Abstract
Background
NY-ESO-1 is a tumor-specific, highly immunogenic, human germ cell antigen of the MAGE-1 family that is a promising vaccine and cell therapy candidate in clinical trial development. The mouse genome does not encode an NY-ESO-1 homolog thereby not subjecting transgenic T-cells to thymic tolerance mechanisms that might impair in-vivo studies. We hypothesized that an NY-ESO-1 T cell receptor (TCR) transgenic mouse would provide the unique opportunity to study avidity of TCR response against NY-ESO-1 for tumor vaccine and cellular therapy development against this clinically relevant and physiological human antigen.
Methods
To study in vitro and in vivo the requirements for shaping an effective T cell response against the clinically relevant NY-ESO-1, we generated a C57BL/6 HLA-A*0201 background TCR transgenic mouse encoding the 1G4 TCR specific for the human HLA-A2 restricted, NY-ESO-1157-165 SLLMWITQC (9C), initially identified in an NY-ESO-1 positive melanoma patient.
Results
The HLA-A*0201 restricted TCR was positively selected on both CD4+ and CD8+ cells. Mouse 1G4 T cells were not activated by endogenous autoimmune targets or a large library of non-cognate viral antigens. In contrast, their activation by HLA-A2 NY-ESO-1157-165 complexes was evident by proliferation, CD69 upregulation, interferon-γ production, and interleukin-2 production, and could be tuned using a twofold higher affinity altered peptide ligand, NY-ESO-1157-165V. NY-ESO-1157-165V recombinant vaccination of syngeneic mice adoptively transferred with m1G4 CD8+ T cells controlled tumor growth in vivo. 1G4 transgenic mice suppressed growth of syngeneic methylcholanthrene (MCA) induced HHD tumor cells expressing the full-length human NY-ESO-1 protein but not MCA HHD tumor cells lacking NY-ESO-1.
Conclusions
The 1G4 TCR mouse model for the physiological human TCR against the clinically relevant antigen, NY-ESO-1, is a valuable tool with the potential to accelerate clinical development of NY-ESO-1-targeted T-cell and vaccine therapies.
Keywords: immunotherapy, adoptive, immunity, cellular, CD8-Positive T-Lymphocytes
Introduction
Cancer immunotherapy has shown great promise in the clinical setting. It consists of three pillars that each have demonstrated variable benefit: checkpoint inhibitors,1 adoptive transfer of tumor specific T cells directly2 or via bone marrow transplantation, and cancer vaccines.3 CD8+ T lymphocytes play an important role in all these therapeutic approaches, as tumor CD8+ T cell infiltration is a hallmark of responsiveness to immunotherapy.4 Further, adoptive transfer of autologous tumor-infiltrating CD8+ lymphocytes (TIL) can mediate antitumor immunity.2 5 6 These cells recognize antigen via the interaction of surface T cell receptors (TCRs) with short peptides presented by MHC class I molecules (pMHC complexes).
Efficient in vivo elimination of tumor cells and virus infected cells is correlated with T cell avidity for target cells,7 8 due primarily to TCR affinity, with contribution from costimulatory and cellular adhesion molecules.9 10 Consequently, the affinity/avidity of the TCR-pMHC interaction has been ascribed a critical role in T cell antigen recognition, activation, expansion, and memory generation.11 12 To date, the main work on parsing the contribution to affinity/avidity antitumor immunity has been limited to in vitro12 13 approaches, and is largely based on OT-I (C57BL/6-Tg(TcraTcrb)1100Mjb/Crl) TCR transgenic cells, which represent a highly artificial system for studying human tumor immunity, as it is based on recognition of a mouse class I molecule binding a chicken derived peptide, an extreme example of an avian to mammalian xenoantigen.11
Here, we describe a new tool for scrutinizing the detailed role of affinity and avidity in shaping the quality of tumor antigen associated CD8+ T cell antitumor effector function and immunogenicity, in vitro and in vivo. To this end we generated and characterized a humanized TCR transgenic mouse (designated A2Eso1G4) encoding the 1G4 TCR specific for HLA-A2 complexed with the NY-ESO-1157-165 peptide SLLMWITQC (9C). This TCR (hereafter referred to as m1G4 TCR) was initially identified from an NY-ESO-1 (New York esophageal squamous cell carcinoma 1) positive melanoma patient by our group.14 NY-ESO-1 is a highly immunogenic, tumor-specific human germ cell antigen of the MAGE-1 (Melanoma-associated antigen 1) family that is a promising vaccine and cell therapy target for tumor immunotherapy.15 NY-ESO-1 is 180 residue protein composed of a hydrophobic C-terminus and glycine-rich N-terminus. It has no identified functional or binding domains, and its presence has been localized mainly to the cytoplasm and somewhat to the nucleus using tissue immunohistochemical stainings.16 In general, NY-ESO-1 is exclusively expressed in germ cells and trophoblasts, but is upregulated in 20%–70% of cancers, including 20%–80% of all melanoma, lung, esophageal, liver, gastric, prostate, ovarian, bladder, and synovial sarcoma cancers—leading it to be classified as a cancer testis (CT) antigen. Rarely, it is expressed by lymphomas, colorectal, pancreatic and renal cancers.17–19
Despite being the basis for 34 human clinical trials to date, including promising NY-ESO-1 T cell phase I/II trials,20–23 no CT, NY-ESO-1-specific, stable TCR mouse model has been described. Existing HLA-A2 TCR transgenic mouse models are specific for differentiation antigens gp10024 and tyrosinase25 rather than CT antigens, and do not have the broad array of high-affinity TCRs((12;26 or the library of high affinity altered peptide ligands (APLs)14 27 available for the 1G4 TCR. Additionally, the mouse genome does not encode an NY-ESO-1 homolog thereby not subjecting transgenic T-cells to thymic tolerance mechanisms that might impair in vivo studies. This allows the unique opportunity to use the 1G4 mouse model to study avidity of TCR response against NY-ESO-1 for tumor vaccine and cellular therapy development against this clinically relevant and physiological human antigen.
Materials and methods
Generation of NY-ESO-1 TCR transgenic mice
Transgenic mice with TIL 1G4 TCR reactive to human NY-ESO-1 were developed on a C57BL/6 background Genomic DNA was isolated from the NY-ESO-1-specific CTL line 1G4, an HLA-A2 +human NY-ESO-1157-165 (SLLMWITQC) specific CD8+ T cell line.28 The genomic V-J and V-D-J genomic regions of the TCR α- and β-chains were cloned, sequenced, and subcloned into the TCR cassette vectors described previously29 using XmaI/SacII and XhoI/SacII respectively. The vectors were then coinjected into C57BL/6 embryos yielding transgenic founder lines using the transgenic core facility at the National Institutes of Health/National Cancer Institute Laboratory Animal Sciences Program (Maryland, USA). Transgenic animals were identified by Southern blot analysis performed on tail snips. Animals were maintained in pathogen-free facilities and under the approved procedures of the Institutional Animal Care and Use Committee. DNA sequence analysis was performed utilizing VectorNTI (Invitrogen, USA). Experiments that were performed in Biomedical services in University of Oxford were carried out under Home office licence PBA43A2E4.
αβ 1G4 Chimeric TCR Construct Transfection of BW 58 TCR α-/β-, CD4 -/-, CD8 -/- mouse thymoma cells
Transfections were carried out utilizing Amaxa (Lonza Cologne AG, Germany) with Solution V and program A-23. 2.25 µg pTα, 2.25 µg pTβ, and 0.5 µg human CD8 pIRES vectors were mixed in 100 ul Sol. V and electroporated together. Cells were grown for 2 days in RPMI 1640 (GIBCO, Invitrogen) +10% fetal bovine serum (FCS) at 5% CO2 and then stably selected in media supplemented with G418 1 mg/mL (Melford, Suffolk, UK). Transfected lines were frozen in freezing media (10% dimethylsulphoxide (DMSO), 90% FCS) and stored in liquid nitrogen. Samples were bulk sorted.
Isolation of mouse genomic DNA for genotyping and transgenic HLA-A*0201 HHD monochain integration site analysis
Mouse ear punches or 0.5 cm tail snips were processed using ArchivePure DNA Mouse Tail Kits (5 Prime, Gaithersburg, Maryland, USA) according to manufacturer’s directions or using in-house lysis buffers and Proteinase K. To genotype for HLA-A*0201 homozygous mice after crossing the founder 1G4 transgenics onto the beta2 microglobulin (beta2m) HLA-A2.1 monochain transgenic H-2Db beta2m double knockout mice (HHD) line,30 the HLA-A*0201 monochain’s site of integration was identified utilizing the GenomeWalker Universal Kit (Clontech and31). PCR primers and conditions for genotyping shown in online supplemental table S1.
jitc-2021-002544supp001.pdf (4.6MB, pdf)
Fluorescent in situ hybridization
Aseptically collected mouse ear biopsies were washed three times in Ca and Mg-free phosphate-buffered normal saline (PBS; pH 7.1), resuspended in 400 µL of collagenase type 1 (4000U/ml in R1 supplemented with 1.5 mL of Dulbecco’s Modified Eagle Media (GIBCO/BRL, Gaithersburg, Maryland, USA) and 100units/ml penicillin and 100 ug/mL streptomycin and 50 ng/mL gentamicin sulfate) in 15 mL Falcon tubes, and incubated for 2 hours at 37° digest tissues. Samples were briefly vortex at 1 hour, 1.5 hours, and 2 hours. After 2 hours, supernatants were collected and 1/10th volume of fetal bovine serum (FBS) was added to neutralize collagenase activity. The resulting cell suspensions was centrifuged at 2600 g for 5 min and cell pellets were resuspended in 10 mL of D0 supplemented with 100 U/mL Penicillin-and/100 U/mL streptomycin (GIBCO), 10% heat-inactivated FBS (HyClone), and 50 µg/mL Gentamicin (Cellgro, Medlatech, Virginia, USA), and cultured in NUNC 50 mL flasks (Nalge Nunc International, Rochester, New York, USA). Fluorescent in situ hybridization (FISH) was performed as previously described.32
pMHC and tetramer production
A2Kb tetramers containing mutations at residues 226 and 227, mutant 226/227AK A2Kb monomer construct, were expressed in Escherichia coli as inclusion bodies and refolded and purified with SLLMWITQV peptide and β2m as previously described.33
Generation of full-length NY-ESO-1 recombinant vaccinia virus encoding SLLMWITQC (9C), SLLMWITQV (9V), and SIINFEKL epitopes
The QuikChange II XL Site-Directed mutagenesis kit was used with a pHR-SIN vector containing the 560Kbp full-length NY-ESO-1 cDNA and primers specified to generate 9V epitope encoding full-length NY-ESO-1 using primers P1 and P2 (online supplemental table S2). Intermediate product NY-ESO-1-Ub-mCherry 9C and 9V constructs were generated using overlapping PCR in four steps as depicted in online supplemental table S2. Step 1 using mCherry as template. Step 2 using ubiquitin cDNA template. Step 3 using NY-ESO-1 products from P1 and P2. Step 4, final constructs from step 3 were fused into individual pSC11 vectors34 previously digested with SalI / NotI using an In-Fusion V.2.0 CF liquid PCR cloning kit (Clontech, Mountain View, California, USA). Recombinant Vaccinia Virus (Western Reserve [WR] Strain) was constructed according to established protocols.35 VV-Ub-SLLMWITQC, VV-Ub-SLLMWITQV, and VV-Ub-SIINFEKL were constructed to allow liberation of peptide by cytosolic ubiquitin hydrolase as previously described36
Peptides
NY-ESO-1157-165 SLLMWITQC and SLLMWITQV, as well as OVA257-264 (SIINFEKL) peptides were synthesized by American Peptide Company (California, USA). Peptides were dissolved at 10 mg/mL in DMSO (ATCC, USA).
Cell culture
The B5 cell line was generated by injecting methylcholanthrene (MCA) dissolved in peanut oil subcutaneously into HHD mice, letting tumors develop spontaneously, and growing out a cell line. The B5 cell line was transduced with pHR SIN lentivirus expressing NY-ESO-1 to form the 1F4 cell line by limited dilution cloning and testing for NY-ESO-1 expression. NY-ESO-1 expressing 1F4 and B5 cell lines were maintained in RPMI media (R-10) with 10% FBS and penicillin/streptomycin (100 units/mL). HHD MCA and T2 (ATCC) cells were cultured in RPMI 1640 (GIBCO, Invitrogen)+10% FCS. Cultures kept at 5% CO2. All reagents were obtained from Sigma-Aldrich (USA).
Preparation of single cell suspensions of blood, spleen and lymph nodes for flow cytometry
After euthanization, blood was quickly collected from mice by cardiac puncture and subsequently subjected to RBC lysis. Then the cells were washed three times in fluorescence-activated cell sorter (FACS) buffer before stained for tetramer or surface molecules. Mice spleen and lymph nodes were carefully harvested and mashed through a 70 µm cell strainer with a plunger. After two washes in FACS buffer (PBS containing 1% FBS and 2 mM Ethylenediaminetetraacetic acid [EDTA]), the cells were subjected to RBC lysis (eBiosciences) for 10 min, followed by two washes with FACS buffer.
Immunophenotyping experiments
After euthanization, murine spleen, mesenteric lymph nodes and inguinal lymph nodes were harvested and crushed gently on a 70 µm cell strainer using a 1 mL syringe plunger. After two washes in fluorescence-activated cell sorter (FACS) buffer (PBS containing 1% FBS and 2 mM EDTA), the cells were subjected to red blood cell (RBC) lysis (eBioSciences)) for 5–7 min, followed by two washes with FACS buffer. The single-cell preparations were resuspended in FACS buffer containing 10 µg/mL Fc receptor block and incubated for 15 min. For dendritic cell (DC), macrophages and B cell analysis, lymph node and splenocytes were stained with antibodies against the following multiple surface antigens: anti-CD11c, anti-MHC-II, F4/80, CD11b and CD19. For T cells, natural killer (NK) and natural killer T cell (NKT) cell analysis, cells were stained with anti CD3, anti CD4, anti CD8, anti NK1.1 and anti NKp46 antibodies. Dead cells were stained with Live/Dead fixable near-infrared staining kit (Life Technologies) in PBS for 30 min at 4°C. ice. Surface-stained samples were fixed with FACS buffer containing 0.1% formaldehyde before acquisition. Data analysis was performed using FlowJo software (Treestar).
1G4 T cells proliferation assay
Pan T cells or CD8+ T cells from 1G4 mice spleen were enriched using MACS beads (Pan T cell isolation kit II and CD8a isolation kit, Miltenyi Biotec) and labeled with 5 µM CellTrace Violet (CTV) (ThermoFisher Scientific) following manufacturer’s instructions. Approximately 2×106 CD8+ T cells or 4×106 pan T cells in 200 µL volume were adoptively transferred to HHD mice. After a day postadoptive transfer, mice were infected with 106 PFU of recombinant vaccinia virus (WR strain) expressing wild type NY-ESO-1 (NY-ESO-1 9C) or NY-ESO-1 with the replacement of cysteine amino acid with valine at 165th position (NY-ESO-1 9V), or vaccinia virus expressing SIINFEKL peptide. Spleen were harvested on day 3 postinfection for the analysis of CD8+T cells or CD4+T cells proliferation, which was determined by cell trace violet dilution.
Ex vivo peptide stimulation and flow cytometry
Single cell suspension from spleen prepared as described previously was stimulated with 1 µM native NY-ESO-1 (9C) (SLLMWITQC), mutant NY-ESO-1 (9V) (SLLMWITQV), or SIINFEKL peptide for 6 hours in the presence of Brefeldin A (BFA) and monensin. For tetramer staining, splenocytes (2×106 cells) were pelleted down in 96 well ‘U’ bottom plate and washed once in R-10. Cells were resuspended in NY-ESO-1 9V HLA A2-Kb tetramer in appropriate dilution and incubated at 37°C for 30 min. After tetramer staining, cells were washed two times in FACS buffer and stained for surface antigens: APC Cy7-antimouse TCR beta, FITC-anti-human TCR Vβ13.1, PerCP Cy5.5 antimouse CD4, PE-Cy7 antimouse CD8, BV421- antimouse CD44 on ice for 30 min. Dead cells were stained with fixable aqua dead cell staining kit following manufacturer’s (Life Technologies).
For intracellular staining, cells were incubated in fixation buffer (Life Technologies) for 45 min on ice, followed by two washes in permeabilization buffer (Life Technologies). Then the cells were stained with PE-antimouse interferon γ (IFN-γ) and BV605-antimouse interleukin 2 (IL-2) antibodies in permeabilization buffer for 30 min on ice. Data analyses were performed using FlowJo software (Treestar).
Tumor models
Syngeneic tumor cells were generated from MCA induced tumors from HHD mice. NY-ESO-1 expressing tumor cells (1F4) were generated by transduction with lentivirus that express full length NY-ESO-1. NY-ESO-1 intracellular expression was confirmed by intracellular staining for NY-ESO-1 protein with 1 µg/mL of anti-NY-ESO-1 antibody (clone E978, Thermo Fisher Scientific) for 30 min at room temperature, followed by staining with Goat antimouse IgG FITC (1:1000 dilution) for 30 min on ice. 1×106 NY-ESO-1 expressing 1F4 cells or control B5 tumor cells (transduced with empty lentiviral vector) were subcutaneously injected into 1G4 mice. Tumor sizes were measured with caliper using the following equation (LxWxW)/2.
For the tumor experiments in HHD mice, 106 1F4 cells were subcutaneously injected into the right flank on day 0. On day 6, some mice received 1×105 naïve CD8+ T cells isolated from 1G4 mice by negative selection and subsequently were infected on next day either with 106 pfu of recombinant vaccinia virus (rVV) NY-ESO-1 or rVV SIINFEKL by intravenous tail administration. In order to analyze for potentially confounding VV induced antitumor response, some tumor cells injected mice were infected with 106 pfu of rVV NY-ESO-1 or rVV SIINFEKL without adoptive transfer of m1G4 CD8+ T cells. Tumor volume was determined by caliper measurements using the following equation (LxWxW)/2. Survival end point was 1.2 cm3.
Confirmation of NY-ESO-1 HLA-A2 dependent presentation: B5 cells or 1F4 cells were washed in FACS buffer and preincubated with anti-HLA-A2 antibody (BB7.2) or isotype antibody in specified concentrations for 45 min before coculture with 1G4 CD8 +T cells at an E:T ratio of 3:1 ratio in Iscove’s Modified Dulbecco’s media media with 10% FBS. BFA and monensin were added in the culture (1/1000) and cells were incubated overnight, followed by intracellular staining for IFN-γ secreting CD8 +T cells.
Statistical analysis
Data were analyzed using Prism GraphPad V.9.0 software and statistical significance was determined by unpaired Student’s t-test with correction for multiple comparisons using the Holm-Sidak method. *denotes a significance of <0.05 in Student’s t-test.
Results
Generating and characterizing human 1G4 TCR HLA A2 transgenic mouse, A2Eso1G4
We generated chimeric TCR constructs composed of variable domains from human 1G4 T cell clone rearranged cDNA, and constant regions and transcriptional regulatory elements from mouse sequences29 37 (online supplemental figure S1A–D). To demonstrate surface expression and correct assembly of the chimeric 1G4 TCR, we cloned the coding sequence of chimeric 1G4 into pTα and pTβ cassettes plasmids (Kouskoff et al) which we cotransfected into BW58 TCRαβ deficient mouse thymoma cells. Correct surface expression was shown by staining transfected BW58 cells with the human Vβ13.1 antibody specific for the human 1G4 variable domain (online supplemental figure S1E). In addition, specific recognition of transfected BW58 cells expressing the 1G4 chimeric TCR by NY-ESO-1157-165/HLA-A2 tetramers and increased levels of CD3 confirmed the surface expression of the assembled TCR complex (online supplemental figure S1F, G). We further demonstrated the proper splicing of the alpha/beta TCR constructs in transfected BW58 cells (online supplemental figure S1H).
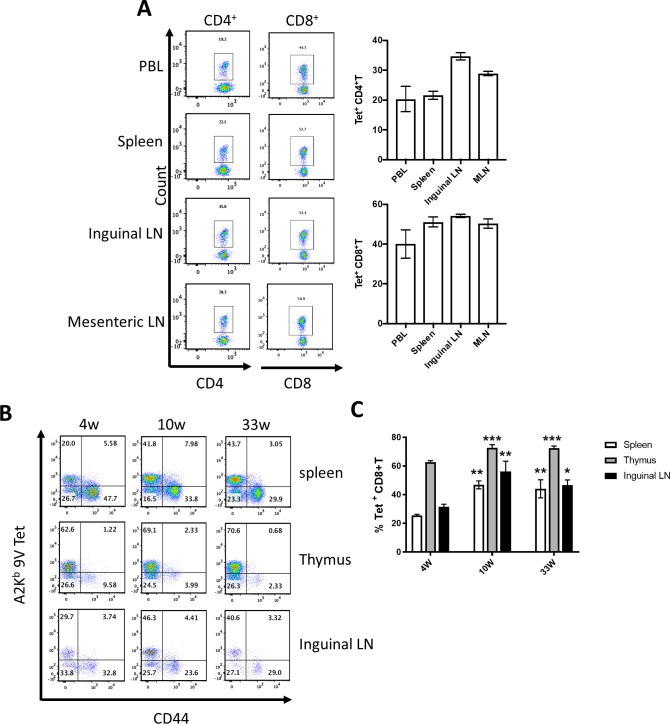
Having confirmed expression of chimeric 1G4 TCR and its ability to bind NY-ESO-1157-165 HLA-A2 tetramers, the 1G4 insert containing pTα and pTβ cassettes were microinjected in equimolar concentrations into C57BL/6 1-day-old embryos, and identified transgenic progeny by Southern blotting. We confirmed germ line integration of the transgenes in two distinct founders, 1G4-A and 1G4-C by FISH and PCR (data not shown and online supplemental figure S1I, J). Founder 1G4-C, with integration of the 1G4 insert into chromosome 9, was chosen for further characterization. As the 1G4 TCR is HLA-A*0201 restricted, we crossed the founder line with HLA–A*0201 expressing HHD mice, (which do not express mouse MHC class-I)30 to obtain mice capable of positively selecting T cells expressing this TCR. These A2Eso1G4 HHD mice were genotyped by PCR during crossing (online supplemental table S1). We confirmed by flow cytometry that mice containing the 1G4 TCRα and β transgenes on HLA-A*0201 (HHD) background were capable of positively selecting T cells, which could be stained with NY-ESO-1157-165/HLA-A2Kb tetramers, engineered to contain the murine CD8 binding site, in peripheral tissues (figure 1A, B).33 Interestingly, while most of these cells were CD8+ (44.3%–53.4%), a substantial population was CD4+CD8- (19.2–35%) T cells. All NY-ESO-1157-165/HLA-A2 tetramer+ mouse T cells (m1G4 T cells) in thymus, spleen and peripheral lymph nodes displayed a naïve T cell phenotype (CD44- negative) in 4, 10 and 33-week-old mice (figure 1B, C). Detailed immunophenotyping revealed generation of all major immune cell populations though significantly decreased levels of B cells and macrophages in mesenteric lymph nodes and spleen, but not inguinal lymph nodes (online supplemental figure S2), and presence of coexpressed murine TCR without allelic exclusion given presence of Vβ13.1+cells negative for tetramer staining (online supplemental figure S3). Negligible NY-ESO-1 reactive T cells are seen by tetramer staining in the endogenous T cell pool of HHD mice (online supplemental figure S3D).
Figure 1.
1G4 transgenic mice develop T cells specific for A2Kb/NY-ESO-1157-165. (A) NY-ESO-1157–165 specific CD4+ and CD8+ T cells from various tissues of naive 9-week-old mice were identified using Tetrameric A2Kb/NY-ESO-1157–165V. Representative FACS plots are shown for peripheral blood lymphocytes (PBL), spleens, and lymph nodes (LN) in the left panels and cumulative data from three mice per group are shown as percentage of CD4+ or CD8+ T cells on the right panels. (B, C) NY-ESO-1157–165 specific CD8+ T cells from various tissues of 4-week-old, 10-week-old, and 33-week-old naïve mice were identified using Tetrameric A2Kb/NY-ESO-1157–165V. Gating strategy for B and C: SSC-A/FSC-A>FSC-A/FSC-H single cells >LiveDead/TCRbeta >TCRbeta/CD8 >Tetramer/CD44. Representative FACS plots are shown in (B) and cumulative data of 3 mice is shown in (C). *P<0.05, **p<0.001, ***p<0.0001 against 4-week-old mice in one way ANOVA, Turkey’s multiple comparisons test. ANOVA, analysis of variance; FACS, fluorescence-activated cell sorter.
We previously demonstrated the specificity of m1G4 T cells for the NY-ESO-1157-165 peptide.22 In this study, we used a library of 5992 synthetic peptides derived from open reading frames of arenaviruses, HBV, HCV, influenza A virus, SARS virus, and vaccinia virus that are predicted to bind HLA-A*0201 with significant affinity (IC50 of <500 nM) to stimulate the m1G4 splenocytes.26 Incubation with target cells exposed to NY-ESO-1157-165C (wild type) or its higher affinity analog NY-ESO-1157-165V, but not any of the other peptides tested induced CD69 expression on m1G4 splenocytes.26
Immune responses of m1G4 CD8+ T cells to APLs of NY-ESO-1
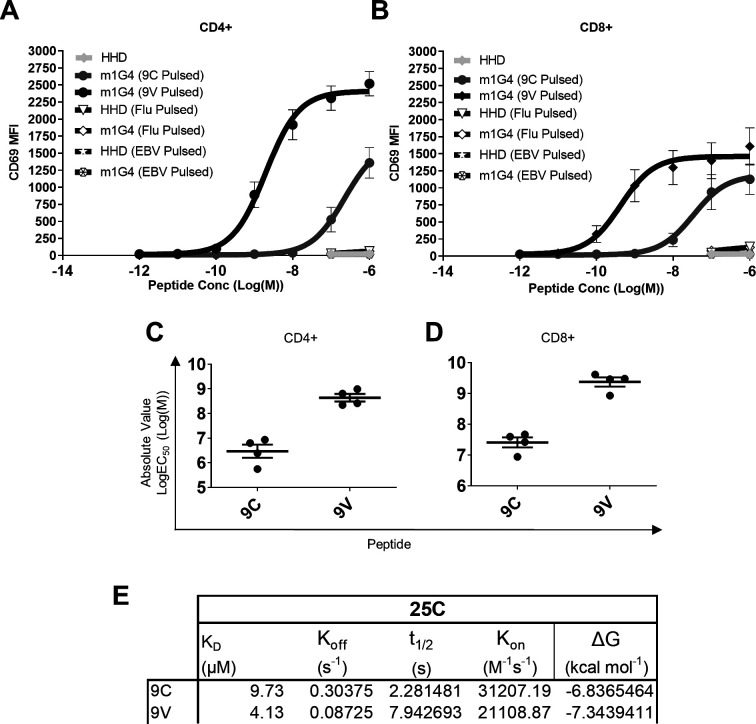
We next investigated the activation, measured by CD69 surface expression, of m1G4 CD4+ (figure 2A) and CD8+ (figure 2B) T cells using increasing concentrations of NY-ESO-1157-165C peptide or its analog NY-ESO-1157-165V. The potency of APLs in the CD69 upregulation assay can be measured by the concentration of the peptide that gives half-maximal response (EC50 value in molar units (M), figure 2C, D) and confirms ~100 fold increased potency of NY-ESO-1157-165V (log10 EC50 ~9) compared with NY-ESO-1157-165C wild-type (log10 EC50 ~7) across both CD4+ and CD8+ T cells. In part this increased sensitivity is due to a twofold increased binding of the 1G4 TCR to the NY-ESO-1157-165V APL, as revealed by surface plasmon resonance binding measurements at 25°C (figure 2C) and 37C°,13(4.1 vs 9.7 µM at 25°, and 7.2 vs 14.4 µM at 37°, respectively).
Figure 2.
Binding properties of the m1G4 TCR to, and m1G4 CD4 +and CD8+T cell responses against, NY-ESO-1 APLs. (A, B) HHD or m1G4 splenocytes were loaded with NY-ESO-1157–165 9C (blue) or 9V (Red) peptides or irrelevant control EBV280-288 or Influenza M158-66 peptides (black). CD69 upregulation on T cells was analyzed by flow cytometry for cells stained with anti-CD4 antibody (A) or anti-CD8 antibody (B). SEM error bars on duplicate or quadruplicate assays are shown. (C, D) EC50 measurement of NY-ESO-1157–165 9C and 9V peptides interaction with m1G4 CD4+ (C) or CD8+ T cells (D) after 17 hour stimulation. Mean with SEM error bars of quadruplicate assays are shown. (E) Surface plasmon resonance affinity measurements for 1G4 TCR binding to A2 complexed with NY-ESO-1157–165 9C or 9V peptides at 25°C. APLs, altered peptide ligand; TCR, T cell receptor.
We then assessed whether HLA-A2 expression on human cells can activate 1G4 T cells. Culturing CD8 +T cells from m1G4 mice overnight with the TAP1/2 deficient T2 cells loaded with different concentrations of NY-ESO-1157-165C wild-type peptide, revealed peptide dose dependent upregulation of CD69 and CD25 (online supplemental figure S4).
In vivo response of A2Eso1G4 m1G4 T cells to infection-mediated antigen presentation
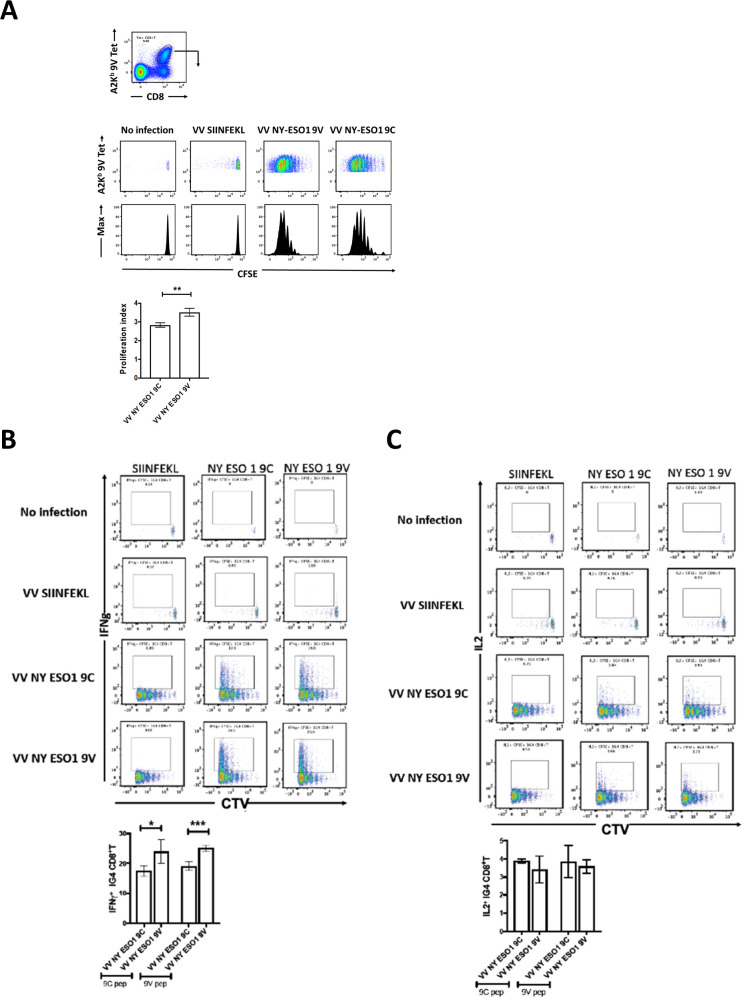
We further investigated m1G4 T cell responses in vivo using by generating recombinant vaccinia viruses (WR) expressing full-length NY-ESO-1 with either wild-type cysteine at position 165 or valine (hereafter referred to as rVV NY-ESO-1 wild type or C165V). The constructs were designed as ubiquitin fusions (Fluorescent protein-Ub-NY-ESO-1 variant) to allow more efficient APL presentation through enhanced proteasomal degradation as previously described.38 39 m1G4 CD8+ T cells were labeled with CTV (a violet version of carboxyfluorescein succinimidyl ester [CFSE]) and transferred to HHD mice that were infected the next day with rVV NY-ESO-1 wild type, C165V, irrelevant SIINFEKL (chicken ovalbumin) peptide, or left uninfected. Three days later the splenic A2Kb/NY-ESO-1157-165 tetramer positive CD8+ m1G4 T cell proliferation fraction was assessed and showed a statistically significant increase in the CTV proliferation index for higher affinity C165V versus wild-type vaccinia infection, and no irrelevant SIINFEKL induced proliferation (figure 3A).
Figure 3.
m1G4 CD8 +T cells show antigen specificity. (A) HHD mice were adoptively transferred via intravenous injection with 2×106 CellTrace Violet (CTV)-labeled naive CD8 +T cells from 1G4 mice and left as uninfected or infected intravenously with 1×106 pfu of rVV expressing full length NY-ESO-1 containing either wild type SLLMWITQC or altered peptide ligand SLLMWITQV. As control, rVV expressing SIINFEKL (from chicken ovalbumin) peptide was used. CTV dilution profiles of splenic m1G4 cells were analyzed at day three postinfection having been stained with A2Kb/NY-ESO-1157–165V tetramer. (B) Splenocytes from the infected mice were restimulated ex vivo with SIINFEKL, SLLMWITQC or SLLMWITQV peptides and IFN-γ expression or (C) IL-2 expression were analyzed by intracellular cytokine staining (ICS) and CTV dilution on day 3. For each panel, representative FACS plots are shown in the top panel and cumulative data for mouse groups (n=5) are plotted below. *P<0.05, **p<0.01, ***p<0.001 in Student’s t-test with GraphPad software. FACS, fluorescence-activated cell sorter; IFN-γ, interferon-γ; IL-2, interleukin 2.
Splenocytes from these mice were then re-stimulated ex vivo with the wild-type NY-ESO-1157-165C peptide (9C), the higher affinity analog NY-ESO-1157-165V peptide (9V) or irrelevant SIINFEKL peptide for 6 hours in the presence of brefeldin-A plus monensin and production of IFN-γ (figure 3B) or IL-2 (figure 3C) was measured. Splenocytes from mock infected or rVV SIINFEKL did not produce any IFN-γ or IL-2, while in mice infected with rVV NY-ESO-1 9C peptide an average of 17% of the m1G4 T cells produced IFN-γ and in mice infected with NY-ESO-1 9V peptide the number of IFN-γ producing m1G4 T cells was statistically higher at an average of 24%. Interestingly, the order of infection and restimulation matters with rVV NY-ESO-1 9V peptide infection producing increased IFN-γ on re-stimulation with either the wild type NY-ESO-1 9C peptide or higher affinity NY-ESO-1 9V peptide. Therefore, it seems that priming the adoptively transferred m1G4 using a higher affinity epitope (rVV NY-ESO-1 C165V) produces T cells that are qualitatively superior (producing more IFN-γ). Note that while we cannot completely exclude the possibility that some of the NY-ESO-1 A2-9V tetramer positive T cells are not adoptively transferred m1G4 but rather derived from endogenous precursor T cells, such cells are highly unlikely to have developed 3 days following priming. The percentage of IL-2 producing m1G4 T cells was much lower than IFN-γ producing, and did not differ between the mice infected with the two different versions of NY-ESO-1 expressing recombinant vaccinia virus.
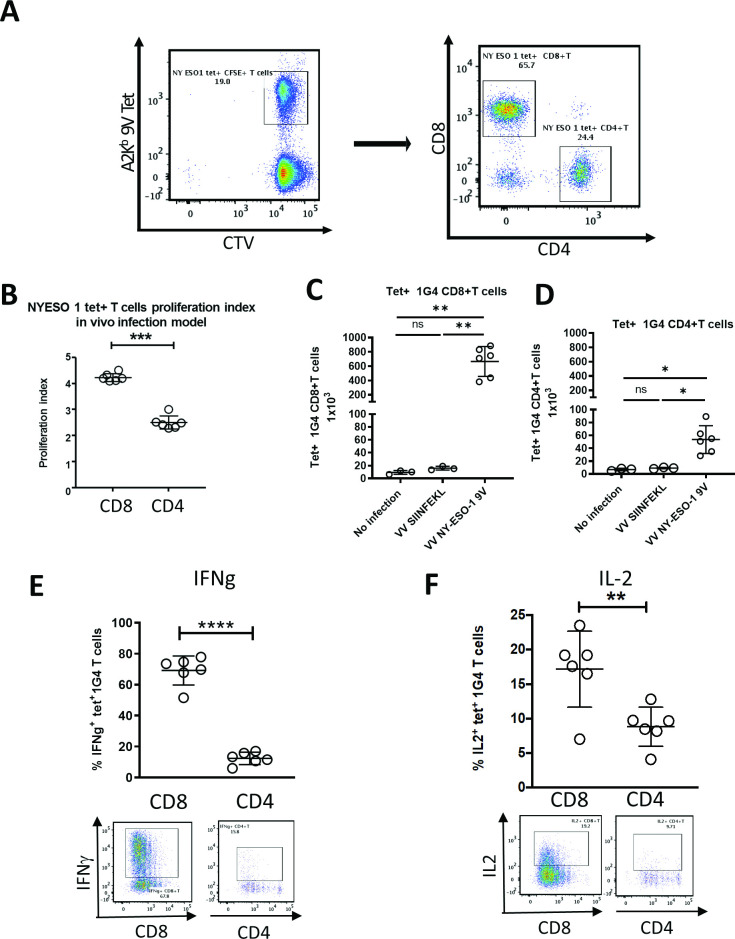
We next compared the rVV-induced activation of CD8 +vs CD4 +m1 G4 cells labeled with cell trace violet adoptively transferred into HHD mice (figure 4A). One day after m1G4 transfer, mice were mock infected or infected with either rVV NY-ESO-1 C165V or rVV SIINFEKL. Three days post infection CD8+ T cells demonstrated increased proliferation in mice infected with rVV NY-ESO-1 C165V compared with CD4+ T cells, while no proliferation was noted in uninfected mice or irrelevantly infected mice with rVV SIINFEKL (figure 4B–D and online supplemental figure S5). Ex vivo splenocyte restimulation with NY-ESO-1157-165V peptide (9V) for 6 hours in the presence of brefeldin-A and monensin demonstrated marked A2-9V tetramer positive m1G4 CD8 +T cell production of IFN-γ (67.8%) and IL-2 (19.2%), but substantially lower production from CD4 +T cells (IFN-γ of 15.8% and IL-2 of 9.71%) and no stimulation with irrelevant rVV SIINFEKL (figure 4E, F and online supplemental figure S5).
Figure 4.
CD4+ and CD8+ T cells from 1G4 mice demonstrate in vivo antigen-specific functionality. m1G4 T cells were isolated by negative selection using Pan T cell isolation Kit (Miltenyi), labeled with CellTrace Violet (CTV) and adoptively transferred into HHD mice. Uninfected mice and mice infected with rVV NY-ESO-1 9V or irrelevant rVV SIINFEKL were then analyzed on day three post infection. (A) Splenic naïve m1G4 T cells before adoptive transfer. (B) CTV labeled splenic 1G4 CD8+ and CD4+ T cell in vivo proliferation index. Absolute number of 1G4 CD8+ (C) and CD4+T cells (D). IFN-γ (E) and IL2 (F) production after ex vivo NY-ESO-1157–165V peptide stimulation. *P<0.05, **p<0.01, ***p<0.001, ****p<0.0001 in Student’s t-test or non-parametric Kruskal-Wallis test with Dunn’s multiple comparisons test with GraphPad software. IFN-γ, interferon-γ; IL2, interleukin 2; ns, not significant.
HHD mice demonstrate NY-ESO-1 specific control of tumor growth in vivo following NY-ESO-1 vaccination
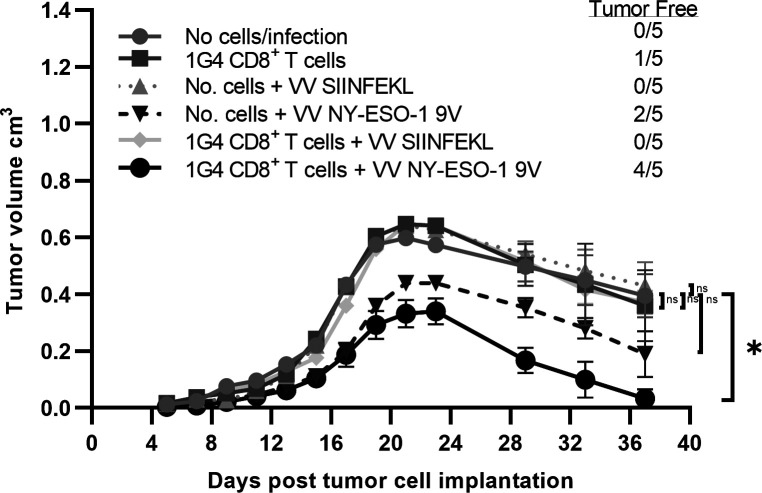
To address the critical issue of whether m1G4 T cells, rather than endogenous CD8 +and CD4+T cells, respond to NY-ESO-1 and can suppress growth of syngeneic tumor cells expressing the full-length human NY-ESO-1 protein, we subcutaneously injected HHD mice with 106 syngeneic tumor cells (MCA induced tumors from HHD mice) expressing NY-ESO-1 (1F4, figure 5). On day 6, some mice received a limited number of 1×105 naïve CD8+ T cells isolated from 1G4 mice by negative selection and subsequently were infected on next day either with no vaccinia, 1×106 pfu of rVV NY-ESO-1 9V or rVV SIINFEKL by intravenous administration to model an NY-ESO-1 directed tumor vaccine. In order to analyze for potentially confounding endogenous T cell response or VV expressed immunogenic antigen directed response, some mice received no m1G4 T cells or tumor cells injected mice were infected with 106 pfu of rVV NY-ESO-1 or rVV SIINFEKL without adoptive transfer of m1G4 CD8+ T cells. Notably, only the HHD mice receiving both m1G4 CD8 +T cells and rVV NY-ESO-1 9V vaccination showed significant tumor clearance with 4/5 mice tumor free on day 37 (p=0.02), while none of the other tumor groups were statistically different from no cells/no infection—indicating that any variation seen was likely due to inherent immunogenicity of the NY-ESO-1-expressing MCA tumor (1F4), which primed the mouse’s endogenous T-cell repertoire against the foreign NY-ESO-1 antigen.
Figure 5.
CD8 +T cells from 1G4 mice control tumor growth in vivo in an antigen specific manner following NY-ESO-1 vaccination. HHD mice (n=5 per group) were subcutaneously injected in the right flank with 1×106 full length NY-ESO-1 cDNA transduced syngeneic tumor cells (1F4 cells) on day 0. On day 6, some mice received 1×105 naïve CD8 +T cells isolated from 1G4 mice by negative selection and subsequently were infected on next day either with 1×106 pfu of rVV NY-ESO-1 9V or rVV SIINFEKL by intravenous administration. In order to analyze for potentially confounding VV induced antitumor response, some tumor cells injected mice were infected with 1×106 pfu of rVV NY-ESO-1 or rVV SIINFEKL without adoptive transfer of m1G4 CD8 +T cells. Error bars represent SEM Tumor growth curves were assessed by two-way analysis of variance (ANOVA) using the Dunnett method for multiple comparison adjustment. *P<0.05 with GraphPad software. ns, not significant.
Direct injection of A2Eso1G4 mice subcutaneously with 106 syngeneic tumor cells of either the parental line (B5) or the human NY-ESO-1 expressing derivative line (1F4) (online supplemental figure S6A) also demonstrated HLA-A2 restricted recognition of NY-ESO-1 (online supplemental figure S6B) with minimal tumor growth in the NY-ESO-1 expressing 1F4 line over 5 weeks compared with the parental B5 line, which grew uncontrolled (online supplemental figure S6C).
Discussion
The use of vaccines to stimulate effective antitumor immune responses in humans has proven to be challenging.40 41 NY-ESO-1 is one of the most attractive broad tumor associated antigen (TAA) targets for human cancer vaccine development, but clinically relevant responses remain elusive.15 42 NY-ESO-1 targeted TCR gene therapy has been more successful with a trial by Robbins et al showing objective clinical responses in of patients with synovial cell sarcoma and 5 of 11 patients with metastatic melanoma.43 To accelerate improvements in NY-ESO-1 vaccination and cellular T-cell therapies we developed a novel TCR transgenic mouse, A2Eso1G4, using an HLA-A2 restricted TCR specific to human NY-ESO-1157-165 SLLMWITQC (9C) initially identified from an NY-ESO-1 positive melanoma patient’s class-I restricted CD8+ TIL.14
The m1G4 TCR was found to be expressed in CD4+ and CD8+ T cells in peripheral blood and lymphoid organs of A2Eso1G4 mice on the RAG+ C57BL/6 HHD genetic background. Immunophenotyping revealed intact presence of all major immune cell populations though differences were observed in numbers of B-cells, macrophages, CD8+ T cells, and CD4+ T cells in lymph node or splenic locations between HHD and 1G4 mice, with differences to be investigated further in RAG competent and knockout settings. Decreased B-cell numbers have been previously observed in mice expressing transgenic TCRs on a RAG competent background, including using the specific cassette vectors used herein, possibly due to apoptosis induced by TCR expression, accumulation, and endoplasmic reticulum stress response.44 Approximately, 20%–35% of CD4+ and 44%–53% of CD8+ T cells in A2Eso1G4 mice express the transgenic TCR and nearly all display the expected naïve phenotype (CD44 negative). This was true even in 33-week-old mice, pointing to the lack of self-antigen expression during this period. Accordingly, no spontaneous autoimmune manifestations were noted, and mice exhibited normal growth and aging. This is notable as a recently described HLA-A2+/human tyrosinase368-376 class I restricted, YMDGTMSQV, high affinity TCR mouse demonstrated spontaneous hair depigmentation and age progressive visual defects due to non-tolerant cross reactivity with the endogenous mouse tyrosinase peptide FMDGTMSQV.25 Other T cells, that are tetramer negative, exist in this RAG competent mice, some of which are CD44 positive cells and consistent with known memory-like T cells arising from presumed physiological homeostatic proliferation.45 The m1G4 T cells were functional, showing proliferation and cytokine secretion after activation with cognate peptide pulsed human T2 cells or mouse syngeneic splenocytes.
A2Eso1G4 m1G4 T cells exhibited NY-ESO-1 specific activation as determined by proliferation, CD69 upregulation, and IL-2 and IFN-γ production. As previously seen in transgenic TCR mice on a RAG competent background we also found absence of allelic exclusion with expression of endogenous TCR and CD4+ T cells expressing the m1G4 TCR,25 37 and demonstrated that CD4+ m1G4 cells can be activated through the TCR as evidenced by CD69 upregulation. However, the resultant cell is less fit compared with a CD8+ m1G4 cell in terms of IFN-γ and IL2 production, demonstrating a role for the CD8 coreceptor in m1G4 activation. Back-crossing of the A2Eso1G4 mice onto a RAG-/-background will be necessary to assess the efficiency of positive selection of the 1G4 TCR.
Interestingly, our adoptive transfer experiments indicate that antigen specific CD4+ T cell presence may improve the response of CD8+ T cells as evidence by improved cytokine production when m1G4 CD4+ T cells were cotransferred with m1G4 CD8+ T cells in our vaccinia infection model (comparison of CD8 functional status in figures 3 and 4). This is consistent with prior data showing transfer of LCMV-specific CD4+ T cells to mice with CD8+ T cell exhaustion from chronic infection improved CD8+ T cell proliferation and function,46 47 and is the focus of an ongoing phase I trial (NCT03691376) evaluating whether NY-ESO-1 directed CD8 TCR responses can benefit from concomitant CD4 TCR mediated help in clearance of chemo resistant ovarian cancer. The A2Eso1G4 mouse model thereby provides the opportunity to study the role of CD4 help in CD8 antitumor responses and TCR signaling in CD4 and CD8 T cells on activation with the same antigen.
Adoptive transfer of m1G4 CD8+ T cells into HHD mice followed by NY-ESO-1 vaccination demonstrated significant tumor clearance despite transfer of suboptimal cell doses (1×105 cells), and future work can build on this by modeling various amounts of CD4 T cell help and varying the NY-ESO-1-TCR affinity dynamics using our described altered peptide libraries.27
A limitation of the m1G4 mouse model presented herein is the current RAG competent background that limits assessment of the efficiency of positive selection of the 1G4 TCR. We intentionally adoptively transferred suboptimal quantities of m1G4 CD8 +T cells to model a cancer vaccine system, and future experiments are required to properly modulate the balance between adoptively transferred T cell quantities, tumor size, and vaccination dose. Finally, to explore the unique properties of this mouse model a range of tumor lines will be required that vary in aggressiveness of growth, and levels of antigen expression and presentation.
In summary, we believe the novel mouse model reported in this study presents a useful TCR transgenic mouse model of tumor reactive T cells that target a clinically relevant mammalian TAA. Absence of murine NY-ESO-1 expression, prevents thymic selection against the TCR, opens the door to future higher affinity TCR mouse variants, and allows our model to serve as a viral or TAA platform to investigate basic T cell antiviral or antitumor responses, and design future tumor immunotherapy protocols, focused on T cell affinity/avidity for tumor vaccine and cellular T-cell therapy development.
Acknowledgments
This paper is dedicated to the loving memory of Vincenzo (Enzo) Cerundolo, a wonderful friend, colleague and inspirational mentor, who will be sorely missed.
Footnotes
Twitter: @JonSilk2
ES and MK contributed equally.
Contributors: ES designed the transgenic mice and reagents, performed experiments, analyzed data, and wrote the manuscript. MK and UG performed experiments, analysed data, and wrote the manuscript. JC, DS, JG, GP, and JDS assisted with reagent preparation and experimental design. JWY and VC designed and supervised the study, secured funding, analyzed data, and edited the manuscript. All authors discussed the results, commented on the manuscript, and agreed on publication.
Funding: ES was supported by the Rhodes Trust, Oxford, UK and the Division of Intramural Research, NIAID, Bethesda, Maryland, USA. VC and UG are supported by MRC and MK JDS and VC are supported by the CRUK. JB and JWY are supported by the Division of Intramural Research, NIAID, Bethesda, Maryland, USA.
Disclaimer: The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available on reasonable request.
Ethics statements
Patient consent for publication
Not required.
Ethics approval
All mouse experiments were performed under approved University of Oxford Institutional Animal care and committee protocols.
References
- 1.Pardoll DM. The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 2012;12:252–64. 10.1038/nrc3239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rosenberg SA, Yang JC, Sherry RM, et al. Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin Cancer Res 2011;17:4550–7. 10.1158/1078-0432.CCR-11-0116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kantoff PW, Higano CS, Shore ND, et al. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med 2010;363:411–22. 10.1056/NEJMoa1001294 [DOI] [PubMed] [Google Scholar]
- 4.Shields BD, Mahmoud F, Taylor EM, et al. Indicators of responsiveness to immune checkpoint inhibitors. Sci Rep 2017;7:807. 10.1038/s41598-017-01000-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tran E, Turcotte S, Gros A, et al. Cancer immunotherapy based on mutation-specific CD4+ T cells in a patient with epithelial cancer. Science 2014;344:641–5. 10.1126/science.1251102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tran E, Robbins PF, Lu Y-C, et al. T-Cell transfer therapy targeting mutant KRAS in cancer. N Engl J Med 2016;375:2255–62. 10.1056/NEJMoa1609279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zeh HJ, Perry-Lalley D, Dudley ME, et al. High avidity CTLs for two self-antigens demonstrate superior in vitro and in vivo antitumor efficacy. J Immunol 1999;162:989–94. [PubMed] [Google Scholar]
- 8.Alexander-Miller MA, Leggatt GR, Berzofsky JA. Selective expansion of high- or low-avidity cytotoxic T lymphocytes and efficacy for adoptive immunotherapy. Proc Natl Acad Sci U S A 1996;93:4102–7. 10.1073/pnas.93.9.4102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Davis MM, Boniface JJ, Reich Z, et al. Ligand recognition by alpha beta T cell receptors. Annu Rev Immunol 1998;16:523–44. 10.1146/annurev.immunol.16.1.523 [DOI] [PubMed] [Google Scholar]
- 10.van der Merwe PA, Davis SJ. Molecular interactions mediating T cell antigen recognition. Annu Rev Immunol 2003;21:659–84. 10.1146/annurev.immunol.21.120601.141036 [DOI] [PubMed] [Google Scholar]
- 11.Zehn D, Lee SY, Bevan MJ. Complete but curtailed T-cell response to very low-affinity antigen. Nature 2009;458:211–4. 10.1038/nature07657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhao Y, Bennett AD, Zheng Z, et al. High-Affinity TCRs generated by phage display provide CD4+ T cells with the ability to recognize and kill tumor cell lines. J Immunol 2007;179:5845–54. 10.4049/jimmunol.179.9.5845 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Aleksic M, Dushek O, Zhang H, et al. Dependence of T cell antigen recognition on T cell receptor-peptide MHC confinement time. Immunity 2010;32:163–74. 10.1016/j.immuni.2009.11.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen J-L, Stewart-Jones G, Bossi G, et al. Structural and kinetic basis for heightened immunogenicity of T cell vaccines. J Exp Med 2005;201:1243–55. 10.1084/jem.20042323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Old LJ. Cancer vaccines: an overview. Cancer Immun 2008;8(Suppl 1):1. [PubMed] [Google Scholar]
- 16.Gnjatic S, Nishikawa H, Jungbluth AA, et al. NY-ESO-1: review of an immunogenic tumor antigen. Adv Cancer Res 2006;95:1–30. 10.1016/S0065-230X(06)95001-5 [DOI] [PubMed] [Google Scholar]
- 17.Jungbluth AA, Chen YT, Stockert E, et al. Immunohistochemical analysis of NY-ESO-1 antigen expression in normal and malignant human tissues. Int J Cancer 2001;92:856–60. 10.1002/ijc.1282 [DOI] [PubMed] [Google Scholar]
- 18.Jungbluth AA, Antonescu CR, Busam KJ, et al. Monophasic and biphasic synovial sarcomas abundantly express cancer/testis antigen NY-ESO-1 but not MAGE-A1 or CT7. Int J Cancer 2001;94:252–6. 10.1002/ijc.1451 [DOI] [PubMed] [Google Scholar]
- 19.Juretic A, Spagnoli GC, Schultz-Thater E, et al. Cancer/testis tumour-associated antigens: immunohistochemical detection with monoclonal antibodies. Lancet Oncol 2003;4:104–9. 10.1016/S1470-2045(03)00982-3 [DOI] [PubMed] [Google Scholar]
- 20.Rapoport AP, Stadtmauer EA, Binder-Scholl GK, et al. NY-ESO-1-specific TCR-engineered T cells mediate sustained antigen-specific antitumor effects in myeloma. Nat Med 2015;21:914–21. 10.1038/nm.3910 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.D'Angelo SP, Melchiori L, Merchant MS, et al. Antitumor Activity Associated with Prolonged Persistence of Adoptively Transferred NY-ESO-1 c259T Cells in Synovial Sarcoma. Cancer Discov 2018;8:944–57. 10.1158/2159-8290.CD-17-1417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stadtmauer EA, Faitg TH, Lowther DE, et al. Long-term safety and activity of NY-ESO-1 spear T cells after autologous stem cell transplant for myeloma. Blood Adv 2019;3:2022–34. 10.1182/bloodadvances.2019000194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ramachandran I, Lowther DE, Dryer-Minnerly R, et al. Systemic and local immunity following adoptive transfer of NY-ESO-1 spear T cells in synovial sarcoma. J Immunother Cancer 2019;7:276. 10.1186/s40425-019-0762-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yu Z, Theoret MR, Touloukian CE, et al. Poor immunogenicity of a self/tumor antigen derives from peptide-MHC-I instability and is independent of tolerance. J Clin Invest 2004;114:551–9. 10.1172/JCI200421695 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mehrotra S, Al-Khami AA, Klarquist J, et al. A coreceptor-independent transgenic human TCR mediates anti-tumor and anti-self immunity in mice. J Immunol 2012;189:1627–38. 10.4049/jimmunol.1103271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ishizuka J, Grebe K, Shenderov E, et al. Quantitating T cell cross-reactivity for unrelated peptide antigens. J Immunol 2009;183:4337–45. 10.4049/jimmunol.0901607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chen JL, Dunbar PR, Gileadi U, et al. Identification of NY-ESO-1 peptide analogues capable of improved stimulation of tumor-reactive CTL. J Immunol 2000;165:948–55. 10.4049/jimmunol.165.2.948 [DOI] [PubMed] [Google Scholar]
- 28.Jäger E, Chen YT, Drijfhout JW, et al. Simultaneous humoral and cellular immune response against cancer-testis antigen NY-ESO-1: definition of human histocompatibility leukocyte antigen (HLA)-A2-binding peptide epitopes. J Exp Med 1998;187:265–70. 10.1084/jem.187.2.265 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Kouskoff V, Signorelli K, Benoist C, et al. Cassette vectors directing expression of T cell receptor genes in transgenic mice. J Immunol Methods 1995;180:273–80. 10.1016/0022-1759(95)00002-R [DOI] [PubMed] [Google Scholar]
- 30.Pascolo S, Bervas N, Ure JM, et al. HLA-A2.1-restricted education and cytolytic activity of CD8(+) T lymphocytes from beta2 microglobulin (beta2m) HLA-A2.1 monochain transgenic H-2Db beta2m double knockout mice. J Exp Med 1997;185:2043–51. 10.1084/jem.185.12.2043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shagin DA, Lukyanov KA, Vagner LL, et al. Regulation of average length of complex PCR product. Nucleic Acids Res 1999;27:i–iii. 10.1093/nar/27.18.e23-i [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Svartman M, Stone G, Stanyon R. Molecular cytogenetics discards polyploidy in mammals. Genomics 2005;85:425–30. 10.1016/j.ygeno.2004.12.004 [DOI] [PubMed] [Google Scholar]
- 33.Choi EM-L, Chen J-L, Wooldridge L, et al. High avidity antigen-specific CTL identified by CD8-independent tetramer staining. J Immunol 2003;171:5116–23. 10.4049/jimmunol.171.10.5116 [DOI] [PubMed] [Google Scholar]
- 34.Chakrabarti S, Brechling K, Moss B. Vaccinia virus expression vector: coexpression of beta-galactosidase provides visual screening of recombinant virus plaques. Mol Cell Biol 1985;5:3403–9. 10.1128/MCB.5.12.3403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Earl PL, Moss B, Wyatt LS. Generation of recombinant vaccinia viruses. Current Protocols in Molecular Biology 2001. [DOI] [PubMed] [Google Scholar]
- 36.Fruci D, Lauvau G, Saveanu L, et al. Quantifying recruitment of cytosolic peptides for HLA class I presentation: impact of TAP transport. J Immunol 2003;170:2977–84. 10.4049/jimmunol.170.6.2977 [DOI] [PubMed] [Google Scholar]
- 37.Madsen LS, Andersson EC, Jansson L, et al. A humanized model for multiple sclerosis using HLA-DR2 and a human T-cell receptor. Nat Genet 1999;23:343–7. 10.1038/15525 [DOI] [PubMed] [Google Scholar]
- 38.Johnson ES, Ma PC, Ota IM, et al. A proteolytic pathway that recognizes ubiquitin as a degradation signal. J Biol Chem 1995;270:17442–56. 10.1074/jbc.270.29.17442 [DOI] [PubMed] [Google Scholar]
- 39.Qian S-B, Ott DE, Schubert U, et al. Fusion proteins with COOH-terminal ubiquitin are stable and maintain dual functionality in vivo. J Biol Chem 2002;277:38818–26. 10.1074/jbc.M205547200 [DOI] [PubMed] [Google Scholar]
- 40.Hu Z, Ott PA, Wu CJ. Towards personalized, tumour-specific, therapeutic vaccines for cancer. Nat Rev Immunol 2018;18:168–82. 10.1038/nri.2017.131 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shenderov E, Antonarakis ES. Reimagining vaccines for prostate cancer: back to the future. Clin Cancer Res 2020;26:5056–8. 10.1158/1078-0432.CCR-20-2257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen J-L, Dawoodji A, Tarlton A, et al. Ny-Eso-1 specific antibody and cellular responses in melanoma patients primed with NY-ESO-1 protein in ISCOMATRIX and boosted with recombinant NY-ESO-1 fowlpox virus. Int J Cancer 2015;136:E590–601. 10.1002/ijc.29118 [DOI] [PubMed] [Google Scholar]
- 43.Robbins PF, Morgan RA, Feldman SA, et al. Tumor regression in patients with metastatic synovial cell sarcoma and melanoma using genetically engineered lymphocytes reactive with NY-ESO-1. J Clin Oncol 2011;29:917–24. 10.1200/JCO.2010.32.2537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lobito AA, Lopes MF, Lenardo MJ. Ectopic T cell receptor expression causes B cell immunodeficiency in transgenic mice. Eur J Immunol 2004;34:890–8. 10.1002/eji.200324675 [DOI] [PubMed] [Google Scholar]
- 45.Haluszczak C, Akue AD, Hamilton SE, et al. The antigen-specific CD8+ T cell repertoire in unimmunized mice includes memory phenotype cells bearing markers of homeostatic expansion. J Exp Med 2009;206:435–48. 10.1084/jem.20081829 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Matloubian M, Concepcion RJ, Ahmed R. CD4+ T cells are required to sustain CD8+ cytotoxic T-cell responses during chronic viral infection. J Virol 1994;68:8056–63. 10.1128/JVI.68.12.8056-8063.1994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Aubert RD, Kamphorst AO, Sarkar S, et al. Antigen-specific CD4 T-cell help rescues exhausted CD8 T cells during chronic viral infection. Proc Natl Acad Sci U S A 2011;108:21182–7. 10.1073/pnas.1118450109 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2021-002544supp001.pdf (4.6MB, pdf)
Data Availability Statement
Data are available on reasonable request.