Abstract
Transforming growth factor-beta2 (TGF-β2) is recognized as a versatile cytokine that plays a vital role in regulation of joint development, homeostasis, and diseases, but its role as a biological mechanism is understood far less than that of its counterpart, TGF-β1. Cartilage as a load-resisting structure in vertebrates however displays a fragile performance when any tissue disturbance occurs, due to its lack of blood vessels, nerves, and lymphatics. Recent reports have indicated that TGF-β2 is involved in the physiological processes of chondrocytes such as proliferation, differentiation, migration, and apoptosis, and the pathological progress of cartilage such as osteoarthritis (OA) and rheumatoid arthritis (RA). TGF-β2 also shows its potent capacity in the repair of cartilage defects by recruiting autologous mesenchymal stem cells and promoting secretion of other growth factor clusters. In addition, some pioneering studies have already considered it as a potential target in the treatment of OA and RA. This article aims to summarize the current progress of TGF-β2 in cartilage development and diseases, which might provide new cues for remodelling of cartilage defect and intervention of cartilage diseases.
Keywords: TGF-β2, Chondrocyte, Cartilage development and diseases
Keywords: cartilage tissue, chondrocytes, cartilage diseases, apoptosis, growth factors, mesenchymal progenitor cells, cartilage defects, cytokines, osteoarthritis (OA), blood vessels
Article focus
The importance of transforming growth factor-beta2 (TGF-β2) in the physiological activities and pathological states of cartilage has not been fully understood. This article summarizes the current progress of TGF-β2 in cartilage development and diseases, which provides us with a total understanding of TGF-β2, particularly with regards to cartilage defects, degenerative changes, and targeted interventions.
Key messages
The basal role of TGF-β2 in chondrocyte life cycle is summarized in this study.
The effect of TGF-β2 on cartilage diseases including osteoarthritis and rheumatoid arthritis is reviewed based on current progress.
Strengths and limitations
TGF-β2 could regulate chondrocyte behaviours including proliferation, migration, differentiation, and cell death, which makes it a potential therapeutic target in gene therapy, tissue engineering, and stem cell therapy.
The lack of understanding of the inner biomechanism of TGF-β2 limits its potential applications in both experimental and clinical trials.
Introduction
In recent decades, an increasing number of studies have been conducted on the role of the transforming growth factor-beta (TGF-β) superfamily in physiological and pathological processes of cartilage, especially TGF-β1. However, few studies focus on the role of TGF-β2 in cartilage development and diseases. One study has revealed that TGF-β2 also plays an important role in maintaining homeostasis of cartilage and pathological progress of cartilage.1 TGF-β2 is known to be a member of the highly conserved TGF-β superfamily, which consists of at least 40 ligand proteins. Moreover, TGF-β2 has two homologous dimeric isoforms: TGF-β1 and TGF-β3, although TGF-β isoforms represent similar structures and different functions.2 TGF-β2 is a pleiotropic cytokine with important roles in embryonic development and tissue homeostasis.3 In addition, TGF-β2 down-regulated osteogenesis when intermittent pressure was applied to the periodontal ligament cells, meaning TGF-β2 exacted its biological functions depending on ambient conditions.4 To date, it is known that TGF-β2 participates in the regulation of cell development, wound healing, immune responses, inflammation, and cancer metastasis.5-9
Articular cartilage is a highly specialized connective tissue that lives in an avascular, aneural, and hypoxic environment.10 This connective tissue consists of chondrocytes and extracellular cartilage matrix. Cartilage matrix contains two major organic substances, type II collagen and proteoglycan.11 Since cartilage tissue lacks blood supply and is a single-cell tissue, the repair ability of articular cartilage is poor.12 Consequently, it is very important to understand the mechanisms of cartilage defects and explore new potential therapeutic targets in articular cartilage diseases by researching signalling pathways of various cytokines in cartilage. Another study has suggested that TGF-β2 could activate not only canonical drosophila mothers against decapentaplegic protein (Smad) signalling, but also activate noncanonical generic mitogen-activated protein kinases (MAPK) signalling in chondrocytes.13 For instance, TGF-β2 could induce redifferentiation of chondrocytes by activating activin receptor-like kinase 5 (ALK5)/Smad3 signalling under hypoxic conditions.14,15 Therefore, TGF-β2 may be a potential therapeutic target for regulating chondrocyte activity and changing the physiological progress of articular cartilage diseases.
Recapitulation
Transforming growth factor-beta2
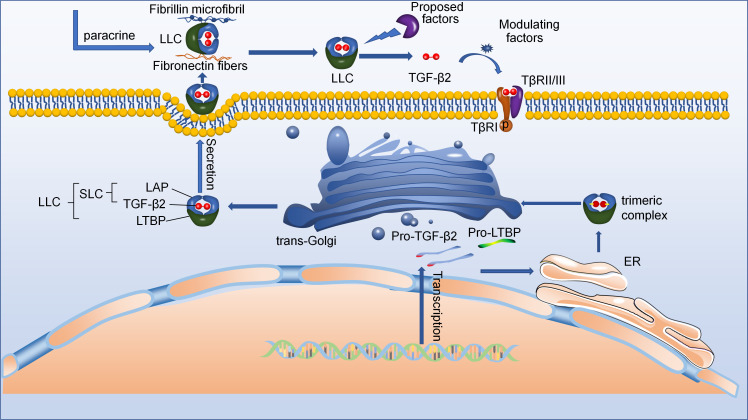
TGF-β2, a member of the highly conserved TGF-β superfamily, including TGF-betas, activins, and nodal and bone morphogenetic proteins (BMPs), is an indispensable cytokine that can regulate the function of many types of cells, and is involved in the pathological initiation and progression of various diseases.16-20 TGF-β2, also known as BSC-1 cell (African green monkey kidney epithelial cell) growth inhibitor or polyergin, was first found following the cloning of TGF-β1 in 1985, and another mammalian isoform, TGF-β3, was also found.21 These TGF-β subtypes exhibit spatial and temporal differences in gene expression, which regulate development of cells and exert profibrotic and anti-inflammatory effects. TGF-β2 is a dimeric protein that consists of two identical disulfide-linked monomers, and its gene has been localized in humans to chromosome 1q41.22,23 Moreover, the α-helix in TGF-β2 is linked to the remainder of the monomer by an additional disulfide bridge, showing great difference of the structure from the other members of the TGF-β superfamily.24 Despite these, TGF-β isoforms have many similar peculiarities due to 72% to 80% similarity in the sequence of the active 25 kDa forms. Secreted TGF-β2 is inactive as a high molecular weight latent complex in most cells.25 Therefore, researching activation mechanisms of latent TGF-β2 is highly important because latent TGF-β also regulates TGF-β biological behaviour.26 Messenger RNA (mRNA) of TGF-β2 is transferred from the nucleus into the cytoplasm (Figure 1).27 mRNA of TGF-β2 is translated as pro-protein including TGF-β2 and latency-associated peptide (LAP).28 Then this pro-protein folds and dimer in the endoplasmic reticulum (ER) with latent TGF-β2-binding protein (LTBP) to form trimeric complex, which is linked to a signal LTBP by a pair of disulfide bonds between LTBP and LAP.29 The LAP is cleaved in Golgi apparatus by a furin-like convertase, forming the large latent complex (LLC) comprising of TGF-β2, LAP, and LTBP.30-32 Moreover, the complex of TGF-β2 and LAP is termed as the small latent complex (SLC).33 After secretion, the LLC can bind to various fibres in the extracellular matrix (ECM) by LTBP.32 Of course, the LLC of ECM can also be derived from paracrine.34 Eventually, the LLC is activated by a number of ECM factors, resulting in the formation of active TGF-β2. TGF-β1 and TGF-β3 contain an argine-glycine-aspartic acid (RGD) motif that is recognized by αv integrins, and LLC is activated by integrin αvβ6 in extracellular matrix.35,36 However, TGF-β2 lacks an RGD motif, resulting in its LLC unable to be activated by integrin αvβ6.37 This indicates that TGF-β2 may contain other conserved motifs that cannot be found in the other two TGF-βs.38 Although no studies showed that TGF-β2 could be activated by integrins, many studies have suggested that latent TGF-β2 could be activated by a number of other activators, such as matrix metalloproteinase-2/3 (MMP-2/3) and thrombospondin-1 (TSP1).37,38 To date, the mechanism by which latent TGF-β2 is activated by various cytokines remains unclear. In the future, further studies of body may determine how to activate latent TGF-β2 and suggest specific activators as drug targets to improve the therapeutic effect of diverse diseases.
Fig. 1.
The schematic diagram showing active transforming growth factor-beta2 (TGF-β2) secretion. Starting at the bottom right: New synthetic pro-TGF-β2 forms dimer/trimeric complexes in the endoplasmic reticulum (ER) with the help of latent TGF-β2-binding protein (LTBP). These dimer/trimeric complexes are then further processed in the trans-Golgi network to form large latent complexes (LLCs). After they are secreted, the LLC may bind to various fibres in the extracellular matrix (ECM) with the help of LTBP. Eventually, the LLC is activated by a number of ECM factors, resulting in the formation of active TGF-β2. LAP, latency-associated peptide.
TGF-β2 receptors
Activated TGF-β2 delivers signals by binding to its receptors, which are a family of transmembrane protein serine/threonine kinases.39 TGF-β receptors are divided into three subfamilies: type I (TβRI), type II (TβRII), and type III (TβRIII) receptors, based on their structural and functional properties.40 These receptors’ properties are described as follows:41-44
Receptor I: TβRI contains extracellular domain (ECD), which is a specific serine/threonine domain, and the L45 loop, which is a nine amino acid sequence in ECD. Next to ECD, there is a glycine-serine-rich juxtamembrane domain (GS-domain), phosphorylated by the type II receptor kinase. TβRI cannot bind ligands directly but can form a complex with TβRII when TGF-β2 is present.
Receptor II: TβRII has the same domain (ECD) as TβRI. TβRII has sustained kinase activity and can phosphorylate TβRI when these two receptors form complexes, resulting in transmission of the downstream signal of TGF-β2.
Receptor III: β-glycan, which is one of the members of TβRIII, another being endoglin, does not activate protein kinase. β-glycan not only concentrates TGF-β but also stabilizes TGF-β, particularly TGF-β2. However, endoglin selectively binds TGF-β1 and TGF-β3.
A great body of evidence suggested that TGF-β2 signals activated canonical ALK5/TβRII/Smad2/3 pathway to exert its functions.13,44,45 However, in recent research, TGF-β2 has been shown to transduce a signal through noncanonical TβRI/TβRIII/TAK1/p38 pathway to regulate proliferation of palatal mesenchyme, resulting in cleft palate.46-48 In addition, some research reported that ALK5 could form a complex with other type II receptors to transduce TGF-β2 signal.49,50 These results indicated that the mechanisms with which TGF-β2 binded to its receptors were highly complex. Therefore, to better understand what affects these mechanisms, further investigation is needed.
TGF-β2 signalling pathway
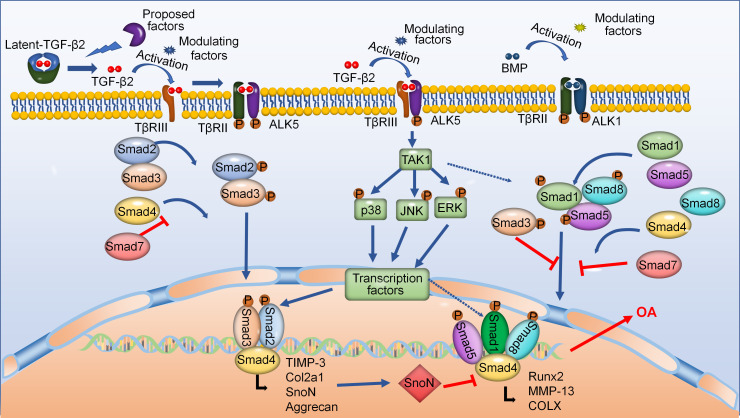
TGF-β2 can regulate cartilage development and maintenance by its canonical and noncanonical signalling pathways (Figure 2). The LLC is activated by various factors in the ECM, and then active TGF-β2 initiates canonical Smad-dependent signalling pathway.32 Active TGF-β2 is presented by type III (TβRIII) to its receptors, which are heteromeric complexes of type II (TβRII) and type I (ALK5) receptors.51 TGF-β2 signals are then transmitted through their downstream signalling pathways that include Smad2 and Smad3.52 Activated and phosphorylated Smad3/Smad2 form a complex with Smad4 and then translocate into the nucleus.53 Finally, this complex regulates target gene expression by interacting with other transcription factors in the nucleus.45 Moreover, active TGF-β2 can also induce the activation of noncanonical MAPK signalling pathways that include p38, extracellular signal-regulated protein kinase (ERK) and Jun N-terminal kinase (JNK), and its receptors are TβRIII/ALK5 complexes that do not contain TβRII.13 Besides, BMP, a member of the TGF-β superfamily, can activate Smad1/5/8 signalling pathways through TGF-β-activated kinase1 (TAK1) receptor.54 Studies indicated that BMP signalling pathways played an indispensable role in the early chondrogenesis.55,56 Interestingly, TGF-β2 signalling pathways generally can resist BMP pathways to maintain cartilage homeostasis by forming mixed Smad3/Smad1/5 complexes, or ski-related novel protein N (SnoN).57 However, whether and how TAK1 mediates the interaction of TGF-β2 signals and Smad1/5/8 remains unclear.
Fig. 2.
Transforming growth factor-beta2 (TGF-β2) regulates cartilage homeostasis and disease progression via its canonical Smad-dependent and noncanonical mitogen-activated protein kinases (MAPK) signalling pathways. During the initiation of the canonical Smad-dependent signalling pathway, active TGF-β2 is binded to its receptors, then activates downstream Smad-dependent signalling pathway to regulate homeostasis of chondrocytes. Moreover, TGF-β2 signals can also transmit to noncanonical TGF-β-activated kinase1 (TAK1)-mediated pathways. Additionally, TGF-β2 signals are involved in bone morphogenetic protein (BMP) signalling pathways. ALK1, activin receptor-like kinase; Col2a1, collagen, type II, alpha 1; COLX, collagen type x; MMP-13, matrix metalloproteinase-13; OA, osteoarthritis; TβR, transforming growth beta receptor; TIMP-3, tissue inhibitor of metalloproteinase 3.
Smad-dependent TGF-β2 signalling pathway
Smad proteins are canonical intracellular mediators of TGF-β2 signalling pathways, and their name is based on the names of the caenorhabditis elegans (Sma) and drosophila (Mad).58 Based on the structural and functional characteristics, these proteins are divided into three groups: regulated Smads (Smads1/2/3/5/8), Co-Smad (Smad4), and inhibitory Smads (Smads6/7).53,59 In addition, there is a unique structure of Smad proteins that contains highly conserved N-terminal (MH1) and CTGF-terminal (MH2) domains and a proline-rich linker region that is of divergent length and sequence.60 These Smad proteins’ properties are described as followed:61-68
R-Smad: The highly important structure of the MH1 domain that is for binding to DNA are the β-hairpin and nuclear localization signals (NLS). A unique insert in Smad2-MH1 domain, which corresponds to exon. The carboxy-terminal MH2 domain contains a nuclear export signal (NES) that inhibits Smad protein from migrating to nucleus, the L3 loop structure (a 17 amino acid region) that can bind to the type receptors, and the Ser-Ser-X-Ser sequence (SSXS motif) that can be phosphorylated by the type I receptor kinases. The proline-tyrosine-rich (PPXY) motif in the linker domain is recognized by the tryptophan-tryptophan (WW) domain of Smurf family proteins.
Co-Smad: Smad4 encompasses the nuclear export signal at the MH1-linker border, the Smad activation domain (SAD) at the linker-MH2 border, and the L3 loop of MH2 domain.
I-Smad: I-Smads lack the MH1 domain and the SSXS motif, retain a conserved MH2 domain that includes the NES and the L3 loop, and negatively regulate signalling through the proline-tyrosine-rich (PPXY) motif.
Upon TGF-β2 ligand binding, the GS domain of TβRI (ALK5) is phosphorylated by TβRII, resulting in a conformational change in TβRI (ALK5) and increasing the binding affinity of receptor for Smad2 and Smad3, which is determined by the L45 loop of TβRI (ALK5) and the L3 loop of the MH2 domain.62,69 Phosphorylation of SSXS at the MH2 domain of Smad2/3 by TβRI kinase enables the formation of a tripolymer that is the complex of Smad2/3 associated with Smad4 consequently; the tripolymer migrates into the nucleus to regulate gene expression by the β-hairpin of the MH1 domain combining with DNA and the MH2 domain binding to transcription factors.62,64,70-72 Of course, this progress can be suppressed by Smad7 of I-Smad.73 In addition, recent studies suggested that the progression of Smad signalling pathway of TGF-β2 migrating to the nucleus can be disturbed by other signalling pathways, including the Wnt or Hippo pathways.74-76 According to these studies, signal transduction of TGF-β2/Smad pathway is highly complex.
Noncanonical TGF-β2 signalling pathways
Generic MAPKs are serine/threonine kinases that can be activated by various cytokines, and their signalling pathways are shared by four distinct cascades that include ERK1/2, JNK1/2/3, p38-MAPK, and ERK5.44,77,78 In recent years, however, studies indicated that the α and β subtypes of p38 were activated by TGF-β2 ligand.79,80 Furthermore, other studies showed that other signalling pathways of MAPK, such as ERK and JNK, were also activated by TGF-β2/TβRI/TβRIII/TAK1 signalling to regulate development of various cells and pathological progression of diseases.81 Of note, TAK1 can activate and cooperate with Smad1/5/8 of BMP pathways to regulate cartilage development, but whether and how TAK1 mediates BMP signalling during chondrogenesis remains to be further investigated.44
The basal role of TGF-β2 in chondrocyte life cycle
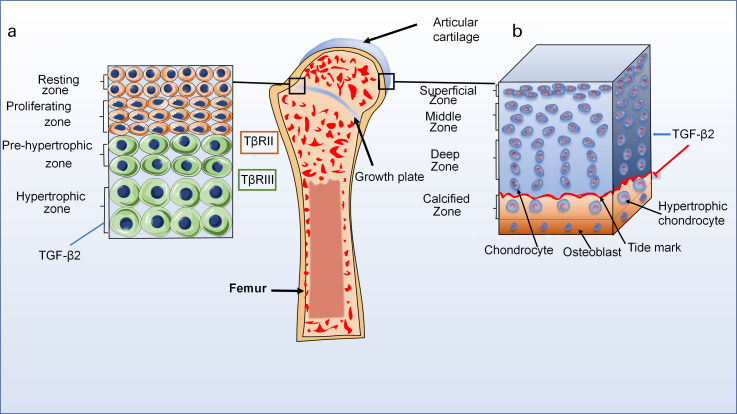
Bone formation in vertebrates is divided into endochondral ossification and intramembranous ossification.82,83 Articular chondrocytes, which do not undergo terminal differentiation, together with growth plate chondrocytes, which are finally replaced by bone in the embryonic stages of endochondral ossification, are known as two types of chondrocytes.84,85 Based on structural differences, growth plate cartilage has four zones, which are the resting zone, proliferative zone, pre-hypertrophic zone, and hypertrophic zone, and most of the terminally differentiated chondrocytes undergo apoptosis, leaving ECM that is good for the invasion of osteoblasts and blood vessels (Figure 3).86,87 Eventually, osteoblasts secrete bone matrix to replace cartilage matrix and realize ossification. TGF-β2 expresses in all zones during cartilage development, but mainly exists in the ECM of hypertrophy zone.88 Interestingly, TGF-β2 has low affinity for TβRII but a strong affinity for TβRIII.89 Besides, Horner et al88 indicated that TβRII achieved maximum expression in the proliferative zone and minimum expression through the hypertrophic zone.
Fig. 3.
The distribution of transforming growth factor-beta2 (TGF-β2) in normal articular cartilage. a) Normal structure diagram of growth plate (left). The growth plate cartilage is divided into four zones: resting zone, proliferative zone, pre-hypertrophic zone, and hypertrophic zone. TGF-β2 can be expressed in all zones during cartilage development, but the highest levels are in hypertrophy zone. Interestingly, TGF-β2 has low affinity for transforming growth factor beta receptor (TβR)II but a strong affinity for TβRIII. b) Normal structure diagram of articular cartilage (right). The articular cartilage is also divided into four zones: superficial zone, middle zone, deep zone, and calcified zone. TGF-β2 signalling pathways can maintain chondrocyte phenotype, and inhibit pre-hypertrophic and hypertrophic differentiation in articular cartilage.
Furthermore, articular cartilage is divided into four zones that incorporate the superficial (tangential) zone, middle (transitional) zone, deep (radial) zone, and calcified zone.87 Articular cartilage is composed of chondrocytes that occupy only 2% of the total tissue volume of the articular cartilage, and ECM which is composed of fibres (collagen and elastin), proteoglycans, and glycoproteins.90 Intriguingly, some studies proposed a concept that the pericellular matrix (PCM) of articular cartilage that surrounded each chondrocyte played a dual role both in normal physiological functions of articular cartilage and in the progression of OA.91,92 Moreover, Jeon et al93 showed that TGF-β superfamily might suppress the progression in PCM. Of note, TGF-β2 signals can maintain chondrocyte phenotypes, and inhibit pre-hypertrophic differentiation in the articular cartilage.13 However, the mechanisms of chondrocyte-PCM-ECM interactions and the role of TGF-β2 in PCM are not well understood. Therefore, TGF-β2 is of great importance to better understand how to maintain homeostasis of articular cartilage and find potential therapeutic targets for various diseases by researching the mechanisms of chondrocyte metabolism and ECM turnover.
Chondrocyte proliferation
Proliferation of chondrocytes plays an important role in the pathophysiological process of cartilage, which includes the maintenance of joint homeostasis and the repair of cartilage tissue defects. However, due to its unique physiological properties, cartilage results in a very poor self-repairing capability when cartilage tissue defects occur.11,12,94 Therefore, exploring the mechanisms that affect chondrocyte proliferation may provide a new method for cartilage repair. Nevertheless, TGF-β2 showed different experimental results in the regulation of chondrocyte proliferation.
Many studies reported that TGF-β2 was expressed in chondrocytes of proliferative and hypertrophic zones of rat growth plates.44,88 Furthermore, Zerath et al95 also reported that systemic infusion of TGF-β2 could alleviate the decrease in chondrocyte proliferation induced by unloading model in rats. This result suggested that TGF-β2 might promote proliferation under certain conditions. Interestingly, in recent studies, TGF-β2 had a negative effect on cell proliferation. For instance, Khaghani et al96 showed that TGF-β2 exerted an inhibitory effect on chondrocyte cell proliferation and might have synergistic effects on joint chondrocytes with other growth factors or proteins, such as basic fibroblast growth factor (bFGF).97 Moreover, cyclin B2 (the onset of proliferation) and proliferating cell nuclear antigen (PCNA) are regarded as proliferation markers.98,99 Tchetina et al98 showed that the downregulation of cyclin B2 expression had a close relationship with TGF-β2. In addition, TGF-β2 can disturb the BMP/Smad1/5/8 signalling pathway that promotes the proliferation of chondrocytes by activating TGF-β2/TAK1/p38 signalling to induce the gene expression of SnoN and Smad7.44,57,100 These results suggest that TGF-β2 regulates the mechanisms of chondrocyte cell proliferation in a highly complex manner.
Chondrocyte differentiation
Looking at chondrocytes in terms of time, chondrocyte differentiation can be divided into early chondrogenesis, which expresses the type X collagen, and terminal differentiation, which expresses vascular endothelial growth factor A (VEGFA), MMP13, and secreted phosphoprotein1 (Spp1).101 Moreover, during the process of endochondral bone formation, the differentiation of chondrocytes is an indispensable phase. Chondrocytes in growth plate and cartilage diseases undergo terminal differentiation.102 However, not all chondrocytes undergo terminal differentiation, such as chondrocytes of articular cartilage. In adult cartilage, most chondrocytes are in a resting state, which has a very poor differentiation capacity. Occasionally, a few chondrocytes are able to differentiate, and can differentiate into activated chondrocytes and hypertrophic chondrocytes. These types of chondrocytes play important roles in the formation of cartilage and the pathological process of cartilage. Interestingly, TGF-β2 signalling pathways can regulate the process of differentiation of chondrocytes. Therefore, exploring the molecular mechanisms of TGF-β2 regulating chondrocyte differentiation is of great significance for cartilage regeneration and treatment of diseases.
Early chondrogenesis
The process of early chondrogenesis in skeletal development of vertebrates is highly complicated because the stage of early chondrogenesis is regulated by multiple transcription factors and signalling pathways, such as Indian hedgehog (Ihh), parathyroid hormone-related peptide (PTHrP), and TGF-β2.98,103,104 Some chondrocytes participate in the formation of cartilage growth plates by differentiating into hypertrophic cells, and play primary roles in endochondral ossification.103 Consequently, early chondrogenesis is regarded as the onset of skeletal development and a precondition for cartilage regeneration.
In recent years, to investigate human early chondrogenesis, many researchers simulated the process by inducing stem cells such as bone marrow-derived mesenchymal stem cells (MSCs) and human adipose-derived stem cells (hASCs) to differentiate into chondrocytes.105 Moreover, most experiments assessed the cell activity and degree of differentiation of chondrocytes by evaluating the quality of typical cartilage differentiation markers such as collagen, type II, alpha 1 (Col2a1), Sox9, and the small intracellular protein S100.101,106 These studies showed that TGF-β2 could positively regulate the early phase of chondrogenesis of chondrocytes by the TGF-β2/TβRIII/Smad3 signalling pathway.104,107 For instance, Kim and Im108 suggested that the combination of TGF-β2 and BMP7 could enhance chondrogenesis from adipose tissue-derived MSCs (ATMSCs). Han et al109 indicated that β-IGH3, which was a TGF-β2-induced extracellular matrix protein, was highly expressed in early chondrogenesis in ATDC5 (a chondrocyte cell line). Kim et al110 suggested that staurosporine could induce chondrogenesis in chick embryos through canonical and noncanonical TGF-β2 pathways, which included the Smad2/3 and p38 MAPK signalling pathways. In addition, Hou et al111 showed that miR-193b negatively regulated the early chondrogenic markers sex-determining region Y box 9 (Sox9) and Col2a1. This indicated that miR-193b played a negative role in early chondrogenesis. Intriguingly, the inhibition of early chondrogenesis, which was activated by miR-193b, bound to the specific seed sequence of the 3'-untranslated regions (UTRs) of TGF-β2 and TβRIII and repressed the expression of TGF-β2 and TβRIII, resulting in the downregulation of early chondrogenesis.
Terminal differentiation
Chondrocytes differentiate into hypertrophic cells, which is the process of terminal differentiation or chondrocyte maturation. However, not all chondrocytes will undergo terminal differentiation. The terminal differentiation of chondrocytes can be seen in the hypertrophic zone of the growth plate and pathological processes of articular cartilage, such as OA.102 There is no doubt that the terminal differentiated chondrocytes are the most abundant in endochondral ossification of vertebrates. Endochondral ossification, the major mechanism in vertebrates, supports skeletal development.83,112 Endochondral ossification begins with the condensation of mesenchymal cells, which is a prerequisite for subsequent chondrogenic differentiation.113 By highly expressing various adhesion molecules, such as neural cadherin and neural cell adhesion molecules, cartilage is formed in situ, and upregulates the expression of Sox9 to form a body of collagen II and proteoglycans in extracellular matrix. Next, the cells surrounded in the aggregation centre rapidly proliferate and differentiate into different morphological characteristics of chondrocytes, forming four zones in the growth plate, including the resting zone, proliferative zone, pre-hypertrophic zone, and hypertrophic zone.86,114 Hypertrophic chondrocytes secrete a large amount of Col10a1 and MMP13, which degrades mineralized matrix and facilitates the invasion of blood vessels.115 Finally, most hypertrophic chondrocytes of terminal differentiation undergo apoptosis, providing space for invasion of osteoblasts, osteoclasts, and blood vessels and resulting in new bone formation.114 It can be easily observed that the terminal differentiation of chondrocytes plays a pivotal role in endochondral osteogenesis. Intriguingly, many researchers showed that TGF-β2 could be expressed in all layers of the growth plate but its highest levels resided in the hypertrophic layer.13,88 These studies indicated that TGF-β2 played an important role in the terminal differentiation of chondrocytes. Moreover, TGF-β2 may repress the expression of markers of the terminal differentiation of chondrocytes that contain Col10a1, MMP-13, and Runx2 by activating their signalling pathways.98,116,117 For example, TGF-β2 can act upstream of PTHrP, inhibiting the terminal differentiation of chondrocytes to repress chondrocyte hypertrophy.98,118 In addition, Alvarez et al119 suggested that TGF-β2 could facilitate Sonic Hedgehog (shh) to block terminal differentiation in vitro metatarsal cultures. Wang et al13 showed that TGF-β2 down-regulated the expression of MMP13 and Runx2 to repress chondrocyte maturation by ALK5/Smad2/3 and p38/MAPK signalling pathways. These studies all showed that TGF-β2 played a highly important role in blocking the terminal differentiation of chondrocytes in vertebrates. However, this function of TGF-β2 still needs to be confirmed in vivo.
Chondrocyte migration
In recent years, many therapies have been used to repair cartilage defects.108,120 However, surgical interventions often promote the formation of inferior fibrocartilage, which lack the mechanical properties of hyaline cartilage.121 Additionally, autologous chondrocyte implantation (ACI), a new therapy, has weak long-term effects and leads to defects in donor sites.122 With the development of cartilage tissue engineering, stem cells from different sources, such as bone marrow mesenchymal stem cells (BMSCs) and adipose tissue-derived MSCs (ATMSCs), are used to regenerate articular cartilage. Although endogenous stem cell regeneration technique of articular cartilage has made great progress, it still faces great challenges, as human-induced pluripotent stem cell (hiPSC) sources are limited and the induction mechanisms of hiPSCs are not well known. Furthermore, migration of hiPSCs to repair sites is a prerequisite step in articular cartilage regeneration. Consequently, it is very important to research how growth factors/chemokines induce stem cells to migrate to cartilage damage sites to repair defects.
At present, the most commonly used cell sources of hiPSCs in cartilage tissue engineering are BMSCs.123,124 Further, ATMSCs have attracted considerable attention because this type of stem cell is abundant and readily available.125 Lee et al126 suggested that MSCs could differentiate into chondrogenic lineage and maintain phenotypic stability during multiple passages. Therefore, the pivotal step is to induce endogenous MSCs to migrate to the site of cartilage defects to facilitate articular cartilage. Studies indicated that TGF-β2 could induce C3H10T1/2 cell (a multipotent mesenchymal precursor cell line) migration at the highest concentration.127 In all-trans-retinoid acid (ATRA)-treated C3H10T1/2 cells, the chemotactic function of TGF-β2 was significantly improved in comparison to untreated cells. Interestingly, the mRNA expression of TβR2 was obviously decreased in ATRA-treated cells, but levels of TβR1 and TβR3 decreased slightly compared with untreated cells. These results indicated that TGF-β2 might lead C3H10T1/2 cell migration through TGF-β2/TβR1/TβR3/MAPK signalling pathway. In addition, Wang et al128 suggested that TGF-β2 might induce human marrow-derived mesenchymal progenitor cells to differentiate into chondrocytes. In this experiment, the transfected progenitor cells, which were recombined by transfection of pcDNA3.1(+)/TGF-β2 into BMSCs, upregulated the expression of collagen type II and aggrecan. Moreover, Jin et al105 showed that TGF-β2 also played an important role in regulating the differentiation of stem cells into chondrocytes. After transfection with a replication-deficient adenovirus carrying h-TGF-β2 (Ad5-h TGF-β2) into adipose derived stem cells (hASCs), the recombinant stem cells were seeded onto poly (DL-lactic-co-glycolic acid) (PLGA)/alginate compounds. The result revealed that the expression of chondrocyte marker genes, Col2a1, and aggrecan was significantly increased in Ad5-h TGF-β2-transduced hASCs and the expression of type I collagen showed a downward trend in comparison to control group. Therefore, these findings indicated that the role of TGF-β2 in promoting cartilage defect repair was very important. On the contrary, Khaghani et al96 indicated that TGF-β2 repressed the wound healing process of chondrocytes by scratch assay. This meant that TGF-β2 might have a negative regulatory effect on chondrocyte migration. Consequently, more research is needed to better understand the mechanisms of TGF-β2 on chondrocyte migration.
Chondrocyte death
The major forms of death of chondrocytes, which include apoptosis, necrosis, and autophagy, are very common in OA.129 Of note, TGF-β2 may be involved in the regulation of these three major methods of chondrocyte death. Apoptosis of chondrocytes, which is very important in endochondral ossification, can be induced by TGF-β2/TAK1/p38 MAPK cascade in conjunction with the Smad-dependent pathway.13 Gibson et al130 showed that chondrocyte apoptosis was up-regulated by TGF-β2 activation in a dose-dependent manner. In several inhibitors, SB203580, a specific inhibitor of p38 kinase, completely suppressed TGF-β2-induced caspase.131 In addition, small interfering RNA (siRNA) knockdown of tumour necrosis factor receptor-associated factor 6 (TRAF6), which was an upstream activator of TAK1, efficiently inhibited apoptosis by TGF-β2/TAK1/p38 MAPK signalling pathway.132
In previous studies, death of chondrocytes by apoptosis was associated with severity of OA.133,134 Moreover, autophagy can protect chondrocyte apoptosis.135 By studying the regulatory mechanisms of autophagy in chondrocytes, new therapeutic targets to ameliorate the progression of cartilage diseases are found. Recently, many studies reported that TGF-β2 regulated autophagy in different types of cells. For instance, TGF-β2 initiated autophagy to promote glioma cells invasion by Smad and non-Smad pathways, and knockdown of Smad2 or inhibition of c-Jun NH2-terminal kinase decreased TGF-β2-induced autophagy.136-138 Moreover, Wu et al137 showed that TGF-β2-treated human retinal pigment epithelium (RPE) cells (ARPE-19 cell line) exhibited increased autophagic flux compared with control cells. Interestingly, TGF-β2-induced EMT (epithelial to mesenchymal transition) is dampened by inhibiting autophagy.138 These results showed that the role of TGF-β2 signalling pathways in regulating the autophagy of various cells might be consistent and that the process was complex. However, the role of TGF-β2 in regulation of autophagy in chondrocytes has not been studied well yet. Studying the regulation mechanism of TGF-β2 in chondrocytes autophagy, therefore, may provide new therapeutic targets to ameliorate the progression of cartilage diseases.
Effect of TGF-β2 on cartilage diseases
The ECM, synthesized by chondrocytes, together with chondrocytes make up the structure of cartilage. The activity of chondrocytes plays a significant role in maintaining homeostasis of cartilage tissues. Furthermore, aetiological factors, such as mechanical trauma, degeneration of ageing, genetic predisposition, can destroy the homeostasis of cartilage tissue. However, the self-repairing capability of cartilage tissue is very poor, resulting in failure to regenerate after destruction.139 If effective treatments are not taken, more serious consequences will result. Therefore, many researchers are searching therapeutic methods by exploring the pathological process of cartilage diseases. According to a large number of studies, TGF-β2 plays an important role in the development of cartilage diseases and may serve as a new therapeutic target.20,140-142 The following sections describe the frontiers of common cartilage diseases and their relationship with TGF-β2.
Osteoarthritis
OA is the most common and age-related joint disease.84 Worldwide, more than 15% (10% of men and 18% of women) of the elderly has been diagnosed with OA.123,143 The common clinical symptoms of OA include long-term chronic pain, joint dysfunction, and deformities.144 To date, the precise molecular mechanism of the pathogenesis of OA remains unclear. Unfortunately, there are currently no effective therapies to prevent the progression of OA. There are only symptomatic treatments, such as those that relieve pain, ameliorate stiffness, decrease patient suffering, and improve their quality of life. For advanced OA, arthroplasty surgery is the only option, and this therapeutic method hardly achieves satisfactory results. Consequently, in recent years, most researchers have explored the molecular mechanisms related to pathogenesis of OA to identify new potential molecular targets for prevention and treatment.
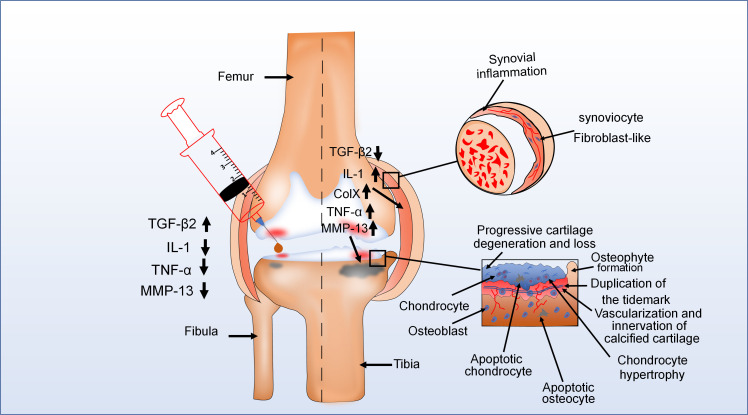
The major pathological features of OA are progressive hyaline cartilage loss, concomitant sclerotic changes in the subchondral bone, synovitis, and the development of osteophytes.20,145 During the advanced stage of OA, the epithelial lining of the joint capsule is thickened due to synovial inflammation, causing most new blood vessel formation and fibroblast-like synoviocyte proliferation (Figure 4). Meanwhile, there are high levels of interleukin-1 (IL-1) in synovial fluids of OA, and the expression level of TGF-β2 significantly decreases.146 In addition, pathological changes occur in different areas of articular cartilage. For instance, various proinflammatory factors such as IL-1, MMP-13, and TNF-α are highly expressed in articular cartilage of OA.147-149 In the deeper zone, chondrocytes undergo terminal differentiation, which can synthesize collagen type X (ColX), and this accelerates the progression of OA.94 The calcified zone exhibited several pathological changes, manifested by vascularization, innervation from subchondral bone, and duplication of tidemark.150 In the subchondral bone, osteocyte apoptosis occurs and osteophytes develop at the joint margins.151 According to most research, one of the earliest changes in OA was swelling of the cartilage ECM.152,153 This indicated that disruption of homeostasis in cartilage tissue was one of the major reasons for the initiation of OA. Moreover, the catabolic and anabolic imbalance of chondrocytes, which is due to the change in chondrocyte phenotype, results in the disorder of cartilage homeostasis.154-156 Thus, the alteration in chondrocyte phenotype may play an important role in the progression of OA. The most common phenotypic change in chondrocytes is hypertrophic phenotype in OA.157 Interestingly, studies indicated that TGF-β2 could effectively inhibit the progression of OA.147-149 TGF-β2 not only inhibits the cleavage of Col2 but also suppresses the expression of hypertrophic markers such as Runx2, Col10a1, and MMP-13.20,158 SnoN, which is induced by the TGF-β2/TAK1/p38 signalling pathway, can inhibit the expression of MMP13 by BMP/Smad1/5/8 signalling pathway in OA.13,57 In addition, research also reported that TGF-β2 could promote the expression of specific tissue inhibitors of MMPs (TIMPs) in different types of cells, especially TIMP-3.140,141 TIMP-3 can protect cartilage tissue by inhibiting MMP-13 degradation of cartilage matrix in OA. TGF-β2 can downregulate IL-1β and TNF-α to inhibit collagenase activity and proteoglycan degradation in OA.155,159,160 Moreover, TGF-β2 also plays an important role in controlling collagen degradation of articular cartilage by downregulating MMP-9 in OA.20 Despite the protective role of TGF-β2 in chondrocyte homeostasis in OA progress, studies have shown that TGF-β2 could destroy normal cartilage structure at high concentration. For instance, Elford et al161 showed that injecting high levels of TGF-β2 into rabbit normal joints caused swelling and significant loss of proteoglycan. This indicated that the regulatory role of TGF-β2 might be dose-dependent in OA. From this evidence, we learn that TGF-β2 can block the pathological changes of OA by regulating the expression of various cytokines, which suggests that TGF-β2 may be a new target for the treatment of OA.
Fig. 4.
The pathological change of knee joint in the osteochondral unit during the evolution of osteoarthritis (OA). OA is typically characterized by cartilage damage, osteophyte formation, and thickening of the joint capsule. For example, in one diagram (upper right), the epithelial lining of the joint capsule is thickened because of synovial inflammation. In another (bottom right), during advanced stages of OA, there are pathological changes in different areas of articular cartilage. Moreover, in the left diagram, injection of transforming growth factor-beta 2 (TGF-β2) into the joint cavity of patients with OA reduced the expression of some proinflammatory factors and the clinical symptoms of OA. This means that TGF-β2 plays an important role in the development of OA. ColX, collagen type X; IL-1, interleukin 1; MMP, matrix metalloproteinase; TNF-α, tumour necrosis factor alpha.
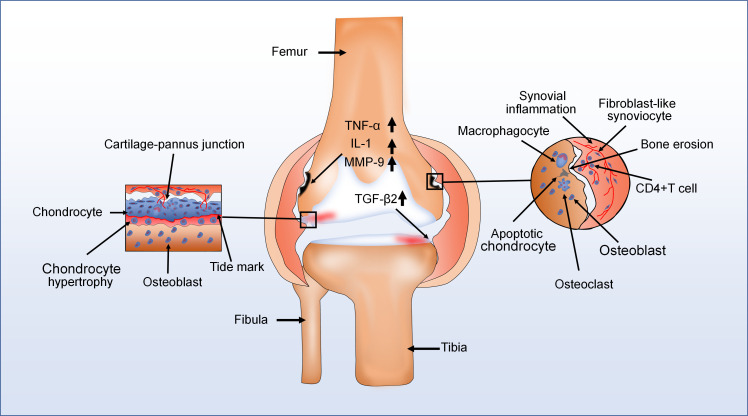
Rheumatoid arthritis
Human rheumatoid arthritis (RA) is a chronic systemic autoimmune inflammatory disease associated with pain, joint dysfunction, and other clinical symptoms.162 The major pathological characteristics of RA are chronic synovitis with hyperplasia, bone erosion, pannus formation, and immune cell infiltration (Figure 5).163,164 During the progression of RA, a variety type of cells exists in the cartilage-pannus of RA, including monocytes, cluster of differentiation 4 (CD4)+ T cells, and fibroblast-like cells. These cells secrete high level of proinflammatory factors such as IL-1, TNF-α, and MMP9 to induce cartilage damage and degradation of collagen.165-167 Interestingly, high expression of TGF-β2 is detected in the cartilage-pannus junction during RA.167 However, little research has been done on the role of TGF-β2 in RA. There is no doubt that TGF-β2 serves an important role in the pathological changes of RA. Müssener et al168 showed that the expression of TGF-β2 was upregulated with time after onset of disease in the synovial tissue. Müssener et al168 also indicated that TGF-β2 not only promoted cartilage tissue repair by promoting collagen and fibronectin synthesis, as well as downregulating synthesis of proteases, but also inhibited the entry of lymphoid cells into the arthritic joints in RA. In addition, TNF-α, a major proinflammatory factor in RA, can induce fibroblast-like synoviocytes (FLSs) to produce IL-6, IL-8, and MMPs to cleave collagen of ECM and break down bone tissue.167,169 According to some studies, evidence suggests that TGF-β2 had an inhibitory effect on TNF-α and IL-1β expression in inflammatory joint diseases.155,159,160 These results indicated that TGF-β2 might slow down the progression of RA by suppressing TNF-α expression. Han et al166 showed that gremlin1 (GREM1) was a pivotal regulator of synoviocyte hyperplasia and invasiveness in RA. In this experiment, TGF-β upregulated GREM1 expression in RA-FLS, and could result in synovial hyperplasia. However, this experiment did not prove the effect of various isoforms of TGF-β on GREM1 in RA-FLS. Therefore, whether and how TGF-β2 plays a promotive or inhibitory role in the progression of RA requires further investigation.
Fig. 5.
The major pathological characteristics of rheumatoid arthritis (RA) are chronic synovitis with hyperplasia, pannus formation, and immune cell infiltration. For instance, in one diagram (left), various cell types in the cartilage-pannus of RA secrete lots of proinflammatory factors to induce cartilage damage and degradation of collagen. In another (right), there are also numerous immune cells infiltration and development of new blood vessels in the synovium of RA with hyperplasia, and the expression of transforming growth factor-beta2 (TGF-β2) also increases. Moreover, bone erosion appeared in the vicinity of the thickened synovium. CD4, cluster of differentiation 4; IL-1, interleukin 1; MMP, matrix metalloproteinase; TNF-α, tumour necrosis factor alpha.
In summary, TGF-β2 is involved in the regulation of the entire process of endochondral ossification, such as cartilage, growth plate development, and joint formation. In addition, TGF-β2 also participates in maintaining homeostasis of cartilage tissue and regulates chondrocyte proliferation, differentiation, apoptosis, and the expression of proinflammatory factors. Altering the TGF-β2 levels or using inhibitors can affect the physiological processes of chondrocytes. Consequently, TGF-β2 exerts a significant influence on physiological and pathological processes of cartilage tissue.
Although considerable progress has been made in the study of the role of TGF-β2 in cartilage, many problems have yet to be solved. For example, the effects of TGF-β2 on the initiation and progression of RA remain unknown. So far, most of the research has been carried out on TGF-β1 and TGF-β3, while research into the role of TGF-β2 in cartilage has been very limited. Therefore, the mechanism by which TGF-β2 regulates chondrogenesis and cartilage maturation is unclear. The regulatory mechanisms in the physiological and pathological processes of cartilage mediated by TGF-β2 are also not well understood. In addition, little evidence is provided to explain how TGF-β2 recruits stem cells to repair cartilage defects through signalling pathways. Hence, in the future, more research needs to be done to detect potential mechanisms of TGF-β2 in joint tissues, which will deepen understanding and recognition of the molecular mechanisms of chondrogenesis and be beneficial to the prevention, diagnosis, and therapy of diseases of cartilage tissue.
Author contributions
M. Duan: Writing - original draft.
Q. Wang: Writing - review and editing.
Y. Liu: Writing - original manuscript.
J. Xie: Conceptualization.
Funding statement
The open access funding for this study was obtained via grants from The National Natural Science Foundation of China (No. 81600840 and 81771047) for this study. No benefits in any form have been received or will be received from a commercial party related directly or indirectly to the subject of this article.
ICMJE COI statement
The authors declare that no competing interests exist.
References
- 1.Watabe T, Miyazono K. Roles of TGF-β family signaling in stem cell renewal and differentiation. Cell Res. 2009;19(1):103–115. [DOI] [PubMed] [Google Scholar]
- 2.Wojciech S, Ahmad R, Belaid-Choucair Z, et al. The orphan GPR50 receptor promotes constitutive TGFβ receptor signaling and protects against cancer development. Nat Commun. 2018;9(1):1216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Seoane J, Gomis RR. TGF-β Family Signaling in Tumor Suppression and Cancer Progression. Cold Spring Harb Perspect Biol. 2017;9(12):a022277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Manokawinchoke J, Pavasant P, Sawangmake C, et al. Intermittent compressive force promotes osteogenic differentiation in human periodontal ligament cells by regulating the transforming growth factor-β pathway. Cell Death Dis. 2019;10:761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Su Y, Feng W, Shi J, Chen L, Huang J, Lin T. circRIP2 accelerates bladder cancer progression via miR-1305/Tgf-β2/smad3 pathway. Mol Cancer. 2020;19(1):23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bohlen CJ, Bennett FC, Tucker AF, Collins HY, Mulinyawe SB, Barres BA. Diverse requirements for microglial survival, specification, and function revealed by Defined-Medium cultures. Neuron. 2017;94(4):759–773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Huang G, Wang Y, Shi LZ, Kanneganti T-D, Chi H, et al. Signaling by the phosphatase MKP-1 in dendritic cells imprints distinct effector and regulatory T cell fates. Immunity. 2011;35(1):45–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Russo I, Cavalera M, Huang S, et al. Protective effects of activated myofibroblasts in the pressure-overloaded myocardium are mediated through Smad-dependent activation of a matrix-preserving program. Circ Res. 2019;124(8):1214–1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dropmann A, Dooley S, Dewidar B, et al. TGF-β2 silencing to target biliary-derived liver diseases. Gut. 2020;69(9):1677–1690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Luo Y, Sinkeviciute D, He Y, et al. The minor collagens in articular cartilage. Protein Cell. 2017;8:560–572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hunziker EB, Lippuner K, Shintani N. How best to preserve and reveal the structural intricacies of cartilaginous tissue. Matrix Biol. 2014;39:33–43. [DOI] [PubMed] [Google Scholar]
- 12.Yu F, Li M, Yuan Z, et al. Mechanism research on a bioactive resveratrol- PLA-gelatin porous nano-scaffold in promoting the repair of cartilage defect. Int J Nanomedicine. 2018;13:7845–7858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang W, Rigueur D, Lyons KM. Tgfβ signaling in cartilage development and maintenance. Birth Defect Res C. 2014;102(1):37–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Basu RK, Hubchak S, Hayashida T, Runyan CE, Schumacker PT, Schnaper HW. Interdependence of HIF-1α and TGF-β/Smad3 signaling in normoxic and hypoxic renal epithelial cell collagen expression. Am J Physiol Renal Physiol. 2011;300(4):F898–F905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Das R, Timur UT, Edip S, et al. TGF-β2 is involved in the preservation of the chondrocyte phenotype under hypoxic conditions. Ann Anat. 2015;198:1–10. [DOI] [PubMed] [Google Scholar]
- 16.Wang Q, Zhou C, Li X, et al. TGF‐β1 promotes gap junctions formation in chondrocytes via Smad3/Smad4 signalling. Cell Prolif. 2019;52(2):e12544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mooney MP, Losken HW, Moursi AM, et al. Anti-TGF-beta2 antibody therapy inhibits postoperative resynostosis in craniosynostotic rabbits. Plast Reconstr Surg. 2007;119(4):1200–1212. [DOI] [PubMed] [Google Scholar]
- 18.Fan Y, Guo L, Wei J, Chen J, Sun H, Guo T. Effects of salidroside on trabecular meshwork cell extracellular matrix expression and mouse intraocular pressure. Invest Ophthalmol Vis Sc. 2019;60(6):2072–2082. [DOI] [PubMed] [Google Scholar]
- 19.Zhang Z, Zhu H, Liu Y, Quan F, Zhang X, Yu L. Lncrna HOTAIR mediates TGF-β2-induced cell growth and epithelial-mesenchymal transition in human lens epithelial cells. Acta Biochim Biophys Sin. 2018;50(10):1028–1037. [DOI] [PubMed] [Google Scholar]
- 20.Tchetina EV, Antoniou J, Tanzer M, Zukor DJ, Poole AR, et al. Transforming growth factor-β2 suppresses collagen cleavage in cultured human osteoarthritic cartilage, reduces expression of genes associated with chondrocyte hypertrophy and degradation, and increases prostaglandin E2 production. Am J Pathol. 2006;168(1):131–140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Derynck R, Jarrett JA, Chen EY, et al. Human transforming growth factor-beta complementary DNA sequence and expression in normal and transformed cells. Nature. 1985;316(6030):701–705. [DOI] [PubMed] [Google Scholar]
- 22.Navarini AA, Simpson MA, Weale M, et al. Genome-wide association study identifies three novel susceptibility loci for severe acne vulgaris. Nat Commun. 2014;5:4020. [DOI] [PubMed] [Google Scholar]
- 23.Ko JM, Bae J-S, Choi JS, et al. Skeletal overgrowth syndrome caused by overexpression of C-type natriuretic peptide in a girl with balanced chromosomal translocation, t(1;2)(q41;q37.1). Am J Med Genet A. 2015;167(5):1033–1038. [DOI] [PubMed] [Google Scholar]
- 24.Liu W, Zhang D, Li X, et al. TGF-β1 facilitates cell-cell communication in osteocytes via connexin43- and pannexin1-dependent gap junctions. Cell Death Discov. 2019;5:141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zou Z, Sun PD. Overexpression of human transforming growth factor-β1 using a recombinant CHO cell expression system. Protein Expr Purif. 2004;37(2):265–272. [DOI] [PubMed] [Google Scholar]
- 26.Todorovic V, Finnegan E, Freyer L, Zilberberg L, Ota M, Rifkin DB. Long form of latent TGF-β binding protein 1 (Ltbp1L) regulates cardiac valve development. Dev Dyn. 2011;240(1):176–187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ofiteru AM, Becheru DF, Gharbia S, et al. Qualifying osteogenic potency assay metrics for human multipotent stromal cells: TGF-β2 a telling eligible biomarker. Cells. 2020;9(12):2559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Paliwal S, Shi J, Dhru U, Zhou Y, Schuger L. P311 binds to the latency associated protein and downregulates the expression of TGF-beta1 and TGF-beta2. Biochem Biophys Res Commun. 2004;315(4):1104–1109. [DOI] [PubMed] [Google Scholar]
- 29.Mercado-Pimentel ME, Runyan RB. Multiple transforming growth factor-β isoforms and receptors function during epithelial-mesenchymal cell transformation in the embryonic heart. Cells Tissues Organs. 2007;185(1-3):146–156. [DOI] [PubMed] [Google Scholar]
- 30.Lawrence DA. Latent-TGF-beta: an overview. Mol Cell Biochem. 2001;219(1-2):163–170. [DOI] [PubMed] [Google Scholar]
- 31.Tran DQ, Andersson J, Wang R, Ramsey H, Unutmaz D, Shevach EM. GARP (LRRC32) is essential for the surface expression of latent TGF- on platelets and activated FOXP3+ regulatory T cells. Proc Natl Acad Sci U S A. 2009;106(32):13445–13450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Robertson IB, Rifkin DB. Regulation of the bioavailability of TGF-β and TGF-β-Related proteins. Cold Spring Harb Perspect Biol. 2016;8(6):a021907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hyytiäinen M, Penttinen C, Keski-Oja J. Latent TGF-β binding proteins: extracellular matrix association and roles in TGF-β activation. Crit Rev Clin Lab Sci. 2004;41(3):233–264. [DOI] [PubMed] [Google Scholar]
- 34.Nilsson EE, Doraiswamy V, Skinner MK. Transforming growth factor-beta isoform expression during bovine ovarian antral follicle development. Mol Reprod Dev. 2003;66(3):237–246. [DOI] [PubMed] [Google Scholar]
- 35.Shi M, Zhu J, Wang R, et al. Latent TGF-β structure and activation. Nature. 2011;474(7351):343–349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Annes JP, Chen Y, Munger JS, Rifkin DB. Integrin αVβ6-mediated activation of latent TGF-β requires the latent TGF-β binding protein-1. J Cell Biol. 2004;165(5):723–734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Annes JP, Rifkin DB, Munger JS. The integrin alphavbeta6 binds and activates latent TGFbeta3. FEBS Lett. 2002;511:65–68. [DOI] [PubMed] [Google Scholar]
- 38.Robertson IB, Rifkin DB. Unchaining the beast; insights from structural and evolutionary studies on TGFβ secretion, sequestration, and activation. Cytokine Growth Factor Rev. 2013;24(4):355–372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yung L-M, Nikolic I, Paskin-Flerlage SD, Pearsall RS, Kumar R, Yu PB. A selective transforming growth factor-β ligand trap attenuates pulmonary hypertension. Am J Respir Crit Care Med. 2016;194(9):1140–1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hinck AP, Mueller TD, Springer TA. Structural biology and evolution of the TGF-β family. Cold Spring Harb Perspect Biol. 2016;8(12):a022103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Yan X, Xiong X, Chen YG. Feedback Regulation of Tgf-β Signaling. Vol 50. Shanghai: Acta Biochim Biophys Sin. 2018: 37–50. [DOI] [PubMed] [Google Scholar]
- 42.Liu IM, Schilling SH, Knouse KA, Choy L, Derynck R, Wang X-F. TGFβ-stimulated Smad1/5 phosphorylation requires the ALK5 L45 loop and mediates the pro-migratory TGFβ switch. Embo J. 2009;28(2):88–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wrana JL, Attisano L, Wieser R, Ventura F, Massagué J. Mechanism of activation of the TGF-beta receptor. Nature. 1994;370(6488):341–347. [DOI] [PubMed] [Google Scholar]
- 44.Guimond A, Sulea T, Pen A, Ear P, O'Connor-McCourt MD, et al. Site-Directed mutagenesis of the type II TGF-β receptor indicates a ligand-binding site distinct from that of the type II activin receptor. FEBS Lett. 2002;515(1-3):13–19. [DOI] [PubMed] [Google Scholar]
- 45.Massagué J, Seoane J, Wotton D. Smad transcription factors. Genes Dev. 2005;19(23):2783–2810. [DOI] [PubMed] [Google Scholar]
- 46.Massagué J. TGF-β signaling in development and disease. FEBS Lett. 1833;2012:586. [DOI] [PubMed] [Google Scholar]
- 47.Iwata J-ichi, Hacia JG, Suzuki A, Sanchez-Lara PA, Urata M, Chai Y. Modulation of noncanonical TGF-β signaling prevents cleft palate in Tgfbr2 mutant mice. J Clin Invest. 2012;122(3):873–885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Nickel J, Ten Dijke P, Mueller TD. TGF-β family co-receptor function and signaling. Acta Biochim Biophys Sin. 2018;50(1):12–36. [DOI] [PubMed] [Google Scholar]
- 49.Yumoto K, Thomas PS, Lane J, et al. TGF-β-activated kinase 1 (TAK1) mediates agonist-induced Smad activation and linker region phosphorylation in embryonic craniofacial neural crest-derived cells. J Biol Chem. 2013;288(19):13467–13480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhao H, Oka K, Bringas P, Kaartinen V, Chai Y, et al. TGF-β type I receptor Alk5 regulates tooth initiation and mandible patterning in a type II receptor-independent manner. Dev Biol. 2008;320(1):19–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Tai T-F, Chan C-P, Lin C-C, Chen L-I, Jeng J-H, Chang M-C. Transforming growth factor β2 regulates growth and differentiation of pulp cells via ALK5/Smad2/3. J Endod. 2008;34(4):427–432. [DOI] [PubMed] [Google Scholar]
- 52.Hinck AP. Structural studies of the TGF-βs and their receptors - insights into evolution of the TGF-β superfamily. FEBS Lett. 2012;586(14):1860–1870. [DOI] [PubMed] [Google Scholar]
- 53.Song B, Estrada KD, Lyons KM. Smad signaling in skeletal development and regeneration. Cytokine Growth Factor Rev. 2009;20(5-6):379–388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wu MY, Hill CS. Tgf-Beta superfamily signaling in embryonic development and homeostasis. Dev Cell. 2009;16(3):329–343. [DOI] [PubMed] [Google Scholar]
- 55.Keller B, Yang T, Chen Y, et al. Interaction of TGFβ and BMP signaling pathways during chondrogenesis. PLoS One. 2011;6(1):e16421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wu M, Chen G, Li Y-P. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 2016;4(1):16009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kawamura I, Maeda S, Imamura K, et al. Snon suppresses maturation of chondrocytes by mediating signal cross-talk between transforming growth factor-β and bone morphogenetic protein Pathways*. J Biol Chem. 2012;287(34):29101–29113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Cao X, Li X, Hu J, Bao Y. Hydrogen peroxide-induced cellular apoptosis is mediated by TGF-β2 signaling pathway in cultured human lens epithelial cells. Int Ophthalmol. 2010;30(3):229–237. [DOI] [PubMed] [Google Scholar]
- 59.Liu J, Tseu I, Wang J, Tanswell K, Post M, et al. Transforming growth factor beta2, but not beta1 and beta3, is critical for early rat lung branching. Dev Dyn. 2000;217(4):343–360. [DOI] [PubMed] [Google Scholar]
- 60.Guca E, Suñol D, Ruiz L, et al. Tgif1 homeodomain interacts with Smad MH1 domain and represses TGF-β signaling. Nucleic Acids Res. 2018;46(17):9220–9235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Savage C, Das P, Finelli AL, et al. Caenorhabditis elegans genes sma-2, sma-3, and sma-4 define a conserved family of transforming growth factor beta pathway components. Proc Natl Acad Sci U S A. 1996;93(2):790–794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Massagué J. Tgfβ signalling in context. Nat Rev Mol Cell Biol. 2012;13(10):616–630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hata A, Chen Y-G. TGF-β Signaling from Receptors to Smads. Cold Spring Harb Perspect Biol. 2016;8(9):a022061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lo R S CYG, Shi Y, et al. The L3 loop: a structural motif determining specific interactions between Smad proteins and TGF-beta receptors. Embo J. 1998;17:996–1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Shi Y, Massagué J. Mechanisms of TGF-β signaling from cell membrane to the nucleus. Cell. 2003;113(6):685–700. [DOI] [PubMed] [Google Scholar]
- 66.Massagué J, Gomis RR. The logic of TGFβ signaling. FEBS Lett. 2006;580(12):2811–2820. [DOI] [PubMed] [Google Scholar]
- 67.Bruce DL, Sapkota GP. Phosphatases in Smad regulation. FEBS Lett. 2012;586(14):1897–1905. [DOI] [PubMed] [Google Scholar]
- 68.Beich-Frandsen M, Aragón E, Llimargas M, et al. Structure of the N-terminal domain of the protein Expansion: an `Expansion’ to the Smad MH2 fold. Acta Crystallogr D Biol Crystallogr. 2015;71(4):844–853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Miyazono K-ichi, Ohno Y, Wada H, et al. Structural basis for receptor-regulated SMAD recognition by MAN1. Nucleic Acids Res. 2018;46(22):12139–12153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Huse M, Muir TW, Xu L, et al. The TGF beta receptor activation process: an inhibitor- to substrate-binding switch. Mol Cell. 2001;8:671–682. [DOI] [PubMed] [Google Scholar]
- 71.Liu X, Sun Y, Constantinescu SN, Karam E, Weinberg RA, Lodish HF. Transforming growth factor -induced phosphorylation of Smad3 is required for growth inhibition and transcriptional induction in epithelial cells. Proc Natl Acad Sci U S A. 1997;94(20):10669–10674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Derynck R, Zhang YE. Smad-Dependent and Smad-independent pathways in TGF-β family signalling. Nature. 2003;425(6958):577–584. [DOI] [PubMed] [Google Scholar]
- 73.Feng X-H, Derynck R. Specificity and versatility in TGF-beta signaling through Smads. Annu Rev Cell Dev Biol. 2005;21:659–693. [DOI] [PubMed] [Google Scholar]
- 74.Pervan CL. Smad-Independent TGF-β2 signaling pathways in human trabecular meshwork cells. Exp Eye Res. 2017;158:137–145. [DOI] [PubMed] [Google Scholar]
- 75.Namachivayam K, Blanco CL, MohanKumar K, et al. Smad7 inhibits autocrine expression of TGF-β 2 in intestinal epithelial cells in baboon necrotizing enterocolitis. Am J Physiol Gastrointest Liver Physiol. 2013;304(2):G167–G180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Zhou W, Lin L, Majumdar A, et al. Modulation of morphogenesis by noncanonical Wnt signaling requires ATF/CREB family-mediated transcriptional activation of TGFbeta2. Nat Genet. 2007;39(10):1225–1234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Futakuchi A, Inoue T, Wei F-Y, et al. YAP/TAZ Are Essential for TGF-β2-Mediated Conjunctival Fibrosis. Invest Ophthalmol Vis Sci. 2018;59(7):3069–3078. [DOI] [PubMed] [Google Scholar]
- 78.Lu M, Wang Y, Zhan X. The MAPK pathway-based drug therapeutic targets in pituitary adenomas. Front Endocrinol. 2019;10:330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sun Y, Liu W-. Z, Liu T, Feng X, Yang N, Zhou H-. F. Signaling pathway of MAPK/ERK in cell proliferation, differentiation, migration, senescence and apoptosis. J Recept Signal Transduct Res. 2015;35(6):600–604. [DOI] [PubMed] [Google Scholar]
- 80.Bragado P, Estrada Y, Parikh F, et al. TGF-β2 dictates disseminated tumour cell fate in target organs through TGF-β-RIII and p38α/β signalling. Nat Cell Biol. 2013;15(11):1351–1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Bian Z-M, Elner SG, Elner VM. Regulation of VEGF mRNA expression and protein secretion by TGF-β2 in human retinal pigment epithelial cells. Exp Eye Res. 2007;84(5):812–822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Lee S-W, Choi K-Y, Cho J-Y, et al. TGF-β2 stimulates cranial suture closure through activation of the Erk-MAPK pathway. J Cell Biochem. 2006;98(4):981–991. [DOI] [PubMed] [Google Scholar]
- 83.Runyan CM, Gabrick KS. Biology of bone formation, fracture healing, and distraction osteogenesis. J Craniofac Surg. 2017;28(5):1380–1389. [DOI] [PubMed] [Google Scholar]
- 84.van der Kraan PM, van den Berg WB. Chondrocyte hypertrophy and osteoarthritis: role in initiation and progression of cartilage degeneration? Osteoarthritis and Cartilage. 2012;20(3):223–232. [DOI] [PubMed] [Google Scholar]
- 85.Pacifici M, Golden EB, Oshima O, Shapiro IM, Leboy PS, Adams SL. Hypertrophic chondrocytes. the terminal stage of differentiation in the chondrogenic cell lineage? Ann N Y Acad Sci. 1990;599:45–57. [DOI] [PubMed] [Google Scholar]
- 86.Kronenberg HM. Developmental regulation of the growth plate. Nature. 2003;423(6937):332–336. [DOI] [PubMed] [Google Scholar]
- 87.Zhou C, Wang Q, Zhang D, Cai L, Du W, Xie J. Compliant substratum modulates vinculin expression in focal adhesion plaques in skeletal cells. Int J Oral Sci. 2019;11(2):18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Horner A, Kemp P, Summers C, et al. Expression and distribution of transforming growth factor-beta isoforms and their signaling receptors in growing human bone. Bone. 1998;23(2):95–102. [DOI] [PubMed] [Google Scholar]
- 89.Baardsnes J, Hinck CS, Hinck AP, O’Connor-McCourt MD. TbetaR-II discriminates the high- and low-affinity TGF-beta isoforms via two hydrogen-bonded ion pairs. Biochemistry. 2009;48(10):2146–2155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Shapiro IM, Adams CS, Freeman T, Srinivas V. Fate of the hypertrophic chondrocyte: microenvironmental perspectives on apoptosis and survival in the epiphyseal growth plate. Birth Defect Res C. 2005;75(4):330–339. [DOI] [PubMed] [Google Scholar]
- 91.Wilusz RE, Sanchez-Adams J, Guilak F. The structure and function of the pericellular matrix of articular cartilage. Matrix Biol. 2014;39:25–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Sanchez-Adams J, Wilusz RE, Guilak F. Atomic force microscopy reveals regional variations in the micromechanical properties of the pericellular and extracellular matrices of the Meniscus. J Orthop Res. 2013;31(8):1218–1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Jeon JE, Schrobback K, Meinert C, Sramek V, Hutmacher DW, Klein TJ. Effect of preculture and loading on expression of matrix molecules, matrix metalloproteinases, and cytokines by expanded osteoarthritic chondrocytes. Arthritis Rheum. 2013;65(9):2356–2367. [DOI] [PubMed] [Google Scholar]
- 94.Goldring SR, Goldring MB. Changes in the osteochondral unit during osteoarthritis: structure, function and cartilage-bone crosstalk. Nat Rev Rheumatol. 2016;12(11):632–644. [DOI] [PubMed] [Google Scholar]
- 95.Zerath E, Holy X, Mouillon JM, et al. TGF-β2 prevents the impaired chondrocyte proliferation induced by unloading in growth plates of young rats. Life Sci. 1997;61(24):2397–2406. [DOI] [PubMed] [Google Scholar]
- 96.Khaghani SAB, Akbarova G, Soon CF, Dilbazi G. Effect of transforming growth factor-β2 on biological regulation of multilayer primary chondrocyte culture. Cell Tissue Bank. 2018;19(4):763–775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Okazaki R, Sakai A, Nakamura T, Kunugita N, Norimura T, Suzuki K. Effects of transforming growth factor beta S and basic fibroblast growth factor on articular chondrocytes obtained from immobilised rabbit knees. Ann Rheum Dis. 1996;55(3):181–186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Tchetina E, Mwale F, Poole A. Distinct phases of coordinated early and late gene expression in growth plate chondrocytes in relationship to cell proliferation, matrix assembly, remodeling, and cell differentiation. J Bone Miner Res. 2003;18(5):844–851. [DOI] [PubMed] [Google Scholar]
- 99.Hayes AJ, MacPherson S, Morrison H, Dowthwaite G, Archer CW, et al. The development of articular cartilage: evidence for an appositional growth mechanism. Anat Embryol. 2001;203(6):469–479. [DOI] [PubMed] [Google Scholar]
- 100.Pal A, Ashworth JC, Collier P, et al. A 3D heterotypic breast cancer model demonstrates a role for mesenchymal stem cells in driving a proliferative and invasive phenotype. Cancers. 2020;12(8):2290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Kozhemyakina E, Lassar AB, Zelzer E. A pathway to bone: signaling molecules and transcription factors involved in chondrocyte development and maturation. Development. 2015;142(5):817–831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Bertrand J, Cromme C, Umlauf D, Frank S, Pap T, et al. Molecular mechanisms of cartilage remodelling in osteoarthritis. Int J Biochem Cell Biol. 2010;42(10):1594–1601. [DOI] [PubMed] [Google Scholar]
- 103.Lefebvre V, Smits P. Transcriptional control of chondrocyte fate and differentiation. Birth Defect Res C. 2005;75(3):200–212. [DOI] [PubMed] [Google Scholar]
- 104.Shimizu H, Yokoyama S, Asahara H. Growth and differentiation of the developing limb bud from the perspective of chondrogenesis. Dev Growth Differ. 2007;49(6):449–454. [DOI] [PubMed] [Google Scholar]
- 105.Jin XB, Sun YS, Zhang K, et al. Tissue engineered cartilage from hTGF beta2 transduced human adipose derived stem cells seeded in PLGA/alginate compound in vitro and in vivo. J Biomed Mater Res A. 2008;86(4):1077–1087. [DOI] [PubMed] [Google Scholar]
- 106.Lehmann M, Martin F, Mannigel K, Kaltschmidt K, Sack U, Anderer U. Three-Dimensional scaffold-free fusion culture: the way to enhance chondrogenesis of in vitro propagated human articular chondrocytes. Eur J Histochem. 2013;57(4):e31:31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Kawai J, Akiyama H, Shigeno C, Ito H, Konishi J, Nakamura T. Effects of transforming growth factor-β signaling on chondrogenesis in mouse chondrogenic EC cells, ATDC5. Eur J Cell Biol. 1999;78(10):707–714. [DOI] [PubMed] [Google Scholar]
- 108.Kim H-J, Im G -I. Combination of Transforming Growth Factor-Beta 2 and Bone Morphogenetic Protein 7 Enhances Chondrogenesis from Adipose Tissue-Derived Mesenchymal Stem Cells. Tissue Eng Part A. 2009;15(7):1543–1551. [DOI] [PubMed] [Google Scholar]
- 109.Han M-S, Kim J-E, Shin H-I, Kim I-S. Expression patterns of βig-h3 in chondrocyte differentiation during endochondral ossification. Exp Mol Med. 2008;40(4):453–460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Kim H, Kei K, Sonn JK. Staurosporine induces chondrogenesis of chick embryo wing bud mesenchyme in monolayer cultures through canonical and non-canonical TGF-β pathways. In Vitro Cell Dev Biol Anim. 2016;52(1):120–129. [DOI] [PubMed] [Google Scholar]
- 111.Hou C, Yang Z, Kang Y, et al. MiR-193b regulates early chondrogenesis by inhibiting the TGF-beta2 signaling pathway. FEBS Lett. 2015;589(9):1040–1047. [DOI] [PubMed] [Google Scholar]
- 112.Garzón-Alvarado DA. A biochemical strategy for simulation of endochondral and intramembranous ossification. Comput Methods Biomech Biomed Engin. 2014;17(11):1237–1247. [DOI] [PubMed] [Google Scholar]
- 113.Long F, Ornitz DM. Development of the endochondral skeleton. Cold Spring Harb Perspect Biol. 2013;5(1):a008334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Hinton RJ, Jing Y, Jing J, Feng JQ. Roles of chondrocytes in endochondral bone formation and fracture repair. J Dent Res. 2017;96(1):23–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Mackie EJ, Ahmed YA, Tatarczuch L, Chen K-S, Mirams M. Endochondral ossification: how cartilage is converted into bone in the developing skeleton. Int J Biochem Cell Biol. 2008;40(1):46–62. [DOI] [PubMed] [Google Scholar]
- 116.Szüts V, Möllers U, Bittner K, et al. Terminal differentiation of chondrocytes is arrested at distinct stages identified by their expression repertoire of marker genes. Matrix Biology. 1998;17(6):435–448. [DOI] [PubMed] [Google Scholar]
- 117.Tchetina EV, Squires G, Poole AR. Increased type II collagen degradation and very early focal cartilage degeneration is associated with upregulation of chondrocyte differentiation related genes in early human articular cartilage lesions. J Rheumatol. 2005;32(5):876–886. [PubMed] [Google Scholar]
- 118.Serra R, Karaplis A, Sohn P. Parathyroid Hormone–related Peptide (PTHrP)-dependent and -independent Effects of Transforming Growth Factor β (TGF-β) on Endochondral Bone Formation. J Cell Biol. 1999;145(4):783–794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Alvarez J, Sohn P, Zeng X, et al. Tgf-Beta2 mediates the effects of hedgehog on hypertrophic differentiation and PTHrP expression. Development. 2002;129:1913–1924. [DOI] [PubMed] [Google Scholar]
- 120.Breinan HA, Martin SD, Hsu H-. P, Spector M. Healing of canine articular cartilage defects treated with microfracture, a type-II collagen matrix, or cultured autologous chondrocytes. J Orthop Res. 2000;18(5):781–789. [DOI] [PubMed] [Google Scholar]
- 121.Pelttari K, Steck E, Richter W. The use of mesenchymal stem cells for chondrogenesis. Injury. 2008;39 Suppl 1:58–65. [DOI] [PubMed] [Google Scholar]
- 122.Lee CR, Grodzinsky AJ, Hsu H-. P, Martin SD, Spector M, et al. Effects of harvest and selected cartilage repair procedures on the physical and biochemical properties of articular cartilage in the canine knee. J Orthop Res. 2000;18(5):790–799. [DOI] [PubMed] [Google Scholar]
- 123.Vinatier C, Guicheux J. Cartilage tissue engineering: from biomaterials and stem cells to osteoarthritis treatments. Ann Phys Rehabil Med. 2016;59(3):139–144. [DOI] [PubMed] [Google Scholar]
- 124.Lee WY-W, Wang B. Cartilage repair by mesenchymal stem cells: Clinical trial update and perspectives. J Orthop Translat. 2017;9(12):76–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Zuk PA, Zhu M, Ashjian P, et al. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13(12):4279–4295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Lee HJ, Choi BH, Min B-H, Park SR. Changes in surface markers of human mesenchymal stem cells during the chondrogenic differentiation and dedifferentiation processes in vitro. Arthritis Rheum. 2009;60(8):2325–2332. [DOI] [PubMed] [Google Scholar]
- 127.Makhijani NS, Bischoff DS, Yamaguchi DT. Regulation of proliferation and migration in retinoic acid treated C3H10T1/2 cells by TGF-? isoforms. J Cell Physiol. 2005;202(1):304–313. [DOI] [PubMed] [Google Scholar]
- 128.Wang W-. G, Lou S-. Q, Ju X-. D, Xia K, Xia J-. H, et al. In vitro chondrogenesis of human bone marrow-derived mesenchymal progenitor cells in monolayer culture: activation by transfection with TGF-β2. Tissue and Cell. 2003;35(1):69–77. [DOI] [PubMed] [Google Scholar]
- 129.D’Arcy MS, D'arcy MS. Cell death: a review of the major forms of apoptosis, necrosis and autophagy. Cell Biol Int. 2019;43(6):582–592. [DOI] [PubMed] [Google Scholar]
- 130.Gibson G, Lin D-L, Wang X, Zhang L. The release and activation of transforming growth factor β2 associated with apoptosis of chick hypertrophic chondrocytes. J Bone Miner Res. 2001;16(12):2330–2338. [DOI] [PubMed] [Google Scholar]
- 131.Yu L, Hébert MC, Zhang YE. Tgf-Beta receptor-activated p38 MAP kinase mediates Smad-independent TGF-beta responses. Embo J. 2002;21(14):3749–3759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Yamashita M, Fatyol K, Jin C, Wang X, Liu Z, Zhang YE. Traf6 mediates Smad-independent activation of JNK and p38 by TGF-β. Mol Cell. 2008;31(6):918–924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Sharif M, Whitehouse A, Sharman P, Perry M, Adams M, et al. Increased apoptosis in human osteoarthritic cartilage corresponds to reduced cell density and expression of caspase-3. Arthritis Rheum. 2004;50(2):507–515. [DOI] [PubMed] [Google Scholar]
- 134.Thomas CM, Fuller CJ, Whittles CE, Sharif M. Chondrocyte death by apoptosis is associated with cartilage matrix degradation. Osteoarthritis and Cartilage. 2007;15(1):27–34. [DOI] [PubMed] [Google Scholar]
- 135.Bohensky J, Shapiro IM, Leshinsky S, Watanabe H, Srinivas V, et al. Pim-2 is an independent regulator of chondrocyte survival and autophagy in the epiphyseal growth plate. J Cell Physiol. 2007;213(1):246–251. [DOI] [PubMed] [Google Scholar]
- 136.Zhang C, Zhang X, Xu R, et al. TGF-β2 initiates autophagy via Smad and non-Smad pathway to promote glioma cells’ invasion. J Exp Clin Cancer Res. 2017;36(1):162. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 137.Wu J, Chen X, Liu X, et al. Autophagy regulates TGF-β2-induced epithelial-mesenchymal transition in human retinal pigment epithelium cells. Mol Med Rep. 2018;17:3607–3614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Dash S, Sarashetti PM, Rajashekar B, Chowdhury R, Mukherjee S, et al. TGF-β2-induced EMT is dampened by inhibition of autophagy and TNF-α treatment. Oncotarget. 2018;9(5):6433–6449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Chen H, Li J, Zhang D, Zhou X, Xie J, et al. Role of the fibroblast growth factor 19 in the skeletal system. Life Sci. 2021;265(Suppl. 2):118804. [DOI] [PubMed] [Google Scholar]
- 140.Kudipudi PK, Galuska SP, Dietze R, Scheiner-Bobis G, Loveland KL, Konrad L. Betaglycan (TβRIII) is a key factor in TGF-β2 signaling in prepubertal rat Sertoli cells. Int J Mol Sci. 2019;20(24):6214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Jia Y, Yue Y, D N H, et al. Human aqueous humor levels of transforming growth factor-β2: association with matrix metalloproteinases/tissue inhibitors of matrix metalloproteinases. Biomed Rep. 2017;7:573–578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Zhu Y, Gu J, Zhu T, Jin C, Hu X, Wang X. Crosstalk between Smad2/3 and specific isoforms of ERK in TGF-β1-induced TIMP-3 expression in rat chondrocytes. J Cell Mol Med. 2017;21(9):1781–1790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Yelin E, Weinstein S, King T. An update on the burden of musculoskeletal diseases in the U.S. Semin Arthritis Rheum. 2019;49(1):1–2. [DOI] [PubMed] [Google Scholar]
- 144.Felson DT. Clinical practice. osteoarthritis of the knee. N Engl J Med. 2006;354:841–848. [DOI] [PubMed] [Google Scholar]
- 145.Onuora S. Osteoarthritis: cartilage matrix stiffness regulates chondrocyte metabolism and OA pathogenesis. Nat Rev Rheumatol. 2015;11:504. [DOI] [PubMed] [Google Scholar]
- 146.Liu YP, Li J, Xin SB, X J. Study the relevance between inflammatory factors and estradiol and their association with knee osteoarthritis in postmenopausal women. Eur Rev Med Pharmacol Sci. 2018;22(2):472–478. [DOI] [PubMed] [Google Scholar]
- 147.Xie J, Zhang D, Lin Y, Yuan Q, Zhou X, et al. Anterior Cruciate Ligament Transection-Induced Cellular and Extracellular Events in Menisci: Implications for Osteoarthritis. Am J Sports Med. 2018;46(5):1185–1198. [DOI] [PubMed] [Google Scholar]
- 148.Wang J, Chen L, Jin S, et al. MiR-98 promotes chondrocyte apoptosis by decreasing Bcl-2 expression in a rat model of osteoarthritis. Acta Biochim Biophys Sin (Shanghai). 2016;48(10):923–929. [DOI] [PubMed] [Google Scholar]
- 149.Li J, Chen H, Zhang D, et al. The role of stromal cell-derived factor 1 on cartilage development and disease. Osteoarthritis Cartilage. 2020;20:31195-X:S1063–4584. [DOI] [PubMed] [Google Scholar]
- 150.Jiang J, Leong NL, Mung JC, Hidaka C, Lu HH, et al. Interaction between zonal populations of articular chondrocytes suppresses chondrocyte mineralization and this process is mediated by PTHrP. Osteoarthritis and Cartilage. 2008;16(1):70–82. [DOI] [PubMed] [Google Scholar]
- 151.Benjamin M, Mcgonagle D. The enthesis organ concept and its relevance to the spondyloarthropathies. Adv Exp Med Biol. 2009;649:57–70. [DOI] [PubMed] [Google Scholar]
- 152.Dwivedi P, Nayak V, Kowshik M. Role of gold nanoparticles as drug delivery vehicles for chondroitin sulfate in the treatment of osteoarthritis. Biotechnol Prog. 2015;31(5):1416–1422. [DOI] [PubMed] [Google Scholar]
- 153.Calvo E, Palacios I, Delgado E, et al. Histopathological correlation of cartilage swelling detected by magnetic resonance imaging in early experimental osteoarthritis. Osteoarthritis Cartilage. 2004;12(11):878–886. [DOI] [PubMed] [Google Scholar]
- 154.Loeser RF, Goldring SR, Scanzello CR, Goldring MB. Osteoarthritis: A disease of the joint as an organ. Arthritis Rheum. 2012;64(6):1697–1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Sandell LJ, Aigner T. Articular cartilage and changes in arthritis: cell biology of osteoarthritis. Arthritis Res Ther. 2001;3(2):107–113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Fukui N, Purple CR, Sandell LJ. Cell biology of osteoarthritis: The chondrocyte’s response to injury. Curr Rheumatol Rep. 2001;3(6):496–505. [DOI] [PubMed] [Google Scholar]
- 157.Singh P, Marcu KB, Goldring MB, Otero M. Phenotypic instability of chondrocytes in osteoarthritis: on a path to hypertrophy. Ann N Y Acad Sci. 2019;1442(1):17–34. [DOI] [PubMed] [Google Scholar]
- 158.Xia B, Di Chen, Zhang J, Hu S, Jin H, Tong P. Osteoarthritis pathogenesis: a review of molecular mechanisms. Calcif Tissue Int. 2014;95(6):495–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Xie J, Fu N, Cai L-Y, et al. The effects of interleukin-1β in modulating osteoclast-conditioned medium’s influence on gelatinases in chondrocytes through mitogen-activated protein kinases. Int J Oral Sci. 2015;7(4):220–231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Poole AR. What type of cartilage repair are we attempting to attain? J Bone Joint Surg Am. 2003;85-A:40–44. [DOI] [PubMed] [Google Scholar]
- 161.Elford PR, Graeber M, Ohtsu H, et al. Induction of swelling, synovial hyperplasia and cartilage proteoglycan loss upon intra-articular injection of transforming growth factor β-2 in the rabbit. Cytokine. 1992;4(3):232–238. [DOI] [PubMed] [Google Scholar]
- 162.Wipke BT, Wang Z, Nagengast W, Reichert DE, Allen PM, et al. Staging the initiation of autoantibody-induced arthritis: a critical role for immune complexes. J Immunol. 2004;172(12):7694–7702. [DOI] [PubMed] [Google Scholar]
- 163.Bevaart L, Vervoordeldonk MJ, Tak PP. Evaluation of therapeutic targets in animal models of arthritis: how does it relate to rheumatoid arthritis? Arthritis & Rheumatism. 2010;62(8):2192–2205. [DOI] [PubMed] [Google Scholar]
- 164.Joo YB, Park Y, Kim K, Bang S-Y, Bae S-C, Lee H-S. Association of CD8 + T-cells with bone erosion in patients with rheumatoid arthritis. Int J Rheum Dis. 2018;21(2):440–446. [DOI] [PubMed] [Google Scholar]
- 165.Bustamante MF, Garcia-Carbonell R, Whisenant KD, Guma M. Fibroblast-Like synoviocyte metabolism in the pathogenesis of rheumatoid arthritis. Arthritis Res Ther. 2017;19(1):110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Han E-J, Yoo S-A, Kim G-M, et al. GREM1 is a key regulator of synoviocyte hyperplasia and invasiveness. J Rheumatol. 2016;43(3):474–485. [DOI] [PubMed] [Google Scholar]
- 167.Chen C, Xie J, Rajappa R, Deng L, Fredberg J, Yang L. Interleukin-1 and tumor necrosis factor- increase stiffness and impair contractile function of articular chondrocytes. Acta Biochim Biophys Sin. 2015;47(2):121–129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Müssener A, Funa K, Kleinau S, Klareskog L. Dynamic expression of transforming growth factor-betas (TGF-beta) and their type I and type II receptors in the synovial tissue of arthritic rats. Clin Exp Immunol. 1997;107:112–119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Liu Y-Y, Wang S-Y, Li Y-N, et al. Activity of fibroblast-like synoviocytes in rheumatoid arthritis was impaired by Dickkopf-1 targeting siRNA. Chin Med J. 2020;51(6):679–686. [DOI] [PMC free article] [PubMed] [Google Scholar]