Abstract
The study aimed to determine clinical presentation, contributing factors, medical and surgical management, and outcome of patients with coronavirus disease 2019 (COVID-19)-associated mucormycosis (CAM). A cross-sectional, single-center study was conducted on patients receiving multidisciplinary treatment for mucormycosis following the second wave of COVID-19 pandemic from April to June 2021 in India. Clinicoepidemiological factors were analyzed, 30-day overall survival and disease-specific survival were determined, and t-test was used to determine the statistical significance. A total of 215 patients were included in the study, the cases were stratified into sino-nasal 95 (44.2%), sino-naso-orbital 32 (14.9%), sino-naso-palatal 55 (25.6%), sino-naso-cerebral 12 (5.6%), sino-naso-orbito-cerebral 16 (7.4%), and sino-naso-orbito-palato-cerebral 5 (2.3%) based on their presentation. A multidisciplinary team treated patients by surgical wound debridement and medical therapy with broad-spectrum antibiotics and amphotericin B. Across all disease stages, cumulative 30-day disease-specific survival is 94% (p < 0.001, intergroup comparison, Breslow (generalized Wilcoxon) CI 95%) and overall 30-day survival is 87.9% (p < 0.001, intergroup comparison, Breslow (generalized Wilcoxon) CI 95%) (censored). Early identification, triaging, and proper multidisciplinary team management with systemic antifungals, surgical debridement, and control of comorbidities lead to desirable outcomes in COVID-associated mucormycosis. The patients with intracranial involvement have a higher chance of mortality compared to the other group.
Supplementary Information
The online version contains supplementary material available at 10.1007/s12262-021-03134-0.
Keywords: Mucormycosis, COVID-19, Opportunistic infection, Amphotericin B, SARS CoV2
Introduction
COVID-19 is one of the most severe and crippling pandemics that humanity has faced in recent times. The B.1.617.1 variant has rapidly spread throughout India and several other countries throughout the world [1]. SARS CoV2 has significantly affected people with comorbidities, specifically diabetes, chronic kidney disease, hypertension, etc. Also, there is a stark rise in the usage of steroids and immune modulators to curtail the collateral damage by the immune system on the pulmonary parenchyma. This led to the outbreak of new opportunistic infection mucormycosis, mainly in the post-COVID patients. These secondary fungal infections or coinfections are critical challenges increasing the patient’s morbidity and mortality. India has reported the highest number of mucormycosis cases globally, especially following the second wave of the pandemic. Before this surge, the estimated prevalence of mucormycosis in India is seventy times more than in the other nations [2]. It is a known fact that diabetes mellitus, ketoacidosis, and steroid use predispose to opportunistic infections. Along with this, COVID-19 forms the unholy trinity of risk factors.
Nearly twenty-eight thousand cases, with almost three thousand deaths all over India, have been reported [3]. Owing to the rapid surge in patients, mucormycosis was declared an epidemic by the Government of India. This article discusses the clinicoepidemiologic characteristics of mucormycosis and its multidisciplinary team management as a fallout of COVID-19.
Methods
The study is a prospective, single-center, interventional study at All India Institute of Medical Sciences, Raipur, Chhattisgarh, India. This cross-sectional study has included the patients with a proven or suspected diagnosis of mucormycosis in a COVID-positive or suspect COVID disease individual during the pandemic and the accrual was done on admission. A total of two hundred and fifteen patients admitted and treated at our center from 1st May to 15th July 2021 were included in the data analysis. A detailed proforma to record the demographic data, clinical presentation, investigations, points in pathogenesis, and pathogenesis of the management in each patient. The data analysis was performed using IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY, IBM Corp. The Institutional Research Cell approval was obtained for the study under reference OW/RC/AIIMS-RPR/2021/571 dated 27th May 2021. A detailed workflow is attached as Supplement sheet 1 (Workflow).
The patients were planned for appropriate surgical debridement as per the disease stage. Supplement Table 1 describes the surgical plan according to the disease stage. Following ablative surgery, a definitive permanent reconstruction procedure was deferred due to the possibility of disease recurrence and contaminated surgical field. A temporary palatal obturator was placed based on the Aramany classification for the palatal defects. The sino-nasal cavity was tightly packed with povidone iodine-soaked ribbon gauze, which was removed after 48 h. The anesthetists faced issues such as post-COVID fibrotic lungs, impaired ventilation and perfusion, electrolyte imbalance, autonomic dysfunction, renal dysfunction, stress cardiomyopathy, post-COVID thromboembolism, and intraoperative hypotension. High-risk patients were started on a prophylactic dose of low molecular weight heparin from the 1st postoperative day to reduce the incidence of thromboembolism [4]. The patients were started on isotonic saline nasal douching to clear the slough and debris upon nasal pack removal. Extensive nasal crusting was debrided in the minor operation theater. Early recovery after surgery (ERAS) guidelines were strictly followed. All patients received pulmonary conditioning exercises, incentive spirometry, and chest physiotherapy. The patients were considered for discharge if they were clinically stable with reasonable control of comorbidities, completed their target dosage of amphotericin B, and had no radiological evidence of disease. The patients were prescribed oral posaconazole tablets at the discharge time, 600 mg loading dose on the first day, followed by 300 mg once daily for 2 to 3 weeks.
Results
A total of two hundred and fifteen patients were admitted for the suspicion of mucormycosis following COVID-19. A total of one hundred eighty-eight patients (87.4%) were diagnosed with mucormycosis by KOH mount (fungal elements/broad aseptate hyphae). Only 25% were culture-positive due to antifungal initiation. Rhizopus species were isolated in 79% of cases, followed by Lichthemia 13% and Apophysomyces 5%. Refer to Supplement Table 2 for descriptive statistics.
Disease Staging and Surgery
Based on the disease presentation, the mucormycosis was classified as sino-nasal 95 (44.2%), sino-naso-orbital 32 (14.9%), Sino-naso-palatal 55 (25.6%), sino-naso-cerebral 12 (5.6%), sino-naso-orbito-cerebral 16 (7.4%), and sino-naso-orbito-palato-cerebral 5 (2.3%) for effective disease stratification and surgical planning (Supplement Fig. 1).
One hundred and ninety patients have been operated (Supplement Fig. 2); a combined (open and endoscopic) approach was commonly used for adequate exposure and disease clearance. The combined approach was utilized due to disease in the maxillary wall with accompanied bony or soft tissue involvement. A purely endoscopic approach was considered in patients with limited disease, which was amenable for complete clearance via endoscopic approach. The debridement entails at least medial maxillectomy (for all sino-nasal cases), infrastructure maxillectomy (for palatal involvement), and a palatal obturator was placed to reconstruct the defect. Medial maxillectomy with orbital exenteration was performed in cases with orbital involvement. The frontal or frontoparietal craniotomy or transnasal approach was utilized for intracranial debridement, along with intraoperative CSF leak repair (if necessary) with tissue glue and knitted collagen (see Supplement Photographs).
Intraoperative Observations
Maxillary sinus was the most affected sinus in our series (70%), followed by ethmoids (60%), sphenoid (40%), and frontal (12%). The majority of patients (60%) had more than one sinus involvement. Middle turbinate was most involved (65%), followed by inferior turbinate (20%) and, lastly, superior turbinate (5%).
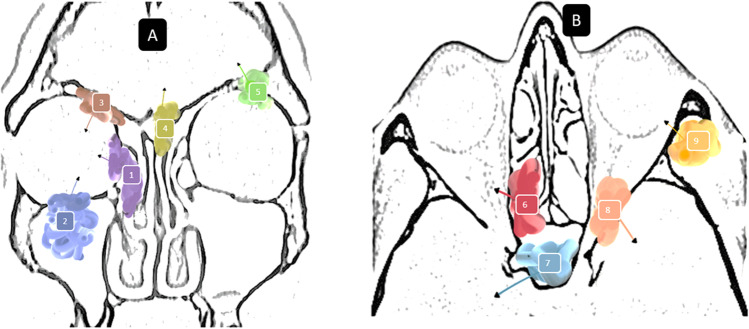
Palatal involvement was seen in sixty cases, of which palatal/gingivobuccal bulge along with loosening of teeth was the most common finding, followed by palatal perforation with debris noticed in the oral cavity 15.7%. Only eight (3.7%) patients had cheek skin involvement. Twenty-two patients had involvement of pterygopalatine fossa. In nineteen patients, osteomyelitis of maxillary bone was seen; in twelve patients, pale nonbleeding sequestrum was found. Infratemporal fossa involvement was seen in fourteen (6.5%) patients. Figure 1 depicts the various routes of spread of mucormycosis into the orbit and brain.
Fig. 1.
Routes of spread. Panel A 1, into the orbit via anterior ethmoids and lamina papyracea; 2, into the orbit via erosion of superior maxillary wall; 3, into the orbit via floor of the frontal sinus; 4, intracranial spread via erosion of cribriform and frontal roof; 5, intracranial extension via erosion of orbital roof. Panel B 6, into the orbital apex and optic nerve via the posterior ethmoids; 7, intracranial and cavernous sinus extension via sphenoid erosion; 8, intracranial extension via superior orbital fissure; 9, intraorbital extension via erosion of lateral orbital wall from the disease in the infratemporal fossa
Intraoperative CSF leak repair with knitted collagen matrix with tissue glue was done in sixteen patients. Ophthalmic findings included ptosis (twelve patients, out of these twelve, one had bilateral ptosis), proptosis (thirteen patients), restricted eye movements, abnormal or absent pupillary reaction, vision deterioration, or vision loss (three patients with bilateral complete vision loss). The various causes of vision loss included fungal corneal ulcer (two patients), CRAO (five patients, in two bilateral), orbital apex syndrome (fifteen patients), cavernous sinus thrombosis (seven patients), and optic neuritis (six patients). Complete ophthalmoplegia was seen in fifteen patients.
In our study, intracranial involvement in the frontal lobe was seen in nine patients and the anterior part of the medial temporal lobe in seven patients. Cavernous sinus involvement was seen in seven patients. Facial nerve involvement was seen in twelve patients due to skull base involvement.
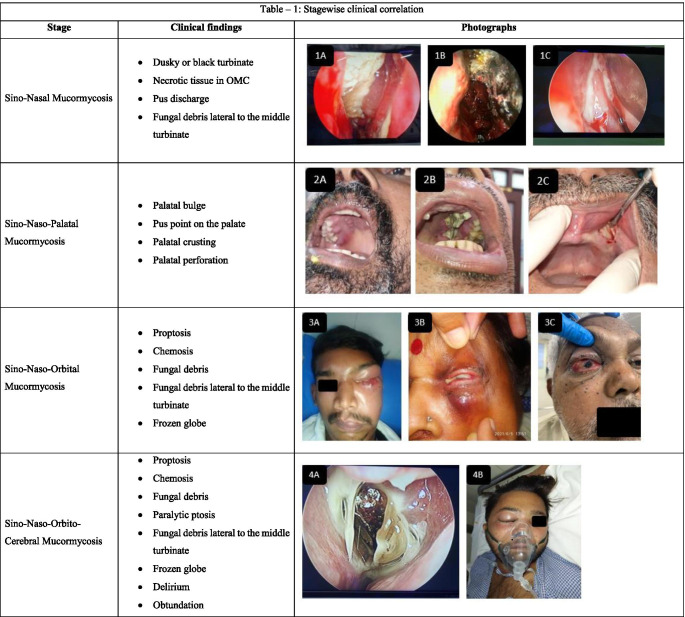
Table 1 shows the correlation of the disease stage with the intraoperative finding.
Table 1.
Stagewise clinical correlation
Panel 1A fungal debris in osteomeatal complex, 1B necrotic and black middle turbinate, 1C pus discharge in nasal cavity. Panel 2A palatal bulge, 2B palatal perforation with fungal debris, 2C gingival ulceration. Panel 3A orbital cellulitis, 3B frozen globe with pus discharge, 3C proptosis and chemosis. Panel 4A fungal debris lateral to middle turbinate, 4B preseptal cellulitis with obtundation. Panel 5A cutaneous involvement, 5B and 5C bilateral paralytic ptosis. Stage sino-naso-orbito-palato-cerebral presents with a combination of sino-naso-orbito-cerebral and sino-naso-palatal stages
Only two out of one hundred and ninety operated patients (0.93%) had craniotomy wound infection, managed by wound exploration under local anesthesia (Clavein Dindo Grade IIIa), with complete resolution following exploration. None of the other patients had any postoperative surgical site complications.
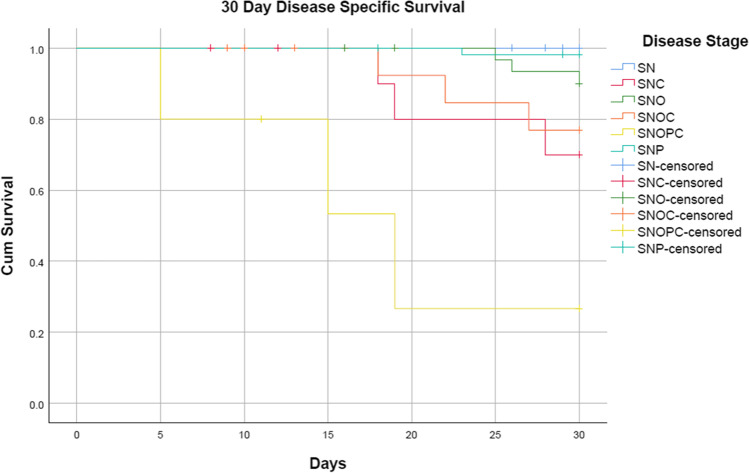
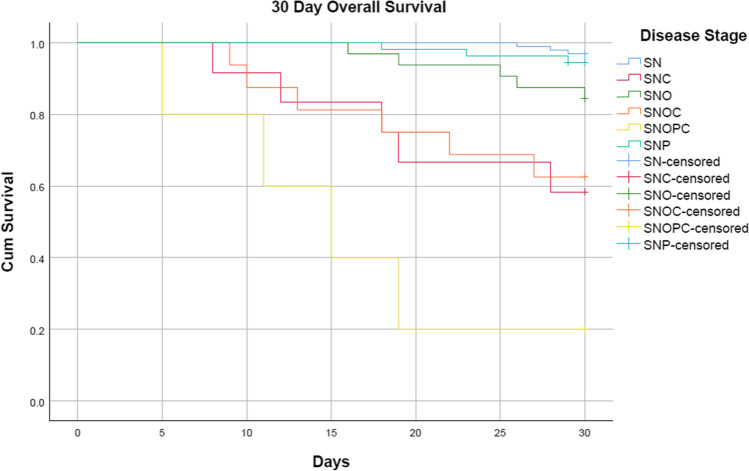
Cumulative 30-day disease-specific survival is 94% (p < 0.001, intergroup comparison, Breslow (generalized Wilcoxon) CI 95%) and overall 30-day survival is 87.9% (p < 0.001, intergroup comparison, Breslow (generalized Wilcoxon) CI 95%) (censored). The terminal stage sino-naso-orbito-palato-cerebral was the deadliest with an overall mortality of 80% and disease-specific mortality of 60%, followed by sino-naso-orbito-cerebral and sino-naso-cerebral with an overall mortality of 37.5% and 41.7%, respectively, and 18.7% and 25% dying due to disease. Our study also shows that cerebral involvement leads to higher rates of death due to disease. The least mortality was seen with sino-nasal disease, with an overall mortality of 3.2% and none dying due to disease.
Table 2 describes the stagewise 30-day disease-specific and overall survival. The Kaplan–Meier survival plots are depicted in Fig. 2 Disease-specific survival and Fig. 3 Overall Survival.
Table 2.
30-day disease specific and overall survival (stagewise)
| Disease stage | Total cases | 30-day disease-specific survival (DSS) | 30-day overall survival |
|---|---|---|---|
| SNOPC | 5 | 40% | 20% |
| SNC | 12 | 75% | 58.3% |
| SNOC | 16 | 81.3% | 62.5% |
| SNO | 32 | 90.6% | 84.4% |
| SNP | 55 | 98.2% | 94.5% |
| SN | 95 | 100.0% | 96.8% |
| Overall | 215 | 94% | 87.9% |
SN sino-nasal, SNO sino-naso-orbital, SNP sino-naso-palatal, SNC sino-naso-cerebral, SNOC sino-naso-orbito-cerebral, SNOPC sino-naso-orbito-palato-cerebral
Fig. 2.
Kaplan–Meier 30-day disease-specific survival
Fig. 3.
Kaplan–Meier 30-day overall survival
A total of eight patients have cerebral infarction due to vascular invasion; three had infarct preoperatively and five postoperatively. Haemorrhagic infarct was seen in two patients, and ischemic infarct was seen in six patients. One patient postoperatively developed ascites, and an ascitic fluid tap suggests mucormycosis. Six patients had fungal meningitis due to dural invasion.
Discussion
Mucormycosis is usually limited to severely immunocompromised people, including those with diabetes mellitus. Risk factors for mucormycosis include immunosuppression and long-term treatment with corticosteroids. Because mucoromycetes grow best in acidic, high-glucose environments, diabetes, primarily if poorly controlled or complicated by ketoacidosis, is also a significant risk factor [5]. Its histopathological hallmark is the invasion of blood vessels, vasculotropism, leading to necrosis.
It is pertinent to note that the same treatment strategies were followed during the first COVID-19 wave last year, and there was no surge of cases during the first wave in contrast to the rising number of patients in a short period during the current second wave. It is also interesting that mucormycosis has not affected our neighboring countries in the subcontinent with the same clinical milieu and socioeconomic factors. Even in India, there is much regional variation in the presentation of the disease, and it substantially differs from the pre-COVID mucormycosis.
The actual case tally may be the tip of the iceberg, as the mucormycosis may not be detected in the fungal Koh swab every time [6]; adequate sampling from the appropriate region is necessary to get accurate results. More clinical suspicion must be maintained in all patients presenting with high-risk signs and symptoms. Certain institutes also consider this fungus as the etiological factor in patients with high-risk symptoms, signs, and predisposing factors.
Mucormycosis is a ubiquitous organism; the nasal cavity is the obvious point of entry. The disease typically begins in the nose and progresses to the paranasal sinuses, orbit, cribriform plate, meninges, and brain through direct extension and intravascular propagation. When dealing with highly immunocompromised individuals, the practitioner must have a high index of suspicion for the disorder. It is critical to recognize early warning signs and symptoms. The high-risk features are described in the workflow part.
Over 90% of our patients had an immediate history of COVID-19 pneumonia following the second wave of the pandemic. Around 88% of these individuals were treated with steroids to curtail the collateral immune-mediated pulmonary injury. Nearly 91% of the patients had diabetes, either previous long-standing or steroid-induced diabetes mellitus.
The most common symptoms of infection include nonspecific headaches, face pain, lethargy, dental discomfort, facial puffiness, decreased vision, and proptosis. Fever can suggest a more severe illness; however, the fever could be related to COVID-19 pneumonia during this pandemic. Loss of smell cannot be attributed to sino-nasal mucormycosis; it may be due to COVID-19. Ptosis, chemosis, proptosis, orbital pain, and vision loss indicate loss of function of the second, third, fourth, and sixth cranial nerves. There have been isolated case reports of facial nerve palsy [7], cavernous sinus thrombosis [8], and abducent nerve palsy [9] following mucormycosis. Nasal endoscopy is the first line of appropriate evaluation for high-risk patients, allowing for detecting changes in mucosal color (dusky blue to gray or black), mucosal texture (presence of slough, crusting, or ulceration), fungal debris, and mold growth. There may be diminished mucosal sensation or reduced bleeding during endoscopy due to early invasion and vasotropism; this should raise red flags for the clinician [10]. In a study by Gravesen, up to 80% of individuals had crusting in the nasal cavity [11].
The involvement of the palatine arteries can cause unilateral ulceration and perforation of the hard and soft palates as described by Gillespie [10]; along with this mode of presentation, our patients also had subtle, hard palate bulge to perforation of the hard palate with necrotic debris hanging out of it (Table 1).
Mucormycosis usually affects the nose and paranasal sinuses; the disease can spread via direct extension or by hematogenous spread. We have identified the following pathways of spread which correlates with the description provided by Hossieni and colleagues [12].
Into the Orbit
Mainly from the ethmoidal air cells, it infiltrates laterally through lamina papyracea into the medial aspect of the extraconal part of orbit (Fig. 1).
By erosion of frontal sinus floor into superior part of the orbit.
Infiltration of maxillary sinus roof to orbit and perineural spread along the infraorbital nerve.
The disease reaches the orbital apex via posterior ethmoids, sphenoid sinus erosion, and further spread from the extraconal part of the orbit.
Erosion of sphenoid sinus wall to the cavernous sinus and both sides optic nerves.
Into the Brain
Direct spread to the brain occurred through the roof of the ethmoid and cribriform plate to skull base to basi frontal lobe.
Disease tracking along the orbital apex into the cavernous sinus.
Through maxillary sinus via infratemporal fossa to skull base to the temporal lobe and additionally through hematogenous spread.
The mucormycosis may lead to disseminated fungemia and lodgement into other visceral organs due to hematogenous spread. Disseminated mucormycosis typically has a dismal prognosis [13].
Patients with mucormycosis that is restricted to the nasal cavity have a greater survival rate. The disease is more susceptible to complete endoscopic surgical resection if detected at this time [10]. Because antifungal medication alone will not suffice, all grossly diseased tissue must be excised. Debridement should be repeated until a healthy tissue margin is found, and it should be done at regular intervals, if necessary. Sinus surgery, wide resection of necrotic soft tissue and bone, and exenteration of the orbit, if necessary, are examples of more comprehensive surgical treatments [14, 15] (Supplement Table 1).
Whether to perform orbital exenteration is perhaps the most challenging in the management of orbital mucormycosis. Complete ophthalmoplegia, varying degrees of proptosis, cranial nerve involvement, and central retinal artery occlusion indicate orbital exenteration [16].
Negative margins and long-term survival are rarely achieved with aggressive surgical debridement to remove disease outside the sino-nasal cavity [10]. According to the available literature, patients with intracranial extension are less likely to react to radical surgery, and their prognosis is poor [17]. However, if surgery is done early on, the chances of survival increase significantly [10]. Patients should be appropriately counseled regarding the dismal prognosis before undergoing radical surgery. Refer to Supplement sheet 2 for our hypothesis on raise in mucormycosis.
It is well established that the management of comorbid conditions is crucial in the successful management of mucormycosis. It is also the single most crucial factor determining the overall prognosis of the patient [18]. Hyperglycaemia and acidosis are likely to have a role in diabetes mellitus patients’ susceptibility to rhinocerebral mucormycosis. The acidic environment is supposed to cause phagocytic impairment and decrease blood iron-binding capacity [18].
Liposomal amphotericin B is the first-line drug of choice for managing invasive mucormycosis, supported by level 1 evidence [19]. Conventional amphotericin B is considered an alternative to liposomal due to its high nephrotoxicity. Our patient’s hypokalemia was most commonly seen in 35% of individuals, followed by infusion reactions and asymptomatic rise in serum creatinine, similar to the Italian cohort [20]. A recent systematic review by Cornely and colleagues provides dose recommendations for various stages of mucormycosis [19]; in general, for uncomplicated invasive mucormycosis, the dose is 3 to 5 mg/kg/dose. The dosage for CNS involvement is a bit controversial; certain animal studies have shown better responses with up to 10 mg/kg/dose [21]. At this dose, there is a substantial risk of renal injury. As per the AMBILOAD trial [22], such higher preparation does not improve the overall outcome compared to the conventional dosing. Before amphotericin B dosage, factors such as existing diabetic and hypertension-induced nephropathy must be kept in mind. There is no strict consensus on the duration and the target dosage of amphotericin B therapy [23]. Based on our institutional expert multidisciplinary team recommendations, we have titrated the target dose based on the extent of the disease, comorbidities, and response to therapy.
Posaconazole alone is the second line of drug and can be used as salvage therapy following the amphotericin B therapy [19].
There is a severe shortage of liposomal amphotericin due to the emergence of mucormycosis during the second wave of the COVID-19 pandemic. In this unique and challenging situation, oral posaconazole loading dose (600 mg/day) followed by salvage dose (300 mg/day) was provided along with amphotericin B in uncomplicated cases to provide a higher dose of amphotericin B to complicated cases. The combination of polyenes with azoles are supported by limited in vitro [24] and clinical studies [25, 26].
Multiple case series have shown that prognosis in a patient with diabetes is superior when compared to immunodeficiencies and malignancies [15, 27]. This would explain the better prognosis among our patients (12.1% overall mortality) along with early debridement and medical management, and the long-term prognosis is yet to be established. In a recent systematic review, 80% mortality was seen with CNS involvement [28]; in our series, 45.4% mortality is seen in individuals with CNS involvement. We want to propose further stratification for CNS involvement which would accurately predict mortality. Among the patients who have survived the CNS, debridement has the minimal or early invasion of the dura and brain.
On the other hand, localized sino-nasal lesions have a lower fatality rate because of earlier identification and surgical debridement [19]. Early diagnosis and implementation of multidisciplinary treatment strategies for aggressive medical and surgical debridement are associated with improved survival [29].
Conclusion
The sino-nasal variety is the most common type of COVID-19-associated mucormycosis and has the best prognosis; the patients with intracranial involvement have a higher chance of mortality than the other groups. Well-controlled studies must confirm a potential therapeutic option of combination therapy with polyenes with azoles. Early identification, triaging, and proper multidisciplinary team management with systemic antifungals, aggressive surgical debridement, and control of comorbidities will lead to desirable outcomes.
Supplementary Information
Below is the link to the electronic supplementary material.
Author Contribution
All authors have contributed towards the development of the manuscript.
Study concept – RM, NN, AJ, RDA, SS, SC, KNR, NKA, AW.
Methodology – RM, NN, AJ, AS, RDA, KNR.
Data acquisition – RM, NN, KNR, RDA, SS, NSB, NKA, SS, SC, AS, AW, PS, PK, AB, PT Surgical therapy – NN, RM, RDA, SS, SC, KNR, AS, NSB, NKA.
Medical therapy—NN, AJ, RM, RDA, SS, SC, KNR, AS, NSB, NKA, PS, PKA, AB, PT.
Data analysis – RM, KNR, NN.
Manuscript writing – KNR, RM, AJ, NN.
Editing—NN, AJ, RM, RDA, SS, SC, KNR, AS, NSB, NKA, PS, PKA, AB, PT.
Manuscript review – all authors.
Declarations
Ethics
Institutional Research Cell approval—OW/RC/AIIMS-RPR/2021/571.
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Rupa Mehta, Email: rmehta0409@yahoo.com.
Nitin M. Nagarkar, Email: directoroffice@aiimsraipur.edu.in
Atul Jindal, Email: dratuljindal@gmail.com.
Karthik Nagaraja Rao, Email: Karthik.nag.rao@gmail.com.
S. B. Nidhin, Email: sbnidhin@gmail.com
Ripu Daman Arora, Email: neelripu@gmail.com.
Anil Sharma, Email: dr.anilsharma02@gmail.com.
Archana Wankhede, Email: archukeche@gmail.com.
Satish Satpute, Email: drsatish3s@gmail.com.
Sharmistha Chakravarty, Email: sharmisthachakravarty06@gmail.com.
N. K. Agrawal, Email: nkagrawal1963@gmail.com
Pranita, Email: dspranita@gmail.com.
Pankaj Kannauje, Email: drpankajkannauje@aiimsraipur.edu.in.
Ajoy Behera, Email: drajoybeherakims@gmail.com.
Pugazhenthan Thangaraju, Email: drpugal23@gmail.com.
References
- 1.Edara V-V, Lai L, Sahoo MK, Floyd K, Sibai M, Solis D, et al. Infection and vaccine-induced neutralizing antibody responses to the SARS-CoV-2 B.1.617.1 variant. bioRxiv [Internet]. 2021 May 10 [cited 2021 Jun 2]; Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8132229/
- 2.Prakash H, Chakrabarti A. Epidemiology of mucormycosis in India. Microorganisms. 2021;9(3):523. doi: 10.3390/microorganisms9030523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sharma NC. 28,252 cases of black fungus detected so far, says Centre [Internet]. mint. 2021 [cited 2021 Aug 30]. Available from: https://www.livemint.com/news/india/28-states-report-over-28-252-cases-of-mucormycosis-11623068251495.html
- 4.Frank GS, Smith JM, Davies BW, Mirsky DM, Hink EM, Durairaj VD. Ophthalmic manifestations and outcomes after cavernous sinus thrombosis in children. J Am Assoc Pediatr Ophthalmol Strabismus. 2015;19(4):358–362. doi: 10.1016/j.jaapos.2015.06.001. [DOI] [PubMed] [Google Scholar]
- 5.Hartnett KP, Jackson BR, Perkins KM, Glowicz J, Kerins JL, Black SR, et al. A guide to investigating suspected outbreaks of mucormycosis in healthcare. J Fungi. 2019;5(3):69. doi: 10.3390/jof5030069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lass-Flörl C (2017) Current challenges in the diagnosis of fungal infections. In: Lion T (eds) Human Fungal Pathogen Identification: Methods and Protocols [Internet]. Springer, New York, [cited 2021 Jun 6]. p 3–15. (Methods in Molecular Biology). Available from: 10.1007/978-1-4939-6515-1_1 [DOI] [PubMed]
- 7.Shekar V, Sikander J, Rangdhol V, Naidu M. Facial nerve paralysis: a case report of rare complication in uncontrolled diabetic patient with mucormycosis. J Nat Sci Biol Med. 2015;6(1):226–228. doi: 10.4103/0976-9668.149195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gelston CD. Rhino-orbital mucormycosis causing cavernous sinus and internal carotid thrombosis treated with posaconazole. Arch Ophthalmol. 2007;125(6):848. doi: 10.1001/archopht.125.6.848. [DOI] [PubMed] [Google Scholar]
- 9.Jain S, Kumar S, Kaushal A. Rhinocerebral mucormycosis with isolated sixth nerve palsy in an immunocompetent patient. Med J Malaysia. 2011;66(4):376–378. [PubMed] [Google Scholar]
- 10.Gillespie MB, O’Malley BW. An algorithmic approach to the diagnosis and management of invasive fungal rhinosinusitis in the immunocompromised patient. Otolaryngol Clin North Am. 2000;33(2):323–334. doi: 10.1016/S0030-6665(00)80008-0. [DOI] [PubMed] [Google Scholar]
- 11.Gravesen S. Fungi as a Cause of Allergic Disease. Allergy. 1979;34(3):135–154. doi: 10.1111/j.1398-9995.1979.tb01562.x. [DOI] [PubMed] [Google Scholar]
- 12.Hosseini SMS, Borghei P. Rhinocerebral mucormycosis: pathways of spread. Eur Arch Oto-Rhino-Laryngol. 2005;262(11):932–938. doi: 10.1007/s00405-005-0919-0. [DOI] [PubMed] [Google Scholar]
- 13.Walsh TJ, Gamaletsou MN, McGinnis MR, Hayden RT, Kontoyiannis DP. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis) Clin Infect Dis. 2012;54(suppl_1):S55–60. doi: 10.1093/cid/cir868. [DOI] [PubMed] [Google Scholar]
- 14.Pakdel F, Ahmadikia K, Salehi M, Tabari A, Jafari R, Mehrparvar G, Rezaie Y, Rajaeih S, Alijani N, Barac A, Abdollahi A, Khodavaisy S. Mucormycosis in patients with COVID-19: A cross-sectional descriptive multicentre study from Iran. Mycoses. 2021;64(10):1238–1252. doi: 10.1111/myc.13334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Scheckenbach K, Cornely O, Hoffmann TK, Engers R, Bier H, Chaker A, et al. Emerging therapeutic options in fulminant invasive rhinocerebral mucormycosis. Auris Nasus Larynx. 2010;37(3):322–328. doi: 10.1016/j.anl.2009.09.001. [DOI] [PubMed] [Google Scholar]
- 16.Hargrove RN, Wesley RE, Klippenstein KA, Fleming JC, Haik BG. Indications for orbital exenteration in mucormycosis. Ophthal Plast Reconstr Surg. 2006;22(4):286–291. doi: 10.1097/01.iop.0000225418.50441.ee. [DOI] [PubMed] [Google Scholar]
- 17.Anaissie EJ, Shikhani AH. Rhinocerebral mucormycosis with internal carotid occlusion: report of two cases and review of the literature. Laryngoscope. 1985;95(9 Pt 1):1107–1113. [PubMed] [Google Scholar]
- 18.Camara-Lemarroy CR, González-Moreno EI, Rodríguez-Gutiérrez R, Rendón-Ramírez EJ, Ayala-Cortés AS, Fraga-Hernández ML, et al. Clinical features and outcome of mucormycosis. Interdiscip Perspect Infect Dis. 2014;2014:e562610. doi: 10.1155/2014/562610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cornely OA, Alastruey-Izquierdo A, Arenz D, Chen SCA, Dannaoui E, Hochhegger B, et al. Global guideline for the diagnosis and management of mucormycosis: an initiative of the European Confederation of Medical Mycology in cooperation with the Mycoses Study Group Education and Research Consortium. Lancet Infect Dis. 2019;19(12):e405–e421. doi: 10.1016/S1473-3099(19)30312-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pagano L, Valentini CG, Posteraro B, Girmenia C, Ossi C, Pan A et al (2009) Zygomycosis in Italy: a survey of FIMUA-ECMM (Federazione Italiana di Micopatologia Umana ed Animale and European Confederation of Medical Mycology). J Chemother Florence Italy 21(3):322–329 [DOI] [PubMed]
- 21.Ibrahim AS, Gebremariam T, Husseiny MI, Stevens DA, Fu Y, Edwards JE, et al. Comparison of lipid amphotericin B preparations in treating murine zygomycosis. Antimicrob Agents Chemother. 2008;52(4):1573–1576. doi: 10.1128/AAC.01488-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cornely OA, Maertens J, Bresnik M, Ebrahimi R, Ullmann AJ, Bouza E, et al. Liposomal amphotericin B as initial therapy for invasive mold infection: a randomized trial comparing a high-loading dose regimen with standard dosing (AmBiLoad trial) Clin Infect Dis Off Publ Infect Dis Soc Am. 2007;44(10):1289–1297. doi: 10.1086/514341. [DOI] [PubMed] [Google Scholar]
- 23.Sipsas NV, Gamaletsou MN, Anastasopoulou A, Kontoyiannis DP. Therapy of mucormycosis. J Fungi. 2018;4(3):90. doi: 10.3390/jof4030090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ibrahim AS, Gebremariam T, Schwartz JA, Edwards JE, Spellberg B. Posaconazole mono- or combination therapy for treatment of murine zygomycosis. Antimicrob Agents Chemother. 2009;53(2):772–775. doi: 10.1128/AAC.01124-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Warkentien T, Rodriguez C, Lloyd B, Wells J, Weintrob A, Dunne JR, et al. Invasive mold infections following combat-related injuries. Clin Infect Dis Off Publ Infect Dis Soc Am. 2012;55(11):1441–1449. doi: 10.1093/cid/cis749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rodriguez CJ, Tribble DR, Malone DL, Murray CK, Jessie EM, Khan M, et al. Treatment of suspected invasive fungal infection in war wounds. Mil Med. 2018;183(suppl_2):142–6. doi: 10.1093/milmed/usy079. [DOI] [PubMed] [Google Scholar]
- 27.Rn H, Re W, Ka K, Jc F, Bg H. Indications for orbital exenteration in mucormycosis. Ophthal Plast Reconstr Surg. 2006;22(4):286–291. doi: 10.1097/01.iop.0000225418.50441.ee. [DOI] [PubMed] [Google Scholar]
- 28.Hoenigl M, Gangneux J-P, Segal E, Alanio A, Chakrabarti A, Chen SC-A, et al (2018) Global guidelines and initiatives from the European Confederation of Medical Mycology to improve patient care and research worldwide: new leadership is about working together. Mycoses 61(11):885–894 [DOI] [PubMed]
- 29.Guinea J, Escribano P, Vena A, Muñoz P, Martínez-Jiménez M del C, Padilla B et al (2017) Increasing incidence of mucormycosis in a large Spanish hospital from 2007 to 2015: epidemiology and microbiological characterization of the isolates. PLOS ONE 12(6):e0179136 [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.