Abstract
Background
Activation of porcine endothelial cells (PECs) is the mechanistic centerpiece of xenograft rejection. This study sought to characterize the immuno-phenotype of human T cells in response to PECs and to explore the immuno-modulation of B7 and mTOR blockade of T cells and/or PECs during xeno-responses.
Methods
Rapid memory T-cell (TM) responses to PECs were assessed by an intracellular cytokine staining. T-cell proliferation to PEC with or without belatacept or rapamycin were evaluated by a mixed lymphocyte-endothelial cell reaction (MLER). Additionally, rapamycin-pretreated PECs were used in MLER. Cell phenotypes were analyzed by flow cytometry.
Results
TNF-α/IFN-γ producers were detected in CD8+ cells stimulated by human endothelium but not PECs. MLER showed proliferation of CD4+ and CD8+ cells with predominantly memory subsets. Purified memory and naïve cells proliferated following PEC-stimulation with an increased frequency of TM in PEC-stimulated naïve cells. Proliferating cells upregulated PD-1 and CD2 expression. Belatacept partially inhibited T-cell proliferation with reduced CD2 expression and frequency of the CD8+CD2highCD28− subset. Rapamycin dramatically inhibited PEC-induced T cell proliferation, and rapamycin-preconditioned PECs failed to induce T-cell proliferation. PD-1 blockade did not restore T-cell proliferation to rapamycin-preconditioned PECs.
Conclusions
Humans lack rapid TM–mediated responses to PECs, but induce T-cell proliferative responses characterized largely as TM with increasing CD2 and PD-1 expression. B7-CD28 and mTOR blockade of T cells exhibit dramatic inhibitory effects in altering xeno-proliferating cells. Rapamycin alter PEC xeno-immunogenicity leading to inhibition of xeno-specific T-cell proliferation independent of PD-1-PD ligand interaction.
1. INTRODUCTION
Clinical allo-transplantation is limited by the supply of donor allografts. The use of pigs has been proposed as a potential source of donors due to their unlimited supply, variable size, similar physiology, and practicality of breeding in a specific pathogen-free environment(1–2). Hyperacute rejection (HAR) of discordant xenografts(3) mediated by natural Gal alpha (1,3) Gal antigen-specific antibodies(4–5) can be overcome via many methods(6–8), exposing cell-mediated immunity and delayed xenograft rejection. This is characterized by activation of endothelial cells with infiltration of activated macrophages and natural killer cells, interstitial fibrin deposition, and eventual microvascular thrombosis(9–10). A significant prolongation of functional xenografts has also been achieved in genetically modified pig–to–nonhuman primates(11–14). In this instance, xenograft rejection occurs by both humoral and cell-mediated mechanisms.
PECs represent the first barrier between hosts and their xenograft. They can be activated during cellular and humoral rejection processes and play a key role in subsequent cellular rejection by acting as antigen-presenting cells, initiating xenospecific T-cell responses by engaging in direct and indirect xenoantigen presentation(15–17) and providing costimulation(18–20). Indeed, infiltrating T cells have been detected in primates with prolonged xenografts(11–12), suggesting that xenoreactive T cells may play a pivotal role directly targeting porcine xenografts(7,9). Unlike naïve T cells, which require antigen recognition and costimulation(21), the stimulation necessary for the activation of pathogen-specific TM is reduced(22), and these T cells respond heterologously to alloantigen(23–24), resulting in costimulation blockade–resistant rejection (CoBRR)(24–25). In contrast, it is unknown whether porcine-specific TM exist in humans without prior xenoantigen exposure, and/or whether heterologously stimulated T cells, characterized as effector and terminally differentiated effector TM (24–27), can recognize and rapidly cross-react with PECs.
Costimulation blockade (CoB) targeting CD154/CD40 pathway has been evaluated in pig-to-primate models and demonstrated significant prolongation of xenograft survival(12–14, 28–29). However, the use of CD154 mAb is not clinically applicable due to thromboembolic complications in human and primates(30–31). Belatacept, a B7 inhibitor, has been approved by FDA for use in clinical kidney allotransplantation(32–33), and the use of belatacept/rapamycin-based maintenance regimen post-depletional induction effectively reduce the risk of TM–mediated CoBRR without significant side effects(34–35). Indeed, B7–CoB reduces the adaptive response in a pig-to-baboon xenotransplant model(37).
Therefore, we sought to characterize the phenotype of xenospecific T cells responding to PECs and evaluate the immunosuppressive agent belatacept and rapamycin, an mTOR inhibitor, in suppressing xenoreactive T cell–mediated responses in vitro. Herein, we have evaluated the rapid heterologous TM–mediated responses(36–37) and T cell–mediated proliferative responses to PECs, characterized their maturation state by surface phenotype, and assessed their susceptibility to belatacept or rapamycin. These agents have been studied given their use in allograft settings as alternative to calcineurin inhibitor-based immunosuppression(32–35). We failed to detect dual cytokine producers, an effector memory T-cell response to PECs. In contrast, naïve and memory cells demonstrated proliferation following PEC stimulation, and proliferating cells were predominately TM subset with significant increase of PD-1 and CD2 expression when compared with nonproliferating cells. Belatacept significantly controlled xenoreactive T-cell proliferation with a concomitant reduction of CD2 expression. mTOR blockade not only alters xenospecific T-cell proliferative responses, but also modulates PEC immunogenicity. Interestingly, the inhibitory effects of PECs with mTOR inhibition was not regulated by PD-1/PD ligand interaction. These findings may be relevant in evaluating belatacept/rapamycin-based maintenance immunosuppressive therapy in preventing xenogeneic T cell–mediated rejection.
2. MATERIALS and METHODS
2.1. Reagents
The fluorochrome-labeled monoclonal antibodies (mAbs) specific for human T-cell surface and intracellular molecules (supplementary document) were used for phenotype analysis. Dulbecco’s modified eagle medium (DMEM), and RPMI-1640 medium were obtained from Life Technologies (Carlsbad, CA). Human endothelial cells (HECs) and culture medium were purchased from Cell Applications (San Diego CA).
2.2. Cells
PECs, isolated from aortas, were cultured in DMEM containing 10% FBS followed by subculture as previously described(21,24). Primary HECs were expanded in endothelial culture medium. HECs and PECs at passage-4 were diluted with FBS containing 10% dimethyl sulfoxide at 5×105 cells/mL followed by step-down frozen in a −80°C freezer, and stored at −180 °C.
Normal human individuals were enrolled under an institutional review board–approved protocol (Pro00062495) following informed consent. Blood was collected into a vacutainer tube containing Ficoll gel followed by centrifugation according to manufacturer’s protocol (BD Biosciences). Peripheral blood mononuclear cells (PBMCs) were collected.
CD14+ monocytes were isolated from PBMCs using positive selection kit (Miltenyi Biotec, Cambridge, MA). Cells were resuspended in medium containing 10% FBS. Naïve and memory cells were purified from PBMCs using negative selection method after purification of CD14+ cells. Briefly, pan-T cells were purified from PBMCs using negative isolation kit. Cells were stained mAbs, and then sorted into naïve (CD45RA+CCR7+) and memory (CD45RA−CCR7+, CD45RA−CCR7−, and CD45RA+CCR7−) subsets using a FACSAria Sorter (BD Biosciences). The purity of T cell subsets was over 90% as measured by flow cytometry.
2.3. Xenogeneic stimulation and intracellular cytokine staining (ICCS) for rapid memory T cell response
Effector memory cells, defined as CD45RA−CCR7− and or CD45RO+ cells, are polyfunctional with rapid production of multi-cytokines following exposure to antigens(36–37). We performed ICCS to assess the rapid TM–mediated response as previously described(26–27). Briefly, defrosted PBMCs (viability >85%) were resuspended in culture medium. 5×105 PBMCs were added to 24-well plates containing PEC monolayers, cultured for 2 hours, 1 μL/mL of GolgiPlug was added to cultures, and continued to stimulate for additional 12 hours. In a selected experiment, PBMCs, stimulated with allogeneic HECs, were used as controls for rapid memory responses. PBMCs were harvested post-stimulation, and surfaced-stained with mAbs followed by fixation/permeabilization with Cytofix/Cytoperm. Cells were intracellularly stained with mAbs specific for TNF-α and IFN-γ and analyzed using flow cytometry (Fortesa, BD Biosciences). Data analysis was performed using FlowJo software (Tree Star, San Carlos, CA).
2.4. Detection of cytokines/chemokines during PBMC–PEC interaction
To evaluate the production of cytokine/chemokine during the initial interaction between PBMCs and PECs, the culture supernatants were collected from MLER and unstimulated negative control PBMCs after 24-hour interaction. The supernatants were diluted with buffer, and the cytokine and chemokine levels were measured by bead-based multiplex kit according to manufacturer’s protocol (Invitrogen, Carlsbad, CA).
2.5. Xenogeneic mixed lymphocyte-endothelial cell reaction (MLER)
To investigate the xenogeneic T cell proliferation, a Violet Proliferation Dye-450 (VPD-450)-based MLER was performed using the method described previously(38). Briefly, PECs were cultured in 24-well plates until confluence. In selected experiments, confluent PECs were pretreated with 10 ng/mL rapamycin, followed by extensive washes to remove rapamycin residual. 5×105 VPD-450-labeled PBMCs were added to each well containing PEC monolayers and cultured for 6 days. In selected experiments, 100 μg/mL of belatacept, 10 ng/mL of rapamycin, and 100 μg/mL of anti-PD-1 mAb were added to MLER. To assess proliferation of naïve and memory cells to PECs, purified naïve and memory cells were labeled with VPD-450, and 5×105 cells were incubated with PECs with or without 1×105 autologous CD14+ cells. Cells were harvested at day 6 of incubation and surface stained with mAb cocktail. Cells were analyzed using flow cytometry, and data analysis was performed using FlowJo software.
2.6. Statistical analysis
Two-sample Student’s t-test was performed to determine the statistical significance for inhibitor effects of regents on both CD4+ and CD8+ cells in response to xenogeneic stimulation. A p value of less than 0.05 was considered statistically significant.
3. RESULTS
3.1. Humans lack rapid TM–mediated responses to porcine xenogeneic antigens
Heterologous T cell–mediated immunity, responses to viral and other antigens that cross-react with allo-antigens, may alter the size of the donor-specific repertoire(23–24). Allo-specific TM–mediated responses are characterized as capable of primarily rapid producing cytokines(32–33). Previous studies have demonstrated that allo-reactive multi-cytokine producers are largely CD8+CCR7−CD45RA− and CCR7−CD45RA+ TM lacking CD28 but not CD57 expression(25–27). Nevertheless, it is unknown whether xenospecific TM are present in humans without previous exposure to xenoantigens. Therefore, we first investigated the xenospecific TM in response to PECs and HECs respectively, using ICCS assays.
We evaluated normal individuals (n=14) for rapid TM–mediated response to PEC. As shown in Figure 1a, PEC-stimulated PBMCs from all individuals did not demonstrate significant increase of TNF-α/IFN-γ dual or single cytokine producers when compared with unstimulated cells. PBMCs from three individuals were tested against PECs and three different lots of HECs followed by ICCS (Figure 1b). As anticipated, these individuals showed rapid TM–mediated response to HECs as determined by increased TNF-α single and TNF-α/IFN-γ dual producers in CCR7−CD45RA− and CCR7−CD45RA+ subsets consistant with previously reported(26–27). In contrast, their TM–mediated responses against PECs were undetectable when compared with resting cells. Additionally, these allo-specific CD8+ TM dual-cytokine producers expressed CD57 but lack CD28 expression (Figure 1c), which confirms previous reports(25). These findings suggest that unlike cross-reactivity of heterologous T-cell immunity to alloantigens, these TM may be unable to mount rapid responses to PECs.
Figure 1. Assessment of xenospecific memory T cell responses.
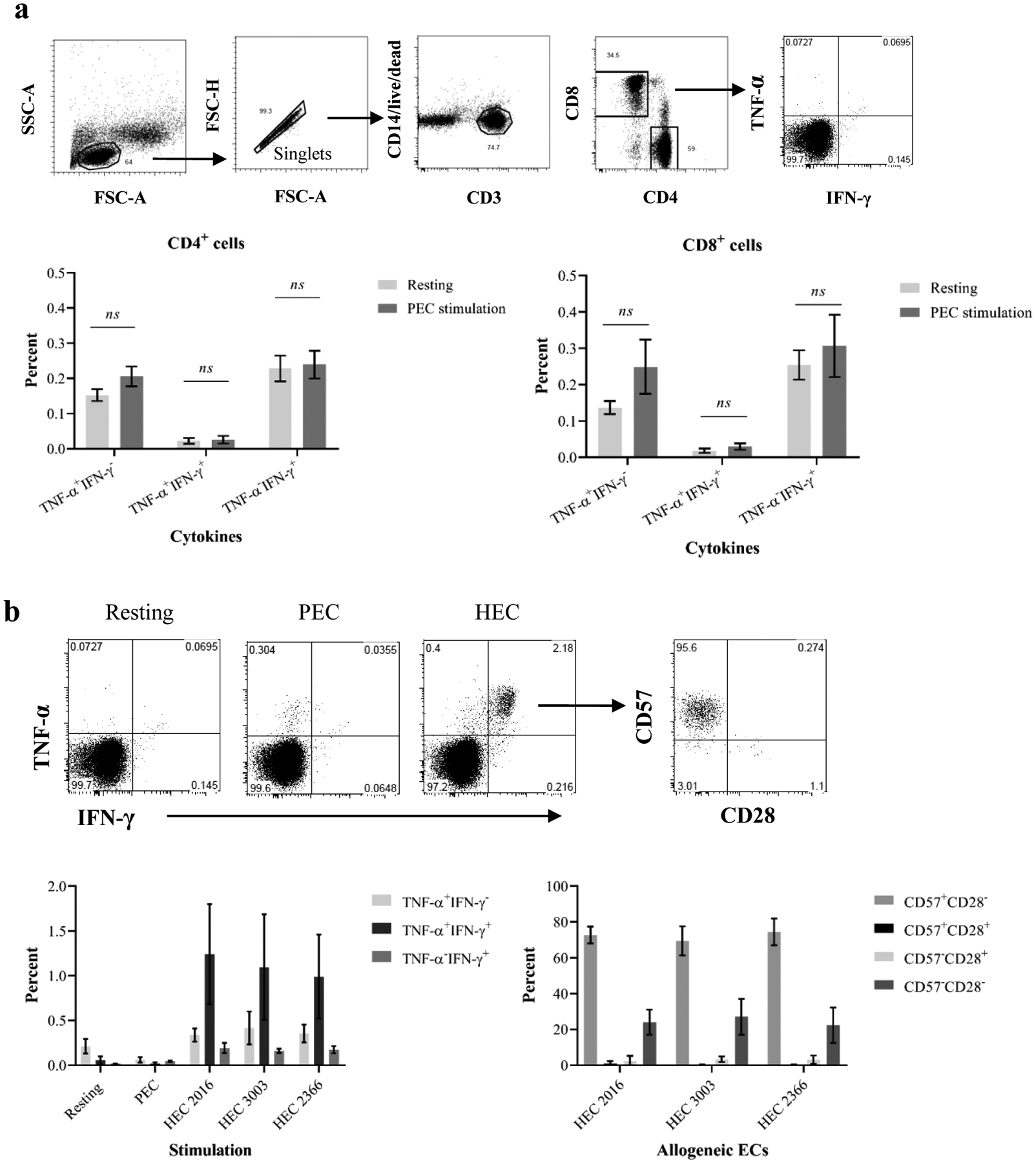
(a) Human PBMCs were stimulated with PEC monolayers in the presence of protein transporter followed by ICCS to detect cytokine producers. T cells were analyzed based on CD3, CD4, and CD8 expression (top panel). The percentage of single and dual cytokine produces in PEC-stimulated CD4+ and CD8+ cells (bottom) is similar to unstimulated cells. (b) Human PBMCs were stimulated by PECs or HECs. CD8+ cells were interrogated for TNF-a and IFN-γ production. Top panel shows a representative sample demonstrating dual cytokine producers following stimulation with HECs but not xenogeneic PECs. The dual cytokine producers were examined for CD57 and CD28 expression. Three individuals demonstrated detectable rapid memory T cell–mediated immune responses to three primary HECs but lacked immune responses to PECs (bottom left). Allo-specific dual cytokine producers are largely CD57-expressing cells but lack CD28 expression.
3.2. Human cytokine/chemokine production during early PBMC–PEC interaction
Previous studies have suggested that initial innate immune response(9–10,15,39) and natural killer (NK) cell–mediated responses(15,39) may play critical roles in DXR. Therefore, we measured cytokines and chemokines in supernatants after 24 hours of PBMC–PEC co-culture. As shown in Figure 2a, all subjects demonstrated significant production of IL-8 when compared with unstimulated cells. In contrast, IL-1-beta, CCL4, and IL-12 were undetectable following stimulation of PBMCs by PECs. Furthermore, the production of TNF-α, IFN-γ, and IL-6 were not observed during 24-hour PBMC–PEC co-culture, confirming the lack of rapid TM–mediated responses to PEC, an observation in ICCS analysis (Figure 2b). CCL5 (Rantes) was also not increased after PEC stimulation when compared with unstimulated PBMCs.
Figure 2. Detection of cytokine and chemokine production during early PBMC–PEC interaction.
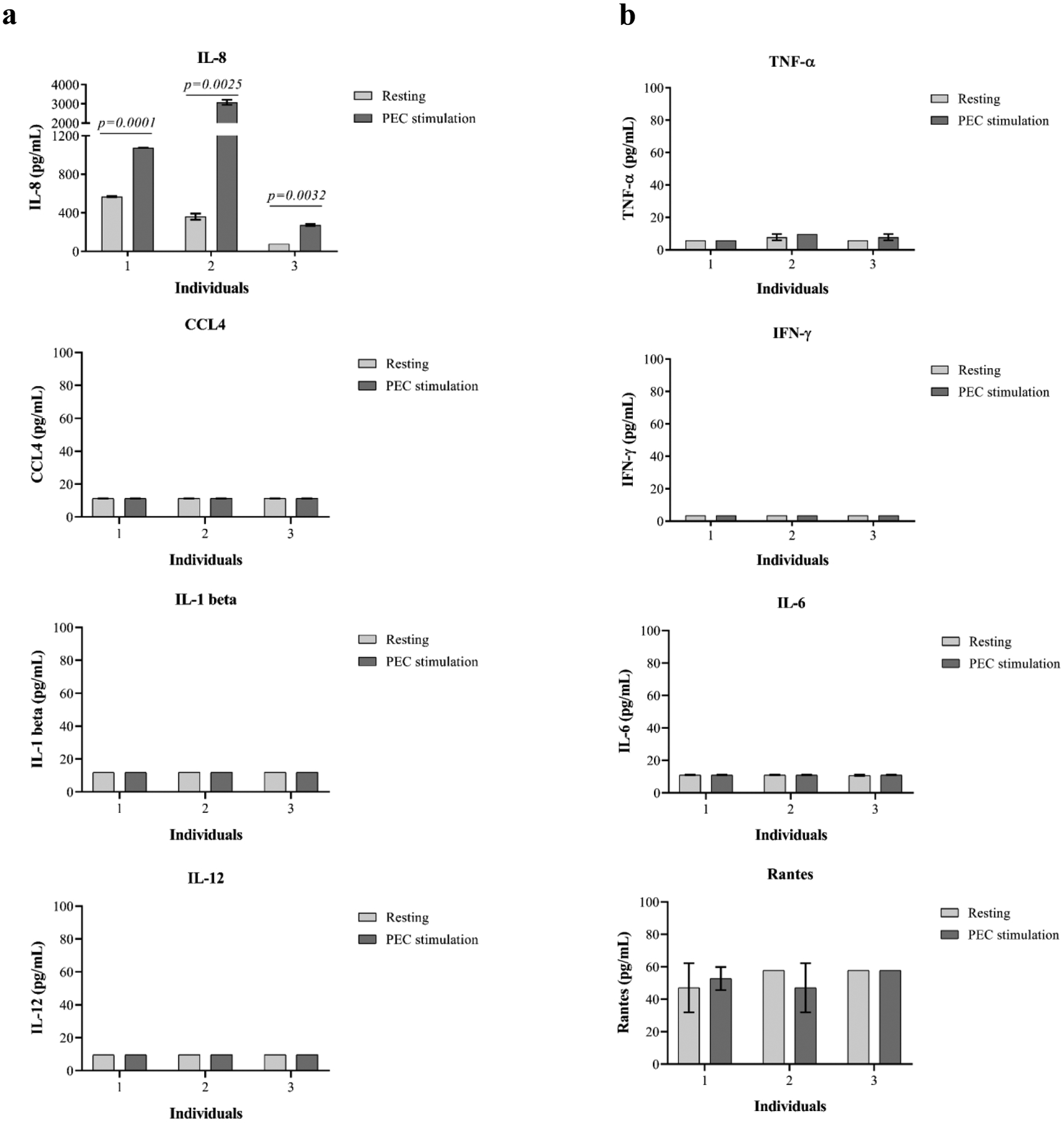
Human PBMCs were stimulated by PECs for 24 hours, and the culture supernatants were measured with multiplex cytokine/chemokine detection kits. (a) A significant increase of IL-8 but not IL-1 beta, CCL4, and IL-12 was detected in PEC-stimulated PBMCs. (b) Cytokines related to memory T cell activation are barely detectable during 24-hour PBMC–PEC interaction.
3.3. The proliferating xenospecific response is mediated by memory cells
We first evaluated the proliferative responses of PBMCs to PECs by a VPD-450–based MLER (Figure S1). As anticipated, both CD4+ and CD8+ cells from PBMCs demonstrated significant proliferative responses to PEC when compared with unstimulated PBMCs, and a highest proliferative response was observed in CD8+ population (Figure 3).
Figure 3. Xenospecific proliferative responses of human PBMCs following PEC stimulation.
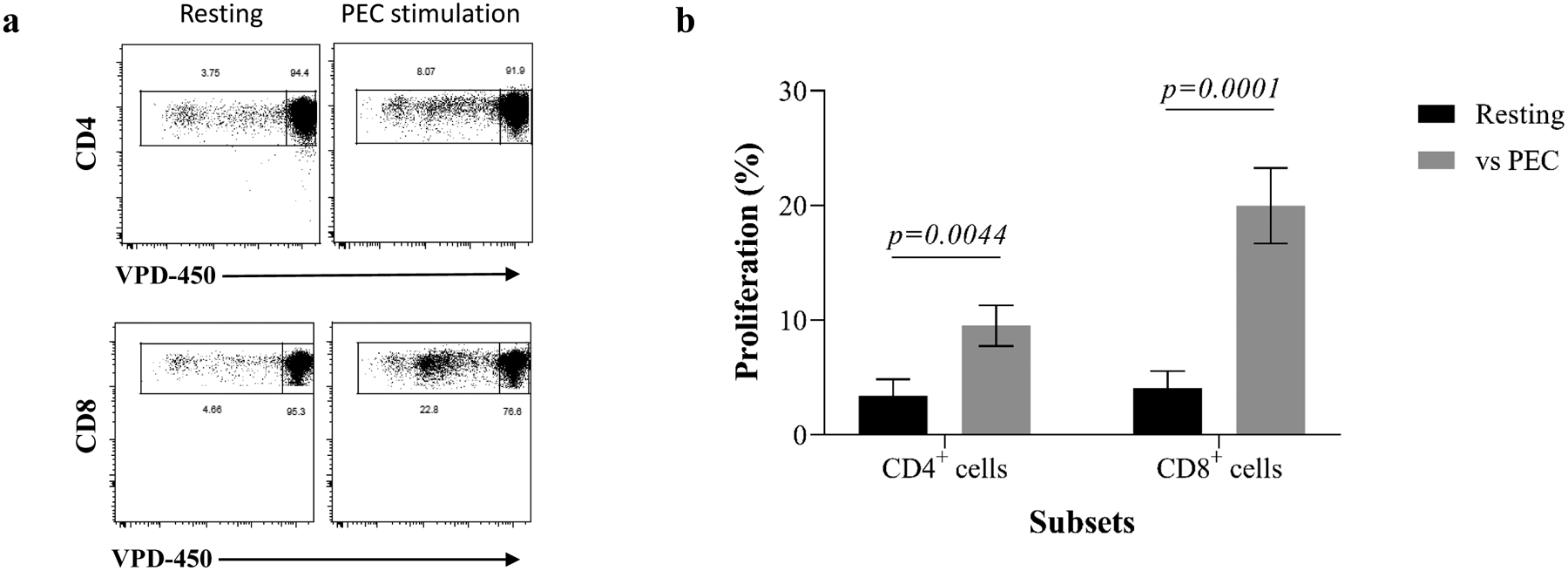
(a) VPD-450–labeled responder PBMCs were incubated with intact PEC monolayers, and CD4+ and CD8+ cells were interrogated for proliferation after 6 days through assessment of VPD-450 dilution. Representative results from one individual are shown in this panel. (b) The magnitude of proliferative responses of xenospecific CD4+ and CD8+ cells from all tested individuals after PEC stimulation are shown.
To assess the phenotype of xeno-proliferating T cells in response to PECs, CD4+, and CD8+ cells were interrogated based on surface markers, including CD2, CD28, CCR7, CD45RA, and PD-1. The proliferating cells were largely TM (Figure 4a). In a comprehensive analysis, CD4+ and CD8+ proliferating cells were segregated into four subsets based on CCR7 (CD197) and CD45RA expression (Figure 4b): naïve (TN), central memory (TC), terminally differential effector memory (TEMRA), and effector memory (TEM)(25–27,40). As shown in Figure 4c, both proliferating CD4+ and CD8+ cells demonstrated significantly higher frequencies of TEM and TC and lower frequencies of TN subsets when compared with nonproliferating and unstimulated PBMCs.
Figure 4. Phenotypical dynamics of primed proliferating T cells in response to porcine endothelial cells.
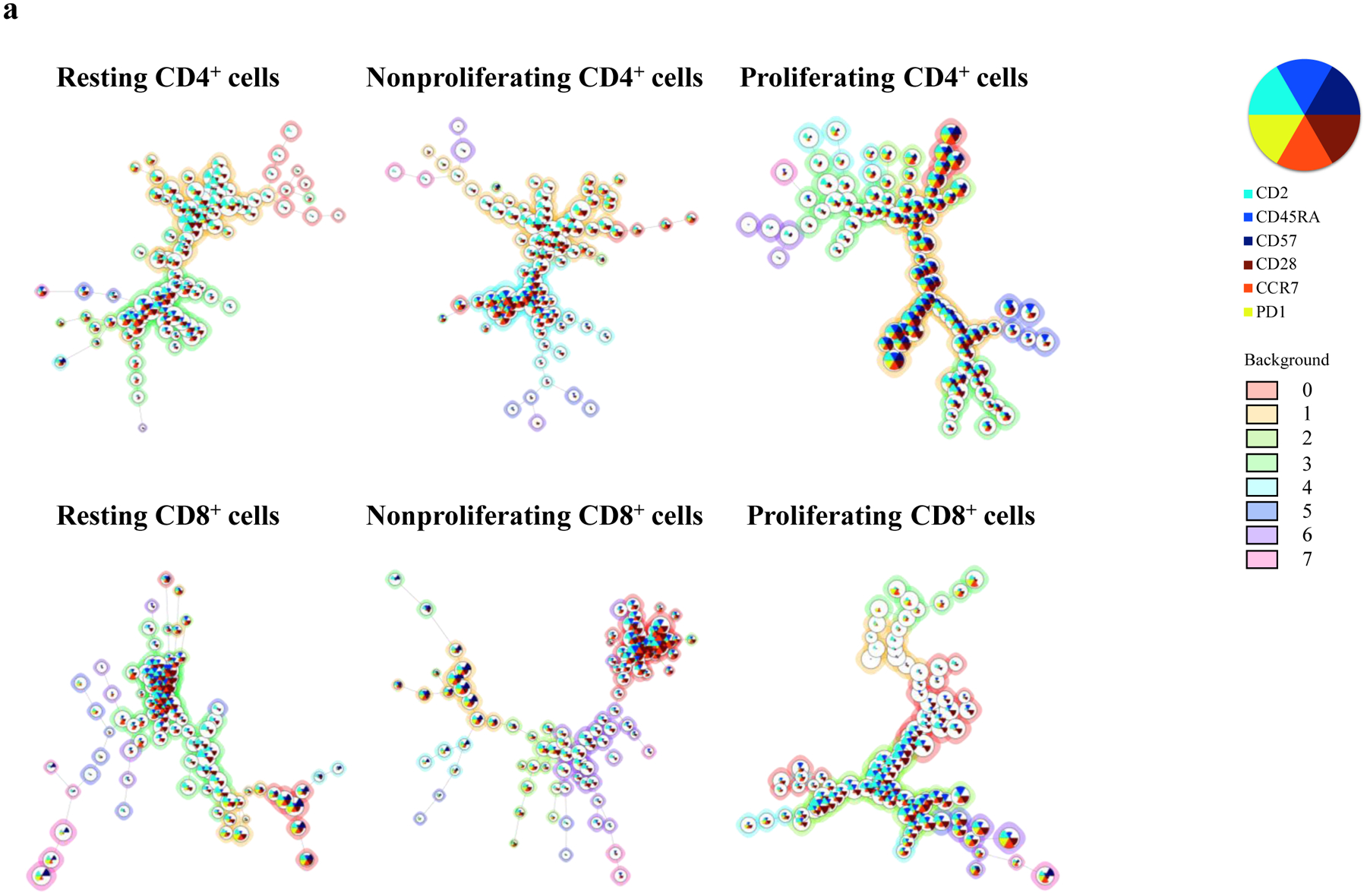
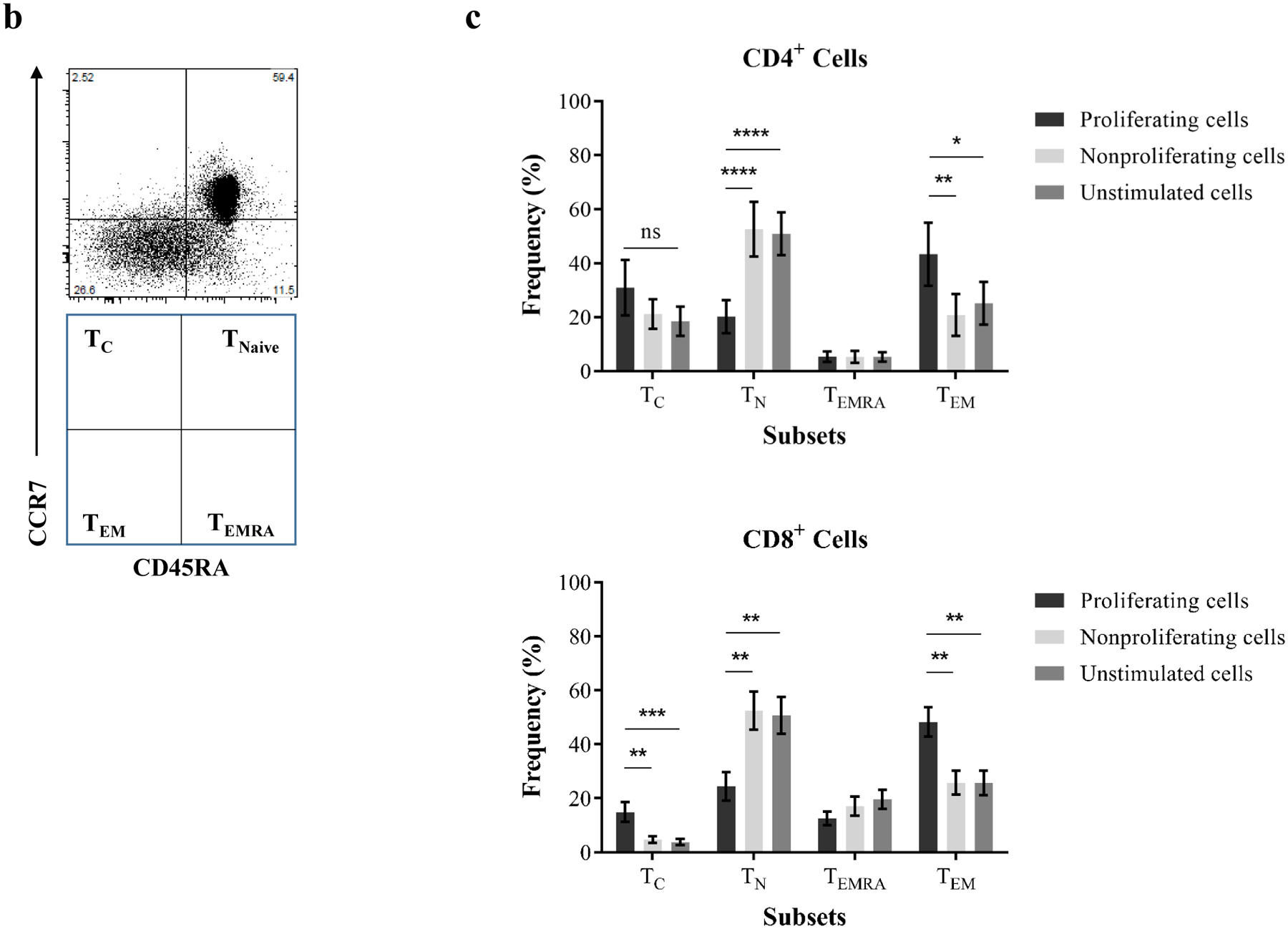
(a) A representative experiment shows the distribution of major surface marker expression on xenospecific proliferating cells, nonproliferating cells, and cells without stimulation. The mean values for markers are visualized by each node using star charts. The height indicates the intensity of surface marker expression, and the specific colors (star charts) from each node represent cell clusters. The proliferating xenoreactive cells show distinguished clusters. An automatic meta clustering of the Flow-SOM nodes is indicated by the background color of the nodes. (b) CD4+ and CD8+ cells are segregated into TN (CCR7+CD45RA+), TC (CCR7+CD45RA−), TEMRA (CCR7−CD45RA+), and TEM (CCR7−CD45RA−) subsets based on surface expression for CD45RA and CCR7. (c) Proliferating CD4+ and CD8+ cells in response to PEC demonstrate significant increase of frequency for effector memory subset when compared with unstimulated cells and nonproliferating cells. (d) Proliferation of purified naïve and memory cells in response to PEC-stimulation in the presence or absence of autologous CD14+ monocytes is assessed by flow cytometry analysis. CD4+ memory T cells (TM) show significantly higher proliferative responses then naïve T cells (TN) following PEC-stimulation. However, the presence of autologous monocytes does not augment anti-PEC proliferation. (e) The CD8+ TN and TM cells proliferate in response to PECs, and the xenogeneic specific proliferation of both TN and TM cells is not enhanced by the presence of autologous CD14+ monocytes. (f) The phenotype of proliferating cells is assessed based on surface CD45RA/CCD7 expression. A significant increase of frequency of TEM subset is observed in PEC-stimulated naïve cells.
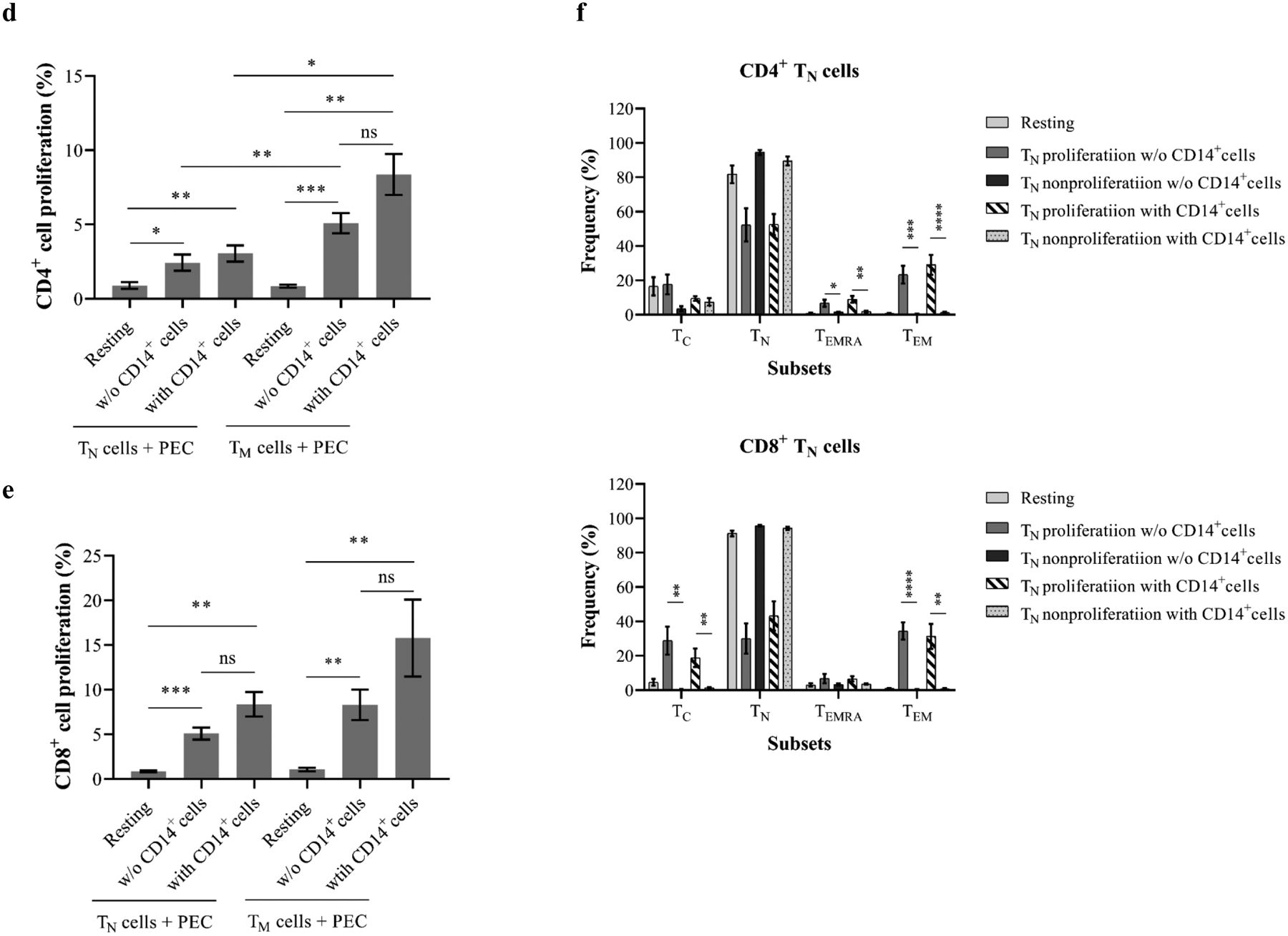
To further investigate the originating phenotype of proliferating cells induced by PEC stimulation, purified naïve and memory cells were stimulated by PECs with or without autologous CD14+ monocytes. Previous studies have suggested that monocytes may act as antigen-presenting cells in inducing T-cell proliferative responses (indirect recognition and costimulation) to alloantigens and xenoantigens(41–42). As shown in Figure 4d, both naïve and memory CD4+ cells proliferated in response to PEC with or without autologous monocytes. The proliferation of memory CD4+ cells was significantly higher than naïve cells. However, the proliferation of CD4+ cells to PECs was not augmented by autologous CD14+ monocytes. Similarly, the significant proliferation of naïve and memory CD8+ cells in response to PECs was observed in the absence of autologous CD14+ monocytes (Figure 4e). Autologous CD14+ monocytes failed to enhance the proliferative responses of naïve and memory CD8+ cells to PECs. Indeed, the proliferating cells from PEC-stimulated CD4+ and CD8+ TM maintained their memory phenotype (data not shown). In contrast, a large proportion of the proliferating cells derived from TN after PEC stimulation became TM particularly TEM phenotype (Figure 4f).
3.4. Proliferating xenoreactive T cells upregulate CD2 expression
Previous studies have suggested that as costimulatory and adhesion molecules, CD2 play an important role in PEC-induced human T cell activation(15–16, 43). Indeed, activated allo-specific T cells characterized as differentiated multi-cytokine producers express a high level of CD2, and this differentiated TM activation may be costimulation-independent and resistant to CoB(25–27, 45). To specifically evaluate the CD2 expression of xenospecific proliferating T cell subsets, PBMCs and purified naïve and memory cells were stimulated with PECs and interrogated for CD2 expression on proliferating and nonproliferating cells. As shown in Figure 5a, the CD2 expression on unstimulated CD4+ and CD8+ cells was similar to nonproliferating cells in PEC-stimulated PBMCs. In contrast, proliferating CD4+ but not CD8+ cells significantly upregulated CD2 expression (Figure 5a). Further analysis of proliferating naïve and memory CD4+ cells following PEC stimulation with or without autologous CD14+ cells demonstrated significant increase of CD2 expression when compared with nonproliferating and unstimulated cells (Figure 5b). In contrast, proliferating CD8+ cells from PEC-stimulated naïve but not memory cells showed higher CD2 expression than nonproliferating and unstimulated cells (Figure 5b).
Figure 5. Proliferating xenoreactive T cells upregulate CD2 expression.
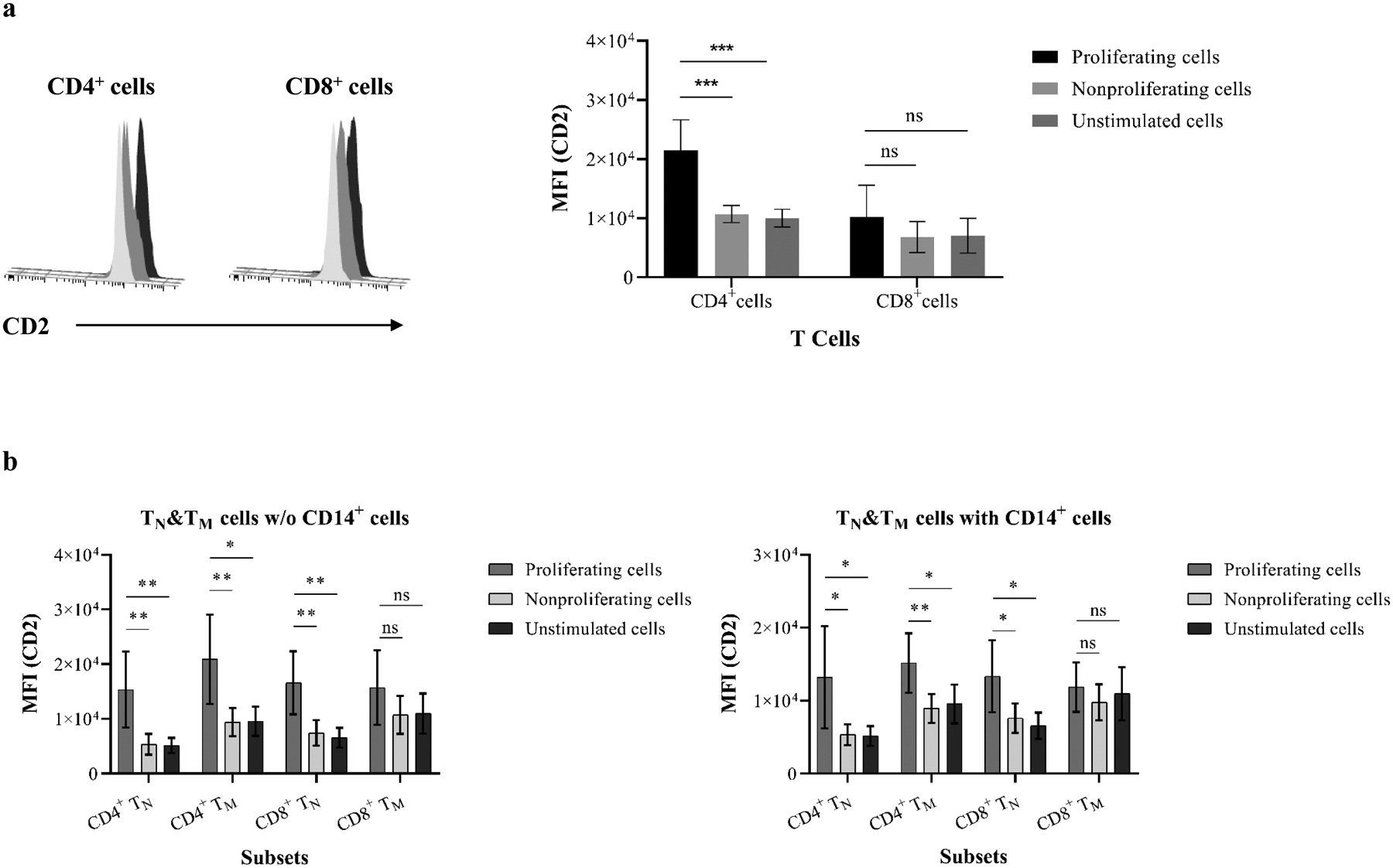
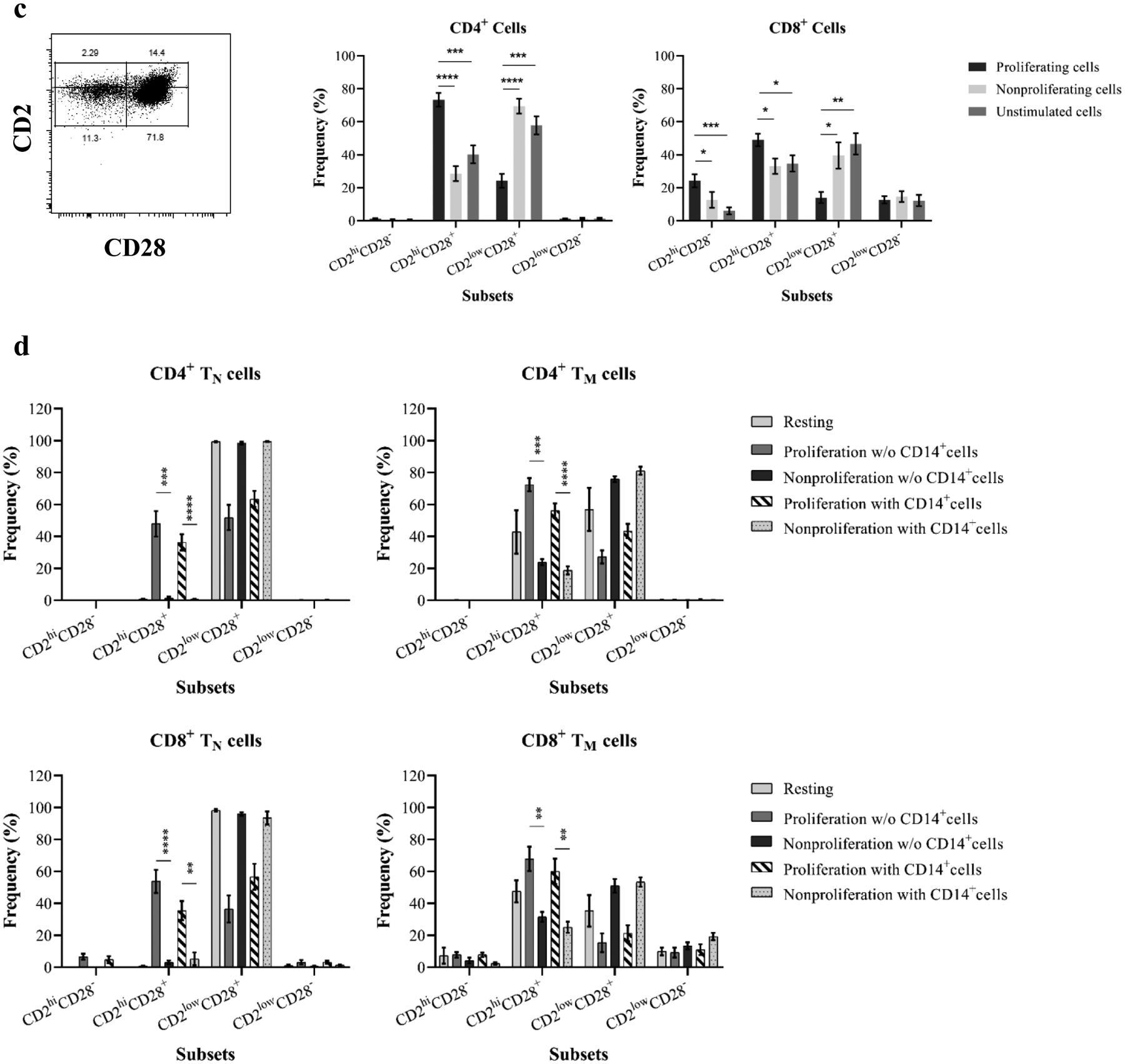
(a) PBMCs were analyzed for mean fluorescence intensity (MFI) of CD2 expression following PEC stimulation. Histogram of representative samples (left) shows CD2 MFI on proliferating CD4+ and CD8+ cells (black) when compared with resting (light gray) and nonproliferating (dark gray) cells. The MFI of CD2 on proliferating CD4+ but not CD8+ cells from all tested individuals (right) is significantly higher than resting and nonproliferating cells. (b) Purified naïve and memory cells in the presence or absence of autologous CD14+ monocytes were assessed for MFI of CD2 expression following stimulation with PEC monolayers. (c) CD4+ and CD8+ cells were segregated into four subsets based on surface CD2 and CD28 expression (left). CD4+ proliferating cells show significantly higher levels of CD2hiCD28+ subpopulation when compared with resting and nonproliferating cells. The proliferating CD8+ cells are predominantly CD2hiCD28− and CD2hiCD28+ subsets. (d) Proliferating cells in PEC-stimulated naive and memory cells in the presence or absence of autologous CD14+ monocytes showed increasing frequency of CD2hiCD28+ cells. (* p<0.05, ** p<0.001, *** p<0.0001, ****p<0.00001)
Activation of differentiated TM may be costimulation-independent as defined by lacking CD28 expression, and this subset is resistant to B7-CoB following exposure to alloantigens(26–27, 44). The xenospecific proliferating cells were analyzed based on CD28 and CD2 expression as described previously(27). As shown in Figure 5c, proliferating CD4+ and CD8+ cells in PEC-stimulated PBMCs were largely CD2hiCD28+ cells. The CD2hiCD28− subset in proliferating CD8+ cells was significantly higher than unstimulated and nonproliferating cells. Moreover, a significant increase of CD2hiCD28+ subset was observed in proliferating cells of PEC-activated naïve and memory cells with or without CD14+ monocytes (Figure 5d).
3.5. Proliferating xenoreactive T cells upregulate PD-1 expression
Programmed death-1 (PD-1), an inducible negative regulator of immune responses(45), may play a critical role in regulating allo-specific immunity(46–47). Recent study has identified that CD4+CD57+PD-1− subset may underline CoBRR in kidney transplant patients receiving belatacept-therapy without depletional induction(25). Both CD4+ and CD8+ proliferating cells from PEC-activated PBMCs, purified naïve, and memory cells were evaluated after segregating into four distinct subsets based on CD57 and PD-1 expression (Figure 6a). As shown in Figure 6b, proliferating CD4+ cells were predominantly PD-1–expressing cells without significant increase of the CD57+PD-1− subset. In contrast, although PD-1+ cells increased significantly in proliferating CD8+ cells when compared with nonproliferating cells, CD57−PD-1− cells were a predominant subset. Furthermore, a significant increase of frequency of PD1+CD57− subset in proliferating CD4+ and CD8+ cells was revealed in both naïve and memory cells following PEC stimulation with or without autologous CD14+ cells (Figure 6c).
Figure 6. Upregulation of PD-1 expression on xenoreactive proliferating T cells.
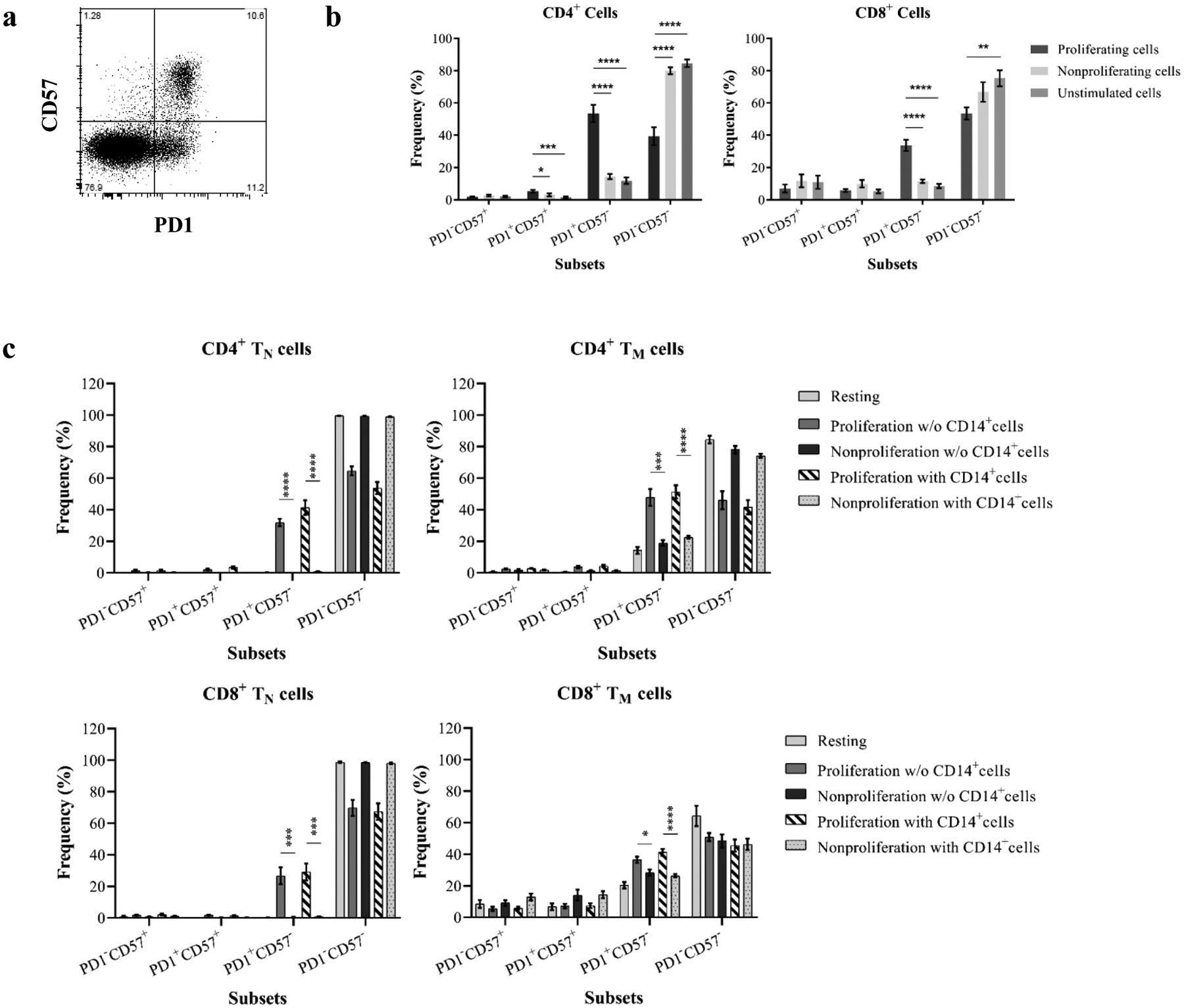
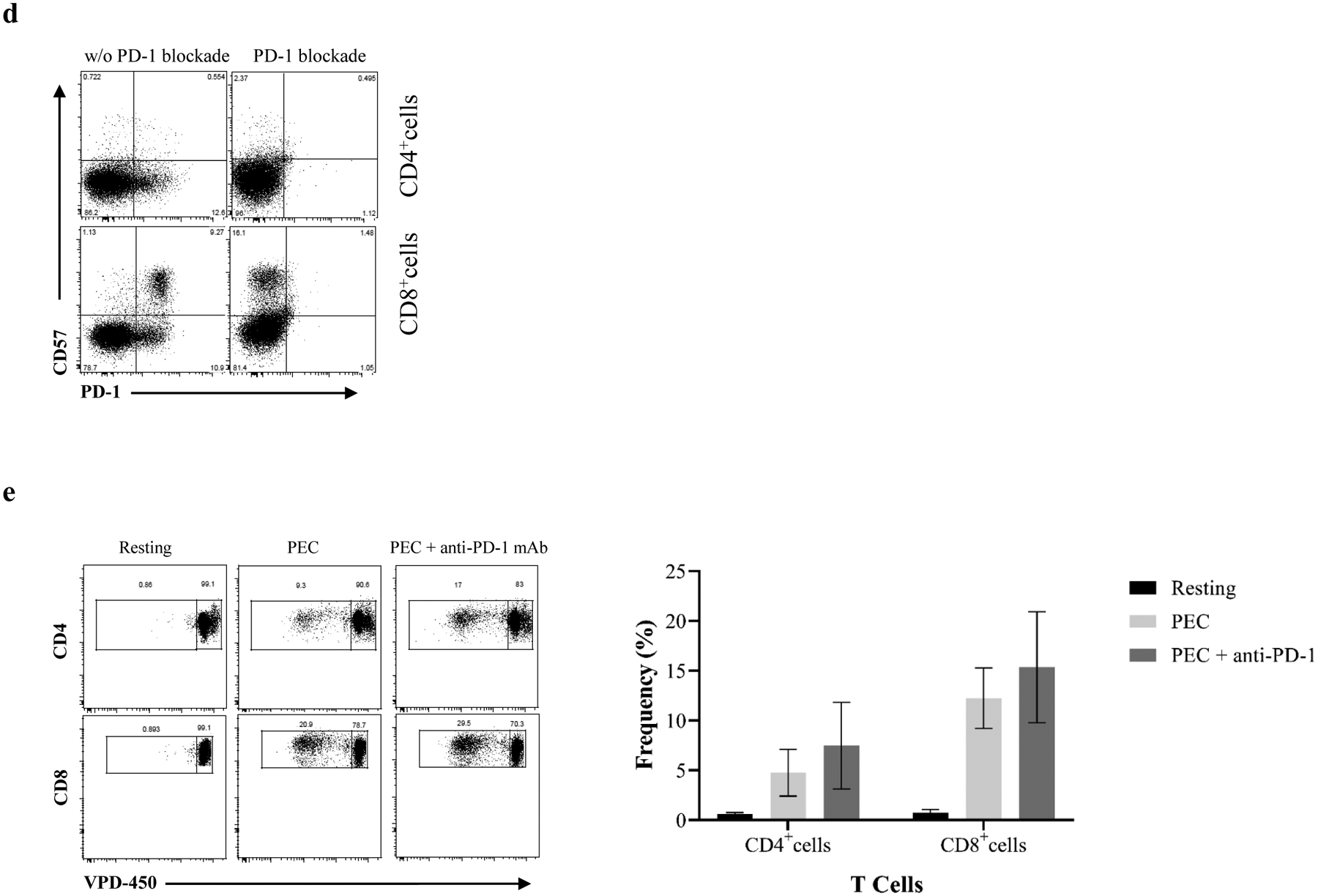
(a) CD4+ and CD8+ cells are segregated into four subsets based on surface expression of PD-1 and CD57. (b) Both CD4+ and CD8+ cells significantly upregulate PD-1but not CD57 expression following stimulation by PECs. (c) Purified naïve and memory cells were analyzed for PD-1 expression following induction of proliferative responses by PECs. An increased PD-1 expression on proliferating cells was detected in CD4+ and CD8+ cells. (d) A representative sample shows that PD-1–specific antibody completely blocks PD-1 expression on CD4+ and CD8+ cells. (e) VPD-450–labeled responder PBMCs were stimulated with PEC in the absence or presence of PD-1 specific antibody for 6 days. A representative from one individual showed the xenoreactive T cell proliferation with or without PD-1 specific antibody (left). The blockade of PD-1 expression with PD-1 specific antibody is not effective to enhance xenoreactive T cell proliferation in response to PEC. (* p<0.05, ** p<0.001, *** p<0.0001, ****p<0.00001)
To further investigate the role of PD-1–expressing cells, which may play a critical role in negatively regulating xenospecific proliferative responses, PD-1 was blocked with PD-1–specific mAb (Figure 6d) during PBMC–PEC co-cultures in selective experiments. As shown in Figure 6e, both CD4+ and CD8+ cells with PD-1 blockade did not demonstrate enhanced proliferative responses when compared with cells without of PD-1–specific antibody.
3.6. Belatacept partially inhibits xenogeneic T cell proliferation in response to PEC
Given that humans may lack cross-reactive heterologous TM-mediated immunity against PECs, the xenoreactive T-cell responses were phenotypically TEM and CD2highCD28+ subsets. Therefore, we postulated that these proliferating T cells may be susceptible to B7 inhibitor belatacept. PBMCs were stimulated with PEC monolayers in the presence or absence of belatacept, followed by detection of T-cell proliferation and evaluation of phenotype of proliferating cells (n=6). As expected (Figure 7a), the proliferation of both CD4+ and CD8+ cells in response to PEC in the presence of belatacept was markedly inhibited. Xeno-specific CD4+ but not CD8+ proliferating cells demonstrated reduction of CD2 expression when compared to proliferating cells without of B7-CoB (Figure 7b). No significant changes were observed in CD2CD28 subsets in proliferating CD4+ cells with or without B7-CoB. Although belatacept did not decrease CD2 expression on proliferating CD8+ cells, the frequencies of CD2highCD28− and CD2highCD28+ subsets were significantly reduced following belatacept treatment when compared to proliferating cells without belatacept (Figure 7c).
Figure 7. B7 blockade dramatically inhibits primary xenospecific T-cell proliferation and alters phenotype of xenospecific proliferating T cells.
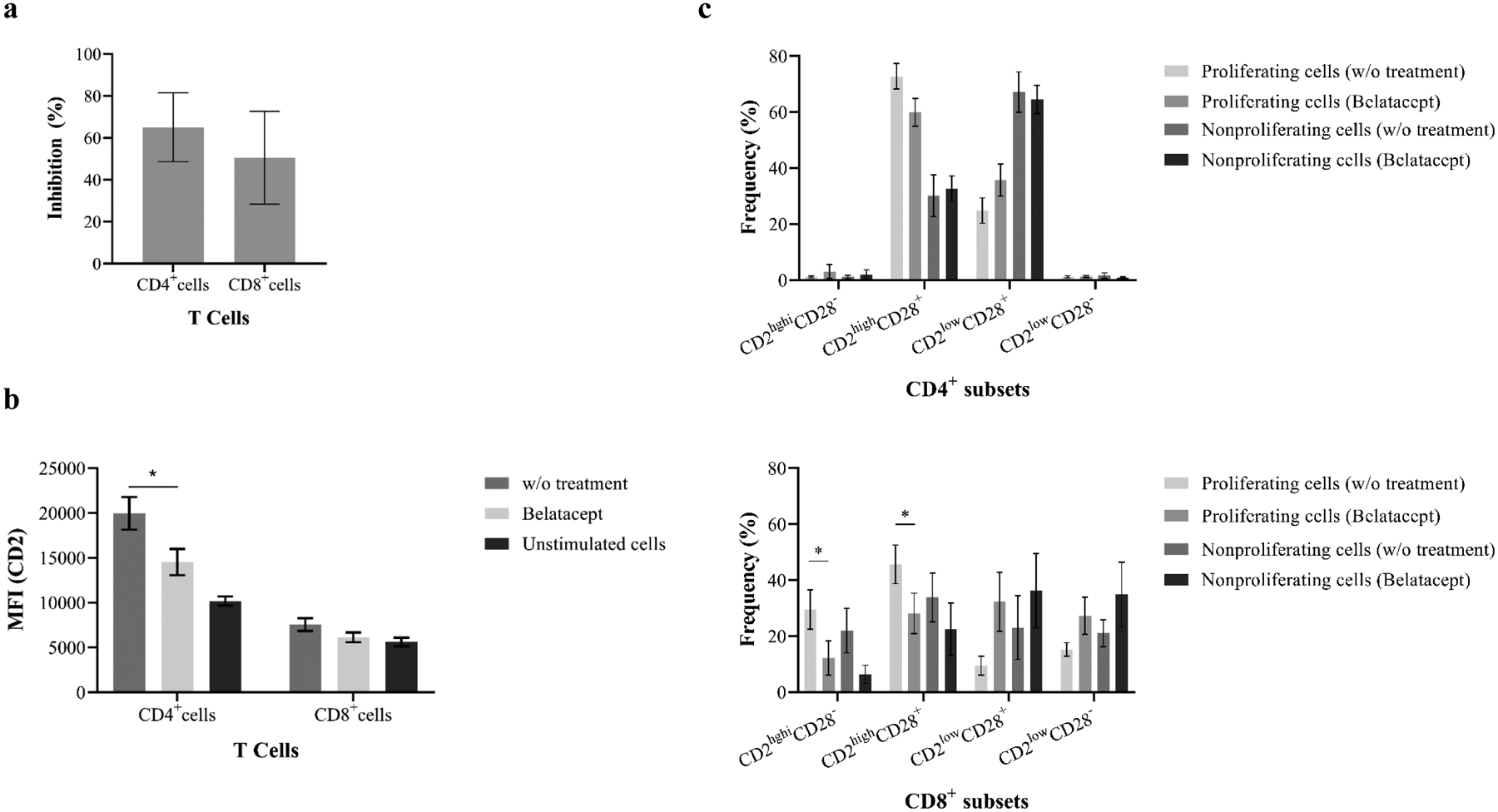
(a) VPD-450–based xenogeneic MLER was performed in the presence or absence of B7 inhibitor belatacept. Human T-cell proliferation in response to PECs was dramatically inhibited by belatacept. (b) Costimulation blockade with belatacept significantly reduced the MFI of CD2 expression on xenoreactive CD4+ proliferating cells when compared with resting and nonproliferating cells. (c) Belatacept significantly alters frequency of CD8+CD2hiCD28− and CD8+CD2hiCD28+ subsets when compared with resting and nonproliferating cells (* p<0.05).
3.7. mTOR inhibitor alters xenoreactive T cells and pig endothelial xeno-immunogenicity
The mTOR inhibitor rapamycin inhibits T cells by blocking IL-2 signaling(48), thereby facilitating activation-induced cell death and elimination of proliferating T cells clones(49). A recent study has suggested that mTOR inhibitor in combination with CD40 blockade is effective in prolongation of pig renal xenografts(50). Increasing evidence has suggested that mTOR inhibition may directly modulate endothelial surface molecules, cytokine production, and allorecognition(51–52). Therefore, we first evaluated the ability of rapamycin to inhibit T-cell proliferation in response to PECs. As shown in Figure 8, the proliferative responses of human CD4+ and CD8+ cells to PECs were dramatically reduced in the presence of rapamycin, with a 74±10.7% inhibition of CD4+ T-cell proliferation and a 76.6±7.8% inhibition of CD8+ T-cell proliferation (Figure 8). To determine if rapamycin modulated this response at the PEC level, PBMCs were stimulated by rapamycin pre-treated PECs with or without PD-1–specific blocking mAb. As shown in Figure 8, rapamycin preconditioned PECs failed to induce CD4+ and CD8+ cell proliferation. Furthermore, blocking PD-1/PD-ligand interaction with PD-1 specific mAb did not restore the proliferation of xenoreactive CD4+ and CD8+ T cells, suggesting that abrogation of human T-cell proliferative responses to PECs, induced by mTOR inhibition of PEC, is independent of PD-1/PD-ligand pathway.
Figure 8. Effects of mTOR blockade in preventing xenoreactive T-cell proliferative responses to PEC.
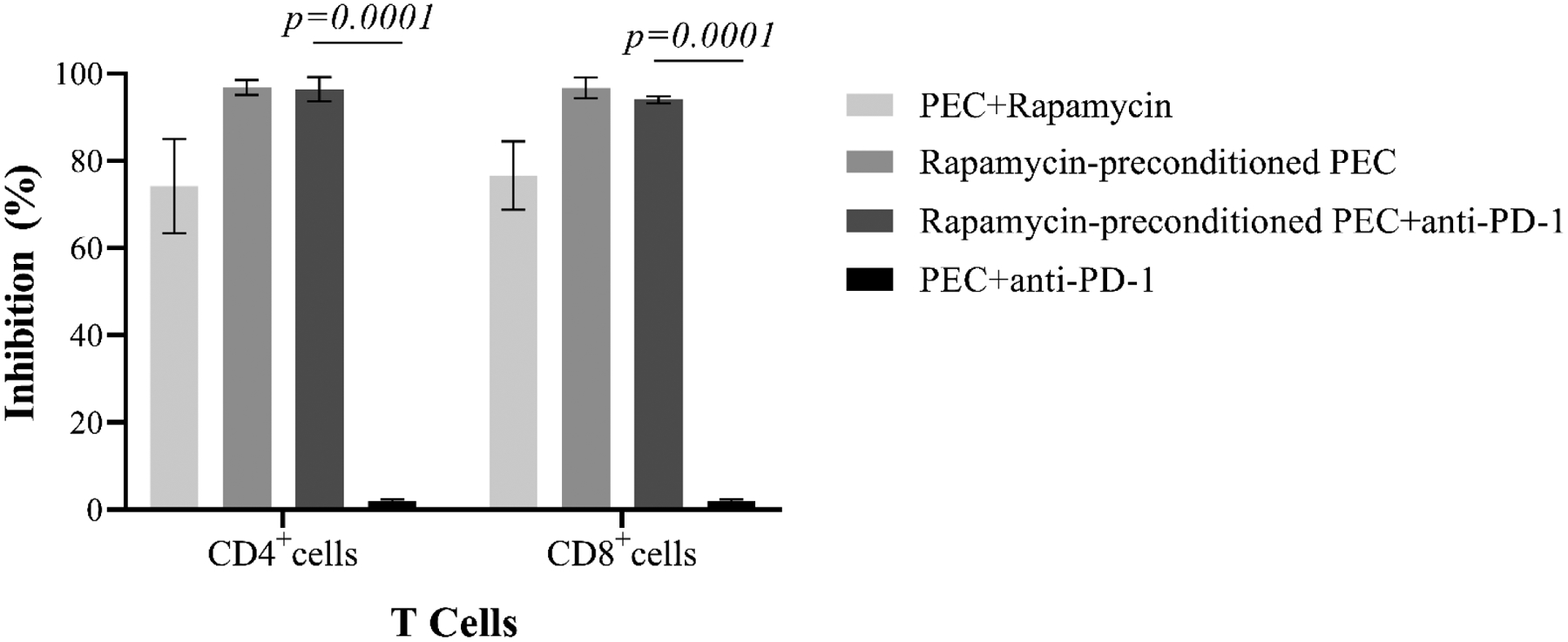
The primary proliferation of xenoreactive T cells in response to PEC was inhibited dramatically in the presence of mTOR inhibitor rapamycin. PECs, pretreated with rapamycin, are unable to induce xenoreactive T cell proliferative responses. The PD-1/PD ligand-1 blockade with PD-1–specific antibody fails to restore T-cell proliferation in response to rapamycin-pretreated PECs. PD-1 blockade does not inhibit PEC-induced T-cell proliferation.
4. DISCUSSION
As the first major interaction point between pig xenograft and host immunity, PECs are the major targets for circulating natural antibody and complement in the development of HAR(3–4). It is believed that PECs acting as antigen-presenting cells may play a key role in initiating T-cell activation via performing both direct and indirect antigen presentation(15–16, 18) and providing costimulation(19–20, 43). Certainly, TEM engage rapid responses following recognition of previously encountered pathogens(29–30). It is well-established that the requirement for costimulation in the activation of pathogen-specific TM is reduced(22), and these heterologous TM can cross-react with alloantigens even without previous exposure(23–24) and mediate CoBRR in transplant patients(24–25). However, it was unknown if heterologous TM–mediated immunity could also recognize and rapidly respond to xenoantigens presented by PECs. In this study, we specifically evaluate rapid TM–mediated responses to PEC, and find that, unlike allo-reactive TM immunity(24–27) that demonstrated rapid responses to alloantigens, humans lack pre-existing xenospecific TM as determined by absence of rapid development of TNF-α/IFN-γ producers after primary exposure to PECs. Certainly, there is a lack of detectable T cell–producing cytokines in culture supernatants within first 24 hours after T cell–PEC interaction. These findings suggest the lack of cross-reactivity between porcine xenoantigens and heterologous TM–mediated immunity. The early cell-mediated response is associated with upregulation of innate cell immunity–related cytokines, including IL-8 production with 24 hours post-stimulation, confirming previous observation in in vivo studies(7–8, 10).
PECs can induce proliferation of human PBMCs(15–16, 18–19, 38) with production of cytokine and chemokine(53–54) via direct and indirect xenorecognition. In this study, we have specifically characterized the phenotype of primary proliferating xenoreactive T cells in response to PECs. First, we show that the xenospecific proliferating CD4+ and CD8+ cells are largely TEM cells when compared with nonproliferating cells and resting cells. In vitro studies have indicated that monocytes, as antigen-presenting cells, may play an important role in indirect xenorecognition and costimulation for T-cell responses(42). We find that purified TM with or without autologous monocytes proliferate in responses to PEC stimulation, and proliferating cells maintain their memory phenotype. In contrast, purified naïve cells also proliferate after PEC stimulation, with a significant proportion of proliferating cells becoming TEM cells. The presence of autologous monocytes does not augment proliferative responses of purified T cells when compared with T cell responses in the absence of monocytes indicating that the direct xenorecognition and costimulation may play a major role in PEC-induced initial T-cell proliferation. We also find that these proliferating cells highly express CD2 molecule, a critical adhesion/costimulatory molecule for T-cell proliferation via direct xenorecognition and costimulation(16, 18 43). Furthermore, we find CD2highCD28+ cells are the predominant subset in primary proliferating CD4+ cells suggesting that this subset may be susceptible to B7-CoB. We also find that the primary proliferating CD8+ cells display an increasing frequency of CD2highCD28− cells, a subset identified previously as B7-CoB–resistant cells(26–27). Indeed, a previous study has found considerable contribution of CD4+CD4RO+ cells in response to PECs, suggesting that effectively controlling this subset may dramatically affect long-term xenograft survival(17). Increasing numbers of studies have suggested that B7-based regimens, as alternatives to calcineurin inhibitors, effectively prevent allograft rejection with depletional induction in human kidney transplantation(33–35), and the inhibition of T cell–dependent immune responses in vivo has been reported in nonhuman primate models(54–55). In this study, the primary proliferation of CD4+ and CD8+ cells in responses to PEC is dramatically inhibited by belatacept. We also find that B7-CoB alters the phenotype of xenoreactive proliferating cells with a significant reduction of CD2 expression on CD4+ cells and CD2highCD28− and CD2highCD28+ subsets in proliferating CD8+ cells.
Rapamycin, an mTOR complex-1 inhibitor, not only effectively prevents T cell–mediated allograft rejection when used as a maintenance immunosuppressive regiment with belatacept(34–35), but also may modulate human endothelium(56–58). In this study, we first demonstrate that rapamycin effectively inhibits primary xenospecific T-cell proliferative response to PECs. Certainly, the allo-specific proliferating cells may undergo activation-induced cells death in the presence of mTOR inhibitor(49). Importantly, we also find that PECs, as xenoantigen presenting cells, are unable to induce primary T-cell proliferative responses after pretreatment of PEC with rapamycin. It has been well established that the interaction between PD-1 and PD ligand-1 can negatively regulate T cell–mediated allo-specific immunity(46–47). Previous studies have showed that HECs express PD-L1 and upregulate PD-L1 expression after pretreatment with rapamycin(58). Indeed, both xenospecific proliferating CD4+ and CD8+ cells upregulate surface PD-1 expression after PEC stimulation. Therefore, we specifically investigated the role of PD-1/PD-L1 pathway in inhibiting PEC-induced primary T cell responses following pre-inhibition of PEC mTOR with rapamycin. We find that blockade of PD-1 expression with PD-1–specific antibody cannot restore the primary proliferative responses of human CD4+ and CD8+ cells to rapamycin-conditioned PECs, suggesting that inhibition of xenospecific T cell–mediated primary responses by rapamycin pretreated PECs is independent of PD-1/PD-L1 interaction. In an in vitro human endothelial-T cell model, rapamycin-treated HECs may active regulatory cells in preventing allo-specific responses(58). We are currently unable to exclude the possibility that human PD-1 may not interact directly with porcine PD-L1. Interestingly, a recent study has successfully generated human PD-L1 transgenic pigs and showed surface expression of PD-L1 on PBMCs, ECs, and fibroblasts exhibiting inhibitory effects on CD4+ cells(59).
Though genetically modified pigs show promise in prolonging xenograft survival, antibody-mediated acute humoral rejection and coagulation dysfunction(11–14, 28–30) have remained barriers. Furthermore, the morbidity and mortality associated with the conventional immunosuppression necessary for xenograft survival is nontrivial(60). Previous study has found that the activation of PECs can be induced by activated human platelets expressing CD154(20) indicating the critical role of CD40/CD154 pathway in xeno-specific T cell activation. Indeed, immunotherapies specifically targeting CD40/CD154 costimulatory molecules have resulted in dramatic prolongation of xenograft in primates(12–14, 28–29); however, blocking CD154/CD40 interaction with CD154 mAb is associated with thromboembolic complications in human and primates(31). The most recent study has demonstrated life-supporting cardiac xenograft for up to 195 days without significant thromboembolic events using monkey-specific anti-CD40 antibody or anti-CD154 PASylated Fab-based immunotherapy(13). Contrastingly, belatacept/rapamycin-based maintenance regimen in combination with depletional induction has been approved as replacement to calcineurin inhibitor in kidney transplantation. This novel regimen not only effectively prevents T cell–mediated acute allograft rejection with preserved cell-mediated immunity, but also inhibits formation of donor-specific alloantibody(34–35). We reasoned that this novel regimen may effectively prevent xenoreactive T cell–mediated rejection and xenoantibody formation when combined with therapies specifically targeting coagulation dysfunction.
In conclusion, this study demonstrates a lack of development of rapid cytokine-producing memory T cell responses in humans during initial interaction between T cells and PECs. Both purified naïve and memory cells with or without autologous CD14+ cells recognize and proliferate in response to PECs. We also define the phenotype of proliferating xenoreactive T cells in response to porcine endothelial xenoantigens with or without B7 CoB. Rapamycin effectively inhibits xenospecific T-cell proliferative responses by either directly interfering with T cells or altering porcine endothelial immunogenicity. We also find that inhibition of xenoreactive T-cell proliferation by porcine endothelium pretreated with rapamycin is independent of negative regulation of PD-1/PD ligand interaction. These findings may be relevant in further investigations of belatacept/rapamycin-based maintenance immunosuppressive regimen in xenotransplantation.
Supplementary Material
Figure S1. Analysis of xenogeneic mixed lymphocyte-endothelial cell reaction. VPD-450-labeled cells were stimulated with PECs, and unstimulated cells were used as controls. The proliferation is determined by the VPD-45 dilution after 6 days of culture. The gating strategy of analysis is shown in this panel.
Funding:
NIH 2UO1A1090956-11
Abbreviation
- PECs
porcine endothelial cells
- TM
memory T cells
- HAR
Hyperacute rejection
- FBS
Fetal Bovine Serum
- MLER
mixed lymphocyte-endothelial cell reaction
- CoBRR
costimulation blockade–resistant rejection
- mAbs
monoclonal antibodies
- CoB
costimulation blockade
- ICCS
intracellular cytokine staining
- HECs
human endothelial cells
- mTOR
mammalian target of rapamycin
- TNF-α
tumor necrosis factor-α
- IFN-γ
interferon-γ
- mAb
monoclonal antibody
- PBMCs
peripheral blood mononuclear cell
- TN
naïve T cells
- TEMRA
terminally differential effector memory T cells
- TEM
effector memory T cells
- TC
central memory T cells
Footnotes
Disclosure:
Conflict of interest: The authors have declared that no conflict of interest exists.
Reference
- 1.Sykes M, Sachs DH. Transplanting organs from pigs to humans. Sci Immunol 2019. Nov 1;4(41): eaau6298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dorling A, Fiesbeck K, Warrens A, Lechler R. Clinical xenotransplantation of solid organs. The Lancet 1997; 349:867–871. [DOI] [PubMed] [Google Scholar]
- 3.Calne RY. Organ transplantation between widely disparate species. Transplantation Proceedings 1970; 2:550. [PubMed] [Google Scholar]
- 4.Platt JL, Fischer RJ, Matas AJ, et al. Immunopathology of hyperacute xenograft rejection in a swine-to-primate model. Transplantation 1991; 52:214–220. [DOI] [PubMed] [Google Scholar]
- 5.Sandrin MS, McKenzie IF. Gal alpha (1–3)Gal, the major xenoantigen(s) recognized in pigs by human natural antibodies. Immuno Rev 1994; 141:169–190. [DOI] [PubMed] [Google Scholar]
- 6.Cooper DK, Human PA, Lexer G, et al. Effects of cyclosporine and antibody absorption on pig cardiac xenograft survival in the baboon. J Heart Transplant 1988; 7:238–246. [PubMed] [Google Scholar]
- 7.Leventhal JR, Dammasso AP, et al. Prolongation of cardiac xenograft survival by depletion of complement. Transplantation 1993; 55:865–866. [DOI] [PubMed] [Google Scholar]
- 8.Xu H, Gundry SR, Hill AC, et al. Prolonged discordant cardiac xenograft survival in newborn recipients. Circulation 1997; 96:364–367. [PubMed] [Google Scholar]
- 9.Xu H, Gundry SR, Hancock WW, et al. Prolonged discordant xenograft survival and delayed xenograft rejection in a pig-to-baboon orthotopic cardiac xenograft model. J Thorac Cardiovasc Surg 1998; 115:1342–1349. [DOI] [PubMed] [Google Scholar]
- 10.Candinas D, Belliveau S, Koyamada N, et al. T cell independence of macrophage and natural killer cell infiltration, cytokine production, and endothelial activation during delayed xenograft rejection. Transplantation 1996; 62:1739–1743 [DOI] [PubMed] [Google Scholar]
- 11.McCurry KR, Kooyman DL, Alvarado CG, et al. Human complement regulatory proteins protect swine-to-primate cardiac xenografts from humoral injury. Mat Med 1995; 5:423–427. [DOI] [PubMed] [Google Scholar]
- 12.Thompson P, Cardona K, Russell M, et al. Anti-CD40-based costimulation blockade enhances neonatal porcine islet survival in nonhuman primates. Am J Transplant 2011; 11:947–957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Langin M, Mayr T, Reichart B, et al. Consistent success in life-supporting porcine cardiac xenotransplantation. Nature 2018; 564:430–433. [DOI] [PubMed] [Google Scholar]
- 14.Adams AB, Kim SC, Martens GR, et al. Xenoantigen depletion and chemical immunosuppression can prolong renal xenograft survival. Ann Surg 2018; 268:564–573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kirk AD, Li RA, Kinch MS, et al. The human antiporcine cellular repertoire. In vitro studies of acquired and innate cellular responsiveness. Transplantation 1993; 55:924–931. [PubMed] [Google Scholar]
- 16.Rollins SA, Kennedy SP, Chodera AJ, et al. Evidence that activation of human T cells by porcine endothelium involves direct recognition of porcine SLA and costimulation by porcine ligands for LFA-1 and CD2. Transplantation 1994; 57:1709–1716. [PubMed] [Google Scholar]
- 17.Kim CH, Oh K, Kim DE, et al. , Both CD45RA+ and CD45RO+ human CD4+ T cells drive direct xenogeneic T-cell responses against porcine aortic endothelial cells. Xenotransplantation 2010; 17:224–243 [DOI] [PubMed] [Google Scholar]
- 18.Vallee I, Guillaumin JM, Thibault G, et al. Human T lymphocyte proliferative response to resting porcine endothelial cells results from an HLA-restricted, IL10-sensitive, indirect presentation pathway but also depends on endothelial-specific costimulatory factors. J Immunol 1998; 161:1652–1658. [PubMed] [Google Scholar]
- 19.Tadaki DK, Craighead N, Saini A, et al. Costimulatory molecules are active in the human xenoreactive T-cell response but not in natural killer-mediated cytotoxicity. Transplantation 2000; 70:162–167. [PubMed] [Google Scholar]
- 20.Xu H, Arnaud F, Tadaki DK, et al. Human platelets activate porcine endothelial cells through a CD154-dependent pathway. Transplantation 2001; 1858–1861. [DOI] [PubMed] [Google Scholar]
- 21.Bretscher P, Cohn M. A theory of self discrimination. Science 1970; 169:1042–1049. [DOI] [PubMed] [Google Scholar]
- 22.Kedl RM, Mescher MF. Qualitative differences between naïve and memory T cells make a major contribution to the more rapid and efficient memory CD8+ T cell response. J Immunol 1998; 161:1674–683. [PubMed] [Google Scholar]
- 23.Welsh R, and Selin L. No one is naïve: the significance of heterologous T-cell immunity. Nat. Rev. Immunol 2002, 2:417–426. [DOI] [PubMed] [Google Scholar]
- 24.Adams AB, Williams MA, Jones TR, et al. Heterologous immunity provides a potent barrier to transplantation tolerance. J Clin Invest 2003; 111:1887–1895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Espinosa J, Herr F, Tharp G, et al. CD57+ CD4 T cells underline belatacept-resistant allograft rejection. Am J Transplant 2016; 16:1102–1112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xu H, Perez SD, Cheeseman J, et al. The allo- and viral-specific immunosuppressive effects of belatacept, but not tacrolimus, attenuates with progressive T cell maturation. Am J Transplant 2014; 14:319–332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Lo DJ, Weaver TA, Stempora L, et al. Selective targeting of human alloresponsive CD8+ effector memory T cells based on CD2 expression. Am J Transplant 2011; 11:22–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shin JS, Kim JM, Kim JS, et al. , Long-term control of diabetes in immunosuppressed nonhuman primates (NHP) by the transplantation of adult porcine islets. Am J Transplant 2015; 11:2837–2850 [DOI] [PubMed] [Google Scholar]
- 29.Zhang T, Pierson 3rd RN, Azimzadeh AM. Update on CD40 and CD154 blockade in transplant models. Immunotherapy 2015; 8:899–911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kawai T, Andrews D, Colvin RB, Sachs DH, Cosimi B. Thromboembolic complications after treatment with monoclonal antibody against CD40 ligand. Nat Med 2000; 6:114. [DOI] [PubMed] [Google Scholar]
- 31.Kirk AD, Harlan DM. Reply to thromboembolic complications after treatment with monoclonal antibody against CD40 ligand. Nat Med 2000; 6:114. [DOI] [PubMed] [Google Scholar]
- 32.Vincenti F, Larsen CP, Alberu, et al. Three-year outcomes form BENEFIT, a randomized active-controlled, parallel-group study in adult kidney transplant recipient. Am J Transplant 2012; 12:210–217. [DOI] [PubMed] [Google Scholar]
- 33.Ferguson R, Grinyó J, Vincenti F, et al. Immunosuppression with belatacept-based, corticosteroid-avoiding regimens in de novo kidney transplant recipient. Am J Transplant 2011; 11:66–76. [DOI] [PubMed] [Google Scholar]
- 34.Kirk A, Guasch A, Xu H, et al. Renal transplantation using belatacept without maintenance steroids or calcineurin inhibitors. Am J Transplant. 2014; 14(5)1142–1151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schmitz R, Fitch ZW, Xu H, et al. Kidney transplantation using alemtuzumab, belatacept and sirolimus - five-year follow-up. Am J Transplant 2020; 12:3609–3619. [DOI] [PubMed] [Google Scholar]
- 36.Akondy RS, Monson ND, Miller JD, et al. The yellow fever virus vaccine induces a broad and polyfunctional human memory CD8+ T cell response. J Immunol 2009; 183:7919–7930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Picker LJ, Singh MK, Zdraveski Z, et al. Direct demonstration of cytokine synthesis heterogeneity among human memory/effector T cells by flow cytometry. Blood 1995; 86:1480–1419. [PubMed] [Google Scholar]
- 38.Xu H, Oluwole SF, Edwards NM, et al. In vitro evaluation of neonatal human immunity against the pig. J Thorac Cardiovasc Surg 1996; 111:920–929. [DOI] [PubMed] [Google Scholar]
- 39.Yang YG, Sykes M. Xenotransplantation: current status and a perspective on the future. Nature Reviews Immunology 2007; 7:519–531 [DOI] [PubMed] [Google Scholar]
- 40.Sallusto F, Lenig D, Forster R, et al. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 1999; 401:708–712. [DOI] [PubMed] [Google Scholar]
- 41.Xu H, Dhanireddy KK, Kirk AD. Human monocytes as intermediaries between allogeneic endothelial cells and allospecific T cells: a role for direct scavenger receptor-mediated endothelial membrane uptake in initiation of alloimmunity. J Immunol 2006; 176:750–761 [DOI] [PubMed] [Google Scholar]
- 42.Brossay A, Harang S, Herault O, Bardos P, Watier H. The active role played by xenogeneic endothelial cells in the indirect presentation pathway is not lymphocyte-trans-co-stimulation. Transpl Int 2005; 17:787–794 [DOI] [PubMed] [Google Scholar]
- 43.Murray AG, Khodadoust MM, Pober JS, et al. Porcine aortic endothelial cells activate human T cells: direct presentation of MHC antigens and costimulation by ligands for human CD2 and CD28. Immunity 1994; 1:57–63. [DOI] [PubMed] [Google Scholar]
- 44.Weaver TA, Charafeddine AH, Agarwal A, et al. Alefacept promotes co-stimulation blockade based allograft survival in nonhuman primates. Nat Med 2009; 15:746–749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zha YY, Blank C, Gajewski TF. Negative regulation of T-cell function by PD-1. Crit Rev Immunol 2004: 24:229–237 [DOI] [PubMed] [Google Scholar]
- 46.Takahashi T, Hsiao HM, Tanaka S, Li W, Higashikubo R, Scozzi D, et al. PD-1 expression on CD8+ T cells regulates their differentiation within lung allografts and is critical for tolerance induction. Am J Transplant 2018; 18:216–225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wang L, Han R, Hancock WW. Programmed cell death 1 (PD-1) and its ligand PD-L1 are required for allograft tolerance. Eur J Immunol 2007; 37:2983–2990 [DOI] [PubMed] [Google Scholar]
- 48.Calvo V, Crews CM, Vik TA, Bierer BE. Interleukin 2 stimulation of p70 S6 kinase activity is inhibited by the immunosuppressant rapamycin. Proc Natl Acad Sci USA 1992; 89:7571–7575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wells AD, Li XC, Li Y, Walsh MC, Zheng XX, Wu Z, et al. Requirement for T-cell apoptosis in the induction of peripheral transplantation tolerance. Nat Med 1999; 11:1303–1307 [DOI] [PubMed] [Google Scholar]
- 50.Yamamoto T, Hara H, Foote J, Wang L, Li Q, Klein EC, et al. Life-supporting kidney xenotransplantation from genetically engineered pigs in baboons: A comparison of two immunosuppressive regimens. Transplantation 2019; 103:2090–2104 [DOI] [PubMed] [Google Scholar]
- 51.Wang C, Qin L, Manes TD, Kirkiles-Smith NC, Tellides G, Pober JS. Rapamycin antagonizes TNF induction of VCAM-1 on endothelial cells by inhibiting mTORC2. J Exp Med 2014; 211:395–404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cantaluppi V, Biancone L, Romannazzi GM, Figliolini F, Beltramo S, Ninniri MS, et al. Antiangiogenic and immunomodulatory effects of rapamycin on islet endothelium: relevance for islet transplantation. Am J Transplant 2006; 6:2601–2611 [DOI] [PubMed] [Google Scholar]
- 53.Buhler L, IIIigens BM-W, Nadazdin O, Tena A, Lee S, Sachs DH, Cooper DKC, Benichou G. Persistence of indirect but not direct T cell xenoresponses in baboon recipients of pig cell and organ transplants. Am J Transplant 2016, 16:1917–1922 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Wilhite T, Ezzelarab C, Hara H, Long C, Ayares D, Cooper DKC, Ezzelarab M. The effect of gal expression on pig cells on the human T-cell xenoresponses. Xenotransplantation 2012; 19:56–63 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Iwase H, Ekser B, Satyananda V, Bhama J, Hara H, Ezzelarab M, et al. Pig-to-baboon heterotopic heart transplantation –exploratory preliminary experience with pigs transgenic for human thrombomodulin and comparison of three costimulation blockade-based regiments. Xenotransplantation 2015; 22:211–220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cantaluppi V, Biancone L, Romannazzi GM, Figliolini F, Beltramo S, Ninniri MS, et al. Antiangiogenic and immunomodulatory effects of rapamycin on islet endothelium: relevance for islet transplantation. Am J Transplant 2006; 6:2601–2611 [DOI] [PubMed] [Google Scholar]
- 57.Fantus D, Thomson AW. Evolving perspectives of mTOR complexes in immunity and transplantation. Am J Transplant. 2011; 15:891–902. [DOI] [PubMed] [Google Scholar]
- 58.Wang C, Yi T, Qin L, Maldonado RA, von Andrian UH, Kulkarni S, Tellides G, Pober JS. Rapamycin-treated human endothelial cells preferentially activate allogeneic regulatory cells. J Clin Invest 2013; 123:1677–1693 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Buermann A, Petkov S, Petersen B, et al. Pigs expressing the human inhibitory ligand PD-L1 (CD274) provide a new source of xenogeneic cells and tissue with low immunogenic properties. Xenotransplantation 2018; 25:e12387. [DOI] [PubMed] [Google Scholar]
- 60.Cozzi E, Vial C, Ostlie D, Farah B, Chavez G, Smith KGC, et al. maintenance triple immunosuppression with cyclosporin A, mycophenolate sodium and steroids allows prolonged survival of primate recipients of hDAF porcine renal xenografts. Xenotransplantation 2003; 10:300–310 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. Analysis of xenogeneic mixed lymphocyte-endothelial cell reaction. VPD-450-labeled cells were stimulated with PECs, and unstimulated cells were used as controls. The proliferation is determined by the VPD-45 dilution after 6 days of culture. The gating strategy of analysis is shown in this panel.


