Abstract
Cancer is associated with a comprehensive burden that significantly affects patient’s quality of life. Even though patients’ disease condition is improving following conventional therapies, researchers are studying alternative tools that can penetrate solid tumours to deliver the therapeutics due to issues of developing resistance by the cancer cells. Treating cancer is not the only the goal in cancer therapy; it also includes protecting non-cancerous cells from the toxic effects of anti-cancer agents. Thus, various advanced techniques, such as cell-based drug delivery, bacteria-mediated therapy, and nanoparticles, are devised for site-specific delivery of drugs. One of the novel methods that can be targeted to deliver anti-cancer agents is by utilising genetically modified non-pathogenic bacterial species. This is due to the ability of bacterial species to multiply selectively or non-selectively on tumour cells, resulting in biofilms that leads to disruption of metastasis process. In preclinical studies, this technology has shown significant results in terms of efficacy, and some are currently under investigation. Therefore, researchers have conducted studies on bacteria transporting the anti-cancer drug to targeted tumours. Alternatively, bacterial ghosts and bacterial spores are utilised to deliver anti-cancer drugs. Although in vivo studies of bacteria-mediated cancer therapy have shown successful outcome, further research on bacteria, specifically their targeting mechanism, is required to establish a complete clinical approach in cancer treatment. This review has focused on the up-to-date understanding of bacteria as a therapeutic carrier in the treatment of cancer as an emerging field.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11095-022-03240-y.
KEY WORDS: bacteria-assisted therapy, cancer treatment, drug delivery, therapeutic carrier for chemotherapy
Introduction
Cancer is defined as a collection of diseases characterised by the uncontrolled proliferation of abnormal cells that are invasive and possess the potential to spread from the primary site to normal healthy tissues (1, 2). The spread of cancer is known as metastasis. Globally, cancer is the second leading cause of death, with an estimated 9.6 million deaths in 2018 (3). Statistics in 2017 reported 1,688,780 cancer cases and approximately 600,920 cancer-related deaths (4). There are three common types of cancer treatment strategies, which include surgery, radiotherapy, and chemotherapy. Recently, efforts were taken to further improve cancer treatment strategies. Biological therapeutics, for example, antibodies, peptide-mimetics, and a few cytokines showed target-specific effects, and they are relatively safer to be used in cancer treatment (5). Furthermore, studies also showed that engineered nanoparticles have selectivity in detecting and destroying tumour cells (6–9). However, physiological and physical limitations for incorporating engineered nanoparticles in cancer treatment still exist. These include protein adsorption, phagocytic sequestration, diffusion, flow, and shear forces which reduce the extent of diffusion of nanoparticles to the tumour cells (6, 10).
On the other hand, the use of bacteria in cancer treatment has been acknowledged many decades ago. William Coley, an American physician, was the first scientist to document the use of bacteria and bacterial toxins in cancer treatment in 1890 (11). He observed that the neck cancer of one of his patients started to recover in the presence of an erysipelas infection. On top of that, he experimented by using a vaccine constructed from killed Streptococcus pyrogens and Serratia marcescens to produce an infection along with fever and found that this vaccine successfully cured sarcomas, lymphomas, carcinomas, and melanomas. Besides, bacterially derived toxins, known as ‘Coley toxins,’ also proved to have anticancer activity (11). In addition, an obligate anaerobe Clostridium novyi and several facultative anaerobic bacterial strains have been observed to localise and grow selectively at the tumour site (12, 13). Although the precise mechanism of selective homing is not well explained, it is believed that the tumour microenvironment may be more suitable for bacterial growth due to the protection from the host immune system and the availability of nutrients (5).
Although it is understood that bacterial use in cancer treatment is workable, there are limitations to this approach. For example, a study demonstrated that some bacterial species could enter and grow inside the tumour cells while carrying the therapeutic agent and the elimination of the bacteria with the administration of antibiotics (14). Studies also showed that using bacteria in treating cancer only provides short-term benefits, and there might be a risk of tumour recurrence. Safety is always a concern for the use of bacteria in biomedical applications due to its pathogenicity. Thus, attenuated bacterial strains are recommended as a carrier in cancer therapy. Intratumoural injection is also recommended to replace systemic injections to minimise the immunogenicity risk (5, 15).
Researchers believe that bacteria can be used as a precisely controlled and highly specific drug delivery carrier by improving its natural characteristics to colonise in tumours and preferentially implement genetic circuitry. This target-specific feature helps in improving cancer therapy by significantly reducing the toxic side effects. Other advantages of using bacteria in cancer treatment include patient tolerance, and intensive immune stimulation by inducing cytotoxic T cells through major histocompatibility complex class I pathway (16).
Researchers have discussed the mechanism behind the use of bacteria as a carrier in cancer treatment. It is established that certain bacterial species such as Salmonella, Clostridium, and Escherichia coli have natural adhesion properties to tumours (12, 17). Upon intra-tumoural or systemic injection, it was found that the bacteria were able to trace the tumour microenvironment, propel and accumulate or even colonise at the tumour site, contributing to regression of cancer (18). Thus, scientists utilised this approach to incorporate/attach drugs to bacteria so that targeted cancer treatment can be achieved. Moreover, researchers also found that mutation in the purI gene, which makes the modified strain unable to synthesise purine, results in tumour-specific localisation. This is because bacteria with mutated purI gene requires external sources to support their replication while cancer cell has a highly detectable rich source of purine as compared to healthy tissues; therefore, tumour-targeted expression can be done by mutating purI gene in bacteria (17, 19). The most commonly used bacteria in treating tumours are Salmonella typhi, Bifido bacterium, Salmonella choleraesuis, Vibrio cholerae, Listeria monocytogenes, and Escherichia coli. Another study revealed the successful application of the attenuated Salmonella strain in suppressing melanoma (11, 20). Besides, liposomes attachment to different types of bacteria also showed fruitful outcomes in cancer treatment and/or diagnosis. For example, bacteriabots created by Kojima et al. consisting of attachment of Vibrio alginolyticus to 20 μm liposomal particles showed significant improvement in the motility function (21). Similarly, researchers developed a bactriobot combined with liposomes loaded with paclitaxel. Liposomal bactriobot was constructed by binding streptavidin on liposomes with biotin molecules on the outer membrane of bacteria. The result of motility analysis showed higher velocity of bacteria-actuated liposomes (3.09 ± 0.44 μm/s) compared to only liposomes as bacteria could drive drug-loaded liposomes to tumour cells. Concurrently, bat robot has shown better tumour killing and tumour targeting properties on breast cancer cells than paclitaxel-loaded liposomes (22). Some concept of on-demand bacterial activation by nanocoating has also been explored to enhance therapeutic effectiveness (23). Thus, the present review highlighted the mechanisms of drug delivery through bacteria, use of live, non-living bacteria, and minicells as activators of carrier for drug delivery with higher specificity than nontargeted nanodrug or conventional anticancer drug formulation in cancer treatment. We have also highlighted the outstanding challenges and clinical implications of bacteria as a drug delivery carrier.
Mechanism of Drug Delivery Through Bacteria
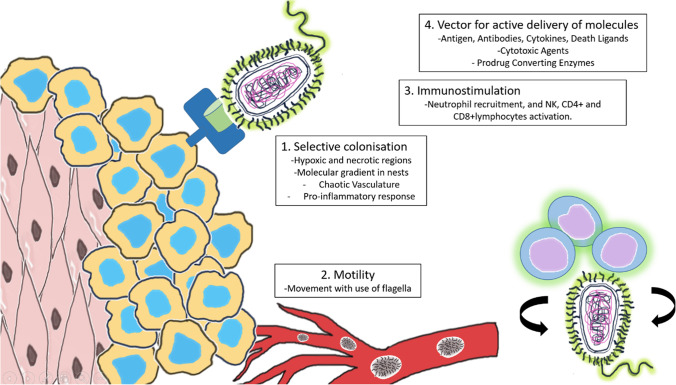
The major concern in bacterial therapeutics is the selection of potential species without pathogenicity. Few bacterial species have been identified, which usually colonise in the tumour region due to the supportive microenvironment from the tumour and protection from the host cells. Although the precise mechanism of bacterial therapeutics is not well understood, there are four different mechanisms have been predicted which might be involved in the process to cause tumour suppression. All these mechanisms of controlling cancerous cells have been summarised in Fig. 1. In addition, literature revealed several evidences of cancer treatment using bacteria-based drug delivery. Such recent studies in the field of bacteria synchronised delivery in cancer therapy have been summarised in Table I (24). These studies are evidencing advancement of this novel deliveries, where numbers of bacteria have shown their potential to deliver therapeutics in cancer microenvironment to control the devastating disease condition.
Fig. 1.
The mode of action of bacteria in anti-cancer therapy.
Table I.
Details of engineered microbes used in cancer therapy studies in past 7 years. Table adopted and modified with permission from Sieow et al. (24)
| Bacteria | Therapeutic payload | Animal model/ cell lines | Outcome | Reference |
|---|---|---|---|---|
| Escherichia coli | CD47 nanobody | BALB/c or C57BL/6 mice bearing A20, 4 T1, and B16-F10 tumours | Intratumoural injection of engineered microbe increased activation of tumour-infiltrating T cells and stimulated significant tumour regression in various tumour-bearing mouse models; treatment also triggered systemic tumour antigen-specific immune responses, because additional tumours that did not receive intratumoural injections experienced slightly weaker growth inhibition. | (25) |
| Expression of myrosinase to convert dietary glucosinolate into sulforaphane | Azoxymethane (AOM) and dextran sulphate sodium (DSS)-induced tumours in BALB/c mice | Significant inhibition of tumour growth and tumour counts (~75% reduction) in colorectal region of induced-colorectal cancer mice. | (26) | |
| Expression of biosynthetic gene clusters of glidobactin, luminmide, and colibactin | Female NMRI nude mice bearing UT-SCC-5 tumours | Colibactin-producing EcN significantly inhibited tumour growth in mice (25% of tumour weight of control group); glidobactin and luminmide-producing EcNs also inhibited tumour growth in mice but to a lesser degree (~50% of tumour weight of control group). |
/ (27) |
|
| E. coli Nissle | Tum-5 | C57BL/6 mice bearing B16-F10 melanoma | Significantly inhibited solid tumour growth in mice; inhibited neovascularization in tumour areas; reduced expression of PECAM-1. | (28) |
| Tum-5 and p53 | BALB/c nude mice bearing SMMC-7721 tumours | Significantly inhibited solid tumour growth in mice; inhibited neovascularization in tumour areas; combined administration of p53 and Tum-5 more potent than single administration of either therapeutic protein. | (29) | |
| PD-L1 and CTLA-4 nanobodies | GM-CSFBALB/c mice bearing A20 and CT26 tumours | Increased systemic memory T cell populations, increased activation of tumour-infiltrating T cells, and stimulated significant tumour regression in mice. Combined administration of PD-L1nb, CTLA-4nb, and GM-CSF was more potent than single or combined administration of the two immune checkpoint nanobodies | (30) | |
| Expression of diadenylate cyclase to synthesize STING-agonist cyclic di-AMP | Specific pathogen-free mice bearing B16-F10 and A20 tumours | Significant dose-dependent tumour rejection of B16-F10 and A20 tumour-bearing mice. Percentage survival of mice also dependent on administered therapeutic dose | (31) | |
| Salmonella typhimurium | IFN-γ | C57BL/6 mice bearing B16-F10 tumours | Significant NK cell-dependent inhibition of B16-F10 melanoma tumour growth and prolonged survival in B16-F10 mouse tumour model | (32) |
| Vibrio vulnificus flagellin | BC57BL/6 mice bearing MC38 and HCT116-luc2 tumours | Significant suppression of MC38 tumour growth and prolonged survival in MC38 mouse tumour model; suppressed tumour growth and metastasis in HCT116 xenograft model; Induction of M1-like macrophage polarization, which secretes tumour-suppressive cytokines and nitric oxide. | (33) | |
| Endostatin | BALB/c mice bearing CT26 tumours and C57BL/6 mice bearing B16-F10 melanoma | Increased apoptosis level and suppression of tumour angiogenesis within tumour tissues. | (34) | |
| hlyE and 5-FU (chemo drug); hlyE, CCL21, and CDD-iRGD | BALB/c mice bearing MC26-LucF tumours | Significantly inhibited tumour growth and prolonged survival of mice. Combined administration of therapeutics showed better efficacy than administration of each therapeutic individually. | (35) | |
| TGFα-PE38 | BALB/c mice bearing CT26 and 4 T1 tumours and C57BL/6 mice bearing MC38 tumours | Release of TGFα-PE38 by phage lysis system and secretion via signal peptide significantly inhibited solid tumour growth in mice and prolonged survival. | (36) | |
| DNase I and triptolide | BALB/c mice bearing B16-F10 tumours | Combination of DNase I delivery and triptolide inhibited tumour growth and enhanced survival rate in mice; at end-point, 26.32% of mice that received both therapeutics had complete elimination of tumours; DNase I monotherapy completely eliminated tumours for 15.79% of treated mice. | (37) | |
| Expression of L-asparaginase to convert asparagine to aspartateC57 | BL/6 mice bearing MC38 tumours | Significant inhibition of tumour growth (75% reduction in size) and prolonged survival in mice (100% survival rate after 20 days) | (38) | |
| IDO (shIDO) and anti-PD-1 antibody | BALB/c mice bearing CT26 tumours and C57BL/6 mice bearing MC38 tumours | Induced intratumoural cell death in colorectal cancer mouse models; co-administration of anti-PD1 and shIDO was not more effective than shIDO monotherapy. | (39) | |
| Alpha subunit of inhibin (sh-INHA) | BALB/c mice bearing CT26 tumours and C57BL/6 mice bearing B16-F10 melanoma | Significant inhibition of tumour growth and prolonged survival in mouse models; suppression of INHA expression; induced caspase activation and downregulated expression of antiapoptotic Bcl-2 and Bcl-xL in in vitro studies. | (40) | |
| sh-PD-1 and Pimozide | C57BL/6 mice bearing B16-F10 melanoma | Combined administration of sh-PD-1 and pimozide induced tumour apoptosis and prolonged survival in mouse models compared with individual monotherapies; also increased tumour infiltration of T cells, increased spleen CD4+ T cells, CD8+ T cells, and NK cells, and reduced spleen Tregs. | (41) | |
| sh-Sox2 and HM-3 peptide | BALB/c mice bearing A549 tumours | Combined administration of sh-Sox2 and HM-3 peptide induced significant inhibition of tumour growth in mouse models and was more effective than sh-Sox2 monotherapy; HM-3 peptide inhibited microvessel formation in tumours. | (42) | |
| Bifidobacterium longum | 420Cell wall-anchored tumour antigen WT1 protein | C57BL/6 N mice bearing C1498-WT1 tumours | Significant inhibition of WT1-expressing tumour growth by WT1 epitope-specific cytotoxic T cells; increased stimulatory cytokine production. |
/ (43) |
| Tumstatin | BALB/c mice bearing CT26 tumours. | Significantly inhibited tumour growth and induced apoptosis of tumour vascular endothelial cells in CT26 mouse model. | (44) |
Endogenous Cell Death-Inducing Agents
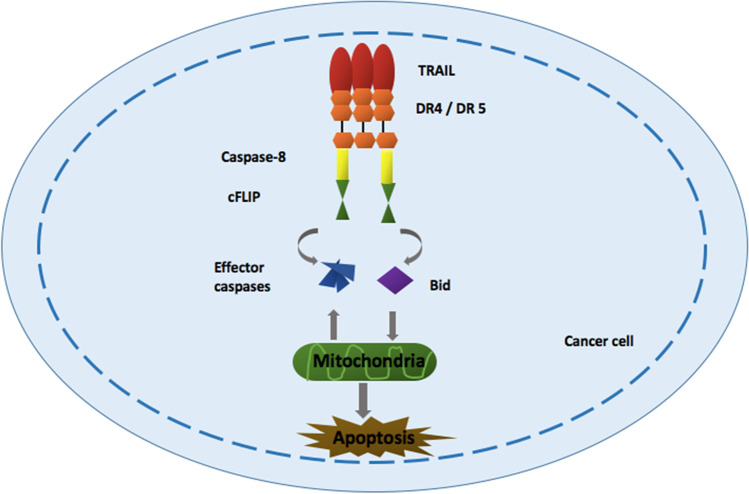
Using endogenous cell-death-inducing molecules will be one of the options as it can be delivered in a localised and controlled manner (e.g., spatio-temporal control). However, it is difficult to reach this objective due to the natural tumour tropism of S. typhimurium to concentrate around tumour. A secreted cytokine is known as the TNF-related apoptosis-inducing ligand (TRAIL) actuate the cell death of tumour cells by activating the route of extrinsic apoptosis (45). TRAIL has been involved in several clinical trials to access the antitumour potential of recombinant human TRAIL, and it emanated as a potential therapeutic agent. This is because of the extraordinary properties of selectively inducing cell death in cancer cells without affecting the normal cells (46). It is a ligand for death receptors 4 and 5 (DR4 and DR5) that will trigger the initiator caspase-8 to stimulate downstream effector caspases (−3, −6, and − 7), which will eventually promote apoptosis (47). The mechanism is elucidated by Fig. 2.
Fig. 2.
Mechanism of apoptotic induction of cancer cells by TNF-related apoptosis-inducing ligand. TRAIL actuate tumour cell death via acting as ligand for DR4 and DR5 that will trigger the initiator caspase-8 to promote apoptosis. Abbreviations: the TNF-related apoptosis-inducing ligand (TRAIL, cellular FLICE-inhibitory protein (cFLIP), death receptors 4 and 5 (DR4 and DR5), Bcl-2 homology 3 interacting-domain death agonist.
Immune Effectors
The host immune system controls the transformed or malignant cells from forming a firm basis for further progression or development. The proper function of the immune system is based on the organised arrangement of cytokine signalling events. Most of the cytokines are not the option to be selected for cancer therapy due to the effects of systemic toxicity that have been identified. However, some cytokines such as interleukin-4 (IL-4), IL-18, and TNFSF14 (or LIGHT) delivered by S. typhimurium have been shown to significantly reduce the tumour growth upon localised synthesis through the triggering of immune response in the region of juxta-tumoural. The activation of IL-4 and/or IL-18 in tumours increase serum IFN-γ. The growth of stromal fibroblast, the major source of angiogenic factors, is inhibited by IL-18 through tumour neo-vascularisation. TNFSF14 is one cytokine from the TNF family that functions to stimulate T cell proliferation, promote the growth of dendritic cells, and activate apoptosis of cancer cells (5, 19).
Tumour Immunization
Stimulation of the immune system is another choice that can be used to limit and or suppress tumour growth. Based on recent studies, the researchers suggested that the stimulation of an adequate innate immune system is essential to efficiently maintain an acquired immune system response. Besides, the researchers also have suggested that those tumours developed in vivo should undergo a process called ‘immunoediting’. There are 3 ‘E’s of cancer immunoediting, which represents elimination, equilibrium, and escape. In the first phase of elimination, the immunogenic tumours are eradicated aggressively in which the size of tumour cells is restricted significantly. However, not all the tumour cells are being killed and the remaining such cells and immune effectors that are specific to the tumour would enter the equilibrium phase. In the equilibrium phase, none of the tumour cells can grow aggressively or is susceptible to attack by the immune system. This forms a small population of immune-attack-resistant tumour cells due to the consequences of clonal evolution. On the other hand, recruiting immune suppressor cells functions to reconfigure the tumour microenvironment. This monitors killer cells and the types of angiogenic cells that engage in proliferation, increasing cancer cells’ survival. Escape represents the last phase in the immunoediting process. This stage is established when tumour undergoes complete mutation to evade the elimination phase and further growth. Hence, antigenic protein might need to resupply to the immune system during the elimination phase to eradicate most of the tumour (48).
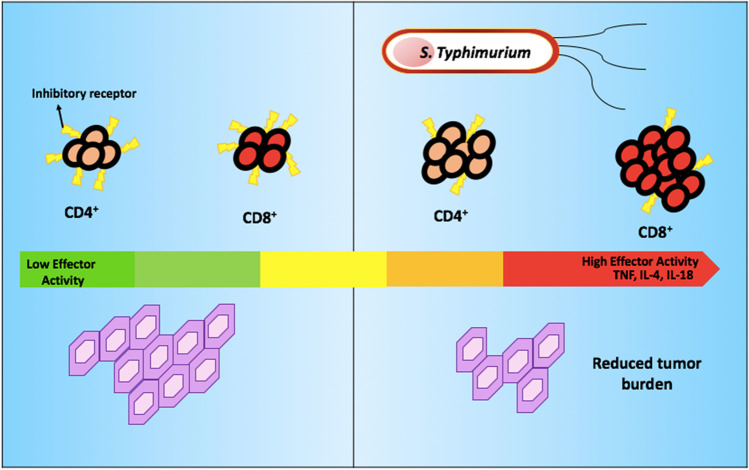
S. typhimurium was involved in the formation of CD8+ cells with significant response. Based on studies, the findings suggested CD8+ cells that are specific to the tumour were found in tumour milieu but it was in an inactive state. However, when S. typhimurium is expressed to antigen, the immune system was activated. This resulted in the re-activation of CD8+ and CD4+ T cells in which the tumour growth was able to be controlled. Other than that, the principles of delivery antigen from S. typhimurium type-3 secretion system (T3SS) would benefit in cancer immunotherapy because advanced, in-depth knowledge regarding the composition of tumour antigen is not required. Based on studies, tumour destruction is dependent on the immune system. A recent report declared that B cells were important in controlling the S. typhimurium (19, 49). For example, heat-killed S. typhimurium given to cancerous mice showed no improvement in the outcome. This is because the oncolytic activity of S. typhimurium and immune serum completely inhibits S. typhimurium from affecting cells in culture due to the formation of neutralising antibodies. However, they show synergism with adoptive T cell therapy in tumour eradication (50). This process has been illustrated in Fig. 3.
Fig. 3.
Presence/absence of S. typhimurium and the responses of CD4+ and CD8+ T cells in tumour burden. S. typhimurium triggers the infiltration of the immune cells in cancer cells and persuades self-destruction with delay in tumour cell migration.
Tumour Growth Control Mechanism
It has been assessed and the advantages have been indicated in the use of mouse models. S. typhimurium can excrete plasmids into the host cytosol. Then the plasmids express the recombinant gene product from vertebrate promoters when it moves into the cell nucleus. As a result, none of the bacteria can be cultured from such cells. Another role of S. typhimurium is to diminish tumour mass by transporting recombinant DNA into it. For instance, mice were orally vaccinated against S. typhimurium through actuation plasmids which involved full-length vascular endothelial growth factor receptor 2 (Vegfr2) cassette. Vegfr2 is known as tyrosine kinase receptor and is activated when Vegf binds to it. Vegfr2 upregulation is required for endothelial cells to undergo angiogenesis effectively to control the tumour growth and transmit to another part of the tissues. Almost all tumour mass can be controlled independently of protein cancer cell expression because Vegfr2 is initiated by the non-cancerous cells in tumour. An interruption of peripheral tolerance against a self-antigen occurred due to the presence of Vegfr2-specific spleen-derived CTLs. A recent update claims that another version of Vegfr2, which is in its mini-gene form, can result in T cell mediated retardation of the formation of new blood vessels, together with a tumour protective immunity (13, 51).
Research evidences of potential cancer treatment using engineered bacterial tool should require further investigation to demonstrate importance of combination therapy, which might be more effective than only bacteria-based therapy and to enhance the possibilities in clinical translation.
Bacteria in Cancer Therapy
This section is focused on three categories of bacterial use in cancer therapy. The first subsection describes the use of bacterial for activation of anticancer prodrug delivery at targeted site, role of bacteria in immunoediting, and bacterial exotoxins as antitumour agents. Second subsection emphasised the use of ghost bacteria as a carrier for the delivery of drug and genetic material. The last subsection describes the use of bacterial as carrier for anticancer nanocarriers.
Bacteria as Activator, Therapeutic Agent, and Carrier for Delivery of Anti-cancer Drug
To develop bacterial therapeutics for cancer, the identification of species with minimal pathogenicity to host as well as maximised selectivity to malignant cells is required. Many Gram-positive and -negative bacteria have been isolated, and its colonising ability in animal tumours has been compared with that in host normal tissues. Although specific mechanisms are unknown, it is found that the microenvironment in the tumour is more bacteria-friendly, as it protects the microorganism from the host’s defences (2, 17).
Unlike normal tissue physiology, tumour cells have leaky capillaries that pose a challenge for the penetration of drugs. Thus, in cancer therapy, the administration of pro-drugs is usually preferred in overactive forms due to increased penetration power. However, upon reaching the tumour site, the pro-drug must be activated by an enzyme (reductase) and the active form should be retained in the tumour site to allow the exertion of its effects. To facilitate the conversion of a pro-drug into its active form, hypoxia, is a significant characteristic of tumour cells that can be exploited. The hypoxic environment activates nitro reductase, which certain bacteria strains can secrete. Anaerobic bacteria such as strains of Clostridium and Salmonella sp. are known to prosper in hypoxic and necrotic tumour areas as they can specifically target oxygen-poor cancerous tissues. Strains of Clostridium sp. have been employed to convert pro-drugs to nitrogen mustard, which has cytotoxic properties and has shown a good effect in mouse cancers (11, 17). In a recent study, bacterial nitro reductase enzyme was explored for the conversion of product to its active form. They have engineered abroad-range nitroreductase, E. coli NfsA, for better activity against dinitrobenzamide aziridine CB1954; duocarmycin analogue nitro-CBI-DEI and 5-nitroimidazole metronidazole. The co-culture of bacteria with CB1954 and nitro-CBI-DEI showed significant fluorescence after conversion of non-florescent prodrug into active fluorescent metabolite. Moreover, cytotoxic study on HEK-293 cells was performed to compare prodrug activation efficacy among wild NfsA and other engineered enzymes and results showed variant 11_78 have significant efficiency towards all three prodrugs with higher reduction in cancer cell viability. This research offers targeted cellular ablation with higher therapeutic outcomes (52).
Some bacterial species of the genus Clostridium are spore-forming. A strategy called ‘combination bacteriolytic therapy’ (COBALT) has been utilised to combine these bacterial spores with therapeutic drugs like docetaxel and mitomycin C. The spores effectively multiply and germinate once they reach the hypoxic tumour, after which the chemotherapeutic agent gets activated (12). To increase its specificity to cancerous tissues, the Clostridial spores’ activity can be genetically engineered to achieve increased 5-fluorouracil concentration. Together with a specific concentration of chemotherapeutic drugs, it would lead to minimal damage to healthy cells. However, bacterial spores have their limitations when treating small metastases (53).
Enterica serovar typhimurium, a flagellated subspecies of the Salmonella enterica (hereafter referred as S. typhimurium), although is not spore-forming, has a natural tendency to concentrate around tumour cells. Since it is not exclusively anaerobic, S. typhimurium is known to pose a threat to both healthy cells as well as cancerous tissues. However, incorporating the bacteria strain with a hypoxia-inducible promoter can overcome its natural lack of specificity to malignant cells. VNP20009, one of the most significant strains of attenuated S. typhimurium has shown a preference for tumour cells over healthy tissues at ratios of 1000:1 (54, 55).
To further enhance the localisation to tumour site, tumour homing peptides can be engineered onto bacterial strains. This protein expressed on the outer membrane of the bacterial cells aids in recognition of subsequent recruitment of antigen to the tumour site. Affibodies (Proteins designed to bind targets such as upregulated receptors in cancer cells) like HER2 are utilised to bind to upregulated receptors on cancer cells, whereas synthetically formulated adhesion molecules called adhesins when expressed in tumour-bearing mice, proved more effective in colonisation compared to natural bacteria (55).
Moreover, several amino acids released by tumours in mice metastatic cancer, such as leucine and arginine have high specificity and sensitivity between Salmonella strains which decreases angiogenesis and enhances tumour necrosis. The local synthesis and delivery of cytokines such as IL2, IL4, IL18 and LIGHT activates the immune response in juxta-tumoural regions and precipitates the synthesis of other immunological agents, which eventually leads to better infiltration into tumour cells and enhanced functions of cytokines. Furthermore, another cytokine known as TRAIL (TNF-related apoptosis-inducing ligand) induces apoptosis by the stimulation of downstream caspases (−8, −3, −6, −7), which was proven when the murine TRAIL expressed by attenuated S. typhimurium effectively reduced the growth of breast tumour in BALB/c mice under certain promoters (46, 56). In another study researcher investigated the intracellular delivery of protein drug to specific tumour site and results indicated the significant reduction in tumour growth (57).
Since the immune system of a healthy individual normally suppresses tumour growth, the tumour cells when grown in vivo, have exhibited a phenomenon called ‘immunoediting’ which is a process that involves elimination, equilibrium, and escape. Following a natural weakening of the immune system, the last stage of the process happens when the tumour cells undergo a sufficient level of mutation, evading elimination and ‘escaping’ the effect of immune cells. Prior to the final stage, supplementing the immune system with appropriate immunogenic proteins may be beneficial. S. typhimurium can be utilised as a carrier to deliver these proteins via the T3SS mechanism, in which the assembled components resemble a syringe and needle. When the fusion protein, sopE-NY-ESO-1 was delivered via T3SS by S. typhimurium, the CD8+ cell-mediated response against melanoma-expressed antigen was significantly enhanced (5, 19).
Other than the utilisation of bacterial spores as a delivery agent, some bacteria strains induce tumour necrosis via more than one mechanism, as studied thus far. Bacteria strains like Bifidobacteria, Clostridium and Salmonella sp. secrete molecules called exotoxins using their T1SS machinery for self-survival. These exotoxins express anti-tumour activity by CD8+ cell - mediated response as observed when the T1SS mechanism was used to introduce chimeric human PSA into mouse mastocytoma. Recombinant S. typhimurium was also used to induce tumour necrosis by secreting Shiga toxins in the tumour microenvironment (5).
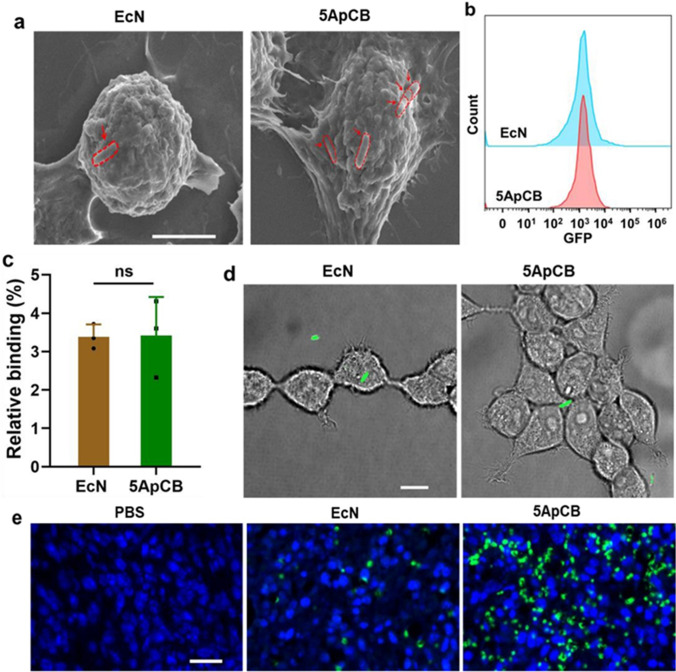
On the same concept, Zeng et al. conjugated aptamers on the bacterial surface (ApCB) via cytocompatible amidation procedure to enhance its tumour specific accumulation which may result in better therapeutic efficacy after systemic administration. Averagely, 2.8 × 105 aptamers conjugated per bacterial cell and showed 2–4 fold enhanced accumulation at tumour site in comparison to unmodified E. coli. This was directly evident from SEM illustration that more bacterial attachment to the surface of 4 T1 cells with ApCB after co-incubation compared to Escherichia coli Nissle 1917 (EcN) (Fig. 4a) which is consistent with flow cytometric analysis results. However, no significant difference in flow cytometric analysis was observed when EcN and ApCB cells were co-incubated with 293 T cells (Fig. 4b, c). The LCMS results also showed that the binding efficiency of ApCB is same for 293 T cells and EcN cells (Fig. 4d). Additionally, binding efficiency of ApCB was further investigated on a 4 T1 tumour model mice after IV dose. After 12 h of injection, tumour were sampled and stained with 4′,6-diamidino-2-phenylindole (DAPI), and FITC-labelled anti-Escherichia coli. A higher accumulation was evident on tumour tissue which was stained with FITC-labelled anti-Escherichia coli (green florescence), which represent specific binding efficacy of ApCB (Fig. 4e). This research supports that ApCB is capable in enhancing cancer cell biotherapy and intertumoural localisation (58).
Fig. 4.
(a) SEM images of 4 T1 cells after incubation with ApCB and EcN cell, bacteria is circled by red dotted lines (b and c) Flow cytometric analysis of 293 T cells after co-incubation for 2 h with EcN and 5ApCB (error bar represent standard deviation at n = 3 independent experiments), Student’s t test (two-tailed), ns- no significance, (d) LSCM images of 293 T cells after incubation with EcN and 5ApCB, green colour denote green fluorescent protein production in EcN (e) Confocal images of tumour tissues sectioned at 12 h after intravenous injection of bacteria. Blue and green colour indicate nuclei stained with 4′,6-diamidino-2-phenylindole and EcN stained with FITC-labelled anti-Escherichia coli, respectively. The adopted figure is under common creative licence and used with permission from Geng et al. (58).
On the other hand, bacteria are used to enhance the durability and uniform distribution of anticancer drug throughout the tumour. In this study researchers used bacteria to reprogram the immune microenvironment by spatiotemporally controllable distribution of therapeutics entrapped in bacterial cells. In-vivo mouse and the ex vivo human study indicates the colonisation of bacteria with drug in tumour due to intertumoural hypoxic condition (59).
Non-Living Bacteria as a Vehicle in Anti-Cancer Therapy
The Bacteria Ghost (BG) is a newer technology to transport drugs, vaccines and DNA via delivery vectors. BGs are non-living, non-denatured empty bacteria that are enclosed by an envelope of gram-negative bacteria by controlled expression of the cloned lysis gene. E.BGs do not contain any cytoplasmic content which includes chromosomal and plasmid DNA but it preserves its’ cellular surface morphological structure; outer membrane, inner membrane and peptidoglycan (56, 60). In 1966, the function of gene E in the lysis of Escherichia coli was well studied and understood. Gene E was the first lethal gene in bacteria that could be silenced within the plasmid. Gene E has the ability to produce a membrane protein with the capability to oligomerise into a transmembrane tunnel structure (61). Thus, there is an analysis conducted on the primary structure of protein E, hydrophobic region at its N-terminal end is involved in co-translational integration to be part of the cytoplasmic membrane of E. coli (62). The analysis of the hydropathicity regions of protein E showed an E-specific lysis tunnel spanning the inner membrane (IM) and outer membrane (OM) adhesion sites within the host cell. Lysis initiated by gene E will cause the release of the entire cytoplasmic content and the drug into the host cell. In contrast, periplasmic content will remain in the empty cell envelope. Schön et al described a three-phase model for the process of E-mediated tunnel formation. Firstly, protein E will be integrated into the IM with C-terminal region. Secondly, protein E will undergo conformational changes which translocates the C-terminal domain to the periplasmic space accompanied by oligomerisation. Lastly, the fusion of IM and OM at the membrane adhesion sites is initiated by the exposition of C-terminus of protein E to the cell surface (63).
Besides E. coli, there are many other gram-negative bacteria which can undergo E-lysis and including Salmonella typhimurium, Klebsiella pneumonia and Pseudomonas putida. Moreover, BGs which contain DNA, tend to have a higher chance of being engulfed by antigen-presenting cells (APC) and tumour cells (64, 65). BG is relatively safe because it is a non-living bacterium that does not contain any internal harmful chemicals to produce cytotoxic and genotoxic effects. BGs also do not have the ability to revert to their pathogenic form to cause harm. Thus, it is thought to be a good alternative for vaccine development. The inner compartment of BGs can be inserted with single or multiple components of peptides, drugs or DNA. BGs have excellent DNA loading capacity which varies from 4000 to 6000 plasmid copies per BG depending on the concentration of the solution used (66). BGs can deliver their content to monocyte derivative dendritic cells, melanoma and macrophages. Cross-presentation of antigens delivered by BGs to dendritic cells will stimulate the activation of CD4+ and CD8+ T cells, producing an immune response. The outer membrane of a bacteria contains a layer of lipopolysaccharide which aids in enhancing maturation of dendritic cells, affecting endosomal acidification of dendritic cells and helping in refining cross-presentation of antigen (60, 67). For example, Chlamydia trachomatis BG vaccine prevents trachomatous conjunctivitis and blindness (68). Besides stimulating CD4+ and CD8+ T cells, BGs intact envelope provides stimulatory effects on tumour cells. Melanoma cells are known to have a similar ability as non-professional APC cells and undergo phagocytosis on both living and apoptotic cells. Melanoma cells have been recently shown to have an active response upon exposure to BGs, resulting in a higher rate of phagocytosis (69). Thus, gene delivery by BGs to immunocompetent cells can restore or initiate an immune response against tumour-associated antigen and induce the expression of the target gene by APC and tumour cells.
BGs will have to deliver the chemotherapy medications specifically to the directed site to exert its action, as these medications have cytotoxic properties. Thus, BGs are non-living and empty bacteria that preserve their morphological structure. Bacteria tend to have the pathogen-associated molecular patterns (PAMP) expressed on the surface. PAMPs are essential in enhancing the targeting properties toward tumour cells. A study revealed that melanoma cells have the ability to bind and internalise BGs without significantly influencing their viability and proliferation (69). Thus, a study was conducted on the efficiency of BGs loaded with DOX to bind and internalise in carcinoma cells and leukaemia cells. In this study, BGs, Mannheimia haemolytica, were loaded with DOX to deliver the medication to human colorectal adenocarcinoma cells (Caco-2 cells). It was assumed that DOX binds non-covalently to the inner membrane through the interaction between the amine sugar region of DOX and the charged membrane protein of BGs or BGs inner membrane and anthraquinone region of DOX. At the site of action, DOX exhibited efficient endocytosis ability after incubation with colon carcinoma cells and leukaemia cells. DOX was released from endo-lysosomal compartment and accumulated in the nucleus. The result obtained from this delivery system was positive where it significantly decreased the viability and proliferative ability of the cancer cells. The efficacy of BGs was two magnitude of order more cytotoxic ability than free DOX in a medium at a similar concentration. Furthermore, the differences in cell proliferation inhibitory effect is clearly seen when both free DOX and BGs loaded with DOX were removed from the cell culture. The free DOX was found to be accumulated at target cells after several hours while BGs were able to pass through the target cells to release their contents into the cytoplasmic region (70). The concentration of DOX in colon carcinoma cells delivered by BGs increased 42 times higher than the cells incubated in equivalent concentration of DOX solution. These results indicated that the loading and delivery capacities of BGs are sufficient to deliver a cytotoxic drug to cells that expresses p-glycoprotein to overcome resistance. P-glycoproteins are atypical translocating ATPase which exhibits low affinity towards ATP and high constitutive ATPase activity. P-glycoprotein tends to have unusually broad specificity for a hydrophobic compound, a common characteristic of chemotherapeutic drugs. Binding of drugs and ATP to p-glycoprotein will induce conformational changes on the transport protein, leading to the removal of drugs from the cells. Thus, detailed analysis conducted showed that BGs loaded with DOX are 300 times more effective in inhibiting cell proliferation at inhibitory concentration, when compared to free DOX. This may occur because BGs loaded with DOX are degraded within the endo-lysosome which allows it to escape from the p-glycoprotein efflux pump and accumulate in the cytoplasm. On the other hand, the free DOX will be pumped out from the cells leading to a lower concentration in the cell and ultimately lower efficacy on the proliferative inhibitory effect (70). In this study, the non-covalently bounded DOX was released from BGs in water for a period of 8 days. It was found that approximately 40% of DOX was released during the initial 24 h and 10% was released constantly for the remaining days. 40% release on the first day was surprisingly a high value because DOX was thought to strongly associate with the cells when the experiment was conducted on standard lysis buffer. BGs were suspected of having a large pore size which could have led to this situation. Thus, the limitation of this delivery system is limited to drugs that are strongly associated with bacterial membrane, membrane protein, and cell wall to achieve sufficient retention of the drug in the body. Drugs that interact weakly with the bacterial membrane, membrane protein, and cell wall of BGs will result in loss of the drug. To prevent loss of a weakly associated drug, the drug needs to interact with bacterial DNA which has been removed initially to create an empty BG, to increase the retention time of the drug in the BG system. This will provide a longer lasting effect and a better therapeutic response as large doses can accumulate in the cells (70).
Besides DOX, a recent study on oxaliplatin loaded in BGs to treat colorectal carcinomatosis is also found. Oxaliplatin is a commonly use systemic medication to treat colorectal cancer, but its usage has declined due to drug resistance. Thus, the study aimed to use the BGs delivery system to deliver the medication by passing the resistance mechanism. In vitro study on BGs loaded with oxaliplatin managed to induce the production of calreticulin. Calreticulin is an integral protein for the production of MHC class I protein. Besides calreticulin, release of ATP is also a classical indication of immunogenic cell death. The in-vitro study showed an increase in the release of ATP during the usage of BGs as a delivery system compared to oxaliplatin monotherapy. Interestingly, empty BGs do not induce significant increase in ATP release. On the other hand, an in vivo study shows a high tumour burden of approximately 1.6 g in the peritoneum of the control and BGs treated rats. Oxaliplatin monotherapy treated rats showed a lower tumour burden of approximately 0.5 g in the rat peritoneum. The combination of oxaliplatin and BGs loaded with oxaliplatin remarkably were tumour free. In addition, an increase in spleen size and mass were clearly seen in BGs treated rats. This may be due to the lipopolysaccharide layer of BGs that activates the immune system through toll-like receptor 4. In short, based on the in vivo study, combination of BGs and oxaliplatin may be a promising immunochemotherapeutic strategy to eliminate colorectal cancer (71).
BGs are potential envelope structures that do not only possess delivery properties for cancer but can also act as adjuvant and potent vaccine. There are currently many studies on the effectiveness of BGs for the delivery of cytotoxic drugs, biotherapeutics, and vaccine. However, the potential of BGs depends on the effectiveness of the delivery system to targeted cells. The possible potential of BGs is the replacement of live or attenuate vaccine with BGs to reduce risk of infection. BGs are empty bacteria which does not possess cytotoxic or genotoxic ability. Thus, it is a safer option as a vaccine. However, BG can mediate immunisation due to their envelope region. It may induce the release of many proinflammatory compound. These effects of BGs may not be suitable for immunocompromised patients (60). Besides that, loss of drugs due to large pores of BG is a concern. Thus, many studies and research can be conducted on improving the mechanism of delivery of BGs.
In conclusion. BG is a novel delivery system that helps to deliver content such as peptides, nucleic acid and drugs into a cell. BGs can also aid in vaccination because it possesses immunostimulatory effects. The lipopolysaccharide layer of the BG could interact with toll-like receptor 4 to induce and activate immune cells such as CD4+ and CD8+ T cells. There are many advantages of using BG drug as delivery system. Firstly, BGs are relatively safe due to absence of cytotoxic and genotoxic properties. Next, BG has a large storage capacity to store one or many peptides. BG is also an essential element that is needed to prevent drug resistance because it has the capability to prevent efflux of chemotherapeutics by p-glycoprotein. Furthermore, the concentration of chemotherapeutic drugs in BG can be reduced to produce a similar effect as the free drug, which ultimately reduces the risk of chemotherapy’s potential adverse effects.
Bacterial Minicells for Drug Delivery
Severe toxicity remains a major setback and a limiting factor in cancer chemotherapeutics, despite advancements in the pharmaceutical industry (72). Frequent high-dose administration of chemotherapeutic agents is considered necessary in chemotherapy to exhibit sufficient clinical response due to its low specificity towards cancerous cells, rapid clearance, and indiscriminate drug distribution. The development of targeted drug delivery systems (DDSs) such as liposomes, polymers, and nanoparticles envelop chemotherapeutic agents in a vehicle and targets receptors on the surface of tumour cells allowing an increased degree of specificity for the cells, at the same time reducing the side effects of chemotherapy (73). Targeting nanoparticles involves the integration of target sections such as antibodies and ligands onto the surface of nanosized carriers that are specific to cancer cell-surface receptors, allowing more selectivity and reduced toxicity resulting from the evasion of nonspecific binding, as well as a reduced multidrug resistance for chemotherapeutic agents. Recent researchers have revealed the potential of bacteria as carriers for chemotherapeutic agents in cancer therapy, whereby they specifically target primary and metastatic tumours (74). New technology has emerged for the packaging of various cytotoxic agents into nanosized particles (diameter of 100–400 nm) derived from bacteria, also known as minicells, a promising approach for the intracellular and selective targeted delivery to cancerous cells (74). In 1967, Howard Adler and colleagues were the first to describe minicells as non-living, nanosized cells that are anucleate, and produced by mutations in genes that regulate bacterial cell divisions, thereby suppressing cell fission sites. In studying various cellular processes such as isolating bacterial cell plasmid DNA, synthesis of proteins and viral infections, minicells were widely used as a vehicle (75). Recently, the focus of minicells has been shifted to their potential in drug delivery and vaccines that activate the immune system, largely due to its ability to preserve the parent cells’ virulence properties and without the ability to proliferate (75).
Generally, minicells are non-living spheres generated from mutated Gram-positive and Gram-negative bacteria such as S. typhimurium, E. coli, and S. flexneri. They are achromosomal and anucleate, allowing chemotherapeutic drugs to be loaded within its structure to target specific tumours. Minicells harbour only RNA, proteins, ribosome, membranes, plasmid DNA and peptidoglycans, hence cell division is absent. However, cellular processes such as replication and transcription of plasmid DNA and translation of mRNA may still be continued (73, 75). The spherical structure of minicells is described as small enough to slither out of the vessels inside the tumours, but large enough to be contained within normal blood vessels. Drugs are loaded into minicells by entrance through the nonspecific porin channels on the membrane of minicells, down the concentration gradient (76).
Minicells specifically target tumour cell surface receptors by using bispecific antibodies (BsAbs). One arm of the antibody is attached to the O-polysaccharide component on the surface of minicells. In contrast, the other arm is directed to the receptors on the surface of tumours. The targeted antibodies bind to the tumour-cell surface receptors, resulting in endocytosis of minicells by the tumour cell. Consequently, the intracellular cells breakdown and the drug is released into the cell (75, 76).
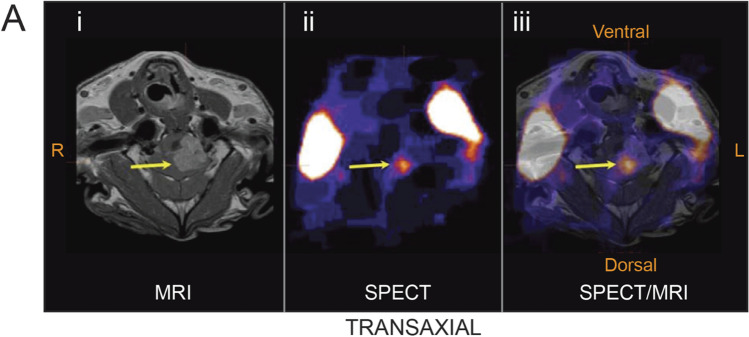
DOX-loaded minicells have exhibited successful tumour growth inhibition in mice breast, lung, leukemia or ovarian in the in vivo experiments (77). In the first human trial, minicells proved to be significantly safe and tolerable. The phase I trial involving patients with stage IV cancer and the infusion of paclitaxel-packaged minicells coated with antibodies targeting the epidermal growth factor receptor (EGFR) protein, ten out of twenty-eight patients showed disease stability after 6 weeks of treatment. Side effects reported were fevers that were short-lived and chills in some cases. Reducing the endotoxins presence in the membranes of minicells may help reduce the associated side effects of the treatment (78). DOX-loaded minicells were continuously tested in dogs with last-stage brain cancer (79). Biodistribution studies were performed to determine the distribution of minicell in brain tumours after systemic administration. Therefore, 123Iodine (123I) radiolabelled minicells were administered to dog with brain tumour and imaged using SPECT and MRI to locate the accumulation of minicells in the brain. Post contrast MRI clearly showed the location of brain tumour mass (Fig. 5i). Although SPECT images illustrate the focal point of accumulated radiolabelled micelles (Fig. 5ii). SPECT/MRI overlay images (Figure 5 iii) revealed the colocalization of minicells on tumour cite in brain (79).
Fig. 5.
Biodistribution of systemically administered 123I labelled minicells Dox in dogs with brain cancer. Arrow showed the tumour location at 3 h post minicells administration. Minicell accumulation was evident with single-photon emission computed tomography (SPECT) imaging. Merge of MRI and SPECT illustrate the location of minicells at the core of brain tumour. Figure adopted with permission from MacDiarmid et al. (79).
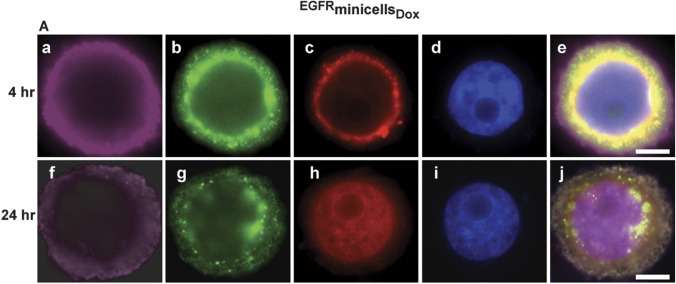
In contrast to other DDSs, minicells are able to package various chemotherapeutic agents with varying charge readily, structure, solubility and hydrophobicity such as cisplatin, DOX, 5-fluorouracil, vinblastine and carboplatin monastrol within its structure, where efforts to package these cytotoxic drugs into liposomes required the vehicle or drug to undergo heavy alterations, leading to reduced potency. It is revealed that as large as 10 million drug molecules can be packaged within minicells, as compared to liposomes’ ability to package only 10,000 drug molecules. Compared to conventional chemotherapy, different BsAbs are able to bind onto the minicell surface, allowing it to target various solid tumours and exhibit efficacy. To determine the targetability, minicells were incubated with MDA-MB-468 human breast adenocarcinoma cells and monitored for 48 h by confocal microscopy. MDA cell membrane was visualised by green fluorescence, Dox by its red autofluorescence and blue fluorescence is for DAPI staining of nuclei. As depicted from Fig. 6, non-targeted minicells did not internalised that was confirmed by absence of green or red fluorescence (Fig. 6a), in contrast BsAb attached to the minicell showed significant internalization (Fig. 6b). This may attributed to EGFR-mediated endocytosis into MDA cells. At 4 h overlaid image (Fig. 6e) showed blue nucleus, similar to DAPI-stained nucleus in Fig. 6d. However, the yellow colour in Fig. 6e suggest that the drug has not yet been released from the minicells. However, colonization and nuclear presence of DOX were confirmed at 24 h via violet fluorescence (Fig. 6j). In addition, minicells allow intracellular delivery of drugs within a tumour cell, steer clear of any leakage of drug from the vector when administered systemically, and provide a significant increase in therapeutic index of drugs with little or no toxic side effects. However, only four to ten drug molecules can be conjugated with one armed antibody. (80).
Fig. 6.
Dynamics of Minicell Attachment. Figure adopted with permission from MacDiarmid et al. (80).
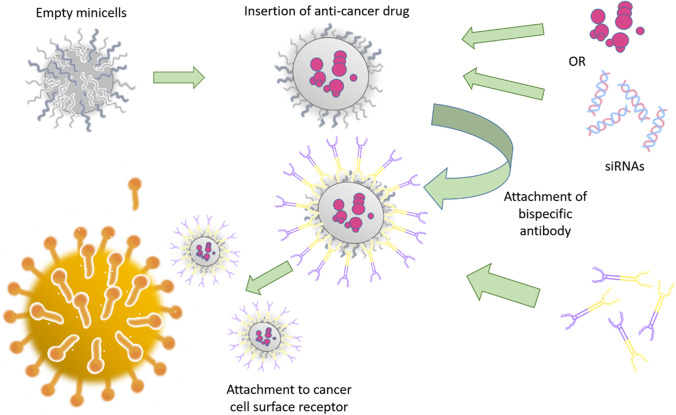
The packaging of drugs into minicells is reliant on the time of incubation and the concentration of drug in the solution (Fig. 7). The most common method used for packaging drugs and genetic material in minicells is co-incubation. Various studies showed that incubating drug or siRNA with minicells for a specific period leads to effective packaging of API into minicells (72, 80, 81). Moreover, electroporation was also explored for drug packaging via creating a pore in the cell membrane with the application of high voltage across the cell membrane. Sonication and Co-Extrusion methods are also significantly considered for drug packaging in minicells (82).
Fig. 7.
The packaging of chemotherapeutic drugs or siRNAs into empty minicells, targeting the tumour-cell surface receptors by using bispecific antibodies.
Clinical Trial
Despite various setbacks such as complex and limited production, side effects, stability, and mutation; several bacterial drug delivery systems for cancer treatment is under clinical trial. Firstly, Dr. William B. Coley in 1891 used Streptococcus pyogenes for treatment of cancer (83). Later, among several bacterial species, Listeria vaccine strains showed promising outcome used alone or in combination in human trial (84). The Vion Pharmaceutics designed S. typhimurium VNP20009 strain which was tested on 24 patients with metastatic melanoma in phase I trial. The maximum tolerated dose was noticed at 3.0 × 108 CFU/m2 after IV administration. No objective tumour regression was found, although several proinflammatory cytokines were increased (85). Another trial on four patients with metastatic melanoma treated with S. typhimurium VNP20009 also showed no objective tumour response (86). Therefore, VNP20009 was modified to express E. coli CD to enhance its therapeutic efficacy. The expressed E. coli CD has the potential to convert 5-FC to toxic 5-FU. Patients suffering from oesophageal adenocarcinoma and neck squamous carcinoma were treated with these bacteria via intratumoural injection and oral route with the frequency of three times a day for multiple cycles. The results showed bacterial colonisation at tumour was 3-folds higher compared to noncolonised patients. Moreover, no significant adverse effects were reported after 6 cycles of treatment. The discrepancies were also observed between clinical (human model) and preclinical (animal model) outcomes owing to different growth rates and structures of tumour that can alter the bacterial invasion, proliferation and clearance. The VNP20009 strain failed to colonise in tumour effectively, due to lack of lipid A function as discussed earlier (87).
On the other hand, application of Clostridium novyi-NT spores has entered Phase I clinical trial, where the study results revealed the promising therapeutic effect on tumour after intratumoural injection. The extensive tumour destruction was observed via the formation of gas pockets by C. novyi-NT spores. However, Clostridium cells was unable to eradicate whole cancerous cell which results in tumour relapse. Therefore, clinical trial on combination of C. novyi-NT strain with pembrolizumab was started to treat refractory advanced solid tumour patients (88). Similarly, bacterial minicell designed to deliver paclitaxel into cancer cells were evaluated for their safety for the first time in human phase I clinical trial on patients with advanced solid tumours. 5 weekly infusions were administered to patients. Dose-escalation design was used for the evaluation of seven dose levels and to determine the safety, tolerability and antitumour effects. Rigors and pyrexia were reported as common treatment-related adverse events; however, no death was reported during the treatment. Results revealed that bacterial minicells are safe to use with modest clinical efficacy (78). Other previous and ongoing clinical trial details are summarized in Table II.
Table II.
On going and Previous clinical trial details on bacterial strain alone or in combination for cancer treatment. This table is adopted and updated with permission from Duong et al. (89)
| Bacterial strain | Phase | Cancer type | Number of patients | References |
|---|---|---|---|---|
|
Salmonella typhimurium VNP20009 (attenuated Salmonella typhimurium) |
I | Metastatic melanoma; metastatic renal cell carcinoma | 25 | (85) |
|
S. typhimurium VNP20009 (Live genetically modified S. typhimurium (VNP 20009). |
I | Melanoma | 4 | (86) |
| S. typhimurium VNP20009 (attenuated Salmonella bacterium expressing the E. coli cytosine deaminase gene | I | Head and neck or esophageal adenocarcinoma | 3 | (90) |
| S. typhimurium VNP20009 (Live, Genetically Modified Salmonella Typhimurium (VNP20009) | I | Patients with advanced or metastatic solid tumours | Not provided |
(91) |
|
S. typhimurium VNP20009 (Live, Genetically Modified Salmonella Typhimurium) |
I | Unspecified adult solid tumours | Not provided |
(92) |
|
S. typhimurium VNP20009 (Live, Genetically Modified Salmonella Typhimurium) |
I | Neoplasm or neoplasm metastatic tumours | 45 |
(93) |
| S. typhimurium (IL-2 Expressing, Attenuated S. typhimurium) | I | Liver cancer | 22 |
(94) |
| S. typhimurium Ty21a VXM01(live attenuated S. typhi carrying an expression plasmid encoding VEGFR) | I | Pancreatic cancer | 26 | (95) |
| Clostridium Novyi-NT Spores | I | Colorectal cancer | 2 |
(96) |
| Clostridium Novyi-NT Spores | I | Solid tumour malignancies | 5 |
(97) |
| Clostridium novyi-NT | I | Solid tumour malignancies | 24 |
(98) |
| C. novyi-NT spores | Ib | Refractory advanced solid tumours | 18 |
(99) |
| Listeria monocytogenes | II | Metastatic pancreatic tumours | 90 | (100) |
| L. monocytogenes | II | Cervical cancer | 109 | (101) |
| L. monocytogenes | III | Cervical cancer | 450- |
(102) |
As discussed, these clinical data showed many key obstacles for clinical application, however, new combination strategies with bacterial drug delivery will improve better intratumoural bacterial colonisation with enhanced therapeutic output.
The Obstacle of Bacterial Therapy
Some of the challenges we may face by using bacteria as a therapeutic or delivery agent in cancer are the adverse effects of introducing an antigen into the human system, may it be live, attenuated or genetically engineered. The toxicity that follows a bacterial systemic infection after administration of live bacteria as carrier is a major cause for concern, especially in immunocompromised patients. This undesired biodistribution of bacterial in normal cells (off target distribution) rather than tumour colonisation can cause side effects and off-target release of anticancer drug.
The extent of toxins uptake dictates the success of cancer therapy into normal cells and its selectivity to tumour cells. Recent advances in biotechnology have suggested that non-pathogenic bacteria and their products deliver components that may result in the necrosis of tumour cells. There has been evidence of strains of viruses carrying altered genes and several bacterial species expressing therapeutic proteins, all of which selectively replicate in malignant cells. However, the effectiveness of microorganisms in the treatment of cancer is still lagging due to several factors as the human body sometimes manages to fight against and eliminate these microorganisms and their anti-tumour effects by producing neutralising antibodies (2).
Another key issue with bacterial drug delivery is the lack of proper understanding of the exact mechanism of preferential tumour colonisation of bacterial cells that showed innate anti-tumour effects. The insufficient colonisation of bacterial was observed in clinical studies that may denote robust immune reaction, which counteracting bacterial accumulation. Other attributes such as rapid clearance of bacterial from the blood before reaching to tumour site. Contrast results were observed in clinical and preclinical data on maintenance of bacteria in the bloodstream for longer duration. Preclinical data showed a high percentage of bacteria maintained in blood after administration for longer period. Other than that, the limited cytotoxic properties of bacteria requiring conjugation with chemotherapeutic agents as well as the risk of mutation due to genetic modification are also obstacles that need to be overcome to utilise bacteria as a potential therapy in cancer patients (5, 103, 104). On the other hand, BGs has its limitation as a delivery system. For example, weakly bound drugs to bacterial membrane, membrane protein, and cell wall can cause significant drug loss due to the large pores on the BG surface. Besides that, immunocompromised patients may not be a suitable candidate for the usage of BG, because BG can induce potent proinflammatory cytokine among immune cells Besides that, bacterial clearance, systemic infection, mutation and toxicity results in losing their therapeutic effects. Additionally, physiochemical and structural complexity of formulation is one of the factors for the slow pace of clinical transition of bacterial drug delivery system. It may require complex fabrication procedures which may have scale-up limitations in large-scale manufacturing (105, 106).
Conclusion
The resistance of cancer cells towards chemotherapy has led to various studies for alternative methods to deliver drugs into the tumour cells. Bacteria-mediated cancer therapy has shown promising benefit of being a controllable and targeted anti-cancer delivery system and is able to alleviate major problems associated with currently available therapy. Thus, the utilisation of bacterial spores, bacteria ghost and combination of bacteria with nanoparticles have been found to be beneficial as anti-cancer agents. However, despite numerous potential benefits offered by these methods, notable challenges are still observed in terms of its distribution as well as toxicity associated with the use of bacterial products in preclinical studies. Hence, future development of bacteria-mediated therapeutic delivery should not only focus on treating cancer but also protect healthy cells from the toxicity of the chemotherapy and the bacterial products/by-products. Therefore, more comprehensive future investigations are required to help overcome current challenges and improve presently available cancer treatments.
Supplementary Information
(DOCX 1166 kb)
Author Contributions
Conceptualization: MNP and HIR; data curation, writing—original draft preparation, PV, PNPL, JT, TJN, NZW, NXZ, CLE, NSNR, NDBK; revision and editing: MCIMA, BG, MC, JM, PAC; supervision, MNP, HIR. All authors have read and agreed to the published version of the manuscript.
Data Availability
Data is freely available.
Declarations
Conflict of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Manisha Pandey and Hira Choudhury contributed equally to this work.
References
- 1.Pucci C, Martinelli C, Ciofani G. Innovative approaches for cancer treatment: current perspectives and new challenges. Ecancermedicalscience [Internet]. 2019 [cited 2021 Nov 2];13:961. Available from: /pmc/articles/PMC6753017/. [DOI] [PMC free article] [PubMed]
- 2.Cao Z, Liu J. Bacteria and bacterial derivatives as drug carriers for cancer therapy. J Control Release. 2020;326:396–407. doi: 10.1016/j.jconrel.2020.07.009. [DOI] [PubMed] [Google Scholar]
- 3.Plummer M, de Martel C, Vignat J, Ferlay J, Bray F, Franceschi S. Global burden of cancers attributable to infections in 2012: a synthetic analysis. Lancet Glob Heal [Internet]. 2016 [cited 2021 Nov 2];4(9):e609–16. Available from: https://pubmed.ncbi.nlm.nih.gov/27470177/ [DOI] [PubMed]
- 4.Papadakis MA, McPhee SJ, editors. Current Medical Diagnosis & Treatment [Internet]. 40th ed. New York: McGraw Hill Medical; 2021 [cited 2021 Nov 2]. Available from: https://accessmedicine.mhmedical.com/content.aspx?bookid=2957§ionid=249359850
- 5.Nallar SC, Xu D-Q, Kalvakolanu DV. Bacteria and genetically modified bacteria as cancer therapeutics: current advances and challenges. Cytokine [Internet]. 2017 [cited 2021 Nov 3];89:160–72. Available from: https://pubmed.ncbi.nlm.nih.gov/26778055/ [DOI] [PubMed]
- 6.Liu G, Wang M, He H, Li J. Doxorubicin-loaded tumor-targeting peptide-decorated polypeptide nanoparticles for treating primary Orthotopic Colon Cancer. Front Pharmacol. 2021;12:2687. doi: 10.3389/fphar.2021.744811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gorain B, Choudhury H, Pandey M, Kesharwani P. Paclitaxel loaded vitamin E-TPGS nanoparticles for cancer therapy. Mater Sci Eng C [Internet]. 2018 Oct 1 [cited 2019 Sep 7];91:868–80. Available from: https://www.sciencedirect.com/science/article/pii/S0928493117335713 [DOI] [PubMed]
- 8.Choudhury H, Gorain B, Pandey M, Kumbhar SA, Tekade RK, Iyer AK, et al. Recent advances in TPGS-based nanoparticles of docetaxel for improved chemotherapy. Int J Pharm. 2017;529(1–2):506–522. Available from: 10.1016/j.ijpharm.2017.07.018 [DOI] [PubMed]
- 9.Kesharwani P, Choudhury H, Meher JG, Pandey M, Gorain B. Dendrimer-entrapped gold nanoparticles as promising nanocarriers for anticancer therapeutics and imaging. Prog Mater Sci. 2019;103:484–508. doi: 10.1016/j.pmatsci.2019.03.003. [DOI] [Google Scholar]
- 10.Rao W, Wang H, Han J, Zhao S, Dumbleton J, Agarwal P, et al. Chitosan-Decorated Doxorubicin-Encapsulated Nanoparticle Targets and Eliminates Tumor Reinitiating Cancer Stem-like Cells. ACS Nano [Internet]. 2015 [cited 2021 Nov 3];9(6):5725–40. Available from: https://pubmed.ncbi.nlm.nih.gov/26004286/ [DOI] [PubMed]
- 11.Awan BN, Fatima N, Riaz S, Malik S, Ahmad W. Bacterial and liposomal vector guided drug delivery system via tumor markers carrier gene to treat neoplasm. J App Pharm. 2015;8:6. [Google Scholar]
- 12.Feng X, He P, Zeng C, Li Y-H, Das SK, Li B, et al. Novel insights into the role of Clostridium novyi-NT related combination bacteriolytic therapy in solid tumors (Review). Oncol Lett [Internet]. 2021 [cited 2021 Nov 3];21(2):1–1. Available from: http://www.spandidos-publications.com/10.3892/ol.2020.12371/abstract [DOI] [PMC free article] [PubMed]
- 13.Staedtke V, Roberts NJ, Bai R-Y, Zhou S. Clostridium novyi-NT in cancer therapy. Genes Dis [Internet]. 2016 [cited 2021 Nov 3];3(2):152. Available from: /pmc/articles/PMC6150096/. [DOI] [PMC free article] [PubMed]
- 14.Sedighi M, Bialvaei AZ, Hamblin MR, Ohadi E, Asadi A, Halajzadeh M, et al. Therapeutic bacteria to combat cancer; current advances, challenges, and opportunities. Cancer Med [Internet]. 2019 [cited 2021 Nov 3];8(6):3181. Available from: /pmc/articles/PMC6558487/. [DOI] [PMC free article] [PubMed]
- 15.Łukasiewicz K, Fol M. Microorganisms in the treatment of Cancer: advantages and limitations. J Immunol Res. 2018;2018:1–8. doi: 10.1155/2018/2397808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Al-Hilu SA, Al-Shujairi WH. Dual role of Bacteria in carcinoma: stimulation and inhibition. Int J Microbiol. 2020;2020:1–15. doi: 10.1155/2020/4639761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gardlik R, Fruehauf JH. Bacterial vectors and delivery systems in cancer therapy. IDrugs [Internet]. 2010 Oct [cited 2019 Jan 10];13(10):701–6. Available from: http://www.ncbi.nlm.nih.gov/pubmed/20878592. [PubMed]
- 18.Mostaghaci B, Yasa O, Zhuang J, Sitti M. Bioadhesive bacterial microswimmers for targeted drug delivery in the urinary and gastrointestinal tracts. Adv Sci [Internet]. 2017 [cited 2021 Nov 3];4(6):170014. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/advs.201700058 [DOI] [PMC free article] [PubMed]
- 19.Mi Z, Feng ZC, Li C, Yang X, Ma MT, Rong PF. Salmonella-mediated cancer therapy: an innovative therapeutic strategy. J Cancer. 2019;10(20):4765–4776. doi: 10.7150/jca.32650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhu C, Ji Z, Ma J, Ding Z, Shen J, Wang Q. Recent Advances of Nanotechnology-Facilitated Bacteria-Based Drug and Gene Delivery Systems for Cancer Treatment. Pharm 2021, Vol 13, Page 940 [Internet]. 2021 Jun 24 [cited 2021 Dec 17];13(7):940. Available from: https://www.mdpi.com/1999-4923/13/7/940/htm [DOI] [PMC free article] [PubMed]
- 21.Kojima M, Zhang Z, Nakajima M, Ooe K, Fukuda T. Construction and evaluation of bacteria-driven liposome. Sensors Actuators B Chem. 2013;183:395–400. doi: 10.1016/j.snb.2013.03.127. [DOI] [Google Scholar]
- 22.Du NV, Han JW, Choi YJ, Cho S, Zheng S, Ko SY, et al. Active tumor-therapeutic liposomal bacteriobot combining a drug (paclitaxel)-encapsulated liposome with targeting bacteria (Salmonella typhimurium) Sensors Actuators B Chem. 2016;224:217–224. doi: 10.1016/j.snb.2015.09.034. [DOI] [Google Scholar]
- 23.Feng P, Cao Z, Wang X, Li J, Liu J. On-demand bacterial reactivation by restraining within a Triggerable Nanocoating. Adv Mater [Internet]. 2020 [cited 2021 Dec 17];32(34):2002406. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/adma.202002406 [DOI] [PubMed]
- 24.Sieow BFL, Wun KS, Yong WP, Hwang IY, Chang MW. Tweak to treat: reprograming Bacteria for Cancer treatment. Trends in Cancer. 2021;7(5):447–464. doi: 10.1016/j.trecan.2020.11.004. [DOI] [PubMed] [Google Scholar]
- 25.Chowdhury S, Castro S, Coker C, Hinchliffe TE, Arpaia N, Danino T. Programmable bacteria induce durable tumor regression and systemic antitumor immunity. Nat Med. 2019;25(7):1057. doi: 10.1038/s41591-019-0498-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ho CL, Tan HQ, Chua KJ, Kang A, Lim KH, Ling KL, et al. Engineered commensal microbes for diet-mediated colorectal-cancer chemoprevention. Nat Biomed Eng. 2018;2(1):27–37. doi: 10.1038/s41551-017-0181-y. [DOI] [PubMed] [Google Scholar]
- 27.Li R, Helbig L, Fu J, Bian X, Herrmann J, Baumann M, et al. Expressing cytotoxic compounds in Escherichia coli Nissle 1917 for tumor-targeting therapy. Res Microbiol. 2019;170(2):74–79. doi: 10.1016/j.resmic.2018.11.001. [DOI] [PubMed] [Google Scholar]
- 28.He L, Yang H, Liu F, Chen Y, Tang S, Ji W, et al. Escherichia coli Nissle 1917 engineered to express Tum-5 can restrain murine melanoma growth. Oncotarget. 2017;8(49):85772–85782. doi: 10.18632/oncotarget.20486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.He L, Yang H, Tang J, Liu Z, Chen Y, Lu B, et al. Intestinal probiotics E. coli Nissle 1917 as a targeted vehicle for delivery of p53 and Tum-5 to solid tumors for cancer therapy. J Biol Eng. 2019;13(1):58. Avaliable at: https://www.pubmed.ncbi.nlm.nih.gov/31297149/ [DOI] [PMC free article] [PubMed]
- 30.Gurbatri CR, Lia I, Vincent R, Coker C, Castro S, Treuting PM, et al. Engineered probiotics for local tumor delivery of checkpoint blockade nanobodies. Sci Transl Med. 2020;12(530):eaax0876. Available at: https://www.pubmed.ncbi.nlm.nih.gov/32051224/ [DOI] [PMC free article] [PubMed]
- 31.Leventhal DS, Sokolovska A, Li N, Plescia C, Kolodziej SA, Gallant CW, et al. Immunotherapy with engineered bacteria by targeting the STING pathway for anti-tumor immunity. Nat Commun. 2020;11(1):1–15. doi: 10.1038/s41467-020-16602-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yoon W, Park YC, Kim J, Chae YS, Byeon JH, Min SH, et al. Application of genetically engineered Salmonella typhimurium for interferon-gamma-induced therapy against melanoma. Eur J Cancer. 2017;70:48–61. doi: 10.1016/j.ejca.2016.10.010. [DOI] [PubMed] [Google Scholar]
- 33.Zheng JH, Nguyen VH, Jiang SN, Park SH, Tan W, Hong SH, et al. Two-step enhanced cancer immunotherapy with engineered Salmonella typhimurium secreting heterologous flagellin. Sci Transl Med. 2017;9(376):eaak9537. Available at: https://www.pubmed.ncbi.nlm.nih.gov/28179508/ [DOI] [PubMed]
- 34.Liang K, Liu Q, Li P, Han Y, Bian X, Tang Y, et al. Endostatin gene therapy delivered by attenuated Salmonella typhimurium in murine tumor models. Cancer Gene Ther. 2018;25(7–8):167–183. doi: 10.1038/s41417-018-0021-6. [DOI] [PubMed] [Google Scholar]
- 35.Din MO, Danino T, Prindle A, Skalak M, Selimkhanov J, Allen K, et al. Synchronized cycles of bacterial lysis for in vivo delivery. Nat. 2016;536(7614):81–85. doi: 10.1038/nature18930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lim D, Kim KS, Kim H-J, Ko K-C, Song JJ, Choi JH, et al. Anti-tumor activity of an immunotoxin (TGFα-PE38) delivered by attenuated Salmonella typhimurium. Oncotarget. 2017;8(23):37550–37560. doi: 10.18632/oncotarget.17197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chen T, Zhao X, Ren Y, Wang Y, Tang X, Tian P, et al. Triptolide modulates tumour-colonisation and anti-tumour effect of attenuated Salmonella encoding DNase I. Appl Microbiol Biotechnol. 2019;103(2):929–939. doi: 10.1007/s00253-018-9481-8. [DOI] [PubMed] [Google Scholar]
- 38.Kim K, Jeong JH, Lim D, Hong Y, Lim HJ, Kim GJ, et al. L-Asparaginase delivered by Salmonella typhimurium suppresses solid tumors. Mol Ther - Oncolytics. 2015;2:15007. doi: 10.1038/mto.2015.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Phan T, Nguyen VH, D’Alincourt MS, Manuel ER, Kaltcheva T, Tsai W, et al. Salmonella-mediated therapy targeting indoleamine 2, 3-dioxygenase 1 (IDO) activates innate immunity and mitigates colorectal cancer growth. Cancer Gene Ther. 2019;27(3):235–245. doi: 10.1158/1535-7163.MCT-18-0768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yoon W, Yoo Y, Chae YS, Kee SH, Kim BM. Therapeutic advantage of genetically engineered Salmonella typhimurium carrying short hairpin RNA against inhibin alpha subunit in cancer treatment. Ann Oncol. 2018;29(9):2010–2017. doi: 10.1093/annonc/mdy240. [DOI] [PubMed] [Google Scholar]
- 41.Zhao T, Wei T, Guo J, Wang Y, Shi X, Guo S, et al. PD-1-siRNA delivered by attenuated Salmonella enhances the antimelanoma effect of pimozide. Cell Death Dis. 2019;10(3):164. Available at: https://www.pubmed.ncbi.nlm.nih.gov/30778049/ [DOI] [PMC free article] [PubMed]
- 42.Zhao C, He J, Cheng H, Zhu Z, Xu H. Enhanced therapeutic effect of an antiangiogenesis peptide on lung cancer in vivo combined with salmonella VNP20009 carrying a Sox2 shRNA construct. J Exp Clin Cancer Res. 2016;35(1):107. Available at: https://www.pubmed.ncbi.nlm.nih.gov/27371094/ [DOI] [PMC free article] [PubMed]
- 43.Kitagawa K, Oda T, Saito H, Araki A, Gonoi R, Shigemura K, et al. Development of oral cancer vaccine using recombinant Bifidobacterium displaying Wilms’ tumor 1 protein. Cancer Immunol Immunother. 2017;66(6):787–798. doi: 10.1007/s00262-017-1984-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wei C, Xun AY, Wei XX, Yao J, Wang JY, Shi RY, et al. Bifidobacteria expressing Tumstatin protein for antitumor therapy in tumor-bearing mice. Technol Cancer Res Treat. 2016;15(3):498–508. doi: 10.1177/1533034615581977. [DOI] [PubMed] [Google Scholar]
- 45.Lv S, Wang X, Zhang N, Sun M, Qi W, Li Y, et al. Autophagy facilitates the development of resistance to the tumor necrosis factor superfamily member TRAIL in breast cancer. Int J Oncol [Internet]. 2015 [cited 2021 Nov 5];46(3):1286–94. Available from: http://www.spandidos-publications.com/10.3892/ijo.2014.2812/abstract [DOI] [PubMed]
- 46.James BR, Griffith TS. Tumor necrosis factor-related apoptosis-inducing ligand-induced apoptotic pathways in cancer immunosurveillance: molecular mechanisms and prospects for therapy. Res Reports Biochem [Internet]. 2014 [cited 2021 Nov 5];5:1–10. Available from: https://www.dovepress.com/tumor-necrosis-factor-related-apoptosis-inducing-ligand-induced-apopto-peer-reviewed-fulltext-article-RRBC
- 47.Setroikromo R, Zhang B, Reis CR, Mistry RH, Quax WJ. Death receptor 5 displayed on extracellular vesicles decreases TRAIL sensitivity of Colon Cancer cells. Front Cell Dev Biol. 2020;8:318. doi: 10.3389/fcell.2020.00318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Daguenet E, Khalifa J, Tolédano A, Borchiellini D, Pointreau Y, Rodriguez-Lafrasse C, et al. To exploit the 5 ‘R’ of radiobiology and unleash the 3 ‘E’ of immunoediting: ‘RE’-inventing the radiotherapy-immunotherapy combination: 10.1177/1758835920913445 [Internet]. 2020 [cited 2021 Nov 5];12. Available from: https://journals.sagepub.com/doi/full/10.1177/1758835920913445 [DOI] [PMC free article] [PubMed]
- 49.Stern C, Kasnitz N, Kocijancic D, Trittel S, Riese P, Guzman CA, et al. Induction of CD4(+) and CD8(+) anti-tumor effector T cell responses by bacteria mediated tumor therapy. Int J cancer [Internet]. 2015 [cited 2021 Nov 5];137(8):2019–28. Available from: https://pubmed.ncbi.nlm.nih.gov/25868911/ [DOI] [PubMed]
- 50.Binder DC, Arina A, Wen F, Tu T, Zhao M, Hoffman RM, et al. Tumor relapse prevented by combining adoptive T cell therapy with Salmonella typhimurium. Oncoimmunology [Internet]. 2016 Jun 2 [cited 2021 Nov 5];5(6):e1130207. Available from: https://pubmed.ncbi.nlm.nih.gov/27471609/ [DOI] [PMC free article] [PubMed]
- 51.Wrande M, Andrews-Polymenis H, Twedt DJ, Steele-Mortimer O, Porwollik S, McClelland M, et al. Genetic Determinants of Salmonella enterica Serovar Typhimurium Proliferation in the Cytosol of Epithelial Cells. Infect Immun [Internet]. 2016 [cited 2021 Nov 5];84(12):3517–26. Available from: https://pubmed.ncbi.nlm.nih.gov/27698022/ [DOI] [PMC free article] [PubMed]
- 52.Sharrock AV, McManaway SP, Rich MH, Mumm JS, Hermans IF, Tercel M, et al. Engineering the Escherichia coli Nitroreductase NfsA to create a flexible enzyme-prodrug activation system. Front Pharmacol. 2021;12:1420. doi: 10.3389/fphar.2021.701456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kubiak AM, Minton NP. The potential of clostridial spores as therapeutic delivery vehicles in tumour therapy. Res Microbiol. 2015;166(4):244–254. doi: 10.1016/j.resmic.2014.12.006. [DOI] [PubMed] [Google Scholar]
- 54.Gao S, Jung J-H, Lin S-M, Jang A-Y, Zhi Y, Bum Ahn K, et al. Development of Oxytolerant Salmonella typhimurium Using Radiation Mutation Technology (RMT) for Cancer Therapy. Sci Reports 2020 101 [Internet]. 2020 [cited 2021 Nov 5];10(1):1–12. Available from: https://www.nature.com/articles/s41598-020-60396-6 [DOI] [PMC free article] [PubMed]
- 55.Chien T, Doshi A, Danino T. Advances in bacterial cancer therapies using synthetic biology. Curr Opin Syst Biol [Internet]. 2017 Oct [cited 2021 Nov 3];5:1–8. Available from: https://pubmed.ncbi.nlm.nih.gov/29881788/ [DOI] [PMC free article] [PubMed]
- 56.Sabzehali F, Azimi H, Goudarzi M. Bacteria as a vehicle in cancer therapy and drug delivery. Arch Adv Biosci [Internet]. 2017 [cited 2021 Nov 5];8(1):52–9. Available from: https://journals.sbmu.ac.ir/aab/article/view/13574
- 57.Raman V, Van Dessel N, Hall CL, Wetherby VE, Whitney SA, Kolewe EL, et al. Intracellular delivery of protein drugs with an autonomously lysing bacterial system reduces tumor growth and metastases. Nat Commun 2021 121 [Internet]. 2021 [cited 2022 Mar 11];12(1):1–14. Available from: https://www.nature.com/articles/s41467-021-26367-9 [DOI] [PMC free article] [PubMed]
- 58.Geng Z, Cao Z, Liu R, Liu K, Liu J, Tan W. Aptamer-assisted tumor localization of bacteria for enhanced biotherapy. Nat Commun 2021 121 [Internet]. 2021 [cited 2021 Dec 17];12(1):1–12. Available from: https://www.nature.com/articles/s41467-021-26956-8 [DOI] [PMC free article] [PubMed]
- 59.Wang L, Cao Z, Zhang M, Lin S, Liu J. Spatiotemporally controllable distribution of combination therapeutics in solid tumors by dually modified Bacteria. Adv Mater [Internet]. 2021 [cited 2021 Dec 17];2106669. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/adma.202106669 [DOI] [PubMed]
- 60.Hajam IA, Dar PA, Won G, Lee JH. Bacterial ghosts as adjuvants: mechanisms and potential. Vet Res 2017 481 [Internet]. 2017 [cited 2021 Nov 5];48(1):1–13. Available from: https://veterinaryresearch.biomedcentral.com/articles/10.1186/s13567-017-0442-5 [DOI] [PMC free article] [PubMed]
- 61.Haidinger W, Mayr UB, Szostak MP, Resch S, Lubitz W. Escherichia coli ghost production by expression of lysis gene E and staphylococcal nuclease. Appl Environ Microbiol [Internet]. 2003 [cited 2021 Nov 5];69(10):6113. Available from: /pmc/articles/PMC201253/. [DOI] [PMC free article] [PubMed]
- 62.Yu S, Peng W, Si W, Yin L, Liu S, Liu H, et al. Enhancement of bacteriolysis of Shuffled phage PhiX174 gene E. Virol J 2011 81 [Internet]. 2011 [cited 2021 Nov 5];8(1):1–6. Available from: https://virologyj.biomedcentral.com/articles/10.1186/1743-422X-8-206 [DOI] [PMC free article] [PubMed]
- 63.Schön P, Schrot G, Wanner G, Lubitz W, Witte A. Two-stage model for integration of the lysis protein E of ΦX174 into the cell envelope of Escherichia coli. FEMS Microbiol Rev [Internet]. 1995 [cited 2021 Nov 5];17(1–2):207–12. Available from: https://academic.oup.com/femsre/article/17/1-2/207/633535 [DOI] [PubMed]
- 64.Kudela P, Kollera VJ, Lubitz W. Bacterial ghosts (BGs)--advanced antigen and drug delivery system. Vaccine [Internet]. 2010 [cited 2021 Nov 5];28(36):5760–7. Available from: https://pubmed.ncbi.nlm.nih.gov/20619379/ [DOI] [PubMed]
- 65.Mayr UB, Walcher P, Azimpour C, Riedmann E, Haller C, Lubitz W. Bacterial ghosts as antigen delivery vehicles. Adv Drug Deliv Rev [Internet]. 2005 [cited 2021 Nov 5];57:1381–91. Available from: www.elsevier.com/locate/addr [DOI] [PubMed]
- 66.Kudela P, Paukner S, Mayr UB, Cholujova D, Schwarczova Z, Sedlak J, et al. Bacterial ghosts as novel efficient targeting vehicles for DNA delivery to the human monocyte-derived dendritic cells. J Immunother [Internet]. 2005 [cited 2021 Nov 5];28(2):136–43. Available from: https://pubmed.ncbi.nlm.nih.gov/15725957/ [DOI] [PubMed]
- 67.Ebensen T, Paukner S, Link C, Kudela P, de Domenico C, Lubitz W, et al. Bacterial ghosts are an efficient delivery system for DNA vaccines. J Immunol [Internet]. 2004 [cited 2021 Nov 5];172(11):6858–65. Available from: https://pubmed.ncbi.nlm.nih.gov/15153504/ [DOI] [PubMed]
- 68.Eko FO, Talin BA, Lubitz W. Development of a Chlamydia trachomatis bacterial ghost vaccine to fight human blindness. Hum Vaccin [Internet]. 2008 [cited 2021 Nov 5];4(3):176–83. Available from: https://pubmed.ncbi.nlm.nih.gov/18382139/ [DOI] [PubMed]
- 69.Kudela P, Paukner S, Mayr UB, Cholujova D, Kohl G, Schwarczova Z, et al. Effective gene transfer to melanoma cells using bacterial ghosts. Cancer Lett [Internet]. 2008 [cited 2021 Nov 5];262(1):54–63. Available from: https://pubmed.ncbi.nlm.nih.gov/18164809/ [DOI] [PubMed]
- 70.Paukner S, Kohl G, Lubitz W. Bacterial ghosts as novel advanced drug delivery systems: antiproliferative activity of loaded doxorubicin in human Caco-2 cells. J Control Release [Internet]. 2004 [cited 2021 Nov 6];94(1):63–74. Available from: https://pubmed.ncbi.nlm.nih.gov/14684272/ [DOI] [PubMed]
- 71.Groza D, Gehrig S, Kudela P, Holcmann M, Pirker C, Dinhof C, et al. Bacterial ghosts as adjuvant to oxaliplatin chemotherapy in colorectal carcinomatosis. Oncoimmunology [Internet]. 2018 [cited 2021 Nov 5];7(5):e1424676. Available from: https://pubmed.ncbi.nlm.nih.gov/29721389/ [DOI] [PMC free article] [PubMed]
- 72.Zhang Y, Ji W, He L, Chen Y, Ding X, Sun Y, et al. E. coli Nissle 1917-Derived Minicells for Targeted Delivery of Chemotherapeutic Drug to Hypoxic Regions for Cancer Therapy. Theranostics [Internet]. 2018 [cited 2021 Nov 6];8(6):1690–705. Available from: https://pubmed.ncbi.nlm.nih.gov/29556350/ [DOI] [PMC free article] [PubMed]
- 73.Nguyen HN, Romero Jovel S, Nguyen THK. Nanosized Minicells Generated by Lactic Acid Bacteria for Drug Delivery. J Nanomater. 2017;2017:6847297. doi: 10.1155/2017/6847297. [DOI] [Google Scholar]
- 74.S Song, Vuai MS, Zhong M. The role of bacteria in cancer therapy - enemies in the past, but allies at present. Infect Agent Cancer [Internet]. 2018 Mar 15 [cited 2021 Nov 6];13(1):9–18. Available from: https://pubmed.ncbi.nlm.nih.gov/29568324/ [DOI] [PMC free article] [PubMed]
- 75.Rang CU, Proenca A, Buetz C, Shi C, Chao L. Minicells as a Damage Disposal Mechanism in Escherichia coli. mSphere [Internet]. 2018 [cited 2021 Nov 6];3(5):1–9. Available from: https://journals.asm.org/doi/abs/10.1128/mSphere.00428-18 [DOI] [PMC free article] [PubMed]
- 76.Farley MM, Hu B, Margolin W, Liu J. Minicells, Back in Fashion. J Bacteriol [Internet]. 2016 [cited 2021 Nov 6];198(8):1186–95. Available from: https://pubmed.ncbi.nlm.nih.gov/26833418/ [DOI] [PMC free article] [PubMed]
- 77.MacDiarmid JA, Amaro-Mugridge NB, Madrid-Weiss J, Sedliarou I, Wetzel S, Kochar K, et al. Sequential treatment of drug-resistant tumors with targeted minicells containing siRNA or a cytotoxic drug. Nat Biotechnol [Internet]. 2009 [cited 2021 Nov 6];27(7):643–51. Available from: https://www.nature.com/articles/nbt.1547 [DOI] [PubMed]
- 78.Solomon BJ, Desai J, Rosenthal M, McArthur GA, Pattison ST, Pattison SL, et al. A first-time-in-human phase I clinical trial of bispecific antibody-targeted, paclitaxel-packaged bacterial Minicells. PLoS One [Internet]. 2015 [cited 2021 Nov 6];10(12):e0144559. Available from: https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0144559 [DOI] [PMC free article] [PubMed]
- 79.MacDiarmid JA, Langova V, Bailey D, Pattison ST, Pattison SL, Christensen N, et al. Targeted doxorubicin delivery to brain tumors via Minicells: proof of principle using dogs with spontaneously occurring tumors as a model. PLoS One [Internet] 2016 [cited 2021 Nov 6];11(4):e0151832. Available from: https://pubmed.ncbi.nlm.nih.gov/27050167/ [DOI] [PMC free article] [PubMed]
- 80.MacDiarmid JA, Mugridge NB, Weiss JC, Phillips L, Burn AL, Paulin RPP, et al. Bacterially derived 400 nm particles for encapsulation and Cancer cell targeting of chemotherapeutics. Cancer Cell. 2007;11(5):431–445. doi: 10.1016/j.ccr.2007.03.012. [DOI] [PubMed] [Google Scholar]
- 81.Brahmbhatt H, MacDiarmid J. Bacterially derived intact minicells that encompass plasmid free functional nucleic acid for in vivo delivery to mammalian cells. Vol. 433. 2010.
- 82.Fazal S, Lee R. Biomimetic bacterial membrane vesicles for drug delivery applications. Pharm 2021, Vol 13, Page 1430 [Internet]. 2021 [cited 2022 Feb 10];13(9):1430. Available from: https://www.mdpi.com/1999-4923/13/9/1430/htm [DOI] [PMC free article] [PubMed]
- 83.Coley WB. Contribution to the Knowledge of Sarcoma. Ann Surg. 1891 [cited 2021 Dec 16];14(3):199–220. Available from: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1428624/ [DOI] [PMC free article] [PubMed]
- 84.Miles B, Safran HP, Monk BJ. Therapeutic options for treatment of human papillomavirus-associated cancers - novel immunologic vaccines: ADXS11–001. Gynecol Oncol Res Pract [Internet]. 2017 [cited 2021 Dec 16];4(1):10. Available from: https://pubmed.ncbi.nlm.nih.gov/28725449/ [DOI] [PMC free article] [PubMed]
- 85.Toso JF, Gill VJ, Hwu P, Marincola FM, Restifo NP, Schwartzentruber DJ, et al. Phase I study of the intravenous administration of attenuated Salmonella typhimurium to patients with metastatic melanoma. J Clin Oncol [Internet]. 2002 [cited 2021 Dec 16];20(1):142–52. Available from: https://pubmed.ncbi.nlm.nih.gov/11773163/ [DOI] [PMC free article] [PubMed]
- 86.Heimann DM, Rosenberg SA. Continuous intravenous administration of live genetically modified salmonella typhimurium in patients with metastatic melanoma. J Immunother [Internet]. 2003 [cited 2021 Dec 16];26(2):179–80. Available from: https://pubmed.ncbi.nlm.nih.gov/12616110/ [DOI] [PMC free article] [PubMed]
- 87.Cunningham C, Nemunaitis J. A phase I trial of genetically modified Salmonella typhimurium expressing cytosine deaminase (TAPET-CD, VNP20029) administered by intratumoral injection in combination with 5-fluorocytosine for patients with advanced or metastatic cancer. Protocol no: CL-. Hum Gene Ther [Internet]. 2001 [cited 2021 Dec 16];12(12):1594–6. Available from: https://pubmed.ncbi.nlm.nih.gov/11529249/ [PubMed]
- 88.Roberts NJ, Zhang L, Janku F, Collins A, Bai RY, Staedtke V, et al. Intratumoral injection of Clostridium novyi-NT spores induces antitumor responses. Sci Transl Med [Internet]. 2014 [cited 2021 Dec 16];6(249):249. Available from: https://pubmed.ncbi.nlm.nih.gov/25122639/ [DOI] [PMC free article] [PubMed]
- 89.Duong MTQ, Qin Y, You SH, Min JJ. Bacteria-cancer interactions: bacteria-based cancer therapy. Exp Mol Med 2019 5112 [Internet]. 2019 [cited 2021 Dec 16];51(12):1–15. Available from: https://www.nature.com/articles/s12276-019-0297-0 [DOI] [PMC free article] [PubMed]
- 90.Nemunaitis J, Cunningham C, Senzer N, Kuhn J, Cramm J, Litz C, et al. Pilot trial of genetically modified, attenuated Salmonella expressing the E. coli cytosine deaminase gene in refractory cancer patients. Cancer Gene Ther. 2003;10(10):737–744. doi: 10.1038/sj.cgt.7700634. [DOI] [PubMed] [Google Scholar]
- 91.VNP20009 in Treating Patients With Advanced or Metastatic Solid Tumors That Have Not Responded to Previous Therapy - Full Text View - ClinicalTrials.gov.
- 92.VNP20009 in Treating Patients With Advanced Solid Tumors - Tabular View - ClinicalTrials.gov.
- 93.Treatment of Patients With Cancer With Genetically Modified Salmonella Typhimurium Bacteria - Tabular View - ClinicalTrials.gov.
- 94.IL-2 Expressing, Attenuated Salmonella Typhimurium in Unresectable Hepatic Spread - Tabular View - ClinicalTrials.gov.
- 95.Schmitz-Winnenthal FH, Hohmann N, Schmidt T, Podola L, Friedrich T, Lubenau H, et al. A phase 1 trial extension to assess immunologic efficacy and safety of prime-boost vaccination with VXM01, an oral T cell vaccine against VEGFR2, in patients with advanced pancreatic cancer. Oncoimmunology. 2018;7(4). [DOI] [PMC free article] [PubMed]
- 96.One Time Injection of Bacteria to Treat Solid Tumors That Have Not Responded to Standard Therapy - Tabular View - ClinicalTrials.gov.
- 97.Safety Study of Clostridium Novyi-NT Spores to Treat Patients With Solid Tumors That Have Not Responded to Standard Therapies - Tabular View - ClinicalTrials.gov.
- 98.Safety Study of Intratumoral Injection of Clostridium Novyi-NT Spores to Treat Patients With Solid Tumors That Have Not Responded to Standard Therapies - Tabular View - ClinicalTrials.gov.
- 99.Pembrolizumab With Intratumoral Injection of Clostridium Novyi-NT - Full Text View - ClinicalTrials.gov.
- 100.Le DT, Wang-Gillam A, Picozzi V, Greten TF, Crocenzi T, Springett G, et al. Safety and survival with GVAX pancreas prime and Listeria monocytogenes-expressing mesothelin (CRS-207) boost vaccines for metastatic pancreatic cancer. J Clin Oncol. 2015;33(12):1325–1333. doi: 10.1200/JCO.2014.57.4244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Basu P, Mehta A, Jain M, Gupta S, Nagarkar RV, John S, et al. A randomized phase 2 study of ADXS11-001 Listeria monocytogenes-Listeriolysin O immunotherapy with or without cisplatin in treatment of advanced cervical Cancer. Int J Gynecol Cancer. 2018;28(4):764–772. doi: 10.1097/IGC.0000000000001235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Study of ADXS11–001 in Subjects With High Risk Locally Advanced Cervical Cancer - Tabular View - ClinicalTrials.gov.
- 103.Shende P, Basarkar V. Recent trends and advances in microbe-based drug delivery systems. DARU J Pharm Sci [Internet] 2019 [cited 2021 Dec 15];27(2):809. Available from: https://www.pmc/articles/PMC6895346/. [DOI] [PMC free article] [PubMed]
- 104.Badie F, Ghandali M, Tabatabaei SA, Safari M, Khorshidi A, Shayestehpour M, et al. Use of Salmonella Bacteria in Cancer therapy: direct, drug delivery and combination approaches. Front Oncol. 2021;11:142. doi: 10.3389/fonc.2021.624759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Ganeshpurkar A, Ganeshpurkar A, Pandey V, Agnihotri A, Bansal D, Dubey N. Harnessing the potential of bacterial ghost for the effective delivery of drugs and biotherapeutics. Int J Pharm Investig [Internet]. 2014 [cited 2021 Dec 15];4(1):10. Available from: https://www.pmc/articles/PMC3944611/. [DOI] [PMC free article] [PubMed]
- 106.Chen H, Ji H, Kong X, Lei P, Yang Q, Wu W, et al. Bacterial ghosts-based vaccine and drug delivery systems. Pharm 2021, Vol 13, Page 1892 [Internet]. 2021 [cited 2021 Dec 15];13(11):1892. Available from: https://www.mdpi.com/1999-4923/13/11/1892/htm [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX 1166 kb)
Data Availability Statement
Data is freely available.