Abstract
Background
Prolonged QT intervals are reported in patients with COVID-19. Additionally, virus particles in heart tissue and abnormal troponin levels have been reported. Consequently, we hypothesize that cardiac electrophysiologic abnormalities may be associated with COVID-19.
Methods
This is a retrospective study between March 15th, 2020 and May 30th, 2020 of 828 patients with COVID-19 and baseline ECG. Corrected QT (QTc) and QRS intervals were measured from ECGs performed prior to intervention or administration of QT prolonging drugs. QTc and QRS intervals were evaluated as a function of disease severity (patients admitted versus discharged; inpatients admitted to medical unit vs ICU) and cardiac involvement (troponin elevation >0.03 ng/ml, elevated B-natriuretic peptide (BNP) or NT pro-BNP >500 pg/ml). Multivariable analysis was used to test for significance. Odds ratios for predictors of disease severity and mortality were generated.
Findings
Baseline QTc of inpatients was prolonged compared to patients discharged (450.1±30.2 versus 423.4±21.7 msec, p<0.0001) and relative to a control group of patients with influenza (p=0.006). Inpatients with abnormal cardiac biomarkers had prolonged QTc and QRS compared to those with normal levels (troponin - QTc: 460.9±34.6 versus 445.3±26.6 msec, p<0.0001, QRS: 98.7±24.6 vs 90.5±16.9 msec, p<0.0001; BNP - QTc: 465.9±33.0 versus 446.0±26.2 msec, p<0.0001, QRS: 103.6±25.3 versus 90.6±17.6 msec, p<0.0001). Findings were confirmed with multivariable analysis (all p<0.05). QTc prolongation independently predicted mortality (8.3% increase in mortality for every 10 msec increase in QTc; OR 1.083, CI [1.002, 1.171], p=0.04).
Interpretation
QRS and QTc intervals are early markers for COVID-19 disease progression and mortality. ECG, a readily accessible tool, identifies cardiac involvement and may be used to predict disease course.
Funding
St. Francis Foundation.
Keywords: COVID-19, SAR-COV-2, Cardiac involvement, QT interval, QTc interval, repolarization, QRS, Activation, Troponin, Mortality, Electrocardiogram, BNP, NTPRO BNP
Research in context.
Evidence before this study
QT interval prolongation, reported in patients with coronavirus disease 2019 (COVID-19), is of concern as it may lead to ventricular arrhythmias. Myocardial injury and elevation in biomarkers such as troponin and b-natriuretic peptide (BNP) have been identified early in COVID-19 presentation.
Added value of this study
Recent studies confirm elevated troponin is an independent predictor of outcomes and mortality in COVID-19. However, these reports have not addressed the importance of changes in the electrophysiological properties of the heart or their clinical impact in these patients. In patients with initial ECGs prior to intervention, cardiac involvement (indicated by troponin release or abnormal BNP) and severity of disease (indexed by triage of patients and mortality) was associated with prolonged QTc. Importantly, there was an 8.3% increase in mortality for every 10 msec increase in QTc and a 9.1% increase in mortality for every 10 msec increase in JTc.
Implications of all the available evidence
Our findings demonstrate a novel, significant, and possibly fundamental effect of COVID-19 on the electrophysiologic properties of the heart. More importantly, taken together with cardiac biomarkers and demographics, these changes appear to have significant prognostic value and may help clinicians triage patients by identifying those more likely to need closer outpatient observation or hospital admission.
Alt-text: Unlabelled box
1. Introduction
QT interval prolongation, reported in patients with coronavirus disease 2019 (COVID-19), is of concern as it may lead to ventricular arrhythmias [1]. Early studies demonstrated marked QT prolongation in critically ill patients, often in the intensive care setting [2], which was largely attributed to hydroxychloroquine with or without concomitant azithromycin [2], [3], [4], [5], [6]. However, cardiac repolarization and QT intervals are affected by other factors such as electrolyte imbalances and other QT prolonging agents [1,7]. In addition, since QRS duration is also part of the measured QT interval, prolongation of the QRS may also impact QT/corrected QT (QTc).
Infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) may itself directly and/or indirectly involve the heart. Viral particles have been identified in myocardial tissue [8, 9]. Myocardial injury [10] and elevation in biomarkers such as troponin and b-natriuretic peptide (BNP) have been identified early in COVID-19 presentation [11], [12], [13], [14]. Viral myocarditis in other settings is rarely associated with QT interval prolongation, but may also be a factor in infected patients [4].
We hypothesize that electrocardiogram (ECG) changes described in patients with COVID-19 are related to direct and indirect cardiac involvement of the virus and to severity of disease. We performed our analysis on the initial ECG's of patients with COVID-19 as they first arrived in the emergency department (ED), prior to interventions or exposure to QT prolonging drugs. Analyzing activation and repolarization (represented on the ECG by QRS and QTc) at this early time point provides unique insight into the impact of SARS-CoV-2 infection on the electrophysiologic properties of the heart.
2. Methods
This is a retrospective observational analysis of patients who tested SARS-CoV-2 positive using reverse transcriptase polymerase chain reaction (RT-PCR) test (Cobas SARS-CoV-2, Roche, Indiana, USA and the Xpert Xpress SARS-CoV-2, Cepheid, California, USA) at St. Francis Hospital between March 15th and May 30th, 2020. Patient demographics, co-morbidities, baseline vitals, lab values, imaging data, medications, ECGs, hospital course, and outcomes were collected. AT, JN, JL, SP and BM had access to the data. The study received a waiver of informed consent from the St. Francis Internal Review Board given its retrospective nature of data analysis in anonymized subjects and ethics approval was obtained.
2.1. Data analysis
Our hospital like others in the metro New York area was inundated with COVID-19 patients during the initial surge; only those with severe illness (severe respiratory distress requiring BIPAP or intubation, sepsis with unstable hemodynamics, renal failure, obtundation, or acute myocardial infarction) were admitted to intensive care units (ICU). Inpatients were subcategorized based on their initial disposition on admission, either to a medical unit or ICU. Inpatients were also subcategorized based on those with cardiac involvement (defined as troponin elevation >0.03 mcg/DL, or elevated BNP or NT pro-BNP >500 pg/mL) and those without.
Baseline QRS, QT/QTc, and JTc (msec) were measured and compared between the following groups: inpatients versus outpatients, inpatients admitted to medical unit versus ICU, inpatients with cardiac involvement versus those without.
In addition to measured ECG values, we also evaluated presence or absence of cardiac involvement, as well as ECG intervals as possible predictors of outcomes (severity of disease and/or death). For presence or absence of cardiac involvement the following were used: elevated versus normal troponin and abnormal versus normal BNP; for ECG intervals the following were used: QRS ≥120 msec, QTc ≥500 msec, QTc as a continuous variable, and JTc as a continuous variable. We also evaluated demographics, laboratory values and cardiac biomarkers (including troponin as a continuous variable to control for degree of cardiac involvement) to determine whether the effect of COVID-19 on ECG intervals remained an independent predictor of outcomes. Also a COVID severity index was created by assigning one point for vasopressors, dialysis, and/or mechanical ventilation during the hospitalization; patients were scored from zero to three. The score and in-hospital mortality were assessed for relevance to baseline electrophysiologic parameters.
2.2. ECG review
ECGs recorded at 25 mm/s were blinded for analysis and were analyzed by two or more electrophysiologists using automated or manual calipers. QT intervals were measured manually and corrected using the Bazett method [15]. Where differences were present, the final QTc was determined by consensus. In cases where heart rate differed among groups (inpatient vs outpatient), we confirmed our results using the Fridericia method of QTc calculation [16].
QT intervals of patients with bundle branch blocks were corrected to the maximal normal QRS of 120 msec. To avoid the potential bias, we also did additional analyses using JTc intervals [17], thus correcting not only for differences in QRS within normal ranges, but also for those exceeding QRS of 120 msec. We chose to omit patients with atrial fibrillation to avoid QTc measure bias. Although others [18] have chosen to measure QTc in 3 consecutive beats, we found marked variations in QTc depending upon which three consecutive beats were chosen. Patients with ventricular pacing were excluded due to its known effects on ventricular repolarization.
QRS duration was measured and confirmed by one or more electrophysiologists. Arrhythmias including atrial fibrillation, atrial flutter, ventricular and atrial arrhythmias, right bundle branch block, and left bundle branch block, were confirmed, counted and classified.
Since repolarization changes may be multifactorial (stress of acute illness, QT prolonging drugs, hospitalization, and electrolyte abnormalities), we measured the QTc of 41 consecutive patients hospitalized during 2019 for influenza A, B, or respiratory syncytial virus (RSV) as a control group of hospitalized patients with acute respiratory illness, during a time where there was no COVID-19.
2.3. Statistics
T-tests were used to generate p-values and 95% confidence intervals to analyze continuous variables. Multivariable logistic regression models were fit to assess the impact of putative predictors of QTc, JTc, and QRS duration on mortality and to generate odds ratios (OR) quantifying that impact. Multivariable analyses controlled for heart involvement, age, gender, BMI, potassium level, calcium level, prior use of QTc prolonging medications (including but not limited to anti-depressants, anti-psychotics, anti-arrhythmics, anti-emetics, and antibiotics), history of smoking, hypertension, diabetes, heart disease, atrial fibrillation or flutter, and ventricular tachycardia. A p-value <0.05 was considered statistically significant. No adjustment due to multiplicity was done. All statistical analyses were performed using SAS 9·4 (Cary, NC).
2.3.1. Role of funding source
Funding, provided by St. Francis Hospital Foundation, was used for data collection and analysis. Authors worked on a voluntary basis. The funding source had no role in study design, data analysis, or interpretation.
3. Results
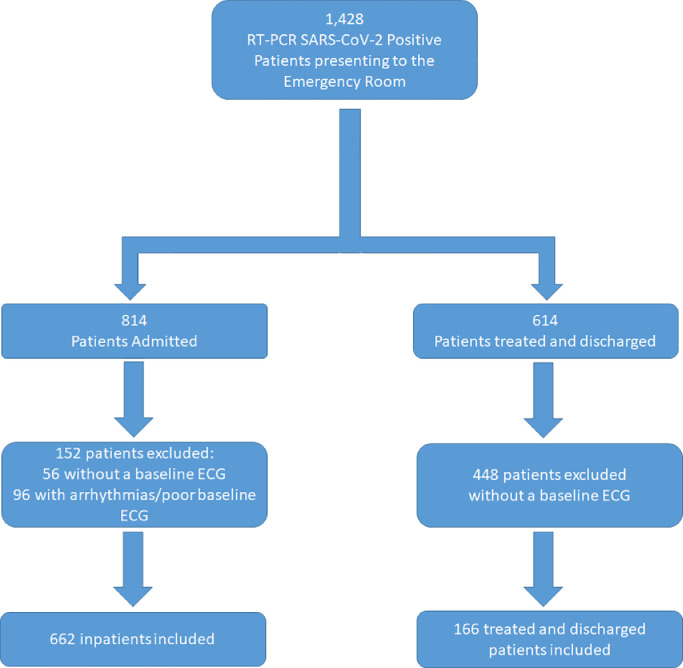
There were 1428 consecutive patients with positive PCR for SARS-CoV-2 treated at St. Francis Hospital Emergency Department (ED) between March 15th to May 30th 2020; 814 were admitted to the hospital from the ED and 614 were treated and discharged (outpatients). Of the 814 patients admitted, 56 were without a baseline ECG and an additional 96 were excluded from analysis due to relative inaccuracy of QTc measurements (atrial fibrillation (62), ventricular paced rhythm (33), or poor baseline ECG (1)), leaving 662 for analysis. Of these 662 inpatients, 579 were admitted to a medical unit, and 83 to ICU. Forty patients had right bundle branch block, 17 left bundle branch block, 11 non-specific interventricular conduction delay, and 37 ventricular ectopy. Of the 614 ED patients treated and discharged, 166 had baseline ECG's. None of these patients had rhythms that would cause relative inaccuracy of QTc measurements and therefore all were included in the analyses. A total of 828 patients with baseline ECGs were included in the final analysis (Fig. 1). The mean age of the entire patient cohort was 66±16 years, 46.5% had hypertension, 27.8% had diabetes, 27.3% had preexisting heart disease, and mean BMI was 28.7±6.4 kg/m2. Demographics and clinical characteristics of the cohort is shown in Table 1.
Fig. 1.
Flow diagram illustrating the total patient cohort of 1428. Of the 814 patients admitted, 152 were excluded, leaving 662 for analysis. Of the 614 patients discharged home from the emergency room, 448 patients were excluded due to not having a baseline ECG, leaving 166 treated and discharged patients.
Table 1.
Demographics and clinical characteristics of our patient population.
| Inpatient(n=662) | Treat and discharge(n=166) | p-value | |
|---|---|---|---|
| Age (years, SD) | 68 ± 15 | 53 ± 16 | <0.001 |
| Body mass Index (kg/m2, SD) | 28·4 ± 6·7 | 28·2 ± 8.3 | 0.908 |
| Female – n (%) | 284 (42.9) | 89 (53.6) | 0.013 |
| Hypertension – n (%) | 411 (62.1) | 49 (30.5) | <0.001 |
| Current or Former smoker | 262 (39.6) | 49 (26.5) | 0.032 |
| Hyperlipidemia – n (%) | 325 (49.1) | 40 (24.1) | <0.001 |
| Diabetes mellitus – n (%) | 210 (31.7) | 15 (9.0) | <0.001 |
| Stroke – n (%) | 44 (6.6) | 1 (0.6) | 0.002 |
| Mean Heart Rate (beats per minute) | 87.3 ± 17.5 | 80·6 ± 14.9 | <0.001 |
| History of Arrhythmia – n (%) | 155 (23.4) | 20 (12.0) | 0.001 |
| Premature Ventricular contraction – n (%) | 28 (4.3) | 2 (1.2) | 0.062 |
| Non-sustained ventricular tachycardia – n (%) | 10 (1.5) | 1 (0.6) | 0.361 |
| Ventricular tachycardia – n (%) | 7 (1.1) | 0 (0) | 0.183 |
| Torsade de pointes – n (%) | 0 (0) | 0 (0) | |
| Ventricular fibrillation – n (%) | 0 (0) | 0 (0) | |
| Atrial fibrillation – n (%) | 79 (11.9) | 6 (3.6) | 0.002 |
| Atrial flutter – n (%) | 17 (2.6) | 5 (3.0) | 0.750 |
| Supraventricular tachycardia – n (%) | 14 (2.1) | 2 (1.2) | 0.446 |
| History of End stage renal disease – n (%) | 1 (0.1) | 0 (0) | 0.616 |
| History of Coronary Artery Disease– n (%) | 176 (26.6) | 17 (10.2) | <0.001 |
| Previous MI – n (%) | 47 (7.1) | 9 (5.4) | 0.441 |
| Previous PCI – n (%) | 100 (15.1) | 9 (5.4) | <0.001 |
| Previous CABG – n (%) | 55 (8.3) | 4 (2.4) | 0.008 |
| Congestive Heart Failure – n (%) | 70 (10.5) | 3 (1.8) | <0.001 |
Abbreviations: MI = myocardial infarction; PCI = percutaneous coronary intervention; CABG = coronary artery bypass graft.
The admitted group was older, predominantly male, more likely to have hypertension, diabetes, hyperlipidemia, previous history of stroke, arrhythmia, and heart disease when compared with outpatients.
3.1. ECG intervals in patients with COVID-19
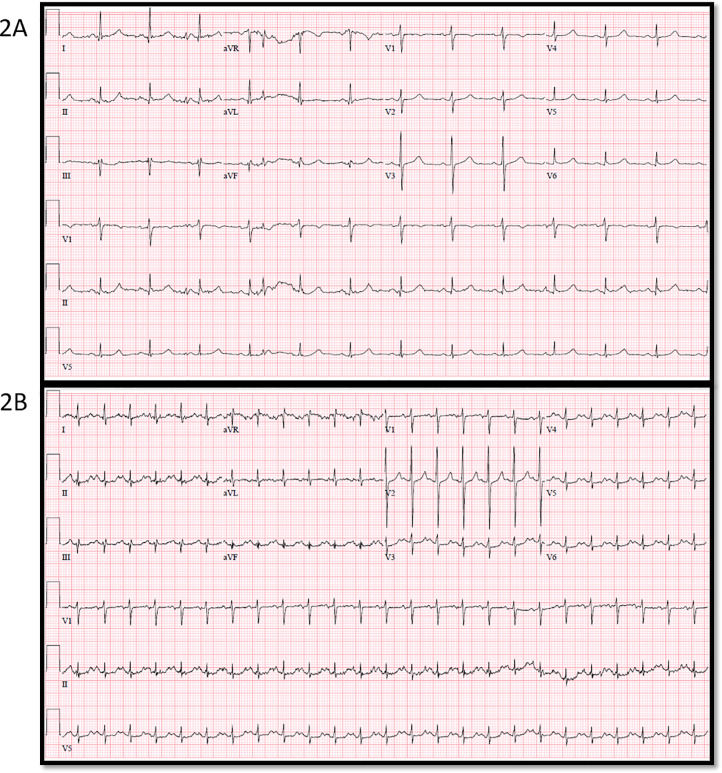
Shown in Fig. 2A is an ECG of a patient taken one month prior to developing COVID-19. Note the normal QTc (436 msec) interval. Fig. 2B is the ECG in the ED one month later when the same patient presented acutely ill with COVID-19. There was markedly prolonged QTc (535 msec) interval despite the patient having normal electrolytes and no exposure to QT prolonging drugs.
Fig. 2.
Example of a 68 year old female, history of congestive heart failure and follicular lymphoma, presenting with shortness of breath, cough and malaise and was found to have COVID-19. Troponin was elevated. 2A: Electrocardiogram done one month prior with normal corrected QT (QTc) (436 msec). 2B: Electrocardiogram on presentation to the emergency room with severely prolonged QTc (535 msec).
In our entire cohort, the average QTc was 444.7±30.6 msec (range 364 to 557 msec corrected for QRS>120). QTc prolongation was longer in those who were older, had history of atrial fibrillation/ flutter, bundle branch block, and history of cardiac disease (Table 2). Prolonged QTc interval (QTc > 460 msec) was noted on the naïve, baseline ECG in 226 (27.3%) of our patients and 49 (5.9%) patients had QTc >500 msec. Average baseline QRS duration was 92.6±19.9 msec (range 56 to 240 msec). There were 78 patients with QRS>120 msec.
Table 2.
Univariate of multiple factors correlated with QTc prolongation.
| Variable | Correlation | p-value |
|---|---|---|
| Age at admit | 0.20 | <0.0001 |
| Overall Cardiac Involvement | 0.26 | <0.0001 |
| Elevated Troponin | 0.24 | <0.0001 |
| Creatinine | 0.20 | <0.0001 |
| History of LBBB | 0.18 | <0.0001 |
| History of RBBB | 0.18 | <0.0001 |
| History of Atrial fibrillation | 0.18 | <0.0001 |
| History of Atrial flutter | 0.13 | 0.0011 |
| History of PVC | 0.12 | 0.0028 |
| History of Ventricular Tachycardia | 0.09 | 0.0198 |
| Potassium level | −0.08 | 0.0534 |
| Calcium level | −0.09 | 0.0228 |
| History of outpatient QT prolonging medications | 0.08 | 0.0319 |
| Hypertension | 0.13 | 0.0005 |
| Stroke | 0.17 | <0.0001 |
| History of cardiac disease | 0.14 | 0.0002 |
| Previous MI | 0.14 | 0.0003 |
| History of CAD | 0.18 | <0.0001 |
| History of Congestive Heart Failure | 0.29 | <0.0001 |
Abbreviations: LBBB = Left bundle branch block; RBBB = Right bundle branch block; PVC = Premature ventricular contraction; CAD = Coronary artery disease.
3.1.1. Effects of cardiac involvement in COVID-19 on cardiac activation and repolarization
In the 339 patients who were tested for cardiac biomarkers (troponin and/or BNP), 126 (37.2%) had elevated troponin, 47 (13.9%) had elevated BNP, and 40 (11.8%) had elevation of both. There were 228 patients classified with cardiac involvement versus 385 without. As troponin abnormalities are likely more specific for active myocardial injury, whereas BNP may be associated with direct or indirect cardiac effects, these factors were analyzed separately.
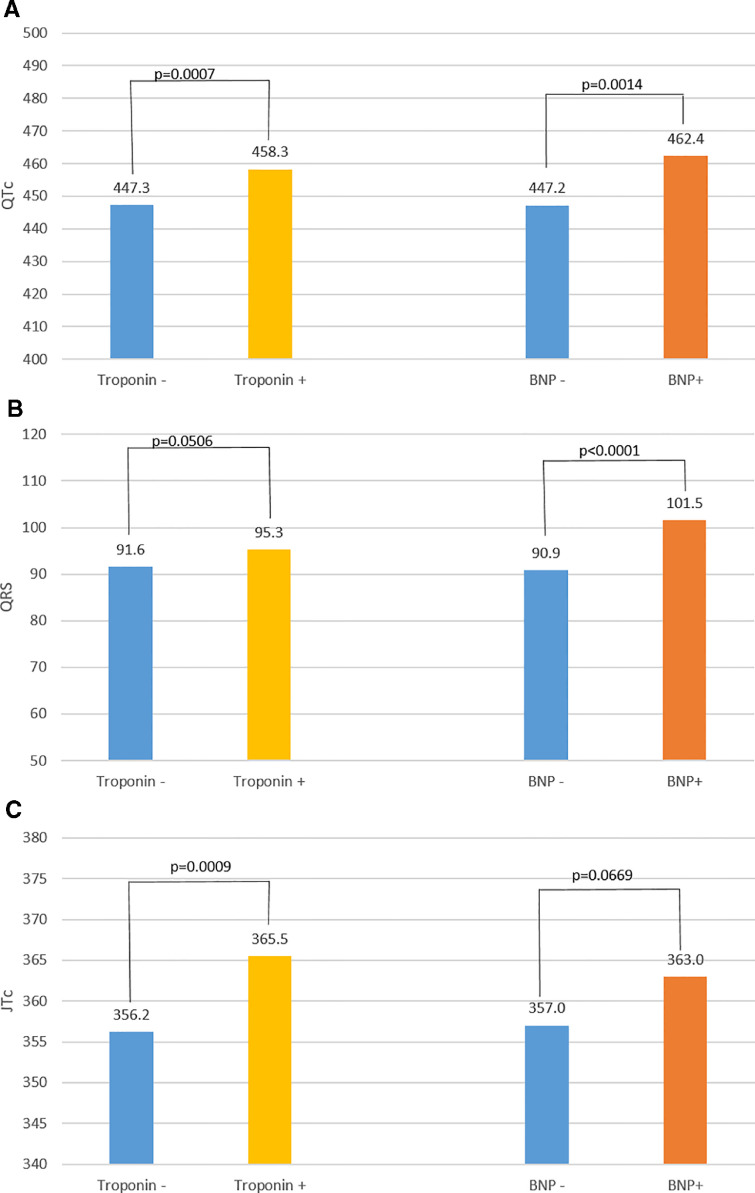
QTc was prolonged in patients with elevated versus normal troponin (460·9±34.6 msec versus 445.3±26.6 msec, p<0.0001) and in patients with abnormal versus normal BNP/ NT-Pro BNP (465.9±33.0 msec versus 446.0±26.2 msec, p<0.0001). These associations remained significant in multivariable analysis when considering other factors found to be significant in the univariate analysis (troponin: p=0.0007; BNP/NT-Pro, BNP: p=0.0014; Fig. 3A).
Fig. 3.
A: Adjusted mean corrected QT (QTc) values, 3B: Adjusted mean QRS values, 3C: Adjusted mean JTc values, for patients with and without cardiac involvement (positive troponin or B-natriuretic peptide [BNP]). Troponin and BNP were evaluated separately. Multivariable analysis was controlled for age, gender, body mass index, history of diabetes mellitus, hypertension, premature ventricular contractions, atrial fibrillation/flutter, ventricular tachycardia, QT prolonging medications, potassium level and QRS duration when applicable.
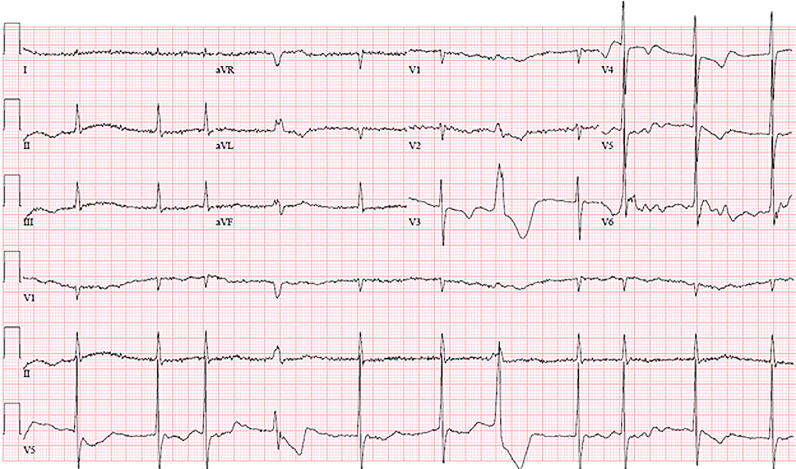
QRS duration was longer in those with elevated troponin (98.7±24.6 versus 90.5±16.9 msec, p<0.0001) and abnormal BNP/NT-Pro BNP (103.6±25.3 versus 90·6±17.6, p<0.0001). In multivariable analysis, QRS duration remained significant with elevated troponin and with abnormal BNP/NT-Pro BNP (p=0.0506 and p<0.0001, respectively; Fig. 3B). Examples of ECG's with these findings in two patients with acute myocarditis associated with COVID-19 are shown in Fig. 4, Fig. 5.
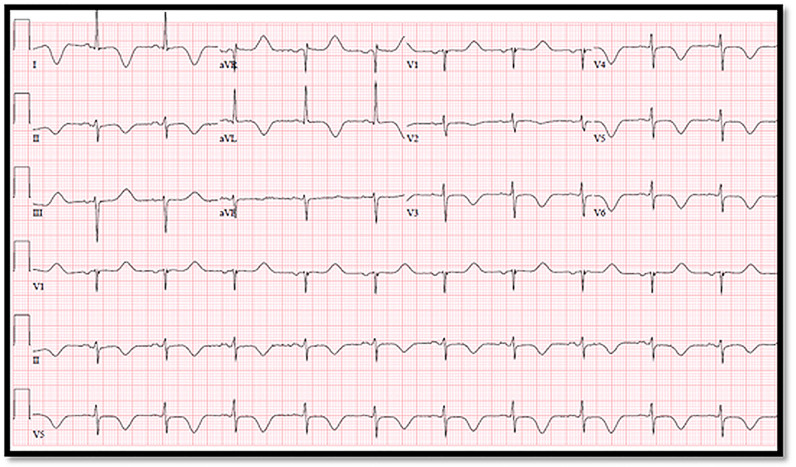
Fig. 4.
56 year old female, history of hypertension and diabetes mellitus, admitted with cough, sore throat and fatigue. Developed an abnormal electrocardiogram and positive troponin thought to be most consistent with COVID-19 myocarditis. Echocardiogram was done showing diffuse wall motion abnormalities. Follow up electrocardiogram showed marked corrected QT prolongation and right ventricular strain pattern.
Fig. 5.
75 year old female, history of hyperlipidemia, congestive heart failure, atrial fibrillation, mechanical mitral valve replacement on warfarin, presented to the emergency room due to poor oral intake and lethargy. Found to have COVID-19. Electrocardiogram on initial presentation is shown above. Both troponin (0.04 ng/mL) and NT Pro-BNP (2651 pg/mL) were abnormal. A later echocardiogram showed left ventricular and right ventricular dysfunction and severely elevated pulmonary artery pressure. Note the prolonged corrected QT interval, late coupled premature ventricular contraction and right ventricular strain pattern.
QRS duration was also an independent predictor of QTc prolongation in these patients, suggesting QT interval prolongation was at least in part due to conduction abnormalities from COVID-19. To better isolate repolarization changes alone, we analyzed JTc (QTc-QRS in msec). JTc was prolonged in patients with elevated troponin (JTc 365.1±32.3 msec versus JTc 355.9±25 msec, p=0.0016) and in those with abnormal BNP/NT-pro BNP (JTc 366.4±29.3 msec versus JTc 357.0±26.1 msec, p=0.0042). Elevated troponin remained significant in multivariable analysis (p=0.0009) while abnormal BNP/NT pro-BNP trended towards significance (p=0.0669) (Fig. 3C).
3.2. Severity of COVID-19 impacts cardiac activation and repolarization
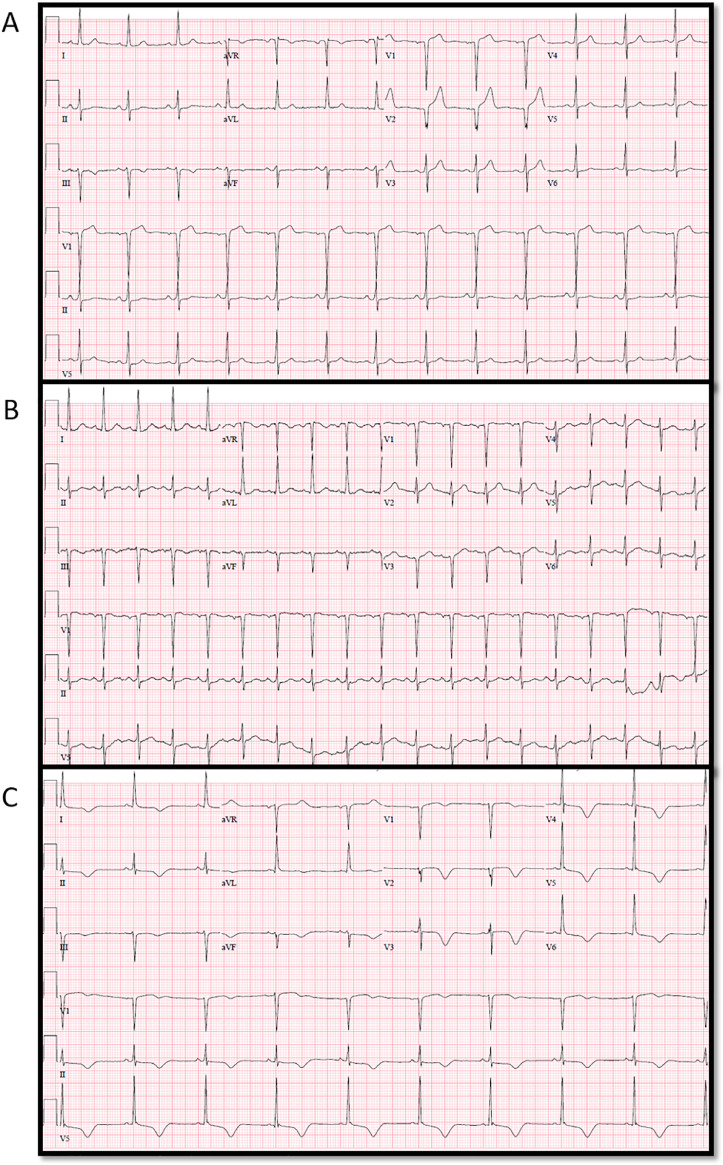
Disease severity was a determinant of QTc, both in individual patients as disease progressed as well as across groups of patients, depending on severity of illness at the time of presentation. Shown in Fig. 6 is a patient who presented with COVID-19 and relatively mild disease on initial presentation to the ED. Note Fig. 6A with normal QRS and QTc interval. He was discharged home. He returned with worsening symptoms two weeks later, but not critically ill. Fig. 6B shows ECG on return and admission to medical unit. He subsequently deteriorated with multiple bouts of sepsis. Fig. 6C shows prolonged QTc with RV strain pattern. He eventually succumbed to the disease.
Fig. 6.
Example of 62 year old male, past medical history of diabetes, hyperlipidemia, coronary artery disease status post percutaneous coronary intervention, presented to the emergency department (ED) with mild symptoms of COVID-19. Fig. 6A is the electrocardiogram from the ED on initial presentation. He was discharged home, however, he worsened while at home and returned to the ED two weeks later. 6B is the electrocardiogram from the return ED visit. He was not critically ill and was admitted to the floor. A few days into his admission, he developed respiratory distress and sepsis, requiring intubation and transfer to the intensive care unit. Troponin was positive and echocardiogram showed new right ventricular dysfunction. 6C is the electrocardiogram after the patient began to deteriorate.
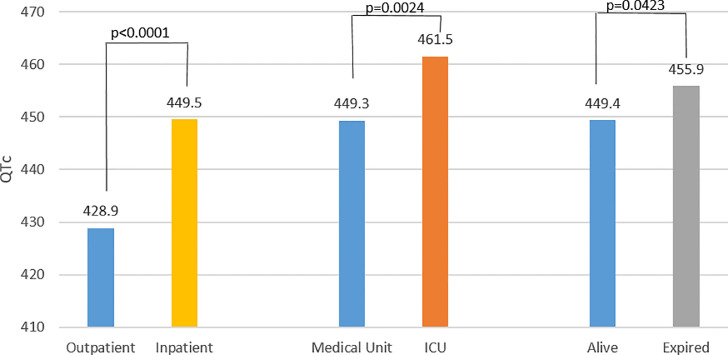
We found these abnormalities across patient populations; that is, QTc varied depending upon severity of illness at the time of ED presentation. The average baseline QTc was significantly longer for inpatients compared to outpatients (450.1±30.2 msec versus 423.4±21.7 msec, p<0.0001). After dichotomization of the inpatients by their disposition from the ED (either to medical unit or ICU), the average QTc of ICU patients was longer than medical unit patients (QTc 458.7±34.0 msec versus 449.2±29.4 msec, p=0.0113). Multivariable analysis confirmed the significance of these relationships (inpatients versus outpatients, p<0.0001; ICU versus medical unit, p=0.0024; Fig. 7). All the findings are noted in Table 3.
Fig. 7.
Correlation of corrected QT (QTc) with severity of disease. First set of bars represent the QTc of the treat and discharged (outpatient) cohort compared to the inpatient cohort, followed by patients admitted to the medical unit versus ICU. The QTc was prolonged as a function of severity of disease as indexed by disposition. This remained significant after controlling for: age, gender, body mass index, history of diabetes mellitus, hypertension, premature ventricular contractions, atrial fibrillation/flutter, ventricular tachycardia, QT prolonging medications, potassium level, and QRS duration.
Table 3.
Comparison of QTc, QRS, and JTc measurements in various subgroup in unadjusted and multivariable model.
| QTC | p-value | QTC Adjusted* | p-value | ||||
|---|---|---|---|---|---|---|---|
| Cardiac Involvement (BNP/NT-Pro BNP and/or Troponin Positive) | 444.5 (25.2) (normal) |
460.7 (35.1) (abnormal) |
<0.0001 | Cardiac Involvement (BNP/NT-Pro BNP and/or Troponin Positive) | 446.4 (normal) |
458.6 (abnormal) |
<0.0001 |
| BNP/NT-Pro BNP Positive Only | 446.0 (26.2) (normal) |
465.9 (33.0) (abnormal) |
<0.0001 | BNP/NT-Pro BNP Positive Only | 447.2 (normal) |
462.4 (abnormal) |
<0.0014 |
| Troponin Positive Only | 445.3 (26.6) (normal) |
460.9 (34.6) (abnormal) |
<0.0001 | Troponin Positive Only | 447.3 (normal) |
458.3 (abnormal) |
0.0007 |
| Discharged from Emergency Department | 423.4 (21.7) (discharged) |
450.1 (30.2) (admitted) |
<0.0001 | Discharged from Emergency Department | 428.9 (discharged) |
449.5 (admitted) |
<0.0001 |
| ICU | 458.7 (34.0) (ICU) |
449.2 (29.4) (Floor) |
0.0113 | ICU | 461.5 (ICU) |
449.3 (Floor) |
0.0024 |
| Survived | 448.1 (28.5) (Survived) |
461.0 (35.2) (Died) |
0.0006 | Survived | 449.4 (Survived) |
455.9 (Died) |
0.0423 |
| QRS | p-value | QRS Adjusted* | p-value | ||||
| Cardiac Involvement (BNP/NT-Pro BNP and/or Troponin Positive) | 90 (16.2) (normal) |
98.8 (24.5) (abnormal) |
<0.0001 | Cardiac Involvement (BNP/NT-Pro BNP and/or Troponin Positive) | 91.1 (normal) |
96.0 (abnormal) |
0.0083 |
| BNP/NT-Pro BNP Positive Only | 90.6 (17.6) (normal) |
103.6 (25.3) (abnormal) |
<0.0001 | BNP/NT-Pro BNP Positive Only | 90.9 (normal) |
101.5 (abnormal) |
<0.0001 |
| Troponin Positive Only | 90.5 (16.9) (normal) |
98.7 (24.6) (abnormal) |
<0.0001 | Troponin Positive Only | 91.6 (normal) |
95.3 (abnormal) |
0·0506 |
| Discharged from Emergency Department | 90.7 (15.6) (discharged) |
93.1 (20.8) (admitted) |
0.101 | Discharged from Emergency Department | 94.9 (discharged) |
92.0 (admitted) |
0.1864 |
| ICU | 94.9 (22.2) (ICU) |
93.0 (20.6) (Floor) |
0.4808 | ICU | 95.3 (ICU) |
92.6 (Floor) |
0.3112 |
| Survived | 92.1 (19·5) (Survived) |
98.9 (25.5) (Died) |
0.0101 | Survived | 92.5 (Survived) |
94.4 (Died) |
0.4079 |
| JTc | p-value | JTc Adjusted* | p-value | ||||
| Cardiac Involvement (BNP/NT-Pro BNP and/or Troponin Positive) | 354.8 (26.3) (normal) |
364.2 (31.2) (abnormal) |
0.0002 | Cardiac Involvement (BNP/NT-Pro BNP and/or Troponin Positive) | 355.9 (normal) |
364.6 (abnormal) |
0.0012 |
| BNP/NT-Pro BNP Positive Only | 357.0 (26.1) (normal) |
366.4 (29.3) (abnormal) |
0.0042 | BNP/NT-Pro BNP Positive Only | 357.0 (normal) |
363.0 (abnormal) |
0.0669 |
| Troponin Positive Only | 355.9 (25.0) (normal) |
365.1 (32.3) (abnormal) |
0.0016 | Troponin Positive Only | 356.2 (normal) |
365.5 (abnormal) |
0.0009 |
| ICU | 363.5 (29.4) (ICU) |
357.2 (28.6) (Floor) |
0.0827 | ICU | 366.1 (ICU) |
357.8 (Floor) |
0.0296 |
| Survived | 356.6 (27.7) (Survived) |
364.9 (33) (Died) |
0.0161 | Survived | 357.6 (Survived) |
363.4 (Died) |
0.0641 |
*adjusted for age, gender, body mass index, history of hypertension, diabetes, atrial fibrillation/flutter, ventricular tachycardia, any cardiac history, QT prolonging medications, calcium level, and potassium level.
Abbreviations: BNP = B-natriuretic peptide; ICU = Intensive Care Unit; QTc = Corrected QT interval.
There was no difference in QRS duration in inpatients versus outpatients (93.1±20.8 msec versus 90.7±15.6 msec, p=0.1010) or ICU versus medical unit patients (94.9±22.2 msec versus 93.0±20.6 msec, p=0.4808). Thus the QTc effect associated with disease severity was entirely attributable to abnormalities of repolarization. Because the heart rate was 7 beats per minute higher in the inpatient group versus the outpatient group, although minimal in magnitude, we confirmed the significance of QTc prolongation using the Fridericia method (less sensitive to overcorrection of the QTc at rapid heart rates) (p<0.0001).
The QTc in inpatients with COVID-19 was prolonged relative to those with influenza/RSV (450.1±30.2 msec vs 436.9±24.4; p=0.006). There were no differences between the groups with respect to serum potassium or abnormal troponin (p>0.10); the influenza group was older and more likely to be taking QT prolonging drugs (p<0.05) (more likely associated with prolonged QT). COVID-19 patients also had minimally lower calcium and albumin levels (p<0.05), which remained in the normal range and likely would not have a significant impact on QTc.
3.3. ECG abnormalities predict outcomes and mortality
For outcomes, we defined a severity index (intubation, dialysis, vasopressors) with each outcome assigned one point. Patients with higher severity index were older and had higher rates of elevated troponin and abnormal BNP. All other variables were similar. Those with indices 1-3 had longer QTc and JTc intervals compared to those with a zero index, (QTc: 457.5±35.6 msec versus 448.2±28.1 msec, p=0.0049; JTc: 363.4±30.2 msec versus 356.0±27.6 msec, p=0.011).
QTc and JTc on baseline ECG predicted a higher severity index. For every 10 msec increase in QTc, there was a 16% increased chance of higher severity index (p=0.0005) and for every 10 msec increase in JTc there was a 13.7% increased chance of a higher severity index (p=0.0032). QTc ≥500 msec further correlated with a higher severity index (p=0.0009), while QRS duration was not a predictor of severity index.
Finally, we evaluated mortality as a function of various factors including abnormal ECG findings. Those who died during admission (112 patients) had longer QTc on their baseline ECG (461.0±35.2 msec versus 448.1±28.5 msec, p=0.0006). This finding remained significant with multivariable analysis (expired versus alive, p=0.04; Figure 7). QTc prolongation >500 msec upon ED admission trended as a predictor of mortality (OR 2.03, CI [0.967, 4.27], p=0.06). Importantly, there was an 8.3% increase in mortality for every 10 msec increase in QTc (OR 1.083, CI [1.002, 1.171], p=0.04) and a 9.1% increase in mortality for every 10 msec increase in JTc (OR 1.091, CI [1.008, 1.181], p=0.03). QRS duration was not a predictor of mortality.
Other factors that predicted mortality include: (elevated troponin (OR 5.59, CI [3.26, 9.62], p<0.0001), admission to ICU (OR 5.49, CI [2.85, 10.58], p<0.0001), and new atrial fibrillation during admission (OR 2.64, CI [1.16, 6.06], p=0.02)). Despite these factors, ECG changes remained independent predictors of outcomes and mortality.
4. Discussion
SARS-CoV-2 infection is associated with prolongation in activation (QRS) and repolarization (QTc). The severity of infection (indexed by triage of patients and mortality) is associated with prolongation in activation and repolarization. Prolonged QRS and QTc intervals are also associated with cardiac involvement, indicated by troponin release or abnormal BNP/NT pro-BNP. Finally, changes in activation and repolarization are independent predictors of disease severity and mortality. Taken together with cardiac biomarkers and other demographics, cardiac electrophysiologic changes seen in COVID-19 provide an early marker for subsequent clinical course and death and may help in initial triage of infected patients.
Our study is unique as we evaluated initial ECGs in patients prior to exposure to QTc prolonging medications such as hydroxychloroquine, macrolide antibiotics or antiviral agents; hence we were able to evaluate ECG parameters independent of interventions that could confound our results. Therefore, our findings demonstrate a novel, significant, and possibly fundamental, effect of COVID-19 on the electrophysiologic properties of the heart.
Although repolarization and QTc changes reported by others in hospitalized and critically ill patients are likely multifactorial, due in part to drugs, interventions and electrolyte imbalance [1], [2], [3], [4], [5], [6], [7] our study shows that direct cardiac involvement by SARS-CoV-2 (indexed by biomarkers) as well as severity of disease, are independent factors in prolongation of these parameters. Our study therefore extends the findings of Russell et al. [[1], [2], [3], [4], [5], [6], [7],19]; not only does QTc prolongation in an ICU population predict mortality, but significant QTc prolongation upon ED presentation in COVID-19 patients predicts the same. Our findings demonstrate a loss of repolarization reserve which may have implications on the safety of a variety of antibiotics, antiviral agents, and other medications affecting QT intervals. Not only is this important for monitoring and choice of therapy in patients with COVID-19 who have evidence of new QT prolongation, but may also be cause to monitor QT intervals in patients with COVID-19 who were previously stable on QT prolonging agents such as antidepressants and antiarrhythmics.
Myocarditis and troponin release have been reported in COVID-19 [20,21], as has direct invasion of myocardium with SARS-CoV-2 particles [8,9]. Recent studies confirm elevated troponin is an independent predictor of outcome and mortality [22,23]. However, these reports have not addressed the importance of changes in the electrophysiological properties of the heart or their clinical impact in these patients. We have shown that QTc and JTc prolongation on the baseline ECG are independent predictors of progression to severe disease and mortality. Strikingly, we found that for every 10 msec increase in QTc, there was an 8.3% increase in mortality. We also confirmed that elevated troponin is associated with five-fold increased mortality in patients with COVID-19. Hence our study confirms the presence of abnormal cardiac biomarkers in patients with COVID-19, and extends the findings of others by demonstrating the relationship between cardiac involvement, ECG intervals, and mortality.
Reports of other forms of myocarditis showing prolongation of the QTc and QRS interval are limited to case reports and small series in which interval prolongation was associated with fulminant disease [24], [25], [26] and did not always predict mortality [25,26]. In our study, we present data in a large patient population suffering from infection of a single type of virus and we confirm and extend their finding that QRS and QTc/JTc intervals are both prolonged in our sickest patients and are independent predictors of mortality.
The mechanism of the ECG changes is unknown. Basic cellular repolarization may be affected by direct and indirect effects of COVID-19 on the heart. Direct cardiac infiltration of the virus may disrupt the cardiomyocyte membrane potential and gap junctions and lead to dysfunction in the conduction system. Cytokines have a direct effect on action potential duration by inducing changes in expression and function of calcium and potassium channels [27,28], and are hallmarks of COVID-19. Myocardial strain (as indexed by abnormal BNP/ NT pro-BNP levels) due to heart failure or resulting from pulmonary and vascular abnormalities caused by COVID-19 may also play a role. Lengthening of action potential duration caused by myocardial cell stretch has been demonstrated in experimental studies, but only with decreased myocardial contractility [28], possibly lengthening in some patients with COVID-19 who have LV or RV dysfunction. Whatever the mechanism, it remains that myocardial activation and repolarization are prolonged in patients with COVID-19, and only rarely reported in other cases of fulminant myocarditis.
That these effects are related to COVID-19 per se is also supported by our finding of prolonged QTc in COVID-19 patients relative to our influenza control group.
Hydroxychloroquine was not a factor in our evaluation as we took care to study initial ECG's before exposure to hydroxychloroquine or other QT prolonging drugs. Early reports that attributed QTc prolongation to hydroxychloroquine with or without azithromycin showed incidence of QTc prolongation of approximately 10–20% [29,30]. The QTc was similarly prolonged on drug naïve ECG's in some of our patients (27.3% of patients had abnormal QTc >460 msec at presentation and 5.9% had QTc >500 msec), therefore COVID-19 per se may contribute to the overall incidence of QTc prolongation noted. We also found that in some cases, there was further prolongation of QTc during hospitalization even without exposure to hydroxychloroquine or other QT prolonging drugs.
Our study provides new insight into cardiac electrophysiologic changes associated with COVID-19 affecting (directly or indirectly) ventricular activation and repolarization. More importantly, taken together with cardiac biomarkers and demographics, these changes appear to have significant prognostic value and may well help clinicians triage patients, identifying those more likely to need closer outpatient observation or hospital admission. This is important in countries with highly developed medical systems, and critical in areas of the world where health care resources are more limited and appropriate utilization is essential.
As a retrospective review of patients, many factors were not controlled for. Nor, in fact, could they be in a prospective study as COVID-19 affects many organ systems, creating marked heterogeneity in presentation. During the initial surge, the threshold for ICU admission may have differed compared to today; however, our results hold for patients who met criteria for ICU admission at the time. In addition, because of limitations in accuracy and availability of SARS-CoV-2 testing early in the pandemic, it was impossible to have a control group from the same time frame with respiratory viral illness proven not to have COVID-19. Hence, we could not perfectly tease out the electrophysiologic effects due directly and purely to SARS-CoV-2 versus those due to other clinical manifestations.
The strength of our study is the large patient population with admitting ECGs studied at an early time of the pandemic allowing us to see the full manifestation and heterogeneity of presentation of the illness prior to confounding interventions.
COVID-19 and/or its clinical manifestations has intrinsic effects on cardiac activation and repolarization related directly to cardiac biomarkers and illness severity and these effects are independent of other confounding variables, notably QT prolonging drugs and electrolyte imbalance. The ECG, a readily available tool, identifies cardiac involvement and may be used to predict cardiac involvement and mortality.
Funding
St. Francis Hospital Foundation (Roslyn, NY)
Data sharing statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Author contributions
AT, JN, SP, SM, JL were involved in the conception/design of the work. AT, JN, SP, BS, DK, BM, JS, JL were involved in the acquisition, analysis, and/or interpretation of the data. AT, JN, JJC, CL, JL were involved in drafting and/or revision of the manuscript. All authors read and approved the final manuscript
Declaration of Competing Interest
All authors have no conflict of interest to report.
References
- 1.Priori SG, Schwartz PJ, Napolitano C, Bloise R, Ronchetti E, Grillo M. Risk stratification in the long-QT syndrome. N Engl J Med. 2003;348(19):1866–1874. doi: 10.1056/NEJMoa022147. [DOI] [PubMed] [Google Scholar]
- 2.Bessiere F, Roccia H, Deliniere A, Charriere R, Chevalier P, Argaud L. Assessment of QT intervals in a case series of patients with Coronavirus disease 2019 (COVID-19) infection treated with Hydroxychloroquine alone or in combination with azithromycin in an intensive care unit. JAMA Cardiol. 2020;5(9):1067–1069. doi: 10.1001/jamacardio.2020.1787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chorin E, Dai M, Shulman E, Wadhwani L, Bar-Cohen R, Barbhaiya C. The QT interval in patients with COVID-19 treated with hydroxychloroquine and azithromycin. Nat Med. 2020;26(6):808–809. doi: 10.1038/s41591-020-0888-2. [DOI] [PubMed] [Google Scholar]
- 4.Maraj I, Hummel JP, Taoutel R, Chamoun R, Workman V, Li C. Incidence and determinants of QT interval prolongation in COVID-19 patients treated with hydroxychloroquine and azithromycin. J Cardiovasc Electrophysiol. 2020;31(8):1904–1907. doi: 10.1111/jce.14594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Voisin O, Lorc’h EL, Mahe A, Azria P, Borie MF, Hubert S. Acute QT interval modifications during hydroxychloroquine-azithromycin treatment in the context of COVID-19 infection. Mayo Clin Proc. 2020;95(8):1696–1700. doi: 10.1016/j.mayocp.2020.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mercuro NJ, Yen CF, Shim DJ, Maher TR, McCoy CM, Zimetbaum PJ. Risk of QT interval prolongation associated with use of hydroxychloroquine with or without concomitant azithromycin among hospitalized patients testing positive for Coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5(9):1036–1041. doi: 10.1001/jamacardio.2020.1834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kramer DB, Zimetbaum PJ. Long-QT syndrome. Cardiol Rev. 2011;19(5):217–225. doi: 10.1097/CRD.0b013e3182203504. [DOI] [PubMed] [Google Scholar]
- 8.Puelles VG, Lutgehetmann M, Lindenmeyer MT, Sperhake JP, Wong MN, Allweiss L. Multiorgan and renal tropism of SARS-CoV-2. N Engl J Med. 2020;383(6):590–592. doi: 10.1056/NEJMc2011400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Xu Z, Shi L, Wang Y, Zhang J, Huang L, Zhang C. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lala A, Johnson KW, Januzzi JL, Russak AJ, Paranjpe I, Richter F. Prevalence and impact of myocardial injury in patients hospitalized with COVID-19 infection. J Am Coll Cardiol. 2020;76(5):533–546. doi: 10.1016/j.jacc.2020.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Doyen D, Moceri P, Ducreux D, Dellamonica J. Myocarditis in a patient with COVID19: a cause of raised troponin and ECG changes. Lancet. 2020;395(10235):1516. doi: 10.1016/S0140-6736(20)30912-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shah P, Doshi R, Chenna A, Owens R, Cobb A, Ivey H. Prognostic value of elevated cardiac troponin i in hospitalized Covid-19 patients. Am J Cardiol. 2020;135:150–153. doi: 10.1016/j.amjcard.2020.08.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sandoval Y, Januzzi JL, Jr., Jaffe AS. Cardiac troponin for assessment of myocardial injury in COVID-19: JACC review topic of the week. J Am Coll Cardiol. 2020;76(10) doi: 10.1016/j.jacc.2020.06.068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Si D, Du B, Ni L, Yang B, Sun H, Jiang N. Death, discharge and arrhythmias among patients with COVID19 and cardiac injury. CMAJ. 2020;192(28):E791–E798. doi: 10.1503/cmaj.200879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bazett H. An analysis of the time-relations of electrocardiograms. Heart. 1920;7:353–370. [Google Scholar]
- 16.Fridericia LS. The duration of systole in an electrocardiogram in normal humans and in patients with heart disease. 1920. Ann Noninvasive Electrocardiol. 2003;8(4):343–351. doi: 10.1046/j.1542-474X.2003.08413.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chiladakis J, Kalogeropoulos A, Koutsogiannis N, Zagkli F, Vlassopoulou N, Chouchoulis K. Optimal QT/JT interval assessment in patients with complete bundle branch block. Ann Noninvasive Electrocardiol. 2012;17(3):268–276. doi: 10.1111/j.1542-474X.2012.00528.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mazzanti A, Briani M, Kukavica D, Bulian F, Marelli S, Trancuccio A. Association of hydroxychloroquine with QTc interval in patients with COVID-19. Circulation. 2020;142(5):513–515. doi: 10.1161/CIRCULATIONAHA.120.048476. [DOI] [PubMed] [Google Scholar]
- 19.Russell H, Churilov L, Toh L, Eastwood GM, Bellomo R. The incidence, predictors and outcomes of QTc prolongation in critically ill patients. J Crit Care. 2019;1(54):244–249. doi: 10.1016/j.jcrc.2019.09.014. [DOI] [PubMed] [Google Scholar]
- 20.Inciardi RM, Lupi L, Zaccone G, Italia L, Raffo M, Tomasoni D. Cardiac involvement in a patient with Coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5(7):819–824. doi: 10.1001/jamacardio.2020.1096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sala S, Peretto G, Gramegna M, Palmisano A, Villatore A, Vignale D. Acute myocarditis presenting as a reverse TakoTsubo syndrome in a patient with SARS-CoV-2 respiratory infection. Eur Heart J. 2020;41(19):1861–1862. doi: 10.1093/eurheartj/ehaa286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guo T, Fan Y, Chen M, Wu X, Zhang L, He T. Cardiovascular implications of fatal outcomes of patients with Coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5(7):811–818. doi: 10.1001/jamacardio.2020.1017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shi S, Qin M, Shen B, Cai Y, Liu T, Yang F. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan, China. JAMA Cardiol. 2020;5(7):802–810. doi: 10.1001/jamacardio.2020.0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ramamurthy S, Talwar KK, Goswami KC, Shrivastava S, Chopra P, Broor S. Clinical profile of biopsy proven idiopathic myocarditis. Int J Cardiol. 1993;41(3):225–232. doi: 10.1016/0167-5273(93)90119-2. [DOI] [PubMed] [Google Scholar]
- 25.Ukena C, Mahfoud F, Kindermann I, Kandolf R, Kindermann M, Bohm M. Prognostic electrocardiographic parameters in patients with suspected myocarditis. Eur J Heart Fail. 2011;13(4):398–405. doi: 10.1093/eurjhf/hfq229. [DOI] [PubMed] [Google Scholar]
- 26.Hung Y, Lin WH, Lin CS, Cheng SM, Tsai TN, Yang SP. The prognostic role of QTc interval in acute myocarditis. Acta Cardiol Sin. 2016;32(2):223–230. doi: 10.6515/ACS20150226A. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lazzerini PE, Capecchi PL, Laghi-Pasini F. Long QT syndrome: an emerging role for inflammation and immunity. Front Cardiovasc Med. 2015;2:26. doi: 10.3389/fcvm.2015.00026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lab MJ. Contraction-excitation feedback in myocardium. Physiological basis and clinical relevance. Circ Res. 1982;50(6):757–766. doi: 10.1161/01.res.50.6.757. [DOI] [PubMed] [Google Scholar]
- 29.Jain S, Workman V, Ganeshan R, Obasare ER, Burr A, DeBiasi RM. Enhanced electrocardiographic monitoring of patients with Coronavirus Disease 2019. Heart Rhythm. 2020;17(9):1417–1422. doi: 10.1016/j.hrthm.2020.04.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Saleh M, Gabriels J, Chang D, Soo Kim B, Mansoor A, Mahmood E. Effect of Chloroquine, Hydroxychloroquine, and Azithromycin on the corrected QT interval in patients with SARS-CoV-2 infection. Circ Arrhythm Electrophysiol. 2020;13(6) doi: 10.1161/CIRCEP.120.008662. [DOI] [PMC free article] [PubMed] [Google Scholar]