Abstract
Delivering therapeutics to the central nervous system (CNS) is difficult because of the blood–brain barrier (BBB). Therapeutic delivery across the tight junctions of the BBB can be achieved through various endogenous transportation mechanisms. Receptor-mediated transcytosis (RMT) is one of the most widely investigated and used methods. Drugs can hijack RMT by expressing specific ligands that bind to receptors mediating transcytosis, such as the transferrin receptor (TfR), low-density lipoprotein receptor (LDLR), and insulin receptor (INSR). Cell-penetrating peptides and viral components originating from neurotropic viruses can also be utilized for the efficient BBB crossing of therapeutics. Exosomes, or small extracellular vesicles, have gained attention as natural nanoparticles for treating CNS diseases, owing to their potential for natural BBB crossing and broad surface engineering capability. RMT-mediated transport of exosomes expressing ligands such as LDLR-targeting apolipoprotein B has shown promising results. Although surface-modified exosomes possessing brain targetability have shown enhanced CNS delivery in preclinical studies, the successful development of clinically approved exosome therapeutics for CNS diseases requires the establishment of quantitative and qualitative methods for monitoring exosomal delivery to the brain parenchyma in vivo as well as elucidation of the mechanisms underlying the BBB crossing of surface-modified exosomes.
Keywords: exosome, brain delivery, BBB crossing, transcytosis
1. Introduction
The central nervous system (CNS) is one of the most in-demand areas for the development of new therapeutics owing to the increasing occurrence rate of neurodegenerative disorders. However, it remains the most difficult area for drug development because of the blood–brain barrier (BBB), which prevents most of the currently developed drugs from entering the brain parenchyma. The BBB functions as a tight barrier to protect the CNS from potential neurotoxic substances, and regulates the selective transport of specific molecules and nutrients to maintain CNS homeostasis. Water molecules and small ions cross brain capillaries through channels, and small molecules under 500 Da can cross the BBB via passive diffusion [1]. However, macromolecules require specific receptors or transport proteins to facilitate receptor- or adsorptive-mediated transport for entry into the brain parenchyma. The increasing need for new therapeutics for CNS diseases has prompted the investigation of various endogenous transportation mechanisms that can deliver macromolecules across the BBB. The development of novel therapeutics utilizing these transportation pathways has been actively validated in numerous preclinical and clinical studies.
Among the novel therapeutics, exosomes have recently gained attention because of their role as therapeutic vehicles for delivering various active pharmaceutical ingredients to the brain. Exosomes, or small extracellular vesicles (EVs), are a subtype of EVs defined as single-membrane lipid bilayer vesicles generated by vesicle budding into endosomes that mature into multivesicular bodies or by direct vesicle budding from the plasma membrane [2]. Different subtypes of EVs have been identified based on their size and density, which allows separation by methods such as tangential flow filtration, size exclusion chromatography, and differential centrifugation [3]. Nevertheless, careful interpretation is necessary when analyzing different groups of EVs because most EV purification methods cannot determine EVs based on their biogenesis pathways, but rather isolate subtypes of EVs based on their physical properties. Among EVs, exosomes are natural nanoparticles with low immunogenicity that can deliver diverse biological molecules, such as nucleic acids, proteins, lipids, and carbohydrates to target cells [4]. Compared with cell therapy, exosomes possess similar therapeutic efficacy with improved safety profiles in various diseases, such as cancer and ischemia [5,6,7,8,9,10]. To induce targeted delivery to the brain, therapeutic exosomes can be engineered to express various targeting moieties via direct modification methods, such as chemical modification of exosomal surfaces, or indirect modification methods via genetic engineering of exosome-producing cells. The aim of this review is to briefly discuss current engineering strategies for delivering therapeutics across the BBB and highlight recent advances in the targeted delivery of exosomes to the brain.
2. Current Strategies for Delivering Therapeutics across the BBB
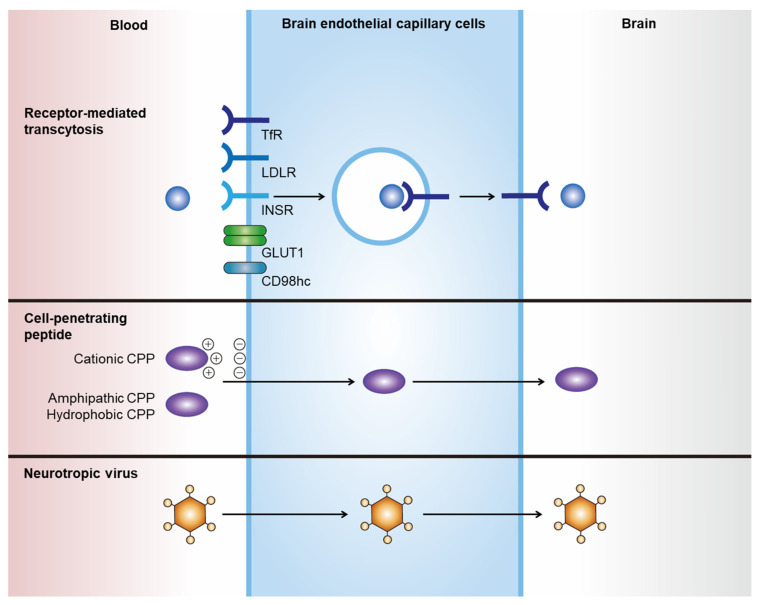
Noninvasive delivery of therapeutics to the CNS can be achieved by hijacking endogenous transport pathways, such as receptor-mediated transcytosis (RMT) and adsorptive-mediated transcytosis (Figure 1, Table 1) [11,12]. Among these, RMT has been the most investigated and applied route for the transportation of drugs through endothelial cells of the BBB [13]. Various therapeutics, including chemicals, antibodies, polymeric nanoparticles, and exosomes, can incorporate these strategies. Their efficacy in brain delivery has been actively tested in numerous preclinical studies and clinical trials [11].
Figure 1.
Strategies for delivering therapeutics across the BBB. Noninvasive delivery of therapeutics across the BBB can be achieved by hijacking endogenous transport pathways. RMT-mediated brain delivery of therapeutics can be achieved by expressing specific ligands that bind to receptors and induce transcytosis, such as TfR, LDLR, INSR, GLUT1, and CD98hc. CPPs are a family of various short peptides (fewer than 30 amino acids) that can induce the translocation of macromolecules across cell membranes without interactions with specific receptors. Neurotropic viruses can cross the BBB and invade the brain parenchyma using specific viral components, such as rabies virus glycoprotein.
Table 1.
Current strategies for delivering therapeutics across the BBB.
| BBB Crossing Strategies | Summary |
|---|---|
| Receptor-mediated transcytosis |
|
| Cell-penetrating peptides |
|
| Neurotropic virus |
|
2.1. Receptor-Mediated Transcytosis
Transcytosis is the vesicular crossing of macromolecules from one side of the cell membrane to another [14]. RMT is mediated by the binding of a ligand to a specific receptor, which subsequently induces receptor-mediated endocytosis and further transports invaginated endosomal compartments to the other side of the membrane. Drugs can hijack RMT by expressing specific ligands that bind to receptors that mediate transcytosis. The optimal receptors to be utilized for RMT-mediated BBB crossing are highly and locally expressed on the membrane of brain capillary endothelial cells (BCECs), with low expression on peripheral endothelial cells. However, to date, no ideal receptor has been identified. Nevertheless, highly and ubiquitously expressed receptors on BCECs have shown promising results in RMT-mediated brain delivery in preclinical studies and several clinical trials.
2.1.1. Transferrin Receptor
The transferrin receptor (TfR) is a widely used and validated receptor for the RMT-mediated BBB crossing of therapeutics. Transferrin is an iron-binding glycoprotein that delivers iron to cells by binding TfR. Although TfR is a ubiquitously expressed receptor, proteomics analysis confirmed that TfR is one of the highest expressed receptors that induces transcytosis in mice [17] and human BCECs [18]. The valency and binding sites of TfR-binding moieties should be carefully considered when developing a TfR-mediated brain delivery system. Recent studies have reported that TfR-targeting antibodies with high valency paradoxically have lower BBB crossing efficacy than low-valency antibodies owing to the lysosomal degradation of antibody-bound TfR [19,20]. Degradation of antibody-bound membrane proteins can also occur in other receptors for RMT-mediated CNS delivery, which warrants further investigation. In addition, it is ideal for TfR-targeting moieties to bind to regions that do not interrupt the endogenous binding of TfR to the receptor. TfR-mediated brain delivery has been applied to various therapeutics, such as liposomes [21,22,23] and chitosan nanospheres [24]. In addition, several clinical trials of TfR-mediated brain delivery of therapeutics have shown promising results. For example, clinical trials of the lysosomal enzyme iduronate 2-sulfatase conjugated with anti-human TfR antibody have shown positive results in Hunter syndrome (NCT0312893, NCT03568175, and NCT04251026).
2.1.2. Low-Density Lipoprotein Receptor
The low-density lipoprotein receptor (LDLR) family is mainly responsible for the endocytosis of low-density lipoproteins (LDLs), such as apolipoprotein B (ApoB) and apolipoprotein E (ApoE). Each LDL particle contains a single apolipoprotein surrounded by fat molecules, such as cholesterol, phospholipids, and triglycerides, which mediate the delivery of these fatty acids into cells in need. LDLR is not only a ubiquitously expressed receptor, but is also widely expressed in the brain, rendering it an efficient transporter of therapeutics. The conjugation of ApoB- and ApoE-derived peptides to proteins, such as lysosomal enzymes, has been demonstrated to successfully transport proteins across the BBB [25,26,27,28]. Nanoparticles, such as liposomes, high-density lipoprotein nanocarriers, and polymersomes, functionalized with ApoE-derived peptides, have also shown enhanced BBB crossing through LDLR- and LDLR-related protein 1 (LRP1)-mediated transcytosis in BCECs [29,30,31,32,33,34,35]. ApoB-derived peptides have also exhibited targeted delivery of siRNA and nanoparticles to the brain parenchyma [36,37]. Angiopep-2 is a 19-amino acid peptide originating from the Kunitz domain of bovine protein aprotinin, which binds to LRP1. LRP1 is widely expressed in human and mouse BCECs [18] and gliomas [38], making angiopep-2 an attractive targeting moiety for various nanoparticles for brain delivery and glioma targeting [39,40,41,42,43,44,45,46,47].
2.1.3. Insulin Receptor
Insulin receptor (INSR) is widely expressed in various tissues. A recent study comparing the expression levels of various receptors mediating transcytosis showed that only INSR was overexpressed in human brain microvessels compared to brain parenchymal and peripheral tissues [48]. A similar pattern was observed in mice, in which INSR, insulin-like growth factor-1 receptor, and LRP8 were highly expressed in brain microvessels compared to those in peripheral tissues [48]. The development of a humanized INSR antibody (HIRMAb), which showed effective delivery in the primate brain after intravenous injection, has accelerated the use of INSR for the brain delivery of drugs [49]. The lysosomal enzyme α-L-iduronidase (IDUA), which is dysfunctional in patients with mucopolysaccharidosis type I, was conjugated to HIRMAb; HIRMAb-conjugated IDUA delivered 1.2% of the injected dose to the brains of rhesus monkeys, whereas IDUA alone resulted in no delivery into the brains [50]. HIRMAb-conjugated IDUA also showed plasma pharmacokinetic profiles comparable to those of human IDUA (laronidase) [51], and an open-label phase 1–2 trial demonstrated a clinical evidence of cognitive and somatic stabilization in patients with mucopolysaccharidosis type I after 52 weeks of intravenous treatment, although few adverse effects, such as infusion-related reactions and transient hypoglycemia, occurred [52].
2.1.4. Other Membrane Proteins
CD98 heavy chain (CD98hc), also known as 4F2 antigen, is a heterodimer membrane protein consisting of a type 2-glycosylated 80-kDa heavy chain linked to a 37-kDa light chain by disulfide bonds [53,54]. CD98hc is highly expressed in human BCECs compared to TfR1, INSR, and LRP1 [18]. CD98hc is expressed on both the apical and basolateral membranes, and can bind and transport amino acids containing CD98 light chains across the BBB [55,56]. A recent study revealed via proteomic analysis that CD98hc is highly expressed in mouse BCECs and that systemic administration of bispecific antibodies targeting CD98hc and β-secretase 1 leads to efficient brain delivery and brain amyloid-beta reduction [17]. This study also showed that targeting CD98hc is more efficient in brain delivery than targeting TfR [17]. CD98hc binding did not alter the endogenous expression and function of CD98hc [17], whereas previous reports have shown that TfR antibodies induce the lysosomal degradation of TfR in an affinity-dependent manner [20].
Glucose transporter 1 (GLUT1), also known as SLC2A1, is a glucose transporter highly expressed on both the apical and basolateral membranes of BCECs [57]. The human brain depends almost entirely on glucose as an energy source, consuming approximately 20% of the total glucose in the body [58], which requires high expression of GLUT1 on the BCECs for efficient glucose transport. Glucose derivatives have been utilized for the BBB crossing of various nanoparticles, such as liposomes [59,60,61,62,63] and micelles [64,65,66], which implies that GLUT1 could be an attractive target receptor for the CNS delivery of therapeutics.
2.2. Cell-Penetrating Peptides
Cell-penetrating peptides (CPPs), or protein transduction domains, are a family of short peptides (<30 amino acids) that can induce the translocation of biologically active macromolecules across cell membranes without interacting with specific receptors [15,16]. Although no consensus has been reached regarding the taxonomy of CPPs, they can generally be categorized into three classes based on their physicochemical properties: cationic, amphipathic, and hydrophobic [15]. The cationic class is mainly composed of peptides with positive charges, such as arginine and lysine, that can interact with negatively charged plasma membranes. The transactivator of transcription (TAT) protein of HIV-1 was the first CPP observed to be internalized into cells in vitro in 1988 [67,68], and it has since been widely investigated as an inducer of intracellular delivery of therapeutics. Amphipathic CPPs are the most commonly found CPPs in nature, and they contain polar and nonpolar amino acid regions [16]. Hydrophobic CPPs contain nonpolar hydrophobic residues that induce cell penetration by interacting with the hydrophobic domains of plasma membranes. The apical surface of cerebral capillaries is densely covered with a negatively charged glycocalyx, which renders positively charged CPPs an efficient transporter of drugs through the BBB [69]. However, several issues must be addressed when using CPPs for brain delivery, such as their low tissue specificity and cellular toxicity. CPP-conjugated drugs show widespread biodistribution owing to their lack of tissue specificity. In addition, the cytotoxicity of CPPs is a major concern [70], as shown in the case of amphipathic CPP model amphipathic peptide, which induces damage to the cellular membrane, resulting in the leakage of cellular components and subsequent cell death [71].
2.3. Neurotropic Virus
Neurotropic viruses can cross the BBB and invade the brain parenchyma, which prompted the investigation of viruses or viral components as transporters for the brain delivery of therapeutics. For instance, peptides derived from rabies virus glycoprotein (RVG) exhibit efficient penetration through the BBB and target neurons [72]. Although the exact BBB crossing mechanism is unknown, it is expected to occur via neuronal acetylcholine receptor-mediated RMT [72]. RVG has been utilized for the delivery of various nanoparticles through the BBB, including liposomes [73], layered double hydroxide [74], porous silicon nanoparticles [75], and exosomes [76]. However, its biological safety and efficacy should be investigated in preclinical studies for clinical translation.
3. Targeted Delivery of Exosomes to the Brain
3.1. Natural Brain Delivery of Exosomes to the Brain
Unmodified exosomes from various cell types show <1% delivery to the brain after systemic injection [77,78], implying that exosomes have a natural tendency to bypass the BBB. This finding was also observed by our group, although the exact mechanism by which naïve exosomes cross the BBB is unknown. Recent studies have shown that exosomes originating from different parental cells have different organ and tissue tropisms [77,79,80,81]. Moreover, the specific membrane proteins or molecules of exosomes responsible for the inclination towards specific organs are not fully known. Nevertheless, altering the cell source of exosomes may be a useful strategy to induce brain delivery. Neural stem cell-derived EVs demonstrated enhanced CNS delivery compared with mesenchymal stem cell-derived EVs in a murine stroke model [82]. Based on these observations, exosomes originating from BCECs or brain tumor cells loaded with doxorubicin were tested for the targeted delivery of doxorubicin to brain tumor in a zebrafish model [83].
Transport across the BBB is enhanced under specific pathological conditions. In mice exhibiting brain inflammation, macrophage-derived exosomes showed over three-fold increased delivery to the brain compared to those in normal mice [84]. Enhanced brain delivery is achieved through the interaction of lymphocyte function-associated antigen 1, intercellular adhesion molecule 1, and C-type lectin receptors expressed on macrophage exosomes with BCECs [84]. In an in vitro transwell assay, unmodified naïve exosomes demonstrated enhanced endocytosis and subsequent crossing through BCECs in a tumor necrosis factor-α-induced stroke-like inflammation model [85]. As unmodified exosomes exhibit potential for brain delivery without additional modifications, their efficacy for BBB crossing should be further validated in preclinical studies.
3.2. Brain Delivery of Engineered Exosomes by Receptor-Mediated Transcytosis
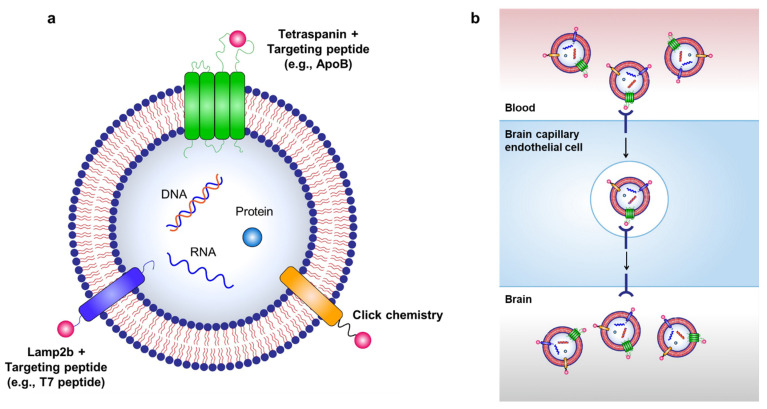
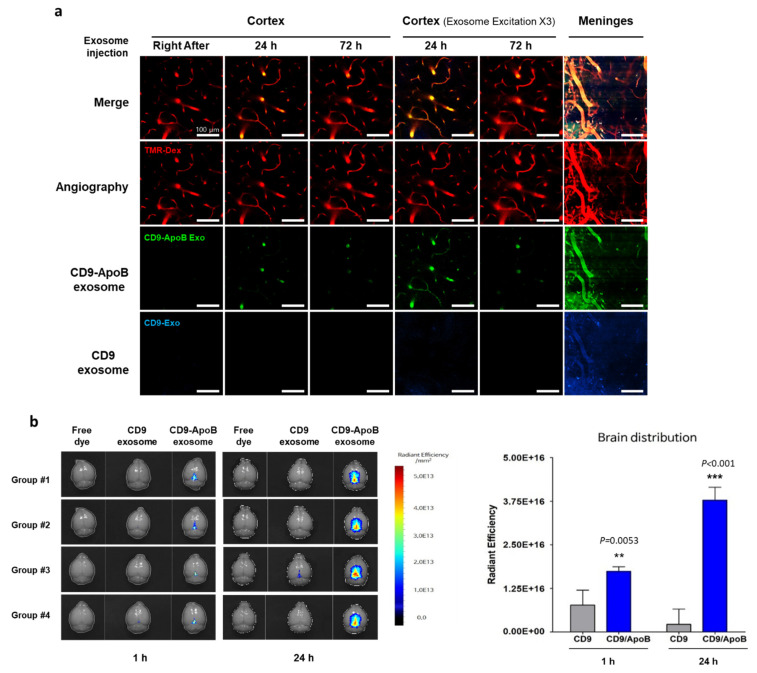
Targeted delivery of exosomes to the brain can be achieved through various exosome surface modifications (Figure 2). As hijacking RMT is a widely used strategy for delivering therapeutics across the BBB, it can also be used for transporting exosomes to the brain via labeling of targeting peptides on the surface of exosomes. For example, Kim et al. used a T7 peptide for the delivery of exosomes (T7-exo) [86]. T7 peptide is a TfR-binding peptide with the sequence HAIYPRH, which does not disturb the binding of transferrin to TfR [87,88]. By conjugating T7 peptide to Lamp2b, T7-exo demonstrated superior targeting of intracranial glioblastoma in rat models after intravenous injection compared to unmodified exosomes or RVG-labeled exosomes [86]. Recently, our group utilized the LDLR-mediated transcytosis pathway for the delivery of exosomes by generating ApoB-labeled exosomes via conjugation of ApoB with tetraspanin CD9 (unpublished data). Tetraspanins, such as CD9, CD63, CD81, and CD82, are abundant transmembrane proteins expressed on exosomes, and they consist of four membrane-spanning domains and two extracellular loops termed the short extracellular loop (SEL) and large extracellular loop (LEL) [89]. Tetraspanins can be modified to contain targeting peptides by incorporating the peptides into the extracellular loops of tetraspanin [90]. We transfected Expi293F cells with plasmids encoding CD9 as a control or CD9/LEL170-ApoB, in which ApoB was inserted between the 170–171 amino acid of CD9, to generate ApoB-expressing exosomes. Control exosomes (CD9 exosomes) or ApoB-expressing exosomes (CD9-ApoB exosomes) were isolated and purified from the supernatants of transfected Expi293F cells. To characterize the tissue distribution of the injected exosomes, we labeled CD9 exosomes with DiO and CD9-ApoB exosomes with DiD lipophilic fluorescent dyes, which were injected intravenously into mice. A laser scanning intravital confocal microscope was used to visualize the labeled exosomes in the cerebral cortex of mice by implanting a cranial window. We observed the accumulation of CD9-ApoB exosomes in the cortical blood vessels compared to CD9 exosomes, which were not detected in the vessels (Figure 3a). Next, we examined the biodistribution of surface-engineered exosomes in the mouse brain. DiD-labeled CD9 or CD9-ApoB exosomes were intravenously injected into mice, and the fluorescence intensity was analyzed using a preclinical optical imaging system. As shown in Figure 3b, the fluorescence intensity CD9-ApoB exosomes in the brain was significantly higher than that of the control CD9 exosomes, indicating prolonged retention in the brain for 24 h. These findings revealed the improved CNS-targeting capability of the surface-modified exosomes that hijacked the RMT pathway. Further studies are needed to determine the delivery efficacy of various other receptors for RMT-mediated brain delivery of exosomes.
Figure 2.
Strategies for targeted delivery of therapeutic exosomes to the brain. (a) Targeted delivery of exosomes to the brain can be achieved by labeling various targeting moieties on the surface of exosomes. Therapeutic exosomes can be engineered to express various targeting moieties via chemical modifications, such as click chemistry, or via genetic modification of exosome-producing cells to express targeting peptides fused with exosomal membrane-associated components, such as Lamp2b and tetraspanins. (b) RMT can be used to transport exosomes to the brain via labeling of targeting peptides on the surface of exosomes.
Figure 3.
Brain-targeted delivery of exosomes modified with ApoB peptide. (a) Exosomes (CD9 or CD9-ApoB) were isolated from transiently transfected Expi293F cells with CD9 or CD9/LEL170-ApoB expression vectors, respectively, and purified using an Amicon Ultra-4 Centrifugal Filter. DiO-labeled CD9 exosomes (blue) and DiD-labeled CD9-ApoB exosomes (green) were intravenously injected (at 1 × 1010 particles each) to C57BL/6 mice. The cortical vascular images of mouse brain in vivo through the cranial window were obtained using intravital confocal microscopy (IVIM Technology, Daejeon, Korea). Cerebral angiography was obtained using TMR-dextran (Red). (b) Exosomes (CD9 or CD9-ApoB) were stained with DiD, and labeled exosomes (1 × 1010 particle number/head) were intravenously injected to C57BL/6 mice. The brain distribution of exosomes was determined via fluorescence imaging by VISQUE® InVivo Smart-LF, an in vivo optical imaging system [91]. Differences between groups were compared using two-way analysis of variance with Bonferroni’s multiple comparison test. Data are expressed as mean ± SEM. ** p < 0.01, *** p < 0.001.
3.3. Other Strategies for Brain Delivery
Neurotropic virus-derived peptides, such as RVG, have been used to induce brain-targeting of exosomes in several preclinical studies. In one study, brain delivery of siRNA-loaded exosomes was achieved by expressing RVG at the exosomal membrane and fusing it with Lamp2b, an exosomal membrane protein [76]. The exact BBB crossing pathway has not been shown; however, modified exosomes demonstrated efficient delivery of siRNA to neurons, microglia, and oligodendrocytes in mouse brain [76]. In another study, the same group used a similar approach to deliver siRNA for α-synuclein (α-Syn) to the brain of α-syn transgenic mice [92]. Further studies are needed to identify safety issues associated with the use of virus-derived peptides as therapeutic agents.
Peptides that bind to specific membrane proteins can also be used for exosome modification. For example, c(RGDyK) peptide, which binds to integrin αvβ3 that is highly expressed in BCECs under ischemic conditions, was labeled on the surface of mesenchymal stem cell-derived exosomes through click chemistry [93]. Click chemistry, also known as copper-catalyzed azide-alkyne cycloaddition, is an efficient covalent reaction of an alkyne and an azide residue to form a stable triazole linkage, and can be applied to attach various targeting moieties to the surface of exosomes [94,95,96,97,98]. c(RGDyK) peptide-labeled exosomes exhibited 11-fold enhanced delivery to the ischemic region of the brain compared with scrambled peptide-labeled exosomes in a mouse stroke model [93].
4. Conclusions
Exosomes are gaining attention because of their potential as next-generation nanoparticles for treating CNS diseases owing to their potential for natural BBB crossing and broad surface-engineering capability. Various technologies to efficiently incorporate drugs and active pharmaceutical ingredients into exosomes are being actively developed [4,99,100]. In addition, various preclinical studies have investigated engineering strategies for targeted delivery of exosomes to specific organs and tissues [101]. Exosomes carry various membrane proteins (e.g., CD9 [102], CD63 [103], PTGFRN [104], and Lamp2b [76]) and lipids (e.g., phosphatidylserine [105]) that can be utilized for the surface engineering of various targeting moieties. Engineered exosomes possessing targetability to the brain have shown promising results for CNS delivery in preclinical studies; however, they also require intense evaluation through well-designed clinical trials. For the successful development of clinically approved exosome therapeutics for CNS diseases, the establishment of imaging methods for quantitative/qualitative monitoring of exosomal delivery to the brain parenchyma in vivo and uncovering the detailed BBB crossing mechanisms of exosomes is needed.
Acknowledgments
We thank Deok-Jin Jang and Cheol Hyoung Park for their constructive suggestions. We also thank Eunjin Kim for performing Nanoparticle Tracking Analysis and DiD labeling of CD9 exosomes and CD9-ApoB exosomes.
Author Contributions
Conceptualization, H.C., K.C. and H.Y.; Methodology, H.Y., D.-H.K. and B.-K.O.; Formal analysis, H.Y., D.-H.K., B.-K.O. and S.J.; Data curation, H.C., K.C., H.Y., D.-H.K. and B.-K.O.; Writing—original draft preparation, H.C. and K.C.; Writing—review and editing, C.C.; Supervision, C.C.; Project administration, C.C.; Funding acquisition, C.C. All authors have read and agreed to the published version of the manuscript.
Funding
This work was supported by the Basic Science Research Program through the National Research Foundation (NRF-2016M3A9B6945831) funded by the Ministry of Science and ICT, Republic of Korea.
Institutional Review Board Statement
Animals were purchased from the specific pathogen-free animal facility of Samtako Bio, Inc. (Osan, Korea). All animal experiments were carried out in accordance with an approved protocol (#ILIAS-IACUC-2021-02) by ILIAS Biologics, Inc. and the Institutional Animal Care and Use Committee (IACUC).
Informed Consent Statement
Not applicable.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Conflicts of Interest
C.C. is the founder and shareholder, and H.C., K.C. and H.Y.Y. are minor shareholders of ILIAS Biologics, Inc. The authors declare no additional financial interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Lipinski C.A., Lombardo F., Dominy B.W., Feeney P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 2001;46:3–26. doi: 10.1016/S0169-409X(00)00129-0. [DOI] [PubMed] [Google Scholar]
- 2.Pegtel D.M., Gould S.J. Exosomes. Annu. Rev. Biochem. 2019;88:487–514. doi: 10.1146/annurev-biochem-013118-111902. [DOI] [PubMed] [Google Scholar]
- 3.Yang D., Zhang W., Zhang H., Zhang F., Chen L., Ma L., Larcher L.M., Chen S., Liu N., Zhao Q., et al. Progress, opportunity, and perspective on exosome isolation—efforts for efficient exosome-based theranostics. Theranostics. 2020;10:3684–3707. doi: 10.7150/thno.41580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kalluri R., LeBleu V.S. The biology, function, and biomedical applications of exosomes. Science. 2020;367:eaau6977. doi: 10.1126/science.aau6977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Escudier B., Dorval T., Chaput N., Andre F., Caby M.P., Novault S., Flament C., Leboulaire C., Borg C., Amigorena S., et al. Vaccination of metastatic melanoma patients with autologous dendritic cell (DC) derived-exosomes: Results of thefirst phase I clinical trial. J. Transl. Med. 2005;3:10. doi: 10.1186/1479-5876-3-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lai R.C., Arslan F., Lee M.M., Sze N.S., Choo A., Chen T.S., Salto-Tellez M., Timmers L., Lee C.N., El Oakley R.M., et al. Exosome secreted by MSC reduces myocardial ischemia/reperfusion injury. Stem Cell Res. 2010;4:214–222. doi: 10.1016/j.scr.2009.12.003. [DOI] [PubMed] [Google Scholar]
- 7.Nikfarjam S., Rezaie J., Kashanchi F., Jafari R. Dexosomes as a cell-free vaccine for cancer immunotherapy. J. Exp. Clin. Cancer Res. 2020;39:258. doi: 10.1186/s13046-020-01781-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Babaei M., Rezaie J. Application of stem cell-derived exosomes in ischemic diseases: Opportunity and limitations. J. Transl. Med. 2021;19:196. doi: 10.1186/s12967-021-02863-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ahmadi M., Rezaie J. Ageing and mesenchymal stem cells derived exosomes: Molecular insight and challenges. Cell Biochem. Funct. 2021;39:60–66. doi: 10.1002/cbf.3602. [DOI] [PubMed] [Google Scholar]
- 10.Nam G.H., Choi Y., Kim G.B., Kim S., Kim S.A., Kim I.S. Emerging Prospects of Exosomes for Cancer Treatment: From Conventional Therapy to Immunotherapy. Adv. Mater. 2020;32:e2002440. doi: 10.1002/adma.202002440. [DOI] [PubMed] [Google Scholar]
- 11.Terstappen G.C., Meyer A.H., Bell R.D., Zhang W. Strategies for delivering therapeutics across the blood-brain barrier. Nat. Rev. Drug Discov. 2021;20:362–383. doi: 10.1038/s41573-021-00139-y. [DOI] [PubMed] [Google Scholar]
- 12.Azarmi M., Maleki H., Nikkam N., Malekinejad H. Transcellular brain drug delivery: A review on recent advancements. Int. J. Pharm. 2020;586:119582. doi: 10.1016/j.ijpharm.2020.119582. [DOI] [PubMed] [Google Scholar]
- 13.Pulgar V.M. Transcytosis to Cross the Blood Brain Barrier, New Advancements and Challenges. Front. Neurosci. 2018;12:1019. doi: 10.3389/fnins.2018.01019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tuma P., Hubbard A.L. Transcytosis: Crossing cellular barriers. Physiol. Rev. 2003;83:871–932. doi: 10.1152/physrev.00001.2003. [DOI] [PubMed] [Google Scholar]
- 15.Guidotti G., Brambilla L., Rossi D. Cell-Penetrating Peptides: From Basic Research to Clinics. Trends Pharmcol. Sci. 2017;38:406–424. doi: 10.1016/j.tips.2017.01.003. [DOI] [PubMed] [Google Scholar]
- 16.Xie J., Bi Y., Zhang H., Dong S., Teng L., Lee R.J., Yang Z. Cell-Penetrating Peptides in Diagnosis and Treatment of Human Diseases: From Preclinical Research to Clinical Application. Front. Pharmcol. 2020;11:697. doi: 10.3389/fphar.2020.00697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Zuchero Y.J., Chen X., Bien-Ly N., Bumbaca D., Tong R.K., Gao X., Zhang S., Hoyte K., Luk W., Huntley M.A., et al. Discovery of Novel Blood-Brain Barrier Targets to Enhance Brain Uptake of Therapeutic Antibodies. Neuron. 2016;89:70–82. doi: 10.1016/j.neuron.2015.11.024. [DOI] [PubMed] [Google Scholar]
- 18.Uchida Y., Ohtsuki S., Katsukura Y., Ikeda C., Suzuki T., Kamiie J., Terasaki T. Quantitative targeted absolute proteomics of human blood-brain barrier transporters and receptors. J. Neurochem. 2011;117:333–345. doi: 10.1111/j.1471-4159.2011.07208.x. [DOI] [PubMed] [Google Scholar]
- 19.Niewoehner J., Bohrmann B., Collin L., Urich E., Sade H., Maier P., Rueger P., Stracke J.O., Lau W., Tissot A.C., et al. Increased brain penetration and potency of a therapeutic antibody using a monovalent molecular shuttle. Neuron. 2014;81:49–60. doi: 10.1016/j.neuron.2013.10.061. [DOI] [PubMed] [Google Scholar]
- 20.Bien-Ly N., Yu Y.J., Bumbaca D., Elstrott J., Boswell C.A., Zhang Y., Luk W., Lu Y., Dennis M.S., Weimer R.M., et al. Transferrin receptor (TfR) trafficking determines brain uptake of TfR antibody affinity variants. J. Exp. Med. 2014;211:233–244. doi: 10.1084/jem.20131660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Sharma G., Modgil A., Layek B., Arora K., Sun C., Law B., Singh J. Cell penetrating peptide tethered bi-ligand liposomes for delivery to brain in vivo: Biodistribution and transfection. J. Control. Release. 2013;167:1–10. doi: 10.1016/j.jconrel.2013.01.016. [DOI] [PubMed] [Google Scholar]
- 22.Zhang Y., Wang Y., Boado R.J., Pardridge W.M. Lysosomal enzyme replacement of the brain with intravenous non-viral gene transfer. Pharm. Res. 2008;25:400–406. doi: 10.1007/s11095-007-9357-6. [DOI] [PubMed] [Google Scholar]
- 23.Zhang Y., Pardridge W.M. Near complete rescue of experimental Parkinson’s disease with intravenous, non-viral GDNF gene therapy. Pharm. Res. 2009;26:1059–1063. doi: 10.1007/s11095-008-9815-9. [DOI] [PubMed] [Google Scholar]
- 24.Karatas H., Aktas Y., Gursoy-Ozdemir Y., Bodur E., Yemisci M., Caban S., Vural A., Pinarbasli O., Capan Y., Fernandez-Megia E., et al. A nanomedicine transports a peptide caspase-3 inhibitor across the blood-brain barrier and provides neuroprotection. J. Neurosci. 2009;29:13761–13769. doi: 10.1523/JNEUROSCI.4246-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Spencer B.J., Verma I.M. Targeted delivery of proteins across the blood-brain barrier. Proc. Natl. Acad. Sci. USA. 2007;104:7594–7599. doi: 10.1073/pnas.0702170104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Sorrentino N.C., D’Orsi L., Sambri I., Nusco E., Monaco C., Spampanato C., Polishchuk E., Saccone P., De Leonibus E., Ballabio A., et al. A highly secreted sulphamidase engineered to cross the blood-brain barrier corrects brain lesions of mice with mucopolysaccharidoses type IIIA. EMBO Mol. Med. 2013;5:675–690. doi: 10.1002/emmm.201202083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bockenhoff A., Cramer S., Wolte P., Knieling S., Wohlenberg C., Gieselmann V., Galla H.J., Matzner U. Comparison of five peptide vectors for improved brain delivery of the lysosomal enzyme arylsulfatase A. J. Neurosci. 2014;34:3122–3129. doi: 10.1523/JNEUROSCI.4785-13.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Spencer B., Valera E., Rockenstein E., Trejo-Morales M., Adame A., Masliah E. A brain-targeted, modified neurosin (kallikrein-6) reduces alpha-synuclein accumulation in a mouse model of multiple system atrophy. Mol. Neurodegener. 2015;10:48. doi: 10.1186/s13024-015-0043-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Re F., Cambianica I., Sesana S., Salvati E., Cagnotto A., Salmona M., Couraud P.O., Moghimi S.M., Masserini M., Sancini G. Functionalization with ApoE-derived peptides enhances the interaction with brain capillary endothelial cells of nanoliposomes binding amyloid-beta peptide. J. Biotechnol. 2011;156:341–346. doi: 10.1016/j.jbiotec.2011.06.037. [DOI] [PubMed] [Google Scholar]
- 30.Re F., Cambianica I., Zona C., Sesana S., Gregori M., Rigolio R., La Ferla B., Nicotra F., Forloni G., Cagnotto A., et al. Functionalization of liposomes with ApoE-derived peptides at different density affects cellular uptake and drug transport across a blood-brain barrier model. Nanomedicine. 2011;7:551–559. doi: 10.1016/j.nano.2011.05.004. [DOI] [PubMed] [Google Scholar]
- 31.Wagner S., Zensi A., Wien S.L., Tschickardt S.E., Maier W., Vogel T., Worek F., Pietrzik C.U., Kreuter J., von Briesen H. Uptake mechanism of ApoE-modified nanoparticles on brain capillary endothelial cells as a blood-brain barrier model. PLoS ONE. 2012;7:e32568. doi: 10.1371/journal.pone.0032568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tamaru M., Akita H., Kajimoto K., Sato Y., Hatakeyama H., Harashima H. An apolipoprotein E modified liposomal nanoparticle: Ligand dependent efficiency as a siRNA delivery carrier for mouse-derived brain endothelial cells. Int. J. Pharm. 2014;465:77–82. doi: 10.1016/j.ijpharm.2014.02.016. [DOI] [PubMed] [Google Scholar]
- 33.Song Q., Song H., Xu J., Huang J., Hu M., Gu X., Chen J., Zheng G., Chen H., Gao X. Biomimetic ApoE-Reconstituted High Density Lipoprotein Nanocarrier for Blood-Brain Barrier Penetration and Amyloid Beta-Targeting Drug Delivery. Mol. Pharm. 2016;13:3976–3987. doi: 10.1021/acs.molpharmaceut.6b00781. [DOI] [PubMed] [Google Scholar]
- 34.Wang Q., Kumar V., Lin F., Sethi B., Coulter D.W., McGuire T.R., Mahato R.I. ApoE mimetic peptide targeted nanoparticles carrying a BRD4 inhibitor for treating Medulloblastoma in mice. J. Control. Release. 2020;323:463–474. doi: 10.1016/j.jconrel.2020.04.053. [DOI] [PubMed] [Google Scholar]
- 35.Ouyang J., Jiang Y., Deng C., Zhong Z., Lan Q. Doxorubicin Delivered via ApoE-Directed Reduction-Sensitive Polymersomes Potently Inhibit Orthotopic Human Glioblastoma Xenografts in Nude Mice. Int. J. Nanomed. 2021;16:4105–4115. doi: 10.2147/IJN.S314895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kim H.R., Andrieux K., Gil S., Taverna M., Chacun H., Desmaele D., Taran F., Georgin D., Couvreur P. Translocation of poly(ethylene glycol-co-hexadecyl)cyanoacrylate nanoparticles into rat brain endothelial cells: Role of apolipoproteins in receptor-mediated endocytosis. Biomacromolecules. 2007;8:793–799. doi: 10.1021/bm060711a. [DOI] [PubMed] [Google Scholar]
- 37.Spencer B., Trinh I., Rockenstein E., Mante M., Florio J., Adame A., El-Agnaf O.M.A., Kim C., Masliah E., Rissman R.A. Systemic peptide mediated delivery of an siRNA targeting alpha-syn in the CNS ameliorates the neurodegenerative process in a transgenic model of Lewy body disease. Neurobiol. Dis. 2019;127:163–177. doi: 10.1016/j.nbd.2019.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Baum L., Dong Z.Y., Choy K.W., Pang C.P., Ng H.K. Low density lipoprotein receptor related protein gene amplification and 766T polymorphism in astrocytomas. Neurosci. Lett. 1998;256:5–8. doi: 10.1016/S0304-3940(98)00734-4. [DOI] [PubMed] [Google Scholar]
- 39.Demeule M., Currie J.C., Bertrand Y., Che C., Nguyen T., Regina A., Gabathuler R., Castaigne J.P., Beliveau R. Involvement of the low-density lipoprotein receptor-related protein in the transcytosis of the brain delivery vector angiopep-2. J. Neurochem. 2008;106:1534–1544. doi: 10.1111/j.1471-4159.2008.05492.x. [DOI] [PubMed] [Google Scholar]
- 40.Regina A., Demeule M., Che C., Lavallee I., Poirier J., Gabathuler R., Beliveau R., Castaigne J.P. Antitumour activity of ANG1005, a conjugate between paclitaxel and the new brain delivery vector Angiopep-2. Br. J. Pharmcol. 2008;155:185–197. doi: 10.1038/bjp.2008.260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Ren J., Shen S., Wang D., Xi Z., Guo L., Pang Z., Qian Y., Sun X., Jiang X. The targeted delivery of anticancer drugs to brain glioma by PEGylated oxidized multi-walled carbon nanotubes modified with angiopep-2. Biomaterials. 2012;33:3324–3333. doi: 10.1016/j.biomaterials.2012.01.025. [DOI] [PubMed] [Google Scholar]
- 42.Xin H., Sha X., Jiang X., Zhang W., Chen L., Fang X. Anti-glioblastoma efficacy and safety of paclitaxel-loading Angiopep-conjugated dual targeting PEG-PCL nanoparticles. Biomaterials. 2012;33:8167–8176. doi: 10.1016/j.biomaterials.2012.07.046. [DOI] [PubMed] [Google Scholar]
- 43.Shao K., Huang R., Li J., Han L., Ye L., Lou J., Jiang C. Angiopep-2 modified PE-PEG based polymeric micelles for amphotericin B delivery targeted to the brain. J. Control. Release. 2010;147:118–126. doi: 10.1016/j.jconrel.2010.06.018. [DOI] [PubMed] [Google Scholar]
- 44.Huile G., Shuaiqi P., Zhi Y., Shijie C., Chen C., Xinguo J., Shun S., Zhiqing P., Yu H. A cascade targeting strategy for brain neuroglial cells employing nanoparticles modified with angiopep-2 peptide and EGFP-EGF1 protein. Biomaterials. 2011;32:8669–8675. doi: 10.1016/j.biomaterials.2011.07.069. [DOI] [PubMed] [Google Scholar]
- 45.He C., Zhang Z., Ding Y., Xue K., Wang X., Yang R., An Y., Liu D., Hu C., Tang Q. LRP1-mediated pH-sensitive polymersomes facilitate combination therapy of glioblastoma in vitro and in vivo. J. Nanobiotechnol. 2021;19:29. doi: 10.1186/s12951-020-00751-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Costagliola di Polidoro A., Zambito G., Haeck J., Mezzanotte L., Lamfers M., Netti P.A., Torino E. Theranostic Design of Angiopep-2 Conjugated Hyaluronic Acid Nanoparticles (Thera-ANG-cHANPs) for Dual Targeting and Boosted Imaging of Glioma Cells. Cancers. 2021;13:503. doi: 10.3390/cancers13030503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xu H., Li C., Wei Y., Zheng H., Zheng H., Wang B., Piao J.G., Li F. Angiopep-2-modified calcium arsenite-loaded liposomes for targeted and pH-responsive delivery for anti-glioma therapy. Biochem. Biophys. Res. Commun. 2021;551:14–20. doi: 10.1016/j.bbrc.2021.02.138. [DOI] [PubMed] [Google Scholar]
- 48.Zhang W., Liu Q.Y., Haqqani A.S., Leclerc S., Liu Z., Fauteux F., Baumann E., Delaney C.E., Ly D., Star A.T., et al. Differential expression of receptors mediating receptor-mediated transcytosis (RMT) in brain microvessels, brain parenchyma and peripheral tissues of the mouse and the human. Fluids Barriers CNS. 2020;17:47. doi: 10.1186/s12987-020-00209-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Pardridge W.M., Kang Y.S., Buciak J.L., Yang J. Human insulin receptor monoclonal antibody undergoes high affinity binding to human brain capillaries in vitro and rapid transcytosis through the blood-brain barrier in vivo in the primate. Pharm. Res. 1995;12:807–816. doi: 10.1023/A:1016244500596. [DOI] [PubMed] [Google Scholar]
- 50.Boado R.J., Pardridge W.M. Brain and Organ Uptake in the Rhesus Monkey in Vivo of Recombinant Iduronidase Compared to an Insulin Receptor Antibody-Iduronidase Fusion Protein. Mol. Pharm. 2017;14:1271–1277. doi: 10.1021/acs.molpharmaceut.6b01166. [DOI] [PubMed] [Google Scholar]
- 51.Pardridge W.M., Boado R.J., Giugliani R., Schmidt M. Plasma Pharmacokinetics of Valanafusp Alpha, a Human Insulin Receptor Antibody-Iduronidase Fusion Protein, in Patients with Mucopolysaccharidosis Type I. BioDrugs. 2018;32:169–176. doi: 10.1007/s40259-018-0264-7. [DOI] [PubMed] [Google Scholar]
- 52.Giugliani R., Giugliani L., de Oliveira Poswar F., Donis K.C., Corte A.D., Schmidt M., Boado R.J., Nestrasil I., Nguyen C., Chen S., et al. Neurocognitive and somatic stabilization in pediatric patients with severe Mucopolysaccharidosis Type I after 52 weeks of intravenous brain-penetrating insulin receptor antibody-iduronidase fusion protein (valanafusp alpha): An open label phase 1-2 trial. Orphanet J. Rare Dis. 2018;13:110. doi: 10.1186/s13023-018-0849-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hemler M.E., Strominger J.L. Characterization of antigen recognized by the monoclonal antibody (4F2): Different molecular forms on human T and B lymphoblastoid cell lines. J. Immunol. 1982;129:623–628. [PubMed] [Google Scholar]
- 54.Kubota H., Sato M., Ogawa Y., Iwai K., Hattori M., Yoshida T., Minato N. Involvement of 4F2 antigen expressed on the MHC-negative target cells in the recognition of murine CD3+CD4-CD8- alpha beta (V alpha 4/V beta 2) T cells. Int. Immuno.l. 1994;6:1323–1331. doi: 10.1093/intimm/6.9.1323. [DOI] [PubMed] [Google Scholar]
- 55.Matsuo H., Tsukada S., Nakata T., Chairoungdua A., Kim D.K., Cha S.H., Inatomi J., Yorifuji H., Fukuda J., Endou H., et al. Expression of a system L neutral amino acid transporter at the blood-brain barrier. Neuroreport. 2000;11:3507–3511. doi: 10.1097/00001756-200011090-00021. [DOI] [PubMed] [Google Scholar]
- 56.Feral C.C., Nishiya N., Fenczik C.A., Stuhlmann H., Slepak M., Ginsberg M.H. CD98hc (SLC3A2) mediates integrin signaling. Proc. Natl. Acad. Sci. USA. 2005;102:355–360. doi: 10.1073/pnas.0404852102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Patching S.G. Glucose Transporters at the Blood-Brain Barrier: Function, Regulation and Gateways for Drug Delivery. Mol. Neurobiol. 2017;54:1046–1077. doi: 10.1007/s12035-015-9672-6. [DOI] [PubMed] [Google Scholar]
- 58.Benarroch E.E. Brain glucose transporters: Implications for neurologic disease. Neurology. 2014;82:1374–1379. doi: 10.1212/WNL.0000000000000328. [DOI] [PubMed] [Google Scholar]
- 59.Xie F., Yao N., Qin Y., Zhang Q., Chen H., Yuan M., Tang J., Li X., Fan W., Zhang Q., et al. Investigation of glucose-modified liposomes using polyethylene glycols with different chain lengths as the linkers for brain targeting. Int. J. Nanomed. 2012;7:163–175. doi: 10.2147/IJN.S23771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Qu B., Li X., Guan M., Li X., Hai L., Wu Y. Design, synthesis and biological evaluation of multivalent glucosides with high affinity as ligands for brain targeting liposomes. Eur. J. Med. Chem. 2014;72:110–118. doi: 10.1016/j.ejmech.2013.10.007. [DOI] [PubMed] [Google Scholar]
- 61.Hao Z.F., Cui Y.X., Li M.H., Du D., Liu M.F., Tao H.Q., Li S., Cao F.Y., Chen Y.L., Lei X.H., et al. Liposomes modified with P-aminophenyl-alpha-D-mannopyranoside: A carrier for targeting cerebral functional regions in mice. Eur. J. Pharm. Biopharm. 2013;84:505–516. doi: 10.1016/j.ejpb.2012.12.020. [DOI] [PubMed] [Google Scholar]
- 62.Du D., Chang N., Sun S., Li M., Yu H., Liu M., Liu X., Wang G., Li H., Liu X., et al. The role of glucose transporters in the distribution of p-aminophenyl-alpha-d-mannopyranoside modified liposomes within mice brain. J. Control. Release. 2014;182:99–110. doi: 10.1016/j.jconrel.2014.03.006. [DOI] [PubMed] [Google Scholar]
- 63.Qin Y., Fan W., Chen H., Yao N., Tang W., Tang J., Yuan W., Kuai R., Zhang Z., Wu Y., et al. In vitro and in vivo investigation of glucose-mediated brain-targeting liposomes. J. Drug Target. 2010;18:536–549. doi: 10.3109/10611861003587235. [DOI] [PubMed] [Google Scholar]
- 64.Shao K., Zhang Y., Ding N., Huang S., Wu J., Li J., Yang C., Leng Q., Ye L., Lou J., et al. Functionalized nanoscale micelles with brain targeting ability and intercellular microenvironment biosensitivity for anti-intracranial infection applications. Adv. Healthc. Mater. 2015;4:291–300. doi: 10.1002/adhm.201400214. [DOI] [PubMed] [Google Scholar]
- 65.Guo Y., Zhang Y., Li J., Zhang Y., Lu Y., Jiang X., He X., Ma H., An S., Jiang C. Cell microenvironment-controlled antitumor drug releasing-nanomicelles for GLUT1-targeting hepatocellular carcinoma therapy. ACS Appl. Mater. Interfaces. 2015;7:5444–5453. doi: 10.1021/am5091462. [DOI] [PubMed] [Google Scholar]
- 66.Niu J., Wang A., Ke Z., Zheng Z. Glucose transporter and folic acid receptor-mediated Pluronic P105 polymeric micelles loaded with doxorubicin for brain tumor treating. J. Drug Target. 2014;22:712–723. doi: 10.3109/1061186X.2014.913052. [DOI] [PubMed] [Google Scholar]
- 67.Frankel A.D., Pabo C.O. Cellular uptake of the tat protein from human immunodeficiency virus. Cell. 1988;55:1189–1193. doi: 10.1016/0092-8674(88)90263-2. [DOI] [PubMed] [Google Scholar]
- 68.Green M., Loewenstein P.M. Autonomous functional domains of chemically synthesized human immunodeficiency virus tat trans-activator protein. Cell. 1988;55:1179–1188. doi: 10.1016/0092-8674(88)90262-0. [DOI] [PubMed] [Google Scholar]
- 69.Ando Y., Okada H., Takemura G., Suzuki K., Takada C., Tomita H., Zaikokuji R., Hotta Y., Miyazaki N., Yano H., et al. Brain-Specific Ultrastructure of Capillary Endothelial Glycocalyx and Its Possible Contribution for Blood Brain Barrier. Sci. Rep. 2018;8:17523. doi: 10.1038/s41598-018-35976-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Saar K., Lindgren M., Hansen M., Eiriksdottir E., Jiang Y., Rosenthal-Aizman K., Sassian M., Langel U. Cell-penetrating peptides: A comparative membrane toxicity study. Anal. Biochem. 2005;345:55–65. doi: 10.1016/j.ab.2005.07.033. [DOI] [PubMed] [Google Scholar]
- 71.Moutal A., Francois-Moutal L., Brittain J.M., Khanna M., Khanna R. Differential neuroprotective potential of CRMP2 peptide aptamers conjugated to cationic, hydrophobic, and amphipathic cell penetrating peptides. Front. Cell Neurosci. 2014;8:471. doi: 10.3389/fncel.2014.00471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wang Q., Cheng S., Qin F., Fu A., Fu C. Application progress of RVG peptides to facilitate the delivery of therapeutic agents into the central nervous system. RSC Adv. 2021;11:8505–8515. doi: 10.1039/D1RA00550B. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Dos Santos Rodrigues B., Arora S., Kanekiyo T., Singh J. Efficient neuronal targeting and transfection using RVG and transferrin-conjugated liposomes. Brain Res. 2020;1734:146738. doi: 10.1016/j.brainres.2020.146738. [DOI] [PubMed] [Google Scholar]
- 74.Chen W., Zuo H., Zhang E., Li L., Henrich-Noack P., Cooper H., Qian Y., Xu Z.P. Brain Targeting Delivery Facilitated by Ligand-Functionalized Layered Double Hydroxide Nanoparticles. ACS Appl. Mater. Interfaces. 2018;10:20326–20333. doi: 10.1021/acsami.8b04613. [DOI] [PubMed] [Google Scholar]
- 75.Kang J., Joo J., Kwon E.J., Skalak M., Hussain S., She Z.G., Ruoslahti E., Bhatia S.N., Sailor M.J. Self-Sealing Porous Silicon-Calcium Silicate Core-Shell Nanoparticles for Targeted siRNA Delivery to the Injured Brain. Adv. Mater. 2016;28:7962–7969. doi: 10.1002/adma.201600634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Alvarez-Erviti L., Seow Y., Yin H., Betts C., Lakhal S., Wood M.J. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 2011;29:341–345. doi: 10.1038/nbt.1807. [DOI] [PubMed] [Google Scholar]
- 77.Wiklander O.P., Nordin J.Z., O’Loughlin A., Gustafsson Y., Corso G., Mager I., Vader P., Lee Y., Sork H., Seow Y., et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J. Extracell. Vesicles. 2015;4:26316. doi: 10.3402/jev.v4.26316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Mirzaaghasi A., Han Y., Ahn S.H., Choi C., Park J.H. Biodistribution and Pharmacokinectics of Liposomes and Exosomes in a Mouse Model of Sepsis. Pharmaceutics. 2021;13:427. doi: 10.3390/pharmaceutics13030427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Sancho-Albero M., Navascues N., Mendoza G., Sebastian V., Arruebo M., Martin-Duque P., Santamaria J. Exosome origin determines cell targeting and the transfer of therapeutic nanoparticles towards target cells. J. Nanobiotechnol. 2019;17:16. doi: 10.1186/s12951-018-0437-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Qiao L., Hu S., Huang K., Su T., Li Z., Vandergriff A., Cores J., Dinh P.U., Allen T., Shen D., et al. Tumor cell-derived exosomes home to their cells of origin and can be used as Trojan horses to deliver cancer drugs. Theranostics. 2020;10:3474–3487. doi: 10.7150/thno.39434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Smyth T., Kullberg M., Malik N., Smith-Jones P., Graner M.W., Anchordoquy T.J. Biodistribution and delivery efficiency of unmodified tumor-derived exosomes. J. Control. Release. 2015;199:145–155. doi: 10.1016/j.jconrel.2014.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Webb R.L., Kaiser E.E., Scoville S.L., Thompson T.A., Fatima S., Pandya C., Sriram K., Swetenburg R.L., Vaibhav K., Arbab A.S., et al. Human Neural Stem Cell Extracellular Vesicles Improve Tissue and Functional Recovery in the Murine Thromboembolic Stroke Model. Transl. Stroke Res. 2018;9:530–539. doi: 10.1007/s12975-017-0599-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yang T.Z., Martin P., Fogarty B., Brown A., Schurman K., Phipps R., Yin V.P., Lockman P., Bai S.H. Exosome Delivered Anticancer Drugs Across the Blood-Brain Barrier for Brain Cancer Therapy in Danio Rerio. Pharm. Res. 2015;32:2003–2014. doi: 10.1007/s11095-014-1593-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Yuan D., Zhao Y., Banks W.A., Bullock K.M., Haney M., Batrakova E., Kabanov A.V. Macrophage exosomes as natural nanocarriers for protein delivery to inflamed brain. Biomaterials. 2017;142:1–12. doi: 10.1016/j.biomaterials.2017.07.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Chen C.C., Liu L.N., Ma F.X., Wong C.W., Guo X.N.E., Chacko J.V., Farhoodi H.P., Zhang S.X., Zimak J., Segaliny A., et al. Elucidation of Exosome Migration Across the Blood-Brain Barrier Model In Vitro. Cell. Mol. Bioeng. 2016;9:509–529. doi: 10.1007/s12195-016-0458-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Kim G., Kim M., Lee Y., Byun J.W., Hwang D.W., Lee M. Systemic delivery of microRNA-21 antisense oligonucleotides to the brain using T7-peptide decorated exosomes. J. Control. Release. 2020;317:273–281. doi: 10.1016/j.jconrel.2019.11.009. [DOI] [PubMed] [Google Scholar]
- 87.Lee J.H., Engler J.A., Collawn J.F., Moore B.A. Receptor mediated uptake of peptides that bind the human transferrin receptor. Eur. J. Biochem. 2001;268:2004–2012. doi: 10.1046/j.1432-1327.2001.02073.x. [DOI] [PubMed] [Google Scholar]
- 88.Han L., Huang R., Liu S., Huang S., Jiang C. Peptide-conjugated PAMAM for targeted doxorubicin delivery to transferrin receptor overexpressed tumors. Mol. Pharm. 2010;7:2156–2165. doi: 10.1021/mp100185f. [DOI] [PubMed] [Google Scholar]
- 89.Umeda R., Satouh Y., Takemoto M., Nakada-Nakura Y., Liu K., Yokoyama T., Shirouzu M., Iwata S., Nomura N., Sato K., et al. Structural insights into tetraspanin CD9 function. Nat. Commun. 2020;11:1606. doi: 10.1038/s41467-020-15459-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Khan H., Pan J.J., Li Y., Zhang Z., Yang G.Y. Native and Bioengineered Exosomes for Ischemic Stroke Therapy. Front. Cell Dev. Biol. 2021;9:619565. doi: 10.3389/fcell.2021.619565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Jeong H., Kim S.-R., Kang Y., Kim H., Kim S.-Y., Cho S.-H., Kim K.-N. Real-Time Longitudinal Evaluation of Tumor Blood Vessels Using a Compact Preclinical Fluorescence Imaging System. Biosensors. 2021;11:471. doi: 10.3390/bios11120471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Cooper J.M., Wiklander P.B., Nordin J.Z., Al-Shawi R., Wood M.J., Vithlani M., Schapira A.H., Simons J.P., El-Andaloussi S., Alvarez-Erviti L. Systemic exosomal siRNA delivery reduced alpha-synuclein aggregates in brains of transgenic mice. Mov. Disord. 2014;29:1476–1485. doi: 10.1002/mds.25978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Tian T., Zhang H.X., He C.P., Fan S., Zhu Y.L., Qi C., Huang N.P., Xiao Z.D., Lu Z.H., Tannous B.A., et al. Surface functionalized exosomes as targeted drug delivery vehicles for cerebral ischemia therapy. Biomaterials. 2018;150:137–149. doi: 10.1016/j.biomaterials.2017.10.012. [DOI] [PubMed] [Google Scholar]
- 94.Smyth T., Petrova K., Payton N.M., Persaud I., Redzic J.S., Graner M.W., Smith-Jones P., Anchordoquy T.J. Surface functionalization of exosomes using click chemistry. Bioconjug. Chem. 2014;25:1777–1784. doi: 10.1021/bc500291r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Ramasubramanian L., Kumar P., Wang A. Engineering Extracellular Vesicles as Nanotherapeutics for Regenerative Medicine. Biomolecules. 2019;10:48. doi: 10.3390/biom10010048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Algar W.R., Prasuhn D.E., Stewart M.H., Jennings T.L., Blanco-Canosa J.B., Dawson P.E., Medintz I.L. The controlled display of biomolecules on nanoparticles: A challenge suited to bioorthogonal chemistry. Bioconjug. Chem. 2011;22:825–858. doi: 10.1021/bc200065z. [DOI] [PubMed] [Google Scholar]
- 97.Villata S., Canta M., Cauda V. EVs and Bioengineering: From Cellular Products to Engineered Nanomachines. Int. J. Mol. Sci. 2020;21:6048. doi: 10.3390/ijms21176048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Nwe K., Brechbiel M.W. Growing applications of “click chemistry” for bioconjugation in contemporary biomedical research. Cancer Biother. Radiopharm. 2009;24:289–302. doi: 10.1089/cbr.2008.0626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Song Y., Kim Y., Ha S., Sheller-Miller S., Yoo J., Choi C., Park C.H. The emerging role of exosomes as novel therapeutics: Biology, technologies, clinical applications, and the next. Am. J. Reprod. Immunol. 2021;85:e13329. doi: 10.1111/aji.13329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Herrmann I.K., Wood M.J.A., Fuhrmann G. Extracellular vesicles as a next-generation drug delivery platform. Nat. Nanotechnol. 2021;16:748–759. doi: 10.1038/s41565-021-00931-2. [DOI] [PubMed] [Google Scholar]
- 101.Choi H., Choi Y., Yim H.Y., Mirzaaghasi A., Yoo J.K., Choi C. Biodistribution of Exosomes and Engineering Strategies for Targeted Delivery of Therapeutic Exosomes. Tissue Eng. Regen. Med. 2021;18:499–511. doi: 10.1007/s13770-021-00361-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Yim N., Ryu S.W., Choi K., Lee K.R., Lee S., Choi H., Kim J., Shaker M.R., Sun W., Park J.H., et al. Exosome engineering for efficient intracellular delivery of soluble proteins using optically reversible protein-protein interaction module. Nat. Commun. 2016;7:12277. doi: 10.1038/ncomms12277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Stickney Z., Losacco J., McDevitt S., Zhang Z., Lu B. Development of exosome surface display technology in living human cells. Biochem. Biophys. Res. Commun. 2016;472:53–59. doi: 10.1016/j.bbrc.2016.02.058. [DOI] [PubMed] [Google Scholar]
- 104.Dooley K., McConnell R.E., Xu K., Lewis N.D., Haupt S., Youniss M.R., Martin S., Sia C.L., McCoy C., Moniz R.J., et al. A versatile platform for generating engineered extracellular vesicles with defined therapeutic properties. Mol. Ther. 2021;29:1729–1743. doi: 10.1016/j.ymthe.2021.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Skotland T., Hessvik N.P., Sandvig K., Llorente A. Exosomal lipid composition and the role of ether lipids and phosphoinositides in exosome biology. J. Lipid Res. 2019;60:9–18. doi: 10.1194/jlr.R084343. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.