Abstract
Neuroglobin (Ngb) is a 17 kDa monomeric hexa-coordinated heme protein belonging to the globin family. Ngb is mainly expressed in neurons of the central and peripheral nervous system, although moderate levels of Ngb have been detected in non-nervous tissues. In the past decade, Ngb has been studied for its neuroprotective role in a large number of neurological disorders such as Alzheimer’s disease, Huntington’s disease, brain ischemia and hypoxia. This review discusses and summarizes the natural compounds and the small synthetic molecules capable of modulating Ngb expression that exhibits a protective role against various neurodegenerative diseases.
Key Words: Neuroglobin, neuroglobin pharmacological induction, neuroprotection, neurodegenerative diseases, natural compounds, synthetic small molecules, Alzheimer's disease, Huntington's disease
Introduction
Globins are heme-proteins that binding oxygen and are present in all living organisms. They play an essential role in bacteria, plant, fungi and animal respiration and oxidative energy production. For several years, hemoglobin (Hb) and myoglobin (Mb) were the only two globins identified in vertebrates (Pesce et al., 2002). Hb is found in the red blood cells, it carries the oxygen (O2) molecule from the lungs to all tissues and it contributes to control the pH of body fluids. Mb is localized in the skeletal and cardiac muscular tissue, and it is involved in the storage and increase in O2 transfer to the mitochondria. In addition, Mb is implicated in the detoxification of nitric oxygen and reactive oxygen species (Rassaf et al., 2014).
In 2000, neuroglobin (Ngb), a third type of globin, was discovered (Burmester et al., 2000). Ngb is a heme-protein that is highly conserved throughout evolution, and human and mouse Ngb have a sequence identity higher than 90% (Ascenzi et al., 2016). Ngb is widely expressed in central and peripheral nervous systems, and several studies suggest that it has a central role in neuroprotection. This review presents the natural and synthetic compounds able to modulate Ngb expression, and which show therefore a possible protective role against various neurodegenerative diseases.
Search Strategy
For the present review, we searched the literature using keywords such as Neuroglobin up-regulation, Neuroglobin pharmacological inductors, natural and synthetic compounds in neurodegeneration, on PubMed, Google Scholar and Sci-Finder. In addition, we also used modifications of the above main keywords to thoroughly search the literature.
Neuroglobin Structure and Functions
Ngb, a monomeric 151 amino-acid protein, is a nerve globin family member widely present in vertebrates and largely expressed in neurons of the central and peripheral nervous system and in endocrine tissue (Burmester et al., 2000). Globins are globular metalloproteins that share a common tertiary structure characterized by six or four α-helices (3/3 or 2/2-fold symmetry, respectively) that make a sandwich around a heme group. They reversibly bind O2 via an iron-containing porphyrin ring.
Human Ngb is distributed widely in the human body, including the hippocampus, thalamus, hypothalamus, cerebral cortex, cerebellum, organs with endocrine function and retinal cells. It has been found that in cerebral hypoxia and ischemia injury Ngb is up-regulated suggesting a neuroprotective role in brain disorders (Li et al., 2010). The Ngb concentration is different across the various human brain sections (Van Acker et al., 2019). It has been reported that Ngb overexpression is related to cytoprotective effects on neurons, anti-apoptotic features on nerve tissue and protection against oxidative stress (Ascenzi et al., 2016; Luyckx et al., 2019). Despite the numerous studies performed, the exact functions of Ngb are difficult to define.
As a globin, the main physiological functions of Ngb are binding and transport of O2, scavenging and detoxification of reactive species (including nitric oxygen, carbon monoxide, or hydrogen sulfide), as well as O2 sensing (Burmester and Hankeln, 2014; Ascenzi et al., 2016; Anna Bilska-Wilkosz et al., 2017). Ngb has the common globin structure consisting of eight α-helices denoted A–H, with the heme prosthetic group located between helices E and F.
While in Hb and Mb proteins the heme iron (heme-Fe) is pentacoordinate, the heme-Fe of Ngb is hexacoordinated by four pyrrole N atoms within the heme plane, the distal His(E7)64 side chain and the proximal His(F8)96 residue (Figure 1). The comparison of the high resolution Ngb crystal structure PDB code: 4MPM (1.74 Å) with the other previous deposited structures (human and murine, wild type (wt) and mutant) suggest that the flexibility of the loop between the helices C and D is crucial for the Ngb ability to bind and stabilize exogenous gaseous ligands (Guimarães et al., 2014).
Figure 1.
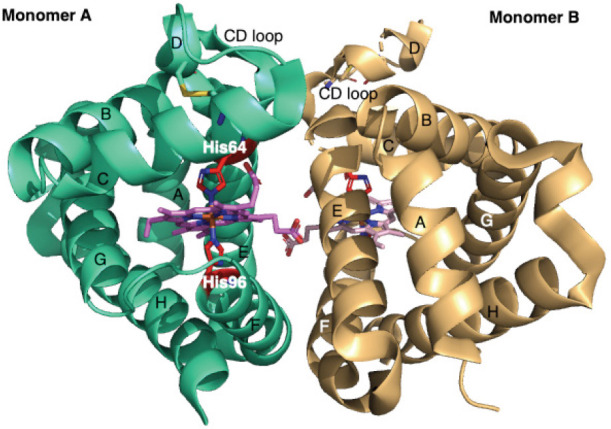
X-ray crystal structure of human Ngb (PDB code: 4MPM).
Graphical representation of the two Ngb monomers (A and B) in the asymmetric unit, light green and light orange respectively. A, B, E, F, G and H α-helices, forming the classical 3/3 sandwich are labelled in both monomers. The hexacoordinated heme group is coloured in violet (Monomer A) and light pink (Monomer B), His64 and His96 are coloured in red. In both monomers, the disulfide bridges located in the CD loop/D helix are labelled. CD loop: The loop between the helices C and D; Ngb: neuroglobin.
Recently, it has been reported that Ngb, as well as Cystatin C, Apolipoprotein A1 and Transthyretin, has a potential protective role in Alzheimer’s disease (AD; Ciccone et al., 2018, 2020a; Van Acker et al., 2019). Previously, in vitro studies showed that Ngb reduced amyloid-β (Aβ) protein toxicity in mouse neurons and pheochromocytoma cell lines suggesting that its antioxidant properties could protect against AD (Khan et al., 2007; Li et al., 2008). Moreover, in vivo investigations showed that Ngb expression increases in AD (Chen et al., 2012), even if the precise mechanisms of Ngb protection against AD remains largely unknown. Experiments, led on human brain of post-mortem AD patients, showed an up-regulation of Ngb expression in hippocampal tissue during early and moderate stages of AD, while the Ngb concentration decreased to control level in advanced AD (Sun et al., 2013). In another study, it has been hypothesized that Ngb through its activation by PI3K/AKt pathway inhibits caspase activity supporting cell survival, an alternative possible strategy to prevent the progression of AD (Li et al., 2016).
The potential neuroprotective function of Ngb has been also investigated in Huntington’s disease (HD), an inherited rare diseases related to a progressive degeneration of neurons in the brain (McColgan and Tabrizi, 2018). It has been reported that huntingtin (Htt) and Ngb take part in neuroprotective axis triggered by the hormone 17β-estradiol (E2) protecting nerve cells from apoptosis (Nuzzo et al., 2017), this synergic pathway fails when huntingtin is mutated. Starting from the idea that neuronal Ngb expression and HD are strictly related, co-localization and interaction between Ngb and Htt were studied in wt- and R6/2 mice models of HD. The result showed a co-localization between Ngb and Htt in wt-mice but not in R6/2 HD models, therefore it has been hypothesized that the Ngb and Htt interaction is mediated by other partners (Cardinale et al., 2018).
The Ngb neuroprotective role is probably related to a synergic mechanism which involves improving mitochondria function, decreasing the secretion of reactive oxygen species and nitric oxygen and inhibiting the intrinsic pathway of cell death (Khan et al., 2007; Li et al., 2008; Jin et al., 2009; Raychaudhuri et al., 2010).
Moreover, it has been suggested that using natural and chemical molecules to induce the Ngb expression in different pathologies, such as ischemia, hypoxia, AD and HD, could be a new therapeutic approach against neurodegenerative diseases (Yu et al., 2009; Jin et al., 2011). Here we summarize the natural and small synthetic molecules that have been studied for their ability to modulate Ngb expression.
Pharmacological Induction of Ngb: Natural and Synthetic Compounds
Various diseases and injuries that affect the central nervous system can be attenuated by Ngb overexpression (Greenberg et al., 2008). Unfortunately, the direct administration of recombinant Ngb in therapy is not suitable because this protein is intracellular and it does not pass through cellular membranes, except in zebrafish (Watanabe and Wakasugi, 2008). However, it has been shown that the use of certain small molecules (natural and synthetic), which are able to cross the blood brain barrier (BBB), can induce the up-regulation of Ngb leading to an improvement in outcome of brain injuries and neurodegenerative disorders in both in vitro and in vivo experiments (Yu et al., 2012).
In Additional Table 1 (783.3KB, pdf) , we list the natural and synthetic molecules able to modulate Ngb, reported in the literature to the best of our knowledge. The compounds have been grouped according to their chemical structure or function into six classes: iron chelators, natural hormones and derivatives, plant derivatives, short fatty acids, nonsteroidal anti-inflammatory drugs, and antidiabetics. Within each class, we specify the origin, natural or synthetic, of the reported compounds.
Furthermore, for the discussed compounds, the structure, the experimental models and the principal clinical uses have been reported in Additional Table 1 (783.3KB, pdf) .
Iron chelators
The first molecule identified as an Ngb inductor was deferoxamine (Dfx; Additional Table 1 (783.3KB, pdf) ), a natural chelating BBB-crossing molecule which is synthesized by Streptomyces pilosus in iron-limiting conditions (Ellenberg et al., 1990; Sun et al., 2001). Dfx is a drug that finds application in the treatment or prevention of iron overload related to thalassemia, and it is a chelating agent able to complex iron favouring its excretion (Olivieri and Brittenham, 1997). It has been shown that Dfx increases the expression of hypoxia inducible gene factor 1, one of the principal transcriptional factors for hypoxia signalling, connected to Ngb expression (Sun et al., 2001).
The literature reports that hemin (Additional Table 1 (783.3KB, pdf) ), a porphyrin containing a ferric iron ion with a chloride ligand, can induce the expression of Hb and Mb (Rutherford et al., 1979; Graber and Woodworth, 1986). Starting from the structural and functional similarity of Ngb with Hb and Mb, it was suggested that hemin can also induce Ngb expression. This hypothesis was confirmed by an in vitro experiment where the HN33 cells (somatic cell fusion of mouse hippocampal neuron and N18TG2 neuroblastoma) treated with hemin showed Ngb up-regulation. It has been hypnotized that, the overexpression mechanism could be regulated by the soluble guanylate cyclase–protein kinase G pathway (Zhu et al., 2002). Moreover, in a recent study, it has been shown that intravenous administration of hemin, in animal models of retinitis pigmentosa, significantly increases the Ngb expression in retina reducing its degeneration (Tao et al., 2018).
Hormones and derivatives
Several studies have suggested that the estrogen hormones have an important physiological role in the brain and protect from neurodegenerative disorders (Vasudevan and Pfaff, 2008). It was highlighted that women in pre or menopause periods are more sensitive to develop AD (Uddin et al., 2020). Particularly, E2 decreased Aβ and glutamate toxicities (Xu et al., 1998). The concept that endogenous modulators such as hormones could also have a role in Ngb regulation levels, prompted scientists to evaluate estrogens in several insults hypothesizing that Ngb can participate in the E2 signalling induction pathway.
The first experimental evidence of the interaction between Ngb and E2 was reported by De Marinis et al. (2010), as shown in Additional Table 1 (783.3KB, pdf) . They demonstrated that E2 increased the Ngb levels up to 300% in both the SK-N-BE human neuroblastoma cell line and mouse hippocampal neurons, showing that E2 can act as an endogenous modulator of Ngb expression (De Marinis et al., 2010, 2013b). The SK-N-BE cells, containing high estrogen receptor (ER)β and low ERα levels, were stimulated with either the specific ERα agonist 4,4′,4′′-(4-propyl-[1H]-pyrazole-1,3,5-triyl)trisphenol or the appropriate ERβ agonist 2,3-bis(4-hydroxyphenyl)propionitrile (DPN) to evaluate the role of each ER subtype in the E2-induced Ngb pathway. Only DPN simulated the E2 effect on Ngb levels, confirming the role of ERβ in Ngb induction (De Marinis et al., 2010; Additional Table 1 (783.3KB, pdf) ).
Also, the upregulation of the Ngb expression induced by E2, in astrocytes, may be related to a mechanism involving estrogen receptor ERβ (De Marinis et al., 2013a).
However, E2 can also bind to the ERα receptor subtype inducing a cancer survival effect and Ngb expression. Experimental data reported that ERα/ERβ ratio is higher in non-neuronal cancers (such as breast and liver) than in normal tissue (Acconcia et al., 2005). Further studies showed that E2 induced Ngb up-regulation both hepatoma and breast adenocarcinoma (MCF-7) cells, which express the ERα receptor subtype (Fiocchetti et al., 2014).
In the literature, controversial results have been reported regarding testosterone, a gonadal hormone. Testosterone alone does not increase the Ngb expression, either in SK-N-BE cells nor mouse in hippocampal neurons (De Marinis et al., 2010), but in another study, it was found that, when T98G cells (human astrocyte cell model) are exposed to glucose deprivation, testosterone induces Ngb production. The latter result suggests that testosterone can regulate the levels of neuroprotective proteins under cellular damaging conditions (Toro-Urrego et al., 2016; Additional Table 1 (783.3KB, pdf) ).
Fucosterol (Fuc) is a phytosterol largely present in brown marine algae and shows several biological effects (anticancer, antidiabetic, antioxidant, antifungal, antihistaminic, anticholinergic, antiadipogenic, etc.) (Abdul et al., 2016). In addition to the previously mentioned endogen hormones, Fuc was studied as a potential Ngb regulator (Additional Table 1 (783.3KB, pdf) ). Human neuroblastoma cell line (SH-SY5Y cells) treated with Fuc showed a remarkable increase of Ngb messenger RNA (mRNA) levels. Moreover, if the SH-SY5Y cells were pre-treated with Fuc before the treatment with Aβ-induced toxicity, the mRNA levels of Ngb increased greatly showing that the neuroprotective effect of Fuc is related to Ngb up-regulation (Gan et al., 2019).
Tibolone (Tib) is a synthetic BBB-crossing steroid hormone that is used in menopause hormone therapy (Kuhl, 2005) and in the prevention of osteoporosis (Ettinger, 2007; Biglia et al., 2010).
Several results, from experimental studies and clinical trials, have suggested that Tib has a neuroprotective effect in CNS (Pinto-Almazán et al., 2017). Tib, such as E2, has a protective effect on astrocytes due to its interaction with ERβ and its ability to upregulate Ngb mRNA (Avila-Rodriguez et al., 2016; Additional Table 1 (783.3KB, pdf) ). Furthermore, it has been shown that Tib has a beneficial effect on BV-2 cells (used as microglia cell model) against inflammation and oxidative stress induced by palmitic acid. The mechanisms by which Tib on its biological activities include the activation of estrogenic pathway via ERβ and the modulation of Ngb levels as well as, reducing mitochondrial damage and inflammation (Hidalgo-Lanussa et al., 2018).
Thyroid hormones have an important role in brain development because they are fundamental for growth and differentiation. An in vivo study evaluated the variation of the levels of two globins located in the brain: Ngb and cytoglobin. It was found that, when rats were treated with high doses of triiodothyronine (T3), both proteins were overexpressed (Oliveira et al., 2015; Additional Table 1 (783.3KB, pdf) ). Even if this study demonstrated an increase in gene and protein expression of the two globins in the brain, further investigations need to be done to elucidate the precise mechanism induced by T3 hormone. Regarding Ngb, one hypothesis is that T3 could upregulate Ngb expression indirectly acting on the hypoxia inducible gene factor 1 (Oliveira et al., 2015).
In addition to the mentioned hormones, erythropoietin also plays a neuroprotective role inducing Ngb up-regulation (Milano and Collomp, 2005). Recently, an in vivo study showed that erythropoietin can stimulate Ngb protein expression in the Mongolian gerbil brain (Gao et al., 2011).
As it is well-known, neuronal response to hypoxic or ischemic injury induces the expression of neuroprotective proteins such as Vascular endothelial growth factor (VEGF). A study, conducted in neuron-enriched murine cerebrocortical cultures, demonstrated that there was a positive correlation between VEGF and Ngb. VEGF upregulated Ngb expression and Ngb downregulated VEGF expression (Jin et al., 2012).
Plant derivatives
Polyphenols are natural compounds containing phenolic groups. They constitute a large area of research in therapies against degenerative diseases including neurodegeneration and cancer (Ortore et al., 2016; Renaud and Martinoli, 2019; Di Meo et al., 2020). The biological effects have been attributed to their antioxidant capacities, their protective capabilities on microcirculation and their estrogen-like anti-inflammatory actions (Poschner et al., 2019).
Naringenin (Nar) is a flavonoid largely studied for its neuroprotective potential in several degenerative pathologies (Chen et al., 2019a; Ciccone et al., 2020b). Conversely, it was found that Nar induced the up-regulation of Ngb in SK-N-BE cells interacting with the ERβ subtype (De Marinis et al., 2010; Additional Table 1 (783.3KB, pdf) ). Interestingly, in another study the Nar effect was investigated in MCF-7 cell line and the result showed that it did not modify the Ngb protein level (Additional Table 1 (783.3KB, pdf) ). Moreover, if E2 was co-administered with Nar, the latter antagonized E2 inducing Ngb upregulation (Cipolletti et al., 2019).
A group of 35 natural compounds were tested using cell based reporter systems (mouse and human) for identification of new Ngb-upregulating molecules. Daidzein (Dzn), genistein, polydatin, biochanin A and especially formononetin were found to result in an increase in Ngb mRNA expression both in mouse and human primary neurons (Sun et al., 2001; Additional Table 1 (783.3KB, pdf) ). Notably, among the five natural inductors, biochanin A, formononetin genistein and Dzn are considered phytoestrogens. They are structurally and functionally similar to estrogen hormones, such as E2, which was previously mentioned as an endogen up-regulator of Ngb gene expression in neurons (De Marinis et al., 2010, 2013b). Among the five new Ngb activators, formononetin displays the highest capability to induce Ngb overexpression. Further investigation has shown that formononetin could induce Ngb up-regulation through activation of cAMP response element-binding protein (Liu et al., 2016).
Recently, Dzn and its metabolites were tested in ERα positive breast cancer cells (MCF-7 and T47D) in order to evaluate the possible correlation between ERα activation and Ngb levels (Montalesi et al., 2020; Additional Table 1 (783.3KB, pdf) ). Contrary to E2, which prompts Ngb up-regulation, the treatment of MCF-7 and T47D cells with Dzn (1–10 μM) reduced Ngb below baseline levels rendering breast cancer cells more sensitive to paclitaxel treatment. The same effect is observed with Dzn-4′-sulfate metabolite, administered at lower concentrations than daidzein (0.1–1 μM) (Montalesi et al., 2020). The other metabolites studied, Equol (Eq), Dzn-7-sulfate (D7S), D-4′,7-disulfate and O-desmethylangolesin had an E2 like effect, increasing Ngb levels proportionally to the concentration used. Precisely, D-4′,7-disulfate (DDS) and O-desmethylangolesin induced Ngb up-regulation at low concentrations, while they showed no effect at high concentrations. The complexity of obtained results suggest that Dzn and its metabolites probably trigger different signal transduction pathways (Montalesi et al., 2020).
Among natural polyphenols, resveratrol (Res) has been largely studied for its numerous potential biological activities including: anti-oxidation, anti-inflammation, anti-cancer, cardioprotection, neuroprotection and estrogenic/antiestrogenic functions (Chen et al., 2019b; Xiao et al., 2019; Ahmadi and Ebrahimzadeh, 2020). Studies have reported that neuronal derivatives SK-N-BE cells, in which ERβ expression is higher than ERα, treated with Res display Ngb up-regulation. This effect persists also when these cells are co-stimulated with E2, showing that Res is an E2 agonist in presence of ERβ and it is an E2 antagonist in presence of ERα (Cipolletti et al., 2019). Due to several reports present in literature suggesting that Ngb also has a protective role in cancer cells (Emara et al., 2010; Ascenzi et al., 2016), Res was tested in two ERα-positive breast cell lines (MCF-7 and T47D). In both experiments Res, acting on E2-induced ERα signalling pathway, decreased Ngb levels rendering cancer cells less resistant to chemotherapeutic drug paclitaxel (Cipolletti et al., 2019).
In addition to Res, quercetin and 8-prenyl-naringeninwere studied as modulators of Ngb levels in ERα positive breast cancer cells (Additional Table 1 (783.3KB, pdf) ). Both of them show Ngb overexpression suggesting that their role as E2 agonists on ERα, without interfering with the E2 effect, if the latter is co-administered (Cipolletti et al., 2019).
In summary, Res, Nar, genistein and Dzn act as antiestrogenic molecules in presence of ERα (i.e., breast cancer cells), and as a consequence, they reduce E2-induced breast cancer cell proliferation. In line with this, these polyphenols reduce the Ngb levels rendering cancer cells more prone to cell death. Note that, different metabolites of these compounds (i.e., daidzein metabolites) act differently from their precursors (i.e., daidzein).
On the other hand, Res and Nar (but also genistein) act as estrogenic molecules in the presence of ERβ subtypes (mainly expressed in neuronal derived cells), increasing the Ngb levels and protecting neurons from oxidative stress injury.
In order to discover new natural compounds capable of inducing Ngb activation, 457 indonesian phytochemicals have been evaluated in silico using the molecular docking technique. It has been reported that Ngb can be activated by 14-3-3 protein, but during hypoxia the 14-3-3 protein shift into the neuron nuclei reducing the Ngb neuroprotective function (Jayaraman et al., 2011). A virtual screening study was used to identify new compounds that mimic 14-3-3 protein in the Ngb activation process. Among all the screened compounds, two phytochemicals: Miraxanthin-III and Strigol (Additional Table 1 (783.3KB, pdf) ) gave good binding scores and similar conformations compared to selected 14-3-3 residues, suggesting that both of these natural products can be considered potential Ngb activators (Pangestu et al., 2018).
Short-chain fatty acid derivatives
Starting from previous results showing that short-chain fatty acid derivatives induced fetal globin expression (Pace et al., 2002), the cinnamic, valproic, butyric, levulinic and succinic acids were studied in HN33 cells in order to investigate if they can act as a regulators of Ngb levels (Jin et al., 2011). Among the fatty acids screened, only cinnamic and valproic acids exhibited relevant induction of Ngb (Jin et al., 2011; Additional Table 1 (783.3KB, pdf) ). Valproic acid is a synthetic compound present in the formulation of several anticonvulsant drugs, while cinnamic acid is a natural product extracted from cinnamic oil. The mechanism by which these fatty acids can up-regulate the Ngb expression is not well understood. Studies suggested that the neuroprotection benefit induced by valproic acid is attributed to histone deacetylase inhibition; however this biological activity seems not related to the induction of Ngb expression (Jin et al., 2011).
Moreover, due to the anticonvulsant activity of valproic aci other anticonvulsants were tested, but none of them showed Ngb induction (Jin et al., 2011).
Nonsteroidal anti-inflammatory drugs
As mentioned above, Ngb is a neuroprotective protein implicated in AD. It supports neuronal cell survival in vitro and in vivo protecting the brain from damage induced by AD progression (Khan et al., 2007; Li et al., 2008; Brittain et al., 2010). In a recent study, a co-drug composed of ibuprofen and α-lipoic acid (IBU-LA) was investigated as a potential inductor of Ngb expression in a rat brain model of AD (Zara et al., 2013; Additional Table 1 (783.3KB, pdf) ). Both IBU and LA have already shown protective action in AD (Weggen et al., 2001; Hager et al., 2007). The IBU-LA co-drug is highly resistant to enzymatic degradation; it crosses the BBB favoring IBU and LA delivery directly to neurons. The results obtained show that the subcutaneous administration of IBU-LA guaranteed a high level of Ngb in the rat brain of the AD model compared to its control (Zara et al., 2013).
Antidiabetic drug
Metformin (Met) is an oral antidiabetic drug largely used in the treatment for type-2 diabetes (Thomas and Gregg, 2017). Recently, Met has been investigated for its potential role in neurodegenerative diseases (Rotermund et al., 2018). In a study performed on adult Wistar rats, the animals were treated with alcohol to induce neurotoxicity and after Met was orally administrated for several days (Additional Table 1 (783.3KB, pdf) ). The result shows that, in response to brain injury induced by alcohol, Met increased Ngb expression in the frontal lobe (Bonea et al., 2020). Moreover, Ngb up-regulation was also found in rats treated only with Met, confirming the antioxidant and protective biological functions of Met (Bonea et al., 2020).
Conclusion
Ngb is a globin discovered 20 years ago. Since then, Ngb has largely been studied for its several biological functions. A considerable amount of experimental evidence shows that Ngb can be considered an endogenous neuroprotective molecule, because its upregulation ameliorates the critical profile that arises after ischemic strokes and brain injuries in general. Moreover, several studies have revealed that Ngb has a protective role in neurogenerative disorders such as AD and HD, where the decreased expression of Ngb has been associated with increased risk of the pathologies’ onset and evolution.
In this review, we collate and discuss the materials available on a number of natural and synthetic compounds, discovered so far, that can act as potential Ngb pharmacological inductors with the aim to summarize all of the principal information in a concise but exhaustive manner.
This overview highlights the chemical structures that can inspire future studies for the design of new molecules able to modulate Ngb level.
The endogenous neuroprotection induced by Ngb overexpression could represent a winning therapeutic strategy. Furthermore, the pharmacological induction of Ngb by natural and synthetic molecules could be useful in the treatment of neurodegenerative diseases that, to date, are lacking in effective therapies.
Additional file:
Additional Table 1 (783.3KB, pdf) : Natural and synthetic compounds as Ngb pharmacological modulators.
Natural and synthetic compounds as Ngb pharmacological modulators
Footnotes
C-Editors: Zhao M, Liu WJ, Wang L; T-Editor: Jia Y
Funding:This work was supported by the Italian Ministero dell’Istruzione, dell’Universitá e della Ricerca PRIN 2017SNRXH3 (to EO and SN); PRA_2018_20 University of Pisa (to EO).
Conflicts of interest:The authors declare no conflicts of interest.
Financial support:This work was supported by the Italian Ministero dell’Istruzione, dell’Universitá e della Ricerca PRIN 2017SNRXH3 (to EO and SN); PRA_2018_20 University of Pisa (to EO).
Copyright license agreement:The Copyright License Agreement has been signed by all authors before publication.
Plagiarism check:Checked twice by iThenticate.
Peer review:Externally peer reviewed.
References
- 1.Abdul QA, Choi RJ, Jung HA, Choi JS. Health benefit of fucosterol from marine algae: a review: Health benefit of fucosterol. J Sci Food Agric. 2016;96:1856–1866. doi: 10.1002/jsfa.7489. [DOI] [PubMed] [Google Scholar]
- 2.Acconcia F, Totta P, Ogawa S, Cardillo I, Inoue S, Leone S, Trentalance A, Muramatsu M, Marino M. Survival versus apoptotic 17beta-estradiol effect: role of ER alpha and ER beta activated non-genomic signaling. J Cell Physiol. 2005;203:193–201. doi: 10.1002/jcp.20219. [DOI] [PubMed] [Google Scholar]
- 3.Ahmadi R, Ebrahimzadeh MA. Resveratrol – A comprehensive review of recent advances in anticancer drug design and development. J Med Chem. 2020;200:112356. doi: 10.1016/j.ejmech.2020.112356. [DOI] [PubMed] [Google Scholar]
- 4.Ascenzi P, di Masi A, Leboffe L, Fiocchetti M, Nuzzo MT, Brunori M, Marino M. Neuroglobin: From structure to function in health and disease. Mol Aspects Med. 2016;52:1–48. doi: 10.1016/j.mam.2016.10.004. [DOI] [PubMed] [Google Scholar]
- 5.Avila-Rodriguez M, Garcia-Segura LM, Hidalgo-Lanussa O, Baez E, Gonzalez J, Barreto GE. Tibolone protects astrocytic cells from glucose deprivation through a mechanism involving estrogen receptor beta and the upregulation of neuroglobin expression. Mol Cell Endocrinol. 2016;433:35–46. doi: 10.1016/j.mce.2016.05.024. [DOI] [PubMed] [Google Scholar]
- 6.Biglia N, Maffei S, Lello S, Nappi RE. Tibolone in postmenopausal women: a review based on recent randomised controlled clinical trials. Gynecol Endocrinol. 2010;26:804–814. doi: 10.3109/09513590.2010.495437. [DOI] [PubMed] [Google Scholar]
- 7.Bilska-Wilkosz A, Iciek M, Górny M, Kowalczyk-Pachel D. The role of hemoproteins: hemoglobin, myoglobin and neuroglobin in endogenous thiosulfate production processes. Int J Mol Sci. 2017;18:1315. doi: 10.3390/ijms18061315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bonea M, Filip GA, Toma VA, Baldea I, Berghian AS, Decea N, Olteanu D, Moldovan R, Crivii C, Vinaşi RC, Micluţia IV. The modulatory effect of metformin on ethanol-induced anxiety, redox imbalance, and extracellular matrix levels in the brains of Wistar rats. J Mol Neurosci. 2020;70:1943–1961. doi: 10.1007/s12031-020-01593-w. [DOI] [PubMed] [Google Scholar]
- 9.Brittain T, Skommer J, Raychaudhuri S, Birch N. An antiapoptotic neuroprotective role for neuroglobin. Int J Mol Sci. 2010;11:2306–2321. doi: 10.3390/ijms11062306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Burmester T, Hankeln T. Function and evolution of vertebrate globins. Acta Physiol (Oxf) 2014;211:501–514. doi: 10.1111/apha.12312. [DOI] [PubMed] [Google Scholar]
- 11.Burmester T, Weich B, Reinhardt S, Hankeln T. A vertebrate globin expressed in the brain. Nature. 2000;407:520–523. doi: 10.1038/35035093. [DOI] [PubMed] [Google Scholar]
- 12.Cardinale A, Fusco FR, Paldino E, Giampà C, Marino M, Nuzzo MT, D’Angelo V, Laurenti D, Straccia G, Fasano D, Sarnataro D, Squillaro T, Paladino S, Melone MAB. Localization of neuroglobin in the brain of R6/2 mouse model of Huntington’s disease. Neurol Sci. 2018;39:275–285. doi: 10.1007/s10072-017-3168-2. [DOI] [PubMed] [Google Scholar]
- 13.Chen C, Wei YZ, He XM, Li DD, Wang GQ, Li JJ, Zhang F. Naringenin produces neuroprotection against LPS-induced dopamine neurotoxicity via the inhibition of microglial NLRP3 inflammasome activation. Front Immunol. 2019a;10:936. doi: 10.3389/fimmu.2019.00936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chen JY, Zhu Q, Zhang S, OuYang D, Lu JH. Resveratrol in experimental Alzheimer’s disease models: A systematic review of preclinical studies. Pharmacol Res. 2019b;150:104476. doi: 10.1016/j.phrs.2019.104476. [DOI] [PubMed] [Google Scholar]
- 15.Chen LM, Xiong YS, Kong FL, Qu M, Wang Q, Chen XQ, Wang JZ, Zhu LQ. Neuroglobin attenuates Alzheimer-like tau hyperphosphorylation by activating Akt signaling. J Neurochem. 2012;120:157–164. doi: 10.1111/j.1471-4159.2011.07275.x. [DOI] [PubMed] [Google Scholar]
- 16.Ciccone L, Fruchart-Gaillard C, Mourier G, Savko M, Nencetti S, Orlandini E, Servent D, Stura EA, Shepard W. Copper mediated amyloid-β binding to Transthyretin. Sci Rep. 2018;8:13744. doi: 10.1038/s41598-018-31808-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ciccone L, Shi C, di Lorenzo D, Van Baelen AC, Tonali N. The positive eptide with Tau, TTR, CysC, and ApoA1. Molecules. 2020a;25:2439. doi: 10.3390/molecules25102439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ciccone L, Tonali N, Nencetti S, Orlandini E. Natural compounds as inhibitors of transthyretin amyloidosis and neuroprotective agents: analysis of structural data for future drug design. J Enzyme Inhib Med Chem. 2020b;35:1145–1162. doi: 10.1080/14756366.2020.1760262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cipolletti M, Montalesi E, Nuzzo MT, Fiocchetti M, Ascenzi P, Marino M. Potentiation of paclitaxel effect by resveratrol in human breast cancer cells by counteracting the 17β-estradiol/estrogen receptor α/neuroglobin pathway. J Cell Physiol. 2019;234:3147–3157. doi: 10.1002/jcp.27309. [DOI] [PubMed] [Google Scholar]
- 20.De Marinis E, Acaz-Fonseca E, Arevalo MA, Ascenzi P, Fiocchetti M, Marino M, Garcia-Segura LM. 17β-Oestradiol anti-inflammatory effects in primary astrocytes require oestrogen receptor β-mediated neuroglobin up-regulation. J Neuroendocrinol. 2013a;25:260–270. doi: 10.1111/jne.12007. [DOI] [PubMed] [Google Scholar]
- 21.De Marinis E, Ascenzi P, Pellegrini M, Galluzzo P, Bulzomi P, Arevalo MA, Garcia-Segura LM, Marino M. 17β-Estradiol -- A new modulator of neuroglobin levels in neurons: role in neuroprotection against H2O2-induced toxicity. Neurosignals. 2010;18:223–235. doi: 10.1159/000323906. [DOI] [PubMed] [Google Scholar]
- 22.De Marinis E, Fiocchetti M, Acconcia F, Ascenzi P, Marino M. Neuroglobin upregulation induced by 17β-estradiol sequesters cytocrome c in the mitochondria preventing H2O2-induced apoptosis of neuroblastoma cells. Cell Death Dis. 2013b;4:e508. doi: 10.1038/cddis.2013.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Di Meo F, Valentino A, Petillo O, Peluso G, Filosa S, Crispi S. Bioactive polyphenols and neuromodulation: molecular mechanisms in neurodegeneration. Int J Mol Sci. 2020;21:2564. doi: 10.3390/ijms21072564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ellenberg R, King AL, Sica DA, Posner M, Savory J. Cerebrospinal fluid aluminum levels following deferoxamine. Am J Kidney Dis. 1990;16:157–159. doi: 10.1016/s0272-6386(12)80572-5. [DOI] [PubMed] [Google Scholar]
- 25.Emara M, Turner AR, Allalunis-Turner J. Hypoxic regulation of cytoglobin and neuroglobin expression in human normal and tumor tissues. Cancer Cell Int. 2010;10:33. doi: 10.1186/1475-2867-10-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ettinger B. Tibolone for prevention and treatment of postmenopausal osteoporosis. Maturitas. 2007;57:35–38. doi: 10.1016/j.maturitas.2007.02.008. [DOI] [PubMed] [Google Scholar]
- 27.Fiocchetti M, Nuzzo MT, Totta P, Acconcia F, Ascenzi P, Marino M. Neuroglobin, a pro-survival player in estrogen receptor α-positive cancer cells. Cell Death Dis. 2014;5:e1449. doi: 10.1038/cddis.2014.418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gan SY, Wong LZ, Wong JW, Tan EL. Fucosterol exerts protection against amyloid β-induced neurotoxicity, reduces intracellular levels of amyloid β and enhances the mRNA expression of neuroglobin in amyloid β-induced SH-SY5Y cells. Int J Biol Macromol. 2019;121:207–213. doi: 10.1016/j.ijbiomac.2018.10.021. [DOI] [PubMed] [Google Scholar]
- 29.Gao Y, Mengana Y, Cruz YR, Muñoz A, Testé IS, García JD, Wu Y, Rodríguez JC, Zhang C. Different expression patterns of Ngb and EPOR in the cerebral cortex and hippocampus revealed distinctive therapeutic effects of intranasal delivery of Neuro-EPO for ischemic insults to the gerbil brain. J Histochem Cytochem. 2011;59:214–227. doi: 10.1369/0022155410390323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Graber S, Woodworth RC. Myoglobin expression in L6 muscle cells. Role of differentiation and heme. J Biol Chem. 1986;261:9150–9154. [PubMed] [Google Scholar]
- 31.Greenberg D, Jin K, Khan A. Neuroglobin: an endogenous neuroprotectant. Current Opin Pharmacol. 2008;8:20–24. doi: 10.1016/j.coph.2007.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Guimarães BG, Hamdane D, Lechauve C, Marden MC, Golinelli-Pimpaneau B. The crystal structure of wild-type human brain neuroglobin reveals flexibility of the disulfide bond that regulates oxygen affinity. Acta Crystallogr D Biol Crystallogr. 2014;70:1005–1014. doi: 10.1107/S1399004714000078. [DOI] [PubMed] [Google Scholar]
- 33.Hager K, Kenklies M, McAfoose J, Engel J, Münch G. Alpha-lipoic acid as a new treatment option for Alzheimer’s disease--a 48 months follow-up analysis. J Neural Transm Suppl. 2007;72:189–193. doi: 10.1007/978-3-211-73574-9_24. [DOI] [PubMed] [Google Scholar]
- 34.Hidalgo-Lanussa O, Ávila-Rodriguez M, Baez-Jurado E, Zamudio J, Echeverria V, Garcia-Segura LM, Barreto GE. Tibolone reduces oxidative damage and inflammation in microglia stimulated with palmitic acid through mechanisms involving estrogen receptor beta. Mol Neurobiol. 2018;55:5462–5477. doi: 10.1007/s12035-017-0777-y. [DOI] [PubMed] [Google Scholar]
- 35.Jayaraman T, Tejero J, Chen BB, Blood AB, Frizzell S, Shapiro C, Tiso M, Hood BL, Wang X, Zhao X, Conrads TP, Mallampalli RK, Gladwin MT. 14-3-3 Binding and phosphorylation of neuroglobin during hypoxia modulate six-to-five heme pocket coordination and rate of nitrite reduction to nitric oxide. J Biol Chem. 2011;286:42679–42689. doi: 10.1074/jbc.M111.271973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Jin K, Mao X, Xie L, Greenberg DA. Interactions between vascular endothelial growth factor and neuroglobin. Neurosci Lett. 2012;519:47–50. doi: 10.1016/j.neulet.2012.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jin K, Mao XO, Xie L, John V, Greenberg DA. Pharmacological Induction of neuroglobin expression. Pharmacology. 2011;87:81–84. doi: 10.1159/000322998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Jin K, Mao XO, Xie L, Khan AA, Greenberg DA. Neuroglobin protects against nitric oxide toxicity. Neurosci Lett. 2009;430:135–137. doi: 10.1016/j.neulet.2007.10.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Khan AA, Mao XO, Banwait S, Jin K, Greenberg DA. Neuroglobin attenuates beta-amyloid neurotoxicity in vitro and transgenic Alzheimer phenotype in vivo. Proc Natl Acad Sci U S A. 2007;104:19114–19119. doi: 10.1073/pnas.0706167104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Kuhl H. Pharmacology of estrogens and progestogens: influence of different routes of administration. Climacteric 8 Suppl. 2005;1:3–63. doi: 10.1080/13697130500148875. [DOI] [PubMed] [Google Scholar]
- 41.Li RC, Guo SZ, Lee SK, Gozal D. Neuroglobin protects neurons against oxidative stress in global ischemia. J Cereb Blood Flow Metab. 2010;30:1874–1882. doi: 10.1038/jcbfm.2010.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Li RC, Pouranfar F, Lee SK, Morris MW, Wang Y, Gozal D. Neuroglobin protects PC12 cells against beta-amyloid-induced cell injury. Neurobiol Aging. 2008;29:1815–1822. doi: 10.1016/j.neurobiolaging.2007.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Li Y, Dai Y, Sun J, Xiang Y, Yang J, Dai S, Zhang X. Neuroglobin attenuates beta amyloid-induced apoptosis through inhibiting caspases activity by activating PI3K/Akt signaling pathway. J Mol Neurosci. 2016;58:28–38. doi: 10.1007/s12031-015-0645-z. [DOI] [PubMed] [Google Scholar]
- 44.Liu N, Yu Z, Gao X, Song YS, Yuan J, Xun Y, Wang T, Yan F, Yuan S, Zhang J, Xiang S, Lo EH, Wang X. Establishment of cell-based neuroglobin promoter reporter assay for neuroprotective compounds screening. CNS Neurol Disord Drug Targets. 2016;15:629–639. doi: 10.2174/1871527315666160321105612. [DOI] [PubMed] [Google Scholar]
- 45.Luyckx E, Van Acker ZP, Ponsaerts P, Dewilde S. Neuroglobin expression models as a tool to study its function. Oxid Med Cell Longev. 2019;2019:5728129. doi: 10.1155/2019/5728129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.McColgan P, Tabrizi SJ. Huntington’s disease: a clinical review. Eur J Neurol. 2018;25:24–34. doi: 10.1111/ene.13413. [DOI] [PubMed] [Google Scholar]
- 47.Milano M, Collomp R. Erythropoietin and neuroprotection: a therapeutic perspective. J Oncol Pharm Pract. 2005;11:145–149. doi: 10.1191/1078155205jp162oa. [DOI] [PubMed] [Google Scholar]
- 48.Montalesi E, Cipolletti M, Cracco P, Fiocchetti M, Marino M. Divergent effects of daidzein and its metabolites on estrogen-induced survival of breast cancer cells. Cancers (Basel) 2020;12:167. doi: 10.3390/cancers12010167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Nuzzo MT, Fiocchetti M, Totta P, Melone MAB, Cardinale A, Fusco FR, Gustincich S, Persichetti F, Ascenzi P, Marino M. Huntingtin polyQ mutation impairs the 17β-estradiol/neuroglobin pathway devoted to neuron survival. Mol Neurobiol. 2017;54:6634–6646. doi: 10.1007/s12035-016-0337-x. [DOI] [PubMed] [Google Scholar]
- 50.Oliveira KC, da Conceição RR, Piedade GC, de Souza JS, Sato MA, de Barros Maciel RM, Giannocco G. Thyroid hormone modulates neuroglobin and cytoglobin in rat brain. Metab Brain Dis. 2015;30:1401–1408. doi: 10.1007/s11011-015-9718-5. [DOI] [PubMed] [Google Scholar]
- 51.Olivieri NF, Brittenham GM. Iron-chelating therapy and the treatment of thalassemia. Blood. 1997;89:739–761. [PubMed] [Google Scholar]
- 52.Ortore G, Orlandini E, Braca A, Ciccone L, Rossello A, Martinelli A, Nencetti S. Targeting different transthyretin binding sites with unusual natural compounds. ChemMedChem. 2016;11:1865–1874. doi: 10.1002/cmdc.201600092. [DOI] [PubMed] [Google Scholar]
- 53.Pace BS, White GL, Dover GJ, Boosalis MS, Faller DV, Perrine SP. Short-chain fatty acid derivatives induce fetal globin expression and erythropoiesis in vivo. Blood. 2002;100:4640–4648. doi: 10.1182/blood-2002-02-0353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Pangestu AP, Indarto D, Balgis Neuroglobin activator In Silico found from Mirabilis Jalapa for stroke treatment. AIP Conference Proceedings. 2018. 2021, 070002 (2018) https://doi.org/10.1063/1.5062800 .
- 55.Pesce A, Bolognesi M, Bocedi A, Ascenzi P, Dewilde S, Moens L, Hankeln T, Burmester T. Neuroglobin and cytoglobin. Fresh blood for the vertebrate globin family. EMBO Rep. 2002;3:1146–1151. doi: 10.1093/embo-reports/kvf248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Pinto-Almazán R, Segura-Uribe JJ, Farfán-García ED, Guerra-Araiza C. Effects of tibolone on the central nervous system: clinical and experimental approaches. Biomed Res Int. 2017;2017:8630764. doi: 10.1155/2017/8630764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Poschner S, Maier-Salamon A, Thalhammer T, Jäger W. Resveratrol and other dietary polyphenols are inhibitors of estrogen metabolism in human breast cancer cells. J Steroid Biochem Mol Biol. 2019;190:11–18. doi: 10.1016/j.jsbmb.2019.03.001. [DOI] [PubMed] [Google Scholar]
- 58.Rassaf T, Totzeck M, Hendgen-Cotta UB, Shiva S, Heusch G, Kelm M. Circulating nitrite contributes to cardioprotection by remote ischemic preconditioning. Circ Res. 2014;114:1601–1610. doi: 10.1161/CIRCRESAHA.114.303822. [DOI] [PubMed] [Google Scholar]
- 59.Raychaudhuri S, Skommer J, Henty K, Birch N, Brittain T. Neuroglobin protects nerve cells from apoptosis by inhibiting the intrinsic pathway of cell death. Apoptosis. 2010;15:401–411. doi: 10.1007/s10495-009-0436-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Renaud J, Martinoli MG. Considerations for the use of polyphenols as therapies in neurodegenerative diseases. Int J Mol Sci. 2019;20:1883. doi: 10.3390/ijms20081883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Rotermund C, Machetanz G, Fitzgerald JC. The therapeutic potential of metformin in neurodegenerative diseases. Front Endocrinol (Lausanne) 2018;9:400. doi: 10.3389/fendo.2018.00400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Rutherford TR, Clegg JB, Weatherall DJ. K562 human leukaemic cells synthesise embryonic haemoglobin in response to haemin. Nature. 1979;280:164–165. doi: 10.1038/280164a0. [DOI] [PubMed] [Google Scholar]
- 63.Sun F, Mao X, Xie L, Greenberg DA, Jin K. Neuroglobin protein is upregulated in Alzheimer’s disease. J Alzheimers Dis. 2013;36:659–663. doi: 10.3233/JAD-130323. [DOI] [PubMed] [Google Scholar]
- 64.Sun Y, Jin K, Mao XO, Zhu Y, Greenberg DA. Neuroglobin is up-regulated by and protects neurons from hypoxic-ischemic injury. Proc Natl Acad Sci U S A. 2001;98:15306–15311. doi: 10.1073/pnas.251466698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Tao Y, Ma Z, Liu B, Fang W, Qin L, Huang YF, Wang L, Gao Y. Hemin supports the survival of photoreceptors injured by N-methyl-N-nitrosourea: The contributory role of neuroglobin in photoreceptor degeneration. Brain Res. 2018;1678:47–55. doi: 10.1016/j.brainres.2017.10.007. [DOI] [PubMed] [Google Scholar]
- 66.Thomas I, Gregg B. Metformin; a review of its history and future: from lilac to longevity: THOMAS AND GREGG. Pediatr Diabetes. 2017;18:10–16. doi: 10.1111/pedi.12473. [DOI] [PubMed] [Google Scholar]
- 67.Toro-Urrego N, Garcia-Segura LM, Echeverria V, Barreto GE. Testosterone protects mitochondrial function and regulates neuroglobin expression in astrocytic cells exposed to glucose deprivation. Front Aging Neurosci. 2016;8:152. doi: 10.3389/fnagi.2016.00152. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 68.Uddin MdS, Rahman MdM, Jakaria Md, Rahman MdS, Hossain MdS, Islam A, Ahmed M, Mathew B, Omar UM, Barreto GE, Ashraf GM. Estrogen signaling in Alzheimer’s disease: molecular insights and therapeutic targets for Alzheimer’s Dementia. Mol Neurobiol. 2020;57:2654–2670. doi: 10.1007/s12035-020-01911-8. [DOI] [PubMed] [Google Scholar]
- 69.Van Acker ZP, Luyckx E, Dewilde S. Neuroglobin expression in the brain: a story of tissue homeostasis preservation. Mol Neurobiol. 2019;56:2101–2122. doi: 10.1007/s12035-018-1212-8. [DOI] [PubMed] [Google Scholar]
- 70.Vasudevan N, Pfaff DW. Non-genomic actions of estrogens and their interaction with genomic actions in the brain. Front Neuroendocrinol. 2008;29:238–257. doi: 10.1016/j.yfrne.2007.08.003. [DOI] [PubMed] [Google Scholar]
- 71.Watanabe S, Wakasugi K. Zebrafish neuroglobin is a cell-membrane-penetrating globin. Biochemistry. 2008;47:5266–5270. doi: 10.1021/bi800286m. [DOI] [PubMed] [Google Scholar]
- 72.Weggen S, Eriksen JL, Das P, Sagi SA, Wang R, Pietrzik CU, Findlay KA, Smith TE, Murphy MP, Bulter T, Kang DE, Marquez-Sterling N, Golde TE, Koo EH. A subset of NSAIDs lower amyloidogenic Abeta42 independently of cyclooxygenase activity. Nature. 2001;414:212–216. doi: 10.1038/35102591. [DOI] [PubMed] [Google Scholar]
- 73.Xiao Q, Zhu W, Feng W, Lee SS, Leung AW, Shen J, Gao L, Xu C. A Review of Resveratrol as a Potent Chemoprotective and Synergistic Agent in Cancer Chemotherapy. Front Pharmacol. 2019;9:1534. doi: 10.3389/fphar.2018.01534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Xu H, Gouras GK, Greenfield JP, Vincent B, Naslund J, Mazzarelli L, Fried G, Jovanovic JN, Seeger M, Relkin NR, Liao F, Checler F, Buxbaum JD, Chait BT, Thinakaran G, Sisodia SS, Wang R, Greengard P, Gandy S. Estrogen reduces neuronal generation of Alzheimer beta-amyloid peptides. Nat Med. 1998;4:447–451. doi: 10.1038/nm0498-447. [DOI] [PubMed] [Google Scholar]
- 75.Yu Z, Liu J, Guo S, Xing C, Fan X, Ning M, Yuan JC, Lo EH, Wang X. Neuroglobin-overexpression alters hypoxic response gene expression in primary neuron culture following oxygen glucose deprivation. Neuroscience. 2009;162:396–403. doi: 10.1016/j.neuroscience.2009.04.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Yu Z, Liu N, Liu J, Yang K, Wang X. Neuroglobin, a novel target for endogenous neuroprotection against stroke and neurodegenerative disorders. Int J Mol Sci. 2012;13:6995–7014. doi: 10.3390/ijms13066995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Zara S, De Colli M, Rapino M, Pacella S, Nasuti C, Sozio P, Di Stefano A, Cataldi A. Ibuprofen and lipoic acid conjugate neuroprotective activity is mediated by Ngb/Akt intracellular signaling pathway in Alzheimer’s disease rat model. Gerontology. 2013;59:250–260. doi: 10.1159/000346445. [DOI] [PubMed] [Google Scholar]
- 78.Zhu Y, Sun Y, Jin K, Greenberg DA. Hemin induces neuroglobin expression in neural cells. Blood. 2002;100:2494–2498. doi: 10.1182/blood-2002-01-0280. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Natural and synthetic compounds as Ngb pharmacological modulators


