Abstract
Background
The added value of radiotherapy following neoadjuvant FOLFIRINOX chemotherapy in patients with resectable or borderline resectable pancreatic cancer ((B)RPC) is unclear. The objective of this meta-analysis was to compare outcomes of patients who received neoadjuvant FOLFIRINOX alone or combined with radiotherapy.
Methods
A systematic literature search was performed in Embase, Medline (ovidSP), Web of Science, Scopus, Cochrane, and Google Scholar. The primary endpoint was pooled median overall survival (OS). Secondary endpoints included resection rate, R0 resection rate, and other pathologic outcomes.
Results
We included 512 patients with (B)RPC from 15 studies, of which 7 were prospective nonrandomized studies. In total, 351 patients (68.6%) were treated with FOLFIRINOX alone (8 studies) and 161 patients (31.4%) were treated with FOLFIRINOX and radiotherapy (7 studies). The pooled estimated median OS was 21.6 months (range 18.4–34.0 months) for FOLFIRINOX alone and 22.4 months (range 11.0–37.7 months) for FOLFIRINOX with radiotherapy. The pooled resection rate was similar (71.9% vs. 63.1%, p = 0.43) and the pooled R0 resection rate was higher for FOLFIRINOX with radiotherapy (88.0% vs. 97.6%, p = 0.045). Other pathological outcomes (ypN0, pathologic complete response, perineural invasion) were comparable.
Conclusions
In this meta-analysis, radiotherapy following neoadjuvant FOLFIRINOX was associated with an improved R0 resection rate as compared with neoadjuvant FOLFIRINOX alone, but a difference in survival could not be demonstrated. Randomized trials are needed to determine the added value of radiotherapy following neoadjuvant FOLFIRINOX in patients with (B)PRC.
Supplementary Information
The online version contains supplementary material available at 10.1245/s10434-021-10276-8.
Pancreatic ductal adenocarcinoma is one of the most aggressive solid tumors.1 Although it is only the 12th most common cancer globally, it is one of the leading causes of cancer-related death in developed countries.2 Around 20–30% of patients have resectable or borderline resectable pancreatic cancer [(B)RPC] at diagnosis. In the most recent National Comprehensive Cancer Network (NCCN) and American Society of Clinical Oncology (ASCO) guidelines, neoadjuvant treatment is recommended for patients with BRPC. For patients with resectable tumors, neoadjuvant treatment is considered an alternative to upfront surgery, especially in patients with biochemical findings suggesting systemic disease (e.g., elevated tumor markers).3–5
In the past two decades, numerous studies on neoadjuvant chemoradiotherapy for pancreatic cancer have been performed.6,7 The rationale behind adding radiotherapy to neoadjuvant chemotherapy is to improve locoregional control by sterilizing vessel margins and enhancing the likelihood of a radical (R0) resection, thereby potentially preventing or postponing locoregional recurrence. Indeed, before the era of FOLFIRINOX (5-fluorouracil with leucovorin, irinotecan, and oxaliplatin), several phase 2 and phase 3 studies of neoadjuvant radiotherapy combined with single- or double-agent chemotherapy have consistently shown high R0 resection rates.8–13
Multidrug regimens including FOLFIRINOX and gemcitabine with nab-paclitaxel have shown superiority to gemcitabine in randomized trials in metastatic and adjuvant settings.14–16 Based on extrapolation of these results, FOLFIRINOX is commonly used in the neoadjuvant setting in many centers worldwide nowadays. Two patient-level meta-analyses of observational studies in patients with locally advanced pancreatic cancer (LAPC) and BRPC treated with FOLFIRINOX ± radiotherapy indeed showed promising results.17,18 Due to limited high-level evidence, current guidelines do not draw final conclusions on whether these multidrug regimens should be combined with radiotherapy.3–5 The role of neoadjuvant radiotherapy in addition to neoadjuvant FOLFIRINOX in patients with (B)RPC remains unclear. Published prospective and retrospective observational studies on this topic are small, precluding definitive conclusions on outcomes.
The aim of this systematic review and meta-analysis was to compare outcomes of (B)RPC patients who received neoadjuvant FOLFIRINOX alone versus FOLFIRINOX with neoadjuvant radiotherapy.
Methods
Search Strategy
This systematic review and meta-analysis was performed according to the PRISMA guidelines.19 An extensive librarian-led literature search of Embase, MEDLINE (via OvidSP), Web-of-Science, Scopus, Cochrane Central, and Google Scholar was performed on 18 December 2020. The search strategy included the following terms: “FOLFIRINOX,” “folinic acid,” “fluorouracil,” “irinotecan,” “oxaliplatin,” “drug combination,” “pancreatic cancer,” and relevant variants. A full description of the search strategy is outlined in Supplementary Table 1. No restrictions on publication dates were applied.
Eligibility
Eligible studies reported outcomes for treatment-naïve patients with resectable or borderline resectable pancreatic cancer [(B)RPC] as defined within each study, and who were either treated with neoadjuvant FOLFIRINOX alone (FOLFIRINOX alone group) or with neoadjuvant FOLFIRINOX followed by any type of radiotherapy (FOLFIRINOX with radiotherapy group). To adequately compare the treatment strategies, additional eligibility criteria were applied. Prospective studies were eligible if patients were scheduled to receive either FOLFIRINOX alone or FOLFIRINOX combined with radiotherapy. Retrospective studies were eligible as FOLFIRINOX with radiotherapy study if at least 90% of patients received radiotherapy following FOLFIRINOX and as FOLFIRINOX alone study if less than 10% of patients received additional radiotherapy. Reviews, letters to the editor, case reports, conference abstracts, and articles written in language other than English were excluded.
Selection Procedure and Data Collection
After removal of duplicates, two authors (Q.J. and I.K.) independently screened the abstracts for eligibility. Full-text assessment was performed for all studies that met the inclusion criteria. Articles were excluded if none of the primary or secondary outcomes were reported or if the same cohort was presented in another study. Discordant judgments were addressed through discussion until consensus was achieved. Data were extracted from the articles separately by the first and second author using a standardized data extraction form.
Methodological Assessment
Risk of bias was assessed using the Critical Appraisal Skill Program (CASP) appraisal system, which is designed to systematically assess the methodological quality of studies.20 Publication bias was assessed using a funnel plot.21
Statistical Analysis
The primary outcome was median OS, as reported by the included articles or extracted from the survival curves. The weighted pooled estimate of median OS was calculated using the formula proposed in a previous meta-analysis, with a study-specific weight function based on the number of patients of interest.6 For the primary analysis, the median OS by intention to treat was used (e.g., excluding studies only reporting outcomes for patients who underwent a resection). Furthermore, the pooled weighted median OS in patients who ultimately underwent resection was calculated. For studies reporting the latter outcome from time of resection, the median OS time was increased with the estimated duration of neoadjuvant treatment based on the reported median number of cycles plus 1 month as estimated time between the end of chemotherapy and surgery date. Confidence intervals for median survival estimates were not calculable, and therefore, the range of medians was provided.
Secondary outcomes were progression-free survival (PFS) in patients who underwent resection, resection rate, adjuvant therapy rate, and postoperative outcomes including R0 resection rate (i.e., among patients who underwent resection and among all patients who started neoadjuvant treatment), ypN0 rate, perineural invasion rate, and pathologic complete response rate. For the adjuvant therapy rate, all patients from prospective studies were included in the denominator, since it is likely that this outcome will be known and reported for prospective studies. Patients from retrospective studies were only included in the denominator for the adjuvant therapy rate if this outcome was reported, since the lack of reporting may be due to information bias. Studies only reporting outcomes for patients who ultimately underwent resection were excluded for calculation of the pooled resection rate, yet included for the pooled R0 resection rate and other pathologic outcomes. Random-effects rather than fixed-effects models were used for all pooled analyses to account for potential between-study heterogeneity and I2 was used as a measure of consistency across studies. Pooled analyses were performed using the meta package for R 3.5.0. All tests were two-sided, and a p-value less than .05 was considered statistically significant.
Results
Included Studies
The literature search identified 6160 records. After removal of duplicates, 2947 records were screened for eligibility. Based on title and abstract, 97 studies were selected for full-text assessment of which 15 fulfilled all inclusion criteria (Fig. 1). The reason for exclusion based on full-text assessment is outlined in Supplementary Table 3.
Fig. 1.
PRISMA flowchart showing selection of articles for systematic review and metaanalysis
Table 1 presents the study characteristics of the 15 included studies. In total, 1081 patients with pancreatic cancer were included, of whom 512 met eligibility criteria based on stage and treatment. Eight studies included 351 patients (68.6%) who received neoadjuvant FOLFIRINOX alone, and 7 other studies included 161 patients (31.4%) who received neoadjuvant FOLFIRINOX followed by radiotherapy. Twelve studies reported outcomes for BRPC patients specifically.22–33 Three studies also or solely reported outcomes for patients with resectable pancreatic cancer.34–36 In total, the FOLFIRINOX alone studies included 310 patients (88.3%) with BRPC and 41 patients (11.7%) with resectable pancreatic cancer, whereas all 161 patients (100.0%) in the FOLFIRINOX with radiotherapy studies had BRPC. Four studies included only patients who underwent a resection after neoadjuvant treatment, 25,32,35,36 while the other 11 studies included all patients who started neoadjuvant treatment.
Table 1.
Study characteristics
| Study (reference) | Country | Evidence | Inclusion period | Total a | Total (B)RPC + FFX | Definition resectability | Radiotherapy, No. (%) | Chemotherapy | Radiotherapy | Adjuvant therapy, No. (%) | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Regimen | Neoadj. cycles, median (range) | Type | CRT regimen | Dose, fractions | |||||||||
| FOLFIRINOX alone | |||||||||||||
| Barenboim22 | Israel | Retrospective | 2008–2017 | 100 | 23 | NCCN | 2 (8.7) | FOLFIRINOX | 8 (5–14) | – | – | – | 16 (70.0) |
| Dhir36 | USA | Retrospective | 2011–2017 | 193 | 73b | NCCN | 0 (0.0) | FOLFIRINOX | 3 (IQR 3–4) | – | – | – | 54 (73.9) |
| Okada23 | Japan | Prospective | 2014–2015 | 10 | 10 | NCCN | 0 (0.0) | mFOLFIRINOX e | 6 (4–8) | – | – | – | 8 (80.0) |
| Tinchon24 | Austria | Prospective | 2010–2012 | 12 | 10 | AHPBA/SSO/SSAT | 0 (0.0) | FOLFIRINOX | 4 (4–6) | – | – | – | 0 |
| De Marsh34 | USA | Prospective | 2013–2015 | 21 | 21c | NCCN | 0 (0.0) | mFOLFIRINOX | 4 (NR–4) | – | – | – | 15 (71.4) |
| Kim35 | USA | Retrospective | 2011–2015 | 26 | 18d | NCCN | 0 (0.0) | FOLFIRINOX | 9 (4–12) | – | – | – | 7 (38.9) |
| Medrano25 | France | Retrospective | 2011–2018 | 139 | 121 | NCCN | 0 (0.0) | FOLFIRINOX | 4 (4–16) | – | – | – | 76 (62.8) |
| Yoo26 | South Korea | Retrospective | 2013–2017 | 199 | 75 | NCCN | 0 (0.0) | (m)FOLFIRINOX | 7 (1–41) | – | – | – | NR |
| Total | 700 | 351 | 2 (0.6) | 5 (3–9) | 176 (58.2) | ||||||||
| FOLFIRINOX with radiotherapy | |||||||||||||
| Christians27 | USA | Retrospective | 2010–2012 | 18 | 18 | MCW | 18 (100.0) | FOLFIRINOX | 4 (3–8) | CRT | Gemcitabine/ capecitabine | 50.4 Gy, 28 | 1 (5.6) |
| Katz28 | USA | Prospective | 2013–2014 | 22 | 22 | ALLIANCE | 21 (95.5) | mFOLFIRINOX | 4 (NR) | CRT | Capecitabine | 50.4 Gy, 28 | 10 (45.5) |
| Murphy29 | USA | Prospective | 2012–2016 | 48 | 48 | NR | 44 (91.7) | FOLFIRINOX | 8 (NR) | CRT | Capecitabine/5-fluorouracil | 25.0 Gy, 5 /30.0 Gy, 10 /50.4 Gy, 28 g | 5 (10.4) |
| Shaib30 | USA | Prospective | NR | 13 | 13 | ALLIANCE | 12 (92.3) | mFOLFIRINOX | 4 (NR) | SBRT | – | 36 - 45 Gy, 3 | 0 |
| Tran31 | USA | Prospective | 2011–2017 | 25 | 25 | NCCN | 19 (76.0) | (m)FOLFIRINOX | 6 (NR) | CRT | Gemcitabine | 50.0 Gy, 25 | 0 |
| Bolton32 | USA | Retrospective | 2008–2015 | 195 | 31 | AHPBA/SSO/SSAT | 28 (90.3)f | FOLFIRINOX | 4 (3–5) | CRT | 5-Fluorouracil | NR | NR |
| Mahaseth33 | USA | Retrospective | 2010–2012 | 60 | 4 | NR | 4 (100.0) | mFOLFIRINOX | NR (2–6) | CRT | Gemcitabine/capecitabine | NR | NR |
| Total | 381 | 161 | 146 (90.7) | 4 (4–8) | 16 (6.0) | ||||||||
AHPBA/SSO/SSAT Americas Hepato-Pancreato-Biliary Association/Society of Surgical Oncology/Society for Surgery of the Alimentary Tract, (B)RPC resectable or borderline resectable pancreatic cancer, CRT chemoradiotherapy, FFX FOLFIRINOX, MCW Medical College of Wisconsin, MDACC MD Anderson Cancer Center, NCCN National Comprehensive Cancer Network, No. number, NR not reported, Neoadj. neoadjuvant, SBRT stereotactic body radiation therapy
aTotal patients included in study, bIncluding upfront resectable pancreatic cancer (n = 15), cAll upfront resectable pancreatic cancer, dIncluding upfront resectable pancreatic cancer (n = 5), eModified FOLFIRINOX without bolus 5-fluorouracil and leucovorin, and with reduced dose of irinotecan, fEstimation based on percentage mentioned in manuscript, gDepending on evaluation by multidisciplinary team; short-course chemotherapy with protons or photons for clearly resectable disease, long-course chemoradiotherapy using intensity modulated radiotherapy in case of persistent vascular involvement
Methodological Assessment
Seven studies were prospective nonrandomized studies, and 8 studies had a retrospective design (Table 1). No randomized controlled trials were identified. Results of the methodological assessment and funnel plot assessing publication bias are shown in the supplementary section. No study was assessed to contain high risk of bias (Suppl. Table 2). Based on the 8 studies reporting the primary outcome, there was no convincing evidence of publication bias, though 2 studies may be considered an outlier (Suppl. Fig. 1). Since there were no randomized studies, confounding by indication cannot be ruled out.
Chemotherapy Regimens and Radiotherapy
Details of the chemotherapy and radiotherapy regimens are presented in Table 1. FOLFIRINOX was administered in 9 studies, modified FOLFIRINOX (mFOLFIRINOX) in 5 studies, and 2 studies administered both [(m)FOLFIRINOX]. Dose modifications consisted of the exclusion of 5-fluorouracil bolus in all 7 studies, 2 studies decreased the dose of irinotecan,23,37 and one study also left out leucovorin.23 The median number of administered neoadjuvant FOLFIRINOX cycles ranged from three to 9 cycles. Adjuvant therapy was administered to 176 patients (58.2%) in the FOLFIRINOX only group (6 studies) and 16 patients (6.0%) in the FOLFIRINOX with radiotherapy group (3 studies). Additional single-agent chemotherapy as radiosensitizer was administered to 133 patients (82.6%) in the FOLFIRINOX with radiotherapy group (6 studies).
In the FOLFIRINOX with radiotherapy group, 146 patients (90.7%) received radiotherapy following FOLFIRINOX, compared with 2 patients (0.6%) in the FOLFIRINOX alone group. Patients were treated with radiation and concurrent chemotherapy (CRT) in 6 studies, while a dose-escalating stereotactic body radiation therapy (SBRT) scheme was used in one study. Total administered dose ranged from 25.0 to 50.4 Gy.
Survival Analysis
The pooled median OS for all studies was 22.0 months (range 11.0–37.7 months). By treatment group, the estimated median OS was 21.6 months (range 18.4–34.0 months) in the FOLFIRINOX only group (3 studies) versus 22.4 months (range 11.0–37.7) in the FOLFIRINOX with radiotherapy group (5 studies) (Table 2). In a sensitivity analysis excluding one study in which a dose-escalating SBRT regimen rather than chemoradiotherapy was used, the median OS for the FOLFIRINOX with radiotherapy group (4 studies) was 25.4 months (range 15.8–37.7 months).
Table 2.
Survival outcomes for (B)RPC patients treated with (m)FOLFIRINOX as first-line treatment with or without additional radiotherapy
| Study (reference) | No. of (B)RPC patients | Median FU, months | Baseline for OS and PFS calculations | Median OS, months (95% CI) | Median PFS, months (95% CI) | |
|---|---|---|---|---|---|---|
| All patients | Resected patients | Resected patients | ||||
| FOLFIRINOX alone | ||||||
| Barenboim22 | 23 | 17.0 | Start treatment | 27.9 (NR) | 34.3 (NR) | 13.7 (NR) |
| Dhir36 | 73a | 35.8 | Diagnosis | # | 38.7 (25.7–50.6) | NR |
| Okada23 | 10 | NR | NR | NR | NR | NR |
| Tinchon24 | 10 | 15.4 | Start treatment | Not reached | Not reached | Not reached |
| De Marsh34 | 21b | 27.7 | Start treatment | 34 (12.3-57.6) | 35.5 (15.0–59.2) | 15.2 (10.5–24.1) |
| Kim35 | 13c | 41.4c | Start treatment | # | 34.2 (NR)c | 19.6 (NR)c |
| Medrano25 | 121 | Mean 39 | Diagnosis | # | 45.0 (NR) | 28.0 (NR) |
| Yoo26 | 75 | 40.3 | Start treatment | 18.4 (16.1–20.8) | NR | NR |
| Estimated median survival (months) | 351 | 33.3 | 21.6 (range 18.4–34.0) | 40.4 (range 34.2–45.0) | 22.1 (range 13.7–28.0) | |
| FOLFIRINOX with radiotherapy | ||||||
| Christians27 | 18 | 22.0 | Diagnosis | 15.8 (NR)d | Not reached | NR |
| Katz28 | 22 | NR | Trial registration | 21.7 (15.7–not reached) | 23.1 (NR)d | At 12 months: 53% (33–86); At 18 months: 40% (19–84) |
| Murphy29 | 48 | 18.0 | Start treatment | 37.7 (19.4–not reached) | Not reached | 48.6 (14.4–not reached) |
| Shaib30 | 13 | 18.0 | Trial registration | 11.0 (5.8–not reached) | Not reached (9.3–not reached) | 29.6 (5.1–not reached) |
| Tran31 | 25 | NR | Trial registration | 24.4 (12.6–40.0) | 37.1 (15.4–not reached) | 21.6 (8.2–31.7) |
| Bolton32 | 31 | NR | Resection | # | 42.5 (NR)e | NR |
| Mahaseth33 | 4 | NR | Start treatment | NR | NR | NR |
| Estimated median survival (months) | 161 | 18.8 | 22.4 (range 11.0–37.7) | 33.5 (range 23.1–42.5) | 28.4 (range 18.0–48.6)f | |
(B)RPC resectable or borderline resectable pancreatic cancer, CI confidence interval, FU follow-up, No. number, NR not reported, OS overall survival, PFS progression-free survival
aIncluding upfront resectable pancreatic cancer (n = 15), bAll upfront resectable pancreatic cancer, cSurvival outcomes for subgroup of BRPC patients specifically, dEstimation based on Kaplan–Meier results, eBased on Kaplan–Meier results of BRPC patients who received 4 or more cycles of neoadjuvant FOLFIRINOX, with 3 months added for duration of neoadjuvant treatment and time to surgery, fPooled estimate including a median PFS of 18 months for Katz et al.,28 since the median PFS certainly did not exceed 18 months
#Study only included patients who underwent resection
Eight studies reported the median OS specifically for those patients who underwent a resection after neoadjuvant treatment. For this subgroup, the estimated median OS was 40.4 months (range 34.2–45.0 months) in the FOLFIRINOX alone group (5 studies) versus 33.5 months (range 23.1–42.5 months) in the FOLFIRINOX with radiotherapy group (3 studies). Median OS was not reached in 4 studies.
Median PFS in patients who underwent a resection after neoadjuvant treatment is presented in Table 2. The pooled estimated median PFS was 22.1 months (range 13.7–28.0 months) in the FOLFIRINOX alone group (4 studies) versus 28.4 months (range 18.0–48.6 months) in the FOLFIRINOX with radiotherapy group (4 studies).
Surgical and Pathological Outcomes
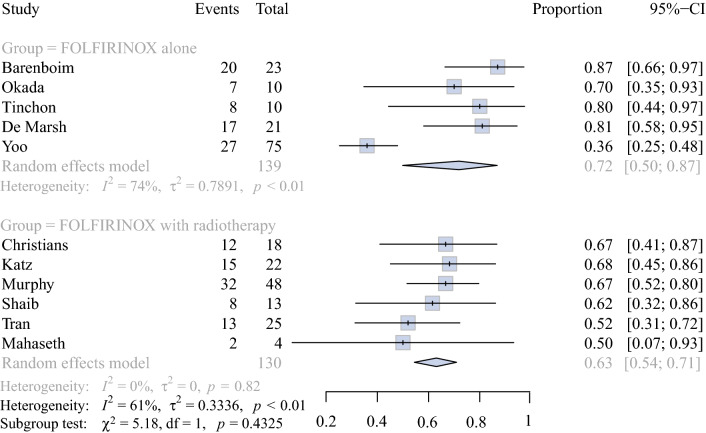
Surgical and pathological outcomes are reported in Table 3. Forest plots of pooled resection and R0 resection rates are shown in Figs. 2 and 3, respectively. The pooled resection rate was 71.9% (79/139 patients, 95% CI: 49.9–86.8%) in the FOLFIRINOX alone group (5 studies) versus 63.1% (82/130 patients, 95% CI: 54.5–70.9%) in the FOLFIRINOX with radiotherapy group (6 studies) (I2 = 61%, p = 0.43) (Fig. 2).
Table 3.
Surgical and pathological outcomes for (B)RPC patients treated with (m)FOLFIRINOX as first-line treatment, with or without additional radiotherapy
| Study (Reference) | No. of (B)RPC patients | Resection rates | ypN0, no. (%)f | Perineural invasion, No. (%)f | Pathological complete response, No. (%)f | |
|---|---|---|---|---|---|---|
| Resection, No. (%) | R0 resection, No. (%)e | |||||
| FOLFIRINOX alone | ||||||
| Barenboim22 | 23 | 20 (87.0) | 20 (100.0) | 16 (80.0)d | 13 (65.0)d | 3 (15.0)d |
| Dhir36 | 73a | # | 62 (84.9) | 32 (43.8) | 57 (78.1) | 3 (4.1) |
| Okada23 | 10 | 7 (70.0) | 5 (71.4) | NR | NR | 0 |
| Tinchon24 | 10 | 8 (80.0) | NR | NR | NR | NR |
| De Marsh34 | 21b | 17 (81.0) | 16 (94.1) | NR | NR | 1 (5.9) |
| Kim35 | 18c | # | 17 (94.4) | 11 (61.1)e | 10 (55.6)e | 0 |
| Medrano25 | 121 | # | 90 (74.4) | 40 (33.1) | 98 (81.0) | 3 (2.5) |
| Yoo26 | 75 | 27 (36.0) | NR | NR | NR | NR |
| Total | 276 | 79 (71.9) | 210 (88.0) | 99 (52.5) | 178 (75.1) | 10 (3.9) |
| FOLFIRINOX with radiotherapy | ||||||
| Christians27 | 18 | 12 (66.7) | 12 (100.0) | 10 (83.3) | NR | 0 |
| Katz28 | 22 | 15 (68.0) | 14 (93.3) | 10 (66.7) | NR | 2 (13.3) |
| Murphy29 | 48 | 32 (66.7) | 31 (96.9) | 20 (62.5) | 22 (68.8) | 0 |
| Shaib30 | 13 | 8 (61.5) | 8 (100.0) | 7 (87.5) | 7 (87.5) | 0 |
| Tran31 | 25 | 13 (52.0) | 13 (100.0) | 6 (46.2) | NR | 0 |
| Bolton32 | 31 | # | NR | NR | NR | 4 (12.9) |
| Mahaseth33 | 4 | 2 (50.0) | 2 (100.0) | 2 (100.0) | NR | NR |
| Total | 161 | 82 (63.1) | 80 (97.6) | 55 (67.1) | 29 (72.5) | 6 (2.9) |
(B)RPC resectable or borderline resectable pancreatic cancer, No. number, NR not reported, ypN0 absence of positive lymph nodes.
aIncluding upfront resectable pancreatic cancer (n = 15), bAll upfront resectable pancreatic cancer, cIncluding upfront resectable pancreatic cancer (n = 5), dEstimated based on percentage of pooled results for borderline resectable (n = 20) and locally advanced pancreatic cancer (n = 3), eEstimated based on percentage of pooled results for resectable and borderline resectable (n = 18) and locally advanced pancreatic cancer (n = 4), fPercentage of patients who underwent a resection
#Studies included only patients who underwent a resection
Fig. 2.
Forest plot showing resection rates in studies with FOLFIRINOX alone versus FOLFIRINOX and radiotherapy (p = 0.43). p-Value calculated using a two-sided Q-test and a random effects model. CI confidence interval, df degrees of freedom
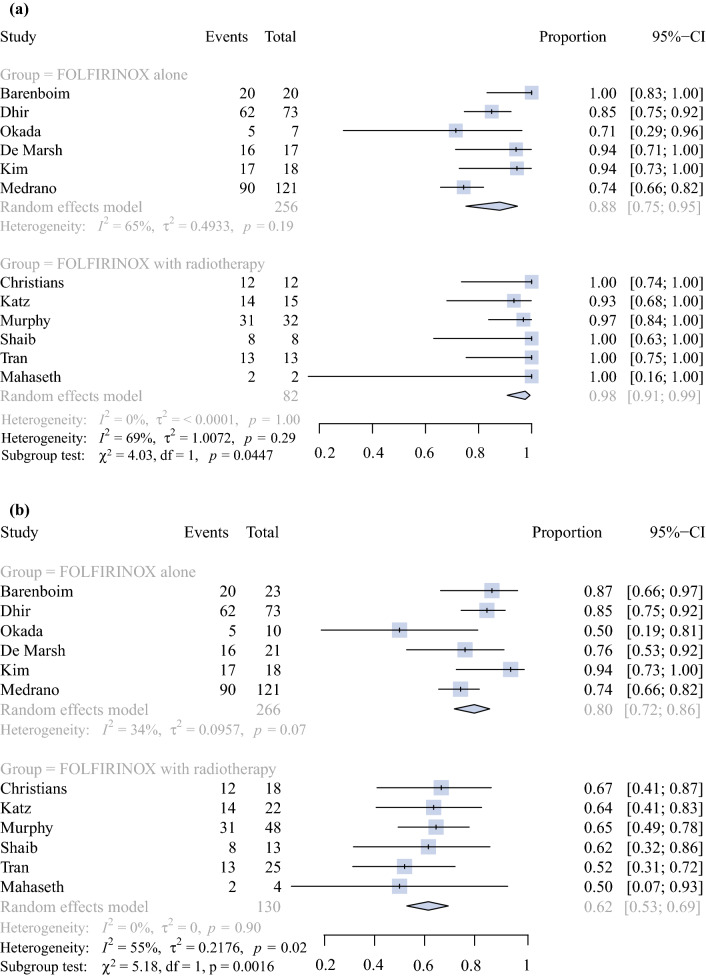
Fig. 3.
Forest plots showing R0 resection rates. (a) Forest plot showing R0 resection rates among patients who underwent a resection in studies with FOLFIRINOX alone versus FOLFIRINOX and radiotherapy (p = 0.04). (b) Forest plot showing R0 resection rates among all patients starting with neoadjuvant treatment in studies with studies with FOLFIRINOX alone versus FOLFIRINOX and radiotherapy (p < 0.01). p-Value calculated using a two-sided Q-test and a random effects model. CI confidence interval, df degrees of freedom
Among the patients who underwent a resection, the pooled R0 resection rate was 88.0% (210/256 patients, 95% CI: 75.2–94.7%) in the FOLFIRINOX alone group (6 studies) versus 97.6% (80/82 patients, 95% CI: 90.8–99.4%) in the FOLFIRINOX with radiotherapy group (6 studies) (I2 = 69%, p = 0.045) (Fig. 3a). The pooled R0 resection rate in all patients starting with FOLFIRINOX was 79.9% (210/266 patients, 95% CI: 71.9–86.1%) in the FOLFIRINOX alone group (6 studies) versus 61.5% (80/130 patients, 95% CI: 52.9–69.5%) in the FOLFIRINOX with radiotherapy group (6 studies) (I2 = 54%, p = 0.002) (Fig. 3b).
The pooled ypN0 rate was 52.5% (99/232 patients, 95% CI: 34.0–70.4%) in the FOLFIRINOX alone group (4 studies) versus 67.1% (55/82 patients, 95% CI: 56.2–76.4%) in the FOLFIRINOX with radiotherapy group (6 studies) (I2 = 73%, p = 0.18). The pooled perineural invasion rate was 75.1% (178/232 patients, 95% CI: 63.9–83.7%) in the FOLFIRINOX alone group (4 studies) versus 72.5% (29/40 patients, 95% CI: 56.8–84.1%) in the FOLFIRINOX with radiotherapy group (2 studies) (I2 = 23%, p = 0.77). Pathologic complete response was rare, considering a pooled estimate of 3.9% (10/256 patients, 95% CI: 2.1–7.1%) in the FOLFIRINOX alone group (6 studies) versus 2.9% (6/111 patients, 95% CI: 0.3–21.2%) in the FOLFIRINOX with radiotherapy group (6 studies) (I2 = 33%, p = 0.80).
Discussion
In this systematic review and meta-analysis including 512 patients with (B)RPC, no difference in survival could be demonstrated between treatment with neoadjuvant FOLFIRINOX with radiotherapy or neoadjuvant FOLFIRINOX alone. The pooled resection rate was also similar, but the pooled R0 resection rate was higher for patients receiving FOLFIRINOX with radiotherapy. These findings support the hypothesis that systemic control remains the most important factor for survival in pancreatic cancer in the era of neoadjuvant FOLFIRINOX. However, these results should be interpreted with caution, since they are based on nonrandomized comparisons of small studies. Considering the small subset of patients with upfront resectable disease, the results of our study are mostly applicable to BRPC patients. A patient-level meta-analysis including 283 BRPC patients who received neoadjuvant FOLFIRINOX found a similar median OS of 22.2 months and a similar resection rate of 67.8%.18
The pooled resection rate was comparable between the treatment groups. In contrast, the pooled R0 resection rate among patients undergoing resection, which is most commonly reported in the literature, was superior for the FOLFIRINOX with radiotherapy group. This is consistent with a large retrospective multicentric cohort study from France including BRPC and LAPC patients who underwent a resection after induction FOLFIRINOX combined with chemoradiotherapy (n = 102) or FOLFIRINOX alone (n = 101). This cohort showed higher R0 (89% vs. 76%, p = 0.017) and ypN0 (77% vs. 49%, p < 0.001) resection rates in patients who received both FOLFIRINOX and chemoradiotherapy. In addition, patients with additional chemoradiotherapy had significantly longer OS (median OS: 57.8 vs. 35.5 months; p = 0.007), which could not be demonstrated in the current meta-analysis.38 This may be explained by the inclusion of LAPC patients in the French study.
Focusing on chemotherapy regimens other than FOLFIRINOX with or without radiotherapy, a large Japanese multicentric cohort study included a prospensity-matched analysis of 376 patients with BRPC who received chemotherapy with radiotherapy (mostly gemcitabine- or S1-based chemoradiotherapy) or neoadjuvant chemotherapy alone (mostly gemcitabine + S1). This study showed a higher ypN0 rate (62.2% vs. 34.0%; p < 0.001) and lower locoregional recurrence rate (20.4% vs. 44.6%; p = 0.002) in the chemotherapy with radiotherapy group, yet no difference in R0 resection rate (87.2% vs. 84.1%, p = 0.50) and survival (median OS: 22.5 vs. 29.2 months; p = 0.130) could be demonstrated.39
No difference in pathological complete response rate could be demonstrated. However, a clinically relevant impact of radiotherapy after FOLFIRINOX on pathologic response cannot be ruled out because of the small number of patients. Two recent retrospective studies found a pathologic complete response rate ranging from 6.8 to 16.3% after systemic chemotherapy and radiotherapy.40,41 A large study from the National Cancer Database showed that preoperative radiation was independently associated with a pathologic complete response on multivariable analysis.42 However, it has not been shown that complete response for a few patients translates into an improvement of survival for all patients who receive neoadjuvant radiation.
Patients in the FOLFIRINOX alone studies have clearly received more adjuvant therapy as compared with patients in the FOLFIRINOX with radiotherapy studies. On the other hand, additional single-agent chemotherapy was used as radiosensitizer in 6 out of the 7 FOLFIRINOX with radiotherapy studies. Since both the neoadjuvant chemoradiotherapy and adjuvant therapy mostly included single-agent chemotherapy regimens, the total systemic treatment may have been comparable in the 2 groups, yet this remains uncertain.
SBRT is a new development in the field of radiotherapy.43 By applying image guidance, the tumor can be followed during the radiation (tracking), or radiation can be interrupted when the tumor moves out of the beam (gating). This allows high doses of radiation in a very short period of time with less toxicity than conventional chemoradiotherapy. Several systematic reviews and large epidemiological studies found good results in LAPC.44–46 Moreover, a recent study in the National Cancer Data Base (NCDB) of over 2000 patients with resected upfront resectable pancreatic cancer who received neoadjuvant multiagent chemotherapy without radiotherapy (n = 1355), with conventional radiotherapy (n = 552), or with SBRT (n = 175), showed superior outcomes for the patients receiving SBRT.47 In the propensity-matched analysis, SBRT was associated with a significantly better survival than chemotherapy alone (HR 0.65, 95% CI: 0.47–0.90, p = 0.01) and chemotherapy plus conventional radiotherapy (HR 0.53, 95% CI: 0.37–0.76, p = 0.001). Furthermore, SBRT was associated with a better R0 resection rate (chemotherapy alone 81% vs. chemotherapy + conventional radiotherapy 86% vs. chemotherapy + SBRT 91%; p = 0.0001) and pathologic complete response rate (respectively 2.2% vs. 4.9% vs. 6.1%; p = 0.0002).47 In line with the current study, this suggests that future randomized studies of neoadjuvant treatment should focus on modern, multiagent chemotherapy in combination with SBRT rather than conventional radiotherapy.
Another new development in the field of radiotherapy for pancreatic cancer is combining radiotherapy with immunotherapeutic agents.48,49 Both in vitro and in vivo studies have shown that radiotherapy may act as an “in situ vaccine” by increasing the expression of cell surface receptors such as major histocompatibility complex class I (MHC-I) and by increasing tumor antigen presentation.50–52 However, due to the immune suppressive tumor microenvironment in pancreatic cancer, the antitumor immune response induced by radiotherapy alone may not be sufficient.53 When combined, the increased release of tumor-specific antigens by radiotherapy may enhance the efficacy of immotherapeutic drugs, potentially resulting in a robust and targeted antitumor immune response.48,54
Four ongoing randomized controlled trials may provide better insights in the individual contributions of systemic chemotherapy and radiotherapy for BRPC patients.55–57 In the ALLIANCE trial A021501, 134 BRPC patients are randomized to neoadjuvant mFOLFIRINOX (8 cycles) or neoadjuvant mFOLFIRINOX (7 cycles) plus SBRT, with surgery and adjuvant FOLFOX in both arms.55 In the French PANDAS-PRODIGE 44 trial (NCT02676349), 90 BRPC patients are randomized to neoadjuvant mFOLFIRINOX (8 cycles) or neoadjuvant mFOLFIRINOX (8 cycles) with subsequent capecitabine-based chemoradiotherapy, followed by surgery and adjuvant gemcitabine or 5-FU in both arms. Results of these 2 studies are expected in 2021. The Chinese BRPCNCC-1 trial is a three-arm trial that randomizes 150 BRPC patients to neoadjuvant gemcitabine plus nab-paclitaxel alone, neoadjuvant gemcitabine plus nab-paclitaxel with SBRT, or neoadjuvant S1 plus nab-paclitaxel with SBRT, with expected results in 2022.56 Finally, the Dutch PREOPANC-2 trial has completed accrual of 368 (B)RPC patients who were randomized to total neoadjuvant FOLFIRINOX (8 cycles) or neoadjuvant gemcitabine-based chemoradiotherapy and adjuvant gemcitabine, with results expected in 2022.57
Some limitations should be taken into account when interpreting the results of our study. First, no randomized trial was included that directly compared FOLFIRINOX with or without radiotherapy. Half of the studies were retrospective studies with potential confounding by indication and information bias. Furthermore, many studies included only small numbers of patients with (B)RPC. Together, these factors have limited the quality of the included studies. Second, our primary endpoint was the estimated median survival time, whereby studies were weighted based on the number of study participants. This weighted estimate of median OS is an imperfect analytical method but a conventional meta-analytical method in the absence of hazard ratios or patient-level data. Third, only one study focused primarily on the addition of radiotherapy to FOLFIRINOX in a dose-finding phase 1 design. This was the only study concerning SBRT. All other studies included conventional chemoradiotherapy, which, as suggested earlier, may not be ideal in this setting. Fourth, heterogeneity across the included studies might have influenced the results, with differences in neoadjuvant FOLFIRINOX treatment (e.g., number of cycles and dose modifications), radiotherapy treatment (e.g., doses, fractions, and concurrent chemotherapy), and different definitions for (B)RPC. This heterogeneity was anticipated by using random effects for all pooled analyses. Last, not all endpoints were reported in several studies, resulting in less precise and potentially biased estimates. Despite these unavoidable limitations, considering the available evidence, the results of the present meta-analysis currently provide the best available comparison of FOLFIRINOX with or without additional radiotherapy in patients with (B)RPC.
In conclusion, radiotherapy following neoadjuvant FOLFIRINOX was associated with an improved R0 resection rate as compared with neoadjuvant FOLFIRINOX alone, but a difference in survival could not be demonstrated. Randomized trials are needed to determine the added value of radiotherapy following neoadjuvant FOLFIRINOX in patients with (B)PRC.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgement
The authors wish to thank W. Bramer from the Erasmus MC Medical Library for developing and updating the search strategies, and the Dutch Cancer Society and ZonMw for their financial support.
Funding
This study is supported by the Dutch Cancer Society (10955) and by ZonMw (843004108). No funding agency was involved in the design of the study, or the collection, analysis, and interpretation of data. The authors are solely responsible for the content of the study and do not necessarily represent the viewpoint of the Dutch Cancer Society or ZonMw.
Disclosure
Alice Wei: Histosonics—consultant, AstraZeneca—consultant, Shire—honorarium, Celgene—honorarium, Bayer—travel, Intuitive Surgical—travel.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics. CA Cancer J Clin. 2020;70(1):7–30. doi: 10.3322/caac.21590. [DOI] [PubMed] [Google Scholar]
- 2.Ferlay J, Ervik M, Lam F, et al. Global Cancer Observatory: cancer today. https://gco.iarc.fr/today. Accessed 20 Aug 2020.
- 3.Tempero MA, Malafa MP, Chiorean EG, et al. Pancreatic adenocarcinoma, Version 1.2019. J Natl Compr Cancer Netw. 2019;17(3):202–210. doi: 10.6004/jnccn.2019.0014. [DOI] [PubMed] [Google Scholar]
- 4.Khorana AA, McKernin SE, Berlin J, et al. Potentially curable pancreatic adenocarcinoma: ASCO clinical practice guideline update. J Clin Oncol. 2019;37(23):2082–2088. doi: 10.1200/JCO.19.00946. [DOI] [PubMed] [Google Scholar]
- 5.Palta M, Godfrey D, Goodman KA, et al. Radiation therapy for pancreatic cancer: executive summary of an ASTRO clinical practice guideline. Pract Radiat Oncol. 2019;9(5):322–332. doi: 10.1016/j.prro.2019.06.016. [DOI] [PubMed] [Google Scholar]
- 6.Gillen S, Schuster T, Meyer Zum Buschenfelde C, Friess H, Kleeff J. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med. 2010;7(4):e1000267. doi: 10.1371/journal.pmed.1000267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Versteijne E, Vogel JA, Besselink MG, et al. Meta-analysis comparing upfront surgery with neoadjuvant treatment in patients with resectable or borderline resectable pancreatic cancer. Br J Surg. 2018;105(8):946–958. doi: 10.1002/bjs.10870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Kim EJ, Ben-Josef E, Herman JM, et al. A multi-institutional phase 2 study of neoadjuvant gemcitabine and oxaliplatin with radiation therapy in patients with pancreatic cancer. Cancer. 2013;119(15):2692–2700. doi: 10.1002/cncr.28117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Van Buren G, Ramanathan RK, Krasinskas AM, et al. Phase II study of induction fixed-dose rate gemcitabine and bevacizumab followed by 30 Gy radiotherapy as preoperative treatment for potentially resectable pancreatic adenocarcinoma. Ann Surg Oncol. 2013;20(12):3787–3793. doi: 10.1245/s10434-013-3161-9. [DOI] [PubMed] [Google Scholar]
- 10.Varadhachary GR, Wolff RA, Crane CH, et al. Preoperative gemcitabine and cisplatin followed by gemcitabine-based chemoradiation for resectable adenocarcinoma of the pancreatic head. J Clin Oncol. 2008;26(21):3487–3495. doi: 10.1200/JCO.2007.15.8642. [DOI] [PubMed] [Google Scholar]
- 11.Casadei R, Di Marco M, Ricci C, et al. Neoadjuvant chemoradiotherapy and surgery versus surgery alone in resectable pancreatic cancer: a single-center prospective, randomized, controlled trial which failed to achieve accrual targets. J Gastrointest Surg. 2015;19(10):1802–1812. doi: 10.1007/s11605-015-2890-4. [DOI] [PubMed] [Google Scholar]
- 12.Jang JY, Han Y, Lee H, et al. Oncological benefits of neoadjuvant chemoradiation with gemcitabine versus upfront surgery in patients with borderline resectable pancreatic cancer: a prospective, randomized, open-label, multicenter phase 2/3 trial. Ann Surg. 2018;268(2):215–222. doi: 10.1097/SLA.0000000000002705. [DOI] [PubMed] [Google Scholar]
- 13.Versteijne E, Suker M, Groothuis K, et al. Preoperative chemoradiotherapy versus immediate surgery for resectable and borderline resectable pancreatic cancer: results of the dutch randomized phase III PREOPANC trial. J Clin Oncol. 2020;38(16):1763–1773. doi: 10.1200/JCO.19.02274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364(19):1817–1825. doi: 10.1056/NEJMoa1011923. [DOI] [PubMed] [Google Scholar]
- 15.Conroy T, Hammel P, Hebbar M, et al. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer. N Engl J Med. 2018;379(25):2395–2406. doi: 10.1056/NEJMoa1809775. [DOI] [PubMed] [Google Scholar]
- 16.Von Hoff DD, Ervin T, Arena FP, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med. 2013;369(18):1691–1703. doi: 10.1056/NEJMoa1304369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Suker M, Beumer BR, Sadot E, et al. FOLFIRINOX for locally advanced pancreatic cancer: a systematic review and patient-level meta-analysis. Lancet Oncol. 2016;17(6):801–810. doi: 10.1016/S1470-2045(16)00172-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Janssen QP, Buettner S, Suker M, et al. Neoadjuvant FOLFIRINOX in Patients with borderline resectable pancreatic cancer: a systematic review and patient-level meta-analysis. JNCI. 2019;111(8):782–794. doi: 10.1093/jnci/djz073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Moher D, Liberati A, Tetzlaff J, Altman DG, Group P Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med. 2009;6(7):e1000097. doi: 10.1371/journal.pmed.1000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Oxford Centre for Triple Value Healthcare. Critical Appraisal Skills Programme (CASP). https://casp-uk.net/wp-content/uploads/2018/03/CASP-Cohort-Study-Checklist-2018_fillable_form.pdf. Accessed Feb 2020.
- 21.Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315(7109):629–634. doi: 10.1136/bmj.315.7109.629. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Barenboim A, Lahat G, Geva R, et al. Neoadjuvant FOLFIRINOX for locally advanced and borderline resectable pancreatic cancer: an intention to treat analysis. Eur J Surg Oncol. 2018;44(10):1619–1623. doi: 10.1016/j.ejso.2018.07.057. [DOI] [PubMed] [Google Scholar]
- 23.Okada KI, Kawai M, Hirono S, et al. Impact of treatment duration of neoadjuvant FIRINOX in patients with borderline resectable pancreatic cancer: a pilot trial. Cancer Chemother Pharmacol. 2016;78(4):719–726. doi: 10.1007/s00280-016-3121-8. [DOI] [PubMed] [Google Scholar]
- 24.Tinchon C, Hubmann E, Pichler A, et al. Safety and efficacy of neoadjuvant FOLFIRINOX treatment in a series of patients with borderline resectable pancreatic ductal adenocarcinoma. Acta Oncol. 2013;52(6):1231–1234. doi: 10.3109/0284186X.2013.771821. [DOI] [PubMed] [Google Scholar]
- 25.Medrano J, Garnier J, Ewald J, et al. Patient outcome according to the 2017 international consensus on the definition of borderline resectable pancreatic ductal adenocarcinoma. Pancreatology. 2020;20(2):223–228. doi: 10.1016/j.pan.2019.12.001. [DOI] [PubMed] [Google Scholar]
- 26.Yoo C, Hwang I, Song TJ, et al. FOLFIRINOX in borderline resectable and locally advanced unresectable pancreatic adenocarcinoma. Ther Adv Med Oncol. 2020;12:1758835920953294. doi: 10.1177/1758835920953294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Christians KK, Tsai S, Mahmoud A, et al. Neoadjuvant FOLFIRINOX for borderline resectable pancreas cancer: a new treatment paradigm? Oncologist. 2014;19(3):266–274. doi: 10.1634/theoncologist.2013-0273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Katz MHG, Shi Q, Ahmad SA, et al. Preoperative modified FOLFIRINOX treatment followed by capecitabine-based chemoradiation for borderline resectable pancreatic cancer alliance for clinical trials in oncology trial A021101. JAMA Surg. 2016;151(8):e161137. doi: 10.1001/jamasurg.2016.1137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Murphy JE, Wo JY, Ryan DP. Total neoadjuvant therapy with FOLFIRINOX followed by individualized chemoradiotherapy for borderline resectable pancreatic adenocarcinoma: a phase 2 clinical trial (vol 4, pg 963, 2018) JAMA Oncol. 2018;4(10):1439–1449. doi: 10.1001/jamaoncol.2018.0329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shaib WL, Hawk N, Cassidy RJ, et al. A phase 1 study of stereotactic body radiation therapy dose escalation for borderline resectable pancreatic cancer after modified FOLFIRINOX ( NCT01446458) Int J Radiat Oncol Biol Phys. 2016;96(2):296–303. doi: 10.1016/j.ijrobp.2016.05.010. [DOI] [PubMed] [Google Scholar]
- 31.Tran NH, Sahai V, Griffith KA, et al. Phase 2 trial of neoadjuvant FOLFIRINOX and intensity modulated radiation therapy concurrent with fixed-dose rate-gemcitabine in patients with borderline resectable pancreatic cancer. Int J Radiat Oncol Biol Phys. 2020;106(1):124–133. doi: 10.1016/j.ijrobp.2019.08.057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Bolton NM, Maerz AH, Brown RE, Bansal M, Bolton JS, Conway WC. Multiagent neoadjuvant chemotherapy and tumor response are associated with improved survival in pancreatic cancer. HPB. 2019;21(4):413–418. doi: 10.1016/j.hpb.2018.08.013. [DOI] [PubMed] [Google Scholar]
- 33.Mahaseth H, Brutcher E, Kauh J, et al. Modified FOLFIRINOX regimen with improved safety and maintained efficacy in pancreatic adenocarcinoma. Pancreas. 2013;42(8):1311–1315. doi: 10.1097/MPA.0b013e31829e2006. [DOI] [PubMed] [Google Scholar]
- 34.Marsh RD, Talamonti MS, Baker MS, et al. Primary systemic therapy in resectable pancreatic ductal adenocarcinoma using mFOLFIRINOX: a pilot study. J Surg Oncol. 2018;117(3):354–362. doi: 10.1002/jso.24872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kim SS, Nakakura EK, Wang ZJ, et al. Preoperative FOLFIRINOX for borderline resectable pancreatic cancer: is radiation necessary in the modern era of chemotherapy? J Surg Oncol. 2016;114(5):587–596. doi: 10.1002/jso.24375. [DOI] [PubMed] [Google Scholar]
- 36.Dhir M, Zenati MS, Hamad A, et al. Folfirinox versus gemcitabine/nab-paclitaxel for neoadjuvant treatment of resectable and borderline resectable pancreatic adenocarcinoma: a propensity matched analysis. Ann Surg Oncol. 2018;25(1):S8. doi: 10.1245/s10434-018-6512-8. [DOI] [PubMed] [Google Scholar]
- 37.Yoo C, Hwang I, Song TJ, et al. FOLFIRINOX in borderline resectable and locally advanced unresectable pancreatic adenocarcinoma. Ther Adv Med Oncol. 2020 doi: 10.1177/1758835920953294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pietrasz D, Turrini O, Vendrely V, et al. How does chemoradiotherapy following induction FOLFIRINOX improve the results in resected borderline or locally advanced pancreatic adenocarcinoma? An AGEO-FRENCH multicentric cohort. Ann Surg Oncol. 2019;26(1):109–117. doi: 10.1245/s10434-018-6931-6. [DOI] [PubMed] [Google Scholar]
- 39.Nagakawa Y, Sahara Y, Hosokawa Y, et al. Clinical impact of neoadjuvant chemotherapy and chemoradiotherapy in borderline resectable pancreatic cancer: analysis of 884 patients at facilities specializing in pancreatic surgery. Ann Surg Oncol. 2019;26(6):1629–1636. doi: 10.1245/s10434-018-07131-8. [DOI] [PubMed] [Google Scholar]
- 40.Zakem SJ, Mueller AC, Meguid C, et al. Impact of neoadjuvant chemotherapy and stereotactic body radiation therapy (SBRT) on R0 resection rate for borderline resectable and locally advanced pancreatic cancer. HPB (Oxford). 2020 doi: 10.1016/j.hpb.2020.11.004. [DOI] [PubMed] [Google Scholar]
- 41.Neyaz A, Tabb ES, Shih A, et al. Pancreatic ductal adenocarcinoma: tumour regression grading following neoadjuvant FOLFIRINOX and radiation. Histopathology. 2020;77(1):35–45. doi: 10.1111/his.14086. [DOI] [PubMed] [Google Scholar]
- 42.Cloyd JM, Ejaz A, Shen C, et al. Pathologic complete response following neoadjuvant therapy for pancreatic ductal adenocarcinoma: defining the incidence, predictors, and outcomes. HPB (Oxford). 2020;22(11):1569–1576. doi: 10.1016/j.hpb.2020.01.013. [DOI] [PubMed] [Google Scholar]
- 43.Ghaly M, Gogineni E, Saif MW. The evolving field of stereotactic body radiation therapy in pancreatic cancer. Pancreas (Fairfax). 2019;3(1):9–14. doi: 10.17140/POJ-3-110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.de Geus SWL, Eskander MF, Kasumova GG, et al. Stereotactic body radiotherapy for unresected pancreatic cancer: a nationwide review. Cancer. 2017;123(21):4158–4167. doi: 10.1002/cncr.30856. [DOI] [PubMed] [Google Scholar]
- 45.Tchelebi LT, Lehrer EJ, Trifiletti DM, et al. Conventionally fractionated radiation therapy versus stereotactic body radiation therapy for locally advanced pancreatic cancer (CRiSP): an international systematic review and meta-analysis. Cancer. 2020;126(10):2120–2131. doi: 10.1002/cncr.32756. [DOI] [PubMed] [Google Scholar]
- 46.Zhong J, Patel K, Switchenko J, et al. Outcomes for patients with locally advanced pancreatic adenocarcinoma treated with stereotactic body radiation therapy versus conventionally fractionated radiation. Cancer. 2017;123(18):3486–3493. doi: 10.1002/cncr.30706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xiang M, Heestand GM, Chang DT, Pollom EL. Neoadjuvant treatment strategies for resectable pancreas cancer: a propensity-matched analysis of the National Cancer Database. Radiother Oncol. 2020;143:101–107. doi: 10.1016/j.radonc.2020.01.007. [DOI] [PubMed] [Google Scholar]
- 48.Formenti SC, Demaria S. Radiation therapy to convert the tumor into an in situ vaccine. Int J Radiat Oncol Biol Phys. 2012;84(4):879–880. doi: 10.1016/j.ijrobp.2012.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Dalgleish AG, Stebbing J, Adamson DJ, et al. Randomised, open-label, phase II study of gemcitabine with and without IMM-101 for advanced pancreatic cancer. Br J Cancer. 2016;115(7):789–796. doi: 10.1038/bjc.2016.271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gaugler MH, Squiban C, van der Meeren A, Bertho JM, Vandamme M, Mouthon MA. Late and persistent up-regulation of intercellular adhesion molecule-1 (ICAM-1) expression by ionizing radiation in human endothelial cells in vitro. Int J Radiat Biol. 1997;72(2):201–209. doi: 10.1080/095530097143428. [DOI] [PubMed] [Google Scholar]
- 51.Garnett CT, Palena C, Chakraborty M, Tsang KY, Schlom J, Hodge JW. Sublethal irradiation of human tumor cells modulates phenotype resulting in enhanced killing by cytotoxic T lymphocytes. Cancer Res. 2004;64(21):7985–7994. doi: 10.1158/0008-5472.CAN-04-1525. [DOI] [PubMed] [Google Scholar]
- 52.Reits EA, Hodge JW, Herberts CA, et al. Radiation modulates the peptide repertoire, enhances MHC class I expression, and induces successful antitumor immunotherapy. J Exp Med. 2006;203(5):1259–1271. doi: 10.1084/jem.20052494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lee Y, Auh SL, Wang Y, et al. Therapeutic effects of ablative radiation on local tumor require CD8+ T cells: changing strategies for cancer treatment. Blood. 2009;114(3):589–595. doi: 10.1182/blood-2009-02-206870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Gandhi SJ, Minn AJ, Vonderheide RH, Wherry EJ, Hahn SM, Maity A. Awakening the immune system with radiation: optimal dose and fractionation. Cancer Lett. 2015;368(2):185–190. doi: 10.1016/j.canlet.2015.03.024. [DOI] [PubMed] [Google Scholar]
- 55.Katz MHG, Ou FS, Herman JM, et al. Alliance for clinical trials in oncology (ALLIANCE) trial A021501: preoperative extended chemotherapy versus chemotherapy plus hypofractionated radiation therapy for borderline resectable adenocarcinoma of the head of the pancreas. BMC Cancer. 2017;17(1):505. doi: 10.1186/s12885-017-3441-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Gao S, Zhu X, Shi X, et al. Comparisons of different neoadjuvant chemotherapy regimens with or without stereotactic body radiation therapy for borderline resectable pancreatic cancer: study protocol of a prospective, randomized phase II trial (BRPCNCC-1) Radiat Oncol. 2019;14(1):52. doi: 10.1186/s13014-019-1254-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Janssen QP, van Dam JL, Bonsing BA, et al. Total neoadjuvant FOLFIRINOX versus neoadjuvant gemcitabine-based chemoradiotherapy and adjuvant gemcitabine for resectable and borderline resectable pancreatic cancer (PREOPANC-2 trial): study protocol for a nationwide multicenter randomized controlled trial. BMC Cancer. 2021;21(1):300. doi: 10.1186/s12885-021-08031-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.