Abstract
Mesenchymal stem cells (MSCs) play an important role in developing bone tissue engineered constructs due to their osteogenic and chondrogenic differentiation potential. MSC-based tissue engineered constructs are generally considered a safe procedure, however, the long-term results obtained up to now are far from satisfactory. The main causes of these therapeutic limitations are inefficient homing, engraftment, and directional differentiation. Flavonoids are a secondary metabolite, widely existed in nature and have many biological activities. For a long time, researchers have confirmed the anti-osteoporosis effect of flavonoids through in vitro cell experiments, animal studies. In recent years the regulatory effects of flavonoids on mesenchymal stem cells (MSCs) differentiation have been received increasingly attention. Recent studies revealed flavonoids possess the ability to modulate self-renewal and differentiation potential of MSCs. In order to facilitate further research on MSCs osteogenic differentiation of flavonoids, we surveyed the literature published on the use of flavonoids in osteogenic differentiation of MSCs, and summarized their pharmacological activities as well as the underlying mechanisms, aimed to explore their promising therapeutic application in bone disorders and bone tissue engineered constructs.
Keywords: tissue engineering, osteogenic differentiation, mesenchymal stem cells, flavonoids, osteoporosis
Introduction
People are living longer than ever before because of medical, social and economic advances in the whole world. However, increasing life expectancy also brings tremendous challenges to the society, like chronic non-communicable diseases including osteogenesis are becoming the leading cause of death and disability (Williams et al., 2018; Garmany et al., 2021). Osteoporosis is a condition in that bones become weaker and more fragile owing to bone mass loss with ageing, diseases and drugs, so the chances are higher they’ll crack or break. It is predicted that osteogenesis fractures will account for over 50% of the total fractures, and unlike bone fractures in young people, osteogenesis fractures induce a large proportion of disability and mortality in elderly people (Patel et al., 2021). Additionally, poor fracture healing can cause critical-sized bone defects (Miller, 2016; Nauth et al., 2018). Mesenchymal stem cells (MSCs)are a kind of adult stem cells with multiple differentiation potentials (Friedenstein, 1976) and exist in a variety of tissues including bone marrow, adipose tissue, umbilical cord, etc., (Baksh et al., 2004). Stand as promising candidates in the treatment of bone defects and other degenerative bone diseases, MSCs have great potential use in the bone repair and regeneration owing to their osteogenic differentiation potential and extensive sources. However, the ability of MSC to differentiate into osteoblasts may become impaired under certain pathophysiological conditions including oxidative stress and inflammation (García-Sánchez et al., 2019). Hence, the strategies aimed to increase cell survival and osteogenic capacity are important for the MSCs-based bone regeneration therapies. Strategies, including promoting MSCs osteogenic differentiation through genetic modification (Armstrong and Stevens, 2019), or providing the appropriate extracellular environmental cues like scaffolds, growth factors or other bioactive molecules, are commonly used (Velasco et al., 2015; Yang et al., 2017). For example, combining β-tricalcium phosphate and BMP-2 has been proven to be effective to enhance the osteogenesis of MSCs (Dimitriou et al., 2011).
Some botanical drugs have been effective and safe in the treatment of fracture healing in China for a long time, and more and more evidences show many ingredients of them are beneficial to bone health. Flavonoids are commonly present in botanicals, they are synthesized in plants as secondary metabolites, and characterized with diverse pharmacological properties (Martens and Mithöfer, 2005). Natural flavonoids and their glycosides have been identified and explored for their therapeutic potentials in different fields including osteoporosis-related complication and disorders. Many flavonoids exerted promoting bone formation and anti-osteoporosis effects through stimulating osteogenic differentiation of MSCs (Huang et al., 2018; Wang et al., 2018; Casado-Díaz and Rodríguez-Ramos, 2021). Also, European nutritional studies demonstrated that daily intake of flavonoids contributed to good bone health (Zamora-Ros et al., 2016), Therefore, combining flavonoids and MSCs would be an efficient strategy to enhance bone formation and increase cell survival in the field of bone tissue engineering.
Basic Structure and Classification of Flavonoids
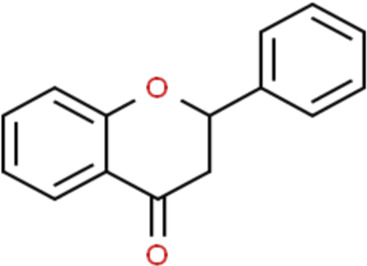
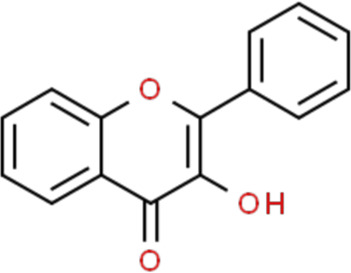
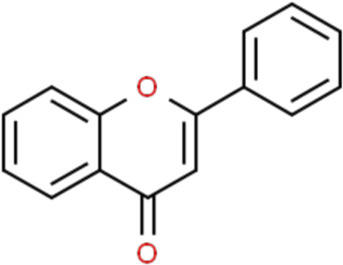
Flavonoids are a kind of polyphenolic compounds widely present in nature and have spectral biological activities. In terms of chemical structure, flavonoids generally refer to a series of hydroxylated phenolic molecules consisting with a C6-C3-C6 units, in which two benzene rings (A and B rings) are linked to each other through three central carbon atoms ring (ring C). These compounds can be divided into many different classes according to the oxidation degree of the central three carbons, whether the three carbons constitute a ring and the connection site of B ring, and so on. Generally, flavonoids are mainly classified into the following subclasses: flavanones, flavonols, flavonones, and isoflavones, anthocyanins, flavanols, (Amarowicz et al., 2009; Kumar and Pandey, 2013), and their basic chemical structure and representative compounds are shown in Table 1.
TABLE 1.
Structure of flavonoids subclasses.
| Subclass name | Core chemical structure | Typical compounds |
|---|---|---|
| Flavanones |
|
Naringin, Hesperetin |
| Flavonols |
|
Quercetin, Kaempferol, Rutin |
| Flavones |
|
Luteolin, Apigenin |
| Isoflavones |
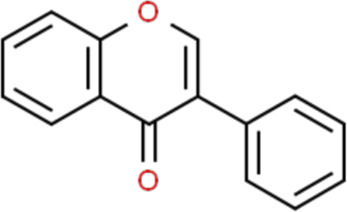
|
Genistein, Daidzein |
| Anthocyanins |
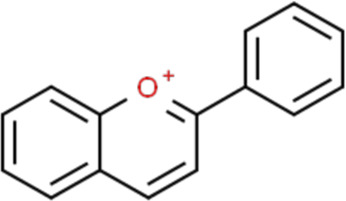
|
Delphinidin, Cyanidin |
| Chalcones |
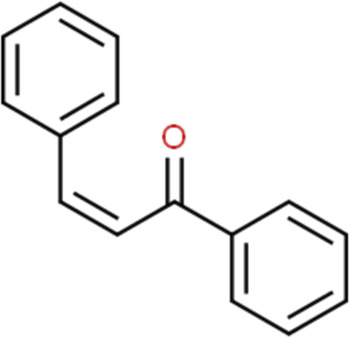
|
Xanthohumol |
Biological Activity of Flavonoids
Flavonoids have a wide range of pharmacological activities including anti-inflammatory, anti-oxidative, anti-microbial, and anti-tumor properties (Kumar and Pandey, 2013; Wen et al., 2017) (Figure 1). These properties are beneficial to bone regeneration. Firstly, many flavonoids, including baicalin (Guo et al., 2019; Huang et al., 2019), Kaempferol (Hwang et al., 2019), exert their anti-inflammatory effects by inhibiting the activation of the nuclear factor NF-κB pathway which is closely associated with inflammation. Kaempferol, a bioflavonoid extracted from Persicaria tinctoria (Aiton) Spach (Polygonaceae), prevented bone resorption through its anti-inflammatory property on osteoclast precursor cells (Hwang et al., 2019). Flavonoids have free radical scavenging activities through inhibiting the formation of free radicals, reducing lipid peroxidation, and stimulating antioxidant enzymes (Pietta, 2000). Secondly, given their anti-oxidative roles of flavonoids, some of them have been applied in clinical treatments. For example, troxerutin, a semi-synthetic flavonoid compound prepared by hydroxymethylation of rutin, is commonly used to treat ischemic cerebrovascular diseases, thrombophlebitis, central retinitis, and so on (Ahmadi and Mohammadinejad, 2021). Finally, flavonoids have been demonstrated anti-tumor effects through inhibiting tumor cell proliferation and metastasis, inducing tumor cell autophagy or apoptosis, and preventing tumor invasion. High intake of dietary flavonols, flavones and anthocyanidins may decrease the risk of cancer (Zamora-Ros et al., 2016; Chang et al., 2018).
FIGURE 1.
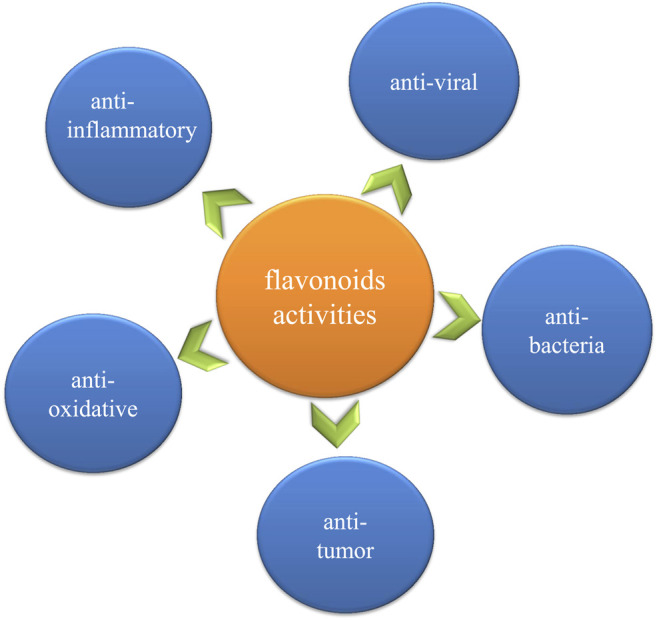
Schematic presentation of the biological activity of flavonoids.
Botanicals containing flavonoids compounds have been extensively used in traditional medicines for centuries, and nowadays many flavonoids have been extracted or synthesized and have been applied to treat various diseases in clinics. For example, diosmin, a semi-synthetic flavonoid drug with, is used to treat chronic venous insufficiency and varicose veins (Zheng et al., 2020). However, even if there are a number of well-tried treatment experiences of botanicals that are safe for clinical use, there are still many disagreements due to their ambiguous mechanisms. Investigating the underlying mechanisms of these herbal extracts will help gain deeper understanding of their beneficial pharmacological activities and facilitate medicinal applications.
Effects of Flavonoids on Osteogenic Differentiation in of Mesenchymal Stem Cells
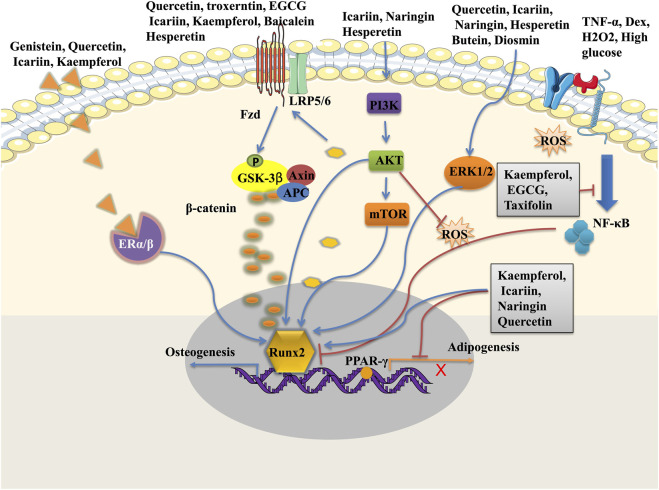
Many herbal medicines for the treatment of fractures and other degenerative bone diseases have been used for several centuries in some countries, and modern pharmacology confirmed their major biologically active ingredients are flavonoids, such as soybean isoflavones, and flavonoids from Drynaria roosii Nakaike (Polypodiaceae), Epimedium brevicornu Maxim (Berberidaceae), etc. (An et al., 2016). Among them, the corresponding active monomers, including naringin, icariin, genistein, and daidzein, have been proved to be able to regulate bone tissues metabolism by enhancing osteogenic differentiation and inhibiting osteoclast-mediated bone resorption. Especially their osteogenic induction potentials make flavonoids potential candidates to interfere with the osteogenic differentiation of MSCs (Table 2). Studies revealed flavonoids modulated the self-renewal and osteogenic differentiation potential of MSCs by targeting multiple signal pathways such as Wnt/β-catenin pathway, ERK pathway, PI3K/Akt pathway, and regulating the bone-specific markers and transcription factors including ALP, Runx2, BMP-2, Cbfa1, Osx (An et al., 2016; Zhang et al., 2016a) (Figure 2). In addition to directly stimulating the osteogenesis of MSCs, flavonoids could also indirectly affect the osteogenic differentiation of MSCs by their well-known antioxidant and anti-inflammatory properties (Schilling et al., 2014; Zhang ND. et al., 2016; Zhang L. et al., 2021). Besides, flavonoids were also loaded on bioscaffolds for the promotion of MSCs self-renewal and differentiation in bone regeneration. The utilization of flavonoids in biomaterials showed to be a great prospect for bone tissue engineering.
TABLE 2.
The list of some flavonoids compounds on MSCs osteogenic differentiation.
| Compound name | Dosage | In vitro | In vivo | Results and mechanism of action |
|---|---|---|---|---|
| Icariin | 0.1–10 μM | hBMSC, hADSC | - | Enhance hBMSC and hADSC osteogenesis (Wu et al., 2017a) |
| 0.1 μM | rADSCs/glass scaffold | rat calvarial bone | Up-regulation VEGF expression, enhance angiogenesis, promote bone formation (Jing et al., 2018) | |
| 5–40 μM | hBMSC | defect models | osteogenesis ↑, adipogenesis ↓; miR-23a ↓, active Wnt/β-catenin (Xu et al., 2021) | |
| 0.01–1 μM | hBMSCs, rBMSCs | - | Osteogenesis ↑; sclerostin ↓, Wnt/β-catenin/ERα activation (Gao et al., 2021) (Wei et al., 2020) | |
| 10–20 μM | rat mandibular MSCs | - | osteogenesis ↑, bone osteoporosis ↓; osteocalcin ↑, STAT 3 pathway activation (Xu et al., 2020) | |
| 1 μM | rBMSCs | OVX rats | proliferation ↑, osteogenesis ↑, adipogenesis ↓; ERα pathway activation (Li et al., 2018) | |
| 0.1–10 μM | rBMSCs | - | protect against iron overload induced dysfunction of BMSCs; active PI3K/AKT/mTOR pathway, inhibit ERK1/2 and JNK pathways (Yao et al., 2019) | |
| 0.1 μM | rBMSCs | - | osteogenesis ↑, TAZ ↑; active ERα and Wnt/β-catenin pathway (Wei et al., 2017) | |
| 50 mg/kg | mBMSCs | - | osteogenesis ↑, bone loss ↓; autophagy activation (Liang et al., 2019) | |
| Quercetin | 2–10 μM | hADSCs | - | proliferation ↓, osteogenesis ↑, ERK activity ↑, ER independent (Kim et al., 2006) |
| 1 μM | rBMSCs/nHA microspheres | OVX fracture rats | proliferation ↑, osteogenesis ↑, angiogenesis ↑; ERK, p38 and AKT activity ↑, RANKL ↓ (Zhou et al., 2017) | |
| 10 μM | hBMSCs/scaffold | - | proliferation ↑, osteogenesis ↑, quercetin-crosslinked nHAp-modified decellularized goat-lung scaffold (Gupta et al., 2017) | |
| 0.03 (wt%) | rabbit BMSC/SF/HAp scaffold | calvarial defect rats | osteogenesis ↑, proliferation ↑, bone regeneration ↑(Song and Tripathy, 2018) | |
| 1–2 (wt%) | hUCMSCs/3D printing scaffold | - | cells growth and mitosis ↑, osteogenesis ↑, calcium deposit ↑(Huang et al., 2021) | |
| 10 μM | rBMSC | - | osteogenesis ↑, adipogenesis ↓, active ERα-mediated circRNA-miR-326-5p-axis (Li et al., 2021) | |
| Quercetin 3-O-β-D-galactopyranoside | 1–25 μM | hBMSCs | - | proliferation ↑, osteogenesis ↑, adipogenesis ↓, active Wnt/BMP pathway, inhibit PPARγ pathways (Oh et al., 2020) |
| Isoquercitrin | 0.1–1 μM | rBMSCs | maxillary expansion rats (10 mg/kg) | proliferation ↑, osteogenesis ↑, BMP2 ↑, bone formation ↑(Li et al., 2019a; Li et al., 2019b) |
| Hesperetin | 1–10 μM | BMSCs | - | DEX-induced osteogenic inhibition ↓, active ERK signal pathway (Liu et al., 2021b) |
| 1 μM | hBMSCs/gelatin scaffold | rat osteotomy model | osteogenesis ↑, active ERK and Smad pathways, accelerate fracture healing (Xue et al., 2017) | |
| 10–100 μM | PDLSCs | - | osteogenesis ↑, ROS ↓, active PI3K/Akt and β-catenin signal pathways (Kim et al., 2013) | |
| Naringin | 1–100 μg/ml | hAFSCs | - | proliferation ↑, osteogenesis ↑, BMP4 ↑, active Wnt/BMP pathway (Liu et al., 2017) |
| 0.03–0.1 (wt%) | hUCMSCs/SF-nHAp scaffolds | rabbit bone defect | proliferation ↑, osteogenesis ↑, angiogenesis ↑, bone regeneration ↑, active PI3K/Akt pathways (Zhao et al., 2021) | |
| 20–100 μM | NPMSC | - | H2O2-induced cell apoptosis ↓; mitochondrial function ↑ (Nan et al., 2020) | |
| 70 μg/ml | rabbit MSC/scaffolds | rabbit bone defect | bone formation ↑, inhibit BMPR-1A signaling (Dong and Ma, 2020) | |
| 0.1 μM | rBMSC | - | restore TNF-α-induced osteogenesis and proliferation inhibition, p-IкBα and nuclear p65 ↓, inhibit NF-кB pathway (Cao et al., 2015) | |
| 1–100 μg/ml | rBMSC | OVX rats | proliferation ↑, osteogenesis ↑, bone loss ↓, inhibit JAK2/STAT3 pathway (Wang et al., 2022) | |
| Kaempferol | 1 μM, 10 mg/kg | rBMSCs | OVX rats | bone density ↑, osteogenesis ↑, CXCL12 ↑, miR-10a-3p ↓(Liu et al., 2021a) |
| 0.1–100 μM or 25–100 mg/kg | rBMSCs | OVX rats bone defect | osteogenesis ↑, prevent OVX-induced osteoporosis, p-4E/BP1 ↓, p-S6K ↑, active mTOR pathway (Zhao et al., 2019) | |
| 20–100 μM | rabbit BMSC | - | cells viability ↑, osteogenesis ↑, adipogenesis ↓, IL-10 ↑, IL6 ↓, inhibit NF-κB pathway (Zhu et al., 2017) | |
| 50 μg/ml | rBMSC/TiO2 implants | rats femur bone defect | cell proliferation ↑, osteogenesis ↑, bone formation ↑, kaempferol-loaded TiO2 implants (Tsuchiya et al., 2018) | |
| 2–10 μM | hADSCs | skull defect mice | cell proliferation ↓, osteogenesis ↑, ERK activity ↑, bone regenerating ability ↑(Kim et al., 2006) | |
| EGCG | 1–10 μM | hBMSCs | rats femoral bone defect | Osteogenesis ↑, Runx2 ↑, BMP2 ↑, bone defect healing ↑ (Lin et al., 2018b; Lin et al., 2019) |
| 5–40 μM | hBMSCs | - | hypoxia-induced apoptosis ↓, ameliorate hypoxia-induced osteogenesis reduction, miR-210 ↑, EFNA3 ↓(Qiu et al., 2016) | |
| 1–10 μM | mBMSCs | - | cell proliferation ↓, osteogenesis ↑, Cbfa1 ↑, Runx2 ↑(Chen et al., 2005) | |
| 1–10 μM | SCAPs | - | Proliferation ↑, osteogenesis ↑, Dspp ↑, Dmp-1 ↑, active BMP-Smad signaling pathway (Liu et al., 2021c) | |
| 1–10 μM | rabbit BMSCs | nude mouse | EGCG/DC/HAp sponges increased cell internalization, attachment proliferation, ALP ↑ (Kook et al., 2018) | |
| Genistein | 0.01–1 μM | hBMSCs | - | Proliferation ↑, osteogenesis ↑, BMP2 ↑, SMAD5 ↑, RUNX2, ER dependent (Dai et al., 2013) |
| 5–20 μM | rBMSCs | - | Proliferation ↑, osteogenesis ↓, PPARγ ↑(Zhang et al., 2016a) | |
| 1 μM | hBMSCs | - | Osteogenesis ↑, adipogenesis ↓, PPARγ ↓, ER-dependent, TGF-β ↑ (Heim et al., 2004) | |
| Ipriflavone | 0.4–0.8 μM | rBMSCs | OVX rats | osteogenesis ↑, osteoporosis ↓, BMD ↑ (Gao et al., 2018) |
| Malvidin | 25 μM | hADSC | - | calcium deposits ↑, BMP-2 and Runx-2 ↑(Saulite et al., 2019) |
| Taxifolin | 15 μM | hBMSC | - | Osteogenesis ↑, inhibit NF-κB pathway (Wang et al., 2017b) |
| Diosmin | 10–100 μM | C3H10T1/2 | - | Osteogenesis ↑, runx2 ↑, active FAK/ERK signaling pathway (Chandran et al., 2019) |
| Tricin | 50–100 μM | hMSC(ATCC) | - | Proliferation ↑, osteogenesis ↑, Wnt3α- mediated (Zhang and Li, 2018) |
| Glabridin | 5 μM | hBMSC | - | osteogenesis ↑, OCT4 gene↑(Heo and Lee, 2017) |
| HYSA | 0.05–0.2 mg/ml | rabbit MSCs | - | prevent glucocorticoid-induced osteoporosis (Wan et al., 2014) |
| 0.1–0.5 mg/ml | rBMSCs/scaffold | rats bone defect | Osteogenesis ↑, HIF-1α ↑, BMP-2 ↑, new bone formation ↑(Deng et al., 2020) | |
| Butein | 1–30 μM | mBMSCs, hBMSCs | - | Osteogenesis ↑, adipogenesis ↓.activate ERK1/2 signaling pathway (Abdallah and Ali, 2020) |
| Baicalein | 0.1–10 μM | TDSCs | tendon-bone healing rat model | Osteogenesis ↑, active Wnt/β-catenin signaling pathway (Tian et al., 2018) |
| Amentoflavone | 0.1–5 μM | hBMSCs | - | Osteogenesis ↑, p-p38 ↑, active JNK and p38 MAPK pathway (Zha et al., 2016) |
| Troxerutin | 25–200 μM | hBMSC | fracture rats model | Osteogenesis ↑, fracture healing ↑, active Wnt/β-catenin signaling (Yang et al., 2021) |
| Fisetin | 200–800 μg/ml | rBMSCs/BC scaffold | - | BC scaffold loaded with fisetin promote osteogenesis (Vadaye Kheiry et al., 2018) proliferation ↓, migration ↓, YAP ↓, osteogenic differentiation ↓(Lorthongpanich and Charoenwongpaiboon, 2021) |
| 1–30 μM | chorion tissue hMSC | - | BC scaffold loaded with fisetin promote osteogenesis (Vadaye Kheiry et al., 2018) proliferation ↓, migration ↓, YAP ↓, osteogenic differentiation ↓(Lorthongpanich and Charoenwongpaiboon, 2021) |
hUCMSCs, human umbilical cord-derived mesenchymal stem cells; hAFSCs, human amniotic fluid-derived stem cells; NPMSC, nucleus pulposus-derived mesenchymal stem cells; NG/SF/HAp, naringin-inlaid composite silk fibroin/hydroxyapatite; SCAPs, Stem cells from apical papilla; TDSCs, tendon-derived stem cells; PDLSCs, periodontal ligament stem cells; HYSA, Hydroxy Safflower Yellow A.
FIGURE 2.
Signalling pathways of flavonoids in MSCs osteogenic differentiation.
Icariin
5Icariin (ICA) is a kind of flavonol glycoside and generally extracted from Epimedium brevicornu Maxim (Berberidaceae), a traditional Chinese herbal medicine for bone repair. A large number of studies have revealed that ICA had protective roles on bone loss and bone regeneration (Fu et al., 2016; Wang J. et al., 2016; Wei et al., 2017; Ye et al., 2017; Wang et al., 2018; Liang et al., 2019; Gao et al., 2021). ICA not only increased the ALP activity and mineralization of BMSCs but also reduced bone resorption mediated by overactivated osteoclasts in OVX-induced osteoporosis mice (Liang et al., 2019). In addition, ICA has been shown to promote proliferation by activating the Wnt/β-catenin signaling pathway (Wang J. et al., 2016; Gao et al., 2021), which is the most important pathway in osteogenesis. In Sprague-Dawley (SD) rats, ICA stimulated BMSCs proliferation by increasing the phosphorylation level of GSK-3β and cyclin D1 protein (Fu et al., 2016). ICA has been reported to promote proliferation and osteogenic differentiation through increasing the expression of transcriptional coactivator with PDZ-binding motif (TAZ) both in rat BMSCs and ADSCs (Wei et al., 2017; Ye et al., 2017). Furthermore, the up-regulation of TAZ induced by ICA could be blocked by ICI 182780 or DKK1 (the Wnt/β-catenin pathway inhibitor), which indicated Wnt/β-catenin signaling pathway and ER signaling pathway were involved in the osteogenic differentiation of rBMSCs induced by ICA. The proliferation of rat BMSCs enhanced by ICA is also achieved through activating ERK and p38 MAPK signaling (Fu et al., 2016).
Similarly, Icariside II, a kind of metabolite of ICA, enhanced osteogenic differentiation of BMSCs by increasing ALP activity and calcium deposition at 10 µM (Luo et al., 2015). Icaritin, another metabolic product of ICA, significantly increased ALP activity and calcium deposition at concentrations 0.1–10 µM in human BMSCs and ADSCs through increasing the expression of BMPs, and showed better osteogenesis induction ability than rhBMP-2 (Wu T. et al., 2017).
Quercetin and Kempferol
Quercetin and kaempferol are the main representatives of flavonols, which are the 3-hydroxy derivatives of flavanones, probably the most ubiquitous subclass of flavonoids in nature. The anti-inflammatory and antioxidant effects of quercetin and kaempferol have been repeatedly reported (Babaei et al., 2018; Dabeek and Marra, 2019; Kashyap et al., 2019).
With the development of tissue engineering, the roles of quercetin on the osteogenic differentiation of MSC gradually attracted more attentions. It was reported quercetin could increase bone mineral density (BMD) and improve bone biomechanical properties in postmenopausal osteoporosis rat models (Yuan et al., 2018). The increase of BMP-2 and TGF-β1, two main osteogenic factors, was observed in rat and mouse BMSCs treated with quercetin (Li et al., 2015). Furthermore, the ability of quercetin to stimulate proliferation and osteogenic differentiation of mouse BMSCs could be blocked by estrogen receptor inhibitor ICI182780 (Pang et al., 2018). This indicates that quercetin enhances osteogenic differentiation of MSCs by an estrogen receptor-dependent mechanism. However, in another study similar effects of quercetin on human adipose tissue-derived stem cells (hADSCs) could not reversed by ICI182780, despite the fact that it up-regulated the expression of BMP2, Runx2, as well as activated ERK phosphorylation (Kim et al., 2006). Quercetin also promoted the differentiation and proliferation of BMSCs through inhibiting NF-κB activation and β-catenin degradation stimulated by TNF-α (Yuan et al., 2018). Bian et al. also observed Wnt/β-catenin pathway activation played an important role in the osteogenic differentiation of quercetin treated-BMSCs (Bian et al., 2021). Quercetin stimulated osteogenic differentiation of BMSCs by increasing connexin 43 expressions (Zhang et al., 2020) which could enhance osteogenic differentiation of BMSCs by promoting GSK-3β/β-catenin signaling pathways (Lin FX. et al., 2018).
Kaempferol, another representative flavonol, had a similar osteogenic induction potential with quercetin in periodontal ligament stem cells (PDLSCs). The treatment with 10–6 M kaempferol increased cell viability, ALP activity, and enhanced calcium mineralization of PDLSCs. Furthermore, these effects of kaempferol could be reversed by XAV939, a tankyrase inhibitor, indicating Wnt/β-catenin signaling pathway was involved (Nie et al., 2020). The activated Wnt/β-catenin signaling by kaempferol, to some extent, depended on estrogen receptors, as the activation of Wnt/β-catenin could be markedly blocked by the ICI 182780, the inhibitor of estrogen receptors (Sharma and Nam, 2019).
Naringin and Hesperetin
Naringin and hesperetin are two of the best-studied compounds in flavanones (Den Hartogh and Tsiani, 2019; Tutunchi et al., 2020). Naringin is rich in grapefruit and also the main active component of Drynaria roosii Nakaike (Polypodiaceae), a Chinese herbal medicine commonly used to treat orthopedic disorders and bone injury (Lavrador et al., 2018). Naringin dose-dependently increased ALP activity and Alizarin red S staining, and decreased PPARγ2 mRNA expression that is the marker of adipogenesis in rat BMSCs under osteogenic induction. Furthermore, this osteogenic effect of naringin could be reversed by the inhibitor of Notch signaling, indicating naringin exerted its role through activating the Notch signaling pathway (Yu G. Y. et al., 2016). In human BMSCs, wang et al. demonstrated naringin promoted proliferation and osteogenesis by activating the ERK signaling pathway (Wang H. et al., 2017). The gene expressions of bone morphogenetic protein 4 (BMP4), runt-related transcription factor 2 (Runx2), β-catenin, and Cyclin D1 were significantly up-regulated by naringin in human amniotic fluid-derived stem cells (Liu et al., 2017). In addition, Naringin alleviates the inhibitory effect of various stimulating factors on osteogenic differentiation of MSC. In a glucocorticoid-induced osteoporosis rat model, naringin not only improved bone mineral density and bone morphology parameters, but also stimulated the expression of autophagy-related factors including Beclin-1 and p62, which indicated autophagy was also involved in the bone protective effect of naringin (Ge and Zhou, 2021). Similarly, Hesperidin and its aglycone, hesperetin, two flavonoids from citrus species, also exerted protective roles in the osteogenesis of MSCs (Parhiz et al., 2015). In dexamethasone-treated BMSCs, the inhibition of MSC osteogenesis was reversed by the intervention of hesperetin through activating the ERK signal pathway (Liu L. et al., 2021). retreatment with 1–100 μM concentration hesperetin significantly increased the osteogenic activity of periodontal ligament stem cells under high glucose conditions. It was considered scavenged intracellular ROS produced and activated PI3K/Akt and β-catenin signaling pathway by hesperetin were responsible for this protective effect (Kim et al., 2013).
Epigallocatechin-3-Gallate
Tea is abundant in flavonoids, mainly including catechins, theaflavins, alkaloids, etc., (-)-epigallocatechin-3-gallate (EGCG) is the major catechin isolated from Green Tea (Friedman, 2007). As an antioxidant and anti-inflammatory agent, EGCG plays an important role in maintaining the balance of bone metabolism through the inhibition of bone resorption as well as the enhancement of bone formation (Nishioku et al., 2020). Although EGCG alone could not induce osteogenic differentiation of MSC, EGCG was able to enhance osteogenesis under osteogenic induction environment through upregulating BMP2 expression (Jin et al., 2021). Lin et al. showed EGCG enhanced osteogenic differentiation at the concentrations range from 1 to 10 µM both in murine and human BMSCs by increasing the expression of osteoblastic genes including BMP2, Runx2, ALP, osteonectin, and osteocalcin, as well as promoting ALP activity and mineral deposits (Lin S. Y. et al., 2018). Furthermore, the effect of EGCG on promoting the mineralization of human MSCs is independent of its antioxidant activity (Lin S. Y. et al., 2018). In human ADSCs, 5 μM EGCG significantly enhanced cell proliferation and ALP activity, experimental data revealed that osteo-inductive effects of EGCG on human ADSCs were associated with the inhibition of adipogenesis-related gene expression (Zhang et al., 2019). For the stem cells from apical papilla (SCAPs), at low concentrations, EGCG promoted the cell proliferation and increased ALP activity as well as mineral deposition through activating the BMP-Smad Signaling Pathway (Liu Z. et al., 2021). In addition to promoting osteogenic differentiation directly, EGCG treatment significantly reversed the inhibition of MSC osteogenesis induced by hydrogen peroxide and inflammatory cytokines. EGCG could enhance osteogenic differentiation by increasing the expression of β-catenin and cyclin D1 in H2O2-induced human BMSCs (Wang D. et al., 2016). A similar protective effect of EGCG was also observed on TNF-α-induced osteogenesis inhibition of MSC, in which EGCG reversed the TNF-α-induced destructive through inhibition of NF-κB signaling (Liu et al., 2016).
Genistein
Genistein is one of the most abundant isoflavones in Glycine max (L.) Merr (Fabaceae), and it is also called a phytoestrogen owing to its similar structure to that of human estrogen. It could bind to ERα and ERβ and exert ER-mediated estrogenic effects including increasing bone formation and repressing adipose tissue (Jaiswal et al., 2019) At the same time, it possessed antiestrogenic effects as well as non-ER-mediated effects like inhibiting tyrosine kinase (Dang et al., 2003). Genistein exerted estrogenic effects mainly by binding to ER α, even with a stronger affinity to ERβ than to ERα, genistein, and 8-prenylgenistein (a prenylated derivative), all of them could inhibit GSK-3β enzymatic activities though inducing GSK-3β phosphorylation at Serine-9 in human BMSCs and murine pre-osteoblast MC3T3-E1 cells. In addition, 8-prenylgenistein showed stronger osteogenic ability than genistein in MC3T3-E1 cells by increasing ERα-dependent β-catenin protein expression (Qiu et al., 2020). It seemed that both Wnt/β-catenin and ERα-associated signaling were involved in the osteogenic activities of genistein. Owing to its well-known estrogenic ability, genistein directly or indirectly affected the osteogenic and adipogenic differentiation of MSCs. In the early stages of differentiation of human primary BMSCs osteogenic markers were strongly up-regulated by genistein, while during adipogenic differentiation, adipogenic regulators, including PPARγ and CCAAT/enhancer-binding protein-α, were down-regulated after genistein treatment (Heim et al., 2004). A lineage shift from adipogenesis to osteogenesis induced by genistein was observed in murine MSCs and pre-osteoblasts isolated from newborn mice (Li et al., 2005). However, in another study, genistein was reported to enhance adipogenesis of human MSCs and suppressed their osteogenesis through regulating the expression of PPARγ (Zhang LY. et al., 2016). These contradictory results may be caused by the dose of genistein, at low concentrations (≦1 μM), genistein acted like estrogen, stimulating osteogenesis and inhibiting adipogenesis, whereas at high concentrations (>1 μM), genistein acted as a ligand of PPARγ, leading to up-regulation of adipogenesis and down-regulation of osteogenesis (Dang et al., 2003).
Other Flavonoids
In addition to the flavonoids mentioned above, other flavonoid extracts like baicalein (Ren et al., 2021), apigenin (Pan et al., 2021), amentoflavone (Zha et al., 2016), and anthocyanins (Saulite et al., 2019) have also been found to enhance osteogenic differentiation of MSCs. In human periodontal ligament cells (hPDLCs), baicalein induced osteogenic differentiation dose-dependently (1.25–10 μM) by activating the Wnt/β-catenin signaling pathway (Chen et al., 2017). Cyanidin-3-O-glucoside (C3G), the most common type of anthocyanin in nature, was shown to increase the expression of osteoblastic markers and osteoblast proliferation rate both in mouse MC3T3-E1 cells and human osteoblasts (extracted from the hip joint of patients with osteoporosis) by regulating ERK1/2 signaling pathway (Hu et al., 2021).
Inhibitory Effects of Flavonoids on Mesenchymal Stem Cells Osteogenic Differentiation
Although most studies showed that flavonoids promoted the osteogenic differentiation of MSC, some reports showed that flavonoids sometimes had an inhibitory effect on the osteogenic differentiation of MSC, and promoted adipogenesis (Hu et al., 2011; Zhang LY. et al., 2016; Casado-Díaz et al., 2016). Some flavonoids showed that they promoted the adipogenesis and inhibited osteogenesis of MSCs (Hu et al., 2011; Casado-Díaz et al., 2016; Lorthongpanich et al., 2021). Two isoprenylated flavonoids isolated from the twigs of Morus alba L (Moraceae; Morus alba L) significantly promoted adipogenesis and induced up-regulation of the expression of adipocyte-specific genes, aP2 and GLUT4 in 3T3L1 cells (Hu et al., 2011). In another study showed that high concentration of quercetin inhibited osteoblastic differentiation and promoted adipogenesis through Wnt/β-catenin inhibition. Which indicate such possible adverse effects of high use concentrations should be taken into account in nutraceutical or pharmaceutical strategies using flavonoids (Casado-Díaz et al., 2016).
The Applications of Flavonoids in Bone Disorders by Promoting Osteogenic Differentiation of Mesenchymal Stem Cells
The effects of flavonoids on bone defects had been extensively established using animal models (Yu et al., 2021; Zhao et al., 2021; Zhou and Xie, 2021). Flavonoids stimulated bone formation by increasing cell viability, matrix mineralization, calcium deposition, and up-regulation of osteogenic genes (Wu Y. et al., 2017; Preethi Soundarya et al., 2018). Meanwhile, flavonoids have great importance in treating bone disorders owing to their anti-inflammatory and anti-oxidative activities as we described above. Many flavonoids have been widely used in ovariectomized (OVX) osteoporotic, age-related osteoporotic models as well as glucocorticoid-induced osteoporosis, by regulating osteoblast-regulated bone formation and/or osteoclast-mediated bone resorption.
The impaired capability of osteogenic differentiation and senescence of MSCs are important pathogeneses of osteoporosis caused by various reasons (Jiang et al., 2021). In the aging process, as well as in glucocorticoid-induced osteoporosis, the ability of MSCs’ commitment towards the osteogenic lineage is impaired, while the adipogenesis is increased. Reactivating the osteogenic differentiation ability of MSCs is considered an important way to osteoporosis treatment. Icariin was effective in preventing postmenopausal osteoporosis through stimulating osteogenic differentiation of BMSCs (Wang et al., 2018), and it also protected against glucocorticoid-induced osteonecrosis of the femoral head in rats (Huang et al., 2018). Hesperetin alleviated glucocorticoid-induced inhibition of osteogenic differentiation through ERK signal pathway in BMSCs (Liu L. et al., 2021). Flavonoids also have great potential for the treatment of diabetes-induced osteoporosis owing to their anti-oxidative and adipogenesis inhibition activities (Nelson-Dooley et al., 2005; Kawser Hossain et al., 2016). Diabetes-induced osteoporosis is caused by chronic hyperglycemia, advanced glycated end products, and oxidative stress (Mohsin et al., 2019). In a rat model of diabetic osteoporosis, icariin could prevent diabetic osteoporosis by reducing blood glucose, inhibiting bone marrow adipogenesis, as well as up-regulation the expression of Runx2 and OPG (Qi et al., 2019).
MSC-based cellular therapy is a promising novel therapeutic strategy for osteonecrosis of the femoral head (ONFH). Flavonoids can increase bone formation in femoral heads by promoting MSCs proliferation and osteogenic differentiation. In methylprednisolone-induced rat ONFH models, the lithium chloride treatment group displayed a higher vessel volume and better trabecular structures as well as more OCN expression compared with methylprednisolone group, MSCs extracted from rats treated with lithium chloride had higher proliferative and osteogenic ability (Zhang Y. L. et al., 2021). Zefeng Yu et al (2016) also demonstrated lithium could enhance angiogenesis and stabilize osteogenic/adipogenic balance in glucocorticoid-induced ONFH rat models by activating the β-catenin pathway.
Combination Use of Flavonoids and Biomaterials in Bone Tissue Engineering
Mesenchymal stem cells combined with biological scaffold materials loaded with flavonoids are an excellent option for the application of flavonoids in the field of bone tissue engineering repair, and incorporation of flavonoids into biomaterials or scaffolds has been proved as a reliable technology for bone tissue regeneration, For example, the quercetin/silk fibroin/hydroxyapatite scaffolds with BMSCs increased the formation of new collagenous tissue and tissue ingrowth in a rat calvarial defect model, quercetin was found to promote cell proliferation and osteogenic differentiation of BMSC cultured in scaffolds in vitro (Song and Tripathy, 2018). Flavonoids also can stabilize collagen and inhibit its degradation in biological systems (Shavandi et al., 2018). The BMSCs-laden quercetin/collagen/hydroxyapatite sponge was proved as an alternative biomaterial for bone regeneration (Song et al., 2020). Kaempferol-immobilized titanium dioxide promotes the formation of new bone and is considered an effective tool for bone regeneration around dental implants (Tsuchiya et al., 2018). Promoting the proliferation and osteogenic differentiation of MSCs on scaffolds is the main role of flavonoids in the construction of bone tissue engineering. Besides, flavonoids enhance bone regeneration by counteracting the negative effect of oxidative stress on MSCs viability and differentiation (Forte et al., 2016; Chu et al., 2018). However, recent developments in bone tissue engineering focusing on flavonoids and their potent biological properties that enhance bone health has been well-reviewed (Preethi Soundarya et al., 2018). The potential of a combination of biomaterials loaded with flavonoids and MSCs might be enormous in bone tissue engineering.
Future Prospective
Extensive evidence showed the roles of flavonoids in regenerative and therapeutic medicine. Flavonoids as stimulants significantly affect the proliferation and osteogenic differentiation of MSCs. To further effectively screen and evaluate the application potential of flavonoids in bone tissue engineering and repair, it is very necessary to establish a standard and effective osteogenic differentiation protocol of MSC induced by flavonoids. Furthermore, the dose-effect relationship between MSCs and flavonoids should also be well established to achieve desired effects and reduce side effects. Given most flavonoids compounds are not having good solubility and low hydrophilicity, delivery systems, such as nanocarriers, with flavonoids are promising strategies for the improvement of cell uptake efficiency. In addition, MSCs combined with biological scaffold materials loaded with flavonoids are an excellent option for the application of flavonoids in the field of bone tissue engineering.
Conclusion
Flavonoids have a wide range of pharmacological activities and widely exist in nature. Flavonoids play a crucial role in the bone repair process not only through direct induction of osteoblastic differentiation, but also through their anti-inflammatory and anti-oxidant effects. MSCs combined with flavonoids are a promising alternative in stem cell therapy and bone tissue engineering construction. Flavonoids can help to increase proliferation and osteogenic differentiation of MSCs as well as modulate the microenvironment in the injured bone. To promote their clinical use, more works need to be done to improve their safety, efficacy, and quality, and to explore the mechanisms underlying their roles.
Author Contributions
JZ, YL, GH, and HC did literature retrieval and prepared the draft. JZ, ZL, and XL wrote the first revision of the manuscript. SQ and AL revised the manuscript.
Funding
This work was supported by grants from Guangzhou Municipal Science and Technology Project (No. 202002030049, 202102010058, 202102010060).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
- Abdallah B. M., Ali E. M. (2020). Butein Promotes Lineage Commitment of Bone Marrow-Derived Stem Cells into Osteoblasts via Modulating ERK1/2 Signaling Pathways. Molecules 25. 10.3390/molecules25081885 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmadi Z., Mohammadinejad R., Roomiani S., Afshar E. G., Ashrafizadeh M. (2021). Biological and Therapeutic Effects of Troxerutin: Molecular Signaling Pathways Come into View. J. Pharmacopuncture 24, 1–13. 10.3831/kpi.2021.24.1.1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amarowicz R., Carle R., Dongowski G., Durazzo A., Galensa R., Kammerer D., et al. (2009). Influence of Postharvest Processing and Storage on the Content of Phenolic Acids and Flavonoids in Foods. Mol. Nutr. Food Res. 53 (Suppl. 2), S151–S183. 10.1002/mnfr.200700486 [DOI] [PubMed] [Google Scholar]
- An J., Yang H., Zhang Q., Liu C., Zhao J., Zhang L., et al. (2016). Natural Products for Treatment of Osteoporosis: The Effects and Mechanisms on Promoting Osteoblast-Mediated Bone Formation. Life Sci. 147, 46–58. 10.1016/j.lfs.2016.01.024 [DOI] [PubMed] [Google Scholar]
- Armstrong J. P. K., Stevens M. M. (2019). Emerging Technologies for Tissue Engineering: From Gene Editing to Personalized Medicine. Tissue Eng. Part. A. 25, 688–692. 10.1089/ten.TEA.2019.0026 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babaei F., Mirzababaei M., Nassiri-Asl M. (2018). Quercetin in Food: Possible Mechanisms of its Effect on Memory. J. Food Sci. 83, 2280–2287. 10.1111/1750-3841.14317 [DOI] [PubMed] [Google Scholar]
- Baksh D., Song L., Tuan R. S. (2004). Adult Mesenchymal Stem Cells: Characterization, Differentiation, and Application in Cell and Gene Therapy. J. Cel Mol Med 8, 301–316. 10.1111/j.1582-4934.2004.tb00320.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bian W., Xiao S., Yang L., Chen J., Deng S. (2021). Quercetin Promotes Bone Marrow Mesenchymal Stem Cell Proliferation and Osteogenic Differentiation through the H19/miR-625-5p axis to Activate the Wnt/β-Catenin Pathway. BMC Complement. Med. Ther. 21, 243. 10.1186/s12906-021-03418-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao X., Lin W., Liang C., Zhang D., Yang F., Zhang Y., et al. (2015). Naringin Rescued the TNF-α-Induced Inhibition of Osteogenesis of Bone Marrow-Derived Mesenchymal Stem Cells by Depressing the Activation of NF-Кb Signaling Pathway. Immunol. Res. 62, 357–367. 10.1007/s12026-015-8665-x [DOI] [PubMed] [Google Scholar]
- Casado-Díaz A., Rodríguez-Ramos Á. (2021). Flavonoid Phloretin Inhibits Adipogenesis and Increases OPG Expression in Adipocytes Derived from Human Bone-Marrow Mesenchymal Stromal-Cells. Nutrients 13, 4185. 10.3390/nu13114185 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casado-Díaz A., Anter J., Dorado G., Quesada-Gómez J. M. (2016). Effects of Quercetin, a Natural Phenolic Compound, in the Differentiation of Human Mesenchymal Stem Cells (MSC) into Adipocytes and Osteoblasts. J. Nutr. Biochem. 32, 151–162. 10.1016/j.jnutbio.2016.03.005 [DOI] [PubMed] [Google Scholar]
- Chandran S. V., Vairamani M., Selvamurugan N. (2019). Osteostimulatory Effect of Biocomposite Scaffold Containing Phytomolecule Diosmin by Integrin/FAK/ERK Signaling Pathway in Mouse Mesenchymal Stem Cells. Sci. Rep. 9, 11900. 10.1038/s41598-019-48429-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang H., Lei L., Zhou Y., Ye F., Zhao G. (2018). Dietary Flavonoids and the Risk of Colorectal Cancer: An Updated Meta-Analysis of Epidemiological Studies. Nutrients 10, 950. 10.3390/nu10070950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen C. H., Ho M. L., Chang J. K., Hung S. H., Wang G. J. (2005). Green tea Catechin Enhances Osteogenesis in a Bone Marrow Mesenchymal Stem Cell Line. Osteoporos. Int. 16, 2039–2045. 10.1007/s00198-005-1995-0 [DOI] [PubMed] [Google Scholar]
- Chen L. J., Hu B. B., Shi X. L., Ren M. M., Yu W. B., Cen S. D., et al. (2017). Baicalein Enhances the Osteogenic Differentiation of Human Periodontal Ligament Cells by Activating the Wnt/β-Catenin Signaling Pathway. Arch. Oral Biol. 78, 100–108. 10.1016/j.archoralbio.2017.01.019 [DOI] [PubMed] [Google Scholar]
- Chu J., Shi P., Yan W., Fu J., Yang Z., He C., et al. (2018). PEGylated Graphene Oxide-Mediated Quercetin-Modified Collagen Hybrid Scaffold for Enhancement of MSCs Differentiation Potential and Diabetic Wound Healing. Nanoscale 10, 9547–9560. 10.1039/c8nr02538j [DOI] [PubMed] [Google Scholar]
- Dabeek W. M., Marra M. V. (2019). Dietary Quercetin and Kaempferol: Bioavailability and Potential Cardiovascular-Related Bioactivity in Humans. Nutrients 11, 2288. 10.3390/nu11102288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai J., Li Y., Zhou H., Chen J., Chen M., Xiao Z. (2013). Genistein Promotion of Osteogenic Differentiation through BMP2/SMAD5/RUNX2 Signaling. Int. J. Biol. Sci. 9, 1089–1098. 10.7150/ijbs.7367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dang Z. C., Audinot V., Papapoulos S. E., Boutin J. A., Löwik C. W. (2003). Peroxisome Proliferator-Activated Receptor Gamma (PPARgamma ) as a Molecular Target for the Soy Phytoestrogen Genistein. J. Biol. Chem. 278, 962–967. 10.1074/jbc.M209483200 [DOI] [PubMed] [Google Scholar]
- Den Hartogh D. J., Tsiani E. (2019). Antidiabetic Properties of Naringenin: A Citrus Fruit Polyphenol. Biomolecules 9, 99. 10.3390/biom9030099 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng Z., Chen J., Lin B., Li J., Wang H., Wang D., et al. (2020). A Novel 3D Printed Bioactive Scaffolds with Enhanced Osteogenic Inspired by Ancient Chinese Medicine HYSA for Bone Repair. Exp. Cel Res 394, 112139. 10.1016/j.yexcr.2020.112139 [DOI] [PubMed] [Google Scholar]
- Dimitriou R., Jones E., Mcgonagle D., Giannoudis P. V. (2011). Bone Regeneration: Current Concepts and Future Directions. BMC Med. 9, 66. 10.1186/1741-7015-9-66 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong G. C., Ma T. Y., Li C. H., Chi C. Y., Su C. M., Huang C. L., et al. (2020). A Study of Drynaria Fortunei in Modulation of BMP–2 Signalling by Bone Tissue Engineering. Turk J. Med. Sci. 50, 1444–1453. 10.3906/sag-2001-148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forte L., Torricelli P., Boanini E., Gazzano M., Rubini K., Fini M., et al. (2016). Antioxidant and Bone Repair Properties of Quercetin-Functionalized Hydroxyapatite: An In Vitro Osteoblast-Osteoclast-Endothelial Cell Co-culture Study. Acta Biomater. 32, 298–308. 10.1016/j.actbio.2015.12.013 [DOI] [PubMed] [Google Scholar]
- Friedenstein A. J. (1976). Precursor Cells of Mechanocytes. Int. Rev. Cytol. 47, 327–359. 10.1016/s0074-7696(08)60092-3 [DOI] [PubMed] [Google Scholar]
- Friedman M. (2007). Overview of Antibacterial, Antitoxin, Antiviral, and Antifungal Activities of tea Flavonoids and Teas. Mol. Nutr. Food Res. 51, 116–134. 10.1002/mnfr.200600173 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu S., Yang L., Hong H., Zhang R. (2016). [Wnt/β-catenin Signaling Is Involved in the Icariin Induced Proliferation of Bone Marrow Mesenchymal Stem Cells]. J. Tradit Chin. Med. 36, 360–368. 10.1016/s0254-6272(16)30050-4 [DOI] [PubMed] [Google Scholar]
- Gao A. G., Zhou Y. C., Hu Z. J., Lu B. B. (2018). Ipriflavone Promotes Osteogenesis of MSCs Derived from Osteoporotic Rats. Eur. Rev. Med. Pharmacol. Sci. 22, 4669–4676. 10.26355/eurrev_201807_15527 [DOI] [PubMed] [Google Scholar]
- Gao J., Xiang S., Wei X., Yadav R. I., Han M., Zheng W., et al. (2021). Icariin Promotes the Osteogenesis of Bone Marrow Mesenchymal Stem Cells through Regulating Sclerostin and Activating the Wnt/β-Catenin Signaling Pathway. Biomed. Res. Int. 2021, 6666836. 10.1155/2021/6666836 [DOI] [PMC free article] [PubMed] [Google Scholar]
- García-Sánchez D., Fernández D., Rodríguez-Rey J. C., Pérez-Campo F. M. (2019). Enhancing Survival, Engraftment, and Osteogenic Potential of Mesenchymal Stem Cells. World J. Stem Cell 11, 748–763. 10.4252/wjsc.v11.i10.748 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garmany A., Yamada S., Terzic A. (2021). Longevity Leap: Mind the Healthspan gap. Npj Regen. Med. 6, 57. 10.1038/s41536-021-00169-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ge X., Zhou G. (2021). Protective Effects of Naringin on Glucocorticoid-Induced Osteoporosis through Regulating the PI3K/Akt/mTOR Signaling Pathway. Am. J. Transl Res. 13, 6330–6341. [PMC free article] [PubMed] [Google Scholar]
- Guo L. T., Wang S. Q., Su J., Xu L. X., Ji Z. Y., Zhang R. Y., et al. (2019). Baicalin Ameliorates Neuroinflammation-Induced Depressive-like Behavior through Inhibition of Toll-like Receptor 4 Expression via the PI3K/AKT/FoxO1 Pathway. J. Neuroinflammation 16, 95. 10.1186/s12974-019-1474-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gupta S. K., Kumar R., Mishra N. C. (2017). Influence of Quercetin and Nanohydroxyapatite Modifications of Decellularized Goat-Lung Scaffold for Bone Regeneration. Mater. Sci. Eng. C Mater. Biol. Appl. 71, 919–928. 10.1016/j.msec.2016.10.085 [DOI] [PubMed] [Google Scholar]
- Heim M., Frank O., Kampmann G., Sochocky N., Pennimpede T., Fuchs P., et al. (2004). The Phytoestrogen Genistein Enhances Osteogenesis and Represses Adipogenic Differentiation of Human Primary Bone Marrow Stromal Cells. Endocrinology 145, 848–859. 10.1210/en.2003-1014 [DOI] [PubMed] [Google Scholar]
- Heo J. S., Lee S. G. (2017). The Flavonoid Glabridin Induces OCT4 to Enhance Osteogenetic Potential in Mesenchymal Stem Cells. Stem Cell Int. 2017, 6921703. 10.1155/2017/6921703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu B., Chen L., Chen Y., Zhang Z., Wang X., Zhou B. (2021). Cyanidin-3-glucoside Regulates Osteoblast Differentiation via the ERK1/2 Signaling Pathway. ACS Omega 6, 4759–4766. 10.1021/acsomega.0c05603 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu X., Ji J., Wang M., Wu J. W., Zhao Q. S., Wang H. Y., et al. (2011). New Isoprenylated Flavonoids and Adipogenesis-Promoting Constituents from Morus Notabilis. Bioorg. Med. Chem. Lett. 21, 4441–4446. 10.1016/j.bmcl.2011.06.020 [DOI] [PubMed] [Google Scholar]
- Huang K. H., Chen C. Y., Chang C. Y., Chen Y. W., Lin C. P. (2021). The Synergistic Effects of Quercetin-Containing 3D-Printed Mesoporous Calcium Silicate/calcium Sulfate/poly-ε-Caprolactone Scaffolds for the Promotion of Osteogenesis in Mesenchymal Stem Cells. J. Formos. Med. Assoc. 120, 1627–1634. 10.1016/j.jfma.2021.01.024 [DOI] [PubMed] [Google Scholar]
- Huang T., Liu Y., Zhang C. (2019). Pharmacokinetics and Bioavailability Enhancement of Baicalin: A Review. Eur. J. Drug Metab. Pharmacokinet. 44, 159–168. 10.1007/s13318-018-0509-3 [DOI] [PubMed] [Google Scholar]
- Huang Z., Cheng C., Cao B., Wang J., Wei H., Liu X., et al. (2018). Icariin Protects against Glucocorticoid-Induced Osteonecrosis of the Femoral Head in Rats. Cell Physiol Biochem 47, 694–706. 10.1159/000490023 [DOI] [PubMed] [Google Scholar]
- Hwang D., Kang M. J., Kang C. W., Kim G. D. (2019). Kaempferol-3-O-β-rutinoside Suppresses the Inflammatory Responses in Lipopolysaccharide-Stimulated RAW264.7 Cells via the NF-Κb and MAPK Pathways. Int. J. Mol. Med. 44, 2321–2328. 10.3892/ijmm.2019.4381 [DOI] [PubMed] [Google Scholar]
- Jaiswal N., Akhtar J., Singh S. P., Ahsan F., Ahsan F. (2019). An Overview on Genistein and its Various Formulations. Drug Res. (Stuttg) 69, 305–313. 10.1055/a-0797-3657 [DOI] [PubMed] [Google Scholar]
- Jiang Y., Zhang P., Zhang X., Lv L. (2021). Advances in Mesenchymal Stem Cell Transplantation for the Treatment of Osteoporosis. Cel Prolif. 54, e12956. 10.1111/cpr.12956 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin P., Li M., Xu G., Zhang K., Zheng L., Zhao J. (2021). Erratum: Role of (-)-Epigallocatechin-3-Gallate in the Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells: An Enhancer or an Inducer? Exp. Ther. Med. 21, 294. 10.3892/etm.2021.9725 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jing X., Yin W., Tian H., Chen M., Yao X., Zhu W., et al. (2018). Icariin Doped Bioactive Glasses Seeded with Rat Adipose-Derived Stem Cells to Promote Bone Repair via Enhanced Osteogenic and Angiogenic Activities. Life Sci. 202, 52–60. 10.1016/j.lfs.2018.02.026 [DOI] [PubMed] [Google Scholar]
- Kashyap D., Garg V. K., Tuli H. S., Yerer M. B. (2019). Fisetin and Quercetin: Promising Flavonoids with Chemopreventive Potential. Biomolecules 9, 174. 10.3390/biom9050174 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawser Hossain M., Abdal Dayem A., Han J., Yin Y., Kim K., Kumar Saha S., et al. (2016). Molecular Mechanisms of the Anti-obesity and Anti-diabetic Properties of Flavonoids. Int. J. Mol. Sci. 17, 569. 10.3390/ijms17040569 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S. Y., Lee J. Y., Park Y. D., Kang K. L., Lee J. C., Heo J. S. (2013). Hesperetin Alleviates the Inhibitory Effects of High Glucose on the Osteoblastic Differentiation of Periodontal Ligament Stem Cells. PLoS One 8, e67504. 10.1371/journal.pone.0067504 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim Y. J., Bae Y. C., Suh K. T., Jung J. S. (2006). Quercetin, a Flavonoid, Inhibits Proliferation and Increases Osteogenic Differentiation in Human Adipose Stromal Cells. Biochem. Pharmacol. 72, 1268–1278. 10.1016/j.bcp.2006.08.021 [DOI] [PubMed] [Google Scholar]
- Kook Y. J., Tian J., Jeon Y. S., Choi M. J., Song J. E., Park C. H., et al. (2018). Nature-derived Epigallocatechin Gallate/duck's Feet Collagen/hydroxyapatite Composite Sponges for Enhanced Bone Tissue Regeneration. J. Biomater. Sci. Polym. Ed. 29, 984–996. 10.1080/09205063.2017.1414480 [DOI] [PubMed] [Google Scholar]
- Kumar S., Pandey A. K. (2013). Chemistry and Biological Activities of Flavonoids: an Overview. ScientificWorldJournal 2013, 162750. 10.1155/2013/162750 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavrador P., Gaspar V. M., Mano J. F. (2018). Bioinspired Bone Therapies Using Naringin: Applications and Advances. Drug Discov. Today 23, 1293–1304. 10.1016/j.drudis.2018.05.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J., Wang X., Wang Y., Lu C., Zheng D., Zhang J. (2019). Isoquercitrin, a Flavonoid Glucoside, Exerts a Positive Effect on Osteogenesis In Vitro and In Vivo . Chem. Biol. Interact 297, 85–94. 10.1016/j.cbi.2018.10.018 [DOI] [PubMed] [Google Scholar]
- Li M., Zhang C., Li X., Lv Z., Chen Y., Zhao J. (2019). Isoquercitrin Promotes the Osteogenic Differentiation of Osteoblasts and BMSCs via the RUNX2 or BMP Pathway. Connect. Tissue Res. 60, 189–199. 10.1080/03008207.2018.1483358 [DOI] [PubMed] [Google Scholar]
- Li X., Chen R., Lei X., Wang P., Zhu X., Zhang R., et al. (2021). Quercetin Regulates ERα Mediated Differentiation of BMSCs through Circular RNA. Gene 769, 145172. 10.1016/j.gene.2020.145172 [DOI] [PubMed] [Google Scholar]
- Li X., Peng B., Pan Y., Wang P., Sun K., Lei X., et al. (2018). Icariin Stimulates Osteogenic Differentiation and Suppresses Adipogenic Differentiation of rBMSCs via Estrogen Receptor Signaling. Mol. Med. Rep. 18, 3483–3489. 10.3892/mmr.2018.9325 [DOI] [PubMed] [Google Scholar]
- Li X. H., Zhang J. C., Sui S. F., Yang M. S. (2005). Effect of Daidzin, Genistin, and Glycitin on Osteogenic and Adipogenic Differentiation of Bone Marrow Stromal Cells and Adipocytic Transdifferentiation of Osteoblasts. Acta Pharmacol. Sin 26, 1081–1086. 10.1111/j.1745-7254.2005.00161.x [DOI] [PubMed] [Google Scholar]
- Li Y., Wang J., Chen G., Feng S., Wang P., Zhu X., et al. (2015). Quercetin Promotes the Osteogenic Differentiation of Rat Mesenchymal Stem Cells via Mitogen-Activated Protein Kinase Signaling. Exp. Ther. Med. 9, 2072–2080. 10.3892/etm.2015.2388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang X., Hou Z., Xie Y., Yan F., Li S., Zhu X., et al. (2019). Icariin Promotes Osteogenic Differentiation of Bone Marrow Stromal Cells and Prevents Bone Loss in OVX Mice via Activating Autophagy. J. Cel Biochem 120, 13121–13132. 10.1002/jcb.28585 [DOI] [PubMed] [Google Scholar]
- Lin F. X., Zheng G. Z., Chang B., Chen R. C., Zhang Q. H., Xie P., et al. (2018). Connexin 43 Modulates Osteogenic Differentiation of Bone Marrow Stromal Cells through GSK-3beta/Beta-Catenin Signaling Pathways. Cel Physiol Biochem 47, 161–175. 10.1159/000489763 [DOI] [PubMed] [Google Scholar]
- Lin S. Y., Kang L., Chen J. C., Wang C. Z., Huang H. H., Lee M. J., et al. (2019). (-)-Epigallocatechin-3-gallate (EGCG) Enhances Healing of Femoral Bone Defect. Phytomedicine 55, 165–171. 10.1016/j.phymed.2018.07.012 [DOI] [PubMed] [Google Scholar]
- Lin S. Y., Kang L., Wang C. Z., Huang H. H., Cheng T. L. (2018). Epigallocatechin-3-Gallate (EGCG) Enhances Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells. Molecules 23, 3221. 10.3390/molecules23123221 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu H., Yi X., Tu S., Cheng C., Luo J. (2021). Kaempferol Promotes BMSC Osteogenic Differentiation and Improves Osteoporosis by Downregulating miR-10a-3p and Upregulating CXCL12. Mol. Cel Endocrinol 520, 111074. 10.1016/j.mce.2020.111074 [DOI] [PubMed] [Google Scholar]
- Liu L., Zheng J., Yang Y., Ni L., Chen H., Yu D. (2021). Hesperetin Alleviated Glucocorticoid-Induced Inhibition of Osteogenic Differentiation of BMSCs through Regulating the ERK Signaling Pathway. Med. Mol. Morphol. 54, 1–7. 10.1007/s00795-020-00251-9 [DOI] [PubMed] [Google Scholar]
- Liu M., Li Y., Yang S. T. (2017). Effects of Naringin on the Proliferation and Osteogenic Differentiation of Human Amniotic Fluid-Derived Stem Cells. J. Tissue Eng. Regen. Med. 11, 276–284. 10.1002/term.1911 [DOI] [PubMed] [Google Scholar]
- Liu W., Fan J. B., Xu D. W., Zhang J., Cui Z. M. (2016). Epigallocatechin-3-gallate Protects against Tumor Necrosis Factor Alpha Induced Inhibition of Osteogenesis of Mesenchymal Stem Cells. Exp. Biol. Med. (Maywood) 241, 658–666. 10.1177/1535370215624020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Z., Lin Y., Fang X., Yang J., Chen Z. (2021c). Epigallocatechin-3-Gallate Promotes Osteo-/Odontogenic Differentiation of Stem Cells from the Apical Papilla through Activating the BMP-Smad Signaling Pathway. Molecules 26, 1580. 10.3390/molecules26061580 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorthongpanich C., Charoenwongpaiboon T. (2021). Fisetin Inhibits Osteogenic Differentiation of Mesenchymal Stem Cells via the Inhibition of YAP. Antioxidants 10, 879. 10.3390/antiox10060879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorthongpanich C., Charoenwongpaiboon T., Supakun P., Klaewkla M., Kheolamai P., Issaragrisil S. (2021). Fisetin Inhibits Osteogenic Differentiation of Mesenchymal Stem Cells via the Inhibition of YAP. Antioxidants 10, 879. 10.3390/antiox10060879 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo G., Gu F., Zhang Y., Liu T., Guo P., Huang Y. (2015). Icariside II Promotes Osteogenic Differentiation of Bone Marrow Stromal Cells in Beagle Canine. Int. J. Clin. Exp. Pathol. 8, 4367–4377. [PMC free article] [PubMed] [Google Scholar]
- Martens S., Mithöfer A. (2005). Flavones and Flavone Synthases. Phytochemistry 66, 2399–2407. 10.1016/j.phytochem.2005.07.013 [DOI] [PubMed] [Google Scholar]
- Miller P. D. (2016). Management of Severe Osteoporosis. Expert Opin. Pharmacother. 17, 473–488. 10.1517/14656566.2016.1124856 [DOI] [PubMed] [Google Scholar]
- Mohsin S., Baniyas M. M., Aldarmaki R. S., Tekes K., Kalász H., Adeghate E. A. (2019). An Update on Therapies for the Treatment of Diabetes-Induced Osteoporosis. Expert Opin. Biol. Ther. 19, 937–948. 10.1080/14712598.2019.1618266 [DOI] [PubMed] [Google Scholar]
- Nan L. P., Wang F., Ran D., Zhou S. F., Liu Y., Zhang Z., et al. (2020). Naringin Alleviates H2O2-Induced Apoptosis via the PI3K/Akt Pathway in Rat Nucleus Pulposus-Derived Mesenchymal Stem Cells. Connect. Tissue Res. 61, 554–567. 10.1080/03008207.2019.1631299 [DOI] [PubMed] [Google Scholar]
- Nauth A., Schemitsch E., Norris B., Nollin Z., Watson J. T. (2018). Critical-Size Bone Defects: Is There a Consensus for Diagnosis and Treatment? J. Orthop. Trauma 32 (Suppl. 1), S7–s11. 10.1097/BOT.0000000000001115 [DOI] [PubMed] [Google Scholar]
- Nelson-Dooley C., Della-Fera M. A., Hamrick M., Baile C. A. (2005). Novel Treatments for Obesity and Osteoporosis: Targeting Apoptotic Pathways in Adipocytes. Curr. Med. Chem. 12, 2215–2225. 10.2174/0929867054864886 [DOI] [PubMed] [Google Scholar]
- Nie F., Zhang W., Cui Q., Fu Y., Li H., Zhang J. (2020). Kaempferol Promotes Proliferation and Osteogenic Differentiation of Periodontal Ligament Stem Cells via Wnt/β-Catenin Signaling Pathway. Life Sci. 258, 118143. 10.1016/j.lfs.2020.118143 [DOI] [PubMed] [Google Scholar]
- Nishioku T., Kubo T., Kamada T., Okamoto K., Tsukuba T., Uto T., et al. (2020). (−)-Epigallocatechin-3-gallate Inhibits RANKL-Induced Osteoclastogenesis via Downregulation of NFATc1 and Suppression of HO-1-HMGB1-RAGE pathwayEpigallocatechin-3-Gallate Inhibits RANKL-Induced Osteoclastogenesis via Downregulation of NFATc1 and Suppression of HO-1-HMGB1-RAGE Pathway. Biomed. Res. 41, 269–277. 10.2220/biomedres.41.269 [DOI] [PubMed] [Google Scholar]
- Oh J. H., Karadeniz F., Seo Y., Kong C. S. (2020). Effect of Quercetin 3-O-β-D-Galactopyranoside on the Adipogenic and Osteoblastogenic Differentiation of Human Bone Marrow-Derived Mesenchymal Stromal Cells. Int. J. Mol. Sci. 21, 8044. 10.3390/ijms21218044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pan F. F., Shao J., Shi C. J., Li Z. P., Fu W. M., Zhang J. F. (2021). Apigenin Promotes Osteogenic Differentiation of Mesenchymal Stem Cells and Accelerates Bone Fracture Healing via Activating Wnt/β-Catenin Signaling. Am. J. Physiol. Endocrinol. Metab. 320, E760–e771. 10.1152/ajpendo.00543.2019 [DOI] [PubMed] [Google Scholar]
- Pang X. G., Cong Y., Bao N. R. (2018). Quercetin Stimulates Bone Marrow Mesenchymal Stem Cell Differentiation through an Estrogen Receptor-Mediated Pathway. Biomed. Res. Int. 2018, 4178021. 10.1155/2018/4178021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel H. B., Lyerly L. J., Horlen C. K. (2021). The Role of Novel Bone Forming Agents in the Treatment of Osteoporosis. J. Pharm. Pract. 34, 952–961. 10.1177/0897190020961227 [DOI] [PubMed] [Google Scholar]
- Pietta P. G. (2000). Flavonoids as Antioxidants. J. Nat. Prod. 63, 1035–1042. 10.1021/np9904509 [DOI] [PubMed] [Google Scholar]
- Preethi Soundarya S., Sanjay V., Haritha Menon A., Dhivya S., Selvamurugan N. (2018). Effects of Flavonoids Incorporated Biological Macromolecules Based Scaffolds in Bone Tissue Engineering. Int. J. Biol. Macromol 110, 74–87. 10.1016/j.ijbiomac.2017.09.014 [DOI] [PubMed] [Google Scholar]
- Qi S., He J., Zheng H., Chen C., Lan S. (2019). Icariin Prevents Diabetes-Induced Bone Loss in Rats by Reducing Blood Glucose and Suppressing Bone Turnover. Molecules 24, 1871. 10.3390/molecules24101871 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qiu Y., Chen Y., Zeng T., Guo W., Zhou W., Yang X. (2016). EGCG Ameliorates the Hypoxia-Induced Apoptosis and Osteogenic Differentiation Reduction of Mesenchymal Stem Cells via Upregulating miR-210. Mol. Biol. Rep. 43, 183–193. 10.1007/s11033-015-3936-0 [DOI] [PubMed] [Google Scholar]
- Qiu Z. C., Zhang Y., Xiao H. H., Chui-Wa Poon C., Li X. L., Cui J. F., et al. (2020). 8-prenylgenistein Exerts Osteogenic Effects via ER α and Wnt-dependent Signaling Pathway. Exp. Cel Res 395, 112186. 10.1016/j.yexcr.2020.112186 [DOI] [PubMed] [Google Scholar]
- Ren M., Zhao Y., He Z., Lin J., Xu C., Liu F., et al. (2021). Baicalein Inhibits Inflammatory Response and Promotes Osteogenic Activity in Periodontal Ligament Cells Challenged with Lipopolysaccharides. BMC Complement. Med. Ther. 21, 43. 10.1186/s12906-021-03213-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saulite L., Jekabsons K., Klavins M., Muceniece R., Riekstina U. (2019). Effects of Malvidin, Cyanidin and Delphinidin on Human Adipose Mesenchymal Stem Cell Differentiation into Adipocytes, Chondrocytes and Osteocytes. Phytomedicine 53, 86–95. 10.1016/j.phymed.2018.09.029 [DOI] [PubMed] [Google Scholar]
- Schilling T., Ebert R., Raaijmakers N., Schütze N., Jakob F. (2014). Effects of Phytoestrogens and Other Plant-Derived Compounds on Mesenchymal Stem Cells, Bone Maintenance and Regeneration. J. Steroid Biochem. Mol. Biol. 139, 252–261. 10.1016/j.jsbmb.2012.12.006 [DOI] [PubMed] [Google Scholar]
- Sharma A. R., Nam J. S. (2019). Kaempferol Stimulates WNT/β-catenin Signaling Pathway to Induce Differentiation of Osteoblasts. J. Nutr. Biochem. 74, 108228. 10.1016/j.jnutbio.2019.108228 [DOI] [PubMed] [Google Scholar]
- Shavandi A., Bekhit A. E. A., Saeedi P., Izadifar Z., Bekhit A. A., Khademhosseini A. (2018). Polyphenol Uses in Biomaterials Engineering. Biomaterials 167, 91–106. 10.1016/j.biomaterials.2018.03.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song J. E., Tian J., Kook Y. J., Thangavelu M., Choi J. H., Khang G. (2020). A BMSCs-Laden Quercetin/duck's Feet Collagen/hydroxyapatite Sponge for Enhanced Bone Regeneration. J. Biomed. Mater. Res. A. 108, 784–794. 10.1002/jbm.a.36857 [DOI] [PubMed] [Google Scholar]
- Song J. E., Tripathy N., Lee D. H., Park J. H., Khang G. (2018). Quercetin Inlaid Silk Fibroin/Hydroxyapatite Scaffold Promotes Enhanced Osteogenesis. ACS Appl. Mater. Inter. 10, 32955–32964. 10.1021/acsami.8b08119 [DOI] [PubMed] [Google Scholar]
- Tian X., Jiang H., Chen Y., Ao X., Chen C., Zhang W., et al. (2018). Baicalein Accelerates Tendon-Bone Healing via Activation of Wnt/β-Catenin Signaling Pathway in Rats. Biomed. Res. Int., 2018, 3849760. 10.1155/2018/3849760 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuchiya S., Sugimoto K., Kamio H., Okabe K., Kuroda K., Okido M., et al. (2018). Kaempferol-immobilized Titanium Dioxide Promotes Formation of New Bone: Effects of Loading Methods on Bone Marrow Stromal Cell Differentiation In Vivo and In Vitro . Int. J. Nanomedicine 13, 1665–1676. 10.2147/IJN.S150786 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tutunchi H., Naeini F., Ostadrahimi A., Hosseinzadeh-Attar M. J. (2020). Naringenin, a Flavanone with Antiviral and Anti-inflammatory Effects: A Promising Treatment Strategy against COVID-19. Phytother Res. 34, 3137–3147. 10.1002/ptr.6781 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vadaye Kheiry E., Parivar K., Baharara J., Fazly Bazzaz B. S., Iranbakhsh A. (2018). The Osteogenesis of Bacterial Cellulose Scaffold Loaded with Fisetin. Iran J. Basic Med. Sci. 21, 965–971. 10.22038/IJBMS.2018.25465.6296 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velasco M. A., Narváez-Tovar C. A., Garzón-Alvarado D. A. (2015). Design, Materials, and Mechanobiology of Biodegradable Scaffolds for Bone Tissue Engineering. Biomed. Res. Int. 2015, 729076. 10.1155/2015/729076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan T., Wu M. R., Qi Z. X. (2014). [Effect of Hydroxy Safflower Yellow A on Glucocorticoid-Induced Bone Marrow Mesenchymal Stem Cells Osteogenic Differentiation]. Zhongguo Gu Shang 27, 224–228. [PubMed] [Google Scholar]
- Wang D., Wang Y., Xu S., Wang F., Wang B., Han K., et al. (2016). Epigallocatechin-3-gallate Protects against Hydrogen Peroxide-Induced Inhibition of Osteogenic Differentiation of Human Bone Marrow-Derived Mesenchymal Stem Cells. Stem Cell Int 2016, 7532798. 10.1155/2016/7532798 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H., Li C., Li J., Zhu Y., Jia Y., Zhang Y., et al. (2017). Naringin Enhances Osteogenic Differentiation through the Activation of ERK Signaling in Human Bone Marrow Mesenchymal Stem Cells. Iran J. Basic Med. Sci. 20, 408–414. 10.22038/IJBMS.2017.8582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J., Tao Y., Ping Z., Zhang W., Hu X., Wang Y., et al. (2016). Icariin Attenuates Titanium-Particle Inhibition of Bone Formation by Activating the Wnt/β-Catenin Signaling Pathway In Vivo and In Vitro . Sci. Rep. 6, 23827. 10.1038/srep23827 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang W., Mao J., Chen Y., Zuo J., Chen L., Li Y., et al. (2022). Naringin Promotes Osteogenesis and Ameliorates Osteoporosis Development by Targeting JAK2/STAT3 Signalling. Clin. Exp. Pharmacol. Physiol. 49, 113–121. 10.1111/1440-1681.13591 [DOI] [PubMed] [Google Scholar]
- Wang Y. J., Zhang H. Q., Han H. L., Zou Y. Y., Gao Q. L., Yang G. T. (2017). Taxifolin Enhances Osteogenic Differentiation of Human Bone Marrow Mesenchymal Stem Cells Partially via NF-Κb Pathway. Biochem. Biophys. Res. Commun. 490, 36–43. 10.1016/j.bbrc.2017.06.002 [DOI] [PubMed] [Google Scholar]
- Wang Z., Wang D., Yang D., Zhen W., Zhang J., Peng S. (2018). The Effect of Icariin on Bone Metabolism and its Potential Clinical Application. Osteoporos. Int. 29, 535–544. 10.1007/s00198-017-4255-1 [DOI] [PubMed] [Google Scholar]
- Wei Q., He M., Chen M., Chen Z., Yang F., Wang H., et al. (2017). Icariin Stimulates Osteogenic Differentiation of Rat Bone Marrow Stromal Stem Cells by Increasing TAZ Expression. Biomed. Pharmacother. 91, 581–589. 10.1016/j.biopha.2017.04.019 [DOI] [PubMed] [Google Scholar]
- Wei Q., Wang B., Hu H., Xie C., Ling L., Gao J., et al. (2020). Icaritin Promotes the Osteogenesis of Bone Marrow Mesenchymal Stem Cells via the Regulation of Sclerostin Expression. Int. J. Mol. Med. 45, 816–824. 10.3892/ijmm.2020.4470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen L., Jiang Y., Yang J., Zhao Y., Tian M., Yang B. (2017). Structure, Bioactivity, and Synthesis of Methylated Flavonoids. Ann. N. Y Acad. Sci. 1398, 120–129. 10.1111/nyas.13350 [DOI] [PubMed] [Google Scholar]
- Williams J., Allen L., Wickramasinghe K., Mikkelsen B., Roberts N., Townsend N. (2018). A Systematic Review of Associations between Non-communicable Diseases and Socioeconomic Status within Low- and Lower-Middle-Income Countries. J. Glob. Health 8, 020409. 10.7189/jogh.08.020409 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu T., Shu T., Kang L., Wu J., Xing J., Lu Z., et al. (2017a). Icaritin, a Novel Plant-Derived Osteoinductive Agent, Enhances the Osteogenic Differentiation of Human Bone Marrow- and Human Adipose Tissue-Derived Mesenchymal Stem Cells. Int. J. Mol. Med. 39, 984–992. 10.3892/ijmm.2017.2906 [DOI] [PubMed] [Google Scholar]
- Wu Y., Cao L., Xia L., Wu Q., Wang J., Wang X., et al. (2017b). Evaluation of Osteogenesis and Angiogenesis of Icariin in Local Controlled Release and Systemic Delivery for Calvarial Defect in Ovariectomized Rats. Sci. Rep. 7, 5077. 10.1038/s41598-017-05392-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu H., Zhou S., Qu R., Yang Y., Gong X., Hong Y., et al. (2020). Icariin Prevents Oestrogen Deficiency-Induced Alveolar Bone Loss through Promoting Osteogenesis via STAT3. Cell Prolif 53, e12743. 10.1111/cpr.12743 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu Y., Jiang Y., Jia B., Wang Y., Li T. (2021). Icariin Stimulates Osteogenesis and Suppresses Adipogenesis of Human Bone Mesenchymal Stem Cells via miR-23a-Mediated Activation of the Wnt/β-Catenin Signaling Pathway. Phytomedicine 85, 153485. 10.1016/j.phymed.2021.153485 [DOI] [PubMed] [Google Scholar]
- Xue D., Chen E., Zhang W., Gao X., Wang S., Zheng Q., et al. (2017). The Role of Hesperetin on Osteogenesis of Human Mesenchymal Stem Cells and its Function in Bone Regeneration. Oncotarget 8, 21031–21043. 10.18632/oncotarget.15473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang J., Zhang Y. S., Yue K., Khademhosseini A. (2017). Cell-laden Hydrogels for Osteochondral and Cartilage Tissue Engineering. Acta Biomater. 57, 1–25. 10.1016/j.actbio.2017.01.036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang X., Shao J., Wu X. M., Pan F. F., Yang S. A., Pan X. H., et al. (2021). Troxerutin Stimulates Osteoblast Differentiation of Mesenchymal Stem Cell and Facilitates Bone Fracture Healing. Front. Pharmacol. 12, 723145. 10.3389/fphar.2021.723145 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yao X., Jing X., Guo J., Sun K., Deng Y., Zhang Y., et al. (2019). Icariin Protects Bone Marrow Mesenchymal Stem Cells against Iron Overload Induced Dysfunction through Mitochondrial Fusion and Fission, PI3K/AKT/mTOR and MAPK Pathways. Front. Pharmacol. 10, 163. 10.3389/fphar.2019.00163 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ye Y., Jing X., Li N., Wu Y., Li B., Xu T. (2017). Icariin Promotes Proliferation and Osteogenic Differentiation of Rat Adipose-Derived Stem Cells by Activating the RhoA-TAZ Signaling Pathway. Biomed. Pharmacother. 88, 384–394. 10.1016/j.biopha.2017.01.075 [DOI] [PubMed] [Google Scholar]
- Yu G. Y., Zheng G. Z., Chang B., Hu Q. X., Lin F. X., Liu D. Z., et al. (2016). Naringin Stimulates Osteogenic Differentiation of Rat Bone Marrow Stromal Cells via Activation of the Notch Signaling Pathway. Stem Cell Int. 2016, 7130653. 10.1155/2016/7130653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu X., Shen G., Shang Q., Zhang Z., Zhao W., Zhang P., et al. (2021). A Naringin-Loaded Gelatin-Microsphere/nano-Hydroxyapatite/silk Fibroin Composite Scaffold Promoted Healing of Critical-Size vertebral Defects in Ovariectomised Rat. Int. J. Biol. Macromol 193, 510–518. 10.1016/j.ijbiomac.2021.10.036 [DOI] [PubMed] [Google Scholar]
- Yu Z., Fan L., Li J., Ge Z., Dang X., Wang K. (2016). Lithium Prevents Rat Steroid-Related Osteonecrosis of the Femoral Head by β-catenin Activation. Endocrine 52, 380–390. 10.1007/s12020-015-0747-y [DOI] [PubMed] [Google Scholar]
- Yuan Z., Min J., Zhao Y., Cheng Q., Wang K., Lin S., et al. (2018). Quercetin Rescued TNF-Alpha-Induced Impairments in Bone Marrow-Derived Mesenchymal Stem Cell Osteogenesis and Improved Osteoporosis in Rats. Am. J. Transl Res. 10, 4313–4321. [PMC free article] [PubMed] [Google Scholar]
- Zamora-Ros R., Knaze V., Rothwell J. A., Hémon B., Moskal A., Overvad K., et al. (2016). Dietary Polyphenol Intake in Europe: the European Prospective Investigation into Cancer and Nutrition (EPIC) Study. Eur. J. Nutr. 55, 1359–1375. 10.1007/s00394-015-0950-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zha X., Xu Z., Liu Y., Xu L., Huang H., Zhang J., et al. (2016). Amentoflavone Enhances Osteogenesis of Human Mesenchymal Stem Cells through JNK and P38 MAPK Pathways. J. Nat. Med. 70, 634–644. 10.1007/s11418-016-0993-1 [DOI] [PubMed] [Google Scholar]
- Zhang H., Li H. (2018). Tricin Enhances Osteoblastogenesis through the Regulation of Wnt/β-Catenin Signaling in Human Mesenchymal Stem Cells. Mech. Dev. 152, 38–43. 10.1016/j.mod.2018.07.001 [DOI] [PubMed] [Google Scholar]
- Zhang J., Wu K., Xu T., Wu J., Li P., Wang H., et al. (2019). Epigallocatechin-3-gallate Enhances the Osteoblastogenic Differentiation of Human Adipose-Derived Stem Cells. Drug Des. Devel Ther. 13, 1311–1321. 10.2147/DDDT.S192683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L., Li X., Ying T., Wang T., Fu F. (2021). The Use of Herbal Medicines for the Prevention of Glucocorticoid-Induced Osteoporosis. Front. Endocrinol. (Lausanne) 12, 744647. 10.3389/fendo.2021.744647 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L. Y., Xue H. G., Chen J. Y., Chai W., Ni M. (2016). Genistein Induces Adipogenic Differentiation in Human Bone Marrow Mesenchymal Stem Cells and Suppresses Their Osteogenic Potential by Upregulating PPARγ. Exp. Ther. Med. 11, 1853–1858. 10.3892/etm.2016.3120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang N. D., Han T., Huang B. K., Rahman K., Jiang Y. P., Xu H. T., et al. (2016). Traditional Chinese Medicine Formulas for the Treatment of Osteoporosis: Implication for Antiosteoporotic Drug Discovery. J. Ethnopharmacol 189, 61–80. 10.1016/j.jep.2016.05.025 [DOI] [PubMed] [Google Scholar]
- Zhang Q., Chang B., Zheng G., Du S., Li X. (2020). Quercetin Stimulates Osteogenic Differentiation of Bone Marrow Stromal Cells through miRNA-206/connexin 43 Pathway. Am. J. Transl Res. 12, 2062–2070. [PMC free article] [PubMed] [Google Scholar]
- Zhang Y. L., Zhu Z. Z., Zhang L. C., Wang G. (2021). Lithium Chloride Prevents Glucocorticoid-Induced Osteonecrosis of Femoral Heads and Strengthens Mesenchymal Stem Cell Activity in Rats. Chin. Med. J. (Engl) 134, 2214–2222. 10.1097/CM9.0000000000001530 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao J., Wu J., Xu B., Yuan Z., Leng Y., Min J., et al. (2019). Kaempferol Promotes Bone Formation in Part via the mTOR Signaling Pathway. Mol. Med. Rep. 20, 5197–5207. 10.3892/mmr.2019.10747 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Z. H., Ma X. L., Zhao B., Tian P., Ma J. X., Kang J. Y., et al. (2021). Naringin-inlaid Silk Fibroin/hydroxyapatite Scaffold Enhances Human Umbilical Cord-Derived Mesenchymal Stem Cell-Based Bone Regeneration. Cell Prolife 54, e13043. 10.1111/cpr.13043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng Y., Zhang R., Shi W., Li L., Liu H., Chen Z., et al. (2020). Metabolism and Pharmacological Activities of the Natural Health-Benefiting Compound Diosmin. Food Funct. 11, 8472–8492. 10.1039/d0fo01598a [DOI] [PubMed] [Google Scholar]
- Zhou Y., Wu Y., Ma W., Jiang X., Takemra A., Uemura M., et al. (2017). The Effect of Quercetin Delivery System on Osteogenesis and Angiogenesis under Osteoporotic Conditions. J. Mater. Chem. B 5, 612–625. 10.1039/c6tb02312f [DOI] [PubMed] [Google Scholar]
- Zhou Y. H., Xie Q. (2021). Total Glycosides from Eucommia Ulmoides Seed Promoted Osteogenic Differentiation of Adipose-Derived Mesenchymal Stem Cells and Bone Formation in Ovariectomized Rats through Regulating Notch Signaling Pathway. J. Orthop. Surg. Res. 16, 660. 10.1186/s13018-021-02797-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu J., Tang H., Zhang Z., Zhang Y., Qiu C., Zhang L., et al. (2017). Kaempferol Slows Intervertebral Disc Degeneration by Modifying LPS-Induced Osteogenesis/adipogenesis Imbalance and Inflammation Response in BMSCs. Int. Immunopharmacol 43, 236–242. 10.1016/j.intimp.2016.12.020 [DOI] [PubMed] [Google Scholar]