Supplemental Digital Content is available in the text.
Keywords: covid-19, homeostasis, inflammation mediators, macrophages, monocyte, neutrophils, omega-3 fatty acids, phenotype, plasma
Rationale:
Resolution mechanisms are central in both the maintenance of homeostasis and the return to catabasis following tissue injury and infections. Among the proresolving mediators, the essential fatty acid-derived specialized proresolving lipid mediators (SPM) govern immune responses to limit disease severity. Notably, little is known about the relationship between the expression and activity of SPM pathways, circulating phagocyte function and disease severity in patients infected with the novel severe acute respiratory syndrome coronavirus 2 leading to coronavirus disease 2019 (COVID-19).
Objective:
Herein, we investigated the link between circulating SPM concentrations and phagocyte activation status and function in patients with COVID-19 (n=39) compared with healthy (n=12) and post-COVID-19 (n=8) volunteers.
Methods and Results:
Lipid mediator profiling demonstrated that plasma SPM concentrations were upregulated in patients with mild COVID-19 and are downregulated in those with severe disease. SPM concentrations were correlated with both circulating phagocyte activation status and function. Perturbations in plasma SPM concentrations and phagocyte activation were retained after the resolution of COVID-19 clinical symptoms. Treatment of patients with dexamethasone upregulated both the expression of SPM biosynthetic enzymes in circulating phagocytes and plasma concentration of these mediators. Notably, incubation of phagocytes from patients with COVID-19 with SPM rectified their phenotype and function. This included a downregulation in the expression of activation markers, a decrease in the tissue factor and inflammatory cytokine expression, and an upregulation of bacterial phagocytosis.
Conclusions:
The present findings suggest that downregulation of systemic SPM concentrations is linked with both increased disease severity and dysregulated phagocyte function. They also identify the upregulation of these mediators by dexamethasone as a potential host protective mechanism elicited by this drug in patients with COVID-19. Taken together, our findings elucidate a role for altered resolution mechanisms in the disruption of phagocyte responses and the propagation of systemic inflammation in COVID-19.
In This Issue, see p 455
The inflammatory response is an essential protective mechanism that evolved to safeguard the host from invading pathogens and facilitate the restoration of tissue function through repair and regeneration of damaged tissues.1–4 This finely tuned response becomes unravelled in disease, often resulting in disseminated inflammation. One such example is coronavirus disease 2019 (COVID-19) caused by the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2).5 Current evidence suggests that in a large part of the population, SARS-CoV-2 infections result in either no or mild symptoms that do not require hospitalization. However, in the remainder of cases, SARS-CoV-2 results in moderate to severe disease requiring hospitalization, with a subset of these patients becoming critically ill and requiring ventilation support.
COVID-19 is characterized by a number of clinical presentations that in severe cases include pneumonia, acute respiratory distress syndrome and multiple organ failure.5 This disseminated inflammatory response is thought to involve alveolar damage that can lead to cardiovascular complications and multiorgan failure. A significant body of effort has now gone into understanding the mechanisms underpinning this dysregulated systemic inflammatory response, since insights into these mechanisms may provide novel therapeutic leads for the treatment of hospitalized patients. This notion is underpinned by results obtained in the RECOVERY trial, which demonstrated a significant reduction in mortality in critically ill patients treated with the anti-inflammatory corticosteroid dexamethasone.6
Initial efforts into detailing these inflammatory mechanisms found a marked upregulation of circulating proinflammatory cytokines as well as an increased inflammatory phenotype of innate immune cells, primarily monocytes, in COVID-19 patients with severe disease.7–12 This phenotype is reminiscent of the dysregulated resolution responses observed following infections by other viral and bacterial pathogens.1–4 These findings suggest that in addition to an increase in proinflammatory mediator production there may also be disruptions in resolution pathways in patients with COVID-19.
Resolution mechanisms are orchestrated by a number of endogenous autacoids that include the omega-3 fatty acid-derived specialized proresolving lipid mediators (LM; SPM).4,13 These molecules are classified into 4 distinct families: lipoxins, resolvins, protectins, and maresins, which are produced via the stereoselective conversion of essential fatty acids. SPM display both host-directed as well as antiviral actions during viral infections. For example, the docosahexaenoic acid (DHA)-derived resolvin D1 (RvD1) and its precursor 17-hydroxy-DHA regulate B-cell response during H1N1 infections in mice by promoting an antibody class switch.14 The protectin (PD) family of mediators regulate viral propagation by inhibiting intracellular viral RNA transport mechanisms.15 While the eicosapentaenoic acid-derived RvE1 reduces effector T cell and neutrophil-mediated propagation of inflammation during herpes simplex virus infections.16 Furthermore, recent studies demonstrate that circulating concentrations of these mediators are linked with disease outcome in sepsis and in treatment responsiveness in humans with chronic inflammatory conditions such as rheumatoid arthritis.17,18
Given the immunoregulatory actions of SPM and the potential diagnostic and prognostic value of measuring peripheral levels of these molecules, we sought to evaluate whether SPM pathways are altered in patients with COVID-19. We also investigated the impact that disruptions in these pathways may have on phagocyte biology and disease propagation in patients with COVID-19.
Methods
Data Availability
All data supporting the findings of this study are available from the corresponding author upon reasonable request. Please see the Data Supplement for the Expanded Materials and Methods and Major Resources Table.
Results
Altered Peripheral Blood LM Profiles in Patients With COVID-19 Pneumonia
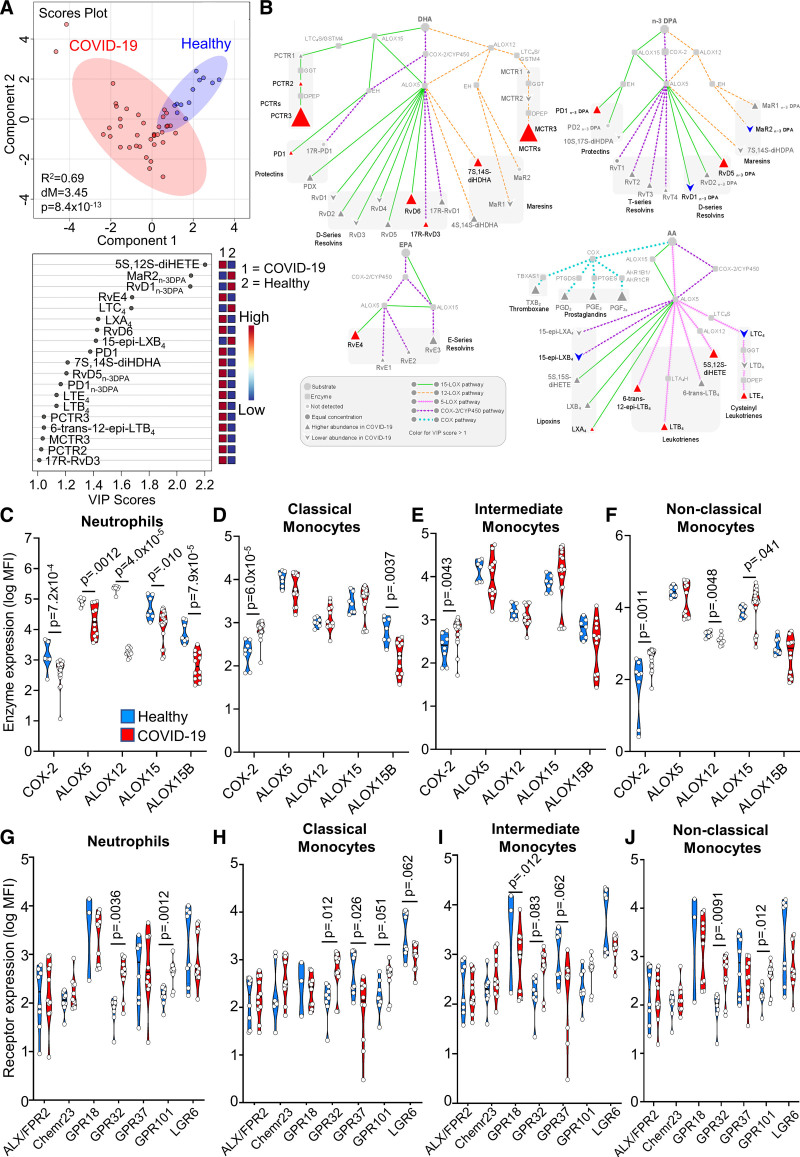
To evaluate whether SPM concentrations are differentially regulated in patients with COVID-19, we first assessed plasma LM profiles in hospitalized patients with COVID-19 (n=38) and compared these with LM profiles obtained from healthy volunteers with no clinical suggestion of COVID-19 infection (n=12; see Table I in the Data Supplement for clinical characteristics). Using liquid chromatography tandem mass spectrometry, we identified LM from all 4 bioactive metabolomes in both hospitalized patients and healthy volunteers. We then assessed their relative levels using Partial Least Squares Discriminant analysis (PLS-DA), a dimensionality-reducing multivariate analysis that creates a linear regression model accounting for multicollinearity and identifies the relationship between samples based on LM concentrations. This demonstrated that LM profiles in hospitalized patients with COVID-19 were different from those found in plasma from healthy volunteers, as illustrated by a separate clustering of LM profiles between the 2 groups (Figure 1A and Table II in the Data Supplement). Assessment of Variable Importance in Projection scores, which identify those mediators that contribute most to the observed separation between the 2 groups, showed that 19 mediators displayed a Variable Importance in Projection score >1 (Figure 1A). Intriguingly, the majority of mediators found to be differentially regulated between patients with COVID-19 and healthy volunteers belonged to the SPM family. This included an upregulation of SPM from the DHA, n-3 docosapentaenoic acid (DPA), and eicosapentaenoic acid bioactive metabolomes in patients with COVID-19, such as DHA-derived maresin conjugates in tissue regeneration (MCTR) 3, protectin conjugates in tissue regeneration (PCTR) 3, and RvD6, and n-3 DPA-derived RvD5n-3 DPA and PD1n-3 DPA.
Figure 1.
Upregulation of specialized proresolving mediator (SPM) pathways in patients with coronavirus disease 2019 (COVID-19). Peripheral blood was collected from patients with COVID-19 or healthy volunteers and (A and B) plasma lipid mediators were identified and quantified using liquid chromatography tandem mass spectrometry-based mediator profiling (n=38 patients with COVID-19, n=12 healthy volunteers). A, Partial least squares discriminant analysis (PLS-DA) was performed on identified mediators. Top: Scores plot, shaded area represents 95% CI; R2, coefficient-of-determination; P, P value from Hotelling T2 test; dM, Mahalanobis distance between groups. Bottom: Variable Importance in Projection (VIP) scores plot. B, Analysis highlighting mediators with VIP scores >1 in PLS-DA and their biosynthetic pathways. C–J, Whole blood from healthy volunteers and patients with COVID-19 was incubated with lineage-marker antibodies against neutrophils (CD16+) and monocyte subsets (classical CD14++CD16+, intermediate CD14++CD16++, nonclassical CD14+CD16++) in combination with antibodies against (C–F) lipid mediator biosynthetic enzymes or (G–J) SPM receptors, and their expression was evaluated using flow cytometry. For (C–F), n=19 patients with COVID-19 (for COX-2, ALOX15), n=12 patients with COVID-19 (for ALOX5 [5-lipoxygenase], ALOX12, ALOX15B), and n=7 healthy volunteers. For (G–J), n=11 patients with COVID-19 (except for GPR18 and GPR37 where n=10) and n=8 healthy volunteers (except for GPR18 where n=3 healthy volunteers). Statistical differences between healthy and COVID-19 groups were established using Mann-Whitney test for each protein, and raw P values are displayed. GPCR indicates G protein-coupled receptor; MCTR, maresin conjugates in tissue regeneration; PCTR, protectin conjugates in tissue regeneration.
To obtain insights into potential changes in LM biosynthetic pathways in patients with COVID-19, we next performed a pathway analysis on mediators that displayed the greatest differences in concentrations between the 2 groups (ie, Variable Importance in Projection scores >1). This demonstrated an upregulation of ALOX5 (5-lipoxygenase)–ALOX15 interaction products as highlighted by increases in RvD6, RvD5n-3 DPA, and RvE4, and a decrease in CYP450 (cytochrome P450)–ALOX5 interaction products evidenced by a downregulation in 15-epi-LXB4 in patients with COVID-19 when compared with healthy volunteers (Figure 1B). Together these findings provide evidence for altered LM, in particular SPM, levels in patients with COVID-19.
Differential Regulation of SPM Biosynthetic Enzymes and Receptors in Peripheral Blood Phagocytes From Patients With COVID-19
Phagocytes play a central role in LM production.19,20 Having identified changes in plasma concentrations of mediators from both ALOX and COX biosynthetic pathways, we next investigated whether these enzymes were differentially expressed in peripheral blood phagocytes from patients with COVID-19 when compared with those from healthy volunteers. Flow cytometric analysis of peripheral blood neutrophils from patients with COVID-19 demonstrated a significant downregulation in the expression of all LM biosynthetic enzymes analyzed (Figure 1C, Figure IA in the Data Supplement). On the contrary, in peripheral blood monocytes we observed an overall trend towards increased expression of these enzymes, which reached statistical significance for COX-2 in all 3 monocyte subsets and for ALOX15 in nonclassical monocytes (Figure 1D through 1F). Together these findings uncover a differential regulation in the expression of LM biosynthetic enzymes in circulating phagocytes from patients with COVID-19.
The biological actions of SPM are mediated by cognate GPCR (G protein-coupled receptors).21,22 Thus, we next evaluated whether phagocytes from patients with COVID-19 displayed altered expression of SPM receptors. Flow cytometric assessment of SPM receptors in peripheral blood neutrophils demonstrated a significant upregulation of GPR32 and GPR101 (Figure 1G). The former receptor mediates the protective actions of RvD1, 17R-RvD1, RvD3, 17R-RvD3, RvD5, 15-epi-LXA4, and LXA4,21,22 whereas the latter is the cognate receptor for RvD5n-3 DPA.23 GPR32 was upregulated in all 3 monocytes subsets, while GPR101 was upregulated in classical and nonclassical monocytes (Figure 1H through 1J). We also observed a significant downregulation of GPR18, the RvD2 receptor,24 as well as a trend towards the downregulation of PD1 receptor GPR3725 in intermediate monocytes and MaR1 receptor LGR626 in all 3 monocytes subsets (Figure 1H through 1J). Together, these findings lend support to the hypothesis that resolution mechanisms are altered in peripheral blood phagocytes from patients with COVID-19.
Peripheral Blood Phagocytes From Patients With COVID-19 Display an Altered Activation Status and Function
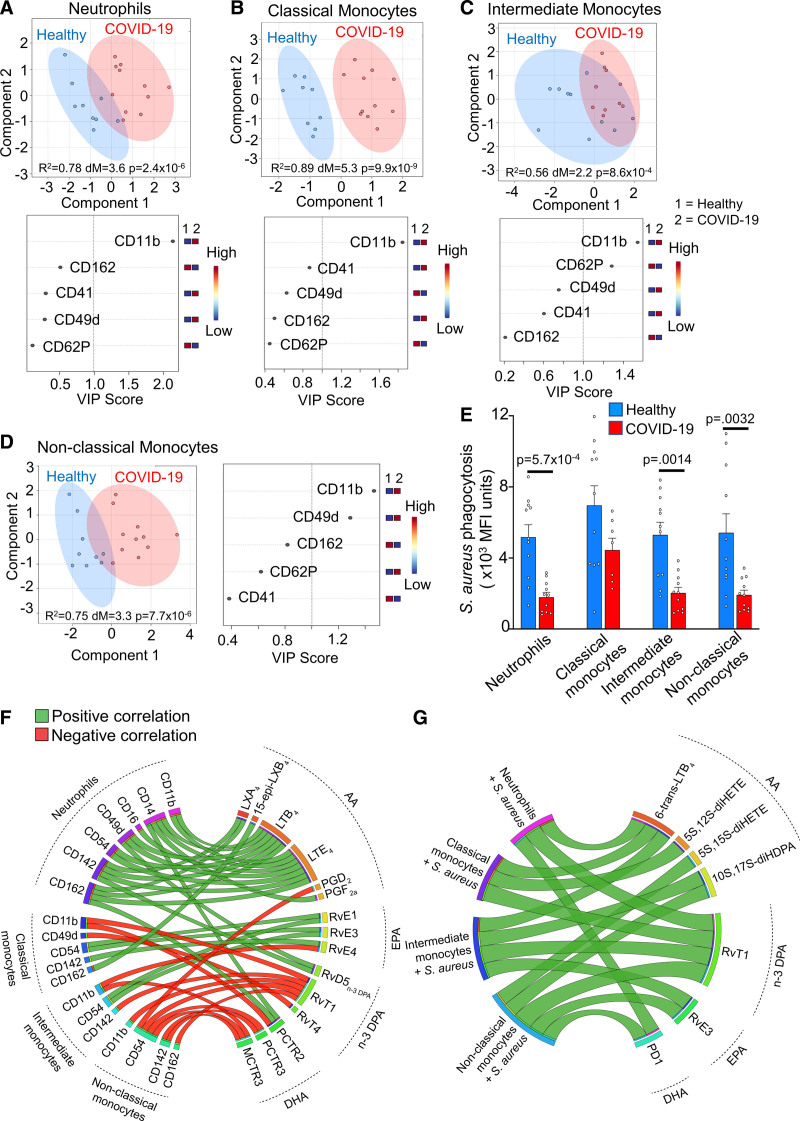
We next assessed whether these changes in SPM pathways were reflected in a dysregulation of circulating phagocyte activation status and function. For this purpose, we evaluated the expression of proteins linked with the activation status of circulating phagocytes. PLS-DA unveiled an overall shift in the activation status of all 4 phagocyte subsets evaluated, as demonstrated by a separation of healthy volunteer and patient with COVID-19 clusters (Figure 2A through 2D). In all phagocyte subsets, this phenotypic shift was primarily driven by upregulation of the integrin Cluster of Differentiation (CD)11b (Figure 2A through 2D). Additionally, we observed a downregulation in the expression of CD62P, a marker of platelet-monocyte heterotypic aggregates19 in intermediate monocytes (Figure 2C). Whereas, in nonclassical monocytes we observed a marked upregulation of the integrin CD49d (Figure 2D).
Figure 2.
Upregulation of activation markers and downregulation of S. aureus phagocytosis in circulating phagocytes from patients with coronavirus disease 2019 (COVID-19). A–D, Partial least squares discriminant analysis (PLS-DA) scores plots with 95% CI and Variable Importance in Projection (VIP) scores for activation markers on circulating phagocytes from healthy volunteers (n=9) and patients with COVID-19 (n=11) assessed by flow cytometry. E, S. aureus phagocytosis by peripheral blood neutrophils and monocyte subsets from healthy volunteers (n=11) and patients with COVID-19 (n=11) evaluated using flow cytometry. Statistical differences between healthy and COVID-19 groups were established using Mann-Whitney test for each cell type and raw P values are displayed. F–G, Circos plots of Pearson correlation coefficients between patient with COVID-19 (n=11) plasma lipid mediator (LM) levels and (F) phagocyte activation markers or (G) bacterial phagocytosis. Connecting bands represent statistically significant correlations (P<0.05), with the width of the band being proportional to the strength of the correlation (range, −0.66 to 0.83) and band color representing positive (green) or negative (red) correlations. AA indicates arachidonic acid; DHA, docosahexaenoic acid; DPA, docosapentaenoic acid; and EPA, eicosapentaenoic acid.
Several studies found that patients with COVID-19 pneumonia may also develop secondary bacterial infections.27,28 Thus, we next evaluated whether phagocytes from patients with COVID-19 displayed altered phagocytic function. We evaluated the ability of peripheral blood phagocytes to uptake and kill Staphylococcus aureus as an exemplar bacterium of clinical relevance using bacterial particles labeled with a pH-sensitive fluorophore to monitor uptake and phagolysosome acidification, a key step in bacterial killing. This analysis demonstrated a significant downregulation in fluorescence levels in neutrophils, intermediate monocytes and nonclassical monocytes from patients with COVID-19 incubated with fluorescently labeled S. aureus (Figure 2E).
To evaluate whether the variations in SPM concentrations observed in patients with COVID-19 were linked to changes in peripheral blood phagocyte activation status and function, we next conducted a correlation analysis (Figure 2F). This analysis demonstrated that arachidonic acid-derived mediator levels, such as those of leukotriene B4 (LTB4), and LTE4, were generally positively correlated with activation marker expression on neutrophils, while n-3 DPA and DHA-derived mediators such as MCTR3, PCTR3, and RvT1 were negatively correlated with expression of activation markers on all three monocytes subsets. MCTR3 and PCTR3 were also significantly negatively correlated with CD54 (ICAM-1) and CD142 (tissue factor) expression on nonclassical monocytes, while RvT1 was negatively correlated with expression of integrin CD11b on classical and nonclassical monocytes (Figure 2F). Plasma SPM concentrations were also linked with the ability of different phagocyte subsets to uptake bacteria. Here, we observed a significant positive correlation between plasma PD1, RvT1, RvE3, and 10S, 17S-diHDPA and the ability of neutrophils and monocytes to uptake S. aureus (Figure 2G). Taken together, these findings suggest that changes in SPM pathways are linked with an alteration in phagocyte activation status and function in patients with COVID-19.
Downregulation of SPM in Patients With Severe COVID-19 Pneumonia
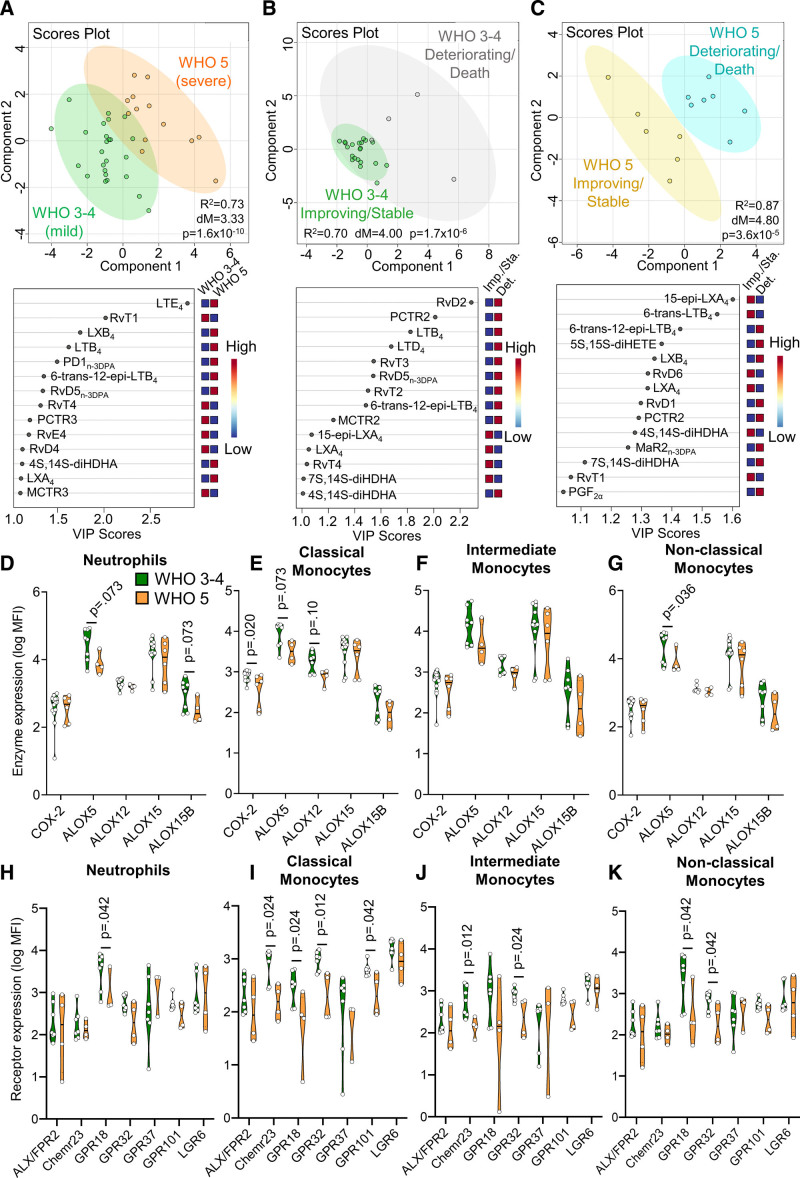
We next investigated whether plasma LM concentrations differed with increasing disease severity in patients with COVID-19. For this purpose, we assessed plasma LM concentration in hospitalized patients with mild disease and compared them with those with severe disease based on their WHO Ordinal Scale of COVID-19 Disease Severity at time of sample collection. Using PLS-DA, we found a marked shift in plasma LM concentrations in patients with a WHO scale of 3 or 4 (WHO 3–4), which indicates hospitalized patients with mild disease symptoms, when compared with those with a WHO scale of 5 (WHO 5), indicative of more severe pneumonia requiring noninvasive ventilation or high-flow oxygen (see Table III in the Data Supplement for clinical characteristics). This shift in plasma LM profiles was due to an overall downregulation in DHA-derived SPM such as PCTR3 and MCTR3 in patients with severe disease, which was coupled with an upregulation of arachidonic acid-derived LM, including the potent leukocyte chemoattractant LTB4, and LTE4, the downstream product of the proinflammatory smooth muscle-contractants LTC4 and LTD4 (Figure 3A and Table IV in the Data Supplement).
Figure 3.
Disruption of specialized proresolving mediator (SPM) pathways with increased disease severity in patients with coronavirus disease 2019 (COVID-19). Peripheral blood was collected from patients with COVID-19 with mild (WHO scale 3–4; WHO 3–4) or severe disease (WHO scale 5; WHO 5). A–C, Plasma lipid mediators were identified and quantified using liquid chromatography tandem mass spectrometry based lipid mediator profiling, and partial least squares discriminant analysis (PLS-DA) was performed on identified mediators for the indicated patient sub-groups. Top: Scores plot with 95% CI. Bottom: Variable Importance in Projection scores plot. For (A), n=25 WHO 3–4 patients and n=13 WHO 5 patients; for (B), n=21 WHO 3–4 improving/stable patients and n=4 WHO 3–4 deteriorating/death patients; for (C), n=6 WHO 5 improving/stable patients and n=7 WHO 5 deteriorating/death patients. D–K, Whole blood from patients with COVID-19 was incubated with lineage-marker antibodies for neutrophils and monocyte subsets in combination with antibodies against (D–G) lipid mediator biosynthetic enzymes or (H–K) SPM receptors and their expression was evaluated using flow cytometry. For (D–G), n=14 WHO 3–4 patients (for COX-2, ALOX15), n=8 WHO 3–4 patients (for ALOX5 [5-lipoxygenase], ALOX12, ALOX15B), n=6 WHO 5 patients (for COX-2, ALOX15), n=6 WHO 5 patients (for ALOX5, ALOX12, ALOX15B). For (H–K), n=7 WHO 3–4 patients and n=4 WHO 5 patients (except for GPR37 where n=3 WHO 5 patients). Statistical differences between WHO 3–4 and WHO 5 patients with COVID-19 were established using Mann-Whitney test for each parameter and raw P values are displayed.
To investigate the impact of disease severity on LM profiles in more detail, we evaluated LM profiles in patients with mild (WHO 3–4) and severe disease (WHO 5) according to their disease trajectory. Here, we separated patients based on whether their symptoms were improving or stable versus those that were either clinically deteriorating at time of sample collection or would ultimately succumb to the disease (referred to as deteriorating; Figure 3B and 3C, Tables III, VII, and VIII in the Data Supplement). PLS-DA demonstrated a shift in LM profiles between clinically improving/stable patients compared with deteriorating patients in patients with mild disease (Figure 3B). Notably, this distinction in LM profiles was observed to be even more pronounced in patients with severe disease (Figure 3C). For patients with mild disease, differences between improving/stable and deteriorating groups were primarily linked with higher concentrations of both SPM (ie, RvD2, PCTR2, RvT2, RvT3, and RvD5n-3 DPA) and leukotrienes (ie, LTB4 and LTD4) in patients who were clinically deteriorating (Figure 3B). On the contrary, differences between these 2 groups in patients with severe disease were primarily driven by higher SPM levels including, 15-epi-LXA4, RvD6, LXA4, and RvT1 in patients that were clinically improving or stable (Figure 3C).
To explore the mechanisms underlying the altered SPM production in patients with severe COVID-19, we analyzed the biosynthetic pathways of mediators with a Variable Importance in Projection score >1 in the PLS-DA (shown in Figure 3A). This analysis identified a downregulation in the levels of DHA and eicosapentaenoic acid-derived ALOX5-ALOX15 interaction products, including RvD4 and RvE4. A reduction that was coupled with an increase in LXA4 and LXB4 concentrations, the arachidonic acid-derived products from these enzymes, in patients at WHO scale 5 (Figure IB in the Data Supplement). We also observed a decrease in peptide-lipid conjugated SPM (PCTR3 and MCTR3), suggesting that there was a reduction in activity of either the initiating ALOX enzymes in each of these pathways (ALOX15 or ALOX12, respectively) or their shared downstream biosynthetic enzymes. Notably, these downstream biosynthetic enzymes also produce cystenyl-leukotrienes such as LTE4, which was increased in patients with severe disease, thereby suggesting a shift in the activity of these enzymes towards the formation of proinflammatory mediators (Figure IB in the Data Supplement).
We next assessed the expression of SPM biosynthetic enzymes in circulating phagocytes. A trend for reduced expression of all enzymes under analysis with increased disease severity could be observed across all 4 phagocyte subsets, which reached statistical significance for ALOX5 and ALOX15B in neutrophils, COX-2 and ALOX5 in classical monocytes, and ALOX5 in nonclassical monocytes (Figure 3D through 3G). These results suggest that the differential regulation of SPM production observed in plasma from severe patients was in part linked with a downregulation of SPM biosynthetic enzyme expression in circulating phagocytes.
We next evaluated whether SPM receptor expression was also altered with increased disease severity. Here, we found reduced expression of multiple SPM receptors on circulating phagocytes from patients with severe COVID-19. This included a significant downregulation of GPR18 on neutrophils, classical monocytes, and nonclassical monocytes, as well as reduced expression of ChemR23, the receptor for RvE1 and RvE2, on classical and intermediate monocytes. We also observed a significant downregulation in GPR32 on intermediate and nonclassical monocytes, and a decrease in GPR101 on classical monocytes in patients with severe disease (Figure 3H through 3K). Together, these findings suggest that resolution pathways become dysregulated in patients with COVID-19 with severe disease.
Altered Resolution Pathways Persist After Resolution of COVID-19 Clinical Symptoms
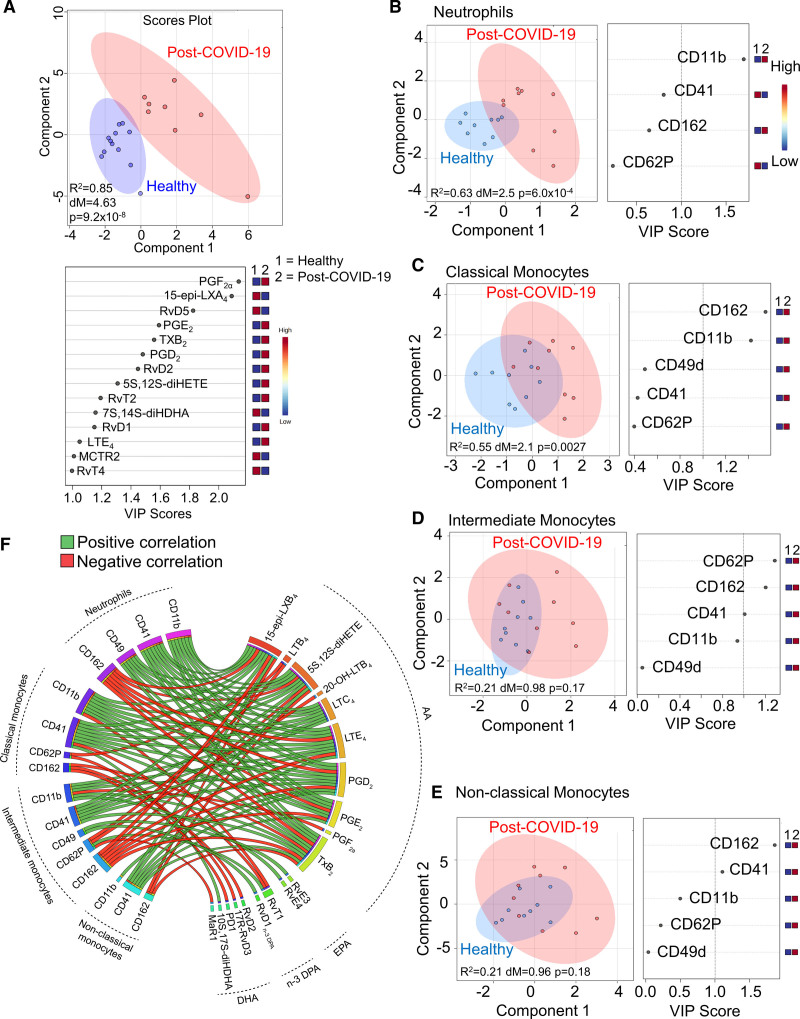
Having observed a marked shift in LM profiles from patients with active COVID-19 pneumonia, we next evaluated whether plasma LM profiles returned to baseline after the resolution of COVID-19 clinical symptoms (referred to as post-COVID-19). We collected peripheral blood from post-COVID-19 volunteers between 12 and 25 days after the resolution of clinical symptoms (n=8) and compared their plasma LM profiles with those obtained from healthy volunteers with no clinical suggestion of COVID-19 infection (n=12; see Table I in the Data Supplement for demographics). PLS-DA demonstrated that LM profiles obtained from post-COVID-19 volunteers were markedly different from those obtained from healthy volunteers (Figure 4A and Table II in the Data Supplement). This separation was primarily driven by differential regulation of 14 mediators that included both proinflammatory eicosanoids, such as prostaglandin (PG)E2, PGF2α, and LTE4 and SPM such as, RvD1, RvD2, and RvT2 (Figure 4A).
Figure 4.
Specialized proresolving mediator (SPM) pathways remain dysregulated after the resolution of clinical symptoms in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infected patients. Peripheral blood was collected from volunteers after the resolution of coronavirus disease 2019 (COVID-19) clinical symptoms (post-COVID-19) or healthy volunteers. A, Plasma lipid mediators were identified and quantified using liquid chromatography tandem mass spectrometry based lipid mediator profiling, and partial least squares discriminant analysis (PLS-DA) was performed on identified mediators (n=12 healthy volunteers, n=8 post-COVID-19 volunteers). Top: Scores plot with 95% CI. Bottom: Variable Importance in Projection (VIP) scores plot. B–E, PLS-DA scores plots and VIP scores for activation markers on circulating phagocytes assessed by flow cytometry of peripheral blood from healthy (n=9) and post-COVID-19 (n=8) volunteers. F, Circos plots of Pearson correlation coefficients between post-COVID-19 volunteer (n=8) plasma lipid mediator (LM) levels and circulating phagocyte activation markers. Connecting bands represent statistically significant correlations (P<0.05), with the width of the band being proportional to the strength of the correlation (range, −0.93 to 0.93) and band color representing positive (green) or negative (red) correlations.
Examination of LM biosynthetic pathways indicated an upregulation of COX activity in post-COVID-19 volunteers, as indicated by an increase in both proinflammatory (ie, PGD2, PGE2, and PGF2a) and proresolving (RvT2) COX-derived products in these volunteers. We also found an overall increase in ALOX5-ALOX15 interaction products, including RvD1 and RvD2, and an increase in the ALOX5 products 5S, 12S-diHETE, and LTE4 in post-COVID-19 volunteers when compared with healthy volunteers (Figure IIA in the Data Supplement). Intriguingly, expression of these biosynthetic enzymes in circulating phagocytes from post-COVID-19 patients was essentially the same as that observed in healthy volunteers (Figure IIC through IIF in the Data Supplement). Thus, these results suggest that the alterations in resolution pathways observed in patients with COVID-19 can persist even after the subsidence of clinical symptoms, and might arise from changes in activity of the LM biosynthetic enzymes.
Changes in Peripheral Blood Phagocyte Activation Status Partially Persist After the Resolution of COVID-19 Clinical Symptoms
We next assessed whether the observed changes in SPM pathways were linked with changes in peripheral blood phagocyte activation status and function in post-COVID-19 volunteers. Flow cytometric analysis demonstrated that post-COVID-19 neutrophils and classical monocytes displayed an activated phenotype as observed by marked shifts in the clusters representing these cells versus the clusters representing cells from healthy volunteers. This separation was primarily linked with an upregulation in CD11b on these cells (Figure 4B and 4C), an observation also previously made with cells from patients with COVID-19 pneumonia (Figure 2A through 2D). Conversely, the activation status of intermediate and nonclassical monocyte from post-COVID-19 volunteers was not markedly different from healthy volunteer cells (Figure 4D and 4E). Comparison of phagocytic ability of cells isolated from post-COVID-19 volunteers demonstrated that phagocytes from these volunteers displayed an essentially similar ability to uptake fluorescently labeled S. aureus when compared with healthy volunteers (Figure IIB in the Data Supplement). We also did not observe marked differences in SPM receptor expression on circulating phagocytes from post-COVD-19 volunteers when compared with healthy volunteers (Figure IIG through IIJ in the Data Supplement).
We next evaluated whether changes in phagocyte activation observed in post-COVID-19 volunteers were correlated with the observed changes in peripheral blood SPM concentrations (Figure 4F). Here, we found that neutrophil and monocyte expression of the platelet-leukocyte heterotypic aggregate marker CD41 and integrin CD11b positively correlated with a number of arachidonic acid-derived leukotrienes and prostaglandins, while the SPM 15-epi-LXB4, 17R-RvD3, and MaR1 negatively correlated with CD62P, another marker of leukocyte-platelet aggregates (Figure 4F). Taken together, these findings indicate that alterations in phagocyte activation status and peripheral LM profiles persist after the resolution of clinical symptoms in SARS-CoV-2 infected individuals.
Dexamethasone Upregulates Peripheral Blood SPM Concentrations in Patients With COVID-19
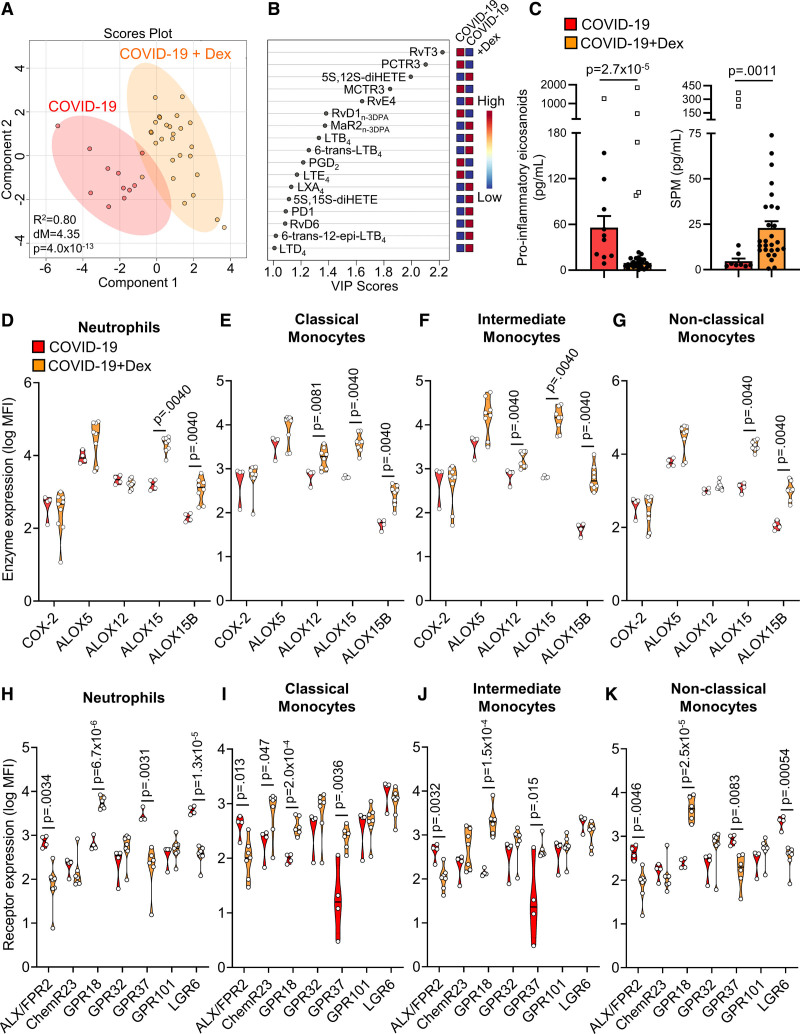
Recent studies demonstrate that dexamethasone upregulates SPM formation in experimental allergic inflammation.29 Therefore, we questioned whether this corticosteroid also regulated SPM formation in patients with COVID-19. Plasma LM concentrations were measured in patients with COVID-19 treated with or without 6 mg/d of dexamethasone (see Table V in the Data Supplement for patient information). Analysis of plasma LM profiles using PLS-DA demonstrated a distinct clustering of the groups representing patients that received dexamethasone (COVID-19+Dex) versus those that did not (COVID-19; Figure 5A and 5B and Table VI in the Data Supplement). This shift in plasma LM profiles was linked with an overall downregulation in plasma proinflammatory eicosanoid concentrations and an upregulation of plasma SPM concentrations (Figure 5C).
Figure 5.
Dexamethasone upregulates plasma specialized proresolving mediator (SPM) in plasma from patients with coronavirus disease 2019 (COVID-19) pneumonia. Peripheral blood was collected from patients with COVID-19 treated with or without dexamethasone (Dex; 6 mg/d). A and B, Plasma lipid mediators were identified and quantified using liquid chromatography tandem mass spectrometry based lipid mediator profiling. Partial least squares discriminant analysis (PLS-DA) was performed on identified mediators, (A) PLS-DA scores Plot with 95% CI and (B) Variable Importance in Projection scores plot. C, Left: cumulative proinflammatory eicosanoids (PG, LT, and TX); right: cumulative SPM (Rv, PD, MaR, LX). Open squares indicate samples identified as statistical outliers using ROUT test (Q=0.2%) and statistical differences were established using Mann-Whitney test (for which outliers were excluded). Patients with n=11 COVID-19 and n=27 COVID-19+Dex. D–K, Blood was collected from patients with COVID-19 or COVID-19+Dex and incubated with lineage-marker antibodies for neutrophils and monocyte subsets in combination with antibodies against (D–G) lipid mediator biosynthetic enzymes or (H–K) SPM receptors and their expression was evaluated using flow cytometry. For (D–G), n=4 patients with COVID-19 and n=8 patients with COVID-19+Dex (except for COX-2 where n=9 patients with COVID-19+Dex). For (H–K), n=4 patients with COVID-19 and n=7 patients with COVID-19+Dex (except for GPR18 and GPR37 where n=6 patients with COVID-19+Dex). Statistical differences between patients with COVID-19 and COVID-19+Dex were established using Mann-Whitney test for each molecule and raw P values are displayed.
Dexamethasone is known to regulate SPM enzyme expression in experimental settings.30 We, therefore, evaluated whether the expression of SPM enzymes in circulating phagocytes was affected by dexamethasone treatment. Flow cytometric evaluation demonstrated a significant upregulation of ALOX15 and ALOX15B in all phagocyte subsets, as well as ALOX12 in classical and intermediate monocytes of patients with COVID-19 treated with dexamethasone (Figure 5D through 5G). These findings suggest that dexamethasone increases peripheral blood SPM concentration in patients with COVID-19 via the upregulation of SPM biosynthetic enzymes in peripheral blood phagocytes.
We next explored whether dexamethasone also regulated the expression of SPM receptors on circulating phagocytes. Flow cytometric analysis demonstrated that while expression of ALX/FPR2 (the receptor for LXA4, RvD1, RvD3, and their aspirin-triggered epimers) was significantly downregulated on all 4 phagocyte subsets, the expression of GPR18 was upregulated on these cells (Figure 5H through 5K). Moreover, GPR37 was downregulated on neutrophils and nonclassical monocytes while it was upregulated on classical and intermediate monocytes (Figure 5H through 5K). Together these findings demonstrate that in addition to upregulating plasma SPM concentrations, dexamethasone also regulates SPM receptor expression on circulating phagocytes from patients with COVID-19.
SPM Rectify Peripheral Blood Phagocyte Responses
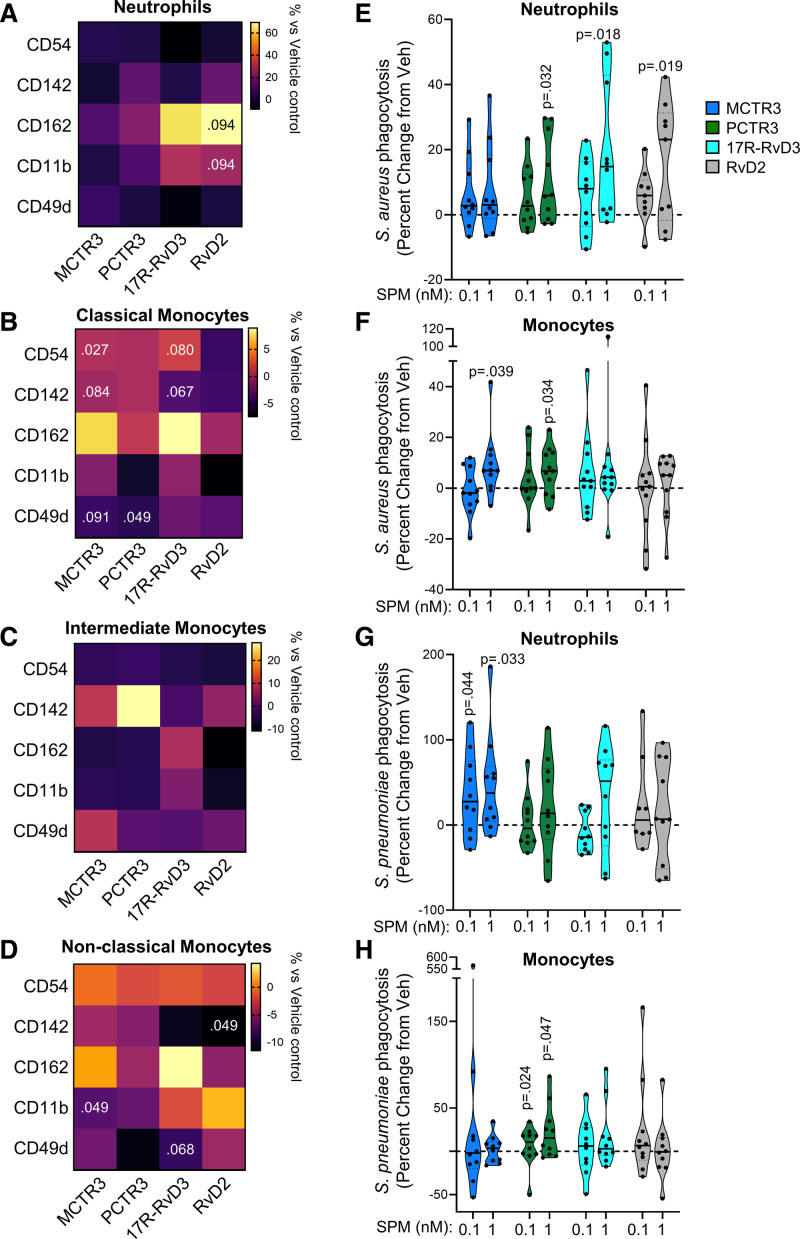
SPM potently regulate phagocyte activation status as well as their ability to uptake and kill bacteria.13,21,24,26,31,32 Having observed that dexamethasone upregulated plasma SPM concentrations, we next evaluated the translational potential of these findings by testing the pharmacological properties of specific SPM in regulating the observed alterations in peripheral blood phagocytes. For this purpose, we focused on MCTR3, PCTR3, 17R-RvD3, and RvD2, given that plasma concentrations of MCTR3 and PCTR3 were found to correlate with phagocyte activation in patients with COVID-19 and their levels were decreased in patients with severe disease. Whereas the receptors for 17R-RvD3 (GPR32) and RvD2 (GPR18) were differentially regulated on phagocytes from these patients. Here, we found that each SPM tested displayed characteristic regulatory activities on the 4 phagocyte subsets (Figure 6). While these mediators did not significantly alter expression of activation markers on neutrophils (Figure 6A), MCTR3 treatment significantly decreased CD54, while PCTR3 downregulated CD49d expression on classical monocytes (Figure 6B). MCTR3 also decreased CD11b expression on nonclassical monocytes (Figure 6D). Recent studies found that expression of the coagulation-initiating CD142 is upregulated on circulating monocytes from patients with COVID-19, an increase that was linked with enhanced disease severity and mortality.33 Incubation of monocytes with all 4 SPM tested led to an overall downregulation of CD142, reaching statistical significance for RvD2 treatment of nonclassical monocytes (Figure 6B through 6D).
Figure 6.
Specialized proresolving mediator (SPM) regulate activation status and bacterial phagocytosis in circulating phagocytes from patients with coronavirus disease 2019 (COVID-19) Neutrophils and monocytes were isolated from peripheral blood of patients with COVID-19 using density centrifugation. A–D, Cells were incubated with 1 nmol/L of indicated SPM or vehicle for 60 min and expression of adhesion molecules was assessed using flow cytometry. Results are reported as percentage change from expression levels in cells incubated with vehicle only. Statistical differences were established using Wilcoxon Signed Rank test for each molecule and raw P values are displayed. n=10 for neutrophils and n=17 for monocytes. E–H, Neutrophils (E and G) and monocytes (F and H) were incubated with indicated SPM or vehicle for 15 min followed by fluorescently labeled (E and F) S. aureus or (G and H) S pneumoniae and phagocytosis assessed in real-time using high-content imaging. Results are expressed as percent change from fluorescence levels recorded in cells incubated with vehicle only. For (E and G), n=10 patients with COVID-19 (except RvD2 0.1 nmol/L where n=8). For (F), n=16 patients with COVID-19. For (H), n=12 patients with COVID-19. Statistical differences between SPM treatments and vehicle were established using Wilcoxon Signed Rank test for each mediator and raw P values are displayed. MCTR indicates maresin conjugates in tissue regeneration; PCTR, protectin conjugates in tissue regeneration; and RvD, D-series resolvin.
Evaluation of the ability of MCTR3, PCTR3, 17R-RvD3, and RvD2 to regulate phagocyte responses based on disease severity (ie, WHO ordinal scale), demonstrated a trend towards upregulation of CD162 on neutrophils from patients with COVID-19 with milder disease. Whereas, these SPM did not regulate neutrophil activation markers on cells from patients with more severe disease (WHO scale 5; Figure IIIA in the Data Supplement). The same outcome emerged when monocytes were analyzed, with SPM being more effective on classical and intermediate monocytes of WHO scale 3 to 4 patients (Supplemental Figure IIIB through 3D in the Data Supplement). Nonclassical monocytes were the only phagocyte subset tested where the activity of SPM on the regulation of activation marker expression appeared to be higher in cells from patients with severe disease (Figure IIID in the Data Supplement).
We next evaluated whether SPM rescued the defect in S. aureus phagocytosis observed in peripheral blood leukocytes isolated from patients with COVID-19. Incubation of all 4 SPM with peripheral blood neutrophils led to an increase in phagocytosis by neutrophils, which reached statistical significance for PCTR3, 17R-RvD3, and RvD2 at 1 nmol/L (Figure 6E). We also observed increases in S. aureus phagocytosis by peripheral blood monocytes when these cells were incubated with 1 nmol/L of either MCTR3 or PCTR3 (Figure 6F). In addition, these mediators significantly increased phagocytosis of Streptococcus pneumoniae, another clinically relevant bacterium in COVID-19,34 by peripheral blood neutrophils and monocytes from patients with COVID-19, respectively (Figure 6G and 6H).
SPM Reprogram Monocyte-Derived Macrophages From Patients With COVID-19
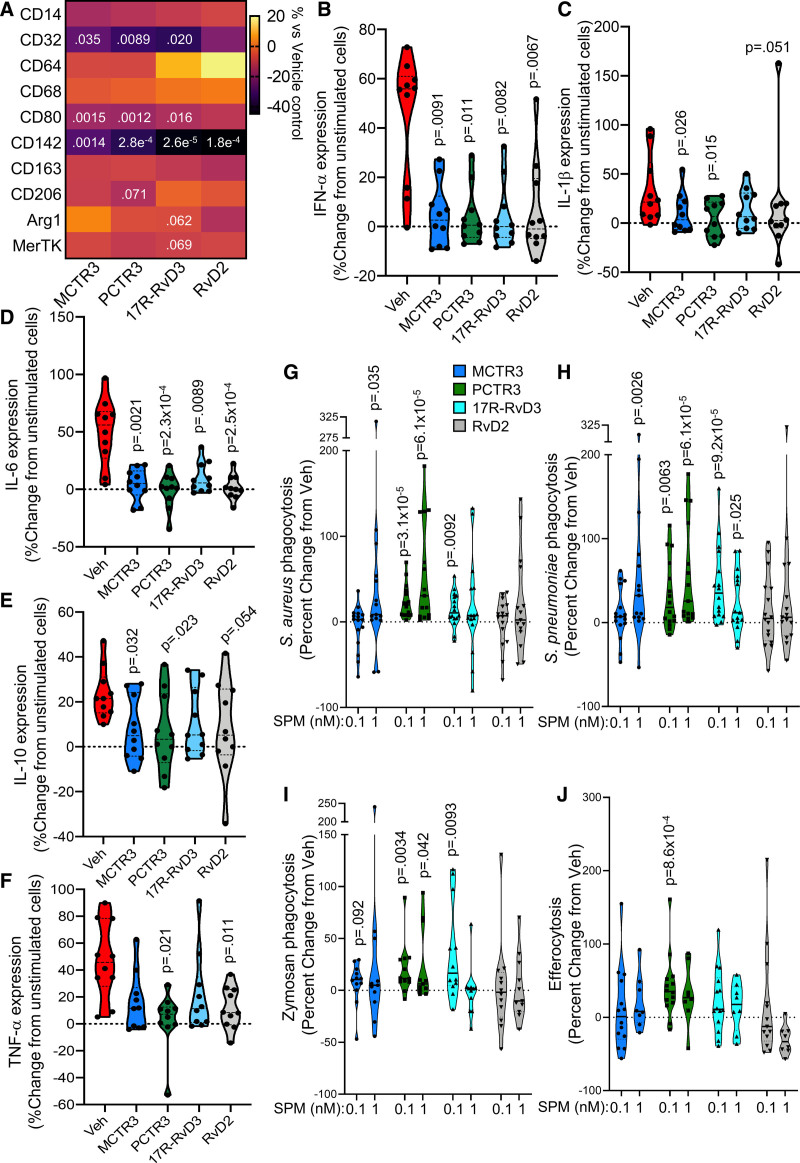
Monocyte-derived macrophages are linked with onset, progression, and resolution of both acute and chronic inflammation.4,13,35 Furthermore, macrophage responses in patients with COVID-19 are dysregulated, with these cells displaying an inflammatory phenotype that is linked with increased tissue inflammation and damage.12 Having found that SPM regulate both monocyte activation and function, we next assessed whether these protective activities extended to monocyte-derived macrophages. For this purpose, we incubated monocytes isolated from peripheral blood of patients with COVID-19 with SPM, differentiated them to macrophages using published protocols,23 and assessed the expression of macrophage phenotypic markers using flow cytometry. Here, we found that MCTR3, PCTR3, and 17R-RvD3 significantly downregulated CD32 and CD80 expression, whereas PCTR3 downregulated CD206 expression and 17R-RvD3 downregulated Arg (arginase)-1 and MerTK expression. Notably, all 4 SPM tested significantly downregulated the expression of CD142 (Figure 7A). Furthermore, the activity of these SPM was found to be comparable in patients with mild and severe disease (Figure IV in the Data Supplement).
Figure 7.
Regulation of coronavirus disease 2019 (COVID-19) patient monocyte-derived macrophage phenotype and function by specialized proresolving mediator (SPM). A–F, Monocytes were isolated from peripheral blood of patients with COVID-19 and differentiated to monocyte-derived macrophages with GM-CSF in the presence of vehicle or 10 nmol/L of the indicated SPM. On day 7, (A) cells were lifted and the expression of the indicated phenotypic markers was assessed using flow cytometry (n=13 patients with COVID-19) or (B–F) cells were incubated with recombinant human S100A8/A9 dimer (1 µg/mL, 24 h) and Brefeldin A (2 µg/mL, for final 18 h) and the expression of indicated cytokines was assessed using flow cytometry (n=12 patients with COVID-19). Results are reported as percentage change in expression from levels in cells incubated with vehicle only. Statistical differences between vehicle and SPM treatments were established using Kruskal-Wallis test with Dunn post hoc correction. G–I, Monocytes from patients with COVID-19 were differentiated to monocyte-derived macrophages with GM-CSF, incubated with the indicated SPM (0.1 or 1 nmol/L) or vehicle for 15 min followed by fluorescently labeled (G) S. aureus, (H) S. pneumoniae, (I) zymosan, or (J) apoptotic cells. Results are reported as percentage change in uptake from levels in cells incubated with vehicle only. For (G–H), n=16 (for 0.1 nmol/L SPM) and n=15 (for 1 nmol/L SPM) patients with COVID-19. For (I), n=12 (for 0.1 nmol/L SPM) and n=11 (for 1 nmol/L SPM) patients with COVID-19. For (J), n=14 (for 0.1 nmol/L SPM) and n=8 (for 1 nmol/L SPM) patients with COVID-19. Statistical differences between SPM treatments and vehicle were established using Wilcoxon Signed Rank test for each mediator and raw P values are displayed. IL indicates interleukin; MCTR indicates maresin conjugates in tissue regeneration; PCTR, protectin conjugates in tissue regeneration; RvD, D-series resolvin; and TNF, tumor necrosis factor.
The excessive production of proinflammatory cytokines, such as IL (interleukin)-1β, IL-6, and TNF (tumor necrosis factor)-α, is one of the hallmarks of severe COVID-19.9 Macrophage-derived cytokines play a central role in the propagation of tissue inflammation. Thus, we next evaluated whether SPM regulated cytokine production in monocyte-derived macrophages. Incubation of these cells with S100A8/A9 dimer, a potent proinflammatory signaling molecule that is abundant in inflamed tissues from patients with COVID-19,8,36,37 upregulated the expression of IFN (interferon)-α, IL-1β, IL-6, IL-10, and TNF-α (Figure 7B through 7F). Incubating macrophages with MCTR3, PCTR3, 17R-RvD3, or RvD2 led to a significant decrease in the expression of S100A8/A9-induced IFN-α, IL-1β, and IL-6 expression, while MCTR3, PCTR3, and RvD2 also significantly decreased expression of IL-10 and TNF-α (Figure 7B through 7F). Of note, assessment of cytokine expression when separating patients with COVID-19 by disease severity based on WHO ordinal scale demonstrated that all 4 SPM were equally effective at reducing expression of these cytokines in cells from both patient groups (Figure V in the Data Supplement).
We next assessed whether SPM regulated phagocytic responses in these cells. Incubation of monocyte-derived macrophages with MCTR3, PCTR3, or 17R-RvD3 increased their ability to clear the Gram-positive bacteria S. aureus and S. pneumoniae in a concentration-dependent manner (Figure 7G and 7H). Given that secondary infections by fungi from the Aspergillus genus are also common in patients with COVID-19,38 we next tested whether these SPM also regulated the clearance of fungi. Here we found that MCTR3, PCTR3, and 17R-RvD3 dose-dependently increased uptake of fluorescently labeled zymosan, a fungal cell wall component (Figure 7I). Notably, while PCTR3 and 17R-RvD3 increased phagocytosis of S. aureus, S. pneumoniae, and zymosan particles to a similar extent in cells from patients with mild disease when compared with those with severe disease, MCTR3 displayed greater activity with cells from patients with mild disease (Figure VI in the Data Supplement).
Since the extensive and unresolved inflammation observed in the lungs of patients with COVID-19 gives rise to widespread tissue damage and apoptosis, we next assessed the ability of SPM to enhance the clearance of apoptotic cells (a process termed efferocytosis). Here, we found that PCTR3 increased efferocytosis of apoptotic cells by monocyte-derived macrophages from patients with COVID-19 in a concentration-dependent manner (Figure 7J). Together these findings demonstrate that SPM alter the function of monocyte-derived macrophages from patients with COVID-19 towards a host protective phenotype.
Discussion
Our study presents evidence that selective determinants of proresolving pathways are present in the blood of patients with COVID-19 pneumonia and suggests that their dysregulation, or defective engagement, may contribute to disease propagation. In peripheral blood from these patients, we observed an upregulation of SPM concentrations and expression of proresolving receptors on circulating phagocytes when compared with healthy volunteers, which became dysregulated with increasing disease severity. These changes were linked with altered neutrophil and monocyte activation status and function when compared with phagocytes from healthy volunteers. Treatment of patients with COVID-19 with dexamethasone upregulated both peripheral blood SPM concentrations and the expression of their biosynthetic enzymes, while incubation of peripheral blood phagocytes with SPM modulated both their activation status and ability to uptake bacteria. In addition, SPM potently tempered monocyte-derived macrophage responses, downregulating the expression of proinflammatory cytokines and upregulating their ability to clear pathogenic microbes and apoptotic cells.
Phagocytes form the first line of defence against invading pathogens.39–41 The behavior of these cells is regulated by environmental cues that include soluble mediators such as cytokines and LM. Mounting evidence demonstrates that phagocyte behavior becomes dysregulated in patients with COVID-19 pneumonia with severe disease, contributing to uncontrolled systemic inflammation with ensuing organ damage.7,33,42–44 Furthermore, these disrupted phagocyte responses are linked with an increased susceptibility to secondary infections.27,28 In the present study, we observed that overall LM concentrations are upregulated in plasma from patients with COVID-19 pneumonia. This concomitant increase in both proinflammatory and proresolving mediators is a feature of several inflammatory conditions, including sepsis and acute respiratory distress syndrome,18 and reflects the activation of counter-regulatory mechanisms by the host as an attempt to control unabated inflammation. Of note, Schwarz et al45 reported that an increase in peripheral blood LM production is retained in serum from patients with COVID-19 pneumonia. The upregulation of SPM in both plasma and serum, together with an increase in the expression of SPM enzymes in peripheral blood monocyte subsets supports the hypothesis that activation of these cells is at least in part responsible for the observed increases in the concentrations of these molecules. Notably, plasma concentrations of SPM were decreased in those patients with more severe disease suggesting that inability of the host to upregulate these molecules leads to disease propagation. This hypothesis is further supported by the observation that in patients treated with dexamethasone, which has been shown to decrease disease severity and mortality in patients with severe COVID-19,6 we observed increased plasma SPM concentrations as well as SPM biosynthetic enzyme expression in circulating phagocytes.
Recent studies demonstrate that the increased platelet-leukocytes heterotypic aggregates found in patients with COVID-19 are linked with an upregulation of CD142 on monocytes.33 CD142 initiates the extrinsic coagulation cascade, is fully functional when expressed on cell surfaces, and increased CD142 expression is linked with a state of hypercoagulability and elevated disease severity in patients with COVID-19 pneumonia.33 Studies investigating the expression of CD142 indicate that the expression of this protein is regulated by both proinflammatory cytokines, such as IL-6,10 and proinflammatory mediators such as thromboxane A2.46,47 In our studies, we found that SPM downregulated the expression of this protein on all 3 monocyte subsets as well as on monocyte-derived macrophages. The regulation of this molecule by SPM was also observed to be comparable between cells from hospitalized patients with mild and severe disease. These results suggest that in addition to regulating leukocyte recruitment and function, SPM may also contribute to reducing the hypercoagulable state observed in patients with COVID-19 by downregulating the expression of CD142 on circulating and tissue-resident phagocytes.
During inflammation, monocytes are recruited into inflamed tissues where they differentiate to monocyte-derived macrophages that either contribute to the propagation of tissue inflammation or promote its resolution and tissue repair to facilitate the re-establishment of tissue function. Studies in patients with COVID-19 as well as in nonhuman primates demonstrate a marked increase in monocyte-derived macrophages numbers in various tissues, including lung, intestine, kidney, and liver.7,42,43 These cells can contribute to the disseminated inflammatory status observed in patients with COVID-19 via the production of both inflammatory cytokines such as IL-6, TNF-α, and IFNs, as well as immunosuppressive agents such as IL-10.11,43 In the present study, we found that SPM potently downregulated the production of several inflammatory cytokines by monocyte-derived macrophages from patients with COVID-19. Intriguingly, type I interferons and PGE2 downregulate bacterial phagocytosis by alveolar macrophages,48,49 suggesting that the decreased bacterial phagocytosis by patient with COVID-19–derived phagocytes might be due to elevated levels of these proinflammatory molecules counteracting endogenous SPM signaling. Consistent with this hypothesis, incubation of patient-derived monocyte-derived macrophages with SPM decreased IFN-α expression and increased their ability to uptake bacteria. Reduced inflammatory cytokine expression was also linked with a downregulation in the expression of phenotypic markers, including CD80 and CD206, which were recently found to be upregulated on monocytes from patients with COVID-19 who express higher levels of proinflammatory cytokines.44
At epithelial surfaces, monocyte-derived macrophages play a central role in the clearance of pathogens. Several studies found that patients with COVID-19 may also develop secondary bacterial or fungal infections.27,28 Here, we found that MCTR3, PCTR3, and 17R-RvD3 significantly upregulated the uptake and killing of both bacteria and fungi by macrophages, an activity that was comparable between cells from patients with mild and severe disease. Taken together, the present findings highlight the potential pharmacological utility of SPM in regulating COVID-19-related immune cell responses. Such SPM-based therapeutics could either involve the use of SPM analogues/mimetics or the enhancement of endogenous SPM production through the administration of SPM substrates and precursors. Of note, we recently found that this latter approach enhances circulating SPM levels in healthy individuals and patients with peripheral artery disease, while it tempered phagocyte phenotype and function.50,51
The strengths of the present study are that it evaluates the correlation between changes in plasma SPM and changes in phagocyte functions in patients with COVID-19. Furthermore, it interrogates the potential pharmacological utility of SPM in rectifying altered phagocyte functions. There are also some limitations that should be considered when evaluating the present findings. First, we were unable to recruit individuals suffering from acute COVID-19 who did not require hospitalization (ie, WHO scale 1–2). Nor were we able to obtain serial samples from the same patient over a period of time. Such patient groups and samples could provide further insight into when and how changes in LM profiles occur during COVID-19 disease progression. Additionally, our study was not sufficiently powered to explore potential interactions between COVID-19 and specific comorbidities known to affect LM concentrations in plasma. Notably, comparisons of LM concentrations in patients with COVID-19 separated by disease severity (Figure 3) or dexamethasone treatment status (Figure 5) showed that there were no marked differences in the comorbidities between these groups (Tables III and V in the Data Supplement). Finally, future studies will be needed to establish the mechanisms that govern the observed changes in SPM biosynthesis. Here, we measured protein expression of key SPM biosynthetic enzymes as one aspect that defines their activity and therefore SPM production. For some of our comparisons, the changes in enzyme expression were consistent with changes observed in LM profiles (Figures 1, 3, and 5). Intriguingly, we found that such a relationship was not observed between plasma SPM concentrations and enzyme expression in leukocytes from post-COVID-19 volunteers. This observation suggests that additional mechanisms, for example, subcellular localization or posttranslational modifications of the enzymes,52 may contribute to the LM profiles obtained in plasma from post-COVID-19 volunteers (Figure II in the Data Supplement).
In summation, in the present study, we provide evidence that the host immune response engages resolution mechanisms as an attempt to limit the over-shooting inflammatory response following SARS-CoV-2 infection. However, these protective mechanisms appear to fail in patients with severe COVID-19 pneumonia, leading to systemic inflammation and dysregulated circulating phagocyte responses, which in turn contribute to secondary organ damage. Such inadequate resolution is linked with the downregulation of SPM receptors on leukocytes. Nonetheless, these receptors remain functional and incubation of phagocytes from patients with COVID-19 with MCTR3, PCTR3, 17R-RvD3, or RvD2 rectified many of the phagocyte responses investigated. Together, these findings shed new light on the mechanisms contributing to the disease propagation in COVID-19 pneumonia.
Acknowledgments
We thank the patients and volunteers who donated blood, Dr Francesco Palmas and members of the QMUL Lipid Mediator Unit for technical support.
Author Contributions
J. Dalli conceived the overall research plan and supervised the work; J. Dalli, D.S. Koenis, and P.R. Souza designed experiments; D.S. Koenis, I. Beegun, P.R. Souza, C.C. Jouvene, M. Gonzalez-Nunez, L. Ly, K. Pistorius, and G. Alusi conducted the experiments and analyzed results; P. Pfeffer, W. Ricketts, G. Thomas, and I. Beegun consented patients and volunteers and collected blood. H.M. Kocher and M. Perretti provided resources and critical insights. All authors contributed to article preparation.
Sources of Funding
This work was supported by funding from the European Research Council (ERC) under the European Union’s Horizon 2020 research and innovation program (grant No. 677542) and the Barts Charity (grant No. MGU0343) to J. Dalli. J. Dalli is also supported by a Sir Henry Dale Fellowship jointly funded by the Wellcome Trust and the Royal Society (grant No. 107613/Z/15/Z).
Disclosures
J. Dalli and M. Perretti are scientific founders and directors of Resolomics Limited. The other authors report no conflicts.
Supplemental Materials
Expanded Materials and Methods
Data Supplement Tables I–IX
Data Supplement Figures I–VI
Supplementary Material
Nonstandard Abbreviations and Acronyms
- ALOX5
- 5-lipoxygenase
- CD
- cluster of differentiation
- COVID-19
- coronavirus disease 2019
- DHA
- docosahexaenoic acid
- DPA
- docosapentaenoic acid
- GPCR
- G protein-coupled receptor
- IFN
- interferon
- IL
- interleukin
- LM
- lipid mediator
- MCTR
- maresin conjugates in tissue regeneration
- PCTR
- protectin conjugates in tissue regeneration
- PLS-DA
- partial least squares discriminant analysis
- RvD
- D-series resolvin
- RvE
- E-series resolvin
- RvT
- 13-series resolvin
- SARS-CoV-2
- severe acute respiratory syndrome coronavirus 2
- SPM
- specialized proresolving mediators
- TNF
- tumor necrosis factor
I. Beegun and C.C. Jouvene contributed equally.
P. Pfeffer and J. Dalli are joint senior authors.
The Data Supplement is available with this article at https://www.ahajournals.org/doi/suppl/10.1161/CIRCRESAHA.121.319142.
For Sources of Funding and Disclosures, see page e70.
Contributor Information
Duco Steven Koenis, Email: d.koenis@qmul.ac.uk.
Issa Beegun, Email: i.beegun@qmul.ac.uk.
Charlotte Camille Jouvene, Email: c.jouvene@qmul.ac.uk.
Gabriel Amador Aguirre, Email: g.amador@qmul.ac.uk.
Patricia Regina Souza, Email: p.soaresdesouza@qmul.ac.uk.
Maria Gonzalez-Nunez, Email: m.gonzaleznunez@qmul.ac.uk.
Lucy Ly, Email: l.ly@qmul.ac.uk.
Kimberly Pistorius, Email: k.c.d.f.pistorius@qmul.ac.uk.
Hemant M. Kocher, Email: h.kocher@qmul.ac.uk.
William Ricketts, Email: william.ricketts@nhs.net.
Gavin Thomas, Email: gavin.thomas5@nhs.net.
Mauro Perretti, Email: m.perretti@qmul.ac.uk.
Ghassan Alusi, Email: alusig@gmail.com.
Paul Pfeffer, Email: p.pfeffer@qmul.ac.uk.
Novelty and Significance
What Is Known?
Specialized proresolving mediators (SPM) are produced via enzymatic conversion of essential fatty acids and promote inflammatory resolution and tissue homeostasis.
Failure to resolve inflammation in a timely manner leads to chronic inflammation.
Severe cases of coronavirus disease 2019 (COVID-19) are characterized by a hyper-inflammatory state that is largely driven by innate immune cell dysfunction.
What New Information Does This Article Contribute?
Circulating SPM concentrations were decreased in patients with severe disease and correlated with the activation status and function of circulating phagocytes.
Perturbations in plasma SPM concentrations and phagocyte activation were retained after the resolution of mild disease, while treatment of patients with severe COVID-19 with dexamethasone increased levels of circulating SPM as well as the expression of their biosynthetic enzymes.
Incubating phagocytes from patients with COVID-19 with specific SPM rectified disease-associated perturbations in their phenotype and function.
A subset of people infected with severe acute respiratory syndrome coronavirus 2 develop severe COVID-19 requiring hospitalization. These severe disease cases are characterized by a disseminated inflammatory response that can lead to cardiovascular complications and multiorgan failure. A better understanding of the mechanisms underpinning this dysregulated inflammatory response may provide novel therapeutic leads for the improved treatment of hospitalized patients. Given the known immunoregulatory actions of SPM, we evaluated whether SPM pathways are altered in patients with COVID-19. The present study suggests that resolution mechanisms are engaged by the host in an attempt to resolve inflammation in the wake of severe acute respiratory syndrome coronavirus 2 infection. However, these protective mechanisms appear to fail in patients with severe COVID-19, leading to systemic inflammation and dysregulated circulating phagocyte responses. Inadequate resolution was found to be linked with downregulation of SPM biosynthetic enzymes and receptors on circulating leukocytes. Nonetheless, these receptors remain functional as incubation of phagocytes from patients with COVID-19 with select SPM rectified many of the pathological phagocyte responses. Taken together, these findings shed new light on the mechanisms contributing to the disease propagation in COVID-19. Therefore, our study provides a basis for future research on the feasibility and potency of targeting SPM-driven resolution mechanisms for the treatment of severe COVID-19.
References
- 1.Gilroy DW, De Maeyer RPH, Tepper M, O’Brien A, Uddin M, Chen J, Goldstein DR, Akbar AN. Treating exuberant, non-resolving inflammation in the lung; Implications for acute respiratory distress syndrome and COVID-19. Pharmacol Ther. 2021;221:107745. doi: 10.1016/j.pharmthera.2020.107745 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Luissint AC, Parkos CA, Nusrat A. Inflammation and the intestinal barrier: leukocyte-epithelial cell interactions, cell junction remodeling, and mucosal repair. Gastroenterology. 2016;151:616–632. doi: 10.1053/j.gastro.2016.07.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Robb CT, Regan KH, Dorward DA, Rossi AG. Key mechanisms governing resolution of lung inflammation. Semin Immunopathol. 2016;38:425–448. doi: 10.1007/s00281-016-0560-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Serhan CN, Levy BD. Resolvins in inflammation: emergence of the pro-resolving superfamily of mediators. J Clin Invest. 2018;128:2657–2669. doi: 10.1172/JCI97943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Poland GA, Ovsyannikova IG, Kennedy RB. SARS-CoV-2 immunity: review and applications to phase 3 vaccine candidates. Lancet. 2020;396:1595–1606. doi: 10.1016/S0140-6736(20)32137-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Horby P, Lim WS, Emberson JR, Mafham M, Bell JL, Linsell L, Staplin N, Brightling C, Ustianowski A, Elmahi E, et al. ; RECOVERY Collaborative Group Dexamethasone in Hospitalized Patients with Covid-19 - Preliminary Report. N Engl J Med. 2020;384:693–704. doi: 10.1056/NEJMoa2021436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fahlberg MD, Blair RV, Doyle-Meyers LA, Midkiff CC, Zenere G, Russell-Lodrigue KE, Monjure CJ, Haupt EH, Penney TP, Lehmicke G, et al. Cellular events of acute, resolving or progressive COVID-19 in SARS-CoV-2 infected non-human primates. Nat Commun. 2020;11:6078. doi: 10.1038/s41467-020-19967-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Guo Q, Zhao Y, Li J, Liu J, Yang X, Guo X, Kuang M, Xia H, Zhang Z, Cao L, et al. Induction of alarmin S100A8/A9 mediates activation of aberrant neutrophils in the pathogenesis of COVID-19. Cell Host Microbe. 2020;29:222, e4–235. doi: 10.1016/j.chom.2020.12.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim JS, Lee JY, Yang JW, Lee KH, Effenberger M, Szpirt W, Kronbichler A, Shin JI. Immunopathogenesis and treatment of cytokine storm in COVID-19. Theranostics. 2021;11:316–329. doi: 10.7150/thno.49713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Loo J, Spittle DA, Newnham M. COVID-19, immunothrombosis and venous thromboembolism: biological mechanisms. Thorax. 2021;76:412–420. doi: 10.1136/thoraxjnl-2020-216243 [DOI] [PubMed] [Google Scholar]
- 11.Lu L, Zhang H, Dauphars DJ, He YW. A potential role of interleukin 10 in COVID-19 pathogenesis. Trends Immunol. 2021;42:3–5. doi: 10.1016/j.it.2020.10.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Merad M, Martin JC. Pathological inflammation in patients with COVID-19: a key role for monocytes and macrophages. Nat Rev Immunol. 2020;20:355–362. doi: 10.1038/s41577-020-0331-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Dalli J. Does promoting resolution instead of inhibiting inflammation represent the new paradigm in treating infections? Mol Aspects Med. 2017;58:12–20. doi: 10.1016/j.mam.2017.03.007 [DOI] [PubMed] [Google Scholar]
- 14.Ramon S, Baker SF, Sahler JM, Kim N, Feldsott EA, Serhan CN, Martínez-Sobrido L, Topham DJ, Phipps RP. The specialized proresolving mediator 17-HDHA enhances the antibody-mediated immune response against influenza virus: a new class of adjuvant? J Immunol. 2014;193:6031–6040. doi: 10.4049/jimmunol.1302795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Morita M, Kuba K, Ichikawa A, Nakayama M, Katahira J, Iwamoto R, Watanebe T, Sakabe S, Daidoji T, Nakamura S, et al. The lipid mediator protectin D1 inhibits influenza virus replication and improves severe influenza. Cell. 2013;153:112–125. doi: 10.1016/j.cell.2013.02.027 [DOI] [PubMed] [Google Scholar]
- 16.Rajasagi NK, Reddy PB, Suryawanshi A, Mulik S, Gjorstrup P, Rouse BT. Controlling herpes simplex virus-induced ocular inflammatory lesions with the lipid-derived mediator resolvin E1. J Immunol. 2011;186:1735–1746. doi: 10.4049/jimmunol.1003456 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gomez EA, Colas RA, Souza PR, Hands R, Lewis MJ, Bessant C, Pitzalis C, Dalli J. Blood pro-resolving mediators are linked with synovial pathology and are predictive of DMARD responsiveness in rheumatoid arthritis. Nat Commun. 2020;11:5420. doi: 10.1038/s41467-020-19176-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dalli J, Colas RA, Quintana C, Barragan-Bradford D, Hurwitz S, Levy BD, Choi AM, Serhan CN, Baron RM. Human sepsis eicosanoid and proresolving lipid mediator temporal profiles: correlations with survival and clinical outcomes. Crit Care Med. 2017;45:58–68. doi: 10.1097/CCM.0000000000002014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Colas RA, Souza PR, Walker ME, Burton M, Zasłona Z, Curtis AM, Marques RM, Dalli J. Impaired production and diurnal regulation of vascular RvDn-3 DPA increase systemic inflammation and cardiovascular disease. Circ Res. 2018;122:855–863. doi: 10.1161/CIRCRESAHA.117.312472 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Norris PC, Libreros S, Serhan CN. Resolution metabolomes activated by hypoxic environment. Sci Adv. 2019;5:eaax4895. doi: 10.1126/sciadv.aax4895 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chiang N, Serhan CN. Structural elucidation and physiologic functions of specialized pro-resolving mediators and their receptors. Mol Aspects Med. 2017;58:114–129. doi: 10.1016/j.mam.2017.03.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Perretti M, Cooper D, Dalli J, Norling LV. Immune resolution mechanisms in inflammatory arthritis. Nat Rev Rheumatol. 2017;13:87–99. doi: 10.1038/nrrheum.2016.193 [DOI] [PubMed] [Google Scholar]
- 23.Flak MB, Koenis DS, Sobrino A, Smith J, Pistorius K, Palmas F, Dalli J. GPR101 mediates the pro-resolving actions of RvD5n-3 DPA in arthritis and infections. J Clin Invest. 2020;130:359–373. doi: 10.1172/JCI131609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chiang N, Dalli J, Colas RA, Serhan CN. Identification of resolvin D2 receptor mediating resolution of infections and organ protection. J Exp Med. 2015;212:1203–1217. doi: 10.1084/jem.20150225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bang S, Xie YK, Zhang ZJ, Wang Z, Xu ZZ, Ji RR. GPR37 regulates macrophage phagocytosis and resolution of inflammatory pain. J Clin Invest. 2018;128:3568–3582. doi: 10.1172/JCI99888 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chiang N, Libreros S, Norris PC, de la Rosa X, Serhan CN. Maresin 1 activates LGR6 receptor promoting phagocyte immunoresolvent functions. J Clin Invest. 2019;129:5294–5311. doi: 10.1172/JCI129448 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ripa M, Galli L, Poli A, Oltolini C, Spagnuolo V, Mastrangelo A, Muccini C, Monti G, De Luca G, Landoni G, et al. ; COVID-BioB study group Secondary infections in patients hospitalized with COVID-19: incidence and predictive factors. Clin Microbiol Infect. 2021;27:451–457. doi: 10.1016/j.cmi.2020.10.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Manohar P, Loh B, Nachimuthu R, Hua X, Welburn SC, Leptihn S. Secondary bacterial infections in patients with viral pneumonia. Front Med (Lausanne). 2020;7:420. doi: 10.3389/fmed.2020.00420 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pyrillou K, Chairakaki AD, Tamvakopoulos C, Andreakos E. Dexamethasone induces ω3-derived immunoresolvents driving resolution of allergic airway inflammation. J Allergy Clin Immunol. 2018;142:691–695.e4. doi: 10.1016/j.jaci.2018.04.004 [DOI] [PubMed] [Google Scholar]
- 30.Colamorea T, Di Paola R, Macchia F, Guerrese MC, Tursi A, Butterfield JH, Caiaffa MF, Haeggström JZ, Macchia L. 5-Lipoxygenase upregulation by dexamethasone in human mast cells. Biochem Biophys Res Commun. 1999;265:617–624. doi: 10.1006/bbrc.1999.1732 [DOI] [PubMed] [Google Scholar]
- 31.Chen J, Shetty S, Zhang P, Gao R, Hu Y, Wang S, Li Z, Fu J. Aspirin-triggered resolvin D1 down-regulates inflammatory responses and protects against endotoxin-induced acute kidney injury. Toxicol Appl Pharmacol. 2014;277:118–123. doi: 10.1016/j.taap.2014.03.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lee S, Nakahira K, Dalli J, Siempos II, Norris PC, Colas RA, Moon JS, Shinohara M, Hisata S, Howrylak JA, et al. NLRP3 inflammasome deficiency protects against microbial sepsis via increased lipoxin B4 synthesis. Am J Respir Crit Care Med. 2017;196:713–726. doi: 10.1164/rccm.201604-0892OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hottz ED, Azevedo-Quintanilha IG, Palhinha L, Teixeira L, Barreto EA, Pão CRR, Righy C, Franco S, Souza TML, Kurtz P, et al. Platelet activation and platelet-monocyte aggregate formation trigger tissue factor expression in patients with severe COVID-19. Blood. 2020;136:1330–1341. doi: 10.1182/blood.2020007252 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ginsburg AS, Klugman KP. COVID-19 pneumonia and the appropriate use of antibiotics. Lancet Glob Health. 2020;8:e1453–e1454. doi: 10.1016/S2214-109X(20)30444-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Motwani MP, Gilroy DW. Macrophage development and polarization in chronic inflammation. Semin Immunol. 2015;27:257–266. doi: 10.1016/j.smim.2015.07.002 [DOI] [PubMed] [Google Scholar]
- 36.Chen L, Long X, Xu Q, Tan J, Wang G, Cao Y, Wei J, Luo H, Zhu H, Huang L, et al. Elevated serum levels of S100A8/A9 and HMGB1 at hospital admission are correlated with inferior clinical outcomes in COVID-19 patients. Cell Mol Immunol. 2020;17:992–994. doi: 10.1038/s41423-020-0492-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shaath H, Vishnubalaji R, Elkord E, Alajez NM. Single-cell transcriptome analysis highlights a role for neutrophils and inflammatory macrophages in the pathogenesis of severe COVID-19. Cells. 2020;9:E2374. doi: 10.3390/cells9112374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Alanio A, Dellière S, Fodil S, Bretagne S, Mégarbane B. Prevalence of putative invasive pulmonary aspergillosis in critically ill patients with COVID-19. Lancet Respir Med. 2020;8:e48–e49. doi: 10.1016/S2213-2600(20)30237-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Campbell EL, Colgan SP. Neutrophils and inflammatory metabolism in antimicrobial functions of the mucosa. J Leukoc Biol. 2015;98:517–522. doi: 10.1189/jlb.3MR1114-556R [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Parkos CA. Neutrophil-epithelial interactions: a double-edged sword. Am J Pathol. 2016;186:1404–1416. doi: 10.1016/j.ajpath.2016.02.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Shalova IN, Lim JY, Chittezhath M, Zinkernagel AS, Beasley F, Hernández-Jiménez E, Toledano V, Cubillos-Zapata C, Rapisarda A, Chen J, et al. Human monocytes undergo functional re-programming during sepsis mediated by hypoxia-inducible factor-1α. Immunity. 2015;42:484–498. doi: 10.1016/j.immuni.2015.02.001 [DOI] [PubMed] [Google Scholar]
- 42.Speranza E, Williamson BN, Feldmann F, Sturdevant GL, Pérez-Pérez L, Meade-White K, Smith BJ, Lovaglio J, Martens C, Munster VJ, et al. Single-cell RNA sequencing reveals SARS-CoV-2 infection dynamics in lungs of African green monkeys. Sci Transl Med. 2021;13:eabe8146. doi: 10.1126/scitranslmed.abe8146 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wang C, Xu J, Wang S, Pan S, Zhang J, Han Y, Huang M, Wu D, Yang Q, Yang X, et al. Imaging mass cytometric analysis of postmortem tissues reveals dysregulated immune cell and cytokine responses in multiple organs of COVID-19 patients. Front Microbiol. 2020;11:600989. doi: 10.3389/fmicb.2020.600989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zhang D, Guo R, Lei L, Liu H, Wang Y, Wang Y, Qian H, Dai T, Zhang T, Lai Y, et al. COVID-19 infection induces readily detectable morphologic and inflammation-related phenotypic changes in peripheral blood monocytes. J Leukoc Biol. 2020;109:13–22. doi: 10.1002/JLB.4HI0720-470R [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Schwarz B, Sharma L, Roberts L, Peng X, Bermejo S, Leighton I, Casanovas-Massana A, Minasyan M, Farhadian S, Ko AI, et al. ; Yale IMPACT Team Cutting edge: severe SARS-CoV-2 infection in humans is defined by a shift in the serum lipidome, resulting in dysregulation of eicosanoid immune mediators. J Immunol. 2021;206:329–334. doi: 10.4049/jimmunol.2001025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bode M, Mackman N. Regulation of tissue factor gene expression in monocytes and endothelial cells: thromboxane A2 as a new player. Vascul Pharmacol. 2014;62:57–62. doi: 10.1016/j.vph.2014.05.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Eilertsen KE, Østerud B. The central role of thromboxane and platelet activating factor receptors in ex vivo regulation of endotoxin-induced monocyte tissue factor activity in human whole blood. J Endotoxin Res. 2002;8:285–293. doi: 10.1179/096805102125000498 [DOI] [PubMed] [Google Scholar]
- 48.Aronoff DM, Canetti C, Peters-Golden M. Prostaglandin E2 inhibits alveolar macrophage phagocytosis through an E-prostanoid 2 receptor-mediated increase in intracellular cyclic AMP. J Immunol. 2004;173:559–565. doi: 10.4049/jimmunol.173.1.559 [DOI] [PubMed] [Google Scholar]
- 49.Nakamura S, Davis KM, Weiser JN. Synergistic stimulation of type I interferons during influenza virus coinfection promotes Streptococcus pneumoniae colonization in mice. J Clin Invest. 2011;121:3657–3665. doi: 10.1172/JCI57762 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Schaller MS, Chen M, Colas RA, Sorrentino TA, Lazar AA, Grenon SM, Dalli J, Conte MS. Treatment with a marine oil supplement alters lipid mediators and leukocyte phenotype in healthy patients and those with peripheral artery disease. J Am Heart Assoc. 2020;9:e016113. doi: 10.1161/JAHA.120.016113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Souza PR, Marques RM, Gomez EA, Colas RA, De Matteis R, Zak A, Patel M, Collier DJ, Dalli J. Enriched marine oil supplements increase peripheral blood specialized pro-resolving mediators concentrations and reprogram host immune responses: a randomized double-blind placebo-controlled study. Circ Res. 2020;126:75–90. doi: 10.1161/CIRCRESAHA.119.315506 [DOI] [PubMed] [Google Scholar]
- 52.Fredman G, Ozcan L, Spolitu S, Hellmann J, Spite M, Backs J, Tabas I. Resolvin D1 limits 5-lipoxygenase nuclear localization and leukotriene B4 synthesis by inhibiting a calcium-activated kinase pathway. Proc Natl Acad Sci U S A. 2014;111:14530–14535. doi: 10.1073/pnas.1410851111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Gomez EA, Colas RA, Souza PR, Hands R, Lewis MJ, Bessant C, Pitzalis C, Dalli J. Blood pro-resolving mediators are linked with synovial pathology and are predictive of DMARD responsiveness in rheumatoid arthritis. Nat Commun. 2020;11:5420. doi: 10.1038/s41467-020-19176-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Xia J, Psychogios N, Young N, Wishart DS. MetaboAnalyst: a web server for metabolomic data analysis and interpretation. Nucleic Acids Res. 2009;37:W652–W660. doi: 10.1093/nar/gkp356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Worley B, Halouska S, Powers R. Utilities for quantifying separation in PCA/PLS-DA scores plots. Anal Biochem. 2013;433:102–104. doi: 10.1016/j.ab.2012.10.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Krzywinski M, Schein J, Birol I, Connors J, Gascoyne R, Horsman D, Jones SJ, Marra MA. Circos: an information aesthetic for comparative genomics. Genome Res. 2009;19:1639–1645. doi: 10.1101/gr.092759.109 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data supporting the findings of this study are available from the corresponding author upon reasonable request. Please see the Data Supplement for the Expanded Materials and Methods and Major Resources Table.