Abstract
Background
Delaying intubation in patients who fail high-flow nasal cannula (HFNC) may result in increased mortality. The ROX index has been validated to predict HFNC failure among pneumonia patients with acute hypoxemic respiratory failure (AHRF), but little information is available for non-pneumonia causes. In this study, we validate the ROX index among AHRF patients due to both pneumonia or non-pneumonia causes, focusing on early prediction.
Methods
This was a retrospective observational study in eight Singapore intensive care units from 1 January 2015 to 30 September 2017. All patients >18 years who were treated with HFNC for AHRF were eligible and recruited. Clinical parameters and arterial blood gas values at HFNC initiation and one hour were recorded. HFNC failure was defined as requiring intubation post-HFNC initiation.
Results
HFNC was used in 483 patients with 185 (38.3%) failing HFNC. Among pneumonia patients, the ROX index was most discriminatory in pneumonia patients one hour after HFNC initiation [AUC 0.71 (95% CI 0.64–0.79)], with a threshold value of <6.06 at one hour predicting HFNC failure (sensitivity 51%, specificity 80%, positive predictive value 61%, negative predictive value 73%). The discriminatory power remained moderate among pneumonia patients upon HFNC initiation [AUC 0.65 (95% CI 0.57–0.72)], non-pneumonia patients at HFNC initiation [AUC 0.62 (95% CI 0.55–0.69)] and one hour later [AUC 0.63 (95% CI 0.56–0.70)].
Conclusion
The ROX index demonstrated moderate discriminatory power among patients with either pneumonia or non-pneumonia-related AHRF at HFNC initiation and one hour later.
Introduction
High-flow nasal cannula therapy (HFNC) provides many benefits to critically ill patients, including reduced work of breathing, reliable oxygen delivery at higher concentrations and enhanced secretion clearance [1–3]. HFNC has gained popularity [4–6] since the landmark FLORALI study for acute hypoxemic respiratory failure (AHRF) among patients with pneumonia [7]. Many centres have broadened indications beyond pneumonia [8,9], using it in pulmonary hemorrhage [10], acute exacerbation of chronic obstructive pulmonary disease (COPD) [9,11–13] and heart failure [14]. Its use has extended beyond the intensive care unit (ICU) to the emergency department (ED), where the aetiology of the hypoxemic respiratory failure may not yet be ascertained [9,14].
Although HFNC has been used among AHRF patients with non-pneumonia indications, existing evidence for HFNC use has been mainly among pneumonia patients. The FLORALI study demonstrated lower intubation rates among pneumonia patients with PaO2:FiO2 ratio (PF ratio) less than 200 mmHg when compared to those receiving conventional oxygen therapy (COT) and non-invasive ventilation (NIV), although up to 40% still required intubation [7]. Prior studies among non-pneumonia patients focused mainly on physiological parameters [8–14], with the failure rate remaining largely unknown. Understanding HFNC failure rates is important, because mortality may be increased if intubation is delayed [15].
In this respect, Roca and colleagues developed the ROX index to help predict HFNC failure in patients with pneumonia [16,17]. The ROX index is the ratio of SpO2/FiO2 (SF ratio) to respiratory rate. It has excellent specificity (98–99%) for predicting HFNC failure at various time points of 2 (<2.85), 6 (<3.47) and 12 hours (<3.85), albeit at the expense of low sensitivity [17]. Physicians have subsequently attempted to improve the ROX index [18] and explore its utility as a predictor of weaning failure [19]. Current evidence supporting the use of the ROX index is limited to pneumonia patients mainly at later time points, limiting its utility.
Understanding the performance of the ROX index at earlier time points, would enhance prediction of HFNC failure, facilitating earlier intubation and potentially averting increased mortality risks. In this study, we aimed to expand the utility of the ROX index as a tool for predicting HFNC failure by validating its use at earlier time points among both pneumonia and non-pneumonia patients. We hypothesized that the ROX index could discriminate patients likely to experience HFNC failure at earlier time points in both pneumonia and non-pneumonia AHRF.
Methods
Study design
We conducted a retrospective observational multi-center study in all public hospital intensive care units (ICU) in Singapore of patients using HFNC for AHRF from 1 January 2015 to 30 September 2017. The National Healthcare Group Domain-Specific Review Board approved the study with a waiver of informed consent due to the non-interventional study design (DSRB 2017/00900).
Patients
Patients were included if they were older than 18 years and treated with HFNC for AHRF. Patients with concomitant hypercapnia were also included. Patients were excluded if they had do-not-intubate orders for the current AHRF episode or if HFNC was used for pre-oxygenation, post-extubation management or peri-procedural purposes. HFNC initiation was at the discretion of the primary physician.
Device description and management
HFNC was provided with one of the following devices: Optiflow device (MR850 heated humidified delivery tubing and nasal cannula; Fisher & Paykel Healthcare, Auckland, New Zealand)™, Bio-med device high flow air-oxygen blender with the heated humidifier (MR850, Fisher & Paykel Healthcare, Auckland, New Zealand)™ or the Airvo 2 device (Fisher & Paykel Healthcare, Auckland, New Zealand)™. Despite the different devices, all the nasal cannula interfaces were similar to the Airvo2 device, where the nasal cannula is able to deliver humidified respiratory gases up to flows of 70L/min. Physicians initiated HFNC at a minimum flow of 30L/min with FiO2 of at least 30% to target a SpO2 of at least 92%.
Data collection
Demographic characteristics were collected for all patients including: age, gender, ethnicity, admission source and comorbidities (diabetes, hypertension, ischemic heart disease, stroke, asthma, COPD, bronchiectasis, interstitial lung disease, liver cirrhosis and chronic kidney disease). Immunocompromised state was also determined, defined as the presence of any solid tumor, hematological malignancy, or usage of steroids (at least 0.3mg/kg/day for at least 1 month) [20] or other immunosuppressants. Illness severity was determined from the Acute Physiology and Chronic Health Evaluation (APACHE) II score and frequency of vasopressor use.
The underlying etiology of AHRF was categorized into one of the following: (a) pneumonia, defined as the presence of respiratory symptoms with radiological evidence on chest radiograph; (b) heart failure; (c) COPD exacerbation; (d) asthma exacerbation; (e) exacerbation of interstitial lung disease; (f) HFNC use post-surgery; (g) others. In cases where multiple etiologies may account for the patient’s AHRF, the leading etiology was determined by the primary physician.
HFNC failure was defined as requiring intubation after HFNC initiation. However, since this was a retrospective observational study, and none of the participating ICUs used a specific protocol to determine intubation decisions among the HFNC patients, the intubation decision occurred at the discretion of the managing physician. ICU mortality, hospital mortality, ICU length-of-stay and hospital length-of-stay were also collected.
As mentioned, the ROX index is the ratio of the SF ratio to respiratory rate. In order to compute the ROX index as defined by Roca and colleagues [17], the following parameters were collected immediately prior to HFNC initiation, after one hour and in the event of HFNC failure, at the point of intubation: SpO2 (%), FiO2 (%), respiratory rate (breaths/min), flow rate (L/min) and arterial partial pressure of carbon dioxide (PaCO2, in mmHg) [16,17].
Statistical analysis
Univariate comparisons of proportions, means and medians were performed using the Chi-square test, Student t, and Wilcoxon rank-sum tests respectively. Separate subgroup analyses were performed for both pneumonia and non-pneumonia populations. We assessed the discrimination of the ROX index using receiving operating characteristic (ROC) curves. Using parameters at HFNC initiation and one hour later, the Youden index method was used to determine the optimal cut-point of the ROX index for HFNC failure. Statistical significance was taken as a two-tailed P<0.05, and analyses were performed with Stata 15.0 (College Station, TX).
Results
Patient characteristics and outcomes
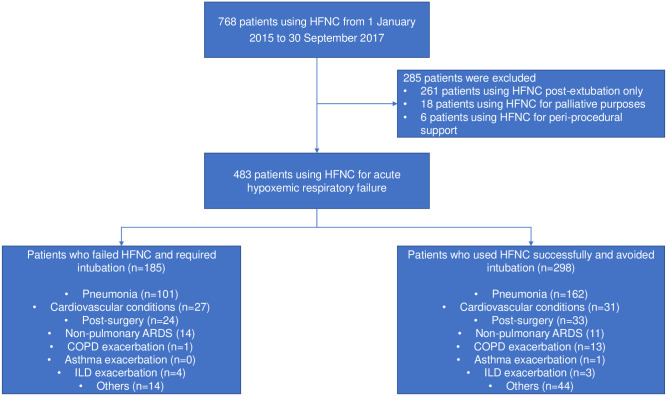
Eight ICUs participated in this study. 483 patients required HFNC for AHRF, of whom 45 patients also had concomitant hypercapnia. 263 patients (54%) had a primary diagnosis of pneumonia (Fig 1). The remaining patients who received HFNC had cardiovascular conditions (n = 58), utilized HFNC post-surgery (n = 57), and for non-pulmonary acute respiratory distress syndrome (n = 25), COPD (n = 14), interstitial lung disease (n = 7) or asthma exacerbations (n = 1). 185 patients (38.3%) failed HFNC. Baseline demographics were mostly similar between both patients with pneumonia and non-pneumonia conditions. Patients with non-pneumonia conditions were more likely to have ischemic heart disease and less likely to be admitted from the operating theatre (Table 1).
Fig 1. Enrolment and outcomes.
Table 1. Baseline characteristics of patients with hypoxemic respiratory failure.
| All patients (n = 483) | Patients with pneumonia (n = 263) | Patients with non-pneumonia conditions (n = 220) | p value | |
|---|---|---|---|---|
| Median Age, (Years) | 64 (55–74) | 65 (55–74) | 63 (56–73) | 0.689 |
| Male | 345 (71.4%) | 190 (72.2%) | 155 (70.5%) | 0.687 |
| Median BMI (kg/m2) | 22.9 (20.0–26.7) | 22.8 (19.6–26.0) | 22.9 (21.2–27.0) | 0.074 |
| Median APACHE II | 19 (13–25) | 19 (13–25) | 20 (13–25) | 0.633 |
| Vasopressor use | 94 (19.5%) | 43 (16.4%) | 51 (23.2%) | 0.065 |
| Smoking status | ||||
| Smoker | 86 (17.8%) | 40 (15.2%) | 46 (20.9%) | 0.271 |
| Ex-smoker | 335 (69.4%) | 188 (71.5%) | 147 (66.8%) | |
| Non-smoker | 62 (12.8%) | 35 (13.3%) | 27 (12.3%) | |
| Admission source | ||||
| Emergency Department | 207 (42.9%) | 83 (37.7%) | 124 (47.2%) | <0.001* |
| General Ward | 233 (48.2%) | 102 (46.4%) | 131 (49.8%) | |
| Operating Theatre | 43 (8.9%) | 35 (15.9%) | 8 (3.0%) | |
| Comorbidities | ||||
| Hypertension | 282 (58.4%) | 149 (56.65%) | 133 (60.5%) | 0.406 |
| Diabetes | 181 (37.5%) | 100 (38.0%) | 81 (36.8%) | 0.850 |
| Immunosuppression | 133 (27.5%) | 67 (25.5%) | 66 (30.0%) | 0.306 |
| Chronic kidney disease | 104 (21.5%) | 59 (22.4%) | 45 (20.5%) | 0.657 |
| Ischemic Heart Disease | 103 (21.3%) | 40 (15.2%) | 63 (28.6%) | 0.001* |
| COPD | 34 (7.0%) | 14 (5.3%) | 20 (9.1%) | 0.112 |
| Stroke | 32 (6.6%) | 17 (6.5%) | 15 (6.8%) | 1.000 |
| Asthma | 28 (5.8%) | 12 (4.6%) | 16 (7.3%) | 0.242 |
| Liver cirrhosis | 15 (3.1%) | 7 (2.7%) | 8 (3.6%) | 0.604 |
| Bronchiectasis | 11 (2.3%) | 8 (3.0%) | 3 (1.4%) | 0.359 |
| Interstitial lung disease | 9 (1.9%) | 4 (1.5%) | 5 (2.3%) | 0.738 |
| Outcomes | ||||
| Median duration of HFNC use (Hrs) | 23 (9–49) | 23 (10–53) | 21 (9–46) | 0.614 |
| Median ICU length of stay (days) | 6 (3–10) | 6 (3–11) | 6 (3–10) | 0.503 |
| Median hospital length of stay (days) | 18 (10–35) | 15 (9–29) | 21 (11–41) | 0.023* |
| ICU mortality | 71 (14.7%) | 47 (17.9%) | 24 (10.9%) | 0.039* |
| Hospital mortality | 120 (24.8%) | 78 (29.7%) | 42 (19.1%) | 0.008* |
* represents those parameters that are statistically significant.
** BMI = Body mass index, APACHE II = Acute Physiology and Chronic Health Evaluation II, ED = Emergency Department, COPD = Chronic obstructive pulmonary disease.
Performance of the ROX index among pneumonia patients only
Out of 263 patients with pneumonia, 101 (38.4%) required intubation. Among those who failed HFNC, they were more tachypneic [27 (23–33) breaths/min vs 24 (20–28) breaths/min, p = 0.006], tachycardic [106 (89–118) beats/min vs 95 (83–107) beats/min, p<0.001] and were initiated on HFNC at lower SF ratio [176 (97–243) vs 208 (115–257), p = 0.006] immediately prior to HFNC initiation. None of the other non-respiratory parameters were predictive of HFNC failure. These differences persisted one hour after HFNC initiation (Table 2). Those who failed HFNC already required higher FiO2 one hour of HFNC initiation [60% (50%-70%) vs 50% (40%-60%), p<0.001]. These results remained consistent when patients with concomitant hypercapnia were excluded (S1 Table).
Table 2. Parameters of patients with hypoxemic respiratory failure at various stages of HFNC use.
| Parameters | Patients with pneumonia only (n = 263) | Patients with non-pneumonia conditions only (n = 220) | ||||
|---|---|---|---|---|---|---|
| Patients who failed HFNC and required intubation (n = 101) | Patients who used HFNC successfully and avoided intubation (n = 162) | p value | Patients who failed HFNC and required intubation (n = 84) | Patients who used HFNC successfully and avoided intubation (n = 136) | p value | |
| Clinical and serological parameters (Median, Inter-quartile range) | ||||||
| Immediately prior to HFNC initiation | ||||||
| Pre-HFNC BIPAP use (%) | 18 (17.8) | 33 (20.5) | 0.634 | 20 (23.8) | 41 (30.2) | 0.354 |
| Pre-HFNC CPAP use (%) | 11 (10.9) | 26 (16.2) | 0.277 | 13 (15.5) | 23 (16.9) | 0.853 |
| Respiratory rate (breaths/min) | 27 (23–33) | 24 (20–28) | 0.006* | 25 (21–29) | 24 (19–29) | 0.417 |
| FiO2 (%) | 50 (40–100) | 50 (36–80) | 0.007* | 45 (36–50) | 40 (35–50) | 0.516 |
| SpO2 (%) | 95 (92–98) | 95 (92–98) | 0.553 | 95 (91–98) | 95 (91–97) | 0.775 |
| SF ratio | 176 (97–243) | 208 (115–257) | 0.006* | 200 (170–257) | 238 (185–277) | 0.035* |
| ROX index | 5.93 (3.93–8.97) | 8.18 (5.00–11.55) | <0.001* | 7.92 (6.06–10.78) | 10.02 (7.23–13.04) | 0.021* |
| PaCO2 (mmHg) | 33 (29–40) | 34 (30–37) | 0.356 | 35 (31–39) | 35 (29–39) | 0.426 |
| Serum HCO3 (mmol/l) | 22.6 (20.0–26.4) | 22.5 (19.3–26.0) | 0.150 | 22.5 (19.2–26.6) | 23.0 (20.2–26.2) | 0.064 |
| pH | 7.44 (7.41–7.48) | 7.43 (7.39–7.48) | 0.208 | 7.43 (7.37–7.48) | 7.41 (7.38–7.46) | 0.617 |
| Heart rate (bpm) | 106 (89–118) | 95 (83–107) | <0.001* | 96 (83–113) | 100 (81–114) | 0.960 |
| Systolic blood pressure (mmHg) | 136 (115–157) | 128 (111–153) | 0.256 | 121 (101–141) | 126 (107–145) | 0.351 |
| Diastolic blood pressure (mmHg) | 75 (63–85) | 72 (61–83) | 0.661 | 66 (52–78) | 70 (60–81) | 0.072 |
| Median GCS | 15 (15–15) | 15 (15–15) | 0.175 | 15 (14–15) | 15 (15–15) | 0.436 |
| 1hr after HFNC administered | ||||||
| Respiratory rate (breaths/min) | 26 (23–31) | 23 (19–30) | 0.004* | 24 (19–28) | 22 (18–27) | 0.338 |
| Flow (L/min) | 50 (50–60) | 50 (40–50) | 0.605 | 50 (45–60) | 50 (40–60) | 0.783 |
| FiO2 (%) | 60 (50–70) | 50 (40–60) | <0.001* | 50 (40–60) | 40 (35–50) | 0.001* |
| SpO2 (%) | 96 (93–98) | 96 (94–99) | 0.436 | 95 (93–97) | 96 (94–98) | 0.877 |
| SF ratio | 164 (134–194) | 190 (158–232) | <0.001* | 196 (165–240) | 240 (186–271) | 0.001* |
| ROX index | 6.74 (4.53–8.70) | 8.56 (6.74–11.19) | <0.001* | 8.03 (6.58–11.06) | 10.35 (7.48–14.00) | 0.001* |
| PaCO2 (mmHg) | 33 (29–38) | 34 (29–38) | 0.823 | 34 (30–39) | 33 (28–38) | 0.814 |
| Serum HCO3 (mmol/l) | 23.1 (20.0–27.0) | 23.0 (20.7–26.0) | 0.223 | 23.8 (20.3–26.0) | 22.0 (20.0–24.5) | 0.259 |
| pH | 7.45 (7.40–7.49) | 7.44 (7.40–7.47) | 0.616 | 7.44 (7.40–7.50) | 7.44 (7.40–7.47) | 0.440 |
| Heart rate (bpm) | 102 (86–118) | 92 (82–103) | <0.001* | 98 (83–112) | 96 (81–106) | 0.289 |
| Systolic blood pressure (mmHg) | 133 (116–150) | 129 (111–149) | 0.727 | 119 (107–137) | 120 (106–137) | 0.916 |
| Diastolic blood pressure (mmHg) | 69 (61–81) | 69 (60–79) | 0.809 | 68 (55–77) | 67 (59–77) | 0.564 |
| Median GCS | 15 (15–15) | 15 (15–15) | 0.682 | 15 (14–15) | 15 (15–15) | 0.389 |
* represents those parameters that are statistically significant.
The ROX index was more discriminatory among pneumonia patients one-hour post-initiation [AUC 0.71 (95% CI 0.64–0.79)] as compared to upon HFNC initiation [AUC 0.65 (0.57–0.72)]. A ROX index <4.58 at HFNC initiation and <6.06 one-hour post-initiation predicted HFNC failure with 80% specificity (Table 3 and S1 Fig).
Table 3. Comparison of cut-off points and ROC curves between ROX index at initiation and after 1 hour of HFNC.
| Index/ score | Cut-off point | Sensitivity at cut-off point | Specificity at cut-off point | Positive predictive value at cut-off point | Negative predictive value at cut-off point | Positive likelihood ratio at cut-off point | Negative likelihood ratio at cut-off point | AUC (95% CI) |
|---|---|---|---|---|---|---|---|---|
| All AHRF patients | ||||||||
| ROX | 6.93 (Youden) | 0.52 | 0.69 | 0.51 | 0.7 | 1.67 | 0.7 | 0.62 (0.57–0.68) |
| 5.18 | 0.31 | 0.8 | 0.49 | 0.65 | 1.55 | 0.86 | ||
| 3.84 | 0.16 | 0.91 | 0.52 | 0.64 | 1.78 | 0.92 | ||
| ROX at 1h | 7.96 (Youden) | 0.59 | 0.65 | 0.51 | 0.72 | 1.69 | 0.63 | 0.65 (0.60–0.71) |
| 6.46 | 0.37 | 0.8 | 0.53 | 0.67 | 1.85 | 0.79 | ||
| 4.94 | 0.22 | 0.91 | 0.6 | 0.66 | 2.44 | 0.86 | ||
| AHRF patients with pneumonia only | ||||||||
| ROX | 6.56 (Youden) | 0.59 | 0.67 | 0.52 | 0.73 | 1.79 | 0.61 | 0.65 (0.57–0.72) |
| 4.58 | 0.41 | 0.81 | 0.57 | 0.69 | 2.16 | 0.73 | ||
| 3.62 | 0.21 | 0.91 | 0.59 | 0.65 | 2.33 | 0.87 | ||
| ROX at 1h | 7.18 (Youden) | 0.65 | 0.68 | 0.55 | 0.76 | 2.03 | 0.51 | 0.71 (0.64–0.79) |
| 6.06 | 0.51 | 0.8 | 0.61 | 0.73 | 2.55 | 0.61 | ||
| 4.64 | 0.33 | 0.91 | 0.69 | 0.69 | 3.67 | 0.74 | ||
| AHRF patients not due to non-pneumonia conditions | ||||||||
| ROX | 7.59 (Youden) | 0.53 | 0.72 | 0.54 | 0.71 | 1.89 | 0.65 | 0.62 (0.55–0.69) |
| 6.24 | 0.33 | 0.80 | 0.51 | 0.66 | 1.65 | 0.84 | ||
| 4.36 | 0.14 | 0.90 | 0.46 | 0.63 | 1.40 | 0.96 | ||
| ROX at 1h | 11.54 (Youden) | 0.82 | 0.42 | 0.47 | 0.79 | 1.41 | 0.43 | 0.63 (0.56–0.70) |
| 6.89 | 0.34 | 0.80 | 0.51 | 0.66 | 1.79 | 0.83 | ||
| 5.44 | 0.13 | 0.91 | 0.45 | 0.63 | 1.44 | 0.96 | ||
AUC: Area under the receiver operating characteristic curve.
CI: Confidence interval.
Performance of the ROX index among non-pneumonia patients only
Among 220 patients with non-pneumonia AHRF, 84 (38.2%) failed HFNC. Patients who failed HFNC predominantly had lower median SF ratio compared to those who used HFNC successfully [200 (170–257) vs 238 (185–277), p = 0.035] immediately prior to HFNC initiation. Unlike pneumonia patients, patients who failed HFNC were not more tachypneic or tachycardic. One-hour after HFNC administration, these clinical characteristics still persisted. Again, higher FiO2 was already required among those who failed HFNC [50% (40%-60%) vs 40% (35%-50%)], p = 0.001] (Table 2). These results remained consistent when patients with concomitant hypercapnia were excluded (S1 Table).
The ROX index still showed moderate discrimination at HFNC initiation [AUC 0.62 (95% CI 0.55–0.69)] and one hour later [AUC 0.63 (95% CI 0.56–0.70)]. Correspondingly, a ROX index of <6.24 at HFNC initiation and <6.89 one-hour later predicted HFNC failure (Table 3 and S1 Fig).
Discussion
Among AHRF patients with pneumonia, the ROX index demonstrated moderate discriminatory power to predict HFNC failure, with greater discrimination one-hour post-initiation compared to when HFNC was initiated. In a similar fashion, the ROX index was also validated as moderately discriminating among non-pneumonia AHRF patients at similar time points.
Roca and colleagues originally validated the ROX index in patients with pneumonia [16,17]. However, at least 20% of all ICU patients with AHRF suffer from non-pneumonia-related conditions [21]. In some subgroups such as immunocompromised patients, the etiology of AHRF is even more varied [20,22–25], and in ED, HFNC may be initiated before the underlying etiology of AHRF is established [9]. Since delayed intubation can increase mortality regardless of the cause of AHRF [7], it is important to understand the performance of the ROX index among patients with non-pneumonia-related conditions. Currently, there are few studies investigating HFNC use among this population and they mainly focused on demonstrating physiological benefits [2,7,26–28]. In contrast, our study focused on understanding the performance of the ROX index among a more diverse HFNC population.
In addition, as the ROX index was originally validated at 2, 6 and 12 hours [16,17], we explored its ability to discriminate patients likely to fail HFNC at earlier time points. This would facilitate early pre-emptive intubation before further deterioration has occurred [15]. We demonstrated that at one-hour post-HFNC initiation, the AUROC of the ROX index for predicting HFNC failure was 0.71 and 0.63 for pneumonia and non-pneumonia patients respectively. Therefore, we have shown that the ROX index calculated immediately prior to HFNC initiation and one-hour post-initiation can help identify those likely to fail. Since HFNC is increasingly initiated while patients are still in ED, predicting HFNC failure at earlier time points can guide the development of practical workflows for close monitoring, especially in settings where staffing resources for close patient monitoring is scarce, or transfer time from ED to ICU is prolonged. Identifying patients most likely to fail HFNC may facilitate earlier intubation decisions and increase safety during patient transport [29].
It is important to highlight that the ROX index is a screening tool with only moderate discriminatory power. In the original study, Roca and colleagues report AUROC values at 2 hours of 0.679, 6 hours of 0.703 and 12 hours of 0.752 [17]. We report similar values in our study (Table 3). To maximize the screening tool potential of the ROX index, the values adopted favoured higher specificity and negative predictive values, so as to reduce false positives. To illustrate, a ROX index of <6.06 (sensitivity 51%, specificity 80%) among pneumonia patients one-hour post-HFNC initiation would have led to an additional 46 patients being intubated. Importantly, our study also highlights the importance of serial measurements of the ROX index given the dynamic evolution of the disease course among these patients. Depending on the study population, the exact ROX index value predicting HFNC failure may differ. This is illustrated with the studies by Roca and Goh separately depicting different values at the same time points at 2 (<2.85 vs <6.55), 6 (<3.47 vs <6.60) and 12 (<3.85 vs <6.55) hours [17,18].
Our study has several strengths. It is a practical real-world evaluation. Our study population was large, from multiple centers and included AHRF patients with multiple etiologies. This allowed us to validate the ROX index at earlier time points and among non-pneumonia patients. To date, this is the largest study population investigating HFNC failure among non-pneumonia patients. However, we acknowledge several limitations in our study. Firstly, it was conducted retrospectively, and in the absence of a standardized protocol, we were unable to study the ROX index at other time points, in particular at later time points, which could be a subject for future studies. Secondly, the lack of a standardized protocol for HFNC practice could have led to biases from individual physician practice patterns and thresholds for determining HFNC failure. Thirdly, as with all retrospective studies, some missing data is inevitable, with the majority related to arterial blood gas findings one-hour post-HFNC initiation. Other studies have already established that these parameters do not predict HFNC failure [16,18,19]. Otherwise, all other parameters were missing <10% of the total data, thus mitigating the bias risk (S1 Table). Fourthly, determination of HFNC failure was clinically-driven rather than protocolized, and some variation of HFNC failure thresholds existed across centers (S3 and S4 Tables). Importantly, our HFNC failure rate of 38.3% is also similar to that of the FLORALI study [7]. Finally, in the absence of randomization, we are unable to account for the effect of unmeasured variables that may affect the ROX index.
Conclusion
There is moderate discriminatory power of the ROX index to predict HFNC failure among both pneumonia and non-pneumonia patients with AHRF upon HFNC initiation and one-hour post-initiation.
Supporting information
(PPTX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(XLSX)
(XLSX)
Acknowledgments
Drs Andrew Li, Matthew Edward Cove, Jason Phua, Ser Hon Puah, Vicky Ng, Amit Kansal, Qiao Li Tan, Juliet Tolentino Sahagun, Juvel Taculod, Addy Yong-Hui Tan, Amartya Mukhopadhyay, Chee Kiang Tay, Kollengode Ramanathan, Yew Woon Chia FRCP11, Duu Wen Sewa, Meiying Chew, Sennen JW Lew, Shirley Goh, Shekhar Dhanvijay, Jonathan Jit-Ern Tan and Kay Choong See form the complete membership of the author group.
We would like to also acknowledge Dan Ong Weijun, Julipie Sumampong Manalansan, Michael Sharey Nocon Zamora and Michael Camba Vidanes for their efforts in the data collection.
Abbreviations
- ABG
Arterial Blood Gas
- AHRF
Acute hypoxemic respiratory failure
- COT
Conventional oxygen therapy
- COVID-19
Coronavirus disease 2019
- ED
Emergency Department
- GCS
Glasgow Coma Scale
- HFNC
High flow nasal cannula
- ICU
Intensive Care Unit
- NIV
Non-invasive ventilation
Data Availability
All relevant data are within the paper and its Supporting information files.
Funding Statement
Author KCS has received honoraria and travel support from Medtronic and GE Healthcare. MC acknowledges travel support and honorarium from Baxter and Medtronic. JP and KR have received travel support from Medtronic. No additional external funding was received for this study. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Goligher EC, Slutsky AS. Not Just Oxygen? Mechanisms of Benefit from High-Flow Nasal Cannula in Hypoxemic Respiratory Failure. Am J Respir Crit Care Med. 2017;195:1128–1130. doi: 10.1164/rccm.201701-0006ED [DOI] [PubMed] [Google Scholar]
- 2.Mauri T, Turrini C, Eronia N, et al. Physiologic Effects of High-Flow Nasal Cannula in Acute Hypoxemic Respiratory Failure. Am J Respir Crit Care Med. 2017;195:1207–1215. doi: 10.1164/rccm.201605-0916OC [DOI] [PubMed] [Google Scholar]
- 3.Ricard JD, Roca O, Lemiale V, et al. Use of nasal high flow oxygen during acute respiratory failure. Intensive Care Med 2020;46:2238–2247. doi: 10.1007/s00134-020-06228-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Messika J, Ben Ahmed K, Gaudry S, et al. Use of high-flow nasal cannula oxygen therapy in subjects with ARDS: a 1-year observational study. Respir Care 2015;60:162–169. doi: 10.4187/respcare.03423 [DOI] [PubMed] [Google Scholar]
- 5.Gaunt KA, Spilman SK, Halub ME, Jackson JA, Lamb KD, Sahr SM. High-flow nasal cannula in a mixed adult ICU. Respir Care 2015;60:1383–1389. doi: 10.4187/respcare.04016 [DOI] [PubMed] [Google Scholar]
- 6.Hyun Cho W, Ju Yeo H, Hoon Yoon S, et al. High-flow nasal cannula therapy for acute hypoxemic respiratory failure in adults: a retrospective analysis. Intern Med 2015;54:2307–2313. doi: 10.2169/internalmedicine.54.4266 [DOI] [PubMed] [Google Scholar]
- 7.Frat JP, Thille AW, Mercat A, et al. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med 2015;372:2185–2196. doi: 10.1056/NEJMoa1503326 [DOI] [PubMed] [Google Scholar]
- 8.Sotello D, Orellana-Barrios M, Rivas AM, Nugent K. High flow nasal cannulas for oxygenation: an audit of its use in a tertiary hospital. Am J Med Sci 2015;350:308–312. doi: 10.1097/MAJ.0000000000000557 [DOI] [PubMed] [Google Scholar]
- 9.Jones PG, Kamona S, Doran O, Sawtell F, Wilsher M. Randomized controlled trial of humidified high-flow nasal oxygen for acute respiratory distress in the emergency department: the HOT-ER study. Respir Care 2016;61:291–299. doi: 10.4187/respcare.04252 [DOI] [PubMed] [Google Scholar]
- 10.Schwabbauer N, Bjorn B, Blumenstock G, et al. Nasal high-flow oxygen therapy in patients with hypoxic respiratory failure: effect on functional and subjective respiratory parameters compared to conventional oxygen therapy and non-invasive ventilation. BMC Anesthesiology 2014;14:66. doi: 10.1186/1471-2253-14-66 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sztrymf B, Messika J, Bertrand F, et al. Beneficial effects of humidified high flow nasal oxygen in critical care patients: a prospective pilot study. Intensive Care Med 2011;37: 1780–1786. doi: 10.1007/s00134-011-2354-6 [DOI] [PubMed] [Google Scholar]
- 12.Braunlich J, Kholer M, Wirtz H. Nasal high flow improves ventilation in patients with COPD. Int J Chron Obstruct Pulmon Dis 2016;11:1077–1085. doi: 10.2147/COPD.S104616 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Fraser JF, Spoonser AJ, Dnuster KR, et al. Nasal high flow oxygen therapy in patients with COPD reduces respiratory rate and tissue carbon dioxide while increasing tidal and end-expiratory lung volumes: a randomised crossover trial. Thorax 2016;71:759–761. doi: 10.1136/thoraxjnl-2015-207962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Makdee O, Monsomboon A, Surabenjawong U, et al. High-flow nasal cannula versus conventional oxygen therapy in emergency department patients with cardiogenic pulmonary edema: a randomized controlled trial. Ann Emerg Med 2017;70:465–472. doi: 10.1016/j.annemergmed.2017.03.028 [DOI] [PubMed] [Google Scholar]
- 15.Kang BJ, Koh Y, Lim CM, et al. Failure of high-flow nasal cannula may delay intubation and increase mortality. Intensive Care Med 2015;41:623–632. doi: 10.1007/s00134-015-3693-5 [DOI] [PubMed] [Google Scholar]
- 16.Roca O, Messika J, Caralt B, et al. Predicting success of high-flow nasal cannula in pneumonia patients with hypoxemic respiratory failure: the utility of the ROX index. J Crit Care 2016;35:200–205. doi: 10.1016/j.jcrc.2016.05.022 [DOI] [PubMed] [Google Scholar]
- 17.Roca O, Caralt B, Messika J, et al. An index combining respiratory rate and oxygenation to predict outcome of nasal high flow therapy. Am J Respir Crit Care Med 2019;199:1368–1376. doi: 10.1164/rccm.201803-0589OC [DOI] [PubMed] [Google Scholar]
- 18.Goh KJ, Chai HZ, Ong TH, et al. Early prediction of high flow nasal cannula therapy outcomes using a modified ROX index incorporating heart rate. Journal of Intensive Care 2020;8:41. doi: 10.1186/s40560-020-00458-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rodriguez M, Thille AW, Boissier F, et al. Predictors of successful separation from high-flow nasal oxygen therapy in patients with acute respiratory failure: a retrospective monocentre study. Ann Intensive Care 2019;9:101. doi: 10.1186/s13613-019-0578-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Azoulay E, Lemiale V, Mokart D, et al. Effect of high-flow nasal oxygen vs standard oxygen on 28-day mortality in immunocompromised patients with acute respiratory failure—the HIGH randomized clinical trial. JAMA 2018;320:2099–2107. doi: 10.1001/jama.2018.14282 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Bellani G, Laffey JG, Pham T, et al. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA 2016;315:788–800. doi: 10.1001/jama.2016.0291 [DOI] [PubMed] [Google Scholar]
- 22.Lemiale V, Resche-Rigon M, Mokart D, et al. High-flow nasal cannula oxygenation in immunocompromised patients with acute hypoxemic respiratory failure: a Groupe de Recherche respiratoire en reanimation onco-hematologique study. Crit Care Med. 2017;45:e274–e280. doi: 10.1097/CCM.0000000000002085 [DOI] [PubMed] [Google Scholar]
- 23.Frat JP, Ragot S, Girault C, et al. Effect of non-invasive oxygenation strategies in immunocompromised patients with severe acute respiratory failure: a post-hoc analysis of a randomised trial. Lancet Respir Med. 2016;4:646–652. doi: 10.1016/S2213-2600(16)30093-5 [DOI] [PubMed] [Google Scholar]
- 24.Azoulay E, Mokart D, Kouatchet A, Demoule A, Lemiale V. Acute respiratory failure in immunocompromised adults. Lancet Respir Med 2019;7:173–186. doi: 10.1016/S2213-2600(18)30345-X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Azoulay E, Pickkers P, Soares M, et al. Acute hypoxemic respiratory failure in immunocompromised patients; the Efraim multinational prospective cohort study. Intensive Care Med 2017;43:1808–1819. doi: 10.1007/s00134-017-4947-1 [DOI] [PubMed] [Google Scholar]
- 26.Pilcher J, Eastlake L, Richards M, et al. Physiological effects of titrated oxygen via nasal high-flow cannulae in COPD exacerbations: a randomized controlled cross-over trial. Respirology 2017;22:1149–1155. doi: 10.1111/resp.13050 [DOI] [PubMed] [Google Scholar]
- 27.Braunlich J, Kohler M, Wirtz H. Nasal high-flow improves ventilation in patients with COPD. Int J COPD 2016;11:1077–1085. doi: 10.2147/COPD.S104616 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Braunlich J, Byer D, Mai D, et al. Effects of nasal high flow on ventilation in volunteers, COPD and idiopathic pulmonary fibrosis patients. Respiration 2013;85:319–325. doi: 10.1159/000342027 [DOI] [PubMed] [Google Scholar]
- 29.Liew MF, Siow WT, Yau YW, See KC. Safe patient transport for COVID-19. Crit Care 2020;24:94. doi: 10.1186/s13054-020-2828-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(PPTX)
(DOCX)
(DOCX)
(DOCX)
(DOCX)
(XLSX)
(XLSX)
Data Availability Statement
All relevant data are within the paper and its Supporting information files.