Abstract
Background
Coronavirus disease (COVID-19) is a systemic illness characterized by raging impact of cytokine storm on multiple organs. This may trigger malignant ventricular arrhythmias and unmask a clinically silent cardiomyopathy.
Case summary
A 57-year-old gentleman, known case of hyperthyroidism and diabetes, was referred to our emergency department with history of two ventricular tachycardia (VT) episodes requiring direct current cardioversion in last 3 h followed by another episode in our emergency department that was cardioverted. There was no past history of cardiac illness. His 12-lead electrocardiogram (during sinus rhythm) along with screening echocardiography suggested Arrhythmogenic right ventricular cardiomyopathy (ARVC). He was coincidentally found to be COVID-19 positive by reverse transcription-polymerase chain reaction (RT-PCR) as part of our routine screening. However, he had no fever or respiratory complaints. We noted raised systemic inflammatory markers and cardiac troponin T which progressively increased over the next 4 weeks paralleled by an increase in ventricular premature contraction burden and thereafter started decreasing and returned to baseline by 6th week when the patient became COVID-19 negative by RT-PCR. Subsequently, a single-chamber automated implantable cardioverter-defibrillator implantation was done following which there was a transient increase in these biomarkers that subsided spontaneously. The patient is asymptomatic during 6 weeks of follow-up.
Discussion
COVID-19-associated cytokine surge triggering VT storm and unmasking a clinically silent ARVC has not yet been reported. The case highlights a life-threatening presentation of COVID-19 and indicates a probable link between inflammation and arrhythmogenicity.
Keywords: COVID-19, ARVC, VT storm, Cytokine surge, AICD, Case report
Learning points
Atypical non-respiratory manifestations of coronavirus disease like ventricular tachycardia should initiate a search for underlying proarrhythmic substrate like cardiomyopathy.
Ventricular arrhythmia burden parallels the systemic inflammatory response, pointing towards a probable role of inflammatory cytokines like interleukin-6 in arrhythmogenesis.
Introduction
The manifestations of coronavirus disease (COVID-19) range from asymptomatic infection to multi-organ failure and death. Among these, COVID-19-related myocarditis and arrhythmias are hypothesized to result from direct viral myocardial injury and/or inflammatory cytokine-induced damage.1–4 We report a patient presenting with ventricular tachycardia (VT) storm induced by COVID-19 infection that unmasked an underlying clinically silent genetic cardiomyopathy.
Timeline
| 3 h before admission | Two episodes of sustained monomorphic ventricular tachycardia (VT) requiring direct current (DC) cardioversion in a peripheral centre. Patient referred on amiodarone infusion for further management |
| In emergency |
One episode of sustained monomorphic VT required DC cardioversion. Patient continued on intravenous amiodarone and oral beta-blocker also started Twelve-lead electrocardiogram in sinus rhythm showed features suggestive of arrhythmogenic right ventricular cardiomyopathy including epsilon waves |
| Reverse transcription-polymerase chain reaction (RT-PCR) for coronavirus disease (COVID-19) from nasopharyngeal swab turned out positive. Levels of inflammatory cytokine interleukin-6, erythrocyte sedimentation rate, C-reactive protein, B-type natriuretic peptide, and high-sensitivity cardiac troponin T (hs-cTnT) were elevated | |
| 1–4 weeks | Weekly RT-PCR tests were positive for COVID-19 associated with progressive increase in inflammatory biomarkers and 24 h ventricular premature contraction (VPC) burden assessed by serial Holter monitoring including couplets and episodes of non-sustained VT (NSVT) |
| 5–6 weeks |
Declining trend noted in inflammatory markers and 24 h VPC burden with no episodes of NSVT RT-PCR for COVID repeated twice at 48 h interval were negative during 6th week Single-chamber transvenous AICD (automated implantable cardioverter-defibrillator) implantation done |
| Week 7 |
Post-AICD implantation, relapse of cytokine surge along with increase in 24 h VPC burden assessed by Holter Any infectious cause ruled out |
| Week 8 | Inflammatory mediators spontaneously declined to normal and associated 24 h VPC burden became insignificant and patient discharged |
| 6 weeks after discharge | Clinically asymptomatic with no recurrence of VT requiring AICD shock |
Case presentation
A 57-year-old gentleman presented to a peripheral centre with sudden onset palpitations associated with dizziness and profound sweating. Two episodes of VT were documented and needed direct current (DC) cardioversion due to haemodynamic instability. After 3 h, he was referred to our emergency on intravenous (IV) amiodarone infusion at 1 mg/min (after initial loading dose of 150 mg).
He had history of Type 2 diabetes mellitus and Grave’s disease for last 10 years (on oral hypoglycaemic drugs and tablet carbimazole 10 mg thrice daily). He had no past history suggestive of cardiac illness but the family history was significant for sudden cardiac death (SCD) in two siblings at ages of 55 and 59 years. However, the patient had not undergone any work-up for familial SCD.
At presentation, he had a pulse rate of 60/min and BP of 120/84 mmHg with no other significant clinical findings. In our emergency, he had another episode of monomorphic VT (with haemodynamic compromise) that was DC cardioverted.
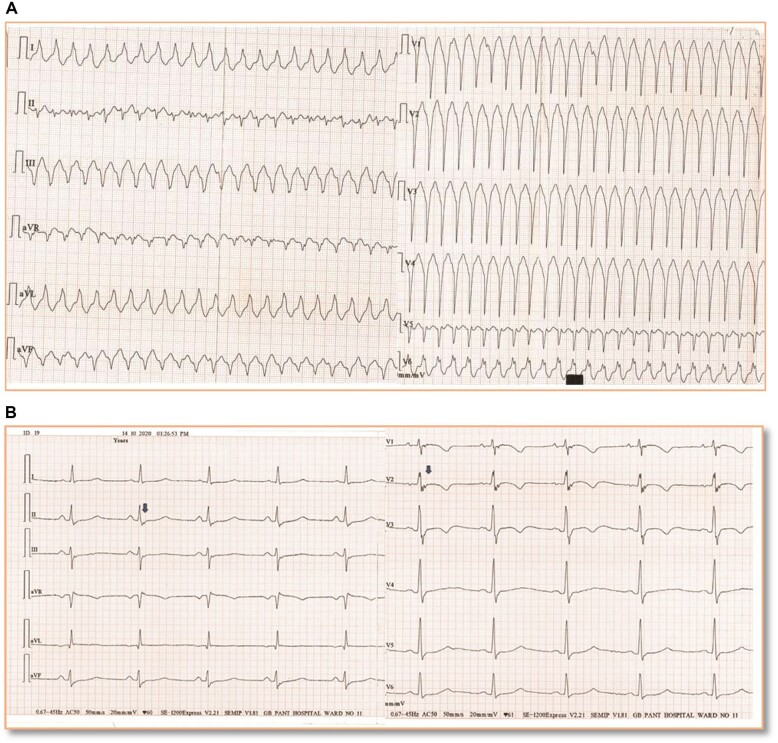
The 12-lead electrocardiogram (ECG) during VT showed ventricular rate of 190 beats/min with left bundle branch block pattern, left axis deviation, and QRS transition in lead V6 with notching in QRS complexes, all suggestive of right ventricle (RV) free wall origin (Figure 1A).5 This was similar in morphology to the ECGs recorded in the peripheral centre. ECG during sinus rhythm showed epsilon waves in leads V1–V3 and inferior limb leads. Leads V1–V3 also showed T-wave inversion along with terminal activation duration of 60 ms (Figure 1B). These findings favoured a diagnosis of arrhythmogenic right ventricular cardiomyopathy (ARVC).5
Figure 1.
(A) Twelve-lead electrocardiogram (at 25 mm/s speed) recording of monomorphic ventricular tachycardia episode recorded at time of presentation to emergency—left axis deviation, QRS duration in lead I >120 ms, QRS transition in V6, notching of QRS in V6 favour a diagnosis ventricular tachycardia of right ventricle origin. (B) Electrocardiogram in sinus rhythm after opening the 40 Hz filter at double amplification and 50 mm/s speed recording. Epsilon waves in leads V1–V3 (blue arrows) with T-wave inversion and terminal activation duration of 60 ms. Epsilon waves are also noted in leads II, III, avF (blue arrows) that suggests concomitant left ventricular involvement.
His blood sugar, serum electrolytes, and thyroid profile were normal. High-sensitivity cardiac-specific troponin T (Elecsys Troponin-T-hs, Cobas®, Roche diagnostics) (hs-cTnT) was mildly raised (Table 1). Patient was reloaded with IV amiodarone 150 mg for breakthrough VT and continued the infusion at 1 mg/min for a total of 6 h followed by 0.5 mg/min for 48 h. After 24 h, the IV infusion was overlapped with oral amiodarone (200 mg tablet thrice daily for 10 days followed by 200 mg twice daily for 5 days, making a cumulative dose of 10.1 g) followed by 200 mg once daily thereafter as per recommendations.6 He was also started on oral carvedilol (50 mg) daily.
Table 1.
Trend of body temperature, various inflammatory, and cardiac biomarkers over the course of hospital stay and their relation to ventricular premature contraction burden
| Week 1 | Week 2 | Week 3 | Week 4 | Week 5 | Week 6 | Week 7 | Week 8 | |
|---|---|---|---|---|---|---|---|---|
| Temperature (°F) | 97.5 | 97.9 | 98.1 | 97.9 | 98.5 | 98.1 | 98.2 | 97.9 |
| ESR (mm/h) | 50 | 52 | 100 | 80 | 40 | 14 | 10 | 21 |
| CRP (mg/L) | 45 | 136 | 37 | 8.25 | 4.3 | 0.52 | 107.18 | 4.45 |
| D-dimer (μg/mL) | 2.93 | 4.3 | 4.9 | 5.55 | 2.93 | 0.27 | 1.6 | 0.23 |
| NLR | 5.2 | 6.3 | 6.6 | 7.55 | 3 | 2.38 | 2.75 | 2.50 |
| IL-6 (pg/mL) | 77.29 | 110 | 180 | 385.4 | 104.1 | 3.9 | 500.1 | 8.23 |
| hs-cTnT (pg/mL) | 20.1 | 41.2 | 51 | 65.5 | 30.1 | 3.5 | 12.1 | 3.9 |
| BNP (pg/mL) | 112 | NA | 140 | NA | 26.4 | NA | 25 | NA |
| VE/h | 11 | 16 | 18 | 51 | 16 | 1 | 86 | 1 |
| Ventricular couplets/24 h | 1 | 3 | 6 | 20 | 0 | 0 | 87 | 0 |
| Ct value | 24.34 | 24.20 | 24.17 | 25.5 |
ICD implantation. Upper limit of normal for the above biomarkers are ESR: 20 mm/h, CRP: <5 mg/L, d-Dimer: 0.5 μg/mL, NLR: 3, IL-6: 5 pg/mL, hs-cTnT: 14 pg/mL, BNP: 100 pg/mL.
Temperature recordings are average of weekly recordings done at 8 a.m. and 8 p.m. everyday.
BNP, B-type natriuretic peptide; CRP, C-reactive protein; Ct, cycle threshold; ESR, erythrocyte sedimentation rate; hs-cTnT, high-sensitivity cardiac troponin T; IL-6, interleukin-6; NA, not available as test was done once in 2 weeks; NLR, neutrophil to lymphocyte ratio; VE/h, ventricular ectopics per hour as recorded on Holter.
Echocardiography showed dilated RV and RV outflow tract (RVOT) (diameter—34.7 mm), RV fractional area change of 20.3%, and RV global strain (free wall + septum) of −14.8%. The left ventricular (LV) dimensions and function were normal. Patient met all major non-histological modified task force criteria for diagnosis of ARVC.7 Automated implantable cardioverter-defibrillator (AICD) for secondary prevention was planned as per European guidelines.8
As part of protocol in our institute to assess COVID-19 status of all emergency patients warranting admission, reverse transcription-polymerase chain reaction (RT-PCR) assay for COVID-19 on nasopharyngeal swab was done which turned out positive with a cycle threshold (Ct) value of 25.4. His laboratory investigations showed high erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), raised neutrophil to lymphocyte ratio (NLR), interleukin 6 (IL-6), D-dimer, and B-Type natriuretic peptide (BNP) (Table 1).
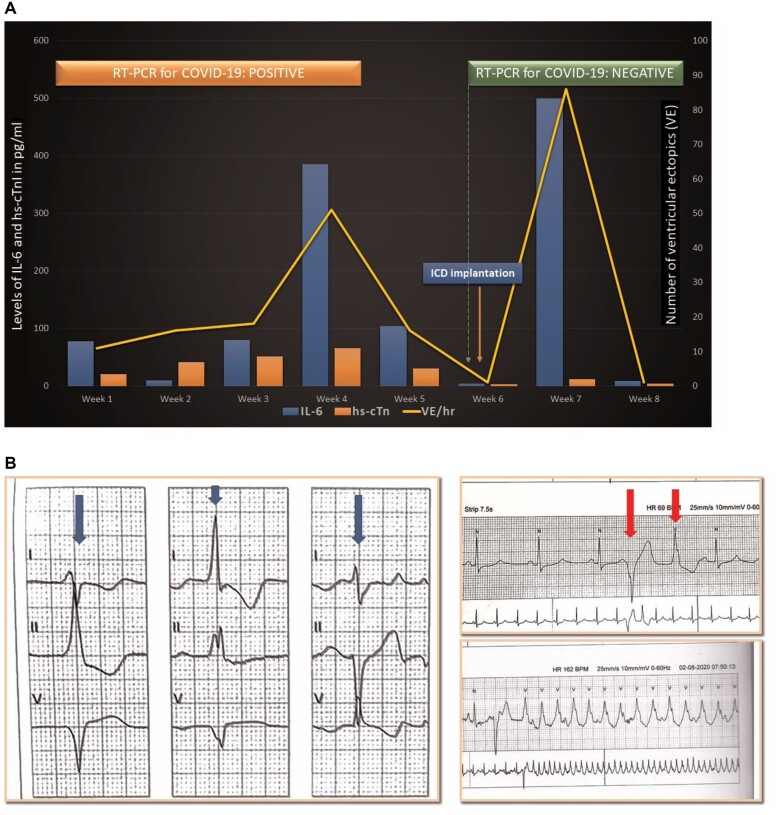
We monitored his inflammatory markers, D-dimer, and NLR along with hs-cTnT twice weekly and BNP every 2 weeks. These parameters showed a surge during the first 4 weeks (Table 1 and Figure 2A). Holter monitoring (for 24–72 h) and RT-PCR for COVID-19 were also done at weekly intervals. RV origin multi-morphic ventricular premature contractions (VPCs) along with couplets and non-sustained VT episodes were noted (Figure 2B) with highest burden during the 4th week (Figure 2A). The RT-PCR was also persistently positive for COVID-19 till 4th week. The Ct value for first 3 weeks suggested increasing viral RNA load (Ct value is inversely proportional to the viral RNA load)9 followed by a decline starting from 4th week (Table 1). No QT prolonging antiviral drug like hydroxychloroquine or azithromycin was given to the patient. At the end of 4th week, the thyroid profile reassessed was within normal limits and amiodarone 200 mg once daily was continued.
Figure 2.
(A) Trend of various inflammatory markers and high-sensitive cardiac troponin T along with ventricular premature contraction burden during the course of hospital stay. y-axis: levels of IL-6 and high-sensitivity cardiac troponin T, x-axis: time since hospitalization, secondary vertical axis: ventricular premature contraction burden. (B) Holter recordings show multiple morphologies of ventricular premature contractions recorded (bold blue arrows in left panel), a multi-morphic ventricular premature contraction couplet (top right panel, red bold arrows) and a run of non-sustained ventricular tachycardia (bottom right panel). COVID-19, coronavirus disease; hs-cTnT, high-sensitivity cardiac troponin T; IL-6, interleukin 6; RT-PCR, reverse transcription-polymerase chain reaction; VE/h: ventricular ectopics per hour as recorded on Holter.
From 5th week onwards, the inflammatory markers started subsiding, mirrored by a similar decrease in the levels of hs-cTnT, BNP, and 24 h VPC burden (Table 1 and Figure 2A). RT-PCR for COVID-19 became negative during the 6th week (assessed twice at 48 h interval). By this time, the inflammatory markers, BNP, and hs-cTnT had also returned to normal range and the VPC burden had become insignificant (Table 1 and Figure 2A). No respiratory symptoms or fever were noted during these 6 weeks.
Following this, cardiac magnetic resonance imaging (MRI) was done, which revealed grossly dilated RV [end-diastolic volume = 114.3 mL/m2 (ULN 110 mL/m2)] with global hypokinesia, reduced RV ejection fraction (20.5%), and dilated RVOT (3.6 cm). Prominent trabeculations in RV apex along with focal outpouchings in RV free wall were also noted (Figure 3A and Videos 1 and 2). The RV mid-cavity free wall, inferior, and apical segment showed late gadolinium enhancement (LGE) suggestive of myocardial fibrosis. The LV basal and mid-cavity (anterior and inferior septal segments) along with mid-cavity (inferior and inferolateral segments) showed T2W hyperintensity due to myocardial oedema which colocalized with selective subepicardial LGE suggestive of myocarditis in evolution with replacement fibrosis (Figure 3B).
Figure 3.
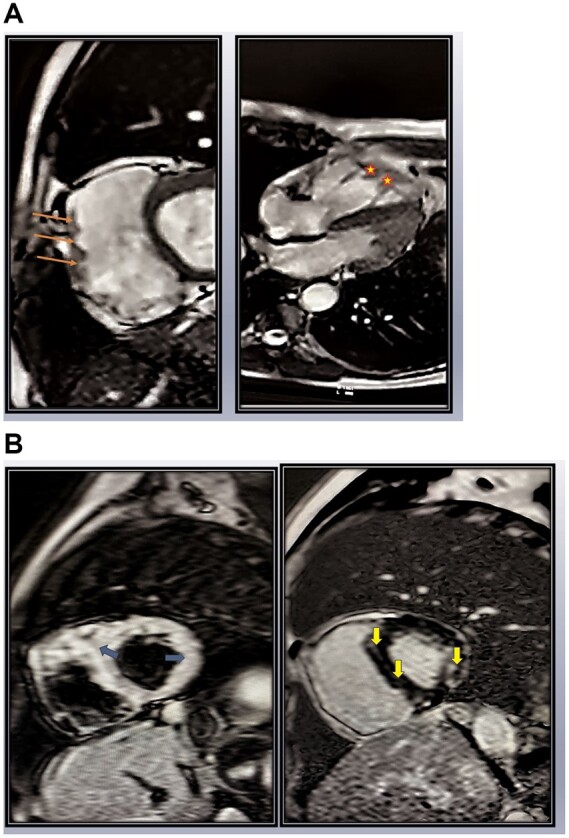
(A) Cardiac magnetic resonance imaging: left panel: short-axis view showing dilated right ventricle with focal outpouchings (orange arrows) in free wall and right panel: four-chamber view showing trabeculations and serrated margins of right ventricle apex (marked as stars). (B) Cardiac magnetic resonance imaging: left panel: short-axis T2w image showing hyperintensity (blue arrows) in segments of left ventricular and interventricular septum suggestive of myocardial oedema. In right panel, the hyperintense segments shown in left panel colocalize with selective subepicardial late gadolinium enhancement (yellow arrows), suggestive of myocarditis in evolution with fibrosis.
After the systemic hyperinflammatory phase had subsided and the patient became COVID negative, a single-chamber AICD was implanted (Fortify Assura™ ICD, St. Jude Medical, Minnesota, USA) at the end of 6th week. Amiodarone, carvedilol, carbimazole, and oral hypoglycaemic drugs were continued in the post-intervention phase along with broad-spectrum IV antibiotics for 72 h.
A transient surge of inflammatory markers and hs-cTnT along with the VPC burden was noted in the post-operative phase (Table 1 and Figure 2A). However, the patient’s total leucocyte count and serum procalcitonin levels were normal together with sterile blood and urine cultures, ruling out any infectious aetiology. All the deranged parameters spontaneously returned to baseline within 1 week. He was discharged thereafter on tablet amiodarone 200 mg once daily, carvedilol 25 mg twice daily, and ramipril 5 mg once daily as per guidelines8 along with anti-thyroid and anti-diabetic drugs. At follow-up of 6 weeks, the patient is asymptomatic and euthyroid without any recurrence of symptoms or AICD interventions.
Discussion
COVID-19 infection primarily causes respiratory symptoms but presentations with malignant ventricular arrhythmias without concomitant respiratory symptoms have been reported, associated with underlying myocarditis or coronary artery disease.10,11 However, to the best of our knowledge VT storm (defined as ≥3 sustained VT episodes in 24 h) due to COVID-19 induced myocarditis that unmasked an underlying clinically silent cardiomyopathy (ARVC) has not yet been reported.
The present case, although representing a rare form of cardiomyopathy, provides insights into the plausible mechanisms of COVID-19-induced malignant ventricular arrhythmias. The patient had never experienced any cardiac symptoms but the cytokine storm following COVID-19 infection unveiled the dormant electrical instability.
IL-6 plays a central role in COVID-19-induced acute myocardial injury.1,4 In our case, persistent viral infection during first 4 weeks had triggered IL-6 surge through immunological activation that led to myocardial injury. There was parallel rise in levels of IL-6, hs-cTnT, and VPC burden (representing severity of acute myocardial injury) during the initial 4 weeks of active infection followed by their decline and normalization by 6th week when the patient turned COVID-19 RT-PCR negative. Myocarditis can manifest solely with symptoms of a new-onset arrhythmia10 without associated symptoms of fever, chest pain, dyspnoea, or fatigability. In our patient, myocarditis had manifested (i) clinically with the sole presentation of a new-onset ventricular arrhythmia associated with (ii) raised level of the cardiac injury biomarker hs-cTnT along with (iii) biomarker evidence of a hyperinflammatory state (raised CRP, ESR, and IL-6) during the 4 weeks of active viral infection together with (iv) MRI evidence of acute myocarditis in the form of myocardial oedema which colocalized with selective subepicardial LGE fulfilling revised Lake-Louis consensus criteria for diagnosing myocarditis.12
It has been reported that IL-6 causes internalization of the desmosomal protein plakoglobin from the cardiomyocyte membrane and promotes electrical instability.13 The reduced surface expression of such desmosomal proteins is also a known cause of arrhythmogenic cardiomyopathies like ARVC,14 thereby partly explaining the concurrent surge of IL-6 levels and VPC burden in our patient.
Cardiac injury markers and systemic inflammatory markers have been reported to rise transiently after mechanical myocardial injury following cardiac device implantation.15 We had performed AICD implantation once the cytokine levels had normalized and the patient was free of active COVID infection. However, the inflammatory markers and VPC burden showed a significant resurge following the procedure. It shows that irrespective of the cause, a rise in IL-6 (viral infection or post-implantation inflammatory stress) promoted electrical instability in our patient with ARVC.
The association between systemic inflammation and ventricular arrhythmias is well-known. Increased IL-6 levels in hyperinflammatory states like myo/endocarditis shows correlation with QT prolongation and subsequent ventricular arrhythmias.16,17 Similarly, an independent correlation between CRP levels and risk of SCD has been reported.18
A wide variety of bradyarrhythmias and supraventricular arrhythmias have also been described with myocarditis. Myocardial and conduction system involvement in infective diseases like diphtheria and autoimmune diseases like sarcoidosis can manifest as varying degrees of heart block.8 Following cardiac surgery, atrial fibrillation, induced by post-surgical inflammation and oxidative stress is a well-known complication.19
The use of amiodarone in our patient of Grave’s disease needs close monitoring. Type 1 amiodarone induced thyrotoxicosis can develop in those with underlying nodular goitre or Grave’s disease as an early (within 12 weeks) or late complication. Our patient after 14 weeks of amiodarone use is still euthyroid. As per guidelines, we plan to assess his thyroid function at six monthly intervals.20
Conclusion
COVID-19 triggered cytokine surge can unmask a dormant cardiomyopathy and manifest solely as malignant ventricular arrhythmias like VT storm in absence of any associated respiratory symptoms.
Lead author biography

Dr Saibal Mukhopadhyay completed his post-graduation in Cardiology in 2002 from the University of Delhi (India). He joined the Department of Cardiology in Gobind Ballabh Pant Hospital as senior research associate and then as faculty in the year 2005. He is currently serving as the Head of Department in this institute. Several of his research articles in the field of cardiology have been published in national and international journals.
Supplementary material
Supplementary material is available at European Heart Journal - Case Reports online.
Slide sets: A fully edited slide set detailing this case and suitable for local presentation is available online as Supplementary data.
Consent: The authors confirm that written consent for submission and publication of this case report including images and associated text has been obtained from the patient in line with COPE guidance.
Conflict of interest: None declared.
Funding: None declared.
Supplementary Material
References
- 1. Siripanthong B, Nazarian S, Muser D, Deo R, Santangeli P, Khanji MY. et al. Recognizing COVID-19-related myocarditis: the possible pathophysiology and proposed guideline for diagnosis and management. Heart Rhythm 2020;17:1463–1471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Turagam MK, Musikantow D, Goldman ME, Bassily-Marcus A, Chu E, Shivamurthy P. et al. Malignant arrhythmias in patients with COVID-19: incidence, mechanisms, and outcomes. Circ Arrhythm Electrophysiol 2020;13:e008920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Bhatla A, Michael M, Adusumalli S, Matthew C, Oh E, Tierney A. et al. COVID-19 and cardiac arrhythmias. Heart Rhythm 2020;17:1439–1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Lazzerini PE, Boutjdir M, Capecchi PL.. COVID-19, arrhythmic risk, and inflammation: mind the gap! Circulation 2020;142:7–9. [DOI] [PubMed] [Google Scholar]
- 5. Gandjbakhch E, Redheuil A, Pousset F, Charron P, Frank R.. The present and future clinical diagnosis, imaging, and genetics of arrhythmogenic right ventricular cardiomyopathy/dysplasia: JACC state-of-the-art review. J Am Coll Cardiol 2018;72:784–804. [DOI] [PubMed] [Google Scholar]
- 6. Muser D, Santangeli P, Liang JJ.. Management of ventricular tachycardia storm in patients with structural heart disease. World J Cardiol 2017;9:521–530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Arrhythmogenic right ventricular cardiomyopathy: current diagnostic criteria - American College of Cardiology [Internet]. https://www.acc.org/latest-in-cardiology/ten-points-to-remember/2019/10/29/11/37/arrhythmogenic-right-ventricular-cardiomyopathy (24 October 2020).
- 8. Priori SG, Blomström-Lundqvist C, Mazzanti A, Bloma N, Borggrefe M, Camm J. et al. 2015 ESC Guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: the task force for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death of the European Society of Cardiology (ESC). Europace 2015;17:1601–1687. [DOI] [PubMed] [Google Scholar]
- 9. Chang MC, Hur J, Park D.. Interpreting the COVID-19 test results: a guide for physiatrists. Am J Phys Med Rehabil 2020;99:583–585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Elsaid O, McCullough PA, Tecson KM, Williams RS, Yoon A.. Ventricular fibrillation storm in coronavirus 2019. Am J Cardiol 2020;135:177–180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Mitacchione G, Schiavone M, Gasperetti A, Forleo GB.. Ventricular tachycardia storm management in a COVID-19 patient: a case report. Eur Hear J Case Rep 2020;4:1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Friedrich MG, Sechtem U, Schulz-Menger J, Holmvang G, Alakija P, Cooper LT. et al. ; International Consensus Group on Cardiovascular Magnetic Resonance in Myocarditis. Cardiovascular magnetic resonance in myocarditis: a JACC white paper. J Am Coll Cardiol 2009;53:1475–1487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Asimaki A, Tandri H, Duffy ER, Winterfield JR, MacKey-Bojack S, Picken MM. et al. Altered desmosomal proteins in granulomatous myocarditis and potential pathogenic links to arrhythmogenic right ventricular cardiomyopathy. Circ Arrhythm Electrophysiol 2011;4:743–752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Gemayel C, Pelliccia A, Thompson PD.. Arrhythmogenic right ventricular cardiomyopathy. J Am Coll Cardiol 2001;38:1773–1781. [DOI] [PubMed] [Google Scholar]
- 15. Varvarousis D, Goulas N, Polytarchou K, Psychari SN, Paravolidakis K, Konstantinidou A. et al. Biomarkers of myocardial injury and inflammation after permanent pacemaker implantation: the lead fixation type effect. J Atr Fibrillation 2018;10:1–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Alí A, Boutjdir M, Aromolaran AS.. Cardiolipotoxicity, inflammation, and arrhythmias: role for interleukin-6 molecular mechanisms. Front Physiol 2019;9:1866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lazzerini PE, Capecchi PL, El-Sherif N, Laghi-Pasini F, Boutjdir M.. Emerging arrhythmic risk of autoimmune and inflammatory cardiac channelopathies. J Am Heart Assoc 2018;7:e010595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Albert CM, Ma J, Rifai N, Stampfer MJ, Ridker PM.. Prospective study of C-reactive protein, homocysteine, and plasma lipid levels as predictors of sudden cardiac death. Circulation 2002;105:2595–2599. [DOI] [PubMed] [Google Scholar]
- 19. Reckman YJ, Creemers EE.. Circulating circles predict postoperative atrial fibrillation. J Am Heart Assoc 2018;7:e8261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Bartalena L, Bogazzi F, Chiovato L, Hubalewska-Dydejczyk A, Links TP, Vanderpump M.. 2018 European Thyroid Association (ETA) guidelines for the management of amiodarone-associated thyroid dysfunction. Eur Thyroid J 2018;7:55–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.