Abstract
Glaucoma is a group of optic neuropathies characterised by the degeneration of retinal ganglion cells, resulting in damage to the optic nerve head (ONH) and loss of vision in one or both eyes. Increased intraocular pressure (IOP) is one of the major aetiological risk factors in glaucoma, and is currently the only modifiable risk factor. However, 30–40% of glaucoma patients do not present with elevated IOP and still proceed to lose vision. The pathophysiology of glaucoma is therefore not completely understood, and there is a need for the development of IOP-independent neuroprotective therapies to preserve vision. Neuroinflammation has been shown to play a key role in glaucoma and, specifically, the NLRP3 inflammasome, a key driver of inflammation, has recently been implicated. The NLRP3 inflammasome is expressed in the eye and its activation is reported in pre-clinical studies of glaucoma. Activation of the NLRP3 inflammasome results in IL-1β processing. This pro inflammatory cytokine is elevated in the blood of glaucoma patients and is believed to drive neurotoxic inflammation, resulting in axon degeneration and the death of retinal ganglion cells (RGCs). This review discusses glaucoma as an inflammatory disease and evaluates targeting the NLRP3 inflammasome as a therapeutic strategy. A hypothetical mechanism for the action of the NLRP3 inflammasome in glaucoma is presented.
Keywords: NLRP3 inflammasome, glaucoma, RGC (retinal ganglion cells), inflammation
1. Introduction
Glaucoma is a neurodegenerative disease and the leading cause of irreversible blindness worldwide. Glaucoma affects more than 70 million people, 10% of which are bilaterally blind [1,2]. The prevalence of glaucoma is estimated to increase to 111.8 million by 2040, which can be attributed to an aging population [3]. Glaucoma is characterised by degeneration of the retinal ganglion cell (RGC) axons resulting in damage or remodelling of the optic nerve head (ONH), as evidenced by the characteristic clinical sign of optic disc cupping [4]. These clinical manifestations lead to disruption of the visual pathway and vision loss in one or both eyes [5]. The most common type of glaucoma is primary open-angle glaucoma (POAG)—referred to as “glaucoma” in this review. POAG is characterised by diminished outflow of aqueous humour (AH) despite an unobstructed or open irideocorneal angle [6]. Many patients with glaucoma present with elevated intraocular pressure (IOP > 21 mmHg); however, this is not a requirement for the diagnosis of glaucoma, with 30–40% presenting with normal IOP [5].
The pathophysiology of glaucoma is not well understood and, currently, IOP is the only modifiable risk factor in the disease [7]. The goal of interventions for glaucoma are to lower IOP to a level that may prevent further damage to the ONH and therefore vision loss [5,6,7,8]. Interventions include topical medications that can be applied to the ocular surface, oral medications, laser treatment, and surgery to regulate AH outflow and production to lower IOP [1]. However, approximately 30–40% of POAG patients exhibit a normal IOP, indicating that elevated IOP is not the sole causative factor of glaucoma [9]. In fact a number of risk factors, such as genetics, age, and lifestyle, trigger common pathological endpoints resulting in glaucomatous optic neuropathy [10].
Impaired axonal transport in the RGCs, observed in both animal and human studies, is reported as a potential mechanism of damage in glaucoma [11]. Ischemia, restriction of blood to the bodies tissues, has also been implicated in the pathogenesis of glaucoma, with reduced ONH blood flow reported in glaucoma patients [12]. Excitotoxicity, which results in neuron damage, has been an area of interest in the pathophysiology of glaucoma; however, the data in this area are contradictory [5,13]. Moreover, there is an emerging body of evidence to support the role of inflammation in glaucoma pathogenesis [14,15].
2. Inflammation in Glaucoma
A number of recent studies have investigated the role of inflammation in glaucoma [16,17]. In experimental glaucoma, several studies reveal a significant induction of inflammatory genes in the ONH and retina in the early stages of glaucoma [18,19,20]. Inflammation in glaucoma primarily occurs in the retina and ONH [21]. Common triggers in glaucoma, such as vascular, mechanical, and immune triggers, all lead to astrocyte and microglial reactivity, neurotrophic factor deprivation, and oxidative stress [10]. Such cascades are believed to result in axonal damage in the ONH, which is the primary site of injury in glaucoma [10,22]. In human and experimental models of glaucoma, activated astrocytes [23,24] and activated microglia [25,26] are detected in the ONH, and coincide with the increased expression of proinflammatory cytokines, such as IL-1β and TNFα, and neurotoxic mediators, such as nitric oxide (NO), reactive oxygen species (ROS), and glutamate [26,27]. Inflammation and oxidative stress co-exist in glaucoma, as inflammation appears to increase the amount of oxidative stress and vice versa, creating a chronic state of inflammation and oxidative stress [28]. ONH astrocytes in glaucoma also overexpress cell adhesion proteins, which may promote the migration of immune cells to the site of ONH damage, further amplifying the immune response [29].
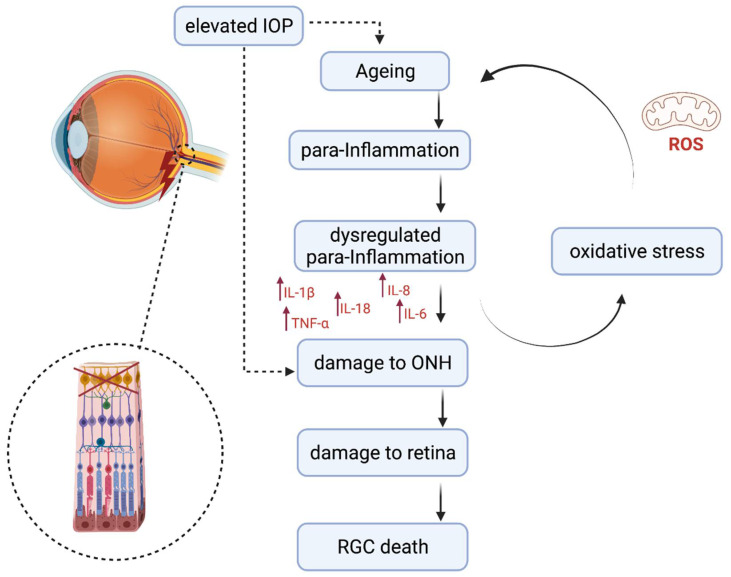
The retina has a self-defence system consisting of microglia, astrocytes, and Müller cells [30]. Chronic oxidative stress appears to drive inflammation through the activation of a para-inflammatory response in the retina [28], and is believed to be a result of aging [31]. Microglia in the aging retina appear to undergo morphological changes and also increase in number [32,33]. Para-inflammation involves low levels of inflammatory activation, which can occur in the microglia and can contribute to damage of the retina if dysregulated (Figure 1) [31]. A number of inflammatory cytokines involved in ageing are also found in the glaucomatous eye [34,35,36,37,38]. An increase in inflammatory cytokines IFN-γ, IL-6, IL-4, IL-10, and IL-1β resulted in a reduction of brn3a+ RGC cells in a glaucoma mouse model, and is thought to be related to microglial activation [39]. There is also evidence of complement activation in the glaucomatous retina [40]. The severity of RGC degeneration in glaucoma models can be correlated with microglial activation [41]. However, it is clear that axon degeneration and RGC death results from a system of complex interactions in glaucoma of many different cells and mediators [10].
Figure 1.
Aging and para-inflammation contributes to retinal ganglion cell death. Ageing in the retina due to ROS is thought to activate a para-inflammatory response, which can become dysregulated and result in damage to the ONH and retina and subsequently RGC death ROS—reactive oxygen species; RG—retinal ganglion cells; ONH—optic nerve head.
Blocking activation of microglia in an experimental glaucoma model reduced RGC death, further strengthening the hypothesis of glial activation and inflammation playing a vital role in glaucoma pathogenesis [25,42]. Targeting inflammation or immune cells as a therapeutic option in glaucoma appears promising. Several novel anti-inflammatory inhibitors have been assessed in different animal models of glaucoma, targeting different inflammatory markers to improve glaucoma disease pathogenesis (Table 1). These inhibitors alter several physical and pathophysiological measures (see Table 1). Drawbacks are associated with some; however, for example, oryzanol failed to control IOP in an acute animal model of glaucoma [43], and lutein is ineffective at reducing increased levels of TNFα in chronic hypoxia [44]. However, the inhibition of specific inflammatory pathways may prove more efficacious than the current tested non-specific inflammatory inhibitors [45,46]. Many anti-inflammatory therapies that specifically target pro-inflammatory cytokines often experience challenges in human trials, such as increasing the risk of fatal infections [47,48].
Table 1.
Anti-inflammatory inhibitors tested in several in-vivo models of glaucoma.
| Inhibitor of Inflammatory Markers | Inflammatory Markers | Glaucoma Model | In-Vivo Findings | References |
|---|---|---|---|---|
| γ-Oryzanol | TNF-α and IL-6 | Subconjunctival injection of phenol in rabbit | Reduces IOP in a chronic glaucoma model by inhibiting the induction of TNF-α and IL-6, and provides protection against glaucoma | [43] |
| Fas inhibitor, ONL1204 | Caspase-8, TNF-α, IL-1β, IL-6, and IL-18 | Intracameral injection of microbeads in C57BL/6J mice | No effect on IOP. Prevents RGC death and axon degradation. Reduces microglial activation and inhibits induction of inflammatory cytokines and chemokines. | [52] |
| Myricetin | IL-1α, IL-1β, IL-6, and TNF-α | Injection of hyaluronic acid into the anterior chamber of the eye in Dawley rats | Lowers IOP level in animals and reduces inflammatory marker levels in in vitro experiments. | [46] |
| Lutein (hydroxycarotenoid) | TNF-α and IL-1β | Mouse model of retinal ischemia | Modulates the overexpression of GFAP in in vivo models of retinal ischemia and inhibits overactivation of NF-κB, IL-1β, and Cox-2 in Müller cells. | [44] |
| Puerarin | IL-1β, IL-17A, and TNF-α | Neovascular glaucoma in C57BL/6 mice | Puerarin reduces high levels of IL-1β, IL-17A, and TNF-α in animal models of glaucoma. It also maintains reactive oxygen species, superoxide dismutase and malondialdehyde, NOS, and inducible NOS and NF-κB to an optimum level. | [45] |
IOP—intraocular pressure; RGCs—retinal ganglion cells; NOS—neuronal nitric oxide synthase; GFAP—glial fibrillary acidic protein; IOP—intraocular pressure; RGC—retinal ganglion cell; NF-κβ—nuclear factor-kappa beta; IL—interluekin; TNF-α—tumour necrosis factor-alpha; Cox-2—cyclooxygenase 2; NOS—nitric oxide synthases.
Therefore, there is a need to develop novel but safe anti-inflammatory therapies [49,50]. Recently, the nucleotide-binding oligomerization domain, Leucine rich Repeat and Pyrin domain containing protein 3 (NLRP3) inflammasome, has gained attention as a potential key orchestrator of inflammation in the aetiology of glaucoma, and may be an attractive therapeutic target [21,51].
3. The NLRP3 Inflammasome
Pattern recognition receptors (PRRs) recognise danger signals, such as, pathogen associated molecular patterns (PAMPs) and danger associated molecular patterns (DAMPs) [53]. Nod like receptors (NLRs) are a type of PRR with a nucleotide-binding and oligomerisation domain, and act as receptors in the cytoplasm [50]. NLRs are categorised into four subgroups based on their N-terminal domain, with the NLRP group containing a pyrin domain [54]. NLRP3 is one member of the NLRP family, of which there are 14 members, all of which are involved in the formation of inflammasomes [55]. NLRP3 is the most well characterised member of the inflammasome family [56], and is an important regulator of inflammatory diseases and plays a key role in the innate immune system [57].
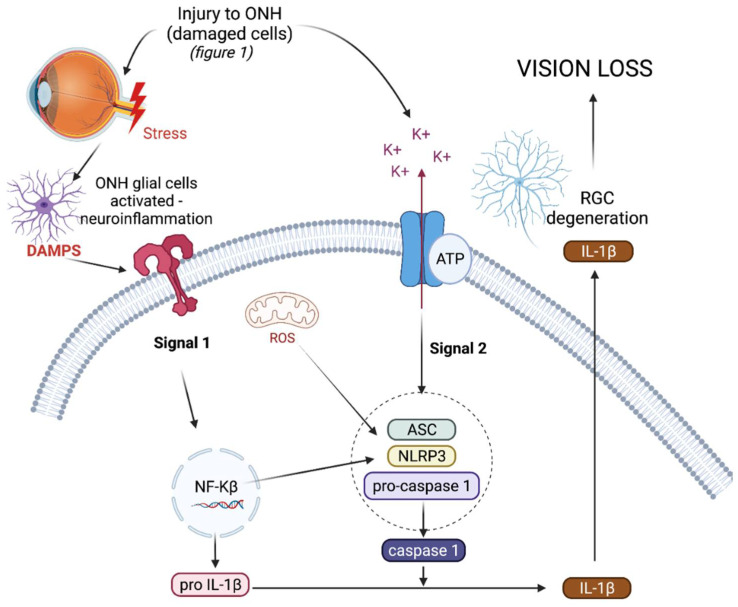
The NLRP3 inflammasome (NOD-, LRR-, and pyrin domain-containing protein 3) is an intracellular, multiprotein signalling complex implicated in a plethora of inflammatory diseases [55]. These integral elements include the sensor NLRP3 protein, an adaptor protein called adaptor molecule apoptosis-associated speck-like protein containing a CARD (ASC) and procaspase-1 [58]. The sensor NLRP3 protein can be triggered by PAMPs, DAMPs, and a range of diverse external stimuli such as infection and injury. NLRP3 inflammasome activation is a tightly regulated system and requires two signals for activation, as depicted in Figure 2. The priming signal results in an increased expression of NLRP3, pro-IL-1β, and pro-IL-18, as the nuclear factor-kappa beta (NF-κβ) pathway is activated in response to a stimulus [58]. The priming step of inflammasome activation is tightly controlled by a series of different post translational modifications of NLRP3, ASC, and caspase-1, including ubiquitination, phosphorylation, and sumoylation [59].
Figure 2.
Hypothetical mechanism for the role of NLRP3 in glaucoma. Activation of the NLRP3 inflammasome requires two signals for activation. Signal 1 results in transcriptional upregulation of Pro-IL-1β and NLRP3 protein via the NF-κβ pathway. Stress to the ONH is thought to activate the NF-κβ pathway. Signal 2 results in the oligomerisation of ASC, NLRP3, and pro-caspase-1 to form the NLRP3 inflammasome. NLRP3 inflammasome oligomerisation results in cleavage of pro-caspase-1 into active caspase-1, which can in turn cleave pro-IL-1β into IL-1β, which can leave the cell to drive inflammation. ROS or extracellular ATP are proposed to result in inflammasome oligomerisation in glaucoma. ONH—optic nerve head; ROS—reactive oxygen species; ATP—adenosine triphosphate; RGC—retinal ganglion cell; DAMPS—danger associated molecular patterns; IL-1β—interluekin-1beta; ASC—apoptosis-associated speck-like protein containing a caspase-recruitment domain; NLRP3—NLR family pyrin domain containing 3; NF-κβ—nuclear factor-kappa beta.
Triggering or activation is the second signal needed for inflammasome activation, and involves the oligomerisation of NLRP3 in its inactive form with procaspase-1 and ASC. Generally the activation step of inflammasome oligomerisation is a result of potassium efflux from the cell [59]. This results in the cleavage of procaspase-1 into caspase-1 and, as a result, pro-IL-1β and pro-IL-18 into active IL-1ß and IL-18, which are subsequently secreted from the cell [60,61]. As well as producing pro-inflammatory cytokines, active caspase-1 also results in the cleavage of gasdermin-D (GSDMD), a pore forming protein, resulting in pyroptosis, a form of cell death [62]. In the chronic diseases driven by inflammation, such as type 2 diabetes and cryopyrin-associated periodic syndromes (CAPS), the NLRP3 inflammasome is dysregulated, resulting in the uncontrolled release of pro-inflammatory cytokine IL-1β, which drives inflammation in such diseases [63,64].
4. IL-1 Signaling in the Eye
Interleukin 1 receptor 1 (IL-1R1) mediates interleukin 1 (IL-1) (IL-1α and IL-1β) signaling. Despite many studies alluding to the activation of microglia by IL-1, there is conflicting evidence in the literature that suggests that microglia do not express IL-1R1 [65]. However, IL-1R1 is clearly expressed in endothelial cells, astrocytes, and neurons in the eye [66]. IL-1α and IL-1β have been detected in various parts of the eye, including in tear fluid and corneal epithelial cells [67]. In the brain, IL-1 stimulates endothelial IL-1R1, which then produces factors that drive microglial inflammatory gene expression [68]. This process may also happen in the retina, as IL-1-stimulated inflammatory cytokine expression was largely abolished after the depletion of microglia, but the restoration of IL-1R1 on endothelial, but not microglial, cells restored IL-1 induced inflammatory gene expression [66]. Retinal pigment epithelial cells and the trabecular meshwork of humans also express IL-1R1 [69]. Fibroblasts are also known to highly express IL-1R1, and may contribute to microglial stimulation. Fibrosis is a known pathological response in the lamina cribrosa in glaucoma [70,71], and it is likely that there is a cross talk between the lamina cribrosa and retinal cells, which ultimately leads to the activation of microglial cells and subsequent RGC death [65,72].
5. NLRP3 in the Eye
The NLRP3 inflammasome is constitutively expressed in various parts of the eye including the retinal pigment epithelium and ONH astrocytes in both human and mice, indicating the importance of this mediator in the defense system of the eye [73,74,75]. NLRP3 is also expressed in many other cells of the eye, including the ONH, retinal microglia, Müller cells [76,77,78], and astrocytes [78,79]. NLRP3 expression is not limited to the retina and ONH, and is found throughout the eye in the conjunctiva, trabecular meshwork, retinal pigment epithelium, and corneal epithelial cells in disease states [80,81,82,83,84].
6. The NLRP3 Inflammasome in Glaucoma
Dysregulation of the NLRP3 inflammasome has been implicated in several neurodegenerative diseases, including Alzheimer’s disease and multiple sclerosis [85]. Increased levels of IL-1β mRNA and protein have also been observed in the blood of glaucoma patients compared with controls, suggesting activation of the NLRP3 inflammasome in glaucoma [35]. Furthermore, activation of the NLRP3 inflammasome has been associated with the induction of IL-1β and death of RGCs in mouse models of acute glaucoma via optic nerve (ON) crush. [64,86]. In an inducible mouse model of glaucoma, the use of fluorescent reporter mice to track inflammasome activation demonstrated that NLRP3 inflammasome activation occurs early in the ONH, following IOP elevation, and coincides with the induction of pro-inflammatory cytokines and Iba1+, a microglia marker, immune cells in the ONH [87]. This upregulation of inflammatory genes occurs as early as 7 days post IOP elevation in the ONH and before the induction of inflammatory genes occurs in the retina [79]. With the use of knockout mice lacking various components of the NLRP3 inflammasome, we can conclude that the NLRP3 inflammasome activation is required for RGC death [64,79,88]. These studies support the hypothesis that “danger” signals in the eye, IOP elevation being one, activates the NLRP3 inflammasome pathway in the glial cells of the ONH and retina, resulting in neurotoxic inflammation, axon degeneration, and subsequent death of RGCs in glaucoma models.
7. Proposed Mechanism for the Role of NLRP3 in Glaucoma
Chronic inflammation from unknown causes, ageing, or genetics may be initiating factors [1], and may directly lead to the activation of glial cells or, alternatively, may lead to damage to the trabecular meshwork [89]. NLRP3 is expressed and activated in the trabecular meshwork as a result of oxidative stress, leading to elevated IOP, further exacerbating the initiation of inflammation [80,90].
Initial injury may also be due to increased IOP resulting in mechanical stress on various cells such as fibroblasts in the sclera [91,92], and subsequently or alternatively, lamina cribrosa astrocytes of the ONH. Fibroblasts and astrocytes are known to express high levels of IL-1R1, and the NLRLP3 inflammasome is known to be activated in these cell types [65,78,93,94,95].
A number of risk factors trigger common pathological endpoints resulting in glaucomatous optic neuropathy [10]. In glaucoma, the ON glial cells become activated, which in turn damage the axons leaving the eye and further trigger inflammatory cell recruitment to the injury sites [79,96]; ONH astrocytes constitutively express NLRP3 and injury to the ONH may also activate the NLRP3 inflammasome pathway in these cells [79,96]. As Figure 2 depicts, the NF-κβ pathway is activated by DAMPs or injury to the ON, resulting in transcriptional upregulation of pro-IL-1β and NLRP3 [58]. A second signal, generally resulting in potassium efflux from the cell, results in the oligomerisation of NLRP3, ASC, and pro-caspase-1 to form the inflammasome complex [30,59]. We hypothesise that extracellular ATP binding to the ligand-gated ion channel purinergic type 2 receptor 7 (p2x7r) or ROS produced by oxidative stress as a result of ageing are some methods of inflammasome activation in glaucoma that may be independent of IOP [30,76,97]. The ATP concentration is higher in the aqueous humour of glaucoma patients compared with the controls, and increased levels of ATP in the glaucomatous eye are thought to be released from cells damaged or stressed by elevated IOP [98,99,100]. However, sheer stress and cell swelling in tissues can also increase extracellular ATP in the absence of elevated IOP [101,102]. Moreover, it has been proposed that ROS produced from reactive astrocytes contribute to early axonal damage within the ONH in glaucoma [103]. Amyloid beta, a known inflammasome activator in the brain microglia, is also increased in the glaucomatous eye compared with normal eyes, and may result in retinal glial cell activation [104,105]. TGF-β, a profibrotic cytokine is found in the aqueous humour and ONH astrocytes in glaucoma patients. TGF-β is involved in damage to the ONH in glaucoma [106] and TGF-β can induce activation of the NLRP3 inflammasome, which has also been demonstrated to be involved in fibrosis [107]. Upon inflammasome activation, pro-caspase-1 is cleaved into its active form caspase-1, which can in turn cleave pro-IL-1β into its active form IL-1β, which is then secreted from the cell [61]. In glaucoma, IL-1β was found to be one of the first inflammatory cytokines upregulated in the ONH, and is a powerful stimulus for immune cell recruitment, further supporting the role of neurotoxic inflammation as a significant contributor to the neurodegenerative process of glaucoma [108,109]. Therefore, pharmacologically targeting NLRP3 may serve as a neuroprotective therapy to prevent the progression of glaucoma.
8. NLRP3 Inflammasome as a Target for Therapy in Glaucoma
There is an obvious need for the development of new medications for glaucoma, given that current treatments work to lower IOP only. However, these treatments are not successful in all patients [110,111]. NLRP3 may be a promising IOP independent target for the treatment of glaucoma [112]. Several studies demonstrate that the inhibition of NLRP3 activation significantly inhibits the death of RGCs in experimental models of retinal ischemia/reperfusion injury (acute glaucoma) [113,114,115,116]. Blockade of the P2x7r, which can act as signal 2 in inflammasome activation with A438079, a P2x7r inhibitor, also attenuates RGC death by inhibiting inflammasome activation. Additionally in an in-vitro model, ATP, which binds to the P2x7r, was shown to induce inflammation in the retina by activating the NLRP3 inflammasome [76]. High-mobility group box 1 (HMGB1), which is often released from damaged cells, is known to be released upon rapid IOP elevation and is also a known DAMP that activates Toll Like Receptor 4 (TLR-4) to activate the NF-kB pathway. In an acute glaucoma mouse model, the release of HMGB1 resulted in increased levels of NLRP3 and IL-1B. Inhibition of HMGB1 resulted in reduced NLRP3 and IL-1B levels, which also reduced RGC death and glaucoma severity [117].
However, in the more clinically relevant microbead-induced mouse model of glaucoma, where IOP was elevated, treatment with a commercially available NLRP3-specific inhibitor (MCC950) [118] was also shown to prevent axon degeneration and death of RGCs [119]. In this microbead-induced model of glaucoma, 4 weeks of elevated IOP resulted in a 25–30% loss of RGCs, which was significantly attenuated following a three-times a week treatment with intraperitoneal MCC950. Mice treated with MCC950 displayed axon and RGC densities equal to that of the non-glaucoma controls. MCC950 injection, however, had no effect on IOP elevation in the treated mice [119]. Currently, this is the only study that has evaluated a NLRP3-specific inhibitor in an inducible model of glaucoma [119]. Collectively, these studies provide evidence that NLRP3 inhibition may be a novel therapeutic strategy to protect RGCs and prevent axon degeneration in glaucoma.
MCC950, a small molecule drug, however, does present with some drawbacks, including the very short half-life of MCC950 of only 3 h [120]. This means the drug must be administered regularly either systemically or by multi-intravitreal injections, both of which are not desirable for diseases of the eye. Other limitations of small molecule inhibitors include that they are often not fully characterised, and many are not completely specific to their targets [121,122]. Therefore, there has been interest in recent years in the development of biologics, such as antibodies for the treatment of many chronic conditions, as they have revolutionised the treatment and modification of many diseases including some cancers and autoimmune rheumatic diseases [121,122]. Biologics are well known to have a longer half-life, allowing for longer dosing intervals because of their larger size, which is an important factor in the treatment of glaucoma [122,123]; they are also recycled by the body and have a high affinity and potency. The major advantage of biologic molecules for treatment is the specificity of their mechanisms of actions preventing off target effects [122]. Financial cost in the long run is also an advantage of biologic therapies, as they are expected to deliver a better economic return than small molecules [121]. The use and approval of biologics in medicine is increasing each year, targeting extracellular or cell membrane proteins [123]. However, the delivery mechanisms of antibodies into cells to target intracellular molecules is a major obstacle [123]. The current inhibitors of NLRP3 and IL-1β are outlined in Table 2. It is important to note that, generally, biologics targeting the NLRP3/IL-1β pathway target secreted IL-1β directly, like Canakinumab to suppress its inflammatory responses, which poses a serious risk of fatal infection, as IL-1β cannot be produced in response to an infection [49]. Targeting NLRP3 to supress IL-1β production means IL-1β can still be produced by other pathways if the body encounters an acute infection [124]. Anakinra is another biologic that targets the NLRP3 pathway binds to IL-1R1 to inhibit IL-1β binding to IL-1R1, as this can further activate the NLRP3 inflammasome [125]. There is a number of small molecule inhibitors that target NLRP3 to suppress its effects or inhibit NLRP3 inflammasome oligomerisation [114,126,127,128,129].
Table 2.
Novel IL-1β and NLRP3 inhibitors.
| Inhibitors | Type | Mechanism of Action | Reference |
|---|---|---|---|
| Canakinumab | Biologic | Inhibits binding of IL-1β to IL-1R1 | [49] |
| Anakinra | Biologic | Binds to IL-1 receptor to inhibit IL-1β binding | [125] |
| MCC950 | Small molecule | Inhibits caspase-1 activation by binding to NLRP3 | [126] |
| CY-09 | Small molecule | Inhibits NLRP3 ATPase activity by binding to the ATP-binding motif of the NACHT domain | [127] |
| Tranilast | Small molecule | Suppresses NLRP3 assembly by binding to NACHT domain | [128] |
| IC-100 | Biologic | Inhibits adaptor ASC | [130] |
| Argablin | Small molecule | Inhibits NLRP3 activation (specific mechanism not known) | [129] |
| Cholchicine | Small molecule | Inhibits expression of components of NLRP3 | [113] |
IL-1β—interleukin-1beta; IL-1R1—interleukin 1 receptor 1; NLRP3—NLR family pyrin domain containing 3; ATP—adenosine triphosphate; NACHT—a central nucleotide-binding and oligomerization domain; ASC—Apoptosis-associated speck-like protein containing a caspase-recruitment domain.
9. Conclusions
Elevated IOP is the only modifiable risk factor in glaucoma, and IOP lowering therapies are unsuccessful in many patients. There is a need for IOP independent therapies to treat or slow glaucoma progression. There is significant evidence to support the role of inflammation in the pathogenesis of glaucoma. In particular, the NLRP3 inflammasome pathway in glial cells of the ONH and retina appears to play a critical role in axon degeneration and death of RGCs in glaucoma. The NLRP3 inflammasome therefore appears to be a strong target for therapy development in glaucoma. A biologic therapy with a long dosing interval would be desirable to avoid multiple dosing. Additional studies are now required to further investigate current and novel inhibitors of the NLRP3 inflammasome pathway for glaucoma treatment.
Author Contributions
Conceptualization, S.C. and V.M. All of the authors have participated in the writing, review, and editing of the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Department for the Economy, NI, and Santen Pharma.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Weinreb R.N., Aung T., Medeiros F.A. The pathophysiology and treatment of glaucoma: A review. JAMA. 2014;311:1901–1911. doi: 10.1001/jama.2014.3192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Quigley H.A., Broman A.T. The number of people with glaucoma worldwide in 2010 and 2020. Br. J. Ophthalmol. 2006;90:262–267. doi: 10.1136/bjo.2005.081224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tham Y.C., Li X., Wong T.Y., Quigley H.A., Aung T., Cheng C.Y. Global prevalence of glaucoma and projections of glaucoma burden through 2040: A systematic review and meta-analysis. Ophthalmology. 2014;121:2081–2090. doi: 10.1016/j.ophtha.2014.05.013. [DOI] [PubMed] [Google Scholar]
- 4.Jonas J.B., Aung T., Bourne R.R., Bron A.M., Ritch R., Panda-Jonas S. Glaucoma. Lancet. 2017;390:2183–2193. doi: 10.1016/S0140-6736(17)31469-1. [DOI] [PubMed] [Google Scholar]
- 5.Weinreb R.N., Khaw P.T. Primary open-angle glaucoma. Lancet. 2004;363:1711–1720. doi: 10.1016/S0140-6736(04)16257-0. [DOI] [PubMed] [Google Scholar]
- 6.Kwon Y.H., Fingert J.H., Kuehn M.H., Alward W.L. Primary Open-Angle Glaucoma. N. Engl. J. Med. 2009;360:1113–1124. doi: 10.1056/NEJMra0804630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Labkovich M., Jacobs E.B., Bhargava S., Pasquale L.R., Ritch R. Ginkgo Biloba Extract in Ophthalmic and Systemic Disease, With a Focus on Normal-Tension Glaucoma. Asia Pac. J. Ophthalmol. 2020;9:215–225. doi: 10.1097/APO.0000000000000279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Garway-Heath D.F., Crabb D.P., Bunce C., Lascaratos G., Amalfitano F., Anand N., Azuara-Blanco A., Bourne R.R., Broadway D.C., Cunliffe I.A., et al. Latanoprost for open-angle glaucoma (UKGTS): A randomised, multicentre, placebo-controlled trial. Lancet. 2015;385:1295–1304. doi: 10.1016/S0140-6736(14)62111-5. [DOI] [PubMed] [Google Scholar]
- 9.Esporcatte B.L.B., Tavares I.M. Normal-tension glaucoma: An update. Arq. Bras. Oftalmol. 2016;79:270–276. doi: 10.5935/0004-2749.20160077. [DOI] [PubMed] [Google Scholar]
- 10.Alqawlaq S., Flanagan J.G., Sivak J.M. All roads lead to glaucoma: Induced retinal injury cascades contribute to a common neurodegenerative outcome. Exp. Eye Res. 2019;183:88–97. doi: 10.1016/j.exer.2018.11.005. [DOI] [PubMed] [Google Scholar]
- 11.Knox D.L., Eagle R.C., Jr., Green W.R. Optic nerve hydropic axonal degeneration and blocked retrograde axoplasmic transport: Histopathologic features in human high-pressure secondary glaucoma. Arch. Ophthalmol. 2007;125:347–353. doi: 10.1001/archopht.125.3.347. [DOI] [PubMed] [Google Scholar]
- 12.Renner M., Stute G., Alzureiqi M., Reinhard J., Wiemann S., Schmid H., Faissner A., Dick H.B., Joachim S.C. Optic Nerve Degeneration after Retinal Ischemia/Reperfusion in a Rodent Model. Front. Cell. Neurosci. 2017;11:254. doi: 10.3389/fncel.2017.00254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hare W.A., WoldeMussie E., Weinreb R.N., Ton H., Ruiz G., Wijono M., Feldmann B., Zangwill L., Wheeler L. Efficacy and Safety of Memantine Treatment for Reduction of Changes Associated with Experimental Glaucoma in Monkey, II: Structural Measures. Investig. Opthalmol. Vis. Sci. 2004;45:2640–2651. doi: 10.1167/iovs.03-0567. [DOI] [PubMed] [Google Scholar]
- 14.Schultheiss M., Voykov B., Klemm M., Gross U., Schultheiss H.-P., Spitzer M.S., Casagrande M. Scleral Inflammation around Collector Channels in Eyes with Primary Open-Angle Glaucoma. Ocul. Immunol. Inflamm. 2020;13:1–7. doi: 10.1080/09273948.2020.1746355. [DOI] [PubMed] [Google Scholar]
- 15.Zaleska-Żmijewska A., Strzemecka E., Wawrzyniak Z.M., Szaflik J.P. Extracellular MMP-9-Based Assessment of Ocular Surface Inflammation in Patients with Primary Open-Angle Glaucoma. J. Ophthalmol. 2019;2019:1–7. doi: 10.1155/2019/1240537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Baudouin C., Kolko M., Melik-Parsadaniantz S., Messmer E.M. Inflammation in Glaucoma: From the back to the front of the eye, and beyond. Prog. Retin. Eye Res. 2021;83:100916. doi: 10.1016/j.preteyeres.2020.100916. [DOI] [PubMed] [Google Scholar]
- 17.Kamat S.S., Gregory M.S., Pasquale L.R. The Role of the Immune System in Glaucoma: Bridging the Divide between Immune Mechanisms in Experimental Glaucoma and the Human Disease. Semin. Ophthalmol. 2016;31:147–154. doi: 10.3109/08820538.2015.1114858. [DOI] [PubMed] [Google Scholar]
- 18.Ahmed F., Brown K.M., Stephan D.A., Morrison J.C., Johnson E.C., Tomarev S.I. Microarray Analysis of Changes in mRNA Levels in the Rat Retina after Experimental Elevation of Intraocular Pressure. Investig. Ophtalmol. Vis. Sci. 2004;45:1247–1258. doi: 10.1167/iovs.03-1123. [DOI] [PubMed] [Google Scholar]
- 19.Qu J., Jakobs T.C. The Time Course of Gene Expression during Reactive Gliosis in the Optic Nerve. PLoS ONE. 2013;8:e67094. doi: 10.1371/journal.pone.0067094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yang Z., Quigley H.A., Pease M., Yang Y., Qian J., Valenta D., Zack D. Changes in Gene Expression in Experimental Glaucoma and Optic Nerve Transection: The Equilibrium between Protective and Detrimental Mechanisms. Investig. Opthalmol. Vis. Sci. 2007;48:5539–5548. doi: 10.1167/iovs.07-0542. [DOI] [PubMed] [Google Scholar]
- 21.Williams P.A., Marsh-Armstrong N., Howell G.R., Bosco A., Danias J., Simon J., Di Polo A., Kuehn M., Przedborski S., Raff M., et al. Neuroinflammation in glaucoma: A new opportunity. Exp. Eye Res. 2017;157:20–27. doi: 10.1016/j.exer.2017.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Oikawa K., Hoeve J.N.V., Teixeira L.B.C., Snyder K.C., Kiland J.A., Ellinwood N.M., McLellan G.J. Sub-region-Specific Optic Nerve Head Glial Activation in Glaucoma. Mol. Neurobiol. 2020;57:2620–2638. doi: 10.1007/s12035-020-01910-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sun D., Qu J., Jakobs T.C. Reversible reactivity by optic nerve astrocytes. Glia. 2013;61:1218–1235. doi: 10.1002/glia.22507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Hernandez M.R., Agapova O.A., Yang P., Salvador-Silva M., Ricard C.S., Aoi S. Differential gene expression in astrocytes from human normal and glaucomatous optic nerve head analyzed by cDNA microarray. Glia. 2002;38:45–64. doi: 10.1002/glia.10051. [DOI] [PubMed] [Google Scholar]
- 25.Bordone M.P., Fleitas M.F.G., Pasquini L.A., Bosco A., Sande P.H., Rosenstein R.E., Dorfman D. Involvement of microglia in early axoglial alterations of the optic nerve induced by experimental glaucoma. J. Neurochem. 2017;142:323–337. doi: 10.1111/jnc.14070. [DOI] [PubMed] [Google Scholar]
- 26.Yuan L., Neufeld A.H. Activated microglia in the human glaucomatous optic nerve head. J. Neurosci. Res. 2001;64:523–532. doi: 10.1002/jnr.1104. [DOI] [PubMed] [Google Scholar]
- 27.Tezel G., Wax M.B. Glial Modulation of Retinal Ganglion Cell Death in Glaucoma. J. Glaucoma. 2003;12:63–68. doi: 10.1097/00061198-200302000-00014. [DOI] [PubMed] [Google Scholar]
- 28.Adornetto A., Russo R., Parisi V. Neuroinflammation as a target for glaucoma therapy. Neural Regen. Res. 2019;14:391–394. doi: 10.4103/1673-5374.245465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Soto I., Howell G.R. The Complex Role of Neuroinflammation in Glaucoma. Cold Spring Harb. Perspect. Med. 2014;4:a017269. doi: 10.1101/cshperspect.a017269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Chen M., Luo C., Zhao J., Devarajan G., Xu H. Immune regulation in the aging retina. Prog. Retin. Eye Res. 2019;69:159–172. doi: 10.1016/j.preteyeres.2018.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xu H., Chen M., Forrester J.V. Para-inflammation in the aging retina. Prog. Retin. Eye Res. 2009;28:348–368. doi: 10.1016/j.preteyeres.2009.06.001. [DOI] [PubMed] [Google Scholar]
- 32.Ramírez A.I., de Hoz R., Albarral J.A.F., Salobrar-Garcia E., Rojas B., Valiente-Soriano F.J., Avilés-Trigueros M., Villegas-Pérez M.P., Vidal-Sanz M., Triviño A., et al. Time course of bilateral microglial activation in a mouse model of laser-induced glaucoma. Sci. Rep. 2020;10:1–17. doi: 10.1038/s41598-020-61848-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ramírez A.I., Albarral J.A.F., De Hoz R., López-Cuenca I., Salobrar-García E., Rojas P., Valiente-Soriano F.J., Avilés-Trigueros M., Villegas-Pérez M.P., Vidal-Sanz M., et al. Microglial changes in the early aging stage in a healthy retina and an experimental glaucoma model. Prog. Brain Res. 2020;256:125–149. doi: 10.1016/bs.pbr.2020.05.024. [DOI] [PubMed] [Google Scholar]
- 34.Ferrucci L., Fabbri E. Inflammageing: Chronic inflammation in ageing, cardiovascular disease, and frailty. Nat. Rev. Cardiol. 2018;15:505–522. doi: 10.1038/s41569-018-0064-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Markiewicz L., Pytel D., Mucha B., Szymanek K., Szaflik J., Szaflik J.P., Majsterek I. Altered Expression Levels of MMP1, MMP9, MMP12, TIMP1, and IL-1β as a Risk Factor for the Elevated IOP and Optic Nerve Head Damage in the Primary Open-Angle Glaucoma Patients. Biomed. Res. Int. 2015;2015 doi: 10.1155/2015/812503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chono I., Miyazaki D., Miyake H., Komatsu N., Ehara F., Nagase D., Kawamoto Y., Shimizu Y., Ideta R., Inoue Y. High interleukin-8 level in aqueous humor is associated with poor prognosis in eyes with open angle glaucoma and neovascular glaucoma. Sci. Rep. 2018;8:1–11. doi: 10.1038/s41598-018-32725-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Echevarria F., Formichella C.R., Sappington R.M. Interleukin-6 Deficiency Attenuates Retinal Ganglion Cell Axonopathy and Glaucoma-Related Vision Loss. Front. Neurosci. 2017;11:318. doi: 10.3389/fnins.2017.00318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Zhou X., Li F., Kong L., Tomita H., Li C., Cao W. Involvement of Inflammation, Degradation, and Apoptosis in a Mouse Model of Glaucoma. J. Biol. Chem. 2005;280:31240–31248. doi: 10.1074/jbc.M502641200. [DOI] [PubMed] [Google Scholar]
- 39.Fernández-Albarral J.A., Salazar J.J., de Hoz R., Marco E.M., Martín-Sánchez B., Flores-Salguero E., Salobrar-García E., López-Cuenca I., Barrios-Sabador V., Avilés-Trigueros M., et al. Retinal Molecular Changes Are Associated with Neuroinflammation and Loss of RGCs in an Experimental Model of Glaucoma. Int. J. Mol. Sci. 2021;22:2066. doi: 10.3390/ijms22042066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Jiang S., Kametani M., Chen D.F. Adaptive Immunity: New Aspects of Pathogenesis Underlying Neurodegeneration in Glaucoma and Optic Neuropathy. Front. Immunol. 2020;11:65. doi: 10.3389/fimmu.2020.00065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bosco A., Romero C.O., Breen K.T., Chagovetz A.A., Steele M.R., Ambati B.K., Vetter M.L. Neurodegeneration severity can be predicted from early microglia alterations monitored in vivo in a mouse model of chronic glaucoma. Dis. Model. Mech. 2015;8:443–455. doi: 10.1242/dmm.018788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Roh M., Zhang Y., Murakami Y., Thanos A., Lee S.C., Vavvas D.G., Benowitz L.I., Miller J.W. Etanercept, a Widely Used Inhibitor of Tumor Necrosis Factor-α (TNF-α), Prevents Retinal Ganglion Cell Loss in a Rat Model of Glaucoma. PLoS ONE. 2012;7:e40065. doi: 10.1371/journal.pone.0040065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Panchal S.S., Patidar R.K., Jha A.B., Allam A.A., Ajarem J., Butani S.B. Anti-Inflammatory and Antioxidative Stress Effects of Oryzanol in Glaucomatous Rabbits. J. Ophthalmol. 2017;2017:1–9. doi: 10.1155/2017/1468716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Li S.-Y., Fung F.K.C., Fu Z.J., Wong D., Chan H.H., Lo A.C.Y. Anti-Inflammatory Effects of Lutein in Retinal Ischemic/Hypoxic Injury: In Vivo and In Vitro Studies. Investig. Opthalmol. Vis. Sci. 2012;53:5976–5984. doi: 10.1167/iovs.12-10007. [DOI] [PubMed] [Google Scholar]
- 45.Wei H.Y., Zhang Y.J., Zhao S.Z. Puerarin regulates neovascular glaucoma through pigment epithelium-derived growth factor-induced NF-κB signaling pathway. Mol. Med. Rep. 2018;17:7866–7874. doi: 10.3892/mmr.2018.8800. [DOI] [PubMed] [Google Scholar]
- 46.Yang Q., Li Y., Luo L. Effect of Myricetin on Primary Open-angle Glaucoma. Transl. Neurosci. 2018;9:132–141. doi: 10.1515/tnsci-2018-0020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Ridker P.M., Everett B.M., Thuren T., MacFadyen J.G., Chang W.H., Ballantyne C., Fonseca F., Nicolau J., Koenig W., Anker S.D., et al. Antiinflammatory Therapy with Canakinumab for Atherosclerotic Disease. N. Engl. J. Med. 2017;377:1119–1131. doi: 10.1056/NEJMoa1707914. [DOI] [PubMed] [Google Scholar]
- 48.Rider P., Carmi Y., Cohen I. Biologics for Targeting Inflammatory Cytokines, Clinical Uses, and Limitations. Int. J. Cell Biol. 2016;2016:9259646. doi: 10.1155/2016/9259646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Alten R., Gomez-Reino J., Durez P., Beaulieu A., Sebba A., Krammer G., Preiss R., Arulmani U., Widmer A., Gitton X., et al. Efficacy and safety of the human anti-IL-1β monoclonal antibody canakinumab in rheumatoid arthritis: Results of a 12-week, Phase II, dose-finding study. BMC Musculoskelet. Disord. 2011;12:153. doi: 10.1186/1471-2474-12-153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Micallef J., Soeiro T., Jonville-Béra A.-P. Non-steroidal anti-inflammatory drugs, pharmacology, and COVID-19 infection. Therapies. 2020;75:355–362. doi: 10.1016/j.therap.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jiang K., Chen J., Tai L., Liu C., Chen X., Wei G., Lu W., Pan W. Inhibition of post-trabeculectomy fibrosis via topically instilled antisense oligonucleotide complexes co-loaded with fluorouracil. Acta Pharm. Sin. B. 2020;10:1754–1768. doi: 10.1016/j.apsb.2020.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Krishnan A., Kocab A.J., Zacks D.N., Marshak-Rothstein A., Gregory-Ksander M. A small peptide antagonist of the Fas receptor inhibits neuroinflammation and prevents axon degeneration and retinal ganglion cell death in an inducible mouse model of glaucoma. J. Neuroinflamm. 2019;16:1–15. doi: 10.1186/s12974-019-1576-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Amarante-Mendes G.P., Adjemian S., Branco L.M., Zanetti L., Weinlich R., Bortoluci K.R. Pattern Recognition Receptors and the Host Cell Death Molecular Machinery. Front. Immunol. 2018;9:2379. doi: 10.3389/fimmu.2018.02379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Kim Y.K., Shin J.-S., Nahm M.H. NOD-Like Receptors in Infection, Immunity, and Diseases. Yonsei Med. J. 2016;57:5–14. doi: 10.3349/ymj.2016.57.1.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zhou W., Chen C., Chen Z., Liu L., Jiang J., Wu Z., Zhao M., Chen Y. NLRP3: A Novel Mediator in Cardiovascular Disease. J. Immunol. Res. 2018;2018:1–8. doi: 10.1155/2018/5702103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Schroder K., Tschopp J. The Inflammasomes. Cell. 2010;140:821–832. doi: 10.1016/j.cell.2010.01.040. [DOI] [PubMed] [Google Scholar]
- 57.Zahid A., Li B., Kombe A.J.K., Jin T., Tao J. Pharmacological Inhibitors of the NLRP3 Inflammasome. Front. Immunol. 2019;10:2538. doi: 10.3389/fimmu.2019.02538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Baldrighi M., Mallat Z., Li X. NLRP3 inflammasome pathways in atherosclerosis. Atherosclerosis. 2017;267:127–138. doi: 10.1016/j.atherosclerosis.2017.10.027. [DOI] [PubMed] [Google Scholar]
- 59.McKee C.M., Coll R.C. NLRP3 inflammasome priming: A riddle wrapped in a mystery inside an enigma. J. Leukoc. Biol. 2020;108:937–952. doi: 10.1002/JLB.3MR0720-513R. [DOI] [PubMed] [Google Scholar]
- 60.Chu J., Thomas L.M., Watkins S.C., Franchi L., Núñez G., Salter R.D. Cholesterol-dependent cytolysins induce rapid release of mature IL-1beta from murine macrophages in a NLRP3 inflammasome and cathepsin B-dependent manner. J. Leukoc. Biol. 2009;86:1227–1238. doi: 10.1189/jlb.0309164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.An N., Gao Y., Si Z., Zhang H., Wang L., Tian C., Yuan M., Yang X., Li X., Shang H., et al. Regulatory Mechanisms of the NLRP3 Inflammasome, a Novel Immune-Inflammatory Marker in Cardiovascular Diseases. Front. Immunol. 2019;10:1592. doi: 10.3389/fimmu.2019.01592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Próchnicki T., Mangan M.S., Latz E. Recent insights into the molecular mechanisms of the NLRP3 inflammasome activation. F1000Research. 2016:5. doi: 10.12688/f1000research.8614.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Menu P., Vince J.E. The NLRP3 inflammasome in health and disease: The good, the bad and the ugly. Clin. Exp. Immunol. 2011;166:1–15. doi: 10.1111/j.1365-2249.2011.04440.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Puyang Z., Feng L., Chen H., Liang P., Troy J.B., Liu X. Retinal Ganglion Cell Loss is Delayed Following Optic Nerve Crush in NLRP3 Knockout Mice. Sci. Rep. 2016;6:20998. doi: 10.1038/srep20998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Liu X., Nemeth D.P., McKim D.B., Zhu L., DiSabato D.J., Berdysz O., Gorantla G., Oliver B., Witcher K.G., Wang Y., et al. Cell-Type-Specific Interleukin 1 Receptor 1 Signaling in the Brain Regulates Distinct Neuroimmune Activities. Immunity. 2019;50:317–333. doi: 10.1016/j.immuni.2018.12.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Todd L., Palazzo I., Suarez L., Liu X., Volkov L., Hoang T.V., Campbell W.A., Blackshaw S., Quan N., Fischer A.J. Reactive microglia and IL1β/IL-1R1-signaling mediate neuroprotection in excitotoxin-damaged mouse retina. J. Neuroinflamm. 2019;16:1–19. doi: 10.1186/s12974-019-1505-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Solomon A., Dursun D., Liu Z., Xie Y., Macri A., Pflugfelder S.C. Pro- and anti-inflammatory forms of interleukin-1 in the tear fluid and conjunctiva of patients with dry-eye disease. Investig. Ophthalmol. Vis. Sci. 2001;42:2283–2292. [PubMed] [Google Scholar]
- 68.Proescholdt M.G., Chakravarty S., Foster J.A., Foti S.B., Briley E.M., Herkenham M. Intracerebroventricular but not intravenous interleukin-1beta induces widespread vascular-mediated leukocyte infiltration and immune signal mRNA expression followed by brain-wide glial activation. Neuroscience. 2002;112:731–749. doi: 10.1016/S0306-4522(02)00048-9. [DOI] [PubMed] [Google Scholar]
- 69.Liu R., Wang T., Zhang B., Qin L., Wu C., Li Q., Ma L. Lutein and Zeaxanthin Supplementation and Association With Visual Function in Age-Related Macular Degeneration. Investig. Opthalmol. Vis. Sci. 2015;56:252–258. doi: 10.1167/iovs.14-15553. [DOI] [PubMed] [Google Scholar]
- 70.Wallace D.M., O’Brien C.J. The role of lamina cribrosa cells in optic nerve head fibrosis in glaucoma. Exp. Eye Res. 2016;142:102–109. doi: 10.1016/j.exer.2014.12.006. [DOI] [PubMed] [Google Scholar]
- 71.Song A., Zhu L., Gorantla G., Berdysz O., Amici S.A., Guerau-De-Arellano M., Madalena K.M., Lerch J.K., Liu X., Quan N. Salient type 1 interleukin 1 receptor expression in peripheral non-immune cells. Sci. Rep. 2018;8:723. doi: 10.1038/s41598-018-19248-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Namvar S., Woolf A.S., Zeef L.A., Wilm T., Wilm B., Herrick S.E. Functional molecules in mesothelial-to-mesenchymal transition revealed by transcriptome analyses. J. Pathol. 2018;245:491–501. doi: 10.1002/path.5101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Malsy J., Alvarado A.C., Lamontagne J.O., Strittmatter K., Marneros A.G. Author response: Distinct effects of complement and of NLRP3- and non-NLRP3 inflammasomes for choroidal neovascularization. Elife. 2020;11:60194. doi: 10.7554/eLife.60194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Doktor F., Prager P., Wiedemann P., Kohen L., Bringmann A., Hollborn M. Hypoxic expression of NLRP3 and VEGF in cultured retinal pigment epithelial cells: Contribution of P2Y2 receptor signaling. Purinergic Signal. 2018;14:471–484. doi: 10.1007/s11302-018-9631-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.AlBalawi F., Lu W., Beckel J.M., Lim J.C., McCaughey S.A., Mitchell C.H. The P2X7 Receptor Primes IL-1β and the NLRP3 Inflammasome in Astrocytes Exposed to Mechanical Strain. Front. Cell. Neurosci. 2017;11:227. doi: 10.3389/fncel.2017.00227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zhang Y., Xu Y., Sun Q., Xue S., Guan H., Ji M. Activation of P2X7R- NLRP3 pathway in Retinal microglia contribute to Retinal Ganglion Cells death in chronic ocular hypertension (COH) Exp. Eye Res. 2019;188:107771. doi: 10.1016/j.exer.2019.107771. [DOI] [PubMed] [Google Scholar]
- 77.Li W., Liu X., Tu Y., Ding D., Yi Q., Sun X., Wang Y., Wang K., Zhu M., Mao J. Dysfunctional Nurr1 promotes high glucose-induced Müller cell activation by up-regulating the NF-κB/NLRP3 inflammasome axis. Neuropeptides. 2020;82:20. doi: 10.1016/j.npep.2020.102057. [DOI] [PubMed] [Google Scholar]
- 78.Pronin A., Pham D., An W., Dvoriantchikova G., Reshetnikova G., Qiao J., Kozhekbaeva Z., Reiser A.E., Slepak V.Z., Shestopalov V.I. Inflammasome Activation Induces Pyroptosis in the Retina Exposed to Ocular Hypertension Injury. Front. Mol. Neurosci. 2019;12:36. doi: 10.3389/fnmol.2019.00036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Gregory M.S., Fei F., Krishnan A., Tzeng T., Deepa S. Destructive neuroinflammation triggered by activation of the NLRP3 inflammasome in the glaucomatous optic nerve head. Investig. Ophthalmol. Vis. Sci. 2017;58:2021. [Google Scholar]
- 80.Li L., Xing C., Zhou J., Niu L., Luo B., Song M., Niu J., Ruan Y., Sun X., Lei Y. Airborne particulate matter (PM2.5) triggers ocular hypertension and glaucoma through pyroptosis. Part Fibre Toxicol. 2021;18:1–13. doi: 10.1186/s12989-021-00403-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Niu L., Li L., Xing C., Luo B., Hu C., Song M., Niu J., Ruan Y., Sun X., Lei Y. Airborne particulate matter (PM2.5) triggers cornea inflammation and pyroptosis via NLRP3 activation. Ecotoxicol. Environ. Saf. 2021;207:111306. doi: 10.1016/j.ecoenv.2020.111306. [DOI] [PubMed] [Google Scholar]
- 82.Park B., Jo K., Lee T.G., Hyun S.W., Kim J.S., Kim C.S. Polydatin Inhibits NLRP3 Inflammasome in Dry Eye Disease by Attenuating Oxidative Stress and Inhibiting the NF-κB Pathway. Nutrients. 2019;11:2792. doi: 10.3390/nu11112792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.McGilligan V.E., Gregory-Ksander M.S., Li D., Moore J.E., Hodges R.R., Gilmore M.S., Moore T., Dartt D.A. Staphylococcus aureus Activates the NLRP3 Inflammasome in Human and Rat Conjunctival Goblet Cells. PLoS ONE. 2013;8:e74010. doi: 10.1371/journal.pone.0074010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Tseng W.A., Thein T., Kinnunen K., Lashkari K., Gregory M.S., D’Amore P.A., Ksander B.R. NLRP3 Inflammasome Activation in Retinal Pigment Epithelial Cells by Lysosomal Destabilization: Implications for Age-Related Macular Degeneration. Investig. Opthalmol. Vis. Sci. 2013;54:110–120. doi: 10.1167/iovs.12-10655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Freeman L.C., Ting J.P.-Y. The pathogenic role of the inflammasome in neurodegenerative diseases. J. Neurochem. 2016;136:29–38. doi: 10.1111/jnc.13217. [DOI] [PubMed] [Google Scholar]
- 86.Chi W., Li F., Chen H., Wang Y., Zhu Y., Yang X., Zhu J., Wu F., Ouyang H., Ge J., et al. Caspase-8 promotes NLRP1/NLRP3 inflammasome activation and IL-1 production in acute glaucoma. Proc. Natl. Acad. Sci. USA. 2014;111:11181–11186. doi: 10.1073/pnas.1402819111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Fei F., Alexander J., Bruce R.K., Ann M.R., Meredith S.G. Gene therapy treatment with AAV-soluble fas ligand protects retinal ganglion cells during development of glaucoma. Investig. Ophthalmol. Vis. Sci. 2015;56:2594. [Google Scholar]
- 88.Wang R., Seifert P., Jakobs T.C. Astrocytes in the Optic Nerve Head of Glaucomatous Mice Display a Characteristic Reactive Phenotype. Investig. Opthalmology Vis. Sci. 2017;58:924–932. doi: 10.1167/iovs.16-20571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Taurone S., Plateroti P., Ripandelli G., Pacella E., Bianchi E., Plateroti A.M., De Vito S., Grippaudo F.R., Cavallotti C., Artico M. Potential regulatory molecules in the human trabecular meshwork of patients with glaucoma: Immunohistochemical profile of a number of inflammatory cytokines. Mol. Med. Rep. 2014;11:1384–1390. doi: 10.3892/mmr.2014.2772. [DOI] [PubMed] [Google Scholar]
- 90.Izzotti A., Longobardi M., Cartiglia C., Saccà S.C. Mitochondrial Damage in the Trabecular Meshwork Occurs Only in Primary Open-Angle Glaucoma and in Pseudoexfoliative Glaucoma. PLoS ONE. 2011;6:e14567. doi: 10.1371/journal.pone.0014567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Jia X., Yu J., Liao S.-H., Duan X.-C. Biomechanics of the sclera and effects on intraocular pressure. Int. J. Ophthalmol. 2016;9:1824–1831. doi: 10.18240/ijo.2016.12.21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Xie Y., Ouyang X., Wang G. Mechanical strain affects collagen metabolism-related gene expression in scleral fibroblasts. Biomed. Pharmacother. 2020;126:110095. doi: 10.1016/j.biopha.2020.110095. [DOI] [PubMed] [Google Scholar]
- 93.Bageghni S.A., Hemmings K.E., Yuldasheva N.Y., Maqbool A., Gamboa-Esteves F.O., Humphreys N.E., Jackson M.S., Denton C.P., Francis S., Porter K.E., et al. Fibroblast-specific deletion of interleukin-1 receptor-1 reduces adverse cardiac remodeling following myocardial infarction. JCI Insight. 2019;5:125074. doi: 10.1172/jci.insight.125074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Ershaid N., Sharon Y., Doron H., Raz Y., Shani O., Cohen N., Monteran L., Leider-Trejo L., Ben-Shmuel A., Yassin M., et al. NLRP3 inflammasome in fibroblasts links tissue damage with inflammation in breast cancer progression and metastasis. Nat. Commun. 2019;10:1–15. doi: 10.1038/s41467-019-12370-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ranzer M.J., Chen L., DiPietro L.A. Fibroblast Function and Wound Breaking Strength Is Impaired by Acute Ethanol Intoxication. Alcohol. Clin. Exp. Res. 2010;35:83–90. doi: 10.1111/j.1530-0277.2010.01324.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Gregory M.S. A New Method to Inhibit Inflammation and Prevent Glaucoma. [(accessed on 6 July 2021)]; Available online: https://www.brightfocus.org/glaucoma/grant/nlrp3-inflammasome-new-target-glaucoma.
- 97.Pérez de Lara M.J., Avilés-Trigueros M., Guzmán-Aránguez A., Valiente-Soriano F.J., de la Villa P., Vidal-Sanz M., Pintor J. Potential role of P2X7 receptor in neurodegenerative processes in a murine model of glaucoma. Brain Res. Bull. 2019;150:61–74. doi: 10.1016/j.brainresbull.2019.05.006. [DOI] [PubMed] [Google Scholar]
- 98.Zhang X., Li A., Ge J., Reigada D., Laties A.M., Mitchell C.H. Acute increase of intraocular pressure releases ATP into the anterior chamber. Exp. Eye Res. 2007;85:637–643. doi: 10.1016/j.exer.2007.07.016. [DOI] [PubMed] [Google Scholar]
- 99.Pérez de Lara M.J., Guzmán-Aránguez A., de la Villa P., Díaz-Hernández J.I., Miras-Portugal M.T., Pintor J. Increased levels of extracellular ATP in glaucomatous retinas: Possible role of the vesicular nucleotide transporter during the development of the pathology. Mol. Vis. 2015;21:1060–1070. [PMC free article] [PubMed] [Google Scholar]
- 100.Dosch M., Gerber J., Jebbawi F., Beldi G. Mechanisms of ATP Release by Inflammatory Cells. Int. J. Mol. Sci. 2018;19:1222. doi: 10.3390/ijms19041222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Burnstock G. Release of vasoactive substances from endothelial cells by shear stress and purinergic mechanosensory transduction. J. Anat. 1999;194:335–342. doi: 10.1046/j.1469-7580.1999.19430335.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Li A., Leung C.T., Peterson-Yantorno K., Stamer W.D., Mitchell C.H., Civan M.M. Mechanisms of ATP release by human trabecular meshwork cells, the enabling step in purinergic regulation of aqueous humor outflow. J. Cell. Physiol. 2012;227:172–182. doi: 10.1002/jcp.22715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Tezel G. Oxidative stress in glaucomatous neurodegeneration: Mechanisms and consequences. Prog. Retin. Eye Res. 2006;25:490–513. doi: 10.1016/j.preteyeres.2006.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Liu Y., Dai Y., Li Q., Chen C., Chen H., Song Y., Hua F., Zhang Z. Beta-amyloid activates NLRP3 inflammasome via TLR4 in mouse microglia. Neurosci. Lett. 2020;736:135279. doi: 10.1016/j.neulet.2020.135279. [DOI] [PubMed] [Google Scholar]
- 105.Guo L., Salt T.E., Luong V., Wood N., Cheung W., Maass A., Ferrari G., Russo-Marie F., Sillito A.M., Cheetham M., et al. Targeting amyloid-beta in glaucoma treatment. Proc. Natl. Acad. Sci. USA. 2007;104:13444–13449. doi: 10.1073/pnas.0703707104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Wang J., Harris A., Prendes M.A., Alshawa L., Gross J.C., Wentz S.M., Rao A.B., Kim N.J., Synder A., Siesky B. Targeting Transforming Growth Factor-β Signaling in Primary Open-Angle Glaucoma. J. Glaucoma. 2017;26:390–395. doi: 10.1097/IJG.0000000000000627. [DOI] [PubMed] [Google Scholar]
- 107.Zhang K., Fan C., Cai D., Zhang Y., Zuo R., Zhu L., Cao Y., Zhang J., Liu C., Chen Y., et al. Contribution of TGF-Beta-Mediated NLRP3-HMGB1 Activation to Tubulointerstitial Fibrosis in Rat With Angiotensin II-Induced Chronic Kidney Disease. Front. Cell Dev. Biol. 2020;8:1. doi: 10.3389/fcell.2020.00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Krasnow S.M., Knoll J.G., Verghese S.C., Levasseur P.R., Marks D.L. Amplification and propagation of interleukin-1β signaling by murine brain endothelial and glial cells. J. Neuroinflamm. 2017;14:1–18. doi: 10.1186/s12974-017-0908-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Wilson G.N., Inman D.M., Dengler Crish C.M., Smith M.A., Crish S.D. Early pro-inflammatory cytokine elevations in the DBA/2J mouse model of glaucoma. J. Neuroinflamm. 2015;12:015–0399. doi: 10.1186/s12974-015-0399-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Mallick J., Devi L., Malik P.K., Mallick J. Update on normal tension glaucoma. J. Ophthalmic Vis. Res. 2016;11:204–208. doi: 10.4103/2008-322X.183914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Cvenkel B., Kolko M. Current Medical Therapy and Future Trends in the Management of Glaucoma Treatment. J. Ophthalmol. 2020;2020:1–14. doi: 10.1155/2020/6138132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Guan L., Li C., Zhang Y., Gong J., Wang G., Tian P., Shen N. Puerarin ameliorates retinal ganglion cell damage induced by retinal ischemia/reperfusion through inhibiting the activation of TLR4/NLRP3 inflammasome. Life Sci. 2020;256:117935. doi: 10.1016/j.lfs.2020.117935. [DOI] [PubMed] [Google Scholar]
- 113.Kwon S., Kim S.H., Khang D., Lee J.Y. Potential Therapeutic Usage of Nanomedicine for Glaucoma Treatment. Int. J. Nanomed. 2020;15:5745–5765. doi: 10.2147/IJN.S254792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Gong Y., Cao X., Gong L., Li W. Sulforaphane alleviates retinal ganglion cell death and inflammation by suppressing NLRP3 inflammasome activation in a rat model of retinal ischemia/reperfusion injury. Int. J. Immunopathol. Pharmacol. 2019;33:2058738419861777. doi: 10.1177/2058738419861777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Chen H., Deng Y., Gan X., Li Y., Huang W., Lu L., Wei L., Su L., Luo J., Zou B., et al. NLRP12 collaborates with NLRP3 and NLRC4 to promote pyroptosis inducing ganglion cell death of acute glaucoma. Mol. Neurodegener. 2020;15:1–16. doi: 10.1186/s13024-020-00372-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Lin C., Wu F., Zheng T., Wang X., Chen Y., Wu X. Kaempferol attenuates retinal ganglion cell death by suppressing NLRP1/NLRP3 inflammasomes and caspase-8 via JNK and NF-κB pathways in acute glaucoma. Eye. 2019;33:777–784. doi: 10.1038/s41433-018-0318-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Yerramothu P., Vijay A.K., Willcox M. Inflammasomes, the eye and anti-inflammasome therapy. Eye. 2017;32:491–505. doi: 10.1038/eye.2017.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Coll R., Robertson A., Chae J.J., Higgins S.C., Muñoz-Planillo R., Inserra M.C., Vetter I., Dungan L.S., Monks B.G., Stutz A., et al. A small-molecule inhibitor of the NLRP3 inflammasome for the treatment of inflammatory diseases. Nat. Med. 2015;21:248–255. doi: 10.1038/nm.3806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Krishnan A., Dua M., Mitchell C.H., Lu W., Gregory-Ksander S.M. Pharmacologic inhibition of the NLRP3 inflammasome-a novel neuroprotective therapy for glaucoma. Investig. Ophthalmol. Vis. Sci. 2019;60:2256. [Google Scholar]
- 120.Ward R., Li W., Abdul Y., Jackson-Cowan L., Dong G., Jamil S., Filosa J., Fagan S.C., Ergul A. NLRP3 inflammasome inhibition with MCC950 improves diabetes-mediated cognitive impairment and vasoneuronal remodeling after ischemia. Pharmacol. Res. 2019;142:237–250. doi: 10.1016/j.phrs.2019.01.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.La Manna S., Di Natale C., Florio D., Marasco D. Peptides as Therapeutic Agents for Inflammatory-Related Diseases. Int. J. Mol. Sci. 2018;19:2714. doi: 10.3390/ijms19092714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Mouyis M., Isenberg D. Overview of biologic therapies in autoimmune rheumatic diseases. Medicine. 2014;42:184–187. doi: 10.1016/j.mpmed.2013.12.008. [DOI] [Google Scholar]
- 123.Gaston J., Maestrali N., Lalle G., Gagnaire M., Masiero A., Dumas B., Dabdoubi T., Radošević K., Berne P.-F. Intracellular delivery of therapeutic antibodies into specific cells using antibody-peptide fusions. Sci. Rep. 2019;9:1–12. doi: 10.1038/s41598-019-55091-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Netea M.G., Simon A., van de Veerdonk F., Kullberg B.J., Van der Meer J.W., Joosten L.A. IL-1beta processing in host defense: Beyond the inflammasomes. PLoS Pathog. 2010;6:1000661. doi: 10.1371/journal.ppat.1000661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Dinarello C.A. The Role of the Interleukin-1–Receptor Antagonist in Blocking Inflammation Mediated by interleukin-1. N. Engl. J. Med. 2000;343:732–734. doi: 10.1056/NEJM200009073431011. [DOI] [PubMed] [Google Scholar]
- 126.Tapia-Abellán A., Angosto D., Banaclocha H.M., de Torre C., Cerón-Carrasco J.P., Pérez-Sánchez H., Arostegui J.I., Pelegrin P. MCC950 closes the active conformation of NLRP3 to an inactive state. Nat. Chem. Biol. 2019;15:560–564. doi: 10.1038/s41589-019-0278-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Jiang H., He H., Chen Y., Huang W., Cheng J., Ye J., Wang A., Tao J., Wang C., Liu Q., et al. Identification of a selective and direct NLRP3 inhibitor to treat inflammatory disorders. J. Exp. Med. 2017;214:3219–3238. doi: 10.1084/jem.20171419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Huang Y., Jiang H., Chen Y., Wang X., Yang Y., Tao J., Deng X., Liang G., Zhang H., Jiang W., et al. Tranilast directly targets NLRP 3 to treat inflammasome-driven diseases. EMBO Mol. Med. 2018;10:e8689. doi: 10.15252/emmm.201708689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Abderrazak A., Couchie D., Mahmood D.F.D., Elhage R., Vindis C., Laffargue M., Matéo V., Büchele B., Ayala M.R., El Gaafary M., et al. Anti-Inflammatory and Antiatherogenic Effects of the NLRP3 Inflammasome Inhibitor Arglabin in ApoE 2.Ki Mice Fed a High-Fat Diet. Circulation. 2015;131:1061–1070. doi: 10.1161/CIRCULATIONAHA.114.013730. [DOI] [PubMed] [Google Scholar]
- 130.Desu H.L., Plastini M., Illiano P., Bramlett H.M., Dietrich W.D., Vaccari J.P.D.R., Brambilla R., Keane R.W. IC100: A novel anti-ASC monoclonal antibody improves functional outcomes in an animal model of multiple sclerosis. J. Neuroinflamm. 2020;17:143. doi: 10.1186/s12974-020-01826-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.