Abstract
Background
Therapist‐delivered trauma‐focused psychological therapies are effective for post‐traumatic stress disorder (PTSD) and have become the accepted first‐line treatments. Despite the established evidence‐base for these therapies, they are not always widely available or accessible. Many barriers limit treatment uptake, such as the number of qualified therapists available to deliver the interventions; cost; and compliance issues, such as time off work, childcare, and transportation, associated with the need to attend weekly appointments. Delivering Internet‐based cognitive and behavioural therapy (I‐C/BT) is an effective and acceptable alternative to therapist‐delivered treatments for anxiety and depression.
Objectives
To assess the effects of I‐C/BT for PTSD in adults.
Search methods
We searched MEDLINE, Embase, PsycINFO and the Cochrane Central Register of Controlled Trials to June 2020. We also searched online clinical trial registries and reference lists of included studies and contacted the authors of included studies and other researchers in the field to identify additional and ongoing studies.
Selection criteria
We searched for RCTs of I‐C/BT compared to face‐to‐face or Internet‐based psychological treatment, psychoeducation, wait list, or care as usual. We included studies of adults (aged over 16 years), in which at least 70% of the participants met the diagnostic criteria for PTSD, according to the Diagnostic and Statistical Manual (DSM) or the International Classification of Diseases (ICD).
Data collection and analysis
Two review authors independently assessed abstracts, extracted data, and entered data into Review Manager 5. The primary outcomes were severity of PTSD symptoms and dropouts. Secondary outcomes included diagnosis of PTSD after treatment, severity of depressive and anxiety symptoms, cost‐effectiveness, adverse events, treatment acceptability, and quality of life. We analysed categorical outcomes as risk ratios (RRs), and continuous outcomes as mean differences (MD) or standardised mean differences (SMDs), with 95% confidence intervals (CI). We pooled data using a fixed‐effect meta‐analysis, except where heterogeneity was present, in which case we used a random‐effects model. We independently assessed the included studies for risk of bias and we evaluated the certainty of available evidence using the GRADE approach; we discussed any conflicts with at least one other review author, with the aim of reaching a unanimous decision.
Main results
We included 13 studies with 808 participants. Ten studies compared I‐C/BT delivered with therapist guidance to a wait list control. Two studies compared guided I‐C/BT with I‐non‐C/BT. One study compared guided I‐C/BT with face‐to‐face non‐C/BT. There was substantial heterogeneity among the included studies.
I‐C/BT compared with face‐to‐face non‐CBT
Very low‐certainty evidence based on one small study suggested face‐to‐face non‐CBT may be more effective than I‐C/BT at reducing PTSD symptoms post‐treatment (MD 10.90, 95% CI 6.57 to 15.23; studies = 1, participants = 40). There may be no evidence of a difference in dropout rates between treatments (RR 2.49, 95% CI 0.91 to 6.77; studies = 1, participants = 40; very low‐certainty evidence). The study did not measure diagnosis of PTSD, severity of depressive or anxiety symptoms, cost‐effectiveness, or adverse events.
I‐C/BT compared with wait list
Very low‐certainty evidence showed that, compared with wait list, I‐C/BT may be associated with a clinically important reduction in PTSD post‐treatment (SMD –0.61, 95% CI –0.93 to –0.29; studies = 10, participants = 608). There may be no evidence of a difference in dropout rates between the I‐C/BT and wait list groups (RR 1.25, 95% CI 0.97 to 1.60; studies = 9, participants = 634; low‐certainty evidence). I‐C/BT may be no more effective than wait list at reducing the risk of a diagnosis of PTSD after treatment (RR 0.53, 95% CI 0.28 to 1.00; studies = 1, participants = 62; very low‐certainty evidence). I‐C/BT may be associated with a clinically important reduction in symptoms of depression post‐treatment (SMD –0.51, 95% CI –0.97 to –0.06; studies = 7, participants = 473; very low‐certainty evidence). Very low‐certainty evidence also suggested that I‐C/BT may be associated with a clinically important reduction in symptoms of anxiety post‐treatment (SMD –0.61, 95% CI –0.89 to –0.33; studies = 5, participants = 345). There were no data regarding cost‐effectiveness. Data regarding adverse events were uncertain, as only one study reported an absence of adverse events.
I‐C/BT compared with I‐non‐C/BT
There may be no evidence of a difference in PTSD symptoms post‐treatment between the I‐C/BT and I‐non‐C/BT groups (SMD –0.08, 95% CI –0.52 to 0.35; studies = 2, participants = 82; very low‐certainty evidence). There may be no evidence of a difference between dropout rates from the I‐C/BT and I‐non‐C/BT groups (RR 2.14, 95% CI 0.97 to 4.73; studies = 2, participants = 132; I² = 0%; very low‐certainty evidence). Two studies found no evidence of a difference in post‐treatment depressive symptoms between the I‐C/BT and I‐non‐C/BT groups (SMD –0.12, 95% CI –0.78 to 0.54; studies = 2, participants = 84; very low‐certainty evidence). Two studies found no evidence of a difference in post‐treatment symptoms of anxiety between the I‐C/BT and I‐non‐C/BT groups (SMD 0.08, 95% CI –0.78 to 0.95; studies = 2, participants = 74; very low‐certainty evidence). There were no data regarding cost‐effectiveness. Data regarding adverse effects were uncertain, as it was not discernible whether adverse effects reported were attributable to the intervention.
Authors' conclusions
While the review found some beneficial effects of I‐C/BT for PTSD, the certainty of the evidence was very low due to the small number of included trials. This review update found many planned and ongoing studies, which is encouraging since further work is required to establish non‐inferiority to current first‐line interventions, explore mechanisms of change, establish optimal levels of guidance, explore cost‐effectiveness, measure adverse events, and determine predictors of efficacy and dropout.
Keywords: Adolescent; Adult; Humans; Young Adult; Bias; Cognitive Behavioral Therapy; Cognitive Behavioral Therapy/methods; Internet-Based Intervention; Randomized Controlled Trials as Topic; Stress Disorders, Post-Traumatic; Stress Disorders, Post-Traumatic/therapy
Plain language summary
Internet‐based cognitive and behavioural therapies for post‐traumatic stress disorder (PTSD)
Why was this review important?
Post‐traumatic stress disorder, or PTSD, is a common mental illness that can occur after a serious traumatic event. Symptoms include re‐experiencing the trauma as nightmares, flashbacks, and distressing thoughts; avoiding reminders of the traumatic event; experiencing negative changes to thoughts and mood; and hyperarousal, which includes feeling on edge, being easily startled, feeling angry, having difficulties sleeping, and problems concentrating. PTSD can be treated effectively with talking therapies that focus on the trauma. Some of the most effective therapies are those based on cognitive behavioural therapy (CBT). Unfortunately, there are a limited number of qualified therapists who can deliver these therapies. There are also other factors that limit access to treatment, such as the need to take time off work to attend appointments, and transportation issues.
An alternative is to deliver psychological therapy on the Internet, with or without guidance from a therapist. Internet‐based cognitive and behavioural therapies (I‐C/BT) have received a great deal of attention and are now used routinely to treat depression and anxiety. There have been fewer studies of I‐C/BT for PTSD, yet research is expanding and there is a growing evidence base for their efficacy.
Who will be interested in this review?
– People with PTSD and their families and friends.
– Professionals working in mental health services.
– General practitioners.
– Commissioners.
What questions did this review try to answer?
In adults with PTSD, we tried to find out if I‐C/BT:
– was more effective than no therapy (wait list);
– was as effective as psychological therapies delivered by a therapist;
– was more effective than other psychological therapies delivered online; or
– was more effective than education about the condition delivered online, at reducing symptoms of PTSD, and improving quality of life; or
– was cost effective, compared to face‐to‐face therapy?
Which studies did the review include?
We searched for randomised controlled trials (clinical studies where people are randomly put into one of two or more treatment groups) that examined I‐C/BT for adults with PTSD, published between 1970 and 5 June 2020.
We included 13 studies with 808 participants.
What did the evidence from the review tell us?
– Analyses including 10 studies found that I‐C/BT was more effective than no therapy (waiting list), at reducing PTSD. However, the certainty of the evidence was very low, which means we have very little confidence in this finding.
– Analyses including two studies found there was no difference between I‐C/BT and another type of psychological therapy delivered online. However, the certainty of the evidence was very low, which means we have very little confidence in this finding.
– One study found that face‐to‐face non‐CBT was more effective than I‐C/BT. However, baseline levels of PTSD symptoms were not controlled for and the certainty of this evidence was very low, which limits our confidence in this finding.
– We found no studies using standardised or validated measures of acceptability to tell us whether people who received I‐C/BT felt it was an acceptable treatment.
– We found no studies that reported the cost‐effectiveness of I‐C/BT.
What should happen next?
The current evidence base is growing but still small. More studies are needed to decide if I‐C/BT should be used routinely for the treatment of PTSD.
Summary of findings
Summary of findings 1. Internet‐based cognitive and behavioural therapy (I‐C/BT) compared to face‐to‐face non‐CBT for post‐traumatic stress disorder (PTSD) in adults.
| I‐C/BT compared to face‐to‐face non‐CBT for PTSD in adults | ||||||
| Patient or population: adults aged ≥ 16 years with PTSD Setting: military Intervention: I‐C/BT Comparison: face‐to‐face non‐CBT | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with wait list | Risk with I‐C/BT | |||||
|
Severity of PTSD symptoms (measured using the IES‐R, CAPS‐5, PCL‐CPSS‐I, and PDS; higher score = worse outcome) Follow‐up: post‐treatment |
The mean severity of PTSD symptoms (post‐treatment) was 30 | MD 10.90 higher (6.57 higher to 15.23 higher) | — | 40 (1 RCT) | ⊕⊝⊝⊝ Very lowa,b | — |
| Dropouts | Study population |
RR 2.49 (0.91 to 6.77) |
40 (1 RCT) | ⊕⊝⊝⊝ Very lowa,b | — | |
| 190 per 1000 | 474 per 1000 (173 to 1000) |
|||||
| Diagnosis of PTSD after treatment | — | — | — | — | Not measured | |
|
Severity of depressive symptoms (measured with the BDI, PHQ and CES‐D; higher score = worse outcome) Follow‐up: post‐treatment |
— | — | — | — | Not measured | |
|
Severity of anxiety symptoms (measured with the BAI and GAD‐7; higher score = worse outcome) Follow‐up: post‐treatment |
— | — | — | — | Not measured | |
| Cost‐effectiveness | — | — | — | — | Not measured | |
| Adverse events | — | — | — | — | Not measured | |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). BAI: Beck Anxiety Inventory; BDI: Beck Depression Inventory; CAPS‐5: Clinician‐Administered PTSD Symptom Scale; CBT: cognitive behavioural therapy; CES‐D: Center for Epidemiologic Studies Depression Scale; CI: confidence interval; GAD‐7: Generalized Anxiety Disorder 7‐Item Scale; I‐C/BT: Internet‐based cognitive and behavioural therapy; IES‐R: Impact of Event Scale – Revised; MD: mean difference; PCL‐CPSS‐I: PTSD Checklist‐Child Posttraumatic Stress Scale – Interview for DSM‐5; PDS: Posttraumatic Diagnostic Scale; PHQ: Patient Health Questionnaire; PTSD: post‐traumatic stress disorder; RCT: randomised controlled trial; RR: risk ratio. | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
aDowngraded two levels due to high risk of performance, attrition, reporting, and other bias. bDowngraded one level for imprecision due to small sample size.
Summary of findings 2. Internet‐based cognitive and behavioural therapy (I‐C/BT) compared to wait list for post‐traumatic stress disorder (PTSD) in adults.
| I‐C/BT compared to wait list for PTSD in adults | ||||||
| Patient or population: adults aged ≥ 16 years with PTSD Setting: recruited through advertisements (including social media, newspapers, flyers, presentations at military defence sites) or referred through primary or secondary care Intervention: I‐C/BT Comparison: wait list | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with wait list | Risk with I‐C/BT | |||||
|
Severity of PTSD symptoms (measured with the IES‐R, CAPS‐5, PCL‐CPSS‐I, and PDS; higher score = worse outcome) Follow‐up: post‐treatment |
The mean severity of PTSD symptoms (post‐treatment) was 40 | SMD 0.61 lower (0.93 lower to 0.29 lower) | — | 608 (10 RCTs) | ⊕⊝⊝⊝ Very lowa,b | — |
| Dropouts | Study population | RR 1.25 (0.97 to 1.60) | 634 (9 RCTs) | ⊕⊕⊝⊝ Lowa | — | |
| 227 per 1000 | 283 per 1000 (220 to 362) | |||||
| Diagnosis of PTSD after treatment | Study population | RR 0.53 (0.28 to 1.00) | 62 (1 RCT) | ⊕⊝⊝⊝ Very lowc,d | — | |
| 548 per 1000 | 291 per 1000 (154 to 548) | |||||
|
Severity of depressive symptoms (measured with the BDI, PHQ and CES‐D; higher score = worse outcome) Follow‐up: post‐treatment |
The mean depression (post‐treatment) was 16 | SMD 0.51 lower (0.97 lower to 0.06 lower) | — | 473 (7 RCTs) | ⊕⊝⊝⊝ Very lowb,e | — |
|
Severity of anxiety symptoms (measured with the BAI and GAD‐7; higher score = worse outcome) Follow‐up: post‐treatment |
The mean anxiety (post‐treatment) was 14 | SMD 0.61 lower (0.89 lower to 0.33 lower) | — | 345 (5 RCTs) | ⊕⊝⊝⊝ Very lowf,g | — |
| Cost‐effectiveness | — | — | — | — | — | Not measured. |
| Adverse events | — | — | — | — | ⊕⊝⊝⊝ Very lowh,i | Lewis 2017 reported an absence of adverse events. Bragesjö 2020 reported adverse events in their study overall; however, we were unable to draw conclusions with respect to the subset of data that was included. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). BAI: Beck Anxiety Inventory; BDI: Beck Depression Inventory; CAPS‐5: Clinician‐Administered PTSD Symptom Scale; CES‐D: Center for Epidemiologic Studies Depression Scale; CI: confidence interval; GAD‐7: Generalized Anxiety Disorder 7‐Item Scale; I‐C/BT: Internet‐based cognitive and behavioural therapy; IES‐R: Impact of Event Scale – Revised; PCL‐CPSS‐I: PTSD Checklist‐Child Posttraumatic Stress Scale – Interview for DSM‐5; PDS: Posttraumatic Diagnostic Scale; PHQ: Patient Health Questionnaire; PTSD: post‐traumatic stress disorder; RCT: randomised controlled trial; RR: risk ratio; SMD: standardised mean difference. | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
aDowngraded two levels due to high risk of performance bias in all 10 studies, high risk of attrition bias in three studies (Allen 2020; Knaevelsrud 2015; Krupnick 2017), and high risk of other bias in five studies (Allen 2020; Bragesjö 2020; Ivarsson 2014; Krupnick 2017; Lewis 2017). bDowngraded one level for inconsistency; high levels of heterogeneity. cDowngraded one level for imprecision due to small sample size and the confidence interval around the effect estimate included both little or no effect. dDowngraded two levels due high risk of performance bias and other bias (Ivarsson 2014). eDowngraded two levels due to high risk of performance bias in all seven studies, high risk of attrition bias in two studies (Allen 2020; Krupnick 2017), and high risk of other bias in four studies (Allen 2020; Bragesjö 2020; Krupnick 2017; Lewis 2017). fDowngraded one level for imprecision due to small sample size. gDowngraded two levels due to high risk of performance bias in all five studies, high risk of attrition bias in two studies (Allen 2020; Knaevelsrud 2015), and high risk of other bias in three studies (Allen 2020; Lewis 2017; Spence 2011). hDowngraded two levels due to high risk of performance bias and other bias in both studies (Bragesjö 2020; Lewis 2017). iDowngraded one level due to small sample size in both studies (Bragesjö 2020: Lewis 2017).
Summary of findings 3. Internet‐based cognitive and behavioural therapy (I‐C/BT) compared to I‐non‐C/BT for post‐traumatic stress disorder (PTSD) in adults.
| I‐C/BT compared to I‐non‐C/BT for PTSD in adults | ||||||
| Patient or population: adults aged ≥ 16 years with PTSD Setting: recruited through advertisements at higher education settings or at military defence sites Intervention: I‐C/BT Comparison: I‐non‐C/BT | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with I‐non‐C/BT | Risk with I‐C/BT | |||||
|
Severity of PTSD symptoms (measured with the IES‐R, CAPS‐5, PCL‐CPSS‐I and PDS; higher score = worse outcome) Follow‐up: post‐treatment |
The mean severity of PTSD symptoms (post‐treatment) was 15 | SMD 0.08 lower (0.52 lower to 0.35 higher) | — | 82 (2 RCTs) | ⊕⊝⊝⊝ Very lowa,b | — |
| Dropouts | Study population | RR 2.14 (0.97 to 4.73) | 132 (2 RCTs) | ⊕⊝⊝⊝ Very lowa,b | — | |
| 113 per 1000 | 242 per 1000 (110 to 534) | |||||
| Diagnosis of PTSD after treatment | — | — | — | — | — | — |
|
Severity of depressive symptoms (measured with the BDI, PHQ and CED‐D; higher score = worse outcome) Follow‐up: post‐treatment |
The mean depression (post‐treatment) was 15 | SMD 0.12 lower (0.78 lower to 0.54 higher) | — | 84 (2 RCTs) | ⊕⊝⊝⊝ Very lowa,b | — |
|
Severity of anxiety symptoms (measured with the BAI and GAD‐7; higher score = worse outcome) Follow‐up: post‐treatment |
The mean anxiety (post‐treatment) was 35.6 | SMD 0.08 higher (0.78 lower to 0.95 higher) | — | 74 (2 RCTs) | ⊕⊝⊝⊝ Very lowa,b,c | — |
| Cost‐effectiveness | — | — | — | — | — | Not measured. |
| Adverse events | — | — | — | — | ⊕⊝⊝⊝ Very lowd,e | Littleton 2016 noted 2 participants in the intervention condition reported a clinically significant increase in depression symptoms post‐treatment, with 1 of these participants also reporting a clinically significant increase in anxiety symptoms. However, it was difficult to attribute this to I‐C/BT as these participants had both experienced the death of an immediate family member during treatment. 3 additional participants reported clinically significant increases in anxiety at post‐treatment. With respect to the control condition 1 participant experienced a clinically significant increase in depression symptoms between post‐treatment and follow‐up. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). BAI: Beck Anxiety Inventory; BDI: Beck Depression Inventory; CAPS‐5: Clinician‐Administered PTSD Symptom Scale; CES‐D: Center for Epidemiologic Studies Depression Scale; CI: confidence interval; GAD‐7: Generalized Anxiety Disorder 7‐Item Scale; I‐C/BT: Internet‐based cognitive and behavioural therapy; IES‐R: Impact of Event Scale; PCL‐CPSS‐I: PTSD Checklist‐Child Posttraumatic Stress Scale – Interview for DSM‐5; PDS: Posttraumatic Diagnostic Scale; PHQ: Patient Health Questionnaire; PTSD: post‐traumatic stress disorder; RCT: randomised controlled trial; RR: risk ratio; SMD: standardised mean difference. | ||||||
| GRADE Working Group grades of evidence High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
aDowngraded two levels due to high risk of performance bias due to lack of blinding participants and personnel in both studies (Littleton 2016; Litz 2007), high risk of detection bias due to lack of blinding outcome assessors in one study (Littleton 2016), and high risk of attrition bias and other bias in one study (Litz 2007). bDowngraded two levels for imprecision due to small sample size and the confidence interval of the effect estimate included both little or no effect. cDowngraded one level for inconsistency due to high levels of heterogeneity. dDowngraded two levels due to high risk of performance bias due to lack of blinding participants and personnel and high risk of detection bias due to lack of blinding outcome assessors in one study (Littleton 2016). eDowngraded one level for imprecision due to small sample size.
Background
Description of the condition
Post‐traumatic stress disorder (PTSD) is characterised by the development of distressing psychological symptoms following exposure to a traumatic event (APA 2013). Exposure can be actual or threatened death, serious injury, or sexual violation either through direct personal experience or witnessing an "extremely threatening or horrific event or series of events" (WHO 2018). PTSD does not represent a valid diagnosis after life‐events such as divorce or losing a job.
Diagnostic symptoms of PTSD include re‐experiencing the trauma as intrusive memories, nightmares, or flashbacks; avoiding thoughts about the trauma or reminders of it; negative alterations in mood or cognitions, including strong negative feelings such as guilt and shame and constricted affect; and heightened physiological arousal, which can manifest as hypervigilance, exaggerated startle responses, and difficulties concentrating or sleeping. Diagnosis is dependent on symptoms that cause clinically significant distress and impairment to the person's capacity to work, socialise, or function in other important domains (APA 2013).
PTSD is a common global mental health disorder that imposes a significant personal and societal burden. Epidemiological research suggests that the lifetime exposure to one or more traumatic events in the general population is 70% (Kessler 2017). Caution must be exercised when considering PTSD prevalence reporting across the literature due to the variable methodological approaches in the field, including diagnostic criteria and assessment tools. Lifetime prevalence of PTSD has been estimated at 1.3% in Japan (Kawakami 2014), 2.2% in Spain (Olaya 2015), 2.3% in South Africa (Atwoli 2013), 8.8% in Northern Ireland (Ferry 2014), 11.2% in Mexico (Norris 2003), and one large‐scale survey in Australia reported a 12‐month prevalence of 1.33% (Creamer 2001). Higher prevalence is apparent in high‐risk professional groups, such as military service members and first responders (Sareen 2013; Wilson 2015), and has been shown to double in populations affected by conflict (Steel 2009). Research has shown that approximately one third of people with PTSD at four to six weeks' post‐trauma exposure are found to naturally remit by three months (Santiago 2013). Many people are exposed to trauma at some point in their lives, yet only a small proportion will develop PTSD, and for many difficulties are subclinical, and most will recover to pretrauma levels of psychological functioning (Bonanno 2015; Giummarra 2018). Research supporting this demonstrates trajectories of the course of PTSD, including findings for a resilient class of people presenting with few PTSD symptoms, as well as a recovery class of people presenting with initial distress followed by gradual remission, among other classes with higher PTSD levels (Bryant 2015).
There is substantial comorbidity between PTSD and other psychiatric disorders. Two commonly cited epidemiological studies, conducted in Australia and the USA, demonstrated high levels of PTSD comorbidity, up to 88% in men and 80% in women, with around 50% experiencing three or more comorbidities. PTSD was often primary to substance use and affective disorders and in half of cases to be primary to anxiety disorders (Creamer 2001; Kessler 1995). Common comorbidities include depression (Rytwinski 2013), panic disorder (Brown 2001), borderline personality disorder (Cattane 2017), substance‐use disorders (Pietrzak 2012), and chronic pain (Siqveland 2017). One UK survey found that people with PTSD were twice as likely as people without PTSD to have at least one other comorbid mood, anxiety, or substance‐use disorder (Ferry 2008).
There are many additional negative potential consequences of PTSD. These include physical health problems, impaired functioning, including social functioning, and maladaptive coping mechanisms (Rauch 2009; Roberts 2016). In addition, there is significant economic burden of PTSD on society, for example a PTSD cost of GBP 172,756,062 was reported in Northern Ireland in 2008 for reasons that included high rates of unemployment due to symptomatology impacting job loss (Ferry 2015). Unfortunately, people with PTSD have 13 times the rate of suicide death than those without PTSD, following adjustment for depression, anxiety, and substance abuse diagnoses (Gradus 2015).
Numerous risk factors for the development of PTSD have been proposed and contested in the literature. These include pretrauma factors, such as genetic variation, psychiatric history and traumatic events; and peritrauma factors, such as trauma severity and dissociation; and post‐trauma factors, such as poor social support (Brewin 2000; Ozer 2003). There is evidence for genetic variation underlying individual differences in risk and resilience to PTSD (Duncan 2018a; Duncan 2018b). There is a higher prevalence of PTSD in women than in men. This is incongruent to the finding that there are gender differences in trauma exposure, with female sex associated with reduced risk of traumatic event exposure, overall (Carmassi 2014; Ferry 2014), though may be explained by the finding that women disproportionately experience the trauma types associated with higher PTSD risk (Pietrzak 2011). Peritraumatic factors have been associated with PTSD risk and resilience. Peritraumatic dissociation (PD), a subjective change in cognitive perception and functioning, feelings of emotional numbness, reduced awareness of surroundings, and derealisation around the time of the traumatic event (APA 1994), is a moderate risk factor for PTSD (Breh 2007). An individual's locus of control, or the extent to which an individual believes they can control events that affect them, is associated with PTSD, where an individual's belief that they have control over their life was a protective factor of resilience against PTSD symptoms (Karstoft 2015). Research has demonstrated that low social support during/after trauma exposure, along with other peritrauma risk factors, such as trauma severity and additional life stresses, has a somewhat stronger effect than pretrauma factors such as demographics (Brewin 2000; Ozer 2003).
Description of the intervention
Guidelines for the prevention and treatment of PTSD developed by the International Society for Traumatic Stress Studies (ISTSS) are based on the most up‐to‐date empirical evidence (ISTSS 2018). These guidelines have developed through rigorous methodology with 361 included randomised controlled trials (RCT) reviewed systematically, and 208 meta‐analyses conducted (Bisson 2019). Strong evidence (at least reasonable quality of evidence and the highest certainty of effect) is available for the use of trauma‐focused psychological treatment: trauma‐focused cognitive behavioural therapy (CBT), either as a category in itself, or the specific interventions of cognitive processing therapy (CPT) (Resick 1996), cognitive therapy (CT) for PTSD (Ehlers 2000), and prolonged exposure (PE) (Foa 1998); and eye movement desensitisation and reprocessing (EMDR) (Shapiro 1989). These ISTSS recommendations converge with the recommendations of four other major guidelines for the treatment of PTSD (ACPMH 2007; APA 2017; Department of Veteran Affairs 2017; NICE 2018). Pharmacological treatments, for example, fluoxetine and paroxetine, are recommended to a low effect level of evidence (at least reasonable quality of evidence and high certainty of a low level of effect). Other Cochrane Reviews have considered combined pharmacotherapy and psychological therapies (Hetrick 2010), and psychological therapies to treat PTSD in children and adolescents (Gillies 2012).
Trauma‐focused psychological therapies are currently the treatments of choice for people with PTSD; however, a shortage of suitably qualified and trained therapists who can deliver these interventions has prompted interest in new interventions that place less reliance on therapist time (Lewis 2013).
There has been a rapid increase in the number of Internet‐based psychological therapies since the late 1990s and early 2000s, helped by mobile phone and Web 2.0 applications, websites, and applications that allow anyone to create and share online information or material they have created (Andersson 2018; Gibbons 2011). Internet‐based approaches offer potential as an adjunct to usual care or a cost‐effective alternative to conventional in‐person face‐to‐face treatment, which has prompted a growing interest in such interventions that place less reliance on therapist time (Lewis 2013).
Individual‐based protocols have been adapted into Internet‐based materials, for use with or without guidance, for several mental health disorders (Andersson 2016). Internet‐based cognitive and behavioural therapy (I‐C/BT) commonly includes a series of modules, often interactive audio and video materials, with agreed homework tasks. For instance, 'Beating the Blues' (BtB), an early example of a computerised CBT (c‐CBT) intervention for depression, includes interactive, multimedia techniques including video vignettes of case study patients, and homework exercises (Proudfoot 2003). More recently in the case of Internet CBT (i‐CBT), modules are typically delivered via website browser or mobile applications (Ebert 2018). Therapists provide sufficient instruction to teach coping skills or bring about improvement in target symptoms with limited therapist input (Spek 2007). Flexible I‐C/BT approaches may be more accessible for people who are unable to commit to in‐person sessions, and may be necessary for all individuals at times, as has been demonstrated during the COVID‐19 pandemic, where social distance measures were in place in many countries across the globe (Békés 2020; Shore 2020). The opportunity for digital therapies to widen access to evidence‐based psychological care is a priority for national and international research, policy, and commissioning (Torous 2019). I‐C/BT programmes have been developed and implemented for a range of disorders, with the aim of reducing healthcare expenditure and broadening access to psychological therapies (Lewis 2010). The content of existing therapies is not usually greatly altered, deviating from traditional psychological treatment mainly in terms of method of delivery (Cuijpers 2010).
The distinction between I‐C/BT and online psychoeducation must be clear. Although the two overlap in content, psychoeducation aims to increase patient knowledge, while I‐C/BT aims to teach skills and techniques that can be used to overcome specific symptoms (IAPT 2010). I‐C/BT programmes are usually based on existing protocols and share many common features (Andersson 2005). Most start treatment with psychoeducation, and then present the rationale for CBT‐based treatment (IAPT 2010). These programmes incorporate cognitive techniques with the aim of identifying and modifying unhelpful patterns of cognition (Newman 2003). Usually, behavioural components are included; for PTSD, they generally encompass imaginal exposure (which creates a narrative of the trauma memory and engages in repeated exposure to it), and in vivo exposure (which involves gradual, repeated exposure to feared or avoided situations; Lewis 2012). Most Internet‐based self‐help programmes conclude with a section on relapse prevention that focuses on staying well, recognising signs of relapse, and offering advice on what to do if problems recur (Gega 2004).
Many different types of I‐C/BT have been developed. They can be distinguished based on the type and level of therapist assistance provided, which can vary widely between interventions. Typically the purpose of guidance is in providing support, including recognising and reinforcing an individual's engagement with the self‐help materials, for example through weekly feedback (Berger 2017), in the form of email, text, telephone, video meetings, or in‐person face‐to‐face sessions. A combination of I‐C/BT and face‐to‐face sessions is also known as 'blended treatment'. Guidance may be from a highly engaged specialist or from a non‐specialist mental health professional. I‐C/BT can also adopt a pure self‐help approach, in which the participant is the sole agent of change, and no therapist assistance is provided.
How the intervention might work
Internet‐based psychological interventions may take a CT or a behavioural therapy (BT) approach, but most are based on CBT (Andersson 2009). The core premise of CBT for PTSD is that fear conditioning and maladaptive cognitions contribute to emotional distress and problematic behaviours. Several disorder‐specific CBT protocols have been developed, and the approach involves a collaborative problem‐solving process, aimed at exploring and challenging unhelpful cognitions, and modifying problematic behaviours. Many I‐C/BT programmes for PTSD have taken a trauma‐focused CBT approach, which relies on general cognitive and behavioural techniques, with additional components aimed at addressing problematic thoughts and behaviours arising from the traumatic event itself.
Face‐to‐face trauma‐focused CBT is evidence based, and protocols draw on four core components: psychoeducation, anxiety management, exposure, and cognitive restructuring (Bisson 2013). These components are incorporated into Internet adaptations of the intervention (Lange 2003). Psychoeducation provides information about the disorder, symptoms, and treatment, and anxiety management techniques strengthen the individual's ability to cope with PTSD symptoms, recollection of traumatic memories, and the therapeutic process. Anxiety management may include breathing techniques, progressive muscle relaxation, or forms of guided imagery. Exposure plays an important role in many trauma‐focused CBT protocols and may be carried out in vivo (real life) or imaginally (Bryant 2003). It is common for both techniques to be used in the treatment of people with PTSD, to target internally and externally feared stimuli (Creamer 2004). The trauma memory itself is often the primary feared stimulus, and exposure to the memory is carried out imaginally. The rationale for the use of imaginal exposure varies according to the specific trauma‐focused CBT protocol applied. Imaginal exposure is based on principles of habituation (reduction of anxiety after PE), information processing (re‐evaluation of old information and incorporation of new information into the trauma memory), or both (Foa 2008). In vivo exposure encourages the person to confront feared situations in real life, and cognitive work seeks to identify and modify unhelpful thoughts by testing and challenging self‐held beliefs (Foa 2007; Wilson 2012).
Why it is important to do this review
PTSD causes clinically significant distress and impacts functioning (APA 2013), and effective interventions are required.
Internet‐based psychological therapies are becoming increasingly popular, but their use for the treatment of people with PTSD has lagged behind that for other disorders (Lewis 2012). I‐C/BT for depression and anxiety have received significant attention, and numerous systematic reviews and meta‐analyses have explored the efficacy of these interventions (e.g. Andersson 2019; Carlbring 2017; Karyotaki 2017; Olthius 2016). Positive findings, and the potential for these Internet‐based treatments to broaden access to psychological therapy and reduce costs, have spurred on the development and evaluation of similar interventions for a wide range of mental health problems (Carlbring 2017). A proliferation of studies evaluating the efficacy of I‐C/BT for PTSD led to the original Cochrane Review on this topic in 2018 (Lewis 2018). These findings were drawn upon for the development of ISTSS guidelines for the prevention and treatment of PTSD (ISTSS 2018). ISTSS have given a standard evidence level (at least reasonable quality of evidence and lower certainty of effect), to multiple session Internet‐based trauma‐focused CBT‐guided self‐help (GSH), as well as multiple session CBT without a trauma‐focus and narrative exposure therapy. Interventions to treat PTSD continue to develop, including interventions that are delivered via the Internet, both GSH and non‐guided, or stand‐alone, therapies. As research into I‐C/BT for PTSD continues to proliferate, a review of the evidence is required to understand the efficacy of interventions delivered through this modality. Furthermore, an increase in the utilisation of digital health due to the COVID‐19 pandemic deems this a good time to conduct an update on this topic (Wind 2020).
Two Cochrane Reviews are related to the current work, including the review of psychological therapies for PTSD, which did not include I‐C/BT treatments (Bisson 2013), and a review of media‐delivered CBT for anxiety disorders in adults, which excluded PTSD, as it was set to be separated from anxiety disorders in the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM‐5), and the International Classification of Diseases, 11th Edition (ICD‐11; Mayo‐Wilson 2013). A third Cochrane Review of I‐C/BT for anxiety disorders included PTSD as an eligible diagnosis, but excluded interventions provided without therapist assistance, and interventions that included face‐to‐face therapist assistance; the stringent inclusion criteria resulted in inclusion of only one trial of I‐C/BT for PTSD (Olthuis 2015). Therefore, there still is the need to summarise the evidence base for I‐C/BT, which is unguided, or uses minimal face‐to‐face guidance.
Objectives
To assess the effects of I‐C/BT for PTSD in adults.
Methods
Criteria for considering studies for this review
Types of studies
RCTs, randomised crossover trials, and cluster‐randomised trials. For crossover trials, we only used data from the first randomisation period to avoid a carry‐over effect. We did not use sample size or publication status to determine whether a study should be included. We included studies published in all languages.
Types of participants
Characteristics
Adults, 16 years of age or older. We did not consider I‐C/BT interventions for children under the age of 16 years for this review. We applied no restrictions on gender or ethnicity.
Diagnosis
Participants had traumatic stress symptoms, and at least 70% of people in any given study were required to meet diagnostic criteria for PTSD according to the Diagnostic and Statistical Manual of Mental Disorders (DSM‐III; APA 1980), DSM‐IIIR (APA 1987), DSM‐IV (APA 2000), DSM‐5 (APA 2013), International Statistical Classification of Diseases and Related Health Problems (ICD‐9; WHO 1979), or ICD‐10 (WHO 1992), assessed by clinical interview or score above a pre‐established threshold on a validated traumatic stress questionnaire. We defined trauma as exposure to a DSM‐qualifying traumatic event. We included studies regardless of the index trauma, severity of symptoms, duration of symptoms, or length of time since trauma. We included studies of participants with PTSD as a comorbid disorder, as long as reduction in PTSD symptoms was the primary aim of the intervention.
Comorbidities
We applied no restrictions on the basis of comorbidity.
Setting
We applied no restrictions on the basis of setting.
Types of interventions
Experimental interventions
We included individual I‐C/BT interventions for the treatment of people with PTSD (with or without therapist guidance), including those delivered online and through applications. We included programmes based on CT, BT, or CBT. These terms were defined as follows.
Interventions based on CT had to be based on established cognitive behavioural models of treatment built around the concept that the way that we think affects how we feel emotionally and how we seek to behave.
Interventions based on BT had to change behaviours associated with unhelpful cognitions or fear conditioning. This might have included exposure‐based work.
Interventions based on CBT must have included a combination of components based on CT and BT.
We drew a distinction between I‐C/BT and online psychoeducation and did not include online psychoeducation.
To be classified as I‐C/BT, programmes had to be delivered via a computer or a mobile device. We included programmes that provided a maximum of five hours of therapist guidance, delivered face‐to‐face or remotely (e.g. telephone, email, instant messaging). We applied no restrictions based on the number of interactions with a therapist, or the length of the online programme.
We excluded interventions based on EMDR and interventions using mindfulness‐based approaches, apart from mindfulness‐based I‐C/BT.
Comparator interventions
Face‐to‐face psychological therapy (CBT‐based).
Face‐to‐face psychological therapy (non‐CBT‐based), categorised EMDR and other therapies (i.e. supportive therapy, non‐directive counselling, psychodynamic therapy, and present‐centred therapy (PCT)) in line with the Cochrane Review of psychological therapies for adults with chronic PTSD (Bisson 2013).
Wait list, repeated assessment, or usual care.
Internet psychoeducation.
Internet psychological therapy (non‐CBT).
Types of outcome measures
We included studies that met the above inclusion criteria, regardless of whether they reported the following outcomes.
Primary outcomes
Severity of PTSD symptoms (measured using a standardised scale, such as the Clinician‐Administered PTSD Symptom Scale (CAPS‐5; Blake 1995), or the PTSD Checklist (PCL‐5; Weathers 2013). When a study reported both a clinician‐administered scale and a self‐report measure, we used the clinician‐administered measure in the meta‐analysis).
Dropouts (measured by the number of participants still receiving treatment at the end of the intervention).
Secondary outcomes
Diagnosis of PTSD after treatment (number of participants who met diagnostic criteria for PTSD in each arm of the study).
Severity of depressive symptoms (using a standardised scale, e.g. Beck Depression Inventory (BDI; Beck 1961)).
Severity of anxiety symptoms (using a standardised scale, e.g. Beck Anxiety Inventory (BAI; Beck 1993)).
Cost‐effectiveness (any measures of cost‐effectiveness).
Adverse events (e.g. symptom worsening (taking into account the measurement error of the instrument), relapses to substance use, hospitalisations, suicide attempts, and work absenteeism).
Standardised measures of treatment acceptability.
Quality of life (any measures of quality of life).
Timing of outcome assessment
We grouped outcome measures according to length of follow‐up as follows.
Immediately post‐treatment.
Follow‐up less than six months' post‐treatment.
Follow‐up between six months' and one year' post‐treatment.
Follow‐up longer than one year' post‐treatment.
Our primary outcome point was immediately post‐treatment.
Hierarchy of outcome measures
We planned to produce hierarchies of standardised measures based on their frequency of use within included studies, so that when a trial reported data from two or more measures of the same outcome, we would only use data from the measure ranked highest. This was only required for one included study (Ivarsson 2014), where the PDS and IES‐R measures were used to measure PTSD symptoms. Given the PDS measure was used in other included studies, whereas the IES‐R was not used in other included studies, only the PDS data were used.
Search methods for identification of studies
Electronic searches
The searches are an update of those conducted to March 2018 for the previous published version of this review (Lewis 2018). The update search was conducted on 5 June 2020, on the following databases (search strategies in Appendix 1):
Cochrane Common Mental Disorders Group's Specialised Register (CCMDCTR) (May/June 2016 only) (Appendix 2).
Cochrane Central Register of Controlled Trials (CENTRAL; 2020, Issue 6); (searched 1 March 2018 to 5 June 2020);
Ovid MEDLINE (2018 to 4 June 2020);
Ovid Embase (2018 to week 22 2020);
Ovid PsycINFO (2018 to 4 June 2020);
Proquest PTSDpubs (previously PILOTS) (1 March 2018 to 5 June 2020);
Proquest Dissertations & Theses (1 March 2018 to 5 June 2020);
We searched international trial registries via the ClinicalTrials.gov to identify unpublished or ongoing studies (to June 2020). The World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) was unavailable at this time, due to heavy usage generated by the COVID‐19 outbreak (apps.who.int/trialsearch/). However, records from this register are periodically added to CENTRAL on the Cochrane Library and some ICTRP records will have been captured there.
We did not restrict the update search by language or publication status.
Searching other resources
Grey literature
We searched sources of grey literature including dissertations and theses, clinical guidelines, and reports from regulatory agencies (when appropriate).
ProQuest Dissertations and Theses Database.
Reference lists
We scrutinised the reference lists of all included studies and relevant systematic reviews to identify additional missed studies. We also conducted a cited reference search on the Web of Science.
Correspondence
We contacted trialists and subject matter experts for information on unpublished or ongoing studies, including studies reported as ongoing in the previous searches, and to request additional trial data.
Data collection and analysis
We followed guidance on data collection and analysis provided by the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2019).
Selection of studies
Two review authors (NS and LR) independently screened titles and abstracts of studies identified by the updated search, and coded them as 'retrieve' or 'do not retrieve'. We retrieved the full‐text publications of all potentially eligible studies, and the same two review authors independently screened and identified studies for inclusion. We recorded reasons for exclusion of ineligible studies. We sought advice and resolution of disagreements through discussions with three review authors (JB, CL, and NR), and recorded the selection process in sufficient detail to complete a PRISMA flow diagram and Characteristics of excluded studies table.
Data extraction and management
We used a data extraction form that was used in the previous search to extract study characteristics and outcome data.
Two review authors (NS and LR) independently extracted the following study characteristics and outcome data from included studies.
Methods: study design, duration of the study, study setting, withdrawals, year of the study.
Participants: number, mean age, age range, gender, primary trauma, time since trauma, severity of condition, diagnostic criteria, inclusion criteria and exclusion criteria, comorbidity, multiple traumas, trauma during childhood.
Interventions: intervention, number of hours of guidance, nature of guidance, training and qualifications of guiding therapists, amount of time spent on the programme, comparison, concomitant and excluded interventions, type of device.
Outcomes: primary and secondary outcomes specified and collected, time points reported.
Notes: funding for trial, notable conflicts of interest of trial authors (e.g. if they were involved in development of the intervention).
We noted in the Characteristics of included studies table if outcome data were not reported in a useable way. We resolved disagreements by consensus, or with involvement of additional review authors (JB, CL, and NR). One review author (LR) transferred data into Review Manager 5 (Review Manager 2014). We double‐checked data by comparing data presented in the systematic review with data provided in the study reports. A second review author (NS) spot‐checked study characteristics for accuracy against the trial report.
Assessment of risk of bias in included studies
Two review authors (NS and LR) independently assessed the risk of bias for each study, using the criteria outlined in the Cochrane Handbook for Systematic Reviews of Interventions and listed below (Higgins 2019). We sought advice and resolution of disagreements through discussions with three review authors (JB, CL, and NR).
Random sequence generation.
Allocation concealment.
Blinding of participants and personnel.
Blinding of outcome assessment.
Incomplete outcome data.
Selective outcome reporting.
Other bias (including baseline imbalances, early termination of the trial, researcher allegiance).
We judged each potential source of bias as high, low, or unclear, and provided a supporting quotation from the study report, together with a justification for the judgement, in the 'Risk of bias' table. We summarised risk of bias judgements across different studies for each of the domains listed. We considered blinding separately for different key outcomes when necessary. When information on risk of bias related to unpublished data or correspondence with a trialist, we noted this in the 'Risk of bias' table.
When considering treatment effects, we took into account risk of bias for studies that contributed to that outcome.
For cross‐over trials, we also took the following into account.
Suitability of the cross‐over design.
Possibility of carry‐over effects.
Whether only first period data were available.
Incorrect analysis.
Comparability of results with those from parallel‐group trials.
Measures of treatment effect
Dichotomous data
We analysed dichotomous data as risk ratios (RR) to allow comparison across studies. We presented all outcomes with 95% confidence intervals (CIs).
Continuous data
We analysed continuous data as mean differences (MDs) or SMDs, to allow comparison across studies. We calculated MDs when all studies within a meta‐analysis used the same outcome measure, and SMDs when studies used different measures. We entered data presented on a scale with a consistent direction of effect. We presented all outcomes using 95% CIs, and undertook meta‐analyses only when it was meaningful to do so (i.e. when treatments, participants, and the underlying clinical question were sufficiently similar). We planned to describe skewed data reported as medians and interquartile ranges in a narrative, and when multiple trial arms were reported in a single trial, we planned to include only the relevant arms.
We assessed clinical significance by taking into account the size of a treatment effect, the severity of the condition being treated, and the adverse effects of the treatment.
Unit of analysis issues
Cluster‐randomised trials
We decided that, when necessary, we would adjust sample sizes, using an estimate of the intracluster or intraclass correlation coefficient (ICC), which describes the similarity of participants within the same cluster. We planned to derive this from the trial if possible, or from another source, such as a similar study, or from a resource providing examples of ICCs, if data were not available in the trial report.
Cross‐over trials
When a study adopted a cross‐over design, we planned to only include outcome data from the first randomisation period, to avoid a carry‐over effect.
Studies with multiple treatment groups
We planned to undertake pair‐wise meta‐analysis with each arm, depending on the nature of the intervention in each arm and its relevance to the review objectives. We aimed to avoid multiple comparisons to limit the risk of false‐positive results. If a study included three or more arms that were relevant to the review, we planned to assess the appropriateness of combining data from two arms if therapies were sufficiently similar, or of using data from the arms of the trial that fit most closely with the review objectives. For studies with multiple treatments arms, some of which were relevant to the review, we still listed the treatment arms in the Characteristics of included studies table. Decisions followed guidance provided by the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2019).
Dealing with missing data
We contacted investigators to verify key study characteristics, and to request missing outcome data. We documented all correspondence with trialists and reported which trialists responded. The protocol described the use of imputation of missing data; however, only published data were presented in the review. Should we find incidents of inadequate reporting of data in future updates of this review, we will attempt to impute missing data from other available information, in line with guidance provided by the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2019).
Assessment of heterogeneity
We assessed studies included in each comparison for clinical heterogeneity in terms of variability in experimental and comparator interventions, participants, settings, and outcomes. To further assess heterogeneity, we used both the I² statistic and the Chi² test of heterogeneity, and visually inspected the forest plots. We used the following scale suggested by the Cochrane Handbook for Systematic Reviews of Interventions as a guide to interpretation of the I² statistic (Higgins 2019).
0% to 40%: might not be important.
30% to 60%: may represent moderate heterogeneity.
50% to 90%: may represent substantial heterogeneity.
75% to 100%: shows considerable heterogeneity.
We interpreted the I² statistic with consideration of the size and direction of effects, as well as the strength of evidence for heterogeneity.
Assessment of reporting biases
We specified that if sufficient studies were available in a meta‐analysis (10 or more), we would prepare funnel plots and examine these for signs of asymmetry. We specified that if asymmetry was identified, we would consider other possible reasons for this.
Data synthesis
We pooled data from more than one study when appropriate. We performed random‐effects meta‐analyses as we anticipated substantial heterogeneity between trials. We conducted fixed‐effect analyses as sensitivity analyses to informally compare the results. When studies could not be combined, we summarised them in a narrative.
We planned the following comparisons:
I‐C/BT versus face‐to‐face CBT;
I‐C/BT versus face‐to‐face non‐CBT;
I‐C/BT versus wait list or usual care;
I‐C/BT versus Internet psychoeducation;
I‐C/BT versus I‐non‐C/BT.
Subgroup analysis and investigation of heterogeneity
We specified that we would consider the following possible causes of clinical heterogeneity for exploration, if sufficient data allowed.
Therapist assistance (e.g. Internet‐based interventions delivered with guidance, Internet‐based interventions delivered without guidance), as this varies substantially between interventions and may impact trial results.
Type of therapist assistance (e.g. guidance face‐to‐face, by telephone, by video conference, by email, by instant messaging).
Participant subgroups (e.g. veterans, female victims of sexual abuse, police officers), as some subgroups are more difficult to treat than others, and may be more or less suited to an online approach to treatment.
Type of recruitment (e.g. from media adverts only, from healthcare services only), as this may influence motivation and symptom severity of trial participants.
Type of CBT (e.g. predominantly CT, predominantly BT, CBT).
Baseline symptom severity (e.g. high versus low baseline mean symptom severity), on the basis that I‐C/BT is commonly thought to be better suited to people with milder symptoms.
Trauma type and context (e.g. war, childhood abuse, motor vehicle accident), on the basis that I‐C/BT is commonly thought to be better suited to people with less complex trauma histories.
Trauma focus (e.g. trauma‐focused versus non‐trauma‐focused I‐C/BT), as findings from the wider PTSD literature support trauma‐focused interventions as most effective.
Type of device (e.g. computer, smartphone), as availability on a smartphone is thought to improve outcomes.
We intended to keep subgroup analyses to a minimum to avoid issues related to multiple testing, and to only conduct these analyses on primary outcome measures.
Sensitivity analysis
We specified that we would consider sensitivity analysis to explore possible causes of methodological heterogeneity, if sufficient data allowed. We planned to base analyses on the following criteria.
Sequence generation.
Allocation concealment.
Blinding of outcome assessment.
Method of diagnosis (e.g. clinician diagnosis, structured interview, screening tool or questionnaire).
We planned to conduct these analyses for primary outcomes by removing studies with high or unknown risk of bias for these domains.
Summary of findings and assessment of the certainty of the evidence
We evaluated the certainty of the evidence using the GRADE approach. For each comparison, we planned to generate a 'Summary of findings' table using GRADEpro GDT software, which imports data from Review Manager 5 (GRADEpro GDT; Review Manager 2014). These tables provided outcome‐specific information concerning the overall certainty of evidence from studies included in the comparison, the magnitude of effect of the interventions examined, and the sum of available data on outcomes considered. We included information on the first seven outcomes of our review: severity of PTSD symptoms post‐treatment, dropouts, diagnosis of PTSD after treatment, severity of depressive symptoms, severity of anxiety symptoms, cost‐effectiveness, and adverse events. We assessed the certainty of evidence using five factors.
Limitations in study design and implementation of available studies.
Indirectness of evidence.
Unexplained heterogeneity or inconsistency of results.
Imprecision of effect estimates.
Potential publication bias.
For each outcome, we classified the certainty of evidence according to the following GRADE categories.
High certainty: further research is very unlikely to change our confidence in the estimate of effect.
Moderate certainty: further research is likely to have an important impact on our confidence in the estimate of effect, and may change the estimate.
Low certainty: further research is very likely to have an important impact on our confidence in the estimate of effect, and is likely to change the estimate.
Very low certainty: we are very uncertain about the estimate.
We downgraded the evidence from high certainty by one level for serious (or by two levels for very serious) study limitations (risk of bias), indirectness of evidence, serious inconsistency, imprecision of effect estimates, or potential publication bias.
Results
Description of studies
See Characteristics of included studies table.
Results of the search
The update search (5 June 2020) identified 1252 records and after the Information Specialist removed 393 duplicate records plus a further 163 records from the earlier 2018 search, this left 696 records to screen. After we assessed the titles, abstracts, and relevant full‐text documents, we identified three additional studies which met the inclusion criteria (Allen 2020; Bragesjö 2020; McLean 2020a).
A previous update search (1 March 2018), conducted for the first published version of this review, identified 861 records and after the Information Specialist removed 359 duplicate records plus 161 reports of uncontrolled trials, this left 341 records to screen. After we assessed the titles, abstracts, and relevant full‐text documents, we identified four additional studies that met the inclusion criteria (Krupnick 2017; Kuhn 2017; Littleton 2016; Miner 2016). These were added to the qualitative and quantitative analyses together with two studies that were previously awaiting classification (Engel 2015; Knaevelsrud 2015).
The first round of searches, conducted shortly after the protocol was published in 2015 (on the CCMDCTR and the PILOTS database only), identified 669 potentially relevant studies for consideration, plus 99 studies from other sources. After removing duplicates, this left 481 records to screen. After we assessed the titles, abstracts, and relevant full‐text documents, we identified four studies which met the inclusion criteria and were included in the qualitative and quantitative analyses (Ivarsson 2014; Lewis 2017; Litz 2007; Spence 2011).
We excluded 45 studies (see Characteristics of excluded studies table).
We identified 21 ongoing studies (see Characteristics of ongoing studies table).
The process of study selection is illustrated in the PRISMA diagram in Figure 1.
1.
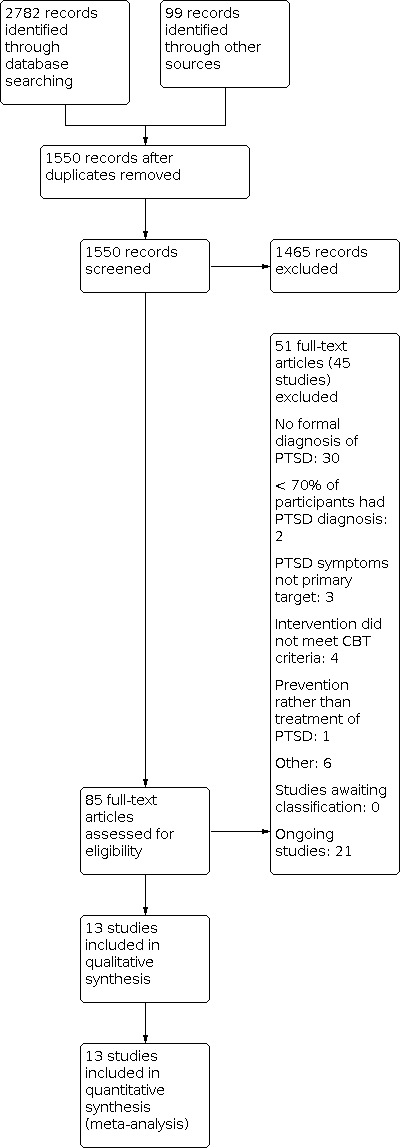
Study flow diagram.
Included studies
The review included 13 RCTs of 808 participants.
Design
All included studies were RCTs. All studies randomly assigned participants rather than clinics or practices.
Sample sizes
The studies had sample sizes of 40 (I‐C/BT 21; wait list 19; Allen 2020); 8 (I‐C/BT 3; wait list 5; Bragesjö 2020); 80 (I‐C/BT 43; optimised usual care 37; Engel 2015); 62 (I‐C/BT 31; delayed treatment 31; Ivarsson 2014); 159 (I‐C/BT 79; wait list 80; Knaevelsrud 2015); 34 (I‐C/BT 17; treatment as usual 17; Krupnick 2017); 120 (I‐C/BT 62; wait list 58; Kuhn 2017); 42 (I‐C/BT 21; wait list 21; Lewis 2017); 87 (I‐C/BT 46; supportive counselling 41; Littleton 2016); 45 (I‐C/BT 24; supportive counselling 21; Litz 2007); 40 (I‐C/BT 20; PCT 20; McLean 2020a); 49 (I‐C/BT 25; wait list 24; Miner 2016); and 42 (I‐C/BT 21; wait list 21; Spence 2011).
Setting
Seven studies were conducted in the US (Engel 2015; Krupnick 2017; Kuhn 2017; Littleton 2016; Litz 2007; McLean 2020a; Miner 2016), two in Sweden (Bragesjö 2020; Ivarsson 2014), two in Australia (Allen 2020; Spence 2011), one in the UK (Lewis 2017), and one in Iraq (Knaevelsrud 2015).
Participants
Three studies included participants who met criteria for DSM‐5 PTSD (Bragesjö 2020; Lewis 2017; McLean 2020a); the other 10 studies included participants who met criteria for DSM‐IV PTSD (Allen 2020; Engel 2015; Ivarsson 2014; Knaevelsrud 2015; Krupnick 2017; Kuhn 2017; Littleton 2016; Litz 2007; Miner 2016; Spence 2011). Four studies included military personnel and veterans traumatised after combat‐related exposure (Engel 2015; Krupnick 2017; Litz 2007; McLean 2020a); one study included only female rape victims (Littleton 2016); and the remainder included participants traumatised after a variety of traumatic events that met DSM criteria. See Characteristics of included studies table for further details. Where reported, the percentage of women in studies ranged from 18.75% to 100%; the percentage of participants with a university education ranged from 14.2% to 62.8%; and the percentage of participants who were unemployed ranged from 8.1% to 40%.
Interventions
Ten studies compared an Internet programme based on CBT with a wait list, treatment as usual, or delayed treatment control group (Allen 2020; Bragesjö 2020; Engel 2015; Ivarsson 2014; Knaevelsrud 2015; Krupnick 2017; Kuhn 2017; Lewis 2017; Miner 2016; Spence 2011). Treatment durations were three weeks (Bragesjö 2020), four weeks (Miner 2016), five weeks (Knaevelsrud 2015), six weeks (Engel 2015), eight weeks (Ivarsson 2014; Lewis 2017; McLean 2020a; Spence 2011), 10 weeks (Allen 2020) and 12 weeks (Kuhn 2017). The study by Krupnick 2017 delivered 10 sessions. Four studies examined i‐CBT interventions that were non‐trauma‐focused (Engel 2015; Kuhn 2017; Miner 2016; Spence 2011). Two studies examined i‐CBT interventions with no provision of guidance, and both were non‐trauma‐focused i‐CBT interventions (Kuhn 2017; Miner 2016).
Two studies compared an Internet programme based on trauma‐focused CBT with Internet psychological therapy (non‐CBT) (Littleton 2016; Litz 2007). In Litz 2007, the duration of treatment was eight weeks, while in Littleton 2016, treatment was for 14 weeks. A therapist guided all the Internet interventions.
One study compared web PE with PCT, a non‐trauma‐focused manualised in‐person therapy, over eight weeks (McLean 2020a).
See Characteristics of included studies table for further details.
Outcomes
Symptoms of PTSD were measured using the Impact of Event Scale (IES‐R), CAPS‐5, PCL‐5 and PTSD Checklist – Civilian Version (PCL‐C), the PTSD Symptom Scale – Interview (PSS‐I), and the Posttraumatic Diagnostic Scale (PDS). Depression was measured using the BDI, the Patient Health Questionnaire (PHQ‐7, PHQ‐8, PHQ‐9, PHQ‐15), the Montgomery‐Åsberg Depression Rating Scale (MADRS), and the Center for Epidemiologic Studies Depression Scale (CES‐D). Anxiety was measured using the BAI and the Generalized Anxiety Disorder 7‐Item Scale (GAD‐7). Quality of life was measured using the Quality of Life Inventory (QOLI), the EuroQol EQ‐5D, and EUROHIS‐QOL.
Excluded studies
For details, see the Characteristics of excluded studies table.
We excluded 45 studies from the update. The most common reasons for exclusion included no formal diagnosis of PTSD (30 studies), the intervention was deemed to not be CBT (four studies), reduction in PTSD symptoms were not the primary target (three studies) and less than 70% of participants had a PTSD diagnosis (three studies). Other reasons for exclusion included the participants requiring to screen positive for drug or alcohol use (one study), the trial did not start (one study), treatment was for anger (one study), treatment was for prevention rather than treatment of PTSD (one study), and use of PTSD Coach app in all four arms and measured the effectiveness of three different add‐on coaching interventions (clinician, peer, and algorithm) (one study).
Studies awaiting classification
There were no studies awaiting classification.
Ongoing studies
Twenty‐one studies were ongoing (ACTRN12611000989943; ACTRN12616000956404; DRKS00010245; DRKS00016931; DRKS00017838; DRKS00020266; Ehlers 2020; Golchert 2019; ISRCTN16806208; Lehavot 2017; McLean 2020b; NCT02929979; NCT04058795; NCT04094922; NCT04101942; NCT04155736; NCT04286165; NCT04333667; Nollett 2018; NTR6912; Sjomark 2018). For details, see the Characteristics of ongoing studies table.
Risk of bias in included studies
For details of the risk of bias judgements for each study, see the Characteristics of included studies table. A graphical representation of the overall risk of bias in included studies is presented in Figure 2 and Figure 3.
2.

'Risk of bias' graph: review authors' judgements about each risk of bias domain presented as percentages across all included studies.
3.

'Risk of bias' summary: review authors' judgements about each risk of bias domain for each included study.
For cluster‐randomised trials, we planned to take the following into account.
Recruitment bias (e.g. if participants had been recruited to the trial after clusters were randomly assigned).
Baseline imbalance.
Loss of clusters.
Incorrect analysis.
Comparability with individual randomised trial.
Allocation
Nine studies provided sufficient information to determine that there was a low risk of bias associated with sequence generation (Allen 2020; Bragesjö 2020; Engel 2015; Ivarsson 2014; Knaevelsrud 2015; Kuhn 2017; Lewis 2017; Littleton 2016; Spence 2011), while four studies did not provide enough information to make a judgement (Krupnick 2017; Litz 2007; McLean 2020a; Miner 2016). Two studies reported the use of sealed, opaque envelopes to conceal the allocation of treatment and were at low risk of selection bias (Allen 2020; Lewis 2017). The remaining 11 studies did not provide information to make a judgement on allocation concealment and were therefore classified at unclear risk of selection bias (Bragesjö 2020; Engel 2015; Ivarsson 2014; Knaevelsrud 2015; Krupnick 2017; Kuhn 2017; Littleton 2016; Litz 2007; McLean 2020a; Miner 2016; Spence 2011).
Blinding
It was impossible to blind participants or therapists, so all studies were at high risk of performance bias.
Eight studies reported adequate blinding of the outcome assessor (Engel 2015; Ivarsson 2014; Knaevelsrud 2015; Krupnick 2017; Kuhn 2017; Lewis 2017; Litz 2007; Miner 2016). In one study, outcome measures were self‐reported and, therefore, at low risk of detection bias (Bragesjö 2020). Two studies did not provide information to make a judgement (Allen 2020; McLean 2020a). The two remaining studies reported that outcome assessors were not blinded to treatment and were, therefore, at high risk of detection bias (Littleton 2016; Spence 2011).
Incomplete outcome data
Seven studies dealt with missing outcome data appropriately and were at low risk of attrition bias (Bragesjö 2020; Engel 2015; Ivarsson 2014; Kuhn 2017; Lewis 2017; Littleton 2016; Miner 2016). Six studies were at high risk of attrition bias (Allen 2020; Knaevelsrud 2015; Krupnick 2017; Litz 2007; McLean 2020a; Spence 2011). One study reported 49 randomised participants; however, only 40 were included in the intention‐to‐treat (ITT) analysis (Allen 2020). One study completed ITT analysis but missing data were more than 30% (Knaevelsrud 2015). One study had a dropout rate of over 75% and did not fully report reasons for dropout (Krupnick 2017). One study reported completer data only, but performed ITT analyses (Litz 2007). One study used the last observation carried forward method to impute missing data (Spence 2011). Reasons for dropout were poorly described.
Selective reporting
Only three studies published study protocols (Allen 2020; Knaevelsrud 2015; McLean 2020a). However, one of these studies was at unclear risk of bias since some secondary outcome measures that were included in the protocol paper were missing (Allen 2020). Of the 10 remaining studies, although the study protocol was not available, it was clear that the published reports included the outcomes for PTSD, depression, and anxiety, that were prespecified and expected in trials of this type in the field of PTSD. Therefore, these 10 studies were at low risk of reporting bias (Bragesjö 2020; Engel 2015; Ivarsson 2014; Krupnick 2017; Kuhn 2017; Lewis 2017; Littleton 2016; Litz 2007; Miner 2016; Spence 2011).
Other potential sources of bias
Five studies were at low risk of other bias (Engel 2015; Knaevelsrud 2015; Kuhn 2017; Littleton 2016; Miner 2016). Seven studies were at high risk of bias (Allen 2020; Bragesjö 2020; Ivarsson 2014; Krupnick 2017; Lewis 2017; Litz 2007; McLean 2020a). We could not rule out potential researcher allegiance, since five included trials were of interventions that were evaluated by their originators (Allen 2020; Ivarsson 2014; Lewis 2017; Litz 2007; McLean 2020a). Sample sizes in six studies were small (Allen 2020; Bragesjö 2020; Krupnick 2017; Lewis 2017; Litz 2007; McLean 2020a), and follow‐up was very limited in one study (Litz 2007). For practical and ethical reasons, longer‐term follow‐up data were not available from the wait list groups. One study was at unclear risk of other bias as it terminated recruitment prematurely and failed to recruit the prespecified number of participants (Spence 2011). One study was at high risk of bias since it terminated RCT design and recruitment prematurely, failing to recruit the prespecified number of participants, and also did not take into account in the analysis the large difference between groups in baseline PCL‐5 total scores (McLean 2020a).
Effects of interventions
See: Table 1; Table 2; Table 3
Comparison 1: Internet‐based cognitive and behavioural therapies versus face‐to‐face cognitive behavioural therapy
None of the included studies compared I‐C/BT versus face‐to‐face CBT.
Comparison 2: Internet‐based cognitive and behavioural therapies versus face‐to‐face non‐cognitive behavioural therapy
One study compared web PE with face‐to‐face non‐CBT (in‐person PCT) (McLean 2020a).
Primary outcomes
2.1. Severity of post‐traumatic stress disorder symptoms
Very low certainty evidence from one study (McLean 2020a) suggested that participants who received web PE had a higher PCL‐5 score post‐treatment than those who received face‐to‐face non‐CBT (MD 10.90, 95% CI 6.57 to 15.23; studies = 1, participants = 40; Analysis 1.1).
1.1. Analysis.

Comparison 1: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus face‐to‐face non‐CBT, Outcome 1: Severity of PTSD symptoms (post‐treatment)
This was also true when PTSD severity was measured < six months' post‐treatment (MD 5.67, 95% CI 1.56 to 9.78; studies = 1, participants = 40; very low‐certainty evidence; Analysis 1.2).
1.2. Analysis.

Comparison 1: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus face‐to‐face non‐CBT, Outcome 2: Severity of PTSD symptoms (follow‐up < 6 months)
2.2. Dropouts
Very low‐certainty evidence from one study (McLean 2020a) suggested no difference in the dropout rates between web PE and face‐to‐face non‐CBT (RR 2.49, 95% CI 0.91 to 6.77; studies = 1, participants = 40; Analysis 1.3).
1.3. Analysis.

Comparison 1: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus face‐to‐face non‐CBT, Outcome 3: Dropouts
Secondary outcomes
2.3. Diagnosis of post‐traumatic stress disorder after treatment
McLean 2020a did not report diagnosis of PTSD after treatment.
2.4. Severity of depressive symptoms
McLean 2020a did not measure severity of depressive symptoms.
2.5. Severity of anxiety symptoms
McLean 2020a did not measure severity of anxiety symptoms.
2.6. Cost‐effectiveness
McLean 2020a did not measure cost‐effectiveness.
2.7. Adverse events
McLean 2020a did not report adverse events.
2.8. Standardised measures of treatment acceptability
McLean 2020a did not report treatment acceptability.
2.9. Quality of life
McLean 2020a found no difference in health functioning between web PE and face‐to‐face non‐CBT.
Comparison 3: Internet‐based cognitive and behavioural therapies versus wait list or usual care
Ten studies including 608 participants compared I‐C/BT versus wait list or usual care (Allen 2020; Bragesjö 2020; Engel 2015; Ivarsson 2014; Knaevelsrud 2015; Krupnick 2017; Kuhn 2017; Lewis 2017; Miner 2016; Spence 2011). See Table 2.
Primary outcomes
3.1. Severity of post‐traumatic stress disorder symptoms
There was very low‐certainty evidence that I‐C/BT was more effective than wait list when the severity of PTSD symptoms were measured post‐treatment (SMD –0.61, 95% CI –0.93 to –0.29; studies = 10, participants = 608; Analysis 2.1). There was substantial heterogeneity in study results (I² = 69%). When duration of follow‐up was less than six months, there was no evidence of a difference between I‐C/BT and wait list groups, with considerable heterogeneity in study results (SMD –0.45, 95% CI –1.29 to 0.39; I² = 82%; studies = 4, participants = 154; Analysis 2.2). The methods of investigating this heterogeneity are reported in the Sensitivity analysis section.
2.1. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 1: Severity of PTSD symptoms (post‐treatment)
2.2. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 2: Severity of PTSD symptoms (follow‐up < 6 months)
A funnel plot did not find evidence of publication bias for severity of PTSD symptoms (Figure 4).
4.

Funnel plot of comparison: 2 Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list; outcome: 2.1 Severity of PTSD symptoms (post‐treatment).
Allen 2020 also measured PCL‐5 at three months' post‐treatment but only in the group who received I‐C/BT. The mean score was 45.29 (standard deviation (SD) 11.51).
3.2. Dropouts
There was low‐certainty evidence of no difference in dropout rates from the I‐C/BT and wait list groups (RR 1.25, 95% CI 0.97 to 1.60; I² = 34%; studies = 9, participants = 634; Analysis 2.3).
2.3. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 3: Dropouts
Secondary outcomes
3.3. Diagnosis of post‐traumatic stress disorder after treatment
There was very low‐certainty evidence that I‐C/BT was no more effective than wait list at reducing the risk of a diagnosis of PTSD after treatment (RR 0.53, 95% CI 0.28 to 1.00; studies = 1, participants = 62; Analysis 2.4).
2.4. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 4: Diagnosis of PTSD after treatment
3.4. Severity of depressive symptoms
There was very low‐certainty evidence that I‐C/BT was more effective than wait list at reducing the severity of depressive symptoms (SMD –0.51, 95% CI –0.97 to –0.06; studies = 7, participants = 473; Analysis 2.5). There was considerable heterogeneity in study results (I² = 80%). There was very low‐certainty evidence that I‐C/BT was still more effective than wait list at follow‐up less than six months (MD –8.75, 95% CI –14.71 to –2.80; studies = 2, participants = 50; Analysis 2.6).
2.5. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 5: Severity of depression (post‐treatment)
2.6. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 6: Severity of depression (follow‐up < 6 months)
Allen 2020 also measured PHQ‐9 at three months' post‐treatment but only in the group who received I‐C/BT. The mean score was 11.7 (SD 5.08).
3.5. Severity of anxiety symptoms
There was very low‐certainty evidence that I‐C/BT was more effective than wait list at reducing symptoms of anxiety (SMD –0.61, 95% CI –0.89 to –0.33; I² = 33%; studies = 5, participants = 345; Analysis 2.7). There was very low‐certainty evidence that I‐C/BT was still more effective than wait list at follow‐up less than six months (MD –12.59, 95% CI –20.74 to –4.44; studies = 1, participants = 42; Analysis 2.8).
2.7. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 7: Severity of anxiety symptoms (post‐treatment)
2.8. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 8: Severity of anxiety symptoms (follow‐up < 6 months)
Allen 2020 also measured GAD‐7 at three months' post‐treatment but only in the group who received I‐C/BT. The mean score was 8.78 (SD 4.8).
3.6. Cost‐effectiveness
None of the studies comparing I‐C/BT to wait list or usual care reported cost‐effectiveness.
3.7. Adverse events
Lewis 2017 reported an absence of adverse events. Bragesjö 2020 reported adverse events in their study overall; however, we were unable to draw conclusions with respect to the subset of data that were included. The other studies did not report the presence or absence of adverse events.
3.8. Standardised measures of treatment acceptability
Lewis 2017 did not measure treatment acceptability.
3.9. Quality of life
There was very low‐certainty evidence of no difference between the I‐C/BT and wait list groups for quality of life post‐treatment (SMD 0.59, 95% CI 0.18 to 1.01; I² = 40%; studies = 3, participants = 229; Analysis 2.9). Bragesjö 2020 also measured quality of life using EQ‐5D at six months (SMD 0.16, 95% CI –1.28 to 1.59; studies = 1, participants = 8).
2.9. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 9: Quality of life (post‐treatment)
Comparison 4: Internet‐based cognitive and behavioural therapies versus Internet psychoeducation
None of the included studies compared I‐C/BT versus Internet psychoeducation.
Comparison 5: Internet‐based cognitive and behavioural therapies versus Internet‐based non‐cognitive and behavioural therapies
Two studies including 82 participants compared I‐C/BT versus I‐non‐C/BT (Littleton 2016; Litz 2007). See Table 3.
5.1. Severity of post‐traumatic stress disorder symptoms
There was very low‐certainty evidence of no difference between the I‐C/BT and I‐non‐C/BT groups in the post‐treatment severity of PTSD symptoms (SMD –0.08, 95% CI –0.52 to 0.35; I² = 19%; studies = 2, participants = 82; Analysis 3.1), or at follow‐up of less than six months (SMD 0.08, 95% CI –0.41 to 0.57; I² = 0%; studies = 2, participants = 65; Analysis 3.2). However, there was evidence of a difference in favour of I‐C/BT at follow‐up of six to 12 months (MD –8.83, 95% CI –17.32 to –0.34; studies = 1, participants = 18; Analysis 3.3).
3.1. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 1: Severity of PTSD symptoms (post‐treatment)
3.2. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 2: Severity of PTSD symptoms (follow‐up < 6 months)
3.3. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 3: Severity of PTSD symptoms (follow‐up 6–12 months)
5.2. Dropouts
There was very low‐certainty evidence of no difference between dropout rates from the I‐C/BT and I‐non‐C/BT groups (RR 2.14, 95% CI 0.97 to 4.73; I² = 0%; studies = 2, participants = 132; Analysis 3.4).
3.4. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 4: Dropouts
Secondary outcomes
5.3. Diagnosis of post‐traumatic stress disorder after treatment
Neither study comparing I‐C/BT versus I‐non‐C/BT reported diagnosis of PTSD after treatment.
5.4. Severity of depressive symptoms
There was very low‐certainty evidence of no difference in severity of depressive symptoms between the I‐C/BT and I‐non‐C/BT groups after treatment (SMD –0.12, 95% CI –0.78 to 0.54; I² = 52%; studies = 2, participants = 84; Analysis 3.5) or when follow‐up was less than six months (SMD 0.20, 95% CI –0.31 to 0.71; I² = 0%; studies = 2, participants = 61; Analysis 3.6). However, there was very low‐certainty evidence of a difference in severity of depressive symptoms between the I‐C/BT and I‐non‐C/BT groups when follow‐up was six to 12 months (MD –8.34, 95% CI –15.83 to –0.85; studies = 1, participants = 18; Analysis 3.7).
3.5. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 5: Severity of depressive symptoms (post‐treatment)
3.6. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 6: Severity of depressive symptoms (follow‐up < 6 months)
3.7. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 7: Severity of depressive symptoms (follow‐up 6–12 months)
5.5. Severity of anxiety symptoms
There was very low‐certainty evidence of no difference in severity of symptoms of anxiety between the I‐C/BT and I‐non‐C/BT groups (SMD 0.08, 95% CI –0.78 to 0.95; I² = 70%; studies = 2, participants = 74; Analysis 3.8) or when follow‐up was less than six months (SMD –0.16, 95% CI –0.67 to 0.35; I² = 9%; studies = 2, participants = 60; Analysis 3.9). However, there was very low‐certainty evidence of a difference in severity of anxiety symptoms between the I‐C/BT and I‐non‐C/BT groups when follow‐up was six to 12 months (MD –8.05, 95% CI –15.20 to –0.90; studies = 1, participants = 18; Analysis 3.10).
3.8. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 8: Severity of anxiety symptoms (post‐treatment)
3.9. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 9: Severity of anxiety symptoms (follow‐up < 6 months)
3.10. Analysis.

Comparison 3: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT, Outcome 10: Severity of anxiety symptoms (follow‐up 6–12 months)
5.6. Cost‐effectiveness
Neither study comparing I‐C/BT versus I‐non‐C/BT reported cost‐effectiveness.
5.7. Adverse events
One study noted two participants in the I‐C/BT group reported a clinically significant increase in depression symptoms post‐treatment, with one of these participants also reporting a clinically significant increase in anxiety symptoms (Littleton 2016). However, it is difficult to attribute this to I‐C/BT as these participants had both experienced the death of an immediate family member during treatment. Three additional participants reported clinically significant increases in anxiety at post‐treatment. With respect to I‐non‐C/BT, one participant experienced a clinically significant increase in depression symptoms between post‐treatment and follow‐up.
5.8. Standardised measures of treatment acceptability
Neither study reported treatment acceptability.
5.9. Quality of life
Neither study comparing I‐C/BT versus I‐non‐C/BT reported quality of life.
Subgroup analyses
There were insufficiently clear data to perform subgroup analyses on the effect of type of therapist assistance, participant subgroups, type of recruitment, baseline symptom severity, trauma type and context, or type of device.
Guided versus non‐guided interventions
Eight studies examined I‐C/BT interventions that were guided (Allen 2020; Bragesjö 2020; Engel 2015; Ivarsson 2014; Knaevelsrud 2015; Krupnick 2017; Lewis 2017; Spence 2011), and two studies examined the intervention PTSD Coach, which is a non‐guided, or stand‐alone, self‐help intervention, originally developed as an adjunct to face‐to‐face treatment (Kuhn 2017; Miner 2016).
There was very low‐certainty evidence that guided I‐C/BT was more effective than wait list at reducing the severity of PTSD symptoms post‐treatment (SMD –0.78, 95% CI –1.09 to –0.47; studies = 8, participants = 439). There was very low‐certainty evidence for no difference between non‐guided I‐C/BT and wait list at reducing the severity of PTSD symptoms post‐treatment (SMD –0.09, 95% CI –0.39 to 0.22; studies = 2, participants = 169). There was a stronger effect for guided I‐C/BT compared to non‐guided I‐C/BT (P = 0.002; Analysis 4.1). There was moderate heterogeneity in the guided intervention results (I² = 52%).
4.1. Analysis.

Comparison 4: Subgroup analysis: guided versus non‐guided, Outcome 1: Severity of PTSD symptoms (post‐treatment)
There was very low‐certainty evidence for no difference for dropouts between guided I‐C/BT and wait list (RR 1.22, 95% CI 0.82 to 1.81; studies = 7, participants = 475), and non‐guided I‐C/BT and wait list (RR 1.95, 95% CI 0.83 to 4.57; studies = 2, participants = 169) (test for subgroup differences: P = 0.32; Analysis 4.2).
4.2. Analysis.

Comparison 4: Subgroup analysis: guided versus non‐guided, Outcome 2: Dropouts
Trauma‐focused versus non‐trauma‐focused intervention
Six studies examined trauma‐focused I‐C/BT interventions (Allen 2020; Bragesjö 2020; Ivarsson 2014; Knaevelsrud 2015; Krupnick 2017; Lewis 2017), and four studies examined non‐trauma focused I‐C/BT (Engel 2015; Kuhn 2017; Miner 2016; Spence 2011).
There was very low‐certainty evidence that trauma‐focused I‐C/BT was more effective than wait list at reducing the severity of PTSD symptoms post‐treatment (SMD –0.94, 95% CI –1.24 to –0.65; studies = 6, participants = 342). There was very low‐certainty evidence for no difference between non‐trauma‐focused I‐C/BT and wait list at reducing the severity of PTSD symptoms post‐treatment (SMD –0.16, 95% CI –0.41 to 0.08; studies = 4, participants = 266). There was a stronger effect for trauma‐focused I‐C/BT compared to non‐trauma‐focused I‐C/BT (P < 0.0001; Analysis 5.1).
5.1. Analysis.

Comparison 5: Subgroup analysis: trauma‐focused versus non‐trauma‐focused, Outcome 1: Severity of PTSD (post‐treatment)
There was very low‐certainty evidence for no significant difference for dropouts between trauma‐focused I‐C/BT and wait list (RR 1.22, 95% CI 0.75 to 1.98; studies = 5, participants = 343), and between non‐trauma‐focused I‐C/BT and wait list (RR 1.78, 95% CI 0.94 to 3.37; studies = 4, participants = 301) (test for subgroup difference: P = 0.89; Analysis 5.2). There was moderate heterogeneity in the trauma‐focused study results (I² = 52%).
5.2. Analysis.

Comparison 5: Subgroup analysis: trauma‐focused versus non‐trauma‐focused, Outcome 2: Dropouts
Sensitivity analyses
Where we had sufficient data, we investigated possible causes of methodological heterogeneity by conducting sensitivity analyses for primary outcomes by excluding any study at high or unclear risk of selection bias or detection bias.
There was substantial heterogeneity (I² = 69%) for the outcome severity of PTSD symptoms post‐treatment (Analysis 2.1). The meta‐analysis included 10 studies but nine studies were at unclear risk of either selection bias (Bragesjö 2020; Engel 2015; Ivarsson 2014; Knaevelsrud 2015; Krupnick 2017; Kuhn 2017; Miner 2016; Spence 2011) or detection bias (Allen 2020; Spence 2011). With the study by Lewis 2017 being the only study at low risk of bias in these domains, it was not possible to conduct a sensitivity analysis for this outcome. For methods of diagnosis, only one study (Lewis 2017) used a clinician‐administered scale (CAPS‐5), the remaining nine studies used a self‐reported scale (PCL‐5: Allen 2020; Bragesjö 2020; Engel 2015; Krupnick 2017; Kuhn 2017; Miner 2016; Spence 2011; or PDS: Ivarsson 2014; Knaevelsrud 2015), and, therefore, it was not feasible to conduct a sensitivity analysis.
There was considerable heterogeneity for the outcome severity of PTSD symptoms at follow‐up of up to 6 months (I² = 82%; Analysis 2.2). There were four studies in this meta‐analysis (Bragesjö 2020; Engel 2015; Lewis 2017; Miner 2016). Three of those studies were at unclear risk selection bias due to insufficient reporting of sequence generation (Miner 2016) or allocation concealment (Bragesjö 2020; Engel 2015; Miner 2016), making it impossible to perform a sensitivity analysis for this outcome. For method of diagnosis, one study used the clinician‐administered scale (CAPS‐5) (Lewis 2017), while the remaining three studies used the self‐reported PCL‐5 (Bragesjö 2020; Engel 2015; Miner 2016). Therefore, due to insufficient data, it was not feasible to conduct a sensitivity analysis.
Discussion
Summary of main results
The review included 13 studies with 808 participants (see Table 1; Table 2; Table 3). Ten studies compared I‐C/BT with a wait‐list control group. Two studies compared I‐C/BT with I‐non‐C/BT and one study compared I‐C/BT with face‐to‐face non‐C/BT.
I‐C/BT may be more effective than a wait list in reducing PTSD symptoms, depression, and anxiety post‐treatment. Guided I‐C/BT appears more effective than non‐guided I‐C/BT, and trauma‐focused I‐C/BT more effective than non‐trauma‐focused I‐C/BT, in reducing PTSD symptoms. However, there was no significant difference between the two groups on measures of quality of life. The magnitude of effect was smaller than that found in comparisons of therapist‐administered trauma‐focused CBT with wait list or usual care (Bisson 2013; Lewis 2020a). This may indicate that while beneficial, this is a less effective form of treatment than equivalent face‐to‐face therapies. There was no significant difference in dropout rates between groups, and this remained when dropout rates were analysed comparing wait list with guided I‐C/BT interventions, non‐guided I‐C/BT interventions, trauma‐focused I‐C/BT, and non‐trauma‐focused I‐C/BT.
There was no difference between I‐C/BT and I‐non‐C/BT on any measure post‐treatment. Only two small studies made this comparison; they found that I‐C/BT was no more effective in reducing severity of PTSD symptoms, depression, and anxiety at 12‐month follow‐up. There was no significant difference in dropout rates between groups, irrespective of whether the I‐C/BT interventions were guided compared with non‐guided, or trauma‐focused compared with non‐trauma‐focused.
Therapist‐guided I‐C/BT appeared less effective than face‐to‐face non‐CBT (PCT) in reducing PTSD severity but was based on one small study of very low certainty. It is important to note that the RCT was abandoned prematurely, failing to recruit the prespecified number of participants, raising concern regarding the study's statistical power. Also of potential concern was the fact that the researchers did not take in to account in the analysis the large difference between the groups in baseline PCL‐5 total scores. Furthermore, selective reporting was at high risk of bias. These limitations aside, the apparent inferiority of the web‐based PE I‐C/BT could also be explained by the lack of cognitive work in the web‐PE intervention.
Overall completeness and applicability of evidence
The field of I‐C/BT for PTSD is growing; however, to date a limited number of RCT studies are available. Drawing together the results of these studies provided an indication of the efficacy of I‐C/BT for PTSD. The small number of studies contributing to each comparison did, however, limit our ability to comprehensively answer the questions that we set out to address. For example, we were unable to draw any conclusions related to cost‐effectiveness. We were also unable to draw clear conclusions comparing the efficacy of I‐C/BT with face‐to‐face psychological therapy or the provision of education. This indicates a need for further research, and an update of the review once further studies, including ongoing and planned studies that were identified in this review, have been completed.
Eligible RCTs included adults who had been exposed to a variety of traumatic events (see Types of participants). We included all studies in which at least 70% of participants had been diagnosed with PTSD. We excluded studies that evaluated I‐C/BT in participants with subthreshold PTSD symptoms, or traumatised people who were not formally diagnosed as having PTSD (see Characteristics of excluded studies table). This approach is in keeping with the Cochrane Review of therapist‐administered psychological therapies for PTSD, and the aim was to ensure the empirical validity of the review (Bisson 2013). However, this did result in the exclusion of studies that would have contributed additional data.
We did not exclude studies on the basis of duration of symptoms and because we did not stipulate that PTSD symptoms would need to be present for at least one month as an eligibility criterion, we cannot be certain of the number of participants fulfilling this diagnostic threshold. Studies were eligible where at least 70% of people met diagnostic criteria for PTSD according to DSM and ICD criteria and this could be assessed by clinical interview or by a score above a pre‐established threshold on a validated traumatic stress questionnaire. Therefore, this criterion enabled the potential inclusion of a wider number of studies, not limited to clinical interview assessment of PTSD, however we acknowledge as a potential concern that those studies that assessed PTSD according to a score above a pre‐established threshold on a questionnaire had been experiencing symptoms for at least one month at the time of assessment.
The included studies were conducted in Sweden, the UK, the USA, Iraq, and Australia, which limits generalisability of results to the rest of the world. Despite Iraq being a low‐ to middle‐income country, the included study conducted in Iraq was not of the general Iraq population, therefore generalisability to low‐ and middle‐income countries is especially limited. The studies did not include participants with comorbidities of substance dependence, psychosis, and severe depression, therefore, excluding people who are arguably more difficult to treat; however, this is also a limitation across studies of all psychological treatment interventions, including those delivered face‐to‐face. Participants included in the 13 studies were predominantly white, employed, and had relatively high levels of education. This limits our confidence that the findings can be generalised to all populations accessing Internet‐based therapies. Furthermore, while some people with these characteristics may prefer an effective treatment option that provides more flexibility, arguably some people with these characteristics would have increased access to resources for face‐to‐face therapies.
None of the eligible studies included data on cost‐effectiveness; however, they hold promise from an economic standpoint, since they would have cost less to deliver than face‐to‐face therapy, given the reduced contact time with therapists. It is important to note, however, that while it is assumed that I‐C/BT are low cost, there may be significant costs associated with developing, hosting, and updating these types of interventions, which should not be ignored. Despite high levels of dropout from the included studies, this is not unique to I‐C/BT approaches, with high dropouts found across psychological interventions for PTSD (Lewis 2020b). There was no difference in dropout rates between the I‐C/BT and wait list groups, indicating I‐C/BT was no less tolerable than waiting for treatment. Further analyses found that this remained when I‐C/BT was compared to wait list for guided and non‐guided I‐C/BT, and for trauma‐focused and non‐trauma‐focused I‐C/BT. However, the reasons given for dropout were rarely described, which precluded indepth consideration related to tolerability and acceptability.
As is common in studies of psychological therapies, concurrent pharmacotherapy was permissible. This caused issues differentiating the effects of medication versus the therapy being trialled. This is largely unavoidable, due to ethical considerations, and all studies stipulated that dosage had been constant for a stipulated duration. Although concurrent psychological therapy was an exclusion criterion for the included studies, we cannot be certain that attempts to formally evaluate whether additional treatment had been sought were taken across studies. Indeed, one study gave the possibility of participants in the control group having engaged in psychological therapy during the study period as a possible explanation for a failure to find between‐group differences, despite large within‐group differences in the treatment group (Spence 2011).
To ensure consistency with other Cochrane Reviews of psychological therapies for PTSD, there was a requirement that at least 70% of participants met diagnostic criteria for PTSD. This resulted in the exclusion of several studies of traumatised people with subthreshold PTSD symptoms from the review. It may be argued that this limits the clinical relevance of the review, since Internet‐based interventions may be useful to people with subthreshold PTSD symptoms, and their use is not limited to those meeting full diagnostic criteria. However, it is intuitive that interventions that are effective for people meeting the criteria for a diagnosis will also be effective in reducing traumatic stress symptoms among people with subthreshold symptomology. Therefore, restricting the review to studies with clinical samples takes a conservative approach, and, consistent with the aims of the review, provides an indication of whether I‐C/BT is effective for the treatment of clinically significant PTSD symptoms. It is also worth noting that people who have volunteered to be part of a trial may engage more with I‐C/BT than the general population of people with PTSD, which may have an impact on results.
There was insufficient data to perform subgroup analyses and determine the potential influence of, type of therapist assistance, participant subgroups, type of recruitment, type of CBT, baseline symptom severity, trauma type and context, or type of device.
Quality of the evidence
We planned to generate a summary of findings table for each of the five comparisons in the review. However, due to lack of data, we were only able to generate tables for three comparisons. We used the five GRADE considerations (study limitations, consistency of effect, imprecision, indirectness, and publication bias) to assess the certainty of the body of evidence for each outcome, and to draw conclusions about the certainty of evidence within the text of the review. The certainty of the evidence was graded as very low to low. One of the main considerations for downgrading GRADE judgements was risk of bias. Concerns over the certainty of the evidence also limit the extent to which conclusions can be generalised. For details of the risk of bias judgements for each study, see the Characteristics of included studies table and the graphical representation of risk of bias presented in Figure 2 and Figure 3.
There was heterogeneity across the I‐C/BT programmes, which varied in content, delivery, and guidance. Although all were based on cognitive‐behavioural principles, the exact nature of what was included varied. The extent and method by which the Internet‐based therapies were guided by a professional also varied.
Potential biases in the review process
The review rigorously followed guidelines set out by Cochrane (Higgins 2019). Two review authors independently screened the abstracts identified by the literature search; read all potentially relevant studies; assessed each study against the inclusion criteria; extracted data from the written reports; and rated each study for risk of bias. We discussed any disagreements with a third review author and reached unanimous decisions for inclusion and classification. We carefully followed guidelines set out by Cochrane on statistical methods and used GRADE to assess the certainty of evidence (Andrews 2013). Following these procedures minimised the potential for bias, but some unavoidable issues remained.
All but one of the included studies in this review were published, which led to the possibility of publication bias. Only one meta‐analysis included more than 10 studies, and while the symmetry of the funnel plot when comparing I‐C/BT and wait list for severity of PTSD symptoms post‐treatment (Figure 4) did not identify evidence of publication bias for this outcome, it was not possible to construct funnel plots for the other comparisons and outcomes (Higgins 2019). In an attempt to overcome this limitation, we searched clinical trial registries with the aim of identifying unpublished trials. That said, we could not rule out the possibility that trials were not registered, which may be the case for smaller studies, pilot work, or unfunded research.
We systematically searched online databases for potentially relevant studies, and scrutinised reference lists of included studies. We contacted experts in the field requesting help to identify missed studies or ongoing work. Nonetheless, we could not fully eliminate the possibility of overlooked RCTs. The included studies appeared to report data for all outcomes of primary interest. However, we could not identify published protocols for the included studies, and, therefore, it was possible that other outcomes of interest were collected but not reported.
There was considerable statistical heterogeneity in two of the four pooled comparisons. In circumstances where heterogeneity was thought to be potentially problematic, we used a random‐effects model. Heterogeneity was also a factor that was taken into consideration in downgrading the certainty of the evidence with GRADE.
Agreements and disagreements with other studies or reviews
Cochrane Reviews have found that trauma‐focused I‐C/BT interventions showed a smaller effect than therapist‐administered interventions in comparison to wait list (Bisson 2013) (Lewis 2020a).
Systematic reviews of I‐C/BT interventions for other disorders have reported better outcomes for guided interventions compared to those without guidance, and the findings of our subgroup analysis suggest greater efficacy for guided I‐C/BT for PTSD. I‐C/BT has been demonstrated as effective for levels of anxiety and depression in people with anxiety and depressive disorders, with smaller effect sizes where the I‐CBT interventions include less therapist time (Cuijpers 2009), and comparable to face‐to‐face psychotherapy, in favour of GSH (Cuijpers 2010).
The comparison of I‐C/BT to wait list in the current review yielded a larger effect size than the equivalent comparison in a previous Cochrane Review of media‐delivered CBT for anxiety disorders in adults (Mayo‐Wilson 2013). The effect size was of a similar magnitude to that obtained in the equivalent comparison in a Cochrane Review of I‐C/BT for anxiety disorders, which excluded interventions provided without therapist assistance, and interventions that included face‐to‐face therapist assistance (Olthuis 2015). However, these comparisons should be treated with caution, due to the small number of studies and high level of heterogeneity in the studies included in the current review.
Authors' conclusions
Implications for practice.
Internet‐based cognitive and behavioural therapy (I‐C/BT) has grown in popularity due to its potential to broaden access to cost‐effective treatment in a climate of limited resources, ever increasing pressure on psychological services, and public preference to use the Internet, not to mention barriers to individual in‐person treatment resulting from COVID‐19 lockdown restrictions. While the extent of therapist support varied considerably across the interventions considered by this review, they all required significantly less therapist time than current first‐line treatments, which can consist of eight to 12 weekly one‐hour sessions. This creates an opportunity to increase therapeutic capacity and optimise access to evidence‐based treatment. It has several potential benefits, including reduced distress and improved functioning. While the review found some beneficial effects of I‐C/BT for post‐traumatic stress disorder (PTSD), the certainty of the evidence was very low due to the small number of included studies. This is reflected in UK (NICE 2018) and international (ISTSS 2018) guidelines, which recommend guided self‐help trauma‐focused I‐C/BT, though only to a moderate level given that the evidence is emerging, with a lower quality of evidence or certainty of effect (or both).
Implications for research.
The field of I‐C/BT for PTSD has lagged behind other disorders, and only a handful of studies have evaluated the efficacy of the approach within a clinical population. Replication of findings is thereby a continued requirement. We are aware of only one randomised controlled trial to date comparing I‐C/BT for PTSD with therapist‐administered non‐CBT psychological therapy, present‐centred therapy (McLean 2020a), which suggested inferiority of I‐C/BT. Apart from Bragesjö 2020, none of the included studies reported adverse events, and only one additional study reported the presence/absence of adverse events (Lewis 2017).
There have been no trials directly comparing I‐C/BT with or without guidance, and no attempts to ascertain the optimal level of therapist input. In addition, we currently have a poor understanding of who can benefit from I‐C/BT for PTSD. All but one of the studies were conducted in high‐income countries and participants in the included trials were predominantly white and highly educated. It is unknown how results would generalise to other populations. There is potential for I‐C/BT to have an important role in the provision of global mental healthcare. It could be especially useful in low‐ and middle‐income countries, where fewer appropriately trained clinicians are available, although Internet connection stability may be a concern in such countries. However, further work is needed to explore the feasibility of using I‐C/BT in these countries, and to determine the acceptability of the approach within other countries.
Dropout from Internet‐based therapies is a concern, and our current understanding of predictors of dropout is limited, indicating the need to further explore this within future trials. There is a need to explore predictors of outcome and dropout, such as participant age, trauma type, levels of computer literacy, and symptom severity. This will provide a greater understanding of the best candidates for I‐C/BT, and enable interventions to be targeted accordingly. I‐C/BT may be most appropriate for people with mild‐to‐moderate PTSD, in a stepped or stratified pathway of care. There is also a need to investigate whether I‐C/BT might be helpful in treating complex PTSD. Future studies exploring the feasibility of I‐C/BT and willingness to engage with Internet‐based therapies would be valuable, as well as knowledge of the factors likely to impact broader implementation of I‐C/BT across the UK National Health Service (NHS), specifically the views of NHS employees involved in intervention commissioning, implementation, and provision. We know little in terms of the long‐term effects of I‐C/BT, indicating a need for future trials to include longer‐term follow‐up. Further work is also needed to back‐up a common assumption that I‐C/BT is cost effective, and to evaluate this for the treatment of PTSD. We need to understand factors associated with I‐C/BT efficacy and acceptability, including the potential role of acceptability in mediating treatment outcome and the optimal type and level of guidance, to develop I‐C/BT targeted to those who might benefit most from this approach to treatment (Klein 2010).
We were unable to draw any conclusions related to cost‐effectiveness. We were also unable to compare the efficacy of I‐C/BT with the provision of psychoeducation, or draw meaningful conclusions with respect to comparing I‐C/BT with face‐to‐face psychological therapy. This indicates a need for further research, and an update of the review once further planned and ongoing studies have been completed.
What's new
| Date | Event | Description |
|---|---|---|
| 18 May 2021 | New citation required but conclusions have not changed | New citation, conclusions remain unchanged. |
| 18 May 2021 | New search has been performed | This is an update of an existing Cochrane Review (Lewis 2018). The update includes 3 new randomised controlled trials. |
History
Protocol first published: Issue 5, 2015 Review first published: Issue 12, 2018
| Date | Event | Description |
|---|---|---|
| 21 May 2020 | Amended | A clarification message from the Co‐ordinating Editor was added to the Declarations of interest statement for an earlier version of the review (Lewis 2018). This was with regard to compliance with the Cochrane conflict of interest policy, which includes the relevant parts of the Cochrane Commercial Sponsorship Policy. It was agreed the review would be updated within 12 months and that the update would have a majority of authors and lead author free of conflicts. |
Acknowledgements
We acknowledge the Cochrane Common Mental Disorders Group for the support they provided during preparation of this review update.
The review authors and the Cochrane Common Mental Disorders Editorial Team are also grateful to the peer reviewers and editors for their time and comments including: N Livingstone and B Rothbaum. They would also like to thank Copy Edit Support.
CRG funding acknowledgement: the National Institute for Health Research (NIHR) is the largest single funder of the Cochrane Common Mental Disorders Group.
Disclaimer: the views and opinions expressed herein are those of the review authors and do not necessarily reflect those of Cardiff University, the NIHR, the National Health Service, or the Department of Health and Social Care.
Appendices
Appendix 1. Database search strategies
Update search (2018 to June 2020)
MEDLINE (2018 to 4 June 2020), 244 records
Embase (2018 to 2020 week 22), 179 records
PsycINFO (2018 to 4 June 2020), 114 records
CENTRAL (1 March 2018 to 5 June 2020), 325 records
CENTRAL Trial Registry Records, 22 records
PTSDpubs (2018 to 5 June 2020), 234 records
ClinicalTrials.gov (all years), 127 records
Total = 1223 Duplicates removed (within this batch): 392 Duplicates removed (from earlier search, March 2018): 163 To screen: 690
[535 Trial Registry records from CLib:CENTRAL were removed and screened separately (22 selected)]
Search strategies: 1. Ovid MEDLINE databases: Ovid MEDLINE(R) Epub Ahead of Print, In‐Process & Other Non‐Indexed Citations, Ovid MEDLINE(R) Daily and Ovid MEDLINE(R) search strategy ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 "trauma and stressor related disorders"/ or stress disorders, traumatic/ or combat disorders/ or psychological trauma/ or stress disorders, post‐traumatic/ or stress disorders, traumatic, acute/ 2 (PTSD or ((posttrauma* or post‐trauma* or post trauma*) adj3 (stress* or disorder* or psych* or symptom?)) or acute stress disorder* or combat disorder* or war neuros*).ti,ab,kf. 3 (((acute or traumatic) adj stress*) and (expos* or psyc*)).ti,ab,kf. 4 (traumati#ed adj (victim? or survivor?)).ti,ab,kf. 5 (trauma* adj2 (event? or memor* or flashback* or nightmare?)).ti,ab,kf. 6 ((trauma* or posttrauma* or post‐trauma* or victim* or survivor?) and (exposure adj3 (therap* or psychotherap* or training or counsel*))).ti,ab,kf. 7 or/1‐6 8 (((internet or web or online) adj3 (cognitive or behavio*)) or iCBT or i‐CBT or ePsych* or e‐Psych* or cCBT or c‐CBT).ti,ab,kf. 9 (android or app or apps or blog* or CD‐ROM or cell phone or cellphone or chat room or computer* or cyber* or digital or technology based or DVD or eHealth or e‐health or electronic health or e‐mail* or email* or e‐Portal or ePortal or eTherap* or e‐therap* or forum* or gaming or information technolog* or instant messag* or messaging or internet* or ipad or i‐pad or iphone or i‐phone or ipod or i‐pod or podcast or smart phone or smartphone or social network* site* or social networking or mHealth or m‐health or mobile or multi‐media or multimedia or online* or on‐line or personal digital assistant or PDA or SMS or social medi* or software or telecomm* or telehealth* or telemed* or telemonitor* or telepsych* or teletherap* or tele‐health* or tele‐med* or tele‐monitor* or tele‐psych* or tele‐therap* or text messag* or texting or virtual* or web* or WWW).ti,ab,kf,hw. 10 computer communication networks/ or internet/ or blogging/ or social media/ 11 cell phones/ or smartphone/ or text messaging/ or videoconferencing/ or webcasts as topic/ or wireless technology/ 12 (telecomm* or tele‐comm*).ti,ab,kf. 13 Telemedicine/ 14 (eLearning or blended learning).ti,kf. 15 (videoconferenc* or video conferenc*).ti,kf. 16 (synchronous or asynchronous or (electronic adj2 deliver*)).ti,kf. 17 or/9‐16 18 (behavio* or cognitive).ti. or (psychotherap* or psychological therap* or cognitive behavio* or ((cognitive or behavio*) adj2 (activat* or component? or defusion or modif* or restructur* or technique* or intervention or treatment* or therap* or train*)) or ((acceptance* or commitment*) adj3 therap*) or rational emotive or RET or problem sol* or PST or problem focus* or solution focus* or trauma focus* or psychoeducat* or psycho‐educat* or psychodrama or psycho‐drama* or mindfulness* or third wave or self‐control or (self* adj3 (control or efficacy)) or stress manage* or exposure or reality therap* or (anxiety adj3 (management or therap* or train*)) or relaxation or guided imagery or present cent* or person cent* or person* construct* or therapeutic process* or schema? or schemata or (thought* adj3 suppress*) or rumination).mp. 19 17 and 18 20 controlled clinical trial.pt. 21 randomized controlled trial.pt. 22 (randomi#ed or randomi#ation or randomi#ing).ti,ab,kf. 23 (RCT or at random or (random* adj3 (assign* or allocat* or control* or crossover or cross‐over or design* or divide* or division or number))).ti,ab,kf. 24 placebo*.ab,ti,kf. 25 trial.ab,ti,kf. 26 groups.ab. 27 (control* and (trial or study or group*) and (placebo or waitlist* or wait* list* or ((treatment or care) adj2 usual))).ti,ab,kf,hw. 28 ((single or double or triple or treble) adj2 (blind* or mask* or dummy)).ti,ab,kf. 29 double‐blind method/ or random allocation/ or single‐blind method/ 30 or/20‐29 31 exp animals/ not humans.sh. 32 30 not 31 33 7 and (8 or 19) and 32 34 (2018* or 2019* or 2020*).yr,dp,dt,ed,ep. 35 33 and 34 *************************** 2. Ovid Embase search strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 posttraumatic stress disorder/ 2 (PTSD or ((posttrauma* or post‐trauma* or post trauma*) adj3 (stress* or disorder* or psych* or symptom?)) or acute stress disorder* or combat disorder* or war neuros*).ti,ab,kw. 3 (((acute or traumatic) adj stress*) and (expos* or psyc*)).ti,ab,kw. 4 (traumati#ed adj (victim? or survivor?)).ti,ab,kw. 5 (trauma* adj2 (event? or memor* or flashback* or nightmare?)).ti,ab,kw. 6 ((trauma* or posttrauma* or post‐trauma* or victim* or survivor?) and (exposure adj3 (therap* or psychotherap* or training or counsel*))).ti,ab,kw. 7 or/1‐6 8 (((internet or web or online) adj3 (cognitive or behavio*)) or iCBT or i‐CBT or ePsych* or e‐Psych* or cCBT or c‐CBT).ti,ab,kw. 9 (android or app or apps or blog* or CD‐ROM or cell phone or cellphone or chat room or computer* or cyber* or digital or technology based or DVD or eHealth or e‐health or electronic health or e‐mail* or email* or e‐Portal or ePortal or eTherap* or e‐therap* or forum* or gaming or information technolog* or instant messag* or messaging or internet* or ipad or i‐pad or iphone or i‐phone or ipod or i‐pod or podcast or smart phone or smartphone or social network* site* or social networking or mHealth or m‐health or mobile or multi‐media or multimedia or online* or on‐line or personal digital assistant or PDA or SMS or social medi* or software or telecomm* or telehealth* or telemed* or telemonitor* or telepsych* or teletherap* or tele‐health* or tele‐med* or tele‐monitor* or tele‐psych* or tele‐therap* or text messag* or texting or virtual* or web* or WWW).ti,ab,kw,hw. 10 internet/ 11 blogging/ or e‐mail/ or social media/ or text messaging/ or videoconferencing/ or webcast/ or wireless communication/ 12 telecommunication/ or teleconference/ 13 telemedicine/ or telehealth/ or telepsychiatry/ or teletherapy/ 14 mobile phone/ or smartphone/ 15 mobile application.hw. 16 *technology/ 17 computer program/ or digital computer/ or personal computer/ or computer assisted therapy/ 18 *computer/ 19 (telecomm* or tele‐comm*).ti,ab,kw. 20 (eLearning or blended learning).ti,ab,kw. 21 (videoconferenc* or video conferenc*).ti,ab,kw. 22 (synchronous or asynchronous or (electronic adj2 deliver*)).ti,ab,kw. 23 or/9‐22 24 (behavio* or cognitive).ti. or (psychotherap* or psychological therap* or cognitive behavio* or ((cognitive or behavio*) adj2 (activat* or component? or defusion or modif* or restructur* or technique* or intervention or treatment* or therap* or train*)) or ((acceptance* or commitment*) adj3 therap*) or rational emotive or RET or problem sol* or PST or problem focus* or solution focus* or trauma focus* or psychoeducat* or psycho‐educat* or psychodrama or psycho‐drama* or mindfulness* or third wave or self‐control or (self* adj3 (control or efficacy)) or stress manage* or exposure or reality therap* or (anxiety adj3 (management or therap* or train*)) or relaxation or guided imagery or present cent* or person cent* or person* construct* or therapeutic process* or schema? or schemata or (thought* adj3 suppress*) or rumination).mp. 25 23 and 24 26 randomized controlled trial/ 27 randomization.de. 28 controlled clinical trial/ and (Disease Management or Drug Therapy or Prevention or Rehabilitation or Therapy).fs. 29 *clinical trial/ 30 placebo.de. 31 placebo.ti,ab. 32 trial.ti. 33 (randomi#ed or randomi#ation or randomi#ing).ti,ab,kw. 34 (RCT or "at random" or (random* adj3 (administ* or allocat* or assign* or class* or control* or determine* or divide* or division or distribut* or expose* or fashion or number* or place* or recruit* or subsitut* or treat*))).ti,ab,kw. (591088) 35 ((singl$ or doubl$ or trebl$ or tripl$) adj3 (blind$ or mask$ or dummy)).mp. 36 (control* and (trial or study or group) and (placebo or waitlist* or wait* list* or ((treatment or care) adj2 usual))).ti,ab,kw,hw. 37 or/26‐36 38 ((animal or nonhuman) not (human and (animal or nonhuman))).de. 39 37 not 38 40 7 and (8 or 25) and 39 41 (2018* or 2019* or 2020*).yr,dc,dd. 42 40 and 41 *************************** 3. Ovid PsycINFO search strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 posttraumatic stress disorder/ or complex ptsd/ or acute stress disorder/ or combat experience/ or “debriefing (psychological)"/ or emotional trauma/ or post‐traumatic stress/ or exp stress reactions/ or traumatic neurosis/ (50805) 2 exp DISASTERS/ 3 (PTSD or ((posttrauma* or post‐trauma* or post trauma*) adj3 (stress* or disorder* or psych* or symptom?)) or acute stress disorder* or combat disorder* or war neuros*).ti,ab,id. 4 (((acute or traumatic) adj stress*) and (expos* or psyc*)).ti,ab,id. 5 (traumati#ed adj (victim? or survivor?)).ti,ab,id. 6 (trauma* adj2 (event? or memor* or flashback* or nightmare?)).ti,ab,id. 7 ((trauma* or posttrauma* or post‐trauma* or victim* or survivor?) and (exposure adj3 (therap* or psychotherap* or training or counsel*))).ti,ab,id,hw. 8 or/1‐7 9 (((internet or web or online) adj3 (cognitive or behavio*)) or iCBT or i‐CBT or ePsych* or e‐Psych* or cCBT or c‐CBT).ti,ab,kf. 10 (android or app or apps or blog* or CD‐ROM or cell phone or cellphone or chat room or computer* or cyber* or digital or technology based or DVD or eHealth or e‐health or electronic health or e‐mail* or email* or e‐Portal or ePortal or eTherap* or e‐therap* or forum* or gaming or information technolog* or instant messag* or messaging or internet* or ipad or i‐pad or iphone or i‐phone or ipod or i‐pod or podcast or smart phone or smartphone or social network* site* or social networking or mHealth or m‐health or mobile or multi‐media or multimedia or online* or on‐line or personal digital assistant or PDA or SMS or social medi* or software or telecomm* or telehealth* or telemed* or telemonitor* or telepsych* or teletherap* or tele‐health* or tele‐med* or tele‐monitor* or tele‐psych* or tele‐therap* or text messag* or texting or virtual* or web* or WWW).ti,ab,id,hw. 11 (telecomm* or tele‐comm*).ti,ab,id. 12 (eLearning or blended learning).ti,ab,id. 13 (videoconferenc* or video conferenc*).ti,ab,id. 14 (synchronous or asynchronous or (electronic adj2 deliver*)).ti,ab,id. 15 internet/ or websites/ 16 mobile devices/ or cellular phones/ 17 social media/ or online social networks/ or blog/ or online community/ or text messaging/ 18 electronic communication/ or exp computer mediated communication/ or electronic learning/ 19 online therapy/ or telemedicine/ 20 telecommunications media/ 21 teleconferencing/ 22 technology/ or information technology/ or exp computer applications/ or computer software/ 23 computers/ or computer games/ or digital computers/ or microcomputers/ 24 or/10‐23 25 (behavio* or cognitive).ti. or (psychotherap* or psychological therap* or cognitive behavio* or ((cognitive or behavio*) adj2 (activat* or component? or defusion or modif* or restructur* or technique* or intervention or treatment* or therap* or train*)) or ((acceptance* or commitment*) adj3 therap*) or rational emotive or RET or problem sol* or PST or problem focus* or solution focus* or trauma focus* or psychoeducat* or psycho‐educat* or psychodrama or psycho‐drama* or mindfulness* or third wave or self‐control or (self* adj3 (control or efficacy)) or stress manage* or exposure or reality therap* or (anxiety adj3 (management or therap* or train*)) or relaxation or guided imagery or present cent* or person cent* or person* construct* or therapeutic process* or schema? or schemata or (thought* adj3 suppress*) or rumination).ti,ab,id,hw. 26 (self adj (care or change or guide* or help or intervention or manag* or support* or train*)).ti,id. 27 24 and (25 or 26) 28 clinical trials.sh. 29 (randomi#ed or randomi#ation or randomi#ing).ti,ab,id. 30 (RCT or at random or (random* adj3 (assign* or allocat* or control* or crossover or cross‐over or design* or divide* or division or number))).ti,ab,id. 31 (control* and (trial or study or group) and (placebo or waitlist* or wait* list* or ((treatment or care) adj2 usual))).ti,ab,id,hw. 32 ((single or double or triple or treble) adj2 (blind* or mask* or dummy)).ti,ab,id. 33 trial.ti. 34 placebo.ti,ab,id,hw. 35 treatment outcome.md. 36 treatment effectiveness evaluation.sh. 37 mental health program evaluation.sh. 38 or/28‐37 39 8 and (9 or 27) and 38 40 (2018*or 2019* or 2020*).yr,an. 41 39 and 40 *************************** 4. PILOTS: Published International Literature on Traumatic Stress PILOTS was searched via ProQuest, using the following terms (RCT filter + Intervention only):
S1. su(randomized clinical trial) S2. ti,ab(randomised OR randomized OR randomisation OR randomization) S3. ti,ab(random* NEAR/2 (allocat* OR assign* OR divid*)) S4. ti,ab((waitlist* OR "wait* list") NEAR/4 (control OR group)) S5. ti,ab(“treatment as usual” OR tau) S6. ti,ab(“care as usual” OR cau) S7. (S1 OR S2 OR S3 OR S4 OR S5 OR S6) S8. su(self help techniques) S9. ti,ab(self NEAR/3 (care OR change OR guid* OR help OR intervention OR manag* OR support* OR train*)) S10. su(computer assisted psychotherapy) S11. ti,ab(android OR app OR apps OR audio* OR blog OR iCBT OR i‐CBT OR cCBT OR c‐CBT OR cdrom OR "cell phone" OR cellphone OR chat OR computer* OR cyber* OR distance* OR DVD OR e‐health OR eHealth OR "electronic health*" OR e‐Portal OR eportal OR eTherap* OR e‐therap* OR forum* OR gaming OR "information technolog*" OR "instant messag*" OR internet* OR interapy OR ipad OR i‐pad OR i‐phone OR ipod OR i‐pod OR web* OR WWW OR "smart phone" OR smartphone OR "mobile phone" OR email* OR email* OR mhealth OR m‐health OR mobile OR multimedia OR multimedia OR online* OR online OR "personal digital assistant" OR PDA OR SMS OR "social medi*" OR Facebook OR software OR telecare OR telecom* OR telehealth* OR telemed* OR telemonitor* OR telepsych*or teletherap* OR "text messag*" OR texting OR tape OR taped OR video* OR YouTube OR podcast OR virtual* OR remote) S12. (S8 OR S9 OR S10 OR S11) S13. (S7 AND S12) Date limited: 01/03/2018 to 05/06/2020 [Key to database field tags: ab:abstract; ti:title; su:subject headings] *************************** 5. Cochrane Central Register of Controlled Trials (CENTRAL) Issue 6 2020
#1 MESH DESCRIPTOR Stress Disorders, Post‐Traumatic #2 MESH DESCRIPTOR Stress Disorders, Traumatic #3 MESH DESCRIPTOR Combat Disorders #4 MESH DESCRIPTOR Stress Disorders, Traumatic, Acute #5 ((posttrauma* or post‐trauma* or post trauma*) adj3 (stress* or disorder* or psych* or symptom*)):TI,AB,KY2809 #6 PTSD:TI,AB,KY #7 ( "acute stress disorder*" or "combat disorder*" or "war neuros*"):TI,AB,KY #8 (#1 OR #2 OR #3 OR #4 OR #5 OR #6 OR #7) #9 ((android or app or apps or audio* or blog or iCBT or cCBT or i‐CBT or c‐CBT or CD‐ROM or ?cell phone? or cellphone or chat or computer* or cyber* or distance* or DVD or eHealth or e‐health or "electronic health*" or e‐Portal or ePortal or eTherap* or e‐therap* or forum* or gaming or ?information technolog*? or "instant messag*" or internet* or interapy or ipad or i‐pad or iphone or i‐phone or ipod or i‐pod or web* or WWW or "smart phone" or smartphone or ?mobile phone? or e‐mail* or email* or mHealth or m‐health or mobile or multi‐media or multimedia or online* or on‐line or?personal digital assistant? or PDA or SMS or "social medi*" or Facebook or software or telecomm* or telehealth* or telemed* or telemonitor* or telepsych*or teletherap* or "text messag*" or texting or tape or taped or video* or YouTube or podcast or virtual* or remote)):TI,AB,KY AND 01/03/2018 TO 05/06/2020:DL #10 ((self near3 (care or change or guide* or help or intervention or manag* or support* or train*))):TI,AB,KY AND 01/03/2018 TO 05/06/2020:DL #11 (#9 OR #10) #12 (#8 AND #11) *************************** 7. International Trial Registers ClinicalTrials.gov (all years to 5 June 2020)
internet OR web OR online OR iCBT | PTSD OR POSTTRAUMATIC STRESS DISORDER OR POST TRAUMATIC STRESS DISORDER WHO International Clinical Trials Registry Platform (ICTRP) to March 2018 only PTSD AND internet OR PTSD AND web OR PTSD AND online OR PTSD AND iCBT OR POSTTRAUMATIC STRESS DISORDER AND internet OR POSTTRAUMATIC STRESS DISORDER AND web OR POSTTRAUMATIC STRESS DISORDER AND online OR POSTTRAUMATIC STRESS DISORDER AND iCBT OR POST TRAUMATIC STRESS DISORDER AND internet OR POST TRAUMATIC STRESS DISORDER AND web OR POST TRAUMATIC STRESS DISORDER AND online OR POST TRAUMATIC STRESS DISORDER AND iCBT ***************************
Appendix 2. Specialised Register
Cochrane Common Mental Disorders Controlled Trials Register (CCMDCTR)
Cochrane Common Mental Disorders (CCMD) maintains two archived clinical trials registers at its editorial base in York (UK): a References Register and a Studies‐based Register. The CCMDCTR‐References Register contains over 40,000 reports of randomised controlled trial (RCTs) in depression, anxiety, and neurosis. Approximately 50% of these references have been tagged to individual, coded trials. The coded trials are held in the CCMDCTR‐Studies‐based Register and records are linked between the two registers using unique Study ID tags. Coding of trials is based on the EU‐Psi coding manual, using a controlled vocabulary (contact the CCMD Information Specialists for further details). Reports of trials for inclusion in the Group's registers are collated from routine (weekly), generic searches of MEDLINE (1950 to 2016), Embase (1974 to 2016), and PsycINFO (1967 to 2016); quarterly searches of the Cochrane Central Register of Controlled Trials (CENTRAL); and review‐specific searches of additional databases. Reports of trials are also sourced from international trial registers via the World Health Organization's trials portal (the International Clinical Trials Registry Platform (apps.who.int/trialsearch/)), pharmaceutical companies, and the handsearching of key journals, conference proceedings, and other (non‐Cochrane) systematic reviews and meta‐analyses.
Details of CCMD's generic search strategies (used to identify RCTs) can be found on the Group's website (cmd.cochrane.org/specialised-register), with an example of the core MEDLINE search (used to inform the register) listed below. The Group's Specialised Register became out of date following the Editorial Group's move from Bristol to York in the summer of 2016.
Core search strategy used to inform the Cochrane Common Mental Disorders Group's Specialised Register: Ovid MEDLINE (to June 2016)
A weekly search alert based on condition + RCT filter only 1. [MeSH Headings]: eating disorders/ or anorexia nervosa/ or binge‐eating disorder/ or bulimia nervosa/ or female athlete triad syndrome/ or pica/ or hyperphagia/ or bulimia/ or self‐injurious behavior/ or self mutilation/ or suicide/ or suicidal ideation/ or suicide, attempted/ or mood disorders/ or affective disorders, psychotic/ or bipolar disorder/ or cyclothymic disorder/ or depressive disorder/ or depression, postpartum/ or depressive disorder, major/ or depressive disorder, treatment‐resistant/ or dysthymic disorder/ or seasonal affective disorder/ or neurotic disorders/ or depression/ or adjustment disorders/ or exp antidepressive agents/ or anxiety disorders/ or agoraphobia/ or neurocirculatory asthenia/ or obsessive‐compulsive disorder/ or obsessive hoarding/ or panic disorder/ or phobic disorders/ or stress disorders, traumatic/ or combat disorders/ or stress disorders, post‐traumatic/ or stress disorders, traumatic, acute/ or anxiety/ or anxiety, castration/ or koro/ or anxiety, separation/ or panic/ or exp anti‐anxiety agents/ or somatoform disorders/ or body dysmorphic disorders/ or conversion disorder/ or hypochondriasis/ or neurasthenia/ or hysteria/ or munchausen syndrome by proxy/ or munchausen syndrome/ or fatigue syndrome, chronic/ or obsessive behavior/ or compulsive behavior/ or behavior, addictive/ or impulse control disorders/ or firesetting behavior/ or gambling/ or trichotillomania/ or stress, psychological/ or burnout, professional/ or sexual dysfunctions, psychological/ or vaginismus/ or Anhedonia/ or Affective Symptoms/ or *Mental Disorders/ 2. [Title/ Author Keywords]: (eating disorder* or anorexia nervosa or bulimi* or binge eat* or (self adj (injur* or mutilat*)) or suicide* or suicidal or parasuicid* or mood disorder* or affective disorder* or bipolar i or bipolar ii or (bipolar and (affective or disorder*)) or mania or manic or cyclothymic* or depression or depressive or dysthymi* or neurotic or neurosis or adjustment disorder* or antidepress* or anxiety disorder* or agoraphobia or obsess* or compulsi* or panic or phobi* or ptsd or posttrauma* or post trauma* or combat or somatoform or somati#ation or medical* unexplained or body dysmorphi* or conversion disorder or hypochondria* or neurastheni* or hysteria or munchausen or chronic fatigue* or gambling or trichotillomania or vaginismus or anhedoni* or affective symptoms or mental disorder* or mental health).ti,kf. 3. [RCT filter]: (controlled clinical trial.pt. or randomized controlled trial.pt. or (randomi#ed or randomi#ation).ab,ti. or randomly.ab. or (random* adj3 (administ* or allocat* or assign* or class* or control* or determine* or divide* or distribut* or expose* or fashion or number* or place* or recruit* or subsitut* or treat*)).ab. or placebo*.ab,ti. or drug therapy.fs. or trial.ab,ti. or groups.ab. or (control* adj3 (trial* or study or studies)).ab,ti. or ((singl* or doubl* or tripl* or trebl*) adj3 (blind* or mask* or dummy*)).mp. or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or randomized controlled trial/ or pragmatic clinical trial/ or (quasi adj (experimental or random*)).ti,ab. or ((waitlist* or wait* list* or treatment as usual or TAU) adj3 (control or group)).ab.) 4. (1 and 2 and 3) Records are screened for reports of RCTs within the scope of the Cochrane Common Mental Disorders Group. Secondary reports of RCTs are tagged to the appropriate study record. Similar weekly search alerts are also conducted on OVID Embase and PsycINFO, using relevant subject headings (controlled vocabularies) and search syntax, appropriate to each resource.
CCMDCTR search for this review
The registered was searched for this review (all years to June 2016, only) using the following terms:
1. The CCMDCTR‐Studies Register: Condition = (PTSD or *trauma* or “acute stress” or “stress reaction”) AND Intervention = (computer* or internet or web* or online or self‐help or self‐manage* or self‐change)
2. The CCMDCTR‐References Register was searched using a more sensitive set of terms to identify additional untagged or uncoded reports of RCTs: #1. (PTSD or *trauma* or “combat disorder*” or “stress reaction” or “acute stress” or "stress disorder" or "war neurosis"):ab,ti,kw,ky,emt,mh,mc #2. (self near3 (care or change or guide* or help or intervention or manag* or support* or train*)):ab,ti,kw,ky,emt,mh,mc #3. (android or app or apps or audio* or blog or iCBT or cCBT or i‐CBT or c‐CBT or CD‐ROM or “cell phone” or cellphone or chat or computer* or cyber* or distance* or DVD or eHealth or e‐health or "electronic health*" or e‐Portal or ePortal or eTherap* or e‐therap* or forum* or gaming or “information technolog*” or "instant messag*" or internet* or interapy or ipad or i‐pad or iphone or i‐phone or ipod or i‐pod or web* or WWW or "smart phone" or smartphone or “mobile phone” or e‐mail* or email* or mHealth or m‐health or mobile or multi‐media or multimedia or online* or on‐line or “personal digital assistant” or PDA or SMS or "social medi*" or Facebook or software or telecomm* or telehealth* or telemed* or telemonitor* or telepsych*or teletherap* or "text messag*" or texting or tape or taped or video* or YouTube or podcast or virtual* or remote):ab,ti,kw,ky,emt,mh,mc #4. (#1 and (#2 or #3)) [Key to CRS field tags: ab: abstract; ti: title; kw: CRG keywords; ky: other keywords; emt: EMTREE headings; mh: MeSH headings; mc: MeSH checkwords]
Data and analyses
Comparison 1. Internet‐based cognitive and behavioural therapy (I‐C/BT) versus face‐to‐face non‐CBT.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Severity of PTSD symptoms (post‐treatment) | 1 | 40 | Mean Difference (IV, Random, 95% CI) | 10.90 [6.57, 15.23] |
| 1.2 Severity of PTSD symptoms (follow‐up < 6 months) | 1 | 40 | Mean Difference (IV, Random, 95% CI) | 5.67 [1.56, 9.78] |
| 1.3 Dropouts | 1 | 40 | Risk Ratio (M‐H, Random, 95% CI) | 2.49 [0.91, 6.77] |
Comparison 2. Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 2.1 Severity of PTSD symptoms (post‐treatment) | 10 | 608 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.61 [‐0.93, ‐0.29] |
| 2.2 Severity of PTSD symptoms (follow‐up < 6 months) | 4 | 154 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.45 [‐1.29, 0.39] |
| 2.3 Dropouts | 9 | 634 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.25 [0.97, 1.60] |
| 2.4 Diagnosis of PTSD after treatment | 1 | 62 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.53 [0.28, 1.00] |
| 2.5 Severity of depression (post‐treatment) | 7 | 473 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.51 [‐0.97, ‐0.06] |
| 2.6 Severity of depression (follow‐up < 6 months) | 2 | 50 | Mean Difference (IV, Fixed, 95% CI) | ‐8.75 [‐14.71, ‐2.80] |
| 2.7 Severity of anxiety symptoms (post‐treatment) | 5 | 345 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.61 [‐0.89, ‐0.33] |
| 2.8 Severity of anxiety symptoms (follow‐up < 6 months) | 1 | 42 | Mean Difference (IV, Fixed, 95% CI) | ‐12.59 [‐20.74, ‐4.44] |
| 2.9 Quality of life (post‐treatment) | 3 | 229 | Std. Mean Difference (IV, Random, 95% CI) | 0.59 [0.18, 1.01] |
| 2.10 Quality of life (6 months) | 1 | 8 | Std. Mean Difference (IV, Random, 95% CI) | 0.16 [‐1.28, 1.59] |
2.10. Analysis.

Comparison 2: Internet‐based cognitive and behavioural therapy (I‐C/BT) versus wait list (WL), Outcome 10: Quality of life (6 months)
Comparison 3. Internet‐based cognitive and behavioural therapy (I‐C/BT) versus I‐non‐C/BT.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 3.1 Severity of PTSD symptoms (post‐treatment) | 2 | 82 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.08 [‐0.52, 0.35] |
| 3.2 Severity of PTSD symptoms (follow‐up < 6 months) | 2 | 65 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.08 [‐0.41, 0.57] |
| 3.3 Severity of PTSD symptoms (follow‐up 6–12 months) | 1 | 18 | Mean Difference (IV, Fixed, 95% CI) | ‐8.83 [‐17.32, ‐0.34] |
| 3.4 Dropouts | 2 | 132 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.14 [0.97, 4.73] |
| 3.5 Severity of depressive symptoms (post‐treatment) | 2 | 84 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.12 [‐0.78, 0.54] |
| 3.6 Severity of depressive symptoms (follow‐up < 6 months) | 2 | 61 | Std. Mean Difference (IV, Fixed, 95% CI) | 0.20 [‐0.31, 0.71] |
| 3.7 Severity of depressive symptoms (follow‐up 6–12 months) | 1 | 18 | Mean Difference (IV, Fixed, 95% CI) | ‐8.34 [‐15.83, ‐0.85] |
| 3.8 Severity of anxiety symptoms (post‐treatment) | 2 | 74 | Std. Mean Difference (IV, Random, 95% CI) | 0.08 [‐0.78, 0.95] |
| 3.9 Severity of anxiety symptoms (follow‐up < 6 months) | 2 | 60 | Std. Mean Difference (IV, Fixed, 95% CI) | ‐0.16 [‐0.67, 0.35] |
| 3.10 Severity of anxiety symptoms (follow‐up 6–12 months) | 1 | 18 | Mean Difference (IV, Fixed, 95% CI) | ‐8.05 [‐15.20, ‐0.90] |
Comparison 4. Subgroup analysis: guided versus non‐guided.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 4.1 Severity of PTSD symptoms (post‐treatment) | 10 | 608 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.61 [‐0.93, ‐0.29] |
| 4.1.1 Guided | 8 | 439 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.78 [‐1.09, ‐0.47] |
| 4.1.2 Non‐guided | 2 | 169 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.09 [‐0.39, 0.22] |
| 4.2 Dropouts | 9 | 644 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [0.90, 1.90] |
| 4.2.1 Guided | 7 | 475 | Risk Ratio (M‐H, Random, 95% CI) | 1.22 [0.82, 1.81] |
| 4.2.2 Non‐guided | 2 | 169 | Risk Ratio (M‐H, Random, 95% CI) | 1.95 [0.83, 4.57] |
Comparison 5. Subgroup analysis: trauma‐focused versus non‐trauma‐focused.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 5.1 Severity of PTSD (post‐treatment) | 10 | 608 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.61 [‐0.93, ‐0.29] |
| 5.1.1 Trauma‐focused | 6 | 342 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.94 [‐1.24, ‐0.65] |
| 5.1.2 Non‐trauma focused | 4 | 266 | Std. Mean Difference (IV, Random, 95% CI) | ‐0.16 [‐0.41, 0.08] |
| 5.2 Dropouts | 9 | 644 | Risk Ratio (M‐H, Random, 95% CI) | 1.31 [0.90, 1.90] |
| 5.2.1 Trauma‐focused | 5 | 343 | Risk Ratio (M‐H, Random, 95% CI) | 1.22 [0.75, 1.98] |
| 5.2.2 Non‐trauma focused | 4 | 301 | Risk Ratio (M‐H, Random, 95% CI) | 1.78 [0.94, 3.37] |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Allen 2020.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 49 Diagnosis: DSM‐IV PTSD Method of recruitment: all participants were self‐referred and recruited via advertisements posted on social media websites, online forums, and flyers. Method of diagnosis: PCL‐C, LEC Trauma type: fire or natural disaster, serious accident (e.g. involving transportation or occurring at home/during a recreational activity), toxic substance(s) exposures, unwanted sexual contact or a sexual assault, assault with a weapon, abduction, combat or a warzone, life‐threatening illness or injury, severe human suffering, sudden violent death of loved ones or others Age (mean): intervention: 41.9 (SD 14.45) years; wait list: 41.26 (SD 13.45) years Sex: intervention: 90.5% women; wait list 89.5% women Location: Australia Comorbidities: MDD at baseline: intervention: 62%; wait list: 74% Adjunctive therapy: not reported Adjunctive medication: not reported Unemployment: intervention: 38.1%; wait list: 26.3% University education: intervention: 47.64%; wait list: 21.05% Exclusion criteria: any of the following: trauma within past 4 weeks; non‐resident of Australia; aged < 18 years; currently receiving treatment for PTSD; frequent suicidal ideation (indicated by a score of 3 on item 9 of the PHQ‐9); regular use of illicit drugs or > 3 standard drinks per day; psychotic disorder or taking antipsychotic medication or benzodiazepines; started medication for anxiety or depression within last 4 weeks or intention to change dose during trial period; highly dissociative (indicated by score ≥ 40 on the Dissociative Experiences Scale; current or pending medicolegal proceedings associated with the trauma; applying for/receiving Workers Compensation associated with the trauma Baseline PTSD: all participants met diagnostic criteria for PTSD Baseline PCL scores (mean): intervention: 59.29 (SD 13.75); wait list: 61.05 (SD 13.76) |
|
| Interventions |
Internet program based on trauma‐focused CBT vs wait list Experimental arm Duration: 10 weeks (6 online sessions) Treatment protocol: content was presented in the form of an illustrated story in which 1 character, experiencing PTSD, learned to self‐manage symptoms with the help of a psychologist. Each lesson consisted of a set of lesson slides, followed by a lesson summary and action plan (including regular homework tasks), and a range of supplementary resources. All lesson material was completed independently by the participant. Participants were able to access 1 lesson per week, with a minimum waiting period of 5 days between lessons. This waiting period was to ensure participants spent time revising lesson content and implementing skills before moving to the next lesson. While 3–4 hours was the recommended weekly commitment per lesson, participants could choose the duration they spent on each lesson and skills practice. Participants were encouraged to stay on schedule via automated email reminders, with support also provided via email or telephone by the study clinician, which involved assisting with understanding the lesson content and implementing the skills. Therapist contact: therapeutic guidance in the form of emails, including automated emails. Study personnel spent a mean of 38.00 minutes (SD 67.29) per participant on email and telephone calls. There was a mean of 9.76 (SD 6.72) email exchanges per participant and a mean of 4.05 (SD 4.88) telephone calls. 3 participants required a higher level of support (> 30 minutes) for assistance with managing ongoing psychosocial stress, suicide risk, and self‐harm risk, and additional skills coaching. Type of contact: participants were provided with therapeutic guidance in the form of automated emails at the conclusion of each lesson, in response to participant request for contact, or if participant symptoms deteriorated based on scores on standardised measures. Support also provided via email or telephone by the study clinician, which involved assisting with understanding the lesson content and implementing the skills. Automated contact: participants were encouraged to stay on schedule via automated email reminders. Type of therapist: not reported Treatment fidelity: not reported Comparator arm Duration: 10 weeks Treatment protocol: wait list |
|
| Outcomes |
Time points for assessment: pretreatment, post‐treatment, 3 months Primary outcomes: PTSD (PCL‐C) Secondary outcomes: Psychological distress (K10), Depression (PHQ‐9), Anxiety (GAD‐7) |
|
| Notes |
Funding source: unclear Declarations of interest among the primary researchers: unclear |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Allocation was generated by an independent person using a random number generator (www.random.org)." |
| Allocation concealment (selection bias) | Low risk | Quote: "Allocation was generated by an independent person using a random number generator (www.random.org), and concealed from the investigators using sequentially numbered opaque envelopes which were assigned manually (by the study coordinator) to each participant." |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Not enough information to permit judgement. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | 49 participants were randomised; however, ITT analysis was reported for only 40 participants. |
| Selective reporting (reporting bias) | Unclear risk | The main outcome measure and most secondary outcome measures noted in the study protocol paper were reported; however, some secondary outcome measures were missing. |
| Other bias | High risk | Intervention evaluated by originators of the intervention. Small sample size. |
Bragesjö 2020.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 8 (subset of participants who met eligibility criteria for inclusion in the review) Diagnosis: DSM‐5 PTSD Method of recruitment: advertisements in newspapers, webpages, and social media; and at primary care, psychiatric, and emergency clinics throughout Sweden Method of diagnosis: PCL‐5 Trauma type: various Age: demographics for the subset of data included in this review not available Sex: demographics for the subset of data included in the review not available Location: Sweden Comorbidities: other serious psychiatric comorbidity (e.g. ongoing substance dependence, untreated bipolar disorder, psychotic symptoms, severe depression, borderline personality disorder, and high suicide risk according to the MINI) Adjunctive therapy: not reported Adjunctive medication: adjunctive medication information for the subset of data included in this review not available Unemployment: unemployment information for the subset of data included in the review not available University education: university education information for the subset of data included in the review not available Exclusion criteria: other serious comorbidity as primary concern (ongoing substance dependence, untreated positive screening for bipolar disorder, psychotic disorder, ongoing substance disorder, or severe suicidality according to MINI 7.0.0); currently receiving CBT for trauma‐related reactions; ongoing trauma‐related threat (e.g. living with a violent spouse). Participants receiving psychotropic medication had to have a stable dose for 1 month prior to inclusion in study. Baseline PTSD: all participants in this subset met diagnostic criteria for PTSD. Baseline PCL‐5 scores (mean): Internet CBT: 54.33 (SD 13.05); wait list: 50.80 (SD 17.60) |
|
| Interventions |
Experimental arm I‐PE Duration: 3 weeks Treatment protocol: active Internet‐based treatment delivered on a safe Internet platform. Treatment divided into 4 modules which the participant gained access to sequentially after completing homework exercises. The I‐PE intervention is a condensed PE protocol adapted to an Internet‐delivered format. All text‐based material was available in an audio‐file format, for flexible access. First module included information about the I‐PE treatment and psychoeducation about common reactions after experiencing psychological trauma. Controlled breathing, as a stress control method, was introduced. Second module included imaginal exposure, where the participant was instructed to approach and process the memory of the traumatic event, either writing it down on paper or recording a verbal account, and revisiting this narrative daily. Third module included information on how to approach the most distressing parts of the memory and how to deal with trauma hot spots, and participants were encouraged to gradually approach safe or low‐risk situations that they had avoided since the traumatic event (exposure in vivo). Fourth module included a summary of the treatment, and the participant was asked to make a relapse‐prevention plan for themselves. Therapist contact: participants were assigned a therapist who they could contact through email within the platform and expect answer within 24 hours on weekdays. Therapists were instructed to guide the participant through the treatment, answer queries, and provide support and encouragement on the progress made. The therapist also sent reminders or in some cases called the participants if they did not log in to the platform for 3 days or were late with submitting homework exercises. The participants were encouraged to have daily contact with their therapist to make full use of the 3‐week treatment period. The researchers report the mean the participant contacted their therapist every other day. Type of contact: email through the online platform. Automated contact: automatic reminders were sent out if participants did not register their intrusive memory diary data. Type of therapist: 3 clinical psychologists with experience in the treatment of PTSD, all of whom had received a short training in the PE protocol used in this study. All could access supervision. Comparator arm Wait list |
|
| Outcomes |
Time points for assessment: post‐treatment and 6 months Primary outcomes: daily occurrence of recurrent, involuntary, and intrusive recollections of the traumatic event or flashback memories was assessed with a smartphone app Secondary outcomes: PTSD (PCL‐5), Depression (MADRS‐S), Quality of Life (EQ‐5D) |
|
| Notes |
Funding source: The Swedish Research Council (grant 2016‐02359), the Swedish Society for Medicine (grant 658811), and Stockholm County Healthcare (grant 20170018). Declarations of interest among the primary researchers: none |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Participants were consecutively randomised (no constraints) by an independent party (using www.random.org) in a 1:1 ratio." |
| Allocation concealment (selection bias) | Unclear risk | Quote: "Participants were allocated to one of three therapists." Comment: it was unclear who was instructing this allocation. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: outcome measures were self‐reported. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "The reason for using mixed effects models was due to their effectiveness in handling missing data as well as in reducing the risk of committing type I errors." |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but it was clear that the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | High risk | As only a subset of data was been included, i.e. those participants who met diagnostic PTSD criteria, the sample size was very small. It is unclear if the researchers evaluating the intervention were developers of the intervention itself. |
Engel 2015.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 80 Diagnosis: DSM‐IV PTSD Method of recruitment: adverts in local and national newspapers Method of diagnosis: CAPS Trauma type: military Age (mean): intervention: 36.2 (SD 7.75) years; usual care: 36.7 (SD 9.75) years Sex: 18.75% women; 81.25% men Location: US Comorbidities: no structured screening for comorbid conditions Adjunctive therapy: excluded Adjunctive medication: stable Unemployment: not reported University education: 62.8% Exclusion criteria: active engagement in trauma‐focused mental health treatment in previous 2 months; recent history of failed speciality mental health treatment for PTSD or associated condition; acute psychosis, psychotic episode, or psychotic disorder diagnosed within the past 2 years; active substance dependence in past year; active suicidal or homicidal ideation within past 2 months; currently taking antipsychotic or mood‐stabilising medication; unstable administration schedule or dosing of any antidepressant, anxiolytic, or sedative‐hypnotic during last month; or acute or unstable physical illness Baseline PTSD: all participants met diagnostic criteria for PTSD Baseline PCL scores (mean): intervention: 58.00 (SD 9.95); usual care: 54.48 (SD 11.23) |
|
| Interventions |
Internet program based on trauma‐focused CBT vs TAU Experimental arm Duration: 6 weeks (with access for 8 weeks, or 10 in exceptional circumstances) Treatment protocol
Therapist contact: participants encouraged to contact nurse for assistance if needed. Nurses were able to access a private portion of the DESTRESS‐PC website where they could monitor compliance and symptom levels. Type of contact: participants contacted nurse for assistance if needed. Nurse had access to a private portion of the DESTRESS‐PC website to monitor compliance and symptom levels. Automated contact: none Type of therapist: nurse Treatment fidelity: unclear Comparator arm Duration: 6 weeks
|
|
| Outcomes |
Time points for assessment: pretreatment, post‐treatment, 6 weeks, 12 weeks, 18 weeks Primary outcomes: PTSD (PCL‐C); Depression (PHQ‐8); Somatic symptoms (PHQ‐15) |
|
| Notes |
Funding source: unclear Declarations of interest among the primary researchers: unclear |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Randomization was performed centrally from Walter Reed's Deployment Health Clinical Center with subsequent notification of the site investigator and the site's DESTRESS Nurse. Stratification of randomization by healthcare site and gender was done to facilitate exploratory analyses on trial outcomes [20]; and employed a random permuted blocking scheme (1:1 allocation ratio with variable block size of four to six) [21] was employed." |
| Allocation concealment (selection bias) | Unclear risk | Not reported. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "the nature of the intervention precluded blinding of providers and participants." |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "we blinded the outcome raters in this trial." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "Because the PROC MIXED procedure uses all of the available data and restricted maximum likelihood to estimate the unknown variance‐covariance parameters, it ensures a robust handling of missing follow‐up data [29]. We examined the effects of time, treatment group, and the treatment group by time interaction on the primary outcome (PCL scores) for the entire sample and for the two types of recruitment sites (VA, DoD) separately." 14/80 participants dropped out. |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but clear that published reports included all expected outcomes, including those that were prespecified. |
| Other bias | Low risk | Study appeared free from other sources of bias. |
Ivarsson 2014.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 62 Diagnosis: DSM‐IV PTSD Method of recruitment: advertisements in local and national newspapers Method of diagnosis: CAPS by telephone Trauma type: various (apart from childhood abuse) Age (mean): immediate treatment: 44.8 (SD 11.2) years; delayed treatment: 47.2 (SD 12.2) years Sex: 82.3% women; 17.7% men Location: Sweden Comorbidities: no structured screening for comorbid conditions Adjunctive therapy: excluded Adjunctive medication: 25.8% (stable for 3 months) Unemployment: 8.1% University education: 56.5% Exclusion criteria: imminent suicide risk; concurrent psychological treatment; presence of alcohol abuse; ongoing trauma or trauma within the past 3 months; symptoms following childhood abuse. Baseline PTSD: all participants met diagnostic criteria for PTSD Baseline IES‐R scores (mean): immediate treatment: 54.65 (SD 13.16); delayed treatment: 54.87 (SD 15.48) |
|
| Interventions |
Internet program based on trauma‐focused CBT vs TAU Experimental arm Duration: 8 weeks Treatment protocol
Therapist contact: once a week and occasional reminders via website. Type of contact: encrypted web‐service Automated contact: none Type of therapist: clinical psychology students (towards the end of course) Treatment fidelity: weekly supervision with an experienced clinical psychologist to monitor fidelity Comparator arm Duration: 8 weeks Participants in treatment group who did not finish all modules within 8 weeks were able to continue using the website without therapist support after post‐treatment data had been collected. Treatment protocol
Therapist contact: kept to a minimum. Therapist screened responses for suicidal ideation and was available to answer questions on the trial. |
|
| Outcomes |
Time points for assessment: pretreatment, post‐treatment, 1 year Primary outcome: PTSD (PDS), Impact of Events (IES‐R) Secondary outcome: Depression (BDI‐II), Anxiety (BAI), Quality of Life (QOLI), Clinical Global Improvement (CGI‐I) |
|
| Notes |
Funding source: grant from Linköping University Declarations of interest among primary researchers: unclear |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Randomization was conducted by an individual who was not otherwise involved in the research project, using an online true random number service (www.random.org)." |
| Allocation concealment (selection bias) | Unclear risk | Quote: "Randomization was conducted by an individual who was not otherwise involved in the research project, using an online true random number service (www.random.org)." |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "The post‐treatment interviewers were blind to participant status (i.e. treatment or control)." Comment: blinding impossible at 1‐year follow‐up since there was no longer a control group. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "All analyses, both the pre‐ to post‐treatment and the pre‐treatment to one year follow‐up analysis, made use of all available data from all randomized participants, following the principle of intention‐to‐treat. Prior to conducting primary analyses, the missing data assumption was tested by exploring associations between baseline characteristics and the presence of missing data." |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but it was clear that the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | High risk | Evaluated by the originators of the intervention. |
Knaevelsrud 2015.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 159 Diagnosis: DSM‐IV PTSD Method of recruitment: advertisements and presentations at department of defence sites Method of diagnosis: PDS used to identify if participants reported the minimum number of symptoms required by DSM‐IV for each of the symptom clusters (≥ 1 intrusion, 3 avoidance, and 2 hyperarousal symptoms). Minimum score of 11 on the PDS required Trauma type: war related Age (mean): interapy: 29.11 (SD 8.20) years; wait list: 27.15 (SD 6.48) years Sex: interapy: 60 women (79%); wait list: 55 women (69%) Location: Iraq Comorbidities: unclear Adjunctive therapy: excluded Adjunctive medication: unclear Unemployment: interapy: 26 (33%); wait list: 29 (36%) University education: interapy: 56 (71%); wait list: 38 (48%) Exclusion criteria: currently receiving treatment elsewhere; substance abuse or dependence; high risk of suicide; psychotic symptoms; low symptom severity Baseline PDS scores (mean): interapy: 30.87 (SD 8.13); wait list: 31.81 (SD 7.13) |
|
| Interventions |
Internet programme based on trauma‐focused CBT vs wait list Experimental arm Interapy Duration: 5 weeks Treatment protocol
Type of contact: short homework reminder by email, if no response contacted by telephone; exact method of communication unclear. Automated contact: unknown, not reported Type of therapist: native speaking psychotherapists Treatment fidelity: weekly supervision sessions, either face‐to‐face or via Skype Comparator arm Duration: 6 weeks Treatment protocol: participants were on a wait list for 6 weeks and then received the intervention. Therapist contact: none |
|
| Outcomes | Primary outcomes: PTSD (PDS); Anxiety and Depression (HSCL‐25); Quality of Life (EUROHIS‐QOL) | |
| Notes |
Funding source: unclear Declarations of interest among the primary researchers: unclear |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Randomisation was based on a computer‐generated randomisation list." |
| Allocation concealment (selection bias) | Unclear risk | Not reported. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Quote: "researchers and psychotherapists were not masked to the intervention." |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: self‐reported measures. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | ITT analysis but missing data > 30%. |
| Selective reporting (reporting bias) | Low risk | Study protocol available (Registered Trial (p.2) – Australian New Zealand Clinical Trial Registry) and the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | Low risk | Study appeared free from other sources of bias. |
Krupnick 2017.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 34 Diagnosis: DSM‐IV PTSD Method of recruitment: clinician referral Method of diagnosis: PCL‐M with cut‐off score 50 Trauma type: military Age (mean): WIRED: 35.44 years; WIRED+TAU: 44.75 years (SDs not reported) Sex: 8.8% women; 91.2% men Location: US Comorbidities: no structured screening for comorbid conditions Adjunctive therapy: a chart review revealed that several participants received adjunctive treatment Adjunctive medication: 50% Unemployment: not reported University education: not reported Exclusion criteria: current substance dependence, acute suicidality, psychosis, gross cognitive impairment, current participation in CBT Baseline PCL‐M scores (mean item score): WIRED: 3.6 (SD 0.3); WIRED+TAU: 3.9 (SD 0.4) |
|
| Interventions |
Internet programme based on trauma‐focused CBT (WIRED) vs TAU Experimental arm WIRED (based on Interapy) Duration: 10 sessions Treatment protocol
Therapist contact: as required. A short response and further instructions sent after each writing session. Type of contact: online Automated contact: none Type of therapist: psychologist Treatment fidelity: unclear Comparator arm TAU
|
|
| Outcomes | Primary outcomes: PTSD (PCL‐M (mean item scores)); Depression (PHQ‐9 (could not be entered into meta‐analysis)) | |
| Notes |
Funding source: grant from Telemedicine and Advanced Technology Research Center, U.S. Department of Defense. Declarations of interest among the primary researchers: unclear |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not reported. |
| Allocation concealment (selection bias) | Unclear risk | Not reported. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | All measures administered after baseline were self‐reported and completed online. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Dropout rate > 75%. Reasons for dropout not fully reported. |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but it was clear that the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | High risk | Small sample size. Participants received other interventions during the trial. |
Kuhn 2017.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 120 Diagnosis: DSM‐IV PTSD Method of recruitment: advertisements through fliers, media coverage, social media and websites (Craigslist) Method of diagnosis: PCL‐C Trauma type: various Age (mean): PTSD Coach: 39.43 (SD 15.16) years; wait list: 39.12 (SD 14.08) years Sex: 69.2% women; 30.8% men Location: US Comorbidities: not reported Adjunctive therapy: not reported Adjunctive medication: not reported Unemployment: not reported University education: 14.2% Exclusion criteria: currently receiving treatment for PTSD Baseline PTSD: all participants met diagnostic criteria for PTSD (no participant had a PCL score < 35) Baseline PCL scores (mean): for PTSD Coach: 63.19 (SD 11.78); wait list: 60.59 (SD 10.24) |
|
| Interventions |
Internet programme based on trauma‐focused CBT (PTSD Coach) vs wait list Experimental arm Duration: 12 weeks Treatment protocol: PTSD Coach PTSD Coach participants were instructed to download the app and use it however they would like to mimic real use. They were emailed links to the post‐treatment assessment 3 months later and follow‐up assessment 6 months. Comparator arm Duration: 12 weeks Treatment protocol: wait list Wait list participants received no intervention during the treatment period. After the post‐treatment assessment, they were told that the app being studied was PTSD Coach, that the app was available in the App Store and Google Play Store, and that they were now free to download and use it if they would like. They were emailed links to the post‐treatment assessment 3 months later and follow‐up assessment 6 months. |
|
| Outcomes |
Time points for assessment: 3 and 6 months Primary outcome: PTSD (PCL) Secondary outcomes: PTSD symptom coping self‐efficacy, Depression (PHQ‐8) and PTSD (B‐IPF) |
|
| Notes |
Funding source: not reported Declarations of interest among the primary researchers: none |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "randomized to conditions by the study coordinator using adaptive randomization, with the probability of condition assignment changing based on the assignment of participants already in the trial using www.randomizer.com." |
| Allocation concealment (selection bias) | Unclear risk | Not reported. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: self‐reported measures. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "Following the intent‐to‐treat principle, data from all randomized participants were analyzed, and multiple imputation was used to replace missing values, with 10 imputed data sets being generated." |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but it was clear that the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | Low risk | Study appeared free from other sources of bias. |
Lewis 2017.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 42 Diagnosis: DSM‐5 PTSD Method of recruitment: primary and secondary care clinicians referred participants in accordance with a brief recruitment protocol and a checklist of inclusion criteria. Study also advertised through local media and by means of posters and leaflets in high‐footfall National Health Service waiting areas. 40 (95%) had been referred to the study by treating clinicians and 2 (5%) were recruited by means of adverts in the media. Method of diagnosis: DSM‐5 PTSD measured by CAPS‐5 Trauma type: transportation accidents (9 participants); witnessing a sudden, violent, or accidental death (9); traumatic childbirth or stillbirth (8); sexual assault or rape (5); physical attack (4); life‐threatening illness or injury (3); serious accident (1); learning of the violent death of a loved one (1); seeing a mutilated body (1); and being held hostage/detained (1) Age (mean): treatment: 38.86 (SD 11.91) years; range 20–65 years; delayed treatment: 37.71 (SD 13.8) years; range 21–64 years Sex: 59.5% women; 40.5% men Location: UK Comorbidities: not reported Adjunctive therapy: excluded Adjunctive medication: stable for 1 month Unemployment: 16.6% University education: 42.8% Exclusion criteria: psychosis, previous trauma‐focused psychological therapy, DSM‐5 severe major depressive episode, substance dependence, inability to read and write fluent English, inability to access the Internet, change in psychotropic medication within 1‐month, concurrent psychological therapy, and suicidal intent Baseline PTSD: all participants met diagnostic criteria for PTSD Baseline CAPS‐5 scores (mean): immediate treatment: 35.99 (SD 6.29); delayed treatment: 37.12 (SD 6.95) |
|
| Interventions |
Internet programme based on trauma‐focused CBT vs wait list Experimental arm Duration: 8 weeks Treatment protocol: interactive, online, guided self‐help intervention, which included 8 online steps designed for delivery over 8 weeks.
The programme was initiated with a 1‐hour session with a therapist‐guide, followed by 30‐minute appointments every 2 weeks. Therapist contact: mean amount of therapist input per participant was 147.53 minutes (SD 57.01). This included a mean of 3.09 (SD 1.84) face‐to‐face meetings, 2.09 (SD 1.85) telephone calls, and 1.00 (SD 1.62) emails. Type of contact: face‐to‐face, telephone, email Automated contact: not reported Type of therapist: 3 male and 2 female therapists who were experienced in the delivery of trauma‐focused CBT guided the programme. They were trained to deliver the Internet‐based therapy, followed an intervention manual, and attended regular supervision meetings to maximise adherence to the manual. Treatment fidelity: therapists attended regular supervision meetings to maximise adherence to the manual. Comparator arm Duration: 8 weeks Treatment protocol: participants were on a wait list for 14 weeks and then received the intervention. Therapist contact: none |
|
| Outcomes |
Time points for assessment: pretreatment, post‐treatment (10 weeks after randomisation), 1 month (14 weeks after randomisation), and 3‐month follow‐up (22 weeks after randomisation). Primary outcome: PTSD (CAPS‐5) Secondary outcomes: Depression (BDI), Anxiety (BAI), Alcohol screening (AUDIT), Social Support (SSQ), Functional impairment (SDS) |
|
| Notes |
Funding source: Knowledge Transfer Partnership (KTP008512) Declarations of interest among the primary researchers: undertaken as part of a Knowledge Transfer Partnership between Cardiff University and Healthcare Learning Smile‐on. If the programme is marketed, royalties will be payable to Cardiff University and Cardiff and Vale University Health Board, with a proportion of these being shared with CL, NK, NR, TV, and JB. DF and VG had no conflicts of interest to disclose. |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Participants were randomly assigned, through a system of sealed, opaque envelopes containing an allocation code generated by an independent statistician, to an immediate or delayed treatment group." |
| Allocation concealment (selection bias) | Low risk | Quote: "Participants were randomly assigned, through a system of sealed, opaque envelopes containing an allocation code generated by an independent statistician, to an immediate or delayed treatment group." |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "A trained postdoctoral researcher who was blind to group allocation carried out all assessments." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "Throughout, varying engagement in the trial and with the treatment was handled using intention‐to‐treat principles." |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but it was clear that the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | High risk | Small sample size. Evaluated by the originators of the intervention. |
Littleton 2016.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 87 Diagnosis: DSM‐IV PTSD Method of recruitment: advertisements at 4 universities/community colleges, including postings on university psychology department participant management websites, and via social media Method of diagnosis: PSS‐SI Trauma type: rape Age (mean): 22 years (range 18–42 years) for whole sample Sex: 100% women Location: US Comorbidities: substance‐use disorder excluded Adjunctive therapy: not reported Adjunctive medication: not reported Unemployment: not reported University education: all students Exclusion criteria: current psychotherapy; changes in psychotropic medication in past 3 months; active suicidality; DSM‐IV criteria for current substance dependence Baseline PTSD: not reported Baseline PSS‐I scores (mean): From Survivor to Thriver: 11.2 (SD 5.8); psychoeducational website: 10.4 (SD 8.5) |
|
| Interventions |
Internet programme based on trauma‐focused CBT (From Survivor to Thriver) with Internet psychological therapy (non‐CBT; psychoeducational website) Experimental arm Duration: 14 weeks Treatment protocol: From Survivor to Thriver From Survivor to Thriver program consisted of 9 programme modules to be completed sequentially, 1 module at a time. The program had 3 phases. The first phase (modules 1–3) was designed to provide psychoeducation about PTSD and the impact of unwanted sex, as well as introduce general distress management strategies (i.e. relaxation, grounding) and healthy coping (e.g. asking others for help, setting an action plan) skills. The second phase (modules 4 and 5) introduced the cognitive model and taught participants to identify distorted and unhelpful automatic thoughts and utilise the challenging questions technique to respond to these thoughts. The third phase (modules 6–9) focused on using a number of cognitive behavioural techniques (e.g. the challenging questions technique, the pros and cons technique, behavioural experiments) to address specific concerns common among women following sexual assault (e.g. difficulties with trust, self‐blame for the assault). Each module contained several components, including: a video clip of the program developers discussing the topic covered in the module, a written description of the skills or techniques being introduced or utilised in the module, and, for modules 4–9, written examples of women modelling using the skills and techniques utilised in the module. The final program component included in all modules was the interactive exercises. These interactive exercises were designed either to help women think about the extent to which the particular issue raised in the module was a concern for them (e.g. the extent to which they blame themselves for the sexual assault) or to enable them to practice a described cognitive‐behavioural skill or technique and receive feedback on their practice. The interactive exercises were completed asynchronously, and were designed in a question and answer format. After a participant completed the interactive exercise, his/her program therapist then provided written feedback to each answer provided, as well as embedded a video message containing more extensive feedback on the interactive exercise page in the program. Therapist feedback focused on encouraging and praising participants for their effort, expressing empathy, encouraging continued program progress, socialising participants to the cognitive model, and providing assistance in challenging automatic thoughts, such as through further use of Socratic questioning. Comparator arm Duration: 14 weeks Treatment protocol: psychoeducational website The psychoeducational website contained the written informational content of the first 3 modules of the interactive program including the symptoms of PTSD, information about relaxation and grounding, and information about healthy coping strategies. The website did not contain any of the multimedia content (videos from the program developers, audio recorded relaxation exercises), or any interactive exercises. Women assigned to the psychoeducational website condition were given access to all the website content and instructed to log in to the program and utilise the content in whatever manner and however frequently they desired to do so over the course of the study. |
|
| Outcomes |
Time points for assessment: 3 months Primary outcome: PTSD (PSS‐I) Secondary outcomes: Depression (CES‐D), Anxiety (FDAS) |
|
| Notes |
Funding source: not reported Declarations of interest among the primary researchers: none |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Participants were randomized to the interactive program or psychoeducational website based on a computerized coin flip." |
| Allocation concealment (selection bias) | Unclear risk | Not reported. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "individuals completing pre‐, post‐ and follow‐up assessments were not blind to participants' intervention condition." |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "In addition, estimates of change from pre‐test to post‐test and follow‐up were estimated for all participants assigned to a program, an Intent to‐Treat (ITT) model. For the ITT analyses, post‐test and follow‐up data for participants who left the study was imputed using the R package. Twenty imputations were used with all outcome variables." |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but it was clear that the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | Low risk | Study appeared free from other sources of bias. |
Litz 2007.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 45 Diagnosis: DSM‐IV PTSD Method of recruitment: advertisements and presentations at department of defence sites Method of diagnosis: PSS‐I Trauma type: combat exposure: 9/11 attack on the Pentagon, or combat in Iraq or Afghanistan Age (mean): I‐CBT: 38.63 (SD 9.41) years; Internet supportive counselling: 39.86 (SD 7.72) years Sex: unclear Location: US Comorbidities: unclear Adjunctive therapy: excluded Adjunctive medication: unclear Unemployment: unclear University education: unclear Exclusion criteria: active substance dependence; current suicidal ideation; history of psychotic disorder; aged < 21 or > 65 years; PTSD or depression immediately before the trauma; current psychiatric treatment; marked ongoing stressors; inadequate social supports; change in medication Baseline PTSD: all participants met diagnostic criteria for PTSD Baseline PSS‐I scores: I‐CBT: 26.71 (SD 9.02); Internet supportive counselling: 29.16 (SD 9.93) |
|
| Interventions |
Internet programme based on trauma‐focused CBT with Internet psychological therapy (non‐CBT) Experimental arm Duration: 8 weeks Treatment protocol
Therapist contact: initial face‐to‐face (2 hours, including baseline assessment); telephone (ad‐lib); email (ad‐lib) Type of contact: online, email, face‐to‐face Automated contact: none Type of therapist: unclear Treatment fidelity: not reported Comparator arm Duration: 8 weeks Treatment protocol
Therapist contact: initial face‐to‐face (2 hours, including baseline assessment), telephone (ad‐lib), email (ad‐lib) Type of contact: online, email, face‐to‐face Automated contact: none Type of therapist: unclear |
|
| Outcomes |
Time points for assessment: pretreatment, post‐treatment, 3 months, and 6 months Primary outcome: PTSD (PSS‐I) Secondary outcomes: Depression (BDI), Anxiety (BAI) |
|
| Notes |
Funding source: supported by grant from NIHM (MH66589) Declarations of interest among the primary researchers: none |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not reported. |
| Allocation concealment (selection bias) | Unclear risk | Not reported. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Quote: "The study therapists conducted the baseline assessments in order to initially establish rapport. Clinicians blind to study arm conducted the follow‐up evaluations." |
| Incomplete outcome data (attrition bias) All outcomes | High risk | Authors stated that they performed ITT analyses, but only completer analyses were reported. |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but it was clear that the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | High risk | Small sample size. Large numbers lost to 3‐month follow‐up and 6‐month follow‐up. Evaluated by the originators of the intervention. |
McLean 2020a.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 40 Diagnosis: DSM‐5 PTSD Method of recruitment: not reported Method of diagnosis: CAPS‐5 clinical interview and CAPS‐5 score ≥ 25 Trauma type: exposure to a combat‐related Criterion A event experienced during deployment. Diagnosis of PTSD may be indexed to the combat‐related Criterion A event, or to another Criterion A event Age (mean): Web PE: 38.7 (SD 8.9) years; PCT: 41.5 (SD 6.5) years Sex: Web PE: 63.2% men; PCT: 85.7% men Location: US Comorbidities: unclear Adjunctive therapy: unclear Adjunctive medication: concomitant medications are not exclusionary; all medication changes were monitored for the duration of the trial. Unemployment: all active or retired military personnel University education: Web PE: graduate degree (5.3%), college 4 years (26.3%), college associate degree (15.8%), some college (42.1%); PCT: graduate degree (0%), college 4 years (23.8%), college associate degree (9.5%), some college (52.4%) Exclusion criteria: recent manic episode or psychotic disorder (determined by the bipolar and psychosis sections of the MINI), current alcohol dependence (determined by the AUDIT), evidence of moderate or severe traumatic brain injury (determined by an inability to comprehend baseline screening questionnaires), current suicidal ideation severe enough to warrant immediate intervention (determined by Depressive Symptom Inventory Suicidality Subscale and corroborated by a clinical risk assessment by a credentialed provider), other psychiatric disorders severe enough to warrant designation as the primary disorder, current engagement in evidence‐based treatment for PTSD Baseline PTSD: all participants met diagnostic criteria for PTSD Baseline PCL scores (mean): for Web PE: 50.37 (SD 3.37); PCT: 44.33 (SD 2.55) |
|
| Interventions |
Web PE vs PCT Experimental arm Duration: 8 weeks Treatment protocol
Therapist contact: 3 scheduled therapist telephone calls, and feedback after each session by text or email. Type of contact: online, email, text Automated contact: none Type of therapist: credentialed providers trained and supervised in PE Treatment fidelity: none Comparator arm Duration: 8 weeks Treatment protocol
Therapist contact: in‐person therapy sessions Type of contact: in‐person Automated contact: none Type of therapist: credentialed providers trained and supervised in PCT. Treatment fidelity: 3 randomly selected videos from each PCT participant were reviewed for treatment adherence by independent raters. Adherence was 99.2% for PCT elements. |
|
| Outcomes |
Time points for assessment: baseline; post‐treatment; 3 and 6 months (additional assessment time points for PCL‐5 following sessions 2, 4, 6, 8, 10) Primary outcome: PTSD (PCL‐5) Secondary measures: PTSD (CAPS‐5), Depression (PHQ‐9), Veterans Health (VR‐12), Treatment Credibility Questionnaire |
|
| Notes |
Funding source: not reported Declarations of interest among the primary researchers: not reported |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "Eligible participants were randomized 1:1 to receive Web‐PE or in‐person PCT using block randomization." |
| Allocation concealment (selection bias) | Unclear risk | No information. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Not possible due to nature of the intervention. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | No information. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | High attrition rates in each group and trial abandoned early due to issues recruiting the required number of participants. |
| Selective reporting (reporting bias) | High risk | Study protocol not available. Secondary outcomes data not reported at time points – just reported as change over time. |
| Other bias | High risk | Evaluated by originators of intervention. Small sample size. The researchers abandoned the RCT design and recruitment prematurely, failing to recruit the prespecified number of participants. Also, the researchers did not take into account in the analysis the large difference between groups in baseline PCL‐5 total scores. |
Miner 2016.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 49 Diagnosis: DSM‐IV PTSD Method of recruitment: advertisements through fliers and websites Method of diagnosis: PCL‐C Trauma type: various Age (mean): total: 45.7 (SD 13.9) years. Age range not reported by group Sex: 81.6% women; 18.4% men Location: US Comorbidities: not reported Adjunctive therapy: not reported Adjunctive medication: not reported Unemployment: not reported University education: not reported Exclusion criteria: currently receiving treatment for PTSD Baseline PTSD: all participants met diagnostic criteria for PTSD (no participant had a PCL score lower than the recommended range (i.e. 30–35) Baseline PSS‐I scores (mean): I‐CBT: 63.00 (SD 11.28); wait list: 59.33 (SD 11.34) |
|
| Interventions |
Internet programme based on trauma‐focused CBT vs wait list Experimental arm Duration: 4 weeks Treatment protocol: PTSD Coach Participants given the app and instructed to use it however they would like for the following month. No specific training, instructions for use, or suggestions of how PTSD Coach might be helpful were provided in attempt to represent real‐world use. Participants completed the postcondition assessment 1 month later. Comparator arm Duration: 4 weeks Treatment protocol: wait list No intervention over 1 month. Participants completed the postcondition assessment 1 month later. Upon completion of the postcondition assessment, participants received the PTSD Coach. |
|
| Outcomes |
Time points for assessment: 4 weeks Primary outcome: PTSD (PCL) Secondary outcomes: acceptability, feasibility |
|
| Notes |
Funding source: not reported Declarations of interest among the primary researchers: none |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "Participants were randomly assigned with an equal chance." Comment: insufficient reporting of methods to determine risk of bias. |
| Allocation concealment (selection bias) | Unclear risk | Not reported. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Comment: all outcomes self‐reported. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Quote: "The condition (PTSD Coach vs. waitlist) by time (baseline and post condition) interaction was used to estimate an effect size and assess if there was significant differential change in PCL scores using intention‐to‐treat (ITT) and completer analyses. For the ITT analysis multiple imputation with 10 imputed data sets was used to replace missing PCL values at the post‐condition (n=5; 10.2%) and follow‐up (n=9; 18.4%) assessments and ranges and averages of statistics across these imputed datasets are presented." Comment: ITT analysis and missing data < 20%. |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but it was clear that the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | Low risk | Study appeared free from other sources of bias. |
Spence 2011.
| Study characteristics | ||
| Methods | RCT | |
| Participants |
Sample size: 42 Diagnosis: DSM‐IV PTSD Method of recruitment: recruitment from a website that offered participation in trials, adverts in local media Method of diagnosis: MINI administered via telephone Trauma type: various Age (mean): I‐CBT: 43.0 (SD 15.2) years; wait list: 42.0 (SD 10.4) years Sex: 81% women; 19% men Location: Australia Comorbidities: unclear Adjunctive therapy: excluded Adjunctive medication: stable for 1 month Unemployment: 40% University education: unclear Exclusion criteria: currently experiencing a psychotic mental illness; severe symptoms of depression; currently highly dissociative; current CBT Baseline PTSD: all participants met diagnostic criteria for PTSD Baseline PCL‐C scores (mean): I‐CBT: 60.78 (SD 10.03); wait list: 57.00 (SD 9.69) |
|
| Interventions |
Internet program based on trauma‐focused CBT vs wait list Experimental arm Duration: 8 weeks Treatment protocol
Therapist contact (mean): 103.91 (SD 96.53) minutes Type of contact: telephone, email, forum Automated contact: reminders and notifications Type of therapist: clinical psychologist Treatment fidelity: not reported Comparator arm Duration: 8 weeks Treatment protocol: wait list |
|
| Outcomes |
Time points for assessment: pretreatment, post‐treatment, and 3 months Primary outcome: PTSD (PCL‐C) Secondary outcomes: Depression (PHQ‐9), Anxiety (GAD‐7), Functional impairment (SDS) |
|
| Notes |
Funding source: supported by a research fellowship from the New South Wales Institute of Psychiatry. Declarations of interest among the primary researchers: contract grant sponsor: New South Wales Institute of Psychiatry |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "Forty‐four applicants met all inclusion criteria and were randomized via a true randomization process (www.random.org), generated by an independent person." |
| Allocation concealment (selection bias) | Unclear risk | Not reported. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Impossible to blind participants or therapists. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Quote: "Treatment group participants had a similar diagnostic assessment at three‐month follow‐up. These assessments were conducted by JS and KS, who were not blind to the participants' condition." |
| Incomplete outcome data (attrition bias) All outcomes | High risk | ITT analyses using LOCF. |
| Selective reporting (reporting bias) | Low risk | Study protocol not available but it was clear that the published reports included all expected outcomes, including those that were prespecified. |
| Other bias | Unclear risk | Small sample size. Recruitment ceased after 4 weeks due to staff availability. Did not meet the recruitment target (indicated by power calculation) of 26 per group. Evaluated by the originators of the intervention. Time‐since‐trauma not recorded, so unsure about the requirement for the trauma to have been at least 1 month previously. |
AUDIT: Alcohol Use Disorders Identification Test; BAI: Beck Anxiety Inventory; BDI‐II: Beck Depression Inventory II; B‐IPF: Brief‐Inventory of Psychosocial Functioning; CAPS: Clinician Administered PTSD Scale; CBT: cognitive behavioural therapy; CES‐D: Center for Epidemiologic Studies Depression Scale; CGI‐I: Clinical Global Impressions – Improvement; DSM‐IV: Diagnostic and Statistical Manual of Mental Disorders; EQ‐5D: EuroQoL 5D; EUROHIS‐QOL: European Health Interview Survey‐Quality of Life; FDAS: Food & Drug Analytical Services; GAD‐7: Generalized Anxiety Disorder 7‐Item Scale; HSCL‐25: Hopkins Symptom Checklist‐25; I‐CBT: Internet‐based cognitive behavioural therapy; I‐PE: I‐CBT based on prolonged exposure; IES‐R: Impact of Event Scale – Revised; ITT: intention to treat; K10: Kessler 10; LEC: Life Events Checklist; LOCF: last observation carried forward; MADRS‐S: Montgomery‐Åsberg Depression Rating Scale – Self‐report; MDD: major depressive disorder; MINI: Mini‐International Neuropsychiatric Interview; PCL: PTSD Checklist; PCL‐C: PTSD Checklist – Civilian Version; PCL‐M: PTSD Checklist – Military Version; PCT: present‐centred therapy; PDS: Posttraumatic Diagnostic Scale; PE: prolonged exposure; PHQ‐9: Patient Health Questionnaire‐9; PHQ‐15: Patient Health Questionnaire‐15; PSS‐I: PTSD Symptom Scale – Interview; PTSD: post‐traumatic stress disorder; QOLI: Quality of Life Inventory; RCT: randomised controlled trial; SD: standard deviation; SDS: Sheehan Disability Scale; SSQ: Social Support Questionnaire; TAU: treatment as usual; VR‐12: Veterans RAND 12‐Item Health Survey; WIRED: Warriors Internet Recovery & EDucation.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Bahena 2016 | No formal diagnosis of DSM/ICD PTSD. |
| Bishop 2012 | Reduction in PTSD symptoms was not the main aim of the intervention. |
| Blankenship 2013 | No formal diagnosis of DSM/ICD PTSD. |
| Bottche 2016 | No formal diagnosis of DSM/ICD PTSD. |
| Brief 2012 | No formal diagnosis of DSM/ICD PTSD. |
| Brief 2013 | No formal diagnosis of DSM/ICD PTSD. |
| Daggett 2014 | No formal diagnosis of DSM/ICD PTSD. |
| de Kleine 2019 | Intervention deemed to be brain‐training rather than CBT. |
| Elbers 2013 | No formal diagnosis of DSM/ICD PTSD. |
| Hirai 2005 | No formal diagnosis of DSM/ICD PTSD. |
| Hirai 2012 | No formal diagnosis of DSM/ICD PTSD. |
| Kersting 2011 | No formal diagnosis of DSM/ICD PTSD. |
| Kersting 2013 | No formal diagnosis of DSM/ICD PTSD. |
| Knaevelsrud 2006 | No formal diagnosis of DSM/ICD PTSD. |
| Knaevelsrud 2010a | No formal diagnosis of DSM/ICD PTSD. |
| Knaevelsrud 2010b | No formal diagnosis of DSM/ICD PTSD. |
| Knaevelsrud 2017 | No formal diagnosis of DSM/ICD PTSD. |
| Lange 2001 | No formal diagnosis of DSM/ICD PTSD. |
| Lange 2003 | No formal diagnosis of DSM/ICD PTSD. |
| Maercker 2004 | No formal diagnosis of DSM/ICD PTSD. |
| McCleary 2019 | Trial used PTSD Coach app in all 4 arms and measured the effectiveness of 3 different add‐on coaching interventions (clinician, peer, and algorithm). |
| McGlinchey 2014 | No formal diagnosis of DSM/ICD PTSD. |
| NCT01508377 | No formal diagnosis of DSM/ICD PTSD. |
| NCT01552278 | Reduction in PTSD symptoms was not the main aim of the intervention. |
| NCT01678196 | No formal diagnosis of DSM/ICD PTSD. |
| NCT01710943 | Participants required to screen positive for PTSD or alcohol/drug use. |
| NCT01760213 | No formal diagnosis of DSM/ICD PTSD. |
| NCT01891734 | No formal diagnosis of DSM/ICD PTSD. |
| NCT03208738 | Correspondence with researchers confirmed that the trial did not start. |
| NCT03733028 | Treatment was for anger rather than PTSD |
| NCT04362358 | Prevention rather than treatment of PTSD. |
| Niemeyer 2020 | < 70% of participants had a PTSD diagnosis at baseline. |
| Nieminen 2016 | < 70% of participants had a PTSD diagnosis at baseline. |
| Possemato 2010 | A brief self‐guided telehealth intervention for PTSD in combat veterans: a pilot study. |
| Possemato 2011 | A brief self‐guided telehealth intervention for PTSD in combat veterans: a pilot study. |
| Sayer 2009 | No formal diagnosis of DSM/ICD PTSD. |
| Smith 2019 | After receiving correspondence from the authors, we determined that the education and symptom management intervention was not suitable. |
| Solzbacher 2012 | No formal diagnosis of DSM/ICD PTSD. |
| Steinmetz 2012 | No formal diagnosis of DSM/ICD PTSD. |
| Stockton 2014 | Reduction in PTSD symptoms was not the main aim of the intervention. |
| Wagner 2007 | No formal diagnosis of DSM/ICD PTSD. |
| Wagner 2012 | No formal diagnosis of DSM/ICD PTSD. |
| Wang 2013 | No formal diagnosis of DSM/ICD PTSD. |
| Zernicke 2014 | No formal diagnosis of DSM/ICD PTSD. |
CBT: cognitive behavioural therapy; DSM: Diagnostic and Statistical Manual of Mental Disorders; ICD: International Classification of Diseases; PTSD: post‐traumatic stress disorder.
Characteristics of ongoing studies [ordered by study ID]
ACTRN12611000989943.
| Study name | A comparison of Internet‐based cognitive behavioural therapy for posttraumatic stress disorder with and without exposure: a randomized controlled trial |
| Methods | Randomised controlled trial using a list generated prior to the study via a software program (www.random.org), at another site, in another country. The list will then be transcribed and details transferred to sealed envelopes. |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm Clinician‐guided I‐CBT with exposure Participants will complete 6 lessons of Internet‐based treatment about management of symptoms of PTSD. 1 lesson will be completed every 7–10 days over 8 weeks. Comparator arm Clinician‐guided I‐CBT without exposure Participants will complete 4 lessons of Internet‐based treatment about management of symptoms of PTSD using the same protocol with 2 lessons about exposure removed. 1 lesson will be completed every 7–14 days over 8 weeks. |
| Outcomes |
Time points for assessment: 1‐day pretreatment; 1‐day post‐treatment; 3 and 12 months' post‐treatment Primary outcomes: PTSD (PSS‐I, PCL‐C) Secondary outcomes: neuroticism (NEO‐FFI), disability (SDS), depression (PHQ‐9), anxiety (GAD‐7), service use (SEQ), health status (EQ‐5D) |
| Starting date | 20 July 2011 |
| Contact information | Prof Nick Titov Department of Psychology, Macquarie University, Australia nick.titov@mq.edu.au |
| Notes | www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=343335 |
ACTRN12616000956404.
| Study name | Internet‐based intervention for posttraumatic stress disorder (PTSD) in soldiers: exploring mechanisms of treatment outcome |
| Methods | Randomised controlled trial. Central randomisation by computer software using a random number generator |
| Participants |
Inclusion criteria: Treatment group/wait list control group
Healthy controls group
Exclusion criteria: Treatment group/wait list control group
Healthy controls group
|
| Interventions |
Experimental arm Therapist‐guided Internet‐based intervention delivered via an online platform lasts 5 weeks and includes 10 structured writing assignments. Participants complete 2 assignments per week. Each assignment requires approximately 45 minutes of writing time. Comparator arm Same treatment as above after an initial waiting period of 6 weeks. Participants seen for the initial assessment and pass through 1 additional assessment before the beginning of the treatment phase. The treatment procedure hereafter is identical to the protocol above. Healthy controls arm |
| Outcomes |
Time points for assessment: post‐treatment; 3, 6, and 12 months Primary outcomes: PTSD (PCL‐5), PTSD (CAPS) Secondary outcomes: Depression (PHQ), Anxiety (GAD‐7), Satisfaction with Life (SWLS), Emotion Regulation (DERS), Post‐traumatic Cognitions (PTCI), Post‐traumatic Growth (PTGI), Coping Style (CSS) |
| Starting date | 22 August 2016 |
| Contact information | Prof Christine Knaevelsrud Freie Universitaet Berlin christine.knaevelsrud@fu‐berlin.de |
| Notes | www.anzctr.org.au/Trial/Registration/TrialReview.aspx?id=370924 |
DRKS00010245.
| Study name | Evaluation of web‐based cognitive and behavioural therapeutic components and its change mechanisms for Arab people with posttraumatic stress disorder |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm 1 Web‐delivered exposure‐focused therapy Experimental arm 2 Web‐delivered cognitive online therapy Comparator arm Untreated (wait list) |
| Outcomes |
Time points for assessment: pretreatment; post‐treatment; 3 months Primary outcomes: PTSD (PCL‐5) Secondary outcomes: Prolonged Grief (PG‐13), Depression (PHQ‐9), Anxiety (GAD‐7), Post‐traumatic Growth (PTGI), DEVS, Body Volume Index (BVI) |
| Starting date | 30 June 2016 |
| Contact information | Freie Universität Berlin |
| Notes | www.drks.de/drks_web/navigate.do?navigationId=trial.HTML&TRIAL_ID=DRKS00010245 |
DRKS00016931.
| Study name | Internet‐based therapy for physicians with post‐traumatic stress |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm 10 structured writing assignments (45 minutes each), divided into 3 treatment phases: self‐confrontation, cognitive restructuring, and social sharing (reflection on the therapeutic process, changes in life, and future perspectives). After each assignment, participants receive feedback and the next writing instruction. Comparator arm Wait list |
| Outcomes |
Time points for assessment: postintervention; and 3, 6, and 12 months Primary outcomes: PTSD (PCL‐5) Secondary outcomes: Depression (PHQ‐9), Anxiety (GAD‐7), Somatic Symptoms (PHQ‐15), Burnout (MBI), KFM, Alcohol (CAGE), Functional Impairment (SDS), Sleep (PSQI), Quality of Life (PGI, and SF‐12) |
| Starting date | 2019 |
| Contact information | |
| Notes |
DRKS00017838.
| Study name | Feasibility, acceptance and effectiveness of Step‐by‐Step, a smartphone‐based self‐help program for Syrian refugees: a pilot study |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm Step by Step A smartphone‐ and internet‐based self‐help program with an introduction session (15 minutes) and 5 weekly sessions (each 30 minutes), a digital mood diary, contact‐on‐demand by trained and supervised non‐specialist, Syrian research assistants. Comparator arm TAU Participants receive basic psychoeducation and information on TAU in Germany. Participants in this group can make use of other care services in parallel. |
| Outcomes |
Time points for assessment: 6 and 12 weeks Primary outcomes: Anxiety and Depression (HSCL‐25), Functioning (WHODAS) Secondary outcomes: PTSD (PCL‐5), Mental Health (PSYCHLOPS), access to health services (questionnaire), costs of care (SRI), user satisfaction (CSQ‐3), Life Events Scale (LEC‐5), Life stressors of assylum seekers (PMLDC), weekly psychological distress (K10), momentary mood ratings, App usage |
| Starting date | |
| Contact information | |
| Notes | Contacted author who informed us this study is still ongoing |
DRKS00020266.
| Study name | Online‐program after adverse childhood experiences in GDR children's homes. A randomized controlled trial |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm Manualised writing therapy through secured digital platform. After every writing exercise, participants are given an individual answer from specially trained psychologists Comparatorarm Wait list |
| Outcomes |
Time points for assessment: 6 weeks; 3, 6, and 12 months Primary outcomes: PTSD (ITQ) Secondary outcomes: Depression (PHQ‐9), Anxiety (GAD‐7), Post‐traumatic Growth (PTGI), Quality of Life (EUROHIS‐QOL) |
| Starting date | January 2020 |
| Contact information | |
| Notes | Public funding institutions financed by tax money/Government funding body (German Research Foundation (DFG), Federal Ministry of Education and Research (BMBF), etc.) BMBF (Research program: GDR‐Research) conducted through DLR, Gesellschaft, Innovation, Technologie, Geistes‐ und Sozialwissenschaften |
Ehlers 2020.
| Study name | A randomised controlled trial of therapist‐assisted online psychological therapies for post‐traumatic stress disorder |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm 1 iCT‐PTSD Experimental arm 2 iStress‐PTSD Comparator arm Wait list |
| Outcomes |
Time points for assessment: 2, 6, 13, 26, 36, and 65 weeks Primary outcomes: PTSD (PCL‐5) Secondary outcomes: PTSD (IES‐R), PTSD (CAPS‐5), Depression (PHQ‐9), Anxiety (GAD‐7), WHO Five Wellbeing Index, Work and Social Adjustment (WSAS), Endicott quality of life, Sleep (ISI), costs (CSRI and PCQ), Quality of Life (EQ‐5D‐DL), Post‐traumatic Cognitions (PTCI), TMQ (Treatment Motivation Questionnaire), Responsibility interpretations Questionnaire (RIQ), Suicide (SBQ), Trait State Dissociation (TSDQ), Self Efficacy (GSES), Intelligence (WAIS), Alcohol (AUDIT), PTSD (ITQ), Emotion Rating Scale |
| Starting date | January 2017 |
| Contact information | Miss Rachel Maddox Department of Experimental Psychology, University of Oxford rachel.maddox@psy.ox.ac.uk |
| Notes | iCT‐PTSD was developed by Anke Ehlers and David M Clark's team and iStress was developed by Gerhard Andersson's team. The trial is funded by the Wellcome Trust, grant 200976, awarded to Anke Ehlers and David M Clark, and is supported by the National Institute of Health Research (NIHR) Oxford Health Biomedical Research Centre. |
Golchert 2019.
| Study name | Development and evaluation of a self‐help app for traumatized Syrian refugees |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm Self‐help app: modular, interactive self‐help app in Arabic, grounded on CBT models for treatment of PTSD Comparator arm Psychoeducational information brochure on traumatisation/PTSD |
| Outcomes |
Time points for assessment: 4 and 12 weeks' postintervention Primary outcomes: PTSD (PDS‐5) Secondary outcomes: Depression (PHQ‐9), Anxiety (GAD‐7), Somatic Symptoms (PHQ‐15), Self‐efficacy (GSES), Quality of Life (PGI), Social Support (ESSI, and LSNS), (Stigma) SSMIS, ambiguous loss, complicated grief, Quality of Life (EQ‐5D‐DL), user acceptance (TAM‐3), app usability |
| Starting date | November 2018 |
| Contact information | Prof Dr Steffi Riedel‐Heller Institut für Sozialmedizin, Arbeitsmedizin und Public Health (ISAP), Medizinische Fakultät, Universität Leipzig steffi.riedel‐heller at medizin.uni‐leipzig.de |
| Notes | Funded by the Innovationsfonds (01VSF16033) of the Federal Joint Committee (Gemeinsamer Bundesausschuss/G‐BA) Contacted trial authors. Trial ongoing. |
ISRCTN16806208.
| Study name | A randomised controlled trial of therapist‐assisted online psychological therapies for post‐traumatic stress disorder |
| Methods | Randomised controlled trial |
| Participants | Aged ≥ 18 years with PTSD resulting from traumatic events experienced in adulthood |
| Interventions |
Experimental arm Internet‐delivered psychological therapy started immediately Comparator arm Internet‐delivered psychological therapy started after a delay of 13 weeks Both therapies involve completing therapy modules online and assignments over 3 months, with guidance via messages and weekly telephone calls with an experienced psychological therapist. |
| Outcomes |
Time points for assessment: baseline; 6 weeks; end of therapy/waiting; and 3, 6, and 12 months after end of therapy Primary outcome: PTSD symptoms (measured by weekly questionnaires and by interviewing participants regarding their experiences) |
| Starting date | January 2017 |
| Contact information | Maxie Scheske maxie.scheske@psy.ox.ac.uk |
| Notes |
Lehavot 2017.
| Study name | Evaluation of web‐based CBT for women Veterans with PTSD |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm DESTRESS‐WV tailored online intervention for PTSD for women Veterans with coach support. Participants will be asked to log on to the website twice per week for 30–60 minutes each time. On 2 occasions, participants will be asked to write about current stressors or hassles. Additionally, on 2 occasions, participants will be asked to write about a traumatic experience and then rewrite it. Participants will be guided in using various coping skills taught in the program during this writing process. Homework assignments will include stress management skills that participants will be asked to practice and apply. A study coach will call each participant once a week for 8 weeks for about 15 minutes to review their progress with the program. Comparator arm Telephone monitoring A study coach will call participants once a week for 8 weeks for approximately 15 minutes. The coach will assess participants' PTSD symptoms and safety. They will encourage participants to use the time on the call to discuss any current life issues or problems that they would like. Active listening and rephrasing will be used, while teaching cognitive‐behavioural strategies will be avoided. |
| Outcomes |
Time points for assessment: baseline; 8–16, 20–28, and 32–40 weeks Primary outcomes: PTSD (PCL‐5) Secondary outcomes: Depression (PHQ‐8), quality of life (Q‐LES‐Q‐SF) |
| Starting date | July 2016 |
| Contact information | Keren Lehavot VA Puget Sound Health Care System Seattle Division, Seattle, US |
| Notes |
McLean 2020b.
| Study name | The efficacy of written exposure therapy versus imaginal exposure delivered online for posttraumatic stress disorder: design of a randomized controlled trial in Veterans |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm Written exposure therapy 1st session: psychoeducation about PTSD, treatment rationale and instructions for writing about the traumatic experience delivered via an online platform, with a peer support specialist providing instructions and feedback before and after each writing session. The therapist reads scripted writing instructions and then has the client complete the 30 minutes of writing without interference; the therapist alerts the client at 30 minutes to stop writing. Clients are instructed to write about their index (worst) trauma during each writing session (same event at each session), providing as much detail as possible; participants are also instructed to include details on thoughts and feelings they experienced during the event. Participants are instructed to write about the experience as they "look back upon it now." At the conclusion of 30 minute of writing, they spends a short amount of time (i.e. ≤ 10 minutes) inquiring about the participants' reaction to their writing session. The written narratives are saved to the participant's Vets Prevail account and are accessible to the participant and the peer support specialists, who is instructed to review the narrative from the prior session to provide feedback to the participant before the next writing exercise (consistent with standard WET procedure). Comparator arm Imaginal exposure Verbally recounting the narrative of the index trauma, typically with closed eyes to promote visualisation and engagement. Each session (referred to as levels on Vets Prevail) is broken into multiple exercises for the participant to complete in a specific order before they can move onto the next session (level). Most sessions include the same components: completion of a short assessment, chatting online with a peer support specialist, entering a pre‐exposure distress rating, completing an imaginal exposure exercise, entering a postexposure distress rating, and talking online with a peer support specialist. There is no between‐session homework. Talk to text software is used to generate a written account for the peer support specialist to review prior to the next session. |
| Outcomes |
Time points for assessment: post‐treatment, 3 months Primary outcomes: PTSD (PCL‐5) Secondary outcomes: Depression (PHQ‐9), Work and Social Adjustment (WSAS), PTC‐9, Depression (CEQ) |
| Starting date | |
| Contact information | |
| Notes | Supported with resources of the National Center for PTSD and the use of facilities at the VA Palo Alto Health Care System. |
NCT02929979.
| Study name | Cognitive remediation for alcohol use disorder (AUD) and posttraumatic stress disorder (PTSD) |
| Methods | Randomised, triple‐blind, factorial assignment controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
Self‐report and collateral history from medical record/primary care physician/outpatient addiction treatment team will be used as necessary to determine inclusion and exclusion. Suicidal and homicidal intent will be assessed in the context of a structured clinical interview. In the unlikely event that respondents endorse active intent they will be referred immediately for treatment and will be excluded from the current study. |
| Interventions |
Experimental arm Cognitive training Participants complete 22.5 hours of cognitive training exercises over 6 weeks (training 5 times a week) using an app‐based program, BrainHQ. Exercises will be packaged into 4 modules (attention skills, memory skills, executive functioning skills, cognitive control skills) comprised of 4 exercises each. All participants will progress through the same fixed schedule of modules. Comparator arm Placebo control Participants play a rotating set of commercial computer games at the same dose and frequency as the cognitive training. |
| Outcomes |
Time points for assessment: baseline, 6 weeks, and 3 months Primary outcomes: neuropsychological assessment (WAIS‐IV, TMT, AAT, HVLT, BVMT‐R, WAIS‐IV WMI, CPT‐II, CWIT, WCST, IGT, DDT, Tower Test (DKEFS)) Secondary outcomes: alcohol use (TLFB), functional status (IPF), PTSD symptom severity (PCL‐M) |
| Starting date | 1 January 2015 |
| Contact information | Adrienne Julie Heinz VA Palo Alto Health Care System, CA, US |
| Notes | NCT02929979 |
NCT04058795.
| Study name | PTSD mobile app for cancer survivors |
| Methods | Randomised controlled, crossover trial |
| Participants |
Inclusion criteria:
|
| Interventions |
CaDC vs control mHealth app CaDC provides tools based on CBT principles to manage stress Experimental arm 1 CaDC and mCoaching CaDC app plus weekly clinician contact to provide support in navigating the app, encouraging adherence to the use of the CaDC, and providing guidance in choosing treatment strategies Experimental arm 2 CaDC plus mCBT 10 mobile sessions with a therapist Comparator arm Mental health services that are available to all people with cancer |
| Outcomes |
Time points for assessment: 1, 3, and 6 months Primary outcomes: PTSD (PCL‐5) Secondary outcomes: distress (Distress thermometer), quality of life (PROMIS QOL), depression (PROMIS), anxiety (PROMIS), self‐efficacy as measured by the Self‐efficacy for Chronic Disease, user satisfaction |
| Starting date | September 2020 |
| Contact information | Sophia K Smith Duke University sophia.smith@duke.edu |
| Notes |
NCT04094922.
| Study name | PTSD Coach Sweden: evaluating a self‐help mobile app for posttraumatic stress in a community sample |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm Swedish PTSD Coach Comparator arm Wait list |
| Outcomes |
Time points for assessment: 3, 6, and 9 months Primary outcomes: PTSD (PCL‐5) Secondary outcomes: Depression (PHQ‐9), Health measure (WHODAS), Patient Reported Outcome Measure (TIC‐P), Somatic Symptoms (PHQ‐15), Negative Effects (NEQ) |
| Starting date | May 2019 |
| Contact information | Filip K Arnberg Uppsala University |
| Notes | We requested an update from researchers on status and received the following response (quote): "our PTSD Coach trial ended recruitment this summer. We expect that the results will be published in 2021." Unsure on eligibility of participants given not sure if PTSD diagnosis required, therefore perhaps will be < 70%. |
NCT04101942.
| Study name | An Internet‐delivered cognitive‐behavioral intervention provided soon after trauma: a RCT |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm I‐CBT: treatment is divided into 4 modules, each containing homework assignments. Participants in the experimental group will be assigned a therapist that they can contact through a message system in the platform and expect answer within 36 hours on weekdays Comparator arm Wait list |
| Outcomes |
Time points for assessment: 1, 6, and 12 months Primary outcomes: PTSD (PCL‐5) Secondary outcomes: Depression (MADRS), Quality of Life (EQ‐5D), Patient Reported Outcome Measure (TIC‐P), adverse events |
| Starting date | |
| Contact information | |
| Notes | We have not emailed Andersson about this as in our last email correspondence with him he informed us of results becoming available in due course for a large‐scale RCT, with promising results, and we believe he may have been referring to this one. We believe study #256 is the first‐phase RCT with this one being the subsequent larger‐scale RCT. |
NCT04155736.
| Study name | Testing a self‐management app for symptoms of posttraumatic stress |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria: Not specified |
| Interventions | RENEW is a behavioural health self‐management app that focuses on 3 core activities: 1. in vivo exposure (to help participants approach safe situations they have been avoiding that they want to feel comfortable in); 2. expressive writing (to help participants emotionally process trauma memories); and 3. self‐care exercises (designed to promote behavioral activation, social connectedness, and relaxation; includes a breathing relaxation tool). Users have access to psychoeducation via a whiteboard video and short texts, and Motivation content (quotes, videos, images, and songs) related to confronting challenges and overcoming fears. For each exercise that is completed, the user earns points that are connected to levels and visually reinforcing images (i.e. a growing tree). Experimental arm 1 RENEW with coaching Coaching: users will be assigned a support staff member who is notified when the user engages with the app or if they have not engaged for 7 days. Support staff are provided with psychoeducation material including information about how to provide effective support for the user and direct messaging capacity to respond to app notifications about user engagement (e.g. user earned X points, user achieved a new level). Experimental arm 2 RENEW with no coaching Comparator arm Wait list |
| Outcomes |
Time points for assessment: 10 weeks' post‐treatment Primary outcomes: PTSD (PCL‐5) Secondary outcomes: Depression (PHQ‐9), a PTSD coping self‐efficacy measure (developed following Bandura's 2006 guidelines, Perceived Social Support (MSPSS), perceived helpfulness of RENEW |
| Starting date | January 2020 |
| Contact information | Carmen McLean Palo Alto Veterans Institute for Research carmen.mclean4@va.gov |
| Notes | We contacted the author who informed us that trial data are unlikely to be available for some time. |
NCT04286165.
| Study name | Brief Peer Supported webSTAIR Compared to Enhanced Wait List (BPSwS) |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm Brief Peer Supported webSTAIR (BPS webSTAIR) The BPS webSTAIR program is an online platform where individuals are taught skills in emotion regulation. In this program, they will learn how to recognise the emotions they are feeling by assessing their own body language, being attentive to their thought processes, and attempting to replace negative behaviours with positive. Throughout the platform they will engage in written exercises to help them solidify the lessons with their own life experience. They will also have the support of peers throughout the program to help them when they need clarification or having a difficult time applying the lesson(s). After randomisation, participants will have 10 weeks to complete the 6 modules. Every time the Veteran logs on they will have the opportunity to engage with a Veteran peer for support through the web program. Contacts can last up to 1 hour. Veterans will receive a series of automated reminders and engagement emails that the Vets Prevail programme sends at various points in the program. Comparator arm Wait list |
| Outcomes |
Time points for assessment: 10 weeks' postrandomisation and 2 months after the participant finishes all 6 levels of the intervention for participants engaged in treatment Primary outcomes: Emotion Regulation (DERS‐16), Depression (PHQ‐8), PTSD (PCL‐5) Secondary outcomes: Work and Social Adjustment (WSAS), PTSD (CAPS), Life Events (LEC‐2) |
| Starting date | January 2020 |
| Contact information | Marylène Cloitre Palo Alto Veterans Institute for Research Marylene.Cloitre@va.gov |
| Notes | We contacted the author and were advised the study is still ongoing. |
NCT04333667.
| Study name | Effectiveness of mindfulness‐based Internet intervention Still Me (Still‐Me) |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm Mindfulness‐based Internet intervention Comparator arm Wait list |
| Outcomes |
Time points for assessment: 8 and 12 weeks' post‐treatment Primary outcomes: PTSD (ITQ) Secondary outcomes: Depression (PHQ‐9), Anxiety (GAD), Adjustment Disorder (ADNM‐8), Emotional and Psychological Mental Health (PMH), Mindfulness (FFMQ‐15) |
| Starting date | April 2020 |
| Contact information | Evaldas Kazlauskas Vilnius University, Lithuania |
| Notes | We queried status with researcher –and received the following response (quote): "We have completed the pilot and currently are in the progress of data analysis and writing report about the effects of the intervention. We do not have a manuscript yet. We expect to have results of the pilot published in 2021. More information about the study could provide clinical psychologist and PhD student Austeja Dumarkaite from our Vilnius trauma group." |
Nollett 2018.
| Study name | A study of trauma‐focused online guided self help versus trauma‐focused cognitive behavioural therapy for post‐traumatic stress disorder |
| Methods | Multicentre, pragmatic, randomised controlled, non‐inferiority trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm Trauma‐focussed CBT In‐session treatment is augmented by daily homework assignments which participants are required to complete between sessions. They will be asked to complete the IES‐R at each session to measure and monitor their PTSD symptoms. Treatment for 8–12 weeks, up to 12 sessions, with each session lasting 60–90 minutes. TFCBT will be delivered face‐to‐face by experienced psychological therapists currently working in the IAPT or psychological services. They come from a variety of backgrounds, including mental health nurses, clinical psychologists, and counsellors, and have varying levels of experience in working therapeutically with people with PTSD. All therapists will attend ≥ 1 × 1‐day workshop on 1 of the TFCBT programmes. Comparator arm SPRING Online GSH programme based on TFCBT 8 steps completed online in participant's own time. Initial 1 hour meeting with therapist then 4 subsequent fortnightly meetings of 30 minutes, normally undertaken face‐to‐face but deliverable by telephone or Internet depending on participant preference. Participant also receives 4 telephone calls or emails between sessions to discuss progress, identify problems, and set new goals. All therapists will receive a half day's training from 2 of the authors who were involved in developing the intervention and delivering it in the pilot study. Therapists also receive ongoing supervision of ≥ 1 training cases until they are regarded as competent. The therapists will work from a manual and, for the duration of the trial, will receive supervision once per month. |
| Outcomes |
Time points for assessment: 16 and 52 weeks' postrandomisation Primary outcomes: CAPS‐5 Secondary outcomes: traumatic stress (IES), EQ‐5DAL, quality of life/functional impairment (WSAS), depression (PHQ‐9), anxiety (GAD‐7), alcohol use (AUDIT‐O), perceived social support (MSPSS), healthcare resource utilisation (CSD, SRIEV), Quality of Life (EQ‐5D), Sleep (ISI), Post‐traumatic Cognitions (PTCI), Self‐efficacy (GSES), treatment satisfaction (CSQ), Therapeutic Alliance (ARM5), therapy adherence, qualitative information |
| Starting date | 8 August 2017 |
| Contact information | Prof Jonathan Bisson Cardiff University, Cardiff, UK |
| Notes | Trial is underway and results are expected in spring 2021. |
NTR6912.
| Study name | Addition of the SUPPORT Coach in PTSD treatment |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm SUPPORT Coach, a mobile app aiming to help people better understand and self‐manage their PTSD symptoms. The SUPPORT Coach includes psychoeducation, a self‐test with a monitoring feature, and exercises and tools to cope with PTSD symptoms Comparator arm Trauma‐focused therapy |
| Outcomes |
Time points for assessment: post‐treatment at 13th session Primary outcomes: PTSD symptoms (measured by the difference in PCL‐5 score reduction (continuous)) Secondary outcomes: feasibility and acceptance of the SUPPORT Coach app |
| Starting date | 15 October 2017 |
| Contact information | Dr M Oiff Academic Medical Center, Amsterdam m.olff@amc.uva.nl |
| Notes | www.amc.nl/supportcoach |
Sjomark 2018.
| Study name | A longitudinal, multi‐centre, superiority, randomized controlled trial of Internet‐based cognitive behavioural therapy (iCBT) versus treatment‐as‐usual (TAU) for negative experiences and posttraumatic stress following childbirth: the JUNO study protocol |
| Methods | Randomised controlled trial |
| Participants |
Inclusion criteria:
Exclusion criteria:
|
| Interventions |
Experimental arm I‐CBT Step 1: 6 weekly modules include homework based on the content. The partner receives identical content and a 'partner instruction' with specific tasks to perform (support and make room for practice; reflective listening; talk about the birth experience and present symptoms, etc.). A psychologist guides, encourages, and responds to questions from the participants. After completion of each module, psychologist writes an email to the participant to inform her that the week's CBT is completed. Women who complete Step 1 and fulfil criteria for PTSD enter Step 2. Step 2: presentation and format of material in Step 2 is the same as in Step 1. Expressive writing (structured written assignments) is used as an alternative to imagery exposure. I‐CBT Step 2 is more individualised than I‐CBT Step 1 with weekly therapeutic support via mail. There is no partner content in this step since the empirical evidence is based on an individual treatment protocol. Comparator arm TAU TAU includes conventional support according to existing practice at the Department of Obstetrics and Gynaecology in the participating hospitals. TAU can include an opportunity for a follow‐up visit with the physician or counselling in a fear of birth‐unit (often called Aurora clinic) where specially trained midwives and obstetricians work together to help the woman deal with negative experiences from childbirth and with fear of childbirth, or both. Although guidelines exist, the nature of counselling offered differs among hospitals in Sweden. However, the intention in all 4 study centres is to offer postpartum counselling if needed before the women leave the hospital. |
| Outcomes |
Time points for assessment: 4 and 6 weeks; 1, 2, 3, and 4 years Primary outcomes: Traumatic Events (TES), Depression (EPDS) Secondary outcomes: Satisfaction with Life (SWLS), Coping (WCQ), marriage and pre‐marriage assessment (ENRICH), Dyadic Adjustment (RDAS), Post‐partum bonding (PBQ), Health (EQ‐5D, SF‐36), Anxiety and Depression (HADS) |
| Starting date | The recruitment phase of this trial began in September 2013. The trial is now closed for inclusion but follow‐up assessment continues. |
| Contact information | |
| Notes | The Regional Research Council supported the study (Regionala Forskningsrådet, RFR, 368901, 308, 451, 480, 141; www.researchweb.org/is/sverige) and Swedish research council funding for clinical research in medicine. |
AAT: Approach Avoidance Tasks; ADNM: Adjustment Disorder – New Module; AFT: Assessment and Feedback Tool; ARM5: Agnew Relationship Measure – 5; AUD: alcohol use disorder; AUDIT: Alcohol Use Disorders Identification Test; AUDIT‐O: Alcohol Use Disorders Identification Test Outcomes; BVI: Bochumer Change Inventory; BVMT‐R: Brief Visuospatial Memory Test – Revised; CaDC: Cancer Distress Coach; CAGE: acronym for Cutting down, Annoyance by criticism, Guilty feeling, and Eye‐openers; CAPS: Clinician Administered PTSD Scale; CBT: cognitive behavioural therapy; CEQ: Credibility/Expectancy Questionnaire; CIWA‐Ar: Clinical Institute Withdrawal Assessment for Alcohol; CPT: cognitive processing therapy; CPT‐II: Conners' Continuous Performance Task II; CSD: Client Satisfaction with Device; CSQ: Client Satisfaction Questionnaire; CSRI: Client Service Receipt Inventory; CSS: Crisis Support Scale; CTQ: Childhood Trauma Questionnaire; CWIT: Color‐Word Interference Test; DAST: Dynamic Application Security Testing; DDT: Delay Discounting Task; DERS: Difficulties in Emotion Regulation Scale; DES: Dissociative Experiences Scale; DEVS: Distress and Endorsement Validation Scale; DKEFS: Delis‐Kaplan Executive Functions System; DoD: Department of Defense; DSI‐SS: Depressive Symptoms Index‐Suicidality Subscale; DSM: Diagnostic and Statistical Manual of Mental Disorders; EQ‐5D: EuroQol‐5D; EQ‐5DAL: EuroQol‐5D Activities of Daily Living; EMDR: eye movement desensitisation and reprocessing; ENRICH: Evaluation and Nurturing Relationship Issues, Communication and Happiness; EPDS: Edinburgh Postnatal Depression Scale; ERQ: Emotion Regulation Questionnaire; ESSI: Enriched Social Support Inventory; EUROHIS‐QOL: European Health Interview Survey‐Quality of Life; F20‐F29: schizophrenia, schizotypal and delusional, and other non‐mood psychotic disorders; FFMQ: Five Facet Mindfulness Questionnaire; GAD‐7: Generalised Anxiety Disorder 7 Item Scale; GSES: General Self‐Efficacy Scale; GSH: guided self‐help; HADS: Hospital Anxiety and Depression Scale; HSCL‐25: Hopkins Symptom Checklist‐25; HVLT: Hopkins Verbal Learning Test; I‐CBT: Internet‐based cognitive behavioural therapy; IAPT: Improving Access to Psychological Therapy; ICD‐10: International Classification of Diseases – 10th edition; IES: Impact of Event Scale; IES‐R: Impact of Event Scale – Revised; IGT: Iowa Gambling Task; IPF: Inventory of Psychosocial Functioning; ISI: Insomnia Severity Index; ITQ: International Trauma Questionnaire; IUS‐12: Intolerance of Uncertainty Scale Score; K10: Kessler 10; KFM: Kurzfragebogen für Medikamentengebrauch; LEC: Life Events Checklist; LSNS: Lubben Social Network Scale; MADRS‐S: Montgomery‐Åsberg Depression Rating Scale – Self‐report; MBI: Maslach Burnout Inventory; MINI: Mini‐International Neuropsychiatric Interview; MMSE: Mini Mental State Examination; MSPSS: Multidimensional Scale of Perceived Social Support; NED: no evidence of disease; NEO‐FFI: NEO‐Five Factor Inventory Neuroticism scale; NEQ: Negative Effects Questionnaire; OEF: Operation Enduring Freedom; OIF: Operation Iragi Freedom; PBQ: Postpartum Bonding Questionnaire; PCQ: Productivity Cost Questionnaire; PCL: PTSD Checklist; PCL‐C: PTSD Checklist – Civilian Version; PCL‐M: PTSD Checklist – Military Version; PCT: present‐centred therapy; PDS‐5: Posttraumatic Diagnostic Scale; PE: prolonged exposure; PET: positron emission tomography; PG‐13: Prolonged Grief Disorder‐13; PGI: Posttramatic Growth Inventory; PHQ: Patient Health Questionnaire; PMH: Positive Mental Health Scale; PMLDC: Post‐Migration Living Difficulties Checklist; PROMIS QOL: Patient‐Reported Outcomes Measurement Information System Quality of Life; PSQI: Pittsburgh Sleep Quality Index; PSS‐I: PTSD Symptom Scale – Interview; PSYCHLOPS: Psychological Outcome Profiles Instrument; PTCI: Posttraumatic Cognition Inventory; PTGI: Post Traumatic Growth Inventory; PTSD: post‐traumatic stress disorder; PWPQ: Perceptions of Web‐PE Questionnaire; QDS: Quick Drinking Screen; Q‐LES‐Q‐SF: Quality of Life Enjoyment and Satisfaction Questionnaire Short Form; RCT: randomised controlled trial; RDAS: Revised Dyadic Adjustment Scale; RIQ: Response to Intrusion Questionnaire; SBQ: Safety Behaviours Questionnaire; SCID: Structured Clinical Interview for DSM‐5; SDS: Sheehan Disability Scale; SEQ: Service Use Questionnaire; SF‐12: 12‐item Short Form; SF‐36: 36‐item Short Form; SIP: Short Inventory of Problems; SRI: Service Receipt Inventory; SRIEV: Service Receipt Inventory European Version; SSMIS: Self‐stigma of Mental Illness Scale; STAXI‐2: State‐Trait Anger Inventory‐2; SWLS: Satisfaction With Life Scale; TAM‐3: Technology Acceptance Model; TAU: treatment as usual; TBI: traumatic brain injury; TES: Traumatic Event Scale; TFCBT: individual trauma‐focused cognitive behavioural therapy; TFPT: trauma‐focused psychological therapy; TIC‐P: Trimbos and Institute of Medical Technology Assessment Cost Questionnaire for Psychiatry; TLFB: Timeline Follow‐Back; TMT: trauma management therapy; TMQ: Trauma Memory Questionnaire; TSDQ: Trait‐State Dissociation Questionnaire; TSQ: Trauma Screening Questionnaire; VA: Veterans Affairs; VR‐12: Veterans RAND 12‐Item Health Survey; WAIS: Wechsler Adult Intelligence Scale; WCQ: Ways of Coping Questionnaire; WCST: Wisconsin Card Sorting Test; WET: Written Exposure Therapy; WHO: World Health Organization; WHODAS: World Health Organization Disability Assessment Schedule; WMI: Working Memory Index; WSAS: Work and Social Adjustment Scale; WTAR: Wechsler Test of Adult Reading.
Differences between protocol and review
Attrition was removed as an outcome measure of adverse events to avoid duplication of dropout as an outcome measure.
Two review authors joined the review team since the last update (N Simon and S Dawson).
Contributions of authors
| Task | Completed by |
| Drafting the protocol | CL, AB, NR, JB |
| Development of a search strategy (in conjunction with CCMD's Information Specialist) | CL |
| Development of a search strategy (review update) | SD |
| Trial selection (original review) | CL, AB, arbitrated by NR |
| Trial selection (review update) | NS, LR, arbitrated by JB, CL, NR |
| Data extraction (original review) | CL, AB, arbitrated by NR |
| Data extraction (review update) | NS, LR, arbitrated by JB, CL, NR |
| 'Risk of bias' assessment (original review) | CL, AB, LR, arbitrated by NR |
| 'Risk of bias' assessment (review update) | NS, LR, arbitrated by JB, CL, NR |
| Data entry (original review) | CL, LR |
| Data entry (review update) | NS, LR |
| Analysis (original review) | CL |
| Analysis (review update) | NS, LR |
| Interpretation of analysis (original review) | CL, AB, NR, JB |
| Interpretation of analysis (review update) | NS, LR, JB, CL, NR |
| Drafting of final review (original review) | CL, AB, NR, JB, LR |
| Drafting of final review (review update) | NS, LR, JB, CL, NR, SD |
| Preparation of 'Summary of findings' tables (original review) | CL, LR |
| Preparation of 'Summary of findings' tables (review update) | NS, LR |
| Checking that final review meets all mandatory MECIR standards before submission (original review) | CL, AB, NR, JB, LR, |
| Checking that final review meets all mandatory MECIR standards before submission (review update) | NS, LR, JB, CL, NR, SD |
| Keeping the review up‐to‐date | NS, LR, CL, AB, NR, JB, SD |
Sources of support
Internal sources
Cardiff University, Other
National Centre of Mental Health (NCMH), Welsh Government, UK
External sources
-
National Institute for Health Research (NIHR), UK
LR and SD time on this review update was funded by Cochrane Infrastructure funding to the Common Mental Disorders Cochrane Review Group
Declarations of interest
NS: is involved in the data collection of one ongoing study (Nollett 2018). If this study is completed and included in future updates of the review, NS will not be involved in data extraction and risk of bias assessments for this study.
LR: none.
CL: involved in the development and evaluation of an online, guided self‐help programme (Spring: a step‐by‐step treatment programme for PTSD) in conjunction with the software development company Healthcare Learning Smile‐on.
NR: involved in the development and evaluation of an online, guided self‐help programme (Spring: a step‐by‐step treatment programme for PTSD) in conjunction with the software development company Healthcare Learning Smile‐on.
AB: none.
SD: none.
JB: involved in the development and evaluation of an online, guided self‐help programme (Spring: a step‐by‐step treatment programme for PTSD) in conjunction with the software development company Healthcare Learning Smile‐on.
New search for studies and content updated (no change to conclusions)
References
References to studies included in this review
Allen 2020 {published data only}
- ACTRN12614001213639. Internet treatment for posttraumatic stress disorder (PTSD). www.anzctr.org.au/ACTRN12614001213639.aspx (first received 18 November 2014.
- Allen A, Loughnan S, Smith J, Hobbs M, Newby J, Mahoney A, et al. Efficacy of iCBT for posttraumatic stress disorder: a randomized controlled trial. Manuscript submitted for publication 2020.
- Allen AR, Newby JM, Smith J, Andrews G. Internet-based cognitive behavioural therapy (iCBT) for posttraumatic stress disorder versus waitlist control: study protocol for a randomised controlled trial. Trials 2015;16:544. [DOI] [PMC free article] [PubMed] [Google Scholar]
Bragesjö 2020 {unpublished data only}
- Bragesjö M, Arnberg FK, Särnholm J, Olofsdotter LK, Andersson E (accepted). Condensed Internet-delivered prolonged exposure provided soon after trauma: a randomised pilot trial. Internet Interventions. [DOI] [PMC free article] [PubMed]
Engel 2015 {published data only}
- Engel CC, Litz B, Magruder KM, Harper E, Gore K, Stein N, et al. Delivery of self training and education for stressful situations (DESTRESS-PC): a randomized trial of nurse assisted online self-management for PTSD in primary care. General Hospital Psychiatry 2015;37(4):323-8. [NCT01474057] [DOI] [PMC free article] [PubMed] [Google Scholar]
Ivarsson 2014 {published data only}
- Ivarsson D, Blom M, Hesser H, Carlbring P, Enderby P, Nordberg R, et al. Guided Internet-delivered cognitive behavior therapy for post-traumatic stress disorder: a randomized controlled trial. Internet Interventions 2014;1:35-40. [Google Scholar]
Knaevelsrud 2015 {published data only}12611001019998
- Knaevelsrud C, Brand J, Lange A, Ruwaard J, Wagner B. Web-based psychotherapy for posttraumatic stress disorder in war-traumatized Arab patients: randomized controlled trial. Journal of Medical Internet Research 2015;17(3):e71. [DOI] [PMC free article] [PubMed] [Google Scholar]
Krupnick 2017 {published data only}
- Krupnick JL, Green BL, Amdur R, Alaoui A, Belouali A, Roberge E, et al. An Internet-based writing intervention for PTSD in veterans: a feasibility and pilot effectiveness trial. Psychological Trauma : Theory, Research, Practice and Policy 2017;9(4):461-70. [DOI] [PubMed] [Google Scholar]
Kuhn 2017 {published data only}
- Kuhn E, Kanuri N, Hoffman JE, Garvet DW, Ruzek JI. A randomized controlled trial of a smartphone app for posttraumatic stress disorder symptoms. Journal of Consulting and Clinical Psychology 2017;85(3):267-73. [DOI] [PubMed] [Google Scholar]
- NCT02445196. PTSD coach app evaluation. ClinicalTrials.gov/show/NCT02445196 (first received 15 May 2015).
Lewis 2017 {published data only}
Littleton 2016 {published data only}
- Littleton H, Grills A. Changes in coping and negative cognitions as mechanisms of change in online treatment for rape‐related posttraumatic stress disorder. Journal of Traumatic Stress 2019;32(6):927-35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Littleton H, Grills AE, Kline KD, Schoemann AM, Dodd JC. The From Survivor to Thriver program: RCT of an online therapist-facilitated program for rape-related PTSD. Journal of Anxiety Disorders 2016;43:41-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NCT02777294. Evaluation of web-based CBT for rape victims. ClinicalTrials.gov/show/NCT02777294 (first received 19 May 2016).
Litz 2007 {published data only}
- Litz B, Engel CC, Bryant R, Papa A. A randomized, controlled proof of concept trial of an Internet-based, therapist-assisted self-management treatment for posttraumatic stress disorder. American Journal of Psychiatry 2007;164:1676-83. [DOI] [PubMed] [Google Scholar]
McLean 2020a {published data only}
- McLean CP, Foa EB, Dondanville KA, Haddock CK, Miller ML, Rauch SA, et al, for the STRONG STAR Consortium. The effects of web-prolonged exposure among military personnel and veterans with posttraumatic stress disorder. Psychological Trauma: Theory, Research, Practice and Policy 2020 Nov 19 [Epub ahead of print]. [DOI: 10.1037/tra0000978] [DOI] [PubMed]
- McLean CP, Rauch SA, Foa EB, Sripada RK, Tannahill HS, Mintz J, et al. Design of a randomized controlled trial examining the efficacy and biological mechanisms of web-prolonged exposure and present-centered therapy for PTSD among active-duty military personnel and veterans. Contemporary Clinical Trials 2017;64:41-8. [DOI] [PubMed] [Google Scholar]
- NCT02556645. A comparison of web-prolonged exposure (Web-PE) and present-centered therapy (PCT) for PTSD among active-duty military personnel. ClinicalTrials.gov/show/NCT02556645 (first received 22 September 2015).
Miner 2016 {published data only}
- Miner A, Kuhn E, Hoffman JE, Owens JE, Ruzik JI. Feasibility, acceptability, and potential efficacy of the PTSD Coach App: a pilot randomized controlled trial with community trauma survivors. Psychological Trauma : Theory, Research, Practice and Policy 2016;8(3):384-92. [DOI] [PubMed] [Google Scholar]
Spence 2011 {published data only}12611000989943
- Spence J, Titov N, Dear BF, Johnston L, Solley K, Lorian C. Randomized controlled trial of Internet-delivered cognitive behavioral therapy for post-traumatic stress disorder. Depression and Anxiety 2011;28(7):541-50. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Bahena 2016 {published data only}
- Bahena S. Efficacy of a mobile application among a sample of veterans with symptoms of post-traumatic stress disorder. Dissertation Abstracts International 2016;77:2. [Google Scholar]
Bishop 2012 {published data only}
- Bishop TM, Possemato K, Acosta M, Lantinga LJ, Maisto SA, Marsch L, et al. Moving forward: update on the development of a web-based cognitive behavioral treatment for OEF/OIF veterans with PTSD symptoms and substance misuse. Alcoholism, Clinical and Experimental Research 2012;36:347A. [Google Scholar]
Blankenship 2013 {published data only}
- Blankenship AE. A prospective examination of mindfulness training on the mitigation of posttraumatic stress symptoms. Dissertations 2013:161. [scholarworks.wmich.edu/dissertations/161]
Bottche 2016 {published data only}
- Bottche M, Kuwert P, Pietrzak RH, Knaevelsrud C. Predictors of outcome of an Internet-based cognitive-behavioural therapy for post-traumatic stress disorder in older adults. Psychology and Psychotherapy 2016;89(1):82-96. [DOI] [PubMed] [Google Scholar]
Brief 2012 {published data only}
- Brief D, Rubin A, Enggasser J, Roy M, Lachowicz M, Helmuth E, et al. Web-based intervention for returning veterans with risky alcohol use abstract. Alcoholism, Clinical and Experimental Research 2012;36:347A. [Google Scholar]
Brief 2013 {published data only}
- Brief DJ, Rubin A, Keane TM, Enggasser JL, Roy M, Helmuth E, et al. Web intervention for OEF/OIF veterans with problem drinking and PTSD symptoms: a randomized clinical trial. Journal of Consulting Psychology 2013;81:890-900. [DOI] [PMC free article] [PubMed] [Google Scholar]
Daggett 2014 {published data only}
- Daggett V, Bakas T, Murray L, Woodward-Hagg H, Williams JG, Maddox K, et al. Feasibility and satisfaction with the VETeranS Compensate, Adapt, REintegrate (VETS-CARE) intervention. Brain Injury 2014;28(5-6):554-5. [Google Scholar]
de Kleine 2019 {published data only}
- de Kleine. Appraisal-based cognitive bias modification in patients with posttraumatic stress disorder: a randomised clinical trial. European Journal of Pscyhotraumatology 2019;10(1):1625690. [DOI] [PMC free article] [PubMed] [Google Scholar]
Elbers 2013 {published data only}
- Elbers NA, Akkermans AJ, Cuijpers P, Bruinvels DJ. Effectiveness of a web-based intervention for injured claimants: a randomized controlled trial. Trials 2013;14:227. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hirai 2005 {published data only}
- Hirai M, Clum GA. An Internet-based self-change programme for traumatic event related fear, distress and maladaptive coping. Journal of Traumatic Stress 2005;18:631-6. [DOI] [PubMed] [Google Scholar]
Hirai 2012 {published data only}
- Hirai M, Skidmore ST, Clum GA, Dolma S. An investigation of the efficacy of online expressive writing for trauma-related psychological distress in Hispanic individuals. Behavior Therapy 2012;43:812-24. [DOI] [PubMed] [Google Scholar]
Kersting 2011 {published data only}
- Kersting A, Kroker K, Schlicht S, Baust K, Wagner B. Efficacy of cognitive behavioral Internet-based therapy in parents after the loss of a child during pregnancy: pilot data from a randomized controlled trial. Archives of Women's Mental Health 2011;14:465-77. [DOI] [PubMed] [Google Scholar]
Kersting 2013 {published data only}
- Kersting A, Dolemeyer R, Steinig J, Walter F, Kroker K, Baust K, et al. Brief Internet-based intervention reduces posttraumatic stress and prolonged grief in parents after the loss of a child during pregnancy: a randomized controlled trial. Psychotherapy and Psychosomatics 2013;82:372-81. [DOI] [PubMed] [Google Scholar]
Knaevelsrud 2006 {published data only}
- Knaevelsrud C, Maercker A. Does the quality of the working alliance predict treatment outcome in online psychotherapy for traumatized patients? Journal of Medical Internet Research 2006;8(4):e31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knaevelsrud C, Maercker A. Internet-based treatment for PTSD reduces distress and facilitates the development of a strong therapeutic alliance: a randomized controlled clinical trial. BMC Psychiatry 2007;7:13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knaevelsrud C, Wagner B, Maercker A. Internet-based treatment of posttraumatic stress: does the working alliance predict long-term therapy outcome. In: Joint Meeting of the European & UK Chapters of the Society for Psychotherapy Research; 2005 Mar 2-5; Lausanne. 2005.
Knaevelsrud 2010a {published data only}
- Knaevelsrud C, Liedl A, Maercker A. Posttraumatic growth, optimism and openness as outcomes of a cognitive-behavioural intervention for posttraumatic stress reactions. Journal of Health Psychology 2010;15:1030-8. [DOI] [PubMed] [Google Scholar]
Knaevelsrud 2010b {published data only}
- Knaevelsrud C, Maercker A. Long-term effects of an Internet-based treatment for posttraumatic stress. Cognitive Behaviour Therapy 2010;39:72-7. [DOI] [PubMed] [Google Scholar]
- NCT01508377. Effects of an Internet-based intervention for posttraumatic stress disorder. clinicaltrials.gov/ct2/show/NCT01508377 (first received 22 December 2011).
Knaevelsrud 2017 {published data only}12608000259347
- Knaevelsrud C, Bottche M, Pietrzak RH, Freyberger HJ, Kuwert P. Efficacy and feasibility of a therapist-guided Internet-based intervention for older persons with childhood traumatization: a randomized controlled trial. American Journal of Geriatric Psychiatry 2017;25(8):878-88. [DOI] [PubMed] [Google Scholar]
Lange 2001 {published data only}
- Lange A, de Ven JP, Schrieken B, Emmelkamp PM. Interapy. Treatment of posttraumatic stress through the Internet: a controlled trial. Journal of Behavior Therapy and Experimental Psychiatry 2001;32(2):73-90. [DOI] [PubMed] [Google Scholar]
Lange 2003 {published data only}
- Lange A, Rietdijk D, Hudcovicova M, de Ven JP, Schrieken B, Emmelkamp PM. Interapy: a controlled randomized trial of the standardized treatment of posttraumatic stress through the Internet. Journal of Consulting and Clinical Psychology 2003;71(5):901-9. [DOI] [PubMed] [Google Scholar]
- Lange A, de Ven JP, Schrieken B. Interapy: treatment of post-traumatic stress via the Internet. Cognitive Behaviour Therapy 2003;32(3):110-24. [DOI] [PubMed] [Google Scholar]
Maercker 2004 {published data only}
- Maercker A, Knaevelsrud C, Wagner B, Lange A. Intherapy: an Internet-based treatment trial of CBT of traumatic stress. In: Twentieth Annual Meeting, International Society for Traumatic Stress Studies; 2004 Nov 14-18; New Orleans (LA). 2004.
McCleary 2019 {published data only}
- McLeary H, Cordova M. Development of Efficacy for Individualized Coaching for Improving Mobile App Engagement and Outcomes for Veterans with Posttraumatic Stress Disorder. Palo Alto (CA): Pacific Graduate School of Psychology, Palo Alto University, 2019. [Google Scholar]
McGlinchey 2014 {published data only}
- McGlinchey R, Rosenblatt A, Mercado R, Esterman M, DeGutis J. Internet-based cognitive training enhances attention and functional outcomes in OEF/OIF/OND Veterans abstract. In: 10th World Congress on Brain Injury of the International Brain Injury Association; 2014 Mar 19-22; San Francisco (CA). 2014.
NCT01508377 {published data only}
- NCT01508377. Effects of an Internet-based intervention for posttraumatic stress disorder. clinicaltrials.gov/ct2/show/NCT01508377 (first received 22 December 2011).
NCT01552278 {published data only}
- NCT01552278. Enhancing cognitive function and reintegration in Iraq and Afghanistan veterans with PTSD using computer-based cognitive training. clinicaltrials.gov/show/NCT01552278 (first received 15 March 2010).
NCT01678196 {published data only}
- NCT01678196. Helping families help veterans with PTSD and alcohol abuse: an RCT of VA-CRAFT. clinicaltrials.gov/ct2/show/NCT01678196 (first received 3 September 2012).
NCT01710943 {published data only}
- NCT01710943. Web-based CBT for recent veterans experiencing problems with trauma symptoms or alcohol/drug use. clinicaltrials.gov/show/NCT01710943 (first received 19 October 2012).
NCT01760213 {published data only}
- NCT01760213. Brief Internet based treatment for PTSD. clinicaltrials.gov/show/NCT01760213 (first received 4 January 2013).
NCT01891734 {published data only}
- NCT01891734. Enhancing delivery of problem solving therapy using SmartPhone technology. clinicaltrials.gov/show/NCT01891734 (first received 25 June 2013).
NCT03208738 {published data only}
- NCT03208738. Pilot evaluation of the VetChange mobile app for veterans with PTSD who engage in problem drinking. clinicaltrials.gov/show/NCT03208738 (first received 5 July 2017).
NCT03733028 {published data only}
- NCT03733028. Mobile intervention for Veterans with PTSD and anger. clinicaltrials.gov/ct2/show/NCT03733028 (first received 7 November 2018).
NCT04362358 {published data only}
- NCT04362358. Online cognitive behavioral therapy (CBT) for stress disorders in health workers involved in the care of patients during the COVID-19 epidemic (REST). clinicaltrials.gov/ct2/show/NCT04362358 (first received 24 April 2020).
Niemeyer 2020 {published data only}
- Niemeyer H, Knaevelsrud C, Schumacher S, Engel S, Kuester A, Burchert S, et al. Evaluation of an Internet-based intervention for service members of the German armed forces with deployment-related posttraumatic stress symptoms. BMC Psychiatry 2020;20:205. [DOI] [PMC free article] [PubMed] [Google Scholar]
Nieminen 2016 {published data only}
- Nieminen K, Berg I, Frankenstein K, Vilta L, Larsson K, Persson U, et al. Internet-provided cognitive behaviour therapy of posttraumatic stress symptoms following childbirth – a randomized controlled trial. Cognitive Behaviour Therapy 2016;45(4):287-306. [DOI] [PubMed] [Google Scholar]
Possemato 2010 {published data only}
- NCT02611843. Peer supported web-based CBT for OEF/OIF veterans with PTSD and substance misuse. clinicaltrials.gov/show/NCT02611843 (first received 23 November 2015).
- Possemato KA, Ouimette PC, Geller PA. Internet-based expressive writing for kidney transplant recipients: effects on posttraumatic stress and quality of life. Traumatology 2010;16:49-54. [Google Scholar]
Possemato 2011 {published data only}
- Possemato K, Ouimette P, Knowlton P. A brief self-guided telehealth intervention for post-traumatic stress disorder in combat veterans: a pilot study. Journal of Telemedicine and Telecare 2011;17:245-50. [DOI] [PubMed] [Google Scholar]
Sayer 2009 {published data only}
- NCT00640445. Military to civilian: RCT of an intervention to promote postdeployment reintegration. clinicaltrials.gov/show/NCT00640445 (first received 18 March 2008).
- Sayer NA, Orazem R, Frazier P, Noorbaloochi S, Schnurr P, Litz B, et al. Military to civilian: RCT of a web-based expressive writing intervention. In: Military Health Research Forum; 2009 Aug 31-Sep 3; Kansas City (MO). 2009.
Smith 2019 {published data only}
- Smith SK. Evolution of a mobile app to manage cancer-related post-traumatic stress. Psycho-oncology 2019;28:19. [Google Scholar]
Solzbacher 2012 {published data only}
- Solzbacher S, Rueddel H. Effectiveness of E-therapy skill training in inpatients with anorexia nervosa, PTSD or personality disorders. Psychosomatic Medicine 2012;74(3):A28-9. [Google Scholar]
Steinmetz 2012 {published data only}
- Steinmetz SE, Benight CC, Bishop SL, James LE. My disaster recovery: a pilot randomized controlled trial of an Internet intervention. Anxiety, Stress, and Coping 2012;25:593-600. [DOI] [PMC free article] [PubMed] [Google Scholar]
Stockton 2014 {published data only}
- Stockton H, Joseph S, Hunt N. Expressive writing and posttraumatic growth: an Internet-based study. Traumatology 2014;20:75-83. [Google Scholar]
Wagner 2007 {published data only}
- Wagner B, Knaevelsrud C, Maercker A. Post-traumatic growth and optimism as outcomes of an Internet-based intervention for complicated grief. Cognitive Behaviour Therapy 2007;36:156-61. [DOI] [PubMed] [Google Scholar]
Wagner 2012 {published data only}
- Wagner B, Brand J, Schulz W, Knaevelsrud C. Online working alliance predicts treatment outcome for posttraumatic stress symptoms in Arab war-traumatized patients. Depression and Anxiety 2012;29:646-51. [DOI] [PubMed] [Google Scholar]
Wang 2013 {published data only}
- Wang Z, Wang J, Maercker A. Chinese my trauma recovery, a Web-based intervention for traumatized persons in two parallel samples: randomized controlled trial. Journal of Medical Internet Research 2013;15:e213. [DOI] [PMC free article] [PubMed] [Google Scholar]
Zernicke 2014 {published data only}
- Zernicke KA, Campbell TS, Speca M, McCabe-Ruff K, Flowers S, Carlson LE. A randomized wait-list controlled trial of feasibility and efficacy of an online mindfulness-based cancer recovery program: the eTherapy for cancer applying mindfulness trial. Psychosomatic Medicine 2014;76(4):257-67. [DOI] [PubMed] [Google Scholar]
References to ongoing studies
ACTRN12611000989943 {published data only}
- ACTRN12611000989943. A comparison of Internet-based cognitive behavioural therapy for posttraumatic stress disorder with and without exposure: a randomized controlled trial. www.anzctr.org.au/ACTRN12611000989943.aspx (first received 16 September 2011).
ACTRN12616000956404 {published data only}
- ACTRN12616000956404. Internet-based intervention for posttraumatic stress disorder (PTSD) in soldiers: exploring mechanisms of treatment outcome. www.anzctr.org.au/ACTRN12616000956404.aspx (first received 20 July 2016).
DRKS00010245 {published data only}
- DRKS00010245. Evaluation of web-based cognitive and behavioural therapeutic components and its change mechanisms for Arab people with posttraumatic stress disorder. www.drks.de/DRKS00010245 (first received 30 March 2016).
DRKS00016931 {published data only}
- Internet-based therapy for physicians with post-traumatic stress. Ongoing study. 2019. Contact author for more information.
DRKS00017838 {published data only}
- Feasibility, acceptance and effectiveness of Step-by-Step, a smartphone-based self-help program for Syrian refugees: a pilot study. Ongoing study. Starting date of trial not provided. Contact author for more information.
DRKS00020266 {published data only}
- Online-program after adverse childhood experiences in GDR children's homes. A randomized controlled trial. Ongoing study. January 2020. Contact author for more information.
Ehlers 2020 {published data only}
- Ehlers A, Wild J, Warnock-Parkes E, Grey N, Murray H, Kerr A. A randomised controlled trial of therapist-assisted online psychological therapies for posttraumatic stress disorder (STOP-PTSD): trial protocol. Trials 2020;21(1):355. [DOI] [PMC free article] [PubMed] [Google Scholar]
Golchert 2019 {published data only}
- Golchert J, Roehr S, Berg F, Grochtdreis T, Hoffman R, Jung F, et al. HELP@APP: development and evaluation of a self-help app for traumatized Syrian refugees in Germany – a study protocol of a randomized controlled trial. BMC Psychiatry 2019;19:131. [DOI] [PMC free article] [PubMed] [Google Scholar]
ISRCTN16806208 {published data only}
- ISRCTN16806208. A randomised controlled trial of therapist-assisted online psychological therapies for post-traumatic stress disorder. isrctn.com/ISRCTN16806208 (first received 5 January 2018).
Lehavot 2017 {published data only}
- Lehavot K, Litz B, Millard SP, Hamilton AB, Sadler A, Simpson T. Study adaptation, design, and methods of a web-based PTSD intervention for women Veterans. Contemporary Clinical Trials 2017;53:68-79. [DOI] [PubMed] [Google Scholar]
McLean 2020b {published data only}
- McLean CP, Miller ML, Gengler R, Henderson J, Sloan DM. The efficacy of written exposure therapy versus imaginal exposure delivered online for posttraumatic stress disorder: design of a randomized controlled trial in Veterans. Contemporary Clinical Trials 2020;91:105990. [DOI] [PubMed] [Google Scholar]
NCT02929979 {published data only}
- NCT02929979. Cognitive remediation for alcohol use disorder and posttraumatic stress disorder. clinicaltrials.gov/show/NCT02929979 (first received 11 October 2016).
NCT04058795 {published data only}
- PTSD mobile app for cancer survivors. Ongoing study. September 2020. Contact author for more information.
NCT04094922 {published data only}
- PTSD Coach Sweden: evaluating a self-help mobile app for posttraumatic stress in a community sample. Ongoing study. May 2019. Contact author for more information.
NCT04101942 {published data only}
- An Internet-delivered cognitive-behavioral intervention provided soon after trauma: a RCT. Ongoing study. Starting date of trial not provided. Contact author for more information.
NCT04155736 {published data only}
- Testing a self-management app for symptoms of posttraumatic stress. Ongoing study. January 2020. Contact author for more information.
NCT04286165 {published data only}
- Brief Peer Supported webSTAIR Compared to Enhanced Wait List (BPSwS). Ongoing study. January 2020. Contact author for more information.
NCT04333667 {published data only}
- Effectiveness of mindfulness-based Internet intervention Still Me (Still-Me). Ongoing study. April 2020. Contact author for more information.
Nollett 2018 {published data only}
- ISRCTN13697710. A study of trauma-focused online guided self help versus trauma-focused cognitive behavioural therapy for post-traumatic stress disorder. isrctn.com/ISRCTN13697710 (first received 21 December 2016).
- Nollett C, Lewis C, Kitchiner N, Roberts N, Addison K, Brookes-Howell L, et al. Pragmatic RAndomised controlled trial of a trauma-focused guided self-help Programme versus InDividual trauma-focused cognitive Behavioural therapy for post-traumatic stress disorder (RAPID): trial protocol. BMC Psychiatry 2018;18(1):77. [DOI] [PMC free article] [PubMed] [Google Scholar]
NTR6912 {published data only}
- NTR6912. Addition of the SUPPORT coach in PTSD treatment. www.trialregister.nl/trialreg/admin/rctview.asp?TC=6912 (first received 22 December 2017).
Sjomark 2018 {published data only}
- Sjomark J, Parling T, Jonsson M, Larsson M, Skoog Svanberg A. A longitudinal, multi-centre, superiority, randomized controlled trial of Internet-based cognitive behavioural therapy (iCBT) versus treatment-as-usual (TAU) for negative experiences and posttraumatic stress following childbirth: the JUNO study protocol. BMC Pregnancy & Childbirth 2018;18(1):387. [DOI] [PMC free article] [PubMed] [Google Scholar]
Additional references
ACPMH 2007
- Phoenix Australia – Centre for Posttraumatic Mental Health. Australian guidelines for the treatment of adults with acute stress disorder and posttraumatic stress disorder, 2007. phoenixaustralia.org/resources/ptsd-guidelines/ (accessed 6 June 2017).
Andersson 2005
- Andersson G, Bergstrom J, Carlbring P, Lindefors N. The use of the Internet in the treatment of anxiety disorders. Current Opinion in Psychiatry 2005;18:73-7. [PubMed] [Google Scholar]
Andersson 2009
- Andersson G. Using the Internet to provide cognitive behaviour therapy. Behaviour Research and Therapy 2009;47(3):175-80. [DOI] [PubMed] [Google Scholar]
Andersson 2016
- Andersson G. Internet-delivered psychological treatments. Annual Review of Clinical Psychology 2016;12:157-79. [DOI] [PubMed] [Google Scholar]
Andersson 2018
- Andersson G. Past, present and future. Internet interventions: the application of information technology in mental and behavioural health. Internet Interventions 2018;12:181-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Andersson 2019
- Andersson G, Carlbring P, Titov N, Lindefors N. Internet interventions for adults with anxiety and mood disorders: a narrative umbrella review of recent meta-analyses. Canadian Journal of Psychiatry 2019;64:465-70. [DOI] [PMC free article] [PubMed] [Google Scholar]
Andrews 2013
- Andrews J, Guyatt G, Oxman AD, Alderson P, Dahm P, Falck-Ytter Y, et al. GRADE guidelines: 14. Going from evidence to recommendations: the significance and presentation of recommendations. Journal of Clinical Epidemiology 2013;66:719-25. [DOI] [PubMed] [Google Scholar]
APA 1980
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-III). Washington (DC): American Psychiatric Association, 1980. [Google Scholar]
APA 1987
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-IIIR). 3rd edition. Washington (DC): American Psychiatric Association, 1987. [Google Scholar]
APA 1994
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th edition. Washington (DC): American Psychiatric Association, 1994. [Google Scholar]
APA 2000
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM IV). 4th edition. Washington (DC): American Psychiatric Association, 2000. [Google Scholar]
APA 2013
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5). 5th edition. Washington (DC): American Psychiatric Association, 2013. [Google Scholar]
APA 2017
- American Psychiatric Association. Clinical practice guideline for the treatment of posttraumatic stress disorder (PTSD) in adults. Washington (DC): American Psychiatric Association, 2017. [Google Scholar]
Atwoli 2013
- Atwoli L, Stein DJ, Williams DR, Mclaughlin KA, Petukhova M, Kessler RC, et al. Trauma and posttraumatic stress disorder in South Africa: analysis from the South African Stress and Health Study. BMC Psychiatry 2013;13:182. [DOI] [PMC free article] [PubMed] [Google Scholar]
Beck 1961
- Beck AT, Ward CH. An inventory for measuring depression. Archives of General Psychiatry 1961;4:561-71. [DOI] [PubMed] [Google Scholar]
Beck 1993
- Beck AT, Steer RA. Beck Anxiety Inventory Manual. San Antonio (TX): The Psychological Corporation, 1993. [Google Scholar]
Békés 2020
- Békés V, Aafjes-Van Doorn K. Psychotherapists' attitudes toward online therapy during the COVID-19 pandemic. Journal of Psychotherapy Integration 2020;30:238-47. [Google Scholar]
Berger 2017
- Berger T. The therapeutic alliance in Internet interventions: a narrative review and suggestions for future research. Psychotherapy Research 2017;27:511-24. [DOI] [PubMed] [Google Scholar]
Bisson 2013
- Bisson JI, Roberts NP, Andrew M, Cooper R, Lewis CE. Psychological therapies for chronic post-traumatic stress disorder (PTSD) in adults. Cochrane Database of Systematic Reviews 2013, Issue 12. Art. No: CD003388. [DOI: 10.1002/14651858.CD003388.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]
Bisson 2019
- Bisson JI, Berliner L, Cloitre M, Forbes D, Jensen TK, Lewis C, et al. The International Society for Traumatic Stress Studies new guidelines for the prevention and treatment of posttraumatic stress disorder: methodology and development process. Journal of Traumatic Stress 2019;32(4):475-83. [DOI] [PubMed] [Google Scholar]
Blake 1995
- Blake DD, Weathers FW, Nagy LM, Kaloupek DG, Gusman FD, Charney DS. The development of a clinician-administered PTSD scale. Journal of Traumatic Stress 1995;8:75-90. [DOI] [PubMed] [Google Scholar]
Bonanno 2015
- Bonanno GA, Romero SA, Klein SI. The temporal elements of psychological resilience: an integrative framework for the study of individuals, families, and communities. Psychological Inquiry 2015;26:139-69. [Google Scholar]
Breh 2007
- Breh DC, Seidler GH. Is peritraumatic dissociation a risk Factor for PTSD? Journal of Trauma & Dissociation 2007;8:53-69. [DOI] [PubMed] [Google Scholar]
Brewin 2000
- Brewin CR, Andrews B, Valentine JD. Meta-analysis of risk factors for posttraumatic stress disorder in trauma-exposed adults. Journal of Consulting and Clinical Psychology 2000;68(5):748-66. [DOI] [PubMed] [Google Scholar]
Brown 2001
- Brown TA, Campbell LA, Lehman CL, Grisham JR, Mancill RB. Current and lifetime comorbidity of the DSM-IV anxiety and mood disorders in a large clinical sample. Journal of Abnormal Psychology 2001;110:585-99. [DOI] [PubMed] [Google Scholar]
Bryant 2003
- Bryant RA, Moulds ML, Guthrie RM, Dang ST, Nixon RD. Imaginal exposure alone and imaginal exposure with cognitive restructuring in treatment of posttraumatic stress disorder. Journal of Consulting & Clinical Psychology 2003;71(4):706-12. [DOI] [PubMed] [Google Scholar]
Bryant 2015
- Bryan RA, Nickerson A, Creamer M, O'Donnell M, Forbes D, Galatzer-Levy I, et al. Trajectory of post-traumatic stress following traumatic injury: 6-year follow-up. British Journal of Psychiatry 2015;206(5):417-23. [DOI] [PubMed] [Google Scholar]
Carlbring 2017
- Carlbring P, Andersson G, Cuijpers P, Riper H, Hedman-Lagerof E. Internet-based vs. face-to-face cognitive behavior therapy for psychiatric and somatic disorders: an updated systematic review and meta-analysis. Cognitive Behaviour Therapy 2017;47:1-18. [DOI] [PubMed] [Google Scholar]
Carmassi 2014
- Carmassi C, Dell'Osso L, Manni C, Candini V, Dagani J, Iozzino L, et al. Frequency of trauma exposure and post-traumatic stress disorder in Italy: analysis from the World Mental Health Survey Initiative. Journal of Psychiatric Research 2014;59:77-84. [DOI] [PMC free article] [PubMed] [Google Scholar]
Cattane 2017
- Cattane N, Rossi R, Lanfredi M, Cattaneo A. Borderline personality disorder and childhood trauma: exploring the affected biological systems and mechanisms. BMC Psychiatry 2017;17:221-41. [DOI] [PMC free article] [PubMed] [Google Scholar]
Creamer 2001
- Creamer M, Burgess P, McFarlane AC. Post-traumatic stress disorder: findings from the Australian national survey of mental health and well-being. Psychological Medicine 2001;31:1237-47. [DOI] [PubMed] [Google Scholar]
Creamer 2004
- Creamer M, Forbes D, Phelps A, Humphreys L. Treating Traumatic Stress: Conducting Imaginal Exposure. Heidelberg West (Vic): Australian Centre for Posttraumatic Mental Health, 2004. [Google Scholar]
Cuijpers 2009
- Cuijpers P, Marks IM, Straten A, Cavanagh K, Gega l, Andersson G. Computer-aided psychotherapy for anxiety disorders: a meta-analytic review. Cognitive Behaviour Therapy 2009;38:66-82. [DOI] [PubMed] [Google Scholar]
Cuijpers 2010
- Cuijpers P, Donker T, Straten A, Li J. Is guided self-help as effective as face-to-face psychotherapy for depression and anxiety disorders? A systematic review and meta-analysis of comparative outcome studies. Psychological Medicine 2010;40(12):1943-57. [DOI] [PubMed] [Google Scholar]
Department of Veteran Affairs 2017
- Department of Veteran Affairs and Department of Defense. VA/DoD Clinical Practice Guideline for the Management of Posttraumatic Stress Disorder and Acute Stress Disorder. Washington (DC): Management of Post-Traumatic Stress Working Group, 2017. [Google Scholar]
Duncan 2018a
- Duncan L, Cooper B, Shen H. Robust findings from 25 years of PTSD genetics research. Current Psychiatry Reports 2018;20:1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
Duncan 2018b
- Duncan L, Ratanatharathorn A, Aiello AE, Almili L, Amstader AB, Ashley-Koch AE, et al. Largest GWAS of PTSD (N=20070) yields genetic overlap with schizophrenia and sex differences in heritability. Molecular Psychiatry 2018;23:666-73. [DOI] [PMC free article] [PubMed] [Google Scholar]
Ebert 2018
- Ebert DD, Van Daele T, Nordgreen T, Karekla M, Compare A, Zarbo C, et al. Internet- and mobile-based psychological interventions: applications, efficacy, and potential for improving mental health a report of the EFPA E-Health Taskforce. European Psychologist 2018;23:167-87. [Google Scholar]
Ehlers 2000
- Ehlers A, Clark DM. A cognitive model of posttraumatic stress disorder. Behaviour Research and Therapy 2000;38:319-45. [DOI] [PubMed] [Google Scholar]
Ferry 2008
- Ferry F, Bolton D, Bunting B, Devine B, McCann S, Murphy S. Trauma, Health and Conflict. A Study of the Epidemiology of Trauma Related Disorders and Qualitative Investigation of the Impact of Trauma on the Individual. Omagh (Northern Ireland): Northern Ireland Centre for Trauma and Transformation, 2008. [Google Scholar]
Ferry 2014
- Ferry F, Bunting B, Murphy S, O'Neill S, Stein D, Koenen K. Traumatic events and their relative PTSD burden in Northern Ireland: a consideration of the impact of the 'Troubles'. Social Psychiatry and Psychiatric Epidemiology 2014;49:435-46. [DOI] [PubMed] [Google Scholar]
Ferry 2015
- Ferry FR, Brady SE, Bunting BP, Murphy SD, Bolton D, O'Neill SM. The economic burden of PTSD in Northern Ireland. Journal of Traumatic Stress 2015;28:191-7. [DOI] [PubMed] [Google Scholar]
Foa 1998
- Foa EB, Rothbaum BO. Treating the Trauma of Rape: Cognitive-behavioral Therapy for PTSD. New York (NY): Guilford Press, 1998. [Google Scholar]
Foa 2007
- Foa E, Hembree E, Rothbaum BO. Prolonged Exposure Therapy for PTSD: Emotional Processing of Traumatic Experiences Therapist Guide. Oxford, UK: Oxford University Press, 2007. [Google Scholar]
Foa 2008
- Foa E, Keane TM, Friedman MJ, Cohen JA. Effective Treatments for PTSD: Practice Guidelines From the International Society for Traumatic Stress Studies. New York (NY): Guilford Press, 2008. [Google Scholar]
Gega 2004
- Gega L, Marks I, Mataix-Cols D. Computer-aided CBT self-help for anxiety and depressive disorders: experience of a London clinic and future directions. Journal of Clinical Psychology 2004;60(2):147-57. [DOI] [PubMed] [Google Scholar]
Gibbons 2011
- Gibbons MC, Fleisher L, Slamon RE, Bass S, Kandadai V, Beck JR. Exploring the potential of Web 2.0 to address health disparities. Journal of Health Communication 2011;16:77-89. [DOI] [PubMed] [Google Scholar]
Gillies 2012
- Gillies D, Taylor F, Gray C, O'Brien L, D'Abrew N. Psychological therapies for the treatment of post-traumatic stress disorder in children and adolescents. Cochrane Database of Systematic Reviews 2012, Issue 12. Art. No: CD006726. [DOI: 10.1002/14651858.CD006726.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Giummarra 2018
- Giummarra MJ, Lennox A, Dali G, Costa B, Gabbe BJ. Early psychological interventions for posttraumatic stress, depression and anxiety after traumatic injury: a systematic review and meta-analysis. Clinical Psychology Review 2018;62:11-36. [DOI] [PubMed] [Google Scholar]
GRADEpro GDT [Computer program]
- GRADEpro GDT. Version (accessed 15 May 2017). Hamilton (ON): GRADE Working Group, McMaster University, 2015.
Gradus 2015
- Gradus JL, Antonsen S, Svensson E, Lash TL, Resick PA, Hansen JG. Trauma, comorbidity, and mortality following diagnoses of severe stress and adjustment disorders: a nationwide cohort study. American Journal of Epidemiology 2015;182:451-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hetrick 2010
- Hetrick SE, Purcell R, Garner B, Parslow R. Combined pharmacotherapy and psychological therapies for posttraumatic stress disorder (PTSD). Cochrane Database of Systematic Reviews 2010, Issue 7. Art. No: CD007316. [DOI: 10.1002/14651858.CD007316.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2019
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al, editor(s). Cochrane Handbook for Systematic Reviews of Interventions version 6.0 (updated August 2019). Cochrane, 2019. training.cochrane.org/handbook/archive/v6. [DOI] [PMC free article] [PubMed]
IAPT 2010
- Baguley C, Farrand P, Hope R, Leibowitz J, Lovell K, Lucock M, et al, IAPT Self-help Conference authors and presenters. Good practice guidance on the use of self-help materials within IAPT services, 2010. www.iapt.nhs.uk/silo/files/good-practice-guidance-on-the-use-of-selfhelp-materials-within-iapt-services.pdf) (accessed 6 June 2017).
ISTSS 2018
- International Society for Traumatic Stress Studies. Posttraumatic stress disorder prevention and treatment guidelines: methodology and recommendations, 2018. www.istss.org/getattachment/Treating-Trauma/New-ISTSS-Prevention-and-Treatment-Guidelines/ISTSS_PreventionTreatmentGuidelines_FNL.pdf.aspx (accessed prior to 23 April 2021).
Karstoft 2015
- Karstoft KI, Armour C, Elklit A, Solomon Z. The role of locus of control and coping style in predicting longitudinal PTSD-trajectories after combat exposure. Journal of Anxiety Disorders 2015;32:89-94. [DOI] [PubMed] [Google Scholar]
Karyotaki 2017
- Karyotaki E, Riper H, Twisk J, Hoogendoorn A, Kleiboer A, Mira A, et al. Efficacy of self-guided Internet-based cognitive behavioral therapy in the treatment of depressive symptoms: a meta-analysis of individual participant data. JAMA Psychiatry 2017;74(4):351-9. [DOI] [PubMed] [Google Scholar]
Kawakami 2014
- Kawakami N, Tsuchiya M, Umeda M, Koenen KC, Kessler RC. Trauma and posttraumatic stress disorder in Japan: results from the World Mental Health Japan Survey. Journal of Psychiatric Research 2014;53:157-65. [DOI] [PMC free article] [PubMed] [Google Scholar]
Kessler 1995
- Kessler RC, Sonnega A, Bromet E, Hughes M, Nelson CB. Posttraumatic stress disorder in the national comorbidity survey. Archives of General Psychiatry 1995;52:1048-60. [DOI] [PubMed] [Google Scholar]
Kessler 2017
- Kessler RC, Aguilar-Gaxiola S, Alonso J, Benjet C, Bromet EJ, Cardoso G, et al. Trauma and PTSD in the WHO World Mental Health Surveys. European Journal of Psychotraumatology 2017;8(5):1353383. [DOI] [PMC free article] [PubMed] [Google Scholar]
Klein 2010
- Klein B, Mitchell J, Abbott J, Shandley K, Austin D, Gilson K, et al. A therapist-assisted cognitive behavior therapy Internet intervention for posttraumatic stress disorder: pre-, post- and 3-month follow-up results from an open trial. Journal of Anxiety Disorders 2010;24:635-44. [DOI] [PubMed] [Google Scholar]
Lewis 2010
- Lewis C, Bisson JI. Health economics of web based mental health interventions to mitigate the effects of trauma. In: Brunet A, Ashbaugh A, Herbert C, editors(s). The Internet in the Aftermath of Trauma. Amsterdam: IOS Press, 2010. [Google Scholar]
Lewis 2012
- Lewis C, Pearce J, Bisson JI. Systematic review of the efficacy, cost effectiveness and acceptability of self-help interventions for individuals diagnosed with an anxiety disorder. British Journal of Psychiatry 2012;200(1):15-21. [DOI] [PubMed] [Google Scholar]
Lewis 2013
- Lewis C, Roberts NR, Vick T, Bisson JI. Development of a guided self-help (GSH) program for the treatment of mild to moderate post-traumatic stress disorder (PTSD). Depression and Anxiety 2013;30(11):1121-8. [DOI] [PubMed] [Google Scholar]
Lewis 2020a
- Lewis C, Roberts NP, Andrew M, Starling E, Bisson JI. Psychological therapies for post-traumatic stress disorder in adults: systematic review and meta-analysis. European Journal of Psychotraumatology 2020;11(1):1729633. [DOI] [PMC free article] [PubMed] [Google Scholar]
Lewis 2020b
- Lewis C, Roberts NP, Gibson S, Bisson JI. Dropout from psychological therapies for post-traumatic stress disorder (PTSD) in adults: systematic review and meta-analysis. European Journal of Psychotraumatology 2020;11:1709709. [DOI] [PMC free article] [PubMed] [Google Scholar]
Mayo‐Wilson 2013
- Mayo-Wilson E, Montgomery P. Media-delivered cognitive behavioural therapy and behavioural therapy (self-help) for anxiety disorders in adults. Cochrane Database of Systematic Reviews 2013, Issue 9. Art. No: CD005330. [DOI: 10.1002/14651858.CD005330.pub4] [DOI] [PMC free article] [PubMed] [Google Scholar]
Newman 2003
- Newman MG, Erickson T, Przeworski A, Dzus E. Self-help and minimal contact therapies for anxiety disorders: is human contact necessary for therapeutic efficacy? Journal of Clinical Psychology 2003;59(3):251-74. [DOI] [PubMed] [Google Scholar]
NICE 2018
- National Institute of Health and Care Excellence. Post-traumatic stress disorder, 2018. www.nice.org.uk/guidance/ng116 (accessed prior to 23 April 2021).
Norris 2003
- Norris FH, Murphy AD, Baker CK, Perilla JL, Rodriguez FG, Rodriguez JD. Epidemiology of trauma and posttraumatic stress disorder in Mexico. Journal of Abnormal Psychology 2003;112(4):646. [DOI] [PubMed] [Google Scholar]
Olaya 2015
- Olaya B, Alonso J, Atwoli L, Kessler RC, Vilagut TG, Haro JM. Association between traumatic events and post-traumatic stress disorder: results from the ESEMeD-Spain study. Epidemiology and Psychiatric Sciences 2015;24:172-83. [DOI] [PMC free article] [PubMed] [Google Scholar]
Olthius 2016
- Olthius JV, Watt MC, Bailey K, Hayden JA, Stewart SH. Therapist-supported Internet cognitive behavioural therapy for anxiety disorders in adults. Cochrane Database of Systematic Reviews 2016, Issue 3. Art. No: CD011565. [DOI: 10.1002/14651858.CD011565.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Olthuis 2015
- Olthuis JV, Watt MC, Bailey K, Hayden JA, Stewart SH. Therapist-supported Internet cognitive behavioural therapy for anxiety disorders in adults. Cochrane Database of Systematic Reviews 2015, Issue 3. Art. No: CD011565. [DOI: 10.1002/14651858.CD011565] [DOI] [PubMed] [Google Scholar]
Ozer 2003
- Ozer EJ, Lipsey TL, Weiss DS. Predictors of posttraumatic stress disorder and symptoms in adults: a meta-analysis. Psychological Bulletin 2003;129(1):52-73. [DOI] [PubMed] [Google Scholar]
Pietrzak 2011
- Pietrzak RH, Goldstein RB, Southwick SM, Grant BF. Prevalence and axis I comorbidity of full and partial posttraumatic stress disorder in the United States: results from wave 2 of the National epidemiologic survey on alcohol and related conditions. Journal of Anxiety Disorders 2011;25:456-65. [DOI] [PMC free article] [PubMed] [Google Scholar]
Pietrzak 2012
- Pietrzak RH, Goldstein RB, Southwick SM, Grant BF. Psychiatric comorbidity of full and partial posttraumatic stress disorder among older adults in the United States: results from wave 2 of the National Epidemiologic Survey on Alcohol and Related Conditions. American Journal of Geriatric Psychiatry 2012;20:380-90. [DOI] [PMC free article] [PubMed] [Google Scholar]
Proudfoot 2003
- Proudfoot J, Goldberg D, Mann A, Everitt B, Marks I, Gray JA. Computerized, interactive, multimedia cognitive-behavioural program for anxiety and depression in general practice. Psychological Medicine 2003;33:217-27. [DOI] [PubMed] [Google Scholar]
Rauch 2009
- Rauch SA, Grunfeld TE, Yadin E, Cahill SP, Hembree E, Foa EB. Changes in reported physical health symptoms and social function with prolonged exposure therapy for chronic posttraumatic stress disorder. Depression and Anxiety 2009;26:732-38. [DOI] [PubMed] [Google Scholar]
Resick 1996
- Resick PA, Schnicke MK. Cognitive Processing Therapy for Rape Victims: a Treatment Manual. 1st edition. London: SAGE Publications, 1996. [Google Scholar]
Review Manager 2014 [Computer program]
- Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Roberts 2016
- Roberts NP, Roberts PA, Jones N, Bisson JI. Psychological therapies for post-traumatic stress disorder and comorbid substance use disorder. Cochrane Database of Systematic Reviews 2016, Issue 4. Art. No: CD010204. [DOI: 10.1002/14651858.CD010204.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Rytwinski 2013
- Rytwinski NK, Scur MD, Feeny NC, Youngstrom EA. The co-occurrence of major depressive disorder among individuals with posttraumatic stress disorder: a meta-analysis. Journal of Traumatic Stress 2013;26:299-309. [DOI] [PubMed] [Google Scholar]
Santiago 2013
- Santiago PN, Ursano RJ, Gray CL, Pynoos RS, Spiegel D, Lewis-Fernandez R, et al. A systematic review of PTSD prevalence and trajectories in DSM-5 defined trauma exposed populations: intentional and non-intentional traumatic events. PloS One 2013;8:e59236. [DOI] [PMC free article] [PubMed] [Google Scholar]
Sareen 2013
- Sareen J, Henriksen C, Bolton S, Afifi T, Stein MB, Asmundson G. Adverse childhood experiences in relation to mood and anxiety disorders in a population-based sample of active military personnel. Psychological Medicine 2013;43:73-84. [DOI] [PubMed] [Google Scholar]
Shapiro 1989
- Shapiro F. Efficacy of the eye movement desensitization procedure in the treatment of traumatic memories. Journal of Traumatic Stress 1989;2:199-223. [Google Scholar]
Shore 2020
Siqveland 2017
- Siqveland J, Hussain A, Lindstrom JC, Ruud T, Hauff E. Prevalence of posttraumatic stress disorder in persons with chronic pain: a meta-analysis. Frontiers in Psychiatry 2017;8:164. [DOI] [PMC free article] [PubMed] [Google Scholar]
Spek 2007
- Spek V, Cuijpers P, Nyklícek I, Riper H, Keyzer J, Pop V. Internet-based cognitive behaviour therapy for symptoms of depression and anxiety: a meta-analysis. Psychological Medicine 2007;37(3):319-28. [DOI] [PubMed] [Google Scholar]
Steel 2009
- Steel Z, Chey T, Silove D, Marnane C, Bryant R, Ommeren M. Association of torture and other potentially traumatic events with mental health outcomes among populations exposed to mass conflict and displacement: a systematic review and meta-analysis. JAMA 2009;302(5):537-49. [DOI] [PubMed] [Google Scholar]
Torous 2019
- Torous J, Andersson G, Bertagnoli A, Christensen H, Cuijpers P, Firth J, et al. Towards a consensus around standards for smartphone apps and digital mental health. World Psychiatry 2019;18:97-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Weathers 2013
- Weathers FW, Litz BT, Keane TM, Palmieri PA, Marx BP, Schnurr PP. The PTSD Checklist for DSM-5 (PCL-5), 2013. www.ptsd.va.gov/professional/assessment/adult-sr/ptsd-checklist.asp (accessed prior to 24 November 2018).
WHO 1979
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems (ICD-9) Mental Disorders. Geneva: World Health Organization, 1979. [Google Scholar]
WHO 1992
- World Health Organization. International Statistical Classification of Diseases and Related Health Problems (ICD-10) Classification of Mental and Behavioural Disorders. Geneva: World Health Organization, 1992. [Google Scholar]
WHO 2018
- World Health Organization. International classification of diseases for mortality and morbidity statistics (11th Revision), 2018. icd.who.int/browse11/l-m/en (accessed prior to 23 April 2021).
Wilson 2012
- Wilson JP, Friedman MJ, Lindy JD, editors. Treating Psychological Trauma and PTSD. New York (NY): Guilford Press, 2012. [Google Scholar]
Wilson 2015
- Wilson LC. A systematic review of probable posttraumatic stress disorder in first responders following man-made mass violence. Psychiatry Research 2015;229:21-6. [DOI] [PubMed] [Google Scholar]
Wind 2020
- Wind T, Rijkeboer M, Andersson G, Riper H. The COVID-19 pandemic: the 'black swan' for mental health care and a turning point for e-health. Internet Interventions 2020;20:100317. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to other published versions of this review
Lewis 2015
- Lewis C, Roberts NP, Bethell A, Bisson JI. Internet-based cognitive and behavioural therapies for post-traumatic stress disorder (PTSD) in adults. Cochrane Database of Systematic Reviews 2015, Issue 5. Art. No: CD011710. [DOI: 10.1002/14651858.CD011710] [DOI] [PMC free article] [PubMed] [Google Scholar]
Lewis 2018
- Lewis C, Roberts N, Bethell A, Robertson L, Bisson J. Internet-based cognitive and behavioural therapies for post-traumatic stress disorder (PTSD) in adults. Cochrane Database of Systematic Reviews 2018, Issue 12. Art. No: CD011710. [DOI: 10.1002/14651858.CD011710.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]


