Abstract
The rising field of RNA modifications is stimulating massive research nowadays. m6A, the most abundant mRNA modification is highly conserved during evolution. Through the last decade, the essential components of this dynamic mRNA modification machinery were found and classified into writer, eraser and reader proteins. m6A modification is now known to take part in diverse biological processes such as embryonic development, cell circadian rhythms and cancer stem cell proliferation. In addition, there is already firm evidence for the importance of m6A modification in stem cell differentiation and gametogenesis, both in males and females. This review attempts to summarize the important results of recent years studying the mechanism underlying stem cell differentiation and gametogenesis processes.
Keywords: RNA modification, N6-methyladenosine, m6A, stem cells, spermatogenesis, oogenesis
1. Introduction
Epigenetics, most commonly, refers to heritable changes in gene expression which are not linked to changes in the DNA sequence. As such, chemical modifications on DNA and histones were studied for decades, revealing the importance of epigenetic modifications in gene expression regulation. In recent years another level of gene expression regulation is under extensive research: the field of post-transcriptional RNA modifications, also known as the epitranscriptome [1]. These RNA modifications, which can be added post-transcriptionally, are another layer of gene expression regulation. For now, more than 170 modifications were found to be added post-transcriptionally to RNA molecules, the vast majority are on tRNA and rRNA [2]. N6-methyladenosine (m6A) is the most abundant modification on polyadenylated RNA [3] (Table 1).
Table 1.
Known mRNA modifications.
| Name | Short Name | Formula | Types of Modified RNA |
|---|---|---|---|
| N6-methyladenosine | m6A | C11O4N5H15 | mRNA, rRNA, snRNA, tRNA |
| N6-formyladenosine | f6A | C11H13N5O5 | mRNA |
| N6,2′-O-dimethyladenosine | m6Am | C12O4N5H17 | mRNA, snRNA |
| N6-hydroxymethyladenosine | hm6A | C11H15N5O5 | mRNA |
| 5-Methylcytidine | m5C | C10O5N3H15 | mRNA, rRNA, tRNA |
| 5-Hydroxymethylcytidine | hm5C | C10O6N3H15 | mRNA |
| 7-Methylguanosine cap (cap 0) | m7Gpp(pN) | C11H15N5O11P2 | mRNA, snRNA |
| Inosine | I | C10O5N4H12 | mRNA, tRNA |
| Pseudouridine | ψ | C9O6N2H12 | mRNA, mRNA, rRNA, snRNA, snoRNA, tRNA |
| N1-mthyladenosine | m1A | C11O4N5H15 | mRNA, rRNA, tRNA |
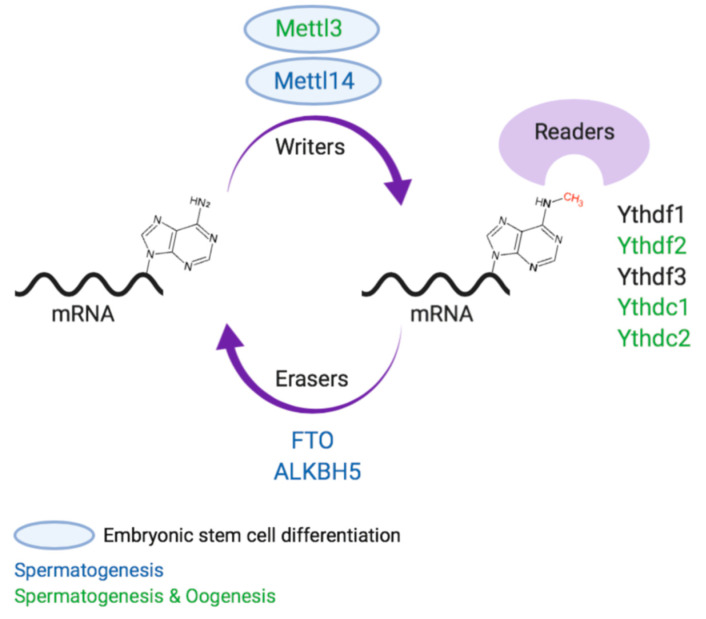
RNA modifications were detected for the first time in the fifties of the last century [4,5]. However, only in the seventies, the most abundant mRNA modification was discovered—the N6-methyladenosine, also known as m6A, in poly(A) RNA fractions [6,7,8,9,10,11]. This modification, which consists of a methyl group added to adenosine (Figure 1) was found in prokaryotes, viruses, plants and mammals [12,13,14,15]; however, at that time there were no available methods to detect m6A sites on mRNA, thus scientist could not determine which specific RNA contains m6A modification. Hence, the research about the RNA modifications was delayed for a few decades.
Figure 1.
Conventions of m6A modification on mRNA, its writer, reader and eraser proteins. Proteins with evidence to have a role in embryonic stem cell differentiation, or in spermatogenesis or oogenesis, are marked in different colors.
In 2012, a leap forward was made in the research of m6A, in the form of the first-ever mapping of the sites of m6A modification in mammals, using a novel method of RNA-IP followed by high-throughput sequencing (MeRIP-Seq/m6A-seq) [16,17]. This method helped to stress the importance of m6A by demonstrating the conservation of its methylated sites during evolution [16,17,18,19]. In addition, by using this method, it has been shown that m6A modification occurs in specific sequence motif DRACH (D = A/G/U, R = G/A, H = A/C/U), and is preferentially localized around stop codons, 3′ UTRs regions and long exons [16,17,20]. Furthermore, these studies showed that the levels of m6A may be dynamic in certain settings, changing due to cellular stress and stages of development.
The notion that the m6A is a dynamic modification was further supported by the discovery of m6A demethylase FTO [21]. It was the first-ever confirmed reversible RNA modification, thus raising the attention to the field. Although nowadays, the latter result is controversial and it is also known that FTO is a demethylase of another modification—N6,2′-O-dimethyladenosine (m6Am) [22]—the idea that m6A modification is dynamically regulated is intriguing. Still, despite the great effort, there is no clear answer so far to whether m6A is dynamically regulated. m6A-seq and meRIP-seq methods are based on antibodies, and as such are limited in their ability to quantify methylation levels, and suffer from false positive hits. A recent work [23] presents a cleaner method for m6A mapping in a single-nucleotide resolution, and shows that the 33%–46% of the methylation is determined by the sequence alone, thus suggesting that the modification is less dynamic than suggested.
Regardless of this question, in recent years new discoveries demonstrated that mRNA modifications play a role in various molecular processes, such as translation efficiency, stability, localization and splicing, thus impacting cell fate [24,25,26,27,28,29,30]. Moreover, these modifications were shown to be crucial for biological processes such as embryonic development, cell circadian rhythms and cancer stem cell proliferation [19,31,32,33,34]. In this review, we focus on m6A mRNA modification and its role in mammalian stem and germ cells. For a review on m6A modification and its mechanisms, we refer you to other comprehensive reviews [35,36,37].
2. m6A Proteins
2.1. m6A “Writers”
In recent years the components necessary for writing m6A modification on RNA molecules were found (Figure 1), allowing the basic research of m6A modification. First to be found, and even before the development of the new methods, was the methyltransferase like 3 protein (METTL3), one part of a larger catalyzing complex [38,39]. METTL3 is widely expressed in different tissues in mouse and human, and when knocking-out METTL3 in mouse embryonic stem cells, no traces of m6A methylation can be found using mass spectrometry [31]. METTL3 was shown to play a major role in development and function of neural cells [40], cardiac cells [41,42], bone and muscle [43], hematopoiesis [44] and gametogenesis [45,46], as well as in cancer progression [47]. It was also shown in 2015 that it is essential for differentiation in vitro and for mouse development in vivo [31].
Later works discovered Wilm’s tumor-associated protein (WTAP) as an METTL3 adaptor protein, a crucial component of the m6A methyltransferase complex [48,49]. WTAP is important for the complex function and when knocked-down, m6A levels in human cells are decreased significantly [25].
Another protein was suggested to be an m6A writer—METTL14 protein. It was shown that METTL3 and METTL14 work in a complex [20]. As such, knock-down of METTL14 also leads to decreased m6A levels [20,25]. Knock-out (KO) of METTL3 or METTL14 results in protein loss of the other writer [31,50], suggesting a coupled stoichiometry. Recent papers demonstrate that METTL14 is not enzymatically active, thus does not have a role as a second methyltransferase, but as an adaptor, helping METTL3 by facilitating RNA binding [51,52,53].
With time, several more proteins have been shown to be important for facilitating the function of the methyltransferase complex and its correct positioning. This includes RNA binding motif protein 15/15B (RBM15/15B), which binds the methylation complex and was shown to methylate XIST [54], Vir like m6A methyltransferase associated (VIRMA), which recruits the methylation complex to 3′ UTR [55], and zinc finger CCCH-type containing 13 (ZC3H13), which modulates the activity of the complex in the nucleus [56]. In addition, METTL16 was discovered as another m6A methyltransferase which adds m6A in a different sequence context and is important for splicing [57,58].
2.2. m6A “Erasers”
There are two proteins which were found to function as m6A mRNA demethylase enzymes (Figure 1)—FTO [21] and α-ketoglutarate-dependent dioxygenase alkB homolog 5 (ALKBH5) [59]. These two m6A erasers have been found to reduce m6A levels following over-expression, and increase m6A levels by 9%–23% following knock-down. The presence of these m6A erasers suggests that m6A modifications can be dynamic and reversible, at least to some extent, in a similar manner to DNA and protein modifications.
However, as mentioned before, the role of FTO as an m6A demethylase is still under debate. It was found that FTO preferentially demethylases m6Am modification, rather than the m6A modification [22]. In 2018 it was shown that FTO bind multiple RNA species and function as demethylase of m1A, m6A and m6Am [60]. In addition, little is known about ALKBH5. Interestingly, ALKBH5-KO mice are viable and normal except for defects in spermatogenesis [59].
2.3. m6A “Readers”
The actual effect of m6A modification is mediated by “readers”, that are m6A binding proteins (Figure 1). m6A reader proteins which predominantly bind to methylated RNA were initially identified in RNA pull-down experiments, including YTHDF2 and YTHDF3 [16]. These cytoplasmatic proteins belong to the YT521-B homology (YTH) domain. Since then, more groups have shown using crystallography and gel shift assays that YTH proteins, including YTHDF2 and YTHDC1, in addition to yeast homolog MRBP1, are m6A binding proteins [61,62,63], eventually concluding that all cytoplasmic YTH domain family (YTHDF1, YTHDF2 and YTHDF3), and nuclear YTH domain containing proteins (YTHDC1, YTHDC2), function as m6A binding proteins [64,65]. YTH domain interacts with methylated adenosine with low affinity, using an aromatic cage that surrounds the methyl group, as concluded from structure studies of MRBP1, YTHDC1 and YTHDF1/2 [64]. Unfortunately, the crystal structure of YTHDF3 has not been achieved so far.
Studies on YTHDF2 demonstrated that its binding motif is identical to the m6A motif, and that by binding to this motif, YTHDF2 regulates m6A-dependent RNA degradation through interaction with CCR4-NOT deadenylase complex [66,67]. YTHDF1 and YTHDF3 were suggested to promote mRNA translation through recruiting translation initiation factors [68,69]. YTHDC1 was linked to mRNA splicing, export and degradation [70,71,72]. YTHDC2 mediates translation and decay specifically in spermatogenesis [65].
With time, more m6A readers were discovered, including HNRNPC/G, HNRNPA2B1, IGF2BP1-3 and FMR1. These proteins contain other types of RNA-binding domains, and a different recognition site (e.g., IGF2BP1-3 bind to GG(m6A)C). They were suggested to be important for mRNA splicing, stability and translation [73,74,75,76,77,78,79]. In 2015 it was suggested that the binding mechanism of HNRNPC/G is different than that of YTH containing readers: the methylation induces a structural change to the mRNA, thus exposing the binding site of HNRNPC/G/A2B1 and facilitating their binding [74]. Interestingly, some m6A binding proteins may work in a competitive manner. For instance, YTHDF2 and IGF2BP1-3 affect mRNA stability in an opposite manner [76]. In this case, they also have different binding preferences—YTHDF2 tends to bind 3′ UTR, and IGF2BP1-3 tends to bind CDS [76]. It is therefore interesting to understand the mechanism that orchestrate the activity of these readers.
m6A modification plays a major role in differentiation. It is therefore natural to use stem cell differentiation, a model which is easy to manipulate, to investigate the different aspects of m6A. Here we will summarize the work that was done to investigate m6A in the context of embryonic stem cells.
3. m6A and Stem Cell Fate
Embryonic stem cells are a unique type of cells, which reside in the early embryo blastocyst, and give rise to all the tissues of the embryo (“pluripotent”). These cells can self-renew, and can be maintained in vitro indefinitely in their pluripotent state. Embryonic stem cells are typically divided into two states—“naïve” state, which is found in the blastocyst inner cell mass, and “primed” state, which is primed to differentiation, and is found in the epiblast. Naïve and primed cells are distinguished in several molecular parameters such as inactivation of X chromosome, activity of OCT4 enhancers and the signaling required for their in vitro maintenance [80]. Key transcription factors such as NANOG, OCT4, SOX2, ESRRB, KLF4, TFAP2C and others form an interconnected circuit orchestrating pluripotency [81].
In 2014, Batista and colleges showed that core pluripotency transcription factors are methylated in mouse and human [19]. Depletion of mouse and human METTL3 was shown to reduce significantly the m6A deposition, resulting in prolonged NANOG and SOX2 expression upon differentiation, and impaired exit from pluripotency [19,31]. By studying m6A modification in vivo using METTL3-KO mice, our group has demonstrated that depletion of m6A in mice is embryonic lethal [31]. The embryos die between embryonic day 3.5 and 6.5, the exact stage in which the cell in the blastocyst inner cell mass are exiting from pluripotent state and starting to differentiate. Teratoma and EB assays confirmed that cells that lack m6A are in a “hyper-pluripotent” state, and had a poor differentiation ability [31]. The erasure of m6A from some of the pluripotent transcripts such as NANOG, SOX2, ZFP42, KLF4 and others, resulted in prolonged half-life. As a consequence, pluripotent circuitry was not turned off, thus emphasizing the importance of m6A in the exit from naïve pluripotent state [31]. Notably, knock-out of METTL14 in mouse ESCs recapitulates METTL3-KO phenotype in vivo and in vitro [82].
Other groups reported that mouse embryonic stem cells with knockdown of METTL3 and METTL14 have lost their self-renewal capability and overexpress developmental regulators [24]. This seeming contradiction can be resolved by the notion that lack of m6A affects the genes that are currently expressed. In the naïve conditions [80], the cells will stay pluripotent, and in primed conditions, in which the cells are primed to differentiation, the cells will lose their self-renewal and differentiate.
Overall, when large-scale changes in the cell are required, such as in differentiation or programming, m6A has an important role in the proper regulation of mRNA. This has been shown in cellular reprogramming, which m6A promotes [83], and during differentiation. For instance, lincRNA1281 was shown to have a crucial role in mESCs differentiation by sequestering let-7 miRNA [84], and this interaction is mediated by m6A modification. In 2018 a novel mechanism of m6A deposition regulation was uncovered by using human embryonic stem cells model. By investigating the interactome of SMAD2/3 during differentiation of human primed pluripotent stem cells, it was shown that SMAD2/3 interacts with the METTL3-METTL14-WTAP complex [85]. Nonetheless, by using nuclear-enriched m6A-methylated-RNA immunoprecipitation followed by deep sequencing, it was demonstrated that activin/TGFβ signaling, mediated by SMAD2/3, promotes m6A deposition on nuclear RNAs. The upregulation of m6A deposition occurs on several transcripts, including transcripts such as NANOG, NODAL and LEFTY1, which are activin signaling targets and function as pluripotency regulators. Thus, inhibition of activin/TGFβ signaling leads to differentiation, meaning that regulation of m6A deposition can be dynamically changed by extracellular signaling [85].
m6A is important for the differentiation of adult stem cells as well. During neural stem cell differentiation, YTHDF2 is responsible for adequate decay of mRNAs that are inhibitory of Jak/Stat cascade [86]. Upon knock-out of YTHDF2, NPCs are unable to produce functional neurites. Other mechanisms that are important for neural differentiation are dependent on m6A [87,88]. Hematopoiesis is also dependent on m6A modification. Hematopoietic stem cells (HSCs) that lack m6A methylation (METTL3-KO) are unable to differentiate, and are accumulated in the bone marrow [44]. Transcriptional analysis revealed that knock-out cells are unable to upregulate m6A-methylated MYC expression, where enforced upregulation of MYC rescued the phenotype [44]. YTHDF2-KO promotes HSCs expansion as well, what has important medical implication [89]. In 2019 it was shown that MYC is responsible for symmetric committed differentiation of HSCs and that upon METTL3-KO, the reduction of MYC brings the cells to be blocked in a multipotent progenitor-like state [90].
To conclude, regulation of mRNA by m6A modification is essential for proper differentiation of embryonic and adult stem cells by multiple pathways. For further reading on m6A in pluripotency and development we refer the readers to the review by Heck and colleges [91].
4. Gametogenesis
Gametogenesis, the process of gametes generation, is another differentiation process which is heavily mediated by m6A modification. Primordial germ cells are the cells that give rise to the gametes of an organism that reproduces sexually. During gametogenesis, these cells undergo meiosis, followed by cellular differentiation into mature gametes—oocytes in females or spermatozoa in males. Early works showed that m6A is essential for proper meiosis in yeast [18,92,93]. It is therefore intriguing to test gametogenesis, the process in which meiosis happens in mammals, and is accompanied by massive degradation of RNA.
4.1. m6A Role in Spermatogenesis
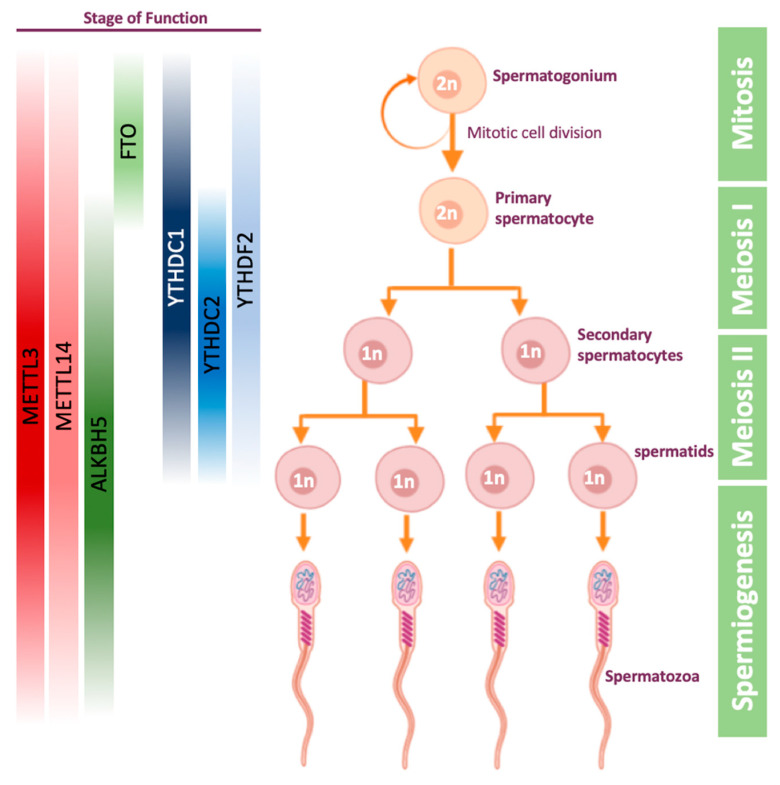
Spermatogenesis is a process undergoing in the seminiferous tubules of the testis, in which germ cells develop into haploid spermatozoa. This process starts with the mitotic division of spermatogonial stem cells, which can either self-review, or give rise to primary spermatocytes (Figure 2). The latter cells then undergo the first meiosis to become secondary spermatocytes. The secondary spermatocytes undergo the second meiosis, and by the end of this process they become haploid spermatids. In the final stage of spermatogenesis, called spermiogenesis, spermatids are transformed into mature and motile spermatozoa, also known as sperm cells (Figure 2).
Figure 2.
Stages of spermatogenesis, and the proteins that have a role during the process. The proteins are highlighted along the stages in which there is some evidence that they play a role. Other stages are yet to be confirmed.
The first evidence for the importance of m6A on gametogenesis was demonstrated in male mice: erasing m6A modification by ALKBH5 was shown to impact mouse spermatogenesis [59]. It was demonstrated that ALKBH5 is highly expressed in mice testis, and that loss of ALKBH5 leads to differentially expressed genes related to spermatogenesis [59]. ALKBH5-deficient male mice were shown to have increased m6A levels on mRNA, significantly smaller testes compared to their WT littermates and impaired fertility due to apoptosis which defects the meiotic metaphase-stage spermatocytes [59]. In 2017, ALKBH5-dependent m6A demethylation was shown to be critical for splicing and stability of 3’ UTR mRNAs in spermatocytes and round spermatids [94]. Later, it was demonstrated that inhibition of FTO using meclofenamic acid leads to an increased level of m6A and decreased expression of few cyclin-dependent kinases, resulting in abnormal spermatogonial proliferation [95].
The understanding that m6A plays a role in spermatogenesis led to other works on writer and reader proteins. It was demonstrated that METTL3 and METTL14 are localized in the nuclei of germ cells in mice testis [96]. Conditional mice knock-out of METTL3 or METTL14 using VASA-Cre line, which activates the knock-out early in migratory primordial germ cells [97], were sterile, with significant reduction of m6A levels in undifferentiated spermatogonia. This led to spermatogonial stem cells defects, causing translational dysregulation of methylated transcripts, including genes that are crucial for proliferation and differentiation of spermatogonial stem cells [96], overall showing that depletion of a single methyltransferase is sufficient to cause a sever phenotype in male mice. In another study, single methyltransferase knock-out mice, METTL3f/fVASA-cre+, were found to have abnormal meiosis initiation and spermatogonial differentiation [46]. These mice were normal in size but completely infertile, with 80% reduction in testis weight at eight weeks old. Furthermore, defects in sperm maturation and motility upon conditional METTL3-KO were detected also in zebrafish, establishing that m6A modification is essential for spermatogenesis outside the mammalian domain [45].
Out of all reader proteins, the protein which studied the most in the context of spermatogenesis, is YTHDC2. YTHDC2 was found to be essential for mouse meiosis, both in males and females, in several different papers [65,98,99,100]. YTHDC2-KO male mice have significantly smaller testes and female mice have significantly smaller ovaries, compared to their littermates [65]. However, it is still unclear whether these mice are sterile due to YTHDC2 ability to recognize the m6A modification using its YTH domain or due to its 3’→5’ RNA helicase activity. YTHDC2 was shown to be expressed in the cytoplasm of mice spermatocytes and interacts with MEIOC, which is an important gene in the meiosis process [101]. When YTHDC2 is knocked-out in male mice, the meiosis is initiated properly but continues aberrantly, finally leading to apoptosis. In addition, it was demonstrated in transcriptome analysis that when YTHDC2 is missing, genes associated with meiosis are downregulated and genes associated with mitotic cells are upregulated, indicating YTHDC2 is essential for ending mitosis and transition to meiotic process [98,99,100].
YTHDC1 is essential for proper embryonic development—YTHDC1-KO mouse embryos die at early post-implantation stages [102]. YTHDC1 is expressed in the nucleus of spermatogonia, spermatocytes, and round spermatids, which are transcriptionally active during spermatogenesis [102]. In addition, YTHDC1f/− VASA-Cre+ mice have reduced spermatogonia on day 8 (post-natal), and no germ cells in their testis from day 25 post-natal and on, indicating that YTHDC1 is not important only for mouse viability, but also for the spermatogenesis process [102].
Few observations yet connect YTHDF readers to spermatogenesis. A work from 2017 showed that YTHDF2-KO male mice are fertile, in contrast to YTHDF2-KO females, and have normal seminiferous tubule histology [103]. Depletion of YTHDF2 leads to downregulation of matrix metallopeptidase, thus affecting cell adhesion and proliferation. This phenotype can be partially rescued by inhibition of MMP13 [104]. However, further examination of all YTHDF readers in spermatogenesis is still required.
4.2. m6A Role in Oogenesis
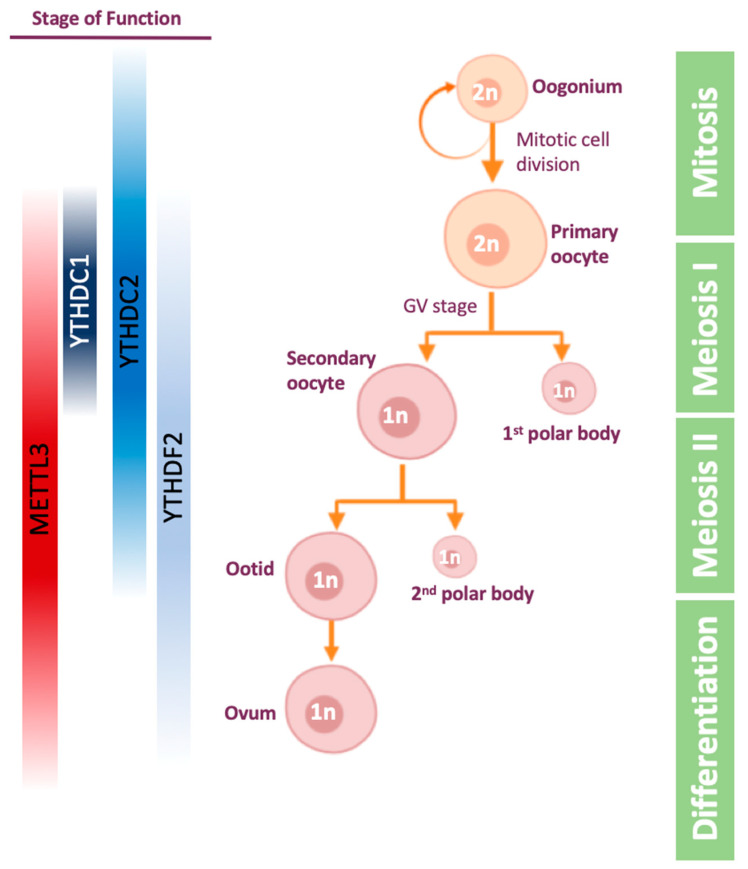
The oogenesis process (Figure 3) is the differentiation of the diploid germ cell, called oogonium, into an ovum (egg cell). The oogonium undergoes transformation into primary oocyte in a process called oocytogenesis. The primary oocyte enters the first meiosis and is arrested in prophase I in germinal vesicle (GV) stage. Following hormonal trigger, the oocyte resumes the first meiosis and becomes secondary oocyte. Then, the secondary oocyte undergoes the second meiosis division and is arrested in metaphase II. If the secondary oocyte is fertilized, meiosis II is resumed and completed.
Figure 3.
Stages of Oogenesis, and the proteins that have a role during the process. The proteins are highlighted along the stages in which there is some evidence that they play a role. Other stages are yet to be confirmed.
The maternal mRNA is transcribed and accumulated during the oocytes’ growth, and transcription ends in GV stage oocytes [105]. Later on, mRNAs are massively degraded when the oocytes proceed the meiosis process following a hormonal trigger [106,107]. The mRNA degradation goes on after fertilization as well, and as a result most of the maternal mRNA is degraded in the 2-cell stage [108]. Thus, mRNA stability appears to be a crucial process in oogenesis and maternal to zygotic transition, highlighting the importance of post-transcription regulation during these processes.
The first evidence for m6A importance during these processes in females was observed in zebrafish, showing that one-third of maternal mRNA is m6A-methylated, and its degradation, mediated by YTHDF2 activity, is important for maternal to zygotic transition and timely development of the zebrafish [109]. A later paper on zebrafish showed that loss of METTL3 results in failed gamete maturation and reduced fertility [45]. In mammals as well, YTHDF2 reader protein was the first link found between m6A and oogenesis: YTHDF2 is expressed throughout mouse oogenesis, as demonstrated by immunostaining [103]. Knocking-out YTHDF2 leads to normal ovulation but an inability to downregulate maternal mRNA. As a result, approximately 270 genes are deregulated and mouse embryo development is defected at or prior to 2-cell stage, leading to infertility [103].
Studying the effect of m6A writers on oogenesis was more challenging due to the fact that METTL3-KO is embryonic lethal in mice [31]. While in spermatogenesis studying this difficulty was solved by using conditional knock-out Cre, METTL3-knock-down female mice were generated by microinjections of siRNAs or morpholino into GV oocytes [110]. The knock-down resulted in reduced translation efficiency, defects in mRNA degradation, and impaired meiotic maturation, including aberrant spindle organization. Overall, it was demonstrated that m6A is crucial for oocyte maturation and maternal-to-zygotic transition [110].
As mentioned before, YTHDC2 also affects oogenesis. It was shown that YTHDC2-KO mice are infertile as their oocytes cannot develop beyond the zygotene stage of meiotic prophase I [65,98,99]. Another m6A reader that was found to be important for mouse oocyte and spermatogonial development, is the nuclear factor YTHDC1. Loss of YTHDC1 leads to extensive polyadenylation and altered 3′ UTR length, in addition to defective alternative splicing [102]. As a result, there are no secondary or antral follicles in the ovaries, indicating YTHDC1-deficient oocytes are blocked at the primary follicle stage. Indeed, YTHDC1 interacts with pre-mRNA 3′ end processing factors CPSF6, SRSF3 and SRSF7 [102].
5. Discussion
To conclude, many discoveries about m6A and its function in stem cells and germ cells were made in recent years. It is clear that m6A play a crucial role when orchestrated major changes in transcription are required, such as in embryonic development, differentiation of adult stem cells and gametogenesis. Looking to the future, much more research is required in order to fully understand the mechanism underlying these processes.
Two main related questions have not been fully answered. First is to what extent the modification is dynamic—on one hand there are multiple proteins which write, read and erase the modification, which suggests the modification is dynamically regulated in different cellular contexts. On the other hand, so far there is no work proving that the modification stoichiometry is changing between cellular states. As mentioned above, one of the reasons for the lack of such work, is that m6A-seq and meRIP-seq, based on antibodies, are limited in their ability to quantify methylation levels. A recent work that is using a cleaner method for m6A mapping in single-nucleotide resolution [23], shows that the methylation is in large not dynamic. However, even in the scenario where modification is not dynamically regulated, it is clear that its role for proper differentiation in vitro and in vivo is indispensable. The second related question is how specificity is achieved during writing, reading and erasing m6A. It is likely that the different m6A proteins interact with other proteins which regulate their activity, and may even direct them to their targets, thus generating specificity. However, more research is required to find those mechanisms.
More specifically, the interplay between YTHDF1, YTHDF2 and YTHDF3 should be studied. The three proteins are highly similar, and share many of their targets [64]. Is there a redundancy between the function of the readers? How do these m6A readers achieve specificity of their targets during gametogenesis and during stem cell differentiation? Are they expressed in different stages or in different levels? Furthermore, a comprehensive understating of other m6A related proteins in oogenesis and spermatogenesis is still missing.
It is also intriguing to investigate the role of m6A in human gametogenesis models. Loss of function of YTHDF2 was not detected in human population (pLI score = 1), however, it is interesting to check whether some cases of human infertility can be associated with aberrant m6A regulation.
Author Contributions
L.L. and N.N. wrote the review; with edits from J.H.H. All authors have read and agreed to the published version of the manuscript.
Funding
J.H.H. and N.N. are funded by Nella and Leon Benoziyo Center for Neurological Diseases; David and Fela Shapell Family Center for Genetic Disorders Research; Kekst Family Institute for Medical Genetics; Helen and Martin Kimmel Institute for Stem Cell Research; Flight Attendant Medical Research Council (FAMRI); Dr. Barry Sherman Center for Medicinal Chemistry; Pascal and Ilana Mantoux; Dr. Beth Rom-Rymer Stem Cell Research Fund; Edmond de Rothschild Foundations; Zantker Charitable Foundation; Estate of Zvia Zeroni; European Research Council (ERC-CoG); Israel Science Foundation (ISF); Minerva; Israel Cancer Research Fund (ICRF) and BSF.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Saletore Y., Meyer K., Korlach J., Vilfan I.D., Jaffrey S., Mason C.E. The birth of the Epitranscriptome: Deciphering the function of RNA modifications. Genome Biol. 2012;13:175. doi: 10.1186/gb-2012-13-10-175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Boccaletto P., MacHnicka M.A., Purta E., Pitkowski P., Baginski B., Wirecki T.K., De Crécy-Lagard V., Ross R., Limbach P.A., Kotter A., et al. MODOMICS: A database of RNA modification pathways. 2017 update. Nucleic Acids Res. 2018;46:D303–D307. doi: 10.1093/nar/gkx1030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Desrosiers R.C., Friderici K.H., Rottman F.M. Characterization of Novikoff Hepatoma Mrna Methylation and Heterogeneity in the Methylated 5′ Terminus. Biochemistry. 1975;14:4367–4374. doi: 10.1021/bi00691a004. [DOI] [PubMed] [Google Scholar]
- 4.Cohn W.E., Volkin E. Nucleoside-5′-phosphates from ribonucleic acid. Nature. 1951;167:483–484. doi: 10.1038/167483a0. [DOI] [Google Scholar]
- 5.DAVIS F.F., ALLEN F.W. Ribonucleic acids from yeast which contain a fifth nucleotide. J. Biol. Chem. 1957;227:907–915. [PubMed] [Google Scholar]
- 6.Perry R.P., Kelley D.E., Friderici K., Rottman F. The methylated constituents of L cell messenger RNA: Evidence for an unusual cluster at the 5′ terminus. Cell. 1975;4:387–394. doi: 10.1016/0092-8674(75)90159-2. [DOI] [PubMed] [Google Scholar]
- 7.Desrosiers R., Friderici K., Rottman F. Identification of methylated nucleosides in messenger RNA from Novikoff hepatoma cells. Proc. Natl. Acad. Sci. USA. 1974;71:3971–3975. doi: 10.1073/pnas.71.10.3971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Perry R.P., Kelley D.E. Existence of methylated messenger RNA in mouse L cells. Cell. 1974;1:37–42. doi: 10.1016/0092-8674(74)90153-6. [DOI] [Google Scholar]
- 9.Lavi S., Shatkin A.J. Methylated simian virus 40 specific RNA from nuclei and cytoplasm of infected BSC 1 cells. Proc. Natl. Acad. Sci. USA. 1975;72:2012–2016. doi: 10.1073/pnas.72.6.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wei C.M., Gershowitz A., Moss B. 5′-Terminal and Internal Methylated Nucleotide Sequences in Hela Cell mRNA. Biochemistry. 1976;15:397–401. doi: 10.1021/bi00647a024. [DOI] [PubMed] [Google Scholar]
- 11.Wei C.M., Gershowitz A., Moss B. Methylated nucleotides block 5′ terminus of HeLa cell messenger RNA. Cell. 1975;4:379–386. doi: 10.1016/0092-8674(75)90158-0. [DOI] [PubMed] [Google Scholar]
- 12.Beemon K., Keith J. Localization of N6-methyladenosine in the Rous sarcoma virus genome. J. Mol. Biol. 1977;113:165–179. doi: 10.1016/0022-2836(77)90047-X. [DOI] [PubMed] [Google Scholar]
- 13.Krug R.M., Morgan M.A., Shatkin A.J. Influenza viral mRNA contains internal N6-methyladenosine and 5′-terminal 7-methylguanosine in cap structures. J. Virol. 1976;20:45–53. doi: 10.1128/JVI.20.1.45-53.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schibler U., Kelley D.E., Perry R.P. Comparison of methylated sequences in messenger RNA and heterogeneous nuclear RNA from mouse L cells. J. Mol. Biol. 1977;115:695–714. doi: 10.1016/0022-2836(77)90110-3. [DOI] [PubMed] [Google Scholar]
- 15.Canaani D., Kahana C., Lavi S., Groner Y. Identification and mapping of N6-methyladenosine containing sequences in simian virus 40 RNA. Nucleic Acids Res. 1979;6:2879–2899. doi: 10.1093/nar/6.8.2879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dominissini D., Moshitch-Moshkovitz S., Schwartz S., Salmon-Divon M., Ungar L., Osenberg S., Cesarkas K., Jacob-Hirsch J., Amariglio N., Kupiec M., et al. Topology of the human and mouse m6A RNA methylomes revealed by m6A-seq. Nature. 2012;485:201–206. doi: 10.1038/nature11112. [DOI] [PubMed] [Google Scholar]
- 17.Meyer K.D., Saletore Y., Zumbo P., Elemento O., Mason C.E., Jaffrey S.R. Comprehensive analysis of mRNA methylation reveals enrichment in 3′ UTRs and near stop codons. Cell. 2012;149:1635–1646. doi: 10.1016/j.cell.2012.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Schwartz S., Agarwala S.D., Mumbach M.R., Jovanovic M., Mertins P., Shishkin A., Tabach Y., Mikkelsen T.S., Satija R., Ruvkun G., et al. High-Resolution mapping reveals a conserved, widespread, dynamic mRNA methylation program in yeast meiosis. Cell. 2013;155:1409–1421. doi: 10.1016/j.cell.2013.10.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Batista P.J., Molinie B., Wang J., Qu K., Zhang J., Li L., Bouley D.M., Lujan E., Haddad B., Daneshvar K., et al. M6A RNA modification controls cell fate transition in mammalian embryonic stem cells. Cell Stem Cell. 2014;15:707–719. doi: 10.1016/j.stem.2014.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Liu J., Yue Y., Han D., Wang X., Fu Y., Zhang L., Jia G., Yu M., Lu Z., Deng X., et al. A METTL3-METTL14 complex mediates mammalian nuclear RNA N6-adenosine methylation. Nat. Chem. Biol. 2014;10:93–95. doi: 10.1038/nchembio.1432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jia G., Fu Y., Zhao X., Dai Q., Zheng G., Yang Y., Yi C., Lindahl T., Pan T., Yang Y.G., et al. N6-Methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat. Chem. Biol. 2011;7:885–887. doi: 10.1038/nchembio.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mauer J., Luo X., Blanjoie A., Jiao X., Grozhik A.V., Patil D.P., Linder B., Pickering B.F., Vasseur J.J., Chen Q., et al. Reversible methylation of m6 Am in the 5′ cap controls mRNA stability. Nature. 2017;541:371–375. doi: 10.1038/nature21022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Garcia-Campos M.A., Edelheit S., Toth U., Safra M., Shachar R., Viukov S., Winkler R., Nir R., Lasman L., Brandis A., et al. Deciphering the “m6A Code” via Antibody-Independent Quantitative Profiling. Cell. 2019;178:731–747. doi: 10.1016/j.cell.2019.06.013. [DOI] [PubMed] [Google Scholar]
- 24.Wang Y., Li Y., Toth J.I., Petroski M.D., Zhang Z., Zhao J.C. N6 -methyladenosine modification destabilizes developmental regulators in embryonic stem cells. Nat. Cell Biol. 2014;16:191–198. doi: 10.1038/ncb2902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schwartz S., Mumbach M.R., Jovanovic M., Wang T., Maciag K., Bushkin G.G., Mertins P., Ter-Ovanesyan D., Habib N., Cacchiarelli D., et al. Perturbation of m6A writers reveals two distinct classes of mRNA methylation at internal and 5′ sites. Cell Rep. 2014;8:284–296. doi: 10.1016/j.celrep.2014.05.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lence T., Akhtar J., Bayer M., Schmid K., Spindler L., Ho C.H., Kreim N., Andrade-Navarro M.A., Poeck B., Helm M., et al. M6A modulates neuronal functions and sex determination in Drosophila. Nature. 2016;540:242–247. doi: 10.1038/nature20568. [DOI] [PubMed] [Google Scholar]
- 27.Meyer K.D., Patil D.P., Zhou J., Zinoviev A., Skabkin M.A., Elemento O., Pestova T.V., Qian S.B., Jaffrey S.R. 5′ UTR m6A Promotes Cap-Independent Translation. Cell. 2015;163:999–1010. doi: 10.1016/j.cell.2015.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shi H., Wang X., Lu Z., Zhao B.S., Ma H., Hsu P.J., Liu C., He C. YTHDF3 facilitates translation and decay of N 6-methyladenosine-modified RNA. Cell Res. 2017;27:315–328. doi: 10.1038/cr.2017.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhou J., Wan J., Gao X., Zhang X., Jaffrey S.R., Qian S.B. Dynamic m6 A mRNA methylation directs translational control of heat shock response. Nature. 2015;526:591–594. doi: 10.1038/nature15377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schwartz S., Bernstein D.A., Mumbach M.R., Jovanovic M., Herbst R.H., León-Ricardo B.X., Engreitz J.M., Guttman M., Satija R., Lander E.S., et al. Transcriptome-wide mapping reveals widespread dynamic-regulated pseudouridylation of ncRNA and mRNA. Cell. 2014;159:148–162. doi: 10.1016/j.cell.2014.08.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Geula S., Moshitch-Moshkovitz S., Dominissini D., Mansour A.A., Kol N., Salmon-Divon M., Hershkovitz V., Peer E., Mor N., Manor Y.S., et al. m6A mRNA methylation facilitates resolution of naïve pluripotency toward differentiation. Science. 2015;347:1002–1006. doi: 10.1126/science.1261417. [DOI] [PubMed] [Google Scholar]
- 32.Fustin J.M., Doi M., Yamaguchi Y., Hida H., Nishimura S., Yoshida M., Isagawa T., Morioka M.S., Kakeya H., Manabe I., et al. XRNA-methylation-dependent RNA processing controls the speed of the circadian clock. Cell. 2013;155:793–806. doi: 10.1016/j.cell.2013.10.026. [DOI] [PubMed] [Google Scholar]
- 33.Zhang C., Samanta D., Lu H., Bullen J.W., Zhang H., Chen I., He X., Semenza G.L. Hypoxia induces the breast cancer stem cell phenotype by HIF-dependent and ALKBH5-mediated m6A-demethylation of NANOG mRNA. Proc. Natl. Acad. Sci. USA. 2016;113:E2047–E2056. doi: 10.1073/pnas.1602883113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lin S., Choe J., Du P., Triboulet R., Gregory R.I. The m 6 A Methyltransferase METTL3 Promotes Translation in Human Cancer Cells. Mol. Cell. 2016;62:335–345. doi: 10.1016/j.molcel.2016.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zaccara S., Ries R.J., Jaffrey S.R. Reading, writing and erasing mRNA methylation. Nat. Rev. Mol. Cell Biol. 2019;20:608–624. doi: 10.1038/s41580-019-0168-5. [DOI] [PubMed] [Google Scholar]
- 36.Shi H., Wei J., He C. Where, When, and How: Context-Dependent Functions of RNA Methylation Writers, Readers, and Erasers. Mol. Cell. 2019;74:640–650. doi: 10.1016/j.molcel.2019.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lee Y., Choe J., Park O.H., Kim Y.K. Molecular Mechanisms Driving mRNA Degradation by m6A Modification. Trends Genet. 2020;36:177–188. doi: 10.1016/j.tig.2019.12.007. [DOI] [PubMed] [Google Scholar]
- 38.Bokar J.A., Rath-Shambaugh M.E., Ludwiczak R., Narayan P., Rottman F. Characterization and partial purification of mRNA N6-adenosine methyltransferase from HeLa cell nuclei. Internal mRNA methylation requires a multisubunit complex. J. Biol. Chem. 1994;269:17697–17704. [PubMed] [Google Scholar]
- 39.Tuck M.T. The formation of internal 6-methyladenine residues in eucaryotic messenger rna. Int. J. Biochem. 1992;24:379–386. doi: 10.1016/0020-711X(92)90028-Y. [DOI] [PubMed] [Google Scholar]
- 40.Widagdo J., Anggono V. The m6A-epitranscriptomic signature in neurobiology: From neurodevelopment to brain plasticity. J. Neurochem. 2018;147:137–152. doi: 10.1111/jnc.14481. [DOI] [PubMed] [Google Scholar]
- 41.Dorn L.E., Lasman L., Chen J., Xu X., Hund T.J., Medvedovic M., Hanna J.H., Van Berlo J.H., Accornero F. The N-Methyladenosine mRNA Methylase METTL3 Controls Cardiac Homeostasis and Hypertrophy. Circulation. 2019;139:533–545. doi: 10.1161/CIRCULATIONAHA.118.036146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Berulava T., Buchholz E., Elerdashvili V., Pena T., Islam M.R., Lbik D., Mohamed B.A., Renner A., von Lewinski D., Sacherer M., et al. Changes in m6A RNA methylation contribute to heart failure progression by modulating translation. Eur. J. Heart Fail. 2020;22:24–66. doi: 10.1002/ejhf.1672. [DOI] [PubMed] [Google Scholar]
- 43.Wu Y., Xie L., Wang M., Xiong Q., Guo Y., Liang Y., Li J., Sheng R., Deng P., Wang Y., et al. Mettl3-mediated m 6 A RNA methylation regulates the fate of bone marrow mesenchymal stem cells and osteoporosis. Nat. Commun. 2018;9:4772. doi: 10.1038/s41467-018-06898-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lee H., Bao S., Qian Y., Geula S., Leslie J., Zhang C., Hanna J.H., Ding L. Stage-specific requirement for Mettl3-dependent m6A mRNA methylation during haematopoietic stem cell differentiation. Nat. Cell Biol. 2019;21:700–709. doi: 10.1038/s41556-019-0318-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Xia H., Zhong C., Wu X., Chen J., Tao B., Xia X., Shi M., Zhu Z., Trudeau V.L., Hu W. Mettl3 mutation disrupts gamete maturation and reduces fertility in zebrafish. Genetics. 2018;208:729–743. doi: 10.1534/genetics.117.300574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Xu K., Yang Y., Feng G.H., Sun B.F., Chen J.Q., Li Y.F., Chen Y.S., Zhang X.X., Wang C.X., Jiang L.Y., et al. Mettl3-mediated m 6 A regulates spermatogonial differentiation and meiosis initiation. Cell Res. 2017;27:1100–1114. doi: 10.1038/cr.2017.100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lan Q., Liu P.Y., Haase J., Bell J.L., Huttelmaier S., Liu T. The critical role of RNA M6A methylation in cancer. Cancer Res. 2019;79:1285–1292. doi: 10.1158/0008-5472.CAN-18-2965. [DOI] [PubMed] [Google Scholar]
- 48.Zhong S., Li H., Bodi Z., Button J., Vespa L., Herzog M., Fray R.G. MTA is an Arabidopsis messenger RNA adenosine methylase and interacts with a homolog of a sex-specific splicing factor. Plant Cell. 2008;20:1278–1288. doi: 10.1105/tpc.108.058883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Agarwala S.D., Blitzblau H.G., Hochwagen A., Fink G.R. RNA methylation by the MIS complex regulates a cell fate decision in yeast. PLoS Genet. 2012;8:E1002732. doi: 10.1371/journal.pgen.1002732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kobayashi M., Ohsugi M., Sasako T., Awazawa M., Umehara T., Iwane A., Kobayashi N., Okazaki Y., Kubota N., Suzuki R., et al. The RNA Methyltransferase Complex of WTAP, METTL3, and METTL14 Regulates Mitotic Clonal Expansion in Adipogenesis. Mol. Cell. Biol. 2018;38:E00116–E00118. doi: 10.1128/MCB.00116-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wang P., Doxtader K.A., Nam Y. Structural Basis for Cooperative Function of Mettl3 and Mettl14 Methyltransferases. Mol. Cell. 2016;63:306–317. doi: 10.1016/j.molcel.2016.05.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Wang X., Feng J., Xue Y., Guan Z., Zhang D., Liu Z., Gong Z., Wang Q., Huang J., Tang C., et al. Structural basis of N6-adenosine methylation by the METTL3-METTL14 complex. Nature. 2016;542:260. doi: 10.1038/nature21073. [DOI] [PubMed] [Google Scholar]
- 53.Śledź P., Jinek M. Structural insights into the molecular mechanism of the m6A writer complex. Elife. 2016;5:E18434. doi: 10.7554/eLife.18434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Patil D.P., Chen C.K., Pickering B.F., Chow A., Jackson C., Guttman M., Jaffrey S.R. M6 A RNA methylation promotes XIST-mediated transcriptional repression. Nature. 2016;537:369–373. doi: 10.1038/nature19342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Liu J., Yue Y., Liu J., Cui X., Cao J., Luo G., Zhang Z., Cheng T., Gao M., Shu X., et al. VIRMA mediates preferential m6A mRNA methylation in 3′UTR and near stop codon and associates with alternative polyadenylation. Cell Discov. 2018:4. doi: 10.1038/s41421-018-0019-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wen J., Lv R., Ma H., Shen H., He C., Wang J., Jiao F., Liu H., Yang P., Tan L., et al. Zc3h13 Regulates Nuclear RNA m6A Methylation and Mouse Embryonic Stem Cell Self-Renewal. Mol. Cell. 2018;69:1028–1038. doi: 10.1016/j.molcel.2018.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Warda A.S., Kretschmer J., Hackert P., Lenz C., Urlaub H., Höbartner C., Sloan K.E., Bohnsack M.T. Human METTL16 is a N 6 --methyladenosine (m 6 A) methyltransferase that targets pre--mRNAs and various non--coding RNAs. EMBO Rep. 2017;18:2004–2014. doi: 10.15252/embr.201744940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Pendleton K.E., Chen B., Liu K., Hunter O.V., Xie Y., Tu B.P., Conrad N.K. The U6 snRNA m6A Methyltransferase METTL16 Regulates SAM Synthetase Intron Retention. Cell. 2017;169:824–835. doi: 10.1016/j.cell.2017.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zheng G., Dahl J.A., Niu Y., Fedorcsak P., Huang C.M., Li C.J., Vågbø C.B., Shi Y., Wang W.L., Song S.H., et al. ALKBH5 Is a Mammalian RNA Demethylase that Impacts RNA Metabolism and Mouse Fertility. Mol. Cell. 2013;49:18–29. doi: 10.1016/j.molcel.2012.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wei J., Liu F., Lu Z., Fei Q., Ai Y., He P.C., Shi H., Cui X., Su R., Klungland A., et al. Differential m 6 A, m 6 A m, and m 1 A Demethylation Mediated by FTO in the Cell Nucleus and Cytoplasm. Mol. Cell. 2018;71:973–985. doi: 10.1016/j.molcel.2018.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhu T., Roundtree I.A., Wang P., Wang X., Wang L., Sun C., Tian Y., Li J., He C., Xu Y. Crystal structure of the YTH domain of YTHDF2 reveals mechanism for recognition of N6-methyladenosine. Cell Res. 2014;24:1493–1496. doi: 10.1038/cr.2014.152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Luo S., Tong L. Molecular basis for the recognition of methylated adenines in RNA by the eukaryotic YTH domain. Proc. Natl. Acad. Sci. USA. 2014;111:13834–13839. doi: 10.1073/pnas.1412742111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Theler D., Dominguez C., Blatter M., Boudet J., Allain F.H.T. Solution structure of the YTH domain in complex with N6-methyladenosine RNA: A reader of methylated RNA. Nucleic Acids Res. 2014;42:13911–13919. doi: 10.1093/nar/gku1116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Patil D.P., Pickering B.F., Jaffrey S.R. Reading m6A in the Transcriptome: m6A-Binding Proteins. Trends Cell Biol. 2018;28:113–127. doi: 10.1016/j.tcb.2017.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hsu P.J., Zhu Y., Ma H., Guo Y., Shi X., Liu Y., Qi M., Lu Z., Shi H., Wang J., et al. Ythdc2 is an N6 -methyladenosine binding protein that regulates mammalian spermatogenesis. Cell Res. 2017;27:1115–1127. doi: 10.1038/cr.2017.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Wang X., Lu Z., Gomez A., Hon G.C., Yue Y., Han D., Fu Y., Parisien M., Dai Q., Jia G., et al. N 6-methyladenosine-dependent regulation of messenger RNA stability. Nature. 2014;505:117–120. doi: 10.1038/nature12730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Du H., Zhao Y., He J., Zhang Y., Xi H., Liu M., Ma J., Wu L. YTHDF2 destabilizes m 6 A-containing RNA through direct recruitment of the CCR4-NOT deadenylase complex. Nat. Commun. 2016;7:12626. doi: 10.1038/ncomms12626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Wang X., Zhao B.S., Roundtree I.A., Lu Z., Han D., Ma H., Weng X., Chen K., Shi H., He C. N6-methyladenosine modulates messenger RNA translation efficiency. Cell. 2015;161:1388–1399. doi: 10.1016/j.cell.2015.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Shi H., Zhang X., Weng Y.L., Lu Z., Liu Y., Lu Z., Li J., Hao P., Zhang Y., Zhang F., et al. m6A facilitates hippocampus-dependent learning and memory through YTHDF1. Nature. 2018;563:249–253. doi: 10.1038/s41586-018-0666-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Xiao W., Adhikari S., Dahal U., Chen Y.S., Hao Y.J., Sun B.F., Sun H.Y., Li A., Ping X.L., Lai W.Y., et al. Nuclear m6A Reader YTHDC1 Regulates mRNA Splicing. Mol. Cell. 2016;61:507–519. doi: 10.1016/j.molcel.2016.01.012. [DOI] [PubMed] [Google Scholar]
- 71.Roundtree I.A., He C. Nuclear m6A Reader YTHDC1 Regulates mRNA Splicing. Trends Genet. 2016;32:320–321. doi: 10.1016/j.tig.2016.03.006. [DOI] [PubMed] [Google Scholar]
- 72.Shima H., Matsumoto M., Ishigami Y., Ebina M., Muto A., Sato Y., Kumagai S., Ochiai K., Suzuki T., Igarashi K. S-Adenosylmethionine Synthesis Is Regulated by Selective N6-Adenosine Methylation and mRNA Degradation Involving METTL16 and YTHDC1. Cell Rep. 2017;21:3354–3363. doi: 10.1016/j.celrep.2017.11.092. [DOI] [PubMed] [Google Scholar]
- 73.Alarcón C.R., Goodarzi H., Lee H., Liu X., Tavazoie S., Tavazoie S.F. HNRNPA2B1 Is a Mediator of m6A-Dependent Nuclear RNA Processing Events. Cell. 2015;162:1299–1308. doi: 10.1016/j.cell.2015.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Liu N., Dai Q., Zheng G., He C., Parisien M., Pan T. N6 -methyladenosine-dependent RNA structural switches regulate RNA-protein interactions. Nature. 2015;518:560–564. doi: 10.1038/nature14234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Wu B., Su S., Patil D.P., Liu H., Gan J., Jaffrey S.R., Ma J. Molecular basis for the specific and multivariant recognitions of RNA substrates by human hnRNP A2/B1. Nat. Commun. 2018;9:420. doi: 10.1038/s41467-017-02770-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Huang H., Weng H., Sun W., Qin X., Shi H., Wu H., Zhao B.S., Mesquita A., Liu C., Yuan C.L., et al. Recognition of RNA N 6 -methyladenosine by IGF2BP proteins enhances mRNA stability and translation. Nat. Cell Biol. 2018;20:285–295. doi: 10.1038/s41556-018-0045-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Edupuganti R.R., Geiger S., Lindeboom R.G.H., Shi H., Hsu P.J., Lu Z., Wang S.Y., Baltissen M.P.A., Jansen P.W.T.C., Rossa M., et al. N6-methyladenosine (m6A) recruits and repels proteins to regulate mRNA homeostasis. Nat. Struct. Mol. Biol. 2017;24:870–878. doi: 10.1038/nsmb.3462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Liu N., Zhou K.I., Parisien M., Dai Q., Diatchenko L., Pan T. N6-methyladenosine alters RNA structure to regulate binding of a low-complexity protein. Nucleic Acids Res. 2017;45:6051–6063. doi: 10.1093/nar/gkx141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zhang F., Kang Y., Wang M., Li Y., Xu T., Yang W., Song H., Wu H., Shu Q., Jin P. Fragile X mental retardation protein modulates the stability of its m6A-marked messenger RNA targets. Hum. Mol. Genet. 2018;27:3936–3950. doi: 10.1093/hmg/ddy292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Weinberger L., Ayyash M., Novershtern N., Hanna J.H. Dynamic stem cell states: Naive to primed pluripotency in rodents and humans. Nat. Rev. Mol. Cell Biol. 2016;17:155–169. doi: 10.1038/nrm.2015.28. [DOI] [PubMed] [Google Scholar]
- 81.Jaenisch R., Young R. Stem Cells, the Molecular Circuitry of Pluripotency and Nuclear Reprogramming. Cell. 2008;132:567–582. doi: 10.1016/j.cell.2008.01.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Meng T.G., Lu X., Guo L., Hou G.M., Ma X.S., Li Q.N., Huang L., Fan L.H., Zhao Z.H., Ou X.H., et al. Mettl14 is required for mouse postimplantation development by facilitating epiblast maturation. FASEB J. 2019;33:1179–1187. doi: 10.1096/fj.201800719R. [DOI] [PubMed] [Google Scholar]
- 83.Chen T., Hao Y.J., Zhang Y., Li M.M., Wang M., Han W., Wu Y., Lv Y., Hao J., Wang L., et al. M6A RNA methylation is regulated by microRNAs and promotes reprogramming to pluripotency. Cell Stem Cell. 2015;16:289–301. doi: 10.1016/j.stem.2015.01.016. [DOI] [PubMed] [Google Scholar]
- 84.Yang D., Qiao J., Wang G., Lan Y., Li G., Guo X., Xi J., Ye D., Zhu S., Chen W., et al. N6-Methyladenosine modification of lincRNA 1281 is critically required for mESC differentiation potential. Nucleic Acids Res. 2018;46:3906–3920. doi: 10.1093/nar/gky130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bertero A., Brown S., Madrigal P., Osnato A., Ortmann D., Yiangou L., Kadiwala J., Hubner N.C., De Los Mozos I.R., Sadée C., et al. The SMAD2/3 interactome reveals that TGFβ controls m 6 A mRNA methylation in pluripotency. Nature. 2018;555:256–259. doi: 10.1038/nature25784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Li M., Zhao X., Wang W., Shi H., Pan Q., Lu Z., Perez S.P., Suganthan R., He C., Bjørås M., et al. Ythdf2-mediated m6A mRNA clearance modulates neural development in mice. Genome Biol. 2018;19:69. doi: 10.1186/s13059-018-1436-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Chen J., Zhang Y.C., Huang C., Shen H., Sun B., Cheng X., Zhang Y.J., Yang Y.G., Shu Q., Yang Y., et al. m6A Regulates Neurogenesis and Neuronal Development by Modulating Histone Methyltransferase Ezh2. Genom. Proteom. Bioinform. 2019;17:154–168. doi: 10.1016/j.gpb.2018.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Edens B.M., Vissers C., Su J., Arumugam S., Xu Z., Shi H., Miller N., Rojas Ringeling F., Ming G.L., He C., et al. FMRP Modulates Neural Differentiation through m6A-Dependent mRNA Nuclear Export. Cell Rep. 2019;28:845–854. doi: 10.1016/j.celrep.2019.06.072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Li Z., Qian P., Shao W., Shi H., He X.C., Gogol M., Yu Z., Wang Y., Qi M., Zhu Y., et al. Suppression of m6A reader Ythdf2 promotes hematopoietic stem cell expansion. Cell Res. 2018;28:904–917. doi: 10.1038/s41422-018-0072-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Cheng Y., Luo H., Izzo F., Pickering B.F., Nguyen D., Myers R., Schurer A., Gourkanti S., Brüning J.C., Vu L.P., et al. m6A RNA Methylation Maintains Hematopoietic Stem Cell Identity and Symmetric Commitment. Cell Rep. 2019;28:1703–1716. doi: 10.1016/j.celrep.2019.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Heck A.M., Wilusz C.J. Small changes, big implications: The impact of m6A RNA methylation on gene expression in pluripotency and development. Biochim. Biophys. Acta-Gene Regul. Mech. 2019;1862:194402. doi: 10.1016/j.bbagrm.2019.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Bushkin G.G., Pincus D., Morgan J.T., Richardson K., Lewis C., Chan S.H., Bartel D.P., Fink G.R. m6A modification of a 3′ UTR site reduces RME1 mRNA levels to promote meiosis. Nat. Commun. 2019;10:3414. doi: 10.1038/s41467-019-11232-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Bodi Z., Bottley A., Archer N., May S.T., Fray R.G. Yeast m6A methylated mRNAs are enriched on translating ribosomes during meiosis, and under rapamycin treatment. PLoS ONE. 2015;10:E0132090. doi: 10.1371/journal.pone.0132090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Tang C., Klukovich R., Peng H., Wang Z., Yu T., Zhang Y., Zheng H., Klungland A., Yan W. ALKBH5-dependent m6A demethylation controls splicing and stability of long 3′-UTR mRNAs in male germ cells. Proc. Natl. Acad. Sci. USA. 2017;115:E325–E333. doi: 10.1073/pnas.1717794115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Huang T., Guo J., Lv Y., Zheng Y., Feng T., Gao Q., Zeng W. Meclofenamic acid represses spermatogonial proliferation through modulating m6A RNA modification. J. Anim. Sci. Biotechnol. 2019;11:10:63. doi: 10.1186/s40104-019-0361-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Lin Z., Hsu P.J., Xing X., Fang J., Lu Z., Zou Q., Zhang K.J., Zhang X., Zhou Y., Zhang T., et al. Mettl3-/Mettl14-mediated mRNA N 6-methyladenosine modulates murine spermatogenesis. Cell Res. 2017;27:1216–1230. doi: 10.1038/cr.2017.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Castrillon D.H., Quade B.J., Wang T.Y., Quigley C., Crum C.P. The human VASA gene is specifically expressed in the germ cell lineage. Proc. Natl. Acad. Sci. USA. 2000;97:9585–9590. doi: 10.1073/pnas.160274797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Bailey A.S., Batista P.J., Gold R.S., Grace Chen Y., de Rooij D.G., Chang H.Y., Fuller M.T. The conserved RNA helicase YTHDC2 regulates the transition from proliferation to differentiation in the germline. Elife. 2017;6:E26116. doi: 10.7554/eLife.26116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wojtas M.N., Pandey R.R., Mendel M., Homolka D., Sachidanandam R., Pillai R.S. Regulation of m6A Transcripts by the 3ʹ→5ʹ RNA Helicase YTHDC2 Is Essential for a Successful Meiotic Program in the Mammalian Germline. Mol. Cell. 2017;68:374–387. doi: 10.1016/j.molcel.2017.09.021. [DOI] [PubMed] [Google Scholar]
- 100.Jain D., Puno M.R., Meydan C., Lailler N., Mason C.E., Lima C.D., Anderson K.V., Keeney S. Ketu mutant mice uncover an essential meiotic function for the ancient RNA helicase YTHDC2. Elife. 2018;7:E30919. doi: 10.7554/eLife.30919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Abby E., Tourpin S., Ribeiro J., Daniel K., Messiaen S., Moison D., Guerquin J., Gaillard J.C., Armengaud J., Langa F., et al. Implementation of meiosis prophase i programme requires a conserved retinoid-independent stabilizer of meiotic transcripts. Nat. Commun. 2016;8:10324. doi: 10.1038/ncomms10324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kasowitz S.D., Ma J., Anderson S.J., Leu N.A., Xu Y., Gregory B.D., Schultz R.M., Wang P.J. Nuclear m6A reader YTHDC1 regulates alternative polyadenylation and splicing during mouse oocyte development. PLoS Genet. 2018;14:E1007412. doi: 10.1371/journal.pgen.1007412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ivanova I., Much C., Di Giacomo M., Azzi C., Morgan M., Moreira P.N., Monahan J., Carrieri C., Enright A.J., O’Carroll D. The RNA m6A Reader YTHDF2 Is Essential for the Post-transcriptional Regulation of the Maternal Transcriptome and Oocyte Competence. Mol. Cell. 2017;67:1059–1067. doi: 10.1016/j.molcel.2017.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Huang T., Liu Z., Zheng Y., Feng T., Gao Q., Zeng W. YTHDF2 promotes spermagonial adhesion through modulating MMPs decay via m 6 A / mRNA pathway. Cell Death Dis. 2020;11:37. doi: 10.1038/s41419-020-2235-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Eppig J.J., Schroeder A.C. Capacity of Mouse Oocytes from Preantral Follicles to Undergo Embryogenesis and Development to Live Young after Growth, Maturation, and Fertilization in Vitro1. Biol. Reprod. 1989;41:268–276. doi: 10.1095/biolreprod41.2.268. [DOI] [PubMed] [Google Scholar]
- 106.Bachvarova R., De Leon V., Johnson A., Kaplan G., Paynton B.V. Changes in total RNA, polyadenylated RNA, and actin mRNA during meiotic maturation of mouse oocytes. Dev. Biol. 1985;108:325–331. doi: 10.1016/0012-1606(85)90036-3. [DOI] [PubMed] [Google Scholar]
- 107.Paynton B.V., Rempel R., Bachvarova R. Changes in state of adenylation and time course of degradation of maternal mRNAs during oocyte maturation and early embryonic development in the mouse. Dev. Biol. 1988;129:304–314. doi: 10.1016/0012-1606(88)90377-6. [DOI] [PubMed] [Google Scholar]
- 108.Tadros W., Lipshitz H.D. The maternal-to-zygotic transition: A play in two acts. Development. 2009;136:3033–3042. doi: 10.1242/dev.033183. [DOI] [PubMed] [Google Scholar]
- 109.Zhao B.S., Wang X., Beadell A.V., Lu Z., Shi H., Kuuspalu A., Ho R.K., He C. M6 A-dependent maternal mRNA clearance facilitates zebrafish maternal-to-zygotic transition. Nature. 2017;542:475–478. doi: 10.1038/nature21355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sui X., Hu Y., Ren C., Cao Q., Zhou S., Cao Y., Li M., Shu W., Huo R. METTL3-mediated m 6 A is required for murine oocyte maturation and maternal-to-zygotic transition. Cell Cycle. 2020;19:391–404. doi: 10.1080/15384101.2019.1711324. [DOI] [PMC free article] [PubMed] [Google Scholar]