Abstract
Objective
Asthma is a common chronic disease that imposes a substantial burden on individuals and society. However, the natural history of childhood asthma in a large population remained to be studied. This study aimed to describe the natural course of childhood asthma and examine the association between early life factors and childhood asthma.
Design
A population-based cohort study.
Setting
This study was based on the national registry data in Denmark.
Participants
All liveborn singletons in Denmark during 1995–1997 were identified and followed them till the end of 2009. Finally, 193 673 children were eligible for our study.
Exposures
The following characteristics were examined as potential early life factors associated with childhood asthma, including parity, maternal asthma history, maternal smoking during pregnancy, maternal social status, delivery method and gender.
Main outcomes and measures
Asthma cases were identified on the basis of hospitalisation for asthma and prescriptions for antiasthmatic medications. Asthma remission was defined as no hospitalisation or prescription recorded for 2 years. Cox proportional hazards’ regression and logistic regression were used to evaluate the association between early life factors and the occurrence and remission of childhood asthma.
Results
The cumulative occurrence rate of asthma in children aged 3–14 years was 13.3% and the remission rate was 44.1%. The occurrence rate decreased with age. Being female had a lower risk of asthma (HR: 0.72, 95% CI 0.70 to 0.74) and higher remission rate of asthma (HR: 1.18, 1.13 to 1.22), while maternal asthma was associated with a higher risk of asthma (HR: 2.15, 2.04 to 2.26) and decreased remission rate of asthma (HR: 0.79, 0.73 to 0.85). These patterns remained the same for early onset asthma.
Conclusions
Female gender had a lower risk of asthma and a better chance of remission, while maternal asthma history had an opposite effect. The early life factors may influence the natural course of childhood asthma.
Keywords: epidemiology, paediatrics, public health
Strengths and limitations of this study.
The definition of asthma was based on the registry information of the pharmacy and hospital, which was likely to be more objective.
The present study was a longitudinal study based on the national registry databases. Therefore, selection bias would be minimised.
However, the follow-up period was not long enough to observe asthma persistent into adulthood.
Background
Asthma, characterised by variable respiratory symptoms and airflow limitation, is one of the most common chronic, non-communicable diseases in children and adults.1 The prevalence of asthma in adults varies from 0.2% to 21.2% in different countries, while the prevalence of asthma in children ranges from 2.8% to 37.6%.2 The wide range of prevalence may be in part attributable to the dynamic nature of asthma, particularly in children, and the age range of the underlying asthmatic population.
Asthmatic status is not constant across the lifecourse of the patient, and patients can experience intermittent asthma attacks.3 A Swedish prospective study recruited 3430 asthmatic children aged 7 to 8 years. After a follow-up for 11 years, 21% were in remission, 38% had periodic asthma and 41% persistent asthma at 19 years of age.4 In a birth cohort conducted among children who had reported wheezing in New Zealand, 27.4% had remission at 26 years old.5 In the Tasmanian Longitudinal Health Study, among children who reported asthma, 65% remitted after 40 years.6 While risk factors for childhood asthma are well studied,7–10 few studies have examined factors related to asthma remission.
The present study aims to examine the natural history of childhood asthma in a large population-based retrospective cohort and the association between early life risk factors and the occurrence and remission of childhood asthma in the Danish population.
Methods
Study population
Our population-based cohort study used data from the Danish Medical Birth Registry, Danish National Patient Registry and Danish National Prescription Database, linked by the unique personal identification number (assigned by the Danish Civil Registration System to all people with permanent residency in Denmark). Using the Danish Medical Birth Registry,11 we identified all liveborn singletons in Denmark during 1995–1997 (N=1 97 697). We excluded 4024 children who died or emigrated before 3 years of age. Finally, 193 673 children were eligible for our study.
The information on maternal age, sex of the child, date of birth, gestational days, birth weight, parity, maternal smoking during pregnancy, mode of delivery and maternal personal identification number was obtained from the Danish Medical Birth Registry. Information on hospital contact, including hospital admission, emergency room and outpatient treatments in which asthma was the primary diagnosis, was obtained from the Danish National Patient Registry. Information on prescription of antiasthmatic medications was from the National Prescription Registry.
Covariates
Early life factors that are potentially associated with asthma were selected a priori based on the previous literature.12–14 They included maternal age at delivery, parity (first, second and higher), maternal socioeconomic status at delivery (not in labour market, unskilled workers, skilled workers and white-collar workers, top-level status), maternal smoking status during pregnancy (yes, no), maternal history of asthma (yes, no), sex of the child (boy, girl), gestational weeks at delivery, maternal residence at delivery (capital or capital suburb, provincial city or town or rural areas), birth weight (an ordinal variable with every 500 g as an interval), delivery method (caesarean section (CS), coded as O82 and O842 in the 10th International Classification of Diseases, ICD-10). A directed acyclic graph is shown in online supplemental figure S1 to illustrate the association between early life determinants and childhood asthma.
bmjopen-2020-045728supp001.pdf (218.7KB, pdf)
Asthma definition
Wheezing is the core symptom for asthma. Diagnosing asthma in children younger than 3 years is difficult because wheezing has many causes in that age group.15 Thus, asthma was diagnosed only at 3 years of age or older. We identified childhood asthma from the Danish national registries in the 12-year period 1998–2009. Asthma cases were identified on the basis of at least one hospitalisation for asthma or two prescriptions for antiasthmatic medications, less than 12 months apart. Hospitalisation for asthma was identified based on the following the ICD-10 codes: J45, J46. The anatomical therapeutical chemical codes for inhaled antiasthmatic drugs were inhaled ß2-agonists (R03AC02-04, R03AC12 and R03AC13), inhaled glucocorticoids (ICS; R03BA01, R03BA02 and R03BA05), fixed-dose combination of inhaled ß2-agonists and glucocorticoids (R03AK06 and R03AK07) and leucotriene receptor antagonists (R03DC03). Two or more medications prescribed on the same day were considered to represent one prescription. The first diagnosis of asthma was defined as the date of first hospitalisation, outpatient contact or first antiasthmatic drugs redeemed in the registers in children aged 3 years or older, whichever came first. Children who did not have documented asthma based on our definition were classified as ‘non-asthmatic’.
Asthma remission was defined as no hospitalisation or prescription recorded in the following 2 years, after the last hospitalisation or prescription of antiasthmatic drugs. The date of remission was defined as 2 years after the last asthma hospitalisation or antiasthmatic drug prescription.16 17 Finally, early onset asthma was defined as the first onset of asthma before 10 years old. Early remission asthma was defined as asthma remised before 12 years old.
We constructed two retrospective cohorts: asthma-onset cohort and remission cohort. The former consisted of all singleton births in 1995–1997. The age-specific and gender-specific occurrence rate of asthma was calculated as the number of newly diagnosed cases in an age-specific and gender-specific group divided by the number of children in that group. The cumulative occurrence rate of asthma was the total number of asthma cases ever diagnosed during the 12-year follow-up period to be divided by the number of children born in these 3 years. The remission cohort was composed of those who were ever been diagnosed as asthma. The remission rate was calculated as the number of children who met the criteria of asthma remission in an age-specific and gender-specific group divided by the number of children still with asthma at the beginning of that age (ie, population-at-risk of remission).
Statistical analysis
Cox proportional hazard regression model was used to examine factors that were associated with asthma onset and remission. HR with a 95% CI was estimated. In addition, logistic regression models were used to examine the association between early life factors and early onset and early remission of childhood asthma. The following variables were included in the multivariate logistic regression model, such as maternal age at delivery, maternal parity, maternal socioeconomic status at delivery, maternal smoking status during pregnancy, maternal history of asthma, sex of the child, gestational weeks at delivery, maternal residence at delivery, birth weight and delivery method. The age of asthma onset was also included in the remission analyses. All the analyses were performed using the SAS software, V.9.4 (SAS Institute, Cary, North Carolina).
Results
From 1998 to 2009, 25 751 asthma cases were observed in this cohort, yielding the cumulative occurrence rate of 13.3% (95% CI 13.1% to 13.5%). Among them, 11 365 patients remised by the end of 2009, and the remission rate was 44.1% (95% CI 43.5% to 44.7%). A flowchart of subject selection in the present study is shown in online supplemental figure S2. Table 1 shows demographic characteristics of the study population. Compared with children without asthma, children with asthma had a higher proportion of men (58.9% vs 50.3%). The asthma group also had a higher proportion of children delivered by CS (9.2% vs 7.8%) and maternal history of asthma (7.0% vs 3.1%), compared with children in the non-asthma group.
Table 1.
Children’s and their mothers’ characteristics (%)
| Non-asthma (N=1 67 922) | Asthma (N=25 751) | Total (N=1 93 673) | |
| Maternal age at delivery, y | |||
| <25 | 15.4 | 16.4 | 15.5 |
| 25–29 | 36.3 | 37.5 | 36.5 |
| 30–34 | 34.4 | 33.0 | 34.2 |
| ≥35 | 13.9 | 13.1 | 13.8 |
| Parity | |||
| 1 | 42.7 | 45.2 | 43.0 |
| 2 | 37.6 | 37.1 | 37.5 |
| ≥3 | 19.7 | 17.7 | 19.4 |
| Maternal socio-economic status | |||
| Not in labour market | 22.0 | 22.3 | 22.0 |
| Unskilled workers | 4.7 | 4.1 | 4.6 |
| Skilled workers and white-collar workers | 30.5 | 31.0 | 30.5 |
| Top level status | 42.0 | 41.9 | 42.0 |
| Unknown | 0.9 | 0.8 | 0.9 |
| Maternal history of asthma | |||
| No | 96.9 | 93.0 | 96.3 |
| Yes | 3.1 | 7.0 | 3.7 |
| Sex of the child | |||
| Boy | 50.3 | 58.9 | 51.4 |
| Girl | 49.7 | 41.0 | 48.6 |
| Calendar year of birth | |||
| 1995 | 33.8 | 36.9 | 34.2 |
| 1996 | 32.9 | 33.1 | 32.9 |
| 1997 | 33.3 | 30.0 | 32.9 |
| Maternal smoking during pregnancy | |||
| No | 69.9 | 66.6 | 69.5 |
| Yes | 24.4 | 27.6 | 24.9 |
| Unknown | 5.6 | 5.8 | 5.6 |
| Gestational age | |||
| 22–28 weeks | 0.2 | 0.4 | 0.2 |
| 29–32 weeks | 0.6 | 0.9 | 0.6 |
| 33–36 weeks | 3.5 | 4.7 | 3.7 |
| 37–38 weeks | 14.2 | 15.6 | 14.4 |
| 39–41 weeks | 71.9 | 69.0 | 71.5 |
| 42–45 weeks | 9.0 | 8.8 | 9.0 |
| Unknown | 0.7 | 0.7 | 0.7 |
| Birth weight | |||
| <1000 g | 0.1 | 0.3 | 0.1 |
| 1000–1499 g | 0.3 | 0.6 | 0.4 |
| 1500–1999 g | 0.7 | 1.0 | 0.7 |
| 2000–2499 g | 2.1 | 3.0 | 2.2 |
| 2500–2999 g | 10.2 | 11.1 | 10.3 |
| 3000–3499 g | 31.4 | 30.8 | 31.3 |
| 3500–3999 g | 34.7 | 33.1 | 34.5 |
| 4000–4499 g | 15.8 | 15.3 | 15.7 |
| 4500–4999 g | 3.3 | 3.3 | 3.3 |
| ≥5000 g | 0.5 | 0.5 | 0.5 |
| Unknown | 1.0 | 1.0 | 1.0 |
| Mode of delivery | |||
| Delivered vaginally | 92.2 | 90.8 | 92.0 |
| Delivered by caesarean section | 7.8 | 9.2 | 8.0 |
| Residence | |||
| Capital or capital suburb | 27.1 | 25.9 | 26.9 |
| Provincial city or town | 12.8 | 13.5 | 12.9 |
| Rural areas | 59.2 | 59.8 | 59.3 |
| Unknown | 0.9 | 0.8 | 0.9 |
bmjopen-2020-045728supp002.pdf (50.8KB, pdf)
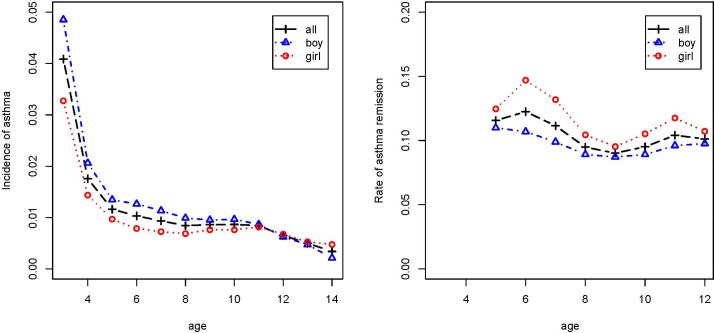
Figure 1 shows the age-specific occurrence of asthma by gender. It ranged from 0.34% to 4.09% and decreased with age. Before 11 years old, boys had a higher occurrence rate of asthma than girls. But after that, the trend seemed reversed. The total remission rate in the study was 44%, and the age-specific rate of asthma remission ranged from 9.02% to 12.3%. The remission rate declined slightly with age. Girls had a higher remission rate than boys.
Figure 1.
The incidence rate (left panel) and remission rate (right panel) of asthma by age. The x-axis is age (year), and y-axis is incidence rate (left panel) and remission rate (right panel). Black line for all children, the red line for girls and the blue line for boys.
Cox proportional hazard regression model assessed the effect of early life factors on childhood asthma (table 2). Having siblings, female gender and living in capital or capital suburb would reduce the risk of asthma, while maternal smoking during pregnancy, maternal asthma history and delivered by CS would increase the risk of asthma. This risk profile is similar to that of early onset asthma (table 3).
Table 2.
Prenatal factors and childhood asthma evaluated by Cox proportional hazards regression model (HR, 95% CI)
| Crude HR | 95% CI | Adjusted HR* | 95% CI | |
| Maternal age | 0.95 | 0.94 to 0.97 | 0.99 | 0.97 to 1.01 |
| Gestational age | 0.95 | 0.94 to 0.96 | 0.97 | 0.96 to 0.97 |
| Birth weight | 0.95 | 0.93 to 0.96 | 0.98 | 0.97 to 0.99 |
| Parity | ||||
| 1 | Ref | Ref | ||
| 2 | 0.93 | 0.91 to 0.96 | 0.94 | 0.92 to 0.97 |
| ≥3 | 0.86 | 0.83 to 0.89 | 0.86 | 0.83 to 0.90 |
| Maternal social status | ||||
| Not in labour market | 1.06 | 1.03 to 1.10 | 1.02 | 0.99 to 1.06 |
| Unskilled workers | 0.92 | 0.86 to 0.98 | 0.91 | 0.86 to 0.98 |
| Skilled workers and white-collar workers | 1.04 | 1.01 to 1.07 | 1.00 | 0.97 to 1.04 |
| Top level status | Ref | Ref | ||
| Residence | ||||
| Capital or capital suburb | 0.96 | 0.93 to 0.99 | 0.95 | 0.92 to 0.98 |
| Provincial city or town | 1.07 | 1.03 to 1.11 | 1.06 | 1.02 to 1.11 |
| Rural areas | Ref | Ref | ||
| Maternal smoking during pregnancy | 1.16 | 1.13 to 1.20 | 1.13 | 1.10 to 1.16 |
| Maternal history of asthma | 2.17 | 2.07 to 2.28 | 2.15 | 2.04 to 2.26 |
| Sex | ||||
| Male | Ref | Ref | ||
| Female | 0.72 | 0.70 to 0.74 | 0.72 | 0.70 to 0.74 |
| Mode of delivery | ||||
| Delivered vaginally | Ref | Ref | ||
| Delivered by caesarean section | 1.19 | 1.14 to 1.25 | 1.12 | 1.07 to 1.17 |
*Cox proportional hazards regression model (HR, 95% CI) adjusting for maternal age at delivery; maternal parity; maternal socio-economic status at delivery; maternal smoking status during pregnancy; maternal history of asthma; sex of the child; gestational weeks at delivery; maternal residence at delivery; birth weight and delivery method.
Table 3.
Prenatal factors and early onset (<10 years of age) of childhood asthma analysed by logistic regression model in asthma patients (OR, 95% CI)
| Crude OR | 95% CI | Adjusted OR* | 95% CI | |
| Maternal age | 0.96 | 0.93 to 1.00 | 1.00 | 0.96 to 1.04 |
| Gestational age | 0.96 | 0.95 to 0.98 | 0.98 | 0.96 to 1.00 |
| Birth weight | 0.97 | 0.94 to 0.99 | 0.98 | 0.95 to 1.02 |
| Parity | ||||
| 1 | Ref | Ref | ||
| 2 | 1.00 | 0.93 to 1.07 | 0.99 | 0.92 to 1.07 |
| ≥3 | 1.04 | 0.95 to 1.14 | 0.99 | 0.90 to 1.10 |
| Maternal social status | ||||
| Not in labour market | 1.32 | 1.22 to 1.44 | 1.28 | 1.17 to 1.40 |
| Unskilled workers | 1.38 | 1.17 to 1.64 | 1.35 | 1.14 to 1.60 |
| Skilled workers and white-collar workers | 1.25 | 1.16 to 1.35 | 1.21 | 1.12 to 1.31 |
| Top level status | Ref | Ref | ||
| Residence | ||||
| Capital or capital suburb | 0.78 | 0.72 to 0.83 | 0.79 | 0.73 to 0.85 |
| Provincial city or town | 1.00 | 0.90 to 1.10 | 0.99 | 0.90 to 1.10 |
| Rural areas | Ref | Ref | ||
| Maternal smoking during pregnancy | 1.10 | 1.03 to 1.18 | 1.04 | 0.97 to 1.12 |
| Maternal history of asthma | 1.32 | 1.16 to 1.61 | 1.32 | 1.15 to 1.51 |
| Sex | ||||
| Male | Ref | Ref | ||
| Female | 0.72 | 0.67 to 0.76 | 0.71 | 0.67 to 0.76 |
| Mode of delivery | ||||
| Delivered vaginally | Ref | Ref | ||
| Delivered by caesarean section | 1.25 | 1.11 to 1.40 | 1.19 | 1.06 to 1.34 |
*Logistic regression model (OR, 95% CI) adjusting for maternal age at delivery; maternal parity; maternal socio-economic status at delivery; maternal smoking status during pregnancy; maternal history of asthma; sex of the child; gestational weeks at delivery; maternal residence at delivery; birth weight and delivery method.
We examined the effects of early life factors on asthma remission (table 4). Children having more siblings, middle social class, lived in capital or capital suburb, female gender, maternal smoking during pregnancy and increasing age of asthma onset had a better chance of asthma remission, while children with maternal asthma history had lower likelihood of remission.
Table 4.
Prenatal factors on childhood asthma remission evaluated by Cox proportional hazards regression model (HR, 95% CI)
| Crude HR | 95% CI | Adjusted HR* | 95% CI | |
| Maternal age | 0.94 | 0.92 to 0.96 | 0.93 | 0.91 to 0.95 |
| Gestational age | 1.00 | 0.99 to 1.01 | 1.00 | 0.99 to 1.01 |
| Birth weight | 0.99 | 0.98 to 1.01 | 1.00 | 0.99 to 1.02 |
| Parity | ||||
| 1 | Ref | Ref | ||
| 2 | 0.98 | 0.94 to 1.03 | 1.04 | 0.99 to 1.08 |
| ≥3 | 0.99 | 0.94 to 1.04 | 1.06 | 1.00 to 1.13 |
| Maternal social status | ||||
| Not in labour market | 1.03 | 0.98 to 1.09 | 1.01 | 0.95 to 1.06 |
| Unskilled workers | 1.14 | 1.04 to 1.26 | 1.17 | 1.06 to 1.29 |
| Skilled workers and white-collar workers | 1.08 | 1.03 to 1.13 | 1.06 | 1.02 to 1.11 |
| Top level status | Ref | Ref | ||
| Residence | ||||
| Capital or capital suburb | 1.06 | 1.02 to 1.11 | 1.07 | 1.02 to 1.12 |
| Provincial city or town | 0.93 | 0.88 to 0.99 | 0.95 | 0.89 to 1.00 |
| Rural areas | Ref | Ref | ||
| Maternal smoking during pregnancy | 1.13 | 1.08 to 1.17 | 1.13 | 1.08 to 1.18 |
| Maternal history of asthma | 0.79 | 0.73 to 0.85 | 0.79 | 0.73 to 0.85 |
| Sex | ||||
| Male | Ref | Ref | ||
| Female | 1.18 | 1.14 to 1.23 | 1.18 | 1.13 to 1.22 |
| Mode of delivery | ||||
| Delivered vaginally | Ref | Ref | ||
| Delivered by caesarean section | 0.95 | 0.89 to 1.02 | 0.98 | 0.92 to 1.05 |
| Age of asthma onset | 1.10 | 1.09 to 1.11 | 1.10 | 1.09 to 1.11 |
*Cox proportional hazards regression model (HR, 95% CI) adjusting for maternal age at delivery; maternal parity; maternal socio-economic status at delivery; maternal smoking status during pregnancy; maternal history of asthma; sex of the child; gestational weeks at delivery; maternal residence at delivery; birth weight, delivery method and age of asthma onset.
We also explored the association of early life factors with early remission of childhood asthma (table 5). Female children, having siblings, middle social status and maternal smoking during pregnancy were associated with a better chance of early remission of asthma. More advanced maternal age at delivery, maternal asthma history, living in middle sized cities and increasing age of asthma onset had a lower probability of early remission of asthma.
Table 5.
Prenatal factors and early remission (<12 years of age) of childhood asthma analysed by logistic regression model in asthma patients (OR, 95% CI)
| Crude OR | 95% CI | Adjusted OR* | 95% CI | |
| Maternal age | 0.91 | 0.88 to 0.94 | 0.91 | 0.87 to 0.94 |
| Gestational age | 0.98 | 0.97 to 1.00 | 0.99 | 0.97 to 1.00 |
| Birth weight | 0.96 | 0.94 to 0.98 | 0.99 | 0.96 to 1.02 |
| Parity | ||||
| 1 | Ref | Ref | ||
| 2 | 1.05 | 0.99 to 1.12 | 1.12 | 1.04 to 1.19 |
| ≥3 | 1.03 | 0.95 to 1.11 | 1.11 | 1.01 to 1.21 |
| Maternal social status | ||||
| Not in labour market | 1.15 | 1.06 to 1.24 | 1.06 | 0.98 to 1.15 |
| Unskilled workers | 1.33 | 1.15 to 1.54 | 1.30 | 1.12 to 1.50 |
| Skilled workers and white-collar workers | 1.27 | 1.19 to 1.36 | 1.20 | 1.12 to 1.28 |
| Top level status | Ref | Ref | ||
| Residence | ||||
| Capital or capital suburb | 0.93 | 0.87 to 0.99 | 0.97 | 0.91 to 1.04 |
| Provincial city or town | 0.86 | 0.79 to 0.93 | 0.89 | 0.82 to 0.97 |
| Rural areas | Ref | Ref | ||
| Maternal smoking during pregnancy | 1.24 | 1.17 to 1.32 | 1.19 | 1.12 to 1.27 |
| Maternal history of asthma | 0.80 | 0.71 to 0.89 | 0.78 | 0.69 to 0.87 |
| Sex | ||||
| Male | Ref | Ref | ||
| Female | 1.20 | 1.13 to 1.27 | 1.20 | 1.13 to 1.27 |
| Mode of delivery | ||||
| Delivered vaginally | Ref | Ref | ||
| Delivered by caesarean section | 1.02 | 0.93 to 1.12 | 1.01 | 0.91 to 1.12 |
| Age of asthma onset | 1.48 | 1.47 to 1.50 | 1.48 | 1.46 to 1.50 |
*Logistic regression model (OR, 95% CI) adjusting for maternal age at delivery; maternal parity; maternal socio-economic status at delivery; maternal smoking status during pregnancy; maternal history of asthma; sex of the child; gestational weeks at delivery; maternal residence at delivery; birth weight, delivery method and age of asthma onset.
Discussion
Our study is one of the largest retrospective cohort studies, showing that the overall occurrence rate of asthma in children decreased with age, with boys having a higher occurrence rate before age 11 than girls. Asthma remised in less than half of the asthmatic children by age 12–14. Girls had a higher remission rate than boys. Being a later born child, female and living in capital or capital suburb had a lower risk of asthma and a better chance of remission. Maternal asthma history increased the risk of asthma and reduced the likelihood of remission in offspring. These patterns remained the same for early-onset asthma. In addition, CS was a risk factor for early-onset asthma while the increasing gestational age at delivery and living in capital or capital suburb was associated with a reduced risk of early-onset asthma.
The accumulative occurrence of asthma was 13.3% in our study. Previous studies from Denmark reported the prevalence of asthma ranging from 4% to 31% between 1996 and 2009. In a cross-sectional study among unselected Danish schoolchildren aged 6–17 years, the prevalence of current physician-diagnosed asthma was 4.0% in 1990–1991 and 3.6% in 2001.18 In a population-based cohort study included all live born singletons from 1996 to 2008 in northern Denmark, the prevalence of asthma was 12.4% by the end of 2009.19 In the longitudinal Danish National Birth Cohort (1996–2002), a total of 17% of children had doctor-diagnosed asthma at 18 months of age. At age 7 years, 6% of children ever had asthma with hospital admission and 31% ever with asthma medication.20 The difference in the prevalence of asthma was mainly due to varying definition of asthma and the age of asthma diagnosis.18 19 21 The definition of asthma based on hospital contact tended to capture more severe cases, while the prescriptions of medication may identify a heterogeneous mix of cases from mild to severe cases.22 In order to include as many asthmatic patients as possible, we defined asthma case as at least one hospital contact for asthma or two prescriptions for asthma medication. The occurrence of asthma in our study was consistent with the previous studies in the similar study period in Denmark.21
The total remission rate was 44% in the present study, while the estimated remission rate varied from 22% to 65% in the literature.23 The remission rate could be influenced by its definition, duration of follow-up and the age of the participants. Asthma remission was defined as absence of asthma symptoms and asthmatic medication use for a period of time, ranging from 1 to 10 years in different studies,24–29 and we defined it as the absence of asthma medication and hospitalisation in 2 years in our study.
Our results are consistent with previous studies on the effects of sex on the occurrence and remission of asthma. A cross-sectional study with 18 873 participants in Italy from 1998 through 2000 found that early-onset asthma, defined as the onset before 10 years of age, occurred more often in boys, and had a better prognosis. While late-onset asthma, generally occurring during or after puberty, more likely affected girls and had a poorer prognosis.25 A similar pattern was found in Asian population.30 In an unselected birth cohort conducted in Dunedin, New Zealand, 613 participants were followed from childhood to 26 years of age. More than 25% of children had persisted wheezing or relapse after remission. Girls and early onset were found to be the factors reducing asthma remission.5
Meanwhile, we also discovered that women had a slightly higher occurrence rate than men after 12 years of age. In the TRacking Adolescents’ Individual Lives Survey, a similar shift in the prevalence of asthma occurred between 11 and 16 years. The authors attributed this crossover to both an increased incidence and decreased remission of asthma in female compared with male subjects.31 This phenomenon was probably owing to the onset of puberty when the increase in progesterone and oestrogen levels during the luteal phase could lead to increased inflammation of the airway wall,32 while androgens may protect against the development of asthma.33 Following the population in the present study forward into adulthood would provide more understanding of the role of sex on the remission of childhood asthma.
In addition, the association of maternal asthma with childhood asthma is consistent with previous studies. The European Community Respiratory Health Survey recruited 18 156 subjects from 16 countries and found that family history of asthma or allergy was associated with a higher risk of developing asthma and a lower chance of remission.17 In the Tasmanian Longitudinal Health Study that enrolled 8583 7-year-old schoolchildren, maternal asthma was also found to adversely affect asthma remission in children.6 The association between maternal history and child asthma may be attributable to genetic factors and child early exposure to the same living environment.34 35 It was postulated that maternal asthma during pregnancy could result in decreased fetal oxygenation during development, leading to asthma in offspring.36 37
As a number of studies have demonstrated the increased risk of asthma by caesarean delivery,38–40 our study further showed that CS was a risk factor for early-onset asthma, and reduced the probability of asthma remission. An increased risk of asthma and asthma exacerbations was also found in the longitudinally followed COPSAC2000 birth cohort among children born by caesarean delivery.41 CS could affect the formation of normal intestinal flora of the newborn,42 which played an important role in the development of the immune system and, therefore, increased the risk of allergic diseases.43 Anaesthetic drugs used during CS were thought to cross the placental barrier and alter the immune system of the offspring.44 The levels of several biomarkers involved in the immune system differed significantly between CS and vaginal births, including IL-4γ, IL-1β, IL-6 and tumour necrosis factor-α.45 46 In addition, CS was associated with offspring obesity after accounting for major confounding factors.47 Obese asthma patients might have an increased risk of more symptoms, more frequent and severe exacerbations, reduced response to several asthma medications and decreased quality of life.48
Consistent with the previous studies,49–52 maternal smoking during pregnancy increased the risk of offspring asthma. In a meta-analysis, prenatal maternal smoking was associated with a 23% increased risk of asthma between the ages of 5 and 18 years (OR=1.23, 95% CI 1.12 to 1.36, 11 studies),53 which is similar to our finding. Paradoxically, maternal smoking during pregnancy was associated with an increased chance of asthma remission in offspring in our study. In the longitudinal study by Andersson et al, persistent asthma was also less common among children of mothers who smoked during pregnancy.4 A possible explanation was that maternal smoking may be more likely to cause non-allergic asthma, which remits more easily than allergic asthma.54 The association of maternal smoking with asthma was also subjected to the ‘healthy smoker’ effect, that is, smokers are less likely to have asthma.55 Besides, the time of maternal smoking, the number of cigarettes smoked per day, maternal home secondhand smoke exposure during pregnancy, postnatal smoke exposure and genetic factors could also impact on the effect of maternal smoking on childhood asthma. Thus, more researches would be warranted to confirm this finding.
Our study has several strengths. First, the definition of asthma was based on the registry information of the pharmacy and hospital rather than parents’ self-report. It was likely to be more objective and credible. Second, it was a longitudinal study with a very large sample size and based on the national registry database. Selection bias was minimized. However, we did not have detailed information on child growth and asthmatic symptoms. Since our study did not have information on child body mass index (BMI), we might have missed an important risk factor for asthma. It remains uncertain whether there is a causal relationship between obesity and asthma in children and adolescents.56 Several studies have shown that obesity increased later onset of asthma, whereas others found a link only among specific subsets of at-risk patients, affected by race, sex, atopic status and timing of obesity.57 At the meantime, several high-quality studies failed to find evidence of obesity–asthma relationship in children.57 A US prospective cohort study showed that children with asthma had a much higher risk of developing obesity during childhood and adolescence compared with children without asthma, suggesting the possible impact of asthma history and asthma medication on the obesity risks in childhood.58 In addition, BMI was not identified as a significant risk factor for asthma non-remission in a population-based retrospective study.14 Further research on this important factor is warranted. Besides, the follow-up period was not long enough to observe asthma persistent into adulthood. Although the socioeconomic status (SES) data represented the status at time of delivery, it could change over time. Smoking status and asthma history of other relatives were not available in the present database. All of these may have resulted in residual confounding to an unknown degree.
Conclusions
Our study demonstrated that the overall occurrence of asthma in children decreased with age. Maternal asthma history, mode of delivery, maternal smoking during pregnancy, and sex of children affected the occurrence and remission of childhood asthma.
Supplementary Material
Footnotes
QC and JC contributed equally.
Contributors: Study concept and design: JZ, YZ, JL. Drafting of the manuscript: QC, LH, JZ. Statistical analysis: JC, QC, YT, YZ, JL, JZ. All authors revised the manuscript and approved the final version. JZ and JL are the guarantors of the content of the manuscript, including the data and analysis.
Funding: This study was supported by grant by 81 530 086 (JZ); 81 803 244 (QC); 11771146, 11 831 008 (YZ); 11 271 136 (YT) from National Natural Science Foundation of China. This study was also supported in part by B14019 from the 111 Project of China (YT).
Competing interests: None declared.
Provenance and peer review: Not commissioned; externally peer reviewed.
Supplemental material: This content has been supplied by the author(s). It has not been vetted by BMJ Publishing Group Limited (BMJ) and may not have been peer-reviewed. Any opinions or recommendations discussed are solely those of the author(s) and are not endorsed by BMJ. BMJ disclaims all liability and responsibility arising from any reliance placed on the content. Where the content includes any translated material, BMJ does not warrant the accuracy and reliability of the translations (including but not limited to local regulations, clinical guidelines, terminology, drug names and drug dosages), and is not responsible for any error and/or omissions arising from translation and adaptation or otherwise.
Data availability statement
Data are available upon reasonable request. No additional data are available.
Ethics statements
Patient consent for publication
Not applicable.
Ethics approval
The study was approved by the Danish Data Protection Agency (Record number 2013-41-2569).
References
- 1.Mims JW. Asthma: definitions and pathophysiology. Int Forum Allergy Rhinol 2015;5:S2–6. 10.1002/alr.21609 [DOI] [PubMed] [Google Scholar]
- 2.Papi A, Brightling C, Pedersen SE. Asthma. Lancet 2017. [DOI] [PubMed] [Google Scholar]
- 3.Holgate ST, Wenzel S, Postma DS, et al. Asthma. Nat Rev Dis Primers 2015;1:15025. 10.1038/nrdp.2015.25 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Andersson M, Hedman L, Bjerg A, et al. Remission and persistence of asthma followed from 7 to 19 years of age. Pediatrics 2013;132:e435–42. 10.1542/peds.2013-0741 [DOI] [PubMed] [Google Scholar]
- 5.Sears MR, Greene JM, Willan AR, et al. A longitudinal, population-based, cohort study of childhood asthma followed to adulthood. N Engl J Med 2003;349:1414–22. 10.1056/NEJMoa022363 [DOI] [PubMed] [Google Scholar]
- 6.Burgess JA, Matheson MC, Gurrin LC, et al. Factors influencing asthma remission: a longitudinal study from childhood to middle age. Thorax 2011;66:508–13. 10.1136/thx.2010.146845 [DOI] [PubMed] [Google Scholar]
- 7.Moffatt MF, Gut IG, Demenais F, et al. A large-scale, consortium-based genomewide association study of asthma. N Engl J Med 2010;363:1211–21. 10.1056/NEJMoa0906312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tegethoff M, Greene N, Olsen J, et al. Stress during pregnancy and offspring pediatric disease: a national cohort study. Environ Health Perspect 2011;119:1647–52. 10.1289/ehp.1003253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Andersen ABT, Farkas DK, Mehnert F, Pedersen L, et al. Use of prescription paracetamol during pregnancy and risk of asthma in children: a population-based Danish cohort study. Clin Epidemiol 2012;4:33–40. 10.2147/CLEP.S28312 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Klopp A, Vehling L, Becker AB, et al. Modes of infant feeding and the risk of childhood asthma: a prospective birth cohort study. J Pediatr 2017;190:192–9. 10.1016/j.jpeds.2017.07.012 [DOI] [PubMed] [Google Scholar]
- 11.Bliddal M, Broe A, Pottegård A, et al. The Danish medical birth register. Eur J Epidemiol 2018;33:27–36. 10.1007/s10654-018-0356-1 [DOI] [PubMed] [Google Scholar]
- 12.Chen W, Marra CA, Lynd LD, et al. The natural history of severe asthma and influences of early risk factors: a population-based cohort study. Thorax 2016;71:267–75. 10.1136/thoraxjnl-2015-207530 [DOI] [PubMed] [Google Scholar]
- 13.Zhang H, Kaushal A, Soto-Ramírez N, et al. Acquisition, remission, and persistence of eczema, asthma, and rhinitis in children. Clin Exp Allergy 2018;48:568–76. 10.1111/cea.13096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Honkamäki J, Piirilä P, Hisinger-Mölkänen H, et al. Asthma remission by age at diagnosis and gender in a population-based study. J Allergy Clin Immunol Pract 2021;9:1950–9. 10.1016/j.jaip.2020.12.015 [DOI] [PubMed] [Google Scholar]
- 15.Liu X, Olsen J, Agerbo E, et al. Maternal preeclampsia and childhood asthma in the offspring. Pediatr Allergy Immunol 2015;26:181–5. 10.1111/pai.12344 [DOI] [PubMed] [Google Scholar]
- 16.Carpaij OA, Burgess JK, Kerstjens HAM, et al. A review on the pathophysiology of asthma remission. Pharmacol Ther 2019;201:8–24. 10.1016/j.pharmthera.2019.05.002 [DOI] [PubMed] [Google Scholar]
- 17.de Marco R, Pattaro C, Locatelli F, et al. Influence of early life exposures on incidence and remission of asthma throughout life☆. J Allergy Clin Immunol 2004;113:845–52. 10.1016/j.jaci.2004.01.780 [DOI] [PubMed] [Google Scholar]
- 18.Zilmer M, Steen NP, Zachariassen G, et al. Prevalence of asthma and bronchial hyperreactivity in Danish schoolchildren: no change over 10 years. Acta Paediatr 2011;100:385–9. 10.1111/j.1651-2227.2010.02036.x [DOI] [PubMed] [Google Scholar]
- 19.Andersen ABT, Erichsen R, Farkas DK, et al. Prenatal exposure to acid-suppressive drugs and the risk of childhood asthma: a population-based Danish cohort study. Aliment Pharmacol Ther 2012;35:1190–8. 10.1111/j.1365-2036.2012.05073.x [DOI] [PubMed] [Google Scholar]
- 20.Maslova E, Strøm M, Oken E, et al. Fish intake during pregnancy and the risk of child asthma and allergic rhinitis - longitudinal evidence from the Danish National Birth Cohort. Br J Nutr 2013;110:1313–25. 10.1017/S000711451300038X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liu X, Liew Z, Olsen J, et al. Association of prenatal exposure to acetaminophen and coffee with childhood asthma. Pharmacoepidemiol Drug Saf 2016;25:188–95. 10.1002/pds.3940 [DOI] [PubMed] [Google Scholar]
- 22.Hansen S, Strøm M, Maslova E, et al. A comparison of three methods to measure asthma in epidemiologic studies: results from the Danish national birth cohort. PLoS One 2012;7:e36328. 10.1371/journal.pone.0036328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brewczyński PZ, Brodziak A. Have recent investigations into remission from childhood asthma helped in understanding the pathogenesis of this disease? Med Sci Monit 2015;21:570–5. 10.12659/MSM.893575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rönmark E, Jönsson E, Lundbäck B. Remission of asthma in the middle aged and elderly: report from the obstructive lung disease in northern Sweden study. Thorax 1999;54:611–3. 10.1136/thx.54.7.611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.De Marco R, Locatelli F, Cerveri I, et al. Incidence and remission of asthma: a retrospective study on the natural history of asthma in Italy. J Allergy Clin Immunol 2002;110:228–35. 10.1067/mai.2002.125600 [DOI] [PubMed] [Google Scholar]
- 26.Westerhof GA, Coumou H, de Nijs SB, et al. Clinical predictors of remission and persistence of adult-onset asthma. J Allergy Clin Immunol 2018;141:104–9. 10.1016/j.jaci.2017.03.034 [DOI] [PubMed] [Google Scholar]
- 27.Bjerg-Bäcklund A, Bäcklund AB, Perzanowski MS, Platts-Mills T, et al. Asthma during the primary school ages--prevalence, remission and the impact of allergic sensitization. Allergy 2006;61:549–55. 10.1111/j.1398-9995.2006.01027.x [DOI] [PubMed] [Google Scholar]
- 28.Holm M, Omenaas E, Gislason T, et al. Remission of asthma: a prospective longitudinal study from northern Europe (Rhine study). Eur Respir J 2007;30:62–5. 10.1183/09031936.00121705 [DOI] [PubMed] [Google Scholar]
- 29.Upham JW, James AL. Remission of asthma: the next therapeutic frontier? Pharmacol Ther 2011;130:38–45. 10.1016/j.pharmthera.2011.01.002 [DOI] [PubMed] [Google Scholar]
- 30.Wu T-J, Wu C-F, Lee YL, et al. Asthma incidence, remission, relapse and persistence: a population-based study in southern Taiwan. Respir Res 2014;15:135. 10.1186/s12931-014-0135-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vink NM, Postma DS, Schouten JP, et al. Gender differences in asthma development and remission during transition through puberty: the tracking adolescents' individual lives survey (trails) study. J Allergy Clin Immunol 2010;126:498–504. 10.1016/j.jaci.2010.06.018 [DOI] [PubMed] [Google Scholar]
- 32.Melgert BN, Ray A, Hylkema MN, et al. Are there reasons why adult asthma is more common in females? Curr Allergy Asthma Rep 2007;7:143–50. 10.1007/s11882-007-0012-4 [DOI] [PubMed] [Google Scholar]
- 33.Chen W, Mempel M, Schober W, et al. Gender difference, sex hormones, and immediate type hypersensitivity reactions. Allergy 2008;63:1418–27. 10.1111/j.1398-9995.2008.01880.x [DOI] [PubMed] [Google Scholar]
- 34.Blumenthal MN. The role of genetics in the development of asthma and atopy. Curr Opin Allergy Clin Immunol 2005;5:141–5. 10.1097/01.all.0000162306.12728.c2 [DOI] [PubMed] [Google Scholar]
- 35.London SJ, James Gauderman W, Avol E, et al. Family history and the risk of early-onset persistent, early-onset transient, and late-onset asthma. Epidemiology 2001;12:577–83. 10.1097/00001648-200109000-00019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bracken MB, Belanger K, Cookson WO, et al. Genetic and perinatal risk factors for asthma onset and severity: a review and theoretical analysis. Epidemiol Rev 2002;24:176–89. 10.1093/epirev/mxf012 [DOI] [PubMed] [Google Scholar]
- 37.Davidson R, Roberts SE, Wotton CJ, et al. Influence of maternal and perinatal factors on subsequent hospitalisation for asthma in children: evidence from the Oxford record linkage study. BMC Pulm Med 2010;10:14. 10.1186/1471-2466-10-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Thavagnanam S, Fleming J, Bromley A, et al. A meta-analysis of the association between caesarean section and childhood asthma. Clin Exp Allergy 2008;38:629–33. 10.1111/j.1365-2222.2007.02780.x [DOI] [PubMed] [Google Scholar]
- 39.Chu S, Chen Q, Chen Y, et al. Cesarean section without medical indication and risk of childhood asthma, and attenuation by breastfeeding. PLoS One 2017;12:e0184920. 10.1371/journal.pone.0184920 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Rusconi F, Zugna D, Annesi-Maesano I, et al. Mode of delivery and asthma at school age in 9 European birth cohorts. Am J Epidemiol 2017;185:465–73. 10.1093/aje/kwx021 [DOI] [PubMed] [Google Scholar]
- 41.Sevelsted A, Stokholm J, Bisgaard H. Risk of asthma from cesarean delivery depends on membrane rupture. J Pediatr 2016;171:10.1016/j.jpeds.2015.12.066:38–42. 10.1016/j.jpeds.2015.12.066 [DOI] [PubMed] [Google Scholar]
- 42.Rutayisire E, Huang K, Liu Y, et al. The mode of delivery affects the diversity and colonization pattern of the gut microbiota during the first year of infants' life: a systematic review. BMC Gastroenterol 2016;16:86. 10.1186/s12876-016-0498-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Wopereis H, Sim K, Shaw A, et al. Intestinal microbiota in infants at high risk for allergy: effects of prebiotics and role in eczema development. J Allergy Clin Immunol 2018;141:10.1016/j.jaci.2017.05.054:1334–42. 10.1016/j.jaci.2017.05.054 [DOI] [PubMed] [Google Scholar]
- 44.Rizzo A, Campanile D, Spedicato M, et al. Update on anesthesia and the immune response in newborns delivered by cesarian section. Immunopharmacol Immunotoxicol 2011;33:581–5. 10.3109/08923973.2010.549137 [DOI] [PubMed] [Google Scholar]
- 45.Malamitsi-Puchner A, Protonotariou E, Boutsikou T, et al. The influence of the mode of delivery on circulating cytokine concentrations in the perinatal period. Early Hum Dev 2005;81:387–92. 10.1016/j.earlhumdev.2004.10.017 [DOI] [PubMed] [Google Scholar]
- 46.Zanardo V, Soldà G, Trevisanuto D. Elective cesarean section and fetal immune-endocrine response. Int J Gynaecol Obstet 2006;95:52–3. 10.1016/j.ijgo.2006.06.022 [DOI] [PubMed] [Google Scholar]
- 47.Yuan C, Gaskins AJ, Blaine AI, et al. Association between cesarean birth and risk of obesity in offspring in childhood, adolescence, and early adulthood. JAMA Pediatr 2016;170:e162385. 10.1001/jamapediatrics.2016.2385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Peters U, Dixon AE, Forno E. Obesity and asthma. J Allergy Clin Immunol 2018;141:1169–79. 10.1016/j.jaci.2018.02.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.den Dekker HT, Voort AMMS-vander, de Jongste JC, et al. Tobacco smoke exposure, airway resistance, and asthma in school-age children: the generation R study. Chest 2015;148:607–17. 10.1378/chest.14-1520 [DOI] [PubMed] [Google Scholar]
- 50.Simons E, To T, Moineddin R, et al. Maternal second-hand smoke exposure in pregnancy is associated with childhood asthma development. J Allergy Clin Immunol 2014;2:201–7. 10.1016/j.jaip.2013.11.014 [DOI] [PubMed] [Google Scholar]
- 51.McEvoy CT, Spindel ER. Pulmonary effects of maternal smoking on the fetus and child: effects on lung development, respiratory morbidities, and life long lung health. Paediatr Respir Rev 2017;21:27–33. 10.1016/j.prrv.2016.08.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Vanker A, Gie RP, Zar HJ. The association between environmental tobacco smoke exposure and childhood respiratory disease: a review. Expert Rev Respir Med 2017;11:661–73. 10.1080/17476348.2017.1338949 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Burke H, Leonardi-Bee J, Hashim A, et al. Prenatal and passive smoke exposure and incidence of asthma and wheeze: systematic review and meta-analysis. Pediatrics 2012;129:735–44. 10.1542/peds.2011-2196 [DOI] [PubMed] [Google Scholar]
- 54.Rönmark E, Jönsson E, Platts-Mills T, et al. Different pattern of risk factors for atopic and nonatopic asthma among children--report from the Obstructive Lung Disease in Northern Sweden Study. Allergy 1999;54:926–35. 10.1034/j.1398-9995.1999.00044.x [DOI] [PubMed] [Google Scholar]
- 55.Becklake MR, Lalloo U. The 'healthy smoker': a phenomenon of health selection? Respiration 1990;57:137–44. 10.1159/000195837 [DOI] [PubMed] [Google Scholar]
- 56.Miethe S, Karsonova A, Karaulov A, et al. Obesity and asthma. J Allergy Clin Immunol 2020;146:685–93. 10.1016/j.jaci.2020.08.011 [DOI] [PubMed] [Google Scholar]
- 57.Lang JE. Obesity and childhood asthma. Curr Opin Pulm Med 2019;25:34–43. 10.1097/MCP.0000000000000537 [DOI] [PubMed] [Google Scholar]
- 58.Chen Z, Salam MT, Alderete TL, et al. Effects of childhood asthma on the development of obesity among school-aged children. Am J Respir Crit Care Med 2017;195:1181–8. 10.1164/rccm.201608-1691OC [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2020-045728supp001.pdf (218.7KB, pdf)
bmjopen-2020-045728supp002.pdf (50.8KB, pdf)
Data Availability Statement
Data are available upon reasonable request. No additional data are available.