Abstract
Kidney transplantation recipients (KTR) with coronavirus disease 2019 (COVID-19) are at higher risk of death than general population. However, mortality risk factors in KTR are still not clearly identified. Our objective was to systematically analyze published evidence for risk factors associated with mortality in COVID-19 KTR. Electronic databases were searched for eligible studies on 1 August 2021. All prospective and retrospective studies of COVID-19 in KTR were considered eligible without language restriction. Since data in case reports and series could potentially be subsets of larger studies, only studies with ≥ 50 patients were included. Random-effects model meta-analysis was used to calculate weighted mean difference (WMD) and pooled odds ratio (OR) of factors associated with mortality. From a total 1,137 articles retrieved, 13 were included in the systematic review and meta-analysis comprising 4,440 KTR. Compared with survivors, non-survivors were significantly older (WMD 10.5 years, 95% CI 9.3–11.8). KTR of deceased donor were at higher risk of death (OR 1.73, 95% CI 1.10–2.74). Comorbidities including diabetes mellitus, cardiovascular disease, and active cancer significantly increased mortality risk. KTR with dyspnea (OR 5.68, 95% CI 2.11–15.33) and pneumonia (OR 10.64, 95% CI 3.37–33.55) at presentation were at higher mortality risk, while diarrhea decreased the risk (OR 0.61, 95% CI 0.47–0.78). Acute kidney injury was associated with mortality (OR 3.24, 95% CI 1.36–7.70). Inflammatory markers were significantly higher in the non-survivors, including C-reactive protein, procalcitonin, and interleukine-6. A number of COVID-19 mortality risk factors were identified from KTR patient characteristics, presenting symptoms, and laboratory investigations. KTR with these risk factors should receive more intensive monitoring and early therapeutic interventions to optimize health outcomes.
Subject terms: Renal replacement therapy, Risk factors, SARS-CoV-2
Introduction
Coronavirus disease 2019 (COVID-19) is an ongoing global pandemic caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). Elderly patients and patients with multiple comorbidities are known to be at higher risk of death1, 2. Immunocompromised patients, particularly solid organ transplantation recipients and those with malignancies, are also at increased risk of severe COVID-19 disease and death3.
The mortality rate of COVID-19 In kidney transplantation patients was 20–40%4–7, compared with 10–15% mortality rate amongst admitted patients overall8–10. The immunosuppressed status in kidney transplantation recipients (KTR) might contribute to the higher mortality rate. Empirical stepwise reductions in immunosuppressive therapy have been recommended in patients at high risk of developing severe disease, and as clinical severity of COVID-19 symptoms increases11, 12. However, temporary reductions in immunosuppression might place these patients at risk for allograft rejection thereafter. More data from clinical studies are urgently needed regarding the management of COVID-19 in KTR, including patient selection criteria for immunosuppressive lowering strategies. Patients with higher mortality risk should be treated more aggressively compared to patients with a lower risk. Moreover, many transplantation programs have been halted during the COVID-19 pandemic. Apart from the strain on hospital facilities by general COVID-19 patients, concerns have been raised regarding donor-derived COVID-19 infection in recipients who might need relatively intensified immunosuppression in the perioperative period, although there are currently no reports of such cases13–15. Induction therapy might also increase the risk of acquiring COVID-19 in the early post-transplantation period16. A clinical tool that identifies patients who are more likely to have a good prognosis of COVID-19 with minimal clinical symptoms after transplantation, might help transplant programs to continue performing kidney transplantation in these low risk patients.
To date, many case reports, case series, and cohort studies of COVID-19 in KTR have been published. However, the clinical risk factors for mortality in KTR with COVID-19 infection are still unclear due to many scattered case reports, and inconsistent reporting with varying quality across larger studies. The objective of this systematic review and meta-analysis was to clarify risk factors for mortality in KTR with COVID-19 infection, to improve of quality of care during the ongoing COVID-19 pandemic.
Methods
Data sources and searches
This systematic review was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA)17. MEDLINE, Scopus, and Cochrane Central Register of Controlled Trials electronic databases were searched for eligible studies on 1 August 2021. The following search strategy was used for MEDLINE: ("Kidney Transplantation"[Mesh]) AND ("COVID-19"[Mesh] OR "SARS-CoV-2"[Mesh]), and the search terms in Scopus were (TITLE-ABS-KEY (COVID-19) AND TITLE-ABS-KEY (kidney AND transplantation)). The MeSH descriptors which exploded all trees of [Coronavirus] and [Kidney Transplantation] were applied in the Cochrane Central register of Controlled Trials. The reference lists in the qualified articles were also reviewed and studies were manually added if deemed appropriate.
Study selection
This systematic review and meta-analysis focused on risk factors contributing to mortality in COVID-19 KTR. Our inclusion criteria were studies of COVID-19 in KTR that included ≥ 50 patients, that reported numbers of survivors and non-survivors, and demographic and/or clinical characteristics by survival group. Baseline demographic characteristics, clinical presentation, laboratory investigations, and treatments related to transplantation and COVID-19 care were extracted separately for survivors and non-survivors. As single case reports and small case series could be subsets of larger clinical registries, we selected only studies with ≥ 50 KTR patients for our review. In addition, meta-analysis of studies that include studies with a small sample size are at risk for bias caused by sampling error and random variation18. Studies with ≥ 50 patients were excluded if they were subsets of other larger studies, based on the study site, start and end date, or if clearly mentioned in the larger studies. If a potentially duplicated population was presented in 2 large studies, the study reporting more information regarding survivors and non-survivors was selected as the main data source. Only studies with adequate information, in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement, were included in the review19. Two authors (S.U. and S.K.) independently screened the titles and abstracts of the electronic citations, and full-text articles were retrieved for comprehensive review. Disagreements were resolved through consensus and arbitration by a third author (N.T.).
Data extraction and quality assessment
The following information was extracted from each study: author names, publication date, journal title, study site, country of origin, study duration, total KTR included, and the number of COVID-19 survivors and non-survivors. Baseline patient characteristics, clinical presentations, laboratory investigations, and treatments were retrieved separately for each study, and grouped by survival group. The Newcastle–Ottawa scale was used for the quality assessment of each individual included study20, 21. The tool evaluates 3 domains which are selection, comparability, and outcome. Each domain is rated total scores of 4 in the selection domain, 2 in the comparability domain, and 3 in the outcome domain.
Data synthesis and analysis
Using data reported in each individual study, we used random-effects models to calculate pooled weighted mean differences (WMD) of continuous variables, and pooled odds ratio (OR) for binary variables, for non-survivors versus survivors. Mean and standard deviations (SD) were estimated by the method of Wan et al.22, if only the median and range, or interquartile range were provided in the study. Pooled OR were calculated using the logarithm of effect size and standard error from each study. Heterogeneity of pooled effect sizes was assessed using the I2 index and the Q-test p-value. An I2 index higher than 75% indicates medium to high heterogeneity. Even when heterogeneity was low or absent, we reported random effects over fixed effects models because clinical care may have differed by different sites, and care practices likely changed as physicians gained more experience in caring for COVID-19 patients. Regression-based Egger’s test was used to test for small-study effects. The mortality change rate over the study period was calculated by regressing the proportion of deaths against the study end dates, using a generalized linear model with a binomial family and logit link functions, and robust variance estimates, after weighting by study size23. The analyses were performed using Stata Statistical Software Release 16.1 (StataCorp LLC, College Station, TX).
Ethical considerations
This meta-analysis and systematic review did not directly obtain data from human or animal subject. All of the included studies’ information was published in the scientific journals without the possibility to identify the individual patients. The clinical and research activities being reported are consistent with the Principles of the Declaration of Istanbul as outlined in the 'Declaration of Istanbul on Organ Trafficking and Transplant Tourism'.
Results
Characteristics of the included studies
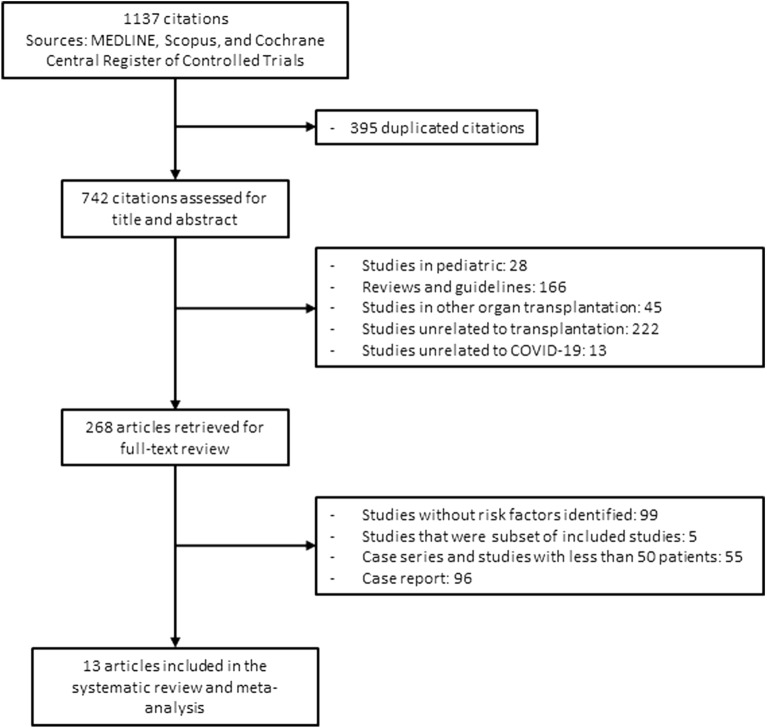
Figure 1 shows the flow diagram of study selection. A total 1,137 studies were retrieved using our search criteria. After duplicate citations and irrelevant studies were excluded, 268 articles underwent full-text review, and 13 articles were included in the final meta-analysis24–36. Details of each study are displayed in Table 1. Most studies were conducted during the first wave of the COVID-19 pandemic: the last study end date was December 2020, and study duration ranged from 1 to 9 months. Ten of 13 studies were multicenter, including 1 multi-continent international study and 1 multi-country European study. KTR numbers included in each study ranged from 52 to 1,680 patients, and mortality rates ranged from 12 to 32%. The total number of KTR reported in our meta-analysis was 4,440. The Newcastle–Ottawa Quality Assessment Scale of the included studies are shown in the Supplementary Table S1.
Figure 1.
Flow diagram of study selection.
Table 1.
Summary characteristics of included studies.
| References | Authors | Journal | First published | Study center | Country of cohort | Study duration | Total kidney transplantation patients included | Survivors | Non-survivors |
|---|---|---|---|---|---|---|---|---|---|
| 24 | Bossini et al | American Journal of Transplantation | July 2020 | Multicenter | Italy | 1/3/20 to 16/4/20 | 53 | 38 (72%) | 15 (28%) |
| 25 | Cravedi et al | American Journal of Transplantation | July 2020 | Multicenter | International | 2/3/20 to 15/5/20 | 144 | 98 (68%) | 46 (32%) |
| 26 | Caillard et al | Kidney International | August 2020 | Multicenter | France | 1/3/20 to 21/4/20 | 243 | 200 (82%) | 43 (18%) |
| 27 | Azzi et al | Kidney International | October 2020 | Albert Einstein College of Medicine, New York | USA | 16/3/20 to 29/7/20 | 229 | 182 (79%) | 47 (21%) |
| 28 | Craig-Schapiro et al | American Journal of Transplantation | October 2020 | Weill Cornell Medicine, New York | USA | 13/3/20 to 20/5/20 | 52 | 39 (75%) | 13 (25%) |
| 29 | Hilbrands et al | Nephrology Dialysis Transplantation | October 2020 | Multicenter | Europe | 1/2/20 to 1/6/20 | 305 | 240 (79%) | 65 (21%) |
| 30 | Mamode et al | Transplantation | November 2020 | Multicenter | UK | 1/3/20 to 27/4/20 | 121 | 85 (70%) | 36 (30%) |
| 31 | Willicombe et al | Transplantation | November 2020 | Imperial College, London | UK | 1/6/20 to 3/7/20 | 113 | 96 (85%) | 17 (15%) |
| 32 | Oto et al | BMC Nephrology | March 2021 | Multicenter | Turkey | 17/4/20 to 1/6/20 | 109 | 95 (87%) | 14 (13%) |
| 33 | Kute et al | Transplantation | April 2021 | Multicenter | India | 23/3/20 to 15/9/20 | 250 | 221 (88%) | 29 (12%) |
| 34 | Villanego et al | American Journal of Transplantation | April 2021 | Multicenter | Spain | 18/3/20 to 5/12/20 | 1,011 | 791 (78%) | 220 (22%) |
| 35 | Alshaqaq et al | Annals of Transplantation | June 2021 | Multicenter | Saudi Arabia | 1/3/20 to 31/8/20 | 130 | 116 (89%) | 14 (11%) |
| 36 | Requiao-Moura et al | PLoS ONE | July 2021 | Multicenter | Brazil | 1/3/20 to 11/11/20 | 1,680 | 90-day cumulative incidence of death 21% | |
Meta-analysis of patient characteristics associated with mortality of KTR with COVID-19 infection
Pooled estimates of patient characteristics associated with morality, with the numbers of studies and patients reporting data are displayed in Table 2. The median number of studies reporting these clinical characteristics was 6 (range 3–10), and median number of patients was 1,514 (range 496–2577). Non-survivors were significantly older than survivors, with WMD of 10.5 years (95% CI 9.3–11.8; p-value < 0.001; I2 0%; Q-test p-value = 0.46). KTR with co-existing medical comorbidities were at higher risk for death, including diabetes mellitus (OR 1.80; 95% CI 1.43–2.26; p-value < 0.001; I2 0%; Q-test p-value 0.58), cardiovascular diseases (OR 2.21; 95% CI 1.60–3.06; p-value < 0.001; I2 15.3%; Q-test p-value 0.35), and active cancer (OR 2.00; 95% CI 1.05–3.80; p-value = 0.034; I2 41.2%; Q-test p-value 0.11). Patients who received kidney allografts from deceased donors had 1.73-fold higher odds of mortality (95% CI 1.10–2.74; p-value = 0.019; I2 5%; Q-test p-value 0.40).
Table 2.
Meta-analysis of patient characteristics between survivor and non-survivor kidney transplantation patients.
| Variables | Survivors | Non-survivors | Number of patients reported | Number of studies reporting | Weighted mean difference (95% CI) | p-value from random effects model | I2 index (%) | Q-test p-value | Egger’s test p-value |
|---|---|---|---|---|---|---|---|---|---|
| Age, years (mean ± SD) | 54.9 ± 15.4 | 67.5 ± 11.8 | 2,577 | 10 | 10.5 (9.3, 11.8) | < 0.001 | 0 | 0.46 | 0.04 |
| BMI, kg/m2 (mean ± SD) | 27.5 ± 5.2 | 27.8 ± 6.0 | 586 | 3 | − 0.1 (− 0.3, 0.2) | 0.46 | 0 | 0.75 | 0.52 |
| Post kidney transplantation duration, months (mean ± SD) | 76.5 ± 78.0 | 79.4 ± 91.0 | 1,909 | 7 | 3.6 (− 4.9, 12.2) | 0.40 | 0 | 0.79 | 0.87 |
| Onset to admission, days (mean ± SD) | 8.0 ± 6.4 | 4.6 ± 3.4 | 496 | 3 | − 3.7 (− 8.2, 0.91) | 0.12 | 94.2 | 0.01 | < 0.001 |
| Variables | Survivors | Non-survivors | Number of patients reported | Number of studies reporting | Pooled odds ratio (95% CI) | p-value from random effects model | I2 index (%) | Q-test p-value | Egger’s test p-value |
|---|---|---|---|---|---|---|---|---|---|
| Male (%) | 1,345 (65.7) | 341 (64.3) | 2,577 | 10 | 0.94 (0.75, 1.19) | 0.63 | 10.6 | 0.37 | 0.38 |
| Hypertension (%) | 928 (76.3) | 243 (81.8) | 1,514 | 8 | 0.99 (0.68, 1.43) | 0.94 | 0 | 0.95 | 0.93 |
| Diabetes mellitus (%) | 548 (26.8) | 204 (38.5) | 2,577 | 10 | 1.80 (1.43, 2.26) | < 0.001 | 1.5 | 0.58 | 0.08 |
| Obesity (%) | 395 (46.8) | 106 (49.8) | 1,063 | 5 | 1.30 (0.92, 1.82) | 0.14 | 0 | 0.24 | 0.15 |
| Cardiovascular diseases (%) | 248 (21.2) | 106 (38.7) | 1,445 | 8 | 2.21 (1.60, 3.06) | < 0.001 | 15.3 | 0.35 | 0.58 |
| Pulmonary diseases (%) | 86 (10.1) | 31 (13.6) | 1,143 | 6 | 1.35 (0.86, 2.13) | 0.19 | 0 | 0.49 | 0.53 |
| Active cancer (%) | 75 (8.2) | 35 (15.1) | 1,143 | 6 | 2.00 (1.05, 3.80) | 0.034 | 41.2 | 0.11 | 0.88 |
| History of smoking (%) | 262 (30.7) | 70 (30.7) | 1,082 | 6 | 0.96 (0.69, 1.34) | 0.82 | 0 | 0.74 | 0.60 |
| Deceased donor kidney transplant (%) | 369 (47.5) | 130 (68.8) | 966 | 6 | 1.73 (1.10, 2.74) | 0.019 | 5.0 | 0.40 | 0.35 |
| Less than 12 months since kidney transplant (%) | 220 (11.4) | 64 (13.3) | 2,404 | 8 | 1.27 (0.83, 1.95) | 0.28 | 33.1 | 0.11 | 0.004 |
| Lymphocyte depleting induction (%) | 432 (68.1) | 87 (56.1) | 789 | 6 | 0.73 (0.49, 1.08) | 0.12 | 0 | 0.90 | 0.44 |
| Tacrolimus (%) | 1,108 (83.9) | 288 (79.6) | 1,682 | 5 | 0.74 (0.50, 1.10) | 0.13 | 15.8 | 0.47 | 0.72 |
| mTORi (%) | 284 (16.4) | 55 (12.1) | 2,183 | 7 | 0.89 (0.61, 1.30) | 0.55 | 11.5 | 0.020 | 0.09 |
| MPA (%) | 1,305 (75.0) | 320 (73.7) | 2,175 | 7 | 0.98 (0.76, 1.25) | 0.86 | 0 | 0.79 | 0.99 |
| Prednisolone (%) | 1,389 (78.0) | 355 (79.4) | 2,227 | 8 | 1.13 (0.86, 1.48) | 0.38 | 0 | 0.37 | 0.84 |
| Fever (%) | 1,217 (74.0) | 324 (77.7) | 2,062 | 6 | 1.19 (0.83, 1.69) | 0.34 | 32.1 | 0.27 | 0.23 |
| Cough (%) | 1,011 (65.4) | 274 (73.9) | 1,918 | 5 | 1.21 (0.73, 2.01) | 0.46 | 65.3 | 0.017 | 0.07 |
| Dyspnea (%) | 298 (34.9) | 146 (74.1) | 1,051 | 5 | 5.68 (2.11, 15.33) | < 0.001 | 84.6 | < 0.001 | 0.19 |
| Diarrhea (%) | 540 (32.8) | 100 (24.0) | 2,062 | 6 | 0.61 (0.47, 0.78) | < 0.001 | 0 | 0.75 | 0.31 |
| Pneumonia (%) | 582 (55.4) | 240 (93.8) | 1,306 | 3 | 10.64 (3.37, 33.56) | < 0.001 | 25.6 | 0.18 | 0.87 |
| Acute kidney injury (%) | 203 (40.2) | 82 (63.1) | 634 | 5 | 3.24 (1.36, 7.70) | 0.008 | 63.2 | 0.030 | 0.002 |
BMI body mass index, CNI calcineurin inhibitor, MPA mycophenolic acid, mTORi mammalian target of rapamycin inhibitor, SpO2 oxygen saturation.
For presenting COVID-19 symptoms, patients with dyspnea and pneumonia were at 5.68-fold (95% CI 2.11–15.33; p-value < 0.001; I2 84.6%; Q-test p-value < 0.001) and 10.64-fold (95% CI 3.37–33.56; p-value < 0.001; I2 25.6%; Q-test p-value 0.18) higher risk of death, respectively. Acute kidney injury also significantly increased mortality risk (OR 3.24; 95% CI 1.36–7.70; p-value = 0.008; I2 63.2%; Q-test p-value 0.030). However, diarrhea significantly lowered the risk with OR of 0.61 (95% CI 0.47–0.78; p-value < 0.001; I2 0%; Q-test p-value 0.75). Supplementary Figure S1 shows the forest plot of patient characteristics that were significantly associated with mortality.
Meta-analysis of baseline laboratory investigations associated with mortality of KTR with COVID-19 infection
Table 3 shows the baseline laboratory results of survivors and non-survivors. Non-survivors had significantly lower estimate glomerular filtration rate (eGFR) compared with the survivors (WMD − 11.4 mL/min/1.73 m2; 95% CI − 15.7, − 7.0; p-value < 0.001; I2 0%; Q-test p-value = 0.55). Other standard laboratory investigations did not show the association with COVID-19 mortality.
Table 3.
Meta-analysis of baseline laboratory investigations between survivor and non-survivor kidney transplantation recipient.
| Variables | Survivors | Non-survivors | Number of patients reported | Number of studies reporting | Weighted mean difference (95% CI) | p-value from random effects model | I2 index (%) | Q-test p-value | Egger’s test p-value |
|---|---|---|---|---|---|---|---|---|---|
| Initial serum Cr, mg/dL (mean ± SD) | 1.79 ± 0.88 | 2.19 ± 1.28 | 825 | 5 | 0.50 (− 0.02, 1.03) | 0.06 | 90.9 | < 0.001 | 0.85 |
| Initial eGFR, mL/min/1.73 m2 (mean ± SD) | 44.4 ± 23.3 | 34.4 ± 21.0 | 562 | 3 | − 11.4 (− 15.7, − 7.0) | < 0.001 | 0 | 0.55 | 0.72 |
| Hemoglobin, g/dL (mean ± SD) | 11.6 ± 2.1 | 11.4 ± 2.2 | 582 | 4 | − 0.3 (− 0.9, 0.2) | 0.22 | 34.0 | 0.20 | 0.09 |
| Platelet, × 103/μL (mean ± SD) | 199 ± 66 | 172 ± 115 | 825 | 5 | − 18.5 (− 39.8, 2.8) | 0.09 | 60.1 | 0.030 | 0.18 |
| WBC, × 103/μL (mean ± SD) | 6.42 ± 2.76 | 7.90 ± 3.80 | 525 | 4 | 1.61 (− 0.14, 3.36) | 0.07 | 88.0 | < 0.001 | 0.67 |
| Lymphocytes, × 103/μL (mean ± SD) | 1.04 ± 1.03 | 0.77 ± 0.50 | 1,182 | 7 | − 0.06 (− 0.15, 0.02) | 0.16 | 34.5 | 0.02 | 0.004 |
| Lactate dehydrogenase, U/L (mean ± SD) | 304.0 ± 85.3 | 425.1 ± 205.3 | 473 | 3 | 117.4 (− 13.2, 248.0) | 0.078 | 96.1 | < 0.001 | 0.94 |
| C-reactive protein, mg/dL (mean ± SD) | 5.66 ± 8.23 | 10.50 ± 9.42 | 830 | 5 | 4.85 (1.18, 8.52) | 0.010 | 88.9 | < 0.001 | 0.18 |
| D-dimer, μg/mL (mean ± SD) | 1.29 ± 1.36 | 1.67 ± 1.53 | 525 | 4 | 0.37 (− 0.22, 0.96) | 0.22 | 88.6 | 0.002 | 0.87 |
| Procalcitonin, ng/mL (mean ± SD) | 0.20 ± 0.57 | 0.97 ± 1.48 | 525 | 4 | 0.60 (0.36, 0.83) | < 0.001 | 64.5 | 0.031 | 0.77 |
| Ferritin, ng/mL (mean ± SD) | 893 ± 1294 | 1232 ± 1041 | 634 | 5 | 128.5 (− 276.1, 533.1) | 0.53 | 71.5 | 0.019 | 0.001 |
| IL-6, pg/mL (mean ± SD) | 31.3 ± 35.1 | 126.0 ± 178.7 | 473 | 3 | 95.4 (54.0, 136.8) | < 0.001 | 76.1 | 0.006 | 0.86 |
Cr creatinine, eGFR estimated glomerular filtration rate, WBC white blood cell.
Details of biomarkers reflecting tissue damage and inflammation at baseline were reported in 3–5 studies representing 473–830 patients in Table 3. Three biomarkers were significantly higher in non-survivors compared with the survived KTR including C-reactive protein (WMD 4.85 mg/dL; 95% CI 1.18–8.52; p-value = 0.010; I2 88.9%; Q-test p-value < 0.001), procalcitonin (WMD 0.60 ng/mL; 95% CI 0.36–0.83; p-value < 0.001; I2 64.5%; Q-test p-value = 0.031), and IL-6 (WMD 95.4 pg/mL; 95% CI 54.0–136.8; p-value < 0.001; I2 76.1%; Q-test p-value = 0.006). Supplementary Figure S2 displays the forest plot of laboratory variables showing a significant association with mortality.
Meta-analysis of treatment received associated with mortality of KTR with COVID-19 infection
Table 4 shows details of the treatment received in the survivors and non-survivors. A significantly higher proportion of KTR non-survivors required ventilator support or intubation (OR 56.45; 95% CI 9.67–329.62; p-value < 0.001; I2 93.1%; Q-test p-value < 0.001). Significantly higher proportions of non-survivors than survivors were treated with hydroxychloroquine, steroids, antibiotics, tocilizumab, and convalescent plasma. Antiviral drugs including lopinavir, remdesivir, darunavir, and favipiravir were given more frequently to non-survivors (OR 1.99; 95% CI 1.36–2.93; p-value < 0.001; I2 15.8%; Q-test p-value = 0.17). The forest plot of each significantly associated variable is illustrated in Supplementary Figure S3.
Table 4.
Meta-analysis of treatment received between survivor and non-survivor kidney transplantation patients.
| Variables | Survivors | Non-survivors | Number of patients reported | Number of studies reporting | Pooled odds ratio (95% CI) | p-value from random effects model | I2 index (%) | Q-test p-value | Egger’s test p-value |
|---|---|---|---|---|---|---|---|---|---|
| Ventilator support or intubation (%) | 68 (5.3) | 224 (64.0) | 1,645 | 6 | 56.45 (9.67, 329.62) | < 0.001 | 93.1 | < 0.001 | 0.007 |
| Withhold CNIs (%) | 32 (6.9) | 46 (39.3) | 582 | 4 | 10.07 (0.76, 132.62) | 0.08 | 93.1 | < 0.001 | < 0.001 |
| Withhold antimetabolites (%) | 379 (75.2) | 110 (84.6) | 634 | 5 | 1.66 (0.92, 2.99) | 0.09 | 0 | 0.39 | 0.46 |
| Hydroxychloroquine (%) | 716 (55.3) | 226 (64.6) | 1,645 | 6 | 1.55 (1.20, 2.00) | < 0.001 | 0 | 0.38 | 0.29 |
| Steroid (%) | 550 (42.5) | 240 (68.6) | 1,645 | 6 | 4.40 (1.70, 11.38) | 0.002 | 81.6 | 0.002 | 0.014 |
| Antibiotics (%) | 553 (44.0) | 169 (50.1) | 1,593 | 5 | 1.91 (1.06, 3.46) | 0.031 | 46.3 | 0.10 | 0.013 |
| Antivirals (%) | 273 (21.1) | 107 (30.6) | 1,645 | 6 | 1.99 (1.36, 2.93) | < 0.001 | 15.8 | 0.17 | 0.15 |
| Tocilizumab (%) | 120 (9.3) | 90 (25.7) | 1,645 | 6 | 5.40 (1.54, 18.88) | 0.008 | 88.1 | < 0.001 | 0.88 |
| Convalescent plasma (%) | 8 (2.6) | 15 (21.4) | 381 | 3 | 8.76 (1.85, 41.5) | 0.006 | 56.2 | 0.10 | 0.86 |
CNI; calcineurin inhibitor.
Multivariable models for mortality in each study
Ten of 13 studies presented adjusted (multivariable) models for factors associated with mortality (Table 5)24–26, 29, 31–36. Statistical methods to analyze multivariable model were different between studies including logistic regression and Cox proportional hazard regression. Thus, the combined or pooled effect sizes of each variable was not executed. The most common variables significantly contributing to mortality after adjustment were age in 8 studies, followed by dyspnea or respiratory rate (5 studies) and renal function (5 studies), and the presence of cardiovascular disease in 3 studies.
Table 5.
Multivariable model for mortality in each study.
| Authors [reference] | Study center | Country of cohort | Significant variables from multivariable model for mortality | Adjusted odds or hazard ratio (95% CI) | p-value | Model selection method |
|---|---|---|---|---|---|---|
| Bossini et al24 | Multicenter | Italy | Age > 60 vs < 60 | 1.12 (1.03–1.24) | 0.01 | Stepwise selection after including all statistically significant variables from univariate logistic regression |
| Shortness of breath | 13.7 (2.7–68.9) | 0.004 | ||||
| Cravedi et al25 | Multicenter | International | Age | 1.07 (1.02–1.14) | 0.022 | Akaike information criterion and Nagelkerke pseudo R2 after logistic regression |
| Respiratory rate ≥ 20 vs < 20 | 6.88 (1.63–41.98) | 0.017 | ||||
| IL-6 | 1 (1–1.01) | 0.04 | ||||
| eGFR | 0.96 (0.93–0.99) | 0.029 | ||||
| Caillard et al26 | Multicenter | France | Age > 60 vs < 60 | 3.81 (1.56–9.31) | 0.003 | Backward selection after including all statistically significant variables from univariate Cox regression |
| Cardiovascular disease | 2.04 (1.07–3.90) | 0.031 | ||||
| Dyspnea on admission | 2.35 (1.23–4.49) | 0.010 | ||||
| Hilbrands et al29 | Multicenter | Europe | Age | 1.07 (1.04–1.10) | < 0.001 | Backward selection after included all statistically significant variables from univariate Cox regression |
| Respiratory rate | 1.07 (1.03–1.11) | < 0.001 | ||||
| > 25% increased creatinine | 1.89 (1.05–3.40) | 0.03 | ||||
| Prednisolone use | 2.88 (1.03–8.03) | 0.04 | ||||
| Willicombe et al31 | Imperial College, London | UK | Age | 1.07 (1.00–1.13) | 0.041 | Backward selection after included all statistically significant variables from univariate logistic regression |
| No diabetes mellitus | 0.27 (0.07–0.99) | 0.047 | ||||
| Living donor transplantation | 0.08 (0.01–0.72) | 0.024 | ||||
| Prednisolone use | 5.98 (1.65–21.60) | 0.006 | ||||
| Oto et al32 | Multicenter | Turkey | Presence of ischemic heart disease | 4.129 (1.104–15.442) | 0.035 | Variables with p-value < 0.05 from univariate logistic regression were adjusted in the multivariable model |
| Creatinine at presentation | 1.681 (1.083–2.608) | 0.021 | ||||
| Kute et al33 | Multicenter | India | Baseline creatinine before COVID-19 | 5.424 (1.294–2.273E7) | 0.043 | Not reported (Cox regression) |
| Villanego et al34 | Multicenter | Spain | Age | 1.06 (1.05–1.08) | < 0.001 | Variables with p-value < 0.1 from univariate analysis were included in the multivariable Cox model |
| Time from transplantation ≤ 6 months | 1.64 (1.07–2.50) | 0.021 | ||||
| Gastrointestinal symptoms | 0.66 (0.48–0.90) | 0.011 | ||||
| Pneumonia | 5.04 (2.81–9.05) | < 0.001 | ||||
| Alshaqaq et al35 | Multicenter | Saudi Arabia | Age | 1.06 (1.013–1.109) | 0.012 | Statistically significant variables and clinically important variables were included in a multivariate Cox regression model |
| Creatinine at presentation | 1.002 (1.00–1.004) | 0.016 | ||||
| Use of azathioprine | 6.38 (1.374–29.630) | 0.018 | ||||
| Acute kidney injury | 18.11 (2.244–146.21) | 0.007 | ||||
| Requiao-Moura et al36 | Multicenter | Brazil | Age | 1.054 (1.040–1.067) | < 0.001 | Variables with p-value ≤ 0.1 from univariate analysis were included in the multivariable logistic regression model |
| Time after transplantation (years) | 1.025 (1.002–1.047) | 0.030 | ||||
| Hypertension | 1.566 (1.070–2.293) | 0.021 | ||||
| Cardiovascular disease | 1.517 (1.047–2.198) | 0.028 | ||||
| CNI-MPA combination | 1.197 (1.022–1.401) | 0.026 | ||||
| Recent high dose of steroids | 1.534 (1.063–2.214) | 0.022 | ||||
| Days of symptom before presentation | 0.954 (0.928–0.981) | 0.001 | ||||
| Dyspnea | 3.437 (2.584–4.571) | < 0.001 | ||||
| Headache | 0.552 (0.371–0.821) | 0.003 | ||||
| Anosmia | 0.563 (0.387–0.821) | 0.003 |
ARDS acute respiratory distress syndrome, CNI calcineurin inhibitor, eGFR estimated glomerular filtration rate, IL-6 interleukine-6, LDH lactate dehydrogenase, MPA mycophenolate.
Mortality trend from the studies of COVID-19 in kidney transplantation recipients
The mortality percentages from all 13 studies included in this meta-analysis were plotted against the study end date, with the back-transformed predicted slope from the generalized linear model in Fig. 2. A gradual decreasing trend in mortality was noted in the prediction plot. The change in the predicted mortality percentage over the all-study period was -5.1% (regression coefficient per hundred days = − 2.2 (95% CI − 2.4, − 2.0; P < 0.001).
Figure 2.
Bubble plot of actual mortality percentage and study end date, with the predicted regression line from a generalized linear model. Bubble size is proportional to the total number of participants in the study.
Discussions
This systematic review and meta-analysis are the first to describe and quantitate the degree of risk for factors associated with mortality in COVID-19 KTR from large cohorts and clinical registries. The results show that a number of baseline patient characteristics increased the risk of patient death, including increasing age, medical comorbidities, and being recipients of deceased donor kidneys. The latter likely relates to inferior allograft function relative to the recipients of living donors37–41, which predisposes patients to an increased risk for severe COVID-19. Non-survivors presented with more dyspnea, pneumonia, and acute kidney injury.
Interestingly, gastrointestinal symptoms significantly more frequent in the survivors. This finding is concordant across studies from France, Spain, the European registry, India, and the international cohort25, 26, 29, 33, 34. The I2 index of the gastrointestinal symptoms was 0% indicating very low heterogeneity. Diarrhea in COVID-19 patients is proposed to result from direct infection of COVID-19 in the intestinal epithelium cells, via angiotensin-converting enzyme 2 (ACE2) receptors that further increase local proinflammatory cytokines and changes in the intestinal flora42, 43. However, association with lower mortality in patients with diarrhea needs further study to assess whether this association is due to more rapid viral clearance through the gastrointestinal tract, or the stimulation of specific immune cells in the intestinal immune-network.
Obesity is an established risk factor for COVID-19 mortality in the general population44, 45. Surprisingly, obesity was not associated with mortality in our meta-analysis of KTR, and the studies reporting on this risk factor had low heterogeneity demonstrated by the I2 index of 0% in Table 2) 25, 26, 29, 30, 33. In addition, no multivariable models in Table 5 included obesity as a significant predictor for mortality. Several mechanisms have been proposed to link obesity to severe COVID-19 illness or death, including lower cardiorespiratory-metabolic reserve and dysfunctional immune response via excess adiposity46. It is possible that in KTR, immunosuppression or previous exposure to uremic status, might override or interfere with the effect of ectopic fat deposition that also leads to higher mortality of COVID-19 in KTR compared with the general population47, 48. However, further studies are needed to evaluate this hypothesis.
To date, many large randomized controlled trials (RCT) have failed to demonstrate a benefit of treatment interventions in lowering COVID-19 mortality49–55. Although there is no evidence that interventions decrease mortality rates, some evidence suggests that treatments including remdesivir, convalescent plasma therapy, and tocilizumab can attenuate the clinical course in COVID-19 patients56–60. Dexamethasone is the only medication proven to lower 28-day mortality in COVID-19 patients who received respiratory support61. The results from this meta-analysis supports that the non-survivors, compared with survivors, exhibit higher inflammatory states, demonstrated from the significantly higher levels of C-reactive protein, pro-calcitonin, and IL-6. This inflammation cannot be explained from the virus itself, and is thought to result from dysregulation of the host immune response leading to a “cytokine storm” and multiple organ dysfunction62–64. The clinical and laboratory results from this study could help identify the COVID-19 KTR with ongoing inflammation who are at risk for multiple organ dysfunction and death, and these patients might be candidates for anti-inflammatory immunomodulator agents. For example, case reports and case series have demonstrated the benefit of tocilizumab as a treatment of COVID-19 KTR65–68. More favorable treatment responses might be achieved if IL-6 levels and/or other biomarkers are used as an inclusion criterion for receiving tocilizumab in future RCTs.
It is important not to misinterpret the associations between treatment effects and mortality found in this meta-analysis. None of the included studies were RCTs specifically designed to evaluate treatment outcomes, the pooled univariable OR presented in the results section do not represent causal relationships, since they are confounded by disease severity, with patients experiencing more severe symptoms more likely to receive aggressive treatment interventions. The bubble plot in Fig. 2 reveals possible improvement of the care of COVID-19 KTR, as has been demonstrated in the non-organ transplant COVID-19 patients8, 69.
The information from this systematic review and meta-analysis could be used in many ways. Clinician could identify patient characteristics suggesting a poor prognosis, and begin early aggressive monitoring and treatment. According to our results, KTR at higher risk of death included the elderly (mean age of the non-survivors was 67.5 years compared with 54.9 years in the survivor group), patients with diabetes mellitus or cardiovascular diseases, patients with active malignancy, history of deceased donor kidney transplantation, dyspneic patients, presence of pneumonia, and patients with acute kidney injury on initial presentation, or with low eGFR (mean eGFR of the non-survivors was 34.4 mL/min/1.73 m2 compared with 44.4 mL/min/1.73 m2 in the survivor group). In addition, elevated concentrations of C-reactive protein, procalcitonin, and IL-6 at presentation should flag the patient as having an increased risk of death. In-hospital treatment with more aggressive and earlier immunosuppression reduction strategies would be reasonable in these high-risk patients, who also might be appropriate targets for the future studies of novel therapeutic interventions. Patients without any of these high-risk features could be closely monitored without significant changes in their immunosuppression. This strategy should be tested in future cohorts or clinical trials. Given the slow role out of vaccinations in many countries, patients at high risk of mortality should be prioritized to receive vaccinations. In addition, transplant centers could resume kidney transplantation programs in potential recipients who are at low COVID-19 mortality risk, based on an assessment of risks in baseline patient characteristics, accompanied by proper decision-making systems70, 71.
There are some limitations in our study. First, the possibility of duplicated information in individual patient level data could not be completely eliminated across our included studies, particularly international study25. However, we carefully selected only large studies with ≥ 50 patients, and thoroughly reviewed all article characteristics to remove studies that were subsets of others, thereby minimizing the chance of data duplication. Using this method, 5 studies were excluded as shown in Fig. 1. No included studies contained identical data and had distinctive information that was valuable for the meta-analysis. Second, SARS-CoV-2 viral load was not routinely measured, so correlations between viral load and KTR outcomes are lacking. Recent studies have demonstrated that the degree of SARS-CoV-2 viral load elevation correlated with disease severity and mortality72, therefore the viral load is likely to be another important factor predicting mortality risk in COVID-19 KTR. Third, the included studies were mainly from the European countries and the USA, with only limited data from Asian countries. Fourth, Egger’s test is not reliable for assessing publication bias when small study numbers are involved. However, given the interest of the medical community in reporting studies about COVID-19, we think that the risk of publication bias is mitigated. Lastly, the complications of COVID-19 contributing to patient death, such as superimposed bacterial infection and thromboembolic phenomenon, were not adequately reported in the included studies73, 74. More robust reporting of these complications would raise the awareness amongst clinicians regarding these potentially fatal complications.
In conclusion, mortality risk of COVID-19 KTR was increased in older patients, with medical comorbidities and deceased donor kidney recipients. Those with acute kidney injury, dyspnea, pneumonia, and increased of inflammatory biomarkers also had an increased risk of dying. Gastrointestinal tract symptoms were associated with lower risk of death. These risk factors could be used for developing clinical scores to further improve the quality of care in COVID-19 kidney transplant patients.
Supplementary Information
Acknowledgements
The authors thank the staff of the medical library of the Faculty of Medicine, Chulalongkorn University for providing the original and full-text articles used in this systematic review and meta-analysis.
Author contribution
S.U., MD: First author, corresponding author, study conceptualization, data search and review, statistical analysis, manuscript writing. S.J.K., PhD: Data search and review, statistical analysis, manuscript review and edit. N.T., MD: Manuscript review and edit. P.S., MD, PhD: Manuscript review and edit. W.T., MD: Manuscript review and edit. K.P., MD: Manuscript review and edit. S.E.-Ong, MD: Manuscript review and edits. Y.A., MD: Manuscript review and edit.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
The online version contains supplementary material available at 10.1038/s41598-021-99713-y.
References
- 1.Grasselli G, Greco M, Zanella A, et al. Risk factors associated with mortality among patients with COVID-19 in intensive care units in Lombardy, Italy. JAMA Intern. Med. 2020;180(10):1345–1355. doi: 10.1001/jamainternmed.2020.3539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet. 2020;395(10229):1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fung M, Babik JM. COVID-19 in immunocompromised hosts: what we know so far. Clin. Infect. Dis. 2020;2:2. doi: 10.1093/cid/ciaa863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mahalingasivam V, Craik A, Tomlinson LA, et al. A systematic review of COVID-19 and kidney transplantation. Kidney Int. Rep. 2020;2:2. doi: 10.1016/j.ekir.2020.10.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Moosavi SA, Mashhadiagha A, Motazedian N, Hashemazar A, Hoveidaei AH, Bolignano D. COVID-19 clinical manifestations and treatment strategies among solid-organ recipients: A systematic review of cases. Transplant Infect. Dis. 2020;22:6. doi: 10.1111/tid.13427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Nacif LS, Zanini LY, Waisberg DR, et al. COVID-19 in solid organ transplantation patients: A systematic review. Clinics. 2020;75:1–11. doi: 10.6061/clinics/2020/e1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Oltean M, Søfteland JM, Bagge J, et al. Covid-19 in kidney transplant recipients: a systematic review of the case series available three months into the pandemic. Infect. Dis. (Lond). 2020;52(11):830–837. doi: 10.1080/23744235.2020.1792977. [DOI] [PubMed] [Google Scholar]
- 8.Asch DA, Sheils NE, Islam MN, et al. Variation in US hospital mortality rates for patients admitted with COVID-19 during the first 6 months of the pandemic. JAMA Intern. Med. 2020;2:2. doi: 10.1001/jamainternmed.2020.8193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Piroth L, Cottenet J, Mariet AS, et al. Comparison of the characteristics, morbidity, and mortality of COVID-19 and seasonal influenza: a nationwide, population-based retrospective cohort study. Lancet Respir. Med. 2020;2:2. doi: 10.1016/S2213-2600(20)30527-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Aziz F, Mandelbrot D, Singh T, et al. Early report on published outcomes in kidney transplant recipients compared to nontransplant patients infected with coronavirus disease 2019. Transplant Proc. 2020;52(9):2659–2662. doi: 10.1016/j.transproceed.2020.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Abu Jawdeh BG. COVID-19 in kidney transplantation: Outcomes, immunosuppression management, and operational challenges. Adv. Chron. Kidney Dis. 2020;27(5):383–389. doi: 10.1053/j.ackd.2020.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Maggiore U, Abramowicz D, Crespo M, et al. How should I manage immunosuppression in a kidney transplant patient with COVID-19? An ERA-EDTA DESCARTES expert opinion. Nephrol. Dial Transplant. 2020;35(6):899–904. doi: 10.1093/ndt/gfaa130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sharma V, Shaw A, Lowe M, Summers A, van Dellen D, Augustine T. The impact of the COVID-19 pandemic on renal transplantation in the UK. Clin. Med. (Lond). 2020;20(4):e82–e86. doi: 10.7861/clinmed.2020-0183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Trubin PA, Azar MM, Malinis M. Diagnostic Testing of COVID-19 in Solid Organ Transplantation: Current Clinical Application and Future Strategies. Curr. Transplant. Rep. 2020;2:1–9. doi: 10.1007/s40472-020-00307-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Alasfar S, Avery RK. The impact of COVID-19 on kidney transplantation. Nat. Rev. Nephrol. 2020;16(10):568–569. doi: 10.1038/s41581-020-00340-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Abuzeineh M, Desai N, Brennan DC, Alasfar S. COVID-19 early after a deceased donor kidney transplant surgery. Transplantation. 2020;104(12):e354–e355. doi: 10.1097/TP.0000000000003439. [DOI] [PubMed] [Google Scholar]
- 17.Liberati A, Altman DG, Tetzlaff J, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: explanation and elaboration. PLoS Med. 2009;6(7):e1000100. doi: 10.1371/journal.pmed.1000100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lin L. Bias caused by sampling error in meta-analysis with small sample sizes. PLoS ONE. 2018;13(9):e0204056. doi: 10.1371/journal.pone.0204056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.von Elm E, Altman DG, Egger M, Pocock SJ, Gøtzsche PC, Vandenbroucke JP. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008;61(4):344–349. doi: 10.1016/j.jclinepi.2007.11.008. [DOI] [PubMed] [Google Scholar]
- 20.Stang A. Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol. 2010;25(9):603–605. doi: 10.1007/s10654-010-9491-z. [DOI] [PubMed] [Google Scholar]
- 21.Zeng X, Zhang Y, Kwong JS, et al. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: A systematic review. J. Evid. Based Med. 2015;8(1):2–10. doi: 10.1111/jebm.12141. [DOI] [PubMed] [Google Scholar]
- 22.Wan X, Wang W, Liu J, Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med. Res. Methodol. 2014;14:135. doi: 10.1186/1471-2288-14-135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Papke LE, Wooldridge JM. Econometric methods for fractional response variables with an application to 401(k) plan participation rates. J. Appl. Economet. 1996;11(6):619–632. doi: 10.1002/(SICI)1099-1255(199611)11:6<619::AID-JAE418>3.0.CO;2-1. [DOI] [Google Scholar]
- 24.Bossini N, Alberici F, Delbarba E, et al. Kidney transplant patients with SARS-CoV-2 infection: The Brescia Renal COVID task force experience. Am. J. Transplant. 2020;20(11):3019–3029. doi: 10.1111/ajt.16176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Cravedi P, Mothi SS, Azzi Y, et al. COVID-19 and kidney transplantation: Results from the TANGO international transplant consortium. Am. J. Transplant. 2020;20(11):3140–3148. doi: 10.1111/ajt.16185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Caillard S, Anglicheau D, Matignon M, et al. An initial report from the French SOT COVID registry suggests high mortality due to COVID-19 in recipients of kidney transplants. Kidney Int. 2020;98(6):1549–1558. doi: 10.1016/j.kint.2020.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Azzi Y, Parides M, Alani O, et al. COVID-19 infection in kidney transplant recipients at the epicenter of pandemics. Kidney Int. 2020;98(6):1559–1567. doi: 10.1016/j.kint.2020.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Craig-Schapiro R, Salinas T, Lubetzky M, et al. COVID-19 outcomes in patients waitlisted for kidney transplantation and kidney transplant recipients. Am. J. Transpl.. 2020;2:2. doi: 10.1111/ajt.16351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hilbrands LB, Duivenvoorden R, Vart P, et al. COVID-19-related mortality in kidney transplant and dialysis patients: Results of the ERACODA collaboration. Nephrol. Dial. Transplant. 2020;35(11):1973–1983. doi: 10.1093/ndt/gfaa261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Mamode N, Ahmed Z, Jones G, et al. Mortality rates in transplant recipients and transplantation candidates in a high-prevalence COVID-19 environment. Transplantation. 2021;105(1):212–215. doi: 10.1097/TP.0000000000003533. [DOI] [PubMed] [Google Scholar]
- 31.Willicombe M, Gleeson S, Clarke C, et al. Identification of patient characteristics associated with SARS-CoV-2 infection and outcome in kidney transplant patients using serological screening. Transplantation. 2021;105(1):151–157. doi: 10.1097/TP.0000000000003526. [DOI] [PubMed] [Google Scholar]
- 32.Oto OA, Ozturk S, Turgutalp K, et al. Predicting the outcome of COVID-19 infection in kidney transplant recipients. BMC Nephrol. 2021;22(1):100. doi: 10.1186/s12882-021-02299-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kute VB, Bhalla AK, Guleria S, et al. Clinical profile and outcome of COVID-19 in 250 kidney transplant recipients: A multicenter cohort study from India. Transplantation. 2021;105(4):851–860. doi: 10.1097/TP.0000000000003593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Villanego F, Mazuecos A, Pérez-Flores IM, et al. Predictors of severe COVID-19 in kidney transplant recipients in the different epidemic waves: Analysis of the Spanish Registry. Am J Transplant. 2021;2:2. doi: 10.1111/ajt.16579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Alshaqaq A, Al Abadi A, Altheaby A, et al. Coronavirus disease 2019 and kidney transplantation in Saudi Arabia: Outcomes and future opportunities. Ann. Transplant. 2021;26:e931832. doi: 10.12659/AOT.931832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Requião-Moura LR, Sandes-Freitas TV, Viana LA, et al. High mortality among kidney transplant recipients diagnosed with coronavirus disease 2019: Results from the Brazilian multicenter cohort study. PLoS ONE. 2021;16(7):e0254822. doi: 10.1371/journal.pone.0254822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Naderi GH, Mehraban D, Kazemeyni SM, Darvishi M, Latif AH. Living or deceased donor kidney transplantation: A comparison of results and survival rates among Iranian patients. Transplant Proc. 2009;41(7):2772–2774. doi: 10.1016/j.transproceed.2009.07.041. [DOI] [PubMed] [Google Scholar]
- 38.Nemati E, Einollahi B, Lesan Pezeshki M, Porfarziani V, Fattahi MR. Does kidney transplantation with deceased or living donor affect graft survival? Nephrourol. Mon. 2014;6(4):e12182. doi: 10.5812/numonthly.12182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Nicholson ML, Metcalfe MS, White SA, et al. A comparison of the results of renal transplantation from non-heart-beating, conventional cadaveric, and living donors. Kidney Int. 2000;58(6):2585–2591. doi: 10.1046/j.1523-1755.2000.00445.x. [DOI] [PubMed] [Google Scholar]
- 40.Pratschke J, Dragun D, Hauser IA, et al. Immunological risk assessment: The key to individualized immunosuppression after kidney transplantation. Transplant Rev (Orlando). 2016;30(2):77–84. doi: 10.1016/j.trre.2016.02.002. [DOI] [PubMed] [Google Scholar]
- 41.Wu WK, Famure O, Li Y, Kim SJ. Delayed graft function and the risk of acute rejection in the modern era of kidney transplantation. Kidney Int. 2015;88(4):851–858. doi: 10.1038/ki.2015.190. [DOI] [PubMed] [Google Scholar]
- 42.D'Amico F, Baumgart DC, Danese S, Peyrin-Biroulet L. Diarrhea during COVID-19 infection: Pathogenesis, epidemiology, prevention, and management. Clin. Gastroenterol. Hepatol. 2020;18(8):1663–1672. doi: 10.1016/j.cgh.2020.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ye Q, Wang B, Zhang T, Xu J, Shang S. The mechanism and treatment of gastrointestinal symptoms in patients with COVID-19. Am. J. Physiol. Gastrointest. Liver Physiol. 2020;319(2):G245–G252. doi: 10.1152/ajpgi.00148.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Gao M, Piernas C, Astbury NM, et al. Associations between body-mass index and COVID-19 severity in 6·9 million people in England: A prospective, community-based, cohort study. Lancet Diabetes Endocrinol. 2021;9(6):350–359. doi: 10.1016/S2213-8587(21)00089-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Poly TN, Islam MM, Yang HC, et al. Obesity and mortality among patients diagnosed with COVID-19: a systematic review and meta-analysis. Front Med. 2021;8:620044. doi: 10.3389/fmed.2021.620044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sattar N, McInnes IB, McMurray JJV. Obesity is a risk factor for severe COVID-19 infection: Multiple potential mechanisms. Circulation. 2020;142(1):4–6. doi: 10.1161/CIRCULATIONAHA.120.047659. [DOI] [PubMed] [Google Scholar]
- 47.Bamgbola O. Metabolic consequences of modern immunosuppressive agents in solid organ transplantation. Ther. Adv. Endocrinol. Metab. 2016;7(3):110–127. doi: 10.1177/2042018816641580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Osmanodja B, Mayrdorfer M, Halleck F, Choi M, Budde K. Undoubtedly, kidney transplant recipients have a higher mortality due to COVID-19 disease compared to the general population. Transpl. Int. 2021;34(5):769–771. doi: 10.1111/tri.13881. [DOI] [PubMed] [Google Scholar]
- 49.Effect of hydroxychloroquine in hospitalized patients with COVID-19. New England Journal of Medicine. 2020;383(21):2030–40. [DOI] [PMC free article] [PubMed]
- 50.A Neutralizing Monoclonal Antibody for Hospitalized Patients with Covid-19. New England Journal of Medicine. 2020. [DOI] [PMC free article] [PubMed]
- 51.Cavalcanti AB, Zampieri FG, Rosa RG, et al. Hydroxychloroquine with or without azithromycin in mild-to-moderate COVID-19. N. Engl. J. Med. 2020;383(21):2041–2052. doi: 10.1056/NEJMoa2019014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Goldman JD, Lye DCB, Hui DS, et al. Remdesivir for 5 or 10 days in patients with severe Covid-19. N. Engl. J. Med. 2020;383(19):1827–1837. doi: 10.1056/NEJMoa2015301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Simonovich VA, Burgos Pratx LD, Scibona P, et al. A Randomized Trial of Convalescent Plasma in Covid-19 Severe Pneumonia. New England Journal of Medicine. 2020. [DOI] [PMC free article] [PubMed]
- 54.Stone JH, Frigault MJ, Serling-Boyd NJ, et al. Efficacy of tocilizumab in patients hospitalized with COVID-19. N. Engl. J. Med. 2020;383(24):2333–2344. doi: 10.1056/NEJMoa2028836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wang Y, Zhang D, Du G, et al. Remdesivir in adults with severe COVID-19: A randomised, double-blind, placebo-controlled, multicentre trial. Lancet. 2020;395(10236):1569–1578. doi: 10.1016/S0140-6736(20)31022-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of COVID-19—final report. N. Engl. J. Med. 2020;383(19):1813–1826. doi: 10.1056/NEJMoa2007764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Kalil AC, Patterson TF, Mehta AK, et al. Baricitinib plus remdesivir for hospitalized adults with Covid-19. N. Engl. J. Med. 2020;2:2. doi: 10.1056/NEJMoa2031994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Libster R, Pérez Marc G, Wappner D, et al. Early high-titer plasma therapy to prevent severe COVID-19 in older adults. N. Engl. J. Med. 2021;2:2. doi: 10.1056/NEJMoa2033700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Salama C, Han J, Yau L, et al. Tocilizumab in patients hospitalized with COVID-19 pneumonia. N. Engl. J. Med. 2020;384(1):20–30. doi: 10.1056/NEJMoa2030340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Spinner CD, Gottlieb RL, Criner GJ, et al. Effect of remdesivir vs standard care on clinical status at 11 days in patients with moderate COVID-19: A randomized clinical trial. JAMA. 2020;324(11):1048–1057. doi: 10.1001/jama.2020.16349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Dexamethasone in Hospitalized Patients with Covid-19 — Preliminary Report. N. Engl. J. Med. 2020. [DOI] [PMC free article] [PubMed]
- 62.Bellgrau D, Modiano JF. The cytokine storm-An appropriate, over-reactive response to SARS-CoV-2 or the wrong immune pathway? Scand. J. Immunol. 2020;2:e12979. doi: 10.1111/sji.12979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Khadke S, Ahmed N, Ahmed N, et al. Harnessing the immune system to overcome cytokine storm and reduce viral load in COVID-19: A review of the phases of illness and therapeutic agents. Virol. J. 2020;17(1):154. doi: 10.1186/s12985-020-01415-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zhao Z, Wei Y, Tao C. An enlightening role for cytokine storm in coronavirus infection. Clin. Immunol. 2020;2:108615. doi: 10.1016/j.clim.2020.108615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Antwi-Amoabeng D, Kanji Z, Ford B, Beutler BD, Riddle MS, Siddiqui F. Clinical outcomes in COVID-19 patients treated with tocilizumab: An individual patient data systematic review. J. Med. Virol. 2020;92(11):2516–2522. doi: 10.1002/jmv.26038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Luo P, Liu Y, Qiu L, Liu X, Liu D, Li J. Tocilizumab treatment in COVID-19: A single center experience. J. Med. Virol. 2020;92(7):814–818. doi: 10.1002/jmv.25801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Thammathiwat T, Tungsanga S, Tiankanon K, et al. A case of successful treatment of severe COVID-19 pneumonia with favipiravir and tocilizumab in post-kidney transplant recipient. Transpl. Infect. Dis. 2020;2:e13388. doi: 10.1111/tid.13388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Pérez-Sáez MJ, Blasco M, Redondo-Pachón D, et al. Use of tocilizumab in kidney transplant recipients with COVID-19. Am. J. Transplant. 2020;20(11):3182–3190. doi: 10.1111/ajt.16192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Horwitz LI, Jones SA, Cerfolio RJ, et al. Trends in COVID-19 risk-adjusted mortality rates. J. Hosp. Med. 2020;2:2. doi: 10.12788/jhm.3552. [DOI] [PubMed] [Google Scholar]
- 70.Clarke C, Lucisano G, Prendecki M, et al. Informing the risk of kidney transplantation versus remaining on the waitlist in the coronavirus disease 2019 era. Kidney Int. Rep. 2021;6(1):46–55. doi: 10.1016/j.ekir.2020.10.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Massie AB, Boyarsky BJ, Werbel WA, et al. Identifying scenarios of benefit or harm from kidney transplantation during the COVID-19 pandemic: A stochastic simulation and machine learning study. Am. J. Transplant. 2020;20(11):2997–3007. doi: 10.1111/ajt.16117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Fajnzylber J, Regan J, Coxen K, et al. SARS-CoV-2 viral load is associated with increased disease severity and mortality. Nat. Commun. 2020;11(1):5493. doi: 10.1038/s41467-020-19057-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Sharifipour E, Shams S, Esmkhani M, et al. Evaluation of bacterial co-infections of the respiratory tract in COVID-19 patients admitted to ICU. BMC Infect. Dis. 2020;20(1):646. doi: 10.1186/s12879-020-05374-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Malas MB, Naazie IN, Elsayed N, Mathlouthi A, Marmor R, Clary B. Thromboembolism risk of COVID-19 is high and associated with a higher risk of mortality: A systematic review and meta-analysis. EClinicalMedicine. 2020;29:100639. doi: 10.1016/j.eclinm.2020.100639. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.