Abstract
Chronic infection with hepatitis B virus (HBV) remains a global health concern despite the availability of vaccines. To date, the development of effective treatments has been severely hampered by the lack of reliable, reproducible, and scalable in vitro modeling systems that precisely recapitulate the virus life cycle and represent virus-host interactions. With the progressive understanding of liver organogenesis mechanisms, the development of human induced pluripotent stem cell (iPSC)-derived hepatic sources and stromal cellular compositions provides novel strategies for personalized modeling and treatment of liver disease. Further, advancements in three-dimensional culture of self-organized liver-like organoids considerably promote in vitro modeling of intact human liver tissue, in terms of both hepatic function and other physiological characteristics. Combined with our experiences in the investigation of HBV infections using liver organoids, we have summarized the advances in modeling reported thus far and discussed the limitations and ongoing challenges in the application of liver organoids, particularly those with multi-cellular components derived from human iPSCs. This review provides general guidelines for establishing clinical-grade iPSC-derived multi-cellular organoids in modeling personalized hepatitis virus infection and other liver diseases, as well as drug testing and transplantation therapy.
Keywords: Hepatitis B virus, Induced pluripotent stem cells, Liver organoid, Multi-cellular organoid, Modeling, Transplantable
Core Tip: The development of an effective treatment for hepatitis B virus relies on reliable and reproducible in vitro modeling systems. Recently, three-dimensional multi-cellular organoid systems have attracted considerable attention owing to their superior susceptibility and capability to precisely recapitulate the virus life cycle. Recent advances in organoid-generating strategies, particularly those derived from human induced pluripotent stem cells, together with future improvements in genetic modification and scalability, will undoubtedly promote personalized disease modeling and drug development.
INTRODUCTION
Hepatitis B virus (HBV), a highly prevalent global health concern, is among the most common causes of advanced chronic liver disease development. Globally, 292 million people (approximately one-third patients in China) reportedly present with chronic HBV infection[1], which is associated with a wide range of clinical manifestations, including liver cirrhosis, liver failure, and hepatocellular carcinoma[2]. Despite the availability of approved vaccines, currently, HBV treatment is mainly restricted to interferon (IFN) and nucleoside analogs, which rarely achieve absolute hepatitis elimination[3]. Notably, clinical treatment usually results in different responses and outcomes due to viral genotype diversity and patient genetic susceptibility, which further increases treatment difficulties[4]. To date, in vitro culture systems have been utilized to determine the characteristics and life cycle of hepatitis C virus. Furthermore, these systems have been proven to substantially contribute to our understanding of the genomic replication subsystems that ultimately led to productive viral infection, as well as successful identification of small molecules having effective activity against various hepatitis C viral enzymes[5]. Therefore, to better understand the HBV life cycle and to promote drug development, a robust personalized in vitro HBV modeling platform is urgently needed to recapitulate the entire HBV replication cycle, especially that of covalently closed circular DNA (cccDNA) infection and formation. Several in vitro modeling systems have been developed, mostly based on in vitro culture systems using primary hepatocytes or hepatic cells from other sources[6-9]. Besides, sodium taurocholate cotransporting polypeptide (NTCP)-transformed hepatocellular carcinoma cell lines are also available. No one system fits all studies, but findings from different systems may be complementary. Maintenance of hepatic function and increase of susceptibility to HBV infection in vitro are the principal focus for most established systems. In this regard, numerous culture systems have also been developed to better fit the in vivo microenvironment.
Considering the demand for clinical applications, the reproducibility and scalability of modeling systems have generated interest in recent years. Accordingly, as a theoretically unlimited source of stem cells without ethical implications, pluripotent stem cells [induced pluripotent stem cell (iPSC)] represent the most promising source that can be used to yield substantial quantities of homogeneous and reproducible cellular components of the liver. Moreover, with a patient-specific genetic background, iPSC-derived hepatocyte-like cells (HLCs) are optimal for individualized disease modeling, which may remarkably benefit mechanistic studies and drug development for hereditary diseases and diseases closely related to the host genetic background, such as HBV infection[10-12]. With considerable expression of major mature hepatocyte markers, as well as host factors required for HBV infection, including the NTCP, iPSC-HLCs could support robust production of HBV particles and viral RNAs[7,13]. More importantly, significant inhibition of HBV infection was detected upon anti-HBV agent treatment, suggesting that iPSC-HLCs could be utilized as a novel HBV infection model for drug testing[14].
However, the drawbacks in maturation and difficulty in maintenance of iPSC-HLCs have raised considerable concerns. Liver organoids (LOs) can be maintained for a longer time, are more susceptible to HBV infection, and exhibit enhanced liver function[3]. Multi-cellular three-dimensional (3D) organoid culture systems have recently become an effective strategy to compensate the deficiencies and also extend the potential modeling duration. With supportive mesenchymal and endothelial cells in the 3D microenvironment, HBV susceptibility was greatly enhanced compared to monolayer culture system, while the duration for HBV propagation and virus production was also prolonged[15]. HBV-infected LOs could serve as a reliable and viable ex vivo infection model for hepatitis research to investigate the role of host genetic background in HBV infection and individual prognosis of infection, enabling personalized hepatitis treatment[15]. To further enhance the reproducibility and scalability, an all-iPSC-based strategy has been adopted most recently in generating multi-cellular LOs with improved hepatic functions almost equivalent to those of adult hepatocytes, although their advantages in HBV modeling remains to be confirmed in future studies[16]. With the development of 3D expansion strategies, together with advances in direct programming or induction of liver fate, novel engineered LOs are expected to be established on a large scale for broad applications in disease modeling, drug screening, and transplantation.
Here, we summarized the recent advances in in vitro modeling systems for HBV infection, especially using multi-cellular LOs. Additionally, we discussed the current and future challenges in the application of advanced organoid generation platforms in terms of efficiency, reproducibility, and scalability. Finally, we explored the future applications of multi-cellular LOs in personalized and precise treatments.
ADVANCES AND LIMITATIONS IN MODELING HEPATITIS B INFECTION
Cell sources
HBV only efficiently infects fully differentiated hepatic cells. Until now, primary human hepatocytes (PHHs) were the only permissive agents used for the study of HBV infection in vitro[17]. This system remains the gold standard and is utilized to model HBV infections in vitro[18]. However, fresh PHHs have markedly limited availability and unpredictable variability among donors. Furthermore, PHHs have extremely limited replication ability in conventional culture systems and cannot be easily subjected to genetic manipulation, which hinders scaled-up manufacturing and targeted gene therapy. Moreover, PHHs show rapid loss of their mature hepatic functions and infection susceptibility after plating. Thus, these cells are unsuitable for utilization as a stable source for long-term modeling and testing[19]. For a reliable and practical platform for long-term disease modeling and drug development, a new gold standard should be established.
To overcome restrictions in the use of PHHs, especially source availability and cost, researchers have shifted their attention to immortalized tumor-derived or transformed hepatocytes and hepatocellular carcinoma cell lines, such as Huh7, HepG2, HepG2.2.15, HepAD38, HepaRG, HepDE19, HepBHAe82, and HepG2-NTCP cells[3,20,21]. HepG2.2.15 cells can stably express HBV viral gene products. However, HBV viral particles are derived from chromosomally integrated DNA rather than cccDNA during in vivo processing. Later, a highly HBV-permissive cell clone of HepAD38 cells was created, which could produce more robust viruses. However, this cell line failed to recapitulate the complete viral replication process[20,21]. In this regard, the liver progenitor cell line HepaRG is a more attractive source for modeling, since these cells show morphological and functional features similar to those of primary hepatocytes. More importantly, HepaRG cells are susceptible to HBV infection upon supplementation with corticoids and DMSO, which maintains the cellular maturation state[6]. However, this infection system has a relatively low infection efficiency, even under the strict culture conditions necessary to prevent dedifferentiation. Additionally, cccDNA amplification did not occur in this system[7]. Subsequently, to meet the requirements for high-throughput screening of cccDNA-targeting drugs, a HepDE19 cell line was developed in which the expression of HBV e antigen (HBeAg) was derived from cccDNA. However, the ELISA antigen shows cross-reactivity with viral HBeAg homologues, which dramatically diminishes the assay specificity[22,23]. To address this problem, the HepBHAe82 cccDNA reporter cell line was developed based on similar principles. Importantly, this line produced high levels of cccDNA-dependent HBeAg with high, specific readout signals and did not disrupt any cis-elements that were essential for HBV replication and HBeAg secretion[21]. As a potential tool for cccDNA-targeted drug screening and testing, it remains to be determined whether cccDNA formation, stability, and transcription are recapitulated in this system. To further achieve high susceptibility to HBV infection, NTCP, an essential receptor for HBV infection[24] was introduced into liver cancer cell lines. Establishment of susceptible cell lines, such as HepG2-NTCP, permitted highly efficient HBV infection and enabled the identification of key events or processes in the viral life cycle, although this system could not be used to elucidate the entire HBV life cycle[25]. Furthermore, the physiological and functional characteristics of tumor-derived or transformed hepatoma cell lines are distinct from those of normal hepatocytes. Comparing the HepG2 transcriptome to cells obtained from liver tissue biopsies showed that most genes with upregulated expression in HepG2 cells were associated with carcinogenesis, while those with upregulated expression in the liver were enriched in heterologous biometabolism[26], thereby implying a fundamental deficiency of hepatocellular carcinoma cell lines for reliable and precise modeling. Besides, a non-hepatic cell line was recently developed for HBV modeling. Constructed by exogenous expression of human NTCP, HNF4α, RXRα, and PPARα, the 293T cell line (293T-NE-3NR) was able to support HBV entry, transcription, and replication, although the HBV production (HBV DNA, cccDNA, and pgRNA) in 293T-NE-3NR remained lower than that in the HepG2 cell line. To date, the use of a non-hepatic model is quite a new concept; however, it may serve as a beneficial complement to the current hepatocellular carcinoma cell models. Without intrinsic liver-related host factors, an exogenous construction strategy may help discover the key factors involved in HBV infection[27].
To achieve functional hepatocytes that closely resemble primary hepatocytes, HLCs differentiated from pluripotent stem cells have attracted considerable attention as a novel cell source. Since the use of embryonic stem cells is markedly hindered due to ethical issues, iPSCs induced by the ectopic expression of defined transcription factors (e.g., Oct4, Sox2, Klf4, and c-Myc) in patient fibroblasts or other somatic cells have emerged as the most promising source for generating HLCs[28-30]. After conduction of stepwise hepatic differentiation[31-33], iPSC-derived HLCs could support effective HBV infection and last for a long time[14,34]. Together with their infinite expansion capability, iPSCs have enabled the provision of numerous hepatic cell sources necessary for stable and scalable modeling with less variability than PHHs[25]. Meanwhile, the epigenetic effects in human iPSCs are minimal and the genetic differences between individual donors contribute to the major heterogeneity between different iPSC lines[35]. In this regard, iPSCs may serve as stable, reliable, and powerful platforms for the precise analysis of the hepatitis virus in specific genotypes and for modeling infection in individuals with different genetic backgrounds[29,36,37]. Additionally, induced hepatocytes (iHeps) generated by direct reprogramming from somatic cells share similar hepatic features and may be potentially used to support productive HBV infection[34].
Despite the progressive improvements reported thus far, in vitro hepatocyte induction with transcription factors, growth factors, or chemicals cannot fully represent the complete phenotypic spectrum of PHHs. Further, inhibition of the innate immune response remains an obstacle to achieve efficient infection[38]. Thus, improvements including optimized induction strategies and niche signaling are warranted to generate transcriptional/functional and hepatitis virus-friendly hepatic sources to recapitulate in vivo virus-host recognition.
In vitro microenvironment
The biological characteristics and functions of primary hepatocytes depend considerably on the in situ liver microenvironment[39,40]. Simply covering hepatocytes with multiple layers of collagen gel can markedly extend the culture time and help retain cellular functions[41]. Nevertheless, utilization of the conventional in vitro culture systems results in a rapid loss of hepatocyte polarity and associated liver function[42], innate immune responses, and susceptibility to HBV infection due to the unsatisfied intrinsic requirements of the liver microenvironment. Consequently, most of these HBV infection systems require the addition of DMSO, PEG[43], or immunomodulatory agents to achieve essential susceptibility, which is not feasible for practical considerations[34]. In vivo, however, HBV can infect more efficiently[44], which highlights that the native hepatic microenvironment, including the physiological liver architecture and niche signaling, markedly impacts productive HBV infection.
As the most abundant non-parenchymal hepatic cell population, liver sinusoidal endothelial cells (LSECs) can efficiently enhance HBV infection by secreting epidermal growth factor (EGF)[45] and can lead to the development of liver fibrosis by constitutively expressing the major histocompatibility complex I-restricted antigens and co-stimulatory molecules, which shifts the immune balance of the liver toward tolerance[46]. Additionally, hepatic stellate cells (HSCs) maintain hepatocyte function via cell signaling and secretion of the necessary factors[47]. Furthermore, HSCs are actively involved in the development of hepatitis virus-induced hepatic fibrosis[48]. Kupffer cells (KCs) and Ly6C+ monocytes are closely associated with host defense in the liver and play crucial roles in the development of anti-HBV immunity. Particularly, KCs are involved in hepatitis virus recognition, suppression of infection, immune cell recruitment and activation, and are correlated with HBV progression and outcomes[49,50]. The integration of multi-cellular interactions as a whole requires precise imitation of the native liver niche. Conventionally, monolayer co-culture systems are used as a simple tool to recapitulate cell-cell interactions and signaling. However, these systems provide non-natural physiological conditions, in which only parts of the hepatocyte surface establish communication with the adjacent cells or the extracellular matrix (ECM), leaving the rest of the cell exposed to the culture medium. Although certain aspects of hepatitis or other viral infections may be elucidated using this system, there remain major concerns regarding a physiologically intact host for virus recognition and replication. Cells assembled in 3D aggregates are more similar to cells in vivo[15]. PHHs cultured in 3D systems preserve certain metabolic functions and permit more accurate hepatotoxicity prediction during in vitro modeling[42]. However, the liver microenvironment includes the 3D mass of hepatocytes and an organized architecture consisting of numerous cell types, which synergistically regulate liver-specific physiology. Therefore, reconstruction of these interactions will theoretically help in the development of a more reliable system for the initiation and long-term maintenance of the hepatitis infection cycle. Recent achievements in generating multi-cellular organoids have paved the way for the establishment of an accurate model for obtaining fundamental knowledge on disease progression, including HBV infection[15], particularly when single patient-derived iPSCs contribute to the cellular composition of desired organoids[51]. iPSC-derived multi-cellular LOs maintain mature hepatic phenotypes and functions to levels comparable to those of PHHs. More importantly, LOs are more susceptible to HBV infection than iPSC-HLCs and maintain long-term HBV propagation while producing infectious viruses. Together with the genetic characteristics inherited from the donor iPSCs, LOs help in the achievement of a promising individualized infection model[15].
In static culture systems, gas exchange, nutrient supply, and waste removal remain significant challenges. The emergence of microfluidic technology has enabled the control of the culture system with optimized temperature, pH, nutrients, and gas exchange, while providing microscale structures and parameters that may help to obtain an approximate simulation of the in vivo microenvironment[52,53]. Thus, the application of these systems permits organoid maintenance in in vivo-like physiological states or even in disease-like conditions in a controlled manner. Notably, improved drug sensitivity can be obtained using microfluidic 3D systems compared to static culture systems. With the utilization of further engineering approaches to control the initial cellular composition, shape, and size of cell aggregates; cell-cell and cell-ECM[54] interactions; and biochemical gradients similar to in vivo microenvironment, the incoming generation of organoids-on-a-chip has considerable potential for large-scale applications in high-throughput drug testing and screening.
Micropatterning
Highly structured organs and tissues provide a fundamental in vivo microenvironment for cells, in which cell polarity and functions are consistently influenced by the specific boundaries imposed by the neighboring cells and the ECM. The mechanical and spatial properties of the microenvironment are tightly correlated with intracellular signaling pathways and affect cell transcriptome status and function. In the past decade, numerous studies have been conducted and have provided insights into the geometrical modeling of the in vitro culture microenvironment by developing engineered substrates that precisely mimic the composition, structure, and mechanical properties of a specific organ or tissue.
To date, accumulative in vitro studies show that cell differentiation can be directed by using micropatterned substrates. The micropattern features on the spreading area and the aspect ratio determine the differentiation status of human mesenchymal stem cells (MSCs)[55]. Moreover, hydroxyapatite bioceramic-based hybrid structures stimulate osteogenic differentiation of MSCs by activating integrins, the BMP2 signaling pathway, and cell-cell communication. The width of micropattern stripes regulates vascular smooth muscle cell orientation by regulating cell elongation[56]. Similarly, mechanical and spatial control was observed during pluripotent stem cell differentiation. When these cells are differentiated within geometrically uniform and circular micropatterns, the patterning of regionalized cell fate can be reproducibly recapitulated[57,58]. Moreover, recent evidence shows that a decreased patterning width promotes iPSC differentiation efficiency, suggesting that stricter topographies direct cell fate specification[59]. Interestingly, the micropattern stiffness, degradability, and biochemical composition can even promote pluripotency reprogramming, thus emphasizing the critical role of micropatterns in morphogenetic and functional remodeling[60].
Although micropatterning strategies are increasingly applied in cell-based modeling and testing, they pose challenges in ensuring compatibility with rapidly advancing organoid technology that requires the use of non-adhesive substrates and extensive cell-cell interactions for self-organization. To this end, scaffold-dependent micropatterning strategies were adopted in recent studies conducted on distinct epithelial organoid generation. Meanwhile, structural and functional improvements have been increasingly reported. For example, a microfilament-based floating scaffold was developed to perform micropatterning and to guide brain organoid self-organization with increased reproducibility and improved tissue architecture[61]. Additionally, a spatially confined hydrogel scaffold was developed to guide self-organization of tube-shaped functional intestinal organoids with an accessible lumen and a physiologically relevant spatial arrangement of crypt- and villus-like domains[62]. Similarly, in terms of LOs, both colloidal crystal and hydrogel-based scaffolds were found to support advanced liver functions[63,64]. However, to date, the mechanism of spatial microenvironmental control of organ/tissue-specific cell fate specification remains undetermined. Once identified, scaffold-based micropatterning systems are expected to offer a potential platform for generating liver-specific and regionalized cell types simultaneously from a single iPSC. These platforms will be of remarkable importance for constructing multi-cellular organoids with native spatial composition, and will help to elucidate the mechanisms by which intra- or intercellular signals regulate distinct cellular identities and maturation patterns, and will fundamentally mimic the in vivo physiological features and hepatic functions for precise hepatitis modeling.
CURRENT CHALLENGES IN GENERATING MULTI-CELLULAR LIVER ORGANOIDS
Liver-like cellular organoid composition and reconstitution
To better mimic an in vivo niche resembling liver-specific function and physiology for disease modeling, vascularized functional multi-cellular LOs were first developed in 2013[65]. However, in this study, human MSCs and umbilical vein endothelial cells (HUVECs) were used to substitute the liver intrinsic stromal cellular components, including HSCs and LSECs, which remain major obstacles for future clinical applications. To address this problem, a complete all-iPSC-based strategy was established, in which hepatic functions were promoted by using iPSC-septum transversum mesenchymal and iPSC-endothelial progenitor cells. In both global transcriptome and function, the new system was more advanced than the HUVEC/MSC-based strategy and was comparable to primary adult hepatocytes[16]. However, this system remains deficient in recapitulating the immune response and inflammation due to the lack of an essential liver component, KCs. Recently, KCs, hepatocytes, stellate cells, and biliary cells were successfully induced synchronously in a multi-cellular system by facilitating co-differentiation from iPSCs, which represents a real and more complex liver-like tissue system that may be used for modeling inflammatory and fibrotic responses[51]. With advanced integration and optimization of cellular composition (Table 1), multi-cellular LOs are expected to support precise and more complex disease modeling.
Table 1.
Current modeling strategies and applications of human multi-cellular liver organoids
|
Model/application
|
Multiple cells (ratio)
|
Culture system
|
Advances/significance
|
Limitations
|
Ref.
|
| ALF | iPSC-HEs, HUVECs, BM-MSCs (10:8:2) | 3D, Matrigel | Multi-cellular LOs with vascularization | Low reproducibility; Time-consuming | 2013[65] |
| ALF | HEs, MCs, ECs (all from iPSCs) (10:8:2) | 3D, ULA | All-iPSC-based strategy | Time-consuming; High cost | 2017[16] |
| ALF | iPSC endoderm cells, HUVECs, UC-MSCs (10:7:1) | 3D, ULA | LOs generated from single donor-derived cells | Low reproducibility; Time-consuming | 2018[75] |
| ALF | iPSCs, HAMECs (3:1) | EB, Agarose | HAMECs improved hepatic functions | Unable to reflect the nature cellular composition of liver | 2019[102] |
| Liver fibrosis | HepaRG, THP-1, hTERT-HSC | 3D, Hanging drop | LOs derived from cell lines | Functional deficiency | 2017[103] |
| Liver fibrosis and steatohepatitis | PHHs, KCs, HSCs, SECs (16:2:1:1) | 3D, ULA | LOs derived from primary cell sources | Low reproducibility; High cost | 2018[104] |
| Steatohepatitis | Hepatocytes, HSCs, BCs, KCs (all from iPSC) | 3D, ULA | Co-differentiation of multiple cell lineages for iPSC- LOs | Functions undetermined; Potential inter/intra-batch variability | 2019[51] |
| HBV infection ex vivo | iPSC endoderm cells, HUVECs, BM- MSCs (10:7:1) | 3D, ULA | Validation of advantages of iPSC-LOs in HBV modeling | Low reproducibility; Time-consuming | 2018[15] |
| Hepatic differentiation | iPSC-HEs, MSCs, HUVECs (10:2:7) | 3D, Matrigel | Platform to identify developmental paracrine signals involved in hepatocyte differentiation | Low reproducibility; Time-consuming | 2017[105] |
| Hepatic differentiation | iHEPs, ECs, HSCs, cholangiocytes (10:7:2:1) | 3D, ULA | Cholangiocytes impaired the hepatic functions in LOs and were associated with the liver disease relevant phenotype | Cholangiocyte activation in LOs was unclear | 2019[106] |
| Liver development and angiogenesis | Hepatocytes, BCs, ECs, HSCs (all from iPSC) | 3D, Matrigel | Engineered iPSC-LOs by programming of the gene regulatory network | Not completed for liver functions | 2021[66] |
ALF: Acute liver failure; BCs: Biliary cells; BM: Bone marrow; EB: Embryoid body; ECs: Endothelial cells; HAMECs: Human adipose microvascular endothelial cells; HBV: Hepatitis B virus; HEs: Hepatic endoderm cells; HSCs: Hepatic stellate cells; HUVECs: Human umbilical vein endothelial cells; iHEPs: Induced hepatocytes; KCs: Kupffer cells; Los: Liver organoids; MCs: Mesenchymal cells; MSCs: Mesenchymal stem cells; NPCs: Non-parenchymal cells; PHHs: Primary human hepatocytes; SECs: Sinusoidal endothelial cells; UC: Umbilical cord; ULA: Ultralow adhesion microwell plate.
With the progressive understanding of liver organogenesis and technical advancements in iPSC differentiation methods toward tissue-specific cellular compositions, the following aspects remain as future challenges: (1) The procedures that can be used to remodel the intrinsic cellular composition proportion and distribution in LOs; (2) Their specific roles in liver development and disease processes; (3) Clarification of biochemical signatures of liver-specific ECM; and (4) The use of clinically compatible ECM hydrogels or microparticles. Overcoming these challenges will be beneficial for maintaining LO physiological characteristics during modeling.
Novel strategies for synchronous induction
Before the all-iPSC-based strategy was adopted, the development of multi-cellular organoids included the collection and separate preparation of each cell type and subsequent self-organization into a 3D structure. Although the emergence of all-iPSC-based strategies facilitated the establishment of a reproducible and stable source for clinical and pharmaceutical applications, the procedure for diverse cell co-culture remains time-consuming, labor-intensive, inefficient, and cannot meet the requirements for large-scale production and practical applications. Moreover, selection of the specific culture medium and ECM to simultaneously maintain multiple cell lineages remains challenging.
To address these limitations, a stepwise organoid induction and maintenance platform was developed, which involved sequential stimulation with the addition of growth factor cocktails into the culture medium. Following the induction of foregut spheroids by retinoic acid (RA) treatment and subsequent hepatic maturation, iPSCs were simultaneously differentiated into multi-cellular LOs. However, this approach lacks precise control over multi-lineage specification and physiological functions. Furthermore, the optimization of medium and growth factors that are used to direct and maintain distinct cell specificity remains an obstacle. In this regard, systematic probing of the molecular pathways and transcriptional networks has emerged as a more precise method for direct organogenesis in vitro. PROX1 and ATF5 overexpression combined with CYP3A4 activation enables gene regulatory network (GRN)-based engineering and facilitates direct programming or induction and the development of iPSC-derived multi-cellular LOs. Importantly, compared to primary mature hepatocytes, the established GRNs not only showed major similarities in natural liver functions, but were also responsive to perturbation and feedback regulation, and thus were superior to immature HLCs and fetal LOs[66]. Although this new method enabled the development of multi-cellular systems via synthetic genetic control, it is difficult to reflect every facet of human liver physiology and function, such as relatively low levels of urea production and CYP2C19 activity. Further global assessment of GRNs may be necessary to identify critical regulatory signatures for advanced liver fate. Additionally, it is important to carefully determine and avoid potential tumorigenesis by transcription factor-based genetic programming and delocalization processes using guide RNAs (gRNAs) to achieve clinical safety standards.
Taken together, synchronous induction or programming systems may substantially improve the efficiency of traditional multi-lineage differentiation, which is a promising strategy for practical applications (Figure 1). However, the lack of maturation, purity, and batch-to-batch variability remains a major challenge. Further optimization of stepwise induction or programming strategies, including medium, supplements, timing, and application of tissue scaffolding and microfluidic devices, is warranted to precisely recapitulate the intrinsic multi-cellular organ system in vivo.
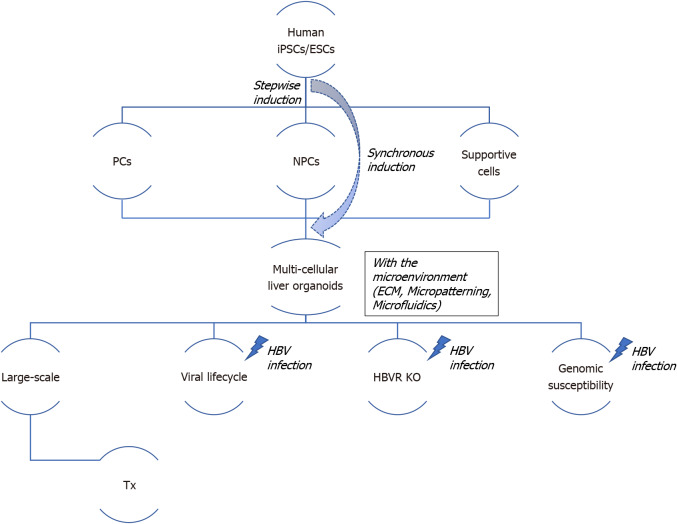
Figure 1.
Schema to generate human liver organoids and strategy regarding their application potential and hepatocyte B virus modeling. Multi-cellular liver organoids are generated and self-organized with parenchyma cells, non-parenchyma cells, and other hematopoietic or/and neuronal supportive cells after stepwise induction from human induced pluripotent stem cells/embryonic stem cells; in contrast, synchronous induction using a CRISPR-based strategy markedly enhances their hepatic functions and improves their reproducibility and scalability. The personalized and population organoid system may provide a reliable platform for high-throughput hepatocyte B virus (HBV) drug screening, allowing to understand novel key points of the HBV lifecycle, gene editing to knock-out the HBV receptor, and distinguish genomic susceptibility in a large population. Transplantable multi-cellular organoids without the HBV receptor have much potential for future applications. ECM: Extracellular matrix; ESCs: Embryonic stem cells; HBV: Hepatocyte B virus; HBVR: Hepatocyte B virus receptor; iPSCs: Induced pluripotent stem cells; KO: Knock-out; NPCs: Non-parenchyma cells, including Kupffer cells, hepatic stellate cells, and liver sinusoidal endothelial cells; Supportive cells: Such as neuronal cells, hematopoietic cells, and vascular endothelial cells; PCs: Parenchyma cells such as hepatocytes; Tx: Transplantation.
Scaling up
The possibility of scaled-up applications is a major advantage of using in vitro organoid systems over animal models. The increasing biomedical and preclinical demands for high-throughput disease modeling, drug testing, and screening, have led to the identification of critical bottleneck of scaling up, which prevents their application to provide more reliable, rapid, and cost-effective modeling.
Using chemical induction, a limited expansion capability of human hepatocytes was successfully induced in 2D monolayer culture systems[67,68]. However, expansion in a 3D system is subjected to complicated factors, such as nutrient/gas exchange, contact inhibition, and complex cell-cell signaling. Although long-term expansion of organoids from human adult biliary epithelial-derived progenitor cells, fetal/adult hepatocytes, and pluripotent stem cell-derived hepatocytes has been successfully established, the expansion efficiency remains markedly lower than that observed with the application of 2D systems[69-73] (Table 2). Recently, a rotating flask-based method was developed for the large-scale expansion of human LGR5-positive liver stem cell organoids. In this system, organoids were subjected to continuous passage and stable maintenance for at least 6 wk, which was mostly achieved with improved oxygenation[74].
Table 2.
Up-to-date methods for obtaining expandable human liver organoids
|
Initial cells
|
Expansion systems (substrate for embedding)
|
Medium
|
Expansion capability (Split ratio/passage days/expansion duration)
|
Ref.
|
| Fetal and adult hepatocytes | 3D, Matrigel | AdDMEM/F12, B27, N-Acetylcysteine, gastrin, RSPO1, Noggin, Wnt, EGF, FGF7, FGF10, HGF, TGFa, Nicotinamide, A83-01, CHIR99021 and Y27632 | 1:3/7-8 d/> 16 passages | 2018[70] |
| EpCAM+ bile duct cells | 3D, Matrigel or BME gel | AdDMEM/F12, N2, B27, N-Acetylcysteine, gastrin, RSPO1, Noggin (d0-3), Wnt (d0-3), EGF, FGF10, FGF19, HGF, Nicotinamide, A83-01, FSK, and Y27632 (d0-3) | 1:4-1:8/7-10 d/6 mo | 2015[69] |
| EpCAM+ bile duct cells | 3D, BME gel | AdDMEM/F12, B27, N-Acetylcysteine, gastrin, RSPO1, Noggin (d0-3), Wnt (d0-3), EGF, FGF10, FGF19, HGF, Nicotinamide, A83-01, FSK, and Y27632 (d0-3) | 1:5/7-10 d/> 6 mo | 2019[107] |
| iPSC derived EpCAM+ hepatic progenitors | 3D, Matrigel | AdDMEM/F12, B27, N-Acetylcysteine, gastrin, RSPO1, Noggin (d0-3), Wnt (d0-3), EGF, FGF10, HGF, Nicotinamide, A83-01, FSK and Y27632 (d0-3) | 1:4-1:8/7-10 d/9-12 mo | 2019[71] |
| PSC-derived hepatocytes | 3D, Matrigel | AdDMEM/F12, N2, B27, N-Acetylcysteine, gastrin, RSPO1, EGF, FGF10, HGF, Nicotinamide, A83-01, FSK | 1:3-1:10/7 d/3 mo | 2019[72] |
| PSCs transduced with PROX1, ATF5 and CYP3A4 | 3D, Matrigel | APEL medium | NA/10 d/17 d | 2021[66] |
APEL: A commercial medium from Stem Cell Technologies; BME: Basement membrane extract; bFGF: Basic fibroblast growth factor; BME: Basement membrane extract; EGF: Epidermal growth factor; FGF: Fibroblast growth factor; FSK: Forskolin; HGF: Hepatocyte growth factor; iPSCs: Induced pluripotent stem cells; PSCs: Including embryonic stem cells and induced pluripotent stem cells; RSPO1: R-spodin1.
Scaling up multi-cellular systems is another major challenge. By developing an omni-well array culture platform, Takebe et al[16] adopted an all-iPSC strategy for the large-scale production of homogenized and vascularized LOs in a clinical setting and achieved a 108-cell grade, which seemed to be feasible for human therapeutic applications. However, this platform involved the performance of labor-intensive work, including separate preparation of diverse cell sources for co-culture, thus limiting the efficacy of scaling. Later, the same group established a synchronous co-differentiation method that facilitated the generation of multi-lineage organoids in a preset growth factor administration strategy, thereby saving substantial time and costs for co-culture procedures[51]. Most recently, organoid programing has emerged as a promising strategy for establishing both functionality and scalability. A tissue-specific set of factors were identified using computational analyses. These factors were then genetically introduced into LOs, and after manipulation of GRNs, multi-lineage tissue fate could be directed to develop multi-cellular systems. Notably, these LOs can be passaged and cryopreserved while retaining their vascular networks[66]. This scalable organoid production system may pave the way for personalized disease modeling, drug testing, and even transplantation. However, there remain challenges in the identification of liver-specific GRNs to directly re-establish the entire profile of human liver physiology and function. Other tissue engineering techniques, such as pinning bioreactors, microfluidics, and bioprinting may be developed before high-throughput and highly efficient platforms can be established to meet the practical demands of pharmaceutical and clinical applications.
Transplantation and gene editing
In addition to demonstrating utility as an in vitro tool in the modeling of a structural and functional organ unit, organoids are also potential transplantation substitutes for organ donors. Indeed, organoids exhibit various advantages over conventional cell-based strategies, as mentioned elsewhere[69,75]. By mimicking liver regeneration after injury, an advanced in vitro system was recently developed for the long-term expansion of functional LOs. Particularly, LOs generated from primary liver sources have demonstrated genetic stability after long-term culture[69,70], suggesting that these organoids are a safe transplantation source for clinical applications. Alternatively, autologous transplantation of iPSC-derived LOs is expected to be promising for the realization of reproducible and personalized liver disease treatment. As a highly prevalent disease, HBV infection remains a strong risk factor for developing liver cirrhosis, hepatocellular carcinoma, or other end-stage liver diseases, thus posing a major threat to health worldwide[76]. To this end, there exists a necessity of developing effective treatments for chronic HBV infection. Currently, the standard therapy for chronic hepatitis B infection includes two major agents. First, nucleotide or nucleoside analogs such as tenofovir and entecavir interfere with viral replication by suppressing the synthesis of reverse transcriptase; second, IFNs such as IFNα induce long-lasting immunological control[76]. However, available drugs show side effects or high response variability, and drug resistance leads to reduced long-term effectiveness. Currently, HBV infection cannot be absolutely cured[77]. Combined with the CRISPR/Cas9 genome editing technology, patient iPSC-derived organoid transplantation may provide a new therapeutic strategy for realizing a highly specific HBV treatment. As the functional regions in key HBV receptors (e.g., NTCP) for HBV binding and post-binding entry have been identified, specific genetic knock-out or modification may markedly change HBV infection susceptibility, thereby permitting the development of transplanted organoids that are resistant to HBV infection. More importantly, the risk of HBV recurrence may be permanently avoided, which remains a major concern after liver donor transplantation and is associated with graft dysfunction or failure and cirrhosis progression[78].
Nevertheless, current research on organoids is in the preclinical stages, and clinical safety and efficacy remain to be established for adult or iPSC-derived LO transplantation. Moreover, the potential risks associated with genetic modification, tumorigenesis, and transplantation techniques should be emphasized in light of individual and societal values. In the context of continuing investigation regarding clinical concerns[79,80], quality-controlled and personalized LOs from authorized patient-derived cell banks are expected to be used routinely in the future.
PERSPECTIVES
Automated and large-scale cultural systems
Robust, consistent, and cost-effective manufacturing is extremely important for facilitating the practical applications of LOs for high-throughput disease modeling, drug testing, and therapeutic purposes. Similarly, automatic LO generation and culture systems may provide promising opportunities to meet critical requirements, especially in reproducibility and scalability.
In recent years, automated culture systems have garnered considerable attention, particularly in the maintenance and differentiation of iPSCs. Since the quality of iPSCs is dependent on the technical skills applied during culture and manipulation[81], automated culture systems can decrease technical variability. With the development of fully automated cell culture systems that have revolutionized cell seeding, medium changing, imaging, harvesting, and analysis, human iPSCs can be maintained in an undifferentiated state for a long period[82]. Additionally, reprogramming and differentiation of cardiomyocytes, hepatocytes, mesenchymal cells, neural stem cells, and retinal pigment epithelial cells have also been successfully performed using automated systems, with high quality and consistency[83-86]. Recently, an automated system was first used to produce LOs. Although the differentiation process was not fully automated, automatic LO self-assembly and maintenance substantially permitted the obtainment of reliable and reproducible results, suggesting potential applications in applied research and industry[87]. With the development of differentiation strategies and medium optimization for iPSC-derived parenchymal and non-parenchymal liver cells, the integration of automated cell production and maintenance systems is expected to further promote a completely standardized high-throughput workflow for LO production. These workflows will enable downstream applications such as HBV and liver cancer modeling, drug screening, and regenerative medicine.
Genetic manipulation
Genetic manipulation seems extremely promising in the revelation of the biological functions of specific genes, their regulation networks, and their relevance to disease progression[88]. Moreover, genetic manipulation facilitates drug testing in various disease states with different genetic backgrounds. In recent years, the CRISPR/Cas9 technology has markedly simplified genetic engineering because of its versatility and broad application potential. Combined with the development of organoid generation and culture technology, CRISPR has enabled the establishment of multiple disease models using genetically engineered tissue-specific organoids. For example, colorectal cancer models were derived from intestinal organoids with mutations in APC, SMAD4, TP53, and KRAS[89]; injury models were derived from kidney organoids via podocalyxin knock-out[90]; brain tumor models were derived from cerebral organoids with mutations in MYC and mutations commonly found in glioblastoma[91]; and liver cancer models including hepatocellular carcinomas and intrahepatic cholangiocarcinomas were derived from LOs with mutations in c-Myc or RAS[92]. Notably, genetically modified iPSC-derived organoids may permit broader practical applications in modeling genetic diseases once consistent patient-derived organoids are developed, such as those with A1AT deficiency or Allagile syndrome. Although not reported, iPSC-derived LOs are expected to acquire altered susceptibility to HBV infection with mutated NTCP expression, which is critical in studying viral replication.
In addition to demonstrating versatility as an in vitro tool used for the elucidation of substantial clues to diseases, gene correction in organoid models provides a potential strategy for future gene therapy. Using the CRISPR/Cas9 genome editing system, the mutation of cystic fibrosis transmembrane conductor receptor was corrected in intestinal organoids derived from cystic fibrosis patients, which restored normal function[93]. Subsequently, mutation correction was achieved in retinal organoids derived from retinitis pigmentosa patients using a similar strategy[94]. With increasing interest in the genetic manipulation of various tissue-specific organoids, the development of a robust genome editing strategy is necessary to further enhance organoid-based disease modeling and related gene correction. Recently, CRISPR–Cas9-mediated homology-independent organoid transgenesis (CRISPR-HOT) was developed to efficiently generate knock-in human organoids[95], thus providing a powerful platform for obtaining reliable and scalable applications in this field.
However, to date, no gene editing has been performed on multi-cellular organoids, partially because of the technical difficulties in targeting specific cell types within the system. Additionally, there are several issues that are encountered with gene therapy, including off-target mutations, delivery difficulties, and the lack of standardized tests to assess anti-HBV gene therapy[96,97]. Undoubtedly, new innovations in the rapidly evolving field of the CRISPR technology may provide exciting possibilities for organoid-based clinical applications.
Susceptibility to HBV infection
The identification of NTCP, the specific receptor for HBV[98], paved the way to construct NTCP-expressing hepatoma cell lines that were originally not susceptible to HBV. Exogenous NTCP expression may render these cell lines vulnerable to HBV infection, providing a more reliable system for studying the HBV life cycle. However, this system does not help elucidate the entire HBV life cycle and permits limited viral spread, with only moderate amounts of detectable cccDNA, partially due to intrinsic deficiencies of these cell lines. As a result, an extremely high multiplicity of infection is necessary to achieve substantial infection, and the addition of polyethylene glycol is needed to enhance infection in most reported cases[99]. Meanwhile, hepatoma cells are physiologically impaired in several intracellular signaling pathways and functions after forced NTBC expression, which limits their use for studying virus-host interactions[38].
In addition to NTCP, HBV infection may also depend on other receptors, such as the sialic acid glycoprotein receptor (ASGPR)[100,101]. A comparison of iPSC-derived hepatic progenitors and HLCs revealed a significant increase in ASGPR expression in iPSC-HLCs, suggesting that ASGPR might be considered a candidate receptor for hepatitis virus infection in mature hepatic cells and might mediate HBV entry into cells. To date, NTCP expression is considered the most important factor for HBV infection in host cells[13]. However, it remains necessary to identify other unknown receptors or media that contribute to hepatitis virus infection susceptibility. The emergence of 3D multi-cellular LOs, which mimic the native liver microenvironment, has attracted substantial attention owing to its superior HBV susceptibility compared to 2D culture[15]. Although the underlying mechanism remains unclear, these organoids may provide an opportunity for the investigation of potential receptors and related pathways involved in viral infection. Computational analyses, followed by sequential knock-out of the candidate targets in engineered multi-cellular LOs, may enable the complete understanding of the key genes contributing to HBV susceptibility. Furthermore, the role of ethnic genotypes in viral susceptibility may be studied using this system. Individuals of different races present with different susceptibilities to hepatitis virus. For example, Chinese populations have a high susceptibility to hepatitis B, while European and American populations are prone to hepatitis C infection. It would be beneficial to use iPSC-derived LOs from different genomic backgrounds to compare HBV infection in different populations with the same risk factors, or to compare susceptibility in the same population with different risk factors. In this regard, drugs, which may account for disparities in different populations and individuals, may also be developed.
CONCLUSION
With the progressive understanding of liver organogenesis, the development of 3D organoid culture systems has helped in the establishment of a novel platform for precise and personalized liver disease modeling. However, it remains particularly challenging to promote the efficiency, reproducibility, and scalability of organoid reconstitution and maintenance. It is expected that future advances in genetical engineering and automated culture system will put 3D multi-cellular organoids into a variety of practical uses, including hepatitis infection modeling and related drug development. Furthermore, the proof of concept for clinical-grade patient iPSC-derived multi-cellular LOs is expected and will contribute much to modeling and treatment of hepatitis virus infection and other liver diseases.
ACKNOWLEDGEMENTS
We thank Dr. Chen SL for helpful discussion, and Drs. Li YM and Liu LP for kind support in Institute of Regenerative Medicine of Jiangsu University.
Footnotes
Conflict-of-interest statement: The authors declare no potential financial interests.
Manuscript source: Invited manuscript
Peer-review started: March 9, 2021
First decision: April 17, 2021
Article in press: July 15, 2021
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Japan
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Wang C, Zhu F S-Editor: Gao CC L-Editor: A P-Editor: Yuan YY
Contributor Information
Di Cao, Institute of Regenerative Medicine and Affiliated Hospital of Jiangsu University, Jiangsu University, Zhenjiang 212001, Jiangsu Province, China.
Jian-Yun Ge, Department of Gastrointestinal and Hepato-Biliary-Pancreatic Surgery, Faculty of Medicine, University of Tsukuba, Tsukuba 305-8575, Ibaraki, Japan; Guangdong Provincial Key Laboratory of Large Animal Models for Biomedicine, and School of Biotechnology and Heath Sciences, Wuyi University, Jiangmen 529020, Guangdong Province, China.
Yun Wang, Institute of Regenerative Medicine and Affiliated Hospital of Jiangsu University, Jiangsu University, Zhenjiang 212001, Jiangsu Province, China.
Tatsuya Oda, Department of Gastrointestinal and Hepato-Biliary-Pancreatic Surgery, Faculty of Medicine, University of Tsukuba, Tsukuba 305-8575, Ibaraki, Japan.
Yun-Wen Zheng, Institute of Regenerative Medicine and Affiliated Hospital of Jiangsu University, Jiangsu University, Zhenjiang 212001, Jiangsu Province, China; Department of Gastrointestinal and Hepato-Biliary-Pancreatic Surgery, Faculty of Medicine, University of Tsukuba, Tsukuba 305-8575, Ibaraki, Japan; Guangdong Provincial Key Laboratory of Large Animal Models for Biomedicine, and School of Biotechnology and Heath Sciences, Wuyi University, Jiangmen 529020, Guangdong Province, China; School of Medicine, Yokohama City University, Yokohama 234-0006, Kanagawa, Japan. ywzheng@md.tsukuba.ac.jp.
References
- 1.Razavi H. Global Epidemiology of Viral Hepatitis. Gastroenterol Clin North Am. 2020;49:179–189. doi: 10.1016/j.gtc.2020.01.001. [DOI] [PubMed] [Google Scholar]
- 2.Yuen MF, Chen DS, Dusheiko GM, Janssen HLA, Lau DTY, Locarnini SA, Peters MG, Lai CL. Hepatitis B virus infection. Nat Rev Dis Primers. 2018;4:18035. doi: 10.1038/nrdp.2018.35. [DOI] [PubMed] [Google Scholar]
- 3.Wose Kinge CN, Bhoola NH, Kramvis A. In Vitro Systems for Studying Different Genotypes/Sub-Genotypes of Hepatitis B Virus: Strengths and Limitations. Viruses. 2020;12 doi: 10.3390/v12030353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kim BK, Revill PA, Ahn SH. HBV genotypes: relevance to natural history, pathogenesis and treatment of chronic hepatitis B. Antivir Ther. 2011;16:1169–1186. doi: 10.3851/IMP1982. [DOI] [PubMed] [Google Scholar]
- 5.Pawlotsky JM, Feld JJ, Zeuzem S, Hoofnagle JH. From non-A, non-B hepatitis to hepatitis C virus cure. J Hepatol. 2015;62:S87–S99. doi: 10.1016/j.jhep.2015.02.006. [DOI] [PubMed] [Google Scholar]
- 6.Hu J, Lin YY, Chen PJ, Watashi K, Wakita T. Cell and Animal Models for Studying Hepatitis B Virus Infection and Drug Development. Gastroenterology. 2019;156:338–354. doi: 10.1053/j.gastro.2018.06.093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Xia Y, Carpentier A, Cheng X, Block PD, Zhao Y, Zhang Z, Protzer U, Liang TJ. Human stem cell-derived hepatocytes as a model for hepatitis B virus infection, spreading and virus-host interactions. J Hepatol. 2017;66:494–503. doi: 10.1016/j.jhep.2016.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hoh A, Heeg M, Ni Y, Schuch A, Binder B, Hennecke N, Blum HE, Nassal M, Protzer U, Hofmann M, Urban S, Thimme R. Hepatitis B Virus-Infected HepG2hNTCP Cells Serve as a Novel Immunological Tool To Analyze the Antiviral Efficacy of CD8+ T Cells In Vitro. J Virol. 2015;89:7433–7438. doi: 10.1128/JVI.00605-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ortega-Prieto AM, Skelton JK, Cherry C, Briones-Orta MA, Hateley CA, Dorner M. "Liver-on-a-Chip" Cultures of Primary Hepatocytes and Kupffer Cells for Hepatitis B Virus Infection. J Vis Exp. 2019 doi: 10.3791/58333. [DOI] [PubMed] [Google Scholar]
- 10.Si-Tayeb K, Noto FK, Nagaoka M, Li J, Battle MA, Duris C, North PE, Dalton S, Duncan SA. Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology. 2010;51:297–305. doi: 10.1002/hep.23354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Choi SM, Kim Y, Liu H, Chaudhari P, Ye Z, Jang YY. Liver engraftment potential of hepatic cells derived from patient-specific induced pluripotent stem cells. Cell Cycle. 2011;10:2423–2427. doi: 10.4161/cc.10.15.16869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rashid ST, Corbineau S, Hannan N, Marciniak SJ, Miranda E, Alexander G, Huang-Doran I, Griffin J, Ahrlund-Richter L, Skepper J, Semple R, Weber A, Lomas DA, Vallier L. Modeling inherited metabolic disorders of the liver using human induced pluripotent stem cells. J Clin Invest. 2010;120:3127–3136. doi: 10.1172/JCI43122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kaneko S, Kakinuma S, Asahina Y, Kamiya A, Miyoshi M, Tsunoda T, Nitta S, Asano Y, Nagata H, Otani S, Kawai-Kitahata F, Murakawa M, Itsui Y, Nakagawa M, Azuma S, Nakauchi H, Nishitsuji H, Ujino S, Shimotohno K, Iwamoto M, Watashi K, Wakita T, Watanabe M. Human induced pluripotent stem cell-derived hepatic cell lines as a new model for host interaction with hepatitis B virus. Sci Rep. 2016;6:29358. doi: 10.1038/srep29358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sakurai F, Mitani S, Yamamoto T, Takayama K, Tachibana M, Watashi K, Wakita T, Iijima S, Tanaka Y, Mizuguchi H. Human induced-pluripotent stem cell-derived hepatocyte-like cells as an in vitro model of human hepatitis B virus infection. Sci Rep. 2017;7:45698. doi: 10.1038/srep45698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nie YZ, Zheng YW, Miyakawa K, Murata S, Zhang RR, Sekine K, Ueno Y, Takebe T, Wakita T, Ryo A, Taniguchi H. Recapitulation of hepatitis B virus-host interactions in liver organoids from human induced pluripotent stem cells. EBioMedicine. 2018;35:114–123. doi: 10.1016/j.ebiom.2018.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Takebe T, Sekine K, Kimura M, Yoshizawa E, Ayano S, Koido M, Funayama S, Nakanishi N, Hisai T, Kobayashi T, Kasai T, Kitada R, Mori A, Ayabe H, Ejiri Y, Amimoto N, Yamazaki Y, Ogawa S, Ishikawa M, Kiyota Y, Sato Y, Nozawa K, Okamoto S, Ueno Y, Taniguchi H. Massive and Reproducible Production of Liver Buds Entirely from Human Pluripotent Stem Cells. Cell Rep. 2017;21:2661–2670. doi: 10.1016/j.celrep.2017.11.005. [DOI] [PubMed] [Google Scholar]
- 17.Allweiss L, Dandri M. Experimental in vitro and in vivo models for the study of human hepatitis B virus infection. J Hepatol. 2016;64:S17–S31. doi: 10.1016/j.jhep.2016.02.012. [DOI] [PubMed] [Google Scholar]
- 18.Zeilinger K, Freyer N, Damm G, Seehofer D, Knöspel F. Cell sources for in vitro human liver cell culture models. Exp Biol Med (Maywood) 2016;241:1684–1698. doi: 10.1177/1535370216657448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Torresi J, Tran BM, Christiansen D, Earnest-Silveira L, Schwab RHM, Vincan E. HBV-related hepatocarcinogenesis: the role of signalling pathways and innovative ex vivo research models. BMC Cancer. 2019;19:707. doi: 10.1186/s12885-019-5916-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Severi T, Ying C, Vermeesch JR, Cassiman D, Cnops L, Verslype C, Fevery J, Arckens L, Neyts J, van Pelt JF. Hepatitis B virus replication causes oxidative stress in HepAD38 Liver cells. Mol Cell Biochem. 2006;290:79–85. doi: 10.1007/s11010-006-9167-x. [DOI] [PubMed] [Google Scholar]
- 21.Cai D, Wang X, Yan R, Mao R, Liu Y, Ji C, Cuconati A, Guo H. Establishment of an inducible HBV stable cell line that expresses cccDNA-dependent epitope-tagged HBeAg for screening of cccDNA modulators. Antiviral Res. 2016;132:26–37. doi: 10.1016/j.antiviral.2016.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cai D, Mills C, Yu W, Yan R, Aldrich CE, Saputelli JR, Mason WS, Xu X, Guo JT, Block TM, Cuconati A, Guo H. Identification of disubstituted sulfonamide compounds as specific inhibitors of hepatitis B virus covalently closed circular DNA formation. Antimicrob Agents Chemother. 2012;56:4277–4288. doi: 10.1128/AAC.00473-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Guo H, Jiang D, Zhou T, Cuconati A, Block TM, Guo JT. Characterization of the intracellular deproteinized relaxed circular DNA of hepatitis B virus: an intermediate of covalently closed circular DNA formation. J Virol. 2007;81:12472–12484. doi: 10.1128/JVI.01123-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yan H, Zhong G, Xu G, He W, Jing Z, Gao Z, Huang Y, Qi Y, Peng B, Wang H, Fu L, Song M, Chen P, Gao W, Ren B, Sun Y, Cai T, Feng X, Sui J, Li W. Sodium taurocholate cotransporting polypeptide is a functional receptor for human hepatitis B and D virus. Elife. 2012;1:e00049. doi: 10.7554/eLife.00049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Thomas E, Liang TJ. Experimental models of hepatitis B and C - new insights and progress. Nat Rev Gastroenterol Hepatol. 2016;13:362–374. doi: 10.1038/nrgastro.2016.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tyakht AV, Ilina EN, Alexeev DG, Ischenko DS, Gorbachev AY, Semashko TA, Larin AK, Selezneva OV, Kostryukova ES, Karalkin PA, Vakhrushev IV, Kurbatov LK, Archakov AI, Govorun VM. RNA-Seq gene expression profiling of HepG2 cells: the influence of experimental factors and comparison with liver tissue. BMC Genomics. 2014;15:1108. doi: 10.1186/1471-2164-15-1108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Yang X, Cai W, Sun X, Bi Y, Zeng C, Zhao X, Zhou Q, Xu T, Xie Q, Sun P, Zhou X. Defined host factors support HBV infection in non-hepatic 293T cells. J Cell Mol Med. 2020;24:2507–2518. doi: 10.1111/jcmm.14944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- 29.Takayama K, Morisaki Y, Kuno S, Nagamoto Y, Harada K, Furukawa N, Ohtaka M, Nishimura K, Imagawa K, Sakurai F, Tachibana M, Sumazaki R, Noguchi E, Nakanishi M, Hirata K, Kawabata K, Mizuguchi H. Prediction of interindividual differences in hepatic functions and drug sensitivity by using human iPS-derived hepatocytes. Proc Natl Acad Sci USA. 2014;111:16772–16777. doi: 10.1073/pnas.1413481111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sa-Ngiamsuntorn K, Wongkajornsilp A, Phanthong P, Borwornpinyo S, Kitiyanant N, Chantratita W, Hongeng S. A robust model of natural hepatitis C infection using hepatocyte-like cells derived from human induced pluripotent stem cells as a long-term host. Virol J. 2016;13:59. doi: 10.1186/s12985-016-0519-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Takayama K, Inamura M, Kawabata K, Sugawara M, Kikuchi K, Higuchi M, Nagamoto Y, Watanabe H, Tashiro K, Sakurai F, Hayakawa T, Furue MK, Mizuguchi H. Generation of metabolically functioning hepatocytes from human pluripotent stem cells by FOXA2 and HNF1α transduction. J Hepatol. 2012;57:628–636. doi: 10.1016/j.jhep.2012.04.038. [DOI] [PubMed] [Google Scholar]
- 32.Takayama K, Inamura M, Kawabata K, Katayama K, Higuchi M, Tashiro K, Nonaka A, Sakurai F, Hayakawa T, Furue MK, Mizuguchi H. Efficient generation of functional hepatocytes from human embryonic stem cells and induced pluripotent stem cells by HNF4α transduction. Mol Ther. 2012;20:127–137. doi: 10.1038/mt.2011.234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Inamura M, Kawabata K, Takayama K, Tashiro K, Sakurai F, Katayama K, Toyoda M, Akutsu H, Miyagawa Y, Okita H, Kiyokawa N, Umezawa A, Hayakawa T, Furue MK, Mizuguchi H. Efficient generation of hepatoblasts from human ES cells and iPS cells by transient overexpression of homeobox gene HEX. Mol Ther. 2011;19:400–407. doi: 10.1038/mt.2010.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Shlomai A, Schwartz RE, Ramanan V, Bhatta A, de Jong YP, Bhatia SN, Rice CM. Modeling host interactions with hepatitis B virus using primary and induced pluripotent stem cell-derived hepatocellular systems. Proc Natl Acad Sci USA. 2014;111:12193–12198. doi: 10.1073/pnas.1412631111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yoshida Y, Yamanaka S. Induced Pluripotent Stem Cells 10 Years Later: For Cardiac Applications. Circ Res. 2017;120:1958–1968. doi: 10.1161/CIRCRESAHA.117.311080. [DOI] [PubMed] [Google Scholar]
- 36.Wang T, Zhang J, Liao J, Zhang F, Zhou G. Donor genetic backgrounds contribute to the functional heterogeneity of stem cells and clinical outcomes. Stem Cells Transl Med. 2020;9:1495–1499. doi: 10.1002/sctm.20-0155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rouhani F, Kumasaka N, de Brito MC, Bradley A, Vallier L, Gaffney D. Genetic background drives transcriptional variation in human induced pluripotent stem cells. PLoS Genet. 2014;10:e1004432. doi: 10.1371/journal.pgen.1004432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Witt-Kehati D, Bitton Alaluf M, Shlomai A. Advances and Challenges in Studying Hepatitis B Virus In Vitro. Viruses. 2016;8 doi: 10.3390/v8010021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Buck LD, Inman SW, Rusyn I, Griffith LG. Co-regulation of primary mouse hepatocyte viability and function by oxygen and matrix. Biotechnol Bioeng. 2014;111:1018–1027. doi: 10.1002/bit.25152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kirchner VA, Tak E, Kim K, LeCluyse EL, Niedernhofer LJ, Soldatow V, Lee J, Kim J, Tolar J, Song GW, Pruett TL. The evolving microenvironment of the human hepatocyte: Healthy vs. cirrhotic liver vs. isolated cells. Tissue Cell. 2020;62:101310. doi: 10.1016/j.tice.2019.101310. [DOI] [PubMed] [Google Scholar]
- 41.Berger DR, Ware BR, Davidson MD, Allsup SR, Khetani SR. Enhancing the functional maturity of induced pluripotent stem cell-derived human hepatocytes by controlled presentation of cell-cell interactions in vitro. Hepatology. 2015;61:1370–1381. doi: 10.1002/hep.27621. [DOI] [PubMed] [Google Scholar]
- 42.Schyschka L, Sánchez JJ, Wang Z, Burkhardt B, Müller-Vieira U, Zeilinger K, Bachmann A, Nadalin S, Damm G, Nussler AK. Hepatic 3D cultures but not 2D cultures preserve specific transporter activity for acetaminophen-induced hepatotoxicity. Arch Toxicol. 2013;87:1581–1593. doi: 10.1007/s00204-013-1080-y. [DOI] [PubMed] [Google Scholar]
- 43.Schulze A, Mills K, Weiss TS, Urban S. Hepatocyte polarization is essential for the productive entry of the hepatitis B virus. Hepatology. 2012;55:373–383. doi: 10.1002/hep.24707. [DOI] [PubMed] [Google Scholar]
- 44.Ortega-Prieto AM, Skelton JK, Wai SN, Large E, Lussignol M, Vizcay-Barrena G, Hughes D, Fleck RA, Thursz M, Catanese MT, Dorner M. 3D microfluidic liver cultures as a physiological preclinical tool for hepatitis B virus infection. Nat Commun. 2018;9:682. doi: 10.1038/s41467-018-02969-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Chen SW, Himeno M, Koui Y, Sugiyama M, Nishitsuji H, Mizokami M, Shimotohno K, Miyajima A, Kido T. Modulation of hepatitis B virus infection by epidermal growth factor secreted from liver sinusoidal endothelial cells. Sci Rep. 2020;10:14349. doi: 10.1038/s41598-020-71453-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ni Y, Li JM, Liu MK, Zhang TT, Wang DP, Zhou WH, Hu LZ, Lv WL. Pathological process of liver sinusoidal endothelial cells in liver diseases. World J Gastroenterol. 2017;23:7666–7677. doi: 10.3748/wjg.v23.i43.7666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Tsuchida T, Friedman SL. Mechanisms of hepatic stellate cell activation. Nat Rev Gastroenterol Hepatol. 2017;14:397–411. doi: 10.1038/nrgastro.2017.38. [DOI] [PubMed] [Google Scholar]
- 48.Shi J, Zhao J, Zhang X, Cheng Y, Hu J, Li Y, Zhao X, Shang Q, Sun Y, Tu B, Shi L, Gao B, Wang FS, Zhang Z. Activated hepatic stellate cells impair NK cell anti-fibrosis capacity through a TGF-β-dependent emperipolesis in HBV cirrhotic patients. Sci Rep. 2017;7:44544. doi: 10.1038/srep44544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wu LL, Peng WH, Wu HL, Miaw SC, Yeh SH, Yang HC, Liao PH, Lin JS, Chen YR, Hong YT, Wang HY, Chen PJ, Chen DS. Lymphocyte Antigen 6 Complex, Locus C+ Monocytes and Kupffer Cells Orchestrate Liver Immune Responses Against Hepatitis B Virus in Mice. Hepatology. 2019;69:2364–2380. doi: 10.1002/hep.30510. [DOI] [PubMed] [Google Scholar]
- 50.Li Y, Li S, Duan X, Yang C, Xu M, Chen L. Macrophage Phenotypes and Hepatitis B Virus Infection. J Clin Transl Hepatol. 2020;8:424–431. doi: 10.14218/JCTH.2020.00046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ouchi R, Togo S, Kimura M, Shinozawa T, Koido M, Koike H, Thompson W, Karns RA, Mayhew CN, McGrath PS, McCauley HA, Zhang RR, Lewis K, Hakozaki S, Ferguson A, Saiki N, Yoneyama Y, Takeuchi I, Mabuchi Y, Akazawa C, Yoshikawa HY, Wells JM, Takebe T. Modeling Steatohepatitis in Humans with Pluripotent Stem Cell-Derived Organoids. Cell Metab. 2019;30:374–384.e6. doi: 10.1016/j.cmet.2019.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Yu F, Hunziker W, Choudhury D. Engineering Microfluidic Organoid-on-a-Chip Platforms. Micromachines (Basel) 2019;10 doi: 10.3390/mi10030165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yu F, Deng R, Hao Tong W, Huan L, Chan Way N, IslamBadhan A, Iliescu C, Yu H. A perfusion incubator liver chip for 3D cell culture with application on chronic hepatotoxicity testing. Sci Rep. 2017;7:14528. doi: 10.1038/s41598-017-13848-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Yang BA, Westerhof TM, Sabin K, Merajver SD, Aguilar CA. Engineered Tools to Study Intercellular Communication. Adv Sci (Weinh) 2021;8:2002825. doi: 10.1002/advs.202002825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wang X, Nakamoto T, Dulińska-Molak I, Kawazoe N, Chen G. Regulating the stemness of mesenchymal stem cells by tuning micropattern features. J Mater Chem B. 2016;4:37–45. doi: 10.1039/c5tb02215k. [DOI] [PubMed] [Google Scholar]
- 56.Nakamoto T, Wang X, Kawazoe N, Chen G. Influence of micropattern width on differentiation of human mesenchymal stem cells to vascular smooth muscle cells. Colloids Surf B Biointerfaces. 2014;122:316–323. doi: 10.1016/j.colsurfb.2014.06.013. [DOI] [PubMed] [Google Scholar]
- 57.Tewary M, Ostblom J, Prochazka L, Zulueta-Coarasa T, Shakiba N, Fernandez-Gonzalez R, Zandstra PW. A stepwise model of reaction-diffusion and positional information governs self-organized human peri-gastrulation-like patterning. Development. 2017;144:4298–4312. doi: 10.1242/dev.149658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Morgani SM, Metzger JJ, Nichols J, Siggia ED, Hadjantonakis AK. Micropattern differentiation of mouse pluripotent stem cells recapitulates embryo regionalized cell fate patterning. Elife. 2018;7 doi: 10.7554/eLife.32839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kusuma S, Smith Q, Facklam A, Gerecht S. Micropattern size-dependent endothelial differentiation from a human induced pluripotent stem cell line. J Tissue Eng Regen Med. 2017;11:855–861. doi: 10.1002/term.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Caiazzo M, Okawa Y, Ranga A, Piersigilli A, Tabata Y, Lutolf MP. Defined three-dimensional microenvironments boost induction of pluripotency. Nat Mater. 2016;15:344–352. doi: 10.1038/nmat4536. [DOI] [PubMed] [Google Scholar]
- 61.Lancaster MA, Corsini NS, Wolfinger S, Gustafson EH, Phillips AW, Burkard TR, Otani T, Livesey FJ, Knoblich JA. Guided self-organization and cortical plate formation in human brain organoids. Nat Biotechnol. 2017;35:659–666. doi: 10.1038/nbt.3906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Nikolaev M, Mitrofanova O, Broguiere N, Geraldo S, Dutta D, Tabata Y, Elci B, Brandenberg N, Kolotuev I, Gjorevski N, Clevers H, Lutolf MP. Homeostatic mini-intestines through scaffold-guided organoid morphogenesis. Nature. 2020;585:574–578. doi: 10.1038/s41586-020-2724-8. [DOI] [PubMed] [Google Scholar]
- 63.Ng SS, Saeb-Parsy K, Blackford SJI, Segal JM, Serra MP, Horcas-Lopez M, No DY, Mastoridis S, Jassem W, Frank CW, Cho NJ, Nakauchi H, Glenn JS, Rashid ST. Human iPS derived progenitors bioengineered into liver organoids using an inverted colloidal crystal poly (ethylene glycol) scaffold. Biomaterials. 2018;182:299–311. doi: 10.1016/j.biomaterials.2018.07.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Wang Y, Liu H, Zhang M, Wang H, Chen W, Qin J. One-step synthesis of composite hydrogel capsules to support liver organoid generation from hiPSCs. Biomater Sci. 2020;8:5476–5488. doi: 10.1039/d0bm01085e. [DOI] [PubMed] [Google Scholar]
- 65.Takebe T, Sekine K, Enomura M, Koike H, Kimura M, Ogaeri T, Zhang RR, Ueno Y, Zheng YW, Koike N, Aoyama S, Adachi Y, Taniguchi H. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature. 2013;499:481–484. doi: 10.1038/nature12271. [DOI] [PubMed] [Google Scholar]
- 66.Velazquez JJ, LeGraw R, Moghadam F, Tan Y, Kilbourne J, Maggiore JC, Hislop J, Liu S, Cats D, Chuva de Sousa Lopes SM, Plaisier C, Cahan P, Kiani S, Ebrahimkhani MR. Gene Regulatory Network Analysis and Engineering Directs Development and Vascularization of Multilineage Human Liver Organoids. Cell Syst. 2021;12:41–55.e11. doi: 10.1016/j.cels.2020.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zhang K, Zhang L, Liu W, Ma X, Cen J, Sun Z, Wang C, Feng S, Zhang Z, Yue L, Sun L, Zhu Z, Chen X, Feng A, Wu J, Jiang Z, Li P, Cheng X, Gao D, Peng L, Hui L. In Vitro Expansion of Primary Human Hepatocytes with Efficient Liver Repopulation Capacity. Cell Stem Cell. 2018;23:806–819.e4. doi: 10.1016/j.stem.2018.10.018. [DOI] [PubMed] [Google Scholar]
- 68.Ge JY, Zheng YW, Liu LP, Isoda H, Oda T. Impelling force and current challenges by chemicals in somatic cell reprogramming and expansion beyond hepatocytes. World J Stem Cells. 2019;11:650–665. doi: 10.4252/wjsc.v11.i9.650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Huch M, Gehart H, van Boxtel R, Hamer K, Blokzijl F, Verstegen MM, Ellis E, van Wenum M, Fuchs SA, de Ligt J, van de Wetering M, Sasaki N, Boers SJ, Kemperman H, de Jonge J, Ijzermans JN, Nieuwenhuis EE, Hoekstra R, Strom S, Vries RR, van der Laan LJ, Cuppen E, Clevers H. Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell. 2015;160:299–312. doi: 10.1016/j.cell.2014.11.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hu H, Gehart H, Artegiani B, LÖpez-Iglesias C, Dekkers F, Basak O, van Es J, Chuva de Sousa Lopes SM, Begthel H, Korving J, van den Born M, Zou C, Quirk C, Chiriboga L, Rice CM, Ma S, Rios A, Peters PJ, de Jong YP, Clevers H. Long-Term Expansion of Functional Mouse and Human Hepatocytes as 3D Organoids. Cell. 2018;175:1591–1606.e19. doi: 10.1016/j.cell.2018.11.013. [DOI] [PubMed] [Google Scholar]
- 71.Akbari S, Sevinç GG, Ersoy N, Basak O, Kaplan K, Sevinç K, Ozel E, Sengun B, Enustun E, Ozcimen B, Bagriyanik A, Arslan N, Önder TT, Erdal E. Robust, Long-Term Culture of Endoderm-Derived Hepatic Organoids for Disease Modeling. Stem Cell Reports. 2019;13:627–641. doi: 10.1016/j.stemcr.2019.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Mun SJ, Ryu JS, Lee MO, Son YS, Oh SJ, Cho HS, Son MY, Kim DS, Kim SJ, Yoo HJ, Lee HJ, Kim J, Jung CR, Chung KS, Son MJ. Generation of expandable human pluripotent stem cell-derived hepatocyte-like liver organoids. J Hepatol. 2019;71:970–985. doi: 10.1016/j.jhep.2019.06.030. [DOI] [PubMed] [Google Scholar]
- 73.Huch M, Koo BK. Modeling mouse and human development using organoid cultures. Development. 2015;142:3113–3125. doi: 10.1242/dev.118570. [DOI] [PubMed] [Google Scholar]
- 74.Schneeberger K, Sánchez-Romero N, Ye S, van Steenbeek FG, Oosterhoff LA, Pla Palacin I, Chen C, van Wolferen ME, van Tienderen G, Lieshout R, Colemonts-Vroninks H, Schene I, Hoekstra R, Verstegen MMA, van der Laan LJW, Penning LC, Fuchs SA, Clevers H, De Kock J, Baptista PM, Spee B. Large-Scale Production of LGR5-Positive Bipotential Human Liver Stem Cells. Hepatology. 2020;72:257–270. doi: 10.1002/hep.31037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Nie YZ, Zheng YW, Ogawa M, Miyagi E, Taniguchi H. Human liver organoids generated with single donor-derived multiple cells rescue mice from acute liver failure. Stem Cell Res Ther. 2018;9:5. doi: 10.1186/s13287-017-0749-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Krause A, Haberkorn U, Mier W. Strategies for the treatment of HBV/HDV. Eur J Pharmacol. 2018;833:379–391. doi: 10.1016/j.ejphar.2018.06.030. [DOI] [PubMed] [Google Scholar]
- 77.European Association for the Study of the Liver. EASL 2017 Clinical Practice Guidelines on the management of hepatitis B virus infection. J Hepatol. 2017;67:370–398. doi: 10.1016/j.jhep.2017.03.021. [DOI] [PubMed] [Google Scholar]
- 78.Teegen EM, Maurer MM, Globke B, Pratschke J, Eurich D. Liver transplantation for Hepatitis-B-associated liver disease - Three decades of experience. Transpl Infect Dis. 2019;21:e12997. doi: 10.1111/tid.12997. [DOI] [PubMed] [Google Scholar]
- 79.Tsuchida T, Murata S, Hasegawa S, Mikami S, Enosawa S, Hsu HC, Fukuda A, Okamoto S, Mori A, Matsuo M, Kawakatsu Y, Matsunari H, Nakano K, Nagashima H, Taniguchi H. Investigation of Clinical Safety of Human iPS Cell-Derived Liver Organoid Transplantation to Infantile Patients in Porcine Model. Cell Transplant. 2020;29:963689720964384. doi: 10.1177/0963689720964384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Tsuchida T, Murata S, Matsuki K, Mori A, Matsuo M, Mikami S, Okamoto S, Ueno Y, Tadokoro T, Zheng YW, Taniguchi H. The Regenerative Effect of Portal Vein Injection of Liver Organoids by Retrorsine/Partial Hepatectomy in Rats. Int J Mol Sci. 2019;21 doi: 10.3390/ijms21010178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Chen KG, Mallon BS, McKay RD, Robey PG. Human pluripotent stem cell culture: considerations for maintenance, expansion, and therapeutics. Cell Stem Cell. 2014;14:13–26. doi: 10.1016/j.stem.2013.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Konagaya S, Ando T, Yamauchi T, Suemori H, Iwata H. Long-term maintenance of human induced pluripotent stem cells by automated cell culture system. Sci Rep. 2015;5:16647. doi: 10.1038/srep16647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Paull D, Sevilla A, Zhou H, Hahn AK, Kim H, Napolitano C, Tsankov A, Shang L, Krumholz K, Jagadeesan P, Woodard CM, Sun B, Vilboux T, Zimmer M, Forero E, Moroziewicz DN, Martinez H, Malicdan MC, Weiss KA, Vensand LB, Dusenberry CR, Polus H, Sy KT, Kahler DJ, Gahl WA, Solomon SL, Chang S, Meissner A, Eggan K, Noggle SA. Automated, high-throughput derivation, characterization and differentiation of induced pluripotent stem cells. Nat Methods. 2015;12:885–892. doi: 10.1038/nmeth.3507. [DOI] [PubMed] [Google Scholar]
- 84.Regent F, Morizur L, Lesueur L, Habeler W, Plancheron A, Ben M'Barek K, Monville C. Automation of human pluripotent stem cell differentiation toward retinal pigment epithelial cells for large-scale productions. Sci Rep. 2019;9:10646. doi: 10.1038/s41598-019-47123-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.McLaren D, Gorba T, Marguerie de Rotrou A, Pillai G, Chappell C, Stacey A, Lingard S, Falk A, Smith A, Koch P, Brüstle O, Vickers R, Tinsley J, Flanders D, Bello P, Craig S. Automated large-scale culture and medium-throughput chemical screen for modulators of proliferation and viability of human induced pluripotent stem cell-derived neuroepithelial-like stem cells. J Biomol Screen. 2013;18:258–268. doi: 10.1177/1087057112461446. [DOI] [PubMed] [Google Scholar]
- 86.Boussaad I, Cruciani G, Bolognin S, Antony P, Dording CM, Kwon YJ, Heutink P, Fava E, Schwamborn JC, Krüger R. Integrated, automated maintenance, expansion and differentiation of 2D and 3D patient-derived cellular models for high throughput drug screening. Sci Rep. 2021;11:1439. doi: 10.1038/s41598-021-81129-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lucendo-Villarin B, Meseguer-Ripolles J, Drew J, Fischer L, Ma WSE, Flint O, Simpson K, Machesky L, Mountford J, Hay D. Development of a cost effective automated platform to produce human liver spheroids for basic and applied research. Biofabrication. 2020 doi: 10.1088/1758-5090/abbdb2. [DOI] [PubMed] [Google Scholar]
- 88.Broutier L, Andersson-Rolf A, Hindley CJ, Boj SF, Clevers H, Koo BK, Huch M. Culture and establishment of self-renewing human and mouse adult liver and pancreas 3D organoids and their genetic manipulation. Nat Protoc. 2016;11:1724–1743. doi: 10.1038/nprot.2016.097. [DOI] [PubMed] [Google Scholar]
- 89.Matano M, Date S, Shimokawa M, Takano A, Fujii M, Ohta Y, Watanabe T, Kanai T, Sato T. Modeling colorectal cancer using CRISPR-Cas9-mediated engineering of human intestinal organoids. Nat Med. 2015;21:256–262. doi: 10.1038/nm.3802. [DOI] [PubMed] [Google Scholar]
- 90.Freedman BS, Brooks CR, Lam AQ, Fu H, Morizane R, Agrawal V, Saad AF, Li MK, Hughes MR, Werff RV, Peters DT, Lu J, Baccei A, Siedlecki AM, Valerius MT, Musunuru K, McNagny KM, Steinman TI, Zhou J, Lerou PH, Bonventre JV. Modelling kidney disease with CRISPR-mutant kidney organoids derived from human pluripotent epiblast spheroids. Nat Commun. 2015;6:8715. doi: 10.1038/ncomms9715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Bian S, Repic M, Guo Z, Kavirayani A, Burkard T, Bagley JA, Krauditsch C, Knoblich JA. Genetically engineered cerebral organoids model brain tumor formation. Nat Methods. 2018;15:631–639. doi: 10.1038/s41592-018-0070-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Sun L, Wang Y, Cen J, Ma X, Cui L, Qiu Z, Zhang Z, Li H, Yang RZ, Wang C, Chen X, Wang L, Ye Y, Zhang H, Pan G, Kang JS, Ji Y, Zheng YW, Zheng S, Hui L. Modelling liver cancer initiation with organoids derived from directly reprogrammed human hepatocytes. Nat Cell Biol. 2019;21:1015–1026. doi: 10.1038/s41556-019-0359-5. [DOI] [PubMed] [Google Scholar]
- 93.Schwank G, Koo BK, Sasselli V, Dekkers JF, Heo I, Demircan T, Sasaki N, Boymans S, Cuppen E, van der Ent CK, Nieuwenhuis EE, Beekman JM, Clevers H. Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell. 2013;13:653–658. doi: 10.1016/j.stem.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 94.Deng WL, Gao ML, Lei XL, Lv JN, Zhao H, He KW, Xia XX, Li LY, Chen YC, Li YP, Pan D, Xue T, Jin ZB. Gene Correction Reverses Ciliopathy and Photoreceptor Loss in iPSC-Derived Retinal Organoids from Retinitis Pigmentosa Patients. Stem Cell Reports. 2018;10:1267–1281. doi: 10.1016/j.stemcr.2018.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Artegiani B, Hendriks D, Beumer J, Kok R, Zheng X, Joore I, Chuva de Sousa Lopes S, van Zon J, Tans S, Clevers H. Fast and efficient generation of knock-in human organoids using homology-independent CRISPR-Cas9 precision genome editing. Nat Cell Biol. 2020;22:321–331. doi: 10.1038/s41556-020-0472-5. [DOI] [PubMed] [Google Scholar]
- 96.Maepa MB, Jacobs R, van den Berg F, Arbuthnot P. Recent developments with advancing gene therapy to treat chronic infection with hepatitis B virus. Curr Opin HIV AIDS. 2020;15:200–207. doi: 10.1097/COH.0000000000000623. [DOI] [PubMed] [Google Scholar]
- 97.Mohammadzadeh I, Qujeq D, Yousefi T, Ferns GA, Maniati M, Vaghari-Tabari M. CRISPR/Cas9 gene editing: A new therapeutic approach in the treatment of infection and autoimmunity. IUBMB Life. 2020;72:1603–1621. doi: 10.1002/iub.2296. [DOI] [PubMed] [Google Scholar]
- 98.Ni Y, Lempp FA, Mehrle S, Nkongolo S, Kaufman C, Fälth M, Stindt J, Königer C, Nassal M, Kubitz R, Sültmann H, Urban S. Hepatitis B and D viruses exploit sodium taurocholate co-transporting polypeptide for species-specific entry into hepatocytes. Gastroenterology. 2014;146:1070–1083. doi: 10.1053/j.gastro.2013.12.024. [DOI] [PubMed] [Google Scholar]
- 99.Li J, Zong L, Sureau C, Barker L, Wands JR, Tong S. Unusual Features of Sodium Taurocholate Cotransporting Polypeptide as a Hepatitis B Virus Receptor. J Virol. 2016;90:8302–8313. doi: 10.1128/JVI.01153-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Zhang X, Lin SM, Chen TY, Liu M, Ye F, Chen YR, Shi L, He YL, Wu LX, Zheng SQ, Zhao YR, Zhang SL. Asialoglycoprotein receptor interacts with the preS1 domain of hepatitis B virus in vivo and in vitro. Arch Virol. 2011;156:637–645. doi: 10.1007/s00705-010-0903-x. [DOI] [PubMed] [Google Scholar]
- 101.Owada T, Matsubayashi K, Sakata H, Ihara H, Sato S, Ikebuchi K, Kato T, Azuma H, Ikeda H. Interaction between desialylated hepatitis B virus and asialoglycoprotein receptor on hepatocytes may be indispensable for viral binding and entry. J Viral Hepat. 2006;13:11–18. doi: 10.1111/j.1365-2893.2005.00648.x. [DOI] [PubMed] [Google Scholar]
- 102.Pettinato G, Lehoux S, Ramanathan R, Salem MM, He LX, Muse O, Flaumenhaft R, Thompson MT, Rouse EA, Cummings RD, Wen X, Fisher RA. Generation of fully functional hepatocyte-like organoids from human induced pluripotent stem cells mixed with Endothelial Cells. Sci Rep. 2019;9:8920. doi: 10.1038/s41598-019-45514-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Prestigiacomo V, Weston A, Messner S, Lampart F, Suter-Dick L. Pro-fibrotic compounds induce stellate cell activation, ECM-remodelling and Nrf2 activation in a human 3D-multicellular model of liver fibrosis. PLoS One. 2017;12:e0179995. doi: 10.1371/journal.pone.0179995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Sendi H, Mead I, Wan M, Mehrab-Mohseni M, Koch K, Atala A, Bonkovsky HL, Bishop CE. miR-122 inhibition in a human liver organoid model leads to liver inflammation, necrosis, steatofibrosis and dysregulated insulin signaling. PLoS One. 2018;13:e0200847. doi: 10.1371/journal.pone.0200847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Asai A, Aihara E, Watson C, Mourya R, Mizuochi T, Shivakumar P, Phelan K, Mayhew C, Helmrath M, Takebe T, Wells J, Bezerra JA. Paracrine signals regulate human liver organoid maturation from induced pluripotent stem cells. Development. 2017;144:1056–1064. doi: 10.1242/dev.142794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Ardalani H, Sengupta S, Harms V, Vickerman V, Thomson JA, Murphy WL. 3-D culture and endothelial cells improve maturity of human pluripotent stem cell-derived hepatocytes. Acta Biomater. 2019;95:371–381. doi: 10.1016/j.actbio.2019.07.047. [DOI] [PubMed] [Google Scholar]
- 107.Artegiani B, van Voorthuijsen L, Lindeboom RGH, Seinstra D, Heo I, Tapia P, López-Iglesias C, Postrach D, Dayton T, Oka R, Hu H, van Boxtel R, van Es JH, Offerhaus J, Peters PJ, van Rheenen J, Vermeulen M, Clevers H. Probing the Tumor Suppressor Function of BAP1 in CRISPR-Engineered Human Liver Organoids. Cell Stem Cell. 2019;24:927–943.e6. doi: 10.1016/j.stem.2019.04.017. [DOI] [PubMed] [Google Scholar]