Abstract
Background
This study aimed to determine the relative and independent contributions of impaired metabolic health and obesity to critical coronavirus disease 2019 (COVID-19).
Methods
We analyzed 4069 COVID-19 patients between January and June 2020 in South Korea, classified into four groups according to metabolic health status and body mass index (BMI): metabolically healthy normal weight (MHNW), metabolically unhealthy normal weight (MUNW), metabolically healthy obesity (MHO), and metabolically unhealthy obesity (MUO). The primary outcome was a composite of intensive care unit (ICU) admission, invasive mechanical ventilation (IMV), extracorporeal membrane oxygenation (ECMO), and death. Multivariable Cox proportional hazard regression models were used to estimate the hazard ratio (HR) for the outcome.
Results
The incidence rate (per 100 person-months) of critical COVID-19 was the lowest in the MHNW group (0.90), followed by the MHO (1.64), MUNW (3.37), and MUO (3.37) groups. Compared with MHNW, a significantly increased risk of critical COVID-19 was observed in MUNW (HR, 1.41; 95% CI, 1.01–1.98) and MUO (HR, 1.77; 95% CI, 1.39–2.44) but not in MHO (HR, 1.48; 95% CI, 0.98–2.23). The risk of ICU admission or IMV/ECMO was increased only in MUO; however, the risk of death was significantly higher in MUNW and MUO. The risk of critical COVID-19 increased insignificantly by 2% per 1 kg/m2 BMI increase but significantly by 13% per 1 metabolically unhealthy component increase, even after mutually adjusting for BMI and metabolic health status.
Conclusions
Metabolic health is more important to COVID-19 outcomes than obesity itself, suggesting that metabolic health status should be considered for a precise and tailored management of COVID-19 patients.
Keywords: COVID-19, Metabolic health, Metabolic syndrome, Body mass index, Adverse clinical outcome
1. Introduction
Earlier observations revealed that patients with severe forms of COVID-19 were more obese than those with non-severe disease [1,2]. Multiple studies have indicated that obesity, which is generally represented by a high body mass index (BMI), is associated with the severity and mortality of COVID-19 [[3], [4], [5], [6], [7]]. However, obesity is a predisposing condition for metabolic derangement and cardiovascular diseases [8], which have also been recognized as important risk factors for adverse outcomes of COVID-19 [9,10]. Therefore, the observed association between obesity and critical COVID-19 might be biased by the effects of comorbidities. Previous studies have attempted to solve this problem by adjusting for comorbidities such as diabetes, hypertension, and dyslipidemia [[11], [12], [13], [14]]; however, this might be insufficient to identify a direct relationship between obesity and COVID-19 outcomes.
Many differences in the determining characteristics of metabolic phenotypes have been observed between East Asians and other ethnic groups. For example, a substantial number of East Asian patients with type 2 diabetes were reported to be nonobese, although the prevalence of type 2 diabetes was similar to that in Caucasians [15]. This observation directed much interest to metabolically unhealthy without obesity phenotypes. However, metabolically healthy with obesity phenotypes have also been observed [16].
We hypothesized that metabolic health beyond obesity might be crucial in determining the risk of COVID-19 outcomes. The objective of this large retrospective study was to investigate the relative contribution of obesity and metabolic health status to critical outcomes of COVID-19 patients in a nationwide cohort in South Korea.
2. Materials and methods
2.1. Data source and study population
The Korean National Health Insurance Service (NHIS), which is the sole mandatory public medical insurance system for all citizens of South Korea, recently released the NHIS-COVID-19 cohort database, which included 8080 patients with COVID-19 confirmed with a SARS-CoV-2 PCR test between January 1, 2020, and June 4, 2020. The database incorporated past longitudinal information of the patients before the diagnosis of COVID-19, including demographic, medical, and pharmaceutical data from 2015 to 2020. The relevant information included medical history based on the International Classification of Disease 10th revision (ICD-10), hospitalization including ICU admission, drug prescriptions, medical procedures, and anthropometric and biochemical laboratory information including body weight, height, waist circumference, systolic and diastolic blood pressure, fasting plasma glucose, and lipid profile. In addition, this database was merged with death records managed by the Korean National Statistical Office. All patients were followed up until 4 months after the diagnosis of COVID-19 or until death. A more detailed protocol has been previously published [17]. We presented the STORBE checklist in Supplementary Table S1.
This study was approved by the Institutional Review Board of Korea University Anam Hospital (approval no. 2020AN0482). The requirement for informed consent was waived because all patient data were anonymized and de-identified.
2.2. Study definition and outcomes
This is a nationwide retrospective cohort study. Supplementary Fig. S1 shows the diagram of the selection of the study patients. Among patients with laboratory confirmed COVID-19, those with any missing variables for the identification of metabolic status were excluded. We divided these patients into four groups according to the obesity and metabolic health status: (i) metabolically healthy normal weight (MHNW), (ii) metabolically unhealthy normal weight (MUNW), (iii) metabolically healthy obesity (MHO), and (iv) metabolically unhealthy obesity (MUO). Obesity was defined as BMI ≥ 25 kg/m2 according to the Asia-Pacific BMI criteria, calculated as weight divided by height squared (kg/m2) [18]. A metabolically unhealthy status was defined as the presence of three or more of the following factors: (i) fasting plasma glucose level ≥ 100 mg/dL or current use of glucose-lowering agents under the ICD-10 codes for diabetes mellitus (E10–E14), (ii) blood pressure ≥ 130/85 mm Hg or use of antihypertensive agents under the ICD-10 codes for hypertension (I10–I15), (iii) serum triglyceride level ≥ 150 mg/dL or current use of lipid-lowering agents under the ICD-10 code for dyslipidemia (E78), (iv) high-density lipoprotein-cholesterol level < 40 mg/dL in men or <50 mg/dL in women or current use of lipid-lowering agents under the ICD-10 code for dyslipidemia (E78), and (v) waist circumference > 90 cm in men or ≥85 cm in women based on the International Diabetes Federation criteria for Asians [18,19].
The primary outcome was a composite of ICU admission, invasive mechanical ventilation (IMV), extracorporeal membrane oxygenation (ECMO), and death of any cause from the diagnosis of COVID-19 to the end of follow-up.
2.3. Statistical analyses
Continuous data are presented as mean ± standard deviation (SD) values for normally distributed variables and as medians and interquartile ranges for nonnormally distributed variables. Categorical data are presented as frequencies and percentages. ANOVA and Pearson's ꭓ2 test were used to compare baseline characteristics among the four groups.
We calculated the incidence of the primary composite outcome by dividing the total number of events by the total follow-up period (person-months). Kaplan–Meier analysis followed by the log-rank test was used to examine differences in the cumulative incidence of the composite primary outcome according to the metabolic health and obesity phenotypes. We also used multivariable Cox proportional hazard regression models to analyze the hazard ratios (HRs) for the study outcomes according to each metabolic health and obesity phenotype. Model 1 was adjusted for age and sex, and model 2 was adjusted for all possible confounders including age, sex, smoking, alcohol consumption, physical activity, socioeconomic status, previous history of chronic pulmonary disease including asthma and chronic obstructive pulmonary disease. As general obesity (represented by a high BMI) and metabolic health are closely associated with each other, we further investigated their independent roles in the development of critical COVID-19 by further adjusting for BMI and metabolically healthy status mutually. For handling the missing data, available-case analyses (also known as pairwise deletion) were used without additional data handing.
All reported p-values were two-sided, and statistical significance was set at p < 0.05. All statistical analyses were performed using SAS software (version 9.4; SAS Institute Inc., Cary, NC, USA).
3. Results
Of patients with laboratory confirmed COVID-19 from the original NHIS-COVID-19 cohort database (n = 8080), those with any missing variables (n = 4011) for the identification of metabolic status, including BMI, waist circumference, fasting plasma glucose, blood pressure, and serum levels of triglyceride and high-density lipoprotein-cholesterol, were excluded. Finally, a total of 4069 patients were selected.
3.1. Baseline patient characteristics
The mean (±SD) age of the patients was 55.5 ± 14.2 years, and 2539 (62.4%) were female. The mean BMI was 24.0 kg/m2. A total of 879 patients (21.6%) had a history of cardiovascular disease, 1310 (32.2%) had hypertension and 1013 (24.9%) had chronic pulmonary disease. The baseline characteristics of patients in the four groups according to metabolic health and obesity status are listed in Table 1 . In general, patients in the metabolically unhealthy groups (MUNW and MUO) were older than those in the metabolically healthy groups (MHNW and MHO). The proportion of male patients was the lowest in MHNW, followed by MUNW, MUO, and MHO. Metabolic parameters, including systolic/diastolic blood pressure and serum glucose/triglyceride levels, were higher in the metabolically unhealthy groups than in the metabolically healthy groups. Metabolically unhealthy patients also had a higher prevalence of comorbidities, including hypertension, diabetes mellitus, dyslipidemia, cardiovascular disease, chronic kidney disease, and chronic pulmonary disease than metabolically healthy patients.
Table 1.
Characteristics of patients with coronavirus disease 2019 according to metabolic health and obesity phenotypes.
| MHNW | MUNW | MHO | MUO | p-Value | |
|---|---|---|---|---|---|
| n (%) | 1871 (46.0) | 728 (17.9) | 595 (14.6) | 875 (21.5) | |
| Follow-up time, mean (SD), days | 118.0 (10.2) | 111.9 (26.2) | 117.0 (14.5) | 113.0 (24.3) | |
| Age group, n (%), years | <0.001 | ||||
| 20–29 | 149 (8.0) | 3 (0.4) | 39 (6.6) | 7 (0.8) | |
| 30–39 | 228 (12.2) | 9 (1.2) | 40 (6.7) | 41 (4.7) | |
| 40–49 | 430 (23.0) | 45 (6.2) | 124 (20.8) | 112 (12.8) | |
| 50–59 | 604 (32.3) | 179 (24.6) | 192 (32.3) | 238 (27.2) | |
| 60–69 | 326 (17.4) | 246 (33.8) | 96 (16.1) | 276 (31.5) | |
| 70–79 | 95 (5.1) | 165 (22.7) | 41 (6.9) | 135 (15.4) | |
| ≥80 | 39 (2.1) | 81 (11.1) | 13 (2.2) | 66 (7.5) | |
| Male sex, n (%) | 522 (27.9) | 300 (41.2) | 298 (50.1) | 410 (46.9) | <0.001 |
| BMI, mean (SD), kg/m2 | 21.8 (2.0) | 22.7 (1.7) | 27.0 (2.1) | 27.9 (2.6) | <0.001 |
| WC, mean (SD), cm | 74.6 (6.8) | 80.2 (6.5) | 85.8 (7.3) | 90.7 (7.4) | <0.001 |
| Men | 79.6 (6.1) | 83.3 (5.5) | 88.8 (6.6) | 92.9 (7.2) | <0.001 |
| Women | 72.7 (6.0) | 78.0 (6.3) | 82.8 (6.8) | 88.7 (7.1) | <0.001 |
| SBP, mean (SD), mm Hg | 115.0 (13.2) | 128.2 (15.2) | 121.0 (13.1) | 130.3 (14.4) | <0.001 |
| DBP, mean (SD), mm Hg | 71.4 (9.2) | 77.6 (9.9) | 75.3 (8.9) | 79.7 (9.9) | <0.001 |
| FPG, mean (SD), mg/dL | 93.4 (15.9) | 113.3 (34.7) | 93.9 (12.1) | 113.6 (31.1) | 0.001 |
| Total cholesterol, mean (SD), mg/dL | 192.0 (34.7) | 196.6 (44.5) | 197.1 (31.7) | 197.3 (43.0) | <0.001 |
| Triglyceride, mean (SD), mg/dL | 92.1 (47.3) | 150.2 (91.1) | 111.8 (60.3) | 165.5 (119.2) | <0.001 |
| HDL-C, mean (SD), mg/dL | 61.4 (17.4) | 53.5 (14.4) | 55.5 (12.8) | 50.4 (12.3) | <0.001 |
| Hemoglobin, mean (SD), g/dL | 13.4 (1.5) | 13.7 (1.5) | 14.1 (1.6) | 14.2 (1.6) | <0.001 |
| Creatinine, mean (SD), mg/dL | 0.80 (0.29) | 0.86 (0.30) | 0.86 (0.20) | 0.90 (0.38) | <0.001 |
| AST, mean (SD), IU/L | 22.5 (8.6) | 26.2 (13.2) | 25.4 (19.0) | 31.1 (37.0) | <0.001 |
| ALT, mean (SD), IU/L | 19.0 (12.1) | 23.8 (13.2) | 25.6 (25.5) | 34.8 (52.5) | <0.001 |
| Smoking, n (%) | <0.001 | ||||
| Never | 1577 (84.3) | 500 (68.7) | 444 (74.6) | 612 (69.9) | |
| Former | 200 (10.7) | 120 (16.5) | 100 (16.8) | 184 (21.0) | |
| Current | 94 (5.0) | 58 (8.0) | 51 (8.6) | 79 (9.0) | |
| Alcohol consumption, n (%) | 313 (16.7) | 106 (14.6) | 159 (26.7) | 203 (23.2) | <0.001 |
| Regular exercise, n (%) | 805 (43.0) | 338 (46.4) | 262 (44.0) | 429 (49.0) | 0.023 |
| Moderate-to-vigorous physical activity, n (%) | 93 (5.0) | 45 (6.2) | 32 (5.4) | 45 (5.1) | 0.663 |
| Low SESa, n (%) | 430 (23.0) | 178 (24.5) | 117 (19.7) | 199 (22.7) | 0.214 |
| Comorbidities, n (%) | |||||
| Hypertension | 200 (10.7) | 464 (63.7) | 95 (16.0) | 551 (63.0) | <0.001 |
| Diabetes mellitus | 33 (1.8) | 264 (36.3) | 8 (1.3) | 287 (32.8) | <0.001 |
| Dyslipidemia | 162 (8.7) | 597 (82.0) | 41 (6.9) | 617 (70.5) | <0.001 |
| Cardiovascular disease | 227 (12.1) | 294 (40.4) | 66 (11.1) | 292 (33.4) | <0.001 |
| Chronic kidney disease | 8 (0.4) | 22 (3.0) | 4 (0.7) | 35 (4.0) | <0.0001 |
| Chronic pulmonary disease | 384 (20.5) | 227 (31.2) | 121 (20.3) | 281 (32.1) | <0.001 |
Abbreviations: MHNW, metabolically healthy normal weight; MUNW, metabolically unhealthy normal weight; MHO, metabolically healthy obesity; MUO, metabolically unhealthy obesity; SD, standard deviation; BMI, body mass index; WC, waist circumference; SBP, systolic blood pressure; DBP, diastolic blood pressure; FPG, fasting plasma glucose; HDL-C, high-density lipoprotein-cholesterol; AST, aspartate aminotransferase; ALT, alanine aminotransferase; SES, socioeconomic status.
Socioeconomic status was identified according to the medical insurance premium in the database, in which low socioeconomic status was defined as the lower 30%.
3.2. Severe COVID-19 outcomes according to metabolic health and obesity status
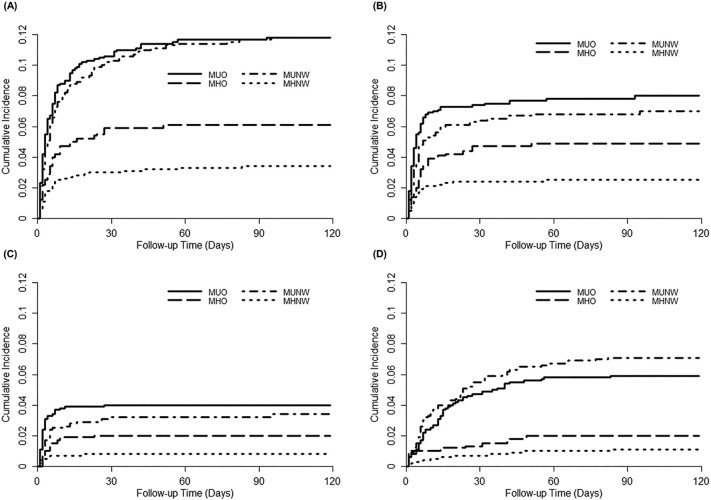
During about 4 months of follow-up, 289 (7.1%) critical COVID-19 outcomes occurred. The incidence rate (per 100 person-months) of the outcomes was higher in metabolically unhealthy patients (3.37 in both MUNW and MUO) than in metabolically healthy patients (0.90 in MHNW and 1.64 in MHO). Table 2 shows the HRs for the composite and individual COVID-19 outcomes in the four groups. Compared with the MHNW group as the reference, all three other groups had significantly higher HRs for the composite of critical COVID-19 outcomes in an unadjusted model (1.79 [1.19–2.70], 3.58 [2.59–4.95], and 3.59 [2.63–4.91] in MHO, MUNW, and MUO, respectively), indicating that both obesity and impaired metabolic health are important risk factors for critical COVID-19. However, after adjusting for confounding variables (in two different models), patients with a metabolically unhealthy status (MUNW and MUO groups) still had a significantly higher risk of critical COVID-19, but patients in the MHO group did not. In model 2, the risk of critical COVID-19 was 41% higher in the MUNW group and 77% higher in the MUO group than in the MHNW group. The MUNW and MUO groups also had a significantly higher risk of death than the MHNW group, although the risks of ICU admission and IMV or ECMO requirement were higher only in the MUO group. Kaplan–Meier curves showed a time-dependent risk of each outcome in the four groups (Fig. 1 ). All outcomes were determined to occur early, mostly within 30 days after the diagnosis of COVID-19.
Table 2.
Severe COVID-19 outcomes according to metabolic health and obesity phenotypes.
| Events, n | Incidence rate, per 100 person-months | Hazard ratio (95% CI) |
|||
|---|---|---|---|---|---|
| Unadjusted | Model 1 | Model 2 | |||
| Composite of severe COVID-19 outcomes | |||||
| MHNW | 64 | 0.90 | 1 (ref.) | 1 (ref.) | 1 (ref.) |
| MUNW | 86 | 3.37 | 3.58 (2.59–4.95) | 1.42 (1.02–1.99) | 1.41 (1.01–1.98) |
| MHO | 36 | 1.64 | 1.79 (1.19–2.70) | 1.42 (0.94–2.14) | 1.48 (0.98–2.23) |
| MUO | 103 | 3.37 | 3.59 (2.63–4.91) | 1.80 (1.31–2.47) | 1.77 (1.29–2.44) |
| ICU admission | |||||
| MHNW | 47 | 0.66 | 1 (ref.) | 1 (ref.) | 1 (ref.) |
| MUNW | 50 | 1.96 | 2.82 (1.89–4.20) | 1.43 (0.94–2.18) | 1.45 (0.95–2.20) |
| MHO | 29 | 1.32 | 1.96 (1.24–3.12) | 1.60 (1.00–2.55) | 1.59 (0.99–2.54) |
| MUO | 69 | 2.26 | 3.26 (2.25–4.72) | 1.92 (1.31–2.81) | 1.92 (1.31–2.82) |
| IMV or ECMO | |||||
| MHNW | 15 | 0.21 | 1 (ref.) | 1 (ref.) | 1 (ref.) |
| MUNW | 24 | 0.91 | 4.19 (2.20–7.98) | 1.83 (0.94–3.58) | 1.87 (0.95–3.65) |
| MHO | 12 | 0.53 | 2.54 (1.19–5.42) | 1.99 (0.93–4.27) | 2.02 (0.94–4.33) |
| MUO | 35 | 1.10 | 5.11 (2.79–9.36) | 2.72 (1.47–5.05) | 2.65 (1.42–4.94) |
| Death | |||||
| MHNW | 21 | 0.29 | 1 (ref.) | 1 (ref.) | 1 (ref.) |
| MUNW | 52 | 1.94 | 6.56 (3.95–10.89) | 1.85 (1.11–3.10) | 1.90 (1.13–3.19) |
| MHO | 12 | 0.52 | 1.81 (0.89–3.67) | 1.30 (0.64–2.65) | 1.44 (0.71–2.94) |
| MUO | 52 | 1.60 | 5.43 (3.27–9.01) | 2.23 (1.34–3.71) | 2.22 (1.33–3.70) |
Abbreviations: COVID-19, coronavirus disease 2019; MHNW, metabolically healthy normal weight; MUNW, metabolically unhealthy normal weight; MHO, metabolically healthy obesity; MUO, metabolically unhealthy obesity; ICU, intensive care unit; IMV, invasive mechanical ventilation; ECMO, extracorporeal membrane oxygenation.
Model 1 was adjusted for age and sex.
Model 2 was adjusted for age, sex, smoking, alcohol consumption, physical activity, socioeconomic status, and previous history of chronic pulmonary disease, including asthma and chronic obstructive pulmonary disease.
Fig. 1.
Kaplan–Meier analysis of severe coronavirus disease 2019 outcomes (A), ICU admission (B), IMV or ECMO therapy (C), and death (D) according to metabolic health and obesity phenotypes.
*Abbreviations: MHNW, metabolically healthy weight; MUNW, metabolically unhealthy normal weight; MHO, metabolically healthy obesity; MUO, metabolically unhealthy obesity; ICU, intensive care unit; IMV, invasive mechanical ventilation; ECMO, extracorporeal membrane oxygenation.
3.3. Severe COVID-19 outcomes according to BMI category and the number of metabolically unhealthy components
In an unadjusted model, the risk of critical COVID-19 was increased by 2% per 1 kg/m2 increase in BMI and by 13% for every 1 component increase in metabolically unhealthy parameters (Table 3 ). However, after reciprocal adjustment for each variable and other confounding factors (in model 2), only the number of metabolically unhealthy parameters was associated with the risk of critical COVID-19.
Table 3.
Severe coronavirus disease 2019 outcomes according to body mass index category and the number of metabolically unhealthy components.
| n | Events, n | Incidence rate, per 100 person-months | Hazard ratio (95% CI) |
|||
|---|---|---|---|---|---|---|
| Unadjusted | Model 1 | Model 2 | ||||
| BMI, kg/m2 | ||||||
| <20 | 393 | 25 | 1.72 | 1.07 (0.68–1.69) | 1.22 (0.77–1.92) | 1.22 (0.77–1.93) |
| 20–22.9 | 1194 | 71 | 1.60 | 1 (ref.) | 1 (ref.) | 1 (ref.) |
| 23–24.9 | 1012 | 54 | 1.43 | 0.90 (0.63–1.28) | 0.78 (0.55–1.11) | 0.72 (0.51–1.03) |
| 25–29.9 | 1279 | 126 | 2.77 | 1.69 (1.27–2.27) | 1.28 (0.96–1.72) | 1.19 (0.88–1.60) |
| ≥30 | 191 | 13 | 1.86 | 1.15 (0.64–2.08) | 1.46 (0.81–2.64) | 1.16 (0.63–2.14) |
| p-Value for trend | 0.002 | 0.129 | 0.118 | |||
| Per 1 kg/m2 increase | 1.04 (1.01–1.08) | 1.03 (0.99–1.07) | 1.02 (0.98–1.06) | |||
| No. of MU components | ||||||
| 0 | 945 | 19 | 0.52 | 1 (ref.) | 1 (ref.) | 1 (ref.) |
| 1 | 793 | 35 | 1.17 | 2.22 (1.27–3.88) | 1.41 (0.81–2.48) | 1.14 (0.80–2.49) |
| 2 | 728 | 46 | 1.71 | 3.21 (1.88–5.48) | 1.47 (0.85–2.52) | 1.43 (0.83–2.49) |
| 3 | 650 | 55 | 2.35 | 4.34 (2.58–7.32) | 1.65 (0.97–2.81) | 1.62 (0.94–2.80) |
| 4 | 649 | 91 | 4.09 | 7.36 (4.49–12.06) | 2.14 (1.28–3.56) | 2.04 (1.20–3.47) |
| 5 | 304 | 43 | 4.16 | 7.47 (4.35–12.82) | 2.02 (1.16–3.54) | 1.84 (0.99–3.41) |
| p-Value for trend | <0.001 | 0.001 | 0.001 | |||
| Per 1 component increase | 1.47 (1.37–1.59) | 1.15 (1.06–1.25) | 1.13 (1.03–1.24) | |||
Abbreviations: BMI, body mass index; MU, metabolically unhealthy; ref., reference.
Model 1 was adjusted for age and sex.
Model 2 was adjusted for age, sex, smoking, alcohol consumption, physical activity, socioeconomic status, and previous history of chronic pulmonary disease (including asthma and chronic obstructive pulmonary disease) and further adjusted for metabolically unhealthy status and BMI.
4. Discussion
In this retrospective cohort study, we found that impaired metabolic health was a stronger predictor of critical COVID-19 than obesity itself in the Korean population. Notably, metabolically unhealthy individuals, either with obesity or not, had a higher risk of critical COVID-19 outcomes, including ICU admission and death, than metabolically healthy individuals without obesity. In contrast, metabolically healthy individuals with obesity were not associated with critical COVID-19. In addition, the risk of COVID-19 outcomes linearly increased with increasing number of metabolically unhealthy components independently of BMI, whereas BMI did not distinguish patients at a risk of critical COVID-19.
Metabolically unhealthy traits, generally manifesting as metabolic syndrome, have been proposed as important risk factors for adverse outcomes of COVID-19. Although the exact mechanisms underlying this association remain unknown, immunomodulation (such as a hyperimmune response) and increased expression of ACE2 (which is the viral entry receptor of SARS-CoV-2) partly explain the association between metabolic syndrome and COVID-19 outcomes [20,21]. However, these mechanisms may also be applicable to obesity-related COVID-19 outcomes. Thus, further studies are required to elucidate this issue.
Obesity is a heterogeneous condition. For example, individuals with the same BMI can show various phenotypes in terms of adipocyte biology, fat distribution, and body composition [22]. One of the important factors determining metabolically healthy and unhealthy obesity is the amount or distribution of visceral adipose tissue (VAT). Previous studies found that Asian diabetic patients had more VAT than Caucasian diabetic patients with the same waist circumference, which can explain the higher susceptibility of lean Asians to type 2 diabetes [23]. Given the close association of VAT with insulin resistance, and further with metabolic syndrome, this parameter could be a determinant of the metabolic health status at a given BMI [24]. Several studies have indicated that VAT could be an important indicator of COVID-19 severity. In a small-sample study, VAT was an independent risk factor for severe COVID-19 outcomes, but not BMI [25]. Another study showed that high VAT (≥128.5 cm2) was a crucial factor in predicting COVID-19 severity [26]. Although we were unable to validate this association in our cohort, we assumed that a higher proportion of VAT in the MUNW group than in the MHNW group might have contributed to the risk of critical COVID-19. Metabolically unhealthy phenotype is also related to adipose tissue dysfunction and inflammation, which ultimately result in ectopic fat deposition and insulin resistance [27]. Indeed, upregulation of inflammatory cytokines including TNF-α and IL-6 was frequently observed in patients with insulin resistance, even without general obesity [20]. This partly explains hyperimmune response to infectious insult including SARS-CoV-2 and adverse outcomes of COVID-19 in MUNW people. Another possible factor is reduced muscle mass characteristic of MUNW individuals [16]. Reduced muscle mass has been proposed to be independently associated with insulin resistance and increased susceptibility to adverse respiratory outcomes, including pneumonia and sepsis [28,29].
Nonetheless, the available evidence from multiple geographical regions generally supports that obesity contributes to the adverse outcomes of COVID-19. However, previous reports had substantial inconsistencies in terms of study population and measured outcomes. A linear association between BMI and the need for ICU admission or IMV was reported in multiple studies [5,30,31]. However, for the mortality risk from COVID-19, J-shaped or U-shaped associations with increasing BMI were frequently observed, indicating that both severe obesity and underweight are risk factors for fatal events related to COVID-19 [5,7,30,32]. Given that the proposed cutoff values of high BMI for death risk ranged from 30 to 40 kg/m2 in previous studies, the possibility exists that the much lower incidence of high BMI in our study population, with only 4.1% having a BMI of >30 kg/m2, may not detect the harmful effects of severe obesity. We also found a J-shaped association between BMI and critical COVID-19, with patients having a BMI of between 20 and 24.9 kg/m2 having the lowest risk of severe outcomes, although it was not significant. Therefore, ethnicity may be a crucial factor in determining the association between BMI and disease severity. Further, the fact that MUO patients still had the highest risk of severe COVID-19 and mortality in this study should not be ignored.
A notable result of our study was that the risk of critical COVID-19 increased with the increasing number of metabolically unhealthy components. This was significant even after adjusting for BMI, suggesting an independent association between metabolic derangement, rather than obesity itself, and COVID-19 severity. We found that individuals with four or more metabolic syndrome components had a two-fold higher risk of COVID-19 outcomes than metabolically healthy individuals.
Taken together, impaired metabolic health, represented by insulin resistance, is closely related to the severity of COVID-19. In this regard, it is worth paying attention to the research results that several drugs related to improvement in insulin resistance, particularly metformin and peroxisome proliferator-activated receptors agonists, are associated with better outcomes in COVID-19 [33,34].
This study had strengths and limitations. The cohort database included anthropometric and laboratory measurements before the diagnosis of COVID-19, which made it possible to identify the underlying metabolic conditions of the patients. In addition, body weight, height, and waist circumference were measured using standardized methods (not self-reported). However, only 50% of the cohort had data on all analyzed metabolic syndrome components, resulting in the exclusion of a large proportion of patients. Our data also included detailed information on patient care, such as dates of hospital admission and ICU admission, medical procedures, and death records. Thus, it was possible to analyze the time-dependent association between exposure and outcomes. Although the follow-up duration was relatively short (up to 4 months), we found that most outcomes occurred within 2 months after the diagnosis of COVID-19, indicating that the follow-up duration would not limit the reliability of the study results. Finally, there might be residual confounding by unmeasured covariates, such as quality of intensive care, occupation, and viral load of SARS-CoV-2 in affected individuals.
In conclusion, our study showed that metabolically unhealthy traits are important risk factors for critical COVID-19 independent of BMI in the Korean population. Considering that COVID-19 remains a tremendous public health problem, this study suggests that the metabolic health status of affected individuals should be evaluated and more attention needs to be paid to managing individuals with metabolically unhealthy traits.
CRediT authorship contribution statement
Nam Hoon Kim: Data curation, Investigation, Writing – original draft, Writing – review & editing. Kyeong Jin Kim: Data curation, Investigation, Writing – original draft. Jimi Choi: Methodology, Formal analysis, Data curation. Sin Gon Kim: Conceptualization, Investigation, Supervision, Project administration.
Declaration of competing interest
The authors report no potential conflicts of interest relevant to this article.
Acknowledgments
Acknowledgments
We thank the participants of the Korean Health Insurance Cohort study and the National Health Insurance Service, which developed the NHIS-COVID-19 cohort database.
Funding
This study was supported by a Korea University grant K1625581.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.metabol.2021.154894.
Appendix A. Supplementary data
Supplementary material
References
- 1.Cai Q., Chen F., Wang T., Luo F., Liu X., Wu Q., et al. Obesity and COVID-19 severity in a designated hospital in Shenzhen, China. Diabetes Care. 2020;43:1392–1398. doi: 10.2337/dc20-0576. [DOI] [PubMed] [Google Scholar]
- 2.Bhatraju P.K., Ghassemieh B.J., Nichols M., Kim R., Jerome K.R., Nalla A.K., et al. Covid-19 in critically ill patients in the Seattle region – case series. N Engl J Med. 2020;382:2012–2022. doi: 10.1056/NEJMoa2004500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Price-Haywood E.G., Burton J., Fort D., Seoane L. Hospitalization and mortality among black patients and white patients with Covid-19. N Engl J Med. 2020;382:2534–2543. doi: 10.1056/NEJMsa2011686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sattar N., Ho F.K., Gill J.M., Ghouri N., Gray S.R., Celis-Morales C.A., et al. BMI and future risk for COVID-19 infection and death across sex, age and ethnicity: preliminary findings from UK biobank. Diabetes Metab Syndr. 2020;14:1149–1151. doi: 10.1016/j.dsx.2020.06.060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gao M., Piernas C., Astbury N.M., Hippisley-Cox J., O’Rahilly S., Aveyard P., et al. Associations between body-mass index and COVID-19 severity in 6·9 million people in England: a prospective, community-based, cohort study. Lancet Diabetes Endocrinol. 2021;9:350–359. doi: 10.1016/S2213-8587(21)00089-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Palaiodimos L., Kokkinidis D.G., Li W., Karamanis D., Ognibene J., Arora S., et al. Severe obesity, increasing age and male sex are independently associated with worse in-hospital outcomes, and higher in-hospital mortality, in a cohort of patients with COVID-19 in the Bronx, New York. Metabolism. 2020;108:154262. doi: 10.1016/j.metabol.2020.154262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Guerson-Gil A., Palaiodimos L., Assa A., Karamanis D., Kokkinidis D., Chamorro-Pareja N., et al. Sex-specific impact of severe obesity in the outcomes of hospitalized patients with COVID-19: a large retrospective study from the Bronx, New York. Eur J Clin Microbiol Infect Dis. 2021;40:1963–1974. doi: 10.1007/s10096-021-04260-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sowers J.R. Obesity and cardiovascular disease. Clin Chem. 1998;44:1821–1825. [PubMed] [Google Scholar]
- 9.Xie J., Zu Y., Alkhatib A., Pham T.T., Gill F., Jang A., et al. Metabolic syndrome and COVID-19 mortality among adult black patients in New Orleans. Diabetes Care. 2020;44:188–193. doi: 10.2337/dc20-1714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lim S., Bae J.H., Kwon H.S., Nauck M.A. COVID-19 and diabetes mellitus: from pathophysiology to clinical management. Nat Rev Endocrinol. 2021;17:11–30. doi: 10.1038/s41574-020-00435-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gao F., Zheng K.I., Wang X.B., Sun Q.F., Pan K.H., Wang T.Y., et al. Obesity is a risk factor for greater COVID-19 severity. Diabetes Care. 2020;43:e72–e74. doi: 10.2337/dc20-0682. [DOI] [PubMed] [Google Scholar]
- 12.Kim L., Garg S., O’Halloran A., Whitaker M., Pham H., Anderson E.J., et al. Risk factors for intensive care unit admission and in-hospital mortality among hospitalized adults identified through the US Coronavirus Disease 2019 (COVID-19)-Associated Hospitalization Surveillance Network (COVID-NET) Clin Infect Dis. 2021;72:e206–e214. doi: 10.1093/cid/ciaa1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yates T., Zaccardi F., Islam N., Razieh C., Gillies C.L., Lawson C.A., et al. Obesity, ethnicity, and risk of critical care, mechanical ventilation, and mortality in patients admitted to hospital with COVID-19: analysis of the ISARIC CCP-UK Cohort. Obesity (Silver Spring) 2021;29:1223–1230. doi: 10.1002/oby.23178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chetboun M., Raverdy V., Labreuche J., Simonnet A., Wallet F., Caussy C., et al. 2020. Association of body mass index and other metabolic risk factors with pneumonia outcomes in critically ill patients with coronavirus disease-19: an international multicenter retrospective cohort study. Available from. [DOI] [Google Scholar]
- 15.Rhee E.J. Diabetes in Asians. Endocrinol Metab (Seoul) 2015;30:263–269. doi: 10.3803/EnM.2015.30.3.263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Stefan N. Metabolically healthy and unhealthy normal weight and obesity. Endocrinol Metab (Seoul) 2020;35:487–493. doi: 10.3803/EnM.2020.301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cho D.H., Lee S.J., Jae S.Y., Kim W.J., Ha S.J., Gwon J.G., et al. Physical activity and the risk of COVID-19 infection and mortality: a nationwide population-based case-control study. J Clin Med. 2021;10 doi: 10.3390/jcm10071539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.The Asia-Pacific perspective: redefining obesity and its treatment [article online] 2000. https://apps.who.int/iris/handle/10665/206936 Available from.
- 19.Alberti K.G., Eckel R.H., Grundy S.M., Zimmet P.Z., Cleeman J.I., Donato K.A., et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation. 2009;120:1640–1645. doi: 10.1161/CIRCULATIONAHA.109.192644. [DOI] [PubMed] [Google Scholar]
- 20.Batabyal R., Freishtat N., Hill E., Rehman M., Freishtat R., Koutroulis I. Metabolic dysfunction and immunometabolism in COVID-19 pathophysiology and therapeutics. Int J Obes (Lond) 2021;45:1163–1169. doi: 10.1038/s41366-021-00804-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Stefan N., Birkenfeld A.L., Schulze M.B. Global pandemics interconnected – obesity, impaired metabolic health and COVID-19. Nat Rev Endocrinol. 2021;17:135–149. doi: 10.1038/s41574-020-00462-1. [DOI] [PubMed] [Google Scholar]
- 22.Stefan N., Schick F., Häring H.U. Causes, characteristics, and consequences of metabolically unhealthy normal weight in humans. Cell Metab. 2017;26:292–300. doi: 10.1016/j.cmet.2017.07.008. [DOI] [PubMed] [Google Scholar]
- 23.Kadowaki T., Sekikawa A., Murata K., Maegawa H., Takamiya T., Okamura T., et al. Japanese men have larger areas of visceral adipose tissue than Caucasian men in the same levels of waist circumference in a population-based study. Int J Obes (Lond) 2006;30:1163–1165. doi: 10.1038/sj.ijo.0803248. [DOI] [PubMed] [Google Scholar]
- 24.Hayashi T., Boyko E.J., McNeely M.J., Leonetti D.L., Kahn S.E., Fujimoto W.Y. Visceral adiposity, not abdominal subcutaneous fat area, is associated with an increase in future insulin resistance in Japanese Americans. Diabetes. 2008;57:1269–1275. doi: 10.2337/db07-1378. [DOI] [PubMed] [Google Scholar]
- 25.Petersen A., Bressem K., Albrecht J., Thieß H.M., Vahldiek J., Hamm B., et al. The role of visceral adiposity in the severity of COVID-19: highlights from a unicenter cross-sectional pilot study in Germany. Metabolism. 2020;110:154317. doi: 10.1016/j.metabol.2020.154317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Favre G., Legueult K., Pradier C., Raffaelli C., Ichai C., Iannelli A., et al. Visceral fat is associated to the severity of COVID-19. Metabolism. 2021;115:154440. doi: 10.1016/j.metabol.2020.154440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Iacobini C., Pugliese G., Fantauzzi C.B., Federici M., Menini S. Metabolically healthy versus metabolically unhealthy obesity. Metabolism. 2019;92:51–60. doi: 10.1016/j.metabol.2018.11.009. [DOI] [PubMed] [Google Scholar]
- 28.Lee S.W., Youm Y., Lee W.J., Choi W., Chu S.H., Park Y.R., et al. Appendicular skeletal muscle mass and insulin resistance in an elderly Korean population: the Korean social life, health and aging project-health examination cohort. Diabetes Metab J. 2015;39:37–45. doi: 10.4093/dmj.2015.39.1.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Okazaki T., Ebihara S., Mori T., Izumi S., Ebihara T. Association between sarcopenia and pneumonia in older people. Geriatr Gerontol Int. 2020;20:7–13. doi: 10.1111/ggi.13839. [DOI] [PubMed] [Google Scholar]
- 30.Tartof S.Y., Qian L., Hong V., Wei R., Nadjafi R.F., Fischer H., et al. Obesity and mortality among patients diagnosed with COVID-19: results from an integrated health care organization. Ann Intern Med. 2020;173:773–781. doi: 10.7326/M20-3742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wu X., Li C., Chen S., Zhang X., Wang F., Shi T., et al. Association of body mass index with severity and mortality of COVID-19 pneumonia: a two-center, retrospective cohort study from Wuhan, China. Aging (Albany NY) 2021;13:7767–7780. doi: 10.18632/aging.202813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Petrilli C.M., Jones S.A., Yang J., Rajagopalan H., O’Donnell L., Chernyak Y., et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. doi: 10.1136/bmj.m1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Khunti K., Knighton P., Zaccardi F., Bakhai C., Barron E., Holman N., et al. Prescription of glucose-lowering therapies and risk of COVID-19 mortality in people with type 2 diabetes: a nationwide observational study in England. Lancet Diabetes Endocrinol. 2021;9:293–303. doi: 10.1016/S2213-8587(21)00050-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yang W., Sun X., Zhang J., Zhang K. The effect of metformin on mortality and severity in COVID-19 patients with diabetes mellitus. Diabetes Res Clin Pract. 2021;178:108977. doi: 10.1016/j.diabres.2021.108977. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material