Abstract
Short telomeres confer risk of degenerative diseases. Chronic psychological stress can lead to disease through many pathways, and research from in vitro studies to human longitudinal studies has pointed to stress-induced telomere damage as an important pathway. However, there has not been a comprehensive model to describe how changes in stress physiology and neuroendocrine pathways can lead to changes in telomere biology. Critically short telomeres or the collapse of the telomere structure caused by displacement of telomere binding protein complex shelterin elicit a DNA damage response and lead to senescence or apoptosis. In this narrative review, we summarize the key roles glucocorticoids, reactive oxygen species (ROS) and mitochondria, and inflammation play in mediating the relationship between psychological stress and telomere maintenance. We emphasis that these mediators are interconnected and reinforce each other in positive feedback loops. Telomere length has not been studied across the lifespan yet, but the initial setting point at birth appears to be the most influential point, as it sets the lifetime trajectory, and is influenced by stress. We describe two types of intergenerational stress effects on telomeres – prenatal stress effects on telomeres during fetal development, and ‘telotype transmission”–the directly inherited transmission of short telomeres from parental germline. It is clear that the initial simplistic view of telomere length as a mitotic clock has evolved into a far more complex picture of both transgenerational telomere influences, and of interconnected molecular and cellular pathways and networks, as hallmarks of aging where telomere maintenance is a key player interacting with mitochondria. Further mechanistic investigations testing this comprehensive model of stress mediators shaping telomere biology and the telomere-mitochondrial nexus will lead to better understanding from cell to human lifespan aging, and could lead to anti-aging interventions.
Keywords: Telomeres, Telomerase, Stress, Glucocorticoid, Reactive oxidative species, Inflammation, Shelterin complex, Mitochondria
1. Introduction
Chronic psychological stress is a major contributor to human health and aging, and is associated with the development of many physical and psychological disorders including cardiovascular disease, type 2 diabetes, metabolic syndrome, autoimmune disease and depression (Cohen et al., 2007). A deeper understanding of the mechanistic pathways by which psychosocial stress affects physiology and disease etiology is crucial to the development of therapeutic and interventional strategies to mitigate the negative impacts of stress. In recent years, telomeres, the DNA-protein structures at the end of chromosomes, have emerged not only as biomarkers, but as mediators through which chronic psychosocial stress leads to diseases. Specifically, over the last seventeen years, a growing literature links stress to alterations in telomerase activity and telomere shortening across many species. But we lack a comprehensive model linking changes in stress physiology to molecular changes in telomere biology. In this selective review, we summarize recent advances in understanding the stress-telomere-disease pathways, with a focus on the molecular mechanisms, including stress hormones, reactive oxidative species and inflammation. We propose that these neuroendocrine and cellular pathways interact with and reinforce each other to form interconnected networks that translate stress to disease risk. Lastly, we discuss methodological issues of telomere length measurement and future research directions.
2. Brief overview of telomere biology basics
Telomeres are DNA-protein structures at the end of the chromosomes, made up of short tandem DNA repeats and a multicomponent protein complex called shelterin (de Lange, 2018; Hockemeyer and Collins, 2015) (Fig. 1). Due to the end replication problem, i.e. the inability of the DNA replication machinery to complete the synthesis of the very tip of linear chromosomes, telomeres shorten as cells divide. Telomere shortening also happens through oxidative damage and other end processing events, in both dividing and non-dividing cells. The attrition of telomeres can be counteracted by telomerase, a specialized enzyme whose main cellular function is to elongate telomeres. Dysfunctional telomeres, caused by telomere shortening, the collapse of the telomere structure, or displacement of shelterin complexes from telomeres, elicit a DNA damage response, and loss of cell proliferation leading to senescence or apoptosis.
Fig. 1.
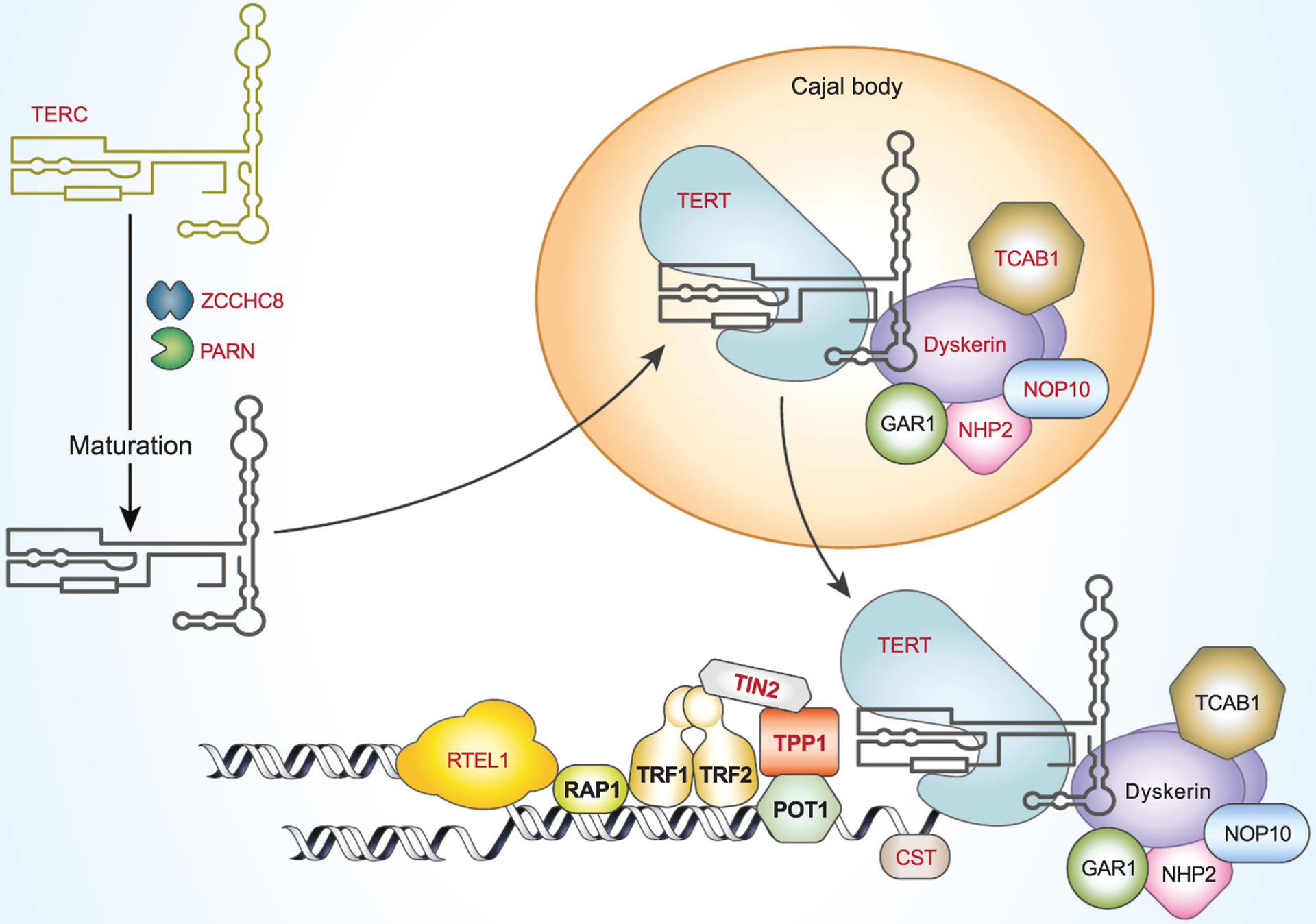
Telomeres and telomerase and shelterin complex and other genes implicated in telomeropathies. Shelterin complex protects and caps the telomere. Shelterin complex proteins are shown in bold. Genes with mutations found in patients with short telomere syndrome are shown in red. Longer forms of telomerase RNA hTERC are transcribed and ZCCHC8 is required for maturation of the 3’ end of the TERC (Gable et al., 2019). PARN is involved in further trimming of the 3’ end before entering Cajal bodies. TERC is assembled with TERT, dyskerin, NHP2, NOP10, GAR1 and TCAB1 to form the telomerase holoenzyme in Cajal bodies before being recruited to telomeres by the shelterin protein TPP1. RTEL1 is required for the unwinding DNA at a replication fork. The CST complex (CTC1, STN1, TEN1) regulates telomerase activity and promotes the C-strand telomerase-independent telomere replication by primase and DNA polymerase alpha (Lyu et al., 2021).
The shelterin complex insulates telomere DNA from inappropriate DNA repair pathways and prevents activation of the DNA damage response. Shelterin also coordinates the regulation of telomerase activity at telomeres and ultimately helps define the set point for telomere length, establishing telomere length homeostasis. The functions of the shelterin complex have been reviewed in detail (Palm and de Lange, 2008; Lim and Cech, 2021) and here we will only provide a brief summary. Telomeric Repeat binding Factors 1 and 2 (TRF1 and TRF2) bind directly to the duplex part of telomeres (Broccoli et al., 1997; Bianchi et al., 1997), whereas the POT1-TPP1 heterodimer binds telomeric TTAGGG repeats at the single strand 3’ overhang (Baumann and Cech, 2001; Liu et al., 2004). TPP1, TIN2 and Rap 1 locate to telomeres through their binding to other proteins of the shelterin complex (Liu et al., 2004; Zhu et al., 2000; Houghtaling et al., 2004). By binding to telomeres, the shelterin complex caps telomeres and blocks access by other proteins. Protected telomeres prevent the ends of chromosomes from being seen as double-strand breaks (DNA damage). Breaks and unprotected chromosomes activate the DNA damage response, and the subsequent repair pathways (including homologous recombination and non-homologous end joining) (Bae and Baumann, 2007; He et al., 2006). Shelterin also protects telomeres from degradation by nucleases (Hockemeyer et al., 2006). Additionally, shelterin plays direct roles in telomere length homeostasis and telomerase regulation by both inhibiting and stimulating telomerase activity. The number of TRF1 and other shelterin components at a telomere proportionally increases with the number of TTAGGG repeats as they are used as a counting mechanism for the length of a telomere (Smogorzewska et al., 2000). Therefore, TRF1, TRF2, TIN2, TPP1, and POT1 behave as negative regulators of telomere length. While the physical presence of POT1 blocks the access of telomerase to the 3’ single strand end of telomeres, thus inhibiting telomere elongation, recent data also indicate that TIN2, POT1 and TPP1 work together to recruit telomerase to the telomere and to stimulate telomerase activity when telomeres need to elongated (Pike et al., 2019). While it is clear that the shelterin complex is critical to telomere length homeostasis and telomerase activity regulation at the telomeres, the specific molecular mechanisms of these processes are still be investigated.
3. Immune cells telomere length’s provide window into aging
In human population research, much of our ability to probe the relationship between telomeres, diseases and associated factors has come through the use of immune cells. Telomere length varies across tissue types, but is correlated to various degrees (Abgrall et al., 2013; Daniali et al., 2013). Immune cells are ideal for telomere research for a number of reasons: they are easy to obtain from blood and readily available, and since they circulate throughout the body, immune cells are exposed to both internal (from cells) and external (from diet and exposures) factors that affect telomere maintenance. Immune cells are also at the center of the interaction between psychological processes and the nervous and immune systems. In turn, a buildup of senescent immune cells contributes to ‘inflammaging’—increases in circulating systemic inflammation (Campisi and Robert, 2014). Senescent cells from adipose tissue, and other tissues, contribute to systemic inflammation, although this is less studied and thus harder to quantify (Hotamisligil et al., 1995).
Circulating peripheral blood cells contains white blood cells, red blood cells and platelets. In humans, only white blood cells have nuclei and are therefore the relevant cell type for assessing telomere biology. White blood cells, also called leukocytes, are composed of lymphocytes, granulocytes and monocytes, which have all been shown to experience age-dependent telomere shortening (Ouyang et al., 2007; Rufer et al., 1998). Telomere length (TL) and telomerase activity (TA) during T and B lymphocyte development have been extensively studied and reviewed in detail elsewhere (Hodes et al., 2002). Briefly, in CD4+ T lymphocytes, TL shortens and telomerase activity decreases as cells differentiate from naïve to memory cells (Rufer et al., 1998, 1999; Weng et al., 1995). Differentiated CD8+ T cells lose expression of the CD28 co-stimulation marker, coinciding with decreased TA (Lin et al., 2010) and shorter TL (Monteiro et al., 1996). It may be that decreases in telomerase precede and trigger the loss of CD28 markers (Dagarag et al., 2004). The proportion of CD4+ and CD8+ T cells that have a memory phenotype increases with age, reflecting the cumulative history of antigen exposure during an individual’s lifetime.
Telomere length and telomerase activity during B cell development have a different trajectory. B cells in germinal centers, where naïve B cells differentiate into antibody producing plasma cell or memory B cells, have telomeres that are substantially longer than the telomeres in either naive or memory B cells, which is due to upregulation of telomerase in these cells in germinal centers (Weng et al., 1997). The rate of age-dependent telomere shortening is lower in B-cells compared to T-cells, possibly due to the increased telomerase activity in the germinal center (Son et al., 2000). Overall, CD4 and CD8 T cells have similar telomere lengths, while B cells have longer telomere lengths than T cells (Lin et al., 2010). Given the complexity of telomere dynamics in T and B cell development, telomere length measured in circulating blood is the cumulative readout of both the composition and the replicative history of all cell types. The vast majority of the research so far has used either total leukocytes or peripheral blood mononuclear cells (PBMCs, a collection of T and B lymphocytes, natural killer cells and monocytes) as the sources for telomere measurements, which does not provide sufficient granularity as to whether and how the differential telomere and senescent profiles of specific cell types may be linked to certain diseases or risks.
In our human studies, when we have been able to measure TL in granulocytes, the most frequent cell type in whole blood, and in other cell types, we have often found that they have similar behavioral correlates. For example, both granulocyte TL and PBMC or lymphocyte TL were associated with weight loss (Mason et al., 2018) and psychological traits (Epel et al., 2013) although we have observed exceptions, where only PBMC telomere length was associated with lifestyle changes (Prather et al., 2015; Tomiyama et al., 2017). Given the different sources of granulocytes (myeloid progenitor cells) vs. lymphocytes (lymphoid progenitor cells), and the fact that granulocytes are short lived, it is logical that they may provide a close reflection of the status of stem cells rather than a reflection of historical exposures.
4. Telomere length and disease
In 2003, Richard Cawthon published the seminal study that examined telomere lengths in a community sample over 60 years old, and demonstrated shorter leukocyte telomere length predicted earlier all-cause mortality as well as mortality from infectious disease and heart disease (Cawthon et al., 2003). Since then, an explosion of literature has shown associations between telomere length, as measured in the blood, and various human diseases in observational studies, leading to many meta-analyses showing small but consistent correlations. The list includes major diseases such as cardiovascular diseases (Yeh and Wang, 2016), diabetes and metabolic syndrome (Cheng et al., 2020), COPD and idiopathic pulmonary fibrosis (Birch et al., 2018), infection (including COVID-19) (Noppert et al., 2020; Asghar et al., 2018, 2016; Helby et al., 2017; Bellon and Nicot, 2017; Froidure et al., 2020; Sanchez-Vazquez et al., 2021), psychological disorders (Epel and Prather, 2018), autoimmune diseases (Habib et al., 2020; Kamycheva et al., 2017; Zeng et al., 2020), and some types of cancers (Zhang et al., 2017; Zhu et al., 2016). Observational studies have been inconsistent with regard to the direction of effect for different types of cancers.
A causal relationship between TL and diseases is bolstered by discoveries of germline mutations in telomere maintenance genes in people with rare genetic diseases, collectively called short telomere syndromes, or telomeropathies (Grill and Nandakumar, 2020). In these short telomere syndromes, disease manifests in multiple tissue types in both high cell turnover tissues such as bone marrow and skin, as well as slow turnover tissues like lung and liver. Mutations in fourteen different genes have been identified to date in patients with short telomere syndromes. These genes are involved in the biogenesis, processing and assembly of the reverse transcriptase enzyme telomerase (TERT, TERC, DKC, NOP10, HNP2, NAF1, WRAP53/TCAB1, PARN and ZCCHC8), telomerase-independent telomere replication (RTEL1, CTC and STN1), or are components of the shelterin complex (TPP1, TIN2). Fig. 1 shows an up to date list of components involved in telomerase and shelterin complex, as well as those that have already been implicated in the telomere syndromes.
Interestingly, while there are many overlapping diseases between short telomere syndromes and those associated with short telomeres in Mendelian Randomization studies (described below), the disease lists are far from identical. The much larger TL shortening effect caused by the mutations in short telomere syndromes, compared to the relatively small difference due to the genetic variants in general populations may provide part of the answer. In telomere syndromes, critically short TL earlier in life may manifest acutely in the high turnover tissues, such as in bone marrow, a phenomenon that is infrequently seen in normal aging with the gradual shortening of telomeres over decades.
Further evidence for a direct, causal relationship between telomere length and diseases come from Mendelian randomization studies where a genetic score made up of several genetic variants of telomere length was used as an instrumental predictor variable. Because these genetic variants are assigned randomly when passed from parents to offspring, this approach mimics a “naturally” randomized controlled trial and provides the strongest evidence of a causal relationship between the genotype (TL) and disease phenotypes without the influence of confounders that are unavoidable in observational studies. Mendelian randomization studies demonstrated that short TL based on the genetic score (what we call gTL) is associated with a wide variety of degenerative diseases (coronary heart disease, abdominal aortic aneurysm, IPF, and Alzheimer’s disease) and several autoimmune diseases (RA, celiac disease, type I diabetes and hypothyroidism) (Zeng et al., 2020; Li et al., 2020; Telomeres Mendelian Randomization et al., 2017; Kuo et al., 2019; Duckworth et al., 2020; Scheller Madrid et al., 2020; Gao et al., 2019). However, long telomere length based on the genetic score is associated with many types of cancer and conditions of excess tissue growth (uterine fibroids, uterine polyps, and benign prostatic hyperplasia) (Li et al., 2020; Zhang et al., 2020; Xu et al., 2020; Muskens et al., 2019; Gao et al., 2020). Thus, there is inherent complexity in understanding TL and disease – it is not a one-way directional path, especially when it comes to genetically determined TL.
The findings with cancer led to notably high odds ratio effect sizes, and confusion about what is worse – having long vs. short telomeres in terms of overall risk of disease. Given that the cancers are much rarer at the population level than chronic degenerative diseases, a direct comparison of odds ratios could be misleading in terms of inferring absolute risk. This prompted our group to examine the overall impact of genetically determined long vs. short telomeres (gTL) on disease incidence and burden. Our analysis revealed that the disease burden of long vs. short telomere on a population, estimated by the Disability Adjusted Life Years (DALY), a metric from the Institute for Health Metrics and Evaluation (IHME) that accounts for both mortality and morbidity, is of approximate equal magnitude (Protsenko et al., 2020). These results demonstrate that odds ratios alone, from a single study or disease, can be misleading, and disease risk should be assessed from both an individual and population level to infer level of disease risk in human aging. These findings can be viewed in the light of the natural trade-off with aging – that with aging, our growth hormones and other proliferative factors may decrease to reduce cancer risk, but this leads to an increase in degeneration of tissues. Thus, the maintenance of human telomere length is balanced by opposing evolutionary forces acting to minimize both neoplastic and non-neoplastic diseases. However, it should be noted that although the length of critically short telomeres has been demonstrated in experimental settings and in telomeropathies, there is no evidence of a clear definition for the cut off of what would define “long telomeres”. We thus use the relative term ‘excessively long telomeres” to describe the long telomeres that bring some level of risk.
5. Physiological to molecular pathways
The transmission of psychological stress to physiological neuroendocrine and autonomic changes, to cellular and molecular targets is linked by multiple processes that are interconnected and often reinforce each other in positive, feedforward loops. In particular, although many changes are involved, we highlight the pivotal role of influential stress mediators – glucocorticoids (GC), reactive oxygen species (ROS) with weakening of antioxidant defenses, and inflammation. Below we review evidence linking stress exposures to TL, and how each individual mediator affects aspects of telomerase and telomere maintenance, and also discuss the links between these processes. Fig. 2 illustrates the molecular and cellular pathways linking psychological stress to short and dysfunctional telomeres. For detailed discussions of mechanistic pathways linking stress and telomerase, we refer the readers to a recent review by de Punder (de Punder et al., 2019). Another potential mechanism of accelerated telomere shortening in aging and aging related diseases could come from intrinsic telomere structure. Telomeres are fragile, chromosomal regions that are difficult to replicate, especially under limited nucleotide pools (Sfeir et al., 2009), The G-quarduplex formed at telomeres by the G-rich strand may cause replication stall and collapse, resulting in telomere shortening (Bryan, 2020). Thus, the intrinsic replication stress in telomeres could accelerate telomere shortening, especially in highly proliferative cells. It is not known if physiological stress could increase replication stress.
Fig. 2.
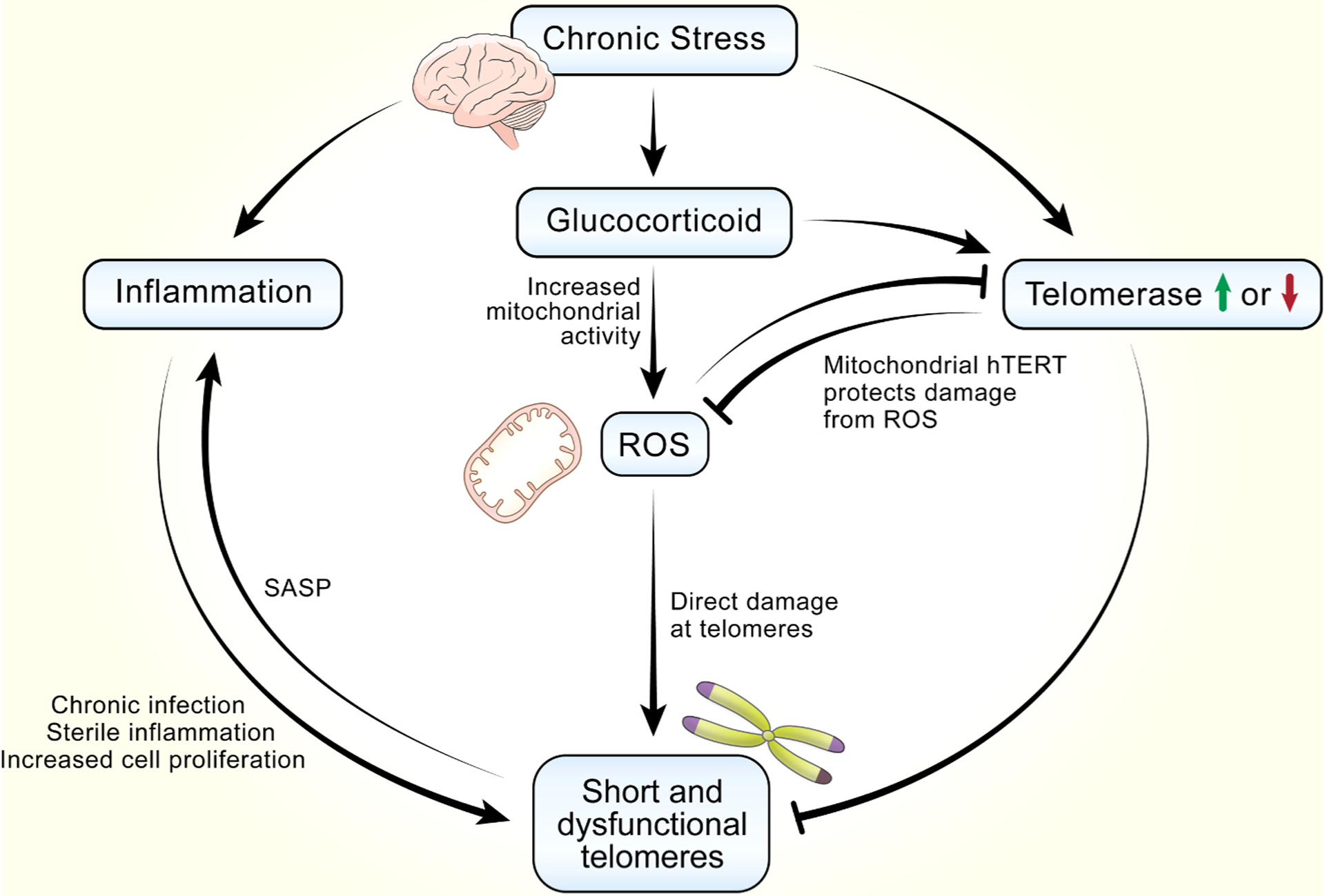
Interconnected molecular and cellular pathways linking psychological stress to short and dysfunctional telomeres. Stress induces secretion of glucocorticoids, which leads to the generation of ROS through increased mitochondrial activity. ROS preferentially damages telomeres and inhibits telomerase activity, and both contribute to short and dysfunctional telomeres. Glucocorticoid can also directly inhibit or induce telomerase activity depending on specific contexts. The roles of telomerase in mitochondria functions are illustrated in more details in Fig. 3. Another important pathway downstream of glucocorticoid is increased inflammation, which lead to short and dysfunctional telomeres viral chronic infection, sterile inflammation and subsequent increased immune cell proliferation. Senescent cells caused by dysfunctional telomeres display a pro-oxidant, pro-inflammatory phenotype known as the senescence associated secretory phenotype (SASP). Thus, inflammation and dysfunctional telomeres form a positive, reinforcing feedback loop, leading to increasing systemic inflammation and increases in number of cells with dysfunctional telomeres.
5.1. Stress and TL-clinical studies
Several recent meta-analyses have confirmed the association of different types of stressors and short telomere length (Schutte and Malouff, 2016; Pepper et al., 2018; Oliveira et al., 2016). Epel and Prather (Epel and Prather, 2018) reviewed studies linking stress, depression, and TL, including animal studies. There appears to be a strong association for early life stress, and for depression, at least cross sectionally. Experimental studies with animals showed causal relations between stress and TL shortening. Corroboratively, in non-humans, a recent meta-analysis of 109 papers showed that the exposure to potentially threatening stressors (physiological or psychological) was associated with shorter telomeres or higher telomere shortening rate in 72 species including birds, mammals, fish and reptiles. The effects of these stressors were stronger in the cases of pathogen infection, competition, reproductive effort and high activity level (Chatelain et al., 2020). The actual mechanisms of how systemic neuroendocrine and neuro-inflammatory factors influence cellular and molecular factors are reviewed below.
5.2. Glucocorticoids and the HPA axis
The major outcome of activation of the hypothalamic–pituitary–adrenal (HPA) axis by stress is the surge of glucocorticoids in the systemic blood stream (Munhoz et al., 2006, 2010). In humans, a meta-analysis showed no association between basal cortisol with TL, but found that higher stress-related cortisol reactivity was associated with shorter TL (Jiang et al., 2019). Several studies have found that severe stress during pregnancy is associated with shorter TL in newborn offspring in most but not all studies (Izano et al., 2020; Entringer et al., 2013; Marchetto et al., 2016; Send et al., 2017; Salihu et al., 2016). One study of 92 mother-infant dyads found that higher cortisol during pregnancy (as assessed by maternal hair cortisol) predicted shorter telomere length in the female offspring, based on telomeres in the infant’s cord blood (Bosquet Enlow et al., 2019). This maternal stress effect could be through prenatal programming via effects of cortisol and other stress mediators on the fetus. Alternatively, or in addition, it might be intergenerational transmission of short telomere length through epigenetic like direct transmission from parental gamete’s short telomere length. The reason for short parental telomere length could be from any causes, including lifetime adversity (Epel, 2020). We are labeling unique direct transmission effect of parental telomeres to offspring “telotype” transmission, to differentiate it from traditional epigenetic transmission.
Some experimental studies in birds have been conducted. In wild birds, six studies showed that exposure to glucocorticoid in experimental settings leads to shorter TL or faster attrition (Haussmann et al., 2012; Schultner et al., 2014; Tissier et al., 2014; Herborn et al., 2014; Casagrande et al., 2020; Pegan et al., 2019). Exposure to psychological stress has also led to telomere shortening in birds (Lewin et al., 2015). However, the timing of exposure may determine effects. Noguera et al. reported that manipulating high levels of egg corticosterone can stimulate telomerase activity and promote longer telomeres during embryo development (Noguera et al., 2020). Similarly, Ost et al. also observed higher egg corticosterone was associated with longer telomeres at hatching (Ost et al., 2020). In wild birds, contradictory results have been reported, showing the relationship between glucocorticoid and TL is clearly context dependent and likely dose dependent (Angelier et al., 2018).
There have been very few in vitro studies to examine the relationship between cortisol exposure with telomerase or TL. Given the known effects of acute vs. chronic cortisol exposure on immune function (stimulatory and then suppressive), one might expect bimodal effects with time. Choi et al. first reported that exposure to elevated cortisol levels in PBMCs and in isolated CD4 and CD8 T cells over 3 days was associated with a downregulation of telomerase activity, with reduced levels of the RNA transcript for hTERT, the telomerase catalytic component. Later studies showed that the relationship between glucocorticoid and telomere maintenance to be context-dependent. For example, Bull et al. reported that treatment of cortisol in human lymphocytes resulted in longer TL and decreased proliferation (Bull et al., 2015). It is possible that in this experimental setting, the long TL is due to fewer cell divisions in the cortisol treatment condition. An vitro study found no TL shortening in fibroblasts treated with cortisol for up to almost two months, and it is unclear if fibroblasts are equally sensitive to cortisol as immune cells are, given that immune cells need to respond acutely to stress (Zannas et al., 2020).
Mechanistically, glucocorticoids alter fundamental aspects of the cell, such as increased metabolic rates and mitochondria activity, which are associated with increased production of ROS (Chatelain et al., 2020). Glucocorticoids can also directly affect oxidative balance through either genomic (Atanasova et al., 2009) or non-genomic mechanisms (Costantini et al., 2011). Elevated glucocorticoids can increase the expression of pro-inflammatory genes (iNOS, IL-1β, TNF-α) and decrease anti-inflammatory genes including IL-1ra, IL-10, and MKP-1 (Escoter-Torres et al., 2019). Further, one study found that in cultured cell lines, the gene expressions of shelterin complex proteins TRF1, TIN2, and POT1 were upregulated by dexamethasone, whereas TERT, TRF2, TRF1, TIN2, and POT1 were upregulated by TNF-α, suggesting the transcriptional regulation of shelterin complex as a potential mechanism of stress related telomere length dynamics (Butler et al., 2012). Clearly more studies are needed to examine the effects of various exposures to stress mediators (GCs and inflammatory) on regulation of the telomere, telomerase, and shelterin complex proteins. The roles of ROS and inflammation on telomere maintenance are discussed below.
5.3. Mitochondria and reactive oxygen species (ROS)
Oxidative stress (reactive oxygen species; ROS) is one of the most potent biological effectors of stress-related endocrine, immune/inflammatory and metabolic pathways on cellular health (Gidron et al., 2006; Black et al., 2017). A recent review showed that 6 out of 8 human population studies that examined markers for oxidative and telomere lengths found a negative correlation between the two (Reichert and Stier, 2017). Early work using in vitro cultured human cells reported that cellular exposures to oxidizing agents led to greater oxidative damage to telomeres compared to microsatellite repeats, another chromosomal region with repeat sequences, and overall genomic DNA (Rhee et al., 2011; Petersen et al., 1998; Coluzzi et al., 2014; Wang et al., 2010). The rate of TL shortening correlated with oxygen concentration (Ahmed et al., 2008; Forsyth et al., 2003; Passos et al., 2007).
Telomeres are thought to be particularly vulnerable to oxidative damage for several reasons (von Zglinicki, 2002). First, telomeric DNA is particularly sensitive to ROS because of its G-rich sequence (Oikawa and Kawanishi, 1999) as 8-oxoguanine (8-oxo-G) is one of the most common DNA lesions resulting from reactive oxygen species. ROS also induce single strand breaks (SSB) at telomeres directly, or as intermediates in lesion repair, leading to replication fork collapse and telomere loss (von Zglinicki, 2002). Second, the presence of the shelterin complex at telomeres prevents recruitment of DNA damage response (DDR) proteins, therefore, damage at telomeres by ROS may not efficiently activate the DDR. While shelterin prevents the ends of the telomere DNA being recognized as DNA damage and the subsequent cell cycle arrest, it also hampers the DNA repair process downstream of the initial DNA damage response (Palm and de Lange, 2008). Third, 8-oxoguanine glycosylase (OGG1), the primary enzyme responsible for excision of 8-oxoG, cannot remove it when present in single stranded DNA or a folded G-quadruplex structure, as in the case of telomere DNA (Zhou et al., 2013). Furthermore, oxidized telomere DNA and nucleotides interfere with telomerase action. Incorporation of free 8-oxoG by telomerase into telomeres serves as a chain terminator to prevent further telomere extension (Ahmed and Lingner, 2018). However, as the shelterin proteins TRF1 and TRF2 bind to ROS-damaged telomere DNA with less affinity, it is also possible that the oxidized telomeres are more accessible to telomerase action (Opresko et al., 2005). But the negative effects of ROS on telomere length outweigh this potential positive effect.
ROS are generated by exogenous and endogenous sources. The primary endogenous source of ROS is inflammation and/or mitochondrial dysfunction, since ROS are generated by immune cells in response to infection or injury and from oxygen metabolism. We will discuss the relationship between inflammation and telomere length in the next section. Telomere maintenance and mitochondrial function are intimately related and form a bidirectional, feedforward loop. As shown in Fig. 3, telomere damage leads to mitochondrial biosynthesis reprogramming and dysfunction, through activation of the tumor repressor gene p53, which in turn binds and represses the PGC-1α and PGC-1β promoters, master regulators of mitochondrial physiology and metabolism (Passos et al., 2010; Sahin et al., 2011). Conversely, mitochondrial dysfunction leads to telomere loss as well as telomere dysfunction-induced foci (TIFs), in which DNA-damage response factors are recruited to critically short and/or uncapped telomeres (Qian et al., 2019). Interestingly, about 10–20% of the total cellular telomerase protein subunit hTERT is localized in mitochondria (Santos et al., 2006; Gordon and Santos, 2010) and shuttles between nucleus and mitochondria. The mitochondrial telomerase protein may protect cells from ROS damage by binding to mitochondrial DNA (mtDNA) as well as mitochondrial tRNA (Ahmed et al., 2008; Haendeler et al., 2009; Sharma et al., 2012). Recently, the telomerase RNA component TERC is found to be imported into mitochondria, processed, and exported back to the cytosol to serve as a signaling molecule, relating the functional states of mitochondria to the nucleus (Cheng et al., 2018; Zheng et al., 2019). Few studies have examined the mechanisms of mitochondria function and telomere regulation at the same time. Measuring TIFs is time consuming but would offer a new outcome linking exposures/predictors to telomere damage due to mitochondrial dysfunction. This may offer new insights into the role of telomere damage vs. average telomere lengths which results from damage but also many other factors.
Fig. 3.

Bidirectional, feedforward loop of telomere maintenance and mitochondrial function. In healthy cells, telomerase protein subunit hTERT is exported from the nucleus to the mitochondria to protect cells from ROS damage by binding to mitochondrial DNA (mtDNA) and mitochondrial transfer RNA. However, telomere damage leads to decreased mitochondrial biogenesis through activation of p53, which represses the PGC-1α and PGC-1β promoters, master regulators of mitochondrial physiology and metabolism. Conversely, mitochondrial dysfunction leads to short and dysfunctional telomeres. Recent evidence also suggests that telomerase RNA TERC is processed in the mitochondria and shuttled back to the nucleus to signal mitochondrial function.
5.4. Inflammation
Chronic inflammation is a major underling mechanism of normal aging and aging related diseases and risks (De Martinis et al., 2006; Furman et al., 2019). The main sources of inflammation include age-dependent chronic activation of tissue macrophages and T cells, leading to increased proinflammatory cytokines with IL-6, TNFα and IL-1β as major players (Wikby et al., 2006). Over the last decade, it has become increasingly clear that many diseases associated with chronic inflammation are also associated with short TL, lending to the proposal that inflammation and telomere dysfunction interact with each other to contribute to disease risk and progression (Kordinas et al., 2016). Indeed, in human studies, elevated levels of pro-inflammatory cytokines have been associated with shorter telomeres (O’Donovan et al., 2011; Pusceddu et al., 2020; Shin et al., 2019). In the Health, Aging and Body Composition Study cohort, with an age range of 70–79 years, cumulative inflammatory load, as indexed by the combination of high levels of IL-6 and TNFα, was associated with increased odds for short LTL (O’Donovan et al., 2011). In the Ludwigshafen Risk and Cardiovascular Health Study, subjects with the longest telomeres had lower concentrations of IL-6, and hs-CRP (Pusceddu et al., 2020). In the National Health and Nutrition Examination Survey (NHANES) 1999–2002 cohort, shorter telomere length was associated with increased levels of CRP, but stratified analysis showed that this association was only present in men without depression (Shin et al., 2019). Depression itself may generate inflammation or result from inflammation, making it a confounder to this relationship.
Several cellular mechanisms have been proposed to link inflammation and telomere maintenance (Fig. 2). Immune activation and clonal expansion due to chronic infection lead to increased cell proliferation, therefore accelerated TL attrition, as has been shown in CMV, HIV, Herpesvirus, hepatitis B and C and other chronic infections (Bellon and Nicot, 2017; Chou and Effros, 2013; Dowd et al., 2017). Inflammation can also be triggered by endogenous ligands, such as molecules from damaged tissues, without exogenous pathogens, a state called sterile inflammation. Sterile inflammation increases as people age and plays a major role in aging-related conditions and diseases. Inflammation and short telomere length appear to work bidirectionally. Mouse models with the telomerase genes TERT or TERC genetically deleted exhibit a pro-inflammatory state, with elevated levels of proinflammatory cytokines, including IL-1, IL-6, CXCL15 (human IL-8 homolog), TNFα, and monocyte chemotactic protein 1 (chemokine ligand 2 (CCL2)) (Chen et al., 2015). On the other hand, one of the major defining features of senescence -and dysfunctional telomeres- is the secretion of proinflammatory cytokines, chemokines, extracellular matrix proteases, growth factors, and other signaling molecules collectively termed the senescence-associated secretory phenotype (SASP) (Campisi and Robert, 2014). SASP is an important mediator of the pathophysiological effects of senescence and may explain, at least in part, this local inflammation within tissues. The functions of SASP are context-dependent and pleiotropic in both autocrine and paracrine fashions (Acosta et al., 2013, 2008; Coppe et al., 2006; da Silva et al., 2019; Herranz and Gil, 2018; Krtolica et al., 2001; Victorelli et al., 2019).
Paradoxically, there exists a positive feedback loop between the expression of the telomerase protein component (hTERT in humans) and NF-κb, the master transcription factor for inflammation. Expression of hTERT is regulated by many transcriptional factors (reviewed in (Ramlee et al., 2016)). The hTERT promoter region has an NF-κb binding site and NF-κb is reported to upregulate telomerase activity in multiple cell lines (Gizard et al., 2011; Aravindan et al., 2011). NF-κb also modulates the nuclear translocation of hTERT in human lymphocytes in response to TNF-α stimulation (Akiyama et al., 2003). On the other hand, hTERT directly regulates NF-κb-dependent gene expression by binding to the NF-κb p65 subunit and to a subset of NF-κb promoters such as IL-6 and TNFα (Ghosh et al., 2012; Ding et al., 2013). In turn, IL-6 and TNF-α upregulate telomerase activity through NF-κb and STAT1 and STAT 3, two other important transcriptional factors (Chung et al., 2017). hTERT also upregulates the expression of matrix metal-loproteinase, another component of SASP, via NF-κb-dependent transcription independently of hTERT telomere elongation activity (Ding et al., 2013). It is not clear how this positive, reinforcing relationship between telomerase activity and inflammation plays out to impact telomere length. It is possible that the apparent telomerase upregulation/activation reflects the immune activation state associated with inflammation, which in turn promotes cell division and leads to telomere shortening. However, even under this scenario, the role of hTERT remains unclear. The upregulation of telomerase activity might be required for NF-κb to exert its full function, but it is also possible that it is a mere bystander of inflammation. Clearly telomerase is acutely responsive to stress mediators, GCs and inflammation, and ROS. It is thus possible that the duration and kinetics of the stress response determines if telomerase remains elevated (such as from acute stress) or becomes suppressed (such as from chronic stress or due to inability to mount a response in an aged or unhealthy organism).
6. Beyond telomere length
Much of the discussion about the role of telomere maintenance on aging focuses on short and dysfunctional telomeres. In addition to stem cell failure due to the inability of senescent cells to divide, short telomeres also have global effects, as dysfunctional telomeres and other hallmarks of aging are interconnected (Chakravarti et al., 2021). Critically short telomeres elicit DNA damage responses, defective mitochondrial biogenesis (Sahin et al., 2011) and downregulation of all sirtuins, linking telomeres to metabolic control (Amano et al., 2019). Recently, a new axis involving short telomeres-NAD metabolism-Sirt1-mitochondrial ROS was reported and lends support to global effects of short telomeres (Sun et al., 2020). Fibroblasts from dyskeratosis congenita patients display lower nicotinamide adenine dinucleotide (NAD) levels, and an imbalance in the NAD metabolome that includes elevated CD38 NADase and reduced poly(ADP-ribose) polymerase and SIRT1 activities. Supplementation with the NAD precursor, nicotinamide riboside, and CD38 inhibition alleviated telomere damage, defective mitochondrial biosynthesis and clearance, cell growth retardation, and cellular senescence of DC fibroblasts. Additionally, changes in telomere structure due to telomere shortening may release shelterin proteins such as Rap1 and TRF2, that might have bound to non-telomere sites, therefore revealing other functions (Martínez et al., 2013; Ye et al., 2014; Cherfils-Vicini et al., 2019; Zizza et al., 2019). Below, we briefly discuss several telomere length independent mechanisms that have been discovered in the last few years.
6.1. Telomere position effect
First described by Baur (Baur et al., 2001), telomere position effect is when the length of a telomere impacts the expression of genes nearby (Lou et al., 2009; Stadler et al., 2013) or long distance genes through chromosome looping (Robin et al., 2014, 2015; Kim et al., 2016), resulting in the repression of gene transcription by long telomeres. Interestingly, hTERT is one of the genes regulated by telomere position effect, providing another potential mechanism for how telomere length and telomerase interact to achieve telomere length homeostasis (Kim et al., 2016). Another interesting example is interferon stimulated gene 15 (ISG15), identified in a screen for genes upregulated by short telomere length (Lou et al., 2009). ISG15 is expressed as part of the innate immunity/stress response pathway and can stimulate the production of the proinflammatory interferon, IFNγ, in immune cells (Recht et al., 1991; D’Cunha et al., 1996). Thus, the upregulation of ISG15 by short telomere length may serve as a mechanistic link between immune aging and age-related inflammation. There is also tentative evidence that TL independent looping occurs during stimulation of CD4 immune cells in vitro, although the biological significance of this phenomenon is not clear (Kim and Shay, 2018).
6.2. TElomeric repeat-containing RNA (TERRA)
Although the telomere region does not encode protein sequences, long non-coding RNAs, are transcribed from telomeres. TElomeric Repeat-containing RNA (TERRA) (Azzalin et al., 2007; Porro et al., 2014) and telomeric damage-induced long non coding RNAs (tdilncRNA) are composed of subtelomeric sequence and variable lengths of telomeric RNA (Rossiello et al., 2017; Nguyen et al., 2018). A tight regulation of TERRA expression and localization is required to maintain telomere and chromosome stability (Lopez de Silanes et al., 2014; Montero et al., 2016). TERRA is upregulated by damaged telomeres (Porro et al., 2014) and interestingly, TERRA has also been reported to regulate subtelomeric genes and internal chromosome genes, indicating a role for TERRA in modulating transcription at genome-wide scale (Chu et al., 2017). TERRA expression is modulated in response to UV radiation, heat shock and oxidative stress. The premature ageing syndrome Hutchinson-Gilford progeria syndrome (HGPS) is associated with increased expression of TERRA and tdilncRNAs (Aguado et al., 2019) and Immunodeficiency, centromeric instability and facial anomalies (ICF) syndrome type I (Yehezkel et al., 2013) and tdilncRNAs is upregulated in HGPS (Aguado et al., 2019). While TERRA expression is regulated during development (Azzalin and Lingner, 2015) and in cancers (Fernandes et al., 2020), it remains to be seen whether changes in TERRA and tdilncRNA levels are associated with aging and diseases in the general population. One study reported described modulation of TERRA expression in skin biopsies in response to the oxidative stress induced by physical exercise and may provide a mechanistic link between exercise and telomere maintenance (Diman et al., 2016).
6.3. Telomere length independent functions of telomerase
Functions of telomerase are not restricted to extension of telomeres. The telomerase protein subunit is reported to regulate the expression of several genes including NF-κb, Myc, SF1, VEGF and DNA methyl-transferases. Independent roles of telomerase were reviewed in details (Segal-Bendirdjian and Geli, 2019). Most relevant to the topic of this review is NF-κb (discussed in Inflammation). It is possible that stress hormones and ROS affect telomerase expression, and through telomerase impact expressions of these other targets.
7. Methodological considerations
Several major methods are utilized to determine telomere length, each with specific strengths and weaknesses. These methods include telomere restriction fragment (TRF) measurement by Southern blot analysis, quantitative PCR (qPCR) amplification of telomere repeats relative to a single copy gene, and fluorescent in situ hybridization (FISH) to quantify telomere repeats in individual cells or chromosomes by flow cytometry (Flow-FISH) or in metaphase cells (Q-FISH). The advantages and disadvantages of each method have been summarized by several excellent reviews recently (Nussey et al., 2014; Montpetit et al., 2014; Lai et al., 2018; Barrett et al., 2015; Aubert et al., 2012) and we will not discuss them in detail here. Because qPCR can be performed with high throughput platforms with relatively small amount of specimen at low cost, it remains the most widely used method for large-scale epidemiological and population studies. However, the high sensitivity of this assay format to the impacts of preanalytical and analytical factors cast some doubts on the accuracy and reproducibility of this method (Boardman et al., 2014; Cunningham et al., 2013; Dagnall et al., 2017; Denham et al., 2014; Hofmann et al., 2014; Lin et al., 2018; Raschenberger et al., 2016; Lindrose et al., 2021). Sample sources, DNA extraction methods and storage conditions, PCR conditions, assay re-agents and data analysis methods have all been reported to influence the final data output. While guidance and recommendations in general and for each step have been provided, based on existing and empirical knowledge (Lin et al., 2018; Morinha et al., 2020), rigorous efforts are needed to provide systematic, evidence-based recommendations for best practices for qPCR TL. Future studies should focus on understanding how each of these various factors contribute to assay variations in order to develop practical guidelines to achieve high accuracy and precision. Current efforts for interdisciplinary collaborations to advance this research is actively ongoing with the support of NIH and NIEHS (Telomere Research Network, https://trn.tulane.edu) and recommendations have been posted on the website. One recommendation so far is that the repeatability of the qPCR telomere length assay, expressed as the intra class correlation (ICC), should be calculated in replicates with the greatest possible independence (e.g. repeated samples from the same individual, repeated DNA extractions, replicates of measurement from the same DNA source) and the ICC of an individual study should be used to estimate power and sample size. A second recommendation is to use the same DNA extraction kit for all samples within a study, or to apply analytical approaches to account for differences in extraction methodologies in situations where extraction differences are unavoidable.
8. Conclusions and future directions
We have reviewed in depth the ways that psychological stress mediators may regulate telomerase activity and telomere shortening vs. repair. This research suggests the importance of the effects of stress related cortisol, ROS and inflammation. Further advances needed to understand the cellular and molecular stress-telomere maintenance interface will require experimental studies with animal models and in vitro studies with cells. With the recent advancement in the last two decades on the consistent and cross species role of telomere maintenance in aging and aging-related diseases, it is clear that the initial simplistic view of telomere length as a mitotic clock has evolved into a far more complex picture of interconnected molecular and cellular pathways and networks as hallmarks of aging. Nevertheless, it is clear that telomere maintenance is a key player in these networks. While mechanistic investigations of these pathways deepened our understanding of the contributions of telomere maintenance to aging and aging related disease, how these various factors play out in human healthy and diseased populations across the lifespan and the relative importance of critical periods such as prenatal development, childhood, menopause and andropause, and late life remains largely unknown.
Furthermore, interrogations of the telomere maintenance system as a biomarker and contributor of human health and disease need to consider the broader, global effects of telomeres and telomerase, beyond average telomere length. Longitudinal studies with systematic approaches that integrate these components are necessary. Methodological advances in measurements for telomere maintenance also require attention. In addition to better assay precision and accuracy for average telomere length in a population of cells, methods to quantify telomere length and dysfunctional telomeres at single cell resolutions should be developed and investigated in human studies.
Acknowledgments
We thank Ms. Dana Smith for insightful discussions and critical reading of the manuscript.
Acknowledgements to women in science and our own story:
Given this special issue on women in science, and our topic on psychological stress, we have a special acknowledgement. We want to acknowledge the greater hardship for women in the world, due to continued misogyny and violence toward women, and lower status given the global gaps in obtaining education and equal pay. Fortunately, the education gap is decreasing globally. Our own careers were shaped by a strong lineage of female mentorship, including the mentorship and support throughout our careers of Dr. Elizabeth Blackburn. The three of us have worked together on many of the studies reviewed in this paper, for the past nearly twenty years.
In our stress studies, one of the most common models of chronic stress used for decades, that we have used as well, is women caregivers. Women most commonly take on the majority of the burden of caregiving, from typical parenting, to caring for children with special needs, to caregiving for a parent with dementia. We are grateful to the women who have donated time (and blood) as participants in our own caregiving studies. During the past year of pandemic stressors, we acknowledge the much greater burden there has been for women in science, due to multiple roles such as caregiving, and applaud AAR and the editorial team for highlighting women scientists at this critical time.
Funding
Dr. Jue Lin is funded by NIH/NIA/NIEHS grant U01AG064785-03 and NIH/NHLBI grant 2R01HL128156-05A1. Dr. Elissa Epel is funded by grant R24AG048024 (stress network) and U24 AG066528-01 (Telomere Research Network).
References
- Abgrall S, Ingle SM, May MT, Costagliola D, Mercie P, Cavassini M, Reekie J, Samji H, Gill MJ, Crane HM, Tate J, Sterling TR, Antinori A, Reiss P, Saag MS, Mugavero MJ, Phillips A, Manzardo C, Wasmuth JC, Stephan C, Guest JL, Gomez Sirvent JL, Sterne JA, Antiretroviral Therapy Cohort Collaboration, 2013. Durability of first ART regimen and risk factors for modification, interruption or death in HIV-positive patients starting ART in Europe and North America 2002–2009. AIDS 27 (5), 803–813. 10.1097/QAD.0b013e32835cb997. [DOI] [PubMed] [Google Scholar]
- Acosta JC, O’Loghlen A, Banito A, Guijarro MV, Augert A, Raguz S, Fumagalli M, Da Costa M, Brown C, Popov N, Takatsu Y, Melamed J, d’Adda di Fagagna F, Bernard D, Hernando E, Gil J, 2008. Chemokine signaling via the CXCR2 receptor reinforces senescence. Cell 133 (6), 1006–1018. 10.1016/j.cell.2008.03.038. [DOI] [PubMed] [Google Scholar]
- Acosta JC, Banito A, Wuestefeld T, Georgilis A, Janich P, Morton JP, Athineos D, Kang TW, Lasitschka F, Andrulis M, Pascual G, Morris KJ, Khan S, Jin H, Dharmalingam G, Snijders AP, Carroll T, Capper D, Pritchard C, Inman GJ, Longerich T, Sansom OJ, Benitah SA, Zender L, Gil J, 2013. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat. Cell Biol 15 (8), 978–990. 10.1038/ncb2784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aguado J, Sola-Carvajal A, Cancila V, Revêchon G, Ong PF, Jones-Weinert CW, Walĺen Arzt E, Lattanzi G, Dreesen O, Tripodo C, Rossiello F, Eriksson M, d’Adda di Fagagna F, 2019. Inhibition of DNA damage response at telomeres improves the detrimental phenotypes of Hutchinson-Gilford Progeria Syndrome. Nat. Commun 10 (1), 4990. 10.1038/s41467-019-13018-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmed S, Passos JF, Birket MJ, Beckmann T, Brings S, Peters H, Birch-Machin MA, von Zglinicki T, Saretzki G, 2008. Telomerase does not counteract telomere shortening but protects mitochondrial function under oxidative stress. J. Cell Sci 121 (Pt 7), 1046–1053. 10.1242/jcs.019372. [DOI] [PubMed] [Google Scholar]
- Ahmed W, Lingner J, 2018. Impact of oxidative stress on telomere biology. Differentiation 99, 21–27. 10.1016/j.diff.2017.12.002. [DOI] [PubMed] [Google Scholar]
- Akiyama M, Hideshima T, Hayashi T, Tai YT, Mitsiades CS, Mitsiades N, Chauhan D, Richardson P, Munshi NC, Anderson KC, 2003. Nuclear factor-kappaB p65 mediates tumor necrosis factor alpha-induced nuclear translocation of telomerase reverse transcriptase protein. Cancer Res. 63 (1), 18–21. Epub 2003/01/09. [PubMed] [Google Scholar]
- Amano H, Chaudhury A, Rodriguez-Aguayo C, Lu L, Akhanov V, Catic A, Popov YV, Verdin E, Johnson H, Stossi F, Sinclair DA, Nakamaru-Ogiso E, Lopez-Berestein G, Chang JT, Neilson JR, Meeker A, Finegold M, Baur JA, Sahin E, 2019. Telomere dysfunction induces sirtuin repression that drives telomere-dependent disease. Cell Metab. 29 (6), 1274–1290. 10.1016/j.cmet.2019.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angelier F, Costantini D, Blevin P, Chastel O, 2018. Do glucocorticoids mediate the link between environmental conditions and telomere dynamics in wild vertebrates? A review. Gen. Comp. Endocrinol 256, 99–111. 10.1016/j.ygcen.2017.07.007. [DOI] [PubMed] [Google Scholar]
- Aravindan N, Veeraraghavan J, Madhusoodhanan R, Herman TS, Natarajan M, 2011. Curcumin regulates low-linear energy transfer gamma-radiation-induced NFkappaB-dependent telomerase activity in human neuroblastoma cells. Int. J. Radiat. Oncol. Biol. Phys 79 (4), 1206–1215. 10.1016/j.ijrobp.2010.10.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asghar M, Palinauskas V, Zaghdoudi-Allan N, Valkiūnas G, Mukhin A, Platonova E, Färnert A, Bensch S, Hasselquist D, 2016. Parallel telomere shortening in multiple body tissues owing to malaria infection. Proc. Biol. Sci 283 (1836) 10.1098/rspb.2016.1184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Asghar M, Yman V, Homann MV, Sondén K, Hammar U, Hasselquist D, Färnert A, 2018. Cellular aging dynamics after acute malaria infection: a 12-month longitudinal study. Aging Cell 17 (1). 10.1111/acel.12702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Atanasova S, Wieland E, Schlumbohm C, Korecka M, Shaw L, von Ahsen N, Fuchs E, Oellerich M, Armstrong V, 2009. Prenatal dexamethasone exposure in the common marmoset monkey enhances gene expression of antioxidant enzymes in the aorta of adult offspring. Stress 12 (3), 215–224. 10.1080/10253890802305075. [DOI] [PubMed] [Google Scholar]
- Aubert G, Hills M, Lansdorp PM, 2012. Telomere length measurement-caveats and a critical assessment of the available technologies and tools. Mutat. Res 730 (1–2), 59–67. 10.1016/j.mrfmmm.2011.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azzalin CM, Lingner J, 2015. Telomere functions grounding on TERRA firma. Trends Cell Biol. 25 (1), 29–36. 10.1016/j.tcb.2014.08.007. [DOI] [PubMed] [Google Scholar]
- Azzalin CM, Reichenbach P, Khoriauli L, Giulotto E, Lingner J, 2007. Telomeric repeat containing RNA and RNA surveillance factors at mammalian chromosome ends. Science 318 (5851), 798–801. 10.1126/science.1147182. [DOI] [PubMed] [Google Scholar]
- Bae NS, Baumann P, 2007. A RAP1/TRF2 complex inhibits nonhomologous endjoining at human telomeric DNA ends. Mol. Cell 26 (3), 323–334. 10.1016/j.molcel.2007.03.023. [DOI] [PubMed] [Google Scholar]
- Barrett JH, Iles MM, Dunning AM, Pooley KA, 2015. Telomere length and common disease: study design and analytical challenges. Hum. Genet 134 (7), 679–689. 10.1007/s00439-015-1563-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baumann P, Cech TR, 2001. Pot1, the putative telomere end-binding protein in fission yeast and humans. Science 292 (5519), 1171–1175. 10.1126/science.1060036. [DOI] [PubMed] [Google Scholar]
- Baur JA, Zou Y, Shay JW, Wright WE, 2001. Telomere position effect in human cells. Science 292 (5524), 2075–2077. 10.1126/science.1062329. [DOI] [PubMed] [Google Scholar]
- Bellon M, Nicot C, 2017. Telomere dynamics in immune senescence and exhaustion triggered by chronic viral infection. Viruses 9 (10). 10.3390/v9100289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bianchi A, Smith S, Chong L, Elias P, de Lange T, 1997. TRF1 is a dimer and bends telomeric DNA. EMBO J. 16 (7), 1785–1794. 10.1093/emboj/16.7.1785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birch J, Barnes PJ, Passos JF, 2018. Mitochondria, telomeres and cell senescence: implications for lung ageing and disease. Pharmacol. Ther 183, 34–49. 10.1016/j.pharmthera.2017.10.005. [DOI] [PubMed] [Google Scholar]
- Black CN, Bot M, Revesz D, Scheffer PG, Penninx B, 2017. The association between three major physiological stress systems and oxidative DNA and lipid damage. Psychoneuroendocrinology 80, 56–66. 10.1016/j.psyneuen.2017.03.003. [DOI] [PubMed] [Google Scholar]
- Boardman LA, Skinner HG, Litzelman K, 2014. Telomere length varies by DNA extraction method: implications for epidemiologic research–response. Cancer Epidemiol. Biomark. Prev 23 (6), 1131. 10.1158/1055-9965.EPI-14-0234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosquet Enlow M, Sideridis G, Bollati V, Hoxha M, Hacker MR, Wright RJ, 2019. Maternal cortisol output in pregnancy and newborn telomere length: evidence for sex-specific effects. Psychoneuroendocrinology 102, 225–235. 10.1016/j.psyneuen.2018.12.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Broccoli D, Smogorzewska A, Chong L, de Lange T, 1997. Human telomeres contain two distinct Myb-related proteins, TRF1 and TRF2. Nat. Genet 17 (2), 231–235. 10.1038/ng1097-231. [DOI] [PubMed] [Google Scholar]
- Bryan TM, 2020. G-Quadruplexes at telomeres: friend or foe? Molecules 25 (16). 10.3390/molecules25163686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bull C, Christensen H, Fenech M, 2015. Cortisol is not associated with telomere shortening or chromosomal instability in human lymphocytes cultured under low and high folate conditions. PLoS One 10 (3), e0119367. 10.1371/journal.pone.0119367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butler KS, Hines WC, Heaphy CM, Griffith JK, 2012. Coordinate regulation between expression levels of telomere-binding proteins and telomere length in breast carcinomas. Cancer Med. 1 (2), 165–175. 10.1002/cam4.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campisi J, Robert L, 2014. Cell senescence: role in aging and age-related diseases. Interdiscip. Top. Gerontol 39, 45–61. 10.1159/000358899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casagrande S, Stier A, Monaghan P, Loveland JL, Boner W, Lupi S, Trevisi R, Hau M, 2020. Increased glucocorticoid concentrations in early life cause mitochondrial inefficiency and short telomeres. J. Exp. Biol 223 (Pt 15) 10.1242/jeb.222513. [DOI] [PubMed] [Google Scholar]
- Cawthon RM, Smith KR, O’Brien E, Sivatchenko A, Kerber RA, 2003. Association between telomere length in blood and mortality in people aged 60 years or older. Lancet 361 (9355), 393–395. 10.1016/S0140-6736(03)12384-7. [DOI] [PubMed] [Google Scholar]
- Chakravarti D, LaBella KA, DePinho RA, 2021. Telomeres: history, health, and hallmarks of aging. Cell 184 (2), 306–322. 10.1016/j.cell.2020.12.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chatelain M, Drobniak SM, Szulkin M, 2020. The association between stressors and telomeres in non-human vertebrates: a meta-analysis. Ecol. Lett 23 (2), 381–398. 10.1111/ele.13426. [DOI] [PubMed] [Google Scholar]
- Chen R, Zhang K, Chen H, Zhao X, Wang J, Li L, Cong Y, Ju Z, Xu D, Williams BR, Jia J, Liu JP, 2015. Telomerase deficiency causes alveolar stem cell senescence-associated low-grade inflammation in lungs. J. Biol. Chem 290 (52), 30813–30829. 10.1074/jbc.M115.681619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng F, Carroll L, Joglekar MV, Januszewski AS, Wong KK, Hardikar AA, Jenkins AJ, Ma RCW, 2020. Diabetes, metabolic disease, and telomere length. Lancet Diabetes Endocrinol. 9, 117–126. 10.1016/S2213-8587(20)30365-X. [DOI] [PubMed] [Google Scholar]
- Cheng Y, Liu P, Zheng Q, Gao G, Yuan J, Wang P, Huang J, Xie L, Lu X, Tong T, Chen J, Lu Z, Guan J, Wang G, 2018. Mitochondrial trafficking and processing of telomerase RNA TERC. Cell Rep. 24 (10), 2589–2595. 10.1016/j.celrep.2018.08.003. [DOI] [PubMed] [Google Scholar]
- Cherfils-Vicini J, Iltis C, Cervera L, Pisano S, Croce O, Sadouni N, Győrffy B, Collet R, Renault VM, Rey-Millet M, Leonetti C, Zizza P, Allain F, Ghiringhelli F, Soubeiran N, Shkreli M, Vivier E, Biroccio A, Gilson E, 2019. Cancer cells induce immune escape via glycocalyx changes controlled by the telomeric protein TRF2. EMBO J. 38 (11) 10.15252/embj.2018100012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chou JP, Effros RB, 2013. T cell replicative senescence in human aging. Curr. Pharm. Des 19 (9), 1680–1698. 10.2174/138161213805219711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu HP, Cifuentes-Rojas C, Kesner B, Aeby E, Lee HG, Wei C, Oh HJ, Boukhali M, Haas W, Lee JT, 2017. TERRA RNA antagonizes ATRX and protects telomeres. Cell 170 (1), 86–101. 10.1016/j.cell.2017.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung SS, Wu Y, Okobi Q, Adekoya D, Atefi M, Clarke O, Dutta P, Vadgama JV, 2017. Proinflammatory cytokines IL-6 and TNF-alpha increased telomerase activity through NF-kappaB/STAT1/STAT3 activation, and withaferin A inhibited the signaling in colorectal cancer cells. Mediat. Inflamm 2017, 5958429 10.1155/2017/5958429. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Cohen S, Janicki-Deverts D, Miller GE, 2007. Psychological stress and disease. JAMA 298 (14), 1685–1687. 10.1001/jama.298.14.1685. [DOI] [PubMed] [Google Scholar]
- Coluzzi E, Colamartino M, Cozzi R, Leone S, Meneghini C, O’Callaghan N, Sgura A, 2014. Oxidative stress induces persistent telomeric DNA damage responsible for nuclear morphology change in mammalian cells. PLoS One 9 (10), e110963. 10.1371/journal.pone.0110963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coppe JP, Kauser K, Campisi J, Beausejour CM, 2006. Secretion of vascular endothelial growth factor by primary human fibroblasts at senescence. J. Biol. Chem 281 (40), 29568–29574. 10.1074/jbc.M603307200. [DOI] [PubMed] [Google Scholar]
- Costantini D, Marasco V, Moller AP, 2011. A meta-analysis of glucocorticoids as modulators of oxidative stress in vertebrates. J. Comp. Physiol. B 181 (4), 447–456. 10.1007/s00360-011-0566-2. [DOI] [PubMed] [Google Scholar]
- Cunningham JM, Johnson RA, Litzelman K, Skinner HG, Seo S, Engelman CD, Vanderboom RJ, Kimmel GW, Gangnon RE, Riegert-Johnson DL, Baron JA, Potter JD, Haile R, Buchanan DD, Jenkins MA, Rider DN, Thibodeau SN, Petersen GM, Boardman LA, 2013. Telomere length varies by DNA extraction method: implications for epidemiologic research. Cancer Epidemiol. Biomark. Prev 22 (11), 2047–2054. 10.1158/1055-9965.EPI-13-0409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- da Silva PFL, Ogrodnik M, Kucheryavenko O, Glibert J, Miwa S, Cameron K, Ishaq A, Saretzki G, Nagaraja-Grellscheid S, Nelson G, von Zglinicki T, 2019. The bystander effect contributes to the accumulation of senescent cells in vivo. Aging Cell 18 (1), e12848. 10.1111/acel.12848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dagarag M, Evazyan T, Rao N, Effros RB, 2004. Genetic manipulation of telomerase in HIV-specific CD8+ T cells: enhanced antiviral functions accompany the increased proliferative potential and telomere length stabilization. J. Immunol 173 (10), 6303–6311. 10.4049/jimmunol.173.10.6303. [DOI] [PubMed] [Google Scholar]
- Dagnall CL, Hicks B, Teshome K, Hutchinson AA, Gadalla SM, Khincha PP, Yeager M, Savage SA, 2017. Effect of pre-analytic variables on the reproducibility of qPCR relative telomere length measurement. PLoS One 12 (9), e0184098. 10.1371/journal.pone.0184098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daniali L, Benetos A, Susser E, Kark JD, Labat C, Kimura M, Desai K, Granick M, Aviv A, 2013. Telomeres shorten at equivalent rates in somatic tissues of adults. Nat. Commun 4, 1597. 10.1038/ncomms2602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- D’Cunha J, Knight E Jr., Haas AL, Truitt RL, Borden EC, 1996. Immunoregulatory properties of ISG15, an interferon-induced cytokine. Proc. Natl. Acad. Sci. USA 93 (1), 211–215. 10.1073/pnas.93.1.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Martinis M, Franceschi C, Monti D, Ginaldi L, 2006. Inflammation markers predicting frailty and mortality in the elderly. Exp. Mol. Pathol 80 (3), 219–227. 10.1016/j.yexmp.2005.11.004. [DOI] [PubMed] [Google Scholar]
- Denham J, Marques FZ, Charchar FJ, 2014. Leukocyte telomere length variation due to DNA extraction method. BMC Res. Notes 7, 877. 10.1186/1756-0500-7-877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diman A, Boros J, Poulain F, Rodriguez J, Purnelle M, Episkopou H, Bertrand L, Francaux M, Deldicque L, Decottignies A, 2016. Nuclear respiratory factor 1 and endurance exercise promote human telomere transcription. Sci. Adv 2 (7), e1600031 10.1126/sciadv.1600031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding D, Xi P, Zhou J, Wang M, Cong YS, 2013. Human telomerase reverse transcriptase regulates MMP expression independently of telomerase activity via NF-kappaB-dependent transcription. FASEB J. 27 (11), 4375–4383. 10.1096/fj.13-230904. [DOI] [PubMed] [Google Scholar]
- Dowd JB, Bosch JA, Steptoe A, Jayabalasingham B, Lin J, Yolken R, Aiello AE, 2017. Persistent herpesvirus infections and telomere attrition over 3 years in the whitehall II cohort. J. Infect. Dis 216 (5), 565–572. 10.1093/infdis/jix255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duckworth A, Gibbons MA, Allen RJ, Almond H, Beaumont RN, Wood AR, Lunnon K, Lindsay MA, Wain LV, Tyrrell J, Scotton CJ, 2020. Telomere length and risk of idiopathic pulmonary fibrosis and chronic obstructive pulmonary disease: a Mendelian randomisation study. Lancet Respir. Med 9, 285–294. 10.1016/S2213-2600(20)30364-7. [DOI] [PubMed] [Google Scholar]
- Entringer S, Epel ES, Lin J, Buss C, Shahbaba B, Blackburn EH, Simhan HN, Wadhwa PD, 2013. Maternal psychosocial stress during pregnancy is associated with newborn leukocyte telomere length. Am. J. Obstet. Gynecol 208 (2), 134. 10.1016/j.ajog.2012.11.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Epel ES, 2020. Can childhood adversity affect telomeres of the next generation? Possible mechanisms, implications, and next-generation research. Am. J. Psychiatry 177 (1), 7–9. 10.1176/appi.ajp.2019.19111161. [DOI] [PubMed] [Google Scholar]
- Epel ES, Prather AA, 2018. Stress, telomeres, and psychopathology: toward a deeper understanding of a triad of early aging. Annu. Rev. Clin. Psychol 14, 371–397. 10.1146/annurev-clinpsy-032816-045054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Epel S, Puterman E, Lin J, Blackburn E, Lazaro A, Mendes WB, 2013. Wandering minds and aging cells. Clin. Psychol. Sci 1 (1), 75–83. 10.1177/2167702612460234. [DOI] [Google Scholar]
- Escoter-Torres L, Caratti G, Mechtidou A, Tuckermann J, Uhlenhaut NH, Vettorazzi S, 2019. Fighting the fire: mechanisms of inflammatory gene regulation by the glucocorticoid receptor. Front. Immunol 10, 1859. 10.3389/fimmu.2019.01859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fernandes SG, Dsouza R, Pandya G, Kirtonia A, Tergaonkar V, Lee SY, Garg M, Khattar E, 2020. Role of telomeres and telomeric proteins in human malignancies and their therapeutic potential. Cancers 12 (7). 10.3390/cancers12071901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forsyth NR, Evans AP, Shay JW, Wright WE, 2003. Developmental differences in the immortalization of lung fibroblasts by telomerase. Aging Cell 2 (5), 235–243. 10.1046/j.1474-9728.2003.00057.x. [DOI] [PubMed] [Google Scholar]
- Froidure A, Mahieu M, Hoton D, Laterre PF, Yombi JC, Koenig S, Ghaye B, Defour JP, Decottignies A, 2020. Short telomeres increase the risk of severe COVID-19. Aging 12 (20), 19911–19922. 10.18632/aging.104097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furman D, Campisi J, Verdin E, Carrera-Bastos P, Targ S, Franceschi C, Ferrucci L, Gilroy DW, Fasano A, Miller GW, Miller AH, Mantovani A, Weyand CM, Barzilai N, Goronzy JJ, Rando TA, Effros RB, Lucia A, Kleinstreuer N, Slavich GM, 2019. Chronic inflammation in the etiology of disease across the life span. Nat. Med 25 (12), 1822–1832. 10.1038/s41591-019-0675-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gable DL, Gaysinskaya V, Atik CC, Talbot CC Jr., Kang B Jr, Stanley SE, Pugh EW, Amat-Codina N, Schenk KM, Arcasoy MO, Brayton C, Florea L, Armanios M, 2019. ZCCHC8, the nuclear exosome targeting component, is mutated in familial pulmonary fibrosis and is required for telomerase RNA maturation. Genes Dev. 33 (19–20), 1381–1396. 10.1101/gad.326785.119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao K, Wei C, Zhu J, Wang X, Chen G, Luo Y, Zhang D, Yue W, Yu H, 2019. Exploring the causal pathway from telomere length to Alzheimer’s disease: an update Mendelian randomization study. Front. Psychiatry 10, 843. 10.3389/fpsyt.2019.00843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao Y, Wei Y, Zhou X, Huang S, Zhao H, Zeng P, 2020. Assessing the relationship between leukocyte telomere length and cancer risk/mortality in UK Biobank and TCGA datasets with the genetic risk score and mendelian randomization approaches. Front. Genet 11, 583106 10.3389/fgene.2020.583106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ghosh A, Saginc G, Leow SC, Khattar E, Shin EM, Yan TD, Wong M, Zhang Z, Li G, Sung WK, Zhou J, Chng WJ, Li S, Liu E, Tergaonkar V, 2012. Telomerase directly regulates NF-kappaB-dependent transcription. Nat. Cell Biol 14 (12), 1270–1281. 10.1038/ncb2621. [DOI] [PubMed] [Google Scholar]
- Gidron Y, Russ K, Tissarchondou H, Warner J, 2006. The relation between psychological factors and DNA-damage: a critical review. Biol. Psychol 72 (3), 291–304. 10.1016/j.biopsycho.2005.11.011. [DOI] [PubMed] [Google Scholar]
- Gizard F, Heywood EB, Findeisen HM, Zhao Y, Jones KL, Cudejko C, Post GR, Staels B, Bruemmer D, 2011. Telomerase activation in atherosclerosis and induction of telomerase reverse transcriptase expression by inflammatory stimuli in macrophages. Arterioscler. Thromb. Vasc. Biol 31 (2), 245–252. 10.1161/ATVBAHA.110.219808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gordon DM, Santos JH, 2010. The emerging role of telomerase reverse transcriptase in mitochondrial DNA metabolism. J. Nucleic Acids 2010, 1–7. 10.4061/2010/390791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grill S, Nandakumar J, 2020. Molecular mechanisms of telomere biology disorders. J. Biol. Chem 296, 100064 10.1074/jbc.REV120.014017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Habib R, Ocklenburg S, Hoffjan S, Haghikia A, Epplen JT, Arning L, 2020. Association between shorter leukocyte telomeres and multiple sclerosis. J. Neuroimmunol 341, 577187 10.1016/j.jneuroim.2020.577187. [DOI] [PubMed] [Google Scholar]
- Haendeler J, Dröse S, Büchner N, Jakob S, Altschmied J, Goy C, Spyridopoulos I, Zeiher AM, Brandt U, Dimmeler S, 2009. Mitochondrial telomerase reverse transcriptase binds to and protects mitochondrial DNA and function from damage. Arterioscler. Thromb. Vasc. Biol 29 (6), 929–935. 10.1161/ATVBAHA.109.185546. [DOI] [PubMed] [Google Scholar]
- Haussmann MF, Longenecker AS, Marchetto NM, Juliano SA, Bowden RM, 2012. Embryonic exposure to corticosterone modifies the juvenile stress response, oxidative stress and telomere length. Proc. Biol. Sci 279 (1732), 1447–1456. 10.1098/rspb.2011.1913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He H, Multani AS, Cosme-Blanco W, Tahara H, Ma J, Pathak S, Deng Y, Chang S, 2006. POT1b protects telomeres from end-to-end chromosomal fusions and aberrant homologous recombination. EMBO J. 25 (21), 5180–5190. 10.1038/sj.emboj.7601294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helby J, Nordestgaard BG, Benfield T, Bojesen SE, 2017. Shorter leukocyte telomere length is associated with higher risk of infections: a prospective study of 75,309 individuals from the general population. Haematologica 102 (8), 1457–1465. 10.3324/haematol.2016.161943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herborn KA, Heidinger BJ, Boner W, Noguera JC, Adam A, Daunt F, Monaghan P, 2014. Stress exposure in early post-natal life reduces telomere length: an experimental demonstration in a long-lived seabird. Proc. Biol. Sci 281 (1782), 20133151 10.1098/rspb.2013.3151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herranz N, Gil J, 2018. Mechanisms and functions of cellular senescence. J. Clin. Investig 128 (4), 1238–1246. 10.1172/JCI95148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hockemeyer D, Collins K, 2015. Control of telomerase action at human telomeres. Nat. Struct. Mol. Biol 22 (11), 848–852. 10.1038/nsmb.3083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hockemeyer D, Daniels JP, Takai H, de Lange T, 2006. Recent expansion of the telomeric complex in rodents: two distinct POT1 proteins protect mouse telomeres. Cell 126 (1), 63–77. 10.1016/j.cell.2006.04.044. [DOI] [PubMed] [Google Scholar]
- Hodes RJ, Hathcock KS, Weng NP, 2002. Telomeres in T and B cells. Nat. Rev. Immunol 2 (9), 699–706. 10.1038/nri890. [DOI] [PubMed] [Google Scholar]
- Hofmann JN, Hutchinson AA, Cawthon R, Liu CS, Lynch SM, Lan Q, Rothman N, Stolzenberg-Solomon R, Purdue MP, 2014. Telomere length varies by DNA extraction method: implications for epidemiologic research-letter. Cancer Epidemiol. Biomark. Prev 23 (6), 1129–1130. 10.1158/1055-9965.EPI-14-0145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hotamisligil GS, Arner P, Caro JF, Atkinson RL, Spiegelman BM, 1995. Increased adipose tissue expression of tumor necrosis factor-alpha in human obesity and insulin resistance. J. Clin. Investig 95 (5), 2409–2415. 10.1172/JCI117936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Houghtaling BR, Cuttonaro L, Chang W, Smith S, 2004. A dynamic molecular link between the telomere length regulator TRF1 and the chromosome end protector TRF2. Curr. Biol 14 (18), 1621–1631. 10.1016/j.cub.2004.08.052. [DOI] [PubMed] [Google Scholar]
- Izano MA, Cushing LJ, Lin J, Eick SM, Goin DE, Epel E, Woodruff TJ, Morello-Frosch R, 2020. The association of maternal psychosocial stress with newborn telomere length. PLoS One 15 (12), e0242064. 10.1371/journal.pone.0242064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang Y, Da W, Qiao S, Zhang Q, Li X, Ivey G, Zilioli S, 2019. Basal cortisol, cortisol reactivity, and telomere length: a systematic review and meta-analysis. Psychoneuroendocrinology 103, 163–172. 10.1016/j.psyneuen.2019.01.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamycheva E, Goto T, Camargo CA Jr., 2017. Celiac disease autoimmunity is associated with leukocyte telomere shortening in older adults: the U.S. National Health and Nutrition Examination Survey. Exp. Gerontol 89, 64–68. 10.1016/j.exger.2017.01.003. [DOI] [PubMed] [Google Scholar]
- Kim W, Shay JW, 2018. Long-range telomere regulation of gene expression: telomere looping and telomere position effect over long distances (TPE-OLD). Differentiation 99, 1–9. 10.1016/j.diff.2017.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim W, Ludlow AT, Min J, Robin JD, Stadler G, Mender I, Lai TP, Zhang N, Wright WE, Shay JW, 2016. Regulation of the human telomerase gene TERT by Telomere Position Effect-Over Long Distances (TPE-OLD): implications for aging and cancer. PLoS Biol. 14 (12), e2000016 10.1371/journal.pbio.2000016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kordinas V, Ioannidis A, Chatzipanagiotou S, 2016. The telomere/telomerase system in chronic inflammatory diseases. Cause or effect? Genes 7 (9). 10.3390/genes7090060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krtolica A, Parrinello S, Lockett S, Desprez PY, Campisi J, 2001. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc. Natl. Acad. Sci. USA 98 (21), 12072–12077. 10.1073/pnas.211053698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo CL, Pilling LC, Kuchel GA, Ferrucci L, Melzer D, 2019. Telomere length and aging-related outcomes in humans: a Mendelian randomization study in 261,000 older participants. Aging Cell 18 (6), e13017. 10.1111/acel.13017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai TP, Wright WE, Shay JW, 2018. Comparison of telomere length measurement methods. Philos. Trans. R. Soc. Lond. B Biol. Sci 373 (1741) 10.1098/rstb.2016.0451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Lange T, 2018. Shelterin-mediated telomere protection. Annu. Rev. Genet 52, 223–247. 10.1146/annurev-genet-032918-021921. [DOI] [PubMed] [Google Scholar]
- Lewin N, Treidel LA, Holekamp KE, Place NJ, Haussmann MF, 2015. Socioecological variables predict telomere length in wild spotted hyenas. Biol. Lett 11 (2), 20140991 10.1098/rsbl.2014.0991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li C, Stoma S, Lotta LA, Warner S, Albrecht E, Allione A, Arp PP, Broer L, Buxton JL, Da Silva Couto Alves A, Deelen J, Fedko IO, Gordon SD, Jiang T, Karlsson R, Kerrison N, Loe TK, Mangino M, Milaneschi Y, Miraglio B, Pervjakova N, Russo A, Surakka I, van der Spek A, Verhoeven JE, Amin N, Beekman M, Blakemore AI, Canzian F, Hamby SE, Hottenga JJ, Jones PD, Jousilahti P, Mägi R, Medland SE, Montgomery GW, Nyholt DR, Perola M, Pietiläinen KH, Salomaa V, Sillanpää E, Suchiman HE, van Heemst D, Willemsen G, Agudo A, Boeing H, Boomsma DI, Chirlaque MD, Fagherazzi G, Ferrari P, Franks P, Gieger C, Eriksson JG, Gunter M, Hägg S, Hovatta I, Imaz L, Kaprio J, Kaaks R, Key T, Krogh V, Martin NG, Melander O, Metspalu A, Moreno C, Onland-Moret NC, Nilsson P, Ong KK, Overvad K, Palli D, Panico S, Pedersen NL, Penninx B, Quirós JR, Jarvelin MR, Rodríguez-Barranco M, Scott RA, Severi G, Slagboom PE, Spector TD, Tjonneland A, Trichopoulou A, Tumino R, Uitterlinden AG, van der Schouw YT, van Duijn CM, Weiderpass E, Denchi EL, Matullo G, Butterworth AS, Danesh J, Samani NJ, Wareham NJ, Nelson CP, Langenberg C, Codd V, 2020. Genome-wide association analysis in humans links nucleotide metabolism to leukocyte telomere length. Am. J. Hum. Genet 106 (3), 389–404. 10.1016/j.ajhg.2020.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim CJ, Cech TR, 2021. Shaping human telomeres: from shelterin and CST complexes to telomeric chromatin organization. Nat. Rev. Mol. Cell Biol 22 (4), 283–298. 10.1038/s41580-021-00328-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin J, Epel E, Cheon J, Kroenke C, Sinclair E, Bigos M, Wolkowitz O, Mellon S, Blackburn E, 2010. Analyses and comparisons of telomerase activity and telomere length in human T and B cells: insights for epidemiology of telomere maintenance. J. Immunol. Methods 352 (1–2), 71–80. 10.1016/j.jim.2009.09.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin J, Smith DL, Esteves K, Drury S, 2018. Telomere length measurement by qPCR -summary of critical factors and recommendations for assay design. Psychoneuroendocrinology 69, 68–75. 10.1016/j.psyneuen.2018.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindrose AR, McLester-Davis LWY, Tristano RI, Kataria L, Gadalla SM, Eisenberg DTA, Verhulst S, Drury S, 2021. Method comparison studies of telomere length measurement using qPCR approaches: a critical appraisal of the literature. PLoS One 16 (1), e0245582. 10.1371/journal.pone.0245582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu D, Safari A, O’Connor MS, Chan DW, Laegeler A, Qin J, Songyang Z, 2004. PTOP interacts with POT1 and regulates its localization to telomeres. Nat. Cell Biol 6 (7), 673–680. 10.1038/ncb1142. [DOI] [PubMed] [Google Scholar]
- Lopez de Silanes I, Grana O, De Bonis ML, Dominguez O, Pisano DG, Blasco MA, 2014. Identification of TERRA locus unveils a telomere protection role through association to nearly all chromosomes. Nat. Commun 5, 4723. 10.1038/ncomms5723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lou Z, Wei J, Riethman H, Baur JA, Voglauer R, Shay JW, Wright WE, 2009. Telomere length regulates ISG15 expression in human cells. Aging 1 (7), 608–621. 10.18632/aging.100066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lyu X, Sang PB, Chai W, 2021. CST in maintaining genome stability: beyond telomeres. DNA Repair 102, 103104. 10.1016/j.dnarep.2021.103104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchetto NM, Glynn RA, Ferry ML, Ostojic M, Wolff SM, Yao R, Haussmann MF, 2016. Prenatal stress and newborn telomere length. Am. J. Obstet. Gynecol 215 (1), 94. 10.1016/j.ajog.2016.01.177. [DOI] [PubMed] [Google Scholar]
- Martínez P, Gómez-López G, García F, Mercken E, Mitchell S, Flores JM, de Cabo R, Blasco MA, 2013. RAP1 protects from obesity through its extratelomeric role regulating gene expression. Cell Rep. 3 (6), 2059–2074. 10.1016/j.celrep.2013.05.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason AE, Hecht FM, Daubenmier JJ, Sbarra DA, Lin J, Moran PJ, Schleicher SG, Acree M, Prather AA, Epel ES, 2018. Weight loss maintenance and cellular aging in the supporting health through nutrition and exercise study. Psychosom. Med 80 (7), 609–619. 10.1097/PSY.0000000000000616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monteiro J, Batliwalla F, Ostrer H, Gregersen PK, 1996. Shortened telomeres in clonally expanded CD28-CD8+ T cells imply a replicative history that is distinct from their CD28+CD8+ counterparts. J. Immunol 156 (10), 3587–3590. Epub 1996/05/15. [PubMed] [Google Scholar]
- Montero JJ, Lopez de Silanes I, Grana O, Blasco MA, 2016. Telomeric RNAs are essential to maintain telomeres. Nat. Commun 7, 12534. 10.1038/ncomms12534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Montpetit AJ, Alhareeri AA, Montpetit M, Starkweather AR, Elmore LW, Filler K, Mohanraj L, Burton CW, Menzies VS, Lyon DE, Jackson-Cook CK, 2014. Telomere length: a review of methods for measurement. Nurs. Res 63 (4), 289–299. 10.1097/NNR.0000000000000037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morinha F, Magalhaes P, Blanco G, 2020. Standard guidelines for the publication of telomere qPCR results in evolutionary ecology. Mol. Ecol. Resour 20 (3) 10.1111/1755-0998.13152. [DOI] [PubMed] [Google Scholar]
- Munhoz CD, Lepsch LB, Kawamoto EM, Malta MB, Lima Lde S, Avellar MC, Sapolsky RM, Scavone C, 2006. Chronic unpredictable stress exacerbates lipopolysaccharide-induced activation of nuclear factor-kappaB in the frontal cortex and hippocampus via glucocorticoid secretion. J. Neurosci 26 (14), 3813–3820. 10.1523/JNEUROSCI.4398-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Munhoz CD, Sorrells SF, Caso JR, Scavone C, Sapolsky RM, 2010. Glucocorticoids exacerbate lipopolysaccharide-induced signaling in the frontal cortex and hippocampus in a dose-dependent manner. J. Neurosci 30 (41), 13690–13698. 10.1523/JNEUROSCI.0303-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muskens IS, Hansen HM, Smirnov IV, Molinaro AM, Bondy ML, Schildkraut JM, Wrensch M, Wiemels JL, Claus EB, 2019. Longer genotypically-estimated leukocyte telomere length is associated with increased meningioma risk. J. Neurooncol 142 (3), 479–487. 10.1007/s11060-019-03119-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen Q, Aguado J, Iannelli F, Suzuki AM, Rossiello F, d’Adda di Fagagna F, Carninci P, 2018. Target-enrichment sequencing for detailed characterization of small RNAs. Nat. Protoc 13 (4), 768–786. 10.1038/nprot.2018.001. [DOI] [PubMed] [Google Scholar]
- Noguera JC, da Silva A, Velando A, 2020. Egg corticosterone can stimulate telomerase activity and promote longer telomeres during embryo development. Mol. Ecol 10.1111/mec.15694. [DOI] [PubMed] [Google Scholar]
- Noppert GA, Feinstein L, Dowd JB, Stebbins RC, Zang E, Needham BL, Meier H, Simanek A, Aiello AE, 2020. Pathogen burden and leukocyte telomere length in the United States. Immun. Ageing 17 (1), 36. 10.1186/s12979-020-00206-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nussey DH, Baird D, Barrett E, Boner W, Fairlie J, Gemmell N, Hartmann N, Horn T, Haussmann M, Olsson M, Turbill C, Verhulst S, Zahn S, Monaghan P, 2014. Measuring telomere length and telomere dynamics in evolutionary biology and ecology. Methods Ecol. Evol 5 (4), 299–310. 10.1111/2041-210X.12161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Donovan A, Pantell MS, Puterman E, Dhabhar FS, Blackburn EH, Yaffe K, Cawthon RM, Opresko PL, Hsueh WC, Satterfield S, Newman AB, Ayonayon HN, Rubin SM, Harris TB, Epel ES, Health Aging and Body Composition, S., 2011. Cumulative inflammatory load is associated with short leukocyte telomere length in the Health, Aging and Body Composition Study. PLoS One 6 (5), e19687. 10.1371/journal.pone.0019687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oikawa S, Kawanishi S, 1999. Site-specific DNA damage at GGG sequence by oxidative stress may accelerate telomere shortening. FEBS Lett. 453 (3), 365–368. 10.1016/s0014-5793(99)00748-6. [DOI] [PubMed] [Google Scholar]
- Oliveira BS, Zunzunegui MV, Quinlan J, Fahmi H, Tu MT, Guerra RO, 2016. Systematic review of the association between chronic social stress and telomere length: a life course perspective. Ageing Res. Rev 26, 37–52. 10.1016/j.arr.2015.12.006. [DOI] [PubMed] [Google Scholar]
- Opresko PL, Fan J, Danzy S, Wilson DM 3rd, Bohr VA, 2005. Oxidative damage in telomeric DNA disrupts recognition by TRF1 and TRF2. Nucleic Acids Res. 33 (4), 1230–1239. 10.1093/nar/gki273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ost M, Noreikiene K, Angelier F, Jaatinen K, 2020. Sex-specific effects of the in ovo environment on early-life phenotypes in eiders. Oecologia 192 (1), 43–54. 10.1007/s00442-019-04569-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ouyang Q, Baerlocher G, Vulto I, Lansdorp PM, 2007. Telomere length in human natural killer cell subsets. Ann. N. Y. Acad. Sci 1106, 240–252. 10.1196/annals.1392.001. [DOI] [PubMed] [Google Scholar]
- Palm W, de Lange T, 2008. How shelterin protects mammalian telomeres. Annu. Rev. Genet 42, 301–334. 10.1146/annurev.genet.41.110306.130350. [DOI] [PubMed] [Google Scholar]
- Passos JF, Saretzki G, Ahmed S, Nelson G, Richter T, Peters H, Wappler I, Birket MJ, Harold G, Schaeuble K, Birch-Machin MA, Kirkwood TB, von Zglinicki T, 2007. Mitochondrial dysfunction accounts for the stochastic heterogeneity in telomere-dependent senescence. PLoS Biol. 5 (5), e110 10.1371/journal.pbio.0050110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Passos JF, Nelson G, Wang C, Richter T, Simillion C, Proctor CJ, Miwa S, Olijslagers S, Hallinan J, Wipat A, Saretzki G, Rudolph KL, Kirkwood TB, von Zglinicki T, 2010. Feedback between p21 and reactive oxygen production is necessary for cell senescence. Mol. Syst. Biol 6, 347. 10.1038/msb.2010.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pegan TM, Winkler DW, Haussmann MF, Vitousek MN, 2019. Brief increases in corticosterone affect morphology, stress responses, and telomere length but not postfledging movements in a wild songbird. Physiol. Biochem. Zool 92 (3), 274–285. 10.1086/702827. [DOI] [PubMed] [Google Scholar]
- Pepper GV, Bateson M, Nettle D, 2018. Telomeres as integrative markers of exposure to stress and adversity: a systematic review and meta-analysis. R. Soc. Open Sci 5 (8), 180744 10.1098/rsos.180744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petersen S, Saretzki G, von Zglinicki T, 1998. Preferential accumulation of single-stranded regions in telomeres of human fibroblasts. Exp. Cell Res 239 (1), 152–160. 10.1006/excr.1997.3893. [DOI] [PubMed] [Google Scholar]
- Pike AM, Strong MA, Ouyang JPT, Greider CW, 2019. TIN2 functions with TPP1/POT1 to stimulate telomerase processivity. Mol. Cell. Biol 39 (21) 10.1128/MCB.00593-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porro A, Feuerhahn S, Delafontaine J, Riethman H, Rougemont J, Lingner J, 2014. Functional characterization of the TERRA transcriptome at damaged telomeres. Nat. Commun 5, 5379. 10.1038/ncomms6379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prather AA, Gurfein B, Moran P, Daubenmier J, Acree M, Bacchetti P, Sinclair E, Lin J, Blackburn E, Hecht FM, Epel ES, 2015. Tired telomeres: poor global sleep quality, perceived stress, and telomere length in immune cell subsets in obese men and women. Brain Behav. Immun 47, 155–162. 10.1016/j.bbi.2014.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Protsenko E, Rehkopf D, Prather AA, Epel E, Lin J, 2020. Are long telomeres better than short? Relative contributions of genetically predicted telomere length to neoplastic and non-neoplastic disease risk and population health burden. PLoS One 15 (10), e0240185. 10.1371/journal.pone.0240185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Punder K, Heim C, Wadhwa PD, Entringer S, 2019. Stress and immunosenescence: the role of telomerase. Psychoneuroendocrinology 101, 87–100. 10.1016/j.psyneuen.2018.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pusceddu I, Herrmann W, Kleber ME, Scharnagl H, Hoffmann MM, WinklhoferRoob BM, März W, Herrmann M, 2020. Subclinical inflammation, telomere shortening, homocysteine, vitamin B6, and mortality: the Ludwigshafen Risk and Cardiovascular Health Study. Eur. J. Nutr 59 (4), 1399–1411. 10.1007/s00394-019-01993-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qian W, Kumar N, Roginskaya V, Fouquerel E, Opresko PL, Shiva S, Watkins SC, Kolodieznyi D, Bruchez MP, Van Houten B, 2019. Chemoptogenetic damage to mitochondria causes rapid telomere dysfunction. Proc. Natl. Acad. Sci. USA 116 (37), 18435–18444. 10.1073/pnas.1910574116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramlee MK, Wang J, Toh WX, Li S, 2016. Transcription regulation of the human telomerase reverse transcriptase (hTERT) gene. Genes 7 (8). 10.3390/genes7080050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raschenberger J, Lamina C, Haun M, Kollerits B, Coassin S, Boes E, Kedenko L, Köttgen A, Kronenberg F, 2016. Influence of DNA extraction methods on relative telomere length measurements and its impact on epidemiological studies. Sci. Rep 6, 25398. 10.1038/srep25398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Recht M, Borden EC, Knight E Jr., 1991. A human 15-kDa IFN-induced protein induces the secretion of IFN-gamma. J. Immunol 147 (8), 2617–2623. Epub 1991/10/25. [PubMed] [Google Scholar]
- Reichert S, Stier A, 2017. Does oxidative stress shorten telomeres in vivo? A review. Biol. Lett 13 (12) 10.1098/rsbl.2017.0463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rhee DB, Ghosh A, Lu J, Bohr VA, Liu Y, 2011. Factors that influence telomeric oxidative base damage and repair by DNA glycosylase OGG1. DNA Repair 10 (1), 34–44. 10.1016/j.dnarep.2010.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robin JD, Ludlow AT, Batten K, Magdinier F, Stadler G, Wagner KR, Shay JW, Wright WE, 2014. Telomere position effect: regulation of gene expression with progressive telomere shortening over long distances. Genes Dev. 28 (22), 2464–2476. 10.1101/gad.251041.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robin JD, Ludlow AT, Batten K, Gaillard MC, Stadler G, Magdinier F, Wright WE, Shay JW, 2015. SORBS2 transcription is activated by telomere position effect-over long distance upon telomere shortening in muscle cells from patients with facioscapulohumeral dystrophy. Genome Res. 25 (12), 1781–1790. 10.1101/gr.190660.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rossiello F, Aguado J, Sepe S, Iannelli F, Nguyen Q, Pitchiaya S, Carninci P, d’Adda di Fagagna F, 2017. DNA damage response inhibition at dysfunctional telomeres by modulation of telomeric DNA damage response RNAs. Nat. Commun 8, 13980. 10.1038/ncomms13980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rufer N, Dragowska W, Thornbury G, Roosnek E, Lansdorp PM, 1998. Telomere length dynamics in human lymphocyte subpopulations measured by flow cytometry. Nat. Biotechnol 16 (8), 743–747. 10.1038/nbt0898-743. [DOI] [PubMed] [Google Scholar]
- Rufer N, Brümmendorf TH, Kolvraa S, Bischoff C, Christensen K, Wadsworth L, Schulzer M, Lansdorp PM, 1999. Telomere fluorescence measurements in granulocytes and T lymphocyte subsets point to a high turnover of hematopoietic stem cells and memory T cells in early childhood. J. Exp. Med 190 (2), 157–167. 10.1084/jem.190.2.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahin E, Colla S, Liesa M, Moslehi J, Müller FL, Guo M, Cooper M, Kotton D, Fabian AJ, Walkey C, Maser RS, Tonon G, Foerster F, Xiong R, Wang YA, Shukla SA, Jaskelioff M, Martin ES, Heffernan TP, Protopopov A, Ivanova E, Mahoney JE, Kost-Alimova M, Perry SR, Bronson R, Liao R, Mulligan R, Shirihai OS, Chin L, DePinho RA, 2011. Telomere dysfunction induces metabolic and mitochondrial compromise. Nature 470 (7334), 359–365. 10.1038/nature09787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Salihu HM, King LM, Nwoga C, Paothong A, Pradhan A, Marty PJ, Daas R, Whiteman VE, 2016. Association between maternal-perceived psychological stress and fetal telomere length. South. Med. J 109 (12), 767–772. 10.14423/SMJ.0000000000000567. [DOI] [PubMed] [Google Scholar]
- Sanchez-Vazquez R, Guio-Carrion A, Zapatero-Gaviria A, Martinez P, Blasco MA, 2021. Shorter telomere lengths in patients with severe COVID-19 disease. Aging 13 (1), 1–15. 10.18632/aging.202463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santos JH, Meyer JN, Van Houten B, 2006. Mitochondrial localization of telomerase as a determinant for hydrogen peroxide-induced mitochondrial DNA damage and apoptosis. Hum. Mol. Genet 15 (11), 1757–1768. 10.1093/hmg/ddl098. [DOI] [PubMed] [Google Scholar]
- Scheller Madrid A, Rasmussen KL, Rode L, Frikke-Schmidt R, Nordestgaard BG, Bojesen SE, 2020. Observational and genetic studies of short telomeres and Alzheimer’s disease in 67,000 and 152,000 individuals: a Mendelian randomization study. Eur. J. Epidemiol 35 (2), 147–156. 10.1007/s10654-019-00563-w. [DOI] [PubMed] [Google Scholar]
- Schultner J, Moe B, Chastel O, Bech C, Kitaysky AS, 2014. Migration and stress during reproduction govern telomere dynamics in a seabird. Biol. Lett 10 (1), 20130889 10.1098/rsbl.2013.0889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schutte NS, Malouff JM, 2016. The relationship between perceived stress and telomere length: a meta-analysis. Stress Health 32 (4), 313–319. 10.1002/smi.2607. [DOI] [PubMed] [Google Scholar]
- Segal-Bendirdjian E, Geli V, 2019. Non-canonical roles of telomerase: unraveling the imbroglio. Front. Cell Dev. Biol 7, 332. 10.3389/fcell.2019.00332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Send TS, Gilles M, Codd V, Wolf I, Bardtke S, Streit F, Strohmaier J, Frank J, Schendel D, Sütterlin MW, Denniff M, Laucht M, Samani NJ, Deuschle M, Rietschel M, Witt SH, 2017. Telomere length in newborns is related to maternal stress during pregnancy. Neuropsychopharmacology 42 (12), 2407–2413. 10.1038/npp.2017.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sfeir A, Kosiyatrakul ST, Hockemeyer D, MacRae SL, Karlseder J, Schildkraut CL, de Lange T, 2009. Mammalian telomeres resemble fragile sites and require TRF1 for efficient replication. Cell 138 (1), 90–103. 10.1016/j.cell.2009.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma NK, Reyes A, Green P, Caron MJ, Bonini MG, Gordon DM, Holt IJ, Santos JH, 2012. Human telomerase acts as a hTR-independent reverse transcriptase in mitochondria. Nucleic Acids Res. 40 (2), 712–725. 10.1093/nar/gkr758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shin D, Shin J, Lee KW, 2019. Effects of inflammation and depression on telomere length in young adults in the United States. J. Clin. Med 8 (5) 10.3390/jcm8050711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smogorzewska A, van Steensel B, Bianchi A, Oelmann S, Schaefer MR, Schnapp G, de Lange T, 2000. Control of human telomere length by TRF1 and TRF2. Mol. Cell. Biol 20 (5), 1659–1668. 10.1128/MCB.20.5.1659-1668.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Son NH, Murray S, Yanovski J, Hodes RJ, Weng N, 2000. Lineage-specific telomere shortening and unaltered capacity for telomerase expression in human T and B lymphocytes with age. J. Immunol 165 (3), 1191–1196. 10.4049/jimmunol.165.3.1191. [DOI] [PubMed] [Google Scholar]
- Stadler G, Rahimov F, King OD, Chen JC, Robin JD, Wagner KR, Shay JW, Emerson CP Jr, Wright WE, 2013. Telomere position effect regulates DUX4 in human facioscapulohumeral muscular dystrophy. Nat. Struct. Mol. Biol 20 (6), 671–678. 10.1038/nsmb.2571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun C, Wang K, Stock AJ, Gong Y, Demarest TG, Yang B, Giri N, Harrington L, Alter BP, Savage SA, Bohr VA, Liu Y, 2020. Re-equilibration of imbalanced NAD metabolism ameliorates the impact of telomere dysfunction. EMBO J. 39 (21), e103420 10.15252/embj.2019103420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Telomeres Mendelian Randomization C, Haycock PC, Burgess S, Nounu A, Zheng J, Okoli GN, Bowden J, Wade KH, Timpson NJ, Evans DM, Willeit P, Aviv A, Gaunt TR, Hemani G, Mangino M, Ellis HP, Kurian KM, Pooley KA, Eeles RA, Lee JE, Fang S, Chen WV, Law MH, Bowdler LM, Iles MM, Yang Q, Worrall BB, Markus HS, Hung RJ, Amos CI, Spurdle AB, Thompson DJ, O’Mara TA, Wolpin B, Amundadottir L, Stolzenberg-Solomon R, Trichopoulou A, Onland-Moret NC, Lund E, Duell EJ, Canzian F, Severi G, Overvad K, Gunter MJ, Tumino R, Svenson U, van Rij A, Baas AF, Bown MJ, Samani NJ, van t’Hof F, Tromp G, Jones GT, Kuivaniemi H, Elmore JR, Johansson M, Mckay J, Scelo G, Carreras-Torres R, Gaborieau V, Brennan P, Bracci PM, Neale RE, Olson SH, Gallinger S, Li D, Petersen GM, Risch HA, Klein AP, Han J, Abnet CC, Freedman ND, Taylor PR, Maris JM, Aben KK, Kiemeney LA, Vermeulen SH, Wiencke JK, Walsh KM, Wrensch M, Rice T, Turnbull C, Litchfield K, Paternoster L, Standl M, Abecasis GR, SanGiovanni JP, Li Y, Mijatovic V, Sapkota Y, Low SK, Zondervan KT, Montgomery GW, Nyholt DR, van Heel DA, Hunt K, Arking DE, Ashar FN, Sotoodehnia N, Woo D, Rosand J, Comeau ME, Brown WM, Silverman EK, Hokanson JE, Cho MH, Hui J, Ferreira MA, Thompson PJ, Morrison AC, Felix JF, Smith NL, Christiano AM, Petukhova L, Betz RC, Fan X, Zhang X, Zhu C, Langefeld CD, Thompson SD, Wang F, Lin X, Schwartz DA, Fingerlin T, Rotter JI, Cotch MF, Jensen RA, Munz M, Dommisch H, Schaefer AS, Han F, Ollila HM, Hillary RP, Albagha O, Ralston SH, Zeng C, Zheng W, Shu XO, Reis A, Uebe S, Hüffmeier U, Kawamura Y, Otowa T, Sasaki T, Hibberd ML, Davila S, Xie G, Siminovitch K, Bei JX, Zeng YX, 2017. Association between telomere length and risk of cancer and non-neoplastic diseases: a Mendelian randomization study. JAMA Oncol. 3 (5), 636–651. 10.1001/jamaoncol.2016.5945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tissier ML, Williams TD, Criscuolo F, 2014. Maternal effects underlie ageing costs of growth in the zebra finch (Taeniopygia guttata). PLoS One 9 (5), e97705. 10.1371/journal.pone.0097705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tomiyama AJ, Milush JM, Lin J, Flynn JM, Kapahi P, Verdin E, Sinclair E, Melov S, Epel ES, 2017. Long-term calorie restriction in humans is not associated with indices of delayed immunologic aging: a descriptive study. Nutr. Healthy Aging 4 (2), 147–156. 10.3233/NHA-160017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Victorelli S, Lagnado A, Halim J, Moore W, Talbot D, Barrett K, Chapman J, Birch J, Ogrodnik M, Meves A, Pawlikowski JS, Jurk D, Adams PD, van Heemst D, Beekman M, Slagboom PE, Gunn DA, Passos JF, 2019. Senescent human melanocytes drive skin ageing via paracrine telomere dysfunction. EMBO J. 38 (23), e101982 10.15252/embj.2019101982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z, Rhee DB, Lu J, Bohr CT, Zhou F, Vallabhaneni H, de Souza-Pinto NC, Liu Y, 2010. Characterization of oxidative guanine damage and repair in mammalian telomeres. PLoS Genet. 6 (5), e1000951 10.1371/journal.pgen.1000951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weng NP, Levine BL, June CH, Hodes RJ, 1995. Human naive and memory T lymphocytes differ in telomeric length and replicative potential. Proc. Natl. Acad. Sci. USA 92 (24), 11091–11094. 10.1073/pnas.92.24.11091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weng NP, Granger L, Hodes RJ, 1997. Telomere lengthening and telomerase activation during human B cell differentiation. Proc. Natl. Acad. Sci. USA 94 (20), 10827–10832. 10.1073/pnas.94.20.10827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wikby A, Nilsson BO, Forsey R, Thompson J, Strindhall J, Löfgren S, Ernerudh J, Pawelec G, Ferguson F, Johansson B, 2006. The immune risk phenotype is associated with IL-6 in the terminal decline stage: findings from the Swedish NONA immune longitudinal study of very late life functioning. Mech. Ageing Dev 127 (8), 695–704. 10.1016/j.mad.2006.04.003. [DOI] [PubMed] [Google Scholar]
- Xu Y, Xu J, Chancoco H, Huang M, Torres KE, Gu J, 2020. Long leukocyte telomere length is associated with increased risks of soft tissue sarcoma: a Mendelian randomization study. Cancers 12 (3). 10.3390/cancers12030594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye J, Renault VM, Jamet K, Gilson E, 2014. Transcriptional outcome of telomere signalling. Nat. Rev. Genet 15 (7), 491–503. 10.1038/nrg3743. [DOI] [PubMed] [Google Scholar]
- Yeh JK, Wang CY, 2016. Telomeres and telomerase in cardiovascular diseases. Genes 7 (9). 10.3390/genes7090058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yehezkel S, Shaked R, Sagie S, Berkovitz R, Shachar-Bener H, Segev Y, Selig S, 2013. Characterization and rescue of telomeric abnormalities in ICF syndrome type I fibroblasts. Front. Oncol 3, 35. 10.3389/fonc.2013.00035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zannas AS, Kosyk O, Leung CS, 2020. Prolonged glucocorticoid exposure does not accelerate telomere shortening in cultured human fibroblasts. Genes 11 (12). 10.3390/genes11121425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng Z, Zhang W, Qian Y, Huang H, Wu DJH, He Z, Ye D, Mao Y, Wen C, 2020. Association of telomere length with risk of rheumatoid arthritis: a meta-analysis and Mendelian randomization. Rheumatology 59 (5), 940–947. 10.1093/rheumatology/kez524. [DOI] [PubMed] [Google Scholar]
- von Zglinicki T, 2002. Oxidative stress shortens telomeres. Trends Biochem. Sci 27 (7), 339–344. 10.1016/s0968-0004(02)02110-2. [DOI] [PubMed] [Google Scholar]
- Zhang C, Ostrom QT, Semmes EC, Ramaswamy V, Hansen HM, Morimoto L, de Smith AJ, Pekmezci M, Vaksman Z, Hakonarson H, Diskin SJ, Metayer C, Glioma International Case-Control Study, Taylor MD, Wiemels JL, Bondy ML, Walsh KM, 2020. Genetic predisposition to longer telomere length and risk of childhood, adolescent and adult-onset ependymoma. Acta Neuropathol. Commun 8 (1), 173. 10.1186/s40478-020-01038-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Zhao Q, Zhu W, Liu T, Xie SH, Zhong LX, Cai YY, Li XN, Liang M, Chen W, Hu QS, Zhang B, 2017. The association of telomere length in peripheral blood cells with cancer risk: a systematic review and meta-analysis of prospective studies. Cancer Epidemiol. Biomark. Prev 26 (9), 1381–1390. 10.1158/1055-9965.EPI-16-0968. [DOI] [PubMed] [Google Scholar]
- Zheng Q, Liu P, Gao G, Yuan J, Wang P, Huang J, Xie L, Lu X, Di F, Tong T, Chen J, Lu Z, Guan J, Wang G, 2019. Mitochondrion-processed TERC regulates senescence without affecting telomerase activities. Protein Cell 10 (9), 631–648. 10.1007/s13238-019-0612-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou J, Liu M, Fleming AM, Burrows CJ, Wallace SS, 2013. Neil3 and NEIL1 DNA glycosylases remove oxidative damages from quadruplex DNA and exhibit preferences for lesions in the telomeric sequence context. J. Biol. Chem 288 (38), 27263–27272. 10.1074/jbc.M113.479055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu X, Han W, Xue W, Zou Y, Xie C, Du J, Jin G, 2016. The association between telomere length and cancer risk in population studies. Sci. Rep 6, 22243. 10.1038/srep22243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu XD, Kuster B, Mann M, Petrini JH, de Lange T, 2000. Cell-cycle-regulated association of RAD50/MRE11/NBS1 with TRF2 and human telomeres. Nat. Genet 25 (3), 347–352. 10.1038/77139. [DOI] [PubMed] [Google Scholar]
- Zizza P, Dinami R, Porru M, Cingolani C, Salvati E, Rizzo A, D’Angelo C, Petti E, Amoreo CA, Mottolese M, Sperduti I, Chambery A, Russo R, Ostano P, Chiorino G, Blandino G, Sacconi A, Cherfils-Vicini J, Leonetti C, Gilson E, Biroccio A, 2019. TRF2 positively regulates SULF2 expression increasing VEGF-A release and activity in tumor microenvironment. Nucleic Acids Res. 47 (7), 3365–3382. 10.1093/nar/gkz041. [DOI] [PMC free article] [PubMed] [Google Scholar]


