Abstract
Emerging and re-emerging zoonotic diseases cause serious illness with billions of cases, and millions of deaths. The most effective way to restrict the spread of zoonotic viruses among humans and animals and prevent disease is vaccination. Recombinant proteins produced in plants offer an alternative approach for the development of safe, effective, inexpensive candidate vaccines. Current strategies are focused on the production of highly immunogenic structural proteins, which mimic the organizations of the native virion but lack the viral genetic material. These include chimeric viral peptides, subunit virus proteins, and virus-like particles (VLPs). The latter, with their ability to self-assemble and thus resemble the form of virus particles, are gaining traction among plant-based candidate vaccines against many infectious diseases. In this review, we summarized the main zoonotic diseases and followed the progress in using plant expression systems for the production of recombinant proteins and VLPs used in the development of plant-based vaccines against zoonotic viruses.
Keywords: recombinant vaccines, zoonotic viruses, virus-like particles, plant molecular farming, zoonotic influenza, emerging coronaviruses, West Nile virus, dengue virus, Zika virus, yellow fever virus, Ebola virus, Crimean–Congo hemorrhagic fever virus, Chikungunya virus, HIV, rabies virus, Hantaviruses, Henipaviruses, Newcastle virus, Hepatitis E virus
1. Introduction
Zoonoses are diseases transmitted from vertebrate animals to humans and are considered one of the most important threats to Public Health [1]. Zoonotic viruses cause illnesses with a high death rate and numerous long-term health issues. In addition, zoonotic diseases also affect the livestock sector and can have a tremendous economic impact [2]. More than 60% of emerging human infectious diseases are zoonoses and 99% of the emerging unknown viruses are zoonotic with a potentially high risk of spreading globally [3,4,5]. The recent outbreak of severe acute respiratory syndrome-associated coronavirus 2 (SARS-CoV-2), causing more than 5 million deaths worldwide by the end of October 2021, demonstrates the significance of zoonoses. Domestic and wild animals serve as reservoirs of zoonotic viruses, which are transmitted via direct or indirect contact [3,6,7]. The indirect transmission occurs by vectors (insects and arthropods), which significantly impacts disease transmission dynamics and complicates the measures taken to control zoonoses. Viruses, which have a transmission cycle between animal reservoirs where they primarily amplify, and their vectors (mosquitoes, ticks, midges) are known as arthropod-borne viruses or Arboviruses. Before transmission to a susceptible host, arboviruses must replicate in the arthropod vectors [8].
Table 1 summarizes important zoonotic viruses with details on the animal reservoirs, distribution, and fatality rate. It is worth noting that some viruses, such as HIV, began as zoonotic viruses, but because of mutations, they are no longer considered zoonotic.
Table 1.
Partial list of important viral zoonoses.
| Virus Family/Name | Main Animal Reservoir | Distribution | Cases/Mortality Rate |
|---|---|---|---|
| Family Orthomyxoviridae
Influenza A virus |
Wide variety of birds and mammals (dogs, cats, pigs, whales, horses) | Worldwide | 3–5 million severe cases annually, ~2% fatal rate |
| Family Coronaviridae
Coronavirus SARS-CoV MERS CoV SARS-CoV-2 |
Bats, pangolins, camels, monkeys, ferrets, minks, pigs, horses, dogs, cats, snakes | Worldwide | SARS-CoV~9.6%; MERS-CoV~34.3%; SARS-CoV-2 ranging from 2% to 7.6% depending on the country |
| Family Flaviviridae
West Nile virus |
Birds and horses | Africa, Europe, the Middle East, North America, and West Asia | ~1% of neuroinvasive disease and 5% of them are fatal |
| Zika virus | Human and non-human primates | Spread from Africa to the Pacific and the Americas | In Brazil, the estimated case fatality rate is 8.3% |
| Japanese encephalitis virus | Ardeid wading birds and pigs | Isolated in Japan. Spread to Southeast Asia, Australia | 35,000–50,000 cases of JE, with a mortality rate of 10,000–15,000 people per year |
| Dengue virus | Monkeys and dogs | Caribbean, Central Africa, the Eastern Mediterranean, Southeast Asia, and the Western Pacific | 390 million dengue virus infections per year, 1.3% fatality rate |
| Yellow fever virus | Humans and non-human primates | West, central and east Africa and in South America | 200,000 cases of YFVD and 30,000 deaths annually |
| Tick-borne encephalitis virus | Small rodents and large woodland animals | Europe, Siberia, northern China, Japan, and South Korea | 10,000–12,000 clinical cases of TBE and mortality rate depending on subtype (1% to 20%) |
| Family Filoviridae
Ebola virus |
Bats, Humans and non-human primates, wild antelopes | West and Central Africa | 2013–2016—28,616 cases. The fatality is ranging from 27% to 79% |
| Marburg virus | Fruit bats and monkeys | Central and South Africa | Fatality ratio of up to 88% |
| Family Nairoviridae
Crime-Congo Hemorrhagic Fever Virus |
Wild and domestic animals such as cattle, sheep, and goats | Africa, Asia, and Europe | Fatality ratio (10–40%) |
| Family Peribunyaviridae Rift Valley fever virus | Buffaloes, camels, cattle, goats, and sheep | Sub-Saharan Africa and Arabian Peninsula | Most human cases are mild. 50% mortality within hemorrhagic form |
| Family Togaviridae
Chikungunya virus |
Monkeys, birds, and rodents | Southeast Asia, Europe, Caribbean, North, Central, and South America | 1 million cases per year, the hospital mortality rate raging (10–26%) |
| Family Rhabdoviridae
Rabies virus |
Dogs and cats, foxes, wolves, mongooses, skunks, raccoons, bats | 95% of cases occurring in Africa and Asia | 59,000 human deaths annually |
| Family Retroviridae
HIV |
Originated in non-human primates | Worldwide | 37.7 million people are with HIV, the fatality rate is 1.8% |
| Family Paramyxoviridae
Henipaviruses (Nipah virus and Hendra henipavirus) |
Large fruit bats, pigs, horses | Southeast Asia, Australia, Papua New Guinea | The fatality rate is estimated at 40% to 75% |
| Newcastle disease virus | Poultry and wild birds | Mild systemic ND can be observed in humans | |
| Family Hepeviridae
Hepatitis E virus |
Pigs, wild boar, rats, rabbits, dears, dromedary camel, birds | Worldwide | 3.3 million symptomatic cases with a fatality rate up to 4% in the general population |
| Family Hantaviridae Hantavirus | Rodents, bats, and insectivores | Asia, Europe, and the Americas |
Mortality rates of 12% |
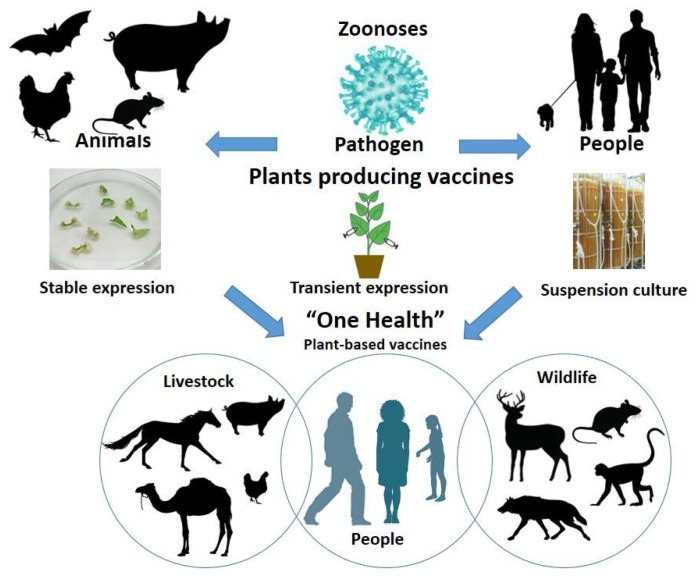
Vaccination is an essential method in eradicating zoonoses and the spread of highly pathogenic viruses and is of great importance to the “Global One Health” paradigm. The Global One Health Initiative’s mission includes efforts to prevent the cross-species transmission of infectious diseases, assess environmental and social impact, and develop adequate science-based risk management policies [9]. An important measure in the One Health Initiative is the creation of widely available vaccines to be used by both humans and animals. The plant expression systems may offer a successful alternative for vaccine production compared to conventional expression systems, especially for animal vaccination (Figure 1).
Figure 1.
Schematic representation of the transmission of zoonotic diseases and the used plant-based production technologies (stable, transient, and suspension cultures) for recombinant vaccine production.
2. Plants as an Expression System for Vaccine Production
For more than three decades, plants have been used for the production of therapeutic recombinant proteins, especially subunit vaccines, and VLP vaccines to cope with emerging and re-emerging diseases. Plants possess great potential in the production of vaccines because they are a safe, cost-effective, and scalable expression system. The achievements of plant molecular farming (PMF) and plant-based production of vaccines and diagnostic reagents have been summarized in a number of review articles [10,11,12,13,14,15,16]. The ability of plants to produce candidate vaccines has been demonstrated in several clinical trials, and two plant-derived vaccines (influenza vaccine and Newcastle disease vaccine) have been approved for commercial use [17,18,19]. Together with the licensing of a plant-derived recombinant human β-glucocerebrosidase, marketed as ELELYSO [20], these achievements promoted plant molecular farming and led to the rapid development of plant expression systems and the overcoming of some of their disadvantages. Increasing the yield [21], modifying plant glycosylation patterns [22], reducing the cost of production and purification [23], and increasing the recombinant protein stability [24,25,26] significantly improved the plant expression systems, making them competitive with the commercially used microbial expression systems. However, despite these achievements, the PMF has not been able to become a technology of choice for the production of pharmaceutical recombinant proteins. There are multiple and complex reasons for the slow progress of PMF. Among them are the strict regulation of GMO and pharma products. In addition, the industry is reluctant to restructure its fermentation infrastructure, and the fact that the productivity of plants, as compared to best practices, is lower also does not help the implementation of PMF in the production of recombinant protein pharmaceuticals [14]. Still, we may be witnessing a breakthrough thanks to the Medicago’s success in launching influenza and SARS-CoV-2 vaccines, which have undergone phase-3 clinical trials and are currently awaiting regulatory approval. As a result of the rapid production of large quantities of recombinant proteins within less than one week due to the transient expression approach, plant-based vaccines are becoming very attractive option when there is urgent need caused by the swift transmission of emerging pandemic viruses.
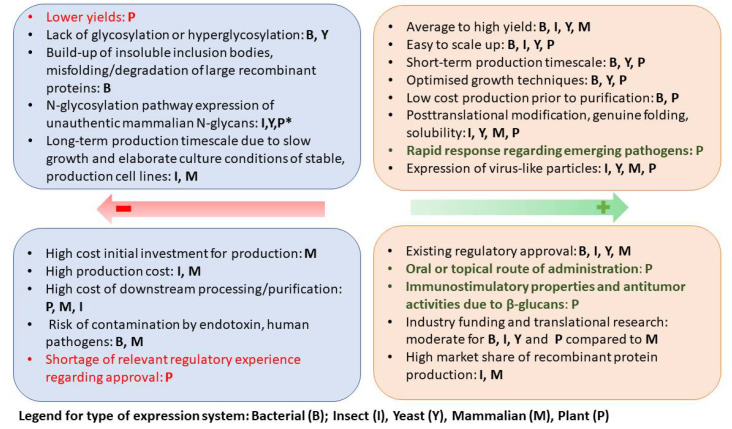
The advantages and disadvantages of the most commonly used systems for vaccine and therapeutic proteins production (Yeast, E. coli, insect cells, Chinese hamster ovary (CHO) cells, and embryonated hen’s eggs (EHE)), and their comparison with plant expression systems have been extensively reviewed by several authors [14,27,28,29]. Figure 2 summarizes the main characteristics of the used bio-pharming systems and highlights the advantages and disadvantages of plant expression systems.
Figure 2.
Comparison between plant expression systems and conventional expression systems. * N-linked glycans in humans differ from plant glycans, and the latter’s strong immunostimulatory effect may cause plant-derived therapeutics to have adverse events; however, these same properties are beneficial for vaccines as they enhance immunogenicity.
The early concept for producing cheap and easy-to-use edible vaccines, created by Charles Arntzen [30], has been studied for decades. Edible vaccines were developed, and some were used in clinical trials [31,32,33] but were later dismissed, especially for humans, mainly because of the impossibility of precise dosage of the recombinant protein within the living plant [14]. This concept of edible vaccines is still being developed for veterinary purposes [34,35,36]. Plant-made vaccines for veterinary use are very promising, mainly because of the possibility of oral delivery of the recombinant vaccine, at low cost and with a long shelf life, especially in cases where cereals are used.
Plant molecular farming is based on three main technological platforms: transient expression (generally in Nicotiana benthamiana), stable transgenic plants (transgenic and transplastomic), and plant cell-suspension cultures [37,38,39,40]. Transient expression in plants provides a higher yield of recombinant proteins within a shorter timeline than the stable modified plants and has been used successfully as a rapid response platform addressing emerging viral diseases such as Influenza and COVID 19. The production of recombinant proteins using transient expression based on RNA viral vectors has gained wide popularity due to numerous advantages: simple and easy engineering, high level of production of the protein of interest, scalability, lower risk, and lower costs [41]. Mainly small (+) RNA viruses such as tobacco mosaic virus (TMV) [42], cowpea mosaic virus (CMV) [43], and potato virus X (PVX) [40] are utilized as viral vectors for commercial protein production [44,45]. Earlier, the virus-based vectors expressed the gene of interest in addition to the virus’s own natural genes. Later on, the viruses were modified by removing all unessential elements for target protein expression, resulting in higher yield and better stability of the viral complex [46]. Transient expression, also known as agroinfiltration, involves the introduction of genes into plant leaves by infiltrating them with disarmed Agrobacterium tumefaciens carrying binary vectors [47]. The advantages of this approach have led several companies (Medicago Inc. (Québec, QC, Canada); iBio/Caliber Therapeutics (Bryan, TX, USA); Kentucky BioProcessing Inc. (KBP), Owensboro, KY, USA; Fraunhofer USA (Plymouth, MI, USA)) to invest in this plant-based platform for rapid and large-scale production of vaccines and therapeutics [48].
Stable transformation of plants takes longer, but once optimized, the transgenic system allows long-term and large-scale recombinant protein production. Various plant species such as tobacco, potato, cereals, lettuce, tomato, carrot, alfalfa, and oilseeds have been used for stable transformation by nuclear or chloroplast genome manipulation. The main approaches used to achieve stably transformed plants are Agrobacterium-mediated transformation or biolistic process [39,49]. The chloroplasts are sometimes a preferable target for stable transformation instead of the nuclei due to their high copy number in the plant cell, leading to high recombinant protein production.
The choice of technology to achieve a plant-based vaccine depends on the route of vaccine administration, the ability to achieve high levels of recombinant protein expression, and a low value of downstream processing. Edible plants (tomato, carrot, lettuce, rice, and maize) are suitable for oral administration. Cereal crops are preferable for long storage. Tobacco plants are preferable as model plants and to achieve high levels of expression.
The use of bioreactors from plant cell cultures in PMF makes it possible to overcome some of the shortcomings of transgenic crops. In vitro growing of plant cells under controlled conditions allows for the precise monitoring of cell growth and protein production and the development of good manufacturing practices. The approach of producing valuable pharmaceutical proteins in plant cell bioreactors is becoming increasingly common due to strong restrictive measures on the use of transgenic plants in the field, especially in Europe [50].
3. Virus-like Particles (VLPs) for Vaccine Development
VLPs are recognized as safe and effective vaccines against viral diseases [11,51]. They resemble regular viruses’ outer structure, composition, and size, but they lack genetic material, which makes them non-infectious [52]. Because VLPs retain the native antigenicity of the viruses they mimic, they can efficiently elicit cellular and humoral immune responses [53]. The virus-like particles can be formed from structural proteins belonging to one type or multiple types of viruses. Those composed of structural proteins or immunogenic epitopes from different viruses are known as chimeric VLPs [54,55,56,57]. Such VLPs can be used as immune modulators and self-adjuvants to provoke strong immune responses against the presented immunogenic epitopes [58,59,60]. The use of virus-like particles has also been recently utilized by the PMF community [54,61,62]. VLPs-based candidate vaccines expressed in plants have been developed against several zoonotic diseases and the efficacy of these vaccines is currently being evaluated. Chimeric plant-derived VLP vaccines are also being developed against diseases such as cancer, allergies, and autoimmune diseases [63].
4. Influenza Viruses
Influenza viruses are members of the family Orthomyxoviridae and include four genera (Influenza virus A, B, C, and D) [64]. Only influenza A and B are clinically significant for human health [65]. While influenza B infects only humans, influenza A infects humans and a wide variety of mammals and birds. The influenza A virus is zoonotic and can generate pandemic viruses by switching the host. Influenza is an enveloped virus with a spherical or pleomorphic shape, and is 80–120 nm in diameter. Its genome contains eight segments of single-stranded negative-sense RNA. The lipoprotein envelope of the influenza A virus contains two glycoproteins, hemagglutinin (HA), and neuraminidase (NA) [66]. After infection, HA and NA are the primary antigens inducing antibody production [67]. To date, 18 HA and 11 NA gene variants have been identified [68]. Of these variants, only four HA (H1, H2, H3, and H5) and two NA (N1 and N2) are considered as potentially serious pandemic threats [69]. HA is a stronger antigen than NA; thus, it is the main object of interest for vaccine development against influenza.
The World Health Organization (WHO) estimated that the flu kills about 250,000 to 500,000 people annually [70]. There is a shortage of influenza vaccines globally, and the ability for a “rapid response” vaccine production against pandemic influenza strains needs to improve [13]. Plant molecular farming technology can address these needs because it provides fast development and manufacturing of vaccines and scalable production. In 2009, the US Defense Advanced Research Projects Agency (DARPA) invested USD 100 million in four companies (Fraunhofer USA, the Center for Molecular Biotechnology in Delaware, Kentucky Bioprocessing, and Medicago Inc.) to produce 100 million influenza vaccines in plants in a month [71]. In 2012, Medicago produced 10 million doses of the H1N1 vaccine in a one-month “rapid fire test” [72]. They were able to achieve that due to many years of scientific development and the use of transient expression vectors [43], providing synthesis and accumulation of large quantities of recombinant proteins in plants within a week. D’Aoust et al. [73] demonstrated the production of VLPs composed only of influenza H5 by means of transient expression in N. benthamiana. Animal studies with low doses HA VLPs showed protective immunity in mice. The development of the candidate monovalent VLP vaccines against H5N1 pandemic strains [74,75] and the polyvalent HA VLPs for seasonal flu, which successfully passed phase-3 clinical trials [17,62,76] demonstrate the great potential plant-based technologies have in the future of vaccine production. These studies show that plant-derived HA VLPs candidate vaccines can provide protection against respiratory illnesses caused by influenza viruses in humans. Medicago Inc. has successfully completed phase-3 clinical studies for a plant-derived VLP quadrivalent flu vaccine (NCT03321968, NCT03301051, NCT03739112) [77,78]. The reported high efficacy of this plant-made flu vaccine is an important milestone in the progress of PMF.
In addition, the efficient production of hemagglutinin-based VLP vaccines in N. benthamiana has been demonstrated in academic studies by Rybicki et al. [13] and Smith et al. [79]. In Smith’s study, H6-subtype VLPs were transiently expressed and evaluated for efficacy in chickens against the heterologous H6N2 virus. Their findings demonstrate the potential of the plant-produced H6N2 HA vaccine for poultry. HA from various influenza strains was expressed in plants, and its immunogenicity was assessed [80,81,82,83,84,85,86,87,88,89]. Modifications of the HA structure were made to achieve a high level of recombinant protein accumulation: the sequences were optimized, the transmembrane domain and native signal peptide were removed, and an endoplasmic retention signal was inserted at the C terminus. The yield of the transiently expressed protein was HA variant dependent. The yield of H3 was 200 mg/kg of fresh weight (FW) tobacco leaves [81], while the yield of H1 was 400–1300 mg/kg FW [82]. Shoji et al. generated trimeric HA, which mimics the authentic HA structure, by introducing a trimerization motif from a heterologous protein into the HA sequence [83]. Immunization with the generated HA induced a protective immune response upon challenge of mice with a lethal viral dose [83]. Musiychuk et al. developed a plant expression system that achieved high-level target antigen expression in plants by engineering a thermostable carrier molecule fused to HA from the influenza A/Vietnam/04 (H5N1) virus [90].
Stable expressions of immunogens from the influenza virus are less used compared with transient expressions. In a review paper, Redkiewicz et al. summarized the results achieved from different plant expression systems for the production of HA [91]. Here, we will only mention some of the latest results achieved by stable expression of HA: avian H5N1 HA was found to be stably expressed in tobacco seeds [92] with a yield of 3.0 mg of the viral antigen per g of seed; and H3N2 nucleoprotein was detected in transgenic maize with a yield of 35 µg of NP/g seed [93]. The relatively low levels of recombinant protein accumulation in stable transgenic plants indicate that transient expression is more promising for developing an influenza vaccine in plants.
The feasibility of plants for the production of chimeric VLPs presenting the extracellular domain of the M2 influenza protein (M2e) as a candidate for universal influenza vaccines has also been investigated. Hepatitis B core protein (HBcAg), Hepatitis E open reading frame 2 (ORF2) capsid protein, Human papillomavirus 16 (HPV-16) L1 protein, Cowpea Mosaic Virus (CPMV), and tobacco mosaic virus (TMV) have been used as scaffolds of the M2e influenza peptide [60,94,95,96,97,98,99]. The immunogenicity of some of the chimeric VLPs presenting M2e was assessed in mice, and they often demonstrated a high immune response with protective activity [88,91,92,93]. Blokina et al. developed a chimeric protein combining the M2e and HA2 influenza A antigens with bacterial flagellin, which has adjuvant properties. The chimeric recombinant protein was expressed using a highly efficient PVX-based expression system in N. benthamiana, resulting in protein accumulation of up to 300 µg/g of fresh leaf tissue. Mice that were intranasally immunized with the purified chimeric protein survived a lethal challenge with the influenza A virus strain A/Aichi/2/68 (H3N2) [100].
5. Emerging Coronaviruses
Within the last twenty years, the emergence of three novel coronaviruses causing severe acute respiratory syndrome was observed: SARS-CoV-1, MERS-CoV, and SARS-CoV-2. They have the crown-like protruding knobs on their surface that are typical for all coronaviruses, a large, positive-strand RNA genome of approximately 30,000 nucleotides, and are classified in the genus Betacoronavirus [101]. Genus Betacoronavirus also harbors the so-called “common cold” human coronaviruses hCoV-OC43 and hCoV-HKU [102]. The virion is composed of four structural proteins, spike (S), envelope (E), membrane (M), and nucleocapsid (N), all of which are immunogenic, but only the spike (S) protein gives rise to neutralizing antibodies [103,104]. The swift spread of the SARS-CoV-2 virus worldwide, infecting billions of people with millions of fatalities, sent an alarming message to the scientific community that there is a need for rapid development of effective vaccines against viruses such as the SARS-CoV-2. Researchers working in plant molecular farming have been prompted to analyze how plant-based production systems can meet this need [28,105,106,107]. The urgent need for a COVID-19 vaccine and diagnostic reagents has been addressed by the rapid and large-scale production of SARS-CoV-2 structural proteins in plants.
The first report of a successful plant-produced vaccine candidate against SARS-CoV-2 was from the Canadian company Medicago Inc. [108]. The strategy included transient expression in N. benthamiana of the modified SARS-CoV-2 S protein with stabilizing point mutations, a plant signal peptide, and the influenza HA transmembrane domain and cytoplasmic tail replacing the equivalent sequences in SARS-CoV-2. Medicago successfully produced and purified modified VLPs built from the S protein. Following positive Phase-1 results, Medicago has launched Phase-2/3 (ClinicalTrials.gov Identifier: NCT04636697) and Phase-3 clinical trials (NCT05040789) (Table 2). Recently, Medicago Inc. and GlaxoSmithKline announced a 71% efficacy rate of their plant-based vaccine against all variants of SARS-CoV-2 [109].
Table 2.
| NCT Number | Study Title | Phase | Responsible Party |
|---|---|---|---|
| NCT04450004 | Safety, Tolerability and Immunogenicity of a Coronavirus-Like Particle COVID-19 Vaccine in Adults Aged 18–55 Years | 1 | Medicago |
| NCT04636697 | Study of a Recombinant Coronavirus-Like Particle COVID-19 Vaccine in Adults | 2/3 | Medicago |
| NCT05040789 | Phase-3 Study to Evaluate the Lot Consistency of a Recombinant Coronavirus-Like Particle COVID-19 Vaccine | 3 | Medicago |
| NCT05065619 | Safety Immunogenicity Study of MT-2766 in Japanese Adults (COVID-19) | 1/2 | Medicago |
| NCT04473690 | KBP-201 COVID-19 Vaccine Trial in Healthy Volunteers | 1/2 | KBP |
| NCT04953078 | A Study to Evaluate Safety, Tolerability, and Reactogenicity of an RBD-Fc-based Vaccine to Prevent COVID-19 | 1 | Baiya Phytopharm |
The American biotech companies iBio [112] and Kentucky BioProcessing (KBP) [113] also announced that they have produced subunit vaccines against SARS-CoV-2 based on plants. KBP used transient expression in N. benthamiana plants for the KBP-201 vaccine production. The KBP-201 with CpG oligonucleotides as an adjuvant is currently in phase-1/2 clinical trials (Table 2; ClinicalTrials.gov Identifier: NCT04473690). iBio produced a VLPs-based vaccine (IBIO-201) containing S protein linked to the LicKMTM booster molecule. IBIO-201 demonstrated that it can elicit an anti-SARS-CoV-2 immune response in preclinical studies [114].
Baiya Phytopharm, a biotech company in Thailand, used N. benthamiana for developing a subunit-based vaccine against SARS-CoV-2. Their candidate vaccines were tested for immunogenicity in mice and monkeys. The Baiya SARS-CoV-2 Vax 1 candidate vaccine is currently in phase-1 clinical trials (Table 2; ClinicalTrials.gov Identifier: NCT04953078) [115].
The Lomonossoff group, John Innes Center, Norwich, also announced SARS-CoV-2 candidate vaccine production based on plants expressing the S, E, and M proteins. They purified “crown-shaped” particles from plant leaves infiltrated with the S protein [116]. Pogrebnyak et al. were also successful in generating S1 protein composed of the N-terminal and the receptor-binding domains in tobacco and tomato plants and observed robust immune response upon immunization of mice [117]. Another study described the expression of the S1 protein in lettuce and transplastomic tobacco. However, immunological studies have not yet been reported [118]. Zheng et al. [119] expressed the SARS-CoV N protein, and Demurtas et al. [120] expressed the M and N proteins in N. benthamiana and demonstrated their immunogenicity in animal models. Mamedov et al. co-expressed the N protein with the receptor-binding domain (RBD) in N. benthamiana to produce the SARS-CoV-2 antigens in plants. The cocktail of these two antigens elicited high-titer antibodies against SARS-CoV-2 [121]. Maharjan et al. also expressed RBD and proved that the plant-derived recombinant RBD elicited a humoral immune response in mice [122]. The overall effort to produce plant-derived SARS-CoV-2 VLPs suitable for vaccine development in a short period of time is unprecedented [123,124,125,126].
6. Filoviruses
6.1. Ebola Virus (EBOV)
EBOV causes severe hemorrhagic fever in humans and non-human primates. It has been associated with periodic outbreaks in Central Africa, and it also has the potential to be used as a bioterrorism agent [127]. The Ebola virus belongs to the family Filoviridae, genus Ebolavirus [128]. EBOV virion has a negative-sense RNA genome encoding seven gene products: four nucleocapsid proteins (NP, VP35, VP30, L); two membrane-associated proteins (VP24, VP40); and a transmembrane glycoprotein (GP) [129,130,131]. The GP mediates the viral entry process into cells and is the key immunogenic protein in vaccine protection [132,133]. In recent years, remarkable success has been achieved in developing candidate vaccines against filoviruses in non-human primate models and clinical trials [134,135,136,137]. Plant expression systems offer cost-effective alternatives to conventional vaccine production systems against EBOV. Nieto-Gómez et al. designed a multiepitopic protein called Zerola, carrying epitopes from the EBOV glycoprotein (GP) [138]. They demonstrated the ability of nuclear-transformed tobacco lines to produce Zerola antigenic protein and created a plant-based, low-cost candidate vaccine against EBOV. Phoolcharoen et al. developed a novel approach by fusing EBOV glycoprotein to the heavy chain of 6D8 mAB and co-expressing it in N. benthamiana [139]. After purification, this product formed immune complexes (EIC). Mice immunized subcutaneously with plant-derived EIC produced anti-EBOV antibodies at levels comparable to those obtained with GP1 virus-like particles, demonstrating the effectiveness of the plant-expressed EIC as a vaccine candidate [139,140].
6.2. Marburg Virus (MV)
The Marburg virus is a part of the same family as the Ebola virus, causing a highly infectious zoonotic hemorrhagic fever. It also has the potential to be used as a biological weapon. To date, there is no published evidence on the creation of a plant-made vaccine against the Marburg virus. Future efforts exploring the generation of such a vaccine can be based on and expand the current knowledge of EBOV candidate vaccines.
7. Bunyavirales
7.1. Crimean–Congo Hemorrhagic Fever Virus (CCHFV)
CCHFV causes tick-borne zoonosis in over 30 countries in Africa, Asia, and Europe [141,142]. Humans are infected mainly by tick bites, particularly from the Hyalomma genus, and by exposure to the tissue and blood of infected animals, which are asymptomatic [143]. The CCHFV is an enveloped single-stranded negative-sense RNA virus with a tri-segmented genome. It belongs to order Bunyavirales, family Nairoviridae, Orthonairovirus genus [144]. The RNA segments, S (small), M (medium), and L (large), encode the viral nucleoprotein (N), the glycoproteins Gn and Gc, and the RNA-dependent RNA polymerase [145]. Due to the lack of approved vaccines, high pathogenicity, and the increasing spread of CCHFV, the development of a vaccine and the use of plants for its production is a viable alternative. Ghiasi et al. expressed the Gn/Gc glycoproteins in transgenic tobacco leaves and hairy roots. Oral immunization of mice with transgenic leaves and roots elicited specific IgA and IgG production [146]. The use of an edible animal vaccine may reduce the risk of zoonotic transmission of CCHFV. An attractive antigen for CCHFV vaccine production is the nucleoprotein (N) [147]. To date, no expression of the N protein in plants and assessment of its immunogenicity have been reported.
7.2. Rift Valley Fever Virus (RVFV)
RVFV causes severe epidemics among ruminants and humans in Africa and the Arabian Peninsula [148]. In animals, RVFV is a vector-borne infection mainly transmitted by the Aedes and Culex mosquito [149,150]. Humans are often infected by close contact with infected animals and rarely through bites from infected mosquitoes [151,152]. The RFV disease symptoms range from uncomplicated acute febrile illness to severe hemorrhagic disease and death [149]. Similarly to the other arboviruses (Zika virus, Chikungunya virus, and Dengue virus), RVFV may emerge worldwide due to the widespread distribution of arthropod vectors. RVFV belongs to the family Peribunyaviridae, genus Phlebovirus [153]. The RVFV genome contains three single negative-stranded RNA, packaged into viral nucleocapsid protein (N) and enveloped by a lipid bilayer containing two viral glycoproteins (Gn/Gc) [154,155]. The main targets for vaccine production are the Gn and N proteins. The Rybicki group described the production of chimeric RVFV virus-like particles transiently expressed in N. benthamiana [156]. They replaced Gn’s ectodomain (Gne) and fused it to the transmembrane domain of avian influenza H5N1 HA, which allowed the chimeric VLPs to be produced and purified. The generation of Gn-HA chimeric RVFV VLPs in plants was the first demonstration of its kind to elicit a specific antibody response in mice [156]. Kalbina et al. expressed the N and Gn proteins in Arabidopsis thaliana and demonstrated that the orally administrated proteins are immunogenic in mice. No VLP formation has been reported [157].
7.3. Hantaviruses
Hantaviruses (HVs) are emerging pathogens belonging to family Bunyaviridae and are known for causing hantavirus pulmonary syndrome or hemorrhagic fever with renal syndrome depending on the geographical location (North America, or Europe and Asia) [158]. HVs are transmitted to humans from their natural reservoirs, rodents, through contact with infected animals and their excrements. Annually, these viruses cause approximately 200,000 infections in humans worldwide [159,160]. The Hantavirus genome comprises three segments, named small (S), medium (M), and large (L). The L segment encodes the viral polymerase, the M segment encodes the envelope glycoproteins Gn and Gc precursor (GPC), and the S segment encodes the viral nucleocapsid protein (N) [161]. The N protein and the Gn and Gc glycoproteins are promising candidates for vaccine development against HVs [159,162]. Kehm et al. generated transgenic tobacco and potato plants expressing the Hantavirus Puumala N protein and observed specific IgG and IgA immune responses upon oral or intraperitoneal immunization of rabbits [163]. Mice that were orally immunized with recombinant Puumala virus N protein did not induce an immune response. Khattak et al. demonstrated that the recombinant viral protein was completely degraded by trypsin and/or pepsin [164], which could explain the negative outcomes of the oral immunizations.
The Hepatitis B nucleocapsid tolerates the insertion of foreign sequences at its c/e1 region and was used to display several immunogenic epitopes for vaccine development in plants [56,94,98,165]. A number of research teams have used a similar approach to create chimeric HBc VLPs presenting various HV immunogenic epitopes, which have never been used in plants [166,167,168,169,170]. Geldmacher et al. successfully inserted a gene fragment encoding 120 amino acids of the N gene from two different hantavirus strains (Dobrava and Hantaan) into the hepatitis B nucleocapsid gene, resulting in a chimeric formation of VLPs exposing the inserted foreign protein segment on the surface [167]. These chimeric VLPs elicited cross-reactive antibody responses to the two HV strains [167,171]. Plant molecular farming researchers can use similar approaches for the generation and production of vaccines against HVs.
8. Togaviruses
Chikungunya Virus (CHIKV)
CHIKV is an arbovirus transmitted by mosquitoes. It causes fever, joint pain, and acute polyarthritis [172,173]. Although the CHIKV disease is non-lethal and self-limiting, more than 30% of infected individuals can develop chronic joint pain [174]. In 2004 the CHIKV had a major outbreak spreading from sub-Saharan Africa and Southeast Asia [175]. The virus is classified as a member of the Togaviridae family, genus Alphavirus [176]. The RNA genome has two open reading frames (ORF); ORF2 encodes five structural proteins (capsid (CP), three envelope glycoproteins (GPs E1, E2, and E3), and 6K viroporin) [177,178]. Since there are no vaccines or treatments against the CHIKV infection, the use of plants can offer a fast and cost-effective platform for vaccine production. The CHIK-VLPs expressed in yeast and insect cells have been explored as vaccine candidates against CIKV. The CHIK-VLPs are morphologically identical with the native virion, and they induce cellular and humoral immune responses [179,180,181,182,183]. These studies were taken a step further by expressing ORF2 in plants to produce CHIK-VLPs. The Lomonossoff group produced capsid-like particles (CLPs) in N. benthamiana through transient expression of ORF2 or a fragment of the ORF2 coding capsid protein [116]. The results show that C proteins can be produced in plants following autoproteolytic cleavage of the expressed polyprotein. Not all envelope glycoproteins could be expressed in plants, only E2 was detected, and no structures resembling full CHIK-VLPs were observed [116].
9. Flaviviruses
The viruses from the genus Flavivirus, family Flaviviridae, cause many diseases transmitted to humans through vectors such as mosquitoes and ticks. West Nile virus (WNV), Dengue virus (DENV), Zika virus (ZIKV), Yellow fever virus (YFV), Japanese encephalitis virus (JEV), and Tick-Borne encephalitis virus (TBEV) are causing the most common flaviviral infections [184,185].
9.1. West Nile Virus (WNV)
West Nile virus is an emerging mosquito-borne flavivirus with a wide geographical distribution that includes part of Europe, Africa, the Middle East, south and central Asia, and Australia [186]. The transmission cycle of WNV is birds–mosquitos–birds, with migratory birds being primarily responsible for the virus dispersal, including the reintroduction of WNV from endemic areas into areas with sporadic outbreaks [186,187,188]. The WNV infection is usually asymptomatic (in around 80% of the infected people), or it could develop into a West Nile fever (headache, sore throat, backache, and diarrhea) or a severe West Nile disease [189,190,191]. WNV is a small (40–50 nm) spherical icosahedral virus. The outer protein shell is enveloped by a lipid membrane derived from the host cell [192]. The RNA genome is enclosed into a polyprotein that is cleaved post-translation to yield three viral structural proteins: nucleocapsid (C), pre-membrane (prM), and envelope glycoprotein (E), and seven nonstructural proteins (NSPs) [193,194,195]. The envelope glycoprotein that mediates viral entry into host cells by binding to cellular receptors is also the major antigenic determinant targeted by the host [196,197]. The WNV E protein shares a similar structure with the flavivirus E glycoproteins. With its C-terminal DIII (wDIII) domain, which contains epitopes recognized by neutralizing antibodies, the E proteins interact with cell receptors during viral transmission [198,199].
The envelope protein and its DIII domain are in the main focus for vaccines against WNV. Vaccines have been developed for horses, but there is no approved vaccine against WNV for human use. As reviewed by Filette et al. [195], the development of a cost-effective vaccine for veterinary use could be a solution to eradicate the virus spread and to control WNF disease in humans and animals. Therefore, many research groups explored making WNV recombinant vaccines made in plants as a safe and cost-effective means of production. The Qiang Chen research group from Arizona State University has a number of candidate vaccines developed against WNV based on the DIII domain of the E protein (wEDIII) [200]. They demonstrated that plants successfully produced wEDIII domains with an average level of 100 µg/g leaf fresh weight. The plant-derived wEDIII protein was immunogenic and potentially protective in mice [201,202,203]. Chen et al. also produced the entire E protein- and prM-E-based WNV VLPs in plants. Only the results of sucrose gradient centrifugation suggested the assembly of prM-E VLPs [51]. They also fused wEDIII to HBcAg, and produced chimeric VLPs, which elicited strong B and T-cell responses against WNV in mice [204].
The latest study of the Rybicki group reported the development of chimeric AP205 phage virus-like particles displaying WNV envelope EDIII domains. They used the SpyTag/SpyCatcher (ST/SC) conjugation system [205] to generate a virus-like particle-display-based vaccine candidate in N. benthamiana. A quantity of 5 μg of purified chimeric VLPs subcutaneously applied in mice elicited a potent antibody response to WNV EDIII [206].
9.2. Dengue Virus (DENV)
Dengue Virus is an emerging arbovirus that is distributed in tropical and subtropical areas of the world by mosquitos (Aedes aegypti and A. albopictus). The WHO estimates that worldwide, more than 3.6 billion people are at risk of being infected by DENV. Of them, annually, more than 200 million will be infected, with 21,000 fatalities [207]. Dengue is an acute febrile viral disease that may proceed asymptomatically or result in self-limited dengue fever, but it could also escalate into severe dengue hemorrhagic fever and dengue shock syndrome [208]. Therefore, the development of a scalable and safe vaccine is needed, and plants may offer the solution. Similar to all other Flaviviruses, DENV’s genome is made up of a positive-sense single strand RNA, which has a single ORF encoding all structural and non-structural proteins into one polyprotein. The polyprotein is then proteolytically cleaved into three structural proteins (capsid, precursor membrane, and envelope) and at least seven non-structural (NS) proteins [209]. DENV has four antigenically distinct serotypes (DENV-1-4) [210]. The dengue DIII domain of the E protein (dEDIII) can induce neutralizing antibody responses and is the main target for vaccine development [211,212]. There are several examples of expression of dEDIII from different serotypes and prM in plants [213,214,215,216,217,218]. dEDIII was expressed in N. tabacum by stable nuclear [219] or chloroplast transformation [216]. Transient expression of dEDIII in N. benthamiana leads to higher recombinant protein quantities than stable expression, 0.28% [212,219] vs. 0.13 to 0.25% [216], respectively [212,216,219]. The development of oral vaccination through the expression of tetravalent EDIII in lettuce chloroplasts was explored by van Eerde et al., showing that EDIII-1-4 can induce an immunogenic effect in rabbits [217].
Martínez et al. [215] used a strategy to display epitopes of dEDIII on the surface of HBcAg capsid-like particles. Using the same strategy, Pang et al. [220] inserted a consensus sequence of dEDIII into the immunodominant c/e1 loop region of tandem HBcAg and demonstrated the production of chimeric VLPs (tHBcAg-cEDIII) in N. benthamiana [165]. A recent study conducted by the Lomonossoff group (Ponndorf et al. [221]) described the production of DENV VLPs in N. benthamiana. Using the transient co-expression of DENV structural proteins (SP) and truncated versions of the non-structural proteins (NSPs), they successfully purified DENV VLPs. Immunogenicity assays revealed that the plant-made DENV VLPs led to a high antibody response in mice.
Existing Ebola recombinant immune complex (RIC) technology [139] was used by Kim et al. to develop a Dengue RIC technology (DERIC). A dEDIII gene fragment was fused with the heavy chain of 6D8 mAB to prepare a chimeric Dengue–Ebola recombinant immune complex. DERIC induced a strong virus-neutralizing anti-cEDIII humoral immune response without using adjuvants in subcutaneously immunized mice and showed the potential to be a candidate vaccine [218].
9.3. Zika Virus (ZIKV)
The vector for Zika virus transmission is primarily Aedes aegypti mosquito, albeit several other species are also involved (A. africanus, A. albopictus, A. hensilli) [222]. Zika virus is also transmitted through sexual contact, blood transfusions, and from mother to fetus during pregnancy [223]. The greatest danger for humans is the vertical transmission during pregnancy, resulting in the development of fetal microcephaly [224]. In 2016, the WHO declared the Zika virus a “public health emergency of international concern” when Brazil reported an association between Zika virus infection and microcephaly [225]. ZIKV infections in humans are usually asymptomatic or characterized by fever and cutaneous rash [226]. Two genotypes of ZIKV are distinguished, African and Asian [227]. ZIKV is a Flavivirus with a genome organization and virion structure similar to the other flaviviruses. The flaviviruses’ genetic similarity can provoke the antibody-dependent enhancement of infection (ADE), and thus, can present challenges for vaccine development [228,229]. The major virion surface glycoprotein E is responsible for attachment to cellular receptors, membrane binding and fusion for viral entry, and mediating viral assembly [230]. The zE glycoprotein, with its DIII domain, has great potential to induce potent neutralizing antibodies, making it a principal candidate for a subunit vaccine [231]. Chen et al. developed a zE-based candidate subunit vaccine by means of transient expression in N. benthamiana plants. The plant-derived envelope protein elicited potent zE-specific antibodies and cellular immune responses that correlate with protective immunity against the Zika virus in mice [232]. Chen et al. also devised VLPs carriers based on the HBcAg that presented zDIII, showing that chimeric VLPs can be easily purified in large quantities from plants and can elicit a potent humoral and cellular immune response in mice [233]. Diamos et al. demonstrated that recombinant immune complexes (RIC) are a potent platform to improve the immunogenicity of weak antigens by fusing a ZIKV envelope domain III (ZEIII) to the IgG heavy chain (N-RIC) [234]. In mice, the IgG fusions elicited up to 150-fold high titers of Zika-specific antibodies, compared to the ZEIII antigen alone, which neutralized ZIKV using only two doses without an adjuvant [234].
Interestingly, codelivery of hepatitis B nucleocapsid/zEDIII VLPs with RIC zEDIII was capable of producing a synergistic effect in terms of enhancing the immune response [235].
The Lomonossoff group transiently expressed ZIKV prM-E proteins in N. benthamiana and showed that plants successfully expressed and accumulated E proteins. The protein yield was low, which led to limited VLP purification due to poor VLP formation [116].
9.4. Yellow Fever Virus (YFV)
The mosquito-transmitted yellow fever virus causes high fatality outbreaks in South America and Africa [236]. According to the WHO, annually, YFV causes 200,000 severe cases of yellow fever (YF) and up to 60,000 deaths [236,237]. The RNA genome of YFV encodes seven non-structural (NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5) and three structural proteins (envelope (E), membrane (M), and capsid (C)) [238]. Although there is an effective live attenuated vaccine against YFV (YF-Vax®, Sanofi-Pasteur, Lyon, France, and 17DD-YFV, Bio-Manguinhos, Rio de Janeiro, Brazil), the increased demand and reports of severe reactions associated with the live vaccines have provoked interest in the development of safer subunit vaccines against YFV.
Two different constructs, (a) the gene coding for the envelope (E) protein alone or (b) a fusion of E with enzyme lichenase gene (YFE LicKM), were transiently expressed in N. benthamiana. Both plant-derived proteins, YFE and YFE-LicKM, elicited virus-neutralizing antibodies in mice and protected over 70% of them from a lethal challenge infection [239]. Currently, there are no reports describing the development of a safe and efficacious vaccine against YFV in plants using the VLP approach. In their review, Hansen et al. describe, in detail, the different platforms for creating a vaccine against YFV [240].
9.5. Japanese Encephalitis Virus (JEV)
Japanese encephalitis is an arbovirus that causes 45,000 human encephalitis cases and 10,000 deaths globally [241]. The primary amplifying hosts are pigs, while water birds are carriers and mosquitoes are vectors [185]. JEV is an enveloped virus with a positive single-stranded RNA genome. The JEV genome has a single ORF encoding three structural proteins (C, prM, and E), and seven non-structural proteins (NS1, NS2A, NS2B, NS3, NS4A, NS4B, and NS5) [242]. The envelope protein (E) is the primary target of neutralizing antibodies. The recombinant JEV E protein, expressed in the baculovirus expression system, induced a neutralizing antibody response and protective immunity against a lethal challenge with JEV [243]. Plants’ expression systems have been used successfully for the production of the E surface glycoprotein. Wang et al. developed transgenic rice expressing the E protein with a yield of 1.1–1.9 μg/mg of total soluble protein. Intraperitoneal administration in mice of plant-derived E protein elicited specific neutralizing antibodies. In addition, also in mice, the oral administration of the E protein induced a mucosal immune response [244]. Chen et al. used a different approach. They created chimeric virus-like particles (CVPs) based on the bamboo mosaic virus (BaMV) to present domain III of the E protein. The CVPs were found to elicit neutralizing antibodies against JEV infection in mice [245].
9.6. Tick-Borne Encephalitis Virus (TBEV)
Tick-Borne encephalitis virus is transmitted by Ixodes spp ticks and causes a potentially fatal neurological infection known as tick-borne encephalitis (TBE). The virus is endemic to Europe, Siberia, northern China, Japan, and South Korea [246]. Each year, more than 10,000 cases of TBE are registered, of which 3000 in Europe, and in terms of morbidity, this frequency is second only to JEV among the neurotropic flaviviruses [247,248]. TBEV has the same genome organization as the other Flaviviruses. The TBEV genome in one open reading frame codes for the expression of three structural (C, prM, and E) and seven non-structural proteins. Specifically, for TBEV, prM and E form smaller, capsid-less virus-like icosahedral particles. These particles do not have the structural protein C and RNA, and they are non-infectious [247]. The recombinant prM and E VLPs and virion-derived particulate immunogens are highly immunogenic and can protect animals. In addition, the M/E protein has also been used in diagnostic assays [249,250,251].
No studies have been reported on the M and E expression in plants, with the exception of the announced project “Development of edible vaccines against Tick-Borne encephalitis” [252] of Örebro University, Sweden.
10. Human Immunodeficiency Viruses (HIV)
Human immunodeficiency virus (HIV) is classified as a member of the Lentivirus genus of the Retroviridae family [253,254]. Phylogenetical analysis showed close simian relatives of HIV-1 in chimpanzees [255] and sooty mangabeys [256], demonstrating the zoonotic origin of this virus [257]. Genetically, there are two types of HIV (HIV-1 and HIV-2). Of the two, HEV-1 has spread rapidly worldwide, and it is the primary cause of the global HIV pandemic [258].
Vaccine development against HIV is still a challenge due to the hyper-variability of the viral envelope (Env) glycoprotein, which results in immune evasion [259]. To address the global needs for HIV prevention, plants represent a viable option as a protein expression system [13,260,261].
A promising approach for an HIV vaccine is the development of artificial proteins based on conservative T-cell and B-cell epitopes. Notably, the gp41 and gp120 regions of HIV-1 are crucial to virus function and immune protection, making them valuable targets for vaccine investigation [262]. Multiple polyvalent HIV proteins have elicited immunological responses in animals (Table 3). For example, the C4(V3)6 protein, based on gp120, expressed in lettuce, induced an immune response in mice [263]. The p24 protein, expressed transiently or via transplastomic or transgenic transformation, induced a humoral immune response [264,265,266]. A small epitope from gp41 displayed on the surface of CPMV stimulated an antibody response in mice [267]. HBsAg has also been utilized as a carrier for HIV polyproteins, providing excellent immunogenic characteristics while being stably expressed in N. benthamiana [268,269]. Soluble gp140 was produced and successfully co-expressed with a human chaperone protein to achieve higher yields of the viral protein [270]. gp40, produced via transient expression in N. benthamiana, formed a trimeric structure and elicited a robust immunological response in rabbits [271]. Despite the substantial number of studies on this topic, none of the HIV plant-based vaccine candidates have reached the clinical trial stages.
Table 3.
Examples of plant-produced HIV proteins that have shown immunogenic effects in animals.
| Antigen | Production System and Expression Level |
Immunogenicity | References |
|---|---|---|---|
| 22 aa epitope from gp41 | Cowpea; CPMV-HIV chimera; n/a. | Mice; Stimulated a strong serum neutralizing antibody response in three strains of mice. | [267,272,273] |
| CTB-P1 | N. benthamiana (transient); >80% of the CTB and CTB-P1 were assembled into functional oligomers. | Mice; Serum IgG response; mucosal IGA response; induction of immunological memory. | [274] |
| p24 | N. benthamiana (transient); 100 μg/g FW. | Rabbit; Induced specific humoral immune response. | [266] |
| NV and GAG | Tomato (transgenic); VLPs composed of the major antigenic protein for the hepatitis B virus (HBV); 0.3 ng/mg powder. | Mice; Induced a humoral immune response. | [275] |
| polHIV-1.op | Nicotiana tabacum (transgenic); VLPs composed of the major antigenic protein for the hepatitis B virus (HBV); 2–26 ng/g FW. | Mice; Elicited anti-HIV-1 specific CD8+ T cell activation detectable in mesenteric lymph nodes. | [268,269] |
| Tat | Tomato (transgenic); 1 μg/mg fruit (dry weight). | Mice; A strong anti-Tat immunological response after either intraperitoneal, intramuscular, or oral application. | [276] |
| p17/p24 | Nicotiana tabacum (transient); >1 mg p24/kg of FW. | Mice; Induced humoral and T cell immune response. | [277] |
| CTB-MPR | N. benthamiana (transgenic); 1–2 mg/kg FW. | Mice; Induction of serum and mucosal antibodies. | [278] |
| p24 | Nicotiana tabacum L. (transplastomic); n/a. | Mice; Serum IgG response. | [265] |
| C4(V3)6 | Lettuce (transgenic); 240 μg/g freeze-dried leaves. | Mice; Induction of humoral and cell-mediated immune response. | [263] |
| p24 | Arabidopsis thaliana (transgenic); 0.2 μg–0.5 μg/g FW. | Mice; Serum IgG response. | [264,279] |
| C4V3 (Multi-HIV) | Tobacco (transplastomic); 16 μg/g FW. | Mice; Induction of systemic mucosal, humoral, and T cell immune response. | [263,280,281] |
| Poly HIV | Physcomitrella patens (transgenic); 3.7 µg/g−1 FW. | Mice; Induction of specific antibody response. | [282] |
| Dgp41 and Gag | N. benthamiana (transient and transgenic respectively); ~9 mg/kg FW and ~22 mg/kg FW, respectively. | Mice; Serum antibodies against both the Gag and gp41 antigens were produced. CD4 and CD8 T cell response. | [283,284] |
| gp140 | N. benthamiana (transient); 21.5 mg/kg FW. | Rabbits; High titers of binding antibodies, including against the V1V2 loop region, and neutralizing antibodies against Tier 1 viruses. | [270,271] |
11. Rabdoviruses
Rabies Virus (RABV)
Rabies virus is a neurotropic agent that causes acute infection of the central nervous system (rabies) in mammals. This can be transmitted to humans by bites of infected animals. The host range predominantly includes carnivores and bats. In humans, mortality is nearly 100% once clinical symptoms of rabies appear [285,286]. Mortality is much lower for the animals that carry the rabies virus; 14% of dogs survive, and bats can survive too [287]. The viral RNA genome encodes five viral proteins: the nucleoprotein (N), phosphoprotein (P), matrix protein (M), glycoprotein (G), and a large RNA polymerase protein (L) [288]. The surface-exposed G protein is responsible for the induction of virus-neutralizing antibodies. There are numerous examples of candidate vaccines based on the G protein or nucleoprotein produced in plants. Many review articles discuss the progress in the development of plant-based vaccines against RABV [13,116,289,290]. Transgenic tobacco plants have been modeled to produce rabies G protein at 0.38% of the total soluble leaf protein, which, upon purification and immunization, provided complete protection in mice after a lethal challenge [291]. Since tobacco cannot be used as an edible vaccine, efforts have been made to use edible plants as potential alternatives for a Rhabdovirus vaccine. Tomato [292,293,294], carrot [295], spinach [296,297], and corn [298] have been used to produce edible vaccines against rabies. Serological studies have shown that oral administration of plant-produced viral proteins caused antigen-stimulated IgG and IgA synthesis and improved the overall health of mice intranasally infected with an attenuated rabies virus [296]. Parenteral immunization of mice with purified N protein produced in transgenic tomato achieved partial protection, while oral immunization with the same product failed [293]. Orally administrated G protein (50 µg in transgenic corn) induced viral neutralizing antibodies and protected 100% of the treated mice against a challenge [298,299]. Loza-Rubio et al. demonstrated that the degree of protection achieved after 2 mg of G protein administrated orally was comparable to that conferred by a commercial vaccine [299]. This study demonstrates that cereals are an attractive platform for oral vaccine production due to the relatively high recombinant protein accumulation and the possibility for long-term storage under ambient conditions.
Oral vaccination with the plant-produced chimeric peptides, containing antigenic determinants from glycoprotein G and nucleoprotein N fused with the alfalfa mosaic virus coat protein (AlMV CP), improved weight gain in mice following a challenge with an attenuated virus strain [296]. Yusibov et al. described the expression of a similar chimeric protein (epitopes from G and N fused with the AlMV coat protein) in tobacco and spinach plants. Studies in human volunteers showed a specific response against the rabies antigens after ingesting raw spinach leaves infected with the recombinant virus [297].
12. Hepatitis E Virus
Hepatitis E virus (HEV) HEV is a positive-sense, single-stranded quasi-enveloped RNA icosahedral virus from the Hepeviridae [300]. Genus Orthohepevirus unite all virus isolates from mammals and birds organized into eight genotypes. Genotypes 1 and 2 are specific for humans, while 3–8 are spread mainly in animals. The main animal reservoir of HEV is the domestic swine, but other animals such as wild boar, dear, camel, etc. can be reservoirs as well [301,302,303,304,305].
HEV ORF2 encodes the viral capsid protein, which is a major immunogenic factor, and for that reason, it is of great interest for anti-HEV vaccine development [306]. Earlier PMF studies were based either on nuclear or plastid stable transformations [307,308,309]. The E2 fragment (394–607 aa) from ORF2 was cloned into a plasmid and successfully transformed into tomato plants using Agrobacterium tumefaciens. Although normal immunoactivity of the recombinant protein was detected, the stable nuclear transformation resulted in low accumulation of the desired product—61.22 ng/g fresh weight (FW) in fruits and 6.37–47.9 ng/g FW in leaves—which makes the methodology unsuitable for scalable production [307]. In another study, the E2 fragment was cloned into a plastid-targeting vector and delivered in tobacco via biolistic particle bombardment. Application of the plastid transformation approach resulted in an increased yield of 13.27 μg/g FW.
The generated recombinant protein was able to elicit a positive antibody response in mice [309]. In the pursuit of an oral vaccine, transgenic potatoes were developed by Agrobacterium-mediated delivery of two truncated ORF2 fragments, truncated 111N and 111N/54C. A yield up to 30 μg/g FW was observed with very few VLPs. The absence of an immune response in mice highlighted the importance of VLP formation for the induction of a proper immune response [308].
The transient expression approach was used in conjunction with the highly productive vectors pEAQ-HT and pEff, based on the Cowpea mosaic virus (CPMV) and the potato virus X (PVX), respectively. When a truncated ORF2 (110–610 aa) sequence, cloned into the pEAQ-HT vector, was expressed, it resulted in a protein production rate of 100 µg/g of FW [310], whereas the same sequence cloned into the pEff vector yielded up to 200 µg/g of FW [311,312]. Both products confirmed that the truncated HEV capsid proteins expressed in plants are suitable for diagnostic purposes [310,312]. VLP formation was also examined by atomic force and electron microscopy. The collected data confirmed self-assembled VLP particles with very heterogeneous sizes [95,312]. The immunogenicity of the plant-produced HEV 110–610 protein was examined in mice, and it was concluded that immunizations with the recombinant protein induced high titers of specific IgG antibodies in comparison to the negative control group [311]. Additionally, HBcAg was used to present an HEV immunogenic epitope. The amino acids 551–607 of the HEV ORF2 capsid protein were successfully inserted into HBV nucleocapsid gene and upon expression in plants, chimeric “ragged” VLPs were produced, which reacted with HEV antibodies [56].
13. Newcastle Disease Virus (NDV)
The NDV belongs to the Paramyxoviridae family and its single-stranded, negative sense RNA genome encodes six structural proteins: nucleocapsid (N), phosphoprotein (P), matrix protein (M), fusion protein (F), haemagglutinin-neuraminidase protein (HN), and large polymerase protein (L) [313]. The surface glycoproteins F and HN are the major immunogenic proteins [314,315] and targets for vaccine design. NDV is highly infectious and often fatal in birds, including domestic poultry [316]. Humans can be infected by NDV through direct contact with infected poultry, which is usually presented as conjunctivitis. No human cases of Newcastle disease have occurred as a result of consuming poultry products [317]. ND severely impacts the poultry industry worldwide because it causes significant morbidity and mortality [318]. To control spreading, prophylactic and emergency vaccination against ND is applied on a large scale in many countries. Various plant systems have been used for NDV vaccine production, focused primarily on the expression of the F and HN glycoproteins (Table 4).
Table 4.
Plant-based candidate vaccines against Newcastle disease virus.
| Antigen | Production System and Expression Level |
Immunogenicity | References |
|---|---|---|---|
| Fusion (F) and haemagglutinin-neuraminidase (HN) protein | Potato/stable transformation/0.3–0.6 µg/mg of total leaf protein | Oral and intraperitoneal delivery of the antigens elicited mucosal and systemic immune response | [319] |
| Fusion (F) | Rice/stable transformation/0.25–0.55 μg of purified NDV in 100 μg of total soluble leaf or seed proteins | Intraperitoneally immunized mice with crude protein extracts from transgenic rice plants elicited specific antibodies | [320] |
| Fusion (F) | Maize/stable transformation/ 0.9–3% TSP |
Orally immunized chicken developed protective immune response | [321] |
| Fusion (F) and hemagglutinin-neuraminidase (HN) proteins | 0.5–0.8% of total seed protein Mize/stable transformation | Induced specific immune response after oral administration to chicken |
[322] |
| Fusion (F) and hemagglutinin-neuraminidase (HN) proteins | Transgenic Canola Seeds | Chickens immunized orally with recombinant HN-F showed a significant rise in specific and hemagglutination inhibition (HI) antibodies | [323] |
| F and HN epitopes fused to Cucumber mosaic virus (CMV) |
N. benthamiana/CMV formed VLPs, which served as carriers for display of neutralizing epitopes of F and HN/20–200 g of infected leaves | No data | [324,325] |
| Hemagglutinin-neuraminidase (HN) | N. benthamiana/transient expression | No data | [326] |
| Hemagglutinin-neuraminidase protein | Transgenic N. tobaccum
0.069% of TSP |
Orally immunized chickens developed low titers of anti-HN serum IgG | [327] |
| Ectodomain of hemagglutinin-neuraminidase protein |
Tobacco cell culture 0.2–0.4% of TSP |
Mice receiving purified eHN protein from transgenic tobacco BY-2 cells produced specific anti-NDV antibodies | [19] |
Table 4 shows that the main research efforts are aimed at creating an oral vaccine against NDV that can be easily administered through food and can be stored long-term under ambient conditions. Immunological studies with seed-based orally administrated vaccines induced a robust immune response. Guerrero-Andrade et al. showed that 100% of the chicken fed with transgenic maize expressing the F glycoprotein survived after a lethal challenge with NDV [321]. These studies show the feasibility of edible vaccines for veterinary application.
A few years back, Dow AgroSciences made a breakthrough in plant-based vaccines. In 2016, they received the first FDA approval for the plant-derived injection application vaccine against NDV, based on HN protein produced in an N. benthamiana cell culture [328].
14. Henipaviruses
Hendra virus (HeV) and Nipah virus (NiV) have recently emerged as zoonotic pathogens affecting domestic animals (horses, pigs) with a documented spillover in humans, sometimes causing lethal disease [318,320]. Based on their genomic organization, they are currently classified in the family Paramyxoviridae [329]. Fruit bats of the Pteropus genus were confirmed as the main zoonotic hosts of Henipaviruses [330]. HeV was initially identified in an outbreak in horses in Australia. NiV was subsequently discovered in an outbreak in pigs in Malaysia [329]. Human cases have been observed after close contact with infected animals (horses and pigs). Food-borne transmission was also reported in people who consumed fruit or palm sap from containers contaminated by fruit bats [331].
HeV and NiV are single-stranded RNA, enveloped viruses. The Henipavirus genome encodes six structural proteins: nucleoprotein (N), phosphoprotein (P), matrix protein (M), fusion glycoprotein (F), attachment glycoprotein (G), and the polymerase protein (L) [332]. The F and G glycoproteins are the main immunogenic proteins and target for vaccine development. Progress in the development of vaccines against the Nipah and Hendra viruses was reported in a review article [333].
Currently, there is no published evidence suggesting the development of a vaccine against Henipaviruses in plants, which presents PMF scientists with an opportunity to make rapid progress in the development of vaccines against HeV and NiV in plants, following the achievements of the plant-based vaccine against the Newcastle disease.
15. Concluding Remarks
The 21st Century has seen remarkable progress in the development of plant-based vaccines against viral diseases. Although the vast majority of these are centered on aca-demic development and are still at the level of pre-clinical trials in animal models, we are also witnessing the significant achievements of vendors such as Medicago Inc and Kentucky Bioprocessing Inc, whose vaccine candidates against influenza and SARS-CoV-2 are likely to be licensed next year. Several factors are contributing to the observed progress in plant-based vaccine development. From a technical perspective, there is a shift from stable expression of viral proteins in transgenic plants to transient expression in N. benthamiana. The latter is much faster and can produce a decent yield of viral antigens within a week after infiltrating plant leaf material with an Agrobacterium suspension containing the target gene, offering the opportunity for a rapid response to the annual emergence of antigenically novel influenza strains or unexpected pandemics. Another very important factor for the current advancement of plant-based vaccines is the recent achievements in plant platforms that not only express target antigens but also facilitate the assembly of VLPs. The advantage of the plant-derived VLPs over simple expression of recombinant proteins in plants is that the former captures the antigenic epitopes in their native conformation, which results in enhanced immunogenicity and ultimately superior efficiency as a potential vaccine. Apart from the above-mentioned technological factors, the potential success of a Medicago plant-derived SARS-CoV-2 vaccine will accelerate the development of other plant vaccines in general.
Fischer and Buyel 2020 [14] describe four factors that affect the selection of plant expression systems: time-to-market factors; the amount of time needed for research and development (R&D); scalability; and regulatory approval. Of the four, the time-to-market factors are the biggest determinant in the selection process. In addition, while plants offer an advantage during R&D (faster transient expression), the production upscaling of plant-produced proteins is usually a challenge. However, the approval of the new plant-based vaccines is likely to open new avenues for improved manufacturing.
The majority of the zoonotic viruses listed in this review disproportionally affect developing countries. The development of new vaccines comes with a cost, which is a challenge for resource-limited countries that need these vaccines most. Plant-based vaccines carry the promise not only to be efficient in preventing zoonotic disease but also, importantly, to be cost-efficient and affordable.
Sustainable long-term cooperation and partnerships with global organizations (the WHO, the World Bank, and others) and governments must be established if plant molecular farming is to become a successful mainstream platform for vaccine generation and production.
Acknowledgments
The authors would like to thank Valeria Tonova (bachelor student, University of Plovdiv) for helping with the graphic abstract preparation and the program “Young scientists and Postdoctoral candidates” from the Bulgarian Ministry of Education and Science for funding Katerina Takova as a young scientist.
Author Contributions
Conceptualization, G.Z.; formal analysis, all authors; investigation, all authors; data curation, K.T.; writing—original draft preparation, all authors; writing—review and editing, G.L.L., A.A. and G.Z.; supervision, G.Z. and A.A.; project administration, V.T. and I.M.; funding acquisition, G.Z. and I.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the European Union’s Horizon 2020 research and innovation programme, project PlantaSYST (SGA-CSA No. 739582 under FPA No. 664620) and by the European Regional Development Fund through the Bulgarian “Science and Education for Smart Growth” Operational Programme (project BG05M2OP001-1.003-001-C01), Research Fund at the University of Plovdiv, competition “Young Scientists and Doctoral Students” MU21-BF-022.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Reed K.D. Viral Zoonoses. Ref. Module Biomed. Sci. 2018 doi: 10.1016/B978-0-12-801238-3.95729-5. [DOI] [Google Scholar]
- 2.Martins S.B., Häsler B., Rushton J. Economic Aspects of Zoonoses: Impact of Zoonoses on the Food Industry. In: Sing A., editor. Zoonoses—Infections Affecting Humans and Animals: Focus on Public Health Aspects. Springer; Dordrecht, The Netherlands: 2015. pp. 1107–1126. [Google Scholar]
- 3.Rahman M.T., Sobur M.A., Islam M.S., Ievy S., Hossain M.J., El Zowalaty M.E., Rahman A.T., Ashour H.M. Zoonotic Diseases: Etiology, Impact, and Control. Microorganisms. 2020;8:1405. doi: 10.3390/microorganisms8091405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Olival K.J., Hosseini P.R., Zambrana-Torrelio C., Ross N., Bogich T.L., Daszak P. Host and Viral Traits Predict Zoonotic Spillover from Mammals. Nature. 2017;546:646–650. doi: 10.1038/nature22975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Carroll D., Watson B., Togami E., Daszak P., Mazet J.A., Chrisman C.J., Rubin E.M., Wolfe N., Morel C.M., Gao G.F., et al. Building a Global Atlas of Zoonotic Viruses. Bull. World Health Organ. 2018;96:292–294. doi: 10.2471/BLT.17.205005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Belay E.D., Kile J.C., Hall A.J., Barton-Behravesh C., Parsons M.B., Salyer S.J., Walke H. Zoonotic Disease Programs for Enhancing Global Health Security. Emerg. Infect. Dis. J. 2017;23:S65. doi: 10.3201/eid2313.170544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rantsios A.T. Zoonoses. Encycl. Food Health. 2016:645–653. doi: 10.1016/B978-0-12-384947-2.00770-4. [DOI] [Google Scholar]
- 8.Institute of Medicine (US) Forum on Microbial Threats . Vector-Borne Diseases: Understanding the Environmental, Human Health, and Ecological Connections, Workshop Summary. National Academies Press; Washington, DC, USA: 2008. [PubMed] [Google Scholar]
- 9.Gebreyes W.A., Dupouy-Camet J., Newport M.J., Oliveira C.J.B., Schlesinger L.S., Saif Y.M., Kariuki S., Saif L.J., Saville W., Wittum T., et al. The Global One Health Paradigm: Challenges and Opportunities for Tackling Infectious Diseases at the Human, Animal, and Environment Interface in Low-Resource Settings. PLoS Negl. Trop. Dis. 2014;8:e3257. doi: 10.1371/journal.pntd.0003257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Schillberg S., Finnern R. Plant Molecular Farming for the Production of Valuable Proteins—Critical Evaluation of Achievements and Future Challenges. J. Plant Physiol. 2021;258–259:153359. doi: 10.1016/j.jplph.2020.153359. [DOI] [PubMed] [Google Scholar]
- 11.Rybicki E.P. Plant Molecular Farming of Virus-like Nanoparticles as Vaccines and Reagents. WIREs Nanomed. Nanobiotechnol. 2020;12:e1587. doi: 10.1002/wnan.1587. [DOI] [PubMed] [Google Scholar]
- 12.Shanmugaraj B., Bulaon C.J.I., Phoolcharoen W. Plant Molecular Farming: A Viable Platform for Recombinant Biopharmaceutical Production. Plants. 2020;9:842. doi: 10.3390/plants9070842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rybicki E.P. Plant-Based Vaccines against Viruses. Virol. J. 2014;11:205. doi: 10.1186/s12985-014-0205-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Fischer R., Buyel J.F. Molecular Farming—The Slope of Enlightenment. Biotechnol. Adv. 2020;40:107519. doi: 10.1016/j.biotechadv.2020.107519. [DOI] [PubMed] [Google Scholar]
- 15.Gómez M.L., Huang X., Alvarez D., He W., Baysal C., Zhu C., Armario-Najera V., Perera A.B., Bennasser P.C., Saba-Mayoral A., et al. Contributions of the International Plant Science Community to the Fight against Human Infectious Diseases–Part 1: Epidemic and Pandemic Diseases. Plant Biotechnol. J. 2021;19:1901–1920. doi: 10.1111/pbi.13657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ma J.K.-C., Drake P.M.W., Christou P. The Production of Recombinant Pharmaceutical Proteins in Plants. Nat. Rev. Genet. 2003;4:794–805. doi: 10.1038/nrg1177. [DOI] [PubMed] [Google Scholar]
- 17.Ward B.J., Makarkov A., Séguin A., Pillet S., Trépanier S., Dhaliwall J., Libman M.D., Vesikari T., Landry N. Efficacy, Immunogenicity, and Safety of a Plant-Derived, Quadrivalent, Virus-like Particle Influenza Vaccine in Adults (18–64 Years) and Older Adults (≥65 Years): Two Multicentre, Randomised Phase 3 Trials. Lancet. 2020;396:1491–1503. doi: 10.1016/S0140-6736(20)32014-6. [DOI] [PubMed] [Google Scholar]
- 18.Vermij P., Waltz E. USDA Approves the First Plant-Based Vaccine. Nat. Biotechnol. 2006;24:234. [Google Scholar]
- 19.Lai K.S., Yusoff K., Mahmood M. Functional Ectodomain of the Hemagglutinin-Neuraminidase Protein Is Expressed in Transgenic Tobacco Cells as a Candidate Vaccine against Newcastle Disease Virus. Plant Cell Tissue Organ Cult. 2013;1:117–121. doi: 10.1007/s11240-012-0214-x. [DOI] [Google Scholar]
- 20.Elelyso® for Gaucher Disease. [(accessed on 19 November 2021)]. Available online: https://protalix.com/about/elelyso/
- 21.Peyret H., Brown J.K.M., Lomonossoff G.P. Improving Plant Transient Expression through the Rational Design of Synthetic 5′ and 3′ Untranslated Regions. Plant Methods. 2019;15:108. doi: 10.1186/s13007-019-0494-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Montero-Morales L., Steinkellner H. Advanced Plant-Based Glycan Engineering. Front. Bioeng. Biotechnol. 2018;6:81. doi: 10.3389/fbioe.2018.00081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Drossard J. Molecular Farming. John Wiley & Sons; Hoboken, NJ, USA: 2004. Downstream Processing of Plant-Derived Recombinant Therapeutic Proteins; pp. 217–231. [Google Scholar]
- 24.Streatfield S.J. Approaches to Achieve High-Level Heterologous Protein Production in Plants. Plant Biotechnol. J. 2007;5:2–15. doi: 10.1111/j.1467-7652.2006.00216.x. [DOI] [PubMed] [Google Scholar]
- 25.Jutras P.V., D’Aoust M.-A., Couture M.M.-J., Vézina L.-P., Goulet M.-C., Michaud D., Sainsbury F. Modulating Secretory Pathway PH by Proton Channel Co-Expression Can Increase Recombinant Protein Stability in Plants. Biotechnol. J. 2015;10:1478–1486. doi: 10.1002/biot.201500056. [DOI] [PubMed] [Google Scholar]
- 26.Jutras P.V., Dodds I., van der Hoorn R.A. Proteases of Nicotiana Benthamiana: An Emerging Battle for Molecular Farming. Curr. Opin. Biotechnol. 2020;61:60–65. doi: 10.1016/j.copbio.2019.10.006. [DOI] [PubMed] [Google Scholar]
- 27.Chung Y.H., Church D., Koellhoffer E.C., Osota E., Shukla S., Rybicki E.P., Pokorski J.K., Steinmetz N.F. Integrating Plant Molecular Farming and Materials Research for Next-Generation Vaccines. Nat. Rev. Mater. 2021:1–17. doi: 10.1038/s41578-021-00399-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.LeBlanc Z., Waterhouse P., Bally J. Plant-Based Vaccines: The Way Ahead? Viruses. 2021;13:5. doi: 10.3390/v13010005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fischer R., Schillberg S. Molecular Farming: Plant-Made Pharmaceuticals and Technical Proteins. Wiley-VCH; Weinheim, Germany: 2004. [Google Scholar]
- 30.Arntzen C. Plant-Made Pharmaceuticals: From “Edible Vaccines” to Ebola Therapeutics. Plant Biotechnol. J. 2015;13:1013–1016. doi: 10.1111/pbi.12460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Tacket C.O., Mason H.S., Losonsky G., Estes M.K., Levine M.M., Arntzen C.J. Human Immune Responses to a Novel Norwalk Virus Vaccine Delivered in Transgenic Potatoes. J. Infect. Dis. 2000;182:302–305. doi: 10.1086/315653. [DOI] [PubMed] [Google Scholar]
- 32.Tacket C.O., Mason H.S., Losonsky G., Clements J.D., Levine M.M., Arntzen C.J. Immunogenicity in Humans of a Recombinant Bacterial Antigen Delivered in a Transgenic Potato. Nat. Med. 1998;4:607–609. doi: 10.1038/nm0598-607. [DOI] [PubMed] [Google Scholar]
- 33.Thanavala Y., Mahoney M., Pal S., Scott A., Richter L., Natarajan N., Goodwin P., Arntzen C.J., Mason H.S. Immunogenicity in Humans of an Edible Vaccine for Hepatitis B. Proc. Natl. Acad. Sci. USA. 2005;102:3378–3382. doi: 10.1073/pnas.0409899102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kolotilin I., Topp E., Cox E., Devriendt B., Conrad U., Joensuu J., Stöger E., Warzecha H., McAllister T., Potter A., et al. Plant-Based Solutions for Veterinary Immunotherapeutics and Prophylactics. Vet. Res. 2014;45:117. doi: 10.1186/s13567-014-0117-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Liew P.S., Hair-Bejo M. Farming of Plant-Based Veterinary Vaccines and Their Applications for Disease Prevention in Animals. Adv. Virol. 2015;2015:936940. doi: 10.1155/2015/936940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rybicki E. History and Promise of Plant-Made Vaccines for Animals. In: MacDonald J., editor. Prospects of Plant-Based Vaccines in Veterinary Medicine. Springer; Cham, Switzerland: 2018. pp. 1–22. [Google Scholar]
- 37.Keshavareddy G., Kumar A.R.V., Ramu V.S. Methods of Plant Transformation—A Review. Int. J. Curr. Microbiol. Appl. Sci. 2018;7:2656–2668. doi: 10.20546/ijcmas.2018.707.312. [DOI] [Google Scholar]
- 38.Barampuram S., Zhang Z.J. Recent Advances in Plant Transformation. Methods Mol. Biol. Clifton. 2011;701:1–35. doi: 10.1007/978-1-61737-957-4_1. [DOI] [PubMed] [Google Scholar]
- 39.Krenek P., Samajova O., Luptovciak I., Doskocilova A., Komis G., Samaj J. Transient Plant Transformation Mediated by Agrobacterium Tumefaciens: Principles, Methods and Applications. Biotechnol. Adv. 2015;33:1024–1042. doi: 10.1016/j.biotechadv.2015.03.012. [DOI] [PubMed] [Google Scholar]
- 40.Mardanova E.S., Blokhina E.A., Tsybalova L.M., Peyret H., Lomonossoff G.P., Ravin N.V. Efficient Transient Expression of Recombinant Proteins in Plants by the Novel PEff Vector Based on the Genome of Potato Virus X. Front. Plant Sci. 2017;8:247. doi: 10.3389/fpls.2017.00247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Naseri Z., Khezri G., Davarpanah S.J., Ofoghi H. Virus-Based Vectors: A New Approach for Production of Recombinant Proteins. J. Appl. Biotechnol. Rep. 2019;6:6–14. doi: 10.29252/JABR.06.01.02. [DOI] [Google Scholar]
- 42.Gleba Y., Klimyuk V., Marillonnet S. Viral Vectors for the Expression of Proteins in Plants. Curr. Opin. Biotechnol. 2007;18:134–141. doi: 10.1016/j.copbio.2007.03.002. [DOI] [PubMed] [Google Scholar]
- 43.Sainsbury F., Thuenemann E.C., Lomonossoff G.P. PEAQ: Versatile Expression Vectors for Easy and Quick Transient Expression of Heterologous Proteins in Plants. Plant Biotechnol. J. 2009;7:682–693. doi: 10.1111/j.1467-7652.2009.00434.x. [DOI] [PubMed] [Google Scholar]
- 44.Venkataraman S., Hefferon K. Application of Plant Viruses in Biotechnology, Medicine, and Human Health. Viruses. 2021;13:1697. doi: 10.3390/v13091697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Peyret H., Lomonossoff G.P. When Plant Virology Met Agrobacterium: The Rise of the Deconstructed Clones. Plant Biotechnol. J. 2015;13:1121–1135. doi: 10.1111/pbi.12412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sainsbury F. Innovation in Plant-Based Transient Protein Expression for Infectious Disease Prevention and Preparedness. Curr. Opin. Biotechnol. 2020;61:110–115. doi: 10.1016/j.copbio.2019.11.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Bally J., Jung H., Mortimer C., Naim F., Philips J.G., Hellens R., Bombarely A., Goodin M.M., Waterhouse P.M. The Rise and Rise of Nicotiana Benthamiana: A Plant for All Reasons. Annu. Rev. Phytopathol. 2018;56:405–426. doi: 10.1146/annurev-phyto-080417-050141. [DOI] [PubMed] [Google Scholar]
- 48.Shahid N., Daniell H. Plant-Based Oral Vaccines against Zoonotic and Non-Zoonotic Diseases. Plant Biotechnol. J. 2016;14:2079–2099. doi: 10.1111/pbi.12604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Twyman R.M., Christou P. Handbook of Plant Biotechnology. John Wiley & Sons; Hoboken, NJ, USA: 2004. Plant Transformation Technology: Particle Bombardment. [Google Scholar]
- 50.Xu J., Zhang N. On the Way to Commercializing Plant Cell Culture Platform for Biopharmaceuticals: Present Status and Prospect. Pharm. Bioprocess. 2014;2:499–518. doi: 10.4155/pbp.14.32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Marsian J., Lomonossoff G.P. Molecular Pharming—VLPs Made in Plants. Curr. Opin. Biotechnol. 2016;37:201–206. doi: 10.1016/j.copbio.2015.12.007. [DOI] [PubMed] [Google Scholar]
- 52.Zeltins A. Protein Complexes and Virus-Like Particle Technology. Subcell. Biochem. 2018;88:379–405. doi: 10.1007/978-981-10-8456-0_16. [DOI] [PubMed] [Google Scholar]
- 53.Wang C., Zheng X., Gai W., Wong G., Wang H., Jin H., Feng N., Zhao Y., Zhang W., Li N., et al. Novel Chimeric Virus-like Particles Vaccine Displaying MERS-CoV Receptor-Binding Domain Induce Specific Humoral and Cellular Immune Response in Mice. Antivir. Res. 2017;140:55–61. doi: 10.1016/j.antiviral.2016.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Chen Q., Lai H. Plant-Derived Virus-like Particles as Vaccines. Hum. Vaccines Immunother. 2013;9:26–49. doi: 10.4161/hv.22218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Rutkowska D.A., Mokoena N.B., Tsekoa T.L., Dibakwane V.S., O’Kennedy M.M. Plant-Produced Chimeric Virus-like Particles—A New Generation Vaccine against African Horse Sickness. BMC Vet. Res. 2019;15:432. doi: 10.1186/s12917-019-2184-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Zahmanova G., Mazalovska M., Takova K., Toneva V., Minkov I., Peyret H., Lomonossoff G. Efficient Production of Chimeric Hepatitis B Virus-Like Particles Bearing an Epitope of Hepatitis E Virus Capsid by Transient Expression in Nicotiana Benthamiana. Life. 2021;11:64. doi: 10.3390/life11010064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Nooraei S., Bahrulolum H., Hoseini Z.S., Katalani C., Hajizade A., Easton A.J., Ahmadian G. Virus-like Particles: Preparation, Immunogenicity and Their Roles as Nanovaccines and Drug Nanocarriers. J. Nanobiotechnol. 2021;19:59. doi: 10.1186/s12951-021-00806-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Lomonossoff G.P. Recent Advances in Plant Virology. Caister Academic Press; Poole, UK: 2011. Virus Particles and the Uses of Such Particles in Bio- and Nanotechnology. [Google Scholar]
- 59.Mardanova E.S., Kotlyarov R.Y., Kuprianov V.V., Stepanova L.A., Tsybalova L.M., Lomonosoff G.P., Ravin N.V. Rapid High-Yield Expression of a Candidate Influenza Vaccine Based on the Ectodomain of M2 Protein Linked to Flagellin in Plants Using Viral Vectors. BMC Biotechnol. 2015;15:42. doi: 10.1186/s12896-015-0164-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Matić S., Rinaldi R., Masenga V., Noris E. Efficient Production of Chimeric Human Papillomavirus 16 L1 Protein Bearing the M2e Influenza Epitope in Nicotiana Benthamiana Plants. BMC Biotechnol. 2011;11:106. doi: 10.1186/1472-6750-11-106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Medicago Inc Plant-Derived Vaccines. Biopharma Dealmakers (Biopharm Deal). 2018. [(accessed on 19 November 2021)]. Available online: https://www.nature.com/articles/d43747-020-00537-y.
- 62.Pillet S., Aubin É., Trépanier S., Bussière D., Dargis M., Poulin J.-F., Yassine-Diab B., Ward B.J., Landry N. A Plant-Derived Quadrivalent Virus like Particle Influenza Vaccine Induces Cross-Reactive Antibody and T Cell Response in Healthy Adults. Clin. Immunol. 2016;168:72–87. doi: 10.1016/j.clim.2016.03.008. [DOI] [PubMed] [Google Scholar]
- 63.Balke I., Zeltins A. Recent Advances in the Use of Plant Virus-Like Particles as Vaccines. Viruses. 2020;12:270. doi: 10.3390/v12030270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Lefkowitz E.J., Dempsey D.M., Hendrickson R.C., Orton R.J., Siddell S.G., Smith D.B. Virus Taxonomy: The Database of the International Committee on Taxonomy of Viruses (ICTV) Nucleic Acids Res. 2018;46:D708–D717. doi: 10.1093/nar/gkx932. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Hay A.J., Gregory V., Douglas A.R., Lin Y.P. The Evolution of Human Influenza Viruses. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2001;356:1861–1870. doi: 10.1098/rstb.2001.0999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cox N.J., Subbarao K. Global Epidemiology of Influenza: Past and Present. Annu. Rev. Med. 2000;51:407–421. doi: 10.1146/annurev.med.51.1.407. [DOI] [PubMed] [Google Scholar]
- 67.Krammer F. The Human Antibody Response to Influenza A Virus Infection and Vaccination. Nat. Rev. Immunol. 2019;19:383–397. doi: 10.1038/s41577-019-0143-6. [DOI] [PubMed] [Google Scholar]
- 68.Shao W., Li X., Goraya M.U., Wang S., Chen J.-L. Evolution of Influenza A Virus by Mutation and Re-Assortment. Int. J. Mol. Sci. 2017;18:1650. doi: 10.3390/ijms18081650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Proença-Módena J.L., Macedo I.S., Arruda E. H5N1 Avian Influenza Virus: An Overview. Braz. J. Infect. Dis. 2007;11:125–133. doi: 10.1590/S1413-86702007000100027. [DOI] [PubMed] [Google Scholar]
- 70.Paget J., Spreeuwenberg P., Charu V., Taylor R.J., Iuliano A.D., Bresee J., Simonsen L., Viboud C. Global Seasonal Influenza-associated Mortality Collaborator Network and GLaMOR Collaborating Teams* Global Mortality Associated with Seasonal Influenza Epidemics: New Burden Estimates and Predictors from the GLaMOR Project. J. Glob. Health. 2019;9:020421. doi: 10.7189/jogh.09.020421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Pellerin C. DARPA Effort Speeds Bio-Threat Response. [(accessed on 19 November 2021)]. Available online: https://www.army.mil/article/47617/darpa_effort_speeds_bio_threat_response.
- 72.Medicago Inc Final Short Form Prospectus. [(accessed on 19 November 2021)]. Available online: https://sec.report/otc/financial-report/93315.
- 73.D’Aoust M.-A., Lavoie P.-O., Couture M.M.-J., Trépanier S., Guay J.-M., Dargis M., Mongrand S., Landry N., Ward B.J., Vézina L.-P. Influenza Virus-like Particles Produced by Transient Expression in Nicotiana Benthamiana Induce a Protective Immune Response against a Lethal Viral Challenge in Mice. Plant Biotechnol. J. 2008;6:930–940. doi: 10.1111/j.1467-7652.2008.00384.x. [DOI] [PubMed] [Google Scholar]
- 74.Landry N., Ward B.J., Trépanier S., Montomoli E., Dargis M., Lapini G., Vézina L.-P. Preclinical and Clinical Development of Plant-Made Virus-Like Particle Vaccine against Avian H5N1 Influenza. PLoS ONE. 2010;5:e15559. doi: 10.1371/journal.pone.0015559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Pillet S., Aubin É., Trépanier S., Poulin J.-F., Yassine-Diab B., ter Meulen J., Ward B.J., Landry N. Humoral and Cell-Mediated Immune Responses to H5N1 Plant-Made Virus-like Particle Vaccine Are Differentially Impacted by Alum and GLA-SE Adjuvants in a Phase 2 Clinical Trial. npj Vaccines. 2018;3:3. doi: 10.1038/s41541-017-0043-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pillet S., Couillard J., Trépanier S., Poulin J.-F., Yassine-Diab B., Guy B., Ward B.J., Landry N. Immunogenicity and Safety of a Quadrivalent Plant-Derived Virus like Particle Influenza Vaccine Candidate—Two Randomized Phase II Clinical Trials in 18 to 49 and ≥50 Years Old Adults. PLoS ONE. 2019;14:e0216533. doi: 10.1371/journal.pone.0216533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Tripathy S., Dassarma B., Bhattacharya M., Matsabisa M.G. Plant-Based Vaccine Research Development against Viral Diseases with Emphasis on Ebola Virus Disease: A Review Study. Curr. Opin. Pharmacol. 2021;60:261–267. doi: 10.1016/j.coph.2021.08.001. [DOI] [PubMed] [Google Scholar]
- 78.Home—ClinicalTrials.Gov. [(accessed on 19 November 2021)]; Available online: https://www.clinicaltrials.gov/
- 79.Smith T., O’Kennedy M.M., Wandrag D.B.R., Adeyemi M., Abolnik C. Efficacy of a Plant-Produced Virus-like Particle Vaccine in Chickens Challenged with Influenza A H6N2 Virus. Plant Biotechnol. J. 2020;18:502–512. doi: 10.1111/pbi.13219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Shoji Y., Bi H., Musiychuk K., Rhee A., Horsey A., Roy G., Green B., Shamloul M., Farrance C.E., Taggart B., et al. Plant-Derived Hemagglutinin Protects Ferrets against Challenge Infection with the A/Indonesia/05/05 Strain of Avian Influenza. Vaccine. 2009;27:1087–1092. doi: 10.1016/j.vaccine.2008.11.108. [DOI] [PubMed] [Google Scholar]
- 81.Shoji Y., Chichester J.A., Bi H., Musiychuk K., de la Rosa P., Goldschmidt L., Horsey A., Ugulava N., Palmer G.A., Mett V., et al. Plant-Expressed HA as a Seasonal Influenza Vaccine Candidate. Vaccine. 2008;26:2930–2934. doi: 10.1016/j.vaccine.2008.03.045. [DOI] [PubMed] [Google Scholar]
- 82.Shoji Y., Farrance C.E., Bautista J., Bi H., Musiychuk K., Horsey A., Park H., Jaje J., Green B.J., Shamloul M., et al. A Plant-Based System for Rapid Production of Influenza Vaccine Antigens. Influenza Other Respir. Viruses. 2012;6:204–210. doi: 10.1111/j.1750-2659.2011.00295.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Shoji Y., Jones R.M., Mett V., Chichester J.A., Musiychuk K., Sun X., Tumpey T.M., Green B.J., Shamloul M., Norikane J., et al. A Plant-Produced H1N1 Trimeric Hemagglutinin Protects Mice from a Lethal Influenza Virus Challenge. Hum. Vaccines Immunother. 2013;9:553–560. doi: 10.4161/hv.23234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Phan H.T., Pham V.T., Ho T.T., Pham N.B., Chu H.H., Vu T.H., Abdelwhab E.M., Scheibner D., Mettenleiter T.C., Hanh T.X., et al. Immunization with Plant-Derived Multimeric H5 Hemagglutinins Protect Chicken against Highly Pathogenic Avian Influenza Virus H5N1. Vaccines. 2020;8:593. doi: 10.3390/vaccines8040593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Phan H.T., Ho T.T., Chu H.H., Vu T.H., Gresch U., Conrad U. Neutralizing Immune Responses Induced by Oligomeric H5N1-Hemagglutinins from Plants. Vet. Res. 2017;48:53. doi: 10.1186/s13567-017-0458-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Phan H.T., Hause B., Hause G., Arcalis E., Stoger E., Maresch D., Altmann F., Joensuu J., Conrad U. Influence of Elastin-Like Polypeptide and Hydrophobin on Recombinant Hemagglutinin Accumulations in Transgenic Tobacco Plants. PLoS ONE. 2014;9:e99347. doi: 10.1371/journal.pone.0099347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Phan H.T., Gresch U., Conrad U. In Vitro-Formulated Oligomers of Strep-Tagged Avian Influenza Haemagglutinin Produced in Plants Cause Neutralizing Immune Responses. Front. Bioeng. Biotechnol. 2018;6:115. doi: 10.3389/fbioe.2018.00115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Phan H.T., Pohl J., Floss D.M., Rabenstein F., Veits J., Le B.T., Chu H.H., Hause G., Mettenleiter T., Conrad U. ELPylated Haemagglutinins Produced in Tobacco Plants Induce Potentially Neutralizing Antibodies against H5N1 Viruses in Mice. Plant Biotechnol. J. 2013;11:582–593. doi: 10.1111/pbi.12049. [DOI] [PubMed] [Google Scholar]
- 89.Mett V., Musiychuk K., Bi H., Farrance C.E., Horsey A., Ugulava N., Shoji Y., de la Rosa P., Palmer G.A., Rabindran S., et al. A Plant-Produced Influenza Subunit Vaccine Protects Ferrets against Virus Challenge. Influenza Other Respir. Viruses. 2008;2:33–40. doi: 10.1111/j.1750-2659.2008.00037.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Musiychuk K., Stephenson N., Bi H., Farrance C.E., Orozovic G., Brodelius M., Brodelius P., Horsey A., Ugulava N., Shamloul A.-M., et al. A Launch Vector for the Production of Vaccine Antigens in Plants. Influenza Other Respir. Viruses. 2007;1:19–25. doi: 10.1111/j.1750-2659.2006.00005.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Redkiewicz P., Sirko A., Kamel K.A., Góra-Sochacka A. Plant Expression Systems for Production of Hemagglutinin as a Vaccine against Influenza Virus. Acta Biochim. Pol. 2014;61:551–560. doi: 10.18388/abp.2014_1877. [DOI] [PubMed] [Google Scholar]
- 92.Ceballo Y., Tiel K., López A., Cabrera G., Pérez M., Ramos O., Rosabal Y., Montero C., Menassa R., Depicker A., et al. High Accumulation in Tobacco Seeds of Hemagglutinin Antigen from Avian (H5N1) Influenza. Transgenic Res. 2017;26:775–789. doi: 10.1007/s11248-017-0047-9. [DOI] [PubMed] [Google Scholar]
- 93.Nahampun H.N., Bosworth B., Cunnick J., Mogler M., Wang K. Expression of H3N2 Nucleoprotein in Maize Seeds and Immunogenicity in Mice. Plant Cell Rep. 2015;34:969–980. doi: 10.1007/s00299-015-1758-0. [DOI] [PubMed] [Google Scholar]
- 94.Thuenemann E.C., Lenzi P., Love A.J., Taliansky M., Bécares M., Zuñiga S., Enjuanes L., Zahmanova G.G., Minkov I.N., Matić S., et al. The Use of Transient Expression Systems for the Rapid Production of Virus-like Particles in Plants. Curr. Pharm. Des. 2013;19:5564–5573. doi: 10.2174/1381612811319310011. [DOI] [PubMed] [Google Scholar]
- 95.Zahmanova G.G., Mazalovska M., Takova K.H., Toneva V.T., Minkov I.N., Mardanova E.S., Ravin N.V., Lomonossoff G.P. Rapid High-Yield Transient Expression of Swine Hepatitis E ORF2 Capsid Proteins in Nicotiana Benthamiana Plants and Production of Chimeric Hepatitis E Virus-Like Particles Bearing the M2e Influenza Epitope. Plants. 2019;9:29. doi: 10.3390/plants9010029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Meshcheryakova Y.A., Eldarov M.A., Migunov A.I., Stepanova L.A., Repko I.A., Kiselev C.I., Lomonossoff G.P., Skryabin K.G. Cowpea Mosaic Virus Chimeric Particles Bearing the Ectodomain of Matrix Protein 2 (M2E) of the Influenza a Virus: Production and Characterization. Mol. Biol. 2009;43:685–694. doi: 10.1134/S0026893309040219. [DOI] [PubMed] [Google Scholar]
- 97.Petukhova N.V., Gasanova T.V., Ivanov P.A., Atabekov J.G. High-Level Systemic Expression of Conserved Influenza Epitope in Plants on the Surface of Rod-Shaped Chimeric Particles. Viruses. 2014;6:1789–1800. doi: 10.3390/v6041789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ravin N.V., Kotlyarov R.Y., Mardanova E.S., Kuprianov V.V., Migunov A.I., Stepanova L.A., Tsybalova L.M., Kiselev O.I., Skryabin K.G. Plant-Produced Recombinant Influenza Vaccine Based on Virus-like HBc Particles Carrying an Extracellular Domain of M2 Protein. Biochem. Mosc. 2012;77:33–40. doi: 10.1134/S000629791201004X. [DOI] [PubMed] [Google Scholar]
- 99.Petukhova N.V., Gasanova T.V., Stepanova L.A., Rusova O.A., Potapchuk M.V., Korotkov A.V., Skurat E.V., Tsybalova L.M., Kiselev O.I., Ivanov P.A., et al. Immunogenicity and Protective Efficacy of Candidate Universal Influenza A Nanovaccines Produced in Plants by Tobacco Mosaic Virus-Based Vectors. Curr. Pharm. Des. 2013;19:5587–5600. doi: 10.2174/13816128113199990337. [DOI] [PubMed] [Google Scholar]
- 100.Blokhina E.A., Mardanova E.S., Stepanova L.A., Tsybalova L.M., Ravin N.V. Plant-Produced Recombinant Influenza A Virus Candidate Vaccine Based on Flagellin Linked to Conservative Fragments of M2 Protein and Hemagglutintin. Plants. 2020;9:162. doi: 10.3390/plants9020162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Pal M., Berhanu G., Desalegn C., Kandi V. Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2): An Update. Cureus. 2020;12:e7423. doi: 10.7759/cureus.7423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Liu D.X., Liang J.Q., Fung T.S. Human Coronavirus-229E, -OC43, -NL63, and -HKU1 (Coronaviridae) Encycl. Virol. 2021:428–440. doi: 10.1016/B978-0-12-809633-8.21501-X. [DOI] [Google Scholar]
- 103.Ying W., Hao Y., Zhang Y., Peng W., Qin E., Cai Y., Wei K., Wang J., Chang G., Sun W., et al. Proteomic Analysis on Structural Proteins of Severe Acute Respiratory Syndrome Coronavirus. Proteomics. 2004;4:492–504. doi: 10.1002/pmic.200300676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Yang Y., Du L. SARS-CoV-2 Spike Protein: A Key Target for Eliciting Persistent Neutralizing Antibodies. Signal Transduct. Target. Ther. 2021;6:95. doi: 10.1038/s41392-021-00523-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Tusé D., Nandi S., McDonald K.A., Buyel J.F. The Emergency Response Capacity of Plant-Based Biopharmaceutical Manufacturing-What It Is and What It Could Be. Front. Plant Sci. 2020;11:1573. doi: 10.3389/fpls.2020.594019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Capell T., Twyman R.M., Armario-Najera V., Ma J.K.-C., Schillberg S., Christou P. Potential Applications of Plant Biotechnology against SARS-CoV-2. Trends Plant Sci. 2020;25:635–643. doi: 10.1016/j.tplants.2020.04.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Rosales-Mendoza S. Will Plant-Made Biopharmaceuticals Play a Role in the Fight against COVID-19? Expert Opin. Biol. Ther. 2020;20:545–548. doi: 10.1080/14712598.2020.1752177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Ward B.J., Gobeil P., Séguin A., Atkins J., Boulay I., Charbonneau P.-Y., Couture M., D’Aoust M.-A., Dhaliwall J., Finkle C., et al. Phase 1 Randomized Trial of a Plant-Derived Virus-like Particle Vaccine for COVID-19. Nat. Med. 2021;27:1071–1078. doi: 10.1038/s41591-021-01370-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Medicago Inc Positive Results from Phase 3 Study of Medicago’s Plant-Based COVID-19 Vaccine Candidate Announced. [(accessed on 14 January 2022)]. Available online: https://www.news-medical.net/news/20211209/Positive-results-from-Phase-3-study-of-Medicagoe28099s-plant-based-COVID-19-vaccine-candidate-announced.aspx.
- 110.Kentucky BioProcessing, Inc A Phase I/II, First-in-Human, Observer-Blinded, Randomized, Placebo-Controlled, Parallel Group Study to Evaluate the Safety and Immunogenicity of TAP-COVID-19 SARS-CoV-2 Vaccine with CpG Adjuvant in Healthy Adults Aged 18–49 and 50–85. Clinicaltrials.gov. 2021. [(accessed on 14 January 2022)]; Available online: https://clinicaltrials.gov/ct2/show/NCT04473690.
- 111.Medicago Inc Medicago and GSK Start Phase 3 Trial of Adjuvanted COVID-19 Vaccine Candidate. [(accessed on 12 January 2022)]. Available online: https://medicago.com/en/press-release/medicago-and-gsk-start-phase-3-trial-of-adjuvanted-covid-19-vaccine-candidate/
- 112.iBio Inc IBio Reports Successful Preclinical Immunization Studies with Next-Gen Nucleocapsid COVID-19 Vaccine Candidate. [(accessed on 20 November 2021)]. Available online: https://ibioinc.com/ibio-reports-successful-preclinical-immunization-studies-with-next-gen-nucleocapsid-covid-19-vaccine-candidate/
- 113.British American Tobacco BAT Makes Progress on COVID-19 Vaccine Provides Community Support. [(accessed on 20 November 2021)]. Available online: https://www.bat.com/group/sites/UK__9D9KCY.nsf/vwPagesWebLive/DOBPMBZC#.
- 114.Hardy A. IBIO-201 Demonstrates Ability to Elicit Anti-SARS-CoV-2 Immune Response in Preclinical Studies. [(accessed on 20 November 2021)]. Available online: https://biotuesdays.com/2020/08/10/ibio-updates-ibio-201-covid-19-vaccine-candidate/
- 115.Baiya Phytopharm Co Ltd Baiya SARS-CoV-2 Vax 1 Vaccine–COVID-19 Vaccine Tracker. [(accessed on 20 November 2021)]. Available online: https://covid19.trackvaccines.org/vaccines/130/
- 116.Peyret H., Steele J.F.C., Jung J.-W., Thuenemann E.C., Meshcheriakova Y., Lomonossoff G.P. Producing Vaccines against Enveloped Viruses in Plants: Making the Impossible, Difficult. Vaccines. 2021;9:780. doi: 10.3390/vaccines9070780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Pogrebnyak N., Golovkin M., Andrianov V., Spitsin S., Smirnov Y., Egolf R., Koprowski H. Severe Acute Respiratory Syndrome (SARS) S Protein Production in Plants: Development of Recombinant Vaccine. Proc. Natl. Acad. Sci. USA. 2005;102:9062–9067. doi: 10.1073/pnas.0503760102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Li H.-Y., Ramalingam S., Chye M.-L. Accumulation of Recombinant SARS-CoV Spike Protein in Plant Cytosol and Chloroplasts Indicate Potential for Development of Plant-Derived Oral Vaccines. Exp. Biol. Med. 2006;231:1346–1352. doi: 10.1177/153537020623100808. [DOI] [PubMed] [Google Scholar]
- 119.Zheng N., Xia R., Yang C., Yin B., Li Y., Duan C., Liang L., Guo H., Xie Q. Boosted Expression of the SARS-CoV Nucleocapsid Protein in Tobacco and Its Immunogenicity in Mice. Vaccine. 2009;27:5001–5007. doi: 10.1016/j.vaccine.2009.05.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Demurtas O.C., Massa S., Illiano E., De Martinis D., Chan P.K.S., Di Bonito P., Franconi R. Antigen Production in Plant to Tackle Infectious Diseases Flare Up: The Case of SARS. Front. Plant Sci. 2016;7:54. doi: 10.3389/fpls.2016.00054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Mamedov T., Yuksel D., Ilgın M., Gürbüzaslan I., Gulec B., Mammadova G., Ozdarendeli A., Yetiskin H., Kaplan B., Islam Pavel S.T., et al. Production and Characterization of Nucleocapsid and RBD Cocktail Antigens of SARS-CoV-2 in Nicotiana Benthamiana Plant as a Vaccine Candidate against COVID-19. Vaccines. 2021;9:1337. doi: 10.3390/vaccines9111337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Maharjan P.M., Cheon J., Jung J., Kim H., Lee J., Song M., Jeong G.U., Kwon Y., Shim B., Choe S. Plant-Expressed Receptor Binding Domain of the SARS-CoV-2 Spike Protein Elicits Humoral Immunity in Mice. Vaccines. 2021;9:978. doi: 10.3390/vaccines9090978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Hemmati F., Hemmati-Dinarvand M., Karimzade M., Rutkowska D., Eskandari M.H., Khanizadeh S., Afsharifar A. Plant-Derived VLP: A Worthy Platform to Produce Vaccine against SARS-CoV-2. Biotechnol. Lett. 2021 doi: 10.1007/s10529-021-03211-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Maharjan P.M., Choe S. Plant-Based COVID-19 Vaccines: Current Status, Design, and Development Strategies of Candidate Vaccines. Vaccines. 2021;9:992. doi: 10.3390/vaccines9090992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Diego-Martin B., González B., Vazquez-Vilar M., Selma S., Mateos-Fernández R., Gianoglio S., Fernández-del-Carmen A., Orzáez D. Pilot Production of SARS-CoV-2 Related Proteins in Plants: A Proof of Concept for Rapid Repurposing of Indoor Farms Into Biomanufacturing Facilities. Front. Plant Sci. 2020;11:2101. doi: 10.3389/fpls.2020.612781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Kumar M., Kumari N., Thakur N., Bhatia S.K., Saratale G.D., Ghodake G., Mistry B.M., Alavilli H., Kishor D.S., Du X., et al. A Comprehensive Overview on the Production of Vaccines in Plant-Based Expression Systems and the Scope of Plant Biotechnology to Combat against SARS-CoV-2 Virus Pandemics. Plants. 2021;10:1213. doi: 10.3390/plants10061213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Feldmann H., Jones S., Klenk H.-D., Schnittler H.-J. Ebola Virus: From Discovery to Vaccine. Nat. Rev. Immunol. 2003;3:677–685. doi: 10.1038/nri1154. [DOI] [PubMed] [Google Scholar]
- 128.King A.M., Lefkowitz E., Adams M.J., Carstens E.B. Virus Taxonomy: Ninth Report of the International Committee on Taxonomy of Viruses. Elsevier; Amsterdam, The Netherlands: 2011. [Google Scholar]
- 129.Beer B., Kurth R., Bukreyev A. Characteristics of Filoviridae: Marburg and Ebola Viruses. Naturwissenschaften. 1999;86:8–17. doi: 10.1007/s001140050562. [DOI] [PubMed] [Google Scholar]
- 130.Feldmann H., Sanchez A., Geisbert T. Fields Virology. 6th ed. Volume 1. Wolters Kluwer Health Adis (ESP); London, UK: 2013. Filoviridae: Marburg and Ebola Viruses. [Google Scholar]
- 131.Mehedi M., Falzarano D., Seebach J., Hu X., Carpenter M.S., Schnittler H.-J., Feldmann H. A New Ebola Virus Nonstructural Glycoprotein Expressed through RNA Editing. J. Virol. 2011;85:5406–5414. doi: 10.1128/JVI.02190-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Baize S. A Single Shot against Ebola and Marburg Virus. Nat. Med. 2005;11:720–721. doi: 10.1038/nm0705-720. [DOI] [PubMed] [Google Scholar]
- 133.Geisbert T.W., Geisbert J.B., Leung A., Daddario-DiCaprio K.M., Hensley L.E., Grolla A., Feldmann H. Single-Injection Vaccine Protects Nonhuman Primates against Infection with Marburg Virus and Three Species of Ebola Virus. J. Virol. 2009;83:7296–7304. doi: 10.1128/JVI.00561-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Sullivan N.J., Sanchez A., Rollin P.E., Yang Z.Y., Nabel G.J. Development of a Preventive Vaccine for Ebola Virus Infection in Primates. Nature. 2000;408:605–609. doi: 10.1038/35046108. [DOI] [PubMed] [Google Scholar]
- 135.Geisbert T.W., Bausch D.G., Feldmann H. Prospects for Immunisation against Marburg and Ebola Viruses. Rev. Med. Virol. 2010;20:344–357. doi: 10.1002/rmv.661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Sridhar S. Clinical Development of Ebola Vaccines. Ther. Adv. Vaccines. 2015;3:125–138. doi: 10.1177/2051013615611017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Wang Y., Li J., Hu Y., Liang Q., Wei M., Zhu F. Ebola Vaccines in Clinical Trial: The Promising Candidates. Hum. Vaccines Immunother. 2017;13:153–168. doi: 10.1080/21645515.2016.1225637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Nieto-Gómez R., Angulo C., Monreal-Escalante E., Govea-Alonso D.O., De Groot A.S., Rosales-Mendoza S. Design of a Multiepitopic Zaire Ebolavirus Protein and Its Expression in Plant Cells. J. Biotechnol. 2019;295:41–48. doi: 10.1016/j.jbiotec.2019.02.003. [DOI] [PubMed] [Google Scholar]
- 139.Phoolcharoen W., Bhoo S.H., Lai H., Ma J., Arntzen C.J., Chen Q., Mason H.S. Expression of an Immunogenic Ebola Immune Complex in Nicotiana Benthamiana. Plant Biotechnol. J. 2011;9:807–816. doi: 10.1111/j.1467-7652.2011.00593.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Phoolcharoen W., Dye J.M., Kilbourne J., Piensook K., Pratt W.D., Arntzen C.J., Chen Q., Mason H.S., Herbst-Kralovetz M.M. A Nonreplicating Subunit Vaccine Protects Mice against Lethal Ebola Virus Challenge. Proc. Natl. Acad. Sci. USA. 2011;108:20695–20700. doi: 10.1073/pnas.1117715108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Papa A., Mirazimi A., Köksal I., Estrada-Pena A., Feldmann H. Recent Advances in Research on Crimean-Congo Hemorrhagic Fever. J. Clin. Virol. 2015;64:137–143. doi: 10.1016/j.jcv.2014.08.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Ergönül Ö. Crimean-Congo Haemorrhagic Fever. Lancet Infect. Dis. 2006;6:203–214. doi: 10.1016/S1473-3099(06)70435-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Ruiz S.I., Zumbrun E.E., Nalca A. Animal Models of Human Viral Diseases. Anim. Models Study Hum. Dis. 2017:853–901. doi: 10.1016/B978-0-12-809468-6.00033-4. [DOI] [Google Scholar]
- 144.Maes P., Alkhovsky S.V., Bào Y., Beer M., Birkhead M., Briese T., Buchmeier M.J., Calisher C.H., Charrel R.N., Choi I.R., et al. Taxonomy of the Family Arenaviridae and the Order Bunyavirales: Update 2018. Arch. Virol. 2018;163:2295–2310. doi: 10.1007/s00705-018-3843-5. [DOI] [PubMed] [Google Scholar]
- 145.Haferkamp S., Fernando L., Schwarz T.F., Feldmann H., Flick R. Intracellular Localization of Crimean-Congo Hemorrhagic Fever (CCHF) Virus Glycoproteins. Virol. J. 2005;2:42. doi: 10.1186/1743-422X-2-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Ghiasi S.M., Salmanian A.H., Chinikar S., Zakeri S. Mice Orally Immunized with a Transgenic Plant Expressing the Glycoprotein of Crimean-Congo Hemorrhagic Fever Virus. Clin. Vaccine Immunol. 2011;18:2031–2037. doi: 10.1128/CVI.05352-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Dowall S.D., Carroll M.W., Hewson R. Development of Vaccines against Crimean-Congo Haemorrhagic Fever Virus. Vaccine. 2017;35:6015–6023. doi: 10.1016/j.vaccine.2017.05.031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Shoemaker T., Boulianne C., Vincent M.J., Pezzanite L., Al-Qahtani M.M., Al-Mazrou Y., Khan A.S., Rollin P.E., Swanepoel R., Ksiazek T.G., et al. Genetic Analysis of Viruses Associated with Emergence of Rift Valley Fever in Saudi Arabia and Yemen, 2000-01. Emerg. Infect. Dis. 2002;8:1415–1420. doi: 10.3201/eid0812.020195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Kwaśnik M., Rożek W., Rola J. Rift Valley Fever—A Growing Threat to Humans and Animals. J. Vet. Res. 2021;65:7–14. doi: 10.2478/jvetres-2021-0009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Linthicum K.J., Britch S.C., Anyamba A. Rift Valley Fever: An Emerging Mosquito-Borne Disease. Annu. Rev. Entomol. 2016;61:395–415. doi: 10.1146/annurev-ento-010715-023819. [DOI] [PubMed] [Google Scholar]
- 151.Balenghien T., Cardinale E., Chevalier V., Elissa N., Failloux A.-B., Jean Jose Nipomichene T.N., Nicolas G., Rakotoharinome V.M., Roger M., Zumbo B. Towards a Better Understanding of Rift Valley Fever Epidemiology in the South-West of the Indian Ocean. Vet. Res. 2013;44:78. doi: 10.1186/1297-9716-44-78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Pepin M., Bouloy M., Bird B.H., Kemp A., Paweska J. Rift Valley Fever Virus (Bunyaviridae: Phlebovirus): An Update on Pathogenesis, Molecular Epidemiology, Vectors, Diagnostics and Prevention. Vet. Res. 2010;41:61. doi: 10.1051/vetres/2010033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Gaudreault N.N., Indran S.V., Balaraman V., Wilson W.C., Richt J.A. Molecular Aspects of Rift Valley Fever Virus and the Emergence of Reassortants. Virus Genes. 2019;55:1–11. doi: 10.1007/s11262-018-1611-y. [DOI] [PubMed] [Google Scholar]
- 154.Faburay B., Wilson W., McVey D.S., Drolet B.S., Weingartl H., Madden D., Young A., Ma W., Richt J.A. Rift Valley Fever Virus Structural and Nonstructural Proteins: Recombinant Protein Expression and Immunoreactivity against Antisera from Sheep. Vector-Borne Zoonotic Dis. 2013;13:619–629. doi: 10.1089/vbz.2012.1285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Bouloy M., Weber F. Molecular Biology of Rift Valley Fever Virus. Open Virol. J. 2010;4 doi: 10.2174/1874357901004010008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Mbewana S., Meyers A.E., Rybicki E.P. Chimaeric Rift Valley Fever Virus-Like Particle Vaccine Candidate Production in Nicotiana Benthamiana. Biotechnol. J. 2019;14:e1800238. doi: 10.1002/biot.201800238. [DOI] [PubMed] [Google Scholar]
- 157.Kalbina I., Lagerqvist N., Moiane B., Ahlm C., Andersson S., Strid Å., Falk K.I. Arabidopsis Thaliana Plants Expressing Rift Valley Fever Virus Antigens: Mice Exhibit Systemic Immune Responses as the Result of Oral Administration of the Transgenic Plants. Protein Expr. Purif. 2016;127:61–67. doi: 10.1016/j.pep.2016.07.003. [DOI] [PubMed] [Google Scholar]
- 158.Chandy S., Mathai D. Globally Emerging Hantaviruses: An Overview. Indian J. Med. Microbiol. 2017;35:165–175. doi: 10.4103/ijmm.IJMM_16_429. [DOI] [PubMed] [Google Scholar]
- 159.Hangaragi P.S. Hantavirus: An Emerging Global Threat. Asian J. Oral Health Allied Sci. 2020;10:4. doi: 10.25259/AJOHAS_6_2020. [DOI] [Google Scholar]
- 160.Liu R., Ma H., Shu J., Zhang Q., Han M., Liu Z., Jin X., Zhang F., Wu X. Vaccines and Therapeutics against Hantaviruses. Front. Microbiol. 2020;10:2989. doi: 10.3389/fmicb.2019.02989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Muyangwa M., Martynova E.V., Khaiboullina S.F., Morzunov S.P., Rizvanov A.A. Hantaviral Proteins: Structure, Functions, and Role in Hantavirus Infection. Front. Microbiol. 2015;6:1326. doi: 10.3389/fmicb.2015.01326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Yoshimatsu K., Arikawa J. Antigenic Properties of N Protein of Hantavirus. Viruses. 2014;6:3097–3109. doi: 10.3390/v6083097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Khattak S., Darai G., Süle S., Rösen-Wolff A. Characterization of Expression of Puumala Virus Nucleocapsid Protein in Transgenic Plants. Intervirology. 2002;45:334–339. doi: 10.1159/000067926. [DOI] [PubMed] [Google Scholar]
- 164.Khattak S., Darai G., Rösen-Wolff A. Puumala Virus Nucleocapsid Protein Expressed in Transgenic Plants Is Not Immunogenic after Oral Administration. Virus Genes. 2004;29:109–116. doi: 10.1023/B:VIRU.0000032794.24032.72. [DOI] [PubMed] [Google Scholar]
- 165.Peyret H., Gehin A., Thuenemann E.C., Blond D., Turabi A.E., Beales L., Clarke D., Gilbert R.J.C., Fry E.E., Stuart D.I., et al. Tandem Fusion of Hepatitis B Core Antigen Allows Assembly of Virus-Like Particles in Bacteria and Plants with Enhanced Capacity to Accommodate Foreign Proteins. PLoS ONE. 2015;10:e0120751. doi: 10.1371/journal.pone.0120751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Ulrich R., Koletzki D., Lachmann S., Lundkvist A., Zankl A., Kazaks A., Kurth A., Gelderblom H.R., Borisova G., Meisel H., et al. New Chimaeric Hepatitis B Virus Core Particles Carrying Hantavirus (Serotype Puumala) Epitopes: Immunogenicity and Protection against Virus Challenge. J. Biotechnol. 1999;73:141–153. doi: 10.1016/S0168-1656(99)00117-0. [DOI] [PubMed] [Google Scholar]
- 167.Geldmacher A., Skrastina D., Petrovskis I., Borisova G., Berriman J.A., Roseman A.M., Crowther R.A., Fischer J., Musema S., Gelderblom H.R., et al. An Amino-Terminal Segment of Hantavirus Nucleocapsid Protein Presented on Hepatitis B Virus Core Particles Induces a Strong and Highly Cross-Reactive Antibody Response in Mice. Virology. 2004;323:108–119. doi: 10.1016/j.virol.2004.02.022. [DOI] [PubMed] [Google Scholar]
- 168.Ulrich R., Lundkvist A., Meisel H., Koletzki D., Sjölander K.B., Gelderblom H.R., Borisova G., Schnitzler P., Darai G., Krüger D.H. Chimaeric HBV Core Particles Carrying a Defined Segment of Puumala Hantavirus Nucleocapsid Protein Evoke Protective Immunity in an Animal Model. Vaccine. 1998;16:272–280. doi: 10.1016/S0264-410X(97)00172-2. [DOI] [PubMed] [Google Scholar]
- 169.Koletzki D., Biel S.S., Meisel H., Nugel E., Gelderblom H.R., Krüger D.H., Ulrich R. HBV Core Particles Allow the Insertion and Surface Exposure of the Entire Potentially Protective Region of Puumala Hantavirus Nucleocapsid Protein. Biol. Chem. 1999;380:325–333. doi: 10.1515/BC.1999.044. [DOI] [PubMed] [Google Scholar]
- 170.Koletzki D., Lundkvist A., Sjölander K.B., Gelderblom H.R., Niedrig M., Meisel H., Krüger D.H., Ulrich R. Puumala (PUU) Hantavirus Strain Differences and Insertion Positions in the Hepatitis B Virus Core Antigen Influence B-Cell Immunogenicity and Protective Potential of Core-Derived Particles. Virology. 2000;276:364–375. doi: 10.1006/viro.2000.0540. [DOI] [PubMed] [Google Scholar]
- 171.Geldmacher A., Skrastina D., Borisova G., Petrovskis I., Krüger D.H., Pumpens P., Ulrich R. A Hantavirus Nucleocapsid Protein Segment Exposed on Hepatitis B Virus Core Particles Is Highly Immunogenic in Mice When Applied without Adjuvants or in the Presence of Pre-Existing Anti-Core Antibodies. Vaccine. 2005;23:3973–3983. doi: 10.1016/j.vaccine.2005.02.025. [DOI] [PubMed] [Google Scholar]
- 172.Simon F., Savini H., Parola P. Chikungunya: A Paradigm of Emergence and Globalization of Vector-Borne Diseases. Med. Clin. N. Am. 2008;92:1323–1343. doi: 10.1016/j.mcna.2008.07.008. [DOI] [PubMed] [Google Scholar]
- 173.Simon F., Javelle E., Oliver M., Leparc-Goffart I., Marimoutou C. Chikungunya Virus Infection. Curr. Infect. Dis. Rep. 2011;13:218–228. doi: 10.1007/s11908-011-0180-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Consuegra-Rodríguez M.P., Hidalgo-Zambrano D.M., Vásquez-Serna H., Jimenez-Canizales C.E., Parra-Valencia E., Rodriguez-Morales A.J. Post-Chikungunya Chronic Inflammatory Rheumatism: Follow-up of Cases after 1 Year of Infection in Tolima, Colombia. Travel Med. Infect. Dis. 2018;21:62–68. doi: 10.1016/j.tmaid.2017.11.013. [DOI] [PubMed] [Google Scholar]
- 175.Cunha M.S., Costa P.A.G., Correa I.A., de Souza M.R.M., Calil P.T., da Silva G.P.D., Costa S.M., Fonseca V.W.P., da Costa L.J. Chikungunya Virus: An Emergent Arbovirus to the South American Continent and a Continuous Threat to the World. Front. Microbiol. 2020;11:1297. doi: 10.3389/fmicb.2020.01297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Rashad A.A., Mahalingam S., Keller P.A. Chikungunya Virus: Emerging Targets and New Opportunities for Medicinal Chemistry. J. Med. Chem. 2014;57:1147–1166. doi: 10.1021/jm400460d. [DOI] [PubMed] [Google Scholar]
- 177.Jin J., Simmons G. Antiviral Functions of Monoclonal Antibodies against Chikungunya Virus. Viruses. 2019;11:305. doi: 10.3390/v11040305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Lum F.-M., Ng L.F.P. Cellular and Molecular Mechanisms of Chikungunya Pathogenesis. Antivir. Res. 2015;120:165–174. doi: 10.1016/j.antiviral.2015.06.009. [DOI] [PubMed] [Google Scholar]
- 179.Gao S., Song S., Zhang L. Recent Progress in Vaccine Development Against Chikungunya Virus. Front. Microbiol. 2019;10:2881. doi: 10.3389/fmicb.2019.02881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Saraswat S., Athmaram T.N., Parida M., Agarwal A., Saha A., Dash P.K. Expression and Characterization of Yeast Derived Chikungunya Virus Like Particles (CHIK-VLPs) and Its Evaluation as a Potential Vaccine Candidate. PLoS Negl. Trop. Dis. 2016;10:e0004782. doi: 10.1371/journal.pntd.0004782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Metz S.W., Gardner J., Geertsema C., Le T.T., Goh L., Vlak J.M., Suhrbier A., Pijlman G.P. Effective Chikungunya Virus-like Particle Vaccine Produced in Insect Cells. PLoS Negl. Trop. Dis. 2013;7:e2124. doi: 10.1371/journal.pntd.0002124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Noranate N., Takeda N., Chetanachan P., Sittisaman P., A-nuegoonpipat A., Anantapreecha S. Characterization of Chikungunya Virus-Like Particles. PLoS ONE. 2014;9:e108169. doi: 10.1371/journal.pone.0108169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Arévalo M.T., Huang Y., Jones C.A., Ross T.M. Vaccination with a Chikungunya Virus-like Particle Vaccine Exacerbates Disease in Aged Mice. PLoS Negl. Trop. Dis. 2019;13:e0007316. doi: 10.1371/journal.pntd.0007316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Cook S., Holmes E.C. A Multigene Analysis of the Phylogenetic Relationships among the Flaviviruses (Family: Flaviviridae) and the Evolution of Vector Transmission. Arch. Virol. 2006;151:309–325. doi: 10.1007/s00705-005-0626-6. [DOI] [PubMed] [Google Scholar]
- 185.Unni S.K., Růžek D., Chhatbar C., Mishra R., Johri M.K., Singh S.K. Japanese Encephalitis Virus: From Genome to Infectome. Microbes Infect. 2011;13:312–321. doi: 10.1016/j.micinf.2011.01.002. [DOI] [PubMed] [Google Scholar]
- 186.Mackenzie J.S., Gubler D.J., Petersen L.R. Emerging Flaviviruses: The Spread and Resurgence of Japanese Encephalitis, West Nile and Dengue Viruses. Nat. Med. 2004;10:S98–S109. doi: 10.1038/nm1144. [DOI] [PubMed] [Google Scholar]
- 187.Sambri V., Capobianchi M., Charrel R., Fyodorova M., Gaibani P., Gould E., Niedrig M., Papa A., Pierro A., Rossini G., et al. West Nile Virus in Europe: Emergence, Epidemiology, Diagnosis, Treatment, and Prevention. Clin. Microbiol. Infect. Off. Publ. Eur. Soc. Clin. Microbiol. Infect. Dis. 2013;19:699–704. doi: 10.1111/1469-0691.12211. [DOI] [PubMed] [Google Scholar]
- 188.Artsob H., Gubler D.J., Enria D.A., Morales M.A., Pupo M., Bunning M.L., Dudley J.P. West Nile Virus in the New World: Trends in the Spread and Proliferation of West Nile Virus in the Western Hemisphere. Zoonoses Public Health. 2009;56:357–369. doi: 10.1111/j.1863-2378.2008.01207.x. [DOI] [PubMed] [Google Scholar]
- 189.Hayes E.B., Gubler D.J. West Nile Virus: Epidemiology and Clinical Features of an Emerging Epidemic in the United States. Annu. Rev. Med. 2006;57:181–194. doi: 10.1146/annurev.med.57.121304.131418. [DOI] [PubMed] [Google Scholar]
- 190.Hubálek Z., Halouzka J. West Nile Fever--a Reemerging Mosquito-Borne Viral Disease in Europe. Emerg. Infect. Dis. 1999;5:643–650. doi: 10.3201/eid0505.990505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Hayes E.B., Sejvar J.J., Zaki S.R., Lanciotti R.S., Bode A.V., Campbell G.L. Virology, Pathology, and Clinical Manifestations of West Nile Virus Disease. Emerg. Infect. Dis. 2005;11:1174–1179. doi: 10.3201/eid1108.050289b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Mukhopadhyay S., Kuhn R.J., Rossmann M.G. A Structural Perspective of the Flavivirus Life Cycle. Nat. Rev. Microbiol. 2005;3:13–22. doi: 10.1038/nrmicro1067. [DOI] [PubMed] [Google Scholar]
- 193.Martín-Acebes M.A., Saiz J.-C. West Nile Virus: A Re-Emerging Pathogen Revisited. World J. Virol. 2012;1:51–70. doi: 10.5501/wjv.v1.i2.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Ulbert S. West Nile Virus: The Complex Biology of an Emerging Pathogen. Intervirology. 2011;54:171–184. doi: 10.1159/000328320. [DOI] [PubMed] [Google Scholar]
- 195.De Filette M., Ulbert S., Diamond M.S., Sanders N.N. Recent Progress in West Nile Virus Diagnosis and Vaccination. Vet. Res. 2012;43:16. doi: 10.1186/1297-9716-43-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Nybakken G.E., Oliphant T., Johnson S., Burke S., Diamond M.S., Fremont D.H. Structural Basis of West Nile Virus Neutralization by a Therapeutic Antibody. Nature. 2005;437:764–769. doi: 10.1038/nature03956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Chu J.J.H., Ng M.L. Infectious Entry of West Nile Virus Occurs through a Clathrin-Mediated Endocytic Pathway. J. Virol. 2004;78:10543–10555. doi: 10.1128/JVI.78.19.10543-10555.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Nybakken G.E., Nelson C.A., Chen B.R., Diamond M.S., Fremont D.H. Crystal Structure of the West Nile Virus Envelope Glycoprotein. J. Virol. 2006;80:11467–11474. doi: 10.1128/JVI.01125-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 199.Modis Y., Ogata S., Clements D., Harrison S.C. Structure of the Dengue Virus Envelope Protein after Membrane Fusion. Nature. 2004;427:313–319. doi: 10.1038/nature02165. [DOI] [PubMed] [Google Scholar]
- 200.Chen Q. Plant-Made Vaccines against West Nile Virus Are Potent, Safe, and Economically Feasible. Biotechnol. J. 2015;10:671–680. doi: 10.1002/biot.201400428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Lai H., Paul A.M., Sun H., He J., Yang M., Bai F., Chen Q. A Plant-Produced Vaccine Protects Mice against Lethal West Nile Virus Infection without Enhancing Zika or Dengue Virus Infectivity. Vaccine. 2018;36:1846–1852. doi: 10.1016/j.vaccine.2018.02.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 202.He J., Lai H., Brock C., Chen Q. A Novel System for Rapid and Cost-Effective Production of Detection and Diagnostic Reagents of West Nile Virus in Plants. J. Biomed. Biotechnol. 2011;2012:e106783. doi: 10.1155/2012/106783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.He J., Peng L., Lai H., Hurtado J., Stahnke J., Chen Q. A Plant-Produced Antigen Elicits Potent Immune Responses against West Nile Virus in Mice. BioMed Res. Int. 2014;2014:e952865. doi: 10.1155/2014/952865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.He J., Lai H., Esqueda A., Chen Q. Plant-Produced Antigen Displaying Virus-Like Particles Evokes Potent Antibody Responses against West Nile Virus in Mice. Vaccines. 2021;9:60. doi: 10.3390/vaccines9010060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 205.Thrane S., Janitzek C.M., Matondo S., Resende M., Gustavsson T., de Jongh W.A., Clemmensen S., Roeffen W., van de Vegte-Bolmer M., van Gemert G.J., et al. Bacterial Superglue Enables Easy Development of Efficient Virus-like Particle Based Vaccines. J. Nanobiotechnol. 2016;14:30. doi: 10.1186/s12951-016-0181-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Stander J., Chabeda A., Rybicki E.P., Meyers A.E. A Plant-Produced Virus-Like Particle Displaying Envelope Protein Domain III Elicits an Immune Response against West Nile Virus in Mice. Front. Plant Sci. 2021;12:1915. doi: 10.3389/fpls.2021.738619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Murugesan A., Manoharan M. Chapter 16—Dengue Virus. In: Ennaji M.M., editor. Emerging and Reemerging Viral Pathogens. Academic Press; Cambridge, MA, USA: 2020. pp. 281–359. [Google Scholar]
- 208.Bäck A.T., Lundkvist A. Dengue Viruses—An Overview. Infect. Ecol. Epidemiol. 2013;3:19839. doi: 10.3402/iee.v3i0.19839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Zhang X., Zhang Y., Jia R., Wang M., Yin Z., Cheng A. Structure and Function of Capsid Protein in Flavivirus Infection and Its Applications in the Development of Vaccines and Therapeutics. Vet. Res. 2021;52:98. doi: 10.1186/s13567-021-00966-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Yung C.-F., Lee K.-S., Thein T.-L., Tan L.-K., Gan V.C., Wong J.G.X., Lye D.C., Ng L.-C., Leo Y.-S. Dengue Serotype-Specific Differences in Clinical Manifestation, Laboratory Parameters and Risk of Severe Disease in Adults, Singapore. Am. J. Trop. Med. Hyg. 2015;92:999–1005. doi: 10.4269/ajtmh.14-0628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Leng C.-H., Liu S.-J., Tsai J.-P., Li Y.-S., Chen M.-Y., Liu H.-H., Lien S.-P., Yueh A., Hsiao K.-N., Lai L.-W., et al. A Novel Dengue Vaccine Candidate That Induces Cross-Neutralizing Antibodies and Memory Immunity. Microbes Infect. 2009;11:288–295. doi: 10.1016/j.micinf.2008.12.004. [DOI] [PubMed] [Google Scholar]
- 212.Chen H.-W., Liu S.-J., Li Y.-S., Liu H.-H., Tsai J.-P., Chiang C.-Y., Chen M.-Y., Hwang C.-S., Huang C.-C., Hu H.-M., et al. A Consensus Envelope Protein Domain III Can Induce Neutralizing Antibody Responses against Serotype 2 of Dengue Virus in Non-Human Primates. Arch. Virol. 2013;158:1523–1531. doi: 10.1007/s00705-013-1639-1. [DOI] [PubMed] [Google Scholar]
- 213.Carlos Marques L.É., da Silva B.B., Lisboa Magalhães I.C., Marques Mendes M.M., de Almeida L.M., Florindo Guedes M.I. Production of Dengue 2 Envelope Domain III in Plant Using CPMV—Based Vector System. BMC Proc. 2014;8:P80. doi: 10.1186/1753-6561-8-S4-P80. [DOI] [Google Scholar]
- 214.Kanagaraj A.P., Verma D., Daniell H. Expression of Dengue-3 Premembrane and Envelope Polyprotein in Lettuce Chloroplasts. Plant Mol. Biol. 2011;76:323–333. doi: 10.1007/s11103-011-9766-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Martínez C.A., Topal E., Giulietti A.M., Talou J.R., Mason H. Exploring Different Strategies to Express Dengue Virus Envelope Protein in a Plant System. Biotechnol. Lett. 2010;32:867–875. doi: 10.1007/s10529-010-0236-6. [DOI] [PubMed] [Google Scholar]
- 216.Gottschamel J., Lössl A., Ruf S., Wang Y., Skaugen M., Bock R., Clarke J.L. Production of Dengue Virus Envelope Protein Domain III-Based Antigens in Tobacco Chloroplasts Using Inducible and Constitutive Expression Systems. Plant Mol. Biol. 2016;91:497–512. doi: 10.1007/s11103-016-0484-5. [DOI] [PubMed] [Google Scholar]
- 217.van Eerde A., Gottschamel J., Bock R., Hansen K.E.A., Munang’andu H.M., Daniell H., Liu Clarke J. Production of Tetravalent Dengue Virus Envelope Protein Domain III Based Antigens in Lettuce Chloroplasts and Immunologic Analysis for Future Oral Vaccine Development. Plant Biotechnol. J. 2019;17:1408–1417. doi: 10.1111/pbi.13065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Kim M.-Y., Reljic R., Kilbourne J., Ceballos-Olvera I., Yang M.-S., Reyes-del Valle J., Mason H.S. Novel Vaccination Approach for Dengue Infection Based on Recombinant Immune Complex Universal Platform. Vaccine. 2015;33:1830–1838. doi: 10.1016/j.vaccine.2015.02.036. [DOI] [PubMed] [Google Scholar]
- 219.Kim M.-Y., Yang M.-S., Kim T.-G. Expression of Dengue Virus E Glycoprotein Domain III in Non-Nicotine Transgenic Tobacco Plants. Biotechnol. Bioprocess Eng. 2009;14:725–730. doi: 10.1007/s12257-009-3011-6. [DOI] [Google Scholar]
- 220.Saejung W., Fujiyama K., Takasaki T., Ito M., Hori K., Malasit P., Watanabe Y., Kurane I., Seki T. Production of Dengue 2 Envelope Domain III in Plant Using TMV-Based Vector System. Vaccine. 2007;25:6646–6654. doi: 10.1016/j.vaccine.2007.06.029. [DOI] [PubMed] [Google Scholar]
- 221.Ponndorf D., Meshcheriakova Y., Thuenemann E.C., Dobon Alonso A., Overman R., Holton N., Dowall S., Kennedy E., Stocks M., Lomonossoff G.P., et al. Plant-Made Dengue Virus-like Particles Produced by Co-Expression of Structural and Non-Structural Proteins Induce a Humoral Immune Response in Mice. Plant Biotechnol. J. 2021;19:745–756. doi: 10.1111/pbi.13501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Gorshkov K., Shiryaev S.A., Fertel S., Lin Y.-W., Huang C.-T., Pinto A., Farhy C., Strongin A.Y., Zheng W., Terskikh A.V. Zika Virus: Origins, Pathological Action, and Treatment Strategies. Front. Microbiol. 2019;9:3252. doi: 10.3389/fmicb.2018.03252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Gregory C.J., Oduyebo T., Brault A.C., Brooks J.T., Chung K.-W., Hills S., Kuehnert M.J., Mead P., Meaney-Delman D., Rabe I., et al. Modes of Transmission of Zika Virus. J. Infect. Dis. 2017;216:S875–S883. doi: 10.1093/infdis/jix396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Antoniou E., Orovou E., Sarella A., Iliadou M., Rigas N., Palaska E., Iatrakis G., Dagla M. Zika Virus and the Risk of Developing Microcephaly in Infants: A Systematic Review. Int. J. Environ. Res. Public Health. 2020;17:3806. doi: 10.3390/ijerph17113806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.de Araújo T.V.B., Rodrigues L.C., de Alencar Ximenes R.A., de Barros Miranda-Filho D., Montarroyos U.R., de Melo A.P.L., Valongueiro S., de Albuquerque M.d.F.P.M., Souza W.V., Braga C., et al. Association between Zika Virus Infection and Microcephaly in Brazil, January to May 2016: Preliminary Report of a Case-Control Study. Lancet Infect. Dis. 2016;16:1356–1363. doi: 10.1016/S1473-3099(16)30318-8. [DOI] [PubMed] [Google Scholar]
- 226.Paniz-Mondolfi A.E., Blohm G.M., Hernandez-Perez M., Larrazabal A., Moya D., Marquez M., Talamo A., Carrillo A., Rothe de Arocha J., Lednicky J., et al. Cutaneous Features of Zika Virus Infection: A Clinicopathological Overview. Clin. Exp. Dermatol. 2019;44:13–19. doi: 10.1111/ced.13793. [DOI] [PubMed] [Google Scholar]
- 227.Faye O., Freire C.C.M., Iamarino A., Faye O., de Oliveira J.V.C., Diallo M., Zanotto P.M.A., Sall A.A. Molecular Evolution of Zika Virus during Its Emergence in the 20th Century. PLoS Negl. Trop. Dis. 2014;8:e2636. doi: 10.1371/journal.pntd.0002636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Pierson T.C., Diamond M.S. The Continued Threat of Emerging Flaviviruses. Nat. Microbiol. 2020;5:796–812. doi: 10.1038/s41564-020-0714-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Valiant W.G., Huang Y.-J.S., Vanlandingham D.L., Higgs S., Lewis M.G., Mattapallil J.J. Zika Convalescent Macaques Display Delayed Induction of Anamnestic Cross-Neutralizing Antibody Responses after Dengue Infection. Emerg. Microbes Infect. 2018;7:1–11. doi: 10.1038/s41426-018-0132-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Chambers T., Monath T. The Flaviviruses: Structure, Replication and Evolution. 1st ed. Academic Press; Cambridge, MA, USA: 2003. [Google Scholar]
- 231.Wang L., Wang R., Wang L., Ben H., Yu L., Gao F., Shi X., Yin C., Zhang F., Xiang Y., et al. Structural Basis for Neutralization and Protection by a Zika Virus-Specific Human Antibody. Cell Rep. 2019;26:3360–3368. doi: 10.1016/j.celrep.2019.02.062. [DOI] [PubMed] [Google Scholar]
- 232.Yang M., Sun H., Lai H., Hurtado J., Chen Q. Plant-Produced Zika Virus Envelope Protein Elicits Neutralizing Immune Responses That Correlate with Protective Immunity against Zika Virus in Mice. Plant Biotechnol. J. 2018;16:572–580. doi: 10.1111/pbi.12796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Yang M., Lai H., Sun H., Chen Q. Virus-like Particles That Display Zika Virus Envelope Protein Domain III Induce Potent Neutralizing Immune Responses in Mice. Sci. Rep. 2017;7:7679. doi: 10.1038/s41598-017-08247-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Diamos A.G., Pardhe M.D., Sun H., Hunter J.G.L., Kilbourne J., Chen Q., Mason H.S. A Highly Expressing, Soluble, and Stable Plant-Made IgG Fusion Vaccine Strategy Enhances Antigen Immunogenicity in Mice Without Adjuvant. Front. Immunol. 2020;11:3140. doi: 10.3389/fimmu.2020.576012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Diamos A.G., Pardhe M.D., Sun H., Hunter J.G.L., Mor T., Meador L., Kilbourne J., Chen Q., Mason H.S. Codelivery of Improved Immune Complex and Virus-like Particle Vaccines Containing Zika Virus Envelope Domain III Synergistically Enhances Immunogenicity. Vaccine. 2020;38:3455–3463. doi: 10.1016/j.vaccine.2020.02.089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Jentes E.S., Poumerol G., Gershman M.D., Hill D.R., Lemarchand J., Lewis R.F., Staples J.E., Tomori O., Wilder-Smith A., Monath T.P. The Revised Global Yellow Fever Risk Map and Recommendations for Vaccination, 2010: Consensus of the Informal WHO Working Group on Geographic Risk for Yellow Fever. Lancet Infect. Dis. 2011;11:622–632. doi: 10.1016/S1473-3099(11)70147-5. [DOI] [PubMed] [Google Scholar]
- 237.WHO Yellow Fever. [(accessed on 20 November 2021)]. Available online: https://www.who.int/news-room/fact-sheets/detail/yellow-fever.
- 238.Jones C.T., Patkar C.G., Kuhn R.J. Construction and Applications of Yellow Fever Virus Replicons. Virology. 2005;331:247–259. doi: 10.1016/j.virol.2004.10.034. [DOI] [PubMed] [Google Scholar]
- 239.Tottey S., Shoji Y., Jones R.M., Chichester J.A., Green B.J., Musiychuk K., Si H., Manceva S.D., Rhee A., Shamloul M., et al. Plant-Produced Subunit Vaccine Candidates against Yellow Fever Induce Virus Neutralizing Antibodies and Confer Protection against Viral Challenge in Animal Models. Am. J. Trop. Med. Hyg. 2017;98:420–431. doi: 10.4269/ajtmh.16-0293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240.Hansen C.A., Barrett A.D.T. The Present and Future of Yellow Fever Vaccines. Pharmaceuticals. 2021;14:891. doi: 10.3390/ph14090891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.van den Hurk A.F., Ritchie S.A., Mackenzie J.S. Ecology and Geographical Expansion of Japanese Encephalitis Virus. Annu. Rev. Entomol. 2009;54:17–35. doi: 10.1146/annurev.ento.54.110807.090510. [DOI] [PubMed] [Google Scholar]
- 242.Sumiyoshi H., Mori C., Fuke I., Morita K., Kuhara S., Kondou J., Kikuchi Y., Nagamatu H., Igarashi A. Complete Nucleotide Sequence of the Japanese Encephalitis Virus Genome RNA. Virology. 1987;161:497–510. doi: 10.1016/0042-6822(87)90144-9. [DOI] [PubMed] [Google Scholar]
- 243.Xu X.-G., Wang Z.-S., Zhang Q., Li Z.-C., Zhao H.-N., Li W., Tong D.-W., Liu H.-J. Baculovirus Surface Display of E Envelope Glycoprotein of Japanese Encephalitis Virus and Its Immunogenicity of the Displayed Proteins in Mouse and Swine Models. Vaccine. 2011;29:636–643. doi: 10.1016/j.vaccine.2010.11.045. [DOI] [PubMed] [Google Scholar]
- 244.Wang Y., Deng H., Zhang X., Xiao H., Jiang Y., Song Y., Fang L., Xiao S., Zhen Y., Chen H. Generation and Immunogenicity of Japanese Encephalitis Virus Envelope Protein Expressed in Transgenic Rice. Biochem. Biophys. Res. Commun. 2009;380:292–297. doi: 10.1016/j.bbrc.2009.01.061. [DOI] [PubMed] [Google Scholar]
- 245.Chen T.-H., Hu C.-C., Liao J.-T., Lee Y.-L., Huang Y.-W., Lin N.-S., Lin Y.-L., Hsu Y.-H. Production of Japanese Encephalitis Virus Antigens in Plants Using Bamboo Mosaic Virus-Based Vector. Front. Microbiol. 2017;8:788. doi: 10.3389/fmicb.2017.00788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Valarcher J.F., Hägglund S., Juremalm M., Blomqvist G., Renström L., Zohari S., Leijon M., Chirico J. Tick-Borne Encephalitis. Rev. Sci. Tech. Int. Off. Epizoot. 2015;34:453–466. doi: 10.20506/rst.34.2.2371. [DOI] [PubMed] [Google Scholar]
- 247.Lindquist L., Vapalahti O. Tick-Borne Encephalitis. Lancet. 2008;371:1861–1871. doi: 10.1016/S0140-6736(08)60800-4. [DOI] [PubMed] [Google Scholar]
- 248.Mansfield K.L., Johnson N., Phipps L.P., Stephenson J.R., Fooks A.R., Solomon T.Y. 2009 Tick-Borne Encephalitis Virus—A Review of an Emerging Zoonosis. J. Gen. Virol. 2009;90:1781–1794. doi: 10.1099/vir.0.011437-0. [DOI] [PubMed] [Google Scholar]
- 249.Jääskeläinen A., Han X., Niedrig M., Vaheri A., Vapalahti O. Diagnosis of Tick-Borne Encephalitis by a μ-Capture Immunoglobulin M-Enzyme Immunoassay Based on Secreted Recombinant Antigen Produced in Insect Cells. J. Clin. Microbiol. 2003;41:4336–4342. doi: 10.1128/JCM.41.9.4336-4342.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Allison S.L., Stadler K., Mandl C.W., Kunz C., Heinz F.X. Synthesis and Secretion of Recombinant Tick-Borne Encephalitis Virus Protein E in Soluble and Particulate Form. J. Virol. 1995;69:5816–5820. doi: 10.1128/jvi.69.9.5816-5820.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Heinz F.X., Allison S.L., Stiasny K., Schalich J., Holzmann H., Mandl C.W., Kunz C. Recombinant and Virion-Derived Soluble and Particulate Immunogens for Vaccination against Tick-Borne Encephalitis. Vaccine. 1995;13:1636–1642. doi: 10.1016/0264-410X(95)00133-L. [DOI] [PubMed] [Google Scholar]
- 252.Andersson S., Asghar N., Davidsson Å., Gunaltay S., Johansson M., Melik W., Persson A., Särndahl E., Tran H. Molecular Flavivirus Research—Örebro University. [(accessed on 20 November 2021)]. Available online: https://www.oru.se/english/research/research-teams/rt/?rdb=g225.
- 253.Barré-Sinoussi F., Chermann J.C., Rey F., Nugeyre M.T., Chamaret S., Gruest J., Dauguet C., Axler-Blin C., Vézinet-Brun F., Rouzioux C., et al. Isolation of a T-Lymphotropic Retrovirus from a Patient at Risk for Acquired Immune Deficiency Syndrome (AIDS) Science. 1983;220:868–871. doi: 10.1126/science.6189183. [DOI] [PubMed] [Google Scholar]
- 254.Gallo R.C., Salahuddin S.Z., Popovic M., Shearer G.M., Kaplan M., Haynes B.F., Palker T.J., Redfield R., Oleske J., Safai B. Frequent Detection and Isolation of Cytopathic Retroviruses (HTLV-III) from Patients with AIDS and at Risk for AIDS. Science. 1984;224:500–503. doi: 10.1126/science.6200936. [DOI] [PubMed] [Google Scholar]
- 255.Huet T., Cheynier R., Meyerhans A., Roelants G., Wain-Hobson S. Genetic Organization of a Chimpanzee Lentivirus Related to HIV-1. Nature. 1990;345:356–359. doi: 10.1038/345356a0. [DOI] [PubMed] [Google Scholar]
- 256.Hirsch V.M., Olmsted R.A., Murphey-Corb M., Purcell R.H., Johnson P.R. An African Primate Lentivirus SIVsmclosely Related to HIV-2. Nature. 1989;339:389–392. doi: 10.1038/339389a0. [DOI] [PubMed] [Google Scholar]
- 257.Sharp P.M., Hahn B.H. Origins of HIV and the AIDS Pandemic. Cold Spring Harb. Perspect. Med. 2011;1:a006841. doi: 10.1101/cshperspect.a006841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.De Cock K.M., Adjorlolo G., Ekpini E., Sibailly T., Kouadio J., Maran M., Brattegaard K., Vetter K.M., Doorly R., Gayle H.D. Epidemiology and Transmission of HIV-2: Why There Is No HIV-2 Pandemic. JAMA. 1993;270:2083–2086. doi: 10.1001/jama.1993.03510170073033. [DOI] [PubMed] [Google Scholar]
- 259.van Schooten J., van Gils M.J. HIV-1 Immunogens and Strategies to Drive Antibody Responses towards Neutralization Breadth. Retrovirology. 2018;15:74. doi: 10.1186/s12977-018-0457-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Tremouillaux-Guiller J., Moustafa K., Hefferon K., Gaobotse G., Makhzoum A. Plant-Made HIV Vaccines and Potential Candidates. Curr. Opin. Biotechnol. 2020;61:209–216. doi: 10.1016/j.copbio.2020.01.004. [DOI] [PubMed] [Google Scholar]
- 261.Scotti N., Buonaguro L., Tornesello M.L., Cardi T., Buonaguro F.M. Plant-Based Anti-HIV-1 Strategies: Vaccine Molecules and Antiviral Approaches. Expert Rev. Vaccines. 2010;9:925–936. doi: 10.1586/erv.10.79. [DOI] [PubMed] [Google Scholar]
- 262.Zolla-Pazner S. Identifying Epitopes of HIV-1 That Induce Protective Antibodies. Nat. Rev. Immunol. 2004;4:199–210. doi: 10.1038/nri1307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Govea-Alonso D.O., Gómez-Cardona E.E., Rubio-Infante N., García-Hernández A.L., Varona-Santos J.T., Salgado-Bustamante M., Korban S.S., Moreno-Fierros L., Rosales-Mendoza S. Production of an Antigenic C4(V3)6 Multiepitopic HIV Protein in Bacterial and Plant Systems. Plant Cell Tissue Organ Cult. 2013;113:73–79. doi: 10.1007/s11240-012-0252-4. [DOI] [Google Scholar]
- 264.Lindh I., Kalbina I., Thulin S., Scherbak N., Sävenstrand H., Bråve A., Hinkula J., Strid Å., Andersson S. Feeding of Mice with Arabidopsis Thaliana Expressing the HIV-1 Subtype C P24 Antigen Gives Rise to Systemic Immune Responses. APMIS. 2008;116:985–994. doi: 10.1111/j.1600-0463.2008.00900.x. [DOI] [PubMed] [Google Scholar]
- 265.Gonzalez-Rabade N., McGowan E.G., Zhou F., McCabe M.S., Bock R., Dix P.J., Gray J.C., Ma J.K.-C. Immunogenicity of Chloroplast-Derived HIV-1 P24 and a P24-Nef Fusion Protein Following Subcutaneous and Oral Administration in Mice. Plant Biotechnol. J. 2011;9:629–638. doi: 10.1111/j.1467-7652.2011.00609.x. [DOI] [PubMed] [Google Scholar]
- 266.Pérez-Filgueira D.M., Brayfield B.P., Phiri S., Borca M.V., Wood C., Morris T.J. Preserved Antigenicity of HIV-1 P24 Produced and Purified in High Yields from Plants Inoculated with a Tobacco Mosaic Virus (TMV)-Derived Vector. J. Virol. Methods. 2004;121:201–208. doi: 10.1016/j.jviromet.2004.06.022. [DOI] [PubMed] [Google Scholar]
- 267.McLain L., Durrani Z., Wisniewski L.A., Porta C., Lomonossoff G.P., Dimmock N.J. Stimulation of Neutralizing Antibodies to Human Immunodeficiency Virus Type 1 in Three Strains of Mice Immunized with a 22 Amino Acid Peptide of Gp41 Expressed on the Surface of a Plant Virus. Vaccine. 1996;14:799–810. doi: 10.1016/0264-410X(95)00229-T. [DOI] [PubMed] [Google Scholar]
- 268.Guetard D., Greco R., Cervantes Gonzalez M., Celli S., Kostrzak A., Langlade-Demoyen P., Sala F., Wain-Hobson S., Sala M. Immunogenicity and Tolerance Following HIV-1/HBV Plant-Based Oral Vaccine Administration. Vaccine. 2008;26:4477–4485. doi: 10.1016/j.vaccine.2008.06.059. [DOI] [PubMed] [Google Scholar]
- 269.Greco R., Michel M., Guetard D., Cervantes-Gonzalez M., Pelucchi N., Wain-Hobson S., Sala F., Sala M. Production of Recombinant HIV-1/HBV Virus-like Particles in Nicotiana Tabacum and Arabidopsis Thaliana Plants for a Bivalent Plant-Based Vaccine. Vaccine. 2007;25:8228–8240. doi: 10.1016/j.vaccine.2007.09.061. [DOI] [PubMed] [Google Scholar]
- 270.Margolin E., Oh Y.J., Verbeek M., Naude J., Ponndorf D., Meshcheriakova Y.A., Peyret H., van Diepen M.T., Chapman R., Meyers A.E., et al. Co-expression of Human Calreticulin Significantly Improves the Production of HIV Gp140 and Other Viral Glycoproteins in Plants. Plant Biotechnol. J. 2020;18:2109–2117. doi: 10.1111/pbi.13369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Margolin E., Chapman R., Meyers A.E., van Diepen M.T., Ximba P., Hermanus T., Crowther C., Weber B., Morris L., Williamson A.-L., et al. Production and Immunogenicity of Soluble Plant-Produced HIV-1 Subtype C Envelope Gp140 Immunogens. Front. Plant Sci. 2019;10:1378. doi: 10.3389/fpls.2019.01378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Porta C., Spall V.E., Loveland J., Johnson J.E., Barker P.J., Lomonossoff G.P. Development of Cowpea Mosaic Virus as a High-Yielding System for the Presentation of Foreign Peptides. Virology. 1994;202:949–955. doi: 10.1006/viro.1994.1417. [DOI] [PubMed] [Google Scholar]
- 273.McLAIN L., Porta C., Lomonossoff G.P., Durrani Z., Dimmock N.J. Human Immunodeficiency Virus Type 1-Neutralizing Antibodies Raised to a Glycoprotein 41 Peptide Expressed on the Surface of a Plant Virus. AIDS Res. Hum. Retrovir. 1995;11:327–334. doi: 10.1089/aid.1995.11.327. [DOI] [PubMed] [Google Scholar]
- 274.Matoba N., Magérus A., Geyer B.C., Zhang Y., Muralidharan M., Alfsen A., Arntzen C.J., Bomsel M., Mor T.S. A Mucosally Targeted Subunit Vaccine Candidate Eliciting HIV-1 Transcytosis-Blocking Abs. Proc. Natl. Acad. Sci. USA. 2004;101:13584–13589. doi: 10.1073/pnas.0405297101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275.Shchelkunov S.N., Salyaev R.K., Pozdnyakov S.G., Rekoslavskaya N.I., Nesterov A.E., Ryzhova T.S., Sumtsova V.M., Pakova N.V., Mishutina U.O., Kopytina T.V., et al. Immunogenicity of a Novel, Bivalent, Plant-Based Oral Vaccine against Hepatitis B and Human Immunodeficiency Viruses. Biotechnol. Lett. 2006;28:959–967. doi: 10.1007/s10529-006-9028-4. [DOI] [PubMed] [Google Scholar]
- 276.Ramírez Y.J.P., Tasciotti E., Gutierrez-Ortega A., Torres A.J.D., Flores M.T.O., Giacca M., Lim M.Á.G. Fruit-Specific Expression of the Human Immunodeficiency Virus Type 1 Tat Gene in Tomato Plants and Its Immunogenic Potential in Mice. Clin. Vaccine Immunol. 2007;14:685–692. doi: 10.1128/CVI.00028-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 277.Meyers A., Chakauya E., Shephard E., Tanzer F.L., Maclean J., Lynch A., Williamson A.-L., Rybicki E.P. Expression of HIV-1 Antigens in Plants as Potential Subunit Vaccines. BMC Biotechnol. 2008;8:53. doi: 10.1186/1472-6750-8-53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Matoba N., Kajiura H., Cherni I., Doran J.D., Bomsel M., Fujiyama K., Mor T.S. Biochemical and Immunological Characterization of the Plant-Derived Candidate Human Immunodeficiency Virus Type 1 Mucosal Vaccine CTB–MPR649–684. Plant Biotechnol. J. 2009;7:129–145. doi: 10.1111/j.1467-7652.2008.00381.x. [DOI] [PubMed] [Google Scholar]
- 279.Lindh I., Bråve A., Hallengärd D., Hadad R., Kalbina I., Strid Å., Andersson S. Oral Delivery of Plant-Derived HIV-1 P24 Antigen in Low Doses Shows a Superior Priming Effect in Mice Compared to High Doses. Vaccine. 2014;32:2288–2293. doi: 10.1016/j.vaccine.2014.02.073. [DOI] [PubMed] [Google Scholar]
- 280.Rubio-Infante N., Govea-Alonso D.O., Romero-Maldonado A., García-Hernández A.L., Ilhuicatzi-Alvarado D., Salazar-González J.A., Korban S.S., Rosales-Mendoza S., Moreno-Fierros L. A Plant-Derived Multi-HIV Antigen Induces Broad Immune Responses in Orally Immunized Mice. Mol. Biotechnol. 2015;57:662–674. doi: 10.1007/s12033-015-9856-3. [DOI] [PubMed] [Google Scholar]
- 281.Rosales-Mendoza S., Rubio-Infante N., Monreal-Escalante E., Govea-Alonso D.O., García-Hernández A.L., Salazar-González J.A., González-Ortega O., Paz-Maldonado L.M.T., Moreno-Fierros L. Chloroplast Expression of an HIV Envelop-Derived Multiepitope Protein: Towards a Multivalent Plant-Based Vaccine. Plant Cell Tissue Organ Cult. 2014;116:111–123. doi: 10.1007/s11240-013-0387-y. [DOI] [Google Scholar]
- 282.Orellana-Escobedo L., Rosales-Mendoza S., Romero-Maldonado A., Parsons J., Decker E.L., Monreal-Escalante E., Moreno-Fierros L., Reski R. An Env-Derived Multi-Epitope HIV Chimeric Protein Produced in the Moss Physcomitrella Patens Is Immunogenic in Mice. Plant Cell Rep. 2015;34:425–433. doi: 10.1007/s00299-014-1720-6. [DOI] [PubMed] [Google Scholar]
- 283.Kessans S.A., Linhart M.D., Matoba N., Mor T. Biological and Biochemical Characterization of HIV-1 Gag/Dgp41 Virus-like Particles Expressed in Nicotiana Benthamiana. Plant Biotechnol. J. 2013;11:681–690. doi: 10.1111/pbi.12058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284.Kessans S.A., Linhart M.D., Meador L.R., Kilbourne J., Hogue B.G., Fromme P., Matoba N., Mor T.S. Immunological Characterization of Plant-Based HIV-1 Gag/Dgp41 Virus-Like Particles. PLoS ONE. 2016;11:e0151842. doi: 10.1371/journal.pone.0151842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Bennett J.E., Dolin R., Blaser M.J. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. Elsevier; Amsterdam, The Netherlands: 2019. [Google Scholar]
- 286.Fooks A.R., Cliquet F., Finke S., Freuling C., Hemachudha T., Mani R.S., Müller T., Nadin-Davis S., Picard-Meyer E., Wilde H., et al. Rabies. Nat. Rev. Dis. Primer. 2017;3:1–19. doi: 10.1038/nrdp.2017.91. [DOI] [PubMed] [Google Scholar]
- 287.Julia Paoli Is Rabies Really 100% Fatal? Viruses101. Learn Science at Scitable. [(accessed on 20 November 2021)]. Available online: https://www.nature.com/scitable/blog/viruses101/is_rabies_really_100_fatal/
- 288.Albertini A.A.V., Ruigrok R.W.H., Blondel D. Rabies Virus Transcription and Replication. Adv. Virus Res. 2011;79:1–22. doi: 10.1016/B978-0-12-387040-7.00001-9. [DOI] [PubMed] [Google Scholar]
- 289.Takeyama N., Kiyono H., Yuki Y. Plant-Based Vaccines for Animals and Humans: Recent Advances in Technology and Clinical Trials. Ther. Adv. Vaccines. 2015;3:139–154. doi: 10.1177/2051013615613272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 290.Loza-Rubio E., Rojas-Anaya E. Edible Rabies Vaccines. In: Howard J.A., Hood E.E., editors. Commercial Plant-Produced Recombinant Protein Products: Case Studies. Springer; Berlin/Heidelberg, Germany: 2014. pp. 153–177. Biotechnology in Agriculture and Forestry. [Google Scholar]
- 291.Ashraf S., Singh P.K., Yadav D.K., Shahnawaz M., Mishra S., Sawant S.V., Tuli R. High Level Expression of Surface Glycoprotein of Rabies Virus in Tobacco Leaves and Its Immunoprotective Activity in Mice. J. Biotechnol. 2005;119:1–14. doi: 10.1016/j.jbiotec.2005.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 292.Singh A., Srivastava S., Chouksey A., Panwar B.S., Verma P.C., Roy S., Singh P.K., Saxena G., Tuli R. Expression of Rabies Glycoprotein and Ricin Toxin B Chain (RGP–RTB) Fusion Protein in Tomato Hairy Roots: A Step Towards Oral Vaccination for Rabies. Mol. Biotechnol. 2015;57:359–370. doi: 10.1007/s12033-014-9829-y. [DOI] [PubMed] [Google Scholar]
- 293.Perea Arango I., Loza Rubio E., Rojas Anaya E., Olivera Flores T., de la Vara L.G., Gómez Lim M.A. Expression of the Rabies Virus Nucleoprotein in Plants at High-Levels and Evaluation of Immune Responses in Mice. Plant Cell Rep. 2008;27:677–685. doi: 10.1007/s00299-007-0324-9. [DOI] [PubMed] [Google Scholar]
- 294.McGarvey P.B., Hammond J., Dienelt M.M., Hooper D.C., Fu Z.F., Dietzschold B., Koprowski H., Michaels F.H. Expression of the Rabies Virus Glycoprotein in Transgenic Tomatoes. Biotechnology. 1995;13:1484–1487. doi: 10.1038/nbt1295-1484. [DOI] [PubMed] [Google Scholar]
- 295.Rojas-Anaya E., Loza-Rubio E., Olivera-Flores M.T., Gomez-Lim M. Expression of Rabies Virus G Protein in Carrots (Daucus Carota) Transgenic Res. 2009;18:911. doi: 10.1007/s11248-009-9278-8. [DOI] [PubMed] [Google Scholar]
- 296.Modelska A., Dietzschold B., Sleysh N., Fu Z.F., Steplewski K., Hooper D.C., Koprowski H., Yusibov V. Immunization against Rabies with Plant-Derived Antigen. Proc. Natl. Acad. Sci. USA. 1998;95:2481–2485. doi: 10.1073/pnas.95.5.2481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 297.Yusibov V., Hooper D.C., Spitsin S.V., Fleysh N., Kean R.B., Mikheeva T., Deka D., Karasev A., Cox S., Randall J., et al. Expression in Plants and Immunogenicity of Plant Virus-Based Experimental Rabies Vaccine. Vaccine. 2002;20:3155–3164. doi: 10.1016/S0264-410X(02)00260-8. [DOI] [PubMed] [Google Scholar]
- 298.Loza-Rubio E., Rojas E., Gómez L., Olivera M.T.J., Gómez-Lim M.A. Development of an Edible Rabies Vaccine in Maize Using the Vnukovo Strain. Dev. Biol. 2008;131:477–482. [PubMed] [Google Scholar]
- 299.Loza-Rubio E., Rojas-Anaya E., López J., Olivera-Flores M.T., Gómez-Lim M., Tapia-Pérez G. Induction of a Protective Immune Response to Rabies Virus in Sheep after Oral Immunization with Transgenic Maize, Expressing the Rabies Virus Glycoprotein. Vaccine. 2012;30:5551–5556. doi: 10.1016/j.vaccine.2012.06.039. [DOI] [PubMed] [Google Scholar]
- 300.Wang B., Meng X.-J. Structural and Molecular Biology of Hepatitis E Virus. Comput. Struct. Biotechnol. J. 2021;19:1907–1916. doi: 10.1016/j.csbj.2021.03.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 301.Yugo D.M., Meng X.-J. Hepatitis E Virus: Foodborne, Waterborne and Zoonotic Transmission. Int. J. Environ. Res. Public Health. 2013;10:4507–4533. doi: 10.3390/ijerph10104507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Forni D., Cagliani R., Clerici M., Sironi M. Origin and Dispersal of Hepatitis E Virus. Emerg. Microbes Infect. 2018;7:11. doi: 10.1038/s41426-017-0009-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Takova K., Koynarski T., Minkov I., Ivanova Z., Toneva V., Zahmanova G. Increasing Hepatitis E Virus Seroprevalence in Domestic Pigs and Wild Boar in Bulgaria. Animals. 2020;10:1521. doi: 10.3390/ani10091521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 304.Tsachev I., Baymakova M., Pepovich R., Palova N., Marutsov P., Gospodinova K., Kundurzhiev T., Ciccozzi M. High Seroprevalence of Hepatitis E Virus Infection Among East Balkan Swine (Sus Scrofa) in Bulgaria: Preliminary Results. Pathogens. 2020;9:911. doi: 10.3390/pathogens9110911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Tsachev I., Baymakova M., Marutsov P., Gospodinova K., Kundurzhiev T., Petrov V., Pepovich R. Seroprevalence of Hepatitis E Virus Infection Among Wild Boars in Western Bulgaria. Vector Borne Zoonotic Dis. 2021;21:441–445. doi: 10.1089/vbz.2020.2756. [DOI] [PubMed] [Google Scholar]
- 306.Ahmad I., Holla R.P., Jameel S. Molecular Virology of Hepatitis E Virus. Virus Res. 2011;161:47–58. doi: 10.1016/j.virusres.2011.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 307.Ma Y., Lin S.-Q., Gao Y., Li M., Luo W.-X., Zhang J., Xia N.-S. Expression of ORF2 Partial Gene of Hepatitis E Virus in Tomatoes and Immunoactivity of Expression Products. World J. Gastroenterol. 2003;9:2211–2215. doi: 10.3748/wjg.v9.i10.2211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Maloney B.J., Takeda N., Suzaki Y., Ami Y., Li T.C., Miyamura T., Arntzen C.J., Mason H.S. Challenges in Creating a Vaccine to Prevent Hepatitis E. Vaccine. 2005;23:1870–1874. doi: 10.1016/j.vaccine.2004.11.020. [DOI] [PubMed] [Google Scholar]
- 309.Zhou Y.-X., Lee M.Y.-T., Ng J.M.-H., Chye M.-L., Yip W.-K., Zee S.-Y., Lam E. A Truncated Hepatitis E Virus ORF2 Protein Expressed in Tobacco Plastids Is Immunogenic in Mice. World J. Gastroenterol. 2006;12:306–312. doi: 10.3748/wjg.v12.i2.306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 310.Mazalovska M., Varadinov N., Koynarski T., Minkov I., Teoharov P., Lomonossoff G.P., Zahmanova G. Detection of Serum Antibodies to Hepatitis E Virus Based on HEV Genotype 3 ORF2 Capsid Protein Expressed in Nicotiana Benthamiana. Ann. Lab. Med. 2017;37:313–319. doi: 10.3343/alm.2017.37.4.313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 311.Mardanova E., Takova K., Toneva V., Zahmanova G., Tsybalova L., Ravin N. A Plant-Based Transient Expression System for the Rapid Production of Highly Immunogenic Hepatitis E Virus-like Particles. Biotechnol. Lett. 2020;42:2441–2446. doi: 10.1007/s10529-020-02995-x. [DOI] [PubMed] [Google Scholar]
- 312.Takova K., Koynarski T., Minkov G., Toneva V., Mardanova E., Ravin N., Lukov G.L., Zahmanova G. Development and Optimization of an Enzyme Immunoassay to Detect Serum Antibodies against the Hepatitis E Virus in Pigs, Using Plant-Derived ORF2 Recombinant Protein. Vaccines. 2021;9:991. doi: 10.3390/vaccines9090991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 313.Gogoi P., Ganar K., Kumar S. Avian Paramyxovirus: A Brief Review. Transbound. Emerg. Dis. 2017;64:53–67. doi: 10.1111/tbed.12355. [DOI] [PubMed] [Google Scholar]
- 314.Kumar S., Nayak B., Collins P.L., Samal S.K. Evaluation of the Newcastle Disease Virus F and HN Proteins in Protective Immunity by Using a Recombinant Avian Paramyxovirus Type 3 Vector in Chickens. J. Virol. 2011;85:6521–6534. doi: 10.1128/JVI.00367-11. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 315.Cox R.M., Plemper R.K. Structure and Organization of Paramyxovirus Particles. Curr. Opin. Virol. 2017;24:105–114. doi: 10.1016/j.coviro.2017.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 316.Brown V.R., Bevins S.N. A Review of Virulent Newcastle Disease Viruses in the United States and the Role of Wild Birds in Viral Persistence and Spread. Vet. Res. 2017;48:68. doi: 10.1186/s13567-017-0475-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 317.Fox J.G., Otto G., Colby L.A. Chapter 28—Selected Zoonoses. In: Fox J.G., Anderson L.C., Otto G.M., Pritchett-Corning K.R., Whary M.T., editors. Laboratory Animal Medicine. 3rd ed. American College of Laboratory Animal Medicine; Academic Press; Boston, MA, USA: 2015. pp. 1313–1370. [Google Scholar]
- 318.Alexander D.J. Gordon Memorial Lecture. Newcastle Disease. Br. Poult. Sci. 2001;42:5–22. doi: 10.1080/713655022. [DOI] [PubMed] [Google Scholar]
- 319.Berinstein A., Vazquez-Rovere C., Asurmendi S., Gómez E., Zanetti F., Zabal O., Tozzini A., Conte Grand D., Taboga O., Calamante G., et al. Mucosal and Systemic Immunization Elicited by Newcastle Disease Virus (NDV) Transgenic Plants as Antigens. Vaccine. 2005;23:5583–5589. doi: 10.1016/j.vaccine.2005.06.033. [DOI] [PubMed] [Google Scholar]
- 320.Yang Z.-Q., Liu Q.-Q., Pan Z.-M., Yu H.-X., Jiao X.-A. Expression of the Fusion Glycoprotein of Newcastle Disease Virus in Transgenic Rice and Its Immunogenicity in Mice. Vaccine. 2007;25:591–598. doi: 10.1016/j.vaccine.2006.08.016. [DOI] [PubMed] [Google Scholar]
- 321.Guerrero-Andrade O., Loza-Rubio E., Olivera-Flores T., Fehérvári-Bone T., Gómez-Lim M.A. Expression of the Newcastle Disease Virus Fusion Protein in Transgenic Maize and Immunological Studies. Transgenic Res. 2006;15:455–463. doi: 10.1007/s11248-006-0017-0. [DOI] [PubMed] [Google Scholar]
- 322.Shahid N., Samiullah T.R., Shakoor S., Latif A., Yasmeen A., Azam S., Shahid A.A., Husnain T., Rao A.Q. Early Stage Development of a Newcastle Disease Vaccine Candidate in Corn. Front. Vet. Sci. 2020;7:499. doi: 10.3389/fvets.2020.00499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 323.Motamedi M.J., Ebrahimi M.M., Shahsavandi S., Amani J., Kazemi R., Jafari M., Salmanian A.-H. The Immunogenicity of a Novel Chimeric Hemagglutinin-Neuraminidase-Fusion Antigen from Newcastle Disease Virus by Oral Delivery of Transgenic Canola Seeds to Chickens. Mol. Biotechnol. 2020;62:344–354. doi: 10.1007/s12033-020-00254-y. [DOI] [PubMed] [Google Scholar]
- 324.Natilla A., Hammond R.W., Nemchinov L.G. Epitope Presentation System Based on Cucumber Mosaic Virus Coat Protein Expressed from a Potato Virus X-Based Vector. Arch. Virol. 2006;151:1373–1386. doi: 10.1007/s00705-005-0711-x. [DOI] [PubMed] [Google Scholar]
- 325.Natilla A., Nemchinov L.G. Improvement of PVX/CMV CP Expression Tool for Display of Short Foreign Antigens. Protein Expr. Purif. 2008;59:117–121. doi: 10.1016/j.pep.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 326.Gómez E., Zoth S.C., Asurmendi S., Vázquez Rovere C., Berinstein A. Expression of Hemagglutinin-Neuraminidase Glycoprotein of Newcastle Disease Virus in Agroinfiltrated Nicotiana Benthamiana Plants. J. Biotechnol. 2009;144:337–340. doi: 10.1016/j.jbiotec.2009.09.015. [DOI] [PubMed] [Google Scholar]
- 327.Hahn B.-S., Jeon I.-S., Jung Y.-J., Kim J.-B., Park J.-S., Ha S.-H., Kim K.-H., Kim H.-M., Yang J.-S., Kim Y.-H. Expression of Hemagglutinin-Neuraminidase Protein of Newcastle Disease Virus in Transgenic Tobacco. Plant Biotechnol. Rep. 2007;1:85–92. doi: 10.1007/s11816-007-0012-9. [DOI] [Google Scholar]
- 328.Vermij P. News In Brief. Nat. Biotechnol. 2006;24:233–234. doi: 10.1038/nbt0306-233. [DOI] [Google Scholar]
- 329.Eaton B.T., Broder C.C., Middleton D., Wang L.-F. Hendra and Nipah Viruses: Different and Dangerous. Nat. Rev. Microbiol. 2006;4:23–35. doi: 10.1038/nrmicro1323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 330.Clayton B.A., Wang L.F., Marsh G.A. Henipaviruses: An Updated Review Focusing on the Pteropid Reservoir and Features of Transmission. Zoonoses Public Health. 2013;60:69–83. doi: 10.1111/j.1863-2378.2012.01501.x. [DOI] [PubMed] [Google Scholar]
- 331.Islam M.S., Sazzad H.M.S., Satter S.M., Sultana S., Hossain M.J., Hasan M., Rahman M., Campbell S., Cannon D.L., Ströher U., et al. Nipah Virus Transmission from Bats to Humans Associated with Drinking Traditional Liquor Made from Date Palm Sap, Bangladesh, 2011–2014. Emerg. Infect. Dis. J. 2016;22:664. doi: 10.3201/eid2204.151747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 332.Enchéry F., Horvat B. Understanding the Interaction between Henipaviruses and Their Natural Host, Fruit Bats: Paving the Way toward Control of Highly Lethal Infection in Humans. Int. Rev. Immunol. 2017;36:108–121. doi: 10.1080/08830185.2016.1255883. [DOI] [PubMed] [Google Scholar]
- 333.Amaya M., Broder C.C. Vaccines to Emerging Viruses: Nipah and Hendra. Annu. Rev. Virol. 2020;7:447–473. doi: 10.1146/annurev-virology-021920-113833. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.