Abstract
Long COVID or post-COVID-19 syndrome first gained widespread recognition among social support groups and later in scientific and medical communities. This illness is poorly understood as it affects COVID-19 survivors at all levels of disease severity, even younger adults, children, and those not hospitalized. While the precise definition of long COVID may be lacking, the most common symptoms reported in many studies are fatigue and dyspnoea that last for months after acute COVID-19. Other persistent symptoms may include cognitive and mental impairments, chest and joint pains, palpitations, myalgia, smell and taste dysfunctions, cough, headache, and gastrointestinal and cardiac issues. Presently, there is limited literature discussing the possible pathophysiology, risk factors, and treatments in long COVID, which the current review aims to address. In brief, long COVID may be driven by long-term tissue damage (e.g. lung, brain, and heart) and pathological inflammation (e.g. from viral persistence, immune dysregulation, and autoimmunity). The associated risk factors may include female sex, more than five early symptoms, early dyspnoea, prior psychiatric disorders, and specific biomarkers (e.g. D-dimer, CRP, and lymphocyte count), although more research is required to substantiate such risk factors. While preliminary evidence suggests that personalized rehabilitation training may help certain long COVID cases, therapeutic drugs repurposed from other similar conditions, such as myalgic encephalomyelitis or chronic fatigue syndrome, postural orthostatic tachycardia syndrome, and mast cell activation syndrome, also hold potential. In sum, this review hopes to provide the current understanding of what is known about long COVID.
Keywords: Long-haul COVID-19, long COVID, post-COVID-19 syndrome, risk factors, pathophysiology, drug repurposing
Introduction
Early into the coronavirus disease 2019 (COVID-19) pandemic, announced in March 2020 by the World Health Organization (WHO), hardly anyone would have thought that the disease might be chronic. The causative agent of COVID-19 is the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). As the ‘A’ in the name implies, the respiratory disease is acute [1]. However, longer-lasting COVID-19 cases started gaining traction among social support groups. At first, doctors dismissed their concerns as symptoms related to mental health, such as anxiety or stress, in a phenomenon called ‘medical gaslighting’ [2,3]. However, that soon changed. The term long COVID (or post-COVID syndrome or long-haul COVID-19) started gaining recognition in the scientific and medical communities [4]. Different descriptions of long COVID have already been proposed, and the most common description is symptoms lasting for more than three months after the first symptom onset (Table 1).
Table 1.
Proposed descriptions of long COVID.
| Reference | Terms | Description |
|---|---|---|
| [4] | Long COVID | Long-term COVID-19 illness that is cyclical, progressive, and multiphasic. |
| [5,6,7] | Long-hauler COVID-19 Long-COVID Chronic COVID syndrome |
Multi-organ symptoms that persist for months after acute COVID-19. |
| [8] | Long-haul COVID Long-tail COVID |
Symptoms lasting for > 100 days. |
| [9,10] | Long COVID | Symptoms lasting for > 2 months. |
| [11,12,13] | Late sequelae of SARS-CoV-2 infection Long-haulers Long-COVID |
Symptoms lasting for > 4 weeks after the initial infection or diagnosis. |
| [14] | Post-acute COVID-19 syndrome | Symptoms lasting for > 4 weeks after the first symptom onset. |
| [15] | Acute post-COVID symptoms | Symptoms lasting for 5-12 weeks. |
| Long post-COVID symptoms | Symptoms lasting for 12-24 weeks. | |
| Persistent post-COVID symptoms | Symptoms lasting for > 24 weeks. | |
| [16, 17,7] | Post-acute COVID-19 On-going symptomatic COVID-19 |
Symptoms lasting for 1-3 months from the first symptom onset. |
| Chronic COVID-19 Long COVID Post-COVID-19 syndrome |
Symptoms lasting for > 3 months from the first symptom onset. |
While the actual definition is lacking, a review identified that the most frequent symptoms of long COVID are fatigue and dyspnoea (i.e. shortness of breath) [17,18]. Other less typical symptoms include cognitive and mental disorders, headache, myalgia, chest and joint pains, smell and taste dysfunctions, cough, hair loss, insomnia, wheezing, rhinorrhea, sputum, and cardiac and gastrointestinal issues. These symptoms may persist for up to six months and counting after hospital discharge or symptom onset (Table 2). Less common symptoms of pernio, chills, flushing, ear pain, and visual impairments associated with long COVID have also been documented [36,45,47]. This illustrates the multifaceted nature of long COVID that involves multiple organ systems.
Table 2.
Demographics, clinical outcomes, and symptom prevalence of long COVID survivors.
| Study | Participant and clinical characteristics | Follow-up duration | Symptom (% prevalence) |
|---|---|---|---|
| [19] | N = 110; median age = 60; 44% females; 100% inpatients; 24.5% had mild disease; 59% had moderate disease; 16.4% had severe disease; Bristol, England. | Median of 83 days after hospital discharge. |
|
| [20] | N = 238; median age = 61; 40.3% females; 100% inpatients; 72.3% needed supplemental O2; 11.8% admitted to ICU; 8.8% needed MV; Novara, Italy. | 4 months after hospital discharge. |
|
| [21] | N = 143; 56.5 ± 14.6 years; 37.1% females; 100% inpatients; 53.8% needed supplemental O2; 12.6% admitted to ICU; 14.7% needed non-invasive ventilation; 4.9% needed MV; Rome, Italy. | Mean of 60 days after hospital discharge. |
|
| [22], preprint. | N = 21,359 (only 233 were COVID-19 survivors; 96.6% outpatients); median age = 56; 63.6% females; 83.7% Europeans. | 90 days after symptom onset. |
|
| [23], preprint | *N = 3,762; age groups of 18-29 (8%), 30-39 (26.1%), 40-49 (33.7%), 50-59 (27.1%), 60-69 (11%), 70-79 (2.5%), 80+ (0.4%) years; 78.9% females; 56.7% did not seek hospital care; 34.9% outpatients; 8.43% were hospitalized; 56 countries (41.6% from U.S.). | 6-7 months after symptom onset. |
|
| [24] | *N = 201; 44 ± 11 years; 71% females; 81.6% outpatients; 18.4% were hospitalized; London, U.K. | Median of 140 days after symptom onset. |
|
| [25] | N = 120; 63.2 ± 15.7 years; 37.5% females; 100% inpatients; 20% admitted to ICU; Clichy, France. | Mean of 110 days after hospital admission. |
|
| [26] | N = 114; 54 ± 12 years; 30% females; 100% inpatients; 21% needed MV; Wuhan, China. | Mean of 175 days after symptom onset. |
|
| [27] | N = 1733, median age = 57; 48% females; 100% inpatients; 67.6% needed supplemental O2; 4% admitted to ICU; Hubei, China. | Median of 186 days after symptom onset. |
|
| [28] | N = 103; median age = 59; 48% females; 100% inpatients; 66% needed supplemental O2; 15% admitted to ICU; 9% needed MV; Lørenskog, Norway. | 3 months after hospital admission. |
|
| [29] | N = 76; 41.3 ± 13.8 years; 72.4% females; 100% inpatients; 9% admitted to ICU. Wuhan, China. | 3 months after hospital discharge. |
|
| [30]. | N = 60; 44.10 ± 16 years; 43.3% females; 100% inpatients; 78.3% had mild disease; 20% had severe disease; 1.7% had critical disease; Anhui Province, China. | 3 months after hospital discharge. |
|
| [31] | N = 63; 48.1 ± 18.5 years; 33.3% females; 100% inpatients; 27% needed supplemental O2; 7.9% needed MV; Tokyo, Japan. | 120 days after symptom onset. |
|
| [32] | N = 180; 39.9 ± 19.4 years; 54% females; 95.6% outpatients; 4.4% were hospitalized; Faroe Islands, Denmark. | 125 days after symptom onset. |
|
| [33] | N = 120; 54.6 ± 16.9 years; 33.3% females; 100% inpatients; 7.5% admitted to ICU; Tehran, Iran. | 6 months after hospital discharge. |
|
| [34] | N = 60; median age = 67; 32% females; 100% inpatients; 46% needed supplemental O2; 20% needed MV; Vancouver, Canada. | 12 weeks after symptom onset. |
|
| [35] | N = 145; 57 ± 14 years; 43% females; 25% outpatients; 75% were hospitalized; 66% needed supplemental O2; 27% needed MV; Innsbruck, Austria. | Mean of 103 days after diagnosis. |
|
| [36] | N = 434; 49.8 ± 15.2 years; 56% females; 100% outpatients; Lørenskog, Norway. | Median of 117 days after symptom onset. |
|
| [37] | N = 4182; median age = 42 years; 71.5% females; Sweden, U.K, and U.S. | 12 weeks after symptom onset. |
|
| [38] | N = 242; 65.9 ± 14.1 years; 40.5% females; 100% inpatients; 18.2% admitted to ICU; 12.8% needed MV; Santiago, Spain. | 6 months after hospital discharge. |
|
| [39] | N = 128; 49.5 ± 15 years; 54% females; 44.5% outpatients; 55.5% were hospitalized; 36.7% needed supplemental O2; 14.1% admitted to ICU; Dublin, Ireland. | Median of 10 weeks after hospital discharge or last acute symptom. |
|
| [40] | N = 22; 54.6 ± 10.9 years; 27.3% females; 100% ICU patients; 64% needed MV; Brussels, Belgium. | 3 months after hospital discharge. |
|
| [41] | N = 124; 59 ± 14 years; 40% females; 100% inpatients; 21.8% had mild disease; 41% had moderate disease; 21% had severe disease; 16.1% had critical disease; Nijmegen, Netherlands. | 3 months after hospital discharge. |
|
| [42] | N = 48; median age = 63; 31.2% females; 100% ICU patients on MV; Maastricht, Netherlands. | 3 months after hospital discharge. |
|
| [43] | N = 78; 62 ± 16 years; 36% females; 100% inpatients; Vancouver, Canada. | 3 months after symptom onset. |
|
| [44] | N = 18; 42.2 ± 14.3 years; 57.9% females; 33.3% outpatients; 22.2% needed supplemental O2; Hamburg, Germany. | Median of 85 days after hospital discharge. |
|
| [45] | N = 538; median age = 52; 54.5% females; 100% inpatients; 33.5% had severe disease; 5% had critical disease; Wuhan, China. | Median of 97 days after hospital discharge. |
|
| [46] | N = 55; 47.5 ± 15.5 years; 41.8% females; 100% inpatients; 7.3% had mild disease, 85.5% had moderate disease; 7.3% had severe disease; 25.5% needed supplemental O2; Henan Province, China. | 3 months after hospital discharge. |
|
Years refer to age presented as mean ± standard deviation, unless otherwise stated as median. * refers to sample size that specifically involved long COVID participants. ICU: intensive care unit; IL; MV: mechanical ventilation; O2: oxygen; PTSD: post-traumatic stress disorder.
Evidently, studies have also reported different persistent symptoms in contrasting durations and frequencies among long COVID survivors (Table 2). This may be due to distinct sample characteristics and data collection methods each study employed or the fact that long COVID is a highly heterogeneous condition [11,48]. Therefore, the precise symptomatic manifestations of long COVID remains elusive and may involve multiple subtypes or phenotypes [49].
One puzzling feature of long COVID is that it affects survivors of COVID-19 at all disease severity. Studies have discovered that long COVID affects even mild-to-moderate cases and younger adults who did not require respiratory support or hospital or intensive care. Patients who were no longer positive for SARS-CoV-2 and discharged from the hospital, as well as outpatients, can also develop long COVID [24,30,31,41,50]. More concerningly, long COVID also targets children, including those who had asymptomatic COVID-19, resulting in symptoms such as dyspnoea, fatigue, myalgia, cognitive impairments, headache, palpitations, and chest pain that last for at least 6 months [51–53].
One known aspect of long COVID is that similar post-viral syndrome was observed with prior human coronavirus diseases. For example, symptoms of fatigue, myalgia, and psychiatric impairments have inflicted survivors of Middle East respiratory syndrome (MERS) and severe acute respiratory syndrome (SARS) for up to four years [54–58]. Even at 7-year and 15-year follow-ups, pulmonary and bone radiological complications were still evident among a proportion of SARS survivors who were mostly younger than 40 years [59,60]. This is rather unsettling as it implies that long COVID may extend beyond just a few months to years.
Presently, there are limited research papers that have voiced discussions about the possible pathophysiology, risk factors, and treatments for long COVID. The current review, hence, seeks to fulfil these gaps.
Methods
The literature search was conducted on PubMed database using the following algorithm: (SARS-CoV-2[tiab] OR COVID*[tiab]) AND (long-haul[tiab] OR long COVID[tiab] OR post-COVID*[tiab] OR post-viral[tiab] OR recover*[ti] OR survivor*[ti] OR discharge*[ti] OR sequela*[ti] OR prolong*[ti] OR persistent[ti] OR long-term[ti]) AND English[la]. The sole author screened the titles and abstracts to identify relevant papers. Long COVID was identified with the presence of at least one persistent symptom that includes either fatigue or dyspnoea for at least three months after symptom onset, hospital admission, or diagnosis [17] (Table 1). Since acute COVID-19 may last for a few weeks before hospital discharge, symptoms lasting for at least two months after discharge is also considered as long COVID. Additional references from reviewers’ recommendations and reference lists of included articles were also incorporated. The last literature search was performed on 5th February 2021, with another brief search update on 15th April 2021.
Studies on long COVID with information on symptomatic prevalence were summarized in Table 2. Studies detailing probable long COVID with a follow-up duration of fewer than three months after symptom onset or two months after hospital discharge were summarized in Supplementary Table 1. The subsequent putative pathophysiology, risk factors, and treatments sections were based on narrative review expanding on studies in Table 2.
Putative pathophysiology
Long-term tissue damage
In a three-month follow-up study of COVID-19 survivors, pulmonary radiological abnormalities and functional impairments were detected in 71% and 25% of participants, respectively, despite that only less than 10% had severe pneumonia [46]. Another study has also observed reduced lung diffusion capacity that correlated with radiological abnormalities in 42% of COVID-19 survivors at three-month post-hospital discharge, regardless of initial disease severity [41]. Even at six months after symptom onset, lung radiological abnormalities associated with persistent symptoms were still present in about half of COVID-19 survivors [27]. Many other reports have also found radiological evidence of lung fibrosis lasting up to six months among COVID-19 survivors after hospital discharge, which also correlated with initial disease severity [20,26,40,61–63].
Using a more advanced xenon gas radiological technique to study lung function, a study discovered defective pulmonary gas-exchange function among discharged patients who had moderate COVID-19 compared to healthy controls [64]. Moreover, in this study, such pulmonary issues were not detectable with standard chest computed tomography (CT), suggesting that routine radiological examinations might have overlooked such pulmonary complications. Notably, a study found reduced maximal aerobic capacity at about 45-day follow-up among young recruits with symptomatic COVID-19 compared to non-COVID-19 recruits [65]. These studies collectively indicate that pulmonary scarring may be a common sequela of COVID-19, which may be responsible for persistent dyspnoea and cough in long COVID [66,67].
However, a separate study has also found that symptoms of long COVID persist even in those with improvements in pulmonary radiological and functional examinations [19]. Thus, long COVID may involve other pathophysiology besides pulmonary lesions, such as lasting neurological complications. For instance, at three-month post-discharge, brain structural and metabolic abnormalities were reported among COVID-19 survivors, which correlated with persistent neurological symptoms such as memory loss, anosmia, and fatigue [30]. This finding is concerning as most participants had mild COVID-19 at baseline, suggesting that even mild COVID-19 could have persistent effects on the brain. Another study documenting 43 cases of COVID-19-induced serious brain diseases (e.g. encephalopathies, delirium, haemorrhage, and stroke) also found that initial COVID-19 severity plays little role in predicting these brain diseases [68].
In more severe cases of COVID-19 resulting in delirium in about 20–30% of hospitalized patients, long-term neurological symptoms are more probable [69–71]. Delirium is also a strong predictor of long-term cognitive impairments, especially among older adults [72,73]. A meta-analysis examining neuropsychiatric outcomes of SARS, MERS, and COVID-19 survivors has found delirium as a common complication in the acute phase of disease, which can lead to various neuropsychiatric sequelae, such as depression, anxiety, post-traumatic stress disorder, memory loss, and fatigue [58]. Indeed, COVID-19-related fatigue has been suggested to result from autonomic nervous system dysfunction [3,74]. In a registry study of 236,379 COVID-19 survivors, about a third received a neuropsychiatric diagnosis (e.g. stroke, dementia, insomnia, and anxiety and mood disorders) within 6 months after the first symptom onset, which was 44% more common than influenza survivors. Moreover, in this study, ICU survivors were 56% more likely to develop a neuropsychiatric disorder compared to non-ICU survivors [75].
As SARS-CoV-2 is a respiratory virus, lung injury can be expected. However, it was only much later that evidence started confirming the neurotropism and replication capacity of SARS-CoV-2 in neuronal cultures, brain organoids, mice, and human brain autopsies [76–79,80]. Notably, damage to the brainstem’s cardiorespiratory centre has been proposed to worsen symptoms of COVID-19 [81,82]. Since neurons rarely regenerate, the resulting brainstem dysfunction may be long-lasting, leading to neurological and cardiorespiratory sequelae that might underlie long COVID [83]. The brainstem expresses higher levels of angiotensin-converting enzyme 2 (ACE2), the receptor for SARS-CoV-2, than other brain regions [84]. Autopsy reports have also found evidence of SARS-CoV-2 genes and proteins, as well as pathological immune and vascular activations, in the brainstem of deceased COVID-19 victims [85–87]. Therefore, on-going neuroinflammatory processes may drive the neurological symptoms and damage in long COVID.
Evidence of cardiac injury in long COVID also exists. A radiological study of 100 discharged COVID-19 patients has found cardiac abnormalities and myocardial inflammation in 78% and 60% of participants, respectively, which were not associated with initial COVID-19 severity [88]. In another study of 26 college athletes with asymptomatic SARS-CoV-2 infection, 46% of them also presented with myocardial inflammation [89]. Even at three-month post-hospital discharge, radiological abnormalities of ventricular remodelling were still evident in 29% of 79 COVID-19 survivors [90]. However, the long-term clinical significance of these radiological findings remains unclear as symptomatic assessments were not performed, warranting further research and surveillance [91,92]. Nevertheless, cardiac symptoms such as chest pain, heart palpitations, and tachycardia commonly persist among COVID-19 survivors for up to six months, suggesting substantial cardiac sequelae [21,24,27,29].
Lastly, the long-term damage of other organs may also be involved in long COVID. One preprint report has found that young adults, mostly free of risk factors for severe COVID-19, often develop long COVID with multi-organ impairment at four-month follow-up. Specifically, at least one radiological abnormality of the lungs, heart, liver, pancreas, kidneys, or spleen was present in 66% of survivors [24]. Similarly, another study involving moderate-to-severe COVID-19 patients has shown radiological evidence of lung, heart, brain, liver and kidney impairments persisting for at least 2–3 month after hospital discharge [93]. Furthermore, a study of over 40,000 discharged COVID-19 patients found increased risks of new events of respiratory, diabetes, and cardiovascular diseases occurring within the subsequent 140 days compared to controls [94]. Therefore, future research of long COVID should consider possible extrapulmonary or multi-organ involvement that may be less obvious.
Pathological inflammation
There have been instances of COVID-19 patients who remained positive for SARS-CoV-2 by reverse transcription real-time polymerase chain reaction (RT-PCR) test for up to three months [95–97,98]. Other studies have documented cases of prolonged SARS-CoV-2 shedding in the respiratory tract via quantitative RT-PCR for up to four months [99,100]. Extended SARS-CoV-2 shedding has also been detected in the faeces, regardless of gastrointestinal symptom manifestation, for up to two months [101,102]. A more recent study has discovered SARS-CoV-2 nucleic acids and proteins in the small intestines of 50% of asymptomatic COVID-19 cases at 4-month post-disease onset [103]. Therefore, these studies showed that SARS-CoV-2 persistence in the body is possible, which may induce some level of immune activation contributing to long COVID.
A review has proposed that T-cells dysfunction may promote long COVID pathophysiology similarly in autoimmune diseases [104]. For instance, SARS-CoV-2 could make antigen-presenting cells present antigens to auto-reactive T-cells in a process called bystander activation. This is consistent with autopsy examinations of deceased COVID-19 patients showing that infiltrates in the lungs and other organs were enriched with CD8+ T cells, one of the crucial mediators of autoimmune reactions [105]. Surprisingly, thyroid dysfunction has been detected in 15–20% of patients with COVID-19 [106,107]. As the thyroid is closely linked to T-cell-mediated autoimmunity, thyroid dysfunction may play a role in the autoimmunity pathophysiology of long COVID [106,108].
B-cells may also be involved in long COVID autoimmunity. In a study analysing serum samples from hospitalized COVID-19 patients, antiphospholipid autoantibodies were detected in 52% of samples, which were further associated with neutrophil hyperactivity and more severe clinical outcomes [109]. Other studies have also identified autoantibodies against interferons, neutrophils, connective tissues, cyclic citrullinated peptides, and cell nucleus in 10–50% of patients with COVID-19 [110–112,113]. While it is unconfirmed if such autoantibodies are long-lasting in COVID-19, research review has strongly linked these autoantibodies to chronic autoimmune diseases, such as antiphospholipid and Sjogren syndromes, lupus erythematosus, and rheumatoid arthritis [114]. Notably, reviews on lupus and rheumatoid arthritis also bear symptomatic resemblances to long COVID: fatigue, joint pain, concentration difficulties, and headache [115,116].
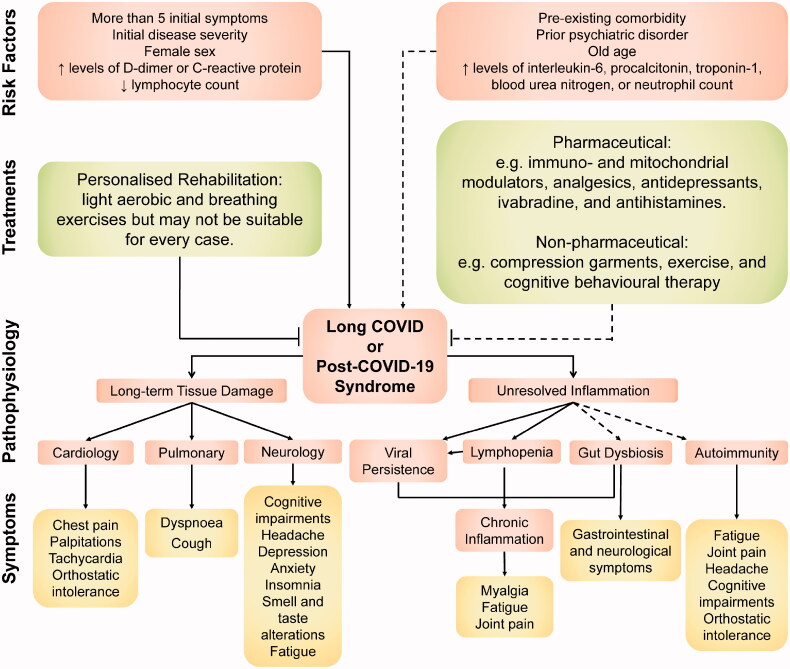
Besides, evidence exists that severe COVID-19 causes lymphopenia (i.e. B-cell and T-cell lymphocytes deficiency) that causes hyperinflammation [117,118]. This is because lymphocytes, particularly T-cells, participate in inflammation resolution following infection [119,120]. Following this, meta-analyses have determined lymphopenia and high pro-inflammatory neutrophil count as independent risk factors of COVID-19 severity and mortality [121–123]. Thus, as B-cell and T-cell lymphocytes are renewed, elevated inflammation from unresolved hyperinflammation may ensue and contribute to long COVID [118,120]. Moreover, decreased T-cell and B-cell numbers have been shown to correlate with persistent SARS-CoV-2 shedding, which may further perpetuate chronic immune activation in long COVID [124,125] (Figure 1).
Figure 1.
An overview of the symptoms, putative pathophysiology, associated risk factors, and potential treatments involved in long COVID. Note: Dashed lines represent areas where evidence is relatively lacking compared to non-dashed lines. (Color online only).
Furthermore, numerous cases of multisystem inflammatory syndrome (MIS) occurring 2–6 weeks after SARS-CoV-2 infection in children and adults have been documented. These patients do not necessarily have a positive SARS-CoV-2 status or severe respiratory disease. Yet they showed elevated levels of systemic pro-inflammatory markers (e.g. CRP, interleukin-6; IL-6, ferritin, and D-dimer) and severe shock, cardiac, gastrointestinal, or neurological symptoms [126–130]. The delayed manifestation of MIS after SARS-CoV-2 infection suggests the involvement of dysregulated adaptive immune system; autoantibodies, in particular, have been strongly suspected in research reviews [9,131]. Therefore, it may be possible that residual inflammation and symptoms from post-SARS-CoV-2 MIS could lead to long COVID in children and adults.
Indeed, increased levels of pro-inflammatory markers (e.g. CRP, IL-6, and D-dimer) and lymphopenia have been associated with long COVID (section ‘Patient and Clinical Characteristics’). Another radiological study of COVID-19 survivors with symptoms persisting for at least 30 days after discharge has revealed increased [18F] FDG uptake, signifying persistent inflammation, in the bone marrow and blood vessels [132]. A recent case–control study found elevated levels of vascular-related pro-inflammatory biomarkers, which correlated with pulmonary damage, among COVID-19 patients discharged three months prior [133]. However, other long COVID studies that found no association with pro-inflammatory biomarkers also exist. These reports imply that unresolved inflammation may only partly explain the pathophysiology of long COVID, particularly and perhaps the inflammation-related symptoms such as myalgia, joint pain, and fatigue [134,135] (Figure 1). Notably, chronic fatigue is a complex syndrome that may have other causes besides inflammation, such as channelopathies, inadequate cerebral perfusion, and autonomic nervous system dysfunction, which may also be involved in long COVID [3,136,137].
Another possible source of unresolved inflammation in long COVID could lie in the gut. SARS-CoV-2 has been known to replicate efficiently in gastric and intestinal cells, owing to the high expression of ACE2 receptors therein, leading to increased faecal shedding of SARS-CoV-2 in patients [138–140]. While the prevalence of gastrointestinal symptom may vary between different study designs, meta-analyses have estimated that gastrointestinal manifestations (e.g. appetite loss, nausea, vomiting, diarrhoea, and abdominal discomfort) affect 10–20% of patients with COVID-19 [141,142]. Importantly, gastrointestinal symptoms have also been reported in up to a third of individuals with long COVID [29,32,46]. Thus, SARS-CoV-2 persistence in the gastrointestinal tract may underlie the gastrointestinal manifestations of long COVID.
Gut microbiome disruption (i.e. gut dysbiosis) has been observed among patients with COVID-19, which persisted for at least ten days up to 30 days after disease resolution [143,144,145]. In these studies, gut dysbiosis also correlated with increased COVID-19 severity and inflammatory biomarkers and prolonged SARS-CoV-2 faecal shedding. However, it is unclear if such gut dysbiosis extends beyond 30 days. Notwithstanding this uncertainty, since the gut is closely intertwined with the immune system, a review has implicated the accompanying gut microbiome in numerous diseases related to chronic inflammation [146]. It has also been reviewed that the gut microbiome modulates the neurotransmitter circuitries in the gut and brain via the microbiota-gut-brain axis [147]. Hence, persistent gut dysbiosis may also contribute to the gastrointestinal and neurological symptoms of long COVID.
Possible risk factors
Biomarkers
Elevated blood urea nitrogen (BUN) and D-dimer levels were found to be risk factors for pulmonary dysfunction among survivors of COVID-19 at three-month post-hospital discharge [46]. Other studies have shown that COVID-19 pulmonary lesions at two-month post-admission were associated with elevated systemic inflammatory biomarkers, such as D-dimer, interleukin-6 (IL-6), and CRP [62,148]. Systemic inflammatory biomarkers (e.g. CRP, procalcitonin, and neutrophil count) also correlated with radiological abnormalities of the heart, liver, and kidney in a 2- to 3-month follow-up study of discharged COVID-19 patients [93]. In another study, increased D-dimer and CRP levels and decreased lymphocytes were more common in COVID-19 survivors who developed persistent symptoms than their fully recovered counterparts [149]. Another report also found that lymphopenia correlated with chest tightness and heart palpitations, whereas elevated troponin-1 correlated with fatigue, among sufferers of long COVID [29]. Therefore, changes in levels of D-dimer, CRP, and lymphocyte appeared consistent in a few studies, and may serve as potential biomarkers of long COVID.
However, other studies have found no changes in pro-inflammatory biomarkers (e.g. CRP, D-dimer, IL-6, CD25, and neutrophil and lymphocyte counts) between COVID-19 cases with and without persistent symptoms [39,41,150,151,46]. Such discrepancies may be due to different study methods as studies differ in their sample characteristics, measured endpoints, and data collection and analyses. Another reason may be the heterogeneous and relapsing-remitting nature of long COVID with multifaceted symptomatic presentations [15] (Table 2). This hints at the possible involvement of multiple pathophysiology, with each type possessing a unique set of biomarkers that may even fluctuate. Indeed, inflammatory biomarkers in autoimmune and other chronic inflammatory diseases are known to fluctuate depending on the disease activity and patient’s characteristics [152,153].
Patient and clinical characteristics
One study has revealed that COVID-19 survivors who developed persistent fatigue at 10-week post-discharge were more likely females and persons with a history of anxiety or depression diagnosis or antidepressant usage [39]. Similarly, in another study of COVID-19 survivors who developed persistent symptoms, the associated risk factors include female sex and prior psychiatric disorder [154]. More recent studies have also found higher rates of long COVID symptoms in females than males a few months after hospital discharge [27,33,155,156]. Interestingly, in the first published case series of five children with long COVID, four were females [53]. However, some studies have found that males were as likely as females to develop long COVID [32,36,150]. Therefore, female sex may be at higher risk for certain long COVID manifestations, which require further studies to clarify.
Another study tracked over 4000 COVID-19 survivors and identified factors that predicted long COVID, which include old age of over 70 years, more than five symptoms during the first week of illness, presence of comorbidities, and female sex [37]. In another preprint study, more than five initial presenting symptoms was also a risk factor for long COVID, but not sex or comorbidities [22]. The manifestation of at least 10 symptoms during acute COVID-19 was also found as a risk factor for long COVID in another four-month follow-up study of 434 COVID-19 survivors [36].
Most studies did not find any association between long COVID and initial disease severity during acute COVID-19 [30,31,39,41,154]. However, a few have reported that patients who suffered severe COVID-19 in need of invasive mechanical ventilation, intensive care unit (ICU) admission or prolonged hospitalization were more likely to suffer long-term tissue damage associated with persistent symptoms [38,93,155]. Studies have also revealed high rates of severe functional disabilities and impaired quality of life among COVID-19 survivors discharged from the ICU three months ago [40,157]. Indeed, survivors of critical disease generally face post-intensive care syndrome (PICS) involving long-term cognitive, mental, and physical sequelae due to extensive tissue damage [158,159]. The possible additive impact of COVID-19 on top of PICS also warrants more investigations.
Therefore, some of the more prominent risk factors of long COVID, supported by at least three studies, are female sex, more than five early symptoms, and initial acute COVID-19 severity. Reasons for the ambiguity in long COVID risk factors may be variances in reporting, study design, and participants’ clinical (e.g. disease severity and treatment received) and demographic (e.g. comorbidities, socioeconomic status, and smoking history) characteristics. Another possibility could be the multifaceted pathophysiology of long COVID, which may target populations with particular phenotypes [49].
Potential treatments
Rehabilitation
The literature thus far has only suggested that rehabilitation may work for treating certain cases of long COVID. According to reviews, in rehabilitation, patients are advised to perform light aerobic exercise paced according to individual capacity. Exercise difficulty levels are increased gradually within tolerated levels until improvements in fatigue and dyspnoea are seen, typically four to six weeks. Rehabilitation also includes breathing exercises that aim to control slow, deep breaths to strengthen respiratory muscles' efficiency, especially the diaphragm. The breath should be inhaled through the nose, expanding the abdominal region, and exhaled via the mouth. Such light aerobic and breathing exercises should be performed daily in 5–10 min sessions throughout the day. Complementary behavioural modification and psychological support may also help improve survivors’ well-being and mental health [16,160,161]. Reviews have also recommended that rehabilitation programs be personalized since long COVID manifestation and pathophysiology may vary in each case [162,163].
In an observational study of 23 discharged COVID-19 patients with on-going symptoms, a personalized multi-disciplinary rehabilitation approach involving breathing, mobilisation, and psychological interventions have improved lung function and physical capacity. However, most participants’ lung function did not heal completely, and persistent neurological symptoms remained [164]. A case series of seven discharged COVID-19 patients with on-going symptoms showed that combined breathing and light exercise rehabilitation healed and improved fatigue symptoms in five and two cases, respectively [165]. Thus far, only one randomized controlled trial (RCT) of 72 elderly COVID-19 survivors has demonstrated that a 6-week rehabilitation program (i.e. involving breathing, stretching and home exercises) improved lung function, exercise capacity, quality of life and anxiety, but not depression [166].
Risks of physical rehabilitation must also be considered. Systematic and scoping reviews have identified that rehabilitation may not be suitable for survivors of critical COVID-19 with severe pulmonary or cardiac damage. Hence, exclusion criteria for post-COVID-19 rehabilitation have been proposed: high resting heart rate (>100 beats/min), low or high blood pressure (<90/60 or >140/90 mmHg), low blood oxygen saturation (<95%), or other conditions where exercise is a contraindication [167]. Indeed, an international survey study found that 85.9% of participants with long COVID experienced symptom relapse following mental or physical activities [23].
Even persons with similar conditions to long COVID (Table 3) may not always respond favourably to physical rehabilitation, which includes patients with postural orthostatic tachycardia syndrome (POTS) or myalgic encephalomyelitis or chronic fatigue syndrome (ME/CFS) with post-exertional malaise [178–180]. Therefore, reviews have emphasised that more RCTs are needed to determine which rehabilitation program would work best for specific groups of long COVID, including those with POTS or ME/CFS [181,182]. The non-pharmaceutical treatment approaches to POTS and ME/CFS may also be repurposed for long COVID cases that share symptoms of POTS and ME/CFS (Table 3). Notably, for ME/CFS, although cognitive behavioural therapy (CBT) and graded exercise therapy (GET) have been the mainstay recommended treatments, they may pose potential harm in some instances [180,183,184].
Table 3.
Descriptions of conditions similar to long COVID with their respective pharmaceutical and non-pharmaceutical treatments.
| Condition | Description | Treatment | Reference |
|---|---|---|---|
| Long COVID or post-COVID-19 syndrome |
|
|
[16,168,17] |
| ME/CFS |
|
|
[169,170,171,172,173] |
| POTS |
|
|
[174,175] |
| MCAS |
|
|
[176,177] |
CBT: cognitive behavioural therapy; GET: graded exercise therapy; CDC: Centres for Disease Control and Prevention; COVID-19: coronavirus disease 2019; IOM: Institute of Medicine; MCAS: mast cell activation syndrome; ME/CFS: myalgic encephalomyelitis or chronic fatigue syndrome; NSAIDs: non-steroidal anti-inflammatory drugs; PEM: post-exertional malaise; POTS: postural orthostatic tachycardia syndrome; TLR3: toll-like receptor 3.
Pharmaceutical treatments
Presently, no pharmaceutical drug has been shown to ameliorate or attenuate symptoms (or radiological and blood biomarker abnormalities) of long COVID in controlled or large-scale cohort studies. However, paracetamol and non-steroidal anti-inflammatory drugs may be used to manage specific symptoms such as fever [16]. However, drugs used to treat similar conditions may hold the potential to be repurposed for long COVID, warranting further research to confirm (Table 3).
Specifically, increasing evidence shows that long COVID resembles ME/CFS and POTS. There have been multiple reports of POTS diagnosis following SARS-CoV-2 infection [174,185–187]. Reviews have suggested that long COVID cases would eventually lead to ME/CFS due to the extensive symptomatic resemblance [136,188,189]. In a survey study of 1146 COVID-19 survivors with persistent symptoms who later sought a medical diagnosis, 13.5% and 10.3% of them received POTS and ME/CFS diagnosis, respectively [23]. A six-month follow-up study discovered that 17.5% of discharged COVID-19 patients still had fatigue, of whom 14.2% met the criteria for ME/CFS [33]. Shared pathophysiology may, therefore, be present among long COVID and POTS or ME/CFS, providing a preliminary basis for further research and potential drug repurposing. Interestingly, a small study of 24 COVID-19 survivors with palpitations or tachycardia found that ivabradine (i.e. a POTS medication) effectively relieved racing heart rate compared to carvedilol (i.e. a blood pressure medication) [168].
Reviews have proposed that mast cell activation syndrome (MCAS) may also underlie long COVID pathophysiology [190,191]. Mast cells serve as a fibroblast-activating factor that could lead to pulmonary fibrosis seen in long COVID sufferers [20,26]. Indeed, SARS-CoV-2 has been shown to trigger inflammatory mast cell responses alongside other immune cells in COVID-19 patients [192,193]. Reviews have also been implicated mast cell activation in the pathophysiology of POTS [175,194]. In contrast to POTS and ME/CFS, there have not been any instances of MCAS diagnosis following COVID-19 in the literature thus far. However, this may be due to the heterogeneous nature of MCAS that has been overlooked in long COVID cases [190]. Hence, the possible involvement of MCAS or mast cell activation pathophysiology in long COVID may need further investigations.
Notably, differences between long COVID and other similar conditions also exist. For example, long COVID entails a myriad of symptoms involving multi-organ systems not usually apparent in POTS and ME/CFS. Dyspnoea, a common symptom of long COVID, is rarely seen in and not part of the diagnostic criteria of ME/CFS and POTS (Table 3). Hence, drug repurposing attempts must consider symptomatic and pathophysiological differences between these similar conditions. Another treatment challenge lies in the heterogeneous nature of long COVID, which likely involves multiple subtypes and complicates accurate diagnosis, as also suggested by other reviews [11,48]. Future studies should also explore and possibly solve these challenges in finding therapeutic drugs for long COVID.
Concluding remarks
This review presents the current understanding of long COVID, a relatively new and puzzling condition that may affect COVID-19 survivors, regardless of initial disease severity or age. The symptoms, putative pathophysiology, associated risk factors, and potential treatments have been discussed. However, much remains ambiguous about long COVID, particularly its risk factors with inconsistent data thus far. This may be due to its multiple symptomatic presentations and pathophysiologies, ranging from long-term damage of multiple organ systems to unresolved inflammation from multiple sources. Hence, future research might be interested in phenotyping subtypes of long COVID [49]. Presently, only rehabilitation has been found as possibly effective in improving symptoms of long COVID, whereas the potential pharmaceutical drugs repurposed from ME/CFS, POTS, and MCAS still require future research to validate.
Evidently, the pandemic has brought us a wave of a new chronic, disabling condition called long COVID that deserves serious attention among the scientific and medical communities to resolve. Assuming at least 10% of COVID-19 survivors develop long COVID, which is likely underestimated (Table 2), it is estimated that 5 million people are facing long COVID globally [5]. The information presented in this review, which has not been communicated extensively elsewhere in the literature, may serve as a starting point for further exploration on long COVID.
Supplementary Material
Acknowledgements
The author thank the editor and peer-reviewers involved in the publication process of this paper.
Funding Statement
No funding was received for this work.
Disclosure statement
The author states that there is no conflict of interest to disclose.
References
- 1.Hu B, Guo H, Zhou P, et al. . Characteristics of SARS-CoV-2 and COVID. Nat Rev Microbiol. 2020;19(3):141–154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Maxwell E. 'Living with Covid19: A dynamic review of the evidence around ongoing Covid19 symptoms (often called Long Covid).', (updated 30th September 2020) <https://evidence.nihr.ac.uk/themedreview/living-with-covid19/>, accessed 31st January 2021.
- 3.Rubin R. As Their Numbers Grow, COVID-19 “Long Haulers” Stump Experts. JAMA. 2020;324(14):1381–1383. [DOI] [PubMed] [Google Scholar]
- 4.Callard F, Perego E.. How and why patients made Long Covid. Social Science & Medicine. 2021;268:113426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Altmann DM, Boyton RJ.. Decoding the unknowns in long covid. BMJ. 2021;372:n132. [DOI] [PubMed] [Google Scholar]
- 6.Baig AM. Deleterious outcomes in long-Hauler COVID-19: the effects of SARS-CoV-2 on the CNS in chronic COVID syndrome. ACS Chem Neurosci. 2020;11(24);4017–4020. [DOI] [PubMed] [Google Scholar]
- 7.Venkatesan P. NICE guideline on long COVID'. The Lancet Respiratory Medicine. 2021;9(2):129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Nath A. Long-Haul COVID. Neurology. 2020;95(13):559–560. [DOI] [PubMed] [Google Scholar]
- 9.Brodin P. Immune determinants of COVID-19 disease presentation and severity. Nat Med. 2021;27(1):28–33. [DOI] [PubMed] [Google Scholar]
- 10.Davido B, et al. . Post-COVID-19 chronic symptoms: a postinfectious entity? Clin Microbiol Infect. 2020;26(11):1448–1449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Datta SD, Talwar A, Lee JT.. A proposed framework and timeline of the spectrum of disease due to SARS-CoV-2 infection: illness beyond acute infection and public health implications. JAMA. 2020;324(22):2251–2252. [DOI] [PubMed] [Google Scholar]
- 12.Mendelson M, Nel J, Blumberg L, et al. . Long-COVID: An evolving problem with an extensive impact. S Afr Med J. 2020;111(1):10–12. [DOI] [PubMed] [Google Scholar]
- 13.Sivan M, Taylor S.. NICE guideline on long covid. BMJ. 2020;371:m4938. [DOI] [PubMed] [Google Scholar]
- 14.Nalbandian A, Sehgal K, Gupta A, et al. . Post-acute COVID-19 syndrome. Nat Med. 2021;27(4):601–615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fernandez-de-Las-Penas C, Palacios-Cena D, Gomez-Mayordomo V, et al. . Defining post-COVID symptoms (post-acute COVID, long COVID, persistent post-COVID): an integrative classification'. Int J Environ Res Public Health. 2021;18(5):2621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Greenhalgh T, Knight M, A’Court C, et al. . Management of post-acute covid-19 in primary care. BMJ. 2020;370:m3026. [DOI] [PubMed] [Google Scholar]
- 17.Shah W, Hillman T, Playford ED, et al. . Managing the long term effects of covid-19: summary of NICE, SIGN, and RCGP rapid guideline. BMJ. 2021;372:n136. [DOI] [PubMed] [Google Scholar]
- 18.Cares-Marambio K, Montenegro-Jiménez Y, Torres-Castro R, et al. . Prevalence of potential respiratory symptoms in survivors of hospital admission after coronavirus disease 2019 (COVID-19): A systematic review and meta-analysis. Chron Respir Dis. 2021;18:14799731211002240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Arnold DT, Hamilton FW, Milne A, et al. . Patient outcomes after hospitalisation with COVID-19 and implications for follow-up: results from a prospective UK cohort. Thorax. 2020;76:399–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Bellan M, Soddu D, Balbo PE, et al. . Respiratory and psychophysical sequelae among patients with COVID-19 four months after hospital discharge. JAMA Netw Open. 2021;4(1):e2036142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Carfi A, et al. . Persistent symptoms in patients after acute COVID-19. JAMA. 2020;324(6):603–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cirulli ET, Schiabor Barrett KM, Riffle S, et al. . Long-term COVID-19 symptoms in a large unselected population. medRxiv. 2020. DOI: 10.1101/2020.10.07.20208702 [DOI] [Google Scholar]
- 23.Davis HE, Assaf GS, McCorkell L, et al. . Characterizing long COVID in an International Cohort: 7 months of symptoms and their impact. medRxiv. 2020. DOI: 10.1101/2020.12.24.20248802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dennis A, Wamil M, Alberts J, et al. . Multiorgan impairment in low-risk individuals with post-COVID-19 syndrome: a prospective, community-based study. BMJ Open. 2021;11(3):e048391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Garrigues E, Janvier P ,Kherabi Y, et al. . Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J Infect. 2020;81(6):e4–e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Han X, Fan Y, Alwalid O, et al. . Six-month follow-up chest CT findings after severe COVID-19 pneumonia. Radiology. 2021;299(1):E177–E186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Huang C, Huang L, Wang Y, et al. . 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. The Lancet. 2021;397(10270):220–232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lerum TV, Aaløkken TM, Brønstad E, et al. . Dyspnoea, lung function and CT findings three months after hospital admission for COVID. Eur Respir J. 2021;57(4):2003448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Liang L, Yang B, Jiang N, et al. . Three-month Follow-up Study of Survivors of Coronavirus Disease 2019 after Discharge. J Korean Med Sci. 2020;35(47):e418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lu Y, Li X, Geng D, et al. . Cerebral micro-structural changes in COVID-19 patients – An MRI-based 3-month follow-up study. EClinicalMedicine. 2020;25:100484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Miyazato Y, Morioka S, Tsuzuki S, et al. . Prolonged and late-onset symptoms of coronavirus disease 2019. Open Forum Infect Dis. 2020;7(11):ofaa507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Petersen MS, Kristiansen MF, Hanusson KD, et al. . Long COVID in the Faroe Islands – a longitudinal study among non-hospitalized patients. Clin Infect Dis. 2020;ciaa1792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Simani L, Ramezani M, Darazam IA, et al. . Prevalence and correlates of chronic fatigue syndrome and post-traumatic stress disorder after the outbreak of the COVID-19. J Neurovirol. 2021;27(1):154–159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shah AS, Wong AW, Hague CJ, et al. . A prospective study of 12-week respiratory outcomes in COVID-19-related hospitalisations. Thorax. 2021;76(4):402–404. [DOI] [PubMed] [Google Scholar]
- 35.Sonnweber T, Sahanic S, Pizzini A, et al. . Cardiopulmonary recovery after COVID-19 – an observational prospective multi-center trial'. ' Eur Respir J. 2020;57(4):2003481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Stavem K, Ghanima W, Olsen MK, et al. . 1.5-6 months after COVID-19 in non-hospitalised subjects: a population-based cohort study. Thorax. 2021;76(4):405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sudre CH, Murray B, Varsavsky T, et al. . Attributes and predictors of long COVID. Nat Med. 2021;27(4):626–631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Taboada M, Carinena A, Moreno E, et al. . Post-COVID-19 functional status six-months after hospitalization. J Infect. 2020;82(4):e31–e33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Townsend L, Dyer AH, Jones K, et al. . Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. PLoS One. 2020;15(11):e0240784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Truffaut L, Demey L, Bruyneel AV, et al. . Post-discharge critical COVID-19 lung function related to severity of radiologic lung involvement at admission. Respir Res. 2021;22(1):29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.van den Borst B, et al. . Comprehensive health assessment three months after recovery from acute COVID-19. Clin Infect Dis. 2020;ciaa1750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.van Gassel RJJ, Bels JLM, Raafs A, et al. . High Prevalence of Pulmonary Sequelae at 3 Months After Hospital Discharge in Mechanically Ventilated COVID-19 Survivors. Am J Respir Crit Care Med. 2021;203(3):371–374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wong AW, Shah AS, Johnston JC, et al. . Patient-reported outcome measures after COVID-19: a prospective cohort study. Eur Respir J. 2020;56(5):2003276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Woo MS, Malsy J, Pottgen J, et al. . Frequent neurocognitive deficits after recovery from mild COVID-19. Brain Commun. 2020;2(2):fcaa205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xiong Q, Xu M, Li J, et al. . Clinical sequelae of COVID-19 survivors in Wuhan, China: a single-centre longitudinal study'. Clin Microbiol Infect. 2021;27(1):89–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhao YM, Shang YM, Song WB, et al. . Follow-up study of the pulmonary function and related physiological characteristics of COVID-19 survivors three months after recovery. EClinicalMedicine. 2020;25:100463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.McMahon DE, Gallman AE, Hruza GJ, et al. . Long COVID in the skin: a registry analysis of COVID-19 dermatological duration. The Lancet Infectious Diseases. 2021;21(3):313–314. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Amenta EM, et al. . Postacute COVID-19: An Overview and Approach to Classification. Open Forum Infect Dis. 2020;7(12):ofaa509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Rando HM, Bennett TD, Byrd JB, et al. . Challenges in defining Long COVID: Striking differences across literature, Electronic Health Records, and patient-reported information. medRxiv. 2021. DOI: 10.1101/2021.03.20.21253896 [DOI] [Google Scholar]
- 50.Townsend L, Dowds J, O'Brien K, et al. . Persistent poor health post-COVID-19 is not associated with respiratory complications or initial disease severity. Ann Am Thorac Soc. 2021. DOI: 10.1513/AnnalsATS.202009-1175OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Buonsenso D, Espuny Pujol F, Munblit D, et al. . Clinical characteristics, activity levels and mental health problems in children with Long COVID: a survey of 510 children. Preprints. 2021. DOI: 10.20944/preprints202103.0271.v1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Buonsenso D, Munblit D, De Rose C, et al. . Preliminary Evidence on Long Covid in children. Acta Paediatr. 2021. DOI: 10.1111/apa.15870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Ludvigsson JF. Case report and systematic review suggest that children may experience similar long-term effects to adults after clinical COVID. Acta Paediatr. 2021;110(3):914–921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Das KM, Lee EY, Singh R, et al. . Follow-up chest radiographic findings in patients with MERS-CoV after recovery. Indian J Radiol Imaging. 2017;27(3):342–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lee SH, Shin H-S, Park HY, et al. . Depression as a Mediator of Chronic Fatigue and Post-Traumatic Stress Symptoms in Middle East Respiratory Syndrome Survivors. Psychiatry Investig. 2019;16(1):59–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Lam MH. Mental morbidities and chronic fatigue in severe acute respiratory syndrome survivors: long-term follow-up. Arch Intern Med. 2009;169(22):2142–2147. [DOI] [PubMed] [Google Scholar]
- 57.Ngai JC, Ko FW, Ng SS, et al. . The long-term impact of severe acute respiratory syndrome on pulmonary function, exercise capacity and health status. Respirology. 2010;15(3):543–550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Rogers JP, Chesney E, Oliver D, et al. . Psychiatric and neuropsychiatric presentations associated with severe coronavirus infections: a systematic review and meta-analysis with comparison to the COVID-19 pandemic. The Lancet Psychiatry. 2020;7(7):611–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang P, Li J, Liu H, et al. . Long-term bone and lung consequences associated with hospital-acquired severe acute respiratory syndrome: a 15-year follow-up from a prospective cohort study. Bone Res. 2020;8(1):8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Zhao FC, Guo KJ, Li ZR.. Osteonecrosis of the femoral head in SARS patients: seven years later. Eur J Orthop Surg Traumatol. 2013;23(6):671–677. [DOI] [PubMed] [Google Scholar]
- 61.Liu D, Zhang W, Pan F, et al. . The pulmonary sequalae in discharged patients with COVID-19: a short-term observational study. Respir Res. 2020;21(1):125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Marvisi M, Ferrozzi F, Balzarini L, et al. . First report on clinical and radiological features of COVID-19 pneumonitis in a Caucasian population: Factors predicting fibrotic evolution. Int J Infect Dis. 2020;99:485–488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wei J, Yang H, Lei P, et al. . Analysis of thin-section CT in patients with coronavirus disease (COVID-19) after hospital discharge. J Xray Sci Technol. 2020;28(3):383–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Li H, Zhao X, Wang Y, et al. . Damaged lung gas-exchange function of discharged COVID-19 patients detected by hyperpolarized (129)Xe MRI. Sci Adv. 2020;7(1):eabc8180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Crameri GAG, Bielecki M, Züst R, et al. . Reduced maximal aerobic capacity after COVID-19 in young adult recruits, Switzerland, May 2020. Euro Surveill. 2020;25(36):2001542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Krishna R, Chapman K, Ullah S.. 2020. 'Idiopathic Pulmonary Fibrosis', StatPearls (Treasure Island (FL)). [Google Scholar]
- 67.Swigris JJ, Streiner DL, Brown KK, et al. . Assessing exertional dyspnea in patients with idiopathic pulmonary fibrosis. Respir Med. 2014;108(1):181–188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Paterson RW, Brown RL, Benjamin L, et al. . The emerging spectrum of COVID-19 neurology: clinical, radiological and laboratory findings. Brain. 2020;143(10):3104–3120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Mao L, Jin H, Wang M, et al. . Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 2020;77(6):683–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Mendez R, Balanzá-Martínez V, Luperdi SC, et al. . Short-term neuropsychiatric outcomes and quality of life in COVID-19 survivors. J Intern Med. 2021. DOI: 10.1111/joim.13262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.O'Hanlon S, Inouye SK.. Delirium: a missing piece in the COVID-19 pandemic puzzle. Age Ageing. 2020;49(4):497–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Girard TD, Jackson JC, Pandharipande PP, et al. . Delirium as a predictor of long-term cognitive impairment in survivors of critical illness. Crit Care Med. 2010;38(7):1513–1520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Gross AL, Jones RN, Habtemariam DA, et al. . Delirium and long-term cognitive trajectory among persons with dementia. Arch Intern Med. 2012;172(17):1324–1331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Dani M, Dirksen A, Taraborrelli P, et al. . Autonomic dysfunction in 'long COVID': rationale, physiology and management strategies. Clin Med. 2021;21(1):e63–e67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Taquet M, Geddes JR, Husain M, et al. . 6-month neurological and psychiatric outcomes in 236 379 survivors of COVID-19: a retrospective cohort study using electronic health records'. The Lancet Psychiatry. 2021;8(5):416–427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ackermann M, Verleden SE, Kuehnel M, et al. . Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. 2020;383(2):120–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Chu H, Chan JF, Yuen TT, et al. . Comparative tropism, replication kinetics, and cell damage profiling of SARS-CoV-2 and SARS-CoV with implications for clinical manifestations, transmissibility, and laboratory studies of COVID-19: an observational study. The Lancet Microbe. 2020;1(1):e14–e23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sun S-H, Chen Q, Gu H-J, et al. . A Mouse Model of SARS-CoV-2 Infection and Pathogenesis. Cell Host Microbe. 2020;28(1):124–133 e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.von Weyhern CH, Kaufmann I, Neff F, et al. . Early evidence of pronounced brain involvement in fatal COVID-19 outcomes. The Lancet. 2020;395(10241):e109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Zhang B-Z, Chu H, Han S, et al. . SARS-CoV-2 infects human neural progenitor cells and brain organoids. Cell Res. 2020;30(10):928–931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Gandhi S, Srivastava AK, Ray U, et al. . Is the collapse of the respiratory center in the brain responsible for respiratory breakdown in COVID-19 patients? ACS Chem Neurosci. 2020;11(10):1379–1381. [DOI] [PubMed] [Google Scholar]
- 82.Li YC, Bai WZ, Hashikawa T.. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J Med Virol. 2020;92(6):552–555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yong SJ. Persistent Brainstem Dysfunction in Long-COVID: A Hypothesis. ACS Chem Neurosci. 2021;12(4):573–580. [DOI] [PubMed] [Google Scholar]
- 84.Lukiw WJ, Pogue A, Hill JM.. SARS-CoV-2 Infectivity and Neurological Targets in the Brain. Cell Mol Neurobiol. 2020. DOI: 10.1007/s10571-020-00947-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Matschke J, Lütgehetmann M, Hagel C, et al. . Neuropathology of patients with COVID-19 in Germany: a post-mortem case series. The Lancet Neurology. 2020;19(11):919–929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Meinhardt J, Radke J, Dittmeyer C, et al. . Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nat Neurosci. 2020;24:168–175. [DOI] [PubMed] [Google Scholar]
- 87.Solomon IH, Normandin E, Bhattacharyya S, et al. . Neuropathological Features of Covid-19. N Engl J Med. 2020;383(10):989–992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Puntmann VO, Carerj ML, Wieters I, et al. . Outcomes of cardiovascular magnetic resonance imaging in patients recently recovered from Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020;5(11):1265–1273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Rajpal S, Tong MS, Borchers J, et al. . Cardiovascular magnetic resonance findings in competitive athletes recovering from COVID-19 infection. JAMA Cardiol. 2020;6(1):116–118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Moody WE, Liu B, Mahmoud-Elsayed HM, et al. . Persisting Adverse Ventricular Remodeling in COVID-19 Survivors: A Longitudinal Echocardiographic Study. Journal of the American Society of Echocardiography. 2021;34(5):562–566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Del Rio C, Collins LF, Malani P.. Long-term Health Consequences of COVID. JAMA. 2020;324(17):1723. ), [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Mitrani RD, Dabas N, Goldberger JJ.. COVID-19 cardiac injury: Implications for long-term surveillance and outcomes in survivors. Heart Rhythm. 2020;17(11):1984–1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Raman B, Cassar MP, Tunnicliffe EM, et al. . Medium-term effects of SARS-CoV-2 infection on multiple vital organs, exercise capacity, cognition, quality of life and mental health, post-hospital discharge. EClinicalMedicine. 2021;31:100683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Ayoubkhani D, Khunti K, Nafilyan V, et al. . Post-covid syndrome in individuals admitted to hospital with covid-19: retrospective cohort study. BMJ. 2021;372:n693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Carmo A, Pereira‐Vaz J, Mota V, et al. . Clearance and persistence of SARS-CoV-2 RNA in patients with COVID. J Med Virol. 2020;92(10):2227–2231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kandetu TB, Dziuban EJ, Sikuvi K, et al. . Persistence of positive RT-PCR results for over 70 days in two travelers with COVID-19. Disaster Med Public Health Prep. 2020. DOI: 10.1017/dmp.2020.450 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Vibholm LK, Nielsen SSF, Pahus MH, et al. . SARS-CoV-2 persistence is associated with antigen-specific CD8 T-cell responses. EBioMedicine. 2021;64:103230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wang X, Huang K, Jiang H, et al. . Long-term existence of SARS-CoV-2 in COVID-19 patients: host immunity, viral virulence, and transmissibility. Virol Sin. 2020;35(6):793–802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Hirotsu Y, Maejima M, Shibusawa M, et al. . Analysis of a persistent viral shedding patient infected with SARS-CoV-2 by RT-qPCR, FilmArray Respiratory Panel v2.1, and antigen detection. J Infect Chemother. 2020;27(2):406–409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Li Q, Zheng XS, Shen XR, et al. . Prolonged shedding of severe acute respiratory syndrome coronavirus 2 in patients with COVID-19. Emerg Microbes Infect. 2020;9(1):1–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Park SK, Lee C, Park D, et al. . Detection of SARS-CoV-2 in fecal samples from patients with asymptomatic and mild COVID-19 in Korea. Clin Gastroenterol Hepatol. 2020. DOI: 10.1016/j.cgh.2020.06.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wu Y, Guo C, Tang L, et al. . Prolonged presence of SARS-CoV-2 viral RNA in faecal samples. Lancet Gastroenterol Hepatol. 2020;5(5):434–435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Gaebler C, Wang Z, Lorenz JCC, et al. . Evolution of antibody immunity to SARS-CoV-2. Nature. 2021;591(7851):639–644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Karlsson AC, Humbert M, Buggert M.. The known unknowns of T cell immunity to COVID. Sci Immunol. 2020;5(53):19. [DOI] [PubMed] [Google Scholar]
- 105.Ehrenfeld M, Tincani A, Andreoli L, et al. . Covid-19 and autoimmunity. Autoimmun Rev. 2020;19(8):102597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Lui DTW, Lee CH, Chow WS, et al. . Thyroid dysfunction in relation to immune profile, disease status and outcome in 191 patients with COVID-19. J Clin Endocrinol Metab. 2020;106(2):e926–e935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Muller I, Cannavaro D, Dazzi D, et al. . SARS-CoV-2-related atypical thyroiditis. The Lancet Diabetes & Endocrinology. 2020;8(9):739–741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Li Q, Wang B, Mu K, et al. . The pathogenesis of thyroid autoimmune diseases: New T lymphocytes – Cytokines circuits beyond the Th1-Th2 paradigm. J Cell Physiol. 2019;234(3):2204–2216. [DOI] [PubMed] [Google Scholar]
- 109.Zuo Y, Estes SK, Ali RA, et al. . Prothrombotic autoantibodies in serum from patients hospitalized with COVID-19. Sci Transl Med. 2020;12:eabd3876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Bastard P, Rosen LB, Zhang Q, et al. . Autoantibodies against type I IFNs in patients with life-threatening COVID-19. Science. 2020;(6515):370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Gao ZW, Zhang H, Liu C, et al. . Autoantibodies in COVID-19: frequency and function. Autoimmun Rev. 2021;20(3):102754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Vlachoyiannopoulos PG, Magira E, Alexopoulos H, et al. Autoantibodies related to systemic autoimmune rheumatic diseases in severely ill patients with COVID-19. Ann Rheum Dis. 2020;79(12):1661–1663. [DOI] [PubMed] [Google Scholar]
- 113.Zhou Y, Han T, Chen J, et al. . Clinical and autoimmune characteristics of severe and critical cases of COVID. Clin Transl Sci. 2020;13(6):1077–1086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Elkon K, Casali P.. Nature and functions of autoantibodies. Nat Clin Pract Rheumatol. 2008;4(9):491–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Cojocaru M, Cojocaru IM, Silosi I, et al. . Manifestations of systemic lupus erythematosus. Maedica (Bucur). 2011;6(4):330–336. [PMC free article] [PubMed] [Google Scholar]
- 116.Guo Q, Wang Y, Xu D, et al. . Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res. 2018;6:15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Fathi N, Rezaei N.. Lymphopenia in COVID-19: therapeutic opportunities. Cell Biol Int. 2020;44(9):1792–1797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Tavakolpour S, Rakhshandehroo T, Wei EX, et al. . Lymphopenia during the COVID-19 infection: What it shows and what can be learned. Immunol Lett. 2020;225:31–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Cheng Y, Zhao H, Song P, et al. . Dynamic changes of lymphocyte counts in adult patients with severe pandemic H1N1 influenza A. J Infect Public Health. 2019;12(6):878–883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Kong M, Zhang H, Cao X, et al. . Higher level of neutrophil-to-lymphocyte is associated with severe COVID-19. Epidemiol Infect. 2020;148:e139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Danwang C, Endomba FT, Nkeck JR, et al. . A meta-analysis of potential biomarkers associated with severity of coronavirus disease 2019 (COVID-19). Biomark Res. 2020;8(1):37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Malik P, Patel U, Mehta D, et al. . Biomarkers and outcomes of COVID-19 hospitalisations: systematic review and meta-analysis. BMJ Evid Based Med. 2020. DOI: 10.1136/bmjebm-2020-111536 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Ou M, Zhu J, Ji P, et al. . Risk factors of severe cases with COVID-19: a meta-analysis. Epidemiol Infect. 2020;148:e175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Hu F, Chen F, Ou Z, et al. . A compromised specific humoral immune response against the SARS-CoV-2 receptor-binding domain is related to viral persistence and periodic shedding in the gastrointestinal tract. Cell Mol Immunol. 2020;17(11):1119–1125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Liu B, Han J, Cheng X, et al. . Reduced numbers of T cells and B cells correlates with persistent SARS-CoV-2 presence in non-severe COVID-19 patients. Sci Rep. 2020;10(1):17718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Amato MK, Hennessy C, Shah K, et al. . Multisystem inflammatory syndrome in an adult. J Emerg Med. 2021. DOI: 10.1016/j.jemermed.2021.02.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Belot A, Antona D, Renolleau S, et al. . SARS-CoV-2-related paediatric inflammatory multisystem syndrome, an epidemiological study, France, 1 March to 17 May 2020. Euro Surveill. 2020;25(22):2001010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Morris SB, Schwartz NG, Patel P, et al. . Case Series of Multisystem Inflammatory Syndrome in Adults Associated with SARS-CoV-2 Infection – United Kingdom and United States, March-August 2020. MMWR Morb Mortal Wkly Rep. 2020;69(40):1450–1456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Toubiana J, Poirault C, Corsia A, et al. . Kawasaki-like multisystem inflammatory syndrome in children during the covid-19 pandemic in Paris, France: prospective observational study. BMJ. 2020;369:m2094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Whittaker E, Bamford A, Kenny J, et al. . Clinical characteristics of 58 children with a pediatric inflammatory multisystem syndrome temporally associated with SARS-CoV-2. JAMA. 2020;324(3):259–269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Roe K. A viral infection explanation for Kawasaki disease in general and for COVID-19 virus-related Kawasaki disease symptoms. Inflammopharmacol. 2020;28(5):1219–1222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Sollini M, et al. . Vasculitis changes in COVID-19 survivors with persistent symptoms: an [(18)F]FDG-PET/CT study'. Eur J Nucl Med Mol Imaging. 2020;48(5):1460–1466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Zhou M, Yin Z, Xu J, et al. . Inflammatory profiles and clinical features of COVID-19 survivors three months after discharge in Wuhan, China. J Infect Dis. 2021;jiab181. DOI: 10.1093/infdis/jiab181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Kucuk A, Cumhur Cure M, Cure E.. Can COVID-19 cause myalgia with a completely different mechanism? A hypothesis. Clin Rheumatol. 2020;39(7):2103–2104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Lacourt TE, Vichaya EG, Chiu GS, et al. . The high costs of low-grade inflammation: persistent fatigue as a consequence of reduced cellular-energy availability and non-adaptive energy expenditure. Front Behav Neurosci. 2018;12:78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Komaroff AL, Bateman L.. Will COVID-19 Lead to Myalgic Encephalomyelitis/Chronic Fatigue Syndrome? Front Med. 2021;7: 1132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Maksoud R, Preez S, Eaton-Fitch N, et al. A systematic review of neurological impairments in myalgic encephalomyelitis/ chronic fatigue syndrome using neuroimaging techniques. PLoS One. 2020;15(4):e0232475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Lamers MM, Beumer J, Vaart J, et al. . SARS-CoV-2 productively infects human gut enterocytes. Science. 2020;369(6499):50–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Xiao F, Tang M, Zheng X, et al. . Evidence for gastrointestinal infection of SARS-CoV-2. Gastroenterology. 2020;158(6):1831–1833 e3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Zang R, Castro MFG, McCune BT, et al. . TMPRSS2 and TMPRSS4 promote SARS-CoV-2 infection of human small intestinal enterocytes. Sci Immunol. 2020;5(47):eabc3582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Cheung KS, Hung IFN, Chan PPY, et al. . Gastrointestinal manifestations of SARS-CoV-2 infection and virus load in fecal samples from a Hong Kong Cohort: systematic review and meta-analysis. Gastroenterology. 2020;159(1):81–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Mao R, Qiu Y, He JS, et al. . Manifestations and prognosis of gastrointestinal and liver involvement in patients with COVID-19: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2020;5(7):667–678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Yeoh YK, Zuo T, Lui GC-Y, et al. . Gut microbiota composition reflects disease severity and dysfunctional immune responses in patients with COVID-19. Gut. 2021;70(4):698–706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Zuo T, Zhan H, Zhang F, et al. . Alterations in Fecal Fungal Microbiome of Patients With COVID-19 During Time of Hospitalization until Discharge. Gastroenterology. 2020;159(4):1302–1310 e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Zuo T, Zhang F, Lui GCY, et al. . Alterations in gut microbiota of patients with covid-19 during time of hospitalization. Gastroenterology. 2020;159(3):944–955 e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Belkaid Y, Hand TW.. Role of the microbiota in immunity and inflammation. Cell. 2014;157(1):121–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Yong SJ, Tong T, Chew J, et al. . Antidepressive mechanisms of probiotics and their therapeutic potential. Front Neurosci. 2019;13:1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Liao B, Liu Z, Tang L, et al. . Longitudinal clinical and radiographic evaluation reveals interleukin-6 as an indicator of persistent pulmonary injury in COVID-19. Int J Med Sci. 2021;18(1):29–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Mandal S, Barnett J, Brill SE, et al. . Long-COVID': a cross-sectional study of persisting symptoms, biomarker and imaging abnormalities following hospitalisation for COVID. Thorax. 2021;76(4):396–398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Moreno-Pérez O, Merino E, Leon-Ramirez J-M, et al. . Post-acute COVID-19 Syndrome. Incidence and risk factors: a Mediterranean cohort study. Journal of Infection. 2021;82(3):378–383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Salmon D, Slama D, Broucker TD, et al. . Clinical, virological and imaging profile in patients with Persistent or Resurgent forms of COVID-19: a cross-sectional study. J Infect. 2020;82(2): e1–e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Katsanos AH, Kyriakidi K, Karassa FB, et al. . Biomarker development in chronic inflammatory diseases'. Biomarkers for Endometriosis. 2017;41–75. [Google Scholar]
- 153.Tektonidou MG, Ward MM.. Validation of new biomarkers in systemic autoimmune diseases. Nat Rev Rheumatol. 2011;7(12):708–717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Poyraz BC, Poyraz CA, Qlgun Y, et al. . Psychiatric morbidity and protracted symptoms after COVID-19. Psychiatry Res. 2020;295:113604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Halpin SJ, Mclvor C, Whyatt G, et al. . Postdischarge symptoms and rehabilitation needs in survivors of COVID-19 infection: A cross-sectional evaluation. J Med Virol. 2021;93(2):1013–1022. [DOI] [PubMed] [Google Scholar]
- 156.Sigfrid L, Drake TM, Pauley E, et al. . Long Covid in adults discharged from UK hospitals after Covid-19: A prospective, multicentre cohort study using the ISARIC WHO Clinical Characterisation Protocol. medRxiv. 2021. DOI:10.1101 [DOI] [PMC free article] [PubMed] [Google Scholar]; /2021.0 [Google Scholar]; 3.18.21253888 [Google Scholar]
- 157.Valent A, Dudoignon E, Ressaire Q, et al. . Three-month quality of life in survivors of ARDS due to COVID-19: A preliminary report from a French academic centre. Anaesth Crit Care Pain Med. 2020;39(6):740–741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Inoue S, Hatakeyama J, Kondo Y, et al. . Post-intensive care syndrome: its pathophysiology, prevention, and future directions. Acute Med Surg. 2019;6(3):233–246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Rawal G, Yadav S, Kumar R.. Post-intensive Care Syndrome: an Overview. J Transl Int Med. 2017;5(2):90–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Greenhalgh T, Knight M.. Long COVID: a primer for family physicians. Am Fam Physician. 2020;102(12):716–717. [PubMed] [Google Scholar]
- 161.Wang TJ, Chau B, Lui M, et al. . Physical Medicine and Rehabilitation and Pulmonary Rehabilitation for COVID-19. Am J Phys Med Rehabil. 2020;99(9):769–774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Barker-Davies RM, Sullivan OO, Senaratne KPP, et al. . The Stanford Hall consensus statement for post-COVID-19 rehabilitation. Br J Sports Med. 2020;54(16):949–959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Sivan M, Rayner C, Delaney B.. Fresh evidence of the scale and scope of long covid'. BMJ. 2021;373:n853. [DOI] [PubMed] [Google Scholar]
- 164.Puchner B, Sahanic S, Kirchmair R, et al. . Beneficial effects of multi-disciplinary rehabilitation in post-acute COVID-19 – an observational cohort study. Eur J Phys Rehabil Med. 2021;57(2):189–198. [DOI] [PubMed] [Google Scholar]
- 165.Ferraro F, Calafiore D, Dambruoso F, et al. . COVID-19 related fatigue: which role for rehabilitation in post-COVID-19 patients? A case series. J Med Virol. 2021;93(4):1896–1899. [DOI] [PubMed] [Google Scholar]
- 166.Liu K, Zhang W, Yang Y, et al. Respiratory rehabilitation in elderly patients with COVID-19: A randomized controlled study. Complement Ther Clin Pract. 2020;39:101166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Demeco A, Marotta N, Barletta M, et al. . Rehabilitation of patients post-COVID-19 infection: a literature review. J Int Med Res. 2020;48(8):300060520948382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Jadhav K, Jariwala P.. ‘Ivabradin’ versus ‘Carvedilol’ in the management of Post-COVID-19 palpitation with sinus tachycardia. Indian Heart Journal. 2020;72:S33. [Google Scholar]
- 169.Fukuda K, Straus SE, Hickie I, et al. . The chronic fatigue syndrome: a comprehensive approach to its definition and study. International Chronic Fatigue Syndrome Study Group. Ann Intern Med. 1994;121(12):953–959. [DOI] [PubMed] [Google Scholar]
- 170.Kim DY, Lee JS, Park SY, et al. . Systematic review of randomized controlled trials for chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME). J Transl Med. 2020;18(1):7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Richman S, Morris MC, Bordrick G, et al. . Pharmaceutical interventions in chronic fatigue syndrome: a literature-based commentary. Clin Ther. 2019;41(5):798–805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Sapra A, Bhandari P.. 2020. 'Chronic Fatigue Syndrome', StatPearls (Treasure Island (FL)). [Google Scholar]
- 173.Strayer DR, Young D, Mitchell WM.. Effect of disease duration in a randomized Phase III trial of rintatolimod, an immune modulator for Myalgic Encephalomyelitis/Chronic Fatigue Syndrome. PLoS One. 2020;15(10):e0240403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Blitshteyn S, Whitelaw S.. Postural orthostatic tachycardia syndrome (POTS) and other autonomic disorders after COVID-19 infection: a case series of 20 patients. Immunol Res. 2021;69(2):212–212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Bryarly M, Philips LT, Fu Q, et al. . Postural orthostatic tachycardia syndrome: JACC focus seminar. J Am Coll Cardiol. 2019;73(10):1207–1228. [DOI] [PubMed] [Google Scholar]
- 176.Akin C, Valent P, Metcalfe DD.. Mast cell activation syndrome: Proposed diagnostic criteria. J Allergy Clin Immunol. 2010;126(6):1099–1104 e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Molderings GJ, Brettner S, Homann J, et al. . Mast cell activation disease: a concise practical guide for diagnostic workup and therapeutic options. J Hematol Oncol. 2011;4(1):10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Fu Q, Levine BD.. Exercise and non-pharmacological treatment of POTS. Auton Neurosci. 2018;215:20–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Larun L, Brurberg KG, Odgaard-Jensen J, et al. . Exercise therapy for chronic fatigue syndrome. Cochrane Database Syst Rev. 2019;10:CD003200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Twisk FN, Maes M.. A review on cognitive behavorial therapy (CBT) and graded exercise therapy (GET) in myalgic encephalomyelitis (ME) / chronic fatigue syndrome (CFS): CBT/GET is not only ineffective and not evidence-based, but also potentially harmful for many patients with ME/CFS. Neuro Endocrinol Lett. 2009;30(3):284–299. [PubMed] [Google Scholar]
- 181.Negrini F, de Sire A, Andrenelli E, The International Multiprofessional Steering Committee of Cochrane Rehabilitation REH-COVER action, et al.. Rehabilitation and COVID-19: a rapid living systematic review 2020 by Cochrane Rehabilitation Field. Update as of October 31st, 2020. Eur J Phys Rehabil Med. 2020;56(5). [DOI] [PubMed] [Google Scholar]
- 182.Siddiq MAB. Pulmonary Rehabilitation in COVID-19 patients: A scoping review of current practice and its application during the pandemic. Turk J Phys Med Rehab. 2020;66(4):480–494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Kindlon T. Do graded activity therapies cause harm in chronic fatigue syndrome? J Health Psychol. 2017;22(9):1146–1154. [DOI] [PubMed] [Google Scholar]
- 184.Torjesen I. NICE backtracks on graded exercise therapy and CBT in draft revision to CFS guidance. BMJ. 2020;371:m4356. [DOI] [PubMed] [Google Scholar]
- 185.Johansson M, Stahlberg M, Runold M, et al. . Long-Haul Post-COVID-19 symptoms presenting as a variant of postural orthostatic tachycardia syndrome: the Swedish experience. JACC Case Rep. 2021;3(4):573–580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Kanjwal K, Jamal S, Kichloo A, et al. . New-onset Postural Orthostatic Tachycardia Syndrome Following Coronavirus Disease 2019 Infection. J Innov Cardiac Rhythm Manage. 2020;11(11):4302–4304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Miglis MG, Prieto T, Shaik R, et al. . A case report of postural tachycardia syndrome after COVID-19. Clin Auton Res. 2020;30(5):449–451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Miller A. COVID-19: not just an acute illness. Trends Urology & Men Health. 2020;11(6):17–19. [Google Scholar]
- 189.Perrin R, Riste L, Hann M, et al. . Into the looking glass: Post-viral syndrome post COVID-19. Med Hypotheses. 2020;144:110055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Afrin LB, Weinstock LB, Molderings GJ.. Covid-19 hyperinflammation and post-Covid-19 illness may be rooted in mast cell activation syndrome. Int J Infect Dis. 2020;100:327–332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Kazama I. Stabilizing mast cells by commonly used drugs: a novel therapeutic target to relieve post-COVID syndrome? Drug Discov Ther. 2020;14(5):259–261. [DOI] [PubMed] [Google Scholar]
- 192.Gebremeskel S, Schanin J, Coyle KM, et al. . Mast cell and eosinophil activation are associated with COVID-19 and TLR-mediated viral inflammation: implications for an anti-Siglec-8 antibody. Front Immunol. 2021;12:650331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Zhou Z, Ren L, Zhang L, et al. . Heightened innate immune responses in the respiratory tract of COVID-19 patients. Cell Host Microbe. 2020;27(6):883–890 e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Shibao C, Arzubiaga C, Roberts LJ, et al. . Hyperadrenergic postural tachycardia syndrome in mast cell activation disorders. Hypertension. 2005;45(3):385–390. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.