Supplemental Digital Content is available in the text.
Keywords: coronavirus, echocardiography, embolism, intensive care units, ischemic stroke
Abstract
Background and Purpose:
Stroke may complicate coronavirus disease 2019 (COVID-19) infection based on clinical hypercoagulability. We investigated whether transcranial Doppler ultrasound has utility for identifying microemboli and clinically relevant cerebral blood flow velocities (CBFVs) in COVID-19.
Methods:
We performed transcranial Doppler for a consecutive series of patients with confirmed or suspected COVID-19 infection admitted to 2 intensive care units at a large academic center including evaluation for microembolic signals. Variables specific to hypercoagulability and blood flow including transthoracic echocardiography were analyzed as a part of routine care.
Results:
Twenty-six patients were included in this analysis, 16 with confirmed COVID-19 infection. Of those, 2 had acute ischemic stroke secondary to large vessel occlusion. Ten non-COVID stroke patients were included for comparison. Two COVID-negative patients had severe acute respiratory distress syndrome and stroke due to large vessel occlusion. In patients with COVID-19, relatively low CBFVs were observed diffusely at median hospital day 4 (interquartile range, 3–9) despite low hematocrit (29.5% [25.7%–31.6%]); CBFVs in comparable COVID-negative stroke patients were significantly higher compared with COVID-positive stroke patients. Microembolic signals were not detected in any patient. Median left ventricular ejection fraction was 60% (interquartile range, 60%–65%). CBFVs were correlated with arterial oxygen content, and C-reactive protein (Spearman ρ=0.28 [P=0.04]; 0.58 [P<0.001], respectively) but not with left ventricular ejection fraction (ρ=−0.18; P=0.42).
Conclusions:
In this cohort of critically ill patients with COVID-19 infection, we observed lower than expected CBFVs in setting of low arterial oxygen content and low hematocrit but not associated with suppression of cardiac output.
Encephalopathy and ischemic stroke, especially large vessel occlusion, are reported neurological complications of coronavirus disease 2019 (COVID-19) infection.1,2 Hypothesized mechanisms include systemic hypercoagulability, vascular inflammation, and microvascular thrombosis.3
Transcranial Doppler ultrasound (TCD) is a portable, noninvasive technique that can detect circulating emboli and evaluate for cardiac embolism or vascular stenosis as potential stroke mechanisms.4 This study investigated occurrence of microemboli and cerebral blood flow velocities (CBFVs) using TCD in patients with and without COVID-19 infection.
Methods
We prospectively evaluated patients with confirmed and suspected COVID-19 infection hospitalized in 2 critical care units in an urban health system between April 4 and May 22, 2020, who underwent a TCD exam as standard of care for severe encephalopathy and suspicion of cerebral emboli. COVID-19 infection was confirmed using reverse transcription polymerase chain reaction. The study was approved by the Johns Hopkins Medicine institutional review board. Patient consent was waived. Data are available upon reasonable request and completion of a data use agreement at http://www.braininjuryoutcomes.com/clear-about.
All patients underwent abbreviated 15-minute manual bilateral TCD monitoring to detect microembolic signals using a 7 dB threshold (Compumedics DWL, San Juan Capistrano, CA). The middle cerebral artery (MCA) was insonated bilaterally using a low-frequency probe (2 mHz), following a validated protocol.5 All examinations were performed by a single experienced neurosonographer (B. Ergin) with the patient in supine position.
Patient characteristics and laboratory data were collected at admission and on day of TCD (±48 hours). Transthoracic echocardiography obtained closest to TCD were analyzed for markers of diastolic function and global longitudinal strain. Arterial oxygen content (CaO2) was calculated from values of hemoglobin and fractional oxygen saturation at time of TCD, using the formula: CaO2 (mL/100 mL)=hemoglobin (mmol/L)×(1.34 mL O2/g hemoglobin)×[(1.61 g hemoglobin/100 mL)/(mmol hemoglobin/L)]×saturation (%/100).
Data are reported as medians and interquartile range. We used Spearman ρ to correlate mean CBFVs in the MCA with markers of viscosity, inflammation, dehydration, and cardiac output: hematocrit, D-dimer, fibrinogen, CRP (C-reactive protein), blood urea nitrogen:creatinine ratio, left ventricular ejection fraction (LVEF), partial pressure of carbon dioxide (PaCO2), and CaO2. Two-sample t test compared differences in means. Statistical significance was determined by P<0.05. Statistical analysis was performed with Stata Version 14 (STATA Corp, College Station, TX).
Results
Twenty-six patients were enrolled after undergoing TCD, 16 with COVID-19 pneumonia (Table 1). The other 10 patients had AIS and had negative reverse transcription polymerase chain reaction for COVID-19; of these, 2 had large vessel occlusion and severe acute respiratory distress syndrome.
Table 1.
Baseline and Laboratory Characteristics at Admission
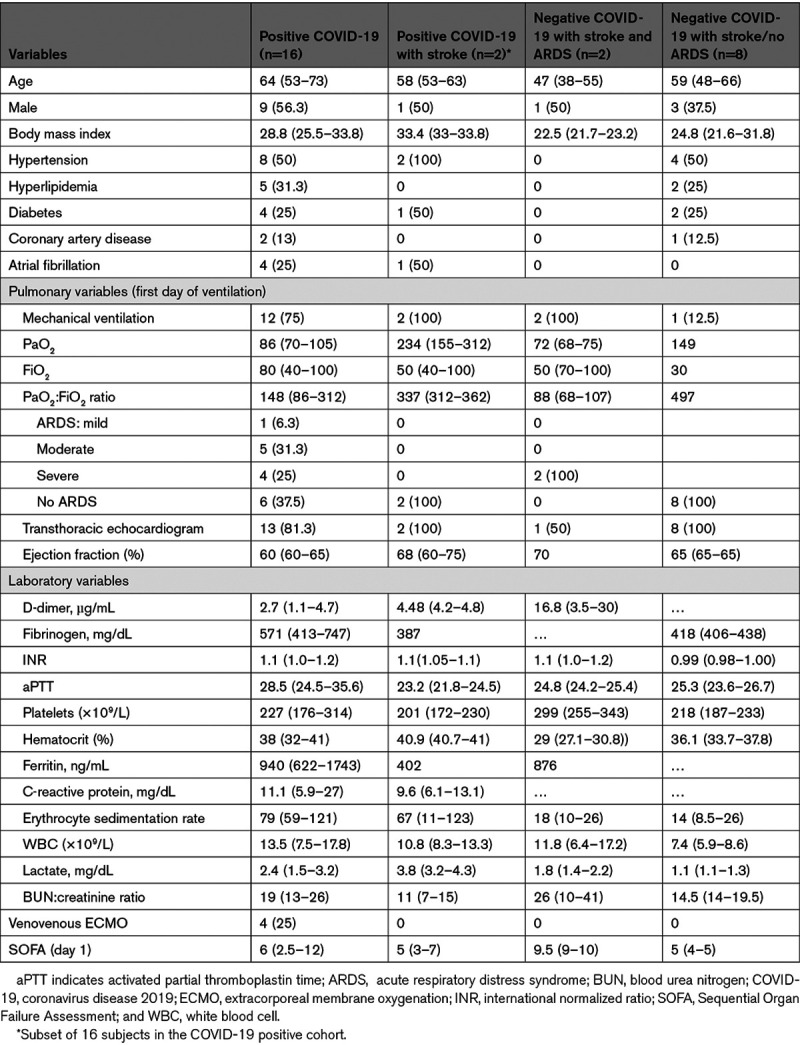
TCD was performed at median 4 (2–5) days after admission. At time of TCD, all patients were afebrile; hematocrit was below normal in all but 4 patients (Table 2). In COVID-19 positive patients CRP, D-dimer, and fibrinogen (except one patient) were all elevated (Table I in the Data Supplement). Median CBFVs were at lower limit of normal relative to age-adjusted normative values in COVID-positive patients and lower relative to all COVID-negative stroke patients including 2 with acute respiratory distress syndrome (Figure). Microembolic signals were not detected in any patient. Mean CBFVs in the M1 division of the MCA were significantly correlated with CaO2 (Spearman ρ=0.28; P=0.04) and CRP (ρ=0.58; P<0.001). Weak inverse correlations were found with blood urea nitrogen:creatinine ratio (ρ=−0.26; P=0.06) and PaCO2 (ρ=−0.31; P=0.09) but not with other variables tested.
Table 2.
Clinical Characteristics at Time of TCD
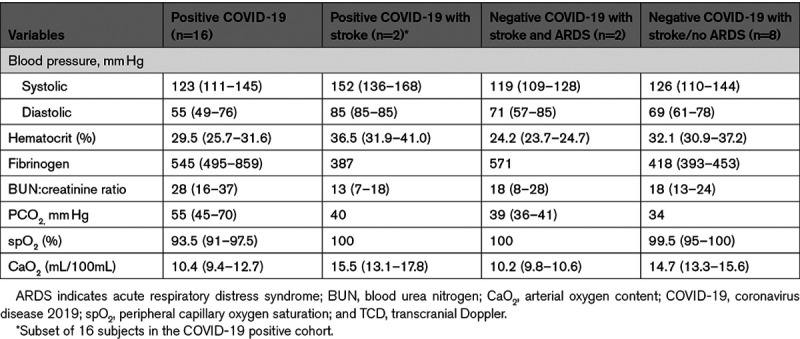
Figure.
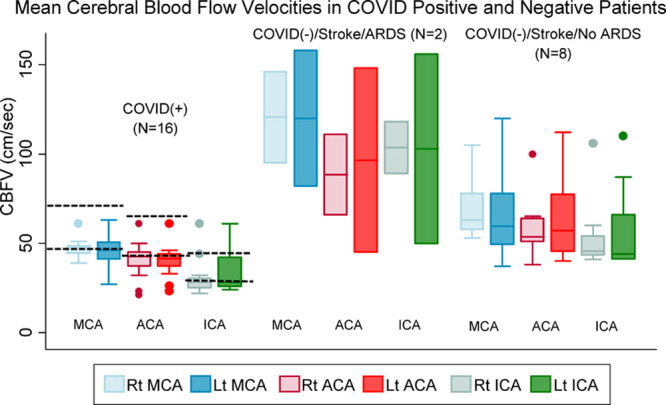
Mean cerebral blood flow velocities (CBFVs) in patients with and without coronavirus disease 2019 (COVID-19) infection. Dashed lines indicate normative CBFV range for age group.
Twenty-two patients had transthoracic echocardiography with median LVEF of 60%. There were no significant differences when comparing LVEF between (1) COVID-19 negative stroke patients and COVID-19 positive patients (LVEF 60%) nor (2) COVID-positive versus COVID-negative stroke patients (LVEF 67.5% versus 65%, respectively). There was no significant correlation between LVEF and MCA CBFVs (ρ=−0.18; P=0.42). Eight patients had global longitudinal strain (median −19.75%, interquartile range, −20% to −15%), with 2 patients having mildly reduced strain (<14%). There was no association between CBFVs and global longitudinal strain. Four patients had smaller than normal left ventricle systolic diameters (<2.5 cm), 3 of whom were COVID-19 positive. Thirteen patients had abnormal mitral e/a ratios (<1 or >2), including 6 of 11 COVID-19 patients with mitral e/a data. Mean CBFVs were not different in those with diastolic dysfunction versus those without (58 versus 62 cm/s).
In this cohort, 8/16 of COVID-19 positive patients and 1/10 COVID-19 negative stroke patients died. Among the COVID-19 positive patients, venous thromboembolism was confirmed in 3 (1 with pulmonary embolism and 2 with deep venous thrombosis).
Discussion
In this first study of TCD in critically ill COVID-19 patients, a short duration bilateral monitoring study did not demonstrate microembolic signals but could not also exclude potential for microembolic signals in these patients. We did observe relatively low CBFVs which appeared unrelated to cardiac ejection fraction and exhibited paradoxical associations with hematocrit, and with arterial oxygen content.
Abnormal hemostasis is a well-recognized complication in COVID-19, including elevated D-dimer, and markedly raised fibrinogen levels.4,6 COVID-19-associated hemostasis abnormality in the lungs appears to result from localized thrombosis, rather than disseminated intravascular coagulation or sepsis-induced coagulopathy.3,6 If a similar microthrombotic pathology occurs in cerebral blood vessels, we would not expect to see embolic high-intensity transient signals on TCD, typically associated with cardiac emboli. Similarly, we did not find TCD signs of vasculitis, which is consistent with autopsy findings.7 Reynolds et al8 performed contrast-enhanced TCD in 18 mechanically ventilated patients with severe COVID-19 pneumonia and found that 83% of patients had detectable microbubbles; the number, which was inversely correlated with the PaO2:FiO2 ratio and with lung compliance, suggests that pulmonary vascular dilatation may be a significant cause of hypoxemia.8
Relatively low CBFVs diffusely in COVID-positive patients were unexpected given the low hematocrit. Hematocrit and viscosity are inversely related to CBFVs which increase by ≈20% with a drop in hematocrit from 40% to 30%.9
Fibrinogen also contributes to variance in CBFVs due to its direct association with plasma viscosity, which contributes to vascular resistance.10 We found no correlation between fibrinogen and MCA velocities, which is contradictory to the described inverse and independent association of fibrinogen and CBFVs.11 Changes in PaCO2, owing to permissive hypercapnia resulted in PaCO2 above normal in 8/15 patients; we observed a weak inverse correlation with MCA velocities. The expected result is vasodilation of resistance vessels and increased CBFVs, although with sustained hypercapnia, there is no adaptation.10 Reduction of Doppler responsiveness to altered PaCO2 has been associated with increased plasma viscosity and fibrinogen.12
At time of TCD, median oxygen saturation in COVID-19 patients was 93.5%. CaO2 was significantly correlated with CBFVs in the MCA, but with a positive rather than inverse correlation which would be expected secondary to cerebral vasodilation. In studies of plasma viscosity manipulation in subjects without cerebrovascular disease, CaO2 was found to be more important than viscosity in determining cerebral blood flow.13 This reflects the importance of oxygen delivery to the brain and local mechanisms in CBF autoregulation, whereas viscosity is an important determinant of CBF when autoregulation is defective. In the latter case (eg, in ischemic tissue), resistance vessels are maximally dilated due to deficient O2 delivery and CBF passively follows perfusion pressure which is low in ischemic regions, where hematocrit may increase significantly, increasing blood viscosity and slowing blood flow.14 This study did not evaluate autoregulation, but positive associations between relatively low CBFVs and low CaO2 might suggest autoregulatory impairment in this cohort of patients experiencing relative hypoxia. Neuropathological findings from one COVID autopsy series included acute hypoxic injury in the cerebrum and cerebellum in all patients, but no thrombi or vasculitis.7
We considered that low CBFVs could be caused by a low flow state from cardiac systolic dysfunction; however, we observed a normal range of LVEF and global longitudinal strain. There was, however, a signal of diastolic dysfunction, measured by LV diameter and mitral e/a ratio. Acute myocarditis has been diagnosed in COVID-19 patients and could present with a normal LVEF and LV diastolic dysfunction.15
Limitations of these data include small sample size, observational design, short duration of TCD monitoring for emboli detection, and lack of brain imaging which prevented us from assessing the association of TCD with neurological complications. However, inability to transport critically ill patients with COVID-19 was in fact the motivation for considering TCD as a diagnostic tool. This study is intended as hypotheses-generating where hypoxic ischemic injury may have important consequences for neurological recovery. We recommend that future evaluation of TCD in COVID-19 patients with pneumonia or neurological sequelae could include a bilateral TCD study supplemented by at least 30 min emboli detection and a TCD bubble test.
Conclusions
We cautiously suggest that TCD provides clinically useful information into mechanisms underlying COVID-19 neurological sequelae, although the significance of relatively low CBFVs in these critically ill patients requires further study.
Sources of Funding
None.
Disclosures
Dr Ziai is supported by the National Institutes of Health (NIH) and has received consulting fees from C.R. Bard Inc and Portola Pharmaceuticals outside of the area of work commented on here. Dr Johansen received grant support from the American Heart Association and NIH. The other authors report no conflicts.
Supplemental Materials
Online Table I
Supplementary Material
Nonstandard Abbreviations and Acronyms
- CaO2
- arterial oxygen content
- CBFV
- cerebral blood flow velocity
- COVID-19
- severe acute respiratory syndrome coronavirus SARS-CoV-2
- CRP
- C-reactive protein
- LVEF
- left ventricular ejection fraction
- MCA
- middle cerebral artery
- PaCO2
- partial pressure of carbon dioxide
- TCD
- transcranial Doppler
The Data Supplement is available with this article at https://www.ahajournals.org/doi/suppl/10.1161/STROKEAHA.120.032150.
For Sources of Funding and Disclosures, see page 2426.
Contributor Information
Sung-Min Cho, Email: csungmi1@jhmi.edu.
Michelle C. Johansen, Email: mjohans3@jhmi.edu.
Bahattin Ergin, Email: bergin1@jhmi.edu.
Mona N. Bahouth, Email: mbahout1@jhmi.edu.
References
- 1.Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, Chang J, Hong C, Zhou Y, Wang D, et al. Neurologic manifestations of hospitalized patients with Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683–690. doi: 10.1001/jamaneurol.2020.1127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Helms J, Kremer S, Merdji H, Clere-Jehl R, Schenck M, Kummerlen C, Collange O, Boulay C, Fafi-Kremer S, Ohana M, et al. Neurologic features in severe SARS-CoV-2 infection. N Engl J Med. 2020;382:2268–2270. doi: 10.1056/NEJMc2008597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Thachil J, Srivastava A. SARS-2 Coronavirus- associated hemostatic lung abnormality in COVID-19: is it pulmonary thrombosis or pulmonary embolism?. Semin Thromb Hemost. 2020;46:777–780. doi: 10.1055/s-0040-1712155 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Alexandrov AV, Sloan MA, Tegeler CH, Newell DN, Lumsden A, Garami Z, Levy CR, Wong LK, Douville C, Kaps M, et al. ; American Society of Neuroimaging Practice Guidelines Committee. Practice standards for transcranial Doppler (TCD) ultrasound. Part II. Clinical indications and expected outcomes. J Neuroimaging. 2012;22:215–224. doi: 10.1111/j.1552-6569.2010.00523.x [DOI] [PubMed] [Google Scholar]
- 5.Ringelstein EB, Droste DW, Babikian VL, Evans DH, Grosset DG, Kaps M, Markus HS, Russell D, Siebler M. Consensus on microembolus detection by TCD. International Consensus Group on Microembolus Detection. Stroke. 1998;29:725–729. doi: 10.1161/01.str.29.3.725 [DOI] [PubMed] [Google Scholar]
- 6.Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost. 2020;18:844–847. doi: 10.1111/jth.14768 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Solomon IH, Normandin E, Bhattacharyya S, Mukerji SS, Keller K, Ali AS, Adams G, Hornick JL, Padera RF, Jr, Sabeti P. Neuropathological features of Covid-19. N Engl J Med. 2020;383:989–992. doi: 10.1056/NEJMc2019373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Reynolds AS, Lee AG, Renz J, DeSantis K, Liang J, Powell CA, Ventetuolo CE, Poor HD. Pulmonary vascular dilatation detected by automated transcranial Doppler in COVID-19 pneumonia. Am J Respir Crit Care Med. 2020;202:1037–1039. doi: 10.1164/rccm.202006-2219LE [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gaehtgens P, Marx P. Hemorheological aspects of the patho-physiology of cerebral ischemia. Cereb Blood Flow Metab. 1987;7:259–265. doi: 10.1038/jcbfm.1987.61 [DOI] [PubMed] [Google Scholar]
- 10.Ameriso SF, Paganini-Hill A, Meiselman HJ, Fisher M. Correlates of middle cerebral artery blood velocity in the elderly. Stroke. 1990;21:1579–1583. doi: 10.1161/01.str.21.11.1579 [DOI] [PubMed] [Google Scholar]
- 11.Poulin MJ, Liang PJ, Robbins PA. Dynamics of the cerebral blood flow response to step changes in end-tidal PCO2 and PO2 in humans. J Appl Physiol (1985). 1996;81:1084–1095. doi: 10.1152/jappl.1996.81.3.1084 [DOI] [PubMed] [Google Scholar]
- 12.Ringelstein EB, Mauckner A, Schneider R, Sturm W, Doering W, Wolf S, Maurin N, Willmes K, Schlenker M, Brückmann H. Effects of enzymatic blood defibrination in subcortical arteriosclerotic encephalopathy. J Neurol Neurosurg Psychiatry. 1988;51:1051–1057. doi: 10.1136/jnnp.51.8.1051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schuurman PR, Albrecht KW. Intraoperative changes of transcranial Doppler velocity: relation to arterial oxygen content and whole-blood viscosity. Ultrasound Med Biol. 1999;25:151–154. doi: 10.1016/s0301-5629(98)00135-5 [DOI] [PubMed] [Google Scholar]
- 14.Korosue K, Heros RC. Mechanism of cerebral blood flow augmentation by hemodilution in rabbits. Stroke. 1992;23:1487–1492. doi: 10.1161/01.str.23.10.1487 [DOI] [PubMed] [Google Scholar]
- 15.Hendren NS, Drazner MH, Bozkurt B, Cooper LT., Jr. Description and proposed management of the acute COVID-19 cardiovascular syndrome. Circulation. 2020;141:1903–1914. doi: 10.1161/CIRCULATIONAHA.120.047349 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


