Abstract
Background
Multiple oral insulin-sensitizing agents, such as metformin, thiazolidinediones, inositols, and berberine, have been proven safe and efficacious in improving the endocrine, metabolic, and reproductive abnormalities seen in polycystic ovary syndrome (PCOS), providing more options for healthcare providers and patients. These oral insulin sensitizers are more convenient, practical, and economic than agents that need to be injected. A comparison of the clinical effectiveness of the four different classes of oral insulin sensitizers in PCOS has not been explored, leading to clinical uncertainty about the optimal treatment pathway. The present study aims to compare the effects of oral insulin sensitizers on endocrine and metabolic profiles in women with PCOS.
Methods
We identified randomized controlled trials for PCOS from a variety of databases, published from January 2005 to October 2020. Outcomes included changes in menstrual frequency, improvements in hyperandrogenism and glucolipid metabolism and adverse side effects. A random-effects network meta-analysis was performed.
Results
Twenty-two trials comprising 1079 patients with PCOS were included in this study. Compared with metformin, treatment with myo-inositol + d-chiro-inositol was associated with a greater improvement in menstrual frequency (odds ratio 14.70 [95% confidence interval (CI) 2.31–93.58]). Myo-inositol + d-chiro-inositol and metformin + thiazolidinediones combination therapies were superior to respective monotherapies in reducing total testosterone levels. Thiazolidinediones, metformin + thiazolidinediones, and myo-inositol + d-chiro-inositol were associated with a lower insulin resistance index (HOMA-IR) compared with that in metformin alone (mean differences: − 0.72 [95% CI (− 1.11)–(− 0.34)] to − 0.89 [95% CI (− 1.460)–(− 0.32)]). Metformin + thiazolidinediones treatment was associated with lower triglyceride levels compared with that in metformin and thiazolidinediones monotherapy, while thiazolidinediones was superior to metformin in increasing high-density lipoprotein cholesterol and decreasing fasting plasma glucose, triglycerides, low-density lipoprotein cholesterol, and gastrointestinal adverse events.
Conclusions
Ours is the first study to report that for women with PCOS, myo-inositol combined with d-chiro-inositol and metformin combined with thiazolidinediones appear superior to metformin alone in improving insulin resistance and decreasing total testosterone. Myo-inositol combined with d-chiro-inositol is particularly efficacious in menstrual recovery. Thiazolidinediones and metformin combined with thiazolidinediones improve lipid metabolism better than metformin alone.
Trial registration PROSPERO CRD42020211524
Supplementary Information
The online version contains supplementary material available at 10.1186/s12978-021-01207-7.
Keywords: Polycystic ovary syndrome, Oral insulin sensitizers, Metformin, Thiazolidinediones, Inositol, Berberine, Endocrine, Metabolic, Network meta-analysis
Plain English summary
This study aimed to compare the effects of oral insulin sensitizers on endocrine and metabolic profiles in women with polycystic ovary syndrome (PCOS). A random-effects network meta-analysis including 22 trials was conducted. For women with PCOS, myo-inositol combined with d-chiro-inositol and metformin combined with thiazolidinediones appear superior to metformin alone in improving insulin resistance and decreasing total testosterone level. Myo-inositol combined with d-chiro-inositol is particularly efficacious in menstrual recovery. Thiazolidinediones and metformin combined with thiazolidinediones improve lipid metabolism better than metformin alone.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12978-021-01207-7.
Background
Polycystic ovary syndrome (PCOS) is a common and complex endocrinopathy that affects 4–21% of women of reproductive age worldwide [1, 2]. It is characterized by ovulatory dysfunction, hyperandrogenism, and a polycystic ovarian morphology. These features are accompanied by various metabolic abnormalities, such as insulin resistance, hyperinsulinemia, and adiposity. PCOS has a long-term impact on overall health, such as an increased risk of endometrial cancer, type 2 diabetes mellitus, and cardiovascular events [3, 4]. Metformin (Met), a recognized insulin sensitizer, has been widely used for women with PCOS due to benefits, such as improving menstruation and hyperinsulinemia, hyperandrogenism, and abnormal metabolism, and it may also have a preventive effect on long-term cardiovascular diseases [5]. However, the persistent use of Met is accompanied by gastrointestinal adverse side effects, such as diarrhea and stomachache [6]. Multiple oral insulin-sensitizing agents, such as thiazolidinediones (TZDs), inositols, and berberine, have been proven safe and efficacious in improving the endocrine, metabolic, and reproductive abnormalities seen in PCOS, providing more options for healthcare providers and patients. These oral insulin sensitizers are more convenient, practical, and economic than agents that need to be injected. However, a comparison of the clinical effectiveness of the four different classes of oral insulin sensitizers in PCOS has not been explored, leading to clinical uncertainty about the optimal treatment pathway. Here, a network meta-analysis (NMA) was designed using metformin as control to compare the efficacy of the four different classes of oral insulin sensitizers in improving menstruation, hyperandrogenism, and abnormal metabolism in patients with PCOS, along with an assessment of their relative safety profiles.
Materials and methods
The methods and results are reported following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement [7]. The protocol was registered at PROSPERO as CRD42020211524.
Search strategy and selection criteria
Systematic literature searches were performed in the following databases from January 2005 to October 23rd, 2020: PubMed, Embase, Web of Science, the Cochrane Central Register of Controlled Trial (CENTRAL), CBM Database, CNKI Database, the WanFang Database, the WeiPu Database, and ClinicalTrials.gov. We used different combinations of the following search terms: “polycystic ovary syndrome,” “metformin,” “thiazolidinediones,” “inositol,” and “berberine.” The PICO framework was used to identify relevant trials [8], without language, ethnicity, or regional restrictions. We included randomized controlled trials (RCTs) in women, aged 18 to 49 years, diagnosed with PCOS based on the Rotterdam consensus, the Androgen Excess and PCOS Society criteria, the National Institute of Health, and Guidelines for diagnosis and treatment of PCOS in China [9–12]. Eligible trials included those where treatments were followed-up for at least 12 weeks, consisting at least one of the predefined outcomes namely, menstruation, hyperandrogenism, and metabolism, and comparison of the effects of insulin sensitizers. Additionally, treatments combined with either oral contraceptives or ovulation-inducing agents, and patients with other diseases, such as nonclassic congenital adrenal hyperplasia, premature ovarian failure, hyperprolactinemia, hypothyroidism, hyperthyroidism, Cushing’s syndrome, and androgen-secreting tumors were excluded.
Outcomes
The outcomes included menstruation frequency, parameters of hyperandrogenism [total testosterone (TT), sex hormone-binding globulin (SHBG), androstenedione (AND), and the modified Ferriman–Gallwey score (mF-G score)], parameters of glucolipid metabolism, including fasting plasma glucose (FPG), fasting insulin (FINS), Homeostatic Model Assessment of Insulin Resistance (HOMA-IR), and lipids [triglyceride (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), and low-density lipoprotein cholesterol (LDL-C)]. Obesity-related indexes, such as body mass index (BMI) and waist–hip ratio (WHR), and adverse events were also considered.
Data extraction and quality assessment
Two independent reviewers (H.Z. and C.X.) screened the titles and abstracts and assessed the full texts of potential reports. The data extracted from the original trials included study, year, region, intervention, sample size, follow-up duration, and outcomes of interest (Table 1). Risk of bias assessment was assigned by two independent investigators (H.Z. and J.Z.) using the revised Cochrane Collaboration risk of bias tool RoB 2.0 across five domains [35]. Each trial was evaluated by two reviewers (H.Z. and C.X.), any discrepancies were resolved by a consensus-based discussion with another author (B.H.).
Table 1.
Characteristics of studies included in the network meta-analysis
| Study | Year | Region | Drugs | Size | F-up (wks) | Efficacy | |||
|---|---|---|---|---|---|---|---|---|---|
| Ahmad [13] | 2008 | India | Met vs TZDs | Met | 850 mg | bid | 31 | 24 w | FPG, FINS, HOMA-IR, BMI, WHR, TT, AND, mF-G score, Menstrual frequency, Adverse events |
| Rosi | 2 mg | bid | 30 | ||||||
| Mohiyididden [14] | 2013 | UK | Met | 500 mg | bid | 17 | 12 w | FPG, FINS, TC, TG, HDL, LDL, BMI, TT, SHBG, Menstrual frequency, Adverse events | |
| Rosi | 4 mg | qd | 18 | ||||||
| Yilmaz [15] | 2005 | Turkey | Met | 850 mg | bid | 25 | 12 w | BMI, WHR, AND, HOMA-IR, TC, TG, LDL, HDL, Menstrual frequency, Adverse events | |
| Rosi | 4 mg | qd | 25 | ||||||
| Sangeeta [16] | 2012 | India | Met | 500 mg | bid | 50 | 24 w | FINS, HOMA-IR, TC, HDL, mF-G score, TT, SHBG, Menstrual frequency, Adverse events | |
| Pio | 15 mg | qd | 50 | ||||||
| Naka [17] | 2011 | Greece | Met | 850 mg | bid | 15 | 24 w | FPG, FINS, TC, TG, HDL, LDL, mF-G score, TT, SHBG, BMI, WHR, Adverse events | |
| Pio | 30 mg | qd | 14 | ||||||
| Jensterl [18] | 2008 | Slovenia | Met | 850 mg | bid | 15 | 24 w | FPG, FINS, HOMA-IR, TC, TG, HDL, LDL, BMI, TT, AND, Menstrual frequency, Adverse events | |
| Rosi | 4 mg | qd | 11 | ||||||
| Ortega [19] | 2005 | Mexico | Met | 850 mg | tid | 18 | 24 w | FPG, FINS, HOMA-IR, TC, TG, HDL, LDL, BMI, WHR, mF-G score, AND, Adverse events | |
| Pio | 30 mg | qd | 17 | ||||||
| Zeng [20] | 2020 | China | Met vs Met + TZDs | Met | 500 mg | tid | 44 | 12 w | FPG, FINS, HOMA-IR, TC, TG, HDL, LDL, BMI, TT, Adverse events |
| Met + Pio | 500 mg + 15 mg | bid | 44 | ||||||
| Wang X [21] | 2014 | China | Met | 1000 mg | tid | 43 | 24 w | FPG, FINS, HOMA-IR, HDL, LDL, BMI, WHR, TT, SHBG, mF-G score, Menstrual frequency, Adverse events | |
| Met + Pio | 1000 mg + 5 mg | tid | 43 | ||||||
| Liang [22] | 2019 | China | Met vs TZDs vs Met + TZDs | Met | 500 mg | tid | 22 | 12 w | FPG, FINS, HOMA-IR, HDL, LDL, TC, TG, BMI, WHR, TT, Menstrual frequency |
| Pio | 30 mg | tid | 21 | ||||||
| Met + Pio | 500 mg + 30 mg | tid | 23 | ||||||
| Sohrevardi [23] | 2016 | Iran | Met | 500 mg | tid | 22 | 12 w | PFG, FINS, HOMA-IR, TC, TG, HDL, LDL, BMI, TT, WHR, Menstrual frequency, Adverse events | |
| Pio | 30 mg | qd | 21 | ||||||
| Met + Pio | 500 mg + 30 mg | tid/qd | 23 | ||||||
| Jamilian [24] | 2017 | Iran | Met vs MI | Met | 500 mg | tid | 30 | 12 w | BMI, SHBG, TT, mF-G score |
| MI | 2 g | bid | 30 | ||||||
| Shokrpour [25] | 2019 | Iran | Met | 500 mg | tid | 27 | 12 w | FPG, FINS, HOMA-IR, BMI, TC, TG, HDL-C, LDL-C | |
| MI | 2 g | bid | 26 | ||||||
| Fruzzetti [26] | 2016 | Italy | Met | 500 mg | tid | 22 | 24 w | HOMA-IR, BMI, mF-G score, Menstrual frequency, Adverse events | |
| MI | 4 g | qd | 24 | ||||||
| Nehra [27] | 2017 | India | Met | 500 mg | tid | 30 | 12–24 w | FPG, FINS, HOMA-IR, TC, TG, HDL, LDL, TT, BMI, WHR | |
| MI | 1 g | bid | 30 | ||||||
| Du [28] | 2018 | China | Met vs MI + DCI | Met | 500 mg | bid | 32 | 24 w | FPG, FINS, HOMA-IR, TC, TG, HDL, LDL, AND, TT, SHBG, Menstrual frequency, Adverse events |
| MI + DCI | 550 mg + 13.8 mg | bid | 32 | ||||||
| Pizzo [29] | 2014 | Italy | MI vs DCI | MI | 4 g | qd | 25 | 24 w | HOMA-IR, BMI, mF-G score, TT, AND, SHGB, Menstrual frequency |
| DCI | 1 g | qd | 25 | ||||||
| Donne [30] | 2019 | Italy | MI vs MI + DCI | MI | 4 g | qd | 10 | 12–24 w | mF-G score, WHR, BMI, Menstrual frequency |
| MI + DCI | 1.1 g + 27.6 mg | qd | 12 | ||||||
| Nordio [31] | 2012 | Italy | MI | 2 g | bid | 24 | 12–24 w | BMI, WHR, FPG, FINS, HOMA-IR, TT, SHBG, AND, Menstrual frequency | |
| MI + DCI | 550 mg + 13.8 mg | bid | 26 | ||||||
| Li [32] | 2017 | China | Met vs BBR | Met | 500 mg | bid | 29 | 12 w | FPG, FINS, HOMA-IR, TC, TG, HDL, LDL, BMI, TT, Adverse events |
| BBR | 300 mg | tid | 26 | ||||||
| Wang P [33] | 2016 | China | Met vs Met + BBR | Met | 500 mg | tid | 42 | 12 w | HOMA-IR, BMI, WHR |
| Met + BBR | 500 mg + 500 mg | tid | 42 | ||||||
| Wang L [34] | 2011 | China | Met | 500 mg | tid | 28 | 12 w | FPG, FINS, HOMA-IR, BMI, TT | |
| Met + BBR | 500 mg + 500 mg | tid | 28 | ||||||
Met Metformin, TZDs thiazolidinediones, Rosi rosiglitazone, Pio pioglitazone, MI myo-inositol, DCI d-chiro-inositol, BBR berberine, TT total testosterone, SHBG sex hormone binding globulin, AND androstenedione, mF-G score modified Ferriman–Gallwey score, BMI body mass index, WHR waist–hip ratio, FPG fasting plasma glucose, FINS fasting insulin, HOMA-IR Homeostatic Model Assessment of Insulin Resistance, TG triglyceride, TC total cholesterol, HDL-C high density lipoprotein cholesterol, LDL-C low density lipoprotein cholesterol
Data synthesis and analyses
The efficacy of different treatment regimens was compared simultaneously using a traditional pairwise meta-analysis (TMA) and network meta-analysis (NMA) [36]. Initially, we conducted a random-effect TMA using Review Manager 5.4 (the Cochrane Collaboration, London, UK). Continuous variables were represented by the weighted mean differences (MDs) with 95% confidence intervals (CIs), and for dichotomous variables, we calculated the combined odds ratios (ORs) with 95% CIs [37]. Between-study heterogeneity was determined using Chi-squared test, combining I2 and P values, for which I2 > 50% or P < 0.05, indicated substantial heterogeneity. The random-effect NMA was subsequently performed to combine the direct and indirect comparisons of agents into one analysis with Stata software (version 15.1, Stata Corp LLC, 4905 Lakeway Drive, College Station, TX, USA). We calculated the pooled estimates of MDs or ORs with 95% CIs in order to compare multiple interventions to each other. Network inconsistency was evaluated using the node-splitting method and inconsistency models, and significance was assessed at the 0.05 level. Finally, to summarize the probabilities, we calculated the surface under the cumulative ranking (SUCRA) curve to provide a summary statistic for cumulative ranking. The efficacy of each intervention was expressed as a percentage [38]. We conducted comparison-adjusted funnel plots using Stata software to examine the publication bias.
Results
Study search and study characteristics
A total of 8645 publications were retrieved; of these, 22 RCTs (n = 1079 participants) were included in the present network meta-analysis. A flow chart depicting the literature search process based on PICO is presented in Fig. 1. Overall, two studies evaluated and compared myo-inositol (MI) and myo-inositol + d-chiro-inositol (MI + DCI) combination treatment (72 women), one study compared MI and DCI (50 women), and the remaining RCTs offered a comparison of Met (19 trials; 455 women) and the following interventions: thiazolidinediones (TZDs) (ten trials; 280 women), Met + TZDs (three trials; 89 women), MI (four trials; 110 women), MI + DCI (one trial; 32 women), berberine (BBR) (one trial; 26 women), and Met + BBR (two trials; 70 women). The evidence map for the aforementioned interventions is shown in Fig. 2.
Fig. 1.
Prisma flow diagram of the study selection process. RCT randomized controlled trial
Fig. 2.

Evidence graph of all agents. The size of the circles is proportional to sample size, and the width of the lines is proportional to the number of trials. Met Metformin, TZDs thiazolidinediones, MI myo-inositol, DCI d-chiro-inositol, BBR Berberine
Table 1 shows the characteristics of the included studies. These RCTs were conducted in various countries, published in English or Chinese, and participants were recruited from an outpatient clinic or a hospital. Trials were generally similar with respect to patient baseline characteristics for most outcomes. In summary, a total of 1079 women with PCOS were randomized to receive eight different interventions (Met, TZDs, BBR, MI, DCI, MI + DCI, Met + TZDs, and Met + BBR).
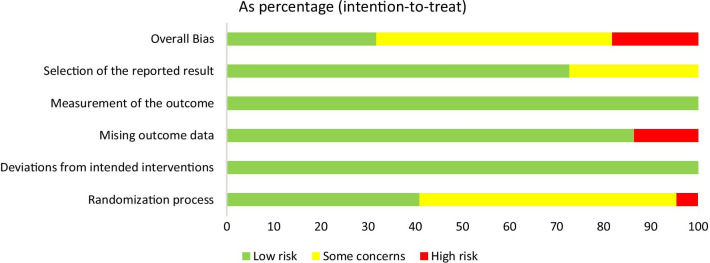
The risk of bias assessment is shown in Fig. 3. Of the 22 included trials, seven were at low risk of bias across all domains, four studies were at a high risk of bias, and we judged the remaining eleven trials to have some concerns. Overall, concerns regarding the randomization process and missing outcome data were the main cause of potential bias. The comparison-adjusted funnel plot for outcomes appeared minor asymmetry or no publication bias (shown in Additional file 1: Appendix S1.1). In addition, the analyses of inconsistency were identified in the network meta-analysis of triglyceride, and the inconsistency model was fitted in the network meta-analysis, the detailed results of the assessment for inconsistency are shown in Additional file 1: Appendix S1.
Fig. 3.
Risk of bias assessment in the RCTs
Efficacy outcomes
Table 2A, B summarizes the NMA and TMA results for positive efficacy outcomes. Furthermore, other efficacy outcomes, details of the Forest plots, and SUCRA curves are presented in Additional file 1: Appendix S2.
Table 2.
(A) NMA and TMA results for menstrual frequency, total testosterone, BMI, and glycometabolism; (B) NMA and TMA results for lipid levels and gastrointestinal adverse events
| Outcomes | Studies | Participants | Traditional pairwise meta-analysis (TMA) | Network meta-analysis (NMA) | ||
|---|---|---|---|---|---|---|
| Heterogeneity | Effect estimate (95% CI) | Studies | Effect estimate (95% CI) | |||
| (A) | ||||||
| Menstrual frequency | ||||||
| TZDs vs Met | 6 | 299 | (P = 0.002); I2 = 74% | 1.17 [0.43, 3.17] | 7 | 1.20 [0.52, 2.76] |
| TZDs + Met vs Met | 3 | 167 | (P = 0.88); I2 = 0% | 3.91 [1.75, 8.72] | 3 | 2.41 [0.75, 7.71] |
| MI vs Met | 1 | 46 | Not applicable | 1.11 [0.24, 5.11] | 4 | 0.89 [0.14, 5.56] |
| MI + DCI vs Met | 1 | 64 | Not applicable | 11.67 [2.37, 57.36] | 3 | 14.70 [2.31, 93.58] |
| MI + DCI vs MI | 2 | 44 | (P = 0.38); I2 = 0% | 21.92 [2.99, 160.44] | 3 | 16.51 [2.56, 106.64] |
| TT | ||||||
| TZDs vs Met | 7 | 322 | (P = 0.22); I2 = 27% | 5.27 [0.98, 9.55] | 8 | 3.90 [1.10, 6.71] |
| TZDs + Met vs Met | 4 | 307 | (P = 0.44); I2 = 0% | − 3.14 [− 6.19, − 0.09] | 4 | − 3.66 [− 6.60, − 0.72] |
| MI vs Met | 2 | 120 | (P = 0.66); I2 = 0% | 0.31 [− 0.69, 1.32] | 6 | 0.37 [− 0.63, 1.37] |
| MI + DCI vs Met | 1 | 64 | Not applicable | − 5.48 [− 10.27, − 0.69] | 3 | − 6.72 [− 10.24, − 3.20] |
| BBR + Met vs Met | 2 | 140 | (P = 1.00); I2 = 0% | − 11.53 [− 16.91, − 6.15] | 2 | − 11.53 [− 16.91, − 6.15] |
| MI + DCI vs MI | 2 | 72 | (P = 0.63); I2 = 0% | − 8.50 [− 13.60, − 3.40] | 3 | − 7.09 [− 10.62, − 3.56] |
| BMI | ||||||
| TZDs vs Met | 8 | 322 | (P = 0.42); I2 = 1% | 1.26 [0.78, 1.75] | 7 | 1.23 [0.75, 1.70] |
| TZDs + Met vs Met | 4 | 171 | (P = 0.68); I2 = 0% | − 0.03 [− 1.02, 0.95] | 3 | 0.22 [− 0.71, 1.14] |
| MI vs Met | 5 | 279 | (P = 0.47); I2 = 0% | 0.28 [0.06, 0.50] | 8 | 0.29 [0.09, 0.49] |
| MI + DCI vs Met | / | / | / | / | 3 | − 0.19 [− 1.61, 1.23] |
| BBR + Met vs Met | 2 | 140 | (P = 0.32); I2 = 0% | − 1.85 [− 2.76, − 0.94] | 2 | − 1.85 [− 2.76, − 0.94] |
| MI + DCI vs MI | 3 | 80 | (P = 0.98); I2 = 0% | − 0.48 [− 1.89, 0.93] | 3 | − 0.48 [− 1.89, 0.93] |
| FPG | ||||||
| TZD vs Met | 7 | 272 | (P < 0.00001); I2 = 86% | − 0.10 [− 0.21, 0.01] | 5 | − 0.12 [− 0.21, − 0.02] |
| TZD + Met vs Met | 5 | 307 | (P = 0.79); I2 = 0% | − 0.05 [− 0.15, 0.06] | 5 | 0.00 [− 0.14, 0.15] |
| MI vs Met | 3 | 173 | (P = 0.22); I2 = 34% | − 0.05 [− 0.12, 0.02] | 5 | − 0.05 [− 0.19, 0.09] |
| MI + DCI vs Met | 1 | 64 | Not applicable | − 0.09 [− 0.17, − 0.01] | 6 | − 0.18 [− 0.37, 0.02] |
| MI + DCI vs MI | 2 | 72 | (P = 0.22); I2 = 35% | − 0.25 [− 0.56, 0.05] | 3 | − 0.13 [− 0.33, 0.08] |
| FINS | ||||||
| TZDs vs Met | 8 | 357 | (P < 0.00001); I2 = 97% | − 2.50 [− 6.23, 1.23] | 7 | − 2.38 [− 4.94, 0.19] |
| TZDs + Met vs Met | 4 | 259 | (P = 0.19); I2 = 37% | − 1.83 [− 3.12, − 0.55] | 4 | − 2.56 [− 6.03, 0.92] |
| MI vs Met | 3 | 173 | (P = 0.23); I2 = 32% | − 0.22 [− 0.75, 0.32] | 5 | − 0.40 [− 4.08, 3.27] |
| MI + DCI vs Met | 1 | 64 | Not applicable | − 1.37 [− 1.56, − 1.18] | 3 | − 1.38 [− 6.14, 3.38] |
| MI + DCI vs MI | 2 | 72 | (P = 0.36); I2 = 0% | − 0.76 [− 1.94, 0.42] | 3 | − 0.98 [− 5.28, 3.33] |
| HOMA-IR | ||||||
| TZDs vs Met | 6 | 317 | (P < 0.00001); I2 = 97% | − 0.92 [− 1.64, − 0.19] | 4 | − 0.72 [− 1.11, − 0.34] |
| TZDs + Met vs Met | 4 | 259 | (P = 0.04); I2 = 65% | − 0.85 [− 1.21, − 0.49] | 4 | − 0.86 [− 1.29, − 0.43] |
| MI vs Met | 4 | 219 | (P = 0.004); I2 = 78% | − 0.20 [− 0.42, 0.01] | 6 | − 0.28 [− 0.66, 0.10] |
| MI + DCI vs Met | 1 | 64 | Not applicable | − 1.15 [− 1.25, − 1.05] | 3 | − 0.89 [− 1.46, − 0.32] |
| BBR + Met vs Met | 2 | 140 | (P = 0.37); I2 = 0% | − 0.25 [− 0.36, − 0.14] | 2 | − 0.25 [− 0.81, 0.31] |
| MI + DCI vs MI | 2 | 72 | (P = 0.97); I2 = 0% | − 0.39 [− 0.83, 0.06] | 3 | − 0.61 [− 1.18, − 0.05] |
| (B) | ||||||
| TG | ||||||
| TZDs vs Met | 7 | 261 | (P = 0.0002); I2 = 77% | − 0.01 [− 0.19, 0.16] | 7 | − 0.66 [− 1.00, − 0.32] |
| TZDs + Met vs Met | 3 | 178 | (P = 0.74); I2 = 0% | − 0.24 [− 0.43, − 0.06] | 3 | − 0.08 [− 0.16, − 0.00] |
| MI vs Met | 3 | 173 | (P = 0.60); I2 = 0% | − 0.03 [− 0.06, 0.00] | 3 | 0.14 [0.07, 0.21] |
| MI + DCI vs Met | 1 | 64 | Not applicable | − 0.08 [− 0.16, − 0.00] | 4 | 0.21 [− 0.26, 0.68] |
| TZDs + Met vs TZDs | 2 | 88 | (P = 0.10); I2 = 0% | 0.15 [− 0.18, 0.48] | 3 | − 0.51 [− 0.88, − 0.14] |
| TC | ||||||
| TZDs vs Met | 8 | 346 | (P < 0.00001); I2 = 93% | − 0.06 [− 0.41, 0.29] | 8 | − 0.18 [− 0.46, 0.10] |
| TZDs + Met vs Met | 3 | 178 | (P = 0.98); I2 = 0% | − 0.30 [− 0.53, − 0.07] | 3 | − 0.15 [− 0.57, 0.27] |
| MI vs Met | 3 | 173 | (P = 0.67); I2 = 0% | 0.03 [− 0.01, 0.07] | 3 | 0.19 [− 0.29, 0.66] |
| MI + DCI vs Met | 1 | 64 | Not applicable | − 0.07 [− 0.16, 0.02] | 4 | − 0.07 [− 0.69, 0.55] |
| HDL | ||||||
| TZDs vs Met | 8 | 346 | (P < 0.00001); I2 = 96% | 0.14 [− 0.03, 0.30] | 8 | 0.13 [0.03, 0.24] |
| TZDs + Met vs Met | 4 | 259 | (P = 0.53); I2 = 0% | − 0.00 [− 0.09, 0.09] | 4 | 0.05 [− 0.10, 0.20] |
| MI vs Met | 3 | 173 | (P = 0.10); I2 = 57% | 0.05 [0.03, 0.07] | 3 | 0.02 [− 0.14, 0.17] |
| MI + DCI vs Met | 1 | 64 | Not applicable | 0.00 [− 0.11, 0.11] | 4 | 0.00 [− 0.28, 0.28] |
| LDL | ||||||
| TZDs vs Met | 7 | 261 | (P = 0.02); I2 = 59% | − 0.11 [− 0.29, 0.08] | 7 | − 0.19 [− 0.27, − 0.11] |
| TZDs + Met vs Met | 4 | 259 | (P = 0.96); I2 = 0% | − 0.10 [− 0.25, 0.05] | 4 | − 0.08 [− 0.24, 0.07] |
| MI vs Met | 3 | 173 | (P = 0.84); I2 = 0% | 0.01 [− 0.03, 0.05] | 3 | 0.01 [− 0.03, 0.05] |
| MI + DCI vs Met | 1 | 64 | Not applicable | − 0.07 [− 0.21, 0.07] | 4 | − 0.07 [− 0.21, 0.07] |
| Gastrointestinal adverse events | ||||||
| TZDs vs Met | 6 | 253 | (P = 0.88); I2 = 0% | 0.11 [0.03, 0.41] | 6 | 0.13 [0.04, 0.46] |
| TZDs + Met vs Met | 3 | 237 | (P = 0.33); I2 = 11% | 0.67 [0.23, 1.93] | 3 | 0.76 [0.25, 2.27] |
| MI vs Met | 1 | 50 | Not applicable | 0.13 [0.01, 2.58] | 1 | 0.13 [0.01, 2.58] |
| MI + DCI vs Met | 1 | 64 | Not applicable | 0.08 [0.00, 1.45] | 1 | 0.08 [0.00, 1.45] |
| BBR vs Met | 1 | 60 | Not applicable | 3.10 [0.12, 79.23] | 1 | 3.10 [0.12, 79.23] |
Bolded results show statistical significance at the 0.05 level
Met Metformin, TZDs thiazolidinediones, MI myo-inositol, DCI d-chiro-inositol, BBR berberine, TT total testosterone, BMI body mass index, FPG fasting plasma glucose, FINS fasting insulin, HOMA-IR Homeostatic Model Assessment of Insulin Resistance, TG triglyceride, TC total cholesterol, HDL-C high density lipoprotein cholesterol, LDL-C low density lipoprotein cholesterol
Menstrual frequency
With respect to improving menstrual frequency, our TMA showed that Met + TZDs (OR 3.91 [95% CI 1.75–8.72]; P = 0.88, I2 = 0%) was more efficacious than Met alone, while MI + DCI (OR 21.92 [95% CI 2.99–160.44]; P = 0.38, I2 = 0%) was more efficacious than MI alone. The NMA revealed that MI + DCI was more efficacious than Met (OR 14.70 [95% CI 2.31–93.58]) and MI (OR 16.51 [95% CI 2.56–106.64]). MI + DCI was ranked best at improving menstrual frequency.
Hyperandrogenism
In terms of reducing TT, the TMA and NMA both revealed that Met + BBR [(TMA: MD − 11.53 [95% CI (− 16.91)–(− 6.15)]; P = 1.00, I2 = 0%; NMA: MD − 11.53 [95% CI (− 17.62)–(− 5.44)]) and Met + TZDs [(TMA: MD − 3.14 [95% CI (− 6.19)–(− 0.09)]; P = 0.44, I2 = 0%; NMA: MD − 3.66 [95% CI (− 6.60)–(− 0.72)] were superior to Met alone. The NMA revealed that MI + DCI was more efficacious than Met, MI, and DCI [MDs ranging from − 6.72 [95% CI (− 10.24)–(− 3.20)] for Met to − 7.70 [95% CI (− 14.90)–(− 0.50)] for DCI]). Treatment with TZDs was less efficacious than Met (MD 3.90 [95% CI 1.10–6.71]). The SUCRA values were as follows: Met + BBR (92.2%), MI + DCI (75.4%), and Met + TZDs (59.4%). No difference was found in comparisons of SHBG, AND, and mF-G scores.
Obesity
With respect to BMI reduction, the TMA showed that Met + BBR (MD − 1.85 [95% CI (− 2.76)–(− 0.94)]; P = 0.32, I2 = 0%) was superior to Met monotherapy, whereas the NMA revealed that Met + BBR was more efficacious than Met, MI, TZDs, and Met + TZDs (MDs ranging from − 1.85 [95% CI (− 2.76)–(− 0.94)] for Met to − 3.08 [95% CI (− 4.10)–(− 2.05)] for TZDs). Met + BBR ranked best among all the treatments at reducing BMI. However, both the TMA and NMA revealed that there was no significant difference between the groups with respect to WHR reduction.
Glycometabolism
With respect to lowering FPG, the TMA revealed that there were no significant differences between each of the investigated agents. The NMA revealed that TZDs (MD − 0.21 [95% CI (− 0.21)–(− 0.02)]) were more efficacious than Met. Additionally, with respect to lowering FINS, the TMA revealed that Met + TZDs (MD − 1.83 [95% CI (− 3.12)–(− 0.55)]; P = 0.19, I2 = 37%) was more efficacious than Met alone. The NMA did not reveal any significant differences among the groups.
With respect to decrease in HOMA-IR, the TMA showed that TZDs (MD − 0.92 [95% CI (− 1.64)–(− 0.19)]; P < 0.00001, I2 = 97%), Met + TZDs (MD − 0.85 [95% CI (− 1.21)–(− 0.49)]; P = 0.04, I2 = 65%), and Met + BBR (MD − 0.25 [95% CI (− 0.36)–(− 0.14)]; P = 0.37, I2 = 0%) were more efficacious than Met alone. The NMA revealed that TZDs, Met + TZDs, and MI + DCI were all superior to Met (MDs ranging from − 0.72 [95% CI (− 1.11)–(− 0.34)] for TZDs to − 0.89 [95% CI (− 1.46)–(− 0.32)] for MI + DCI), with MI + DCI being ranked the best with a SUCRA value of 80.8%. The SUCRA values for the other agents were as follows: 79.9% and 68.6% for Met + TZDs and TZDs, respectively.
Lipid levels
Parameters assessed for improvement in blood lipids included TG, TC, HDL-C, and LDL-C. In terms of reducing TG and TC levels, the TMA revealed that Met + TZDs (TG: MD − 0.24 [95% CI (− 0.43)–(− 0.06)]; P = 0.74, I2 = 0%; TC: MD − 0.30 [95% CI (− 0.53)–(− 0.07)]; P = 0.98, I2 = 0%) was more efficacious than Met alone. In terms of reducing levels of TG, the NMA showed that Met + TZDs was superior to Met (MD − 0.08 [95% CI (− 0.16)–(0.00)]) and TZDs (MD − 0.51 [95% CI (− 0.88)–(− 0.14)]), and was the best intervention among treatments. The NMA also showed that BBR (MD − 0.03 [95% CI (− 0.06)–(0.00)]) was more efficacious than Met. However, for TC no significant differences were found in the NMA.
Furthermore, the TMA also suggested that MI (MD 0.05 [95% CI 0.03–0.07]; P = 0.10, I2 = 57%) was associated with higher HDL-C than Met. The NMA revealed that treatment with TZDs was superior to Met in increasing HDL-C (MD 0.13 [95% CI 0.03–0.24]) and decreasing LDL-C (MD − 0.19 [95% CI (− 0.27)–(− 0.11)]) and was the best intervention among the different treatments.
Adverse events
In terms of the frequency of gastrointestinal adverse events during treatment, both TMA and NMA revealed that treatment with TZDs (TMA: OR 0.11 [95% CI 0.03–0.41]; P = 0.88, I2 = 0%; NMA: OR 0.13 [95% CI 0.04–0.46]) was inferior to Met. With respect to the incidence of peripheral edema, the TMA revealed that TZDs were more frequent than Met (OR 67.89 [95% CI 3.96, 1163.28]; P&I2 NA), no significant differences were found in the NMA. Furthermore, both the TMA and NMA did not show any significant differences between the different treatment regimens for the incidence rate of muscle spasms, while no drug increased odds of transaminase abnormalities. Of note, these results should be interpreted with caution since results for MI, MI + DCI, and BBR were based on a single trial, and in some of the trials the description of adverse events was subjective and unclear.
Discussion
To our knowledge, this is the first report on an NMA used to assess the efficacy and safety of oral insulin sensitizers (metformin, thiazolidinediones, inositol, and berberine) as an adjunct therapy to improve irregular menses, hyperandrogenism, and glucolipid metabolism abnormalities in women with PCOS. The results obtained are based on 22 trials that included 1079 women, randomly assigned to eight different interventions. Overall, treatment with MI + DCI was associated with the best improvement in menstrual frequency. MI + DCI, Met + TZDs, and Met + BBR were superior to Met for TT reduction, while MI + DCI, Met + TZDs, and TZDs significantly lowered HOMA-IR than Met alone. TZDs were superior to Met in decreasing FPG, TG, LDL-C levels, and increasing HDL-C level, while Met + TZDs was associated with lower TG levels compared to Met and TZDs monotherapy. Furthermore, Met + BBR was more efficacious than Met alone in reducing BMI.
Metformin is a classic insulin sensitizer, that inhibits hepatic glucose production, thereby decreasing glucose levels [39]. Many studies have already demonstrated its ability to improve menstruation frequency, reduce androgen excess, and decrease insulin resistance in PCOS. However, it also often associated with gastrointestinal side effects, such as diarrhea, nausea, and abdominal discomfort [40]. In the current study, in four of the RCTs, a total of 12 women withdrew due to intolerance rising from gastrointestinal side effects [14, 19, 23, 26]. There were other reports of mild stomach discomfort that resolved spontaneously within a few weeks [13, 15, 16, 18, 20, 21, 28].
Thiazolidinediones (TZDs) decrease hepatic and peripheral insulin resistance directly through activation of the nuclear hormone receptor PPARγ and have a well-documented effect of improving hyperglycemia and dyslipidemia. TZDs also improve the menstrual cycle and ovulation and reduce androgen levels in women with PCOS [41, 42]. However, these clinical benefits have largely been ignored due to safety issues and side effects, such as weight gain, peripheral edema, and even heart failure [43]. In our study, one trial with pioglitazone showed 40% of women with mild peripheral edema and 11% with muscle spasms [16], although other adverse events were not found and gastrointestinal adverse events were less frequent with TZDs than Met. Furthermore, compared with that by Met, TZDs had a more beneficial effect on improving glucolipid metabolism; our NMA revealed that treatment with TZDs was associated with lower FPG, TG, LDL-C levels, and higher HDL-C level. The SUCRA analysis revealed that treatment with TZDs was the best (among these treatments) for decreasing LDL-C and increasing HDL-C, indicating its potential ability to reduce a patients’ risk of cardiovascular diseases in PCOS.
Our NMA revealed that the combination of Met + TZDs was more effective than Met alone at improving menstruation frequency, and reducing TT, HOMA-IR, and TG. The SUCRA value showed that Met + TZDs was the best intervention for reducing TG level. The TMA also demonstrated that Met + TZDs was superior to Met monotherapy at reducing FINS and TC level. Our previous NMA in overweight women with PCOS revealed that Met + TZDs was superior to Met in recovering menstrual function, whereas there were no evident differences in TT and FINS [44]. Different inclusion criteria might explain these discrepancies. Additionally, three RCTs reported gastrointestinal discomfort in the Met + TZDs group [20, 21, 23], which was not significantly different than the Met alone group.
Inositol acts as a second messenger with insulin-like functions and is safe and well-tolerated [45]. The two most common isomers of inositol are MI, which has been shown to significantly improve ovulatory function [46], and DCI, which is able to reduce peripheral insulin resistance in patients with PCOS [47]. Our NMA is in line with other pairwise meta-analyses, showing that there is no apparent benefit of MI or DCI alone compared with Met [48–50]. Recent studies have proposed that a combination of both MI and DCI, at a plasma ratio of 40:1, can restore normal hormonal function quicker than MI or DCI alone [51]. This is the first report on the analysis of efficacy of Met and MI + DCI in women with PCOS; it revealed that among all of the agents investigated here MI + DCI appeared to be the best intervention for restoring regular menses and decreasing HOMA-IR, while the combination was also able to reduce TT better than Met. No side effects have been described in clinical studies examining the effect of inositol [26, 44].
Berberine (BBR), a natural isoquinoline alkaloid, has been studied in various randomized clinical trials in patients with PCOS and has been shown to be safe and promising for decreasing insulin resistance, lowering blood lipids, and restoring ovulation [52–55]. In the only previous pairwise meta-analysis that compared Met, BBR, and their combination in insulin resistant patients with PCOS, there were no significant differences between the different treatment groups [56]. In the current study, only one trial that examined BBR and Met was included. As seen from the NMA, BBR was superior to Met in reducing TG. Met + BBR was associated with a greater reduction in TT and BMI than that by Met alone and was the best intervention among the investigated agents tested here. These findings should be interpreted with caution since they are based on data from only two head-to-head trials of low quality [33, 34]. Further high-quality trials to verify these potential favorable effects in PCOS are therefore required. One person in the berberine group withdrew due to gastrointestinal side effects [32].
Strengths and limitations
Our findings reflect the comparative efficacy and safety of monotherapy versus a combination of different oral insulin sensitizers (metformin, thiazolidinediones, inositol, and berberine) in the treatment of PCOS. Previous related meta-analyses have mainly been of pairwise design that focused on individual agents and have given inconsistent results, our NMA incorporated both direct and indirect comparisons of interventions into a single analysis and provided a ranking of the available interventions. Through a comprehensive search and a rigorous review of the design of the randomized clinical trials, we aimed to reduce the likelihood of selection bias. We focused on the analysis of menstrual and ovulation abnormalities, hyperandrogenism, obesity, glucolipid metabolism, and adverse events in women with PCOS to provide a comprehensive reference for its clinical treatment.
However, the present study still has some limitations that must be noted. First, very few literature reports met the inclusion criteria of treatment without contraceptives and/or intervention of ovulation inducing drugs to effectively verify the independent effects of drugs on efficacy. Second, there were markedly different dosages, durations of treatment, and small sample sizes that could contribute to sample bias, selection bias, and high heterogeneity. Third, some of trials did not have a specific analysis of women who dropped out or missed a follow-up.
Conclusions
In conclusion, for women with PCOS, MI combined with DCI and Met combined with TZDs appear to be superior than Met alone in improving insulin resistance and decreasing total testosterone. MI combined with DCI appears to be particularly efficacious in menstrual recovery. TZDs and TZDs combined with Met seem to offer the additional advantage of improving lipid metabolism. However, our findings are limited by the small number and low quality of available studies, and therefore more rigorous and high-level RCTs are needed to further guide the clinical management of women with PCOS, especially those with insulin resistance.
Supplementary Information
Additional file 1: Appendix S1. Details on study methods.
Acknowledgements
Not applicable.
Abbreviations
- PCOS
Polycystic ovary syndrome
- Met
Metformin
- TZDs
Thiazolidinediones
- MI
Myo-inositol
- DCI
D-chiro-inositol
- BBR
Berberine
- TT
Total testosterone
- SHBG
Sex hormone binding globulin
- AND
Androstenedione
- mF-Gscore
Modified Ferriman–Gallwey score
- BMI
Body mass index
- WHR
Waist–hip ratio
- FPG
Fasting plasma glucose
- FINS
Fasting insulin
- HOMA-IR
Homeostatic Model Assessment of Insulin Resistance
- TG
Triglyceride
- TC
Total cholesterol
- HDL-C
High density lipoprotein cholesterol
- LDL-C
Low density lipoprotein cholesterol
Authors’ contributions
HZ, CX, JZ and BH conceived the study. HZ, CX and BH designed the study. HZ wrote the study protocol and registered the study protocol. HZ, CX selected the articles and extracted the data. HZ and CX, JZ, and BH developed the statistical methods and analyzed the data. HZ wrote the first draft of the manuscript. BH critically checked its content and approved its final version. All authors agreed with the results and conclusions of this article. All authors read and approved the final manuscript.
Funding
This work was supported by a grant from the National Natural Science Foundation of China (Grant no. 81570765), and the 345 Talent Project of ShengJing Hospital of China Medical University.
Availability of data and materials
The datasets supporting the conclusions of this article are included within the article and its additional file.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Han Zhao, Email: 1014136426@qq.com.
Chuan Xing, Email: 1148434087@qq.com.
Jiaqi Zhang, Email: 904330413@qq.com.
Bing He, Email: hebing7557@163.com.
References
- 1.Azziz R, Carmina E, Chen Z, Dunaif A, Laven JS, Legro RS, Lizneva D, Natterson-Horowtiz B, Teede HJ, Yildiz BO. Polycystic ovary syndrome. Nat Rev Dis Primers. 2016;267(2):2056–2676. doi: 10.1038/nrdp.2016.57. [DOI] [PubMed] [Google Scholar]
- 2.Lizneva D, Suturina L, Walker W, Brakta S, Gavrilova-Jordan L, Azziz R. Criteria, prevalence, and phenotypes of polycystic ovary syndrome. Fertil Steril. 2016;106(1):6–15. doi: 10.1016/j.fertnstert.2016.05.003. [DOI] [PubMed] [Google Scholar]
- 3.Teede HJ, Misso ML, Costello MF, Dokras A, Laven J, Moran L, Piltonen T, Norman RJ, International PCOS Network Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Fertil Steril. 2018;110(3):364–379. doi: 10.1016/j.fertnstert.2018.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Aversa A, La Vignera S, Rago R, Gambineri A, Nappi RE, Calogero AE, Ferlin A. Fundamental concepts and novel aspects of polycystic ovarian syndrome: expert consensus resolutions. Front Endocrinol (Lausanne) 2020;11:516. doi: 10.3389/fendo.2020.00516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.McCartney CR, Marshall JC. CLINICAL PRACTICE. Polycystic Ovary Syndrome. N Engl J Med. 2016;375(1):54–64. 10.1056/NEJMcp1514916 [DOI] [PMC free article] [PubMed]
- 6.Renato P. Metformin in women with PCOS, Pros. Endocrine. 2015;48(2):422–426. doi: 10.1007/s12020-014-0311-1. [DOI] [PubMed] [Google Scholar]
- 7.Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schardt C, Adams MB, Owens T, Keitz S, Fontelo P. Utilization of the PICO framework to improve searching PubMed for clinical questions. BMC Med Inform Decis Mak. 2007;7:16. doi: 10.1186/1472-6947-7-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS) Hum Reprod. 2004;19(1):41–47. doi: 10.1093/humrep/deh098. [DOI] [PubMed] [Google Scholar]
- 10.Azziz R, Carmina E, Dewailly D, Diamanti-Kandarakis E, Escobar-Morreale HF, Futterweit W, Janssen OE, Legro RS, Norman RJ, Taylor AE, Witchel SF, Androgen Excess Society Positions statement: criteria for defining polycystic ovary syndrome as a predominantly hyperandrogenic syndrome: an Androgen Excess Society guideline. J Clin Endocrinol Metab. 2006;91(11):4237–4245. doi: 10.1210/jc.2006-0178. [DOI] [PubMed] [Google Scholar]
- 11.Chinese Endocrinologist Association. Chinese Medical Doctor Association Endocrinologist consensus on the management of polycystic ovary syndrome. Chin J Endocrinol Metab. 2018;34(1):1–7. doi: 10.3760/cma.j.issn.1000-6699.2018.01.001. [DOI] [Google Scholar]
- 12.Zawadri J. Diagnostic criteria for polycystic ovary syndrome: towards a rational approach. Polycystic ovary syndrome. Current issues in endocrinology and metabolism. 1992.
- 13.Ahmad J, Shukla N, Khan AR, Ahmed F, Siddiqui MA. Comparison of metabolic effects of metformin and rosiglitazone in the management of polycystic ovary syndrome [PCOS]: a prospective, parallel, randomized, open-label study. Diabetes Metab Syndr. 2008;2(1):37–46. doi: 10.1016/j.dsx.2007.12.003. [DOI] [Google Scholar]
- 14.Mohiyiddeen L, Watson AJ, Apostolopoulos NV, Berry R, Alexandraki KI, Jude EB. Effects of low-dose metformin and rosiglitazone on biochemical, clinical, metabolic and biophysical outcomes in polycystic ovary syndrome. J Obstet Gynaecol. 2013;33(2):165–170. doi: 10.3109/01443615.2012.745839. [DOI] [PubMed] [Google Scholar]
- 15.Yilmaz M, Bukan N, Ayvaz G, Karakoç A, Törüner F, Cakir N, Arslan M. The effects of rosiglitazone and metformin on oxidative stress and homocysteine levels in lean patients with polycystic ovary syndrome. Hum Reprod. 2005;20(12):3333–3340. doi: 10.1093/humrep/dei258. [DOI] [PubMed] [Google Scholar]
- 16.Sangeeta S. Metformin and pioglitazone in polycystic ovarian syndrome: a comparative study. J Obstet Gynaecol India. 2012;62(5):551–556. doi: 10.1007/s13224-012-0183-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Naka KK, Kalantaridou SN, Kravariti M, Bechlioulis A, Kazakos N, Calis KA, Makrigiannakis A, Katsouras CS, Chrousos GP, Tsatsoulis A, Michalis LK. Effect of the insulin sensitizers metformin and pioglitazone on endothelial function in young women with polycystic ovary syndrome: a prospective randomized study. Fertil Steril. 2011;95(1):203–209. doi: 10.1016/j.fertnstert.2010.06.058. [DOI] [PubMed] [Google Scholar]
- 18.Jensterle M, Sebestjen M, Janez A, Prezelj J, Kocjan T, Keber I, Pfeifer M. Improvement of endothelial function with metformin and rosiglitazone treatment in women with polycystic ovary syndrome. Eur J Endocrinol. 2008;159(4):399–406. doi: 10.1530/EJE-08-0507. [DOI] [PubMed] [Google Scholar]
- 19.Ortega-González C, Cardoza L, Coutiño B, Hidalgo R, Arteaga-Troncoso G, Parra A. Insulin sensitizing drugs increase the endogenous dopaminergic tone in obese insulin-resistant women with polycystic ovary syndrome. J Endocrinol. 2005;184(1):233–239. doi: 10.1677/joe.1.05844. [DOI] [PubMed] [Google Scholar]
- 20.Zeng H, Huang Y, Wu J, Xie TQ, Liu DK, Huang QL, Chen Z, Liu JP. Effects of pioglitazone hydrochloride and metformin hydrochloride tablets on metabolic parameters and hormones in polycystic ovary syndrome. Prog Obstet Gynecol. 2020;29(11):7–10. doi: 10.13283/j.cnki.xdfckjz.2020.11.031. [DOI] [Google Scholar]
- 21.Wang X, Gao S, Zhang X. Metformin and pioglitazone plus metformin for patients with polycystic ovary syndrome combined with insulin resistance. Cent South Pharm. 2014 doi: 10.7539/j.issn.1672-2981.2014.02.023. [DOI] [Google Scholar]
- 22.Liang N, Zhang Z. Effects of metformin and pioglitazone on sexual hormones and metabolic level in patients with polycystic ovary syndrome. Guangxi Med J. 2019;41:148–152. doi: 10.11675/j.issn.0253-4304.2019.02.02. [DOI] [Google Scholar]
- 23.Sohrevardi SM, Nosouhi F, Hossein Khalilzade S, Kafaie P, Karimi-Zarchi M, Halvaei I, Mohsenzadeh M. Evaluating the effect of insulin sensitizers metformin and pioglitazone alone and in combination on women with polycystic ovary syndrome: an RCT. Int J Reprod Biomed. 2016;14(12):743–754. [PMC free article] [PubMed] [Google Scholar]
- 24.Jamilian M, Farhat P, Foroozanfard F, Afshar Ebrahimi F, Aghadavod E, Bahmani F, Badehnoosh B, Jamilian H, Asemi Z. Comparison of myo-inositol and metforminon clinical, metabolic and genetic parameters in polycystic ovary syndrome: a randomized controlled clinical trial. Clin Endocrinol. 2017;87(2):194–200. doi: 10.1111/cen.13366. [DOI] [PubMed] [Google Scholar]
- 25.Shokrpour M, Foroozanfard F, Afshar Ebrahimi F, Vahedpoor Z, Aghadavod E, Ghaderi A, Asemi Z. Comparison of myo-inositol and metformin on glycemic control, lipid profiles, and gene expression related to insulin and lipid metabolism in women with polycystic ovary syndrome: a randomized controlled clinical trial. Gynecol Endocrinol. 2019;35(5):406–411. doi: 10.1080/09513590.2018.1540570. [DOI] [PubMed] [Google Scholar]
- 26.Fruzzetti F, Perini D, Russo M, Bucci F, Gadducci A. Comparison of two insulin sensitizers, metformin and myo-inositol, in women with polycystic ovary syndrome (PCOS) Gynecol Endocrinol. 2017;33(1):39–42. doi: 10.1080/09513590.2016.1236078. [DOI] [PubMed] [Google Scholar]
- 27.Nehra J, Kaushal J, Singhal SR, Ghalaut VS. A comparative study of myo inositol versus metformin on biochemical profile in polycystic ovarian syndrome in women. Int J Pharm Res. 2017;8(4):1664–1670. doi: 10.13040/IJPSR.0975-8232.8(4).1664-70. [DOI] [Google Scholar]
- 28.Du J, Wu RR, Lin XF, Ke WN, Du Y. Clinical effect of myo-inositol combined with D-chiro-inositol on insulin resistant polycystic ovary syndrome. China Med. 2018;13(9):1389–1393. doi: 10.3760/j.issn.1673-4777.2018.09.027. [DOI] [Google Scholar]
- 29.Pizzo A, Laganà AS, Barbaro L. Comparison between effects of myo-inositol and D-chiro-inositol on ovarian function and metabolic factors in women with PCOS. Gynecol Endocrinol. 2014;30:205–208. doi: 10.3109/09513590.2013.860120. [DOI] [PubMed] [Google Scholar]
- 30.Le Donne M, Metro D, Alibrandi A, Papa M, Benvenga S. Effects of three treatment modalities (diet, myoinositol or myoinositol associated with D-chiro-inositol) on clinical and body composition outcomes in women with polycystic ovary syndrome. Eur Rev Med Pharmacol Sci. 2019;23:2293–2301. doi: 10.26355/eurrev_201903_17278. [DOI] [PubMed] [Google Scholar]
- 31.Nordio M, Proietti E. The combined therapy with myo-inositol and D-chiro-inositol reduces the risk of metabolic disease in PCOS overweight patients compared to myo-inositol supplementation alone. Eur Rev Med Pharmacol Sci. 2012;16:575–581. [PubMed] [Google Scholar]
- 32.Li XB, Kuang H, Luo Y, Chen QX. Clinical observation of berberine in intervening insulin resistance of polycystic ovary syndrome. J Guangzhou Univ Tradit Chin Med. 2017;1(2):172–177. doi: 10.13359/j.cnki.gzxbtcm.2017.02.006. [DOI] [Google Scholar]
- 33.Wang P, Wang H, Wang Y. Clinical analysis of metformin combined with berberine in the treatment of obese polycystic ovary syndrome. Chin Prim Health Care. 2016;30:77–78. doi: 10.3969/j.issn.1001-568X.2016.10.0032. [DOI] [Google Scholar]
- 34.Wang LX, Kong YZ, Ren YW, Shen M. Therapeutic effect of berberine combined with dfformin for women with polycystic ovary syndrome and insulin resistance. J Zhejiang Chin Med Univ. 2011;5:713–715. doi: 10.3969/j.issn.1005-5509.2011.05.034. [DOI] [Google Scholar]
- 35.Sterne JAC, Savović J, Page MJ, Elbers RG, Blencowe NS, Boutron I, Cates CJ, Cheng H-Y, Corbett MS, Eldridge SM, Emberson JR, Hernán MA, Hopewell S, Hróbjartsson A, Junqueira DR, Jüni P, Kirkham JJ, Lasserson T, Li T, McAleenan A, Reeves BC, Shepperd S, Shrier I, Stewart LA, Tilling K, White IR, Whiting PF, Higgins JPT. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 36.Dias S, Sutton AJ, Ades AE, Welton NJ. Evidence synthesis for decision making 2: a generalized linear modeling framework for pairwise and network meta-analysis of randomized controlled trials. Med Decis Mak. 2013;33(5):607–617. doi: 10.1177/0272989X12458724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5:13. doi: 10.1186/1471-2288-5-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Salanti G, Ades AE, Ioannidis JP. Graphical methods and numerical summaries for presenting results from multiple-treatment meta-analysis: an overview and tutorial. J Clin Epidemiol. 2011;64(2):163–171. doi: 10.1016/j.jclinepi.2010.03.016. [DOI] [PubMed] [Google Scholar]
- 39.Diamanti-Kandarakis E, Christakou CD, Kandaraki E, Economou FN. Metformin: an old medication of new fashion: evolving new molecular mechanisms and clinical implications in polycystic ovary syndrome. Eur J Endocrinol. 2010;162(2):193–212. doi: 10.1530/EJE-09-0733. [DOI] [PubMed] [Google Scholar]
- 40.Rena G, Hardie DG, Pearson ER. The mechanisms of action of metformin. Diabetologia. 2017;60(9):1577–1585. doi: 10.1007/s00125-017-4342-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Macut D, Bjekić-Macut J, Rahelić D, Doknić M. Insulin and the polycystic ovary syndrome. Diabetes Res Clin Pract. 2017;130:163–170. doi: 10.1016/j.diabres.2017.06.011. [DOI] [PubMed] [Google Scholar]
- 42.Panunzi S, Maltese S, Verrastro O, Labbate L, De Gaetano A, Pompili M, Capristo E, Bornstein SR, Mingrone G. Pioglitazone and bariatric surgery are the most effective treatments for non-alcoholic steatohepatitis: a hierarchical network meta-analysis. Diabetes Obes Metab. 2020 doi: 10.1111/dom.14304.10.1111/dom.14304. [DOI] [PubMed] [Google Scholar]
- 43.Lebovitz HE. Thiazolidinediones: the forgotten diabetes medications. Curr Diabetes Rep. 2019;19(12):151. doi: 10.1007/s11892-019-1270-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Xing C, Li CZ, He B. Insulin sensitizers for improving the endocrine and metabolic profile in overweight women with PCOS. J Clin Endocrinol Metab. 2020;105(9):2950–2963. doi: 10.1210/clinem/dgaa337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sortino MA, Salomone S, Carruba MO, Drago F. Polycystic ovary syndrome: insights into the therapeutic approach with inositols. Front Pharmacol. 2017;8:341. doi: 10.3389/fphar.2017.00341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bevilacqua A, Bizzarri M. Inositols in insulin signaling and glucose metabolism. Int J Endocrinol. 2018;2018:1968450. doi: 10.1155/2018/1968450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ravanos K, Monastra G, Pavlidou T, Goudakou M, Prapas N. Can high levels of D-chiro-inositol in follicular fluid exert detrimental effects on blastocyst quality? Eur Rev Med Pharmacol Sci. 2017;21(23):5491–5498. doi: 10.26355/eurrev_201712_13940. [DOI] [PubMed] [Google Scholar]
- 48.Facchinetti F, Orrù B, Grandi G, Unfer V. Short-term effects of metformin and myo-inositol in women with polycystic ovarian syndrome (PCOS): a meta-analysis of randomized clinical trials. Gynecol Endocrinol. 2019;35(3):198–206. doi: 10.1080/09513590.2018.1540578. [DOI] [PubMed] [Google Scholar]
- 49.Arentz S, Smith CA, Abbott J, Bensoussan A. Nutritional supplements and herbal medicines for women with polycystic ovary syndrome; a systematic review and meta-analysis. BMC Complement Altern Med. 2017;17(1):500. doi: 10.1186/s12906-017-2011-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Morley LC, Tang T, Yasmin E, Norman RJ, Balen AH. Insulin-sensitising drugs (metformin, rosiglitazone, pioglitazone, D-chiro-inositol) for women with polycystic ovary syndrome, oligo amenorrhoea and subfertility. Cochrane Database Syst Rev. 2017;11(11):CD003053. doi: 10.1002/14651858.CD003053.pub6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gateva A, Unfer V, Kamenov Z. The use of inositol(s) isomers in the management of polycystic ovary syndrome: a comprehensive review. Gynecol Endocrinol. 2018;34(7):545–550. doi: 10.1080/09513590.2017.1421632. [DOI] [PubMed] [Google Scholar]
- 52.Wei W, Zhao H, Wang A, Sui M, Liang K, Deng H, Ma Y, Zhang Y, Zhang H, Guan Y. A clinical study on the short-term effect of berberine in comparison to metformin on the metabolic characteristics of women with polycystic ovary syndrome. Eur J Endocrinol. 2012;166(1):99–105. doi: 10.1530/EJE-11-0616. [DOI] [PubMed] [Google Scholar]
- 53.Rondanelli M, Infantino V, Riva A, Petrangolini G, Faliva MA, Peroni G, Naso M, Nichetti M, Spadaccini D, Gasparri C, Perna S. Polycystic ovary syndrome management: a review of the possible amazing role of berberine. Arch Gynecol Obstet. 2020;301(1):53–60. doi: 10.1007/s00404-020-05450-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ju J, Li J, Lin Q, Xu H. Efficacy and safety of berberine for dyslipidaemias: a systematic review and meta-analysis of randomized clinical trials. Phytomedicine. 2018;50:25–34. doi: 10.1016/j.phymed.2018.09.212. [DOI] [PubMed] [Google Scholar]
- 55.Xie L, Zhang D, Ma H, He H, Xia Q, Shen W, Chang H, Deng Y, Wu Q, Cong J, Wang CC, Wu X. The effect of berberine on reproduction and metabolism in women with polycystic ovary syndrome: a systematic review and meta-analysis of randomized control trials. Evid Based Complement Altern Med. 2019 doi: 10.1155/2019/7918631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Li MF, Zhou XM, Li XL. The effect of berberine on polycystic ovary syndrome patients with insulin resistance (PCOS-IR): a meta-analysis and systematic review. Evid Based Complement Altern Med. 2018;2018:2532935. doi: 10.1155/2018/2532935. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Appendix S1. Details on study methods.
Data Availability Statement
The datasets supporting the conclusions of this article are included within the article and its additional file.