Abstract
Recognition of β-cell antigens by autoreactive T cells is a critical step in the initiation of autoimmune type1 diabetes. A complete protection from diabetes development in NOD mice harboring a point mutation in the insulin B-chain 9–23 epitope points to a dominant role of insulin in diabetogenesis. Generation of NOD mice lacking the chromogranin A protein (NOD.ChgA−/−) completely nullified the autoreactivity of the BDC2.5 T cell and conferred protection from diabetes onset. These results raised the issue concerning the dominant antigen that drives the autoimmune process. Here we revisited the NOD.ChgA−/− mice and found that their lack of diabetes development may not be solely explained by the absence of chromogranin A reactivity. NOD.ChgA−/− mice displayed reduced presentation of insulin peptides in the islets and periphery, which corresponded to impaired T-cell priming. Diabetes development in these mice was restored by antibody treatment targeting regulatory T cells or inhibiting transforming growth factor-β and programmed death-1 pathways. Therefore, the global deficiency of chromogranin A impairs recognition of the major diabetogenic antigen insulin, leading to broadly impaired autoimmune responses controlled by multiple regulatory mechanisms.
Introduction
Type 1 diabetes (T1D) is a chronic autoimmune disease that results from progressive destruction of β-cells in the pancreatic islets. Recognition of specific autoantigens by T cells is crucial for the initiation of the disease. Several islet-specific antigens have been identified as the targets of autoimmunity in humans with T1D and NOD mice (1). Major progress was achieved with the generation of a NOD mouse harboring a point mutation in the insulin B-chain 9–23 (InsB:9–23) epitope (2). This mutation nullified the immunogenicity of insulin without influencing its biological activity. The resulting mouse was completely resistant to T1D and lacked any signs of autoimmunity in the islets (2). Subsequent studies indicated that the earliest T cells that entered the islet were directed to the InsB:9–23 peptide (3). T cells specific to InsB:9–23 transferred diabetes in mice (4), and in humans, these T cells were detected in the inflamed islets (5,6). Recently, by an unbiased screen, we found that MHC class II (MHC-II)-bound peptides derived from insulin were the major antigens giving rise to T-cell reactivity in the islets and periphery (pancreatic lymph nodes [panLNs] and spleen) (7). These findings establish that insulin is a primary autoantigen critical for initiating diabetogenesis in NOD mice.
Other antigens, such as GAD65, islet-specific glucose-6-phosphatase catalytic subunit-related protein (IGRP), islet amyloid polypeptide (IAPP), islet antigen 2 (IA-2), and zinc transporter 8 (ZnT8), have been identified, but these seem to have a lesser impact on diabetogenesis (1,8–10). NOD mice lacking GAD65, IA-2, IGRP, and IAPP did not show obvious changes in the development of diabetes (1,8–10). Therefore, although T-cell responses to these antigens are involved in diabetes development, they may not be the major drivers required for the initiation of the autoimmune process. Our attention was called to chromogranin A (ChgA) by the studies from the Haskins laboratory (11), which indicated that the absence of diabetes development was associated with a lack of presentation of ChgA peptides. A signature diabetogenic CD4 T cell, BDC2.5, responded weakly to the naturally occurring ChgA cleavage peptide WE14 (12). Recently, BDC2.5 T cells were shown to recognize a hybrid peptide generated by the fusion of the ChgA WE14 N-terminal sequence and insulin C-peptide, representing a novel mechanism underlying T-cell autoreactivity (13).
Therefore, the important question is whether the protection conferred by deletion of the ChgA gene is only due to the lack of ChgA reactivity. ChgA has diverse biological functions. As a granulogenic protein, ChgA is expressed in many endocrine and neuroendocrine tissues, including chromaffin cells of the adrenal gland, enterochromaffin cells of the gut, and β-cells of the islets (14,15). ChgA is involved in the initiation and regulation of dense-core granule biogenesis and sequestration of the peptide hormone in the neuroendocrine and endocrine cells (15,16). Moreover, ChgA is synthesized as a proprotein, giving rise to several biologically active peptides that have diverse functions (16). Genetic ablation of ChgA in mice resulted in reduced size and number of adrenal medullary chromaffin granules, resulting in altered catecholamines levels and high blood pressure (17,18). In the pancreas, loss of ChgA affected islet volume, composition, distribution, and nuclear size of islet cell types (19). Within the secretory granule, peptides derived from ChgA, such as betagranin and pancreastatin, were shown to regulate insulin secretion (20,21). A consistent reduction in the plasma insulin levels was observed in B6.ChgA−/− mice upon glucose challenge, indicating a defect in insulin production and secretion (22,23). Although B6.ChgA−/− mice maintained a euglycemic state at steady state, this impairment of insulin production/secretion may influence the presentation of insulin peptides. Many of the immunogenic insulin peptides were shown to be released from the islets, and these sensitized peripheral lymphoid tissues, resulting in T-cell priming (24,25). Therefore, the impact of ChgA on insulin presentation should be considered in the context of diabetes resistance of the NOD.ChgA−/− mice.
In this report, we evaluated the NOD.ChgA−/− mice and found a reduced level of insulin presentation and impaired priming of insulin-reactive T cells. We hypothesized that such a broad defect of autoreactivity may be subjected to immunoregulation, resulting in the protection of diabetes. Indeed, targeting regulatory T cells or inhibiting programmed death-1 (PD-1) and transforming growth factor-β (TGF-β) pathways resulted in the development of diabetes in NOD.ChgA−/− mice. This study indicates that in addition to the lack of CD4 T-cell reactivity to ChgA, other factors may contribute to the profound phenotype of the NOD.ChgA−/− mice because of the broad biological functions of ChgA.
Research Design and Methods
Mice
NOD.ChgA−/− mice were a gift from Kathryn Haskins, University of Colorado Anschutz School of Medicine (Aurora, CO). NOD/ShiLtJ (NOD) mice were originally obtained from The Jackson Laboratory. The mice were bred, maintained, and used for experiments under specific pathogen-free conditions in our animal facility in accordance with the Division of Comparative Medicine, Washington University School of Medicine (St. Louis, MO).
Antibody Treatment and Diabetes Monitoring
For depletion of regulatory T cells, female NOD or NOD.ChgA−/− mice were given two injections (0.5 mg per mouse i.p.) of rat anti-mouse CD25 monoclonal antibody, clone PC61 (Leinco Technologies, Fenton, MO). The injections were given at 14 and 21 days of age, and mice were then followed for diabetes development. For PD-1 inhibition and TGF-β neutralization, NOD or NOD.ChgA−/− mice were injected i.p. with 250 μg anti–PD-1(RMP1-14) and 500 μg anti–TGF-β (1D11.16.8) antibodies either alone or in combination on days 0, 3, 6, and 9. All the antibodies were diluted in sterile PBS prior to injection. Urine glucose levels in mice were monitored every day following injection until days 30. Mice were considered diabetic following two consecutive readings >250 mg/dL.
Islet Isolation
NOD or NOD.ChgA−/− mice were killed, and the peritoneal cavity was dissected to expose the common bile duct. Under a dissection microscope, the bile duct leading into the duodenum was clamped. Through the common bile duct, 5 mL type XI collagenase (0.4 mg/mL; Sigma-Aldrich) was injected to inflate the pancreas. The inflated pancreas was carefully removed and digested at 37°C for 12 min. Following digestion, crude islets were washed and collected into a petri dish. Islets were handpicked under a microscope.
Flow Cytometry
Islets from NOD or NOD.ChgA−/− mice were dispersed into a single-cell suspension using a nonenzymatic cell dissociation solution (Sigma-Aldrich) for 3 min at 37°C. Cells were incubated in a buffer containing the FcR blocking antibody (2.4G2) for 15 min at 4°C. Surface staining was performed with fluorescently labeled antibodies (1:200 [vol/vol]) by incubating at 4°C for 30 min. Cells were washed twice and analyzed by BD FACSCanto II Cell Analyzer (BD Biosciences). For intracellular tumor necrosis factor-α (TNF-α) and interleukin-1β (IL-1β) staining, single-cell suspension of islet cells was incubated at 37°C with 10 μg/mL brefeldin A for 4 h. Following incubation, cells were washed twice, and surface staining was performed as described above. Following surface staining, cells were fixed (Cytofix/Cytoperm; BD Biosciences) for 30 min at 4°C. Cells were then washed twice with the perm buffer (Cytofix/Cytoperm; BD Biosciences) and incubated with TNF-α and IL-1β antibodies for 30 min at 4°C. Data were analyzed using FlowJo 10.0 software (TreeStar).
Tetramer Staining
Single-cell suspensions of pooled secondary lymphoid tissues (spleen, pancreatic, mesenteric, inguinal, and axillary LNs) from individual NOD or NOD.ChgA−/− mice were prepared, followed by removal of the red blood cells. Tetramer staining was then performed as described previously (3). Briefly, cells were washed twice with 1× PBS and stained with allophycocyanin-labeled I-Ag7/InsB12–20 tetramer for 1 h at room temperature (RT) at a concentration of 10 μg/mL. Cells were washed and then labeled with anti-allophycocyanin microbeads and purified using a MACS column (Miltenyi Biotec). Cells bound to the column were then eluted and washed twice with FACS buffer, and surface staining was performed using fluorescently conjugated antibodies as described above. Following surface staining, cells were fixed using Foxp3/transcription buffer (eBioscience) for 30 min at 4°C, washed twice with 1× permeabilization buffer (eBioscience), and then stained with fluorescently conjugated Foxp3 antibody for 30 min at RT. Cells were washed twice with 1× permeabilization buffer and analyzed by BD FACSCanto II Cell Analyzer (BD Biosciences).
Isolation of Peripheral Blood Mononuclear Cells From Mice
NOD or NOD.ChgA−/− mice were fasted overnight and injected i.p. with glucose (4 g/kg body wt). Mice were anesthetized 30 min after the glucose challenge, and blood was drawn from the heart. Blood was diluted by an equal volume of DMEM media containing 10% FBS and then underlayered by an equal volume of Histopaque-1077 (catalog number H8889; Sigma-Aldrich) in a 5-mL conical tube. Samples were spun for 35 min at 400g at RT, and the buffy coat from the interface was collected. The cells were then washed twice and subsequently used for antigen presentation assay.
Antigen Presentation Assay
For islet presentation assays, islets from 2–3 NOD or NOD.ChgA−/− mice were pooled and dispersed nonenzymatically. Together with 5 × 104 T-cell hybridoma cells per well, 5 × 104 islet cells per well were plated in DMEM containing 10% FBS in a 96-well plate. After 24 h, supernatants were collected, and the levels of IL-2 in the supernatant were measured by culturing with CTLL-2, an IL-2–dependent cell line. Proliferation of CTLL-2 was measured by incorporation 3[H] thymidine.
For presentation by white blood cells, isolated white blood cells were cultured overnight with the T-cell hybridomas in a 96-well plate. In some wells, anti–I-Ag7 antibody (1 μg/mL) was added as a control for I-Ag7–specific response. The T-cell response was probed by IL-2 production by the CTLL-2 cell line as described above.
ELISPOT Assay
PanLNs from female NOD or NOD.ChgA−/− mice were harvested and pooled. Single-cell suspensions were made by digestion of panLNs with Liberase (Roche) (125 μg/mL) and DNAse (Roche) (50 μg/mL). Cells were cultured in DMEM with 10% FBS at a concentration of 2 × 106/mL culture media and supplemented with 20 units/mL IL-2 and 1 μmol/L InsB:9–23 peptide at 37°C. After 7 days, total live cells were collected from the culture using Histopaque-1119 (Sigma-Aldrich) and then subjected to the second round of amplification. In a fresh culture media supplemented with 20 units/mL IL-2 and 1 μmol/L InsB:9–23 peptide, live cells were added along with irradiated splenocytes (3,000 rads) as antigen-presenting cells (APCs) (2 × 106/mL). After 3 days, cells were harvested and then assayed for reactivity to InsB:9–23 by eliciting a recall response on either IL-2– or interferon-γ–coated 96-well multiscreen plates (MilliporeSigma) for ELISPOT assay. A total of 2 × 105 cells per well were plated with 10 μmol/L InsB:9–23 peptide, and the reactive cells (spots) were counted using ImmunoSpot software.
Serum Insulin ELISA
The capture mouse anti-insulin antibody (1 μg/mL insulin) (clone 3A6; Novus Biologicals) was used to coat 96-well ELISA plates overnight at 4°C. Plates were washed and then blocked with 5% FBS for 1 h at RT. Following blocking, serum samples were added for 2 h at 37°C. Plates were then washed, and a detection mouse anti-insulin antibody (1 μg/mL) (clone 8E2-HRP; Novus Biologicals) was added for 2 h at 4°C. Antibody binding was visualized using OptEIA TMB substrate (BD Biosciences). The reaction was stopped using a diluted phosphoric acid, and absorbance was read at 450 nm.
Pancreatic Insulin Content
Pancreata dissected from 5-week-old NOD or NOD.ChgA−/− mice were weighed and minced in a prechilled acid ethanol solution. Tissues were then homogenized, and the supernatant was collected after centrifugation. A 100-μL aliquot of the aqueous solution was neutralized with 100 μL 1M Tris (pH 7.5). Neutralized samples were diluted (1:1,000) with insulin ELISA sample diluent, and insulin content was measured by ELISA as described above. Insulin content was expressed as ng/mg of pancreatic tissue.
Statistical Analysis
The mice were age and sex matched for all the experiments. Unpaired two-tailed Student t test was used to determine the statistical significance in different biological replicates between two experimental groups. Log-rank (Mantel-Cox) test was performed to compare the diabetes incidence in mice from different treatment groups. Data are represented as mean ± SEM, and all of the P values were calculated using GraphPad PRISM 9.1.0, with the following significance: *P < 0.05, **P < 0.01, ***P < 0.001, and ****P < 0.0001.
Data and Resource Availability
The data generated and analyzed during this study are included in the article, and additional data generated are available from the corresponding author upon reasonable request. No applicable data sets/resources were generated or analyzed during the current study.
Results
ChgA Deficiency Results in Reduced Insulin Presentation
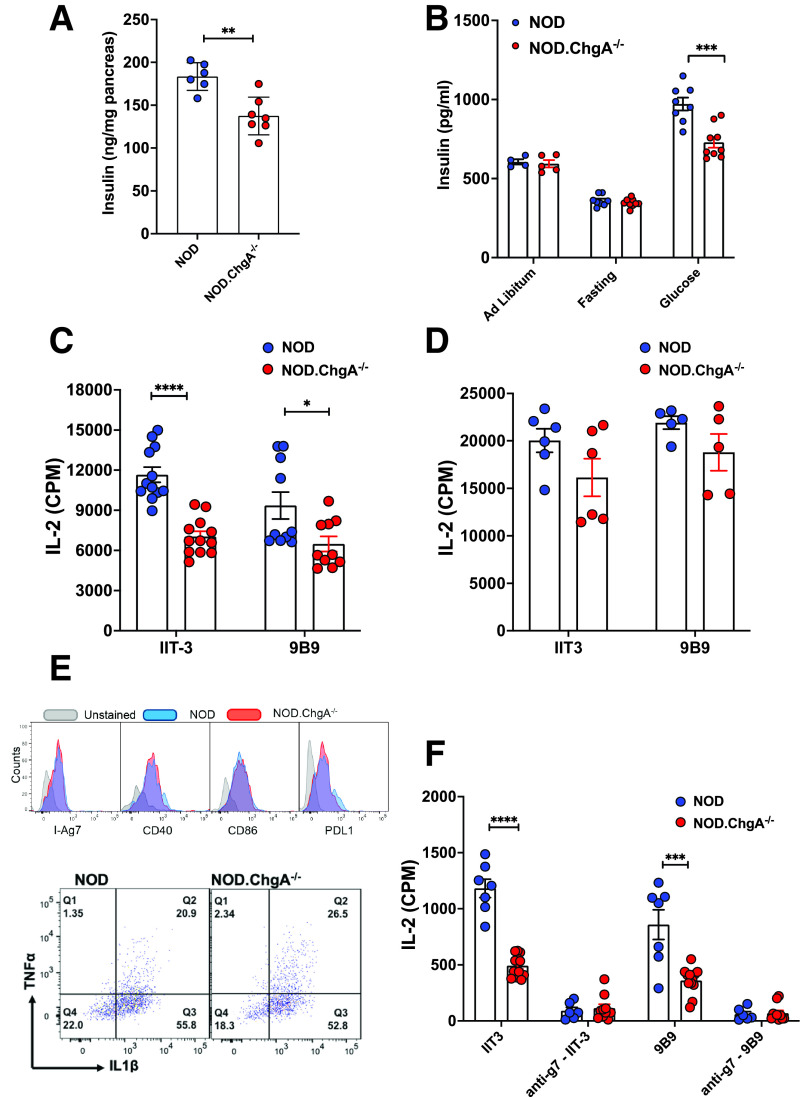
NOD.ChgA−/− mice showed a reduction in the insulin content of the pancreas relative to the wild-type (WT) NOD (NOD) mice (Fig. 1A), suggesting a defect in insulin biosynthesis. We also found a decrease in the serum insulin levels in the NOD.ChgA−/− mice upon glucose challenge (Fig. 1B). This observation may be explained by impaired insulin biosynthesis or secretion resulting from ChgA deficiency. To correlate these findings with the presentation of insulin and its degradative peptides, we assessed islet presentation of insulin epitopes. Two different sets of insulin-reactive CD4 T cells recognizing the InsB:12–20 (9B9) or InsB:13–21 (IIT-3) epitope, as described previously (4,26), were tested. We used young male mice (4–6 weeks of age) for the experiment. At this stage, the major APC in the islet was the islet-resident macrophage, while the infiltration of other APCs was minimal. The absolute numbers of the islet macrophages were comparable between the NOD.ChgA−/− and NOD mice (data not shown). We first cultured the T-cell hybridomas with dispersed islet cells from NOD or NOD.ChgA−/− mice without pulsing with exogenous antigens. This spontaneous presentation of both insulin epitopes was significantly reduced in the islets of NOD.ChgA−/− mice (Fig. 1C). Such a difference was not due to different antigen-presenting capacities, because the exogenous pulse of islet cells from NOD and NOD.ChgA−/− mice with the InsB:9–23 peptide elicited similar responses from either T-cell clone (Fig. 1D). Consistent with the previous study (11), we confirmed that presentation to the BDC2.5 T cells was absent in the islets of the NOD.ChgA−/− mice (Supplementary Fig. 1A).
Figure 1.
NOD.ChgA−/− mice display impaired insulin presentation. A: Pancreatic insulin content in 5-week-old NOD or NOD.ChgA−/− mice assessed by ELISA. Data (mean ± SEM) summarize results from three independent experiments. Each dot represents individual mouse. B: ELISA analysis of serum insulin levels in NOD or NOD.ChgA−/− mice under indicated conditions. Data (mean ± SEM) are representative of two independent experiments. Each dot represents individual mouse. C: Islet cells from 4- to 6-week-old male NOD or NOD.ChgA−/− mice are evaluated for their spontaneous presentation to two different CD4 T-cell hybridomas (IIT3 and 9B9). Islet cells (5 × 104) were cultured with the IIT3 or 9B9 T cells (5 × 104) without pulse with exogenous antigens. The T-cell responses were probed by measuring IL-2 production in the culture supernatants. Data (mean ± SEM) summarize results from 2–4 independent experiments. Each dot represents a biological replicate of islet cells pooled from 2–3 mice. D: Presentation to the IIT-3 and 9B9 T cells by islet cells from NOD or NOD.ChgA−/− mice pulsed with 10 μmol/L InsB:9–23 peptide. Data (mean ± SEM) summarize results from three independent experiments. Each dot represents a biological replicate of islet cells pooled from 2–3 mice. E: Flow cytometry analysis of I-Ag7, CD40, CD86, and PD-1 ligand (PD-L1) expression, as well as TNF-α and IL-1β production by islet macrophages (gated on live CD45+F4/80+CD11c+) from 5-week-old male NOD or NOD.ChgA−/− mice. Data are representative of two independent experiments. F: Spontaneous presentation of insulin peptides to IIT3 and 9B9 CD4 T-cell hybridomas by blood leukocytes isolated from NOD or NOD.ChgA−/− mice after glucose challenge. Data (mean ± SEM) summarize results from three independent experiments. Each dot represents a biological replicate of blood leukocytes from 1–3 mice. A–F: P values were calculated by unpaired two-tailed Student t test. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001. CPM, counts per minute.
The major MHC-II+ APC in the islets is the islet macrophage. To ascertain that the reduced insulin presentation was not caused by intrinsic differences in the islet macrophages of the NOD.ChgA−/− mice, we examined the expression level of MHC-II, costimulatory molecules (CD40 and CD80), and PD-1 ligand, as well as the production of IL-1β and TNF-α (Fig. 1E). The MHC-II expression and the differentiation state in the islet macrophages were comparable between NOD and NOD.ChgA−/− mice. Thus, the reduced insulin presentation was correlated with impaired insulin production in the islets of NOD.ChgA−/− mice.
In addition to islets, we examined the presentation of insulin peptides by blood leukocytes isolated from mice upon glucose challenge. We have recently shown that upon glucose challenge, insulin and its degradative peptides are rapidly captured by the circulating APCs and can then prime autoreactive T cells in the lymphoid tissue (24,25). Mice were fasted overnight and then i.p. injected with glucose. After 30 min, blood leukocytes were isolated and cultured with the insulin-reactive CD4 T-cell hybridomas in the absence of exogenous antigen. We found a dramatic reduction in the presentation to both IIT-3 and 9B9 T cells by the blood leukocytes in NOD.ChgA−/− mice (Fig. 1F). The presentation was completely blocked by the addition of the anti–I-Ag7 antibody, indicating that the uptake and the presentation of insulin peptides were MHC-II dependent (Fig. 1F). Thus, ChgA deficiency also resulted in a decrease of immunogenic insulin peptides that can seed the peripheral lymphoid tissues.
Reduced Priming of Insulin-Reactive T Cells in the NOD.ChgA−/− Mice
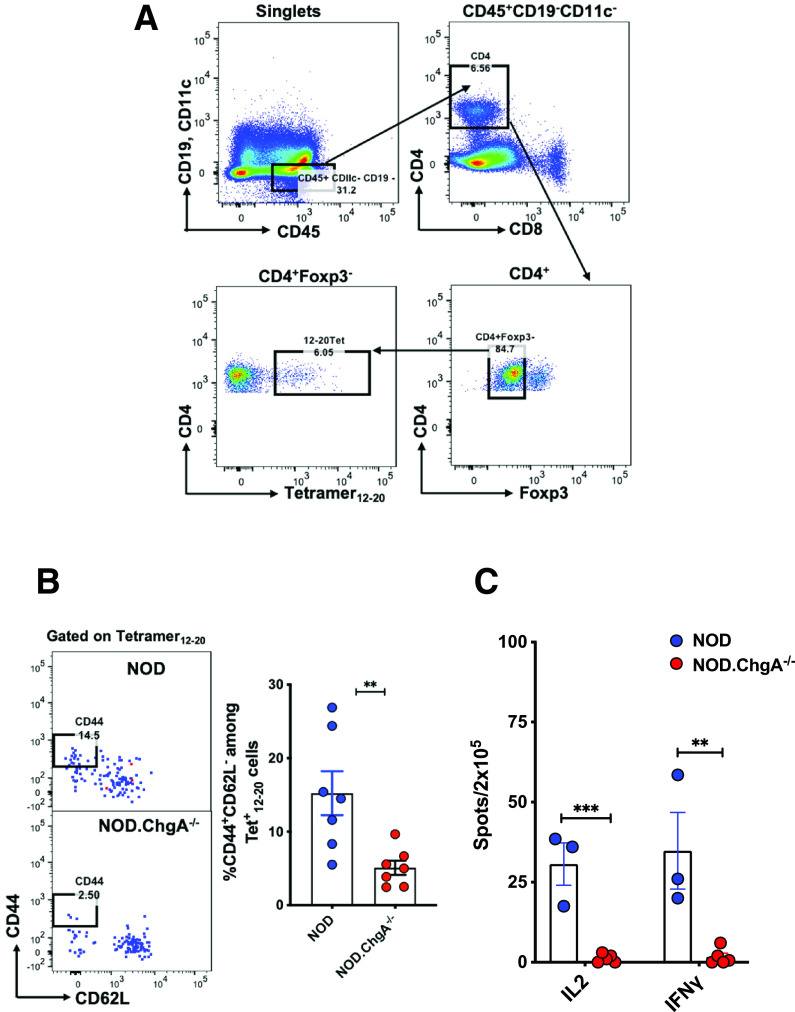
We next examined whether the reduced presentation of insulin peptides affected the activation of peripheral insulin-reactive CD4 T cells in NOD.ChgA−/− mice. The InsB:12–20 and InsB:13–21 epitopes, contained in the InsB:9–23 peptide, corresponded to two specific sets of CD4 T cells. The T cells specific to InsB:12–20 were primed in the peripheral LNs, invaded islets, and caused pathogenic responses, whereas those reactive to the InsB:13–21 epitope remained largely unresponsive (3,4,7,24,27–29). We therefore, examined the activation status of the T cells reactive to InsB:12–20 by using a specific I-Ag7–based tetramer (3). We assessed the T cells pooled from secondary lymphoid tissues and found a significant reduction in the frequency of the T cells specific to InsB:12–20 expressing CD44+CD62L− in NOD.ChgA−/− mice, indicating impaired T-cell priming (Fig. 2A and B).
Figure 2.
Defective priming of insulin-specific T cells in the secondary lymphoid tissues of NOD.ChgA−/− mice. A: FACS plots depicting the gating strategy of the InsB12–20 tetramer+ CD4 T cells in the secondary lymphoid tissue. B: Representative FACS plots showing the activation status of the InsB12–20 tetramer+ CD4 T cells in the secondary lymphoid tissues of NOD or NOD.ChgA−/− mice. Data show the percentage of the CD44+CD62L− cells among the InsB12–20 tetramer+ CD4 T cells as gated in panel A. The bar graph summarizes results from three independent experiments. Each dot represents individual mouse. C: ELISPOT assay showing IL-2 and interferon-γ (IFN-γ) production by CD4 T cells from the panLN cells of female NOD or NOD.ChgA−/− mice challenged and recalled with the InsB:9–23 peptide. Data (mean ± SEM) summarize results from three independent experiments. Each dot represents a biological replicate of panLN cells pooled from 3–5 mice. B and C: P values were calculated by unpaired two-tailed Student t test. **P < 0.01, ***P < 0.001.
Priming of naïve autoreactive T cells in the panLNs is essential for the initiation of diabetes in NOD mice (30,31). It has been shown that excision of panLNs in young NOD mice or treatment of pregnant mice with LTβR-Ig protein, which completely ablates lymph node development, protects the mice from diabetes (30,31). We next examined whether the priming of insulin-reactive T cells is affected in the panLNs of the NOD.ChgA−/− mice using a previously described protocol (7). Specifically, we cultured panLN cells isolated from NOD or NOD.ChgA−/− mice with the InsB:9–23 peptide for 10 days followed by recall with the same peptide. The T-cell responses were indicated by interferon-γ and IL-2 production measured by ELISPOT. We found no T-cell reactivity to InsB:9–23 in the panLNs of the NOD.ChgA−/− mice, indicating impaired priming of insulin-specific T cells in the draining lymph node (Fig. 2C). The impaired T-cell priming correlated with limited T-cell infiltration into the islets of the NOD.ChgA−/− mice. Consistent with the previous results (11), we found a very low level of CD45+ leukocytes (Supplementary Fig. 1B) as well as CD4 T cells (Supplementary Fig. 1C) in the islets of 8-week-old female NOD.ChgA−/− mice. This corresponded to a lack of diabetes development in these mice in our colony (Supplementary Fig. 1D).
Targeting Regulatory Mechanisms Promotes Diabetes Development in NOD.ChgA−/− Mice
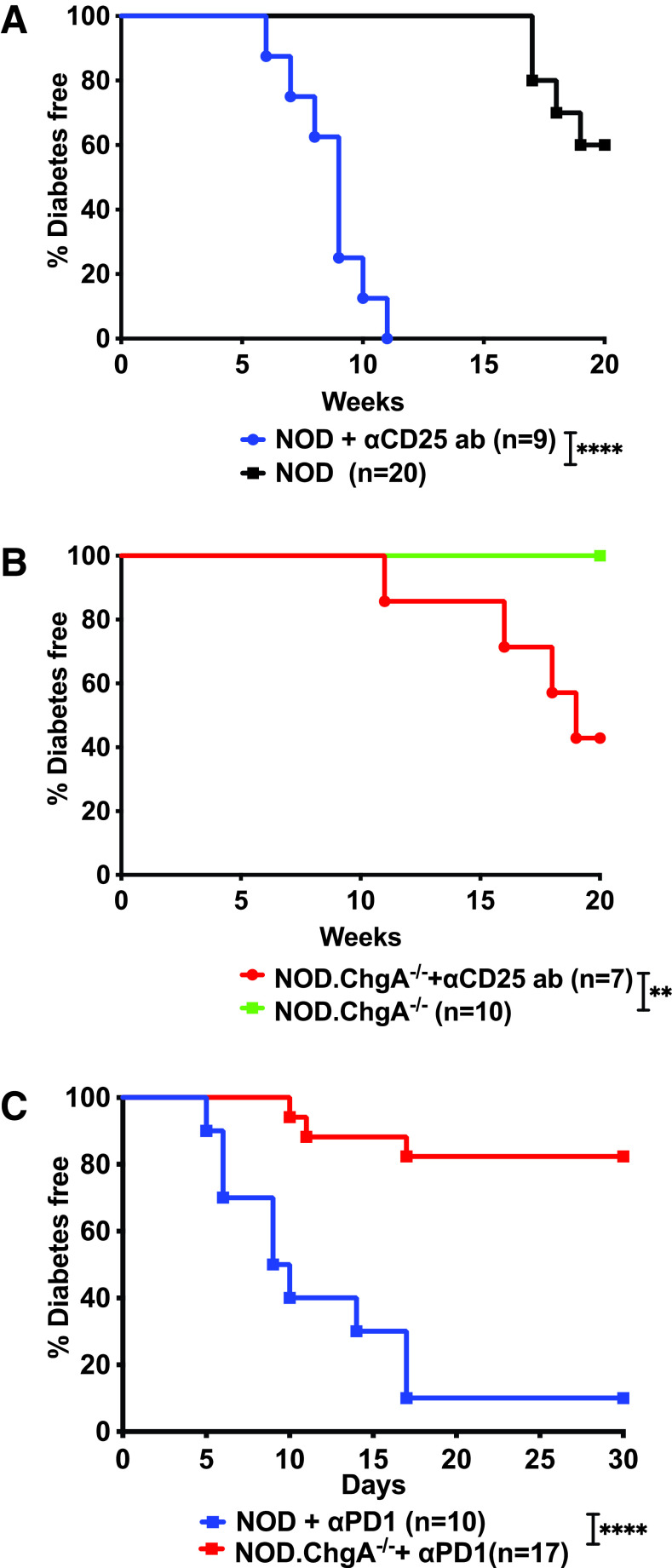
The above findings suggest that ChgA deficiency may broadly impair T-cell autoreactivity. The autoimmune process in the NOD mice is known to be controlled by diverse regulatory mechanisms. We therefore hypothesized that the lack of diabetes development in the NOD.ChgA−/− mice may be due to dominant immunoregulation. We first targeted regulatory T cells that were shown to control the onset of diabetes in NOD mice (32,33). Treatment of young NOD.ChgA−/− mice with the anti-CD25 antibody (clone PC61) rendered them susceptible to autoimmune diabetes, although the progression of diabetes was slower compared with that in WT NOD mice (Fig. 3A and B).
Figure 3.
Depletion of regulatory T cells promotes diabetes development in NOD.ChgA−/− mice. A and B: Diabetes incidence in female NOD (A) or NOD.ChgA−/− (B) mice that were either left untreated or treated with two injections of the anti-CD25 antibody (ab) at age 14 and 21 days. Data are from two independent experiments. C: Diabetes incidence in female NOD or NOD.ChgA−/− mice treated with the anti–PD-1 ab. Mice were given four injections of the anti–PD-1 ab every 3 days and then followed for diabetes. Data are from three independent experiments. P values were calculated by the log-rank (Mantel-Cox) test. **P < 0.01, ****P < 0.0001.
We next examined other immunoregulatory mechanisms. Inhibition of PD-1 has been shown to induce acute diabetes in NOD mice (34,35). We administrated the monoclonal PD-1 blockade antibody to female NOD and NOD.ChgA−/− mice (8–10 weeks of age) and found that although the NOD mice rapidly became diabetic, the NOD.ChgA−/− mice did not respond to the treatment (Fig. 3C). This phenomenon could be explained by the lack of intraislet T cells in the NOD.ChgA−/− mice, which were the major target of the anti–PD-1 antibody for triggering acute diabetes (36).
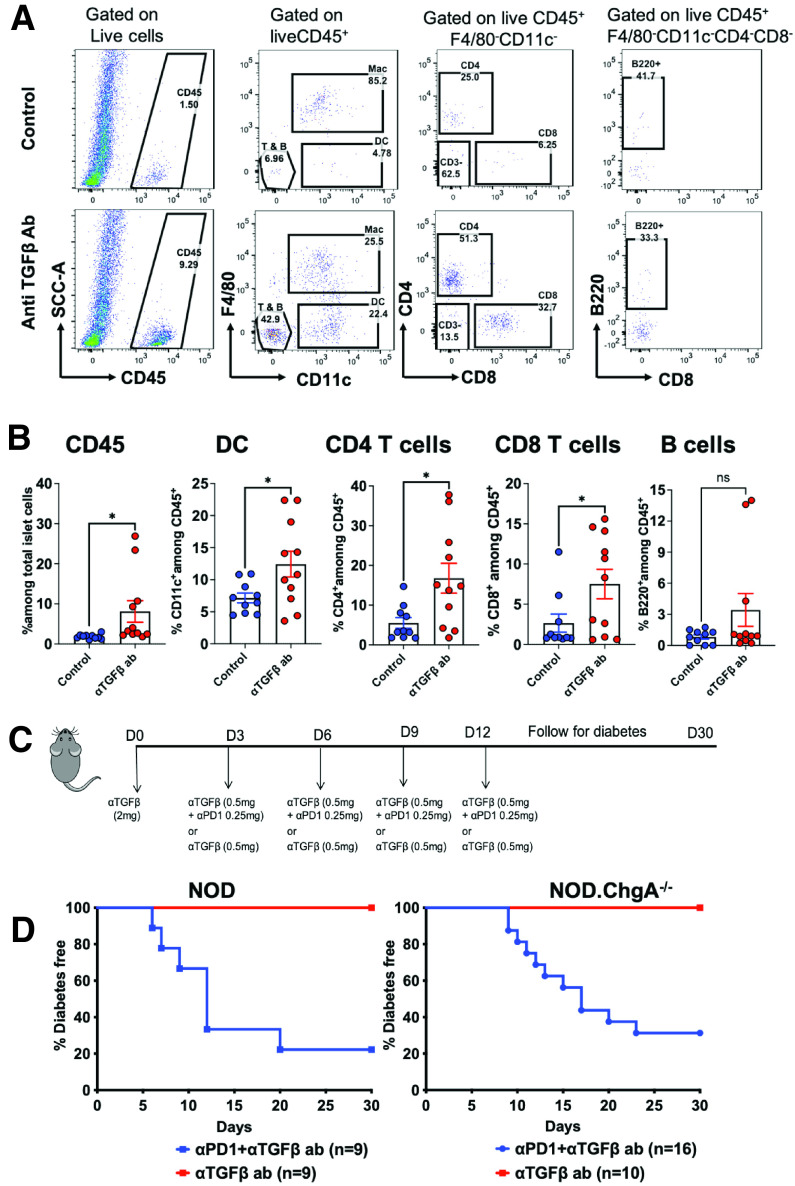
We reasoned whether the failure of T cells to enter the islets of NOD.ChgA−/− mice was due to an additional regulatory mechanism in the tissue microenvironment, as previously shown in various models of solid tumors (37,38). Selected tumors did not respond to checkpoint blockade because of a regulation mediated by TGF-β–activated stroma, which prevented T-cell entry into tumor parenchyma (37–39). In tumor-bearing mice, coinhibition of TGF-β and PD-1 ligand pathways led to increased infiltration of lymphocytes in the tumor and induced a pronounced antitumor response (37,38). To explore this possibility in NOD.ChgA−/− mice, we initiated a short-term treatment of TGF-β neutralization using a monoclonal antibody that inhibits all three isoforms of TGF-β (TGF-β1, 2, and 3). Analysis of islets of NOD.ChgA−/− mice following treatment with the anti–TGF-β antibody revealed a significant increase in the infiltration of immune cells (Fig. 4A and B). We observed increased frequencies of CD4 and CD8 T cells as well as dendritic cells in anti–TGF-β antibody–treated NOD.ChgA−/− mice as compared with the untreated mice (Fig. 4A and B).
Figure 4.
Anti–TGF-β and anti–PD-1 dual antibody (ab) treatment render NOD.ChgA−/− mice susceptible to autoimmune diabetes. A: Representative FACS plots showing the percentage of indicated immune cell populations in the islets of NOD.ChgA−/− mice either treated with four injections of anti–TGF-β ab every 3 days or left untreated (control). Infiltration of leukocytes was analyzed 15 days postinitiation of treatment. B: Quantification of the percentage of indicated cell populations among total islet cells as described in panel A. Data (mean ± SEM) are pooled from three independent experiments. Each dot represents individual mouse. C: Experimental design of the anti–TGF-β and anti–PD-1 ab treatment. D: Incidence of diabetes in 8–10 week old NOD or NOD.ChgA−/− mice treated with anti–TGF-β either alone or in combination with anti–PD-1. Data are pooled from three independent experiments. P values were calculated by unpaired two-tailed Student t test. *P < 0.05. DC, dendritic cell; ns, not significant.
We next treated NOD and NOD.ChgA−/− mice with anti–TGF-β blocking antibody either alone or in combination with the anti–PD-1 antibody and followed diabetes progression for 30 days (Fig. 4C). As shown above, anti–PD-1 treatment alone in NOD.ChgA−/− mice had no effect; however, neutralization of TGF-β together with anti–PD-1 rendered most of the NOD.ChgA−/− mice susceptible to diabetes within a few days of treatment. Importantly, under the dual antibody treatment, the incidence of T1D as well as the progression of diabetes in the NOD.ChgA−/− mice followed kinetics similar to those in the NOD mice (Fig. 4D). Although TGF-β neutralization alone increased infiltration of T cells into the islets of NOD.ChgA−/− mice, this treatment did not cause diabetes onset in either strain during the monitoring period (Fig. 4C and D). Notably, the synergistic effect of coadministration of anti–TGF-β and anti–PD-1 antibodies on diabetes incidence was specific to NOD.ChgA−/− mice; there was no acceleration of diabetes progression in WT NOD mice treated with anti–TGF-β and anti–PD-1 antibodies as compared with anti–PD-1 antibody alone (compare Fig. 3C with Fig. 4D). In summary, neutralization of TGF-β enabled autoreactive T cells to enter the islet and synergistically acted with PD-1 blockade to promote the development of clinical diabetes in NOD.ChgA−/− mice.
Discussion
This study shows that ablation of ChgA in NOD mice caused a reduction in the presentation of the other major autoantigen, insulin. A significant decrease in insulin peptide presentation subsequently led to impaired priming of insulin-specific autoreactive T cells, an unexpected consequence of ChgA deficiency. The NOD.ChgA−/− mice manifested a broad impairment in autoimmune responses, which were dominated by regulatory pathways. Targeting such pathways, including regulatory T-cell, TGF-β, and PD-1 pathways, rendered NOD.ChgA−/− mice susceptible to diabetes.
Antigen presentation is affected by the availability of the antigen and the activation state of the antigen-presenting cells. Upon glucose challenge, there was a profound reduction in insulin presentation by white blood cells of NOD.ChgA−/− mice. A partial reduction in the presentation of diabetogenic antigens may have a profound impact. Diabetes development was absent in I-Ag7+/− NOD mice, indicating that reducing the presentation by half was sufficient to completely block the autoimmune process (3). Impaired insulin presentation in the islets and peripheral blood correlated with the reduction of the insulin levels in both the pancreas and serum of the NOD.ChgA−/− mice. Although these findings support an important role of ChgA in regulating insulin biosynthesis and/or secretion, the underlying molecular mechanisms remain largely unexplored. Considering that many of the insulin epitopes are derived from the degradative compartment, the crinosome (7,24), we speculate that the defect in the secretory granule also affected the crinophagic pathway and therefore the overall reduced presentation of insulin epitopes.
In addition to the impairment in the local presentation, we found a major defect in the peripheral priming of insulin reactive T cells in NOD.ChgA−/− mice. Previous studies have also demonstrated the absence of BDC2.5 T-cell activation in the periphery of NOD.ChgA−/−. These findings together show impaired peripheral priming of T cells reactive to multiple diabetogenic epitopes. Because peripheral priming is essential for the mobilization of T cells into the islet, this may be a major mechanism for the lack of entrance T cells in the islet of NOD.ChgA−/− mice.
Although we did not find major differences in the activation state of islet macrophages in young NOD.ChgA−/− mice, other studies have documented a role for ChgA-derived peptides in the regulation of inflammation (15,16,40). Peritoneal macrophages from NOD.ChgA−/− mice produced significantly lower amounts of TNF-α, IL-1β, IL-12, IL-6, MCP1, and iNOS upon lipopolysaccharide stimulation (41,42). Treatment of mice with pancreastatin, a dysglycemic peptide derived from ChgA, significantly increased proinflammatory cytokines in peritoneal macrophages and in the white adipose tissue (42). Importantly, NOD.ChgA−/− mice are also protected from dextran sulfate sodium–induced colitis (41). However, anti-inflammatory functions of some ChgA-derived peptides have been also reported (43). Thus, in addition to antigen recognition, ChgA may also directly regulate autoreactivity via a complex mechanism. Whether and how these bioactive peptides contribute to diabetogenesis has not been examined, but it is possible that their deficiency may be associated with diabetes resistance in the NOD.ChgA−/− mice.
We consider that the impaired presentation of insulin in the NOD.ChgA−/− mice results from multiple factors and may not be limited to insulin. Other β-cell antigens that are part of the secretory granule may also be affected and require future investigation.
Article Information
Acknowledgments. The authors thank all the members of the Unanue laboratory for providing critical advice and discussion on many aspects of this project. The authors appreciate the help of Katherine Fredericks (Department of Pathology and Immunology, Washington University School of Medicine, St. Louis, MO) for the maintenance of the mouse colony and Pavel Zakharov (Department of Pathology and Immunology, Washington University School of Medicine, St. Louis, MO) for his assistance in some experiments.
Funding. This study is supported by grants from the National Institutes of Health (DK058177 and AI114551), Juvenile Diabetes Research Foundation, and Kilo Diabetes and Vascular Research Foundation.
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. N.S., X.W., and E.R.U. designed the study, analyzed the data, and wrote and edited the manuscript. N.S., H.H., A.N.V., and O.J.P. performed all the experiments. R.L.B., K.H., and L.T. critically reviewed the manuscript. K.H. provided NOD.ChgA−/− mice. L.T. provided Ins12–20 tetramers. N.S., X.W., and E.R.U. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This article contains supplementary material online at https://doi.org/10.2337/figshare.16554321.
References
- 1. Roep BO, Peakman M. Antigen targets of type 1 diabetes autoimmunity. Cold Spring Harb Perspect Med 2012;2:a007781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Nakayama M, Abiru N, Moriyama H, et al. Prime role for an insulin epitope in the development of type 1 diabetes in NOD mice. Nature 2005;435:220–223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Gioia L, Holt M, Costanzo A, et al. Position β57 of I-Ag7 controls early anti-insulin responses in NOD mice, linking an MHC susceptibility allele to type 1 diabetes onset. Sci Immunol 2019;4:eaaw6329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mohan JF, Levisetti MG, Calderon B, Herzog JW, Petzold SJ, Unanue ER. Unique autoreactive T cells recognize insulin peptides generated within the islets of Langerhans in autoimmune diabetes. Nat Immunol 2010;11:350–354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Babon JA, DeNicola ME, Blodgett DM, et al. Analysis of self-antigen specificity of islet-infiltrating T cells from human donors with type 1 diabetes. Nat Med 2016;22:1482–1487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Michels AW, Landry LG, McDaniel KA, et al. Islet-derived CD4 T cells targeting proinsulin in human autoimmune diabetes. Diabetes 2017;66:722–734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Wan X, Vomund AN, Peterson OJ, Chervonsky AV, Lichti CF, Unanue ER. The MHC-II peptidome of pancreatic islets identifies key features of autoimmune peptides. Nat Immunol 2020;21:455–463 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Baker RL, Delong T, Barbour G, Bradley B, Nakayama M, Haskins K. Cutting edge: CD4 T cells reactive to an islet amyloid polypeptide peptide accumulate in the pancreas and contribute to disease pathogenesis in nonobese diabetic mice. J Immunol 2013;191:3990–3994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Yamamoto T, Yamato E, Tashiro F, et al. Development of autoimmune diabetes in glutamic acid decarboxylase 65 (GAD65) knockout NOD mice. Diabetologia 2004;47:221–224 [DOI] [PubMed] [Google Scholar]
- 10. Alkemade GM, Clemente-Casares X, Yu Z, et al. Local autoantigen expression as essential gatekeeper of memory T-cell recruitment to islet grafts in diabetic hosts. Diabetes 2013;62:905–911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Baker RL, Bradley B, Wiles TA, et al. Cutting edge: nonobese diabetic mice deficient in chromogranin A are protected from autoimmune diabetes. J Immunol 2016;196:39–43 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Stadinski BD, Delong T, Reisdorph N, et al. Chromogranin A is an autoantigen in type 1 diabetes. Nat Immunol 2010;11:225–231 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Delong T, Wiles TA, Baker RL, et al. Pathogenic CD4 T cells in type 1 diabetes recognize epitopes formed by peptide fusion. Science 2016;351:711–714 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Eissa N, Hussein H, Hendy GN, Bernstein CN, Ghia JE. Chromogranin-A and its derived peptides and their pharmacological effects during intestinal inflammation. Biochem Pharmacol 2018;152:315–326 [DOI] [PubMed] [Google Scholar]
- 15. Mahata SK, Corti A. Chromogranin A and its fragments in cardiovascular, immunometabolic, and cancer regulation. Ann N Y Acad Sci 2019;1455:34–58 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. D’amico MA, Ghinassi B, Izzicupo P, Manzoli L, Di Baldassarre A. Biological function and clinical relevance of chromogranin A and derived peptides. Endocr Connect 2014;3:R45–R54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Mahapatra NR, O’Connor DT, Vaingankar SM, et al. Hypertension from targeted ablation of chromogranin A can be rescued by the human ortholog. J Clin Invest 2005;115:1942–1952 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Kim T, Zhang CF, Sun Z, Wu H, Loh YP. Chromogranin A deficiency in transgenic mice leads to aberrant chromaffin granule biogenesis. J Neurosci 2005;25:6958–6961 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Portela-Gomes GM, Gayen JR, Grimelius L, Stridsberg M, Mahata SK. The importance of chromogranin A in the development and function of endocrine pancreas. Regul Pept 2008;151:19–25 [DOI] [PubMed] [Google Scholar]
- 20. Schmid GM, Meda P, Caille D, et al. Inhibition of insulin secretion by betagranin, an N-terminal chromogranin A fragment. J Biol Chem 2007;282:12717–12724 [DOI] [PubMed] [Google Scholar]
- 21. Tatemoto K, Efendić S, Mutt V, Makk G, Feistner GJ, Barchas JD. Pancreastatin, a novel pancreatic peptide that inhibits insulin secretion. Nature 1986;324:476–478 [DOI] [PubMed] [Google Scholar]
- 22. Gayen JR, Saberi M, Schenk S, et al. A novel pathway of insulin sensitivity in chromogranin A null mice: a crucial role for pancreastatin in glucose homeostasis. J Biol Chem 2009;284:28498–28509 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Wollam J, Mahata S, Riopel M, et al. Chromogranin A regulates vesicle storage and mitochondrial dynamics to influence insulin secretion. Cell Tissue Res 2017;368:487–501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Wan X, Zinselmeyer BH, Zakharov PN, et al. Pancreatic islets communicate with lymphoid tissues via exocytosis of insulin peptides. Nature 2018;560:107–111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Vomund AN, Lichti CF, Peterson OJ, Arbelaez AM, Wan X, Unanue ER. Blood leukocytes recapitulate diabetogenic peptide-MHC-II complexes displayed in the pancreatic islets. J Exp Med 2021;218:e20202530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Mohan JF, Unanue ER. Unconventional recognition of peptides by T cells and the implications for autoimmunity. Nat Rev Immunol 2012;12:721–728 [DOI] [PubMed] [Google Scholar]
- 27. Mohan JF, Petzold SJ, Unanue ER. Register shifting of an insulin peptide-MHC complex allows diabetogenic T cells to escape thymic deletion. J Exp Med 2011;208:2375–2383 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Mohan JF, Calderon B, Anderson MS, Unanue ER. Pathogenic CD4+ T cells recognizing an unstable peptide of insulin are directly recruited into islets bypassing local lymph nodes. J Exp Med 2013;210:2403–2414 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wan X, Thomas JW, Unanue ER. Class-switched anti-insulin antibodies originate from unconventional antigen presentation in multiple lymphoid sites. J Exp Med 2016;213:967–978 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Levisetti MG, Suri A, Frederick K, Unanue ER. Absence of lymph nodes in NOD mice treated with lymphotoxin-beta receptor immunoglobulin protects from diabetes. Diabetes 2004;53:3115–3119 [DOI] [PubMed] [Google Scholar]
- 31. Gagnerault MC, Luan JJ, Lotton C, Lepault F. Pancreatic lymph nodes are required for priming of beta cell reactive T cells in NOD mice. J Exp Med 2002;196:369–377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Wildin RS, Freitas A. IPEX and FOXP3: clinical and research perspectives. J Autoimmun 2005;25(Suppl.):56–62 [DOI] [PubMed] [Google Scholar]
- 33. Salomon B, Lenschow DJ, Rhee L, et al. B7/CD28 costimulation is essential for the homeostasis of the CD4+CD25+ immunoregulatory T cells that control autoimmune diabetes. Immunity 2000;12:431–440 [DOI] [PubMed] [Google Scholar]
- 34. Wang J, Yoshida T, Nakaki F, Hiai H, Okazaki T, Honjo T. Establishment of NOD-Pdcd1-/- mice as an efficient animal model of type I diabetes. Proc Natl Acad Sci USA 2005;102:11823–11828 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Ansari MJ, Salama AD, Chitnis T, et al. The programmed death-1 (PD-1) pathway regulates autoimmune diabetes in nonobese diabetic (NOD) mice. J Exp Med 2003;198:63–69 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Hu H, Zakharov PN, Peterson OJ, Unanue ER. Cytocidal macrophages in symbiosis with CD4 and CD8 T cells cause acute diabetes following checkpoint blockade of PD-1 in NOD mice. Proc Natl Acad Sci USA 2020;117:31319–31330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Mariathasan S, Turley SJ, Nickles D, et al. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 2018;554:544–548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Tauriello DVF, Palomo-Ponce S, Stork D, et al. TGFβ drives immune evasion in genetically reconstituted colon cancer metastasis. Nature 2018;554:538–543 [DOI] [PubMed] [Google Scholar]
- 39. Calon A, Espinet E, Palomo-Ponce S, et al. Dependency of colorectal cancer on a TGF-β-driven program in stromal cells for metastasis initiation. Cancer Cell 2012;22:571–584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Muntjewerff EM, Dunkel G, Nicolasen MJT, Mahata SK, van den Bogaart G. Catestatin as a target for treatment of inflammatory diseases. Front Immunol 2018;9:2199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Eissa N, Hussein H, Kermarrec L, et al. Chromogranin-A regulates macrophage function and the apoptotic pathway in murine DSS colitis. J Mol Med (Berl) 2018;96:183–198 [DOI] [PubMed] [Google Scholar]
- 42. Bandyopadhyay GK, Lu M, Avolio E, et al. Pancreastatin-dependent inflammatory signaling mediates obesity-induced insulin resistance. Diabetes 2015;64:104–116 [DOI] [PubMed] [Google Scholar]
- 43. Ying W, Mahata S, Bandyopadhyay GK, et al. Catestatin inhibits obesity-induced macrophage infiltration and inflammation in the liver and suppresses hepatic glucose production, leading to improved insulin sensitivity. Diabetes 2018;67:841–848 [DOI] [PMC free article] [PubMed] [Google Scholar]