Abstract
This research was designed to evaluate the CNS depressant, anxiolytic, and analgesic action of aqueous and ethanol extract of Ganoderma applanatum, a valuable medicinal fungus used in multiple disorders belongs to Ganodermataceae family. Two extracts of G. applanatum were prepared using distilled water and ethanol as solvents and named AEGA and EEGA. Open field method, rotarod method, tail suspension method, and hole cross method were utilized for the CNS depressant action. In contrast, elevated plus-maze test and hole board method were utilized for the anxiolytic action. For determining the analgesic potential, acetic acid-induced writhing test, hot plate method, and tail immersion test were used. Besides, molecular docking has been implemented by using Discovery studio 2020, UCSF Chimera and PyRx autodock vina. At both doses (200 and 400 mg/kg) of AEGA and EEGA showed significant CNS depressant effect (p < 0.05 to 0.001) against all four tests used for CNS depressant activity. Both doses of AEGA and EEGA exhibited important anxiolytic activity effect (p < 0.05 to 0.001)against the EPM and hole board test. Both doses of AEGA and EEGA also exhibited a potential analgesic effect (p < 0.05 to 0.001) against all three tests used for analgesic action. In addition, in the molecular docking the compounds obtained the scores of −5.2 to −12.8 kcal/mol. Ganoapplanin, sphaeropsidin D and cytosporone C showed the best binding affinity to the selected recptors. It can be concluded that AEGA and EEGA have potential CNS depressant, anxiolytic, and analgesic action, which can be used as a natural antidepressant, anxiolytic, and analgesic source.
Keywords: Ganodermataceae, Ganoderma applanatum, CNS depressant, Anxiolytic, Analgesic, Receptors
Abbreviations: WHO, World health organization; NSAID, Nonsteroidal anti-inflammatory drugs; AEGA, Aqueous extract of G. applanatum; EEGA, Ethanol extract of G. applanatum; MPE, Maximum possible effect; PRT, Pain reaction time
Highlights
-
•
The mushroom extracts were found to possess dose-dependent potentiality in antidepressant and anxiolytic test on mice model.
-
•
The mushroom extracts revealed significant inhibition in pain.
-
•
The mushroom extract is non-toxic evident from acute toxicity study.
-
•
Ganoderma applanatum can be a prominent source of CNS depressant, anxiety and pain management.
-
•
Ganoderma applanatum is a bracket fungus with a cosmopolitan distribution.
1. Introduction
Depression and anxiety are frequently found in mental disorders due to lifestyle stress [1]. Day by day, the number of patients suffering from mental disorders have been expanding, remarkably in emerging nations [2]. According to the report of the WHO, approximately 322 million persons are experiencing melancholy. This report also includes that anxiety disorders affect nearly 3.6% of the global people (more than 260 million people) [3]. Anxiety and depression can be influenced by genetic, epigenetic and environmental factors which orient further pathophysiological complexes [4]. Among the psychiatric disorders, anxiety and depression are the most common illness and together they are highly considered to the part of the wider internalizing condition. Based on data from Mental Health Care Administration and Alcohol Abuse, in 2017 the 12-month incidence of major depression disease was measured at 7.1% for adults and 13.3% in youth among the total population [5]. Currently, several antidepressant and anxiolytic medications are attainable from the market. They are still associated with various side effects, including sexual dysfunction, hypotension, gaining weight, sleeping difficulty, physical dependence, tolerance, and cardio-toxicity [6].
The severity, consistency, and period of pain can differ greatly and have different pathophysiological mechanisms and definitions. The clear and succinct description of the idea of pain is, therefore a challenge [7]. Pain is a psychic, sensory, and obnoxious event that mostly results from tissue injury [8]. Although analgesics like NSAIDs and opiates are frequently utilized to alleviate the pain without modifying sentience, they are liable for generating various side effects [9]. Ulceration, bleeding, gastrointestinal irritation, etc. are produced by NSAIDs, while opiates frequently create dependence and tolerance [9]. For this reason, researchers have shown concern to discover analgesic drugs from natural origins with fewer side effects [10].
Computational biology represents the immense opportunity that biologists are currently generating and validate experiments through this approach. The computer-aided drug development (CADD) techniques and molecular docking have been demonstrated under in silico process to develop and investigate drug design within a limited period [11]. A successful molecular docking should be able to establish the status of ligands on the binding site and the physicochemical relationship of the protein structure [12].
Drugs from natural origins have few side effects, but they are comparable in their ability to treat diseases as the synthetic derivate. Searching for alternative drug therapies for psychiatric illness from mushroom sources are recently improved dramatically [13,14]. Medicinal plants are a promising source of life-saving therapeutic products worldwide, owing to their diverse pharmacological functions [15].
Ganoderma is also identified as reishi (in Japan) or lingzhi (in China), relating to the family Ganodermataceae, scattered throughout the world [16]. Over 250 species of this fungal genus are identified. Unique double-walled basidiospores are the characteristic of this fungal genus [17,18]. Latterly pharmaceutical industries have shown concern in Ganoderma species because of their enormous folkloric medicinal uses for thousands of years [19]. Ganoderma applanatum is a wood-decay fungus and generally found at the bases of tree stumps [20]. The different parts of the G. applanatum contains Applanoxidic acid E, Applanatumol A, Applanatumin A, Applanatumol E, Applanatumol P, Applanatumol Q, Cytosporone C, Stemphone B, Condidymic acid, Nigragillin, Ganoapplanin, Myrocin C, Sphaeropsidin D, Epoxy-4,4,14-trimethyl3,7,11,15,20-pentaoxo-pregnane, Comazaphilone D, Applanoxidic acid (C-G), and etc; as its biochemical compositions [[20], [21], [22], [23]]. Several studies represented that G. applanatum contains compounds with potent anti-tumor, antibacterial, and antifibrotic properties [24]. Various medicines have been prepared from this fungus to treat the liver injury, cirrhosis, and hepatitis [25]. G. applanatum is also found to have antiobesity, antiviral, antioxidative, and immunomodulating activities [20]. Therefore, in this study, in vivo antidepressant, anxiolytic, and analgesic effects of the two extract of G. applanatum along with phytochemical evaluation were analyzed. Besides, computational investigation (molecular docking) of the chemical composition with the receptors associated with the antidepressant, anxiolytic, and analgesic effects also have been implemented as these studies were not analyzed previously.
2. Materials and methods
2.1. Drugs and chemicals
Analytical grade chemicals and solvents were utilized during this experiment. Diclofenac sodium and diazepam were purchased from the Bangladeshi pharmaceutical company named Square Pharmaceuticals Limited. Department of Pharmacy, University of Chittagong, had provided the remaining chemicals.
2.2. Experimental animal
4–5 weeks old albino rodents of both genders within 20–25 g weight range were obtained from BCSIR, Chattogram, Bangladesh. They remained housed in room temperature conditions for a week before commencing the test to acclimatize the environment. During the experimental period, a balanced diet and tap water were provided to the Swiss albino mice.
2.3. Animal euthanasia
The animals were treated in compliance with the values of the Swiss Academy of Sciences and Swiss Academy of Medical Sciences and have been euthanized according to the recommendations for animal euthanasia: 2013 edition [26]. This research has been approved by the ethical review board of the Department of Pharmacy, University of Chittagong, Bangladesh under the consent number: CUDP:March 16, 2019:17).
2.4. Collection and identification of the plant
The mushroom was identified by fungus expert Dr. Akther Jahan Kakon, Mushroom Specialist, Mushroom Development Institute, Savar, Dhaka, and systemic herbarium was prepared for authentication and further correspondence if necessary. The identified mushroom is G. applanatum. Herbarium Accession number: 2018/004/Fungi/CU/DP.
2.5. Preparation of the plant extract
The gathered mushrooms were milled into the powders for effective extraction. The powders were entered into a clean glass container for soaking in solvents. The sealed vessel was retained for 14 days with occasional shaking. Whatman filter paper no. 1 was used to filter the mixture while a rotary evaporator was used for the evaporation of the solvents. Finally, the blackish crude extracts were obtained and kept in the refrigerator for further use. Two extracts of G. applanatum were prepared by using distilled water and absolute ethanol as solvent named as the aqueous extract of G. applanatum (AEGA) and ethanol extract of G. applanatum (EEGA).
2.6. Phytochemical screening
Standard methods were adopted for the qualitative determination of active constituents present in the crude ethanol and aqueous extract of G. applanatum [27,28].
2.7. CNS anti-depressant activity of the plant extracts
2.7.1. Open field test
For assessing the CNS depressant activity, the process illustrated by Gupta and Hussain et al. [29,30] was used with a small adjustment. After splitting the animals into six classes (n = 5), negative control (1% Tween-80 in saline, 10 mL/kg; p. o.), positive control (diazepam, 1 mg/kg, i. p.) AEGA and EEGA (200 and 400 mg/kg; p. o.) was administered to different classes. The number of fields covered by individual mice of different groups was noted for 3 min at 0, 30, 60, 90, 120 min throughout the experimental phase.
2.7.2. Rotarod test
Dunham and Miya outlined the method used for this experiment with small adjustments [31]. A rotarod apparatus with a four-section design was used to perform this research. Rodents were split into six classes (n = 5), and each class was managed with similar doses, as mentioned in the previous method. The extracts were orally administered 60 min before commencing the test, while diazepam was intraperitoneally administered 30 min before starting the experiment. The time at which the animals fell off from the rotating rod at a speed of 24 rpm was noted at 30, 60, and 90 min.
2.7.3. Tail suspension test
This test was performed with slight modifications. In this experiment, rodents were divided into six classes, with five mice in any class. The extracts of G. applanatum (200 and 400 mg/kg, p. o.) was used to treat the test groups (Group III, Group IV, Group V, and Group VI). The diazepam (1 mg/kg, i. p.) was used as a positive control, while 1% Tween-80 in saline was negative (10 mL/kg, p. o.). Thirty minutes after the drug administration to the animals, rodents were hung from 50 cm above the ground, utilizing sticky tape lodged nearly 1 cm from their tails' apex. The period during which the immobility of animals recorded was 6 min. When the mice silently hung or remained motionless, they were considered immobile [32].
2.7.4. Hole cross test
The process was performed as defined by Islam et al. [33]. Group I received 1% Tween-80 in saline orally, while Group II received diazepam (1 mg/kg, i. p.). On the other hand, the remaining experimental groups received G. applanatum extracts at the doses of 200 mg/kg and 400 mg/kg body weight orally. The number of holes crossed by the mice in 3 min was recorded at 0, 30, 60, 90, and 120 min [34].
2.8. Anxiolytic activity of the plant extracts
2.8.1. Hole board test
The hole board device formed of a wooden chamber (40 × 40 × 25 cm) with 16 equally aligned holes (3 cm diameter). The device was constructed to a climax of 25 cm from the ground so that the mice could peep through the holes. The rodents were treated with G. applanatum (200 and 400 mg/kg, p. o.), the vehicle (10 mL/kg, p. o.) or diazepam (1 mg/kg, i. p.), 30 min before commencing the experiment. The numbers and the duration of the head poking of each animal were counted during the 5 min observation period [35].
2.8.2. Elevated pluz-maze test
This widely approved experiment was conducted for evaluating anxiolytic activities in rodents [36]. The elevated plus-maze was positioned 50 cm above the ground. The animals were split into negative control, positive control, and test groups (n = 5). After treating the test extracts (200 and 400 mg/kg), diazepam (1 mg/kg), and vehicle, every mice was located in the middle of the maze approaching the closed arms. The time spent in closed and open arms and the total numbers of open and closed arm entries were noted for 5 min at 0, 30, 60, 90, and 120 min. The entire operation was managed in a sound-attenuated place.
2.9. Analgesic activity of the plant extract
2.9.1. Acetic acid-induced writhing test
The acetic acid-induced writhing method was widely used to evaluate the analgesic potential of G. applanatum [37,38]. After overnight fasting, two different doses (200 and 400 mg/kg body weight) of the test extracts, vehicle (10 mL/kg body weight), diclofenac sodium (10 mg/kg body weight) were treated orally 40 min before the intraperitoneal injection of 0.7% acetic acid. The total numbers of writhing were counted for each animal for 15 min, 5 min after acetic acid administration.
2.9.2. Hot plate test
When the rodents were put on Eddy's hot plate [39], the heat was kept at 55 ± 0.5 °C. To avoid the harmful effect on mice's skin, a cutoff time of 30 s was maintained [40]. After oral treatment of the specimens, the response period was noted at 0, 30, 60, and 90, 120, 180, and 240 min. The experiment groups' mice were treated at two doses (200 and 400 mg/kg body weight). The positive control group received diclofenac sodium (10 mg/kg body weight) while the negative control received vehicle (10 mL/kg body weight).
2.9.3. Tail immersion test
The tail immersion method was done to assess the central analgesic property and followed the developed method mentioned by Gawel et al. [41]. This method is based on the outcome that drugs that show a morphine-like effect can extend the abandonment of the mice tail from warm water. After treating the animal with diclofenac sodium or extracts, one to two cm of the rodents' tail was submerged in hot water, maintaining a temperature of 54 ± 0.5 °C. After administering the drug and extracts, the tail-withdrawal reaction's latency duration was recorded at 30, 60, 90, and 120 min. The latency phase was kept to 20 s so that the damage of tail tissue could be bypassed.
2.10. In silico computer aid virtual screening
2.10.1. Software tools
During the process of this analysis, the following were used: UCSF Chimera, AutodockVina, Discovery Studio Visualizer 2020 (BIOVIA), drug banking, MGL instruments, Protein Data Bank (PDB) and PubChem.
2.10.2. Selection of the target proteins
A systematic review for the current scientific research has mainly accomplished the collection of possible treatment objectives of anxiety, depression and pain. From the protein data bank (PDB) (large biological molecules database viz.), the selected targets were confirmed again [42].
2.10.3. Preparation of target proteins
Cyclooxygenase-1 (PDB: 2OYE) [43], Potassium channel KCSA-FAB (PDB: 4UUJ) [44] and human serotonin transporter (PDB: 5I6X) [45] have been downloaded from the RCSB Protein Data Bank (PDB). These constructs have been identified as acceptable and connected to Homo sapiens, Mus musculus, Streptomyces lividans and Ovis aries. In addition, these structures were also used in recent research works and have been found in a various reputed published journal. This ensures that the findings are reproducible by using relevant tools. PDB ID 2OYE, 4UUJ and 5I6X are well-authenticated receptors for activated pain, anxiety and depression. This ensures that the findings are reproducible by using relevant tools. PDB ID 2OYE, 4UUJ and 5I6X are well-authenticated receptors for activated pain, anxiety and depression. Beginning of the study, the protein ligands were detached from the complexes via Discovery Studio Visualizer (BIOVIA). For optimization and preparation of these proteins in the docking process, UCSF Chimera has been used. Water molecules were eliminated from structures and polar hydrogen was added to the necessary H-atoms. After the gasteiger charge estimation, these proteins were converted and saved into pdbqt format with AutodockVina docking [46].
2.10.4. Virtual filtering
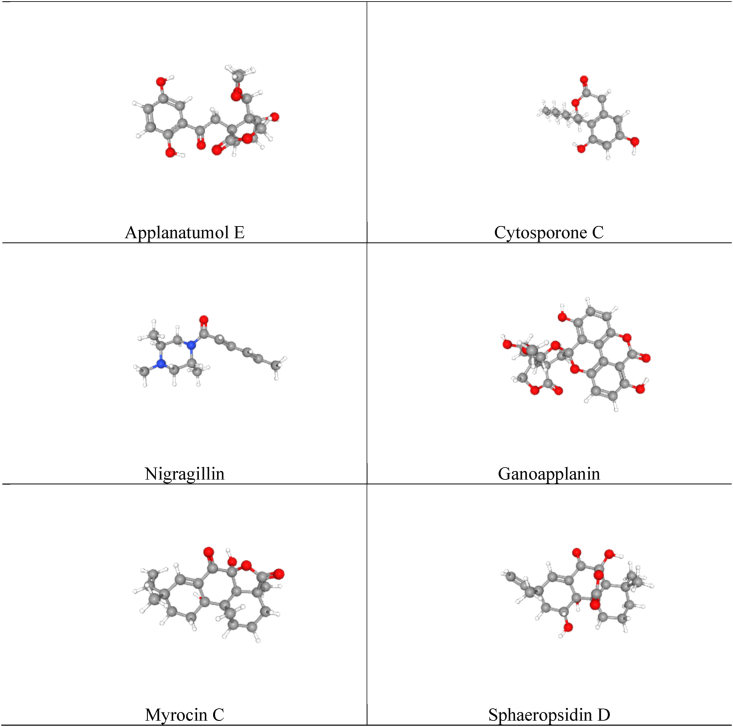
To complete the virtual screening, PyRx-autodockvina software has been operated [46]. The ligands (Applanatumol E, Cytosporone C, Nigragillin, Ganoapplanin, Myrocin C, Sphaeropsidin D) of Ganoderma applanatum mushroom (Fig. 1) were minimized and optimized by PyRX-autodock vina software and were docked with the selected receptor targets (2OYE, 4UUJ and 5I6X). The implementation of the PyRx autogrid program, ligand centered grid box has covered the binding site of the target pockets of the receptors. The selection of the grid box enables the ligand to pass about freely within the allocated values in planes (X, Y and Z). The coordinate values of x, y and z were obtained using the Discovery Studio Visualizer from the respective PDB crystals, i.e. 2OYE, 4UUJ and 5I6X. Grid scale was 60 Å × 60 Å Å x 60 Å as in previous publications. Finally, the docking (ligand-protein interactions) have been visualized by BIOVIA Discovery Studio Visualizer 2020.
Fig. 1.
The chemical structures of the selected compounds (Applanatumol E, Cytosporone C, Nigragillin, Ganoapplanin, Myrocin C, Sphaeropsidin D) of Ganoderma applanatum mushroom.
3. Statistical analysis
The data were manifested as mean ± SEM. Statistical comparisons were conducted using one-way ANOVA followed by Dunnett's multiple comparison tests. The resulting values were compared with the positive control group and were statistically significant when p < 0.05, p < 0.01, p < 0.001.
4. Results
4.1. Phytochemical screening
The results of the phytochemical screening has been presented in Table 1. The screening showed the presence of Alkaloids, Glycosides, Triterpenes, Carbohydrate, Flavonoids and Steroids in the ethanol extract of the G. applanatum. Bsides, the primary screening of the aqueous extract of the G. applanatum yielded the presence of Alkaloids, Cardiac glycosides, Triterpenes, Carbohydrate, Flavonoids and Steroids.
Table 1.
Observation and results of chemical screening.
| Secondary Metabolites |
Name of the test |
Observation |
Result |
|
|---|---|---|---|---|
| EEGA | AEGA | |||
| Alkaloids | Wagner's test | Brown or deep brown precipitate | + | + |
| Glycosides | General test | Yellow color | + | – |
| Cardiac glycosides | 1. Legal's test 2. Baljet's test |
1. No pink or red color 2. No yellow orange color |
_ | + |
| Triterpenes | Salkowsky test | Reddish coloration | + | _ |
| Carbohydrate | Molisch test | Red-violet layer at the interface between the acid (bottom) and aqueous (upper) layers | + | + |
| Flavonoids | 1. General test 2. Specific test |
1. A yellow coloration 2. Orange to red color |
+ | + |
| Steroids | Libermann- Burchard's test | Greenish color | + | + |
| Tannins | FeCl3 test | No brownish green color | – | – |
| Saponins | Frothing test | Change is observed | – | – |
“+” stands for the presence and “-” indicates the absence of secondary metabolites.
4.2. Open field test
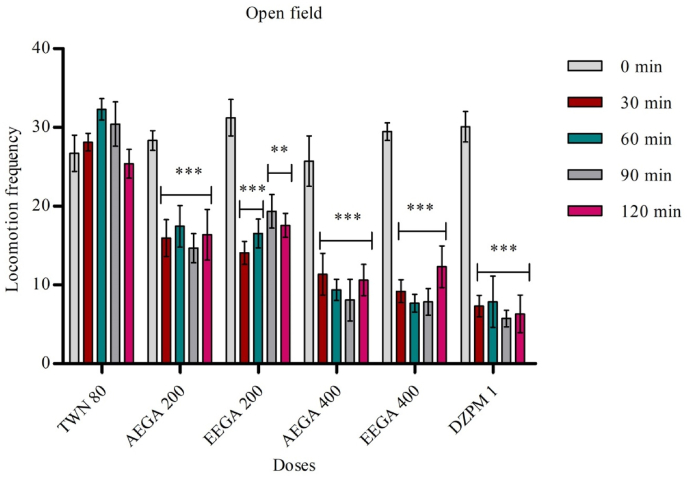
Both extract (water and ethanol) of G. applanatum fungus were subjected to screening for the open-field method's anxiolytic activity. The test was performed by taking aqueous extract at 200 mg/kg and 400 mg/kg body weight. The AEGA and EEGA significantly (p < 0.05, <0.01, <0.001) reduced the number of squares passed by the animals. The study results are shown in the following Fig. 2.
Fig. 2.
Effect of test samples in open field test. Results are expressed as mean ± SEM (n = 5) and statistically analyzed using ANOVA followed by Dunnett's comparison test. *p < 0.05, **p < 0.01 ***p < 0.001 have been considered as significant, when compared with control (n = 5). AEGA = aqueous extract of Ganoderma applanatum. EEAA = ethanol extract of Ganoderma applanatum.
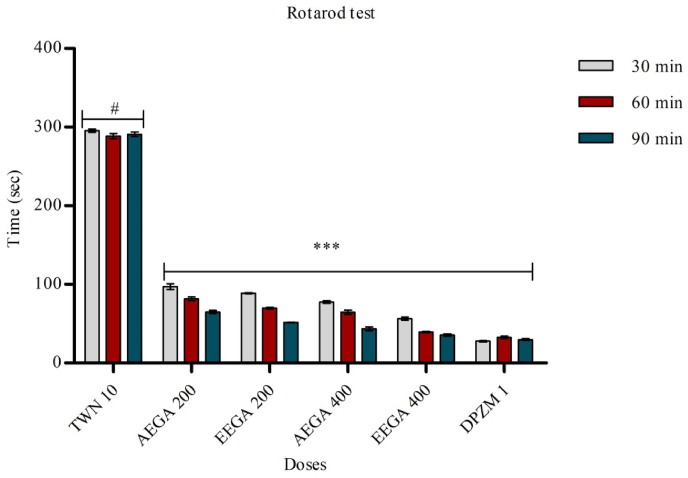
4.3. Rotarod test
In this method, mice falling from the rotarod in both diazepam and AEGA, EEGA treated groups exhibited a reduction in time spent by the mice in the rotarod test compared with the control group. The result was found statistically highly significant (p < 0.05, <0.01, <0.001). The experimental results were shown in Fig. 3.
Fig. 3.
Effects of test samples in rotarod test. Results are expressed as mean ± SEM (n = 5) and statistically analyzed using ANOVA followed by Dunnett's comparison test. *p < 0.05, **p < 0.01 ***p < 0.001 have been considered as significant, when compared with control (n = 5). AEGA = aqueous extract of Ganoderma applanatum. EEAA = ethanol extract of Ganoderma applanatum.
4.4. Tail suspension test
The extracts at the dose of 200 mg/kg found a significant (p < 0.01) rise in the immobility times when compared to the control group and at the dose of 400 mg/kg found a (p < 0.001) rise in the immobility times when compared to control group. In this test, the influence of the extract on behavior is shown in Table 2.
Table 2.
Effects of test samples in tail suspension test.
| Group | Dose (mg/kg) | Immobility time (sec) |
|---|---|---|
| Tween 80 | 0.1 mL/kg | 111.2 ± 2.81 |
| Diazepam | 1 | 212 ± 1.63*** |
| AEGA | 200 | 182 ± 2.65** |
| AEGA | 400 | 252 ± 3.61*** |
| EEGA | 200 | 198 ± 2.65** |
| EEGA | 400 | 263 ± 3.35*** |
Results are expressed as mean ± SEM (n = 5) and statistically analyzed using ANOVA followed by Dunnett's comparision test. *p < 0.05, **p < 0.01 ***p < 0.001 have been considered as significant, when compared with control (n = 5). AEGA = aqueous extract of Ganoderma applanatum. EEAA = ethanol extract of Ganoderma applanatum.
4.5. Hole cross test
The aqueous and absolute ethanol extract of G. applanatum was exposed to screen the anti-depressant potential by hole cross method. The result was statistically significant (p < 0.001) presented in Table 3.
Table 3.
Effects of test samples in hole cross test.
| Group | Dose (mg/kg) | Number of hole crossed in minutes |
||||
|---|---|---|---|---|---|---|
| 0 min | 30 min | 60 min | 90 min | 120 min | ||
| Tween 80 | 10 mL/kg | 16.33 ± 1.60 | 14.67 ± 1.25 | 15.0 ± 1.00 | 16.67 ± 0.76 | 15.00 ± 0.58 |
| Diazepam | 1 | 19.00 ± 0.86*** | 6.00 ± 1.32*** | 5.67 ± 1.52*** | 5.00 ± 1.00*** | 3.67 ± 0.57*** |
| AEGA | 200 | 16.93 ± 1.11*** | 4.47 ± 0.88*** | 4.12 ± 1.11*** | 3.41 ± 1.70*** | 5.12 ± 0.78*** |
| AEGA | 400 | 22.45 ± 1.42*** | 3.97 ± 0.95*** | 3.10 ± 1.53*** | 2.43 ± 1.77*** | 2.89 ± 0.99*** |
| EEGA | 200 | 20.33 ± 1.45*** | 4.67 ± 1.25*** | 3.00 ± 1.k60*** | 3.33 ± 1.50*** | 5.00 ± 1.00*** |
| EEGA | 400 | 20.68 ± 1.89*** | 3.00 ± 2.17*** | 2.33 ± 1.25*** | 2.33 ± 1.25*** | 3.33 ± 1.25*** |
Results are expressed as mean ± SEM (n = 5) and statistically analyzed using ANOVA followed by Dunnett's comparision test. *p < 0.05, **p < 0.01 ***p < 0.001 have been considered as significant, when compared with control (n = 5). AEGA = aqueous extract of Ganoderma applanatum. EEAA = ethanol extract of Ganoderma applanatum.
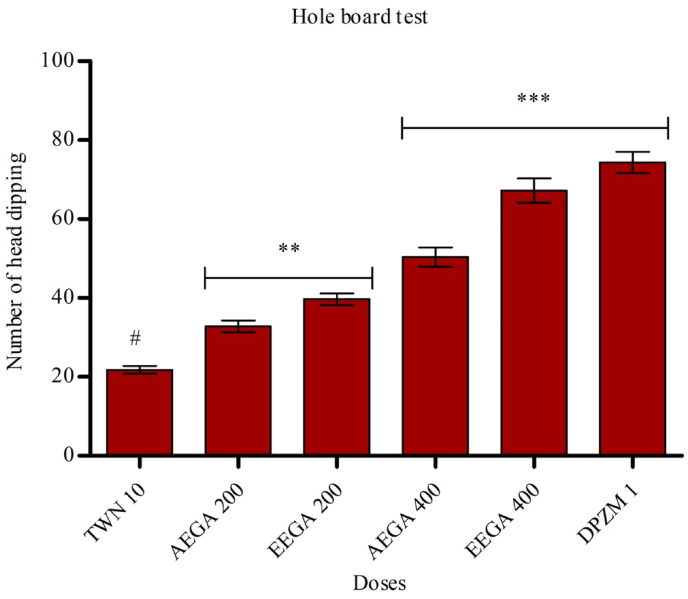
4.6. Hole board test
The test results are captured in Fig. 4, and it represents the anxiolytic effect of aqueous extract of G. applanatum. The extract at the doses of 400 mg/kg has shown most significantly (p < 0.001) improved the head dipping numbers compared to control. At the doses of 200 mg/kg, the head dipping number also increased significantly (p < 0.01).
Fig. 4.
Effects of test samples in hole board test. Results are expressed as mean ± SEM (n = 5) and statistically analyzed using ANOVA followed by Dunnett's comparison test. *p < 0.05, **p < 0.01 ***p < 0.001 have been considered as significant, when compared with control (n = 5). AEGA = aqueous extract of Ganoderma applanatum. EEAA = ethanol extract of Ganoderma applanatum.
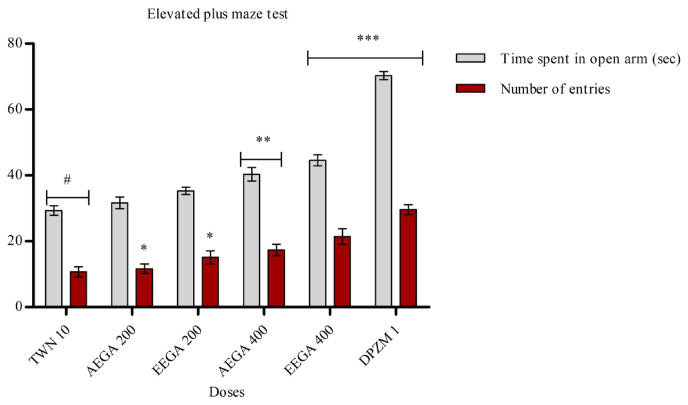
4.7. Elevated pluz-maze test
The extracts of G. applanatum significantly decreased the number of entrances of mice into the open arms. The staying time in the open arms of the EPM is presented in Fig. 5. The standard drug (diazepam 1 mg/kg, i. p) found a significant (p < 0.01) rise in time spent in open arm and a significant (p < 0.01) rise in the number of open arm entries.
Fig. 5.
Effect of test samples in elevated plus-maze test. Results are expressed as mean ± SEM (n = 5) and statistically analyzed using ANOVA followed by Dunnett's comparison test. *p < 0.05, **p < 0.01 ***p < 0.001 have been considered as significant, when compared with control (n = 5). AEGA = aqueous extract of Ganoderma applanatum. EEAA = ethanol extract of Ganoderma applanatum.
4.8. Acetic acid induced writhing
The test was executed by taking aqueous and ethanol extracts (AEGA and EEGA) at doses of 200 mg/kg and 400 mg/kg body weight. The result was found statistically significant. Experimental results are tabulated in Table 4.
Table 4.
Effect of test samples in acetic acid induced writhing test.
| Group | Dose (mg/kg) | Writhing count | % of Writhing | % of Inhibition |
|---|---|---|---|---|
| Tween 80 | 10 mL/kg | 48.8 ± 1.40 | – | – |
| Diclofenac sodium | 10 | 7.2 ± 0.35*** | 15.36 | 84.63 |
| AEGA | 200 | 39.3 ± 0.78* | 78.55 | 21.45 |
| AEGA | 400 | 30.5 ± 0.98* | 62.25 | 37.75 |
| EEGA | 200 | 32.9 ± 0.87* | 67.41 | 32.58 |
| EEGA | 400 | 27.2 ± 0.98* | 79.09 | 44.26 |
Results are expressed as mean ± SEM (n = 5) and statistically analyzed using ANOVA followed by Dunnett's comparision test. *p < 0.05, **p < 0.01 ***p < 0.001 have been considered as significant, when compared with control. AEGA = aqueous extract of Ganoderma applanatum. EEAA = ethanol extract of Ganoderma applanatum.
4.9. Tail immersion test
The test was performed by taking extract at doses of 200 mg/kg and 400 mg/kg body weight, and the tail withdrawal reflex time represented almost the same as increasing the dose of the sample compared to the standard drug. Highly potential (p < 0.001) action was originated by the treatment of AEGA and EEGA at a dose of 200 mg/kg in 60 min, which was 4.38 ± 0.56, and the administration of AEGA and EEGA at a dose of 400 mg/kg in 60 min and 90 min, which was 4.11 ± 0.07 and 4.79 ± 0.06 respectively (Table 5).
Table 5.
Effect of test samples in tail immersion test.
| Group | Dose (mg/kg) | Response time (sec) |
||||
|---|---|---|---|---|---|---|
| 0 min | 30 min | 60 min | 90 min | 120 min | ||
| Tween 80 | 10 mL/kg | 2.85 ± 0.02 | 2.63 ± 0.03 | 2.83 ± 0.15 | 2.64 ± 0.03 | 2.70 ± 0.09 |
| Diclofenac sodium | 10 | 2.64 ± 0.03 | 4.13 ± 0.08*** | 5.36 ± 0.14*** | 4.38 ± 0.05*** | 3.41 ± 0.11 |
| AEGA | 200 | 2.69 ± 0.07 | 3.53 ± 0.17* | 4.38 ± 0.56*** | 3.41 ± 0.02 | 2.87 ± 0.09 |
| AEGA | 400 | 2.63 ± 0.03 | 3.33 ± 0.16 | 4.11 ± 0.07*** | 4.79 ± 0.06** | 3.15 ± 0.08 |
| EEGA | 200 | 2.71 ± 0.06 | 3.63 ± 0.10* | 4.21 ± 0.22** | 3.31 ± 0.05 | 2.88 ± 0.07 |
| EEGA | 400 | 2.73 ± 0.06 | 3.41 ± 0.12 | 4.31 ± 0.07*** | 4.99 ± 0.04*** | 3.24 ± 0.07 |
Results are expressed as mean ± SEM (n = 5) and statistically analyzed using ANOVA followed by Dunnett's test. *p < 0.05, **p < 0.01 ***p < 0.001 have been considered as significant, when compared with control. AEGA = aqueous extract of Ganoderma applanatum. EEAA = ethanol extract of Ganoderma applanatum.
4.10. Hot plate test
AEGA and EEGA augmented the latency time at doses of 200 and 400 mg/kg after 60, 120, and 180 min, respectively. At a dose of 400 mg/kg, the extract showed significant (p < 0.001) at a time interval of 120 and 180 min (Table 6).
Table 6.
Effect of test samples in hot plate test.
| Group | Dose | Response time |
|||||
|---|---|---|---|---|---|---|---|
| 0 min | 30 min | 60 min | 120 min | 180 min | 240 min | ||
| Control | 10 mL/kg | 8.0 ± 0.71 | 9.4 ± 0.51 | 6.8 ± 0.58 | 8.4 ± 0.51 | 10 ± 0.71 | 7.8 ± 0.97 |
| Diclofenac sodium | 10 | 10.6 ± 0.51 | 14.4 ± 0.43*** | 12.6 ± 0.51*** | 13.3 ± 0.68*** | 8.2 ± 0.86 | 8 ± 0.89 |
| AEGA | 200 | 9.4 ± 0.51 | 10.4 ± 1.14 | 12.2 ± 0.58** | 11.00 ± 0.71 | 14 ± 1.41** | 13 ± 0.71** |
| AEGA | 400 | 8.5 ± 0.71 | 11.44 ± 1.6 | 13.54 ± 1.18** | 14.7 ± 1.99*** | 18.28 ± 1.37*** | 11 ± 1.58 |
| EEGA | 200 | 8.1 ± 0.21 | 8.5 ± 1.10 | 11.4 ± 0.40 | 12.5 ± 0.52 | 13.5 ± 1.01** | 13.4 ± 0.53** |
| EEGA | 400 | 9.75 ± 0.11 | 8.1 ± 0.11 | 12.3 ± 0.35** | 13.5 ± 0.41 | 16.31 ± 0.45*** | 12.47 ± 0.81** |
Results are expressed as mean ± SEM (n = 5) and statistically analyzed using ANOVA followed by Dunnett's test. *p < 0.05, **p < 0.01 ***p < 0.001 have been considered as significant, when compared with control. AEGA = aqueous extract of Ganoderma applanatum. EEAA = ethanol extract of Ganoderma applanatum.
4.11. In silico computer aid virtual screening
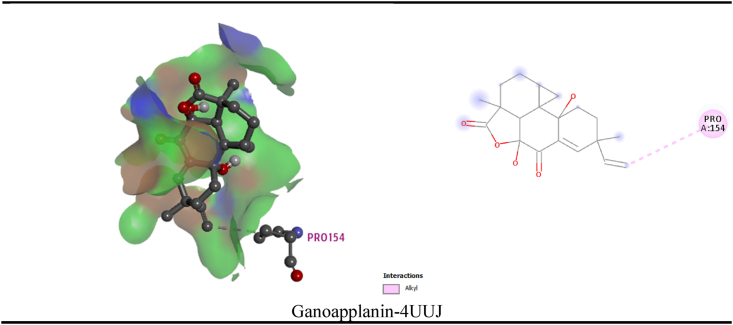
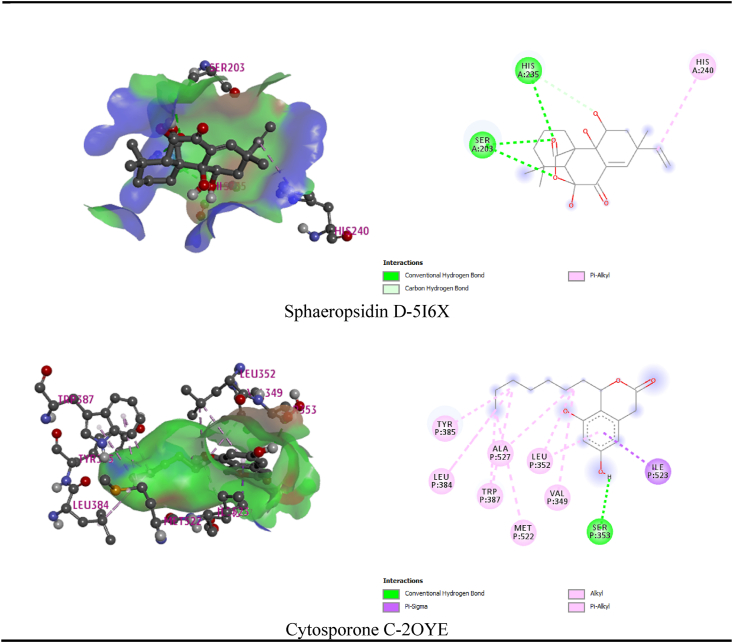
In the current review, the maximum and least interaction with Potassium channel KCSA-FAB (PDB: 4UUJ), was demonstrated by ganoapplanin and nigragillin with docking scores of −10.5 kcal/mol and 5.7 kcal/mol respectively, whereas the standard drug Diazepam attained −7.3 kcal/mol binding affinity. By interacting with pro154 residue, Ganoapplanin had interacted with the KCSA-FAB Potassium channel. Table 7 and Fig. 6 displays the relationships of the compounds. Nigragilline has the least docking score (−5.2 kcal/mol) on this simulation of molecular docking with the selected compounds and human serotonin transporter (PDB: 5I6X). Sphaeropsidin D also demonstrated strongest binding affinity (−9.2 kcal/mol). Sphaeropsidin D interacted with the drug pocket via a number of residues (ser203, his236 and his240). In addition, Cytosporone C displayed strongest (−12.8 kcal/mol) binding affinity to the Cyclooxygenase-1 (PDB: 2OYE) receptor where the standard drug showed binding affinity to the same receptor as (−6.9 kcal/mol). Cytosporone C binds to the Cyclooxygenase-1 through a series: conventional hydrogen bond (ser353), pi-sigma (ile523), alkyl bond (val349, met522, trp387, leu384, tyr385, ala527, leu 352) of bonds.
Table 7.
Docking scores of the selected compounds of Ganoderma applanatum mushroom with the potassium channel KCSA-FAB (PDB: 4UUJ) and human serotonin transporter (PDB: 5I6X) and Cyclooxygenase-1 (PDB: 2OYE) receptors.
| Docking Score (kcal/mol) | ||||
|---|---|---|---|---|
| Compounds | PubChem CID | Anxiolytic |
Antidepressant |
Analgesic |
| 4UUJ | 5I6X | 2OYE | ||
| Applanatumol E | 132542814 | −7.1 | −6.0 | −6.4 |
| Cytosporone C | 10778975 | −5.7 | −5.8 | −12.8 |
| Nigragillin | 15939563 | −5.7 | −5.2 | −7.3 |
| Ganoapplanin | 11067914 | −10.5 | −6.8 | −7.1 |
| Myrocin C | 132581256 | −7.5 | −6.8 | −7.9 |
| Sphaeropsidin D | 636779 | −7.9 | −10.5 | −6.9 |
| Standard drugs (Diazepam/Diclofenac sodium) | 3016/5018304 | −7.3 | −6.4 | −6.9 |
Fig. 6.
Best interaction (3D and 2D) of the selected compounds of Ganoderma applanatum mushroom with the Potassium channel KCSA-FAB (PDB: 4UUJ) and human serotonin transporter (PDB: 5I6X) and Cyclooxygenase-1 (PDB: 2OYE) receptors.
5. Discussion
WHO has reported that 80% of people worldwide utilize medicinal plants to treat various diseases due to the presence of phytoconstituents [47]. G. applanatum has a wide range of therapeutic activities.
Many mushrooms have been used in previous research to overcome anxiety [48], depressant [49] and pain [50] in different ways. Our study is the first systemic pharmacological and computational evaluation of the efficacy of methanol and aqueous extracts of Ganoderma applanatum (MEGA and AEGA) for the treatments of anxiety, depression and so that, this investigation was designed to estimate the CNS depressant, anxiolytic, and analgesic activity of AEGA and EEGA.
CNS excitability levels are observed by a reaction, which is known as locomotion. An increase in locomotors activity represents the anxiolytic effect, while a decrease in locomotor activity indicates the sedative effect [51]. Our current study showed that AEGA and EEGA possessed strong CNS depressant potential in the open field, rotarod, tail suspension, and hole cross tests. The extracts' CNS depressant action is assured when the locomotor activity and animals' exploratory behavior are reduced in a dose-dependent manner. Both doses of EEGA and AEGA showed CNS depressant activity in all tests.
The hole board and EPM are the most feasible model for the evaluation of anxiolytic effect. This study also planned to determine of the anxiolytic effect of AEGA and EEGA by using the hole board and EPM test on Swiss albino mice. For the determination of anxiolytic actions of numerous medicinal plant extracts, the hole board is frequently utilized. A high level of anxiolytic behavior is confirmed when head dipping of animals in the hole board increases. On the other hand, a fall in head dipping in the hole board exhibits a high anxiety behavior [52]. In this study, both doses of AEGA and EEGA significantly increased the head dipping in the hole board compared to control. Standard drug diazepam also increased head dipping in the hole board when compared to control. There is a tendency of animals to leave the open area in EPM. This leaving tendency of mice from an open arm reflects the anxiolytic effect. Therefore, an increase in open arm activity of mice expresses an increase in anxiolytic activity [1]. In this study, both doses of AEGA and EEGA significantly decreased both the stay time and the number of entries in open arms when compared with control. In contrast, diazepam increased both the stay time and the number of entries in open arms than control. So, the extracts showed anxiolytic activity only in the hole board method.
Various earlier researches suggested that phytoconstituents like alkaloids, flavonoids, tannins, sterol, saponins, and terpenoids are accountable for CNS depressants anxiolytic effect [1,47,51]. Gamma aminobutyric acid is a chief neurochemical in the CNS. G. applanatum showed actions might be due to the enhancement of GABAergic suppression in the CNS via hyperpolarization of membrane. Hyperpolarization might cause a reduction in the rate of firing of some crucial neurons within the brain. G. applanatum could also work by direct activation of GABA receptors [53]. Another study also stated that the reduction of serotonin and noradrenaline also plays a part in motor activity. Decrease secretion of serotonin and noradrenaline indicates CNS depressant activity [54]. It is reported that plants containing saponins, tannins, and flavonoids have a beneficial role in numerous CNS disorders [47,55]. The supposed anxiolytic action of the AEGA and EEGA might be due to the binding of plant constituents like flavonoids and saponins to the GABA-A and benzodiazepine complex [56].
The most valuable method for screening the peripheral analgesic effect is the acetic acid-induced writhing test [6]. Endogenous pain mediators, such as prostaglandins, histamine, and bradykinin, are induced when a 0.7% solution of acetic acid is administered intraperitoneally in mice. As a result, pain sensation occurs in a localized area of mice [57]. At the time of the writhing test, the concentration of lipoxygenase products and prostanoids (PGE2 and PGF2α) was significantly raised in the peritoneal fluid [38]. In this study, both doses of AEGA and EEGA showed significant analgesic activity. So the observed analgesic activity of the AEGA and EEGA might be due to its possible interruption with those mentioned pain mediators' actions. A study revealed that alkaloids possess excellent analgesic activity [58]. The phytochemical study of AEGA and EEGA revealed alkaloids, which might act as major contributors to the anti-nociceptive effect.
Tail immersion and hot plate methods are suitable methods for screening centrally acting analgesic effects [59]. According to the procedure used in the tail immersion test, the reaction time of the tail withdrawal reflex in mice will be prolonged if the drugs show morphine-like activity [60]. On the contrary, in the hot plate model, the sensitiveness of animal paws is found at 50–55 ± 1 °C temperatures [40]. The analgesic effect of the drug is increased when the PRT time is increased [61]. Both doses of AEGA and EEGA significantly increased the PRT when compared to control. So, the extracts are believed to have centrally acting analgesic effects. In order to produce new medicines, the development of computer-aided drugs (CADD) plays a key role. Two types of drug design approaches are primarily available: structural and ligand based drug design [62]. In the previous research, researchers used ligand-based relations to pick a lead compound that demonstrated a strong affinity with the potassium channel (PDB: 4UUJ) [63], human serotonin receptor (PDB: 5I6X) [32] and COX-1 (PDB: 2OYE) receptors [37,38]. The MAO receptor is used in this study because MAO-A is typically aimed at treating depression and anxieties [64]. In this research, molecular docking was carried out for six main compounds of G. applanatum with the active site of the enzymes: 4UUJ, 5I6X and 2OYE. The analysis demonstrated that, Ganoapplanin, Sphaeropsidin D and Cytosporone C have the best docking score of −10.5, −10.5 and −12.8 with the 4UUJ, 5I6X and 2OYE enzymes correspondingly, which also recommends that they have a superior interface with the 4UUJ, 5I6X and 2OYE receptors than the other bioactive constituents. However, more experiments to confirm the pharmacological implications of these isolated substances are required.
6. Conclusion
Our analysis exhibited that the AEGA and EEGA have substantial medicinal property via its CNS depressant, anxiolytic, and analgesic activity, all of the activity measured with the standard reference drug. This study suggests that the biological constituents present in the plant are worthy of further investigation and structure elucidation.
Author's contribution
SMMH: conceptualization, research design, acquisition, supervision. MJI: investigation, formal analysis, data curation. MSH: formal analysis, data curation, wrote the manuscript. MR: formal analysis, data curation. AB: formal analysis, data curation. MGU: visualization, validation, review and editing. NUE: methodology, investigation, research design, software, writing the original manuscript, review and editing. Finally all the authors reviewed and agreed to publish this research.
Funding
The research work received no grant or fund to execute this research.
Conflicts of interest
The authors have no known competing interests.
Ethical consideration
In keeping with the ethical principles set forth in the Helsinki 2013 Declaration [65,66] all biological activity evaluation was carried out.
Declaration of competing interest
The authors declare no conflict of interest for this research.
Acknowledgements
We are thankful to the Department of Pharmacy, University of Chittagong, for technical and laboratory support.
Contributor Information
S M Moazzem Hossen, Email: hossen.pharmacy@cu.ac.bd.
Nazim Uddin Emon, Email: nazim7emon@gmail.com.
References
- 1.Hasan M.S., Uddin M.G., Shoibe M., Al Mahmud A., Banik S. Evaluation of anxiolytic and hypoglycemic potential of Cissus adnata Roxb. in animal model. J. Compl. Integr. Med. 2019;1 doi: 10.1515/jcim-2018-0130. [DOI] [PubMed] [Google Scholar]
- 2.Chen U., Hussain M.S., Mazumder T., Uddin S.N., Banik S. Neuropharmacological evaluation of methanolic extract of Costus speciosus Linn. rhizome in Swiss albino mice. Asian Pacific Journal of Tropical Biomedicine. 2019;9:217. [Google Scholar]
- 3.Krause K.R., Bear H.A., Edbrooke-Childs J., Wolpert M. What outcomes count? Outcomes measured for adolescent depression between 2007 and 2017. J. Am. Acad. Child Adolesc. Psychiatry. 2019;58:61–71. doi: 10.1016/j.jaac.2018.07.893. [DOI] [PubMed] [Google Scholar]
- 4.Krishnan V., Nestler E.J. The molecular neurobiology of depression. Nature. 2008;455:894–902. doi: 10.1038/nature07455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kalin N.H. The critical relationship between anxiety and depression. Am Psychiatric Assoc. 2020;177:365–367. doi: 10.1176/appi.ajp.2020.20030305. [DOI] [PubMed] [Google Scholar]
- 6.Islam A., Hussain M.S., Sen N., Abedin F., Millat M.S., Islam M.S., Das A., Kar A., Hossain M.M. Investigation of in vitro thrombolytic and anti-helminthic activity and in vivo anxiolytic and antidepressant potentiality with phytochemical nature of methanolic extract of Leucas lavandulifolia. Sustainable Chemistry and Pharmacy. 2017;6:61–66. [Google Scholar]
- 7.Raja S.N., Carr D.B., Cohen M., Finnerup N.B., Flor H., Gibson S., Keefe F.J., Mogil J.S., Ringkamp M., Sluka K.A., Song X.-J., Stevens B., Sullivan M.D., Tutelman P.R., Ushida T., Vader K. PAIN; 2020. The Revised International Association for the Study of Pain Definition of Pain: Concepts, Challenges, and Compromises; p. 161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Paliwal S.K., Sati B., Faujdar S., Sharma S. Studies on analgesic, anti-inflammatory activities of stem and roots of Inula cuspidata CB Clarke. Journal of traditional and complementary medicine. 2017;7:532–537. doi: 10.1016/j.jtcme.2016.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sen N., Bulbul L., Hussain M.S., Banik S., Choudhuri M.S.K. An in vivo study regarding analgesic and anxiolytic activity of methanolic extract of Typha elephantina Roxb. Phytomedicine. 2018;4:1–9. [Google Scholar]
- 10.Shojaii A., Motaghinejad M., Norouzi S., Motevalian M. Evaluation of anti-inflammatory and analgesic activity of the extract and fractions of Astragalus hamosus in animal models. Iran. J. Pharm. Res. (IJPR): IJPR. 2015;14:263. [PMC free article] [PubMed] [Google Scholar]
- 11.Guedes I.A., de Magalhães C.S., Dardenne L.E. Receptor–ligand molecular docking. Biophys rev. 2014;6:75–87. doi: 10.1007/s12551-013-0130-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Alam S., Emon N.U., Shahriar S., Richi F.T., Haque M.R., Islam M.N., Sakib S.A., Ganguly A. Pharmacological and computer-aided studies provide new insights into Millettia peguensis Ali (Fabaceae) Saudi Pharmaceut. J. 2020;28:1777–1790. doi: 10.1016/j.jsps.2020.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kothari S., Minda M., Tonpay S. Anxiolytic and antidepressant activities of methanol extract of Aegle marmelos leaves in mice. Indian J. Physiol. Pharmacol. 2010;54:318–328. [PubMed] [Google Scholar]
- 14.Zhang Z.-J. Therapeutic effects of herbal extracts and constituents in animal models of psychiatric disorders. Life Sci. 2004;75:1659–1699. doi: 10.1016/j.lfs.2004.04.014. [DOI] [PubMed] [Google Scholar]
- 15.Alamgir A. Therapeutic use of medicinal plants and their extracts. Springer. 2017;1 [Google Scholar]
- 16.Paterson R.R.M. Ganoderma–a therapeutic fungal biofactory. Phytochemistry. 2006;67:1985–2001. doi: 10.1016/j.phytochem.2006.07.004. [DOI] [PubMed] [Google Scholar]
- 17.Mohanta Y.K., Singdevsachan S.K., Parida U.K., Panda S.K., Mohanta T.K., Bae H. Green synthesis and antimicrobial activity of silver nanoparticles using wild medicinal mushroom Ganoderma applanatum (Pers.) Pat. from Similipal Biosphere Reserve, Odisha, India. IET Nanobiotechnol. 2016;10:184–189. doi: 10.1049/iet-nbt.2015.0059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Smith B.J., Sivasithamparam K. Morphological studies of Ganoderma (Ganodermataceae) from the australasian and pacific regions. Aust. Syst. Bot. 2003;16:487–503. [Google Scholar]
- 19.Loyd A.L., Richter B.S., Jusino M.A., Truong C., Smith M.E., Blanchette R.A., Smith J.A. Identifying the “mushroom of immortality”: assessing the Ganoderma species composition in commercial Reishi products. Front. Microbiol. 2018;9:1557. doi: 10.3389/fmicb.2018.01557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Luo Q., Di L., Dai W.-F., Lu Q., Yan Y.-M., Yang Z.-L., Li R.-T., Cheng Y.-X. Applanatumin A, a new dimeric meroterpenoid from Ganoderma applanatum that displays potent antifibrotic activity. Org. Lett. 2015;17:1110–1113. doi: 10.1021/ol503610b. [DOI] [PubMed] [Google Scholar]
- 21.Luo Q., Yang X.-H., Yang Z.-L., Tu Z.-C., Cheng Y.-X. Miscellaneous meroterpenoids from Ganoderma applanatum. Tetrahedron. 2016;72:4564–4574. [Google Scholar]
- 22.Elkhateeb W.A., Zaghlol G.M., El-Garawani I.M., Ahmed E.F., Rateb M.E., Abdel Moneim A.E. Ganoderma applanatum secondary metabolites induced apoptosis through different pathways: in vivo and in vitro anticancer studies. Biomed. Pharmacother. 2018;101:264–277. doi: 10.1016/j.biopha.2018.02.058. [DOI] [PubMed] [Google Scholar]
- 23.Li L., Li H., Peng X.-R., Hou B., Yu M.-Y., Dong J.-R., Li X.-N., Zhou L., Yang J., Qiu M.-H. (±)-Ganoapplanin, a pair of polycyclic meroterpenoid enantiomers from Ganoderma applanatum. Org. Lett. 2016;18:6078–6081. doi: 10.1021/acs.orglett.6b03064. [DOI] [PubMed] [Google Scholar]
- 24.Gao Z., Yuan F., Li H., Feng Y., Zhang Y., Zhang C., Zhang J., Song Z., Jia L. The ameliorations of Ganoderma applanatum residue polysaccharides against CCl4 induced liver injury. Int. J. Biol. Macromol. 2019;137:1130–1140. doi: 10.1016/j.ijbiomac.2019.07.044. [DOI] [PubMed] [Google Scholar]
- 25.Peng X., Li L., Dong J., Lu S., Lu J., Li X., Zhou L., Qiu M. Lanostane-type triterpenoids from the fruiting bodies of Ganoderma applanatum. Phytochemistry. 2019;157:103–110. doi: 10.1016/j.phytochem.2018.10.011. [DOI] [PubMed] [Google Scholar]
- 26.Leary S.L., Underwood W., Anthony R., Cartner S., Corey D., Grandin T., Greenacre C., Gwaltney-Brant S., McCrackin M., Meyer R. American Veterinary Medical Association Schaumburg; IL: 2013. AVMA Guidelines for the Euthanasia of Animals: 2013 Edition. [Google Scholar]
- 27.Hernandez J., Torres-Espin A., Navarro X. Adult stem cell transplants for spinal cord injury repair: current state in preclinical research. Curr. Stem Cell Res. Ther. 2011;6:273–287. doi: 10.2174/157488811796575323. [DOI] [PubMed] [Google Scholar]
- 28.Ghosh A., Banik S., Islam M.A. In vitro thrombolytic, anthelmintic, anti-oxidant and cytotoxic activity with phytochemical screening of methanolic extract of Xanthium indicum leaves. Bangladesh J. Pharmacol. 2015;10:854–859. [Google Scholar]
- 29.Gupta B., Dandiya P., Gupta M. A psycho-pharmacological analysis of behaviour in rats. Jpn. J. Pharmacol. 1971;21:293–298. doi: 10.1254/jjp.21.293. [DOI] [PubMed] [Google Scholar]
- 30.Hussain M.S., Ibrahim M., Hasan M.M., Aziz M.T., Suchi S.A., Uddin M.G. An in vivo study of the pharmacological activities of a methanolic acetate fraction of Pistia stratiotes L. An ethno‐medicinal plant used in Bangladesh, Animal models and experimental medicine. 2018;1:221–227. doi: 10.1002/ame2.12037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Shiotsuki H., Yoshimi K., Shimo Y., Funayama M., Takamatsu Y., Ikeda K., Takahashi R., Kitazawa S., Hattori N. A rotarod test for evaluation of motor skill learning. J. Neurosci. Methods. 2010;189:180–185. doi: 10.1016/j.jneumeth.2010.03.026. [DOI] [PubMed] [Google Scholar]
- 32.Emon N.U., Alam S., Rudra S., Riya S.R., Paul A., Hossen S.M.M., Kulsum U., Ganguly A. Antidepressant, anxiolytic, antipyretic, and thrombolytic profiling of methanol extract of the aerial part of Piper nigrum: in vivo, in vitro, and in silico approaches. Food Sci. Nutr. 2021:833–846. doi: 10.1002/fsn3.2047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Islam M.R., Naima J., Proma N.M., Hussain M.S., Uddin S.N., Hossain M.K. In-vivo and in-vitro evaluation of pharmacological activities of Ardisia solanacea leaf extract. Phytomedicine. 2019;5:1–11. [Google Scholar]
- 34.Nagandla K., De S. Restless legs syndrome: pathophysiology and modern management. Postgrad. Med. 2013;89:402–410. doi: 10.1136/postgradmedj-2012-131634. [DOI] [PubMed] [Google Scholar]
- 35.Emon N.U., Alam S., Rudra S., Chowdhury S., Rajbangshi J.C., Ganguly A. Evaluation of pharmacological potentials of the aerial part of Achyranthes aspera L.: in vivo, in vitro and in silico approaches. Adv Tradit Med. 2020;25:1–4. [Google Scholar]
- 36.Emon N.U., Kaiser M., Islam M., Kabir M.F.I., Jamir M., Uddin M.A.J., Islam M. Anxiolytic and thrombolytic investigation of methanol extract of Piper nigrum L. fruits and Sesamum indicum L. seeds. J. Adv. Biotechnol. Exp. Ther. 2020;3:158–164. doi: 10.1007/s13596-020-00528-5. [DOI] [Google Scholar]
- 37.Alam S., Emon N.U., Shahriar S., Richi F.T., Haque M.R., Islam M.N., Sakib S.A., Ganguly A. Saudi Pharmaceutical Journal; 2020. Pharmacological and Computer-Aided Studies Provide New Insights into Millettia Peguensis Ali (Fabaceae) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Emon N.U., Jahan I., Sayeed M.A. Investigation of antinociceptive, anti-inflammatory and thrombolytic activity of Caesalpinia digyna (Rottl.) leaves by experimental and computational approaches. Adv Tradit Med. 2020:1–9. [Google Scholar]
- 39.Gaur K., Rana A., Nema R., Kori M., Sharma C. Anti-inflammatory and analgesic activity of hydro-alcoholic leaves extract of Euphorbia neriifolia Linn. Asian J. Pharmaceut. Clin. Res. 2009;2:26–28. [Google Scholar]
- 40.Franzotti E., Santos C., Rodrigues H., Mourao R., Andrade M., Antoniolli A. Anti-inflammatory, analgesic activity and acute toxicity of Sida cordifolia L.(Malva-branca) J. Ethnopharmacol. 2000;72:273–277. doi: 10.1016/s0378-8741(00)00205-1. [DOI] [PubMed] [Google Scholar]
- 41.Gawel K., Jenda-Wojtanowska M., Gibula-Bruzda E., Kedzierska E., Filarowska J., Marszalek-Grabska M., Wojtanowski K., Komsta L., Talarek S., Kotlinska J. The influence of AMN082, metabotropic glutamate receptor 7 (mGlu7) allosteric agonist on the acute and chronic antinociceptive effects of morphine in the tail-immersion test in mice: comparison with mGlu5 and mGlu2/3 ligands. Physiol. Behav. 2018;185:112–120. doi: 10.1016/j.physbeh.2017.12.035. [DOI] [PubMed] [Google Scholar]
- 42.Goodsell D.S., Zardecki C., Di Costanzo L., Duarte J.M., Hudson B.P., Persikova I., Segura J., Shao C., Voigt M., Westbrook J.D. RCSB Protein Data Bank: enabling biomedical research and drug discovery. Protein Sci. 2020;29:52–65. doi: 10.1002/pro.3730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Harman C.A., Turman M.V., Kozak K.R., Marnett L.J., Smith W.L., Garavito R.M. Structural basis of enantioselective inhibition of cyclooxygenase-1 by S-alpha-substituted indomethacin ethanolamides. J. Biol. Chem. 2007;282:28096–28105. doi: 10.1074/jbc.M701335200. [DOI] [PubMed] [Google Scholar]
- 44.Lenaeus M.J., Burdette D., Wagner T., Focia P.J., Gross A. Structures of KcsA in complex with symmetrical quaternary ammonium compounds reveal a hydrophobic binding site. Biochemistry. 2014;53:5365–5373. doi: 10.1021/bi500525s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Coleman J.A., Green E.M., Gouaux E. X-ray structures and mechanism of the human serotonin transporter. Nature. 2016;532:334–339. doi: 10.1038/nature17629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Masters L., Eagon S., Heying M. Evaluation of consensus scoring methods for AutoDock Vina, smina and idock. J. Mol. Graph. Model. 2020;96:107532. doi: 10.1016/j.jmgm.2020.107532. [DOI] [PubMed] [Google Scholar]
- 47.Hasan I., Hussain M.S., Millat M.S., Sen N., Rahman M.A., Rahman M.A., Islam S., Moghal M.M.R. Ascertainment of pharmacological activities of Allamanda neriifolia Hook and Aegialitis rotundifolia Roxb used in Bangladesh: an in vitro study. Journal of traditional and complementary medicine. 2018;8:107–112. doi: 10.1016/j.jtcme.2017.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Socala K., Nieoczym D., Grzywnowicz K., Stefaniuk D., Wlaz P. Evaluation of anticonvulsant, antidepressant-, and anxiolytic-like effects of an aqueous extract from cultured mycelia of the Lingzhi or Reishi medicinal mushroom Ganoderma lucidum (Higher Basidiomycetes) in mice. Int. J. Med. Mushrooms. 2015;17 doi: 10.1615/intjmedmushrooms.v17.i3.10. [DOI] [PubMed] [Google Scholar]
- 49.Sánchez C. Food bioactives; Springer: 2017. Bioactives from Mushroom and Their Application; pp. 23–57. [Google Scholar]
- 50.Khan M.A., Tania M. Nutritional and medicinal importance of Pleurotus mushrooms: an overview. Food Rev. Int. 2012;28:313–329. [Google Scholar]
- 51.Kolawole O., Makinde J. Central nervous system depressant activity of Russelia equisetiformis. Niger. J. Physiol. Sci. 2007;22 [PubMed] [Google Scholar]
- 52.Rey A.A., Purrio M., Viveros M.-P., Lutz B. Biphasic effects of cannabinoids in anxiety responses: CB1 and GABA B receptors in the balance of GABAergic and glutamatergic neurotransmission. Neuropsychopharmacology. 2012;37:2624–2634. doi: 10.1038/npp.2012.123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Momin M.A.M., Bellah S.F., Rahman S.M.R., Rahman A.A., Murshid G.M.M., Emran T.B. Phytopharmacological evaluation of ethanol extract of Sida cordifolia L. roots. Asian Pac. J. Trop. Biomed. 2014;4:18–24. doi: 10.1016/S2221-1691(14)60202-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Amin K.R., Uddin M.G., Rashid M.M.O., Sharmin T. New insight in neuropharmacological activities of Dioscorea alata. Discovery Phytomedicine. 2018;5:1–6. [Google Scholar]
- 55.Yadav G., Garg V.K., Thakur N., Khare P. Locomotor activity of methanolic extract of Saraca indica bark. Adv. Biol. Res. 2013;7:1–3. [Google Scholar]
- 56.Kumar B.A., Lakshman K., Velmurugan C., Sridhar S., Gopisetty S. Antidepressant activity of methanolic extract of Amaranthus spinosus. Basic Clin. Neurosci. 2014;5:11. [PMC free article] [PubMed] [Google Scholar]
- 57.Trongsakul S., Panthong A., Kanjanapothi D., Taesotikul T. The analgesic, antipyretic and anti-inflammatory activity of Diospyros variegata Kruz. J. Ethnopharmacol. 2003;85:221–225. doi: 10.1016/s0378-8741(03)00020-5. [DOI] [PubMed] [Google Scholar]
- 58.Uche F., Aprioku J. The Phytochemical Constituents, Analgesic and Anti-inflammatory effects of methanol extract of Jatropha curcas leaves in Mice and Wister albino rats. J. Appl. Sci. Environ. Manag. 2008;12 [Google Scholar]
- 59.Bachhav R., Gulecha V., Upasani C. Analgesic and anti-inflammatory activity of Argyreia speciosa root. Indian J. Pharmacol. 2009;41:158. doi: 10.4103/0253-7613.56066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Toma W., Gracioso J.d.S., Hiruma-Lima C., Andrade F.d., Vilegas W., Brito A.S. Evaluation of the analgesic and antiedematogenic activities of Quassia amara bark extract. J. Ethnopharmacol. 2003;85:19–23. doi: 10.1016/s0378-8741(02)00334-3. [DOI] [PubMed] [Google Scholar]
- 61.Ramabadran K., Bansinath M. A critical analysis of the experimental evaluation of nociceptive reactions in animals. Pharmaceut. Res. 1986;3:263–270. doi: 10.1023/A:1016355200944. [DOI] [PubMed] [Google Scholar]
- 62.Śledź P., Caflisch A. Protein structure-based drug design: from docking to molecular dynamics. Curr. Opin. Struct. Biol. 2018;48:93–102. doi: 10.1016/j.sbi.2017.10.010. [DOI] [PubMed] [Google Scholar]
- 63.Emon N.U., Alam S., Rudra S., Chowdhury S., Rajbangshi J.C., Ganguly A. Evaluation of pharmacological potentials of the aerial part of Achyranthes aspera L.: in vivo, in vitro and in silico approaches. Adv Tradit Med. 2020:1–14. [Google Scholar]
- 64.Fajemiroye J.O., da Silva D.M., de Oliveira D.R., Costa E.A. Treatment of anxiety and depression: medicinal plants in retrospect. Fund. Clin. Pharmacol. 2016;30:198–215. doi: 10.1111/fcp.12186. [DOI] [PubMed] [Google Scholar]
- 65.Ndebele P. The declaration of Helsinki, 50 years later. Jama. 2013;310:2145–2146. doi: 10.1001/jama.2013.281316. [DOI] [PubMed] [Google Scholar]
- 66.Emanuel E.J. Reconsidering the declaration of Helsinki. Lancet. 2013;381:1532–1533. doi: 10.1016/s0140-6736(13)60970-8. [DOI] [PubMed] [Google Scholar]