Abstract
Emergent scientific evidence indicates the central role of cancer-associated fibroblasts in determining whether the microenvironment of cancer works as friend or foe of the host; however, there is no unified histological evaluation framework of fibrotic stroma in colorectal cancers. Myxoid stroma and keloid-like collagen are site-specific histopathological features generated by cancer-associated fibroblasts, which appear exclusively in the tumor front during desmoplastic reaction. On the basis of these two stromal components, desmoplastic reaction is categorized into three patterns—immature, intermediate and mature—using hematoxylin and eosin staining. In January 2020, a prospective randomized clinical trial, JCOG1805, to elucidate the value of adjuvant chemotherapy in stage II colorectal cancer patients with pathological risk factors of recurrence was launched in Japan, in which intermediate/immature desmoplastic reaction is one of the four risk factors selected as inclusion criteria. This paper covers the diagnostic criteria for the desmoplastic reaction classification being used in the JCOG1805 study.
Keywords: cancer stroma, cancer-associated fibroblasts, extracellular matrix, myxoid stroma, keloid-like hyalinized collagen
The present article illustrates the diagnostic criteria of desmoplastic reaction classification, a novel prognostic factor in colorectal cancer, used in the JCOG1805 trial conducted by the Japan Clinical Oncology Group.
Introduction
A number of molecular biology studies have clarified the central role of stromal environment in cancer development, and cancer-associated fibroblasts (CAFs) have recently been regarded as key players of modulating cancer microenvironment (1). Biological behavior of at least part of CAFs is closely associated with epithelial–mesenchymal transition (EMT) and cancer stemness via modulating growth factor signaling and extracellular matrix remodeling, thereby promoting acquisition of proinvasion/metastasis phenotype and chemoresistance of cancer cells (2).
Figure 1.
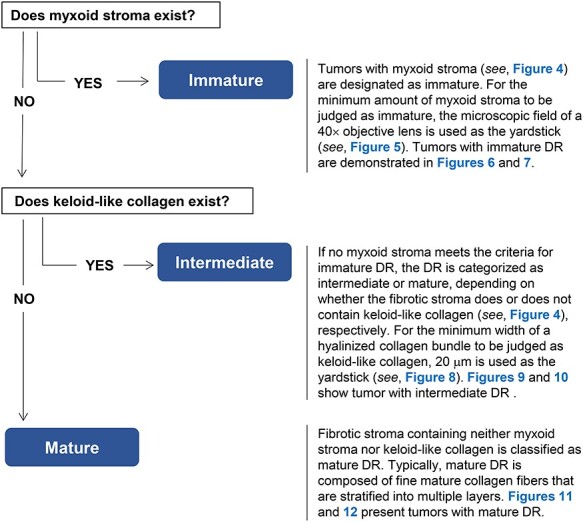
Diagnostic criteria for the desmoplastic reaction (DR) classification.
Being different from advances in the field of cancer biological studies, where genes closely associated with poor-prognosis subtypes are shown to be expressed in the stroma rather than in cancer cells (3,4), tumor factors are still major determinants in clinical practice for colorectal cancer (CRC). A major example would be judging the indication of adjuvant chemotherapy in stage II CRCs, wherein patients are considered at high-risk of recurrence if they present at least one of the following clinical characteristics: lymph-node sampling <12; poorly differentiated tumor, vascular, lymphatic or perineural invasion; tumor presentation with obstruction or tumor perforation; or pT4 staging in international guidelines such as the National Comprehensive Cancer Network (NCCN) guidelines(5) and European Society for Medical Oncology (ESMO) guidelines (6). No stromal factors are included in the decision-making process of the treatment. The lack of appropriate assessment criteria for cancer stroma may be a major reason for expanding the gap in the approach to understand the nature of individual tumors between the bench and the clinic.
In recent years, an increasing number of studies have highlighted the higher prognostic value of the histological desmoplastic reaction (DR) classification over conventional risk factors in CRCs, including two multicenter retrospective studies analyzing stage II CRC populations in Japan (7) and the UK (8). The prognostic value of the DR characterization was also assessed in the prospective randomized SACURA (surgical adjuvant chemotherapy with UFT for curatively resected stage II colon cancer) trial (9). As a result, the DR classification was demonstrated to play an essential role in a prognostic model predicting relapse-free survival (RFS), and this factor was identified as the best single discriminator of RFS events, followed by T-stage, microsatellite instability status and budding (10).
In January 2020, a prospective phase III trial—JCOG1805 [randomized controlled study of adjuvant chemotherapy for stage II CRC patients at high-risk of developing recurrence according to T-stage and three selected pathological factors (Pn, DR and BD)]—was initiated by the Japanese Clinical Oncology Group. In the process of developing a study protocol, the evidence levels for prognostic values of candidate risk factors, including those adopted in the NCCN and ESMO guidelines, had been reviewed; consequently, the DR classification was selected, together with T-stage, perineural invasion, tumor budding, as a prognostic factor to be used as an inclusion criterion (Supplemental Fig. 1).
Myxoid stroma and keloid-like, hyalinized collagen bundles are two criteria for judging the DR pattern (11). These are site-specific, histopathological features produced by CAFs and exclusively appearing at the front of the tumor. Their association with a dedifferentiated tumor phenotype, reduced distribution of immune cells, extracellular matrix changes and high incidence of metastasis (12,13) suggests that underlying molecular mechanisms generating these features are essential for cancer development, such as EMT (14,15). This article covers the diagnostic criteria of the DR classification described in the diagnostic manual prepared for quality control in the JCOG1805. We believe that the DR classification greatly contributes to validating the link between cells’ acquired functions and tissues’ structural changes and further provides new insights into a better histological understanding of cancer microenvironment.
Evaluation criteria for the DR classification
Myxoid stroma and keloid-like collagen are two components of the DR classification criteria, by which DR is histologically categorized into three patterns—immature, intermediate and mature. A summary of the diagnostic criteria is shown in Fig. 1.
Figure 2.
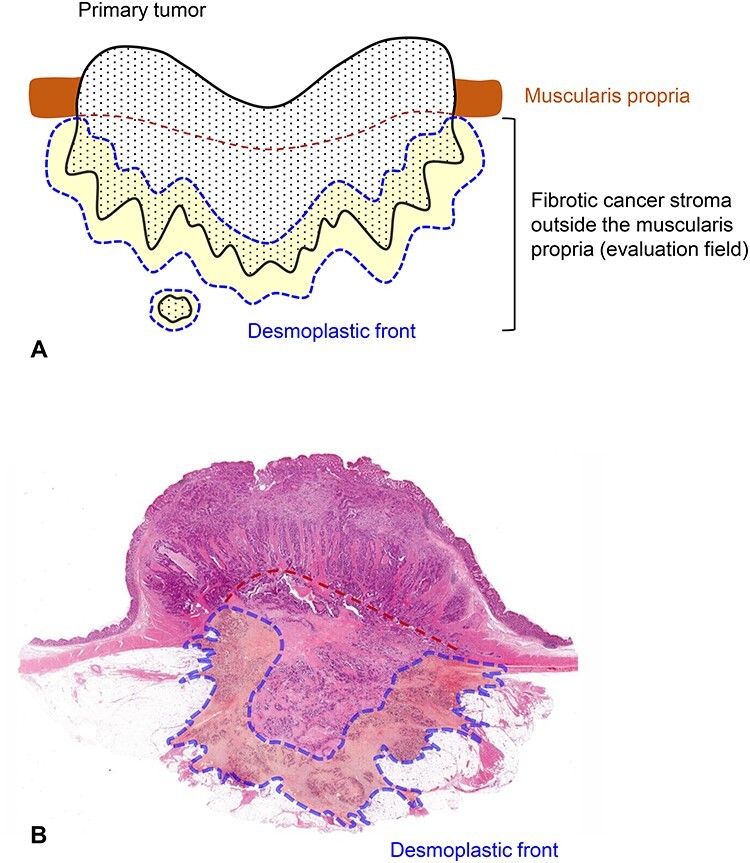
Field in the primary tumor to be evaluated for the DR classification. Fibrotic cancer stroma outside the muscularis propria is evaluated on pathological slides prepared in routine practice. Cancer stroma in the submucosa or those in the muscularis propria are excluded from the evaluation. Fibrotic stroma along the leading edge of the primary tumors (desmoplastic front) is the area subject to intensive evaluation because both myxoid stroma and keloid-like hyalinized collagen mostly appear in this lesion. When a tumor nodule exists in the fatty tissue attached to the primary tumor, the fibrotic stroma of the lesion should be evaluated entirely for the DR classification. (A) Schema and (B) a whole slide image [Hematoxylin–eosin (H&E) stained] of the primary tumor. Area circled with blue dotted line, desmoplastic front; brown dotted line, estimated lower border of the muscularis propria.
Site of cancer stroma to be evaluated
Hematoxylin–eosin (H&E) glass slides of the primary tumor prepared in routine pathology practice are used to determine the DR category. Fibrotic cancer stroma outside the muscularis propria layer is the area to be evaluated (Fig. 2). Findings in the submucosa and muscularis propria are not considered in classifying the DR pattern. The fibrotic stroma along the leading edge of the primary tumor (desmoplastic front), where myxoid stroma and keloid-like collagen most often appear, is the highly important field to be evaluated. When tumor nodules exist in the fatty tissue attached to the primary tumor, the fibrotic stroma of the nodule must be examined in its entirety. Areas of stroma with localized inflammation suspected of being caused by external pathogens, such as that around microscopic abscesses by intratumoral perforation, are not taken into consideration (Fig. 3).
Figure 3.
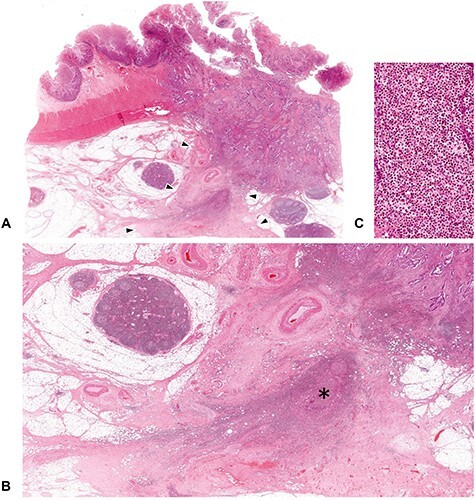
Fibrotic area to be excluded from the DR classification evaluation. The DR pattern is evaluated at fibrotic stroma exclusively induced by tumor–stroma interaction. Acute inflammation such as microscopic abscess with intratumoral perforation can evoke a dense fibrosis, but this area should be distinguished from the desmoplastic reaction to be assessed for the DR classification. In figure (A), dense fibrotic tissue exists beyond the desmoplastic front (area surrounded by the triangle symbols), in which a remnant of microscopic abscess (shown with an asterisk) is observed, thereby indicates this fibrosis was induced by the external pathogens, rather than tumor–stroma interaction. (B; ×1.5 objective) is photograph magnifying the part of the extramural area indicated by the triangles in (A; whole slide image) and (C; ×40 objective) is that magnifying that part of asterisk. All, H&E staining.
Figure 4.
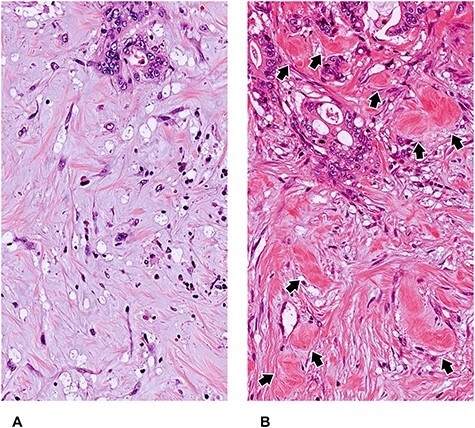
Myxoid stroma and keloid-like hyalinized collagen bundles as two elemental components to categorize DR pattern. The DR pattern is categorized as immature, intermediate, or mature, based on two site-specific histological products of fibroblasts—myxoid stroma and hyalinized keloid-like collagens—that appear exclusively at the desmoplastic front. (A) Myxoid stroma: a fibrotic stroma with an amorphous mucinous stromal substance composed of a basophilic or amphophilic extracellular material. (B) Keloid-like collagen bundles are thick, hyalinized bundles of hypocellular collagen with bright eosinophilic hyalinization (arrow) (A, B: Original magnification, ×40 objective; H&E staining).
Myxoid stroma
Myxoid stroma is defined as stroma accompanied by an amorphous mucinous substance, which is typically a basophilic or amphophilic, vacuolated extracellular material among the collagen fibers (Fig. 4). The proportion of stromal mucin to cancer stroma varies depending on the individual tumor. For the minimum amount of myxoid stroma to be judged as immature, the microscopic field of a ×40 objective lens is used as a yardstick (Fig. 5). Generally, the myxoid stroma exists in relatively limited areas at the desmoplastic front, even in cases with an immature DR in the CRC (Fig. 6). This is different from the situation in pancreatic cancer, a notoriously aggressive desmoplastic tumor with a dismal prognosis, in which myxoid stroma is more commonly and extensively observed. It is not difficult to distinguish stromal mucin as a component of myxoid stroma from stromal mucin leakage associated with mucinous cancer, since the former is accompanied by other DR components including fibroblasts, collagen fibers, inflammatory cells and endothelial cells in stromal mucin (Fig. 7).
Figure 5.
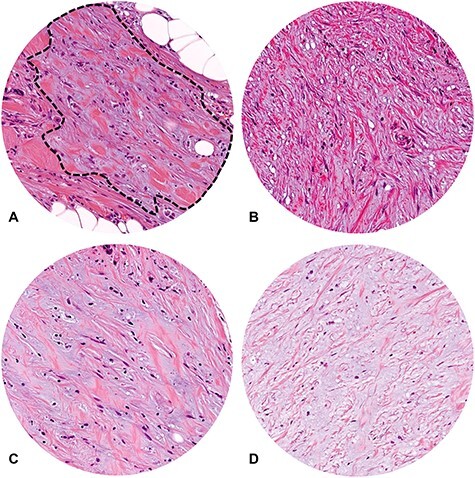
Extent of myxoid stroma area to be judged as in the immature DR category. For the minimum amount of myxoid stroma to be judged as immature, the microscopic field of a ×40 objective lens is used as a yardstick. (A) Although there is an area with stromal mucin, as circled with a black dotted line, it does not completely fill the field of a ×40 objective lens, thereby does not meet the criteria to place it in the immature category; (B–D) Increased stromal mucin forms the area of myxoid stroma, which fully occupies the field of a ×40 objective lens with various proportions of stromal mucin-to-collagen fibers. These DR lesions are classified in the immature category. (A–D) Microscopic fields of a ×40 objective lens (field number, 20; H&E staining).
Figure 6.
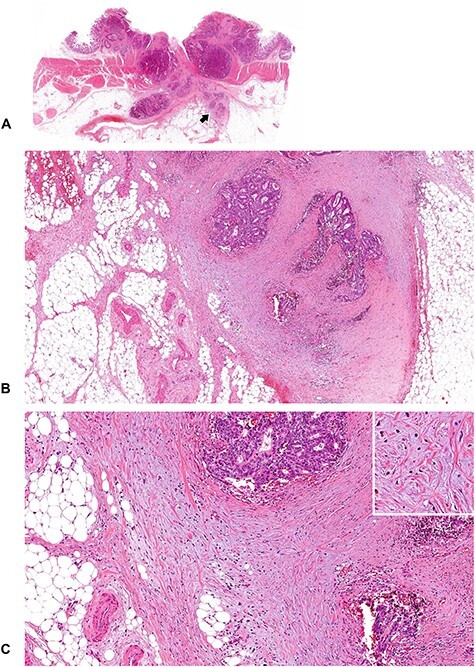
A tumor with immature DR (1). An amorphous and vacuolated, basophilic, myxoid stroma is observed in some areas within the desmoplastic front. Note that there is an intratumoral heterogeneity in the DR pattern, and in the case of colorectal cancer, myxoid stroma generally occupies a limited area of the desmoplastic front. (B; ×4 objective) and (C; ×10 objective) are photographs magnifying that part of the desmoplastic front indicated by the arrow in (A; whole slide image). Insert in (C), ×20 objective. All, H&E staining.
Figure 7.
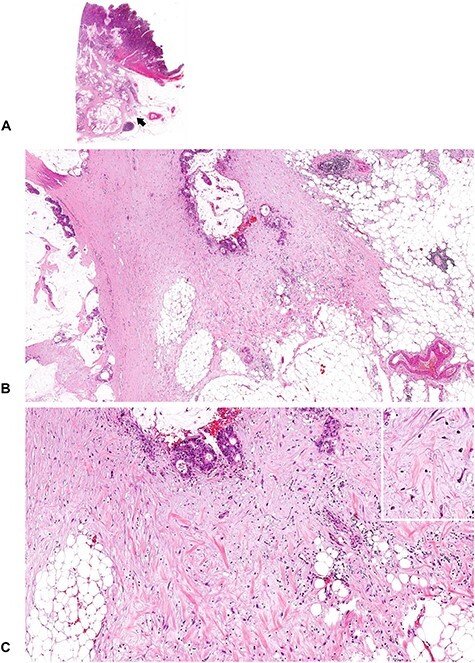
A tumor with immature DR (2). An amorphous, vacuolated, amphophilic myxoid material extends to the desmoplastic front. Note that although the tumor cells produce abundant extracellular mucin, myxoid stroma can be differentiated from the mucin pool in the stroma. The mucin leakage is devoid of desmoplastic reaction, whereas the myxoid stroma is intermingled with other desmoplastic components such as fibroblasts, collagen fibers, inflammatory cells and endothelial cells. (B; ×4 objective) and (C; ×10 objective) are magnified photographs of the portion of the desmoplastic front indicated by the arrow in (A; whole slide image). Insert in (C), ×20 objective. All, H&E staining.
Figure 8.
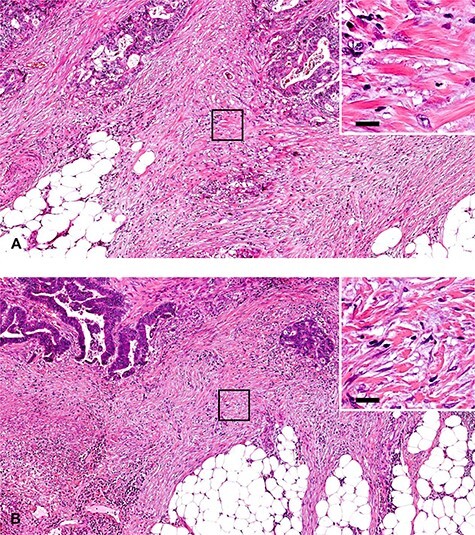
Size of hyalinized collagen bundles to be judged as keloid-like collagen, the criterion of the intermediate DR category. Keloid-like collagen is a thick, hyalinized, collagen bundle, and is recognized at low or middle magnification. A yardstick of 20 μm can be used for the minimum width of the bundle to be judged as keloid-like collagen. In cases (A) and (B), fragmentary collagen fibers are observed at the desmoplastic front, but they are not judged as keloid-like collagen because the thickness of the fibers is <20 μm. These DR lesions are classified as the mature category. (A, B): ×10 objective (H&E staining). Upper right photographs are magnified images of the area boxed with black lines in photos (A) and (B). Bars indicate 20 μm.
Keloid-like collagen
Keloid-like collagen is a thick bundle of hypocellular collagen with bright eosinophilic hyalinization (Fig. 4). To differentiate it from fragmentary collagen fiber, a 20-μm width can be used as a yardstick for the minimum thickness of hyalinized collagen (Fig. 8). Although keloid-like collagen appears exclusively at the desmoplastic front, not in the superficial portion or center of the tumor, there is heterogeneity in terms of its anatomical distribution at the desmoplastic front. Some keloid-like collagen is distributed near tumor nests (Fig. 9), and other keloid-like collagen is found at the most desmoplastic front, which is slightly distant from the tumor nests (Fig. 10).
Figure 9.
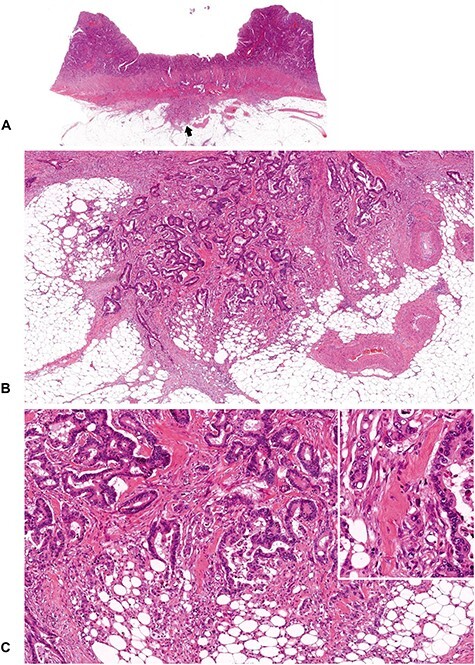
A tumor with intermediate DR (1). Keloid-like, hyalinized collagen bundles are observed at the desmoplastic front. Note that some keloid-like collagen is located adjacent to the cancer nests, and some is located distant from the cancer nests (Figure 10), depending on the individual tumors. In this case, keloid-like collagen is distributed close to the cancer nests as if coiled around them. (B; ×4 objective) and (C; ×10 objective) are magnifications of the desmoplastic front, as indicated by the arrow in (A) (whole slide image). Insert in (C), ×20 objective. All, H&E staining.
Figure 10.
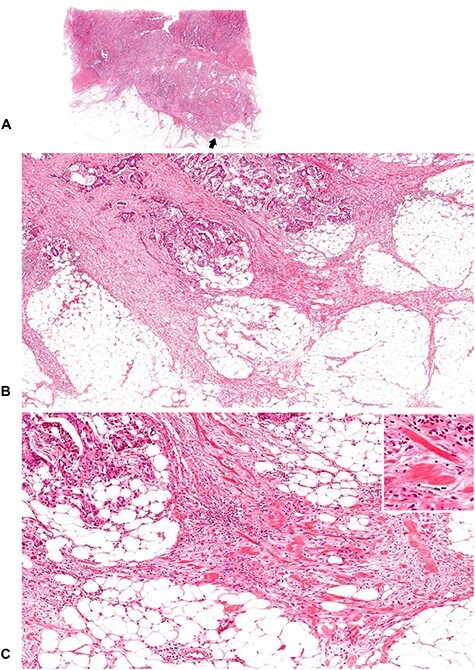
A tumor with intermediate DR (2). Keloid-like collagen bundles appear at the leading edge of the desmoplastic stroma. (B; ×4 objective) and (C; ×10 objective) are magnifications of the area of the desmoplastic front as indicated by the arrow in (A) (whole slide image). Insert in (C), ×20 objective. All, H&E staining.
Figure 11.
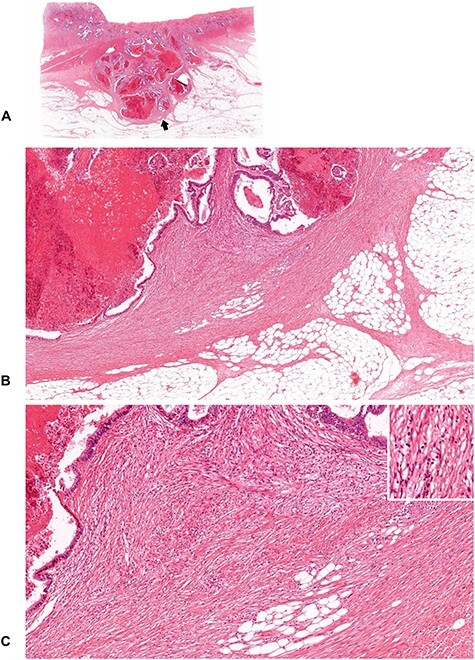
A tumor with mature DR (1). Cancer nests are encompassed by fine, mature collagen fibers, which are developing in a multilayer form. No myxoid stroma or keloid-like collagen bundles are seen. (B; ×4 objective) and (C; ×10 objective) are magnifications of the area of the desmoplastic front as indicated by the arrow in (A; whole slide image). Insert in (C), ×20 objective. All, H&E staining.
Diagnostic flow of DR category
The flowchart of categorizing DR is shown in Fig. 1. Extramural stroma of the primary tumor is first scanned at low or middle-power magnification to search for myxoid stroma and keloid-like collagen. The category of ‘immature’ is designated for the pattern of DR accompanying myxoid stroma. In cases without myxoid stroma that satisfy the criteria for the immature DR category, the DR pattern is categorized as ‘intermediate’ if the fibrotic stroma contains keloid-like collagen. Only one focus with such a collagen bundle is enough for the classification as intermediate. Note that DR pattern of the coexistence of the myxoid stroma which fully occupies the field of a ×40 objective lens and keloid-like collagen is classified as immature category. DR is classified as ‘mature’ if there is no myxoid stroma or keloid-like collagen in the extramural stroma. Mature DR is typically characterized by fine, multilayered, mature collagen fibers, which may develop to encompass tumor nests or develop radially to form the most desmoplastic front (Fig. 11). Immune-cell infiltration status does not affect the DR categorization. Figure 12 exhibits the finding of a desmoplastic front of a tumor having conspicuous immune cells, in which no myxoid stroma or keloid-like collagen existed, thereby being diagnosed as mature DR.
Figure 12.
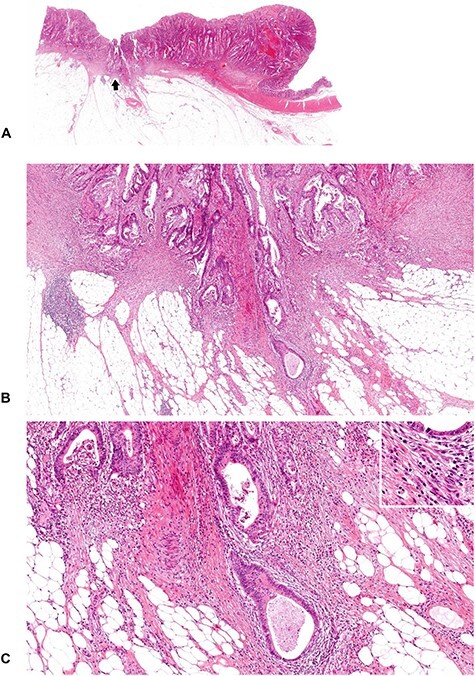
A tumor with mature DR (2). Desmoplastic stroma with relatively abundant lymphocyte infiltration is developing at the leading edge of the tumor. Neither myxoid stroma nor keloid-like collagen bundles are observed, and the DR pattern is classified as the mature category. (B; ×4 objective) and (C; ×10 objective) are magnifications of the area of the desmoplastic front as indicated by the arrow in (A; whole slide image). Insert in (C), ×20 objective. All, H&E staining.
Discussion
The roles of cancer stroma are complex and multifaceted and cannot be defined simply as helpful or harmful to tumors (16,17). In general, tumor stroma is thought to act in a direction of supporting and promoting the growth of cancer, but it is reported that some stromal components can act to restrain tumor growth (18,19). The potential of tumor growth and metastasis may be determined by how tumor cells orchestrate stromal cells for their benefit through various preexisting molecular mechanisms for tissue remodeling. In this regard, although there have been some attempts to categorize cancer stroma on the basis of its amount (20,21) or the percentage of stroma in a tumor (22,23), it would be difficult to distinguish between ‘prohost DR’ or ‘protumor DR’ with these morphometric approaches.
Myxoid stroma and keloid-like hyalinized collagen bundles are histological features of fibrotic stroma, supposedly generated by specific subgroups of activated fibroblasts, given that these are not universally observed in the field of DR. These are not malignant neoplasm-specific, and they can be generated in other contexts, for example, inflammatory responses to infections, pathogens or soft tissue tumors. As for the terminology for the categories in DR characterization, ‘mature’ was originally used for the highly fibrogenic pattern of DR that was being organized regularly with fine, uniform and multilayered collagen fibers, whereas ‘immature’ was for the DR pattern with poor fibrogenicity with mucopolysaccharide that comprised the major portion of extracellular matrix (11). In the DR categorization system, the ‘intermediate’ category was provided for the DR pattern which was fibrogenic (similar to ‘mature’ pattern), but disorderly in terms of the morphology of collagen fibers; i.e. state of existence of hyalinized, thickened, ‘keloid-like’ collagens which were deviated from collagen fibers that form the ‘mature’ DR pattern and observed in most tumors with immature DR. In the preceding studies, the frequency distributions of tumors that were classified as mature, intermediate and immature DR patterns were 63–64%, 27–30% and 6–10%, respectively, in stage II CRC (7,10).
An increasing number of pathological studies show that the DR classification in the CRC (7,8,10,24–28) and that in the colorectal metastatic lesions (29,30) have greatly influenced on prognostic outcomes. Similar prognostic impact of the DR classification is also observed in other malignancies, including pancreatic ductal adenocarcinoma (31), cutaneous squamous cell carcinoma [SCC; (32)] and cervical SCC (33). Notably, it has been consistently shown that mature DR is a characteristic of tumors behaving favorably, whereas non-mature DR (especially immature DR) is a characteristic of aggressive tumors. These results indicate that myxoid stroma and keloid-like collagen are histological biomarkers of a desmoplastic microenvironment, indicating that cancer cells have successfully used molecular mechanisms of the stromal remodeling process for their development.
Lack of a robust prognosticator is a major source of uncertainty regarding the optimal indication of adjuvant chemotherapy in stage II CRC, although it is generally believed that the higher the recurrence risk, the more likely that patients derive larger benefits from adjuvant chemotherapy (34). The JCOG1805 trial is a multicenter, randomized controlled trial (RCT) evaluating the superiority of postoperative adjuvant therapy over surgery alone in stage II CRC, with four selected pathological risk factors—pT4, grade 3 tumor budding (BD3), presence of perineural invasion (Pn1) and non-mature (i.e. intermediate or immature) DR patterns (Supplemental Fig. 1). pT4, BD3 and Pn1 are routinely diagnosed and recorded in pathology reports throughout Japan according to the Japanese Classification of Colorectal, Appendiceal, and Anal Carcinoma, published by the Japanese Society for Cancer of the Colon and Rectum [JSCCR; (35)]. On the other hand, the DR classification has yet to be described there, although it was featured in the 77th JSCCR Biannual Meeting in 2018 (http://jsccr.umin.jp/88/dr.html). Nevertheless, the non-mature DR feature was determined to be adopted for use as an inclusion criterion in the JCOG1805 because of its high prognostic value validated in a prospective multicenter study [SACURA trial; (10)]. The JCOG1805 will become the first RCT to determine clinical values of pathological risk factors, including a stromal factor, as treatment indicators in high-risk stage II CRC patients.
Future studies will elucidate the underlying molecular background of adverse prognostic results in tumors with non-mature DR, which is highly suggestive of a link to the protumor function of the cancer microenvironment that switches indolent tumor cells to cells with a high potential for growth and metastasis. Most likely, one would be transforming growth factor-β (TGF-β) family signaling. For example, thick, hyalinized collagen bundles, which are observed not only in tumors with intermediate DR but in most tumors classified as immature DR, are typically observed in keloids, scars that invade adjacent healthy tissue and rarely regress over time. As compared with normal fibroblasts, keloid fibroblasts are associated with increased expression in various growth factors, including TGF-β (36). Since TGF-β is a well-established inducer of EMT (37), and, reportedly, TGF-β levels in CRC are robust predictors of disease relapse in CRC (38), it is presumed that prometastatic program induced by TGF-β exists in the microenvironment of non-mature DR tumors with keloid-like collagen bundles.
In contrast, some previous biological studies showing that some stromal components can act to restrain tumor growth may include key clues to understanding molecular mechanisms of favorable clinical outcomes associated with mature DR. Rhim et al. have demonstrated that reduced stromal desmoplasia by modulating the hedgehog signaling pathway is associated with undifferentiated histology and increased proliferation and metastasis of cancer cells (18). Özdemir et al. have observed that myofibroblast depletion leads to immunosuppression and increased tumor invasion in transgenic mice with delete αSMA+ myofibroblasts in pancreatic cancer (19). These findings obviously indicate that some CAF populations and the microenvironment induced by them act as a potent barrier for tumor growth. In this regard, the heterogeneity of the CAF population may be expressed morphologically as the differential DR pattern, which is associated with tumor differentiation and immune surveillance status. Specifically, mature DR is characterized by a high incidence of well differentiated carcinoma without the dedifferentiation phenotype (11,13,26) or the conspicuous lymphocytes (11,13,39). The issues to be resolved in the future are to verify the hypothesis that the CAF population is different between the mature and non-mature DR patterns; the former act as a prohost by restraining tumors, whereas the latter act as a protumor by supporting tumor cell growth and metastasis.
In conclusion, the DR pattern of the desmoplastic front may be a new stromal prognostic factor to be reported in routine pathology practice for CRC patients. Increasing evidence indicates that it is certainly an essential prognostic determinant independent of the anatomical extent of disease or conventional tumor factors, and its predictive value for chemotherapy efficacy will be established by the JCOG1805 trial, which is the first RCT incorporating the stromal factor as an inclusion criterion to determine the value of adjuvant chemotherapy in CRCs. Further, molecular investigation focusing on the DR classification would lead to better understanding of the heterogeneity of CAFs and to the development of strategies for personalized CRC management.
Funding
This work was supported by the Practical Research for Innovative Cancer Control from Japan Agency for Medical Research and Development (AMED) under Grant Number jp20ck0106584.
Conflict of interest statement
The authors declare that there are no conflicts of interest.
Supplementary Material
Contributor Information
Hideki Ueno, Department of Surgery, National Defense Medical College, Saitama, Japan.
Yoshiki Kajiwara, Department of Surgery, National Defense Medical College, Saitama, Japan.
Yoich Ajioka, Division of Molecular and Diagnostic Pathology, Graduate School of Medical and Dental Sciences, Niigata University, Niigata, Japan.
Tamotsu Sugai, Department of Molecular Diagnostic Pathology, School of Medicine, Iwate Medical University, Iwate, Japan.
Shigeki Sekine, Division of Pathology and Clinical Laboratories, National Cancer Center Hospital, Tokyo, Japan.
Megumi Ishiguro, Department of Translational Oncology, Tokyo Medical and Dental University Graduate School, Tokyo, Japan.
Atsuo Takashima, Gastrointestinal Medical Oncology Division, National Cancer Center Hospital, Tokyo, Japan.
Yukihide Kanemitsu, Department of Colorectal Surgery, National Cancer Center Hospital, Tokyo, Japan.
References
- 1. Chen X, Song E. Turning foes to friends: targeting cancer-associated fibroblasts. Nat Rev Drug Discov. 2019;18:99–115. [DOI] [PubMed] [Google Scholar]
- 2. Erdogan B, Webb DJ. Cancer-associated fibroblasts modulate growth factor signaling and extracellular matrix remodeling to regulate tumor metastasis. Biochem Soc Trans. 2017;45:229–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Isella C, Terrasi A, Bellomo SE, et al. Stromal contribution to the colorectal cancer transcriptome. Nat Genet. 2015;47:312–9. [DOI] [PubMed] [Google Scholar]
- 4. Calon A, Lonardo E, Berenguer-Llergo A, et al. Stromal gene expression defines poor-prognosis subtypes in colorectal cancer. Nat Genetics. 2015;47:320–9. [DOI] [PubMed] [Google Scholar]
- 5. National Comprehensive Cancer Network . NCCN Clinical Practice Guidelines in Oncology; Colon Cancer Version 3. 2020; (4 June 2020, date last accessed)..
- 6. Labianca R, Nordlinger B, Beretta GD, et al. Early colon cancer: ESMO clinical practice guidelines for diagnosis, treatment, and follow-up. Ann Oncol 2013;24:vi64–72. [DOI] [PubMed] [Google Scholar]
- 7. Ueno H, Kanemitsu Y, Sekine S, et al. A multicenter study of the prognostic value of desmoplastic reaction categorization in stage II colorectal cancer. Am J Surg Pathol. 2019;43:1015–22. [DOI] [PubMed] [Google Scholar]
- 8. Nearchou IP, Kajiwara Y, Mochizuki S, Harrison DJ, Caie PD, Ueno H. Novel internationally verified method reports desmoplastic reaction as the most significant prognostic feature for disease-specific survival in stage II colorectal cancer. Am J Surg Pathol. 2019;43:1239–48. [DOI] [PubMed] [Google Scholar]
- 9. Ishiguro M, Mochizuki H, Tomita N, et al. Study protocol of the SACURA trial: a randomized phase III trial of efficacy and safety of UFT as adjuvant chemotherapy for stage II colon cancer. BMC Cancer. 2012;12:281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Ueno H, Ishiguro M, Nakatani E, et al. Prognostic value of desmoplastic reaction characterization in stage II colon cancer: prospective validation in a phase III study (SACURA trial). Br J Cancer. 2021;124:1088–1097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Ueno H, Jones AM, Wilkinson KH, Jass JR, Talbot IC. Histological categorisation of fibrotic cancer stroma in advanced rectal cancer. Gut. 2004;53:581–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ueno H, Jones A, Jass JR, Talbot IC. Clinicopathological significance of the 'keloid-like' collagen and myxoid stroma in advanced rectal cancer. Histopathology. 2002;40:327–34. [DOI] [PubMed] [Google Scholar]
- 13. Ueno H, Shinto E, Shimazaki H, et al. Histological categorization of desmoplastic reaction: its relevance to the colorectal cancer microenvironment and prognosis. Ann Surg Oncol. 2015;22:1504–12. [DOI] [PubMed] [Google Scholar]
- 14. Thiery JP, Acloque H, Huang RYJ, Nieto MA. Epithelial-mesenchymal transitions in development and disease. Cell. 2009;139:871–90. [DOI] [PubMed] [Google Scholar]
- 15. Ueno H, Shinto E, Kajiwara Y, et al. Prognostic impact of histological categorisation of epithelial-mesenchymal transition in colorectal cancer. Br J Cancer. 2014;111:2082–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Mueller MM, Fusenig NE. Friends or foes-bipolar effects of the tumour stroma in cancer. Nat Rev Cancer. 2004;4:839–49. [DOI] [PubMed] [Google Scholar]
- 17. Ronca R, Van Ginderachter JA, Turtoi A. Paracrine interactions of cancer-associated fibroblasts, macrophages and endothelial cells: tumor allies and foes. Curr Opin Oncol. 2018;30:45–53. [DOI] [PubMed] [Google Scholar]
- 18. Rhim AD, Oberstein PE, Thomas DH, et al. Stromal elements act to restrain, rather than support, pancreatic ductal adenocarcinoma. Cancer Cell. 2014;25:735–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Özdemir BC, Pentcheva-Hoang T, Carstens JL, et al. Depletion of carcinoma-associated fibroblasts and fibrosis induces immunosuppression and accelerates pancreas cancer with reduced survival. Cancer Cell. 2014;25:719–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Halvorsen TB, Seim EVA. Association between invasiveness, inflammatory reaction, desmoplasia and survival in colorectal cancer. J Clin Pathol. 1989;42:162–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Japanese Society for Cancer of the Colon and Rectum . Japanese Classification of Colorectal Carcinoma (second English edition). Tokyo: Kanehara & Co., Ltd., 2009. [Google Scholar]
- 22. Sis B, Sarioglu S, Sokmen S, Sakar M, Kupelioglu A, Fuzun M. Desmoplasia measured by computer assisted image analysis: an independent prognostic marker in colorectal carcinoma. J Clin Pathol. 2005;58:32–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Mesker WE, Junggeburt JMC, Szuhai K, et al. The carcinoma-stromal ratio of colon carcinoma is an independent factor for survival compared to lymph node status and tumor stage. Cell Oncol. 2007;29:387–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Ueno H, Konishi T, Ishikawa Y, et al. Histological categorization of fibrotic cancer stroma in the primary tumor is an independent prognostic index in resectable colorectal liver metastasis. Am J Surg Pathol. 2014;38:1380–6. [DOI] [PubMed] [Google Scholar]
- 25. Zippi M, Toma GD, Minervini G, et al. Desmoplasia influenced recurrence of disease and mortality in stage III colorectal cancer within five years after surgery and adjuvant therapy. Saudi J Gastroenterol. 2017;23:39–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Ueno H, Kanemitsu Y, Sekine S, et al. Desmoplastic pattern at the tumor front defines poor-prognosis subtypes of colorectal cancer. Am J Surg Pathol. 2017;41:1506–12. [DOI] [PubMed] [Google Scholar]
- 27. Ueno H, Sekine S, Oshiro T, et al. Disentangling the prognostic heterogeneity of stage III colorectal cancer through histologic stromal categorization. Surgery. 2018;163:777–83. [DOI] [PubMed] [Google Scholar]
- 28. Konishi T, Shimada Y, Lee LH, et al. Poorly differentiated clusters predict colon cancer recurrence: an in-depth comparative analysis of invasive-front prognostic markers. Am J Surg Pathol. 2018;42:705–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Ao T, Kajiwara Y, Yonemura K, et al. Prognostic significance of histological categorization of desmoplastic reaction in colorectal liver metastases. Virchows Arch. 2019;475:341–8. [DOI] [PubMed] [Google Scholar]
- 30. Ao T, Kajiwara Y, Yonemura K, et al. Morphological consistency of desmoplastic reactions between the primary colorectal cancer lesion and associated metastatic lesions. Virchows Arch. 2020;477:47–55. [DOI] [PubMed] [Google Scholar]
- 31. Wang LM, Silva MA, D'Costa Z, et al. The prognostic role of desmoplastic stroma in pancreatic ductal adenocarcinoma. Oncotarget. 2016;7:4183–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Hernández-Ruiz E, Hernández-Muñoz I, Masferrer E, et al. A myxoid fibrotic reaction pattern is associated with metastatic risk in cutaneous squamous cell carcinoma. Acta Derm Venereol. 2019;99:89–94. [DOI] [PubMed] [Google Scholar]
- 33. Cao L, Sun P-L, He Y, Yao M, Gao H. Desmoplastic reaction and tumor budding in cervical squamous cell carcinoma are prognostic factors for distant metastasis: a retrospective study. Cancer Manag Res. 2020;12:137–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Venook AP, Niedzwiecki D, Lopatin M, et al. Biologic determinants of tumor recurrence in stage II colon cancer: validation study of the 12-gene recurrence score in cancer and leukemia group B (CALGB) 9581. J Clin Oncol. 2013;31:1775–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Japanese Society for Cancer of the Colon and Rectum . Japanese Classification of Colorectal, Appendiceal, and Anal Carcinoma (third English edition). Tokyo: Kanehara & Co., Ltd., 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Bran GM, Goessler UR, Hormann K, Riedel F, Sadick H. Keloids: current concepts of pathogenesis (review). Int J Mol Med. 2009;24:283–93. [DOI] [PubMed] [Google Scholar]
- 37. Lamoulle S, Xu J, Derynck R. Molecular mechanisms of epithelial–mesenchymal transition. Nat Rev Mol Cell Biol. 2014;15:178–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Calon A, Espinet E, Palomo-Ponce S, et al. Dependency of colorectal cancer on a TGF-beta-driven programme in stromal cells for metastasis initiation. Cancer Cell. 2012;22:571–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Ueno H, Shinto E, Hashiguchi Y, et al. In rectal cancer, the type of desmoplastic response after preoperative chemoradiotherapy is associated with prognosis. Virchows Arch. 2015;466:655–63. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


