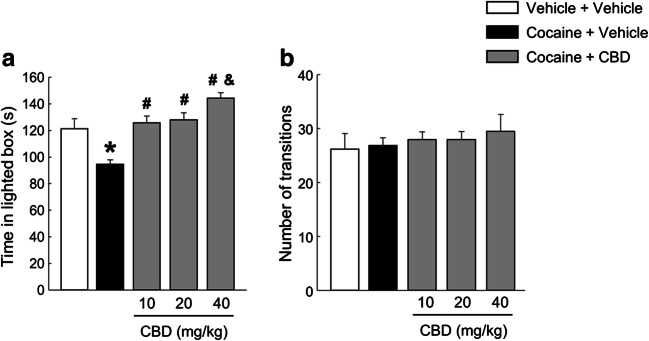
After the cessation of cocaine treatment, cocaine + vehicle-treated mice exhibited a significant decrease in the time spent in the lighted box. The administration of 10 and 20 mg/kg doses of CBD fully blocked this anxiety-like behaviour, while the dose of 40 mg/kg induced an additional anxiolytic effect (Fig. 3(A), one-way ANOVA, F(4, 48) = 12.573, P < 0.001). Furthermore, no changes were observed in the number of transitions between the five groups (Fig. 3(B), one-way ANOVA, F(4, 48) = 0.326, P = 0.859).
Fig. 3.
Evaluation of CBD (10, 20 and 40 mg/kg) effects on increased anxiety-like behaviour induced by the spontaneous cocaine withdrawal model, in the light-dark box paradigm. Columns represent the means and vertical lines ± SEM of the time spent in the lighted box (s) (A) and the number of transitions (B). *, values from COCA-VEH-treated mice that are significantly different from VEH-VEH-treated mice (one-way ANOVA, P < 0.001). #, values from COCA-CBD-treated groups that are significantly different from COCA-VEH-treated group (one-way ANOVA, P < 0.001). &, values from COCA-CBD (40 mg/kg)-treated group that are significantly different from COCA-CBD (10 and 20 mg/kg) groups (one-way ANOVA, P < 0.05). Number of mice per group n = 9–10
Effects of CBD on Changes in the DAT, TH, CNR1, and CNR2 Gene Expression Induced by Spontaneous Cocaine Withdrawal
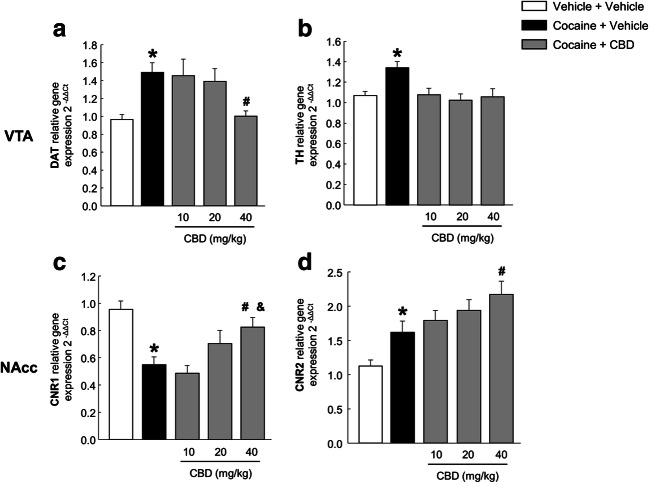
Compared with control mice, mice exposed to the animal model of spontaneous cocaine withdrawal showed an increase in DAT relative gene expression, which was normalized with CBD (40 mg/kg) (Fig. 4(A), one-way ANOVA, F(4, 47) = 5.019, P < 0.01). In addition, spontaneous cocaine withdrawal induced an increase in TH relative gene expression which was completely normalized with all three administered doses of CBD (10, 20, and 40 mg/kg) (Fig. 4(B), one-way ANOVA, F(4, 42) = 4.878, P < 0.01).
Fig. 4.
Relative gene expression analyses of dopamine transporter (DAT) (A) and tyrosine hydroxylase (TH) in the ventral tegmental area (VTA) (B), and cannabinoid receptors 1 (CNR1) (C) and 2 (CNR2) (D) in the nucleus accumbens (NAcc). Columns represent the means and vertical lines ± SEM of 2−ΔΔCt. *, values from COCA-VEH-treated group that are significantly different from VEH-VEH-treated group (one-way ANOVA, P < 0.01). #, values from COCA-CBD-treated group that are significantly different from those treated with COCA-VEH (one-way ANOVA, P < 0.05). &, values from COCA-CBD (40 mg/kg)-treated group that are significantly different from groups treated with COCA-CBD (10 mg/kg) (one-way ANOVA, P < 0.01). Number of mice per group n = 8–10
One-way ANOVA revealed decreased CNR1 relative gene expression in mice exposed to the animal model of spontaneous cocaine withdrawal. CBD produced a significant upregulation of CNR1 gene expression at the dose of 40 mg/kg (Fig. 4(C), one-way ANOVA, F(4, 47) = 7.292, P < 0.001). In addition, CNR2 relative gene expression increased in mice with spontaneous cocaine withdrawal, and CBD administration produced an additional increase, reaching statistical significance at the dose of 40 mg/kg (Fig. 4(D), one-way ANOVA, F(4, 45) = 8.484, P < 0.001).
Discussion
The results of the present study reveal that the administration of CBD significantly regulated the behavioural and gene expression alterations induced in a new animal model of spontaneous cocaine withdrawal. These findings are supported by the following observations: (i) 6 h after cessation of cocaine administration, mice presented increased motor activity, number of rearings, and diggings, and anxiety-like behaviour, along with decreased rubbing and grooming behaviours; (ii) the administration of CBD reduced motor activity, normalized the spontaneous cocaine withdrawal-induced somatic signs and induced an anxiolytic effect; and (iii) the administration of CBD blocked the increase of DAT and TH relative gene expression in the VTA, modulated the decrease of CNR1 gene expression in the NAcc and produced an additional increase of CNR2 gene expression in the NAcc induced by the spontaneous cocaine withdrawal.
The development of new animal models of cocaine withdrawal is critical for identifying new therapeutic targets. In this study, we designed a new animal model of spontaneous cocaine withdrawal by using increasingly high doses of cocaine for 12 days. The escalation was carefully planned to increase the morning dose first to avoid coinciding with the peak night-time activity and to minimize adverse cardiac effects [32]. Furthermore, the administration of cocaine every 5 h during the light cycle contrasts with other reported cocaine withdrawal models where cocaine was given three times in the morning, 1 h apart [14, 15, 33]. We believe that the pattern used in our study simulates more closely the intermittent consumption of cocaine described in cocaine users avoiding long periods of abstinence [34]. Finally, considering the pharmacokinetic properties of cocaine and the clinical onset of the abstinence, the evaluation of cocaine withdrawal–induced disturbances was carried out 6 h after the last administration. This allowed the detection of enhanced withdrawal signs such as increased motor activity and anxiety-like behaviour [13–15]. Interestingly, during cocaine withdrawal, we found an increase in the number of rearings and diggings and a decrease in the number of rubbings and groomings. Furthermore, the evaluation of spontaneous cocaine withdrawal (6 h after the last administration) revealed significantly increased anxiety-like behaviour unlike other models where anxiety-like behaviour was assessed days after the last administration [13–15].
In recent years, several studies have focused on the potential of CBD for treatment of substance use disorders. This is probably because of its anxiolytic, antidepressant, antipsychotic, and neuroprotective actions [35] and the lack of risk for becoming a drug of abuse [18]. Indeed, CBD effectively modulates the reinforcing and motivational properties of different drugs of abuse such as alcohol, cannabis, or opiates [20, 21, 36–38], along with context- and stress-induced cocaine seeking in rats [23], and cocaine self-administration [25]. In this study, the administration of CBD normalized the increase in motor activity induced by spontaneous cocaine withdrawal. The intervention significantly reduced the number of rearings and diggings at intermediate and high doses and normalized the number of rubbings and groomings at the lowest dose. This dose-dependent modulation of withdrawal signs could be explained, at least in part, by the bell-shaped dose-response curve of CBD-mediated regulation of anxiety-like behaviour [35]. In a previous study, our group found that CBD regulated hypothalamic-pituitary-adrenal (HPA) axis activation after an acute stress only at the lowest dose [27]. Moreover, CBD normalized the increased anxiety-like behaviour in mice exposed to the spontaneous cocaine withdrawal model, reaching an additional anxiolytic action at the highest dose.
Gene expression analyses were performed to identify alterations in specific targets that may give rise to the withdrawal-induced behavioural disturbances and their regulation by CBD. We found increased DAT relative gene expression in vehicle-treated mice exposed to the cocaine withdrawal model. The main mechanism of action of cocaine is the blockade of the DAT function preventing dopamine reuptake and leading to an upregulation after repeated administration [39–42]. Interestingly, CBD (40 mg/kg) completely normalized DAT gene expression, suggesting an inhibitory action consistent with a previous study that inferred that CBD may act as a low-potency inhibitor of DAT in rat striatal terminals [43]. Considering its crucial role in the actions induced by cocaine, it is possible to hypothesise that this effect may be closely related with the CBD-mediated improvement of cocaine withdrawal behaviours.
Previous results suggested that CBD modulates dopaminergic neurotransmission in the mesolimbic system. First of all, microdialysis perfusion of CBD into the lateral hypothalamus increases dopamine extracellular levels in the NAcc [44]; secondly, the administration of CBD into the NAcc blocks VTA dopaminergic neuronal sensitization induced by the amphetamine [45], and finally, CBD attenuates cocaine-induced increases in extracellular dopamine in the NAcc [46]. Accordingly, cocaine withdrawal also significantly increases TH gene expression in the VTA and this action is completely blocked by all doses of CBD. Our group has already reported the potential of CBD to reduce enhanced TH gene expression in animal models of alcohol consumption [21, 37]. Taken together, it is tempting to suggest that the CBD-mediated regulation of cocaine-induced mesolimbic dopaminergic activation may be involved in the regulation of behavioural alterations during cocaine withdrawal.
Since both CB1r and CB2r may be involved in the mechanism of action of CBD and in cocaine addiction, several studies measured their gene expressions in the NAcc [47–53]. Some authors reported the involvement of CB1r in the reinforcing and motivational properties of cocaine [54–57]. CNR1 gene expression was lower in mice exposed to the cocaine withdrawal model and upregulated with CBD. A similar effect was found in the hippocampus of CBD-treated mice exposed to a cocaine self-administration paradigm [58]. Our group already reported on the normalization of CNR1 gene expression changes in the NAcc [20, 21, 37]. Indeed, CBD acts as an indirect agonist of CB1r by the inhibition of FAAH activity and the blockade of anandamide reuptake [59]. Some reports also suggest that CBD might act as a negative allosteric modulator of CB1r [60, 61]. Further studies are needed to identify the mechanism involved in the actions of CBD on CNR1 gene.
Cocaine withdrawal syndrome was associated with increased CNR2 gene expression in the NAcc. Overexpression of CB2r has resulted in cocaine-induced conditioned place aversion and reduced cocaine-self-administration in mice [49]. Furthermore, the use of conditional knockout mice to delete the CNR2 gene in midbrain DA neurons revealed the critical involvement of CB2r in attenuating psychomotor and rewarding effects of cocaine [56]. Recently, it was reported that the CB2r agonist JWH133 inhibited the acquisition and expression of cocaine sensitization and conditioned place preference [57]. A plausible hypothesis is that cocaine withdrawal–induced upregulation of CNR2 gene expression may be related with a homeostatic compensatory effect. Interestingly, in this study, the administration of CBD additionally increased CNR2 gene expression. This effect may be due, at least in part, to the inverse agonism, or antagonism on CB2r produced by CBD [62]. A recent report suggested that the attenuation of cocaine rewarding effects induced by CBD was mediated by CB2r [46]. It is plausible that CBD-induced CNR2 upregulation is at least partly responsible for modulating cocaine withdrawal.
In conclusion, the results of this study provide unequivocal evidence of the efficacy of CBD for improving anxiety-like behaviour, motor activity, and somatic sign alterations induced by a new animal model of spontaneous cocaine withdrawal. Gene expression analyses provide relevant information about the neurobiological basis of this model and of the mechanisms involved in the actions of CBD. The most important limitation of our study was the use of a single strain of male mice, so further research aimed at assessing the suitability of this promising animal model of cocaine withdrawal could include female mice to evaluate gender-dependent effects or explore potential strain-dependent differences in the behavioural and brain gene expression alterations induced by spontaneous cocaine withdrawal. In addition, future studies are needed to clarify the neurobiological mechanisms involved in cocaine withdrawal and facilitate the development of new drugs to prevent relapse and to explore the potential of CBD to treat cocaine withdrawal.
Supplementary Information
(PDF 1196 kb)
Acknowledgments
The authors would like to thank the technical support provided by José Mulet in this study.
This research was supported by ‘Instituto de Salud Carlos III’ (RETICS, RD12/0028/0019 and RD16/0017/0014), ‘Plan Nacional Sobre Drogas’ (PNSD 2016/016) and ‘Ministerio de Economía y Competitividad’ (FIS, PI14/00438) to J.M.
Required Author Forms
Disclosure forms provided by the authors are available with the online version of this article.
Abbreviations
- CBD
Cannabidiol
- CBr
Cannabinoid receptor
- CNR
Cannabinoid receptor gene
- CUD
Cocaine use disorder
- DAT
Dopamine transporter
- NAcc
Nucleus accumbens
- SMART
Spontaneous Motor Activity Recording and Tracking
- TH
Tyrosine hydroxylase
- VTA
Ventral tegmental area
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Richards JR, Garber D, Laurin EG, Albertson TE, Derlet RW, Amsterdam EA, et al. Treatment of cocaine cardiovascular toxicity: a systematic review. Clin Toxicol (Phila). 2016;54(5):345–64. doi: 10.3109/15563650.2016.1142090. [DOI] [PubMed] [Google Scholar]
- 2.Mena G, Giraudon I, Alvarez E, Corkery JM, Matias J, Grasaasen K, et al. Cocaine-related health emergencies in Europe: a review of sources of information, trends and implications for service development. Eur Addict Res. 2013;19(2):74–81. doi: 10.1159/000341719. [DOI] [PubMed] [Google Scholar]
- 3.Ryan SA. Cocaine Use in Adolescents and Young Adults. Pediatr Clin North Am. 2019;66(6):1135–47. doi: 10.1016/j.pcl.2019.08.014. [DOI] [PubMed] [Google Scholar]
- 4.Agurto C, Norel R, Pietrowicz M, Parvaz M, Kinreich S, Bachi K, et al. Speech Markers for Clinical Assessment of Cocaine Users. Proc IEEE Int Conf Acoust Speech Signal Process. 2019;2019:6391–4. doi: 10.1109/icassp.2019.8682691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sofuoglu M, Dudish-Poulsen S, Poling J, Mooney M, Hatsukami DK. The effect of individual cocaine withdrawal symptoms on outcomes in cocaine users. Addict Behav. 2005;30(6):1125–34. doi: 10.1016/j.addbeh.2004.10.010. [DOI] [PubMed] [Google Scholar]
- 6.Mulvaney FD, Alterman AI, Boardman CR, Kampman K. Cocaine abstinence symptomatology and treatment attrition. J Subst Abuse Treat. 1999;16(2):129–35. doi: 10.1016/s0740-5472(98)00017-8. [DOI] [PubMed] [Google Scholar]
- 7.Sofuoglu M, Poling J, Gonzalez G, Gonsai K, Kosten T. Cocaine withdrawal symptoms predict medication response in cocaine users. Am J Drug Alcohol Abuse. 2006;32(4):617–27. doi: 10.1080/00952990600920680. [DOI] [PubMed] [Google Scholar]
- 8.Kampman KM, Volpicelli JR, Mulvaney F, Rukstalis M, Alterman AI, Pettinati H, et al. Cocaine withdrawal severity and urine toxicology results from treatment entry predict outcome in medication trials for cocaine dependence. Addict Behav. 2002;27(2):251–60. doi: 10.1016/s0306-4603(01)00171-x. [DOI] [PubMed] [Google Scholar]
- 9.Kampman KM, Alterman AI, Volpicelli JR, Maany I, Muller ES, Luce DD, et al. Cocaine withdrawal symptoms and initial urine toxicology results predict treatment attrition in outpatient cocaine dependence treatment. Psychol Addict Behav. 2001;15(1):52–9. doi: 10.1037/0893-164x.15.1.52. [DOI] [PubMed] [Google Scholar]
- 10.Kampman KM, Volpicelli JR, McGinnis DE, Alterman AI, Weinrieb RM, D'Angelo L, et al. Reliability and validity of the Cocaine Selective Severity Assessment. Addict Behav. 1998;23(4):449–61. doi: 10.1016/s0306-4603(98)00011-2. [DOI] [PubMed] [Google Scholar]
- 11.Sofuoglu M, Dudish-Poulsen S, Brown SB, Hatsukami DK. Association of cocaine withdrawal symptoms with more severe dependence and enhanced subjective response to cocaine. Drug Alcohol Depend. 2003;69(3):273–82. doi: 10.1016/s0376-8716(02)00328-9. [DOI] [PubMed] [Google Scholar]
- 12.Li M, Xu P, Xu Y, Teng H, Tian W, Du Q, et al. Dynamic Expression Changes in the Transcriptome of the Prefrontal Cortex after Repeated Exposure to Cocaine in Mice. Front Pharmacol. 2017;8:142. doi: 10.3389/fphar.2017.00142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Valzachi MC, Teodorov E, Marcourakis T, Bailey A, Camarini R. Enhancement of behavioral sensitization, anxiety-like behavior, and hippocampal and frontal cortical CREB levels following cocaine abstinence in mice exposed to cocaine during adolescence. PLoS One. 2013;8(10):e78317. doi: 10.1371/journal.pone.0078317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ledesma JC, Aguilar MA, Gimenez-Gomez P, Minarro J, Rodriguez-Arias M. Adolescent but not adult ethanol binge drinking modulates cocaine withdrawal symptoms in mice. PLoS One. 2017;12(3):e0172956. doi: 10.1371/journal.pone.0172956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Aguilar MA, Ledesma JC, Rodriguez-Arias M, Penalva C, Manzanedo C, Minarro J, et al. Adolescent Exposure to the Synthetic Cannabinoid WIN 55212-2 Modifies Cocaine Withdrawal Symptoms in Adult Mice. Int J Mol Sci. 2017;18(6). [DOI] [PMC free article] [PubMed]
- 16.Stoker AK, Markou A. Withdrawal from chronic cocaine administration induces deficits in brain reward function in C57BL/6J mice. Behav Brain Res. 2011;223(1):176–81. doi: 10.1016/j.bbr.2011.04.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Benuck M, Lajtha A, Reith ME. Pharmacokinetics of systemically administered cocaine and locomotor stimulation in mice. J Pharmacol Exp Ther. 1987;243(1):144–9. [PubMed] [Google Scholar]
- 18.Viudez-Martinez A, Garcia-Gutierrez MS, Medrano-Relinque J, Navarron CM, Navarrete F, Manzanares J. Cannabidiol does not display drug abuse potential in mice behavior. Acta Pharmacol Sin. 2019;40(3):358–64. doi: 10.1038/s41401-018-0032-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Elsaid S, Le Foll B. The complexity of pharmacology of cannabidiol (CBD) and its implications in the treatment of brain disorders. Neuropsychopharmacology. 2019. [DOI] [PMC free article] [PubMed]
- 20.Navarrete F, Aracil-Fernandez A, Manzanares J. Cannabidiol regulates behavioural alterations and gene expression changes induced by spontaneous cannabinoid withdrawal. Br J Pharmacol. 2018;175(13):2676–88. doi: 10.1111/bph.14226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Viudez-Martinez A, Garcia-Gutierrez MS, Navarron CM, Morales-Calero MI, Navarrete F, Torres-Suarez AI, et al. Cannabidiol reduces ethanol consumption, motivation and relapse in mice. Addict Biol. 2018;23(1):154–64. doi: 10.1111/adb.12495. [DOI] [PubMed] [Google Scholar]
- 22.Prud'homme M, Cata R, Jutras-Aswad D. Cannabidiol as an Intervention for Addictive Behaviors: A Systematic Review of the Evidence. Subst Abuse. 2015;9:33–8. doi: 10.4137/SART.S25081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gonzalez-Cuevas G, Martin-Fardon R, Kerr TM, Stouffer DG, Parsons LH, Hammell DC, et al. Unique treatment potential of cannabidiol for the prevention of relapse to drug use: preclinical proof of principle. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology. 2018;43(10):2036–45. doi: 10.1038/s41386-018-0050-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Parker LA, Burton P, Sorge RE, Yakiwchuk C, Mechoulam R. Effect of low doses of delta9-tetrahydrocannabinol and cannabidiol on the extinction of cocaine-induced and amphetamine-induced conditioned place preference learning in rats. Psychopharmacology (Berl). 2004;175(3):360–6. doi: 10.1007/s00213-004-1825-7. [DOI] [PubMed] [Google Scholar]
- 25.Lujan MA, Castro-Zavala A, Alegre-Zurano L, Valverde O. Repeated Cannabidiol treatment reduces cocaine intake and modulates neural proliferation and CB1R expression in the mouse hippocampus. Neuropharmacology. 2018;143:163–75. doi: 10.1016/j.neuropharm.2018.09.043. [DOI] [PubMed] [Google Scholar]
- 26.Deiana S, Watanabe A, Yamasaki Y, Amada N, Arthur M, Fleming S, et al. Plasma and brain pharmacokinetic profile of cannabidiol (CBD), cannabidivarine (CBDV), Delta(9)-tetrahydrocannabivarin (THCV) and cannabigerol (CBG) in rats and mice following oral and intraperitoneal administration and CBD action on obsessive-compulsive behaviour. Psychopharmacology (Berl). 2012;219(3):859–73. doi: 10.1007/s00213-011-2415-0. [DOI] [PubMed] [Google Scholar]
- 27.Viudez-Martinez A, Garcia-Gutierrez MS, Manzanares J. Cannabidiol regulates the expression of hypothalamus-pituitary-adrenal axis-related genes in response to acute restraint stress. J Psychopharmacol. 2018;32(12):1379–84. doi: 10.1177/0269881118805495. [DOI] [PubMed] [Google Scholar]
- 28.Paxinos G, Franklin KBJ. The mouse brain in stereotaxic coordinates. New York: Academic Press. Harcourt Science and Technology Company; 2001. [Google Scholar]
- 29.Palkovits M. Punch sampling biopsy technique. Methods Enzymol. 1983;103:368–76. doi: 10.1016/s0076-6879(83)03025-6. [DOI] [PubMed] [Google Scholar]
- 30.Navarrete F, Perez-Ortiz JM, Manzanares J. Pregabalin and topiramate mediated regulation of cognitive and motor impulsivity in DBA/2 mice. Br J Pharmacol. 2012. [DOI] [PMC free article] [PubMed]
- 31.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25(4):402–8. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 32.Richards JR, Hollander JE, Ramoska EA, Fareed FN, Sand IC, Izquierdo Gomez MM, et al. beta-Blockers, Cocaine, and the Unopposed alpha-Stimulation Phenomenon. J Cardiovasc Pharmacol Ther. 2017;22(3):239–49. doi: 10.1177/1074248416681644. [DOI] [PubMed] [Google Scholar]
- 33.Zhang Y, Schlussman SD, Rabkin J, Butelman ER, Ho A, Kreek MJ. Chronic escalating cocaine exposure, abstinence/withdrawal, and chronic re-exposure: effects on striatal dopamine and opioid systems in C57BL/6J mice. Neuropharmacology. 2013;67:259–66. doi: 10.1016/j.neuropharm.2012.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kawa AB, Allain F, Robinson TE, Samaha AN. The transition to cocaine addiction: the importance of pharmacokinetics for preclinical models. Psychopharmacology (Berl). 2019;236(4):1145–57. doi: 10.1007/s00213-019-5164-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Campos AC, Moreira FA, Gomes FV, Del Bel EA, Guimaraes FS. Multiple mechanisms involved in the large-spectrum therapeutic potential of cannabidiol in psychiatric disorders. Philos Trans R Soc Lond B Biol Sci. 2012;367(1607):3364–78. doi: 10.1098/rstb.2011.0389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Ren Y, Whittard J, Higuera-Matas A, Morris CV, Hurd YL. Cannabidiol, a nonpsychotropic component of cannabis, inhibits cue-induced heroin seeking and normalizes discrete mesolimbic neuronal disturbances. J Neurosci. 2009;29(47):14764–9. doi: 10.1523/JNEUROSCI.4291-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Viudez-Martinez A, Garcia-Gutierrez MS, Fraguas-Sanchez AI, Torres-Suarez AI, Manzanares J. Effects of cannabidiol plus naltrexone on motivation and ethanol consumption. Br J Pharmacol. 2018. [DOI] [PMC free article] [PubMed]
- 38.Katsidoni V, Anagnostou I, Panagis G. Cannabidiol inhibits the reward-facilitating effect of morphine: involvement of 5-HT1A receptors in the dorsal raphe nucleus. Addict Biol. 2013;18(2):286–96. doi: 10.1111/j.1369-1600.2012.00483.x. [DOI] [PubMed] [Google Scholar]
- 39.Fang Y, Ronnekleiv OK. Cocaine upregulates the dopamine transporter in fetal rhesus monkey brain. J Neurosci. 1999;19(20):8966–78. doi: 10.1523/JNEUROSCI.19-20-08966.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Daws LC, Callaghan PD, Moron JA, Kahlig KM, Shippenberg TS, Javitch JA, et al. Cocaine increases dopamine uptake and cell surface expression of dopamine transporters. Biochem Biophys Res Commun. 2002;290(5):1545–50. doi: 10.1006/bbrc.2002.6384. [DOI] [PubMed] [Google Scholar]
- 41.Mash DC, Pablo J, Ouyang Q, Hearn WL, Izenwasser S. Dopamine transport function is elevated in cocaine users. J Neurochem. 2002;81(2):292–300. doi: 10.1046/j.1471-4159.2002.00820.x. [DOI] [PubMed] [Google Scholar]
- 42.Kahlig KM, Galli A. Regulation of dopamine transporter function and plasma membrane expression by dopamine, amphetamine, and cocaine. Eur J Pharmacol. 2003;479(1-3):153–8. doi: 10.1016/j.ejphar.2003.08.065. [DOI] [PubMed] [Google Scholar]
- 43.Pandolfo P, Silveirinha V, dos Santos-Rodrigues A, Venance L, Ledent C, Takahashi RN, et al. Cannabinoids inhibit the synaptic uptake of adenosine and dopamine in the rat and mouse striatum. Eur J Pharmacol. 2011;655(1-3):38–45. doi: 10.1016/j.ejphar.2011.01.013. [DOI] [PubMed] [Google Scholar]
- 44.Murillo-Rodriguez E, Palomero-Rivero M, Millan-Aldaco D, Mechoulam R, Drucker-Colin R. Effects on sleep and dopamine levels of microdialysis perfusion of cannabidiol into the lateral hypothalamus of rats. Life Sci. 2011;88(11-12):504–11. doi: 10.1016/j.lfs.2011.01.013. [DOI] [PubMed] [Google Scholar]
- 45.Renard J, Norris C, Rushlow W, Laviolette SR. Neuronal and molecular effects of cannabidiol on the mesolimbic dopamine system: Implications for novel schizophrenia treatments. Neurosci Biobehav Rev. 2017;75:157–65. doi: 10.1016/j.neubiorev.2017.02.006. [DOI] [PubMed] [Google Scholar]
- 46.Galaj E, Bi GH, Yang HJ, Xi ZX. Cannabidiol attenuates the rewarding effects of cocaine in rats by CB2, 5-HT1A and TRPV1 receptor mechanisms. Neuropharmacology. 2020;167:107740. doi: 10.1016/j.neuropharm.2019.107740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Arnold JC. The role of endocannabinoid transmission in cocaine addiction. Pharmacol Biochem Behav. 2005;81(2):396–406. doi: 10.1016/j.pbb.2005.02.015. [DOI] [PubMed] [Google Scholar]
- 48.Gobira PH, Oliveira AC, Gomes JS, da Silveira VT, Asth L, Bastos JR, et al. Opposing roles of CB1 and CB2 cannabinoid receptors in the stimulant and rewarding effects of cocaine. Br J Pharmacol. 2019;176(10):1541–51. doi: 10.1111/bph.14473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Aracil-Fernandez A, Trigo JM, Garcia-Gutierrez MS, Ortega-Alvaro A, Ternianov A, Navarro D, et al. Decreased cocaine motor sensitization and self-administration in mice overexpressing cannabinoid CB(2) receptors. Neuropsychopharmacology. 2012;37(7):1749–63. doi: 10.1038/npp.2012.22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Alvaro-Bartolome M, Garcia-Sevilla JA. Dysregulation of cannabinoid CB1 receptor and associated signaling networks in brains of cocaine addicts and cocaine-treated rodents. Neuroscience. 2013;247:294–308. doi: 10.1016/j.neuroscience.2013.05.035. [DOI] [PubMed] [Google Scholar]
- 51.Bystrowska B, Frankowska M, Smaga I, Pomierny-Chamiolo L, Filip M. Effects of Cocaine Self-Administration and Its Extinction on the Rat Brain Cannabinoid CB1 and CB2 Receptors. Neurotox Res. 2018. [DOI] [PMC free article] [PubMed]
- 52.Li X, Peng XQ, Jordan CJ, Li J, Bi GH, He Y, et al. mGluR5 antagonism inhibits cocaine reinforcement and relapse by elevation of extracellular glutamate in the nucleus accumbens via a CB1 receptor mechanism. Sci Rep. 2018;8(1):3686. doi: 10.1038/s41598-018-22087-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Xi ZX, Peng XQ, Li X, Song R, Zhang HY, Liu QR, et al. Brain cannabinoid CB(2) receptors modulate cocaine's actions in mice. Nat Neurosci. 2011;14(9):1160–6. doi: 10.1038/nn.2874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Vlachou S, Nomikos GG, Panagis G. WIN 55,212-2 decreases the reinforcing actions of cocaine through CB1 cannabinoid receptor stimulation. Behav Brain Res. 2003;141(2):215–22. doi: 10.1016/s0166-4328(02)00370-4. [DOI] [PubMed] [Google Scholar]
- 55.Soria G, Mendizabal V, Tourino C, Robledo P, Ledent C, Parmentier M, et al. Lack of CB1 cannabinoid receptor impairs cocaine self-administration. Neuropsychopharmacology. 2005;30(9):1670–80. doi: 10.1038/sj.npp.1300707. [DOI] [PubMed] [Google Scholar]
- 56.Liu QR, Canseco-Alba A, Zhang HY, Tagliaferro P, Chung M, Dennis E, et al. Cannabinoid type 2 receptors in dopamine neurons inhibits psychomotor behaviors, alters anxiety, depression and alcohol preference. Sci Rep. 2017;7(1):17410. doi: 10.1038/s41598-017-17796-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lopes JB, Bastos JR, Costa RB, Aguiar DC, Moreira FA. The roles of cannabinoid CB1 and CB2 receptors in cocaine-induced behavioral sensitization and conditioned place preference in mice. Psychopharmacology (Berl). 2020;237(2):385–94. doi: 10.1007/s00213-019-05370-5. [DOI] [PubMed] [Google Scholar]
- 58.Lujan MA, Cantacorps L, Valverde O. The pharmacological reduction of hippocampal neurogenesis attenuates the protective effects of cannabidiol on cocaine voluntary intake. Addict Biol. 2019:e12778. [DOI] [PubMed]
- 59.Bisogno T, Hanus L, De Petrocellis L, Tchilibon S, Ponde DE, Brandi I, et al. Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br J Pharmacol. 2001;134(4):845–52. doi: 10.1038/sj.bjp.0704327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Laprairie RB, Bagher AM, Kelly ME, Denovan-Wright EM. Cannabidiol is a negative allosteric modulator of the cannabinoid CB1 receptor. Br J Pharmacol. 2015;172(20):4790–805. doi: 10.1111/bph.13250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chung H, Fierro A, Pessoa-Mahana CD. Cannabidiol binding and negative allosteric modulation at the cannabinoid type 1 receptor in the presence of delta-9-tetrahydrocannabinol: An In Silico study. PLoS One. 2019;14(7):e0220025. doi: 10.1371/journal.pone.0220025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Thomas A, Baillie GL, Phillips AM, Razdan RK, Ross RA, Pertwee RG. Cannabidiol displays unexpectedly high potency as an antagonist of CB1 and CB2 receptor agonists in vitro. Br J Pharmacol. 2007;150(5):613–23. doi: 10.1038/sj.bjp.0707133. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PDF 1196 kb)