Abstract
Cellular senescence is a persistently hypoproliferative state with diverse stressors in a specific aging microenvironment. Senescent cells have a double‐edged sword effect: they can be physiologically beneficial for tissue repair, organ growth, and body homeostasis, and they can be pathologically harmful in age‐related diseases. Among the hallmarks of senescence, the SASP, especially SASP‐related extracellular vesicle (EV) signalling, plays the leading role in aging transmission via paracrine and endocrine mechanisms. EVs are successful in intercellular and interorgan communication in the aging microenvironment and age‐related diseases. They have detrimental effects on downstream targets at the levels of immunity, inflammation, gene expression, and metabolism. Furthermore, EVs obtained from different donors are also promising materials and tools for antiaging treatments and are used for regeneration and rejuvenation in cell‐free systems. Here, we describe the characteristics of cellular senescence and the aging microenvironment, concentrating on the production and function of EVs in age‐related diseases, and provide new ideas for antiaging therapy with EVs.
Keywords: age‐related diseases, aging microenvironment, antiaging therapy, cellular senescence, extracellular vesicles
1. INTRODUCTION
Cellular senescence is a physiologically stable state of cell cycle arrest that strictly regulates cell proliferation and lifespan. The discovery of this phenomenon can be traced to the 1960s. Hayflick and Moorhead cultivated human fibroblasts and observed that they underwent a limited number of cell divisions, which is now called the “Hayflick limitation” (Hayflick & Moorhead, 1961). Cellular senescence is a transitional state of cell death that precisely regulates survival and death at the cellular level. The expected cellular senescence trajectory is cell elimination, which plays an essential role in promoting tissue repair and regulating organ growth and body homeostasis (Mosteiro et al., 2016; Ritschka et al., 2017). However, in senescence‐associated diseases, excessive accumulation of senescent cells during aging becomes burdensome, disrupting the trajectory of programmed cell death and leading to a decline in regeneration (Masaldan et al., 2018). These findings indicate that short‐term accumulation of senescent cells in tissues is beneficial to the renewal of the internal environment and the maintenance of organs but that long‐term accumulation of senescent cells in tissues triggers an imbalance in the internal environment and organ disability.
HIGHLIGHTS
The aging microenvironment can be unstable with aging and transform into a pathological state.
The SASP, with the involvement of EVs, amplifies senescent signals in autocrine, paracrine, and endocrine ways.
Senescent EVs alter the levels of immunity, inflammation, gene expression and metabolism of target cells.
Therapeutic strategies incorporating EVs from various materials and donors are feasible for age‐related diseases.
This double‐edged sword effect of cellular senescence is specifically attributed to the senescence‐associated secretory phenotype (SASP), which can be beneficial by inducing the recruitment of immune cells against stressors and can be detrimental by inducing the transmission of aging signals to cause inflammation (Ovadya et al., 2018). Among these secretory factors, extracellular vesicles (EVs), diverse small vesicles with lipid bilayer membranes, are released by most cell types and have been detected in virtually all body fluids (Saugstad et al., 2017). Their omnidirectional communication among cells and organs indicates their tight association with the transmission of aging signals in the context of age‐related diseases. Transmission through EVs can induce immune and inflammatory activation, genomic instability, telomere attrition, epigenetic alterations, mitochondrial and lysosomal system dysfunction, ROS accumulation, loss of proteostasis and nutrition, and stem cell exhaustion in target cells. Their potential for clinical application suggests that they can be used therapeutically to extend lifespan. In this review, we focus on the role of EVs in the aging microenvironment and age‐related diseases and the value of EVs as biomarkers and treatments of pathological aging states.
2. EXTRACELLULAR VESICLES IN THE CELLULAR SENESCENCE AND AGING MICROENVIRONMENT
2.1. Cellular senescence and the aging microenvironment
From a longitudinal perspective, single‐cell aging can be considered a fate‐decision process according to Li's single‐cell analysis of yeast performed with microfluidics and time‐lapse microscopy. Although cells carrying identical genetic materials were cultured in uniform medium, they embarked on two distinct paths of aging, bifurcating early: one pathway led to rDNA silencing and nucleolar decline, and the other led to heme abundance and mitochondrial aggregation (Li et al., 2020b). The findings pinpointed two essential events of cellular senescence: changes in nuclear genes and the transformation of mitochondria (Figure 1).
FIGURE 1.
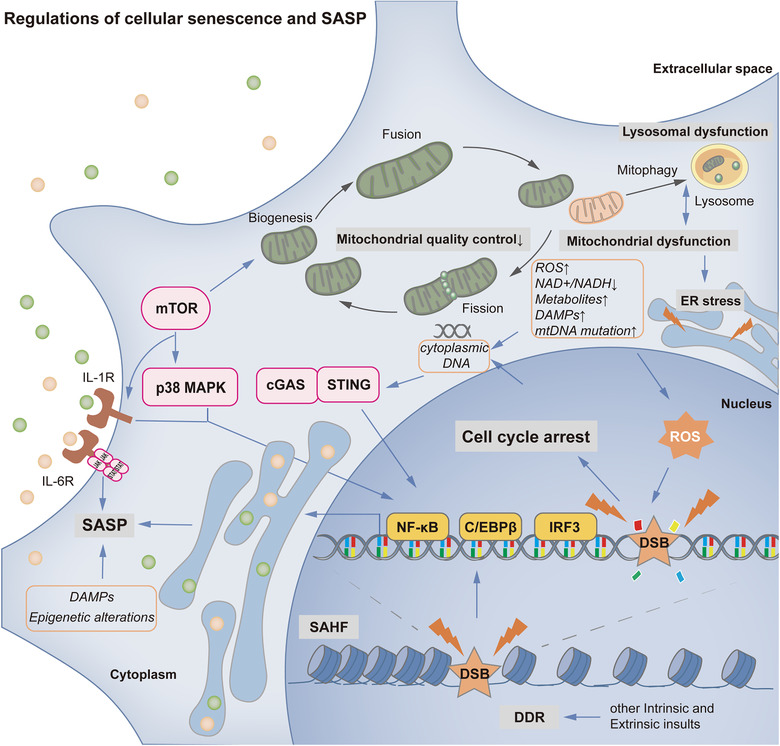
Regulation of cellular senescence and SASP. In cellular senescence, there are two essential events: changes in nuclear genes (associated with DDR, SAHF and other epigenetic alterations) and the transformation of mitochondria (unbalanced mitochondrial quality control and mitochondrial dysfunction). Mitochondrial changes, which may be induced by lysosomal dysfunction and mTOR signaling, can result in elevated ROS levels, reduced NAD+/NADH ratios, increased abundance of metabolites, release of DAMPs, mutation of mtDNA, lysosomal dysfunction and ER stress. The accumulation of ROS can trigger and accelerate DNA damage and cell cycle arrest. In addition to ROS, many intrinsic and extrinsic insults can also trigger DDR and cell cycle arrest. In the SASP process, several signaling pathways (including mTOR, p38 MAPK, cGAS/STING, DDR/PIKK superfamily, epigenetic alteration, and DAMP pathways) are involved. Downstream transcription factors, such as NF‐κB, C/EBPβ and IRF3, are consistently recruited to regulate SASP‐related gene expression. Functionally, the SASP can transmit aging signals in autocrine, paracrine, and endocrine ways. Hallmarks of senescence are shown in grey. Controlling productions or events in aging are shown in orange. Important signaling pathways are shown in pink. Essential transcription factors are shown in yellow. Receptors are shown in brown
As mitochondria are crucial double‐membrane organelles in the progression of the tricarboxylic acid (TCA) cycle and electron transport chain, the production of ATP, the synthesis and breakdown of lipids and the regulation of apoptosis, they are regarded as the key drivers and regulators of senescence (Habiballa et al., 2019). They undergo biogenesis, dynamic fusion and fission, and mitophagy to ensure structural and functional homeostasis in response to stimuli and stresses, and disruption of any of these steps can cause mitochondrial dysfunction and detrimental oxidative metabolism leading to cellular senescence (Picca et al., 2018). The role of mitochondrial biogenesis in aging has been controversial. Mostly, pathologically senescent cells have been shown to exhibit increased mitochondrial mass with increasing oxygen consumption and ROS production by transcriptional coactivator PGC‐1, which is a regulator of mitochondrial biogenesis (Correia‐Melo et al., 2016). However, senescent CD4+ T cells display a higher mitochondrial content and biogenesis than CD8+ T cells, while retaining better glucose and lipid uptake, nutrient usage, and proliferative and migratory capacity (Callender et al., 2020). The significance of mitochondrial dynamics has been shown in a Pgam5 deficiency mouse model in vivo and PGAM5–/– cells in vitro (PGAM5 is a mitochondrial Ser/Thr phosphatase that dephosphorylates Drp1 and promotes mitochondrial fission). In these models, reduced mitochondrial fission increased ATP and ROS levels, elevated signaling via mTOR and IFN pathways and resulted in an age‐related reduction in antioxidation capability (Yu et al., 2020). Like mitochondrial fission and fusion, mitophagy is a selective process of mitochondrial quality control; however, mitophagy determines dysfunctional mitochondrial degradation by lysosomes. In yeast, defects in lysosome‐like vacuoles induce intracellular cysteine toxicity, collectively hampering iron bioavailability and mitochondrial respiration via a ROS‐based mechanism (Hughes et al., 2020). Altogether, these data indicate that impaired mitochondrial homeostasis may result in elevated ROS levels and senescence. In addition to elevated ROS levels, senescence‐associated mitochondrial metabolism can be characterized by reduced NAD+/NADH ratios, abundance of metabolites, release of mitochondrial damage‐associated molecular patterns (DAMPs) and mutation of mtDNA (Habiballa et al., 2019).
Accordingly, several studies have indicated that the accumulation of ROS is not a simple consequence of cellular senescence but also a trigger and accelerator of DNA damage and aging (Bai et al., 2020; Jia et al., 2014; Qian et al., 2019; Yin et al., 2013). The presence of DNA damage activates a robust response named the DNA damage response (DDR) (Calcinotto et al., 2019). Common DDR outcomes realized from intrinsic insults include telomere shortening (Azarm et al., 2020), oncogene mutation (Hafez et al., 2017), and oxidative damage (Oh et al., 2017). Various DNA‐damaging agents are also effectively applied to induce different types of senescence, for example, chemotherapy‐induced senescence (e.g., that induced by doxorubicin (Yao et al., 2020) or etoposide (Zurgil et al., 2014)). The DDR activates the ATM/ATR pathway to block cell cycle progression via stabilization of the p53 protein (Stewart‐Ornstein & Lahav, 2017) or alternative splicing of TP53 mRNA (Chen et al., 2017), which then stimulates cyclin‐dependent kinase inhibitor p21WAF1/CIP1 transcription (Thurn et al., 2013). The p21 signal is essential at the onset of cell cycle arrest, while it is replaced by the subsequently increasing p16 level, both of which can prevent Rb phosphorylation to slow cell division in aging (Figure 2) (Prasnikar et al., 2021). Moreover, the retrograde signal between mitochondria and the nucleus impacts nuclear gene expression in the direction of senescence. In an aged mouse model, the RNA component of telomerase TERC was found to be imported into mitochondria and then exported to the cytosol as a short form, TERC‐53, which reveals mitochondrial function and induces cognitive decline by changing nuclear gene expression (Zheng et al., 2019). Most genes can also display changes in the epigenome and chromatin organization, typically at senescence‐associated heterochromatin foci (SAHF), to drive aging. These changes can be triggered by disruption of mitochondrial function, glucose metabolism and autophagy (Di Micco et al., 2021; Zhang et al., 2021).
FIGURE 2.
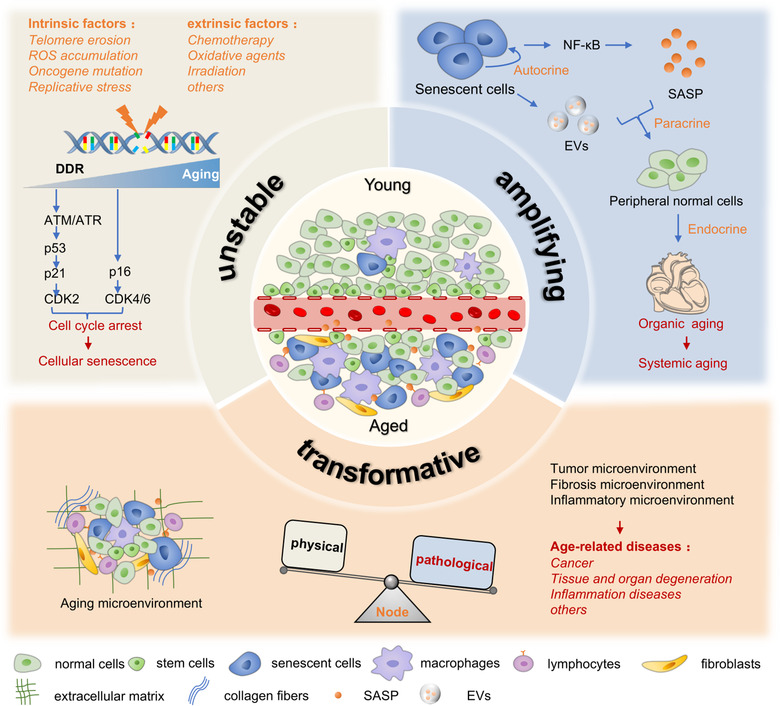
Three characteristics of the aging microenvironment. The aging microenvironment contains fewer proliferative stem cells and more senescent functional cells and immunosuppressive cells with age. It is increasingly unstable because of intrinsic and extrinsic stressors, leading to the DNA damage response and cell cycle arrest. The SASP, which includes EVs, can amplify senescent signals from accumulating senescent cells to peripheral normal cells or distant organs via autocrine, paracrine and endocrine pathways. The aging microenvironment can be transformative and reprogrammed through specific nodes. The physical microenvironment can be transformed into pathological tumours, fibrosis and inflammatory microenvironments, inducing age‐related diseases, such as cancer, degenerative diseases and inflammation
However, in a special type of senescence, PTEN loss‐induced senescence (PICS), mammalian target of rapamycin complexes 1 and 2 (mTORC1 and mTORC2) can directly stabilize p53 and initiate the p21‐associated senescent pathway through PI3K/AKT activation and MDM2 inhibition without triggering the DDR (Jung et al., 2019). The mTOR signaling pathway also controls mitochondrial activities to regulate energy metabolism and oxidative phosphorylation progress. It has been documented that mTORC1 can promote nuclear‐encoded mitochondria‐related gene expression through inhibition of eukaryotic translation initiation factor 4E (elF4E)‐binding proteins (4E‐BPs) or interaction with PCG‐1α, consequently increasing mitochondrial biogenesis (de la Cruz Lopez et al., 2019; Morita et al., 2013). Simultaneously, mTORC1 can selectively stimulate the translation of mitochondrial fission process 1 (MTFP1) via 4E‐BPs, recruit the fission GTPase dynamin‐related protein (DRP1) and govern mitochondrial dynamics (Morita et al., 2017). In multiform senescence, mTORC1‐related mitochondrial biogenesis can activate the ROS‐induced DDR through the ATM/Akt/mTORC1 phosphorylation cascade and then upregulate PCG‐1β, which has impacts on senescent deterioration and SASP persistence in vitro and in vivo (Correia‐Melo et al., 2016). In addition, mTOR signaling in aging also interferes with protein metabolism and blocks autophagy and regeneration to accelerate cellular and tissue senescence (Liu & Sabatini, 2020).
With mitochondrial dysfunction, elevated ROS levels can also induce endoplasmic reticulum (ER) stress via unfolded protein response (UPR) signaling, in which FGF21 as the culprit systemically triggers premature senescence (Tezze et al., 2017). Whereas mitochondria suffer mitophagy and dysfunction, lysosomes undergo deterioration with the accumulation of lipofuscins (Park et al., 2018). In this vicious feedback cycle, autophagic flux also consequently shows impaired proteolytic ability, and this impairment is responsible for chronic senescence (Tai et al., 2017). Thus, due to the mechanistic complexity and intersection of the causes and types of single‐cell senescence, the currently known senescent hallmarks do not have specificity.
At eye level, senescence does not merely affect a single group of cells. Senescence is always the consequence of the coaction of different cells, cytokines, extracellular matrix components, and other elements. This combination is regarded as the aging microenvironment and is mainly composed of proliferating cells, functional and metabolic cells, and surveillance and elimination factors (Fane & Weeraratna, 2020). Here, we conclude three characteristics of the aging microenvironment (Figure 2). First, it grows increasingly unstable with age. The body confronts various risk factors over time, which can be external or internal, genetic or acquired, and with sudden and powerful effects or cumulative and weak effects. When systems are disturbed and illness strikes, stem cell exhaustion, metabolic dysfunction, byproduct accumulation, and an imbalance of the microenvironment can emerge, ultimately leading to cancer or other age‐related diseases. Fane and Weeraratna indicated that compared to the young, healthy microenvironment, the aging microenvironment demonstrates accumulation of SASP factors and immunosuppressive cells, such as regulatory T cells and myeloid‐derived suppressor cells, which accelerate the instability of the aging microenvironment and induce tumour‐permissive conditions (Fane & Weeraratna, 2020). A metabolic study on elderly individuals has also confirmed this process. In the serum, the levels of methylmalonic acid, a byproduct of propionate metabolism, increase with age. This metabolic alteration changes the aging microenvironment and favours the epithelial‐mesenchymal transition in lung and breast cancer through the TGF‐β and SOX4 pathways (Gomes et al., 2020).
Second, the aging microenvironment has an amplifying effect. Senescent cells can transmit aging signals via the SASP involving extracellular vesicles to magnify aging signals among cells and organs (Al Suraih et al., 2020). In ischemic retinal models, senescent retinal ganglion cells, microglia and endothelial cells can release many SASP factors, such as TGF‐β, IL‐1, IL‐6 and VEGF, to reinforce their senescence in autocrine ways and propagate senescent alterations via the paracrine pathway in the whole retina, contributing to pathological retinal angiogenesis (Oubaha et al., 2016).
Finally, the aging microenvironment can be transformative and reprogrammed. With the above characteristics, the aging microenvironment can be easily transformed into a pathological microenvironment. For example, markers in the aging microenvironment can also be detected in the tumour microenvironment (Han et al., 2020). There are even specific controllable nodes in this transformation. Chen et al. confirmed that neuropilin‐1 (NRP1) and HIF2α were highly expressed in aging lungs and significantly downregulated endothelial cell protein C receptor (EPCR), a regulator of the circulating microenvironment. Inhibition of the EPCR pathway had important effects on a variety of functions from regeneration to fibrosis in the context of the circulating aging microenvironment (Chen et al., 2020). Selectively inhibiting neuropilin‐1 (NRP1) and HIF2α in vascular endothelial cells (ECs) significantly attenuated the fibrosis process, indicating that this research area is fertile ground for targeted therapy development.
These findings show that organ aging in the aging microenvironment is multidirectional and multimembered. Systemic aging is characterized by multiformity associated with alterations and combinations of various molecules, cells, and organs. Based on this theory, individual ageotypes (depicted and formulated as different types of aging patterns in diverse populations) will provide molecular assessment data (including genes, plasma proteins, metabolites, cytokines, and microbes) that precisely reflect personal aging levels (Ahadi et al., 2020).
2.2. SASP and cellular senescence
Among ageotypes, various soluble molecules marking the presence of senescent cells, which together are termed the SASP, have attracted our attention because of their heterogeneity and availability for clinical application. High levels of immune modulators, proinflammatory cytokines, growth factors, chemokines, and proteases are involved in the SASP in a cell non‐autonomous manner (Calcinotto et al., 2019). Proteomic analysis results obtained from human fibroblasts, renal epithelial cells, bone marrow, and adipose mesenchymal stromal cells have suggested that the SASP can be categorized into different groups depending on the senescence inducers and cell derivations (Basisty et al., 2020; Ozcan et al., 2016). In contrast to genotoxic and oncogenic senescence, the work by Wiley et al. presented a distinct senescent phenotype of mitochondrial dysfunction in progeroid mice with POLGD257A mitochondrial DNA (mtDNA) mutation, termed mitochondrial dysfunction‐associated senescence (MiDAS), which had a specific secretion profile without IL‐1‐dependent factors through an NADH/AMPK/p53‐dependent pathway (Wiley et al., 2016). Additionally, the SASP Atlas website (www.SASPAtlas.com), constructed by Basisty's team, can aid in a more comprehensive understanding of the complex and dynamic SASP network (Basisty et al., 2020).
Paradoxically, the SASP can be beneficial to organismal homeostasis or detrimental in different biological contexts (Oubaha et al., 2016; Watanabe et al., 2017). When primary mouse keratinocytes are transiently subjected to SASP conditions, the levels of stem cell markers and regenerative capacity increase in vivo. In contrast, under prolonged exposure, cell plasticity and stemness are attenuated to some extent (Ritschka et al., 2017). Therefore, it can be assumed that the roles of the SASP can autonomously switch depending on the concentration and duration of treatment. Moreover, high levels of SASP proteins are positively correlated with the age and physical conditions of patients. Among these SASP factors, the levels of 7 SASP factors (GDF15, FAS, OPN, TNFR1, ACTIVIN A, CCL3 and IL‐15) may predict the postsurgery outcomes of patients with severe aortic stenosis and ovarian cancer (Schafer et al., 2020).
When the SASP is stimulated, several signaling pathways are activated, and two downstream transcription factors, NF‐κB and C/EBPβ, are consistently recruited to regulate SASP‐related gene expression (Figure 1 and Table 1). With regard to DNA damage‐induced senescence, mounting evidence has indicated that the phosphatidylinositol 3‐kinase‐related kinase (PIKK) superfamily (ATM, ATR and DNA‐PKcs protein kinases included) is crucial for the onset of senescence and transcription of SASP genes upstream of NF‐κB signaling, responding to the DDR (Strzeszewska et al., 2018). Pharmacologic inhibition and genetic depletion of ATM can suppress NF‐κB activation, diminish inflammatory cytokine levels, and reverse senescence (Zhao et al., 2020). ATM and ATR also inhibit p62‐dependent autophagic degradation to stabilize the GATA4 protein, which evokes the NF‐κB signaling pathway, thereby contributing to the SASP (Kang et al., 2015). In addition, the transcription‐associated methyltransferase and oncoprotein MLL1 also mobilizes the ATM‐NF‐κB axis to control SASP genes in the context of the DDR (Capell et al., 2016). With a prolonged DDR, the accumulated histone variant H2A.J colocalizes with 53BP1 to be incorporated into SAHF formation and competes with H1 to increase the expression of repeated DNA sequences while activating STAT/IRF transcription factors and inducing the interferon‐related SASP (Isermann et al., 2020; Mangelinck et al., 2020).
TABLE 1.
Signaling pathways that are associated with SASP‐related secretion
| Trigger | Signaling pathway | Transcription factor(s) | Secretory factors | Ref. |
|---|---|---|---|---|
| DNA damage response | ||||
| H2A.J accumulation | Incorporation into SAHFs; H1 decrease | STAT, IRF | IL‐6, IL‐8, CXCL8, CSF2, CM‐CSF, MCP1, CCL2, IFN | (Isermann et al., 2020); (Mangelinck et al., 2020) |
| MLL1 increase | ATM | NF‐κB | IL‐8, IL‐1B | (Capell et al., 2016) |
| Autophagy decrease | GATA4 | NF‐κB | IL‐6, IL‐8, CXCL1/2/3, CM‐CSF, ICAM1, CCL2, TNF | (Kang et al., 2015) |
| Mitochondrial dysfunction | ||||
| NAD+/NADH ratio decrease | NADH/AMPK/p53 | / | IL‐10, TNFα, CCL27 | (Wiley et al., 2016) |
| ROS and CCF increases | cGAS/STING | NF‐κB, IRF3 | IL‐6, CXCL10 | (Vizioli et al., 2020) |
| Cell‐free mtDNA | cGAS/STING; TLR9‐DC activation | IRF3; T‐bet, RORγt | IFN; IL‐17, IFN‐γ | (Aarreberg et al., 2019); (Iske et al., 2020) |
| Chromatin remodeling | ||||
| LBR decrease | CCFs/cGAS/STING | / | IL‐6, IL‐8, MMP1 | (En et al., 2020) |
| LINE1 transcription increase | cGAS/STING | NF‐κB, IRF3 | IFN, IL‐6, IL‐8, IL‐1, MMP3, ISG, MCP1 | (Bi et al., 2020) |
| HDAC inhibition | ATM/MRN | NF‐κB | IL‐6, IL‐8, CM‐CSF, IL‐1 | (Malaquin et al., 2020) |
| BRD4 increase | Acetylation of superenhancers of the SASP | / | IL‐6, IL‐8, CM‐CSF, IL‐1, CXCL1, G‐CSF | (Tasdemir et al., 2016) |
| Other triggers | ||||
| Oncogenic Ras increase | MAPK/PEA3/SCCA; P38 MAPK | NF‐κB | IL‐6, IL‐8, CXCL1, G‐CSF, CM‐CSF | (Catanzaro et al., 2014); (Freund et al., 2011) |
| HMGA protein increase | NAMPT/NAD+/p38 MAPK | NF‐κB | IL‐6, IL‐8, IL‐1B | (Nacarelli et al., 2019) |
| HMGB1 protein increase | TLR4 signaling | NF‐κB | TNFα, IL‐6, IL‐8, | (Davalos et al., 2013) |
| Inflammasome increase | IL‐1 signaling | / | IL‐1, IL‐6, IL‐8, CCL2 | (Acosta et al., 2013) |
| PTBP1 increase | Alternative splicing of EXOC7 | / | IL‐6, IL‐8 | (Georgilis et al., 2018) |
| S100A14 and Cu2+ increases | IL‐1 signaling | NF‐κB | IL‐6, IL‐8, IL‐1, MMP3, CXCL1/2, CCL2 | (Su et al., 2019b) |
| mTOR increase | IL‐1 signaling | NF‐κB | IL‐6, IL‐8, IL‐1, CXCL1/2, CCL2 | (Laberge et al., 2015) |
| NOTCH1 decrease | NOTCH | C/EBPβ | IL‐6, IL‐8, IL‐1 | (Ito et al., 2017) |
| DNase2 and TREX1 decrease | CCFs/cGAS/STING | NF‐κB, IRF3 | IL‐6, IL‐8, IL‐1, CXCL10, IFN | (Takahashi et al., 2018) |
Epigenetically, bromodomain‐containing protein 4 (BRD4), an acetylated histone‐binding protein, is directly recruited to superenhancers adjacent to SASP genes, and myriad cytokines are released (Tasdemir et al., 2016). Nevertheless, histone deacetylase inhibitors (HDACis) are required for the recruitment of ATM and MRN complexes, and NF‐κB binds to chromatin to modulate the expression of SASP genes, which is independent of DDR (Malaquin et al., 2020). Regarding the sirtuin family, a group of histone deacetylases, their declining expression with age determines the detachment of nuclear lamina proteins and the loss of heterochromatin proteins, such as SIRT7. SIRT7 downregulation promotes the transcription and accumulation of the LINE1 retrotransposon (LINE1), which triggers the innate immune response through the cGAS‐STING signaling pathway (Bi et al., 2020). Besides the decrease in SIRT7, the decrease in the laminar protein lamin B receptor (LBR) also causes dysregulation of heterochromatin and generation of cytoplasmic chromatin fragments (CCFs) to activate the cGAS‐STING pathway in senescent cells (En et al., 2020).
Thus, the cyclic GMP‐AMP synthase (cGAS)‐stimulator of interferon genes (STING) pathway is critical for SASP modulation in response to the accumulation of cytoplasmic DNA (CCFs, cDNA and mtDNA). Diverse triggers of senescence (such as oxidative stress, oncogene mutation, irradiation, and pro‐senescent drug treatment) can cause extrusion of cytoplasmic DNA and activation of the innate immune sensing cGAS‐STING pathway, which next activates TBK1 and IκBα to promote IRF3 and NF‐κB expression (Gluck et al., 2017; Loo et al., 2020). Moreover, ROS generated by mitochondrial dysfunction can promote linking of the kinase JNK with 52BP1 (a nuclear protein that impedes DNA double‐strand break end resection and CCF formation) for incorporation during CCF formation. This mechanism constitutes another mitochondria‐to‐nucleus retrograde signaling pathway that can activate the cGAS‐STING pathway and develop the SASP (Vizioli et al., 2020). In senescent cells, downregulation of DNases (for example, DNase2 and TREX1) in the cytoplasm can also result in the accumulation of cytoplasmic DNA to aberrantly activate the cGAS‐STING pathway (Takahashi et al., 2018).
Among the origins of cytoplasmic DNA are mammalian mitochondria, in which cytoplasmic DNA is created through the coordinated regulation of nuclear and mitochondrial genes. The mutation rate and progeronic potency of mtDNA are significantly higher than those of nuclear DNA, mostly owing to the ROS‐rich mitochondrial microenvironment, the poor fidelity of mitochondrial DNA polymerase and the electron transport chain (ETC)‐encoded ability (Li et al., 2019). Although only 13 of the 1500 proteins constituting mammalian mitochondria are encoded by mtDNA, they are all important components of the ETC that regulate oxidative phosphorylation (Taanman, 1999). Mutations in mtDNA with aging are positively linked with mitochondrial dysfunction, defects in ATP synthesis, and the exaggeration of ROS‐induced oxidative stress (Niemann et al., 2017). It has also been proven that the declining mass of mtDNA in Transcription Factor A Mitochondrial (TFAM) knockout mice can induce systemic cytokine storms and multiorgan senescence featuring including muscular dystrophy, cardiovascular alterations and neurological disability through TNFα (Desdin‐Mico et al., 2020). In the aging microenvironment, cell‐free mtDNA acting as a DAMP or localized in EVs is released by senescent cells and can be recognized through the cGAS‐STING pathway or pattern recognition receptors (PRRs), such as TRL9, to activate immune responses and increase the release of systemic inflammatory factors (Iske et al., 2020; Schwartz et al., 2021). In addition to mtDNA, DAMPs are dynamically generated, translocated and released into the extracellular milieu to increase senescence‐related stressors, especially ROS, driving the initiation and development of aging and the SASP. With aging, the declining levels of intracellular DAMPs cause genomic instability, metabolic disorder, and chromatin modification, whereas increased levels of extracellular DAMPs can elicit immune and inflammatory responses via PRR recognition (Huang et al., 2015). For example, the nuclear high‐mobility group box 1 (HMGB1) protein regulates DNA replication, transcription and repair, the loss of which can cause DNA damage and genome instability in the aging state (Jia et al., 2020). In contrast, extracellular HMGB1 is associated with TLR4‐induced senescence‐associated inflammation, which activates NF‐κB signaling pathways and IL‐6, IL‐1 and TNFα secretion (Davalos et al., 2013).
It has also been reported that senescent CD8+ T cells display the SASP but that it is governed by p38 MAPK signaling (Callender et al., 2018). This signaling pathway is dispensable along with the canonical DDR, and it can be directly mediated by mitochondrial dysfunction with decreased NAD+/NADH ratios. After activation of p38 MAPK, the levels of p53 and NF‐κB also increase and then restrain and enhance the SASP, respectively (Freund et al., 2011; Wiley et al., 2016). In addition, the p38 MAPK signaling pathway can exert profound effects on oncogene‐induced senescence models to increase the levels of secretory factors, an effect that is driven by high‐mobility group A (HMGA)‐mediated NAMPT expression and then NAD+‐dependent AMPK derepression to enhance NF‐κB activity (Nacarelli et al., 2019). Oncogenic Ras can also upregulate squamous cell carcinoma antigens 1 and 2 (SCCA1/2) through MAPK and the ETS transcription factor PEA3 to mediate NF‐κB activation and inflammatory cytokine production (Catanzaro et al., 2014).
Furthermore, IL‐1α and its receptors also reinforce NF‐κB activity in an mTOR‐dependent or S100A13‐Cu2+‐dependent manner, even from the inflammasome, which accelerate the migration of IL‐1α to the cell membrane and the binding of IL‐1α with IL‐1R in a juxtacrine fashion (Acosta et al., 2013; Ito et al., 2017; Laberge et al., 2015; Su et al., 2019b).
Regarding C/EBPβ, Notch signaling can regulate its temporal expression by regulating the level of TGF‐β between an immunosuppressive profibrotic secretory state and a proinflammatory fibrolytic secretory state (Ito et al., 2017). In a RASG12V‐induced senescence model, C/EBPβ can be rescued from 3′UTR regulation of protein activity (UPA) disruption and amplified by AMPK activation, leading to the SASP (Salotti & Johnson, 2019).
Accordingly, the SASP involves the release of a plethora of molecules to modify the aging microenvironment in a spatial and temporal manner via numerous transcription factors and signaling pathways. Expanding the SASP database and later improving the accuracy of SASP targeting through clinical trials may enable giant leaps to be made in aging research.
2.3. Diversity of extracellular vesicles in cellular senescence
Initially, EVs were considered fragments and discarded units of dead and degenerated cells, but it was subsequently hypothesized that EVs are inevitably released during physiological and pathological processes (Misawa et al., 2020). EV classification is based on size, density, purification methods, formation progress, surface markers, and even release mechanisms. Although classification is still challenging, the International Society for Extracellular Vesicles (ISEV) holds that, based on the mechanisms of EV biogenesis, two identifiable subtypes of EVs have been largely confirmed: one from the plasma membrane (i.e., microvesicles, ectosomes, microparticles) and another from the endosomal system/multivesicular bodies (MVBs) (exosomes, which are vital) (Russell et al., 2019).
Vesicles directly formed by plasma membrane budding usually range from 100 to 1,000 nm in diameter (Moller & Lobb, 2020). They are rich in miRNAs, especially after oxidative stress, which enables them to be sorted into microvesicles by the hnRNPA2B1/caveolae‐1 complex upon separate O‐GlcNAcylation and phosphorylation (Tkach & Thery, 2016).
Exosomes are known for their nanoscale size and versatility, notably in cancer. Their biogenesis begins with endocytosis and invagination of the plasma membrane to form early‐sorting endosomes (ESEs). After interaction with organelles and maturation into late‐sorting endosomes (LSEs), they ultimately assemble multivesicular endosomes (MVEs). Intraluminal vesicles (ILVs) are then produced by secondary invagination of the endosomal limiting membrane in MVEs, a process mediated by the endosomal sorting complex required for transport (ESCRT) complex, sphingomyelinase, ceramide, RAB family members, and KIBRA (or their combinations) and are eventually released as exosomes. MVEs can merge with either lysosomes or autophagosomes to be degraded (Figure 3) (Kalluri & LeBleu, 2020; Moller & Lobb, 2020; Russell et al., 2019; Song et al., 2019; Wei et al., 2020a). Importantly, in either senescent or nonsenescent cells, inhibition of exosomal biogenesis through silencing of Alix or Rab27a, two key factors of EV production in the ESCRT‐dependent pathway, can provoke ROS‐induced DDR, cell cycle arrest and apoptosis owing to cytoplasmic DNA accumulation (Takahashi et al., 2017). This indicates that EV secretion pathways are essential to maintain cellular homeostasis.
FIGURE 3.

Mechanisms of EVs in the aging microenvironment. Senescence‐associated EVs are produced from the plasma membrane and the endosomal system, and their levels can be increased due to DNA damage, mitochondrial damage and lipid peroxidation with age. These EVs contain typical markers (tetraspanins, ESCRT proteins, MHC, etc.) and specific proteins, glycoproteins, nucleic acids, and lipids (such as ceramide and sphingomyelin). They carry destructive signals and shuttle from senescent cells to target cells, which causes aging transmission. Target cells internalize these EVs through receptor‐ligand combinations or endocytosis. Senescence‐associated EVs can alter and induce senescence in target cells through immune and inflammatory activation, genomic instability, telomere attrition, epigenetic alterations, mitochondrial and lysosomal system dysfunction, ROS accumulation, loss of proteostasis and nutrition, and stem cell exhaustion. Here, we only present the route of exosome production. Early‐sorting endosomes, ESEs; late‐sorting endosomes, LSEs; multivesicular endosomes, MVEs; and intraluminal vesicles, ILVs
Secreted EVs interact with or are internalized by recipient cells via classic receptor‐ligand combination or endocytosis, including caveolae‐dependent, clathrin‐dependent and lipid raft‐mediated endocytosis; receptor‐mediated endocytosis; macropinocytosis; and phagocytosis (Feng et al., 2010; Gonda et al., 2019; Lia et al., 2020; Nanbo et al., 2013; Svensson et al., 2013; Tian et al., 2014). Specifically, oxidative stress‐generated EVs can rapidly reduce transepithelial resistance in recipient cells through neuraminidase activity to enable their internalization (Nicholson et al., 2020).
EVs, as constituents of the SASP, show significant changes in senescent cells (Figure 3). These changes are primarily stimulated by irreparable DNA damage with aging that promotes sphingomyelinase and ceramide activity and increases Golgi dispersal and endoplasmic reticulum dilation accompanied by increasing inflammation and decreasing autophagy (Goulielmaki et al., 2020; Hitomi et al., 2020; Takahashi et al., 2017). In the DDR process, the production of EVs still relies on functional p53 and p65. Inhibition of p53 has been verified to reduce the secretion of CD63+ EVs, which plays a pivotal role in senescence and apoptosis of adjacent cells (Tesei et al., 2021). It is possible that p53 activation enhances the expression of genes involved in endosome regulation and exosome generation during exposure to multiple stresses, such as CHMP4, caveolin‐1 and nSMase2, which are important components of ESCRT‐dependent and ceramide‐dependent signaling pathways (Buratta et al., 2017; Yu et al., 2009). Inhibition of p65 also prevents age‐induced inflammatory message transmission through EVs among mesenchymal stem cells, which reflects the importance of NF‐κB in the SASP (Mato‐Basalo et al., 2021).
EVs purified from PD donors are packed with low levels of mitochondrial markers (i.e., ATP5A, NDUFS3, and SDHB) related to systemic inflammation (i.e., CRP, MIP‐1β, and TNF‐α) (Picca et al., 2020). The existence of these mitochondrial markers as cargos suggest that mitochondrial dysfunction and impaired mitochondrial quality control (MQC) exist in PD in the presence of oxidative stress. The MQC system depends on the mitochondrial‐lysosomal axis to achieve fusion and fission, which can dilute damaged mitochondria or clear them through mitophagy. If the system is compromised or overwhelmed, these damaged mitochondria enter the endolysosomal system to produce EVs with oxidized components requiring PINK1 and Parkin factors (Picca et al., 2019). These oxidized components, including proteins, lipids, nucleic acids and even ROS, can induce an inflammatory state in targeted cells (Borras et al., 2020). Furthermore, under oxidative stress, ROS in senescent cells can regulate cargo sorting into EVs by increasing the phosphorylation of proteins such as the receptor tyrosine kinase ephrin‐A2 (EphA2) (Takasugi et al., 2017). Due to the vital role of mitochondrial dysfunction in aging, these mitochondrial components within EVs may become ideal molecular markers for individual ageotypes. However, in mitochondria‐free red blood cells (RBCs), aging EVs are released through membrane shedding by haemoglobin (Hb)‐generated ROS (Fibach, 2021). In addition, the lipid peroxidation product 4‐hydroxynonenal (HNE) can also elevate the production and internalization of EVs containing cytotoxic α‐synuclein to propagate pathology transneuronally in Parkinson's disease (Zhang et al., 2018b). Although several senescence‐associated pathways have been confirmed to participate in EV secretion, the detailed molecular mechanism by which EV biogenesis is regulated with aging remains largely unexplored.
The particle number and size of EVs have been tracked and compared under senescent and nonsenescent conditions in several experiments. However, the results have been controversial, possibly due to different cell derivations, senescence triggers, and scale ranges. Even though one report has supported categorization based on a concomitant increase in the number of relatively small EVs (Basisty et al., 2020; Chanda et al., 2019; Lei et al., 2017), another report has argued for categorization of EVs of similar number and size (Alberro et al., 2016; Fafian‐Labora et al., 2020a). Notably, the heterogeneity of EV cargos determines the diversity of EV functions in the aging microenvironment. EVs may contain typical markers (tetraspanins, ESCRT proteins, MHC, etc.) and specific proteins, glycoproteins, noncoding RNAs, and lipids (such as ceramide and sphingomyelin) during aging (Khayrullin et al., 2019; Pienimaeki‐Roemer et al., 2017; Sagini et al., 2018; Willis et al., 2020). The cargos can be expressed either ubiquitously or in a tissue‐specific manner and used as biomarkers to capture aging complexity. We describe the diverse cargos and multiple functions of EVs in the aging microenvironment and age‐related diseases in detail below.
3. MECHANISM OF AGING TRANSMISSION VIA EXTRACELLULAR VESICLES
With aging or senescence‐associated stress, the dynamic cargos of EVs reach their targets through paracrine or endocrine patterns and exhibit destructive or supportive behaviours that affect aging signal transmission. This transmission refers to the passing of information via cell‐to‐cell or organ‐to‐organ communication through EV trafficking (Figure 3). Borghesan et al. confirmed that IFITM3, a protein messenger linked with the IFN pathway, was selectively encapsulated in small EVs during oncogene‐induced senescence (OIS). IFITM3 is critical for early genotoxic damage and paracrine senescence in neighbouring fibroblasts (Borghesan et al., 2019). Moreover, shuttling of EVs between the liver and vasculature in a non‐alcoholic fatty liver disease (NAFLD) and cardiovascular disease (CVD) model has revealed the distant interorgan communication of EVs. Steatotic hepatocyte‐derived EVs containing miR‐1 suppress KLF4 expression but activate the NF‐κB pathway to facilitate endothelial inflammation and senescence and participate in the development of atherosclerosis (Jiang et al., 2020). In these ways, transmission through EVs from senescent cells has negative impacts on the aging microenvironment, promoting chronic low‐grade inflammation and cellular senescence in recipient cells and organs and progressively inducing senescence‐associated diseases.
EVs isolated from senescent cells can transform recipient cells’ immune state to mediate cellular viability through information delivery (Figure 3). When treated with oxidative agents, retinal pigment epithelial cells, a type of phagocytic cell, can produce more CD36‐associated microparticles, impeding phagocytosis and diminishing the viability of neighbourhood cells, as indicated by increased SA‐β‐gal staining and p21 and p15 expression (Yang et al., 2020). A cell‐free form of telomeric repeat‐containing RNA (cfTERRA) is abundant in EVs and is related to telomere dysfunction and DNA damage of parent cells. It is regarded as a telomere‐associated molecular pattern (TAMP) or DAMP and is detected in the blood plasma of control subjects and cancer patients, directly revealing that exosomal cfTERRA is a potential marker for telomere metabolism in senescence (Azzalin & Lingner, 2015; Cusanelli et al., 2013; Porro et al., 2014). It can also induce the transport of alarmin to elicit a recipient inflammatory response (Wang & Lieberman, 2016). Incubation of peripheral blood mononuclear cells with exosomes containing cfTERRA stimulates innate immune signaling and upregulates inflammatory cytokines, such as TNFα, IL‐6, and CXCL10 (Wang et al., 2015). In addition to cfTERRA, other exosomal DAMPs modulate innate immunity and cell conditions in the aging microenvironment. CD63+ EVs from irradiated cancer cells loaded with DNA:RNA hybrids and LINE 1 retrotransposons trigger the senescence of adjacent cells and induce innate immune signaling to polarize M1 macrophages (Tesei et al., 2021). EVs from senescent oral keratinocytes can carry mtDNA and nuclear DNA to activate the cGAS‐STING pathway and interferon expression in monocytes (Schwartz et al., 2021). Hence, these DAMPs in EVs consequently contribute to chronic inflammation and age‐related diseases through the innate immune signaling pathway, which can be considered a positive feedback loop of SASP modulation.
In genotoxic aging models with either irradiated breast cancer cells or irradiated mice, EVs can directly induce telomere attrition and DDR in recipients, causing genomic instability and aging. However, this effect is reduced by RNase treatment or thermal denaturation by boiling, which suggests that protein‐ and RNA‐containing EVs indeed have effects on telomere shortening and DNA damage (Al‐Mayah et al., 2017; Hargitai et al., 2021). In addition, EVs can mediate genomic instability through senescence‐associated genome‐wide hypomethylation to spread senescence characteristics to neighbouring cells. In a model consisting of replicative senescent human umbilical vein endothelial cells (HUVECs), EVs carried miR‐21‐5p, and miR‐217 downregulated DNA methyltransferase 1 (DNMT1) and Sirtuin 1 (SIRT1) expression in young cells, leading to high levels of SASP molecules and cell cycle inhibitors (Mensa et al., 2020). In nature, DNMT1 cooperates with SIRT1 (a well‐known longevity‐related gene) to maintain genetic methylation patterns in order to ensure genome integrity through mitosis (Lee et al., 2019; Zheng et al., 2015). Therefore, EVs transmit aging signals at the genetic and epigenetic levels (Figure 3).
Cargos and their corresponding EVs can co‐destroy energetic and proteinic networks to upset the intracellular balance of the targeted cells, promoting aging (Figure 3). In a model of recipient mitochondrial dysfunction, lung fibroblast‐derived EVs from idiopathic pulmonary fibrosis (IPF) patients carried elevated levels of miR‐23b‐3p and miR‐494‐3p to suppress SIRT3 expression. These EVs are critical for increasing mitochondrial ROS, causing damage, and inducing subsequent lung epithelial cell senescence, which verifies that EVs can destroy the energetic networks of target cells to induce senescence (Kadota et al., 2020). With aging, overload of the local lysosomal system can lead to a decrease in cellular degradation capacity, especially for some negligibly digested proteins, such as α‐synuclein, in PD. In transgenic Caenorhabditis elegans, these misfolded proteins are transported into EVs migrating from neurons or from muscle to the hypodermis, which disrupts intracellular protein homeostasis of distant cells after rupture (Sandhof et al., 2020). EVs can also abrogate proteostasis through autophagic disruption of recipient cells. In a study using Caenorhabditis elegans to study EV trafficking, miR‐83‐enriched EVs, which drift from the intestine to other tissues in pseudocoelomic fluids, was found to limit the expression of CUP‐5 (an autophagic regulator), obviously reducing autophagy in distinct tissues and causing protein aggregation and senescent phenotype acquisition (Zhou et al., 2019). In addition, EVs can modulate targets’ anabolic activities through accumulation of the very‐long‐chain ceramide C24:1 (Khayrullin et al., 2019), which negatively affects amino acid transporters to starve cells to death (Guenther et al., 2008).
Stem cell exhaustion is another pivotal factor playing a role in EV‐linked aging transmission (Figure 3). Mesenchymal stem cells (MSCs) from bone marrow are among the widely reported stem cells affected by EVs. Incubation of young MSCs with old MSC‐derived EVs stimulates the mTOR pathway, promotes aging marker expression and reduces pluripotency marker levels ( Fafian‐Labora et al., 2020a). Incubation of bone marrow stem cells (BMSCs) with EVs from aged bone marrow interstitial fluids consistently induces stem cell senescence and impedes osteogenic differentiation via the miR‐183 cluster (Davis et al., 2017). Moreover, mouse myoblasts and primary human myotubes treated with hydrogen peroxide generate miR‐34a‐carrying circulating EVs to suppress SIRT1 and induce BMSC senescence and death (Fulzele et al., 2019). Therefore, these shuttled EVs seem to dictate stem cell differentiation, maturation, cessation, and even expiration. However, more convincing in vivo data to verify the impacts of EVs on stem cells are still lacking.
In conclusion, EVs and their cargos play important roles in transmitting aging signals through intersecting networks, partially inducing immune and inflammatory activation, genomic instability, telomere attrition, epigenetic alterations, mitochondrial and lysosomal system dysfunction, ROS accumulation, loss of proteostasis and nutrition, and stem cell exhaustion in targeted cells (Figure 3). All these mechanisms are also prime aging hallmarks of parent cells, but the transmission through EVs is not just a senescent copy. It is a fine‐tuned informative delivery and systemic response. Loss of balance may contribute to senescence‐associated diseases. However, more experiments should be performed to strengthen the evidence, and more attention should be given to the molecular function of EVs in aging transmission and pathology.
4. IMPACTS OF EXTRACELLULAR VESICLES ON AGE‐RELATED DISEASES
Aging is one of the risk factors for chronic diseases and dysfunctional pathologies. In age‐related diseases, senescent cells exert central effector functions to transmit SASP factors to neighbouring cells and tissues, among which different EVs could be representative. However, senescent cells are highly heterogeneous and form multiple colonies; thus, senescent cell‐secreted EVs have different cellular origins and tissue specificities (Table 2).
TABLE 2.
Roles of extracellular vesicles in age‐related diseases
| Disease | Senescent cell sources | Cargo(s) | Mechanism(s) and Effect(s) | Ref. |
|---|---|---|---|---|
| Musculoskeletal diseases | ||||
| Osteopenia and osteoporosis | Osteoclasts | miR‐214‐3p | Promoted osteoclastogenesis and reduced bone formation through PI3K/AKT pathway | (Li et al., 2016) |
| BMSCs | miR‐31a‐5p | Increased adipogenesis and aging phenotypes; Promoted osteoclastogenesis and bone resorption | (Xu et al., 2018b) | |
| HUVECs | miR‐31 | Inhibited osteogenic differentiation of mesenchymal stem cells via Frizzled‐3 restraint | (Weilner et al., 2016) | |
| Osteoarthritis | Chondrocytes | / | Reduced cartilage production and increased the inflammatory state and pain | (Jeon et al., 2019) |
| Degraded cartilage | miR‐449a‐5p | Repressed the expression of the ATG4B gene, leading to inhibition of autophagy; Increased ROS accumulation and inflammasome and IL‐1β activation | (Ni et al., 2019) | |
| Neurodegeneration | ||||
| Alzheimer's disease | Astrocytes and neurons | / | Induced MAC formation and neurotoxicity on recipient neurons | (Nogueras‐Ortiz et al., 2020) |
| Microglia | Tau protein | Microglia phagocytosed Tau protein and facilitated tau propagation to neurons | (Asai et al., 2015) | |
| CSF | BIN1 protein | Reduced microglial Tau clearance, increased Tau release and propagation | (Crotti et al., 2019) | |
| SHswe cells | miRNAs | Induced proinflammatory factor expression and aging in microglia | (Fernandes et al., 2018) | |
| Neuroblastoma cells; AD CSF | Tau protein | Facilitated Tau protein transmission depending on synaptic connectivit; Promoted Tau aggregation in secondary neurons and microglia | (Wang et al., 2017b) | |
| Parkinson's disease | Neuroblastoma cells | Gangliosides | Catalyzed the conversion of α‐synuclein monomeric pattern to fibrillar aggregates | (Grey et al., 2015) |
| HEK293 cells | α‐Synuclein | Facilitated α‐synuclein uptake and dysregulation in monocytes and microglia | (Grozdanov et al., 2019) | |
| LPS‐treated erythrocytes | α‐Synuclein | Traversed the blood‐brain barrier and upregulated microglial inflammatory responses; Promoted the process of synucleinopathy pathogenesis | (Matsumoto et al., 2017) | |
| Microglia | α‐Synuclein | Dysregulated autophagy of the patient cells and accelerated secretion of α‐synuclein | (Xia et al., 2019) | |
| Cardiovascular diseases | ||||
| Vascular calcification | ECs and plasma | Annexins, Ca2+ | Promoted calcification in vascular smooth muscle cells | (Alique et al., 2017) |
| Atherosclerosis | TNF‐α treated ECs | / | Disseminated SASP molecules and promoted the monocyte inflammatory mode; Promoted atherosclerotic plaque formation | (Hosseinkhani et al., 2018) |
| Atherogenic macrophages | miR‐146a | Repressed the expression of IGF2BP1 and the HuR gene; Decreased migration and promoted entrapment of macrophages | (Nguyen et al., 2018) | |
| Cardiac hypertrophy | Cardiac fibroblasts | miR‐21‐3p | Mediated cardiac hypertrophy and remodeling through downregulation of the genes SORBS2 and PDLIM5 | (Bang et al., 2014) |
| Angiotensin‐II treated fibroblasts | / | Upregulated the expression of renin, angiotensinogen, Ang II receptor, and Ang II in cardiomyocytes via the MAPK and AKT pathways | (Lyu et al., 2015) | |
| Fibroblasts | miR‐27a | Induced ROS with hypertrophic gene expression and suppressed PDLIM5 gene expression | (Tian et al., 2020) | |
| Heart failure | Fibroblasts | miR‐146a | Suppressed SUMO1 expression, SERCA2a SUMOylation and cardiac contractility | (Oh et al., 2018a) |
| Cardiac fibrosis | Hypertrophied myocytes | Hsp90 | Activated STAT‐3 signaling of cardiac fibroblasts and enhanced excess collagen synthesis | (Datta et al., 2017) |
| Myocytes under hypoxia | LncRNA Neat1 | Mediated fibroblastic cell survival, fibrosis, proliferation and migration via the AKT pathway | (Kenneweg et al., 2019) | |
| Type 2 diabetes mellitus | Diabetic plasma | inflammatory proteins and VEGF‐A | Increased lamellipodia formation and migration in endothelial cells; Increased the potential for peripheral vascular diseases | (Wu et al., 2020) |
| Erythrocytes | / | Altered the expression of genes related to cell survival, oxidative stress and immune function in leukocytes; increased inflammatory cytokine levels | (Freeman et al., 2018) | |
| BMSCs | miR‐29b‐3p | Inhibited the insulin signaling pathway; Exacerbated insulin resistance of young BMSCs through SIRT1 dysfunction | (Su et al., 2019a) | |
| Adipose tissue macrophages | miR‐155 | Reduced insulin sensitivity of insulin target cells through PPARγ suppression; Induced insulin resistance and glucose intolerance | (Ying et al., 2017) | |
| Adipocytes | miR‐27a | Decreased IRS‐1, GLUT4 and AKT in insulin target cells through PPARγ inhibition; Impaired glucose consumption and insulin sensitivity of skeletal muscle cells | (Yu et al., 2018) | |
| High glucose‐treated ECs | Notch 3 | Increased the expression of osteocalcin, p21 and SA‐β‐gal in vascular smooth muscle cells; Increased VSMC calcification, aging and the potential for cardiovascular diseases | (Lin et al., 2019) | |
| Albumin‐treated renal tubular epithelial cells | miR‐4756 | Increased epithelial‐to‐mesenchymal transition and endoplasmic reticulum stress in tubular epithelial cells through Sestrin2 inhibition; Accelerated diabetic kidney disease progression | (Jia et al., 2019b) | |
| Reproductive diseases | ||||
| Ovarian aging | Follicular fluid of elderly women | miR‐21‐5p; miR‐134; miR‐190b | Regulated heparan‐sulfate proteoglycan expression and deregulated follicle morphogenesi; Increased apoptosis of human granulosa cells through regulation of the p53 pathway, bcl2, and IKBKG | (Diez‐Fraile et al., 2014) |
| Follicular fluid of elderly women | miR‐16; miR‐214; miR‐372 | Increased apoptosis and cellular senescence though activation of the p53 pathway; Increased EV production through feedback | (Battaglia et al., 2020) | |
| Follicular fluid of aged mares | miR‐181a | Decreased proliferating cell nuclear antigen (PCNA) levels of granulosa cells; Suppressed the TGF‐β pathway and impaired oocyte maturation | (da Silveira et al., 2015) | |
| Plasma in POF patients | miR‐23a | Increased apoptosis of granulosa cells through XIAP inhibition and caspase‐3 activation | (Yang et al., 2012) | |
| Uterine aging | Endometrial stromal cells | PAI‐1 | Transmitted senescence patterns in 2D and 3D senescent and young ESC coculture systems; Perhaps dysregulated endometrial function and impaired embryo implantation | (Griukova et al., 2019) |
4.1. Bone diseases
Bone is a dynamic tissue regulated by life‐long formation and resorption that requires stem cells, osteoclasts, and osteoblasts to cooperate precisely. Once bone equilibrium is disrupted, redundant bone resorption and reduced bone formation can cause osteopenia and osteoporosis (Li et al., 2016). Bone loss is a more typical feature of elderly individuals than young individuals. This pattern of disease indicates that impaired intercellular contacts strongly collaborate with the senescence process in bone loss.
Mesenchymal stem cells (MSCs) in bone marrow are multipotent stem cells based on their ability to differentiate into chondrocytes, osteoblasts, adipocytes, and stromal cells to maintain osseous tissue homeostasis and effective performance in bone remodelling and regeneration (Wolock et al., 2019). However, aged BMSCs show a decline in proliferation rate, loss of stemness and viability, decreased osteogenic and chondrogenic differentiation, and increased adipogenesis (Baker et al., 2015). With aging, BMSCs are susceptible to excessive ROS deposition and energy disorder on the path to exhaustion and anergy (Denu & Hematti, 2016). The two main intrinsic factors (ROS deposition and energy disorder) may be effectively controlled by depletion of inositol hexakisphosphate kinase 1 (Ip6k1), enhancing BMSC growth and survival. Ip6k1 is usually described as a regulator of fat accumulation through AMPK‐mediated adipose energy metabolism (Boregowda et al., 2017; Zhu et al., 2016).
As mentioned previously, extrinsic factors, such as EVs, also generate harmful impacts on the aging process by transmitting miRNA signals. In osteoporosis, it has been experimentally proven that the crosstalk between BMSCs and osteoclasts relies on EV conveyance of information in bone marrow. MiR‐31a‐5p derived from aged BMSCs mechanically binds to the 3′UTR of the RhoA gene to inhibit osteoclast cytoskeletal organization in a paracrine manner while directly attaching to the SATB2 and IE2F2 genes in an autocrine manner to decrease self‐stemness and osteogenic differentiation (Xu et al., 2018b; Zhou et al., 2016). Osteoclasts purified from ovariectomized mice transport EVs containing miR‐214‐3p to cocultured osteoblasts, ultimately inducing osteoclastogenesis through the PI3K/AKT pathway (Li et al., 2016). Other cells, such as senescent endothelial cells, promote osteoporosis development via miR‐31 transmission to MSCs, inhibiting their capacity for differentiation (Weilner et al., 2016). Notably, exosome functions in the serum of osteoporosis and osteopenia patients are contradictory. The latter demonstrate a compensatory elevation in bone formation. These contradictory findings suggest that greater attention should be given to the ageotypes of elderly patients when considering the use of EVs as diagnostic biomarkers.
Osteoarthritis (OA) is another common age‐associated bone disease characterized by distinct chondrocyte catabolism and death, cartilage matrix loss, and an inflammatory state due to oxidative stress and the SASP mode (Loeser et al., 2016). The contents of EVs in synovial fluids differ significantly under osteoarthritis conditions and by sex. Patients with severe OA tend to show stimulated release of proinflammatory factors from M1 macrophages (Domenis et al., 2017; Kolhe et al., 2017). In females, EVs are involved in the ovarian steroidogenesis signaling pathway and metabolic process (Kolhe et al., 2017). In OA‐affected joints, chondrocytes and immune cells also deliver pathogenic signals reciprocally via EV shuttling (Zhou et al., 2020). Taking miR‐449a‐5p as an example, senescent OA chondrocytes produce miR‐449a‐5p vesicles to repress ATG4B gene expression in macrophages and then inhibit their autophagy. Subsequently, ROS deposition leads to inflammasome activation and IL‐1β release (Ni et al., 2019). Therefore, eliminating senescent cells or interfering with EV contents may be beneficial to osteoporosis and osteoarthritis.
4.2. Neurodegeneration
As the frequencies of neurodegenerative diseases exponentially increase with age, postmitotic cells in the brain show extreme sensitivity to systemic and cellular senescence. Alzheimer's disease (AD) and Parkinson's disease (PD) are the most extensively studied neuronal senescence‐associated conditions to date, and both are mainly attributed to insoluble misfolded protein aggregation in specific regions of the brain. These neuropathic proteins are closely linked with senescence‐associated autophagy and lysosomal dysfunction, which lead to their stable presence in specific EVs (Chung et al., 2019; Kapogiannis et al., 2019; Lamontagne‐Proulx et al., 2019; Minakaki et al., 2018). Additionally, evidence has shown that injection of EVs isolated from AD patients into the hippocampi of wild‐type mice is neurotoxic in vivo, leading to increased tau phosphorylation (Aulston et al., 2019). After such EVs are intravenously injected from the serum of PD patients into mice, protein aggregation, dopamine neuron degeneration, microglial activation, and mouse movement defects are also observed (Han et al., 2019a). This directly indicates that EVs packaged with a pathogenic pattern of molecules may play vital roles in neural aging phenotype transmission.
To precisely evaluate and diagnose age‐associated neurodegeneration, secreted EVs in various body fluids may be the optimal biomarkers. Taking advantage of blood‐brain barrier permeability to EVs, brain‐derived exosomes can be detected in the peripheral circulation. Neuron‐derived EVs in serum, sorted by L1CAM and neural cell adhesion molecule expression, were found to exhibit elevated phosphorylated tau‐181 and 231, Aβ42 and insulin receptor substrate 1 levels in an AD group (Kapogiannis et al., 2019). Compared to exosome‐unbound Aβ, exosome‐bound Aβ can better reflect amyloid plaques of the brain and be used to differentiate various clinical groups via amplification techniques (Lim et al., 2019). The levels of advanced glycation end products N‐(1‐carboxymethyl)‐L‐lysine (CML) in EVs present dynamic changes in early and moderate AD (Haddad et al., 2019). However, there is little correlation in the miRNA contents of EVs in brain and blood samples obtained from AD subjects (Cheng et al., 2020), while similar levels of exosomal Aβ42 and tau in CSF and blood have been verified (Jia et al., 2019a). A novel tau fragment in AD, N‐224 tau, can be packaged in neuron‐specific EVs and secreted in CSF to upregulate cognitive decline (Cicognola et al., 2019). In PD, in addition to α‐synuclein detected in CSF, numerous noncoding RNAs are significantly upregulated in EVs, including miR‐153, miR‐409‐3p, and let‐7 g‐3p (Gui et al., 2015; Stuendl et al., 2016). Interestingly, the α‐synuclein oligomer and the total ratio of α‐synuclein in salivary EVs can be noninvasively assessed in PD patients, and α‐synuclein levels increase with disease severity (Cao et al., 2019). Consequently, it is valuable to classify specific EVs in biofluids as ageotypes and obtain profiles of AD and PD patients for early and safe diagnosis.
In particular, EVs can contribute to the development and aggravation of these neurodegenerative diseases through the intercellular transmission of misfolded proteins or other pathological molecules. In AD, EVs with tau protein are released by primarily affected neurons after depolarization and internalized by the next neurons through synaptic transmission to cause protein folding errors in normal cells (Wang et al., 2017b). SHswe cells (a neuroblastoma cell line transfected with the Swedish mutant of amyloid precursor protein) are capable of stimulating the microglial immune response and increasing senescence hallmark activation through EV‐containing miRNA transmission between neurons and microglia (Fernandes et al., 2018). Microglia, which are tissue‐resident macrophages, not only facilitate tau release in EVs but also facilitate neuroinflammation development via EVs (Asai et al., 2015). With age, the accumulation of internal antigen‐antibody compounds and misfolded proteins stimulates microglia to overexpress several complement components involved in the complement cascade and maladaptive neuroinflammatory effects (Spani et al., 2015). High complement levels can also be found in astrocytic EVs and neuronal EVs (Goetzl et al., 2018). These inflammatory EVs cause cellular membrane disruption and neurite density reduction in rat cortical neurons by activating membrane attack complex (MAC) formation (Nogueras‐Ortiz et al., 2020). Age‐associated chronic inflammatory states are similarly evident during PD progression. α‐Synuclein in EVs, whether from the brain or the periphery, can incessantly evoke innate immune responses and interfere with the clearance system, resulting in synucleinopathy pathogenesis (Grozdanov et al., 2019; Matsumoto et al., 2017; Xia et al., 2019). In a sense, pathological pattern transmission via EVs is equivalent to aging phenotype transmission, as both involve crosstalk of diverse cells in the brain microenvironment and induce systemic immune responses to low‐grade inflammation.
4.3. Cardiovascular diseases
Cardiovascular diseases are the most prevalent types of age‐associated conditions and manifest as structural, cellular, molecular, and functional changes in the heart that occur with aging. Numerous circulating progeronic and antigeronic factors from different systems orchestrate the aging process simultaneously in the cardiovascular system. They can cause vascular aging and subsequently impair angiogenesis and induce chronic inflammation, pathological remodelling, atherogenesis, and myocardial injury (Fajemiroye et al., 2018; Ungvari et al., 2018). In the endocrine system, glucose stimulation aggravates endothelial dysfunction and increases NADP (nicotinamide adenine dinucleotide phosphate) oxidase activity and ROS levels, producing more senescent endothelial EVs that mediate cardiovascular diseases in diabetes (Carracedo et al., 2019). Other factors, such as proinflammatory cytokines (IL‐1, TNF‐α, and INF‐γ), coagulant factors (thrombin and PAI‐1), hypoxia, and low shear stress, can also stimulate endothelial cells to release progeronic EVs (Hromada et al., 2017).
Vascular calcification, a risk factor for atherosclerosis and myocardial injury, is markedly induced by age‐related EVs. EVs extracted from replicative senescent endothelial cells (ECs) and the plasma of elderly patients contained increased levels of annexins, BMP2 (a bone morphogenic protein) and Ca2+, which induce the initiation and propagation of calcification in human aortic smooth muscle cells (Alique et al., 2017; Pescatore et al., 2019). The interaction between monocytes and ECs also plays a crucial role in inflammatory atherosclerosis. TNF‐α‐induced ECs can produce more EVs containing chemotactic mediators, such as ICAM‐1, CCL‐2, CXCL‐10, and CCL‐5, which are taken up by monocytes to promote the expression of their inflammatory markers (IL‐6 and IL‐8) and their adhesion and migration (Hosseinkhani et al., 2018). On the other hand, activated monocytes constitute mitochondria‐rich EVs and initiate type I INF and TNF signaling pathways in ECs, leading to chronic inflammation (Puhm et al., 2019). In addition, atherogenic macrophages can decrease migration and promote the entrapment of self‐ or naïve macrophages through inhibition of IGF2BP1 and HuR expression by miR‐146a‐associated EVs (Nguyen et al., 2018). Elucidating the intercellular trajectory, mediators, and cargos of EVs will aid in attenuation of vascular calcification and atherosclerosis.
In acute myocardial infarction, EVs are transiently increased and mainly originate in myocytes and ECs. Monocytes can readily take up these EVs to regulate inflammatory responses in the local region (Loyer et al., 2018). The evidence supports the idea that the ischemic myocardium selectively releases EVs from leukocytes, ECs and myocytes in vivo. Concomitantly, EVs from myocytes upregulate MMP3 and other genes associated with apoptosis and senescence in ECs (Rodriguez et al., 2018). These age‐affected myocardial injured tissues undergo cardiomyocyte hypertrophy, apoptosis, cardiac fibrosis, and angiogenesis in the interplay among myocytes, ECs, fibroblasts, and immune cells. Between myocytes and fibroblasts, EVs from fibroblasts join in the pathological process of cardiac hypertrophy and heart failure. MiR‐21‐3p and miR‐27a have been proven to suppress the PDLIM5 gene of myocytes and decrease cardiac contractility, which mainly mediates cardiomyocyte hypertrophy and remodelling, ultimately leading to heart failure (Bang et al., 2014; Tian et al., 2020). Together, the evidence indicates that myocytes exert a profound influence on cardiac fibrosis through EV trafficking. Deposition of extracellular matrix is markedly reduced in fibroblasts conditioned with supernatant from Hsp90‐inhibited hypertrophied myocytes. In fibroblasts, Hsp90 inhibition decreases IL‐6 levels and STAT‐3 activation to ameliorate cardiac fibrosis (Datta et al., 2017). Interestingly, EVs from aged mouse serum contain reduced levels of Hsp70. However, the inhibition of Hsp70 promotes fibroblast proliferation and fibroblast‐to‐myofibroblast differentiation (Yang et al., 2019). Fortunately, EVs from stem cells usually display positive phenotypes in cardiac trauma repair to ameliorate postinfarction remodelling and attenuate ischemic progression. We discuss the functions of EVs from stem cells in further detail below. Blockade of pathological EV communication among these cells or utilization of stem cell therapy are methods that can be used to attenuate myocardial injury and ischemic diseases.
4.4. Type 2 diabetes mellitus (T2DM)
It has been confirmed that aging greatly increases susceptibility to chronic and refractory age‐related diseases. In turn, age‐related diseases, in a mechanism similar to positive feedback, exacerbate the aging microenvironment and accelerate the aging process. In particular, a diabetic microenvironment is replete with factors leading to senescence and hyperglycaemia, both of which collaborate to induce long‐standing diabetes mellitus and severe complications. In the endocrine system, message delivery via EVs is essential for realization of aging transmission. In a diabetic microenvironment, EVs from MSCs are aging factor carriers that induce renal tubular cell senescence, fibrosis and dysfunction (Li et al., 2020c). These outcomes indicate that stem cells problems should be monitored when they are utilized in tissue repair of T2DM patients.
The increased EVs from the circulation and cells induce diabetic senescence and organ injury, subsequently causing systemic inflammation and insulin resistance. Evidence has shown that mouse islets under attack from mixed cytokines and streptozotocin can produce dramatically elevated amounts of injury‐related exosomal miR‐375‐3p in serum prior to the development of blood glucose and insulin disorders (Fu et al., 2018). In addition, CD31+/annexin V‐labelled EVs from damaged endothelial cells show higher sensitivity in T2DM than in metabolic syndrome, which leads to immune cytokine overproduction and vascular aging (Berezin et al., 2015). Research on EVs obtained from T2DM plasma, mainly erythrocyte‐derived EVs, has revealed the immune function of these EVs, which are internalized by circulating leukocytes; this internalization alters their gene expression related to cell survival, oxidative stress and cytokine release (Freeman et al., 2018).
Bone can be regarded as another endocrine tissue that regulates systemic hormone metabolism. BMSCs from aged mice target SIRT1 in adipocytes, myocytes and hepatocytes through exosomal miR‐29b‐3p to significantly induce age‐related insulin resistance in vitro and increase blood glucose in vivo (Su, Xiao et al., 2019). Moreover, EVs from obese tissue engage in age‐related DM. Excessive calorie intake in mice can induce cellular senescence in adipose tissue through accumulation of ROS and activation of p53, which have been proven to play roles in insulin resistance (Minamino et al., 2009). Thereinto, exosomal miR‐27a in adipocytes has been verified to participate in the insulin resistance of muscle cells through repression of downstream PPARγ (Yu et al., 2018). In addition to adipocytes, macrophages from adipose tissue can release EVs to modulate cellular insulin action via the same target gene (Ying et al., 2017). These findings suggest that controlling energy intake not only maintains glucose homeostasis but also ameliorates senescence.
However, the true risk of diabetes involves its complications, especially microvascular and macrovascular diseases. Long‐term exposure to high glucose causes endothelial cells to transfer exosomal Notch3 to vascular smooth muscle cells, consequently increasing senescence and osteocalcin expression to induce vascular calcification and aging (Lin et al., 2019). Additionally, renal tubular epithelial cells are injured by albumin in diabetic macroalbuminuria. Exosomal miR‐4756 accelerates the aging and injury of these cells by suppressing Sestrin2 to induce endoplasmic reticulum stress and the epithelial‐to‐mesenchymal transition (Jia et al., 2019b). Despite the multiorgan hazards and refractoriness of T2DM, metformin therapy has been confirmed as a jack‐of‐all‐trades drug to achieve antiaging, antidiabetes, and antiobesity effects (Cabreiro et al., 2013; Onken & Driscoll, 2010). A spectrum of circulating miRNAs in EVs in T2DM subjects treated with metformin has been found to be surprisingly similar to that of healthy controls (Ghai et al., 2019). However, caution should be exercised when considering the use of metformin as an antiaging agent for treating healthy elderly patients. A recent study warns that it will enhance mitochondrial dysfunction and metabolic disturbance in late life, shortening lifespan (Espada et al., 2020).
4.5. Reproductive diseases
Reproductive aging, including ovarian aging and uterine aging, is the earliest type of system senescence in women, as the ovaries exhibit gradual declines in follicle quantity and quality from birth onwards. Detrimental effects of reproductive aging mainly affect oocyte quality and reserves, female hormonal balance, energy metabolism, and pathological changes. Evidence has highlighted that reduced fertilization and implantation and increased miscarriage occur in women of child‐bearing age with ovarian aging. A single‐cell transcriptomic analysis of the ovaries in young and old nonhuman primates demonstrated that the expression of genes involved in oxidative phosphorylation and oxidoreductase activity pathways was reduced in aging oocytes and granulosa cells, contributing to oxidative damage and apoptosis (Wang et al., 2020). Another single‐cell transcriptomic analysis in the ovaries of aged mice after supplementation with nicotinamide mononucleotide (NMN), an NAD+ intermediate, showed that mitochondrial function was significantly restored with ROS elimination after NMN supplementation (Miao et al., 2020). Both studies showed that mitochondrial dysfunction had adverse effects on oocyte function with aging.
In addition to mitochondrial dysfunction in the ovaries, apoptosis causes infertility and ovarian aging through senescence transmission by EVs. Downregulation of X‐linked inhibitor of apoptosis protein (XIAP) has been found to occur in aged granulosa cells via miR‐23a, which activates caspase‐3 and increases apoptosis of granulosa cells (Yang et al., 2012). In follicular fluid obtained from older women, exosomal miR‐134 has also been found to be potentially involved in an apoptotic pathway upon targeting of Bcl2 and IKBKG (Diez‐Fraile et al., 2014).
In endometrial stromal cells, EVs also transmit a senescent phenotype in 2D and 3D cell coculture systems using aged and young cells, which dysregulates uterine function during pregnancy and impairs embryo implantation (Griukova et al., 2019). However, there is little evidence to confirm that senescent EVs are strongly linked with uterine aging during a woman's life. Nevertheless, inflammasome‐containing EVs with specific Nik‐related kinases present only in the reproductive system have been detected in the CSF and serum of replicative senescent rats. These inflammasomes can trigger an innate immune response in senescent female reproductive organs and then the brain (Raval et al., 2019). This result indicates that women with replicative aging are more susceptible to neurodegenerative diseases and that EVs might be the pacemakers of female aging, delivering information from the ovaries to the whole body. By monitoring the cargos and pathways of these EVs in the reproductive system with aging, we will be able to elucidate the trajectories of aging transmission in female individuals, predict pathological changes, and intervene in the occurrence of age‐related diseases. These findings show that for fertility preservation, we can increase the reproductive rate and quality and decrease infertility and reproductive abnormalities through age‐related EV control.
5. EXPLOITING SENESCENCE FOR THERAPEUTICS
5.1. Targeting senescence through senotherapy
There is a growing consensus that researching and developing therapeutic interventions for age‐related disorders is necessary to alleviate the severe economic, healthcare and humanitarian challenges of the global aging society (Zhao & Stambler, 2020). Our aging population must cope with increasing threats of senescence‐related dysfunction, among which mortality is the top concern and the most challenging. COVID‐19 results in markedly higher in‐hospital mortality in patients with advanced chronological age than in younger patients, possibly due to involvement of CD26 and ACE‐2 (Koff & Williams, 2020; Li et al., 2020a; Radzikowska et al., 2020) (two host receptors that show a significant association with senescence (Li et al., 2018; Takeshita et al., 2018)). In experimental animals, inhibition of cellular senescence with the transgenic suicide gene INK‐ATTAC can expand lifespan and healthspan through inducible elimination of p16INK4a‐positive senescent cells upon drug treatment (Baker et al., 2011). Using small molecules to therapeutically target exclusive markers of aging cells will likely have positive effects on the quality of life and the burden of age‐related diseases. Hence, there is a growing interest in the development of antiaging treatment, termed senotherapy (Figure 4). In principle, senotherapeutic strategies can be broadly classified into two categories: senolytic strategies (antiaging strategies for selective removal of senescent cells (Jeon et al., 2017)) and senomorphic strategies (antiaging strategies that block senescent phenotypes by inhibiting the SASP without compromising cell death (Short et al., 2019)).
FIGURE 4.

Approaches for exploiting senescence for therapeutics: senolytics, senomorphics and EV‐mediated regeneration and rejuvenation. There is a growing interest in developing antiaging therapies, termed senotherapies. Senotherapeutic strategies can be traditionally classified into two categories: senolytic strategies (antiaging strategies for selective removal of senescent cells, including compound treatment and immune system‐mediated clearance) and senomorphic strategies (antiaging strategies that block senescent phenotypes by inhibiting the SASP without compromising cell death). Recently, EVs, as next‐generation candidates for antiaging therapy, have been well studied for their preventive, rejuvenating and anti‐inflammatory effects on senescence. There are several natural sources (from stem cells, young or centenarian donors), modified sources and artificial sources of therapeutic EVs
5.1.1. Senolytics
Senolytic therapy specifically kills senescent cell populations depending on apoptotic resistance, an essential hallmark of cellular senescence. Using apoptosis‐inducing drugs, several intracellular prosurvival pathway members have been targeted to eliminate senescent cells, including Bcl‐2 family members (including Bcl‐2, Bcl‐W, and Bcl‐XL), p53/FOXO4/p21 pathway members, PI3K/Akt pathway members, receptor tyrosine kinases, HSP90, HDACs, and glutamine metabolism pathway members (Song et al., 2020).
The senolytic agent ABT‐737 and its orally bioavailable formulation ABT‐263 (navitoclax), which belong to the class of BH3 mimetics, can inhibit Bcl‐2, Bcl‐W, and Bcl‐XL and initiate apoptosis of senescent cells through a mitochondria‐dependent pathway (Basu, 2021). The highly potent and selective Bcl‐XL inhibitors A‐1155462 and A‐1331852 have also been proven to have proapoptotic ability but less hematological toxicity than navitoclax (Zhu et al., 2017). In multiple senescence events, the transcription factor FOXO4 is upregulated and interacts with p53 in the nucleus, preventing apoptosis. Administration of a peptide of FOXO4, FOXO4‐DRI, can perturb this interaction and cause p53 nuclear exclusion and cell‐intrinsic apoptosis in vitro and in vivo (Baar et al., 2017). In senescent liver cancer cells with p53 mutations, the antidepressant sertraline exerts a selective proapoptotic function via inhibition of the DNA‐replication kinase CDC7 and suppression of mTOR signaling (Wang et al., 2019). In addition, quercetin and fisetin, two natural flavonoids, have also been shown to have senolytic properties in the modulation of several pathways, such as the PI3K/Akt/mTOR and NF‐κB pathways (Ngoi et al., 2021). There exists a classic senolytic cocktail of dasatinib, a tyrosine kinase inhibitor, plus quercetin (D+Q), which has been well studied in the elimination of a wide range of senescent cells (Zhu et al., 2015). The combination of D+Q treatment can alleviate physical dysfunction and improve lifespan and healthspan in naturally aged mice by reducing the numbers of senescent cells and the levels of cytokines (Xu et al., 2018a). This senolytic combination has been verified to be effective in phase I clinical trials for diabetic kidney disease (NCT02848131) and idiopathic pulmonary disease (NCT02874989) (Hickson et al., 2019; Justice et al., 2019). In apoptosis, HSP90 is an important chaperone protein that induces the stabilization and degradation of proteins and impacts mTOR and NF‐κB signaling. Its primary inhibitor 17‐DMAG is a part of several classes of senolytics that delay age‐related pathologies in progeroid mice (Fuhrmann‐Stroissnigg et al., 2017). The HDAC inhibitor panobinostat has also been repurposed to target persistent senescent cells that accumulate after chemotherapy due to the link between senescence and histone deacetylation (Samaraweera et al., 2017). Recently, glutamine metabolism has been noted for its association with senescence. Glutaminase 1 (GLS1) not only mediates glutaminolysis but also promotes senescent cell survival, inhibition of which can clear senescent cells and improve age‐related disorders in geriatric mice (Johmura et al., 2021).
In the aging microenvironment, senescent cells can be subjected to the intrinsic immunosurveillance system, which presents a suppressive trend with age. Restoring the ability of the immunosurveillance system or using modified immune cells (such as CAR‐T cells (Amor et al., 2020)) might be successful in the targeted clearance of senescent cells from tissues (Di Micco et al., 2021). For example, to enhance the cytotoxic activity of NK cells, polyinosinic‐polycytidylic acid (polyI:C, a TLR3 ligand) has been administered to induce IFN‐γ and activate the NK cell receptor NKG2D, resulting in clearance of senescent activated stellate cells and amelioration of liver fibrosis (Krizhanovsky et al., 2008).
5.1.2. Senomorphics
Senomorphic therapy interferes with the proinflammatory nature of senescent cells and normalizes the SASP process in the aging microenvironment through the regulation of various biochemical pathways involving mTOR, p38 MAPK, NF‐κB, JAK/STAT, ROCK, glucocorticoid receptors, and neutralizing antibodies (Prasnikar et al., 2021).
As stated previously, several stress response signaling cascades are activated to induce the secretion of inflammatory components, including NF‐κB, C/EBPβ and their upstream components. Through efficient blockade of the signaling cascades or direct inhibition of the activity of SASP components, the amplifying outcomes of senescence can be ameliorated in the aging microenvironment. mTOR, one of the most critical upstream signaling pathways in the process of cellular senescence and the SASP, can be inhibited with rapamycin and its analogs, such as RAD001, and can present a senomorphic function. In normal human fibroblasts and breast cells, rapamycin can suppress IL‐6 secretion, selectively decrease IL‐1α translation and diminish NF‐κB transcriptional activity (Laberge et al., 2015). In Nrf2KO mice and WI‐38 fibroblasts, it can also significantly decrease IL‐6 and IL‐1 transcription through Stat3 inhibition in a Nrf2‐independent mechanism (Wang et al., 2017a). Inhibition of another essential SASP‐modulated pathway, the p38 MAPK pathway, with SB203580 and the next‐generation compounds UR‐13756 and BIRB796 has also been proven to reduce NF‐κB transcriptional activation, thereby inhibiting the paracrine effects of senescence (Alimbetov et al., 2016; Freund et al., 2011). In addition to NF‐κB, the JAK/STAT pathway can sustain cytokine production through C/EBPβ activation. In human preadipocytes, endothelial cells and aged mice, the JAK1/2 inhibitor ruxolitinib attenuates SASP‐related secretion and reduces systemic inflammation, leading to improvements in metabolic function and physical capacity (Xu et al., 2015). However, given the diversity of the signaling pathways of the SASP in different cell types, interference with any one pathway may not affect all components.
Another strategy of senomorphics is to achieve the antiaging goal through direct modulation of NF‐κB binding and activation. Among senomorphics with this strategy, metformin has been used the most extensively. Metformin, an FDA‐approved oral hypoglycaemic medication for T2DM, was the first drug to be tested in a large clinical trial for age‐related diseases termed the TAME (Targeting Aging by Metformin) trial (Barzilai et al., 2016). It can significantly interfere with IKK/NF‐κB activity and reduce NF‐κB nuclear translocation and activation to decrease SASP‐related production independently of AMPK (Moiseeva et al., 2013). Additionally, it inhibits mTOR activity and modulates certain downstream cellular effects to extend lifespan across multiple model systems (Soukas et al., 2019). However, its mechanisms in aging and the SASP are not yet clearly defined. The SASP can also be regulated by a class of anti‐inflammatory steroid hormones, the glucocorticoids. Both corticosterone and cortisol are efficacious in suppressing the SASP via glucocorticoid receptors by preventing IL‐1α positive feedback and inhibiting NF‐κB binding and transactivation (Laberge et al., 2012). Although reduction of SASP component production is an aspect of traditional senomorphic treatment, the release process of SASP is rarely targeted for interference. Recently, it was reported that inhibition of Rho/Rho kinase with CT04 and Y27632 may not only alter the morphology of senescent cells but also affect the secretion of senescent cells via changes in the ER/Golgi system (Simay Demir et al., 2021). Alternatively, more direct inhibition can be achieved by specific neutralizing monoclonal antibodies against SASP factors, such as MABp1 for IL‐1α, ABX‐IL‐8 for IL‐8 and olokizumab for IL‐6 (von Kobbe, 2019).
Collectively, these strategies take advantage of a broad spectrum of cellular pathways, indicating that senescence can be modulated and controlled via numerous avenues. However, some of these drugs are in fact repurposed anticancer medicaments, the adverse effects of which cannot be ignored. Additionally, inhibition of senescence through clearance or disruption does not directly reverse aging. The efficacy of these senotherapies is still limited for rejuvenation and regeneration. Hence, other antiaging therapies urgently need to be developed creatively from novel perspectives of aging and translated rapidly from the bench to the bedside.
5.2. Achieving rejuvenation and regeneration via EVs
Extracellular vesicles are able to program their target cells to follow a Trojan‐horse approach, which enables them to exert pathogenic and curative effects depending on their source cells. In the aging microenvironment, EVs can transmit aging phenotypes from senescent cells in amplifying ways. However, they can also be cell‐free therapeutic materials and tools with no risk of tumour induction, and they show a low probability of immune rejection during regeneration and rejuvenation (Adamiak et al., 2018; Zhai et al., 2020). It has been determined that EVs are next‐generation candidates for use in achieving antiaging goals.
5.2.1. Natural sources of EVs
There are natural, modified and artificial sources of therapeutic EVs (Table 3). Among natural sources, stem cells are the primary sources, and young donors are the main population group supplying these sources. EVs originating from stem cells contain diverse cargos that can execute antioxidative stress and anti‐inflammatory activities in order to induce antiaging effects (Figure 4).
TABLE 3.
Novel antiaging treatments via extracellular vesicles
| Treatments and sources | Cargo(s) | Subjects | Mechanism(s) and Effect(s) | Ref. |
|---|---|---|---|---|
| Stem cell‐derived therapeutics | ||||
| Hypothalamic stem cells | miRNAs | Aged mice | Blocked NF‐κB activation and restored GnRH secretion in neurons; Reduced age‐related physiological deficits and extended lifespan | (Zhang et al., 2017) |
| Human umbilical cord MSCs | LncRNA MALAT1 | Cardiomyocytes with H2O2‐induced aging; Mice with D‐galactose‐induced aging | Inhibited the NF‐κB/TNFα pathway; increased TERT expression; Increased the left ventricular ejection fraction and fractional shorting in aging mice | (Zhu et al., 2019) |
| Human umbilical cord MSCs | / | UVB‐irradiated HDFs | Reduced production of ROS and increased glutathione peroxidase; Inhibited UVB‐induced senescence and induced fibroblast proliferation | (Deng et al., 2020) |
| ADSCs | / | IL‐1β‐treated OA osteoblasts | Reduced inflammatory mediators including IL‐6 and prostaglandin E2; Controlled mitochondrial membrane stability and reduced oxidative stress | (Tofino‐Vian et al., 2017) |
| ADSCs | miRNAs Proteins | UVB‐irradiated HDFs | Induced senescent fibroblast proliferation and DNA repair and attenuated aging; Suppressed the expression levels of MMP‐1, 2, 3, and 9 and enhanced collagen; Provided protection from UVB irradiation and enhanced recovery from skin photoaging | (Choi et al., 2019) |
| ADSCs | / | Myocardial infarction mice, myocytes, fibroblasts, and macrophages | Activated S1P/SK1/S1PR1 signaling and promoted M2 polarization; Suppressed cardiac dysfunction, apoptosis, fibrosis, and the inflammatory state | (Deng et al., 2019) |
| ADSCs | / | Photoaging mice and fibroblasts | Attenuated macrophage infiltration and ROS production; Promoted cell proliferation and decreased skin wrinkles | (Xu et al., 2020) |
| Human iPSCs | / | Aged and UVB‐irradiated HDFs | Reduced the expression levels of SA‐β‐Gal and MMP1, 3; Restored collagen type I and ameliorated UVB‐induced skin aging | (Oh et al., 2018b) |
| Human urine‐derived stem cells | CTHRC1; OPG | Osteoporotic mice | Enhanced osteoblastic bone formation and suppressed bone resorption | (Chen et al., 2019b) |
| Human embryonic stem cells | miR‐200a | Aged mice with pressure‐induced ulcers; Senescent endothelial cells | Downregulated Keap1 and activated the Nrf2 pathway; Enhanced EC proliferation and angiogenesis; Accelerated wound closure and ameliorated senescent phenotypes | (Chen et al., 2019a) |
| Human embryonic stem cells | 4122 proteins | Senescent BMSCs | Rescued the function of senescent BMSCs and promoted proliferation; Induced osteogenic differentiation and reduced age‐related bone loss | (Gong et al., 2020) |
| Embryonic stem cells | Several miRNAs | Vascular dementia mice | Alleviated senescence and loss of hippocampal stem cells; Reduced cognitive decline by activating mTORC1 and promoting TFEB translocation and lysosome resumption | (Hu et al., 2020) |
| Embryonic stem cells | SMAD4, 5 | Aged mice | Activated MYT1 and improved HIF‐2α, NAMPT and SIRT1 levels; Alleviated hippocampal stem cell senescence, reversed cognitive impairment | (Hu et al., 2021) |
| BMSCs | Neprilysin | APP/PS1 AD mice | Reduced Aβ plaques and dystrophic neurites in the cortex and hippocampus | (Elia et al., 2019) |
| Human decidua‐derived MSCs | / | High glucose‐treated fibroblasts; Diabetic mice | Rescued the senescent state via inhibition of RAGE and activation of Smad; Accelerated collagen deposition and enhanced diabetic wound healing | (Bian et al., 2020) |
| Amniotic fluid MSCs | miR‐21 | Mice with premature ovarian dysfunction | Increased total follicular counts and AMH levels and restored regular oestrous cycles by modulating the PTEN and caspase 3 apoptotic pathway | (Thabet et al., 2020) |
| Amniotic fluid MSCs | miR‐320a | Mice with premature ovarian insufficiency; POI human granulosa cells | Decreased SIRT4 and ROS levels; Improved proliferation, inhibited apoptosis and elevated ovarian function | (Ding et al., 2020) |
| Dental pulp stem cells | miR‐302b | Senescent dental pulp stem cells | Improved stemness through HIF‐1α and OSKM upregulation; Switched energetic metabolism toward glycolysis through the ERK pathway | (Mas‐Bargues et al., 2020) |
| Modified stem cell therapeutics | ||||
| GAG‐coated matrix vesicles | / | BMSCs | Promoted differentiation of osteoblasts from hBMSCs and reduced osteoclasts | (Schmidt et al., 2016) |
| Serially extruded human iPSCs | Oct4 Nanog | Naturally senescent HDFs | Reduced the senescent state and ameliorated skin aging | (Lee et al., 2020) |
| SHS‐coated MS Exos | / | Photoaging mice | Enhanced skin absorption of exosomes by creating microchannels; Reduced wrinkles and promoted extracellular matrix constituents | (Zhang et al., 2020) |
| BMSC Exos with aptamer | miR‐26a | Mice with OVX‐induced osteoporosis; Femur fracture mouse model | Promoted bone regeneration and accelerated bone healing; Enhanced bone targeting and bone generation | (Luo et al., 2019) |
| MSC Exos in silk fibroin hydrogel | miR‐675 | Aged animal model; H2O2‐treated H9C2 cells | Prolonged the half‐life of exosomes in vivo; Reduced aging hallmarks and promoted blood perfusion | (Han et al., 2019b) |
| ACE2‐primed endothelial progenitor cell Exos | ACE2 miR‐18a | Aging ECs | Protected ECs from hypoxia/reoxygenation‐induced injury through downregulation of Nox2/ROS | (Zhang et al., 2018a) |
| LPS‐preconditioned BMSC Exos | / | MI mouse model | Increased M2 macrophage polarization upon LPS stimulation; Attenuated inflammation and cardiomyocyte apoptosis | (Xu et al., 2019) |
| Cytokine‐preconditioned MSC Exos | / | APP/PS1 AD mice | Reduced activation of microglia and increased the dendritic spine density; Induced immunomodulatory and neuroprotective effects | (Losurdo et al., 2020) |
| Predifferentiated MSC Exos in 3D‐printed titanium scaffolds | / | BMSCs | Induced osteogenesis and regenerated bone tissue through the PI3K/Akt and MAPK pathways | (Zhai et al., 2020) |
| Other cell‐derived therapeutics | ||||
| Wnt4‐transgenic TECs | Wnt4; miR‐27b | TECs with steroid‐induced aging | Counteracted thymic adipose involution; Reduced age‐related thymic dysfunction | (Banfai et al., 2019) |
| Endothelial cells | / | Senescent fibroblasts; Diabetic mice | Reduced senescent phenotypes through YAP translocation and the PI3K/Akt pathway and promoted wound healing in diabetic mice | (Wei et al., 2020b) |
| 3D human dermal fibroblasts | TIMP‐1 | Photoaging mice | Induced collagen synthesis and antiaging effects | (Hu et al., 2019) |
| Young donor‐derived therapeutics | ||||
| Young mouse serum | eNAMPT | Aged mice | Enhanced NAD+ biosynthesis and controlled oxidative stress; Enhanced physical activity and extended lifespan | (Yoshida et al., 2019) |
| Primary fibroblasts of young human donors | GSTM2 | HGPS fibroblasts; Aged mice | Reduced senescent phenotypes by restoring antioxidant capacity; Prevented lipid peroxidation | (Fafia, Rodriguez‐Navarron‐Labora et al., 2020) |
| Young human serum | / | DOX‐treated cardiomyocytes | Retarded cellular senescence through the miR‐34a/PNUTS pathway | (Liu et al., 2019b) |
| Young mouse serum | miRNAs | Aged mice | Rejuvenated thymic aging and enhanced thymic negative selection | (Wang et al., 2018) |
| Young mouse serum | miRNAs | Aged mice | Reduced aging gene expression and induced telomerase gene expression | (Lee et al., 2018) |
| Centenarian donor‐derived therapeutics | ||||
| Centenarian fibroblasts | RNAseH2C enzyme | Young HDFs | Reduced inflammatory cytokines and upregulated anti‐inflammatory enzymes; Induced M2 polarization and genomic stability | (Storci et al., 2019) |
| Modified nanomedicines | ||||
| Catalase‐loaded exosomes | Catalase | PD mice | Prolonged circulation time and induced neuroprotective and antioxidant effects | (Haney et al., 2015) |
Stem cell‐derived therapeutics
UV radiation is an extrinsic factor of skin photoaging that disturbs human dermal fibroblasts (HDFs) via ROS production and extracellular matrix degradation (Xu et al., 2020). Pretreatment and posttreatment with EVs from human umbilical cord mesenchymal stem cells (MSCs), adipose‐derived stem cells (ADSCs), and induced pluripotent stem cells (iPSCs) are linked to a broad range of antiphotoaging functions, including fibroblast proliferation, collagen biosynthesis, DNA repair and UV protection (Choi et al., 2019; Deng et al., 2020; Oh et al., 2018b; Xu et al., 2020). Because of their antioxidant proteins, such as glutathione peroxidase 1, superoxide dismutase and catalase, EVs from the aforementioned cell sources are well suited to control the mitochondrial membrane and clear accumulated ROS (Deng et al., 2020; Xu et al., 2020). Human iPSCs show stronger antiphotoaging abilities than mesenchymal stem cells, with 16‐fold more EV production (Liu et al., 2019a).
However, in wound healing and inflammaging diseases, inhibition of a robust immune response is the critical mechanism of stem cell‐based EV treatment (Bian et al., 2020). After myocardial infarction or oxidant treatment, senescent cardiomyocytes are enriched during local inflammatory responses (Deng et al., 2019; Zhu et al., 2019). The exosomal lncRNA MALAT1 derived from MSCs inhibits the NF‐κB/TNFα pathway to rescue myocytes from aging and increases the left ventricular ejection fraction and fractional shortening (Zhu et al., 2019). ADSC exosome treatment activates S1P/SK1/S1PR1 signaling and promotes macrophage M2 polarization to mitigate cardiac damage (Deng et al., 2019). ADSC exosomes can also downregulate inflammatory cytokines and PGE2 expression in OA osteoblasts (Tofino‐Vian et al., 2017). Systemic inflammaging is associated with the loss of hypothalamic stem cells with increased age. Core treatment using exosomal miRNAs from hypothalamic stem cells can inhibit NF‐κB function and restore GnRH secretion, consequently reducing physiological deficits and extending lifespan (Meyer & Yankner, 2017; Zhang et al., 2017).
EVs from stem cells also inherit the ability to rescue the stemness of target stem cells. Human embryonic stem cell‐derived EVs can alleviate the senescence of tissue‐resident stem cells, such as BMSCs, to prevent bone loss with aging (Gong et al., 2020). MSCs can also rescue themselves from senescence and improve stemness through miR‐302b and HIF‐1 activation (Mas‐Bargues et al., 2020). To treat neurodegenerative diseases, EVs obtained from stem cells mainly transfer miRNAs to promote lysosome resumption and cytotoxic protein degradation (Elia et al., 2019; Hu et al., 2020). Interestingly, EVs noninvasively harvested from urine‐derived stem cells contain collagen triple‐helix repeat‐containing 1 (CTHRC1) and osteoprotegerin (OPG), which effectively reduce age‐related bone loss (Chen et al., 2019b). Treatment with these EVs is another regenerative therapy for osteoporosis patients who have unlimited access to the key treatment component.
Young donor‐derived therapeutics
In bone marrow transplantation, donor age is an essential concern because of engraftment failure associated with hematopoietic stem cell (HSC) senescence. Nonetheless, young donor‐derived vesicles can rejuvenate aged HSCs to improve autologous transplantation (Kulkarni et al., 2018). EVs from young donor serum contain high levels of nicotinamide phosphoribosyltransferase (eNAMPT) and glutathione‐related protein (GSTM2), both of which play roles in antioxidation, and their levels decline with age. NAMPT enhances NAD+ biogenesis and has antiaging effects on the hypothalamus, pancreas, retinas, and hippocampus. GSTM2 enhances intrinsic glutathione‐S‐transferase activity to scavenge ROS and prevent lipid peroxidation (Fafian‐Labora et al., 2020b; Yoshida et al., 2019). With regard to anti‐inflammation, EVs from young mouse serum can rejuvenate the aging thymus and enhance central negative selection signals to reinforce central tolerance. Then, they can ameliorate peripheral and CNS self‐immune responses by passing through the blood‐brain barrier (Wang et al., 2018). miRNAs from young donor‐derived EVs can also reduce telomere shortening and DNA damage (Lee et al., 2018; Liu et al., 2019b).
Centenarian donor‐derived therapeutics
The aging microenvironment, which is faced with external and internal risk factors, is equipped with considerable antioxidant machinery to eliminate ROS in endothelial EVs and HUVEC EVs, which mainly contain antioxidant enzymes and peptides as well as NADPH‐synthesizing enzymes (Bodega et al., 2017, 2018). Given this adaptive ability, centenarian donor‐derived EVs may have more potent antioxidant agents to maintain organ and systemic balance. In fibroblasts of centenarians, EVs contain high levels of the enzyme RNAseH2C, which can restrain DNA damage‐induced inflammation and ameliorate age‐associated low‐grade chronic inflammation (Storci et al., 2019). This phenomenon also implies that the antioxidant machinery is another controllable node of pathological transformation in the aging microenvironment. To some degree, intervention with antioxidant elements can support microenvironment stability.
5.2.2. Modified/artificial sources of EVs
Although EVs purified from cells or the serum of healthy donors can directly exert profound effects for rejuvenation, there are still inefficiency issues with natural cell‐free therapy, including low production, weak targeting, short half‐lives, and fast clearance. To address these problems, modified and artificial EVs should be designed well to promote the sensitivity, specificity, and safety of antiaging therapy.
Increasing the yield of EVs from stem cells is the first step toward promoting antiaging sensitivity. A serial extrusion method in which human iPSCs are passed through membrane filters with diminishing pore sizes can generate more cell‐engineered nanovesicles, showing an excellent ability to reduce senescence hallmarks of naturally aging HDFs and ameliorate skin aging (Lee et al., 2020). In addition to quantity‐promoting methods, methods that produce high‐quality EVs are crucial and valuable for clinical applications. Coatings on implants can facilitate EV penetration through tight physiological barriers to enable the EVs to subsequently exhibit dynamic functions. A skin coating made from SHSs (from the spicules of the marine sponge Haliclona) and used with MSC‐derived exosomes enhances skin penetration by creating microchannels, exhibiting significant antiphotoaging effects with slight skin irritation (Zhang et al., 2020). Another coating is made with sulfated glycosaminoglycans (GAGs), which synergize with EVs to strengthen extracellular matrix formation and mineralization in age‐related bone loss (Schmidt et al., 2016). Preconditioned cells and EVs also have roles in quality improvement. MSCs osteogenically predifferentiated for 10–15 days can be efficiently harvested as sources of osteogenic exosomes, which induce bone tissue regeneration through osteogenic miRNAs after loading onto 3D‐printed titanium alloy scaffolds (Zhai et al., 2020). Cells cultured in vitro as 3D spheroids can also produce high‐quality EVs (Hu et al., 2019). Directly encapsulating antiaging medicines into EVs as artificial nanomedicines is a method for developing high‐affinity and high‐quality antiaging systems. For example, a new exosomal delivery system for catalase, an antioxidant, has visibly neuroprotective effects in PD after intranasal delivery (Haney et al., 2015).
Prolonging the half‐life of functional EVs would address sensitivity and specificity problems by preventing ectopic distribution and clearance. Age‐ and tissue‐specific miRNAs, such as miR‐675, mainly remain and function in muscle tissue; these miRNAs can be encapsulated in EVs and used to decorate silk fibroin hydrogel in order to increase the retention time in muscle cells and thereby cure ischemic diseases (Han et al., 2019b). Using these methods, the targeting problem can also be resolved. Through combination with specific aptamers, EVs containing miR‐26a from BMSCs can be directionally driven to bone marrow to accelerate bone healing after fracture (Luo et al., 2019).
For safety, side effects, primarily those involving excessive inflammation, should be minimized. After myocardial infarction, cardiomyocytes treated with EVs from lipopolysaccharide (LPS)‐preconditioned BMSCs can reduce apoptosis and inflammation by mediating macrophage M2 polarization (Xu et al., 2019). In AD, cytokine‐preconditioned MSCs produce immunomodulatory and neuroprotective EVs, achieving antiaging and anti‐inflammatory goals after intranasal delivery (Losurdo et al., 2020).
In conclusion, as burgeoning biomaterials for antiaging therapy, EVs play roles in treating age‐related diseases. However, questions related to their tumorigenicity and manoeuvrability should be addressed. With the diversity of EVs and their possible applications in nanomedicines, it is anticipated that more specific senolytic and senomorphic strategies can be developed that rely on EVs.
6. CONCLUSION AND PERSPECTIVES
Cellular senescence can be a potential launching point for various diseases, not only because of its association with organ/systemic aging but also because of its nature. As a relatively stable intermediate state between survival and death, senescence is associated with specific markers and cell morphological changes, which is convenient for qualitative and quantitative detection and evaluation (Hernandez‐Segura et al., 2018). There are significant differences and hidden weaknesses in chromatin structures, gene expression, and metabolic profiles of senescent cells compared with nonsenescent cells, which can be monitored by the immune system (Liu et al., 2020; Wang et al., 2019). Moreover, for tumours, the characteristics of cell cycle arrest can be leveraged for a physiological anticancer effect.
In this review, we have discussed the characteristics of the aging microenvironment for the first time, highlighting new ideas that might be developed for the treatment of age‐related diseases. On the one hand, in view of the instability of the aging microenvironment, prompt prevention and exclusion of interfering factors must be the focal points and be managed accordingly. On the other hand, considering the amplification effect of the aging microenvironment, inhibiting the secretion of harmful molecules and cutting off routes of aging transmission can buffer the adverse effects of the inflammatory microenvironment. In addition, in light of the transformative nature of the aging microenvironment, exploring the key mechanism of transformation and intervening in key events can effectively forestall and reverse the establishment and progression of pathological states.
Accordingly, EVs play undeniable roles in cellular senescence and age‐related diseases and can be used in promising approaches to identify and therapeutically target senescent cells. However, there are still several challenges associated with the development of this young but emerging field of research, including a lack of optimal and verified experimental models, details on the biogenetic mechanism, a comprehensive age‐related EV atlas and a rapid capture and visualization system. Given the long aging period of humans, fast‐aging model organisms, such as Caenorhabditis elegans and yeasts, and animals, such as naturally aging mice or gene‐edited mice, are widely used in geroscience. However, they do not faithfully recapitulate human states due to the complex multicell and multiorgan networks of senescence. Better‐suited animal models (e.g., nonhuman primates) or human organoids are required for research on the topics of EVs and aging. As secretion marks the onset of EV function, clarifying the detailed molecular mechanism of EV secretion may be helpful for elucidating the aging transmission pathway and developing efficient therapeutic interventions. In addition, given the heterogeneity of senescence and EVs, creation of a comprehensive atlas of age‐related EVs from different cells under different stresses would be enormously helpful for this field. It is possible that a specific ageotype may indicate an individual physiological and pathological status as a more distinctive biomarker than others now available. Senescence‐associated EVs can now be detected in blood, urine, CSF, and saliva, which provides us with new inspiration for diagnostic and prognostic tools using liquid biopsy (Machida et al., 2015; Perrotte et al., 2020; Santelli et al., 2019). Due to the prion‐like behaviour of EVs, in vivo imaging techniques have been designed, such as techniques involving EV labelling combined with CT that longitudinally and quantitatively trace aberrant neurons in the brains of PD, AD, and stroke patients (Perets et al., 2019). However, a full understanding of which cells are senescent and which cells secrete and take up aging EVs in vivo is lacking owing to the unsuitable and inaccurate capture and visualization systems.
To develop clinical antiaging therapies, safety should first be considered, followed by sensitivity and specificity. Because of the specificity of immune recognition, senolytic CAR‐T and senescent CD153 vaccines have been proven feasible and efficient (Amor et al., 2020; Yoshida et al., 2020). This finding suggests that immune‐modified EVs may be able to preferentially target senescent cells and tissues to promote senotherapy. However, before EV transplantation is performed, the routes of administration, reasonable dosages, dosing intervals, and host nutritional status should be strictly verified. EVs can be considered promising materials and tools for antiaging treatments after rigorous trials.
ACKNOWLEDGEMENTS
This work was supported by grants from the Ministry of Science and Technology of China (2020YFC2002800).
Yin, Y., Chen, H., Wang, Y., Zhang, L., & Wang, X. (2021). Roles of extracellular vesicles in the aging microenvironment and age‐related diseases. Journal of Extracellular Vesicles, 10,e12154. 10.1002/jev2.12154
REFERENCES
- Aarreberg, L. D., Esser‐Nobis, K., Driscoll, C., Shuvarikov, A., Roby, J. A., & Gale, M.Jr. (2019). Interleukin‐1beta induces mtDNA Release to activate innate immune signaling via cGAS‐STING. Molecular Cell, 74, 801–815 e806. 10.1016/j.molcel.2019.02.038 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Acosta, J. C., Banito, A., Wuestefeld, T., Georgilis, A., Janich, P., Morton, J. P., Athineos, D., Kang, T. W., Lasitschka, F., Andrulis, M., Pascual, G., Morris, K. J., Khan, S., Jin, H., Dharmalingam, G., Snijders, A. P., Carroll, T., Capper, D., Pritchard, C., … Gil J . (2013). A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nature Cell Biology, 15, 978–990; 10.1038/ncb2784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Adamiak, M., Cheng, G., Bobis‐Wozowicz, S., Zhao, L., Kedracka‐Krok, S., Samanta, A., Karnas, E., Xuan, Y. T., Skupien‐Rabian, B., Chen, X., Jankowska, U., Girgis, M., Sekula, M., Davani, A., Lasota, S., Vincent, R. J., Sarna, M., Newell, K. L., Wang, Ou‐Li, … Zuba‐Surma, E. K. (2018). Induced Pluripotent Stem Cell (iPSC)‐derived extracellular vesicles are safer and more effective for cardiac repair than iPSCs. Circulation Research, 122, 296–309. 10.1161/CIRCRESAHA.117.311769 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahadi, S., Zhou, W., Schussler‐Fiorenza Rose, S. M., Sailani, M. R., Contrepois, K., Avina, M., Ashland, M., Brunet, A., & Snyder, M. (2020). Personal aging markers and ageotypes revealed by deep longitudinal profiling. Nature Medicine, 26, 83–90. 10.1038/s41591-019-0719-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al Suraih, M. S., Trussoni, C. E., Splinter, P. L., LaRusso, N. F., & O'Hara, S. P. (2020). Senescent cholangiocytes release extracellular vesicles that alter target cell phenotype via the epidermal growth factor receptor. Liver International, 40(10), 2455–2468. 10.1111/liv.14569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Al‐Mayah, A. H., Bright, S. J., Bowler, D. A., Slijepcevic, P., Goodwin, E., & Kadhim, M. A. (2017). Exosome‐mediated telomere instability in human breast epithelial cancer cells after x irradiation. Radiation Research, 187, 98–106. 10.1667/RR14201.1 [DOI] [PubMed] [Google Scholar]
- Alberro, A., Saenz‐Cuesta, M., Munoz‐Culla, M., Mateo‐Abad, M., Gonzalez, E., Carrasco‐Garcia, E., Arauzo‐Bravo, M. J., Matheu, A., Vergara, I., & Otaegui, D. (2016). Inflammaging and frailty status do not result in an increased extracellular vesicle concentration in circulation. International Journal of Molecular Science, 17, 1168 10.3390/ijms17071168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alimbetov, D., Davis, T., Brook, A. J., Cox, L. S., Faragher, R. G., Nurgozhin, T., Zhumadilov, Z., & Kipling, D. (2016). Suppression of the senescence‐associated secretory phenotype (SASP) in human fibroblasts using small molecule inhibitors of p38 MAP kinase and MK2. Biogerontology, 17, 305–315. 10.1007/s10522-015-9610-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alique, M., Ruiz‐Torres, M. P., Bodega, G., Noci, M. V., Troyano, N., Bohorquez, L., Luna, C., Luque, R., Carmona, A., Carracedo, J., & Ramirez, R. (2017). Microvesicles from the plasma of elderly subjects and from senescent endothelial cells promote vascular calcification. Aging (Albany NY), 9, 778–789. 10.18632/aging.101191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amor, C., Feucht, J., Leibold, J., Ho, Y. J., Zhu, C., Alonso‐Curbelo, D., Mansilla‐Soto, J., Boyer, J. A., Li, X., Giavridis, T., Kulick, A., Houlihan, S., Peerschke, E., Friedman, S. L., Ponomarev, V., Piersigilli, A., Sadelain, M., & Lowe, S. W. (2020). Senolytic CAR T cells reverse senescence‐associated pathologies. Nature, 583, 127–132. 10.1038/s41586-020-2403-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asai, H., Ikezu, S., Tsunoda, S., Medalla, M., Luebke, J., Haydar, T., Wolozin, B., Butovsky, O., Kugler, S., & Ikezu, T. (2015). Depletion of microglia and inhibition of exosome synthesis halt tau propagation. Nature Neuroscience, 18, 1584–1593. 10.1038/nn.4132 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aulston, B., Liu, Q., Mante, M., Florio, J., Rissman, R. A., & Yuan, S. H. (2019). Extracellular vesicles isolated from familial Alzheimer's disease neuronal cultures induce aberrant tau phosphorylation in the wild‐type mouse brain. Journal of Alzheimers Disease, 72, 575–585. 10.3233/JAD-190656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azarm, K., Bhardwaj, A., Kim, E., & Smith, S. (2020). Persistent telomere cohesion protects aged cells from premature senescence. Nature Communications, 11, 3321. 10.1038/s41467-020-17133-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azzalin, C. M., & Lingner, J. (2015). Telomere functions grounding on TERRA firma. Trends in Cell Biology, 25, 29–36. 10.1016/j.tcb.2014.08.007 [DOI] [PubMed] [Google Scholar]
- Baar, M. P., Brandt, R. M. C., Putavet, D. A., Klein, J. D. D., Derks, K. W. J., Bourgeois, B. R. M., Stryeck, S., Rijksen, Y., van Willigenburg, H., Feijtel, D. A., Pluijm, I., Essers, J., Cappellen, W. A., IJcken, W. F., Houtsmuller, A. B., Pothof, J., Bruin, R. W. F., Madl, T., Hoeijmakers, J. H. J. … Keizer, P. L. J. (2017). Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell, 169, 132–147 e116. 10.1016/j.cell.2017.02.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bai, J., Wang, Y., Wang, J., Zhai, J., He, F., & Zhu, G. (2020). Irradiation‐induced senescence of bone marrow mesenchymal stem cells aggravates osteogenic differentiation dysfunction via paracrine signaling. American Journal of Physiology: Cell Physiology, 318, C1005–C1017. 10.1152/ajpcell.00520.2019 [DOI] [PubMed] [Google Scholar]
- Baker, D. J., Wijshake, T., Tchkonia, T., LeBrasseur, N. K., Childs, B. G., van de Sluis, B., Kirkland, J. L., & van Deursen, J. M. (2011). Clearance of p16Ink4a‐positive senescent cells delays ageing‐associated disorders. Nature, 479, 232–236. 10.1038/nature10600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baker, N., Boyette, L. B., & Tuan, R. S. (2015). Characterization of bone marrow‐derived mesenchymal stem cells in aging. Bone (New York), 70, 37–47. 10.1016/j.bone.2014.10.014 [DOI] [PubMed] [Google Scholar]
- Banfai, K., Garai, K., Ernszt, D., Pongracz, J. E., & Kvell, K. (2019). Transgenic exosomes for thymus regeneration. Frontiers in Immunology, 10, 862. 10.3389/fimmu.2019.00862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bang, C., Batkai, S., Dangwal, S., Gupta, S. K., Foinquinos, A., Holzmann, A., Just, A., Remke, J., Zimmer, K., Zeug, A., Ponimaskin, E., Schmiedl, A., Yin, X., Mayr, M., Halder, R., Fischer, A., Engelhardt, S., Wei, Y., Schober, A., … Thum, T. (2014). Cardiac fibroblast‐derived microRNA passenger strand‐enriched exosomes mediate cardiomyocyte hypertrophy. Journal of Clinical Investigation, 124, 2136–2146. 10.1172/JCI70577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barzilai, N., Crandall, J. P., Kritchevsky, S. B., & Espeland, M. A. (2016). Metformin as a tool to target aging. Cell Metabolism, 23, 1060–1065. 10.1016/j.cmet.2016.05.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basisty, N., Kale, A., Jeon, O. H., Kuehnemann, C., Payne, T., Rao, C., Holtz, A., Shah, S., Sharma, V., Ferrucci, L., Campisi, J., & Schilling, B. (2020). A proteomic atlas of senescence‐associated secretomes for aging biomarker development. PLoS Biology, 18, e3000599. 10.1371/journal.pbio.3000599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Basu, A. (2021). The interplay between apoptosis and cellular senescence: Bcl‐2 family proteins as targets for cancer therapy. Pharmacology Therapy, 107943. 10.1016/j.pharmthera.2021.107943 [DOI] [PubMed] [Google Scholar]
- Battaglia, R., Musumeci, P., Ragusa, M., Barbagallo, D., Scalia, M., Zimbone, M., Lo Faro, J. M., Borzi, P., Scollo, P., Purrello, M., Vento, E. M., & Di Pietro, C. (2020). Ovarian aging increases small extracellular vesicle CD81(+) release in human follicular fluid and influences miRNA profiles. Aging (Albany NY), 12, 12324–12341. 10.18632/aging.103441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berezin, A. E., Kremzer, A. A., Samura, T. A., Berezina, T. A., & Kruzliak, P. (2015). Impaired immune phenotype of circulating endothelial‐derived microparticles in patients with metabolic syndrome and diabetes mellitus. Journal of Endocrinological Investigation, 38, 865–874. 10.1007/s40618-015-0273-z [DOI] [PubMed] [Google Scholar]
- Bi, S., Liu, Z., Wu, Z., Wang, Z., Liu, X., Wang, S., Ren, J., Yao, Y., Zhang, W., Song, M., Liu, G.‐H., & Qu, J. (2020). SIRT7 antagonizes human stem cell aging as a heterochromatin stabilizer. Protein Cell, 11, 483–504. 10.1007/s13238-020-00728-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bian, X., Li, B., Yang, J., Ma, K., Sun, M., Zhang, C., & Fu, X. (2020). Regenerative and protective effects of dMSC‐sEVs on high‐glucose‐induced senescent fibroblasts by suppressing RAGE pathway and activating Smad pathway. Stem Cell Reserach Therapy, 11(1), 166, 10.1186/s13287-020-01681-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bodega, G., Alique, M., Bohorquez, L., Ciordia, S., Mena, M. C., & Ramirez, M. R. (2017). The antioxidant machinery of young and senescent human umbilical vein endothelial cells and their microvesicles. Oxidative Medicine and Cellular Longevity, 2017, 7094781. 10.1155/2017/7094781 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bodega, G., Alique, M., Bohorquez, L., Moran, M., Magro, L., Puebla, L., Ciordia, S., Mena, M. C., Arza, E., & Ramirez, M. R. (2018). Young and especially senescent endothelial microvesicles produce NADPH: The fuel for their antioxidant machinery. Oxidative Medicine and Cellular Longevity, 2018, 3183794. 10.1155/2018/3183794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boregowda, S. V., Ghoshal, S., Booker, C. N., Krishnappa, V., Chakraborty, A., & Phinney, D. G. (2017). IP6K1 reduces mesenchymal stem/stromal cell fitness and potentiates high fat diet‐induced skeletal involution. Stem Cells, 35, 1973–1983. 10.1002/stem.2645 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borghesan, M., Fafian‐Labora, J., Eleftheriadou, O., Carpintero‐Fernandez, P., Paez‐Ribes, M., Vizcay‐Barrena, G., Swisa, A., Kolodkin‐Gal, D., Ximenez‐Embun, P., Lowe, R., Martín‐Martín, B., Peinado, H., Muñoz, J., Fleck, R. A., Dor, Y., Ben‐Porath, I., Vossenkamper, A., Muñoz‐Espin, D., & O'Loghlen, A. (2019). Small extracellular vesicles are key regulators of non‐cell autonomous intercellular communication in senescence via the interferon protein IFITM3. Cell Reports, 27, 3956–3971 e3956. 10.1016/j.celrep.2019.05.095 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borras, C., Mas‐Bargues, C., Sanz‐Ros, J., Roman‐Dominguez, A., Gimeno‐Mallench, L., Ingles, M., Gambini, J., & Vina, J. (2020). Extracellular vesicles and redox modulation in aging. Free Radical Biology and Medicine, 149, 44–50. 10.1016/j.freeradbiomed.2019.11.032 [DOI] [PubMed] [Google Scholar]
- Buratta, S., Urbanelli, L., Sagini, K., Giovagnoli, S., Caponi, S., Fioretto, D., Mitro, N., Caruso, D., & Emiliani, C. (2017). Extracellular vesicles released by fibroblasts undergoing H‐Ras induced senescence show changes in lipid profile. Plos One, 12, e0188840. 10.1371/journal.pone.0188840 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cabreiro, F., Au, C., Leung, K. Y., Vergara‐Irigaray, N., Cocheme, H. M., Noori, T., Weinkove, D., Schuster, E., Greene, N. D., & Gems, D. (2013). Metformin retards aging in C. elegans by altering microbial folate and methionine metabolism. Cell, 153, 228–239. 10.1016/j.cell.2013.02.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calcinotto, A., Kohli, J., Zagato, E., Pellegrini, L., Demaria, M., & Alimonti, A. (2019). Cellular senescence: Aging, cancer, and injury. Mitochondrial Membrane Permeabilization in Cell Death, 99, 1047–1078. 10.1152/physrev.00020.2018 [DOI] [PubMed] [Google Scholar]
- Callender, L. A., Carroll, E. C., Beal, R. W. J., Chambers, E. S., Nourshargh, S., Akbar, A. N., & Henson, S. M. (2018). Human CD8(+) EMRA T cells display a senescence‐associated secretory phenotype regulated by p38 MAPK. Aging Cell, 17(1), e12675, 10.1111/acel.12675 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Callender, L. A., Carroll, E. C., Bober, E. A., Akbar, A. N., Solito, E., & Henson, S. M. (2020). Mitochondrial mass governs the extent of human T cell senescence. Aging Cell, 19, e13067. 10.1111/acel.13067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao, Z., Wu, Y., Liu, G., Jiang, Y., Wang, X., Wang, Z., & Feng, T. (2019). alpha‐Synuclein in salivary extracellular vesicles as a potential biomarker of Parkinson's disease. Neuroscience Letters 696, 114–120. 10.1016/j.neulet.2018.12.030 [DOI] [PubMed] [Google Scholar]
- Capell, B. C., Drake, A. M., Zhu, J., Shah, P. P., Dou, Z., Dorsey, J., Simola, D. F., Donahue, G., Sammons, M., Rai, T. S., Natale, C., Ridky, T. W., Adams, P. D., & Berger, S. L. (2016). MLL1 is essential for the senescence‐associated secretory phenotype. Genes & Development, 30, 321–336. 10.1101/gad.271882.115 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carracedo, J., Alique, M., Ramirez‐Carracedo, R., Bodega, G., and Ramirez, R. (2019). Endothelial extracellular vesicles produced by senescent cells: pathophysiological role in the cardiovascular disease associated with all types of diabetes mellitus. Current Vascular Pharmacology, 17, 447–454. 10.2174/1570161116666180820115726 [DOI] [PubMed] [Google Scholar]
- Catanzaro, J. M., Sheshadri, N., Pan, J. A., Sun, Y., Shi, C., Li, J., Powers, R. S., Crawford, H. C., & Zong, W. X. (2014). Oncogenic Ras induces inflammatory cytokine production by upregulating the squamous cell carcinoma antigens SerpinB3/B4. Nature Communications, 5, 3729. 10.1038/ncomms4729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chanda, D., Otoupalova, E., Hough, K. P., Locy, M. L., Bernard, K., Deshane, J. S., Sanderson, R. D., Mobley, J. A., & Thannickal, V. J. (2019). Fibronectin on the surface of extracellular vesicles mediates fibroblast invasion. The American Journal of Respiratory Cell and Molecular Biology, 60, 279–288. 10.1165/rcmb.2018-0062OC [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen, B., Sun, Y., Zhang, J., Zhu, Q., Yang, Y., Niu, X., Deng, Z., Li, Q., & Wang, Y. (2019a). Human embryonic stem cell‐derived exosomes promote pressure ulcer healing in aged mice by rejuvenating senescent endothelial cells. Stem Cell Research Therapy, 10, 142. 10.1186/s13287-019-1253-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen, C. Y., Rao, S. S., Tan, Y. J., Luo, M. J., Hu, X. K., Yin, H., Huang, J., Hu, Y., Luo, Z. W., Liu, Z. Z., Wang, Z. ‐. X., Cao, J., Liu, Yi‐W., Li, H. ‐. M., Chen, Y., Du, W., Liu, J. ‐. H., Zhang, Y., Chen, T. ‐. H., … Xie, H. (2019b). Extracellular vesicles from human urine‐derived stem cells prevent osteoporosis by transferring CTHRC1 and OPG. Bone Research, 7, 18. 10.1038/s41413-019-0056-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen, J., Crutchley, J., Zhang, D., Owzar, K., & Kastan, M. B. (2017). Identification of a DNA damage‐induced alternative splicing pathway that regulates p53 and cellular senescence markers. Cancer Discovery, 7, 766–781. 10.1158/2159-8290.CD-16-0908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen, Y., Pu, Q., Ma, Y., Zhang, H., Ye, T., Zhao, C., Huang, X., Ren, Y., Qiao, L., Liu, H. ‐ M., Esmon, C. T., Ding, B. ‐ S., & Cao, Z. (2020). Aging reprograms the hematopoietic‐vascular niche to impede regeneration and promote fibrosis. Cell Metabolism, 33, 395–410. 10.1016/j.cmet.2020.11.019 [DOI] [PubMed] [Google Scholar]
- Cheng, L., Vella, L. J., Barnham, K. J., McLean, C., Masters, C. L., & Hill, A. F. (2020). Small RNA fingerprinting of Alzheimer's disease frontal cortex extracellular vesicles and their comparison with peripheral extracellular vesicles. Journal of Extracellular Vesicles, 9, 1766822. 10.1080/20013078.2020.1766822 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi, J. S., Cho, W. L., Choi, Y. J., Kim, J. D., Park, H. A., Kim, S. Y., Park, J. H., Jo, D. G., & Cho, Y. W. (2019). Functional recovery in photo‐damaged human dermal fibroblasts by human adipose‐derived stem cell extracellular vesicles. Journal of Extracellular Vesicles, 8, 1565885. 10.1080/20013078.2019.1565885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung, K. M., Hernandez, N., Sproul, A. A., & Yu, W. H. (2019). Alzheimer's disease and the autophagic‐lysosomal system. Neuroscience Letters, 697, 49–58. 10.1016/j.neulet.2018.05.017 [DOI] [PubMed] [Google Scholar]
- Cicognola, C., Brinkmalm, G., Wahlgren, J., Portelius, E., Gobom, J., Cullen, N. C., Hansson, O., Parnetti, L., Constantinescu, R., Wildsmith, K., Chen, H. ‐. H., Beach, T. G., Lashley, T., Zetterberg, H., Blennow, K., & Höglund, K. (2019). Novel tau fragments in cerebrospinal fluid: Relation to tangle pathology and cognitive decline in Alzheimer's disease. Acta Neuropathologica, 137, 279–296. 10.1007/s00401-018-1948-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Correia‐Melo, C., Marques, F. D., Anderson, R., Hewitt, G., Hewitt, R., Cole, J., Carroll, B. M., Miwa, S., Birch, J., Merz, A., Rushton, M. D., Charles, M., Jurk, D., Tait, S. W. G., Czapiewski, R., Greaves, L., Nelson, G., Bohlooly‐Y, M., Rodriguez‐Cuenca, S., … Passos, J. F. (2016). Mitochondria are required for pro‐ageing features of the senescent phenotype. EMBO Journal, 35, 724–742. 10.15252/embj.201592862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crotti, A., Sait, H. R., McAvoy, K. M., Estrada, K., Ergun, A., Szak, S., Marsh, G., Jandreski, L., Peterson, M., Reynolds, T. L., Dalkilic‐LiddleI.,Cameron, A., Cahir‐McFarland, E., & Ransohoff, R. M. (2019). BIN1 favors the spreading of Tau via extracellular vesicles. Scientific Reports, 9, 9477. 10.1038/s41598-019-45676-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cusanelli, E., Romero, C. A., & Chartrand, P. (2013). Telomeric noncoding RNA TERRA is induced by telomere shortening to nucleate telomerase molecules at short telomeres. Molecular Cell, 51, 780–791. 10.1016/j.molcel.2013.08.029 [DOI] [PubMed] [Google Scholar]
- da Silveira, J. C., Winger, Q. A., Bouma, G. J., & Carnevale, E. M. (2015). Effects of age on follicular fluid exosomal microRNAs and granulosa cell transforming growth factor‐beta signalling during follicle development in the mare. Reproduction, Fertility, and Development, 27, 897–905. 10.1071/RD14452 [DOI] [PubMed] [Google Scholar]
- Datta, R., Bansal, T., Rana, S., Datta, K., Datta Chaudhuri, R., Chawla‐Sarkar, M., & Sarkar, S. (2017). Myocyte‐derived Hsp90 modulates collagen upregulation via biphasic activation of STAT‐3 in fibroblasts during cardiac hypertrophy. Molecular Cell Biology, 37, e00611–e00616. 10.1128/MCB.00611-16 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davalos, A. R., Kawahara, M., Malhotra, G. K., Schaum, N., Huang, J., Ved, U., Beausejour, C. M., Coppe, J. P., Rodier, F., & Campisi, J. (2013). p53‐dependent release of Alarmin HMGB1 is a central mediator of senescent phenotypes. Journal of Cell Biology, 201, 613–629. 10.1083/jcb.201206006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis, C., Dukes, A., Drewry, M., Helwa, I., Johnson, M. H., Isales, C. M., Hill, W. D., Liu, Y., Shi, X., Fulzele, S., & Hamrick, M. W. (2017). MicroRNA‐183‐5p increases with age in bone‐derived extracellular vesicles, suppresses bone marrow stromal (stem) cell proliferation, and induces stem cell senescence. Tissue Engineering Part A, 23, 1231–1240. 10.1089/ten.TEA.2016.0525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de la Cruz Lopez, K. G., Toledo Guzman, M. E., Sanchez, E. O., & Garcia Carranca, A. (2019). mTORC1 as a regulator of mitochondrial functions and a therapeutic target in cancer. Frontiers in Oncology, 9, 1373. 10.3389/fonc.2019.01373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng, M., Yu, Z., Li, D., Wang, X., Zhou, G., Liu, W., Cao, Y., Xia, W., Li, W., & Zhang, J. W. (2020). Human umbilical cord mesenchymal stem cell‐derived and dermal fibroblast‐derived extracellular vesicles protect dermal fibroblasts from ultraviolet radiation‐induced photoaging in vitro. Photochemistry and Photobiology Science, 19, 406–414. 10.1039/c9pp00421a [DOI] [PubMed] [Google Scholar]
- Deng, S., Zhou, X., Ge, Z., Song, Y., Wang, H., Liu, X., & Zhang, D. (2019). Exosomes from adipose‐derived mesenchymal stem cells ameliorate cardiac damage after myocardial infarction by activating S1P/SK1/S1PR1 signaling and promoting macrophage M2 polarization. International Journal of Biochemistry and Cell Biology, 114, 105564. 10.1016/j.biocel.2019.105564 [DOI] [PubMed] [Google Scholar]
- Denu, R. A., & Hematti, P. (2016). Effects of oxidative stress on mesenchymal stem cell biology. Oxidative Medicine and Cellular Longevity, 2016, 2989076. 10.1155/2016/2989076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desdin‐Mico, G., Soto‐Heredero, G., Aranda, J. F., Oller, J., Carrasco, E., Gabande‐Rodriguez, E., Blanco, E. M., Alfranca, A., Cusso, L., Desco, M., Ibañez, B., Gortazar, A. R., Fernández‐Marcos, P., Gortazar, A. R., Fernández‐Marcos, P., Navarro, M. N., Hernaez, B., Alcamí, A., Baixauli, F. & Mittelbrunn, M. (2020). T cells with dysfunctional mitochondria induce multimorbidity and premature senescence. Science, 368, 1371–1376. 10.1126/science.aax0860 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Micco, R., Krizhanovsky, V., Baker, D., & d'Adda di Fagagna, F. (2021). Cellular senescence in ageing: From mechanisms to therapeutic opportunities. Nature Reviews Molecular Cell Biology, 22, 75–95. 10.1038/s41580-020-00314-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diez‐Fraile, A., Lammens, T., Tilleman, K., Witkowski, W., Verhasselt, B., De Sutter, P., Benoit, Y., Espeel, M., & D'Herde, K. (2014). Age‐associated differential microRNA levels in human follicular fluid reveal pathways potentially determining fertility and success of in vitro fertilization. Human Fertility (Cambridge, England), 17, 90–98. 10.3109/14647273.2014.897006 [DOI] [PubMed] [Google Scholar]
- Ding, C., Qian, C., Hou, S., Lu, J., Zou, Q., Li, H., & Huang, B. (2020). Exosomal miRNA‐320a is released from hAMSCs and regulates SIRT4 to prevent reactive oxygen species generation in POI. Molecular Therapy Nucleic Acids, 21, 37–50. 10.1016/j.omtn.2020.05.013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Domenis, R., Zanutel, R., Caponnetto, F., Toffoletto, B., Cifu, A., Pistis, C., Di Benedetto, P., Causero, A., Pozzi, M., Bassini, F., Fabris, M., Niazi, K. R., Soon‐Shiong, P., & Curcio, F. (2017). Characterization of the proinflammatory profile of synovial fluid‐derived exosomes of patients with osteoarthritis. Mediators of Inflammation, 2017, 4814987. 10.1155/2017/4814987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elia, C. A., Tamborini, M., Rasile, M., Desiato, G., Marchetti, S., Swuec, P., Mazzitelli, S., Clemente, F., Anselmo, A., Matteoli, M., Malosio, M. L., & Coco, S. (2019). Intracerebral injection of extracellular vesicles from mesenchymal stem cells exerts reduced abeta plaque burden in early stages of a preclinical model of Alzheimer's disease. Cells, 8, 1059. 10.3390/cells8091059 [DOI] [PMC free article] [PubMed] [Google Scholar]
- En, A., Takauji, Y., Ayusawa, D., & Fujii, M. (2020). The role of lamin B receptor in the regulation of senescence‐associated secretory phenotype (SASP). Experimental Cell Research, 390, 111927. 10.1016/j.yexcr.2020.111927 [DOI] [PubMed] [Google Scholar]
- Espada, L., Dakhovnik, A., Chaudhari, P., Martirosyan, A., Miek, L., Poliezhaieva, T., Schaub, Y., Nair, A., Doring, N., Rahnis, N., Werz, O., Koeberle, A., Kirkpatrick, J., Ori, A., & Ermolaeva, M. A. (2020). Loss of metabolic plasticity underlies metformin toxicity in aged Caenorhabditis elegans. Nature Metabolism, 2, 1316–1331. 10.1038/s42255-020-00307-1 [DOI] [PubMed] [Google Scholar]
- Fafian‐Labora, J., Morente‐Lopez, M., Sanchez‐Dopico, M. J., Arntz, O. J., van de Loo, F. A. J., De Toro, J., & Arufe, M. C. (2020a). Influence of mesenchymal stem cell‐derived extracellular vesicles in vitro and their role in ageing. Stem Cell Research Therapy, 11, 13. 10.1186/s13287-019-1534-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fafian‐Labora, J. A., Rodriguez‐Navarro, J. A., & O'Loghlen, A. (2020b). Small extracellular vesicles have GST activity and ameliorate senescence‐related tissue damage. Cell Metabolism, 32, 71–86 e75. 10.1016/j.cmet.2020.06.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fajemiroye, J. O., da Cunha, L. C., Saavedra‐Rodriguez, R., Rodrigues, K. L., Naves, L. M., Mourao, A. A., da Silva, E. F., Williams, N. E. E., Martins, J. L. R., Sousa, R. B., Rebelo, A. C. S., Reis, A. A. S., Santos, R. S., Ferreira‐Neto, M. L., & Pedrino, G. R. (2018). Aging‐induced biological changes and cardiovascular diseases. BioMed Research International, 2018, 7156435. 10.1155/2018/7156435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fane, M., & Weeraratna, A. T. (2020). How the ageing microenvironment influences tumour progression. Nature Reviews Cancer, 20, 89–106. 10.1038/s41568-019-0222-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng, D., Zhao, W. L., Ye, Y. Y., Bai, X. C., Liu, R. Q., Chang, L. F., Zhou, Q., & Sui, S. F. (2010). Cellular internalization of exosomes occurs through phagocytosis. Traffic (Copenhagen, Denmark), 11, 675–687. 10.1111/j.1600-0854.2010.01041.x [DOI] [PubMed] [Google Scholar]
- Fernandes, A., Ribeiro, A. R., Monteiro, M., Garcia, G., Vaz, A. R., & Brites, D. (2018). Secretome from SH‐SY5Y APPSwe cells trigger time‐dependent CHME3 microglia activation phenotypes, ultimately leading to miR‐21 exosome shuttling. Biochimie, 155, 67–82. 10.1016/j.biochi.2018.05.015 [DOI] [PubMed] [Google Scholar]
- Fibach, E. (2021). The redox balance and membrane shedding in RBC production, maturation, and senescence. Frontiers in Physiology, 12, 604738. 10.3389/fphys.2021.604738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freeman, D. W., Noren Hooten, N., Eitan, E., Green, J., Mode, N. A., Bodogai, M., Zhang, Y., Lehrmann, E., Zonderman, A. B., Biragyn, A., Egan, J., Becker, K. G., Mattson, M. P., Ejiogu, N., & Evans, M. K. (2018). Altered extracellular vesicle concentration, cargo, and function in diabetes. Diabetes, 67, 2377–2388. 10.2337/db17-1308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freund, A., Patil, C. K., & Campisi, J. (2011). p38MAPK is a novel DNA damage response‐independent regulator of the senescence‐associated secretory phenotype. EMBO Journal, 30, 1536–1548. 10.1038/emboj.2011.69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu, Q., Jiang, H., Wang, Z., Wang, X., Chen, H., Shen, Z., Xiao, L., Guo, X., & Yang, T. (2018). Injury factors alter miRNAs profiles of exosomes derived from islets and circulation. Aging (Albany NY), 10, 3986–3999. 10.18632/aging.101689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuhrmann‐Stroissnigg, H., Ling, Y. Y., Zhao, J., McGowan, S. J., Zhu, Y., Brooks, R. W., Grassi, D., Gregg, S. Q., Stripay, J. L., Dorronsoro, A., Corbo, L., Tang, P., Bukata, C., Ring, N., Giacca, M., Li, X., Tchkonia, T., Kirkland, J. L., Niedernhofer, L. J., & Robbins, P. D. (2017). Identification of HSP90 inhibitors as a novel class of senolytics. Nature Communications, 8, 422. 10.1038/s41467-017-00314-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fulzele, S., Mendhe, B., Khayrullin, A., Johnson, M., Kaiser, H., Liu, Y., Isales, C. M., and Hamrick, M. W. (2019). Muscle‐derived miR‐34a increases with age in circulating extracellular vesicles and induces senescence of bone marrow stem cells. Aging (Albany NY), 11, 1791–1803. 10.18632/aging.101874 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Georgilis, A., Klotz, S., Hanley, C. J., Herranz, N., Weirich, B., Morancho, B., Leote, A. C., D'Artista, L., Gallage, S., Seehawer, M., Carroll, T., Dharmalingam, G., Wee, K. B., Mellone, M., Pombo, J., Heide, D., Guccione, E., Arribas, J., Barbosa‐Morais, N. L., … Gil, J. (2018). PTBP1‐mediated alternative splicing regulates the inflammatory secretome and the pro‐tumorigenic effects of senescent cells. Cancer Cell, 34, 85–102 e109. 10.1016/j.ccell.2018.06.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghai, V., Kim, T. K., Etheridge, A., Nielsen, T., Hansen, T., Pedersen, O., Galas, D., & Wang, K. (2019). Extracellular vesicle encapsulated microRNAs in patients with type 2 diabetes are affected by metformin treatment. Journal of Clinical Medicine, 8, 617. 10.3390/jcm8050617 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gluck, S., Guey, B., Gulen, M. F., Wolter, K., Kang, T. W., Schmacke, N. A., Bridgeman, A., Rehwinkel, J., Zender, L., & Ablasser, A. (2017). Innate immune sensing of cytosolic chromatin fragments through cGAS promotes senescence. Nature Cell Biology, 19, 1061–1070. 10.1038/ncb3586 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goetzl, E. J., Schwartz, J. B., Abner, E. L., Jicha, G. A., & Kapogiannis, D. (2018). High complement levels in astrocyte‐derived exosomes of Alzheimer disease. Annals of Neurolofy, 83, 544–552. 10.1002/ana.25172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gomes, A. P., Ilter, D., Low, V., Endress, J. E., Fernandez‐Garcia, J., Rosenzweig, A., Schild, T., Broekaert, D., Ahmed, A., Planque, M., Elia, I., Han, J., Kinzig, C., Mullarky, E., Mutvei, A. P., Asara, J., Cabo, R., Cantley, L. C., Dephoure, N., … Blenis, J. (2020). Age‐induced accumulation of methylmalonic acid promotes tumour progression. Nature, 585, 283–287. 10.1038/s41586-020-2630-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonda, A., Kabagwira, J., Senthil, G. N., & Wall, N. R. (2019). Internalization of exosomes through receptor‐mediated endocytosis. Molecular Cancer Research, 17, 337–347. 10.1158/1541-7786.MCR-18-0891 [DOI] [PubMed] [Google Scholar]
- Gong, L., Chen, B., Zhang, J., Sun, Y., Yuan, J., Niu, X., Hu, G., Chen, Y., Xie, Z., Deng, Z., Li, Q., & Wang, Y. (2020). Human ESC‐sEVs alleviate age‐related bone loss by rejuvenating senescent bone marrow‐derived mesenchymal stem cells. Journal of Extracellular Vesicles, 9, 1800971. 10.1080/20013078.2020.1800971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goulielmaki, E., Ioannidou, A., Tsekrekou, M., Stratigi, K., Poutakidou, I. K., Gkirtzimanaki, K., Aivaliotis, M., Evangelou, K., Topalis, P., Altmuller, J., Gorgoulis, V. G., Chatzinikolaou, G., & Garinis, G. A. (2020). Tissue‐infiltrating macrophages mediate an exosome‐based metabolic reprogramming upon DNA damage. Nature Communications, 11, 42. 10.1038/s41467-019-13894-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grey, M., Dunning, C. J., Gaspar, R., Grey, C., Brundin, P., Sparr, E., & Linse, S. (2015). Acceleration of alpha‐synuclein aggregation by exosomes. Journal of Biological Chemistry, 290, 2969–2982. 10.1074/jbc.M114.585703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griukova, A., Deryabin, P., Shatrova, A., Burova, E., Severino, V., Farina, A., Nikolsky, N., & Borodkina, A. (2019). Molecular basis of senescence transmitting in the population of human endometrial stromal cells. Aging (Albany NY), 11, 9912–9931. 10.18632/aging.102441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grozdanov, V., Bousset, L., Hoffmeister, M., Bliederhaeuser, C., Meier, C., Madiona, K., Pieri, L., Kiechle, M., McLean, P. J., Kassubek, J., Behrends, C., Ludolph, A. C., Weishaupt, J. H., Melki, R., & Danzer, K. M. (2019). Increased immune activation by pathologic alpha‐synuclein in Parkinson's disease. Annals of Neurology, 86, 593–606. 10.1002/ana.25557 [DOI] [PubMed] [Google Scholar]
- Guenther, G. G., Peralta, E. R., Rosales, K. R., Wong, S. Y., Siskind, L. J., & Edinger, A. L. (2008). Ceramide starves cells to death by downregulating nutrient transporter proteins. Proceedings of the National Academy of Sciences of the United States of America, 105, 17402–17407. 10.1073/pnas.0802781105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gui, Y., Liu, H., Zhang, L., Lv, W., & Hu, X. (2015). Altered microRNA profiles in cerebrospinal fluid exosome in Parkinson disease and Alzheimer disease. Oncotarget, 6, 37043–37053. 10.18632/oncotarget.6158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Habiballa, L., Salmonowicz, H., & Passos, J. F. (2019). Mitochondria and cellular senescence: Implications for musculoskeletal ageing. Free Radical Biology and Medicine, 132, 3–10. 10.1016/j.freeradbiomed.2018.10.417 [DOI] [PubMed] [Google Scholar]
- Haddad, M., Perrotte, M., Landri, S., Lepage, A., Fulop, T., & Ramassamy, C. (2019). Circulating and extracellular vesicles levels of N‐(1‐Carboxymethyl)‐L‐Lysine (CML) differentiate early to moderate Alzheimer's disease. Journal of Alzheimers Disease, 69, 751–762. 10.3233/JAD-181272 [DOI] [PubMed] [Google Scholar]
- Hafez, A. Y., Messinger, J. E., McFadden, K., Fenyofalvi, G., Shepard, C. N., Lenzi, G. M., Kim, B., & Luftig, M. A. (2017). Limited nucleotide pools restrict Epstein‐Barr virus‐mediated B‐cell immortalization. Oncogenesis, 6, e349. 10.1038/oncsis.2017.46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han, C., Xiong, N., Guo, X., Huang, J., Ma, K., Liu, L., Xia, Y., Shen, Y., Li, J., Jiang, H., Wang, L., Guo, S., Xu, X., Zhang, G., Liu, J., Cao, X., Zhang, Z., Lin, Z., & Wang, T. (2019a). Exosomes from patients with Parkinson's disease are pathological in mice. Journal of Molecular Medicine (Berl), 97, 1329–1344. 10.1007/s00109-019-01810-z [DOI] [PubMed] [Google Scholar]
- Han, C., Zhou, J., Liu, B., Liang, C., Pan, X., Zhang, Y., Zhang, Y., Wang, Y., Shao, L., Zhu, B., Wang, J., Yin, Q., Yu, Xi‐Y., & Li, Y. (2019b). Delivery of miR‐675 by stem cell‐derived exosomes encapsulated in silk fibroin hydrogel prevents aging‐induced vascular dysfunction in mouse hindlimb. Material Science Engineering C Material Biology Applied, 99, 322–332. 10.1016/j.msec.2019.01.122 [DOI] [PubMed] [Google Scholar]
- Han, L., Long, Q., Li, S., Xu, Q., Zhang, B., Dou, X., Qian, M., Jiramongkol, Y., Guo, J., Cao, L., Eugene Chin, Y., Lam, E. W.‐.F., Jiang, J., & Sun, Yu (2020). Senescent stromal cells promote cancer resistance through SIRT1 loss‐potentiated overproduction of small extracellular vesicles. Cancer Research, 80, 3383–3398. 10.1158/0008-5472.CAN-20-0506 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haney, M. J., Klyachko, N. L., Zhao, Y., Gupta, R., Plotnikova, E. G., He, Z., Patel, T., Piroyan, A., Sokolsky, M., Kabanov, A. V., & Batrakova, E. V. (2015). Exosomes as drug delivery vehicles for Parkinson's disease therapy. Journal of Controlled Release, 207, 18–30. 10.1016/j.jconrel.2015.03.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hargitai, R., Kis, D., Persa, E., Szatmari, T., Safrany, G., & Lumniczky, K. (2021). Oxidative stress and gene expression modifications mediated by extracellular vesicles: An in vivo study of the radiation‐induced bystander effect. Antioxidants (Basel), 10(2), 156. 10.3390/antiox10020156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayflick, L., & Moorhead, P. S. (1961). The serial cultivation of human diploid cell strains. Experimental Cell Research, 25, 585–621. 10.1016/0014-4827(61)90192-6 [DOI] [PubMed] [Google Scholar]
- Hernandez‐Segura, A., Nehme, J., & Demaria, M. (2018). Hallmarks of cellular senescence. Trends in Cell Biology, 28, 436–453. 10.1016/j.tcb.2018.02.001 [DOI] [PubMed] [Google Scholar]
- Hickson, L. J., Langhi Prata, L. G. P., Bobart, S. A., Evans, T. K., Giorgadze, N., Hashmi, S. K., Herrmann, S. M., Jensen, M. D., Jia, Q., Jordan, K. L., Kellogg, T. A., Khosla, S., Koerber, D. M., Lagnado, A. B., Lawson, D. K., LeBrasseur, N. K., Lerman, L. O., McDonald, K. M., McKenzie, T. J., … Kirkland, J. L. (2019). Senolytics decrease senescent cells in humans: Preliminary report from a clinical trial of Dasatinib plus Quercetin in individuals with diabetic kidney disease. EBioMedicine, 47, 446–456. 10.1016/j.ebiom.2019.08.069 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hitomi, K., Okada, R., Loo, T. M., Miyata, K., Nakamura, A. J., & Takahashi, A. (2020). DNA damage regulates senescence‐associated extracellular vesicle release via the ceramide pathway to prevent excessive inflammatory responses. International Journal of Molecular Science, 21, 3720. 10.3390/ijms21103720 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hosseinkhani, B., Kuypers, S., van den Akker, N. M. S., Molin, D. G. M., & Michiels, L. (2018). Extracellular vesicles work as a functional inflammatory mediator between vascular endothelial cells and immune cells. Frontiers in Immunology, 9, 1789. 10.3389/fimmu.2018.01789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hromada, C., Muhleder, S., Grillari, J., Redl, H., & Holnthoner, W. (2017). Endothelial extracellular vesicles‐promises and challenges. Frontiers in Physiology, 8, 275. 10.3389/fphys.2017.00275 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu, G., Xia, Y., Chen, B., Zhang, J., Gong, L., Chen, Y., Li, Q., Wang, Y., & Deng, Z. (2021). ESC‐sEVs rejuvenate aging hippocampal NSCs by transferring SMADs to regulate the MYT1‐Egln3‐Sirt1 axis. Molecular Therapy, 29, 103–120. 10.1016/j.ymthe.2020.09.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu, G., Xia, Y., Zhang, J., Chen, Y., Yuan, J., Niu, X., Zhao, B., Li, Q., Wang, Y., & Deng, Z. (2020). ESC‐sEVs rejuvenate senescent hippocampal NSCs by activating lysosomes to improve cognitive dysfunction in vascular dementia. Advanced science (Weinheim, Baden‐Wurttemberg, Germany), 7, 1903330. 10.1002/advs.201903330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu, S., Li, Z., Cores, J., Huang, K., Su, T., Dinh, P. U., & Cheng, K. (2019). Needle‐free injection of exosomes derived from human dermal fibroblast spheroids ameliorates skin photoaging. ACS Nano, 13, 11273–11282. 10.1021/acsnano.9b04384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang, J., Xie, Y., Sun, X., Zeh, H. J.3rd, Kang, R., Lotze, M. T., & Tang, D. (2015). DAMPs, ageing, and cancer: The ‘DAMP Hypothesis’. Ageing Research Reviews, 24, 3–16. 10.1016/j.arr.2014.10.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes, C. E., Coody, T. K., Jeong, M. Y., Berg, J. A., Winge, D. R., & Hughes, A. L. (2020). Cysteine toxicity drives age‐related mitochondrial decline by altering iron homeostasis. Cell, 180, 296–310 e218. 10.1016/j.cell.2019.12.035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Isermann, A., Mann, C., & Rube, C. E. (2020). Histone variant H2A.J marks persistent DNA damage and triggers the secretory phenotype in radiation‐induced senescence. International Journal of Molecular Science, 21, 9130. 10.3390/ijms21239130 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iske, J., Seyda, M., Heinbokel, T., Maenosono, R., Minami, K., Nian, Y., Quante, M., Falk, C. S., Azuma, H., Martin, F., Passos, J. F., Niemann, C. U., Tchkonia, T., Kirkland, J. L., Elkhal, A., & Tullius, S. G. (2020). Senolytics prevent mt‐DNA‐induced inflammation and promote the survival of aged organs following transplantation. Nature Communications, 11, 4289. 10.1038/s41467-020-18039-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito, Y., Hoare, M., & Narita, M. (2017). Spatial and temporal control of senescence. Trends in Cell Biology, 27, 820–832. 10.1016/j.tcb.2017.07.004 [DOI] [PubMed] [Google Scholar]
- Jeon, O. H., Kim, C., Laberge, R. M., Demaria, M., Rathod, S., Vasserot, A. P., Chung, J. W., Kim, D. H., Poon, Y., David, N., Baker, D. J., Deursen, J. M., Campisi, J., & Elisseeff, J. H. (2017). Local clearance of senescent cells attenuates the development of post‐traumatic osteoarthritis and creates a pro‐regenerative environment. Nature Medicine, 23, 775–781. 10.1038/nm.4324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeon, O. H., Wilson, D. R., Clement, C. C., Rathod, S., Cherry, C., Powell, B., Lee, Z., Khalil, A. M., Green, J. J., Campisi, J., Santambrogio, L., Witwer, K. W., & Elisseeff, J. H. (2019). Senescence cell‐associated extracellular vesicles serve as osteoarthritis disease and therapeutic markers. JCI Insight, 4, e125019. 10.1172/jci.insight.125019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia, J., Ma, L., Wu, M., Zhang, L., Zhang, X., Zhai, Q., Jiang, T., Wang, Q., & Xiong, L. (2014). Anandamide protects HT22 cells exposed to hydrogen peroxide by inhibiting CB1 receptor‐mediated type 2 NADPH oxidase. Oxidative Medicine and Cellular Longevity, 2014, 893516. 10.1155/2014/893516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia, L., Qiu, Q., Zhang, H., Chu, L., Du, Y., Zhang, J., Zhou, C., Liang, F., Shi, S., Wang, S., Li, F., Wang, Q., Li, Y., Shen, L., Wei, Y., & Jia, J. (2019a). Concordance between the assessment of Abeta42, T‐tau, and P‐T181‐tau in peripheral blood neuronal‐derived exosomes and cerebrospinal fluid. Alzheimers & Dementia, 15, 1071–1080. 10.1016/j.jalz.2019.05.002 [DOI] [PubMed] [Google Scholar]
- Jia, L., Song, H., Fan, W., Song, Y., Wang, G., Li, X., He, Y., & Yao, A. (2020). The association between high mobility group box 1 chromatin protein and mitotic chromosomes in glioma cells. Oncology Letters, 19, 745–752. 10.3892/ol.2019.11170 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia, Y., Zheng, Z., Yang, Y., Zou, M., Li, J., Wang, L., Guan, M., & Xue, Y. (2019b). MiR‐4756 promotes albumin‐induced renal tubular epithelial cell epithelial‐to‐mesenchymal transition and endoplasmic reticulum stress via targeting Sestrin2. Journal of Cellular Physiology, 234, 2905–2915. 10.1002/jcp.27107 [DOI] [PubMed] [Google Scholar]
- Jiang, F., Chen, Q., Wang, W., Ling, Y., Yan, Y., & Xia, P. (2020). Hepatocyte‐derived extracellular vesicles promote endothelial inflammation and atherogenesis via microRNA‐1. Journal of Hepatology, 72, 156–166. 10.1016/j.jhep.2019.09.014 [DOI] [PubMed] [Google Scholar]
- Johmura, Y., Yamanaka, T., Omori, S., Wang, T. W., Sugiura, Y., Matsumoto, M., Suzuki, N., Kumamoto, S., Yamaguchi, K., Hatakeyama, S., Tomoyo Takami, R., Yamaguchi, E., Shimizu, K., Ikeda, N., Okahashi, R., Mikawa, M., Suematsu, M., Arita, M. S., & Nakanishi, M. (2021). Senolysis by glutaminolysis inhibition ameliorates various age‐associated disorders. Science, 371, 265–270. 10.1126/science.abb5916 [DOI] [PubMed] [Google Scholar]
- Jung, S. H., Hwang, H. J., Kang, D., Park, H. A., Lee, H. C., Jeong, D., Lee, K., Park, H. J., Ko, Y. G., & Lee, J. S. (2019). mTOR kinase leads to PTEN‐loss‐induced cellular senescence by phosphorylating p53. Oncogene, 38, 1639–1650. 10.1038/s41388-018-0521-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Justice, J. N., Nambiar, A. M., Tchkonia, T., LeBrasseur, N. K., Pascual, R., Hashmi, S. K., Prata, L., Masternak, M. M., Kritchevsky, S. B., Musi, N., & Kirkland, J. L. (2019). Senolytics in idiopathic pulmonary fibrosis: Results from a first‐in‐human, open‐label, pilot study. EBioMedicine, 40, 554–563. 10.1016/j.ebiom.2018.12.052 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadota, T., Yoshioka, Y., Fujita, Y., Araya, J., Minagawa, S., Hara, H., Miyamoto, A., Suzuki, S., Fujimori, S., Kohno, T., Fujii, T., Kishi, K., Kuwano, K., & Ochiya, T. (2020). Extracellular vesicles from fibroblasts induce epithelial cell senescence in pulmonary fibrosis. The American Journal of Respiratory Cell and Molecular Biology, 63, 623–636. 10.1165/rcmb.2020-0002OC [DOI] [PubMed] [Google Scholar]
- Kalluri, R., & LeBleu, V. S. (2020). The biology, function, and biomedical applications of exosomes. Science, 367, eaau6977. 10.1126/science.aau6977 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang, C., Xu, Q., Martin, T. D., Li, M. Z., Demaria, M., Aron, L., Lu, T., Yankner, B. A., Campisi, J., & Elledge, S. J. (2015). The DNA damage response induces inflammation and senescence by inhibiting autophagy of GATA4. Science, 349, aaa5612. 10.1126/science.aaa5612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kapogiannis, D., Mustapic, M., Shardell, M. D., Berkowitz, S. T., Diehl, T. C., Spangler, R. D., Tran, J., Lazaropoulos, M. P., Chawla, S., Gulyani, S., Eitan, E., An, Y., Huang, C. ‐. W., Oh, E. S., Lyketsos, C. G., Resnick, S. M., Goetzl, E. J., & Ferrucci, L. (2019). Association of extracellular vesicle biomarkers with alzheimer disease in the baltimore longitudinal study of aging. JAMA Neurology, 76, 1340–1351. 10.1001/jamaneurol.2019.2462 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kenneweg, F., Bang, C., Xiao, K., Boulanger, C. M., Loyer, X., Mazlan, S., Schroen, B., Hermans‐Beijnsberger, S., Foinquinos, A., Hirt, M. N., Eschenhagen, T., Funcke, S., Stojanovic, S., Genschel, C., Schimmel, K., Just, A., Pfanne, A., Scherf, K., Dehmel, S., & Raemon‐Buettner, S. M. (2019). Long noncoding RNA‐enriched vesicles secreted by hypoxic cardiomyocytes drive cardiac fibrosis. Molecular Therapy Nucleic Acids, 18, 363–374. 10.1016/j.omtn.2019.09.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khayrullin, A., Krishnan, P., Martinez‐Nater, L., Mendhe, B., Fulzele, S., Liu, Y., Mattison, J. A., & Hamrick, M. W. (2019). Very long‐chain c24:1 ceramide is increased in serum extracellular vesicles with aging and can induce senescence in bone‐derived mesenchymal stem cells. Cells, 8, 37. 10.3390/cells8010037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koff, W. C., & Williams, M. A. (2020). Covid‐19 and immunity in aging populations ‐ A new research agenda. New England Journal of Medicine, 383, 804–805. 10.1056/NEJMp2006761 [DOI] [PubMed] [Google Scholar]
- Kolhe, R., Hunter, M., Liu, S., Jadeja, R. N., Pundkar, C., Mondal, A. K., Mendhe, B., Drewry, M., Rojiani, M. V., Liu, Y., Isales, C. M., Guldberg, R. E., Hamrick, M. W., & Fulzele, S. (2017). Gender‐specific differential expression of exosomal miRNA in synovial fluid of patients with osteoarthritis. Scientific Reports, 7, 2029. 10.1038/s41598-017-01905-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krizhanovsky, V., Yon, M., Dickins, R. A., Hearn, S., Simon, J., Miething, C., Yee, H., Zender, L., & Lowe, S. W. (2008). Senescence of activated stellate cells limits liver fibrosis. Cell, 134, 657–667. 10.1016/j.cell.2008.06.049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kulkarni, R., Bajaj, M., Ghode, S., Jalnapurkar, S., Limaye, L., & Kale, V. P. (2018). Intercellular transfer of microvesicles from young mesenchymal stromal cells rejuvenates aged murine hematopoietic stem cells. Stem Cells, 36, 420–433. 10.1002/stem.2756 [DOI] [PubMed] [Google Scholar]
- Laberge, R. M., Sun, Y., Orjalo, A. V., Patil, C. K., Freund, A., Zhou, L., Curran, S. C., Davalos, A. R., Wilson‐Edell, K. A., Liu, S., Limbad, C., Demaria, M., Li, P., Hubbard, G. B., Ikeno, Y., Javors, M., Desprez, P. ‐. Y., Benz, C. C., Kapahi, P., … Campisi, J. (2015). MTOR regulates the pro‐tumorigenic senescence‐associated secretory phenotype by promoting IL1A translation. Nature Cell Biology, 17, 1049–1061. 10.1038/ncb3195 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laberge, R. M., Zhou, L., Sarantos, M. R., Rodier, F., Freund, A., de Keizer, P. L., Liu, S., Demaria, M., Cong, Y. S., Kapahi, P., Desprez, P. ‐. Y., Hughes, R. E., & Campisi, J. (2012). Glucocorticoids suppress selected components of the senescence‐associated secretory phenotype. Aging Cell, 11, 569–578. 10.1111/j.1474-9726.2012.00818.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lamontagne‐Proulx, J., St‐Amour, I., Labib, R., Pilon, J., Denis, H. L., Cloutier, N., Roux‐Dalvai, F., Vincent, A. T., Mason, S. L., Williams‐Gray, C., Duchez, A ‐ C., Droit, A., Lacroix, S., Dupré, N., Langlois, M., Chouinard, S., Panisset, M., Barker, R. A., Boilard, E., … Cicchetti, F. (2019). Portrait of blood‐derived extracellular vesicles in patients with Parkinson's disease. Neurobiology of Disease, 124, 163–175. 10.1016/j.nbd.2018.11.002 [DOI] [PubMed] [Google Scholar]
- Lee, B. R., Kim, J. H., Choi, E. S., Cho, J. H., & Kim, E. (2018). Effect of young exosomes injected in aged mice. International Journal of Nanomedicine 13, 5335–5345. 10.2147/IJN.S170680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee, H., Cha, H., & Park, J. H. (2020). Derivation of cell‐engineered nanovesicles from human induced pluripotent stem cells and their protective effect on the senescence of dermal fibroblasts. International Journal of Molecular Science, 21, 343. 10.3390/ijms21010343 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee, S. ‐ H., Lee, J. ‐ H., Lee, H. ‐ Y., & Min, K. ‐ J. (2019). Sirtuin signaling in cellular senescence and aging. BMB Reports, 52, 24–34. 10.5483/BMBRep.2019.52.1.290 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei, Q., Liu, T., Gao, F., Xie, H., Sun, L., Zhao, A., Ren, W., Guo, H., Zhang, L., Wang, H., Zhichao, C., Guo, An‐Y., & Li, Q. (2017). Microvesicles as potential biomarkers for the identification of senescence in human mesenchymal stem cells. Theranostics, 7, 2673–2689. 10.7150/thno.18915 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, D., Liu, J., Guo, B., Liang, C., Dang, L., Lu, C., He, X., Cheung, H. Y., Xu, L., Lu, C., de Wit, E., Soares, R. J., Nielsen, B. S., Holmstrøm, K., Martínez‐Herrera, D. J., Huarte, M., Louloupi, A., Drost, J., Elkon, R., & Agami, R. (2016). Osteoclast‐derived exosomal miR‐214‐3p inhibits osteoblastic bone formation. Nature Communications 7, 10872. 10.1038/ncomms10872 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, G., He, X., Zhang, L., Ran, Q., Wang, J., Xiong, A., Wu, D., Chen, F., Sun, J., & Chang, C. (2020a). Assessing ACE2 expression patterns in lung tissues in the pathogenesis of COVID‐19. Journal of Autoimmunity, 112, 102463. 10.1016/j.jaut.2020.102463 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, H., Slone, J., Fei, L., & Huang, T. (2019). Mitochondrial DNA variants and common diseases: A mathematical model for the diversity of age‐related mtDNA mutations. Cells, 8, 608. 10.3390/cells8060608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, L., van Breugel, P. C., Loayza‐Puch, F., Ugalde, A. P., Korkmaz, G., Messika‐Gold, N., Han, R., Lopes, R., Barbera, E. P., Teunissen, H., de Wit, E., Soares, R. J., Nielsen, B. S., Holmstrøm, K., Martínez‐Herrera, D. J., Huarte, M., Louloupi, A., Drost, J., Elkon, R., & Agami, R. (2018). LncRNA‐OIS1 regulates DPP4 activation to modulate senescence induced by RAS. Nucleic Acids Research, 46, 4213–4227. 10.1093/nar/gky087 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, Y., Jiang, Y., Paxman, J., O'Laughlin, R., Klepin, S., Zhu, Y., Pillus, L., Tsimring, L. S., Hasty, J., & Hao, N. (2020b). A programmable fate decision landscape underlies single‐cell aging in yeast. Science, 369, 325–329. 10.1126/science.aax9552 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li, Y., Meng, Y., Zhu, X., Saadiq, I. M., Jordan, K. L., Eirin, A., & Lerman, L. O. (2020c). Metabolic syndrome increases senescence‐associated micro‐RNAs in extracellular vesicles derived from swine and human mesenchymal stem/stromal cells. Cell Communication Signalling, 18, 124. 10.1186/s12964-020-00624-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lia, G., Di Vito, C., Cerrano, M., Brunello, L., Calcaterra, F., Tapparo, M., Giaccone, L., Mavilio, D., & Bruno, B. (2020). Extracellular vesicles after allogeneic hematopoietic cell transplantation: Emerging role in post‐transplant complications. Frontiers in Immunology, 11, 422. 10.3389/fimmu.2020.00422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim, C. Z. J., Zhang, Y., Chen, Y., Zhao, H., Stephenson, M. C., Ho, N. R. Y., Chen, Y., Chung, J., Reilhac, A., Loh, T. P., Chen, C. L. H., & Shao, H. (2019). Subtyping of circulating exosome‐bound amyloid beta reflects brain plaque deposition. Nature Communications, 10, 1144. 10.1038/s41467-019-09030-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin, X., Li, S., Wang, Y. J., Wang, Y., Zhong, J. Y., He, J. Y., Cui, X. J., Zhan, J. K., & Liu, Y. S. (2019). Exosomal Notch3 from high glucose‐stimulated endothelial cells regulates vascular smooth muscle cells calcification/aging. Life Science, 232, 116582. 10.1016/j.lfs.2019.116582 [DOI] [PubMed] [Google Scholar]
- Liu, G. Y., & Sabatini, D. M. (2020). mTOR at the nexus of nutrition, growth, ageing and disease. Nature Reviews Molecular Cell Biology, 21, 183–203. 10.1038/s41580-019-0199-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, P., Tang, Q., Chen, M., Chen, W., Lu, Y., Liu, Z., & He, Z. (2020). Hepatocellular senescence: Immunosurveillance and future senescence‐induced therapy in hepatocellular carcinoma. Frontiers in Oncology, 10, 589908. 10.3389/fonc.2020.589908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, S., Mahairaki, V., Bai, H., Ding, Z., Li, J., Witwer, K. W., & Cheng, L. (2019a). Highly purified human extracellular vesicles produced by stem cells alleviate aging cellular phenotypes of senescent human cells. Stem Cells, 37, 779–790. 10.1002/stem.2996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu, Y., Liu, Z., Xie, Y., Zhao, C., & Xu, J. (2019b). Serum extracellular vesicles retard H9C2 cell senescence by suppressing miR‐34a expression. Journal of Cardiovascular Translational Research, 12, 45–50. 10.1007/s12265-018-9847-4 [DOI] [PubMed] [Google Scholar]
- Loeser, R. F., Collins, J. A., & Diekman, B. O. (2016). Ageing and the pathogenesis of osteoarthritis. Nature Reviews Rheumatology, 12, 412–420. 10.1038/nrrheum.2016.65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loo, T. M., Miyata, K., Tanaka, Y., & Takahashi, A. (2020). Cellular senescence and senescence‐associated secretory phenotype via the cGAS‐STING signaling pathway in cancer. Cancer Science, 111, 304–311. 10.1111/cas.14266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Losurdo, M., Pedrazzoli, M., D'Agostino, C., Elia, C. A., Massenzio, F., Lonati, E., Mauri, M., Rizzi, L., Molteni, L., Bresciani, E., Dander, E., D'Amico, G., Bulbarelli, A., Torsello, A., Matteoli, M., Buffelli, M., & Coco, S. (2020). Intranasal delivery of mesenchymal stem cell‐derived extracellular vesicles exerts immunomodulatory and neuroprotective effects in a 3xTg model of Alzheimer's disease. Stem Cells Translational Medicine, 9, 1068–1084. 10.1002/sctm.19-0327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loyer, X., Zlatanova, I., Devue, C., Yin, M., Howangyin, K. Y., Klaihmon, P., Guerin, C. L., Kheloufi, M., Vilar, J., Zannis, K., Fleischmann, B. K., Hwang, Do W., Park, J., Lee, H., Menasché, P., Silvestre, J. ‐. S., & Boulanger, C. M. (2018). Intra‐cardiac release of extracellular vesicles shapes inflammation following myocardial infarction. Circulation Research, 123, 100–106. 10.1161/CIRCRESAHA.117.311326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo, Z. W., Li, F. X., Liu, Y. W., Rao, S. S., Yin, H., Huang, J., Chen, C. Y., Hu, Y., Zhang, Y., Tan, Y. J., Yuan, L. ‐. Q., Chen, T. ‐. H., Liu, H. ‐. M., Cao, J., Liu, Z. ‐. Z., Wang, Z. ‐. X., & Xie, H. (2019). Aptamer‐functionalized exosomes from bone marrow stromal cells target bone to promote bone regeneration. Nanoscale, 11, 20884–20892. 10.1039/c9nr02791b [DOI] [PubMed] [Google Scholar]
- Lyu, L., Wang, H., Li, B., Qin, Q., Qi, L., Nagarkatti, M., Nagarkatti, P., Janicki, J. S., Wang, X. L., & Cui, T. (2015). A critical role of cardiac fibroblast‐derived exosomes in activating renin angiotensin system in cardiomyocytes. The Journal of Molecular Cell Cardiology, 89, 268–279. 10.1016/j.yjmcc.2015.10.022 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Machida, T., Tomofuji, T., Ekuni, D., Maruyama, T., Yoneda, T., Kawabata, Y., Mizuno, H., Miyai, H., Kunitomo, M., & Morita, M. (2015). MicroRNAs in salivary exosome as potential biomarkers of aging. International Journal of Molecular Science, 16, 21294–21309. 10.3390/ijms160921294 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malaquin, N., Olivier, M. A., Martinez, A., Nadeau, S., Sawchyn, C., Coppe, J. P., Cardin, G., Mallette, F. A., Campisi, J., & Rodier, F. (2020). Non‐canonical ATM/MRN activities temporally define the senescence secretory program. EMBO Reports, 21, e50718. 10.15252/embr.202050718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mangelinck, A., Coudereau, C., Courbeyrette, R., Ourarhni, K., Hamiche, A., Redon, C., Bonner, W. M., van Dijk, E., Derbois, C., Olaso, R., Deleuze, J. ‐. F., Fenaille, F., Rübe, C. E., Thuret, J. ‐. Y., & Mann, C. (2020). The H2A.J histone variant contributes to Interferon‐Stimulated Gene expression in senescence by its weak interaction with H1 and the derepression of repeated DNA sequences. bioRxiv, 2020.2010.2029.361204. 10.1101/2020.10.29.361204</bib> [DOI] [Google Scholar]
- Mas‐Bargues, C., Sanz‐Ros, J., Roman‐Dominguez, A., Gimeno‐Mallench, L., Ingles, M., Vina, J., & Borras, C. (2020). Extracellular vesicles from healthy cells improves cell function and stemness in premature senescent stem cells by miR‐302b and HIF‐1alpha activation. Biomolecules, 10, 957. 10.3390/biom10060957 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masaldan, S., Clatworthy, S. A. S., Gamell, C., Meggyesy, P. M., Rigopoulos, A. T., Haupt, S., Haupt, Y., Denoyer, D., Adlard, P. A., Bush, A. I., & Cater, M. A. (2018). Iron accumulation in senescent cells is coupled with impaired ferritinophagy and inhibition of ferroptosis. Redox Biology, 14, 100–115. 10.1016/j.redox.2017.08.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mato‐Basalo, R., Morente‐Lopez, M., Arntz, O. J., van de Loo, F. A. J., Fafian‐Labora, J., & Arufe, M. C. (2021). Therapeutic potential for regulation of the nuclear factor kappa‐B transcription factor p65 to prevent cellular senescence and activation of pro‐inflammatory in mesenchymal stem cells. International Journal of Molecular Science, 22, 3367. 10.3390/ijms22073367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsumoto, J., Stewart, T., Sheng, L., Li, N., Bullock, K., Song, N., Shi, M., Banks, W. A., & Zhang, J. (2017). Transmission of alpha‐synuclein‐containing erythrocyte‐derived extracellular vesicles across the blood‐brain barrier via adsorptive mediated transcytosis: Another mechanism for initiation and progression of Parkinson's disease? Acta Neuropathologica Communications, 5, 71. 10.1186/s40478-017-0470-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mensa, E., Guescini, M., Giuliani, A., Bacalini, M. G., Ramini, D., Corleone, G., Ferracin, M., Fulgenzi, G., Graciotti, L., Prattichizzo, F., Sorci, L., Battistelli, M., Monsurrò, V., Bonfigli, A. R., Cardelli, M., Recchioni, R., Marcheselli, F., Latini, S., Maggio, S., … Olivieri, F. (2020). Small extracellular vesicles deliver miR‐21 and miR‐217 as pro‐senescence effectors to endothelial cells. Journal of Extracellular Vesicles, 9, 1725285. 10.1080/20013078.2020.1725285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer, K., & Yankner, B. A. (2017). Slowing down aging. Cell Metabolism, 26, 592–593. 10.1016/j.cmet.2017.09.012 [DOI] [PubMed] [Google Scholar]
- Miao, Y., Cui, Z., Gao, Q., Rui, R., & Xiong, B. (2020). Nicotinamide mononucleotide supplementation reverses the declining quality of maternally aged oocytes. Cell Reproduction. 32, 107987. 10.1016/j.celrep.2020.107987 [DOI] [PubMed] [Google Scholar]
- Minakaki, G., Menges, S., Kittel, A., Emmanouilidou, E., Schaeffner, I., Barkovits, K., Bergmann, A., Rockenstein, E., Adame, A., Marxreiter, F., Galasko, D., Buzás, E. I., Schlötzer‐Schrehardt, U., Marcus, K., Xiang, W., Lie, D. C., Vekrellis, K., Masliah, E., Winkler, J., & Klucken, J. (2018). Autophagy inhibition promotes SNCA/alpha‐synuclein release and transfer via extracellular vesicles with a hybrid autophagosome‐exosome‐like phenotype. Autophagy, 14, 98–119. 10.1080/15548627.2017.1395992 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Minamino, T., Orimo, M., Shimizu, I., Kunieda, T., Yokoyama, M., Ito, T., Nojima, A., Nabetani, A., Oike, Y., Matsubara, H., Ishikawa, F., & Komuro, I. (2009). A crucial role for adipose tissue p53 in the regulation of insulin resistance. Nature Medicine, 15, 1082–1087. 10.1038/nm.2014 [DOI] [PubMed] [Google Scholar]
- Misawa, T., Tanaka, Y., Okada, R., & Takahashi, A. (2020). Biology of extracellular vesicles secreted from senescent cells as senescence‐associated secretory phenotype factors. Geriatrics and Gerontology International, 20, 539–546. 10.1111/ggi.13928 [DOI] [PubMed] [Google Scholar]
- Moiseeva, O., Deschenes‐Simard, X., St‐Germain, E., Igelmann, S., Huot, G., Cadar, A. E., Bourdeau, V., Pollak, M. N., & Ferbeyre, G. (2013). Metformin inhibits the senescence‐associated secretory phenotype by interfering with IKK/NF‐kappaB activation. Aging Cell 12, 489–498. 10.1111/acel.12075 [DOI] [PubMed] [Google Scholar]
- Moller, A., & Lobb, R. J. (2020). The evolving translational potential of small extracellular vesicles in cancer. Nature Reviews Cancer, 20, 697–709. 10.1038/s41568-020-00299-w [DOI] [PubMed] [Google Scholar]
- Morita, M., Gravel, S. P., Chenard, V., Sikstrom, K., Zheng, L., Alain, T., Gandin, V., Avizonis, D., Arguello, M., Zakaria, C., McLaughlan, S., Nouet, Y., Pause, A., Pollak, M., Gottlieb, E., Larsson, O., St‐Pierre, J., Topisirovic, I., & Sonenberg, N. (2013). mTORC1 controls mitochondrial activity and biogenesis through 4E‐BP‐dependent translational regulation. Cell Metabolism, 18, 698–711. 10.1016/j.cmet.2013.10.001 [DOI] [PubMed] [Google Scholar]
- Morita, M., Prudent, J., Basu, K., Goyon, V., Katsumura, S., Hulea, L., Pearl, D., Siddiqui, N., Strack, S., McGuirk, S., St‐Pierre, J., Larsson, O., Topisirovic, I., Vali, H., McBride, H. M., Bergeron, J. J., & Sonenberg, N. (2017). mTOR controls mitochondrial dynamics and cell survival via MTFP1. Molecular Cell, 67, 922–935 e925. 10.1016/j.molcel.2017.08.013 [DOI] [PubMed] [Google Scholar]
- Mosteiro, L., Pantoja, C., Alcazar, N., Marion, R. M., Chondronasiou, D., Rovira, M., Fernandez‐Marcos, P. J., Munoz‐Martin, M., Blanco‐Aparicio, C., Pastor, J., Gómez‐López, G., De Martino, A., Blasco, M. A., Abad, M., & Serrano, M. (2016). Tissue damage and senescence provide critical signals for cellular reprogramming in vivo. Science, 354, aaf4445. 10.1126/science.aaf4445 [DOI] [PubMed] [Google Scholar]
- Nacarelli, T., Lau, L., Fukumoto, T., Zundell, J., Fatkhutdinov, N., Wu, S., Aird, K. M., Iwasaki, O., Kossenkov, A. V., Schultz, D., Noma, K. ‐. I., Baur, J. A., Schug, Z., Tang, H. ‐. Y., Speicher, D. W., David, G., & Zhang, R. (2019). NAD(+) metabolism governs the proinflammatory senescence‐associated secretome. Nature Cell Biology, 21, 397–407. 10.1038/s41556-019-0287-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nanbo, A., Kawanishi, E., Yoshida, R., & Yoshiyama, H. (2013). Exosomes derived from Epstein‐Barr virus‐infected cells are internalized via caveola‐dependent endocytosis and promote phenotypic modulation in target cells. Journal of Virology, 87, 10334–10347. 10.1128/JVI.01310-13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ngoi, N. Y., Liew, A. Q., Chong, S. J. F., Davids, M. S., Clement, M. V., & Pervaiz, S. (2021). The redox‐senescence axis and its therapeutic targeting. Redox Biology, 45, 102032. 10.1016/j.redox.2021.102032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen, M. A., Karunakaran, D., Geoffrion, M., Cheng, H. S., Tandoc, K., Perisic Matic, L., Hedin, U., Maegdefessel, L., Fish, J. E., & Rayner, K. J. (2018). Extracellular vesicles secreted by atherogenic macrophages transfer MicroRNA to inhibit cell migration. Arteriosclerosis, Thrombosis, and Vascular Biology, 38, 49–63. 10.1161/ATVBAHA.117.309795 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ni, Z., Kuang, L., Chen, H., Xie, Y., Zhang, B., Ouyang, J., Wu, J., Zhou, S., Chen, L., Su, N., Tan, Q. Y., Luo, X., Chen, Bo, Chen, S., Yin, L., Huang, H., Du, X., & Chen, L. (2019). The exosome‐like vesicles from osteoarthritic chondrocyte enhanced mature IL‐1beta production of macrophages and aggravated synovitis in osteoarthritis. Cell Death & Disease, 10, 522. 10.1038/s41419-019-1739-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicholson, C., Shah, N., Ishii, M., Annamalai, B., Brandon, C., Rodgers, J., Nowling, T., & Rohrer, B. (2020). Mechanisms of extracellular vesicle uptake in stressed retinal pigment epithelial cell monolayers. Biochim Biophys Acta Molecular Basis Disease, 1866, 165608. 10.1016/j.bbadis.2019.165608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niemann, J., Johne, C., Schroder, S., Koch, F., Ibrahim, S. M., Schultz, J., Tiedge, M., & Baltrusch, S. (2017). An mtDNA mutation accelerates liver aging by interfering with the ROS response and mitochondrial life cycle. Free Radical Biology and Medicine, 102, 174–187. 10.1016/j.freeradbiomed.2016.11.035 [DOI] [PubMed] [Google Scholar]
- Nogueras‐Ortiz, C. J., Mahairaki, V., Delgado‐Peraza, F., Das, D., Avgerinos, K., Eren, E., Hentschel, M., Goetzl, E. J., Mattson, M. P., & Kapogiannis, D. (2020). Astrocyte‐ and neuron‐derived extracellular vesicles from Alzheimer's disease patients effect complement‐mediated neurotoxicity. Cells, 9, 1618. 10.3390/cells9071618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oh, B. M., Lee, S. J., Cho, H. J., Park, Y. S., Kim, J. T., Yoon, S. R., Lee, S. C., Lim, J. S., Kim, B. Y., Choe, Y. K., & Lee, H. G. (2017). Cystatin SN inhibits auranofin‐induced cell death by autophagic induction and ROS regulation via glutathione reductase activity in colorectal cancer. Cell Death & Disease, 8, e3053. 10.1038/cddis.2017.446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oh, J. G., Watanabe, S., Lee, A., Gorski, P. A., Lee, P., Jeong, D., Liang, L., Liang, Y., Baccarini, A., Sahoo, S., Brown, B. D., Hajjar, R. J., & Kho, C. (2018a). miR‐146a suppresses SUMO1 expression and induces cardiac dysfunction in maladaptive hypertrophy. Circulation Research, 123, 673–685. 10.1161/CIRCRESAHA.118.312751 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oh, M., Lee, J., Kim, Y. J., Rhee, W. J., & Park, J. H. (2018b). Exosomes derived from human induced pluripotent stem cells ameliorate the aging of skin fibroblasts. International Journal of Molecular Science 19(6), 1715. 10.3390/ijms19061715 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onken, B., & Driscoll, M. (2010). Metformin induces a dietary restriction‐like state and the oxidative stress response to extend C. elegans Healthspan via AMPK, LKB1, and SKN‐1. Plos One, 5, e8758. 10.1371/journal.pone.0008758 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oubaha, M., Miloudi, K., Dejda, A., Guber, V., Mawambo, G., Germain, M. A., Bourdel, G., Popovic, N., Rezende, F. A., Kaufman, R. J., Mallette, F. A., & Sapieha, P. (2016). Senescence‐associated secretory phenotype contributes to pathological angiogenesis in retinopathy. Science Translational Medicine, 8, 362ra144. 10.1126/scitranslmed.aaf9440 [DOI] [PubMed] [Google Scholar]
- Ovadya, Y., Landsberger, T., Leins, H., Vadai, E., Gal, H., Biran, A., Yosef, R., Sagiv, A., Agrawal, A., Shapira, A., Windheim, J., Tsoory, M., Schirmbeck, R., Amit, I., & Geiger, H. (2018). Impaired immune surveillance accelerates accumulation of senescent cells and aging. Nature Communications, 9, 5435. 10.1038/s41467-018-07825-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozcan, S., Alessio, N., Acar, M. B., Mert, E., Omerli, F., Peluso, G., & Galderisi, U. (2016). Unbiased analysis of senescence associated secretory phenotype (SASP) to identify common components following different genotoxic stresses. Aging (Albany NY), 8, 1316–1329. 10.18632/aging.100971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park, J. T., Lee, Y. S., Cho, K. A., & Park, S. C. (2018). Adjustment of the lysosomal‐mitochondrial axis for control of cellular senescence. Ageing Research Reviews, 47, 176–182. 10.1016/j.arr.2018.08.003 [DOI] [PubMed] [Google Scholar]
- Perets, N., Betzer, O., Shapira, R., Brenstein, S., Angel, A., Sadan, T., Ashery, U., Popovtzer, R., & Offen, D. (2019). Golden exosomes selectively target brain pathologies in neurodegenerative and neurodevelopmental disorders. Nano Letter, 19, 3422–3431. 10.1021/acs.nanolett.8b04148 [DOI] [PubMed] [Google Scholar]
- Perrotte, M., Haddad, M., Le Page, A., Frost, E. H., Fulop, T., & Ramassamy, C. (2020). Profile of pathogenic proteins in total circulating extracellular vesicles in mild cognitive impairment and during the progression of Alzheimer's disease. Neurobiology of Aging, 86, 102–111. 10.1016/j.neurobiolaging.2019.10.010 [DOI] [PubMed] [Google Scholar]
- Pescatore, L. A., Gamarra, L. F., & Liberman, M. (2019). Multifaceted mechanisms of vascular calcification in aging. Arteriosclerosis, Thrombosis, and Vascular Biology, 39, 1307–1316. 10.1161/ATVBAHA.118.311576 [DOI] [PubMed] [Google Scholar]
- Picca, A., Guerra, F., Calvani, R., Bucci, C., Lo Monaco, M. R., Bentivoglio, A. R., Coelho‐Junior, H. J., Landi, F., Bernabei, R., & Marzetti, E. (2019). Mitochondrial dysfunction and aging: Insights from the analysis of extracellular vesicles. International Journal of Molecular Science, 20, 805. 10.3390/ijms20040805 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picca, A., Guerra, F., Calvani, R., Marini, F., Biancolillo, A., Landi, G., Beli, R., Landi, F., Bernabei, R., Bentivoglio, A. R., Monaco, M. R. L, Bucci, C., & Marzetti, E. (2020). Mitochondrial signatures in circulating extracellular vesicles of older adults with Parkinson's disease: Results from the EXosomes in PArkiNson's Disease (EXPAND) Study. Journal of Clinical Medicine, 9, 504. 10.3390/jcm9020504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Picca, A., Mankowski, R. T., Burman, J. L., Donisi, L., Kim, J. S., Marzetti, E., & Leeuwenburgh, C. (2018). Mitochondrial quality control mechanisms as molecular targets in cardiac ageing. Nature Reviews Cardiology, 15, 543–554. 10.1038/s41569-018-0059-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pienimaeki‐Roemer, A., Konovalova, T., Musri, M. M., Sigruener, A., Boettcher, A., Meister, G., & Schmitz, G. (2017). Transcriptomic profiling of platelet senescence and platelet extracellular vesicles. Transfusion, 57, 144–156. 10.1111/trf.13896 [DOI] [PubMed] [Google Scholar]
- Porro, A., Feuerhahn, S., Delafontaine, J., Riethman, H., Rougemont, J., & Lingner, J. (2014). Functional characterization of the TERRA transcriptome at damaged telomeres. Nature Communications, 5, 5379. 10.1038/ncomms6379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prasnikar, E., Borisek, J., & Perdih, A. (2021). Senescent cells as promising targets to tackle age‐related diseases. Ageing Research Reviews, 66, 101251. 10.1016/j.arr.2020.101251 [DOI] [PubMed] [Google Scholar]
- Puhm, F., Afonyushkin, T., Resch, U., Obermayer, G., Rohde, M., Penz, T., Schuster, M., Wagner, G., Rendeiro, A. F., Melki, I., Kaun, C., Wojta, J., Bock, C., Jilma, B., Mackman, N., Boilard, E., & Binder, C. J. (2019). Mitochondria are a subset of extracellular vesicles released by activated monocytes and induce type I IFN and TNF responses in endothelial cells. Circulation Research 125, 43–52. 10.1161/CIRCRESAHA.118.314601 [DOI] [PubMed] [Google Scholar]
- Qian, W., Kumar, N., Roginskaya, V., Fouquerel, E., Opresko, P. L., Shiva, S., Watkins, S. C., Kolodieznyi, D., Bruchez, M. P., & Van Houten, B. (2019). Chemoptogenetic damage to mitochondria causes rapid telomere dysfunction. Proceedings of the National Academy of Sciences of the United States of America, 116, 18435–18444. 10.1073/pnas.1910574116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Radzikowska, U., Ding, M., Tan, G., Zhakparov, D., Peng, Y., Wawrzyniak, P., Wang, M., Li, S., Morita, H., Altunbulakli, C., Reiger, M., Neumann, A. U., Lunjani, N., Traidl‐Hoffmann, C., Nadeau, K. C., O'Mahony, L., Akdis, C., & Sokolowska, M. (2020). Distribution of ACE2, CD147, CD26, and other SARS‐CoV‐2 associated molecules in tissues and immune cells in health and in asthma, COPD, obesity, hypertension, and COVID‐19 risk factors. Allergy, 75, 2829–2845. 10.1111/all.14429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raval, A. P., Martinez, C. C., Mejias, N. H., & de Rivero Vaccari, J. P. (2019). Sexual dimorphism in inflammasome‐containing extracellular vesicles and the regulation of innate immunity in the brain of reproductive senescent females. Neurochemistry International, 127, 29–37. 10.1016/j.neuint.2018.11.018 [DOI] [PubMed] [Google Scholar]
- Ritschka, B., Storer, M., Mas, A., Heinzmann, F., Ortells, M. C., Morton, J. P., Sansom, O. J., Zender, L., & Keyes, W. M. (2017). The senescence‐associated secretory phenotype induces cellular plasticity and tissue regeneration. Genes & Development, 31, 172–183. 10.1101/gad.290635.116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez, J. A., Orbe, J., Saenz‐Pipaon, G., Abizanda, G., Gebara, N., Radulescu, F., Azcarate, P. M., Alonso‐Perez, L., Merino, D., Prosper, F., Paramo, J. A., & Roncal, C. (2018). Selective increase of cardiomyocyte derived extracellular vesicles after experimental myocardial infarction and functional effects on the endothelium. Thrombosis Research, 170, 1–9. 10.1016/j.thromres.2018.07.030 [DOI] [PubMed] [Google Scholar]
- Russell, A. E., Sneider, A., Witwer, K. W., Bergese, P., Bhattacharyya, S. N., Cocks, A., Cocucci, E., Erdbrugger, U., Falcon‐Perez, J. M., Freeman, D. W., Gallagher, T. M., Hu, S., Huang, Y., Jay, S. M., Kano, S.‐.I., Lavieu, G., Leszczynska, A., Llorente, A. M., Lu, Q., … Vader, P. (2019). Biological membranes in EV biogenesis, stability, uptake, and cargo transfer: An ISEV position paper arising from the ISEV membranes and EVs workshop. Journal of Extracellular Vesicles, 8, 1684862. 10.1080/20013078.2019.1684862 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sagini, K., Urbanelli, L., Costanzi, E., Mitro, N., Caruso, D., Emiliani, C., & Buratta, S. (2018). Oncogenic H‐ras expression induces fatty acid profile changes in human fibroblasts and extracellular vesicles. International Journal of Molecular Sciences, 19, 3515. 10.3390/ijms19113515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salotti, J., & Johnson, P. F. (2019). Regulation of senescence and the SASP by the transcription factor C/EBPbeta. Experimental Gerontology, 128, 110752. 10.1016/j.exger.2019.110752 [DOI] [PubMed] [Google Scholar]
- Samaraweera, L., Adomako, A., Rodriguez‐Gabin, A., & McDaid, H. M. (2017). A novel indication for panobinostat as a senolytic drug in NSCLC and HNSCC. Scientific Reports, 7, 1900. 10.1038/s41598-017-01964-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandhof, C. A., Hoppe, S. O., Druffel‐Augustin, S., Gallrein, C., Kirstein, J., Voisine, C., & Nussbaum‐Krammer, C. (2020). Reducing INS‐IGF1 signaling protects against non‐cell autonomous vesicle rupture caused by SNCA spreading. Autophagy, 16, 878–899. 10.1080/15548627.2019.1643657 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santelli, A., Sun, I. O., Eirin, A., Abumoawad, A. M., Woollard, J. R., Lerman, A., Textor, S. C., Puranik, A. S., & Lerman, L. O. (2019). Senescent kidney cells in hypertensive patients release urinary extracellular vesicles. Journal of the American Heart Association, 8, e012584. 10.1161/JAHA.119.012584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saugstad, J. A., Lusardi, T. A., Van Keuren‐Jensen, K. R., Phillips, J. I., Lind, B., Harrington, C. A., McFarland, T. J., Courtright, A. L., Reiman, R. A., Yeri, A. S., Kalani, Y. S., Adelson, D., Arango, J., Nolan, J. P., Duggan, E., Messer, K., Akers, J. C., Galasko, D. R., Quinn, J. F., … Hochberg, F. H. (2017). Analysis of extracellular RNA in cerebrospinal fluid. Journal of Extracellular Vesicles, 6, 1317577. 10.1080/20013078.2017.1317577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schafer, M. J., Zhang, X., Kumar, A., Atkinson, E. J., Zhu, Y., Jachim, S., Mazula, D. L., Brown, A. K., Berning, M., Aversa, Z., Kotajarvi, A., Bruce, C. J., Greason, K. L., Suri, R. M., Tracy, R. P., Cummings, S. R., White, T. A., & LeBrasseur, N. K. (2020). The senescence‐associated secretome as an indicator of age and medical risk. JCI Insight, 5, e133668. 10.1172/jci.insight.133668 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt, J. R., Kliemt, S., Preissler, C., Moeller, S., von Bergen, M., Hempel, U., & Kalkhof, S. (2016). Osteoblast‐released matrix vesicles, regulation of activity and composition by sulfated and non‐sulfated glycosaminoglycans. Molecular & Cellular Proteomics, 15, 558–572. 10.1074/mcp.M115.049718 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwartz, R. E., Shokhirev, M. N., Andrade, L. R., Gutkind, J. S., Iglesias‐Bartolome, R., & Shadel, G. S. (2021). Insights into epithelial cell senescence from transcriptome and secretome analysis of human oral keratinocytes. Aging, 13, 4747–4777. 10.18632/aging.202658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Short, S., Fielder, E., Miwa, S., & von Zglinicki, T. (2019). Senolytics and senostatics as adjuvant tumour therapy. EBioMedicine, 41, 683–692. 10.1016/j.ebiom.2019.01.056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simay Demir, Y. D., Ozdemir, A., Sucularli, C., Benhur, E., & Ark, M. (2021). The implication of ROCK 2 as a potential senotherapeutic target via the suppression of the harmful effects of the SASP: Do senescent cancer cells really engulf the other cells? Cellular Signalling, 84, 110007. 10.1016/j.cellsig.2021.110007 [DOI] [PubMed] [Google Scholar]
- Song, L., Tang, S., Han, X., Jiang, Z., Dong, L., Liu, C., Liang, X., Dong, J., Qiu, C., Wang, Y., & Du, Y. (2019). KIBRA controls exosome secretion via inhibiting the proteasomal degradation of Rab27a. Nature Communication, 10, 1639. 10.1038/s41467-019-09720-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song, S., Lam, E. W., Tchkonia, T., Kirkland, J. L., & Sun, Y. (2020). Senescent cells: Emerging targets for human aging and age‐related diseases. Trends in Biochemical Sciences, 45, 578–592. 10.1016/j.tibs.2020.03.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soukas, A. A., Hao, H., & Wu, L. (2019). Metformin as antiaging therapy: Is it for everyone? Trends in Endocrinology & Metabolism, 30, 745–755. 10.1016/j.tem.2019.07.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spani, C., Suter, T., Derungs, R., Ferretti, M. T., Welt, T., Wirth, F., Gericke, C., Nitsch, R. M., & Kulic, L. (2015). Reduced beta‐amyloid pathology in an APP transgenic mouse model of Alzheimer's disease lacking functional B and T cells. Acta Neuropathologica Communications, 3, 71. 10.1186/s40478-015-0251-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart‐Ornstein, J., & Lahav, G. (2017). p53 dynamics in response to DNA damage vary across cell lines and are shaped by efficiency of DNA repair and activity of the kinase ATM. Science Signaling, 10, eaah6671. 10.1126/scisignal.aah6671 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Storci, G., De Carolis, S., Papi, A., Bacalini, M. G., Gensous, N., Marasco, E., Tesei, A., Fabbri, F., Arienti, C., Zanoni, M., Sarnelli, A., Santi, S., Olivieri, F., Mensà, E., Latini, S., Ferracin, M., Salvioli, S., Garagnani, P., Franceschi, C., & Bonafè, M. (2019). Genomic stability, anti‐inflammatory phenotype, and up‐regulation of the RNAseH2 in cells from centenarians. Cell Death & Differentiation, 26, 1845–1858. 10.1038/s41418-018-0255-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strzeszewska, A., Alster, O., Mosieniak, G., Ciolko, A., & Sikora, E. (2018). Insight into the role of PIKK family members and NF‐small ka, CyrillicB in DNAdamage‐induced senescence and senescence‐associated secretory phenotype of colon cancer cells. Cell Death & Disease, 9, 44. 10.1038/s41419-017-0069-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stuendl, A., Kunadt, M., Kruse, N., Bartels, C., Moebius, W., Danzer, K. M., Mollenhauer, B., & Schneider, A. (2016). Induction of alpha‐synuclein aggregate formation by CSF exosomes from patients with Parkinson's disease and dementia with Lewy bodies. Brain, 139, 481–494. 10.1093/brain/awv346 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su, T., Xiao, Y., Xiao, Y., Guo, Q., Li, C., Huang, Y., Deng, Q., Wen, J., Zhou, F., & Luo, X. H. (2019a). Bone marrow mesenchymal stem cells‐derived exosomal MiR‐29b‐3p regulates aging‐associated insulin resistance. ACS Nano, 13, 2450–2462. 10.1021/acsnano.8b09375 [DOI] [PubMed] [Google Scholar]
- Su, Y., Xu, C., Sun, Z., Liang, Y., Li, G., Tong, T., & Chen, J. (2019b). S100A13 promotes senescence‐associated secretory phenotype and cellular senescence via modulation of non‐classical secretion of IL‐1alpha. Aging (Albany NY), 11, 549–572. 10.18632/aging.101760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svensson, K. J., Christianson, H. C., Wittrup, A., Bourseau‐Guilmain, E., Lindqvist, E., Svensson, L. M., Morgelin, M., & Belting, M. (2013). Exosome uptake depends on ERK1/2‐heat shock protein 27 signaling and lipid Raft‐mediated endocytosis negatively regulated by caveolin‐1. The Journal of Biological Chemistry, 288, 17713–17724. 10.1074/jbc.M112.445403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taanman, J. W. (1999). The mitochondrial genome: Structure, transcription, translation and replication. Biochimica Et Biophysica Acta, 1410, 103–123. 10.1016/s0005-2728(98)00161-3 [DOI] [PubMed] [Google Scholar]
- Tai, H., Wang, Z., Gong, H., Han, X., Zhou, J., Wang, X., Wei, X., Ding, Y., Huang, N., Qin, J., Zhang, J., Wang, S., Gao, F., Chrzanowska‐Lightowlers, Z. M., Xiang, R., & Xiao, H. (2017). Autophagy impairment with lysosomal and mitochondrial dysfunction is an important characteristic of oxidative stress‐induced senescence. Autophagy, 13, 99–113. 10.1080/15548627.2016.1247143 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi, A., Loo, T. M., Okada, R., Kamachi, F., Watanabe, Y., Wakita, M., Watanabe, S., Kawamoto, S., Miyata, K., Barber, G. N., Ohtani, N., & Hara, E. (2018). Downregulation of cytoplasmic DNases is implicated in cytoplasmic DNA accumulation and SASP in senescent cells. Nature Communications, 9, 1249. 10.1038/s41467-018-03555-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takahashi, A., Okada, R., Nagao, K., Kawamata, Y., Hanyu, A., Yoshimoto, S., Takasugi, M., Watanabe, S., Kanemaki, M. T., Obuse, C., & Hara, E. (2017). Exosomes maintain cellular homeostasis by excreting harmful DNA from cells. Nature Communications, 8, 15287. 10.1038/ncomms15287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takasugi, M., Okada, R., Takahashi, A., Virya Chen, D., Watanabe, S., & Hara, E. (2017). Small extracellular vesicles secreted from senescent cells promote cancer cell proliferation through EphA2. Nature Communications, 8, 15729. 10.1038/ncomms15728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takeshita, H., Yamamoto, K., Nozato, S., Takeda, M., Fukada, S. I., Inagaki, T., Tsuchimochi, H., Shirai, M., Nozato, Y., Fujimoto, T., Imaizumi, Y., Yokoyama, S., Nagasawa, M., Hamano, Go, Hongyo, K., Kawai, T., Hanasaki‐Yamamoto, H., Takeda, S., Takahashi, T., … Rakugi, H. (2018). Angiotensin‐converting enzyme 2 deficiency accelerates and angiotensin 1–7 restores age‐related muscle weakness in mice. Journal of Cachexia Sarcopenia Muscle, 9, 975–986. 10.1002/jcsm.12334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tasdemir, N., Banito, A., Roe, J. S., Alonso‐Curbelo, D., Camiolo, M., Tschaharganeh, D. F., Huang, C. H., Aksoy, O., Bolden, J. E., Chen, C. C., Fennell, M., Thapar, V., Chicas, A., Vakoc, C. R., & Lowe, S. W. (2016). BRD4 connects enhancer remodeling to senescence immune surveillance. Cancer Discovery, 6, 612–629. 10.1158/2159-8290.CD-16-0217 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tesei, A., Arienti, C., Bossi, G., Santi, S., De Santis, I., Bevilacqua, A., Zanoni, M., Pignatta, S., Cortesi, M., Zamagni, A., Storci, G., Bonafè, M., Sarnelli, A., Romeo, A., Cavallo, C., Bartolazzi, A., Rossi, S., Soriani, A., & Strigari, L. (2021). TP53 drives abscopal effect by secretion of senescence‐associated molecular signals in non‐small cell lung cancer. Journal of Experimental & Clinical Cancer Research, 40, 89. 10.1186/s13046-021-01883-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tezze, C., Romanello, V., Desbats, M. A., Fadini, G. P., Albiero, M., Favaro, G., Ciciliot, S., Soriano, M. E., Morbidoni, V., Cerqua, C., Loefler, S., Kern, H., Franceschi, C., Salvioli, S., Conte, M., Blaauw, B., Zampieri, S., Salviati, L., Scorrano, L., & Sandri, M. (2017). Age‐associated loss of OPA1 in muscle impacts muscle mass, metabolic homeostasis, systemic inflammation, and epithelial senescence. Cell Metabolism, 25, 1374–1389 e1376. 10.1016/j.cmet.2017.04.021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thabet, E., Yusuf, A., Abdelmonsif, D. A., Nabil, I., Mourad, G., & Mehanna, R. A. (2020). Extracellular vesicles miRNA‐21: A potential therapeutic tool in premature ovarian dysfunction. Molecular Human Reproduction, 26, 906–919. 10.1093/molehr/gaaa068 [DOI] [PubMed] [Google Scholar]
- Thurn, K. T., Thomas, S., Raha, P., Qureshi, I., & Munster, P. N. (2013). Histone deacetylase regulation of ATM‐mediated DNA damage signaling. Molecular Cancer Therapeutics, 12, 2078–2087. 10.1158/1535-7163.MCT-12-1242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian, C., Hu, G., Gao, L., Hackfort, B. T., & Zucker, I. H. (2020). Extracellular vesicular MicroRNA‐27a* contributes to cardiac hypertrophy in chronic heart failure. The Journal of Molecular and Cellular Cardiology, 143, 120–131. 10.1016/j.yjmcc.2020.04.032 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian, T., Zhu, Y. L., Zhou, Y. Y., Liang, G. F., Wang, Y. Y., Hu, F. H., & Xiao, Z. D. (2014). Exosome uptake through clathrin‐mediated endocytosis and macropinocytosis and mediating miR‐21 delivery. Journal of Biological Chemistry, 289, 22258–22267. 10.1074/jbc.M114.588046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tkach, M., & Thery, C. (2016). Communication by extracellular vesicles: Where we are and where we need to go. Cell, 164, 1226–1232. 10.1016/j.cell.2016.01.043 [DOI] [PubMed] [Google Scholar]
- Tofino‐Vian, M., Guillen, M. I., Perez Del Caz, M. D., Castejon, M. A., & Alcaraz, M. J. (2017). Extracellular vesicles from adipose‐derived mesenchymal stem cells downregulate senescence features in osteoarthritic osteoblasts. Oxidative Medicine and Cellular Longevity, 2017, 7197598. 10.1155/2017/7197598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ungvari, Z., Tarantini, S., Donato, A. J., Galvan, V., & Csiszar, A. (2018). Mechanisms of vascular aging. Circulation Research, 123, 849–867. 10.1161/CIRCRESAHA.118.311378 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vizioli, M. G., Liu, T., Miller, K. N., Robertson, N. A., Gilroy, K., Lagnado, A. B., Perez‐Garcia, A., Kiourtis, C., Dasgupta, N., Lei, X., Kruger, P. J., Nixon, C., Clark, W., Jurk, D., Bird, T. G., Passos, J. F., Berger, S. L., Dou, Z., & Adams, P. D. (2020). Mitochondria‐to‐nucleus retrograde signaling drives formation of cytoplasmic chromatin and inflammation in senescence. Genes & Development, 34, 428–445. 10.1101/gad.331272.119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Kobbe, C. (2019). Targeting senescent cells: Approaches, opportunities, challenges. Aging (Albany NY), 11, 12844–12861. 10.18632/aging.102557 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, C., Vegna, S., Jin, H., Benedict, B., Lieftink, C., Ramirez, C., de Oliveira, R. L., Morris, B., Gadiot, J., Wang, W., Chatinier, A., Wang, L., Gao, D., Evers, B., Jin, G., Xue, Z., Schepers, A., Jochems, F., Sanchez, A. M., … Bernards, R. (2019). Inducing and exploiting vulnerabilities for the treatment of liver cancer. Nature, 574, 268–272. 10.1038/s41586-019-1607-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, R., Yu, Z., Sunchu, B., Shoaf, J., Dang, I., Zhao, S., Caples, K., Bradley, L., Beaver, L. M., Ho, E., Löhr, C. V., & Perez, V. I. (2017a). Rapamycin inhibits the secretory phenotype of senescent cells by a Nrf2‐independent mechanism. Aging Cell, 16, 564–574. 10.1111/acel.12587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, S., Zheng, Y., Li, J., Yu, Y., Zhang, W., Song, M., Liu, Z., Min, Z., Hu, H., Jing, Y., He, X., Sun, L., Ma, L., Esteban, C. R., Chan, P., Qiao, J., Zhou, Qi, Belmonte, J. C. I., Qu, J., & Liu, G.‐.H. (2020). Single‐cell transcriptomic atlas of primate ovarian aging. Cell, 180, 585–600 e519. 10.1016/j.cell.2020.01.009 [DOI] [PubMed] [Google Scholar]
- Wang, W., Wang, L., Ruan, L., Oh, J., Dong, X., Zhuge, Q., & Su, D. M. (2018). Extracellular vesicles extracted from young donor serum attenuate inflammaging via partially rejuvenating aged T‐cell immunotolerance. FASEB Journal, 32, fj201800059R. 10.1096/fj.201800059R [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, Y., Balaji, V., Kaniyappan, S., Kruger, L., Irsen, S., Tepper, K., Chandupatla, R., Maetzler, W., Schneider, A., Mandelkow, E., & Mandelkow, E. M. (2017b). The release and trans‐synaptic transmission of Tau via exosomes. Molecular Neurodegeneration, 12, 5. 10.1186/s13024-016-0143-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, Z., Deng, Z., Dahmane, N., Tsai, K., Wang, P., Williams, D. R., Kossenkov, A. V., Showe, L. C., Zhang, R., Huang, Q., Conejo‐Garcia, J. R., & Lieberman, P. M. (2015). Telomeric repeat‐containing RNA (TERRA) constitutes a nucleoprotein component of extracellular inflammatory exosomes. Proceedings of the National Academy of Sciences of the United States of America, 112, E6293–6300. 10.1073/pnas.1505962112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang, Z., & Lieberman, P. M. (2016). The crosstalk of telomere dysfunction and inflammation through cell‐free TERRA containing exosomes. RNA Biology, 13, 690–695. 10.1080/15476286.2016.1203503 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe, S., Kawamoto, S., Ohtani, N., & Hara, E. (2017). Impact of senescence‐associated secretory phenotype and its potential as a therapeutic target for senescence‐associated diseases. Cancer Science. 108, 563–569. 10.1111/cas.13184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei, D., Zhan, W., Gao, Y., Huang, L., Gong, R., Wang, W., Zhang, R., Wu, Y., Gao, S., & Kang, T. (2020a). RAB31 marks and controls an ESCRT‐independent exosome pathway. Cell Research, 31, 157–177. 10.1038/s41422-020-00409-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei, F., Wang, A., Wang, Q., Han, W., Rong, R., Wang, L., Liu, S., Zhang, Y., Dong, C., & Li, Y. (2020b). Plasma endothelial cells‐derived extracellular vesicles promote wound healing in diabetes through YAP and the PI3K/Akt/mTOR pathway. Aging (Albany NY), 12, 12002–12018. 10.18632/aging.103366 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weilner, S., Schraml, E., Wieser, M., Messner, P., Schneider, K., Wassermann, K., Micutkova, L., Fortschegger, K., Maier, A. B., Westendorp, R., Resch, H., Wolbank, S., Redl, H., Jansen‐Dürr, P., Pietschmann, P., Grillari‐Voglauer, R., & Grillari, J. (2016). Secreted microvesicular miR‐31 inhibits osteogenic differentiation of mesenchymal stem cells. Aging Cell, 15, 744–754. 10.1111/acel.12484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wiley, C. D., Velarde, M. C., Lecot, P., Liu, S., Sarnoski, E. A., Freund, A., Shirakawa, K., Lim, H. W., Davis, S. S., Ramanathan, A., Gerencser, A. A., Verdin, E., & Campisi, J. (2016). Mitochondrial dysfunction induces senescence with a distinct secretory phenotype. Cell Metabolism, 23, 303–314. 10.1016/j.cmet.2015.11.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Willis, C. M., Nicaise, A. M., Bongarzone, E. R., Givogri, M., Reiter, C. R., Heintz, O., Jellison, E. R., Sutter, P. A., TeHennepe, G., Ananda, G., Vella, A. T., & Crocker, S. J. (2020). Astrocyte support for oligodendrocyte differentiation can be conveyed via extracellular vesicles but diminishes with age. Scientific Reports, 10, 828. 10.1038/s41598-020-57663-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wolock, S. L., Krishnan, I., Tenen, D. E., Matkins, V., Camacho, V., Patel, S., Agarwal, P., Bhatia, R., Tenen, D. G., Klein, A. M., & Welner, R. S. (2019). Mapping distinct bone marrow niche populations and their differentiation paths. Cell Reproduction, 28, 302–311 e305. 10.1016/j.celrep.2019.06.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu, S. F., Noren Hooten, N., Freeman, D. W., Mode, N. A., Zonderman, A. B., & Evans, M. K. (2020). Extracellular vesicles in diabetes mellitus induce alterations in endothelial cell morphology and migration. Journal of Translational Medicine, 18, 230. 10.1186/s12967-020-02398-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia, Y., Zhang, G., Han, C., Ma, K., Guo, X., Wan, F., Kou, L., Yin, S., Liu, L., Huang, J., Xiong, N., & Wang, T. (2019). Microglia as modulators of exosomal alpha‐synuclein transmission. Cell Death & Disease, 10, 174. 10.1038/s41419-019-1404-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu, M., Pirtskhalava, T., Farr, J. N., Weigand, B. M., Palmer, A. K., Weivoda, M. M., Inman, C. L., Ogrodnik, M. B., Hachfeld, C. M., & Fraser, D. G. et al. (2018a).Senolytics improve physical function and increase lifespan in old age. Nature Medicine, 24, 1246–1256. 10.1038/s41591-018-0092-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu, M., Tchkonia, T., Ding, H., Ogrodnik, M., Lubbers, E. R., Pirtskhalava, T., White, T. A., Johnson, K. O., Stout, M. B., Mezera, V., Giorgadze, N., Jensen, M. D., LeBrasseur, N. K., & Kirkland, J. L. (2015). JAK inhibition alleviates the cellular senescence‐associated secretory phenotype and frailty in old age. Proceedings of the National Academy of Sciences of the United States of America, 112, E6301–6310. 10.1073/pnas.1515386112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu, P., Xin, Y., Zhang, Z., Zou, X., Xue, K., Zhang, H., Zhang, W., & Liu, K. (2020). Extracellular vesicles from adipose‐derived stem cells ameliorate ultraviolet B‐induced skin photoaging by attenuating reactive oxygen species production and inflammation. Stem Cell Research & Therapy, 11, 264. 10.1186/s13287-020-01777-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu, R., Shen, X., Si, Y., Fu, Y., Zhu, W., Xiao, T., Fu, Z., Zhang, P., Cheng, J., & Jiang, H. (2018b). MicroRNA‐31a‐5p from aging BMSCs links bone formation and resorption in the aged bone marrow microenvironment. Aging Cell, 17, e12794. 10.1111/acel.12794 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu, R., Zhang, F., Chai, R., Zhou, W., Hu, M., Liu, B., Chen, X., Liu, M., Xu, Q., Liu, N., & Liu, S. (2019). Exosomes derived from pro‐inflammatory bone marrow‐derived mesenchymal stem cells reduce inflammation and myocardial injury via mediating macrophage polarization. Journal of Cellular and Molecular Medicine, 23, 7617–7631. 10.1111/jcmm.14635 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang, C., Shani, S., Tahiri, H., Ortiz, C., Gu, M., Lavoie, J. C., Croteau, S., & Hardy, P. (2020). Extracellular microparticles exacerbate oxidative damage to retinal pigment epithelial cells. Experimental Cell Research 390, 111957. 10.1016/j.yexcr.2020.111957 [DOI] [PubMed] [Google Scholar]
- Yang, J., Yu, X. F., Li, Y. Y., Xue, F. T., & Zhang, S. (2019). Decreased HSP70 expression on serum exosomes contributes to cardiac fibrosis during senescence. European Review for Medical and Pharmacological Sciences, 23, 3993–4001. 10.26355/eurrev_201905_17829 [DOI] [PubMed] [Google Scholar]
- Yang, X., Zhou, Y., Peng, S., Wu, L., Lin, H. Y., Wang, S., & Wang, H. (2012). Differentially expressed plasma microRNAs in premature ovarian failure patients and the potential regulatory function of mir‐23a in granulosa cell apoptosis. Reproduction (Cambridge, England), 144, 235–244. 10.1530/REP-11-0371 [DOI] [PubMed] [Google Scholar]
- Yao, Z., Murali, B., Ren, Q., Luo, X., Faget, D. V., Cole, T., Ricci, B., Thotala, D., Monahan, J., van Deursen, J. M., Baker, D., Faccio, R., Schwarz, J. K., & Stewart, S. A. (2020). Therapy‐induced senescence drives bone loss. Cancer Research, 80, 1171–1182. 10.1158/0008-5472.CAN-19-2348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin, Y., Li, W., Son, Y. O., Sun, L., Lu, J., Kim, D., Wang, X., Yao, H., Wang, L., Pratheeshkumar, P., Hitron, A. J., Luo, J., Gao, N., Shi, X., Zhang, Z., & (2013). Quercitrin protects skin from UVB‐induced oxidative damage. Toxicology and Applied Pharmacology 269, 89–99. 10.1016/j.taap.2013.03.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ying, W., Riopel, M., Bandyopadhyay, G., Dong, Y., Birmingham, A., Seo, J. B., Ofrecio, J. M., Wollam, J., Hernandez‐Carretero, A., Fu, W., Li, P., Olefsky, J. M. (2017). Adipose tissue macrophage‐derived exosomal miRNAs can modulate in vivo and in vitro insulin sensitivity. Cell, 171, 372–384 e312. 10.1016/j.cell.2017.08.035 [DOI] [PubMed] [Google Scholar]
- Yoshida, M., Satoh, A., Lin, J. B., Mills, K. F., Sasaki, Y., Rensing, N., Wong, M., Apte, R. S., & Imai, S. I. (2019). Extracellular vesicle‐contained eNAMPT delays aging and extends lifespan in mice. Cell Metabolism, 30, 329–342.e325. 10.1016/j.cmet.2019.05.015 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida, S., Nakagami, H., Hayashi, H., Ikeda, Y., Sun, J., Tenma, A., Tomioka, H., Kawano, T., Shimamura, M., Morishita, R., & Rakugi, H. (2020). The CD153 vaccine is a senotherapeutic option for preventing the accumulation of senescent T cells in mice. Nature Communications, 11, 2482. 10.1038/s41467-020-16347-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu, B., Ma, J., Li, J., Wang, D., Wang, Z., & Wang, S. (2020). Mitochondrial phosphatase PGAM5 modulates cellular senescence by regulating mitochondrial dynamics. Nature Communications, 11, 2549. 10.1038/s41467-020-16312-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu, X., Riley, T., & Levine, A. J. (2009). The regulation of the endosomal compartment by p53 the tumor suppressor gene. FEBS Journal, 276, 2201–2212. 10.1111/j.1742-4658.2009.06949.x [DOI] [PubMed] [Google Scholar]
- Yu, Y., Du, H., Wei, S., Feng, L., Li, J., Yao, F., Zhang, M., Hatch, G. M., & Chen, L. (2018). Adipocyte‐derived exosomal MiR‐27a induces insulin resistance in skeletal muscle through repression of PPARgamma. Theranostics, 8, 2171–2188. 10.7150/thno.22565 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhai, M., Zhu, Y., Yang, M., & Mao, C. (2020). Human mesenchymal stem cell derived exosomes enhance cell‐free bone regeneration by altering their miRNAs profiles. Advanced science (Weinheim, Baden‐Wurttemberg, Germany), 7, 2001334. 10.1002/advs.202001334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, C., Wang, J., Ma, X., Wang, W., Zhao, B., Chen, Y., Chen, C., & Bihl, J. C. (2018a). ACE2‐EPC‐EXs protect ageing ECs against hypoxia/reoxygenation‐induced injury through the miR‐18a/Nox2/ROS pathway. Journal of Cellular and Molecular Medicine, 22, 1873–1882. 10.1111/jcmm.13471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, K., Yu, L., Li, F. R., Li, X., Wang, Z., Zou, X., Zhang, C., Lv, K., Zhou, B., Mitragotri, S., & Chen, M. (2020). Topical application of exosomes derived from human umbilical cord mesenchymal stem cells in combination with sponge spicules for treatment of photoaging. Int J Nanomedicine, 15, 2859–2872. 10.2147/IJN.S249751 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, S., Eitan, E., Wu, T. Y., & Mattson, M. P. (2018b). Intercellular transfer of pathogenic alpha‐synuclein by extracellular vesicles is induced by the lipid peroxidation product 4‐hydroxynonenal. Neurobiology of Aging, 61, 52–65. 10.1016/j.neurobiolaging.2017.09.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, S., Yu, X., Zhang, Y., Xue, X., Yu, Q., Zha, Z., Gogol, M., Workman, J. L., & Li, S. (2021). Metabolic regulation of telomere silencing by SESAME complex‐catalyzed H3T11 phosphorylation. Nature Communications, 12, 594. 10.1038/s41467-020-20711-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang, Y., Kim, M. S., Jia, B., Yan, J., Zuniga‐Hertz, J. P., Han, C., & Cai, D. (2017). Hypothalamic stem cells control ageing speed partly through exosomal miRNAs. Nature, 548, 52–57. 10.1038/nature23282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao, J., Zhang, L., Lu, A., Han, Y., Colangelo, D., Bukata, C., Scibetta, A., Yousefzadeh, M. J., Li, X., Gurkar, A. U., McGowan, S. J., Angelini, L., O'Kelly, R., Li, H., Corbo, L., Sano, T., Nick, H., Pola, E.,Pilla, S. P. S., … Robbins, P. D. (2020). ATM is a key driver of NF‐κB‐dependent DNA‐damage‐induced senescence, stem cell dysfunction and aging. Aging (Albany NY), 12, 4688–4710. 10.18632/aging.102863 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao, R. C., & Stambler, I. (2020). The urgent need for international action for antiaging and disease prevention. Aging Disease, 11, 212–215. 10.14336/AD.2019.1230 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng, L. D., Linarelli, L. E., Liu, L., Wall, S. S., Greenawald, M. H., Seidel, R. W., Estabrooks, P. A., Almeida, F. A., & Cheng, Z. (2015). Insulin resistance is associated with epigenetic and genetic regulation of mitochondrial DNA in obese humans. Clinical Epigenetics, 7, 60. 10.1186/s13148-015-0093-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng, Q., Liu, P., Gao, G., Yuan, J., Wang, P., Huang, J., Xie, L., Lu, X., Di, F., Tong, T., Chen, J., Lu, Z., Guan, J., & Wang, G. (2019). Mitochondrion‐processed TERC regulates senescence without affecting telomerase activities. Protein Cell, 10, 631–648. 10.1007/s13238-019-0612-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou, P., Wu, G., Zhang, P., Xu, R., Ge, J., Fu, Y., Zhang, Y., Du, Y., Ye, J., Cheng, J., & Jiang, H. (2016). SATB2‐Nanog axis links age‐related intrinsic changes of mesenchymal stem cells from craniofacial bone. Aging (Albany NY), 8, 2006–2011. 10.18632/aging.101041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou, Q. F., Cai, Y. Z., & Lin, X. J. (2020). The dual character of exosomes in osteoarthritis: Antagonists and therapeutic agents. Acta Biomaterial, 105, 15–25. 10.1016/j.actbio.2020.01.040 [DOI] [PubMed] [Google Scholar]
- Zhou, Y., Wang, X., Song, M., He, Z., Cui, G., Peng, G., Dieterich, C., Antebi, A., Jing, N., & Shen, Y. (2019). A secreted microRNA disrupts autophagy in distinct tissues of Caenorhabditis elegans upon ageing. Nature Communications, 10, 4827. 10.1038/s41467-019-12821-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu, B., Zhang, L., Liang, C., Liu, B., Pan, X., Wang, Y., Zhang, Y., Zhang, Y., Xie, W., Yan, B., Liu, F., Yip, H ‐ K., Yu, X ‐ Y., & Li, Y. (2019). Stem cell‐derived exosomes prevent aging‐induced cardiac dysfunction through a novel exosome/lncRNA MALAT1/NF‐kappaB/TNF‐alpha signaling pathway. Oxidative Medicine and Cellular Longevity, 2019, 9739258. 10.1155/2019/9739258 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu, Q., Ghoshal, S., Rodrigues, A., Gao, S., Asterian, A., Kamenecka, T. M., Barrow, J. C., & Chakraborty, A. (2016). Adipocyte‐specific deletion of Ip6k1 reduces diet‐induced obesity by enhancing AMPK‐mediated thermogenesis. Journal of Clinical Investigation, 126, 4273–4288. 10.1172/JCI85510 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu, Y., Doornebal, E. J., Pirtskhalava, T., Giorgadze, N., Wentworth, M., Fuhrmann‐Stroissnigg, H., Niedernhofer, L. J., Robbins, P. D., Tchkonia, T., & Kirkland, J. L. (2017). New agents that target senescent cells: The flavone, fisetin, and the BCL‐X(L) inhibitors, A1331852 and A1155463. Aging (Albany NY), 9, 955–963. 10.18632/aging.101202 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu, Y., Tchkonia, T., Pirtskhalava, T., Gower, A. C., Ding, H., Giorgadze, N., Palmer, A. K., Ikeno, Y., Hubbard, G. B., Lenburg, M., O'Hara, S. P., LaRusso, N. F., Miller, J. D., Roos, C. M., Verzosa, G. C., LeBrasseur, N. K., Wren, J. D., Farr, J. N., Khosla, S., … Kirkland, J. L. (2015). The Achilles' heel of senescent cells: From transcriptome to senolytic drugs. Aging Cell, 14, 644–658. 10.1111/acel.12344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zurgil, U., Ben‐Ari, A., Atias, K., Isakov, N., Apte, R., & Livneh, E. (2014). PKCeta promotes senescence induced by oxidative stress and chemotherapy. Cell Death & Disease, 5, e1531. 10.1038/cddis.2014.481 [DOI] [PMC free article] [PubMed] [Google Scholar]


