Abstract
Coronavirus Disease 2019 (COVID-19) is upsetting the world and innovative therapeutic solutions are needed in an attempt to counter this new pandemic. Great hope lies in vaccines, but drugs to cure the infected patient are just as necessary. In the most severe forms of the disease, a cytokine storm with neuroinflammation occurs, putting the patient's life at serious risk, with sometimes long-lasting sequelae. Palmitoylethanolamide (PEA) is known to possess anti-inflammatory and neuroprotective properties, which make it an ideal candidate to be assumed in the earliest stage of the disease. Here, we provide a mini-review on the topic, pointing out phospholipids consumption in COVID-19, the possible development of an antiphospholipid syndrome secondary to SARS-CoV-2 infection, and reporting our preliminary single-case experience concerning to a 45-year-old COVID-19 female patient recently treated with success by micronized / ultramicronized PEA.
Keywords: Coronavirus disease 2019 (COVID-19), Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), Neuroinflammation, Phospholipids, Antiphospholipid syndrome, Palmitoylethanolamide (PEA)
1. Palmitoylethanolamide (PEA)
PEA is an endogenous fatty acid amide able to bind the peroxisome proliferator-activated receptor-α (PPAR-α), also known as nuclear receptor subfamily 1 - group C - member 1 (NR1C1), a major regulator of lipid metabolism [1]. In addition, PEA shows affinity for vanilloid receptor 1 (VR1) and for cannabinoid-like G protein-coupled receptors 55 (GPR55) and 119 (GPR119) [2], enhancing anandamide activity by means of «entourage effect» [3,4]. In 1993, the Nobel prize in Medicine Rita Levi-Montalcini (Turin, April 22, 1909 – Rome, December 30, 2012) provided scientific evidence that PEA downregulates hyperactive mastocytes in a dose-dependent manner, through a feedback mechanism defined “autacoid local inflammation antagonism” (ALIA); for this reason, PEA is nicknamed «ALIAmide» [5]. By virtue of these multiple interactions, PEA is endowed of anti-inflammatory [6,7], anti-nociceptive [8,9], neuroprotective [[10], [11], [12]], and anti-convulsant properties [13].
2. PEA in respiratory viral infections
Twenty years after the Levi-Montalcini's work, a review article on the clinical efficacy and safety of PEA in the treatment of influenza and common cold, based on the data from 6 double-blind, placebo, randomized controlled trials (about 4.000 people), was successfully published [14]. Nowadays, its use has been repurposed to mitigate the neuroinflammatory state and cytokine storm in Coronavirus Disease 2019 (COVID-19) [14,16], and two clinical trials in this regard have been launched in Italy and United States by exploiting micronized and ultramicronized PEA [17,18].
3. PEA micronization technique
PEA micronization is a patented technique that allows to reduce PEA particle diameter up to a micronized size of 2 ± 6 μm (mPEA), optimally absorbable along the intestine, or to an ultramicronized size of 0.8 ± 2 μm (umPEA), able to cross the blood-brain barrier too [19].
4. COVID-19 and severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)
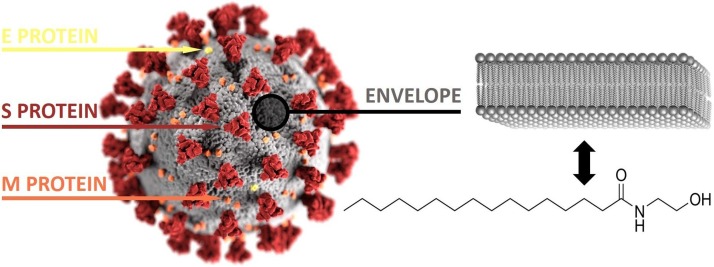
COVID-19 is a complex disease with many clinicopathological issues, including respiratory, vasculo-coagulative and immune ones [[20], [21], [22]]. In those patients requiring intensive care, there is a significant impairment of T-helper 1 (Th1) immune function (cell-mediated immunity); consequently, the immune system is forced to polarize towards a T-helper 2 (Th2) immune response (humoral immunity), whose effectors are mastocytes, basophils, eosinophils, and plasma cells [23,24]. Besides to show antiviral power, mastocytes express angiotensin converting enzyme 2 (ACE2), the principal receptor exploited by SARS-CoV-2 to enter the human cells, becoming themselves a possible target of the viral attack [25,26]. Therefore, an active and passive role of mastocytes in COVID-19 has been documented [25]. Scientific research has been mainly focused on SARS-CoV-2 spike protein, capable of binding ACE2 receptors; however, the outer surface of the virion consists of two other structural proteins, known as envelope and membrane proteins, and of an important phospholipidic envelope, which derives from the cytoplasmatic membrane of the infected cell via budding (Fig. 1 ).
Fig. 1.
Like other coronaviruses, the outer surface of SARS-CoV-2 consists of an envelope (colored in gray) and of 3 structural proteins, namely envelope (E, yellow), membrane (M, orange) and spike (S, red) proteins. The envelope is made up of a phospholipid bilayer, which derives via budding from membrane portions of the host cell infected by the virus. Each phospholipid contains 1 hydrophilic phosphate group and 2 hydrophobic tails of fatty acids, precursors of PEA, as shown by PEA skeletal formula [the 3D illustration of SARS-CoV-2 has been created by Alissa Eckert, MS, and Dan Higgins, MAM, at the Centers for Disease Control and Prevention (CDC) of Atlanta, Georgia, USA, placed in the public domain and thus free of any copyright restrictions].
5. Secondary antiphospholipid syndrome and phospholipids consumption from SARS-CoV-2 infection
It is precisely against this phospholipidic envelope that autoantibodies (e.g. lupus anticoagulant, anti-cardiolipin, anti-β2glycoprotein) can be developed, establishing a serious antiphospholipid syndrome secondary to SARS-CoV-2 infection, able to explain, at the same time, the reported episodes of demyelination and the concomitant occurrence of arterial and venous thromboses in a percentage of COVID-19 patients just under 5% [27]. Since SARS-CoV-2 consumes the membrane phospholipids of infected cells to equip its envelope, the expected logical consequence is that the organism suffers from an acute deficiency of precursor (phosphatidylethanolamine) to synthetize endogenous PEA, essential for controlling neuroinflammation and systemic hyperreactivity. Moreover, through ACE2 receptors, SARS-CoV-2 infects also olfactory neurons until to reach the olfactory bulb, where it can activate microglia with consequent release of pro-inflammatory mediators [28,29]. This can lead to transient anosmia plus ageusia, especially in female patients, and to symptoms and signs of cerebral involvement, such as anxiety, depression, nervous asthenia, post-traumatic stress disorder, headache, insomnia, cognitive dysfunctions (brain fog, memory lapses), delirium or encephalitis, sometimes in the long-term (≥ 4 weeks), the so called «long COVID» (Table 1 ).
Table 1.
According to the British National Institute for Health and Care Excellence (NICE), COVID-19 can be subdivided into 3 clinical definitions: acute COVID-19 for symptoms and signs within the first 4 weeks following SARS-CoV-2 infection; ongoing symptomatic COVID-19 (sub-acute COVID-19) for effects from 4 to 12 weeks from the disease onset; and post−COVID-19 syndrome (chronic COVID-19) for signs and clusters of symptoms that persist 12 or more weeks after SARS-CoV-2 infection, and not explained by an alternative diagnosis. The patient-created term «long COVID» (long-haul COVID) includes both ongoing symptomatic COVID-19 and post−COVID-19 syndrome [available at:https://www.nice.org.uk/guidance/ng188/chapter/Context].
 |
6. Preliminary single-case experience to treat COVID-19 by micronized / ultramicronized PEA
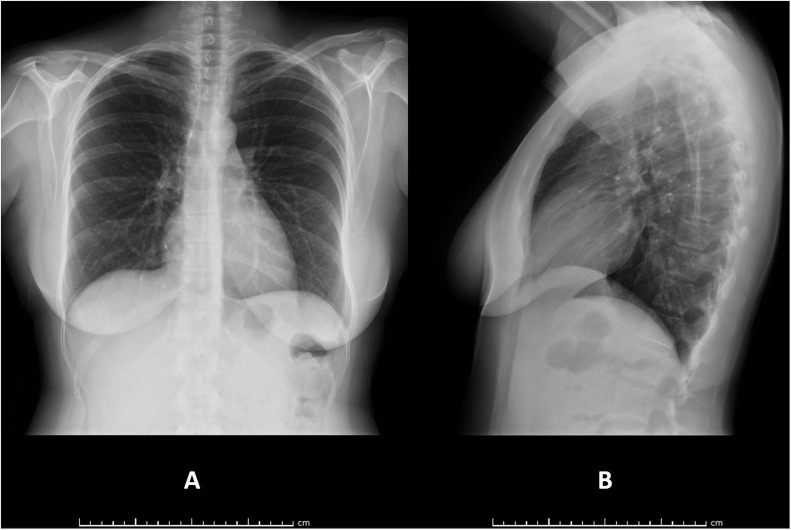
On the basis of all these arguments, after informed consent, we have recently treated a 45-year-old COVID-19 female patient, poliallergic, tested positive to rapid antigenic swab then confirmed positive by real-time reverse transcription polymerase chain reaction (rRT-PCR), with mPEA 300 mg plus umPEA 600 mg in tablets (Normast® MPS), 3 times a day up to rRT-PCR negativization occurred after 17 days, from the very beginning of the appearance of headache, prostration, myalgia and respiratory symptoms (cough, cold). The initial chest X-ray performed at the emergency room showed no signs of interstitial pneumonia in progress (Fig. 2 ), and the blood tests highlighted only mild leukocytosis with high total immunoglobulins E (412 UI/mL) due to the known atopy. Given the absence of relevant comorbidities, the patient was sent back to her home in quarantine, where she immediately started PEA therapy under constant medical supervision with, in addition, antipyretic (paracetamol) as needed. The disease course has been favorable with no hyperpyrexia or hyperperistalsis, normal oxygen saturation, and no loss of smell or taste.
Fig. 2.
Frontal (A) and lateral (B) projections of the patient’s chest X-ray at the early onset of SARS-CoV-2 infection: no signs of interstitial pneumonia in progress are noticeable. The patient was sent back to her home in quarantine, where she immediately started PEA assumption under constant medical supervision.
7. Conclusion and perspective
Our single-case experience supports the administration of mPEA plus umPEA as anti-inflammatory and neuroprotective adjuvant in the earliest stage of COVID-19, and lays the foundations for further research on larger case series. From a speculative point of view, its introduction into multidrug protocol against severe COVID-19 could be also useful to mitigate, at some extent, the cytokine storm in these critical patients with the advantage of not weakening cell-mediated immunity, as conversely do immunosuppressive agents already used in this setting, like corticosteroids or tocilizumab [30]. The virus-induced lymphocytopenia is in fact an unfavorable prognostic parameter in COVID-19, able to exacerbate, if further aggravated by therapy, serious opportunistic infections or fatal herpetic reactivations [30,31]. Precisely this parameter could be considered for PEA introduction in the multidrug COVID-19 regimen, instead of an increase in the dosage of existing immunosuppressive agent or the introduction of a new one; in this way, the antioxidant properties of PEA could be fully exploited, since it reduces the expression of cyclooxygenase 2 and nitric oxide synthase 2, which play a key role in regulating the immune response to infections [15,16]. The most suitable daily dose would be 2700 mg as an oral suspension (mPEA 300 mg plus umPEA 600 mg 3 times a day) in add-on to standard therapy established for each patient. However, the main goal must remain to treat the patient when the first symptoms or fever lines appear, in order to prevent the onset of an hyperinflammatory state, then difficult to manage in clinical practice, especially in older and frail patients.
Author contributions
LR conceived, designed and supervised the study, interpreted the data, prepared the figures and the table with the related legends, and wrote the manuscript; BL performed the literature search; FP revised critically the final version of the manuscript; LC provided ideas for the research study.
Declaration of Competing Interest
The authors declare no competing interest.
Acknowledgment
The authors thank Giuliano Claps, ISF Specialist, EPITECH Group SpA, for his information support.
References
- 1.Lo Verme J., Fu J., Astarita G., La Rana G., Russo R., Calignano A., et al. The nuclear receptor peroxisome proliferator-activated receptor-alpha mediates the anti-inflammatory actions of palmitoylethanolamide. Mol. Pharmacol. 2005;67(1):15–19. doi: 10.1124/mol.104.006353. [DOI] [PubMed] [Google Scholar]
- 2.Godlewski G., Offertáler L., Wagner J.A., Kunos G. Receptors for acylethanolamides - GPR55 and GPR119. Prostaglandins Other Lipid Mediat. 2009;89(3-4):105–111. doi: 10.1016/j.prostaglandins.2009.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ho W.S., Barrett D.A., Randall M.D. ’Entourage’ effects of N-palmitoylethanolamide and N-oleoylethanolamide on vasorelaxation to anandamide occur through TRPV1 receptors. Br. J. Pharmacol. 2008;155(6):837–846. doi: 10.1038/bjp.2008.324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Jonsson K.O., Vandevoorde S., Lambert D.M., Tiger G., Fowler C.J. Effects of homologues and analogues of palmitoylethanolamide upon the inactivation of the endocannabinoid anandamide. Br. J. Pharmacol. 2001;133(8):1263–1275. doi: 10.1038/sj.bjp.0704199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aloe L., Leon A., Levi-Montalcini R. A proposed autacoid mechanism controlling mastocyte behaviour. Agents Actions. 1993;39:145–147. doi: 10.1007/BF01972748. [DOI] [PubMed] [Google Scholar]
- 6.Endocannabinoid Research Group, De Filippis D., D’Amico A., Cipriano M., Petrosino S., Orlando P., et al. Levels of endocannabinoids and palmitoylethanolamide and their pharmacological manipulation in chronic granulomatous inflammation in rats. Pharmacol. Res. 2010;61(4):321–328. doi: 10.1016/j.phrs.2009.11.005. [DOI] [PubMed] [Google Scholar]
- 7.Lambert D.M., Vandevoorde S., Jonsson K.O., Fowler C.J. The palmitoylethanolamide family: a new class of anti-inflammatory agents? Curr. Med. Chem. 2002;9(6):663–674. doi: 10.2174/0929867023370707. [DOI] [PubMed] [Google Scholar]
- 8.Calignano A., La Rana G., Piomelli D. Antinociceptive activity of the endogenous fatty acid amide, palmitylethanolamide. Eur. J. Pharmacol. 2001;419(2-3):191–198. doi: 10.1016/s0014-2999(01)00988-8. [DOI] [PubMed] [Google Scholar]
- 9.Walker J.M., Krey J.F., Chu C.J., Huang S.M. Endocannabinoids and related fatty acid derivatives in pain modulation. Chem. Phys. Lipids. 2002;121(1-2):159–172. doi: 10.1016/s0009-3084(02)00152-4. [DOI] [PubMed] [Google Scholar]
- 10.Koch M., Kreutz S., Böttger C., Benz A., Maronde E., Ghadban C., et al. Palmitoylethanolamide protects dentate gyrus granule cells via peroxisome proliferator-activated receptor-α. Neurotox. Res. 2011;19(2):330–340. doi: 10.1007/s12640-010-9166-2. [DOI] [PubMed] [Google Scholar]
- 11.D’Agostino G., Russo R., Avagliano C., Cristiano C., Meli R., Calignano A. Palmitoylethanolamide protects against the amyloid-β25-35-induced learning and memory impairment in mice, an experimental model of Alzheimer disease. Neuropsychopharmacology. 2012;37(7):1784–1792. doi: 10.1038/npp.2012.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Roncati L., Pusiol T., Piscioli F., Lavezzi A.M. Neurodevelopmental disorders and pesticide exposure: the northeastern Italian experience. Arch. Toxicol. 2017;91(2):603–604. doi: 10.1007/s00204-016-1920-7. [DOI] [PubMed] [Google Scholar]
- 13.Lambert D.M., Vandevoorde S., Diependaele G., Govaerts S.J., Robert A.R. Anticonvulsant activity of N-palmitoylethanolamide, a putative endocannabinoid, in mice. Epilepsia. 2001;42(3):321–327. doi: 10.1046/j.1528-1157.2001.41499.x. [DOI] [PubMed] [Google Scholar]
- 14.Keppel Hesselink J.M., de Boer T., Witkamp R.F. Palmitoylethanolamide: a natural body-own anti-inflammatory agent, effective and safe against influenza and common cold. Int. J. Inflam. 2013;2013:151028. doi: 10.1155/2013/151028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gigante A., Aquili A., Farinelli L., Caraffa A., Ronconi G., Enrica Gallenga C., et al. Sodium chromo-glycate and palmitoylethanolamide: a possible strategy to treat mast cell-induced lung inflammation in COVID-19. Med. Hypotheses. 2020;143 doi: 10.1016/j.mehy.2020.109856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pesce M., Seguella L., Cassarano S., Aurino L., Sanseverino W., Lu J., et al. Phytotherapics in COVID-19: why palmitoylethanolamide? Phytother. Res. 2020;9(December) doi: 10.1002/ptr.6978. [DOI] [PubMed] [Google Scholar]
- 17.U.S. National Library of Medicine, Micronized and ultramicronized palmitoylethanolamide in COVID-19 patients, ClinicalTrials.gov Identifier: NCT04568876.
- 18.U.S. National Library of Medicine, Ultramicronized palmitoylethanolamide (PEA) treatment in hospitalized participants with COVID-19, ClinicalTrials.gov Identifier: NCT04619706.
- 19.Impellizzeri D., Bruschetta G., Cordaro M., Crupi R., Siracusa R., Esposito E., et al. Micronized/ultramicronized palmitoylethanolamide displays superior oral efficacy compared to nonmicronized palmitoylethanolamide in a rat model of inflammatory pain. J. Neuroinflammation. 2014;11:136. doi: 10.1186/s12974-014-0136-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Roncati L., Manenti A., Manco G., Farinetti A. Abdominal aortic thrombosis complicating COVID-19 pneumonia. Ann. Vasc. Surg. 2020;67:8–9. doi: 10.1016/j.avsg.2020.05.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Roncati L., Ligabue G., Nasillo V., Lusenti B., Gennari W., Fabbiani L., et al. A proof of evidence supporting abnormal immunothrombosis in severe COVID-19: naked megakaryocyte nuclei increase in the bone marrow and lungs of critically ill patients. Platelets. 2020;31(8):1085–1089. doi: 10.1080/09537104.2020.1810224. [DOI] [PubMed] [Google Scholar]
- 22.Roncati L., Ligabue G., Fabbiani L., Malagoli C., Gallo G., Lusenti B., et al. Type 3 hypersensitivity in COVID-19 vasculitis. Clin. Immunol. 2020;217 doi: 10.1016/j.clim.2020.108487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Roncati L., Nasillo V., Lusenti B., Riva G. Signals of Th2 immune response from COVID-19 patients requiring intensive care. Ann. Hematol. 2020;99(6):1419–1420. doi: 10.1007/s00277-020-04066-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Roncati L., Vadalà M., Corazzari V., Palmieri B. COVID-19 vaccine and boosted immunity: nothing ad interim to do? Vaccine. 2020;38(48):7581–7584. doi: 10.1016/j.vaccine.2020.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Afrin L.B., Weinstock L.B., Molderings G.J. Covid-19 hyperinflammation and post-Covid-19 illness may be rooted in mast cell activation syndrome. Int. J. Infect. Dis. 2020;100:327–332. doi: 10.1016/j.ijid.2020.09.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roncati L., Gallo G., Manenti A., Palmieri B. Renin-angiotensin system: the unexpected flaw inside the human immune system revealed by SARS-CoV-2. Med. Hypotheses. 2020;140 doi: 10.1016/j.mehy.2020.109686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Roncati L., Manenti A., Manco G., Farinetti A., Mattioli A. The Covid-19 arterial thromboembolic complications: from inflammation to immunothrombosis through anti-phospholipid autoantibodies. Ann. Vasc. Surg. 2020;30(December) doi: 10.1016/j.avsg.2020.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Meng X., Deng Y., Dai Z., Meng Z. COVID-19 and anosmia: a review based on up-to-date knowledge. Am. J. Otolaryngol. 2020;41(5):102581. doi: 10.1016/j.amjoto.2020.102581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zahra S.A., Iddawela S., Pillai K., Choudhury R.Y., Harky A. Can symptoms of anosmia and dysgeusia be diagnostic for COVID-19? Brain Behav. 2020;10(11):e01839. doi: 10.1002/brb3.1839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Roncati L., Manenti A., Fabbiani L., Malagoli C., Nasillo V., Lusenti B., et al. HSV1 viremia with fulminant hepatitis as opportunistic sequela in severe COVID-19. Ann. Hematol. 2021;18(January) doi: 10.1007/s00277-021-04417-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Roncati L., Lusenti B., Nasillo V., Manenti A. Fatal SARS-CoV-2 coinfection in course of EBV-associated lymphoproliferative disease. Ann. Hematol. 2020;99(8):1945–1946. doi: 10.1007/s00277-020-04098-z. [DOI] [PMC free article] [PubMed] [Google Scholar]