SUMMARY
Objectives:
We sought to collate and summarize high-quality data on non-typhoidal Salmonella invasive disease (iNTS) incidence to provide contemporary incidence estimates by location and year.
Methods:
We systematically searched the databases Embase + MEDLINE, Web of Science, and PubMed for articles published on the incidence of iNTS from inception of the database through 8 May 2020 with no language, country, date, or demographic restrictions applied. A meta-analysis was performed to report pooled iNTS incidence as a rate of cases per 100,000 per year.
Results:
Among 13 studies eligible for analysis, there were 68 estimates of incidence. Overall pooled incidence (95% CI) was 44.8 (31.5–60.5) per 100,000 persons per year. When stratified by region, pooled incidence was significantly higher in Africa than Asia, 51.0 (36.3–68.0) compared to 1.0 (0.2–2.5), respectively. Incidence was consistently higher in children aged <5 years compared with older age groups. Incidence displayed considerable heterogeneity in both place and time, varying substantially between locations and over consecutive years in the same location.
Conclusions:
iNTS incidence varies by region, location, age group, and over time. Concerted efforts are needed to address the limited high-quality data available on iNTS disease incidence.
Keywords: Systematic review, Meta-analysis, Non-typhoidal Salmonella, Incidence
Introduction
Non-typhoidal Salmonella (NTS) are an important cause of self-limited diarrheal disease often transmitted by food or water.1 However, in some patients NTS cause serious, life-threatening invasive infections involving the bloodstream, meninges, and other normally sterile sites.2,3 Patients with non-typhoidal Salmonella invasive disease (iNTS) often present with a non-specific febrile illnesses in the absence of recent or current diarrhea that is difficult to distinguish from other infectious diseases including malaria and typhoid fever.2 iNTS is a serious illness with a case fatality ratio of approximately 15%40 and was estimated to have caused 535,000 illnesses and more than 77,000 deaths in 2017.4 In a recent systematic review on the prevalence of community-onset bloodstream infections (BSI), NTS were among the most frequently isolated pathogens.5 Salmonella enterica subspecies enterica serovars Typhimurium and Enteritidis, accounted for more than 80% of serovars causing iNTS.5,6
Regionally, iNTS disease is concentrated in sub-Saharan Africa where it is a major cause of illness and death.4 Host risk factors including HIV, malaria, and malnutrition are thought to drive the disproportionate burden of iNTS in Africa compared to other regions.7,8 Treatment is proving increasingly problematic with widespread antimicrobial resistance among NTS isolates.9 Salmonella Typhimurium sequence type (ST) 313 accounts for the majority of Salmonella Typhimurium causing invasive disease in Africa,10 is predominately multi-drug resistant, and may also be extensively-drug resistant.11 Additionally, vaccine development has been slow to progress because of the limited data on burden of disease, as well as economic and technical challenges.12
In 2010 and 2017 population-based surveillance or national surveillance data were reviewed, and extrapolated to areas without incidence data based on host risk factors.4,13,14 However, a number of studies have been published since that time. We sought to collate and summarize high-quality data on iNTS incidence to provide contemporary incidence estimates by location and year for policymakers to support investments in vaccine development and non-vaccine intervention efforts.
Methods
Search strategy
We performed a search of the databases Embase + MEDLINE, Web of Science, and PubMed for articles published on the incidence of iNTS from inception of the database through 8 May 2020. No language, country, date, or demographic restrictions were applied to the search strategy (Box 1 and Supplementary Appendix A). We used key words of non-typhoidal Salmonella, non-Typhi, salmonellosis, incidence, epidemiology, burden, and specific serovars including Typhimurium, Enteritidis, Heidelberg, Dublin, Choleraesuis, Newport, Virchow, Concord, Brancaster, Freetown, Infantis, and Isangi. Specific serovars were selected based on previous reviews of prevalence of bloodstream infections5,6 and knowledge (JAC and CSM) of common Salmonella serovars that cause iNTS. Additionally, we screened citations of included full text articles. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses was followed.15 The protocol was submitted to PROPSERO International Prospective Register of Systematic Reviews on 14 May 2020 and registered on 10 July 2020 (CRD42020186362). As an analysis of published data, this study was exempt from requiring institutional review board approval.
Study selection
We included study designs based on active household or population-based surveillance, sentinel site surveillance using healthcare utilization surveys to adjust for under-ascertainment (i.e., hybrid surveillance or multiplier studies),16,17 prospective observational studies, or vaccine clinical trials for other invasive bacterial diseases with relevant control arm data. Studies recruited participants of any age reporting the number of cases of iNTS identified using cultures of a normally sterile site (e.g., blood, bone marrow) for confirmation. Raw data were required to calculate incidence rate as number of cases per 100,000 per year. We excluded study designs based on case reports, case series, and surveillance studies where collection of blood cultures from febrile patients was not systematic. We also excluded studies using only clinical indication (i.e., symptoms and signs), culture of a non-sterile site (e.g., stool or urine), or serology alone to classify a case of iNTS.
Text files for each database search result were downloaded and imported into Endnote X8 (Clarivate Analytics, Boston, MA, United States) and combined into one reference list. Duplicates of titles and abstracts were removed by Endnote, and uploaded to the online systematic review tool Rayyan (Qatar Computing Research Institute, Doha, Qatar) for screening.18 Titles and abstracts, and full text were screened in parallel for inclusion (CSM, FF, and EP). Data were then abstracted (CSM, FF, and EP) using Google Forms (Google LLC, Mountain View, CA, USA). The data abstraction form is available in Supplementary Appendix B. A third author (JAC) was consulted when discrepancies could not be resolved through discussion and reviewed the final dataset for completeness and accuracy.
Data abstraction and analysis
Abstracted study characteristics included study country and location, United Nations (UN) region and sub-region, study design, data collection start and end date, duration of surveillance in months, normally sterile sites cultured, eligibility criteria for culture request, and age group of participants (children ≤ 15 years, adults >15 years, or mixed ages). Age groups were categorized based on inclusion criteria or age range data provided in results.
Study designs were stratified into two groups: (1) Active, household, or population-based surveillance or hybrid surveillance that used multipliers for adjustment, and (2) Unadjusted sentinel site surveillance. Hybrid surveillance studies were defined as those using one or more of the three multipliers described by Andrews and colleagues to adjust the crude incidence for under-ascertainment.16 These multipliers made adjustments for culture sensitivity, enrollment capture, or facility coverage estimated by an household survey. We defined unadjusted sentinel site surveillance as all other studies that were not active, population-based, or hybrid surveillance. To be eligible, unadjusted surveillance studies were required to have well-defined catchment population information where we were confident that the authors were able to capture a large proportion of potential iNTS cases through systematic testing.
Guided by bias assessment tools for prevalence, incidence, and non-randomized studies,19–21 we assessed the risk of bias in two main domains. For the selection and recruitment domain, we evaluated study design, incidence multipliers, and patient selection. In the measurement and reporting domain, we evaluated eligibility criteria for receiving a culture, data for incidence calculations, and microbiology methods. Each domain question was scored low or high risk for bias, and an overall score of low, moderate, or high risk of bias was assigned to each study. Definitions for scoring and each question are provided in Supplementary Appendix C.
We recorded the number of cases of iNTS, including by serovar when available, the population under surveillance, and the duration of surveillance in months. The number of cases were divided by the number of months of surveillance and then multiplied by 12 to calculate the number of cases per year. Cases per year were then divided by the population under surveillance and multiplied by 100,000 to report incidence as a rate of cases per 100,000 per year. All incidence data presented were per 100,000 persons per year, unless otherwise noted. When data were available for multiple study years, incidence was calculated by individual year when it was possible to do so. We performed a meta-analysis in MetaXL version 5.3 (Epigear International) using DerSimonian-Lairdrandom-effects model with double arcsine transformation to report pooled incidence estimates.22 We evaluated heterogeneity using Cochran Q-test and I2.
Results
Our search strategy returned 9,779 articles (Fig. 1) to be screened. After 3,690 duplicate articles were removed, we screened 6,089 titles and abstracts for inclusion. Of these, 158 (2.6%) proceeded for full text review. We excluded 145 articles after reviewing the full text; the most common reason for exclusion was insufficient data available to calculate incidence. Thirteen articles were eligible for analysis.23–35
Fig. 1.
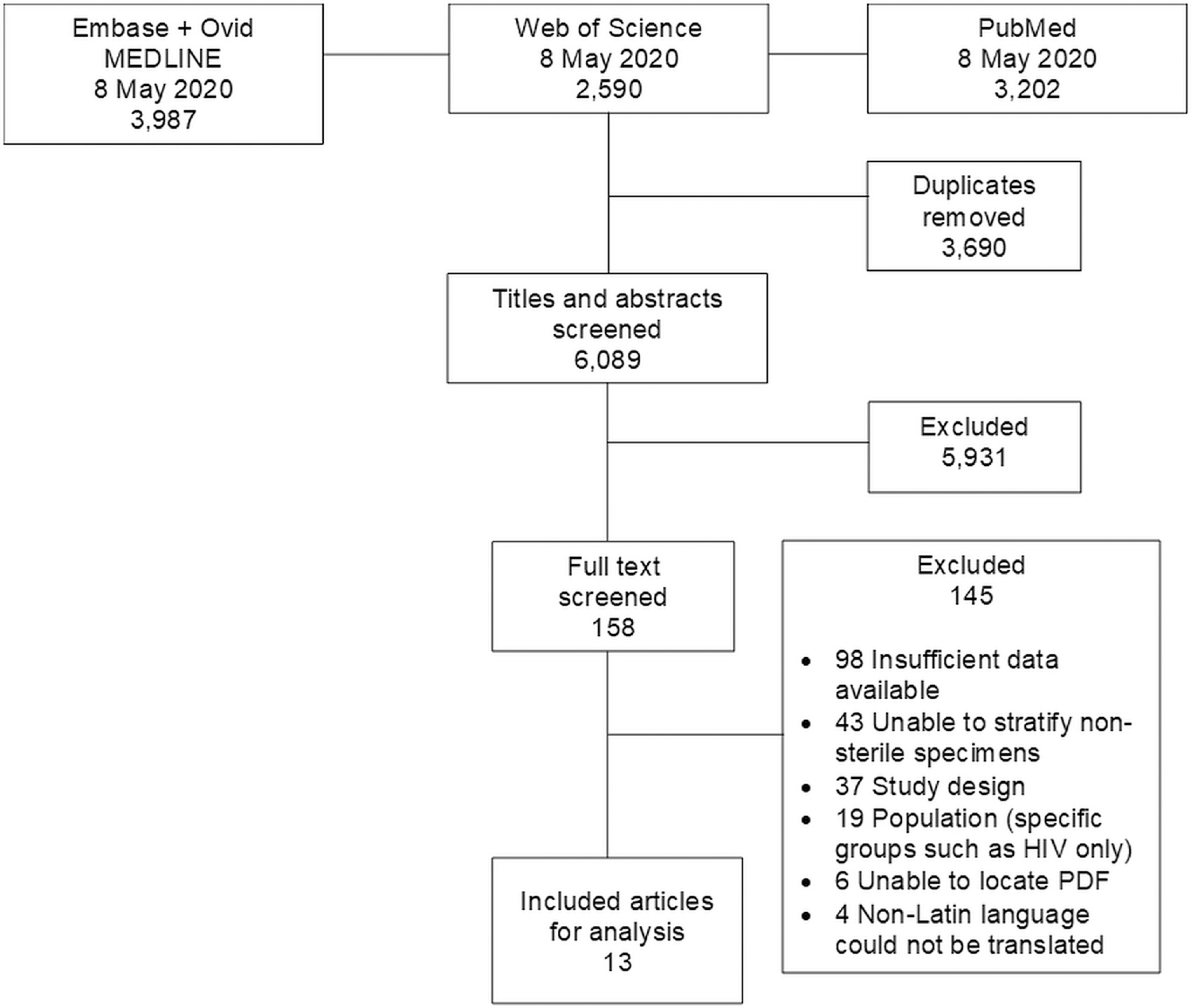
Preferred reporting items for systematic reviews and meta-analyses flow diagram of search strategy and selection of articles for incidence of non-typhoidal Salmonella invasive disease, 1996–2016.
Study characteristics and quality assessment
Among the 13 included studies, data were collected from 1 January 1996 through 31 December 2016 in 19 countries from Africa and Asia. There were no included studies from any other UN region (Table 1). Eight studies collected data either from multiple locations or during multiple consecutive years,23–25,27,29,33–35 resulting in 68 separate estimates of iNTS incidence. The median (range) population under surveillance was 571,000 (5,333 to 850,000). There were 63 (92.6%) incidence estimates from Africa and 5 (7.4%) from Asia. Of the 68 estimates, 53 (77.9%) were from the Eastern Africa sub-region. Among 53 estimates from Eastern Africa, 25 (47.2%) were from Kenya, 17 (32.1%) from Malawi, 6 (11.3%) from Tanzania, 2 (3.8%) from Madagascar, and one (1.9%) each from Ethiopia, Mozambique, and Uganda.
Table 1.
Characteristics of included studies of non-typhoidal Salmonella incidence by United Nations sub-region, 1996 through 2016.
| UNsub-region | Study location, Country | Study design | Years of data collection | Cultures | Inclusion age group | NTS serovar | Type of multiplier |
|---|---|---|---|---|---|---|---|
| Eastern Africa | Butajira, Ethiopia 27 | Hybrid surveillance | 2012–2014 | Blood | Mixed ages | NTS | None |
| Kilifi, Kenya 30 | Hybrid surveillance | 1996–2014 | Blood | Mixed ages | NTS | F, E | |
| Kilifi, Kenya 33 | Unadjusted sentinel site surveillance | 1999–2007 | Blood | Children | NTS | None | |
| Asembo, Kenya 34 | Hybrid surveillance | 2006–2009 | Blood | Mixed ages | NTS Salmonella Enteritidis Salmonella Heidelberg Salmonella Typhimurium |
F, E | |
| Kibera, Kenya 34 | Hybrid surveillance | 2007–2009 | Blood | Mixed ages | NTS Salmonella Enteritidis Salmonella Typhimurium |
F, E | |
| Lwak, Kenya 35 | Hybrid surveillance | 2009–2014 | Blood | Mixed ages | NTS | F, E | |
| Kibera, Kenya 35 | Hybrid surveillance | 2009–2014 | Blood | Mixed ages | NTS | F, E | |
| Siaya County, Kenya 32 | Vaccine trial control arms | 2009–2013 | Blood | Children | NTS | None | |
| Kibera, Kenya 27 | Hybrid surveillance | 2012–2013 | Blood | Mixed ages | NTS | F | |
| Imerintsiatosika, Madagascar 27 | Hybrid surveillance | 2011–2013 | Blood | Mixed ages | NTS | F | |
| Isotry, Madagascar 27 | Hybrid surveillance | 2012–2013 | Blood | Mixed ages | NTS | F | |
| Blantyre, Malawi 24 | Unadjusted sentinel site surveillance | 1998–2014 | Blood;CSF | Mixed ages | NTS Salmonella Enteritidis Salmonella Typhimurium |
T* | |
| Manhiça District, Mozambique 26 | Unadjusted sentinel site surveillance | 2001–2014 | Blood;CSF | Children | NTS Salmonella Enteritidis Salmonella Typhimurium |
None | |
| Muheza, Tanzania 29 | Unadjusted sentinel site surveillance | 2006–2010 | Blood | Children | NTS | None | |
| Moshi Rural District, Tanzania 27 | Hybrid surveillance | 2011–2013 | Blood | Mixed ages | NTS | F, E | |
| Moshi Urban District, Tanzania 27 | Hybrid surveillance | 2011–2013 | Blood | Mixed ages | NTS | F, E | |
| Rural southwest, Uganda 28 | Unadjusted sentinel site surveillance | 1996–2007 | Blood | Mixed ages | NTS | None | |
| Northern Africa | East Wad Medani, Sudan 27 | Hybrid surveillance | 2012–2013 | Blood | Mixed ages | NTS | F |
| Southern Africa | Pietermaritzburg, South Africa 27 | Hybrid surveillance | 2012–2014 | Blood | Mixed ages | NTS | None |
| Western Africa | Nioko II, Burkina Faso 27 | Hybrid surveillance | 2012–2013 | Blood | Mixed ages | NTS | F |
| Polesgo, Burkina Faso 27 | Hybrid surveillance | 2012–2013 | Blood | Mixed ages | NTS | F | |
| Ashanti Region, Ghana31 | Hybrid surveillance | 2007–2009 | Blood | Children | NTS | None | |
| Asante Akim, Ghana 23 | Hybrid surveillance | 2010–2012 | Blood | Children | NTS | F, E | |
| Asante Akim, Ghana27 | Hybrid surveillance | 2010–2012 | Blood | Children | NTS | F | |
| Bandim, Guinea-Bissau 27 | Hybrid surveillance | 2011–2013 | Blood | Mixed ages | NTS | F | |
| Pikine, Senegal 27 | Hybrid surveillance | 2011–2013 | Blood | Mixed ages | NTS | None | |
| Eastern Asia | Hechi, China 25 | Unadjusted sentinel site surveillance | 2001–2002 | Blood | Mixed ages | NTS | None |
| South-eastern Asia | North Jakarta, Indonesia 25 | Unadjusted sentinel site surveillance | 2002–2003 | Blood | Mixed ages | NTS | None |
| Hue, Vietnam 25 | Unadjusted sentinel site surveillance | 2002–2003 | Blood | Mixed ages | NTS | None | |
| Southern Asia | Kolkata, India 25 | Unadjusted sentinel site surveillance | 2003–2004 | Blood | Mixed ages | NTS | None |
| Karachi, Pakistan 25 | Unadjusted sentinel site surveillance | 2002–2004 | Blood | Children | NTS | None |
NTS = Non-typhoidal Salmonella; CSF = Cerebrospinal fluid; F = Facility coverage; E = Enrollment capture; T = Test sensitivity
Described adjusting incidence to account for blood culture sensitivity but did not provide data for the adjusted rates.
Data for 38 (55.9%) of 68 estimates were collected using an unadjusted sentinel site surveillance study design, while 30 (44.1%) estimates used hybrid surveillance design. There were no active, population-based studies that did not also include multipliers. Six studies reported a multiplier-adjusted incidence estimate.23,27,30,31,34,35 No article used all three multiplier adjustments described by Andrews, et al.16 In our bias assessment, five (38.5%) of 13 studies scored as high risk of bias,23,25,30,31,33 seven (53.8%) as moderate risk,24,26–29,32,34 and one (7.7%) as low risk (Fig. 2).35
Fig. 2.
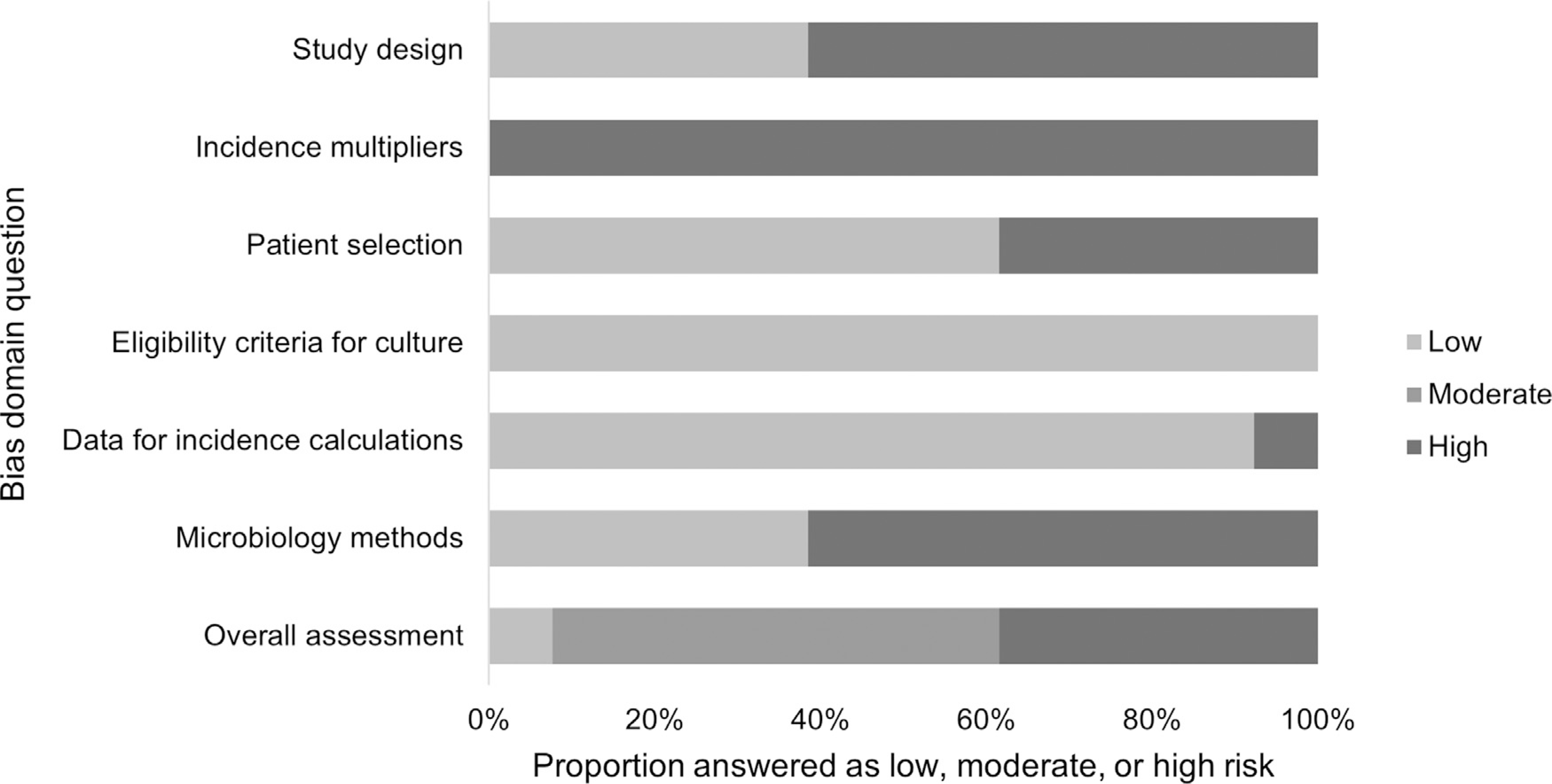
Quality assessment for risk of bias of included studies on incidence of non-typhoidal Salmonella invasive disease by domain, 1996 through 2016.
Incidence of non-typhoidal Salmonella invasive disease
Among 68 estimates of incidence, six (8.8%) reported no cases of iNTS isolated from a normally sterile site25,27 and the highest incidence reported was 1262.0 in Ghana (Supplement Table S1).31 Overall pooled incidence (95% CI) was 44.8 (31.5–60.5) per 100,000 persons per year. When stratified by region, pooled incidence was significantly higher in Africa than Asia, 51.0 (36.3–68.0) compared to 1.0 (0.2–2.5), respectively. Among sub-regions in Africa, pooled incidence was 71.3 (18.4–138.0) in Western Africa, 52.1 (36.7–70.0) in Eastern Africa, <0.1 (0.0–3.7) in Northern Africa, and <0.1 (0.0–0.4) in Southern Africa (Fig. 3). No included study reported estimates from Middle Africa. Among the three countries with the most estimates of incidence, pooled incidence was 85.6 (55.8–121.5), 56.7 (38.1–79.0), and 12.1 (0.0–32.4) in Malawi, Kenya, and Tanzania, respectively. Among the locations of Kibera, Kilifi, and Lwak, Kenya; and Blantyre, Malawi; where there were multiple consecutive years of incidence data, there was a pattern of lower incidence in more recent studies (Fig. 3).
Fig. 3.
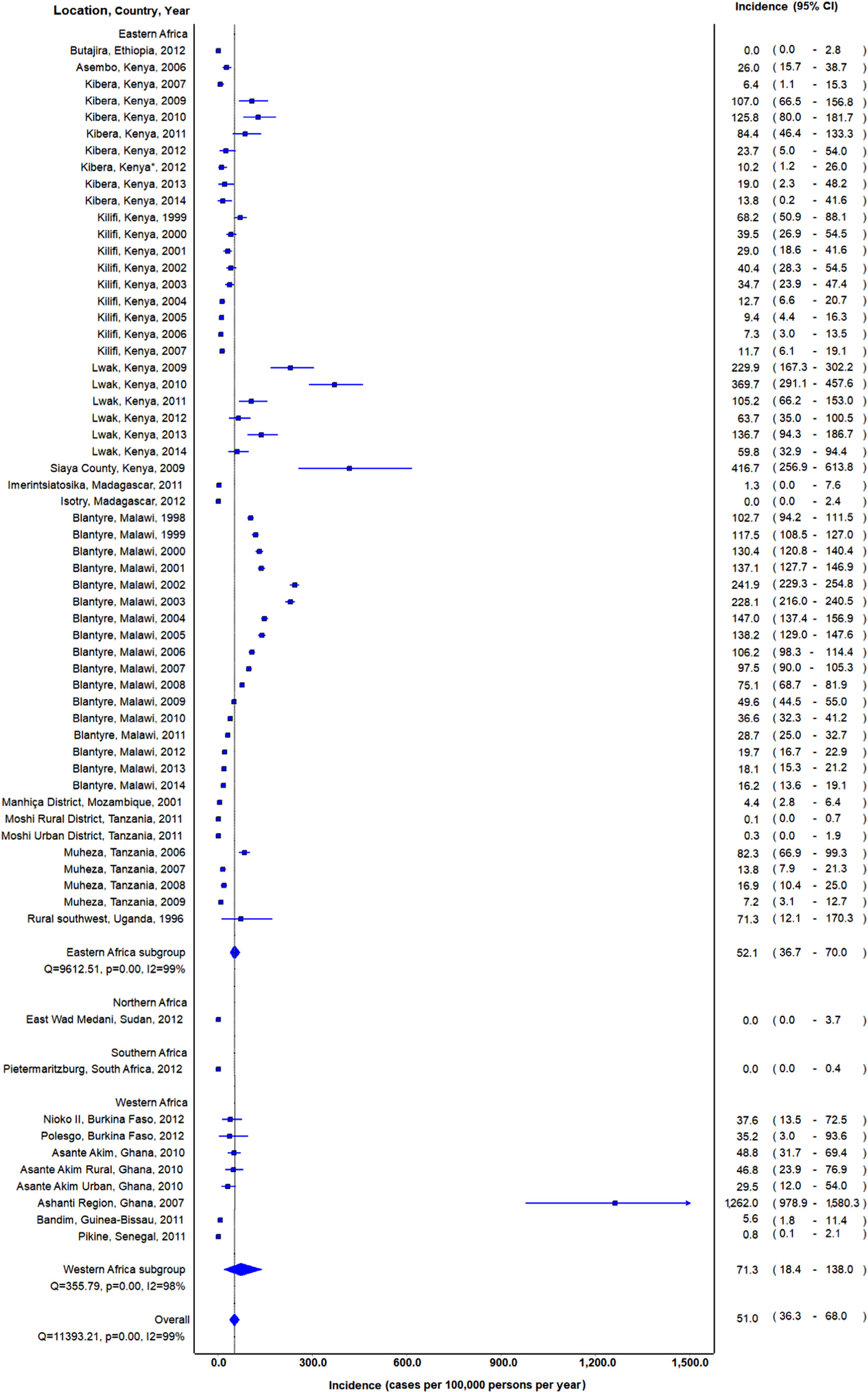
Forest plot of non-typhoidal Salmonella invasive disease incidence in Africa by United Nations sub-regions, 1996–2016
* Data from Marks et al. is same location and year as Verani et al.
By study design, pooled incidence among studies using hybrid surveillance was 42.2 (25.1–63.7), and among unadjusted sentinel surveillance studies was 45.5 (29.6–64.9). Eight studies provided age-stratified crude incidence estimates23,26,27,30–32,34,35 and five provided adjusted incidence using one or more multiplier (Table 2).23,27,30,34,35 Younger age groups between zero and five years consistently had higher iNTS incidence than older populations. The highest reported crude incidence among age-stratified studies was 4,133.2 among 1–11 month old infants in Siaya County, Kenya.32 Among the five studies using a multiplier, four (80.0%) used both facility coverage and enrollment capture adjustments23,30,34,35 and one (20.0%) facility coverage only.27 One study described adjusting the incidence to account for blood culture sensitivity but did not provide data for the adjusted rates.24
Table 2.
Age stratified incidence and adjusted incidence of non-typhoidal Salmonella invasive disease by United Nations sub-region and year, 1996 through 2016.
| UN sub-region | Study location, Country | Year surveillance started | Age stratified crude incidence, 100,000 per PYO | Type of multipliers | Age stratified adjusted or overall adjusted incidence, 100,000 per PYO |
|---|---|---|---|---|---|
| Eastern Africa | Kilifi, Kenya 30 | 1996 | 0–4y: 25.6; 5–14y: 1.9; > = 15y: 1.0 |
F, E | 0–4y: 32.6; 5–14y: 2.4; > = 15y: 1.7 |
| Manhiça District, Mozambique 26 | 2001 | 0–11m: 217.7; 12–59m: 172.7; > = 60m: 7.8 |
NA | NR | |
| Asembo (rural), Kenya 34 | 2006 | 0–4y: 206.0; 5–9y: 53.0; 10–17y: 6.0; 18–49: 76.0; > 50: 58.0 |
F, E | 0–4y: 2,085.0; 5–9y: 389.0; 10–17y: 24.0; 18–49y: 367.0; > 50y: 232.0; All ages: 580.0 |
|
| Kibera (urban), Kenya 34 | 2007 | 0–4y: 52.0; 5–9y: 12.0; 10–17y: 0.0; 18–49: 3.7; > 50: 0.0 |
F, E | 0–4y: 260.0; 5–9y: 37.0; 10–17y: 0.0; 18–49y: 11.0; > 50y: 0.0; All ages: 57.0 |
|
| Siaya County, Kenya 32 | 2009 | 1–11m: 4,133.2; 12–23m: 2,253.5; 24–35m: 1,279.2; 36–70m: 733.8 |
NA | NR | |
| Lwak (rural), Kenya 35 | 2009 | 0–4y: 501.8 a; 5–9y: 118.3 a; 10–17y: 62.8 a; 18–49: 115.7 a; > 50: 69.2 a |
F, E | < 12m: 3,533.0 a; 12–23m: 6,419.1 a; 24–35m: 3,888.3 a; 36–47m: 3,771.7 a; 48–59m: 1,788.9 a; 5–9y: 374.5 a; 10–17y: 216.1 a; 18–49y: 325.7 a; > 50y: 249.5 a All ages: 1,428.7 |
|
| Kibera (urban), Kenya 35 | 2009 | 0–4y: 254.9 a; 5–9y: 41.8 a; 10–17y: 10.5 a; 18–49: 28.0 a; > 50: 0 a |
F, E | < 12m: 2,210.0 a; 12–23m: 1,483.8 a; 24–35m: 805.1 a; 36–47m: 636.6 a; 48–59m: 185.4 a; 5–9y: 82.5 a; 10–17y: 21.3 a; 18–49y: 62.2 a; > 50y: 0.0 a All ages: 185.5 |
|
| Lwak (rural), Kenya 35 | 2010 | NR | F, E | 1,927.3 | |
| Kibera (urban), Kenya 35 | 2010 | NR | F, E | 218.5 | |
| Lwak (rural), Kenya 35 | 2011 | NR | F, E | 608.5 | |
| Kibera (urban), Kenya 35 | 2011 | NR | F, E | 220.5 | |
| Imerintsiatosika, Madagascar 27 | 2011 | 0–1y: 77.7; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0 |
F | 0–1y: 100.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0; All ages: 9.0 |
|
| Moshi Rural District, Tanzania 27 | 2011 | 0–1y: 0.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 21.8 |
F, E | 0–1y: 0.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 28.0; All ages: 7.0 |
|
| Moshi Urban District, Tanzania 27 | 2011 | 0–1y: 336.1; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0 |
F, E | 0–1y: 427.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0; |
|
| Butajira, Ethiopia 27 | 2012 | 0–1y: 0.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0 |
NA | NR | |
| Kibera, Kenya 27 | 2012 | 0–1y: 49.2; 2–4y: 49.0; 5–14y: 17.5; > = 15y: 32.5 |
F | 0–1y: 49.0; 2–4y: 49.0; 5–14y: 17.0; > = 15y: 33.0; All ages: 32.0 |
|
| Lwak (rural), Kenya 35 | 2012 | NR | F, E | 303.3 | |
| Kibera (urban), Kenya 35 | 2012 | NR | F, E | 62.5 | |
| Isotry, Madagascar 27 | 2012 | 0–1y: 0.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0 |
F | 0–1y: 0.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0; All ages: 0.0 |
|
| Lwak (rural), Kenya 35 | 2013 | NR | F, E | 745.5 | |
| Kibera (urban), Kenya 35 | 2013 | NR | F, E | 93.4 | |
| Lwak (rural), Kenya 35 | 2014 | NR | F, E | 337.8 | |
| Kibera (urban), Kenya 35 | 2014 | NR | F, E | 87.2 | |
| Northern Africa | East Wad Medani, Sudan 27 | 2012 | 0–1y: 0.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0 |
F | 0–1y: 0.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0; All ages: 0.0 |
| Southern Africa | Pietermaritzburg, South Africa 27 | 2012 | 0–1y: 0.0; 2–4y: 0.0; 5–14y: 0.0; > = 15y: 0.0 |
NA | NR |
| Western Africa | Ashanti Region, Ghana 31 | 2007 | < 1m: 37.5; 1–11m: 862.6; 12–23m: 843.8; 24–35m: 337.5; 36–47m: 281.3; 48–60m: 56.3 |
NA | NR |
| Asante Akim (urban), Ghana 23 | 2010 | 0–1y: 380.0; 2–4y: 316.4; 5–14y: 24.2 |
F, E | 0–1y: 927.3; 2–4y: 769.9; 5–14y: 64.2; < 15y: 346.4 |
|
| Asante Akim (rural), Ghana 23 | 2010 | 0–1y: 966.2; 2–4y: 1,150.2; 5–14y: 46.6 |
F, E | 0–1y: 2353.3; 2–4y: 2,808.4; 5–14y: 123.4; < 15y: 1,012.1 |
|
| Asante Akim, Ghana 27 | 2010 | 0–1y: 710.8; 2–4y: 782.3; 5–14y: 55.8 |
F | 0–1y: 1,733.0; 2–4y: 1,908.0; 5–14y: 147.0; < 15y: 742.0 |
|
| Bandim, Guinea-Bissau 27 | 2011 | 0–1y: 96.2 2–4y: 25.9 5–14y: 18.0 > = 15y: 0.0 |
F | 0–1y: 291.0; 2–4y: 53.0; 5–14y: 53.0; > = 15y: 0.0; All ages: 37.0 |
|
| Pikine, Senegal 27 | 2011 | 0–1y: 0.0 2–4y: 0.0 5–14y: 2.3 > = 15y: 3.6 |
NA | NR | |
| Nioko II, Burkina Faso 27 | 2012 | 0–1y: 143.1; 2–4y: 143.1; 5–14y: 61.4; > = 15y: 9.4 |
F | 0–1y: 753.0; 2–4y: 753.0; 5–14y: 236.0; > = 15y: 35.0; All ages: 237.0 |
|
| Polesgo, Burkina Faso 27 | 2012 | 0–1y: 107.6; 2–4y: 201.6; 5–14y: 0.0 > = 15y: 20.3 |
F | 0–1y: 431.0; 2–4y: 630.0; 5–14y: 0.0; > = 15y: 54.0; All ages: 144.0 |
Multipliers: F = Facility coverage: eligible participants not seeking care at facility; E = Enrollment: eligible participants did not have a blood culture collected; NA: Not applicable; NR: Not reported; m: month; y: year; PYO: person-years observed.
Age stratified adjusted incidence data is for entire surveillance period from 2009 through 2014. Age stratified adjusted incidence was not reported for each individual year.
Three studies provided incidence by individual serovars in addition to iNTS incidence overall.24,26,34 Among these three studies, there were 20 estimates of incidence for Salmonella Typhimurium, 20 estimates of Salmonella Enteritidis (Supplement Figs. S1 and S2, respectively), and one estimate of Salmonella Heidelberg. The median (range) incidence of Salmonella Typhimurium was 68.8 (3.1–204.7) and 7.0 (0.8–55.7) for Salmonella Enteritidis. The single incidence estimate for Salmonella Heidelberg was 0.4 from Kenya in 2006.34
Prevalence of non-typhoidal Salmonella serovars
Five (38.5%) of the 13 studies provided data on prevalence of iNTS serovars among isolates from normally sterile sites; all were studies located in Africa. Among the five studies, 8,726 (77.4%) of 11,271 iNTS were Salmonella Typhimurium, followed by 1919 (17.0%) Salmonella Enteritidis, and 10 (0.1%) Salmonella Dublin. The remaining 14 serovars each accounted for <0.1% of NTS reported (Table 3). For 588 (5.2%) isolates, serotyping was performed but serovars could not be determined, or the authors provided only the most common serovars and not all serovars that were identified.
Table 3.
Prevalence of non-typhoidal Salmonella enterica serovars in Africa, 1998 through 2016.
| Salmonella enterica serovar | Cases | Proportion of isolates,% |
|---|---|---|
| Typhimurium | 8,726 | 77.4 |
| Enteritidis | 1,919 | 17.0 |
| Dublin | 10 | 0.1 |
| Heidelberg | 5 | < 0.1 |
| Choleraesuis | 4 | < 0.1 |
| Infantis | 4 | < 0.1 |
| Virchow | 3 | < 0.1 |
| Derby | 2 | < 0.1 |
| Panama | 2 | < 0.1 |
| Bovismorbificans | 1 | < 0.1 |
| Hadar | 1 | < 0.1 |
| Isangi | 1 | < 0.1 |
| Kibusi | 1 | < 0.1 |
| Senegal | 1 | < 0.1 |
| Stanleyville | 1 | < 0.1 |
| Umbilo | 1 | < 0.1 |
| Urbana | 1 | < 0.1 |
| Other Salmonella * | 588 | 5.2 |
| Total | 11,271 | 100.0 |
Serotyping performed but could not identify serovars or authors provided only most common serovars and did not describe all serovars that were serotyped.
Discussion
Our systematic review of iNTS incidence demonstrated varying levels of incidence between countries, locations in close proximity, and consecutive years in the same location, displaying considerable heterogeneity in both place and time. Similar heterogeneity of incidence has also been observed for typhoid fever.36 Incidence in Africa was significantly higher than in Asia, and no data were available from other regions. Serovars isolated were predominately Salmonella Typhimurium and Enteritidis, accounting for more than 90% of all iNTS that were serotyped.
The pooled incidence estimate of 51 per 100,000 per year in Africa in our review was similar to one provided by a 2017 estimate4 but substantially lower to an estimate for 2010.13 Lower recent incidence estimates may reflect improvements in host risk factors for iNTS disease, including expanded coverage of HIV prevention and care services, and declining malaria incidence in Africa.37,38 It is possible that variations in the prevalence of host risk factors for iNTS disease such as HIV, malaria, and malnutrition, and presence or absence of key serovars and sequence types may contribute to the heterogeneity between and within each review. Additionally, the methods between reviews varied, with lower-quality national surveillance data used in previous reviews, as well as the application of differing extrapolation methods to estimate incidence in areas that lacked data.
Among studies stratifying iNTS incidence by age, children aged <5 years regularly had incidence rates higher than older children and adults, and incidence was highest among infants. Infants and younger children represent a key target for iNTS vaccines. However, data in eligible incidence studies lacked sufficient resolution to examine differences in incidence by narrower age bands during the first 12 months of life.
Since Salmonella Typhimurium and Enteritidis accounted for >90% of iNTS infections, a bivalent vaccine would address the majority of NTS serovars causing invasive disease. However, it is known that some NTS serovars demonstrate geographic localization and that given the small number of eligible studies, we cannot rule out the presence of unstudied locations where otherwise rare serovars predominate. Antimicrobial resistance in NTS causing invasive disease has recently been reviewed by others.9 The prevalence of antimicrobial resistance to widely used antimicrobial classes in NTS causing invasive disease further underscores the need for prevention interventions.
Our review had several limitations. First, there were substantial gaps in data available from the published literature. Robust incidence estimates data were not available for the majority of countries in Africa. Countries and areas with known high prevalence of HIV, malaria incidence, and malnutrition lacking data on iNTS are potential high priority targets for future studies. Hospital-based prevalence studies of NTS BSI could be used as a lower-cost alternative to more resource intensive population-based incidence studies to gain insights into the role of NTS as a cause of bacteremia in unstudied locations.39 Second, available data were subject to moderate or high risk of bias. Varying types and numbers of multipliers were used across studies. There is a need to establish a standard design for hybrid surveillance studies. We also observed substantial heterogeneity in our meta-analyses. Lastly, since high typhoidal and non-typhoidal Salmonella invasive disease incidence occur uncommonly at the same site,5,6 the inclusion of data from the Typhoid Fever Surveillance in Africa Program (TSAP) that targeted areas with known occurence of typhoid fever27 may have biased out review towards sites with less iNTS disease.
We found that iNTS incidence varies by region, location, age group, and over time. While a large number of Salmonella enterica serovars cause iNTS, Salmonella Typhimurium and Enteritidis predominate. Concerted efforts are needed to address the limited high-quality data available on iNTS disease incidence. Increased sentinel site surveillance, as well as prevalence studies, are needed to better understand iNTS epidemiology. Bivalent vaccines targeting Salmonella Typhimurium and Enteritidis have the potential to prevent considerable iNTS disease among African infants and children.
Supplementary Material
Role of the funder
The funders of the study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Financial disclosure
This project has received funding from the European Union’s Horizon 2020 research and innovation program under the project vacc-iNTS, grant agreement number 815439. CSM and JAC also received support from Bill & Melinda Gates Foundation (BMGF) grant OPP1151153. JAC received support from received support from BMGF (grant numbers OPP1125993 and OPP1158210), the US National Institutes of Health (Grant No. R01AI121378), and the New Zealand Health Research Council through the e-ASIA Joint Research Program (Grant No. 16/697).
Footnotes
Vacc-iNTS consortium collaborators
Laura B. Martin, GSK Vaccines Institute for Global Health; Gianluca Breghi, Fondazione Achille Sclavo; Rocio Canals, GSK Vaccines Institute for Global Health; Melita A Gordon, University of Liverpool; Brama Hanumunthadu, University of Oxford; Jan Jacobs, Institute of Tropical Medicine Antwerp; Samuel Kariuki, Kenya Medical Research Institute (KEMRI); Jong-Hoon Kim, International Vaccine Institute; Stefano Malvolti, MM Global Health Consulting; Carsten Mantel, MM Global Health Consulting; Florian Marks, University of Cambridge; Donata Medaglini, Università di Siena and Sclavo Vaccines Association; Vittal Mogasale, International Vaccine Institute; Esther Muthumbi, KEMRI-Wellcome Trust Research Programme; Robert Onsare, KEMRI; Ellis Owusu-Dabo, Kwame Nkrumah University of Science and Technology Kumasi; Michael Owusu, Kwame Nkrumah University of Science and Technology Kumasi; Tonney Niyrenda, University of Malawi; Maheshi Ramasamy, University of Oxford; J. Anthony Scott, KEMRI-Wellcome Trust Research Programme; Bassiahi Abdramane Soura, University of Ouagadougou; Tiziana Spadafina, Sclavo Vaccines Association; Bieke Tack, Institute of Tropical Medicine Antwerp.
Declaration of Competing Interest
None
Supplementary materials
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.jinf.2021.06.029.
References
- 1.Roth GA, Abate D, Abate KH, Abay SM, Abbafati C, Abbasi N, et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: a systematic analysis for the global burden of disease study 2017. Lancet 2018; 392 (10159):1736–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Crump JA, Sjölund-Karlsson M, Gordon MA, Parry CM. Epidemiology, clinical presentation, laboratory diagnosis, antimicrobial resistance, and antimicrobial management of invasive Salmonella infections. Clin Microbiol Rev 2015; 28 (4):901–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gilchrist J, MacLennan C. Invasive nontyphoidal Salmonella disease in Africa. Ecosal Plus 2019; 8 (2):1–23. [DOI] [PubMed] [Google Scholar]
- 4.Stanaway JD, Parisi A, Sarkar K, Blacker BF, Reiner RC, Hay SI, et al. The global burden of non-typhoidal Salmonella invasive disease: a systematic analysis for the global burden of disease study 2017. Lancet Infect Dis 2019; 19 (12):1312–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Marchello CS, Dale AP, Pisharody S, Rubach MP, Crump JA. A systematic review and meta-analysis of the prevalence of community-onset bloodstream infections among hospitalized patients in Africa and Asia. Antimicrob Agents Chemother 2019; 64 (1):e01974–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Reddy EA, Shaw AV, Crump JA. Community-acquired bloodstream infections in Africa: a systematic review and meta-analysis. Lancet Infect Dis 2010; 10 (6):417–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Park SE, Pak GD, Aaby P, Adu-Sarkodie Y, Ali M, Aseffa A, et al. The relationship between invasive nontyphoidal Salmonella disease, other bacterial bloodstream infections, and malaria in sub-Saharan Africa. Clin Infect Dis 2016; 62 (1):S23–31 suppl_. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Crump JA, Heyderman RS. A perspective on invasive Salmonella disease in Africa. Clin Infect Dis 2015; 61 (suppl_4):S235–SS40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tack B, Vanaenrode J, Verbakel JY, Toelen J, Jacobs J. Invasive non-typhoidal Salmonella infections in sub-Saharan Africa: a systematic review on antimicrobial resistance and treatment. BMC Med 2020; 18 (1):212 Epub 2020/07/18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Feasey N, Cain A, Msefula C, Pickard D, Alaerts M, Aslett M, et al. Drug resistance in Salmonella enterica ser. Typhimurium bloodstream infection, Malawi. Emerg Infect Dis J 2014; 20 (11):1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Van Puyvelde S, Pickard D, Vandelannoote K, Heinz E, Barbé B, de Block T, et al. An African Salmonella Typhimurium ST313 sublineage with extensive drug-resistance and signatures of host adaptation. Nat Commun 2019; 10 (1):4280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.MacLennan CA, Martin LB, Micoli F. Vaccines against invasive Salmonella disease: current status and future directions. Hum Vaccin Immunother 2014; 10 (6):1478–93 Epub 2014/05/09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ao TT, Feasey NA, Gordon MA, Keddy KH, Angulo FJ, Crump JA. Global burden of invasive nontyphoidal Salmonella disease, 2010. Emerg Infect Dis 2015; 21 (6):941–9 Epub 2015/04/11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kirk MD, Pires SM, Black RE, Caipo M, Crump JA, Devleesschauwer B, et al. World health organization estimates of the global and regional disease burden of 22 foodborne bacterial, protozoal, and viral diseases, 2010: a data synthesis. PLoS Med 2015; 12 (12):e1001921 Epub 2015/12/04. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Moher D, Liberati A, Tetzlaff J, Altman DG, Group P. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 20 09; 6 (7):e10 0 0 097 Epub 2009/07/22. [PMC free article] [PubMed] [Google Scholar]
- 16.Andrews JR, Barkume C, Yu AT, Saha SK, Qamar FN, Garrett D, et al. Integrating facility-based surveillance with healthcare utilization surveys to estimate enteric fever incidence: methods and challenges. J Infect Dis 2018; 218 (suppl_4):S268–SS76 Epub 2018/09/06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Crump JA, Youssef FG, Luby SP, Wasfy MO, Rangel JM, Taalat M, et al. Estimating the incidence of typhoid fever and other febrile illnesses in developing countries. Emerg Infect Dis 2003; 9 (5):539–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ouzzani M, Hammady H, Fedorowicz Z, Elmagarmid A. Rayyan-a web and mobile app for systematic reviews. Syst Rev 2016; 5 (1):210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Munn Z, Moola S, Lisy K, Riitano D, Tufanaru C. Methodological guidance for systematic reviews of observational epidemiological studies reporting prevalence and cumulative incidence data. JBI Evid Implement 2015; 13 (3). [DOI] [PubMed] [Google Scholar]
- 20.Viswanathan M, Patnode CD, Berkman ND, Bass EB, Chang S, Hartling L, et al. Recommendations for assessing the risk of bias in systematic reviews of health-care interventions. J Clin Epidemiol 2018; 97:26–34 Epub 2017/12/19. [DOI] [PubMed] [Google Scholar]
- 21.Sterne JAC, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016; 355:i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.EpiGear International Pty L. MetaXL. 2016. [cited 2020 1 October]; Available from: https://www.epigear.com/index_files/metaxl.html.
- 23.Cruz Espinoza LM, Nichols C, Adu-Sarkodie Y, Al-Emran HM, Baker S, Clemens JD, et al. Variations of invasive Salmonella infections by population size in Asante Akim North Municipal, Ghana. Clin Infect Dis 2016; 62:S17–22 Suppl 1Epub 2016/03/05. [DOI] [PubMed] [Google Scholar]
- 24.Feasey NA, Masesa C, Jassi C, Faragher EB, Mallewa J, Mallewa M, et al. Three epidemics of invasive multidrug-resistant Salmonella bloodstream infection in Blantyre, Malawi, 1998–2014. Clin Infect Dis 2015; 61:S363–71 Suppl 4Epub 2015/10/10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Khan MI, Ochiai RL, von Seidlein L, Dong B, Bhattacharya SK, Agtini MD, et al. Non-typhoidal Salmonella rates in febrile children at sites in five Asian countries. Trop Med Int Health 2010; 15 (8):960–3 Epub 2010/06/16. [DOI] [PubMed] [Google Scholar]
- 26.Mandomando I, Bassat Q, Sigauque B, Massora S, Quinto L, Acacio S, et al. Invasive Salmonella infections among children from rural Mozambique, 2001–2014. Clin Infect Dis 2015; 61:S339 Suppl 4–45Epub 2015/10/10. [DOI] [PubMed] [Google Scholar]
- 27.Marks F, von Kalckreuth V, Aaby P, Adu-Sarkodie Y, El Tayeb MA, Ali M, et al. Incidence of invasive Salmonella disease in sub-Saharan Africa: a multicentre population-based surveillance study. Lancet Glob Health 2017; 5 (3):e310–ee23 Epub 2017/02/15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mayanja BN, Todd J, Hughes P, Van der Paal L, Mugisha JO, Atuhumuza E, et al. Septicaemia in a population-based HIV clinical cohort in rural Uganda, 1996–2007: incidence, aetiology, antimicrobial drug resistance and impact of antiretroviral therapy. Trop Med Int Health 2010; 15 (6):697–705 Epub 2010/04/22. [DOI] [PubMed] [Google Scholar]
- 29.Mtove G, Amos B, Nadjm B, Hendriksen IC, Dondorp AM, Mwambuli A, et al. Decreasing incidence of severe malaria and community-acquired bacteraemia among hospitalized children in Muheza, north-eastern Tanzania, 2006–2010. Malar J 2011; 10:320 Epub 2011/10/28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Muthumbi E, Morpeth SC, Ooko M, Mwanzu A, Mwarumba S, Mturi N, et al. Invasive salmonellosis in Kilifi, Kenya. Clin Infect Dis 2015; 61:S290–301 Suppl 4Epub 2015/10/10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nielsen MV, Sarpong N, Krumkamp R, Dekker D, Loag W, Amemasor S, et al. Incidence and characteristics of bacteremia among children in rural Ghana. PLoS ONE 2012; 7 (9):e44063 Epub 2012/09/13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Oneko M, Kariuki S, Muturi-Kioi V, Otieno K, Otieno VO, Williamson JM, et al. Emergence of community-acquired, Multidrug-resistant invasive nontyphoidal Salmonella disease in rural western Kenya, 2009–2013. Clin Infect Dis 2015; 61:S310–16 Suppl 4Epub 2015/10/10. [DOI] [PubMed] [Google Scholar]
- 33.Scott JA, Berkley JA, Mwangi I, Ochola L, Uyoga S, Macharia A, et al. Relation between falciparum malaria and bacteraemia in Kenyan children: a population-based, case-control study and a longitudinal study. Lancet 2011; 378 (9799):1316–23 Epub 2011/09/10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tabu C, Breiman RF, Ochieng B, Aura B, Cosmas L, Audi A, et al. Differing burden and epidemiology of non-Typhi Salmonella bacteremia in rural and urban Kenya, 20 06–20 09. PLoS ONE 2012; 7 (2):e31237 Epub 2012/03/01. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Verani JR, Toroitich S, Auko J, Kiplang’at S, Cosmas L, Audi A, et al. Burden of invasive nontyphoidal Salmonella disease in a rural and urban site in Kenya, 2009–2014. Clin Infect Dis 2015; 61:S302 Suppl 4–9Epub 2015/10/10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Marchello CS, Hong CY, Crump JA. Global typhoid fever incidence: a systematic review and meta-analysis. Clin Infect Dis 2019; 68 (2):S105–SS16 Supplement_. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.World Health Organization (WHO). 2018. World malaria report 2018. ISBN: 978–92-4–156565-3. Available from: https://apps.who.int/iris/bitstream/handle/10665/275867/9789241565653-eng.pdf. Accessed 8 Sep 2021.
- 38.Murray CJL, Ortblad KF, Duber HC, Naghavi M, Dicker D, Dandona L, et al. Global, regional, and national incidence and mortality for HIV, tuberculosis, and malaria during 1990–2013: a systematic analysis for the global burden of disease study 2013. Lancet 2014; 384 (9947):1005–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Marchello CS, Dale AP, Pisharody S, Crump JA. Using hospital-based studies of community-onset bloodstream infections to make inferences about typhoid fever incidence. Trop Med Int Health 2019; 24 (12):1369–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Marchello CS, Birkhold M, Crump JA Vacc-iNTS Consortium Collaborators Complications and mortality of non-typhoidal Salmonella invasive disease: a global systematic review and meta-analysis. Lancet Infect Dis 2021. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


