Abstract
Post-acute sequelae of COVID (PASC), usually referred to as ‘Long COVID’ (a phenotype of COVID-19), is a relatively frequent consequence of SARS-CoV-2 infection, in which symptoms such as breathlessness, fatigue, ‘brain fog’, tissue damage, inflammation, and coagulopathies (dysfunctions of the blood coagulation system) persist long after the initial infection. It bears similarities to other post-viral syndromes, and to myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS). Many regulatory health bodies still do not recognize this syndrome as a separate disease entity, and refer to it under the broad terminology of ‘COVID’, although its demographics are quite different from those of acute COVID-19. A few years ago, we discovered that fibrinogen in blood can clot into an anomalous ‘amyloid’ form of fibrin that (like other β-rich amyloids and prions) is relatively resistant to proteolysis (fibrinolysis). The result, as is strongly manifested in platelet-poor plasma (PPP) of individuals with Long COVID, is extensive fibrin amyloid microclots that can persist, can entrap other proteins, and that may lead to the production of various autoantibodies. These microclots are more-or-less easily measured in PPP with the stain thioflavin T and a simple fluorescence microscope. Although the symptoms of Long COVID are multifarious, we here argue that the ability of these fibrin amyloid microclots (fibrinaloids) to block up capillaries, and thus to limit the passage of red blood cells and hence O2 exchange, can actually underpin the majority of these symptoms. Consistent with this, in a preliminary report, it has been shown that suitable and closely monitored ‘triple’ anticoagulant therapy that leads to the removal of the microclots also removes the other symptoms. Fibrin amyloid microclots represent a novel and potentially important target for both the understanding and treatment of Long COVID and related disorders.
Keywords: amyloid, clotting, COVID
Introduction
In many cases, individuals infected with the SARS-CoV-2 virus and suffering from COVID-19 continue (or in some case begin) to display symptoms long after the acute phase. Depending on the demographics of the hosts and variant of SARS-CoV-2, these post-acute sequelae of COVID-19 (PASC [1]) may affect ∼30% of all infected individuals [2,3], are starting to be observed even in children [4,5], and are commonly known as long COVID [6–10]. Many regulatory health bodies still do not recognize this syndrome as a separate disease entity, and refers to it under the broad terminology of ‘COVID’. The symptoms of long COVID are multifarious [11,12] and include breathlessness, fatigue, chest pain, myalgia, cognitive dysfunction, innate immune responses coupled to inflammatory cytokine production, and a pro-coagulant state. Our focus is on the latter.
At present there is no established treatment for long COVID (e.g. [13,14], so from a systems point of view it is important to understand which symptoms are ‘primary’, and which are simply secondary effects of the primary symptoms themselves. This would allow treatment strategies to focus on the primary symptoms and their causes. We here make the case (with evidence) that much of the aetiology of long COVID can be attributed to the formation of aberrant amyloid fibrin microclots, triggered in particular by the SARS-Cov-2 spike protein, and that by inhibiting the transport of erythrocytes to capillaries, and hence O2 transfer, it is these amyloid microclots that are mainly responsible for the various long COVID symptoms observed. The microclots may also present novel antigens that lead to the production of autoantibodies, that can exacerbate symptoms further. This understanding of the role of such microclots may be expected to lead to an effective strategy for treating long COVID (and probably for other, related conditions such as myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS)).
Protein conformation, prions, and amyloid structures
Classically, it was assumed that proteins folded into their conformational states of lowest free energy, because unfolded proteins typically refolded into their original forms [15]. However, the discovery of prion proteins in particular showed that this was not always the case; the more common and native form of the prion protein PrPC could be converted — with no change in primary sequence — into a thermodynamically more stable, ‘rogue’ version referred to as PrPSc (Figure 1), and this transformation was itself catalyszd by PrPSc (e.g. [16,17]). These ‘rogue’ versions commonly contain a large number of ordered β-sheets in a cross-β architecture [17–23], and are referred to generally as amyloid forms. A great many proteins are now known to be able to adopt such amyloid forms, and over 50 have been implicated in a variety of diseases (‘amyloidoses’) [24–26]. These kinds of amyloid structures may be stained with fluorogenic dyes [27,28] such as congo red [29], thioflavin T [30–35], which is much more visible than congo red, or a variety of conjugated oligothiophenes [36] commercialized as AmytrackerTM dyes (e.g. [37,38]).
Figure 1. Many proteins can adopt more than more thermodynamically stable microstate with no change in primary structure (sequence), in which the more stable contains an ordered β-sheet ‘amyloid’ structure.
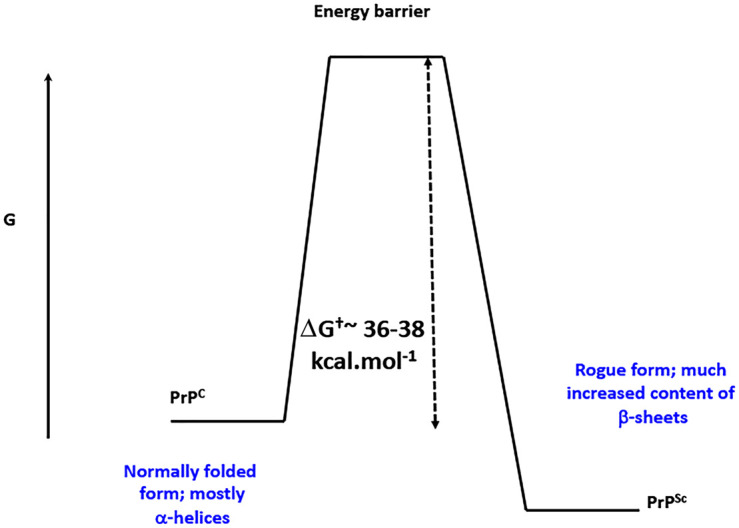
Normally, however, it is present in a less stable state that is kinetically more accessible during and following its synthesis. The more stable (labeled PrPSc) is separated from the initial state (PrPC) via a large energy barrier. This is true for amyloid proteins generally, and is illustrated here for classical prion proteins. Redrawn from a CC-BY publication at [26].
Amyloidogenic blood clotting
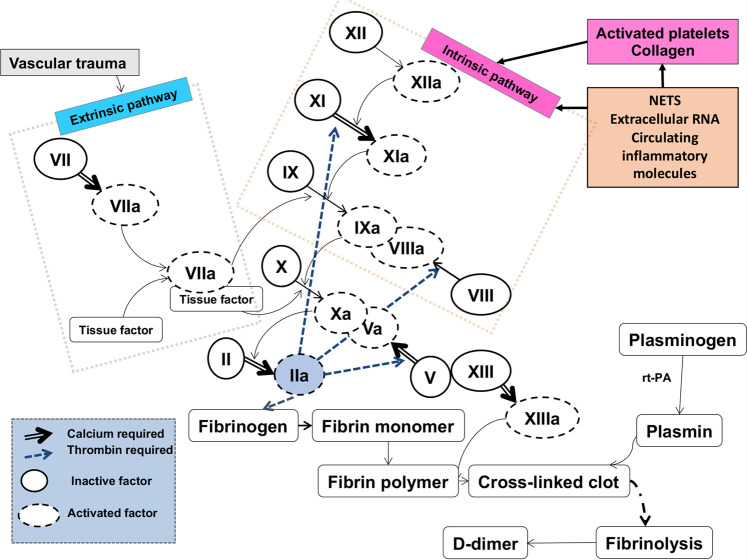
The normal blood clotting cascade is well established (e.g. [39–41] (Figure 2), with the terminal stages of both ‘intrinsic’ and ‘extrinsic’ pathways involving the self-organised polymerization of soluble fibrinogen to insoluble fibrin, catalysed by thrombin [42]. Fibrinogen, a cigar-shaped molecule of some 5 × 45 nm, is one of the most abundant proteins in plasma (commonly present at 1.5–3.5 g L−1 [41]).
Figure 2. Representation of the classical blood-clotting cascades, ending in the removal of two fibrinopeptides from fibrinogen, and its self organization to produce fibrin (that may then be cross-linked).
The eventual result of fibrinolysis, that acts as a record of its extent, is d-dimer. Redrawn from a CC-BY publication at [26].
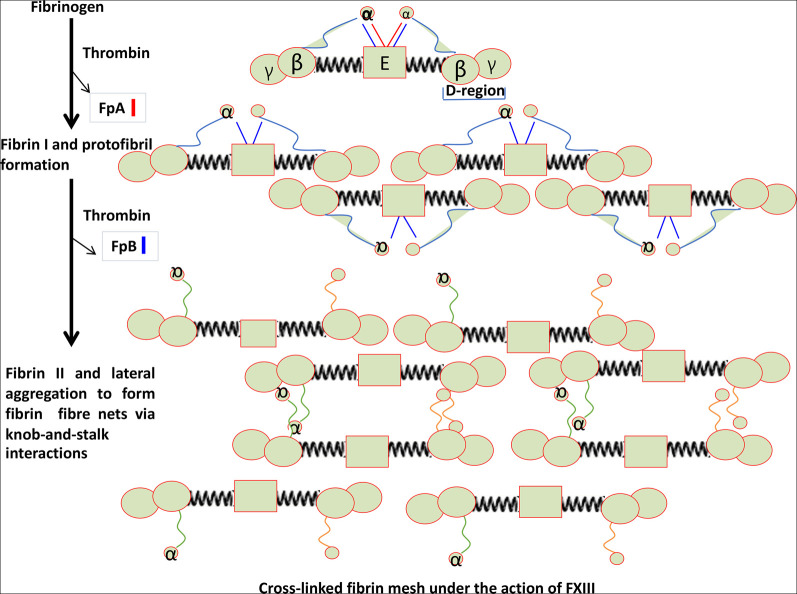
The action of thrombin on fibrinogen leads to the removal of two small fibrinopeptides (Figure 3), and this initiates the thermodynamically favorable conversion to fibrin, first as small protofibrils and then as long fibrils, typically with a diameter of 50–100 nm. Factor XIII is a transglutaminase which inserts cross-links into the developing clot, and also serves to cross-link α2-antiplasmin (α2-AP) to fibrin. α2-AP and other similar molecules are major inhibitors of plasmin, the enzyme which is mainly responsible for clot proteolysis (fibrinolysis).
Figure 3. Self organization of fibrinogen into fibrin under the action of thrombin in removing two fibrinopeptides.
Redrawn from a CC-BY publication at [26].
The structure of fibrin clots is typically characterized by the diameter of the spaghetti-like fibres and by the size of the residual pores [43]. However, in some cases these spaghetti-like structures with pores are absent. We initially characterized this phenomenon, observed in the electron microscope, as ‘dense matted deposits’ (e.g. [44–46]). We later discovered that such structures could be induced by highly substoichiometric amounts of bacterial lipopolysaccharide (LPS) (1 molecule LPS per 100,000,000 fibrinogen molecules), and it emerged that they too were, in fact, amyloid in character [26,47–50]. This highly substoichiometric induction (of a thermodynamically favourable process) is very important, as it effectively provides a means for a massive amplification of what may be a tiny amount of trigger material.
The list of molecules that could induce this anomalous clotting, additional to the bacterial LPS that was then our main focus, included iron ions [51–56], oestrogens [46,57], lipoteichoic acid [49,52], and serum amyloid A [58].
While the list of the many other molecules that can plausibly effect this anomalous amyloid-type clotting is unclear, such clots may also be observed in the blood of individuals with inflammatory diseases such as Alzheimer's [37,50,59–61], Parkinson's [37,48], type 2 diabetes [37,38,62–64], and rheumatoid arthritis [65–68]. Similar phenomena have also been observed in the pregnancy disorder pre-eclampsia [69], where there is also strong evidence for a microbial component [70,71].
Although this was a novel finding for ‘normal’ blood, it had previously been shown that β-sheet structures could be induced in fibrin artificially by mechanical means [72]. There have also been rare reports (see [26,73,74]) of amyloidogenic mutations (alleles) in the fibrinogen Aα chain. Additionally, it has long been known that aged fibrin deposits can bind amyloid dyes in tissue sections [75] and that fibrin-derived peptides can form cross-beta structures [76]. The amyloid form of the prion protein is highly resistant to proteolysis (resistance to proteolysis by proteinase K is used in an assay for PrPSc [77–79]), and so this provided a ready explanation for the different nature and persistence of the amyloid fibrin clots [52].
A note on ‘amyloid’ terminology
We recognize that there has occasionally been confusion in our use of the term ‘amyloid’ to describe amyloidogenic blood clotting, as blood is not seen as a source of the classical amyloidoses. While we could have used another term, the structures and staining of these blood clot fibres, presumably consisting of the ordered β-sheet architectures necessary (i) to bind fluorogenic amyloid stains and (ii) to be resistant to the normal sources of proteolysis, do reflect the well-established term ‘amyloid’. In addition, there is a very simple discrimination between ‘classical’ amyloids and those fibrin-rich amyloids that are our focus here: this is that the amyloid fibrils seen in classical amyloidoses tend to be ca 5–25 nm or so in diameter [21,22,80,81] whereas those in fibrin amyloid microclots tend to be in the range 50–150 nm or even more. They are thus easily distinguished microscopically, even without the greater analytic power afforded by proteomics [82], antibody staining [83], and so on. However, to avoid such ambiguity in the future, we consider it sensible to refer to the kinds of fibrin-based amyloids we are speaking about as fibrinaloids.
Amyloid fibrin microclots (fibrinaloids) in COVID-19 and long COVID
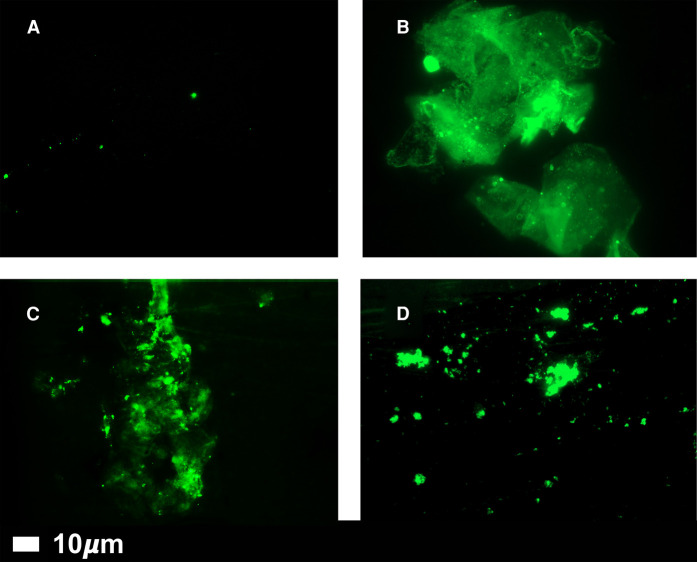
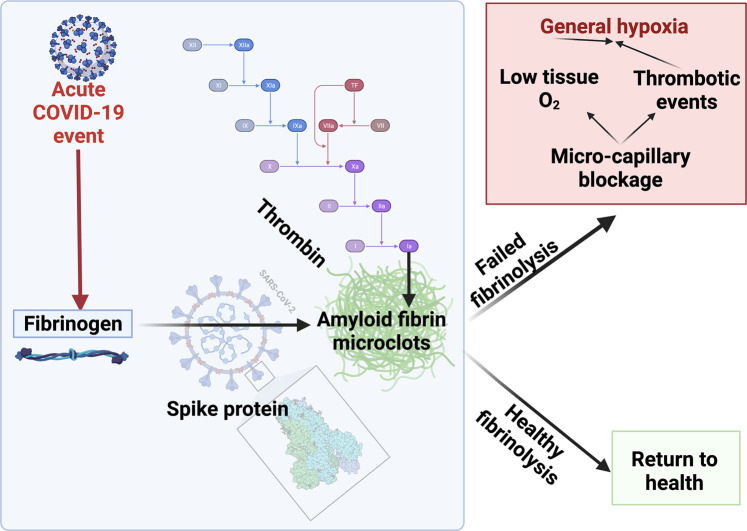
Coagulopathies [84–102], and especially the formation of extensive microclots in vivo, are a hallmark of both COVID [85,103–115] and long COVID [116,117], and we have demonstrated that these microclots too are amyloid in character [108,109,116]. Importantly, the addition of purified, recombinant SARS-CoV-2 S1 spike protein to coagulation-competent normal plasma is sufficient to induce the formation of anomalous clots [118] that adopt amyloid states that are also resistant to fibrinolysis [108]. Note that the observations of the microclots in (platelet-poor) plasma are performed without the addition of exogenous thrombin; they are naturally there in the circulation of patients with both acute and long COVID. The size of these amyloid microclots, that can be observed microscopically and stained e.g. with thioflavin T [108,109,116] (Figure 4; control vs LC plasma), are typically anywhere from 1–200 µm; this means that they can effectively block up, and inhibit blood flow through, all kinds of microcapillaries, thereby strongly lowering the availability of oxygen in tissues. As expected, they consist mainly of fibrin, but also contain many other proteins, including α2-antiplasmin [108] (and even the virus itself [119]). They also have heightened pro-inflammatory activity and elicit fibrin autoantibodies [118] (and maybe others). Elements of at least some spike protein variants of SARS-CoV-2 can also stimulate (in silico) the amyloidogenic aggregation of serum amyloid A [120]. More generally, there is considerable evidence for the induction of amyloid production by amyloidogenic proteins by viruses such as those of the Herpesviridae family, e.g. in Alzheimer's disease [121–125].
Figure 4. Fluorescence microscopy of representative micrographs showing microclots in the circulation of controls (A) and in patients with Long COVID (B–D).
Absence of significant amyloid microclots in the plasma of ‘normal’ individuals, and their significant presence in the plasma of individuals with long COVID. Platelet-poor plasma was produced by centrifugation at 3000×g for 15 min, stained with 5 µM thioflavin T, and imaged in a fluorescence microscope (Zeiss Axio Observer 7 with a Plan-Apochromat 63×/1.4 Oil DIC M27 objective (Carl Zeiss Microscopy, Munich, Germany). Wavelengths were Exc 450–488 nm/emission 499–529 nm, all as in [108].
‘Ground-glass’ opacities in COVID and long COVID
One of the early hallmarks of acute COVID-19 was the observation of extensive, diffuse opacities in high-resolution computed tomography (HRCT) scans of the lungs of patients, consistent with the breathing difficulties widely observed. These opacities resembled ground glass [126], and indeed are commonly referred to as ‘ground-glass opacities’ (GGO) [127–132]. They are largely formed of fibrin, though we are not aware that anyone has yet stained them for amyloid structures (which is what we believe they must be, and there is ample precedent for this in other diseases (e.g. [133–136])). In COVID patients they persist well beyond the acute phase [137].
More on the size and properties of microclots
As mentioned, the fibrinaloid microclots that we observe are typically in the range 1–200 µm on their longest axis. This is consistent with the ‘ground glass’ appearance, It is also true for ‘artificial’ amyloid-type protein structures [28], and for the kinds of amyloid seen as deposits in thrombotic microangiopathies [138–142]. However, the size distribution differs markedly between individuals (Figure 4), and while hard to pin down it does provide a ready general explanation for the very different manifestations of Long COVID (including suggestions that the term covers ‘multiple’ diseases). This is also true for prion diseases, where specific ‘strains’ based on particular conformations can propagate in the same form. In some cases, there can be differential staining of individual amyloids by different dyes [26,49,143–145], which also allows a certain degree of differentiation of the structure of the clots. In the case of Long COVID, we are probably not quite ready for such subtleties.
Role of d-dimer
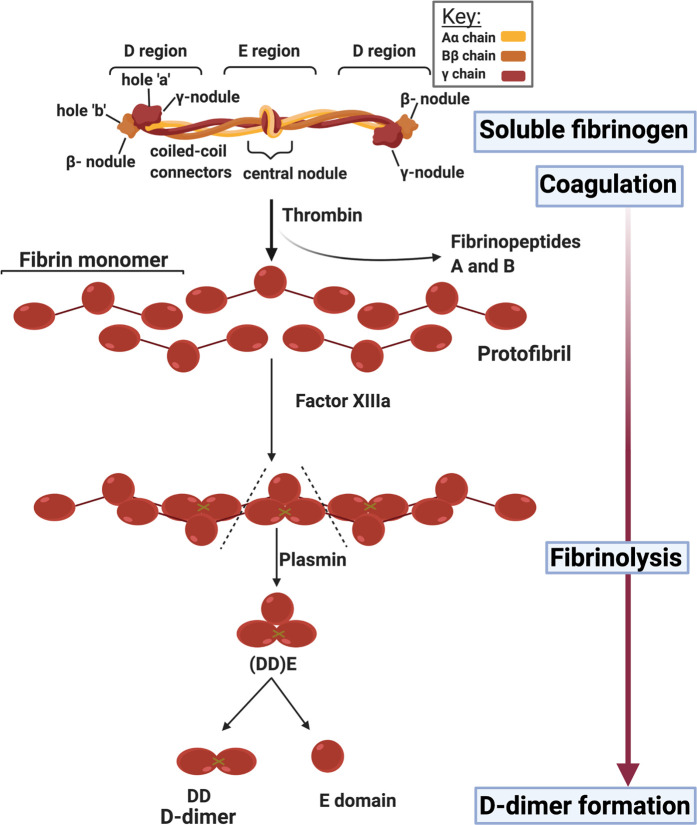
A major marker for fibrinolytic activity (Figure 2) is a polypeptide referred to as d-dimer Figure 5. It is a strong prognostic indicator for disease outcome (survival) in acute COVID [85,146–154]. It is, in effect, a composite measure of how much fibrinogen there was, how much was converted to fibrin clots (whether normal or fibrinaloids), and how much these then went on to be lysed. Each of these general steps may of course be regulated independently (i.e. proceed at a ‘fast’ or slow’ rate, as encoded simplistically in Figure 6), so the analysis of d-dimer measurements must take all of these issues into account. Thus high levels of d-dimer must of necessity represent the production of many more clots (and sufficient fibrinolysis to be occurring) but cannot of themselves reflect how many remained after their lysis nor which of them were ‘normal’ and which amyloid (see Figure 7 for a simplified pathway explanation).This explains why d-dimer levels are significantly raised following infection with many of the earlier variants of SARS-CoV-2, which produce multiple fibrinaloid microclots and Long COVID. However, d-dimer is massively increased in the case of the omicron variant where much clotting takes place but seemingly not to a fibrinaloid form, and thus effective clot fibrinolysis takes place. That there may be strong dependencies on the precise variant is not really surprising (omicron differs from both alpha and delta variants by more than 20 mutations [155], more than enough to vary a protein's activity 1000-fold in typical directed evolution experiments [156]). High d-dimer thus may, but does not have to [157], reflect disease severity given a particular SARS-CoV-2 variant; it depends on the context, and in particular the type of fibrin that can serve as the substrate for its production.
Figure 5. d-dimer production by fibrinolysis.
It is believed to be similar when produced from fibrinaloid clots, but the rate is considered to be slower. Image created with BioRender (https://biorender.com/).
Figure 6. d-dimer levels reflect both the rate of production and rate of degradation of clots, whether the clots are ‘normal’ or fibrinaloid in nature.
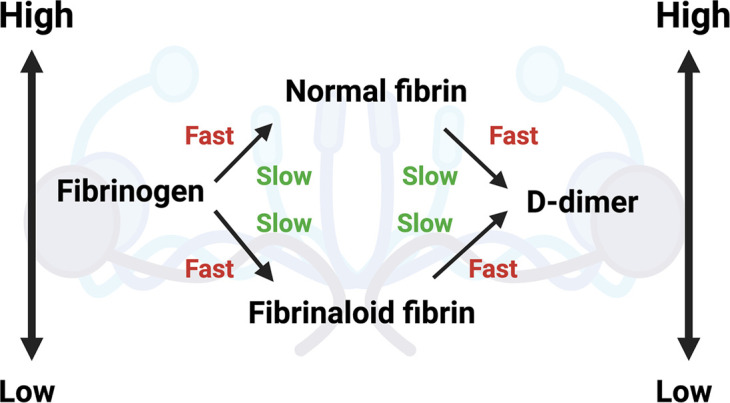
Image created with BioRender (https://biorender.com/.
Figure 7. A simplified diagram to explain microclot formation that might either be resolved via fibrinolytic processes after acute COVID-19 or, in some patients, result in a failed fibrinolytic process.
Image created with BioRender (https://biorender.com/).
Assessment of clotting
Why TEG rather than APTT, INR and other clotting measures?
A variety of optical clotting tests are in more-or-less common usage [158]. These include the aPTT (activated partial thromboplastin clotting time), prothrombin time (PT) and International normalized ratio (INR), but they are considered to have limited predictive value for bleeding and are not cost-effective [159,160]. INR and APTT are of limited value to predict clotting (and identifying patients that will bleed) in patients with acute and Long COVID. The test uses platelet poor plasma after discarding the cellular component of blood (including the platelets).
As blood clots, the viscosity of the clot increases, and measuring the time-dependent rate and extent of this viscoelasticity change directly in whole blood provides a more comprehensive and convenient in vitro assessment of the clotting behavior of the blood at the time. Note that clots of fibrinaloid that have already formed are effectively inert and invisible to such methods. Here the method of choice is known as Thromboelastography (TEG) [48,52,56,58,59,65,66,108,109,161–165] (or a related version known as ROTEM [166–173]). Figure 8 shows stylized traces of TEG, as typically found in health and disease. There are three main generalized traces: healthy (normocoagulable), hypercoagulable (as commonly seen in acute COVID-19) and hypocoagulable (during bleeding risk).
Figure 8. TEG® traces with the main parameters visualized.
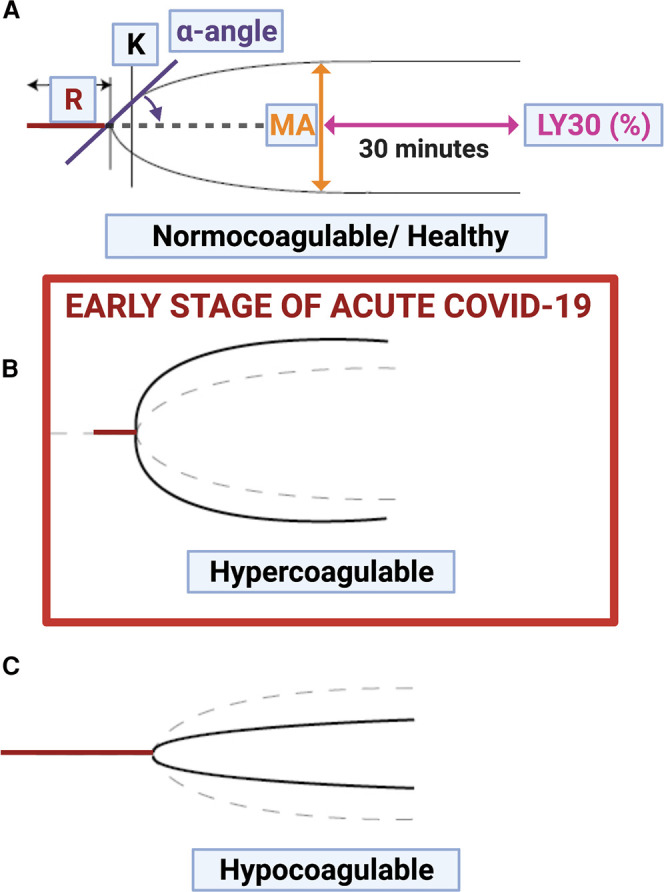
(A) Healthy (normocoagulable) trace; (B) Hypercoagulable trace (seen during early stages of acute COVID-19) and (C) Hypocoagulable trace. Image created with BioRender (https://biorender.com/). In B and C the dotted lines represent the normocoagulable case. R represents the time taken to initiate clot formation, α and K reflect the rate and extent of clot formation (e.g. [52]).
The TEG is a well-known point-of-care method, lately suggested as a good method for following blood clotting status in acute COVID-19, especially when anticoagulation regimes are prescribed to patients [109]. However, in our experience, it is not useful for determining (hyper)coagulation status in Long COVID, and should be reserved for those Long COVID patients where there is either a bleeding concern or actual bleeding. TEG should then be used in conjunction with platelet and microclot mapping, to follow patients closely when bleeding might be expected. Because microclots already contain clotted plasma proteins, and TEG only measures unclotted (soluble) plasma proteins, the TEG might not be suitable to determine the actual existing coagulation status (consisting of both clotted and soluble plasma proteins).
TEG trace ranges might in fact be within normal ranges, and our take on this is that the fibrin inside the microclots has already been coagulated, and trapped inside the microclots, leaving only a lower amount of soluble fibrinogen that might partake in the TEG assay. Therefore both the already-coagulated fibrin in the microclots and the soluble fibrinogen levels should in fact be considered, and preferably measured. Unfortunately the TEG cannot do that. However, we consider that TEG is vital for assessing the potential for coagulation when Long COVID patients are treated with anticoagulation therapy, or in any individual undergoing treatment designed to lower the levels of fibrinaloid microclots. In our view, TEG is therefore a much more true evaluation of the patient's coagulation state, as it uses whole blood, thus bringing in the very important role played by platelets.
INR, APTT d-dimer, fibrinogen levels and platelet count may be of help in the very late event of end stage acute COVID-19, where acute disseminated intravascular coagulopathy (DIC)-coagulopathy is seen. Interestingly, it is now accepted that DIC-bleeding events are uncommon in acute COVID-19 [111]. Even in this event, TEG is still of more use to manage this situation [109].
Mechanical properties of amyloid microclots
While we do not yet know the details of the mechanical properties of the fibrin amyloid microclots, it is known that amyloid fibrils can typically exhibit unusually high mechanical strength and resistance to deformation (e.g. [174–177]. The stiffness also increases with the thickness of the fibres [178]. This implies strongly that the fibrinaloids are likely to be more prone to ‘getting stuck’ in capillaries.
Bradford Hill criteria and microclots
The Bradford Hill criteria for causation of a disease Y by an environmental factor X [70,179] represent a useful framework for assessing the role of fibrin amyloid microclots in Long COVID, and are as follows: (1) strength of association between X and Y, (2) consistency of association between X and Y, (3) specificity of association between X and Y, (4) experiments verify the relationship between X and Y, (5) modification of X alters the occurrence of Y, and (6) biologically plausible cause and effect relationship. We think that the evidence is very strongly consistent with these criteria when X is represented by microclots and Y by Long COVID and potentially a variety of other conditions.
Ability of amyloid microclots to explain the symptoms of long COVID
While long COVID is a multi-system disorder with multiple symptoms of varying severity, it remains possible that there is in fact a particular major underlying cause (or that a very small number are the main contributors). Our view is as follows: given that amyloid microclots can clog up capillaries and inhibit the transport of O2 to tissues, this alone can in fact more or less self-evidently serve to explain a great many observations, and in particular the symptoms of both acute and long COVID. These obviously include breathlessness due to low O2 directly, and thrombotic events such as acute myocardial infarction [180,181], stroke [181–183], etc due to the microclots. The lack of O2 transport to tissues explains straightforwardly how a nominally respiratory disease also leads to the dysfunction of organs such as the kidney [184,185], PoTS (postural tachycardia syndrome [186]), myalgia in skeletal muscle [187,188], neurological disorders [189], and lactic acidosis [190,191] (the mass of lactate is too low to have been detected in our own COVID untargeted metabolomics experiments [192]), and potentially the benefits of hyperbaric O2 therapy [193]. It is also worth stressing that amyloid structures themselves tend to be cytotoxic, often via membrane disruption [194–198].
What makes long COVID long?
For any deterministic system to change its behavior there has to be a change of its parameters [199]. In the case of long COVID we need mechanisms that can explain how something that was initiated a long time ago can somehow persist. One source of the continuous production of a stimulus is represented by microbes, including virions, that persist in a largely dormant state (often in intracellular reservoirs) but can occasionally continue to replicate [1,200]. There is now considerable evidence for the persistence of SARS-CoV-2 [201]. Another is the continued release of sequestered microbially derived substances than can act as stimuli for continuing microclot formation. Here, the finding [202] that S1 spike protein can itself persist in CD16+ Monocytes in PASC for up to 15 months post-infection is highly relevant, as the amplification of trigger proteins to make microclots as part of the clotting mechanism means that miniscule (and highly substoichiometric) amounts of suitable triggers can suffice [26,47]. This alone is sufficient to account for the chronic nature of such diseases.
Microclot sequestration of biomarkers
An extra consequence of the production of microclots that sequester other proteins whose concentration would otherwise appear elevated is that those proteins do not then appear in plasma from which the microclots have been removed (e.g. by centrifugation), and thus they do not manifest, and cannot usefully be used, as biomarkers. Thus, our experience [108,116] is (i) that many such proteins including α2-AP and autoantibodies are so sequestered, and (ii) that the degradation of these protease-resistant microclots for classical proteomic analyses requires multiple rounds of trypsinization.
The role of autoantibodies and biomimicry
Autoantibodies are a feature of many chronic, inflammatory diseases, such as rheumatoid arthritis [68] (where Proteus spp are strongly implicated and where the cross-reactive epitopes leading to ‘mimicry’ by and of host protein targets are understood [68,203,204]). Importantly, it has already been shown that both acute [205,206] and Long COVID [207] are accompanied by immunological dysfunction, and by novel antibodies [1,206], including in the latter case to (an ‘abnormal’ but unspecified form of) fibrin [118]. We recognise that any change in the conformation of a protein can result in the generation of novel epitopes that can thereby lead to the production of novel antibodies; indeed the use of secondary antibodies in the detection of small molecules [208] relies precisely on this fact. Although we consider that the more primary event is the generation of fibrinaloid microclots, we also recognise that they are likely to be able to change the natural conformation of many proteins that might normally present as harmless (seen as ‘self’). However, the detailed nature of these autoantibodies is not known (but a subject of considerable present interest [1]).
Epidemiological aspects
One approach to understanding the mechanisms of long COVID is to analyse epidemiological data, since the characteristics of those experiencing acute COVID differ markedly from those with long COVID [11]. Among these (and in significant contrast with the case of acute COVID [209]), is a striking over-representation of long COVID in women, and especially younger women [3,10,210]. From this point of view, it is of considerable interest that female sex hormones can induce anomalous blood clotting [46,57,211].
Haemophiliacs (and others with hypocoagulatory disorders) represent a complex test case, because depending on the stage of the disease [85], COVID can be both hyper- and hypocoagulatory [212]. However, we are not presently aware of any analyses of fibrinaloid microclots in such individuals.
Strain differences
While it has been somewhat slowed by the generally sluggish recognition of Long COVID, it is to be expected that different strains of SARS-CoV-2 may have different tendencies to induce it. To this end, it is reasonable that if microclots are important to LC their prevalence should also vary with the severity or frequency of LC induced by different strains of SARS-CoV-2. In a sense this would provide a very important kind of ‘control’, since the only thing varying as the stimulus is the strain of SARS-CoV-2.
Other causes of amyloid microclots
While we here focus on SARS-CoV-2, we note that all kinds of molecules have been shown to affect the extent of fibrinaloid clot formation, including iron, other amyloids, bacterial cell wall components, etc., and that we have observed them in a variety of chronic, inflammatory diseases including Alzheimer's [37,50,60–62], Parkinson's disease [37,48], Type 2 diabetes [37,38,62,63,213,214] (where the amyloid protein amylin [215–217] is of course a well-known player) and rheumatoid arthritis [65,67]. Although not yet tested directly, we consider it likely that this will also be true for infectious diseases known to be causing similar post-infection syndromes, such as Dengue [218–220], Ebola [221–223], Lyme [224], Zika and others where viruses persist and can cause microangiopathies [139] that we suspect are also amyloid in character. There is also likely to be a role for molecules raised in pre-existing inflammatory diseases, as well as substances produced by dyregulation microbiomes [225], and Leiden factor V [226,227].
Similarities to ME/CFS
As well as the post-infection diseases referred to above, the emergence of long COVID has brought to the fore its similarities to other even more widely established syndromes such as myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) [1,228], and full details are available in these recent reviews [1,228]. Berg and colleagues have also highlighted the role of coagulopathies in ME/CFS [229]. We note, too, that other ‘anomalous’ diseases bearing symptoms that overlap with those of ME/CFS include Gulf war syndrome [230], where again we would hazard that the analysis of fibrinaloid microclot formation would have a positive outcome. Thus, while we consider it likely that the phenomena we describe will also broadly be true for ME/CFS, we focus here on PASC/Long COVID.
Endotheliopathy
An important component of severe acute COVID-19 disease accompanying pathological clotting, is virus-induced endotheliopathy resulting in systemic endotheliitis [111,231–235]. Endotheliitis is central to initiating a state of failing normal clotting physiology and it is also known to be significantly linked to coagulopathies, as it activates microthrombotic pathways and initiates endotheliopathy-associated intravascular microthrombi [236].
A role for iron dysregulation
A consequence of cell death, such as occurs in endotheliopathy, is the release in COVID patients of the normally intracellular iron storage protein ferritin [237,238], which can then release free iron [239]. Iron dysregulation is an accompaniment to a huge number of chronic, inflammatory diseases [200,240,241], and iron dysregulation is a significant accompaniment in COVID [238,242–245]. Indeed, there is evidence that SARS-CoV-2 can release iron from haemoglobin directly [246]. The involvement of iron dysregulation would be consistent with the potentially protective effects of chelating it, e.g. with lactoferrin [247–250] or other chelators [251–254].
Role of platelets
The important role that platelets play in acute COVID-19 (Figure 9), has been discussed in the context of disease severity, the development of endotheliopathy and as general drivers of coagulation pathology [232,233,255–257]. Platelets interact with circulating inflammatory molecules, the (damaged) endothelium itself, and also with immune cells, resulting in platelet/cellular and platelet/molecule complexes [258,259]. Platelet complexes are mediated by membrane-membrane interactions via receptor binding. Two of the molecules central in this discussion are fibrinogen and VWF, and platelets form significant complexes with them both [260]. Ultimately, platelet–cell and platelet–coagulation molecule protein complexes form part of platelet activation mechanisms and vascular remodeling and these complexes drive granule secretion, surface glycoprotein expression, and molecular activation platelet hyperactivation pathways [258,259].
Figure 9. Platelet interactions with viruses.
Figure adapted from [259]. Various platelet receptors can mediate binding to viral particles, where pattern recognition receptors recognize viral signals, viral products and also modulate platelet function. Platelets mediate viral attack by secreting virucidal proteins and by engulfing viral particles, as well as by interacting with immune cells and enhancing the immune response. Virus-platelet aggregates and platelets with a viral load are targeted by leukocytes, and platelets are ultimately cleared from the circulation (Figure created with https://biorender.com/).
Consequences for treatment protocols
Given that there is a definite background removal rate of fibrinaloid microclots, albeit slower than that of normal non-amyloid clots, it is then mostly necessary to ensure that they do not form further. Consequently, we do not see a role for ‘clot-busters’ as have occasionally been used when acute COVID-19 is accompanied by Acute Respiratory Distress Syndrome [261]. However, addressing only one of the pathologies (either clot pathologies or platelet hyperactivation) will always tend to fail, as was also noted in recent clinical trials where just one of the two therapies was trialed in Acute COVID-19. Lawler and co-workers [262] investigated the use of therapeutic anticoagulation with heparin in noncritically ill patients with COVID-19. (We note too that heparin, like lactoferrin [250], may have antiviral properties [263–266].) Lopes and coworkers [267] also investigated the use of therapeutic versus prophylactic anticoagulation for patients admitted to hospital with COVID-19. Earlier, Viecca and colleagues reported [268] on a single-center, investigator-initiated, proof-of-concept, case control, phase IIb study. The study [268] explored the effects of anti-platelet therapy on arterial oxygenation and on clinical outcomes in patients with severe COVID-19 with hypercoagulability. Outcomes were not significantly positive in any of the trials mentioned.
However, it must be noted that the trials were done on acute COVID-19 patients. This has led us to suggest a multipronged approach and a regime of triple anticoagulation treatment [231], where Long COVID patients might be treated by one month of dual antiplatelet therapy (DAPT) (Clopidogrel 75 mg/Aspirin 75 mg) once a day, as well as a direct oral anticoagulant (DOAC) (Apixiban) 5 mg twice a day, together with a proton pump inhibitor (PPI) (e.g. pantoprazole 40 mg/day for gastric protection). Such a treatment regime showed promise under condition of clinical practice (there were no treatment-free ‘controls’) [231]. However, especially because of the potential for hypocoagulation (bleeding), it must only be followed under strict and qualified medical guidance in which we recommend the regular assessment of coagulation status before and during treatment. It (as do any other methods [269]) now needs to be studied in a proper randomized controlled trial.
We do note that the use of triple therapy is not new in clinical practice. Its uses predate COVID-19, where it is successfully prescribed in patients where thromboses (which may or may not involve fibrinaloid clots) are particularly worrisome and in various coronary diseases accompanied by atrial fibrillation. Here strong anticoagulant treatments tailored to the needs of the individual patient are recommended [270], commonly involving dual or triple treatments with various anticoagulant agents. The present authors have focused on the ‘triple treatment’ for anticoagulation that involves an oral anticoagulant plus two drugs designed to decrease platelet activation (usually the P2Y12 inhibitor clopidogrel, plus low-dose aspirin). (In addition, a gastric proton pump inhibitor is also given to reduce the likelihood of gastric bleeding.)
What is needed next?
Many pieces of research-level evidence (especially [85,101,106,108,109,116,231,271]) suggest strongly that fibrin amyloid microclots, driven by the presence of the SARS-CoV-2 spike protein, are an inevitable accompaniment to (and a likely cause of) Long COVID. A biologically coherent explanation [272,273] can link such observations with the other observable symptoms of Long COVID, and thus serve to satisfy the logic normally required [179,274,275] to provide a causative explanation.
However, we are still potentially far from translating this kind of understanding into both diagnostics and therapeutics. Some of the elements that are needed both to make the evidence even more robust and to pass regulatory approval more widely include the following:
Our fibrin amyloid diagnostic methods, presently semi-quantitative, must be subject to further optimization, made robust and quantitative, and reproduced more widely
Work is required to provide a simple, cheap and widely available point-of-care instrument to effect the necessary measurements of the number, size and nature of fibrinaloid micrclots
Longitudinal studies, and those relating fibrinaloid presence to the severity of Long COVID, will help recognise the role of different fibrinaloids in causing different symptoms; these may vary predictively between SARS-CoV-2 strains
There would be value in assessing other blood parameters simultaneously (e.g. fibrinogen, d-dimer, von Willebrand Factor) plus suitable inflammatory cytokines
Armed with the above, we should reasonably expect clinicians to be able to obtain ethics for a suitable designed Randomized Controlled Trial of anticoagulant and platelet inhibitor regimes, coupled to the assessment, using viscoelastic assays such as Thromboelastography (TEG or ROTEM or Sonoclot), before and during such treatments of coagulation potential so as to guard against any hypocoagulation and the dangers of bleeding.
Such individuals should be followed for a period after the end of treatment to assess the degree of permanence
Of course it is easy to design detailed and expensive trials, accompanied by a great many other measurements of covariates, but the above sets out what we would consider as the minimum necessary to strengthen the claims that the analysis and removal of fibrin amyloid microclots should have major utility in providing substantial benefits to those with Long COVID, and likely in related conditions as well.
Conclusions
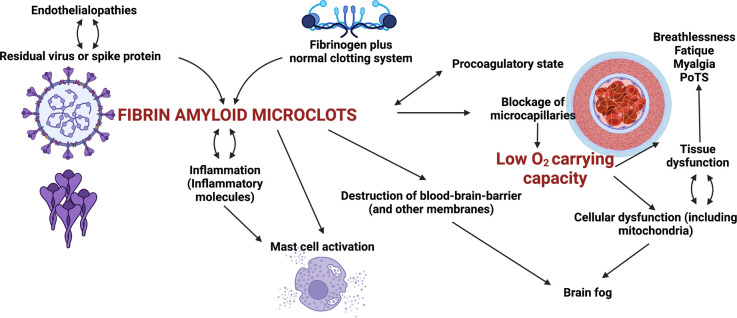
Here we have argued, and focus on the fact, that Long COVID is characterized by the presence of persistent fibrin amyloid microclots that might block capillaries and inhibit the transport of O2 to tissues, entrapping numerous inflammatory molecules, including those that prevent clot breakdown (as we have indeed recently shown) (Figure 10).
Figure 10. Some of the sequelae of fibrinaloid microclot formation in the symptomology of Long COVID.
Many others, such as a role for auto-antibodies, are not shown.
In addition to microclot formation, significant platelet dysfunction and a systemic endotheliitis drive systemic cellular hypoxia. These pathologies can explain most, if not all, of the lingering symptoms to which individuals with long COVID refer. We have noted that amyloid microclots, platelet hyperactivation and endothelial dysfunction, might form a suitable set of foci for the clinical treatment of the symptoms of long COVID [231]. Therefore, if fibrinaloid microclots are largely responsible for the symptoms of Long COVID, their removal is to be seen as paramount for relieving these symptoms and allowing the body to repair itself.
Acknowledgements
D.B.K. thanks the Novo Nordisk Foundation for financial support (grant NNF20CC0035580).
Abbreviations
- DAPT
dual antiplatelet therapy
- DIC
disseminated intravascular coagulopathy
- INR
International normalized ratio
- LPS
lipopolysaccharide
- PASC
Post-acute sequelae of COVID
- PPP
platelet-poor plasma
- TEG
Thromboelastography
Competing Interests
E.P. is a named inventor on a patent application covering the use of fluorescence methods for microclot detection in Long COVID.
Open Access Statement
Open access for this article was enabled by the participation of University of Liverpool in an all-inclusive Read & Publish pilot with Portland Press and the Biochemical Society under a transformative agreement with JISC.
References
- 1.Proal, A.D. and VanElzakker, M.B. (2021) Long COVID or post-acute sequelae of COVID-19 (PASC): an overview of biological factors that may contribute to persistent symptoms. Front. Microbiol. 12, 698169 10.3389/fmicb.2021.698169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Logue, J.K., Franko, N.M., McCulloch, D.J., McDonald, D., Magedson, A., Wolf, C.R.et al. (2021) Sequelae in adults at 6 months after COVID-19 infection. JAMA Netw. Open. 4, e210830 10.1001/jamanetworkopen.2021.0830 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nalbandian, A., Sehgal, K., Gupta, A., Madhavan, M.V., McGroder, C., Stevens, J.S.et al. (2021) Post-acute COVID-19 syndrome. Nat. Med. 27, 601–615 10.1038/s41591-021-01283-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Asadi-Pooya, A.A., Nemati, H., Shahisavandi, M., Akbari, A., Emami, A., Lotfi, M.et al. (2021) Long COVID in children and adolescents. World J. Pediatr. 17, 495–499 10.1007/s12519-021-00457-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gurdasani, D., Akrami, A., Bradley, V.C., Costello, A., Greenhalgh, T., Flaxman, S.et al. (2022) Long COVID in children. Lancet Child Adolesc. Health 6, e2 10.1016/S2352-4642(21)00342-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Alwan, N.A. (2021) The road to addressing long COVID. Science 373, 491–493 10.1126/science.abg7113 [DOI] [PubMed] [Google Scholar]
- 7.Di Toro, A., Bozzani, A., Tavazzi, G., Urtis, M., Giuliani, L., Pizzoccheri, R.et al. (2021) Long COVID: long-term effects? Eur. Heart J. Suppl. 23, E1–E5 10.1093/eurheartj/suab080 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Marshall, M. (2021) The four most urgent questions about long COVID. Nature 594, 168–170 10.1038/d41586-021-01511-z [DOI] [PubMed] [Google Scholar]
- 9.Michelen, M., Manoharan, L., Elkheir, N., Cheng, V., Dagens, A., Hastie, C.et al. (2021) Characterising long COVID: a living systematic review. BMJ Glob. Health 6, e005427 10.1136/bmjgh-2021-005427 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sigfrid, L., Drake, T.M., Pauley, E., Jesudason, E.C., Olliaro, P., Lim, W.S.et al. (2021) Long COVID in adults discharged from UK hospitals after COVID-19: a prospective, multicentre cohort study using the ISARIC WHO clinical characterisation protocol. Lancet Reg. Health Eur. 8, 100186 10.1016/j.lanepe.2021.100186 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Davis, H.E., Assaf, G.S., McCorkell, L., Wei, H., Low, R.J., Re'em, Y.et al. (2021) Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. EClinicalMedicine 38, 101019 10.1016/j.eclinm.2021.101019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Deer, R.R., Rock, M.A., Vasilevsky, N., Carmody, L., Rando, H., Anzalone, A.J.et al. (2021) Characterizing long COVID: deep phenotype of a complex condition. EBioMedicine 74, 103722 10.1016/j.ebiom.2021.103722 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Décary, S., Gaboury, I., Poirier, S., Garcia, C., Simpson, S., Bull, M.et al. (2021) Humility and acceptance: working within Our limits With long COVID and myalgic encephalomyelitis/chronic fatigue syndrome. J. Orthop. Sports Phys. Ther. 51, 197–200 10.2519/jospt.2021.0106 [DOI] [PubMed] [Google Scholar]
- 14.Greenhalgh, T., Knight, M., A'Court, C., Buxton, M. and Husain, L. (2020) Management of post-acute COVID-19 in primary care. BMJ 370, m3026 10.1136/bmj.m3026 [DOI] [PubMed] [Google Scholar]
- 15.Anfinsen, C.B. (1973) Principles that govern the folding of protein chains. Science 181, 223–230 10.1126/science.181.4096.223 [DOI] [PubMed] [Google Scholar]
- 16.Prusiner, S.B. (1998) Prions. Proc. Natl Acad. Sci. U.S.A. 95, 13363–13383 10.1073/pnas.95.23.13363 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Baral, P.K., Yin, J., Aguzzi, A. and James, M.N.G. (2019) Transition of the prion protein from a structured cellular form (PrP(C)) to the infectious scrapie agent (PrP(Sc)). Protein Sci. 28, 2055–2063 10.1002/pro.3735 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tycko, R. (2016) Structure of aggregates revealed. Nature 537, 492–493 10.1038/nature19470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ke, P.C., Zhou, R., Serpell, L.C., Riek, R., Knowles, T.P.J., Lashuel, H.A.et al. (2020) Half a century of amyloids: past, present and future. Chem. Soc. Rev. 49, 5473–5509 10.1039/C9CS00199A [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Collinge, J. (2016) Mammalian prions and their wider relevance in neurodegenerative diseases. Nature 539, 217–226 10.1038/nature20415 [DOI] [PubMed] [Google Scholar]
- 21.Iadanza, M.G., Jackson, M.P., Hewitt, E.W., Ranson, N.A. and Radford, S.E. (2018) A new era for understanding amyloid structures and disease. Nat. Rev. Mol. Cell Biol. 19, 755–773 10.1038/s41580-018-0060-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Fitzpatrick, A.W. and Saibil, H.R. (2019) Cryo-EM of amyloid fibrils and cellular aggregates. Curr. Opin. Struct. Biol. 58, 34–42 10.1016/j.sbi.2019.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Carta, M. and Aguzzi, A. (2021) Molecular foundations of prion strain diversity. Curr. Opin. Neurobiol. 72, 22–31 10.1016/j.conb.2021.07.010 [DOI] [PubMed] [Google Scholar]
- 24.Blancas-Mejía, L.M. and Ramirez-Alvarado, M. (2013) Systemic amyloidoses. Annu. Rev. Biochem. 82, 745–774 10.1146/annurev-biochem-072611-130030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Palladini, G. and Merlini, G. (2013) Systemic amyloidoses: what an internist should know. Eur. J. Intern. Med. 24, 729–739 10.1016/j.ejim.2013.10.007 [DOI] [PubMed] [Google Scholar]
- 26.Kell, D.B. and Pretorius, E. (2017) Proteins behaving badly. Substoichiometric molecular control and amplification of the initiation and nature of amyloid fibril formation: lessons from and for blood clotting. Progr. Biophys. Mol. Biol. 123, 16–41 10.1016/j.pbiomolbio.2016.08.006 [DOI] [PubMed] [Google Scholar]
- 27.Cao, K.J. and Yang, J. (2018) Translational opportunities for amyloid-targeting fluorophores. Chem. Commun (Camb) 54, 9107–9118 10.1039/C8CC03619E [DOI] [PubMed] [Google Scholar]
- 28.Apter, B., Lapshina, N., Barhom, H., Fainberg, B., Handelman, A., Accardo, A.et al. (2020) Fluorescence phenomena in amyloid and amyloidogenic bionanostructures. Crustals 10, 668 10.3390/cryst10080668 [DOI] [Google Scholar]
- 29.Howie, A.J. and Brewer, D.B. (2009) Optical properties of amyloid stained by Congo red: history and mechanisms. Micron 40, 285–301 10.1016/j.micron.2008.10.002 [DOI] [PubMed] [Google Scholar]
- 30.Peccati, F., Pantaleone, S., Riffet, V., Solans-Monfort, X., Contreras-Garcia, J., Guallar, V.et al. (2017) Binding of thioflavin T and related probes to polymorphic models of amyloid-beta fibrils. J. Phys. Chem. B 121, 8926–8934 10.1021/acs.jpcb.7b06675 [DOI] [PubMed] [Google Scholar]
- 31.Sulatskaya, A.I., Kuznetsova, I.M. and Turoverov, K.K. (2012) Interaction of thioflavin T with amyloid fibrils: fluorescence quantum yield of bound dye. J. Phys. Chem. B 116, 2538–2544 10.1021/jp2083055 [DOI] [PubMed] [Google Scholar]
- 32.Sulatskaya, A.I., Rodina, N.P., Sulatsky, M.I., Povarova, O.I., Antifeeva, I.A., Kuznetsova, I.M.et al. (2018) Investigation of alpha-Synuclein amyloid fibrils using the fluorescent probe thioflavin T. Int. J. Mol. Sci. 19, 2486 10.3390/ijms19092486 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Khurana, R., Coleman, C., Ionescu-Zanetti, C., Carter, S.A., Krishna, V., Grover, R.K.et al. (2005) Mechanism of thioflavin T binding to amyloid fibrils. J. Struct. Biol. 151, 229–238 10.1016/j.jsb.2005.06.006 [DOI] [PubMed] [Google Scholar]
- 34.Amdursky, N., Erez, Y. and Huppert, D. (2012) Molecular rotors: what lies behind the high sensitivity of the thioflavin-T fluorescent marker. Acc. Chem. Res. 45, 1548–1557 10.1021/ar300053p [DOI] [PubMed] [Google Scholar]
- 35.Biancalana, M. and Koide, S. (2010) Molecular mechanism of Thioflavin-T binding to amyloid fibrils. Biochim. Biophys. Acta 1804, 1405–1412 10.1016/j.bbapap.2010.04.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Klingstedt, T. and Nilsson, K.P.R. (2012) Luminescent conjugated poly- and oligo-thiophenes: optical ligands for spectral assignment of a plethora of protein aggregates. Biochem. Soc. Trans. 40, 704–710 10.1042/BST20120009 [DOI] [PubMed] [Google Scholar]
- 37.de Waal, G.M., Engelbrecht, L., Davis, T., de Villiers, W.J.S., Kell, D.B. and Pretorius, E. (2018) Correlative light-electron microscopy detects lipopolysaccharide and its association with fibrin fibres in Parkinson's disease, Alzheimer's disease and type 2 diabetes mellitus. Sci. Rep. 8, 16798 10.1038/s41598-018-35009-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pretorius, E., Page, M.J., Engelbrecht, L., Ellis, G.C. and Kell, D.B. (2017) Substantial fibrin amyloidogenesis in type 2 diabetes assessed using amyloid-selective fluorescent stains. Cardiovasc. Diabetol. 16, 141 10.1186/s12933-017-0624-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Baskurt, O.K., Hardeman, M.R., Rampling, M.W. and Meiselman, H.J. (2007) Handbook of Hemorheology and Hemodynamics, IOS Press, Amsterdam [Google Scholar]
- 40.Cilia La Corte, A.L., Philippou, H. and and Ariëns, R.A.S. (2011) Role of fibrin structure in thrombosis and vascular disease. Adv. Protein Chem. Struct. Biol. 83, 75–127 10.1016/B978-0-12-381262-9.00003-3 [DOI] [PubMed] [Google Scholar]
- 41.Litvinov, R.I., Pieters, M., de Lange-Loots, Z. and Weisel, J.W. (2021) Fibrinogen and fibrin. Subcell. Biochem. 96, 471–501 10.1007/978-3-030-58971-4_15 [DOI] [PubMed] [Google Scholar]
- 42.Weisel, J.W. and Litvinov, R.I. (2017) Fibrin formation, structure and properties. Subcell. Biochem. 82, 405–456 10.1007/978-3-319-49674-0_13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Undas, A. (2016) How to assess fibrinogen levels and fibrin clot properties in clinical practice? Semin. Thromb. Hemost. 42, 381–388 10.1055/s-0036-1579636 [DOI] [PubMed] [Google Scholar]
- 44.Pretorius, E., Briedenhann, S., Marx, J. and Franz, R.C. (2006) Structural changes in the fibrin network of a Pretoria family with dysfibrinogenemia: a scanning electron microscopical study. Ultrastruct. Pathol. 30, 167–176 10.1080/01913120600689772 [DOI] [PubMed] [Google Scholar]
- 45.Pretorius, E. and Lipinski, B. (2013) Differences in morphology of fibrin clots induced with thrombin and ferric ions and its pathophysiological consequences. Heart Lung Circ. 22, 447–449 10.1016/j.hlc.2012.10.010 [DOI] [PubMed] [Google Scholar]
- 46.Swanepoel, A.C., Visagie, A., de Lange, Z., Emmerson, O., Nielsen, V.G. and Pretorius, E. (2016) The clinical relevance of altered fibrinogen packaging in the presence of 17beta-estradiol and progesterone. Thromb. Res. 146, 23–34 10.1016/j.thromres.2016.08.022 [DOI] [PubMed] [Google Scholar]
- 47.Pretorius, E., Mbotwe, S., Bester, J., Robinson, C.J. and Kell, D.B. (2016) Acute induction of anomalous and amyloidogenic blood clotting by molecular amplification of highly substoichiometric levels of bacterial lipopolysaccharide. J. R. Soc. Interface. 123, 20160539 10.1098/rsif.2016.0539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Pretorius, E., Page, M.J., Mbotwe, S. and Kell, D.B. (2018) Lipopolysaccharide-binding protein (LBP) can reverse the amyloid state of fibrin seen or induced in Parkinson's disease. PLoS ONE 13, e0192121 10.1371/journal.pone.0192121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pretorius, E., Page, M.J., Hendricks, L., Nkosi, N.B., Benson, S.R. and Kell, D.B. (2018) Both lipopolysaccharide and lipoteichoic acids potently induce anomalous fibrin amyloid formation: assessment with novel amytracker™ stains. J. R. Soc. Interface 15, 20170941 10.1098/rsif.2017.0941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pretorius, E., Bester, J., Page, M.J. and Kell, D.B. (2018) The potential of LPS-binding protein to reverse amyloid formation in plasma fibrin of individuals with Alzheimer-type dementia. Front. Aging Neurosci. 10, 257 10.3389/fnagi.2018.00257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jankun, J., Landeta, P., Pretorius, E., Skrzypczak-Jankun, E. and Lipinski, B. (2014) Unusual clotting dynamics of plasma supplemented with iron(III). Int. J. Mol. Med. 33, 367–372 10.3892/ijmm.2013.1585 [DOI] [PubMed] [Google Scholar]
- 52.Kell, D.B. and Pretorius, E. (2015) The simultaneous occurrence of both hypercoagulability and hypofibrinolysis in blood and serum during systemic inflammation, and the roles of iron and fibrin(ogen). Integr. Biol. 7, 24–52 10.1039/c4ib00173g [DOI] [PubMed] [Google Scholar]
- 53.Lipinski, B., Pretorius, E., Oberholzer, H.M. and Van Der Spuy, W.J. (2012) Iron enhances generation of fibrin fibers in human blood: implications for pathogenesis of stroke. Microsc. Res. Tech. 75, 1185–1190 10.1002/jemt.22047 [DOI] [PubMed] [Google Scholar]
- 54.Pretorius, E., Vermeulen, N., Bester, J. and Lipinski, B. (2013) Novel use of scanning electron microscopy for detection of iron-induced morphological changes in human blood. Microsc. Res. Tech. 76, 268–271 10.1002/jemt.22163 [DOI] [PubMed] [Google Scholar]
- 55.Pretorius, E., Bester, J., Vermeulen, N., Lipinski, B., Gericke, G.S. and Kell, D.B. (2014) Profound morphological changes in the erythrocytes and fibrin networks of patients with hemochromatosis or with hyperferritinemia, and their normalization by iron chelators and other agents. PLoS ONE 9, e85271 10.1371/journal.pone.0085271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Pretorius, E. and Kell, D.B. (2014) Diagnostic morphology: biophysical indicators for iron-driven inflammatory diseases. Integr. Biol. 6, 486–510 10.1039/C4IB00025K [DOI] [PubMed] [Google Scholar]
- 57.Swanepoel, A.C., Lindeque, B.G., Swart, P.J., Abdool, Z. and Pretorius, E. (2014) Estrogen causes ultrastructural changes of fibrin networks during the menstrual cycle: a qualitative investigation. Microsc. Res. Tech. 77, 594–601 10.1002/jemt.22378 [DOI] [PubMed] [Google Scholar]
- 58.Page, M.J., Thomson, G.J.A., Nunes, J.M., Engelbrecht, A.M., Nell, T.A., de Villiers, W.J.S.et al. (2019) Serum amyloid A binds to fibrin(ogen), promoting fibrin amyloid formation. Sci. Rep. 9, 3102 10.1038/s41598-019-39056-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bester, J., Soma, P., Kell, D.B. and Pretorius, E. (2015) Viscoelastic and ultrastructural characteristics of whole blood and plasma in Alzheimer-type dementia, and the possible role of bacterial lipopolysaccharides (LPS). Oncotarget Gerontol. 6, 35284–35303 10.18632/oncotarget.6074 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Lipinski, B. and Pretorius, E. (2013) The role of iron-induced fibrin in the pathogenesis of Alzheimer's disease and the protective role of magnesium. Front. Hum. Neurosci. 7, 735 10.3389/fnhum.2013.00735 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lipinski, B. and Pretorius, W. (2014) Iron-induced fibrin formation may explain vascular pathology in Alzheimer's disease. Folia Neuropathol. 52, 205 10.5114/fn.2014.43792 [DOI] [PubMed] [Google Scholar]
- 62.Pretorius, E., Oberholzer, H.M., van der Spuy, W.J., Swanepoel, A.C. and Soma, P. (2011) Qualitative scanning electron microscopy analysis of fibrin networks and platelet abnormalities in diabetes. Blood Coagul. Fibrinol. 22, 463–467 10.1097/MBC.0b013e3283468a0d [DOI] [PubMed] [Google Scholar]
- 63.Pretorius, E., Bester, J., Vermeulen, N., Alummoottil, S., Soma, P., Buys, A.V.et al. (2015) Poorly controlled type 2 diabetes is accompanied by significant morphological and ultrastructural changes in both erythrocytes and in thrombin-generated fibrin: implications for diagnostics. Cardiovasc. Diabetol. 13, 30 10.1186/s12933-015-0192-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Soma, P. and Pretorius, E. (2015) Interplay between ultrastructural findings and atherothrombotic complications in type 2 diabetes mellitus. Cardiovasc. Diabetol. 14, 96 10.1186/s12933-015-0261-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Bezuidenhout, J., Venter, C., Roberts, T., Tarr, G., Kell, D. and Pretorius, E. (2020) The atypical fibrin fibre network in rheumatoid arthritis and its relation to autoimmunity, inflammation and thrombosis. bioRxiv 2020.05.28.121301v1 10.1101/2020.05.28.121301 [DOI] [Google Scholar]
- 66.Bezuidenhout, J., Venter, C., Roberts, T., Tarr, G., Kell, D. and Pretorius, E. (2020) Detection of citrullinated fibrin in plasma clots of RA patients and its relation to altered structural clot properties, disease-related inflammation and prothrombotic tendency. Front. Immunol. 11, 577523 10.3389/fimmu.2020.577523 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Pretorius, E., Oberholzer, H.M., van der Spuy, W.J., Swanepoel, A.C. and Soma, P. (2012) Scanning electron microscopy of fibrin networks in rheumatoid arthritis: a qualitative analysis. Rheumatol. Int. 32, 1611–1615 10.1007/s00296-011-1805-2 [DOI] [PubMed] [Google Scholar]
- 68.Pretorius, E., Akeredolu, O.-O., Soma, P. and Kell, D.B. (2017) Major involvement of bacterial components in rheumatoid arthritis and its accompanying oxidative stress, systemic inflammation and hypercoagulability. Exp. Biol. Med. 242, 355–373 10.1177/1535370216681549 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Buhimschi, I.A., Nayeri, U.A., Zhao, G., Shook, L.L., Pensalfini, A., Funai, E.F.et al. (2014) Protein misfolding, congophilia, oligomerization, and defective amyloid processing in preeclampsia. Sci. Transl. Med. 6, 245ra292 10.1126/scitranslmed.3008808 [DOI] [PubMed] [Google Scholar]
- 70.Kell, D.B. and Kenny, L.C. (2016) A dormant microbial component in the development of pre-eclampsia. Front. Med. Obs. Gynecol. 3, 60 10.3389/fmed.2016.00060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kenny, L.C. and Kell, D.B. (2018) Immunological tolerance, pregnancy and pre-eclampsia: the roles of semen microbes and the father. Front. Med. Obs. Gynecol. 4, 239 10.3389/fmed.2017.00239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Litvinov, R.I., Faizullin, D.A., Zuev, Y.F. and Weisel, J.W. (2012) The alpha-helix to beta-sheet transition in stretched and compressed hydrated fibrin clots. Biophys. J. 103, 1020–1027 10.1016/j.bpj.2012.07.046 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Benson, M.D., Liepnieks, J., Uemichi, T., Wheeler, G. and Correa, R. (1993) Hereditary renal amyloidosis associated with a mutant fibrinogen alpha-chain. Nat. Genet. 3, 252–255 10.1038/ng0393-252 [DOI] [PubMed] [Google Scholar]
- 74.Haidinger, M., Werzowa, J., Kain, R., Antlanger, M., Hecking, M., Pfaffenberger, S.et al. (2013) Hereditary amyloidosis caused by R554L fibrinogen aalpha-chain mutation in a spanish family and review of the literature. Amyloid 20, 72–79 10.3109/13506129.2013.781998 [DOI] [PubMed] [Google Scholar]
- 75.Lendrum, A.C., Slidders, W. and Fraser, D.S. (1972) Renal hyalin. A study of amyloidosis and diabetic fibrinous vasculosis with new staining methods. J. Clin. Pathol. 25, 373–396 10.1136/jcp.25.5.373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kranenburg, O., Bouma, B., Kroon-Batenburg, L.M.J., Reijerkerk, A., Wu, Y.P., Voest, E.E.et al. (2002) Tissue-type plasminogen activator is a multiligand cross-beta structure receptor. Curr. Biol. 12, 1833–1839 10.1016/S0960-9822(02)01224-1 [DOI] [PubMed] [Google Scholar]
- 77.Aguzzi, A. and Calella, A.M. (2009) Prions: protein aggregation and infectious diseases. Physiol. Rev. 89, 1105–1152 10.1152/physrev.00006.2009 [DOI] [PubMed] [Google Scholar]
- 78.Silva, C.J., Vazquez-Fernández, E., Onisko, B. and Requena, J.R. (2015) Proteinase K and the structure of PrPSc: the good, the bad and the ugly. Virus Res. 207, 120–126 10.1016/j.virusres.2015.03.008 [DOI] [PubMed] [Google Scholar]
- 79.Wang, F., Wang, X., Abskharon, R. and Ma, J. (2018) Prion infectivity is encoded exclusively within the structure of proteinase K-resistant fragments of synthetically generated recombinant PrP(Sc). Acta Neuropathol. Commun. 6, 30 10.1186/s40478-018-0534-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Fitzpatrick, A.W.P., Falcon, B., He, S., Murzin, A.G., Murshudov, G., Garringer, H.J.et al. (2017) Cryo-EM structures of tau filaments from Alzheimer's disease. Nature 547, 185–190 10.1038/nature23002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Li, D. and Liu, C. (2020) Structural diversity of amyloid fibrils and advances in their structure determination. Biochemistry 59, 639–646 10.1021/acs.biochem.9b01069 [DOI] [PubMed] [Google Scholar]
- 82.Lavatelli, F., di Fonzo, A., Palladini, G. and Merlini, G. (2016) Systemic amyloidoses and proteomics: the state of the art. EuPA Open Proteom. 11, 4–10 10.1016/j.euprot.2016.02.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Linke, R.P. (2012) On typing amyloidosis using immunohistochemistry. Detailled illustrations, review and a note on mass spectrometry. Prog. Histochem. Cytochem. 47, 61–132 10.1016/j.proghi.2012.03.001 [DOI] [PubMed] [Google Scholar]
- 84.Altschul, D.J., Unda, S.R., de La Garza Ramos, R., Zampolin, R., Benton, J., Holland, R.et al. (2020) Hemorrhagic presentations of COVID-19: risk factors for mortality. Clin. Neurol. Neurosurg. 198, 106112 10.1016/j.clineuro.2020.106112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Grobler, C., Maphumulo, S.C., Grobbelaar, L.M., Bredenkamp, J., Laubscher, J., Lourens, P.J.et al. (2020) COVID-19: the rollercoaster of fibrin(ogen), D-dimer, von willebrand factor, P-selectin and their interactions with endothelial cells, platelets and erythrocytes. Int. J. Mol. Sci. 21, 5168 10.3390/ijms21145168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Iba, T., Levy, J.H., Levi, M. and Thachil, J. (2020) Coagulopathy in COVID-19. J. Thromb. Haemost. 18, 2103–2109 10.1111/jth.14975 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Klok, F.A., Kruip, M., van der Meer, N.J.M., Arbous, M.S., Gommers, D., Kant, K.M.et al. (2020) Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: an updated analysis. Thromb. Res. 191, 148–150 10.1016/j.thromres.2020.04.041 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Malas, M.B., Naazie, I.N., Elsayed, N., Mathlouthi, A., Marmor, R. and Clary, B. (2020) Thromboembolism risk of COVID-19 is high and associated with a higher risk of mortality: a systematic review and meta-analysis. EClinicalMedicine 29, 100639 10.1016/j.eclinm.2020.100639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Thachil, J., Tang, N., Gando, S., Falanga, A., Cattaneo, M., Levi, M.et al. (2020) ISTH interim guidance on recognition and management of coagulopathy in COVID-19. J. Thromb. Haemost. 18, 1023–1026 10.1111/jth.14810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Gómez-Mesa, J.E., Galindo-Coral, S., Montes, M.C. and Muñoz Martin, A.J. (2021) Thrombosis and coagulopathy in COVID-19. Curr. Probl. Cardiol. 46, 100742 10.1016/j.cpcardiol.2020.100742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Leentjens, J., van Haaps, T.F., Wessels, P.F., Schutgens, R.E.G. and Middeldorp, S. (2021) COVID-19-associated coagulopathy and antithrombotic agents-lessons after 1 year. Lancet Haematol. 8, e524–e533 10.1016/S2352-3026(21)00105-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Lorini, F.L., Di Matteo, M., Gritti, P., Grazioli, L., Benigni, A., Zacchetti, L.et al. (2021) Coagulopathy and COVID-19. Eur. Heart J. Suppl. 23, E95–E98 10.1093/eurheartj/suab100 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Polimeni, A., Leo, I., Spaccarotella, C., Mongiardo, A., Sorrentino, S., Sabatino, J.et al. (2021) Differences in coagulopathy indices in patients with severe versus non-severe COVID-19: a meta-analysis of 35 studies and 6427 patients. Sci. Rep. 11, 10464 10.1038/s41598-021-89967-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Sarkar, M., Madabhavi, I.V., Quy, P.N. and Govindagoudar, M.B. (2021) COVID-19 and coagulopathy. Clin. Respir. J. 15, 1259–1274 10.1111/crj.13438 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Vincent, J.L., Levi, M. and Hunt, B.J. (2021) Prevention and management of thrombosis in hospitalised patients with COVID-19 pneumonia. Lancet Respir. Med. 10, 214–220 10.1016/S2213-2600(21)00455-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Ortega-Paz, L., Capodanno, D., Montalescot, G. and Angiolillo, D.J. (2021) Coronavirus disease 2019-associated thrombosis and coagulopathy: review of the pathophysiological characteristics and implications for antithrombotic management. J. Am Heart Assoc. 10, e019650. 10.1161/JAHA.120.019650 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Wang, Z., Gao, X., Miao, H., Ma, X. and Ding, R. (2021) Understanding COVID-19-associated coagulopathy: from PIC to SIC or DIC. J Intens. Med. 1, 35–41 10.1016/j.jointm.2021.03.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Douillet, D., Riou, J., Penaloza, A., Moumneh, T., Soulie, C., Savary, D.et al. (2021) Risk of symptomatic venous thromboembolism in mild and moderate COVID-19: a comparison of two prospective European cohorts. Thromb. Res. 208, 4–10 10.1016/j.thromres.2021.10.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Levi, M. and Thachil, J. (2020) Coronavirus disease 2019 coagulopathy: disseminated intravascular coagulation and thrombotic microangiopathy-Either, neither, or both. Semin. Thromb. Hemost. 46, 781–784 10.1055/s-0040-1712156 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Thachil, J. (2021) Lessons learnt from COVID-19 coagulopathy. EJHaem 2, 577–584 10.1002/jha2.228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Venter, C., Bezuidenhout, J.A., Laubscher, G.J., Lourens, P.J., Steenkamp, J., Kell, D.B.et al. (2020) Erythrocyte, platelet, serum ferritin and P-selectin pathophysiology implicated in severe hypercoagulation and vascular complications in COVID-19. Int. J. Mol. Sci. 21, 8234 10.3390/ijms21218234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Fogarty, H., Townsend, L., Ni Cheallaigh, C., Bergin, C., Martin-Loeches, I., Browne, P.et al. (2020) COVID19 coagulopathy in Caucasian patients. Br. J. Haematol. 189, 1044–1049 10.1111/bjh.16749 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Bray, M.A., Sartain, S.E., Gollamudi, J. and Rumbaut, R.E. (2020) Microvascular thrombosis: experimental and clinical implications. Transl. Res. 225, 105–130 10.1016/j.trsl.2020.05.006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Levi, M., Thachil, J., Iba, T. and Levy, J.H. (2020) Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. 7, e438–e440 10.1016/S2352-3026(20)30145-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Willyard, C. (2020) Coronavirus blood-clot mystery intensifies. Nature 581, 250 10.1038/d41586-020-01403-8 [DOI] [PubMed] [Google Scholar]
- 106.Pretorius, E., Venter, C., Laubscher, G.J., Lourens, P.J., Steenkamp, J. and Kell, D.B. (2020) Prevalence of amyloid blood clots in COVID-19 plasma. medRxiv 10.1101/2020.07.28.20163543 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Asakura, H. and Ogawa, H. (2021) COVID-19-associated coagulopathy and disseminated intravascular coagulation. Int. J. Hematol. 113, 45–57 10.1007/s12185-020-03029-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Grobbelaar, L.M., Venter, C., Vlok, M., Ngoepe, M., Laubscher, G.J., Lourens, P.J.et al. (2021) SARS-CoV-2 spike protein S1 induces fibrin(ogen) resistant to fibrinolysis: implications for microclot formation in COVID-19. Biosci. Rep. 41, BSR20210611 10.1042/BSR20210611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Laubscher, G.J., Lourens, P.J., Venter, C., Kell, D.B. and Pretorius, E. (2021) TEG®, microclot and platelet mapping for guiding early management of severe COVID-19 coagulopathy. J. Clin. Med. 10, 5381 10.3390/jcm10225381 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Wygrecka, M., Birnhuber, A., Seeliger, B., Michalick, L., Pak, O., Schultz, A.S.et al. (2021) Altered fibrin clot structure and dysregulated fibrinolysis contribute to thrombosis risk in severe COVID-19. Blood Adv. 6, 1074–1087 10.1182/bloodadvances.2021004816 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Wool, G.D. and Miller, J.L. (2021) The impact of COVID-19 disease on platelets and coagulation. Pathobiology 88, 15–27 10.1159/000512007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Iba, T., Levy, J.H., Connors, J.M., Warkentin, T.E., Thachil, J. and Levi, M. (2020) The unique characteristics of COVID-19 coagulopathy. Crit. Care 24, 360 10.1186/s13054-020-03077-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Loo, J., Spittle, D.A. and Newnham, M. (2021) COVID-19, immunothrombosis and venous thromboembolism: biological mechanisms. Thorax 76, 412–420 10.1136/thoraxjnl-2020-216243 [DOI] [PubMed] [Google Scholar]
- 114.Manolis, A.S., Manolis, T.A., Manolis, A.A., Papatheou, D. and Melita, H. (2021) COVID-19 Infection: viral macro- and micro-Vascular coagulopathy and thromboembolism/Prophylactic and therapeutic management. J. Cardiovasc. Pharmacol. Ther. 26, 12–24 10.1177/1074248420958973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Poor, H.D. (2021) Pulmonary thrombosis and thromboembolism in COVID-19. Chest 160, 1471–1480 10.1016/j.chest.2021.06.016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Pretorius, E., Vlok, M., Venter, C., Bezuidenhout, J.A., Laubscher, G.J., Steenkamp, J.et al. (2021) Persistent clotting protein pathology in long COVID/ post-Acute sequelae of COVID-19 (PASC) is accompanied by increased levels of antiplasmin. Cardiovasc. Diabetol. 20, 172 10.1186/s12933-021-01359-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Zuin, M., Engelen, M.M., Barco, S., Spyropoulos, A.C., Vanassche, T., Hunt, B.J.et al. (2021) Incidence of venous thromboembolic events in COVID-19 patients after hospital discharge: a systematic review and meta-analysis. Thromb. Res. 209, 94–98 10.1016/j.thromres.2021.11.029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Ryu, J.K., Sozmen, E.G.. Dixit, K., Montano, M., Matsui, Y., Liu, Y.et al. (2021) SARS-CoV-2 spike protein induces abnormal inflammatory blood clots neutralized by fibrin immunotherapy. bioRxiv 2021.10.12.464152 10.1101/2021.10.12.464152 [DOI] [Google Scholar]
- 119.Marfella, R., Paolisso, P., Sardu, C., Palomba, L., D'Onofrio, N., Cesaro, A.et al. (2021) SARS-COV-2 colonizes coronary thrombus and impairs heart microcirculation bed in asymptomatic SARS-CoV-2 positive subjects with acute myocardial infarction. Crit. Care 25, 217 10.1186/s13054-021-03643-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Jana, A.K., Greenwood, A.B. and Hansmann, U.H.E. (2021) Presence of a SARS-CoV-2 protein enhances amyloid formation of serum amyloid A. J. Phys. Chem. B 125, 9155–9167 10.1021/acs.jpcb.1c04871 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Itzhaki, R.F., Lathe, R., Balin, B.J., Ball, M.J., Braak, H., Bearer, E.L.et al. (2016) Microbes and Alzheimer's disease. J. Alzheimers Dis. 51, 979–984 10.3233/JAD-160152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Brothers, H.M., Gosztyla, M.L. and Robinson, S.R. (2018) The physiological roles of amyloid-beta peptide hint at New ways to treat Alzheimer's disease. Front. Aging Neurosci. 10, 118 10.3389/fnagi.2018.00118 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Eimer, W.A., Vijaya Kumar, D.K., Navalpur Shanmugam, N.K., Rodriguez, A.S., Mitchell, T., Washicosky, K.J.et al. (2018) Alzheimer's disease-Associated beta-Amyloid Is rapidly seeded by herpesviridae to protect against brain infection. Neuron 99, 56–63.e53 10.1016/j.neuron.2018.06.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Bortolotti, D., Gentili, V., Rotola, A., Caselli, E. and Rizzo, R. (2019) HHV-6A infection induces amyloid-beta expression and activation of microglial cells. Alzheimers Res. Ther. 11, 104 10.1186/s13195-019-0552-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Sait, A., Angeli, C., Doig, A.J. and Day, P.J.R. (2021) Viral involvement in Alzheimer's disease. ACS Chem. Neurosci. 12, 1049–1060 10.1021/acschemneuro.0c00719 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Hansell, D.M., Goldin, J.G., King, Jr, T.E., Lynch, D.A., Richeldi, L. and Wells, A.U. (2015) CT staging and monitoring of fibrotic interstitial lung diseases in clinical practice and treatment trials: a position paper from the fleischner society. Lancet Respir. Med. 3, 483–496 10.1016/S2213-2600(15)00096-X [DOI] [PubMed] [Google Scholar]
- 127.Parekh, M., Donuru, A., Balasubramanya, R. and Kapur, S. (2020) Review of the chest CT differential diagnosis of ground-Glass opacities in the COVID Era. Radiology 297, E289–E302 10.1148/radiol.2020202504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Warman, A., Warman, P., Sharma, A., Parikh, P., Warman, R., Viswanadhan, N.et al. (2020) Interpretable artificial intelligence for COVID-19 diagnosis from chest CT reveals specificity of ground-Glass opacities. medRxiv 10.1101/2020.05.16.20103408 [DOI] [Google Scholar]
- 129.Zhao, W., Zhong, Z., Xie, X., Yu, Q. and Liu, J. (2020) Relation between chest CT findings and clinical conditions of coronavirus disease (COVID-19) pneumonia: a multicenter study. AJR Am. J. Roentgenol. 214, 1072–1077 10.2214/AJR.20.22976 [DOI] [PubMed] [Google Scholar]
- 130.Cozzi, D., Cavigli, E., Moroni, C., Smorchkova, O., Zantonelli, G., Pradella, S.et al. (2021) Ground-glass opacity (GGO): a review of the differential diagnosis in the era of COVID-19. Jpn J. Radiol. 39, 721–732 10.1007/s11604-021-01120-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Roig-Marín, N. and Roig-Rico, P. (2021) Ground-glass opacity on emergency department chest X-ray: a risk factor for in-hospital mortality and organ failure in elderly admitted for COVID-19. Postgrad. Med. 10.1080/00325481.2021.2021741 [DOI] [PubMed] [Google Scholar]
- 132.Wang, D., Shang, Y., Chen, Y., Xia, J., Tian, W., Zhang, T.et al. (2021) Clinical value of COVID-19 chest radiography and high-resolution CT examination. Curr. Med. Imaging 10.2174/1573405618666211229143121 [DOI] [PubMed] [Google Scholar]
- 133.Pickford, H.A., Swensen, S.J. and Utz, J.P. (1997) Thoracic cross-sectional imaging of amyloidosis. AJR Am. J. Roentgenol. 168, 351–355 10.2214/ajr.168.2.9016204 [DOI] [PubMed] [Google Scholar]
- 134.Milani, P., Basset, M., Russo, F., Foli, A., Palladini, G. and Merlini, G. (2017) The lung in amyloidosis. Eur. Respir. Rev. 26, 170046 10.1183/16000617.0046-2017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Liu, Y., Jin, Z., Zhang, H., Zhang, Y., Shi, M., Meng, F.et al. (2018) Diffuse parenchymal pulmonary amyloidosis associated with multiple myeloma: a case report and systematic review of the literature. BMC Cancer 18, 802 10.1186/s12885-018-4565-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Baumgart, J.V., Stuhlmann-Laeisz, C., Hegenbart, U., Nattenmüller, J., Schönland, S., Krüger, S.et al. (2018) Local vs. systemic pulmonary amyloidosis-impact on diagnostics and clinical management. Virchows. Arch. 473, 627–637 10.1007/s00428-018-2442-x [DOI] [PubMed] [Google Scholar]
- 137.Frija-Masson, J., Debray, M.P., Boussouar, S., Khalil, A., Bancal, C., Motiejunaite, J.et al. (2021) Residual ground glass opacities three months after COVID-19 pneumonia correlate to alteration of respiratory function: the post COVID M3 study. Respir. Med. 184, 106435 10.1016/j.rmed.2021.106435 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Zheng, X.L. and Sadler, J.E. (2008) Pathogenesis of thrombotic microangiopathies. Annu. Rev. Pathol. 3, 249–277 10.1146/annurev.pathmechdis.3.121806.154311 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Lopes da Silva, R. (2011) Viral-associated thrombotic microangiopathies. Hematol. Oncol. Stem Cell Ther. 4, 51–59 10.5144/1658-3876.2011.51 [DOI] [PubMed] [Google Scholar]
- 140.Bommer, M., Wolfle-Guter, M., Bohl, S. and Kuchenbauer, F. (2018) The differential diagnosis and treatment of thrombotic microangiopathies. Dtsch. Arztebl. Int. 115, 327–334 10.3238/arztebl.2018.0327 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Fakhouri, F. (2016) Pregnancy-related thrombotic microangiopathies: clues from complement biology. Transfus. Apher. Sci. 54, 199–202 10.1016/j.transci.2016.04.009 [DOI] [PubMed] [Google Scholar]
- 142.Rosove, M.H. (2014) Thrombotic microangiopathies. Semin. Arthritis Rheum. 43, 797–805 10.1016/j.semarthrit.2013.11.004 [DOI] [PubMed] [Google Scholar]
- 143.Magnusson, K., Simon, R., Sjölander, D., Sigurdson, C.J., Hammarström, P. and Nilsson, K.P.R. (2014) Multimodal fluorescence microscopy of prion strain specific PrP deposits stained by thiophene-based amyloid ligands. Prion 8, 319–329 10.4161/pri.29239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Magnusson, K., Appelqvist, H., Cieslar-Pobuda, A., Back, M., Kågedal, B., Jonasson, J.A.et al. (2015) An imidazole functionalized pentameric thiophene displays different staining patterns in normal and malignant cells. Front. Chem. 3, 58 10.3389/fchem.2015.00058 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Wegenast-Braun, B.M., Skodras, A., Bayraktar, G., Mahler, J., Fritschi, S.K., Klingstedt, T.et al. (2012) Spectral discrimination of cerebral amyloid lesions after peripheral application of luminescent conjugated oligothiophenes. Am. J. Pathol. 181, 1953–1960 10.1016/j.ajpath.2012.08.031 [DOI] [PubMed] [Google Scholar]
- 146.Düz, M.E., Balci, A. and Menekşe, E. (2020) D-dimer levels and COVID-19 severity: systematic review and meta-Analysis. Tuberk. Toraks. 68, 353–360 10.5578/tt.70351 [DOI] [PubMed] [Google Scholar]
- 147.Favaloro, E.J. and Thachil, J. (2020) Reporting of D-dimer data in COVID-19: some confusion and potential for misinformation. Clin. Chem. Lab. Med. 58, 1191–1199 10.1515/cclm-2020-0573 [DOI] [PubMed] [Google Scholar]
- 148.Gerber, G.F. and Chaturvedi, S. (2021) How to recognize and manage COVID-19-associated coagulopathy. Hematol. Am. Soc. Hematol. Educ. Program 2021, 614–620 10.1182/hematology.2021000297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Mehrdad, R., Zahra, K. and Mansouritorghabeh, H. (2021) Hemostatic system (Fibrinogen level, D-Dimer, and FDP) in severe and Non-Severe patients With COVID-19: a systematic review and meta-Analysis. Clin. Appl. Thromb. Hemost. 27, 10760296211010973 10.1177/10760296211010973 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Paliogiannis, P., Mangoni, A.A., Dettori, P., Nasrallah, G.K., Pintus, G. and Zinellu, A. (2020) D-Dimer concentrations and COVID-19 severity: a systematic review and meta-Analysis. Front. Public Health 8, 432 10.3389/fpubh.2020.00432 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Poudel, A., Poudel, Y., Adhikari, A., Aryal, B.B., Dangol, D., Bajracharya, T.et al. (2021) D-dimer as a biomarker for assessment of COVID-19 prognosis: D-dimer levels on admission and its role in predicting disease outcome in hospitalized patients with COVID-19. PLoS ONE 16, e0256744 10.1371/journal.pone.0256744 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Rostami, M. and Mansouritorghabeh, H. (2020) D-dimer level in COVID-19 infection: a systematic review. Expert Rev. Hematol. 13, 1265–1275 10.1080/17474086.2020.1831383 [DOI] [PubMed] [Google Scholar]
- 153.Shah, S., Shah, K., Patel, S.B., Patel, F.S., Osman, M., Velagapudi, P.et al. (2020) Elevated D-Dimer levels are associated with increased risk of mortality in COVID-19: a systematic review and meta-Analysis. Cardiol. Rev. 28, 295–302 10.1097/CRD.0000000000000330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Wang, Q., Cheng, J., Shang, J., Wang, Y., Wan, J., Yan, Y.Q.et al. (2021) Clinical value of laboratory indicators for predicting disease progression and death in patients with COVID-19: a retrospective cohort study. BMJ Open 11, e043790 10.1136/bmjopen-2020-043790 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Kupferschmidt, K. (2021) Startling new variant raises urgent questions. Science 374, 1178–1180 10.1126/science.acx9737 [DOI] [PubMed] [Google Scholar]
- 156.Currin, A., Swainston, N., Day, P.J. and Kell, D.B. (2015) Synthetic biology for the directed evolution of protein biocatalysts: navigating sequence space intelligently. Chem. Soc. Rev. 44, 1172–1239 10.1039/C4CS00351A [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Townsend, L., Fogarty, H., Dyer, A., Martin-Loeches, I., Bannan, C., Nadarajan, P.et al. (2021) Prolonged elevation of D-dimer levels in convalescent COVID-19 patients is independent of the acute phase response. J. Thromb. Haemost. 19, 1064–1070 10.1111/jth.15267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Perry, D.J. (2021) A pratial guide to haemostasis
- 159.Weil, I.A., Seicean, S., Neuhauser, D., Schiltz, N.K. and Seicean, A. (2015) Use and utility of hemostatic screening in adults undergoing elective, non-cardiac surgery. PLoS ONE 10, e0139139 10.1371/journal.pone.0139139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Loizou, E., Mayhew, D.J., Martlew, V. and Murthy, B.V.S. (2018) Implications of deranged activated partial thromboplastin time for anaesthesia and surgery. Anaesthesia 73, 1557–1563 10.1111/anae.14344 [DOI] [PubMed] [Google Scholar]
- 161.Chitlur, M., Sorensen, B., Rivard, G.E., Young, G., Ingerslev, J., Othman, M.et al. (2011) Standardization of thromboelastography: a report from the TEG-ROTEM working group. Haemophilia 17, 532–537 10.1111/j.1365-2516.2010.02451.x [DOI] [PubMed] [Google Scholar]
- 162.Nielsen, V.G. and Pretorius, E. (2014) Iron-enhanced coagulation is attenuated by chelation: thrombelastographic and ultrastructural analysis. Blood Coagul. Fibrinol. 25, 845–850 10.1097/MBC.0000000000000160 [DOI] [PubMed] [Google Scholar]
- 163.de Villiers, S., Bester, J., Kell, D.B. and Pretorius, E. (2019) Erythrocyte health and the possible role of amyloidogenic blood clotting in the evolving haemodynamics of female migraine-with-aura pathophysiology: results from a pilot study. Front. Neurol. 10, 1262 10.3389/fneur.2019.01262 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Kell, D.B. and Pretorius, E. (2015) On the translocation of bacteria and their lipopolysaccharides between blood and peripheral locations in chronic, inflammatory diseases: the central roles of LPS and LPS-induced cell death. Integr. Biol. 7, 1339–1377 10.1039/c5ib00158g [DOI] [PubMed] [Google Scholar]
- 165.Erdoes, G., Schloer, H., Eberle, B. and Nagler, M. (2018) Next generation viscoelasticity assays in cardiothoracic surgery: feasibility of the TEG6s system. PLoS ONE 13, e0209360 10.1371/journal.pone.0209360 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Da Luz, L.T., Nascimento, B., Shankarakutty, A.K., Rizoli, S. and Adhikari, N.K. (2014) Effect of thromboelastography (TEG(R)) and rotational thromboelastometry (ROTEM(R)) on diagnosis of coagulopathy, transfusion guidance and mortality in trauma: descriptive systematic review. Crit. Care 18, 518 10.1186/s13054-014-0518-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Franz, R.C. (2009) ROTEM analysis: a significant advance in the field of rotational thrombelastography. S. Afr. J. Surg. 47, 2–6 [PubMed] [Google Scholar]
- 168.Jackson, G.N.B., Ashpole, K.J. and Yentis, S.M. (2009) The TEG((R)) vs the ROTEM((R)) thromboelastography/thromboelastometry systems. Anaesthesia 64, 212–215 10.1111/j.1365-2044.2008.05752.x [DOI] [PubMed] [Google Scholar]
- 169.Lang, T., Bauters, A., Braun, S.L., Potzsch, B., von Pape, K.W., Kolde, H.J.et al. (2005) Multi-centre investigation on reference ranges for ROTEM thromboelastometry. Blood Coagul. Fibrinol. 16, 301–310 10.1097/01.mbc.0000169225.31173.19 [DOI] [PubMed] [Google Scholar]
- 170.Nielsen, V.G. (2007) A comparison of the Thrombelastograph and the ROTEM. Blood Coagul. Fibrinol. 18, 247–252 10.1097/MBC.0b013e328092ee05 [DOI] [PubMed] [Google Scholar]
- 171.Tanaka, K.A., Bolliger, D., Vadlamudi, R. and Nimmo, A. (2012) Rotational thromboelastometry (ROTEM)-based coagulation management in cardiac surgery and major trauma. J. Cardiothorac. Vasc. Anesth. 26, 1083–1093 10.1053/j.jvca.2012.06.015 [DOI] [PubMed] [Google Scholar]
- 172.Theusinger, O.M., Schroder, C.M., Eismon, J., Emmert, M.Y., Seifert, B., Spahn, D.R.et al. (2013) The influence of laboratory coagulation tests and clotting factor levels on Rotation Thromboelastometry (ROTEM(R)) during major surgery with hemorrhage. Anesth. Analg. 117, 314–321 10.1213/ANE.0b013e31829569ac [DOI] [PubMed] [Google Scholar]
- 173.Wikkelsø, A., Wetterslev, J., Møller, A.M. and Afshari, A. (2016) Thromboelastography (TEG) or thromboelastometry (ROTEM) to monitor haemostatic treatment versus usual care in adults or children with bleeding. Cochrane Database Syst. Rev. 2016, CD007871 10.1002/14651858.CD007871.pub3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Smith, J.F., Knowles, T.P., Dobson, C.M., Macphee, C.E. and Welland, M.E. (2006) Characterization of the nanoscale properties of individual amyloid fibrils. Proc. Natl Acad. Sci. U.S.A. 103, 15806–15811 10.1073/pnas.0604035103 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Knowles, T.P., Fitzpatrick, A.W., Meehan, S., Mott, H.R., Vendruscolo, M., Dobson, C.M.et al. (2007) Role of intermolecular forces in defining material properties of protein nanofibrils. Science 318, 1900–1903 10.1126/science.1150057 [DOI] [PubMed] [Google Scholar]
- 176.Paul, T.J., Hoffmann, Z., Wang, C., Shanmugasundaram, M., DeJoannis, J., Shekhtman, A.et al. (2016) Structural and mechanical properties of amyloid beta fibrils: a combined experimental and theoretical approach. J. Phys. Chem. Lett. 7, 2758–2764 10.1021/acs.jpclett.6b01066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Lamour, G., Nassar, R., Chan, P.H.W., Bozkurt, G., Li, J., Bui, J.M.et al. (2017) Mapping the broad structural and mechanical properties of amyloid fibrils. Biophys. J. 112, 584–594 10.1016/j.bpj.2016.12.036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Yoon, G., Lee, M., Kim, K., Kim, J.I., Chang, H.J., Baek, I.et al. (2015) Morphology and mechanical properties of multi-stranded amyloid fibrils probed by atomistic and coarse-grained simulations. Phys. Biol. 12, 066021 10.1088/1478-3975/12/6/066021 [DOI] [PubMed] [Google Scholar]
- 179.Bradford Hill, A. (1965) Environment and disease: association or causation? Proc. R. Soc. Med. 58, 295–300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Fanaroff, A.C., Garcia, S. and Giri, J. (2021) Myocardial infarction during the COVID-19 pandemic. JAMA 326, 1916–1918 10.1001/jama.2021.19608 [DOI] [PubMed] [Google Scholar]
- 181.Katsoularis, I., Fonseca-Rodríguez, O., Farrington, P., Lindmark, K. and Fors Connolly, A.M. (2021) Risk of acute myocardial infarction and ischaemic stroke following COVID-19 in Sweden: a self-controlled case series and matched cohort study. Lancet 398, 599–607 10.1016/S0140-6736(21)00896-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Ntaios, G., Michel, P., Georgiopoulos, G., Guo, Y., Li, W., Xiong, J.et al. (2020) Characteristics and outcomes in patients With COVID-19 and acute ischemic stroke: the global COVID-19 stroke registry. Stroke 51, e254–e258 10.1161/STROKEAHA.119.026907 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 183.Perry, R.J., Smith, C.J., Roffe, C., Simister, R., Narayanamoorthi, S., Marigold, R.et al. (2021) Characteristics and outcomes of COVID-19 associated stroke: a UK multicentre case-control study. J. Neurol. Neurosurg. Psychiatry 92, 242–248 10.1136/jnnp-2020-324927 [DOI] [PubMed] [Google Scholar]
- 184.Bowe, B., Xie, Y., Xu, E. and Al-Aly, Z. (2021) Kidney outcomes in long COVID. J. Am. Soc. Nephrol. 32, 2851–2862 10.1681/ASN.2021060734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Yende, S. and Parikh, C.R. (2021) Long COVID and kidney disease. Nat. Rev. Nephrol. 17, 792–793 10.1038/s41581-021-00487-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Raj, S.R., Arnold, A.C., Barboi, A., Claydon, V.E., Limberg, J.K., Lucci, V.M.et al. (2021) Long-COVID postural tachycardia syndrome: an American autonomic society statement. Clin. Auton. Res. 31, 365–368 10.1007/s10286-021-00798-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Agergaard, J., Leth, S., Pedersen, T.H., Harbo, T., Blicher, J.U., Karlsson, P.et al. (2021) Myopathic changes in patients with long-term fatigue after COVID-19. Clin. Neurophysiol. 132, 1974–1981 10.1016/j.clinph.2021.04.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Soares, M.N., Eggelbusch, M., Naddaf, E., Gerrits, K.H.L., van der Schaaf, M., van den Borst, B.et al. (2021) Skeletal muscle alterations in patients with acute COVID-19 and post-acute sequelae of COVID-19. J. Cachexia Sarcopenia Muscle 13, 11–22 10.1002/jcsm.12896 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Huang, C., Huang, L., Wang, Y., Li, X., Ren, L., Gu, X.et al. (2021) 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet 397, 220–232 10.1016/S0140-6736(20)32656-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Vassiliou, A.G., Jahaj, E., Ilias, I., Markaki, V., Malachias, S., Vrettou, C.et al. (2020) Lactate kinetics reflect organ dysfunction and Are associated with adverse outcomes in intensive care unit patients with COVID-19 pneumonia: preliminary results from a GREEK single-Centre study. Metabolites 10, 386 10.3390/metabo10100386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Nechipurenko, Y.D., Semyonov, D.A., Lavrinenko, I.A., Lagutkin, D.A., Generalov, E.A., Zaitceva, A.Y.et al. (2021) The role of acidosis in the pathogenesis of severe forms of COVID-19. Biology (Basel) 10, 852 10.3390/biology10090852 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Roberts, I., Wright Muelas, M., Taylor, J.M., Davison, A.S., Xu, Y., Grixti, J.M.et al. (2022) Untargeted metabolomics of COVID-19 patient serum reveals potential prognostic markers of both severity and outcome. Metabolomics 18, 6 10.1007/s11306-021-01859-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Robbins, T., Gonevski, M., Clark, C., Baitule, S., Sharma, K., Magar, A.et al. (2021) Hyperbaric oxygen therapy for the treatment of long COVID: early evaluation of a highly promising intervention. Clin. Med. (Lond) 21, e629–e632 10.7861/clinmed.2021-0462 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Chen, G.F., Xu, T.H., Yan, Y., Zhou, Y.R., Jiang, Y., Melcher, K.et al. (2017) Amyloid beta: structure, biology and structure-based therapeutic development. Acta Pharmacol. Sin. 38, 1205–1235 10.1038/aps.2017.28 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Drabik, D., Chodaczek, G. and Kraszewski, S. (2020) Effect of amyloid-beta monomers on lipid membrane mechanical parameters-Potential implications for mechanically driven neurodegeneration in Alzheimer's disease. Int. J. Mol. Sci. 22, 18 10.3390/ijms22010018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 196.Milanesi, L., Sheynis, T., Xue, W.F., Orlova, E.V., Hellewell, A.L., Jelinek, R.et al. (2012) Direct three-dimensional visualization of membrane disruption by amyloid fibrils. Proc. Natl Acad. Sci. U.S.A. 109, 20455–20460 10.1073/pnas.1206325109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Fusco, G., Chen, S.W., Williamson, P.T.F., Cascella, R., Perni, M., Jarvis, J.A.et al. (2017) Structural basis of membrane disruption and cellular toxicity by alpha-synuclein oligomers. Science. 358, 1440–1443 10.1126/science.aan6160 [DOI] [PubMed] [Google Scholar]
- 198.Scheckel, C. and Aguzzi, A. (2018) Prions, prionoids and protein misfolding disorders. Nat. Rev. Genet. 19, 405–418 10.1038/s41576-018-0011-4 [DOI] [PubMed] [Google Scholar]
- 199.Kell, D.B. and Knowles, J.D. (2006) The role of modeling in systems biology. In System Modeling in Cellular Biology: From Concepts to Nuts and Bolts (Szallasi, Z., Stelling, J. and Periwal, V., eds). pp. 3–18, MIT Press, Cambridge [Google Scholar]
- 200.Kell, D.B. and Pretorius, E. (2018) No effects without causes. The iron dysregulation and dormant microbes hypothesis for chronic, inflammatory diseases. Biol. Rev. 93, 1518–1557 10.1111/brv.12407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Stein, S.R., Ramelli, S.C., Grazioli, A., Chung, J.-Y., Singh, M., Yinda, C.K.et al. (2021) SARS-CoV-2 infection and persistence throughout the human body and brain. Res. Square 10.21203/rs.3.rs-1139035/v1 [DOI] [Google Scholar]
- 202.Patterson, B.K., Francisco, E.B., Yogendra, R., Long, E., Pise, A., Rodrigues, H.et al. (2022) Persistence of SARS CoV-2 S1 protein in CD16+ monocytes in post-Acute sequelae of COVID-19 (PASC) Up to 15 months post-Infection. Front. Immunol. 12, 746021 10.3389/fimmu.2021.746021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Ebringer, A., Rashid, T. and Wilson, C. (2010) Rheumatoid arthritis, proteus, anti-CCP antibodies and karl popper. Autoimmun. Rev. 9, 216–223 10.1016/j.autrev.2009.10.006 [DOI] [PubMed] [Google Scholar]
- 204.Ebringer, A. (2012) Rheumatoid Arthritis and Proteus, Springer, London [Google Scholar]
- 205.Leonardi, A.J. and Proenca, R.B. (2020) Akt-Fas to quell aberrant T cell differentiation and apoptosis in COVID-19. Front. Immunol. 11, 600405 10.3389/fimmu.2020.600405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Woodruff, M.C., Ramonell, R.P., Lee, F.E.-H. and Sanz, I. (2020) Clinically identifiable autoreactivity is common in severe SARS-CoV-2 infection. medRxiv 10.1101/2020.10.21.20216192 [DOI] [Google Scholar]
- 207.Phetsouphanh, C., Darley, D.R., Wilson, D.B., Howe, A., Munier, C.M.L., Patel, S.K.et al. (2022) Immunological dysfunction persists for 8 months following initial mild-to-moderate SARS-CoV-2 infection. Nat. Immunol. 23, 210–216 10.1038/s41590-021-01113-x [DOI] [PubMed] [Google Scholar]
- 208.Guntas, G., Mansell, T.J., Kim, J.R. and Ostermeier, M. (2005) Directed evolution of protein switches and their application to the creation of ligand-binding proteins. Proc. Natl Acad. Sci. U.S.A. 102, 11224–11229 10.1073/pnas.0502673102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Vizcaychipi, M.P., Shovlin, C.L., McCarthy, A., Howard, A., Brown, A., Hayes, M.et al. (2020) Development and implementation of a COVID-19 near real-time traffic light system in an acute hospital setting. Emerg. Med. J. 37, 630–636 10.1136/emermed-2020-210199 [DOI] [PubMed] [Google Scholar]
- 210.Stewart, S., Newson, L., Briggs, T.A., Grammatopoulos, D., Young, L. and Gill, P. (2021) Long COVID risk - a signal to address sex hormones and women's health. Lancet Reg. Health Eur. 11, 100242 10.1016/j.lanepe.2021.100242 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Swanepoel, A.C., Lindeque, B.G., Swart, P.J., Abdool, Z. and Pretorius, E. (2014) Ultrastructural changes of fibrin networks during three phases of pregnancy: a qualitative investigation. Microsc. Res. Tech. 77, 602–608 10.1002/jemt.22385 [DOI] [PubMed] [Google Scholar]
- 212.Pipe, S.W., Kaczmarek, R., Srivastava, A., Pierce, G.F., Makris, M. and Hermans, C. and Interim Guidance Coagulation Products Safety Supply Access Committee of the World Federation of H. (2021) Management of COVID-19-associated coagulopathy in persons with haemophilia. Haemophilia 27, 41–48 10.1111/hae.14191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Pretorius, E., Lipinski, B., Bester, J., Vermeulen, N. and Soma, P. (2013) Albumin stabilizes fibrin fiber ultrastructure in low serum albumin type 2 diabetes. Ultrastruct. Pathol. 37, 254–257 10.3109/01913123.2013.778929 [DOI] [PubMed] [Google Scholar]
- 214.Pretorius, E., Mbotwe, S. and Kell, D.B. (2017) Lipopolysaccharide-binding protein (LBP) reverses the amyloid state of fibrin seen in plasma of type 2 diabetics with cardiovascular comorbidities. Sci. Rep. 7, 9680 10.1038/s41598-017-09860-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Cooper, G.J.S., Leighton, B., Dimitriadis, G.D., Parrybillings, M., Kowalchuk, J.M., Howland, K.et al. (1988) Amylin found in amyloid deposits in human type-2 diabetes-Mellitus May Be a hormone that regulates glycogen-Metabolism in skeletal-Muscle. Proc. Natl Acad. Sci. U.S.A. 85, 7763–7766 10.1073/pnas.85.20.7763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 216.Roberts, A.N., Leighton, B., Todd, J.A., Cockburn, D., Schofield, P.N., Sutton, R.et al. (1989) Molecular and functional characterization of amylin, a peptide associated with type 2 diabetes mellitus. Proc. Natl Acad. Sci. U.S.A. 86, 9662–9666 10.1073/pnas.86.24.9662 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Konarkowska, B., Aitken, J.F., Kistler, J., Zhang, S. and Cooper, G.J.S. (2006) The aggregation potential of human amylin determines its cytotoxicity towards islet beta-cells. FEBS J. 273, 3614–3624 10.1111/j.1742-4658.2006.05367.x [DOI] [PubMed] [Google Scholar]
- 218.Seet, R.C.S., Quek, A.M.L. and Lim, E.C.H. (2007) Post-infectious fatigue syndrome in dengue infection. J. Clin. Virol. 38, 1–6 10.1016/j.jcv.2006.10.011 [DOI] [PubMed] [Google Scholar]
- 219.Hung, T.M., Wills, B., Clapham, H.E., Yacoub, S. and Turner, H.C. (2019) The uncertainty surrounding the burden of post-acute consequences of dengue infection. Trends Parasitol. 35, 673–676 10.1016/j.pt.2019.06.004 [DOI] [PubMed] [Google Scholar]
- 220.Umakanth, M. (2018) Post dengue fatigue syndrome (PDFS) among dengue IgM-Antibody positive patients at batticaloa teaching hospital, Sri Lanka. Open Acc. Lib. J. 5, e4798 10.4236/oalib.1104798 [DOI] [Google Scholar]
- 221.Bond, N.G., Grant, D.S., Himmelfarb, S.T., Engel, E.J., Al-Hasan, F., Gbakie, M.et al. (2021) Post-Ebola syndrome presents With multiple overlapping symptom clusters: evidence from an ongoing cohort study in eastern Sierra Leone. Clin. Infect. Dis. 73, 1046–1054 10.1093/cid/ciab267 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Burki, T.K. (2016) Post-Ebola syndrome. Lancet Infect. Dis. 16, 780–781 10.1016/S1473-3099(15)00259-5 [DOI] [PubMed] [Google Scholar]
- 223.Scott, J.T., Sesay, F.R., Massaquoi, T.A., Idriss, B.R., Sahr, F. and Semple, M.G. (2016) Post-Ebola syndrome, Sierra Leone. Emerg. Infect. Dis. 22, 641–646 10.3201/eid2204.151302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Rebman, A.W. and Aucott, J.N. (2020) Post-treatment lyme disease as a model for persistent symptoms in lyme disease. Front. Med. (Lausanne) 7, 57 10.3389/fmed.2020.00057 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Potgieter, M., Bester, J., Kell, D.B. and Pretorius, E. (2015) The dormant blood microbiome in chronic, inflammatory diseases. FEMS Microbiol. Rev. 39, 567–591 10.1093/femsre/fuv013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Bots, M.L., Breteler, M.M., van Kooten, F., Haverkate, F., Meijer, P., Koudstaal, P.J.et al. (1998) Coagulation and fibrinolysis markers and risk of dementia. The Dutch vascular factors in dementia study. Haemostasis 28, 216–222 [DOI] [PubMed] [Google Scholar]
- 227.Cortes-Canteli, M., Paul, J., Norris, E.H., Bronstein, R., Ahn, H.J., Zamolodchikov, D.et al. (2010) Fibrinogen and beta-amyloid association alters thrombosis and fibrinolysis: a possible contributing factor to Alzheimer's disease. Neuron 66, 695–709 10.1016/j.neuron.2010.05.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Wong, T.L. and Weitzer, D.J. (2021) Long COVID and myalgic encephalomyelitis/Chronic fatigue syndrome (ME/CFS)-A systemic review and comparison of clinical presentation and symptomatology. Medicina (Kaunas) 57, 418 10.3390/medicina57050418 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Berg, D., Berg, L.H., Couvaras, J. and Harrison, H. (1999) Chronic fatigue syndrome and/or fibromyalgia as a variation of antiphospholipid antibody syndrome: an explanatory model and approach to laboratory diagnosis. Blood Coagul. Fibrinol. 10, 435–438 10.1097/00001721-199910000-00006 [DOI] [PubMed] [Google Scholar]
- 230.Hannan, K.L., Berg, D.E., Baumzweiger, W., Harrison, H.H., Berg, L.H., Ramirez, R.et al. (2000) Activation of the coagulation system in gulf War illness: a potential pathophysiologic link with chronic fatigue syndrome. A laboratory approach to diagnosis. Blood Coagul. Fibrinol. 11, 673–678 10.1097/00001721-200010000-00013 [DOI] [PubMed] [Google Scholar]
- 231.Pretorius, E., Venter, C., Laubsher, G.J., Kotze, M.J., Moremi, K., Oladejo, S.et al. (2021) Combined triple treatment of fibrin amyloid microclots and platelet pathology in individuals with long COVID/Post-Acute sequelae of COVID-19 (PASC) can resolve their persistent symptoms. Res. Square 10.21203/rs.3.rs-1205453/v1 [DOI] [Google Scholar]
- 232.Barrett, T.J., Cornwell, M., Myndzar, K., Rolling, C.C., Xia, Y., Drenkova, K.et al. (2021) Platelets amplify endotheliopathy in COVID-19. Sci. Adv. 7, eabh2434 10.1126/sciadv.abh2434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Barrett, T.J., Bilaloglu, S., Cornwell, M., Burgess, H.M., Virginio, V.W., Drenkova, K.et al. (2021) Platelets contribute to disease severity in COVID-19. J. Thromb. Haemost. 19, 3139–3153 10.1111/jth.15534 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Nishikawa, M., Kanno, H., Zhou, Y., Xiao, T.H., Suzuki, T., Ibayashi, Y.et al. (2021) Massive image-based single-cell profiling reveals high levels of circulating platelet aggregates in patients with COVID-19. Nat. Commun. 12, 7135 10.1038/s41467-021-27378-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Zhang, S., Liu, Y., Wang, X., Yang, L., Li, H., Wang, Y.et al. (2020) SARS-CoV-2 binds platelet ACE2 to enhance thrombosis in COVID-19. J. Hematol. Oncol. 13, 120 10.1186/s13045-020-00954-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Chang, J.C. (2018) Disseminated intravascular coagulation: is it fact or fancy? Blood Coagul. Fibrinolysis 29, 330–337 10.1097/MBC.0000000000000727 [DOI] [PubMed] [Google Scholar]
- 237.Perricone, C., Bartoloni, E., Bursi, R., Cafaro, G., Guidelli, G.M., Shoenfeld, Y.et al. (2020) COVID-19 as part of the hyperferritinemic syndromes: the role of iron depletion therapy. Immunol. Res. 68, 213–224 10.1007/s12026-020-09145-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238.Vargas-Vargas, M. and Cortés-Rojo, C. (2020) Ferritin levels and COVID-19. Rev. Panam. Salud. Publica 44, e72 10.26633/RPSP.2020.72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Kell, D.B. and Pretorius, E. (2014) Serum ferritin is an important disease marker, and is mainly a leakage product from damaged cells. Metallomics 6, 748–773 10.1039/C3MT00347G [DOI] [PubMed] [Google Scholar]
- 240.Kell, D.B. (2009) Iron behaving badly: inappropriate iron chelation as a major contributor to the aetiology of vascular and other progressive inflammatory and degenerative diseases. BMC Med. Genom. 2, 2 10.1186/1755-8794-2-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 241.Kell, D.B. (2010) Towards a unifying, systems biology understanding of large-scale cellular death and destruction caused by poorly liganded iron: Parkinson's, huntington's, Alzheimer's, prions, bactericides, chemical toxicology and others as examples. Arch. Toxicol. 577, 825–889 10.1007/s00204-010-0577-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Cavezzi, A., Troiani, E. and Corrao, S. (2020) COVID-19: hemoglobin, iron, and hypoxia beyond inflammation. A narrative review. Clin. Pract. 10, 1271 10.4081/cp.2020.1271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Edeas, M., Saleh, J. and Peyssonnaux, C. (2020) Iron: innocent bystander or vicious culprit in COVID-19 pathogenesis? Int. J. Infect. Dis. 97, 303–305 10.1016/j.ijid.2020.05.110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.McLaughlin, K.-M., Bechtel, M., Bojkova, D., Münch, C., Ciesek, S., Wass, M.N.et al. (2020) COVID-19-Related coagulopathy—Is transferrin a missing link? Diagnostics 10, 539 10.3390/diagnostics10080539 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Chakurkar, V., Rajapurkar, M., Lele, S., Mukhopadhyay, B., Lobo, V., Injarapu, R.et al. (2021) Increased serum catalytic iron may mediate tissue injury and death in patients with COVID-19. Sci. Rep. 11, 19618 10.1038/s41598-021-99142-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Liu, W. and Li, H. (2022) COVID-19: attacks the 1-beta chain of hemoglobin to disrupt respiratory function and escape immunity. chemRxiv 10.26434/chemrxiv-2021-dtpv3-v [DOI] [Google Scholar]
- 247.Campione, E., Cosio, T., Rosa, L., Lanna, C., Di Girolamo, S., Gaziano, R.et al. (2020) Lactoferrin as protective natural barrier of respiratory and intestinal mucosa against coronavirus infection and inflammation. Int. J. Mol. Sci. 21, 4903 10.3390/ijms21144903 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Campione, E., Lanna, C., Cosio, T., Rosa, L., Conte, M.P., Iacovelli, F.et al. (2020) Pleiotropic effect of Lactoferrin in the prevention and treatment of COVID-19 infection: in vivo, in silico and in vitro preliminary evidences. bioRxiv 2020.08.11.244996 10.1101/2020.08.11.244996 [DOI] [Google Scholar]
- 249.Dalamaga, M., Karampela, I. and Mantzoros, C.S. (2020) Commentary: could iron chelators prove to be useful as an adjunct to COVID-19 treatment regimens? Metabolism 108, 154260 10.1016/j.metabol.2020.154260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250.Kell, D.B., Heyden, E.L. and Pretorius, E. (2020) The biology of lactoferrin, an iron-binding protein that can help defend against viruses and bacteria. Front. Immunol. 11, 1221 10.3389/fimmu.2020.01221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251.Abobaker, A. (2020) Can iron chelation as an adjunct treatment of COVID-19 improve the clinical outcome? Eur. J. Clin. Pharmacol. 76, 1619–1620 10.1007/s00228-020-02942-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Carota, G., Ronsisvalle, S., Panarello, F., Tibullo, D., Nicolosi, A. and Li Volti, G. (2021) Role of iron chelation and protease inhibition of natural products on COVID-19 infection. J. Clin. Med. 10, 2306 10.3390/jcm10112306 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Naseef, P.P., Elayadeth-Meethal, M., Salim, K.T.M., Anjana, A., Muhas, C., Vajid, K.A.et al. (2022) Therapeutic potential of induced iron depletion using iron chelators in COVID-19. Saudi J. Biol. Sci. 10.1016/j.sjbs.2021.11.061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254.Vlahakos, V.D., Marathias, K.P., Arkadopoulos, N. and Vlahakos, D.V. (2021) Hyperferritinemia in patients with COVID-19: an opportunity for iron chelation? Artif. Organs. 45, 163–167 10.1111/aor.13812 [DOI] [PubMed] [Google Scholar]
- 255.Fogarty, H., Townsend, L., Morrin, H., Ahmad, A., Comerford, C., Karampini, E.et al. (2021) Persistent endotheliopathy in the pathogenesis of long COVID syndrome. J. Thromb. Haemost. 19, 2546–2553 10.1111/jth.15490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256.Goshua, G., Pine, A.B., Meizlish, M.L., Chang, C.H., Zhang, H., Bahel, P.et al. (2020) Endotheliopathy in COVID-19-associated coagulopathy: evidence from a single-centre, cross-sectional study. Lancet Haematol. 7, e575–e582 10.1016/S2352-3026(20)30216-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Shovlin, C.L. and Vizcaychipi, M.P. (2020) Vascular inflammation and endothelial injury in SARS-CoV-2 infection: the overlooked regulatory cascades implicated by the ACE2 gene cluster. QJM 10.1093/qjmed/hcaa241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258.Gremmel, T., Frelinger, III, A.L. and Michelson, A.D. (2016) Platelet physiology. Semin Thromb Hemost. 42, 191–204 10.1055/s-0035-1564835 [DOI] [PubMed] [Google Scholar]
- 259.Page, M.J. and Pretorius, E. (2020) A champion of host defense: a generic large-Scale cause for platelet dysfunction and depletion in infection. Semin. Thromb. Hemost. 46, 302–319 10.1055/s-0040-1708827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 260.Rubenstein, D.A. and Yin, W. (2018) Platelet-Activation mechanisms and vascular remodeling. Compr. Physiol. 8, 1117–1156 10.1002/cphy.c170049 [DOI] [PubMed] [Google Scholar]
- 261.Wang, J., Hajizadeh, N., Moore, E.E., McIntyre, R.C., Moore, P.K., Veress, L.A.et al. (2020) Tissue plasminogen activator (tPA) treatment for COVID-19 associated acute respiratory distress syndrome (ARDS): a case series. J. Thromb. Haemost. 18, 1752–1755 10.1111/jth.14828 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.ATTACC Investigators, ACTIV-4a Investigators, REMAP-CAP Investigators, Lawler, P.R., Goligher, E.C., Berger, J.S.et al. (2021) Therapeutic anticoagulation with heparin in noncritically Ill patients with COVID-19. N. Engl. J. Med. 385, 790–802 10.1056/NEJMoa2105911 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 263.Mycroft-West, C.J., Su, D., Pagani, I., Rudd, T.R., Elli, S., Gandhi, N.S.et al. (2020) Heparin inhibits cellular invasion by SARS-CoV-2: structural dependence of the interaction of the spike S1 receptor-Binding domain with heparin. Thromb. Haemost. 120, 1700–1715 10.1055/s-0040-1721319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Tandon, R., Sharp, J.S., Zhang, F., Pomin, V.H., Ashpole, N.M., Mitra, D.et al. (2021) Effective inhibition of SARS-CoV-2 entry by heparin and enoxaparin derivatives. J. Virol. 95, e01987-20 10.1128/JVI.01987-20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265.Tree, J.A., Turnbull, J.E., Buttigieg, K.R., Elmore, M.J., Coombes, N., Hogwood, J.et al. (2021) Unfractionated heparin inhibits live wild type SARS-CoV-2 cell infectivity at therapeutically relevant concentrations. Br. J. Pharmacol. 178, 626–635 10.1111/bph.15304 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Paiardi, G., Richter, S., Oreste, P., Urbinati, C., Rusnati, M. and Wade, R.C. (2022) The binding of heparin to spike glycoprotein inhibits SARS-CoV-2 infection by three mechanisms. J. Biol. Chem. 298, 101507 10.1016/j.jbc.2021.101507 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267.Lopes, R.D., de Barros, E.S.P.G.M., Furtado, R.H.M., Macedo, A.V.S., Bronhara, B., Damiani, L.P.et al. (2021) Therapeutic versus prophylactic anticoagulation for patients admitted to hospital with COVID-19 and elevated D-dimer concentration (ACTION): an open-label, multicentre, randomised, controlled trial. Lancet 397, 2253–2263 10.1016/S0140-6736(21)01203-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 268.Viecca, M., Radovanovic, D., Forleo, G.B. and Santus, P. (2020) Enhanced platelet inhibition treatment improves hypoxemia in patients with severe COVID-19 and hypercoagulability. A case control, proof of concept study. Pharmacol. Res. 158, 104950 10.1016/j.phrs.2020.104950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.van der Togt, V., McFarland, S., Esperti, M., Semper, E., Citrenbaum, S. and Rossman, J.S. (2021) Promotion of non-evidence-based therapeutics within patient-led long COVID support groups. Nat. Med. 27, 2068–2069 10.1038/s41591-021-01589-y [DOI] [PubMed] [Google Scholar]
- 270.Kozieł, M., Potpara, T.S. and Lip, G.Y.H. (2020) Triple therapy in patients with atrial fibrillation and acute coronary syndrome or percutaneous coronary intervention/stenting. Res. Pract. Thromb. Haemost. 4, 357–365 10.1002/rth2.12319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Pretorius, E., Venter, C., Laubscher, G.J., Lourens, P.J., Steenkamp, J. and Kell, D.B. (2020) Prevalence of readily detected amyloid blood clots in ‘unclotted’ type 2 diabetes mellitus and COVID-19 plasma: a preliminary report. Cardiovasc. Diabetol. 19, 193 10.1186/s12933-020-01165-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272.Thagard, P. (2007) Coherence, truth, and the development of scientific knowledge. Philos. Sci. 74, 28–47 10.1086/520941 [DOI] [Google Scholar]
- 273.Thagard, P. (2008) Explanatory Coherence. Reasoning: Studies of Human Inference and Its Foundations, 471–513
- 274.Bradford Hill, A. and Hill, I.D. (1991) Bradford Hill's Principles of Medical Statistics, Edward Arnold, London [Google Scholar]
- 275.Schaffner, K.F. (1993) Discovery and Explanation in Biology and Medicine, University of Chicago Press, Chicago [Google Scholar]