Abstract
Background
Posterior capsule opacification (PCO) is a clouding of the posterior part of the lens capsule, a skin‐like transparent structure, which surrounds the crystalline lens in the human eye. PCO is the most common postoperative complication following modern cataract surgery with implantation of a posterior chamber intraocular lens (IOL). The main symptoms of PCO are a decrease in visual acuity, 'cloudy', blurred vision and reduced contrast sensitivity. PCO is treated with a neodymium:YAG (Nd:YAG) laser to create a small opening in the opaque capsule and regain a clear central visual axis. This capsulotomy might cause further ocular complications, such as raised intraocular pressure or swelling of the central retina (macular oedema). This procedure is also a significant financial burden for health care systems worldwide. In recent decades, there have been advances in the selection of IOL materials and optimisation of IOL designs to help prevent PCO formation after cataract surgery. These include changes to the side structures holding the lens in the centre of the lens capsule bag, called IOL haptics, and IOL optic edge designs.
Objectives
To compare the effects of different IOL optic edge designs on PCO after cataract surgery.
Search methods
We searched CENTRAL, Ovid MEDLINE, Ovid Embase, Latin American and Caribbean Health Sciences Literature Database (LILACS), the ISRCTN registry, ClinicalTrials.gov and the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) up to 17 November 2020.
Selection criteria
We included randomised controlled trials (RCTs) that compared different types of IOL optic edge design. Our prespecified primary outcome was the proportion of eyes with Nd:YAG capsulotomy one year after surgery. Secondary outcomes included PCO score, best‐corrected distance visual acuity (BCDVA) and quality of life score at one year. Due to availability of important long‐term data, we also presented data at longer‐term follow‐up which is a post hoc change to our protocol.
Data collection and analysis
We used standard methods expected by Cochrane and the GRADE approach to assess the certainty of the evidence.
Main results
We included 10 studies (1065 people, 1834 eyes) that compared sharp‐ and round‐edged IOLs. Eight of these studies were within‐person studies whereby one eye received a sharp‐edged IOL and the fellow eye a round‐edged IOL. The IOL materials were acrylic (2 studies), silicone (4 studies), polymethyl methacrylate (PMMA, 3 studies) and different materials (1 study). The studies were conducted in Austria, Germany, India, Japan, Sweden and the UK. Five studies were at high risk of bias in at least one domain. We judged two studies to be at low risk of bias in all domains.
There were few cases of Nd:YAG capsulotomy at one year (primary outcome): 1/371 in sharp‐edged and 4/371 in round‐edged groups. The effect estimate was in favour of sharp‐edged IOLs but the confidence intervals were very wide and compatible with higher or lower chance of Nd:YAG capsulotomy in sharp‐edged compared with round‐edged lenses (Peto odds ratio (OR) 0.30, 95% CI 0.05 to 1.74; I2 = 0%; 6 studies, 742 eyes). This corresponds to seven fewer cases of Nd:YAG capsulotomy per 1000 sharp‐edged IOLs inserted compared with round‐edged IOLs (95% CI 9 fewer to 7 more). We judged this as low‐certainty evidence, downgrading for imprecision and risk of bias.
A similar reduced risk of Nd:YAG capsulotomy in sharp‐edge compared with round‐edge IOLs was seen at two, three and five years but as the number of Nd:YAG capsulotomy events increased with longer follow‐up this effect was more precisely measured at longer follow‐up: two years, risk ratio (RR) 0.35 (0.16 to 0.80); 703 eyes (6 studies); 89 fewer cases per 1000; three years, RR 0.21 (0.11 to 0.41); 538 eyes (6 studies); 170 fewer cases per 1000; five years, RR 0.21 (0.10 to 0.45); 306 eyes (4 studies); 331 fewer cases per 1000. Data at 9 years and 12 years were only available from one study.
All studies reported a PCO score. Four studies reported the AQUA (Automated Quantification of After‐Cataract) score, four studies reported the EPCO (Evaluation of PCO) score and two studies reported another method of quantifying PCO. It was not possible to pool these data due to the way they were reported, but all studies consistently reported a statistically significant lower average PCO score (of the order of 0.5 to 3 units) with sharp‐edged IOLs compared with round‐edged IOLs. We judged this to be moderate‐certainty evidence downgrading for risk of bias.
The logMAR visual acuity score was lower (better) in eyes that received a sharp‐edged IOL but the difference was small and likely to be clinically unimportant at one year (mean difference (MD) ‐0.06 logMAR, 95% CI ‐0.12 to 0; 2 studies, 153 eyes; low‐certainty evidence). Similar effects were seen at longer follow‐up periods but non‐statistically significant data were less fully reported: two years MD ‐0.01 logMAR (‐0.05 to 0.02); 2 studies, 311 eyes; three years MD ‐0.09 logMAR (‐0.22 to 0.03); 2 studies, 117 eyes; data at five years only available from one study.
None of the studies reported quality of life. Very low‐certainty evidence on adverse events did not suggest any important differences between the groups.
Authors' conclusions
This review provides evidence that sharp‐edged IOLs are likely to be associated with less PCO formation than round‐edged IOLs, with less Nd:YAG capsulotomy. The effects on visual acuity were less certain. The impact of these lenses on quality of life has not been assessed and there are only very low‐certainty comparative data on adverse events.
Plain language summary
Does the design of replacement lenses used in cataract surgery help to stop clouding of the new lens once inside the eye?
Key messages In cataract surgery, replacement lenses that have a sharp edge design may be less likely to develop clouding on their surface than lenses with a round edge. People given new lenses with sharp edges may have clearer eyesight, and may be less likely to need laser surgery, than people given lenses with rounded edges.
How are cataracts treated? The lens is a small, clear disc inside the eye that focuses light rays to make clear images of objects seen. A cataract starts when cloudy patches develop on the lens. As the cloudy patches get bigger over time, sight becomes misty and blurred.
Surgery is the only way to improve your eyesight if you have cataracts. In cataract surgery, a tiny cut is made in your eye; the old, cloudy lens is removed and a new, plastic lens is put in its place.
After cataract surgery A common problem after cataract surgery is that the back of the new lens develops cloudy patches on the surface, causing blurred or misty sight again. The problem is treated by using a laser to create a small opening in the back of the lens, to allow light through. However, this procedure could cause further problems, such as raising pressure inside the eye, or could damage other parts of the eye.
Why we did this Cochrane Review Different styles of plastic lenses have been designed to try to stop or reduce clouding once they are in the eye. Changes include making the edges of the lens round or sharp. We wanted to find out if differently shaped edges on the lens would make it less likely to cloud.
What did we do? We searched for studies that tested different designs of plastic lenses used in cataract surgery. We wanted to know how differently shaped edges on the lens affected the need for laser eye treatment one year after cataract surgery.
Search date: we included evidence published up to 17 November 2020.
What we found We found 10 studies involving 1065 people with age‐related cataracts who had surgery to replace the lens in 1834 eyes. The studies took place in Austria, Germany, India, Japan, Sweden and the UK. The studies lasted for at least one year. Some studies followed participants for up to 12 years after their surgery.
The studies compared replacement lenses with rounded edges against replacement lenses with sharp edges.
A pharmaceutical company funded one study and partly funded another study.
What are the main results of our review? In all 10 studies, eyes given a sharp‐edged lens showed less clouding on the lens, compared with eyes given a round‐edged lens.
‐ After two, three and five years, eyes given a sharp‐edged lens may be less likely to need laser surgery than those given a round‐edged lens (evidence from six studies involving 742 people). ‐ After one year and three years, vision may be clearer in eyes given sharp‐edged lenses compared with eyes given round‐edged lenses (evidence from two studies involving 260 people). ‐ At one year after surgery, it was not clear if the design of lens (sharp‐ or round‐edged) affected how many people needed to have laser surgery to treat cloudy patches on the new lens. This might be because not many people in either group needed laser surgery after one year.
We are uncertain how the two designs of lens affected the numbers of any unwanted effects people experienced, because these were not consistently reported. We found no evidence of any important differences between the two lens types in the numbers of complications reported.
None of the studies measured people's well‐being.
Our confidence in our results We are moderately confident about lenses with sharp edges showing a lower average score for clouding than lenses with round edges. Further evidence may increase our confidence in this result. We are less confident about vision being clearer and the lower need for laser surgery for eyes given sharp‐edged lenses. Further research is likely to increase our confidence in these results.
Summary of findings
Summary of findings 1. Sharp‐edged intraocular lens (IOL) compared to round‐edged IOL for the prevention of posterior capsule opacification after cataract surgery.
| Sharp‐edged compared to round‐edged IOLS for the prevention of posterior capsule opacification after cataract surgery | ||||||
| Patient or population: people undergoing cataract surgery Setting: eye hospital Intervention: sharp‐edged IOL Comparison: round‐edged IOL | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with round‐edged IOL | Risk with sharp‐edged IOL | |||||
| Nd:YAG capsulotomy at one year | Study population | OR 0.30 (0.05 to 1.74) | 742 (6 RCTs) | ⊕⊕⊝⊝ LOWa,b | At 2 years: RR 0.35 (0.16 to 0.80); 703 eyes (6 studies); 89 fewer cases per 1000. At 3 years: RR 0.21 (0.11 to 0.41); 538 eyes (6 studies); 170 fewer cases per 1000. At 5 years: RR 0.21 (0.10 to 0.45); 306 eyes (4 studies); 331 fewer cases per 1000. Data at 9 years and 12 years were only available from one study. |
|
| 10 per 1000 | 3 per 1000 (1 to 17) | |||||
| Posterior capsule opacification score at one year As measured by AQUA score, Evaluation of PCO (EPCO) score and other methods of quantifying PCO |
All studies consistently reported a lower average PCO score (of the order of 0.5 to 3 units) with sharp‐edged IOLs compared with round‐edged IOLs | (10 RCTs) | ⊕⊕⊕⊝ MODERATEa | It was not possible to pool these data due to the way they were reported | ||
| BCDVA at one year, measured using logMAR score (range ‐0.3 to 1.3, low scores = better vision, a score of 0 is equivalent to 6/6 or 20/20 vision) | The mean logMAR BCDVA at one year ranged from 0 to 0.13 | MD 0.06 logMAR units lower (better) (0.12 lower to 0 lower) | ‐ | 153 (2 RCTs) | ⊕⊕⊝⊝ LOWa,b | At 2 years: MD ‐0.01 logMAR (‐0.05 to 0.02; 2 studies, 311 eyes. At 3 years MD ‐0.09 logMAR (‐0.22 to 0.03; 2 studies, 117 eyes. Data at 5 years were only available from one study. |
| Quality of life | None of the studies reported this outcome | |||||
| Adverse effects | Adverse effects were reported inconsistently. There was no evidence of any important differences in Intraocular complications, entopic phenomena, IOL tilt/decentration or anterior capsule fibrosis between groups. | ⊕⊝⊝⊝ VERY LOWa,c | ‐ | |||
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). BCDVA: best‐corrected distance visual acuity; CI: confidence interval; OR: odds ratio; PCO: posterior capsule opacification; RR: risk ratio | ||||||
| GRADE Working Group grades of evidence High‐certainty: We are very confident that the true effect lies close to that of the estimate of the effect. Moderate‐certainty: We are moderately confident in the effect estimate: the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low‐certainty: Our confidence in the effect estimate is limited: the true effect may be substantially different from the estimate of the effect. Very low‐certainty: We have very little confidence in the effect estimate: the true effect is likely to be substantially different from the estimate of effect. | ||||||
aDowngraded for risk of bias (‐1): only 2 studies at low risk of bias in all domains bDowngraded for imprecision (‐1): confidence intervals include benefit and harm cDowngraded for imprecision (‐2): studies were small and underpowered to assess rare outcomes
Background
Extracapsular cataract extraction (ECCE) with posterior chamber intraocular lens (IOL) implantation is the preferred surgical technique for the treatment of opacification of the crystalline lens (cataract) in high‐income countries, and most surgeons in low‐ and middle‐income countries are now being trained in this method (Ang 2014; Riaz 2013). In this technique, the lens contents are removed, leaving the posterior lens capsule intact. The lens capsule forms an anatomical barrier between the anterior and posterior segments of the eye. This is thought to reduce the risk of posterior segment complications following surgery (such as swelling of the central area of the retina (cystoid macular oedema) and retinal detachment) compared to intracapsular cataract extraction (ICCE), in which the whole lens with intact capsule is removed from the eye. Whereas phacoemulsification with implantation of a posterior chamber IOL is standard for cataract treatment in high‐income countries, this technique represents only 10% of all cataract surgeries in low‐ and middle‐income countries (LMICs) (Khanna 2011). The main reason for such a low percentage may be the high costs of phacoemulsification equipment (Findl 2010; Riaz 2006). However, leaving the capsule intact may result in long‐term complications, mainly that of posterior capsule opacification (PCO) (Apple 1992; Schaumberg 1998). People with PCO experience decreased visual acuity, impaired contrast sensitivity and glare.
Description of the condition
PCO is the most common postoperative complication after cataract surgery, and PCO proportions of 11% to 43% have been described within the first year after surgery, depending on surgical technique, IOL design and material, and concomitant ocular diseases (Findl 2010; Riaz 2013; Schaumberg 1998; Wormstone 2002). Clinically, PCO (or after‐cataract) is subdivided into two types: regeneratory and fibrotic PCO. Regeneratory PCO is much more common and is the main cause of a decrease in visual function after cataract surgery. Regeneratory PCO is a result of migration of lens epithelial cells along the posterior capsule, behind the IOL. These cells proliferate to form layers of lens material and Elschnig pearls, leading to opacification of the posterior capsule. In fibrotic PCO, lens epithelial cells of the anterior capsule undergo transformation to myofibroblasts, causing fibrosis and contraction of the capsule bag. This can lead to decentration of the IOL and hinder visualisation of the peripheral retina. If present on the posterior capsule, this may cause vision to deteriorate. Diagnosis is made by slit lamp examination by an ophthalmologist. PCO is usually treated by cutting an opening into the posterior capsule with a neodymium:YAG (Nd:YAG) laser, or rarely by surgical opening (capsulotomy). However, laser treatment may damage the IOL optic, transiently increase intraocular pressure (IOP), induce cystoid macular oedema, increase the incidence of retinal detachment and often does not improve visualisation of the peripheral retina (Javitt 1992). Additionally, Nd:YAG laser treatment is associated with high costs for the healthcare system. In the USA, Nd:YAG capsulotomy is the second most commonly performed ophthalmic surgical procedure after cataract surgery. The annual cost of Nd:YAG capsulotomy was estimated to be USD 250 million in the USA alone (Apple 1992). The lack of availability of laser treatment is one of the major reasons why ECCE with IOL implantation is not extensively used in low‐ and middle‐income countries.
Description of the intervention
To prevent PCO in eyes after cataract surgery with implantation of an IOL, industry and clinicians make efforts to prevent PCO by modifying IOL loops (or haptics) and optic design. The IOL optic edge may be round or sharp. A sharp edge means that the posterior surface and the lateral edge of the IOL meet at a right angle. The firm contact with the posterior capsule created by the sharp optic edge seems to be important in the inhibition of cell migration (Kohnen 2008; Nishi 2007). The sharpness of the optic edge may vary according to the type of material used to manufacture the lens (Nanavety 2008; Nanavety 2019).
How the intervention might work
IOL optic edges with a square edge reduce the migration of remnant lens epithelial cells from the equatorial zone of the capsular bag. Lens epithelial cells proliferating from the periphery of the capsule stop when they reach the sharply bent edge of the capsule in contact with the sharp edge of the IOL. As a result, this optic edge design may reduce progression of PCO towards the centre of the capsule and, therefore, towards the visual axis. Various manufacturers are offering IOLs with sharp optic edges in order to reduce postoperative PCO proportion (Nishi 2007).
The introduction of sharp optic edges appears to reduce the incidence of PCO (Findl 2010). It is unclear whether differences in the style of IOL haptics play a role in PCO inhibition.
Beside this inhibiting effect on PCO formation, the square edge of IOL optics has been shown to be a potential source for the occurrence of entopic phenomena such as positive and negative dysphotopsia. Several ray‐tracing models and in vivo studies have investigated sharp IOL optic edges as cause for these optic side effects in pseudophakic eyes (Holladay 2012; Masket 1993; Masket 2020; Schwiegerling 2016). Sharp and truncated edges may induce dysphotopsia, especially in combination with material of a high index of refraction (Radmall 2015). Therefore, some manufacturers have enhanced their IOL designs ‐ for example, by rounding the anterior optic edge.
Why it is important to do this review
Three Cochrane Reviews evaluating different surgical interventions for age‐related cataract have already been published (Ang 2014; de Silva 2014; Riaz 2013). There is an extensive body of published data on interventions to reduce the incidence of PCO. Several factors seem to influence the interaction of the IOL with the capsule bag and therefore play an important role in the prevention of PCO progression (Findl 2010). These include surgical technique (Hollick 1999; Khalifa 1992), IOL material and biocompatibility (Auffarth 2004; Findl 2010), and different IOL optic edge designs (Findl 2010; Hollick 1999; Nishi 1998; Nishi 2004). Additionally, Walker 2008 has investigated intraocular pharmaceutical agents for their influence on lens epithelial cell growth and PCO formation. In order to draw conclusions regarding the different IOL optic edge designs' influence on preventing PCO, we have undertaken a systematic review of randomised controlled trials (RCTs). This will be important to inform cataract surgeons and patients as to the best choice of IOL during cataract surgery.
Objectives
To compare the effects of different IOL optic edge designs on PCO after cataract surgery.
Methods
Criteria for considering studies for this review
Types of studies
This review included only randomised controlled trials (RCTs) with a follow‐up period of at least 12 months.
Types of participants
Participants in the trials were people with age‐related cataract who underwent cataract surgery (all extracapsular surgical techniques, including femtosecond laser‐assisted cataract surgery) with implantation of an IOL into the capsule bag. We did not exclude people with ocular comorbidities such as age‐related macular degeneration (AMD), diabetes, glaucoma or uveitis, and other sight‐threatening diseases, because low PCO rates and therefore better postoperative visual outcomes might also be an advantage for these individuals.
Types of interventions
We compared sharp (or square) IOL optic edges to round‐edge IOLs, including IOLs with special optical properties, like toric, multifocal and accommodating IOLs.
Types of outcome measures
We considered the following outcome measures.
Primary outcomes
Proportion of eyes receiving Nd:YAG laser capsulotomy at 12 months postoperatively. Because PCO formation is very uncommon in the first months after surgery, a minimum of 12 months follow‐up time was defined.
Nd:YAG capsulotomy may depend on a clinician's subjective evaluation, whereas PCO scores are objective measurement techniques. However, PCO scores do not indicate whether PCO is located on the central or peripheral posterior capsule and they may not reflect individuals' subjective visual impairment (Li 2013). Therefore, we selected the capsulotomy proportion as the primary outcome.
Secondary outcomes
Mean intensity of PCO, as assessed with scoring techniques or quantification by image analysis at 12 months and 24 months postoperatively.
Mean best‐corrected distance visual acuity (BCDVA) (logMAR). The minimum length of follow‐up to be included for meta‐analysis was 12 months. Data from earlier follow‐up were not included.
Mean quality of life score as measured by any validated questionnaire.
Assessment of PCO intensity or severity is complicated by the fact that there is no commonly accepted scoring method for PCO (Findl 2003). There are several well‐known subjective scoring systems, such as the EPCO (Evaluation of PCO) computer‐aided scoring system, as well as 'semi‐objective' or objective systems, such as POCO (posterior capsule opacification) software and AQUA (Automated Quantification of After‐Cataract) systems, both based on automated analysis of retro‐illumination PCO images. In addition, there is a frequently used objective PCO assessment method based on analysis of Scheimpflug images.
Analysis both of PCO scores and visual acuity data may be additionally complicated by the fact that some people may have an Nd:YAG capsulotomy. This problem is sometimes handled by estimating the missing PCO scores after a capsulotomy (Buehl 2005; Findl 2005). We documented how the problem of Nd:YAG was handled in each study.
Adverse effects
Intraoperative surgical complications (e.g. capsule rupture)
Entopic phenomena (e.g. negative dysphotopsia)
Clinically relevant IOL tilt and decentration
Distinctive anterior capsule fibrosis and capsule phimosis
Any other adverse effects as reported by studies
We collected data at 12, 24, 36 months and longer time periods where available.
Search methods for identification of studies
Electronic searches
The Cochrane Eyes and Vision Information Specialist searched the following electronic databases for randomised controlled trials and controlled clinical trials. There were no restrictions on language or year of publication. The electronic databases were last searched on 17 November 2020.
Cochrane Central Register of Controlled Trials (CENTRAL; 2019; Issue 11) (which contains the Cochrane Eyes and Vision Trials Register) in the Cochrane Library (searched 17 November 2020) (Appendix 1).
MEDLINE Ovid (1946 to 17 November 2020) (Appendix 2).
Embase Ovid (1980 to 17 November 2020) (Appendix 3).
Latin American and Caribbean Health Sciences Literature Database (LILACS) (1982 to 17 November 2020) (Appendix 4).
ISRCTN registry (www.isrctn.com/editAdvancedSearch; searched 17 November 2020) (Appendix 5).
US National Institutes of Health Ongoing Trials Register ClinicalTrials.gov (www.clinicaltrials.gov; searched 17 November 2020) (Appendix 6).
World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) (www.who.int/ictrp; searched 17 November 2020) (Appendix 7).
Searching other resources
We manually searched reference lists of relevant trial reports. We did not manually search any conference abstracts for this review.
Data collection and analysis
Selection of studies
Two review authors (SM, AH) reviewed the titles and abstracts resulting from the searches. The same authors obtained full‐text copies of possibly and definitely relevant trials and assessed them according to the criteria set out above. There were no restrictions concerning publication date. Both authors worked independently from each other, and resolved discrepancies through discussion.
Data extraction and management
Three review authors (SM, AH, JE) independently extracted data using a pre‐piloted customised data extraction template in Covidence. The review authors compared these and resolved discrepancies through discussion. Data were imported directly from Covidence into Review Manager 5 (Review Manager 2014). When data were missing, we tried to contact the study authors by email and asked them to provide the missing information within a period of two months. One review author (SM) checked data entered in Review Manager 5 for accuracy.
Assessment of risk of bias in included studies
We assessed risk of bias according to the methods set out in Chapter 8 of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2017). We used five components to determine risk of bias: selection bias (sequence generation, allocation concealment), performance bias, detection bias and attrition bias, and selective outcome reporting. We graded each component as low risk of bias, high risk of bias or unclear risk of bias. Masking (blinding) of study participants and personnel may reduce the risk of performance bias, and masking of outcome assessors may reduce risk of detection bias. Therefore, both measures, if applicable, were of special interest in our assessment of risk of bias in the studies included for this review. Two review authors (SM, AH) independently assessed the risk of bias and resolved disagreements through discussion, the use of additional referees, or both. The review authors were not masked to any trial details during the assessment. If we were unable to make a decision about the classification of a study due to lack of information, we tried to contact the study authors.
Measures of treatment effect
We presented dichotomous outcomes, such as the proportion of people needing capsulotomy, as risk ratios, and continuous outcomes, such as BCDVA and PCO scores, as the mean difference. For the analysis of ND:YAG capsulotomy at one year, there were only five events so we used the Peto odds ratio, following guidance in Chapter 10 of the Cochrane Handbook for Systematic Reviews of Interventions (Sterne 2011).
Unit of analysis issues
In our protocol, we anticipated a variety of unit of analysis issues (Differences between protocol and review). For the review, the majority of studies are within‐person studies whereby one eye receives one type of lens and the other eye receives the other. This is a paired design and, ideally, the included studies should have reported a paired analysis. For some outcomes (such as PCO score) in some studies, this was done. However, in general, the paired design was ignored by the included studies. We have used the data as reported. This is a conservative approach ‐ confidence intervals will be wider than if a paired analysis had been done.
Dealing with missing data
Most missing data were due to loss of follow‐up of participants. To assess whether the data were 'missing at random', whenever possible, we tried to contact the original investigators to identify reasons for loss of follow‐up and request incomplete data, if available.
Assessment of heterogeneity
We examined heterogeneity between trial results using a Chi2 test and the I2 statistic. Due to the fact that it has low power in the situation of a meta‐analysis when studies have small sample size or are few in number, a P value of 0.10 was used to determine statistical significance. According to Chapter 9 of the Cochrane Handbook for Systematic Reviews of Interventions, concerning the I2 statistic, values of 30% to 60% may represent moderate heterogeneity, 50% to 90% may represent substantial heterogeneity, and values between 75% and 100% may indicate considerable heterogeneity (Deeks 2017).
Assessment of reporting biases
The main reporting biases that we considered were publication bias and outcome reporting bias. For publication bias, there were not enough trials contributing data to undertake our plan to do a funnel plot to assess whether small trials had different effects. To assess outcome reporting bias we were guided by the ORBIT classification (Kirkham 2010).
Data synthesis
We summarised data from studies collecting comparable outcome measures with similar follow‐up times. We used a random‐effects model, unless there were fewer than three trials in a comparison or the data were sparse, when we used a fixed‐effect model.
Subgroup analysis and investigation of heterogeneity
We planned three subgroup analyses: firstly, to investigate whether effects varied by IOL optic material; secondly, to see whether effects varied by surgical technique; and thirdly, to see the effect of ocular co‐morbidities. There were only data available to assess the first of these planned subgroup analyses.
Sensitivity analysis
We investigated the effect of excluding studies at risk of bias. We repeated the analyses removing studies judged to be at high risk of selection bias, performance bias or detection bias.
Summary of findings and assessment of the certainty of the evidence
We created a 'Summary of findings' table including the following outcomes at one year: Nd:YAG capsulotomy, PCO score, BCDVA, quality of life and adverse effects. Two authors (SM, JE) working together assessed the certainty of the evidence for each outcome using the GRADE approach (GRADEpro GDT).
Results
Description of studies
See Characteristics of included studies and Characteristics of excluded studies.
Results of the search
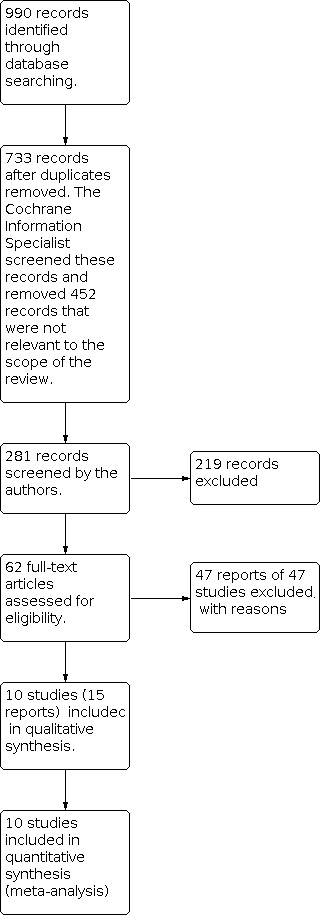
The electronic searches yielded a total of 990 records (Figure 1). After 257 duplicate records were removed, the Cochrane Information Specialist (CIS) screened the remaining 733 records and removed 452 records which were not relevant to the scope of the review. We screened the remaining 281 records and identified 62 references that potentially met the inclusion criteria and obtained full‐text copies of these for further investigation. After assessing the full‐text references, we included 15 reports of 10 studies (Characteristics of included studies), and excluded 47 reports of 47 studies (Characteristics of excluded studies).
1.
Included studies
We included 10 studies (1065 people, 1834 eyes) that compared sharp‐ and round‐edged IOLs (Table 2). The studies were conducted in Austria, Germany, India, Japan, Sweden and the UK.
1. Included studies.
| Study | Country | Number of people | Number of eyes | Type of study | Type of lens material | |
| 1 | Buehl 2005 | Austria | 53 | 106 | Within‐person | Acrylic |
| 2 | Buehl 2007 | Austria | 52 | 104 | Within‐person | Silicone |
| 3 | Findl 2005 | Austria | 32 | 64 | Within‐person | PMMA |
| 4 | Haripriya 2017 | India | 100 | 200 | Within‐person | PMMA |
| 5 | Hayashi 2005 | Japan | 75 | 150 | Within‐person | Acrylic |
| 6 | Mester 2004 | Germany | 288 | 576 | Within‐person | Silicone |
| 7 | Sacu 2005 | Austria | 51 | 102 | Within‐person | Silicone |
| 8 | Shah 2007 | India, UK | 118 | 236 | Within‐person | PMMA |
| 9 | Sundelin 2005 | Sweden | 116 | 116 | Parallel group | Silicone |
| 10 | Wejde 2003 | Sweden | 180 | 180 | Parallel group | Acrylic, PMMA, silicone |
| Total | 1065 | 1834 |
PMMA: polymethyl methacrylate
Types of study
Eight of the studies were within‐person studies, whereby eyes within one person were allocated to sharp‐ or round‐edged lenses. Two studies were parallel group studies whereby people were randomly allocated to treatment and one eye per person included in the study (Sundelin 2005; Wejde 2003).
Types of participants
People with bilateral age‐related cataract. In most of the studies, participants with ocular sight‐threatening diseases (glaucoma, diabetic retinopathy, age‐related macular degeneration or corneal pathologies) or other circumstances that would make a postoperative visual acuity of 20/40 (decimal equivalent 0.5) or better unlikely, were excluded.
Types of interventions
Uncomplicated cataract surgery in both eyes with implantation of an IOL with sharp‐edged optic or round‐edged optic. Different studies used different IOLs. Four studies used silicone IOLs (Buehl 2007; Mester 2004; Sacu 2005; Sundelin 2005), three studies used polymethyl methacrylate (PMMA) (Findl 2005; Haripriya 2017; Shah 2007), two studies used acrylic (Buehl 2005; Hayashi 2005), and one study used all three types (Wejde 2003).
Types of outcome measures
Nd:YAG capsulotomy rates; intensity of PCO (PCO scores ‐ POCO, EPCO, AQUA Score ‐ fraction of PCO); anterior capsule opacification (ACO); visual acuity (BCDVA); contrast sensitivity; glare sensitivity; IOL centration; and capsulorhexis‐IOL overlap.
Excluded studies
Altogether, we excluded 47 studies (please see Characteristics of excluded studies for further information):
19 studies, because they didn't have the correct study design (e.g. were not RCTs, or they were reviews) (Abela‐Formanek 2002; Auffarth 2003; Beltrame 2002; Casprini 2002; Cumming 1994; Hancox 2007; Iashinskas 2005; Kruger 2000; Li 2008; Miyata 2007; Morgan‐Warren 2013; Nishi 2007; Nixon 2004; Oner 2000; Sacu 2004b; Sacu 2005; Spyridaki 2010; Vyas 2007; Zemaitiene 2004);
20 studies, because IOLs of the same optic edge design were being compared (Alio 2016; Alio 2018; Cardona 2018; Gangwani 2011; Gundersen 2017; Hirnschall 2013; Hirnschall 2018; Iwase 2011; Kahraman 2015; Kugelberg 2008; Leydolt 2013; Leydolt 2017; Miháltz 2018; Prosdocimo 2003; Schrecker 2018; Voronin 2017; Yang 2017; Yu 2016; Zemaitiene 2007; Zemaitiene 2011);
three studies, because of the wrong outcomes (e.g. intraocular inflammatory response) (Sacu 2004b; Sacu 2004c; Wejde 2004);
two studies, because no copy of the paper was available after trying to contact the author (Krajcova 2007; Zhang 2002);
two studies because of wrong indication (IOL design comparison: three‐piece versus one‐piece IOL) (Leydolt 2007; Nejima 2006);
one study, because the last follow‐up was only six months postoperatively (Choi 2018).
Risk of bias in included studies
See Figure 2.
2.

Risk of bias summary: review authors' judgements about each risk of bias item for each included study
Allocation
Only two studies used and reported methods of generating and concealing the allocation schedule sufficient for a low risk of bias judgment (Findl 2005; Hayashi 2005). Three studies described a procedure for generating a random allocation but did not describe its concealment (Haripriya 2017; Shah 2007; Sundelin 2005). The remaining studies did not report this aspect in sufficient detail.
Blinding
Two studies reported procedures to mask participants, personnel and outcome assessment (Findl 2005; Hayashi 2005). Three studies were not masked (Mester 2004; Sundelin 2005; Wejde 2003). The remaining studies provided insufficient information.
Incomplete outcome data
In cases of incomplete outcome data reporting, we tried to contact the authors of the studies to gain additional information. We graded all studies with incomplete outcome data as 'unclear risk'.
Selective reporting
We did not identify any obvious cases of selective reporting, but in the absence of access to protocols, it is likely that we could have missed instances of selective reporting. All studies reported Nd:YAG capsulotomy and PCO score. None of the studies reported quality of life but it is likely that this outcome was not measured. Adverse effects were reported inconsistently. Three studies reported visual acuity as a continuous measure and are included in the analyses (Findl 2005; Shah 2007; Sundelin 2005), two studies reported visual acuity as a dichotomous variable (Haripriya 2017; Wejde 2003), two studies reported only on lack of statistical significance (ORBIT classification A, high risk of bias) (Buehl 2007; Mester 2004) and three studies reported on lack of statistical significance and reported the data in a form that could not be included in the review (ORBIT classification C, low risk of bias) (Buehl 2005; Hayashi 2005; Sacu 2005).
Other potential sources of bias
We detected no additional potential sources of bias among the studies included in the review.
Effects of interventions
See: Table 1
See Table 1.
Nd:YAG capsulotomy
Six studies reported Nd:YAG capsulotomy proportions one year postoperatively (Analysis 1.1). There were few events: 1/371 in sharp‐edged and 4/371 in round‐edged IOL groups. The effect estimate was in favour of sharp‐edged IOLs but the confidence intervals were very wide and compatible with higher or lower chance of Nd:YAG capsulotomy in sharp‐edged compared with round‐edged lenses (Peto odds ratio (OR) 0.30, 95% CI 0.05 to 1.74; I2 = 0%; 6 studies, 742 eyes). This corresponds to seven fewer cases of Nd:YAG capsulotomy per 1000 sharp‐edged IOLs inserted compared with round‐edged IOLs (95% CI 9 fewer to 7 more). We judged this as low‐certainty evidence, downgrading for imprecision and risk of bias.
1.1. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 1: Nd:YAG capsulotomy at one year
Similar effects were seen at other time points (Analysis 1.2; Analysis 1.3; Analysis 1.4; Analysis 1.5; Analysis 1.6), and are summarised in the following table. As the number of events in the round‐edged comparator arm increased over time, the number of cases of Nd:YAG capsulotomy potentially prevented increased over time with 89 fewer cases per 1000 at two years, 161 fewer cases per 1000 at three years, and 331 fewer cases per 1000 at five years. Data at 9 years and 12 years were only available from one study.
1.2. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 2: Nd:YAG capsulotomy at two years
1.3. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 3: Nd:YAG capsulotomy at three years
1.4. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 4: Nd:YAG capsulotomy at five years
1.5. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 5: Nd:YAG capsulotomy at 9 years
1.6. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 6: Nd:YAG capsulotomy at 12 years
| Time point | Eyes (studies) | Risk ratio (95% CI) | Fewer cases of Nd:YAG capsulotomy in sharp‐edged IOLs per 1000 (95% CI) |
| 2 years | 703 (6) | 0.35 (0.16 to 0.80) |
89 (27 to 115) |
| 3 years | 538 (6) | 0.21, (0.11 to 0.41) |
170 (127 to 191) |
| 5 years | 306 (4) | 0.21 (0.10 to 0.45) |
331 (230 to 377) |
| 9 years | 66 (1) | 0.06 (0.01 to 0.42) |
484 (299 to 510) |
| 12 years | 179 (1) | 0.74 (0.49 to 1.13) |
113 (56 more to 221 fewer) |
Subgroup analyses
There was no evidence that these effects varied by type of IOL lens material (test for interaction at one year, P = 0.77; two years, P = 0.80; three years, P = 0.53; five years, P = 0.92), but the number of studies in each group was small and the analyses were underpowered to detect differences (Analysis 1.1; Analysis 1.2; Analysis 1.3; Analysis 1.4).
Sensitivity analyses
We investigated the effect of excluding studies at risk of bias. We repeated the analyses removing studies judged to be at high risk of selection bias, performance bias or detection bias (Table 3). The estimates of effect for the outcome Nd:YAG capsulotomy were reasonably robust and did not change much when excluding studies at high risk of bias.
2. Sensitivity analyses: excluding studies at high risk of bias.
| Including all studies | Excluding studies at high risk of selection, performance or detection bias | |||||
| Analysis | Number of studies | Effect estimate | 95% CI | Number of studies | Effect estimate | 95% CI |
| Analysis 1.1 Nd:YAG capsulotomy at one year | 6 | Peto OR 0.30 | 0.05 to 1.74 | 4 | Peto OR 0.14 | 0.00 to 6.82 |
| Analysis 1.2 Nd:YAG capsulotomy at two years | 6 | RR 0.35 | 0.16 to 0.80 | 4 | RR 0.37 | 0.12 to 1.18 |
| Analysis 1.3 Nd:YAG capsulotomy at three years | 6 | RR 0.21 | 0.11 to 0.41 | 3 | RR 0.19 | 0.07 to 0.52 |
| Analysis 1.4 Nd:YAG capsulotomy at five years | 4 | RR 0.21 | 0.10 to 0.45 | 2 | RR 0.30 | 0.13 to 0.72 |
| Analysis 1.5 Nd:YAG capsulotomy at nine years | Not applicable only one study | ‐ | ‐ | ‐ | ‐ | ‐ |
| Analysis 1.6 Nd:YAG capsulotomy at 12 years | Not applicable only one study | ‐ | ‐ | ‐ | ‐ | ‐ |
| Analysis 1.8 BCDVA at one year | 2 | MD ‐0.06 logMAR | ‐0.12 to 0 | 1 | MD ‐0.06 logMAR | ‐0.14 to 0.02 |
| Analysis 1.8 BCDVA at two years | 2 | MD ‐0.01 logMAR | ‐0.05 to 0.02 | 1 | MD ‐0.01 logMAR | ‐0.04 to 0.03 |
| Analysis 1.8 BCDVA at three years | 2 | MD ‐0.09 logMAR | ‐0.22 to 0.03 | 1 | MD 0.03 logMAR | ‐0.19 to 0.25 |
| Analysis 1.8 BCDVA at five years | Not applicable only one study | ‐ | ‐ | ‐ | ‐ | ‐ |
CI: confidence interval; OR: odds ratio; RR: risk ratio; MD: mean difference
PCO score
Ten studies reported a PCO score (Analysis 1.7). Four studies reported the AQUA score, four studies reported the EPCO score and two studies reported another method of quantifying PCO. It was not possible to pool these data due to the way they were reported, but all studies consistently reported a statistically significant lower average PCO score (of the order of 0.5 to 3 units) with sharp‐edged IOLs compared with round‐edged IOLs. We judged this to be moderate‐certainty evidence, downgrading for risk of bias.
1.7. Analysis.
Comparison 1: Sharp versus round optic design, Outcome 7: PCO score
| PCO score | ||
| Study | Method of measuring PCO and method of dealing with people who had Nd:YAG capsulotomy | Mean difference between sharp and round edge (95% CI or P value) |
| Buehl 2005 | AQUA Score 0=clear capsule to 10=exceptionally severe PCO |
Mean difference (P value) 1 year: ‐1.18 (P < 0.05) 2 years: ‐1.38 (P < 0.05) 3 years: ‐1.36 (P < 0.05) including YAG cases 3 years: ‐1.05 (P < 0.05) excluding YAG cases |
| Buehl 2007 | AQUA Score 0=clear capsule to 10=exceptionally severe PCO |
Mean difference (P value) Excluding YAG cases 1 year: ‐0.83 (P < 0.05) 2 years: ‐0.86 (P < 0.05) 3 years: ‐0.88 (P < 0.05) Including YAG cases 2 years: ‐1.07 (P < 0.05) 3 years: ‐1.4 (P < 0.05) |
| Findl 2005 | AQUA Score 0=clear capsule to 10=exceptionally severe PCO |
Mean within‐person differences (95% CI) 1 year: ‐1.29 (‐0.51 to ‐2.07) 3 years: ‐2.31 (‐1.62 to ‐3.00) 5 years: ‐2.83 (‐1.66 to ‐4.00) |
| Haripriya 2017 | EPCO Score Density of opacification (0 to 4) times fractional area. Missing data due to YAG imputed (how?) |
Mean difference (P value) 1 year: ‐0.37 2 years: ‐0.056 (P < 0.05) 3 years: ‐0.362 (P < 0.05) 4 years: ‐0.432 (P < 0.05) 5 years: ‐0.673 (P < 0.05) 9 years: ‐0.999 (P < 0.05) |
| Hayashi 2005 | PCO value calculated by subtracting the scattering light density of the anterior IOL surface area from that of the posterior capsule area and averaged over 4 meridians. | "The PCO value in the sharp‐edge group was significantly less than that in the rounded‐edge group throughout the follow‐up period". |
| Mester 2004 | EPCO Score | Median within‐person difference (range) 1 year: ‐0.001 (‐0.855 to 0.328) 3 years: ‐0.069 (‐0.874 to 0.442) |
| Sacu 2005 | AQUA score | Mean difference (P value) 5 years: ‐1.2 (P = 0.001). |
| Shah 2007 | PCO score based on the percentage of the area within the capsulorhexis showing opacity using the POCO system | Median PCO 1 year:
"The square‐edged IOL eyes had significantly less PCO than the round‐edged IOL eyes (p<0.001, Wilcoxon signed rank test)." 2 years:
"The square‐edged IOL eyes had significantly less PCO than the round‐edged IOL eyes (p=0.006, Wilcoxon signed rank test)." |
| Sundelin 2005 | EPCO | "There was a statistically significant difference between the EPCO values for the two different IOLs at 2 (p=0.00014) and 3 years (p=0.02), but not at 1 year." |
| Wejde 2003 | EPCO | 2 years:
5 years:
12 years (Rönbeck 2014)
(note: HSM PMMA and Silicone IOLs were round‐edged, AcrySof group was sharp‐edged). |
Subgroup and sensitivity analyses could not be performed.
Best‐corrected distance visual acuity (BCDVA)
Three studies reported visual acuity as a continuous measure and are included in the analyses (Findl 2005; Shah 2007; Sundelin 2005), two studies reported visual acuity as a dichotomous variable (Haripriya 2017; Wejde 2003), three studies reported data in a form that could not be included in the review (Buehl 2005; Hayashi 2005; Sacu 2005) and two studies reported only on lack of statistical significance (Buehl 2007; Mester 2004).
Several studies showed better BCDVA in eyes with IOLs with sharp optic edges but the difference was small: one year postoperatively MD ‐0.06 logMAR (95% CI ‐0.12 to 0; 2 studies, 153 eyes; Analysis 1.8); two years postoperatively MD ‐0.01 logMAR (95% CI ‐0.05 to 0.02; 2 studies, 311 eyes; Analysis 1.9); three years postoperatively MD ‐0.09 logMAR (95% CI ‐0.22 to 0.03; 2 studies, 117 eyes; Analysis 1.10), and five years postoperatively MD ‐0.08 logMAR (95% CI ‐0.22 to 0.06; 1 study, 34 eyes; Analysis 1.11).
1.8. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 8: BCDVA (logMAR) at one year
1.9. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 9: BCDVA (logMAR) at two years
1.10. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 10: BCDVA (logMAR) at three years
1.11. Analysis.

Comparison 1: Sharp versus round optic design, Outcome 11: BCDVA (logMAR) at five years
Subgroup analyses
Subgroup analyses could not be performed due to the low number of studies in each IOL lens material group.
Sensitivity analyses
We investigated the effect of excluding studies at risk of bias. We repeated the analyses removing studies judged to be at high risk of selection bias, performance bias or detection bias (Table 3). The estimates of effect for BCDVA were not affected by excluding high risk of bias studies at one and two years but the effect was reduced (changed) at three years, however, the number of studies was small.
Quality of life
None of the studies reported quality of life data or other subjective outcomes.
Adverse events
Table 4 summarises the available information on adverse events. The included studies reported adverse effects inconsistently. There was little evidence of any important differences between groups.
3. Adverse events.
| Study | Intraoperative surgical complications | Entopic phenomena (e.g. negative dysphotopsia) | Clinically relevant IOL tilt and decentration | Distinctive anterior capsule fibrosis and capsule phimosis |
| Buehl 2005 | Not reported | "Concerning subjective visual symptoms, there were no differences between the groups. There were 1 case of day glare in the AR40 group and 2 cases in the AR40e group and 2 cases of halos and/or photophobia in each group." | "There was no significant difference between the 2 groups at any time for decentration of the IOL or capsulorhexis–IOL overlap (1 case of buttonholing in each group)." | "There was no difference between groups in cellular ongrowth on the IOL from the anterior capsule. There were 34 cases in both groups after 1 month, 6 cases in both groups after 1 year, and 1 case in both groups after 2 years; however, there was no case in either group 3 years after surgery." |
| Buehl 2007 | "There were no surgical complications that would have led to patient exclusion." | "There was also no statistically significant difference in subjective visual symptoms described by the patients (one case of day glare in the SI40 group and two cases in the Clariflex group; three cases of halos and/or photophobia in each group)." | "Two patients were excluded after surgery, one patient because of endophthalmitis and one patient because of pronounced fibrosis‐induced decentration of an SI40 IOL that required resurgery." | Not reported |
| Findl 2005 | "There were no surgical complications that would have led to patient exclusion." | Not reported | "We found no significant difference between the 2 groups at any time regarding decentration of the IOL (1 case in the sharp‐edge group), rhexis/IOL overlap (1 case of buttonholing in the round‐edge group), ..." | "We found no significant difference between the 2 groups at any time regarding... cases of cellular ongrowth onto the anterior IOL surface from the rhexis margin (chi‐square tests, all Ps % 0.05)." |
| Haripriya 2017 | "There were no surgical complications, such as posterior capsular rupture, leading to subject disqualification." | Not reported | Not reported | Not reported |
| Hayashi 2005 | "Furthermore, 1 surgeon performed all of the operations, and there were no surgical complications" | Not reported | Not reported | Not reported |
| Mester 2004 | "Of the 288 randomized patients, 41 (14%) had to be excluded from analyses due to various reasons: severe complications during surgery (10), no surgery or adverse events before second surgery (7)," | Not reported | "In all eyes, slitlamp examination revealed correct IOL positioning in the bag with no significant tilt or decentration." | "There was no difference between the 3 IOLs in anterior chamber cells, flare, or chronic inflammatory cells on the IOL surface by slitlamp examination." |
| Sacu 2005 | "There were no surgical complications that would have led to patient exclusion." | Not reported | Not reported | "One week postoperatively, full circumferential overlap of the peripheral optic by the anterior capsule leaf as a prerequisite for capsular bend formation was verified in all cases. In cases in which part of the optic circumference was exposed at the 5‐year follow‐up examination (one eye in the sharp edge and four eyes in the round edge optic group, P = 0.13), this had resulted from secondary buttonholing generally occurring during months 1 and 6." |
| Shah 2007 | "No serious surgical complications were seen." | Not reported | 6/107 in round‐edge group had decentered IOL compared with 4/107 in the sharp‐edge group. | 1 case of iris atrophy, 1 case of posterior synechiae, 41 cases capsulorhexis contact with IOL surface in round‐edge group. 51 cases of capsulorhexis contact with IOL surface in square‐edge IOL group. |
| Sundelin 2005 | "Surgical complications" 1 case in each group given as a reason for incomplete follow‐up. | Not reported | "At the 2‐month visit it was noticed that in some patients the optic was partly outside the capsulorhexis rim. This was due either to an asymmetric or overly large capsulorhexis, or to the IOL being slightly decentred in the capsular bag. In many patients the pupil could not be dilated sufficiently to inspect the entire optic/ capsulorhexis relationship at the 2‐month visit. A total of 113 patients attended the 2‐month visit; in 55 patients the entire capsulorhexis was on the optic, in 17 patients some part of the capsulorhexis was outside the optic edge, and in 41 patients the optic/capsulorhexis relationship could not be assessed" | Not reported |
| Wejde 2003 | Not reported | Not reported | Not reported | Not reported |
Intraoperative complications
Two studies did not report intraoperative complications (Buehl 2005; Wejde 2003).
Six studies reported that there were either no intraoperative complications (Hayashi 2005; Shah 2007), or no intraoperative complications leading to exclusion (Buehl 2007; Findl 2005; Haripriya 2017; Sacu 2005).
Two studies reported intraoperative complications. In Mester 2004, 10/288 people were excluded due to severe complications during surgery but it was not clear to which groups these people were allocated. In Sundelin 2005, one case in each group had "surgical complications" given as a reason for incomplete follow‐up.
Entopic phenomena ‐ negative dysphotopsia
Two studies reported cases of positive dysphotopsia (Buehl 2005; Buehl 2007). However, the number of reported cases was low and no difference could be found between different IOL optic edge designs.
Most studies did not report either the presence or absence of entopic phenomena. Two studies reported glare, halos and photophobia but these occurred rarely and in both groups (Buehl 2005; Buehl 2007).
Clinically relevant IOL tilt and decentration
Four studies did not report on IOL tilt or decentration (Haripriya 2017; Hayashi 2005; Sacu 2005; Wejde 2003).
One study reported that there was correct IOL position in all eyes with no important tilt or decentration (Mester 2004).
In the remaining five studies, IOL tilt or decentration was reported but in most cases this occurred rarely and evenly between groups.
Distinctive anterior capsule fibrosis and capsule phimosis
Five studies did not report on this (Buehl 2007; Haripriya 2017; Hayashi 2005; Sundelin 2005; Wejde 2003).
The remaining studies reported no important differences between groups.
Discussion
Summary of main results
We included 10 studies (1834 eyes) that compared sharp‐ and round‐edged IOLs. Eight of these studies were within‐person studies: one eye received a sharp‐edged IOL and the fellow eye a round‐edged IOL. The IOL materials were acrylic (two studies), silicone (four studies), PMMA (three studies) and different materials (one study). The studies were conducted in Austria, Germany, India, Japan, Sweden and the UK. We judged five studies to be at high risk of bias in at least one domain. We judged only two studies to be at low risk of bias in all domains.
Low‐certainty evidence suggested a reduction in the chance of Nd:YAG capsulotomy at one year with sharp‐edged IOLs compared to round‐edged IOLs: there were 7 fewer cases of Nd:YAG capsulotomy per 1000 (95% confidence interval (CI) 9 fewer to 7 more) (Peto odds ratio (OR) 0.30, 95% CI 0.05 to 1.74; 6 studies, 742 eyes). A similar relative effect was seen at two, three and five years. As the number of Nd:YAG capsulotomy events increased with longer follow‐up, the number of cases of Nd:YAG capsulotomy prevented increased over time. There was no evidence that these effects varied by type of IOL lens material but the number of studies in each group was small and the analyses underpowered to detect any differences in effect.
All of the studies reported a PCO score. Four studies reported the AQUA score, four studies reported the EPCO score and two studies reported another method of quantifying PCO. It was not possible to pool these data due to the way they were reported, but all studies consistently reported a lower average PCO score (of the order of 0.5 to 3 units) with sharp‐edged IOLs compared with round‐edged IOLs. We judged this to be moderate‐certainty evidence, downgrading for risk of bias.
Low‐certainty evidence suggested the logMAR visual acuity score was lower (better) in eyes that received a sharp‐edged IOL but the difference was small at one year and probably clinically unimportant. There was a greater difference observed at longer follow‐up times but statistically non‐significant results were less likely to be reported fully and incorporated in the analyses.
Very low‐certainty evidence on adverse events did not suggest any important differences between the groups. None of the studies reported quality of life.
Overall completeness and applicability of evidence
Participants in the included studies were likely to be representative of a standard cataract population. Although we did not exclude participants with ocular comorbidities in this review, most of the included studies excluded people with ocular comordibities. We believe that people with age‐related macular degeneration (AMD), glaucoma, diabetes and other sight‐threatening diseases should also be part of these study populations, as they are part of cataract populations in daily clinical practice. We assume that most authors excluded ophthalmic comorbidities to reduce noise, especially for visual acuity data, and make results comparable.
All of the study authors describe modern standard cataract surgery with implantation of an IOL. Nevertheless, many of the IOLs mentioned (including most PMMA IOLs) in the included studies are no longer being used and are unavailable on the market. Some of the included studies were published more than 15 years ago. In the meantime, scientists and manufacturers have produced advances in IOL technology. As a result, many new foldable IOLs with better biocompatibility, lower PCO rates and possibly with better visual acuity are on the market today.
We found that, independent of the IOL material and design, only very few Nd:YAG capsulotomies were reported one year after cataract surgery. We decided to include longer term follow‐ups at two, three, five, nine and 12 years, in addition to our initially defined primary time point. At these later time points, significant differences concerning PCO formation between different IOL designs could be highlighted. The effect of higher PCO proportion with longer postoperative follow‐ups corresponds well with clinical experience, because ‐ with modern IOL designs ‐ only very few laser treatments need to be performed within the first year of cataract surgery.
Only two studies reported cases of dysphotopsia. In both studies, the number of participants affected was low and equal for both IOLs with sharp and round optic edge design. Nevertheless, according to the literature, the number of people experiencing optical side effects associated with IOL material and design is high, and dysphotopsia in pseudophakic eyes is a leading cause of low postoperative patient satisfaction (Kinard 2013). Therefore, the studies included in this review – which focused on objective outcomes such as PCO and capsulotomy rates – may not sufficiently capture subjective outcomes.
Quality of the evidence
The studies were poorly reported in general, and several studies were at risk of bias in one or more domain. In general, we downgraded for risk of bias. Some of the outcomes were imprecisely measured, particularly at shorter follow‐up times with fewer events. The results were largely consistent. We did not have enough studies to reliably assess publication bias.
Potential biases in the review process
Our prespecified primary outcome was the proportion of eyes with Nd:YAG capsulotomy one year after surgery. However, the number of events at this time point was low, and so we made a post hoc decision to include and present data for longer time periods. We think this is justified but it should be noted as a post hoc decision.
The majority of studies included in this review were within‐person studies whereby one eye received one type of lens and the other eye received the other type. This is a paired design and, as we specified in our methods, ideally the included studies should have reported a paired analysis. However, a paired analysis was not done consistently for all outcomes. We have used the data as reported. This is a conservative approach ‐ confidence intervals are wider than if a paired analysis had been done.
Agreements and disagreements with other studies or reviews
Other reviews and meta‐analyses have shown similar findings to the current review; namely, that IOLs with sharp optic edges are associated with less PCO, lower laser capsulotomy rates and better visual acuity compared with IOLs with round optic edges (Cheng 2007). A recent network meta‐analysis sponsored by Alcon compared sharp‐edged lens Acrysof with other lenses and concluded that this sharp‐edged lens had a lower risk of Nd:YAG capsulotomy compared to all the other round‐edged IOLs (Thom 2019). This network meta‐analysis included more studies than the current review. This is because we restricted our search to studies that formally compared optic edge design.
Authors' conclusions
Implications for practice.
This review provides evidence that sharp‐edged IOLs are likely to be associated with less PCO formation than round‐edged IOLs and less Nd:YAG capsulotomy but the evidence on visual acuity was less certain. The impact of these lenses on quality of life has not been assessed and there is only very low‐certainty evidence on adverse events.
Most of the IOLs with round optic edges used in the studies included in this review have been removed from the market and are no longer implanted during cataract surgery.
Implications for research.
Study authors used different PCO scores to describe the amount of PCO in their studies which meant we were unable to do a meta‐analysis for this outcome. Ideally, the measurement of PCO should be standardised between studies in order to make meta‐analyses possible in the future.
Additionally, none of the included studies used questionnaires to assess the impact of PCO on participants' quality of life and subjective impairment. We suggest that the use of subjective parameters, such as quality of life scores, would be important for future studies.
At present, new IOLs are licensed and introduced without adequate evidence of safety and effectiveness. Surgical clinical registries are important for collecting longer‐term data. Severe adverse events may occur rarely and may not be observed in sufficient numbers to robustly assess differences between IOLs in small, short‐term RCTs, such as those included in this review.
Eight of the 10 studies included in this review had a paired design but did not consistently report measures of effect based on a paired analysis. In addition, quality of life cannot be assessed in such designs. We recommend parallel‐group studies whereby people are randomly allocated to treatment in order to get a complete picture of the effects of these lenses.
All pre‐specified outcomes should be fully reported, irrespective of whether the results are statistically significant or not.
History
Protocol first published: Issue 1, 2017
Notes
None.
Acknowledgements
The Cochrane Eyes and Vision editorial team developed and executed the electronic searches. We thank Catey Bunce and Mayank Nanavaty for comments on the protocol and full review, and Anupa Shah for her assistance with the editorial process.
Appendices
Appendix 1. CENTRAL search strategy
#1 MeSH descriptor: [Cataract Extraction] explode all trees #2 phacoemulsificat* or capsulorhexis #3 cataract* near/3 (extract* or surg*) #4 MeSH descriptor: [Lenses, Intraocular] explode all trees #5 MeSH descriptor: [Lens Implantation, Intraocular] explode all trees #6 MeSH descriptor: [Lens Capsule, Crystalline] explode all trees #7 MeSH descriptor: [Capsule Opacification] explode all trees #8 posterior near/3 capsul* near/3 opaci* #9 PCO #10 #1 or #2 or #3 or #4 or #5 or #6 or #7 or #8 or #9 #11 round‐edged #12 sharp‐edged #13 square‐edged #14 sharp‐optic* #15 (round* or sharp* or optic* or haptic*) near/6 (edge*) #16 (round* or sharp* or optic* or haptic*) near/6 (design*) #17 #11 or #12 or #13 or #14 or #15 or #16 #18 #10 and #17
Appendix 2. MEDLINE Ovid search strategy
1. randomized controlled trial.pt. 2. (randomized or randomised).ab,ti. 3. placebo.ab,ti. 4. dt.fs. 5. randomly.ab,ti. 6. trial.ab,ti. 7. groups.ab,ti. 8. or/1‐7 9. exp animals/ 10. exp humans/ 11. 9 not (9 and 10) 12. 8 not 11 13. exp cataract extraction/ 14. (phacoemulsificat$ or capsulorhexis).tw. 15. ((extract$ or surg$) adj3 cataract$).tw. 16. exp lenses, intraocular/ 17. exp lens implantation, intraocular/ 18. exp lens capsule crystalline/ 19. Capsule Opacification/ 20. (posterior adj3 capsul$ adj3 opaci$).tw. 21. PCO.tw. 22. or/13‐21 23. round‐edged.tw. 24. sharp‐edged.tw. 25. square‐edged.tw. 26. sharp‐optic$.tw. 27. ((round$ or sharp$ or optic$ or haptic$) adj6 edge$).tw. 28. ((round$ or sharp$ or optic$ or haptic$) adj6 design$).tw. 29. ((lens or lenses) adj3 design$).tw. 30. or/23‐29 31. 22 and 30 32. 12 and 31 33. or/23‐28 34. 22 and 33 35. 12 and 34
The search filter for trials at the beginning of the MEDLINE strategy was from the published paper by Glanville 2006.
Appendix 3. Embase Ovid search strategy
1. exp randomized controlled trial/ 2. exp randomization/ 3. exp double blind procedure/ 4. exp single blind procedure/ 5. random$.tw. 6. or/1‐5 7. (animal or animal experiment).sh. 8. human.sh. 9. 7 and 8 10. 7 not 9 11. 6 not 10 12. exp clinical trial/ 13. (clin$ adj3 trial$).tw. 14. ((singl$ or doubl$ or trebl$ or tripl$) adj3 (blind$ or mask$)).tw. 15. exp placebo/ 16. placebo$.tw. 17. random$.tw. 18. exp experimental design/ 19. exp crossover procedure/ 20. exp control group/ 21. exp latin square design/ 22. or/12‐21 23. 22 not 10 24. 23 not 11 25. exp comparative study/ 26. exp evaluation/ 27. exp prospective study/ 28. (control$ or prospectiv$ or volunteer$).tw. 29. or/25‐28 30. 29 not 10 31. 30 not (11 or 23) 32. 11 or 24 or 31 33. exp cataract extraction/ 34. (phacoemulsificat$ or capsulorhexis).tw. 35. ((extract$ or surg$) adj3 cataract$).tw. 36. lens implantation/ 37. lens implant/ 38. exp lens capsule/ 39. exp aftercataract/ 40. (posterior adj3 capsul$ adj3 opaci$).tw. 41. PCO.tw. 42. or/33‐41 43. round‐edged.tw. 44. sharp‐edged.tw. 45. square‐edged.tw. 46. sharp‐optic$.tw. 47. ((round$ or sharp$ or optic$ or haptic$) adj6 edge$).tw. 48. ((round$ or sharp$ or optic$ or haptic$) adj6 design$).tw. 49. ((lens or lenses) adj3 design$).tw. 50. or/43‐49 51. 42 or 50 52. 32 and 51
Appendix 4. LILACS search strategy
cataract or phacoemulsificat$ or capsule opacification and round$ or sharp$ or optic$ or haptic and lens$ or design$
Appendix 5. ISRCTN search strategy
(sharp or round or square) and lens
Appendix 6. ClinicalTrials.gov search strategy
(Sharp OR Round OR Square) AND Lens
Appendix 7. WHO ICTRP search strategy
Three searches run on basic search screen.
Sharp AND Lens Round AND Lens Square AND Lens
Data and analyses
Comparison 1. Sharp versus round optic design.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1.1 Nd:YAG capsulotomy at one year | 6 | 742 | Peto Odds Ratio (Peto, Fixed, 95% CI) | 0.30 [0.05, 1.74] |
| 1.1.1 Acrylic | 1 | 92 | Peto Odds Ratio (Peto, Fixed, 95% CI) | 0.14 [0.00, 6.82] |
| 1.1.2 PMMA | 3 | 366 | Peto Odds Ratio (Peto, Fixed, 95% CI) | 0.14 [0.00, 6.82] |
| 1.1.3 Silicone | 2 | 284 | Peto Odds Ratio (Peto, Fixed, 95% CI) | 0.51 [0.05, 4.96] |
| 1.2 Nd:YAG capsulotomy at two years | 6 | 703 | Risk Ratio (M‐H, Random, 95% CI) | 0.35 [0.16, 0.80] |
| 1.2.1 Acrylic | 2 | 204 | Risk Ratio (M‐H, Random, 95% CI) | 0.13 [0.02, 0.72] |
| 1.2.2 PMMA | 2 | 298 | Risk Ratio (M‐H, Random, 95% CI) | 0.34 [0.02, 4.80] |
| 1.2.3 Silicone | 1 | 68 | Risk Ratio (M‐H, Random, 95% CI) | 0.25 [0.03, 2.12] |
| 1.2.4 Mixed materials | 1 | 133 | Risk Ratio (M‐H, Random, 95% CI) | 0.38 [0.12, 1.19] |
| 1.3 Nd:YAG capsulotomy at three years | 6 | 538 | Risk Ratio (M‐H, Random, 95% CI) | 0.21 [0.11, 0.41] |
| 1.3.1 Acrylic | 1 | 62 | Risk Ratio (M‐H, Random, 95% CI) | 0.08 [0.01, 0.60] |
| 1.3.2 PMMA | 2 | 116 | Risk Ratio (M‐H, Random, 95% CI) | 0.15 [0.02, 1.13] |
| 1.3.3 Silicone | 3 | 360 | Risk Ratio (M‐H, Random, 95% CI) | 0.27 [0.12, 0.62] |
| 1.4 Nd:YAG capsulotomy at five years | 4 | 306 | Risk Ratio (M‐H, Random, 95% CI) | 0.21 [0.10, 0.45] |
| 1.4.1 PMMA | 2 | 106 | Risk Ratio (M‐H, Random, 95% CI) | 0.13 [0.01, 2.20] |
| 1.4.2 Silicone | 1 | 48 | Risk Ratio (M‐H, Random, 95% CI) | 0.11 [0.01, 1.96] |
| 1.4.3 Mixed material | 1 | 152 | Risk Ratio (M‐H, Random, 95% CI) | 0.19 [0.07, 0.50] |
| 1.5 Nd:YAG capsulotomy at 9 years | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 1.6 Nd:YAG capsulotomy at 12 years | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 1.7 PCO score | 10 | Other data | No numeric data | |
| 1.8 BCDVA (logMAR) at one year | 2 | 153 | Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.12, 0.00] |
| 1.8.1 PMMA | 1 | 46 | Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.14, 0.02] |
| 1.8.2 Silicone | 1 | 107 | Mean Difference (IV, Fixed, 95% CI) | ‐0.06 [‐0.16, 0.04] |
| 1.9 BCDVA (logMAR) at two years | 2 | 311 | Mean Difference (IV, Fixed, 95% CI) | ‐0.01 [‐0.05, 0.02] |
| 1.9.1 PMMA | 1 | 214 | Mean Difference (IV, Fixed, 95% CI) | ‐0.01 [‐0.04, 0.03] |
| 1.9.2 Silicone | 1 | 97 | Mean Difference (IV, Fixed, 95% CI) | ‐0.11 [‐0.25, 0.03] |
| 1.10 BCDVA (logMAR) at three years | 2 | 117 | Mean Difference (IV, Fixed, 95% CI) | ‐0.09 [‐0.22, 0.03] |
| 1.10.1 PMMA | 1 | 34 | Mean Difference (IV, Fixed, 95% CI) | 0.03 [‐0.19, 0.25] |
| 1.10.2 Silicone | 1 | 83 | Mean Difference (IV, Fixed, 95% CI) | ‐0.15 [‐0.30, 0.00] |
| 1.11 BCDVA (logMAR) at five years | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected |
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Buehl 2005.
| Study characteristics | ||
| Methods |
Study design: within‐person study Eyes: bilateral age‐related cataract, enrolled for surgery in both eyes and good overall physical condition, no history of other ocular diseases or intraocular surgery, laser treatment, diabetes requiring medical control, glaucoma, or retinal pathology that would make a postoperative visual acuity of 20/40 (decimal equivalent 0.5) or better unlike Cataract surgery: standardised technique, one surgeon Postoperative treatment: prednisolone acetate 1% (Ultracortenol) and diclofenac (Voltaren Ophtha) eyedrops 4 times a day for 1 month Criteria for Nd:YAG capsulotomy: visual acuity less than 20/30 (decimal equivalent 0.7) or subjective symptoms attributable to the slit lamp finding |
|
| Participants |
Country: Austria Number of participants (eyes) randomised: 53 (106) Inclusion criteria: bilateral age‐related cataract, good overall physical condition Exclusion criteria: history of other ocular disease or intraocular surgery, laser treatment, diabetes requiring medical control, glaucoma, retinal pathology that would make a postoperative visual acuity of 20/40 (decimal equivalent 0.5) or better unlikely. Average age (range): 72 years (range not reported) % women: not reported |
|
| Interventions |
Intervention characteristics IOL with sharp optic edge design (46 participants, 46 eyes)
IOL with round optic edge design (46 participants, 46 eyes)
|
|
| Outcomes |
Primary outcome: PCO (subjective slit lamp grading, PCO AQUA score) Secondary outcome: YAG capsulotomy proportion, visual acuity (Snellen) Follow‐up: 1 week, 1 month, 6 months, 1 year, 2 years and 3 years after surgery |
|
| Identification |
Setting: Department of Ophthalmology, Medical University of Vienna, Austria Author's name: Oliver Findl, MD Institution: Department of Ophthalmology, Medical University of Vienna, Austria Email: oliver.findl@meduniwien.ac.at Address: Medical University of Vienna, Dep. of Ophthalmology, Währinger Gürtel 18‐20, 1090 Wien, Austria Contact with investigators: O.Findl is co‐author of this review |
|
| Notes |
Funding: institution driven Conflict of interest: none Date study conducted: not available Trial registration ID: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "The IOL type for the first surgically treated eye of each patient was randomly assigned prior to patient recruitment." Judgement comment: unclear how the allocation schedule was generated |
| Allocation concealment (selection bias) | Unclear risk | Judgement comment: exact procedure of randomisation and allocation not reported |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Judgement comment: stated as "patient‐ and examiner‐masked" ‐ no exact methods to guarantee masking were described |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Judgement comment: "examiner‐masked" ‐ exact technique not stated |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Judgement comment: problem of missing PCO score and BCVA after capsulotomy is addressed with estimation of missing values |
| Selective reporting (reporting bias) | Low risk | Judgement comment: no SD for PCO scores available. No absolute BCVA values available ‐ these are only mentioned in the discussion section. |
| Other bias | Low risk | None |
Buehl 2007.
| Study characteristics | ||
| Methods |
Study design: within‐person study Eyes: bilateral age‐related cataract, enrolled for surgery in both eyes and good overall physical condition, no history of other ocular diseases or intraocular surgery, laser treatment, diabetes requiring medical control, glaucoma, or retinal pathology that would make a postoperative visual acuity of 20/40 (decimal equivalent 0.5) or better unlike Cataract surgery: standardised technique, one surgeon Postoperative treatment: prednisolone acetate 1% (Ultracortenol; Ciba Vision, Duluth, Georgia, USA) and diclofenac (Voltaren Ophtha; Ciba Vision) eyedrops 4 times a day for 1 month Criteria for Nd:YAG capsulotomy: visual acuity less than 20/30 (decimal equivalent 0.7) or subjective symptoms attributable to the slit lamp findings |
|
| Participants |
Country: Austria Number of participants (eyes) randomised: 52 (104) Overall Inclusion criteria: bilateral age‐related cataract scheduled for surgery on both eyes, good overall physical condition. Exclusion criteria: history of other ocular disease or intraocular surgery, laser treatment, diabetes requiring medical control, glaucoma, or retinal pathology that would make a postoperative visual acuity of 20/40 (decimal equivalent 0.5) or better unlikely Pretreatment: Average age (range): 77 years (range not reported) % women: not reported |
|
| Interventions |
Intervention characteristics IOL with sharp optic edge design
IOL with round optic edge design
|
|
| Outcomes |
Primary outcome: PCO (subjective slit lamp grading, PCO AQUA score) Secondary outcome: YAG capsulotomy proportion, visual acuity (Snellen) Follow‐up: 1 week, 1 month, 6 months, 1 year, 2 years, and 3 years after surgery |
|
| Identification |
Setting: Department of Ophthalmology, Medical University of Vienna, Vienna, Austria. Comments: Author's name: Rupert Menapace Institution: Department of Ophthalmology, Medical University of Vienna Email: rupert.menapace@meduniwien.ac.at Address: Department of Ophthalmology, Medical University of ViennaWähringer Gürtel 18‐20 1090 Wien, Austria Contact with investigators: O. Findl is co‐author of this review |
|
| Notes |
Funding: institution driven Conflict of interest: none Date study conducted: surgery was performed between June 2001 and February 2002 Trial registration ID: NCT00428363 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "The IOL type for the first operated eye of each patient was randomly assigned before patient recruitment." Judgement comment: unclear how the allocation schedule was generated |
| Allocation concealment (selection bias) | Unclear risk | Judgement comment: study was described as masked for patient and examiner but allocation concealment not clearly describedIOL chosen by randomisation process of randomisation not described IOL |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Judgement comment: "patient and examiner‐masked"; no exact description of masking process |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Judgement comment: "patient and examiner‐masked"; no exact description of masking process |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | within‐person study and no indication of attrition bias |
| Selective reporting (reporting bias) | High risk | Judgement comment: missing absolute BCVA values (ORBIT classification A, high risk of bias), missing SD for PCO score |
| Other bias | Low risk | None |
Findl 2005.
| Study characteristics | ||
| Methods |
Study design: within‐person study Eyes: bilateral, age‐related cataract and good overall physical constitution (i.e. they should be able to attend all follow‐up examinations without the help of others, and they should not suffer from a major eye disease except cataract) Cataract surgery: standardised surgical technique, one surgeon Postoperative treatment: prednisolone acetate (1%) eyedrops (Ultracortenol, Ciba Vision, Atlanta, GA) and diclofenac (Voltaren Ophtha, Ciba Vision, Atlanta, GA) 4 times a day for 1 month |
|
| Participants |
Country: Austria Number of participants (eyes) randomised: 32 (64) Overall Inclusion criteria: bilateral, age‐related cataract, good overall physical constitution Exclusion criteria: Exclusion criteria were a history of ocular disease or intraocular surgery, laser treatment, diabetes requiring medical control, glaucoma, and severe retinal pathology that would make a postoperative visual acuity of 20/40 (decimal equivalent, 0.5) or better unlikely Average age (range): 74 years (range not reported) % women: not reported |
|
| Interventions |
Intervention characteristics IOL with sharp optic edge design (23 participants, 23 eyes)
IOL with round optic edge design (23 participants, 23 eyes)
|
|
| Outcomes |
Primary outcome: PCO (subjective slit lamp grading, PCO AQUA score) Secondary outcome: YAG capsulotomy proportion, visual acuity (Snellen), other slit lamp findings, IOL position and centration, rhexis/IOL overlap Follow‐up: 1 week, 1 month, 1 year, 3 years, and 5 years after surgery |
|
| Identification |
Setting: Department of Ophthalmology, Medical University of Vienna, Vienna, Austria Comments: Author's name: Oliver Findl, MD Institution: Department of Ophthalmology, Medical University of Vienna, Austria Email: oliver.findl@meduniwien.ac.at. Address: Department of Ophthalmology, Medical University of Vienna, Waehringer Guertel 18–20, Wien, 1090 Austria. Contact with investigators: O.Findl is co‐author of this review |
|
| Notes |
Funding: institution driven Conflict of interest: none Date study conducted: not available Trial registration ID: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "The IOL type for the first operated eye of each patient was randomly assigned prior to patient recruitment. Randomization blocks of 4 were derived from a list of random numbers to ensure an equal number of first eyes to be operated on in each group." |
| Allocation concealment (selection bias) | Low risk | Judgement comment: IOL type for first eye to be operated was randomly assigned prior to participant recruitment. Randomisation in blocks to ensure equal number of first eyes to be operated on each group |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Judgement comment: participants, examiner and surgeon were masked. Surgeon was unmasked after irrigation/aspiration |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Judgement comment: examiners were masked to IOL choice; participants and examiners were masked to the order of the implanted IOLs. The surgeon was unmasked to allocation after completion of irrigation/aspiration of cortical remnants. Examiners were masked to IOL choice |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Judgement comment: PCO score values were estimated for eyes after capsulotomy |
| Selective reporting (reporting bias) | Low risk | Judgement comment: no evidence for risk of bias |
| Other bias | Low risk | Judgement comment: none |
Haripriya 2017.
| Study characteristics | ||
| Methods |
Study design: within‐person study Eyes: bilateral age‐related cataract, pupillary dilation greater than 7 mm, being scheduled for second‐eye surgery within 3 months of the first‐eye surgery Surgical technique: standardised surgical technique, 3 surgeons Postoperative treatment: unknown Indication for capsulotomy: visually significant PCO accounting for BCVA of 6/9 or less |
|
| Participants |
Country: India Number of participants (eyes) randomised: 100 (200) Inclusion criteria: bilateral age‐related cataract; pupillary dilation greater than 7 mm; being scheduled for second‐eye surgery within 3 months of the first‐eye surgery Exclusion criteria: corneal pathology, glaucoma, shallow anterior chamber, expected zonulopathy, pseudoexfoliation, any vision‐impairing posterior segment pathology, traumatic cataract, complicated cataract, dense posterior subcapsular cataract, and posterior polar cataract Average age (range): 53 years (40 to 65) % women: 68% |
|
| Interventions |
Intervention characteristics Group A: ‐ IOL with sharp optic edge design (50 participants, 50 eyes)
‐ IOL with round optic edge design (50 participants, 50 eyes)
(Group B (not included in review) ‐ IOL with sharp optic edge design (50 participants, 50 eyes)
‐ IOL with round optic edge design (50 participants, 50 eyes)
|
|
| Outcomes |
Primary outcome: PCO (EPCO PCO Score (0‐4)) Secondary outcome: YAG capsulotomy proportion, visual acuity (Snellen) Follow‐up: 1 day, 1 month, and years 1, 2, 3, 4, 5, and 9 after surgery |
|
| Identification |
Setting: Aravind Eye Hospital, Madurai, India Author's name: Aravind Haripriya, MD Institution: Aravind Eye Hospital, Madurai, India Email: haripriya@aravind.org Address: Aravind Eye Hospital, 1, Anna Nagar, Madurai 625020, India. |
|
| Notes |
Funding: institution driven Conflict of interest: none Date study conducted: 2006‐2016 Trial registration Id: NCT00312299 |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "After complete preoperative workup, the patients were randomized into 2 groups of 50 patients each using a computer generated randomization list." "The 2 subrandomization tables were used to determine which of the 2 IOLs was implanted in the first and second eyes." |
| Allocation concealment (selection bias) | Unclear risk | Judgement comment: participants were randomised to different groups, no exact statement about generation of randomisation list |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Judgement comment: The participants were masked as to which eye had which IOL. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Judgement comment: no masking of examiners (single‐masked study) |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Judgement comment: missing PCO score values due to capsulotomy were estimated. Judgement comment: BCVA values missing ‐ only percentages |
| Selective reporting (reporting bias) | Low risk | None |
| Other bias | Low risk | None |
Hayashi 2005.
| Study characteristics | ||
| Methods |
Study design: within‐person study Eyes: bilateral cataract planned to undergo bilateral phacoemulsification surgery and IOL implantation Surgical technique: standardised surgical technique, performed by one surgeon Postoperative treatment: unclear Indication for Nd:YAG capsulotomy: loss of 2 or more lines of visual acuity or blurred vision |
|
| Participants |
Country: Japan Number of participants (eyes) randomised: 75 (150) Inclusion criteria: bilateral phacoemulsification surgery and IOL implantation scheduled Exclusion criteria: ocular pathologic features other than senile cataract, history of prior ocular surgery or inflammation, eyes scheduled for planned extracapsular cataract extraction with nucleus expression, a pupil diameter less than 6.0 mm after full dilation, people with diabetes mellitus, and people who could not be available for follow‐up. Pretreatment: Average age (range): 70 years (53 to 88) % women: 68% |
|
| Interventions |
Intervention characteristics IOL with sharp optic edge design (75 participants, 75 eyes)
IOL with round optic edge design (75 participants, 75 eyes)
|
|
| Outcomes |
Primary outcome: PCO (customized PCO scoring with Scheimpflug imaging and measurement of scattering light density, unit: CCT (computer‐compatible‐tape steps), 0‐255 only reported in graphs, no absolute values ‐ cannot be used for analysis Secondary outcome: YAG capsulotomy proportion, visual acuity (Snellen), contrast sensitivity and glare sensitivity Follow‐up: 1, 3, 6, 12, 18, and 24 months after surgery Contact with investigators: none |
|
| Identification |
Setting: Hayashi Eye Hospital, Fukuoka, Japan. Author's name: Ken Hayashi, MD Institution: Hayashi Eye Hospital, Fukuoka, Japan Email: hayashi‐ken@hayashi.or.jp Address: Hayashi EyeHospital, 4‐7‐13 Hakataekimae, Hakata‐Ku, Fukuoka 812, Japan |
|
| Notes |
Funding: unknown Conflict of interest: none Date study conducted: not available Trial registration ID: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "The controller of this clinical trial generated a randomization code with equal numbers using random number tables and kept concealed the assignment schedule until all data were collected to ensure allocation concealment." |
| Allocation concealment (selection bias) | Low risk | Quote: "The controller of this clinical trial generated a randomization code with equal numbers using random number tables and kept concealed the assignment schedule until all data were collected to ensure allocation concealment." |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Judgement comment: participants, surgeon and examiners were masked, operating theatre personnel were not informed about purpose of study |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Judgement comment: participants, surgeon and examiners were masked, operating theatre personnel were not informed about purpose of study |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Judgement comment: within‐person study and equal numbers followed up in each group |
| Selective reporting (reporting bias) | Low risk | Judgement comment: no evidence for selective outcome reporting |
| Other bias | Low risk | Judgement comment: no evidence for other sources of bias |
Mester 2004.
| Study characteristics | ||
| Methods |
Study design: within‐person study Eyes: eyes of participants, who were 50 years or older at the time of surgery, were of any race and gender, in need of bilateral surgery for senile cataract, had potential to achieve 0.5 (20/40) visual acuity in both eyes, were in good overall physical constitution, and gave informed consent Cataract surgery: one surgeon for each centre, standardised surgical technique Postoperative treatment: unknown Indication for Nd:YAG capsulotomy: no standardised criteria |
|
| Participants |
Country: Germany Number of participants (eyes) randomised: 288 (576) Inclusion criteria: age of 50 years or older, bilateral age‐related cataract, potential visual acuity of 0.5 (20/40) in both eyes, good overall physical constitution, and a signed informed consent Exclusion criteria: history of ocular disease or surgery and intraoperative complications Average age (range): 71 years (range not reported) % women: 55% |
|
| Interventions |
Intervention characteristics IOL with sharp optic edge design (102 participants, 102 eyes)
IOL with round optic edge design (102 participants, 102 eyes)
Group 2 (not included in review) IOL with sharp optic edge design (127 participants, 127 eyes)
IOL with sharp optic edge design (127 participants, 127 eyes)
|
|
| Outcomes |
Primary outcome: PCO score (EPCO Score) Secondary outcome: YAG capsulotomy proportion, visual acuity Follow‐ups: 1 day, 1 week, 6 months, and 1 year postoperatively |
|
| Identification |
Setting: 7 German ophthalmological hospitals Author's name: Ulrich Mester, MD Institution: Department of Ophthalmology, Bundesknappschaft’s Hospital, Sulzbach, Germany Email: mester@kksulzbach.de Address: Department of Ophthalmology, Bundesknappschaft’s Hospital, An der Klinik 10, 66280 Sulzbach, Germany |
|
| Notes |
Funding: research grant by Pharmacia, Erlangen, Germany Conflict of interest: none Date study conducted: not available Trial registration ID: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "The IOL type for the first operated eye was randomly assigned." |
| Allocation concealment (selection bias) | Unclear risk | Judgement comment: not reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Judgement comment: no masking of participants or examiners |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Judgement comment: no masking of participants or examiners |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | Judgement comment: low risk of bias due to incomplete outcome data ‐ small no. of excluded eyes |
| Selective reporting (reporting bias) | High risk | Judgement comment: ORBIT classification A (high risk of bias) for visual acuity. |
| Other bias | Low risk | None |
Sacu 2005.
| Study characteristics | ||
| Methods |
Study design: within‐person study Eyes: bilateral age‐related cataract Cataract surgery: standardised surgical technique, one surgeon Postoperative treatment: prednisolone ointment. Therapy was started after the first visit 18 to 24 hours postoperatively with diclofenac and prednisolone acetate 0.5% 4 times a day for 1 month Indication for Nd:YAG capsulotomy: reported complaints, visual acuity of 20/25 (decimal equivalent 0.8) or less that was attributable to central PCO formation at slit lamp exam |
|
| Participants |
Country: Austria Number of participants (eyes) randomised: 51 (102) Inclusion criteria: bilateral age‐related cataract and good overall physical constitution Exclusion criteria: history of other ocular diseases or intraocular surgery, previous laser treatment, diabetes requiring medical control, glaucoma, and retinal pathology that would make a postoperative visual acuity of 20/40 (decimal equivalent 0.5) or better unlikely. Average age (range): 73 years (range not reported) % women: not reported |
|
| Interventions |
Intervention characteristics IOL with sharp optic edge design (24 participants, 24 eyes)
IOL with round optic edge design (24 participants, 24 eyes)
|
|
| Outcomes |
Primary outcome: BCDVA (Snellen) PCO (PCO AQUA score) Secondary outcomes: slit lamp examination results (rhexis or IOL overlap, amount of anterior capsule opacification (ACO), and the amount and subtypes of fibrotic PCO), Nd:YAG capsulotomy proportion Follow‐up: 5 years after surgery |
|
| Identification |
Setting: Medical University of Vienna, Department of Ophthalmology, Vienna, Austria. Author's name: Rupert Menapace, MD Institution: Medical University of Vienna, Department of Ophthalmology, Vienna, Austria Email: rupert.menapace@meduniwien.ac.at Address: Medical University of Vienna, Department of Ophthalmology, Waehringer Guertel 18–20, A‐1090 Vienna, Austria Contact with the investigator: O.Findl is co‐author of this review |
|
| Notes |
Funding: institutional driven Conflict of interest: none Date study conducted: surgery was performed between November 1998 and June 1999 Trial registration ID: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Quote: "The IOL model (S or R) for the first operated eye of each patient was randomly assigned before surgery." |
| Allocation concealment (selection bias) | Unclear risk | Judgement comment: IOL choice according to randomisation; no detailed information about allocation concealment |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Judgement comment: study was described as double‐masked but unclear if all personnel were maskedstudy participant and examiner‐masked? was surgeon masked? |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Judgement comment: participant‐ examiners were masked |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | None |
| Selective reporting (reporting bias) | Low risk | Judgement comment: no evidence for selective outcome reporting could be found |
| Other bias | Low risk | Judgement comment: no evidence for other sources of bias |
Shah 2007.
| Study characteristics | ||
| Methods |
Study design: within‐person study Eyes: bilateral age‐related cataracts in otherwise normal eyes Cataract surgery: standardised surgical technique, one surgeon Postoperative treatment: prednisolone 1% and ciprofloxacin 0.3% eyedrops 4 times a day for 1 month and then tapered, with tropicamide 1% eyedrops once at night for 1 week. Systemic ciprofloxacin 500 mg was given orally twice a day for 5 days starting 1 day before surgery, and diclofenac sodium 50 mg was given orally twice a day for 5 days after surgery Indications for Nd:YAG capsulotomy: no statement concerning standardised indications |
|
| Participants |
Country: India, United Kingdom Number of participants (eyes) randomised: 118 (236) Inclusion criteria: bilateral age‐related cataracts in otherwise normal eyes, informed consent, preoperative dilated pupil diameter greater than 6.0 mm, willingness to have the second eye operated on within 1 month, ability to attend follow‐up visits, and expectation of postoperative visual acuity better than 6/12. Exclusion criteria: previous eye surgery, intraocular disease, significant corneal scarring, topical ophthalmic medication, medical treatment for diabetes, and capsule complications at surgery. Average age (range): 58 years (45 to 75) % women: 41% |
|
| Interventions |
Intervention characteristics IOL with sharp optic edge design (115 participants, 115 eyes)
IOL with round optic edge design (115 participants, 115 eyes)
|
|
| Outcomes |
Primary outcome: PCO score (POCO system) Secondary outcomes: uncorrected (UDVA) and corrected distance visual acuity (BCDVA), intraocular pressure, slit lamp examination findings of the anterior chamber, capsulorhexis contact with the anterior IOL surface, Nd:YAG capsulotomy proportion Follow‐up: 1 and 2 years after surgery |
|
| Identification |
Setting: eye hospital in Dholka, Ahmedabad, Gujarat, India Author's name: DJ Spalton, MD Institution: St. Thomas’ Hospital, London, United Kingdom Email: dspalton@hotmail.com Address: St. Thomas’ Hospital, Lambeth Palace Road, London, United Kingdom Contact with the investigator: none |
|
| Notes |
Funding: Supported by a grant from the Star Foundation, London, and from Sight Savers, Haywards Heath, United Kingdom. Rayner and Co., Hove, United Kingdom, donated the intraocular lenses. Conflict of interest: none Date study conducted: unknown Trial registration ID: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "A simple blocked randomization schedule was used for this purpose." |
| Allocation concealment (selection bias) | Unclear risk | Judgement comment: not reported |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Judgement comment: participants and examiner were masked to choice of IOL |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | Judgement comment: examiner was masked to IOL choice. It was not possible to identify IOL type during exams. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Judgement comment: within‐person study and no evidence of attrition biasestimation? |
| Selective reporting (reporting bias) | Low risk | Judgement comment: no evidence for selective outcome reporting |
| Other bias | Low risk | Judgement comment: no evidence of other sources of bias |
Sundelin 2005.
| Study characteristics | ||
| Methods |
Study design: parallel group study Eyes: senile cataract requiring surgery Cataract surgery: standardised surgical technique, two surgeons Postoperative treatment: topical dexamethasone 0.1% was prescribed 3 times a day for 3 weeks Indications for Nd:YAG capsulotomy: PCO present at slit lamp exam, if participant's perceived visual acuity had declined and visual acuity had deteriorated by at least two lines at the visual acuity exam |
|
| Participants |
Country: Sweden Number of participants (eyes) randomised: 116 (116) Inclusion criteria: age above 50 years, senile cataract requiring surgery, presume possibility of a 3 year follow‐up Exclusion criteria: intraocular pressure (IOP) of 25 mmHg or above in the study eye, glaucoma in either eye, corneal pathology in the study eye, a history of uveitis and pre‐operative dislocated lens or zonular rupture, participants with diabetes mellitus Average age (range): 72 years (51 to 90) % women: 58% |
|
| Interventions |
Intervention characteristics IOL with sharp optic edge design
IOL with round optic edge design
|
|
| Outcomes |
Primary outcome: PCO score (EPCO system) Secondary outcome: Nd:YAG capsulotomy proportion Follow‐up: 2 months, 1, 2 and 3 years after surgery |
|
| Identification |
Setting: Department of Ophthalmology, Moelndal’s Hospital, Moelndal, Sweden Author's name: Karin Sundelin, MD Institution: Department of Ophthalmology, Moelndal’s Hospital, Moelndal, Sweden Email: karin.sundelin@oft.gu.se Address: Department of Ophthalmology, Moelndal’s HospitalS‐431 80 Moelndal, Sweden Contact with the investigators: E‐mail contact to evaluate EPCO means and SD. According to corresponding author, KS's data not accessible any more |
|
| Notes |
Funding: supported in part by Pharmacia & Upjohn Sverige AB, the Gothenburg Medical Society and the Medical Faculty of the University of Gothenburg Date study conducted: the operations were all carried out in 2000 and in January 2001 Trial registration ID: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Quote: "The randomization was conducted in blocks to ensure that the IOLs were approximately equally distributed between the surgeons." |
| Allocation concealment (selection bias) | Unclear risk | Judgement comment: not reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Judgement comment: open‐label ‐ no masking |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Judgement comment: open‐label ‐ no masking |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Judgement comment: 116 participants enrolled, 107 (92%) followed to 1 year, 97 (84%) to 2 years and 83 (72%) followed to 3 years procession. |
| Selective reporting (reporting bias) | Low risk | Judgement comment: no evidence for selective outcome reporting |
| Other bias | Low risk | Judgement comment: no evidence for other sources of bias |
Wejde 2003.
| Study characteristics | ||
| Methods |
Study design: parallel group study Eyes: eyes with senile cataract who had uneventful phacoemulsification Cataract surgery: standardised technique, one surgeon Postoperative treatment: topical dexamethasone for 3 weeks in a tapered dose Indication for Nd:YAG capsulotomy: substantial PCO in the visual axis (diagnosed during slit lamp exam, participant describing relevant symptoms, BCDVA declined by at least 1 line |
|
| Participants |
Country: Sweden Number of participants (eyes) randomised: 180 (180) Inclusion criteria: senile cataract that had uneventful phacoemulsification Exclusion criteria: glaucoma, exfoliation syndrome, corneal pathology, a history of uveitis or intraocular surgery, preoperative oral steroid therapy, and diabetes mellitus. In addition, the participant could have no ocular pathology other than cataract and had to have a potential distance visual acuity of 20/40. Average age (range): 73 years (60 to 83) % women: 59% |
|
| Interventions |
Intervention characteristics IOL with sharp optic edge design ‐ Acrylic (59 participants, 59 eyes)
IOL with round optic edge design ‐ PMMA (61 participants, 61 eyes)
IOL with round optic edge design ‐ silicone (60 participants, 60 eyes)
|
|
| Outcomes |
Primary outcome: BCDVA Secondary outcome: YAG capsulotomy proportion, PCO score (POCOman computer‐analysis), position of the anterior capsulorhexis Follow‐up: 2 years after surgery (Wejde 2003) / 5 years after surgery(Rönbeck 2009)/ 12 years after surgery (Rönbeck 2014) |
|
| Identification |
Setting: St. Erik’s Eye Hospital, Stockholm, Sweden Comments: partial publication 5 years postoperatively Rönbeck 2014 Author's name: Gisela Wejde, MD Institution: St. Erik’s Eye Hospital, Stockholm, Sweden Email: gisela.wejde@ sankterik.se Address: St. Erik’s Eye Hospital, SE‐112 82 StockholmSweden Contact with investigators: none |
|
| Notes |
Funding: Financial support provided through the regional agreement on medical training and clinical research (ALF) between Stockholm County Council and the Karolinska Institute. Supported by grants from Stiftelsen Synfrämjandets Forskningsfond, Stockholm, Sweden Conflict of interest: none Date study conducted: surgery between May 1995 and March 1998 Trial registration ID: not available |
|
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Judgement comment: not reported |
| Allocation concealment (selection bias) | Unclear risk | Judgement comment: not reported |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Judgement comment: open‐label. No masking of participants, examiners, surgeons |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Judgement comment: open‐label. No masking of participants, examiners, surgeons |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Judgement comment: SD for PCO score and BCVA missing |
| Selective reporting (reporting bias) | Low risk | None |
| Other bias | Low risk | Judgement comment: no evidence for other source of bias |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Abela‐Formanek 2002 | Wrong study design (not randomised) |
| Alio 2016 | Wrong interventions (not IOL optic edge comparison) |
| Alio 2018 | Wrong interventions (not IOL optic edge comparison) |
| Auffarth 2003 | Wrong study design (prospective comparative observational case series ‐ not randomised) |
| Beltrame 2002 | Wrong study design (retrospective analysis) |
| Cardona 2018 | Wrong interventions (not IOL optic edge comparison) |
| Casprini 2002 | Wrong study design (retrospective analysis) |
| Choi 2018 | Follow‐up too short (6 months) |
| Cumming 1994 | Wrong study design |
| Gangwani 2011 | Wrong comparator (both square‐edged IOLs) |
| Gundersen 2017 | Wrong interventions (not IOL optic edge comparison) |
| Hancox 2007 | Wrong comparator (both squared optic edge) |
| Hirnschall 2013 | Wrong comparator (both sharp‐edged IOLs) |
| Hirnschall 2018 | Wrong interventions (not IOL optic edge comparison) |
| Iashinskas 2005 | Wrong study design |
| Iwase 2011 | Wrong comparator (both sharp‐edged IOLs) |
| Kahraman 2015 | Wrong comparator (both sharp‐edged IOLs) |
| Krajcova 2007 | No full‐text available |
| Kruger 2000 | Wrong study design (not randomised) |
| Kugelberg 2008 | Wrong comparator (both sharp‐edged IOLs) |
| Leydolt 2007 | Wrong indication (1‐piece vs. 3‐piece IOLs) |
| Leydolt 2013 | Wrong comparator (both sharp‐edged IOLs) |
| Leydolt 2017 | Wrong interventions (not IOL optic edge comparison) |
| Li 2008 | Wrong study design (meta‐analysis) |
| Miháltz 2018 | Wrong interventions (not IOL optic edge comparison) |
| Miyata 2007 | Wrong study design (not randomised) |
| Morgan‐Warren 2013 | Wrong study design (not randomised) |
| Nejima 2006 | Wrong intervention |
| Nishi 2007 | Wrong study design (not randomised) |
| Nixon 2004 | Wrong study design (not randomised) |
| Oner 2000 | Wrong study design (not randomised) |
| Prosdocimo 2003 | Wrong comparator (both IOLs sharp‐edged) |
| Sacu 2004a | PCO not measured |
| Sacu 2004b | PCO not measured |
| Sacu 2004c | Wrong study design |
| Sacu 2005a | Wrong study design (not RCT) |
| Schrecker 2018 | Wrong interventions (not IOL optic edge comparison) |
| Spyridaki 2010 | Wrong study design (not RCT‐ retrospective analysis) |
| Voronin 2017 | Wrong interventions (not IOL optic edge comparison) |
| Vyas 2007 | Wrong study design (not RCT) |
| Wejde 2004 | PCO not measured |
| Yang 2017 | Wrong interventions (not IOL optic edge comparison) |
| Yu 2016 | Wrong interventions (not IOL optic edge comparison) |
| Zemaitiene 2004 | Wrong study design (not randomised) |
| Zemaitiene 2007 | Wrong comparator (all sharp optic edges) |
| Zemaitiene 2011 | Wrong comparator (all sharp optic edges) |
| Zhang 2002 | No full‐text available |
Differences between protocol and review
Types of participants: We amended the inclusion criteria to include participants with ocular morbidity.
Unit of analysis: We planned to analyse within‐person studies as paired data but due to reporting difficulties this was not possible.
Contributions of authors
OF, SM and AH conceived the review question. SM and AH screened potential studies resulting from the searches, extracted data and assessed risk of bias for all studies.
SM and JE performed data analysis and interpretation of data. SM, AH and JE wrote drafts of the review, and responded to peer review comments and comments from the editorial base. OF reviewed the paper.
Sources of support
Internal sources
No sources of support provided
External sources
-
National Institute for Health Research (NIHR), UK
Richard Wormald, the former Co‐ordinating Editor for Cochrane Eyes and Vision (CEV) received financial support for his CEV research sessions from the Department of Health through the award made by the National Institute for Health Research to Moorfields Eye Hospital NHS Foundation Trust and UCL Institute of Ophthalmology for a Specialist Biomedical Research Centre for Ophthalmology.
Up to March 2021, this review was supported by the NIHR, via Cochrane Infrastructure funding to the CEV UK editorial base.
The views expressed in this publication are those of the authors and not necessarily those of the NIHR, NHS, or the Department of Health.
-
Public Health Agency, UK
As of April 2021, the completion of this review was supported by the HSC Research and Development (R&D) Division of the Public Health Agency which funds the Cochrane Eyes and Vision editorial base at Queen's University Belfast.
-
Queen's University Belfast, UK
Gianni Virgili, Co‐ordinating Editor for Cochrane Eyes and Vision’s work is funded by the Centre for Public Health, Queen’s University of Belfast, Northern Ireland
Declarations of interest
AH and JE: no financial interest in any of the products and methods mentioned.
SM has received grants, payment for lectures and meeting expenses from Alcon, Rayner and Johnson & Johnson Vision (formerly Abbot Medical Optics) but these payments were unrelated to the topic of the current review
OF is a scientific advisor to Alcon, Carl Zeiss Meditec AG, Croma Pharma, Johnson & Johnson and Merck.
OF is a trialist for three included studies ‐ Buehl 2005; Buehl 2007; Findl 2005 ‐ but did not take part in study selection, data extraction or assessment of risk of bias.
As per guidance from the Research Integrity Editor, Cochrane Eyes and Vision note that due to conflict of interest declarations for OF and SM for both the protocol and full review, this published review does not comply with the Cochrane Commercial Sponsorship Policy (2014) and an update of this review with an author team that complies with the policy will be required.
New
References
References to studies included in this review
Buehl 2005 {published data only}
- Buehl W, Findl O, Menapace R, Rainer G, Sacu S, Kiss B, et al. Effect of an acrylic intraocular lens with a sharp posterior optic edge on posterior capsule opacification. Journal of Cataract and Refractive Surgery 2002;28(7):1105-11. [DOI] [PubMed] [Google Scholar]
- Buehl W, Findl O, Menapace R, Sacu S, Kriechbaum K, Koeppl C, et al. Long-term effect of optic edge design in an acrylic intraocular lens on posterior capsule opacification. Journal of Cataract and Refractive Surgery 2005;31(5):954-61. [DOI] [PubMed] [Google Scholar]
Buehl 2007 {published data only}
- Buehl W, Menapace R, Findl O, Neumayer T, Bolz M, Prinz A. Long-term effect of optic edge design in a silicone intraocular lens on posterior capsule opacification. American Journal of Ophthalmology 2007;143(6):913-9. [DOI] [PubMed] [Google Scholar]
- Buehl W, Menapace R, Sacu S, Kriechbaum K, Koeppl C, Wirtitsch M, et al. Effect of a silicone intraocular lens with a sharp posterior optic edge on posterior capsule opacification. Journal of Cataract and Refractive Surgery 2004;30(8):1661-7. [DOI] [PubMed] [Google Scholar]
Findl 2005 {published data only}
- Findl O, Buehl W, Menapace R, Sacu S, Georgopoulos M, Rainer G. Long-term effect of sharp optic edges of a polymethyl methacrylate intraocular lens on posterior capsule opacification: a randomized trial. Ophthalmology 2005;112(11):2004-8. [DOI] [PubMed] [Google Scholar]
Haripriya 2017 {published data only}
- Haripriya A, Chang DF, Vijayakumar B, Niraj A, Shekhar M, Tanpreet S, et al. Long-term posterior capsule opacification reduction with square-edge polymethylmethacrylate intraocular lens: randomized controlled study. Ophthalmology 2017;124(3):295-302. [DOI] [PubMed] [Google Scholar]
Hayashi 2005 {published data only}
- Hayashi K, Hayashi H. Posterior capsule opacification in the presence of an intraocular lens with a sharp versus rounded optic edge. Ophthalmology 2005;112(9):1550-6. [DOI] [PubMed] [Google Scholar]
Mester 2004 {published data only}
- Kohnen T, Fabian E, Gerl R, Hunold W, Hutz W, Strobel J, et al. Optic edge design as long-term factor for posterior capsular opacification rates. Ophthalmology 2008;115(8):1308-14. [DOI] [PubMed] [Google Scholar]
- Mester U, Fabian E, Gerl R, Hunold W, Hutz W, Strobel J, et al. Posterior capsule opacification after implantation of CeeOn Edge 911A, PhacoFlex SI-40NB, and AcrySof MA60BM lenses: one-year results of an intraindividual comparison multicenter study. Journal of Cataract and Refractive Surgery 2004;30(5):978-85. [DOI] [PubMed] [Google Scholar]
Sacu 2005 {published data only}
- Sacu S, Menapace R, Findl O, Kiss B, Buehl W, Georgopoulos M. Long-term efficacy of adding a sharp posterior optic edge to a three-piece silicone intraocular lens on capsule opacification: five-year results of a randomized study. American Journal of Ophthalmology 2005;139(4):696-703. [DOI] [PubMed] [Google Scholar]
Shah 2007 {published data only}
- Shah A, Spalton DJ, Gilbert C, Vasavada A, Boyce JF, Minassian D, et al. Effect of intraocular lens edge profile on posterior capsule opacification after extracapsular cataract surgery in a developing country. Journal of Cataract and Refractive Surgery 2007;33(7):1259-66. [DOI] [PubMed] [Google Scholar]
Sundelin 2005 {published data only}
- Sundelin K, Shams H, Stenevi U. Three-year follow-up of posterior capsule opacification with two different silicone intraocular lenses. Acta Ophthalmologica Scandinavica 2005;83(1):11-9. [DOI] [PubMed] [Google Scholar]
Wejde 2003 {published data only}
- Ronbeck M, Kugelberg M. Posterior capsule opacification with 3 intraocular lenses: 12-year prospective study. Journal of Cataract and Refractive Surgery 2014;40(1):70-6. [DOI] [PubMed] [Google Scholar]
- Ronbeck M, Zetterstrom C, Wejde G, Kugelberg M. Comparison of posterior capsule opacification development with 3 intraocular lens types: five-year prospective study. Journal of Cataract and Refractive Surgery 2009;35(11):1935-40. [DOI] [PubMed] [Google Scholar]
- Wejde G, Kugelberg M, Zetterstrom C. Posterior capsule opacification: comparison of 3 intraocular lenses of different materials and design. Journal of Cataract and Refractive Surgery 2003;29(8):1556-9. [DOI] [PubMed] [Google Scholar]
References to studies excluded from this review
Abela‐Formanek 2002 {published data only}
- Abela-Formanek C, Amon M, Schauersberger J, Schild G, Kolodjaschna J, Barisani-Asenbauer, et al. Uveal and capsular biocompatibility of 2 foldable acrylic intraocular lenses in patients with uveitis or pseudoexfoliation syndrome: comparison to a control group. Journal of Cataract and Refractive Surgery 2002;28(7):1160-72. [DOI] [PubMed] [Google Scholar]
Alio 2016 {published data only}
- Alio JL, Simonov A, Plaza-Puche AB, Angelov A, Angelov Y, Van Lawick W, et al. Visual outcomes and accommodative response of the Lumina accommodative intraocular lens. American Journal of Ophthalmology 2016;164:37-48. [DOI] [PubMed] [Google Scholar]
Alio 2018 {published data only}
- Alio J, Simonov AN, Romero D, Angelov A, Angelov Y, Van Lawick W, et al. Analysis of accommodative performance of a new accommodative intraocular lens. Journal of Refractive Surgery 2018;34(2):78‐83. [DOI] [PubMed] [Google Scholar]
Auffarth 2003 {published data only}
- Auffarth GU, Golescu A, Becker KA, Volcker HE. Quantification of posterior capsule opacification with round and sharp edge intraocular lenses. Ophthalmology 2003;110(4):772-80. [DOI] [PubMed] [Google Scholar]
Beltrame 2002 {published data only}
- Beltrame G, Salvetat ML, Chizzolini M, Driussi GB, Busatto P, Di Giorgio G, et al. Posterior capsule opacification and Nd:YAG capsulotomy rates after implantation of silicone, hydrogel and soft acrylic intraocular lenses: a two-year follow-up study. European Journal of Ophthalmology 2002;12(5):388-94. [DOI] [PubMed] [Google Scholar]
Cardona 2018 {published data only}
- Cardona G, Vega F, Gil MA, Varón C, Buil JA, Millán MS. Visual acuity and image quality in 5 diffractive intraocular lenses. European Journal of Ophthalmology 2018;28(1):36-41. [DOI] [PubMed] [Google Scholar]
Casprini 2002 {published data only}
- Casprini F, Tosi GM, Quercioli PP, Caporossi A. Comparison of AcrySof MA30BA and Sensar AR40 acrylic intraocular lenses. Journal of Cataract and Refractive Surgery 2002;28(7):1130-4. [DOI] [PubMed] [Google Scholar]
Choi 2018 {published data only}76566080
- Choi M, Lazo MZ, Kang M, Lee J, Joo CK. Effect of number and position of intraocular lens haptics on anterior capsule contraction: a randomized, prospective trial. BMC Ophthalmology 2018;1:78. [DOI] [PMC free article] [PubMed] [Google Scholar]
Cumming 1994 {published data only}
- Cumming JS. Capsulotomy rate in silicon plate haptic and prolene loop IOLs. European Journal of Implant and Refractive Surgery 1994;6(4):200-4. [Google Scholar]
Gangwani 2011 {published data only}
- Gangwani V, Hirnschall N, Koshy J, Crnej A, Nishi Y, Maurino V, et al. Posterior capsule opacification and capsular bag performance of a microincision intraocular lens. Journal of Cataract and Refractive Surgery 2011;37(11):1988-92. [DOI] [PubMed] [Google Scholar]
Gundersen 2017 {published data only}
- Gundersen KG, Potvin R. Trifocal intraocular lenses: A comparison of the visual performance and quality of vision provided by two different lens designs. Clinical Ophthalmology 2017;11:1081-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Hancox 2007 {published data only}
- Hancox J, Spalton D, Heatley C, Jayaram H, Yip J, Boyce J, et al. Fellow-eye comparison of posterior capsule opacification rates after implantation of 1CU accommodating and AcrySof MA30 monofocal intraocular lenses. Journal of Cataract and Refractive Surgery 2007;33(3):413-7. [DOI] [PubMed] [Google Scholar]
Hirnschall 2013 {published data only}
- Hirnschall N, Nishi Y, Crnej A, Koshy J, Gangwani V, Maurino V, et al. Capsular bag stability and posterior capsule opacification of a plate-haptic design microincision cataract surgery intraocular lens: 3-year results of a randomised trial. British Journal of Ophthalmology 2013;97(12):1565-8. [DOI] [PubMed] [Google Scholar]
Hirnschall 2018 {published data only}
- Hirnschall N, Farrokhi S, Amir-Asgari S, Hienert J, Findl O. Intraoperative optical coherence tomography measurements of aphakic eyes to predict postoperative position of 2 intraocular lens designs. Journal of Cataract and Refractive Surgery 2018;44(11):1310-6. [DOI] [PubMed] [Google Scholar]
Iashinskas 2005 {published data only}
- Iashinskas VP, Zhemaĭtene R, Barzzhiukas V. Secondary cataract and IOL type and the shape of its optic part. Vestnik Oftalmologii 2005;121(1):13-6. [PubMed] [Google Scholar]
Iwase 2011 {published data only}
- Iwase T, Nishi Y, Oveson BC, Jo YJ. Hydrophobic versus double-square-edged hydrophilic foldable acrylic intraocular lens: effect on posterior capsule opacification. Journal of Cataract and Refractive Surgery 2011;37(6):1060-8. [DOI] [PubMed] [Google Scholar]
Kahraman 2015 {published data only}
- Kahraman G, Amon M, Ferdinaro C, Nigl K, Walch M. Intraindividual comparative analysis of capsule opacification after implantation of 2 single-piece hydrophobic acrylic intraocular lenses models: three-year follow-up. Journal of Cataract and Refractive Surgery 2015;41(5):990-6. [DOI] [PubMed] [Google Scholar]
Krajcova 2007 {published data only}
- Krajcová P, Chynoranský M, Strmen P. Posterior capsule opacification (PCO) following implantation of various types of IOLs--part one: the uncomplicated course. Ceska a Slovenska Oftalmologie 2007;63(6):379-89. [PubMed] [Google Scholar]
Kruger 2000 {published data only}
- Kruger AJ, Schauersberger J, Abela C, Schild G, Amon M. Two year results: sharp versus rounded optic edges on silicone lenses. Journal of Cataract and Refractive Surgery 2000;26(4):566-70. [DOI] [PubMed] [Google Scholar]
Kugelberg 2008 {published data only}
- Kugelberg M, Wejde G, Jayaram H, Zetterström C. Two-year follow-up of posterior capsule opacification after implantation of a hydrophilic or hydrophobic acrylic intraocular lens. Acta Ophthalmologica 2008;86(5):533-6. [DOI] [PubMed] [Google Scholar]
Leydolt 2007 {published data only}
- Leydolt C, Davidovic S, Sacu S, Menapace R, Neumayer T, Prinz A, et al. Long-term effect of 1-piece and 3-piece hydrophobic acrylic intraocular lens on posterior capsule opacification: a randomized trial. Ophthalmology 2007;114(9):1663-9. [DOI] [PubMed] [Google Scholar]
Leydolt 2013 {published data only}
- Leydolt C, Kriechbaum K, Schriefl S, Pachala M, Menapace R. Posterior capsule opacification and neodymium:YAG rates with 2 single-piece hydrophobic acrylic intraocular lenses: three-year results. Journal of Cataract and Refractive Surgery 2013;39(12):1886-92. [DOI] [PubMed] [Google Scholar]
Leydolt 2017 {published data only}
- Leydolt C, Schartmüller D, Schwarzenbacher L, Schranz M, Schriefl S, Menapace R. Comparison of posterior capsule opacification development with 2 single-piece intraocular lens types. Journal of Cataract and Refractive Surgery 2017;43(6):774-80. [DOI] [PubMed] [Google Scholar]
Li 2008 {published data only}
- Li N, Chen X, Zhang J, Zhou Y, Yao X, Du L, et al. Effect of AcrySof versus silicone or polymethyl methacrylate intraocular lens on posterior capsule opacification. Ophthalmology 2008;115(5):830-8. [DOI] [PubMed] [Google Scholar]
Miháltz 2018 {published data only}
- Miháltz K, Lasta M, Burgmüller M, Vécsei-Marlovits PV, Weingessel B. Comparison of two toric IOLs with different haptic design: optical quality after 1 year. Journal of Ophthalmology 2018;11:Article ID 4064369. [DOI] [PMC free article] [PubMed] [Google Scholar]
Miyata 2007 {published data only}
- Miyata K, Kato S, Nejima R, Miyai T, Honbo M, Ohtani S. Influences of optic edge design on posterior capsule opacification and anterior capsule contraction. Acta Ophthalmologica Scandinavica 2007;85(1):99-102. [DOI] [PubMed] [Google Scholar]
Morgan‐Warren 2013 {published data only}
- Morgan-Warren PJ, Smith JA. Intraocular lens-edge design and material factors contributing to posterior-capsulotomy rates: comparing Hoya FY60aD, PY60aD, and AcrySof SN60WF. Clinical Ophthalmology 2013;7:1661-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Nejima 2006 {published data only}
- Nejima R, Miyai T, Kataoka Y, Miyata K, Honbou M, Tokunaga T, et al. Prospective intrapatient comparison of 6.0-millimeter optic single-piece and 3-piece hydrophobic acrylic foldable intraocular lenses. Ophthalmology 2006;113(4):585-90. [DOI] [PubMed] [Google Scholar]
Nishi 2007 {published data only}
- Nishi Y, Rabsilber TM, Limberger IJ, Reuland AJ, Auffarth GU. Influence of 360-degree enhanced optic edge design of a hydrophilic acrylic intraocular lens on posterior capsule opacification. Journal of Cataract and Refractive Surgery 2007;33(2):227-31. [DOI] [PubMed] [Google Scholar]
Nixon 2004 {published data only}
- Nixon DR. In vivo digital imaging of the square-edged barrier effect of a silicone intraocular lens. Journal of Cataract and Refractive Surgery 2004;30(12):2574-84. [DOI] [PubMed] [Google Scholar]
Oner 2000 {published data only}
- Oner FH, Gunenc U, Ferliel ST. Posterior capsule opacification after phacoemulsification: foldable acrylic versus poly(methyl methacrylate) intraocular lenses. Journal of Cataract and Refractive Surgery 2000;26(5):722-6. [DOI] [PubMed] [Google Scholar]
Prosdocimo 2003 {published data only}
- Prosdocimo G, Tassinari G, Sala M, Di Biase A, Toschi PG, Gismondi M, et al. Posterior capsule opacification after phacoemulsification: silicone CeeOn Edge versus acrylate AcrySof intraocular lens. Journal of Cataract and Refractive Surgery 2003;29(8):1551-5. [DOI] [PubMed] [Google Scholar]
Sacu 2004a {published data only}
- Sacu S, Menapace R, Findl O, Georgopoulos M, Buehl W, Kriechbaum K, et al. Influence of optic edge design and anterior capsule polishing on posterior capsule fibrosis. Journal of Cataract and Refractive Surgery 2004;30(3):658-62. [DOI] [PubMed] [Google Scholar]
Sacu 2004b {published data only}
- Sacu S, Menapace R, Buehl W, Rainer G, Findl O. Effect of intraocular lens optic edge design and material on fibrotic capsule opacification and capsulorhexis contraction. Journal of Cataract and Refractive Surgery 2004;30(9):1875-82. [DOI] [PubMed] [Google Scholar]
Sacu 2004c {published data only}
- Sacu S, Findl O, Menapace R, Buehl W, Wirtitsch M. Comparison of posterior capsule opacification between the 1-piece and 3-piece Acrysof intraocular lenses: two-year results of a randomized trial. Ophthalmology 2004;111(10):1840-6. [DOI] [PubMed] [Google Scholar]
Sacu 2005a {published data only}
- Sacu S, Findl O, Menapace R, Buehl W. Influence of optic edge design, optic material, and haptic design on capsular bend configuration. Journal of Cataract and Refractive Surgery 2005;31(10):1888-94. [DOI] [PubMed] [Google Scholar]
Schrecker 2018 {published data only}
- Schrecker J, Langenbucher A, Seitz B, Eppig T. First results with a new intraocular lens design for the individual correction of spherical aberration. Journal of Cataract and Refractive Surgery 2018;44(10):1211-9. [DOI] [PubMed] [Google Scholar]
Spyridaki 2010 {published data only}
- Spyridaki M, Höh H. Comparison of four MICS intraocular lenses regarding their rates of neodymium:YAG laser capsulotomy. Klinische Monatsblatter fur Augenheilkunde 2010;227(3):208-14. [DOI] [PubMed] [Google Scholar]
Voronin 2017 {published data only}
- Voronin GV, Mamikonyan VR, Sheludchenko VM, Narbut MN. Clinical results of aphakia correction with multifocal intraocular lenses. Vestnik Oftalmologii 2017;133(1):37-41. [DOI] [PubMed] [Google Scholar]
Vyas 2007 {published data only}
- Vyas AV, Narendran R, Bacon PJ, Apple DJ. Three-hundred-sixty degree barrier effect of a square-edged and an enhanced-edge intraocular lens on centripetal lens epithelial cell migration: two-year results. Journal of Cataract and Refractive Surgery 2007;33(1):81-7. [DOI] [PubMed] [Google Scholar]
Wejde 2004 {published data only}
- Wejde G, Kugelberg M, Zetterström C. Position of anterior capsulorhexis and posterior capsule opacification. Acta Ophthalmologica Scandinavica 2004;82(5):531-4. [DOI] [PubMed] [Google Scholar]
Yang 2017 {published data only}
- Yang S, Lim SA, Na KS, Joo CK. Comparison of anterior capsule stability following implantation of three single piece acrylic intraocular lenses with different haptic design. Korean Journal of Ophthalmology 2017;31(1):32-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Yu 2016 {published data only}
- Yu P. Safety and efficacy of implantation of two multifocal intraocular lenses in the treatment of senile cataract: Study protocol for a randomized controlled clinical trial.. Chinese Journal of Tissue Engineering Research 2017;20:7139-44. [Google Scholar]
Zemaitiene 2004 {published data only}
- Zemaitiene R, Jasinskas V, Barzdziukas V, Auffarth GU. Prevention of posterior capsule opacification using different intraocular lenses (results of one-year clinical study). Medicina 2004;40(8):721-30. [PubMed] [Google Scholar]
Zemaitiene 2007 {published data only}
- Zemaitiene R, Jasinskas V, Auffarth GU. Influence of three-piece and single-piece designs of two sharp-edge optic hydrophobic acrylic intraocular lenses on the prevention of posterior capsule opacification: a prospective, randomised, long-term clinical trial. British Journal of Ophthalmology 2007;91(5):644-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Zemaitiene 2011 {published data only}
- Zemaitiene R, Jasinskas, V. Prevention of posterior capsule opacification with 3 intraocular lens models: a prospective, randomized, long-term clinical trial. Medicina 2011;47(11):595-9. [PubMed] [Google Scholar]
Zhang 2002 {published data only}
- Zhang Z. A clinical study of posterior capsular opacification after implantation of foldable intraocular lenses with different edges of optics. Chinese Journal of Ophthalmology 2002;38(10):606-9. [PubMed] [Google Scholar]
Additional references
Ang 2014
- Ang M, Evans JR, Mehta JS. Manual small incision cataract surgery (MSICS) with posterior chamber intraocular lens versus extracapsular cataract extraction (ECCE) with posterior chamber intraocular lens for age-related cataract. Cochrane Database of Systematic Reviews 2014, Issue 11. Art. No: CD008811. [DOI: 10.1002/14651858.CD008811.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Apple 1992
- Apple DJ, Solomon KD, Tetz MR, Assia EI, Holland EY, Legler UF, et al. Posterior capsule opacification. Survey of Ophthalmology 1992;37(2):73-116. [DOI] [PubMed] [Google Scholar]
Auffarth 2004
- Auffarth GU, Brezin A, Caporossi A, Lafuma A, Mendicute J, Berdeaux G, et al. Comparison of Nd:YAG capsulotomy rates following phacoemulsification with implantation of PMMA, silicone, or acrylic intra-ocular lenses in four European countries. Ophthalmic Epidemiology 2004;11(4):319-29. [DOI] [PubMed] [Google Scholar]
Cheng 2007
- Cheng JW, Wei RL, Cai JP, Xi GL, Zhu H, Li Y, et al. Efficacy of different intraocular lens materials and optic edge designs in preventing posterior capsular opacification: a meta-analysis. American Journal of Ophthalmology 2007;143(3):428-36. [DOI] [PubMed] [Google Scholar]
Covidence [Computer program]
- Veritas Health Innovation Covidence. Version accessed 18 November 2020. Melbourne, Australia: Veritas Health Innovation. Available at covidence.org.
Deeks 2017
- Deeks JJ, Higgins JP, Altman DG, (editors), on behalf of the Cochrane Statistical Methods Group. Chapter 9: Analysing data and undertaking meta-analyses. In: Higgins JP, Churchill R, Chandler J, Cumpston MS (editors), Cochrane Handbook for Systematic Reviews of Interventions version 5.2.0 (updated June 2017), Cochrane, 2017. Available from www.training.cochrane.org/handbook.
de Silva 2014
- Silva SR, Riaz Y, Evans JR. Phacoemulsification with posterior chamber intraocular lens versus extracapsular cataract extraction (ECCE) with posterior chamber intraocular lens for age‐related cataract. Cochrane Database of Systematic Reviews 2014, Issue 1. Art. No: CD008812. [DOI: 10.1002/14651858.CD008812.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Findl 2003
- Findl O, Buehl W, Menapace R, Georgopoulos M, Rainer G, Siegl H, et al. Comparison of 4 methods for quantifying posterior capsule opacification. Journal of Cataract and Refractive Surgery 2003;29(1):106-11. [DOI] [PubMed] [Google Scholar]
Glanville 2006
- Glanville JM, Lefebvre C, Miles JN, Camosso-Stefinovic J. How to identify randomized controlled trials in MEDLINE: ten years on. Journal of the Medical Library Association 2006;94(2):130-6. [PMC free article] [PubMed] [Google Scholar]
GRADEpro GDT [Computer program]
- McMaster University (developed by Evidence Prime) GRADEpro GDT. Version accessed 21 January 2021. Hamilton (ON): McMaster University (developed by Evidence Prime). Available at gradepro.org.
Higgins 2017
- Higgins JP, Altman DG, Sterne JA (editors). Chapter 8: Assessing risk of bias in included studies. In: Higgins JP, Churchill R, Chandler J, Cumpston MS (editors), Cochrane Handbook for Systematic Reviews of Interventions version 5.2.0 (updated June 2017), Cochrane, 2017. Available from www.training.cochrane.org/handbook.
Holladay 2012
- Holladay JT, Zhao H, Reisin CR. Negative dysphotopsia: the enigmatic penumbra. Journal of Cataract and Refractive Surgery 2012;38(7):1251–65. [DOI] [PubMed] [Google Scholar]
Hollick 1999
- Hollick EJ, Spalton DJ, Meacock WR. The effect of capsulorhexis size on posterior capsular opacification: one-year results of a randomized prospective trial. American Journal of Ophthalmology 1999;128(3):271-9. [DOI] [PubMed] [Google Scholar]
Javitt 1992
- Javitt JC, Tielsch JM, Canner JK, Kolb MM, Sommer A, Steinberg EP, et al. National outcomes of cataract extraction: increased risk of retinal complications associated with Nd:YAG laser capsulotomy. Ophthalmology 1992;99(10):1487-98. [DOI] [PubMed] [Google Scholar]
Khalifa 1992
- Khalifa MA. Polishing the posterior capsule after extracapsular extraction of senile cataract. Journal of Cataract and Refractive Surgery 1992;18(2):170-3. [DOI] [PubMed] [Google Scholar]
Khanna 2011
- Khanna R, Pujari S, Sangwan V. Cataract surgery in developing countries. Current Opinion in Ophthalmology 2011;22(1):10-4. [DOI] [PubMed] [Google Scholar]
Kinard 2013
- Kinard K, Jarstad A, Olson RJ. Correlation of visual quality with satisfaction and function in a normal cohort of pseudophakic patients. Journal of Cataract and Refractive Surgery 2013;39(4):590-7. [DOI] [PubMed] [Google Scholar]
Kirkham 2010
- Kirkham JJ, Dwan KM, Altman DG, Gamble C, Dodd S, Smyth R, et al. The impact of outcome reporting bias in randomised controlled trials on a cohort of systematic reviews. BMJ 2010;340:c365. [DOI] [PubMed] [Google Scholar]
Kohnen 2008
- Kohnen T, Fabian E, Gerl R, Hunold W, Hutz W, Strobel J, et al. Optic edge design as long-term factor for posterior capsular opacification rates. Delete Edit. Ophthalmology 2008;115(8):1308-14. [DOI] [PubMed] [Google Scholar]
Li 2013
- Li Y, Wang J, Chen Z, Tang X. Effect of hydrophobic acrylic versus hydrophilic acrylic intraocular lens on posterior capsule opacification: meta-analysis. PLoS One 2013;8(11):e77864. [DOI] [PMC free article] [PubMed] [Google Scholar]
Masket 1993
- Masket S, Geraghty E, Crandall AS, Davison JA, Johnson SH, Koch DD, et al. Undesired light images associated with ovoid intraocular lenses. Journal of Cataract and Refractive Surgery 1993;19(6):690-4. [DOI] [PubMed] [Google Scholar]
Masket 2020
- Masket S, Fram NR. Pseudophakic dysphotopsia: review of incidence, cause, and treatment of positive and negative dysphotopsia. Ophthalmology 2020;12:S0161-6420(20)30787-9. [DOI] [PubMed] [Google Scholar]
Nanavety 2008
- Nanavaty MA, Spalton DJ, Boyce J, Brain A, Marshall J. Edge profile of commercially available square-edged intraocular lenses. Journal of Cataract and Refractive Surgery 2008;34(4):677-86. [DOI] [PubMed] [Google Scholar]
Nanavety 2019
- Nanavaty MA, Zukaite I, Salvage J. Edge profile of commercially available square-edged intraocular lenses: part 2. Journal of Cataract and Refractive Surgery 2019;45(6):847-53. [DOI] [PubMed] [Google Scholar]
Nishi 1998
- Nishi O, Nishi K, Sakanishi K. Inhibition of migrating lens epithelial cells at the capsular bend created by the rectangular optic edge of a posterior chamber intraocular lens. Ophthalmic Surgery and Lasers 1998;29(7):587-94. [PubMed] [Google Scholar]
Nishi 2004
- Nishi O, Nishi K, Osakabe Y. Effect of intraocular lenses on preventing posterior capsule opacification: design versus material. Journal of Cataract and Refractive Surgery 2004;30(10):2170-6. [DOI] [PubMed] [Google Scholar]
Radmall 2015
- Radmall BR, Floyd A, Oakey Z, Olson RJ. Refractive index and its impact on pseudophakic dysphotopsia. Clinical Ophthalmology 2015;9:1353-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Review Manager 2014 [Computer program]
- Nordic Cochrane Centre, The Cochrane Collaboration Review Manager 5 (RevMan 5). Version 5.4. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2020.
Riaz 2006
- Riaz Y, Mehta JS, Wormald R, Evans JR, Foster A, Ravilla T, et al. Surgical interventions for age-related cataract. Cochrane Database of Systematic Reviews 2006, Issue 4. Art. No: CD001323. [DOI: 10.1002/14651858.CD001323.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Riaz 2013
- Riaz Y, Silva SR, Evans JR. Manual small incision cataract surgery (MSICS) with posterior chamber intraocular lens versus phacoemulsification with posterior chamber intraocular lens for age-related cataract. Cochrane Database of Systematic Reviews 2013, Issue 10. Art. No: CD008813. [DOI: 10.1002/14651858.CD008813.pub2] [DOI] [PMC free article] [PubMed] [Google Scholar]
Schaumberg 1998
- Schaumberg DA, Dana MR, Christen WG, Glynn RJ. A systematic overview of the incidence of posterior capsule opacification. Ophthalmology 1998;105(7):1213-21. [DOI] [PubMed] [Google Scholar]
Schwiegerling 2016
- Schwiegerling J. Recent developments in pseudophakic dysphotopsia. Current Opinion in Ophthalmology 2016;17(1):27-30. [DOI] [PubMed] [Google Scholar]
Sterne 2011
- Sterne JA, Egger M, Moher D (editors). Chapter 10: Addressing reporting biases. In: Higgins JP, Green S (editors). Cochrane Handbook for Systematic Reviews of Intervention version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from www.handbook.cochrane.org.
Thom 2019
- Thom H, Ender F, Samavedam S, Perez Vivez C, Gupta S, Dhariwal M, et al. Effect of AcrySof versus other intraocular lens properties on the risk of Nd:YAG capsulotomy after cataract surgery: a systematic literature review and network meta-analysis. Plos ONE 2019;14(8):e0220498. [DOI] [PMC free article] [PubMed] [Google Scholar]
Walker 2008
- Walker TD. Pharmacological attempts to reduce posterior capsule opacification after cataract surgery-a review. Clinical and Experimental Ophthalmology 2008;36(9):883-90. [DOI] [PubMed] [Google Scholar]
Wormstone 2002
- Wormstone IM. Posterior capsule opacification: a cell biological perspective. Experimental Eye Research 2002;74(3):337-47. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Findl 2010
- Findl O, Buehl W, Bauer P, Sycha T. Interventions for preventing posterior capsule opacification. Cochrane Database of Systematic Reviews 2010, Issue 2. Art. No: CD003738. [DOI: 10.1002/14651858.CD003738.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Maedel 2017
- Maedel S, Buehl W, Findl O. Intraocular lens optic edge design for the prevention of posterior capsule opacification after cataract surgery. Cochrane Database of Systematic Reviews 2017, Issue 1. Art. No: CD012516. [DOI: 10.1002/14651858.CD012516] [DOI] [PMC free article] [PubMed] [Google Scholar]


