Abstract
Background
Bipolar disorder is a chronic mental disorder with repetitive mania/hypomania as well as depressive episodes, which eventually results in marked impairment in overall functioning and health‐related quality of life. A worldwide prevalence rate of 2.4% has been reported. The risk of suicide is higher in people with bipolar disorder than those with other mental disorders. Therefore, effective management of bipolar disorder in the maintenance period is warranted to minimize the risk of relapse or recurrence. Although lithium has been the standard treatment of bipolar disorder for many years, it is associated with adverse effects and teratogenicity. Lamotrigine is approved to be expected for prevention of recurrence for the maintenance treatment of bipolar disorder. In addition, lamotrigine is as effective as lithium. Therefore, we performed a systematic review to confirm the efficacy and safety of lamotrigine in the maintenance treatment of bipolar disorder.
Objectives
To assess the efficacy and tolerability of lamotrigine in the maintenance treatment of bipolar disorder.
Search methods
We searched Ovid MEDLINE, Embase, PsycINFO, the Cochrane Common Mental Disorders Group's Specialized Register (CCMDCTR) and the Cochrane Central Register of Controlled Trials (CENTRAL) from inception to 21 May 2021. We also searched international trial registries and contacted experts in the field.
Selection criteria
We included randomized controlled trials enrolling adults with bipolar disorder who were treated with lamotrigine, placebo or lithium.
Data collection and analysis
Two reviews authors independently checked the eligibility of studies and extracted data using a standardized form. Data extracted included study characteristics, participant characteristics, intervention details, settings, and outcome measures in the term of efficacy and tolerability. Study information were then entered into RevMan web.
Main results
We included 11 studies with a total of 2314 participants in this review; 1146 were randomized to lamotrigine, 869 were randomized to placebo and, 299 to lithium.
We rated all studies as having an unclear risk of bias in at least one domain of Cochrane's tool for assessing risk of bias, with the most commonly observed weakness being selection bias (random sequence generation and allocation concealment). We judged five studies to be at a high risk of detection bias (blinding of outcome assessment). These potential biases pose as major threat to the validity of the included studies in this review.
Outcomes of efficacy showed a possible advantage of lamotrigine over placebo. The estimated risk ratio (RR) for recurrence of manic symptom at one year as measured by the Young Mania Rating Scale (YMRS) was 0.67, (95% confidence interval (CI) 0.51 to 0.87; 3 studies, 663 participants; low‐certainty evidence) in favor of lamotrigine. The RR of clinical worsening with the need for additional psychotropic treatment (RR 0.82, 95% CI 0.70 to 0.98; 4 studies, 756 participants) based on moderate‐certainty evidence. The possible benefits of lamotrigine were also seen for the outcome of treatment withdrawal due to any reason at 6‐12 months after treatment (RR 0.88, 95% CI 0.78 to 0.99; 4 studies, 700 participants; moderate‐certainty evidence). Regarding tolerability, our analyses showed that the incidence rates of adverse effects were similar between the lamotrigine group and the placebo group (short‐term effect: RR 1.07, 95% CI 0.81 to 1.42; 5 studies, 1138 participants; very low‐certainty evidence; long‐term effect: RR 0.97, 95% CI 0.77 to 1.23; 4 studies, 756 participants; moderate‐certainty evidence).
In the comparison between lamotrigine and lithium, efficacy was similar between groups except for recurrence of mania episode at one year. Recurrence of manic symptoms was higher in the lamotrigine group than that of the lithium group (RR 2.13, 95% CI 1.32 to 3.44; 3 studies, 602 participants; moderate‐certainty evidence). Analysis of adverse effects at 6‐12 months showed that a lower proportion of participants experienced at least one adverse effect when treated with lamotrigine compared to lithium (RR 0.70, 95% CI 0.51 to 0.96; 4 studies, 691 participants; moderate‐certainty evidence).
Authors' conclusions
Low‐ to moderate‐certainty evidence collectively suggests that lamotrigine may be superior to placebo as a treatment modality for bipolar disorder. In comparison to lithium, people with bipolar disorder seem to tolerate lamotrigine better in the long run; however, the demonstrated efficacy in the maintenance of bipolar disorder was similar between the two groups.
Keywords: Adult, Humans, Anticonvulsants, Anticonvulsants/therapeutic use, Bipolar Disorder, Bipolar Disorder/drug therapy, Lamotrigine, Lamotrigine/adverse effects, Quality of Life, Randomized Controlled Trials as Topic
Plain language summary
Lamotrigine as a treatment for prevention of recurrence of bipolar disorder
Review question
To investigate the clinical effectiveness (benefits and harms) of lamotrigine for maintenance therapy of bipolar disorder in comparison with placebo, combination therapy or existing drugs (e.g. lithium, olanzapine).
Background
Lamotrigine is approved for the maintenance treatment (treatment for prevention of recurrence) of bipolar disorder. It could be a viable and effective treatment strategy for the maintenance of bipolar disorder, where it demonstrated a lower risk of recurrence than placebo. In addition, it has been reported to be as effective as lithium. Therefore, we performed a systematic review to investigate the efficacy and safety of lamotrigine in the maintenance treatment (treatment for prevention of recurrence) of bipolar disorder.
Key findings
The evidence is current to May 2021. We included 11 studies in this review with a total 2314 participants. 1146 participants were assigned to lamotrigine, and 1168 participants were assigned to control intervention (869 received placebo and 299 received lithium).
Our review identified the following.
Lamotrigine versus placebo
Benefits: lamotrigine was found to be superior over placebo in the following outcomes.
1) Reduced the rate of recurrence of manic symptoms
2) Suppressed depressive symptoms
3) Less need for additional therapeutic agents for the recurrence of all symptoms
4) Reduction in the withdrawal rate due to any causes six months or more after the initiating of intervention
Harms: adverse‐event profile of lamotrigine was similar to that of placebo.
Lamotrigine versus lithium
Benefits: lamotrigine was as effective as lithium, except for the recurrence of manic symptoms, for which the rate was higher in people who received lamotrigine than people treated with lithium.
Harms: the rate of adverse events associated with lamotrigine was lower than that of lithium.
Certainty of the evidence
We assessed the certainty of our included evidence to be very low to moderate. This is because majority of the included trials did not describe how treatment allocation was concealed. Given that we were unable to identify high‐confidence evidence the overall findings of this systematic review should be interpreted carefully.
Conclusions
Lamotrigine may be superior to placebo in efficacy and may be comparable in safety. And then, lamotrigine was as effective as lithium except for its recurrence rate of mania, and was superior to lithium in terms of safety. Future studies in this field with robust methods and transparent reporting are needed to confirm our results and to fully answer our pre‐specified review question.
Summary of findings
Summary of findings 1. Lamotrigine versus placebo for the maintenance treatment of bipolar disorder.
| Lamotrigine versus placebo in the maintenance treatment of bipolar disorder | ||||||
| Patient or population: bipolar disorder Setting: hospital outpatients Intervention: lamotrigine Comparison: placebo | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with placebo | Risk with lamotrigine | |||||
| Recurrence of any episodes at one year (hospitalization for any episodes) | Study population | ‐ | (0 studies) | ‐ | The outcome was not reported in any studies. | |
| see comment | see comment | |||||
| Recurrence of any episodes at one year (Young Mania Rating Scale (YMRS) total score ≥15 for manic episode) | Study population | RR 0.67 (0.51 to 0.87) | 663 (3 RCTs) | ⊕⊕⊝⊝ LOW 1 2 3 | 2 studies reported that no recurrence of any episodes for one year Young Mania Rating Scale total score 15 or greater for manic episode was recorded in the lamotrigine and control groups. | |
| 217 per 1,000 | 87 per 1,000 (48 to 159) | |||||
| Recurrence of any episodes at one year (Montgomery‐Asberg Depression Rating Scale (MADRS) total score ≥15 for depressive episode; Hamilton Depression Rating Scale (HDRS) total score ≥14 for depressive episode) | Study population | RR 0.85 (0.70 to 1.02) | 1606 (7 RCTs) | ⊕⊕⊝⊝ LOW 1 4 5 | ||
| 516 per 1,000 | 438 per 1,000 (340 to 536) | |||||
| Recurrence of any episodes at one year for clinical worsening with additional psychotropics (mood stabilizers, antidepressants, antipsychotics or benzodiazepines) | Study population | RR 0.82 (0.70 to 0.98) | 756 (4 RCTs) | ⊕⊕⊕⊝ MODERATE 1 6 | ||
| 595 per 1,000 | 500 per 1,000 (428 to 577) | |||||
| Recurrence of any episodes at one year (active suicidal behavior) | Study population | not estimable | (1 study) | ‐ | The outcome was measured in one study, but no numeric data were provided. | |
| 0 per 1,000 | 0 per 1,000 (0 to 0) | |||||
| Withdrawal from treatment due to any reason, up to 12 weeks after intervention commencement (short term) | Study population | RR 1.10 (0.70 to 1.74) | 195 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 1 7 8 | ||
| 288 per 1,000 | 317 per 1,000 (196 to 472) | |||||
| Withdrawal from treatment due to any reason, 6‐12 months after intervention initiation (long term) | Study population | RR 0.88 (0.78 to 0.99) | 700 (4 RCTs) | ⊕⊕⊕⊝ MODERATE 1 6 | ||
| 686 per 1,000 | 529 per 1,000 (426 to 625) | |||||
| Any reported adverse effects up to 12 weeks after starting treatment (short term) | Study population | RR 1.07 (0.81 to 1.42) | 1138 (5 RCTs) | ⊕⊝⊝⊝ VERY LOW 1 9 10 | ||
| 239 per 1,000 | 256 per 1,000 (208 to 311) | |||||
| Any reported adverse effects 6‐12 months after initiating the intervention (long term) | Study population | RR 0.97 (0.77 to 1.23) | 756 (4 RCTs) | ⊕⊕⊕⊝ MODERATE 1 6 | ||
| 536 per 1,000 | 514 per 1,000 (428 to 594) | |||||
| Recurrence of manic episode at one year | Study population | RR 0.91 (0.66 to 1.26) | 574 (3 RCTs) | ⊕⊕⊕⊝ MODERATE 1 11 | ||
| 229 per 1,000 | 211 per 1,000 (149 to 288) | |||||
| Recurrence of depressive episode at one year | Study population | RR 0.75 (0.53 to 1.05) | 574 (3 RCTs) | ⊕⊕⊕⊝ MODERATE 1 11 | ||
| 382 per 1,000 | 294 per 1,000 (225 to 374) | |||||
| Quality of life as measured by the 36‐Item Short Form Health Survey | ‐ | see comment | ‐ | (0 study) | ‐ | The outcome was not reported in any studies. |
| Total severity score from depression and mania symptom scores, such as HDRS and the YMRS | ‐ | MD 0 (0 to 0 ) | ‐ | (0 study) | ‐ | The outcome was not reported in any studies. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk ratio; | ||||||
| High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect. | ||||||
1 Publication bias was not evaluated due to limited data.
2 Downgraded by one level for risk of bias. Calabrese 1999, Calabrese 2000 and Calabrese 2003 had unclear to high risk of detection bias (blinding of outcome assessment).
3 Downgraded by one level. Only Calabrese 2000 provided OR and 95% CI.
4 Downgraded by one level for inconsistency (I2= 60%).
5 Downgraded by one level for risk of bias. Calabrese 1999, Calabrese 2000, Calabrese 2003 and Calabrese 2008 had unclear to high risk of detection bias (blinding of outcome assessors).
6 Downgraded by one level for risk of bias. Bowden 2003, Calabrese 2000, Calabrese 2003 and Koyama 2011 had unclear to high risk of detection bias (blinding of outcome assessment).
7 Downgraded by one level for risk of bias (only Calabrese 1999 was included with unclear risk of bias).
8 Downgraded by one level. Only Calabrese 1999 provided OR and 95% CI.
9 Downgraded by one level for risk of bias. Calabrese 1999 and Calabrese 2008 had unclear to high risk of detection bias (blinding of outcome assessment).
10 Downgraded by two levels for very serious inconsistency (I2= 82%)
11 Downgraded by one level for risk of bias. Bowden 2003, Calabrese 2003 and Koyama 2011 had unclear to high risk of detection bias (blinding of outcome assessment).
Summary of findings 2. Lamotrigine versus lithium for the maintenance treatment of bipolar disorder.
| Lamotrigine versus lithium in the maintenance treatment of bipolar disorder | ||||||
| Patient or population: bipolar disorder Setting: hospital outpatients Intervention: lamotrigine Comparison: lithium | ||||||
| Outcomes | Anticipated absolute effects* (95% CI) | Relative effect (95% CI) | № of participants (studies) | Certainty of the evidence (GRADE) | Comments | |
| Risk with lithium | Risk with lamotrigine | |||||
| Recurrence of any episodes at one year (hospitalization for any episodes) | Study population | ‐ | (0 studies) | ‐ | The outcome was not reported in any studies. | |
| see comment | see comment | |||||
| Recurrence of any episodes at one year (Young Mania Rating Scale (YMRS) total score ≥15 for manic episode) | Study population | RR 3.57 (0.15 to 85.39) | 376 (2 RCTs) | ⊕⊝⊝⊝ VERY LOW 1 2 3 | ||
| 0 per 1,000 | 0 per 1,000 (0 to 0) | |||||
| Recurrence of any episodes at one year (Montgomery‐Asberg Depression Rating Scale (MADRS) total score ≥15 for depressive episode; Hamilton Depression Rating Scale (HDRS) total score ≥14 for depressive episode) | Study population | RR 1.40 (0.70 to 2.79) | 376 (2 RCTs) | ⊕⊕⊝⊝ LOW 1 3 4 | ||
| 65 per 1,000 | 92 per 1,000 (44 to 179) | |||||
| Recurrence of any episodes at one year for clinical worsening with additional psychotropics (mood stabilizers, antidepressants, antipsychotics or benzodiazepines) | Study population | RR 1.11 (0.92 to 1.35) | 602 (3 RCTs) | ⊕⊕⊕⊝ MODERATE 3 5 | ||
| 408 per 1,000 | 461 per 1,000 (380 to 543) | |||||
| Recurrence of any episodes at one year (active suicidal behavior) | Study population | RR 1.01 (0.06 to 15.91) | 155 (1 RCT) | ⊕⊝⊝⊝ VERY LOW 2 3 6 | ||
| 13 per 1,000 | 13 per 1,000 (1 to 176) | |||||
| Withdrawal from treatment due to any reason, up to 12 weeks after intervention commencement (short term) | Study population | ‐ | (0 studies) | ‐ | The outcome was not reported in any studies. | |
| see comment | see comment | |||||
| Withdrawal from treatment due to any reason, 6‐12 months after intervention initiation (long term) | Study population | RR 0.96 (0.88 to 1.05) | 636 (4 RCTs) | ⊕⊕⊕⊝ MODERATE 3 7 | ||
| 663 per 1,000 | 590 per 1,000 (491 to 677) | |||||
| Any reported adverse effects up to 12 weeks after starting treatment (short term) | Study population | ‐ | (0 studies) | ‐ | The outcome was not reported in any studies. | |
| see comment | see comment | |||||
| Any reported adverse effects 6‐12 months after initiating the intervention (long term) | Study population | RR 0.70 (0.51 to 0.96) | 691 (4 RCTs) | ⊕⊕⊕⊝ MODERATE 3 7 | ||
| 382 per 1,000 | 226 per 1,000 (164 to 294) | |||||
| Recurrence of manic episode at one year | Study population | RR 2.13 (1.32 to 3.44) | 602 (3 RCTs) | ⊕⊕⊕⊝ MODERATE 3 7 | ||
| 102 per 1,000 | 206 per 1,000 (136 to 298) | |||||
| Recurrence of depressive episode at one year | Study population | RR 0.83 (0.63 to 1.09) | 602 (3 RCTs) | ⊕⊕⊕⊝ MODERATE 3 7 | ||
| 355 per 1,000 | 295 per 1,000 (227 to 373) | |||||
| Quality of life as measured by the 36‐Item Short Form Health Survey | ‐ | see comment | ‐ | (0 studies) | ‐ | The outcome was not reported in any studies. |
| Total severity score from depression and mania symptom scores, such as HDRS and the YMRS | ‐ | see comment | ‐ | (0 studies) | ‐ | The outcome was not reported in any studies. |
| *The risk in the intervention group (and its 95% confidence interval) is based on the assumed risk in the comparison group and the relative effect of the intervention (and its 95% CI). CI: Confidence interval; RR: Risk ratio; | ||||||
| High certainty: we are very confident that the true effect lies close to that of the estimate of the effect. Moderate certainty: we are moderately confident in the effect estimate; the true effect is likely to be close to the estimate of the effect, but there is a possibility that it is substantially different. Low certainty: our confidence in the effect estimate is limited; the true effect may be substantially different from the estimate of the effect. Very low certainty: we have very little confidence in the effect estimate; the true effect is likely to be substantially different from the estimate of effect. | ||||||
1 Downgraded by one level for risk of bias (only Suppes 2008a was included).
2 Downgraded by two levels due to wide CIs.
3 Publication bias was not evaluated due to limited data.
4 Downgraded by one level due to wide CIs.
5 Downgraded by one level for risk of bias. Bowden 2003 and Calabrese 2003 were judged to be at high risk of detection bias (blinding of outcome assessment).
6 Downgraded by one level for risk of bias (only Licht 2010 was included).
7 Downgraded by one level for risk of bias. Calabrese 2003 and Licht 2010 were judged to be at high risk of detection bias (blinding of outcome assessment).
Background
Description of the condition
Bipolar disorder is a chronic mental disorder with repetitive cycles of mania/hypomania as well as depressive episodes, which eventually results in marked impairment in overall functioning and health‐related quality of life (de Hert 2011). It is described by the American Psychiatric Association's Diagnostic and Statistical Manual of Mental Disorders (DSM‐5) tool as a type of mental health condition that leads to extreme fluctuation in a person's mood, energy, and ability to function (American Psychiatric Association 2013). Bipolar disorder was the 46th greatest causes of disability‐adjusted life years (DALYs) in the world, placing it ahead of Alzheimer's disease and other dementias among the 291 disorders included in the Global Burden of Disease Study 2010 (Murray 2012), and it has a worldwide prevalence of 2.4% (Merikangas 2011). Bipolar disorder affects approximately 60 million people worldwide (WHO 2019a). The risk of suicide is higher in people with bipolar disorder than those with other mental disorders (Nordentoft 2011). The estimated rate of death by suicide was 0.2 people per year to 0.4 people per year among people with bipolar disorder (Sondergard 2008), and one‐year recurrence rate was 26.3% (Vazquez 2015). Therefore, in addition to treating recurrent mania/hypomania and depressive episodes, effective management of bipolar disorder after acute treatment of mood episodes using long‐term continuation therapy is warranted in order to minimize the risk of relapse or recurrence (Calabrese 2006).
Description of the intervention
For many years, lithium has been the standard treatment of bipolar disorder with acute mood episodes, polarity change prevention, prophylactic treatment, and suicide prevention (BALANCE investigators and collaborators 2010; Smith 2007; Yildiz 2011). However, questions were raised over the potential overestimation of the effectiveness of lithium and the frequency of use of lithium for bipolar disorder is decreasing (Blanco 2002; Lyall 2019). Furthermore, lithium has been associated with congenital malformations during pregnancy for the fetus (Poels 2018). It has been reported that rates of recurrence after lithium discontinuation sharply increased postpartum (Viguera 2000). Sodium valproate and carbamazepine, both antiepileptic drugs, are effective for the management of the bipolar mania and acute symptoms (Bowden 2005; Greil 1998); however, they are relatively less effective against depression of maintenance phase and depression of bipolar disorder (Ng 2007). Atypical antipsychotics including olanzapine (Berk 1999b), quetiapine (Calabrese 2005; Ketter 2007), aripiprazole (Keck 2003), and risperidone (Segal 1998) have also demonstrated efficacy in the treatment of mania. For the new anticonvulsants such as gabapentin (Vieta 2006), topiramate (Pigott 2016), zonisamide (Dauphinais 2011), and levetiracetam (Kaufman 2004), sufficient efficacy has not been demonstrated for bipolar disorder. Thus, there remains a need to identify additional pharmacological agents with sufficient and up‐to‐date evidence to demonstrate their effectiveness in preventing and managing relapse of bipolar disorder.
Lamotrigine is approved for the maintenance treatment of adults with bipolar disorder. Lamotrigine is indicated for use in numerous clinical guidelines as a first‐line pharmacological agent in the treatment of bipolar depression (Nivoli 2011). Lamotrigine is licensed by the US Food and Drug Administration (FDA) for the prevention of relapses in people with bipolar disorder. Although the evidence base regarding long‐term efficacy of lamotrigine is reasonably robust, five pivotal trials conducted in acute‐phase therapy reported relatively neutral findings and they found no statistically significant benefits (Calabrese 2008). Previous studies found that lamotrigine could be a viable and effective treatment modality for the maintenance of bipolar disorder, where it illustrated a lower risk of recurrence than placebo (Geddes 2009; Miura 2014; Oya 2019). It has also been reported to be equally effective as lithium, which is the standard treatment for bipolar disorder (Suppes 2008b). In addition, lamotrigine demonstrated a better safety profile compared to lithium in postpartum teratogenicity (Graham 2018), although available evidence from a population‐based cohort study found that it was equivalent to lithium in the prevention of (hypo) manic or depressive episodes (Wesseloo 2017). It is worth highlighting that lamotrigine has been proposed as a treatment option for bipolar disorder during pregnancy among mothers who are at risk of depression. For example, Kong 2018 indicated that lamotrigine could be a safe mood stabilizer for use during pregnancy based on available clinical evidence; Veroniki 2017 conducted a systematic review on the safety of antiepileptic drugs in pregnancy which included 96 (n=58,461 patients) studies, most were cohort studies. The risk of congenital malformations with lamotrigine was comparable to placebo (OR, 0.96; 95% CI, 0.72–1.25). and less than with other antiepileptic drugs (ethosuximide (OR, 3.04; 95% CI, 1.23–7.07), valproate (OR, 2.93; 95% CI, 2.36–3.69), topiramate (OR, 1.90; 95% CI, 1.17–2.97), phenobarbital (OR, 1.83; 95% CI, 1.35–2.47), phenytoin (OR, 1.67; 95% CI, 1.30–2.17), carbamazepine (OR, 1.37; 95% CI, 1.10–1.71).
How the intervention might work
Lamotrigine is an antiepileptic drug belonging to the phenyltriazine class used in the treatment of epilepsy and bipolar disorder. Lamotrigine works by inhibiting voltage‐sensitive sodium channels, stabilizing presynaptic neuronal membranes and inhibiting glutamate release (Verrotti 2018). The proposed mechanisms of action to explain the treatment of lamotrigine for bipolar disorder include inhibiting voltage‐sensitive sodium channels, glutamate release and calcium channel blockade (Andreazza 2014). To reduce the risk of life‐threatening skin rashes, lamotrigine needs to be titrated slowly. Thus, lamotrigine is more effective as maintenance therapy than in the acute treatment of bipolar disorder (Bobo 2017; Calabrese 2008). Although the effects of lamotrigine on pregnancy have been reported in a previous systematic review, there was no association with birth defects or related disorders (Pariente 2017). Lamotrigine has been shown to demonstrate more predictable pharmacokinetics than other antiepileptic drugs such as carbamazepine and valproic acid or valproate, which have a pronounced interindividual variability in their pharmacokinetics and a narrow therapeutic range (Johannessen 2006). As with other antiepileptic drugs, there are demonstrable effects of metabolic enzymes but oral bioavailability is almost 100%, with negligible influence from diets (Garnett 1997). Consequently, treatment of lamotrigine is associated with less burden in blood sampling/therapeutic monitoring inflicted on people with bipolar disorder.
Why it is important to do this review
Reviews exploring the effectiveness of lamotrigine are available (Bowden 2012b; Yatham 2018). However, these existing reviews considered non‐maintenance therapies as part of their scope, and the numbers of included studies and study participants were small. Consequently, we planned to conduct this Cochrane Review to synthesize the latest available evidence in order to provide a comprehensive update on the effectiveness of lamotrigine for maintenance treatment of bipolar disorder. Although lithium is used as the first‐line drug for the treatment of bipolar disorder, results from randomized studies on the comparative efficacy of lamotrigine versus lithium are inconsistent and thus we hoped to review and synthesize evidence using rigorous and systematic methods. We examined the effectiveness of lamotrigine against placebo as well as combination therapy and existing medications.
Objectives
To determine efficacy and safety of lamotrigine for the maintenance treatment of manic, depressive, and mixed episodes of bipolar disorder.
Methods
Criteria for considering studies for this review
Types of studies
We included individually‐randomized controlled trials. Cluster‐randomized trials were eligible for inclusion but none were identified. If studies employing a cross‐over design had been identified and included, we would only have used only data from the first active treatment (i.e. first randomized phase before crossing over). Studies published as full texts or only as abstracts as well as unpublished data were eligible for inclusion.
Types of participants
We included people with bipolar I or II disorder in remission, with a diagnosis based on the International Classification of Diseases 11th Revision (ICD‐11) coding system (WHO 2019b), or the Diagnostic and Statistical Manual of Mental Disorders Fifth Edition (DSM‐5) tool (or previous versions of these diagnostic manuals) (American Psychiatric Association 2013). Study participants were aged 18 years or older of either gender with concurrent primary diagnosis of Axis I or Axis II disorder. We included participants with any co‐morbidities except for those with dementia and personality disorder or cyclothymia, which was defined as a disorder not meeting the requirements to be classified as a major episode of hypomanic and depressive state.
Types of interventions
We included studies comparing lamotrigine with usual care, placebo or no treatment; the daily dosage of lamotrigine maintenance treatment was defined to range from 100 mg to 500 mg with a treatment duration of more than 12 weeks.
We were interested in the following treatment comparisons:
lamotrigine versus no treatment;
lamotrigine versus placebo;
lamotrigine versus lithium;
lamotrigine versus valproic acid or valproate (or both);
lamotrigine versus olanzapine;
lamotrigine versus quetiapine;
lamotrigine plus lithium versus lithium;
lamotrigine plus lithium versus lamotrigine;
lamotrigine plus valproic acid or valproate (or both) versus valproic acid or valproate (or both);
lamotrigine plus valproic acid or valproate (or both) versus lamotrigine;
lamotrigine plus olanzapine versus olanzapine;
lamotrigine plus olanzapine versus lamotrigine;
lamotrigine plus quetiapine versus quetiapine;
lamotrigine plus quetiapine versus lamotrigine.
Types of outcome measures
Timing of outcome assessment
We anticipated that authors of studies would report response rates at various time points during and post‐intervention. Therefore, we subdivided the timing of outcome assessment as follows:
short‐term effects, within 12 weeks after treatment initiation;
long‐term effects, at 6 to 12 months after treatment initiation.
Hierarchy of outcome measures
If several measures were available for each outcome, we used results from the Hamilton Depression Rating Scale (HDRS). If HDRS results were not available, we used results from the Montgomery‐Asberg Depression Rating Scale (MADRS) for depressive episode as the primary outcome. However, if outcomes were measured by other rating scales, we included and extracted these results with explanations on the components of the scale used by the respective included studies.
Primary outcomes
Our primary outcome measures of interests were as follows.
-
Recurrence of any episode at one year:
hospitalization for any mood episodes;
Young Mania Rating Scale (YMRS) total score ≥15 for manic episode (Young 1978);
Montgomery‐Asberg Depression Rating Scale (MADRS) total score ≥ 15 for depressive episode (Montgomery 1979); Hamilton Depression Rating Scale (HDRS) total score ≥ 14 for depressive episode (Hamilton 1960);
clinical worsening with the need for additional treatment (e.g. mood stabilizers, antidepressants, antipsychotics or benzodiazepines);
active suicidal behavior.
-
Withdrawal from treatment due to any reason:
short term, up to 12 weeks after treatment initiation (range: 7 to 16 weeks);
long term, at least six months after treatment initiation (range: 6 to 16 months).
-
Adverse effects:
short term, up to 12 weeks after treatment initiation (range: 7 to 16 weeks);
long term, at 6 to 12 months after treatment initiation (range: 6 to 16 months).
Secondary outcomes
We included the following secondary outcome measures:
recurrence of manic episode at one year (based on assessor's judgement without using an evaluation scale);
recurrence of depressive episode at one year (based on assessor's judgement without using an evaluation scale) ;
quality of life as measured by the mental component summary of the 36‐Item Short Form Health Survey (SF‐36) (Ware 1993);
total severity score calculated from adding depression and manic symptom scores, such as the HDRS and the YMRS (Hamilton 1960; Young 1978).
For timing of outcome assessment, we anticipated that authors of studies would report response rates at various time points during and post‐intervention. Therefore, we subdivided the timing of outcome assessment as follows:
short term effects, up to 12 weeks after treatment initiation (range: 7 to 16 weeks);
long term effects, at least six months after treatment initiation (range: 6 to 16 months).
If several measures were available for each outcome, we used results from the HDRS. If HDRS results were unavailable, we used results from MADRS. However, if outcomes were measured by other rating scales not listed above, we included and extracted the relevant results with explanations on the components of the scale used by the respective included studies.
Search methods for identification of studies
Electronic searches
An information specialist from the Cochrane Common Mental Disorders Group searched the following databases on 21 May 2021 using relevant keywords, subject headings (controlled vocabularies) and search syntax, appropriate to each resource (Appendix 1):
Cochrane Central Register of Controlled Trials (CENTRAL), (Issue 5 of 12, 2021) on the Cochrane Library;
Cochrane Database of Systematic Reviews (CDSR) (Issue 5 of 12, 2021) on the Cochrane Library;
Ovid MEDLINE (1946 to May 21, 2021);
Ovid Embase (1974 to 2021 Week 21);
Ovid PsycINFO (to May Week 3 2021).
Cochrane Common Mental Disorders Controlled Trials Register (CCMDCTR) (all years to June 2016) (Appendix 2).
We searched ClinicalTrials.gov (clinicaltrials.gov/), and the World Health Organization (WHO) International Clinical Trials Registry Platform (ICTRP) Search Portal (apps.who.int/trialsearch/), to identify unpublished or ongoing studies.
We did not impose any restrictions on date, language or publication status to the searches.
Searching other resources
We screened reference lists of relevant narrative reviews and included studies for further relevant information. For information on unpublished data or ongoing studies, we attempted to contact the study investigators or content experts in the field (or both) in an attempt to identify additional trial data.
Data collection and analysis
Selection of studies
Two review authors (YH and KK) independently screened titles and abstracts from the systematic literature search and excluded clearly irrelevant records. For any disagreements, we consulted other review authors (TF or SS, or both). We identified and excluded duplicates and collated multiple reports of the same study so that each study, not each reference, was the unit of interest in the review.
We retrieved full‐text versions of all 'to include' or 'unclear' records for further assessment against our pre‐defined eligibility criteria. We attempted to obtain translations of articles that were published in languages other than English or Japanese. For conference abstracts, we attempted to retrieve relevant subsequent full‐text publication or contacted the study investigators for clarification. Two review authors (YH and KK) independently screened the full texts against the pre‐specified inclusion/exclusion criteria. We resolved disagreements by discussion or by consulting other review authors (TF or SS, or both)
Data extraction and management
Two review authors (YH and KK) independently extracted the following information from included studies using a pre‐standardized data collection form.
Methods: study design, total duration of study, methods of randomization, methods of allocation concealment, withdrawals.
Participants: number, mean age, age range, gender, inclusion and exclusion criteria, diagnosis (bipolar disorder I type or type II), baseline comparability between two groups, severity of condition, mean scores on HDRS, or MADRS, or any other episode scale at baseline and end of studies, time from onset and losses to follow‐up.
Interventions: intervention, comparison, concomitant medications and excluded medications.
Outcomes: primary and secondary outcome data and characteristics.
Others: setting, publication year, sources of funding, intention‐to‐treat (ITT) analysis.
We resolved any disagreements regarding the extracted study information by discussion or by consulting other review authors (TF or SS, or both).
Assessment of risk of bias in included studies
Two review authors (YH and KK) independently assessed risk of bias in included studies using Cochrane's tool for assessing risk of bias as indicated in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2020). We resolved disagreements by discussion or by consulting other review authors (TF or SS, or both).
We assessed risk of bias in included studies according to the following seven domains:
random sequence generation;
allocation concealment;
blinding of participants and personnel;
blinding of outcome assessment;
incomplete outcome data;
selective outcome reporting;
other bias.
We judged each potential source of bias as 'high risk', 'low risk' or 'unclear risk', and provided a supporting quotation from the study report together with a justification for our judgements in the risk of bias table. We summarized our judgements across different studies for each domain.
Measures of treatment effect
If the included studies reported continuous data such as total severity scores using the same measurement scale, we planned to synthesize the effect measures using mean differences (MDs) with 95% confidence intervals (CIs); had studies used different measurement tools, we planned to calculate the standardized mean differences (SMDs). For dichotomous data such as the number of reported adverse effects, we calculated the risk ratios (RRs) with 95% CIs.
Unit of analysis issues
Cross‐over studies
One concern of cross‐over trials is the carry‐over effect, where the anticipated and unprecedented pharmacological, physiological and psychological effects of the study treatment intervention in the first phase may affect the results of the second phase of the study. As a result, in the second phase, participants can differ systematically from their initial state, even after a washout period.
For the purpose of this Cochrane Review, had we included cross‐over studies, we would have considered only results from the first phase, i.e. prior to crossing over.
Cluster‐randomized trials
For cluster‐randomized trials, we planned to make an adjustment to the sample size for each intervention based on the method described in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2020), using an estimate of the intraclass correlation coefficient (ICC) derived from the study (where available) or from a similar study or from a study of a similar population. We intended to conduct a sensitivity analysis to explore the effects of variation in ICCs and the overall robustness of our findings (Sensitivity analysis). Impact of studies for which no ICC was reported on the overall review findings would have been explored in a sensitivity analysis (Sensitivity analysis).
Studies with multiple treatment groups
Where a relevant study involved more than two treatments groups, we included data from the additional arms for comparisons. If the data were binary, we combined them in a 2 × 2 table. If data were continuous, we combined them using the formula provided in the Cochrane Handbook for Systematic Reviews of Interventions (Higgins 2020).
Dealing with missing data
We recorded missing data for each included study and attempted to contact the study investigators for the missing information. Where possible, we performed all meta‐analyses using an intention‐to‐treat (ITT) approach i.e. we analyzed all participants and their outcomes within the groups to which they were originally allocated, regardless of whether they received the intervention.
Assessment of heterogeneity
We assessed heterogeneity by visually inspecting the calculated effect estimates and CIs in the forest plots. We used the Chi2 test (statistical significance at P < 0.1) and the I2 statistic to investigate and quantify statistical heterogeneity in each meta‐analysis. We interpreted the I2 statistic as follows (Higgins 2020):
0% to 40% might not be important;
30% to 60% may represent moderate heterogeneity;
50% to 90% may represent substantial heterogeneity;
75% to 100% represents considerable heterogeneity.
Assessment of reporting biases
Reporting biases arise when dissemination of research findings is often influenced by the nature and direction of the results (Higgins 2020). We planned to conduct a funnel plot to investigate publication bias through visual inspection of asymmetry should sufficient evidence be available (10 or more included studies) for each outcome, also planned to perform a statistical test for funnel plot asymmetry as proposed by Egger and Rücker (Egger 1997; Rücker 2008).
Data synthesis
One review author (KK) entered data into the RevMan web, and a second review author (YH) checked the entries. Due to variations in participants, interventions or outcomes, we used a random‐effects meta‐analysis to produce an overall summary.
Subgroup analysis and investigation of heterogeneity
We planned to perform the following four subgroup analyses for our Primary outcomes should data be sufficient:
with or without mental disorder co‐morbidities;
duration of treatment (up to six months and longer than six months);
setting (community versus hospital);
for each pharmacological modality such as mood stabilizer, antipsychotic and antidepressant.
Sensitivity analysis
We planned to conduct a sensitivity analysis to examine the impact of the ICCs (Unit of analysis issues) mentioned above.
In addition, sensitivity analyses were also planned under the following conditions to determine if the risk area of bias would affect the overall robustness of the findings:
excluding studies with inadequate allocation concealment and random sequence generation;
excluding studies in which outcome evaluation was not blinded;
excluding studies in which loss to follow‐up was not reported or was greater than 10%;
excluding studies funded by the pharmaceutical company marketing lamotrigine.
Summary of findings and assessment of the certainty of the evidence
We prepared a summary of findings table based on the GRADE assessment system using the GRADEpro GDT software for each of the main comparisons, considering the two most clinically important primary outcomes (Guyatt 2011). Two review authors (YH and KK) independently graded the body of evidence using adapted decision rules. We explored the following five domains:
risk of bias;
inconsistency;
indirectness;
imprecision;
and publication bias.
We graded the overall strength of evidence for each outcome as 'high', 'moderate', 'low' or 'very low', and resolved any disagreement through discussion or by consulting a third author (TF or NW).
We included the following outcomes in the summary of findings tables.
-
Recurrence of any episode at one year:
hospitalization for any mood episodes;
YMRS total score ≥15 for manic episode;
MADRS total score ≥15 for depressive episode; and HDRS total score ≥14 for depressive episode;
clinical worsening with the need for addition of a mood stabilizer, antidepressant treatment, antipsychotic medication or benzodiazepine; or
active suicidal behavior.
-
Withdrawal from treatment due to any reason:
short term, up to 12 weeks after treatment initiation (range: 7 to 16 weeks);
long term, at least six months after treatment initiation (range: 6 to 16 months).
Results
Description of studies
Lamotrigine in the maintenance treatment of bipolar disorder
Results of the search
Searches of the Cochrane Library (CENTRAL and CDSR), MEDLINE, Embase, PsycINFO, and CCMDCTR databases yielded a total of 1708 records (updated to include searches up to May 2021). Koyama 2011 written in Japanese was not retrieved by searching these electrical database, but from screening of the reference list of Oya 2019. Of those initial 1709 studies, we identified 142 studies as potentially eligible for inclusion after title and abstract screening. We retrieved full‐text articles for these 142 studies for full inspection, of which 134 were excluded and finally, 11 studies (eight papers) fulfilled our inclusion criteria and were included in the review. Our study selection process is illustrated in Figure 1 (Moher 2009).
1.
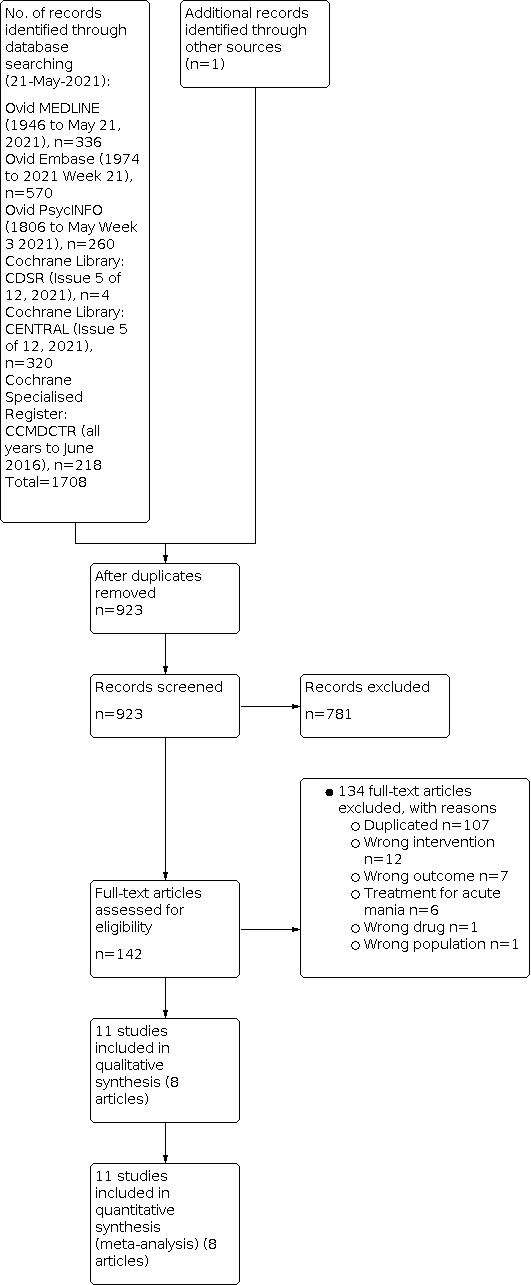
PRISMA flow diagram.
Included studies
Please refer to Characteristics of included studies for further information.
We included 11 studies in this review (Bowden 2003; Calabrese 1999; Calabrese 2000; Calabrese 2003; Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]; Koyama 2011; Licht 2010; Suppes 2008a). We identified one publication which reported findings from five RCTs; of these, one (GW602/SCAB2001) was the duplication of Calabrese 1999. Therefore, this was excluded and the remaining four studies were included in this review (Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]). Please note we have listed these trial names as per the original publication.
Study design and settings
All included studies were multicenter randomized trials; all but one study applied double‐blind methodology (Suppes 2008a). We did not identify any cluster‐randomized studies.
Study duration
The length of study ranged from seven to 302 weeks: Calabrese 1999, seven weeks; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910], eight weeks; Calabrese 2008 [GW603/SCAA2010], 10 weeks. Treatment duration ranged from 16 weeks to 5.8 years: Suppes 2008a, 16 weeks; Koyama 2011, 26 weeks; Calabrese 2000, six months; Bowden 2003 and Calabrese 2003, 76 weeks; and in Licht 2010 treatment continued for 5.8 years. Our analysis was stratified according to the study period (short term and long term). We defined study duration of seven to 16 weeks as "short term" and duration of over six months as "long term".
Sample sizes
The 11 included studies involved a total 2314 participants: 1146 were randomized to lamotrigine, 869 were randomized to placebo and 299 to lithium.
Bowden 2003, Calabrese 2003, Licht 2010, and Koyama 2011 were the only studies that reported sample size calculations.
Participants
Of the 11 included studies, participants were diagnosed with bipolar I disorder in seven studies (Bowden 2003; Calabrese 1999; Calabrese 2003; Koyama 2011; Licht 2010; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]). Participants from Calabrese 2000, and Calabrese 2008 [GW603/SCAA2010] were diagnosed with bipolar I as well as bipolar II disorders. Two studies included patients with bipolar II disorder (Calabrese 2008 [SCA100223]; Suppes 2008a).
In the open‐label phase, 38.5% of participants were treated with lamotrigine prior to double‐blinded randomization phase (Bowden 2003; Calabrese 2000; Calabrese 2003; Koyama 2011); in Calabrese 2008 [SCA40910], the proportion of pre‐randomization use of lamotrigine was 53.9%. No description of medications used prior to randomization was available in Calabrese 1999, Calabrese 2008 [GW603/SCAA2010], Licht 2010, Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]. Only Suppes 2008a was a single‐blind randomized study. Therefore, it was an open‐label study throughout the study duration (4.1%).
Participants included in each of the 11 included studies were hospital outpatients of both genders; in six studies, participants were aged at least 18 years old (Bowden 2003; Calabrese 2003; Calabrese 1999; Calabrese 2000; Licht 2010; Suppes 2008a). The inclusion criterion for Koyama 2011 was limited to adults aged 20 years or above (Koyama 2011). In the remaining four studies, no description of specific age range/cut‐offs were provided besides describing their participants as “adults” (Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]).
All the study participants were diagnosed with bipolar disorder according to Diagnostic and Statistical Manual of Mental Disorders (DSM) criteria (DSM‐IV, DSM‐IV‐TR), assessed by a variety of instruments including the Structured Clinical Interview for DSM Disorders (SCID) as illustrated in Calabrese 1999 and Suppes 2008a.
Interventions and comparators
Types of comparisons were as follows: seven studies comparing lamotrigine with placebo (Calabrese 1999; Calabrese 2000; Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]; Koyama 2011), two investigated lamotrigine versus lithium (Licht 2010; Suppes 2008a), and two were three‐arm studies comparing lamotrigine with lithium as well as placebo (Bowden 2003; Calabrese 2003).
During the open‐label phase, the dosing schedule of lamotrigine used in Suppes 2008a was as follows: weeks 1 to 2, 25 mg/day; weeks 3 to 4, 50 mg/day; week 5, 75 mg/day; week 6, 100 mg/day; week 7, 150 mg/day; and week 8, 200 mg/day. In the other included studies, lamotrigine dose was escalated to reach a target dose as follows: weeks to ‐2, 25 mg/day; weeks 3 to 4, 50 mg/day; week 5, 10 0mg/day; and week 6, 200 mg/day. After six weeks, the daily dose was adjusted depending on tolerability, with a flexible dosing regimen from 100 mg to 500 mg daily in the maintenance phase.
Outcomes
The 11 included studies reported the following outcome measures.
Recurrence of manic episodes at one year measured by the Young Mania Rating Scale (YMRS) (Suppes 2008a) and Mania Rating Scale (MRS) (Calabrese 1999; Calabrese 2000; Calabrese 2003).
Recurrent of depressive episodes at one year by the Hamilton Depression Rating Scale (HDRS) (Calabrese 1999; Calabrese 2000; Calabrese 2003; Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]; Suppes 2008a).
Addition of any psychotropics for recurrence of any episodes at one year (Bowden 2003; Calabrese 2003; Calabrese 2000; Koyama 2011; Licht 2010).
Active suicidal behavior (Licht 2010).
Withdrawal from treatment by any reason up to 12 weeks after the intervention i.e. short‐term withdrawal (Calabrese 1999).
Withdrawal from treatment due to any reason at six‐ to12 months after the intervention i.e. long‐term withdrawal (Bowden 2003; Calabrese 2000; Calabrese 2003; Koyama 2011; Licht 2010; Suppes 2008a).
Any reported adverse effects (number of participants who experienced at least one adverse effect) up to 12 weeks after the intervention i.e. short‐term safety (Calabrese 1999; Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]).
Any reported adverse effects (number of participants who experienced at least one adverse effect) six to 12 months after the intervention i.e. long‐term safety (Bowden 2003; Calabrese 2003; Calabrese 2000; Koyama 2011; Licht 2010; Suppes 2008a).
Recurrence of manic episode (Bowden 2003; Calabrese 2003; Koyama 2011; Licht 2010).
Recurrence of depressive episode (Bowden 2003; Calabrese 2003; Koyama 2011; Licht 2010).
None of the included studies reported data on quality of life or satisfaction with treatment outcomes.
Excluded studies
We excluded 27 studies from this review (see Figure 1). The most common reason of exclusion was inappropriate study inclusion criteria. Other reasons were wrong type(s) of intervention(s), wrong outcome measures, wrong study designs (e.g. non‐randomized controlled trials (RCTs)), and the lack of relevant data (Characteristics of excluded studies).
Risk of bias in included studies
Findings of our assessment of risk of bias in included studies are illustrated in Figure 2 and Figure 3 for the specific judgments. Further details and justification of our judgments are presented in the Characteristics of included studies section.
2.

Figure 2. Risk of bias graph: review authors’ judgements about each risk of bias item presented as percentages across all included studies.
3.

Figure 3. Risk of bias table
Allocation
Nine studies did not report details on the sequence generation and were judged as having an unclear risk of bias (Bowden 2003; Calabrese 1999; Calabrese 2000; Calabrese 2003; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]; Koyama 2011; Suppes 2008a). Two studies provided sufficient information on sequence generation (Calabrese 2008 [GW603/SCAA2010]; Licht 2010).
Eight studies failed to provide details on how allocation was concealed and were thus judged as having an unclear risk of bias (Bowden 2003; Calabrese 1999; Calabrese 2000; Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]; Koyama 2011). Two studies provided sufficient information on allocation concealment and we judged them as having low risk of bias (Calabrese 2003; Licht 2010). Suppes 2008a was described as an open‐label study and thus we classified it as high risk of bias.
Blinding
Blinding of participants and personnel (performance bias)
We classified nine studies included in the review as being at low risk of performance bias, as participants and personal were explicitly described as blinded in the study report (Bowden 2003; Calabrese 1999; Calabrese 2000; Calabrese 2003; Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]; Koyama 2011). We classified one trial as being unclear because it provided no information on blinding participants or personnel (Licht 2010). Suppes 2008a was described as an open‐label study and thus we classified it as high risk of bias.
Blinding of outcome assessors (detection bias)
We rated one trial as having a low risk of detection bias (Suppes 2008a); five trials as having a high risk of detection bias (Calabrese 2000; Calabrese 2003; Calabrese 2008 [SCA30924] Koyama 2011; Licht 2010). The remaining trials did not provide details on the blinding of outcome assessors and thus were judged to be of unclear risk of bias (Bowden 2003; Calabrese 1999; Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA40910]).
Incomplete outcome data
We rated six trials to be adequate in terms of addressing incomplete outcome data (Bowden 2003; Calabrese 1999; Calabrese 2000; Calabrese 2003; Licht 2010; Suppes 2008a). One trial had unclear risk of attrition bias (Koyama 2011), and the remaining four trials were assessed to be at high risk of bias since they lacked clear description of study methodology (Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]).
Selective reporting
On the whole, study authors diligently reported study data and thus we assessed all included studies to be at low risk of reporting bias (Bowden 2003; Calabrese 1999; Calabrese 2000; Calabrese 2003; Calabrese 2008 [GW603/SCAA2010]; Koyama 2011; Licht 2010; Suppes 2008a; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]).
Other potential sources of bias
We did not identify other potential sources of bias amongst the 11 included studies.
Effects of interventions
Comparison 1: Lamotrigine versus no treatment
See Table 1.
Primary outcomes
Hospitalization for any mood episode
No trial measured this outcome.
Young Mania Rating Scale (YMRS) total score ≥ 15 for manic episode
Three trials provided data on the YMRS total score ≥ 15 for manic episode, enrolling a total of 663 participants. But only one study contributed to the meta‐analysis because the other two studies had zero events. We found a difference between lamotrigine and placebo (RR 0.67, 95% CI 0.51 to 0.87; P = 0.003) in favor of lamotrigine; low‐certainty evidence (Analysis 1.1).
1.1. Analysis.

Comparison 1: Lamotrigine versus placebo, Outcome 1: Recurrence of any episodes at one year (Young Mania Rating Scale total score ≥15 for manic episode)
Montgomery‐Asberg Depression Rating Scale (MADRS) total score ≥ 15 depressive episode; Hamilton Depression Rating Scale (HDRS) total score ≥ 14 for depressive episode
Seven trials provided data on the MADRS total score ≥ 15 for depressive episodes or HDRS total score ≥ 14 for depressive episodes, including a total of 1606 participants. There was no difference between lamotrigine and placebo (RR 0.85, 95% CI 0.70 to 1.02; P = 0.08; low‐certainty evidence Analysis 1.2), with substantial heterogeneity (I2 = 71%; P = 0.002).
1.2. Analysis.

Comparison 1: Lamotrigine versus placebo, Outcome 2: Recurrence of any episodes at one year (Montgomery‐Asberg Depression Rating Scale total score ≥15 for depressive episode; Hamilton Depression Rating Scale total score ≥14 for depressive episode)
Clinical worsening with the need for additional treatment (mood stabilizers, antidepressants, antipsychotics or benzodiazepines)
Four trials (n=756) provided data on clinical worsening with the need of additional treatment. We found a difference between lamotrigine and placebo (RR 0.82, 95% CI 0.70 to 0.98; P = 0.03) in favor of lamotrigine; moderate‐certainty evidence (Analysis 1.3). Heterogeneity was found to be not important (I2 = 31%; P = 0.23).
1.3. Analysis.

Comparison 1: Lamotrigine versus placebo, Outcome 3: Recurrence of any episodes at one year for clinical worsening with additional psychotropics (mood stabilizers, antidepressants, antipsychotics or benzodiazepines)
Active suicidal behavior
None of the included trials measured this a priori outcome.
Withdrawal from treatment due to any reason (short term)
Only one trial of 195 participants provided data on treatment withdrawal due to any reason (short term), and we found no difference between lamotrigine and placebo (RR 1.10, 95% CI 0.70 to 1.74; P = 0.67; very low‐certainty evidence Analysis 1.4).
1.4. Analysis.

Comparison 1: Lamotrigine versus placebo, Outcome 4: Withdrawal from treatment due to any reason, up to 12 weeks after intervention commencement (short‐term)
Withdrawal from treatment due to any reason (long term)
Four trials of 700 participants assessed treatment withdrawal due to any reason (long term). Treatment of lamotrigine was associated with a reduced incidence of treatment withdrawal as compared to placebo (RR 0.88, 95% CI 0.78 to 0.99; P = 0.03; moderate‐certainty evidence Analysis 1.5), with moderate heterogeneity (I2 = 37%; P = 0.19).
1.5. Analysis.

Comparison 1: Lamotrigine versus placebo, Outcome 5: Withdrawal from treatment due to any reason, 6‐12 months after intervention initiation (long‐term)
Adverse effects (short term)
Five trials reported adverse effects (short term), enrolling a total of 1138 participants. There was no difference between lamotrigine and placebo (RR 1.07, 95% CI 0.81 to 1.42; P = 0.61; very low‐certainty evidence Analysis 1.6); heterogeneity was moderate (I2 = 42%; P = 0.14).
1.6. Analysis.

Comparison 1: Lamotrigine versus placebo, Outcome 6: Adverse effects up to 12 weeks after starting treatment (short‐term)
Adverse effects (long term)
Four trials provided data on adverse effects (long term), including a total of 756 participants. We found no difference between the two groups (RR 0.97, 95% CI 0.77 to 1.23; P = 0.83; moderate‐certainty evidence Analysis 1.7), with moderate heterogeneity (I2 = 45%; P = 0.14).
1.7. Analysis.

Comparison 1: Lamotrigine versus placebo, Outcome 7: Adverse effects 6‐12 months after initiating the intervention (long‐term)
Secondary outcomes
Recurrence of manic episode at one year
Three trials of 574 participants assessed recurrence of manic episode. We found no difference between lamotrigine and placebo (RR 0.91, 95% CI 0.66 to 1.26; P = 0.58; moderate‐certainty evidence Analysis 1.8). We found no heterogeneity (I2 = 0%; P = 0.59).
1.8. Analysis.

Comparison 1: Lamotrigine versus placebo, Outcome 8: Recurrence of manic episode at one year
Recurrence of depressive episode at one year
Three trials provided data on the recurrence of depressive episode, enrolling a total of 574 participants. We found a difference between lamotrigine and placebo (RR 0.75, 95% CI 0.53 to 1.05; P = 0.09) in favor of lamotrigine moderate‐certainty evidence (Analysis 1.9). Heterogeneity was moderate for this outcome (I2 = 37%; P = 0.21).
1.9. Analysis.

Comparison 1: Lamotrigine versus placebo, Outcome 9: Recurrence of depressive episode at one year
Quality of life
No trial measured this outcome.
Total severity score
No trial measured this outcome.
Comparison 2: Lamotrigine versus lithium
See Table 2.
Primary outcomes
Hospitalization for any mood episode
No trial measured this outcome.
Young Mania Rating Scale (YMRS) total score ≥ 15 for manic episode
Two trials of 376 participants provided data on the YMRS total score 15 or greater for manic episode and there was no difference between the two groups. (RR 3.57, 95% CI 0.15 to 85.39; P = 0.43; very low‐certainty evidence Analysis 2.1). Heterogeneity was not applicable.
2.1. Analysis.

Comparison 2: Lamotrigine versus lithium, Outcome 1: Recurrence of any episodes at one year (Young Mania Rating Scale total score ≥15 for manic episode)
Montgomery‐Asberg Depression Rating Scale (MADRS) total score ≥ 15 depressive episode; Hamilton Depression Rating Scale (HDRS) total score ≥ 14 for depressive episode
Two trials reported the MADRS total score ≥ 15 for depressive episode or HDRS total score ≥14 for depressive episode, including a total of 376 participants. We found no difference between lamotrigine and placebo (RR 1.40, 95% CI 0.70 to 2.79; P = 0.34; low‐certainty evidence Analysis 2.2). Heterogeneity was not applicable.
2.2. Analysis.

Comparison 2: Lamotrigine versus lithium, Outcome 2: Recurrence of any episodes at one year (Montgomery‐Asberg Depression Rating Scale total score ≥15 for depressive episode; Hamilton Depression Rating Scale total score ≥14 for depressive episode)
Clinical worsening with the need for additional treatment (mood stabilizers, antidepressants, antipsychotics or benzodiazepines)
Three trials of 602 participants assessed clinical worsening with the use of additional treatment. There was no difference between lamotrigine and lithium (RR 1.11, 95% CI 0.92 to 1.35; P = 0.28; moderate‐certainty evidence Analysis 2.3). We found no important heterogeneity (I2 = 0%; P = 0.73).
2.3. Analysis.

Comparison 2: Lamotrigine versus lithium, Outcome 3: Recurrence of any episodes at one year for clinical worsening with additional psychotropics (mood stabilizers, antidepressants, antipsychotics or benzodiazepines)
Active suicidal behavior
We identified just one trial (n = 155) with data on active suicidal behavior. There was no difference between lamotrigine and placebo (RR 1.01, 95% CI 0.06 to 15.91; P = 0.99; very low‐certainty evidence Analysis 2.4).
2.4. Analysis.

Comparison 2: Lamotrigine versus lithium, Outcome 4: Recurrence of any episodes at one year (active suicidal behavior)
Withdrawal from treatment due to any reason (short term)
None the included trials measured this pre‐specified outcome.
Withdrawal from treatment due to any reason (long term)
Four trials provided data on treatment withdrawal due to any reason (long term), including a total of 636 participants. No difference between groups was observed (RR 0.96, 95% CI 0.88 to 1.05; P = 0.34; moderate‐certainty evidence Analysis 2.5); heterogeneity was found to be minimal (I2 = 4%; P = 0.37).
2.5. Analysis.

Comparison 2: Lamotrigine versus lithium, Outcome 5: Withdrawal from treatment due to any reason, 6‐12 months after intervention initiation (long‐term)
Adverse effects (short term)
None of the included trial measured this outcome.
Adverse effects (long term)
Four trials assessed long‐term adverse effect amongst 691 participants. We found a difference between lamotrigine and lithium (RR 0.70, 95% CI 0.51 to 0.96; P = 0.02) in favor of lamotrigine moderate‐certainty evidence (Analysis 2.6). Heterogeneity was found to be moderate (I2 = 37%; P = 0.19).
2.6. Analysis.

Comparison 2: Lamotrigine versus lithium, Outcome 6: Adverse effects 6‐12 months after initiating the intervention (long‐term)
Secondary outcomes
Recurrence of manic episode at one year
Three trials provided data on the recurrence of manic episode, including a total of 602 participants. We noted a higher incidence of recurrent manic episode with lamotrigine versus lithium (RR 2.13, 95% CI 1.32 to 3.44; P = 0.002; moderate‐certainty evidence Analysis 2.7), with no heterogeneity (I2 = 0%; P = 0.43).
2.7. Analysis.

Comparison 2: Lamotrigine versus lithium, Outcome 7: Recurrence of manic episode at one year
Recurrence of depressive episode at one year
Three trials (602 participants) assessed the recurrence of depressive episode. There was no difference between the two groups (RR 0.83, 95% CI 0.63 to 1.09; P = 0.18; moderate‐certainty evidence Analysis 2.8). We found no important heterogeneity (I2 = 2%; P = 0.36).
2.8. Analysis.

Comparison 2: Lamotrigine versus lithium, Outcome 8: Recurrence of depressive episode at one year
Quality of life
None of the included trials measured this outcome.
Total severity score
None of the included trials measured this a priori outcome.
Discussion
Summary of main results
Our review included 11 studies involving 2314 participants. We found low‐certainty evidence supporting the use of lamotrigine over placebo for people with bipolar disorder. Lamotrigine was found to be more effective than placebo for minimizing recurrence of bipolar depression at one year. Moderate‐certainty evidence indicated that lamotrigine a similar safety profile compared to placebo. Treatment withdrawal at 6 to 12 months was more frequent amongst participants in the placebo groups when compared with the lamotrigine groups.
Compared to lithium, we found low‐certainty evidence indicating that lamotrigine was comparable to lithium in the outcomes of bipolar disorder symptoms except for recurrence of bipolar mania. Current evidence also found that lamotrigine increased incidence of exacerbated bipolar manic symptoms when compared to lithium. In addition, adverse events experienced by participants treated with lamotrigine were lower than those reported in the lithium groups.
Overall completeness and applicability of evidence
We conducted a comprehensive and systematic literature search to identify all available published and unpublished studies fulfilling our pre‐specified inclusion criteria, and 11 studies were eventually included. Of these, seven studies compared lamotrigine with placebo, two were three‐arm studies that investigated the effects of lamotrigine versus lithium versus placebo, and the remaining two studies compared lamotrigine with lithium.
Overall, all the included studies reported our efficacy outcomes of interest. We performed data conversion (from continuous to dichotomous data) using indicated average, standard deviation, and sample numbers (Bowden 2003; Calabrese 1999; Calabrese 2000; Calabrese 2003; Calabrese 2008 [GW603/SCAA2010]; Calabrese 2008 [SCA100223]; Calabrese 2008 [SCA30924]; Calabrese 2008 [SCA40910]; Suppes 2008a). As a result, we were able to synthesize evidence for the various efficacy outcomes in the comparisons of lamotrigine versus placebo and lamotrigine versus lithium. None of the included studies assessed hospitalization for any mood episode, quality of life, or total change score Young Mania Rating Scale (YMRS) plus: Hamilton Depression Rating Scale (HDRS)). Reporting of adverse events was generally adequate, which allowed for evaluation of safety outcomes.
All our studies enrolled adults with clinically‐diagnosed bipolar disorder (as confirmed by: Diagnostic and Statistical Manual of Mental Disorders, Fourth edition (DSM‐IV)) and were conducted in the outpatient setting from several countries.
In this review we attempted to ensure the highest possible level of certainty of the evidence, we excluded non‐randomized studies or randomized controlled trials (RCTs) that did not use a standard diagnostic process. Our review findings showed that lamotrigine was more effective than placebo on recurrence of bipolar disorder, and the incidence of adverse events was comparable between groups. Furthermore, lamotrigine was found to be more tolerable than lithium, which has been the standard treatment approach in clinical practice. The key to treatment during the maintenance phase of bipolar disorder is continuity of treatment. Considering these facts, we would like to highlight that lamotrigine poses as a viable treatment option.
Quality of the evidence
We assessed the certainty of the evidence collected using the GRADE approach, which takes into consideration five domains: risk of bias, inconsistency, indirectness, imprecision and publication bias. Elaboration of our GRADE assessment for each outcome is illustrated in Table 1 and Table 2.
Overall GRADE assessment
Lamotrigine versus placebo: evidence on efficacy outcomes was generally of low to moderate certainty. Overall, the number of included studies was small and thus we could not assess publication bias. Included evidence regarding adverse effects of lamotrigine was judged to be of very low certainty in the short‐term duration and of moderate certainty in the long term.
Lamotrigine versus lithium: certainty of evidence on efficacy outcomes was moderate. Only limited study findings were available and we decided to not investigate publication bias. Certainty of evidence on the comparative adverse effect profiles of lamotrigine was assessed to be moderate.
Risk of bias
We rated all included studies as having an unclear risk of bias in at least one domain of the Cochrane Collaboration’s tool for assessing risk of bias (Higgins 2020), with the most commonly observed weakness being selection bias (random sequence generation and allocation concealment). We judged seven of the nine included studies as having a high risk of bias in at least one domain; among these, six studies showed a high risk of detection bias (blinding of outcome assessment). These potential biases pose a major threat to the validity of the review findings.
Inconsistency
Substantial statistical heterogeneity across studies was noted in the outcome of recurrence of any episodes at one year (MADRS total score ≥15 for depressive episode or HDRS total score ≥14 for depressive episode) for the comparison of lamotrigine versus placebo, which limited the reliability of the included evidence and we downgraded the level of certainty accordingly.
Indirectness
Overall, the available evidence matched well with our pre‐defined clinical questions and review scope. The included study subjects were all hospital outpatients during the maintenance phase of bipolar I or II disorder.
Imprecision
The precision of our outcome estimates was significantly hampered by several factors, such as very wide confidence intervals and small size of some of studies.
Publication bias
We did not assess publication bias using a funnel plot as none of the analyses included more than 10 studies.
Potential biases in the review process
We conducted an extensive search for randomized studies meeting our pre‐defined eligibility criteria. Nevertheless, we found insufficient data available to fully answer our review questions and numerous outcomes had only a small number of included studies. As a result, we were unable to conduct subgroup analyses and sensitivity analyses as planned at the protocol stage; however, we did not find any sources of bias that might be expected to affect the study results. On the other hand, of the 11 studies we included, eight were funded by the same sponsor and thus we cannot rule out the potential of industry sponsorship bias, which could lead to an overestimation of interventions.
Agreements and disagreements with other studies or reviews
Our results are consistent with the results of four previous systematic reviews (Beynon 2009; Miura 2014; Oya 2019; Smith 2007). Lamotrigine was shown to be superior over placebo in reducing relapse of any mood episode. Although lamotrigine showed no benefits in bipolar manic episode, lamotrigine was more effective than placebo in reducing bipolar depressive episode (Beynon 2009; Smith 2007). As for adverse effects, lamotrigine was found to be better tolerated than lithium (Miura 2014; Smith 2007).
Authors' conclusions
Implications for practice.
Findings from our review showed a potential superiority of lamotrigine over placebo in terms of recurrence prevention and treatment continuation. The incidence of adverse effects was similar between lamotrigine and placebo regardless of the duration of the studies. Therefore, lamotrigine is considered to be sufficiently useful in the maintenance treatment for bipolar disorder. In comparison with lithium, change in rating scale mania as well as depression, addition of therapeutic intervention for any mood episodes, treatment continuation, and recurrence of depressive episode for one year were similar in lamotrigine. Although lamotrigine worsened the recurrence of mania episode for one year, tolerability was superior to lamotrigine for lithium. These results are consistent with current guidelines for treatment of bipolar disorder (Yatham 2018). In the pharmacotherapy of bipolar disorder, the use of antipsychotic agents as well as mood stabilizers has been increased in recent years. However, these are primarily treatments for manic symptoms of bipolar disorder; there is no robust treatment strategy for depressive symptoms during the maintenance phase. Lithium, which has been used as standard first‐line treatment, is associated with severe adverse effects and thus prone to treatment discontinuation and non‐compliance. Furthermore, in Japan, lithium is contraindicated for pregnant and lactating women. In this review, we could not comprehensively evaluate the safety profile of lamotrigine for pregnant and lactating women due to insufficient data. Nonetheless, our findings on relapse prevention by lamotrigine suggests that it is a viable maintenance treatment option for people with bipolar disorder.
Implications for research.
The overall low‐ to moderate‐certainty evidence is indicative of the need to design and execute robust, large‐scale randomized studies of lamotrigine amongst people with bipolar disorder, with particular considerations on the methodological shortcomings in randomization method, allocation concealment and blinding of outcome assessment as identified in our review. For effective clinical decision‐making, results of our present review will contribute to an ongoing network meta‐analysis of drug treatments for people with bipolar disorder, which aims to provide a solid evidence base in regards to efficacy and safety of the wide array of drug treatment options available for bipolar disorder.
We initially planned to explore the efficacy and safety profile of lamotrigine (as monotherapy or in combination with other pharmacological agents) in pregnant and lactating women, and assessing the quality of life or satisfaction amongst treated individuals. However, we did not identify eligible studies involving pregnant and lactating women and none of the studies assessed quality of life or patient satisfaction. These are important knowledge gaps and their evidence is critical for effective clinical practice management, in particular to enhance treatment safety and compliance. We urge study investigators in the field to continue, and should be systematically reviewed.
History
Protocol first published: Issue 4, 2020
Acknowledgements
We thank the editorial team of the Cochrane Common Mental Disorders Group (CCMD) for providing guidance.
The review authors and the CCMD Editorial Team are grateful to the following peer reviewers for their time and comments: Marianna Purgato, S Rees, and Lindsay Robertson. They would also like to thank Cochrane Japan for language support and Cochrane Copy Edit Support.
This project was supported by the National Institute for Health Research, via Cochrane Infrastructure funding to the Cochrane Common Mental Disorders Review Group. The views and opinions expressed therein are those of the authors and do not necessarily reflect those of the Evidence Synthesis Programme, the NIHR, NHS or the Department of Health and Social Care.
Appendices
Appendix 1. Database search strategies
The following databases were searched on 21 May 2021.
Ovid MEDLINE (1946 to May 21, 2021), n = 336.
Ovid Embase (1974 to 2021 Week 21), n = 570.
Ovid PsycINFO (1806 to May Week 5 2021), n = 260.
Cochrane Library: CDSR (Issue 3 of 12, 2021), n = 4.
Cochrane Library: CENTRAL (Issue 5 of 12, 2021), n = 320.
Cochrane Specialised Register: CCMDCTR (all years to June 2016), n = 218.
Total = 1670; duplicates removed=769; records screened = 901.
Ovid MEDLINE(R) and Epub Ahead of Print, In‐Process & Other Non‐Indexed Citations and Daily <1946 to May 21, 2021> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 Lamotrigine/ (3037) 2 lamotrigin*.mp. (5626) 3 1 or 2 (5626) 4 "bipolar and related disorders"/ or bipolar disorder/ (39841) 5 (mania or manic or hypomani*).mp. (19374) 6 affective psychosis.mp. (914) 7 (rapid cycling or schizoaffective).mp. (6868) 8 (psychos* or psychotic or anti‐psycho* or antipsycho*).ti,kf. (94530) 9 or/4‐8 (138653) 10 3 and 9 (911) 11 controlled clinical trial.pt. (93602) 12 randomized controlled trial.pt. (503313) 13 (randomi#ed or randomi#ation or randomi#ing).ti,ab,kf. (627562) 14 (RCT or "at random" or (random* adj3 (administ* or allocat* or assign* or class* or control* or determine* or divide* or division or distribut* or expose* or fashion or number* or place* or recruit* or split or subsitut* or treat*))).ti,ab,kf. (524211) 15 placebo*.ab,ti,kf. (214313) 16 trial.ab,ti,kf. (591673) 17 groups.ab. (2030691) 18 (control* and (trial or study or group*) and (placebo or waitlist* or wait* list* or ((treatment or care) adj2 usual))).ti,ab,kf,hw. (196791) 19 ((single or double or triple or treble) adj2 (blind* or mask* or dummy)).ti,ab,kf. (171880) 20 double‐blind method/ or random allocation/ or single‐blind method/ (276541) 21 or/11‐20 (3007719) 22 exp animals/ not humans.sh. (4686331) 23 21 not 22 (2551396) 24 10 and 23 (322) *************************** Embase <1974 to 2021 Week 21> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 Lamotrigin*.ti,ot,ab,kw. (8545) 2 *Lamotrigine/ (4000) 3 1 or 2 (9104) 4 exp bipolar disorder/ or exp mania/ (73118) 5 (bipolar or mania* or manic* or hypomani* or rapid cycling or schizoaffective or affective psychosis).ti,ot,ab,kw. (111321) 6 4 or 5 (131534) 7 3 and 6 (1723) 8 randomized controlled trial/ (597675) 9 randomization.de. (86485) 10 controlled clinical trial/ and Drug Therapy.fs. (200577) 11 phase 2 clinical trial/ or phase 3 clinical trial/ (119767) 12 placebo/ (348394) 13 placebo.ti,ab. (303480) 14 trial.ti. (295884) 15 (randomi#ed or randomi#ation or randomi#ing).ti,ab,kw. (901687) 16 (RCT or "at random" or (random* adj3 (administ* or allocat* or assign* or class* or control* or determine* or divide* or division or distribut* or expose* or fashion or number* or place* or recruit* or split or subsitut* or treat*))).ti,ab,kw. (720109) 17 double blind procedure/ or single blind procedure/ or triple blind procedure/ (207863) 18 ((singl* or doubl* or trebl* or tripl*) adj3 (blind* or mask* or dummy)).mp. (305491) 19 (control* and (trial or study or group?) and (waitlist* or wait* list* or ((treatment or care) adj2 usual))).ti,ab,kw,hw. (39340) 20 or/8‐19 (1727289) 21 ((animal or nonhuman) not human).de. (5649578) 22 20 not 21 (1571827) 23 7 and 22 (554) *************************** APA PsycInfo <1806 to May Week 3 2021> Search Strategy: ‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐ 1 lamotrigin*.mp. (2030) 2 exp bipolar disorder/ (30063) 3 mania/ or hypomania/ (6149) 4 (bipolar or mania* or manic* or hypomani* or rapid cycling or schizoaffective or affective psychosis).ti,ot,ab,id. (56480) 5 or/2‐4 (56803) 6 1 and 5 (829) 7 clinical trials.sh. (11603) 8 (randomi#ed or randomi#ation or randomi#ing).ti,ab,id. (84626) 9 (RCT or at random or (random* adj3 (administ* or allocat* or assign* or class* or control* or crossover or cross‐over or determine* or divide* or division or distribut* or expose* or fashion or number* or place* or recruit* or split or subsitut* or treat*))).ti,ab,id. (100841) 10 (control* and (trial or study or group) and (placebo or waitlist* or wait* list* or ((treatment or care) adj2 usual))).ti,ab,id,hw. (28729) 11 ((single or double or triple or treble) adj2 (blind* or mask* or dummy)).ti,ab,id. (25971) 12 trial.ti. (29835) 13 placebo.ti,ab,id,hw. (39858) 14 treatment outcome.md. (20242) 15 treatment effectiveness evaluation.sh. (24046) 16 mental health program evaluation.sh. (2110) 17 or/7‐16 (193390) 18 6 and 17 (257) *************************** The Cochrane Library, Issue 5 of 12, 2021 (lamotrigin* and (bipolar or mania or manic or hypomani* or “affective psychosis” or “rapid cycling” or schizoaffective)):ti,ab,kw *************************** Cochrane Common Mental Disorders Controlled Trials Register (CCMDCTR) (lamotrigin* and (bipolar or mania or manic or hypomani* or “affective psychosis” or “rapid cycling” or schizoaffective)) [all fields] ***************************
Appendix 2. Description of the CCMDCTR
Cochrane Common Mental Disorders Controlled Trials Register (CCMDCTR)
The Cochrane Common Mental Disorders Group (CCMD) retains two clinical trials registers at its editorial base (current to June 2016); a References Register and a studies‐based register. The CCMDCTR‐References Register contains over 40,000 reports of randomised controlled trials (RCTs) in disorders of anxiety, bipolar, depression, eating and psychological trauma, together with self‐harm. Approximately half of these references have been tagged to individual, coded trials. The coded trials are held in the CCMDCTR‐Studies Register and records are linked between the two registers through the use of unique Study ID tags. Coding of trials is based on the EU‐Psi coding manual, using a controlled vocabulary. (Please contact the CCMD Information Specialists for further details). Reports of trials for inclusion in the Group's registers were collated from routine (weekly), generic searches of MEDLINE (1950 ‐), Embase (1974 ‐) and PsycINFO (1967 ‐), quarterly searches of the Cochrane Central Register of Controlled Trials (CENTRAL) and review‐specific searches of additional databases. Reports of trials were also sourced from international trial registers, pharmaceutical companies, conference proceedings and other (non‐Cochrane) systematic reviews and meta‐analyses.
Details of CCMD's generic search strategies (used to identify RCTs) can be found on the Group's website, with an example of the core MEDLINE search displayed below.
Core search strategy used to inform the Cochrane Common Mental Disorders Controlled Trials Register (CCMDCTR): OVID MEDLINE A weekly search alert based on condition + RCT filter only 1. [MeSH Headings]: eating disorders/ or anorexia nervosa/ or binge‐eating disorder/ or bulimia nervosa/ or female athlete triad syndrome/ or pica/ or hyperphagia/ or bulimia/ or self‐injurious behavior/ or self mutilation/ or suicide/ or suicidal ideation/ or suicide, attempted/ or mood disorders/ or affective disorders, psychotic/ or bipolar disorder/ or cyclothymic disorder/ or depressive disorder/ or depression, postpartum/ or depressive disorder, major/ or depressive disorder, treatment‐resistant/ or dysthymic disorder/ or seasonal affective disorder/ or neurotic disorders/ or depression/ or adjustment disorders/ or exp antidepressive agents/ or anxiety disorders/ or agoraphobia/ or neurocirculatory asthenia/ or obsessive‐compulsive disorder/ or obsessive hoarding/ or panic disorder/ or phobic disorders/ or stress disorders, traumatic/ or combat disorders/ or stress disorders, post‐traumatic/ or stress disorders, traumatic, acute/ or anxiety/ or anxiety, castration/ or koro/ or anxiety, separation/ or panic/ or exp anti‐anxiety agents/ or somatoform disorders/ or body dysmorphic disorders/ or conversion disorder/ or hypochondriasis/ or neurasthenia/ or hysteria/ or munchausen syndrome by proxy/ or munchausen syndrome/ or fatigue syndrome, chronic/ or obsessive behavior/ or compulsive behavior/ or behavior, addictive/ or impulse control disorders/ or firesetting behavior/ or gambling/ or trichotillomania/ or stress, psychological/ or burnout, professional/ or sexual dysfunctions, psychological/ or vaginismus/ or Anhedonia/ or Affective Symptoms/ or *Mental Disorders/
2. [Title/ Author Keywords]: (eating disorder* or anorexia nervosa or bulimi* or binge eat* or (self adj (injur* or mutilat*)) or suicide* or suicidal or parasuicid* or mood disorder* or affective disorder* or bipolar i or bipolar ii or (bipolar and (affective or disorder*)) or mania or manic or cyclothymic* or depression or depressive or dysthymi* or neurotic or neurosis or adjustment disorder* or antidepress* or anxiety disorder* or agoraphobia or obsess* or compulsi* or panic or phobi* or ptsd or posttrauma* or post trauma* or combat or somatoform or somati#ation or medical* unexplained or body dysmorphi* or conversion disorder or hypochondria* or neurastheni* or hysteria or munchausen or chronic fatigue* or gambling or trichotillomania or vaginismus or anhedoni* or affective symptoms or mental disorder* or mental health).ti,kf.
3. [RCT filter]: (controlled clinical trial.pt. or randomized controlled trial.pt. or (randomi#ed or randomi#ation).ab,ti. or randomly.ab. or (random* adj3 (administ* or allocat* or assign* or class* or control* or determine* or divide* or distribut* or expose* or fashion or number* or place* or recruit* or subsitut* or treat*)).ab. or placebo*.ab,ti. or drug therapy.fs. or trial.ab,ti. or groups.ab. or (control* adj3 (trial* or study or studies)).ab,ti. or ((singl* or doubl* or tripl* or trebl*) adj3 (blind* or mask* or dummy*)).mp. or clinical trial, phase ii/ or clinical trial, phase iii/ or clinical trial, phase iv/ or randomized controlled trial/ or pragmatic clinical trial/ or (quasi adj (experimental or random*)).ti,ab. or ((waitlist* or wait* list* or treatment as usual or TAU) adj3 (control or group)).ab.)
4. (1 and 2 and 3)
Records were screened for reports of RCTs within the scope of the Cochrane Common Mental Disorders Group. Secondary reports of RCTs were tagged to the appropriate study record. Similar weekly search alerts were also conducted on OVID Embase and PsycINFO, using relevant subject headings (controlled vocabularies) and search syntax, appropriate to each resource.
For this review, the CCMDCTR (Studies and References Register) was cross‐searched using the following terms: (lamotrigin* and (bipolar or mania or manic or hypomani* or “affective psychosis” or “rapid cycling” or schizoaffective)) [all fields]
Data and analyses
Comparison 1. Lamotrigine versus placebo.
Comparison 2. Lamotrigine versus lithium.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Bowden 2003.
| Study characteristics | ||
| Methods | Study design: randomized, double‐blind, parallel‐group, placebo‐controlled, multicenter study | |
| Participants | Diagnosis: bipolar I disorder Method of diagnosis: DSM‐IV or had been manic or hypomanic within 60 days of the screening visit, had manic or hypomanic symptoms at enrollment, and had at least 1 additional manic or hypomanic episode and 1 depressed episode within 3 years of enrollment. Age: for lamotrigine, median = 40.7 (SD = 12.6) years; for lithium, median = 41.9 (SD = 11.3) years; for placebo, median = 40.9 (SD = 11) years; range = not specified Sex: lamotrigine 55% women; 45% men, lithium 52% women; 48% men, placebo 51% women; 49% men Location: Australia, Austria, Belgium, Canada, Greece, New Zealand, Norway, Poland, the UK, the USA, Yugoslavia Co‐morbidities: not described Adjunctive therapy: none Adjunctive medication: concomitant antiepileptic drugs and other psychotropic medications were permitted during the open‐label phase. For patients receiving lithium during the open‐label phase, the dosage was tapered over at least 3 weeks and discontinued a minimum of 1 week before they entered the double‐blind phase of the study. |
|
| Interventions | Participants were randomly assigned to either:
|
|
| Outcomes | Time points for assessment:clinic visits were scheduled to occur weekly during the first 4 weeks of the double‐blind phase, biweekly through week 8, and every 4 weeks thereafter, through week 76. At each clinic visit, psychiatric evaluations from the screening visit were administered again, and patients were queried regarding the occurrence of adverse events. Primary outcome:the time to intervention (addition of pharmacotherapy or electroconvulsive therapy) for any mood episode. Secondary outcome: time to early discontinuation for any reason (i.e. survival in study); time to intervention for a manic, hypomanic, or mixed episode; time to intervention for a depressive episode; and mean change from baseline (defined as day 1 of the double‐blind phase) in scores on the MRS, HAM‐D, CGI, and GAS scales during double‐blind treatment. | |
| Notes | Date of study: unknown Funding source: GlaxoSmithKline Declarations of interest among the primary researchers: Dr Bowden has received research support from Abbott Laboratories, Bristol‐MyersSquibbCo, GlaxoSmithKline, Janssen Pharmaceutica, Lilly Research Laboratories, the National Institute of Mental Health, Parke‐Davis Pharmaceuticals, the RobertWoodJohnson Pharmaceutical Institute, GlaxoSmith‐ Kline, and the Stanley Medical Research Institute. Heis a paid consultant for Abbott Laboratories, GlaxoSmithKline, Janssen Pharmaceutica, Lilly Research Laboratories, Sanofi‐Synthélabo, and UCBPharma and is on the speakers bureau for Abbott Laboratories, AstraZeneca, Glaxo Wellcome, Janssen Pharmaceutica, Lilly Research Laboratories, and Pfizer. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not described in sufficient detail |
| Allocation concealment (selection bias) | Unclear risk | Not described in sufficient detail |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | The protocol for this randomized, double‐blind, parallel‐ group, placebo‐controlled, multicenter study conducted at 64 centers (Glaxo Wellcome protocol SCAB2006, GW606) was approved by an institutional review board or ethics committee at each study site. In both cases, these concomitant antiepileptic drugs were gradually withdrawn so that lamotrigine monotherapy at target dosages was received for at least 1 week before the double‐blind phase, with the lamotrigine dosage immediately doubled after discontinuation of valproate and gradually halved after discontinuation of carbamazepine. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | The primary efficacy end point was the time to intervention (addition of pharmacotherapy or electroconvulsive therapy) for any mood episode. Secondary efficacy measures included time to early discontinuation for any reason (i.e., survival in study); time to intervention for a manic, hypomanic, or mixed episode; time to intervention for a depressive episode; and mean change from baseline (defined as day 1 of the double‐blind phase) in scores on the MRS, HAM‐D, CGI, and GAS scales during double‐blind treatment. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | In most cases, efficacy and safety can be evaluated. |
| Selective reporting (reporting bias) | Low risk | The content described in the method is reflected in the result |
| Other bias | Low risk | No special instruction |
Calabrese 1999.
| Study characteristics | ||
| Methods | Study design: randomized, double‐blind, parallel‐group, placebo‐controlled, multicenter study | |
| Participants | Diagnosis:bipolar I depression Method of diagnosis: bipolar I disorder, as defined by DSM‐IV criteria, and had at least 1 of which was a manic or mixed episode. The diagnosis was confirmed by the Structual Clinical Interview for DSM‐IV. Age:for lamotrigine 50 mg/day, median = 41 years; range =19 to 75; for lamotrigine 200 mg/day, median = 42 years; range =21 to 66; for placebo, median = 42 years; range =21 to 71 Sex: lamotrigine 50 mg/day 67% women; 33% men, lamotrigine 200 mg/day 56% women; 44% men, placebo 59% women; 41% men Location:15 centers in the USA and 6 centers in the UK, France, and Australia. Co‐morbidities: not described Adjunctive therapy: none Adjunctive medication:none |
|
| Interventions | Participants were randomly assigned to either:
Experimental arm 1 ‐ lamotrigine 50 mg/day
N = 66
Duration: 7 weeks
Treatment protocol: 50mg/day. Therapist/face‐to‐face contact: At the last treatment visit, patients were given physical examinations and clinical laboratory tests. Experimental arm 2 ‐ lamotrigine 200 mg/day N = 63 Duration: 7 weeks Treatment protocol: 200mg/day. Therapist/face‐to‐face contact: At the last treatment visit, patients were given physical examinations and clinical laboratory tests. Comparator arm ‐ placebo N = 66 Duration: 7 weeks Treatment protocol: The number of placebo tablets was adjusted at each week and for each lamotrigine dose so that the total number of tablets administrated per day was always 8. Therapist/face‐to‐face contact: At the last treatment visit, patients were given physical examinations and clinical laboratory tests. |
|
| Outcomes | Time points for assessment: clinical visits were conducted at screening, baseline, on the fourth day of treatment, and the end of every week for the 7‐week duration of treatment. Primary outcome: 17‐item HAM‐D Secondary outcome:1‐item HAM‐D, 31‐item HAM‐D, MADRS, CGI‐S, CGI‐I, MRS | |
| Notes | Date of study: unknown Funding source: GlaxoSmithKline Declarations of interest among the primary researchers: not described | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | It is stratified as described below, but the selection bias is unknown. To balance the effects of recent use of lithium, randomization was stratified according to intensity of treatment with lithium during the 5 months preceding study entry. |
| Allocation concealment (selection bias) | Unclear risk | Not described in sufficient detail |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | The number of placebo tablets was adjusted at each week and for each lamotrigine dose so that the total number of tablets administrated per day was always 8. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | No mention of outcome assessors |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | In most cases, efficacy and safety can be evaluated. |
| Selective reporting (reporting bias) | Low risk | The content described in the method is reflected in the result |
| Other bias | Low risk | No special instruction. |
Calabrese 2000.
| Study characteristics | ||
| Methods | Study design: double‐blind, flexible‐dose, placebo‐controlled, parallel‐group design. | |
| Participants | Diagnosis: Diagnostic and Statistical Manual of Mental Disorders, Fourth edition (DSM‐IV) criteria. Method of diagnosis: bipolar disorder I or II with rapid cycling Age: for lamotrigine, median = 38.5 years; range =2 to ‐61 ; for placebo, median = 37.4 years; range =18 to 64 Sex: lamotrigine 55% women; 45% men, placebo 59% women; 41% men, Location:the USA and Canada Co‐morbidities: not described Adjunctive therapy: none Adjunctive medication: If patients did not respond to study treatment and pharmacotherapy was clinically indicated to treat emerging symptoms of a mood episode, the investigator was encouraged to add lithium or divalproex. |
|
| Interventions | Participants were randomly assigned to either:
Experimental arm ‐ lamotrigine
N = 93
Duration: 6 months
Treatment protocol: from 100 mg to 500 mg/day Comparator arm ‐ placebo N = 89 Duration: 6 months Treatment protocol:not described Therapist/face‐to‐face contact: not described |
|
| Outcomes | Time points for assessment: Primary outcome: at least 1 dose of study drug, had at least post‐baseline primary outcome assessment during the randomized phase, or required additional therapy for symptoms of emerging mood episode. Secondary outcome: survival in study, percentage of patients stable without relapse for 6 months, and changes in the Global Assessment Scale (GAS) and Clinical Global Impressions‐Severity (CGI‐S) scale. | |
| Notes | Date of study: unknown Funding source: Glaxo Wellcome, Eli Lilly, Novartis, Janssen, AstraZeneca, Bristol Myers Squibb, Abbott Laboratories, Stanley Foundation, the Lattner Foundation, Timberlawn, Abbott Laboratories, Lilly Research, Sanofi Synthelabo, UBC Pharma, Lilly Research, Parke‐Davis, Johnson Pharmaceutical Institute, SmithKline Beecham, Solvay, Upjohn Declarations of interest among the primary researchers: not described | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | It is stratified as described below, but the selection bias is unknown. Patients were randomly assigned in a 1:1 ratio to treatment with lamotrigine or matching placebo in a double‐blind fashion. Additionally, patients were stratified by diagnosis of bipolar I or IIdisorder. |
| Allocation concealment (selection bias) | Unclear risk | Not described in sufficient detail |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Patients were randomly assigned in a 1:1 ratio to treatment with lamotrigine or matching placebo in a double‐blind fashion. Additionally, patients were stratified by diagnosis of bipolar I or II disorder. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | If patients did not respond to study treatment and pharmacotherapy was clinically indicated to treat emerging symptoms of a mood episode, the investigator was encouraged to add lithium or divalproex. In the event that lithium or divalprorex was ineffective or inappropriate, the investigator could choose any regimen of pharmacotherapy or ECT. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | In most cases, efficacy and safety can be evaluated. |
| Selective reporting (reporting bias) | Low risk | The content described in the method is reflected in the result |
| Other bias | Low risk | No special instruction. |
Calabrese 2003.
| Study characteristics | ||
| Methods | Study design: randomized, double‐blind, parallel‐group, placebo‐controlled, multicenter study | |
| Participants | Diagnosis:bipolar I disorder Method of diagnosis: currently experiencing a major depressive episode as defined by DSM‐IV and ascertained by clinical interview or if their most recent mood episode was a major depressive episode and occurred within 60 days of the screening visit with depressive symptoms still present at enrollment; had at least 1 manic or hypomanic episode within 3 years of enrollment; and had at least 1 additional depressed episode within 3 years of enrollment. Age: for lamotrigine, median = 44.1 (SD = 11.7) years; for lithium, median = 43.6 (SD = 12.3) years; for placebo, median = 42.1 (SD = 13.0) years; range = not specified Sex: lamotrigine 59% women; 41% men, lithium 60% women; 40% men, placebo 50% women; 50% men Location:15 countries Co‐morbidities: not described Adjunctive therapy: electroconvulsive therapy if the treating psychiatrist determined clinically that developing illness symptomatology required such additional intervention. Adjunctive medication: antidepressants, antipsychotics, anticonvulsants/mood stabilizers if the treating psychiatrist determined clinically that developing illness symptomatology required such additional intervention. |
|
| Interventions | Participants were randomly assigned to either:
Experimental arm ‐ lamotrigine
N = 221
Duration: 76 weeks
Treatment protocol:50, 200, or 400 mg/day Therapist/face‐to‐face contact: not described Comparator arm 1 ‐ lithium N = 121 Duration: 76 weeks Treatment protocol:Lithium titrated to serum levels of 0.8 to 1.1 mEq/L Therapist/face‐to‐face contact: not described Comparator arm 2 ‐ placebo Duration: 76 weeks Treatment protocol: not described Therapist/face‐to‐face contact: not described |
|
| Outcomes | Time points for assessment: Clinic visits were scheduled to occur weekly during the first 4 weeks of the double‐blind phase, biweekly through week 8, and every 4 weeks thereafter, through week 76. At each clinic visit, psychiatric evaluations from the screening visit were administered again, and patients were queried regarding the occurrence of adverse events. Primary outcome: Time to intervention for any mood episode Secondary outcome: time to intervention for a manic or hypomanic episode; time to intervention for a depressive episode; mean change from baseline on the HAM‐D, MRS, CGI‐S and GAS scores; and CGI‐I scores for double‐blind treatment | |
| Notes | Date of study: unknown Funding source: GlaxoSmithKline, Abbott Laboratories, Glaxo Wellcome, Janssen, Lilly Research, Sanof Synthelabo, UBC Pharma, Bristol Myers Squibb, Lilly Research, Parke‐Davis, Johnson Pharmaceutical Institute, SmithKline Beecham, Stanley Foundation, AstraZeneca, Pfizer, Novartis, Elan Sanofi, Sigma‐Tau, Solvay, Janssen‐Cilag, Declarations of interest among the primary researchers: GlaxoSmithKline | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not described in sufficient detail |
| Allocation concealment (selection bias) | Low risk | All patients, regardless of treatment group, had serum drawn for lithium levels at no less than 8‐week intervals. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | To maintain the blind, each instruction to adjust the lithium dose was accompanied by a corresponding instruction to adjust the lithium placebo dose in a lamotrigine and placebo patient. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | From the following description, the evaluator determined that it was open‐label All patients, regardless of treatment group, had serum drawn for lithium levels at no less than 8‐week intervals. To maintain the blind, each instruction to adjust the lithium dose was accompanied by a corresponding instruction to adjust the lithium placebo dose in a lamotrigine and placebo patient. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | In most cases, efficacy and safety can be evaluated. |
| Selective reporting (reporting bias) | Low risk | The content described in the method is reflected in the result. |
| Other bias | Low risk | No special instruction. |
Calabrese 2008 [GW603/SCAA2010].
| Study characteristics | ||
| Methods | Study design: randomized, double‐blind, parallel‐group, placebo‐controlled trials of lamotrigine monotherapy. | |
| Participants | Diagnosis:bipolar I and II disorder Method of diagnosis: Diagnostic and Statistical Manual, 4th edition (DSM‐IV), and diagnosis was confirmed by the Structured Clinical Interview for DSM‐IV (SCID). Age: for lamotrigine, median = 40.5 (SD = 11.3) years; for placebo, median = 40.9 (SD = 11.2) years; range = not specified Sex: lamotrigine 64% women; 36% men, placebo 59% women; 41% men Location: not described Co‐morbidities: not described Adjunctive therapy: not described Adjunctive medication: not described |
|
| Interventions | Experimental arm ‐ lamotrigine
N = 103
Duration: 10 weeks
Treatment protocol: Weeks 1 and 2: 25 mg, Weeks 3 and 4: 50 mg, Week 5: 100 mg, Week 6: flexible, 100 mg to 200 mg, Week 7: flexible, 100 mg mto 300 mg, Weeks 8 to 10: flexible, 100 mg to 400 mg Comparator arm ‐ placebo N = 103 Duration: 10 weeks Treatment protocol:not described Therapist/face‐to‐face contact: not described |
|
| Outcomes | Primary outcome: 17‐item HAM‐D Secondary outcome: HAM‐D Item 1, 31‐item HAM‐D, MADRS, CGI‐S, CGI‐I | |
| Notes | Date of study: unknown Funding source: GlaxoSmithKline Declarations of interest among the primary researchers: not described | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | Randomization of bipolar disorder Ⅰ and Ⅱ has been stratified. |
| Allocation concealment (selection bias) | Unclear risk | No description of randomization methods was found. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Any participant whose study treatment was unblinded was withdrawn from the study. If an investigator broke the blind, the reason was documented in a report forwarded to the sponsor company. In the event of a subject withdrawing from the study once randomization had occurred, that subject number was not reallocated. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Any subject whose study treatment was unblinded was withdrawn from the study. If an investigator broke the blind, the reason was documented in a report forwarded to the sponsor company. In the event of a participant withdrawing from the study once randomization had occurred, that participant number was not reallocated. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | The evaluator of the effect is unknown, and follow‐up is not possible. |
| Selective reporting (reporting bias) | Low risk | All the results described in the method have been published. |
| Other bias | Unclear risk | no special instruction |
Calabrese 2008 [SCA100223].
| Study characteristics | ||
| Methods | Study design: randomized, double‐blind, parallel‐group, placebo‐controlled trials of lamotrigine monotherapy. | |
| Participants | Diagnosis: bipolar II disorder Method of diagnosis: Diagnostic and Statistical Manual, 4th edition (DSM‐IV), and diagnosis was confirmed by the Structured Clinical Interview for DSM‐IV (SCID). Age: for lamotrigine, median = 38.1 (SD = 11.5) years; for placebo, median = 36.5 (SD = 11.9) years; range = not specified Sex: lamotrigine 64% women; 36% men, placebo 63% women; 37% men Location: not described Co‐morbidities: not described Adjunctive therapy: not described Adjunctive medication: not described |
|
| Interventions | Experimental arm ‐ lamotrigine
N = 111
Duration: 8 weeks
Treatment protocol: Weeks 1 and 2: 25 mg, Weeks 3 and 4: 50 mg, Week 5: 100 mg, Weeks 6 to 8: 200 mg Comparator arm ‐ placebo N = 110 Duration: 8 weeks Treatment protocol: not described Therapist/face‐to‐face contact: not described |
|
| Outcomes | Primary outcome:MADRS Secondary outcome:HAM‐D Item 1, 17‐item HAM‐D, 31‐item HAM‐D, CGI‐S, CGI‐I | |
| Notes | Date of study: unknown Funding source: GlaxoSmithKline Declarations of interest among the primary researchers: not described | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No description of randomization methods was found. |
| Allocation concealment (selection bias) | Unclear risk | No description of randomization methods was found. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Any participant whose study treatment was unblinded was withdrawn from the study. If an investigator broke the blind, the reason was documented in a report forwarded to the sponsor company. In the event of a subject withdrawing from the study once randomization had occurred, that participant number was not reallocated. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Any participant whose study treatment was unblinded was withdrawn from the study. If an investigator broke the blind, the reason was documented in a report forwarded to the sponsor company. In the event of a participant withdrawing from the study once randomization had occurred, that participant number was not reallocated. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | The evaluator of the effect is unknown, and follow‐up is not possible. |
| Selective reporting (reporting bias) | Low risk | All the results described in the method have been published. |
| Other bias | Unclear risk | no special instruction |
Calabrese 2008 [SCA30924].
| Study characteristics | ||
| Methods | Study design: randomized, double‐blind, parallel‐group, placebo‐controlled trials of lamotrigine monotherapy. | |
| Participants | Diagnosis:bipolar I disorder Method of diagnosis: Diagnostic and Statistical Manual, 4th edition (DSM‐IV), and diagnosis was confirmed by the Structured Clinical Interview for DSM‐IV (SCID). Age: for lamotrigine, median = 40.5 (SD = 12.5) years; for placebo, median = 38.2 (SD = 12.1) years; range = not specified Sex: lamotrigine 54% women; 46% men, placebo 54% women; 46% men Location: not described Co‐morbidities: not described Adjunctive therapy: not described Adjunctive medication: not described |
|
| Interventions | Experimental arm ‐ lamotrigine
N = 131
Duration: 8 weeks
Treatment protocol: Weeks 1 and 2: 25 mg Weeks 3 and 4: 50 mg, Week 5: 100 mg, Weeks 6 to 8: 200 mg Comparator arm ‐ placebo N = 128 Duration: 8 weeks Treatment protocol: not described Therapist/face‐to‐face contact: not described |
|
| Outcomes | Primary outcome: MADRS Secondary outcome: HAM‐D Item 1, 17‐item HAM‐D, 31‐item HAM‐D, CGI‐S, CGI‐I | |
| Notes | Date of study: unknown Funding source: GlaxoSmithKline Declarations of interest among the primary researchers: not described | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No description of randomization methods was found. |
| Allocation concealment (selection bias) | Unclear risk | No description of randomization methods was found. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Any participant whose study treatment was unblinded was withdrawn from the study. If an investigator broke the blind, the reason was documented in a report forwarded to the sponsor company. In the event of a participant withdrawing from the study once randomization had occurred, that participant number was not reallocated. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | Subjects must have had a history of full or partial hospitalization for a mood episode, or incarceration with or without formal charges as a result of mania‐related behavior. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | The evaluator of the effect is unknown, and follow‐up is not possible. |
| Selective reporting (reporting bias) | Low risk | All the results described in the method have been published. |
| Other bias | Unclear risk | no special instruction |
Calabrese 2008 [SCA40910].
| Study characteristics | ||
| Methods | Study design: randomized, double‐blind, parallel‐group, placebo‐controlled trials of lamotrigine monotherapy. | |
| Participants | Diagnosis: bipolar I disorder Method of diagnosis: Diagnostic and Statistical Manual, 4th edition (DSM‐IV), and diagnosis was confirmed by the Structured Clinical Interview for DSM‐IV (SCID). Age: for lamotrigine, median = 37.6 (SD = 12.6) years; for placebo, median = 37.3 (SD = 11.5) years; range = not specified Sex: lamotrigine 57% women; 43% men, placebo 53% women; 47% men Location: not described Co‐morbidities: not described Adjunctive therapy: not described Adjunctive medication: not described |
|
| Interventions | Experimental arm ‐ lamotrigine
N = 133
Duration: 8 weeks
Treatment protocol: Weeks 1 and 2: 25 mg, Weeks 3 and 4: 50 mg, Week 5: 100 mg, Weeks 6 to 8: 200 mg N = 124 Duration: 8 weeks Treatment protocol: not described Therapist/face‐to‐face contact: not described |
|
| Outcomes | Primary outcome: MADRS Secondary outcome: HAM‐D Item 1, 17‐item HAM‐D, 31‐item HAM‐D, CGI‐S, CGI‐I | |
| Notes | Date of study: unknown Funding source: GlaxoSmithKline Declarations of interest among the primary researchers: not described | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | No description of randomization methods was found. |
| Allocation concealment (selection bias) | Unclear risk | No description of randomization methods was found. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | Any participant whose study treatment was unblinded was withdrawn from the study. If an investigator broke the blind, the reason was documented in a report forwarded to the sponsor company. In the event of a participant withdrawing from the study once randomization had occurred, that participant number was not reallocated. |
| Blinding of outcome assessment (detection bias) All outcomes | Unclear risk | Any participant whose study treatment was unblinded was withdrawn from the study. If an investigator broke the blind, the reason was documented in a report forwarded to the sponsor company. In the event of a participant withdrawing from the study once randomization had occurred, that participant number was not reallocated. |
| Incomplete outcome data (attrition bias) All outcomes | High risk | The evaluator of the effect is unknown, and follow‐up is not possible. |
| Selective reporting (reporting bias) | Low risk | All the results described in the method have been published. |
| Other bias | Unclear risk | no special instruction |
Koyama 2011.
| Study characteristics | ||
| Methods | Study design: a placebo‐controlled, multicenter, double‐blind parallel‐group study. | |
| Participants | Diagnosis: bipolar disorder Method of diagnosis: DSM‐IV‐TR Age: for lamotrigine, median = 42.4 (SD = 11.79) years; for lithium, median = 43.1 (SD = 12.68) years; range = not specified Sex: lamotrigine 60% women; 40% men, placebo 53% women; 47% men, Location: Japan Co‐morbidities: not described Adjunctive therapy: prohibition of psychotherapy, cognitive behavior therapy, electroconvulsive therapy, sleep deprivation therapy, light irradiation therapy, and St. John's Wort. Adjunctive medication: throughout the second phase, the use of antipsychotics for the treatment of mania or depressive episodes and drugs that enhance the therapeutic effect on depression or mania was banned. From the 2nd transitional period, it was possible to use any one of the 3 denominations, triazolam (0.5 mg/dose or less), zopiclone (10 mg/dose or less) or brotizolam (0.25 mg/dose or less), as a single dose. However, it was used 3 times or less per week. |
|
| Interventions | Participants were randomly assigned to either:
Experimental arm ‐ lamotrigine
N = 45
Duration: 26 weeks
Treatment protocol: 100 mg or 200 mg/day. Once a day in the evening. Comparator arm ‐ placebo N = 58 Duration: 26 weeks Treatment protocol: not described Therapist/face‐to‐face contact: not described |
|
| Outcomes | Time points for assessment: not described Primary outcome: Time to Withdrawal from Study (TWS) Secondary outcome: TIme to Intervention for Mood episode (TIME), TIme to Intervention for Depressive Episode (TIDep), TIme to Intervention for Mainic, Hypomanic, or Mixed Episode (TIMan) | |
| Notes | Date of study: unknown Funding source: GlaxoSmithKline Declarations of interest among the primary researchers: not described | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not described in sufficient detail |
| Allocation concealment (selection bias) | Unclear risk | Participants whose symptoms were stable after administration of 200 mg/day of lamotrigine in the first phase were randomly assigned to the placebo group or the lamotrigine group and transferred to the second phase. |
| Blinding of participants and personnel (performance bias) All outcomes | Low risk | The investigational products were allocated 1:1 using an allocation table prepared in advance by the investigational drug allocation manager. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | TIME/TIDep/TIMan requires the investigator to share some drug therapy other than therapeutic drugs due to recurrence/relapse of bipolar disorder depression, mania, hypomania or mixed episodes after the 2nd stage judgment was made until the first enforcement. |
| Incomplete outcome data (attrition bias) All outcomes | Unclear risk | Details of the reason for discontinuation are unknown |
| Selective reporting (reporting bias) | Low risk | The content described in the method is reflected in the result |
| Other bias | Low risk | No special instruction. |
Licht 2010.
| Study characteristics | ||
| Methods | Study design: an open, randomized effectiveness study mimicking clinical practice. | |
| Participants | Diagnosis: bipolar I disorder Method of diagnosis: DSM‐IV Age: for lamotrigine, median = 38.2 (SD = 11.8) years; for lithium, median = 37.3 (SD = 11.5) years; range = not specified Sex: lamotrigine 52% women; 48% men, lithium 46% women; 54% men, Location: Denmark and Sweden Co‐morbidities: not described Adjunctive therapy: none Adjunctive medication: additional antipsychotic or antidepressant drugs were allowed within the first six months after randomization, with no restrictions in terms of type, number of dose of medications, and no attempt were made to encouraged to achieve monotherapy with study drugs at month 6. Supplemental treatment with benzodiazepines and benzodiazepine‐like drugs, e.g. zolpidem was allowed throughout the study. |
|
| Interventions | Participants were randomly assigned to either:
Experimental arm ‐ lamotrigine
N = 77
Duration: 5.8 years
Treatment protocol: 25 mg/day during week 1 and 2, 50 mg/day during weeks 3 and 4, and 100 mg/day during week 5. After week 45 the daily dose was increased by 100 mg weekly to a maximum of 400 mg daily. Comparator arm ‐ lithium N = 78 Duration: 5.8 years Treatment protocol: patients received lithium citrate or lithium sulphate (each tablet containing 6 mmol lithium ion), one tablet twice daily through week 1, with subsequent adjustment of dose until a serum lithium level (12‐hour value) of 0.5 mmol/L to 1.0 mmol/L was achieved. Therapist/face‐to‐face contact: not described |
|
| Outcomes | Time points for assessment:the patients were evaluated at visits with their treating psychiatrist (investigator) as clinically needed and in accordance with the protocol, which required a minimum of fixed visits at month 1, month 3, and month 6, and subsequently at least one visit per each three months of follow‐up.
Primary outcome:psychotropic treatment in addition to study drugs and benzodiazepines still required at month 6. Hospitalization still required at month 6. Psychotropic treatment during at least one week required after month 6. Hospitalization during at least one week required after month 6. Secondary outcome:patient discontinued study drug. Consent withdrawn. Protocol violation. Severe abuse of alcohol or drugs. Adverse events. Lost to follow‐up. Other Suicide Suicide attempt Pregnancy Censoring due to fixed study end. |
|
| Notes | Date of study: unknown Funding source: Stanley Medical Institute, GlaxoSmithKline, Eli Lilly, Janssen, Bristol‐Myers Squibb, AstraZeneca, Lundbeck, Declarations of interest among the primary researchers: not described | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Low risk | The randomization was done in blocks of four within each centre, with the block size kept hidden for the investigators. |
| Allocation concealment (selection bias) | Low risk | The index episode was treated at the discretion of the clinician, and, when it was timely, the patients were randomly allocated to either lithium or lamotrigine as continuation and prophylactic. |
| Blinding of participants and personnel (performance bias) All outcomes | Unclear risk | Lithium twice daily through week 1 Lamotrigine was given in the morning. |
| Blinding of outcome assessment (detection bias) All outcomes | High risk | At each visit the investigator evaluated whether an endpoint had been reached since the last visit and recorded its date. If the endpoint indicated insufficient maintenance treatment. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | In most cases, efficacy and safety can be evaluated. |
| Selective reporting (reporting bias) | Low risk | The content described in the method is reflected in the result |
| Other bias | Low risk | No special instruction. |
Suppes 2008a.
| Study characteristics | ||
| Methods | Study design: single‐blind, 16‐week, randomized comparison of open‐label lithium and lamotrigine in the acute treatment of patients | |
| Participants | Diagnosis: bipolar II disorder Method of diagnosis:SCID‐research version interview and met criteria for DSM‐IV bipolar II disorder Age: for lamotrigine, median = 36.9 (SD = 12.3) years; for lithium, median = 36.2 (SD = 11.4) years; range = not specified Sex: lamotrigine 68.2% women; 31.8% men, lithium 57.4% women; 42.6% men Location: two sites in Texas (Dallas and Houston). Co‐morbidities: not described Adjunctive therapy: none Adjunctive medication: short‐term use of limited benzodiazepines/hypnotics for a maximum of 5 consecutive days, on no more than one occasion over the course of the 16‐week trial, was permitted. |
|
| Interventions | Participants were randomly assigned to either:
|
|
| Outcomes | Time points for assessment: at every visit Primary outcome: change in depression symptoms from baseline to week 16 Secondary outcome: incidence and severity of hypomanic and/or depressive symptoms, medication tolerability, response (defined as a 50% reduction on the Ham‐D17), remission (defined as Ham‐D17 or MADRS ≤ 12), and switch into hypomania (defined as CGI‐BP Mania severity score of 4 or greater). | |
| Notes | Date of study: unknown Funding source: not described Declarations of interest among the primary researchers: not described | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Random sequence generation (selection bias) | Unclear risk | Not described in sufficient detail |
| Allocation concealment (selection bias) | High risk | Patients were randomized to open‐label monotherapy. Patients and physicians knew their group assignment. |
| Blinding of participants and personnel (performance bias) All outcomes | High risk | Patients were randomized to open‐label monotherapy. Patients and physicians knew their group assignment. |
| Blinding of outcome assessment (detection bias) All outcomes | Low risk | All patients met with a psychiatrist or mental health nurse practitioner and symptom severity was assessed by a blinded research assistant at biweekly visits for the duration of the 16‐week study. |
| Incomplete outcome data (attrition bias) All outcomes | Low risk | No special instruction. |
| Selective reporting (reporting bias) | Low risk | The content described in the method is reflected in the result |
| Other bias | High risk | A single‐blind comparison of lithium and lamotrigine for the treatment of bipolar II depression |
CGI‐I: Clinical Global Impressions scale‐Improvement scale; CGI‐S: Clinical Global Impressions scale; CGI‐S: Clinical Global Impressions‐Severity scale; DSM‐IV: Diagnostic and Statistical Manual of Mental Disorders, Fourth edition;ECT: electroconvulsive therapy; GAS: Global Assessment Scale; HAM‐D: Hamilton Depression Rating Scale; MADRS: Montgomery‐Asberg Depression Rating Scale; MRS: Mania Rating Scale; SD: standard deviation.
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Berk 1999a | The patient was in the acute mania stage. |
| Bowden 2012a | wrong drug |
| ChiCTR2000038166 | wrong population |
| EudraCT 2006‐001317‐15 | The patient was in the acute mania stage. Target patients have different age groups |
| Frangou 1999 | The patient was in the acute mania stage. |
| Gao 2020 | wrong intervention |
| Gardiner 2011 | wrong outcome |
| Geddes 2005 | wrong intervention |
| Geddes 2014 | wrong outcome |
| Geddes 2015 | wrong outcome |
| Geddes 2015b | wrong outcome |
| Geddes 2016 | wrong outcome |
| Ketter 2006 | wrong intervention |
| Kwon 2001 | Acute bipolar disorder |
| NCT00074776 | Acute treatment of Bipolar II depression |
| NCT01195363 | wrong intervention |
| NCT01587066 | wrong intervention |
| NCT01674010 | wrong intervention |
| Parikh 2012 | wrong intervention |
| PER‐122‐12 | wrong intervention |
| PER‐123‐12 | wrong intervention |
| Simon 2018 | wrong outcome |
| Swann 2005 | wrong intervention |
| Tolliver 2014 | wrong outcome |
| Tolliver 2018 | wrong intervention |
| van der Loos 2009 | Acute bipolar disorder |
| van der Loos 2011 | wrong intervention |
Differences between protocol and review
Types of outcome measures In the protocol, we had specified the observation period for the outcome as follows:
short term, up to 12 weeks after treatment initiation;
long term, from six months after treatment initiation.
For the review, we revised the timeline of outcome measures as follows:
short term, up to 12 weeks after treatment initiation (range: 7 to 16 weeks);
long term, from six months after treatment initiation (range: 6 months to 16 months).
Subgroup analysis
Due to limited data we could not pursue our planned subgroup analyses of mental disorder co‐morbidities, duration of treatment, setting and pharmacological modality.
Sensitivity analysis Due to limited data we did not perform sensitivity analysis as planned at the protocol stage.
Contributions of authors
Conceiving the review: YH, KK.
Co‐ordinating the review: YH, KK, TF, SS.
Undertaking manual searches: YH, KK.
Organizing retrieval of papers: YH, KK.
Screening retrieved papers against inclusion criteria: YH, KK, TF, SS.
Appraising quality of papers: YH, KK, NW, TF.
Extracting data from papers: YH, KK, TF, SS.
Providing additional data about papers: YH, KK.
Obtaining and screening data on unpublished studies: YH, KK.
Data management for the review: YH, KK.
Entering data into RevMan web: YH, KK.
RevMan web statistical data: YH, KK.
Other statistical analysis not using RevMan web: YH, KK.
Double entry of data: data entered by person one, YH; data entered by person two, KK.
Interpretation of data: YH, KK, NW, TF, SS.
Statistical inferences: YH, KK, NW, TF, SS.
Writing the review: YH, KK.
Securing funding for the review: YH, KK.
Performing previous work that was the foundation of the present study: YH, KK, TF, SS.
Guarantor for the review: YH.
Person responsible for reading and checking review before submission: YH.
Sources of support
Internal sources
-
Kobe Gakuin University, Japan
Research funding
External sources
-
None, Other
No sources of support supplied
Declarations of interest
YH: none. KK: none. NW: none. TF: none. SS: none.
New
References
References to studies included in this review
Bowden 2003 {published data only}
- Bowden CL, Calabrese JR, Sachs G, Yatham LN, Asghar SA, Hompland M, et al, Lamictal 606 Study Group. A placebo-controlled 18-month trial of lamotrigine and lithium maintenance treatment in recently manic or hypomanic patients with bipolar I disorder. Archives of General Psychiatry 2003;60(4):392-400. [DOI] [PubMed] [Google Scholar]
Calabrese 1999 {published data only}
- Calabrese JR, Bowden CL, Sachs GS, Ascher JA, Monaghan E, Rudd GD, Lamictal 602 Study Group. A double-blind placebo-controlled study of lamotrigine monotherapy in outpatients with bipolar I depression. Journal of Clinical Psychiatry 1999;60(2):79-88. [DOI] [PubMed] [Google Scholar]
- Calabrese JR, Huffman RF, White RL, Edwards S, Thompson TR, Ascher JA, et al. Lamotrigine in the acute treatment of bipolar depression: results of five double-blind, placebo-controlled clinical trials. Bipolar Disorders 2008;10(2):323-33. [DOI] [PubMed] [Google Scholar]
Calabrese 2000 {published data only}
- Calabrese JR, Suppes T, Bowden CL, Sachs GS, Swann AC, McElroy SL, et al, Lamictal 614 Study Group. A double-blind, placebo-controlled, prophylaxis study of lamotrigine in rapid-cycling bipolar disorder. Journal of Clinical Psychiatry 2000;61(11):841-50. [DOI] [PubMed] [Google Scholar]
Calabrese 2003 {published data only}
- Calabrese JR, Bowden CL, Sachs G, Yatham LN, Behnke K, Mehtonen OP, et al, Lamictal 605 Study Group. A placebo-controlled 18-month trial of lamotrigine and lithium maintenance treatment in recently depressed patients with bipolar I disorder. Journal of Clinical Psychiatry 2003;64(9):1013-24. [DOI] [PubMed] [Google Scholar]
Calabrese 2008 [GW603/SCAA2010] {published data only}
- Calabrese JR, Huffman RF, White RL, Edwards S, Thompson TR, Ascher JA, et al. Lamotrigine in the acute treatment of bipolar depression: results of five double-blind, placebo-controlled clinical trials. Bipolar Disorders 2008;10(2):323-33. [DOI] [PubMed] [Google Scholar]
Calabrese 2008 [SCA100223] {published data only}
- Calabrese JR, Huffman RF, White RL, Edwards S, Thompson TR, Ascher JA, et al. Lamotrigine in the acute treatment of bipolar depression: results of five double-blind, placebo-controlled clinical trials. Bipolar Disorders 2008;10(2):323-33. [DOI] [PubMed] [Google Scholar]
- NCT00274677. Depression and bipolar disorder [A multicenter, double-blind, placebo-controlled, fixed-dose, 8-week evaluation of the efficacy and safety of lamotrigine in the treatment of depression in patients with type II bipolar disorder]. clinicaltrials.gov/ct2/show/NCT00274677 (first received 11 January 2006).
Calabrese 2008 [SCA30924] {published data only}
- Calabrese JR, Huffman RF, White RL, Edwards S, Thompson TR, Ascher JA, et al. Lamotrigine in the acute treatment of bipolar depression: results of five double-blind, placebo-controlled clinical trials. Bipolar Disorders 2008;10(2):323-33. [DOI] [PubMed] [Google Scholar]
- NCT00056277. Bipolar disorder study for men and women [Double blind placebo controlled study of lamictal in acute bipolar depression]. clinicaltrials.gov/ct2/show/NCT00056277 (first received 11 March 2003).
Calabrese 2008 [SCA40910] {published data only}
- Calabrese JR, Huffman RF, White RL, Edwards S, Thompson TR, Ascher JA, et al. Lamotrigine in the acute treatment of bipolar depression: results of five double-blind, placebo-controlled clinical trials. Bipolar Disorders 2008;10(2):323-33. [DOI] [PubMed] [Google Scholar]
Koyama 2011 {published data only}
- Koyama T, Higuchi T, Yamawaki S, Kanba S, Terao T, Shinohara A. Study SCA104779, an evaluation of BW430C (lamotrigine) versus placebo in the prevention of mood episodes in bipolar Ⅰ disorder patients. Japanese Journal of Clinical Psychiatry 2011;40:369-83. [Google Scholar]
- NCT00550407. An evaluation of BW430C (lamotrigine) versus placebo in the prevention of mood episodes in bipolar I disorder patients. clinicaltrials.gov/ct2/show/NCT00550407 (first received 27 July 2010).
Licht 2010 {published data only}
- Licht RW, Nielsen JN, Gram LF, Vestergaard P, Bendz H. Lamotrigine versus lithium as maintenance treatment in bipolar I disorder: an open, randomized effectiveness study mimicking clinical practice. The 6th trial of the Danish University Antidepressant Group (DUAG-6). Bipolar Disorders 2010;12(5):483-93. [DOI] [PubMed] [Google Scholar]
- NCT00226135. Prophylactic effect of lamotrigine compared with lithium in bipolar disorder. clinicaltrials.gov/ct2/show/NCT00226135 (first received 26 September 2005).
Suppes 2008a {published data only}
- Suppes T, Marangell LB, Bernstein IH, Kelly DI, Fischer EG, Zboyan HA, et al. A single blind comparison of lithium and lamotrigine for the treatment of bipolar II depression. Journal of Affective Disorders 2008;111(2-3):334-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
References to studies excluded from this review
Berk 1999a {published data only}
- Berk M. Lamotrigine and the treatment of mania in bipolar disorder. European Neuropsychopharmacology 1999;9:119-23. [DOI] [PubMed] [Google Scholar]
Bowden 2012a {published data only}
- Bowden CL, Singh V, Weisler R, Thompson P, Chang X, Quinones M, et al. Lamotrigine vs. lamotrigine plus divalproex in randomized, placebo-controlled maintenance treatment for bipolar depression. Acta Psychiatrica Scandinavica 2012;126(5):342-50. [DOI] [PubMed] [Google Scholar]
ChiCTR2000038166 {published data only}
- ChiCTR2000038166. Lithium carbonate and lamotrigine monotherapy or combination therapy as maintenance treatment for childbearing women with bipolar disorder in remission: a randomized controlled study. www.chictr.org.cn/showprojen.aspx?proj=61129 (first received 11 September 2020).
EudraCT 2006‐001317‐15 {published data only}
- EudraCT 2006-001317-15. BALANCE 2: Bipolar disorder:Antidepressant/Lamotrigine/ANtipsychotic Comparative Evaluation. www.clinicaltrialsregister.eu/ctr-search/trial/2006-001317-15/GB (first received 9 May 2006).
Frangou 1999 {published data only}
- Frangou S. A multicentre, double-blind, placebo-controlled, fixed-dose evaluation of the safety and efficacy of lamotrigine compared to placebo and lithium in the treatment of an acute manic episode in patients who have bipolar disorder: incorporating participants. In: National Research Register. 1999.
Gao 2020 {published data only}
- Gao K, Arnold JG, Prihoda TJ, Quinines M, Singh V, Schinagle M, et al. Sequential Multiple Assignment Randomized Treatment (SMART) for Bipolar Disorder at Any Phase od Illness and at least Mild Symptom Severity. Psychopharmacology Bulletin 2020;50(2):8-25. [NCT01588457] [DOI] [PMC free article] [PubMed] [Google Scholar]
- NCT01588457. Sequential multiple assignment treatment for bipolar disorder (SMART) [Sequential multiple assignment randomized treatment (SMART) for bipolar disorder]. clinicaltrials.gov/ct2/show/NCT01588457 (first received 1 May 2012).
Gardiner 2011 {published data only}
- Gardiner A, Rendell J, Stephens W, Hainsworth J, Yu LM, Brown S, et al. CEQUEL: A Comparative Evaluation of QUEtiapine-Lamotrigine combination versus quetiapine monotherapy, (and folic acid versus placebo) in people with bipolar depression: A 2x2 factorial randomised trial [conference abstract RC3] [ISRCTN17054996]. In: Bipolar Disorders [Abstracts of the Ninth International Conference on Bipolar Disorder. June 9-11, 2011. Pittsburgh, Pennsylvania, USA]. Vol. 13. 2011:21.
Geddes 2005 {published data only}
- Geddes JR, Rendell JM. BALANCE 2: international trial of treatment for bipolar depression. Bipolar Disorders 2005;7:57. [Google Scholar]
Geddes 2014 {published data only}
- Geddes JR, Hinds C, Rendell J, Gardiner A, Voysey M, Attenburrow MJ, et al. Comparative evaluation of quetiapine plus lamotrigine versus quetiapine monotherapy in bipolar depression: a randomized placebo controlled trial (CEQUEL). In: Neuropsychopharmacology. Vol. 39. 2014:S371.
Geddes 2015 {published data only}
- Geddes JR, Hinds C, Rendell J, Gardiner A, Voysey M, Attenburrow MJ, et al. Comparative evaluation of quetiapine plus lamotrigine versus quetiapine monotherapy in people with bipolar depression: a randomized trial (CEQUEL). In: Bipolar Disorders. Vol. 17. 2015:25.
Geddes 2015b {published data only}
- Geddes JR, Rendell J, Hinds C, Voysey M, Gardiner A, Attenburrow MJ, et al. Comparative evaluation of quetiapine plus lamotrigine versus quetiapine monotherapy in people with bipolar depression: a randomized trial (CEQUEL). Bipolar Disorders 2015;17:50-1. [Google Scholar]
Geddes 2016 {published data only}
- Geddes JR, Gardiner A, Rendell J, Voysey M, Tunbridge E, Hinds C, et al, CEQUEL Investigators and Collaborators. Comparative evaluation of quetiapine plus lamotrigine combination versus quetiapine monotherapy (and folic acid versus placebo) in bipolar depression (CEQUEL): a 2 × 2 factorial randomised trial. Lancet Psychiatry 2016;3(1):31-9. [DOI] [PubMed] [Google Scholar]
Ketter 2006 {published data only}
- Ketter TA, Greist JH, Graham JA, Roberts JN, Thompson TR, Nanry KP. The effect of dermatologic precautions on the incidence of rash with addition of lamotrigine in the treatment of bipolar I disorder: a randomized trial. Journal of Clinical Psychiatry 2006;67(3):400-6. [DOI] [PubMed] [Google Scholar]
Kwon 2001 {published data only}
- Kwon YJ, Jeong HY, Park IJ. Lamotrigine and lithium in the treatment of acute bipolar disorder. Journal of Korean Neuropsychiatric Association 2001;40(5):885-92. [Google Scholar]
NCT00074776 {published data only}
- NCT00074776. Acute treatment of bipolar II depression. clinicaltrials.gov/show/NCT00074776 (first received 22 December 2003).
NCT01195363 {published data only}
- NCT01195363. Quetiapine sr as adjunctive treatment in mixed states of bipolar disorder. clinicaltrials.gov/show/NCT01195363 (first received 6 September 2010).
NCT01587066 {published data only}
- NCT01587066. Efficacy of quetiapine xr versus divalproex on clinical outcome quality of sleep and quality of life in bipolar depression. clinicaltrials.gov/show/NCT01587066 (first received 27 April 2012).
NCT01674010 {published data only}
- NCT01674010. Safety and efficacy study of ELND005 as an adjunctive maintenance treatment in bipolar I disorder. clinicaltrials.gov/show/NCT01674010 (first posted 28 August 2012).
Parikh 2012 {published data only}
- Parikh SV, Zaretsky A, Beaulieu S, Yatham LN, Young LT, Patelis-Siotis I, et al. A randomized controlled trial of psychoeducation or cognitive-behavioral therapy in bipolar disorder: a Canadian Network for Mood and Anxiety Treatments (CANMAT) study. Journal of Clinical Psychiatry 2012;73(6):803-10. [DOI] [PubMed] [Google Scholar]
PER‐122‐12 {published data only}
- PER-122-12. A randomized, double-blind, placebo-controlled, phase 3 study to evaluate the efficacy and safety of once a day, tak-375sl 0.1, 0.4, and 0.8 mg as an adjunctive therapy to treatment-as-usual in the maintenance treatment of bipolar 1 disorder in adult subjects. www.who.int/trialsearch/Trial2.aspx?TrialID=PER-122-12.
PER‐123‐12 {published data only}
- PER-123-12. A randomized, double-blind, placebo-controlled, phase 3 study to evaluate the efficacy and safety of once a day, tak-375 (ramelteon) tablet for sublingual administration (tak-375sl tablet) 0.1, 04, and 0.8 mg as adjunctive therapy in the treatment of acute depressive episodes associated with bipolar 1 disorder in adult subjects. http://www.who.int/trialsearch/Trial2.aspx?TrialID=PER-123-12.
Simon 2018 {published data only}
- Simon J, Geddes JR, Gardiner A, Rendell J, Goodwin GM, Mayer S. Comparative economic evaluation of quetiapine plus lamotrigine combination vs quetiapine monotherapy (and folic acid vs placebo) in patients with bipolar depression (CEQUEL). Bipolar Disorders 2018;20(8):733-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
Swann 2005 {published data only}
- Swann AC. A trial analyzing polypharmacy incorporating extended-release carbamazepine in patients with bipolar I disorder. Bipolar Disorders 2005;7:105. [Google Scholar]
Tolliver 2014 {published data only}
- Tolliver BK. Treatment of co-occurring alcohol dependence and bipolar disorder: preliminary results from a randomized controlled trial of lamotrigine. Alcoholism: Clinical and Experimental Research 2014;38:212. [Google Scholar]
Tolliver 2018 {published data only}
- Tolliver B, Prisciandaro J, Baker N, Brown D, Brenner H, Brady K. A double-blind, placebo-controlled randomized controlled trial of lamotrigine in adults with co-occurring bipolar disorder and alcohol dependence: effects on drinking and mood outcomes. Bipolar Disorders 2018;20:92. [Google Scholar]
van der Loos 2009 {published data only}
- Loos ML, Mulder PG, Hartong EG, Blom MB, Vergouwen AC, Keyzer HJ, et al. Efficacy and safety of lamotrigine as add-on treatment to lithium in bipolar depression: a multicenter, double-blind, placebo-controlled trial. Journal of Clinical Psychiatry 2009;70(2):223-31. [DOI] [PubMed] [Google Scholar]
van der Loos 2011 {published data only}
- Loos ML, Mulder P, Hartong EG, Blom MB, Vergouwen AC, Noorden MS, et al. Long-term outcome of bipolar depressed patients receiving lamotrigine as add-on to lithium with the possibility of the addition of paroxetine in nonresponders: a randomized, placebo-controlled trial with a novel design. Bipolar Disorders 2011;13(1):111-7. [DOI] [PubMed] [Google Scholar]
Additional references
American Psychiatric Association 2013
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition. Arlington, VA: American Psychiatric Association. 2013. Available from dsm.psychiatryonline.org/doi/book/10.1176/appi.books.9780890425596.
Andreazza 2014
- Andreazza AC, Young LT. The neurobiology of bipolar disorder: identifying targets for specific agents and synergies for combination treatment. International Journal of Neuropsychopharmacology 2014;17(7):1039-52. [DOI] [PubMed] [Google Scholar]
BALANCE investigators and collaborators 2010
- BALANCE investigators and collaborators, Geddes JR, Goodwin GM, Rendell J, Azorin JM, Cipriani A, Ostacher MJ, et al. Lithium plus valproate combination therapy versus monotherapy for relapse prevention in bipolar I disorder (BALANCE): a randomised open-label trial. Lancet 2010;375(9712):385-95. [DOI] [PubMed] [Google Scholar]
Berk 1999b
- Berk M, Ichim L, Brook S. Olanzapine compared to lithium in mania: a double-blind randomized controlled trial. International Clinical Psychopharmacology 1999;14(6):339-43. [PMID: ] [DOI] [PubMed] [Google Scholar]
Beynon 2009
- Beynon S, Soares-Weiser K, Woolacott N, Duffy S, Geddes JR. Pharmacological interventions for the prevention of relapse in bipolar disorder: a systematic review of controlled trials. Journal of Psychopharmacology 2009;23(5):574-91. [DOI] [PubMed] [Google Scholar]
Blanco 2002
- Blanco C, Laje G, Olfson M, Marcus SC, Pincus HA. Trends in the treatment of bipolar disorder by outpatient psychiatrists. American Journal of Psychiatry 2002;159(6):1005-10. [PMID: ] [DOI] [PubMed] [Google Scholar]
Bobo 2017
- Bobo WV. The diagnosis and management of bipolar I and II disorders: clinical practice update. Mayo Clinic Proceedings 2017;92(10):1532-51. [PMID: ] [DOI] [PubMed] [Google Scholar]
Bowden 2005
- Bowden CL, Singh V. Valproate in bipolar disorder: 2000 onwards. Acta Psychiatrica Scandinavica 2005;111(Suppl 426):13-20. [PMID: ] [DOI] [PubMed] [Google Scholar]
Bowden 2012b
- Bowden CL, Singh V. Lamotrigine (Lamictal IR) for the treatment of bipolar disorder. Expert Opinion on Pharmacotherapy 2012;13(17):2565-71. [PMID: ] [DOI] [PubMed] [Google Scholar]
Calabrese 2005
- Calabrese JR, Keck PE Jr, Macfadden W, Minkwitz M, Ketter TA, Weisler RH, et al. A randomized, double-blind, placebo-controlled trial of quetiapine in the treatment of bipolar I or II depression. American Journal of Psychiatry 2005;162(7):1351-60. [PMID: ] [DOI] [PubMed] [Google Scholar]
Calabrese 2006
- Calabrese JR, Goldberg JF, Ketter TA, Suppes T, Frye M, White R, et al. Recurrence in bipolar I disorder: a post hoc analysis excluding relapses in two double-blind maintenance studies. Biological Psychiatry 2006;59(11):1061-4. [DOI] [PubMed] [Google Scholar]
Calabrese 2008
- Calabrese JR, Huffman RF, White RL, Edwards S, Thompson TR, Ascher JA, et al. Lamotrigine in the acute treatment of bipolar depression: results of five double-blind, placebo-controlled clinical trials. Bipolar Disorders 2008;10(2):323-33. [PMID: ] [DOI] [PubMed] [Google Scholar]
Dauphinais 2011
- Dauphinais D, Knable M, Rosenthal J, Polanski M, Rosenthal N. Zonisamide for bipolar disorder, mania or mixed states: a randomized, double blind, placebo-controlled adjunctive trial. Psychopharmacology Bulletin 2011;44(1):5-17. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
de Hert 2011
- Hert M, Correll CU, Bobes J, Cetkovich-Bakmas M, Cohen D, Asai I, et al. Physical illness in patients with severe mental disorders. I. Prevalence, impact of medications and disparities in health care. World Psychiatry 2011;10(1):52-77. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Egger 1997
- Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997;315(7109):629-34. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Garnett 1997
- Garnett WR. Lamotrigine: pharmacokinetics. Journal of Child Neurology 1997;12 Suppl 1:S10-5. [PMID: ] [DOI] [PubMed] [Google Scholar]
Geddes 2009
- Geddes JR, Calabrese JR, Goodwin GM. Lamotrigine for treatment of bipolar depression: independent meta-analysis and meta-regression of individual patient data from five randomised trials. British Journal of Psychiatry 2009;194(1):4-9. [PMID: ] [DOI] [PubMed] [Google Scholar]
GRADEpro GDT [Computer program]
- GRADEpro GDT. Version accessed 6 July 2021. Hamilton (ON): McMaster University (developed by Evidence Prime), accessed 6 July 2021. Available at gradepro.org.
Graham 2018
- Graham RK, Tavella G, Parker GB. Is there consensus across international evidence-based guidelines for the psychotropic drug management of bipolar disorder during the perinatal period? Journal of Affective Disorders 2018;228:216-21. [PMID: ] [DOI] [PubMed] [Google Scholar]
Greil 1998
- Greil W, Kleindienst N, Erazo N, Muller-Oerlinghausen B. Differential response to lithium and carbamazepine in the prophylaxis of bipolar disorder. Journal of Clinical Psychopharmacology 1998;18(6):455-60. [PMID: ] [DOI] [PubMed] [Google Scholar]
Guyatt 2011
- Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction – GRADE evidence profiles and summary of findings tables. Journal of Clinical Epidemiology 2011;64(4):383-94. [PMID: ] [DOI] [PubMed] [Google Scholar]
Hamilton 1960
- Hamilton M. A rating scale for depression. Journal of Neurology, Neurosurgery, and Psychiatry 1960;23:56-62. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2020
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, et al (editors). Cochrane Handbook for Systematic Reviews ofInterventions version 6.1 (updated September 2020). Cochrane,2020. Available from www.training.cochrane.org/handbook.
Johannessen 2006
- Johannessen SI, Tomson T. Pharmacokinetic variability of newer antiepileptic drugs: when is monitoring needed? Clinical Pharmacokinetics 2006;45(11):1061-75. [PMID: ] [DOI] [PubMed] [Google Scholar]
Kaufman 2004
- Kaufman KR. Monotherapy treatment of bipolar disorder with levetiracetam. Epilepsy & Behavior 2004;5(6):1017-20. [PMID: ] [DOI] [PubMed] [Google Scholar]
Keck 2003
- Keck PE Jr, Marcus R, Tourkodimitris S, Ali M, Liebeskind A, Saha A, et al, Aripiprazole Study Group. A placebo-controlled, double-blind study of the efficacy and safety of aripiprazole in patients with acute bipolar mania. American Journal of Psychiatry 2003;160(9):1651-8. [PMID: ] [DOI] [PubMed] [Google Scholar]
Ketter 2007
- Ketter TA, Jones M, Paulsson B. Rates of remission/euthymia with quetiapine monotherapy compared with placebo in patients with acute mania. Journal of Affective Disorders 2007;100 Suppl 1:S45-53. [PMID: ] [DOI] [PubMed] [Google Scholar]
Kong 2018
- Kong L, Zhou T, Wang B, Gao Z, Wang C. The risks associated with the use of lamotrigine during pregnancy. International Journal of Psychiatry in Clinical Practice 2018;22(1):2-5. [PMID: ] [DOI] [PubMed] [Google Scholar]
Lyall 2019
- Lyall LM, Penades N, Smith DJ. Changes in prescribing for bipolar disorder between 2009 and 2016: national-level data linkage study in Scotland. British Journal of Psychiatry 2019;215:415-21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Merikangas 2011
- Merikangas KR, Jin R, He JP, Kessler RC, Lee S, Sampson NA, et al. Prevalence and correlates of bipolar spectrum disorder in the world mental health survey initiative. Archives of General Psychiatry 2011;68(3):241-51. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Miura 2014
- Miura T, Noma H, Furukawa TA, Mitsuyasu H, Tanaka S, Stockton S, et al. Comparative efficacy and tolerability of pharmacological treatments in the maintenance treatment of bipolar disorder: a systematic review and network meta-analysis. Lancet Psychiatry 2014;1(5):351-9. [PMID: ] [DOI] [PubMed] [Google Scholar]
Moher 2009
- Moher D, Liberati A, Tetzlaff J, Altman DG, PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Annals of Internal Medicine 2009;151(4):264-9. [PMID: ] [DOI] [PubMed] [Google Scholar]
Montgomery 1979
- Montgomery SA, Asberg M. A new depression scale designed to be sensitive to change. British Journal of Psychiatry 1979;134(4):382-9. [PMID: ] [DOI] [PubMed] [Google Scholar]
Murray 2012
- Murray CJ, Vos T, Lozano R, Naghavi M, Flaxman AD, Michaud C, et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990-2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012;380(9859):2197-223. [DOI] [PubMed] [Google Scholar]
Ng 2007
- Ng F, Hallam K, Lucas N, Berk M. The role of lamotrigine in the management of bipolar disorder. Neuropsychiatric Disease and Treatment 2007;3(4):463-74. [PMID: ] [PMC free article] [PubMed] [Google Scholar]
Nivoli 2011
- Nivoli AM, Colom F, Murru A, Pacchiarotti I, Castro-Loli P, Gonzalez-Pinto A, et al. New treatment guidelines for acute bipolar depression: a systematic review. Journal of Affective Disorders 2011;129(1-3):14-26. [PMID: ] [DOI] [PubMed] [Google Scholar]
Nordentoft 2011
- Nordentoft M, Mortensen PB, Pedersen CB. Absolute risk of suicide after first hospital contact in mental disorder. Archives of General Psychiatry 2011;68(10):1058-64. [PMID: ] [DOI] [PubMed] [Google Scholar]
Oya 2019
- Oya K, Sakuma K, Esumi S, Hashimoto Y, Hatano M, Matsuda Y, et al. Efficacy and safety of lithium and lamotrigine for the maintenance treatment of clinically stable patients with bipolar disorder: a systematic review and meta-analysis of double-blind, randomized, placebo-controlled trials with an enrichment design. Neuropsychopharmacology Reports 2019;39(3):241-6. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Pariente 2017
- Pariente G, Leibson T, Shulman T, Adams-Webber T, Barzilay E, Nulman I. Pregnancy outcomes following in utero exposure to lamotrigine: a systematic review and meta-analysis. CNS Drugs 2017;31(6):439-50. [PMID: ] [DOI] [PubMed] [Google Scholar]
Pigott 2016
- Pigott K, Galizia I, Vasudev K, Watson S, Geddes J, Young AH. Topiramate for acute affective episodes in bipolar disorder in adults. Cochrane Database of Systematic Reviews 2016, Issue 9. Art. No: CD003384. [DOI: 10.1002/14651858.CD003384.pub3] [DOI] [PMC free article] [PubMed] [Google Scholar]
Poels 2018
- Poels EM, Bijma HH, Galbally M, Bergink V. Lithium during pregnancy and after delivery: a review. International Journal of Bipolar Disorders 2018;6(1):26. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Rücker 2008
- Rücker G, Schwarzer G, Carpenter J. Arcsine test for publication bias in meta-analyses with binary outcomes. Statistics in Medicine 2008;27(5):746-63. [PMID: ] [DOI] [PubMed] [Google Scholar]
Segal 1998
- Segal J, Berk M, Brook S. Risperidone compared with both lithium and haloperidol in mania: a double-blind randomized controlled trial. Clinical Neuropharmacology 1998;21(3):176-80. [PMID: ] [PubMed] [Google Scholar]
Smith 2007
- Smith LA, Cornelius V, Warnock A, Bell A, Young AH. Effectiveness of mood stabilizers and antipsychotics in the maintenance phase of bipolar disorder: a systematic review of randomized controlled trials. Bipolar Disorders 2007;9(4):394-412. [PMID: ] [DOI] [PubMed] [Google Scholar]
Sondergard 2008
- Sondergard L, Lopez AG, Andersen PK, Kessing LV. Mood-stabilizing pharmacological treatment in bipolar disorders and risk of suicide. Bipolar Disorders 2008;10(1):87-94. [PMID: ] [DOI] [PubMed] [Google Scholar]
Suppes 2008b
- Suppes T, Marangell LB, Bernstein IH, Kelly DI, Fischer EG, Zboyan HA, et al. A single blind comparison of lithium and lamotrigine for the treatment of bipolar II depression. Journal of Affective Disorders 2008;111(2-3):334-43. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Vazquez 2015
- Vazquez GH, Holtzman JN, Lolich M, Ketter TA, Baldessarini RJ. Recurrence rates in bipolar disorder: systematic comparison of long-term prospective, naturalistic studies versus randomized controlled trials. European Neuropsychopharmacology 2015;25(10):1501-12. [PMID: ] [DOI] [PubMed] [Google Scholar]
Veroniki 2017
- Veroniki AA, Cogo E, Rios P, Straus SE, Finkelstein Y, Kealey R, et al. Comparative safety of anti-epileptic drugs during pregnancy: a systematic review and network meta-analysis of congenital malformations and prenatal outcomes. BMC Medicine 2017;15(95):1-20. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Verrotti 2018
- Verrotti A, Striano P, Iapadre G, Zagaroli L, Bonanni P, Coppola G, et al. The pharmacological management of Lennox-Gastaut syndrome and critical literature review. Seizure 2018;63:17-25. [PMID: ] [DOI] [PubMed] [Google Scholar]
Vieta 2006
- Vieta E, Manuel Goikolea J, Martinez-Aran A, Comes M, Verger K, Masramon X, et al. A double-blind, randomized, placebo-controlled, prophylaxis study of adjunctive gabapentin for bipolar disorder. Journal of Clinical Psychiatry 2006;67(3):473-7. [PMID: ] [DOI] [PubMed] [Google Scholar]
Viguera 2000
- Viguera AC, Nonacs R, Cohen LS, Tondo L, Murray A, Baldessarini RJ. Risk of recurrence of bipolar disorder in pregnant and nonpregnant women after discontinuing lithium maintenance. American Journal of Psychiatry 2000;157(2):179-84. [PMID: ] [DOI] [PubMed] [Google Scholar]
Ware 1993
- Ware JE, Snow KK, Kosinski M, Gandek B. SF-36 Health Survey: Manual and Interpretation Guide. Boston (MA): The Health Institute, New England Medical Centre, 1993. [Google Scholar]
Wesseloo 2017
- Wesseloo R, Liu X, Clark CT, Kushner SA, Munk-Olsen T, Bergink V. Risk of postpartum episodes in women with bipolar disorder after lamotrigine or lithium use during pregnancy: a population-based cohort study. Journal of Affective Disorders 2017;218:394-7. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
WHO 2019a
- World Health Organization. Mental disorders. www.who.int/news-room/fact-sheets/detail/mental-disorders (accessed 6 July 2021).
WHO 2019b
- World Health Organization. ICD-11. International Classification of Diseases 11th Revision. icd.who.int/en (accessed 6 July 2021).
Yatham 2018
- Yatham LN, Kennedy SH, Parikh SV, Schaffer A, Bond DJ, Frey BN, et al. Canadian Network for Mood and Anxiety Treatments (CANMAT) and International Society for Bipolar Disorders (ISBD) 2018 guidelines for the management of patients with bipolar disorder. Bipolar Disorders 2018;20(2)::97-170. [DOI] [PMC free article] [PubMed] [Google Scholar]
Yildiz 2011
- Yildiz A, Vieta E, Leucht S, Baldessarini RJ. Efficacy of antimanic treatments: meta-analysis of randomized, controlled trials. Neuropsychopharmacology 2011;36(2):375-89. [PMID: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Young 1978
- Young RC, Biggs JT, Ziegler VE, Meyer DA. A rating scale for mania: reliability, validity and sensitivity. British Journal of Psychiatry 1978;133:429-35. [DOI] [PubMed] [Google Scholar]
References to other published versions of this review
Hashimoto 2020
- Hashimoto Y, Kotake K, Watanabe N, Fujiwara T, Sakamoto S. Lamotrigine in the maintenance treatment of bipolar disorder. Cochrane Database of Systematic Reviews 2020, Issue 4. Art. No: CD013575. [DOI: 10.1002/14651858.CD013575] [DOI] [PMC free article] [PubMed] [Google Scholar]


