Abstract
The pandemic of novel coronavirus disease 2019 (COVID-19) is caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Diabetes mellitus is a risk factor for developing severe illness and a leading cause of death in patients with COVID-19. Diabetes can precipitate hyperglycaemic emergencies and cause prolonged hospital admissions. Insulin resistance is thought to cause endothelial dysfunction, alveolar capillary micro-angiopathy and interstitial lung fibrosis through pro-inflammatory pathways. Autopsy studies have also demonstrated the presence of microvascular thrombi in affected sections of lung, which may be associated with diabetes. Chest imaging using x-ray (CXR) and computed tomography (CT) of chest is used to diagnose, assess disease progression and severity in COVID-19. This article reviews current literature regarding chest imaging findings in patients with diabetes affected by COVID-19. A literature search was performed on PubMed. Patients with diabetes infected with SARS-CoV-2 are likely to have more severe infective changes on CXR and CT chest imaging. Severity of airspace consolidation on CXR is associated with higher mortality, particularly in the presence of co-morbidities such as ischaemic heart disease. Poorly controlled diabetes is associated with more severe acute lung injury on CT. However, no association has been identified between poorly-controlled diabetes and the incidence of pulmonary thromboembolism in patients with COVID-19.
Keywords: Diabetes mellitus, COVID-19, Chest X-Ray, Chest imaging using x-ray, Computed tomography of chest
Core Tip: COVID-19 infection can present as multifocal peripheral airspace changes on chest imaging using x-ray (CXR). Ground-glass opacities are the most common computed tomography finding in coronavirus disease 2019 (COVID-19). Post admission daily bloody glucose readings are a strong predictor for COVID-19 CXR changes that indicate poorer outcomes. Poorly controlled diabetes is associated with increased volumes of ground-glass opacity and consolidation. Diabetes is also linked with endothelial dysfunction and hypercoagulability, which may result in the formation of microvascular thrombi in peripheral segments of lung.
INTRODUCTION
The world is currently undergoing a significant healthcare crisis due to the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic. In March 2020, World Health Organisation declared a pandemic caused by SARs-CoV-2. SARS-CoV-2 was named novel coronavirus disease 2019 (COVID-19). Hospitals in different countries have been overwhelmed with patients suffering from COVID-19. So far, 2.78 million people have died as of 29th March 2021[1].
Diabetes mellitus (DM) is a risk factor associated with severe illness in SARS-CoV-2 infection, precipitating hyperglycaemic emergencies such as diabetic ketoacidosis (DKA) and hyperosmolar hyperglycaemic state (HHS)[2]. A third of deaths in England up to May 2020 related to COVID-19 occurred in people with DM[3]. Patients with DM are more likely to stay longer in hospital[4]. DM can cause a deregulated immune system predisposing to infection; the endothelial angiotensin-converting enzyme 2 (ACE2) receptor responsible for SARS-CoV-2 invasion in human cells has reduced expression in patients of DM, possibly due to glycosylation[5]. Insulin resistance and altered glucose homeostasis have been thought to cause alveolar capillary micro-angiopathy and interstitial fibrosis via over-inflammation[6].
A normal chest radiograph does not exclude COVID-19 pneumonia, and no single feature on a radiograph is diagnostic[7]. However, a combination of multifocal peripheral airspace changes often found bilaterally may be present in COVID-19. Due to limited PCR testing capacity in the early d of the pandemic, in addition to its low sensitivity and waiting period of up to 2 d, many clinicians turned to chest computed tomography (CT) for early detection of COVID-19.
Studies have reported the negative predictive value of using CT to be above 90%[8,9]. Chest CT was used to detect subtle radiological changes consistent with COVID-19 in patients where the chest radiograph was reported to be normal or indeterminate. Typical CT findings seen in patients with COVID-19 include peripheral ground-glass opacities (GGO), which progresses to consolidation and interstitial thickening within GGO areas known as ‘crazy paving pattern’[10,11]. These non-specific imaging findings of acute lung injury are indistinguishable from other types of viral pneumonia or interstitial lung diseases, thereby limiting the use of CT as a confirmatory diagnostic test in COVID-19.
This article reviews current literature regarding chest imaging changes in patients with DM affected by COVID-19.
LITERATURE SEARCH
A literature search was conducted on PubMed using the keywords of COVID-19 or Coronavirus; CXR or x-ray or radiograph; CT chest; CTPA or pulmonary embolism or PE; and diabetes mellitus or diabetes within the title or abstract.
Chest Radiography
Studies have shown chest radiographs of patients with DM to have increased bilateral airspace consolidation compared to patients without DM[12,13]. The severity of chest radiograph changes in patients with DM has indicated a significant correlation with mortality, as evidenced in multivariate analysis by Cellina et al[14]. Patients with bilateral peripheral alveolar disease (Figure 1) often present at a later stage and have a worse outcome. However, some patients with COVID-19 have preserved lung compliance despite being acutely hypoxaemic, suggesting poorer outcomes result from processes other than alveolar damage[15].
Figure 1.
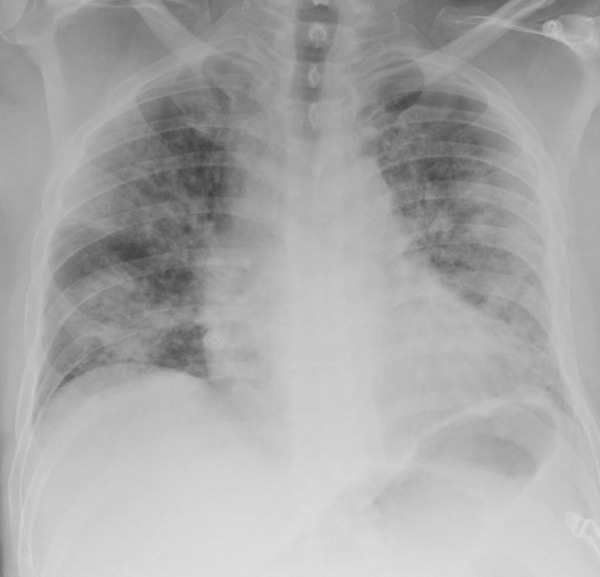
The Chest X-Ray demonstrates multiple bilateral peripheral predominant airspace opacities. There is no pleural effusion.
In some studies, DM alone was not associated with an increased risk of intensive care unit admission or death. Still, it was associated with cardiovascular disease as a driver of poorer outcomes. Izzi-Engbeaya et al[16] studied 889 patients admitted to London hospitals with COVID-19, and their outcomes found patients with DM were found to have a 33% increased risk of death or ICU admission if they also have ischaemic heart disease. Surprisingly, a similar severity of CXR changes was demonstrated for patients with and without DM. Mozzini et al[17] (2021) studied 50 Italian patients with COVID-19, 32% of which had DM. Patients with hypertension or DM had 8 times greater risk of having more severe CXR changes.
COVID-19 infection in patients with DM leads to hyperglycaemia, and in some cases leads to DKA and/or HHS[2]. It has been shown that there is a positive correlation between daily average blood glucose readings and CXR findings. Similarly, post-admission day-1 hyperglycaemia was found to be the strongest independent predictor for COVID-19 CXR changes. This was a stronger predictor than age, body mass index, and temperature[18].
Chest computed tomography
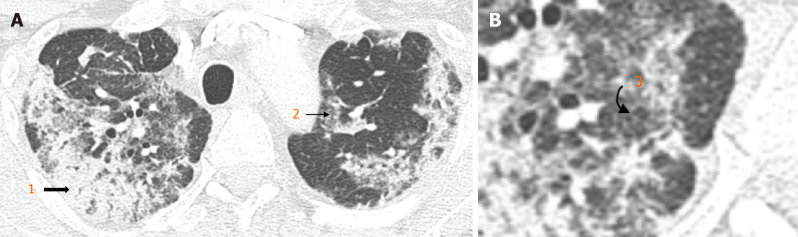
Earlier studies employed semi-quantitative methods to analyse chest computed tomography (CT) findings (Figure 2) in patients with COVID-19[19,20]. This involved a single, or multiple experienced radiologists blinded to clinical parameters and assigning a score based on the severity of findings. Higher chest CT scores have been found in patients with DM, suggesting more severe COVID-19 pneumonia when compared with patients without DM[19]. Findings by Iacobellis et al[18] suggested day-1 hyperglycaemia as a predictor of COVID-19 severity on CXR were confirmed on CT.
Figure 2.
Chest X-Ray. A: Typical appearances of COVID-19 infection: Bilateral peripheral consolidation (1. block arrow), multifocal groundglass opacities (2. straight arrow); B: Some areas of smooth intralobular septal thickening (3. curved arrow).
Patients with poorly-controlled DM are likely to have more severe COVID-19 pneumonia. A recent study by Lu et al[21] using a quantitative artificial intelligence algorithm found parameters including the percentage of ground glass volume (PGV) and percentage of consolidation volume (PCV), positively correlated with fasting blood glucose and HbA1c. Unlike semi-quantitative methods, results using this approach were not affected by inter- and intra-observer variability. Raoufi et al[20] used a semi-quantitative method to study 117 patients with DM in Iran and found no significant difference in patients with well-controlled (defined as maintaining glycaemic variability between 3.9-10 mmol/L) and poorly-controlled DM. However, the poorly-controlled group contained almost 4 times the number of patients (93 vs 24). Furthermore, the median age of patients in the well-controlled group were older (75 vs 62 years) which may have been a confounding factor for this negative result[20].
Studies have shown mortality rates to be higher among patients with poorly-controlled DM and COVID-19 than the general population with COVID-19[22,23]. In particular, high HbA1c levels have been linked with inflammation and hypercoagulability, resulting in an increased mortality rate in patients with DM suffering from COVID-19[24]. However, the accuracy of these results may be influenced by other co-morbidities such as ischaemic heart disease and stroke. No large-scale studies have yet shown an association between worse CT findings and mortality in DM.
A high incidence of venous and arterial thrombotic complications in critically ill patients with COVID-19 has been reported previously[25]. Recent literature based on autopsy studies shows that the origin of thrombotic lesions in COVID-19 is largely unknown. Lung histopathological analysis found multiple thrombi in small to medium pulmonary arteries giving rise to the theory of COVID-19 associated immunothrombosis, contrary to the conventional thromboembolic pathomechanism of PE[26,27]. In situ microvascular thrombosis or immunothrombosis occurs due to alveolar injury, inflammatory storm and disruption of the thromboprotective pulmonary vascular endothelium. COVID-19 clinical outcomes are worse in patients with diseases associated with endothelial dysfunction such as systemic hypertension, DM and obesity[28].
The radiological finding of subsegmental or segmental thrombi in peripheral segments of lung affected by acute lung injury and the absence of deep vein thrombosis (DVT) in patients with COVID-19 infection, assumes the theory of immunothrombosis[27]. Monfardini et al[29] found 76% of patients with a moderate-high pre-test probability of PE and positive D-dimer level (a fibrin degradation product measured to help diagnose thrombosis), had positive CTPA findings. Nevertheless, only 15% of these patients were associated with ultrasound detected lower limb DVT[29], suggesting the remainder probably represented immunothrombosis. A meta-analysis of twenty-seven studies by Suh et al[30] revealed DVT was only found in 42% of patients with PE.
As yet, no large-scale studies have reported a link between pulmonary thromboembolism and DM in patients with COVID-19. Kaminetzky et al[31] found patients with DM were significantly less frequently observed to have CTPA examinations. Of 23 patients identified to have PE in this study, only 3 had DM; however, this finding may be attributed to the small sample size.
CONCLUSION
DM predisposes to immune deregulation and reduced expression of the ACE2 receptor, leading to severe acute lung injury[5,6]. Studies have proven a link between DM and more severe airspace consolidation based on chest x-ray findings[12,13]. Furthermore, CXR evidence suggests DM is associated with higher mortality in COVID-19. The exact pathogenesis of this is unclear but may be related to microvascular immunothrombosis[26,28].
There is now quantitative evidence to suggest poorly controlled DM is associated with more severe lung injury on CT[21]. However, no large-scale studies have investigated a direct link between CT findings and mortality in DM. Although the incidence of PE is greater in critically ill patients with COVID-19[25], no link has been established between poorly controlled DM and the risk of PE.
As new research into COVID-19 is produced and evidence emerges from autopsy studies, the understanding of pathobiology of the disease has evolved. However, there remains scope for future research; particularly whether small pulmonary thromboses represent venous thromboembolism, immunothrombosis, or a combination of both. Furthermore, a direct link between DM and immunothrombosis may help to guide future management strategies.
ACKNOWLEDGEMENT
We will like to thank Kirresh OZ for providing radiographs.
Footnotes
Conflict-of-interest statement: The authors declare that they have no competing or conflicts of interests.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Peer-review started: March 30, 2021
First decision: October 17, 2021
Article in press: December 28, 2021
Specialty type: Radiology, nuclear medicine and medical imaging
Country/Territory of origin: United Kingdom
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Liu D S-Editor: Wang LL L-Editor: A P-Editor: Wang LL
Contributor Information
Sunay Gangadharan, Department of Radiology, University Hospitals Sussex NHS Foundation Trust, Brighton BN2 5BE, United Kingdom.
Storm Parker, Department of Radiology, University Hospitals Sussex NHS Foundation Trust, Brighton BN2 5BE, United Kingdom.
Fahad Wali Ahmed, Department of Medical Oncology, King Faisal Specialist Hospital and Research Centre, Madinah 42522, Saudi Arabia. fahadwali@yahoo.com.
References
- 1.CCSE Dashboard [Internet] COVID-19 Dashboard by the Center for Systems Science and Engineering (CSSE) at Johns Hopkins University (JHU). [cited 20 February 2021]. Available from: https://www.arcgis.com/apps/opsdashboard/index.html#/bda7594740fd40299423467b48e9ecf6 .
- 2.Rafique S, Ahmed FW. A Case of Combined Diabetic Ketoacidosis and Hyperosmolar Hyperglycemic State in a Patient With COVID-19. Cureus . 2020;12:e8965. doi: 10.7759/cureus.8965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Barron E, Bakhai C, Kar P, Weaver A, Bradley D, Ismail H, Knighton P, Holman N, Khunti K, Sattar N, Wareham NJ, Young B, Valabhji J. Associations of type 1 and type 2 diabetes with COVID-19-related mortality in England: a whole-population study. Lancet Diabetes Endocrinol . 2020;8:813–822. doi: 10.1016/S2213-8587(20)30272-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ahmed FW, Kirresh OZ, Robinson AV. A Retrospective Study Assessing the Effect of Diabetes on Mortality in Patients With COVID-19 at a Teaching Hospital in the United Kingdom [Internet]. 2021. [cited 20 February 2021]. Available from: https://www.cureus.com/articles/54241 . [DOI] [PMC free article] [PubMed]
- 5.Sartore G, Ragazzi E, Faccin L, Lapolla A. A role of glycation and methylation for SARS-CoV-2 infection in diabetes? Med Hypotheses . 2020;144:110247. doi: 10.1016/j.mehy.2020.110247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sardu C, Gargiulo G, Esposito G, Paolisso G, Marfella R. Impact of diabetes mellitus on clinical outcomes in patients affected by Covid-19. Cardiovasc Diabetol . 2020;19:76. doi: 10.1186/s12933-020-01047-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cleverley J, Piper J, Jones MM. The role of chest radiography in confirming covid-19 pneumonia. BMJ . 2020;370:426. doi: 10.1136/bmj.m2426. [DOI] [PubMed] [Google Scholar]
- 8.Ai T, Yang Z, Hou H, Zhan C, Chen C, Lv W, Tao Q, Sun Z, Xia L. Correlation of Chest CT and RT-PCR Testing for Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology . 2020;296:E32–E40. doi: 10.1148/radiol.2020200642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Herpe G, Lederlin M, Naudin M, Ohana M, Chaumoitre K, Gregory J, Vilgrain V, Freitag CA, De Margerie-Mellon C, Flory V, Ludwig M, Mondot L, Fitton I, Jacquier ARR, Ardilouze P, Petit I, Gervaise A, Bayle O, Crombe A, Mekuko Sokeng M, Thomas C, Henry G, Bliah V, Le Tat T, Guillot MS, Gendrin P, Garetier M, Bertolle E, Montagne C, Langlet B, Kalaaji A, Kayayan H, Desmots F, Dhaene B, Saulnier PJ, Guillevin R, Bartoli JM, Beregi JP, Tasu JP. Efficacy of Chest CT for COVID-19 Pneumonia Diagnosis in France. Radiology . 2021;298:E81–E87. doi: 10.1148/radiol.2020202568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ufuk F, Savaş R. Chest CT features of the novel coronavirus disease (COVID-19) Turk J Med Sci . 2020;50:664–678. doi: 10.3906/sag-2004-331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ye Z, Zhang Y, Wang Y, Huang Z, Song B. Chest CT manifestations of new coronavirus disease 2019 (COVID-19): a pictorial review. Eur Radiol . 2020;30:4381–4389. doi: 10.1007/s00330-020-06801-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Elemam NM, Hannawi H, Salmi IA, Naeem KB, Alokaily F, Hannawi S. Diabetes mellitus as a comorbidity in COVID-19 infection in the United Arab Emirates. Saudi Med J . 2021;42:170–180. doi: 10.15537/smj.2021.2.25700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bhandari S, Rankawat G, Singh A, Gupta V, Kakkar S. Impact of glycemic control in diabetes mellitus on management of COVID-19 infection. Int J Diabetes Dev Ctries . 2020:1–6. doi: 10.1007/s13410-020-00868-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cellina M, Gibelli D, Valenti Pittino C, Toluian T, Marino P, Oliva G. Risk Factors of Fatal Outcome in Patients With COVID-19 Pneumonia. Disaster Med Public Health Prep . 2020:1–8. doi: 10.1017/dmp.2020.346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gattinoni L, Coppola S, Cressoni M, Busana M, Rossi S, Chiumello D. COVID-19 Does Not Lead to a "Typical" Acute Respiratory Distress Syndrome. Am J Respir Crit Care Med . 2020;201:1299–1300. doi: 10.1164/rccm.202003-0817LE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Izzi-Engbeaya C, Distaso W, Amin A, Yang W, Idowu O, Kenkre JS, Shah RJ, Woin E, Shi C, Alavi N, Bedri H, Brady N, Blackburn S, Leczycka M, Patel S, Sokol E, Toke-Bjolgerud E, Qayum A, Abdel-Malek M, Hope DCD, Oliver NS, Bravis V, Misra S, Tan TM, Hill NE, Salem V. Adverse outcomes in COVID-19 and diabetes: a retrospective cohort study from three London teaching hospitals. BMJ Open Diabetes Res Care . 2021;9 doi: 10.1136/bmjdrc-2020-001858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mozzini C, Cicco S, Setti A, Racanelli V, Vacca A, Calciano L, Pesce G, Girelli D. Spotlight on Cardiovascular Scoring Systems in Covid-19: Severity Correlations in Real-world Setting. Curr Probl Cardiol . 2021;46:100819. doi: 10.1016/j.cpcardiol.2021.100819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Iacobellis G, Penaherrera CA, Bermudez LE, Bernal Mizrachi E. Admission hyperglycemia and radiological findings of SARS-CoV2 in patients with and without diabetes. Diabetes Res Clin Pract . 2020;164:108185. doi: 10.1016/j.diabres.2020.108185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guo W, Li M, Dong Y, Zhou H, Zhang Z, Tian C, Qin R, Wang H, Shen Y, Du K, Zhao L, Fan H, Luo S, Hu D. Diabetes is a risk factor for the progression and prognosis of COVID-19. Diabetes Metab Res Rev . 2020:e3319. doi: 10.1002/dmrr.3319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Raoufi M, Khalili S, Mansouri M, Mahdavi A, Khalili N. Well-controlled vs poorly-controlled diabetes in patients with COVID-19: Are there any differences in outcomes and imaging findings? Diabetes Res Clin Pract . 2020;166:108286. doi: 10.1016/j.diabres.2020.108286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lu X, Cui Z, Pan F, Li L, Liang B, Yang L, Zheng C. Glycemic status affects the severity of coronavirus disease 2019 in patients with diabetes mellitus: an observational study of CT radiological manifestations using an artificial intelligence algorithm. Acta Diabetol . 2021;58:575–586. doi: 10.1007/s00592-020-01654-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wu ZH, Tang Y, Cheng Q. Diabetes increases the mortality of patients with COVID-19: a meta-analysis. Acta Diabetol . 2021;58:139–144. doi: 10.1007/s00592-020-01546-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhu L, She ZG, Cheng X, Qin JJ, Zhang XJ, Cai J, Lei F, Wang H, Xie J, Wang W, Li H, Zhang P, Song X, Chen X, Xiang M, Zhang C, Bai L, Xiang D, Chen MM, Liu Y, Yan Y, Liu M, Mao W, Zou J, Liu L, Chen G, Luo P, Xiao B, Zhang Z, Lu Z, Wang J, Lu H, Xia X, Wang D, Liao X, Peng G, Ye P, Yang J, Yuan Y, Huang X, Guo J, Zhang BH. Association of Blood Glucose Control and Outcomes in Patients with COVID-19 and Pre-existing Type 2 Diabetes. Cell Metab . 2020;31:1068–1077.e3. doi: 10.1016/j.cmet.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang Z, Du Z, Zhu F. Glycosylated hemoglobin is associated with systemic inflammation, hypercoagulability, and prognosis of COVID-19 patients. Diabetes Res Clin Pract . 2020;164:108214. doi: 10.1016/j.diabres.2020.108214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Klok FA, Kruip MJHA, van der Meer NJM, Arbous MS, Gommers D, Kant KM, Kaptein FHJ, van Paassen J, Stals MAM, Huisman MV, Endeman H. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: An updated analysis. Thromb Res . 2020;191:148–150. doi: 10.1016/j.thromres.2020.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Patel BV, Arachchillage DJ, Ridge CA, Bianchi P, Doyle JF, Garfield B, Ledot S, Morgan C, Passariello M, Price S, Singh S, Thakuria L, Trenfield S, Trimlett R, Weaver C, Wort SJ, Xu T, Padley SPG, Devaraj A, Desai SR. Pulmonary Angiopathy in Severe COVID-19: Physiologic, Imaging, and Hematologic Observations. Am J Respir Crit Care Med . 2020;202:690–699. doi: 10.1164/rccm.202004-1412OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.van Dam LF, Kroft LJM, van der Wal LI, Cannegieter SC, Eikenboom J, de Jonge E, Huisman MV, Klok FA. Clinical and computed tomography characteristics of COVID-19 associated acute pulmonary embolism: A different phenotype of thrombotic disease? Thromb Res . 2020;193:86–89. doi: 10.1016/j.thromres.2020.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Loo J, Spittle DA, Newnham M. COVID-19, immunothrombosis and venous thromboembolism: biological mechanisms. Thorax . 2021;76:412–420. doi: 10.1136/thoraxjnl-2020-216243. [DOI] [PubMed] [Google Scholar]
- 29.Monfardini L, Morassi M, Botti P, Stellini R, Bettari L, Pezzotti S, Alì M, Monaco CG, Magni V, Cozzi A, Schiaffino S, Bnà C. Pulmonary thromboembolism in hospitalised COVID-19 patients at moderate to high risk by Wells score: a report from Lombardy, Italy. Br J Radiol . 2020;93:20200407. doi: 10.1259/bjr.20200407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Suh YJ, Hong H, Ohana M, Bompard F, Revel MP, Valle C, Gervaise A, Poissy J, Susen S, Hékimian G, Artifoni M, Periard D, Contou D, Delaloye J, Sanchez B, Fang C, Garzillo G, Robbie H, Yoon SH. Pulmonary Embolism and Deep Vein Thrombosis in COVID-19: A Systematic Review and Meta-Analysis. Radiology . 2021;298:E70–E80. doi: 10.1148/radiol.2020203557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kaminetzky M, Moore W, Fansiwala K, Babb JS, Kaminetzky D, Horwitz LI, McGuinness G, Knoll A, Ko JP. Pulmonary Embolism at CT Pulmonary Angiography in Patients with COVID-19. Radiol Cardiothorac Imaging . 2020;2:e200308. doi: 10.1148/ryct.2020200308. [DOI] [PMC free article] [PubMed] [Google Scholar]