Abstract
Background and purpose
The aim was to evaluate potential predictive factors of smell recovery in a clinical series of 288 patients presenting olfactory dysfunction (OD) related to coronavirus disease 2019 (COVID‐19). Potential correlations were sought between epidemiological, clinical and immunological characteristics of patients and the persistence of OD at 60 days.
Methods
COVID‐19 positive patients presenting OD were prospectively recruited from three European hospitals. Baseline clinical and olfactory evaluations were performed within the first 2 weeks after OD onset and repeated at 30 and 60 days. In a subgroup of patients, anti‐severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) antibodies were measured in serum, saliva and nasal secretions at 60 days.
Results
A total of 288 COVID‐19 patients with OD were included in the study. Two weeks after the onset of the loss of smell, 52.4% of patients had OD on psychophysical tests, including 113 cases (39.2%) of anosmia and 38 cases (13.2%) of hyposmia. At 60‐day follow‐up, 25.4% of the patients presented persistent OD. There was no significant correlation between sex, age, viral load on nasopharyngeal swab or COVID‐19 severity and poor olfactory outcome. In a subgroup of 63 patients, it was demonstrated that patients with poor olfactory outcomes at 60 days had lower levels of salivary and nasal immunoglobulin G (IgG) and IgG1, but similar levels of antibodies in the serum.
Conclusions
No clinical markers predicted the evolution of OD at 60 days. Patients with poor olfactory outcome at 60 days had lower saliva and nasal antibodies, suggesting a role for local immune responses in the persistence of COVID‐19 related OD.
Keywords: COVID‐19, IgG, IgG1, olfactory dysfunction, SARS‐COV‐2
The aim of this study was to evaluate potential predictive factors of smell recovery in a clinical series of 288 patients presenting olfactory dysfunction (OD) related to COVID‐19. No clinical markers predicted the evolution of OD at 60 days. Patients with poor olfactory outcome at 60 days had lower saliva and nasal antibodies, suggesting a role for local immune responses in the persistence of COVID‐19 related OD

INTRODUCTION
Olfactory dysfunction (OD) is one of the most frequent clinical manifestations of coronavirus disease 2019 (COVID‐19), affecting more than 70% of patients infected with severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) [1, 2, 3, 4]. In the vast majority of patients, these disorders completely subside within a few weeks [5, 6, 7]. However, the first long‐term follow‐up studies are showing a prevalence of persistent severe olfactory disturbances ranging between 4.5% and 11% 6 months after onset of symptoms [8, 9, 10, 11]. This frequency, given the high prevalence of infection in the population, means that in the near future there could be a very high number of subjects with long‐term morbidity and a significant impact on quality of life [12].
The pathogenesis of OD in COVID‐19 seems to be linked to inflammatory responses involving support cells of the olfactory epithelium which, in some patients, can persist after the end of the infection with progressive damage to the sustentacular cells and olfactory neurons [7, 13, 14, 15, 16, 17, 18]. On this basis, corticosteroids were proposed to interrupt this pathological process before it affects cells that do not have regenerative capacity [19, 20, 21, 22]. To guide therapeutic interventions and avoid over‐treatment of patients who will spontaneously recover, it is necessary to identify risk factors of persistent of OD. Unfortunately, no risk factor has been identified yet.
The aim of this multicentre study was to evaluate whether epidemiological, clinical and immunological characteristics of the patients correlate with the persistence of anosmia or severe hyposmia at 60 days from clinical onset.
MATERIALS AND METHODS
This prospective study was approved by three Institutional Review Boards (CHU Saint‐Pierre, Brussels, Belgium, CHUSP210207; Epicura, Baudour, Belgium, P2020011; Sassari University Hospital, Italy, PG/2021/5471). Patients provided written electronic or paper informed consent.
From 22 March 2020 to 15 November 2020 patients with a diagnosis of COVID‐19 and OD confirmed by psychophysical tests were recruited within 2 weeks of onset of symptoms in three European hospitals, in Belgium and Italy. In Belgian hospitals patients were recruited between March and May 2020, and in Sassari Hospital patients were recruited from May to November 2020. SARS‐CoV‐2 infection was diagnosed by nasal swabbing and reverse transcriptase polymerase chain reaction (RT‐PCR) or serology (immunoglobulin M [IgM] and/or IgG). All patients were invited to perform olfactory training at least twice daily with >3 daily odours (e.g., coffee, perfume, essential oils) for each olfactory training session.
Epidemiological and clinical outcomes
The following epidemiological data were collected through a standardized online questionnaire or medical records: gender, age, smoking and comorbidities. The following comorbidities were evaluated: hypercholesterolaemia, reflux, heart, respiratory, kidney, liver, autoimmune diseases, diabetes, neurological, hypertension, allergy, chronic rhinitis, chronic rhinosinusitis, depression, thyroid and skin disorders. Symptoms were evaluated during the clinical course of the disease with the COVID‐19 Symptom Index, which is a 100‐point clinical instrument evaluating common COVID‐19 symptoms [23]. In addition, nasal symptoms and the potential relationship between nasal complaints and OD were assessed with the French and Italian versions of the sinonasal outcome tool 22 (SNOT‐22) [24].
Inclusion criteria consisted of adults with SARS‐CoV‐2 infection identified through nasal swabs and positive RT‐PCR or positive SARS‐CoV‐2 serology and COVID‐19 related OD. Patients with the following criteria were excluded: normosmia at the psychophysical evaluation, history of pre‐COVID‐19 pandemic OD, chronic or self‐reported acute rhinosinusitis (with regard to the European Position Paper on Rhinosinusitis and Nasal Polyps guidelines) at the time of evaluation, dementia, or other conditions associated with an inability to complete the evaluations.
Subjective and psychophysical olfactory evaluations
Subjective olfactory and gustatory functions were evaluated with the smell and taste component of the National Health and Nutrition Examination Survey, as previously described [9]. Psychophysical olfactory assessments were performed with the identification component of Sniffin’ Sticks tests (Medisense, Groningen, Netherlands), which is a validated psychophysical olfactory test using 16 smell pens. Each pen is presented to individuals who have to choose the adequate smell between four given options. The final score ranges from 0 (none correctly identified) to 16 (all correctly identified). Normative values established normosmia as a score ranging between 12 and 16, hyposmia between 9 and 11 and anosmia between 0 and 8 [25].
Study design
Figure 1 describes the chart flow of our study. For patients suffering from mild COVID‐19, baseline clinical and olfactory evaluations were performed within the first 2 weeks after OD onset and repeated 1 and 2 months later. For patients suffering from severe COVID‐19, it was not possible to perform objective olfactory evaluations during the hospitalization (due to the sanitary situation) but baseline clinical and subjective olfaction were assessed. At 2 months after OD onset, psychophysical olfactory evaluations were performed.
FIGURE 1.

The flow chart of our study including 288 COVID‐19 patients with olfactory dysfunction (OD). From 22 March 2020 to 15 November 2020, patients with a confirmed diagnosis of COVID‐19 and OD confirmed by psychophysical tests were recruited within 2 weeks of onset of symptoms in three European hospitals, in Belgium and Italy. Baseline clinical and semi‐objective olfactory evaluations of patients suffering from mild COVID‐19 were performed within the first 2 weeks after OD onset and repeated 1 and 2 months later. For patients suffering from severe COVID‐19, it was not possible to perform semi‐objective olfactory evaluations during hospitalization (due to the sanitary situation) but baseline clinical and subjective olfaction evaluations were assessed. Then, semi‐objective olfactory evaluations were assessed at 2 months after OD onset. Patients from Epicura Hospital (Baudour, Belgium) participated in a nested study which aimed to assess the antibody responses to SARS‐CoV‐2 in the serum, saliva and nasal secretions 2 months after COVID‐19 onset
Patients from Epicura Hospital (Baudour, Belgium) participated in a nested study which aimed to assess the antibody responses to SARS‐CoV‐2 in the serum, saliva and nasal secretions 2 months after COVID‐19 onset. During this visit, patients were invited to perform a sniffing test and to donate blood, saliva and nasal secretion samples (nasal washing with physiological saline solution was carried out and nasal secretions were sterile aspirated).
Definition of good or poor evolution of COVID‐19 related olfactory dysfunction
Two groups of patients were identified having either a good evolution or a persistence of severe OD. The ‘good olfactory outcome’ group corresponds to patients presenting either a normal sniffing test (i.e., ≥12/16) at one of the tests carried out during the 2 months of follow‐up (the sniffing test being performed at 15, 30 and 60 days of the onset OD, except for patients presenting a severe form of COVID‐19 for whom the sniffing test was performed at 2 months) or a sniffing test ≥10 at 60 days. The ‘poor olfactory outcome’ group corresponds to patients with a score between 0 and 9 on the sniffing test carried out 2 months after the onset of symptoms.
SARS‐CoV‐2 IgG and IgA assays
Recombinant SARS‐CoV‐2 spike antigens S1, S2 and RBD, purchased from SinoBiological (40591‐VO8H, 40590‐VO8B and 40592‐VNAH respectively), and nucleocapsid protein (NP) (kindly provided by André Matagne, University of Liège) were covalently coupled to fluorescent magnetic beads (Luminex Corporation) by a two‐step carbodiimide reaction, with 5 µg of antigens for 1 million beads. 1000 beads/antigen/well diluted in assay wash buffer (PBS‐1X, 0.1% BSA, 0.05% Tween 20, pH 7.4) was used for antibody screening. Based on preliminary experiments, the serum/mucosal fluid dilution giving the best signal‐to‐noise ratio for IgG, IgG1 and IgG3 was 1/200 and 1/50 for IgA. PBS‐diluted serum/mucosal fluid samples were incubated for 2 h at room temperature on an orbital shaker with antigen‐coupled beads. Antigen‐specific antibody titres were detected using a phycoerythrin‐coupled detection antibody for each subclass and isotype, namely IgG, IgG1, IgG3 and IgA from Southern Biotech (2040‐09, 9052‐09, 9210‐09 and 2050‐09 respectively) at a final concentration of 0.65 µg/ml. Detection antibodies were incubated with the complex antigen‐coated bead‐antigen‐specific antibody for 1 h at room temperature on an orbital shaker. Antigen–antibody binding was measured using BioPlex‐200 equipment (Bio‐Rad) and the results were expressed as median fluorescence intensity.
Statistical analyses
Statistical analyses were performed using the Statistical Package for the Social Sciences for Windows version 22.0 (IBM Corp.). According to the type of outcomes, the following tests were used to compare severity groups: Kruskal–Wallis, Fisher's exact test and Mann–Whitney U test. The pre‐ to post‐intervention changes in Sniffin’ Sticks tests were evaluated within group with the Mann–Whitney U test whilst the inter‐group differences were assessed through the differences between Sniffin’ Sticks test scores by means of the Wilcoxon signed‐rank test. Multivariate analysis was used to study the associations between outcomes.
RESULTS
In all, 288 COVID‐19 patients were included in the study (Table 1). The cohort included 101 men (35.1%) and 187 women (64.9%) with an average age of 44.4 ± 13.2 years. The most frequently reported symptoms were asthenia (143 cases, 49.6%), headache (131 cases, 45.5%), cough (127 cases, 44.1%) and myalgia (114 cases, 39.6%). COVID‐19 was staged as mild in 208 patients, moderate in 31 and severe in 49 cases according to Tian et al. [26]. At the time of infection, 52.4% of patients had OD on psychophysical tests, including 113 cases (39.2%) of anosmia and 38 cases (13.2%) of hyposmia. Mean olfactory score was 9.6 ± 4.6. At 60‐day follow‐up, 9.4% and 16% of the patients were anosmic and hyposmic, respectively, whilst 74.6% of the subjects had normal olfactory function (Table 2). The mean olfactory score at the end of the observation period was 12.7 ± 2.8. Overall, 34 subjects (11.8%) therefore presented a poor outcome (i.e., anosmia or severe hyposmia) at 60 days.
TABLE 1.
General and clinical features of the study population
| No. of patients | 288 |
|
Gender No. of patients (%) | |
| Male | 101 (35.1%) [95% CI 29.6%–40.9%] |
| Female | 187 (64.9%) [95% CI 59.1%–70.4%] |
|
Age (years) Mean ± SD |
44.4 ± 13.2 |
|
Clinical severity No. of patients (%) | |
| Mild | 203 (70.5%) [95% CI 64.8%–75.7%] |
| Moderate | 31 (10.8%) [95% CI 7.4%–14.9%] |
| Severe | 49 (17%) [95% CI 12.9%–21.9%] |
| Missing data | 5 (1.7%) [95% CI 0.06%–4%] |
|
Cycle threshold (N = 51) Mean ± SD |
31.4 ± 8.1 |
|
Referred symptoms (N = 194) No. of patients (%) | |
| Asthenia | 143 (73.7%) [95% CI 66.9%–79.8%] |
| Headache | 131 (67.5%) [95% CI 60.4%–74.1%] |
| Cough | 127 (65.5%) [95% CI 58.3%–72.1%] |
| Myalgia | 114 (58.8%) [95% CI 51.5%–65.8%] |
| Nasal obstruction | 108 (55.7%) [95% CI 48.4%–62.8%] |
| Loss of appetite | 105 (54.1%) [95% CI 46.8%–61.3%] |
| Fever | 93 (47.9%) [95% CI 40.7%–55.2%] |
| Diarrhoea | 92 (47.4%) [95% CI 40.2%–54.7%] |
| Arthralgia | 86 (44.3%) [95% CI 37.2%–51.6%] |
| Chest pain | 82 (42.3%) [95% CI 35.2%–49.5%] |
| Dyspnoea (assessed on 121 patients) | 49 (40.5%) [95% CI 31.7%–39.8%] |
| Sore throat | 60 (30.9%) [95% CI 24.5%–37.9%] |
| Sticky mucous | 52 (26.8%) [95% CI 20.7%–33.6%] |
| Abdominal pain | 49 (25.3%) [95% CI 19.3%–32%] |
| Nausea | 47 (24.2%) [95% CI 18.4%–30.9%] |
| Voice issues | 45 (23.2%) [95% CI 17.4%–29.8%] |
| Face pain | 43 (22.2%) [95% CI 16.5%–28.7%] |
| Conjunctivitis | 42 (21.6%) [95% CI 16.7%–28.1%] |
| Dysphagia | 38 (19.6%) [95% CI 14.2%–25.9%] |
Abbreviation: CI, confidence interval.
TABLE 2.
Olfactory function assessment results
| Baseline | |
|
Sniffin' Sticks score Mean ± SD |
9.6 ± 4.6 |
| Normal | 137 (47.6%) [95% CI 41.7%–53.5%] |
| Hyposmia | 38 (13.2%) [95% CI 9.5%–17.7%] |
| Anosmia | 113 (39.2%) [95% CI 33.6%–45.1%] |
| 60 days | |
|
Sniffin' Sticks score Mean ± SD |
12.7 ± 2.8 |
| Normal | 215 (74.6%) [95% CI 69.2%–79.6%] |
| Hyposmia | 46 (16%) [95% CI 11.9%–20.7%] |
| Anosmia | 27 (9.4%) [95% CI 6.3%–13.3%] |
Abbreviation: CI, confidence interval.
As indicated in Table 3, there was no significant association between poor olfactory outcome and female gender (odds ratio [OR] 0.605, 95% confidence interval [CI] 0.293–1.248; p = 0.174), age (OR 0.997, 95% CI 0.970–1.025; p = 0.832), viral load on nasopharyngeal swab (OR 1.048, 95% CI 0.942–1.165; p = 0.393) or COVID‐19 severity (χ 2(1, N = 283) = 5.97, p = 0.08). Persistent OD was inversely associated with the presence of cough (OR 0.355, 95% CI 0.146–0.860; p = 0.022), dyspnoea (OR 0.176, 95% CI 0.038–0.815; p = 0.026), asthenia (OR 0.400, 95% CI 0.163–0.981; p = 0.045) and voice alterations (OR 0.127, 95% CI 0.016–0.971; p = 0.047).
TABLE 3.
Cross‐tabulation and logistic regression analysis for clinical characteristics
| Odds ratio | 95% confidence interval | p value | ||
|---|---|---|---|---|
| Upper limit | Lower limit | |||
| Female gender | 0.605 | 0.293 | 1.248 | 0.174 |
| Age | 0.997 | 0.970 | 1.025 | 0.832 |
| Viral load | 1.048 | 0.942 | 1.165 | 0.393 |
| Asthenia | 0.400 | 0.163 | 0.981 | 0.045 |
| Headache | 0.691 | 0.281 | 1.698 | 0.421 |
| Cough | 0.355 | 0.146 | 0.860 | 0.022 |
| Myalgia | 0.488 | 0.202 | 1.117 | 0.11 |
| Nasal obstruction | 0.358 | 0.617 | 3.807 | 0.358 |
| Loss of appetite | 0.452 | 0.169 | 1.204 | 0.112 |
| Fever | 0.422 | 0.165 | 1.079 | 0.072 |
| Diarrhoea | 0.661 | 0.273 | 1.622 | 0.371 |
| Arthralgia | 0.577 | 0.319 | 1.891 | 0.577 |
| Chest pain | 0.685 | 0.276 | 1.701 | 0.415 |
| Dyspnoea (assessed on 121 patients) | 0.176 | 0.038 | 0.815 | 0.026 |
| Sore throat | 0.758 | 0.283 | 2.03 | 0.582 |
| Sticky mucous | 0.133 | 0.017 | 1.022 | 0.525 |
| Abdominal pain | 0.795 | 0.279 | 2.270 | 0.669 |
| Nausea | 0.847 | 0.296 | 2.42 | 0.756 |
| Voice issues | 0.127 | 0.016 | 0.971 | 0.047 |
| Face pain | 0.138 | 0.018 | 1.059 | 0.057 |
| Conjunctivitis | 0.737 | 0.236 | 2.297 | 0.599 |
| Dysphagia | 0.579 | 0.163 | 2.058 | 0.399 |
Relationship between OD recovery and serum IgG, IgG1, IgG3 and IgA responses
The relationship between olfactory outcome and the magnitude of serum IgG and IgA responses to SARS‐CoV‐2 antigens was analysed in a subgroup of 144 patients (18 patients with poor recovery versus 126 with good recovery). Serum IgG, IgG1, IgG3 and IgA responses were not significantly different in patients with good or poor olfactory outcome at 2 months (Figure 2a–d).
FIGURE 2.

Relationship between OD recovery and serum IgG, IgG1, IgG3 and IgA responses: serum IgG and IgA responses to SARS‐CoV‐2 antigens were analysed in a subgroup of 144 patients (18 patients with poor recovery vs. 126 with good recovery). Serum IgG, IgG1, IgG3 and IgA responses were not significantly different in patients with good or poor olfactory outcome at 2 months. The results are expressed as median fluorescence intensity (MFI). Red box plots correspond to patients with poor olfactory outcomes at 60 days whereas black box plots correspond to patients with good olfactory outcomes at 60 days. ns, not significant
Relationship between OD recovery and salivary IgG, IgG1, IgG3 and IgA responses
The relationship between olfactory outcome and the magnitude of salivary IgG, IgG1, IgG3 and IgA responses to SARS‐CoV‐2 antigens was analysed in a subgroup of 63 patients (15 patients with persistent severe OD vs. 48 with complete recovery). (Figure 3a) shows that the poor olfactory outcome group had lower levels of saliva S1 (Mann–Whitney, p = 0.037) and RBD IgG (Mann–Whitney, p = 0.035) than the good outcome group. Patients from the poor olfactory outcome group also presented lower levels of saliva S1 (p = 0.0055), RBD (p = 0.007), S2 (p = 0.03) and NP (p = 0.008) IgG1 compared to the good olfactory outcome group (Figure 3b). No association between olfactory outcome and salivary IgG3 or IgA responses to SARS‐CoV‐2 antigens was observed.
FIGURE 3.
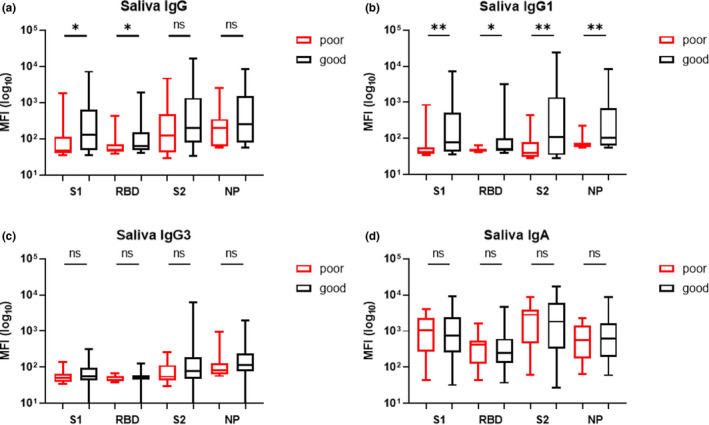
Relationship between OD recovery and salivary IgG (a), IgG1 (b), IgG3 (c) and IgA (d) responses to SARS‐CoV‐2 antigens in a subgroup of 63 patients (15 patients with persistent severe OD vs. 48 with good olfactory outcomes). (a) Patients with poor olfactory outcome had lower levels of saliva S1 and RBD IgG than patients with good olfactory outcomes. Patients from the poor olfactory outcome group also presented lower levels of saliva S1, RBD, S2 and NP IgG1 compared to the good olfactory outcome group (b). No association between olfactory outcome and salivary IgG3 (c) or IgA (d) responses to SARS‐CoV‐2 antigens was observed. The results are expressed as median fluorescence intensity (MFI). Red box plots correspond to patients with poor olfactory outcomes at 60 days whereas black box plots correspond to patients with good olfactory outcomes at 60 days. ns, not significant. *p < 0.05; **p < 0.01
Relationship between OD recovery and nasal IgG, IgG1, IgG3 and IgA responses
In a subgroup of 66 patients (15 patients with persistent severe OD vs. 51 with good recovery), nasal IgG and IgG1 responses were associated with 2 months’ olfactory recovery. Individuals from the poor olfactory outcome group had lower levels of nasal RBD (Mann–Whitney, p = 0.032), S2 (Mann–Whitney, p = 0.02) and NP IgG (p = 0.024) than patients from the good olfactory outcome group. Individuals from the poor olfactory outcome group also had lower levels of nasal S1 (p = 0.037) and S2 (p = 0.036) IgG1 than the good olfactory outcome group (Figure 4b). A similar trend was observed with nasal RBD (p = 0.051) and NP IgG3 (p = 0.08) whereas no association between olfactory recovery and nasal IgA was observed.
FIGURE 4.

Relationship between OD recovery and nasal IgG (a), IgG1 (b), IgG3 (c) and IgA (d) responses to SARS‐CoV‐2 antigens in a subgroup of 66 patients (15 patients with persistent severe OD vs. 51 with good olfactory outcomes at 60 days). (a) Individuals from the poor olfactory outcome group had lower levels of nasal RBD, S2 and NP IgG than patients from the good olfactory outcome group. The poor olfactory outcome group also had lower levels of nasal S1 and S2 IgG1 than the good olfactory outcome group (b). A similar trend was observed with nasal RBD and NP IgG3 (c). No association was observed between olfactory recovery and nasal IgA response (d). The results are expressed as median fluorescence intensity (MFI). Red box plots correspond to patients with poor olfactory outcomes at 60 days whereas black box plots correspond to patients with good olfactory outcomes at 60 days. ns, not significant. *p < 0.05
DISCUSSION
With 152 million cases of COVID‐19 (3 May 2021) and based on the meta‐analysis of Saniasiaya et al. [27] reporting a prevalence of OD of 48%, it can be estimated that worldwide over 70 million people will present with OD. Amongst these, it seems crucial to be able to detect patients at risk of prolonged anosmia or hyposmia. In this regard, it has recently been described in a clinical series of 751 COVID‐19 patients with OD that 37% still reported a persistent subjective loss of smell and 14% reported partial recovery after a mean follow‐up of 47 days [28]. Using objective olfactory evaluations, it was shown also in a clinical series of 118 COVID‐19 patients that 15% did not fully recover olfaction at 60 days [9]. The first 6‐month follow‐up studies reported a prevalence of severe and persistent OD ranging between 4.5% and 11% [8, 9, 10, 11, 29]. This high incidence of OD persistence calls for therapies that could prevent this long‐term morbidity. Considering the likely role of progressive inflammatory damage to the olfactory epithelium [14, 16], these therapies could potentially be more effective if started as early as possible after onset of symptoms [18, 19, 20, 21, 22]. However, some authors reported that micro‐haemorrhage might be another cause of persistent OD [30]. The use of corticosteroids in these patients can be detrimental due to their anti‐angiogenic effect which can worsen the vascular supply resulting in induced hypoxia. Moreover, as many patients recover quickly, guidelines published on the management of COVID‐19 OD propose use of oral corticosteroids as an option in those with symptoms persistent for more than 2 weeks [31]. Guiding the best use of these ‘optional’ interventions requires the identification of predictive factors associated with poor functional recovery. However, no clinical, epidemiological or laboratory risk factors have yet been identified [32, 33]. In this study including 288 patients with COVID‐19 OD, it is confirmed that no clinical or epidemiological factor predicts the evolution of OD at 2 months. Patients with OD must therefore be counselled on the potential risks versus benefits of corticosteroids and the chance of spontaneous recovery before use of corticosteroids; this remains a difficult decision for both clinicians and patients and is an area where further research is needed to guide decision making.
Based on the lack of relationship between severity and duration of OD and systemic inflammatory markers, it has been hypothesized that local factors are involved in the persistence of OD [15]. However, there is a shortage of studies investigating the correlation between immune or viral local factors and the persistence of OD. Cho et al. [34] investigated the relationship between viral load at baseline nasopharyngeal swab and persistence of OD without detecting significant correlations. The first pathological studies on patients with COVID‐19 induced OD revealed a destruction of the olfactory epithelium associated with chronic inflammatory infiltrate [14, 16]. Such persistent inflammation could be caused by the persistence of virus in the olfactory epithelium even after recovery from the infection. Dias De Melo et al. [35], in a study of four patients with COVID‐19 related OD for more than 3 months, did not detect SARS‐CoV‐2 RNA in nasopharyngeal samples, but all patients had detectable SARS‐CoV‐2 RNA in olfactory mucosa cytological samples. This finding could be related to an inefficient immune response at the level of the olfactory epithelium and stresses the possibility that antiviral treatments may have an effect on promoting the recovery of olfactory function. Recently, Zhou et al. [36] treated 77 COVID‐19 patients with nebulized interferon α2b (IFN‐α2b). Interestingly, IFN‐α2b significantly reduced the duration of elevated blood levels of interleukin‐6 and C‐reactive protein. Based on these preliminary results, it could be supposed that patients with severe OD could benefit from taking nebulized IFN‐α2b.
Whilst information on systemic immunity to SARS‐CoV‐2 continues to rapidly accrue [37, 38], the role of mucosal immunity within the nasal cavities and the upper respiratory tract, the primary site of SARS‐CoV‐2 infection and replication, remains largely unexplored. Butler et al. [39] and Cervia et al. [40] found an inverse correlation between the levels of serum and nasal antibody levels and severity of COVID‐19. Patients with severe disease had higher serum antibody levels whilst nasal antibodies were higher in milder disease.
Our study indicates that patients with good olfactory outcomes at 60 days had higher levels of salivary and nasal IgG and IgG1 compared to patients with poor outcomes. Interestingly, no association was observed with the levels of serum antibodies, suggesting a role for local immune response. Although IgG is often considered as a systemic antibody, it is increasingly recognized that it is also produced at the mucosal level and can participate in mucosal immunity to pathogens [41]. In contrast to IgG, the levels of mucosal IgA were not different in the two groups. This dissociation between the two types of antibodies is intriguing and could be related to a more transient production of IgA compared to IgG.
The inverse association between local IgG levels and duration of OD suggests a role for antibodies in the control of SARS‐CoV‐2 replication and prevention of epithelial damage. Indeed, studies have shown that in patients with mild infection and olfactory impairment, viral clearance was delayed compared to patients with normal olfaction [42]. Longitudinal studies are needed to characterize the antibody response to SARS‐CoV‐2 soon after the onset of symptoms and to identify the predictive value of these parameters for the duration of OD.
Our study has some limitations. As already mentioned, there were only a small number of patients with poor olfactory outcome and further studies with larger case series will be needed to confirm these results. Secondly, the 60‐day follow‐up is still too short to declare the OD permanent, and these patients may still recover. However, other authors have reported that improvements in olfactory scores tend to become non‐significant 2 months after clinical onset [10, 11]. In order to minimize contact time, only the identification component of the Sniffin’ Sticks test was performed, and it is possible that whilst some patients had normal function with this component of the test they could have persistent threshold deficits that might impair their quality of life.
CONCLUSIONS
In patients with a loss of smell related to COVID‐19, no clinical markers predicted the evolution of symptoms at 60 days. Patients with poor olfactory outcome at 60 days had lower saliva and nasal antibodies, suggesting a role for local immune responses in the persistence of COVID‐19 related OD.
CONFLICT OF INTEREST
None of the authors has a financial interest in any of the products, devices or drugs mentioned in this paper.
AUTHOR CONTRIBUTIONS
Sven Saussez: Conceptualization (equal); data curation (equal); formal analysis (equal); investigation (equal); methodology (equal); project administration (equal); supervision (equal); validation (equal); writing—original draft (equal); writing—review and editing (equal). Shilpee Sharma: Data curation (equal); investigation (equal); methodology (equal); resources (equal). Anais Thiriard: Data curation (equal); formal analysis (equal); investigation (equal); methodology (equal); software (equal). Veronique Olislagers: Data curation (equal); resources (equal). Ines Vu Duc: Data curation (equal); resources (equal); software (equal). Serge Daniel Le Bon: Data curation (supporting); investigation (supporting). Mohamad Khalife: Data curation (supporting); investigation (supporting); project administration (supporting). Stephane Hans: Writing—review and editing (supporting). Giacomo De Riu: Conceptualization (supporting); data curation (supporting). Claire Hopkins: Writing—review and editing (supporting). Jerome Lechien: Conceptualization (equal); data curation (equal); methodology (supporting). Luigi Vaira: Conceptualization (equal); data curation (equal); investigation (equal); methodology (equal); writing—original draft (equal); writing—review and editing (equal). Arnaud Marchant: Conceptualization (equal); formal analysis (equal); funding acquisition (equal); writing—review and editing (equal).
ETHICAL APPROVAL
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
ACKNOWLEDGEMENTS
The study was co‐funded by the F.R.S.‐FNRS and the Université libre de Bruxelles, Belgium. Arnaud Marchant is Research Director of the F.R.S.‐FNRS, Belgium.
Saussez S, Sharma S, Thiriard A, et al. Predictive factors of smell recovery in a clinical series of 288 coronavirus disease 2019 patients with olfactory dysfunction. Eur J Neurol. 2021;28:3702–3711. 10.1111/ene.14994
Dr Saussez and Dr Sharma have equally contributed to this work and should be regarded as joint first authors. Dr Vaira and Dr Marchant have equally contributed to the work supervision and are co‐senior authors.
DATA AVAILABILITY STATEMENT
Research data are not shared.
REFERENCES
- 1. Parma V, Ohla K, Veldhuizen MG, et al. More than smell—COVID‐19 is associated with severe impairment of smell, taste, and chemesthesis. Chem Senses. 2020;45:609‐622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Spinato G, Fabbris C, Polesel J, et al. Alterations in smell or taste in mildly symptomatic outpatients with SARS‐CoV‐2 infection. JAMA. 2020;323:2089‐2090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lechien JR, Chiesa‐Estomba CM, Vaira LA, et al. Epidemiological, otolaryngological, olfactory and gustatory outcomes according to the severity of COVID‐19: a study of 2579 patients. Eur Arch Otorhinolaryngol. 2021;1–9. 10.1007/s00405-020-06548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Vaira LA, Lechien JR, Khalife M, et al. Psychophysical evaluation of the olfactory function: European multi‐center study on 774 COVID‐19 patients. Pathogens. 2021;10:E62. 10.3390/pathogens10010062 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Lechien JR, Journe F, Hans S, et al. Severity of anosmia as an early symptom of COVID‐19 infection may predict lasting loss of smell. Front Med (Lausanne). 2020;7:582802. 10.3389/fmed.2020.582802 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Parente‐Arias P, Barreira‐Fernandez P, Quintana‐Sanjuas A, Patiño‐Castiñeira B. Recovery rate and factors associated with smell and taste disruption in patients with coronavirus disease 2019. Am J Otolaryngol. 2020;42:102648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Vaira LA, Hopkins C, Petrocelli M, et al. Smell and taste recovery in coronavirus disease 2019 patients: a 60‐day objective and prospective study. J Laryngol Otol. 2020;134:703‐709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Boscolo‐Rizzo P, Menegaldo A, Fabbris C, et al. Six‐month psychophysical evaluation of olfactory dysfunction in patients with COVID‐19. Chem Senses. 2021;46:1–9. 10.1093/chemse/bjab006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Lechien JR, Chiesa‐Estomba CM, Beckers E, et al. Prevalence and 6‐month recovery of olfactory dysfunction: a multicentre study of 1363 COVID‐19 patients. J Intern Med. 2021;1–11. 10.1111/joim.13209 [DOI] [PubMed] [Google Scholar]
- 10. Petrocelli M, Cutrupi S, Salzano G, et al. Six‐month smell and taste recovery rates in COVID‐19 patients: a prospective psychophysical study. J Laryngol Otol. 2021;135 5:436–441. 10.1017/S002221512100116X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Riestra‐Ayora J, Yanes‐Diaz J, Esteban‐Sanchez J, et al. Long‐term follow‐up of olfactory and gustatory dysfunction in COVID‐19: 6 months case–control study of health workers. Eur Arch Otorhinolaryngol. 2021; 1–7. 10.1007/s00405-021-06764-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Zou LQ, Hummel T, Otte MS, et al. Association between olfactory function and quality of life in patients with olfactory disorders: a multicenter study over 760 patients. Rhinology. 2021;59:164‐172. [DOI] [PubMed] [Google Scholar]
- 13. Bilinska K, Butowt R. Anosmia in COVID‐19: a bumpy road to establishing a cellular mechanism. ACS Chem Neurosci. 2020;11:2152‐2155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Kirschenbaum D, Imbach LL, Ulrich S, et al. Inflammatory olfactory neuropathy in two patients with COVID‐19. Lancet. 2020;396:166. 10.1016/S0140-6736(20)31525-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Torabi A, Mohammadbagheri E, Akbari Dilmaghani N, et al. Proinflammatory cytokines in the olfactory mucosa result in COVID‐19 induced anosmia. ACS Chem Neurosci. 2020;11:1909‐1913. [DOI] [PubMed] [Google Scholar]
- 16. Vaira LA, Hopkins C, Sandison A, et al. Olfactory epithelium histopathological findings in long‐term COVID‐19 related anosmia. J Laryngol Otol. 2020;134:1123‐1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lechien JR, Radulesco T, Calvo‐Henriquez C, et al. ACE2 and TMPRSS2 expressions in head and neck tissues: a systematic review. Head Neck Pathol. 2021;15:225‐235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Vaira LA, Salzano G, Fois AG, Piombino P, De Riu G. Potential pathogenesis of ageusia and anosmia in COVID‐19 patients. Int Forum Allergy Rhinol. 2020;10:1103‐1104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Abdelalim AA, Mohamady AA, Elsayed RA, Elawady MA, Ghallab AF. Corticosteroid nasal spray for recovery of smell sensation in COVID‐19 patients: a randomized controlled trial. Am J Otolaryngol. 2021;42:102884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Le Bon S‐D, Konopnicki D, Pisarski N, Prunier L, Lechien JR, Horoi M. Efficacy and safety of oral corticosteroids and olfactory training in the management of COVID‐19‐related loss of smell. Eur Arch Otorhinolaryngol. 2021;1–5. 10.1007/s00405-020-06520-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Vaira LA, Hopkins C, Petrocelli M, et al. Efficacy of corticosteroid therapy in the treatment of long‐lasting olfactory disorders in COVID‐19 patients. Rhinology. 2021b;59:21‐25. [DOI] [PubMed] [Google Scholar]
- 22. Saussez S, Vaira LA, Chiesa‐Estomba CM, et al. Short‐term efficacy and safety of oral and nasal corticosteroids in COVID‐19 patients with olfactory dysfunction: a European multicenter study. Pathogens. 2021;10(6):698. 10.3390/pathogens10060698 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Lechien JR, Chiesa‐Estomba CM, Hans S, et al. Validity and reliability of the COVID‐19 symptom index, an instrument evaluating severity of general and otolaryngological symptoms. Acta Otolaryngol. 2021;141(6):615‐620. 10.1080/00016489.2021.1899282 [DOI] [PubMed] [Google Scholar]
- 24. Piccirillo JF, Merritt MG Jr, Richards ML. Psychometric and clinimetric validity of the 20‐Item Sino‐Nasal Outcome Test (SNOT‐20). Otolaryngol Head Neck Surg. 2002;126:41‐47. [DOI] [PubMed] [Google Scholar]
- 25. Oleszkiewicz A, Schriever VA, Croy I, Hähner A, Hummel T. Updated Sniffin’ Sticks normative data based on an extended sample of 9139 subjects. Eur Arch Otorhinolaryngol. 2019;276:719‐728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Tian S, Hu N, Lou J, et al. Characteristics of COVID‐19 infection in Beijing. J Infect. 2020;80:401‐406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Saniasiaya J, Islam MA, Abdullah B. Prevalence of olfactory dysfunction in coronavirus disease 2019 (COVID‐19): a meta‐analysis of 27,492 patients. Laryngoscope. 2021;131:865‐878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Chiesa‐Estomba CM, Lechien JR, Radulesco T, et al. Patterns of smell recovery in 751 patients affected by the COVID‐19 outbreak. Eur J Neurol. 2020;27:2318‐2321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Hopkins C, Surda P, Vaira LA, et al. Six month follow‐up of self‐reported loss of smell during the COVID‐19 pandemic. Rhinology. 2021;59:26‐31. [DOI] [PubMed] [Google Scholar]
- 30. Kandemirli SG, Altundag A, Yildirim D, Tekcan Sanli DE, Saatci O. Olfactory bulb MRI and paranasal sinus CT findings in persistent COVID‐19 anosmia. Acad Radiol. 2021;28:28‐35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Hopkins C, Alanin M, Philpott C, et al. Management of new onset loss of sense of smell during the COVID‐19 pandemic – BRS Consensus Guidelines. Clin Otolaryngol. 2021;46:16‐22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Vaira LA, De Vito A, Deiana G, et al. Correlations between IL‐6 serum level and olfactory dysfunction severity in COVID‐19 patients: a preliminary study. Eur Arch Otorhinolaryngol. 2021;1–6. 10.1007/s00405-021-06868-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Vaira LA, De Vito A, Deiana G, et al. Systemic inflammatory markers and psychophysical olfactory scores in COVID‐19 patients. Is there any correlation? J Laryngol Otol. 2021;1–6. 10.1017/S0022215121001651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Cho RHW, To ZWH, Yeung ZWC, et al. COVID‐19 viral load in the severity and recovery from olfactory and gustatory function. Laryngoscope. 2020;130:2680‐2685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Dias De Melo G, Lazarini F, Levallois S, et al. COVID‐19‐associated olfactory dysfunction reveals SARS‐CoV‐2 neuroinvasion and persistence in the olfactory system. bioRxiv. 2020;1–37. 10.1101/2020-11-18-388819 [DOI] [Google Scholar]
- 36. Zhou Q, Chen V, Shannon CP, et al. Interferon‐α2b treatment for COVID‐19. Front Immunol. 2020;11:615275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Klein SL, Pekosz A, Park HS, et al. Sex, age, and hospitalization drive antibody responses in a COVID‐19 convalescent plasma donor population. J Clin Invest. 2020;130:6141‐6150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Gerhards C, Thiaucourt M, Kittel M, et al. Longitudinal assessment of anti‐SARS‐CoV‐2 antibody dynamics and clinical features following convalescent from COVID‐19 infection. Int J Infect Dis. 2021;107:221–227. 10.1016/j.ijid.2021.04.080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Butler SE, Crowley AR, Natarajan H, et al. Distinct features and functions of systemic and mucosal humoral immunity among SARS‐CoV‐2 convalescent individuals. Front Immunol. 2021;11:618685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Cervia C, Nilsson J, Zurbuchen Y, et al. Systemic and mucosal antibody responses specific to SARS‐CoV‐2 during mild versus severe COVID‐19. J Allergy Clin Immunol. 2021;147:545‐557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Chen K, Magri G, Grasset EK, Cerutti A. Rethinking mucosal antibody responses: IgM, IgG and IgD join IgA. Nat Rev Immunol. 2020;20:427‐441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Nakagawara K, Masaki K, Uwamino Y, et al. Acute onset olfactory/taste disorders are associated with a high viral burden in mild or asymptomatic SARS‐CoV‐2 infections. Int J Infect Dis. 2020;99:19‐22. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Research data are not shared.


