SIGNIFICANCE
This investigation examines the effectiveness of several common contact lens solutions in the disinfection of Acanthamoeba, which causes a serious eye infection most often resulting from dysfunctional or improper use of contact lens products.
PURPOSE
Acanthamoeba keratitis is an eye infection caused by a free-living amoeba, which can lead to extensive corneal damage and frequently blindness. Acanthamoeba keratitis is linked with contact lens use combined with noncompliance with contact lens care cleaning regimens. The patient's choice and use of multipurpose solutions (MPSs) continue to be a risk factor for Acanthamoeba keratitis. Thus, it is critical that the Acanthamoeba disinfection efficacy of the popular MPSs be determined. Here we compare the efficacy of seven major MPSs on the global market.
METHODS
Using standard methods of Acanthamoeba disinfection and quantification, Acanthamoeba ATCC 30461, 30868, 50370, and 50676 trophozoites were inoculated into each MPS and held for the manufacturer's recommended disinfection time. Acanthamoeba recovery plates were incubated for 14 days, after which positive wells were identified and cell concentrations determined using the 50% endpoint method.
RESULTS
Members of the OPTI-FREE products (Express, Replenish, and Puremoist [Alcon, Fort Worth, TX]) demonstrated significantly higher percentages of antimicrobial activity compared with the renu Advanced Formula (Bausch + Lomb, Rochester, NY), Biotrue (Bausch + Lomb), Acuvue RevitaLens (Johnson & Johnson, Santa Ana, CA), and Lite products (Cooper Vision, Scottsville, NY) for four of the trophozoite strains tested.
CONCLUSIONS
Many of the popular MPS biocides maintain little or no antimicrobial activity against Acanthamoeba trophozoites, and the number of biocides in an MPS does not necessarily indicate its antimicrobial activity.

Acanthamoeba is a pervasive microorganism. However, Acanthamoeba keratitis is a fortunately rare condition, as this infection can lead to blindness in as many as 41% of the afflicted patients.1,2 Outbreaks of Acanthamoeba keratitis in the United States in 20073 and in the United Kingdom since 20104 have been directly linked to particular multipurpose solution contact lens care products.3–6 These outbreaks resulted in the withdrawal of those products.3–6 Although the current International Standard (ISO 14729)7 does not presently recommend acanthamoebicidal testing, this standard is currently being updated by the American National Standards Institute and the International Standards Committee.8
The differences in the products' formulations and the biocides used in each product maintain the clear differentiation in the Acanthamoeba disinfection capabilities of each multipurpose solution. In general, Acanthamoeba is susceptible to some common biocides, which may have a variety of disinfection mechanisms. For instance, Acanthamoeba pseudopodia, nucleolar structure, mitochondria, and endoplasmic reticulum are susceptible to chlorhexidine gluconate.9 Chlorhexidine diacetate has also been shown to produce shrinkage from the cyst wall, whereas polyhexamethylene biguanide induced both withdrawal of the cytoplasm from the cyst wall and swelling of cysts.10 Finally, chlorine treatment has led to size reduction, permeabilization, and retraction of pseudopods.11
The biocides available for use within any multipurpose solution provide a unique challenge. Alongside the use of cleaning, wetting, and comfort agents, the biocides included in every multipurpose solution reach toward the goal of maintaining sufficient antimicrobial activity against potentially pathogenic microorganisms while promoting ocular health. Biocides effective against Acanthamoeba may not be suitable for patient safety as part of a multipurpose solution because of the potential for corneal damage. Furthermore, because Acanthamoeba is a unique pathogen in the field of potentially infectious ocular agents, biocides that may be effective against other common microorganisms and can be included in multipurpose solutions may have little to no effect against amoeba in either the trophozoite or cyst formation.12,13 Therefore, the goal of this study was to determine the Acanthamoeba disinfection efficacy of seven of the most common multipurpose solutions on the global market.
METHODS
As previously described,14,15 axenic culture media (containing 20 g biosate peptone, 5 g glucose, 0.3 KH2PO4, 10 μg vitamin B12, and 15 mg l-methionine per liter of distilled deionized water) was used to produce homogenous populations of Acanthamoeba trophozoites. Axenic culture media was adjusted to a pH of 6.6 to 6.95 with 1 M of NaOH and autoclaved at 121°C for 20 minutes before storing at room temperature for use within 2 months. One-fourth Ringer's solution was used to harvest organisms and for seeding trophozoites into 96-well plates.
Antimicrobial efficacy of contact lens disinfecting solutions against Acanthamoeba trophozoites was conducted as previously published,14 per a modified version of ISO standard 14729. Acanthamoeba strains were obtained from ATCC (American Type Culture Collection, Manassas, VA): ATCC 30461 (Acanthamoeba polyphaga, group T4, isolated from human corneal scrapings; Houston, TX, 1973), ATCC 30868 (Acanthamoeba castellanii, group T4, isolated from human cornea; Cambridge, England, 1974), ATCC 50370 (A. castellanii, group T4, isolated from human eye infection; New York, NY, 1978), and ATCC 50676 (Acanthamoeba mauritaniensis, group T4, isolated from human eye infection; Namibia or South Africa, 1990). These strains belong to the T4 group, which is the most commonly associated genotype with Acanthamoeba keratitis.16Acanthamoeba trophozoites were subcultured in axenic media with the final 24 hours of growth in fresh media to promote uniform Acanthamoeba trophozoite proliferation, before testing. After scale-up, cells were collected and centrifuged at 500g for 5 minutes at room temperature, and washed three times with Ringer's solution. Pellets were then resuspended in Ringer's solution, and the count seeding density was confirmed via hemocytometer. Trophozoites were inoculated into each multipurpose solution for a final cell density between 2 × 105 and 5 × 105 cells per well. Each multipurpose solution was held at room temperature for the manufacturer's soaking time (Table 1). At disinfection time, 1 mL of multipurpose solution was placed in 9 mL of neutralizing broth (Ringer's solution with lecithin and polysorbate 80 to neutralize quaternaries, phenolics, hexachlorophene, and formalin)17 and serially diluted in Ringer's solution. Each dilution was plated in quadruplicate on a 12-well plate containing 2 mL of nonnutrient agar with 100 μL of Escherichia coli (108 colony-forming unit/mL; ATCC 8739). Plates were incubated for 14 days at 28°C ± 2°C. After incubation, positive wells were identified and surviving cells quantified using the 50% end point following the Reed and Muench computation.18 This computation calculates the concentration of a test substance or culture that produces an effect of interest in half of the test units. Antimicrobial efficacy was determined by calculating the log and percent reduction of the cell densities recovered from the multipurpose solution at disinfection time, compared with the inoculum control of the respective strain. Each Acanthamoeba strain was tested in one to two independent trials, in triplicate, on different days, and the results were averaged. All multipurpose solutions were tested simultaneously using the same inoculum stock as a direct comparison. To understand the differences between groups, log reduction quantifications were analyzed via the Student two-tailed t test and are represented as mean ± standard error.
TABLE 1.
Multipurpose solutions used and their manufacturers, biocides, and stated disinfection times
| Contact lens care product | Manufacturer | Biocides | Disinfection time (h) |
|---|---|---|---|
| OPTI-FREE Puremoist | Alcon, Fort Worth, TX | Polyquaternium-1 (0.001%), myristamidopropyl dimethylamine (0.0006%) | 6 |
| OPTI-FREE Express | Alcon, Fort Worth, TX | Polyquaternium-1 (0.001%), myristamidopropyl dimethylamine (0.0005%) | 6 |
| OPTI-FREE Replenish | Alcon, Fort Worth, TX | Polyquaternium-1 (0.001%), myristamidopropyl dimethylamine (0.0005%) | 6 |
| Acuvue RevitaLens | Johnson & Johnson, New Brunswick, NJ | Polyquaternium-1 (0.0003%), alexidine dihydrochloride (0.00016%) | 6 |
| renu Advanced Formula | Bausch + Lomb, Rochester, NY | Polyquaternium (0.00015%), alexidine dihydrochloride (0.0002%), polyaminopropyl biguanide (0.00005%) | 4 |
| Biotrue | Bausch + Lomb, Rochester, NY | Polyaminopropyl biguanide (0.00013%), polyquaternium (0.0001%) | 4 |
| Lite | CooperVision, Lake Forest, CA | Polyhexanide (0.0001%) | 6 |
To obtain images of the Acanthamoeba following disinfection efficacy of each solution, propidium iodide staining (as propidium iodide only binds to exposed cellular DNA of amoeba with damaged cell walls; Invitrogen, Carlsbad, CA) was used with three of the Acanthamoeba strains: ATCC 30461, ATCC 50370, and ATCC 50676.15 Propidium iodide staining was performed on separate cultures from those used for quantification via the 50% endpoint method. Briefly, Acanthamoeba were seeded into a black clear-bottom 96-well plate at a density of 1 × 104 cells per well. Cells were allowed to adhere for 2 hours. Media was removed, and multipurpose solutions were added to appropriate wells (0.2 mL/well) in 15 replicates to confirm that representative images were indicative of quantitative log calculations. Blanks and untreated Acanthamoeba wells were included as controls. Following the multipurpose solution manufacturer's listed disinfection time, multipurpose solutions were removed, and 0.2 mL of 2 μg/mL propidium iodide diluted in ¼ Ringer's solution was added to each well of the 15 replicates. This was followed by confocal imaging (Nikon Ti Eclipse Microscope; Nikon, Minato City, Tokyo, Japan) at ×20 magnification using the Nikon NIS-Elements platform.
RESULTS
To determine the Acanthamoeba disinfection efficacy of each multipurpose solution, trophozoites were tested in each multipurpose solution according to the manufacturer's stated disinfection time at room temperature (Fig. 1, Table 2). The disinfection efficacy of each multipurpose solution was determined by calculating the percent reduction compared with the inoculum control. Inoculum controls for each strain and replicate ranged between 2 × 105 and 3.8 × 105 log cells/mL. Each multipurpose solution was tested against ATCC 30461 (Fig. 1A), ATCC 30868 (Fig. 1B), ATCC 50370 (Fig. 1C), ATCC 50676 (Fig. 1D). In all four strains, OPTI-FREE Express Multi-Purpose Disinfecting Solution and OPTI-FREE Puremoist Multi-Purpose Disinfecting Solution (Alcon, Fort Worth, TX) maintained significantly higher antimicrobial activity than the renu Advanced Formula (P < .05; Bausch + Lomb, Rochester, NY), Biotrue (P < .05; Bausch + Lomb), and Lite products (P < .05; Cooper Vision, Scottsville, NY). In the ATCC 30461 strain, OPTI-FREE Express Multi-Purpose Disinfecting Solution and OPTI-FREE Puremoist Multi-Purpose Disinfecting Solution maintained a significantly higher antimicrobial activity than Acuvue RevitaLens (P < .001; Johnson & Johnson, Santa Ana, CA). This was also true for strain ATCC 50370 (P < .001) and in strain ATCC 30868 (P ≤ .001). Similarly, for the ATCC 30461 strain, OPTI-FREE Replenish Multi-Purpose Disinfecting Solution demonstrated significantly greater antimicrobial activity versus Biotrue (P = .003); for the ATCC 30868 strain versus renu Advanced Formula (P = .04) and Lite (P = .003); for the ATCC 50370 strain versus renu Advanced Formula (P = .005), Biotrue (P < .001), Acuvue RevitaLens (P < .001), and Lite (P < .001); and for the ATCC 50676 strain versus Biotrue (P = .004) and Lite (P = .001).
FIGURE 1.


OPTI-FREE products (Express, Replenish, and Puremoist Multi-Purpose Disinfecting Solutions) maintain a significantly higher percentage of Acanthamoeba antimicrobial efficacy vs. other global market products. Quantifications are represented as mean ± standard error percent reduction vs. inoculum controls. All seven products were tested against ATCC 30461 (A), ATCC 30868 (B), ATCC 50370 (C), and ATCC 50676 (D). n = 3 to 6 per group. aaP < .05 vs. renu Advanced Formula, aP < .005 vs. renu Advanced Formula, bP < .005 vs. Biotrue, cP < .005 vs. Acuvue RevitaLens, and dP < .005 vs. Lite.
TABLE 2.
CIs for multipurpose solution disinfection efficacy comparisons with P < .05, as related to Figs. 1 to 4
| Contact lens care product | Compared with | Strain tested | 95% CI |
|---|---|---|---|
| OPTI-FREE Express MPS | renu Advanced Formula | ATCC 30461 | −3.127 to −1.674 |
| OPTI-FREE Express MPS | Biotrue | ATCC 30461 | −4.244 to −2.983 |
| OPTI-FREE Express MPS | Acuvue RevitaLens | ATCC 30461 | −3.681 to −2.394 |
| OPTI-FREE Express MPS | Lite | ATCC 30461 | −3.325 to −2.018 |
| OPTI-FREE Replenish MPS | Biotrue | ATCC 30461 | −1.885 to −0.5141 |
| OPTI-FREE Puremoist MPS | renu Advanced Formula | ATCC 30461 | −3.102 to −1.701 |
| OPTI-FREE Puremoist MPS | Biotrue | ATCC 30461 | −4.215 to −3.014 |
| OPTI-FREE Puremoist MPS | Acuvue RevitaLens | ATCC 30461 | −3.653 to −2.425 |
| OPTI-FREE Puremoist MPS | Lite | ATCC 30461 | −3.253 to −2.092 |
| OPTI-FREE Express MPS | renu Advanced Formula | ATCC 30868 | −3.459 to −1.079 |
| OPTI-FREE Express MPS | Biotrue | ATCC 30868 | −4.129 to −2.045 |
| OPTI-FREE Express MPS | Acuvue RevitaLens | ATCC 30868 | −4.129 to −2.045 |
| OPTI-FREE Express MPS | Lite | ATCC 30868 | −4.938 to −2.917 |
| OPTI-FREE Replenish MPS | renu Advanced Formula | ATCC 30868 | 0.067 to 1.535 |
| OPTI-FREE Replenish MPS | Lite | ATCC 30868 | −1.235 to −0.479 |
| OPTI-FREE Puremoist MPS | renu Advanced Formula | ATCC 30868 | −3.385 to −2.091 |
| OPTI-FREE Puremoist MPS | Biotrue | ATCC 30868 | −3.852 to −3.260 |
| OPTI-FREE Puremoist MPS | Acuvue RevitaLens | ATCC 30868 | −3.852 to −3.260 |
| OPTI-FREE Puremoist MPS | Lite | ATCC 30868 | −4.457 to −4.246 |
| OPTI-FREE Express MPS | renu Advanced Formula | ATCC 50370 | −1.789 to −0.560 |
| OPTI-FREE Express MPS | Biotrue | ATCC 50370 | −2.113 to −1.237 |
| OPTI-FREE Express MPS | Acuvue RevitaLens | ATCC 50370 | −1.841 to −0.349 |
| OPTI-FREE Express MPS | Lite | ATCC 50370 | −2.117 to −1.780 |
| OPTI-FREE Replenish MPS | renu Advanced Formula | ATCC 50370 | −1.598 to −0.373 |
| OPTI-FREE Replenish MPS | Biotrue | ATCC 50370 | −1.920 to −1.055 |
| OPTI-FREE Replenish MPS | Acuvue RevitaLens | ATCC 50370 | −1.648 to −0.753 |
| OPTI-FREE Replenish MPS | Lite | ATCC 50370 | −1.958 to −1.623 |
| OPTI-FREE Puremoist MPS | renu Advanced Formula | ATCC 50370 | −2.076 to −0.0660 |
| OPTI-FREE Puremoist MPS | Biotrue | ATCC 50370 | −2.430 to −1.308 |
| OPTI-FREE Puremoist MPS | Acuvue RevitaLens | ATCC 50370 | −2.155 to −1.008 |
| OPTI-FREE Puremoist MPS | Lite | ATCC 50370 | −2.753 to −1.591 |
| OPTI-FREE Express MPS | renu Advanced Formula | ATCC 50676 | −1.855 to −0.039 |
| OPTI-FREE Express MPS | Biotrue | ATCC 50676 | −3.367 to −2.041 |
| OPTI-FREE Express MPS | Lite | ATCC 50676 | −4.977 to −3.098 |
| OPTI-FREE Replenish MPS | Biotrue | ATCC 50676 | −3.318 to −1.240 |
| OPTI-FREE Replenish MPS | Lite | ATCC 50676 | −4.846 to −2.379 |
| OPTI-FREE Puremoist MPS | renu Advanced Formula | ATCC 50676 | −1.913 to −0.575 |
| OPTI-FREE Puremoist MPS | Biotrue | ATCC 50676 | −3.255 to −2.742 |
| OPTI-FREE Puremoist MPS | Lite | ATCC 50676 | −5.045 to −3.618 |
CI = confidence interval; MPS = multipurpose solution.
For three of these strains, representative images of the antimicrobial efficacy were obtained using a novel rapid method, which was recently outlined.15 Propidium iodide, which stains red any cellular DNA components that are exposed because of cell death and loss of cell membrane integrity, or spilled because of cell lysis, was used to examine Acanthamoeba cultures immediately after completion of the multipurpose solution exposure time as a separate qualitative examination from the log reduction quantitative experiments. These representative examinations were performed for all seven multipurpose solutions with the ATCC 30461 strain (Fig. 2), the ATCC 50370 strain (Fig. 3), and the ATCC 50676 strain (Fig. 4). The control samples (after 6 hours of incubation) demonstrated minimal red staining. However, natural cell death is evident even in the control samples, indicating a base level of expected mortality without intervention. Like the quantifications noted in Fig. 1A, the Lite multipurpose solution demonstrated a strong degree of cell death within the ATCC 30461 strain (Fig. 2), but not in the ATCC 50370 or 50676 strains (Figs. 3, 4). Acuvue RevitaLens and renu Advanced Formula seemed to cause the highest amount of cell death within the ATCC 50676 strain (Fig. 4), but not the other two strains. Biotrue seemed to induce a minimal amount of cell death in all three of the tested strains compared with the other products. Among all three strains, the three OPTI-FREE products demonstrated a high amount of red staining, indicating high amounts of cell death. Overall, these visual observations were in line with the 50% endpoint quantifications found in Fig. 1.
FIGURE 2.

Representative images of ATCC 30461 Acanthamoeba cell death after exposure to multipurpose solutions. Propidium iodide staining results in red stain indicating cell death or lysis, whereas gray color indicates living cells. Scale bar, 100 μm; ×20 magnification.
FIGURE 3.
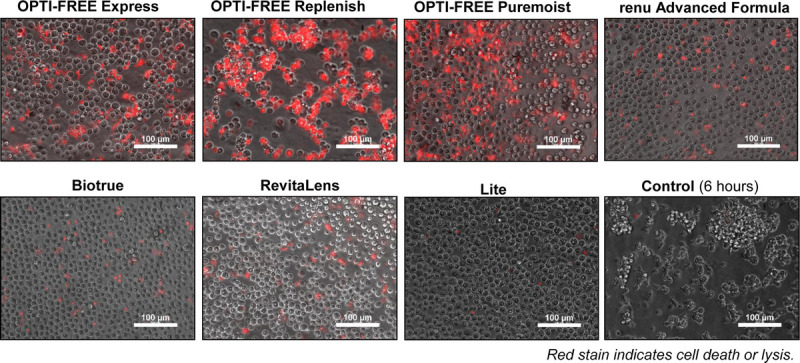
Representative images of ATCC 50370 Acanthamoeba cell death after exposure to multipurpose solutions. Propidium iodide staining results in red stain indicating cell death or lysis, whereas gray color indicates living cells. Scale bar, 100 μm; ×20 magnification.
FIGURE 4.

Representative images of ATCC 50676 Acanthamoeba cell death after exposure to multipurpose solutions. Propidium iodide staining results in red stain indicating cell death or lysis, whereas gray color indicates living cells. Scale bar, 100 μm; ×20 magnification.
DISCUSSION
Acanthamoeba keratitis is a dangerous ocular infection largely because of the difficulties in diagnosing, treating, and managing the disease. Critically, it can lead to severe corneal damage and, in many cases, permanent blindness without corneal transplant.1,2 Although it is a fortunately rare affliction, misuse of contact lens care products or ineffective contact lens care products have been directly linked to Acanthamoeba keratitis infections and outbreaks.3–6 Thus, it is integral to public health that information regarding the efficacy of common contact lens multipurpose solutions be both robust and available. The goal of this study was therefore to use widely reported methods of Acanthamoeba testing and quantification19–24 resulting from multiple replicates, using both standard and clinical strains of Acanthamoeba,25–27 and a wide range of multipurpose solutions on the global market. Notably, this study focuses on the trophozoite form of Acanthamoeba, as the biocides in non-hydrogen peroxide–based systems have been shown to be ineffective against the Acanthamoeba cyst.28
The field of Acanthamoeba keratitis research and contact lens disinfection agents sometimes contains contentious or differing results, often attributable to varying methods of investigation and quantification. Fortunately, the methods used in this investigation demonstrated similar results to what has been reported for other global products.29 Furthermore, despite a field of varied reports, some consistencies in the use of certain biocides against Acanthamoeba persist. For instance, the OPTI-FREE products have been previously demonstrated as highly effective against Acanthamoeba trophozoites.12,30,31 OPTI-FREE products have also previously been found to produce little or no encystment,32,33 ensuring that the more vulnerable trophozoite form persists and is able to be acted upon by multipurpose solutions. Finally, the biocides used in the OPTI-FREE products, namely, polyquaternium-1 and myristamidopropyl dimethylamine, have been shown to have the greatest efficacy against Acanthamoeba (and other ophthalmological pathogens) as compared with other biocides.34–36
The results of the current investigation indicate similar results: products containing polyquaternium-1 and myristamidopropyl dimethylamine demonstrated significantly greater Acanthamoeba trophozoite disinfection efficacy than other biocides or combinations of biocides. According to the results demonstrated here, less effective biocides include polyaminopropyl biguanide or alexidine dihydrochloride combined with polyquaternium or polyquaternium-1. In addition, although having a greater number of biocides does not necessarily infer greater antimicrobial efficacy, as in the case of renu Advanced Formula, the higher concentration of polyquaternium-1 in the OPTI-FREE products does seem to impart a higher percentage of disinfection. Indeed, products containing 0.001% polyquaternium versus those containing less (0.0003% in Acuvue RevitaLens, 0.00015% in renu Advanced Formula, 0.0001% in Biotrue) maintained significantly greater antimicrobial activity. In addition, the OPTI-FREE products are the only products examined that contain myristamidopropyl dimethylamine. These data imply that either polyquaternium acts on Acanthamoeba in a dose-dependent manner, whereas other biocides are ineffective against this particular pathogen, or myristamidopropyl dimethylamine is a critical component to Acanthamoeba antimicrobial activity, or both.
The visual examination of the antimicrobial activity of these multipurpose solutions demonstrated similar results to the 50% endpoint quantifications performed here. Propidium iodide staining has been previously established as an efficient method for identifying dead or lysed amoeboid cells.15,37,38 Visually, it is easy to assess that there is very little cell death in control samples (although a minor amount of cell death persists because of natural cell cycles) based on the very small amount of red staining. Furthermore, the visual observations gathered from these representative stains are in agreement with published data regarding the morphological changes of Acanthamoeba after exposure to biocides, such as a rounding of the cell wall, retraction of pseudopodia, and encystment.11,39,40 The renu Advanced Formula, Biotrue, Acuvue RevitaLens, and Lite products (which contain a maximum of three parts per million of polyquaternium and/or any other biocide) overall demonstrated a low to moderate amount of propidium iodide staining between the three Acanthamoeba strains examined. In general, the antimicrobial quantifications aligned with the visual representations of Acanthamoeba cell death. Notably, within each of these multipurpose solutions, it was clear in both the 50% endpoint quantifications and the visual staining that the antimicrobial activity of each multipurpose solution can be altered by which Acanthamoeba strain is tested, as the results were not universal between strains. However, even within the differences noticed between strains, the OPTI-FREE products, which contain 10 parts per million of polyquaternium-1, consistently demonstrated both quantifiably high levels of antimicrobial activity and visually the greatest amount of propidium iodide-based cell death.
In conclusion, the difference and concentration of biocides between multipurpose solutions are critically important in the endeavor to determine the contact lens care product antimicrobial activity. After the examination of multiple laboratory and clinical isolates of Acanthamoeba, and seven of the most widely used multipurpose solutions on the global market, this investigation demonstrated that the biocides used within the OPTI-FREE products are the most efficacious regarding Acanthamoeba trophozoite disinfection.
Footnotes
Funding/Support: This work was funded by Alcon Research, LLC.
Conflict of Interest Disclosure: All authors are employees of Alcon Research LLC. The authors have full access and control of the data reported in the study and take full responsibility for their presentation in this article. Alcon Research LLC funded the study.
Study Registration Information: No study registration or protocol. No human subjects.
Author Contributions and Acknowledgments: Conceptualization: RW, EM, AC, MMG, PS, CM, MC; Data Curation: RW, EM, AC, CM, MC; Formal Analysis: EM, AC, MMG, MC; Investigation: EM, AC, MMG, PS, CM, MC; Methodology: RW, EM, AC, MMG, PS, CM, MC; Project Administration: PS, MC; Resources: PS, MC; Supervision: MC; Validation: RW, MC; Visualization: MMG, CM, MC; Writing – Original Draft: RW, AC; Writing – Review & Editing: RW, EM, MMG, PS, CM, MC.
The authors wish to thank Valerie Harris, Megan Thomas, Jamie King, and Melissa Martin for their technical assistance.
Contributor Information
Rhonda Walters, Email: rhonda.walters@alcon.com.
Elise Miller, Email: elise.miller@alcon.com.
Allison Campolo, Email: allison.campolo@alcon.com.
Manal M. Gabriel, Email: manal.gabriel@alcon.com.
Paul Shannon, Email: stephen.shannon@alcon.com.
Cindy McAnally, Email: Cynthia.mcanally@alcon.com.
REFERENCES
- 1.Szentmary N Daas L Shi L, et al. Acanthamoeba Keratitis—Clinical Signs, Differential Diagnosis and Treatment. J Curr Ophthalmol 2019;31:16–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Scruggs BA Quist TS Salinas JL, et al. Notes from the Field: Acanthamoeba Keratitis Cases—Iowa, 2002–2017. MMWR Morb Mortal Wkly Rep 2019;68:448–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Verani JR Lorick SA Yoder JS, et al. National Outbreak of Acanthamoeba Keratitis Associated with Use of a Contact Lens Solution, United States. Emerg Infect Dis 2009;15:1236–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Carnt N Hoffman JJ Verma S, et al. Acanthamoeba Keratitis: Confirmation of the UK Outbreak and a Prospective Case-Control Study Identifying Contributing Risk Factors. Br J Ophthalmol 2018;102:1621–8. [DOI] [PubMed] [Google Scholar]
- 5.Datta A, Willcox MD, Stapleton F. In Vivo Efficacy of Silver-impregnated Barrel Contact Lens Storage Cases. Cont Lens Anterior Eye 2021;44:101357. [DOI] [PubMed] [Google Scholar]
- 6.Tu EY, Joslin CE. Recent Outbreaks of Atypical Contact Lens–related Keratitis: What Have We Learned? Am J Ophthalmol 2010;150:602–8.e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.International Organization for Standardization (ISO) . Ophthalmic Optics—Contact Lens Care Products—Microbiological Requirements and Test Methods for Products and Regimens for Hygienic Management of Contact Lenses; ISO 14729:2001/A1:2010. Geneva, Switzerland: ISO; 2010. [Google Scholar]
- 8.American National Standards Institute (ANSI) . Minutes of the Parent Committee Meeting, February 27, 2018, Clearwater Beach, Florida; ASC Z80. Available at: https://www.thevisioncouncil.org/sites/default/files/ASCZ80_ParentCommitteeMinutes_February_27_2018_FINALMar19-2018.pdf. Accessed June 14, 2021.
- 9.Fatima H, Nakisah MA. Visualization on the Effect of Chlorhexidine Gluconate, a Biocide on Acanthamoeba sp. by Electron Microscopy. Malaysian J Microscopy 2013;9:154–9. [Google Scholar]
- 10.Khunkitti W Hann AC Lloyd D, et al. Biguanide-induced Changes in Acanthamoeba castellanii: An Electron Microscopic Study. J Appl Microbiol 1998;84:53–62. [DOI] [PubMed] [Google Scholar]
- 11.Mogoa E Bodet C Morel F, et al. Cellular Response of the Amoeba Acanthamoeba castellanii to Chlorine, Chlorine Dioxide, and Monochloramine Treatments. Appl Environ Microbiol 2011;77:4974–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Borazjani RN, Kilvington S. Efficacy of Multipurpose Solutions against Acanthamoeba species. Cont Lens Anterior Eye 2005;28:169–75. [DOI] [PubMed] [Google Scholar]
- 13.Shoff ME Joslin CE Tu EY, et al. Efficacy of Contact Lens Systems against Recent Clinical and Tap Water Acanthamoeba Isolates. Cornea 2008;27:713–9. [DOI] [PubMed] [Google Scholar]
- 14.Crary MJ Walters R Shannon P, et al. Variables Affecting the Recovery of Acanthamoeba Trophozoites. Pathogens 2021;10:221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Campolo A, Shannon P, Crary M. Evaluating Alternate Methods of Determining the Antimicrobial Efficacy of Contact Lens Care Products against Acanthamoeba Trophozoites. Pathogens 2021;10:126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Arnalich-Montiel F Lumbreras-Fernández B Martín-Navarro CM, et al. Influence of Acanthamoeba Genotype on Clinical Course and Outcomes for Patients with Acanthamoeba Keratitis in Spain. J Clin Microbiol 2014;52:1213–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rutala WA Weber DJ, Healthcare Infection Control Practices Advisory Committee (HICPAC), Guideline for Disinfection and Sterilization in Healthcare Facilities, 2008 . US Department of Health and Human Services, Centers of Disease Control and Prevention (CDC). Available at: https://www.in.gov/health/files/Tab_1_Resource_CD.pdf. Accessed June 14, 2021.
- 18.Reed LJ, Muench H. A Simple Method of Estimating Fifty per Cent Endpoints. Am J Epidemiol 1938;27:493–7. [Google Scholar]
- 19.Niszl IA, Markus MB. Anti-Acanthamoeba Activity of Contact Lens Solutions. Br J Ophthalmol 1998;82:1033–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hughes R, Kilvington S. Comparison of Hydrogen Peroxide Contact Lens Disinfection Systems and Solutions against Acanthamoeba polyphaga. Antimicrob Agents Chemother 2001;45:2038–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hiti K Walochnik J Haller-Schober EM, et al. Viability of Acanthamoeba After Exposure to a Multipurpose Disinfecting Contact Lens Solution and Two Hydrogen Peroxide Systems. Br J Ophthalmol 2002;86:144–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kilvington S, Lam A. Development of Standardized Methods for Assessing Biocidal Efficacy of Contact Lens Care Solutions against Acanthamoeba Trophozoites and Cysts. Invest Ophthalmol Vis Sci 2013;54:4527–37. [DOI] [PubMed] [Google Scholar]
- 23.Johnston SP Sriram R Qvarnstrom Y, et al. Resistance of Acanthamoeba Cysts to Disinfection in Multiple Contact Lens Solutions. J Clin Microbiol 2009;47:2040–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kolar SS Manarang JC Burns AR, et al. Contact Lens Care Solution Killing Efficacy against Acanthamoeba castellanii by in Vitro Testing and Live-imaging. Cont Lens Anterior Eye 2015;38:442–50. [DOI] [PubMed] [Google Scholar]
- 25.Thomson S Rice CA Zhang T, et al. Characterisation of Sterol Biosynthesis and Validation of 14α-Demethylase as a Drug Target in Acanthamoeba. Sci Rep 2017;7:8247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Dobrowsky PH, Khan S, Khan W. Resistance of Legionella and Acanthamoeba mauritaniensis to Heat Treatment as Determined by Relative and Quantitative Polymerase Chain Reactions. Environ Res 2017;158:82–93. [DOI] [PubMed] [Google Scholar]
- 27.Alves Dde S Moraes AS Alves LM, et al. Experimental Infection of T4 Acanthamoeba Genotype Determines the Pathogenic Potential. Parasitol Res 2016;115:3435–40. [DOI] [PubMed] [Google Scholar]
- 28.Rayamajhee B Willcox MD Henriquez FL, et al. Acanthamoeba Keratitis: An Increasingly Common Infectious Disease of the Cornea. Lancet Microbe 2021;2:E345–6. [DOI] [PubMed] [Google Scholar]
- 29.Borazjani RN, Kilvington S. Effect of a Multipurpose Contact Lens Solution on the Survival and Binding of Acanthamoeba Species on Contact Lenses Examined with a No-rub Regimen. Eye Contact Lens 2005;31:39–45. [DOI] [PubMed] [Google Scholar]
- 30.Lakhundi S, Khan NA, Siddiqui R. Inefficacy of Marketed Contact Lens Disinfection Solutions against Keratitis-causing Acanthamoeba castellanii Belonging to the T4 Genotype. Exp Parasitol 2014;141:122–8. [DOI] [PubMed] [Google Scholar]
- 31.Kal A, Toker MI, Kaya S. The Comparison of Antimicrobial Effectiveness of Contact Lens Solutions. Int Ophthalmol 2017;37:1103–14. [DOI] [PubMed] [Google Scholar]
- 32.Lonnen J Heaselgrave W Nomachi M, et al. Disinfection Efficacy and Encystment Rate of Soft Contact Lens Multipurpose Solutions against Acanthamoeba. Eye Contact Lens 2010;36:26–32. [DOI] [PubMed] [Google Scholar]
- 33.Padzik M Chomicz L Szaflik JP, et al. In Vitro Effects of Selected Contact Lens Care Solutions on Acanthamoeba castellanii Strains in Poland. Exp Parasitol 2014;145(Suppl):S98–101. [DOI] [PubMed] [Google Scholar]
- 34.Codling CE, Maillard JY, Russell AD. Aspects of the Antimicrobial Mechanisms of Action of a Polyquaternium and an Amidoamine. J Antimicrob Chemother 2003;51:1153–8. [DOI] [PubMed] [Google Scholar]
- 35.Moon EK Lee S Quan FS, et al. Effect of 2, 6-Dichlorobenzonitrile on Amoebicidal Activity of Multipurpose Contact Lens Disinfecting Solutions. Korean J Parasitol 2018;56:491–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Gabriel MM, McAnally C, Bartell J. Antimicrobial Efficacy of Multipurpose Disinfecting Solutions in the Presence of Contact Lenses and Lens Cases. Eye Contact Lens 2018;44:125–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hillmann F Novohradská S Mattern DJ, et al. Virulence Determinants of the Human Pathogenic Fungus Aspergillus fumigatus Protect against Soil Amoeba Predation. Environ Microbiol 2015;17:2858–69. [DOI] [PubMed] [Google Scholar]
- 38.Radosa S Ferling I Sprague JL, et al. The Different Morphologies of Yeast and Filamentous Fungi Trigger Distinct Killing and Feeding Mechanisms in a Fungivorous Amoeba. Environ Microbiol 2019;21:1809–20. [DOI] [PubMed] [Google Scholar]
- 39.Khunkitti W Lloyd D Furr JR, et al. Acanthamoeba castellanii: Growth, Encystment, Excystment and Biocide Susceptibility. J Infect 1998;36:43–8. [DOI] [PubMed] [Google Scholar]
- 40.Mogoa E Bodet C Legube B, et al. Acanthamoeba castellanii: Cellular Changes Induced by Chlorination. Exp Parasitol 2010;126:97–102. [DOI] [PubMed] [Google Scholar]


