Abstract
Plant aldehyde oxidases (AOs) are multi-functional enzymes, and they could oxidize abscisic aldehyde into ABA (abscisic acid) or indole acetaldehyde into IAA (indoleacetic acid) as the last step, respectively. AOs can be divided into four groups based on their biochemical and physiological functions. In this review, we summarized the recent studies about AOs in plants including the motif information, biochemical, and physiological functions. Besides their role in phytohormones biosynthesis and stress response, AOs could also involve in reactive oxygen species homeostasis, aldehyde detoxification and stress tolerance.
Keywords: Aldehyde oxidases, Abscisic acid, Indoleacetic acid, Stress
Introduction
Abscisic acid (ABA) acts as a ubiquitous signal that regulates diverse processes and plant responses, including seed maturation/germination, dormancy, leaf senescence, stomatal aperture, apical dominance, and the regulation of phototropic and gravitropic behaviour, as well as adaptation to a variety of environmental stresses, such as drought and salinity (Verslues & Zhu, 2005; Normanly, Slovin & Cohen, 1995; Merlot & Giraudat, 1997; Srivastava et al., 2017).
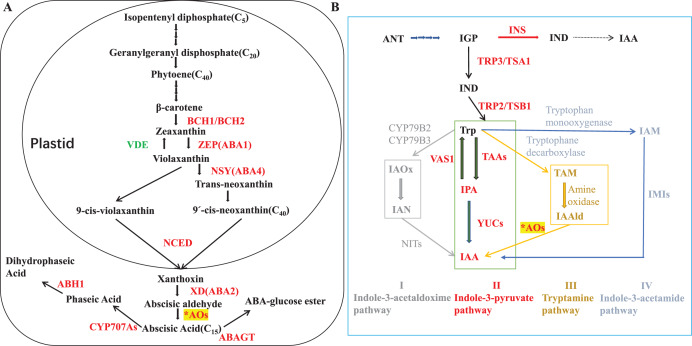
ABA biosynthetic genes and enzymes have been identified in various plants species (Taylor, Burbidge & Thompson, 2000; Milborrow, 2001; Seo & Koshiba, 2002; Nambara & Marion-Poll, 2005). ABA synthesis starts from C40 epoxycarotenoids that derived from isopentenyl diphosphate in methylerythritol phosphate pathway. Zeaxanthin epoxidase (ZEP) is responsible for catalyzing the two-step epoxidation of all-trans-zeaxanthin to all-trans-violaxanthin (Finkelstein, 2013; Marin et al., 1996; Audran et al., 1998, 2001; Xiong, Schumaker & Jian-Kang, 2002). The latter is then converted into the cis-isomers of neoxanthin and violaxanthin, which are cleaved by a 9-cis-epoxycarotenoid dioxygenase (NCED) to its direct precursor, xanthoxin (Schwartz et al., 1997; Qin & Zeevaart, 1999), and this oxidative cleavage process is a key rate-limiting step for ABA biosynthesis (Estrada-Melo et al., 2015). Conversion of xanthoxin to abscisic aldehyde is catalyzed by a short-chain dehydrogenase/reductase (SDR) (Cheng et al., 2002; González-Guzmán et al., 2002), and aldehyde oxidases (AOs; EC 1.2.3.1) oxidize abscisic aldehyde into ABA as the final step (Seo et al., 2000a, 2000b; Barrero et al., 2006; Melhorn et al., 2008; Ando et al., 2006). Pathway for ABA biosynthesis was summarized in Fig. 1A.
Figure 1. ABA and IAA biosynthesis.
(A) ABA precursor is synthesized from the methylerythritol phosphate (MEP) pathway. Enzymes are shown in red colour. BCH1/BCH2: β-carotene hydroxylases; ZEP: Zeaxanthin epoxidase; NSY: Neoxanthin synthase; NCED: 9-cis-epoxycarotenoid dioxygenase; XD: Xanthoxin dehydrogenase; ABAO: Abscisic aldehyde oxidase; CYP707A: ABA 8′-hydroxylase; ABH1: Phaseic acid reductase 1; ABAGT: ABA glucosyltransferase; βG: β-glucosidase; VDE: violaxanthin de-epoxidase, AOs: aldehyde oxidases were indicated with an asterisk (*). Adapted from Dejonghe, Okamoto & Cutler (2018); Song et al. (2020b); Finkelstein (2013). (B) ANT, anthranilate; IAA, indole-3-acetic acid; IAAld, indole-3-acetaldehyde; IAN, indole-3-acetonitrile; IGP, indole-3-glycerol phosphate; IND, indole; AAO, aldehydeoxidase; CYP79B2/3, cytochrome P450 monooxygenases2/3; IMI, amidase; INS, indole synthase; NIT, nitrilase; TAA, tryptophan aminotransferase; TRP3/TSA1, Trp synthase α-subunit; TRP2/TSB1, Trp synthase β-subunit; VAS1, pyridoxal phosphate-dependentaminotransferase1; YUC, YUCCA flavin-containing monooxygenase. Adapted from Kasahara (2016); Song et al. (2020a).
AOs are widespread cytosolic molybdo-iron-flavo enzymes that oxidize a variety of aldehydes to their corresponding carboxylic acids (Koshiba et al., 1996; Ori et al., 1997; Seo et al., 1998, 2000a, 2000b; Garattini, Fratelli & Terao, 2008; Zdunek-Zastocka, 2008). Except involved in ABA synthesis, AO enzyme could also oxidize indole acetaldehyde into indoleacetic acid (IAA, most important form of auxin). Though the tryptophan (Trp)-independent pathway has been proposed for IAA biosynthesis alternatively, Trp-dependent pathway is the main one including four proposed pathways for IAA biosynthesis in plants: (I) indole-3-acetaldoxime (IAOx) pathway, (II) the indole-3-pyruvic acid (IPA) pathway, (III) tryptamine (TAM) pathway, and (IV) the indole-3-acetamide (IAM) pathway. Based on evolutionarily conserved core mechanisms, it is thought that the pathway via IAM or IPA are the major route(s) to IAA in plants (Mano & Nemoto, 2012). AO is responsible for the final step of IPA pathway in auxin biosynthesis (Fig. 1B). Auxin is deeply implicated in most aspects of plant growth and development, and the detail of AO in IAA production will discuss in the part of aldehyde oxidase 1 later.
The nucleotide and amino acid of the AO multigene family shares high similarity with xanthine dehydrogenase (XDH), and AO is likely derived from XDH by gene duplication and neofunctionalization, though their function could be divergent. Both AO and XDH comprise FAD, Fe-S, and molybdenum cofactor (Moco) as prosthetic groups (Koshiba et al., 1996; Hille, Nishinoab & Bittner, 2011). However, different physiological electron acceptors distinguish AO and XDH enzymes: AO enzymes does not contain NAD-binding site, and AO can exclusively use molecular oxygen as a physiological electron acceptor and XDH use NAD+ as the final electron acceptor (Nishino & Nishino, 1989; Turner, Weiner & Tayior, 1995).
Plants AOs catalyze the oxidation of a variety of different aromatic and aliphatic aldehydes (Zdunek-Zastocka et al., 2004). The conversion of abscisic aldehyde to ABA and indole acetaldehyde into indoleacetic acid by AOs, respectively, may constitute important regulatory element; however, its regulatory role is still being debated and we summarize the biochemical and physiological function of AO1, AO2, AO3, and AO4.
Survey Methodology
Primary and secondary literature relevant to the topic of this paper was assessed using Web of Science (Clarivate Analytics). Key words such as “aldehyde oxidase,” “abscisic acid,” “indoleacetic acid,” “plants” and “stress,” were searched between 1 March and 31 October, 2021.
The information of stress induced genes, interaction proteins and function predictions were generated with the interaction viewer at bar.utoronto.ca/eplant by Waese et al. (2017). The detail of experiment information can be found on this website. Take AO1 gene (AT5G20960) in Arabidopsis as an example, search the gene number, and choose Abiotic Stress II eFP, and we could find the total RNA and polysomal mRNA level was upregulated after hypoxia stress.
Biochemical and physiological function of AO1
Plant AOs have been studied from several sources, including oat (Avena sativa) coleoptiles (Rajagopal, 1971), potato tubers (Rothe, 1974), cucumber (Cucumis sativus) seedlings (Bower, Brown & Purves, 1978), pea seedlings (Miyata et al., 1981; Zdunek-Zastocka, 2010), and maize (Zea mays L.) coleoptiles (Koshiba et al., 1996). Both animal and plant AOs possess relatively wide substrate specificity and can oxidize a number of different aldehydes, and this trait indicates that AOs have different biological roles (Koshiba et al., 1996; Ori et al., 1997; Akaba et al., 1999; Omarov et al., 1999; Koiwai et al., 2000, 2004; Seo et al., 2000a; Ibdah et al., 2009). AO isoforms have been largely characterized in Arabidopsis, and the AO gene family consists of at least four aldehyde oxidase genes, AO1–AO4 by forming homodimers and heterodimers (Akaba et al., 1999; Koiwai et al., 2000, 2004; Seo et al., 2000a; Zdunek-Zastocka et al., 2004).
In Arabidopsis AOα (homodimers of AAO1) showed a relatively high efficiency for indole-3-acetaldehyde, a precursor of IAA and it indicates AOα is a possible candidate aldehyde oxidase for IAA biosynthesis, and this activity is higher in IAA-overproducing sur1 mutant seedlings (Akaba et al., 1999; Seo et al., 1998). The study of AO in maize also confirms its role in IAA biosynthesis (Koshiba et al., 1996; Sekimoto et al., 1997). Fedorova et al. (2005) proposed that the local synthesis of IAA in the root nodule meristem and the modulation of AO expression and activity are involved in the regulation of Lupinus albus and Medicago truncatula nodule development. Two isoforms, BrAO-a, and BrAO-d, were suggested to be involved in auxin overproduction during clubroot development induced by pathogen infection in Chinese cabbage (Ando et al., 2006). In Arabidopsis, the expression patterns of AO1 are tissue specific and are expressed predominantly in seedlings, roots, and seeds.
Based on the publicly available microarray data (http://bar.utoronto.ca/eplant/, Kilian et al., 2007), we can predict AO1 might be involved in hypoxia response in Arabidopsis because it was induced significantly by hypoxia stress. Waterlogging or flooding is one of the main abiotic stresses that limit crops production and deficiency of oxygen is the main reason responsible for the damages caused by waterlogging or flooding stress, and according to the extent of lack of oxygen, it can be divided into anoxia (nearly no oxygen) or hypoxia (low oxygen). In most cases, waterlogging or flooding stress cause hypoxia conditions and it is detrimental to plants due to the energy crisis caused by anaerobic respiration and the accumulation of toxic substances (Xu, Pan & Zhang, 2020).
Biochemical and physiological function of AO2
AO2 is highly expressed in hypocotyls, seedlings, roots and senescing leaves. In Arabidopsis, AO2 homodimers have a strong preference for 1-naphthaldehyde as a substrate, which is oxidized to 1-naphthyl acetic acid, whereas the AO1/AO2 heterodimer has a preference for indole-3-acetaldehyde and 1-naphthyl acetic acid (Akaba et al., 1999). The deduced amino acid sequence of aldehyde oxidase 2 (AhAO2) in Arachis hypogaea L. showed high similarity with other plant AOs. AhAO2 was dominantly expressed in leaves, and its transcript level was greatly increased under exogenous ABA application; overexpression of AhAO2 in Arabidopsis led to improved ABA levels and drought tolerance after drought treatment (Yang et al., 2011). ZmAO-2 in maize was expressed at a higher level in coleoptiles than in roots (Sekimoto et al., 1997).
From the prediction of the website, AtAO2 in Arabidopsis (AT3G43600) could interact with two proteins (gene number: AT1G19730 and AT5G42980) which are belong to thioredoxin superfamily and it indicates AO2 might involve in redox reaction.
Biochemical and physiological function of AO3
Among the AOs, AO3 has gained great attention owing to its possible involvement in ABA biosynthesis and its importance under normal and stress conditions (Seo et al., 2000a, 2000b; González-Guzmán et al., 2004). The AO3 gene can efficiently catalyse the final step in ABA synthesis in Arabidopsis leaves (Seo et al., 2000b) and seeds (González-Guzmán et al., 2004), in barley roots (Omarov et al., 2003), and in pea plant leaves and roots (Zdunek-Zastocka et al., 2004).
AO3 is mainly localized in vegetative organs such as the vascular tissues of roots, hypocotyls as well as leaves and is also slightly expressed within guard cells and surrounding cells (Koiwai et al., 2004; Nambara & Marion-Poll, 2005). Rice OsAO3 was expressed in germinated seeds, roots, leaves, and floral organs, particularly in vascular tissues and guard cells, and its expression was significantly induced by exogenous ABA and mannitol treatment. The osao3 mutant accumulated less ABA and exhibited earlier seed germination, increased seedling growth and grain yield, and decreased drought tolerance than the wild-type, and OsAO3-overexpressing lines exhibited the opposite phenotype. Mutation and overexpression of OsAO3 increased and decreased grain yield, respectively, by affecting panicle number per plant, spikelet number per panicle, and spikelet fertility (Shi et al., 2021).
In Arabidopsis, AO3 mRNA is mainly expressed and most AOd enzyme activity is detected in rosette leaves. The ao3-1 mutant exhibited a wilted phenotype under normal conditions, reduced ABA levels accompanied by excessive water loss and retarded vegetative growth and reduced stress tolerance, but this mutant showed a less prominent dormancy-associated phenotype in seeds compared with aba3-2, with impaired function of all AOs due to the dysfunction of Moco (Seo et al., 2000b; González-Guzmán et al., 2004). These findings indicated that AOd is involved in ABA biosynthesis in leaves and that other aldehyde oxidases are involved in ABA biosynthesis in other organs, such as roots, silique, and seeds (Seo et al., 2004). Consistently, mutations in AO1 or AO4 in the AO3 mutant background aggravated ABA deficiency in seeds, demonstrating that both gene products contribute partially to ABA biosynthesis and act as a ‘back-up system’ to AO3 in the AO3 mutant background, while this effect might be negligible in wild-type Arabidopsis. Absence of AAO3 oxidation activity (rather than the lower ABA) and its associated function is responsible for the earlier senescence symptoms in aao3 mutant. Arabidopsis AAO3 knockout mutant aao3 exhibited earlier senescence compared with wild-type during normal growth or upon application of UV-C irradiation. Different aldehydes accumulated prominently in aao3 mutants compared with WT leaves under normal growth conditions, upon UV-C irradiation and after exogenous aldehydes application (Nurbekova et al., 2021).
AO3 can be slightly induced by stresses; for example, an increase in abscisic aldehyde oxidizing activity was observed for the PsAO isoform in the leaves and roots of pea plants exposed to salinity or ammonium treatments (Zdunek-Zastocka et al., 2004). Drought stress increases AO3 transcript levels and the activity of AOd (Seo et al., 2000b; Bittner, Oreb & Mendel, 2001) and high AO3 expression was sufficient to induce stomatal closure (Melhorn et al., 2008).
Biochemical and physiological function of AO4
A study on Arabidopsis AO4 knockout mutants and overexpressing lines demonstrated that AO4 plays a critical role in delaying senescence and protecting silique against toxic aldehydes (Ibdah et al., 2009). Single loss-of-function mutants for AO4 failed to show significant changes in endogenous ABA levels in seeds compared with wild type. Plants produce toxic aldehydes under normal and stress conditions (Mano, 2012; Srivastava et al., 2017) and excess aldehydes may lead to reactive oxygen species (ROS) accumulation that could damage proteins, lipids and other molecular compounds (Bartels, 2001; Kotchoni et al., 2006; Zhang et al., 2012; Biswas & Mano, 2015). AO4 was expressed most abundantly in developing silique, and AO4 was involved in the oxidation of benzaldehyde in silique, contributing to the synthesis of the benzoic acid (BA) pool in seed capsules (Ibdah et al., 2009). The AO4 mutant seeds showed significant reductions in the total contents of benzaldehyde and 4-benzoyloxybutylglucosinolate, as well as a slight reductions in the levels of 3-benzoyloxypropylglucosinolate (Ibdah et al., 2009).
AO4 can oxidize various aromatic and aliphatic aldehydes and it differentially generates superoxide (O2−) and hydrogen peroxide (H2O2) in an aldehyde-dependent manner. In addition, AO4 transcript levels and activity in silique were induced by H2O2 application, indicating that the ROS generated by the activity of AO4 can self-amplify, enabling further detoxification of toxic aldehydes and delaying silique senescence of Arabidopsis plants (Srivastava et al., 2017). Functions of aldehyde oxidases in plants were summarized in Table 1.
Table 1. Aldehyde oxidase in plants.
| Species | Substrate/localization | Protein | Function | Reference |
|---|---|---|---|---|
| Cucumber | Benzaldehyde, Phenylacetaldehyde Indole-3-acetaldehyde (major) |
AO1 | IAA biosynthesis | Bower, Brown & Purves (1978) |
| Pea | Indole-3-acetaldehyde | AO1 | IAA biosyntesis | Miyata et al. (1981) |
| Pea | Indole-3-aldehyde | AO3 | AO activity and ABA induced by suboptimal conditions | Zdunek & Lips (2001) |
| Maize | Indole-3-acetaldehyde/apical region of coleoptiles | AO1 | IAA production | Koshiba et al. (1996) |
| Barley | indole-3-aldehyde, acetaldehyde, heptaldehyde, benzaldehyde | AOs | Omarov et al. (1999) | |
| Oat | indoleacetaldehyde | IAA biosynthesis | Rajagopal (1971) | |
| Arabidopsis | Indole-3-aldehyde | AO1 | ABA biosyntesis | Seo et al. (1998), Böttcher et al. (2014) |
| Arabidopsis | Abscisic aldehyde/rosette leaves | AO3 | ABA production | González-Guzmán et al. (2004) |
| Arabidopsis | Abscisic aldehyde | AO3 | ABA and ROS production, drought and water stress | Yesbergenova et al. (2005) |
| Arabidopsis | Aromatic and aliphatic aldehydes/silique | AO4 | Aldehyde detoxification | Srivastava et al. (2017) |
| Arabidopsis | Abscisic aldehyde, hexanal, and acetaldehyde | AO3 | ABA production, Aldehyde detoxification | Nurbekova et al. (2021) |
| Arabidopsis | Abscisic aldehyde/leaves | AO3 | ABA biosyntesis | Seo et al. (2000a) |
| Arabidopsis | Abscisic aldehyde/seeds | AO3 | ABA biosyntesis | Seo et al. (2004) |
| Arabidopsis | Benzaldehyde/sillique | AO4 | Benzoic acid | Ibdah et al. (2009) |
| wheat | Abscisic aldehyde | AO3 | carotenoid pigments accumulation | Colasuonno et al. (2017) |
| Rice | Abscisic aldehyde/germinated seeds, roots, leaves, floral organs, vascular tissues, guard cells | AO3 | ABA production, drought stress | Shi et al. (2021) |
Summary and prospect
Phylogenic tree showed at least two conserved clades for AO enzymes involved in the biosynthesis of ABA and auxin synthesis (Abu-Zaitoon, 2014) and it supports the proposed function of ABA and auxin biosynthesis. Studies about AOs are quite scant; however, AOs are very import enzymes merit better understandings due to two reasons. Firstly, they may involve in biosynthesis of important phytohormones, and it may be a potential target for genetic control in agriculture as genes like GA20-oxidase in green revolution. Phytohormones are vital substances regulates multifaced function of plants and mutants in the early steps of biosynthetic pathway could be lethal and useful tools in scientific researches while mutation in late nodes could be important regulating points and easier for genetic manipulation in agriculture. Secondly, AOs could have multifaced functions due to their wide substrate of AOs, thus pathway or traits influenced by AOs also deserve further studies. In this review, we summarized the recent studies about AO in plants including the motif information, biochemical, and physiological functions which may provide useful information for botanist and biologist and it is also beneficial for agriculture.
AO may be involved in metabolic processes not only phytohormone synthesis but also cellular ROS homeostasis under normal and stress conditions (Wendehenne et al., 2014; Gilroy et al., 2014). Recombinantly expressed Arabidopsis AO1 and AO3 in the methylotrophic yeast Pichia pastoris are capable of producing not only H2O2 but also O2− during oxidation of aldehydes (Zarepour et al., 2012). In addition to their aldehyde oxidation activity, AO1 and AO3 were found to exhibit NADH oxidase activity (Yergaliyeva et al., 2016; Brikis et al., 2018; Zhong et al., 2010; Hesberg et al., 2004; Diallinas et al., 1997; Yesbergenova et al., 2005). In this respect, conditions such as natural senescence and stress-induced stomatal movement, which both require simultaneously elevated levels of abscisic acid and hydrogen peroxide/superoxide, are likely to benefit from AOs by formation of abscisic acid and ROS.
As mentioned above, Moco is an important component of several enzymes including nitrate reductase, AOs, XDH. The activity of AO enzymes requires a sulfurated Moco, which is converted from the desulfo- to the sulfo-form by the Moco sulfurase ABA3 (Xiong et al., 2001; Bittner, Oreb & Mendel, 2001). Deficiency in the biosynthesis of Moco might result in pleiotropic effect or even lethal consequences for the respective organisms (Mendel, 2013). For example, Moco deficient mutants of barley Az34 (Walker, Kudrna & Warner, 1989), tobacco (Leydecker et al., 1995), tomato flacca and sitiens1 (Sagi, Fluhr & Lips, 1999; Sagi, Scazzocchio & Fluhr, 2002), and Arabidopsis aba3 (Akaba et al., 1999; Bittner, Oreb & Mendel, 2001) had severely impaired ABA production.
The availability of Moco and its sulfuration may therefore represent a potential point of interaction between cytokinin, ABA and IAA metabolism in plants to control phytohormone balance, which may determine the stress tolerance of plants (Cowan & Tayor, 2001). In addition, the plant Moco-pool size (enzymes with Moco) varies under different nutritional and environmental conditions (Sagi et al., 1997; Sagi, Omarov & Lips, 1998). Thus, increased activity of Mo-hydroxylases (i.e., AO and XDH) in response to salt stress and ammonium treatment could be one mechanism for stress adaptation, which could include enhanced activity of the Moco-containing enzymes responsible for ABA synthesis (Sagi, Omarov & Lips, 1998). When XDH activity or nitrate assimilation is reduced or inhibited, more Moco might therefore be available for sulfuration (Sagi, Fluhr & Lips, 1999; Sagi, Scazzocchio & Fluhr, 2002; Bittner, Oreb & Mendel, 2001) and thus AOs enzymes were more activated. One the other hand, when nitrate reductase has been induced by its substrate, the AO activity and ABA content in plants were reduced (Omarov, Sagi & Lips, 1998). Molybdenum as one of the trace essential elements for plants, and the effect of external sources molybdenum on AO, XDH, and other enzymes as well as phytohormone biosynthesis could be a promising research direction.
In addition, AO1 was predict to involve in hypoxia stress of Arabidopsis and AO2 was assumed to interact with proteins belong to thioredoxin superfamily. We can speculate AOs might regulate the abiotic stress of plants including drought, and osmotic, stress. The study about AO enzymes thrived around 20 years ago, and they need further study for possible manipulation in agriculture for suitable phytohormone biosynthesis, ROS homeostasis, stress tolerance, development, and aldehyde detoxification.
Acknowledgments
We thank Dr. Qisen Zhang for useful discussion.
Funding Statement
The work was supported by the funding from the National Natural Science Foundation of China (31901438) and crop varietal improvement and insect pests control by nuclear radiation program. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Additional Information and Declarations
Competing Interests
The authors declare that they have no competing interests.
Author Contributions
Jun Wu performed the experiments, authored or reviewed drafts of the paper, and approved the final draft.
Blair Moses Kamanga analyzed the data, authored or reviewed drafts of the paper, and approved the final draft.
Wenying Zhang analyzed the data, authored or reviewed drafts of the paper, and approved the final draft.
Yanhao Xu conceived and designed the experiments, prepared figures and/or tables, authored or reviewed drafts of the paper, and approved the final draft.
Le Xu conceived and designed the experiments, performed the experiments, prepared figures and/or tables, authored or reviewed drafts of the paper, and approved the final draft.
Data Availability
The following information was supplied regarding data availability:
There is no data to publish; this article is a literature review.
References
- Abu-Zaitoon (2014).Abu-Zaitoon YM. Phylogenetic analysis of putative genes involved in the tryptophan-dependent pathway of auxin biosynthesis in rice. Applied Biochemistry and Biotechnology. 2014;172(5):2480–2495. doi: 10.1007/s12010-013-0710-4. [DOI] [PubMed] [Google Scholar]
- Akaba et al. (1999).Akaba S, Seo M, Dohmae N, Takio K, Sekimoto H, Kamiya Y, Furuya N, Romano T, Koshiba T. Production of homo- and hetero-dimeric isozymes from two aldehyde oxidase genes of Arabidopsis thaliana. The Journal of Biochemistry. 1999;126(2):395–401. doi: 10.1093/oxfordjournals.jbchem.a022463. [DOI] [PubMed] [Google Scholar]
- Ando et al. (2006).Ando S, Tsushima S, Tagili A, Kamachi S, Konagaya K, Hagio T, Tabei Y. Increase in BrAO1 gene expression and aldehyde oxidase activity during clubroot development in Chinese cabbage (Brassica rapa L.) Molecular Plant Biology. 2006;7(4):223–234. doi: 10.1111/j.1364-3703.2006.00333.x. [DOI] [PubMed] [Google Scholar]
- Audran et al. (1998).Audran C, Borel C, Frey A, Sotta B, Meyer C, Simonneau T, Marion-Poll A. Expression studies of the zeaxanthin epoxidase gene in Nicotiana plumbaginifolia. Plant Physiology. 1998;118(3):1021–1028. doi: 10.1104/pp.118.3.1021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Audran et al. (2001).Audran C, Liotenberg S, Gonneau M, North H, Frey A, Tap-Waksman K, Vartanian N, Marion-Poll A. Localisation and expression of zeaxanthin epoxidase mRNA in Arabidopsis in response to drought stress and during seed development. Australian Journal of Plant Physiology. 2001;28(12):1161–1173. doi: 10.1071/pp00134. [DOI] [Google Scholar]
- Barrero et al. (2006).Barrero JM, Rodríguez PL, Quesada V, Piqueras P, Ponse MR, Micol JL. Both abscisic acid (ABA)-dependent and ABA-independent pathways govern the induction of NCED3, AAO3 and ABA1 in response to salt stress. Plant Cell and Environment. 2006;29(10):2000–2008. doi: 10.1111/j.1365-3040.2006.01576.x. [DOI] [PubMed] [Google Scholar]
- Bartels (2001).Bartels D. Targeting detoxification pathways: an efficient approach to obtain plants with multiple stress tolerance? Trends Plant Sciences. 2001;6(7):284–286. doi: 10.1016/S1360-1385(01)01983-5. [DOI] [PubMed] [Google Scholar]
- Biswas & Mano (2015).Biswas MS, Mano JI. Lipid peroxide-derived short-chain carbonyls mediate hydrogen peroxide-induced and salt-induced programmed cell death in plants. Plant Physiology. 2015;168(3):885–898. doi: 10.1104/pp.115.256834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bittner, Oreb & Mendel (2001).Bittner F, Oreb M, Mendel RR. ABA3 Is a molybdenum cofactor sulfurase required for activation of aldehyde oxidase and xanthine dehydrogenase in Arabidopsis thaliana. The Journal of Biological Chemistry. 2001;276(44):40381–40384. doi: 10.1074/jbc.C100472200. [DOI] [PubMed] [Google Scholar]
- Böttcher et al. (2014).Böttcher C, Chapman A, Fellermeier F, Choudhary M, Scheel D, Glawischnig E. The biosynthetic pathway of indole-3-carbaldehyde and indole-3-carboxylic acid derivatives in arabidopsis. Plant Physiology. 2014;165(2):841–853. doi: 10.1104/pp.114.235630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bower, Brown & Purves (1978).Bower PJ, Brown HM, Purves WK. Cucumber seedling indoleacetaldehyde oxidase. Plant Physiology. 1978;61(1):107–110. doi: 10.1104/pp.61.1.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brikis et al. (2018).Brikis CJ, Zarei A, Chiu GZ, Deyman KL, Liu JL, Trobacher CP, Hoover GJ, Subedi S, DeEll JR, Bozzo GG, Shelp BJ. Targeted quantitative profiling of metabolites and gene transcripts associated with 4-aminobutyrate (GABA) in apple fruit stored under multiple abiotic stresses. Horticulture Research. 2018;61(5):1–18. doi: 10.1038/s41438-018-0069-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng et al. (2002).Cheng WH, Endo A, Zhou L, Penney J, Chen HC, Arroyo A, Leon P, Nambara E, Asami T, Seo M, Koshiba T, Sheen J. A unique short-chain dehydrogenase/reductase in Arabidopsis glucose signaling and abscisic acid biosynthesis and functions. The Plant Cell. 2002;14(11):2723–2743. doi: 10.1105/tpc.006494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colasuonno et al. (2017).Colasuonno P, Marcotuli I, Lozito ML, Simeone R, Blanco A, Gadaleta A. Characterization of aldehyde oxidase (ao) genes involved in the accumulation of carotenoid pigments in wheat grain. Frontiers in Plant Science. 2017;8:863. doi: 10.3389/fpls.2017.00863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cowan & Tayor (2001).Cowan K, Tayor N. Plant hormone homeostasis and the control of avocado fruit size. Plant Growth Regulation. 2001;35(3):247–255. doi: 10.1023/A:1014463300850. [DOI] [Google Scholar]
- Dejonghe, Okamoto & Cutler (2018).Dejonghe W, Okamoto M, Cutler SR. Small molecule probes of ABA biosynthesis and signaling. Plant Cell and Physiology. 2018;59(8):1490–1499. doi: 10.1093/pcp/pcy126. [DOI] [PubMed] [Google Scholar]
- Diallinas et al. (1997).Diallinas G, Pateraki I, Sanmartin M, Scossa A, Stilianou E, Panopoulos NJ, Kanellis AK. Melon ascorbate oxidase: cloning of a multigene family, induction during fruit development and repression by wounding. Plant Molecular Biology. 1997;34(5):759–770. doi: 10.1023/A:1005851527227. [DOI] [PubMed] [Google Scholar]
- Estrada-Melo et al. (2015).Estrada-Melo AC, Chao, Reid MS, Jiang C-Z. Overexpression of an ABA biosynthesis gene using a stress-inducible promoter enhances drought resistance in petunia. Horticulture Research. 2015;2015(2):15013. doi: 10.1038/hortres.2015.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fedorova et al. (2005).Fedorova E, Redondo FJ, Koshiba T, Pueyo JJ, Rosario de Felipe M, Lucas MM. Aldehyde oxidase (AO) in the root nodules of Lupinus albus and Medicago truncatula: identification of AO in meristematic and infection zones. The American Phytopathological Society. 2005;18(5):405–413. doi: 10.1094/MPMI-18-0405. [DOI] [PubMed] [Google Scholar]
- Finkelstein (2013).Finkelstein R. Abscisic acid synthesis and response. Arabidopsis Book. 2013;11:e0166. doi: 10.1199/tab.0166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garattini, Fratelli & Terao (2008).Garattini E, Fratelli M, Terao M. Mammalian aldehyde oxidases: genetics, evolution and biochemistry. Cellular and Molecular Life Sciences. 2008;65(7–8):1019–1048. doi: 10.1007/s00018-007-7398-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilroy et al. (2014).Gilroy S, Suzuki N, Miller G, Choi WG, Toyota M, Devireddy AR, Mittler R. A tidal wave of signals: calcium and ROS at the forefront of rapid systemic signaling. Trends in Plant Science. 2014;19(10):623–630. doi: 10.1016/j.tplants.2014.06.013. [DOI] [PubMed] [Google Scholar]
- González-Guzmán et al. (2004).González-Guzmán M, Abia D, Salinas J, Serrano R, Rodríguez PL. Two new alleles of the abscisic aldehyde oxidase 3 gene reveal its role in abscisic acid biosynthesis in seeds. Plant Physiology. 2004;135(1):325–333. doi: 10.1104/pp.103.036590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- González-Guzmán et al. (2002).González-Guzmán M, Apostolova N, Bellés JM, Barrero JM, Piqueras P, Ponce MR, Micol JL, Serrano R, Rodríguez PL. The short-chain alcohol dehydrogenase aba2 catalyzes the conversion of xanthoxin to abscisic aldehyde. The Plant Cell. 2002;14(8):1833–1846. doi: 10.1105/tpc.002477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hesberg et al. (2004).Hesberg C, Hänsch R, Mendel RR, Bittner F. Tandem orientation of duplicated xanthine dehydrogenase genes from Arabidopsis thaliana differential gene expression and enzyme activities. Journal of Biological Chemistry. 2004;279(14):13547–13554. doi: 10.1074/jbc.M312929200. [DOI] [PubMed] [Google Scholar]
- Hille, Nishinoab & Bittner (2011).Hille R, Nishinoab T, Bittner F. Molybdenum enzymes in higher organisms. Coordination Chemistry Reviews. 2011;255(9–10):1179–1205. doi: 10.1016/j.ccr.2010.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ibdah et al. (2009).Ibdah M, Chen YT, Wilkerson CG, Pichersky E. An aldehyde oxidase in developing seeds of Arabidopsis converts benzaldehyde to benzoic acid. Plant Physiology. 2009;150(1):416–423. doi: 10.1104/pp.109.135848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasahara (2016).Kasahara H. Current aspects of auxin biosynthesis in plants. Bioscience Biotechnology and Biochemistry. 2016;80(1):34–42. doi: 10.1080/09168451.2015.1086259. [DOI] [PubMed] [Google Scholar]
- Kilian et al. (2007).Kilian J, Whitehead D, Horak J, Wanke D, Weinl S, Batistic O, D’Angelo C, Bornberg-Bauer E, Kudla J, Harter K. The AtGenExpress global stress expression data set: protocols, evaluation and model data analysis of UV-B light, drought and cold stress responses. Plant Journal. 2007;50(2):347–363. doi: 10.1111/j.1365-313X.2007.03052.x. [DOI] [PubMed] [Google Scholar]
- Koiwai et al. (2000).Koiwai H, Akaba S, Seo M, Komano T, Koshiba T. Functional expression of two Arabidopsis aldehyde oxidases in the yeast Pichia pastoris. The Journal of Biochemistry. 2000;127(4):659–664. doi: 10.1093/oxfordjournals.jbchem.a022654. [DOI] [PubMed] [Google Scholar]
- Koiwai et al. (2004).Koiwai H, Nakaminami K, Seo M, Mitsuhashi W, Koshiba TT. Tissue-specific localization of an abscisic acid biosynthetic enzyme, aao3, in Arabidopsis. Plant Physiology. 2004;134(4):1697–1707. doi: 10.2307/4281702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koshiba et al. (1996).Koshiba T, Saito E, Ono N, Yamamoto N, Sato M. Purification and properties of flavin—and molybdenum- containing aldehyde oxidase from coleoptiles of maize. Plant Physiology. 1996;110(3):781–789. doi: 10.1104/pp.110.3.781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kotchoni et al. (2006).Kotchoni SO, Kuhns C, Ditzer A, Kirch HH, Bartels D. Over-expression of different aldehyde dehydrogenase genes in Arabidopsis thaliana confers tolerance to abiotic stress and protects plants against lipid peroxidation and oxidative stress. Plant Cell Environment. 2006;29(6):1033–1048. doi: 10.1111/j.1365-3040.2005.01458.x. [DOI] [PubMed] [Google Scholar]
- Leydecker et al. (1995).Leydecker MT, Moureaux T, Kraepiel Y, Schnorr K, Caboche M. Molybdenum cofactor mutants, specifically impaired in xanthine dehydrogenase activity and abscisic acid biosynthesis, simultaneously overexpress nitrate reductase. Plant Physiology. 1995;107(4):1427–1431. doi: 10.1104/pp.107.4.1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mano (2012).Mano J. Reactive carbonyl species: their production from lipid peroxides, action in environmental stress, and the detoxification mechanism. Plant Physiology Biochemistry. 2012;59(1):90–97. doi: 10.1016/j.plaphy.2012.03.010. [DOI] [PubMed] [Google Scholar]
- Mano & Nemoto (2012).Mano Y, Nemoto K. The pathway of auxin biosynthesis in plants. Journal of Experimental Botany. 2012;63(8):2853–2872. doi: 10.1093/jxb/ers091. [DOI] [PubMed] [Google Scholar]
- Marin et al. (1996).Marin E, Nussaume L, Quesada A, Gonneau M, Sotta B, Hugueney P, Frey A, Marion-Poll A. Molecular identification of zeaxanthin epoxidase of Nicotiana plumbaginifolia, a gene involved in abscisic acid biosynthesis and corresponding to the ABA locus of Arabidopsis thaliana. EMBO Journal. 1996;15(10):2331–2342. doi: 10.1002/j.1460-2075.1996.tb00589.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melhorn et al. (2008).Melhorn V, Matsumi K, Koiwai H, Ikegami K, Okamoto M, Nambara E, Bittner F, Koshiba T. Transient expression of AtNCED3 and AAO3 genes in guard cells causes stomatal closure in Vicia faba. Journal of Plant Research. 2008;121(1):125–131. doi: 10.1007/s10265-007-0127-7. [DOI] [PubMed] [Google Scholar]
- Mendel (2013).Mendel RR. The molybdenum cofactor. The Journal of Biological Chemistry. 2013;288(19):13165–13172. doi: 10.1074/jbc.R113.455311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merlot & Giraudat (1997).Merlot S, Giraudat J. Genetic analysis of abscisic acid signal transduction. Plant Physiology. 1997;114(3):751–757. doi: 10.1104/pp.114.3.751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milborrow (2001).Milborrow BV. The pathway of biosynthesis of abscisic acid in vascular plants: a review of the present state of knowledge of ABA biosynthesis. Journal of Experimental Botany. 2001;52(359):1143–1164. doi: 10.1093/jexbot/52.359.1145. [DOI] [PubMed] [Google Scholar]
- Miyata et al. (1981).Miyata S, Suzuki Y, Kamisaka S, Masuda Y. Indole-3-acetaldehyde oxidase of pea seedlings. Physiologia Plantarum. 1981;51(4):402–406. doi: 10.1111/j.1399-3054.1981.tb05577.x. [DOI] [Google Scholar]
- Nambara & Marion-Poll (2005).Nambara E, Marion-Poll A. Abscisic acid biosynthesis and catabolism. Annual Review of Plant Biology. 2005;56(1):165–185. doi: 10.1146/annurev.arplant.56.032604.144046. [DOI] [PubMed] [Google Scholar]
- Nishino & Nishino (1989).Nishino T, Nishino T. The nicotinamide adenine dinucleotide-binding site of chicken liver xanthine dehydrogenase: evidence for alteration of the redox potential of the flavin by NAD binding or modification of the NAD-binding site and isolation of a modified peptide. Journal of Biology Chemistry. 1989;264(10):5468–5473. doi: 10.1016/S0021-9258(18)83568-2. [DOI] [PubMed] [Google Scholar]
- Normanly, Slovin & Cohen (1995).Normanly J, Slovin JP, Cohen JD. Rethinking auxin biosynthesis and metabolism. Plant Physiology. 1995;107(2):323–329. doi: 10.1104/pp.107.2.323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nurbekova et al. (2021).Nurbekova Z, Srivastava S, Standing D, Kurmanbayeva A, Bekturova A, Soltabayeva A, Oshanova D, Tureckova V, Strand M, Biswas MS, Mano J, Sagi M. Arabidopsis aldehyde oxidase 3, known to oxidize abscisic aldehyde to abscisic acid, protects leaves from aldehyde toxicity. The Plant Journal. 2021;108(5):1439–1455. doi: 10.1111/tpj.15521. [DOI] [PubMed] [Google Scholar]
- Omarov et al. (2003).Omarov R, Dräger D, Tischner R, Lips H. Aldehyde oxidase isoforms and subunit composition in roots of barley as affected by ammonium and nitrate. Physiologia Plantarum. 2003;117(3):337–342. doi: 10.1034/j.1399-3054.2003.00043.x. [DOI] [PubMed] [Google Scholar]
- Omarov, Sagi & Lips (1998).Omarov RT, Sagi M, Lips SH. Regulation of aldehyde oxidase and nitrate reductase in roots of barley (Hordeum vulgare L.) by nitrogen source and salinity. Journal of Experimental Botany. 1998;49(322):897–902. doi: 10.1093/jxb/49.322.897. [DOI] [Google Scholar]
- Omarov et al. (1999).Omarov RT, Shuichi A, Tomokazu K, Herman LS. Aldehyde oxidase in roots, leaves and seeds of barley (Hordeum vulgare L.) The Journal of Experimental Botany. 1999;50(330):63–69. doi: 10.1093/jexbot/50.330.63. [DOI] [Google Scholar]
- Ori et al. (1997).Ori N, Eshed Y, Paran I, Zamir D, Fluhr R. TAO1, a representative of the molybdenum cofactor containing hydroxylases from tomato. Journal of Biological Chemistry. 1997;272(2):1019–1025. doi: 10.1074/jbc.272.2.1019. [DOI] [PubMed] [Google Scholar]
- Qin & Zeevaart (1999).Qin X, Zeevaart JAD. The 9-cis-epoxycarotenoid cleavage reaction is the key regulatory step of abscisic acid biosynthesis in water-stressed bean. Proceedings of the National Academy of Sciences of the United States of America. 1999;96(26):15354–15361. doi: 10.1073/pnas.96.26.15354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajagopal (1971).Rajagopal R. Metabolism of Indole-3-acetaldehyde. III. Some characteristics of the aldehyde oxidase of avena coleoptiles. Physiologia Plantarum. 1971;24(2):272–281. doi: 10.1111/j.1399-3054.1971.tb03491.x. [DOI] [Google Scholar]
- Rothe (1974).Rothe GM. Aldehyde oxidase isoenzymes (E.C. 1.2.3.1) in potato tubers (Solanum tuberosum) Plant and Cell Physiology. 1974;15(3):493–499. doi: 10.1103/PhysRevD.7.3821.3. [DOI] [Google Scholar]
- Sagi et al. (1997).Sagi M, Dovrat A, Kipnis T, Lips H. Ionic balance, biomass production, and organic nitrogen as affected by salinity and nitrogen source in annual ryegrass. Journal of Plant Nutrition. 1997;20(10):1291–1316. doi: 10.1080/01904169709365336. [DOI] [Google Scholar]
- Sagi, Fluhr & Lips (1999).Sagi M, Fluhr R, Lips SH. Aldehyde oxidase and xanthine dehydrogenase in a flacca tomato mutant with deficient abscisic acid and wilty phenotype. Plant Physiology. 1999;121(2):315. doi: 10.1104/pp.120.2.571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sagi, Omarov & Lips (1998).Sagi M, Omarov RT, Lips SH. The Mo-hydroxylases xanthine dehydrogenase and aldehyde oxidase in ryegrass as affected by nitrogen and salinity. Plant Science. 1998;135(2):125–135. doi: 10.1016/S0168-9452(98)00075-2. [DOI] [Google Scholar]
- Sagi, Scazzocchio & Fluhr (2002).Sagi M, Scazzocchio C, Fluhr R. The absence of molybdenum cofactor sulfuration is the primary cause of the flacca phenotype in tomato plants. The Plant Journal. 2002;31(3):305–317. doi: 10.1046/j.1365-313x.2002.01363.x. [DOI] [PubMed] [Google Scholar]
- Schwartz et al. (1997).Schwartz SH, Tan BC, Gage DA, Zeevaart JAD, Mccarty DA. Specific oxidative cleavage of carotenoids by VP14 of maize. Science. 1997;276(5320):1872–1874. doi: 10.1126/science.276.5320.1872. [DOI] [PubMed] [Google Scholar]
- Sekimoto et al. (1997).Sekimoto H, Seo M, Dohmae N, Takio K, Kamiya Y, Koshiba T. Cloning and molecular characterization of plant aldehyde oxidase. Journal of Biological Chemistry. 1997;272(24):15280–15285. doi: 10.1074/jbc.272.24.15280. [DOI] [PubMed] [Google Scholar]
- Seo et al. (1998).Seo M, Akaba S, Oritani T, Delarue M, Bellini C, Caboche M, Koshiba T. Higher activity of an aldehyde oxidase in the auxin-overproducing superroot1 mutant of Arabidopsis thaliana. Plant Physiology. 1998;116(2):687–693. doi: 10.1104/pp.116.2.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seo et al. (2004).Seo M, Aoki H, Koiwai H, Kamiya Y, Nambara E, Koshiba T. Comparative studies on the Arabidopsis aldehyde oxidase (AAO) gene family revealed a major role of AAO3 in ABA biosynthesis in seeds. Plant Cell Physiology. 2004;45(11):1694–1703. doi: 10.1093/pcp/pch198. [DOI] [PubMed] [Google Scholar]
- Seo et al. (2000a).Seo M, Koiwai H, Akaba S, Komano T, Oritani T, Kamiya Y, Koshiba T. Abscisic aldehyde oxidase in leaves of Arabidopsis thaliana. The Plant Journal. 2000a;23(4):481–488. doi: 10.1046/j.1365-313x.2000.00812.x. [DOI] [PubMed] [Google Scholar]
- Seo & Koshiba (2002).Seo M, Koshiba T. Complex regulation of ABA biosynthesis in plants. Trends in Plant Science. 2002;7(1):41–48. doi: 10.1016/s1360-1385(01)02187-2. [DOI] [PubMed] [Google Scholar]
- Seo et al. (2000b).Seo M, Peeters AJM, Koiwai H, Oritani T, Marion-Poll A, Zeevaart JAD, Koornneef M, Kamiya Y, Koshiba T, Koshiba T. The Arabidopsis aldehyde oxidase 3 (AAO3) gene product catalyzes the final step in abscisic acid biosynthesis in leaves. Proceedings of the National Academy of Sciences of the United States of America. 2000b;97(23):12908–12913. doi: 10.1073/pnas.220426197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi et al. (2021).Shi X, Tian Q, Deng P, Zhang W, Jing W. The rice aldehyde oxidase osao3 gene regulates plant growth, grain yield, and drought tolerance by participating in aba biosynthesis. Biochemical and Biophysical Research Communications. 2021;548(9):189–195. doi: 10.1016/j.bbrc.2021.02.047. [DOI] [PubMed] [Google Scholar]
- Song et al. (2020a).Song SQ, Liu J, Tang CF, Zhang WH, Xu HH, Zhang Q, Gao JD. Metabolism and signaling of auxins and their roles in regulating seed dormancy and germination (in Chinese) Chinese Science Bulletin. 2020a;65(34):3924–3943. doi: 10.1360/TB-2020-0509. [DOI] [Google Scholar]
- Song et al. (2020b).Song SQ, Liu J, Xu HH, Xu L, Hui H. ABA metabolism and signaling and their molecular mechanism regulating seed dormancy and germination. Scientia Agricultura Sinica. 2020b;53(5):857–873. doi: 10.3864/j.issn.0578-1752.2020.05.001. [DOI] [Google Scholar]
- Srivastava et al. (2017).Srivastava S, Brychkova G, Yarmolinsky D, Soltabayeva A, Samani T, Sagi M. Aldehyde oxidase 4 plays a critical role in delaying silique senescence by catalyzing aldehyde detoxification. Plant Physiology. 2017;173(4):1977–1997. doi: 10.1104/pp.16.01939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor, Burbidge & Thompson (2000).Taylor IB, Burbidge A, Thompson AJ. Control of abscisic acid synthesis. Journal of Experimental Botany. 2000;51(350):1563–1574. doi: 10.1093/jexbot/51.350.1563. [DOI] [PubMed] [Google Scholar]
- Turner, Weiner & Tayior (1995).Turner RJ, Weiner JH, Tayior DE. The tellurite-resistance determinants tehAtehB and klaAklaBtelB have different biochemical requirements. Microbiology. 1995;141(Pt. 12):3133–3140. doi: 10.1099/13500872-141-12-3133. [DOI] [PubMed] [Google Scholar]
- Verslues & Zhu (2005).Verslues PE, Zhu JK. Before and beyond ABA: upstream sensing and internal signals that determine ABA accumulation and response under abiotic stress. Biochemical Society Transactions. 2005;33(Pt. 2):375–379. doi: 10.1042/BST0330375. [DOI] [PubMed] [Google Scholar]
- Waese et al. (2017).Waese J, Fan J, Pasha A, Yu H, Geoffrey Fucile G, Shi R, Cumming M, Kelley LA, Sternberg MJ, Krishnakumar V, Ferlanti E, Miller J, Town C, Stuerzlinger W, Provart NJ. ePlant: visualizing and exploring multiple levels of data for hypothesis generation in plant biology. The Plant Cell. 2017;29(8):1806–1821. doi: 10.1105/tpc.17.00073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker, Kudrna & Warner (1989).Walker SM, Kudrna DA, Warner RL. Reduced accumulation of ABA during water stress in a molybdenum cofactor mutant of barley. Plant Physiology. 1989;90(2):728–733. doi: 10.1104/pp.90.2.728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wendehenne et al. (2014).Wendehenne D, Gao QM, Kachroo A, Kachroo P. Free radical-mediated systemic immunity in plants Author links open overlay panel. Current Opinion in Plant Biology. 2014;20:127–134. doi: 10.1016/j.pbi.2014.05.012. [DOI] [PubMed] [Google Scholar]
- Xiong et al. (2001).Xiong L, Ishitani M, Lee H, Zhu JK. The Arabidopsis LOS5/ABA3 locus encodes a molybdenum cofactor sulfurase and modulates cold stress- and osmotic stress-responsive gene expression. The Plant Cell. 2001;13(9):2063–2083. doi: 10.1105/tpc.13.9.2063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong, Schumaker & Jian-Kang (2002).Xiong L, Schumaker KS, Jian-Kang Z. Cell signaling during cold, drought, and salt stress. The Plant Cell. 2002;14(Suppl. 1):S165–S183. doi: 10.1105/tpc.000596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu, Pan & Zhang (2020).Xu L, Pan R, Zhang W. Membrane lipids are involved in plant response to oxygen deprivation. Plant Signaling & Behavior. 2020;15:7. doi: 10.1080/15592324.2020.1771938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang et al. (2011).Yang L, Liang J, Zhou W, Su L, Zhang B, Li L. Isolation and characterization of the aldehyde oxidase2 gene from Arachis hypogaea L. Plant Molecular Biology. 2011;29(3):544–553. doi: 10.1007/s11105-010-0259-0. [DOI] [Google Scholar]
- Yergaliyeva et al. (2016).Yergaliyeva TM, Nurbekova Z, Mukiyanova G, Akbassova A, Omarov RT. The involvement of ROS producing aldehyde oxidase in plant response to Tombusvirus infection. Plant Physiology and Biochemistry. 2016;109:36–44. doi: 10.1016/j.plaphy.2016.09.001. [DOI] [PubMed] [Google Scholar]
- Yesbergenova et al. (2005).Yesbergenova Z, Yang G, Oron E, Soffer D, Fluhr R, Sagi M. The plant Mo-hydroxylases aldehyde oxidase and xanthine dehydrogenase have distinct reactive oxygen species signatures and are induced by drought and abscisic acid. Plant Journal. 2005;42(6):862–876. doi: 10.1111/j.1365-313X.2005.02422.x. [DOI] [PubMed] [Google Scholar]
- Zarepour et al. (2012).Zarepour M, Simon K, Wilch M, Nieländer U, Koshiba T, Seo M, Lindel T, Bittner F. Identification of superoxide production by Arabidopsis thaliana aldehyde oxidases AAO1 and AAO3. Plant Molecular Biology. 2012;80(6):671. doi: 10.1007/s11103-012-9975-1. [DOI] [PubMed] [Google Scholar]
- Zdunek & Lips (2001).Zdunek E, Lips HS. Transport and accumulation rates of abscisic acid and aldehyde oxidase activity in Pisum sativum L. in response to suboptimal growth conditions. Journal of Experimental Botany. 2001;359(52):1269–1276. doi: 10.1093/jexbot/52.359.1269. [DOI] [PubMed] [Google Scholar]
- Zdunek-Zastocka et al. (2004).Zdunek-Zastocka E, Omarov RT, Koshiba T, Lips HS. Activity and protein level of AO isoforms in pea plants (Pisum sativum L.) during vegetative development and in response to stress conditions. Journal of Experimental Botany. 2004;55(401):1361–1369. doi: 10.1093/jxb/erh134. [DOI] [PubMed] [Google Scholar]
- Zdunek-Zastocka (2008).Zdunek-Zastocka E. Molecular cloning, characterization and expression analysis of three aldehyde oxidase genes from Pisum sativum L. Plant Physiology and Biochemistry. 2008;46(1):19–28. doi: 10.1016/j.plaphy.2007.09.011. [DOI] [PubMed] [Google Scholar]
- Zdunek-Zastocka (2010).Zdunek-Zastocka E. The activity pattern and gene expression profile of aldehyde oxidase during the development of Pisum sativum seeds. Plant Science. 2010;179(5):543–548. doi: 10.1016/j.plantsci.2010.08.005. [DOI] [PubMed] [Google Scholar]
- Zhang et al. (2012).Zhang Y, Mao L, Wang H, Brocker C, Yin X, Vasiliou V, Fei Z, Wang X, Blazquez MA. Genome-wide identification and analysis of grape aldehyde dehydrogenase (ALDH) gene superfamily. PLOS ONE. 2012;7(2):e32153. doi: 10.1371/journal.pone.0032153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong et al. (2010).Zhong R, Thompson J, Ottesen E, Lamppa GK. A forward genetic screen to explore chloroplast protein import in vivo identifies Moco sulfurase, pivotal for ABA and IAA biosynthesis and purine turnover. The Plant Journal. 2010;63(1):44–59. doi: 10.1111/j.1365-313X.2010.04220.x. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The following information was supplied regarding data availability:
There is no data to publish; this article is a literature review.