Abstract
Background:
Pertussis is a current contagious bacterial disease caused by Bp. Given the prevalence of pertussis, development of new vaccines is important. This study was attempted to evaluate the expression of main virulence factors (PTX, PRN, and FHA) from Bp predominant strains and also compare the expression of these factors in the OMVs obtained from predominant circulating Bp isolate.
Methods:
The physicochemical features of the prepared OMVs were analyzed by electron microscopy and SDS-PAGE. The presence of the mentioned virulence factors was confirmed by Western blotting. BALB/c mice (n = 21) immunized with characterized OMVs were challenged intranasally with sublethal doses of Bp, to examine their protective capacity.
Results:
Electron microscopic examination of the OMVs indicated vesicles within the range of 40 to 200 nm. SDS-PAGE and Western blotting demonstrated the expression of all three main protective immunogens (PTX, PRN, and FHA), prevalent in the predominant, challenge, and vaccine strains, and OMVs of the predominant IR37 strain and BP134 vaccine strain. Significant differences were observed in lung bacterial counts between the immunized mice with OMV (30 CFU/lung) compared to the negative control group ((6 104 CFU/lung; p < 0.001). In mice immunized with OMVs (3 µg), the number of lungs recovered colonies after five days dropped at least five orders of magnitude compared to the control group.
Conclusion:
OMVs obtained from circulating isolates with the predominant profile may constitute a highly promising vaccine quality. They also can be proposed as a potential basic material for the development of new pertussis vaccine candidate.
Key Words: Bordetella pertussis, Vaccines, Virulence factors
INTRODUCTION
Pertussis or whooping cough is a current respiratory bacterial disease caused by Bp. Before the introduction of high efficient vaccines in the second half of the 20th century, pertussis was recognized as a childhood disease with high mortality rates. Later, following the success of the infant immunization programs, pertussis was speculated to be under control[1]. Unexpectedly, during the last decade, the disease was still among the public-health problems, particularly in countries with a regular vaccination program[2,3]. Recently, a large number of pertussis outbreaks were observed in several countries[4,5]. Although it is not a life-threatening disease in adults, the affected people can act as reservoirs for the transmission of the disease among children[6-8]. Based on existing evidence, the resurgence of the disease is prevalent mostly in developing countries and developed world[9,10] due to adaptation of pathogen populations, reduced long-term immunity of pertussis vaccine in prolonged period of time, and the use of aP instead of wP[10,11].
Studies have reported genetic differences between the current circulating Bp and vaccine strains, particularly in genes encoding the vaccine component proteins, including PTX, (an AB5 toxin composed of the A [S1] subunit, and the B oligomer) and adhesions, including PRN, FHA, and fimbriae. These differences could result in inadequate immunity induction to protect against new isolates[11,12]. This genetic adaptation can affect bacterial protein expression, as well as vaccine-induced immunity against pertussis[10,13,14].
The re-emergence of the disease has pointed out the need for the development of new vaccines that are able to overcome the disadvantages of the conventional vaccines[15]. To this end, the OMVs obtained from Bp are possible vaccine candidates that can successfully avoid the weaknesses of current vaccines against Bp [16-18]. These vesicles naturally contain bacterial surface antigens and exhibit an acceptable level of protection. It is worth noting that the new vaccine, established based on circulating strains isolated from patients, conferred protection against circulating Bp isolates and also induced long-lasting immunity[18-20]. Since virulence factors have been considered as the main protective antigens to the formulation of the pertussis vaccine, studies on the expressed protein level could help to provide an insight into the pertussis resurgence problem[21,22]. Considering the above-mentioned data, the current study aimed to evaluate the expression of main virulence factors, including PTX, PRN, and FHA from Bp predominant strains and compare to the expression of these factors in the OMVs obtained from predominant circulating Bp isolate. In addition, the physicochemical properties of OMVs, as a probable vaccine candidate, were assessed.
MATERIALS AND METHODS
Bacteria and culture medium
Bacterial strains used in this study consisted of four predominant isolates (IR37, IR77, IR124, and IR148). The challenge strain (18283) and the vaccine strain (BP134) were obtained from the Pertussis Reference Laboratory at Pasteur Institute of Iran (Tehran) and the RVSRI, respectively. The bacteria were cultured on BGA supplemented with 10% defibrinated sheep blood[23,24].
Isolation of OMVs and preparation of growth curve
OMWs were obtained from bacterial cells. Briefly, bacteria were sub-cultured into MSS liquid medium containing methyl-β-cyclodextrin at 36 °C with aeration at 150 rpm (HYSC, Korea)[24]. Since the time of OMV harvesting is very crucial for the product yield, it is important to determine the bacterial growth curve before OMV extraction. For this purpose, after 21-h incubation at 3 ×g and 36 ºC, the culture was inoculated into a fresh MSS medium at OD600 = 0.05, 3×g and 36 ºC. Thereafter, the samples were collected continuously at fixed intervals for 70 h to plot the growth curve. At the end of the logarithmic phase, (OD600 = 0.75-1), 10 ml was inoculated into a 500-ml fresh MSS medium with initial turbidity at OD600 = 0.05 and then incubated at 3 ×g at 36ºC for 34 h[25]. OMVs were prepared as previously described with modifications to avoid the need for ultracentrifuge with high speed[24-26]. Briefly, the bacterial strains were pelleted from MSS (at 8000 ×g at 4 °C for 30 min). The pellets were washed with TE and centrifuged, and then the supernatants were pelleted at 60,000 ×g at 4 °C for 2 h. Subsequently, these resulting pellets were resuspended in a TE buffer containing deoxycholate (5 g/L) and mixed again several times by pipetting to make a homogenized suspension. The mixture was incubated for 10 min and then centrifuged at 60,000 g at 4 °C for 2 h. Afterward, the supernatant was separated carefully in a new tube and treated with TE buffer and centrifuged again at 60,000 ×g at 4 °C for 1 h. The pellets were suspended in 6 ml of 3% sucrose and filtered with 0.22-µm pore size filters (polyvinylidene difluoride, syringe filters, Sigma-Aldrich, Germany)[24,27]. The OMV sample was inactivated in a bain-marie (Memmert, Germany) at 56 °C for 30 min. The sterility and viability of the samples were controlled by incubating the suspension on BGA and blood agar plates. The OMVs were examined by TEM[24-26].
Gel electrophoresis and Western blot analysis
The four predominant strains, challenge strain, vaccine strain were grown on BGA. A loop full of Bp bacteria from a 48h-old culture on BGA was taken and resuspended in 1 ml of PBS and incubated at 56 °C for 30 min. The suspensions were then diluted in a sample buffer and boiled for 15 min before being subjected to electrophoresis[26], In addition, OMVs (5 µg) obtained from predominant and vaccine strains were resuspended in a sample buffer, boiled by heating at 100 °C for 10 min and run on 10% SDS gels[24]. Electrophoresis was performed at RT with constant voltage of 80 V. Polypeptides were stained with a solution of Coomassie Blue R250 (DNA biotech, Iran) with gentle shaking for 1 h. The gels were then washed three times in methanol 40% (v/v) and acetic acid 10% (v/v) in double distilled water for 20 min[28]. For Western blot analysis, the separated protein bands were transferred from polyacrylamide SDS gels to a polyvinylidene difluoride membrane. The membranes were then blocked with 5% skim milk in PBS overnight, washed three times with PBS in 0.05% Tween 20 and incubated with PRN, FHA, and PTX monoclonal antibodies (NIBSC No. 97/558, 97/564, and 97/572, respectively) at RT for 1 h. The washing steps were repeated thrice, followed by incubation with horseradish peroxidase-conjugated rabbit anti-sheep antibody (PADZA Company, Iran) at a 1:1000 dilution, at RT for 1 h. After additional three times washing with PBST, the membranes were developed with Metal Enhanced DAB Substrate kit (Sigma-Aldrich)[ 28].
LAL test
The LPS toxicity content in OMVs was determined by the LAL test. The chromogenic LAL assay (Thermo Fisher Scientific, USA) was performed to determine the presence of LPS and measure the endotoxins in the samples[29,30].
Animal model
Female BALB/c mice at the age of 6-8 weeks old (weighing 15-20 g) were provided by RVSRI. In order to be adapted to the environment, all the animals were kept under standard conditions one week before the experiments.
MWG test
The safety of OMVs obtained from predominant strain IR37 was determined by the MWG test, according to the WHO guidelines[31]. A group of eight BALB/c mice were injected intraperitoneally with detoxified OMVs acquired from circulating strain with predominant profile (IR37). OMVs were detoxified with 0.4% formalin. Two different concentrations of OMVs (3 µg and 20 µg in 40 µl PBS) were used in this experiment, along with PBS as negative control. The weight of mice was measured at specific intervals (16 h, three and seven days). The non-toxicity of extracted OMVs were confirmed by passing the WHO requirement as follows: (1) the total weight of the mice from the vaccine group three days after injection was higher than or same as the initial weight; (2) the weight gain average was compared with the control group at the end of the day seven, which should not be less than 60% of the control group, based on the WHO guidelines; (3) more than 95% of the animals survived during the test. The side effects of the injection site and the rate of mortality were examined, as well. ANOVA test was used to compare the data obtained[28,31,32].
Immunization regimen
To evaluate the immunological responses to OMVs obtained from the predominant strain IR37 of Bp, vaccination was carried out in three groups (seven mice per group). Each immunization dose for injection comprised 3 µg of protein in 40 µl of PBS, according to the result of the MWG test[32]. The mice were injected with OMVs, PBS as negative control, and commercial Tdap vaccine (ADACEL, Sanofi Pasteur, France; 1:10 of human equivalent dose) as the positive control. All the mice were immunized intraperitoneally on days 0, 14, and 28[27,32].
B. pertussis challenge
To evaluate the immunization efficacy, the mice were challenged with the intranasal injection of sublethal dose challenge strain 18323) 1.4 × 107 Bp( two weeks after the last immunization. For bacterial count, the lungs of the injected mice were collected after seven days. The lungs were homogenized in the sterile PBS and were diluted, and then cultured on BGA containing 10% defibrinated sheep blood to determine bacterial recoveries. The experiments were performed three times[27,31,32].
Statistical analysis
Standard deviations and means were calculated from Log10-transformed CFU numbers. Differences between means were analyzed by one-way ANOVA with statistical significance accepted at the p< 0.05 level[28].
Ethical statement
All animal experiments, including toxicity MWG test, immunization, and challenge test were approved by the Ethical Committee of Animal Care of RVSRI and Shahid Beheshti University (Ethical code: IR.SBU.REC.1398.014).
RESULTS
In liquid cultures, various parameters including medium composition, stress conditions, and cell harvest time, affect the amount of bacterial OMVs vesiculation. Considering the importance of cell harvest time in the process of OMV preparation, our result showed that the optimum time for collecting OMVs would be the end of the logarithmic phase, as in this time point the bacterial growth will be stopped and t the OMV extraction process started. The turbidity of liquid culture was approximately 0.7 to 1 at 600 nm wavelengths, which was about 30 h after the initial culture for vaccine strain and about 36 h for predominant strain IR37 (Fig. 1)[25]. The OMVs were obtained from predominant strain IR37 and vaccine strain BP134 as previously described. The electron microscopic examinations indicated that the mean size of extracted OMVs were 70 nm and ranged from 40 to 200 nm (Fig. 2). The OMV extraction procedure repeated five times, and in all cases the results were similar.
Fig. 1.

The growth curve of the circulating strain BP IR37 and vaccine strain BP134. Inoculations were carried out in triplicate, and the OD was recorded at 600 nm
Fig. 2.

Negatively stained Bordetella pertussis OMVs examined with an electron microscope
Samples of bacteria, including the four predominant strains, challenge strain, vaccine strain and OMV, were obtained from predominant strain. Vaccine strains were screened by SDS-PAGE and Western immunoblot for the expression of PTX, PRN, and FHA. Western immunoblot results showed the presence of all three main protective immunogens in all predominant strains, challenge strain, vaccine strain, and also in the OMVs of predominant strain IR37 and OMVs of vaccine strain BP134 (Figs. 3-5).
Fig. 3.
Western blot of samples. Lane 1-4, predominant strains (IR148, IR77, IR124, and IR37); lane 5, challenge strain; lane 6, vaccine strain; M, molecular marker
Fig. 5.
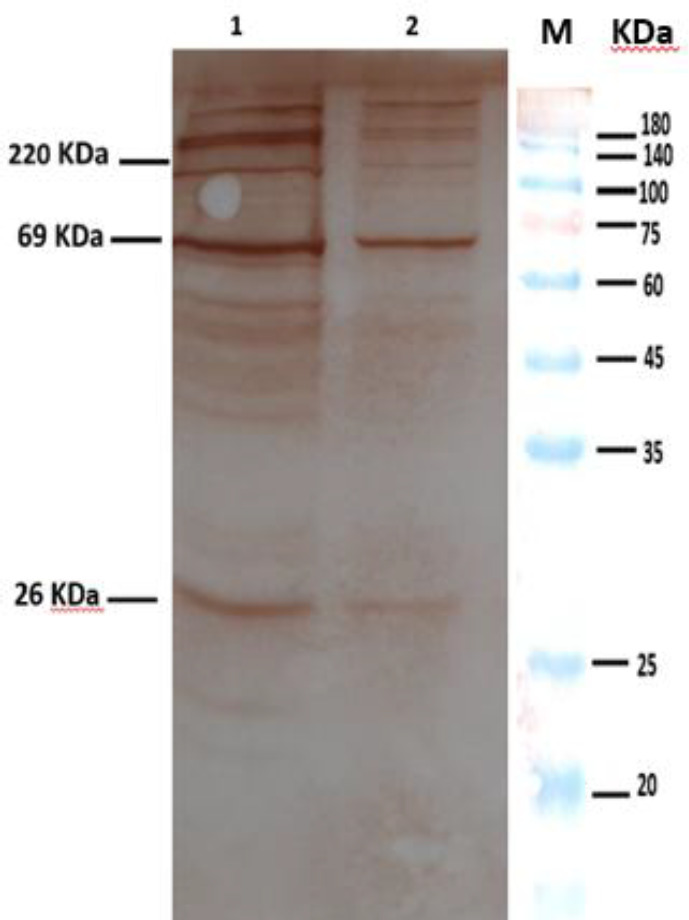
Western blot of OMVs using anti-FHA, anti-PRN, and anti-PTX. Lane 1, the OMV obtained from vaccine strain; lane 2, the OMV obtained from predominant strain IR37; M, molecular marker
The LPS toxicity of the OMV final lot samples was tested in a fivefold dilution and contained 15,000 EU/ml. The endotoxin activity was within the range of DPT/polio vaccines and is therefore regarded as safe[31]. The MWG test is used to evaluate the toxicity of the injected OMVs. According to Table 1, the extracted OMVs were injected with a lower dose of 3 μg, which showed no AT, no significant weight loss of the mice, and no abnormal local tolerance at the injection site, and all mice survived during the experiment period. Therefore, it means the amounts of OMVs applied were non-toxic based on the WHO criteria for the MWG test[31]. However, the results of higher dose (20 µg) showed weight loss three days post vaccination. According to these observations, the safety dose of OMVs was about 3 µg. This amount of OMVs considered for further examination.
Table 1.
Toxicity test (mouse weight gain test)
| Dose of infection | Average weight gain per mouse (g) | ||
|---|---|---|---|
| Day 3 | Day 7 | Weight gain compared to the control (%) | |
| 3 | 0.8 | 1 | 74 |
| 20 | -1.4 | -0.5 | -32 |
| PBS | 0.6 | 1.4 | ---- |
The average weight gain per mouse was determined after three and seven days after intraperitoneal injection.
Fig. 4.

The SDS-PAGE pattern of OMVs containing FHA, PRN, and PTX. Lane 1, the OMV obtained from vaccine strain; lane 2, the OMV obtained from predominant strain IR37; M, molecular marker
Murine challenge
Based on the immunization program, seven BALB/c mice per group were immunized either by OMVs extracted from predominant strain or positive control, as well as negative control., The immunized mice were then challenged with IR37 strain intranasally at a concentration of 1.4 × 107, two weeks after immunization. The survival time was also monitored for the next 10 days (Fig. 6). As expected, significant differences in lung bacterial counts between the control group and immunized mice were observed (p < 0.001). In mice injected with PBS, the number of recovered colonies was 6 104 CFU/lung, while in mice immunized with OMVs (3 µg), the number of recovered colonies (30 CFU/lung) after five days dropped at least 3 orders of magnitude in comparison to the negative control group.
Fig. 6.

Comparison of the lung clearance efficiency of OMVs with Tdap and PBS
DISCUSSION
Currently, there are two different kinds of pertussis vaccines. The wP that is the first generation vaccine developed in the 1940s and comprises of killed bacteria. Despite the reactogenicity and side effects, this type of vaccine has resulted in a significant decrease in pertussis incidence. However, the above mentioned drawbacks encouraged the researchers to introduce the second generation pertussis vaccine, namely aP, which was developed in the early 1990s. The aP vaccines include three to five antigens purified from Bp and are broadly used around the world; however, these vaccines have some disadvantages such as low efficacy and short-term immunity[33,34].
Despite vaccination against pertussis with a high rate of coverage, there are concerns regarding the recurrence of the disease[2,21]. Among the possible explanations for the re-emergence of the disease, pathogen adaptation is one of the main issues. Investigations focusing on pathogen adaptation have reported the antigenic divergence between the isolates used for vaccine development and the currently circulating isolates in different regions, affecting the expression of virulence factors[3,35]. This antigenic divergence commonly occurs during the bacterial genome evolution to escape from the immune system, which increases the adaptation of the bacterial population. It leads to waning the efficacy of the vaccine-induced immunity against the new isolates over the time and decreases the period in which pertussis vaccine is effective[36,37].
In a study performed by Fry et al.[38]on the allelic shift of the PTX gene, ptxA1 had the most allelic diversity. Similar allelic diversity in different countries has been reported, as well. Herein, the ptxA1, which was different from the vaccine strain alleles, was the predominant circulating allele in different countries. Mooi et al.[35,39,40] demonstrated polymorphisms in vaccine antigen genes such as ptxP, ptxA, prn, and fim.
In Iran, the disease incidence has been increasing despite the high rate (more than 96%) of wP vaccination using the same strains in vaccines without any changes during the last 60 years[41]. According to previous studies conducted in Iran, the allelic profiles of predominant circulating isolates were ptxA1, ptxP3, prn2, fim2-1, and fim3-2. It is important to note that this pattern is not completely equivalent to the vaccine strain BP134 profiles ptxP1, ptxA2, prn1, and fim3-2[41-43]. Moreover, The presence of ptxP3, ptxA1, and prn2 alleles results in enhancing the capacity of the strain to spread among the immunized populations[12,44]. Another genomic change in Bp isolates has been the deletion of the PRN gene. PRN is the main antigen component of the pertussis vaccines, and those Bp strains that lack the expression of PRN protein are called PRN negative[45,46]. Therefore, it was important to evaluate the presence of PRN and other virulence factors in the native predominant strains.
In this research, some circulating isolates with predominant profiles were selected from the Biobank of the Pasteur Institute of Iran, which based on Western blot results, all expressed PTX, PRN, FHA similar to vaccine strains and challenge strain (Fig. 3). Despite the presence of main protective antigens to overcome the drawbacks related to the conventional vaccines, using predominant circulating strains in vaccine might be effective to decrease the incidence of pertussis and potentially eradicate Bp worldwide[47,48]. In this regard, the use of predominant strain in vaccine development will be a first step forward to control pertussis worldwide.
One of the drawbacks of aP pertussis vaccines is decreased vaccine efficacy, due to the waning vaccine immunity by the rapid reduction of antibodies following vaccination and long-term immunity[11]. Although aP vaccines provide immunity against specific antigens used in the vaccine formulation, wP vaccines provide immunity against the most common antigens of bacterial whole cells[11,49]. aP vaccines also elicit humoral immune response Th2/Th17. Because Bp is an intracellular bacterium, the cellular immune response Th1/Th17 plays a main role in the clearance of the bacteria from the respiratory tract. Moreover, the humoral response is an insufficient immune response needed for complete protective immunity against pertussis[49,50]. Thus, studies have attempted to develop the third generation of new protein-based vaccines, such as OMVs vaccines, which besides having several immunogenic proteins, they can induce Th2 , Th1, and Th17 responses[50,51]. A significant alternative protective subunit is the OMV produced by the Gram-negative bacteria as a natural budding element, which can be obtained by detergent extraction from Bp[52]. The component of the OMVs is complex, which the main constituents include outer membrane proteins, periplasmic proteins, phospholipids, lipopolysaccharides, and nucleic acids. The interaction of the above components with each other determines the physicochemical properties of OMV and its stability[17].
The size range of OMVs also indicated that OMVs were nanoparticles that can be taken up by the antigen-presenting cells[53]. OMVs act as pathogen-associated molecular patterns, which are detected by the immune system and elicit the innate immune response and have adjuvant activity, as well[54]. The OMV vaccines can be considered effective and also safe vaccines[17,55]. The first attempts to apply OMVs in the field of vaccines were in response to the serogroup B meningococcal disease by the Finlay Institute in Cuba[56].
In recent years, numerous studies around the world have evaluated the potential use of OMVs, as vaccine candidate for pertussis[16,17,19,27,54]. Selective pressure on circulating strains cannot affect OMV contents, therefore making the emergence of escape isolates more difficult, as was observed for pertussis aP vaccines made with purified protein antigen[17,51].
In this study, OMVs were obtained from Bp strain IR37 with predominant genomic profile and also vaccine strain BP134. OMVs were extracted by the modified method to avoid the need for ultracentrifuge with high speed, which is expensive and is not generally available in laboratories and many research centers[26]. The TEM result indicated that the extracted OMVs were stable in conformation, the same as previous studies[19,27].
Since OMVs have the ability to induce immune response as an immunogen, due to carrying several important antigens, the presence of the main virulence factors such as PTX, PRN, and FHA in the OMVs is very important[16,27]. In this context, we investigated the expression of the PTX, PRN, and FHA in OMVs obtained from predominant Bp and vaccine strains. Our results confirmed the existence of these antigens in investigated OMVs. The presence of surface immunogens such as PTX, PRN, and FHA allowed us to verify that the OMVs obtained from the outer membrane, as described before[16]. The OMV extraction procedure repeated five times, and in all cases, the same results were observed, which was similar to the previous studies[18,27]. Furthermore, the presence of main virulence factors was detected in all samples. The safety of using these extracted OMVs was evaluated by the abnormal toxicity test. The LAL test results showed that the extracted OMVs were safe[30]. The MWG test, which is often used to determine the toxicity of the injected OMVs, using low dose (3 μg), showed no difference in the weight gain curve, meaning that this amount of OMVs passed toxicity criteria successfully and considered as non-toxic. This finding is in agreement with former studies[19,32]. The results indicated that the samples were not contaminated with impurities during the OMV preparation process.
In order to determine the protection capacity induced by extracted OMVs, animal studies using intranasal Bp challenge were conducted. Adequate clearance rates (p< 0.005) were detected in mice immunized with extracted OMVs and commercial Tdap as a positive control. However, in mice injected with PBS (as negative control), the number of recovered colonies was high ((6 104 per lung).). Our results showed statistically significant differences between immunized animals with OMVs and the control group (p < 0.001), which is comparable to other studies[18,27,32].
In conclusion, according to OMVs features and due to the data presented herein and previous studies, it can be suggested that OMV-based vaccines derived from circulating isolates with the predominant profile may constitute a very promising vaccine quality. Moreover, it could be proposed as a potential basic material for developing the new generation of pertussis vaccine candidate and could be successful in the disease control.
CONFLICT OF INTEREST.
None declared.
ACKNOWLEDGEMENTS
The authors would like to appreciate the staff of the Aerobic Bacterial Research and Vaccine Production Department and Department of Immunology and Microbiology of Razi Vaccine and Serum Research Institute (Karaj, Iran), and Microbiology Department of Pasteur Institute of Iran (Tehran) for their cooperation.
References
- 1.Kuchar E, Karlikowska-Skwarnik M, Han S, Nitsch-Osuch A. Pertussis: history of the disease and current prevention failure. Advances in experimental medicine biology. 2016;934:77–82. doi: 10.1007/5584_2016_21. [DOI] [PubMed] [Google Scholar]
- 2.Sala-Farré MR, Arias-Varela C, Recasens-Recasens A, Simó-Sanahuja M, Muñoz-Almagro C, Pérez-Jové J. Pertussis epidemic despite high levels of vaccination coverage with a cellular pertussis vaccine. Enfermedades infecciosasy microbiologia clinica. 2015;33(1):27–31. doi: 10.1016/j.eimc.2013.09.013. [DOI] [PubMed] [Google Scholar]
- 3.Barkoff AM, He Q. Molecular epidemiology of Bordetella pertussis. Advances in experimental medicine biology. 2019;1183:19–33. doi: 10.1007/5584_2019_402. [DOI] [PubMed] [Google Scholar]
- 4.Clark TA. Changing pertussis epidemiology: everything old is new again. The journal of infectious diseases. 2014;209(7):978–981. doi: 10.1093/infdis/jiu001. [DOI] [PubMed] [Google Scholar]
- 5.Sealey KL, Belcher T, Preston A. Bordetella pertussis epidemiology and evolution in the light of pertussis resurgence. Infection, genetics and evolution. 2016;40:136–143. doi: 10.1016/j.meegid.2016.02.032. [DOI] [PubMed] [Google Scholar]
- 6.Hoey J. Pertussis in adults. CMAJ. 2003;168(4):453–454. [PMC free article] [PubMed] [Google Scholar]
- 7.Guiso N. Whooping cough from infants to adults. Bulletin de l'Academie nationale de medecine. 2008;192(7):1437–1449. [PubMed] [Google Scholar]
- 8.Tan T, Dalby T, Forsyth K, Halperin SA, Heininger U, Hozbor D, Plotkin S, Ulloa-Gutierrez R, Wirsing von König CH. Pertussis across the globe: recent epidemiologic trends from 2000 to 2013. The pediatric infectious disease journal. 2015;34(9):222–232. doi: 10.1097/INF.0000000000000795. [DOI] [PubMed] [Google Scholar]
- 9.Bart MJ, van Gent M, van der Heide HG, Boekhorst J, Hermans P, Parkhill J, Mooi FR. Comparative genomics of prevaccination and modern Bordetella pertussis strains. BMC genomics. 2010;11:1–8. doi: 10.1186/1471-2164-11-627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Mooi F, Van Der Maas NA, De Melker HE. Pertussis resurgence: waning immunity and pathogen adaptation-two sides of the same coin. Epidemiology and infection. 2014;142(4):685–694. doi: 10.1017/S0950268813000071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bart MJ, Harris SR, Advani A, Arakawa Y, Bottero D Bouchez V, Cassiday PK, Chiang CS, Dalby T, Fry NK, Gaillard ME, van Gent M, Guiso N, Hallander HO, Harvill ET, He Q, van der Heide HG, Heuvelman K, Hozbor DF, Kamachi K, Karataev GI, Lan R, Lutyńska A, Maharjan RP, Mertsola J, Miyamura T, Octavia S, Preston A, Quail MA, Sintchenko V, Stefanelli P, Tondella ML, Tsang RS, Xu Y, Yao SM, Zhang S, Parkhill J, Mooi FR. Global population structure and evolution of Bordetella pertussis and their relationship with vaccination. MBio. 2014;5(2):1–9. doi: 10.1128/mBio.01074-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Pawloski L, Queenan AM, Cassiday PK, Lynch AS, Harrison MJ, Shang W, Williams MM, Bowden KE, Burgos-Rivera B, Qin X, Messonnier N, Tondella ML. Prevalence and molecular characterization of pertactin-deficient Bordetella pertussis in the United States. Clinical and vaccine immunology. 2014;21(2):119–125. doi: 10.1128/CVI.00717-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Williamson YM, Moura H, Whitmon J, Woolfitt AR, Schieltz DM, Rees JC, Guo S, Kirkham H, Bouck D, Ades EW, Tondella ML, Carlone GM, Sampson JS, Barr JR. A proteomic characterization of Bordetella pertussis clinical isolates associated with a California state pertussis outbreak. International journal of proteomics. 2015:2015: 1–12. doi: 10.1155/2015/536537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hozbor D. New Pertussis vaccines: A need and a challenge. Advances in experimental medicine and biology. 2019;1183:115–126. doi: 10.1007/5584_2019_407. [DOI] [PubMed] [Google Scholar]
- 15.Ormazábal M, Bartel E, Gaillard ME, Bottero D, Errea A, Zurita ME, Moreno G, Rumbo M, Castuma C, Flores D, Martín MJ, Hozbor D. Characterization of the key antigenic components of pertussis vaccine based on outer membrane vesicles. Vaccine. 2014;32(46):6084–6090. doi: 10.1016/j.vaccine.2014.08.084. [DOI] [PubMed] [Google Scholar]
- 16.Hozbor DF. Outer membrane vesicles: an attractive candidate for pertussis vaccines. Expert review of vaccines. 2017;16(3):193–196. doi: 10.1080/14760584.2017.1276832. [DOI] [PubMed] [Google Scholar]
- 17.Tan K, Li R, Huang X, Liu Q. Outer membrane vesicles: current status and future direction of these novel vaccine adjuvants. Frontiers in immunology. 2018;9:783–792. doi: 10.3389/fmicb.2018.00783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Gaillard ME, Bottero D, Errea A, Ormazábal M, Zurita ME, Moreno G, Rumbo M, Castuma C, Bartel E, Flores D, van der Ley P, van der Ark A, F Hozbor D. Acellular pertussis vaccine based on outer membrane vesicles capable of conferring both long-lasting immunity and protection against different strain genotypes. Vaccine. 2014;32(8):931–937. doi: 10.1016/j.vaccine.2013.12.048. [DOI] [PubMed] [Google Scholar]
- 19.Zurita ME, Wilk MM, Carriquiriborde F, Bartel E, Moreno G, Misiak A, Mills KHG, Hozbor D. A pertussis outer membrane vesicle-based vaccine induces lung-resident memory CD4 T cells and protection against Bordetella pertussis, including pertactin deficient strains. Frontiers in cellular and infection microbiology. 2019;9:125–134. doi: 10.3389/fcimb.2019.00125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Carbonetti NH. Bordetella pertussis: new concepts in pathogenesis and treatment. Current opinion in infectious diseases. 2016;29(3):287–294. doi: 10.1097/QCO.0000000000000264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Smith AM, Guzmán CA, Walker MJ. The virulence factors of Bordetella pertussis: a matter of control. FEMS microbiology review. 2001;25(3):309–333. doi: 10.1111/j.1574-6976.2001.tb00580.x. [DOI] [PubMed] [Google Scholar]
- 22.Mooi FR, He Q, van Oirschot H, Mertsola J. Variation in the Bordetella pertussis virulence factors pertussis toxin and pertactin in vaccine strains and clinical isolates in Finland. Infection and immunity. 1999;67(6):3133–3134. doi: 10.1128/iai.67.6.3133-3134.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Badamchi A, Bahrami F, Tasbiti AH, Yari S, Shafiei M, Shahcheraghi F, Siadat SD. Immuno-proteomics analysis between OMV of vaccine and dominant wild type strains of Bordetella pertussis in Iran. Iranianjournal of microbiology. 2020;12(2):77–88. [PMC free article] [PubMed] [Google Scholar]
- 24.Hozbor D, Rodriguez ME, Fernández J, Lagares A, Guiso N, Yantorno O. Release of outer membrane vesicles from Bordetella pertussis. Current microbiology. 1999;38(5):273–278. doi: 10.1007/pl00006801. [DOI] [PubMed] [Google Scholar]
- 25.Klimentová J, Stulík J. Methods of isolation and purification of outer membrane vesicles from gram-negative bacteria. Microbiological research. 2015;170:1–9. doi: 10.1016/j.micres.2014.09.006. [DOI] [PubMed] [Google Scholar]
- 26.Soltani MS, Eftekhar F, Noofeli M, Banihashemi SR, Shahcheraghi F. Comparison of two different methods in the extraction of outer membrane vesicles from the Bordetella pertussis as a vaccine candidate. Archive of Razi Institute. 2021;76(3):411–419. doi: 10.22092/ARI.2020.342861.1487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Roberts R, Moreno G, Bottero D, Gaillard ME, Fingermann M, Graieb A, Rumbo M, Hozbor D. Outer membrane vesicles as acellular vaccine against pertussis. Vaccine. 2008;26(36):4639–4646. doi: 10.1016/j.vaccine.2008.07.004. [DOI] [PubMed] [Google Scholar]
- 28.Weber C, Boursaux-Eude C, Coralie G, Caro V, Guiso N. Polymorphism of Bordetella pertussis isolates circulating for the last 10 years in France, where a single effective whole-cell vaccine has been used for more than 30 years. Journal of clinical microbiology. 2001;39(12):4396–4403. doi: 10.1128/JCM.39.12.4396-4403.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Surendran N, Pichichero M. Genetically detoxified pertussis toxin induces superior antigen specific CD4 T cell responses compared to chemically detoxified pertussis toxin. Human vaccines and immunotherapeutics. 2019;15(5):1167–1170. doi: 10.1080/21645515.2019.1565270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ochiai M, Kataoka M, Toyoizumi H, Kamachi K, Yamamoto A, Horiuchi Y. Evaluation of endotoxin content of diphtheria–tetanus-acellular pertussis combined (DTaP) vaccines that interfere with the bacterial endotoxin test. Vaccine. 2003;21:1862–1866. doi: 10.1016/s0264-410x(03)00006-9. [DOI] [PubMed] [Google Scholar]
- 31.Horiuchi Y, Takahashi M, Konda T, Ochiai M, Yamamoto Y, Kataoka M, Toyoizumi H, Arakawa Y. Quality control of diphtheria tetanus acellular pertussis combined (DTaP) vaccines in Japan. Japanese Journal of Infectious diseases. 2001;54:167–180. [PubMed] [Google Scholar]
- 32.Asensio CJ, Gaillard ME, Moreno G, Bottero D, Zurita E, Rumbo M, van der Ley P, van der Ark A, Hozbor D. Outer membrane vesicles obtained from Bordetella pertussis tohama expressing the lipid A deacylase pagL as a novel acellular vaccine candidate. Vaccine. 2011;29:1649–1656. doi: 10.1016/j.vaccine.2010.12.068. [DOI] [PubMed] [Google Scholar]
- 33.Kilgore PE, Salim AM, Zervos MJ, Schmitt HJ. Pertussis: microbiology, disease, treatment, and prevention. Clinical microbiology reviews. 2016;29:449–486. doi: 10.1128/CMR.00083-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Guiso N. Bordetella pertussis and pertussis vaccines. Clinical infectious diseases. 2009;49(10):1565–1569. doi: 10.1086/644733. [DOI] [PubMed] [Google Scholar]
- 35.Mooi FR, Van Loo I, King AJ. Adaptation of Bordetella pertussis to vaccination: a cause for its reemergence? Emerging infectious diseases. 2001;7(3 Suppl):526–531. doi: 10.3201/eid0707.017708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Althouse BM, Scarpino SV. Asymptomatic transmission and the resurgence of Bordetella pertussis. BMC medicine. 2015;13:1–8. doi: 10.1186/s12916-015-0382-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.He Q, Mertsola J. Factors contributing to pertussis resurgence. Future microbiology. 2008;3:329–339. doi: 10.2217/17460913.3.3.329. [DOI] [PubMed] [Google Scholar]
- 38.Fry NK, Neal S, Harrison TG, Miller E, Matthews R, George RC. Genotypic variation in the Bordetella pertussis virulence factors pertactin and pertussis toxin in historical and recent clinical isolates in the United Kingdom. Infection and immunity. 2001;69(9):5520–5528. doi: 10.1128/IAI.69.9.5520-5528.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Mooi FR, van Oirschot H, Heuvelman K, van der Heide HG, Gaastra W, Willems RJ. Polymorphism in the Bordetella pertussis virulence factors P 69/pertactin and pertussis toxin in the Netherlands: Temporal trends and evidence for vaccine-driven evolution. Infection and immunity. 1998;66(2):670–675. doi: 10.1128/iai.66.2.670-675.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Mooi FR, van Loo HMI, van Gent M, He Q, Bart MJ, Heuvelman KJ, de Greeff SC, Diavatopoulos D, Teunis P, Nagelkerke N, Mertsola J. Bordetella pertussis strains with increased toxin production associated with pertussis resurgence. Emerging infectious diseases. 2009;15(8):1206–1213. doi: 10.3201/eid1508.081511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Safarchi A, Octavia S, Nikbin VS, Lotfi MN, Zahraei SM, Tay CY, Lamichhane B, Shahcheraghi F, Lan R. Genomic epidemiology of Iranian Bordetella pertussis: 50 years after the implementation of whole cell vaccine. Emerging microbes and infections. 2019;8(1):1416–1427. doi: 10.1080/22221751.2019.1665479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Nikbin VS, Ahmadi NJ, Hosseinpour M, Lotfi NM, Shooraj F, Sadeghpour F, Shahcheraghi F. Virulence factors variation among Bordetella pertussis isolates in Iran. International journal of molecular and cellular medicine. 2015;4(2):138–144. [PMC free article] [PubMed] [Google Scholar]
- 43.Heravi FS, Nikbin VS, Lotfi MN, Badiri P, Ahmadi NJ, Zahraei SM, Shahcheraghi F. Strain variation and antigenic divergence among Bordetella pertussis circulating strains isolated from patients in Iran. European journal of clinical microbiology and infectious diseases. 2018;37(10):1893–1900. doi: 10.1007/s10096-018-3323-6. [DOI] [PubMed] [Google Scholar]
- 44.de Gouw D, Hermans PW, Bootsma HJ, Zomer A, Heuvelman K, Diavatopoulos DA, Mooi FR. Differentially expressed genes in Bordetella pertussis strains belonging to a lineage which recently spread globally. PLoS one. 2014;9(1):1–13. doi: 10.1371/journal.pone.0084523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Barkoff AM, Mertsola J, Pierar D, Dalby T, Hoegh SV, Guillot S, Stefanelli P, van Gent M, Berbers G, Vestrheim D, Greve-Isdahl M, Wehlin L, Ljungman M, Fry NK, Markey K, He Q. Pertactin-deficient Bordetella pertussis isolates: evidence of increased circulation in Europe, 1998 to 2015. Euro surveillance. 2019;24(7):1–8. doi: 10.2807/1560-7917.ES.2019.24.7.1700832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Williams MM, Sen K, Weigand MR, Skoff TH, Cunningham VA, Halse TA, Tondella MC. Bordetella pertussis strain lacking pertactin and pertussis toxin. Emerging infectious diseases. 2016;22(2):319–327. doi: 10.3201/eid2202.151332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Mills KH, Ross PJ, Allen AC, Wilk MM. Do we need a new vaccine to control the re-emergence of pertussis? Trends in microbiology. 2014;22(2):49–52. doi: 10.1016/j.tim.2013.11.007. [DOI] [PubMed] [Google Scholar]
- 48.Klein NP, Bartlett J, Rowhani-Rahbar A, Fireman B, Baxter R. Waning protection after fifth dose of acellular pertussis vaccine in children. The New England journal of medicine. 2012;367(11):1012–1019. doi: 10.1056/NEJMoa1200850. [DOI] [PubMed] [Google Scholar]
- 49.Warfel JM, Zimmerman LI, Merkel TJ. Acellular pertussis vaccines protect against disease but fail to prevent infection and transmission in a nonhuman primate model. Proceedings of the national academy of sciences of the United States of America. 2014;111(2):787–792. doi: 10.1073/pnas.1314688110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Locht C. Pertussis: acellular, whole-cell, new vaccines, what to choose? Expert review of vaccines. 2016;15(6):671–673. doi: 10.1586/14760584.2016.1161511. [DOI] [PubMed] [Google Scholar]
- 51.van der Ley P, van den Dobbelsteen G. Next-generation outer membrane vesicle vaccines against Neisseria meningitidis based on nontoxic LPS mutants. Human vaccines. 2011;7(8):886–890. doi: 10.4161/hv.7.8.16086. [DOI] [PubMed] [Google Scholar]
- 52.Schwechheimer C, Kuehn MJ. Outer-membrane vesicles from gram-negative bacteria: biogenesis and functions. Nature reviews. microbiology. 2015;13(10):605–609. doi: 10.1038/nrmicro3525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Cai W, Kesavan DK, Wan J, Abdelaziz MH, Su Z, Xu H. Bacterial outer membrane vesicles, a potential vaccine candidate in interactions with host cells based. Diagnostic pathology. 2018;13(1):95–104. doi: 10.1186/s13000-018-0768-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Acevedo R, Fernandez S, Zayas C, Acosta A, Sarmiento ME, Ferro VA, Rosenqvist E, Campa C, Cardoso D, Garcia L, Perez JL. Bacterial outer membrane vesicles and vaccine applications. Frontiers in immunology. 2014;5:121–129. doi: 10.3389/fimmu.2014.00121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.van der Pol L, Stork M, van der Ley P. Outer membrane vesicles as platform vaccine technology. Biotechnology Journal. 2015;10(11):1689–1706. doi: 10.1002/biot.201400395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Holst J, Oster P, Arnold R, Tatley M, Næss L, Aaberge I, Galloway Y, McNicholas A, O'hallahan J, Rosenqvist E, Black S. Vaccines against meningococcal serogroup B disease containing outer membrane vesicles (OMV): lessons from past programs and implications for the future. Human vaccines and immunotherapeutics. 2013;9(6):1241–1253. doi: 10.4161/hv.24129. [DOI] [PMC free article] [PubMed] [Google Scholar]



