Abstract
Background
The worldwide epidemics of diseases as dengue and Zika have triggered an intense effort to repurpose drugs and search for novel antivirals to treat patients as no approved drugs for these diseases are currently available. Our aim was to screen plant-derived extracts to identify and isolate compounds with antiviral properties against dengue virus (DENV) and Zika virus (ZIKV).
Methods
Seven thousand plant extracts were screened in vitro for their antiviral properties against DENV-2 and ZIKV by their viral cytopathic effect reduction followed by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method, previously validated for this purpose. Selected extracts were submitted to bioactivity-guided fractionation using high- and ultrahigh-pressure liquid chromatography. In parallel, high-resolution mass spectrometric data (MSn) were collected from each fraction, allowing compounds into the active fractions to be tracked in subsequent fractionation procedures. The virucidal activity of extracts and compounds was assessed by using the plaque reduction assay. EC50 and CC50 were determined by dose response experiments, and the ratio (EC50/CC50) was used as a selectivity index (SI) to measure the antiviral vs. cytotoxic activity. Purified compounds were used in nuclear magnetic resonance spectroscopy to identify their chemical structures. Two compounds were associated in different proportions and submitted to bioassays against both viruses to investigate possible synergy. In silico prediction of the pharmacokinetic and toxicity (ADMET) properties of the antiviral compounds were calculated using the pkCSM platform.
Results
We detected antiviral activity against DENV-2 and ZIKV in 21 extracts obtained from 15 plant species. Hippeastrum (Amaryllidaceae) was the most represented genus, affording seven active extracts. Bioactivity-guided fractionation of several extracts led to the purification of lycorine, pretazettine, narciclasine, and narciclasine-4-O-β-D-xylopyranoside (NXP). Another 16 compounds were identified in active fractions. Association of lycorine and pretazettine did not improve their antiviral activity against DENV-2 and neither to ZIKV. ADMET prediction suggested that these four compounds may have a good metabolism and no mutagenic toxicity. Predicted oral absorption, distribution, and excretion parameters of lycorine and pretazettine indicate them as candidates to be tested in animal models.
Conclusions
Our results showed that plant extracts, especially those from the Hippeastrum genus, can be a valuable source of antiviral compounds against ZIKV and DENV-2. The majority of compounds identified have never been previously described for their activity against ZIKV and other viruses.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12985-022-01751-z.
Keywords: Antiviral, Dengue virus, Zika virus, Alkaloids, Natural products, Plant extract, Hippeastrum, Virucidal, Bioactivity-guided fractionation, Compounds
Introduction
Zika virus (ZIKV) and Dengue virus (DENV), members of the Flaviviridae family, are arboviruses of great importance in human Public Health worldwide. Primarily asymptomatic, ZIKV and DENV infected patients may have mild clinical symptoms such as fever, headache, and rashes. However, ZIKV can also cause severe neurologic manifestations in fetuses and newborns after congenital infection and Guillain-Barré syndrome in adults [1–3]. There are no currently approved vaccines for ZIKV infection [4]. This scenario is worsened by the imminent risk of DENV infections, which, although it already has a licensed vaccine, CYD-TDV (Dengvaxia), its indication is not well established. CYD-TDV efficacy varies according to DENV serotype, serostatus (with detectable dengue antibodies), and age [5, 6]. According to the World Health Organization (WHO), Dengvaxia is recommended for people living in regions where dengue is endemic, from 9 to 45 years old, and who have had at least one previous DENV infection [5]. Unfortunately, there are an estimated 100–400 million dengue infections each year, and severe dengue is a potentially deadly complication due to plasma leaking, fluid accumulation, respiratory distress, severe bleeding, or organ impairment. Furthermore, without proper medical care, severe dengue can reach up to 10% case fatality [5, 7, 8]. As for Zika, there are no approved antivirals for dengue treatment. Treatments are palliative and mainly directed toward alleviating symptoms, such as fever and rash, by administering antipyretics, antihistamine, and fluids against dehydration [9–11]. Therefore, Zika and dengue outbreaks have been triggered intense efforts to drug repurposing and identification of novel antiviral agents for specific treatment [12, 13]. Different approaches and methodologies have been used for drug repurposing to find antivirals against ZIKV and DENV, such as testing specific compounds with known antiviral activity in other virus models and screening of libraries composed of hundreds of bioactive molecules, many of them already approved for human use. These molecules target viral and cellular components, including nucleosides analogues, nucleoside synthesis inhibitors, drugs targeting viral enzymes, anticancer and anti-inflammatory molecules, antibiotics, antiparasitics, among others [11, 14].
Compounds derived from plants have long been used worldwide as a source of medicines since ancient times until today [15, 16]. Indeed, natural products derived from plants and other living organisms represent a huge reservoir of bioactive chemical diversity and have been successful sources of potential drug leads to new therapeutic agents for many human diseases, including those caused by viruses [17–19]. Our strategy was to screen large numbers of plant extracts for their anti-flavivirus activities to identify compounds that exhibit antiviral activity against DENV and ZIKV. Other studies have shown that higher plants represent a recognized source of antiviral drug leads [19, 20]. Thus, the aim of our study was threefold: (1) to identify extracts of plants collected in distinct Brazilian biomes as potential sources of antiviral compounds; (2) to identify and purify the active compounds present in the selected extracts and (3) assess the antiviral activity of the purified compounds against DENV-2 and ZIKV.
Methods
Reagents
Solvents and reagents used in analytical methods were purchased from Merck (Brazil). 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT), cell culture media, and supplements were purchased from Thermo (USA). Trypsin–EDTA and carboxymethyl cellulose were purchased from Sigma-Aldrich (USA). Formic acid (> 98%) was purchased from Fluka (USA). Lycorine (HCl Salt Form) was purchased from BOC Sciences (USA).
Cells, viruses, and Interferon-α 2B
C6/36 (ATCC #CRL-1660) cells derived from larvae of Aedes albopictus mosquito were used to obtain working stock of DENV-2 and ZIKV. Baby Hamster Kidney 21 cells (BHK-21; ATCC #CCL-10) and Vero cells (ATCC #CCL-81) were used, respectively, for virus titration and antiviral assays activity against DENV-2 and ZIKV. The ZIKV used in this study (PE243/2015; GenBank: KX197192.1) was kindly given by Dr. Marli Tenório Cordeiro from Aggeu Magalhães Institute, Fiocruz Pernambuco, Brazil. The working stock of low passage ZIKV (7.4 × 108 PFU/ml) was prepared and stored at − 80 °C in our lab at the René Rachou Institute, Fiocruz Minas, Brazil. ZIKV stock titration was performed by plaque formation assay in Vero cells. The DENV-2 sample was provided by Dr. Luiz Tadeu Figueiredo from University of São Paulo, Ribeirão Preto, Brazil. DENV-2 was multiplied in C6/36 cells and titrated by plaque formation assay in BHK-21 cells. Interferon-α 2B (INREC, Uruguay) was used as a positive control in antiviral assays.
Plant material
Plant specimens were collected in different Brazilian biomes by Dr. Marcos Sobral, from Universidade Federal de São João del-Rei/MG. Hippeastrum glaucescens (H. glaucescens) and Hippeastrum puniceum (H. puniceum) were also collected by Carlos Alberto Ferreira Junior at Fundação Zoobotânica de Belo Horizonte-MG. Both researchers were also responsible for the identification of the plant specimens. Voucher codes are shown in Table 1.
Table 1.
Plant extracts with anti-DENV-2 and anti-ZIKV activity: EC50, CC50, SI and virucidal activity
| Family | Plant species | Voucher | Part | Extract | EC50 DENV-2 | CC50 BHK-21 | SI | Virucidal activity | EC50 ZIKV | CC50 Vero | SI | Virucidal activity |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amaryllidaceae | Hippeastrum glaucescens | HUFSJ-2914 | Flower | 13356 | 27.9 | > 100 | > 3.6 | No | 56.8 | > 100 | > 1.8 | No |
| Amaryllidaceae | Hippeastrum glaucescens | HUFSJ-2914 | Bulb | 13358 | 3.9 | 8.6 | 2.2 | No | 8.9 | 53.0 | 5.9 | No |
| Amaryllidaceae | Hippeastrum sp. | HUFSJ-3007 | Root | 13418 | 27.5 | 76.2 | 2.8 | No | 41.8 | > 100 | > 2.4 | No |
| Amaryllidaceae | Hippeastrum glaucescens | HUFSJ-4494 | Bulb | 17006 | 6.2 | 8.4 | 1.4 | No | 10.6 | 44.1 | 4.2 | No |
| Amaryllidaceae | Hippeastrum puniceum | BHZB-12069 | Flower | 17007 | 3.1 | 8.7 | 2.8 | No | 6.8 | 18.2 | 2.8 | No |
| Amaryllidaceae | Hippeastrum puniceum | BHZB-12069 | Root | 17010 | 3.1 | 5.9 | 1.9 | No | 5.5 | 16.5 | 3.0 | No |
| Amaryllidaceae | Hippeastrum puniceum | BHZB-12069 | Bulb | 17011 | 0.8 | 1.3 | 1.7 | No | 1.1 | 4.2 | 3.8 | No |
| Annonaceae | Duguetia furfuracea | HUFSJ-2345 | Fruit | 12829 | 9.8 | > 100 | > 10.2 | + | 34.7 | > 100 | > 2.9 | + |
| Asteraceae | Chrysolaena sp. | HUFSJ-2686 | Flower | 13211 | 8.9 | 16.4 | 1.8 | + | 35.1 | > 100 | > 2.8 | + |
| Erythroxylaceae | Erythroxylum sp. | HUFSJ-2580 | Stem | 13023 | 9.1 | 41.1 | 4.5 | + | 29.3 | > 100 | > 3.4 | + |
| Fabaceae | Chamaecrista sp. | HUFSJ-2193 | Fruit | 11279 | 2.9 | 35.4 | 11.9 | + | 53.7 | > 100 | > 1.9 | + |
| Fabaceae | Chamaecrista sp. | HUFSJ-2193 | Stem | 11280 | 7.8 | > 100 | > 20.5 | No | > 100 | > 100 | > 1.0 | No |
| Malpighiaceae | Banisteriopsis argyrophylla | HUFSJ-2201 | Stem | 11298 | 12.5 | 36.6 | 2.9 | + | 48.9 | > 100 | > 2.0 | + |
| Malpighiaceae | Banisteriopsis sp. | HUFSJ-1995 | Stem | 11117 | 34.8 | 80.5 | 2.3 | + | 63.2 | > 100 | > 1.6 | + |
| Malpighiaceae | Byrsonima coccolobifolia | HUFSJ-2683 | Flower | 13203 | 9.4 | 26.7 | 2.8 | + | 43.4 | > 100 | > 2.3 | + |
| Primulaceae | Ardisia sp. | HUFSJ-2368 | Stem | 12875 | 11.8 | 31.1 | 2.6 | + | 43.9 | > 100 | > 2.3 | + |
| Rubiaceae | Palicourea sp. | HUFSJ-2360 | Flower | 12860 | 16.5 | > 100 | > 6.0 | + | 62.4 | > 100 | > 1.6 | + |
| Sapindaceae | Urvillea sp. | HUFSJ-1997 | Stem | 11123 | 23.8 | 31.5 | 1.3 | + | 43.1 | > 100 | > 2.3 | + |
| Sapindaceae | Cupania sp. | HUFSJ-1992 | Leaf | 11107 | 19.5 | 38.7 | 2.0 | + | 52.7 | > 100 | > 1.9 | + |
| Sapindaceae | Cupania sp. | HUFSJ-1992 | Stem | 11108 | 17.8 | 47.9 | 2.7 | + | 21.6 | > 100 | > 4.6 | + |
| Ochnaceae | Ouratea sp. | HUFSJ-2555 | Flower | 12969 | 5.2 | 51.7 | 9.9 | + | 26.2 | > 100 | > 3.8 | + |
EC50: Half maximum effective concentration; CC50: Half maximum cytotoxic concentration; SI: Selectivity Index = ratio CC50/EC50; ( +) positive virucidal activity
Preparation of extracts
In brief, fresh plant parts were separated, cut into small pieces, and approximately 20 g placed in a 50 ml flask. The flask was then filled with absolute ethanol (Merck, Brazil) and kept at room temperature in the dark for at least one week. The extract was filtered and dried under vacuum at 45 °C. Stock solutions of the extracts at 20 mg/ml were prepared using 90% dimethyl sulfoxide (DMSO) and stored at –20 °C. For isolation of active compounds, about 2 kg of H. puniceum bulbs were used.
Validation of the MTT method for antiviral screening against ZIKV and DENV-2
The validation of the MTT method [21–23] for High Throughput Screening (HTS) of antivirals against ZIKV and DENV was performed in accordance with the Assay Guidance Manual published by the National Institutes of Health (NIH) [24]. Briefly, tests were made with three levels of signals as follows: high signal (H) for cell control, medium signal (M) for antiviral control and, low signal (L) for virus infection control. the MTT assays were done in sets of 96-well plates containing positive and negative controls (Additional file 1: Fig. S1). For the assays with ZIKV and DENV-2, 1.0 × 104 Vero cells per well and 8.0 × 103 BHK-21 cells per well, respectively, were seeded in 96-well plates and incubated for 24 h in MEM with 5% FBS. We used 200 µL of MEM with 0.25% DMSO for the high signal wells (H); 100 µL of ZIKV or DENV-2 suspension at m.o.i. of 2 and 100 µL of IFN-α 2b (100 IU/ml) for the medium signal (M); and for low signal (L), 100 µL of ZIKV or DENV-2 suspension at m.o.i. of 2 were added 100 µl of MEM medium. After three days post-infection (p.i.) with DENV-2 and 4 days p.i. with ZIKV, the reduction of viral CPE was verified by optic microscopy using a scoring system as described [23] and by the MTT method. The evaluation of the test and the variability between wells and between plates was done using the software available at the NIH website [24].
Screening of antiviral activity by cytopathic effect reduction and by the MTT method
Plant extracts were screened for their cytotoxic and anti-ZIKV and anti-DENV activities in Vero cells and BHK-21, respectively, in 96-well culture plates by observing the reduction of the viral cytopathic effect (CPE) [23] and then measured by the MTT method [21, 22]. The crude extracts were dissolved in 4% DMSO aqueous solution, and their concentration was normalized to 50 µg/ml with Eagle's minimum essential medium (MEM) supplemented with 2% fetal bovine serum (FBS). Briefly, 8 × 103 of BHK-21 cells or 1 × 104 of Vero cells per well were seeded in a 96-well plate and incubated for 24 h at 37 °C and 5% CO2 atmosphere. Extract solution and virus suspension, at a multiplicity of infection (m.o.i.) of 2, for both viruses, were simultaneously added on 70% confluent monolayers. Each extract was tested in duplicate. Controls of untreated infected and untreated uninfected cells incubated with or without DMSO were included. Interferon-α 2B, in concentrations of 500 and 1000 IU/mL, was used as a positive antiviral control in all antiviral assays. After three days post-infection (p.i.) with DENV-2 and 4 days p.i. with ZIKV, the reduction of viral CPE was verified by optic microscopy using a scoring system as described by Kudi and Myint (1999) [23] and by the MTT method. Briefly, the supernatants were removed, and 30 µl of a stock solution of MTT (2 mg/ml) dissolved in phosphate-buffered saline were added to each well. After 90 min of incubation at 37 °C, 130 µl of DMSO were added to each well, and plates were homogenized for 5 min at 500 rpm. The absorbance values of each reaction were measured in an ELISA reader (Spectra Max, Molecular Devices, USA) at λ540 nm. Extracts were considered active when they showed at least 50% of protection or reduction of the viral CPE by at least 50%. Cytotoxicity (CC50) and anti-ZIKV/DENV-2 (CE50) experiments were performed in duplicate and repeated at least three times.
Determination of CC50
The half-maximal cytotoxic concentration (CC50) was calculated as the extract/compound concentration at which only 50% of cells were alive, in comparison with non-treated cell controls. Compounds and selected extracts that tested positive for anti-ZIKV or anti-DENV-2 activity at the screening step had their CC50 determined by using an 8‐point dose range in 96 well microplate containing Vero or BHK-21 cell monolayers. Cell viability was observed by optic microscopy and measured by the MTT method as described. The experiments were performed in duplicate and repeated at least three times.
Determination of EC50
Viral CPE and cell viability were observed by optic microscopy and measured by the MTT method as previously described. The half-maximal effective concentration (EC50) was calculated as the extract/compound concentration at which only 50% of cells were alive, in comparison with non-infected cell controls. Compounds and extracts that tested positive for anti-ZIKV or anti-DENV-2 activity at the screening step had their EC50 determined using an 8‐point dose range in 96 well microplates in Vero cells or BHK-21 monolayers, respectively. The experiments were performed in duplicate and repeated at least three times.
Virucidal mechanism of action
The virucidal activity of extracts and compounds against ZIKV and DENV was assessed by plaque reduction assay [25] in Vero and BHK-21 cells, respectively, in 24 well microplates. Prior viral adsorption, equal volumes of viral suspension and extract or compound dilutions, corresponding to its EC50, EC50/2, and two times EC50, were mixed and incubated for 60 min at 37 °C and 5% CO2 atmosphere. Virus suspension without the extract/compound was included as a control. Surviving infectious viral particles were assessed by viral plaque formation assay. Briefly, the infected monolayers were overlaid with 1.5% carboxymethyl cellulose containing MEM with 2% FBS and incubated for five days. After that, cell monolayers were fixed with 10% formaldehyde and stained with 1% crystal violet 1% in an 30% ethanol aqueous solution. Extract and compounds were considered virucidal when their EC50 reduced at least by 90% the number of virus plaques compared to the untreated virus control [26, 27].
Bioassay-guided chromatographic fractionation
Selected extracts were fractionated in a Nexera UHPLC-system (Shimadzu, Japan) hyphenated to a MaXis ETD high-resolution ESI-QTOF mass spectrometer (Bruker, Germany) controlled by the Compass 1.7 software package (Bruker, Germany). Samples were diluted to final 5 mg/ml, and 1–3 µl (5–15 µg) injected on a Shim-Pack XR-ODS-III column (C18, 2.2 µm, 2.0 × 150 mm) (Shimadzu, Japan) at 40 °C under a flow rate of 400 μl/min. An additional identical run was performed with injection of 10 μl (50 µg). Instead of being sent to the mass spectrometer, the column effluent was collected in 80 wells of a polypropylene microtiter plate (200 µl/well/fraction). The mobile phases A and B (0.1% formic acid in distilled water and acetonitrile, respectively) formed an eluent gradient of initial 5 min 5% B, a linear gradient to 100% B in 40 min, and hold at 100% B for 5 min. The mass spectra were acquired in positive mode at a spectra rate of 2–5 Hz. Ion-source parameters were set to 500 V endplate offset, 4500 V capillary voltage, 3.0 bar nebulizer pressure, 8 l/min, and 200 °C dry gas flow and temperature, respectively. Data-dependent fragment spectra were recorded using a collision energy range between 15 and 60 eV. Ion cooler settings were optimized for an m/z 40–1000 range using a calibrant solution of 1 mM sodium formate in 50% 2-propanol. Mass calibration was achieved by initial ion-source infusion of 20 µl calibrant solution and post-acquisition recalibration of the raw data. Compound detection was performed by chromatographic peak dissection with subsequent formula determination according to exact mass and isotope pattern (MS1). Putative identification was based on the comparison of compound fragment spectra (MS2) with reference spectra of an in-house database of standard compounds, the public spectra database MassBank [28] as well as in silico fragment spectra generated from the Universal Natural Product Database (UNPD-ISDB) [29]. The collected fractions were dried in a vacuum centrifuge at 45 °C before being used in the bioassays. Compounds in the active fractions were identified by their retention time, exact m/z, and MS/MS fragmentation. These features were then used to track these compounds during the extract's preparative fractionation steps, aiming to isolate the active compounds for structural elucidation and further bioassays. The antiviral activity of all fractions was accessed using the bioassays with DENV-2 or ZIKV by the MTT method as described. IFN alpha 2B and the original crude extract were positive controls for antiviral activity.
Nuclear magnetic resonance analysis
Mono and bidimensional 1H and inversed detection 13C NMR experiments were run on a Bruker Avance 400 MHz spectrometer (Bruker Daltonics, Germany) using TMS as an internal standard. Samples were dissolved in chloroform-d. The experiments were run at 27 °C. Data were analysed using TopSpin software from Bruker.
Synthesis of Lycorine mono and di-acetylated derivatives
The methodology described by Wang et al. [30] was used. The chemical structures of the mono and di-acetylated derivatives were confirmed by comparing their MS and NMR spectral data with those described by the mentioned authors. The compounds were dissolved in DMSO and assayed at 0.5% DMSO final concentration.
Investigation of pretazettine and lycorine synergism against DENV-2 and ZIKV
The antiviral effect of the association of lycorine and pretazettine against DENV-2 and ZIKV was evaluated in vitro by a modified isobologram method [31, 32]. The results were analysed by nonlinear regression in the GraphPad Prism 5 software (GraphPad Software, Inc.). The EC50 and CC50 of each combination were assessed by the MTT method as described previously. Thus, two EC50 and CC50 for each of the five combination curves were calculated separately using the known concentration ratios of lycorine and pretazettine. The fractional inhibitory concentration of lycorine (FICL) and pretazettine (FICP) were calculated for each point, i.e. [EC50 when in combination]/[EC50 of drug alone]. The sum of the FIC (ΣFIC) with the equation ΣFIC = FIC lycorine + FIC pretazettine was calculated [33]. From the median of the sum of all FIC (xΣFIC), the fractional inhibitory concentration index (FICI) value was obtained and used to classify the interaction between lycorine and pretazettine. There would be synergy when xΣFIC ≤ 0.5; indifference when xΣFIC between 0.5 and ≤ 4 or antagonism if xΣFIC was ≥ 4 [33]. Isobolograms were built by plotting the FIC of lycorine vs. pretazettine. The ratio reveals the effect of their association on toxicity and antiviral activity [31, 32, 34, 35].
ADMET prediction
Pharmacokinetics and toxicity properties of compounds were predicted using the pkCSM platform [36]. pkCSM is a comprehensive web-based, user-friendly resource that allows for the rapid screening of 30 different properties regarding absorption, distribution, metabolism, excretion, and toxicity (http://biosig.unimelb.edu.au/pkcsm). pkCSM relies on the concept of graph-based signatures [37, 38] to model small molecule physicochemical and geometrical properties. These are used as evidence to train machine learning-based models, which have been invaluable to predict and optimize ligand pharmacokinetic properties [39]. Compounds structures were provided to the pkCSM web server as their canonical SMILES representation.
Data analysis and selectivity index
Bioassay data were normalized to untreated uninfected cells and the EC50 and CC50 values were obtained using GraphPad Prism 5 based on nonlinear logistic regression of the experimental dose–response curves. The values correspond to three independent assays' average and standard deviation with at least 8 sample distinct concentrations. The CC50 and EC50 values ratio was used as a selectivity index (SI). The extracts and compounds with higher SI values were considered the most promising and selected for further investigation if available in sufficient quantities.
Results
The MTT method is a reliable tool for antiviral high-throughput screening (HTS) against DENV and ZIKV
The results of the validation of the MTT method for antiviral HTS against ZIKV in Vero cells and DENV-2 in BHK-21 cells are shown in Additional file 1: Figs. S2 and S3. We observed no effects of systematic errors, which could be verified if the difference of over 20% in the values of each column or observing the distribution of points (each point is equivalent to the reading of a well). We noticed a considerable edge effect in both ZIKV and DENV assays. Indeed, edge effects are usually due to evaporation in the wells on the sides of the plate, especially when the assay requires incubation for very long periods. To reduce this variability, the incubation time was reduced from 96 to 72 h. Finally, for the assessment of the MTT assay and the variability between plates, the acceptance criterion is that the L or MIN signal (infected and untreated cells) should not show any significant change. Furthermore, the coefficient of variation between the H or MAX signals (uninfected and untreated cells) and between the M or MED signals (infected cells and treated with the positive control) must not exceed 20%, and the Z' factor must be above of 0.4 (data not shown). All criteria listed in the statistical analysis were met. Finally, it is worth mentioning that we demonstrated that the CE50 values of several extracts obtained from the assessment of viral CPE protection against DENV-2 and ZIKV were similar to those obtained by the MTT assay (data not shown).
Antiviral, EC50, CC50 , and virucidal activities of crude plant extracts
A total of 7,000 extracts obtained from plants occurring in different Brazilian biomes were screened at 20 µg/ml on cells infected with DENV-2. A smaller number (1,000 extracts) were also tested against ZIKV. As a result, 21 plant extracts from ten plant families displayed consistently in vitro activity against DENV-2 and ZIKV, and their EC50, CC50, and virucidal properties were determined (Table 1). Among the 21 selected extracts, 12 exhibited good DENV-2 activity, disclosing EC50 values < 10 µg/ml, while nine had a moderate activity with an EC50 ranging from 10 to 35 µg/ml. Four extracts exhibited good anti-ZIKV activity and showed EC50 values < 10 µg/ml, while 17 had a moderate anti-ZIKV activity with an EC50 ranging from 10 to 100 µg/ml. The extracts of the Amaryllidaceae family were cytotoxic in very low concentrations, with CC50 values < 10 µg/ml, while extracts from other families presented CC50 > 10 µg/ml. The SI values of the extracts ranged from 1.3 to > 20.5 µg/ml for DENV-2 and 1.0 to 5.9 for ZIKV. Afterward, inhibition of infection due to virucidal effect on DENV and ZIKV particles was tested using at least three non-cytotoxic concentrations. Thirteen extracts were considered virucidal since they caused a 90% or higher reduction in plaque numbers (Table 1).
Bioassay-guided fractionation
The choice of extracts for bioassay-guided fractionation was based on their antiviral activity and sample availability. Thirteen active extracts, eight virucidal and five non-virucidal (listed in Tables 1, 2) were subjected to bioactivity-guided fractionation to identify their antiviral compounds. The extracts from fruits of Duguetia furfuracea (Annonaceae), stems of Banisteriopsis argyrophylla (Malpighiaceae), and flowers of Palicourea sp. (Rubiaceae) did not yield fractions with significant activity against ZIKV and DENV-2 (Table 2). The flower from Ouratea sp. (Ochnaceae) afforded an extract for which the activity appeared only in the first experiment. The fractionation of this extract yielded highly polar fractions of the UHPLC RP-18 chromatography; however, no useful MS or MS/MS spectra could be obtained. In the active fractions of stem extract of Chamaecrista sp. (Fabaceae) the presence of agarobiose and leuhistin was proposed based on the HRMS/MS data. Similarly, fragment spectra indicated the presence of leuhistin and plumbagin in the active fractions of its fruit extracts. Extracts of H. glaucescens (Amaryllidaceae) afforded several active fractions, from which HRMS/MS data suggested the presence of pancratine (flowers), pretazettine (flowers and bulbs), rutine (flower), lycorine (flowers and bulbs), pseudolycorine, pancracine and nangustine (bulbs) (Table 2). HRMS/MS spectra of the active fractions from the extracts of H. puniceum suggested the presence of incartine and pseudolycorine from its flowers. The MS/MS spectra of root extract were compatible with lycoramine, narcissidine acetate, and 6-hydroxy-hippeastrine. narciclasine, kalbreclasine, NXP, pseudolycorine, lycoranine E, lycoranine C, N-norlycoramine, incartine, lycorine, crisarnine, acetylnerbowdine, and narcissidine acetate were detected in the bulbs extract of H. puniceum (Table 2). Lycorine, narciclasine, and NXP were isolated, and their identity was confirmed based on their NMR spectra or by comparison with a commercial standard. Pretazettine, as well, was compared with 1H NMR data from those described by Moraes [40]. Commercial rutine was compared with the MS/MS spectra of its respective flower extract fraction of H. glaucescens.
Table 2.
Compounds identified by UHPLC-HRMS/MS in active extract fractions against DENV-2 and ZIKV
| Extract number | Plant family | Species | Part | Chemical compound |
|---|---|---|---|---|
| 13356 | Hippeastrum glaucescens | Flower | Pancratine, pretazettine, rutine and lycorine | |
| 13358 | Amaryllidaceae | Hippeastrum glaucescens | Bulb | Lycorine and pretazettine |
| 13418 | Amaryllidaceae | Hippeastrum sp. | Root | Lycorine |
| 17006 | Amaryllidaceae | Hippeastrum glaucescens | Bulb | Lycorine, pseudolycorine, pancracine, nangustine and pretazettine |
| 17007 | Amaryllidaceae | Hippeastrum puniceum | Flower | Pseudolycorine, incartine |
| 17010 | Amaryllidaceae | Hippeastrum puniceum | Root | Lycorine, lycoramine, narcissidine acetate and 6-hydroxy-hippeastrine |
| 17011 | Amaryllidaceae | Hippeastrum puniceum | Bulb | Narcissidine acetate, narciclasine, lycorine, kalbreclasine, lycoranine E, lycoranine C, acetylnerbowdine, incartine, crisarnine, pseudolycorine, N-norlycoramine and narciclasine-4-O-β-D-xylopyranoside (NXP) |
| 12829 | Annonaceae | Duguetia furfuracea | Fruit | No active fractions |
| 11280 | Fabaceae | Chamaecrista sp. | Stem | Agarobiose and leuhistin |
| 11279 | Fabaceae | Chamaecrista sp. | Fruit | Leuhistin and plumbagin |
| 11298 | Malpighiaceae | Banisteriopsis argyrophylla | Stem | No active fractions |
| 12860 | Rubiaceae | Palicourea sp. | Flower | No active fractions |
| 12969 | Ochnaceae | Ouratea sp. | Flower | Mix of polar or ionizable compounds |
Anti-DENV-2 and anti-ZIKV activity of rutine, lycorine and its derivatives, pretazettine, narciclasine and NXP
Among the 22 compounds detected in the bioactive fractions, three were isolated and identified: pretazettine, narciclasine, and NXP. Rutine and lycorine were acquired from commercial sources. Lycorine derivatives were synthesized in our lab. These compounds were tested for their antiviral and cytotoxic properties. Rutine showed low toxicity against BHK-21 and Vero cells, with CC50 of > 300 µM and no antiviral activity against DENV-2 and ZIKV (Table 3). Pretazettine showed antiviral activity against DENV-2 (EC50 of 0.8 µM and SI of 6.8) and ZIKV (EC50 of 1.9 µM and SI of 3.8) with CC50 of 5.4 µM for BHK-21 cells and 7.2 µM for Vero cells. Narciclasine, isolated from H. puniceum, showed anti-DENV and anti-ZIKV activity at very low concentrations, with EC50 of 0.02 µM for both viruses, but also a CC50 in the low micromolar range (0.09 µM for BHK-21 and 0.12 µM for Vero cells), resulting in SI values of 4.5 and 6.0 for DENV-2 and ZIKV, respectively. Its sugar conjugate, narciclasine-4-O-β-D-xylopyranoside or NXP, described recently by Katoch et al. [41], was much less active against these viruses (EC50 = 7.9 µM) and less toxic to BHK-21 and Vero cells (CC50 of 39.3 µM and 51.8 µM, respectively) (Table 3). Lycorine was active against DENV-2 (EC50 of 0.5 µM and SI of 8.6) and ZIKV (EC50 of 0.9 µM and SI of 3.8). Similarly, to other Amarillydaceae alkaloids, lycorine was toxic at low concentrations to BHK-21 and Vero cells, with CC50 of 4.3 µM and 3.4 µM, respectively. Using commercial lycorine, we synthesized 1-acetyl-lycorine and 1,2-diacetyl-lycorine derivatives to test them against DENV-2 and ZIKV. 1,2-diacetyl-lycorine showed CC50 > 100 µM on BHK-21 and Vero cells but was inactive against DENV-2 and ZIKV. Likewise, the 1-acetyl-lycorine showed decreased cytotoxicity in BHK-21 cells (CC50 of 7.8 µM) and in Vero cells (CC50 of 19.8 µM) when compared to lycorine. However, 1-acetyl-lycorine was inactive against DENV-2 and showed very weak activity against ZIKV, with EC50 of 19.3 µM and SI of 1.0. The CC50 and EC50 titration curves of lycorine, pretazettine, narciclasine, against ZIKV and DENV are shown in the Additional file 1: Fig. S3.
Table 3.
Antiviral activities of compounds against DENV-2 and ZIKV
| Compounds | DENV-2 | ZIKV | CC50 (µM) | |||||
|---|---|---|---|---|---|---|---|---|
| EC50 (µM) | Virucidal activity | SI | EC50 (µM) | Virucidal activity | SI | BHK-21 | Vero | |
| Lycorine | 0.5 | No | 8.6 | 0.9 | No | 3.8 | 4.3 | 3.4 |
| Rutine | Inactive | No | – | Inactive | NT | – | > 300 | > 300 |
| Pretazettine | 0.8 | No | 6.8 | 1.9 | No | 3.8 | 5.4 | 7.2 |
| Narciclasine | 0.02 | No | 4.5 | 0.02 | No | 6.0 | 0.09 | 0.12 |
| Narciclasine-4-O-β-D-xylopyranoside | 7.9 | No | 4.9 | 7.9 | No | 6.5 | 39.3 | 51.8 |
| 1-acetyl-lycorine* | Inactive | No | – | 19.3 | No | 1.0 | 7.8 | 19.8 |
| 1,2-diacetyl-lycorine* | Inactive | No | – | Inactive | No | – | > 100 | > 100 |
EC50: half maximum effective concentration; CC50: half maximum cytotoxic concentration; SI: Selectivity Index = ratio CC50/EC50; *Synthetic derivatives from lycorine
Pretazettine and lycorine association against DENV-2 and ZIKV
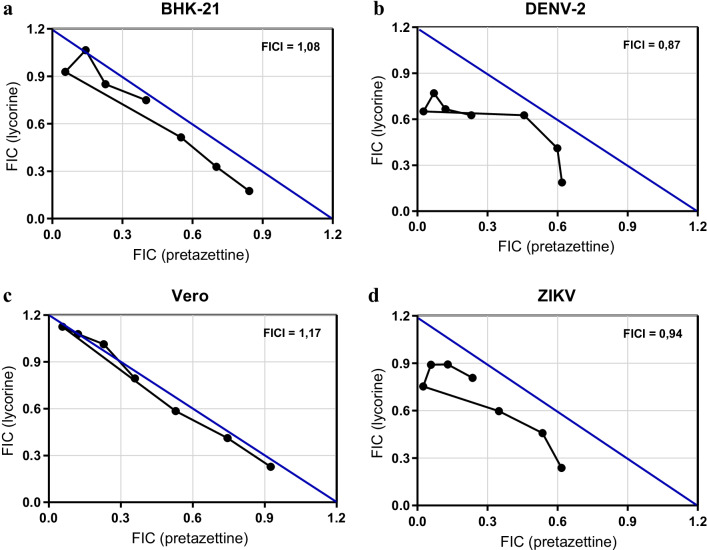
Since pretazettine and lycorine were active against both viruses and could act by different mechanisms of action, the combinations at different proportions of these compounds were tested against DENV-2 and ZIKV to assess possible synergistic effects. Data analyses resulted in FICI values of 0.87 for DENV-2, 0.94 for ZIKV, 1.08 for BHK-21 cells, and 1.17 for Vero cells. According to Odds, 2003 [33], such interactions with 0.5 < FICI > 4.0, both in terms of antiviral activity and cytotoxicity, are considered indifferent. The absence of interactions between lycorine and pretazettine is most evident when the FIC was plotted and illustrated through the isobolograms. No synergistic or antagonistic interactions were identified (Fig. 1). Results are from two experiments performed with duplicates.
Fig. 1.
Isobolograms representing in vitro interactions of lycorine and pretazettine. The compounds were combined in fixed ration generating solutions which were tested for their cytotoxicity in BHK-21 (a) and Vero (c) cells and in parallel for their antiviral activity against DENV-2 (b) and ZIKV (d). The pair of doses were evaluated at the level of EC50 and CC50, which were determined by regression curves. Subsequently, their FIC was determined and plotted on the graphs above. All points on isobole represent dose pairs, FIC of pretazettine (x-axis) and lycorine (y-axis), that are expected to give the EC50 (in the case of viruses) or CC50 (in the case of cells). Plots are observed very close to the theoretical diagonal line, which means, in short, the indifferent effect of both compounds used together. The illustration strengthens the results obtained of statistical analyses, from which the fractional inhibitory concentration index (FICI) was obtained (shown at the upper right corner) and used to define interactions as indifferent for being between 0.5 and 4
Predicted pharmacokinetic and toxicity profiles
The pharmacokinetics and toxicity profiles, comprising 30 different ADMET properties, were calculated for lycorine, pretazettine, narciclasine, and NXP using the pkCSM platform. Kalbreclasine was also included, but its activity against ZIKV and DENV was not tested yet. The complete set of their predicted properties is available in Table 4. Pretazettine was predicted to have the highest intestinal absorption by both Caco2 cells permeability (log Papp > 0.90), which models the absorption of orally administered drugs, and by the intestinal absorption model (predicted to be 95% absorbed). The other compounds were predicted to have intermediate to low intestinal absorption. Pretazettine was also the molecule with the highest predicted steady-state volume of distribution (VDss) (VDss = 0.80), meaning better distribution in tissue rather than plasma, followed closely by lycorine (VDss = 0.79). Also, in terms of distribution properties, none of the molecules was predicted to readily cross the blood–brain barrier and penetrate the central nervous system, with kalbreclasine and NXP predicted to be very poorly distributed in the brain (log BB = -1.66 and -1.78, respectively). None of the compounds were predicted to be P450 inhibitors, and, instead, lycorine and pretazettine were predicted to be P450 substrates. In line with the predicted metabolism profiles of these two molecules were their excretion profiles. Both presented the highest total clearance (logCL = 1.03 for lycorine and logCL = 0.97 for pretazettine). In terms of their toxicity profile, none of the molecules were predicted to be mutagenic (AMES toxicity) or elicit skin sensitization. Three out of four were predicted to be hepatotoxic; however (NXP) was expected not to disrupt normal liver function. Three out of four molecules were also predicted to have low maximum recommended tolerated doses (MRTD), with only narciclasine presenting a high MRTD [0.60 log (mg/kg/day)] and NXP presenting an intermediate MRTD [0.34 log (mg/kg/day)].
Table 4.
Predicted pharmacokinetic and toxicity profiles (ADMET) of the studied compounds
| Compound/pkCSM property | LYC | PRZ | NCL | NXP | KBL |
|---|---|---|---|---|---|
| Water solubility | − 2.982 | − 2.935 | − 2.667 | − 2.431 | − 2.468 |
| Caco2 permeability | 0.517 | 1.192 | 0.586 | − 0.47 | − 0.417 |
| Intestinal absorption (human) | 68.852 | 95.146 | 68.248 | 47.934 | 35.249 |
| Skin Permeability | − 3.319 | − 3.810 | − 2.931 | − 2.749 | − 2.736 |
| P-Glycoprotein substrate | No | No | Yes | Yes | Yes |
| P-Glycoprotein I inhibitor | No | No | No | No | No |
| P-Glycoprotein II inhibitor | No | No | No | No | No |
| VDss (human) | 0.790 | 0.802 | − 0.561 | − 0.540 | − 0.537 |
| Fraction unbound (human) | 0.446 | 0.458 | 0.536 | 0.609 | 0.576 |
| BBB permeability | − 0.153 | − 0.084 | − 1.315 | − 1.784 | − 1.660 |
| CNS permeability | − 2.518 | − 2.502 | − 3.788 | − 4.649 | − 4.883 |
| CYP2D6 substrate | Yes | No | No | No | No |
| CYP3A4 substrate | Yes | Yes | No | No | No |
| CYP1A2 inhibitor | No | No | No | No | No |
| CYP2C19 inhibitor | No | No | No | No | No |
| CYP2C9 inhibitor | No | No | No | No | No |
| CYP2D6 inhibitor | No | No | No | No | No |
| CYP3A4 inhibitor | No | No | No | No | No |
| Total Clearance | 1.031 | 0.974 | 0.223 | 0.480 | 0.491 |
| Renal OCT2 substrate | No | Yes | No | No | No |
| AMES toxicity | No | No | No | No | No |
| Max. tolerated dose (human) | − 0.386 | − 0.566 | 0.597 | 0.335 | 0.181 |
| hERG I inhibitor | No | No | No | No | No |
| hERG II inhibitor | No | No | No | No | No |
| Oral Rat Acute Toxicity (LD50) | 2.674 | 2.417 | 1.982 | 2.854 | 2.621 |
| Oral Rat Chronic Toxicity (LOAEL) | 0.977 | 1.739 | 2.896 | 4.310 | 3.692 |
| Hepatotoxicity | Yes | Yes | Yes | No | No |
| Skin Sensitisation | No | No | No | No | No |
| T. pyriformis toxicity | 0.526 | 0.443 | 0.276 | 0.285 | 0.285 |
| Minnow toxicity | 1.877 | 2.583 | 4.660 | 7.014 | 8.766 |
LYC: lycorine; PRZ: pretazettine; NCL: narciclasine; NXP: narciclasine-4-O-β-D-xylopyranoside; \: kalbreclasine; ADMET: Absorption, Distribution, Metabolism, Excretion, and Toxicity
Discussion
Numerous human viral diseases have a significant economic impact worldwide. The lack of licensed drugs to treat patients affected by many viral diseases such as dengue and Zika urges the need to discover, develop, and reposition drugs with innovative viral targets and mechanisms of action. In this work, in vitro assays were established and employed to identify extracts with antiviral properties against DENV and ZIKV and used to test thousands of extracts from different parts of hundreds of plant species from different Brazilian biomes.
Some of the active extracts showed virucidal properties since their activity relied on direct inactivation of the viral particles before virus adsorption. It is also speculated that compounds in the crude extracts could interfere with the virion envelope, masking the E and M proteins necessary for viral adsorption and entry into the host cells [42–45]. However, the composition and the precise mechanism of action of the virucidal extracts identified in this study needs further investigation.
One of our study's limitations is that some active non-virucidal extracts from Annonaceae, Malpighiaceae, and Rubiaceae families, when submitted to bioassay-guided fractionation, afforded no active fractions against DENV-2 and ZIKV. It is known that the phenomenon of synergistic effects of compounds present in an extract frequently causes loss-of-activity during bioassay-guided fractionation efforts, precluding identification or characterization of a relevant fraction for further evaluation, which is also a subject of further study. Therefore, we can infer that the antiviral activity verified in those extracts may have been due to the association of compounds separated in the fractionation steps or even their degradation along these procedures. Indeed, extracts and even their fractions may contain complex mixtures of substances which can result in a combination of different effects, not allowing reliable "go/no-go" decisions for further processing.
For those extracts presented active fractions against DENV-2 and ZIKV, we identified 18 known alkaloids and one flavonoid. However, few reports about the antiviral properties of these natural products were published. Only lycorine was detected in extracts of three species of Hippeastrum. Pseudolycorine was detected in H. glaucescens and H. puniceum. Among the 16 alkaloids identified, 12 were detected exclusively in H. puniceum and four in H. glaucescens. These results emphasize the importance of investigating this class of alkaloids as antivirals. Noteworthy, the alkaloids pretazettine, narciclasine, narciclasine-4-O-β-D-xylopyranoside, and the flavonoid rutine have never been tested against ZIKV. Just recently, the in vitro and in vivo antiviral properties of lycorine, found in more than 30 genera of plants, were reported against ZIKV [46]. Still, any lycorine derivatives have never been tested against this virus. Lycorine has broad-spectrum inhibitory activities against several RNA and DNA viruses, such as poliovirus [47], severe acute respiratory syndrome-associated coronavirus (SARS-CoV) [48], Herpes simplex virus (type 1) [49], DENV, West Nile virus and Yellow fever virus [50]. Some studies have put considerable effort into identifying related compounds or analogues from lycorine [30, 51, 52] by investigating its structure–activity relationships and analogues. These strategies have allowed us to discover promising less toxic molecules with higher or comparable levels of antiviral activity. Wang et al. [30] showed that a lycorine analogue, 1‐acetyl-lycorine, exhibited the most potent anti‐DENV activity with reduced cytotoxicity in A549 cells. In addition, 1-acetyl-lycorine inhibited hepatitis C virus (HCV) and suppressed the proliferation of multiple strains of Enterovirus 71 (EV71) through targeting viral proteases [53]. Here, we showed that 1-acetyl-lycorine and 1,2-diacetyl-lycorine have no antiviral activity against DENV-2 and a weak activity against ZIKV. We believe that different cell lines and assays could explain this discordance among those studies. Pretazettine, another alkaloid detected in active fractions of Hippeastrum, was isolated for the first time by Furusawa et al. [54] from Narcissus tazetta, an Amaryllidaceae species. Pretazettine strongly inhibited the activity of RNA-dependent DNA polymerase (reverse transcriptase) of the avian myeloblastosis virus [55]. Furthermore, this alkaloid exhibited consistent in vitro activity against Japanese encephalitis virus (JEV), YFV, Punta toro virus (PTV), Rift Valley fever virus [50], and human herpesvirus 1 (HHV-1) [49]. Interestingly, pretazettine inhibited the growth of the Rauscher virus and cellular protein synthesis in eukaryotic cells by a mechanism that does not affect DNA and RNA synthesis [50, 56–60].
In our work, we reasoned that the combination between pretazettine and lycorine could be more beneficial, given a possible synergistic action between them. This is supported by the fact that two compounds could act in different steps of the viral cycle and increase their effectiveness as antivirals. Indeed, combination therapies with multiple drugs have been demonstrated as effective approaches to treat several diseases such as malaria, tuberculosis, leishmaniasis, HIV, and HCV [61–67]. However, according to our results and the isobologram analyses, the antiviral effects of pretazettine-lycorine combinations in vitro showed to be indifferent against DENV-2 and ZIKV. The same was observed for their toxicity in Vero and BHK-21 cells.
Narciclasine, isolated from H. puniceum in this work, was reported to have selective and highly potent cytotoxic action on cancer cells, therefore being intensively investigated as an antitumor compound, in vitro and in vivo [68–72], as well as anti-inflammatory [73], anti-Alzheimer [74] and antiviral against JEV, YFV and DENV [50]. We have shown that narciclasine, though more toxic than lycorine and pretazettine was active against DENV-2 at a concentration 25 times less than presented by lycorine and 40 times less than presented by pretazettine. Similarly, the same was observed for ZIKV, being narciclasine activity verified at concentrations 45 times less than presented by lycorine and 95 times less than presented by pretazettine. Our data are similar to those reported by Gabrielsen et al. [50] for other flaviviruses as JEV, YFV, and DENV-4. Even though our study showed that minimum toxic doses and therapeutic doses were very similar, indicating a very narrow window for prophylaxis and therapy, there is room for improvement. As already mentioned, studies of the structure–activity relationship can be a promising area for exploring chemical radicals that can lead to increased antiviral activity and reduced cytotoxicity. Until now, the synthesis of narciclasine derivatives did not result in an optimized compound, at least concerning its antimitotic properties [75]. However, the discovery of new natural congeners of narciclasine with conserved antiviral activity can spur new studies of the antiviral activity of this molecule. Here we report the anti-DENV-2 and anti-ZIKV activity of narciclasine-4-O-β-D-xylopyranoside (NXP) isolated from bulbs of H. puniceum. NXP was 430 times less toxic than narciclasine in BHK-21 and Vero cells. Its EC50 values against DENV-2 and ZIKV were at concentrations 395 times those detected for narciclasine for both viruses. NXP was recently reported and characterized by Katoch et al. [41] in another species of Amaryllidaceae. It's relevant to mention that not all alkaloids identified in active extracts of Hippeastrum against DENV-2 and/ZIKV were tested in this work, and some of them, as narcissidine acetate, N-norlycoramine, 6-hidroxi-hippeastrine, lycoranine E, lycoranine C, crisarnine and acetyl-nerbowdine were not reported yet having any biological activity. The others, as pseudolycorine [76], pancratine and pancracine have already been shown to contain antiproliferative activities [77, 78]. Several studies indicate that alkaloids trigger a range of biological activities. Kalbreclasine, for example, exhibited a potent mitogenic action on splenic lymphocytes in healthy adult mice, stimulating their extensive proliferation [79, 80]. Nangustine has shown weak activities against trypanosome, leishmania, and plasmodium parasites [81]. Incartine [82–84] has shown significant neuroprotective effects against cell injury models in dopaminergic neuroblastoma SHSY5Y cells [85]. Lycoramine is a reversible cholinesterase inhibitor [86] and a modulator of nicotinic receptors, an important property in treating Alzheimer's and Parkinson's disease or neuroprotection against neurodegenerative disorders [87]. Nonetheless, these metabolites are isolated only in low amounts and therefore are not commercially available, representing an issue for thorough biological investigation.
Thankfully, in silico techniques have been introduced to drug discovery and development as tools to predict and optimize the ADMET properties of candidates at early stages [88]. These use physicochemical properties of compounds and advanced computational modelling to generate predicted models. Computational approaches may help minimize risks in following studies or taken important information for the decision to advance, hold or terminate a drug candidate [89]. Here, we used the pkCSM platform, which uses graph-based signatures to develop predictive models of central ADMET properties, [36] to analyse the compounds lycorine, pretazettine, narciclasine, NXP that exhibited some antiviral against DENV and ZIKV. ADMET properties of kalbreclasine were also analysed. The results supported these molecules as candidates for in vivo studies. Hopefully, chemical modifications studies can bring new derivatives that could be well optimized to present better bioavailability, oral absorption, clearance, volume of distribution, less toxicity, increased antiviral activity, and penetration into the central nervous system. Our findings corroborate the potential of plants to produce antiviral compounds. Indeed, there is still a lot of work to be done since we identified many other compounds, not tested yet, in the active extracts reported by this study. A natural progression of this work is to assess the potential antiviral properties of all these compounds against ZIKV, DENV, and other viruses. Surely, innovative strategies are required to reveal and contribute the full range of chemical diversity of these valuable natural products to the antiviral drugs discovery process.
Conclusions
The laborious screening of 7,000 plant extracts for antiviral activity led to the identification of extracts from the Amaryllidaceae family, which allowed the identification of several antiviral drug candidates. Our study supports the importance of exploring random crude extracts of plants to unveil new antiviral agents. We demonstrated the anti-ZIKV activity of pretazettine, narciclasine, and narciclasine-4-O-β-D-xylopyranoside. Finally, our findings indicate that plant species mainly of the genus Hippeastrum are a useful source of antiviral compounds against DENV-2 and ZIKV.
Supplementary Information
Additional file 1: Figure S1. Map of the plates used for the validation assays of antiviral activity against DENV-2 and ZIKV using the MTT method. The validation was performed as per the High Throughput Screening (HTS) protocol described by Iversen et al., 2012 [24]. The model shows a combination of wells that produce the different intercalated signals, namely: uninfected cells (H), infected cells (L) and infected and treated cells (M), suitable for statistical analysis of absorbance readings of the product of the reduction of MTT by the cells. Figure S2. Validation of the antiviral HTS assay against DENV-2 and ZIKV using the MTT method. The validation was performed as per the High Throughput Screening (HTS) protocol described by Iversen et al., 2012 [24]. Raw data values of the plates on the day 3 (endpoint) of plate uniformity study with interleaved distribution of MIN (infected cells), MED (treated and infected cells) and MAX (cell control) signals analyzed by row (A and C) and by column (B and D). Figure S3. CC50 and EC50 titration curves of pretazettine (PTZ),lycorine (LYC), narciclasine (NCL), and narciclasine-4-O-β-D-xylopiranoside (NXP) against DENV-2 and ZIKV. The values were determined by regression curve using GraphPad Prism 5 based on nonlinear logistic regression of the dose-response curves. The values correspond to the average and standard deviation of three independent assays with at least 8 concentrations of the substance. The red dots correspond to the concentration at which the substance has reached host cell toxicity in antiviral assays.
Acknowledgements
The authors would like to acknowledge Carlos Alberto Ferreira Junior from Fundação Zoobotânica—Belo Horizonte-MG for the Hippeastrum collection. We are also grateful to the technologists and resources of the Fiocruz Bioprospection and Nuclear Magnetic Resonance technological platforms.
Abbreviations
- ADMET
Stands for Absorption, Distribution, Metabolism, Excretion and Toxicity
- ATCC
American Type Culture Collection
- CAPES
Coordenação de Aperfeiçoamento de Pessoal de Nível Superior
- CC50
Half maximum cytotoxic concentration
- CNPq
Conselho Nacional de Desenvolvimento Científico e Tecnológico
- CPE
Cytopathic effect
- DENV
Dengue virus
- DENV-2
Dengue virus serotype 2
- DENV-4
Dengue virus serotype 4
- DMSO
Dimethyl sulfoxide
- EC50
Half maximum effective concentration
- EDTA
Ethylenediamine tetraacetic acid
- FBS
Fetal bovine serum
- FIC
Fractional inhibitory concentration
- FICI
Fractional inhibitory concentration index
- H. puniceum
Hippeastrum puniceum
- H. glaucescens
Hippeastrum glaucescens
- HCV
Hepatitis C virus
- HHV-1
Human herpesvirus 1
- HIV
Human Immunodeficiency virus
- HRMS/MS
High resolution mass spectrometry in tandem with mass spectrometry
- JEV
Japanese encephalitis virus
- MEM
Minimum Eagle's Medium
- m.o.i.
Multiplicity of infection
- MTT
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide
- MSn
Mass spectrometry
- MRTD
Maximum recommended tolerated doses
- NMR
Nuclear magnetic resonance
- NXP
Narciclasine-4-O-β-D-xylopyranoside
- PFU
Plaque forming unit
- p.i.
Post infection
- UHPLC
Ultra high pressure liquid chromatography
- YFV
Yellow fever virus
- ZIKV
Zika virus
Authors' contributions
JGO, ECB, EGK, CLZ, CECS, LHR, and TMAA participated in the conception, design, and acquisition of funds for the study. ECB, ACCF, EARS, STGT, and JGO performed the infection procedures and antiviral data analysis and interpretation. MS was responsible for plant harvesting and identification. MK, TMAA, and CLZ carried out the experiments, analysis, and interpretation of chromatography and mass spectrometry studies. TMAA and CLZ carried out NMR data analysis and interpretation. CLZ and DEVP carried out the ADMET data analysis and interpretation. JGO, CLZ, ECB, EGK, STGJ; CECS, DEVP; EARA, LHR, and TMAA made substantial contributions in the interpretation of data and revising the manuscript for intellectual content. All authors read and approved the final manuscript.
Funding
This work was supported by Fundação de Amparo à Pesquisa do Estado de Minas Gerais – (Fapemig) #APQ-02797-16, Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES); MCTIC/FNDC-CNPq/MEC-CAPES/MS-Decit award #440911/2016-8, and Fundação Oswaldo Cruz (Fiocruz). Emerson C. Barbosa received scholarship from CAPES and Luiz H Rosa, Erna G Kroon, Carlos L Zani and Tânia M A Alves received research fellowships from CNPq.
Availability of data and materials
All data generated or analysed during this study are included in this published article.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Carlos Leomar Zani, Email: carlos.zani@fiocruz.br.
Jaquelline Germano de Oliveira, Email: jaquelline.oliveira@fiocruz.br.
References
- 1.Aliota MT, et al. Zika in the Americas, year 2: What have we learned? What gaps remain? A report from the Global Virus Network. Antiviral Res. 2017;144:223–246. doi: 10.1016/j.antiviral.2017.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Baud D, et al. An update on Zika virus infection. Lancet. 2017;390(10107):2099–2109. doi: 10.1016/s0140-6736(17)31450-2. [DOI] [PubMed] [Google Scholar]
- 3.Dos Santos T, et al. Zika virus and the Guillain-Barre syndrome - case series from seven countries. N Engl J Med. 2016;375(16):1598–1601. doi: 10.1056/NEJMc1609015. [DOI] [PubMed] [Google Scholar]
- 4.FDA. Zika Virus Response Updates from FDA|FDA. Available online: https://www.fda.gov/emergency-preparedness-and-response/mcm-issues/zika-virus-response-updates-fda Accessed on 03 March 2021.
- 5.WHO. Dengue and Severe Dengue. Available online: https://www.who.int/health-topics/dengue-and-severe-dengue#tab=tab_1 Accessed on 03 March 2021.
- 6.Thomas SJ, Yoon IK. A Review of Dengvaxia®: development to deployment. Hum Vaccine Immunother. 2019;15(10):2295–2314. doi: 10.1080/21645515.2019.1658503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ong A, et al. Fatal dengue hemorrhagic fever in adults during a dengue epidemic in Singapore. Int Infect Dis. 2007;11(3):263–267. doi: 10.1016/j.ijid.2006.02.012. [DOI] [PubMed] [Google Scholar]
- 8.Guo C, et al. Global epidemiology of dengue outbreaks in 1990–2015: a systematic review and meta-analysis. Front Cell Infect Microbiol. 2017;7:317. doi: 10.3389/fcimb.2017.00317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Musso D, Gubler DJ. Zika Virus. Clin Microbiol Rev. 2016;29(3):487–524. doi: 10.1128/CMR.00072-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.CDC. Treatment Zika Virus CDC. Available online: https://www.cdc.gov/zika/symptoms/treatment.html Accessed on 3 Mar 2021.
- 11.Baz M, Boivin G. Antiviral agents in development for Zika virus infections. Pharmaceuticals (Basel, Switzerland) 2019;12(3):101. doi: 10.3390/ph12030101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Barrows NJ, et al. A screen of FDA-approved drugs for inhibitors of Zika virus infection. Cell Host Microbe. 2016;20:259–270. doi: 10.1016/j.chom.2016.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Boldescu V, et al. Broad-spectrum agents for flaviviral infections: dengue, Zika and beyond. Nat Rev Drug Discov. 2017;16(8):565–586. doi: 10.1038/nrd.2017.33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Saiz JC, Martín-Acebes MA. The race to find antivirals for Zika virus. Antimicrob Agents Chemother. 2017;61(6):e00411–e00417. doi: 10.1128/AAC.00411-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Teixeira AH, et al. Conhecimento popular sobre o uso de plantas medicinais no município de Sobral-Ceará, Brasil. SANARE. 2014;13(1):23–28. [Google Scholar]
- 16.Atanasov AG, et al. Discovery and resupply of pharmacologically active plant-derived natural products: a review. Biotechnol Adv. 2015;33(8):1582–1614. doi: 10.1016/j.biotechadv.2015.08.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.DeCorte BL. Underexplored opportunities for natural products in drug discovery. J Med Chem. 2016;59(20):9295–9304. doi: 10.1021/acs.jmedchem.6b00473. [DOI] [PubMed] [Google Scholar]
- 18.Chaudhuri J. The role of advanced glycation end products in aging and metabolic diseases: bridging association and causality. Cell Metab. 2018;28(3):337–352. doi: 10.1016/j.cmet.2018.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Newman DJ, Cragg GM. Natural products as sources of new drugs over the nearly four decades from 01/1981 to 09/2019. J Nat Prod. 2020;83:770–803. doi: 10.1021/acs.jnatprod.9b01285. [DOI] [PubMed] [Google Scholar]
- 20.Palombo EA. Phytochemicals from traditional medicinal plants used in the treatment of diarrhoea: modes of action and effects on intestinal function. Phytother Res. 2006;20(9):717–724. doi: 10.1002/ptr.1907. [DOI] [PubMed] [Google Scholar]
- 21.Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65(1–2):55–63. doi: 10.1016/0022-1759(83)90303-4. [DOI] [PubMed] [Google Scholar]
- 22.Betancur-Galvis L, et al. Antitumor and antiviral activity of Colombian medicinal plant extracts. Mem Inst Oswaldo Cruz. 1999;94(4):531–535. doi: 10.1590/s0074-02761999000400019. [DOI] [PubMed] [Google Scholar]
- 23.Kudi AC, Myint MS. Antiviral activity of some Nigerian medicinal plant extracts. J Ethnopharmocol. 1999;68:289–294. doi: 10.1016/S0378-8741(99)00049-5. [DOI] [PubMed] [Google Scholar]
- 24.Iversen PW, Beck B, Chen YF, et al., HTS Assay Validation. 2012 May In: Markossian S, Grossman A, Brimacombe K, et al., editors. Assay Guidance Manual [Internet]. Bethesda (MD): Eli Lilly & Company and the National Center for Advancing Translational Sciences; 2004-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK83783/ [PubMed]
- 25.Dulbecco R, Vogt M. Some problems of animal virology as studied by the plaque technique. Cold Spring Harb Symp Quant Biol. 1953;18:273–279. doi: 10.1101/sqb.1953.018.01.039. [DOI] [PubMed] [Google Scholar]
- 26.Faral-Tello P, et al. Cytotoxic, virucidal, and antiviral activity of South American plant and algae extracts. Sci World J. 2012;1(5):174837–174842. doi: 10.1100/2012/174837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zandi K, et al. Novel antiviral activity of baicalein against dengue virus. BMC Complement Altern Med. 2012;12(214):1–9. doi: 10.1186/1472-6882-12-214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Horai H, et al. Massbank: a public repository for sharing mass spectral data for life sciences. J Mass Spectrom. 2010;45(7):703–714. doi: 10.1002/jms.1777. [DOI] [PubMed] [Google Scholar]
- 29.Allard PM, et al. Integration of molecular networking and in-silico Ms/Ms fragmentation for natural products dereplication. Anal Chem. 2016;88(6):3317–3323. doi: 10.1021/acs.analchem.5b04804. [DOI] [PubMed] [Google Scholar]
- 30.Wang P, et al. Anti-dengue virus activity and structure-activity relationship studies of lycorine derivatives. ChemMedChem. 2014;9(7):1522–1533. doi: 10.1002/cmdc.201300505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Fivelman QL, Adagu IS, Warhurst DC. Modified fixed-ratio isobologram method for studying in vitro interactions between atovaquone and proguanil or dihydroartemisinin against drug-resistant strains of Plasmodium falciparum. Antimicrob Agents Chemother. 2004;48(11):4097–4102. doi: 10.1128/AAC.48.11.4097-4102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Seifert K, Croft SL. In vitro and in vivo interactions between miltefosine and other antileishmanial drugs. Antimicrob Agents Chemother. 2006;50(1):73–79. doi: 10.1128/AAC.50.1.73-79.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Odds FC. Synergy, antagonism, and what the chequerboard puts between them. J Antimicrob Chemother. 2003;52(1):1. doi: 10.1093/jac/dkg301. [DOI] [PubMed] [Google Scholar]
- 34.Tallarida RJ. Drug synergism: its detection and applications. J Pharmacol Exp Ther. 2001;298(3):865–872. [PubMed] [Google Scholar]
- 35.Tallarida RJ. Revisiting the isobole and related quantitative methods for assessing drug synergism. J Pharm Exp Ther. 2012;342(1):2–8. doi: 10.1124/jpet.112.193474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pires DEV, Blundell TL, Ascher DB. pkCSM: Predicting small-molecule pharmacokinetic and toxicity properties using graph-based signatures. J Med Chem. 2015;58(9):4066–4072. doi: 10.1021/acs.jmedchem.5b00104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Pires DEV, et al. Cutoff Scanning Matrix (CSM): structural classification and function prediction by protein inter-residue distance patterns. BMC Genomics. 2011 doi: 10.1186/1471-2164-12-S4-S12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pires DEV, et al. aCSM: noise-free graph-based signatures to large-scale receptor-based ligand prediction. Bioinformatics. 2013;29(7):855–861. doi: 10.1093/bioinformatics/btt058. [DOI] [PubMed] [Google Scholar]
- 39.Pires DEV, Ascher DB. mycoCSM: using graph-based signatures to identify safe potent hits against Mycobacteria. J Chem Inf Model. 2020;60(7):3450–3456. doi: 10.1021/acs.jcim.0c00362. [DOI] [PubMed] [Google Scholar]
- 40.Moraes CT. Estudo fitoquímico das aéreas de Worsleya procesra (Lem.) Traub (Amaryllidaceae). 2016. Dissertação (Mestrado em Química) Centro de Ciências Exatas da Universidade Federal do Espírito Santo, Espírito Santo. https://repositorio.ufes.br/bitstream/10/7319/1/tese_10338_Disserta%C3%A7%C3%A3o%20FINAL%20Carolina%20PDF%20%281%29.pdf
- 41.Katoch D. Narciclasine-4-O-β-D-xylopyranoside, a new narciclasine glycoside from Zephyranthes minuta. Nat Prod Res. 2019;34(2):233–240. doi: 10.1080/14786419.2018.1527836. [DOI] [PubMed] [Google Scholar]
- 42.Schuhmacher A, Reichling J, Schnitzler P. Virucidal effect of peppermint oil on the enveloped viruses Herpes simplex virus type 1 and type 2 in vitro. Phytomedicine. 2003;10(6–7):504–510. doi: 10.1078/094471103322331467. [DOI] [PubMed] [Google Scholar]
- 43.Gao F, et al. Novel binding between pre-membrane protein and claudin-1 is required for efficient dengue virus entry. Biochem Biophys Res Commun. 2010;391(1):952–957. doi: 10.1016/j.bbrc.2009.11.172. [DOI] [PubMed] [Google Scholar]
- 44.Zhang Q, et al. The stem region of premembrane protein plays an important role in the virus surface protein rearrangement during dengue maturation. J Biol Chem. 2012;287(48):40525–40534. doi: 10.1074/jbc.M112.384446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Guo M, et al. ZIKV viral proteins and their roles in virus-host interactions. Sci China Life Sci. 2020;14:1–11. doi: 10.1007/s11427-020-1818-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chen H, et al. Antiviral activity of lycorine against Zika virus in vivo and in vitro. Virology. 2020;546:88–97. doi: 10.1016/j.virol.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hwang YC, et al. Rapid identification of inhibitors that interfere with poliovirus replication using a cell-based assay. Antiviral Res. 2008;77(3):232–236. doi: 10.1016/j.antiviral.2007.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Li S, et al. Identification of natural compounds with antiviral activities against SARS-associated coronavirus. Antiviral Res. 2005;67(1):18–23. doi: 10.1016/j.antiviral.2005.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Renard-Nozaki J, et al. effect of alkaloids isolated from Amaryllidaceae on Herpes simplex virus. Res Virol. 1989;140:115–128. doi: 10.1016/s0923-2516(89)80089-5. [DOI] [PubMed] [Google Scholar]
- 50.Gabrielsen B, et al. Antiviral (RNA) activity of selected Amaryllidaceae isoquinoline constituents and synthesis of related substances. J Nat Prod. 1992;55(11):1569–1581. doi: 10.1021/np50089a003. [DOI] [PubMed] [Google Scholar]
- 51.Cedrón JC, et al. Synthesis and antiplasmodial activity of lycorine derivatives. Bioorg Med Chem. 2010;18(13):4694–4701. doi: 10.1016/j.bmc.2010.05.023. [DOI] [PubMed] [Google Scholar]
- 52.Chen D, et al. Design, synthesis and structure-activity relationship optimization of lycorine derivatives for HCV inhibition. Sci Rep. 2015;5(1):14972. doi: 10.1038/srep14972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Guo Y, et al. A conserved inhibitory mechanism of a lycorine derivative against enterovirus and Hepatitis C virus. Antimicrob Agents Chemother. 2015;60(2):913–924. doi: 10.1128/aac.02274-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Furusawa E. Isolation of pretazettine from Narciussus tazetta L. Chem Pharm Bull. 1976;24(2):336–338. doi: 10.1248/cpb.24.336. [DOI] [Google Scholar]
- 55.Papas TS, et al. Inhibition of DNA polymerase of avian myeloblastosis virus by an alkaloid extract from Narcissus tazetta L. Biochem Biophys Res Commun. 1973;52(1):88–92. doi: 10.1016/0006-291x(73)90957-1. [DOI] [PubMed] [Google Scholar]
- 56.Suzuki N, et al. Therapeutic activity of Narcissus alkaloids on Rauscher leukemia: antiviral effect in vitro and rational drug combination in vivo. Proc Soc Exp Biol Med. 1974;145(3):771–777. doi: 10.3181/00379727-145-37892. [DOI] [PubMed] [Google Scholar]
- 57.Jimenez A, et al. Inhibitors of protein synthesis in eukaryotic cells: comparative effects of some Amaryllidaceae alkaloids. Biochim Biophys Acta. 1976;425(3):342–348. doi: 10.1016/0005-2787(76)90261-6. [DOI] [PubMed] [Google Scholar]
- 58.Martin SF. The Amaryllidaceae alkaloids. Chapter 3. In: Brossi A, editor. The alkaloids: chemistry and pharmacology. New York: Academic Press; 1987. pp. 251–376. [Google Scholar]
- 59.Furusawa E, et al. Therapeutic activity of pretazettine on Rauscher leukemia: combination of antiviral activity and cellular protein inhibition. Chemotherapy. 1978;24(4):259–266. doi: 10.1159/000237790. [DOI] [PubMed] [Google Scholar]
- 60.Furusawa E, et al. Therapeutic activity of pretazettine on Rauscher leukemia: comparison with the related Amaryllidaceae alkaloids. Chemotherapy. 1980;26(1):36–45. doi: 10.1159/000237881. [DOI] [PubMed] [Google Scholar]
- 61.Scott LJ, Perry CM. Interferon-alpha-2b plus ribavirin. Drugs. 2002;62(3):507–556. doi: 10.2165/00003495-200262030-00009. [DOI] [PubMed] [Google Scholar]
- 62.Whegang SY, Tahar R, Foumane VN, et al. Efficacy of non-artemisinin- and artemisinin-based combination therapies for uncomplicated falciparum malaria in Cameroon. Malaria J. 2010 doi: 10.1186/1475-2875-9-56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Costa-Gouveia J, et al. Combination therapy for tuberculosis treatment: pulmonary administration of ethionamide and booster co-loaded nanoparticles. Sci Rep. 2017;7:5390. doi: 10.1038/s41598-017-05453-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Diro E, et al. A randomized trial of Am Bisome monotherapy and Am Bisome and miltefosine combination to treat visceral leishmaniasis in HIV co-infected patients in Ethiopia. PLOS Negl Trop Dis. 2019;13(1):e0006988. doi: 10.1371/journal.pntd.0006988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gonçalves-Oliveira LF, et al. The combination therapy of meglumine antimoniate and oxiranes (epoxy-α-lapachone and epoxymethyl-lawsone) enhance the leishmanicidal effect in mice infected by Leishmania (Leishmania) amazonensis. Int J Parasitol Drugs Drug Resist. 2019;10:101–108. doi: 10.1016/j.ijpddr.2019.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Moreno S, et al. Two-drug vs. three-drug combinations for HIV-1: Do we have enough data to make the switch? HIV Med. 2019;4:2–12. doi: 10.1111/hiv.12716. [DOI] [PubMed] [Google Scholar]
- 67.Alven S, Aderibigbe B. Combination therapy strategies for the treatment of malaria. Molecules. 2019;24(19):3601. doi: 10.3390/molecules24193601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Ceriotti G. Narciclasine: an antimitotic substance from Narcissus bulbs. Nature. 1967;213:595–596. doi: 10.1038/213595a0. [DOI] [PubMed] [Google Scholar]
- 69.Pettit GR, et al. Antineoplastic agents, 256. Cell growth inhibitory isocarbostyrils from Hymenocallis. J Nat Prod. 1993;56(10):1682–1687. doi: 10.1021/np50100a004. [DOI] [PubMed] [Google Scholar]
- 70.Van Goietsenoven G, et al. Narciclasine as well as other Amaryllidaceae isocarbostyrils are promising GTP-ase targeting agents against brain cancers. Med Res Rev. 2013;33(2):439–455. doi: 10.1002/med.21253. [DOI] [PubMed] [Google Scholar]
- 71.Dumont P, et al. The Amaryllidaceae isocarbostyril narciclasine induces apoptosis by activation of the death receptor and/or mitochondrial pathways in cancer cells but not in normal fibroblasts. Neoplasia. 2007;9(9):766–776. doi: 10.1593/neo.07535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Ingrassia L, et al. Structure-activity relationship analysis of novel derivatives of narciclasine (an Amaryllidaceae isocarbostyril derivative) as potential anticancer agents. J Med Chem. 2009;52(4):1100–1114. doi: 10.1021/jm8013585. [DOI] [PubMed] [Google Scholar]
- 73.Mikami M, et al. Suppressive activity of lycoricidinol (narciclasine) against cytotoxicity of neutrophil-derived calprotectin, and its suppressive effect on rat adjuvant arthritis model. Biol Pharm Bull. 1999;22(7):674–678. doi: 10.1248/bpb.22.674. [DOI] [PubMed] [Google Scholar]
- 74.Kim J, et al. Effect of Lycoris chejuensis and its active components on experimental models of Alzheimer's Disease. J Agricult Food Chem. 2015;63(31):6979–6988. doi: 10.1021/acs.jafc.5b00889. [DOI] [PubMed] [Google Scholar]
- 75.Fürst R. Narciclasine – an Amaryllidaceae alkaloid with potent antitumor and anti-inflammatory properties. Planta Med. 2016;82(16):1389–1394. doi: 10.1055/s-0042-115034. [DOI] [PubMed] [Google Scholar]
- 76.Llabres JM, et al. Two alkaloids from Narcissus requienii. Phytochemistry. 1986;25(6):1453–1459. doi: 10.1016/S0031-9422(00)81308-4. [DOI] [Google Scholar]
- 77.Cedrón JC, et al. Antiproliferative and structure activity relationships of Amaryllidaceae alkaloids. Molecules. 2015;20(8):13854–13863. doi: 10.3390/molecules200813854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Katoch D, et al. Pseudolycorine N-oxide, a new N-oxide from Narcissus tazetta. Nat Prod Res. 2019;34(14):2051–2058. doi: 10.1080/14786419.2019.1574785. [DOI] [PubMed] [Google Scholar]
- 79.Ghosal S, et al. Alkaloids of Haemanthus kalbreyeri. Phytochemistry. 1985;24(8):1825–1828. doi: 10.1016/S0031-9422(00)82560-1. [DOI] [Google Scholar]
- 80.Ghosal S, Singh SK, Srivastava RS. Alkaloids of Zephyranthes flava. Phytochemistry. 1986;25(8):1975–1978. doi: 10.1016/S0031-9422(00)81187-5. [DOI] [Google Scholar]
- 81.Labraña J, et al. Alkaloids from Narcissus angustifolius subsp. transcarpathicus (Amaryllidaceae) Phytochemistry. 2002;60(8):847–852. doi: 10.1016/s0031-9422(02)00154-1. [DOI] [PubMed] [Google Scholar]
- 82.Kihara M, et al. Incartine, a biosynthetic intermediate, from the flowers of Lycoris incarnata. Heterocycles. 1992;34(7):1299–1301. doi: 10.3987/COM-92-6068. [DOI] [Google Scholar]
- 83.Kihara M, et al. Isolation and structure elucidation of a novel alkaloid, incartine, a supposed biosynthetic intermediate, from flowers of Lycoris incarnata. Chem Pharm Bull. 1994;42(2):289–292. doi: 10.1248/cpb.42.289. [DOI] [Google Scholar]
- 84.Kihara M, Ozaki T, Kobayashi S, Shingu T. Alkaloidal constituents of Leucojum autumnale L. (Amaryllidaceae) Chem Pharm Bull. 1995;43(2):318–320. doi: 10.1248/cpb.43.318. [DOI] [Google Scholar]
- 85.Zhu YY, Li X, Yu HY, et al. Alkaloids from the bulbs of Lycoris longituba and their neuroprotective and acetylcholinesterase inhibitory activities. Arch Pharm Research. 2015;38(5):604–613. doi: 10.1007/s12272-014-0397-2. [DOI] [PubMed] [Google Scholar]
- 86.Stephenson GR, Roe C, Sandoe EJ. Electrophilic C12 Building Blocks for Alkaloids: 1,1 Iterative Organoiron-Mediated Routes to (±)-Lycoramine and (±)-Maritidine. Eur J Org Chem. 2011;9:1664–1681. doi: 10.1002/ejoc.201001394. [DOI] [Google Scholar]
- 87.European Patent Office (EP2311463A1). Analogues of galanthamine and lycoramine have activity as modulators of nicotinic receptors, see: a) B. Davis, US patent US 148253, 2002; b) B. Davis, PCT Int. Appl. WO 2001043697, 2001.
- 88.Boobis A, Gundert-Remy U, Kremers P, et al. In silico prediction of ADME and pharmacokinetics. Eur J Pharm Sci. 2002;17(4–5):183–193. doi: 10.1016/s0928-0987(02)00185-9. [DOI] [PubMed] [Google Scholar]
- 89.Zhang D, Luo G, Ding X, et al. Preclinical experimental models of drug metabolism and disposition in drug discovery and development. Acta Pharm Sinica B. 2012;2(6):549–561. doi: 10.1016/j.apsb.2012.10.004. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1. Map of the plates used for the validation assays of antiviral activity against DENV-2 and ZIKV using the MTT method. The validation was performed as per the High Throughput Screening (HTS) protocol described by Iversen et al., 2012 [24]. The model shows a combination of wells that produce the different intercalated signals, namely: uninfected cells (H), infected cells (L) and infected and treated cells (M), suitable for statistical analysis of absorbance readings of the product of the reduction of MTT by the cells. Figure S2. Validation of the antiviral HTS assay against DENV-2 and ZIKV using the MTT method. The validation was performed as per the High Throughput Screening (HTS) protocol described by Iversen et al., 2012 [24]. Raw data values of the plates on the day 3 (endpoint) of plate uniformity study with interleaved distribution of MIN (infected cells), MED (treated and infected cells) and MAX (cell control) signals analyzed by row (A and C) and by column (B and D). Figure S3. CC50 and EC50 titration curves of pretazettine (PTZ),lycorine (LYC), narciclasine (NCL), and narciclasine-4-O-β-D-xylopiranoside (NXP) against DENV-2 and ZIKV. The values were determined by regression curve using GraphPad Prism 5 based on nonlinear logistic regression of the dose-response curves. The values correspond to the average and standard deviation of three independent assays with at least 8 concentrations of the substance. The red dots correspond to the concentration at which the substance has reached host cell toxicity in antiviral assays.
Data Availability Statement
All data generated or analysed during this study are included in this published article.