Abstract
Overall, 12% of the global population (800 million) suffers from liver disease, which causes 2 million deaths every year. Liver injury involving characteristic reactive oxygen/nitrogen species (RONS) and inflammation plays a key role in progression of liver disease. As a key metabolic organ of the human body, the liver is susceptible to injury from various sources, including COVID-19 infection. Owing to unique structural features and functions of the liver, most current antioxidants and anti-inflammatory drugs are limited against liver injury. However, the characteristics of the liver could be utilized in the development of nanodrugs to achieve specific enrichment in the liver and consequently targeted treatment. Nanodrugs have shown significant potential in eliminating RONS and regulating inflammation, presenting an attractive therapeutic tool for liver disease through controlling liver injury. Therefore, the main aim of the current review is to provide a comprehensive summary of the latest developments contributing to our understanding of the mechanisms underlying nanodrugs in the treatment of liver injury via harnessing RONS and inflammation. Meanwhile, the prospects of nanodrugs for liver injury therapy are systematically discussed, which provides a sound platform for novel therapeutic insights and inspiration for design of nanodrugs to treat liver disease.
Keywords: RONS-Scavenging, Anti-inflammatory, Liver injury, Nanozyme, Nanocarriers
Graphical abstract
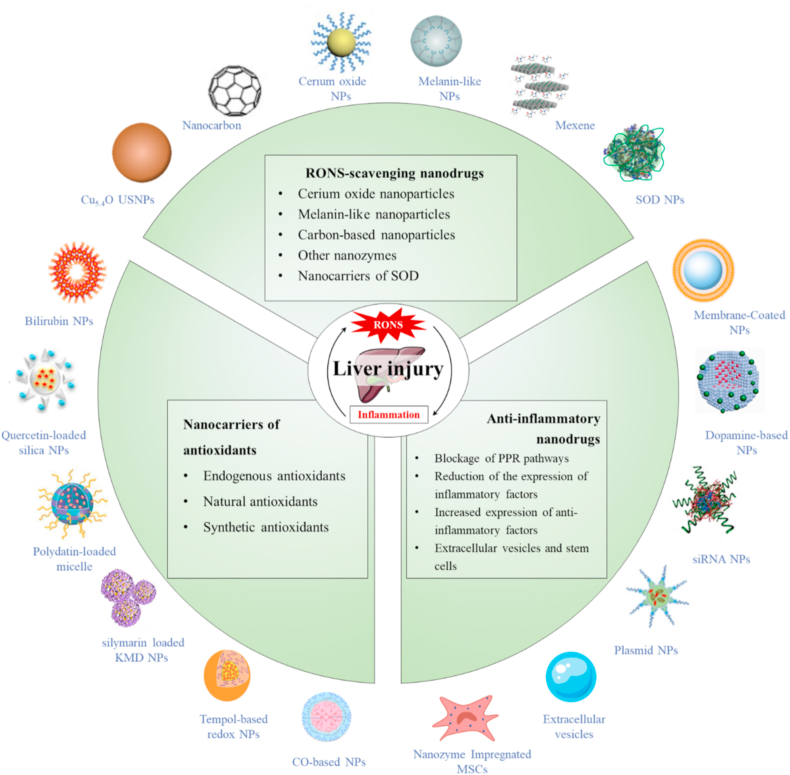
The scope and focus of this article. RONS overproduction and inflammation are the major features of multiple types of liver injury, with known crosstalk between RONS and inflammation. Nanoparticles with RONS-scavenging or inflammation-regulating function could ameliorate liver injury through eliminating RONS and inhibiting inflammation. These therapeutic nanodrugs used for treatment of liver disease are divided into three categories according to structure and mechanism of action.
Highlights
-
•
RONS and inflammation play an essential role in the liver injury progression.
-
•
The development of nanodrugs harnessing RONS and inflammation is expected to achieve superior efficacy for liver injury.
-
•
Nanodrugs that scavenging RONS and inhibiting inflammation are systematically reviewed.
1. Introduction
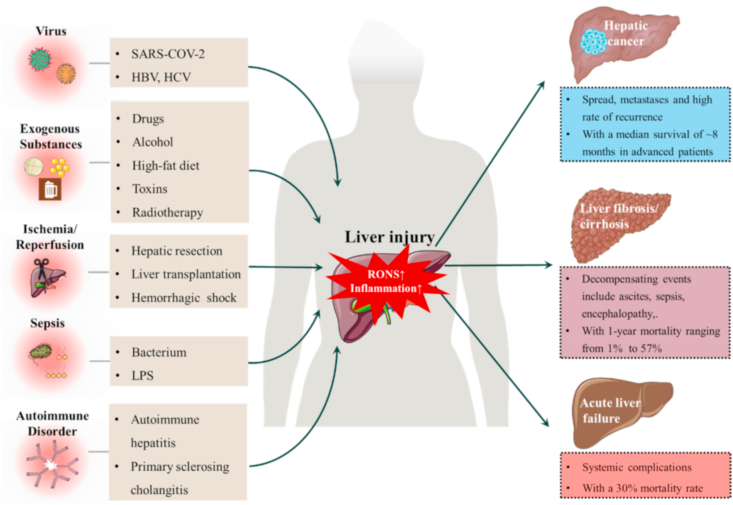
Liver injury is one of the main ongoing hot topics of medical research. Recently, the high incidence of liver injury triggered by coronavirus disease 2019 (COVID-19) infection has attracted the attention of several research groups [1,2]. A large proportion of COVID-19 patients (over 45%) experience liver injury of varying severity, with the mortality rate of severe COVID-19-induced liver injury reported to be as high as 42% [3]. As an essential organ of the human body, the liver plays an irreplaceable role in numerous physiological processes [[4], [5], [6], [7]]. Consequently, the liver is significantly vulnerable to multiple extraneous factors and external stimuli [[8], [9], [10]]. Liver injury is the main factor in pathogenesis of most liver disorders, including hepatitis, acute liver failure, liver fibrosis and liver cancer. Liver disease has a high morbidity rate, with more than 800 million cases reported worldwide (up to 12% of the global population) resulting in about 2 million deaths each year [11,12]. Depending on etiology, liver injury can be classified into injuries caused by exogenous substances (including drug-induced liver injury, alcoholic liver injury, and non-alcoholic fatty liver disease), disease or external stimuli (including COVID-19, viral hepatitis, hepatic ischemia-reperfusion injury (IRI), sepsis, and radiation), and others [[12], [13], [14], [15], [16]]. However, available treatments are limited, mainly involving methods targeting etiology (including diet control, abstinence from alcoholic drink, drug withdrawal, and antiviral and antilipemic therapy) [[17], [18], [19]]. Liver injury can rapidly progress to acute liver failure, fibrosis, or even cancer if not treat effectively [[20], [21], [22]]. In cases where liver injury develops into acute liver failure and cirrhosis, transplantation is the only feasible option. However, only a small percentage of patients benefit from liver transplantation due to the very limited source of donors [23,24]. A diagram depicting detailed pathogenesis of various forms of liver injury and clinical progression is presented in Fig. 1.
Fig. 1.
Pathogenesis of various forms of liver injury and clinical characteristics of severe outcomes. As a core organ with important metabolic and detoxification functions, the liver is vulnerable to RONS/inflammation-related injury owing to significant exposure to harmful substances and stimuli. As illustrated above, viruses, drugs, hemorrhagic shock, and other factors trigger varying degrees of liver injury that can further develop into hepatic cancer, liver cirrhosis and acute liver injury (shown on the right).
Emerging evidence supports a key role of oxidative stress and inflammation in all liver injury types, among which reactive oxygen/nitrogen species (RONS) are crucial [[25], [26], [27]] As an important metabolic organ of the human body, the liver requires high levels of energy with comparable energy consumption per unit mass to that of the brain [28]. Therefore, hepatocytes contain a high abundance of mitochondria. When liver injury occurs, excessive RONS are produced from damaged mitochondria. Hepatotoxic compounds, their active metabolites and other factors (such as hypoxia and reoxygenation) interfere with the electron transport chain located in the mitochondrial membrane, leading to excessive RONS production. In addition, inflammation is initiated by liver injury, followed by derivation of RONS mainly from NADPH oxidase 2 (NOX2), inducible nitric oxide synthase (iNOS), and myeloperoxidase (MPO) in activated inflammatory cells and liver sinusoidal endothelial cells (LSECs), further dysregulating the redox balance of liver [[29], [30], [31]]. More importantly, persistent inflammation with elevated inflammatory cytokines will lead to extensive hepatic damage and the progression of chronic liver disease, even liver fibrosis [27,32].
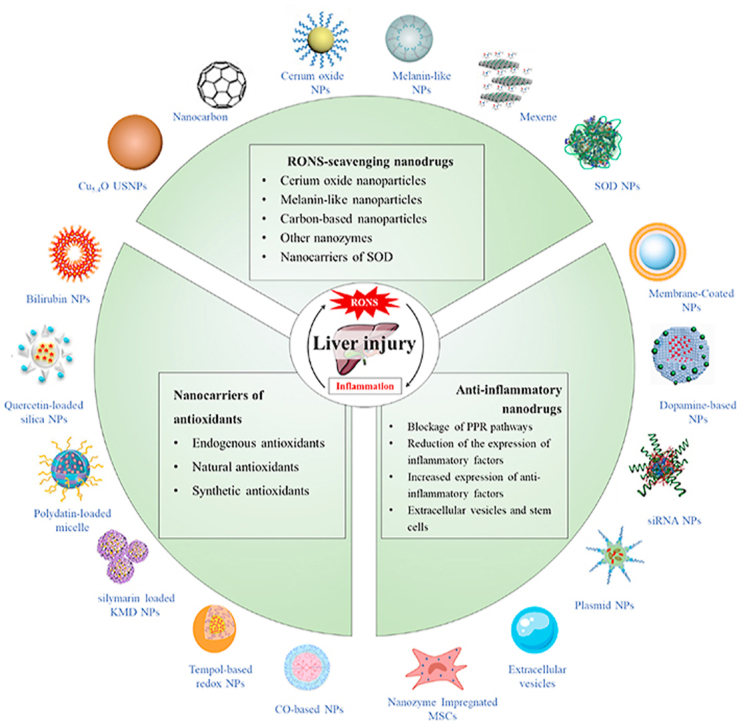
Currently available antioxidant and anti-inflammatory drugs have limited efficacy in treatment of liver injury [33,34]. Owing to the unique structure and metabolic functions of liver, the therapeutic effects of almost all antioxidant and anti-inflammatory drugs are limited, occasionally even aggravating liver injury. For example, only 34% of patients who received pioglitazone and 43% of those who received vitamin E partially benefit in a clinical trial of nonalcoholic steatohepatitis [35]. In recent years, the latest advances of nanodrugs made in clinical shed a light on the treatment of liver injury. Nanodrugs have recently achieved excellent results as therapeutic agents for various diseases [[36], [37], [38], [39], [40], [41], [42]]. There were several reviews focusing on a kind of nanodrug, a type of liver injury or influence of nanodrugs on liver after retrieving and evaluating relevant articles [[43], [44], [45], [46], [47]], instead of the RONS and inflammation regulatory properties of nanodrugs for all kinds of liver injury. Therefore, the mechanisms associated with RONS and inflammation-mediated liver injury and related documenting collective findings have been summarized in the current review, which would be very valuable in providing a comprehensive outline and platform for further investigation of therapeutic nanodrugs targeting RONS and inflammation. The therapeutic effects of newly developed nanodrugs on RONS and inflammation are further described specifically. The available nanodrugs are classified into three categories: RONS-scavenging nanodrugs, nanocarriers with antioxidants, and anti-inflammatory nanodrugs (Fig. 2 & Table 1). Finally, the current challenges of nanodrug application in the field of liver injury treatment are discussed.
Fig. 2.
The scope and focus of this article. RONS overproduction and inflammation are the major features of multiple types of liver injury, with known crosstalk between RONS and inflammation. Nanoparticles with RONS-scavenging or inflammation-regulating function could ameliorate liver injury through eliminating RONS and inhibiting inflammation. These therapeutic nanodrugs used for treatment of liver injury are divided into three categories according to structure and mechanism of action.
Table 1.
Main nanodrugs with RONS-scavenging function for liver injury.
| Category | Nanoparticles | Size | Main Components | Experimental Model | Effect Mechanism | Reference |
|---|---|---|---|---|---|---|
| RONS-scavenging nanodrugs | ||||||
| Cerium oxide nanoparticles | CeO2NPs | 4–20 nm | CeO2 | CCl4-induced liver fibrosis | RONS↓ TNF-α, IL1β, COX-2, iNOS↓ GPx, CAT↑, Ncf1, Ncf2↓ Atf3, Hspa5↓ |
[153] |
| Ceria NPs | <20 nm | hydrophobic ceria, PEG | Hepatic IRI | RONS↓ IL-1, IL-12, TNF-α, IL-6, iNOS, MPO↓ |
[175] | |
| CeNZs | ∼12 nm | ceria ions, DSPE-PEG2000 | APAP-induced liver injury | ROS↓ GPx, HO-1↑, NOX2↓ Keap↓, Nrf2↑ IL-1β, TNF-α, HIF-1α↓ |
[176] | |
| Melanin-like nanoparticles | PADN | 106.6 ± 5.4 nm | PEGylated phenylboronic-acid-protected l-DOPA | APAP-induced liver injury | RONS↓ Lipid peroxidation↓ GPx↑ MPO, TNF-α, IL-6, IL-1β↓ |
[182] |
| Carbon-based nanoparticles | C3 NPs | 175 nm | carboxyfullerene | Hepatic IRI | Lipid peroxidation↓, SOD↑ MPO, TNF-α, IL-6↓, NF-κB↓ |
[186] |
| Large GQDs | 20–60 nm | graphene | ConA induced hepatitis | RONS↓ Lipid peroxidation↓ IFN-γ, T-beta↓, ST2↑ p-ERK, p-JNK↓, p38 MAPK↑ |
[187] | |
| C-NP | 78 ± 11.3 nm | a hydrophilic carbohydrate-derived nanoparticle | Hepatic IRI | ROS↓, SOD↑ IL-1, TNF-α, IL-6↓ |
[190] | |
| Other nanozymes | Nb2C-PVP NSs | ∼150 nm 0.5–1 nm (thickness) |
ultrathin 2D Nb2C (MXene), PVP | Hepatic IRI | ROS↓, SOD↑ | [200] |
| SeNPs-C/C | ∼50 nm | SeNPs, polysaccharide chitosan | D-GalN-induced liver injury | ROS↓ | [206] | |
| Cu5·4O USNPs | ∼4.5 nm | Cu5·4O | APAP-induced liver injury | ROS↓ | [200] | |
| MoS2 nanosheet | ∼500 nm 10–20 nm (thickness) |
MoS2 | CCl4-induced liver fibrosis | ROS↓ IL1β↓ |
[214] | |
| PBZs | ∼119 nm | PVP, PB nanozymes | Anthracycline-induced liver injury | RONS↓ SOD, GPx↑ Nrf2, Nqo1, HO-1↑ IL1β, IL6, TNF-α, MPO↓ |
[215] | |
| Nanocarriers of SOD | d-HA/SOD/USCaP NPs | 194.6 ± 3.2 nm | SOD molecules, d-HA, USCaP | APAP-induced liver injury | ROS↓, SOD↑ | [219] |
| Nanocarriers of antioxidants | ||||||
| Nanocarriers of antioxidants | BRNP | 90 ± 13 nm | bilirubin, PEG | Hepatic IRI | RONS↓ IL1β, IL6, iNOS, Ptgs2↓ Sele, Ccl2, Icam↓ MPO, NF-κB↓ |
[261] |
| Q-ORMOSIL | ∼103.5 nm | silica nanoparticles, lactobionic acid, quercetin |
Cyclophosphamide nduced liver injury | ROS↓ GSH↑ |
[235] | |
| PD-MC | 84 ± 5 nm | PBEM-co-DPA, polydatin | CCl4-induced liver fibrosis | ROS↓ NOX4↓ TLR/NF-κB↓ |
[232] | |
| Silymarin-loaded KMD NPs | ∼440 nm | ketalized maltodextrin, silymarin | APAP-induced liver injury | ROS↓ TNF-α↓ |
[258] | |
| Anti-inflammatory nanodrugs | ||||||
| Blocking the PPR pathways | M-NPs | 171.4 ± 17.3 nm | Macrophage membrane, PLGA | Hepatic IRI | MyD88, IRAK1, p-p65, TNF-α, IL-6↓ | [268] |
| SW@DSeSeD | 50–200 nm | SW033291, diselenide-containing molecule | APAP-induced liver injury | ROS↓ TNF-α, IL-1β ↓ |
[270] | |
| Reducing the expression of inflammatory factors | PPABLG HNPs | ∼100 nm | TNF-α siRNA, PPABLG (α helix), PAOBLG-MPA | LPS/d-GalN-induced hepatic sepsis | TNF-α, IL-1β, IL-6↓ MPO↓ |
[278] |
| PNSDS | ∼146 nm | TNF-α siRNA, AAPEG | LPS-induced liver injury | TNF-α↓ | [279] | |
| Fuc-liposomes/NF-κB decoy | 64.5 ± 1.84 nm | NF-κB decoy, Fuc-C4-Chol, DOPE | LPS-induced liver injury | TNF-α, NF-κB↓ | [285] | |
| HMGB1-siRNA@SNALP-pPB | ∼110 nm | HMGB1 siRNA, stable nucleic acid lipid nanoparticles, pPB peptide | CCl4/TAA-induced liver fibrosis | HMGB1↓ TNF-α↓ |
[280] | |
| Increasing the expression of anti-inflammatory factors | LiposIA | <200 nm | IL-22/Apop A-I fusion-protein expression plasmid, cationic liposome | APAP-induced liver injury | RONS↓ iNOS, p-JNK↓ STAT3/Erk, Akt/mTOR↑ TNF-α↓ |
[287] |
| CDPIA | ∼100 nm | pIA, biguanide-modified Chitosan, DSPE-PEG2000, penetratin | NAFLD | STAT3/ERK, cyclinD1, Bcl2↑ Nrf2/SOD1, Acox1, Cpt1a↑ |
[288] | |
| pMMP9-DGNS | 107.2 ± 1.5 nm | MMP9 expression plasmid, graphene nanostars, PAMAM-G5 dendrimer | CCl4-induced liver fibrosis | MMP9↑ ARG1, MRC1, RETN1A↑ COX-2, IL-1β↓ |
[290] | |
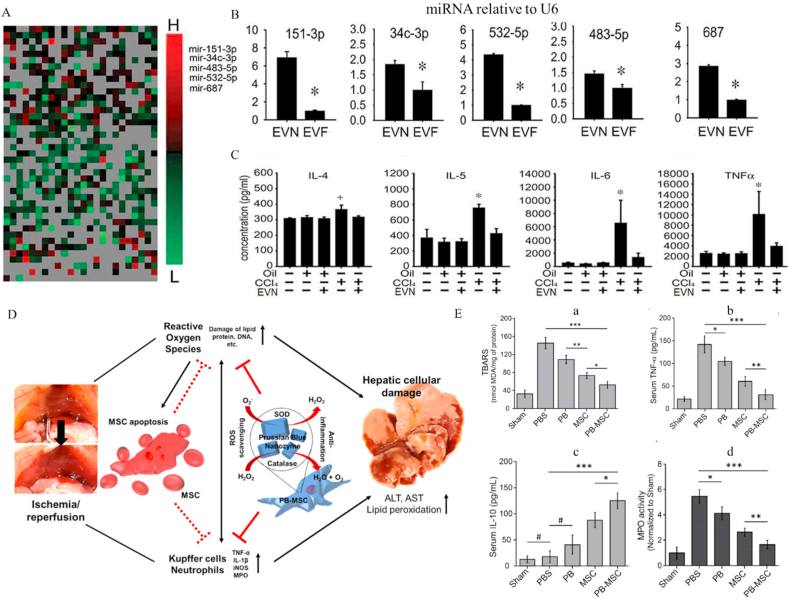
| Extracellular vesicles and stem cells with anti-inflammatory properties | Serum EV | 115±8 nm | microRNA-34c, -151–3p, -483–5p, -532–5p and −68 | CCl4/TAA-induced liver fibrosis | CCN2, α-SMA, Colα1↓ TNF-α, IL-1β ↓ |
[299] |
| PB-MSC | / | MSC, PB | Hepatic IRI | RONS, Lipid peroxidation↓ TNF-α, IL-1β, iNOS↓ IL-10↑ |
[306] | |
2. RONS in liver injury
Due to their strong oxidative properties, RONS act as important inflammation mediators and cause damage to cells at high concentrations [30,32]. The representative RONS predominantly include reactive oxygen species (ROS), such as superoxide anion radicals (O2•−), hydroxyl radical (•OH), hydrogen peroxide (H2O2), and, hypochlorous acid (HOCl), and reactive nitrogen species (RNS), such as peroxynitrite (ONOO−) and nitric oxide (NO) [48,49]. For treatment of liver injury, analysis of the mechanisms underlying RONS generation is critical. Among the various factors causing liver injury, RONS are mainly derived from mitochondria and different types of inflammatory cells.
2.1. Mitochondria-derived RONS generation
Liver is one of the organs with the most abundant mitochondria and a high metabolic rate [26,50]. Each hepatocyte contains an estimated 1000–2000 mitochondria, accounting for 18% of the total volume [[50], [51], [52]]. In all eukaryotic cells, mitochondria are the main energy-supplying organelles, producing ATP mainly through oxidative phosphorylation (OXPHOS) in the mitochondrial respiratory chain, also known as mitochondrial electron transport chain (ETC) [53,54]. Under normal physiological conditions, RONS are produced as a by-product of OXPHOS, requiring the consumption of up to 2% oxygen (O2) in mitochondria [55]. Following disruption of ETC and hindrance of ATP synthesis, high levels of RONS are formed by electrons and O2. Therefore, mitochondria may serve as the main source of RONS. Most liver diseases, including alcoholic liver disease, nonalcoholic fatty liver disease (NAFLD), nonalcoholic steatohepatitis (NASH), drug-induced hepatotoxicity, and viral hepatitis, are characterized by mitochondrial dysfunction [16,26,50,56].
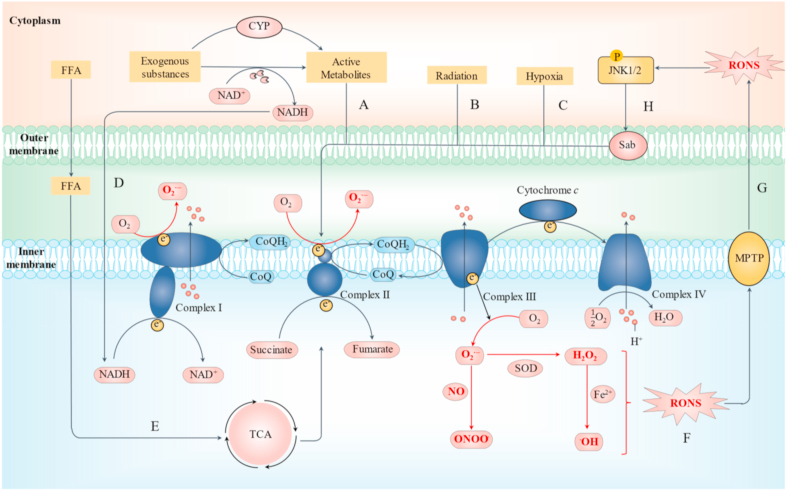
O2•− are produced uncontrollably via ETC in liver injury due to the influence of foreign substances and irritants (Fig. 3A–C) [[57], [58], [59]]. Interference with the electron transport chain in mitochondria is considered the main reason for the initial excessive production of O2•− mediated by exogenous compounds and active metabolites (such as acetaldehyde, NADPQI, ω-hydroxylated fatty acids) [30,[60], [61], [62], [63], [64]]. NADH produced in the P450 enzyme metabolic process is also transported into mitochondria to promote electron leakage (Fig. 3D) [32,65]. In addition, accumulation of excessive free fatty acids in hepatocytes can interfere with ETC by affecting the tricarboxylic acid (TCA) cycle (Fig. 3E) [66,67]. O2•− further generates other RONS with higher reactivity (Fig. 3F) [26,30,68]. Excessive RONS cause damage to the mitochondrial membrane, mitochondrial DNA (mDNA), and ETC (to release free cytochrome c) in mitochondria. Among these, mitochondrial membrane damage leads to alterations in permeability, and ultimately to the release of RONS, mDNA, and cytochrome c in mitochondria into the cytoplasm [26,69]. RONS entering the cytoplasm activate JNK1/2 (Fig. 3G). Phosphorylated JNK is subsequently transported to mitochondria and combined with SH3 homology-associated BTK-binding protein (Sab) on the outer mitochondrial membrane, promoting inactivation of Src under the DOK4 platform in the inner membrane (Fig. 3H), disruption of ETC, and O2•− leakage. This cycle of damage leads to continuous activation of JNK and amplification of the oxidative stress effect [70,71].
Fig. 3.
Mitochondria-derived RONS generation in damaged hepatocytes. Each hepatocyte, the main cell type involved in the metabolic functions of the liver, contains thousands of mitochondria. Excessive O2 in mitochondria is consumed to produce ATP via OXPHOS in ETC while a small amount of O2•− is a by-product of the OXPHOS process during normal physiological conditions. (A–C) Active metabolites and other harmful stimuli directly interfere with ETC, leading to overproduction of O2•−. (D) NADH produced during the P450 enzyme metabolic process is also transported into mitochondria to promote electron leakage. (E) Excessive free fatty acid disrupts the OXPHOS process through promoting the TCA cycle. (F) O2•− generates H2O2 under the action of mitochondrial SOD, which is further converted to •OH via Fenton reaction. Meanwhile O2•− may react with NO from iNOS to form ONOO−. (G) Due to RONS generation, changes in membrane permeability (MPT) based on membrane permeability pore (MPTP) and damage to mitochondrial DNA result in release of RONS into the cytoplasm, in turn, activating JNK1/2. (H) Phosphorylated JNK is transported to the mitochondria and disrupts ETC via Sab activity, promoting RONS generation. This cycle of damage leads to continuous activation of JNK and constant amplification of the oxidative stress effect.
Hemorrhagic shock, liver transplantation, liver resection, and external stimuli often lead to hepatic ischemia reperfusion injury (IRI) [72,73]. Interference with the mitochondrial electron transport chain is the main contributory factor to excessive RONS production in hepatocytes. Under ischemic and hypoxic conditions, accumulated electrons are stored in succinic acid, causing damage to mitochondria. Moreover, reverse electron transport (RET; converse to the conventional forward transport that drives ATP synthesis) occurs due to the recovery of oxygen during reperfusion, inducing abundant O2•− generation from accumulated succinic acid [74,75].
2.2. RONS from inflammation
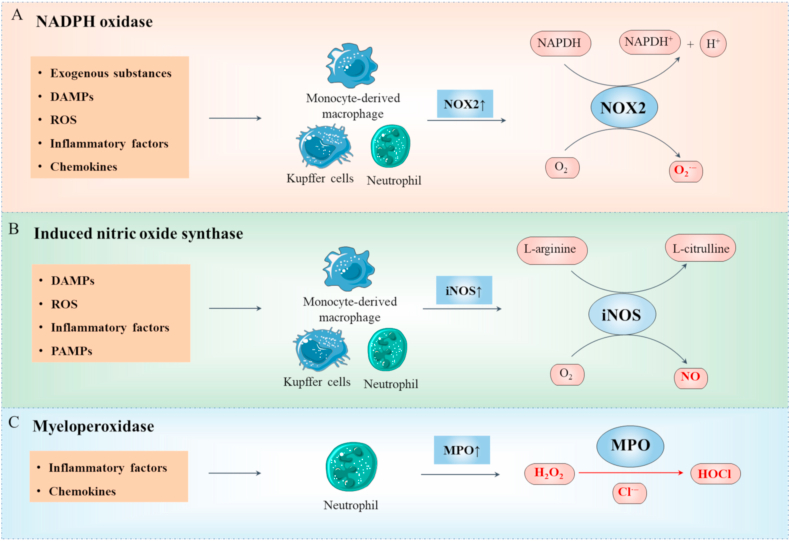
Liver serves as a frontline immune organ that can rapidly generate a robust inflammatory response and efficient adaptive immunity [4]. Continuous inflammation is strongly associated with liver injury and disease progression [76,77]. Exacerbation of injury facilitates RONS-induced cell necrosis, leading to release of RONS and damage-associated molecular patterns (DAMPs) [78]. DAMPs and pathogen-associated molecular patterns (PAMPs) produce several inflammatory factors through the pattern recognition receptor (PRR) pathway of inflammatory cells, mainly including Toll-like receptors (TLR) and Nod-like receptors (NLR) [32,[77], [78], [79]]. As a result, inflammation triggers overproduction of RONS [80,81]. Notably, O2 consumption of neutrophils is increased to 100 times basal metabolic activity to generate RONS [80]. RONS are regulated and overproduced mainly by NADPH oxidase (NOX), nitric oxide synthase (NOS), and myeloperoxidase (MPO) in various inflammatory cells (Fig. 4).
Fig. 4.
Inflammation-derived RONS generation. NOX2 (A), iNOS (B), and MPO (C) are the three main molecules that mediate the generation and regulation of RONS in various inflammatory cells. After receiving multiple factors including RONS, DAMPs and chemokines, NOX2 and iNOS in monocyte-derived macrophages, KCs, and neutrophils are activated while MPO is restricted to neutrophils. NOX2 transfers electrons from NADPH to O2 to form O2•−., iNOS converts l-arginine and O2 into l-citrulline and NO, and MPO catalyzes interactions of chloride ions with H2O2 to generate HOCl.
NOX is a membrane-bound enzyme complex facing the extracellular space, which mainly includes NOX1, NOX2, NOX3, NOX4, NOX5, DUOX1, and DUOX2. NOX2 is responsible for O2•− production in inflammatory cells including macrophages (hepatic resident KC, monocyte-derived macrophages) and neutrophils [[82], [83], [84]]. Several inflammatory factor receptors couple with NOX2 to generate O2•− in inflammatory cells (Fig. 4A) [85]. NOX2 is a multiprotein complex containing a catalytic subunit gp91phox (NOX2) and small subunit p22phox located on the cell membrane and p40phox, p47phox, and p67phox located in the cytoplasm. Protein kinase C (PKC) translocates to the cell membrane and phosphorylates P47. Simultaneously, C3 botulinum toxin substrate 1 (Rac1) facilitates exchange of GTP and GDP, leading to translocation of subunits from the cytoplasmic to transmembrane domain of NOX2. Subsequently, active enzyme complexes transfer electrons from NADPH to O2 via flavin adenine dinucleotide (FAD) to form O2•− [86]. In addition to NOX2 [84,86,87], NOX1, and NOX4 expressed in HSC and hepatocytes are related to progression of liver injury [87,88].
NOS is classified into three types: endothelial NOS (eNOS; NOS3), inducible NOS (iNOS; NOS2), and neuronal NOS (nNOS; NOS1). iNOS plays an important role in liver injury and is prevalent in phagocytes (KC and monocyte-derived macrophages) [89]. iNOS converts l-arginine and O2 into l-citrulline and NO through complex redox reactions via stimulation of inflammatory ROS, DAMPs, and PAMPs (Fig. 4B) [81,90,91]. NO further react with O2 and other reactive intermediates to form RNOS, including •NO2, ONOO−, HNO2, and NO2+ [90].
Myeloperoxidase (MPO), a cationic heme-containing enzyme stored in azurophilic granules of neutrophils, is released into the extracellular space during degranulation. This lysosomal protein catalyzes the formation of HOCl [68,92] utilized by neutrophils to kill bacteria and other pathogens but causes oxidative damage in host tissue [93]. HOCl is derived from chloride ions and H2O2 due to sufficiently high reduction potential provided by MPO (Fig. 4C) [94] and interacts with other small molecules, including NH3, to form monochloramines (NH2Cl) or with other ROS to yield ONOO−, •OH, singlet oxygen (1O2), and ozone (O3) [95].
3. Crosstalk between RONS and inflammation in liver injury
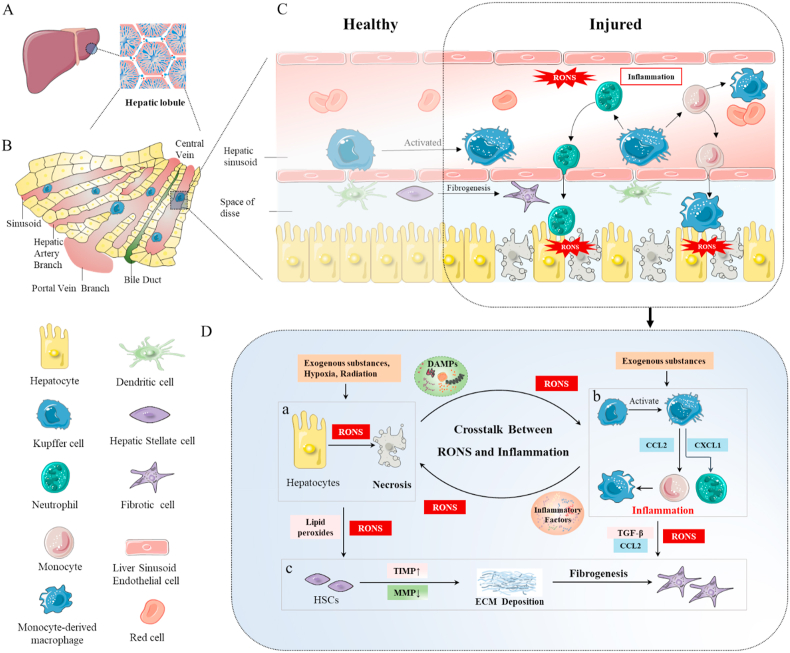
As the crucial organ responsible for metabolism and innate immunity [[96], [97], [98]], the liver has a distinct structure from other organs to meet the needs of various physiological functions, which hidden dangers inherent to a rapid burst of RONS in liver injury as well [99,100]. Hepatic lobule, the functional structural unit of liver, is typically hexagonal in shape and formed by hepatocyte chords (Fig. 5A) [101,102]. The central vein is in the core site of the hexagon while the grouped branches of the hepatic artery, portal vein, and bile ducts form portal triads in vertices (Fig. 5B). In addition to hepatocytes, four other cells types exist in normal liver, including bile duct epithelial cells, hepatic stellate cells (HSCs), Kupffer cells (KCs), and liver sinusoidal endothelial cells (LSECs) [5] (Fig. 5C). Remarkably, LSECs form fenestrated sieve plates at the sinusoidal lumen distinct to the normal endothelial population [31,103]. Blood plasma enters the space of Disse via fenestrae between LSECs, facilitating uptake of plasma substances and excretion of secreted products by hepatocytes. This characteristic feature of endothelial cells endows hepatocytes with the superior capability of retrieving O2 and processing nutrients from blood. As a key metabolic organ, the liver can respond to pathogens and toxins with highly specialized detoxification capacity [104]. During the process, RONS injurious to hepatocytes are excessively produced. Due to their enormous capacity for regeneration, hepatocytes and bile epithelial cells (BECs) undergo proliferation after injury [105,106]. However, hepatic regeneration is impaired in severe acute and chronic liver injury [105,107,108]. As mentioned above, exogenous compounds and stimuli, such as drugs, alcohol, hypoxia, and radiation, directly trigger hepatocyte injury [32,109,110].
Fig. 5.
Overview of the mechanisms underlying liver injury. (A) Hepatic lobule with a hexagonal shape represents the structural and functional unit of liver. (B) The hepatic artery, portal vein, and bile ducts surrounding the lobule provide oxygen, supply nutrients, and store bile for hepatocytes, respectively. The influx of vessels forms capillary-like structures known as sinusoids, which exchange O2 and nutrients directly with hepatocytes. These vessels finally drain into a central vein, the core site of the lobule. (C) Compared with healthy status, RONS, and inflammation-induced induced damage is evident at the injury sites. (D) In terms of crosstalk between RONS and inflammation, RONS could activate inflammatory cells and promote inflammation via direct or indirect pathways. Active inflammatory cells, in turn, generate RONS or interfere with hepatocyte function. (a) Following exposure to exogenous substances or stimuli, excessive RONS generated in hepatocytes induce necrosis, which promotes the release of RONS and DAMPs. (b) RONS and DAMPs activate KCs and recruit monocytes and neutrophils, resulting in inflammation. Active KCs, monocyte-derived macrophages, and recruited neutrophils produce RONS and inflammatory factors (TNF-α, IL-6, IL-1β, etc.) that cause damage to hepatocytes. (c) During the continuous cycle, RONS, lipid peroxides, inflammatory factors, and chemokines constantly stimulate HSCs. Accumulation of the extracellular matrix further leads to liver fibrosis.
To this end, drug-induced liver injury (DILI) is used as an example to illustrate the correlation between RONS and inflammation. DILI is an unexpected harm to the liver resulting from drugs or other xenobiotics and their reactive metabolites [14,111]. Based on their structural characteristics, hepatocytes can obtain and remove drugs from the circulatory system [111,112], which subsequently undergo a series of metabolic processes. First, most drugs are metabolized by cytochrome P450 enzymes [113,114]. This process, also known as Phase I reaction, generates reactive oxidative metabolites that exert potential toxicity on cells via covalent binding with cell proteins during which drug-protein adducts are formed. These intermediate biologically reactive oxidative metabolites interact with various organelles (in particular, mitochondria) [115]. Next, combination of drug metabolites and endogenous molecules (such as glutathione (GSH) and glucuronic acid) inactivate these potentially toxic intermediate products, designated Phase II reaction. Finally, non-toxic metabolites are eliminated from cells through an ATP-dependent efflux pump [116]. However, upon overexposure to drugs (such as acetaminophen or APAP), it is difficult for hepatocytes to tackle exceedingly toxic reactive metabolites from the Phase I reaction [30]. Endogenous molecules of the detoxification system (Phase II) are consumed or absent, leading to accumulation of toxic metabolites. Consequently, mitochondrial function is damaged and large amounts of RONS are produced. Accumulation of RONS further triggers intracellular damage and necrosis (Fig. 5D–a). Eventually, necrosis of hepatocytes leads to release of mitochondrial DNA, HMGB1 (High-mobility group box 1), and ATP [12,55,117], which are known DAMPs. Subsequently, KCs are activated by RONS, DAMPs, and other factors. Simultaneously, monocytes and neutrophils are recruited by pro-inflammatory factors, chemokines, and DAMPs. Monocytes further differentiate into macrophages to promote an inflammatory environment (Fig. 5D–b). RONS are directly produced and released into activated inflammatory cells [81,118]. Inflammation mediates cell death through two main pathways: (1) inflammatory factors induce cell necrosis through receptor-interacing protein kinase 3 (RIPK3)/mixed lineage kinase domain-like protein (MLKL) and other pathways [119,120] and (2) RONS released by various inflammatory cells cause bystander damage, along with proteases and antibacterial proteins [121,122]. Collectively, RONS either directly or indirectly induce inflammation in liver injury. The crosstalk between RONS and inflammation induces a vicious cycle of hepatocyte injury.
Hepatocyte injury is considered a key trigger for progression of various liver diseases. Severe hepatocyte necrosis, if not effectively controlled, will quickly lead acute liver failure or even multiple organ failure, and eventually, death [121,123]. Chronic liver injury may develop into liver fibrosis, cirrhosis, and ultimately, cancer due to relatively low-grade cell death and sterile inflammation [124,125]. Chronic liver injury and liver cirrhosis are major risks responsible for the liver cancer, but the mechanism of liver cancer is quite different. Antioxidant and anti-inflammatory drugs are not main modalities of liver cancer therapy. Many reviews have summarized therapies of liver cancer [[126], [127], [128], [129]]. Above all, nanodrugs for liver cancer are not included in this review. The mechanisms are mainly related to fibroblast activation, including HSCs and portal vein fibroblasts, induced by the long-term action of RONS, cytokines (TGF-β), chemokines (CCL2), and lipid peroxides [125]. Imbalance in the expression of extracellular matrix (ECM) remodeling-related genes in activated fibroblasts results in upregulation of tissue inhibitors of metalloproteinases (TIMP) and reduction in the clearance efficiency of matrix metalloproteinases (MMP). Consequently, ECM proteins (including fibrillary collagen, α-SMA, and LAMININ) accumulate, in turn, leading to tissue injury and liver fibrosis (Fig. 5D–c) [32,125]. In addition, the pro-inflammatory mechanism of myofibroblasts involving RONS-mediated migration (via the production of polymorphic chemotactic substances, platelet-derived growth factor (PDGF), CCL2, vascular endothelial growth factor (VEGF), angiotensin II, etc.) and promotion of angiogenesis further enhance progression of fibrosis [124,125].
4. Advantages of nanodrugs for treatment of liver injury
Due to its complex function, liver receives ∼25% of the whole cardiac output despite only constituting 2.5% of total body weight [130,131]. While a massive amount of blood flows to hepatic lobules, hepatocytes are only in contact with conventional therapeutic reagents for a short time (few seconds to minutes) [11]. These reagents are rapidly metabolized and inactivated by hepatocytes. In addition, small-molecule drugs move into and pass through cells mainly via passive diffusion, lacking selectivity in their biodistribution [132,133]. Thus, systemic side effects of drugs are hard to avoid during the process of reaching the lesion site through the vascular endothelial system. Moreover, numerous anti-oxidant and anti-inflammatory agents fail to exert clinical effects [134,135]. Only one antioxidant, N-acetyl-l-cysteine (NAC) is currently approved to treat DILI, but its treatment time window is narrow and efficacy is limited [30]. At present, treatment of various liver injuries is still confined to conventional supportive therapy and removal of etiologic factors [30,43,136]. For instance, extracorporeal liver support systems relieve liver metabolic pressure and use activated carbon purification to treat APAP-induced liver injury [137].
The accumulation of nanodrugs in healthy tissues is undeniable harmful, given that RONS and inflammation play a meaningful role in maintaining the whole body's normal metabolic process and defending defense against invading pathogens [[138], [139], [140]]. The dose as low as possible to the normal tissues could avoid corresponding side effects of nanodrugs and no significant toxicity was reported in the treatment of nanodrugs to liver injury. Therefore, the sequestration effect of the liver provides significant advantages for nanodrug therapy of liver injury. The endothelial system of liver with fenestrae is different from other tissues, which endows nanodrugs with unique advantages for therapeutic purposes (Fig. 6A). Furthermore, modification of nanodrugs in terms of physical (including size, shape, and uniformity), chemical (including composition, charge, and surface coating) and biological (including encapsulated compounds and specific conjugated ligands) features facilitate their efficient and selective targeting of pathology sites [141,142]. Nanodrugs cannot penetrate the vascular endothelial system of normal non-hepatic tissues (Fig. 6B) and nanoparticles tend to accumulate and target lesions with incomplete endothelial lining and basement membrane [[143], [144], [145], [146]]. Overall, 30–99% nanodrugs are reported to aggregate in the liver after administration into the body via different routes [147].
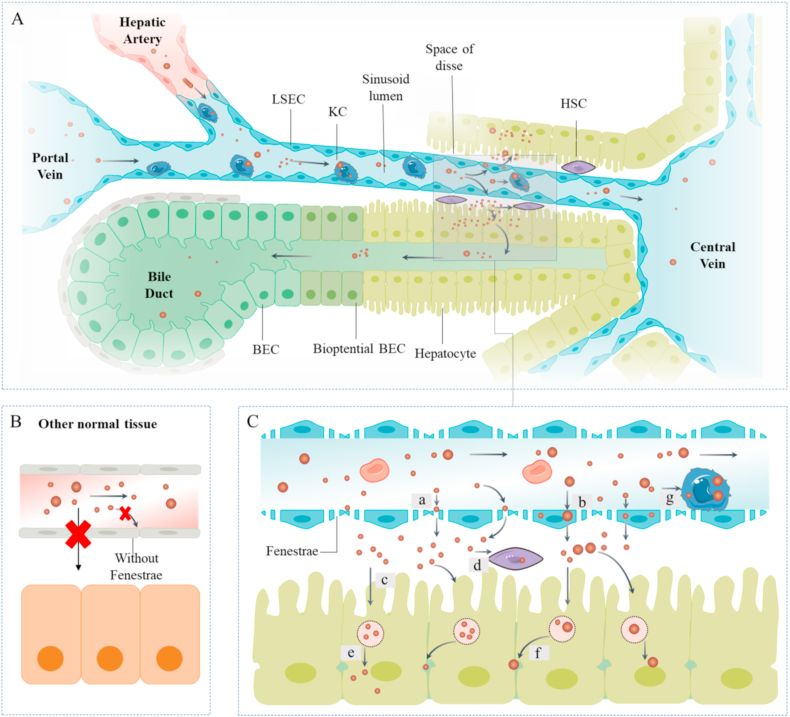
Fig. 6.
Schematic of metabolic processing of nanoparticles in liver. (A) Nanoparticles injected intravenously drain into liver sinusoid through the portal vein and hepatic artery after entry into the systemic circulation. The nanoparticles in sinusoid are taken up by distinct cell types via various pathways or excreted into bile. (B) Notably, no fenestrae exist between vascular endothelial cells in other normal tissues, which restricts entry of nanoparticles into target tissue. (C) Fate of nanoparticles in liver. Smaller nanoparticles exit the fenestrae directly into space of disse (a). Some nanoparticles are taken up by LSECs through receptor–ligand interactions and released if not removed by LSECs (b). Nanoparticles accumulating in space of disse are taken up by hepatocytes (c). Some target nanoparticles are absorbed by HSCs (d). Nanoparticles in hepatocytes escape from the endosome into cytoplasm (e) or are excreted into bile (f). Nanoparticles may be taken by KCs, especially those with larger sizes (g).
Due to the fenestrated sieve plates between LSECs, which range from 50 to 180 nm in humans or up to 280 nm in mice and rats [5], smaller nanodrugs directly pass the sinusoid into disse of space. Nanodrugs are also taken up by LSECs through receptor–ligand interactions and subsequently LSECs transported to disse of space if not consumed. Subsequently, nanodrugs in the disse interact with hepatocytes, leading to their uptake. Modification of nanodrugs, such as alterations in targeting molecules, PEGylation, and imparting positive charges on the surface, can also greatly increase uptake by hepatocytes [148,149]. In addition, larger nanoparticles tend to be captured by KCs after entering hepatic sinusoids through portal vein or hepatic artery due to their greater surface area for interactions [147,150]. Considering the critical role of KC-related inflammation in liver injury, enrichment of anti-inflammatory and antioxidant nanoparticles in KCs also provides favorable conditions for treatment (Fig. 6C). Notably, sinusoid capillarization occurs when LSECs are injured during liver injury, especially chronic liver injury and liver fibrosis. The capillarization process leads to loss of fenestrae and development of a basement membrane, which impedes the entry of nanoparticles through fenestrae to some extent [31]. Antioxidant and anti-inflammatory molecules are reported to prevent and alleviate sinusoid capillarization through protecting LSECs as potential therapeutic options to relieve chronic liver disease and its complications [151,152]. In the articles collected, cerium oxide nanoparticles are demonstrated to reduce portal hypertension, a kind of chronic liver disease complication promoted by sinusoid capillarization, and accumulate in liver parenchyma [153]. Therefore, the nanoparticles with RONS scavenging and anti-inflammatory effects could reach hepatocytes through alleviating ameliorating loss of fenestrae as one important aspect. But a further investigation is necessary to support the underlying mechanism.
Using the above pathways for transport into hepatocytes, nanodrugs significantly improve the bioavailability of drugs, prolong their therapeutic effects, and achieve combination or targeted therapy. For example, lipidized somatostatin analogue can integrate with the hepatocyte membrane to a greater extent, which promotes accumulation of drugs in the liver (by 3.8 times) [154]. The significant success of ONPATTRO, the first liver-targeted lipid nanodrug approved by the FDA, substantiates the potential application value of nanodrugs in the future [155].
5. RONS-scavenging nanodrugs
Powerful antioxidant enzyme systems for scavenging RONS, such as SOD, catalase (CAT), peroxidase (POD) and glutathione peroxidase (GPx), are present in cells [26,30,156], but insufficient to cope with liver injury. The utility of antioxidant enzymes in liver injury has attracted widespread research attention from the beginning of the 21st century [157,158]. However, these antioxidant enzymes are often ineffective in vivo owing to their instability in the blood circulatory system [33,159,160]. Two strategies have been developed to resolve this bottleneck: (1) nanozymes with good stability and multiple antioxidant enzyme activities can be used to replace natural antioxidant enzymes and (2) nanocarriers containing natural antioxidant enzymes can be developed to increase bioavailability by improving stability in the blood system.
5.1. Antioxidant nanozymes
Oxidative stress is triggered by a variety of RONS in liver injury. Elimination of a single type of RONS therefore has limited efficacy for treatment of liver injury. Antioxidant nanozyme, a type of artificial enzyme based on nanomaterials, can achieve multiple functions (including multiple antioxidant enzyme activities, regulation of catalytic activity, and targeting of liver) based on regulation of the structure, composition, size, and surface modification [161], and is therefore useful as a therapeutic option. Antioxidant nanozymes for treatment of liver injury are mainly based on cerium oxide, melanin-like carbon (fullerene, graphene, and other carbon nanomaterials) and other nanomaterials, such as selenium and MXene.
5.1.1. Cerium oxide nanoparticles
Upon reduction of the size of cerium (IV) oxide to the nanometer scale, many oxygen vacancies are formed, leading to two valence states of +3 and + 4 of cerium on the nanoparticle surface. The valence state ratio of +3/+4 becomes higher with smaller nanoparticle size. Cerium oxide nanoparticles (CeO2 NPs) represent a widely investigated type of multi-antioxidant with potent nanozyme activities, including SOD and CAT mimetic properties [162]. SOD mimetics are reflected in the elimination of O2•− through cerium (III) sites (Ce3+ + 2H+ + O2•− → H2O2 + Ce4+). Cerium (III) sites are additionally accountable for removal of •OH via redox reactions (2Ce3+ + 2•OH → 2Ce4+ + H2O+1/2 O2). Cerium (IV) sites achieve clearance of H2O2 through CAT mimetic activity (Ce4+ + H2O2 + OH− → 1/2 O2 + Ce3+ + H2O) [163]. Although no obvious toxicity was reported in the treatment of CeO2 NPs to liver injury, previous literature indicated the development of rare earth pneumoconiosis accompanied by granulomatous inflammation and interstitial fibrosis when the lungs are constantly exposed to cerium oxide nanoparticles [[164], [165], [166]]. Given Ce in lung tissue was only 33–36% pure Ce4+ after administration of CeO2 NPs [167], the increasing toxicity determined by the Ce3+/Ce4+ ratio in vivo should be carefully considered. Since the lung is not an enriched target of CeO2 NPs in liver injury treatment and increasing studies focus on its mechanism of inducing pulmonary fibrosis, the double-edged sword may be a sharp weapon in the future.
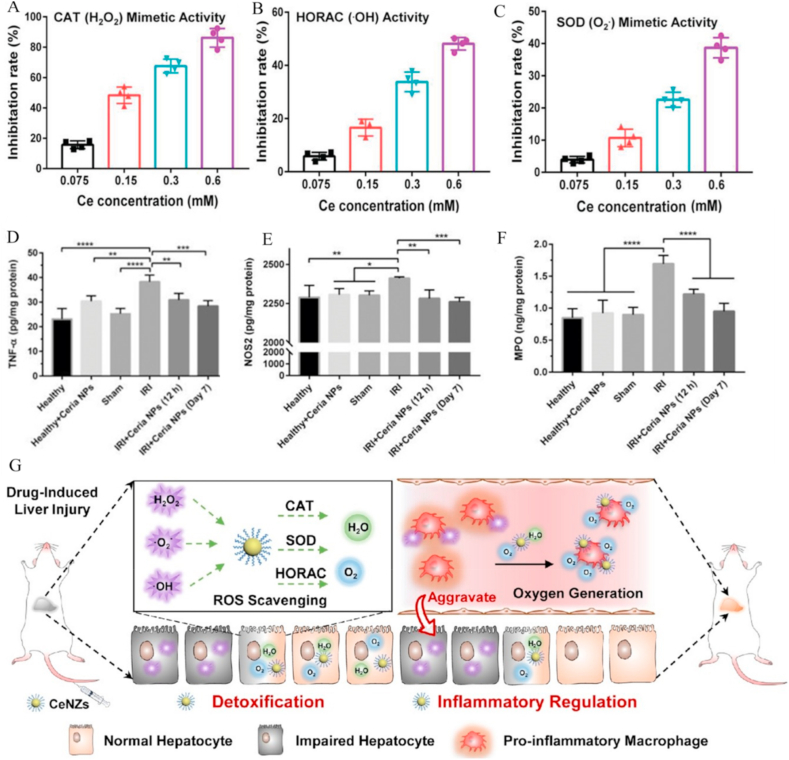
CeO2 NPs have been widely used in the treatment of liver disease owing to their strong ability to eliminate a variety of free radicals, including hepatic IRI, injury induced by drugs or toxins, radiation, inflammation, and liver fibrosis [153,[168], [169], [170], [171], [172], [173], [174]]. For example, a study by Denise and co-workers demonstrated that CeO2 NPs could reduce inflammation in rats with liver fibrosis [153]. The majority of CeO2 NPs effectively localized in the liver with antioxidant and anti-inflammatory effects after intravenous administration. Notably, mRNA expression of signals related to oxidative (GPx, CAT, Ncf1, Ncf2) or endoplasmic reticulum stress (Atf3, Hspa5) signaling pathways and expression of inflammatory cytokines (TNF-α, IL-1β, COX-2) was significantly changed owing to the efficient ability of CeO2 NPs to eliminate a variety of RONS in liver injury. CeO2 NPs have also been developed to reduce acute liver injury caused by ischemia and reperfusion. Ni et al. [175] modified the surface of CeO2 NPs with PEG (PEG-CeO2 NPs) to improve their biocompatibility. PEG-CeO2 NPs could effectively capture ROS (O2•−, •OH, and H2O2) to reduce lipid peroxidation and inhibit the production of pro-inflammatory cytokines (IL-1, IL-12, TNF-α, INF-γ, and NOS2) (Fig. 7A–F). After PEG-CeO2 NP treatment, liver injury was efficiently repaired and function returned to normal.
Fig. 7.
Cerium oxide nanoparticles for liver injury. (A–C) Scavenging performance of ceria NPs via mimicking catalase (CAT) (A), elimination of •OH (B) and SOD mimics (C). (D–F) Anti-inflammatory activities of ceria NPs with TNF-α (D), NOS2 (E), and MPO (F) in liver. Reprinted with permission from Ref. [175]. (G) Schematic illustration of the mechanisms underlying DSPE-PEG-CeO2 NP activity against DILI. Reprinted with permission from Ref. [176].
Recently, Ling et al. [176] developed CeO2 NPs modified with 1,2-distearoyl-sn-glycero-3-phosphoethanolamine-N-[methoxy (polyethylene glycol)-2000] (DSPE-PEG) to treat APAP-induced liver injury. DSPE-PEG greatly improved the bioavailability and biocompatibility of CeO2 NPs. DSPE-PEG-CeO2 NPs exhibited high SOD mimetic, •OH elimination and CAT mimetic activities. In particular, the CAT mimic activity of DSPE-PEG-CeO2 NPs converted harmful H2O2 into O2, which alleviated the hypoxic environment to decrease inflammation (Fig. 7G). Compared with NAC, the first-line clinical drug for treatment of DILI, DSPE-PEG-CeO2 NPs had a long-lasting therapeutic effect and wider therapeutic time window [177]. Therefore, DSPE-PEG-CeO2 NPs with a longer therapeutic window have significant clinical application prospects in the treatment of DILI.
5.1.2. Melanin-like nanoparticles
Melanin is a naturally occurring biological polymer present in living organisms displaying superior properties of biocompatibility, biodegradability, and renewability that has attracted widespread attention. To overcome water insolubility for applicability in biomedicine, melanin nanoparticles (MNP) and melanin-like nanoparticles (MLNP) have been generated to treat various diseases [[178], [179], [180]]. Owing to multiple functional groups, such as catechol, amine, and imine, MNPs and MLNPs possess potent antioxidant activity. Our group showed that MNPs could scavenge multiple RONS, including O2•−, H2O2, •OH, •NO, and ONOO−. The catalytic performance of SOD mimetics, manifested by half-reactions (Melanin• + O2•− → Melanin−+ O2, Melanin• + O2•− +2H2O→ Melanin−+ H2O + 2OH−), has also been verified [181].
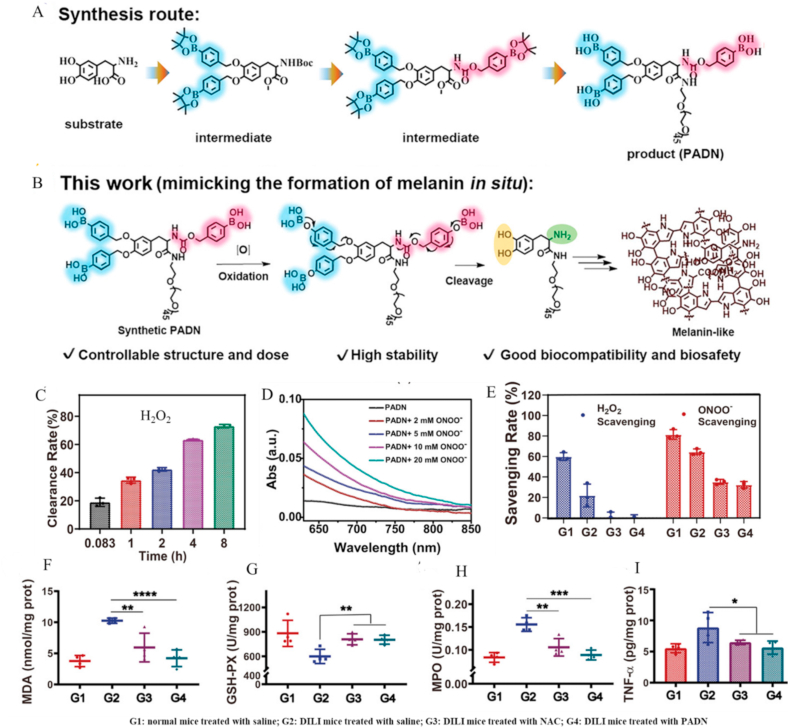
Recently, Shi et al. [182] developed well-defined melanin-like nanoparticles (PADN) for acute liver injury therapy. PADNs were obtained via self-assembly of a PEGylated, phenylboronic-acid-protected l-DOPA precursor by mimicking the biosynthesis process of melanin (Fig. 8A&B). Due to their stable chemical structure, the actual composition and dosage of PADNs could be precisely controlled. In addition, antioxidant functional groups on the surface of l-DOPA were protected by phenylboronic acid groups conjugated via RONS-responsive self-immolative linkages. As a result, PADNs displayed controllable and enhanced antioxidative activity along with superior physiological stability. Specifically, PADNs could scavenge H2O2 and ONOO− in a time- and concentration-dependent manner that was significantly better than PEG-DOPA, oxidized PADN, and MNPs (Fig. 8C–E). Surprisingly, ∼100% ONOO− was eliminated only after 1 h of co-incubation with PADNs in vitro. PADNs could prevent and treat drug-induced acute liver injury/failure in APAP-administered mice through reducing oxidative stress and the inflammatory response (Fig. 8F–I).
Fig. 8.
Melanin-like nanoparticles as therapy for APAP-induced acute liver injury. (A) Schematic illustration of the synthesis process of PEGylated phenylboronic-acid-protected l-DOPA nanoparticles (PADN). (B) PADN acts as a tyrosine mimic to scavenge ROS via melanin-like nanoparticles similar to natural tyrosine. (C) H2O2 scavenging effect of PADN measured at different time-points (PADN concentration of 20 × 10−6 M). (D) ONOO− scavenging effect of PADN measured at different concentrations. (E) RONS scavenging effect of different treatments (G1: PADN; G2: PEG-DOPA; G3: ONOO−-oxidized PADN; G4: Mel-NPs). (F–I) Mechanism of PADN action in DILI of mice. Levels of MDA (F), GSH-PX (G), MPO (H), and TNF-α (I) measured in liver from each group (G1: normal mice treated with saline; G2: DILI mice treated with saline; G3: DILI mice treated with NAC; G4: DILI mice treated with PADN). Reprinted with permission from Ref. [182].
5.1.3. Carbon-based nanoparticles
Compared with natural enzymes, carbon-based nanomaterials have significant advantages, such as low cost and stability [183]. These nanomaterials used in liver injury to date are mainly divided into three categories: carbon nanomaterials, graphene nanomaterials, and fullerenes. All nanomaterials contain a large area of conjugated π-electronic structure domain. Among these, carbon and graphene-based nanomaterials have two-dimensional conjugated structural domains while fullerenes have three-dimensional conjugated structural domains [184,185]. Electrons can flow freely in conjugated structure domains, which combine with free electrons on RONS, facilitating efficient elimination of RONS. Moreover, carbon-based nanomaterials do not contain metal elements and have good biocompatibility, further supporting their utility in the treatment of liver injury.
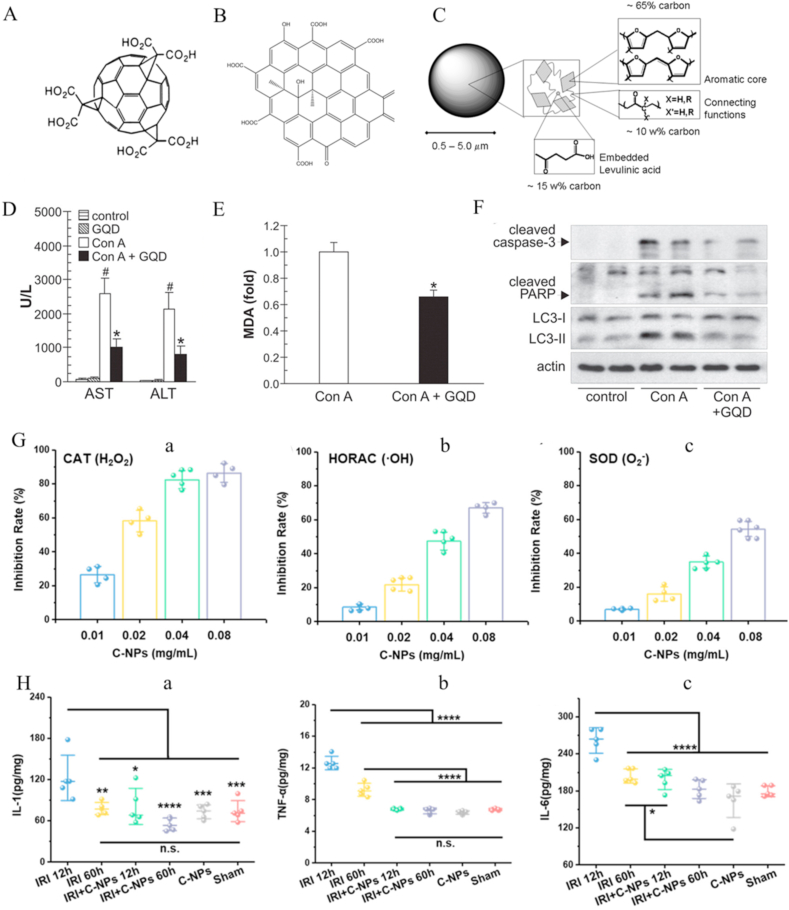
Fullerene, the first carbon-based nanomaterial for liver disease, is hydrophobic and cannot be used directly [[186], [187], [188], [189], [190], [191], [192], [193], [194]]. Carboxylation and hydroxylation of fullerenes significantly improve their aqueous solubility. For instance, Chen et al. [186] developed a carboxy-fullerene (C3) to reduce oxidative stress and inflammation in mice with hepatic IRI (Fig. 9A). C3 had higher aqueous solubility than fullerene and could scavenge multiple RONS to reduce lipid peroxidation in liver tissue after hemorrhagic shock. Moreover, the levels of key inflammatory molecules (TNF-α and IL-6), expression of NF-κB (a key signaling protein for inflammation), and activity of MPO in neutrophils were significantly reduced in liver after C3 treatment. Both graphene and carbon materials can also scavenge RONS, similar to fullerenes, since they contain a π-electronic structure domain (Fig. 9B&C). Vladislav and co-workers showed that graphene quantum dots (GQD) could effectively scavenge O2•− and •OH [187]. In addition to the π-conjugated structure domain, the ROS quenching potential of graphene is derived from defects and unpaired electrons at the surface. GQDs inhibited lipid peroxidation, apoptosis, and autophagy in concanavalin A (ConA)-induced mouse hepatitis (Fig. 9D–F) [187]. Recently, Long et al. [190] developed hydrophilic carbohydrate-derived carbon nanoparticles (C-NPs) to prevent hepatic IRI injury. The abundant π-electronic structure of domains of C-NPs could effectively scavenge H2O2, •OH, and O2•− (Fig. 9G). After C-NP treatment, activation of KCs and macrophages was suppressed by eliminating excessive ROS and levels of key inflammatory factors (IL-1, TNF-α, and IL-6) were reduced in the liver (Fig. 9H). Finally, the key indicators of liver injury (aspartate aminotransferase (AST) and alanine aminotransferase (ALT)) were restored, suggesting recovery of liver function.
Fig. 9.
Carbon-based nanoparticles for liver injury. (A) Chemical structure of carboxyfullerene. Reprinted with permission from Ref. [195]. (B) Structure of graphene quantum dots. Reprinted with permission from Ref. [196]. (C) Structural model of carbohydrate-derived nanoparticles. Reprinted with permission from Ref. [197]. (D–F) Single intravenous injection of GQDs (50 mg/kg) significantly suppresses Con A hepatotoxicity by reducing serum levels of liver transaminases (D), lipid peroxidation (MDA, malondialdehyde) (E), and expression of proapoptotic and autophagy-related proteins (F). Reprinted with permission from Ref. [187] (G) In vitro scavenging activity of C-NPs as CAT (a), HORC (b), and SOD (d). (H) ELISA results of IL-1 (a), TNF-α (b), and IL-6 (c) from activated monocytes/macrophages and KCs measured in liver homogenates from each group subjected to different treatments. Reprinted with permission from Ref. [190].
5.1.4. Other nanozymes
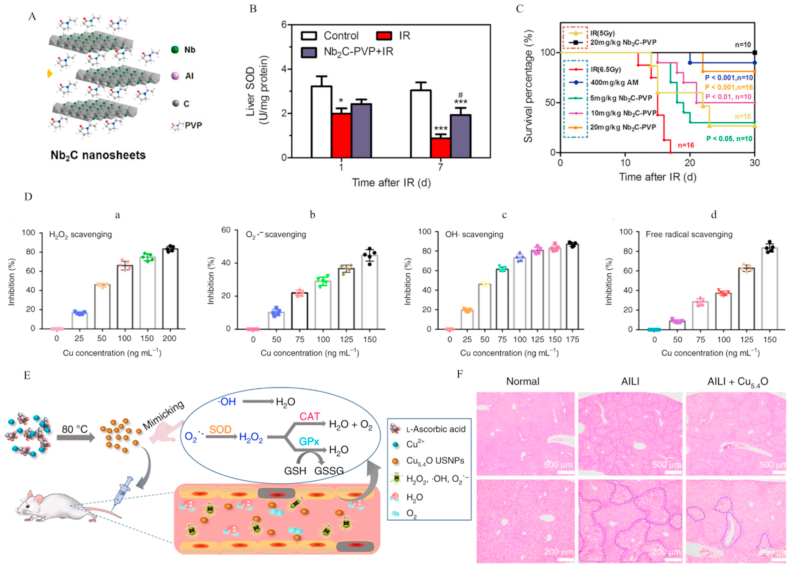
Mxene is an ultra-thin nanosheet with a two-dimensional structure composed of an early transition metal, carbon, and nitrogen [162,198]. This unique structure imparts unique physical and chemical properties and biological effects [199]. Recently, Ren et al. [200] developed ultrathin two-dimensional niobium carbide nanosheets functionalized with biocompatible polyvinylpyrrolidone (2D ultrathin Nb2C-PVP MXenes) as a radiotherapeutic protective agent for ionizing radiation (IR)-induced liver injury (Fig. 10A). IR induced large amounts of ROS (such as O2•− and •OH) in liver. Nb2C eliminated O2•− via direct reaction. In addition, •OH specifically bound the [CNb3] site of the Nb2C plane and was efficiently eliminated. The 2D ultrathin Nb2C-PVP MXenes could effectively eliminate oxidative damage caused by IR, restore activity of SOD, and improve liver injury (Fig. 10B). The survival rates of mice were greatly improved (from 30% to 81.25%) under 5 Gy irradiation after 2D ultrathin Nb2C-PVP MXene treatment (Fig. 10C).
Fig. 10.
Other nanozymes for treatment of liver injury. (A) Schematic presentation of Nb2C-PVP structures. (B) ROS scavenging activity of Nb2C-PVP, reflected by total SOD activities in liver of BALB/C mice (C) Protective effect of Nb2C-PVP in BALB/C mice exposed to sublethal TBI (5 Gy) and lethal TBI (6.5 Gy). Overall, 30-day survival rates were measured in different groups. Reprinted with permission from Ref. [200] (D) ROS scavenging ability of Cu5·4O USNP:H2O2 (a), O2•− (b), •OH (c), and free radicals (d). (E) Schematic preparation and mechanisms of action of Cu5·4O USNPs. (F) Hepatoprotective effects of Cu5·4O USNPs in APAP-induced liver injury determined via analysis of H&E staining of liver tissues from each group. Blue dashed lines indicate the range of hepatic necrosis. Reprinted with permission from Ref. [207]. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
Selenium (Se) and copper (Cu) nanoparticles have recently been used for treatment of liver injury [[201], [202], [203], [204], [205], [206], [207], [208], [209]]. Selenium, an essential trace element for animals and humans, is a core component of antioxidant enzyme GPx [210]. However, traditional selenium supplements usually have lower rates of absorption [211]. Nanoparticles greatly improve the bioavailability of selenium. Moreover, selenium nanoparticles (SeNPs) have superior properties in direct removal of RONS. Recently, Kaikai and co-workers demonstrated that Se nanoparticles modified with a positively charged natural polysaccharide chitosan (CS-SeNPs) could directly eliminate O2•− and •OH. In liver injury mediated by Con A, CS-SeNPs induced a significant reduction in liver necrosis and restored liver function [202,206]. Similarly, copper is an essential trace element that plays a key role in the activities of many enzymes, including tyrosinase and Cu–Zn SOD [212,213]. Recently, Song et al. [207] synthesized ultrasmall Cu5·4O nanoparticles (Cu5·4O USNPs) with prior biocompatibility and enzymatic ROS scavenging abilities (Fig. 10E). The proportions of Cu and Cu2O in Cu5·4O USNPs were ∼3.4. Cu5.4O USNPs could exert multienzyme-like antioxidative activity to quench H2O2, O2•−, and •OH (Fig. 10D). In particular, the H2O2-scavenging efficiency was ∼2000-fold stronger than that of traditional small molecular antioxidants, such as vitamin C. Overall, Cu5·4O USNPs showed excellent hepatoprotective activities against DILI (Fig. 10F).
Very recently, two novel nanozymes, molybdenum disulfide (MoS2) and prussian blue (PB), were shown to be a very good treatment effect for liver fibrosis and anthracycline-induced liver injury, respectively. MoS2 nanosheets, as typical two-dimensional 2D transition metal dichalcogenide nanomaterials, were reported to exhibit SOD, POD, CAT mimetic activity. Zhou et al. [214] prepared MoS2 nanosheets via liquid exfoliation of bulk MoS2 in N, N-dimethylformamide (DMF). In addition to multienzyme mimetic activities, including SOD, CAT, POD and GPx, MoS2 nanosheets were indicated with protective potential of mitochondrial ETC. Of note, prior CAT activity in acidic environment was revealed by studying the interaction between MoS2 nanosheets and H2O2 and O2•−. Eventually, MoS2 nanosheets inhibited inflammation and ameliorated fibrosis in CCl4-treated murine model. PB have been approved by FDA as an antidote agent with excellent biosafety. Similarly, multienzyme-like activity of PB nanozyme has attracted intensive attention. Zhang et al. [215] synthesized monodispersed polyvinylpyrrolidone (PVP)-modified PB nanozymes (PBZs) to prevent anthracycline-induced liver injury, which is an increasingly serious and potential clinical complication in tumor patients treated with anthracyclines. In general, PBZs could attenuate oxidative stress and inflammation by scavenging RONS and regulating antioxidative genes and MPO.
5.2. Nanocarriers of SOD
Superoxide dismutase (SOD) is a powerful antioxidant enzyme that functions in eliminating O2•− but has a short half-life in the blood circulation and cannot penetrate the cell membrane. The half-life of SOD can be extended in the blood circulatory system through specific chemical modifications (mannosylation or coupling to divinyl ether and maleic anhydride) to circumvent clearance in the reticuloendothelial system [216]. Using this strategy, Piter et al. [217] showed that chemically modified SOD eliminates O2•− to reduce inflammation in rats with liver fibrosis. However, activity of chemically modified SOD may still be lost in lysosomes of hepatocytes.
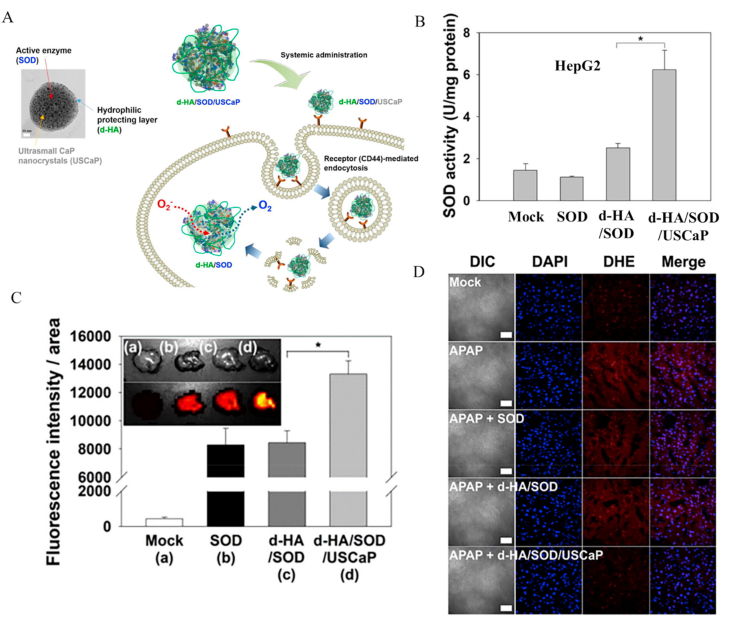
Nanocarriers not only effectively improve the stability of SOD [218] but also aid in escape from lysosomes into the liver cytoplasm. Recently, Lee et al. [219] developed a nanocarrier of SOD (d-HA/SOD/USCaP NPs) to treat APAP-induced liver injury. d-HA/SOD/USCaP NPs were composed of two parts: l-dopa-derivatized hyaluronic acid (d-HA) polymer network associated with SOD and small calcium phosphate nanoparticles dispersed in the polymer network. HA not only effectively protected SOD in the blood circulation but also targeted hepatocytes by binding to HA receptors (CD44) on the surface (Fig. 11A). When d-HA/SOD/USCaP NPs enters lysosomes of hepatocytes, constituent calcium phosphate is decomposed into calcium ions and phosphate ions in the acidic environment, which leads to changes in the osmotic pressure in lysosomes and release of SOD into the liver cytoplasm. d-HA/SOD/USCaP NPs successfully delivered SOD efficiently in vivo and significantly reduced the liver toxicity of APAP by eliminating O2•− (Fig. 11B–D).
Fig. 11.
Nanocarriers of SOD for treatment of APAP-induced liver injury. (A) Uptake and release process of d-HA/SOD/USCaP. (B) Radical scavenging activities of SOD, d-HA/SOD, and d-HA/SOD/USCaP in HepG2 cells. (C) Accumulation of SOD, d-HA/SOD, and d-HA/SOD/USCaP in liver. (D) ROS levels in liver tissues of the APAP-induced mouse model treated with SOD, d-HA/SOD, and d-HA/SOD/USCaP. Reprinted with permission from Ref. [219].
6. Nanocarriers of small-molecule antioxidants
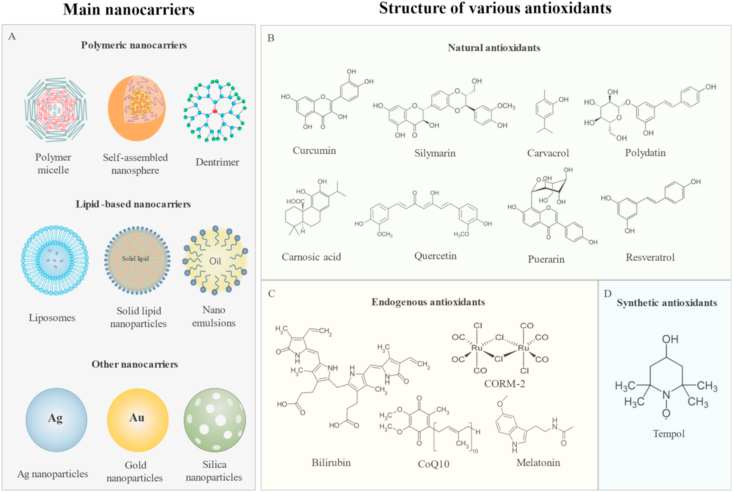
Currently, several small-molecular antioxidants are clinically used to prevent excessive oxidation in a number of diseases. However, biocompatibility of many antioxidants is not ideal for treatment of liver injury. NAC is the only approved first-line drug [177] and serves as a prodrug of l-cysteine, a precursor of the biological antioxidant glutathione. However, Several reports have documented patients with DILI that have missed the time window of NAC treatment and developed acute liver failure leading to death [220]. While natural antioxidants have better biocompatibility and lower side effects than their synthetic counterparts, neither synthetic nor natural antioxidants meet the requirements for effective treatment of liver injury. The major problems are low bioavailability and poor targeting. Numerous therapeutic nanocarriers have been developed to improve loading of antioxidants to date. In these systems, nanocarriers are generally composed of polymers and liposomes, along with a small percentage of other nanocarriers (Fig. 12A). The loaded small-molecule antioxidants can be divided into three categories: natural antioxidants (such as curcumin [[221], [222], [223], [224], [225]], silymarin [[226], [227], [228], [229]], resveratrol [230], carvacrol [231], polydatin [232], carnosic acid [233], quercetin [234,235], and other compounds [231,[236], [237], [238], [239], [240], [241], [242], [243], [244], [245], [246], [247], [248], [249], [250], [251], [252]]), synthetic antioxidants (nitroxide 4-amino-2,2,6,6-tetramethylpiperidine-N-oxyl, tempol [[253], [254], [255], [256], [257], [258]]), and human endogenous antioxidants (melatonin [259,260], bilirubin [261], ubiquinone (CoQ10) [262], and carbon monoxide (CO) [263]) (Fig. 12B–C). All antioxidant-containing nanocarriers exert enhanced therapeutic effects relative to conventional antioxidants by greatly improving bioavailability and reducing side effects.
Fig. 12.
Schematic representation of the structures of main nanocarriers and various antioxidants. (A) Nanocarriers of antioxidants for liver injury therapy are mainly composed of polymers and liposomes, along with a small proportion of other nanocarriers. (B) The main feature of plant polyphenols is one or more aromatic rings with one or more hydroxyl groups attached. The utility of curcumin, resveratrol, quercetin, and other plant polyphenols were explored in different liver injury models after loading onto nanocarriers. Although plant polyphenol compounds play a key role in the field of natural antioxidants, the important functions of other types of natural antioxidants, such as puerarin, cannot be overlooked. (C) Melatonin, bilirubin, CoQ10 and carbon monoxide releasing molecule-2 (CORM-2) acting as a carbon monoxide-releasing molecule are important endogenous non-enzymatic antioxidants in the body. (D) As a membrane-permeable radical scavenger, nitroxide 4-amino-2,2,6,6-tetramethylpiperidine-N-oxyl (tempol) effectively promotes metabolism or inhibits generation of various RONS based on the characteristics of NO.
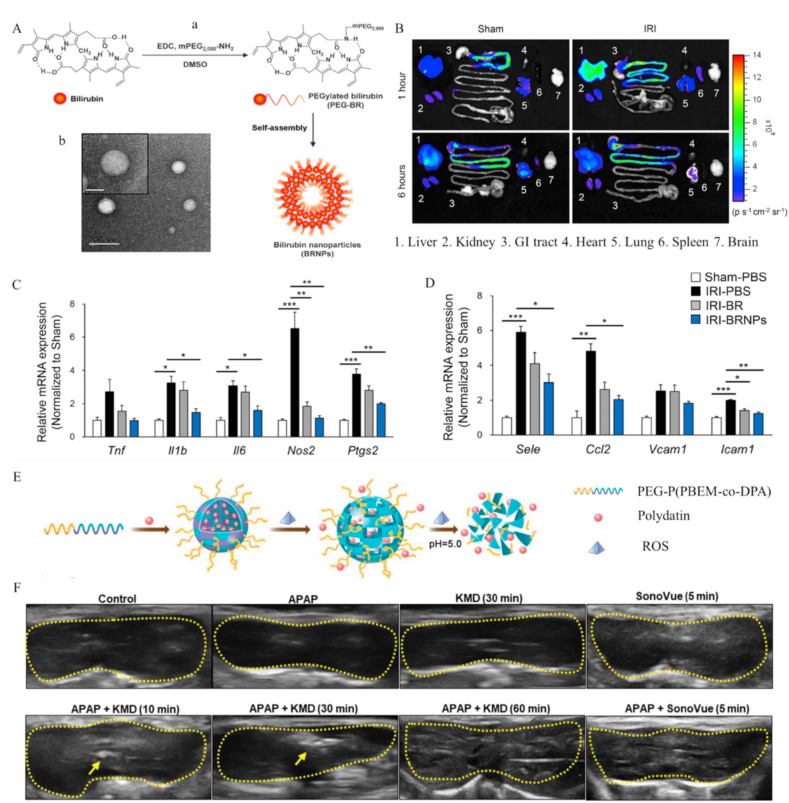
Bilirubin is an important endogenous non-enzymatic antioxidant in the body similar to glutathione. However, owing to its water insoluble and easily oxidizable properties, bilirubin is unsuitable for treatment of liver injury [264,265]. Earlier, Kim and co-workers [261] developed a PEG-based nanocarrier loaded with bilirubin (BR NP) for treatment of hepatic IRI. PEG was covalently bonded to biliverdin via an amide bond to form an amphiphilic PEG-bilirubin complex (Fig. 13A) that could self-assemble in an aqueous environment, generating BR NPs. PEG improved the bioavailability of bilirubin through prolonging systemic circulation time. Moreover, BR NPs were specifically enriched at the site of hepatic IRI. Relative accumulation of BR NPs at the liver injury site was 3.4 times higher than that in normal liver at 1 h after injection (Fig. 13B). Notably, BR NPs effectively suppressed ROS levels, production of inflammatory factors (IL-1β, IL-6, NOS2, Ptgs2, NF-κB), and infiltration of neutrophils (Fig. 13C&D). After treatment, the necrotic and apoptotic areas of the liver after ischemia and reperfusion were greatly reduced and liver function was restored [261].
Fig. 13.
Nanocarriers with antioxidants for treatment of liver injury. (A) Schematic illustration of fabrication of BRNPS [264] (a). Representative TEM images of BRNPs (b). Reprinted with permission from Ref. [261] (B) Representative images of major organs extracted from sham-operated control and IRI-induced mice at 1 h and 6 h post injection illustrating biodistribution of BRNPs in a hepatic IRI model. (C) Relative expression of mRNAs of pro-inflammatory mediators, including TNF, IL1b, IL6, Nos2, and Ptgs2 and (D) cell adhesion molecules (Sele, Ccl2, Vcam1, and Icam1) in liver after IRI [261]. (E) Schematic synthesis of PEG-P (PBEM-co-DPA)-polydatin and illustration of the release process of PEG-P (PBEM-co-DPA)-polydatin as a ROS and pH dual-responsive nanodrug. Reprinted with permission from Ref. [232] (F) Representative ultrasound images of liver in APAP-induced liver injury mice. Dotted lines indicate liver contours and arrows indicate echogenic KMD nanoparticles. Reprinted with permission from Ref. [258].
Multifunctional property is another major advantage of nanodrugs [266]. Owing to the flexibility of nanocarrier design, it is possible to integrate targeting and imaging functions with RONS-scavenging nanodrugs. Fundamentally, two distinct methods have been proposed for targeting of damaged liver: modifying the nanocarrier with targeting molecules and designing RONS-sensitive nanocarriers. For example, Swaran and co-workers added lactobionic acid (LA), a molecule targeting the asialoglycoprotein receptor (ASGPR) on hepatocytes, to quercetin-loaded organically modified silica nanoparticles (Q-ORMOSIL) [235]. Targeted quercetin nanoparticles with improved solubility and bioavailability exerted hepatoprotective effects at a 1000 times lower dose (50 μg/kg/day) compared to bulk quercetin alone (50 mg/kg/day). Zhu and co-workers further synthesized ROS and pH dual-sensitive block polymer PEG-P (PBEM-co-DPA) as a nanocarrier for polydatin [232]. The pinacol-type boronic ester on side-chains hydrolyzed into quinone methide at acidic pH or in the presence of ROS (Fig. 13E). Thus, polydatin-loaded micelles (PD-MC) could be released in fibrotic liver tissue after entering acidic lysosomal compartments. Due to the ROS elimination property of micelles and antioxidant property of polydatin, PD-MC suppressed TLR4/NF-κB p65 signaling in macrophages, reducing NOX-4 activity in HSCs, and finally alleviating liver fibrosis through inhibition of inflammation and oxidative stress. In addition, combination of imaging capability provides a novel prospect in the diagnosis and treatment of liver injury. To achieve effective treatment of liver injury along with further analysis of the condition through ultrasound imaging, Lee and co-workers attempted to construct polymer nanoparticles with ultrasound imaging capacity [[245], [246], [247], [248], [249]]. More recently, the group designed a ketalized maltodextrin nanocarrier with silymarin (KMD NP) that could generate CO2 bubbles through disrupting carbonate bonds in acidic sites [258]. KMD NPs were formed by conjugating acid-cleavable hydrophobic moieties to maltodextrin via carbonate bonds. The formation of CO2 bubbles at pH acid sites endowed KMD NPs with echogenicity, so that ultrasound imaging could be performed (Fig. 13F). KMD NPs significantly enhanced ultrasound contrast in drug-injured liver and ameliorated hepatic damage by inhibiting ROS and expression of TNF-α [258].
7. Anti-inflammatory nanodrugs
Reduction of inflammation can effectively decrease oxidative stress damage by disrupting the vicious circle of RONS and inflammation in liver injury. Currently, anti-inflammatory drugs are widely adopted in the treatment of several diseases. However, these drugs are almost ineffective and may even aggravate liver damage, since they are metabolized by liver and cause further damage to hepatocytes. For example, non-steroidal anti-inflammatory drugs cause acute liver injury of varying severity [17]. Moreover, corticosteroid anti-inflammatory drugs have limited efficacy for liver injury. The survival rate of groups with drug-induced acute liver failure treated with corticosteroid anti-inflammatory drugs was not significantly different to that of non-treated groups (69% vs 66%) [267].
Compared with traditional anti-inflammatory drugs, nanodrugs provide a better and safer therapeutic option to suppress inflammation The current anti-inflammatory nanodrugs for liver injury mainly include the following four categories: (1) nanodrugs reduce inflammation in liver injury by blocking pattern recognition receptor (PPR) pathways, (2) nanocarriers with nucleic acid reduce the expression of inflammatory factors, or (3) increase the expression of anti-inflammatory factors, and (4) nanoscale exosomes and stem cells reduce inflammation at the site of liver injury.
7.1. Blockage of PRR pathways
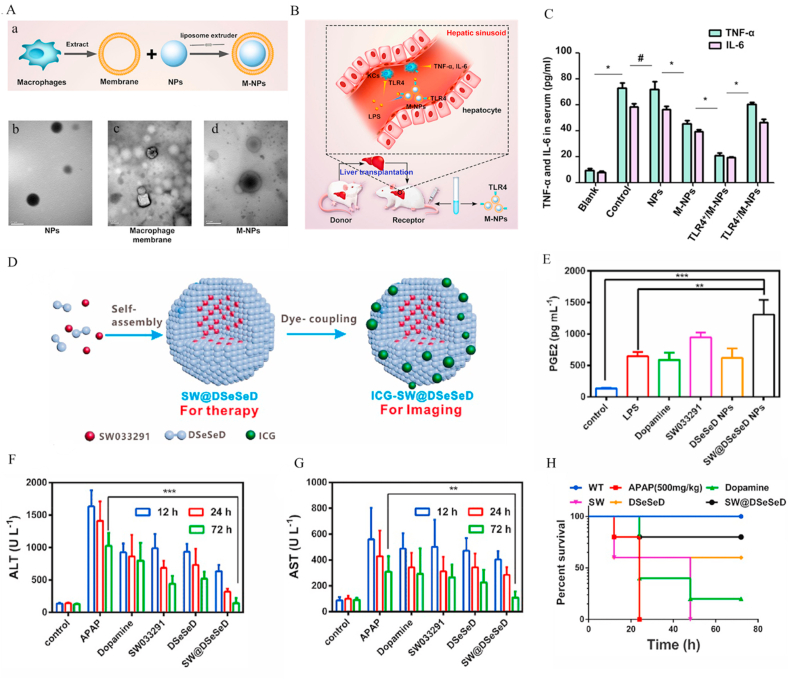
Blockage of the PPR pathway is an effective method to reduce inflammation in liver injury. Recently, Ou and co-workers developed biomimetic nanoparticles (M-NPs) by coating the surface of polylactic acid nanoparticles with the membrane of KCs to reduce hepatic IRI [268] (Fig. 14A). Cell membranes on the M − NP surface contained several TLR4 receptors that could bind DAMPs and PAMPs (Fig. 14B). Consequently, M-NPS competed with KCs to interact with PAMPs and DAMPs, which inhibited activation of the inflammatory pathways of KCs and reduced levels of inflammatory factors. In a rat hepatic IRI model, M-NPs significantly reduced the lipopolysaccharide concentration (LPS) in serum and suppressed activation of KCs via inhibiting the TLR4/MyD88/IRAK1/NF-κB signaling pathway along with reducing the levels of inflammatory factors TNF-α and IL-6 (Fig. 14C).
Fig. 14.
Anti-inflammatory nanodrugs for liver injury exert their effects through blocking PPR pathways. (A) (a) Schematic illustration of M − NP formulation. TEM images of (b) NPs, (c) macrophage membrane and (d) M-NPs. (Scale bar: 200 nm) (B) Schematic mechanisms of M − NP action in alleviation of hepatic IRI caused by liver transplantation. (C) ELISA analysis of TNF-α and IL-6 in serum. Reprinted with permission from Ref. [268] (D) Schematic synthesis of SW@DSeSeD. (E) PGE2 levels in LPS-stimulated RAW-264.7 cells after 20 h incubation with different formulations. (F, G) ELISA assay of serum ALT and AST levels in WT (control) and treatment groups at 12 h, 24 h and 72 h. (H) Survival curves of mice subjected to different treatments after injection of a lethal dose of APAP (500 mg kg−1). Reprinted with permission from Ref. [270].
Blockage of the NLR pathway can also effectively reduce inflammation at the site of liver injury. NLRP3 belongs to the NLR subfamily of PRRs. NLRP3, adaptor (ASC protein), and effector (caspase-1) are combined to form the NLRP3 inflammasome [269]. DAMPs and PAMPs activate the NLRP3 inflammasome and trigger inflammation in the liver injury site. Dopamine has recently been reported to inhibit NLRP3 inflammasomes. However, dopamine has a short half-life in the body and its direct use in the treatment of liver injury is difficult. Recently, Wu et al. [270] prepared a new type of dopamine-based nanodrug (SW@DSeSeD) for drug-induced acute liver injury. SW@DSeSeD was formed by self-assembly of a disselenium molecule with dopamine at both ends and prostaglandin degrading enzyme 2 (PGE2) inhibitor (SW033291) in aqueous solution (Fig. 14D). In the presence of RONS, the Se–Se bond was easily disrupted. SW033291 could induce rapid tissue regeneration by inhibiting prostaglandin degrading enzyme activity. SW@DSeSeD effectively lysed and released active ingredients (dopamine and SW033291) within the high RONS-containing environment of liver injury, significantly inhibited the NLRP3 inflammasomes to attenuate release of pro-inflammatory cytokines, such as IL-1β and IL-18, and promoted regeneration by increasing the level of PGE2 (Fig. 14E). After SW@DSeSeD treatment, all mice survived and their liver functions returned to normal (Fig. 14F–H). All untreated mice died owing to liver failure. These results support the excellent therapeutic effects of SW@DSeSeD against liver injury.
7.2. Reduction of the expression of inflammatory factors
Delivery of siRNA to inflammatory cells presents an effective strategy for achieving anti-inflammatory effects in liver injury by interfering with the expression of inflammatory factors [[271], [272], [273]]. siRNAs are difficult to transport through the cell membrane and commonly degraded by nucleases, poor targeting, and immunogenicity in vivo [274]. Nanocarriers containing siRNAs can effectively protect against rapid enzymatic degradation in vivo, reduce immunogenicity, and increase blood retention time and cellular uptake [275].
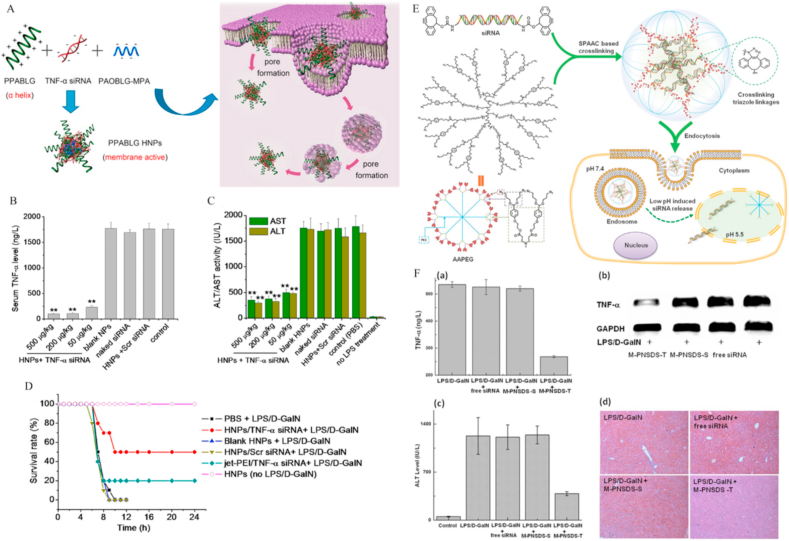
Positively charged polypeptides, cationic polymers and so on [276,277] have been developed as nanocarriers of siRNAs (polyanions), with the aim of reducing the TNF-α level at the site of liver injury. TNF-α is a critical pro-inflammatory factor and liver injury can be effectively alleviated through suppression of its expression in inflammatory cells. In a previous study, Yin et al. [278] adopted the α-helical structures PPABLG and PAOBLG-MPA as nanocarriers of TNF-α siRNA. PPABLG was a cell-penetrating peptide with high molecular weight and high cationic charge density (Fig. 15A). Notably, the α-helical structure of PPABLG facilitated vigorous transmembrane activity and effective delivery of siRNA from lysosomes to the cytoplasm. In addition, interactions between cationic polymers and RNA could be enhanced by introducing the polyanionic polypeptide, PAOBLG-MPA, which promoted TNF-α siRNA encapsulation in nanocarriers. PPABLG hybrid nanoparticles (HNP) protected against LPS/d-galactosamine (d-GalN)- and sepsis-induced liver injury in mice and improved survival rates via downregulation of TNF-α (Fig. 15B–D). Recently, Tang and co-workers developed a pH-sensitive mannose nanocarrier with no positive charge (M-PNSDS-T NP) to improve delivery of TNF-α siRNA to the cytoplasm [279]. M-PNSDS-T NPs were cross-linked with alkyne-functionalized siRNA and azide-modified multi-arm PEG through hydrophobic unstable acetal bonds. Upon endocytosis of M-PNSDS-T NPs from the extracellular region to the lysosome, the pH value of the environment dropped from 7.4 to ∼4.5. Acetal bonds in M-PNSDS-T NPs were stable under physiological conditions but disrupted under acidic conditions (pH 4.5), leading to release of TNF-α siRNA into the cytoplasm (Fig. 15E). Moreover, modification of mannose moieties facilitated targeting of macrophages and minimized aggregation with serum proteins. M-PNSDS-T NPs could selectively deliver TNF-α siRNA to liver macrophages for treatment of acute liver failure induced by LPS and d-GalN, which protected against injury to a significant extent (Fig. 15F). As for liver cirrhosis, HMGB1 protein has gradually become a research hotspot as pro-inflammatory factor and fibroblast chemokine. Zhang et al. constructed an HMGB1-siRNA-loaded stable nucleic acid lipid nanoparticle that were modified with pPB peptide (HMGB1-siRNA@SNALP-pB) to target HSCs, which indicated dual antifibrotic and anti-inflammatory effects in TAA- and CCl4-induced liver cirrhosis mouse model. More importantly, the results revealed that the therapeutic effect of HMGB1-siRNA@SNALP-pB were superior to that of HSP47-siRNA@SNALP-pPB with only antifibrotic activity, which provided significant insights toward the treatment of hepatic cirrhosis [280].
Fig. 15.
Anti-inflammatory nanodrugs for liver injury act by reducing the expression of inflammatory factors. (A) Schematic illustration of PPABLG/PAOBLG-MPA/siRNA HNP fabrication and intracellular delivery. (B) Serum levels of TNF-α in LPS/d-GalN-treated mice receiving i. v. administration of HNPs. LPS/d-GalN was i. p.-injected 24 h after administration. (C) Serum ALT and AST levels were measured 5 h after LPS/d-GalN stimulation. (D) Survival of mice in different treatment groups. Reprinted with permission from Ref. [279] (E) Schematic illustration of siRNA cross-linked nanoparticles (PNSDS). (F) M-PNSDS-T protects against liver injury in LPS/d-GalN-treated mice via suppressing TNF-α expression. Serum TNF-α levels in mice administered siRNA (50 μg kg−1). (a). Relative TNF-α mRNA levels in liver (b). ALT levels in mice 6 h after LPS/d-GalN treatment (c). H&E staining of mouse liver 6 h post LPS/d-GalN treatment (d). Reprinted with permission from Ref. [285].
Synthetic decoy oligodeoxynucleotides can bind specific transcription factors based on mimicking DNA complementary sequences [281]. These oligodeoxynucleotides show high affinity for their respective targets and hinder gene transcription upon transfection into target cells [282]. As a result, NF-κB decoys prevent initiation of related gene expression by entrapping NF-κB, which translocates to the nucleus and binds cis-elements of target genes, including inflammation-related genes, such as TNF-α and IL-6 [283]. Recently, NF-κB decoy with no significant immunogenic effects has been effectively utilized as an alternative treatment for inflammation-related liver injury. Due to the low rate of uptake of naked NF-κB decoy by cells, several researchers have explored the use of nanocarriers [284]. For instance, Hashida et al. [285] developed fucosylated cationic liposomes (Fuc-NPs) for loading NF-κB decoy while dioleoylphosphatidy-lethanolamine (DOPE) was used to protect NF-κB from endosomal activity. Fucosylated proteins acquired targeting through fucose receptors on KCs. The Fuc-NP surface had a positive charge, which achieved better selective transport of NF-κB decoy by KCs. Fuc-NPs induced a significant decrease in serum levels of TNF-α, IFN, ALT, and AST in LPS-treated mice [266].
7.3. Increased expression of anti-inflammatory factors
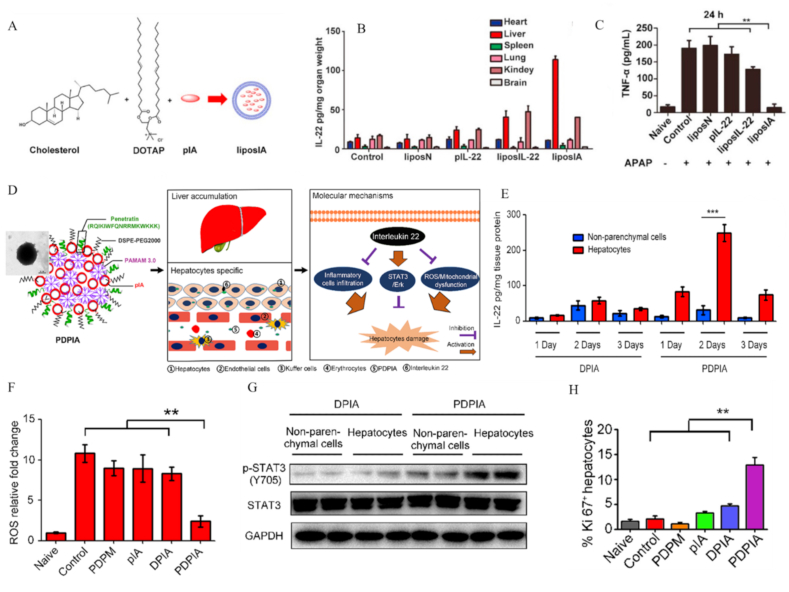
Anti-inflammatory factors exert significant antioxidant effects through inhibiting RONS production in hepatocytes. For example, interleukin-22 (IL-22), a member of the interleukin 10 family, has recently been shown to induce hepatocyte production of endogenous antioxidants and promote expression of anti-apoptotic factors in liver injury by activating STAT3 signaling [286]. However, IL-22-mediated treatment of liver injury can cause systemic side-effects, since IL-22 receptors are distributed throughout the body. Coupling of IL-22 to liver-targeted proteins represents a very effective strategy to reduce systemic side-effects. Apolipoprotein A-I additionally has excellent liver targeting properties. IL-22 can be effectively delivered to hepatocytes by coupling with apolipoprotein A-I. Ju and co-workers constructed cationic liposomes (liposIA NPs) composed of DOTAP and cholesterol as gene expression vectors for delivery of interleukin-22/ApoA-I fusion protein-expressing plasmid (Fig. 16A). Upon transfection of liposIA NPs, IL-22/ApoA-I fusion protein was expressed in vivo [287], which effectively accumulated in liver (Fig. 16B). After liposIA NP treatment in DILI, levels of pro-inflammatory factors (such as TNF-α) were markedly reduced and liver function restored (Fig. 16C).
Fig. 16.
Anti-inflammatory nanodrugs for liver injury that exert effects via promoting the expression of anti-inflammatory factors. (A) Schematic illustration of liposIA NPs. (B) ELISA analysis of IL-22 in selected organs (heart, liver, lung, spleen, kidney and brain) 2 days after liposIA administration. (C) Serum levels of TNF-α in mice subjected to APAP treatment. Various drugs were intramuscularly injected, followed by intraperitoneal injection of APAP after 48 h. Reprinted with permission from Ref. [287] (D) Schematic representation of hepatocellular expression of interleukin-22 platform against hepatitis. (E) ELISA analysis of IL-22 in hepatocytes and non-parenchymal cells. (F) Quantitative analysis of ROS in liver sections among different groups. (G) Western blot analysis of p-STAT3 and STAT3 in hepatocytes and non-parenchymal cells. (H) PDPIA increased hepatocyte regeneration, as indicated by the percentage of Ki-67-positive hepatocytes. Reprinted with permission from Ref. [288].
Specific delivery of IL-22 plasmid nanocarriers to hepatocytes presents another effective strategy. These nanocarriers are generally prepared by self-assembly of cationic polymers or proteins and IL-22 plasmid. Chen and co-workers prepared a composite nanocarrier (PDPIA NPs) composed of cationic polypeptide penetration protein (penetratin), PEGylated lipid (DSPE-PEG2000), and poly (amidoamine) dendrimer for delivery of IL-22 plasmids to liver [288] (Fig. 16D). PDPIA NPs effectively accumulated in hepatocytes instead of KCs (Fig. 16E), inhibited generation of RONS and promoted hepatocyte regeneration through activated STAT3/Erk signal transduction in the severe ConA-induced hepatitis model (Fig. 16F–H).
MMP9 is the largest member of MMPs well known for degradation and remodeling of the extracellular matrix proteins. The anti-inflammatory effect of MMP9 has drawn attention of researchers [289]. Melgar-Lesmes et al. delivered a plasmid expressing MMP 9 (pMMP9) to inflammatory macrophages in cirrhotic livers through constructing a nanocarrier based on graphene nanostars (GNS) linked to PAMAM-G5 dendrimer (DGNS) [290]. In such ingenious design, the addition of CD11b promoter (TNF-α is a CD11b promoter activator) as well as the binding of two biocompatible materials, GNS with PAMAM-G5 dendrimers, achieved well targeting of macrophages. The overexpression of MMP 9 inhabited inflammation by inducing the conversion of macrophages from M1 type to M2 type. M1 pro-inflammatory cells helped to clear the infection but promote liver injury in aseptic inflammation, while M2 anti-inflammatory cells have a repair phenotype that can promote the regression phase [291]. Eventually, pMMP9-DGNP alleviated liver fibrosis through heightened collagen degradation and inflammation inhibition.
In addition, immune cells play an irreplaceable role in the pathogenesis of liver injury and even cirrhosis, especially macrophages [292]. It can effectively control inflammation with siRNA, decoy technology, and delivering plasmid to knockdown pro-inflammatory factors or to overexpress anti-inflammatory cytokine in immune cells. Nanocarriers should be effectively improve the pharmacokinetics and targeting of these nucleic acid drugs. However, the use of nanodrugs to treat liver injury is an emerging field, and nanodrugs for targeting immune cells have not been reported. We believe more surprising target immune cell nanodrugs will have tremendous power in the field of liver injury with the deepening of research.
7.4. Extracellular vesicles and stem cells with anti-inflammatory properties
Extracellular vesicles are cell-derived membrane vesicles, including exosomes (50–100 nm) and microvesicles (about 50–500 nm, with the largest reaching 1 μm). Vesicles can transport various substances, such as proteins, lipids, and nucleic acids, leading to modification of transcription or translation in recipient cells. All cells secrete various types of extracellular vesicles. Specific cell-derived extracellular vesicles exert anti-inflammatory and antioxidant therapeutic effects in liver injury [293]. Compared with many artificially prepared nanoparticles, exosomes have excellent biocompatibility, supporting their utility in the treatment of liver injury [[294], [295], [296], [297], [298]]. Anti-inflammatory exosomes from different sources have been developed, such as serum extracellular vesicles (SEV) [299], stem cell-derived exosomes [300], plant-derived exosomes [301,302], honey-derived exosomes [303], and exosomes from shiitake mushrooms [304]. For example, Brigstock and co-workers [299] demonstrated that SEVs from healthy humans exert a therapeutic effect against liver fibrosis. SEVs were directly derived from circulating cell types and tissues (such as endothelial cells) or indirect contact systemic circulation (through interstitial fluid). Compared with SEV of fibrotic patients, that of healthy people contained higher levels of miR-34c, -151–3p, -483–5p, and -532–5p, and could significantly inhibit liver fibrosis by reducing inflammation (Fig. 17A&B). Moreover, inflammatory factors, such as IFN-γ, and infiltration of activated immune cells were significantly reduced by SEV treatment of healthy mice in a carbon tetrachloride (CCl4)-induced liver fibrosis model (Fig. 17C).
Fig. 17.
Extracellular vesicles and stem cells with anti-inflammatory functions for treatment of liver injury. (A) Differential expression of miRNAs (miRs) between EVN and Exo F (left panel). Ranking of differential miRs from highest (“H”) to lowest (“L) led to identification of miR-34c-3p, -151–3p, -483–5p, -532–5p, and −687 as the most highly differentially expressed between the groups (right panel). (B) RT-PCR validation of reduced expression of selected miRs in EVF versus EVN. (C) Anti-inflammatory effect of EVN determined using the cytokine multiplex assay for serum IL-4, IL-5, IL-6, and TNF-α. Reprinted with permission from Ref. [299] (D) Schematic representation of the mechanisms of nanozyme impregnation of mesenchymal stem cells (PB-MSC) for hepatic IRI. (E) Analysis of lipid peroxidation and inflammation in hepatic tissue homogenates via TBARS assay (a), serum concentrations of TNF-α (b) and IL-10 (c), and MPO activity (d). Reprinted with permission from Ref. [306].
In addition to exosomes, mesenchymal stem cells (MSCs) have recently been developed for treatment of liver injury [305]. MSCs release a variety of cytokines, such as hepatocyte growth factor (HGF), through paracrine mechanisms. HGF inhibits apoptosis, regulates inflammation, promotes angiogenesis, and reduces tissue fibrosis in liver. Despite these promising outcomes, several limitations are associated with the use of MSCs for treatment of liver injury. The high RONS content makes MSC survival at injury sites difficult, leading to failure to secrete relevant cytokines and loss of therapeutic effects. Recently, Tae et al. [306] combined MSCs with Prussian blue nanoparticles (PB) comprising a biocompatible nanozyme with multiple ROS-scavenging mimetic activity to protect against oxidative stress in liver with I/R injury (Fig. 17D). Following impregnation of MSCs with PB, RONS from the external environment was obviously decreased. Compared with injection of PBS, PB, and MSCs, MSCs with PB reduced pro-inflammatory factors and liver tissue necrosis to a greater extent and effectively restored liver function in hepatic IRI mice (Fig. 17E).
8. Conclusion
Starting with the mechanisms of liver injury from various sources, we have comprehensively summarized the key roles of RONS and inflammation and limitations of traditional anti-inflammatory and antioxidant drugs in the treatment of liver injury. Due to the unique structural features and functions of liver, the numerous antioxidant and anti-inflammatory drugs currently available are relatively ineffective for therapy. Based on these characteristics, nanodrugs specifically enriched in liver may achieve efficient targeted treatment of liver injury and therefore have significant potential to overcome the limitations of traditional anti-inflammatory and antioxidant drugs.
Although significant progress has been made in this emerging field, continued efforts are required to address the huge challenges in clinical translation. The application of nanodrugs for liver disease is an emerging field in the past few years, and only three nanodrugs have entered clinical trials (data from the USA National Library of Medicine database, https://clinicaltrials.gov/). (Table 2). In fact, those nanodrugs consist of approved active pharmaceutical ingredients (API) and simple liposomal or polymer carrier are easier to be translated into clinic in the field of liver disease [307]. Above all, nanocarriers of antioxidants or anti-inflammatory drugs mentioned above are of greater feasibility in clinical translation, such as BRNP synthesized by Kim [261] and co-workers and TNF-α siRNA carried nanodrugs fabricated by Yin et al. [278]. The road ahead is full of thorns, but it is a considerable deal if nanodrug harnessing RONS and inflammation achieve success in the control of liver injury, which further promote the progress of most nanodrugs. The success of some nanodrugs has set a precedent [155], and four main challenges should be considered seriously learned from previous experiences [308,309] (Fig. 18), which is benefit for the meaningful clinical translation of nanodrug harnessing RONS and inflammation for liver disease.
Table 2.
Antioxidant and anti-inflammatory nanodrug clinical trials on liver disease treatment. Data source: https://www.clinicaltrials. gov.
| Study title | Nanodrug | Liver Disease | Starting Time | Status | Number of Participants | Identifier |
|---|---|---|---|---|---|---|
| The Effect of Curcumin on the Development of Prednisolone-induced Hepatic Insulin Resistance (CURPRED) | Curcumin Liposome | NAFLD | December 2019 | Not applicable Recruiting |
36 | NCT04315350 |
| Study to Evaluate the Safety, Tolerability, PDs, and Efficacy of CNP-104 in Subjects With Primary Biliary Cholangitis | CNP-104 (comprised of PDC-E2 peptide dispersed within a negatively charged polymer matrix of PLGA particles) | Primary Biliary Cholangitis | December 2021 | Phase 1, 2 Not yet recruiting |
40 | NCT05104853 |
| Phase 1b/2, Open Label, Repeat Dose, Dose Escalation Study of ND-L02-s0201 Injection in Subjects With Moderate to Extensive Fibrosis (METAVIR F3-4) | ND-L02-s0201 (A Vitamin A-coupled Lipid Nanoparticle Containing siRNA Against HSP47) | Hepatic Fibrosis | October 2014 | Phase 1 Completed |
25 | NCT02227459 |
Fig. 18.
Challenges in nanodrug translation of liver disease.
Firstly, toxicity of nanodrugs is the biggest obstacle that inhibit their application translating to clinic. Nanodrugs that harnessing RONS and inflammation for liver disease are no exception. Most materials will be more active once scaled down to nano-regime due to the dramatic increase in total surface area [310]. The altered biological activity possesses serious potential toxicities towards cells, tissues and organs in the body [311]. The long-term side effects of nanodrugs should be further established. The successful clinical translation of some nanodrug provide an excellent example for this challenge. For instance, it is very important to continuously optimize nanoparticles for addressing problems in preclinical experiments throughout the story of Onpattro [155].
Secondly, the mechanism of action and metabolism of nanodrugs in liver requires further research. Many nanodrugs have not yet entered clinical trials due to lack of data on their behavioral mechanisms in vivo. The studies on behavioral mechanisms of nanodrugs are thought to contribute to the optimization of drug efficacy on one hand and the prediction and reduction of nanotoxicity [46,47,312]. Given the significant role of liver played in metabolism, further studies of the influence of existing liver injury on the action and metabolism of the nanodrugs are needed. In addition, the effect of ROS scavengers under pathological conditions on its metabolism also requires further studies on behavioral mechanisms. Accumulating studies have focused on the mechanisms of nanomaterials, including their transformation and therapeutic actions in the body. For instance, Chen and co-workers clarified the whole process of nanomaterial transport-biotransformation-bioutilization and fate in the body with the aid of advanced technologies, such as high sensitivity, high resolution in situ characterization, proteomics, and molecular dynamics simulation [313]. Cai et al. [314] established a multi-dimensional method for evaluation of nanobiological effects using protein metabonomic technology. The collective studies provide a good reference for further research on the basic principles of nanodrug behavior in vivo.
Thirdly, the limitation of preclinical animal model results in the difference of nanodrugs efficacy. The large difference in physiological and pathological settings of the injured area caused by species divergence between humans and animals is difficult to avoid. And some animal models cannot manifest the clinical progress of various liver disease [315]. In addition, rodents’ models of alcoholic liver disease are unlikely to develop through drinking alone due to the rapid ethanol metabolism and short-term feeding period. Many of the symptoms observed in severe alcoholic liver disease are induced by secondary effects of severe injury and inflammation, rather than the main effects of alcohol-induced hepatoxicity [316].
Finally, the complex, inefficient, and costly synthesis process provides a big challenge for nanodrugs translate from laboratory to large-scale manufacturing. First of all, many elaborately fabricated nanodrugs integrate different functional species or components into a single nano-system, instead of using a primitive nanomaterial [266,317]. Then, complex, inefficient, and costly synthesis of nanoparticles is proposed, which is difficult to implement in large-scale manufacturing. At the same time, the addition of uncertain new components will also make the activity of nanodrug in the body more complicated or increase the risk of potential toxicity [318]. In summary, the complexity of the components makes chemistry, manufacturing, and quality control more comprehensive and stricter. In addition, chemistry, manufacturing, and quality control is hard to be guaranteed due to lack of standards and norms. Therefore, the key quality attribute should be taken into account in the early stage of formulation design. And the production process also needs to be conducted in strict accordance with Good Manufacturing Practice (GMP) guidelines. It is necessary to carefully evaluate the ingredients, excipients and key quality attributes as early as possible in the design of nanodrug [307,319].
Notwithstanding, the development of nanodrugs provides a promising outlook for treating liver injury compared with conventional drug development. It is reported that the estimated total success rate of nanodrugs against cancer from the first stage to clinical approval is 6%, which is much higher than the 3.4% of conventional new drugs [320]. Notably, it can't be ignored many of those approved nanodrugs are based on the FDA approved API instead of a completely new chemical entity. Nanodrugs achieved the superior performance for optimized pharmacokinetics and/or pharmaceutical formulations when compared with conventional new drugs development. But nanodrug developments largely benefit from conventional new drugs developments as well. As early as 1965, liposomes were proposed for drug delivering [321]. And the success of liposomal nanodrugs were evident in the clinical application in the past 40 plus years. The annual sales AmBisome® and Doxil® have both reached hundreds of millions of dollars [322]. Thereby, nanodrugs based on regulation of RONS and inflammation have extended the frontier of research on treatment of liver injury from various sources and been expected to replace large part of the traditional drugs when further studies on toxicity, metabolism in the body, animal model and manufacturing are warranted. Nanodrug treatment is expected to significantly reduce liver injury and prevent progression to liver failure, fibrosis, and cancer, thus paving the way for reducing mortality from liver disease.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (No.21974134, 81974508, 81770739, 81673492), the Hunan Science Fund for Distinguished Young Scholar of China (No. 2021JJ10067), Innovation-Driven Project of Central South University (No. 202045005), Hunan Provincial Natural Science Foundation of China (No. 2021JJ31066), Changsha Science and Technology Project (No. kq2001048), and Key Research Project of Ningxia Hui Autonomous Region in 2021 of China (Major Project) (2021BEG01001).
Contributor Information
Zhicheng Gong, Email: gongzhicheng@csu.edu.cn.
Kelong Ai, Email: aikelong@csu.edu.cn.
References
- 1.Marjot T., Webb G.J., Barritt ASt, Moon A.M., Stamataki Z., Wong V.W., et al. COVID-19 and liver disease: mechanistic and clinical perspectives. Nat. Rev. Gastroenterol. Hepatol. 2021;18:348–364. doi: 10.1038/s41575-021-00426-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jothimani D., Venugopal R., Abedin M.F., Kaliamoorthy I., Rela M. COVID-19 and the liver. J. Hepatol. 2020;73:1231–1240. doi: 10.1016/j.jhep.2020.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Phipps M.M., Barraza L.H., LaSota E.D., Sobieszczyk M.E., Pereira M.R., Zheng E.X., et al. Acute liver injury in COVID-19: prevalence and association with clinical outcomes in a large U.S. Cohort. Hepatology. 2020;72:807–817. doi: 10.1002/hep.31404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kubes P., Jenne C. Immune responses in the liver. Annu. Rev. Immunol. 2018;36:247–277. doi: 10.1146/annurev-immunol-051116-052415. [DOI] [PubMed] [Google Scholar]
- 5.Trefts E., Gannon M., Wasserman D.H. The liver. Curr. Biol. 2017;27:R1147–R1151. doi: 10.1016/j.cub.2017.09.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wei Y., Wang Y.G., Jia Y., Li L., Yoon J., Zhang S., et al. Liver homeostasis is maintained by midlobular zone 2 hepatocytes. Science. 2021:371. doi: 10.1126/science.abb1625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gola A., Dorrington M.G., Speranza E., Sala C., Shih R.M., Radtke A.J., et al. Commensal-driven immune zonation of the liver promotes host defence. Nature. 2021;589:131–136. doi: 10.1038/s41586-020-2977-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Abdelmalek M.F. Nonalcoholic fatty liver disease: another leap forward. Nat. Rev. Gastroenterol. Hepatol. 2021;18:85–86. doi: 10.1038/s41575-020-00406-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pawlotsky J.M. COVID-19 and the liver-related deaths to come. Nat. Rev. Gastroenterol. Hepatol. 2020;17:523–525. doi: 10.1038/s41575-020-0328-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Weaver R.J., Blomme E.A., Chadwick A.E., Copple I.M., Gerets H.H.J., Goldring C.E., et al. Managing the challenge of drug-induced liver injury: a roadmap for the development and deployment of preclinical predictive models. Nat. Rev. Drug Discov. 2020;19:131–148. doi: 10.1038/s41573-019-0048-x. [DOI] [PubMed] [Google Scholar]
- 11.Huang X., Lee F., Teng Y., Lingam C.B., Chen Z., Sun M., et al. Sequential drug delivery for liver diseases. Adv. Drug Deliv. Rev. 2019;149–150:72–84. doi: 10.1016/j.addr.2019.11.001. [DOI] [PubMed] [Google Scholar]
- 12.Asrani S.K., Devarbhavi H., Eaton J., Kamath P.S. Burden of liver diseases in the world. J. Hepatol. 2019;70:151–171. doi: 10.1016/j.jhep.2018.09.014. [DOI] [PubMed] [Google Scholar]
- 13.Singal A.K., Mathurin P. Diagnosis and treatment of alcohol-associated liver disease: a review. JAMA. 2021;326:165–176. doi: 10.1001/jama.2021.7683. [DOI] [PubMed] [Google Scholar]
- 14.Hoofnagle J.H., Bjornsson E.S. Drug-induced liver injury - types and phenotypes. N. Engl. J. Med. 2019;381:264–273. doi: 10.1056/NEJMra1816149. [DOI] [PubMed] [Google Scholar]
- 15.Han H., Desert R., Das S., Song Z., Athavale D., Ge X., et al. Danger signals in liver injury and restoration of homeostasis. J. Hepatol. 2020;73:933–951. doi: 10.1016/j.jhep.2020.04.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mouzannar K., Liang T.J. Hepatitis B virus - recent therapeutic advances and challenges to cure. J. Hepatol. 2020;73:694–695. doi: 10.1016/j.jhep.2020.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.European Association for the Study of the Liver Electronic address eee, clinical Practice guideline panel C, panel m, representative EGB. EASL clinical Practice guidelines: drug-induced liver injury. J. Hepatol. 2019;70:1222–1261. [Google Scholar]
- 18.Stefan N., Haring H.U., Cusi K. Non-alcoholic fatty liver disease: causes, diagnosis, cardiometabolic consequences, and treatment strategies. Lancet Diabetes Endocrinol. 2019;7:313–324. doi: 10.1016/S2213-8587(18)30154-2. [DOI] [PubMed] [Google Scholar]
- 19.Singal A.K., Bataller R., Ahn J., Kamath P.S., Shah V.H. ACG clinical guideline: alcoholic liver disease. Am. J. Gastroenterol. 2018;113:175–194. doi: 10.1038/ajg.2017.469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ma L., Craig A.J., Heinrich S. Hypoxia is a key regulator in liver cancer progression. J. Hepatol. 2021;73:736–737. doi: 10.1016/j.jhep.2021.05.032. [DOI] [PubMed] [Google Scholar]
- 21.Gustot T., Stadlbauer V., Laleman W., Alessandria C., Thursz M. Transition to decompensation and acute-on-chronic liver failure: role of predisposing factors and precipitating events. J. Hepatol. 2021;75(Suppl 1):S36–S48. doi: 10.1016/j.jhep.2020.12.005. [DOI] [PubMed] [Google Scholar]
- 22.Llovet J.M., Kelley R.K., Villanueva A., Singal A.G., Pikarsky E., Roayaie S., et al. Hepatocellular carcinoma. Nat. Rev. Dis. Prim. 2021;7:6. doi: 10.1038/s41572-020-00240-3. [DOI] [PubMed] [Google Scholar]
- 23.Burra P., Samuel D., Sundaram V., Duvoux C., Petrowsky H., Terrault N., et al. Limitations of current liver donor allocation systems and the impact of newer indications for liver transplantation. J. Hepatol. 2021;75(Suppl 1):S178–S190. doi: 10.1016/j.jhep.2021.01.007. [DOI] [PubMed] [Google Scholar]
- 24.Barnett R. Liver cirrhosis. Lancet. 2018;392:275. doi: 10.1016/S0140-6736(18)31659-3. [DOI] [PubMed] [Google Scholar]
- 25.Zhao Q., Liu J., Deng H., Ma R., Liao J.Y., Liang H., et al. Targeting mitochondria-located circRNA SCAR alleviates NASH via reducing mROS output. Cell. 2020;183:76–93 e22. doi: 10.1016/j.cell.2020.08.009. [DOI] [PubMed] [Google Scholar]
- 26.Mansouri A., Gattolliat C.H., Asselah T. Mitochondrial dysfunction and signaling in chronic liver diseases. Gastroenterology. 2018;155:629–647. doi: 10.1053/j.gastro.2018.06.083. [DOI] [PubMed] [Google Scholar]
- 27.Parola M., Pinzani M. Liver fibrosis: pathophysiology, pathogenetic targets and clinical issues. Mol. Aspect. Med. 2019;65:37–55. doi: 10.1016/j.mam.2018.09.002. [DOI] [PubMed] [Google Scholar]
- 28.Wang Z., Ying Z., Bosy-Westphal A., Zhang J., Schautz B., Later W., et al. Specific metabolic rates of major organs and tissues across adulthood: evaluation by mechanistic model of resting energy expenditure. Am. J. Clin. Nutr. 2010;92:1369–1377. doi: 10.3945/ajcn.2010.29885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Boussif A., Rolas L., Weiss E., Bouriche H., Moreau R., Perianin A. Impaired intracellular signaling, myeloperoxidase release and bactericidal activity of neutrophils from patients with alcoholic cirrhosis. J. Hepatol. 2016;64:1041–1048. doi: 10.1016/j.jhep.2015.12.005. [DOI] [PubMed] [Google Scholar]
- 30.Yan M., Huo Y., Yin S., Hu H. Mechanisms of acetaminophen-induced liver injury and its implications for therapeutic interventions. Redox Biol. 2018;17:274–283. doi: 10.1016/j.redox.2018.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gracia-Sancho J., Caparros E., Fernandez-Iglesias A., Frances R. Role of liver sinusoidal endothelial cells in liver diseases. Nat. Rev. Gastroenterol. Hepatol. 2021;18:411–431. doi: 10.1038/s41575-020-00411-3. [DOI] [PubMed] [Google Scholar]
- 32.Seitz H.K., Bataller R., Cortez-Pinto H., Gao B., Gual A., Lackner C., et al. Alcoholic liver disease. Nat. Rev. Dis. Prim. 2018;4:16. doi: 10.1038/s41572-018-0014-7. [DOI] [PubMed] [Google Scholar]
- 33.Bottger R., Pauli G., Chao P.H., Al Fayez N., Hohenwarter L., Li S.D. Lipid-based nanoparticle technologies for liver targeting. Adv. Drug Deliv. Rev. 2020;154–155:79–101. doi: 10.1016/j.addr.2020.06.017. [DOI] [PubMed] [Google Scholar]
- 34.Maqsoudlou A., Assadpour E., Mohebodini H., Jafari S.M. Improving the efficiency of natural antioxidant compounds via different nanocarriers. Adv. Colloid Interface Sci. 2020;278 doi: 10.1016/j.cis.2020.102122. [DOI] [PubMed] [Google Scholar]
- 35.Sanyal A.J., Chalasani N., Kowdley K.V., McCullough A., Diehl A.M., Bass N.M., et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N. Engl. J. Med. 2010;362:1675–1685. doi: 10.1056/NEJMoa0907929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sang Y., Cao F., Li W., Zhang L., You Y., Deng Q., et al. Bioinspired construction of a nanozyme-based H2O2 homeostasis disruptor for intensive chemodynamic therapy. J. Am. Chem. Soc. 2020;142:5177–5183. doi: 10.1021/jacs.9b12873. [DOI] [PubMed] [Google Scholar]
- 37.Cao F., Zhang L., You Y., Zheng L., Ren J., Qu X. An enzyme-mimicking single-atom catalyst as an efficient multiple reactive oxygen and nitrogen species scavenger for sepsis management. Angew Chem. Int. Ed. Engl. 2020;59:5108–5115. doi: 10.1002/anie.201912182. [DOI] [PubMed] [Google Scholar]
- 38.Kim J., Kim H.Y., Song S.Y., Go S.H., Sohn H.S., Baik S., et al. Synergistic oxygen generation and reactive oxygen species scavenging by manganese ferrite/ceria Co-decorated nanoparticles for rheumatoid arthritis treatment. ACS Nano. 2019;13:3206–3217. doi: 10.1021/acsnano.8b08785. [DOI] [PubMed] [Google Scholar]
- 39.Lee Y., Sugihara K., Gillilland M.G., 3rd, Jon S., Kamada N., Moon J.J. Hyaluronic acid-bilirubin nanomedicine for targeted modulation of dysregulated intestinal barrier, microbiome and immune responses in colitis. Nat. Mater. 2020;19:118–126. doi: 10.1038/s41563-019-0462-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Zhang M., Zhu J., Qin X., Zhou M., Zhang X., Gao Y., et al. Cardioprotection of tetrahedral DNA nanostructures in myocardial ischemia-reperfusion injury. ACS Appl. Mater. Interfaces. 2019;11:30631–30639. doi: 10.1021/acsami.9b10645. [DOI] [PubMed] [Google Scholar]
- 41.Zhao T., Wu W., Sui L., Huang Q., Nan Y., Liu J., et al. Reactive oxygen species-based nanomaterials for the treatment of myocardial ischemia reperfusion injuries. Bioact. Mater. 2021;7:47–72. doi: 10.1016/j.bioactmat.2021.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Chen L., Huang Q., Zhao T., Sui L., Wang S., Xiao Z., et al. Nanotherapies for sepsis by regulating inflammatory signals and reactive oxygen and nitrogen species: new insight for treating COVID-19. Redox Biol. 2021;45 doi: 10.1016/j.redox.2021.102046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jin Y., Wang H., Yi K., Lv S., Hu H., Li M., et al. Applications of nanobiomaterials in the therapy and imaging of acute liver failure. Nano-Micro Lett. 2020;13:25. doi: 10.1007/s40820-020-00550-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Casals G., Perramon M., Casals E., Portoles I., Fernandez-Varo G., Morales-Ruiz M., et al. Cerium oxide nanoparticles: a new therapeutic tool in liver diseases. Antioxidants. 2021;10 doi: 10.3390/antiox10050660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Guan Y., Yao W., Yi K., Zheng C., Lv S., Tao Y., et al. Nanotheranostics for the management of hepatic ischemia-reperfusion injury. Small. 2021;17 doi: 10.1002/smll.202007727. [DOI] [PubMed] [Google Scholar]
- 46.Cornu R., Beduneau A., Martin H. Influence of nanoparticles on liver tissue and hepatic functions: a review. Toxicology. 2020;430 doi: 10.1016/j.tox.2019.152344. [DOI] [PubMed] [Google Scholar]
- 47.Li J., Chen C., Xia T. Understanding nanomaterial-liver interactions to facilitate the development of safer nanoapplications. Adv. Mater. 2022 doi: 10.1002/adma.202106456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sies H., Jones D.P. Reactive oxygen species (ROS) as pleiotropic physiological signalling agents. Nat. Rev. Mol. Cell Biol. 2020;21:363–383. doi: 10.1038/s41580-020-0230-3. [DOI] [PubMed] [Google Scholar]
- 49.Zorov D.B., Juhaszova M., Sollott S.J. Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol. Rev. 2014;94:909–950. doi: 10.1152/physrev.00026.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.An P., Wei L.L., Zhao S., Sverdlov D.Y., Vaid K.A., Miyamoto M., et al. Hepatocyte mitochondria-derived danger signals directly activate hepatic stellate cells and drive progression of liver fibrosis. Nat. Commun. 2020;11:2362. doi: 10.1038/s41467-020-16092-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Wiesner R.J., Ruegg J.C., Morano I. Counting target molecules by exponential polymerase chain reaction: copy number of mitochondrial DNA in rat tissues. Biochem. Biophys. Res. Commun. 1992;183:553–559. doi: 10.1016/0006-291x(92)90517-o. [DOI] [PubMed] [Google Scholar]
- 52.Di Ciaula A., Passarella S., Shanmugam H., Noviello M., Bonfrate L., Wang D.Q., et al. Nonalcoholic fatty liver disease (NAFLD). Mitochondria as players and targets of therapies? Int. J. Mol. Sci. 2021;22 doi: 10.3390/ijms22105375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hernansanz-Agustin P., Choya-Foces C., Carregal-Romero S., Ramos E., Oliva T., Villa-Pina T., et al. Na(+) controls hypoxic signalling by the mitochondrial respiratory chain. Nature. 2020;586:287–291. doi: 10.1038/s41586-020-2551-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Barbier-Torres L., Fortner K.A., Iruzubieta P., Delgado T.C., Giddings E., Chen Y., et al. Silencing hepatic MCJ attenuates non-alcoholic fatty liver disease (NAFLD) by increasing mitochondrial fatty acid oxidation. Nat. Commun. 2020;11:3360. doi: 10.1038/s41467-020-16991-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Brenner C., Galluzzi L., Kepp O., Kroemer G. Decoding cell death signals in liver inflammation. J. Hepatol. 2013;59:583–594. doi: 10.1016/j.jhep.2013.03.033. [DOI] [PubMed] [Google Scholar]
- 56.Fisicaro P., Barili V., Montanini B., Acerbi G., Ferracin M., Guerrieri F., et al. Targeting mitochondrial dysfunction can restore antiviral activity of exhausted HBV-specific CD8 T cells in chronic hepatitis B. Nat. Med. 2017;23:327–336. doi: 10.1038/nm.4275. [DOI] [PubMed] [Google Scholar]
- 57.Pond S.M., Tozer T.N. First-pass elimination. Basic concepts and clinical consequences. Clin. Pharmacokinet. 1984;9:1–25. doi: 10.2165/00003088-198409010-00001. [DOI] [PubMed] [Google Scholar]
- 58.Cederbaum A.I. Alcohol metabolism. Clin. Liver Dis. 2012;16:667–685. doi: 10.1016/j.cld.2012.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Shen D.D., Kunze K.L., Thummel K.E. Enzyme-catalyzed processes of first-pass hepatic and intestinal drug extraction. Adv. Drug Deliv. Rev. 1997;27:99–127. doi: 10.1016/s0169-409x(97)00039-2. [DOI] [PubMed] [Google Scholar]
- 60.Niemela O., Parkkila S., Juvonen R.O., Viitala K., Gelboin H.V., Pasanen M. Cytochromes P450 2A6, 2E1, and 3A and production of protein-aldehyde adducts in the liver of patients with alcoholic and non-alcoholic liver diseases. J. Hepatol. 2000;33:893–901. doi: 10.1016/s0168-8278(00)80120-8. [DOI] [PubMed] [Google Scholar]
- 61.Leung T.M., Nieto N. CYP2E1 and oxidant stress in alcoholic and non-alcoholic fatty liver disease. J. Hepatol. 2013;58:395–398. doi: 10.1016/j.jhep.2012.08.018. [DOI] [PubMed] [Google Scholar]
- 62.Kloss M.W., Rosen G.M., Rauckman E.J. N-demethylation of cocaine to norcocaine. Evidence for participation by cytochrome P-450 and FAD-containing monooxygenase. Mol. Pharmacol. 1983;23:482–485. [PubMed] [Google Scholar]
- 63.Zangar R.C., Davydov D.R., Verma S. Mechanisms that regulate production of reactive oxygen species by cytochrome P450. Toxicol. Appl. Pharmacol. 2004;199:316–331. doi: 10.1016/j.taap.2004.01.018. [DOI] [PubMed] [Google Scholar]
- 64.Vitcheva V. Cocaine toxicity and hepatic oxidative stress. Curr. Med. Chem. 2012;19:5677–5682. doi: 10.2174/092986712803988929. [DOI] [PubMed] [Google Scholar]
- 65.Derry P.J., Nilewski L.G., Sikkema W.K.A., Mendoza K., Jalilov A., Berka V., et al. Catalytic oxidation and reduction reactions of hydrophilic carbon clusters with NADH and cytochrome C: features of an electron transport nanozyme. Nanoscale. 2019;11:10791–10807. doi: 10.1039/c9nr00807a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Pawlak M., Lefebvre P., Staels B. Molecular mechanism of PPARalpha action and its impact on lipid metabolism, inflammation and fibrosis in non-alcoholic fatty liver disease. J. Hepatol. 2015;62:720–733. doi: 10.1016/j.jhep.2014.10.039. [DOI] [PubMed] [Google Scholar]
- 67.Bessone F., Razori M.V., Roma M.G. Molecular pathways of nonalcoholic fatty liver disease development and progression. Cell. Mol. Life Sci. 2019;76:99–128. doi: 10.1007/s00018-018-2947-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zhang L., Wang X., Cueto R., Effi C., Zhang Y., Tan H., et al. Biochemical basis and metabolic interplay of redox regulation. Redox Biol. 2019;26 doi: 10.1016/j.redox.2019.101284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Masubuchi Y., Suda C., Horie T. Involvement of mitochondrial permeability transition in acetaminophen-induced liver injury in mice. J. Hepatol. 2005;42:110–116. doi: 10.1016/j.jhep.2004.09.015. [DOI] [PubMed] [Google Scholar]
- 70.Win S., Than T.A., Min R.W., Aghajan M., Kaplowitz N. c-Jun N-terminal kinase mediates mouse liver injury through a novel Sab (SH3BP5)-dependent pathway leading to inactivation of intramitochondrial Src. Hepatology. 2016;63:1987–2003. doi: 10.1002/hep.28486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Levada K., Guldiken N., Zhang X., Vella G., Mo F.R., James L.P., et al. Hsp72 protects against liver injury via attenuation of hepatocellular death, oxidative stress, and JNK signaling. J. Hepatol. 2018;68:996–1005. doi: 10.1016/j.jhep.2018.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Dickson I. Improving hepatic ischaemia-reperfusion injury outcomes. Nat. Rev. Gastroenterol. Hepatol. 2019;16:583. doi: 10.1038/s41575-019-0194-y. [DOI] [PubMed] [Google Scholar]
- 73.Rao J., Cheng F., Zhou H., Yang W., Qiu J., Yang C., et al. Nogo-B is a key mediator of hepatic ischemia and reperfusion injury. Redox Biol. 2020;37 doi: 10.1016/j.redox.2020.101745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chouchani E.T., Pell V.R., James A.M., Work L.M., Saeb-Parsy K., Frezza C., et al. A unifying mechanism for mitochondrial superoxide production during ischemia-reperfusion injury. Cell Metabol. 2016;23:254–263. doi: 10.1016/j.cmet.2015.12.009. [DOI] [PubMed] [Google Scholar]
- 75.Milliken A.S., Kulkarni C.A., Brookes P.S. Acid enhancement of ROS generation by complex-I reverse electron transport is balanced by acid inhibition of complex-II: relevance for tissue reperfusion injury. Redox Biol. 2020;37 doi: 10.1016/j.redox.2020.101733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Woolbright B.L., Jaeschke H. Role of the inflammasome in acetaminophen-induced liver injury and acute liver failure. J. Hepatol. 2017;66:836–848. doi: 10.1016/j.jhep.2016.11.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Kazankov K., Jorgensen S.M.D., Thomsen K.L., Moller H.J., Vilstrup H., George J., et al. The role of macrophages in nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Nat. Rev. Gastroenterol. Hepatol. 2019;16:145–159. doi: 10.1038/s41575-018-0082-x. [DOI] [PubMed] [Google Scholar]
- 78.Gao B., Ahmad M.F., Nagy L.E., Tsukamoto H. Inflammatory pathways in alcoholic steatohepatitis. J. Hepatol. 2019;70:249–259. doi: 10.1016/j.jhep.2018.10.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Zhou R., Yazdi A.S., Menu P., Tschopp J. A role for mitochondria in NLRP3 inflammasome activation. Nature. 2011;469:221–225. doi: 10.1038/nature09663. [DOI] [PubMed] [Google Scholar]
- 80.Winterbourn C.C., Kettle A.J., Hampton M.B. Reactive oxygen species and neutrophil function. Annu. Rev. Biochem. 2016;85:765–792. doi: 10.1146/annurev-biochem-060815-014442. [DOI] [PubMed] [Google Scholar]
- 81.Weigert A., von Knethen A., Fuhrmann D., Dehne N., Brune B. Redox-signals and macrophage biology. Mol. Aspect. Med. 2018;63:70–87. doi: 10.1016/j.mam.2018.01.003. [DOI] [PubMed] [Google Scholar]
- 82.Oberkampf M., Guillerey C., Mouries J., Rosenbaum P., Fayolle C., Bobard A., et al. Mitochondrial reactive oxygen species regulate the induction of CD8(+) T cells by plasmacytoid dendritic cells. Nat. Commun. 2018;9:2241. doi: 10.1038/s41467-018-04686-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cappenberg A., Margraf A., Thomas K., Bardel B., McCreedy D.A., Van Marck V., et al. L-selection shedding affects bacterial clearance in the lung: a new regulatory pathway for integrin outside-in signaling. Blood. 2019;134:1445–1457. doi: 10.1182/blood.2019000685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Bedard K., Krause K.H. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol. Rev. 2007;87:245–313. doi: 10.1152/physrev.00044.2005. [DOI] [PubMed] [Google Scholar]
- 85.Griffiths H.R., Gao D., Pararasa C. Redox regulation in metabolic programming and inflammation. Redox Biol. 2017;12:50–57. doi: 10.1016/j.redox.2017.01.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kim S.Y., Jeong J.M., Kim S.J., Seo W., Kim M.H., Choi W.M., et al. Pro-inflammatory hepatic macrophages generate ROS through NADPH oxidase 2 via endocytosis of monomeric TLR4-MD2 complex. Nat. Commun. 2017;8:2247. doi: 10.1038/s41467-017-02325-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Choi J., Corder N.L., Koduru B., Wang Y. Oxidative stress and hepatic Nox proteins in chronic hepatitis C and hepatocellular carcinoma. Free Radic. Biol. Med. 2014;72:267–284. doi: 10.1016/j.freeradbiomed.2014.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Liang S., Kisseleva T., Brenner D.A. The role of NADPH oxidases (NOXs) in liver fibrosis and the activation of myofibroblasts. Front. Physiol. 2016;7:17. doi: 10.3389/fphys.2016.00017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Bogdan C. Nitric oxide synthase in innate and adaptive immunity: an update. Trends Immunol. 2015;36:161–178. doi: 10.1016/j.it.2015.01.003. [DOI] [PubMed] [Google Scholar]
- 90.Iwakiri Y., Kim M.Y. Nitric oxide in liver diseases. Trends Pharmacol. Sci. 2015;36:524–536. doi: 10.1016/j.tips.2015.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Greuter T., Shah V.H. Hepatic sinusoids in liver injury, inflammation, and fibrosis: new pathophysiological insights. J. Gastroenterol. 2016;51:511–519. doi: 10.1007/s00535-016-1190-4. [DOI] [PubMed] [Google Scholar]
- 92.Antonelou M., Michaelsson E., Evans R.D.R., Wang C.J., Henderson S.R., Walker L.S.K., et al. Therapeutic myeloperoxidase inhibition attenuates neutrophil activation, ANCA-mediated endothelial damage, and crescentic GN. J. Am. Soc. Nephrol. 2020;31:350–364. doi: 10.1681/ASN.2019060618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lehman H.K., Segal B.H. The role of neutrophils in host defense and disease. J. Allergy Clin. Immunol. 2020;145:1535–1544. doi: 10.1016/j.jaci.2020.02.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Nahon P., Sutton A., Rufat P., Ziol M., Akouche H., Laguillier C., et al. Myeloperoxidase and superoxide dismutase 2 polymorphisms comodulate the risk of hepatocellular carcinoma and death in alcoholic cirrhosis. Hepatology. 2009;50:1484–1493. doi: 10.1002/hep.23187. [DOI] [PubMed] [Google Scholar]
- 95.Maiocchi S.L., Ku J., Thai T., Chan E., Rees M.D., Thomas S.R. Myeloperoxidase: a versatile mediator of endothelial dysfunction and therapeutic target during cardiovascular disease. Pharmacol. Ther. 2021;221 doi: 10.1016/j.pharmthera.2020.107711. [DOI] [PubMed] [Google Scholar]
- 96.Heymann F., Tacke F. Immunology in the liver--from homeostasis to disease. Nat. Rev. Gastroenterol. Hepatol. 2016;13:88–110. doi: 10.1038/nrgastro.2015.200. [DOI] [PubMed] [Google Scholar]
- 97.Smith R.L., Soeters M.R., Wust R.C.I., Houtkooper R.H. Metabolic flexibility as an adaptation to energy resources and requirements in health and disease. Endocr. Rev. 2018;39:489–517. doi: 10.1210/er.2017-00211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Cappel D.A., Deja S., Duarte J.A.G., Kucejova B., Inigo M., Fletcher J.A., et al. Pyruvate-carboxylase-mediated anaplerosis promotes antioxidant capacity by sustaining TCA cycle and redox metabolism in liver. Cell Metabol. 2019;29 doi: 10.1016/j.cmet.2019.03.014. 1291-12305 e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Prieto I., Monsalve M. ROS homeostasis, a key determinant in liver ischemic-preconditioning. Redox Biol. 2017;12:1020–1025. doi: 10.1016/j.redox.2017.04.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Wu L., Liu J., Tian X., Groleau R.R., Bull S.D., Li P., et al. Fluorescent probe for the imaging of superoxide and peroxynitrite during drug-induced liver injury. Chem. Sci. 2021;12:3921–3928. doi: 10.1039/d0sc05937d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Hong G., Kim J., Oh H., Yun S., Kim C.M., Jeong Y.M., et al. Production of multiple cell-laden microtissue spheroids with a biomimetic hepatic-lobule-like structure. Adv. Mater. 2021 doi: 10.1002/adma.202102624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Chen F., Jimenez R.J., Sharma K., Luu H.Y., Hsu B.Y., Ravindranathan A., et al. Broad distribution of hepatocyte proliferation in liver homeostasis and regeneration. Cell Stem Cell. 2020;26:27–33 e4. doi: 10.1016/j.stem.2019.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Koch P.S., Lee K.H., Goerdt S., Augustin H.G. Angiodiversity and organotypic functions of sinusoidal endothelial cells. Angiogenesis. 2021;24:289–310. doi: 10.1007/s10456-021-09780-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Wang B., Wangkahart E., Secombes C.J., Wang T. Insights into the evolution of the suppressors of cytokine signaling (SOCS) gene family in vertebrates. Mol. Biol. Evol. 2019;36:393–411. doi: 10.1093/molbev/msy230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Campana L., Esser H., Huch M., Forbes S. Liver regeneration and inflammation: from fundamental science to clinical applications. Nat. Rev. Mol. Cell Biol. 2021;22:608–624. doi: 10.1038/s41580-021-00373-7. [DOI] [PubMed] [Google Scholar]
- 106.Michalopoulos G.K., DeFrances M.C. Liver regeneration. Science. 1997;276:60–66. doi: 10.1126/science.276.5309.60. [DOI] [PubMed] [Google Scholar]
- 107.Forbes S.J., Newsome P.N. Liver regeneration - mechanisms and models to clinical application. Nat. Rev. Gastroenterol. Hepatol. 2016;13:473–485. doi: 10.1038/nrgastro.2016.97. [DOI] [PubMed] [Google Scholar]
- 108.Michalopoulos G.K., Bhushan B. Liver regeneration: biological and pathological mechanisms and implications. Nat. Rev. Gastroenterol. Hepatol. 2021;18:40–55. doi: 10.1038/s41575-020-0342-4. [DOI] [PubMed] [Google Scholar]
- 109.Gadd V.L., Aleksieva N., Forbes S.J. Epithelial plasticity during liver injury and regeneration. Cell Stem Cell. 2020;27:557–573. doi: 10.1016/j.stem.2020.08.016. [DOI] [PubMed] [Google Scholar]
- 110.Shen T., Liu Y., Shang J., Xie Q., Li J., Yan M., et al. Incidence and etiology of drug-induced liver injury in mainland China. Gastroenterology. 2019;156 doi: 10.1053/j.gastro.2019.02.002. 2230-2241 e11. [DOI] [PubMed] [Google Scholar]
- 111.Andrade R.J., Chalasani N., Bjornsson E.S., Suzuki A., Kullak-Ublick G.A., Watkins P.B., et al. Drug-induced liver injury. Nat. Rev. Dis. Prim. 2019;5:58. doi: 10.1038/s41572-019-0105-0. [DOI] [PubMed] [Google Scholar]
- 112.Zhang J., Wang H., Fan Y., Yu Z., You G. Regulation of organic anion transporters: role in physiology, pathophysiology, and drug elimination. Pharmacol. Ther. 2021;217 doi: 10.1016/j.pharmthera.2020.107647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Zanger U.M., Schwab M. Cytochrome P450 enzymes in drug metabolism: regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol. Ther. 2013;138:103–141. doi: 10.1016/j.pharmthera.2012.12.007. [DOI] [PubMed] [Google Scholar]
- 114.Backman J.T., Filppula A.M., Niemi M., Neuvonen P.J. Role of cytochrome P450 2C8 in drug metabolism and interactions. Pharmacol. Rev. 2016;68:168–241. doi: 10.1124/pr.115.011411. [DOI] [PubMed] [Google Scholar]
- 115.Almazroo O.A., Miah M.K., Venkataramanan R. Drug metabolism in the liver. Clin. Liver Dis. 2017;21:1–20. doi: 10.1016/j.cld.2016.08.001. [DOI] [PubMed] [Google Scholar]
- 116.Jetter A., Kullak-Ublick G.A. Drugs and hepatic transporters: a review. Pharmacol. Res. 2020;154 doi: 10.1016/j.phrs.2019.04.018. [DOI] [PubMed] [Google Scholar]
- 117.Gong T., Liu L., Jiang W., Zhou R. DAMP-sensing receptors in sterile inflammation and inflammatory diseases. Nat. Rev. Immunol. 2020;20:95–112. doi: 10.1038/s41577-019-0215-7. [DOI] [PubMed] [Google Scholar]
- 118.Jaeschke H., Ramachandran A. Pleiotropic roles of platelets and neutrophils in cell death and recovery during acetaminophen hepatotoxicity. Hepatology. 2020;72:1873–1876. doi: 10.1002/hep.31490. [DOI] [PubMed] [Google Scholar]
- 119.Tang Y., Li H., Li J., Liu Y., Li Y., Zhou J., et al. Macrophage scavenger receptor 1 contributes to pathogenesis of fulminant hepatitis via neutrophil-mediated complement activation. J. Hepatol. 2018;68:733–743. doi: 10.1016/j.jhep.2017.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Kondylis V., Kumari S., Vlantis K., Pasparakis M. The interplay of IKK, NF-kappaB and RIPK1 signaling in the regulation of cell death, tissue homeostasis and inflammation. Immunol. Rev. 2017;277:113–127. doi: 10.1111/imr.12550. [DOI] [PubMed] [Google Scholar]
- 121.Schwabe R.F., Luedde T. Apoptosis and necroptosis in the liver: a matter of life and death. Nat. Rev. Gastroenterol. Hepatol. 2018;15:738–752. doi: 10.1038/s41575-018-0065-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kolaczkowska E., Kubes P. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 2013;13:159–175. doi: 10.1038/nri3399. [DOI] [PubMed] [Google Scholar]
- 123.Koch D.G., Speiser J.L., Durkalski V., Fontana R.J., Davern T., McGuire B., et al. The natural history of severe acute liver injury. Am. J. Gastroenterol. 2017;112:1389–1396. doi: 10.1038/ajg.2017.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Cho Y., Szabo G. Two faces of neutrophils in liver disease development and progression. Hepatology. 2021;74:503–512. doi: 10.1002/hep.31680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Kisseleva T., Brenner D. Molecular and cellular mechanisms of liver fibrosis and its regression. Nat. Rev. Gastroenterol. Hepatol. 2021;18:151–166. doi: 10.1038/s41575-020-00372-7. [DOI] [PubMed] [Google Scholar]
- 126.Petrowsky H., Fritsch R., Guckenberger M., De Oliveira M.L., Dutkowski P., Clavien P.A. Modern therapeutic approaches for the treatment of malignant liver tumours. Nat. Rev. Gastroenterol. Hepatol. 2020;17:755–772. doi: 10.1038/s41575-020-0314-8. [DOI] [PubMed] [Google Scholar]
- 127.Pinter M., Scheiner B., Peck-Radosavljevic M. Immunotherapy for advanced hepatocellular carcinoma: a focus on special subgroups. Gut. 2021;70:204–214. doi: 10.1136/gutjnl-2020-321702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Wang H., Lu Z., Zhao X. Tumorigenesis, diagnosis, and therapeutic potential of exosomes in liver cancer. J. Hematol. Oncol. 2019;12:133. doi: 10.1186/s13045-019-0806-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Yang J.D., Heimbach J.K. New advances in the diagnosis and management of hepatocellular carcinoma. BMJ. 2020;371:m3544. doi: 10.1136/bmj.m3544. [DOI] [PubMed] [Google Scholar]
- 130.Chirinos J.A., Segers P., Hughes T., Townsend R. Large-artery stiffness in health and disease: JACC state-of-the-art review. J. Am. Coll. Cardiol. 2019;74:1237–1263. doi: 10.1016/j.jacc.2019.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Greenway C.V., Stark R.D. Hepatic vascular bed. Physiol. Rev. 1971;51:23–65. doi: 10.1152/physrev.1971.51.1.23. [DOI] [PubMed] [Google Scholar]
- 132.Vargason A.M., Anselmo A.C., Mitragotri S. The evolution of commercial drug delivery technologies. Nat. Biomed. Eng. 2021;5:951–967. doi: 10.1038/s41551-021-00698-w. [DOI] [PubMed] [Google Scholar]
- 133.Madden S.K., de Araujo A.D., Gerhardt M., Fairlie D.P., Mason J.M. Taking the Myc out of cancer: toward therapeutic strategies to directly inhibit c-Myc. Mol. Cancer. 2021;20:3. doi: 10.1186/s12943-020-01291-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Forman H.J., Zhang H. Targeting oxidative stress in disease: promise and limitations of antioxidant therapy. Nat. Rev. Drug Discov. 2021;20:689–709. doi: 10.1038/s41573-021-00233-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Petroni M.L., Brodosi L., Bugianesi E., Marchesini G. Management of non-alcoholic fatty liver disease. BMJ. 2021;372:m4747. doi: 10.1136/bmj.m4747. [DOI] [PubMed] [Google Scholar]
- 136.Lackner C., Tiniakos D. Fibrosis and alcohol-related liver disease. J. Hepatol. 2019;70:294–304. doi: 10.1016/j.jhep.2018.12.003. [DOI] [PubMed] [Google Scholar]
- 137.Lautt W.W. Morgan & Claypool Life Sciences Copyright © 2010 by Morgan & Claypool Life Sciences; San Rafael (CA): 2009. Colloquium Series on Integrated Systems Physiology: from Molecule to Function to Disease. Hepatic Circulation: Physiology and Pathophysiology. [PubMed] [Google Scholar]
- 138.Radi R. Oxygen radicals, nitric oxide, and peroxynitrite: redox pathways in molecular medicine. Proc. Natl. Acad. Sci. U. S. A. 2018;115:5839–5848. doi: 10.1073/pnas.1804932115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Pizzino G., Irrera N., Cucinotta M., Pallio G., Mannino F., Arcoraci V., et al. Oxidative stress: harms and benefits for human health. Oxid. Med. Cell. Longev. 2017;2017 doi: 10.1155/2017/8416763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Lushchak V.I. Free radicals, reactive oxygen species, oxidative stress and its classification. Chem. Biol. Interact. 2014;224:164–175. doi: 10.1016/j.cbi.2014.10.016. [DOI] [PubMed] [Google Scholar]
- 141.Sousa de Almeida M., Susnik E., Drasler B., Taladriz-Blanco P., Petri-Fink A., Rothen-Rutishauser B. Understanding nanoparticle endocytosis to improve targeting strategies in nanomedicine. Chem. Soc. Rev. 2021;50:5397–5434. doi: 10.1039/d0cs01127d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Baig N., Kammakakam I., Falath W. Nanomaterials: a review of synthesis methods, properties, recent progress, and challenges. Mater. Adv. 2021;2:1821–1871. [Google Scholar]
- 143.Izci M., Maksoudian C., Manshian B.B., Soenen S.J. The use of alternative strategies for enhanced nanoparticle delivery to solid tumors. Chem. Rev. 2021;121:1746–1803. doi: 10.1021/acs.chemrev.0c00779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Mitchell M.J., Billingsley M.M., Haley R.M., Wechsler M.E., Peppas N.A., Langer R. Engineering precision nanoparticles for drug delivery. Nat. Rev. Drug Discov. 2021;20:101–124. doi: 10.1038/s41573-020-0090-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Cabral H., Kinoh H., Kataoka K. Tumor-targeted nanomedicine for immunotherapy. Acc. Chem. Res. 2020;53:2765–2776. doi: 10.1021/acs.accounts.0c00518. [DOI] [PubMed] [Google Scholar]
- 146.Poon W., Zhang Y.N., Ouyang B., Kingston B.R., Wu J.L.Y., Wilhelm S., et al. Elimination pathways of nanoparticles. ACS Nano. 2019;13:5785–5798. doi: 10.1021/acsnano.9b01383. [DOI] [PubMed] [Google Scholar]
- 147.Zhang Y.N., Poon W., Tavares A.J., McGilvray I.D., Chan W.C.W. Nanoparticle-liver interactions: cellular uptake and hepatobiliary elimination. J. Contr. Release. 2016;240:332–348. doi: 10.1016/j.jconrel.2016.01.020. [DOI] [PubMed] [Google Scholar]
- 148.Mahdinloo S., Kiaie S.H., Amiri A., Hemmati S., Valizadeh H., Zakeri-Milani P. Efficient drug and gene delivery to liver fibrosis: rationale, recent advances, and perspectives. Acta Pharm. Sin. B. 2020;10:1279–1293. doi: 10.1016/j.apsb.2020.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Peng W., Cheng S., Bao Z., Wang Y., Zhou W., Wang J., et al. Advances in the research of nanodrug delivery system for targeted treatment of liver fibrosis. Biomed. Pharmacother. 2021;137 doi: 10.1016/j.biopha.2021.111342. [DOI] [PubMed] [Google Scholar]
- 150.Zhu S., Zhang J., Zhang L., Ma W., Man N., Liu Y., et al. Inhibition of kupffer cell autophagy abrogates nanoparticle-induced liver injury. Adv. Healthc. Mater. 2017;6 doi: 10.1002/adhm.201601252. [DOI] [PubMed] [Google Scholar]
- 151.Maeso-Diaz R., Ortega-Ribera M., Lafoz E., Lozano J.J., Baiges A., Frances R., et al. Aging influences hepatic microvascular biology and liver fibrosis in advanced chronic liver disease. Aging Dis. 2019;10:684–698. doi: 10.14336/AD.2019.0127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Guillaume M., Rodriguez-Vilarrupla A., Gracia-Sancho J., Rosado E., Mancini A., Bosch J., et al. Recombinant human manganese superoxide dismutase reduces liver fibrosis and portal pressure in CCl4-cirrhotic rats. J. Hepatol. 2013;58:240–246. doi: 10.1016/j.jhep.2012.09.010. [DOI] [PubMed] [Google Scholar]
- 153.Oro D., Yudina T., Fernandez-Varo G., Casals E., Reichenbach V., Casals G., et al. Cerium oxide nanoparticles reduce steatosis, portal hypertension and display anti-inflammatory properties in rats with liver fibrosis. J. Hepatol. 2016;64:691–698. doi: 10.1016/j.jhep.2015.10.020. [DOI] [PubMed] [Google Scholar]
- 154.Yuan L., Wang J., Shen W.C. Reversible lipidization of somatostatin analogues for the liver targeting. Eur. J. Pharm. Biopharm. 2008;70:615–620. doi: 10.1016/j.ejpb.2008.05.004. [DOI] [PubMed] [Google Scholar]
- 155.Akinc A., Maier M.A., Manoharan M., Fitzgerald K., Jayaraman M., Barros S., et al. The Onpattro story and the clinical translation of nanomedicines containing nucleic acid-based drugs. Nat. Nanotechnol. 2019;14:1084–1087. doi: 10.1038/s41565-019-0591-y. [DOI] [PubMed] [Google Scholar]
- 156.Lu J., Holmgren A. The thioredoxin antioxidant system. Free Radic. Biol. Med. 2014;66:75–87. doi: 10.1016/j.freeradbiomed.2013.07.036. [DOI] [PubMed] [Google Scholar]
- 157.Ferret P.J., Hammoud R., Tulliez M., Tran A., Trebeden H., Jaffray P., et al. Detoxification of reactive oxygen species by a nonpeptidyl mimic of superoxide dismutase cures acetaminophen-induced acute liver failure in the mouse. Hepatology. 2001;33:1173–1180. doi: 10.1053/jhep.2001.24267. [DOI] [PubMed] [Google Scholar]
- 158.Malassagne B., Ferret P.J., Hammoud R., Tulliez M., Bedda S., Trebeden H., et al. The superoxide dismutase mimetic MnTBAP prevents Fas-induced acute liver failure in the mouse. Gastroenterology. 2001;121:1451–1459. doi: 10.1053/gast.2001.29590. [DOI] [PubMed] [Google Scholar]
- 159.Yu Z., Lou R., Pan W., Li N., Tang B. Nanoenzymes in disease diagnosis and therapy. Chem. Commun. 2020;56:15513–15524. doi: 10.1039/d0cc05427e. [DOI] [PubMed] [Google Scholar]
- 160.Poilil Surendran S., George Thomas R., Moon M.J., Jeong Y.Y. Nanoparticles for the treatment of liver fibrosis. Int. J. Nanomed. 2017;12:6997–7006. doi: 10.2147/IJN.S145951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Zhang R., Yan X., Fan K. 2021. Nanozymes Inspired by Natural Enzymes. [Google Scholar]
- 162.Yang B., Chen Y., Shi J. Reactive oxygen species (ROS)-Based nanomedicine. Chem. Rev. 2019;119:4881–4985. doi: 10.1021/acs.chemrev.8b00626. [DOI] [PubMed] [Google Scholar]
- 163.Casals E., Zeng M., Parra-Robert M., Fernandez-Varo G., Morales-Ruiz M., Jimenez W., et al. Cerium oxide nanoparticles: advances in biodistribution, toxicity, and preclinical exploration. Small. 2020;16 doi: 10.1002/smll.201907322. [DOI] [PubMed] [Google Scholar]
- 164.Ma J., Mercer R.R., Barger M., Schwegler-Berry D., Cohen J.M., Demokritou P., et al. Effects of amorphous silica coating on cerium oxide nanoparticles induced pulmonary responses. Toxicol. Appl. Pharmacol. 2015;288:63–73. doi: 10.1016/j.taap.2015.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Ma J.Y., Young S.H., Mercer R.R., Barger M., Schwegler-Berry D., Ma J.K., et al. Interactive effects of cerium oxide and diesel exhaust nanoparticles on inducing pulmonary fibrosis. Toxicol. Appl. Pharmacol. 2014;278:135–147. doi: 10.1016/j.taap.2014.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Ma J.Y., Mercer R.R., Barger M., Schwegler-Berry D., Scabilloni J., Ma J.K., et al. Induction of pulmonary fibrosis by cerium oxide nanoparticles. Toxicol. Appl. Pharmacol. 2012;262:255–264. doi: 10.1016/j.taap.2012.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Annangi B., Lu Z., Bruniaux J., Ridoux A., da Silva V.M., Vantelon D., et al. Macrophage autophagy protects mice from cerium oxide nanoparticle-induced lung fibrosis. Part. Fibre Toxicol. 2021;18:6. doi: 10.1186/s12989-021-00398-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Li H., Yang Z.Y., Liu C., Zeng Y.P., Hao Y.H., Gu Y., et al. PEGylated ceria nanoparticles used for radioprotection on human liver cells under gamma-ray irradiation. Free Radic. Biol. Med. 2015;87:26–35. doi: 10.1016/j.freeradbiomed.2015.06.010. [DOI] [PubMed] [Google Scholar]
- 169.Hashem R.M., Rashd L.A., Hashem K.S., Soliman H.M. Cerium oxide nanoparticles alleviate oxidative stress and decreases Nrf-2/HO-1 in D-GALN/LPS induced hepatotoxicity. Biomed. Pharmacother. 2015;73:80–86. doi: 10.1016/j.biopha.2015.05.006. [DOI] [PubMed] [Google Scholar]
- 170.Adebayo O.A., Akinloye O., Adaramoye O.A. Cerium oxide nanoparticles attenuate oxidative stress and inflammation in the liver of diethylnitrosamine-treated mice. Biol. Trace Elem. Res. 2020;193:214–225. doi: 10.1007/s12011-019-01696-5. [DOI] [PubMed] [Google Scholar]
- 171.Kobyliak N., Virchenko O., Falalyeyeva T., Kondro M., Beregova T., Bodnar P., et al. Cerium dioxide nanoparticles possess anti-inflammatory properties in the conditions of the obesity-associated NAFLD in rats. Biomed. Pharmacother. 2017;90:608–614. doi: 10.1016/j.biopha.2017.03.099. [DOI] [PubMed] [Google Scholar]
- 172.Manne N., Arvapalli R., Graffeo V.A., Bandarupalli V.V.K., Shokuhfar T., Patel S., et al. Prophylactic treatment with cerium oxide nanoparticles attenuate hepatic ischemia reperfusion injury in sprague dawley rats. Cell. Physiol. Biochem. 2017;42:1837–1846. doi: 10.1159/000479540. [DOI] [PubMed] [Google Scholar]
- 173.Ibrahim H.G., Attia N., Hashem F., El Heneidy M.A.R. Cerium oxide nanoparticles: in pursuit of liver protection against doxorubicin-induced injury in rats. Biomed. Pharmacother. 2018;103:773–781. doi: 10.1016/j.biopha.2018.04.075. [DOI] [PubMed] [Google Scholar]
- 174.Cordoba-Jover B., Arce-Cerezo A., Ribera J., Pauta M., Oro D., Casals G., et al. Cerium oxide nanoparticles improve liver regeneration after acetaminophen-induced liver injury and partial hepatectomy in rats. J. Nanobiotechnol. 2019;17:112. doi: 10.1186/s12951-019-0544-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Ni D., Wei H., Chen W., Bao Q., Rosenkrans Z.T., Barnhart T.E., et al. Ceria nanoparticles meet hepatic ischemia-reperfusion injury: the perfect imperfection. Adv. Mater. 2019;31 doi: 10.1002/adma.201902956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Li F.Y., Qiu Y.P., Xia F., Sun H., Liao H.W., Xie A., et al. Dual detoxification and inflammatory regulation by ceria nanozymes for drug-induced liver injury therapy. Nano Today. 2020;35 [Google Scholar]
- 177.Rushworth G.F., Megson I.L. Existing and potential therapeutic uses for N-acetylcysteine: the need for conversion to intracellular glutathione for antioxidant benefits. Pharmacol. Ther. 2014;141:150–159. doi: 10.1016/j.pharmthera.2013.09.006. [DOI] [PubMed] [Google Scholar]
- 178.Caldas M., Santos A.C., Veiga F., Rebelo R., Reis R.L., Correlo V.M. Melanin nanoparticles as a promising tool for biomedical applications - a review. Acta Biomater. 2020;105:26–43. doi: 10.1016/j.actbio.2020.01.044. [DOI] [PubMed] [Google Scholar]
- 179.Liu Y., Ai K., Liu J., Deng M., He Y., Lu L. Dopamine-melanin colloidal nanospheres: an efficient near-infrared photothermal therapeutic agent for in vivo cancer therapy. Adv. Mater. 2013;25:1353–1359. doi: 10.1002/adma.201204683. [DOI] [PubMed] [Google Scholar]
- 180.Ai K., Liu Y., Ruan C., Lu L., Lu G.M. Sp2 C-dominant N-doped carbon sub-micrometer spheres with a tunable size: a versatile platform for highly efficient oxygen-reduction catalysts. Adv. Mater. 2013;25:998–1003. doi: 10.1002/adma.201203923. [DOI] [PubMed] [Google Scholar]
- 181.Liu Y., Ai K., Ji X., Askhatova D., Du R., Lu L., et al. Comprehensive insights into the multi-antioxidative mechanisms of melanin nanoparticles and their application to protect brain from injury in ischemic stroke. J. Am. Chem. Soc. 2017;139:856–862. doi: 10.1021/jacs.6b11013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Zhao C., Li Z., Chen J., Su L., Wang J., Chen D.S., et al. Site-specific biomimicry of antioxidative melanin formation and its application for acute liver injury therapy and imaging. Adv. Mater. 2021 doi: 10.1002/adma.202102391. [DOI] [PubMed] [Google Scholar]
- 183.Sun H., Zhou Y., Ren J., Qu X. Carbon nanozymes: enzymatic properties, catalytic mechanism, and applications. Angew Chem. Int. Ed. Engl. 2018;57:9224–9237. doi: 10.1002/anie.201712469. [DOI] [PubMed] [Google Scholar]
- 184.Markovic Z., Trajkovic V. Biomedical potential of the reactive oxygen species generation and quenching by fullerenes (C60) Biomaterials. 2008;29:3561–3573. doi: 10.1016/j.biomaterials.2008.05.005. [DOI] [PubMed] [Google Scholar]
- 185.Gharbi N., Pressac M., Hadchouel M., Szwarc H., Wilson S.R., Moussa F. [60]fullerene is a powerful antioxidant in vivo with no acute or subacute toxicity. Nano Lett. 2005;5:2578–2585. doi: 10.1021/nl051866b. [DOI] [PubMed] [Google Scholar]
- 186.Chen G., Song X., Wang B., You G., Zhao J., Xia S., et al. Carboxyfullerene nanoparticles alleviate acute hepatic injury in severe hemorrhagic shock. Biomaterials. 2017;112:72–81. doi: 10.1016/j.biomaterials.2016.10.022. [DOI] [PubMed] [Google Scholar]
- 187.Volarevic V., Paunovic V., Markovic Z., Simovic Markovic B., Misirkic-Marjanovic M., Todorovic-Markovic B., et al. Large graphene quantum dots alleviate immune-mediated liver damage. ACS Nano. 2014;8:12098–12109. doi: 10.1021/nn502466z. [DOI] [PubMed] [Google Scholar]
- 188.Umezaki Y., Iohara D., Anraku M., Ishitsuka Y., Irie T., Uekama K., et al. Preparation of hydrophilic C60(OH)10/2-hydroxypropyl-beta-cyclodextrin nanoparticles for the treatment of a liver injury induced by an overdose of acetaminophen. Biomaterials. 2015;45:115–123. doi: 10.1016/j.biomaterials.2014.12.032. [DOI] [PubMed] [Google Scholar]
- 189.Zhou Y., Zhen M., Guan M., Yu T., Ma L., Li W., et al. Amino acid modified [70] fullerene derivatives with high radical scavenging activity as promising bodyguards for chemotherapy protection. Sci. Rep. 2018;8:16573. doi: 10.1038/s41598-018-34967-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Long Y., Wei H., Li J., Li M., Wang Y., Zhang Z., et al. Prevention of hepatic ischemia-reperfusion injury by carbohydrate-derived nanoantioxidants. Nano Lett. 2020;20:6510–6519. doi: 10.1021/acs.nanolett.0c02248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Iohara D., Umezaki Y., Anraku M., Uekama K., Hirayama F. In vitro and in vivo evaluation of hydrophilic C60(OH)10/2-Hydroxypropyl-beta-cyclodextrin nanoparticles as an antioxidant. J. Pharmacol. Sci. 2016;105:2959–2965. doi: 10.1016/j.xphs.2016.04.033. [DOI] [PubMed] [Google Scholar]
- 192.Elshater A.A., Haridy M.A.M., Salman M.M.A., Fayyad A.S., Hammad S. Fullerene C60 nanoparticles ameliorated cyclophosphamide-induced acute hepatotoxicity in rats. Biomed. Pharmacother. 2018;97:53–59. doi: 10.1016/j.biopha.2017.10.134. [DOI] [PubMed] [Google Scholar]
- 193.Kuznietsova H.M., Dziubenko N.V., Lynchak O.V., Herheliuk T.S., Zavalny D.K., Remeniak O.V., et al. Effects of pristine C60 fullerenes on liver and pancreas in alpha-naphthylisothiocyanate-induced cholangitis. Dig. Dis. Sci. 2020;65:215–224. doi: 10.1007/s10620-019-05730-3. [DOI] [PubMed] [Google Scholar]
- 194.Kuznietsova H., Dziubenko N., Hurmach V., Chereschuk I., Motuziuk O., Ogloblya O., et al. Water-soluble pristine C60 fullerenes inhibit liver fibrotic alteration and prevent liver cirrhosis in rats. Oxid. Med. Cell. Longev. 2020;2020 doi: 10.1155/2020/8061246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Dugan L.L., Tian L., Quick K.L., Hardt J.I., Karimi M., Brown C., et al. Carboxyfullerene neuroprotection postinjury in Parkinsonian nonhuman primates. Ann. Neurol. 2014;76:393–402. doi: 10.1002/ana.24220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Mansuriya B.D., Altintas Z. Graphene quantum dot-based electrochemical immunosensors for biomedical applications. Materials. 2019;13 doi: 10.3390/ma13010096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Baccile N., Laurent G., Babonneau F., Fayon F., Titirici M.-M., Antonietti M. Structural characterization of hydrothermal carbon spheres by advanced solid-state MAS 13C NMR investigations. J. Phys. Chem. C. 2009;113:9644–9654. [Google Scholar]
- 198.Karahan H.E., Goh K., Zhang C.J., Yang E., Yildirim C., Chuah C.Y., et al. MXene materials for designing advanced separation membranes. Adv. Mater. 2020;32 doi: 10.1002/adma.201906697. [DOI] [PubMed] [Google Scholar]
- 199.Liu J., Lu W., Lu X., Zhang L., Dong H., Li Y. Versatile Ti3C2T x MXene for free-radical scavenging. Nano Res. 2021:1–9. doi: 10.1007/s12274-021-3751-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Ren X., Huo M., Wang M., Lin H., Zhang X., Yin J., et al. Highly catalytic niobium carbide (MXene) promotes hematopoietic recovery after radiation by free radical scavenging. ACS Nano. 2019;13:6438–6454. doi: 10.1021/acsnano.8b09327. [DOI] [PubMed] [Google Scholar]
- 201.Guo L., Xiao J., Liu H., Liu H. Selenium nanoparticles alleviate hyperlipidemia and vascular injury in ApoE-deficient mice by regulating cholesterol metabolism and reducing oxidative stress. Metallomics. 2020;12:204–217. doi: 10.1039/c9mt00215d. [DOI] [PubMed] [Google Scholar]
- 202.Bai K., Hong B., He J., Huang W. Antioxidant capacity and hepatoprotective role of chitosan-stabilized selenium nanoparticles in concanavalin A-induced liver injury in mice. Nutrients. 2020;12 doi: 10.3390/nu12030857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Mohamed A.A., Khater S.I., Hamed Arisha A., Metwally M.M.M., Mostafa-Hedeab G., El-Shetry E.S. Chitosan-stabilized selenium nanoparticles alleviate cardio-hepatic damage in type 2 diabetes mellitus model via regulation of caspase, Bax/Bcl-2, and Fas/FasL-pathway. Gene. 2021;768 doi: 10.1016/j.gene.2020.145288. [DOI] [PubMed] [Google Scholar]
- 204.Amin K.A., Hashem K.S., Alshehri F.S., Awad S.T., Hassan M.S. Antioxidant and hepatoprotective efficiency of selenium nanoparticles against acetaminophen-induced hepatic damage. Biol. Trace Elem. Res. 2017;175:136–145. doi: 10.1007/s12011-016-0748-6. [DOI] [PubMed] [Google Scholar]
- 205.Li B., Li D., Jing W., Fan J., Dahms H.U., Lee S.C., et al. Biogenic selenium and its hepatoprotective activity. Sci. Rep. 2017;7:15627. doi: 10.1038/s41598-017-13636-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Bai K., Hong B., Hong Z., Sun J., Wang C. Selenium nanoparticles-loaded chitosan/citrate complex and its protection against oxidative stress in D-galactose-induced aging mice. J. Nanobiotechnol. 2017;15:92. doi: 10.1186/s12951-017-0324-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Liu T., Xiao B., Xiang F., Tan J., Chen Z., Zhang X., et al. Ultrasmall copper-based nanoparticles for reactive oxygen species scavenging and alleviation of inflammation related diseases. Nat. Commun. 2020;11:2788. doi: 10.1038/s41467-020-16544-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Aljuhr S.A., Abdelaziz G., Essa B.M., Zaghary W.A., Sakr T.M. Hepatoprotective, antioxidant and anti-inflammatory potentials of Vit-E/C@SeNPs in rats: synthesis, characterization, biochemical, radio-biodistribution, molecular and histopathological studies. Bioorg. Chem. 2021;117 doi: 10.1016/j.bioorg.2021.105412. [DOI] [PubMed] [Google Scholar]
- 209.Wang C., Gu Z., Gu X., Tan X., Wang S., Zhang R., et al. Nano-selenium attenuates mitochondrial-associated apoptosis via the PI3K/AKT pathway in nickel-induced hepatotoxicity in vivo and in vitro. Environ. Toxicol. 2021;37:101–119. doi: 10.1002/tox.23381. [DOI] [PubMed] [Google Scholar]
- 210.Huang B., Zhang J., Hou J., Chen C. Free radical scavenging efficiency of Nano-Se in vitro. Free Radic. Biol. Med. 2003;35:805–813. doi: 10.1016/s0891-5849(03)00428-3. [DOI] [PubMed] [Google Scholar]
- 211.Hosnedlova B., Kepinska M., Skalickova S., Fernandez C., Ruttkay-Nedecky B., Peng Q., et al. Nano-selenium and its nanomedicine applications: a critical review. Int. J. Nanomed. 2018;13:2107–2128. doi: 10.2147/IJN.S157541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Yang P., Tao J., Chen F., Chen Y., He J., Shen K., et al. Multienzyme-mimic ultrafine alloyed nanoparticles in metal organic frameworks for enhanced chemodynamic therapy. Small. 2021;17 doi: 10.1002/smll.202005865. [DOI] [PubMed] [Google Scholar]
- 213.Dong C., Feng W., Xu W., Yu L., Xiang H., Chen Y., et al. The coppery age: copper (Cu)-Involved nanotheranostics. Adv. Sci. 2020;7 doi: 10.1002/advs.202001549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Zhang X., Zhang S., Yang Z., Wang Z., Tian X., Zhou R. Self-cascade MoS2 nanozymes for efficient intracellular antioxidation and hepatic fibrosis therapy. Nanoscale. 2021;13:12613–12622. doi: 10.1039/d1nr02366g. [DOI] [PubMed] [Google Scholar]
- 215.Bai H., Kong F., Feng K., Zhang X., Dong H., Liu D., et al. Prussian blue nanozymes prevent anthracycline-induced liver injury by attenuating oxidative stress and regulating inflammation. ACS Appl. Mater. Interfaces. 2021;13:42382–42395. doi: 10.1021/acsami.1c09838. [DOI] [PubMed] [Google Scholar]
- 216.Natarajan G., Perriotte-Olson C., Casey C.A., Donohue T.M., Jr., Talmon G.A., Harris E.N., et al. Effect of nanoformulated copper/zinc superoxide dismutase on chronic ethanol-induced alterations in liver and adipose tissue. Alcohol. 2019;79:71–79. doi: 10.1016/j.alcohol.2018.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Swart P.J., Hirano T., Kuipers M.E., Ito Y., Smit C., Hashida M., et al. Targeting of superoxide dismutase to the liver results in anti-inflammatory effects in rats with fibrotic livers. J. Hepatol. 1999;31:1034–1043. doi: 10.1016/s0168-8278(99)80316-x. [DOI] [PubMed] [Google Scholar]
- 218.Gopal T., Kumar N., Perriotte-Olson C., Casey C.A., Donohue T.M., Jr., Harris E.N., et al. Nanoformulated SOD1 ameliorates the combined NASH and alcohol-associated liver disease partly via regulating CYP2E1 expression in adipose tissue and liver. Am. J. Physiol. Gastrointest. Liver Physiol. 2020;318:G428–G438. doi: 10.1152/ajpgi.00217.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Lee M.S., Kim N.W., Lee J.E., Kim M.G., Yin Y., Kim S.Y., et al. Targeted cellular delivery of robust enzyme nanoparticles for the treatment of drug-induced hepatotoxicity and liver injury. Acta Biomater. 2018;81:231–241. doi: 10.1016/j.actbio.2018.09.023. [DOI] [PubMed] [Google Scholar]
- 220.Heard K.J. Acetylcysteine for acetaminophen poisoning. N. Engl. J. Med. 2008;359:285–292. doi: 10.1056/NEJMct0708278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Singh N., Khullar N., Kakkar V., Kaur I.P. Attenuation of carbon tetrachloride-induced hepatic injury with curcumin-loaded solid lipid nanoparticles. BioDrugs. 2014;28:297–312. doi: 10.1007/s40259-014-0086-1. [DOI] [PubMed] [Google Scholar]
- 222.Choudhury S.T., Das N., Ghosh S., Ghosh D., Chakraborty S., Ali N. Vesicular (liposomal and nanoparticulated) delivery of curcumin: a comparative study on carbon tetrachloride-mediated oxidative hepatocellular damage in rat model. Int. J. Nanomed. 2016;11:2179–2193. doi: 10.2147/IJN.S101886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Bisht S., Khan M.A., Bekhit M., Bai H., Cornish T., Mizuma M., et al. A polymeric nanoparticle formulation of curcumin (NanoCurc) ameliorates CCl4-induced hepatic injury and fibrosis through reduction of pro-inflammatory cytokines and stellate cell activation. Lab. Invest. 2011;91:1383–1395. doi: 10.1038/labinvest.2011.86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Kushwaha P., Yadav A., Samim M., Flora S.J.S. Combinatorial drug delivery strategy employing nano-curcumin and nano-MiADMSA for the treatment of arsenic intoxication in mouse. Chem. Biol. Interact. 2018;286:78–87. doi: 10.1016/j.cbi.2018.03.006. [DOI] [PubMed] [Google Scholar]
- 225.El-Naggar M.E., Al-Joufi F., Anwar M., Attia M.F., El-Bana M.A. Curcumin-loaded PLA-PEG copolymer nanoparticles for treatment of liver inflammation in streptozotocin-induced diabetic rats. Colloids Surf. B Biointerfaces. 2019;177:389–398. doi: 10.1016/j.colsurfb.2019.02.024. [DOI] [PubMed] [Google Scholar]
- 226.Yang K.Y., Hwang du H., Yousaf A.M., Kim D.W., Shin Y.J., Bae O.N., et al. Silymarin-loaded solid nanoparticles provide excellent hepatic protection: physicochemical characterization and in vivo evaluation. Int. J. Nanomed. 2013;8:3333–3343. doi: 10.2147/IJN.S50683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Kandimalla R., Dash S., Bhowal A.C., Kalita S., Talukdar N.C., Kundu S., et al. Glycogen-gold nanohybrid escalates the potency of silymarin. Int. J. Nanomed. 2017;12:7025–7038. doi: 10.2147/IJN.S142497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Liang J., Liu Y., Liu J., Li Z., Fan Q., Jiang Z., et al. Chitosan-functionalized lipid-polymer hybrid nanoparticles for oral delivery of silymarin and enhanced lipid-lowering effect in NAFLD. J. Nanobiotechnol. 2018;16:64. doi: 10.1186/s12951-018-0391-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Abdullah A.S., El Sayed I.E.T., El-Torgoman A.M.A., Alghamdi N.A., Ullah S., Wageh S., et al. Preparation and characterization of silymarin-conjugated gold nanoparticles with enhanced anti-fibrotic therapeutic effects against hepatic fibrosis in rats: role of MicroRNAs as molecular targets. Biomedicines. 2021;9 doi: 10.3390/biomedicines9121767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Lee C.W., Yen F.L., Huang H.W., Wu T.H., Ko H.H., Tzeng W.S., et al. Resveratrol nanoparticle system improves dissolution properties and enhances the hepatoprotective effect of resveratrol through antioxidant and anti-inflammatory pathways. J. Agric. Food Chem. 2012;60:4662–4671. doi: 10.1021/jf2050137. [DOI] [PubMed] [Google Scholar]
- 231.Hussein J., El-Banna M., Mahmoud K.F., Morsy S., Abdel Latif Y., Medhat D., et al. The therapeutic effect of nano-encapsulated and nano-emulsion forms of carvacrol on experimental liver fibrosis. Biomed. Pharmacother. 2017;90:880–887. doi: 10.1016/j.biopha.2017.04.020. [DOI] [PubMed] [Google Scholar]
- 232.Lin L., Gong H., Li R., Huang J., Cai M., Lan T., et al. Nanodrug with ROS and pH dual-sensitivity ameliorates liver fibrosis via multicellular regulation. Adv. Sci. 2020;7 doi: 10.1002/advs.201903138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Li H., Sun J.J., Chen G.Y., Wang W.W., Xie Z.T., Tang G.F., et al. Carnosic acid nanoparticles suppress liver ischemia/reperfusion injury by inhibition of ROS, Caspases and NF-kappaB signaling pathway in mice. Biomed. Pharmacother. 2016;82:237–246. doi: 10.1016/j.biopha.2016.04.064. [DOI] [PubMed] [Google Scholar]
- 234.Verma S.K., Rastogil S., Arora I., Javed K., Akhtar M., Samim M. Nanoparticle based delivery of quercetin for the treatment of carbon tetrachloride mediated liver cirrhosis in rats. J. Biomed. Nanotechnol. 2016;12:274–285. doi: 10.1166/jbn.2016.2153. [DOI] [PubMed] [Google Scholar]
- 235.Naqvi S., Sharma H., Flora S.J. Lactobionic acid conjugated quercetin loaded organically modified silica nanoparticles mitigates cyclophosphamide induced hepatocytotoxicity. Int. J. Nanomed. 2019;14:8943–8959. doi: 10.2147/IJN.S218577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Giri T.K., Mukherjee P., Barman T.K., Maity S. Nano-encapsulation of capsaicin on lipid vesicle and evaluation of their hepatocellular protective effect. Int. J. Biol. Macromol. 2016;88:236–243. doi: 10.1016/j.ijbiomac.2016.03.056. [DOI] [PubMed] [Google Scholar]
- 237.Xiao Y., Huang J., Xu J., Zeng L., Tian J., Lou Y., et al. Targeted delivery of puerarin/glycyrrhetinic acid-PEG-PBLA complex attenuated liver ischemia/reperfusion injury via modulating Toll-like receptor 4/nuclear factor-kappaB pathway. Ther. Deliv. 2018;9:245–255. doi: 10.4155/tde-2017-0106. [DOI] [PubMed] [Google Scholar]
- 238.Roy P., Das S., Auddy R.G., Mukherjee A. Engineered andrographolide nanosystems for smart recovery in hepatotoxic conditions. Int. J. Nanomed. 2014;9:4723–4735. doi: 10.2147/IJN.S65262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Mishra N., Yadav K.S., Rai V.K., Yadav N.P. Polysaccharide encrusted multilayered nano-colloidal system of andrographolide for improved hepatoprotection. AAPS PharmSciTech. 2017;18:381–392. doi: 10.1208/s12249-016-0512-4. [DOI] [PubMed] [Google Scholar]
- 240.Wang S., Du L.B., Jin L., Wang Z., Peng J., Liao N., et al. Nano-oleanolic acid alleviates metabolic dysfunctions in rats with high fat and fructose diet. Biomed. Pharmacother. 2018;108:1181–1187. doi: 10.1016/j.biopha.2018.09.150. [DOI] [PubMed] [Google Scholar]
- 241.Singh A., Ahmad I., Akhter S., Jain G.K., Iqbal Z., Talegaonkar S., et al. Nanocarrier based formulation of Thymoquinone improves oral delivery: stability assessment, in vitro and in vivo studies. Colloids Surf. B Biointerfaces. 2013;102:822–832. doi: 10.1016/j.colsurfb.2012.08.038. [DOI] [PubMed] [Google Scholar]
- 242.Sayeed S., Imam S.S., Najmi A.K., Aqil M., Akhtar M. Nonionic surfactant based thymoquinone loaded nanoproniosomal formulation: in vitro physicochemical evaluation and in vivo hepatoprotective efficacy. Drug Dev. Ind. Pharm. 2017;43:1413–1420. doi: 10.1080/03639045.2017.1318903. [DOI] [PubMed] [Google Scholar]
- 243.Kalam M.A., Raish M., Ahmed A., Alkharfy K.M., Mohsin K., Alshamsan A., et al. Oral bioavailability enhancement and hepatoprotective effects of thymoquinone by self-nanoemulsifying drug delivery system. Mater. Sci. Eng. C Mater. Biol. Appl. 2017;76:319–329. doi: 10.1016/j.msec.2017.03.088. [DOI] [PubMed] [Google Scholar]
- 244.Rathore C., Upadhyay N., Kaundal R., Dwivedi R.P., Rahatekar S., John A., et al. Enhanced oral bioavailability and hepatoprotective activity of thymoquinone in the form of phospholipidic nano-constructs. Expet Opin. Drug Deliv. 2020;17:237–253. doi: 10.1080/17425247.2020.1716728. [DOI] [PubMed] [Google Scholar]
- 245.Kwon J., Kim J., Park S., Khang G., Kang P.M., Lee D. Inflammation-responsive antioxidant nanoparticles based on a polymeric prodrug of vanillin. Biomacromolecules. 2013;14:1618–1626. doi: 10.1021/bm400256h. [DOI] [PubMed] [Google Scholar]
- 246.Lee D., Bae S., Hong D., Lim H., Yoon J.H., Hwang O., et al. H2O2-responsive molecularly engineered polymer nanoparticles as ischemia/reperfusion-targeted nanotherapeutic agents. Sci. Rep. 2013;3:2233. doi: 10.1038/srep02233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Ko E., Jeong D., Kim J., Park S., Khang G., Lee D. Antioxidant polymeric prodrug microparticles as a therapeutic system for acute liver failure. Biomaterials. 2014;35:3895–3902. doi: 10.1016/j.biomaterials.2014.01.048. [DOI] [PubMed] [Google Scholar]
- 248.Kang C., Cho W., Park M., Kim J., Park S., Shin D., et al. H2O2-triggered bubble generating antioxidant polymeric nanoparticles as ischemia/reperfusion targeted nanotheranostics. Biomaterials. 2016;85:195–203. doi: 10.1016/j.biomaterials.2016.01.070. [DOI] [PubMed] [Google Scholar]
- 249.Berwin Singh S.V., Jung E., Noh J., Yoo D., Kang C., Hyeon H., et al. Hydrogen peroxide-activatable polymeric prodrug of curcumin for ultrasound imaging and therapy of acute liver failure. Nanomedicine. 2019;16:45–55. doi: 10.1016/j.nano.2018.11.003. [DOI] [PubMed] [Google Scholar]
- 250.Nag S., Manna K., Saha M., Das Saha K. Tannic acid and vitamin E loaded PLGA nanoparticles ameliorate hepatic injury in a chronic alcoholic liver damage model via EGFR-AKT-STAT3 pathway. Nanomedicine. 2020;15:235–257. doi: 10.2217/nnm-2019-0340. [DOI] [PubMed] [Google Scholar]
- 251.Ravi G.S., Charyulu R.N., Dubey A., Prabhu P., Hebbar S., Mathias A.C. Nano-lipid complex of rutin: development, characterisation and in vivo investigation of hepatoprotective, antioxidant activity and bioavailability study in rats. AAPS PharmSciTech. 2018;19:3631–3649. doi: 10.1208/s12249-018-1195-9. [DOI] [PubMed] [Google Scholar]
- 252.Jiang X., Gu Y., Huang Y., Zhou Y., Pang N., Luo J., et al. CBD alleviates liver injuries in alcoholics with high-fat high-cholesterol diet through regulating NLRP3 inflammasome-pyroptosis pathway. Front. Pharmacol. 2021;12:724747. doi: 10.3389/fphar.2021.724747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Eguchi A., Yoshitomi T., Lazic M., Johnson C.D., Vong L.B., Wree A., et al. Redox nanoparticles as a novel treatment approach for inflammation and fibrosis associated with nonalcoholic steatohepatitis. Nanomedicine. 2015;10:2697–2708. doi: 10.2217/nnm.15.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Boonruamkaew P., Chonpathompikunlert P., Nagasaki Y. Redox nanoparticle therapeutics for acetaminophen-induced hepatotoxicity in mice. Oxid. Med. Cell. Longev. 2016;2016 doi: 10.1155/2016/4984597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 255.Wilcox C.S., Pearlman A. Chemistry and antihypertensive effects of tempol and other nitroxides. Pharmacol. Rev. 2008;60:418–469. doi: 10.1124/pr.108.000240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Thangavel S., Yoshitomi T., Sakharkar M.K., Nagasaki Y. Redox nanoparticle increases the chemotherapeutic efficiency of pioglitazone and suppresses its toxic side effects. Biomaterials. 2016;99:109–123. doi: 10.1016/j.biomaterials.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 257.Li C., Hu Y., Nie Q., Chen S., Li G., Li L., et al. A reactive oxygen species-responsive antioxidant nanotherapy for the treatment of drug-induced tissue and organ injury. Biomater. Sci. 2020;8:7117–7131. doi: 10.1039/d0bm01660h. [DOI] [PubMed] [Google Scholar]
- 258.Go Y., Lee H., Jeong L., Sun S., Hong E., Jung E., et al. Acid-triggered echogenic nanoparticles for contrast-enhanced ultrasound imaging and therapy of acute liver failure. Biomaterials. 2018;186:22–30. doi: 10.1016/j.biomaterials.2018.09.034. [DOI] [PubMed] [Google Scholar]
- 259.Wang H., Wei W., Zhang S.Y., Shen Y.X., Wang N.P., Yue L., et al. Melatonin-selenium nanoparticles protects liver against immunological injury induced by bacillus Calmette-Guerin and lipopolysaccharide. Acta Pharmacol. Sin. 2005;26:745–752. doi: 10.1111/j.1745-7254.2005.00745.x. [DOI] [PubMed] [Google Scholar]
- 260.Chen G., Deng H., Song X., Lu M., Zhao L., Xia S., et al. Reactive oxygen species-responsive polymeric nanoparticles for alleviating sepsis-induced acute liver injury in mice. Biomaterials. 2017;144:30–41. doi: 10.1016/j.biomaterials.2017.08.008. [DOI] [PubMed] [Google Scholar]
- 261.Kim J.Y., Lee D.Y., Kang S., Miao W., Kim H., Lee Y., et al. Bilirubin nanoparticle preconditioning protects against hepatic ischemia-reperfusion injury. Biomaterials. 2017;133:1–10. doi: 10.1016/j.biomaterials.2017.04.011. [DOI] [PubMed] [Google Scholar]
- 262.Quagliariello V., Vecchione R., De Capua A., Lagreca E., Iaffaioli R.V., Botti G., et al. Nano-encapsulation of coenzyme Q10 in secondary and tertiary nano-emulsions for enhanced cardioprotection and hepatoprotection in human cardiomyocytes and hepatocytes during exposure to anthracyclines and trastuzumab. Int. J. Nanomed. 2020;15:4859–4876. doi: 10.2147/IJN.S245170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Song B., Zhang C., Hu W., Guo C., Xia Z., Hu W., et al. Nano-designed carbon monoxide donor SMA/CORM2 exhibits protective effect against acetaminophen induced liver injury through macrophage reprograming and promoting liver regeneration. J. Contr. Release. 2021;331:350–363. doi: 10.1016/j.jconrel.2021.01.025. [DOI] [PubMed] [Google Scholar]
- 264.Lee Y., Kim H., Kang S., Lee J., Park J., Jon S. Bilirubin nanoparticles as a nanomedicine for anti-inflammation therapy. Angew Chem. Int. Ed. Engl. 2016;55:7460–7463. doi: 10.1002/anie.201602525. [DOI] [PubMed] [Google Scholar]
- 265.Chen Z., Vong C.T., Gao C., Chen S., Wu X., Wang S., et al. Bilirubin nanomedicines for the treatment of reactive oxygen species (ROS)-Mediated diseases. Mol. Pharm. 2020;17:2260–2274. doi: 10.1021/acs.molpharmaceut.0c00337. [DOI] [PubMed] [Google Scholar]
- 266.Wang J., Li Y., Nie G. Multifunctional biomolecule nanostructures for cancer therapy. Nat. Rev. Mater. 2021:1–18. doi: 10.1038/s41578-021-00315-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Karkhanis J., Verna E.C., Chang M.S., Stravitz R.T., Schilsky M., Lee W.M., et al. Steroid use in acute liver failure. Hepatology. 2014;59:612–621. doi: 10.1002/hep.26678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Ou Z., Zhong H., Zhang L., Deng M., Zhang W., Wang J., et al. Macrophage membrane-coated nanoparticles alleviate hepatic ischemia-reperfusion injury caused by orthotopic liver transplantation by neutralizing endotoxin. Int. J. Nanomed. 2020;15:4125–4138. doi: 10.2147/IJN.S253125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Swanson K.V., Deng M., Ting J.P. The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat. Rev. Immunol. 2019;19:477–489. doi: 10.1038/s41577-019-0165-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Zhan C., Lin G., Huang Y., Wang Z., Zeng F., Wu S. A dopamine-precursor-based nanoprodrug for in-situ drug release and treatment of acute liver failure by inhibiting NLRP3 inflammasome and facilitating liver regeneration. Biomaterials. 2021;268 doi: 10.1016/j.biomaterials.2020.120573. [DOI] [PubMed] [Google Scholar]
- 271.Lim H., Noh J., Kim Y., Kim H., Kim J., Khang G., et al. Acid-degradable cationic poly(ketal amidoamine) for enhanced RNA interference in vitro and in vivo. Biomacromolecules. 2013;14:240–247. doi: 10.1021/bm301669e. [DOI] [PubMed] [Google Scholar]
- 272.Chenitz K.B., Fernando M., Shea J.A. In-center hemodialysis attendance: patient perceptions of risks, barriers, and recommendations. Hemodial. Int. 2014;18:364–373. doi: 10.1111/hdi.12139. [DOI] [PubMed] [Google Scholar]
- 273.He C., Yin L., Song Y., Tang C., Yin C. Optimization of multifunctional chitosan-siRNA nanoparticles for oral delivery applications, targeting TNF-alpha silencing in rats. Acta Biomater. 2015;17:98–106. doi: 10.1016/j.actbio.2015.01.041. [DOI] [PubMed] [Google Scholar]
- 274.Dong Y., Siegwart D.J., Anderson D.G. Strategies, design, and chemistry in siRNA delivery systems. Adv. Drug Deliv. Rev. 2019;144:133–147. doi: 10.1016/j.addr.2019.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Zhang W., Zhou Y., Li X., Xu X., Chen Y., Zhu R., et al. Macrophage-targeting and reactive oxygen species (ROS)-responsive nanopolyplexes mediate anti-inflammatory siRNA delivery against acute liver failure (ALF) Biomater. Sci. 2018;6:1986–1993. doi: 10.1039/c8bm00389k. [DOI] [PubMed] [Google Scholar]
- 276.Liu H., Liu C., Ye L., Ma D., He X., Tang Q., et al. Nanoassemblies with effective serum tolerance capability achieving robust gene silencing efficacy for breast cancer gene therapy. Adv. Mater. 2021;33 doi: 10.1002/adma.202003523. [DOI] [PubMed] [Google Scholar]
- 277.Van Bruggen C., Hexum J.K., Tan Z., Dalal R.J., Reineke T.M. Nonviral gene delivery with cationic glycopolymers. Acc. Chem. Res. 2019;52:1347–1358. doi: 10.1021/acs.accounts.8b00665. [DOI] [PubMed] [Google Scholar]
- 278.He H., Zheng N., Song Z., Kim K.H., Yao C., Zhang R., et al. Suppression of hepatic inflammation via systemic siRNA delivery by membrane-disruptive and endosomolytic helical polypeptide hybrid nanoparticles. ACS Nano. 2016;10:1859–1870. doi: 10.1021/acsnano.5b05470. [DOI] [PubMed] [Google Scholar]
- 279.Tang Y., Zeng Z., He X., Wang T., Ning X., Feng X. SiRNA crosslinked nanoparticles for the treatment of inflammation-induced liver injury. Adv. Sci. 2017;4 doi: 10.1002/advs.201600228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Zhang J., Shen H., Xu J., Liu L., Tan J., Li M., et al. Liver-targeted siRNA lipid nanoparticles treat hepatic cirrhosis by dual antifibrotic and anti-inflammatory activities. ACS Nano. 2020;14:6305–6322. doi: 10.1021/acsnano.0c02633. [DOI] [PubMed] [Google Scholar]
- 281.Tehran M.M., Rezaei S., Jalili A., Aghaee-Bakhtiari S.H., Sahebkar A. Decoy oligodeoxynucleotide technology: an emerging paradigm for breast cancer treatment. Drug Discov. Today. 2020;25:195–200. doi: 10.1016/j.drudis.2019.10.008. [DOI] [PubMed] [Google Scholar]
- 282.Crinelli R., Bianchi M., Gentilini L., Magnani M. Design and characterization of decoy oligonucleotides containing locked nucleic acids. Nucleic Acids Res. 2002;30:2435–2443. doi: 10.1093/nar/30.11.2435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283.Zhu X., Lu L., Currier B.L., Windebank A.J., Yaszemski M.J. Controlled release of NFkappaB decoy oligonucleotides from biodegradable polymer microparticles. Biomaterials. 2002;23:2683–2692. doi: 10.1016/s0142-9612(01)00409-4. [DOI] [PubMed] [Google Scholar]
- 284.Akao C., Tanaka T., Onodera R., Ohyama A., Sato N., Motoyama K., et al. Potential use of fucose-appended dendrimer/alpha-cyclodextrin conjugates as NF-kappaB decoy carriers for the treatment of lipopolysaccharide-induced fulminant hepatitis in mice. J. Contr. Release. 2014;193:35–41. doi: 10.1016/j.jconrel.2014.07.004. [DOI] [PubMed] [Google Scholar]
- 285.Higuchi Y., Kawakami S., Yamashita F., Hashida M. The potential role of fucosylated cationic liposome/NFkappaB decoy complexes in the treatment of cytokine-related liver disease. Biomaterials. 2007;28:532–539. doi: 10.1016/j.biomaterials.2006.08.045. [DOI] [PubMed] [Google Scholar]
- 286.Chen W., Luan J., Wei G., Zhang X., Fan J., Zai W., et al. In vivo hepatocellular expression of interleukin-22 using penetratin-based hybrid nanoparticles as potential anti-hepatitis therapeutics. Biomaterials. 2018;187:66–80. doi: 10.1016/j.biomaterials.2018.09.046. [DOI] [PubMed] [Google Scholar]
- 287.Chen W., Zhang X., Fan J., Zai W., Luan J., Li Y., et al. Tethering interleukin-22 to apolipoprotein A-I ameliorates mice from acetaminophen-induced liver injury. Theranostics. 2017;7:4135–4148. doi: 10.7150/thno.20955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 288.Zai W., Chen W., Wu Z., Jin X., Fan J., Zhang X., et al. Targeted interleukin-22 gene delivery in the liver by polymetformin and penetratin-based hybrid nanoparticles to treat nonalcoholic fatty liver disease. ACS Appl. Mater. Interfaces. 2019;11:4842–4857. doi: 10.1021/acsami.8b19717. [DOI] [PubMed] [Google Scholar]
- 289.Zhang H., Liu L., Jiang C., Pan K., Deng J., Wan C. MMP9 protects against LPS-induced inflammation in osteoblasts. Innate Immun. 2020;26:259–269. doi: 10.1177/1753425919887236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Melgar-Lesmes P., Luquero A., Parra-Robert M., Mora A., Ribera J., Edelman E.R., et al. Graphene-dendrimer nanostars for targeted macrophage overexpression of metalloproteinase 9 and hepatic fibrosis precision therapy. Nano Lett. 2018;18:5839–5845. doi: 10.1021/acs.nanolett.8b02498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Ouyang W., O'Garra A. IL-10 family cytokines IL-10 and IL-22: from basic science to clinical translation. Immunity. 2019;50:871–891. doi: 10.1016/j.immuni.2019.03.020. [DOI] [PubMed] [Google Scholar]
- 292.Tacke F., Zimmermann H.W. Macrophage heterogeneity in liver injury and fibrosis. J. Hepatol. 2014;60:1090–1096. doi: 10.1016/j.jhep.2013.12.025. [DOI] [PubMed] [Google Scholar]
- 293.Tang T.T., Wang B., Lv L.L., Liu B.C. Extracellular vesicle-based Nanotherapeutics: emerging frontiers in anti-inflammatory therapy. Theranostics. 2020;10:8111–8129. doi: 10.7150/thno.47865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 294.Damania A., Jaiman D., Teotia A.K., Kumar A. Mesenchymal stromal cell-derived exosome-rich fractionated secretome confers a hepatoprotective effect in liver injury. Stem Cell Res. Ther. 2018;9:31. doi: 10.1186/s13287-017-0752-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Povero D., Pinatel E.M., Leszczynska A., Goyal N.P., Nishio T., Kim J., et al. Human induced pluripotent stem cell-derived extracellular vesicles reduce hepatic stellate cell activation and liver fibrosis. JCI Insight. 2019;5 doi: 10.1172/jci.insight.125652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Zhao H., Shang Q., Pan Z., Bai Y., Li Z., Zhang H., et al. Exosomes from adipose-derived stem cells attenuate adipose inflammation and obesity through polarizing M2 macrophages and beiging in white adipose tissue. Diabetes. 2018;67:235–247. doi: 10.2337/db17-0356. [DOI] [PubMed] [Google Scholar]
- 297.Li X., Chen R., Kemper S., Brigstock D.R. Extracellular vesicles from hepatocytes are therapeutic for toxin-mediated fibrosis and gene expression in the liver. Front. Cell Dev. Biol. 2019;7:368. doi: 10.3389/fcell.2019.00368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298.Lee J., Kim S.R., Lee C., Yi Jun, Bae S., Yoon Y.J., et al. Extracellular vesicles from in vivo liver tissue accelerate recovery of liver necrosis induced by carbon tetrachloride. J. Extracell. Vesicles. 2021;10 doi: 10.1002/jev2.12133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 299.Chen L., Chen R., Kemper S., Cong M., You H., Brigstock D.R. Therapeutic effects of serum extracellular vesicles in liver fibrosis. J. Extracell. Vesicles. 2018;7 doi: 10.1080/20013078.2018.1461505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 300.Haga H., Yan I.K., Takahashi K., Matsuda A., Patel T. Extracellular vesicles from bone marrow-derived mesenchymal stem cells improve survival from lethal hepatic failure in mice. Stem Cells Transl. Med. 2017;6:1262–1272. doi: 10.1002/sctm.16-0226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Zhuang X., Deng Z.B., Mu J., Zhang L., Yan J., Miller D., et al. Ginger-derived nanoparticles protect against alcohol-induced liver damage. J. Extracell. Vesicles. 2015;4:28713. doi: 10.3402/jev.v4.28713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Liu B., Lu Y., Chen X., Muthuraj P.G., Li X., Pattabiraman M., et al. Protective role of shiitake mushroom-derived exosome-like nanoparticles in D-galactosamine and lipopolysaccharide-induced acute liver injury in mice. Nutrients. 2020;12 doi: 10.3390/nu12020477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Chen X., Liu B., Li X., An T.T., Zhou Y., Li G., et al. Identification of anti-inflammatory vesicle-like nanoparticles in honey. J. Extracell. Vesicles. 2021;10 doi: 10.1002/jev2.12069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Haga H., Yan I.K., Borrelli D.A., Matsuda A., Parasramka M., Shukla N., et al. Extracellular vesicles from bone marrow-derived mesenchymal stem cells protect against murine hepatic ischemia/reperfusion injury. Liver Transplant. 2017;23:791–803. doi: 10.1002/lt.24770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Zhang L., Ma X.J., Fei Y.Y., Han H.T., Xu J., Cheng L., et al. Stem cell therapy in liver regeneration: focus on mesenchymal stem cells and induced pluripotent stem cells. Pharmacol. Ther. 2021 doi: 10.1016/j.pharmthera.2021.108004. [DOI] [PubMed] [Google Scholar]
- 306.Sahu A., Jeon J., Lee M.S., Yang H.S., Tae G. Nanozyme impregnated mesenchymal stem cells for hepatic ischemia-reperfusion injury alleviation. ACS Appl. Mater. Interfaces. 2021;13:25649–25662. doi: 10.1021/acsami.1c03027. [DOI] [PubMed] [Google Scholar]
- 307.Min Y., Caster J.M., Eblan M.J., Wang A.Z. Clinical translation of nanomedicine. Chem. Rev. 2015;115:11147–11190. doi: 10.1021/acs.chemrev.5b00116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Huang X., He D., Pan Z., Luo G., Deng J. Reactive-oxygen-species-scavenging nanomaterials for resolving inflammation. Mater. Today Bio. 2021;11 doi: 10.1016/j.mtbio.2021.100124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 309.Germain M., Caputo F., Metcalfe S., Tosi G., Spring K., Aslund A.K.O., et al. Delivering the power of nanomedicine to patients today. J. Contr. Release. 2020;326:164–171. doi: 10.1016/j.jconrel.2020.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Khan I., Saeed K., Khan I. Nanoparticles: properties, applications and toxicities. Arab. J. Chem. 2019;12:908–931. [Google Scholar]
- 311.Xue H.Y., Liu S., Wong H.L. Nanotoxicity: a key obstacle to clinical translation of siRNA-based nanomedicine. Nanomedicine. 2014;9:295–312. doi: 10.2217/nnm.13.204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 312.Yang W., Wang L., Mettenbrink E.M., DeAngelis P.L., Wilhelm S. Nanoparticle toxicology. Annu. Rev. Pharmacol. Toxicol. 2021;61:269–289. doi: 10.1146/annurev-pharmtox-032320-110338. [DOI] [PubMed] [Google Scholar]
- 313.Cao M., Cai R., Zhao L., Guo M., Wang L., Wang Y., et al. Molybdenum derived from nanomaterials incorporates into molybdenum enzymes and affects their activities in vivo. Nat. Nanotechnol. 2021;16:708–716. doi: 10.1038/s41565-021-00856-w. [DOI] [PubMed] [Google Scholar]
- 314.Cai X., Dong J., Liu J., Zheng H., Kaweeteerawat C., Wang F., et al. Multi-hierarchical profiling the structure-activity relationships of engineered nanomaterials at nano-bio interfaces. Nat. Commun. 2018;9:4416. doi: 10.1038/s41467-018-06869-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Yokoi T., Oda S. Models of idiosyncratic drug-induced liver injury. Annu. Rev. Pharmacol. Toxicol. 2021;61:247–268. doi: 10.1146/annurev-pharmtox-030220-015007. [DOI] [PubMed] [Google Scholar]
- 316.Avila M.A., Dufour J.F., Gerbes A.L., Zoulim F., Bataller R., Burra P., et al. Recent advances in alcohol-related liver disease (ALD): summary of a Gut round table meeting. Gut. 2020;69:764–780. doi: 10.1136/gutjnl-2019-319720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Ouyang Z., Gao Y., Shen M., Shi X. Dendrimer-based nanohybrids in cancer photomedicine. Mater. Today Bio. 2021;10 doi: 10.1016/j.mtbio.2021.100111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 318.Hua S., de Matos M.B.C., Metselaar J.M., Storm G. Current trends and challenges in the clinical translation of nanoparticulate nanomedicines: pathways for translational development and commercialization. Front. Pharmacol. 2018;9:790. doi: 10.3389/fphar.2018.00790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 319.Metselaar J.M., Lammers T. Challenges in nanomedicine clinical translation. Drug Deliv. Transl. Res. 2020;10:721–725. doi: 10.1007/s13346-020-00740-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 320.He H., Liu L., Morin E.E., Liu M., Schwendeman A. Survey of clinical translation of cancer nanomedicines-lessons learned from successes and failures. Acc. Chem. Res. 2019;52:2445–2461. doi: 10.1021/acs.accounts.9b00228. [DOI] [PubMed] [Google Scholar]
- 321.Patra J.K., Das G., Fraceto L.F., Campos E.V.R., Rodriguez-Torres M.D.P., Acosta-Torres L.S., et al. Nano based drug delivery systems: recent developments and future prospects. J. Nanobiotechnol. 2018;16:71. doi: 10.1186/s12951-018-0392-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 322.Allen T.M., Cullis P.R. Liposomal drug delivery systems: from concept to clinical applications. Adv. Drug Deliv. Rev. 2013;65:36–48. doi: 10.1016/j.addr.2012.09.037. [DOI] [PubMed] [Google Scholar]