ABSTRACT
Background:
The pathomechanisms of hypoxemia and treatment strategies for type H and type L acute respiratory distress syndrome (ARDS) in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)-induced coronavirus disease 2019 (COVID-19) have not been elucidated.
Main text:
SARS-CoV-2 mainly targets the lungs and blood, leading to ARDS, and systemic thrombosis or bleeding. Angiotensin II-induced coagulopathy, SARS-CoV-2-induced hyperfibrin(ogen)olysis, and pulmonary and/or disseminated intravascular coagulation due to immunothrombosis contribute to COVID-19-associated coagulopathy. Type H ARDS is associated with hypoxemia due to diffuse alveolar damage-induced high right-to-left shunts. Immunothrombosis occurs at the site of infection due to innate immune inflammatory and coagulofibrinolytic responses to SARS-CoV-2, resulting in microvascular occlusion with hypoperfusion of the lungs. Lung immunothrombosis in type L ARDS results from neutrophil extracellular traps containing platelets and fibrin in the lung microvasculature, leading to hypoxemia due to impaired blood flow and a high ventilation/perfusion (VA/Q) ratio. COVID-19-associated ARDS is more vascular centric than the other types of ARDS. D-dimer levels have been monitored for the progression of microvascular thrombosis in COVID-19 patients. Early anticoagulation therapy in critical patients with high D-dimer levels may improve prognosis, including the prevention and/or alleviation of ARDS.
Conclusions:
Right-to-left shunts and high VA/Q ratios caused by lung microvascular thrombosis contribute to hypoxemia in type H and L ARDS, respectively. D-dimer monitoring-based anticoagulation therapy may prevent the progression to and/or worsening of ARDS in COVID-19 patients.
Keywords: Acute respiratory distress syndrome (ARDS), compliance, COVID-19, hypoxemia, neutrophil extracellular traps (NETs), shunt, thrombosis, VA/Q
Abbreviations: ACE2, angiotensin converting enzyme 2, Ang II, angiotensin II, ARDS, acute respiratory distress syndrome, AT1R, angiotensin II type 1 receptor, BAL, bronchoalveolar lavage, COVID-19, coronavirus disease 2019, Crs, respiratory system compliance, CT, computed tomography, DIC, disseminated intravascular coagulation, FDP, fibrin/fibrinogen degradation product, NETs, neutrophil extracellular traps, PAI-1, plasminogen activator inhibitor-1, SARS-CoV, Severe acute respiratory syndrome coronavirus, SARS-CoV-2, Severe acute respiratory syndrome coronavirus 2, t-PA, tissue-type plasminogen activator, u-PA, urokinase-type plasminogen activator, VA/Q, ventilation/perfusion
INTORODUCTION
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) causes coronavirus disease 2019 (COVID-19), which results in multiple organ dysfunctions throughout the body (1). The lungs and blood mainly effect the prognosis of COVID-19 patients. Severe cases of SARS-CoV-2 infection frequently result in pneumonia, which in turn leads to acute respiratory distress syndrome (ARDS). Reports from China discussed abnormal hematological tests associated with increases in inflammatory cytokines and high incidence of thromboembolic complications leading to disseminated intravascular coagulation (DIC) (2, 3). The close interplay between coagulation and inflammation in acute lung injuries including ARDS is well established (4). Notably, participation of pulmonary intravascular coagulation involving alveolar epithelial and lung endothelial injury has been implicated in COVID-19-associated pneumonia (5, 6). It has been speculated that early stage of COVID-19 does not lead to typical ARDS (7) and that COVID-19-associated ARDS is of two phenotypes, type L and type H, which show the same levels of hypoxemia but have different computed tomography (CT) findings, lung elastances (compliances), and pathomechanisms of low partial pressure to fractional inspired oxygen (PaO2/FiO2) (8). Using the database of the “LUNG SAFE” study, Panwar et al. (9) determined the respiratory system compliance (Crs)-based ARDS phenotypes and showed that 12% of ARDS patients exhibited preserved Crs and that 55% of those patients exhibited a low PaO2/FiO2 ratio less than 150. The results suggest that the preserved compliance-phenotype (type L) seen in COVID-19-associated ARDS is also present in patients with ARDS not associated with COVID-19. However, the pathomechanisms of hypoxemia in two phenotypes of ARDS, especially type L, that have linked to low ventilation/perfusion (VA/Q) ratios in original study, have not yet been fully understood (10, 11).
This narrative review discusses the pathomechanisms of underlying hypoxemia in two phenotypes of COVID-19-associated ARDS based on the insights from thrombosis and hemostasis and shows that a high VA/Q ratio is a primary pathomechanism of hypoxemia in type L ARDS, while low VA/Q ratio is not. Thereafter, the management of COVID-19-associated ARDS with respect to thrombosis and hemostasis has been discussed.
SEARCH STRATEGIES
Using the search terms “COVID-19 or SARS-CoV-2” and “pneumonia or ARDS, acute respiratory distress syndrome, phenotype, pathology, autopsy, inflammation, coagulation, fibrinolysis, neutrophil,” studies published from January 1, 2020 to May 20, 2021 were identified in PubMed. We manually checked the references of the selected articles to identify additional relevant articles. We limited our search to articles that were published in English. The selected relevant articles were reviewed henceforth.
BRONCHOALVEOLAR COAGULATION AND FIBRINOLYSIS IN PATIENTS WITH ARDS
Bronchoalveolar thrombosis and hemostasis are crucial to the pathogenesis of ARDS (4, 12). Alveolar epithelial cells express the procoagulant tissue factor, which binds to Factor VII to initiate tissue factor-dependent coagulation pathway, and this has been confirmed in bronchoalveolar lavage (BAL) fluid in ARDS patients (13, 14). Furthermore, increased levels of fibrinopeptide A, which is a direct marker of thrombin generation and its action on fibrinogen, were observed in BAL samples from ARDS patients (15). Decreased levels of protein C and increased levels of soluble thrombomodulin, probably degraded from alveolar epithelial thrombomodulin, were also observed in the fluid of pulmonary edemas in patients with acute lung injury and ARDS (16), indicating the low levels of protein C/thrombomodulin and impaired function of associated anticoagulation systems in the alveolus. High levels of plasminogen activator inhibitor-1 (PAI-1) in association with low levels of tissue-type plasminogen activator (t-PA) and urokinase-type plasminogen activator (u-PA) in the BAL fluid from ARDS patients indicate a suppression of fibrinolysis in alveolar spaces (14, 15). Plasma samples from ARDS patients also showed increased levels of fibrinopeptide A, PAI-1, and soluble thrombomodulin, as well as low levels of protein C (15, 16). Taken together, the procoagulant and depressed fibrinolytic activities associated with the impaired function of anticoagulation systems in patients with ARDS provoke intra-alveolar and lung intravascular fibrin formation.
COAGULOPATHY IN COVID-19-ASSOCIATED ARDS
Angiotensin-converting enzyme 2 (ACE2) is widely expressed throughout the body, including lung epithelial cells and endothelial cells, especially in type II alveolar epithelial cells (17, 18). SARS-CoV-2 invades cells using the spike glycoprotein that binds to the ACE2, which acts as an entry receptor, resulting in the downregulation and inactivation of ACE2 and elevation of angiotensin II (Ang II) levels (19, 20). Elevation of plasma Ang II levels in parallel with an increase in SARS-CoV-2 viral load is negatively correlated with PaO2/FiO2, while an increase in the viral load of SARS-CoV-2 is positively correlated with the Murray lung injury score (21). The spike glycoprotein of SARS-CoV, belonging to the coronavirus family, binds to ACE2, downregulating its expression, and leading to worsening of lung function, as measured by elastances, lung wet/dry ratio, Murray lung injury score, and other pathological findings, via angiotensin II type 1 receptor (AT1R) (22). In addition, recombinant human ACE2 has been shown to exhibit protective effects in the lungs of patients with acute lung injury who are associated with markedly elevated Ang II levels in the lung and plasma (23). This study further confirmed that Ang II effects through AT1R are involved in the ACE2-mediated pathological conditions of the lung. These results support the notion that Ang II may play a role in SARS-CoV-2-induced ARDS through intra-alveolar and microvascular thrombosis in the lungs (5).
High levels of the plasmin antiplasmin complex, fibrin/fibrinogen degradation product (FDP) and D-dimer, and increased FDP/D-dimer ratio in patients with COVID-19, especially in critically ill and non-survivors, indicate increased fibrin(ogen)olysis (2, 3, 24, 25). However, few studies have investigated coagulopathy, including bronchoalveolar hemostasis, in COVID-19-associated ARDS. Hunt and Levi (26) suggested intra-alveolar fibrinolysis in patients with COVID-19, which they have shown is distinct from impaired fibrinolysis caused by PAI-1 observed in other types of ARDS (14, 15). It is speculated that the increased intra-alveolar fibrin(ogen)olysis may be triggered by SARS-CoV-2 RNA (27), neutrophil extracellular traps (NETs) comprising neutrophil DNA and elastase (28, 29), activated alveolar macrophages (30), and dysregulated Serpine1-mediated inhibition of t-PA and u-PA (31). In severe cases of SARS-CoV-2 infection, intra-alveolar hyperfibrin(ogen)olysis disseminates to the whole body, similar to that during influenza virus infections (32).
Immunothrombosis is an innate-immune inflammatory and coagulofibrinolytic response to a pathogenic insult with compartmentalization of “non-self” and “altered-self” and suppression of their spread to locally maintain homeostasis with limited host damage (33). The main pathophysiology of immunothrombosis is NET-induced activation of coagulation, suppression of anticoagulation, and impairment of fibrinolysis (33–35). Immunothrombosis in the lung vasculature causes pulmonary intravascular coagulation in COVID-19 pneumonia, suggesting a stage of type L ARDS (5). If the insult is sufficiently severe, immunothrombosis disseminates into systemic circulation, giving rise to systemic microvascular thrombosis, namely DIC (33, 35), which is associated with diffuse alveolar damage. This stage of ARDS may be classified as type H ARDS. The prevalence of DIC in COVID-19 is very low, approximately 5% in COVID-19-associated coagulopathy; however, when present, this complication is frequently fatal due to systemic microvascular thrombosis and bleeding (36, 37).
PATHOMECHANISMS OF HYPOXEMIA IN COVID-19-ASSOCIATED ARDS
COVID-19 patients with type H ARDS show high elastance, high right-to-left shunt, and high lung weight, and these are also characteristics of patients with typical ARDS showing diffuse alveolar damage. In contrast, those with type L ARDS show the following characteristics: low elastance, low VA/Q ratio, and low lung weight, and broadly spared lung with normally aerated areas and a few restricted areas with ground-glass opacity, as seen using computed tomography (CT) findings (8). The low VA/Q ratio in patients with type L ARDS may be attributed to loss of hypoxic vasoconstriction with impaired regulation of blood flow, probably leading to hyper-perfusion of gasless regions (7, 8). However, many previous studies have reported contradicting findings. Microvascular and intra-alveolar fibrin formation in the lungs are hallmarks of ARDS (38), including COVID-19-associated ARDS (5, 6), and fibrin and FDP play important roles in neutrophil recruitment to the lungs, as well as during endothelial injury associated with increased permeability (39, 40). Lung microvascular activation of neutrophils results in NETosis, leading to microvascular thrombosis involving platelets, neutrophils, and fibrin in COVID-19-associated ARDS (41, 42). These phenomena consequently lead to high VA/Q ratio and impaired perfusion due to microvascular thrombosis, which are likely to occur in patients with type L ARDS.
Lung biopsies performed at the early stages of acute respiratory insufficiency in patients with viral or bacterial pneumonia revealed fibrin and fibrinogen debris, as well as microvascular thrombosis in multiple biopsies taken during the same operation (43). In patients with acute respiratory failure, of which 20% were caused by viral pneumonia, balloon occlusion angiography demonstrated multiple pulmonary artery filling defects, regardless of whether the areas were opacified or normal as seen in the chest X-ray and severity of failure (44). A histologic study showed that 83% of patients with filling defects showed the occurrence of thrombosis in small and large pulmonary arteries (44). Dual-energy CT imaging in patients with COVID-19-asociated pneumonia with D-dimer levels >1.0 μg/mL showed decreased perfusion in peripheral areas with ground-glass-opacity (45), and these results are inconsistent with the results observed in patients with type L ARDS (7, 8). Patients with COVID-19-associated ARDS can be divided into four subgroups depending on the median D-dimer level (1.8 μg/mL) and static compliance (41 mL/cmH20) (46). CT angiogram demonstrated that 94% of patients with high D-dimer levels were associated with diffuse bilateral hypoperfusion areas consistent with the areas of thrombosis or emboli, irrespective of their compliance. Notably, dead space measured by ventilatory ratio in these patients was high, which indicated that high VA/Q ratio due to intravascular thrombosis contributes to hypoxemia with preserved compliance in patients with COVID-19-associated ARDS (46). Lung perfusion can be assessed using subtraction CT angiography based on iodine distribution maps. A high incidence of perfusion abnormality (87.8%), mainly hypoperfusion, in areas of apparently normal lung tissue was observed in COVID-19 patients with abnormal CT findings (ground-glass opacities and/or consolidation) associated with low PaO2/FiO2 and high D-dimer levels (47). Typical patients with type L ARDS showed large areas of severe hypoperfusion in the apparently healthy lung and increased perfusion in small areas of ground-glass opacities (47). The results suggest that VA/Q mismatch, mainly high VA/Q and partly low VA/Q, is attributable to the pathomechanisms of hypoxemia in type L ARDS. Bimodal distributions of blood flow and ventilation using the J.B. West model in five COVID-19-associated ARDS patients support this notion (48). This study showed extreme mismatch in perfusion and ventilation; high perfusion with low ventilation in the low VA/Q area, and high ventilation with low perfusion in the high VA/Q area, which indicates that not only shunt fraction, but also VA/Q mismatches are related to hypoxemia in COVID-19-associated ARDS.
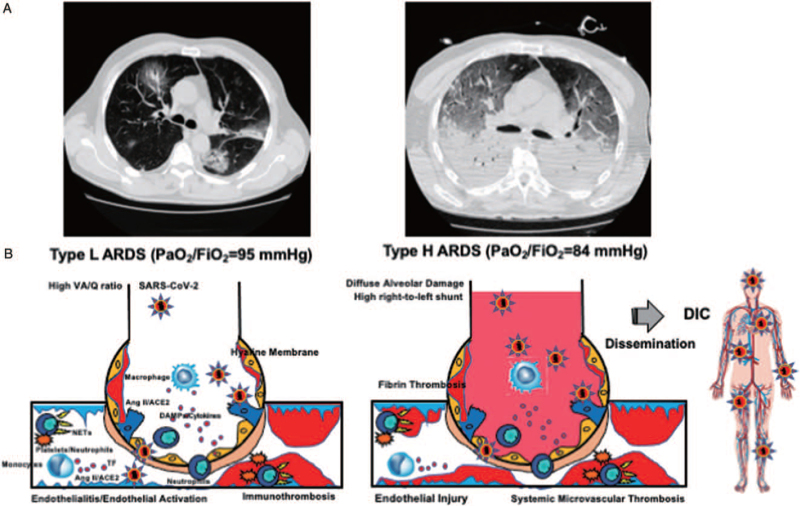
Initial pulmonary intravascular coagulation is characteristic of COVID-19-associated pneumonia (5). Many autopsy studies have demonstrated the presence of platelets, neutrophils, and fibrin thrombosis in small vessels, arterioles, venules, and capillaries in the lungs of COVID-19 patients (49–52). Importantly, fibrin microvascular thrombosis could be found both in injured and more preserved lung parenchyma (52). Furthermore, microvascular thrombosis with neutrophils forming NETs with platelets and fibrin was confirmed in patients with COVID-19-asscoaited pneumonia and ARDS (41, 42). Aggregated NETs occluded a range of pulmonary vessels, from capillaries to small- and middle-size vessels, with endothelial injuries (53). These autopsy findings clearly indicate the impaired perfusion in the normal and injured areas of the lung, indicating that a high VA/Q ratio is involved in the pathology of low PaO2/FiO2 seen in type L ARDS. Figure 1 summarizes the pathologies of type L and type H ARDS associated with COVID-19.
Fig. 1.
Pathomechanisms of COVID-19-associated ARDS. A, Typical CT findings of two phenotypes of COVID-19-associated ARDS. B, SARS-CoV-2 binds to ACE2, leading to its downregulation and increase in Ang II levels, resulting in Ang II-induced coagulopathy. SARS-CoV-2 RNA, which is a pathogen-associated molecular pattern, and damage-associated molecular pattern generated by pyroptosis and necroptosis of virus-infected cells that bind to pattern recognition receptors and induce the expression of inflammatory cytokines and activation of platelets and neutrophils, which results in NETosis-induced coagulopathy. Both coagulopathies activate coagulation and impair the function of anticoagulation factors that are associated with endothelial activation and injury. Fibrinolytic pathways are activated or inhibited based on the balance of SARS-CoV-2-induced fibrin(ogen)olysis and PAI-1-mediated inhibition of fibrinolysis. Type L ARDS represents early phase of COVID-19, which is characterized microvascular immunothrombosis-induced hypoperfusion both in damaged and normal lung alveoli. The main pathomechanism of hypoxemia in type L ARDS is high VA/Q ratio with preserved compliance. Type H ARDS represents the more typical ARDS with hypoxemia, which is caused by diffuse alveolar damage-induced high right-to-left shunt with deteriorated compliance. Type H ARDS, in severe cases, may lead to systemic microvascular thrombosis, also called disseminated intravascular coagulation, as a result of dysregulated immunothrombosis disseminating to the whole body. ACE2 indicates angiotensin-converting enzyme 2; Ang II, angiotensin II; ARDS, acute respiratory distress syndrome; COVID19, coronavirus disease 2019; DAMPs, damage-associated molecular pattern; DIC, disseminated intravascular coagulation; NETs, neutrophil extracellular traps; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; TF, tissue factor; VA/Q, ventilation/perfusion. A, Adapted from (8) with modification (Creative Commons Attribution-NonCommercial 4.0 International License).
MANAGEMENT
Monitoring. Reports from China showed markedly higher levels of D-dimer and FDP in critical cases of COVID-19 than in moderate and severe cases and in non-survivors than in survivors (2, 3). High D-dimer levels are independent predictors of ARDS and death due to COVID-19 (54). High values of D-dimer and FDP are due to intrapulmonary coagulation or DIC followed by increased fibrin(ogen)olysis (3, 5, 26). A systematic review and meta-analysis revealed that D-dimer levels are associated with the severity of COVID-19 and contribute to increased aggression of disease and progression to ARDS (55–57). COVID-19 patients with severe hypoperfusion in areas of normal lung parenchyma in subtraction CT angiography with high D-dimer levels mimicking type L ARDS were associated with increased probability of admission to the ICU and initiation of invasive mechanical ventilation (47). COVID-19-associated ARDS patients with high D-dimer levels and low compliance that mimics type H ARDS showed significantly lower survival probability and higher hazard ratio for 28-day mortality than those with high compliance (type L ARDS) (46).
In brief, D-dimer levels should be seriously evaluated to monitor progression to and/or worsening of ARDS, to enable timely initiation of mechanical ventilation, and for effective prognosis of the COVID-19 patients. Hypoxemia with restricted ground-glass opacities and large, normally aerated areas in CT finding associated with elevated D-dimer levels is highly suggestive of type L ARDS.
Treatment. All guidelines published during the search period recommend thromboprophylaxis using various anticoagulation drugs with varying regimens; however, these recommendations lack clear evidence (58). Three randomized controlled trials that studied different doses of heparin as anticoagulant therapy with end points of organ-support-free days, arterial and venous thrombosis, and/or hospital mortality have not reached robust conclusions (58). A multicenter international prospective registry including 5,480 COVID-19 patients showed a lower mortality rate with the use of anticoagulation therapy in those with respiratory failure requiring invasive ventilation (59). Although single-center retrospective in nature, the study included 4,389 COVID-19 patients and confirmed that both prophylactic and therapeutic doses of anticoagulation therapies initiated within 48 h of admission decreased the incidence of intubation and hospital mortality compared with the non-anticoagulation therapy group (60).
Early anticoagulation therapy in critical COVID-19 patients with high D-dimer levels may improve prognosis, including prevention and/or alleviation of ARDS. However, the dosing regimen and time to start anticoagulation therapy as well as the threshold of D-dimer levels are still unclear. The risk of bleeding should always be considered.
CONTROVERSIES
Many controversies exist regarding the ventilatory strategies in COVID-19-associated ARDS, especially in the type L ARDS with preserved compliance (61–64). However, this topic is beyond the scope of this review, which focuses on thrombosis and hemostasis. Next, do the phenotypes of COVID-19-associated ARDS exist? If they exist, does type L progress to type H ARDS?
COVID-19-associated ARDS is a dysregulated innate immune, inflammatory, and coagulofibrinolytic responses to SARS-CoV-2 infection, which leads to organ dysfunction associated with intrapulmonary and/or disseminated intravascular coagulation. This pathology is similar to sepsis-induced ARDS, in which a genome-wide association of a patient's susceptibility to the condition has been confirmed (65). The use of genomic approximation can identify predictors for early and precise ARDS development and the risk of critical illness (66). Genetic variants associated with alterations in immune responses and susceptibility to SARS-CoV-2 have also been reported in COVID-19 infection (67). Therefore, the phenotypes of COVID-19 may develop according to the patient's genetic predisposition.
More than 80% of experts agreed that the pathophysiology of COVID-19 is similar to that of other forms of ARDS (68). Grasselli et al. (46) also concluded that the pathophysiology of COVID-19-associated ARDS is similar to that of ARDS unrelated to COVID-19. However, their median Crs was 28% (41 mL/cmH2O) higher than classical ARDS, and CT angiography confirmed more normally ventilated areas similar to type L ARDS than ARDS from classical etiologies. These inconsistencies were pointed out by Gattinoni et al., the original advocate of type L ARDS (69). Furthermore, another phenotype of COVID-19-associated ARDS has been proposed (70). Do type L and type H phenotypes really exist? Further clarification is needed. In the real clinical settings, a variety of patterns of development of types of ARDS have been experienced, such as sudden onset of type H, gradual progression from type L to type H, or recovery from both types without progressing to another type of ARDS. These diversities mean that it is not conclusive whether sequential progression from type L to type H always occurs.
CONCLUSIONS
Local immunothrombosis occurs in the alveolus and microvasculature of the lungs. Immunothrombosis in the lung microvasculature gives rise to type L ARDS with high VA/Q ratio and preserved compliance. Diffuse alveolar damage associated with systemic microvascular thrombosis leads to type H ARDS with a high right-to-left shunt and deteriorated compliance. Therefore, the main pathomechanisms of low PaO2/FiO2 are due to VA/Q mismatch (mainly high VA/Q, in part low VA/Q ratio) and right-to-left shunt in patients with type L and type H ARDS, respectively. COVID-19-associated ARDS is a highly vascular-centric disease that requires serial monitoring of D-dimer levels to initiate anticoagulation therapy without delay. The high risk of bleeding observed in COVID-19-associated ARDS should also be considered.
Footnotes
Revised 10 May, 2021
SG was personally funded by Grifols outside the submitted work.
The authors report no conflicts of interest.
This work was supported in part by JSPS KAKENHI (Grant-in-Aid (C) 2020, 20K09280).
Authors’ Contributions: SG and TW contributed equally to this article. SG and TW planned, cowrote, improved, critically revised, and approved this review.
REFERENCES
- 1.Wadman M, Couzin-Frankel J, Kaiser J, Matacic C. A rampage through the body. Science 368 (6489):356–360, 2020. [DOI] [PubMed] [Google Scholar]
- 2.Liao D, Zhou F, Luo L, Xu M, Wang H, Xia J, Gao Y, Cai L, Wang Z, Yin P, et al. Haematological characteristics and risk factors in the classification and prognosis evaluation of COVID-19: a retrospective cohort study. Lancet Haematol 7 (9):e671–e678, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tang N, Li D, Wang X, Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J Thromb Haemost 18 (4):844–847, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Welty-Wolf KE, Carraway MS, Ortel TL, Piantadosi CA. Coagulation and inflammation in acute lung injury. Thromb Haemost 88 (1):17–25, 2002. [PubMed] [Google Scholar]
- 5.McGonagle D, O’Donnell JS, Sharif K, Emery P, Bridgewood C. Immune mechanisms of pulmonary intravascular coagulopathy in COVID-19 pneumonia. Lancet Rheumatol 2 (7):e437–e445, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Teuwen LA, Geldhof V, Pasut A, Carmeliet P. COVID-19: the vasculature unleashed. Nat Rev Immunol 20 (7):389–391, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gattinoni L, Coppola S, Cressoni M, Busana M, Rossi S, Chiumello D. COVID-19 does not lead to a “Typical” Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med 201 (10):1299–1300, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Gattinoni L, Chiumello D, Caironi P, Busana M, Romitti F, Brazzi L, Camporota L. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med 46:1099–1102, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Panwar R, Madotto F, Laffey JG, van Haren FMP. Compliance phenotypes in early acute respiratory distress syndrome before the COVID-19 pandemic. Am J Respir Crit Care Med 202 (9):1244–1252, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jha AK. Anatomical, physiological and clinical similarities and differences in ARDS physiological subtypes. Intensive Care Med 47 (4):493–494, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chiumello D, Camporota L, Gattinoni L, Marini JJ. Complexity and unanswered questions in the pathophysiology of COVID-19 ARDS. Intensive Care Med 47 (4):495–496, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hofstra JJ, Haitsma JJ, Juffermans NP, Levi M, Schultz MJ. The role of bronchoalveolar hemostasis in the pathogenesis of acute lung injury. Semin Thromb Hemost 34 (5):475–484, 2008. [DOI] [PubMed] [Google Scholar]
- 13.Marshall BC, Brown BR, Rothstein MA, Rao NV, Hoidal JR, Rodgers GM. Alveolar epithelial cells express both plasminogen activator and tissue factor. Potential role in repair of lung injury. Chest 99: (3 suppl): 25s–27s, 1991. [PubMed] [Google Scholar]
- 14.Idell S, James KK, Levin EG, Schwartz BS, Manchanda N, Maunder RJ, Martin TR, McLary J, Fair DS. Local abnormalities in coagulation and fibrinolytic pathways predispose to alveolar fibrin deposition in the adult respiratory distress syndrome. J Clin Invest 84 (2):695–705, 1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Idell S, Koenig KB, Fair DS, Martin TR, McLarty J, Maunder RJ. Serial abnormalities of fibrin turnover in evolving adult respiratory distress syndrome. Am J Physiol 261 (4 pt 1):L240–48, 1991. [DOI] [PubMed] [Google Scholar]
- 16.Ware LB, Fang X, Matthay MA. Protein C and thrombomodulin in human acute lung injury. Am J Physiol Lung Cell Mol Physiol 285 (3):L514–521, 2003. [DOI] [PubMed] [Google Scholar]
- 17.Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol 203 (2):631–637, 2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Zhao Y, Zhao Z, Wang Y, Zhou Y, Ma Y, Zuo W. Single-Cell RNA expression profiling of ACE2, the receptor of SARS-CoV-2. Am J Respir Crit Care Med 202 (5):756–759, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, Si HR, Zhu Y, Li B, Huang CL, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579 (7798):270–273, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Verdecchia P, Cavallini C, Spanevello A, Angeli F. The pivotal link between ACE2 deficiency and SARS-CoV-2 infection. Eur J Intern Med 76:14–20, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Liu Y, Yang Y, Zhang C, Huang F, Wang F, Yuan J, Wang Z, Li J, Li J, Feng C, et al. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci 63 (3):364–374, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kuba K, Imai Y, Rao S, Gao H, Guo F, Guan B, Huan Y, Yang P, Zhang Y, Deng W, et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat Med 11 (8):875–879, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Imai Y, Kuba K, Rao S, Huan Y, Guo F, Guan B, Yang P, Sarao R, Wada T, Leong-Pol H, et al. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature 436 (7047):112–116, 2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jin X, Duan Y, Bao T, Gu J, Chen Y, Li Y, Mao S, Chen Y, Xie W. The values of coagulation function in COVID-19 patients. PLoS One 15 (10):e0241329, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Blasi A, von Meijenfeldt FA, Adelmeijer J, Calvo A, Ibañez C, Perdomo J, Reventer JC, Lisman T. In vitro hypercoagulability and ongoing in vivo activation of coagulation and fibrinolysis in COVID-19 patients on anticoagulation. J Thromb Haemost 18 (10):2646–2653, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Hunt BJ, Levi M. Re The source of elevated plasma D-dimer levels in COVID-19 infection. Br J Haematol 190 (3):e133–e134, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kannemeier C, Shibamiya A, Nakazawa F, Trusheim H, Ruppert C, Markart P, Song Y, Tzima E, Kennerknecht E, Niepmann M, et al. Extracellular RNA constitutes a natural procoagulant cofactor in blood coagulation. Proc Natl Acad Sci U S A 104 (15):6388–6393, 2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Oehmcke S, Mörgelin M, Herwald H. Activation of the human contact system on neutrophil extracellular traps. J Innate Immun 1 (3):225–230, 2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hayakawa M, Katabami K, Wada T, Sugano M, Hoshino H, Sawamura A, Gando S. Sivelestat (selective neutrophil elastase inhibitor) improves the mortality rate of sepsis associated with both acute respiratory distress syndrome and disseminated intravascular coagulation patients. Shock 33 (1):14–18, 2010. [DOI] [PubMed] [Google Scholar]
- 30.Loscalzo J. The macrophage and fibrinolysis. Semin Thromb Hemost 22 (6):503–506, 1996. [DOI] [PubMed] [Google Scholar]
- 31.Gralinski LE, Bankhead A 3rd, Jeng S, Menachery VD, Proll S, Belisle SE, Matzke M, Webb-Robertson BJ, Luna ML, Shukla AK, et al. Mechanisms of severe acute respiratory syndrome coronavirus-induced acute lung injury. mBio 4 (4):e00271–13, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Berri F, Rimmelzwaan GF, Hanss M, Albina E, Foucault-Grunenwald ML, Lê VB, Vogelzang-van Trierum SE, Gil P, Camerer E, Martinez D, et al. Plasminogen controls inflammation and pathogenesis of influenza virus infections via fibrinolysis. PLoS Pathog 9 (3):e1003229, 2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Engelmann B, Massberg S. Thrombosis as an intravascular effector of innate immunity. Nat Rev Immunol 13 (1):34–45, 2013. [DOI] [PubMed] [Google Scholar]
- 34.Alhamdi Y, Toh CH. Recent advances in pathophysiology of disseminated intravascular coagulation: the role of circulating histones and neutrophil extracellular traps. F1000Res 6:2143, 2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Gando S, Otomo Y. Local hemostasis, immunothrombosis, and systemic disseminated intravascular coagulation in trauma and traumatic shock. Crit Care 19 (1):72, 2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Uaprasert N, Moonla C, Sosothikul D, Rojnuckarin P, Chiasakul T. Systemic coagulopathy in hospitalized patients with coronavirus disease 2019: a systematic review and meta-analysis. Clin Appl Thromb Hemost 27:1076029620987629, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Jin S, Jin Y, Xu B, Hong J, Yang X. Prevalence and impact of coagulation dysfunction in COVID-19 in China: a meta-analysis. Thromb Haemost 120 (11):1524–1535, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Matthay MA, Zemans RL, Zimmerman GA, Arabi YM, Beitler JR, Mercat A, Herridge M, Randolph AG, Calfee CS. Acute respiratory distress syndrome. Nat Rev Dis Primers 5 (1):18, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Dang CV, Bell WR, Kaiser D, Wong A. Disorganization of cultured vascular endothelial cell monolayers by fibrinogen fragment D. Science 227 (4693):1487–1490, 1985. [DOI] [PubMed] [Google Scholar]
- 40.Leavell KJ, Peterson MW, Gross TJ. The role of fibrin degradation products in neutrophil recruitment to the lung. Am J Respir Cell Mol Biol 14 (1):53–60, 1996. [DOI] [PubMed] [Google Scholar]
- 41.Nicolai L, Leunig A, Brambs S, Kaiser R, Weinberger T, Weigand M, Muenchoff M, Hellmuth JC, Leddeross S, Schulz H, et al. Immunothrombotic Dysregulation in COVID-19 pneumonia is associated with respiratory failure and coagulopathy. Circulation 142 (12):1176–1189, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Middleton EA, He XY, Denorme F, Campbell RA, Ng D, Salvatore SP, Mostyka M, Baxter-Stollzius A, Borczuk AC, Loda M, et al. Neutrophil extracellular traps contribute to immunothrombosis in COVID-19 acute respiratory distress syndrome. Blood 136 (10):1169–1179, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hill JD, Ratliff JL, Parrott JC, Lamy M, Fallat RJ, Koeniger E, Yaeger EM, Whutmer G. Pulmonary pathology in acute respiratory insufficiency: lung biopsy as a diagnostic tool. J Thorac Cardiovasc Surg 71 (1):64–71, 1976. [PubMed] [Google Scholar]
- 44.Greene R, Zapol WM, Snider MT, Reid L, Snow R, O’Connell RS, Novelline RA. Early bedside detection of pulmonary vascular occlusion during acute respiratory failure. Am Rev Respir Dis 124 (5):593–601, 1981. [DOI] [PubMed] [Google Scholar]
- 45.Lang M, Som A, Mendoza DP, Flores EJ, Reid N, Carey D, Li MD, Witkin A, Rodriguez-Lopez JM, Shepard JQ, et al. Hypoxaemia related to COVID-19: vascular and perfusion abnormalities on dual-energy CT. Lancet Infect Dis 20 (12):1365–1366, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Grasselli G, Tonetti T, Protti A, Langer T, Girardis M, Bellani G, Laffey J, Carrafiello G, Carsana L, Rizzuto C, et al. Pathophysiology of COVID-19-associated acute respiratory distress syndrome: a multicentre prospective observational study. Lancet Respir Med 8 (12):1201–1208, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Santamarina MG, Boisier Riscal D, Beddings I, Contreras R, Baque M, Volpacchio M, Martinez Lomakkin F. COVID-19: what iodine maps from perfusion CT can reveal-A prospective cohort study. Crit Care 24 (1):619, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Busana M, Giosa L, Cressoni M, Gasperetti A, Di Girolamo L, Martinelli A, Sonzogni A, Lorini L, Palumbo MM, Romitti F, et al. The impact of ventilation-perfusion inequality in COVID-19: a computational model. J Appl Physiol 130 (3):865–876, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, Vanstapel A, Weriein C, Stark H, Tzankov A, et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med 383:120–128, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Fox SE, Akmatbekov A, Harbert JL, Li G, Quincy Brown J, Vander Heide RS. Pulmonary and cardiac pathology in African American patients with COVID-19: an autopsy series from New Orleans. Lancet Respir Med 8 (7):681–686, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Schurink B, Roos E, Radonic T, Barbe E, Bouman CSC, de Boer HH, de Bree GJ, Bulle EB, Aronica EM, Florquin S, et al. Viral presence and immunopathology in patients with lethal COVID-19: a prospective autopsy cohort study. Lancet Microbe 1:e290–299, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Dolhnikoff M, Duarte-Neto AN, de Almeida Monteiro RA, da Silva LFF, de Oliveira EP, Saldiva PHN, Mauad T, Negri EM. Pathological evidence of pulmonary thrombotic phenomena in severe COVID-19. J Thromb Haemost 18 (6):1517–1519, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Leppkes M, Knopf J, Naschberger E, Lindemann A, Singh J, Herrmann I, Stüzl M, Staats L, Mahajan A, Schauer C, et al. Vascular occlusion by neutrophil extracellular traps in COVID-19. EBioMedicine 58:102925, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wu C, Chen X, Cai Y, Xia J, Zhou X, Xu S, Huang H, Zhang L, Zhou X, Du C, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med 180 (7):934–943, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Lin J, Yan H, Chen H, He C, Lin C, He H, Zhang S, Shi S, Kin K. COVID-19 and coagulation dysfunction in adults: a systematic review and meta-analysis. J Med Virol 93:934–944, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Vidali S, Morosetti D, Cossu E, Luisi MLE, Pancani S, Semeraro V, Consales G. D-dimer as an indicator of prognosis in SARS-CoV-2 infection: a systematic review. ERJ Open Res 6 (2):00260–2020, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bansal A, Singh AD, Jain V, Aggarwal M, Gupta S, Padappayil RP, Nadeem M, Joshi S, Mian A, Greathouse T, et al. The association of D-dimers with mortality, intensive care unit admission or acute respiratory distress syndrome in patients hospitalized with coronavirus disease 2019 (COVID-19): A systematic review and meta-analysis. Heart Lung 50 (1):9–12, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Leentjens J, van Haaps TF, Wessels PF, Schutgens REG, Middeldorp S. COVID-19-associated coagulopathy and antithrombotic agents-lessons after 1 year. Lancet Haematol S2352-3026 (21):00105–00108, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Santoro F, Núñez-Gil IJ, Viana-Llamas MC, Maroun Eid C, Romero R, Fernández Rozas I, Aparisi A, Becerra-Munoz VM, Garcia AM, Huang J, et al. Anticoagulation therapy in patients with coronavirus disease 2019: results from a multicenter international prospective registry (Health Outcome Predictive Evaluation for Corona Virus Disease 2019 [HOPE-COVID19]). Crit Care Med 49 (6):e624–e633, 2021. [DOI] [PubMed] [Google Scholar]
- 60.Nadkarni GN, Lala A, Bagiella E, Chang HL, Moreno PR, Pujadas E, Arvind V, Bose S, Chamey AW, Chen MD, et al. Anticoagulation, bleeding, mortality, and pathology in hospitalized patients with COVID-19. J Am Coll Cardiol 76 (16):1815–1826, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chiumello D, Busana M, Coppola S, Romitti F, Formenti P, Bonifazi M, Pozzi T, Palumbo MM, Cressoni M, Hermann P, et al. Physiological and quantitative CT-scan characterization of COVID-19 and typical ARDS: a matched cohort study. Intensive Care Med 46 (12):2187–2196, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Goligher EC, Ranieri VM, Slutsky AS. Is severe COVID-19 pneumonia a typical or atypical form of ARDS? And does it matter? Intensive Care Med 47 (1):83–85, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Fan E, Beitler JR, Brochard L, Calfee CS, Ferguson ND, Slutsky AS, Brodie D. COVID-19-associated acute respiratory distress syndrome: is a different approach to management warranted? Lancet Respir Med 8 (8):816–821, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Robba C, Battaglini D, Ball L, Patroniti N, Loconte M, Brunetti I, Vena A, Glaccobe DR, Bassetti M, Rocco PRM, et al. Distinct phenotypes require distinct respiratory management strategies in severe COVID-19. Respir Physiol Neurobiol 279:103455, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Guillen-Guio B, Lorenzo-Salazar JM, Ma SF, Hou PC, Hernandez-Beeftink T, Corrales A, Garcia-Laorden MI, Jou J, Epinosa E, Muirei A, et al. Sepsis-associated acute respiratory distress syndrome in individuals of European ancestry: a genome-wide association study. Lancet Respir Med 8 (3):258–266, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Hernández-Beeftink T, Guillen-Guio B, Villar J, Flores C. Genomics and the acute respiratory distress syndrome: current and future directions. Int J Mol Sci 20 (16):4004, 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Pollard CA, Morran MP, Nestor-Kalinoski AL. The COVID-19 pandemic: a global health crisis. Physiol Genomics 52 (11):549–557, 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Nasa P, Azoulay E, Khanna AK, Jain R, Gupta S, Javeri Y, Juneja D, Rangappa P, Sundararajan K, Alhazzani W, et al. Expert consensus statements for the management of COVID-19-related acute respiratory failure using a Delphi method. Crit Care 25 (1):106, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Camporota L, Chiumello D, Busana M, Gattinoni L, Marini JJ. Pathophysiology of COVID-19-associated acute respiratory distress syndrome. Lancet Respir Med 9 (1):e1, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ranjeva S, Pinciroli R, Hodell E, Mueller A, Hardin CC, Thompson BT, Berra L. Identifying clinical and biochemical phenotypes in acute respiratory distress syndrome secondary to coronavirus disease. EClinicalMedicine 34:100829, 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]