Summary
Oxidative mechanisms are not only involved in chronic degenerative diseases but also in infectious diseases, among which viral respiratory diseases. Antioxidants have the capability to counteract the action of oxidants by scavenging reactive oxygen species (ROS) and by inhibiting oxidant generating enzymes. Overproduction of ROS and deprivation of antioxidant systems play a major role in COVID-19 occurrence, progression, and severity. Interconnected pathways account for the relationships between oxidative damage and inflammation resulting from an interplay between transcription factors having opposite effects. For instance, Nrf2 downregulates inflammation by inhibiting endogenous antioxidant enzymes such as NQO-1 and HO-1. On the other hand, NF-κB upregulates pro-inflammatory cytokines and chemokines, such as IL-1β, IL-6, IL-8, PGE-2, COX-2, TNF-α, MMP-3, and MMP-4. A central protective role against oxidants is played by reduced glutathione (GSH), which is depleted in SARS-CoV-2 infection. N-acetylcysteine (NAC), a precursor of GSH, is of particular interest as an anti-COVID-19 agent. GSH and NAC hamper binding of the S1 subunit of SARS-CoV-2 spike proteins to the angiotensin-converting enzyme 2 (ACE2) receptor. In addition, NAC and its derivatives possess a broad array of antioxidant and antiinflammatory mechanisms that could be exploited for COVID-19 prevention and adjuvant therapy. In particular, as demonstrated in a previous clinical trial evaluating influenza and influenza-like illnesses, the oral administration of NAC may be expected to decrease the risk of developing COVID-19. Furthermore, at the very high doses used worldwide as an antidote against paracetamol intoxication, intravenous NAC is likely to attenuate the pulmonary and systemic symptoms of COVID-19.
Keywords: COVID-19, Oxidative stress, Antioxidants, N-Acetylcysteine, Glutathione
Balance between oxidants and antioxidants in human diseases
Oxidative mechanisms play a key role in the pathogenesis of virtually all human diseases, and consequently antioxidants share a broad range of protective effects. It should be made clear that, like the exposure to oxidants just increases the risk of contracting a given disease, generally in association with other pathogenetic determinants, antioxidants alone are not likely to fully prevent a pathological condition but are just expected to contribute to lower its risk and to attenuate the severity of its consequences. Together with deprivation of antioxidant mechanisms, oxidative stress is also involved in the aging process, especially when causing mutations in the mitochondrial DNA.
Free radicals include reactive oxygen species (ROS), such as the superoxide anion (O2–) and the hydroxyl radical (•OH), an extremely reactive species, as well as ‘non-radical reactive molecules’, such as hydrogen peroxide (H2O2), and reactive nitrogen species (RNS), such as peroxynitrite (ONOO−). These species cause redox-modulated signaling cascades involving the transcription factors AP-1 (activator protein-1), NF-κB (nuclear factor kappa-light-chain-enhancer of activated B cells) and/or Nrf2 (nuclear factor erythroid 2–related factor 2), which can mediate a variety either of physiological functions or alterations of macromolecules leading to pathological conditions [1]. Free radicals may either be introduced into the body from exogenous sources (for instance, smoking one cigarette produces 1016 radicals) [2] or be generated in the body via biochemical reactions, such as the Fenton reaction and the Haber-Weiss reaction. While a controlled production of reactive molecules is essential for normal physiological and cellular functions, their uncontrolled or excessive production can cause ‘oxidative/nitrosative stress’ [1].
Natural or synthetic antioxidants have the capability to counteract the actions of oxidants either by directly scavenging ROS or by inhibiting oxidant generating enzymes, e.g. xanthine oxidase, or by stimulating ROS metabolizing enzymes, such as catalase, superoxide dismutase, or glutathione peroxidase, or by regulating the aforementioned redox-sensitive transcription factors. Therefore, in principle, agents possessing antioxidant properties can prevent the generation of ROS and hamper their deleterious effects. However, an indiscriminate use of antioxidants should be avoided because under certain conditions and at certain doses some of them may become pro-oxidants [1]. A typical example is provided by ascorbic acid that, at high doses and in the presence of transition metal ions, such as iron and copper, can acquire pro-oxidative properties, in spite of the fact that the interaction between ascorbic acid and iron is of nutritional, physiological, and pharmacological interest [3].
Involvement of oxidative stress in respiratory viral diseases and protection by antioxidants
Oxidative stress is strongly involved in the pathogenesis not only of chronic degenerative diseases, such as cancer, atherosclerosis, arterial hypertension and other cardiovascular diseases, neurological disorders, dysmetabolic conditions such as diabetes mellitus, rheumatoid arthritis, etc. [4], but also of infectious diseases. Respiratory viral diseases are often associated with cytokine production, inflammation, and other pathophysiological processes resulting from a redox imbalance, disruption of the thiol redox cycle and other redox circuits [5]. Thus, overproduction of ROS and antioxidant mechanisms deprivation are one of the key events that is linked to viral replication and the subsequent virus-associated disease [6].
Respiratory viruses cause infections of the upper or lower respiratory tract that affect every year millions of people. They include influenza viruses (Orthomyxoviridae family), human respiratory syncytial viruses (HRSV, Pneumoviridae family), human rhinoviruses (HRV, Picornaviridae family), human metapneumoviruses (HMPV) and parainfluenza viruses (both belonging to the Paramyxoviridae family), adenoviruses (Adenoviridae family), and coronaviruses (Coronaviridae family). With the exception of adenoviruses, having a DNA genome, all the other respiratory viruses are RNA viruses. Many lines of evidence suggest that marked signs of increased production of ROS accompany all respiratory viral infections, along with disturbance of antioxidant defences [6]. The sources of ROS in airway epithelial cells infected with viruses are mainly NADPH oxidases, dual oxidase, xanthine oxidase, and Nox2 (NADPH oxidase), which is mainly expressed in macrophages. Alterations of ROS-producing and scavenging pathways that are caused by respiratory viral infections are implicated in inflammation, lung epithelial disruption, tissue damage, and cell death resulting in macrophage activation. In addition, oxidative stress triggers an antiviral immune response, whose excess may lead to a cytokine storm and a severe inflammation [5]. Respiratory viral infections have been associated with inhibition of Nrf2 pathways and/or NF-kB signaling activation, leading to inflammation and oxidative damage [7].
The role of oxidative mechanisms is also supported by experimental and clinical findings that show protective effects of antioxidants such as vitamin C, vitamin E, and N-acetylcysteine (NAC). NAC is the only agent that has been shown to attenuate the risk of respiratory viral diseases in humans. In particular, a double-blind trial involved 262 subjects of both genders who were enrolled in 20 Italian Centres. These subjects were randomized to receive either placebo or NAC tablets (600 mg) twice daily for 6 months. The results demonstrated that administration of NAC during the cold season can attenuate the incidence and severity of influenza and influenza-like illnesses, as shown by the fact that both local and systemic symptoms were sharply and significantly reduced in the NAC group. Moreover, only 25% of A/H1N1 influenza virus-infected subjects receiving NAC developed a symptomatic form versus 79% in the placebo group [8]. In the same study, a time-related shift of anergic condition to normoergic condition was observed in the NAC group thereby showing a beneficial effect of NAC administration on cell-mediated immunity [8].
The protective effects of NAC towards respiratory viral diseases have also been confirmed and explored from a mechanistic point of view in experimental test systems. Influenza A and B viruses and HRSV are responsible for COPD (chronic obstructive pulmonary diseases) by increasing apoptosis and inflammatory events through mechanisms that involve ROS generation and release of mucins from epithelial cells. NAC inhibited the replication of influenza A and B viruses and HRSV and restored the normal functions of alveolar type II A549 cells by modulating MUC5AC overexpression and release and by inhibiting IL-8, IL-6 and TNF- α as well as NF-κB translocation to the nucleus and phosphorylation of MAPK p38 [9]. In addition, NAC inhibited virus replication and expression of pro-inflammatory molecules in the same cells infected with the highly pathogenic H5N1 influenza virus [10]. In vivo, NAC attenuated the pulmonary inflammation and oedema and decreased myeloperoxidase activity, neutrophils, macrophages, TNF-α, IL-6, IL-1β and chemokine ligand-10 in the bronchoalveolar lavage fluid of mice inoculated intranasally with A/swine/HeBei/012/2008/ H9N2 influenza virus [11]. Interestingly, in the perspective of using NAC for COVID-19 treatment in association with antivirals, NAC exerted protective effects towards influenza viruses when administered in association with either ribavirin [12] or oseltamivir [13].
Oxidative mechanisms in COVID-19 pathogenesis
The clinical patterns in most SARS-CoV-2-infected subjects are similar to those of other respiratory diseases. The forms are often paucisymptomatic or even evolve as an asymptomatic infection. However, approximately 15% of COVID-19 patients suffer from impairment of gas exchange and pneumonia, and 5% undergo acute respiratory distress syndrome (ARDS), the leading cause of death in COVID-19 patients, and can experience septic shock and/or multiple organ failure that require hospitalization in intensive care units (ICU) [14]. ARDS involves a systemic inflammatory response that has been attributed to the release of mediators triggering an attack by the immune system [15]. Like other viruses, SARS-CoV-2 stimulates the massive production of proinflammatory cytokines and chemokines, such as TNFα, IL6, and IL8, referred to as “cytokine storm”, which is responsible for lung tissue damage and causes cell death [5]. Although SARS-CoV-2 primarily replicates in the respiratory tract, autopsies demonstrate that this virus can infect cells in multiple organs, including the lungs, pharynx, heart, liver, brain, and kidneys [16]. Cardiovascular alterations include a plethora of disorders, such as stroke, diffuse thrombosis, acute changes in myocardial demand and supply due to tachycardia, hypotension, hypoxemia resulting in type 2 myocardial infarction, acute coronary syndrome due to acute atherothrombosis, microvascular dysfunction due to diffuse microthrombi or vascular injury, stress-related cardiomyopathy (Takotsubo syndrome), direct viral cardiomyocyte toxicity, and myocarditis [17]. Moreover, some COVID-19 patients exhibit widespread neurological manifestations including acute ischemic stroke, intracerebral haemorrhage, cerebral venous sinus thrombosis and anosmia. COVID-19-associated coagulopathy is increasingly recognized as a result of acute infection and is likely caused by the inflammatory cytokine storm [18].
Similarly to influenza virus infection, infection of experimental animals with coronaviruses has provided evidence for the involvement of the oxidative stress machinery, with enhancement of ROS production and weakening of defence mechanisms [19]. Many lines of evidence suggest that overproduction of ROS and a deprivation of antioxidant system play a major role also in the pathogenesis of SARS-CoV and SARS-CoV-2 infections in humans as well as in the progression and severity of the related diseases [4]. Activated neutrophils and mononuclear phagocytic cells are to a large extent responsible for the massive release of ROS into the lung tissue [20]. In addition, the massive TNF-α release during the cytokine storm could exacerbate ROS production via a positive feedback loop by activating NADPH oxidases [21], and TNF-α induced ROS production could contribute to the extension of COVID-19 effects to distant tissues [2].
The importance of oxidative stress in COVID-19 is also reinforced by the role of ROS production in associated co-morbidities. Many studies highlighted the importance of redox-sensitive pathways as novel cell-based targets for therapies aimed at blocking both viral replication and virus-induced inflammation [23]. As discussed in the next session, the prominent role of oxidative mechanisms in the pathogenesis of COVID-19 is corroborated by the perspective of implementing antioxidative strategies in the prevention and therapy of COVID-19 [24].
Some antioxidants proposed for COVID-19 prevention and treatment
Several agents endowed with antioxidant properties have been assayed or proposed with the goals of lowering the risk of being affected by SARS-CoV-2 infection and/or of being used as an adjunct treatment in case of severe COVID-19 forms [25]. Often, these compounds share multiple mechanisms, which may render them capable of exerting broad-spectrum protective effects. On the other hand, antioxidants working with different mechanisms of action may complement each other thereby enhancing their protective properties in a synergistic fashion. Most antioxidant compounds are molecules from natural sources and especially of dietary origin [26], a poor nutrient status being associated with oxidative stress, inflammation, and impairment of the immune system [27, 28]. Therefore, a balanced diet and supplementation with proper nutrients may play a vital role in prevention, treatment, and management of COVID-19 [29].
The scientific literature in this area of research is evolving very rapidly. Some examples of protective antioxidants are shown in Figure 1. Among phytochemicals, polyphenols have been shown both in silico and in vitro to interfere with various stages of coronavirus entry into cells and replication [30] and disclose inhibitory activities towards viral components, which may render them potentially suitable to counteract the SARS-CoV-2 infection [31]. For instance, flavonoids, which are a class of polyphenols including quercetin (Fig. 1), baicalin, luteolin, hesperetin, gallocatechin gallate, epigallocatechin gallate (EGCG,), etc., are able to inhibit key proteins involved in the coronavirus infective cycle, such as PLpro, 3CLpro, and NTPase/helicase [30]. It has also been postulated that quercetin may exert a synergistic antiviral action with vitamin C due to the overlapping antiviral and immunomodulatory properties of these agents and to the capacity of ascorbate to recycle quercetin [32]. In molecular simulations, quercetin has been hypothesized to inhibit the protease 3CLpro, which is an essential player in the coronavirus replication cycle [33]. It is noteworthy, however, that the U.S. Food and Drug Administration has cautioned against advertisement of unauthorized health claims of quercetin.
Fig. 1.
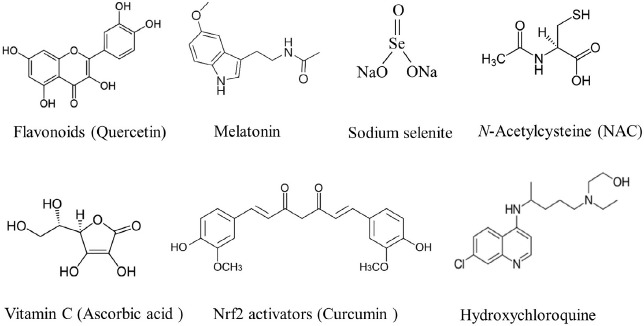
Some agents endowed with antioxidant properties that have been shown or suspected to play a role in COVID-19 prevention and/or adjuvant therapy. The quercetin formula is shown as an example of flavonoids and curcumin is shown as an example of Nrf2 activators.
In theory, vitamin C (ascorbic acid) may represent a suitable tool against SARS-CoV-2 infection because this vitamin is a reducing agent and antioxidant acting by electron transfer reactions and therefore it reacts with ROS such as •OH. However, the appropriateness of its use in COVID-19 is still uncertain [34] also because the use of vitamin C in ARDS or sepsis is still a matter of debate [35]. A multicentre prospective randomised placebo-controlled trial has been recently designed in order to evaluate the effects of high dose intravenous vitamin C in COVID-19 patients hospitalized in ICUs [36].
A promising agent for the treatment of viral diseases, including COVID-19, is melatonin, a potent multifunctional signalling hormone secreted by the pineal gland that acts as an antioxidant with immunomodulatory and anti-inflammatory properties [37]. Melatonin can reduce oxidative stress and efficiently combat the cytokine storm and sepsis. In addition, melatonin is an inhibitor of calmodulin, an essential intracellular component to maintain angiotensin-converting enzyme 2 (ACE-2) on the cell surface [38]. Melatonin has been proposed in subjects with obesity and diabetes, who may undergo severe inflammation and oxidative stress following infection with SARS-CoV-2 infection [39]. It has also hypothesized that children do not suffer from COVID-19 as much as their grandparents because they have lower levels of melatonin, which is lost with age [40]. A clinical trial with an injectable formulation of melatonin for intravenous perfusion in ICU patients suffering from COVID-19 has been designed [41].
Drugs that are being tested for the treatment of COVID-19 may also possess antioxidant properties. For instance, hydroxychloroquine, an old medication for malaria that is also used to treat autoimmune disorders such as rheumatoid arthritis and systemic lupus erythematosus, shares a number of protective effects, also including antioxidant mechanisms [42]. Note however that, on the contrary, the same drug has also been reported to have oxidative properties due to a decrease of GSH levels [43].
A Chinese medicinal formula consisting of 21 herbs (QFPDT) has been recommended in the 6th and 7th versions of Clinical Practice Guideline on COVID-19 in China due to its antioxidant, immunomodulatory and antiviral mechanisms [44].
Nrf2 activators are a broad category of antioxidant agents that could potentially inhibit SARS-CoV-2. In fact, Nrf2 is a transcription factor that regulates the expression of antioxidant cytoprotective enzymes via a promoter sequence known as antioxidant response element (ARE). Regulation of the redox state by Nrf2 results in the modulation of genes involved in immunity and inflammation, also including antiviral mechanisms. Nrf-2 activators include a variety of food-derived compounds that have extensively been investigated for their protective properties, such as curcumin, capsaicin, gingerol, EGCG, genistein, the carotenoid lycopene, resveratrol, caffeic acid phenethyl ester, diallyl sulphide, indole-3-carbinol, and sulphoraphane. Moreover, using primary human pulmonary artery endothelial cells, the synthetic Nrf2 activator PB125® was found to downregulate 36 genes encoding cytokines, such as IL-1-beta, IL-6, TNF-α, the cell adhesion molecules ICAM-1, VCAM-1, and E-selectin, as well as a group of IFN-γ-induced genes, many of which have specifically been identified in the cytokine storm observed in fatal cases of COVID-19 [45].
Among natural Nrf-2 activators, it has been hypothesized that, in the light of its low toxicity and of its antioxidant, anti-inflammatory, and antiviral activity, curcumin may be used as a therapeutic drug for viral pneumonia and ALI (acute lung injury)/ARDS. Curcumin exerts protective effects by regulating the expression of both pro- and anti-inflammatory factors such as IL-6, IL-8, IL-10, and COX-2, by promoting the apoptosis of polymorphonucleate cells, and by scavenging the ROS that exacerbate the inflammatory response [46]. EGCG, the most abundant ingredient in green tea leaves and a well-known antioxidant, has been proposed as a supplementation therapy in COVID-19 patients. Besides some antiviral and anti-sepsis actions, the major EGCG benefits lie in its anti-fibrotic effect and in the ability to downregulate expression and signaling of many inflammatory mediators [47]. Flavonoid supplements, combined with vitamin D3, are expected to activate Nrf2, which may be a potential target to prevent and/or decrease SARS-CoV-2 infection severity, reducing oxidative stress and inflammation, enhancing innate immunity, and downregulating ACE2 receptors [48]. As it will be specifically discussed in more detail below, the thiols GSH and NAC are additional activators of Nrf2 [49].
Selenium (Se) is another important antioxidant whose deficiency is likely to play a role in affecting SARS-CoV-2 virulence and COVID-19 severity. A positive association has been reported between COVID-19 rates and previously measured population Se status in 17 cities across China [50], and Se deficiency has been associated with an increased mortality risk from COVID-19 [51]. Se is a component of glutathione peroxidase 1 (GPx1), a cytosolic selenoenzyme with known antiviral properties (see below), and the interaction between the GPx1 detoxifying system and the main protease (Mpro) of SARS-CoV-2 represents a novel molecular target for COVID-19 [52]. Low Se status is a common finding in conditions considered at risk for severe COVID-19, especially in the elderly, and Se might be beneficial via restoration of the host antioxidant capacity, reduction of apoptosis and endothelial cell damages as well as platelet aggregation [53]. As inferred from an online search for articles published in the period 2010-2020, the direct evidence that the micronutrients zinc, Se, and vitamin D might be involved in the course and outcome of the COVID-19 disease was evaluated to be observational and weak. However, based on experiences from treatments of SARS and other viral infections, it was postulated that nutritive supplements administered at an early stage of the infection would be important in order to enhance the host resistance against RNA viral infections, which might also include severe COVID-19 [54]. Another Se compound of interest is ebselen, an organoselenium compound exhibiting hydroperoxide- and peroxynitrite-reducing activity that behaves as a GPx and peroxiredoxin enzyme mimetic. Ebselen reacts with a multitude of protein thiols, forming a selenosulfide bond, which results in pleiotropic effects of antiviral, antibacterial and anti-inflammatory nature that may potentially be beneficial in COVID-19 [55].
The glutathione (GSH) system
Reduced glutathione (GSH, γ-glutamylcysteinylglycine) provides key protective effects against toxic substances and infectious agents. GSH is available at micromolar concentrations in biological fluids and at millimolar concentrations in cells within the endoplasmic reticulum, mitochondria and nucleus. The thiol redox circuit involves interconversions between GSH and glutathione disulfide (GSSG), which are in an approximately 100:1 ratio inside cells. As shown in Figure 2, GSH is oxidized to GSSG via the enzyme glutathione peroxidase (GPx), whereas GSSG is reduced to GSH via glutathione reductase (GR). The main enzyme involved in GSH synthesis is glutamate cysteine ligase (GCL). Besides being a potent antioxidant, GSH is a nucleophile that can block reactive molecules either per se or via glutathione-S-transferases (GST)-catalyzed conjugation.
Fig. 2.
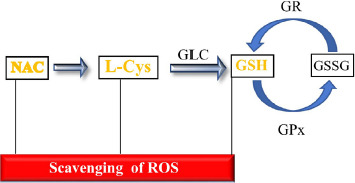
Schematic representation of the glutathione cycle and ability of thiols to scavenge ROS. Abbreviations: GSH, reduced glutathione; GSSG, oxidized glutathione; GR, glutathione reductase; GPx, glutathione peroxidase; GLC, glutamate cysteine ligase; L-Cys, L-Cysteine; NAC, N-acetylcysteine; ROS, reactive oxygen species.
Literature data support the concept that an endogenous deficiency in GSH may underlie the serious manifestations and death due to COVID-19 [56]. A common denominator in all conditions associated with COVID-19 appears to be the impaired redox homeostasis responsible for ROS accumulation. Therefore, GSH levels could be critical in extinguishing the exacerbated inflammation that triggers organ failure in COVID-19 [57]. In addition, GSH plays a central role in the pathophysiology of most human diseases [58], including those that occur as co-morbidities with COVID-19.
It is noteworthy that GR was found to be significantly increased in the blood serum of COVID-19 patients, especially when admitted to ICUs [59]. This alteration reflects an oxidative stress imbalance, being an attempt to replenish the GSH stores that are depleted by the infection. GPx, the other enzyme of the glutathione circuit, belongs to a family of antioxidant selenoenzymes that functionally link selenium and glutathione, both of them showing correlations with clinical outcomes in COVID-19 [60]. Cytosolic GPx1 has been shown to interact with an inactive C145A mutant of Mpro, the main cysteine protease of SARS-CoV-2, but not with catalytically active wild-type Mpro. In addition, Mpro may be targeting not only GPx1 but also several other selenoproteins as well as GCL, the rate-limiting enzyme for glutathione synthesis [60]. Thus, Mpro is a potential drug target, and a screen with over 10,000 compounds identified ebselen as a particularly promising inhibitor of this protease [55].
Liposomal GSH has been proposed as an adjunctive treatment in COVID-19 patients [61]. Moreover, a case report study showed that the repeated use of both oral administration and intravenous injection of GSH was effective in relieving the severe respiratory symptoms of COVID-19, suggesting for the first time the efficacy of this antioxidant therapy for COVID-19 [62].
Inhibition of ACE2 by antioxidant thiols
The S1 subunit of SARS-CoV-2 spike proteins binds to the angiotensin-converting enzyme 2 (ACE2) receptor thereby starting the virus replication cycle in cells. Both the receptor binding domain of the viral spike proteins and ACE2 have several cysteine residues, and the binding affinity is decreased when the disulfide bonds of ACE2 and SARS-CoV-2 spike proteins are reduced to sulfhydryl groups. Therefore, the redox environment of cell surface receptors is regulated by the thiol–disulfide equilibrium in the extracellular region [63]. ACE2, which is expressed in epithelial, endothelial and myocardial cells as well as in T lymphocytes, macrophages, and hepatocytes [64], is a protease that is involved in the renin/angiotensin system together with the angiotensin-converting enzyme (ACE), which has opposite effects. In fact, while ACE causes vasoconstriction, inflammation, apoptosis and oxidative stress due to the production of ROS through the activation of NADPH oxidase and the generation of peroxynitrite anions, ACE2 causes vasodilatation, angiogenesis, antioxidative and antiapoptotic effects [65].
Both animal studies and clinical studies suggest that treatment with the GSH precursor NAC, which is known to attenuate the tolerance to nitrates, modifies the function of the renin/angiotensin system in vivo, an effect that is probably mediated by inhibition of ACE activity [66]. Therefore, by modulating the renin/angiotensin system activity, GSH and its precursor thiols are likely to inhibit entrance of SARS-CoV-2 into cells.
N-Acetyl-L-cysteine (NAC). A promising anti-COVID-19 agent
The thiol NAC easily penetrates cells where it is deacetylated to yield L-cysteine (L-Cys), the only naturally occurring amino acid that carries a thiol-containing side chain (sulfhydryl group). Alternatively, at least in the blood, NAC acts by freeing in the plasma L-Cys that then enters erythrocytes [67]. L-Cys is the rate-limiting substrate for GSH biosynthesis, which is mainly achieved through activation and upregulated production of GCL (Fig. 2) [68]. Therefore, replenishment of depleted GSH stores occurs both by GSH recycling and by de novo synthesis of this tripeptide (Fig. 2).
The physiological recycling of GSH is increased but cannot match its high consumption in COVID-19 lung disease. NAC works both per se in the extracellular environment and as a substrate and precursor of GSH inside cells. Accordingly, all its intracellular effects are mediated by GSH replenishment. It is known since a long time that rescue of GSH through NAC is a treatment strategy for a broad array of different diseases, all of which have in common a pathogenetically relevant loss of GSH [69]. It may be possible to discriminate whether the effects of NAC are either due to NAC itself or to GSH replenishment by comparatively testing its unnatural D-isomer that is not a precursor of L-Cys and GSH [70].
NAC has been in clinical use since the 1960s as a mucolytic agent, usually at the oral dose of 600 mg, due to its ability to break the disulfide bonds of mucus and to depolymerize mucin. Later on, this drug has been proposed or used for the therapy and/or prevention of a variety of diseases involving GSH depletion and redox status imbalances, such as heart diseases, diabetes, AIDS, neurodegenerative diseases, neuropsychiatric disorders, and several other conditions, which generally are treated with 2 daily oral doses of 600 mg [71-73]. Even higher dose regimens (3 daily oral doses of 600 mg) have been used for several years for the treatment of idiopathic pulmonary fibrosis, whose pathogenesis has been ascribed to oxidative agent-mediated alveolar epithelial cell injury, accompanied by an abnormal fibroblast response [74]. Note that COVID-19 pneumonia may present as an acute exacerbation of idiopathic pulmonary fibrosis [75] and that most of the COVID-19 patients undergo postinflammatory pulmonary fibrosis on the follow-up CT scan when discharged [76]. Moreover, high-dose intravenous NAC exerts protective effects in ARDS [77, 78] that, as previously mentioned, is the leading cause of death in COVID-19 patients. ROS play a key role in the pathogenesis of the acute lung injury, and the alveolar epithelial lining fluid (ELF) of patients with ARDS is deficient in GSH [79]. Likewise, GSH is depleted in the ELF of patients with sepsis [79]., which is another condition associated with COVID-19 [80]. In addition, the intravenous administration of NAC at very high doses (150 mg/kg b.w.) is used to treat inflammatory conditions, such as contrast-induced nephropathy [81], and it is in clinical practice as an antidote against paracetamol (acetaminophen) overdosage. For this reason, NAC is quoted in the WHO Model List of Essential Medicines as an antidote in poisonings, being almost 100% effective against paracetamol intoxication when injected within 8 hours after intake of the drug [81]. Furthermore, intravenous NAC may have a role in the management of acute liver failure (ALF) attributable to administration of remdesivir, a direct-acting nucleoside RNA polymerase inhibitor with activity against the novel SARS-CoV-2 virus used in the treatment of COVID-19 pneumonia [82].
Mechanisms of NAC
NAC works via a broad variety of mechanisms, which could be exploited for COVID-19 prevention and treatment. First of all, as previously reported, thiols can hamper penetration of SARS-CoV-2 into cells. Another important property is their ability to block free radicals and reactive molecules responsible either for acute effects or long-term effects. Nucleophilicity of thiols is related to the property of sulfhydryl groups to react with electrophilic metabolites. An example of reactive intermediate is the paracetamol metabolite N-acetyl-p-benzoquinone imine (NAPQI) formed via cytochrome P450 enzymes, which is bound by GSH and excreted in conjugated form. Likewise, NAC and its thiol derivatives have the ability to competitively block electrophilic derivatives of carcinogens that are capable of binding DNA. This may be particularly important in the case of smokers, since there is evidence that smoking is associated with a negative progression and adverse outcomes of COVID-19 [83]. In fact, exposure to cigarette smoke increases ROS levels and causes a depletion of GSH intracellular concentrations [84] by reacting with nonreducible glutathione-aldehyde derivatives [85] thereby accelerating cigarette smoke-induced inflammation and airspace enlargement [86]. NAC has the ability to modulate a large variety of smoking-related end-points, due to many mechanisms and effects demonstrated in experimental test systems. They include NAC nucleophilicity, antioxidant activity, modulation of metabolism, effects in mitochondria, decrease of the biologically effective dose of carcinogens, modulation of DNA repair, inhibition of genotoxicity and cell transformation, modulation of gene expression and signal transduction pathways, regulation of cell survival and apoptosis, antiinflammatory activity, anti-angiogenetic activity, immunological effects, inhibition of progression to malignancy, influence on cell cycle progression, inhibition of preneoplastic and neoplastic lesions, and inhibition of invasion and metastasis [87]. In addition, NAC was shown to modulate several biomarkers in a randomized, double-blind, placebo-controlled, Phase II chemoprevention trial in heavy smokers who received NAC tablets (600 mg) twice daily for 6 months [88].
The key mechanisms shared by NAC, L-Cys and GSH are related to their antioxidant activity, which is mainly due to a potent ability to scavenge ROS (Fig. 2), and especially hypochlorous acid (HOCl) and •OH, and additionally hydrogen peroxide [89]. The SH-groups within the NAC molecule can also scavenge several RNS that play a role in the oxidation of lipids, proteins, and DNA [90].
The antioxidant effects are interconnected with antiinflammatory effects, which is crucial for COVID-19 control [49]. First of all, oxidative stress is linked with inflammation via two parallel biochemical channels that are modulated in opposite direction by NAC (Fig. 3). The first one involves inhibition by NAC of the ROS-mediated activation of NF-κB and consequently the hindrance of biochemical pathways upregulating pro-inflammatory genes [71] involved in COVID-19 pathogenesis, such as interleukins (IL-1β, IL-6, IL-8), prostaglandins (PGE-2), cyclooxygenases (COX-2), tumor necrosis factor (TNF-α), matrix metalloproteinases (MMP-3, MMP-4), and intercellular adhesion molecule (ICAM-1). NAC also inhibits NF-κB translocation to the cellular nucleus and phosphorylation of MAPK p38 (p38 mitogen-activated protein kinase) by reducing the intracellular hydrogen peroxide concentration and by restoring the intracellular total thiol contents [91]. On the other hand, NAC further enhances the stimulation of Nrf2 by oxidative stress [92]. Nrf2 downregulates inflammation by favouring the ARE-mediated transcription of phase II enzyme genes. These include endogenous antioxidant enzymes such as heme oxygenase 1 (HO-1), NAD(P)H dehydrogenase [quinone] 1 (NQO-1) and additionally GCL [93]. It is noteworthy that the heme-HO-1 system has been proposed as a target to prevent severe complications following SARS-CoV-2 infection [94].
Fig. 3.
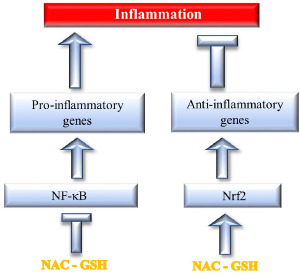
NAC and GSH trigger two parallel channels that attenuate inflammation either by inhibiting NFκB-dependent pro-inflammatory genes and/or by stimulating Nrf2-dependent anti-inflammatory genes (see Text for details).
Besides acting as antioxidants by scavenging ROS, NAC and GSH elicit antioxidant and antiinflammatory effects via other mechanisms that may bear relevance for the control of COVID-19 and associated co-morbidities. For instance, NAC can exert antioxidant activity via p53 mediated apoptosis [95]. In the cardiovascular domain, NAC is well known to interact with nitric oxide by potentiating its vasodilator and antiaggregatory effects [96]. Moreover, NAC inhibits the ROS-producing vascular NAD(P)H oxidases, which bears relevance in the prevention of hypertension and atherosclerosis [97]. NAC has also been proposed with the goal of preserving endothelial function and limiting microthrombosis in severe forms of COVID-19 [98]. Of particular interest in the framework of the COVID-19 clinical picture is inhibition of epidermal growth factor receptor (EGFR), a tyrosine kinase involved in inflammation, which also results in a decreased inactivation of α1-antitrypsin that, together with oxidative stress, plays an important role in the pathogenesis of COPD and its exacerbations [99, 100]. In addition, NAC is a hydrogen sulfide donor, because L-Cys, derived by NAC catabolism, is the substrate for this vasodilator, anti-inflammatory and readily diffusible compound [101], which is a pleiotropic mediator having effects on many elements in the inflammatory cascade and promoting the resolution of inflammation and injury [102].
Conclusions
Oxidative stress represents a major mechanism in the pathogenesis not only of chronic degenerative diseases but also of infectious diseases, also including viral respiratory diseases. Oxidation, inflammation and immune response impairment are strictly interconnected and are key determinants in COVID-19. Various antioxidants have been proposed as anti-SARS-CoV-2 agents. One of the most promising drugs is NAC, a precursor of GSH, both because the redox environment of the ACE2 receptor of SARS-CoV-2 spikes is regulated by the thiol-disulfide balance in the extracellular region and because replenishment of depleted GSH stores by NAC exerts formidable antioxidant and antiinflammatory effects. These effects are potentially suitable to control the cytokine storm that is characteristic of COVID-19. Due to their pleiotropic mechanisms, both NAC and GSH have been evaluated or are under scrutiny for a variety of end-points in a large number of clinical trials (162 studies for GSH and 714 studies for NAC) [57]. We [49] and several other authors [43, 103-115] have proposed the use of NAC in the prevention and/or the treatment of COVID-19. Due to its low toxicity profiles, the 60 year-experience of clinical use and the fact that NAC is approved by FDA under various formulations and is popular as a health supplement, this drug may be repurposed as an anti-COVID-19 agent.
In particular, two strategies can be envisaged. The first one is the oral administration of NAC, at the dose of 600 mg twice per day, in order to decrease the risk of developing COVID-19 and to attenuate its severity, especially during epidemic periods and in high risk individuals because of age and/or concomitant pathological conditions or because they have been in contact with infected SARS-CoV-2 carriers. Interestingly, having previously demonstrated that this protocol is effective in lowering the incidence and severity of influenza and influenza-like illnesses [8], the hypothesis that NAC administration may confer a broad spectrum protection against different respiratory viral diseases is mechanistically sound. It is also noteworthy that oral NAC (600 mg/twice daily) was safe and effective to prevent and delay ventilator-associated pneumonia, and improved its complete recovery rate in a selected, high-risk ICU population [116]. At the same NAC dose, a cross sectional study evaluating 164 COVID-19 patients in Kolkata (India) found that moderate-severe patients who received NAC along with standard therapy had a variety of benefits from the clinical standpoint [117].
The second perspective, in case of manifest COVID-19 forms, is to use NAC as an adjuvant therapy, possibly in association with other drugs, at the high intravenous doses that are commonly used as an antidote against paracetamol intoxication. It is noteworthy that paracetamol, which is the preferred drug for the symptomatic and domiciliary management of the early stages of COVID-19, may cause GSH depletion, especially in people at higher COVID-19 risk, thereby increasing further the risk of developing severe COVID-19 forms [118]. Accordingly, the preferential use of paracetamol in COVID-19 as a safer alternative to nonsteroidal antiinflammatory drugs (NSAIDs) should be carefully reconsidered [119] and it would be important to further investigate whether NAC supplementation should be adopted, irrespective of COVID-19, in case of prolonged administration of high doses of this antipyretic and analgesic compound [118]. A case of severe COVID-19 infection treated with hydroxychloroquine in a patient deficient in glucose 6-phosphate dehydrogenase (G6PD), which facilitates human coronavirus infection due to GSH depletion, had benefit from the intravenous administration of NAC [43]. On the other hand, a double-blind, randomized, placebo-controlled trial enrolling 135 patients with severe COVID-19 (confirmed or suspected), conducted in São Paulo, Brazil, did not show any benefit from the intravenous injection of 21 g NAC (approximately 300 mg/kg) for 20 hours, at least in terms of need for endotracheal intubation and mechanical ventilation [120].
Several clinical trials evaluating the efficacy and safety of NAC treatment in COVID-19 are now in progress in various countries, including Brazil, China, Iran, Nigeria, Saudi Arabia and USA (http://www.ensaiosclinicos.gov.br/rg/RBR-8969zg/; https://clinicaltrials.gov/; https://doi.org/10.1186/ISRCTN60069084; https://clinicaltrials.gov/show/NCT04279197; http://ethics.research.ac.ir/PortalProposalListEn.php?code=&title=acetylcysteine&name).
Figures and tables
Acknowledgements
Writing of this article did not receive any economical support.
Footnotes
Conflict of interest statement
The authors declare no conflict of interest.
Authors’ contributions
SDF wrote the manuscript. RB and SLM made literature search and revised the manuscript.
References
- [1].Oter S, Jin S, Cucullo L, Dorman HJ. Oxidants and antioxidants: friends or foes? Oxid Antioxid Med Sci 2012;1:1-4. https://doi.org/10.5455/oams.080612.ed.001 10.5455/oams.080612.ed.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Pryor WA, Prier DG, Church DF. Electron-spin resonance study of mainstream and sidestream cigarette smoke: nature of the free radicals in gas-phase smoke and in cigarette tar. Environ Health Perspect 1983;47:345-55. https://doi.org/10.1289/ehp.8347345 10.1289/ehp.8347345 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Kontoghiorghes GJ, Kolnagou A, Kontoghiorghe CN, Mourouzidis L, Timoshnikov VA, Polyakov NE. Trying to solve the puzzle of the interaction of ascorbic acid and iron: redox, chelation and therapeutic implications. Medicines (Basel) 2020;7:E45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].García-Sánchez A, Miranda-Díaz AG, Cardona-Muñoz EG. The role of oxidative stress in physiopathology and pharmacological treatment with pro- and antioxidant properties in chronic diseases. Oxid Med Cell Longev 2020;2020:2082145. https://doi.org/10.1155/2020/2082145 10.1155/2020/2082145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Delgado-Roche L, Mesta F. Oxidative stress as key player in severe acute respiratory syndrome coronavirus (SARS-CoV) infection. Arch Med Res 2020;51:384-7. https://doi.org/10.1016/j.arcmed.2020.04.019 10.1016/j.arcmed.2020.04.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Khomich OA, Kochetkov SN, Bartosch B, Ivanov AV. Redox biology of respiratory viral infections. Viruses 2018;10:392. https://doi.org/10.3390/v10080392 10.3390/v10080392 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Komaravelli N, Casola A. Respiratory viral infections and subversion of cellular antioxidant defenses. J Pharmacogenomics Pharmacoproteomics 2014;5:1000141. https://doi.org/10.4172/2153-0645.1000141 10.4172/2153-0645.1000141 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].De Flora S, Grassi C, Carati L. Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment. Eur Respir J 1997;10:1535-41. [DOI] [PubMed] [Google Scholar]
- [9].Mata M, Morcillo E, Gimeno C, Cortijo J. N-acetyl-L-cysteine (NAC) inhibit mucin synthesis and pro-inflammatory mediators in alveolar type II epithelial cells infected with influenza virus A and B and with respiratory syncytial virus (RSV). Biochem Pharmacol 2011;82:548-55. https://doi.org/10.1016/j.bcp.2011.05.014 10.1016/j.bcp.2011.05.014 [DOI] [PubMed] [Google Scholar]
- [10].Geiler J, Michaelis M, Naczk P, Leutz A, Langer K, Doerr HW, Cinatl J, Jr. N-acetyl-L-cysteine (NAC) inhibits virus replication and expression of pro-inflammatory molecules in A549 cells infected with highly pathogenic H5N1 influenza A virus. Biochem Pharmacol 2010;1:79:413-20. https://doi.org/10.1016/j.bcp.2009.08.025 10.1016/j.bcp.2009.08.025 [DOI] [PubMed] [Google Scholar]
- [11].Zhang RH, Li CH, Wang CL, Xu M-J, Xu T, Wei D, Liu B-J, Wang G-H, Tian S-F. N-acetyl-l-cystine (NAC) protects against H9N2 swine influenza virus-induced acute lung injury. Int Immunopharmacol 2014;22:1-8. https://doi.org/10.1016/j.intimp.2014.06.013 10.1016/j.intimp.2014.06.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Ghezzi P, Ungheri D. Synergistic combination of N-acetylcysteine and ribavirin to protect from lethal influenza viral infection in a mouse model. Int J Immunopathol Pharmacol 2004;17:99-102. https://doi.org/10.1177/039463200401700114 10.1177/039463200401700114 [DOI] [PubMed] [Google Scholar]
- [13].Garozzo A, Tempera G, Ungheri D, Timpanaro R, Castro A. N-acetylcysteine synergizes with oseltamivir in protecting mice from lethal influenza infection. Int J Immunopathol Pharmacol 2007;20:349-54. [DOI] [PubMed] [Google Scholar]
- [14].Cao X. COVID-19: immunopathology and its implications for therapy. Nat Rev Immunol 2020;20:269-70. https://doi.org/10.1038/s41577-020-0308-3 10.1038/s41577-020-0308-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Li X, Geng M, Peng Y, Meng L, Lu S. Molecular immune pathogenesis and diagnosis of COVID-19. J Pharm Anal 2020;10:102-8. https://doi.org/10.1016/j.jpha.2020.03.001. 10.1016/j.jpha.2020.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Puelles VG, Lütgehetmann M, Lindenmeyer MT, Sperhake JP, Wong MN, Allweiss L, Chilla S, Heinemann A, Wanner N, Liu S, Braun F, Lu S, Pfefferle S, Schröder AS, Edler C, Gross O, Glatzel M, Wichmann D, Wiech T, Kluge S, Pueschel K, Aepfelbacher M, Huber TB. Multiorgan and Renal Tropism of SARS-CoV-2. N Engl J Med 2020;383:590-592. https://doi.org/10.1056/NEJMc2011400. 10.1056/NEJMc2011400 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Lang JP, Wang X, Moura FA, Siddiqi HK, Morrow DA, Bohula EA. A current review of COVID-19 for the cardiovascular specialist. Am Heart J 2020;226:29-44. https://doi.org/10.1016/j.ahj.2020.04.025. 10.1016/j.ahj.2020.04.025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [18].Divani AA, Andalib S, Di Napoli M, Lattanzi S, Hussain MS, Biller J, McCullough LD, Azarpazhooh MR, Seletska A, Mayer SA, Torbey M. Coronavirus Disease 2019 and stroke: clinical manifestations and pathophysiological insights. J Stroke Cerebrovasc Dis 2020;29:104941. https://doi.org/10.1016/j.jstrokecerebrovasdis.2020.104941 10.1016/j.jstrokecerebrovasdis.2020.104941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].van den Brand JM, Haagmans BL, van Riel D, Osterhaus AD, Kuiken T. The pathology and pathogenesis of experimental severe acute respiratory syndrome and influenza in animal models. J Comp Pathol 2014;151:83-112. https://doi.org/10.1016/j.jcpa.2014.01.004 10.1016/j.jcpa.2014.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Schönrich G, Raftery MJ, Samstag Y. Devilishly radical NETwork in COVID-19: Oxidative stress, neutrophil extracellular traps (NETs), and T cell suppression. Adv Biol Regul 2020;77:100741. https://doi.org/10.1016/j.jbior.2020.100741 10.1016/j.jbior.2020.100741 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [21].Blaser H, Dostert C, Mak TW, Brenner D. TNF and ROS Crosstalk in Inflammation. Trends Cell Biol 2016;26:249-61. https://doi.org/10.1016/j.tcb.2015.12.002 10.1016/j.tcb.2015.12.002 [DOI] [PubMed] [Google Scholar]
- [22].Sandoval R, Lazcano P, Ferrari F, Pinto-Pardo N, González-Billault C, Utreras E. TNF-α increases production of reactive oxygen species through Cdk5 activation in nociceptive neurons. Front Physiol 2018;9:65. https://doi.org/10.3389/fphys.2018.00065 10.3389/fphys.2018.00065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [23].Checconi P, De Angelis M, Marcocci ME, Fraternale A, Magnani Palamara AT, Nencioni L. Redox-modulating agents in the treatment of viral infections. Int J Mol Sci 2020;21:4084. https://doi.org/10.3390/ijms21114084 10.3390/ijms21114084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Wu J. Tackle the free radicals damage in COVID-19. Nitric Oxide 2020;102:39-41. https://doi.org/10.1016/j.niox.2020.06.002 10.1016/j.niox.2020.06.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [25].Soto ME, Guarner-Lans V, Soria-Castro E, Manzano Pech L, Pérez-Torres I. Is Antioxidant therapy a useful complementary measure for Covid-19 treatment? An algorithm for its application. Medicina (Kaunas) 2020;56:386. https://doi.org/10.3390/medicina56080386 10.3390/medicina56080386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Diniz LRL, Bezerra Filho CDSM, Fielding BC, de Sousa DP. Natural antioxidants: a review of studies on human and animal coronavirus. Oxid Med Cell Longev 2020;2020:3173281. https://doi.org/10.1155/2020/3173281 10.1155/2020/3173281 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [27].Iddir M, Brito A, Dingeo G, Fernandez Del Campo S, Samouda H, La Frano MR, Bohn T. Strengthening the immune system and reducing inflammation and oxidative stress through diet and nutrition: considerations during the COVID-19 crisis. Nutrients 2020;12:1562. https://doi.org/10.3390/nu12061562 10.3390/nu12061562 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [28].Emerald M. Nutrients in prevention and maintenance of COVID-19 and other viral diseases. In: Functional Foods and Viral Diseases, First Edition, Chapter 8. Dallas, TX, USA: Food Science Publisher; 2020, pp. 175-202. [Google Scholar]
- [29].BourBour F, Mirzaei Dahka S, Gholamalizadeh M, Akbari ME, Shadnoush M, Haghighi M, Taghvaye-Masoumi H, Ashoori N, Doaei S. Nutrients in prevention, treatment, and management of viral infections; special focus on Coronavirus. Arch Physiol Biochem 2020:1-10. https://doi.org/10.1080/13813455.2020.1791188 10.1080/13813455.2020.1791188 [DOI] [PubMed] [Google Scholar]
- [30].Russo M, Moccia S, Spagnuolo C, Tedesco I, Russo GL. Roles of flavonoids against coronavirus infection. Chem Biol Interact. 2020;328:109211. https://doi.org/10.1016/j.cbi.2020.109211 10.1016/j.cbi.2020.109211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Levy E, Delvin E, Marcil V, Spahis S. Can phytotherapy with polyphenols serve as a powerful approach for the prevention and therapy tool of novel coronavirus disease 2019 (COVID-19)? Am J Physiol Endocrinol Metab 2020;319:E689-E708. https://doi.org/10.1152/ajpendo.00298.2020 10.1152/ajpendo.00298.2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Colunga Biancatelli RML, Berrill M, Catravas JD, Marik PE. Quercetin and vitamin C: an experimental, synergistic therapy for the prevention and treatment of SARS-CoV-2 related disease (COVID-19). Front Immunol 2020;11:1451. https://doi.org/10.3389/fimmu.2020.01451 10.3389/fimmu.2020.01451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Abian O, Ortega-Alarcon D, Jimenez-Alesanco A, Ceballos Laita L, Vega S, Reyburn HT, Rizzuti B, Velazquez-Campoy A. Structural stability of SARS-CoV-2 3CLpro and identification of quercetin as an inhibitor by experimental screening. Int J Biol Macromol 2020;164:1693-703. https://doi.org/10.1016/j.ijbiomac.2020.07.235 10.1016/j.ijbiomac.2020.07.235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Loffredo L, Violi F. COVID-19 and cardiovascular injury: a role for oxidative stress and antioxidant treatment? Int J Cardiol 2020;312:136. https://doi.org/10.1016/j.ijcard.2020.04.066 10.1016/j.ijcard.2020.04.066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Obi J, Pastores SM, Ramanathan LV, Yang J, Halpern NA. Treating sepsis with vitamin C, thiamine, and hydrocortisone: exploring the quest for the magic elixir. J Crit Care 2020;57:231-9. https://doi.org/10.1016/j.jcrc.2019.12.011 10.1016/j.jcrc.2019.12.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Liu F, Zhu Y, Zhang J, Li Y, Peng Z. Intravenous high-dose vitamin C for the treatment of severe COVID-19: study protocol for a multicentre randomised controlled trial. BMJ Open 2020;10:e039519. https://doi.org/10.1136/bmjopen-2020-039519 10.1136/bmjopen-2020-039519 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Bahrampour Juybari K, Pourhanifeh MH, Hosseinzadeh A, Hemati K, Mehrzadi S. Melatonin potentials against viral infections including COVID-19: current evidence and new findings. Virus Res 2020;287:198108. https://doi.org/10.1016/j.virusres.2020.198108 10.1016/j.virusres.2020.198108 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Feitosa EL, Júnior FTDSS, Nery Neto JAO, Matos LFL, Moura MHS, Rosales TO, De Freitas GBL. COVID-19: rational discovery of the therapeutic potential of melatonin as a SARS-CoV-2 main protease inhibitor. Int J Med Sci 2020;17:2133-46. https://doi.org/10.7150/ijms.48053 10.7150/ijms.48053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [39].El-Missiry MA, El-Missiry ZMA, Othman AI. Melatonin is a potential adjuvant to improve clinical outcomes in individuals with obesity and diabetes with coexistence of Covid-19. Eur J Pharmacol 2020;882:173329. https://doi.org/10.1016/j.ejphar.2020.173329 10.1016/j.ejphar.2020.173329 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [40].Shneider A, Kudriavtsev A, Vakhrusheva A. Can melatonin reduce the severity of COVID-19 pandemic? Int Rev Immunol 2020;39:153-62. https://doi.org/10.1080/08830185.2020.1756284 10.1080/08830185.2020.1756284 [DOI] [PubMed] [Google Scholar]
- [41].Acuña-Castroviejo D, Escames G, Figueira JC, de la Oliva P, Borobia AM, Acuña-Fernández C. Clinical trial to test the efficacy of melatonin in COVID-19. J Pineal Res 2020;69:e12683. https://doi.org/10.1111/jpi.12683 10.1111/jpi.12683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [42].Pahan P, Pahan K. Smooth or risky revisit of an old malaria drug for COVID-19? J Neuroimmune Pharmacol 2020;15:174-80. https://doi.org/10.1007/s11481-020-09923-w 10.1007/s11481-020-09923-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- [43].Ibrahim H, Perl A, Smith D, Lewis T, Kon Z, Goldenberg R, Yarta K, Staniloae C, Williams M. Therapeutic blockade of inflammation in severe COVID-19 infection with intravenous N-acetylcysteine. Clin Immunol 2020;219:108544. https://doi.org/10.1016/j.clim.2020.108544 10.1016/j.clim.2020.108544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [44].Zhong LLD, Lam WC, Yang W, Chan KW, Sze SCW, Miao J, Yung KKL, Bian Z, Wong VT. Potential targets for treatment of Coronavirus Disease 2019 (COVID-19): a review of Qing-Fei-Pai-Du-Tang and its major herbs. Am J Chin Med 2020;48:1051-71. https://doi.org/10.1142/S0192415X20500512 10.1142/S0192415X20500512 [DOI] [PubMed] [Google Scholar]
- [45].McCord JM, Hybertson BM, Cota-Gomez A, Geraci KP, Gao B. Nrf2 Activator PB125® as a potential therapeutic agent against COVID-19. Antioxidants (Basel) 2020;9:518. https://doi.org/10.3390/antiox9060518 10.3390/antiox9060518 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [46].Liu Z, Ying Y. The inhibitory effect of curcumin on virus-induced cytokine storm and its potential use in the associated severe pneumonia. Front Cell Dev Biol 2020;8:479. https://doi.org/10.3389/fcell.2020.00479 10.3389/fcell.2020.00479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Menegazzi M, Campagnari R, Bertoldi M, Crupi R, Di Paola R, Cuzzocrea S. Protective effect of epigallocatechin-3-gallate (EGCG) in diseases with uncontrolled immune activation: could such a scenario be helpful to counteract COVID-19? Int J Mol Sci 2020;21:5171. https://doi.org/10.3390/ijms21145171 10.3390/ijms21145171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Mendonca P, Soliman KFA. Flavonoids activation of the transcription factor Nrf2 as a hypothesis approach for the prevention and modulation of SARS-CoV-2 infection severity. Antioxidants (Basel) 2020;9:659. https://doi.org/10.3390/antiox9080659 10.3390/antiox9080659 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].De Flora S, Balansky R, La Maestra S. Rationale for the use of N-acetylcysteine in both prevention and adjuvant therapy of COVID-19. FASEB J 2020;34:13185-93. https://doi.org/10.1096/fj.202001807 10.1096/fj.202001807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [50].Zhang J, Taylor EW, Bennett K, Saad R, Rayman MP. Association between regional selenium status and reported outcome of COVID-19 cases in China. Am J Clin Nutr 2020;111:1297-9. https://doi.org/10.1093/ajcn/nqaa095 10.1093/ajcn/nqaa095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [51].Moghaddam A, Heller RA, Sun Q, Seelig J, Cherkezov A, Seibert L, Hackler J, Seemann P, Diegmann J, Pilz M, Bachmann M, Minich WB, Schomburg L. Selenium deficiency Is associated with mortality risk from COVID-19. Nutrients 2020;12:2098. https://doi.org/10.3390/nu12072098 10.3390/nu12072098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [52].Seale LA, Torres DJ, Berry MJ, Pitts MW. A role for selenium-dependent GPX1 in SARS-CoV-2 virulence. Am J Clin Nutr 2020;112:447-8. https://doi.org/10.1093/ajcn/nqaa177 10.1093/ajcn/nqaa177 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [53].Hiffler L, Rakotoambinina B. Selenium and RNA virus interactions: potential implications for SARS-CoV-2 infection (COVID-19). Front Nutr 2020;7:164. https://doi.org/10.3389/fnut.2020.00164 10.3389/fnut.2020.00164 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [54].Alexander J, Tinkov A, Strand TA, Alehagen U, Skalny A, Aaseth J. Early nutritional interventions with zinc, selenium and vitamin D for raising anti-viral resistance against progressive COVID-19. Nutrients 2020;12:2358. https://doi.org/10.3390/nu12082358 10.3390/nu12082358 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [55].Sies H, Parnham MJ. Potential therapeutic use of ebselen for COVID-19 and other respiratory viral infections. Free Radic Biol Med. 2020;156:107-12. https://doi.org/10.1016/j.freeradbiomed.2020.06.032 10.1016/j.freeradbiomed.2020.06.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [56].Polonikov A. Endogenous Deficiency of Glutathione as the Most Likely Cause of serious manifestations and death in COVID-19 patients. ACS Infect Dis 2020;6:1558-62. https://doi.org/10.1021/acsinfecdis.0c00288 10.1021/acsinfecdis.0c00288 [DOI] [PubMed] [Google Scholar]
- [57].Silvagno F, Vernone A, Pescarmona GP. The role of glutathione in protecting against the severe inflammatory response triggered by COVID-19. Antioxidants (Basel) 2020;9:624. https://doi.org/10.3390/antiox9070624 10.3390/antiox9070624 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [58].Franco R, Schoneveld OJ, Pappa A, Panayiotidis MI. The central role of glutathione in the pathophysiology of human diseases. Arch Physiol Biochem 2007;113:234-58. https://doi.org/10.1080/13813450701661198 10.1080/13813450701661198 [DOI] [PubMed] [Google Scholar]
- [59].Cao M, Zhang D, Wang Y, Lu Y., Zhu X, Ying Li, Xue H, Lin Y, Zhang M, Sun Y, Yang Z, Shi J, Wang Y, Zhou C, Dong Y, Liu P, Dudek SM, Xiao Z, Lu H, Peng L. Clinical features of patients infected with the 2019 novel coronavirus (COVID-19) in Shanghai, China. medRxiv 2020. https://doi.org/10.1101/2020.03.04.20030395 10.1101/2020.03.04.20030395 [DOI] [Google Scholar]
- [60].Taylor EW, Radding W. Understanding selenium and glutathione as antiviral factors in COVID-19: does the viral Mpro protease target host selenoproteins and glutathione synthesis? Front Nutr 2020;7:143. https://doi.org/10.3389/fnut.2020.00143 10.3389/fnut.2020.00143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [61].Guloyan V, Oganesian B, Baghdasaryan N, Yeh C, Singh M, Guilford F, Ting YS, Venketaraman V. Glutathione supplementation as an adjunctive therapy in COVID-19. Antioxidants (Basel) 2020;9:E914. https://doi.org/10.3390/antiox9100914 10.3390/antiox9100914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [62].Horowitz RI, Freeman PR, Bruzzese J. Efficacy of glutathione therapy in relieving dyspnea associated with COVID-19 pneumonia: a report of 2 cases. Respir Med Case Rep 2020;30:101063. https://doi.org/10.1016/j.rmcr.2020.101063 10.1016/j.rmcr.2020.101063 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [63].Hati S, Bhattacharyya S. Impact of thiol-disulfide balance on the binding of Covid-19 spike protein with Angiotensin Converting Enzyme 2 receptor. bioRxiv 2020. https://doi.org/10.1101/2020.05.07.083147 10.1101/2020.05.07.083147 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [64].Capettini LS, Montecucco F, Mach F, Stergiopulos N, Santos RA, da Silva RF. Role of renin-angiotensin system in inflammation, immunity and aging. Curr Pharm Des 2012;18:963-70. https://doi.org/10.2174/138161212799436593 10.2174/138161212799436593 [DOI] [PubMed] [Google Scholar]
- [65].Basi Z, Turkoglu V. In vitro effect of oxidized and reduced glutathione peptides on angiotensin converting enzyme purified from human plasma. J Chromatogr B Analyt Technol Biomed Life Sci 2019;1104:190-5. https://doi.org/10.1016/j.jchromb.2018.11.023 10.1016/j.jchromb.2018.11.023 [DOI] [PubMed] [Google Scholar]
- [66].Boesgaard S, Aldershvile J, Poulsen HE, Christensen S, Dige-Petersen H, Giese J. N-acetylcysteine inhibits angiotensin converting enzyme in vivo. J Pharmacol Exp Ther 1993;265:1239-44. [PubMed] [Google Scholar]
- [67].Raftos JE, Whillier S, Chapman BE, Kuchel PW. Kinetics of uptake and deacetylation of N-acetylcysteine by human erythrocytes. Int J Biochem Cell Biol 2007;39:1698-706. https://doi.org/10.1016/j.biocel.2007.04.014 10.1016/j.biocel.2007.04.014 [DOI] [PubMed] [Google Scholar]
- [68].Lu SC. Regulation of glutathione synthesis. Mol Aspects Med 2009;30:42-59. https://doi.org/10.1016/j.mam.2008.05.005 10.1016/j.mam.2008.05.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [69].Ruffmann R, Wendel A. GSH rescue by N-acetylcysteine. Klin Wochenschr 1991;69:857-62. https://doi.org/10.1007/BF01649460 10.1007/BF01649460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [70].Wong BK, Chan HC, Corcoran GB. Selective effects of N-acetylcysteine stereoisomers on hepatic glutathione and plasma sulfate in mice. Toxicol Appl Pharmacol 1986;86:421-9. https://doi.org/10.1016/0041-008x(86)90369-8 10.1016/0041-008x(86)90369-8 [DOI] [PubMed] [Google Scholar]
- [71].Sadowska AM. N-Acetylcysteine mucolysis in the management of chronic obstructive pulmonary disease. Ther Adv Respir Dis 2012;6:127-35. https://doi.org/10.1177/1753465812437563 10.1177/1753465812437563 [DOI] [PubMed] [Google Scholar]
- [72].Šalamon Š, Kramar B, Marolt TP, Poljšak B, Milisav I. Medical and dietary uses of N-acetylcysteine. Antioxidants (Basel) 2019;8:111. https://doi.org/10.3390/antiox8050111 10.3390/antiox8050111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [73].Haddad JJ. Glutathione depletion is associated with augmenting a proinflammatory signal: evidence for an antioxidant/pro-oxidant mechanism regulating cytokines in the alveolar epithelium. Cytokines Cell Mol Ther 2000;6:177-87. https://doi.org/10.1080/mccm.6.4.177.187 10.1080/mccm.6.4.177.187 [DOI] [PubMed] [Google Scholar]
- [74].Feng F, Zhang J, Wang Z, Wu Q, Zhou X. Efficacy and safety of N-acetylcysteine therapy for idiopathic pulmonary fibrosis: an updated systematic review and meta-analysis. Exp Ther Med 2019;18:802-16. https://doi.org/10.3892/etm.2019.7579 10.3892/etm.2019.7579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [75].Lee YH, Kim CH, Lee J. Coronavirus disease 2019 pneumonia may present as an acute exacerbation of idiopathic pulmonary fibrosis. J Thorac Dis 2020;12:3902-4. https://doi.org/10.21037/jtd-20-1658 10.21037/jtd-20-1658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [76].Zhan X, Liu B, Tong ZH. Postinflammatroy pulmonary fibrosis of COVID-19: the current status and perspective. Zhonghua Jie He He Hu Xi Za Zhi 2020;43:728-32. https://doi.org/10.3760/cma.j.cn112147-20200317-00359 10.3760/cma.j.cn112147-20200317-00359 [DOI] [PubMed] [Google Scholar]
- [77].Bernard GR, Wheeler AP, Arons MM, Morris PE, Paz HL, Russell JA, Wright PE. A trial of antioxidants N-acetylcysteine and procysteine in ARDS. The Antioxidant in ARDS Study Group. Chest 1997;112:164-72. https://doi.org/10.1378/chest.112.1.164 10.1378/chest.112.1.164 [DOI] [PubMed] [Google Scholar]
- [78].Soltan-Sharifi MS, Mojtahedzadeh M, Najafi A, Reza Khajavi M, Reza Rouini M, Moradi M, Mohammadirad A, Abdollahi M. Improvement by N-acetylcysteine of acute respiratory distress syndrome through increasing intracellular glutathione, and extracellular thiol molecules and anti-oxidant power: evidence for underlying toxicological mechanisms. Hum Exp Toxicol 2007;26:697-703. https://doi.org/10.1177/0960327107083452 10.1177/0960327107083452 [DOI] [PubMed] [Google Scholar]
- [79].Pacht ER, Timerman AP, Lykens MG, Merola AJ. Deficiency of alveolar fluid glutathione in patients with sepsis and the adult respiratory distress syndrome. Chest 1991;100:1397-403. [DOI] [PubMed] [Google Scholar]
- [80].Dharsandiya M, Shah K, Patel K, Patel T, Patel A, Patel A. SARS-CoV-2 viral sepsis with meningoencephalitis. Indian J Med Microbiol 2020;38:219-21. https://doi.org/10.4103/ijmm.IJMM_20_291 10.4103/ijmm.IJMM_20_291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [81].Ershad M, Wermuth HR, Vearrier D. N Acetylcysteine. In: StatPearls. Treasure Island (FL): StatPearls Publishing; 2020. [Google Scholar]
- [82].Carothers C, Birrer K, Vo M. Acetylcysteine for the treatment of suspected remdesivir-associated acute liver failure in COVID-19: a case series. Pharmacotherapy 2020:10.1002/phar.2464. https://doi.org/10.1002/phar.2464 10.1002/phar.2464 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [83].Vardavas CI, Nikitara K. COVID-19 and smoking: A systematic review of the evidence. Tob Induc Dis 2020;18:20. https://doi.org/10.18332/tid/119324 10.18332/tid/119324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [84].Bazzini C, Rossetti V, Civello DA, Sassone F, Vezzoli V, Persani L, Tiberio L, Lanata L, Bagnasco M, Paulmichl M, Meyer G, Garavaglia ML. Short- and long- term effects of cigarette smoke exposure on glutathione homeostasis in human bronchial epithelial cells. Cell Physiol Biochem 2013;32:129-45. https://doi.org/10.1159/000356633 10.1159/000356633 [DOI] [PubMed] [Google Scholar]
- [85].van der Toorn M, Smit-de Vries MP, Slebos DJ, de Bruin HG, Abello N, van Oosterhout AJ, Bischoff R, Kauffman HF. Cigarette smoke irreversibly modifies glutathione in airway epithelial cells. Am J Physiol Lung Cell Mol Physiol 2007;293:L1156-62. https://doi.org/10.1152/ajplung.00081.2007 10.1152/ajplung.00081.2007 [DOI] [PubMed] [Google Scholar]
- [86].Gould NS, Min E, Huang J, Chu HW, Good J, Martin RJ, Day BJ. Glutathione depletion accelerates cigarette smoke-induced inflammation and airspace enlargement. Toxicol Sci 2015;147:466-74. https://doi.org/10.1093/toxsci/kfv143 10.1093/toxsci/kfv143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [87].De Flora S, Izzotti A, D’Agostini F, Balansky RM. Mechanisms of N-acetylcysteine in the prevention of DNA damage and cancer, with special reference to smoking-related end-points. Carcinogenesis 2001;22:999-1013. [DOI] [PubMed] [Google Scholar]
- [88].van Schooten FJ, Besaratinia A, De Flora S, D’Agostini F, Izzotti A, Camoirano A, Balm AJ, Dallinga JW, Bast A, Haenen GR, Van’t Veer L, Baas P, Sakai H, Van Zandwijk N. Effects of oral administration of N-acetyl-L-cysteine: a multi-biomarker study in smokers. Cancer Epidemiol Biom Prev 2002:11;167-75. [PubMed] [Google Scholar]
- [89].Aruoma OI, Halliwell B, Hoey BM, Butler J. The antioxidant action of N-acetylcysteine: its reaction with hydrogen peroxide, hydroxyl radical, superoxide, and hypochlorous acid. Free Radic Biol Med 1989;6:593-7. https://doi.org/10.1016/0891-5849(89)90066-x 10.1016/0891-5849(89)90066-x [DOI] [PubMed] [Google Scholar]
- [90].Tardiolo G, Bramanti P, Mazzon E. Overview on the effects of N-acetylcysteine in neurodegenerative diseases. Molecules 2018;23:3305. https://doi.org/10.3390/molecules23123305 10.3390/molecules23123305 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [91].Hui DS, Lee N. Adjunctive therapies and immunomodulating agents for severe influenza. Influenza Other Respir Viruses 2013;7,3:52-9. https://doi.org/10.1111/irv.12171 10.1111/irv.12171 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [92].Zhou Y, Wang HD, Zhou XM, Fang J, Zhu L, Ding K.N-acetylcysteine amide provides neuroprotection via Nrf2-ARE pathway in a mouse model of traumatic brain injury. Drug Des Devel Ther 2018;12:4117-27. https://doi.org/10.2147/DDDT.S179227 10.2147/DDDT.S179227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [93].Zhao N, Guo FF, Xie KQ, Zeng T. Targeting Nrf-2 is a promising intervention approach for the prevention of ethanol-induced liver disease. Cell Mol Life Sci 2018;75:3143-57. https://doi.org/10.1007/s00018-018-2852-6 10.1007/s00018-018-2852-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [94].Wagener FADTG, Pickkers P, Peterson SJ, Immenschuh S, Abraham NG. Targeting the heme-heme oxygenase system to prevent severe complications following COVID-19 infections. Antioxidants (Basel) 2020;9:E540. https://doi.org/10.3390/antiox9060540 10.3390/antiox9060540 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [95].Liu M, Pelling JC, Ju J, Chu E, Brash DE. Antioxidant action via p53-mediated apoptosis. Cancer Res 1998;58:1723-9. [PubMed] [Google Scholar]
- [96].Loscalzo J. Nitric oxide insufficiency, platelet activation, and arterial thrombosis. Circ Res 2001;88:756-62. https://doi.org/10.1161/hh0801.089861 10.1161/hh0801.089861 [DOI] [PubMed] [Google Scholar]
- [97].Griendling KK, Sorescu D, Ushio-Fukai M. NAD(P)H oxidase: role in cardiovascular biology and disease. Circ Res 2000;86:494-501. https://doi.org/10.1161/01.res.86.5.494 10.1161/01.res.86.5.494 [DOI] [PubMed] [Google Scholar]
- [98].Guglielmetti G, Quaglia M, Sainaghi PP, Castello LM, Vaschetto R, Pirisi M, Corte FD, Avanzi GC, Stratta P, Cantaluppi V. “War to the knife” against thromboinflammation to protect endothelial function of COVID-19 patients. Crit Care 2020;24:365. https://doi.org/10.1186/s13054-020-03060-9 10.1186/s13054-020-03060-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [99].Zuin R, Palamidese A, Negrin R, Catozzo L, Scarda A, Balbinot M. High-dose N-acetylcysteine in patients with exacerbations of chronic obstructive pulmonary disease. Cl Drug Invest 2005;25:401-8. https://doi.org/10.2165/00044011-200525060-00005 10.2165/00044011-200525060-00005 [DOI] [PubMed] [Google Scholar]
- [100].Qi Q, Ailiyaer Y, Liu R, Zhang Y, Li C, Liu M, Wang X, Jing L, Li Y. Effect of N-acetylcysteine on exacerbations of bronchiectasis (BENE): a randomized controlled trial. Respir Res 2019;20:73. https://doi.org/10.1186/s12931-019-1042-x 10.1186/s12931-019-1042-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- [101].Aldini G, Altomare A, Baron G, Vistoli G, Carini M, Borsani L, Sergio F. N-Acetylcysteine as an antioxidant and disulphide breaking agent: the reasons why. Free Radic Res 2018;52:751-62. https://doi.org/10.1080/10715762.2018.1468564. 10.1080/10715762.2018.1468564 [DOI] [PubMed] [Google Scholar]
- [102].Wallace JL, Blackler RW, Chan MV, Da Silva GJ, Elsheikh W, Flannigan KL, Gamaniek I, Manko A, Wang L, Motta JP, Buret AG. Anti-inflammatory and cytoprotective actions of hydrogen sulfide: translation to therapeutics. Antioxid Redox Signal 2015;22:398-410. https://doi.org/10.1089/ars.2014.5901 10.1089/ars.2014.5901 [DOI] [PubMed] [Google Scholar]
- [103].Andreou A, Trantza S, Filippou D, Sipsas N, Tsiodras S. COVID-19: The potential role copper and N-acetylcysteine (NAC) in a combination of candidate antiviral treatments against SARS-CoV-2. In Vivo 2020;34:1567-88. https://doi.org/10.21873/invivo.11946.1007/s40121-020-00303-8 10.21873/invivo.11946.1007/s40121-020-00303-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [104].Bauer SR, Kapoor A, Rath M, Thomas SA. What is the role of supplementation with ascorbic acid, zinc, vitamin D, or N-acetylcysteine for prevention or treatment of COVID-19? Cleve Clin J Med 2020. https://doi.org/10.3949/ccjm.87a.ccc046 10.3949/ccjm.87a.ccc046 [DOI] [PubMed] [Google Scholar]
- [105].Gasmia A, Tippairoteb T, Mujawdiyad PK, Peana M, Menzelf A, Dadarg M, Gasmi Benahmedh A, Bjørklund G. Micronutrients as immunomodulatory tools for COVID-19 management. Clin Immunol 2020;220:108545. https://doi.org/10.1016/j.clim.2020.108545 10.1016/j.clim.2020.108545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [106].Jaiswal N, Bhatnagarb M, Shahc H. N-acetycysteine: A potential therapeutic agent in COVID-19 infection. Medical Hypotheses 2020;144:110133. https://doi.org/10.1016/j.mehy.2020.110133 10.1016/j.mehy.2020.110133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [107].McCarty MF, DiNicolantonio JJ. Nutraceuticals have potential for boosting the type 1 interferon response to RNA viruses including influenza and coronavirus. Prog Cardiovasc Dis 2020;S0033-0620(20)30037-2. https://doi.org/10.1016/j.pcad.2020.02.007 10.1016/j.pcad.2020.02.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [108].Nasi A, McArdle S, Gaudernack G, Westman G, Melief C, Rockberg J, Arens R, Kouretas D, Sjölin J, Mangsbo S. Reactive oxygen species as an initiator of toxic innate immune responses in retort to SARS-CoV-2 in an ageing population, consider N-acetylcysteine as early therapeutic intervention. Toxicol Rep 2020;18:768-71. https://doi.org/10.1016/j.toxrep.2020.06.003 10.1016/j.toxrep.2020.06.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [109].Poe FL, Corn J. N-Acetylcysteine: a potential therapeutic agent for SARS-CoV-2. Med Hypotheses 2020;143:109862. https://doi.org/10.1016/j.mehy.2020.109862. 10.1016/j.mehy.2020.109862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [110].Jorge-Aarón RM, Rosa-Ester MP. N-acetylcysteine as a potential treatment for COVID-19. Future Microbiol 2020;15:959-62. https://doi.org/10.2217/fmb-2020-0074 10.2217/fmb-2020-0074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [111].Sahebnasagh A, Saghafi F, Avan R, A, Khoshi A, Khataminia M, Safdari M, Habtemariam S, Rezai Ghaleno H, Nabavi SM. The prophylaxis and treatment potential of supplements for COVID-19. Eur J Pharmacol 2020;1:173530. https://doi.org/10.1016/j.ejphar.2020.173530. 10.1016/j.ejphar.2020.173530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [112].Spearow JL, Copeland L. Review: improving therapeutics for COVID-19 with glutathione-boosting treatments that improve immune responses and reduce the severity of viral infections. 2020. https://doi.org/10.31219/osf.io/y7wc2 10.31219/osf.io/y7wc2 [DOI] [Google Scholar]
- [113].Tolosa MTS, Genuino RF. Should N-acetylcysteine be used as adjunct treatment for COVID-19? Asia Pacific Center for Evidence Based Healthcare 2020. Available at: https://www.psmid.org/wp-content/uploads/2020/05/NAC-Abridged-25May2020-MTS-.pdf (accessed on: October 3, 2020). [Google Scholar]
- [114].van Hecke O, Lee J. N-acetylcysteine: A rapid review of the evidence for effectiveness in treating COVID-19. 2020. - cebm.net. Available at: https://www.cebm.net/covid-19/n-acetylcysteine-a-rapid-review-of-the-evidence-for-effectiveness-in-treating-covid-19 (accessed on: October 6, 2020). [Google Scholar]
- [115].Al-Horani RA, Kar S. Potential anti-SARS-CoV-2 therapeutics that target the post-entry stages of the viral life cycle: a comprehensive review. Viruses 2020;12:E1092. https://doi.org/10.3390/v12101092 10.3390/v12101092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [116].Sharafkhah M, Abdolrazaghnejad A, Zarinfar N, Mohammadbeigi A, Massoudifar A, Abaszadeh S. Safety and efficacy of N-acetyl-cysteine for prophylaxis of ventilator-associated pneumonia: a randomized, double blind, placebo-controlled clinical trial. Med Gas Res 2018;8:19-23. https://doi.org/10.4103/2045-9912.229599 10.4103/2045-9912.229599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [117].Bhattacharya R, Mondal M, Naiya SB, Lyngdoh L, Mukherjee R, Singh PK. The beneficial role of N-acetylcysteine as an adjunctive drug in treatment of COVID-19 patients in a tertiary care hospital in India: an observational study. Int J Res Med Sci 2020;8:3518-22. 10.18203/2320-6012.ijrms20204010 [DOI] [Google Scholar]
- [118].Sestili P, Fimognari C. Paracetamol use in COVID-19: friend or enemy? Preprints 2020,2020080186. [Google Scholar]
- [119].Sestili P, Fimognari C. Paracetamol-induced glutathione consumption: is there a link with severe COVID-19 illness? Front Pharmacol 2020. 10.3389/fphar.2020.579944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- [120].de Alencar JCG, de Lucena Moreira C, Dudy Müller A, Chaves CE, Akemi Fukuhara M, da Silva EA, Miyamoto MFS, Barbosa Pinto V, Gasparotto Bueno C, Neto FL, Gomez LM, Saad Menezes MC, Meirelles Marchini JF, Oliveira Marino L, Neto RAB, Souza HP, Covid Register Group . Double-blind, randomized, placebo-controlled trial with N-acetylcysteine for treatment of severe acute respiratory syndrome caused by COVID-19. Clin Inf Dis 2020;72:e736-e741. https://doi.org/10.1093/cid/ciaa1443 10.1093/cid/ciaa1443 [DOI] [PMC free article] [PubMed] [Google Scholar]


