Abstract
Introduction
Maintenance dialysis patients (MDP) are at higher risk of exposure with increased mortality from COVID‐19 with generalized immunization becoming the cornerstone in prevention. This study aims to compare humoral response between hemodialysis (HD) and peritoneal dialysis (PD) patients.
Materials and Methods
Observational prospective study following HD and PD programs from a Portuguese Center receiving BNT162b2 vaccine. Specific anti‐Spike IgG quantification to compare both for absolute value and non‐responders (NR) between modalities and against risk factors.
Results
Of 67 MDP, 42 were HD and 25 PD patients. PD developed higher antibody titers after both first (median 5.44 vs. 0.99 AU/ml, p < 0.01) and second dose (median 170.43 vs. 65.81 AU/ml; p < 0.01). HD associated with NR after the first dose (p < 0.01).
Conclusion
This study demonstrated improved humoral immunogenicity with BNT162b2 in PD compared to HD patients. These differences are attributed to comorbidity burden and age differences, rather than dialysis modality.
Keywords: BNT162b2 (Pfizer BionNTech), Charlson comorbidity index, COVID‐19, hemodialysis, peritoneal dialysis, vaccination
1. INTRODUCTION
Worldwide vaccination against coronavirus 2019 (COVID‐19) has become the cornerstone in prevention of Severe Acute Respiratory Syndrome Coronavirus 2 (Sars‐CoV‐2) associated with this pandemic. Several vaccines with different acting mechanisms have been developed, namely BNT162b2 (Pfizer BioNTech) [1] and mRNA‐1273 (Moderna) [2]—mRNA‐based vaccines; ChAdOx1 nCov‐19 (AstraZeneca) [3] and Ad26.COV2.S (Johnson & Johnson/Janssen) [4]—recombinant adenovirus vectors encoding Sars‐CoV‐2 Spike glycoprotein, among others.
The need for mandatory regular contact with health care services in maintenance dialysis patients (MDP), coupled with worse disease severity and increased mortality [5, 6, 7], establish MDP as a high‐risk population. Following this assessment and international recommendations, the Portuguese government implemented vaccination for MDP early in the immunization plan, starting on February 2021. A known challenge in end‐stage kidney disease (ESKD) then reemerged: impaired immune system function translating in low immunogenicity from vaccination [8]. Influenza, pneumococcal and Hepatitis B immunization have systematically shown some level of weakened response compared to the general population [9, 10, 11, 12, 13]. New protocols involving follow‐up antibody measurements, adjuvant improvement, and repeated inoculations have minimized this problem, particularly well documented for Hepatitis B [14, 15].
Immunodeficiency in ESKD is multifactorial and affects both the innate and adaptative system [16]. Uremic toxins, malnutrition, chronic inflammation and dialysis technique contribute to this deficiency [17]. Cellularly, all immune cells show a disturbance in ESKD, both quantitative and qualitative, starting early as impaired response to stimuli by monocytes and lymphocytes [17, 18, 19, 20]. Specific humoral response requires antigen presenting dendritic cells which are both reduced in number and dysfunctional in ESKD and is now proposed as one of the main mechanisms of immunodeficiency in this population [21, 22, 23, 24]. Additionally, antigen‐specific memory CD4 T‐cell, responsible for lasting immunity, is also functionally defective [25, 26, 27]. On a molecular level, dysregulation of toll‐like receptors and upregulation of inflammatory cytokines contribute to immune system stunning [28, 29].
Dialysis modality also affects the immune system differently. Peritoneal dialysis specific factors for immune dysfunction include: (a) intra‐abdominal catheter; (b) high glucose/glucose degradation products or endotoxins on dialysate; (c) constitutive complement activation; (d) repetitive peritonitis and exit‐site infection; (e) protein loss, including immunoglobulin in spent dialysate (controversial) [30, 31]. For the HD population: (a) central venous catheter as vascular access; (b) use of conventional hemodialysis over hemodiafiltration; (c) use of bioincompatible dialysis membranes; (d) complement activation during session secondary to loss of inhibitory molecules [32, 33, 34]. Immune system dysfunction in ESKD is still a matter of active investigation and, at this moment, is not clear if any modality is superior to the other.
Specifically, for SARS‐CoV‐2 vaccination, efficacy studies described above did not include MDP. Humoral response to BNT162b2 in HD patients has been a target in multiple studies [35, 36, 37]. Data on PD, however, is still lacking, and differences between modalities have not been studied. This study aims to describe and compare the initial humoral response to the BNT162b2 between a group of PD and HD patients in a nephrology unit.
2. MATERIALS AND METHODS
The study design consisted of an observational prospective study. It included a group of 46 HD and 27 PD patients who were scheduled to receive two doses of BNT162b2 in a 3‐week interval, in accordance with pharmaceutical guidelines for administration, between February and March 2021.
The assessment of humoral vaccination response was done as part of the internal policy of the center's contingency protocol and informed consent was obtained from each participant, regarding the use and access to these analytical results, as well as to the remaining clinical and demographic information for scientific research. Blood collection and analysis was made at two distinct phases: (1) 3 weeks after the administration of the first dose and (2) 3 weeks after the administration of the second dose. Because the recommended dosing interval is 3 weeks, the first collection was coincidentally performed with the second dose administration.
Humoral response, measured as IgG anti‐Spike for SARS‐CoV‐2, in addition to IgM anti‐Spike and anti‐Nucleocapside for tracking possible contacts, even if asymptomatic. Titers were measured as arbitrary units per milliliter (AU/ml) using the MAGLUMI® SARS‐CoV‐2 S‐RBD IgG chemiluminescence kit. Response was considered significant for titers superior to 1 AU/ml, in accordance with manufacturer's indications. Inclusion criteria included capacity to understand and provide informed consent and no significant increase in specific IgM anti‐N for SARS‐CoV‐2 during follow‐up.
Primary end point was established as comparison between the median of achieved titers to both the first and second dose and rate of non‐responders (NR) for each modality. Secondary end points focused on comparison of clinical and demographic data including age, sex, Charlson comorbidity index (CCI) and dialysis vintage. Further subgroup exploratory analysis was performed based on age (under 70) and factors for weak response (IgG anti‐Spike below percentile 25 for each modality) to vaccination.
2.1. Statistical analysis
Statistical analysis was carried out using Microsoft Excel 2016 and IBM SPSS Statistics 25 software.
Study variables, divided in demographic/clinical and humoral, are summarized in Table 1. Descriptive analysis was performed using frequencies and percentages for categorical variables, whereas continuous variables are presented using means with standard deviations, if normal distributed, or medians with interquartile range, for skewed distribution.
TABLE 1.
Humoral and demographic/clinical variables in the study
| Continuous | Categorical |
|---|---|
| Humoral response associated variables | |
| IgG anti‐Spike S1 levels 1st dose (AU/ml) |
1st dose NR (<1 AU/ml) 2nd dose NR (<1 AU/ml) |
| IgG anti‐Spike S1 levels 2nd dose (AU/ml) | |
| Demographic/clinical variables | |
| Age | Sex |
| CCI | Diabetes |
| Dialysis vintage | |
Abbreviations: AU/ml: arbitrary units per milliliter; CCI: Charlson comorbidity index; NR, non‐responders.
Demographic and clinical data, anti‐Spike IgG levels and NR rate were compared between both modality groups. Differences in continuous variables between groups were analyzed with Student's t test for means (parametric) and Mann–Whitney U test for medians (non‐parametric). Response rate was analyzed between modalities using Fisher's exact test and Phi correlation coefficient for association strength. Subgroup analysis using the same tests and Spearman's correlation test between antibody levels and continuous variables were also performed in an exploratory basis to further understand the primary outcome results. A binary logistic regression was performed using age, CCI and dialysis vintage to predict the likelihood weak response across the entire sample.
3. RESULTS
Of the 46 HD patients enrolled, 42 were eligible for the study: (1) two patients discontinued dialysis before the administration of the second dose; (2) one patient died from an unrelated cause, and (3) one patient, despite asymptomatic, showed a significant increase in IgM titer raising the possibility of contact with the virus. Similarly, two patients from the PD group showed significant increase in specific IgM and were considered to have asymptomatic infection, resulting in 25 PD patients being enrolled into the statistical analysis, a total of 67 MDP.
Demographic and clinical data is summarized in Table 2, both for descriptive and comparative analyses. PD group was younger (60.5 vs. 75.1 years; t [65] = 5.1; p < 0.01), with shorter dialysis vintage (23.9 vs. 41.1 months; p = 0.02) and lower CCI (5.2 vs. 7.8, p < 0.01). There was no statistical difference for gender or diabetes between both groups. Humoral response data is summarized in Table 3 with boxplot graph representation in Figures 1 and 2. Overall, IgG titers were higher in PD patients both for the first (5.44 vs. 0.99; U = 844; p < 0.01) and second doses (170.43 vs. 65.81; U = 766; p < 0.01). HD was weakly associated with NR for the first dose (Phi 0.383; p < 0.01) and showed no association with the second dose (p = 0.08) when compared to PD. However, after complete vaccination schedule, there were no NR patients in the PD group, an outcome that was observed in six patients (14%) in the HD group.
TABLE 2.
Description and comparison of clinical and demographic data by modality
| HD | PD | Comparison p value | |
|---|---|---|---|
| Age, years, mean (σ) | 75.1 (11.7) | 60.5 (10.7) | <0.001 (t test) |
| Sex, female, n (%) | 17 (40.5) | 7 (28) | 0.43 (Fish) |
| Diabetes, n (%) | 19 (45.2) | 7 (28) | 0.2 (Fish) |
| Dialysis vintage, mo, Med (IQR) | 35 (44.8) | 18 (19.5) | 0.02 (MW‐U) |
| CCI, mean (σ) | 7.8 (2.4) | 5.2 (2.4) | <0.001 (MW‐U) |
Abbreviations: CCI, Charlson comorbidity index; Fish, Fisher's exact test; HD, hemodialysis; IQR, interquartile range; mo, months; Med, median; MW‐U, Mann–Whitney U test; PD, peritoneal dialysis; t test, Student's t test.
TABLE 3.
Humoral response titters and rate of non‐responders by modality
| HD | PD | Comparison p‐value | |
|---|---|---|---|
| First dose | |||
| IgG anti‐Spike | |||
| Med (IQR) | 0.99(3.16) | 5.44 (13.45) | <0.01 (MW‐U) |
| Min/Max | 0.1/49.1 | 0.6/68.53 | |
| IgM | |||
| Med (IQR) | 0.62 (0.15) | 0.66 (0.15) | |
| Non‐responders | |||
| N (%) | 21 (50%) | 3 (12%) | <0.01 (Fish) |
| OR (95% CI) | 7.35 (1.9–28.57) | ||
| Second dose | |||
| IgG anti‐Spike | |||
| Med (IQR) | 65.81 (120.56) | 170.43 (199.06) | <0.01 (MW‐U) |
| Min/Max | 0.1/411.6 | 16.04/912 | |
| IgM | |||
| Med (IQR) | 0.53 (0.22) | 0.62 (0.24) | |
| Non‐responders | |||
| N (%) | 6 (14%) | 0 (0) | 0.08 (Fish) |
| OR (95% CI) | N/A | ||
| Sub‐analysis | |||
| Under 70 years old | |||
| N | 14 | 22 | |
| Age, mean (σ) | 61.9 (8.7) | 57.8 (8.1) | 0.12 (t test) |
| CCI, mean (σ) | 6.1 (2.2) | 5.0 (1.9) | 0.22 (t test) |
| IgG, Med (IQR) | 89.9 (189.9) | 173.3 (167.4) | 0.08 (MW‐U) |
Note: Humoral titters are expressed in arbitrary units per milliliter (AU/ml).
Abbreviations: Fish, Fisher's exact test; HD, hemodialysis; IQR, interquartile range; Med, median; Min, minimum value; Max, maximum value; MW‐U, Mann–Whitney U test; OR, odds ratio; PD, peritoneal dialysis; St t: Student's t test.
FIGURE 1.
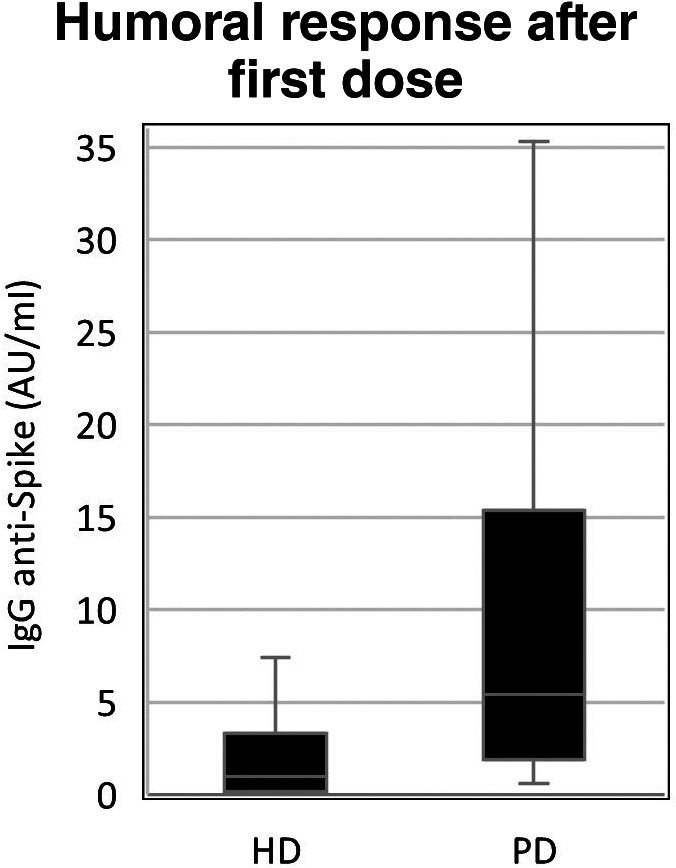
Boxplot graph for first dose humoral response between modalities. AU/ml, arbitrary units per milliliter; HD, hemodialysis group; PD, peritoneal dialysis group
FIGURE 2.
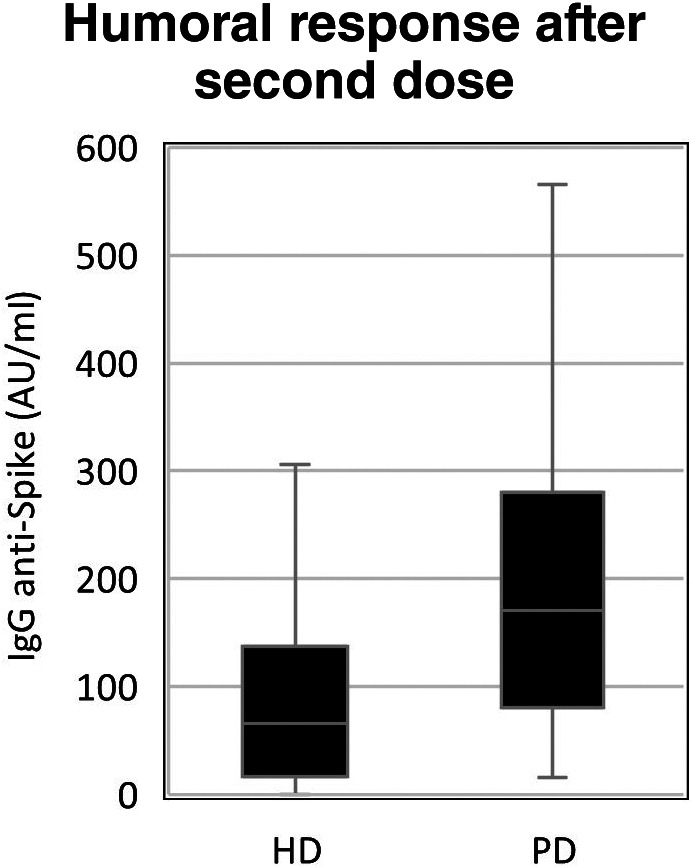
Boxplot graph for second dose humoral response between modalities. AU/ml: arbitrary units per milliliter; HD, hemodialysis group; PD, peritoneal dialysis group
Additional exploratory analysis was performed to better clarify the influence of these factors in humoral response: (1) correlation analysis between CCI, age and achieved humoral titers after both doses was performed using Spearman's test. CCI correlated moderately (ρ = −0.62, p < 0.001), and age weakly (ρ = − 0.38, p = 0.001) with humoral titers after both doses. Separately, however, this correlation was still verified in the HD subgroup (CCI: ρ = − 0.66, p < 0.001; age: ρ = − 0.3, p = 0.05), but not for PD (CCI: p = 0.4; age: p = 0.49). (2) A sub‐analysis was performed for the same outcomes while restricting investigation to patients under 70 years of age. Differently from the global analysis, age and CCI means were not statistically different between subgroups (p = 0.12 and p = 0.22, respectively), while dialysis vintage remained shorter in the PD subgroup (21.6 vs. 45.5 months, p = 0.04). Humoral response was not significant after complete vaccination (173.3 vs 89.9 AU/ml, p = 0.08). (3) A binary logistic regression was performed to ascertain the effects of age, dialysis vintage and CCI on the likelihood that patients develop a low response to vaccination, defined by IgG anti‐Spike under percentile 25 after both doses in each modality. The model was statistically significant, χ 2 (3) = 19, p < 0.001 and explained 37% of the variance in low response (Nagelkerke R 2) while also correctly classifying 82.1% of cases. Increasing CCI was associated with increased likelihood of low response (OR 1.56; 1.11–2.18 95% CI, p = 0.01), whereas increasing dialysis vintage was protective (OR 0.96; 0.93–0.99 95% CI, p = 0.03). Age alone did not contribute significantly to this model (p = 0.56).
4. DISCUSSION
Humoral response to BNT162b2 was significant in this MDP sample with only 9% of the total sample failing to achieve measurable response, coherent with current studies [37, 38]. All of these non‐responders were from the HD group, but, nevertheless, the difference in NR rate was non‐significant between modalities, illustrating the most important limitation of this study: a small sample size. Additionally, coupling this limitation with the non‐parametric distribution of resulting humoral titers impaired statistical analysis and restricted feasible testing, namely regression analysis.
The first studies of BNT162b2 in HD soon showed a reduce in humoral response, namely when compared to healthy cohorts [35, 36, 37]. While these outcomes were expected, our study supports a lower response in HD also when compared to PD in a real‐world environment. The reason for this difference, however, does not appear to reside simply on modality. Age and use of immunosuppressive therapy have both been associated with low seroconversion and humoral titers in this population [35, 36, 37, 38, 39]. The analysis of clinical and demographic data favors PD patients as a younger, with less dialysis vintage and less comorbidity burden (as evaluated by CCI) when compared to HD. These differences were expected, given the nature and requirements of each modality. The performed sub analysis of humoral response in younger patients (under 70 years of age), was executed to normalize subgroups for comorbidity burden and age, since the difference for these variables that was verified for the primary analysis lost its significance in this subgroup. As a result, and differently from the primary comparison, this sub analysis showed no significant difference in IgG titers after complete vaccination, hence supporting a secondary role of dialysis modality with age and comorbidity burden taking primacy. The second part of sub analysis was to use binary logistic regression using low responders (IgG anti‐S under percentile 25), rather than non‐responders (IgG anti‐S under 1 AU/ml) in order to identify common risk factors in low responders across both modalities. This resulted in a significant model, where CCI and dialysis vintage contributed, but age did not. Shorter dialysis vintage was associated with worse response in this model, even though PD patients have significant better quantitative responses and shorter dialysis vintage in the main analysis, suggesting CCI as stronger predictor. Still, early adaptation to dialysis, with lower dialysis adequacy and uremic toxin removal could play a role in these results [40]. Comorbidity burden, measured by CCI, is suggested as the most complete form of assessment for predicting humoral response. It contributed significantly to the likelihood of low response in the regression model and, although both CCI and age correlated significantly with IgG titers, CCI showed a stronger correlation coefficient across the entire sample of MDP. These results were sustained in a sub analysis for HD after separation by modality, but was not verified for PD alone, suggesting that CCI is particularly important in HD and can be used as a better marker of likelihood of insufficient response to vaccination over age individually, for this modality.
Taken together, these results suggest a superior response to vaccination with BNT162b2 in PD compared to HD, most likely due to comorbidity and clinical status over modality‐related factors, while also suggesting CCI as a better predictor of humoral response, over age alone, specifically in the HD population. It is licit to differentiate contingency protocols and to implement adequate policies for each modality, instead of a “one‐fits‐all” method for MDP, allowing for better allocation of resources and promoting the protection of those at higher risk of contagium and death. Consideration on a third or subsequent doses, similarly to other countries, is warranted, particularly in high‐risk patients since several recent studies have shown a faster decline in humoral titers when compared to the general population [41]. A recent study from France involving humoral response to BNT162b2 after three doses in a small sample of MDP (both PD and HD) revealed beneficial in patients that had not achieved a response with two doses [42]. Further investigation relating comorbidity burden and immune dysfunction is required to better establish causality and evaluate the usefulness of CCI quantification as a surrogate marker of immune deficiency in ESKD.
It is worth noting that, during the follow‐up, no patient developed clinically significant COVID‐19 disease, neither was diagnosed with it by nasopharyngeal swab.
5. CONCLUSION
This study suggests that humoral response in MDP is significantly different for BNT162b2 vaccination depending on modality, with PD patients achieving higher titers after both first and second doses when compared to their HD counterpart. Nevertheless, dialysis modality is not the crucial factor, with clinical and demographic differences between the two groups taking precedence, particularly age and comorbidity burden. CCI, as a quantitative measure of comorbidity burden, is a strong predictor for low response in MDP, and correlates with achieved humoral titers in HD patients, being suggested as a surrogate marker of immunodeficiency in this modality. Additional investigation with larger study groups is required to further explore immunogenicity factors and their influence on overall humoral response.
Regardless of the abovementioned differences and limitations, our study supports a quick and expeditious vaccination of all MDP, irrespective of modality, for the yield of elicited immunity is high, and therefore immunization should not be discouraged or postponed for fear of a lack of response. The identification and signalization of higher risk subgroups in MDP for no response to vaccination is essential, providing institutions an opportunity to develop and establish protective and follow‐up protocols in this pandemic, namely closer monitoring, adequate isolation, vigilance of symptoms and as a possible indication for booster doses.
To the best of our knowledge, this is the first study comparing humoral response to BNT162b2 between PD and HD patients.
CONFLICTS OF INTEREST
There are no relevant disclosures or conflicts of interest for any of the authors of this work.
Duarte R, Roldão M, Figueiredo C, Luz I, Ferrer F, Gonçalves H, et al. Humoral response to BNT162b2 mRNA COVID‐19 vaccine in peritoneal and hemodialysis patients: A comparative study. Ther Apher Dial. 2022;26:790–796. 10.1111/1744-9987.13766
REFERENCES
- 1. Polack FP, Thomas SJ, Kitchin N, Absalon J, Gurtman A, Lockhart S, et al. Safety and efficacy of the BNT162b2 mRNA Covid‐19 vaccine. N Engl J Med. 2020;383(27):2603–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Anderson EJ, Rouphael NG, Widge AT, Jackson LA, Roberts PC, Makhene M, et al. Safety and immunogenicity of SARS‐CoV‐2 mRNA‐1273 vaccine in older adults. N Engl J Med. 2020;383(25):2427–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Ramasamy MN, Minassian AM, Ewer KJ, Flaxman AL, Folegatti PM, Owens DR, et al. Safety and immunogenicity of ChAdOx1 nCoV‐19 vaccine administered in a prime‐boost regimen in young and old adults (COV002): a single‐blind, randomised, controlled, phase 2/3 trial. Lancet (London, England). 2021;396(10267):1979–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Stephenson KE, Le Gars M, Sadoff J, et al. Immunogenicity of the Ad26.COV2.S vaccine for COVID‐19. JAMA. 2021;325(15):1535–44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Wu C, Chen X, Cai Y, Xia J', Zhou X, Xu S, et al. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;180(7):934–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Flythe JE, Assimon MM, Tugman MJ, Chang EH, Gupta S, Shah J, et al. Characteristics and outcomes of individuals with pre‐existing kidney disease and COVID‐19 admitted to intensive care units in the United States. Am J Kidney Dis Off J Natl Kidney Found. 2021;77(2):190–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Shimada N, Shimada H, Itaya Y, Tomino Y. Novel coronavirus disease in patients with end‐stage kidney disease. Ther Apher Dial. 2021;25:544–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kato S, Chmielewski M, Honda H, Pecoits‐Filho R, Matsuo S, Yuzawa Y, et al. Aspects of immune dysfunction in end‐stage renal disease. Clin J Am Soc Nephrol. 2008. Sep;3(5):1526–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Cohen‐Hagai K, Kotliroff A, Rozenberg I, et al. Effectiveness of influenza vaccine in hemodialyzed patients: a retrospective study. Ther Apher Dial. 2019;23:38–43. [DOI] [PubMed] [Google Scholar]
- 10. Son J, Lee SB, Lee DW, Kim IY, Lee SJ, Lee SM, et al. Immunogenicity of low‐dose MF59‐adjuvanted 2009 influenza a/H1N1 vaccine in dialysis patients. Clin Exp Nephrol. 2013. Apr;17(2):275–83. [DOI] [PubMed] [Google Scholar]
- 11. Mitra S, Stein GE, Bhupalam S, Havlichek DH. Immunogenicity of 13‐valent conjugate pneumococcal vaccine in patients 50 years and older with end‐stage renal disease and on dialysis. Clin Vaccine Immunol. 2016;23(11):884–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Stevens CE, Alter HJ, Taylor PE, Zang EA, Harley EJ, Szmuness W, et al. Hepatitis B vaccine in patients receiving hemodialysis. N Engl J Med. 1984;311(8):496–501. [DOI] [PubMed] [Google Scholar]
- 13. Ramezani A, Velayati AA, Eslamifar A, Banifazl M, Ahmadi F, Maziar S, et al. Persistence of hepatitis B vaccine immunity in hemodialysis patients. Ther Apher Dial. 2008;12:143–6. [DOI] [PubMed] [Google Scholar]
- 14. Krueger M, Ison CG. Practical guide to vaccination in all stages of CKD, including patients treated by dialysis or kidney transplantation. Am J Kidney Dis. 2020;75:4176–425. [DOI] [PubMed] [Google Scholar]
- 15. Noh JY, Song JY, Choi WS, Lee J, Seo YB, Kwon YJ, et al. Immunogenicity of trivalent influenza vaccines in patients with chronic kidney disease undergoing hemodialysis: MF59‐adjuvanted versus non‐adjuvanted vaccines. Hum Vaccin Immunother. 2016;12(11):2902–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Espi M, Koppe L, Fouque D, Thaunat O. Chronic kidney disease‐associated immune dysfunctions: impact of protein‐bound uremic retention solutes on immune cells. Toxins (Basel). 2020;12(5):300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kim JU, Kim M, Kim S, Nguyen TT, Kim E, Lee S, et al. Dendritic cell dysfunction in patients with end‐stage renal disease. Immune Netw. 2017;17(3):152–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Girndt M, Trojanowicz B, Ulrich C. Monocytes in uremia. Toxins (Basel). 2020;12(5):340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Girndt M, Sester U, Sester M, Kaul H, Köhler H. Impaired cellular immune function in patients with end‐stage renal failure. Nephrol Dial Transplant. 1999;14(12):2807–10. [DOI] [PubMed] [Google Scholar]
- 20. Ando M, Shibuya A, Tsuchiya K, Akiba T, Nitta K. Reduced expression of toll‐like receptor 4 contributes to impaired cytokine response of monocytes in uremic patients. Kidney Int. 2006. Jul;70(2):358–62. [DOI] [PubMed] [Google Scholar]
- 21. Choi HM, Woo YS, Kim MG, Jo SK, Cho WY, Kim HK. Altered monocyte‐derived dendritic cell function in patients on hemodialysis: a culprit for underlying impaired immune responses. Clin Exp Nephrol. 2011;15:546–53. [DOI] [PubMed] [Google Scholar]
- 22. Hoyo G, Martín P, Anjuère F, et al. Origin and differentiation of dendritic cells. Trends Immunol. 2001;22:691–700. [DOI] [PubMed] [Google Scholar]
- 23. Merad M, Manz MG. Dendritic cell homeostasis. Blood. 2009;113:3418–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Verkade MA, van Druningen CJ, Vaessen LM, et al. Functional impairment of monocyte‐derived dendritic cells in patients with severe chronic kidney disease. Nephrol Dial Transplant. 2007;22:128–38. [DOI] [PubMed] [Google Scholar]
- 25. Verkade MA, van Druningen CJ, de Hoek OP, et al. Decreased antigen‐specific T‐cell proliferation by moDC among hepatitis B vaccine non‐responders on haemodialysis. Clin Exp Med. 2007;7:65–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Yoon J‐W, Gollapudi S, Pahl MV, Vaziri ND. Naive and central memory T‐cell lymphopenia in end‐stage renal disease. Kidney Int. 2006;70(2):371–6. ISSN 0085‐2538. [DOI] [PubMed] [Google Scholar]
- 27. Litjens NH, Huisman M, van den Dorpel M, Betjes MG. Impaired immune responses and antigen‐specific memory CD4+ T cells in hemodialysis patients. J Am Soc Nephrol. 2008;19(8):1483–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Cohen G. Immune dysfunction in uremia. Toxins (Basel). 2020;12(7):439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Gollapudi P, Yoon JW, Gollapudi S, Pahl MV, Vaziri ND. Leukocyte toll‐like receptor expression in end‐stage kidney disease. Am J Nephrol. 2010;31(3):247–54. [DOI] [PubMed] [Google Scholar]
- 30. Wang AY‐M. Consequences of chronic inflammation in peritoneal dialysis. Semin Nephrol. 2011;31(2):159–71. [DOI] [PubMed] [Google Scholar]
- 31. Wang X, Grobe N, Patel A, et al. Presence of SARS‐CoV‐2 antibodies in spent peritoneal dialysate. J Am Soc Nephrol. 2021;32(8): 1865 LP ‐ 1867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Stepniewska J, Dolegowska B, Golembiewska E, et al. The activation of complement system in different types of renal replacement therapy. J Physiol Pharmacol. 2020;71(2):275–81. [DOI] [PubMed] [Google Scholar]
- 33. Hoedt C, Bots M, Grooteman M, et al. Online hemodiafiltration reduces systemic inflammation compared to low‐flux hemodialysis. Kidney Int. 2014;86(2):423–32. [DOI] [PubMed] [Google Scholar]
- 34. Sit D, Esen B, Atay AE, Kayabaşı H. Is hemodialysis a reason for unresponsiveness to hepatitis B vaccine? Hepatitis B virus and dialysis therapy. World J Hepatol. 2015;7(5):761–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Grupper A, Sharon N, Finn T, et al. Humoral response to the Pfizer BNT162b2 vaccine in patients undergoing maintenance hemodialysis. Clin J Am Soc Nephrol. 2021;16(7):1037–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Attias P, Sakhi H, Rieu P, Soorkia A, Assayag D, Bouhroum S, et al. Antibody response to the BNT162b2 vaccine in maintenance hemodialysis patients. Kidney Int. 2021;99(6):1490–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Ikizler TA, Coates PT, Rovin BH, Ronco P. Immune response to SARS‐CoV‐2 infection and vaccination in patients receiving kidney replacement therapy. Kidney Int. 2021;99(6):1275–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Broseta JJ, Rodríguez‐Espinosa D, Rodríguez N, Mosquera MM, Marcos MÁ, Egri N, et al. Humoral and cellular responses to mRNA‐1273 and BNT162b2 SARS‐CoV‐2 vaccines administered to hemodialysis patients. Am J Kidney Dis. 2021;78(4):571–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Frantzen L, Cavaillé G, Thibeaut S, el‐Haik Y. Efficacy of the BNT162b2 mRNA COVID‐19 vaccine in a haemodialysis cohort. Nephrol Dial Transplant. 2021;36(9):1756–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Broers NJ, Cuijpers AC, van der Sande FM, et al. The first year on haemodialysis: a critical transition. Clin Kidney J. 2015;8(3):271–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Sakhi H, Dahmane D, Attias P, Kofman T, Bouvier M, Lapidus N, et al. Kinetics of anti–SARS‐CoV‐2 IgG antibodies in hemodialysis patients six months after infection. J Am Soc Nephrol. 2021;32:1033–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Bensouna I, Caudwell V, Kubab S, Acquaviva S, Pardon A, Vittoz N, et al. SARS‐CoV‐2 antibody response after a third dose of the BNT162b2 vaccine in patients receiving maintenance hemodialysis or peritoneal dialysis. Am J Kidney Dis. 2021;S0272‐6386(21):833–7. [DOI] [PMC free article] [PubMed] [Google Scholar]


