Abstract
Acute radiation syndrome (ARS) is the radiation toxicity that can affect the hematopoietic, gastrointestinal, and nervous systems upon accidental radiation exposure within a short time. Currently, there are no effective and safe approaches to treat mass population exposure to ARS. Our study aimed to evaluate the therapeutic potential of allogeneic adipose‐derived stem cells (ASCs) for total body irradiation (TBI)‐induced ARS and understand the underlying mitigation mechanism. We employed 9.25 Gy TBI dose to C57BL/6 mice and studied the effect of allogeneic ASCs on mice survival and regeneration of the hematopoietic system. Our results indicate that intraperitoneal‐injected ASCs migrated to the bone marrow, rescued hematopoiesis, and improved the survival of irradiated mice. Our transwell coculture results confirmed the migration of ASCs to irradiated bone marrow and rescue hematopoietic activity. Furthermore, contact coculture of ASCs improved the survival and hematopoiesis of irradiated bone marrow in vitro. Irradiation results in DNA damage, upregulation of inflammatory signals, and apoptosis in bone marrow cells, while coculture with ASCs reduces apoptosis via activation of DNA repair and the antioxidation system. Upon exposure to irradiated bone marrow cells, ASCs secrete prosurvival and hematopoietic factors, such as GM‐CSF, MIP1α, MIP1β, LIX, KC, 1P‐10, Rantes, IL‐17, MCSF, TNFα, Eotaxin, and IP‐10, which reduces oxidative stress and rescues damaged bone marrow cells from apoptosis. Our findings suggest that allogeneic ASCs therapy is effective in mitigating TBI‐induced ARS in mice and may be beneficial for clinical adaptation to treat TBI‐induced toxicities. Further studies will help to advocate the scale‐up and adaptation of allogeneic ASCs as the radiation countermeasure.
Keywords: acute radiation syndrome, adipose stem cells, cytokines, hematopoiesis, intraperitoneal injection, total body irradiation

Significance statement.
Acute radiation syndrome (ARS) is the radiation toxicity that can affect the hematopoietic, gastrointestinal, and nervous systems upon accidental radiation exposure within a short time period. Currently, there are no effective and safe approaches to treat mass population exposure to ARS. This study revealed a possible application of allogeneic adipose‐derived stem cells (ASCs) as a mitigator. These cells are available in large quantities and can be stored for application at mass levels in case of radiation accidents. Future studies in higher animals will enable the clinical adaptation of ASCs‐based therapies.
1. INTRODUCTION
Acute radiation syndrome (ARS) is characterized as an acute illness caused by total body irradiation (TBI) or exposure of the majority of the body to a high radiation dose in a very short time span. 1 ARS manifestation mainly involves hematopoietic and gastrointestinal organ failure, followed by neurological and other organ damage. 1 , 2 , 3 , 4 , 5 Events leading to the development of ARS can be a radiation accident like in Chernobyl (1986) or Fukushima (2011), a therapeutic misadventure, or nuclear weapon detonation during war or in a terroristic attack. 1 , 6 , 7 The threats for such events have increased sharply in recent years. 8 , 9 , 10 As such, events are modeled to impact the mass population, so individual patient management is not possible. Rather, it requires therapeutic approaches that can be administered within 24 hours with minimal expert staff supervision.
The extent of damage caused by TBI depends on the mode of exposure, irradiation dose, and the rate of exposure. TBI can cause harmful effects such as fatigue, lightheartedness, and vomiting. At high doses, the patient can die within 24 to 48 hours due to cerebrovascular syndrome. Lower doses can cause a hematopoietic and gastrointestinal syndrome that can lead to death in less than 10 days. 11 In almost all instances of TBI exposure, the primary life‐threatening damage is inflicted on the hematopoietic system. Long‐term effects of TBI include problems with senses, cognition, communication, behavior, and emotions. Research suggests TBI increases the chances of degenerative illness due to breakdown and damage of brain cells. Such problems include Alzheimer's disease, Parkinson's disease, and chronic traumatic encephalopathy. 12
At present, few treatment options are available. Matched hematopoietic stem cell transplant is the therapy of choice. Although bone marrow transplantation is beneficial, maintenance of hematopoietic stem cells, radiation dose determination, and the lack of matched stem cell donor for allogeneic therapy applications are limiting factors. In a mass population exposure scenario, several hundred to millions of individuals can be exposed; from a practical standpoint, hematopoietic stem cell transfusion is an impossible way forward. 2 In case such scenario develops, it will be difficult to treat all individuals at the same time, and treatment will be delayed for a large number of affected individuals, which may lead to an increased mortality rate. Other possible treatment options include administration of prohematopoietic cytokines, such as granulocyte‐macrophage colony‐stimulating factor (GM‐CSF) and granulocyte stimulating factor (G‐CSF). 13 , 14 , 15 Both of these factors are approved by the United States Food and Drug Administrative Authority (FDA) for bone marrow regeneration following radiotherapy. 10 Other drugs at an earlier stage of development include Amifostine/WR2721, Genistein, Gamma‐tocotrienol, CBLB502 (bioengineered truncated Salmonella sp. Flagellin), anti‐inflammatory cytokines, growth factors, prostaglandins have been proposed for mitigation of ARS. 10 Despite the progress, none of these drugs provide complete protection and their use is associated with side effects, so there is a need to research alternative therapy approaches.
An attractive alternative to hematopoietic stem cells is the development of allogenic cell therapies that can indirectly support the recovery of hematopoiesis. Bone marrow‐derived mesenchymal stem cells from nonhematopoietic linage are important players for repairing damaged tissue and can differentiate into multilineages such as adipocytes, osteocytes, and chondrocytes. Studies have reported that bone marrow‐derived mesenchymal stem cells mitigate ARS. 16 , 17 Other approaches employing placental‐derived and Wharton's jelly derived mesenchymal stem cells rescue from radiation by regenerating damaged bone marrow from TBI. 18 , 19 , 20 Low yield and difficulty in harvest and mass production for stockpiling are some of the drawbacks associated with the therapeutic application of these cells.
Adipose tissue‐derived stem cells (ASCs) have self‐renewal capacity and can undergo differentiation to mature cells, such as adipocytes and osteocytes. 21 , 22 ASCs are found abundantly in adipose tissue, can be easily harvested in vast quantities, and are very easy to propagate in cell culture compared to other cell types. 22 Upon enzymatic digestion, adipose tissue can mainly be divided into two different fractions: mature adipocytes and the stromal vascular fraction. 23 The stromal vascular fraction is composed of ASCs, preadipocytes, endothelial cells, lymphocytes, and pericytes. 24 ASCs are plastic adherent and exhibit fibroblastic or spindle‐shaped morphology and can be maintained up to 12 to 18 passages in cell culture. 25 ASCs are characterized by the expression of cell surface markers such as CD34, CD26, CD29, and CD90. 22 , 26 , 27 , 28 Reports show that ASCs acts as an anti‐fibrotic agent to ameliorate the radiation‐induced fibrosis. 28 An important attribute of ASCs is the immune‐modulatory effect on the tissue microenvironment. ASCs are less or nonimmunogenic due to their lack of MHCII expression and relatively low MHCI expression. 22 Moreover, secretory factors produced by ASCs exert anti‐inflammatory and immune‐suppressive effects and play a vital role in the regeneration of damaged tissues. Importantly, autologous SVF from adipose tissue has been reported to regenerate the damaged bone marrow from TBI and improve survival. 29 The limiting factors for using allogeneic SVF is the presence of immune cells in SVF, which can cause graft vs host disease in recipients, and the requirement for a very large tissue volume for cell harvest. Earlier research suggested that ASCs can regenerate the hematopoietic system from damaged bone marrow. 30 Therefore, regeneration or reparation of damage caused by TBI is possible through allogeneic, cell culture propagated ASCs that can be a novel and alternative strategy for mitigation of ARS.
We are showing for the first time the use of allogeneic ASCs therapy to mitigate TBI‐induced ARS and that intraperitoneally injected ASCs migrate to bone marrow and facilitate repair by secreting hematopoietic and prosurvival factors. This therapy can be conveniently upscaled for a national stockpile storage for mass applications.
2. METHODS
2.1. Mice
Female, 6 to 8 weeks aged wild type and transgenic for GFP, C57 BL/6, and FVB mice (Taconic Biosciences, Wayne, Pennsylvania) were used in this study. Animals were housed in an Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC)‐approved facility and were treated according to the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
2.2. Irradiation
A cesium‐137 gamma cell irradiator was used for delivering TBI to mice at a rate of 310 cGy/min. Beam flatness, homogeneity, and spectrum of gamma‐ray dosimetry was analyzed as published. 31
2.3. Isolation and maintenance of ASCs
C57BL/6 and FVB transgenic mice expressing green fluorescent protein (GFP) were used for the ASCs transplantation studies. ASCs were isolated, as published previously. Briefly, subcutaneous fat tissues were harvested from the transgenic mice of either C57BL/6 or FVB‐GFP mice, washed with 1× phosphate‐buffered saline (PBS) (Sigma, St. Louis, Missouri), and chopped into pieces using scissors. The minced tissue was subjected to collagenase (Worthington, New Jersey) treatment and incubated at 37°C for 45 minutes in shaking incubator. Following incubation, the digested tissue was filtered through a 100 μm cell strainer and centrifuged at 300g for 10 minutes at room temperature. Thereafter, the supernatant containing mature adipocytes was discarded, and the sediment containing the stromal vascular fraction (SVF) was incubated with erythrocyte lysis buffer (Thermo Fisher Scientific, Waltham, Massachusetts) for 5 minutes at room temperature, filtered using a 70 μm cell strainer, and washed with PBS. After centrifugation, the supernatant was discarded and the SVF was suspended in Dulbecco's Modified Eagle's medium (DMEM; Sigma, St. Louis) with 10% FBS (Sigma, St. Louis) and filtered through 40 μm nylon mesh (Thermo Fisher Scientific, Waltham). The cell suspension was subjected to centrifugation at 300g for 10 minutes. Following centrifugation, the supernatant was discarded and the pellet containing the SVF was suspended in DMEM with 10% FBS and plated in a 75 cm2 tissue culture flask at a seeding density of 30 000 cells/cm2 and maintained at 37°C in 5% carbon dioxide (CO2). The medium was changed every 3 days, and the ASCs were harvested at 80% confluence using trypsinization. ASCs expanded to passage four were used for cell therapy and coculture studies.
2.4. Phenotyping of ASCs
For surface marker expression, 1× 105 ASCs of either C57BL/6‐GFP or FVB‐GFP origin were trypsinized and centrifuged. The cell pellet was washed with 1× PBS and fixed with 4% paraformaldehyde for 20 minutes. Subsequently, the cells were washed using 1× PBS, and the surface marker antibodies against CD34, CD29, CD90, CD26 (BD Biosciences, California), and CD45 (BioLegend, California) were added to the cells and incubated for 30 minutes at 4°C in the dark. The cells were washed using 2% FBS in 1× PBS and analyzed using a FACS Fortessa flow cytometer (BD Biosciences). Data were analyzed using FlowJo software.
2.5. Transplantation of ASCs
For the autologous and allogeneic GFP‐ASCs transplantation studies, we used wild‐type C57BL/6 and FVB mice as recipients. Mice were subjected to TBI using a radiation dose of 9.25 Gy for C57BL/6 and 8.75 Gy for FVB mice. Twenty four hours post TBI, the mice were intraperitoneally injected with 5 × 106 GFP expressing autologous or allogeneic ASCs or PBS as control. Mice were monitored for survival for 30 days.
2.6. ASCs migration to bone marrow
To study the migration of ASCs from the peritoneal cavity to bone marrow, irradiated mice surviving at the end of the ASCs transplantation studies, injected with either ASCs or PBS, were sacrificed at day 35 postirradiation. The bone marrow cells were isolated following the protocol with slight modifications. 32 , 33 Using aseptic techniques, mice hind limbs were surgically removed and shifted to the lab for further processing under sterile conditions. Skin and muscles were removed from the tibia and femur. After washing with PBS, the tibia and femur were separated by cutting the joint, and bone ends were cut open using scissors. The bones were flushed with DMEM medium using 3 mL syringes and 28‐gauge needles. The marrow was thoroughly resuspended with a pipette and centrifuged at 300g for 10 minutes. Red blood cells were lysed using ACK buffer. The cells were then filtered, centrifuged, resuspended in DMEM 10% FBS, and cultured in six‐well plates, with individual wells for each mouse. Bone marrow cells were cultured and screened for GFP positive cells for 60 days.
2.7. In vitro transwell migration assay
Transwell migration assays were performed to evaluate ASCs (C57BL/6‐GFP and FVB‐GFP) migration in transwell chambers (six‐well insert, pore size: 0.4 mm). At day 0, 2 × 105 ASCs of either C57BL/6‐GFP or FVB‐GFP origin were plated in the top basket of the transwell and incubated overnight to allow the cells to attach. After 24 hours, irradiated (3 × 105) and nonirradiated bone marrow cells (3 × 105) were plated in the lower chamber of the transwell inserts and cocultured for 2 weeks. After 2 weeks of culture, the lower chamber cells were used for testing bone marrow survival and ASC migration. Monoculture ASCs or bone marrow cells were used as controls.
2.8. Contact coculture assay
ASCs originating from C57BL/6 mice or FVB mice were grown to confluency in six‐well plates. 3 × 105 bone marrow cells from irradiated and nonirradiated mice were overlaid on ASCs and cocultured for 2 weeks. Number of cobble stone formation was documented. In addition, the effect of contact coculture on cell survival was monitored.
2.9. Actin phalloidin staining and imaging
Briefly, cultured ASCs or ASC‐bone marrow cocultures from transwell migration experiments were fixed with 4% paraformaldehyde and incubated for 20 minutes at room temperature. Cells were washed using 1× PBS three times for 5 minutes each, permeabilized with 0.1% Triton X‐100 for 10 minutes, followed by washes with 1× PBS three times for 5 minutes each. Afterward, the cells were blocked with 5% bovine serum albumin (BSA) for 1 hour at room temperature in the dark. F‐actin was stained using phalloidin (Abcam, Cambridge, Massachusetts) labeled with TRITC by incubation overnight at 4°C in the dark. The cell nuclei were stained blue with 1 μg/mL Hoechst 33342 or DAPI (Thermo Fisher Scientific, Waltham). Images were documented by fluorescent microscopy (Keyence, Osaka, Japan).
2.10. Apoptosis analysis
Bone marrow cells and ASCs were harvested from the transwell migration or contact coculture experiments after 2 weeks of coculture. The cells were washed with 1× PBS and stained with APC‐conjugated annexin‐V and propidium iodide (PI) in binding buffer (BioLegend kit) for 15 minutes at room temperature. For bone marrow cell survival, GFP positive and propidium iodide negative cells were gated and analyzed for their expression of annexin‐V. Samples were acquired on a FACS Fortessa and were analyzed using FlowJo software.
2.11. Preparation and injection of conditioned media
ASCs‐conditioned media was prepared to study the effects of cytokines and growth factors released from ASCs (C57BL/6 and FVB). 2 × 105 of ASCs were plated in a 75 cm2 flask and cultured to 80% confluency. DMEM with 10% FBS was added to the flasks and cultured for 72 hours at 37°C after being washed with PBS twice. The conditioned medium was filtered and stored at −80°C until further use. Culture medium without ASCs served as a negative control.
To test the effect of ASCs derived conditioned medium on mouse survival, we used wild‐type C57BL/6 mice and subjected for TBI with 9.25 Gy. After 24 hours, irradiated mice were injected daily with conditioned medium from C57BL/6 or FVB‐GFP ASCs or control medium or PBS for 1 week. After 1 week, mice were injected every 3 days and continued for 30 days with continuous mouse survival monitoring.
2.12. Bone marrow culture in ASCs conditioned media
Irradiated or nonirradiated bone marrow cells were cultured in ASCs conditioned media for 2 weeks. Cobblestone formation was visualized by microscopy and cell survival by Annexin/PI FACS staining.
2.13. Protein measurements by Luminex assay
The multiplex Luminex assay analyzed the concentration of secreted proteins in the conditioned medium according to the manufacturer's protocol (Milliplex, Millipore Sigma, Massachusetts). Cell culture supernatant samples were centrifuged for 4 minutes at 16 000g immediately before use or dilution. Samples were diluted twofold by adding 75 μL supernatant to 75 μL of Calibrator Diluent mix (Kit). A 50 μL of the diluted sample or standard (Kit) was added to 96‐well plates according to assay scheme. Incubation was performed for 2 hours at room temperature after adding 50 μL Microparticle Cocktail (Kit). Each was washed three times with 100 μL wash buffer (Kit). A 50 μL diluted biotin antibody cocktail was added to each well and incubated for 1 hour with shaking at 800 rpm. Each well was then washed three times with wash buffer. A 50 μL diluted Streptavidin‐PE was added to each well and incubated for 30 minutes with shaking at 800 rpm, after which each well was again washed three times with wash buffer. Readings were taken using the Luminex 100/200 analyzer (Luminex Inc, Austin, Texas) and protein concentrations reported.
2.14. Real‐time polymerase chain reaction (PCR)
RNA was extracted from cells using the RNeasy plus Mini kit (Qiagen, Germany). RNA was reverse transcribed using High Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Thermo Fisher Scientific, Waltham) according to manufacturer's protocol. Using cDNA as a template and gene specific primers (KiCqStart SYBR Green Primers Millipore Sigma) expression of target genes was quantified by quantitative real time PCR using the Fast SYBR Green Master Mix Applied Biosystems (Thermo Fisher Scientific). Data for each gene transcript were normalized by calculating the difference (∆Ct) from the Ct‐housekeeping and Ct‐target genes. The relative increase or decrease in expression was calculated by comparing the reference gene with target gene calculated by comparing the reference gene with the target gene (∆∆Ct) and using the formula for relative expression (=2∆∆Ct).
2.15. Data analysis
FlowJo was used to analyze the flow cytometry data. GraphPad Prism and Microsoft Excel were used for statistical analysis. One‐way ANOVA or Student t tests were performed to compare the groups. The statistically significant P‐value considered for analysis was <.05.
3. RESULTS
3.1. Characterization of ASCs
We first verified the presence of characteristic ASCs features in cells isolated from subcutaneous adipose tissue of C57BL/6 and FVB mice. ASCs were plastic adherent, grew as a monolayer, exhibited fibroblastic, spindle‐shaped, or mesenchymal morphology, and maintained up to eight passages without losing their characteristics (Figure 1A). To examine the actin cytoskeleton of ASCs from two different mice strains, the F‐actin was visualized using phalloidin‐TRITC staining and assessed. ASCs deriving from C57BL/6 and FVB showed a similar parallel arrangement of F‐actin and no alterations in actin cytoskeleton distribution or integrity were observed (Figure 1B). A hallmark phenotypic identification feature of ASCs is the expression of surface markers such as CD34, CD90, CD73, CD29, and CD26. 26 , 27 , 34 To confirm the phenotype of ASCs used in this study, we analyzed the surface marker expression by flow cytometry. Immunophenotyping analysis showed that GFP‐expressing ASCs were positive for CD34, CD29, CD26, and CD90 and negative for CD45 (Figure 1C). Moreover, ASCs showed robust differentiation to adipocytes upon treatment with adipogenic medium, demonstrated by the accumulation of Oil Red O stained lipid droplets (Figure 1D).
FIGURE 1.

Characterization of allogeneic adipose‐derived stem cells (ASCs). Phase‐contrast microscopic images represent the morphology of ASCs derived from adipose tissues of C57BL/6‐GFP and FVB‐GFP (A). The microscopic fluorescence images of GFP positive ASCs fixed and stained with TRITC‐phalloidin for F‐actin (red) and Hoechst to visualize cell nuclei (blue) (B). Scale bars = 100 μm. Magnification 10x. C, Representative flow cytometry histograms. Expression of CD34, CD29, CD90, CD105, CD26, and CD45 was analyzed. D, Isolated ASCs were subjected to adipocyte differentiation for 14 days using the adipocyte differentiation medium. Oil‐Red‐O staining to confirm differentiation was performed and shown
3.2. Intraperitoneal injection of allogeneic ASCs rescues mice from severe ARS
We aimed to evaluate the efficacy of allogeneic ASCs to mitigate ARS. To achieve this goal, we compared the potential of intraperitoneal (i.p.) injection of autologous (C57BL/6) (Figure 2A) or allogeneic (FVB) (Figure 2C) ASCs or PBS control to rescue 9.25 Gy TBI C57BL/6 mice. Injections were performed 24 hours postirradiation and mice survival was observed for 30 days. Kaplan‐Meier estimator survival analysis showed that a single injection of 5 × 106 autologous (Figure 2B) or allogeneic ASCs (Figure 2D) rescued the mice from TBI‐induced ARS. We observed significant increase in white blood cell counts in ASCs treated mice (Figure 2E). Cell dose was selected based on a previously published study. 35
FIGURE 2.

Allogeneic and autologous allogeneic adipose‐derived stem cells (ASCs) transplant improve the survival of total body irradiated mice. Schematic representation of autologous ASCs (A) and allogeneic ASCs (C) transplanted to irradiated C57 BL/6 recipients. (B) The survival curve of irradiated C57BL/6 mice injected with autologous ASCs or phosphate‐buffered saline (PBS) as control is shown. Survival was monitored for 30 days (n = 20/group). D, Survival curve of irradiated C57BL/6 mice injected with allogeneic ASCs or PBS as control is shown. Survival was monitored for 30 days (n = 20/group). E, White blood count analyses at day 14 post‐ASCs injection. The results are presented as mean ± SD; *P < .05. F,G, Schematic representation of autologous ASCs (F) and allogeneic ASCs (G) transplanted to irradiated FVB recipients. H, Survival curve of irradiated FVB mice injected with either autologous ASCs, allogeneic ASCs, or PBS. Survival was monitored for 30 days (n = 10/group). I, Survival curve of 9.25 Gy irradiated C57BL/6 mice injected with either C57BL/6 ASCs CM, FVB ASCs CM, control culture media, or PBS. Survival was monitored for 10 days (n = 10/group)
The use of allogeneic ASCs as a mitigator of ARS is the practical therapeutic approach. We observed that allogeneic ASCs improved the survival of TBI C57BL/6 mice, and we were interested in verifying our results in a different mice strain. So, we reversed the recipient mice strain and employed TBI FVB mice as the recipient and test the efficacy of allogeneic (C57BL/6) ASCs to rescue TBI mice. For this, FVB mice were subject to TBI with a dose of 8.75 Gy as 9.25 Gy proved fatal for FVB mice. After 24 hours, autologous ASCs (FVB) (Figure 2F), allogeneic (C57BL/6) (Figure 2G) ASCs, or PBS as control was injected i.p. into the irradiated FVB mice, and mice survival was observed for 30 days. Both autologous (FVB) and allogeneic (C57BL/6) ASCs improved mice survival (Figure 2H). Our findings suggested that allogeneic ASCs mitigate TBI‐induced ARS irrespective of the host species, and the mitigation efficacy is comparable to autologous cells.
ASCs are well known to secrete prohematopoietic factors (Supplementary Table 1). 36 Next, we investigated the therapeutic effect of ASCs conditioned medium on the survival of TBI mice to test whether the factors released by the ASCs are capable to mitigate ARS. For this, medium from cultured C57BL/6 or FVB ASCs was injected daily for the first 7 days and twice a week afterward to 9.25 Gy TBI C57BL/6 mice for 30 days starting 24 hours postirradiation. Fresh cell culture media was used as a control. Observation for mice survival lasted for 30 days. Results revealed that although ASCs have shown to secrete hematopoietic factors in the cell culture medium, the concentration of these factors in the conditioned media was not high enough to rescue hematopoiesis and prolong the survival of TBI mice (Figure 2I).
3.3. Intraperitoneal‐injected ASCs migrate to the bone marrow of TBI mice
Failure of ASCs conditioned media to rescue TBI mice pointed toward the need for a highly concentrated and sustained presence of factors released by ASCs for mitigation. High concentrations and sustainability are most likely achievable if the ASCs migrate to the mitigation site, that is, bone marrow. To determine whether GFP positive injected ASCs migrated from the peritoneal cavity to bone marrow in vivo, we performed cell tracing studies. Bone marrow was isolated from surviving irradiated mice injected with either ASCs or PBS and control nonirradiated mice after 35 days postirradiations and cultured for up to 60 days. Our observation revealed that both autologous and allogeneic GFP positive ASCs migrated to irradiated bone marrow (Figure 3A). Bone marrow is composed of heterogeneous populations consisting of hematopoietic stem cells and mesenchymal stem cells. 37 Cells were fixed with paraformaldehyde and stained with Phalloidin TRITC (Red), and nuclei (blue) with Hoechst to identify the GFP positive injected ASCs from non‐GFP positive host bone marrow populations. Microscopic images represent the cells that were positive for GFP, Phalloidin TRITC, and Hoechst and referred as migrated ASCs, whereas bone marrow cells were positive for only Phalloidin TRITC and Hoechst (Figure 3A). In the earlier time points of culture, that is, day 24, the GFP positive ASCs appeared rounded (Figure 3A) and stressed; but by day 52, transplanted ASCs attained their original spindle‐shaped or mesenchymal morphology (Figure 3B). These results suggested that the mitigation of ARS is mediated by the migration of the transplanted ASCs to irradiated bone marrow.
FIGURE 3.
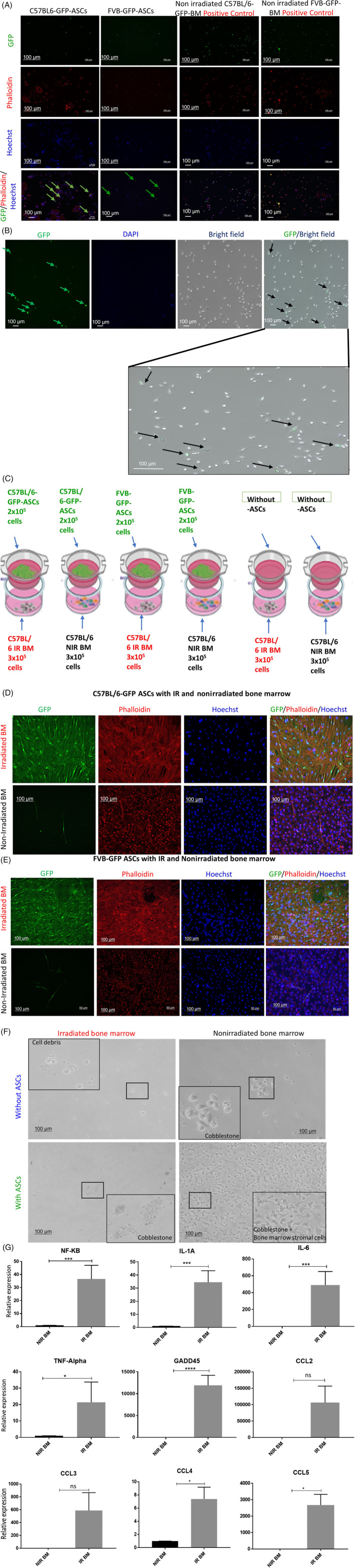
Transplanted allogeneic adipose‐derived stem cells (ASCs) migrate to the bone marrow of irradiated mice. A, Bone marrow from day 35 postirradiation surviving mice was isolated and cultured. Day 24 postculture bone marrow cells were fixed and stained with TRITC‐phalloidin for F‐actin (red) and Hoechst to visualize cell nuclei (blue). B, Day 52 postculture images for green fluorescent protein (GFP) positive cells. C, Schematic representation of transwell migration experiment. D,E, Fluorescence microscopy images of the ASCs isolated from C57BL/6‐GFP (D) and FVB‐GFP (E) mice cocultured with irradiated and nonirradiated bone marrow. The cells were fixed with 4% paraformaldehyde and stained with Phalloidin‐TRITC for F‐actin (red). Hoechst was used for staining nuclei (blue). ASCs: GFP+, Phalloidin‐TRITC+, and Hoechst+. Bone marrow cells: GFP−, Phalloidin‐TRITC+, and Hoechst+. Scale bars = 100 μm. Magnification 10x. F, Bone marrow from 24 hours postirradiated mice, or nonirradiated mice were cocultured with either allogeneic or autologous ASCs for 2 weeks. ASCs were seeded in the top chamber, and bone marrow cells from irradiated and nonirradiated were seeded in the lower chamber. Phase‐contrast microscopic images of cobblestone formation during coculture. G, Ex vivo real‐time quantitative gene expression analysis of irradiated bone marrow cells 24 hours postirradiation. The results are presented as mean ± SD; **P < .01, *P < .05
3.4. ASCs migrate and rescue irradiated bone marrow in transwell cultures
To further confirm that ASCs migrate toward the damaged bone marrow, we performed an in vitro transwell migration assay. As shown in the experimental scheme (Figure 3C), GFP expressing autologous (C57BL/6) and allogeneic (FVB) ASCs were cocultured for 2 weeks with bone marrow harvested from 9.25 Gy irradiated mice 24 hours postirradiation and nonirradiated C57BL/6 mice. Bone marrow cells without ASCs served as the control. Cells in the lower compartment were fixed, and stained with Phalloidin TRITC to visualize F‐actin and Hoechst for nuclear stain. Migrated ASCs were identified as being GFP positive. As shown in Figure 3D,E, a large number of ASCs migrated through the transwell porous membranes in response to irradiated cells cocultured in the bottom chamber, while very few ASCs migrated upon coculture with nonirradiated bone marrow. ASCs either exposed to UV light or fixed with ethanol or formaldehyde failed to show any migration in response to coculture with irradiated or nonirradiated bone marrow cells (data not shown). Of note, we used a transwell pore size of 0.4 μm in our experiments, and we observed a drastic morphological change in the freshly migrating ASCs to a needle‐like morphology, indicating an intense migration pressure on cells which support our in vivo ASCs migration observations. To correlate the functional impact of migrating ASCs on the rescue of the hematopoietic character of bone marrow cells in vitro, we observed that ASCs coculture improved cobblestone formation, a characteristic feature of in vitro hematopoietic activity, in irradiated bone marrow wells compared to wells without ASCs (Figure 3F). To determine the possible signals generated by damaged bone marrow cells which attract the ASCs, we analyzed the gene expression of inflammatory chemokines and cytokines. Real‐time quantitation of genes revealed upregulation of NF‐kB, IL‐6, IL‐1, TNFα, CCL2, CCL3, CCL4, and CCL5 (Figure 3G). A significant upregulation in DNA damage‐induced gene GADD45 was recorded in irradiated bone marrow cells (Figure 3G).
3.5. ASCs migration positively correlated with improved survival of irradiated bone marrow cells
Radiation exposure results in hematopoietic cell damage and apoptosis. Since our transwell experiments demonstrated that ASCs migrated to the irradiated bone marrow, we next sought to analyze the impact of ASCs migration on the survival of irradiated bone marrow cells. To achieve this goal, we trypsinized the cells at the bottom of the transwells and stained them for Annexin V and PI. First, we compared the number of migrating GFP positive ASCs upon coculture with irradiated and nonirradiated bone marrow by counting 10 000 cells by flow cytometry. Data revealed that the number of migrated autologous (C57BL/6) (Figure 4A) and allogeneic (FVB) (Figure 4E) ASCs were higher in irradiated bone marrow cocultures compared to nonirradiated bone marrow cocultures. Next, we analyzed the effect of ASCs migration on the survival of bone marrow cells and determined the Annexin V/PI positive percentage by gating on GFP negative cells. Results revealed only cell debris in the irradiated bone marrow culture without ASCs; however, upon coculture with ASCs, we observed the majority of the irradiated bone marrow cells survived (Supplementary Figure 1). Interestingly, the percentage of irradiated bone marrow cell survival (Annexin V/PI negative cells) correlated positively to the percentage of ASCs migrating through the transwell membranes (Figure 4A,B,E,F). On the other hand, only a small number of autologous (Figure 4D) and allogeneic (Figure 4H) ASCs migrated in the nonirradiated bone marrow cocultures. Nevertheless, they improved the bone marrow cell survival. To analyze the possible mechanism by which ASCs coculture reduce the apoptosis in irradiated bone marrow cells, we cocultured irradiated bone marrow isolated from C57BL/6 mice with ASCs isolated from GFP+Luc+ mice. We sorted the GFP negative bone marrow cells following coculture and analyzed the expression of DNA damage repair, antioxidant, and inflammation related genes by real‐time quantitative PCR. Results showed a significant upregulation in the expression of DNA damage repair gene BRCA2, antioxidant gene superoxide dismutase (SOD1) and downregulation in NF‐kB and TNF expression (Figure 4I). Our results indicated that irradiated bone marrow stimulates ASCs migration, and migrated ASCs improve the bone marrow recovery and protect them from oxidative DNA damage‐induced apoptosis.
FIGURE 4.

Allogeneic adipose‐derived stem cells (ASCs) migrate through the transwell and protect irradiated bone marrow from apoptosis. A,C, Migration of C57BL/6 ASCs across the transwell membrane toward irradiated bone marrow (A) and nonirradiated bone marrow (C) was analyzed by FACS. The percentage of GFP negative (bone marrow cells) and GFP positive (ASCs) as determined. B,D, Apoptosis of cocultured irradiated (B) and nonirradiated (D) bone marrow cells was analyzed by Annexin V and PI staining by FACS. E,G, Migration of FVB ASCs across the transwell membrane toward irradiated bone marrow (E) and nonirradiated bone marrow (G) was analyzed by FACS. The percentage of GFP negative (bone marrow cells) and GFP positive (ASCs) was determined. F,H, Apoptosis of cocultured irradiated (F) and nonirradiated (H) bone marrow cells was analyzed by Annexin V and PI staining by FACS. I, Real‐time quantitative gene expression analysis of sorted cocultured irradiated bone marrow cells. The results are presented as mean ± SD; **P < .01, *P < .05
3.6. Contact coculture improves irradiated bone marrow survival
As our in vivo and transwell cocultures demonstrated that ASCs migrated to irradiated bone marrow cells, which seems to be a crucial factor for mitigation; next, we analyzed the impact of the cell to cell contact of ASCs to irradiated bone marrow on cell survival. For this, bone marrow from irradiated and nonirradiated mice was cocultured with autologous (C57BL/6) and allogeneic (FVB) ASCs for 2 weeks, and cell death was determined in GFP negative bone marrow cells. ASCs contact coculture improves irradiated bone marrow cells survival and cobblestone formation compared to irradiated bone marrow cells cultured without ASCs (Figure 5A). Annexin V/PI staining of cocultured cells revealed that ASCs protect irradiated bone marrow cells from apoptosis, whereas irradiated bone marrow cultured without ASCs showed a higher percentage of Annexin V/PI positive apoptotic cells (Figure 5B,C). We further tested the capacity of growth factors and cytokines present in the ASCs condition medium to support irradiated bone marrow survival in cell culture. Irradiated bone marrow cultured with or without condition medium for 2 weeks showed no differences in cell survival or cobblestone formation (Figure 5D). We conclude that cell to cell contact improves the efficiency of ASCs to mitigate radiation‐induced bone marrow damage.
FIGURE 5.

The contact coculture of allogeneic adipose‐derived stem cells (ASCs) improves the survival of the irradiated bone marrow. A, Morphology of cocultured irradiated bone marrow with or without ASCs. Scale bars = 100 μm. Magnification 10x. B, Irradiated bone marrow cocultured with or without ASCs were stained with Annexin‐V‐APC/Propidium iodide and analyzed by FACS. Flow cytometry histograms of non‐GFP positive gated bone marrow cells apoptosis. C, The percentage of apoptosis during contact coculture. The results are presented as mean ± SD; **P < .01. D, Phase‐contrast images of irradiated and nonirradiated bone marrow cultured in ASCs conditioned media or fresh media. Scale bars = 50 μm. Magnification 10x
3.7. ASCs mitigate irradiation damage by secreting hematopoietic cytokines
Stromal cells secreted cytokines, chemokines, and growth factors are known to be involved in the hematopoietic stem cell maintenance and survival, thus supporting bone marrow recovery after injury. 38 , 39 , 40 , 41 Our allogeneic ASCs therapy results show the successful rescue of TBI mice mitigated by the migration of ASCs to bone marrow. To determine the mitigatory factors released by ASCs upon interaction with irradiated bone marrow cells, we performed a Luminex screen for cytokines involved in hematopoiesis, cell survival, and chemotaxis using cell culture supernatant collected at day 7 and day 14 from the allogeneic transwell experiments. In support to our findings of enhanced mice survival, reduced irradiated bone marrow cells damage, and enhanced cobblestones formation, we observed a significant increase in the production of hematopoietic cytokines GM‐CSF, MIP1α, MIP1β, LIX, KC,1P‐10, Rantes, IL‐17, MCSF, TNFα, Eotaxin, and IP‐10 (Figure 6A‐L). These cytokines were secreted at low levels by ASCs cultured with or without nonirradiated bone marrow cells, but significant upregulation was observed upon interaction with irradiated bone marrow cells. Besides, we also analyzed several other cytokines shown in the literature to be involved in hematopoiesis but observed no notable changes in supernatant from nonirradiated or irradiated bone marrow cocultures with ASCs (Supplementary Figure 2). Our results suggested that allogeneic ASCs support irradiated bone marrow survival via secreting a set of specific hematopoietic and prosurvival cytokines shown in Supplementary Table 2.
FIGURE 6.

Allogeneic adipose‐derived stem cells (ASCs) secrete prohematopoietic factors to support the recovery of damaged bone marrow cells. A‐L, Luminex assay readout of the factors released by monoculture and cocultured irradiated and nonirradiated bone marrow cells with ASCs (n = 3). The results are presented as mean ± SD; *P < .05
4. DISCUSSION
ARS is radiation poisoning caused by TBI during radiation accidents. Radiation exposure causes damage to the hematopoietic, gastrointestinal, and nervous systems. High doses of radiation can cause irreparable damage to the bone marrow, affecting the immune system and potentially causing inflammation and infection. Currently, there are no effective therapies available, and there is an urgent need to add effective therapies in the national stockpile.
We investigated an allogeneic cell therapy approach, and our results show that intraperitoneally injected allogeneic ASCs migrate to the irradiated bone marrow and mitigate ARS by extending the survival of TBI mice. Similarly, in our transwell assays, we observed that irradiated bone marrow promoted ASCs migration which improved bone marrow survival ex vivo. We used 9.25 Gy for C57BL/6 and 8.75 Gy irradiation dose for FVB mice for the TBI model. The hematopoietic system is sensitive to ionizing radiation, which causes irreversible damage to the bone marrow. Therefore, regeneration of damaged bone marrow using allogeneic ASCs may increase the survival rate of ARS victims.
Bone marrow serves as a pool for the blood‐forming hematopoietic stem cells, which are supported by other nonhematopoietic cells in the bone marrow niche, mainly mesenchymal stem cells, endothelial cells, pericytes, and stromal cells. Secretory factors such as cytokines and growth factors from the cellular compartment of the bone marrow niche are known to regulate hematopoietic stem cell proliferation and differentiation. 42 , 43 It is well established that bone marrow populations are vulnerable to radiation exposure resulting in extreme impairment to bone marrow populations. Still, the severity of even sublethal doses can lead to irreversible tissue damage of the bone marrow microenvironment. 44 , 45 Hence, bone marrow retrieval might be crucial to repair an injury from ARS. Several studies have proposed that using cytokines, growth factors, drugs, and stem cells can mitigate the damage caused to bone marrow. 10 Still, the clinical use of the above therapies for mass applications finds limitations.
Previous studies have shown that ASCs support hematopoietic stem cell expansion and maintenance. 30 , 46 Yet, the mechanism of action of ASCs' therapeutic effect is unknown. Therefore, in our current study, we explored whether treatment with ASCs could mitigate radiation‐induced bone marrow failure and the mechanisms associated with the rescue of damaged bone marrow.
Various publications demonstrated that mesenchymal stem cells derived from bone marrow, Wharton jelly, and placenta are effective in mitigating ARS. 10 , 13 , 14 , 15 Still, the problems associated with tissue availability, cell harvesting, and propagation limit mass applications. Moreover, autologous stromal vascular fraction from adipose tissue was shown to reconstitute the bone marrow in TBI mice, but this approach has limitations due to the autologous nature of the cells, which is not feasible in accidental exposure scenarios. 35 In this context, we found that allogeneic ASCs protected the mice and improved the survival percentage from ARS similar to autologous ASCs. Our studies suggest that the easily accessible and abundant source of subcutaneous derived ASCs is an attractive candidate for allogeneic transplantation. They find attraction because of their numerous characteristic features, including being easy to harvest, high in vitro expansion, ease of maintenance, no or low immunogenicity, and higher differentiation potential. 22
Migration and homing of transplanted cells to the site of injury are vital for tissue regeneration. Most of the cell therapies failed due to impairment in their movement and homing abilities. Besides, unfavorable conditions such as oxidative stress, hypoxia, and inflammation show adverse effects on survival. Moreover, the efficacy and functionality of transplanted cells depend on the method of injection and the way it is administered. Previous reports demonstrated that intravenous infusion of stem cells led to their entrapment in other tissues, mainly in the lungs, resulted in a poor outcome. 35 , 47 , 48 Intraperitoneal injected allogeneic ASCs migrated to the irradiated bone marrow, improving the mice's survival from radiation damage. Our result suggested that immune‐modulatory, anti‐inflammatory, and antioxidant properties of allogeneic ASCs can overcome unfavorable conditions. Consistent with our studies, earlier reports also found that the migration of transplanted cells increased the functionality of damaged bone marrow. 49 , 50
Bone marrow exposed to radiation induces expression of proinflammatory molecules TNF‐α, growth factors EGF, pro‐apoptotic markers, and free radicals. 51 , 52 Inflammatory molecules serve as a chemoattractant and enhance the migration of hematopoietic and mesenchymal stem cells to the injury site for regeneration. 53 , 54 Our data also show that ASCs were attracted and migrated to the irradiated bone marrow through transwell inserts. In contrast, few ASCs got relocated to the nonirradiated bone marrow. Besides, we observed that migrated ASCs promoted the survival of irradiated bone marrow cells. Our results indicate that ASCs could act as an antiapoptotic agent, downregulate the expression levels of proapoptotic molecules, and prevent cell death. Cell to cell interaction is vital for the maintenance of the hematopoietic system in the bone marrow niche. ASCs have been shown to protect bone marrow cells from radiation damage and support the hematopoietic activity in vitro and in vivo. 46 Similarly, our studies demonstrated cobblestone formation and improved survival of radiation‐exposed cells during contact coculture and robust transmigration.
The bone marrow niche represents a complex microenvironment. Cellular and noncellular compartments control the self‐renewal and differentiation of hematopoietic stem cells. Soluble factors such as cytokines, growth factors, and chemokines regulate stem cell behavior through an autocrine, paracrine, and juxtacrine signaling mechanism. Soluble factors act as a messenger and are essential for the establishment of a communicational network between the cells. 55 Our transwell experiments have shown that activation of ASCs with the inflammatory environment from irradiated bone marrow significantly increased the secretion of prohematopoiesis and prosurvival cytokines. They include GM‐CSF, MIP1α, MIP1β, LIX, KC, 1P‐10, Rantes, IL‐17, MCSF, TNFα, Eotaxin, and IP‐10 which supported the cobblestone formation and improved the survival of bone marrow cells from radiation damage. Similarly, more recently, it was reported that ARS‐related stress signals activated placental stem cells, and the secretome of activated placental cells rescue the mice from ARS. 56
Our data strongly supported several documented reports and relate to the functional role of soluble factors in various stages of blood cell development by a direct or indirect mechanism. GM‐CSF is an FDA‐approved cytokine regime for blood cell development and immunomodulation. Apart from its role in the stimulation of granulocyte and macrophage differentiation, it also acts as a radioprotector and reduces the damage caused by radiation. 57 MIP‐1α increases the production of peripheral blood by inhibiting proliferation and promotes the differentiation of hematopoietic stem cells. Moreover, MIP‐1α acts as an anti‐inflammatory agent to reduce inflammation. 58 LIX is a chemokine essential for the maintenance of long‐term cultures. It preserves the quiescent state of hematopoietic stem cells and maintains stem cell‐enriched populations in the bone marrow. 40 LIX also supports the angiogenesis by neovascularization. 59 Interleukin‐10 (IL‐10) is an immunosuppressive cytokine and regulates the self‐renewal capacity of hematopoietic stem cells. 60 Apart from maintaining long term cultures of hematopoietic stem cells, 1L‐10 also acts as an anti‐inflammatory cytokine and resolves tissue from inflammation. 61 Interleukin 17 is a proinflammatory cytokine and supports the regeneration of hematopoietic progenitor cells and improves the survival of radiation damage. 62 , 63 Also, it promotes angiogenesis for neovascularization for injured tissue. 64 Interleukin 1α regulates the proliferation of hematopoietic stem cells and stimulates the growth factors required for hematopoiesis. 65 , 66 Moreover, IL‐1 acts as a radioprotective agent and restores the function of bone marrow from radiation damage. 67 MCSF is an inflammatory cytokine and inhibitor of stem cells and promotes maturation of hematopoietic progenitor cells. MCSF expresses high levels and stimulates the expression of other inflammatory molecules during stress or inflammation. 68 Angiogenesis is essential for blood circulation and communication between tissues and cells. Exotoxin is a hematopoietic stem cell‐derived proangiogenic factor necessary for neovascularization and reduces the inflammation. 69 , 70
ASCs conditioned media has been used in various therapeutic applications such as wound healing, aging, inflammation, and regeneration. 71 , 72 , 73 , 74 ASCs secrete several paracrine factors such as VEGF, G‐CSF, GM‐CSF, TGF‐β, MIP1α, MIP1β, MCP2, and Eotaxin. Although conditioned medium from bone marrow‐derived mesenchymal stem cells and ASCs treated various degenerative diseases, the therapeutic application for ARS is limited due to lower factors concentrations, which results in a poor outcome. Recent study has shown successful mitigation of TBI mice using purified extracellular vesicles (EVs) derived from MSCs. 75 ASCs are well known to secrete EVs having regenerative potential. 76 The most likely reason that conditioned media failed to mitigate in our study is due to the lower concentration of growth factors and EVs in culture media.
Cellular therapies have the potential to mitigate both acute and late complications from radiation exposure. 77 Critical challenges for adapting cellular therapies for mass population applications in real‐life scenarios include mass production of cellular product, quality control, storage, stability, logistics, and injection to affected individuals. Adipose tissue is a readily available source in bulk quantities and ASCs show robust expansion capabilities in cell culture that enables mass production of these cells. The in vitro assays for characterization of these cells based on phenotype marker expression are very well established. Previous studies have shown feasibility of storing these cells up to 10 years in liquid nitrogen without loss of functionality. 78 Adaptation of cell therapies as a radiation countermeasure require development of bomb and radiation proof cell banks in different parts of the country that enables transportation of stored cells in dry ice to the site of accident within 10 to 15 hours and delivery to affected individuals within 24 hours of the exposure. Further studies investigating different cell numbers, frequency of injection, and anatomical sites will guide real‐world adaptation.
In conclusion, our study demonstrated a proof‐of‐concept allogeneic stem cell therapy and the mitigation mechanism of ARS using ASCs, which are readily available in abundance and can be easily propagated to sufficient quantities for national stockpile storage. We show that the mitigation is dependent on ASCs migration to damaged bone marrow and is positively correlated to the survival of damaged cells. The migration of ASCs is dependent on signals generated from damaged bone marrow cells. Mechanistically, ASCs secrete hematopoietic and prosurvival factors that rescue irradiated damaged bone marrow cells.
CONFLICT OF INTEREST
The authors declared no potential conflicts of interest.
AUTHOR CONTRIBUTIONS
S.C.: collection and assembly of data, data analysis and interpretation, manuscript writing; K.S.Y., Y.S., W.H.: collection of data; M.W.E.: collection and assembly of data, data analysis and interpretation; J.S.G.: conception and design, data interpretation, manuscript writing; A.E.: conception and design, collection and assembly of data, data analysis and interpretation, manuscript writing and final approval; P.J.R.: conception and design, data analysis and interpretation.
Supporting information
Supplementary Figure 1 ASCs migrate through the transwell and promote survival
Morphology of migrated ASCs to the irradiated bone marrow. Scale bars: 100 μm. Magnification 10x.
Supplementary Figure 2: ASCs secrete pro‐hematopoietic factors to support the recovery of damaged bone marrow cells. (A‐T) Luminex assay readout of the factors released by monoculture and co‐cultured irradiated and non‐irradiated bone marrow cells with ASCs (n = 3). The results are presented as mean ± SD; *P < 0.05.
Supplementary Table 1: Characterization of C57BL/6 (Syngenetic) compared to FVB (Allogeneic) stem cells
Supplementary Table 2: Change in the expression pattern of cytokines in ASCs‐irradiated bone marrow cells compared to ASCs‐non‐irradiated bone marrow cells co‐culture
ACKNOWLEDGMENTS
This study was supported by CMCR NIAID/NIH Grant U19A168021, NCI/NIH Grant R01‐CA114246‐08A1, PSF/MTF grant and DOD Grant W81XWH‐19‐PRMRP‐DA.
Chinnapaka S, Yang KS, Samadi Y, et al. Allogeneic adipose‐derived stem cells mitigate acute radiation syndrome by the rescue of damaged bone marrow cells from apoptosis. STEM CELLS Transl Med. 2021;10:1095–1114. 10.1002/sctm.20-0455
Funding information DOD, Grant/Award Number: W81XWH‐19‐PRMRP‐DA; PSF/MTF; NCI/NIH, Grant/Award Number: R01‐CA114246‐08A1; CMCR NIAID/NIH, Grant/Award Number: U19A168021
Contributor Information
Asim Ejaz, Email: ejaza@upmc.edu.
J. Peter Rubin, Email: rubinjp@upmc.edu.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Donnelly EH, Nemhauser JB, Smith JM, et al. Acute radiation syndrome: assessment and management. South Med J. 2010;103(6):541‐546. [DOI] [PubMed] [Google Scholar]
- 2. Gaberman E, Pinzur L, Levdansky L, et al. Mitigation of lethal radiation syndrome in mice by intramuscular injection of 3D cultured adherent human placental stromal cells. PLoS One. 2013;8(6):e66549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Baker JE, Moulder JE, Hopewell JW. Radiation as a risk factor for cardiovascular disease. Antioxid Redox Signal. 2011;15(7):1945‐1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Pan CC, Kavanagh BD, Dawson LA, et al. Radiation‐associated liver injury. Int J Radiat Oncol Biol Phys. 2010;76(3):S94‐S100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Dawson LA, Kavanagh BD, Paulino AC, et al. Radiation‐associated kidney injury. Int J Radiat Oncol Biol Phys. 2010;76(3):S108‐S115. [DOI] [PubMed] [Google Scholar]
- 6. Williams JP, McBride WH. After the bomb drops: a new look at radiation‐induced multiple organ dysfunction syndrome (MODS). Int J Radiat Biol. 2011;87(8):851‐868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Meineke V, Fliedner T. Radiation‐induced multi‐organ involvement and failure: challenges for radiation accident medical management and future research. Br J Radiol. 2005;1:196‐200. [Google Scholar]
- 8. Peters MA. The Threat of Nuclear War: Peace Studies in an Apocalyptic Age. London: Taylor & Francis; 2019. [Google Scholar]
- 9. Andersson KG, Mikkelsen T, Astrup P, et al. Estimation of health hazards resulting from a radiological terrorist attack in a city. Radiat Prot Dosimetry. 2008;131(3):297‐307. [DOI] [PubMed] [Google Scholar]
- 10. Singh VK, Romaine PL, Seed TM. Medical countermeasures for radiation exposure and related injuries: characterization of medicines, FDA‐approval status and inclusion into the strategic national stockpile. Health Phys. 2015;108(6):607‐630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Wills C, Cherian S, Yousef J, Wang K, Mackley HB. Total body irradiation: a practical review. Appl Radiat Oncol. 2016;5:11‐17. [Google Scholar]
- 12. Dropcho EJ. Central nervous system injury by therapeutic irradiation. Neurol Clin. 1991;9(4):969‐988. [PubMed] [Google Scholar]
- 13. Gale RP, Armitage JO. Use of molecularly‐cloned haematopoietic growth factors in persons exposed to acute high‐dose, high‐dose rate whole‐body ionizing radiations. Blood Rev. 2020;45:100690. [DOI] [PubMed] [Google Scholar]
- 14. Hofer M, Pospíšil M, Komůrková D, Hoferová Z. Granulocyte colony‐stimulating factor in the treatment of acute radiation syndrome: a concise review. Molecules. 2014;19(4):4770‐4778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Bertho J‐M, Frick J, Prat M, et al. Comparison of autologous cell therapy and granulocyte‐colony stimulating factor (G‐CSF) injection vs. G‐CSF injection alone for the treatment of acute radiation syndrome in a non‐human primate model. Int J Radiat Oncol Biol Phys. 2005;63(3):911‐920. [DOI] [PubMed] [Google Scholar]
- 16. Aghamir SMR, Mehrabani D, Amini M, et al. The regenerative effect of bone marrow‐derived stem cells on cell count and survival in acute radiation syndrome. World J Plast Surg. 2017;6(1):111‐113. [PMC free article] [PubMed] [Google Scholar]
- 17. Mortazavi SMJ, Shekoohi‐Shooli F, Aghamir SMR, et al. The healing effect of bone marrow‐derived stem cells in acute radiation syndrome. Pak J Med Sci. 2016;32(3):646‐651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Gan J, Meng F, Zhou X, et al. Hematopoietic recovery of acute radiation syndrome by human superoxide dismutase–expressing umbilical cord mesenchymal stromal cells. Cytotherapy. 2015;17(4):403‐417. [DOI] [PubMed] [Google Scholar]
- 19. Shim S, Lee SB, Lee J‐G, et al. Mitigating effects of hUCB‐MSCs on the hematopoietic syndrome resulting from total body irradiation. Exp Hematol. 2013;41(4):346‐353.e342. [DOI] [PubMed] [Google Scholar]
- 20. Bandekar M, Maurya DK, Sharma D, et al. Xenogeneic transplantation of human WJ‐MSCs rescues mice from acute radiation syndrome via Nrf2 dependent regeneration of damaged tissues. Am J Transplant. 2020;20:2044‐2057. [DOI] [PubMed] [Google Scholar]
- 21. Zuk PA, Zhu M, Ashjian P, et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13(12):4279‐4295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Ejaz A, Greenberger JS, Rubin PJ. Understanding the mechanism of radiation induced fibrosis and therapy options. Pharmacol Ther. 2019;204:107399. [DOI] [PubMed] [Google Scholar]
- 23. Han J, Koh YJ, Moon HR, et al. Adipose tissue is an extramedullary reservoir for functional hematopoietic stem and progenitor cells. Blood. 2010;115(5):957‐964. [DOI] [PubMed] [Google Scholar]
- 24. Yoshimura K, Shigeura T, Matsumoto D, et al. Characterization of freshly isolated and cultured cells derived from the fatty and fluid portions of liposuction aspirates. J Cell Physiol. 2006;208(1):64‐76. [DOI] [PubMed] [Google Scholar]
- 25. Mitterberger MC, Mattesich M, Zwerschke W. Bariatric surgery and diet‐induced long‐term caloric restriction protect subcutaneous adipose‐derived stromal/progenitor cells and prolong their life span in formerly obese humans. Exp Gerontol. 2014;56:106‐113. [DOI] [PubMed] [Google Scholar]
- 26. Hörl S, Ejaz A, Ernst S, et al. CD146 (MCAM) in human cs‐DLK1−/cs‐CD34+ adipose stromal/progenitor cells. Stem Cell Res. 2017;22:1‐12. [DOI] [PubMed] [Google Scholar]
- 27. Zwierzina ME, Ejaz A, Bitsche M, et al. Characterization of DLK1 (PREF1)+/CD34+ cells in vascular stroma of human white adipose tissue. Stem Cell Res. 2015;15(2):403‐418. [DOI] [PubMed] [Google Scholar]
- 28. Ejaz A, Epperly MW, Hou W, Greenberger JS, Rubin JP. Adipose‐derived stem cell therapy ameliorates ionizing irradiation fibrosis via hepatocyte growth factor‐mediated transforming growth factor‐β downregulation and recruitment of bone marrow cells. Stem Cells. 2019;37(6):791‐802. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Cousin B, André M, Arnaud E, Pénicaud L, Casteilla L. Reconstitution of lethally irradiated mice by cells isolated from adipose tissue. Biochem Biophys Res Commun. 2003;301(4):1016‐1022. [DOI] [PubMed] [Google Scholar]
- 30. Corre J, Barreau C, Cousin B, et al. Human subcutaneous adipose cells support complete differentiation but not self‐renewal of hematopoietic progenitors. J Cell Physiol. 2006;208(2):282‐288. [DOI] [PubMed] [Google Scholar]
- 31. Steinman J, Epperly M, Hou W, et al. Improved total‐body irradiation survival by delivery of two radiation mitigators that target distinct cell death pathways. Radiat Res. 2018;189(1):68‐83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Banki Z, Posch W, Ejaz A, et al. Complement as an endogenous adjuvant for dendritic cell‐mediated induction of retrovirus‐specific CTLs. PLoS Pathog. 2010;6(4):e1000891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Ejaz A, Ammann CG, Werner R, et al. Targeting viral antigens to CD11c on dendritic cells induces retrovirus‐specific T cell responses. PLoS One. 2012;7(9):e45102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Berry R, Rodeheffer MS. Characterization of the adipocyte cellular lineage in vivo. Nat Cell Biol. 2013;15(3):302‐308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Cousin B, Andre M, Arnaud E, Penicaud L, Casteilla L. Reconstitution of lethally irradiated mice by cells isolated from adipose tissue. Biochem Biophys Res Commun. 2003;301(4):1016‐1022. [DOI] [PubMed] [Google Scholar]
- 36. Kilroy GE, Foster SJ, Wu X, et al. Cytokine profile of human adipose‐derived stem cells: expression of angiogenic, hematopoietic, and pro‐inflammatory factors. J Cell Physiol. 2007;212(3):702‐709. [DOI] [PubMed] [Google Scholar]
- 37. Robles H, Park S, Joens MS, Fitzpatrick JAJ, Craft CS, Scheller EL. Characterization of the bone marrow adipocyte niche with three‐dimensional electron microscopy. Bone. 2019;118:89‐98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Sun L, Rautela J, Delconte RB, et al. GM‐CSF quantity has a selective effect on granulocytic vs. monocytic myeloid development and function. Front Immunol. 2018;9:1922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Staversky RJ, Byun DK, Georger MA, et al. The chemokine CCL3 regulates myeloid differentiation and hematopoietic stem cell numbers. Sci Rep. 2018;8(1):1‐9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Choong ML, Yong YP, Tan AC, Luo B, Lodish HF. LIX: a chemokine with a role in hematopoietic stem cells maintenance. Cytokine. 2004;25(6):239‐245. [DOI] [PubMed] [Google Scholar]
- 41. Sidahmed AM, León AJ, Bosinger SE, et al. CXCL10 contributes to p38‐mediated apoptosis in primary T lymphocytes in vitro. Cytokine. 2012;59(2):433‐441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Prockop DJ. Marrow stromal cells as stem cells for nonhematopoietic tissues. Science. 1997;276(5309):71‐74. [DOI] [PubMed] [Google Scholar]
- 43. Cluitmans FH, Esendam BH, Veenhof WF, Landegent JE, Willemze R, Falkenburg JH. The role of cytokines and hematopoietic growth factors in the autocrine/paracrine regulation of inducible hematopoiesis. Ann Hematol. 1997;75(1–2):27‐31. [DOI] [PubMed] [Google Scholar]
- 44. Green DE, Rubin CT. Consequences of irradiation on bone and marrow phenotypes, and its relation to disruption of hematopoietic precursors. Bone. 2014;63:87‐94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Till JE, Mc CE. A direct measurement of the radiation sensitivity of normal mouse bone marrow cells. Radiat Res. 1961;14:213‐222. [PubMed] [Google Scholar]
- 46. De Toni F, Poglio S, Youcef AB, et al. Human adipose‐derived stromal cells efficiently support hematopoiesis in vitro and in vivo: a key step for therapeutic studies. Stem Cells Dev. 2011;20(12):2127‐2138. [DOI] [PubMed] [Google Scholar]
- 47. Eggenhofer E, Benseler V, Kroemer A, et al. Mesenchymal stem cells are short‐lived and do not migrate beyond the lungs after intravenous infusion. Front Immunol. 2012;3:297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Eggenhofer E, Luk F, Dahlke MH, Hoogduijn MJ. The life and fate of mesenchymal stem cells. Front Immunol. 2014;5:148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Wynn RF, Hart CA, Corradi‐Perini C, et al. A small proportion of mesenchymal stem cells strongly expresses functionally active CXCR4 receptor capable of promoting migration to bone marrow. Blood. 2004;104(9):2643‐2645. [DOI] [PubMed] [Google Scholar]
- 50. Zong Z‐W, Cheng T‐M, Su Y‐P, et al. Crucial role of SDF‐1/CXCR4 interaction in the recruitment of transplanted dermal multipotent cells to sublethally irradiated bone marrow. J Radiat Res. 2006;47(3–4):287‐293. [DOI] [PubMed] [Google Scholar]
- 51. Schaue D, Kachikwu EL, McBride WH. Cytokines in radiobiological responses: a review. Radiat Res. 2012;178(6):505‐523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Hallahan DE, Spriggs DR, Beckett MA, Kufe DW, Weichselbaum RR. Increased tumor necrosis factor alpha mRNA after cellular exposure to ionizing radiation. Proc Natl Acad Sci USA. 1989;86(24):10104‐10107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Ponte AL, Marais E, Gallay N, et al. The in vitro migration capacity of human bone marrow mesenchymal stem cells: comparison of chemokine and growth factor chemotactic activities. Stem Cells. 2007;25(7):1737‐1745. [DOI] [PubMed] [Google Scholar]
- 54. Ji JF, He BP, Dheen ST, Tay SS. Interactions of chemokines and chemokine receptors mediate the migration of mesenchymal stem cells to the impaired site in the brain after hypoglossal nerve injury. Stem Cells. 2004;22(3):415‐427. [DOI] [PubMed] [Google Scholar]
- 55. Zhao Y, Zhan Y, Burke KA, Anderson WF. Soluble factor(s) from bone marrow cells can rescue lethally irradiated mice by protecting endogenous hematopoietic stem cells. Exp Hematol. 2005;33(4):428‐434. [DOI] [PubMed] [Google Scholar]
- 56. Pinzur L, Akyuez L, Levdansky L, et al. Rescue from lethal acute radiation syndrome (ARS) with severe weight loss by secretome of intramuscularly injected human placental stromal cells. J Cachexia Sarcopenia Muscle. 2018;9(6):1079‐1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Patni N, Patni S, Bapna A. The optimal use of granulocyte macrophage colony stimulating factor in radiation induced mucositis in head and neck squamous cell carcinoma. J Cancer Res Ther. 2005;1(3):136‐141. [DOI] [PubMed] [Google Scholar]
- 58. Cook DN. The role of MIP‐1 alpha in inflammation and hematopoiesis. J Leukoc Biol. 1996;59(1):61‐66. [DOI] [PubMed] [Google Scholar]
- 59. Addison CL, Daniel TO, Burdick MD, et al. The CXC chemokine receptor 2, CXCR2, is the putative receptor for ELR+ CXC chemokine‐induced angiogenic activity. J Immunol. 2000;165(9):5269‐5277. [DOI] [PubMed] [Google Scholar]
- 60. Burmeister AR, Marriott I. The interleukin‐10 family of cytokines and their role in the CNS. Front Cell Neurosci. 2018;12:458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Kang YJ, Yang SJ, Park G, et al. A novel function of interleukin‐10 promoting self‐renewal of hematopoietic stem cells. Stem Cells. 2007;25(7):1814‐1822. [DOI] [PubMed] [Google Scholar]
- 62. Mojsilovic S, Jaukovic A, Santibanez JF, Bugarski D. Interleukin‐17 and its implication in the regulation of differentiation and function of hematopoietic and mesenchymal stem cells. Mediators Inflamm. 2015;2015:470458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Piryani SO, Kam AYF, Vu UT, Chao NJ, Doan PL. CCR5 signaling promotes murine and human hematopoietic regeneration following ionizing radiation. Stem Cell Rep. 2019;13(1):76‐90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Liu GT, Huang YL, Tzeng HE, Tsai CH, Wang SW, Tang CH. CCL5 promotes vascular endothelial growth factor expression and induces angiogenesis by down‐regulating miR‐199a in human chondrosarcoma cells. Cancer Lett. 2015;357(2):476‐487. [DOI] [PubMed] [Google Scholar]
- 65. Hestdal K, Jacobsen SE, Ruscetti FW, et al. In vivo effect of interleukin‐1 alpha on hematopoiesis: role of colony‐stimulating factor receptor modulation. Blood. 1992;80(10):2486‐2494. [PubMed] [Google Scholar]
- 66. Orelio C, Peeters M, Haak E, van der Horn K, Dzierzak E. Interleukin‐1 regulates hematopoietic progenitor and stem cells in the midgestation mouse fetal liver. Haematologica. 2009;94(4):462‐469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Neta R, Douches S, Oppenheim JJ. Interleukin 1 is a radioprotector. J Immunol. 1986;136(7):2483‐2485. [PubMed] [Google Scholar]
- 68. Kremlev SG, Chapoval AI, Evans R. CSF‐1 (M‐CSF) enhances the inflammatory response of fibronectin‐primed macrophages: pathways involved in activation of the cytokine network. Nat Immun. 1998;16(5–6):228‐243. [DOI] [PubMed] [Google Scholar]
- 69. Rose JA, Erzurum S, Asosingh K. Biology and flow cytometry of proangiogenic hematopoietic progenitors cells. Cytometry A. 2015;87(1):5‐19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Asosingh K, Vasanji A, Tipton A, et al. Eotaxin‐rich proangiogenic hematopoietic progenitor cells and CCR3+ endothelium in the atopic asthmatic response. J Immunol. 2016;196(5):2377‐2387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Zhou BR, Xu Y, Guo SL, et al. The effect of conditioned media of adipose‐derived stem cells on wound healing after ablative fractional carbon dioxide laser resurfacing. Biomed Res Int. 2013;2013:519126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Platas J, Guillen MI, del Caz MD, Gomar F, Mirabet V, Alcaraz MJ. Conditioned media from adipose‐tissue‐derived mesenchymal stem cells downregulate degradative mediators induced by interleukin‐1beta in osteoarthritic chondrocytes. Mediators Inflamm. 2013;2013:357014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Jha KA, Pentecost M, Lenin R, et al. Concentrated conditioned media from adipose tissue derived mesenchymal stem cells mitigates visual deficits and retinal inflammation following mild traumatic brain injury. Int J Mol Sci. 2018;19(7). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Li L, Ngo HTT, Hwang E, et al. Conditioned medium from human adipose‐derived mesenchymal stem cell culture prevents UVB‐induced skin aging in human keratinocytes and dermal fibroblasts. Int J Mol Sci. 2019;21(1). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Accarie A, l'Homme B, Benadjaoud MA, et al. Extracellular vesicles derived from mesenchymal stromal cells mitigate intestinal toxicity in a mouse model of acute radiation syndrome. Stem Cell Res Ther. 2020;11(1):1‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Figliolini F, Ranghino A, Grange C, et al. Extracellular vesicles from adipose stem cells prevent muscle damage and inflammation in a mouse model of hind limb ischemia: role of Neuregulin‐1. Arterioscler Thromb Vasc Biol. 2020;40(1):239‐254. [DOI] [PubMed] [Google Scholar]
- 77. DiCarlo AL, Tamarat R, Rios CI, et al. Cellular therapies for treatment of radiation injury: report from a NIH/NIAID and IRSN workshop. Radiat Res. 2017;188(2):e54‐e75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Shaik S, Wu X, Gimble J, Devireddy R. Effects of decade long freezing storage on adipose derived stem cells functionality. Sci Rep. 2018;8(1):1‐12. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Figure 1 ASCs migrate through the transwell and promote survival
Morphology of migrated ASCs to the irradiated bone marrow. Scale bars: 100 μm. Magnification 10x.
Supplementary Figure 2: ASCs secrete pro‐hematopoietic factors to support the recovery of damaged bone marrow cells. (A‐T) Luminex assay readout of the factors released by monoculture and co‐cultured irradiated and non‐irradiated bone marrow cells with ASCs (n = 3). The results are presented as mean ± SD; *P < 0.05.
Supplementary Table 1: Characterization of C57BL/6 (Syngenetic) compared to FVB (Allogeneic) stem cells
Supplementary Table 2: Change in the expression pattern of cytokines in ASCs‐irradiated bone marrow cells compared to ASCs‐non‐irradiated bone marrow cells co‐culture
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


